 UBS Global Life Sciences Conference September 22, 2009 Exhibit 99.1 |
 2 Forward-Looking Statements This presentation contains forward-looking statements as defined by the Private Securities Litigation Reform Act of 1995, which convey management's expectations, beliefs, plans and objectives regarding future performance. These statements discuss matters that are not facts, and may include words to indicate their uncertain nature such as "believe," "expect," "anticipate," "intend," "plan," "estimate," "project," and “target." Forward-looking statements are subject to significant risks and uncertainties that could affect such future performance including those relating to product development, revenue, expense and earnings expectations, intellectual property rights, adverse litigation developments, competitive products, results and timing of clinical trials, success of marketing efforts, the need for additional research and testing, delays in manufacturing, funding, and the timing and content of decisions made by regulatory authorities, including the U.S. Food and Drug Administration. Actual results could differ materially from those described in this presentation. Information on various factors that could affect the information contained in this presentation and Inspire’s performance are detailed in the reports we file with the SEC. |
 3 Robust Pipeline with Multiple Catalysts * Received two approvable letters from FDA ** Currently approved for the treatment of bacterial conjunctivitis *** Inspire 2009 guidance reaffirmed on 07/31/09 Innovative Late-Stage Pipeline Extensive Intellectual Property R&D and Commercial Capabilities $80-90 mm Revenue Stream*** Core Technology P2Y receptor platform; role in the treatment of airway and ocular disease Over 80 U.S. patents Focused on two therapeutic areas: ophthalmology and pulmonology Multiple Novel Programs Denufosol for cystic fibrosis in Phase 3 Prolacria for dry eye in Phase 3* for blepharitis in Phase 2** Three Ophthalmology Products for bacterial conjunctivitis for prevention of itching associated with allergic conjunctivitis for dry eye TM 2 |
 4 Novel Clinical Pipeline * NDA filed and received two approvable letters from FDA ** Currently approved for the treatment of bacterial conjunctivitis Pre-Clinical Phase 1 Phase 2 Phase 3 Prolacria for dry eye * Denufosol for cystic fibrosis AzaSite for blepharitis ** INS115644 for glaucoma INS117548 for glaucoma TM ® |
