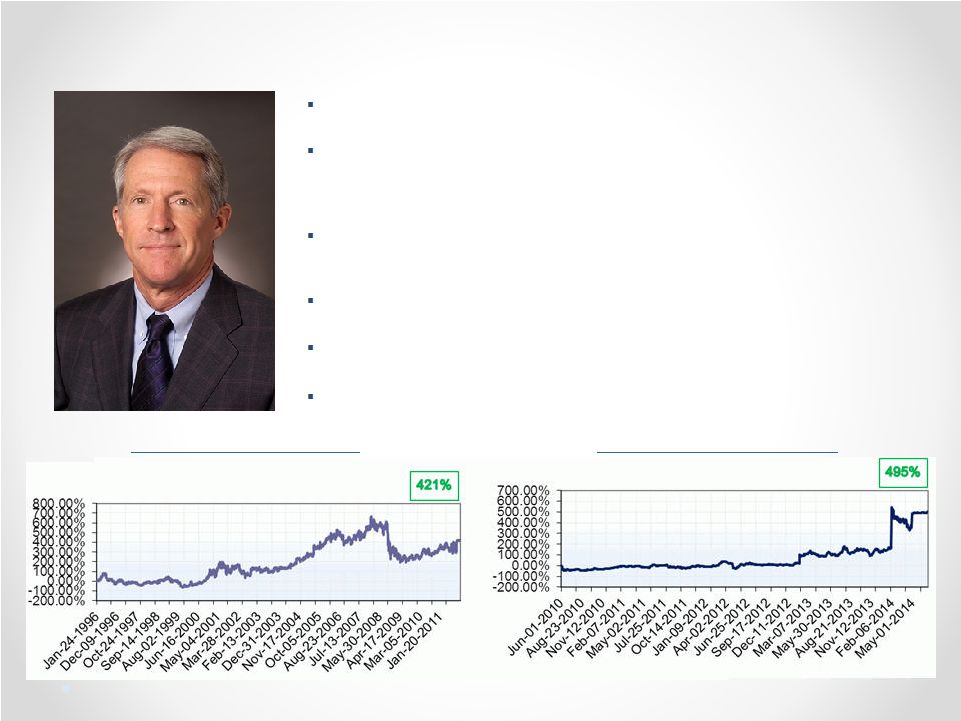
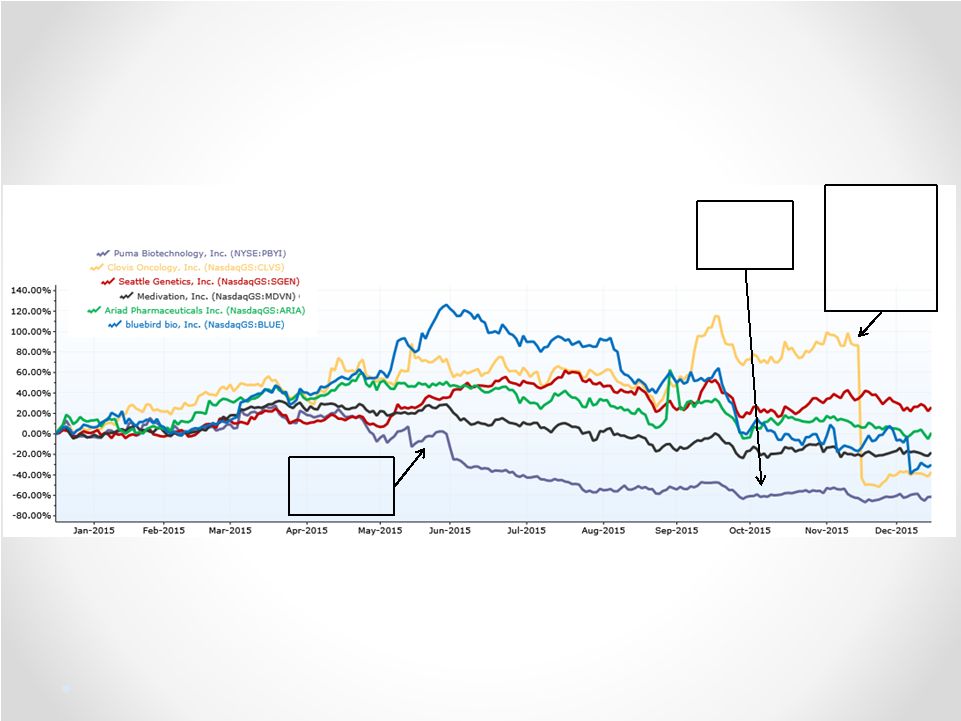
Optimal Board Size: Nine is Fine Of 31 peer companies identified by either ISS, Capital IQ, or Bloomberg: • Seven peers have boards with nine directors. • Notably, CEO Alan Auerbach sits on the board of Radius Health Inc., which has nine members. Radius Health’s market cap and product pipeline are similar to Puma’s. • 13 peer boards have nine or more members. • NONE of Puma’s peers has a board with fewer than six members. 42 ISS: “A board of between nine and 12 members is considered ideal.” Glass Lewis: “[F]ive directors is almost always a minimum for an effective and properly functioning board.” (2015 Puma Biotechnology Proxy Paper) Council of Institutional Investors Corporate Governance Policies: “[A] board should have no fewer than five and no more than 15 members.” Puma’s current board would have you believe that a five member board “is appropriate for effectively governing the Company” and that the board’s current size “provides for efficient decision-making.” Puma’s claims contradict best practices and industry norms: Best Practices Industry Norms Numerous companies, including Fortune 500 companies, have boards with nine directors: United Natural Foods Inc., Windstream Holdings, Inc., Dr. Pepper Snapple Group, Inc., Lennar Corporation, Laboratory Corp of America Holdings, Cliffs Natural Resources, PulteGroup Inc, Graybar Electric Company Inc. Wynn Resorts, Limited, St. Jude Medical, Inc., Asbury Automotive Group, Big Lots, Inc., Tractor Supply Company, Insight