UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
Filed by the Registrantx
Filed by a Party other than the Registrant¨
Check the appropriate box:
| ¨ | Preliminary Proxy Statement |
| ¨ | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) |
| x | Definitive Proxy Statement |
| ¨ | Definitive Additional Materials |
| ¨ | Soliciting Material Pursuant to Rule 14a-12 |
Inspire Pharmaceuticals, Inc.
(Name of Registrant as Specified In Its Charter)
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
Payment of Filing Fee (Check the appropriate box):
| x | No fee required. |
| ¨ | Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11. |
| 1) | Title of each class of securities to which transaction applies: |
| 2) | Aggregate number of securities to which transaction applies: |
| 3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): |
| 4) | Proposed maximum aggregate value of transaction: |
| 5) | Total fee paid: |
| ¨ | Fee paid previously with preliminary materials. |
| ¨ | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. |
| 1) | Amount Previously Paid: |
| 2) | Form, Schedule or Registration Statement No.: |
| 3) | Filing Party: |
| 4) | Date Filed: |

April 17, 2003
To Our Stockholders:
You are most cordially invited to attend the 2003 Annual Meeting of Stockholders of Inspire Pharmaceuticals, Inc. at 9:00 a.m., local time, on Monday, June 9, 2003, at the Wentworth Mansion, 149 Wentworth Street, Charleston, South Carolina 29401. Please contact the company by telephone at (919) 941-9777, extension 220, for more specific details regarding the venue for the meeting.
The enclosed Notice of Annual Meeting of Stockholders and proxy statement on the following pages describe the matters to be presented at the Annual Meeting.
It is important that your shares be represented at the Annual Meeting to assure the presence of a quorum. Whether or not you plan to attend the Annual Meeting, we hope that you will have your stock represented by signing, dating and returning your proxy in the enclosed envelope, which requires no postage if mailed in the United States,as soon as possible. Your stock will be voted in accordance with the instructions you have given in your proxy.
Thank you for your continued support.
Sincerely,
Christy L. Shaffer, Ph.D.
Chief Executive Officer
INSPIRE PHARMACEUTICALS, INC.
4222 Emperor Boulevard, Suite 470
Durham, North Carolina 27703-8466
NOTICE OF ANNUAL MEETING OF STOCKHOLDERS
To Be Held On June 9, 2003
Dear Stockholders:
The Annual Meeting of the Stockholders of Inspire Pharmaceuticals, Inc. will be held on June 9, 2003, at 9:00 a.m., at the Wentworth Mansion, 149 Wentworth Street, Charleston, South Carolina 29401, to:
| (1) | Elect three directors to serve until the Annual Meeting of the Stockholders to be held in 2006 and until their successors have been duly elected and qualified; |
| (2) | Ratify the selection of PricewaterhouseCoopers LLP as our independent auditors for the fiscal year ending December 31, 2003; and |
| (3) | Transact such other business as may properly come before the Annual Meeting or any adjournments thereof. |
Only stockholders of record at the close of business on April 10, 2003 are entitled to notice of, and to vote at, the Annual Meeting, or any adjournment or adjournments thereof. A complete list of such stockholders will be open to the examination of any stockholder during ordinary business hours at our executive offices at 4222 Emperor Boulevard, Suite 470, Durham, North Carolina 27703-8466 for a period of 10 days prior to the Annual Meeting and a copy shall be available for examination at the time and place of the Annual Meeting. The Annual Meeting may be adjourned from time to time without notice other than by announcement to such effect at the Annual Meeting.
It is important that your shares be represented at the Annual Meeting. Your shares cannot be voted unless they are represented by proxy or you make other arrangements to have them represented at the Annual Meeting. Whether or not you plan to attend the Annual Meeting, please fill in, date and sign the enclosed proxy and return it promptly in the enclosed envelope. The prompt return of proxies will ensure a quorum and save us the expense of further solicitation.
By Order of the Board of Directors,
Gregory J. Mossinghoff
President, Secretary and Treasurer
Durham, North Carolina
April 17, 2003
INSPIRE PHARMACEUTICALS, INC.
4222 Emperor Boulevard, Suite 470
Durham, North Carolina 27703-8466
PROXY STATEMENT
General
These proxy materials are furnished in connection with the solicitation of proxies by the Board of Directors of Inspire Pharmaceuticals, Inc., a Delaware corporation, for use at our 2003 Annual Meeting of Stockholders (the “Annual Meeting”), and at any continuation or adjournment thereof. The Annual Meeting will be held June 9, 2003 at the Wentworth Mansion, 149 Wentworth Street, Charleston, South Carolina 29401 at 9:00 a.m.
We maintain principal executive offices at 4222 Emperor Boulevard, Suite 470, Durham, North Carolina 27703-8466. This proxy statement and the accompanying form of proxy are being mailed to stockholders on or about April 17, 2003. Our Annual Report for 2002, including financial statements for the year ended December 31, 2002, is being mailed to stockholders at the same time.
Stockholders Entitled To Vote
Holders of shares of our common stock of record at the close of business on April 10, 2003 are entitled to notice of, and to vote at, the Annual Meeting and at any and all adjournments or postponements of the Annual Meeting. Each share entitles its owner to one vote. The holders of a majority of the shares entitled to vote at the Annual Meeting must be present in person or represented by proxy in order to constitute a quorum for all matters to come before the Annual Meeting. On the record date there were 31,688,893 shares outstanding. Our Amended and Restated Certificate of Incorporation, as amended, does not provide for cumulative voting.
Other than the election of directors, which requires a plurality of the votes cast, each matter to be submitted to the stockholders requires the affirmative vote of a majority of the votes cast at the meeting. For purposes of determining the number of votes cast with respect to a particular matter, only those cast “For” or “Against” are included. Abstentions and broker non-votes are counted only for purposes of determining whether a quorum is present at the meeting.
How To Vote
If you are a stockholder of record (i.e. a stockholder who holds shares in one’s own name), you can vote by signing, dating and returning your proxy card in the enclosed postage-paid envelope. If you sign and return your proxy card but do not give voting instructions, the shares represented by that proxy will be voted as recommended by the Board of Directors.
If your shares are held in the name of a bank, broker or other holder of record (that is, “street name”), you will receive instructions from the holder of record that you must follow in order for your shares to be voted.
Changing Your Vote
You may change your vote at any time before the proxy is exercised. If you voted by mail, you may revoke your proxy at any time before it is voted by executing and delivering a timely and valid later-dated proxy, by voting by ballot at the meeting or by giving written notice to the Secretary of Inspire. Attendance at the Annual Meeting will not have the effect of revoking a proxy unless you give proper written notice of revocation to the Secretary before the proxy is exercised or you vote by written ballot at the Annual Meeting.
1
Reduce Duplicate Mailings
We are required to provide an Annual Report to all stockholders who receive this proxy statement. If you are a stockholder of record and have more than one account in your name or at the same address as other stockholders of record, you may authorize us to discontinue mailings of multiple proxy statements, Annual Reports and other information statements. To do so, mark the designated box on each proxy card for which you wish to discontinue to receive duplicate documents. Your consent to cease delivery of the Annual Report, proxy statements and other information statements shall be effective for five (5) years or until you revoke your consent. You may revoke your consent at any time by contacting Gregory J. Mossinghoff, in writing, at our offices located at 4222 Emperor Boulevard, Suite 470, Durham, North Carolina 27703-8466, or by calling (919) 941-9777. Delivery of individual copies of the documents shall resume within 30 days of receipt of your request.
Stockholder Proposals
To be included in the Board of Directors’ proxy statement and proxy card for the 2004 Annual Meeting of Stockholders, a stockholder proposal must be received by us on or before December 19, 2003. Proposals should be directed to the attention of Gregory J. Mossinghoff at our offices located at 4222 Emperor Boulevard, Suite 470, Durham, North Carolina 27703-8466.
In addition, if a stockholder wishes to present a proposal at our 2004 Annual Meeting of Stockholders which is not intended to be included in the proxy statement for that meeting, we must receive written notice of the stockholder proposal by March 3, 2004. If we do not receive timely notice of such a stockholder proposal, we will retain discretionary authority to vote proxies on such proposals even if it is not specifically reflected on the proxy card and stockholders have not had an opportunity to vote on the proposal by proxy.
PROPOSAL NO. 1
ELECTION OF DIRECTORS
At the Annual Meeting three directors are to be duly elected to hold office until the Annual Meeting of Stockholders to be held in 2006 and until their successors shall have been elected and qualified. The nominees for election to the Board of Directors are W. Leigh Thompson, M.D., Ph.D., D.Sc., Gary D. Novack, Ph.D. and William R. Ringo, Jr.