 5 Cystic Fibrosis: Life Threatening and Underserved CF is caused by a genetic mutation that disrupts the CFTR protein, an ion channel, and leads to poorly hydrated lungs and severely impaired mucociliary clearance ~30,000 U.S. patients; 70,000 worldwide* Life expectancy: ~37 yrs* Mean FEV 1 % predicted is 75.9* 55% of CF population is less than 18 years old* 90% of CF patients die of respiratory failure * Cystic Fibrosis Foundation Patient Registry 2007 Annual Data Report ** All information is for 12 months ended 12/31/08; Source: IMS Disease Quick Facts Current CF Lung Disease Treatment Options TOBI - inhaled antibiotic for Pseudomonas aeruginosa - $238 mm in U.S. sales** Pulmozyme - inhaled enzyme that thins and loosens mucus - $302 mm in U.S. sales** Chest physiotherapy No existing approved drug targets CF ion channel defect Denufosol is latest stage ion channel modulator in development and is not limited to a specific genetic mutation ® ® |
 6 Denufosol’s Mechanism of Action Restores Mucosal Hydration and Mucociliary Clearance in CF CFTR Defect Inefficient Ion Transport Thickened Airway Secretions Bacterial Colonization Irreversible Inflammation Lung Structural Damage Gene Therapy, Potentiator/ Corrector Ion Transport Restoration denufosol Mucus Regulation Antibiotic Anti- inflammatory Lung Transplant P2Y 2 Receptor Activation chloride ion and fluid secretion sodium absorption cilia beat frequency mucin secretion from goblet cells surfactant release from type II alveolar cells |
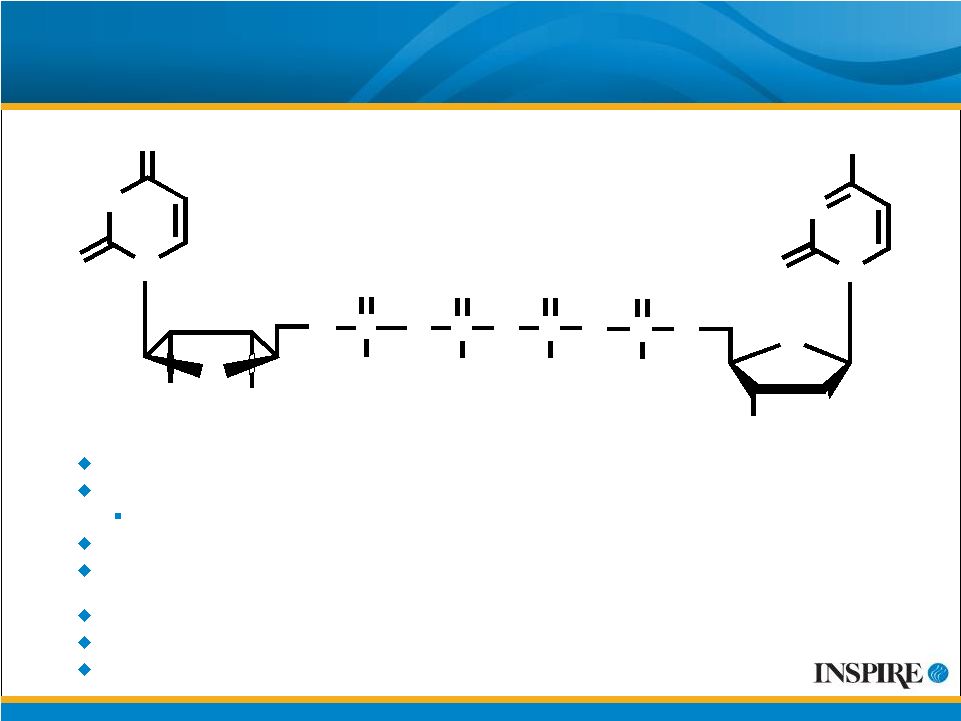 7 Global Phase 3 Program with Denufosol for CF Two Phase 3 pivotal trials: TIGER-1 and TIGER-2 TIGER-1 met primary efficacy endpoint in 24-week placebo-controlled portion Showed increasing lung function in open-label 24-week safety extension TIGER-2 initiated in February 2008 with enrollment expected to be complete in 2009 Histopathology results from two-year carcinogenicity study in rodents indicated no evidence of carcinogenic effect U.S. and European Orphan Drug Status and U.S. Fast Track Designation IP coverage through at least 2017 Inspire owns worldwide rights for denufosol; seeking ex-North American partner O O 4 Na + O OH O P - O P O - O O P O O O - P O O - O N HN N O O OH OH O N O NH 2 |
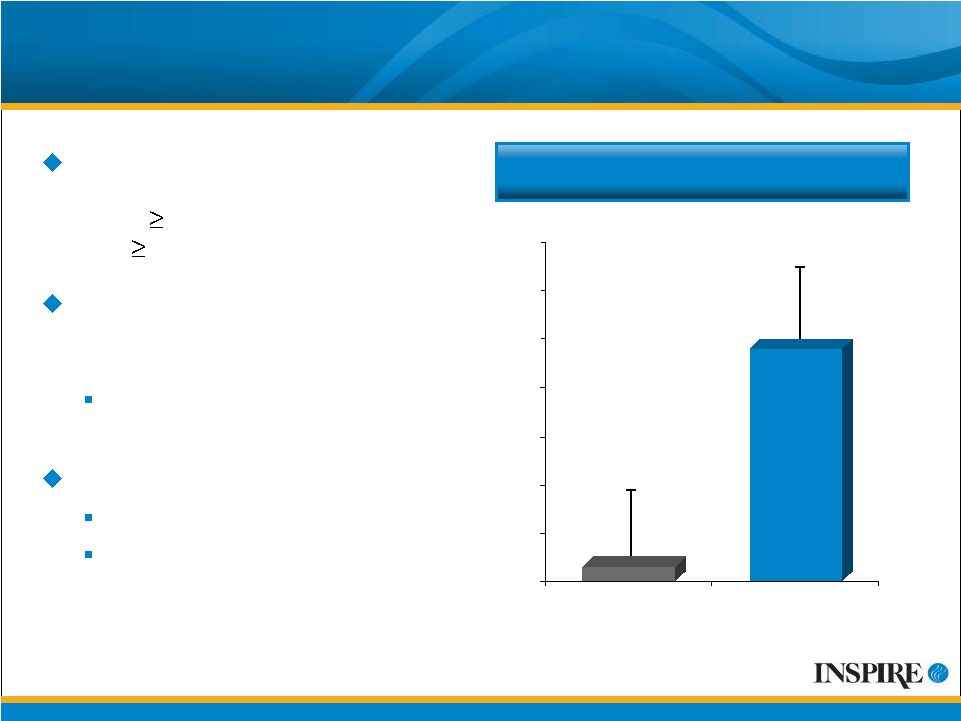 8 Mean (SE) Change in FEV 1 (mL) Primary Efficacy Results from TIGER-1: Placebo-Controlled (ITT) Enrolled 352 patients with early stage CF lung disease (FEV 1 75% predicted normal) and 5 years old Met primary efficacy endpoint: change in FEV 1 from baseline to endpoint* 45 mL treatment effect (p = 0.047) “Real world” design Broader population Patients on multiple therapies that improve FEV 1 3 48 0 10 20 30 40 50 60 70 Placebo Denufosol *Endpoint is the Week 24 value or the last observation carried forward for patients who withdrew early. # Values displayed are adjusted means from the pre-specified analysis of covariance model that included baseline FEV1 as a covariate. |
