Exhibit 99.1

INSPIRE

Christy L. Shaffer, Ph.D. President and Chief Executive Officer Inspire Pharmaceuticals
Inspire R&D Investor Meeting – March 21, 2006

Forward-Looking Statements
This presentation contains forward-looking statements as defined by the Private Securities Litigation Reform Act of 1995 which convey management’s expectations, beliefs, plans and objectives regarding future performance. These statements discuss matters that are not facts, and may include words to indicate their uncertain nature such as “believe,” “expect,” “anticipate,” “intend,” “plan,” “estimate,” “project,” and “target.” Forward-looking statements are subject to significant risks and uncertainties that could affect such future performance including those relating to product development, revenue, expense and earnings expectations, intellectual property rights, adverse litigation developments, adverse developments in the U.S. Securities and Exchange Commission (SEC) investigation, competitive products, results and timing of clinical trials, success of marketing efforts, the need for additional research and testing, delays in manufacturing, funding, and the timing and content of decisions made by regulatory authorities, including the U.S. Food and Drug Administration. Actual results could differ materially from those described in this presentation. Information on various factors that could affect the information contained in this presentation and Inspire’s performance are detailed in the reports we file with the SEC.
Inspire R&D Investor Meeting – March 21, 2006

Inspire R&D Investor Meeting Agenda
Corporate Overview
Scientific Overview of Cystic Fibrosis (CF)
CF Program Overview
Scientific Overview of Allergy
Intranasal Epinastine Program Overview
INS50589 Program Overview
Christy Shaffer, Ph.D.
Richard Boucher, M.D.
Lynn Smiley, M.D.
William Berger, M.D., M.B.A.
Don Kellerman, Pharm.D.
Ben Yerxa, Ph.D.
Inspire Pharmaceuticals President and CEO
UNC-Chapel Hill, Kenan Professor of Medicine, Dir., CF/Pulmonary Research and Treatment Center Inspire Pharmaceuticals Chief Medical Officer
University of CA, Irvine, Clinical Professor, Dept. of Pediatrics Division of Allergy and Immunology
Inspire Pharmaceuticals Sr. VP, Development
Inspire Pharmaceuticals Chief, Scientific Operations & Alliances
Inspire R&D Investor Meeting – March 21, 2006
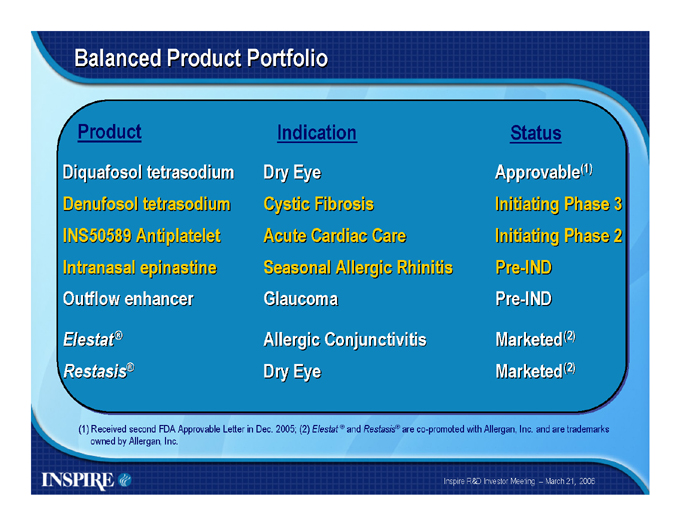
Balanced Product Portfolio
Product
Diquafosol tetrasodium Denufosol tetrasodium INS50589 Antiplatelet Intranasal epinastine Outflow enhancer
Elestat® Restasis®
Indication
Dry Eye Cystic Fibrosis Acute Cardiac Care Seasonal Allergic Rhinitis Glaucoma
Allergic Conjunctivitis Dry Eye
Status
Approvable(1) Initiating Phase 3 Initiating Phase 2 Pre-IND
Pre-IND
Marketed(2) Marketed(2)
(1) Received second FDA Approvable Letter in Dec. 2005; (2) Elestat ® and Restasis® are co-promoted with Allergan, Inc. and are trademarks owned by Allergan, Inc.
Inspire R&D Investor Meeting – March 21, 2006

Financial Overview
Receiving co-promotion revenues on Elestat® and Restasis® Potential to achieve revenues from out-licensing deals, including potential additional milestone payments from Allergan $122 million cash balance at 12/31/05 2006 Guidance*
Aggregate revenue—$31 to $39 million Operating expenses—$77 to $86 million 2+ years of operating capital
*Revenue and expense guidance provided on 2/28/06
Inspire R&D Investor Meeting – March 21, 2006
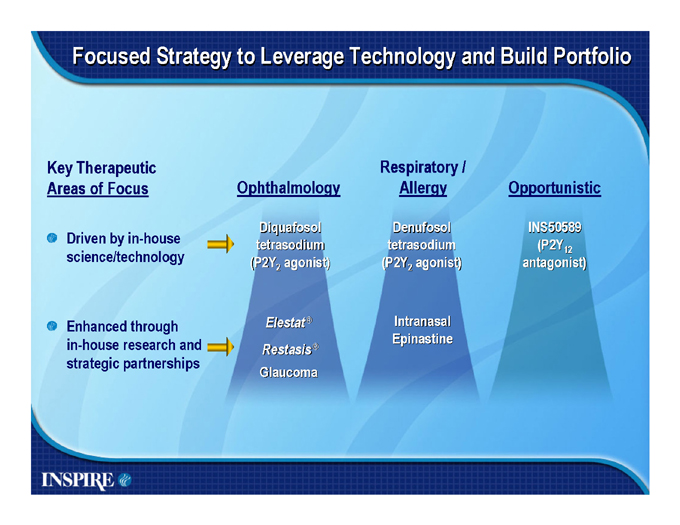
Focused Strategy to Leverage Technology and Build Portfolio
Key Therapeutic Areas of Focus
Driven by in-house science/technology
Enhanced through in-house research and strategic partnerships
Ophthalmology
Diquafosol tetrasodium
(P2Y2 agonist)
Elestat® Restasis®
Glaucoma
Respiratory / Allergy
Denufosol tetrasodium
(P2Y2 agonist)
Intranasal Epinastine
Opportunistic
INS50589
(P2Y12
antagonist)
Creating Successful Collaborations
Inspire R&D Investor Meeting – March 21, 2006

Overview of Cystic Fibrosis
Richard C. Boucher, M.D.
Kenan Professor of Medicine
Director, CF/Pulmonary Research and Treatment Center University of North Carolina at Chapel Hill

Disclosure
The presenter is a co-founder of Inspire Pharmaceuticals and has served as a consultant and clinical investigator for Inspire.
The presenter owns stock and stock options in Inspire Pharmaceuticals.
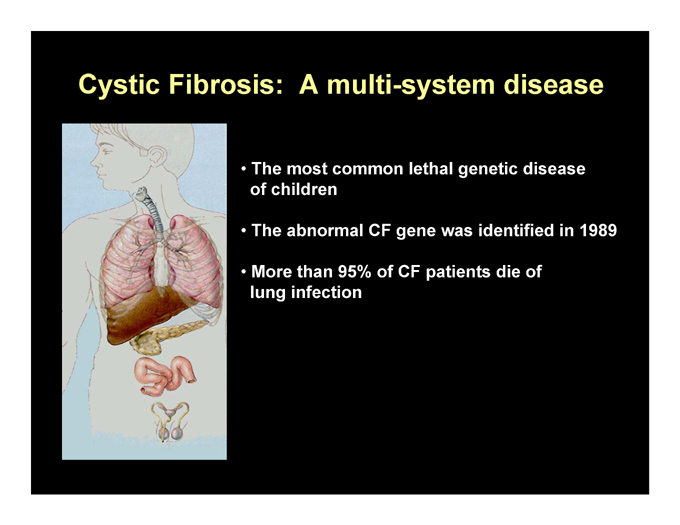
Cystic Fibrosis: A multi-system disease
The most common lethal genetic disease of children
The abnormal CF gene was identified in 1989
More than 95% of CF patients die of lung infection
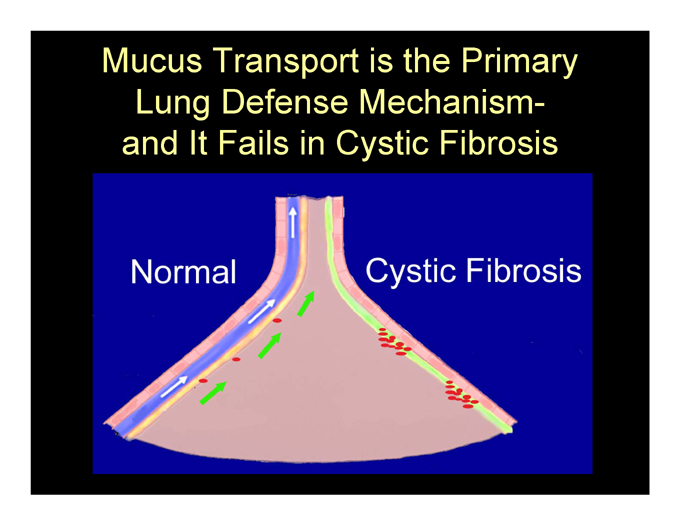
Mucus Transport is the Primary Lung Defense Mechanism-and It Fails in Cystic Fibrosis
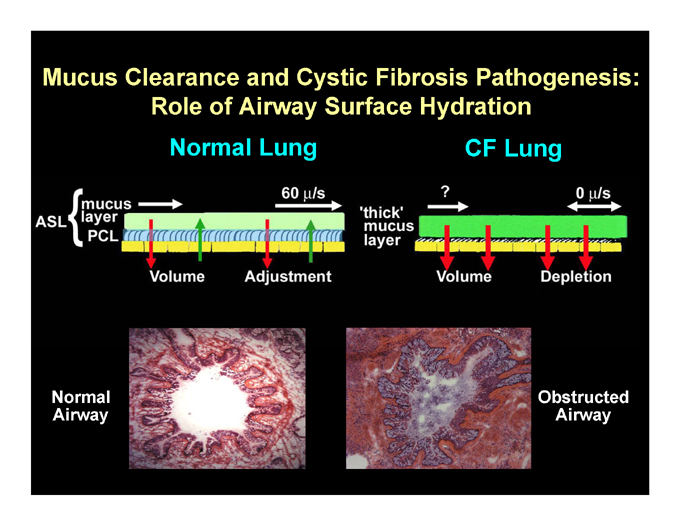
Mucus Clearance and Cystic Fibrosis Pathogenesis: Role of Airway Surface Hydration Normal Lung CF Lung
Normal Cystic Fibrosis