Pursuant to our Amended and Restated Certificate of Incorporation, as amended, the Board of Directors is divided into three classes, each class consisting, as nearly as possible, of an equal number of directors. There are currently seven members of the Board of Directors. Class A consists of Dr. Shaffer and Mr. Mossinghoff, whose terms will expire at the Annual Meeting of Stockholders in 2004. Class B consists of Kip Frey and Jesse I. Treu, Ph.D., whose terms will expire at the Annual Meeting of Stockholders in 2005. The Corporate Governance Committee of the Board of Directors has nominated, and the Board of Directors has ratified, the nomination of W. Leigh Thompson, M.D., Ph.D., D.Sc., Gary D. Novack, Ph.D. and William R. Ringo, Jr., for election at the Annual Meeting as Class C directors, whose terms will expire at the Annual Meeting of Stockholders in 2006. The Corporate Governance Committee does not currently consider nominees recommended by stockholders. Richard Boucher, M.D., a current Class C director, has informed the Board of Directors that he does not intend to stand for re-election at the Annual Meeting but will instead accept a one-year non-voting position as an Emeritus Board Observer to provide us with scientific expertise. In addition, Dr. Boucher continues to serve as Co-Chairman of our Scientific Advisory Board.
2
Set forth below is certain information about the members of our Board of Directors and the nominees, including their name, age and current positions:
Name | Age | Position | ||
Christy L. Shaffer, Ph.D. | 45 | Chief Executive Officer and Director | ||
Gregory J. Mossinghoff | 42 | President, Secretary, Treasurer and Director | ||
W. Leigh Thompson, M.D., Ph.D., D.Sc. | 64 | Chairman of the Board, Director and Nominee | ||
Richard Boucher, M.D. | 58 | Director | ||
Kip Frey | 44 | Director | ||
Gary D. Novack, Ph.D. . | 49 | Director and Nominee | ||
William Ringo, Jr. . | 57 | Nominee | ||
Jesse I. Treu, Ph.D. | 56 | Director |
If the enclosed proxy is properly executed and received in time for the Annual Meeting, it is the intention of the persons named in the proxy to vote the shares represented thereby for the persons nominated for election as directors unless authority to vote shall have been withheld. If any nominee should refuse or be unable to serve, an event not anticipated, the proxy will be voted for such person as shall be designated by the Board of Directors to replace such nominee or, in lieu thereof, the Board of Directors may reduce the number of directors. The Board of Directors has no reason to believe that the nominees will be unable to serve if elected. The nominees have consented to being named in this proxy statement and to serve if elected.
Set forth below is biographical information for each member of the Board of Directors and each nominee for election to the Board of Directors, including a summary of their background and principal occupations. If elected, each nominee would serve until the Annual Meeting of Stockholders in 2006 and until his or her successor is duly elected and qualified, or until such director’s earlier death, resignation, or removal.
Christy L. Shaffer, Ph.D., has served as our Chief Executive Officer and as a director since January 1999 and previously served as our President, as well as CEO, from January 1999 through June 2002, at which time she proposed that Mr. Mossinghoff assume the role of President. Dr. Shaffer is also a member of our Corporate Governance Committee. Dr. Shaffer joined us in June 1995 as our first full-time employee, Director, Clinical Operations. She was promoted to Senior Director, Development in June 1996 and to Vice President, Development and Chief Operating Officer in January 1998. Dr. Shaffer has fifteen years of experience in drug development within the pharmaceutical industry. She previously served in a variety of positions in the clinical research division of Burroughs Wellcome Co. including International Project Leader for cardiopulmonary programs. She served as the Associate Director of pulmonary research in the department of pulmonary/critical care medicine at Burroughs Wellcome immediately prior to joining Inspire in June 1995. Dr. Shaffer coordinated several IND submissions and one NDA submission at Burroughs Wellcome. Dr. Shaffer received a Ph.D. in pharmacology from the University of Tennessee and completed two years of postdoctoral training in cardiovascular research in the Biochemistry Department at the Chicago Medical School before her one-year postdoctoral appointment at University of North Carolina.
Gregory J. Mossinghoff has served as our President since June 2002 and a director since June 2001. Mr. Mossinghoff joined us in June 1998 as our Senior Director of Strategic Planning and Operations and was promoted to Vice President, Corporate Development in January 1999; Chief Business Officer in December 1999; and Senior Vice President in November 2000. Mr. Mossinghoff has also served as our Secretary since October 1998 and as our Treasurer since March 2000. In his current role he helps us develop and realize strategic objectives, expand our corporate partnerships in the United States and abroad, and oversees all business-related activities including operations and finance. Before joining us, from February 1996 to June 1998, Mr. Mossinghoff was worldwide Director of Business Analysis at Glaxo Wellcome plc. Before joining Glaxo Wellcome, Mr. Mossinghoff held various roles with increasing responsibility at Hoffmann LaRoche Inc., from June 1988 to February 1996, including Manager, Business Development and Strategic Planning from 1994 to 1996. Mr. Mossinghoff received a BA degree in Economics from the University of Virginia, Charlottesville, VA and an MBA in Financial Management & Analysis from George Mason University, Fairfax, VA.
3
W. Leigh Thompson, M.D., Ph.D., D.Sc., has served as a director since April 1996, is a member of our Corporate Governance Committee, the chairman of our Compensation Committee and has served as our Chairman of the Board since June 2002. In December 1994, Dr. Thompson retired from Eli Lilly and Co. where he served as Chief Scientific Officer and a member of the management committee. Dr. Thompson has enjoyed a distinguished career in both academic medicine and the pharmaceutical industry and has published extensively, particularly in the area of critical care medicine. He is a member of numerous corporate, academic, and civic boards, and consults in the areas of health informatics, enterprise strategic planning, and related areas and has been inducted into the Johns Hopkins Society of Scholars. Since 1995, Dr. Thompson has been the Chief Executive Officer of Profound Quality Resources, Ltd., a worldwide scientific consulting firm. He is currently a director of Bioanalytical Systems Inc., DepoMed Inc., Guilford Pharmaceuticals, Inc., Medarex Inc., Diabetogen Biosciences, Inc., La Jolla Pharmaceutical Company and Sontra Medical Corporation.
Richard Boucher, M.D., has served as a director since March 1995, and is a Co-Chairman of our Scientific Advisory Board and a member of our Corporate Governance Committee. Dr. Boucher has informed the Board of Directors that he does not intend to stand for re-election at the Annual Meeting. Dr. Boucher has been appointed as an Emeritus Board Observer effective on the date of the Annual Meeting and will serve in that capacity until the date of the 2004 Annual Meeting. Dr. Boucher is the William Rand Kenan Professor of Medicine, Chief of Pulmonary Medicine and Director of the Cystic Fibrosis/Pulmonary Research and Treatment Center at The University of North Carolina at Chapel Hill School of Medicine. Dr. Boucher obtained his M.D. degree from Columbia University College of Physicians and Surgeons. Following residency training, he joined the Faculty of Medicine at The University of North Carolina at Chapel Hill in 1977. Dr. Boucher has authored or co-authored more than 200 original research articles and more than 100 additional publications including book chapters. He received the Doris Tulcin and Paul Di Sant’Agnese CF Research Awards and the Julius Comroe Award from The American Physiology Society. He is an established principal investigator with the National Institutes of Health, and is a member of the American College of Physicians and the Association of American Physicians. In recent years, Dr. Boucher has pioneered new approaches for the treatment of cystic fibrosis.
Kip Frey has served as a director since June 2002, is a member of our Audit Committee, Compensation Committee and the Chairman of our Corporate Governance Committee. He is currently a Professor of the Practice in Entrepreneurial Management & Law at Duke University with joint appointments from the Fuqua School of Business and Duke Law School. He served as 2001-2002 Chairman of the Council for Entrepreneurial Development in Research Triangle Park, NC. He is the former President and Chief Executive Officer of OpenSite, a software company acquired by Siebel Systems in May 2000. Over the past five years, Mr. Frey has been the leader and architect of three of RTP’s most notable entrepreneurial ventures, Ventana Communications Group, Accipiter, Inc. and OpenSite. He is a member of the Board of Directors of the Raleigh Chamber of Commerce, the Council for Entrepreneurial Development, Duke Law School’s Board of Visitors, and several local civic and charitable organizations. He is also a Venture Partner at Intersouth Partners, a North Carolina venture capital firm. In 1999, Digital South Magazine named him the Southeast’s top CEO. Mr. Frey is a Phi Beta Kappa graduate of the University of Southern California Film School and of Duke Law School.
Gary D. Novack, Ph.D., joined the board as a director in January 2003 and is a member of our Audit Committee. Dr. Novack is the founder and President of PharmaŸLogic Development, Inc., a pharmaceutical and medical device development consulting firm that he started in 1989. He has worked with Inspire as a consultant, providing important strategic input to our pre-clinical and clinical ophthalmology programs. Dr. Novack is a board-certified clinical pharmacologist and has over 23 years experience in pharmaceutical research and development. He has consulted with over 150 pharmaceutical and medical device firms, and has authored over 220 abstracts and publications in pharmacology, ophthalmology, neurology, dermatology and medical communications. He serves on the editorial board of The Ocular Surface, and is a member of the scientific advisory board of the Foundation Fighting Blindness. He received his bachelor’s degree in Biology from the University of California, Davis, his Ph.D. in Pharmacology and Toxicology from the University of California at Davis, and was an NIH postdoctoral trainee in neurophysiology at the University of California, Los Angeles. As of July 1, 2003, Dr. Novack will join the Board of Regents of the University of California. He has held managerial positions at Allergan and Merrell Dow Pharmaceuticals and has served on several boards of not-for-profit organizations.
