 A Phase 3 Study of Denufosol for A Phase 3 Study of Denufosol for Cystic Fibrosis: Cystic Fibrosis: TIGER-1 Results TIGER-1 Results Lead PI TIGER-1 Lead PI TIGER-1 1 Frank Accurso, MD Frank Accurso, MD Professor of Pediatrics Professor of Pediatrics Cystic Fibrosis Center Director Cystic Fibrosis Center Director The Children’s Hospital The Children’s Hospital University of Colorado Denver University of Colorado Denver Transport of Transport of Generate Generate Ions to Ions to Epithelial Epithelial Rehydration Rehydration Exhibit 99.2 |
 2 Cystic Fibrosis Therapies Significant need for agent that addresses basic defect of ion transport prior to onset of infection and inflammation: Antibiotic Mucus Regulation Anti- Inflammatory Gene Therapy, Potentiator /Corrector Lung structural damage CFTR defect Thickened airway secretions Bacterial colonization Irreversible inflammation Lung Transplant Inefficient ion transport Inefficient ion transport Ion Transport Restoration denufosol P2Y 2 Receptor Activation Inhaled P2Y 2 receptor agonists increase mucosal hydration and the natural mechanism of mucociliary clearance in the lung sodium absorption chloride ion and fluid secretion cilia beat frequency mucin secretion from goblet cells surfactant release from type II alveolar cells |
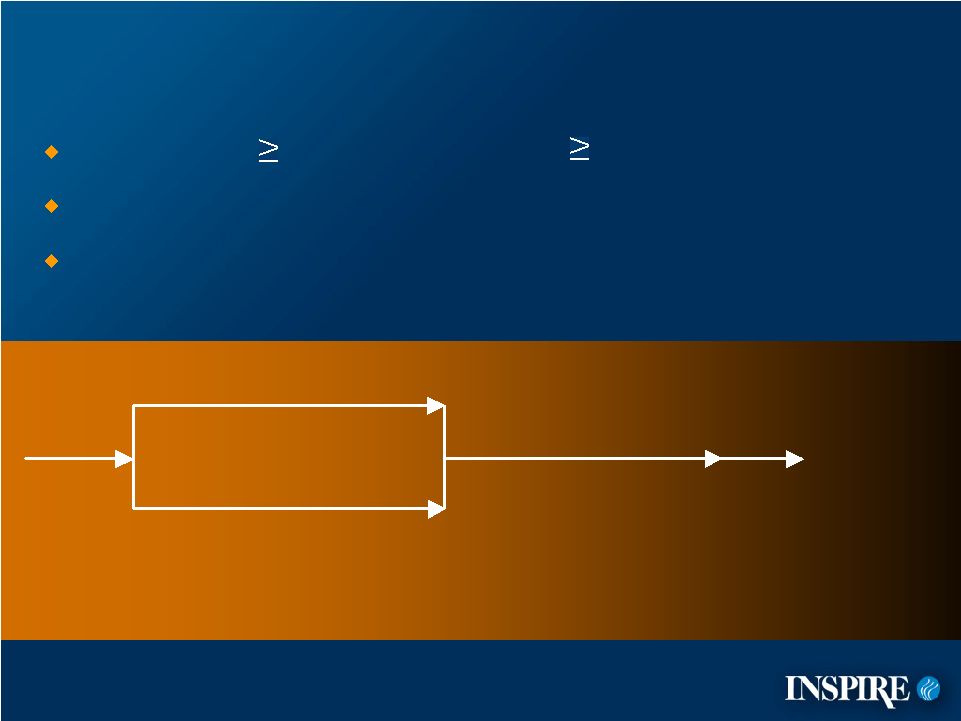 3 TIGER-1 Clinical Study Design CF patients 5 years with FEV 1 75% predicted Standard of care allowed 352 CF patients randomized at 62 treatment centers denufosol 60 mg TID (n=314) period treatment period 24-week safety extension period 1-week post-treatment follow-up placebo TID (n=174) Placebo-controlled 24-week Screening denufosol 60 mg TID (n=178) |
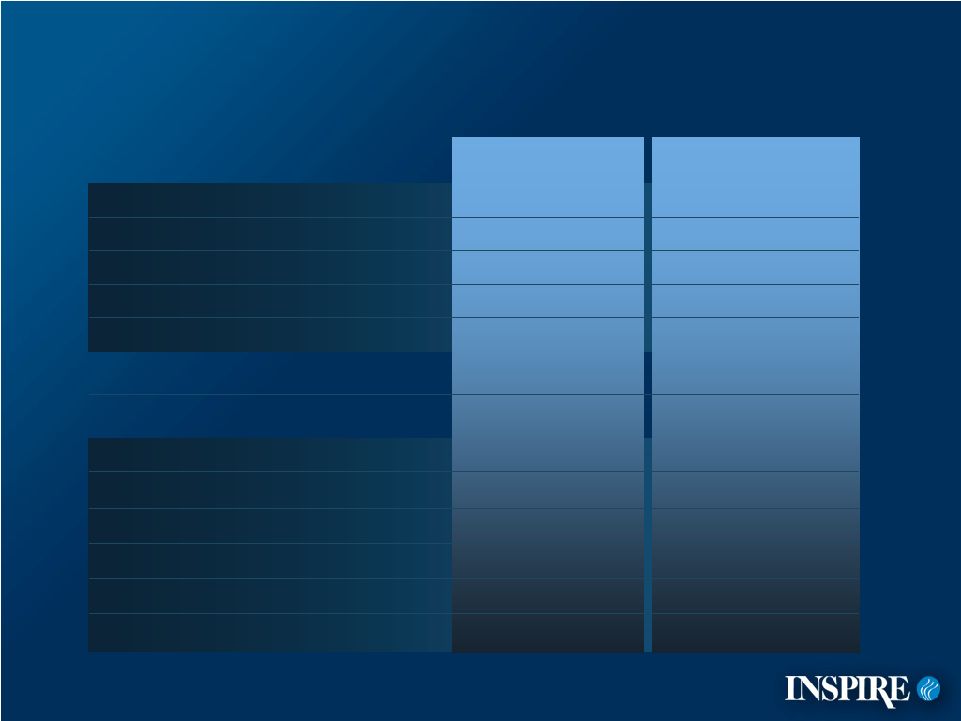 4 Demographic and Background Characteristics (Intent to Treat Population: ITT*) 89% 95% Pancreatic enzymes 82% 79% Bronchodilators 39% 40% Chronic macrolides 37% 38% Chronic inhaled antibiotics 77% 77% Dornase alfa 42% 48% PA + status 81% 84% FEF 25%-75% % Predicted 93% 92% FEV 1 % Predicted 54% 49% % Males 38 (21%) 33 (19%) 19 n (%) 59 (33%) 64 (37%) 12-18, n (%) 81 (46%) 77 (44%) 5-11, n (%) 14.3 14.9 Mean age, yrs Denufosol (n=178) Placebo (n=174) *ITT=All patients randomized |