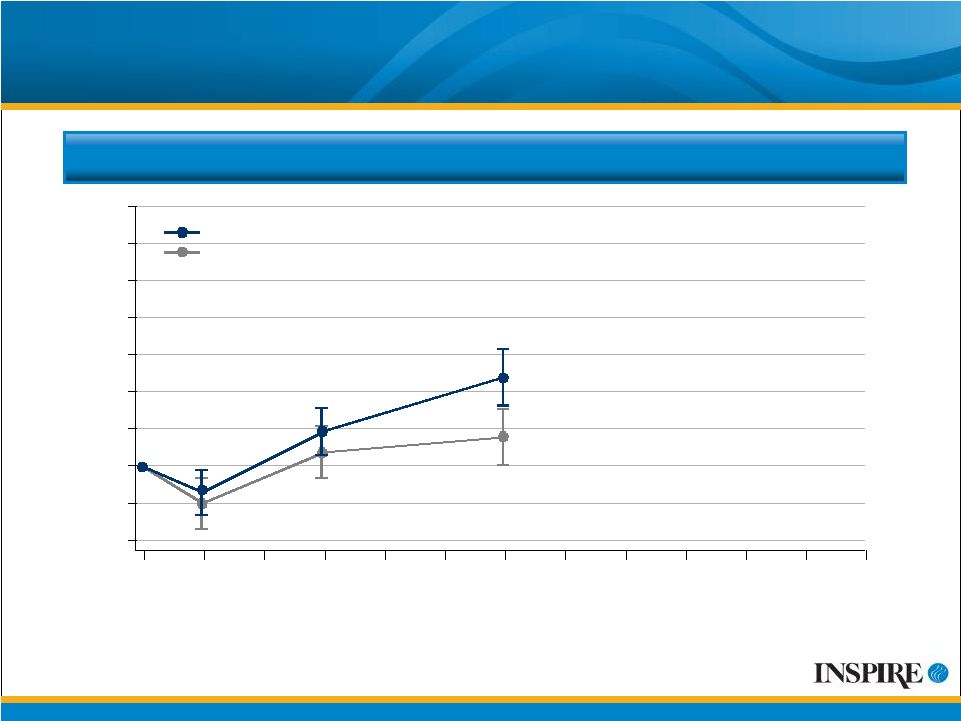 9 Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population # Values displayed are mean observed values at each time point without adjustment for discontinuations or covariates. Mean (SE) Change from Baseline FEV 1 (mL) 140 -40 -20 0 20 40 60 80 100 120 0 4 12 24 28 36 48 Treatment Week Placebo Denufosol |
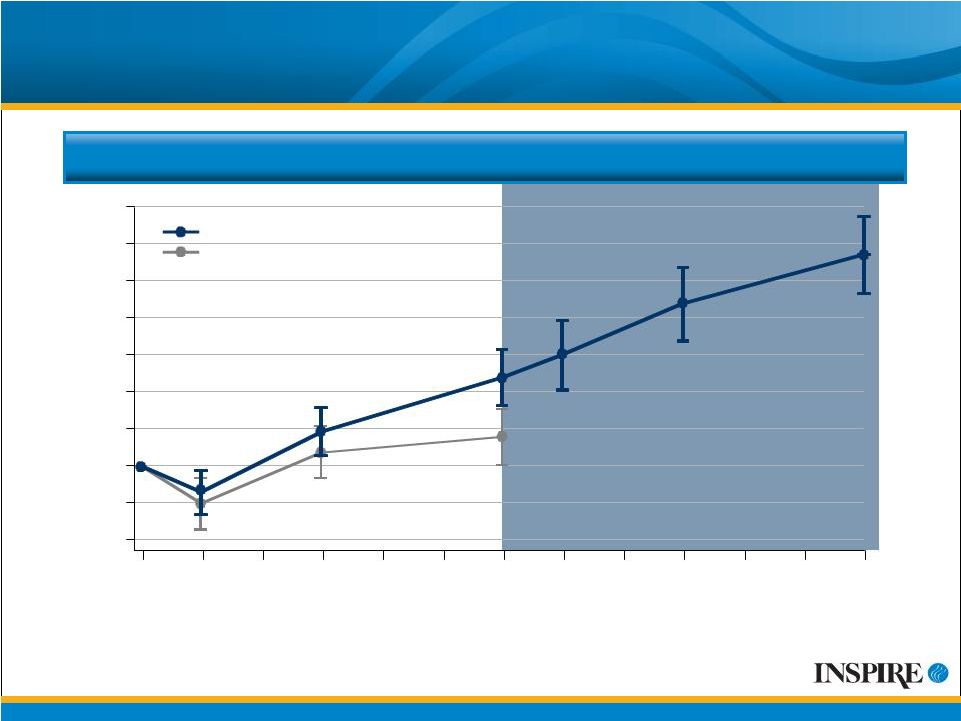 10 Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population # Values displayed are mean observed values at each time point without adjustment for discontinuations or covariates. Mean (SE) Change from Baseline FEV 1 (mL) 140 -40 -20 0 20 40 60 80 100 120 0 4 12 24 28 36 48 Treatment Week Placebo Denufosol Denufosol-only, Open-label Phase |
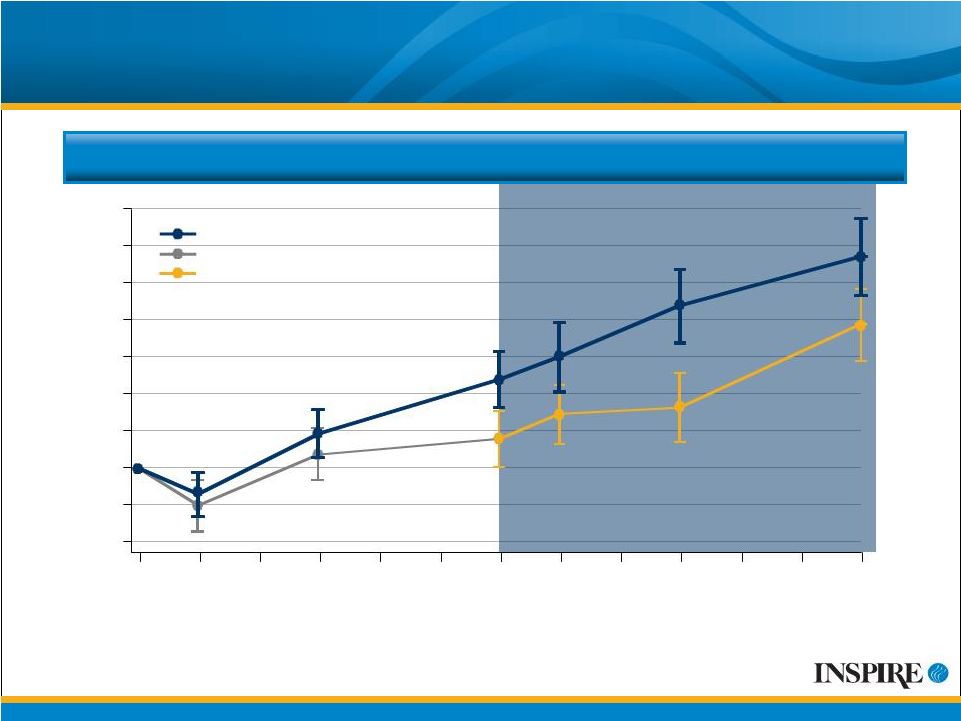 11 Mean (SE) Change from Baseline FEV 1 (mL) Mean Change from Baseline FEV 1 CFB=Change from Baseline ITT Population # Values displayed are mean observed values at each time point without adjustment for discontinuations or covariates. 140 -40 -20 0 20 40 60 80 100 120 0 4 12 24 28 36 48 Treatment Week Placebo Denufosol Placebo Switched to Denufosol Denufosol-only, Open-label Phase |
 12 TIGER-2 Phase 3 Clinical Trial (08-110) 350 patients enrolled as of July 24, 2009 Patients who complete TIGER-2 will be eligible for open-label, denufosol 48-week trial Expect to complete enrollment by end of year 2009 FEV 1 75% and 110% predicted normal for age, gender and height Age 5 or older Key Inclusion Criteria Participated in TIGER-1 Standard CF meds allowed, except hypertonic saline Key Exclusion Criteria Planned Trial Size: 450 Change in FEV 1 in liters at 48-week endpoint Primary Endpoint Pulmonary exacerbation, antibiotic use, incidence of hospitalization/ER visits, health resource utilization, quality of life Secondary Endpoint Denufosol 60 mg TID N = 225 Placebo TID N = 225 R A N D O M I Z A T I O N 48-week Treatment Period |
 13 TM Dry Eye: Affects More Than 30 Million Patients Worldwide *Market Scope Report on the Dry Eye Market, July 2004; **Inspire estimate based on extrapolation of U.S. Data ***Allergan 2008 financial results on 02/04/09 Caused by environmental or genetic factors, aging/menopause, immune/inflammatory disease, lid margin disease, certain medications Disease of the tears and ocular surface that leads to instability of the tear film, symptoms of discomfort and visual disturbance and, if left untreated, can result in damage to the ocular surface and visual impairment Characterized by decrease in tear production and/or imbalance in the composition of the tear film components Inspire estimates 9 mm patients in North America*, 30 mm patients worldwide** Disease Quick Facts Current Treatment Options Limited treatment options, which include artificial tears/lubricant drops and punctal plugs and one prescription product in the U.S. Worldwide artificial tear sales over $1 billion*** Restasis ® – prescription eye drop to help increase tear production reduced by inflammation – $444 mm in 2008 sales*** Since Prolacria and Restasis have different mechanisms of action, they may be complementary products with commercial opportunity for both |