The in Vitro Model System that Enabled the Study of Airways Defense

The Model System Maintains Mucus Transport in Vitro
Note: This slide contains a video clip that is not available in PDF format or webcast.
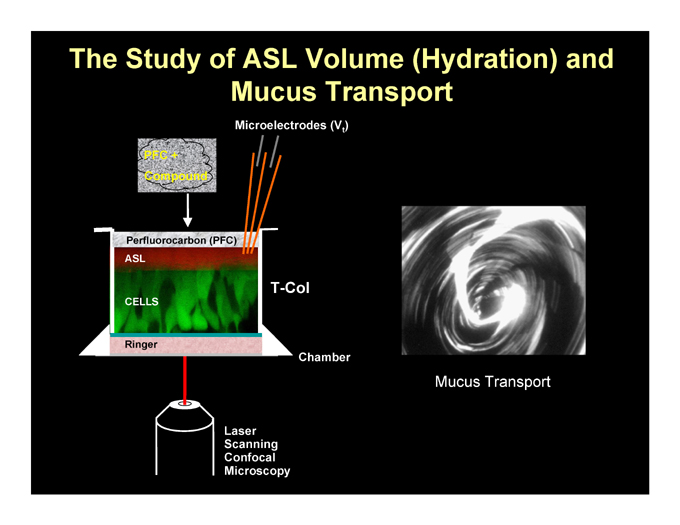
The Study of ASL Volume (Hydration) and Mucus Transport
Microelectrodes (Vt)
T-Col
Chamber
Mucus Transport
Laser Scanning Confocal Microscopy
Ringer
Perfluorocarbon (PFC)
ASL
CELLS
PFC + Compound
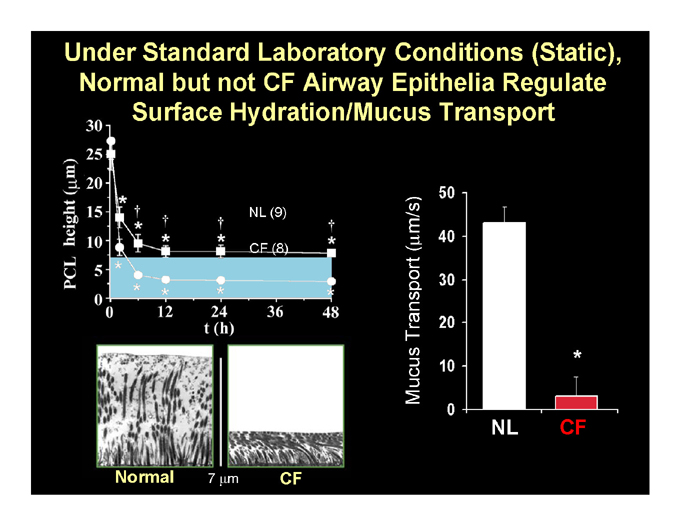
Under Standard Laboratory Conditions (Static), Normal but not CF Airway Epithelia Regulate Surface Hydration/Mucus Transport
NL CF
Normal 7 ?m CF
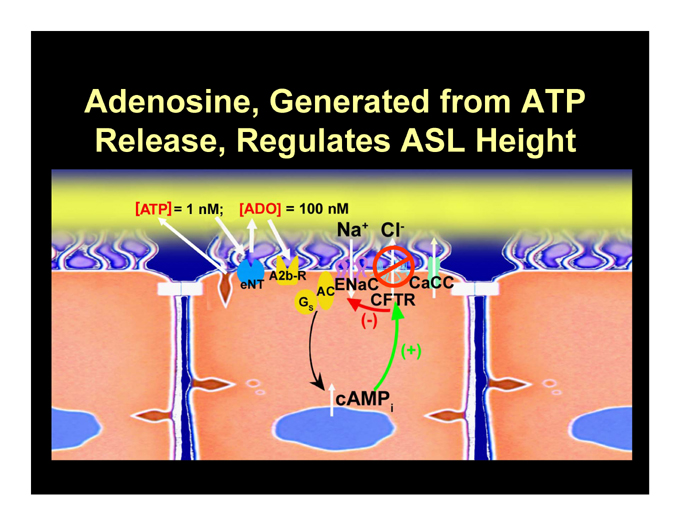
Adenosine, Generated from ATP Release, Regulates ASL Height

Adenosine Regulates ASL Volume Homeostasis in Normal but not CF Airway Epithelia
NL
[ADO] = 100 ± 19 nM (n=10)
CF
[ADO] = 189 ± 71 nM (n=3)
Control (9)
8-SPT (5)
Control (8)
8-SPT (6)
8-SPT = Adenosine Receptor Antagonist

Mucus Transport is Preserved for Long Periods of Time in Normal Cultures
T = 0
T = 24 hour
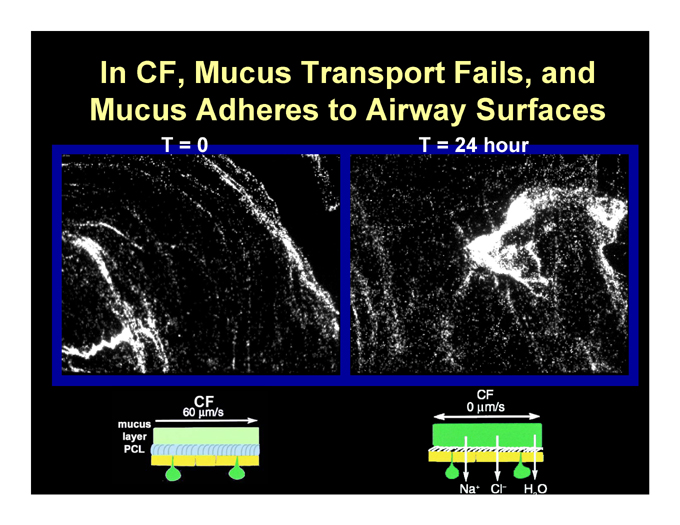
In CF, Mucus Transport Fails, and Mucus Adheres to Airway Surfaces
T = 0
T = 24 hour
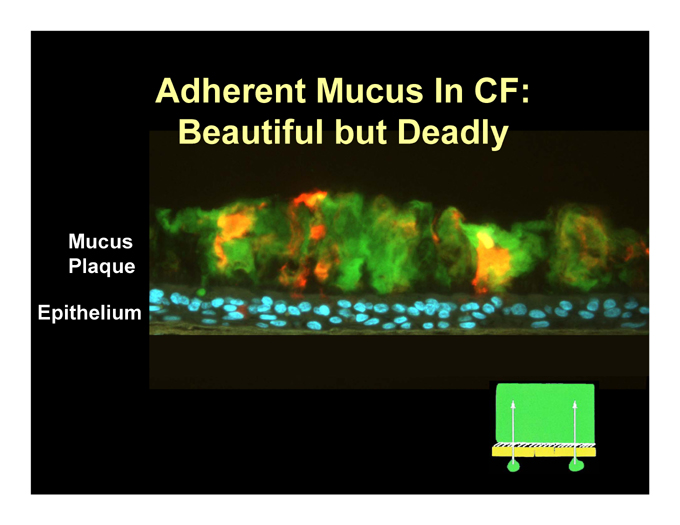
Adherent Mucus In CF: Beautiful but Deadly
Mucus Plaque
Epithelium
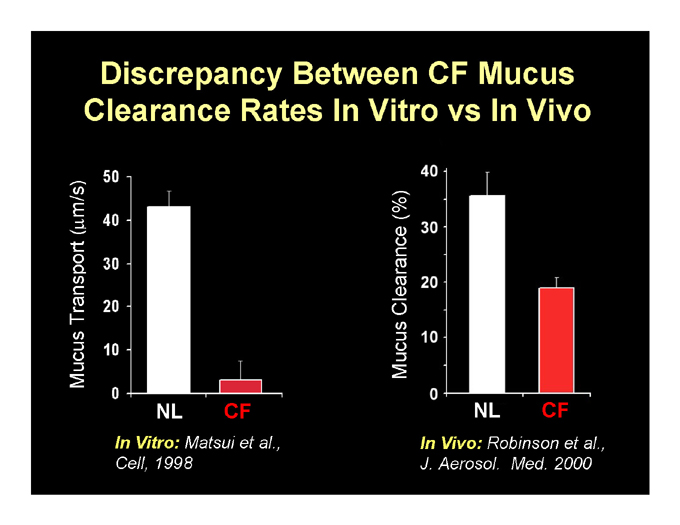
Discrepancy Between CF Mucus Clearance Rates In Vitro vs In Vivo
NL CF
NL CF
In Vitro: Matsui et al., Cell, 1998
In Vivo: Robinson et al., J. Aerosol. Med. 2000
Mucus Transport (%)
Mucus Clearance (?m/s)
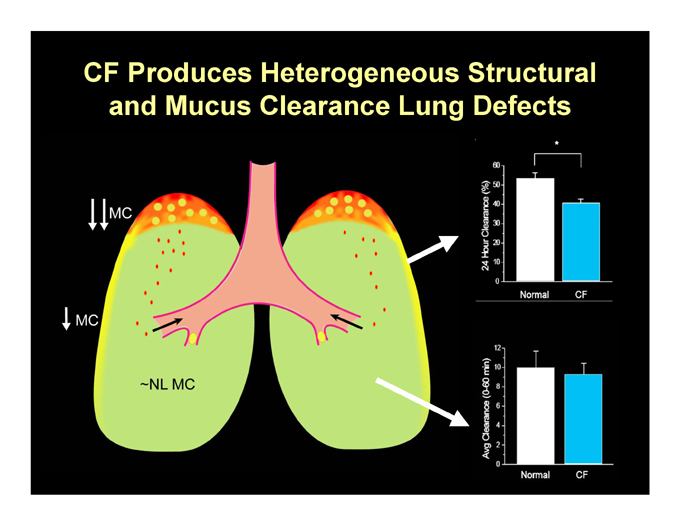
CF Produces Heterogeneous Structural and Mucus Clearance Lung Defects