 5 Disposition by Treatment Group 5 (3%) 2 (1%) Discontinued from placebo-controlled phase due to an AE, n (%) 19 (11%) 18 (10%) Discontinued early: placebo-controlled phase, n (%) 159 (89%) 156 (90%) Completed through Week 24, n (%) 178 (100%) 174 (100%) Intent-to-treat population, n (%) 178 174 Randomized, n Denufosol Placebo |
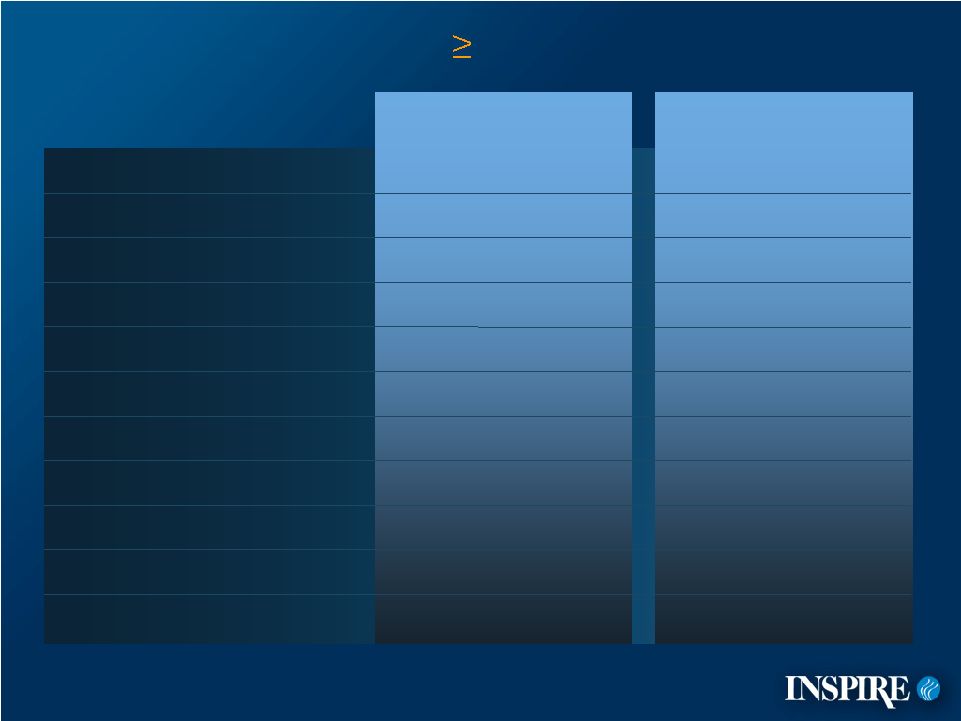
 6 Primary Efficacy Results – Placebo–Controlled (ITT) 0 10 20 30 40 50 60 70 Placebo Denufosol Met primary efficacy endpoint: change in FEV 1 from baseline to endpoint* 45 mL treatment effect (p = 0.047) “Real world” design Broader population Patients on multiple therapies that improve FEV 1 3 48 *Endpoint is the Week 24 value or the last observation carried forward for patients who withdrew early. |
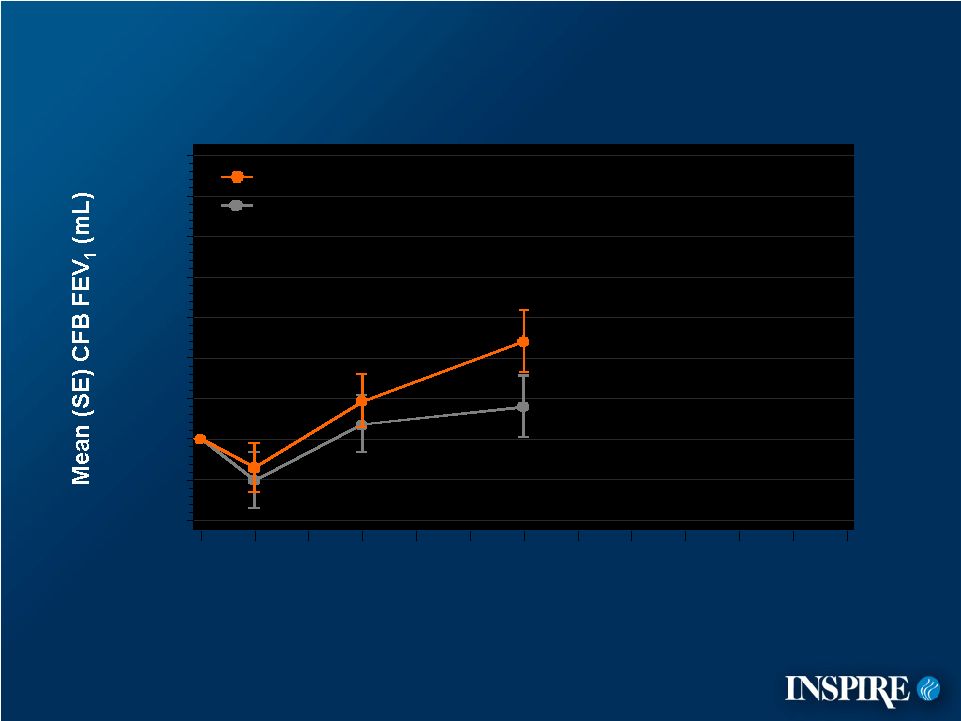 7 -40 -20 0 20 40 60 80 100 120 140 Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population Placebo Denufosol Treatment Week 0 4 12 24 28 36 48 |
 8 -40 -20 0 20 40 60 80 100 120 140 Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population Placebo Denufosol Treatment Week 0 4 12 24 28 36 48 |