4
William R. Ringo, Jr., has been nominated to serve as a member of our Board of Directors. Mr. Ringo is a health care consultant with Barnard Life Sciences, which he founded in January 2001. From 1973 until his retirement in March 2001, Mr. Ringo was employed at Eli Lilly and Company, a global pharmaceutical company where he held a variety of positions, most recently serving as the President of Oncology and Critical Care Products from 1999 to March 2001, as the President of Internal Medicine Products from 1998 to March 2001 and as a member of Eli Lilly’s Corporate Operations Committee from 1995 to March 2001. From 1995 to 1997, Mr. Ringo served as the President of Eli Lilly’s Infectious Disease Business Unit. Prior to that time, Mr. Ringo held senior positions in the Sales and Marketing and Business Planning groups. Mr. Ringo also serves as a director of La Jolla Pharmaceutical Company, Praecis Pharmaceuticals Incorporated, Intermune, Inc. and Texas Biotechnology Corporation. In addition, Mr. Ringo is a Trustee of the Heart Hospital of Indiana and a member of the Burrill & Company Biotech Advisory Board.
Jesse I. Treu, Ph.D., has served as a director since March 1995, and is a member of our Compensation Committee and the Chairman of our Audit Committee. Dr. Treu has been a General Partner and Managing Member of Domain Associates, L.L.C. since its inception eighteen years ago. He has been a Director of over twenty early stage health care companies, thirteen of which have so far become successful public companies. These include Biosite Incorporated, GelTex Pharmaceuticals, Inc., OraPharma, Inc., Sepracor Inc. and Trimeris, Inc. He has served as a Founder, President and Chairman of numerous venture stage companies. Prior to the formation of Domain, Dr. Treu had twelve years health care industry experience. He was CEO of MicroSonics, Inc. and at GE and Technicon Corporation served in a number of research, management and corporate staff positions. Dr. Treu received his B.S. from Rensselaer Polytechnic Institute, and from Princeton University his M.A. and Ph.D. degrees in physics.
THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” EACH NOMINEE.
DIRECTOR COMPENSATION
In March 2000, we granted each non-employee director an option to purchase 5,714 shares of our common stock at an exercise price equal to the initial public offering price of the common stock, which was $12.00 per share. These options vested as to one-third of the option on the one year anniversary of the grant date and monthly thereafter. In June 2002, we granted each director an option to purchase 30,000 shares of our common stock and we granted members of our Audit Committee and our Chairman an option to purchase an additional 10,000 shares of our common stock. All of the June 2002 options have an exercise price of $2.76 per share and vest as to one-third of the shares on the dates of our Annual Meetings of Stockholders in 2003, 2004 and 2005. In March 2003, we granted Dr. Novack an option to purchase 6,667 shares of our common stock. Dr. Novack’s option has an exercise price of $14.72 per share and will vest on the date of the Annual Meeting. All of the options described above will cease to vest if the director no longer serves on our Board.
In March 2003, we implemented a policy for the compensation of non-employee directors, pursuant to which we will provide both cash and stock option compensation to such directors for their services. Each non-employee director who is not a member of the Audit Committee will receive annual cash compensation of $15,000 and each director who is a member of the Audit Committee will receive annual cash compensation of $25,000. In addition, non-employee directors will receive $2,500 per quarterly Board meeting attended in-person. In the future, following the full vesting of any existing option for service on the Board, each non-employee director who is not a member of the Audit Committee will receive a stock option to purchase 30,000 shares of common stock upon election or re-election to the Board and each director who is a member of the Audit Committee will receive an option to purchase 40,000 shares of common stock upon their election or re-election to the Board. The stock options will be issued pursuant to our Amended and Restated 1995 Stock Plan, as amended. The stock options will vest over a director’s three year term in office, assuming continued service as a director during such term. The compensation is intended to cover the general availability of directors between quarterly meetings and the directors’ participation in meetings. Directors are also reimbursed for expenses incurred in connection with attendance at Board and committee meetings.
5
MEETINGS AND COMMITTEES OF THE BOARD
During the year ended December 31, 2002, there were nine (9) meetings of the Board of Directors. Each incumbent director attended at least 75% of the meetings of the Board and its Committees. The Board has established an Audit Committee, Corporate Governance Committee and a Compensation Committee.
The members of the Audit Committee are Dr. Treu, as chairperson, Dr. Novack and Mr. Frey. Drs. Treu and Novack and Mr. Frey are “independent” as defined by the rules regarding the listing standards of the NASD. Pursuant to guidelines established by a charter adopted by the Board, the Audit Committee assists the Board of Directors in fulfilling its responsibilities of ensuring that management is maintaining an adequate system of internal controls such that there is reasonable assurance that assets are safeguarded and that financial reports are properly prepared; that there is consistent application of generally accepted accounting principles; and that there is compliance with management’s policies and procedures. In March 2003, the Board adopted an amended charter, which is attached to this proxy statement as an appendix. In performing these functions, the Audit Committee meets periodically with the independent auditors and management to review their work and confirm that they are properly discharging their respective responsibilities. In addition, the Audit Committee recommends the independent auditors for appointment by the Board of Directors. The Audit Committee met five (5) times during 2002.
The Corporate Governance Committee, which was formerly known as the Nominating Committee, consists of Mr. Frey, as chairperson, and Drs. Boucher, Thompson and Shaffer. This committee identifies individuals qualified to become members of the Board, reviews the qualifications of candidates, selects the director nominees for each annual meeting of stockholders, develops and recommends to the Board corporate governance principles, plans for and assists in the transitioning of directors on to and off of the Board, provides a review function for members of the Board, helps prepare for management succession, and leads the Board in complying with its corporate governance principles. The Corporate Governance Committee met two (2) during 2002.
The Compensation Committee currently consists of Dr. Thompson, as chairperson, and Dr. Treu and Mr. Frey. The Compensation Committee considers executive compensation of our key officers and compensation of directors. The Committee considers and reviews and approves issues and matters concerning the compensation of employees and consultants and the objectives and policies instituted by the Board of Directors. The Compensation Committee met three (3) times in 2002.
AUDIT AND OTHER FEES
The independent auditor of Inspire during the fiscal year ended December 31, 2002 was PricewaterhouseCoopers LLP. The fees billed to us by PricewaterhouseCoopers LLP for the fiscal year ended December 31, 2002 were as follows:
Audit Fees
The aggregate fees billed by PricewaterhouseCoopers LLP in connection with their review of the financial statements contained in our quarterly reports on Form 10-Q for the quarters ended March 31, June 30 and September 30, 2002, and the audit of the financial statements for the fiscal year ended December 31, 2002 included in our Annual Report on Form 10-K were $72,865.
Financial Information Systems Design and Implementation
PricewaterhouseCoopers LLP did not perform any financial information systems design, implementation or related services for us during the year ended December 31, 2002.
All Other Fees
The aggregate fees billed for all other services rendered to Inspire by PricewaterhouseCoopers LLP for the fiscal year ended December 31, 2002 were $8,950 which relate to recurring and non-recurring tax services.
6
AUDIT COMMITTEE REPORT
The Audit Committee of the Board of Directors has furnished the following report, in accordance with rules established by the Securities and Exchange Commission, for inclusion in this proxy statement.
The Audit Committee of the Board of Directors, which consists of Dr. Treu, as chairperson, Mr. Frey and Dr. Novack, reviews the results and scope of the annual audit and the services provided by PricewaterhouseCoopers LLP, the independent auditors. As part of its ongoing activities, the Audit Committee has:
| • | Reviewed and discussed with Inspire’s management the audited financial statements for the fiscal year ended December 31, 2002; |
| • | Discussed with PricewaterhouseCoopers LLP the matters required to be discussed by Statement on Auditing Standards No. 61, Communications with Audit Committees, as amended; and |
| • | Received the written disclosures and the letter from PricewaterhouseCoopers LLP required by Independence Standards Board Standard No. 1, Independence Discussions with Audit Committees, and has discussed with PricewaterhouseCoopers LLP their independence. |
Based on the review and discussions referred to above, the Audit Committee recommended to the Board of Directors that the audited financial statements be included in the Annual Report on Form 10-K for the fiscal year ended December 31, 2002.