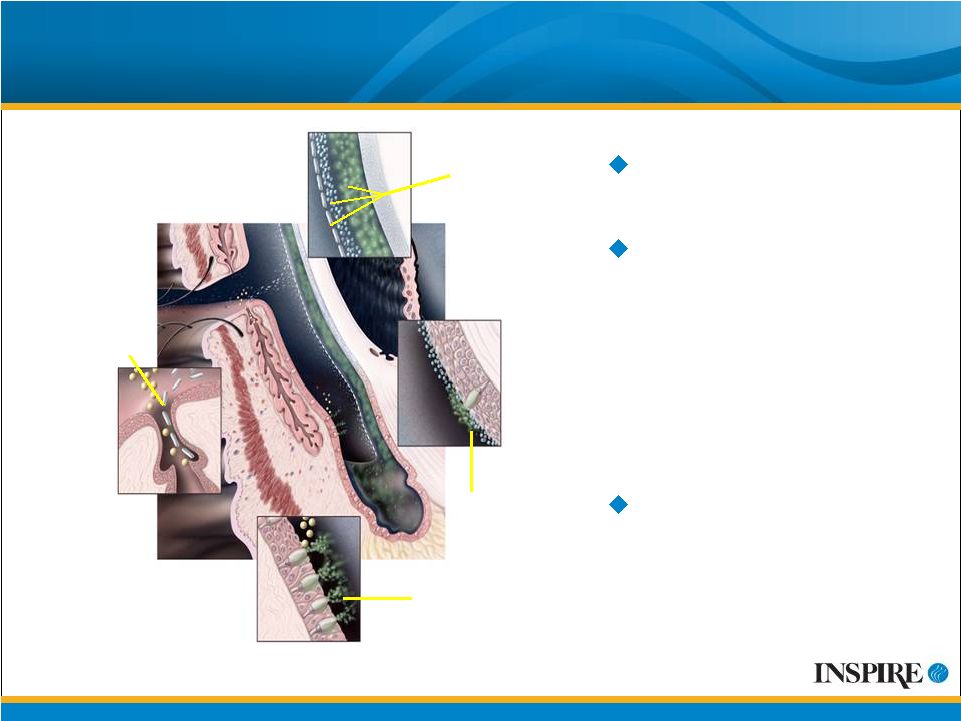 14 Prolacria : Mechanism of Action for Dry Eye Internally developed P2Y 2 agonist Receptor activation results in increased intracellular calcium concentration, which triggers a series of intracellular events resulting in mucosal surface secretion Promotes release of components of natural tear-film (fluid, mucin and lipids) Lipid secretion Mucin, aqueous & lipid layers Water & mucin secretion Mucin secretion Prolacria™ is the proposed U.S. trade name for diquafosol tetrasodium ophthalmic solution 2% TM |
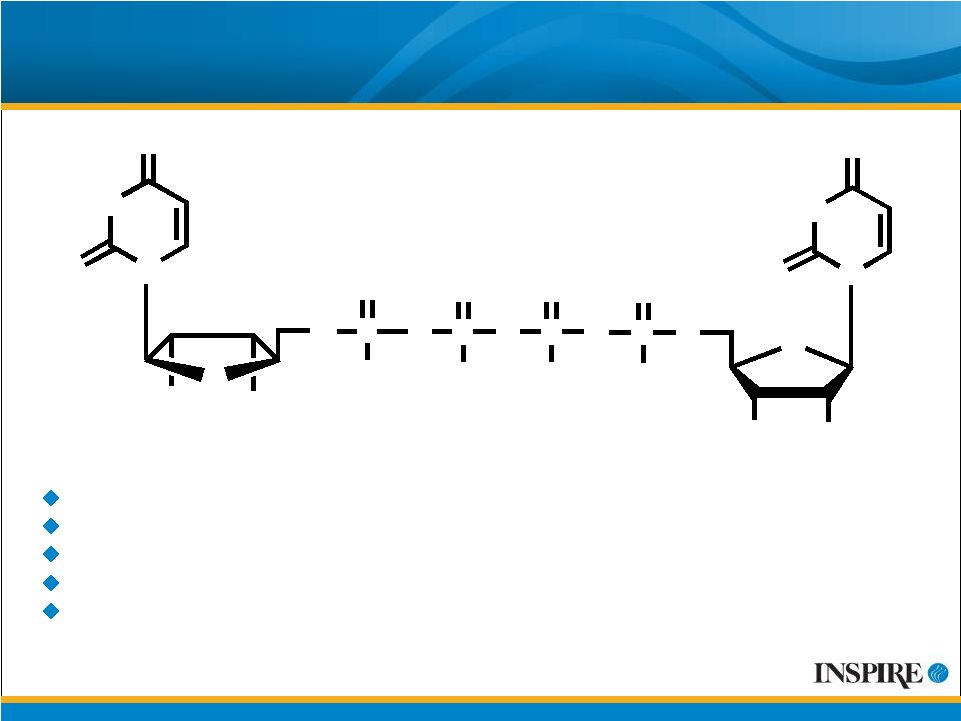 15 Phase 3 Program with Prolacria™ for Dry Eye O O 4 Na + O OH OH O P - O P O - O O P O O O - P O O - O N HN N O O OH OH O HN O O Four Phase 3 clinical trials conducted to date Tolerability and safety profile similar to placebo Received two approvable letters from U.S. FDA Initiated additional Phase 3 trial under SPA agreement in January 2009 IP coverage through at least 2017 |
 16 Clearing of Central Corneal Staining Proportion of all patients, regardless of baseline staining, that achieved clearing of central corneal staining (NEI score of 0) at Week 6 Endpoint in the Phase 3 environmental trials (Intent-to-Treat Population) Post-hoc analysis shows statistically significant differences in central corneal clearing scores shown for Prolacria treatment vs. placebo in two Phase 3 environmental trials (03-105 and 03-109) Post-hoc analysis based on Week 6 value or last observation carried forward for patients who withdrew early. P-value is from a Pearson Chi-Square Test using each patient’s study eye. Brazzell R.K., et al., ARVO, 2007; Tauber J., et al., Cornea, 2004 0.526 0.150 94/180 (52%) 38/186 (20%) 85/174 (49%) 47/175 (27%) <0.001 209/318 (66%) 164/322 (51%) 0.029 89/175 (51%) 67/171 (39%) Week 6 Endpoint 0.593 114/318 (36%) 122/322 (38%) 0.358 32/175 (18%) 25/171 (15%) Baseline Placebo N=175 Prolacria N=186 p-value Placebo N=171 Prolacria N=175 p-value Placebo N=322 Prolacria N=318 p-value 03-104 03-105 03-109 |