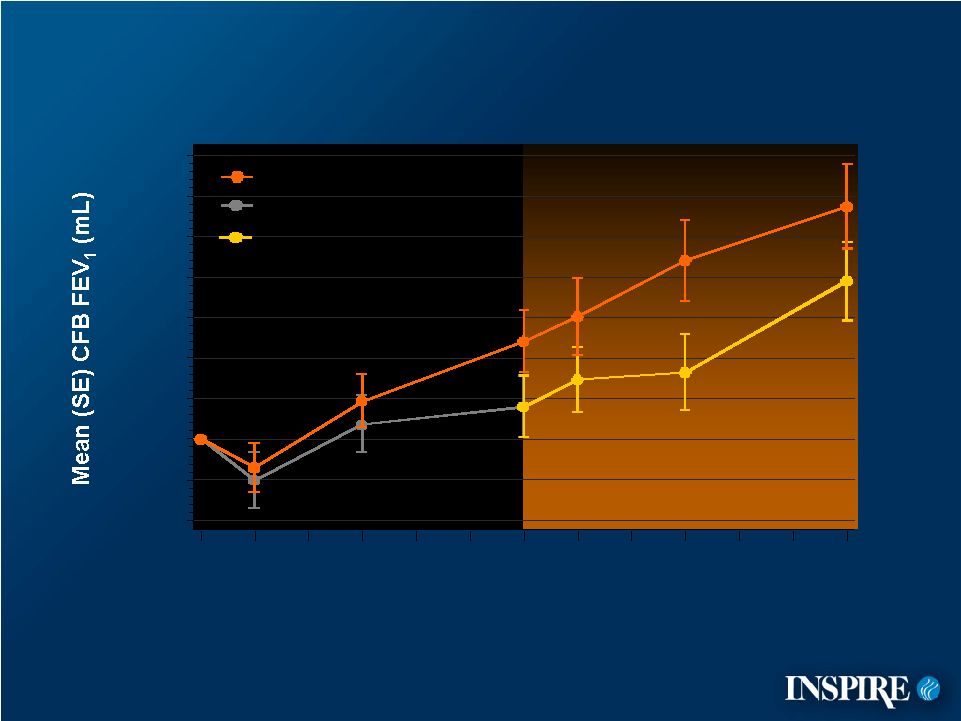 9 -40 -20 0 20 40 60 80 100 120 140 0 4 12 24 28 36 48 Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population Treatment Week Placebo Denufosol Placebo Switched to Denufosol denufosol-only, open-label phase |
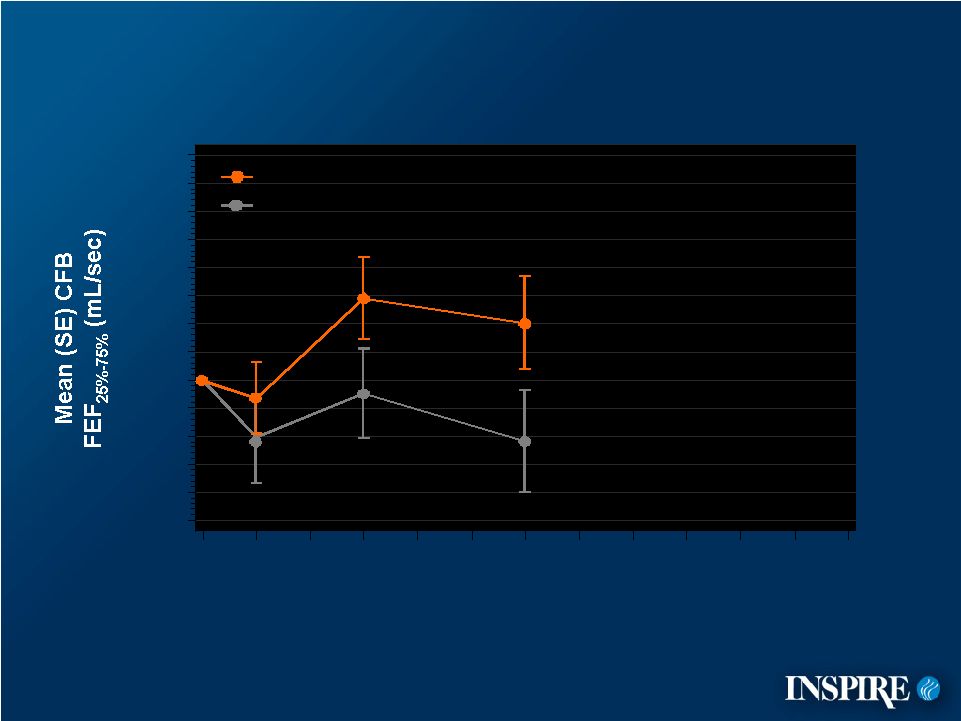 10 Placebo Denufosol Mean Change from Baseline FEF 25%-75% -100 -80 -60 -40 -20 0 20 40 60 80 100 120 140 160 0 4 12 24 28 36 48 CFB=Change from Baseline ITT Population Treatment Week |
 11 Placebo Denufosol Mean Change from Baseline FEF 25%-75% -100 -80 -60 -40 -20 0 20 40 60 80 100 120 140 160 0 4 12 24 28 36 48 CFB=Change from Baseline ITT Population Treatment Week |
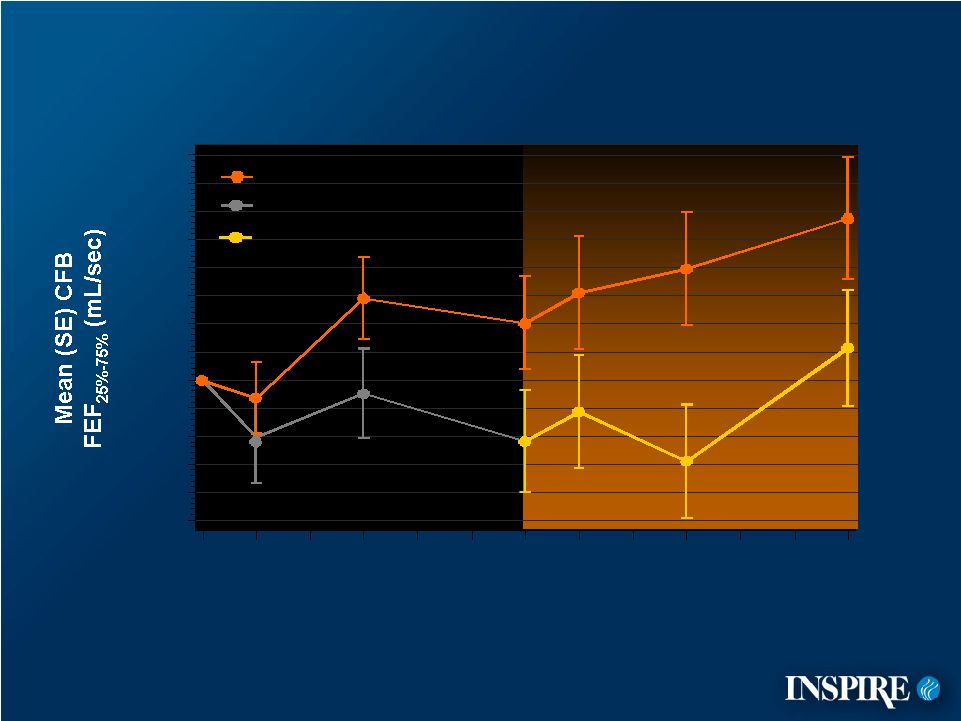 12 Placebo Denufosol Placebo Switched to Denufosol Mean Change from Baseline FEF 25%-75% -100 -80 -60 -40 -20 0 20 40 60 80 100 120 140 160 0 4 12 24 28 36 48 denufosol-only, open-label phase CFB=Change from Baseline ITT Population Treatment Week |