In Vitro Modeling of the Phasic Motion and Shear Stress Associated with Respiration
shear = h r
t
Where:
p= density h = ASL height q= angular velocity r = distance from the center of rotation t = time of acceleration
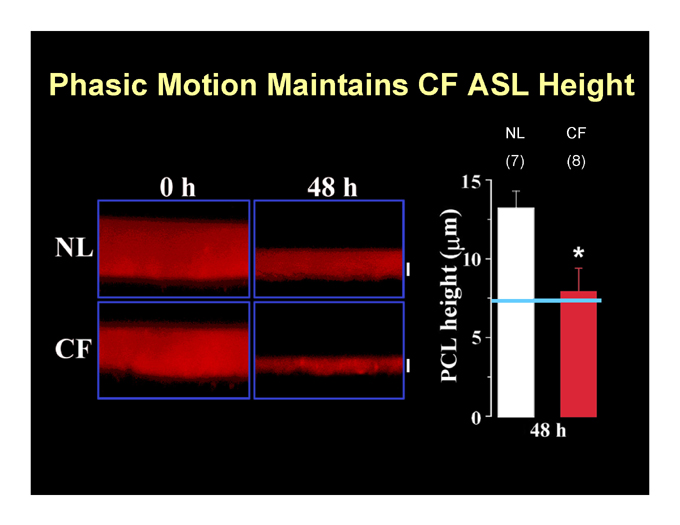
Phasic Motion Maintains CF ASL Height
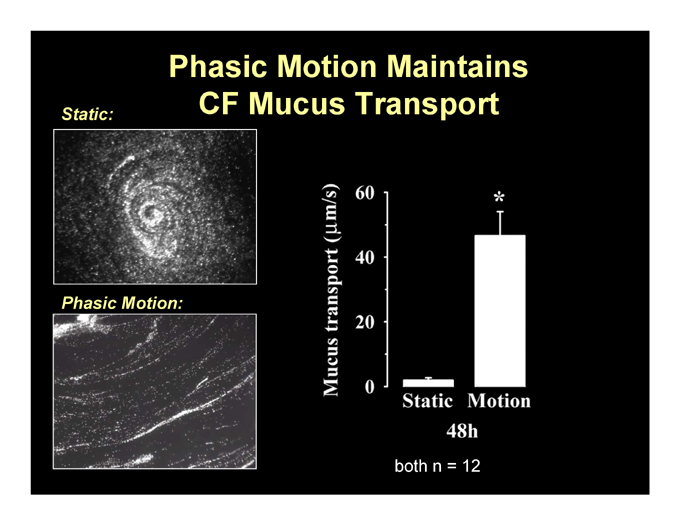
Phasic Motion Maintains CF Mucus Transport
Static:
Phasic Motion: both n = 12
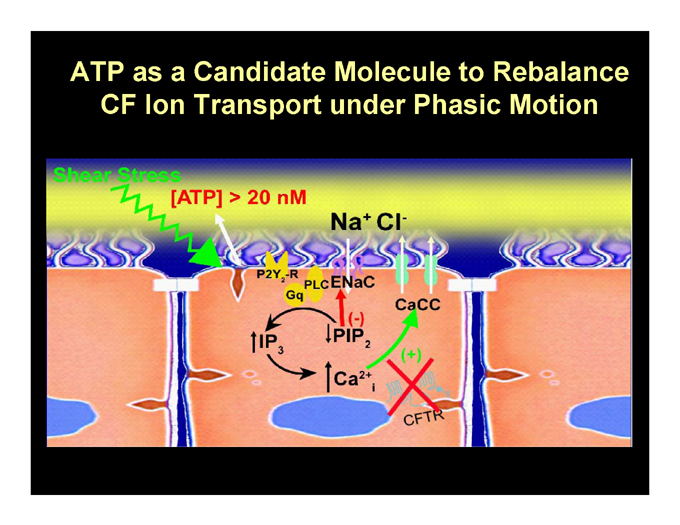
ATP as a Candidate Molecule to Rebalance CF Ion Transport under Phasic Motion
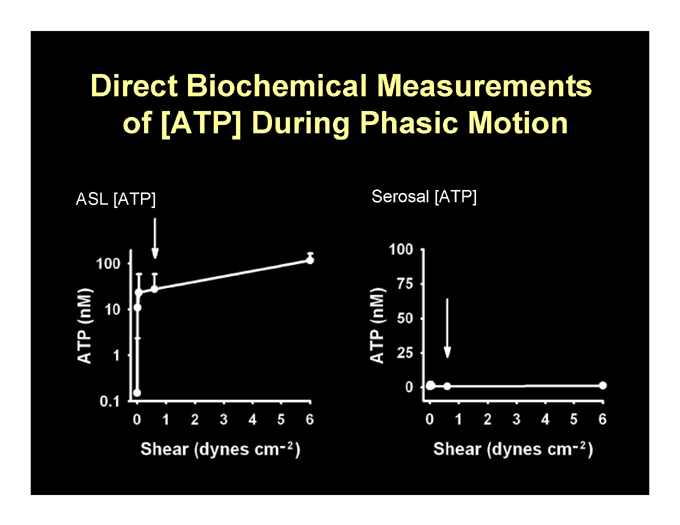
Direct Biochemical Measurements of [ATP] During Phasic Motion
ASL [ATP] Serosal [ATP]
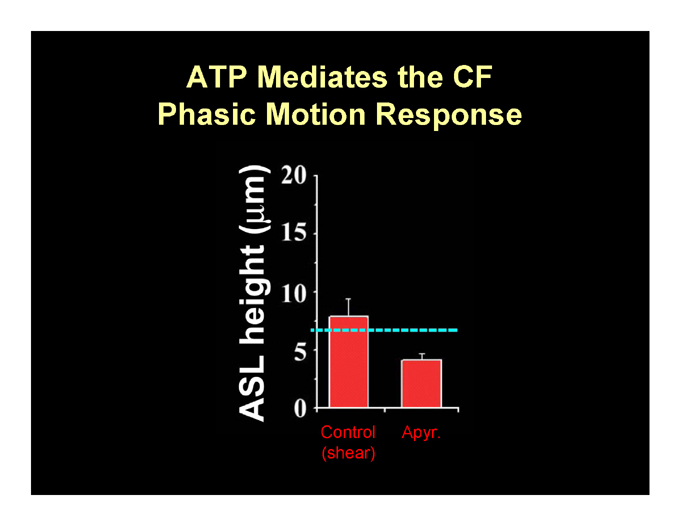
ATP Mediates the CF Phasic Motion Response
Control Apyr. (shear)
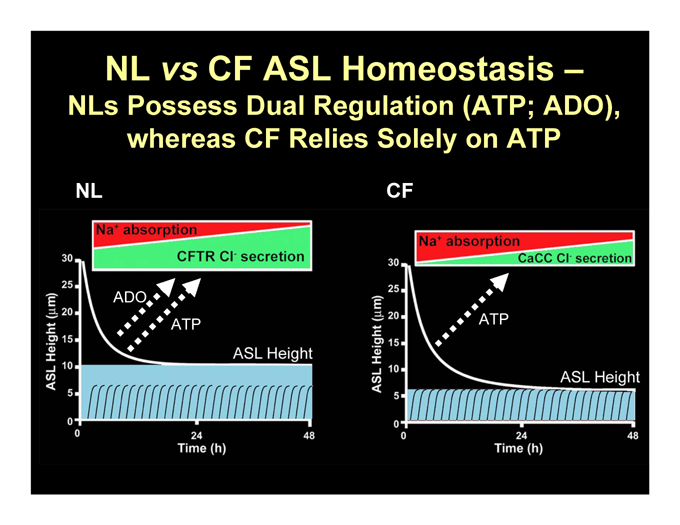
NL vs CF ASL Homeostasis –
NLs Possess Dual Regulation (ATP; ADO), whereas CF Relies Solely on ATP
NL
CF
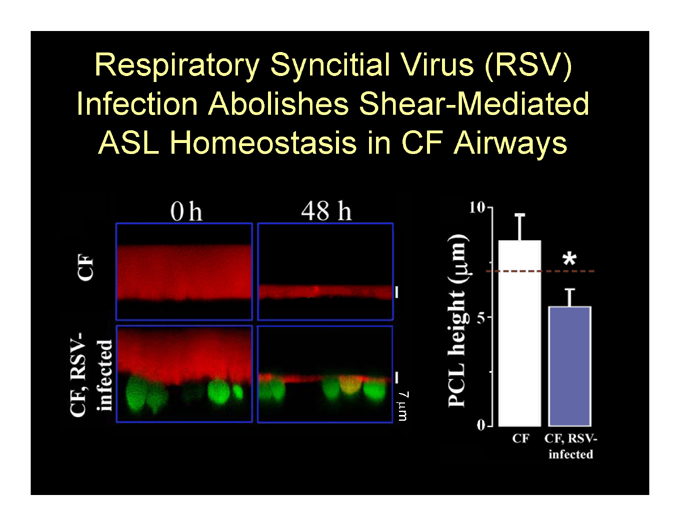
Respiratory Syncitial Virus (RSV) Infection Abolishes Shear-Mediated ASL Homeostasis in CF Airways
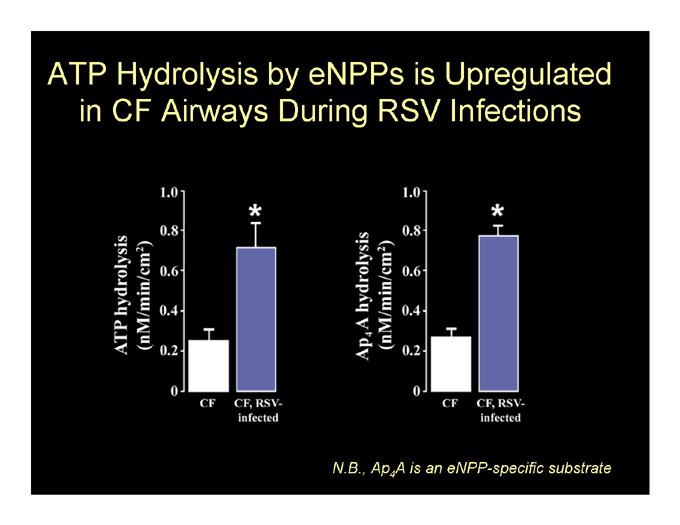
ATP Hydrolysis by eNPPs is Upregulated in CF Airways During RSV Infections
N.B., Ap4A is an eNPP-specific substrate
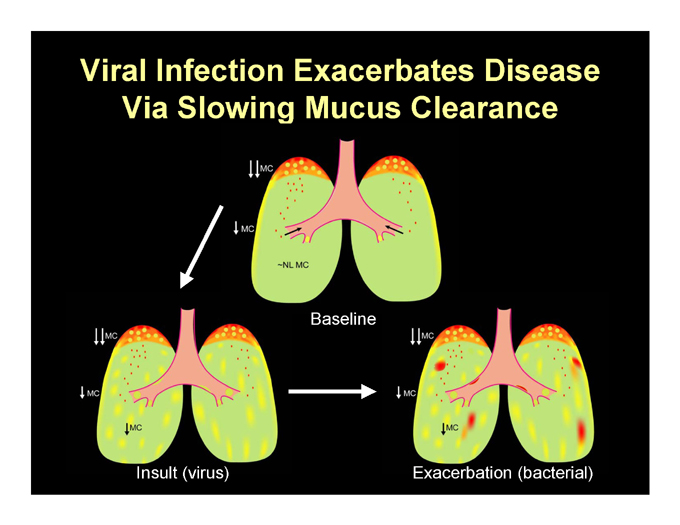
Viral Infection Exacerbates Disease Via Slowing Mucus Clearance
Baseline
Insult (virus)
Exacerbation (bacterial)