Respectfully submitted,
Audit Committee
Jesse Treu
Kip Frey
Gary D. Novack
7
COMPENSATION COMMITTEE REPORT
The Compensation Committee of the Board of Directors furnishes the following report on the compensation of Inspire’s executive officers. The Compensation Committee, which consists of Dr. Thompson, as chairperson, and Dr. Treu and Mr. Frey, reviews and approves executive officer salaries and bonuses, administers the Amended and Restated 1995 Stock Plan, as amended, and approves all other compensation benefits of Inspire’s executive officers. In addition, the Compensation Committee consults with management regarding its benefit plans, performance evaluation and career development initiatives and is responsible for reviewing overall compensation policies and practices of Inspire.
Compensation Philosophies
Inspire intends that each employee will have continuous growth in skills and performance aided by appropriate mentoring and challenging career opportunities and rewarded by appropriate performance-based compensation. Compensation is a means of aligning the objectives of employees and stockholders.
Inspire provides competitive base salaries based on contribution and performance. In addition, many executive officers are granted bonuses and stock options in response to short-term corporate and personal performance and as incentives to continued contributions. Stock options are generally granted at the time of employment and periodically thereafter, with an appropriate schedule of vesting. Executive officers tend to have greater fractions of their total compensation from performance-based bonuses and stock options.
In 2002, executive compensation consisted of base salary, cash bonuses and stock options. The following describes in more detail the elements of compensation that are part of the Compensation Committee’s policies.
Base Salaries
Compensation of all Inspire employees is based on their: (i) demonstrated skills, experience, education and continued career development; (ii) impact within and outside of Inspire; (iii) the linkage of the employee’s performance with Inspire’s success; (iv) Inspire’s corporate achievement of annual and long-term goals; and (v) compensation of comparable employees within peer companies.
In considering compensation during 2002, the following objectives were among those considered: (i) preparations to file a New Drug Application (NDA) for diquafosol tetrasodium (INS365) for dry eye and the successful completion of Phase II and Phase III clinical trials for the same; (ii) the initiation of a Phase III clinical trial of INS37217 Intranasal in patients with perennial allergic rhinitis (PAR); and (iii) the positive results from a Phase II clinical trial for INS37217 Intranasal in patients with upper respiratory infection.
Bonuses
Cash bonuses are used to attract, retain and motivate executives. These bonuses are paid taking into account the executive officer’s achievement of individual objectives and the contribution of the executive to the overall success and achievements of the company and its management team. In determining the level of the cash bonus during 2002, the Compensation Committee considered certain significant accomplishments, including: preparations to file a NDA for diquafosol tetrasodium (INS365) for dry eye and the successful completion of Phase II and Phase III clinical trials for the same; the initiation of a Phase III clinical trial of INS37217 Intranasal in patients with PAR; the positive results from a Phase II clinical trial for INS37217 Intranasal in patients with upper respiratory infection; the appointment of executive officers and other key employees; and scientific publications and presentations.
Stock Options
The Compensation Committee believes that stock options are an excellent long-term incentive for executives that aligns executive and stockholder interests and assists in retention of key officers and employees. At the recommendation of the Compensation Committee to the Board, stock options are issued to new employees, including executive officers, following the commencement of employment.
8
Stock options granted under the Amended and Restated 1995 Stock Plan, as amended, generally vest over four years. The Compensation Committee has and may in the future, determine to more closely link the vesting of stock options with an executive’s achievement of a particular objective.
When determining additional stock option awards, the Compensation Committee considers the executive’s current responsibilities and contributions to Inspire’s performance, the anticipated contribution to meeting Inspire’s long-term strategic performance goals, his or her position with Inspire and industry practice in companies of similar size, market and capitalization. The direct link between the value of a stock option to an executive and an increase in the price of Inspire’s stock makes stock option awards a key method for aligning executive compensation with stockholder value.
Chief Executive Officer Compensation
Dr. Shaffer’s compensation is determined by the Compensation Committee without Dr. Shaffer’s participation, based upon the same factors as those used by the Compensation Committee and management for other executives. Dr. Shaffer participates in the same compensation arrangements available to the other senior executives. Accordingly, her compensation also consists of an annual base salary, a potential cash bonus, and stock option grants. The Compensation Committee’s general approach in establishing Dr. Shaffer’s compensation is to be competitive with the compensation paid to chief executive officers in peer companies, but to have a significant percentage of her compensation based upon performance criteria established by the Compensation Committee.
Dr. Shaffer’s compensation as Chief Executive Officer for the year ended December 31, 2002 included $260,300 in base salary and a stock option to purchase an aggregate of 100,000 shares of common stock at an exercise price of $2.76. In determining the base salary in 2002, the Compensation Committee reviewed Dr. Shaffer’s salary in relation to the salaries of other chief executive officers at emerging biopharmaceutical companies and Inspire’s achievement of certain goals during 2002, including: continued research and development efforts with respect to its product candidates; promotion of investor confidence; the readiness to file a NDA for diquafosol tetrasodium (INS365) for dry eye; the progress of the clinical research of INS37217 Intranasal in patients with PAR and INS37217 Intranasal in patients with upper respiratory infection.
Deductibility of Compensation
Section 162(m) of the Internal Revenue Code of 1986, as amended, generally disallows a federal income deduction to public companies for certain compensation over $1,000,000 paid to a company’s chief executive officer and four other most highly compensated executive officers. Qualifying performance-based compensation will not be subject to the deduction limit if certain requirements are met. The Compensation Committee intends to review the potential effects of Section 162(m) periodically and intends to structure its stock option grants and certain other equity-based awards in a manner that is intended to avoid disallowances under Section 162(m) of the Code unless the Compensation Committee believes that such compliance would not be in the best interests of Inspire or its stockholders.
Respectfully submitted,
Compensation Committee
Kip Frey
W. Leigh Thompson
Jesse I. Treu
9
RELATIVE STOCK PERFORMANCE
Set forth below is a line graph comparing the percentage change in the cumulative total stockholder return on our common stock to the cumulative total return of the NASDAQ Stock Market (U.S.) Index and the NASDAQ Biotechnology Stock Index for the period commencing August 3, 2000, the commencement of trading on the NASDAQ Market, and ended December 31, 2002:
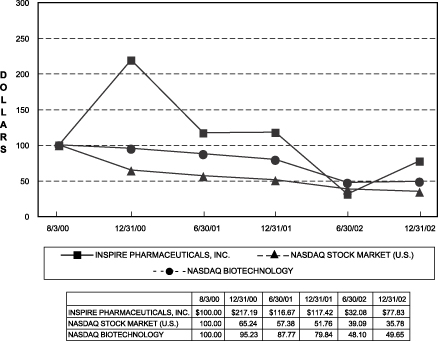
The graph assumes $100 was invested on August 3, 2000, in our common stock, and each of the indices, and that dividends were reinvested. The comparisons in the graph are required by the Securities and Exchange Commission and are not intended to forecast or be indicative of possible future performance of our common stock.
10
PROPOSAL NO. 2
RATIFICATION OF SELECTION OF AUDITORS
The Board of Directors has appointed PricewaterhouseCoopers LLP as the independent auditors for the fiscal year 2003. Stockholder ratification of the appointment is not required by our Amended and Restated Certificate of Incorporation, as amended, or Bylaws or otherwise, but the Board has decided to ascertain the position of the stockholders on the appointment. The Board of Directors will reconsider the appointment if it is not ratified. The affirmative vote of a majority of the shares voted at the meeting is required for ratification.
One or more representatives of PricewaterhouseCoopers LLP is expected to attend the Annual Meeting and have an opportunity to make a statement and/or respond to appropriate questions from stockholders.
THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL.
EXECUTIVE OFFICERS WHO ARE NOT DIRECTORS OR NOMINEES
The name, age and position of each person who is currently serving as an executive officer, who served as an executive officer in 2002 and who has been chosen to become an executive officer, and who is not a director is listed below, followed by summaries of their background and principal occupations. Executive officers are elected annually, and serve at the discretion of the Board of Directors.
Name | Age | Position | ||
Donald J. Kellerman, Pharm.D. | 48 | Senior Vice President, Development | ||
Benjamin R. Yerxa, Ph.D. | 37 | Senior Vice President, Discovery | ||
Richard M. Evans, Ph.D. | 43 | Vice President, Pharmaceutical Development | ||
Mary B. Bennett | 45 | Vice President, Operations and Communications | ||
Joseph K. Schachle | 38 | Vice President, Marketing and Sales | ||
Thomas R. Staab, II, CPA | 35 | Vice President and Chief Financial Officer designate |
Donald J. Kellerman, Pharm.D., has served as our Senior Vice President, Development since May 2000. He is responsible for the clinical and regulatory affairs aspects of our programs. Dr. Kellerman joined us in July 1999 as Vice President, Development. Before joining us, Dr. Kellerman spent 11 years with Glaxo Wellcome, from August 1997 to July 1999 and from April 1988 to August 1996, where he was director of various groups, including International OTC, U.S. Infectious Diseases, and the Inhaled Corticosteroid Group. He was clinical project leader for Flovent® from first U.S. clinical studies in 1989 to approval in 1996. From September 1996 to August 1997, he was Vice President of Clinical Research at Sepracor, Inc., where he was project leader for the Xopenex® NDA team. Before Glaxo Wellcome, Dr. Kellerman worked at E.R. Squibb and Sons, Inc. and Ciba-Geigy Corporation on several cardiovascular products. Dr. Kellerman holds a Doctor of Pharmacy and Bachelor of Science degree from the University of Minnesota.