 13 0 20 40 60 80 100 120 140 160 180 Effect of Denufosol in ITT Population and Patients 110% Predicted 56 45 ITT p-value = 0.047 (n=352) 0 20 40 60 80 100 120 140 160 180 110% Predicted p-value = 0.015 (n=329) 112 88 ITT p-value = 0.072 (n=352) 110% Predicted p-value = 0.025 (n=329) FEV 1 FEF 25%-75% |
 14 Secondary Efficacy Endpoint: Pulmonary Exacerbation Incidence as expected in this patient population (i.e. low) No significant difference between groups May be confounded by denufosol’s pharmacology Longer treatment time may be needed |
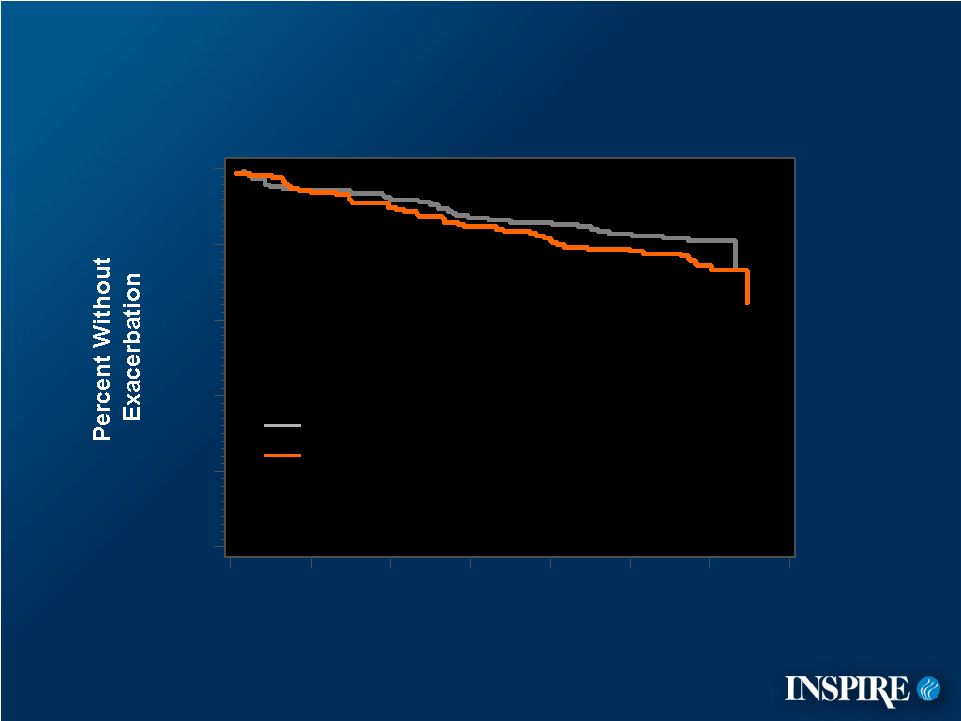 15 Pulmonary Exacerbations (Fuchs’ Criteria) During Placebo–Controlled Phase 0 20 40 60 80 100 0 4 8 12 16 20 24 28 Placebo Denufosol 60 mg Log rank statistic = 2.379 p-value = 0.123 Time (Weeks) ITT Population |
 16 Hospitalizations/Missed School and Work During Placebo–Controlled Phase 3.6 (7.7) 13 (7.3%) Denufosol (n=178) 0.610 3.4 (7.9) Mean Days of CF-Related Missed School or Work (SD) 0.843 14 (8.0%) Hospitalizations/ ER visits for respiratory complaint, n (%) P-value Placebo (n=174) ITT Population |