 17 Clearing of Central Corneal Staining in Patients With Baseline Central Corneal Staining Score of 3 0.109 0.320 >0.999 0/24 (0%) 0/17 (0%) 0/7 (0%) 3/23 (13%) 03-104 + 03-105 1/8 (13%) 03-105 2/15 (13%) 03-104 Study Prolacria Placebo p-value Proportion of patients with a baseline NEI staining score of 3 in the central cornea that achieved clearing of central corneal staining (NEI staining score of 0) at Week 6 Endpoint in the Phase 3 environmental trials (Intent-to-Treat Population) Post-hoc analysis based on Week 6 value or last observation carried forward for patients who withdrew early. P-value is from a Fisher’s Exact Test using each patient’s study eye. There were no patients with a central corneal staining score of 3 at baseline in Trial 03-109, as per the protocol inclusion/exclusion criteria. Post-hoc analysis shows trend in favor of Prolacria; not statistically significant due to small patient sample size; individual trials not designed for this endpoint |
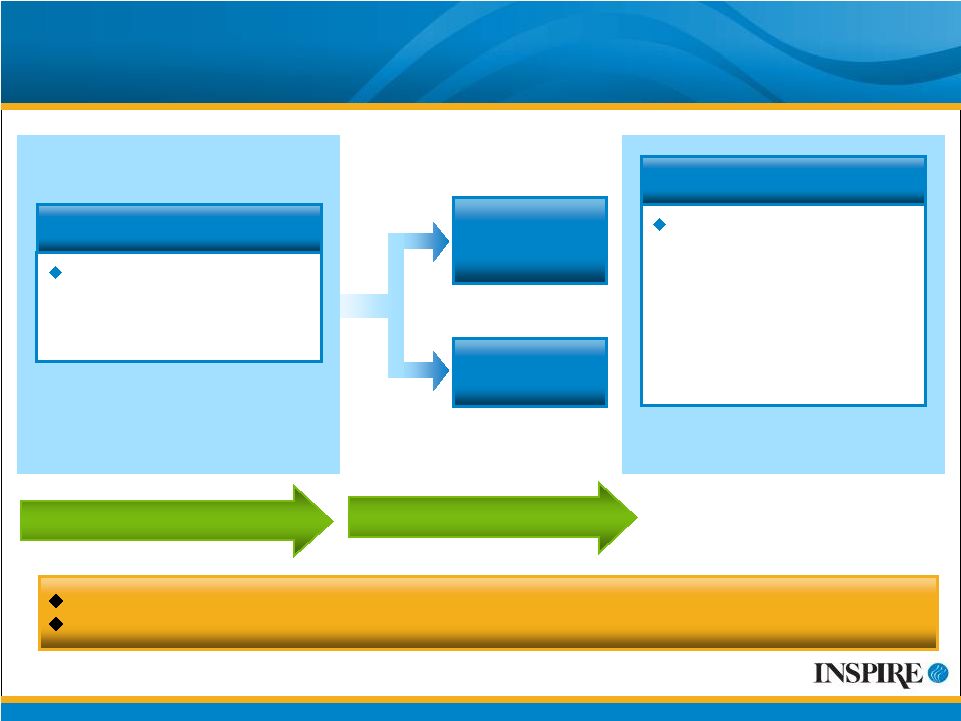 18 Prolacria ™ Phase 3 Environmental Clinical Trial (03-113) 1-week Placebo Run-in Period 6-week Treatment Period Planned Trial Size: 450 Corneal fluorescein staining score of 3 in the central region of the subject’s study eye using the NEI scale Key Inclusion Criteria Prolacria (diquafosol tetrasodium ophthalmic solution 2%) QID N = 225 Placebo QID N = 225 R A N D O M I Z A T I O N Proportion of subjects receiving Prolacria that achieve clearing of fluorescein staining of the central region of the cornea (a score of zero on the NEI scale) at the six-week trial endpoint, compared to those receiving placebo Primary Endpoint 345 patients enrolled as of July 24, 2009 Expect to complete enrollment in Fall 2009 |
 19 Global Partnerships for Prolacria™/DE-089 Partnered with Allergan, Inc., a leading global ophthalmic company Inspire responsible for development and approval of Prolacria™ in the United States, with right to co-promote Allergan responsible for commercialization and all post-marketing activities in the U.S., and development and commercialization in rest of world, except Asia Partnered with Santen Pharmaceutical Co., a premier ophthalmic company with 80% market share in dry eye in Japan Santen responsible for development and commercialization of diquafosol (DE-089) in Japan and nine other Asian countries Santen filed regulatory application for DE-089 in Japan in May 2008 Meaningful royalty stream for Inspire, if product is approved and launched |
 20 AzaSite ® : Differentiated Ocular Anti-Infective First ocular form of azithromycin AzaSite combines well-known, broad spectrum macrolide antibiotic, azithromycin (1%), with DuraSite ® drug delivery technology Anti-inflammatory properties Approved by FDA in April 2007, with indication for treatment of bacterial conjunctivitis IP coverage through 2019 Broad spectrum coverage against most common pathogens High tissue concentration on ocular surface and eyelids Easiest dosing regimen among all ocular anti-infectives (9 vs. 21+ drops) |