How Do You Restore ASL Volume and Mucus Clearance in CF
Therapeutically? Add Salt.
Osmotically: HS
Pharmacologically: ATP/UTP t (min) t (min)
ASL Height (?m)
40 35 30 25 20 15 10 5 0
HS
0
20
40
60
ASL Height (?m)
20 15 10 5 0
0
20
40
60
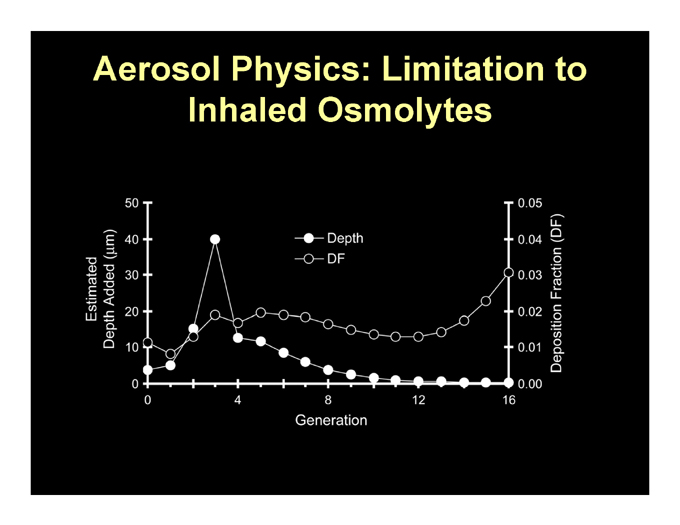
Aerosol Physics: Limitation to Inhaled Osmolytes
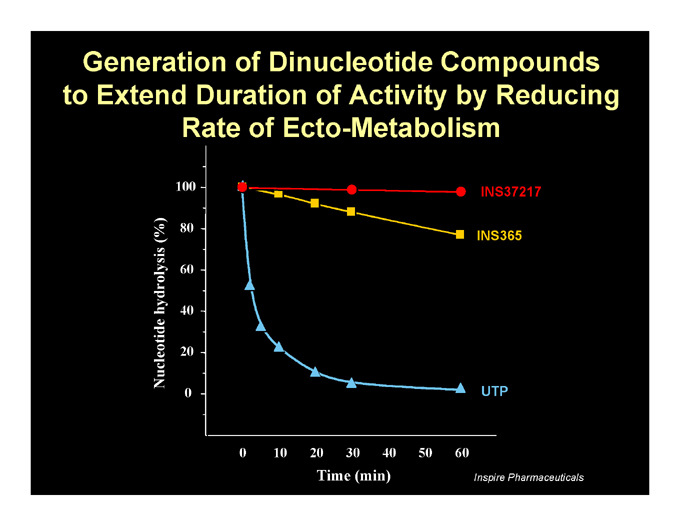
Generation of Dinucleotide Compounds to Extend Duration of Activity by Reducing Rate of Ecto-Metabolism
INS37217
INS365
UTP
Nucleotide hydrolysis (%)
100 80 60 40 20 0
0 10 20 30 40 50 60
Time (min)
Inspire Pharmaceuticals
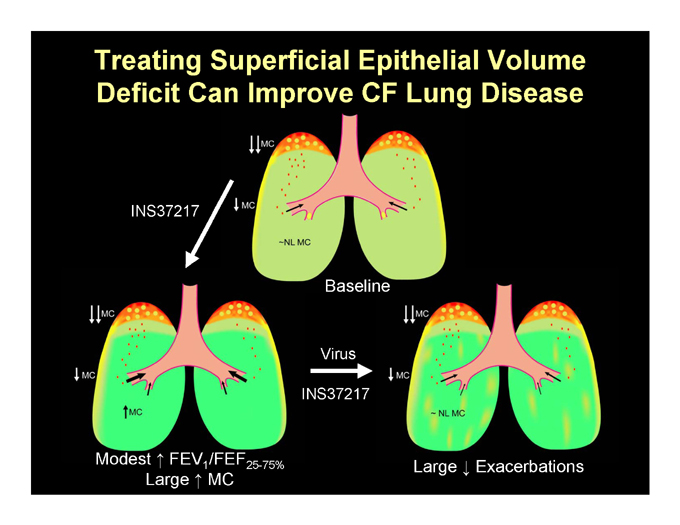
Treating Superficial Epithelial Volume Deficit Can Improve CF Lung Disease
INS37217
Baseline
Virus
INS37217
Modest FEV1/FEF25-75% Large Exacerbations Large MC

Denufosol Tetrasodium: A Potential Breakthrough
08-103 Inspire Trial Summary of Spirometry Results
Significant differences1 in favor of active doses (pooled) for all 4 spirometry parameters
FEV1 (max): p-value = 0.006 FEF25%-75% : p-value = 0.008 FVC (max): p-value = 0.022
FEV1 /FVC : p-value = 0.047
1ITT analysis, adjusting for pooled site, baseline spirometry, age, gender, and Pa status.
Deterding, et al., NACF 2004
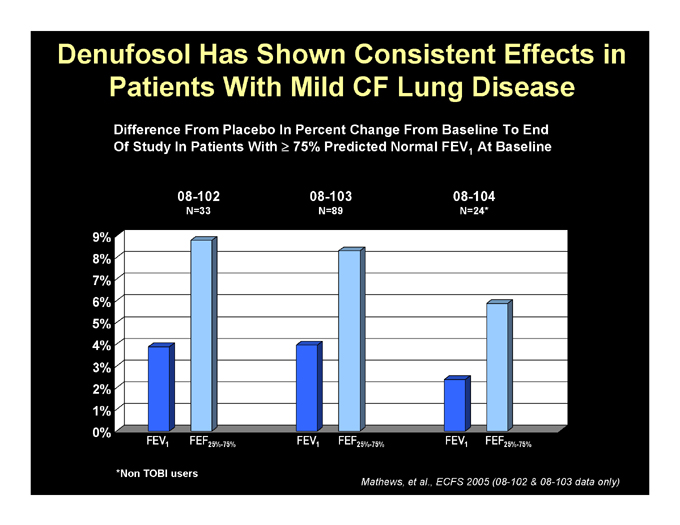
Denufosol Has Shown Consistent Effects in Patients With Mild CF Lung Disease
Difference From Placebo In Percent Change From Baseline To End
Of Study In Patients With 75% Predicted Normal FEV1 At Baseline
08-102
N=33
08-103
N=89
08-104
N=24*
9% 8% 7% 6% 5% 4% 3% 2% 1% 0%
FEV1 FEF25%-75% FEV1 FEF25%-75% FEV1 FEF25%-75%
*Non TOBI users
Mathews, et al., ECFS 2005 (08-102 & 08-103 data only)
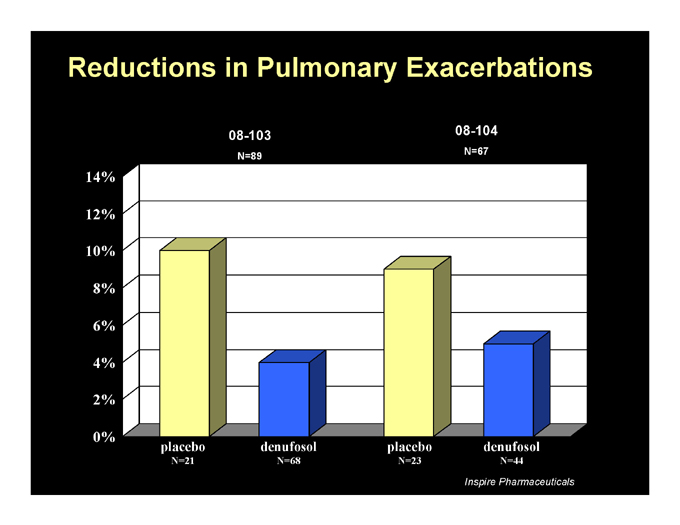
Reductions in Pulmonary Exacerbations
08-103
N=89
08-104
N=67
14% 12% 10% 8% 6% 4% 2% 0% placebo
N=21 denufosol
N=68 placebo
N=23 denufosol
N=44
Inspire Pharmaceuticals
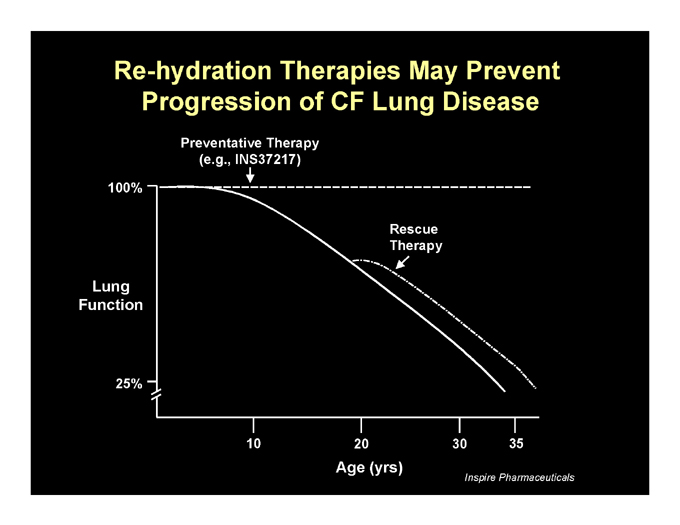
Re-hydration Therapies May Prevent Progression of CF Lung Disease
Preventative Therapy (e.g., INS37217)
100%
Lung Function
25%
Rescue Therapy
10
20
30
35
Age (yrs)
Inspire Pharmaceuticals

Overview of Cystic Fibrosis
Richard C. Boucher, M.D.
Kenan Professor of Medicine
Director, CF/Pulmonary Research and Treatment Center University of North Carolina at Chapel Hill

M. Lynn Smiley, M.D. Chief Medical Officer Inspire Pharmaceuticals
Inspire R&D Investor Meeting – March 21, 2006

Regulatory Status of Denufosol Tetrasodium
Advancing to Phase 3 based on Phase 1 & 2 safety and efficacy findings and completion of End-of-Phase 2 FDA meeting
U.S. and European Union Orphan Drug designation
Fast track status in U.S.
European Medicines Agency (EMEA) protocol assistance process initiated
Inspire R&D Investor Meeting – March 21, 2006

Overview of Clinical Development Program to Date
Over 300 patients/volunteers studied in 6 completed clinical trials
Phase 1 trial (volunteers)
08-101
Phase 2 trials (CF patients)
Dose-ranging nasal potential difference study (UNC) 08-102 08-103 08-104
2 PK studies (volunteers)
08-105 08-106
Inspire R&D Investor Meeting – March 21, 2006
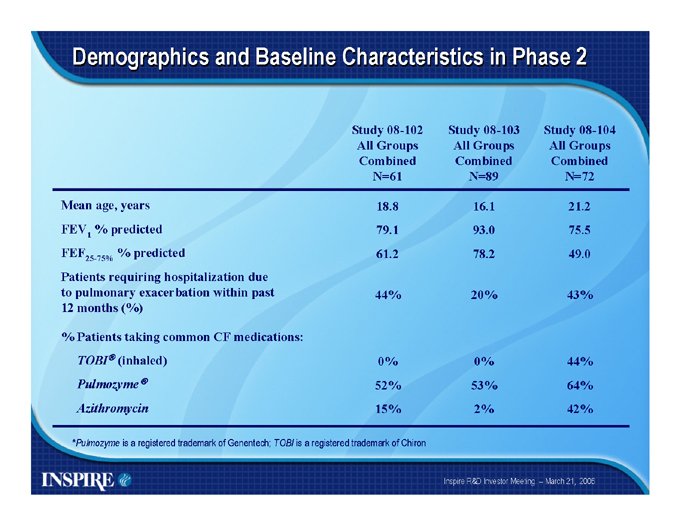
Demographics and Baseline Characteristics in Phase 2
Study |
| 08-102 All Groups Combined N=61 Study 08-103 All Groups Combined N=89 Study 08-104 All Groups Combined N=72 |
Mean |
| age, years 18.8 16.1 21.2 |
FEV1 |
| % predicted 79.1 93.0 75.5 |
FEF25-75% |
| % predicted 61.2 78.2 49.0 |
Patients |
| requiring hospitalization due |
to pulmonary exacerbation within past 44% 20% 43%
12 months (%)
% Patients taking common CF medications:
TOBIR(inhaled) |
| 0% 0% 44% |
Pulmozyme® |
| 52% 53% 64% |
Azithromycin |
| 15% 2% 42% |
*Pulmozyme is a registered trademark of Genentech; TOBI is a registered trademark of Chiron
Inspire R&D Investor Meeting – March 21, 2006