Benjamin R. Yerxa, Ph.D., has served as our Senior Vice President, Discovery since March 2003 and as a Co-Chairman of our Scientific Advisory Board since June 2000. Dr. Yerxa joined us in August 1995 and previously held several positions, including Vice President, Discovery from February 2000 until March 2003, and Senior Director of Preclinical Programs. He supervises the molecular pharmacology, chemistry, pre-clinical and intellectual property teams. He created a new strategic opportunity for us by developing the concept of ophthalmic uses for our core P2Y2technology. Before being promoted to the position of Senior Director of Preclinical Programs in December 1999, Dr. Yerxa was Director of Preclinical Programs and, before that, Senior Research Chemist. While in chemistry, he served as the preclinical project leader for INS365. As a Senior Research Chemist his work focused on designing and synthesizing new P2Y receptor agonists. Before joining us, from October 1993 to August 1995, Dr. Yerxa was a Research Scientist at Burroughs Wellcome Co. Dr. Yerxa worked at Biophysica, Inc. for over two years, synthesizing radiocontrast agents. He developed scale-up procedures for the industrial production of Oxilan®, a marketed imaging product. Dr. Yerxa received his Ph.D. in Organic Chemistry from UC Irvine in 1993.
11
Richard M. Evans, Ph.D., has served as our Vice President, Pharmaceutical Development since June 2000. Dr. Evans joined us in October 1996 and has previously held several positions including Director and Senior Director of Pharmaceutical Development. He is responsible for all activities related to the manufacture, formulation development and testing of our products in development and for the identification and development of relevant drug delivery technologies. He also serves as Vice-Chair of the Inhalation Technology Focus Group for the American Association of Pharmaceutical Scientists. Prior to joining Inspire, Dr. Evans was Section Manager of Inhalation Dosage Forms at Rhône-Poulenc Rorer. He has over 13 years experience in the pharmaceutical industry, including positions at Delphi Pharmaceuticals and Rhône-Poulenc Rorer. Dr. Evans holds both a Bachelor of Pharmacy degree and a Doctorate in Pharmaceutical Chemistry, in the field of inhalation drug delivery, from the Welsh School of Pharmacy, University of Wales College of Cardiff.
Mary B. Bennetthas served as our Vice President, Operations and Communications since March 2001. As Vice President, Operations and Communications, she manages a variety of areas for Inspire, including Human Resources, Communications, Investor Relations and Operations. Mrs. Bennett comes to Inspire from Glaxo Wellcome, where she served as Worldwide Director for R&D Human Resources Effectiveness, Planning and Communication. Prior to her role in Human Resources, Mrs. Bennett held various roles during her 13 years at Glaxo Wellcome, including Worldwide Director for R&D Communications, Director of R&D Operations and Department Head of Comparative Medicine. She also led communications for major company redesign initiatives, and for the Glaxo Wellcome merger. Mrs. Bennett holds a BA in Behavioral Science from National Louis University and an MBA from Duke University.
Joseph K. Schachle re-joined us in February 2003 as our Vice President, Marketing and Sales. Mr. Schachle is responsible for the development of a marketing and sales infrastructure in North America to commercialize potential products from the Company’s pipeline, as well as partnered products. Mr. Schachle has extensive experience in pharmaceutical marketing and sales, and has had responsibility for the successful launch of major new products including the marketing of Glaxo SmithKline’s Advair. His experience includes marketing and sales positions of significant responsibility at The Upjohn Company, Glaxo SmithKline, Inspire and MedImmune. He most recently served as Director of Marketing for infectious disease products at MedImmune from April 2002 until January 2003, where he was responsible for strategic brand management and promotion of Synagis®, currently MedImmune’s largest product, and Cytogam®. In his previous role at Inspire from April 2001 until April 2002, Mr. Schachle was responsible for the new product planning and market research that laid the groundwork for the commercialization of diquafosol tetrasodium (INS365), the first potential product from Inspire’s pipeline. Mr. Schachle will now be responsible for implementing these plans, and for developing and executing a co-promotion strategy with Inspire’s partner, Allergan. Mr. Schachle holds a BBA in Marketing from James Madison University and an MBA from Old Dominion University.
Thomas R. Staab, II, CPA, will join Inspire in May 2003 as our Vice President and Chief Financial Officer. Currently, he holds senior financial positions with Triangle Pharmaceuticals, Inc., a biotechnology company listed on the NASDAQ National Market prior to its being acquired in 2003. Mr. Staab has served five years with Triangle, most recently as Vice President/Treasurer and Acting Chief Financial Officer. His experience includes equity financing, strategic alliances, mergers and acquisitions, drug commercialization, SEC reporting, treasury management and investor relations. Prior to joining Triangle, Mr. Staab was employed by Price Waterhouse LLP where he worked with publicly and privately held companies focusing on audits, business advisory and tax services. Mr. Staab received his BS in Business Administration and Master of Accounting degrees from The University of North Carolina, Chapel Hill.
12
EXECUTIVE COMPENSATION
The following table shows, for the fiscal years ended December 31, 2002, 2001 and 2000, the annual and long-term compensation we paid to our chief executive officer and our four most highly compensated executive officers who were serving at December 31, 2002 (collectively, the “named executive officers”). Following the rules of the Securities and Exchange Commission, the compensation described in the table does not include medical, group life insurance or some other benefits which are available generally to all of our salaried employees.
SUMMARY COMPENSATION TABLE
Year | Annual Compensation | Long Term Compensation | ||||||||||
Name and Principal Position | Salary ($) | Bonus ($) | Other Annual Compensation ($) | Shares Underlying Options (#) | ||||||||
Christy L. Shaffer, Ph.D. Chief Executive Officer and Director | 2002 2001 2000 | $ $ $ | 260,300 245,716 212,500 |
$ $ | — 70,000 50,000 | — — — | 130,000 5,000 114,285 | |||||
Gregory J. Mossinghoff President, Secretary, Treasurer and Director | 2002 2001 2000 | $ $ $ | 211,458 186,375 168,333 | $ $ $ | 40,000 60,000 50,000 | — — — | 130,000 5,000 85,714 | |||||
Donald J. Kellerman, Pharm.D. Senior Vice President, Development | 2002 2001 2000 | $ $ $ | 179,379 169,633 155,300 | $ $ $ | 60,000 60,000 50,000 | — — — | 20,000 5,000 71,428 | |||||
Mary B. Bennett Vice President, Operations and Communications | 2002 2001 2000 | $ $
| 156,886 127,631 — | $ $
| 50,000 50,000 — | — — — | 20,000 65,000 — | |||||
Benjamin R. Yerxa, Ph.D. Senior Vice President, Discovery | 2002 2001 2000 | $ $ $ | 163,546 152,911 140,172 | $ $ $ | 60,000 50,000 40,000 | — — — | 30,000 5,000 85,714 | |||||
13
OPTION GRANTS IN 2002
The following stock options grants were made to the named executive officers during the fiscal year ended December 31, 2002 pursuant to the terms of our Amended and Restated 1995 Stock Plan, as amended. The options listed below have exercise prices equal to the fair market value on the date of the grant.
