Exhibit 99.1

Exhibit 99.1
Pharsight Corporation
Investor Presentation
August 2004
Shawn O’Connor President & CEO
Cynthia Stephens SVP & CFO
Copyright © 2004 Pharsight. All rights reserved.

Forward-Looking Statements
This presentation contains forward-looking statements related to future events or our future financial performance, including forward-looking statements regarding our competitive position, trends in customer demand, potential business growth, expansion of scope and increasing efficiency of consulting engagements, new service offerings, market conditions, future revenue growth and profitability, and revenue trends.
Forward-looking statements are inherently speculative, and actual results may differ materially from our expectations due to a variety of factors, including: timing of completion of large software installations and related revenue recognition may cause revenue and net income to fluctuate significantly in individual quarters; unpredictable fluctuations in the valuation of Pharsight’s preferred stock dividends may cause net income attributable to common stockholders in any future period to be significantly less than net income before the effect of the preferred dividend; changing economic conditions may affect the demand Pharsight expects for its products and services and may prevent the company from sustaining positive cash flow; changes in Pharsight research and development focus or operating strategies due to new market opportunities or conditions may delay the company’s growth or ability to sustain profitability or the company’s ability to sustain positive cash flow; we may be unable to sufficiently anticipate market needs and offer products and services that achieve market acceptance, we may have higher than anticipated expenses in future quarters; and other risks disclosed under the caption “Risk Factors” in Pharsight’s Quarterly Report on Form 10-Q, as filed with the Securities and Exchange Commission on August 12, 2004. Although we believe that expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. All forward-looking statements are based on information available to Pharsight as of the date hereof, and Pharsight assumes no obligation to update such statements, whether as a result of new developments or otherwise.
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL

Pharsight® is…
Delivering breakthrough improvement in the clinical drug development process to pharmaceutical companies, by accelerating client product time-to-market and improving client development efficiencies
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL
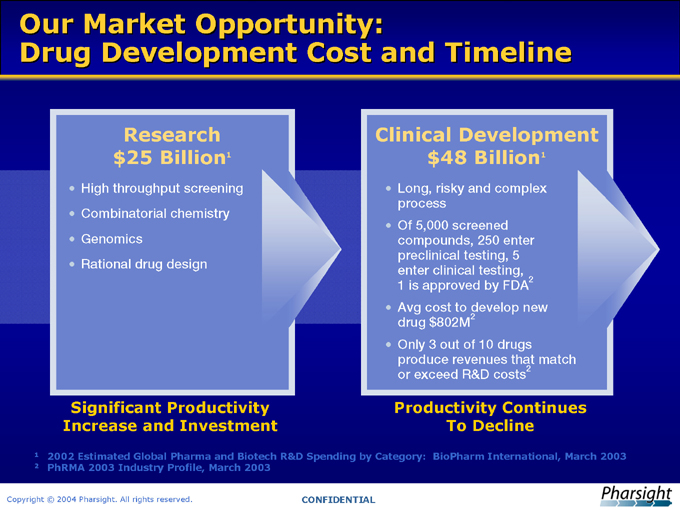
Our Market Opportunity:
Drug Development Cost and Timeline
Research
$25 Billion1
High throughput screening
Combinatorial chemistry
Genomics
Rational drug design
Significant Productivity Increase and Investment
Clinical Development
$48 Billion1
Long, risky and complex
process
Of 5,000 screened compounds, 250 enter preclinical testing, 5 enter clinical testing, 1 is approved by FDA2
Avg cost to develop new drug $802M2
Only 3 out of 10 drugs produce revenues that match or exceed R&D costs2
Productivity Continues To Decline
1 2002 Estimated Global Pharma and Biotech R&D Spending by Category: BioPharm International, March 2003
2 PhRMA 2003 Industry Profile, March 2003
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL

Profile
Background
Founded 1995; IPO August 2000
Headquarters—Mountain View, CA
Satellite Office—Cary, NC
Headcount—approx 80
US, Europe, Australia and Japan
OTCBB: PHST
??Business and Products
Unique provider of strategic scientific consulting and decision analysis delivering significant impact to customers’ clinical development process
Market leading provider of analysis application for PK/PD modeling (WNL Suite) and Data Repository for PK data and results
Opportunity to leverage and accelerate use of scientific methodology through new software application—Drug Model Explorer TM (“DMX TM”)
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL

Executive Team
Shawn O’Connor President & Chief Executive Officer QRS, Diasonics Ultrasound, Peat Marwick
Cynthia Stephens SVP & Chief Financial Officer Rainmaker Systems, Calico, Quiver, Infoseek/ The Walt Disney Company
Mark Robillard SVP Software Products Business Unit EMAX Solutions, VWR Scientific Products
Mona Sowiski SVP Drug Development Consulting Services Mitchell Madison Consulting, CSC/APM Healthcare, Stanford, Pitt
Daniel Weiner, PhD SVP Business Development IVAX, Merrell Dow, Syntex, Quintiles
William Gillespie, PhD VP Consulting Services, East Coast GloboMax, FDA (CDER), Univ. of Texas at Austin-College of Pharmacy
Greg Lee, PhD VP Research & Development Sunrise Test Systems, Weitek, Schlumberger
Nancy Risch VP Global Sales BBN Corporation, Interleaf, GE, Wang
Russ Wada, PhD VP Consulting Services, West Coast and Japan Stanford, MiniMed Infusion Systems, Hughes Aircraft, TRW
Rene Bruno, PhD Managing Director, Consulting Services, Europe Genentech, Rhone-Poulenc Rorer, Syntex
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL

Software Products
Copyright © 2004 Pharsight. All rights reserved.

Key Software Offerings
WinNonLin® (“WNL™”)
Industry Standard for Pharmacokinetic (“PK”), Pharmacodynamic (“PD”) Modeling and Non Compartmental Analysis
Key Benefits
Used to simulate effects of different dosing regimens and changes in
pharmacokinetic parameters required in regulatory submissions
Increases productivity in modeling, analysis
and reporting activities
?Potential Growth Drivers
Maintain market share with subscription model
Provide upgrade path to support incremental revenue opportunity
?Sales Model
Annual subscription fee
ASP $3,000—$6,500 per seat
Copyright © 2004 Pharsight. All rights reserved.
CONFIDENTIAL

Key Software Offerings
Pharsight Knowledge Server™ (“PKS™”)
Enterprise Data Management Systems for Secure Storage and Management of PK/PD Data
Key Benefits
Dramatically improves data management and data access
Increases productivity in analysis and reporting tasks
Supports compliance with FDA requirements for electronic records
and signature, CFR 21 part 11
Potential Growth Drivers
Leverage installed base of clients
Optimize adoption pattern of industry
Expand utility and seat requirements for application
Sales Model
Subscription and perpetual license model
Deployment services, customization/automation
Post-contract maintenance & support
Average contract size $500K
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Key Software Offerings
Drug Model Explorer™ (“DMX™”) Software-Based Communication Technology, Designed Quantitative Decision-Making in Drug Development to Facilitate
Introduced in fiscal 2004
Key Benefits
Facilitates more efficient collaboration within project teams to explore key drug attributes, and their respective uncertainties
Enables more flexible scenarios & generates views of program data from underlying model outputs & simulated responses over defined problem-space
Potential Growth Drivers
Expand client base of early adopters
Expand utility and footprint of application
Sales Model
Annual subscription fee
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Software Products Market Acceptance
Over 900 customers (FY2004)
All top 20 Pharma apply our computer-assisted drug development software products
Our software applications are licensed for use on more than 2,900 researcher desktops
3 of the top 10 Pharma are early adopters of our new DMX TM product
4 of the top 10 Pharma are PKS TM customers
7 of the top 20 Pharma
13 PKS customers to-date
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services
Copyright © 2004 Pharsight. All rights reserved.