Enterprises Inc., Quintiles Transnational Holdings Inc., Joy Global Inc., Lorillard, Inc., Sanmina, First American Financial Corporation, Avery Dennison Corporation, Allergan Inc., Omnicare Inc., Dick’s Sporting Goods, NCR Corporation, Waste Management, Inc., PPG Industries Inc., Marathon Oil Corporation, Casey’s General Stores, Inc., Agilent Technologies Inc., Symantec Corporation, HollyFrontier Corporation, PBF Energy Inc., Kohl’s Corporation, AbbVie Inc, Vertex Pharmaceuticals Incorporated, Clovis Oncology, Inc., Tesoro Corporation, ARIAD Pharmaceuticals, Inc., Dynavax Technologies Corporation, Lexicon Pharmaceuticals, Inc., Seattle Genetics, Inc., Tesaro, Inc., World Fuel Services Corporation, INTL FCStone Inc., Best Buy Co. Inc, Reliance Steel & Aluminum Co., Stryker Corporation, Cognizant Technology Solutions, Ball Corporation, Broadcom Corporation, CenterPoint Energy, Franklin Resources, Inc., Oaktree Capital Group LLC, Jarden Corporation, Mohawk Industries, Inc., UGI Corporation, The Pantry, Inc., Tyson Foods Inc., Jabil Circuit, Inc., AutoNation Inc., Lear Corporation, Automatic Data Processing Inc., Liberty Interactive Corporation, Ameriprise Financial, Inc., Centene Corporation, Huntsman Corporation, Devon Energy Corporation, Publix Super Markets, Inc., Tech Data Corporation, RiteAid Corporation, National Oilwell Varco, Inc., Xerox Corporation, Arrow Electronics, Gentherm Inc., Cree, Inc Belden Inc., Eastgroup Properties Inc., La-Z-Boy Inc., Amphenol Corporation, CVR Energy Corp, International Bancshares Corp. 1-800-Flowers.com Inc. |