Safety Summary Across Phase 2 Trials
Denufosol (up to 60 mg) was generally well tolerated, especially in CF patients with less severe lung function impairment Cough was most frequently reported adverse event Slight transient decline in lung function was noted immediately following administration of placebo or denufosol but was generally resolved by 2 hours post-dose Systemic exposure to denufosol appeared to be minimal
Inspire R&D Investor Meeting – March 21, 2006

Phase 3 Program
TIGER : Transport of Ions to Generate Epithelial Rehydration
R&D Investor Meeting – March 21, 2006

Global Phase 3 Program: Initial Indication for Early Intervention
Tiger-1 trial – targeting mid-year 2006 initiation Tiger-2 trial – 1st Half 2007 projected start
One 2-year carcinogenicity study required (3 years to complete) Patients with early CF lung disease – FEV1> 75% predicted
Primary endpoint of clinical trials expected to be FEV1
Anticipated secondary endpoints include pulmonary exacerbations, other lung function parameters and quality of life Age > 5 years and standard approved meds allowed—Pulmozyme® and/or TOBI ®
Will tightly manage patients on TOBI by initiating and ending during off cycle Robust pharmacoeconomic and Quality of Life data analyses planned
Inspire R&D Investor Meeting – March 21, 2006
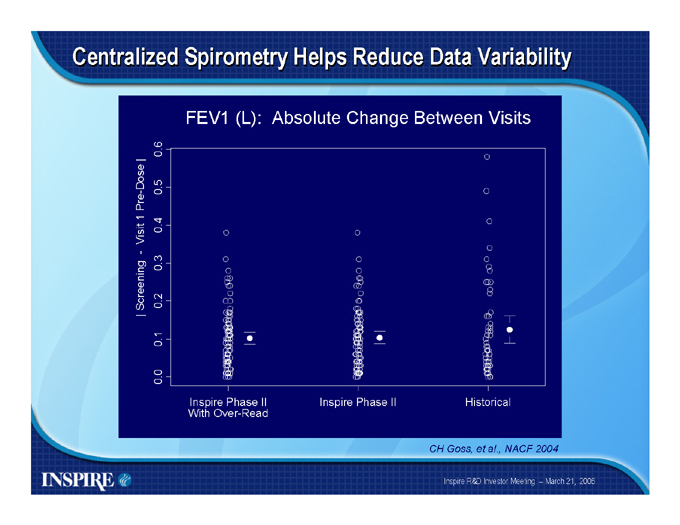
Centralized Spirometry Helps Reduce Data Variability
FEV1 (L): Absolute Change Between Visits
| Screening—Visit 1 Pre-Dose |
0.0 0.1 0.2 0.3 0.4 0.5 0.6
Inspire Phase II With Over-Read
Inspire Phase II
Historical
CH Goss, et al., NACF 2004
Inspire R&D Investor Meeting – March 21, 2006
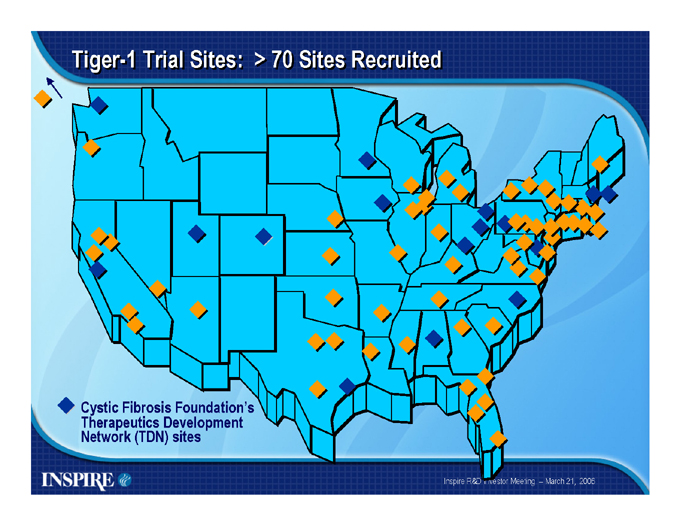
Tiger-1 Trial Sites: > 70 Sites Recruited
Cystic Fibrosis Foundation’s Therapeutics Development Network (TDN) sites
Inspire R&D Investor Meeting – March 21, 2006

Summary
Denufosol tetrasodium for Cystic Fibrosis
First in class, novel compound from Inspire’s discovery efforts
Strong intellectual property with composition of matter patent through 2017 Attractive market opportunity with existing CF lung therapies generating
> $500 mm in annual revenues*
Plan to commercialize in North America with internal resources
Initiating Phase 3 program
Conducted Tiger-1 Investigator Meeting Submitted Tiger-1 trial protocol to FDA Carcinogenicity study protocol in development
Working with CFF and TDN to generate awareness of trials Presenting Phase 2 data at European CF Meeting in June
Collaborating with EMEA to discuss European development strategy
Continuing partnering discussions for ex-North America commercialization
*Information is for 12 months ended 9/30/05; Source: IMS National Prescription Audit data
Inspire R&D Investor Meeting – March 21, 2006

Overview of Allergic Rhinitis
William E. Berger, MD, MBA
Clinical Professor Department of Pediatrics
Division of Allergy and Immunology University of California, Irvine

Disclosure
The presenter has served as a consultant and clinical investigator for Inspire. The presenter does not own stock or stock options in Inspire Pharmaceuticals.

Allergic Rhinitis: Definition
Symptomatic disorder of the nose induced by an IgE-mediated inflammation after allergen exposure of the membranes lining the nose; “hay fever”
Seasonal allergic rhinitis (SAR) 1-3
Intermittent
Patients may be referred to as “sneezers and runners”
Perennial allergic rhinitis (PAR) 1-3
Persistent
Patients may be referred to as “blockers”
1. Bousquet J, et al; Aria Workshop Group; WHO. J Allergy Clin Immunol. 2001;108(pt 2 no 5 suppl):S147-S334. 2. Skoner DP. J Allergy Clin Immunol. 2001;108(1 suppl):S2-8. 3. Khanna P, Shah A. Ann Allergy Asthma Immunol. 2005;94:60-64.

Rhinitis: Classification
Classification
Non-inflammatory (rhinitis medicamentosa, hormonal, vasomotor)
Structural (septal deviation, neoplasia, foreign body, trauma, adenoids, choanal atresia) Inflammatory
Non-allergic (NARES, infectious, polyps, associated with- systemic disease) Allergic (SAR, PAR)
NARES=non-allergic rhinitis with eosinophilia syndrome; SAR=seasonal allergic rhinitis; PAR=perennial allergic rhinitis. Bousquet J, et al: Aria Workshop Group; WHO. J Allergy Clin Immunol. 2001;108(pt 2 no 5 suppl):S147-S334.

Allergic Rhinitis: Widespread Impact
More than 50 million people suffer from allergic diseases in the US 1 Allergies are the 6th leading cause of chronic disease in the US 1 AR is the 2nd leading cause of chronic disease in the US2 AR affects 35 million Americans annually 2
10% to 30% of adults 33
Accounts for 3 million lost work days 22
Up to 40% of children 33
Accounts for 2 million lost school days 22
1. AAAAI. The Allergy Report: Science Based Findings on the Diagnosis & Treatment of Allergic Disorders, 1996–2001. 2. Strachan D, et al. Pediatr Allergy Immunol. 1997;8:161-176. 3. Dykewicz MS, et al. Ann Allergy Asthma Immunol. 1998;81:478-518.

Allergic Rhinitis: Widespread Impact
Approximately 14 million office visits to healthcare providers were attributed to allergic rhinitis in 20001 Morbidity of AR is significant Financial impact of AR is substantial
Productivity loss estimated at >$3 billion per year2 AR medication costs estimated at $6.3 billion in 20003
1. CDC. Fast Stats A-Z, Advanced Data from Vital and Health Statistics, no. 346, Table 13. August 26, 2004. At: http://www.cdc.gov/nchs/fastats/allergies.htm. 2. Rossoff LJ, et al. Am J Manag Care. 1997;3:309-315. 3. Retail and Provider 2000. IMS Health. (represents total manufacturer sales to pharmacies for AR products)

Link Between Allergic Rhinitis and Other Chronic Disorders
Comorbidities1,2
Asthma
Allergic rhinoconjunctivitis Sinusitis Otitis media
Complications1,3
Sleep-Disordered Otitis Media Breathing
Allergic Dentofacial Rhinitis Abnormalities
Asthma
Sinusitis
1. Spector SL, et al. J Allergy Clin Immunol. 1997;99:S773-S780.
2. O’Connell EJ. Allergy. 2004;78:7-11.
3. Rachelefsky GR. Ann Allergy Asthma Immunol 1998;82:1-10

Allergic Rhinitis: Therapeutic Approaches
Environmental control Treatments
Pharmacotherapy
Antihistamines (oral/topical) Decongestants Corticosteroids (oral/topical) Intranasal cromolyn Intranasal anticholinergics
Leukotriene receptor antagonists (LTRAs)
Allergen immunotherapy

Allergic Rhinitis: Role of Medications
Antihistamines
Sedating antihistamines* (chlorpheniramine, diphenhydramine)
Competitive antagonists of H 1 1-mediated effects
Relieve rhinorrhea, pruritus, sneezing, and ocular symptoms Oral, ocular, and intranasal preparations Use is limited by sedation, performance impairment, and anticholinergic effects
Nonsedating or low-sedating antihistamines † (azelastine, cetirizine, desloratadine, fexofenadine, loratadine, olopatadine)
Equally efficacious compared with sedating antihistamines Lack prominent CNS and anticholinergic effects Desloratadine, fexofenadine, loratadine, and olopatadine are nonsedating Cetirizine and azelastine are associated with mild sedation
*Sometimes referred to as first-generation antihistamines. †Sometimes referred to as second-generation antihistamines. Philip G, Togias A. J Respir Dis. 1999;20:580-588. Simons FE. N Engl J Med. 2004;351:2203-2217. Dykewicz MS, et al. Ann Allergy Asthma Immunol. 1998;81:463-468. Dykewicz MS. J Allergy Clin Immunol. 2003;111:S520-S529.