 17 Cystic Fibrosis Questionnaire – Respiratory Subscale -3.08 (15.43) -1.48 (16.74) CFB Week 24 81.4 (14.56) (n=173) Mean (SD) Denufosol 79.3 (15.98) Baseline (n=162) Mean (SD) CFQ - Respiratory Placebo ITT Population |
 18 High Level of Compliance with TID Dosing Compliance during the placebo-controlled phase ~90% of patients were compliant - Comparable between denufosol and placebo Average number of daily doses = 2.7 Excellent patient retention throughout study 352 patients were randomized ~90% completed the 24-week placebo-controlled phase ~99% of those who completed the placebo-controlled phase elected to continue into the open-label extension |
 19 Most Common AEs ( 10% in any group) 20 (11%)* 43 (25%) Headache 36 (20%) 27 (15%) Pyrexia 61 (34%) 51 (29%) Pulmonary exacerbation 15 (8%) 23 (13%) Pseudomonas infection 17 (10%)* 31 (18%) Sinusitis 29 (16%) 30 (17%) Productive cough 18 (10%)* 31 (18%) Rhinorrhea 34 (19%) 32 (18%) Pharyngolaryngeal pain 26 (15%) 34 (19%) Nasal congestion 96 (54%) 103 (59%) Cough 165 (93%) 169 (97%) Any AE, n (%) Denufosol (n=177) Placebo (n=175) *p-value <0.05 vs placebo Safety Population |
