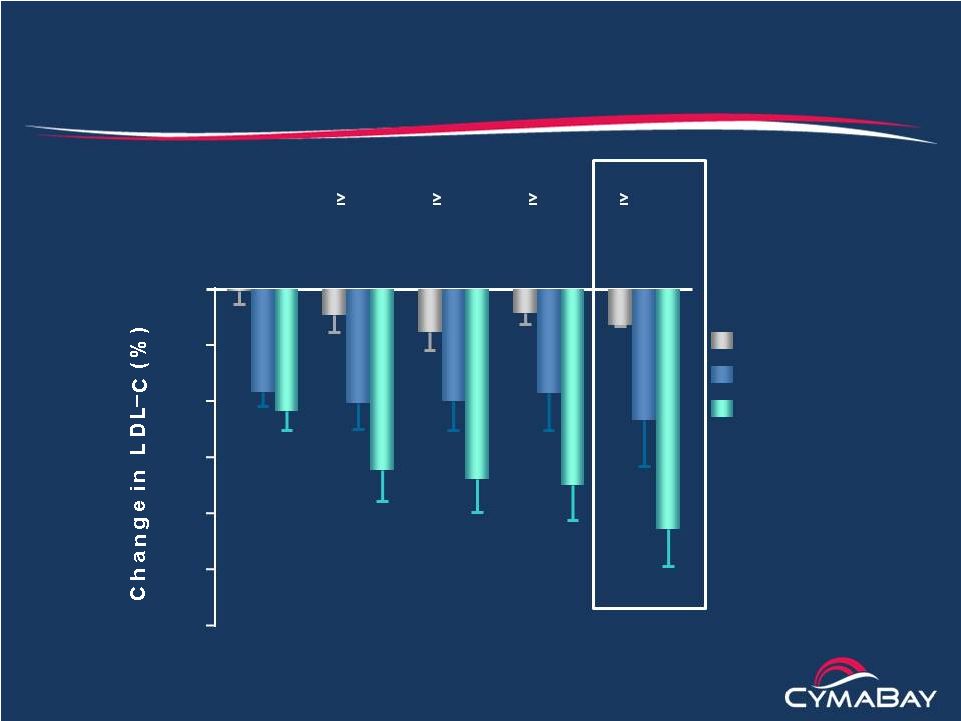
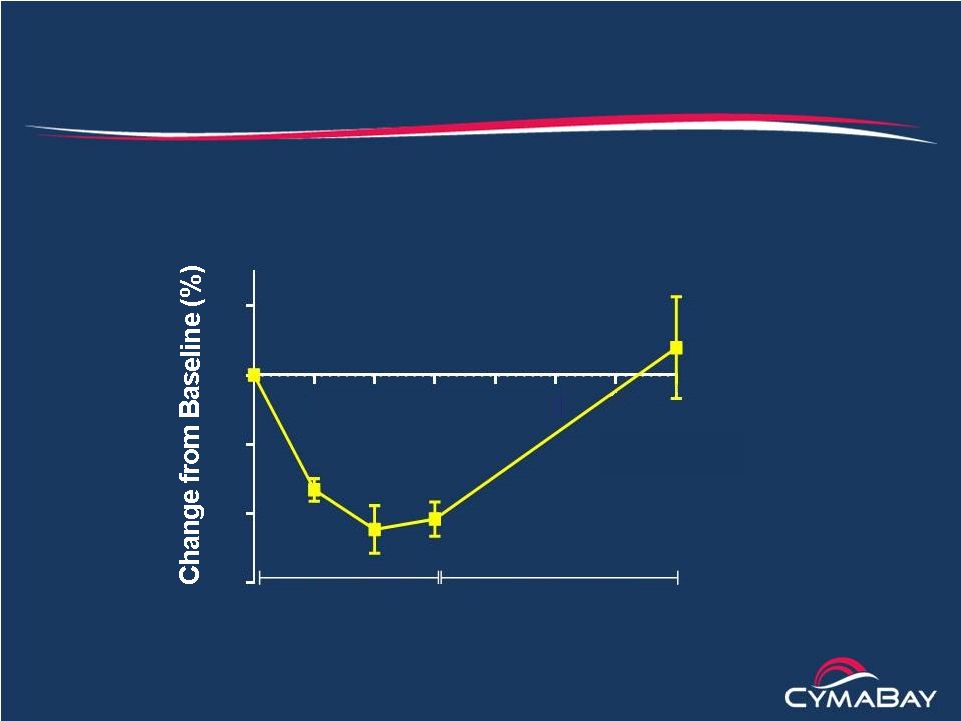
2 Safe Harbor Statement This presentation contains "forward-looking" statements that involve risks, uncertainties and assumptions, and actual results may differ substantially from those projected or expected in the forward-looking statements. Forward-looking statements include, but are not limited to: any projections of financial information; any statements about future development, clinical or regulatory events; any statements concerning CymaBay's plans, strategies or objectives; and any other statements of expectation or belief regarding future events. These statements are based on estimates and information available to CymaBay at the time of this presentation and are not guarantees of future performance. Actual results could differ materially from CymaBay's current expectations as a result of many factors including, but not limited to: CymaBay's ability to obtain additional financing to fund its operations; unexpected delays or results in clinical trials; uncertainties regarding obtaining regulatory approvals; uncertainties regarding the ability to protect CymaBay's intellectual property; uncertainties regarding market acceptance of any products for which CymaBay is able to obtain regulatory approval; the effects of competition; and other market and general economic conditions. You should read CymaBay's Quarterly Report on Form 10-Q filed with the SEC on November 14, 2014, especially under the caption “Risk Factors,” which is available on the SEC web site at http://www.sec.gov, for a fuller discussion of these and other risks relating to an investment in CymaBay’s common stock. CymaBay assumes no obligation for and does not intend to update these forward-looking statements, except as required by law. |