 21 Recent Development in Ocular Antibiotic Market Current manufacturing supply shortage of erythromycin ophthalmic ointment Generic macrolide antibiotic routinely used in neonates for prophylaxis of ophthalmia neonatorum, a form of bacterial conjunctivitis that may be contracted by newborns during delivery CDC, FDA and AAO have provided guidance to healthcare professionals in dealing with shortage Reserve current supplies of erythromycin ophthalmic ointment for neonatal prophylaxis use Use alternative drugs for treatment of superficial ocular infections AzaSite is an acceptable substitute for neonatal prophylaxis use where erythromycin ophthalmic ointment is not available Increased production of AzaSite in September 2009 to fill treatment gap as needed CDC made the following recommendation: "Our experts indicate that AzaSite ® (Azithromycin Ophthalmic Solution 1%, Inspire Pharmaceuticals) is an acceptable substitute, if Erythromycin Ophthalmic Ointment (0.5%) is not available".* *AzaSite has not been approved by the FDA for the treatment of prophylaxis of ophthalmia neonatorum and no clinical trials have been conducted using AzaSite in this population. |
 22 Blepharitis: A New Potential Indication Common condition characterized by inflammation of the eyelids, which is often secondary to infection Often involves significant patient discomfort leading to associated ocular surface inflammation, chronic ocular irritation, unstable tear film and damage to the ocular surface Blepharitis is an under-diagnosed and under-treated condition, which may be as prevalent as dry eye disease Survey data indicates that ophthalmologists and optometrists report that blepharitis is commonly seen in clinical practice in 37% and 47% of their patients, respectively* In a survey of 5,000 adults in the U.S., app. 15% reported having one to three symptoms of blepharitis at least half of the time in the past 12 months, implying as many as 34 million adults might suffer from some form of blepharitis (based on overall U.S. adult population data)* Disease Quick Facts Current Blepharitis Treatment Options No prescription products indicated for blepharitis Limited treatment options at present Warm compresses Lid hygiene Topical antibiotic ointments Topical steroids or oral antibiotics, when condition exacerbated * The Ocular Surface, May 2009 |
 23 0.0 1.0 2.0 3.0 4.0 Baseline After 2 Weeks of Treatment Warm Compresses (WC) AzaSite + WC Two-Week Phase 4 Trial in Posterior Blepharitis: Effect on Lid Margin Hyperemia p < 0.001 69% Improvement in Mean from Baseline with AzaSite vs. 10% with WC Alone (n=21) Luchs J, Adv Ther, Sept 25 (9):858-870, 2008 (0) Normal: no redness (3) Severe: increased vascularity of the eyelid margin (1) Mild: slightly dilated blood vessels (4) Very severe: clearly increased vascularity of the eyelid margin (2) Moderate: more apparent dilation of blood vessels ® |