Drug Development Consulting Methodology
A formal, quantitative, model-based decision-making method
Increase drug development productivity
Decrease late stage attrition
Decrease time-to-market
Increase # of drugs reaching market/$ invested
Improve clinical quality and commercial performance of final product
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
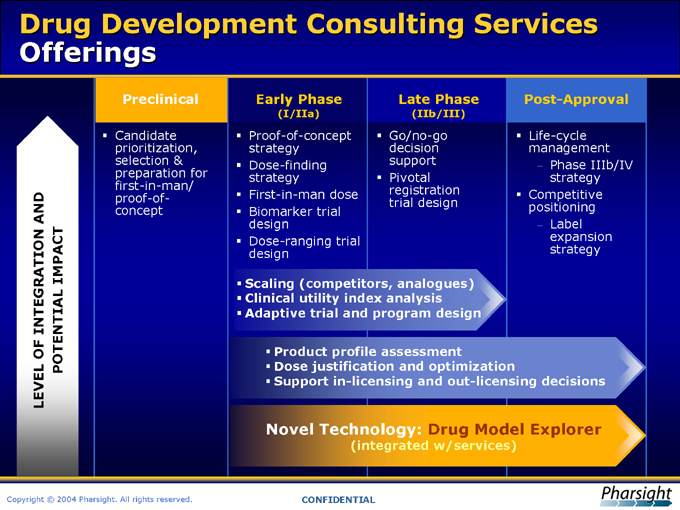
Drug Development Consulting Services Offerings
LEVEL OF INTEGRATION AND
POTENTIAL IMPACT
Preclinical
?Candidate prioritization, selection & preparation for first-in-man/ proof-of-concept
Early Phase
(I/IIa)
Proof-of-concept strategy
Dose-finding strategy
First-in-man dose
Biomarker trial design
Dose-ranging trial design
Late Phase
(IIb/III)
Go/no-go decision support ?Pivotal registration trial design
Post-Approval
Life-cycle management
Phase IIIb/IV strategy ?Competitive positioning
Label expansion strategy
Scaling (Competitors, analogues)
Clinical utility index analysis
Adaptive trial and program design
Product profile assessment
Dose justification and optimization
Support in-licensing and out-licensing decisions
Novel Technology: Drug Model Explorer
(integrated w/services)
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services
Case Study: Trial Design Optimization/Label Expansion Strategy
Critical Business Issue
Exploration of new indication for flagship product franchise
Approach & Technologies Utilized
Predictive modeling of key clinical measures using literature, public data and customer study data
Optimized trial design using Trial SimulatorTM
Results
Optimized study design (dose-arms, trial duration) based on predicted likelihood of positive result
Impact
Estimated savings of 25% in direct clinical costs on multi-million dollar trial, plus substantial time-to-market savings
Trial was success as predicted; FDA Advisory Committee recommended
approval for new indication
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services
Case Study: Trial Design Optimization/Phase II Go/No-Go Decision
Critical Business Issue
Optimal Phase II proof-of-concept trial design for novel treatment
??Approach & Technologies Utilized
Drug-disease models developed from available Phase I data and public literature for competing treatments
Alternate trial designs explored via simulation to predict significant drug effect; optimal design selected and success criteria defined for making
conclusive ‘go/no go’ decision
??Results
Trial conducted based on smaller, more efficient design
Results failed to meet success criteria; development was terminated early
??Impact
Early attrition: $1.4-$3.8 million estimated savings in direct trial costs, plus earlier redeployment of project team to new programs
Novel design yielded no go decision 8-12 months earlier than traditional development strategy
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services
Case Study: Use of DMX to Support
Program Strategy & Decision Making
Critical Business Issue
For new compound in core franchise, evaluation of product attributes and positioning as mono/combination-therapy
??Approach & Technologies Utilized
Modeled dose-response for multiple endpoints, evaluated safety and efficacy tradeoffs in competitive landscape
Model insights made available via Drug Model ExplorerTM; predictive models and DMXTM were rapidly updated to incorporate new clinical study
results
??Results
DMXTM used in ‘real-time’ decision-making environment to build team consensus, rapidly answer what-if questions, and support dose selection
??Impact
Supported critical question of product viability and competitive positioning through optimal dose selection balancing safety and efficacy
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services
Case Study: Go/No-Go Decision
Critical Business Issue
Product positioning vs. first-generation competitor
??Approach & Technologies Utilized
Predictive treatment response-models linked with quantitative framework (Clinical Utility Index) to provide integrated metric of clinical benefit vs. competitor on key safety/efficacy attributes
??Results
Failure to demonstrate equivalent clinical utility vs. competitor at all doses examined
??Impact
Further development was halted, saving an estimated $50M-$100M in development costs
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

FORBES ASAP | October 7, 2002
Aventis turned to a computer model from a small company named Pharsight. Using its own animal and human test data, Aventis was able to simulate later-stage clinical trials. Based on that simulation, “We stopped funding development of the compound. The ratio between the therapeutic benefit and side effect demonstrated that this compound was not as beneficial as Evista.” Douglas estimates that the Pharsight computer model saved Aventis $50M to $100M, the cost of later-stage clinical trials. “We were able to switch to another project with a greater chance of success.”
Frank Douglas Aventis Chief Scientific Officer and
Executive VP of Drug Innovation and Approval
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services Potential Growth Drivers
??Increased acceptance of modeling and simulation as an alternative to clinical trial results in Pharma’s development cycle
Migration from one-off projects to systematic application within portfolio
??Expanding scope and increasing efficiency of consulting engagements
??Broaden services offering
??Ability to recruit and retain skilled scientific staff to support client demand
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Drug Development Consulting Services: Market Acceptance
??18 of top 20 Pharma utilize our strategic consulting services
??All 5 of the top 5 Pharma utilize our services
Average duration of client relationships – 4 years
9 of the top 10 Pharma utilize our services
Average duration of client relationships – 3 years
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Financial Overview
Copyright © 2004 Pharsight. All rights reserved.