Allergic Rhinitis: Role of Medications
Decongestants: adrenergic agonists (oral: phenylephrine, pseudoephedrine; topical: phenylephrine, xylometazoline, oxymetazoline)
Stimulate receptors to induce local vasoconstriction
Decrease blood volume in the nasal mucosa capacitance vessels
Reduce blood supply to mucosa, decrease mucosal edema, improve nasal patency
Intranasal corticosteroids (beclomethasone, budesonide, flunisolide, fluticasone, mometasone, triamcinolone)
Affect inflammatory response by reducing number of eosinophils and mast cells
Intranasal cromolyn sodium1 (mast cell stabilizer)
Prevents release of prechemical and newly formed mediators to prevent degranulation
Dykewicz MS, et al. Ann Allergy Asthma Immunol. 1998;81:478-518.

Allergic Rhinitis: Role of Medications and Immunotherapy
Medications
Anticholinergic agents 1 (ipratropium bromide)
Inhibit parasympathetic transmission to submucosal glands Have low lipid solubility; do not cross blood-brain barrier
Leukotriene (LT) modifiers 2 (montelukast)
Inhibit LT production and/or block activity
Impact inflammatory cells recruited/activated by LTs
Immunotherapy
Allergen immunotherapy (allergy shots)
May decrease risk of developing asthma 3
May reduce symptoms and medication reliance on a long-term basis 44
1. Dykewicz MS, et al. Ann Allergy Asthma Immunol. 1998;81:478-518. 2. Philip G, et al. Clin Exp Allergy. 2002;32:1020-1028. 3. Valovirta E. J Invest Allergol Clin Immunol. 1997;7:369-370. 4. Dykewicz M, et al. Ann Allergy Asthma Immunol. 1998;81:478-518.
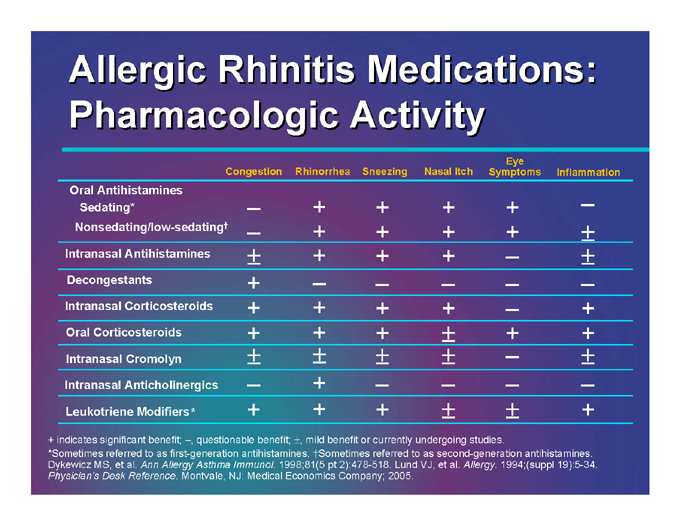
Allergic Rhinitis Medications: Pharmacologic Activity
Congestion |
| Rhinorrhea Sneezing Nasal Itch Eye Symptoms Inflammation |
Oral |
| Antihistamines |
Sedating* |
| – + + + + – |
Nonsedating/low-sedating |
| † – + + + + ± |
Intranasal |
| Antihistamines ± + + + – ± |
Decongestants |
| + – – – – – |
Intranasal |
| Corticosteroids + + + + – + |
Oral |
| Corticosteroids + + + ± + + |
Intranasal |
| Cromolyn ± ± ± ± – ± |
Intranasal |
| Anticholinergics – + – – – – |
Leukotriene |
| Modifiers* + + + ± ± + |
+ indicates significant benefit; –, questionable benefit; ±, mild benefit or currently undergoing studies.
*Sometimes referred to as first-generation antihistamines. †Sometimes referred to as second-generation antihistamines. Dykewicz MS, et al. Ann Allergy Asthma Immunol. 1998;81(5 pt 2):478-518. Lund VJ, et al. Allergy. 1994;(suppl 19):5-34. Physician’s Desk Reference. Montvale, NJ: Medical Economics Company; 2005.

Allergic Rhinitis: Emerging Therapies
Sublingual immunotherapy Anti-IgE therapy New topical nasal antihistamines New topical nasal corticosteroids

Epinastine Multi-Action
Inhibits histamine release
Inhibits release of inflammatory mediators including cytokines Binds to histamine receptors Inhibits neutrophil activation Inhibits eosinophil activation and cytokine release Inhibits mediator release from basophils
Amon U et al. Inflamm Res. 2000;49:112-116; Kamei C et al. Immunopharm Immunotox. 1992;14:191-205; Fugner A et al. Arzneimittelforschung. 1988;38:1446-1453; Matsushita N et al. Jpn J Pharmacol. 1998;78:11-22; Calder VL, Ajayi Y, Galatowicz G. IgE-stimulated cytokine production by human cord blood-derived and conjunctival mast cells: Effects of epinastine. ACAAI 2005; Ohmori K et al. Arzneimittelforschung. 1998;48:43-46; Koyhama T et al. Biochem Biophys Res Comm. 1997;230:125-128; Fukuishi N et al. Jpn J Pharmacol. 1995;68:449-452; Mita H, Shida T. Arzneimittelforschung. 1995;45:36-40.

Allergic Rhinitis: Expanding Awareness and Meeting Expectations
What physicians know…
How patients use their medications
Nasal steroids not used properly or consistently 64% forget to take their medications 33% felt drugs were ineffective
Patient concerns about time to onset of action, safety, cost, etc
What patients want from treatment…
Fast onset & long duration of action, targeted relief of TNSS Nonsteroidal, non-habit forming Few adverse effects, does not cause drowsiness Improved “real world” quality of life
TNSS=total nasal symptom score. Gallup Poll, 2005.
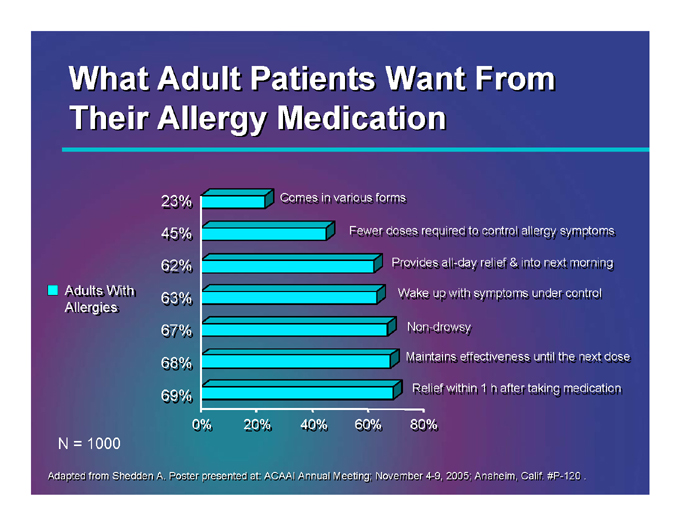
What Adult Patients Want From Their Allergy Medication
Adults With Allergies
23% 45% 62% 63% 67% 68% 69%
Comes in various forms
Fewer doses required to control allergy symptoms
Provides all-day relief & into next morning all-
Wake up with symptoms under control
Non-drowsy
Maintains effectiveness until the next dose
Relief within 1 h after taking medication
0% 20% 40% 60% 80%
N = 1000
Adapted from Shedden A. Poster presented at: ACAAI Annual Meeting; November 4-9, 2005; Anaheim, Calif. #P-120.
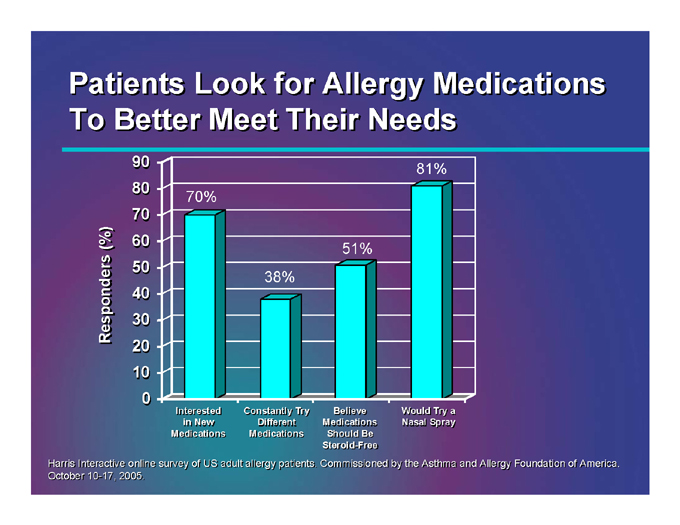
Patients Look for Allergy Medications To Better Meet Their Needs
Responders (%)
90 80 70 60 50 40 30 20 10 0
70%
38%
51%
81%
Interested in New Medications
Constantly Try Different Medications
Believe Medications Should Be Steroid-Free
Would Try a Nasal Spray
Harris Interactive online survey of US adult allergy patients. Commissioned by the Asthma and Allergy Foundation of America. October 10-17, 2005.
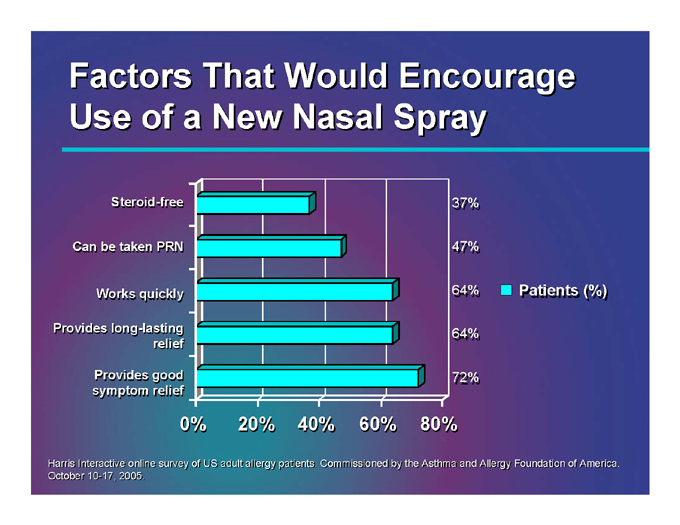
Factors That Would Encourage Use of a New Nasal Spray
Steroid-free
Can be taken PRN
Works quickly
Provides long-lasting relief
Provides good symptom relief
0% 20% 40% 60% 80%
37% 47% 64% 64% 72%
Patients (%)
Harris Interactive online survey of US adult allergy patients. Commissioned by the Asthma and Allergy Foundation of America. October 10-17, 2005.