Name | Number of Securities Underlying Options Granted | % of Total Options Granted to Employees In Fiscal Year | Exercise or Base Price ($/Share) | Expiration Date | Potential Realizable Value of Assumed Annual Rates of Stock Price Appreciation for Option Term (1) | ||||||||||
5% | 10% | ||||||||||||||
Christy L. Shaffer | 30,000(2) 100,000(3) | 3.1% 10.3% | $ $ | 2.76 2.76 | 6/4/12 6/4/12 | $ $ | 52,072 173,575 | $ $ | 131,962 439,873 | ||||||
Gregory J. Mossinghoff | 30,000(2) 100,000(3) | 3.1% 10.3% | $ $ | 2.76 2.76 | 6/4/12 6/4/12 | $ $ | 52,072 173,575 | $ $ | 131,962 439,873 | ||||||
Donald J. Kellerman | 20,000(4) | 2.1% | $ | 3.96 | 9/10/12 | $ | 34,715 | $ | 87,975 | ||||||
Benjamin R. Yerxa | 30,000(4) | 3.1% | $ | 3.96 | 9/10/12 | $ | 52,072 | $ | 131,962 | ||||||
Mary B. Bennett | 20,000(4) | 2.1% | $ | 3.96 | 9/10/12 | $ | 34,715 | $ | 87,975 | ||||||
| (1) | The potential realizable value is calculated based on the fair market value of our common stock on the date of the grant. These amounts only represent certain assumed rates of appreciation established by the SEC. There can be no assurance that the amounts reflected in this table or the associated rates of appreciation will be achieved. |
| (2) | Each option is exercisable as to 10,000 or one-third (1/3) of the shares underlying such option at any time on or after the date of the Company’s Annual Meeting of Stockholders in 2003, as to an additional 10,000 or one-third (1/3) of the shares at any time on or after the date of the Company’s Annual Meeting of Stockholders in 2004, and the final 10,000 or one-third (1/3) of the shares at any time on or after the date of the Company’s Annual Meeting of Stockholders in 2005. |
| (3) | Each option is exercisable as to 33,333 or one-third (1/3) of the shares underlying such option at any time after June 4, 2004 and as to the remaining 66,667 or two-thirds (2/3) shares at any time on or after June 4, 2005. |
| (4) | Each option is exercisable as to one-quarter (1/4) of the shares underlying such option at any time after September 10, 2003, as to one-forty-eighth (1/48) of the shares each month for 35 months thereafter and as to the remaining shares on the last month of vesting. |
14
AGGREGATED STOCK OPTION EXERCISES IN LAST FISCAL YEAR
AND FISCAL YEAR-END STOCK OPTION VALUES
The following table provides certain information as to certain stock options exercisable by the named executive officers for the fiscal year ended December 31, 2002, and the value of such options held by them at December 31, 2002, measured in terms of the closing price of our common stock on The NASDAQ Stock Market on December 31, 2002 which was $9.34 per share.
Shares Acquired on Exercise (#) | Value Realized(1) | Number of Unexercised Options at December 31, 2002 | Value of Unexercised In-the-Money Options at December 31, 2002 | |||||||||||||
Name | Exercisable | Unexercisable | Exercisable | Unexercisable | ||||||||||||
Christy L. Shaffer | 10,000 | $ | 24,575 | 427,907 | 166,376 | $ | 3,129,019 | $855,400 | ||||||||
Gregory J. Mossinghoff | — |
| — | 190,663 | 172,906 | $ | 1,129,889 | $981,778 | ||||||||
Donald J. Kellerman | — |
| — | 100,596 | 54,260 | $ | 408,213 | $196,026 | ||||||||
Mary B. Bennett | — |
| — | 29,458 | 55,542 | $ | 42,044 | $157,356 | ||||||||
Benjamin R. Yerxa | — |
| — | 91,963 | 61,008 | $ | 264,389 | $187,892 | ||||||||
| (1) | The value realized has been calculated by determining the difference between the fair market value underlying the options and the exercise price of the options at exercise. The fair market value of the options was determined by taking the average of the high and low price quoted on the NSADAQ National Market System on the day of exercise. |
401(k) PROFIT SHARING PLAN
We have adopted a tax-qualified employee savings and retirement 401(k) Profit Sharing Plan (the “401(k) Plan”) covering all qualified employees. The effective date of the 401(k) Plan is August 1, 1995. Participants may elect a salary reduction of at least 1% as a contribution to the 401(k) Plan, up to the statutorily prescribed annual limit for tax-deferred contributions ($11,000 in 2002). Modification of salary reductions can be made quarterly. The 401(k) Plan permits employer matching of up to 8% of a participant’s salary. If employer matching is implemented, participants will begin vesting 100% immediately in employer contributions. In 2002, we elected a safe harbor contribution at 3.0% of annual compensation. All of our safe harbor contributions vest 100% immediately.
15
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information with respect to the beneficial ownership of our common stock as of March 31, 2003, except as otherwise indicated, by: (i) each of our directors and nominees; (ii) the named executive officers; (iii) all beneficial owners of greater than 5% of our outstanding common stock; and (iv) all of our directors and officers as a group.
Name and Address of Beneficial Owner | Number of Shares Beneficially Owned(1) | Percentage Beneficially Owned (2) | ||
Arnold H. Snider (3) Deerfield Capital, L.P. Deerfield Management Company 780 Third Avenue, 37th Floor New York, NY 10017 | 2,400,000 | 7.6% | ||
Delaware Investments entities (4) 2005 Market Street Philadelphia, PA 19103 | 2,169,541 | 6.8% | ||
Domain Partners entities and related individuals (5) Domain Associates, L.L.C. One Palmer Square, Suite 515 Princeton, NJ 08542 | 2,077,484 | 6.6% | ||
Mary B. Bennett (6) | 41,144 | * | ||
Richard Boucher, M.D. (7) | 594,951 | 1.9% | ||
Kip Frey (8) | 0 | * | ||
Donald J. Kellerman, Pharm.D. (9) | 117,707 | * | ||
Gregory J. Mossinghoff (10) | 204,173 | * | ||
Gary D. Novack, Ph.D. (11) | 12,218 | * | ||
William R. Ringo, Jr. | 0 | * | ||
Christy L. Shaffer, Ph.D. (12) | 490,642 | 1.5% | ||
W. Leigh Thompson, M.D., Ph.D., D.Sc. (13) | 5,714 | * | ||
Jesse I. Treu, Ph.D. (14) | 2,093,462 | 6.6% | ||
Benjamin R. Yerxa, Ph.D. (15) | 137,353 | * | ||
All directors and executive officers as a group (12 persons) (16) | 3,780,861 | 11.6% |
| * | Less than one percent |
| (1) | Except as indicated by footnote, beneficial ownership includes all options which are exercisable within 60 days of March 31, 2003. Except as indicated by footnote, and subject to community property laws where applicable, the persons named in the table have sole voting and investment power for all shares of common stock shown as beneficially owned by them. |
| (2) | Applicable percentage of ownership is based on 31,688,893 shares of common stock outstanding on March 31, 2003, unless noted as otherwise. |
| (3) | The number of shares reported is based upon information disclosed by Arnold Snider, Deerfield Capital, L.P., Deerfield Partners, L.P., Deerfield Management Company and Deerfield International Limited on a Schedule 13G/A filed with the Securities and Exchange Commission on February 11, 2003. The Schedule 13G/A indicates that 1,344,000 shares are beneficially owned by Deerfield Capital, L.P., 1,344,000 shares are beneficially owned by Deerfield Partners, L.P., 1,056,000 shares are beneficially owned by Deerfield Management Company, 1,056,000 shares are beneficially owned by Deerfield International Limited and 2,400,000 shares are beneficially owned by Arnold H. Snider. Mr. Snider, |
16
Deerfield Capital, L.P., Deerfield Partners, L.P., Deerfield Management Company and Deerfield International Limited have shared voting and investment power with respect to all shares beneficially owned. |
| (4) | The number of shares reported is based upon information disclosed by Delaware Management Business Trust and Delaware Management Holdings, Inc. on a Schedule 13G/A filed with the Securities and Exchange Commission on February 7, 2003. The Schedule 13G/A indicates that 2,169,541 shares are beneficially owned by Delaware Management Business Trust and by Delaware Management Holdings, Inc. Delaware Management Holdings, Inc. and Delaware Management Business Trust have shared investment power with respect to 4,700 shares. Delaware Management Holdings, Inc. beneficially owns those shares which are beneficially owned by Delaware Management Business Trust. Lincoln National Corp. is the ultimate parent of Delaware Management Business Trust and Delaware Management Holdings, Inc. |
| (5) | The number of shares reported is based upon information disclosed by Domain Partners III, L.P. and DP III Associates, L.P. on a Schedule 13G/A filed with the Securities and Exchange Commission on January 21, 2003. The Schedule 13G/A indicates that 2,014,456 shares are beneficially owned by Domain Partners III, L.P. and 63,028 shares are beneficially owned by DP III Associates, L.P. One Palmer Square Associates III, L.P. is the general partner of Domain Partners III, L.P. and DP III Associates, L.P. Dr. Treu is a general partner of One Palmer Square Associates III, L.P., which is the sole general partner of Domain Partners III, L.P. and DP III Associates, L.P., and disclaims beneficial ownership of these securities, and any proceeds thereof, that exceed his pecuniary interest therein, and/or that are not actually distributed to him. See footnote (14) below. |
| (6) | Includes 5,000 shares of common stock and 36,144 shares of common stock underlying stock options granted to Ms. Bennett which will have vested within sixty days after March 31, 2003. Does not include 48,856 shares of common stock underlying stock options granted to Ms. Bennett which will not have vested within sixty days after March 31, 2003. |