 20 No Difference in Growth Between Groups 45.3 44.5 Baseline 0.922 1.01 1.03 CFB at 6 months 19.8 0.03 147.5 1.90 Denufosol (n=177) 0.769 19.3 0.07 BMI, kg/m Baseline CFB at 6 months Weight, kg 0.871 148.1 1.83 Height, cm Baseline CFB at 6 months P-value Placebo (n=175) Mean Change from Baseline (CFB) Safety Population 2 |
 21 Safety Overview Safety and tolerability profile - favorable Relatively few SAEs and withdrawals due to AEs AE profile Comparable between treatment groups SAEs comparable between treatment groups Placebo: 14 (8%) Denufosol: 16 (9%) No evidence of systemic adverse effects (labs, vital signs) Minimal to no systemic absorption following dosing No significant effect on growth (height/weight/BMI) |
 22 TIGER-1 Summary Large multicenter, randomized, double-blind, placebo-controlled trial Used with other therapies No significant interactions with concomitant medications Significant improvement in FEV 1 Comparable exacerbation rates Well-tolerated Good adherence to TID regimen |
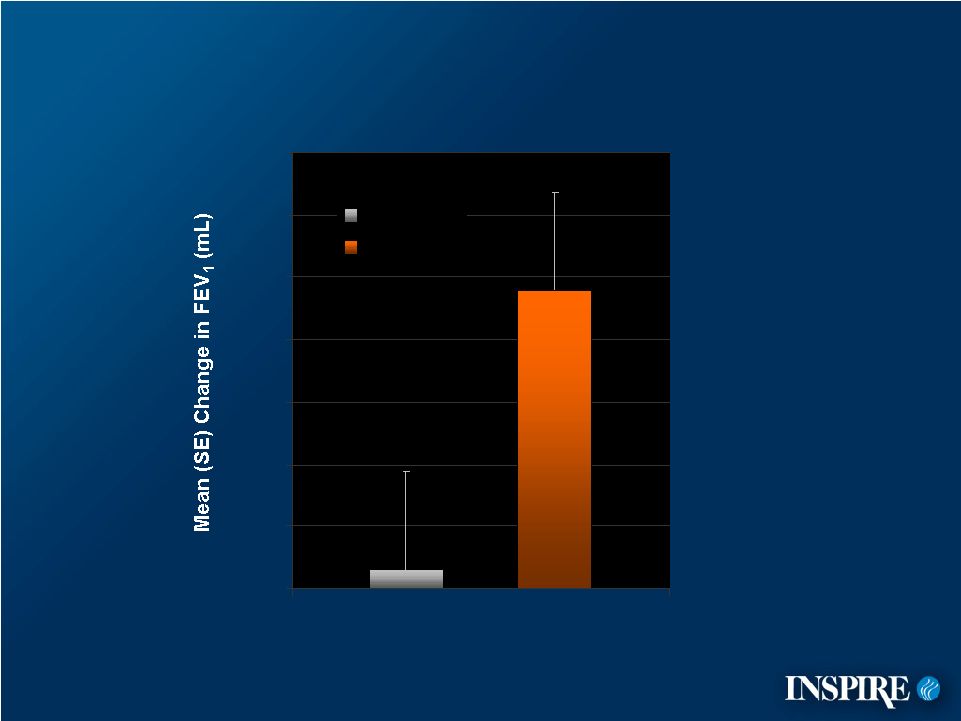 23 0 10 20 30 40 50 60 70 Placebo Denufosol Primary Efficacy Results – Placebo–Controlled (ITT) Endpoint is the Week 24 value or the last observation carried forward for patients who withdrew early. |