FY04 Accomplishments FY05 Objectives
Fiscal Year 2004 Results
Positive revenue momentum
?Y-Y revenue growth of 27%
Effected operating efficiencies
?Y-Y reductions in operating loss of 84%
Achieved profitability in Q4
Achieved positive annual operating cash flow
Introduced new product offering (DMX)
Fiscal Year 2005 Objectives
Continued revenue growth of 27%-30% Y/Y
Profitability
Full year net income (5-10% of revenue)
Excluding impact of preferred stock dividends
Expansion of DMX capabilities
Additional new product development
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
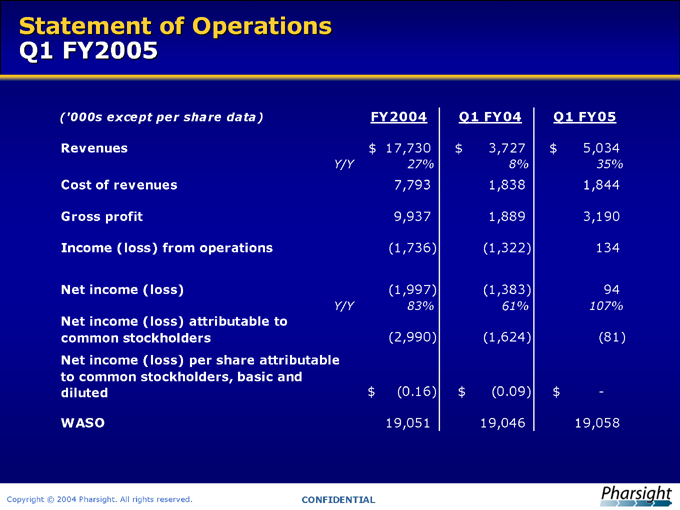
Statement of Operations Q1 FY2005
(‘000s except per share data) FY2004 Q1 FY04 Q1 FY05
Revenues $ 17,730 $ 3,727 $ 5,034
Y/Y 27% 8% 35%
Cost of revenues 7,793 1,838 1,844
Gross profit 9,937 1,889 3,190
Income (loss) from operations (1,736) (1,322) 134
Net income (loss) (1,997) (1,383) 94
Y/Y 83% 61% 107%
Net income (loss) attributable to common stockholders (2,990) (1,624) (81)
Net income (loss) per share attributable to common stockholders, basic and diluted $ (0.16) $ (0.09) $ -
WASO 19,051 19,046 19,058
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
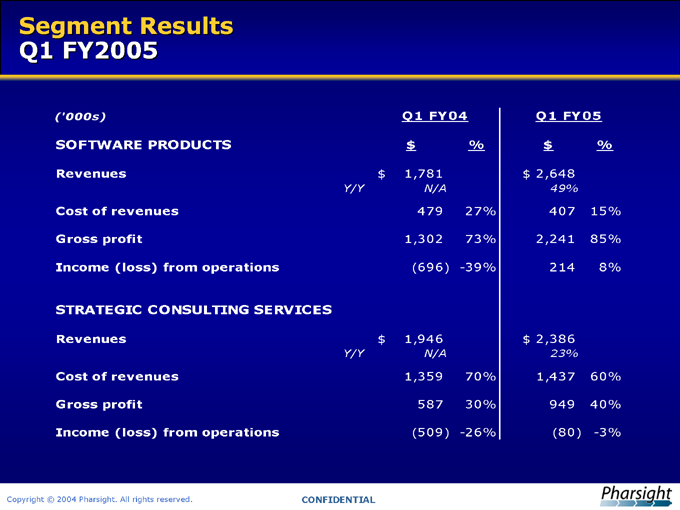
Segment Results Q1 FY2005
(‘000s) Q1 FY04 Q1 FY05
SOFTWARE PRODUCTS $ % $ %
Revenues $ 1,781 $ 2,648
Y/Y N/A 49%
Cost of revenues 479 27% 407 15%
Gross profit 1,302 73% 2,241 85%
Income (loss) from operations (696) -39% 214 8%
STRATEGIC CONSULTING SERVICES
Revenues $ 1,946 $ 2,386
Y/Y N/A 23%
Cost of revenues 1,359 70% 1,437 60%
Gross profit 587 30% 949 40%
Income (loss) from operations (509) -26% (80) -3%
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