Summary
Emerging allergic rhinitis treatments focus on:
Meeting patient needs:
Rapid onset, long-lasting & convenient dosing Safety and cost-effectiveness Improving patient adherence/compliance
Meeting physician needs:
Providing newer, enhanced treatment choices that improve patient quality of life Immune tolerance Recognizing and treating comorbidities of AR

Thank You

Donald J. Kellerman, Pharm.D. Senior Vice President, Development Inspire Pharmaceuticals
Inspire R&D Investor Meeting – March 21, 2006
New Opportunity in Allergic Rhinitis with Proven Molecule
Licensed U.S. and Canadian rights for rhinitis from Boehringer Ingelheim Known molecule Well-characterized safety
Extensive patient experience with oral and ocular forms
College of Allergy presentation
Proven efficacy following topical administration Extensive pre-clinical and pharmacokinetics package available
H2N
N
x HCl
N
Inspire R&D Investor Meeting – March 21, 2006

Rationale for Nasal Antihistamines for Allergic Rhinitis
Clinician interest in having additional options for patients Topical nasal spray can provide quick relief to affected areas; reduced systemic side effects Allergic rhinitis development path is well-defined Large number of patients available to participate in trials
40-50 million patients suffer from nasal allergies*
Large population of experienced U.S. investigators
*Source: American Academy of Allergy, Asthma & Immunology
Inspire R&D Investor Meeting – March 21, 2006

Advantages of Direct Topical vs. Systemic Administration
Little or no systemic effects
Sedation, QT interval, alcohol interaction
Amount of drug administered is much less High local concentrations in the nose More effective in blocking triggers of rhinitis Potential for more rapid onset
Inspire R&D Investor Meeting – March 21, 2006
Well-Established Profile for Epinastine in Ocular Use
Prevents ocular allergy symptoms by
Blocking H1 and H2 receptors
Stabilizing mast cells
Stopping the progression of pro-inflammatory mediators
Important features
Low affinity for M3
Soothing upon instillation
Rapid relief and sustained duration
Good tolerability profile
Elestat is #2 in national weekly TRx market share*
*Information as of 3/03/06; Source: IMS National Prescription Audit data
Inspire R&D Investor Meeting – March 21, 2006

Types of Allergic Rhinitis Indications
Seasonal Allergic Rhinitis (SAR) -allergens seasonally present
Tree, grass, weed pollens
Perennial Allergic Rhinitis (PAR) -allergens always present
Animal dander, dust mites, molds
Non-allergic Rhinitis
Perfume, Cigarette smoke
Inspire R&D Investor Meeting – March 21, 2006

FDA Guidance on Development Pathway Well Defined
Written by current FDA Pulmonary Allergy Drug Product Division director Discusses extent of program required Discusses use in pediatric patients Double-blind, placebo-controlled, parallel group studies recommended Double-blind period – 2 weeks for SAR, 4 weeks for PAR Pollen counts at each of the study centers PAR trials done when seasonal pollens less abundant
Inspire R&D Investor Meeting – March 21, 2006

FDA Guidance on Development Pathway
Symptoms are primary efficacy endpoint Instantaneous and reflective symptoms
Rhinorrhea, nasal congestion, nasal itching, sneezing Patient symptoms – rated on Diary Cards
0 = absent 1 = mild symptoms 2 = moderate symptoms 3 = severe symptoms
Score at least as frequent as dosing interval
Inspire R&D Investor Meeting – March 21, 2006
Types of Allergic Rhinitis Trials
Traditional 2-week SAR
Ragweed
August 15—October 15, most of the U.S.
Mountain Cedar
December 15 – February 15, San Antonio – Austin
Trees and Grass
April – June, much of U.S.
Park setting study
Chamber study (Environmental Exposure Unit)
Good for both onset of effect and dosing interval
Not pivotal
Inspire R&D Investor Meeting – March 21, 2006

Substantial Experience in Allergy & Respiratory Programs
Development Related Expertise
Don Kellerman, Pharm.D. – Flonase®, Beconase®, Techastemizole Richard Evans, Ph.D. – Formulations of Nasacort Amy Schaberg, M.S., R.N – Flonase®, Beconase® Ramesh Krishnamoorthy, Ph.D. – Various formulations of nasal spray solutions
Marketing and Sales Related Expertise
Joe Schachle – Advair Diskus® Jeff Sampere – Flovent® Kim Morris – Flonase®
*Flonase, Beconase, Advair Diskus and Flovent are registered trademarks of GlaxoSmithKline; Nasacort is a registered trademark of Sanofi-Aventis
Inspire R&D Investor Meeting – March 21, 2006

Advantages of Intranasal Epinastine Program
Strategic expansion of respiratory/ allergy franchise Existing data package of pharmacological and toxicological testing and human experience Extensive internal development and scientific expertise for allergy products Straightforward regulatory pathway Experienced investigators and readily-available patient population for trials
Inspire R&D Investor Meeting – March 21, 2006

Summary
Intranasal Epinastine
Enhances focus in the respiratory and allergy therapeutic area Well-known molecule in defined market where Inspire has extensive experience Minimal initial cash investment to in-license asset Plan to commercialize in North America
Moving toward IND filing and Phase 2 program
Scheduled Pre-IND meeting with FDA in late May Submit IND filing to FDA
Finalize formulation and delivery system
Target initiation of first Phase 2 trial by year-end 2006
Plan to pursue various possible forms of intellectual property protection to support commercialization of intranasal epinastine product
Drug delivery, formulation, method of use
Inspire R&D Investor Meeting – March 21, 2006

Benjamin R. Yerxa, Ph.D.
Chief, Scientific Operations and Alliances Inspire Pharmaceuticals
Inspire R&D Investor Meeting – March 21, 2006

Overview of INS50589 Antiplatelet
Role of platelets in clotting and bleeding
Role of P2Y12 receptor in platelet function
INS50589
Chemistry and pharmacology Phase 1 results
Platelet dysfunction in cardiopulmonary bypass procedures Phase 2 program
Strategic partnering
Inspire R&D Investor Meeting – March 21, 2006
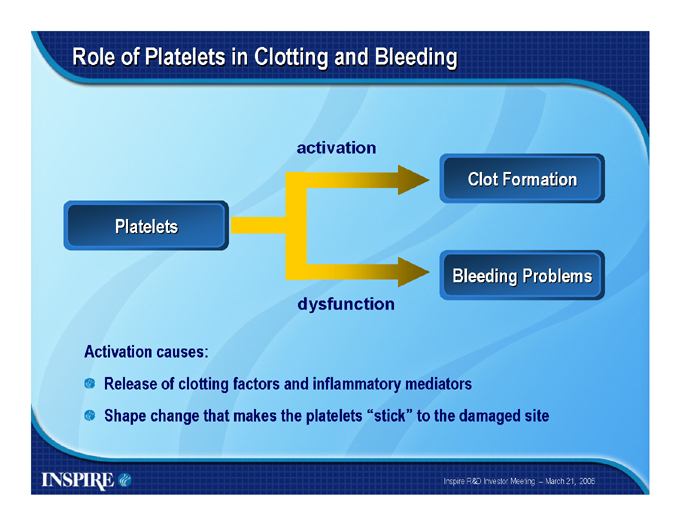
Role of Platelets in Clotting and Bleeding activation
Platelets
Clot Formation
Bleeding Problems dysfunction
Activation causes:
Release of clotting factors and inflammatory mediators
Shape change that makes the platelets “stick” to the damaged site
Inspire R&D Investor Meeting – March 21, 2006
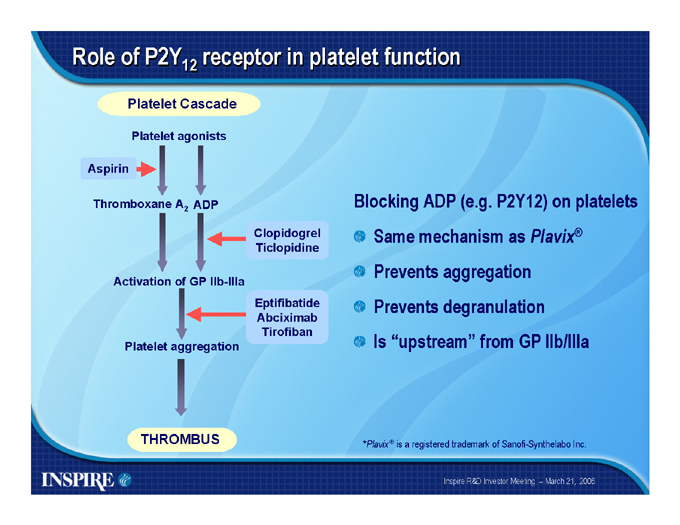
Role of P2Y12 receptor in platelet function
Platelet Cascade
Platelet agonists
Aspirin
Thromboxane A2 ADP
Clopidogrel Ticlopidine
Activation of GP IIb-IIIa
Eptifibatide Abciximab Tirofiban Platelet aggregation
Blocking ADP (e.g. P2Y12) on platelets Same mechanism as Plavix® Prevents aggregation Prevents degranulation Is “upstream” from GP IIb/IIIa
THROMBUS
*Plavix® is a registered trademark of Sanofi-Synthelabo Inc.
Inspire R&D Investor Meeting – March 21, 2006
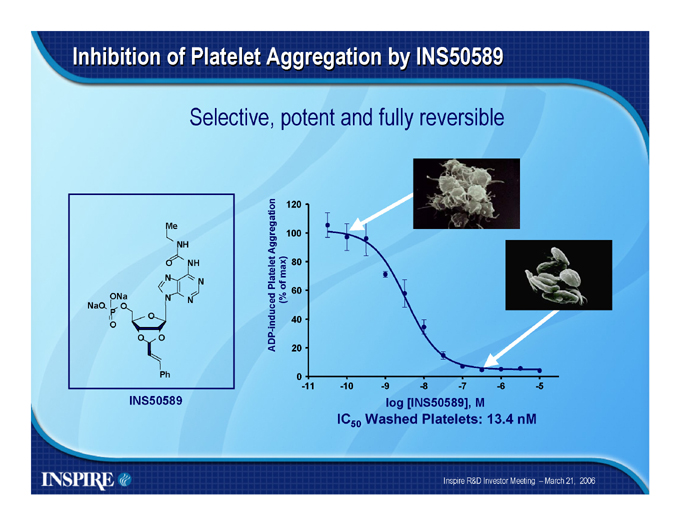
Inhibition of Platelet Aggregation by INS50589
Selective, potent and fully reversible
ADP- induced Platelet Aggregation (% of max)
120 100 80 60 40 20 0
-11 -10 -9 -8 -7 -6 -5 log [INS50589], M
IC50 Washed Platelets: 13.4 nM
Me
NH
O NH N N
ONa N N NaO O
P O O
O O
Ph
INS50589
Inspire R&D Investor Meeting – March 21, 2006