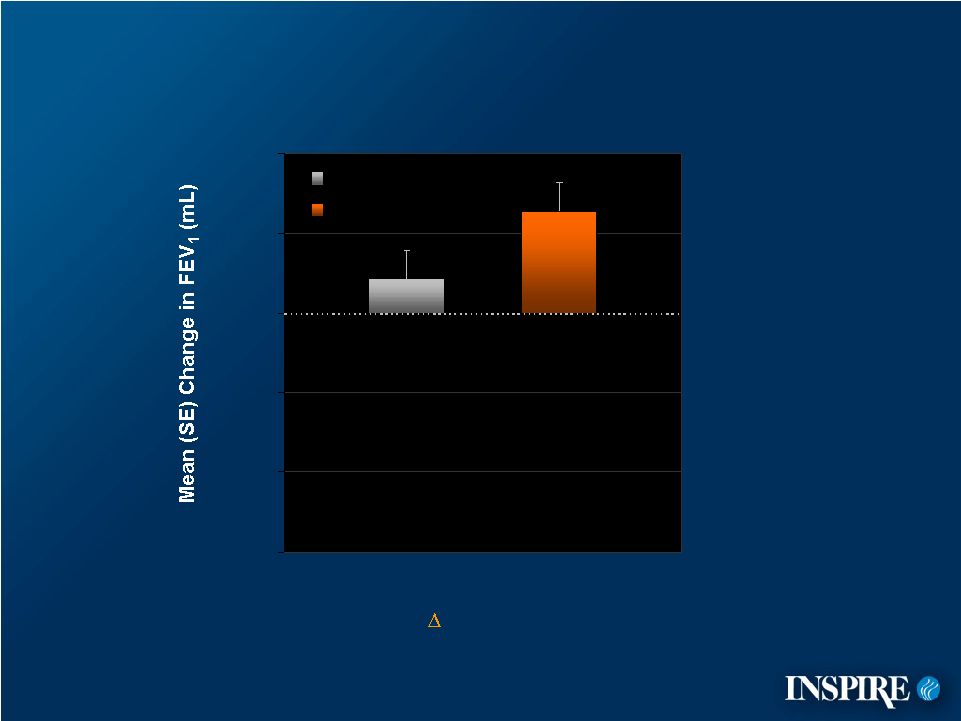 24 -150 -100 -50 0 50 100 = 41 mL p-value = 0.101 Subgroup: FEV in Patients Who Did NOT Experience an Exacerbation (Fuchs’ Criteria) n=141 (81%) n=131 (74%) Placebo Denufosol 1 |
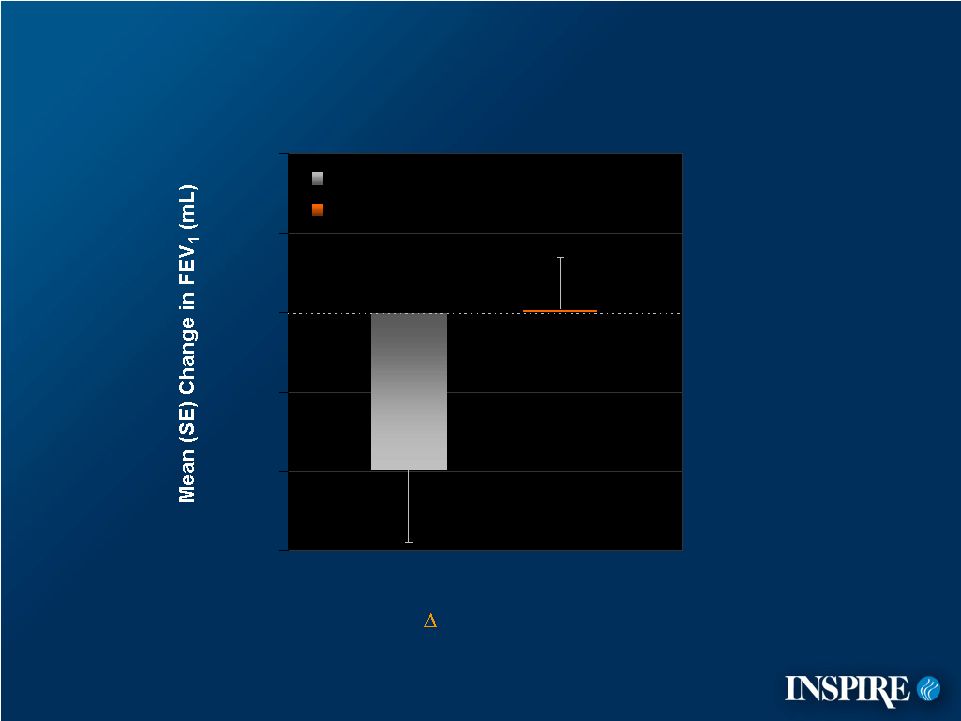 25 -150 -100 -50 0 50 100 Subgroup: FEV in Patients Who Did Experience an Exacerbation (Fuchs’ Criteria) n=33 (19%) n=47 (26%) = 101 mL p-value = 0.062 Placebo Denufosol 1 |
 26 Projected Long-term Impact of Slowing the Rate of Decline Konstan., Pediatr Pulmonol., 2008. 43, S24-28 |
 27 TIGER-2 Study Design Current Status ~100 sites in North America, Australia and New Zealand As of October 17, 133 patients randomized at 41 sites TIGER-2 Study Design Primary outcome: FEV 1 48 weeks; placebo-controlled Planned sample size n=~450 Open-label extension to follow |
 28 Acknowledgements Cystic Fibrosis Foundation CFFT Therapeutics Development Network CFFT Data Monitoring Committee TIGER-1 Principal Investigators and Research Coordinators |