 24 Four-Week Phase 4 Trial in Anterior/Posterior Blepharitis *p value listed is for the comparison to baseline for each visit (n=26) Shapiro A., et al., American Society of Cataract and Refractive Surgery (ASCRS) Annual Meeting 2009. 4 Wks After Dosing Stopped* 2 Wks After Dosing Stopped* After 4 Wks Dosing* Baseline AzaSite ® 1.6 p=0.037 1.3 p<0.001 1.3 p<0.001 2.2 Ocular Dryness 0.8 p<0.001 0.5 p<0.001 0.6 p<0.001 2.8 Swollen/Heavy Eyelids 0.3 p<0.001 0.2 p<0.001 0.4 p<0.001 1.7 Ocular Burning 0.8 p<0.001 0.9 p<0.001 0.7 p<0.001 2.2 Foreign Body Sensation 0.6 p<0.001 1.1 p<0.001 0.9 p<0.001 2.2 Eyelid Itching |
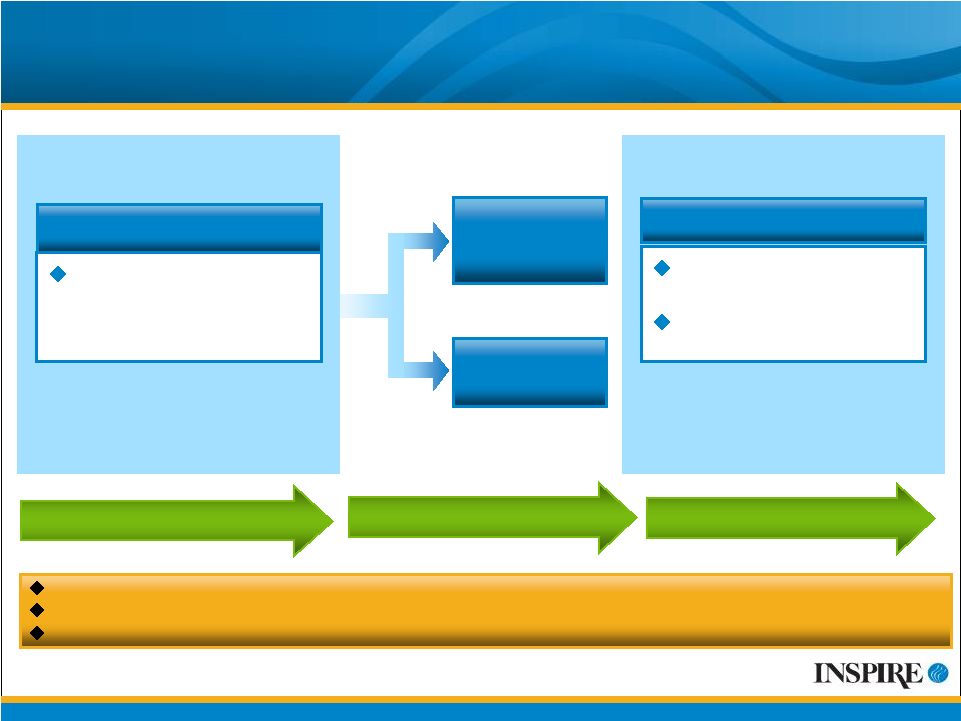 25 AzaSite Phase 2 for Blepharitis Clinical Trials (044-101 and 044-102) 6-week Optional Washout Period 2 or 4-week Treatment Period Two trials initiated in May 2009 with a two week treatment period (044-101) or four weeks (044-102) 90 patients enrolled in Trial 044-101 as of July 24, 2009 130 patients enrolled in Trial 044-102 as of July 24, 2009 Planned Trial Size: 300 Have current diagnosis of blepharitis in one or both eyes Key Inclusion Criteria AzaSite (azithromycin ophthalmic solution) 1% 1 drop BID for 2 days, then 1 drop QD for remainder N = 150 Placebo 1 drop BID for 2 days, then 1 drop QD for remainder N = 150 R A N D O M I Z A T I O N Signs and symptoms of blepharitis Safety and tolerability Endpoints 2 or 4-week Follow-up Period ® |
 26 Glaucoma: Largest Ophthalmic Market * Glaucoma Research Foundation ** IMS for the 12 months ended 12/31/08 Glaucoma is the leading cause of blindness in the world and the leading cause of preventable blindness in the U.S., according to the World Health Organization Glaucoma is a group of eye diseases that gradually reduces the field of vision A variety of factors cause loss of retinal ganglion cells leading to permanent damage to the optic nerve Associated with increased intraocular pressure (IOP) 65 mm worldwide, 4 mm in the U.S.* Disease Quick Facts Current Treatment Options Agents that reduce IOP: Beta blockers, prostaglandin F2 analogues, carbonic anhydrase inhibitors, alpha-2 adrenoreceptor agonists, combination agents No existing approved drug targets the primary outflow pathway Largest market in ophthalmic pharmaceuticals: $2.1 billion in U.S. prescription sales** |
 27 Glaucoma Program Update Novel approach to lowering intraocular pressure (IOP) by targeting primary outflow pathway (trabecular meshwork) Top-line Phase 1 clinical trial results in patients with elevated IOP INS115644 (Latrunculin B) – observed dose-dependant IOP-lowering effects in 56 randomized subjects; compound was generally well tolerated INS117548 (Rho kinase inhibitor) – observed mild IOP-lowering effects in 84 randomized subjects; tolerability issues (ocular discomfort) with this molecule Targeting potential presentation of key Phase 1 data at 2010 medical conferences Evaluating next steps for overall program, based on clinical and preclinical data, expert opinions and resource availability |