INS50589 Phase 1 Trial Objectives
Evaluate the safety and tolerability of a four-hour, continuous infusion of up to four doses of INS50589 in 36 healthy volunteers Evaluate the pharmacokinetics of INS50589 and its major metabolite(s) Evaluate the effects of INS50589 on platelet function Identify a pharmacologically effective dose of INS50589 for further clinical development
Inspire R&D Investor Meeting – March 21, 2006
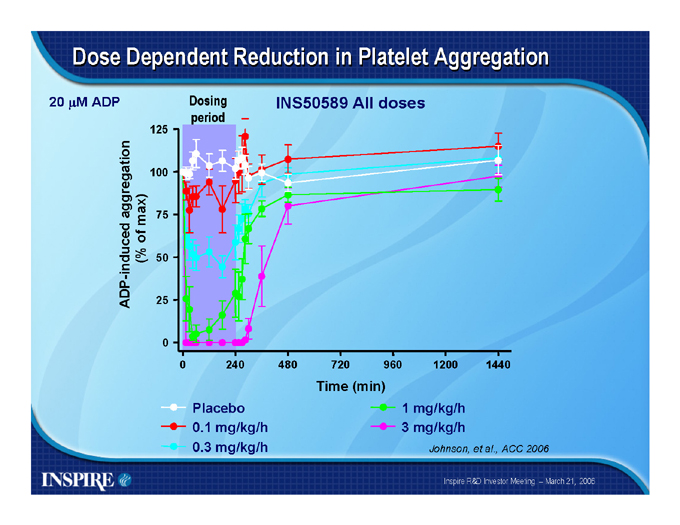
Dose Dependent Reduction in Platelet Aggregation
20 ?M ADP
ADP- induced aggregation (% of max)
125 100 75 50 25 0
Dosing period
INS50589 All doses
0 240 480 720 960 1200 1440
Time (min)
Placebo 0.1 mg/kg/h 0.3 mg/kg/h
1 mg/kg/h 3 mg/kg/h
Johnson, et al., ACC 2006
Inspire R&D Investor Meeting – March 21, 2006
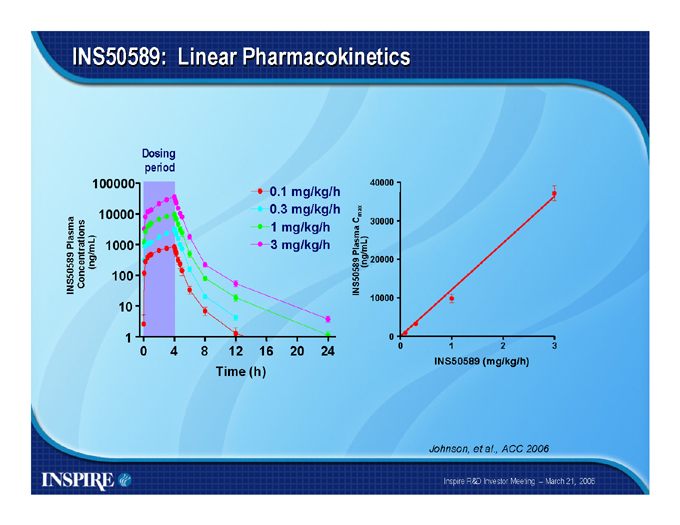
INS50589: Linear Pharmacokinetics
INS50589 Plasma Concentrations (ng/mL)
100000
10000
1000 100 10 1
Dosing period
0 4 8 12 16 20 24
Time (h)
0.1 mg/kg/h 0.3 mg/kg/h 1 mg/kg/h 3 mg/kg/h
INS50589 Plasma Cmax (ng/mL)
40000 30000 20000 10000 0
0 1 2 3
INS50589 (mg/kg/h)
Johnson, et al., ACC 2006
Inspire R&D Investor Meeting – March 21, 2006

Phase 1 Trial Conclusions
INS50589 was well-tolerated, no treatment-related serious adverse effects were observed and all subjects dosed completed the study Intravenous administration of INS50589 produced a rapid and reversible dose-related inhibition of platelet aggregation Results may support use where a rapid and strict control of platelet function is required
Inspire R&D Investor Meeting – March 21, 2006
Potential Clinical Indications
Cardiovascular surgery involving cardiopulmonary bypass: Coronary Artery Bypass Graft (CABG) and valve replacements Acute Coronary Syndrome (ACS) Percutaneous Coronary Interventions (PCI) Stroke Clinical conditions where blood is exposed to artificial surfaces such as artificial heart, lung, renal dialysis, apheresis, etc.
Pediatric cardiovascular surgery
Inspire R&D Investor Meeting – March 21, 2006

Lead Clinical Program: Cardiopulmonary Bypass
In the U.S., approximately 450,000 open heart surgeries expected to be performed in 2006* Clinical adverse effects associated with CPB include:
Bleeding complications
Excessive post-operative blood loss requiring transfusion Embolism Production of vasoactive and inflammatory substances Stroke and neurocognitive disorders
CPB development program
Smaller than ACS or PCI
Less crowded area
*Source: Defined Health report
Inspire R&D Investor Meeting – March 21, 2006
Normal Blood Vessel
Inspire R&D Investor Meeting – March 21, 2006
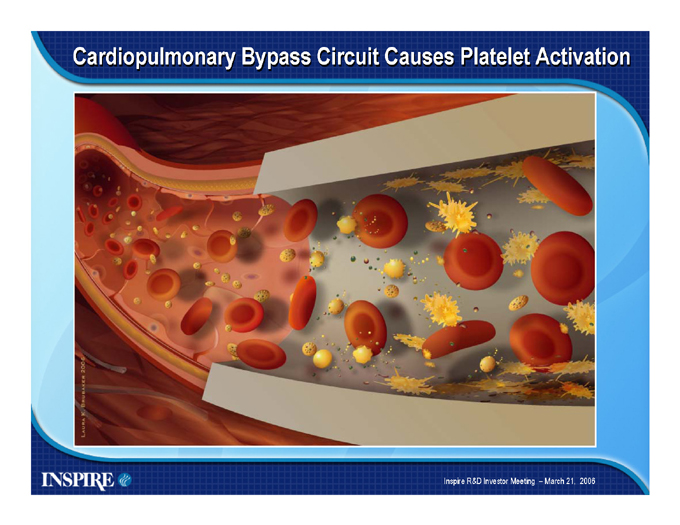
Cardiopulmonary Bypass Circuit Causes Platelet Activation
Inspire R&D Investor Meeting – March 21, 2006
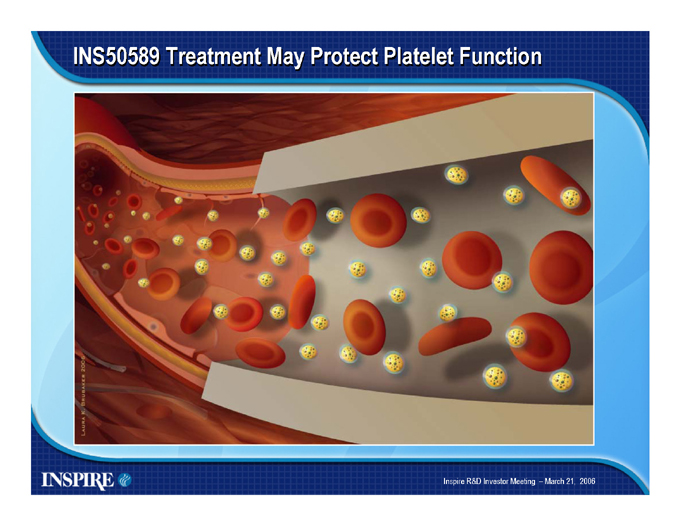
INS50589 Treatment May Protect Platelet Function
Inspire R&D Investor Meeting – March 21, 2006
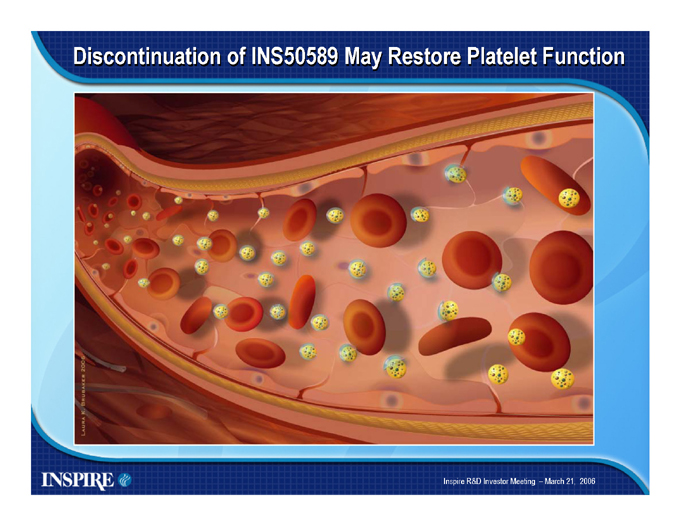
Discontinuation of INS50589 May Restore Platelet Function
Inspire R&D Investor Meeting – March 21, 2006
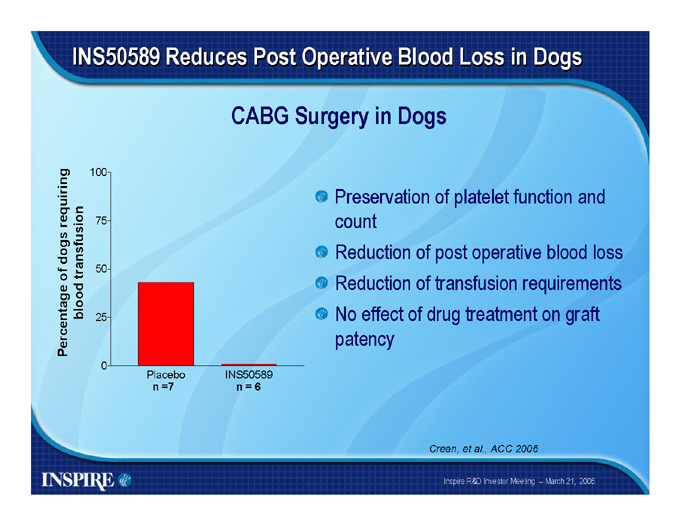
INS50589 Reduces Post Operative Blood Loss in Dogs
CABG Surgery in Dogs
Percentage of dogs requiring blood transfusion
100 75 50 25 0
Placebo n =7
INS50589 n = 6
Preservation of platelet function and count Reduction of post operative blood loss Reduction of transfusion requirements No effect of drug treatment on graft patency
Crean, et al., ACC 2006
Inspire R&D Investor Meeting – March 21, 2006

INS50589 Phase 2 Trial
Randomized, Multicenter, Placebo-Controlled Double-Blind, Parallel Group Study of INS50589 Intravenous Infusion in Subjects Undergoing Coronary Artery Bypass Graft Surgery Involving Cardiopulmonary Bypass
Primary Objective:
To identify a dose(s) of INS50589 that is well tolerated and significantly decreases postoperative blood loss following surgery Targeting approximately 160 subjects at 20 U.S. sites
Inspire R&D Investor Meeting – March 21, 2006
Strategic Partnering Discussions are Ongoing
Actively seeking partnerships for:
INS50589 licensing globally
Will consider regional carve outs for the right partner Orally bioavailable candidates included or separate
Partnering during Phase 2 allows us to:
Continue to build value while simultaneously seeking partnerships Stay focused long term on core therapeutic areas
Inspire R&D Investor Meeting – March 21, 2006

Summary
INS50589
Potent, selective and reversible P2Y12 antagonist
Fast on and fast off kinetics
Initial use is in cardiopulmonary bypass surgeries Many other potential indications to explore Plan to commercialize through partnership
Phase 2 starting
Initiating dose-ranging Phase 2 trial in CABG with CPB in Q2 2006 Blood loss endpoint
Inspire R&D Investor Meeting – March 21, 2006

INSPIRE