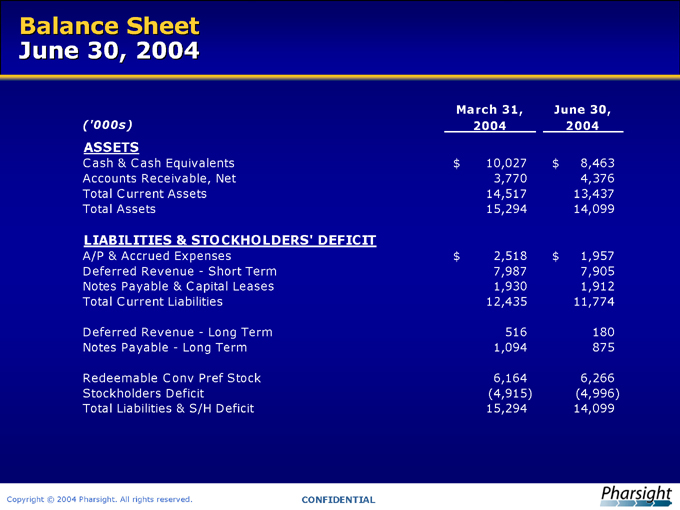
Balance Sheet June 30, 2004
(‘000s) March 31, 2004 June 30, 2004
ASSETS
Cash & Cash Equivalents $ 10,027 $ 8,463
Accounts Receivable, Net 3,770 4,376
Total Current Assets 14,517 13,437
Total Assets 15,294 14,099
LIABILITIES & STOCKHOLDERS’ DEFICIT
A/P & Accrued Expenses $ 2,518 $ 1,957
Deferred Revenue—Short Term 7,987 7,905
Notes Payable & Capital Leases 1,930 1,912
Total Current Liabilities 12,435 11,774
Deferred Revenue—Long Term 516 180
Notes Payable—Long Term 1,094 875
Redeemable Conv Pref Stock 6,164 6,266
Stockholders Deficit (4,915) (4,996)
Total Liabilities & S/H Deficit 15,294 14,099
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Equity Structure
(All share data as of July 31, 2004)
Common Shares O/S: 19,058,453
?Preferred Shares O/S: 1,869,085
Series A 1,814,662 / Series B 54,423
?Common Equivalent Shares (as converted): 26,534,793
PIPE Financing 6/02 and 9/02 = $7.5M
1,814,662 Units Series A redeemable convertible
stock + warrants
Preferred conversion ratio 1:4
Warrants exercisable for 1 share common @ $1.15
Dividend rights: 8%/annum cash or Series B shares
Conversion:
Automatically upon consent of 75% of preferred holders, or
Upon close of public offering of at least $3.006 per share and proceeds of $40M
Redemption:
At election of at least 75% of preferred holders (or at Pharsight’s option) after 5th anniversary of initial issuance at $4.008 per share
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
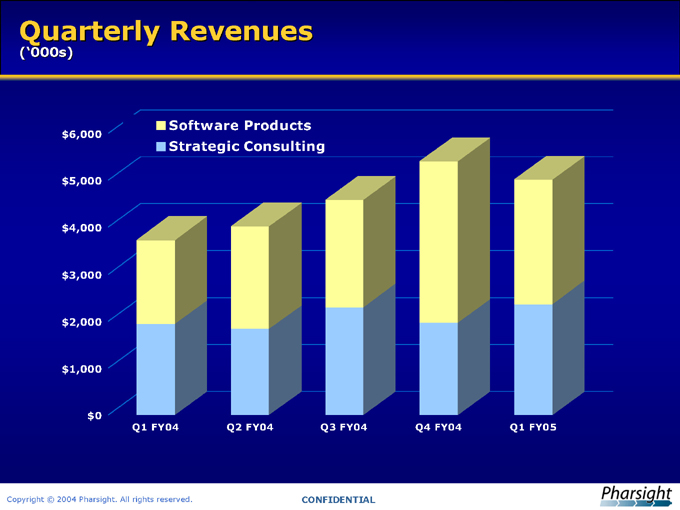
Quarterly Revenues
(‘000s)
Software Products Strategic Consulting
$6,000
$5,000
$4,000
$3,000
$2,000
$1,000
$0
Q1 FY04 Q2 FY04 Q3 FY04 Q4 FY04 Q1 FY05