| (7) | Includes 474,952 shares of common stock and 119,999 shares of common stock underlying stock options granted to Dr. Boucher which will have vested within sixty days after March 31, 2003. Does not include 30,000 shares of common stock underlying stock options granted to Dr. Boucher which will not have vested within sixty days after March 31, 2003. |
| (8) | Mr. Frey does not currently beneficially own any securities. Does not include 40,000 shares of common stock underlying stock options granted to Mr. Frey which will not have vested within sixty days after March 31, 2003. |
| (9) | Includes 300 shares of common stock held by Dr. Kellerman’s spouse, of which he disclaims beneficial ownership, and 117,407 shares of common stock underlying stock options granted to Dr. Kellerman which will have vested within sixty days after March 31, 2003. Does not include 40,450 shares of common stock underlying stock options granted to Dr. Kellerman which will not have vested within sixty days after March 31, 2003. |
| (10) | Includes 71,427 shares of common stock and 132,746 shares of common stock underlying stock options granted to Mr. Mossinghoff which will have vested within sixty days after March 31, 2003. Does not include 159,396 shares of common stock underlying stock options granted to Mr. Mossinghoff which will not have vested within sixty days after March 31, 2003. |
| (11) | Includes 725 shares of common stock held jointly by Dr. Novack and his spouse and 65 shares of common stock held by Dr. Novack’s daughter. Dr. Novack disclaims beneficial ownership of the 65 shares held by his daughter. Also includes 11,428 shares of common stock underlying a warrant which will have vested within sixty days after March 31, 2003. Does not include 6,667 shares of common stock underlying stock options granted to Dr. Novack which will not have vested within sixty days after March 31, 2003. |
| (12) | Includes 51,999 shares of common stock, 800 shares of common stock held by Dr. Shaffer’s children and 437,843 shares of common stock underlying stock options granted to Dr. Shaffer which will have vested within sixty days after March 31, 2003. Does not include 156,440 shares of common stock underlying stock options granted to Dr. Shaffer which will not have vested within sixty days after March 31, 2003. |
| (13) | Includes 5,714 shares of common stock underlying stock options granted to Dr. Thompson which will have vested within sixty days after March 31, 2003. Does not include 40,000 shares of common stock underlying stock options granted to Dr. Thompson which will not have vested within sixty days after March 31, 2003. |
17
| (14) | Includes 2,014,456 shares held by Domain Partners III, L.P., 63,028 shares held by DP III Associates, L.P., 10,264 shares of common stock and 5,714 shares of common stock underlying stock options granted to Dr. Treu which will have vested within sixty days after March 31, 2003. One Palmer Square Associates III, L.P. is the general partner of Domain Partners III, L.P. and DP III Associates, L.P. Dr. Treu is a general partner of One Palmer Square Associates III, L.P. See footnote (5) above. Dr. Treu shares voting and investment power with respect to these shares and disclaims beneficial ownership of such shares except to the extent of his proportional interest therein. Does not include 40,000 shares of common stock underlying stock options granted to Dr. Treu which will not have vested within sixty days after March 31, 2003. |
| (15) | Includes 35,454 shares of common stock and 101,899 shares of common stock underlying stock options granted to Dr. Yerxa which will have vested within sixty days after March 31, 2003. Does not include 51,072 shares of common stock underlying stock options granted to Dr. Yerxa which will not have vested within sixty days after March 31, 2003. |
| (16) | Includes an aggregate of 993,903 shares of common stock underlying stock options and warrants granted to all of the directors and executive officers as a group which will have vested within sixty days after March 31, 2003. |
18
CERTAIN TRANSACTIONS
We entered into a Consultation and Scientific Advisory Board Agreement with Dr. Richard Boucher, a member of the Board of Directors, in March 1995. The terms of the agreement provide that Dr. Boucher will serve as the Co-Chairman of our Scientific Advisory Board for an initial term of three years. The agreement automatically renews itself thereafter for successive one year terms unless terminated by either party. Under the agreement, Dr. Boucher also agreed to consult on the field of airway diseases and the development of low molecular weight molecules for therapeutic or diagnostic purposes. During the fiscal year ended December 31, 2002, we paid Dr. Boucher $50,000 for his services. In addition, he received an option to purchase 30,000 shares of our common stock at the exercise price of $2.76 per share as partial compensation for his service.
In addition, we fund research programs with The University of North Carolina at Chapel Hill (“UNC”) relating to uses of the P2Y receptor family. Dr. Boucher is currently serving as one of the three principal investigators with respect to the research. Inspire paid approximately $116,363 during the fiscal year ended December 31, 2002 for the work performed by UNC under the research program that Dr. Boucher leads.
COMPLIANCE WITH SECTION 16(a) OF THE EXCHANGE ACT
Under the securities laws of the United States, directors, executive officers and any person holding more than ten percent (10%) of our common stock are required to report their ownership of common stock and any changes in that ownership to the Securities and Exchange Commission on Forms 3, 4 and 5. Based on our review of the copies of such forms we have received, all required reports have been timely filed except for one Form 4 filed by H. Jefferson Leighton, a former director, reporting one transaction.
19
OTHER MATTERS
Management knows of no matters other than those described above that are to be brought before the meeting. However, if any other matter properly comes before the meeting, the persons named in the enclosed proxy will vote the proxy in accordance with their best judgment on the matter.
We will bear the cost of preparing and mailing the enclosed material. We may use the services of our officers and employees (who will receive no additional compensation) to solicit proxies. We intend to request banks and brokers holding shares of our common stock to forward copies of the proxy materials to those persons for whom they hold shares and to request authority for the execution of proxies. We will reimburse banks and brokers for their out-of-pocket expenses. We have retained our transfer agent, Computershare Trust Company, Inc., to aid in the mailing and solicitation of proxies, at an estimated cost of approximately $2,200.
Certain information contained in this proxy statement relating to the occupations and security holdings of our directors and officers is based upon information received from the individual directors and officers.
Upon request, we will furnish, without charge, by first class mail a copy of our Annual Report on Form 10-K for the year ended December 31, 2002, including financial statements and schedules thereto, to each of our stockholders of record on April 10, 2003. Such requests are to be made to Gregory J. Mossinghoff, President, Secretary and Treasurer, at our offices located at 4222 Emperor Boulevard, Suite 470, Durham, North Carolina 27703-8466 or by telephone at (919) 941-9777. We will mail such materials within one business day of our receipt of the request. A reasonable fee will be charged for copies of requested exhibits.
By Order of the Board of Directors, | ||
Gregory J. Mossinghoff President, Secretary and Treasurer | ||
Durham, North Carolina
April 17, 2003
20
APPENDIX
INSPIRE PHARMACEUTICALS, INC.
AMENDED AUDIT COMMITTEE CHARTER
Adopted: March 25, 2003
Organization and Membership
The Audit Committee (the “Committee”) is a committee of the Board of Directors (the “Board”) of Inspire Pharmaceuticals, Inc. (the “Company”). The Committee shall be comprised of at least three directors, each of whom is independent of management and the Company under the standards applied to companies listed on The Nasdaq Stock Market, Inc. (“Nasdaq”). All Committee members will be financially literate to the extent required by applicable law, rules or regulation and at least one member will have accounting or related financial management expertise.
Purpose
The purpose of the Committee is to assist the Board in fulfilling its duty to the stockholders with respect to overseeing the accounting and financial reporting processes of the Company and audits of financial statements of the Company. In so doing, it is the responsibility of the Committee to maintain a posture of effective and independent oversight and to promote free and open communication among the directors, the independent auditors and the financial management of the Company. The Committee shall prepare the report required by the rules of the Securities and Exchange Commission (the “SEC”) to be included in the Company’s annual proxy statement.
Meetings
The Committee shall meet at least quarterly and maintain minutes of each meeting. The Committee shall report the matters discussed at each meeting to the Board.
Authority
The Committee shall have the authority, to the extent it deems it necessary or appropriate, to retain independent legal, accounting or other advisors. The Committee shall have the authority and responsibility to make regular reports to the Board as outlined below.