 28 Financing Summary Raised $115 million in common stock offering in August 2009 Financing fully funds current strategic plan Provides strong foundation to advance Company to the next level Enables a rapid succession of multiple catalysts Top-line clinical results in four programs within two years Three potential regulatory filings toward product approval |
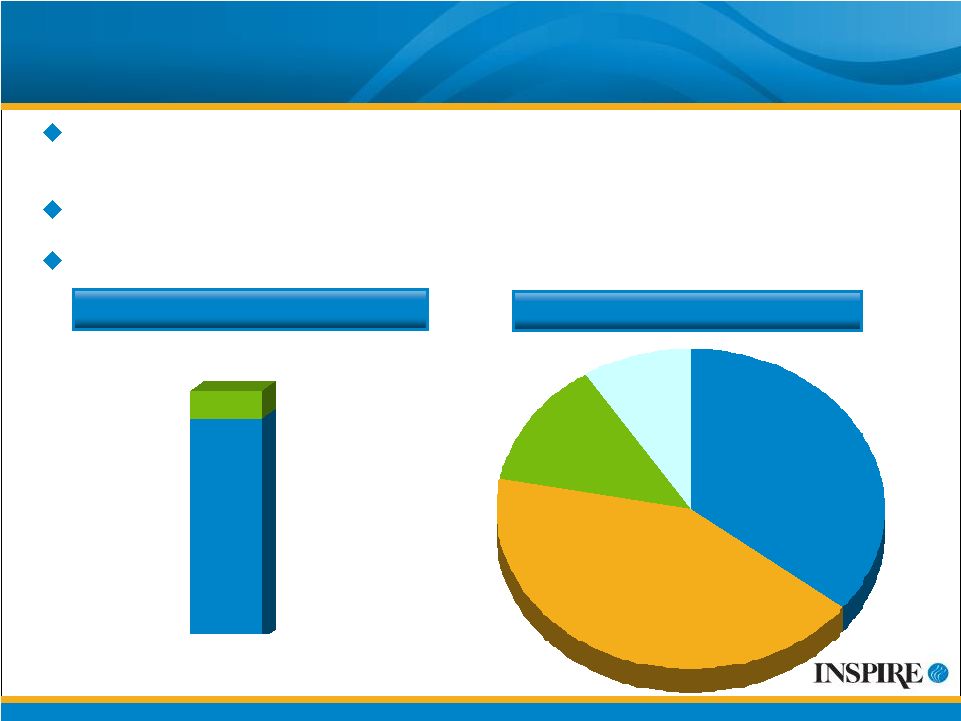 29 Current Financial Summary ~ $150 MM pro forma cash and investments balance, based on 6/30/09 balance plus $109 MM net proceeds from recent common stock offering 82.3 million common shares outstanding after common stock offering 2009 guidance reaffirmed on 07/31/09 Expenses - $120-135 mm Revenue - $80-90 mm M&S $45 - 50 mm R&D $50- 60 mm COGS $8-13 mm G&A $14 - 18 mm |
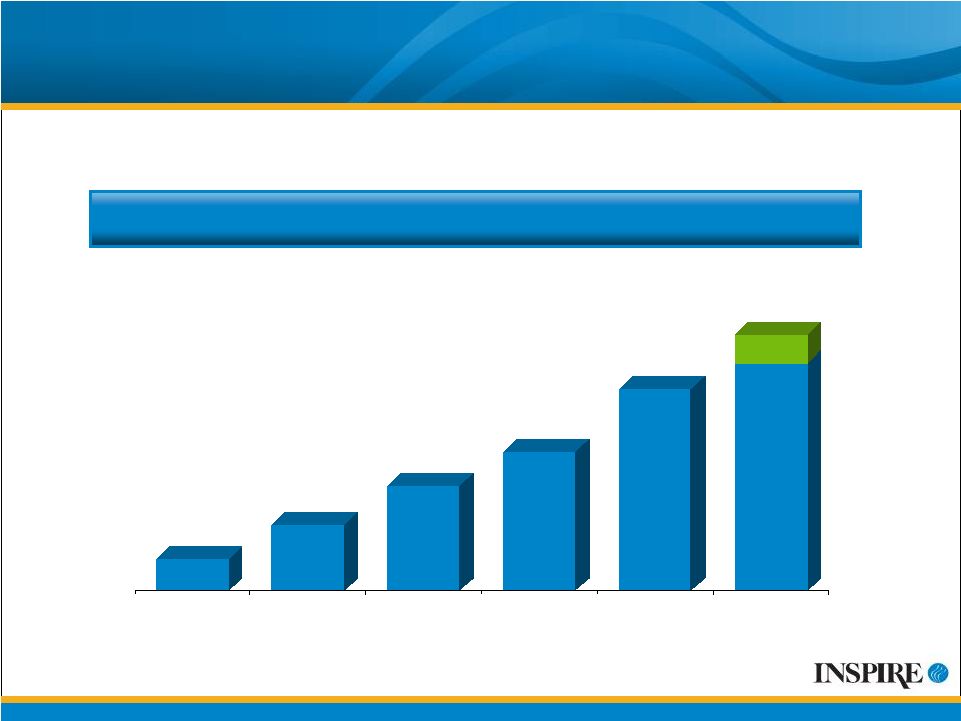 30 2004 2005 2006 2007 2008 2009 Guidance Inspire Has Achieved Double-Digit Annual Growth Each Year Growing Stream of Revenues Inspire Receives Revenues from Three Products* ($ Millions) $23 $49 $11 $37 $80-90** $71 31% increase 110% increase 59% increase 45% increase * Inspire receives revenues related to the promotion of AzaSite, co-promotion of Elestat and royalties on Restasis ** Guidance reaffirmed on 07/31/09 |
 31 Denufosol for Cystic Fibrosis – Phase 3 Complete enrollment in TIGER-2 CF trial (year-end 2009) Report top-line results of TIGER-2 CF trial Identify ex-North American partner for denufosol Prolacria for Dry Eye – Phase 3 Complete enrollment in Prolacria dry eye trial (Fall 2009) Report top-line results of Prolacria dry eye trial File NDA amendment with FDA including new Prolacria trial data Potential regulatory approval in Japan AzaSite ® for Blepharitis – Phase 2 Complete enrollment in AzaSite Phase 2 blepharitis trials (Q1 2010) Report top-line results of AzaSite Phase 2 blepharitis trials Initiate Phase 3 trials of AzaSite for blepharitis, if Phase 2 is positive Multiple Significant Events Over the Next Two Years TM |
 32 Inspire Summary Vision to build long-term value Therapeutically focused with unique, innovative pipeline and products Recent financing fully funds current strategic plan and provides strong foundation for future growth Multiple upcoming catalysts Top-line clinical results in four programs within two years Three potential regulatory filings toward product approval |
 UBS Global Life Sciences Conference September 22, 2009 |