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
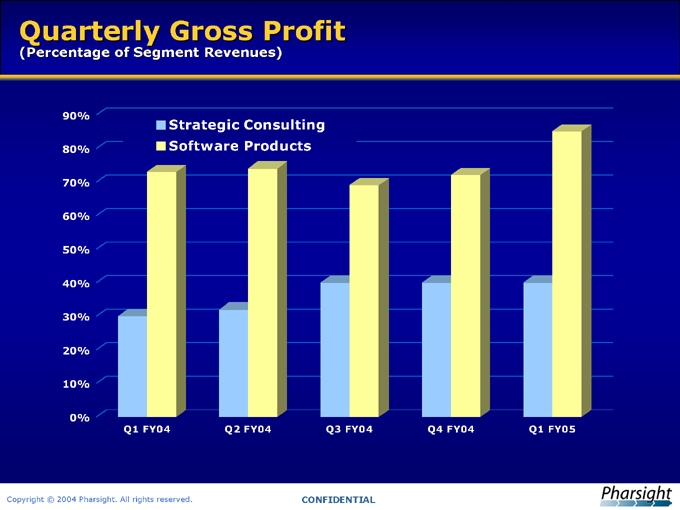
Quarterly Gross Profit
(Percentage of Segment Revenues)
Strategic Consulting Software Products
90% 80% 70% 60% 50% 40% 30% 20% 10%
0%
Q1 FY04 Q2 FY04 Q3 FY04 Q4 FY04 Q1 FY05
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
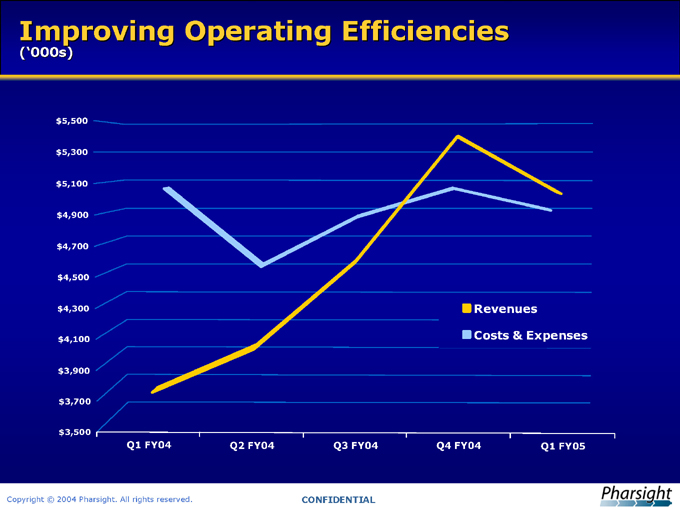
Improving Operating Efficiencies
(‘000s)
Revenues Costs & Expenses
$5,500
$5,300
$5,100
$4,900
$4,700
$4,500
$4,300
$4,100
$3,900
$3,700
$3,500
Q1 FY04 Q2 FY04 Q3 FY04 Q4 FY04 Q1 FY05
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Summary
Current Pharma Market Trends…
Continuing Pharsight Investment in Revenue Optimization & Product Development…
Increased Financial Stability Achieved in FY04…
And Continued Positive Momentum Demonstrated in Q1 FY05…
All Support Our Drive Towards
Sustainable Growth and Profitability in
FY05
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL

Notices
Proprietary Notice
All contents Copyright ©2004 Pharsight Corporation. All rights reserved. The copyright for this document is owned by Pharsight Corporation.
No part of this document may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying, for any purpose, without the express written permission of Pharsight Corporation. WinNonLin® and Pharsight® are registered trademarks of Pharsight Corporation. Pharsight Knowledgebase ServerTM, PKS TM, PKS Reporter TM, Drug Model Explorer TM, DMX TM and Trial Simulator TM are trademarks of Pharsight Corporation.
All other brand and product names are trademarks or registered trademarks of their respective holders.
Copyright © 2004 Pharsight. All rights reserved. CONFIDENTIAL