Responsibilities
Annual Audit
| 1. | Recommend to the Board the independent auditors to be selected to audit the financial statements of the company and any divisions and subsidiaries it may have from time to time. |
| 2. | On an annual basis, obtain from the independent auditors a written statement delineating all relationships between the independent auditor and the Company, as required by Independence Standards Board Standard No. 1 (Independence Discussions with Audit Committees); discuss with the independent auditors any disclosed relationships or professional services that may impact on the objectivity or independence of the independent auditors; and take appropriate action to ensure the independence of the independent auditors. |
| 3. | The Committee shall preapprove all auditing services and permitted non-audit services (including the fees and terms thereof) to be performed for the Company by its independent auditor, subject to the de minimis exceptions for non-audit services described in Section 10A(i)(1)(B) of the Exchange Act which are approved by the Committee prior to the completion of the audit. The Committee may form and delegate authority to subcommittees consisting of one or more members when appropriate, including the authority to |
A-1
grant preapprovals of audit and permitted non-audit services, provided that decisions of such subcommittee to grant preapprovals shall be presented to the full Committee at its next scheduled meeting.
| 4. | Meet with the independent auditors and financial management of the Company to (a) review and approve the scope and timing of the annual audit and the quarterly reviews for the current year, (b) discuss the planning and staffing of the audit, (c) review and approve the independent auditor’s compensation, and (d) discuss other matters required to be reviewed under applicable corporate and securities legal and regulatory requirements, including Nasdaq requirements. |
| 5. | At the conclusion of the annual audit, discuss with the independent auditors and the financial management of the Company the results of the audit, including recommendations of the independent auditors and management’s response. Attention should be given to the following matters: |
| (a) | the Company’s audited financial statements and related footnotes; |
| (b) | matters which are required to be communicated to the Committee under SAS 61, including: |
| • | methods used to account for significant, unusual transactions, |
| • | the effect of significant accounting policies in controversial or emerging areas for which there is a lack of authoritative guidance or consensus, |
| • | the process used by management in formulating particularly sensitive accounting estimates and basis for the independent auditor’s conclusions regarding the reasonableness of those estimates, and |
| • | disagreements with management over the application of accounting principles, the basis for management’s accounting estimates, or the disclosures in the financial statements; |
| (c) | the adequacy and effectiveness of the accounting and financial controls of the Company. The Committee should elicit any recommendations for the improvement of such internal controls or particular areas where new or more detailed controls or procedures are desirable. Particular emphasis should be given to the adequacy of internal controls to expose any payments, transactions, or procedures that might be deemed illegal or otherwise improper; |
| (d) | the status of recommendations made in previous years by the independent auditors; and |
| (e) | recent or impending changes in accounting principles and disclosure practices. |
| 6. | Meet separately with the independent auditors, without members of management present, to discuss the following matters: |
| (a) | the independent auditors’ evaluation of the Company’s financial and accounting personnel; |
| (b) | the cooperation that the independent auditors received during the course of the audit; |
| (c) | the independent auditors’ evaluation of the degree of aggressiveness or conservatism of the Company’s accounting principles, the underlying estimates, and the significant judgments made by management in preparing financial statements; |
| (d) | the clarity of the financial disclosures; and |
| (e) | any other matters the independent auditors wish to raise. |
| 7. (a) | Report the results of the annual audit to the Board. Such report may include the attendance of the independent auditors at a full meeting of the Board or, alternatively, the attendance of other directors at the Committee meeting at which the results of the annual audit are presented. |
| (b) | Review disclosures made to the Committee by the Company’s Chief Executive Officer (“CEO”) and Chief Financial Officer (or such person performing the equivalent function) (the “CFO”) during their certification process for the annual report on Form 10-K (“Form 10-K”) and quarterly report on Form 10-Q (“Form 10-Q”) about any significant deficiencies in the design or operation of internal controls or |
A-2
material weaknesses therein and any fraud involving management or other employees who have a significant role in the Company’s internal controls.
Financial Reports
| 8. | Review and discuss with management and the independent auditor the proxy statement and the Form 10-K, including the audited annual financial statements and the disclosures made in management’s discussion and analysis, and, prior to the submission of such documents to the SEC, determine that the independent auditors are satisfied that the disclosures in these reports are consistent with the Company’s audited financial statements. |
| 9. | Review and discuss with management and the independent auditor the Company’s quarterly financial statements prior to the filing of its Form 10-Q and determine that, prior to filing, interim financial statements included in its Form 10-Q have been reviewed by the independent auditors under the standards of SAS 71 Interim Financial Review. The objective of this review is not to perform an audit but rather to provide the independent auditors with a basis for reporting whether material modifications should be made for the interim financial statements to conform with generally accepted accounting principles (“GAAP”). The report of the independent auditor on this review must be filed with the Form 10-Q. |
| 10. | With the assistance of management, prepare an annual Audit Committee Report for inclusion in the Company’s proxy statement and in the Form 10-K stating whether: (a) the Committee has reviewed and discussed the audited financial statements with management; (b) the Committee has discussed with the independent auditors the matters required to be discussed by SAS 61; (c) the Committee has received the written disclosures and the letter from the independent auditors required by Independence Standards Board Standard No. 1 and discussed with the independent auditors the independent auditors’ independence; and (d) based on the review and discussions in (a), (b) and (c) above, the Committee recommended to the Board that the audited financial statements be included in the Company’s Form 10-K for the last fiscal year for filing with the SEC. The annual report must also state the name of each member of the Company’s Committee. |
| 11. | Assure that management includes in the proxy statement for the annual meeting of stockholders a statement that the Committee has a formal written Charter; include a copy of this Charter as an appendix to the proxy statement every three years, or sooner in the event of a significant amendment to the Charter. |
| 12. | Review and discuss quarterly reports from the independent auditors on: |
| • | critical accounting policies and practices to be used; |
| • | alternative treatments of financial information within GAAP that have been discussed with management, ramifications of the use of such alternative disclosures and treatments, and the treatment preferred by the independent auditor; and |
| • | other material written communications between the independent auditor and management, such as any management letter or schedule of unadjusted differences. |
Oversight of the Company’s Relationship with the Independent Auditor
| 13. | To the extent required by law or otherwise deemed necessary by the Committee, the Committee shall at least annually review whether the provision of permitted non-audit services is compatible with maintaining the independent auditor’s independence. |
| 14. | Ensure the rotation of the lead (or coordinating) audit partner having primary responsibility for the audit and any other member of the audit engagement team as required, if at all, by law. |
| 15. | Recommend to the Board policies for the Company’s hiring of employees or former employees of the independent auditor who participated in any capacity in the audit of the Company. |
A-3
Other Duties
| 16. | Obtain the full Board’s approval of this Charter, and review and reassess this Charter at least annually. |
| 17. | Beginning at such time as an internal audit function is warranted, review and concur with management’s appointment of a director of internal audit, and include the internal audit function in this Charter. |
| 18. | Investigate any matter brought to its attention within the scope of its duties, with the power to engage and determine funding for independent legal counsel, accounting or other necessary expert advisors for this purpose if, in its judgment, that is appropriate. |
| 19. | Obtain from the independent auditor assurance that Section 10A(b) of the Exchange Act has not been implicated. |
A-4
INSPIRE PHARMACEUTICALS, INC.
Revocable Proxy for 2003 Annual Meeting
This Proxy is Solicited on Behalf of The Board of Directors
The undersigned hereby appoints Christy L. Shaffer, Ph.D. and Gregory J. Mossinghoff or either of them, each with power of substitution, his or her full and lawful agents and proxies to vote all shares of common stock which the undersigned would possess if personally present at the Annual Meeting of Stockholders (including all adjournments thereof) of Inspire Pharmaceuticals, Inc. to be held on Monday, June 9, 2003, at the Wentworth Mansion, 149 Wentworth Street, Charleston, South Carolina 29401.
The Board of Directors recommends a vote FOR each of the proposals listed below and more fully described in the Notice of Annual Meeting of Stockholders and proxy statement for the meeting (receipt of which is hereby acknowledged). Unless otherwise specified, the vote represented by this proxy will be cast FOR proposals 1 and 2.
INSPIRE PHARMACEUTICALS, INC.
Please mark vote in box in the following manner using dark ink only | x | ¨ | Mark this box with an X if you have made changes to your name or address details. |
Proxy Card | Proxy Card |
1. Election of Directors. Nominees:
For | Withhold | For | Against | Abstain | ||||||||||
A. W. Leigh Thompson, M.D., Ph.D., D.Sc. | ¨ | ¨ | 2. | Ratification of the selection of PricewaterhouseCoopers LLP as auditors for the fiscal year ending December 31, 2003. | ¨ | ¨ | ¨ | |||||||
B. Gary D. Novack, Ph.D. | ¨ | ¨ | 3. | In their discretion, the proxies are authorized to vote upon such other business as may properly come before the meeting. | ||||||||||
C. William R. Ringo, Jr. | ¨ | ¨ | PLEASE CHECK THE BOX below if you, and any persons related or unrelated to you at the same address, are currently receiving multiple copies of the proxy statement and Annual Report and you wish to cease future deliveries of the proxy statement and Annual Report for the shares represented hereby. PLEASE NOTE you will continue to receive a proxy card and be able to vote the shares represented hereby and can revoke this consent at any time by following the procedures set forth in the proxy statement. | |||||||||||
¨ I hereby CONSENT to discontinue delivery of the proxy statement and Annual Report for the shares represented hereby. | ||||||||||||||
STOCKHOLDERS ARE REQUESTED TO SIGN AND DATE THIS PROXY AND RETURN IT PROMPTLY IN THE ENCLOSED ENVELOPE. NO POSTAGE IS REQUIRED IF MAILED IN THE UNITED STATES.
PLEASE SIGN HERE exactly as your name appears on this proxy. When shares are held by joint tenants, each joint tenant should sign. When signing as attorney, executor, administrator, trustee, guardian or other fiduciary, please give full title as such. If the signer is a corporation, please sign in full corporate name by a duly authorized officer; if a partnership, please sign in the partnership name by an authorized person.
Signature of Stockholder | Signature if held jointly | Date | ||