EXHIBIT 99.1

January 2019 Investor Presentation Driving Value to Dermatology Partners Matching Patients and Clinics

This presentation includes “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 that are subject to risks, uncertainties and other factors. All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including any statements of the plans, strategies and objectives of management for future operations; any statements regarding product development, product extensions, product integration or product marketing; any statements regarding continued compliance with government regulations, changing legislation or regulatory environments; any statements of expectation or belief and any statements of assumptions underlying any of the foregoing. In addition, there are risks and uncertainties related to successfully integrating the products and employees of the Company, as well as the ability to ensure continued regulatory compliance, performance and/or market growth. These risks, uncertainties and other factors, and the general risks associated with the businesses of the Company described in the reports and other documents filed with the SEC, could cause actual results to differ materially from those referred to, implied or expressed in the forward-looking statements. The Company cautions readers not to rely on these forward-looking statements. All forward-looking statements are based on information currently available to the Company and are qualified in their entirety by this cautionary statement. The Company anticipates that subsequent events and developments will cause its views to change. The information contained in this presentation speaks as of the date hereof and the Company has or undertakes no obligation to update or revise these forward-looking statements, whether as a result of new information, future events or otherwise. Safe Harbor Statement 2

STRATA Investment Highlights Seasoned Management Team – with Deep Domain KnowledgeDr. Dolev Rafaeli, CEO returns with the Accelmed transaction Excimer Laser Technology for Dermatology IndicationsPsoriasis, Vitiligo, Eczema, Atopic Dermatitis and Leukoderma Large Market Opportunity Over 35 Million available patients in the United States alone Proven Strategy and Unique Business ModelBest in class XTRAC and VTRAC Excimer Laser technologies Features stable base of recurring revenuePatient generation to dermatology practices using DTC techniques 3
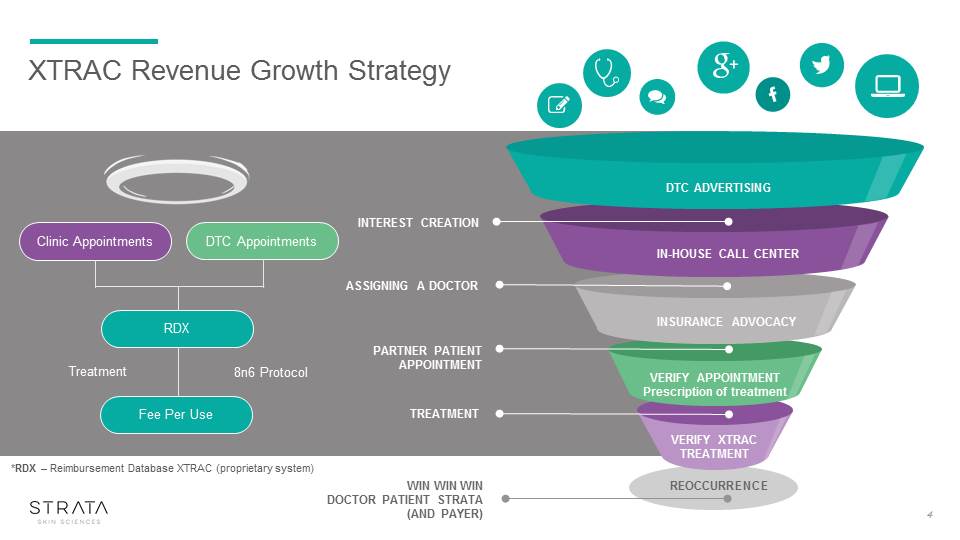
XTRAC Revenue Growth Strategy 8n6 Protocol Treatment *RDX – Reimbursement Database XTRAC (proprietary system) REOCCURRENCE DTC ADVERTISING ASSIGNING A DOCTOR PARTNER PATIENTAPPOINTMENT TREATMENT WIN WIN WIN DOCTOR PATIENT STRATA (AND PAYER) INTEREST CREATION IN-HOUSE CALL CENTER INSURANCE ADVOCACY VERIFY APPOINTMENTPrescription of treatment VERIFY XTRACTREATMENT Clinic Appointments DTC Appointments RDX Fee Per Use 4

XTRAC DTC Advertising 5
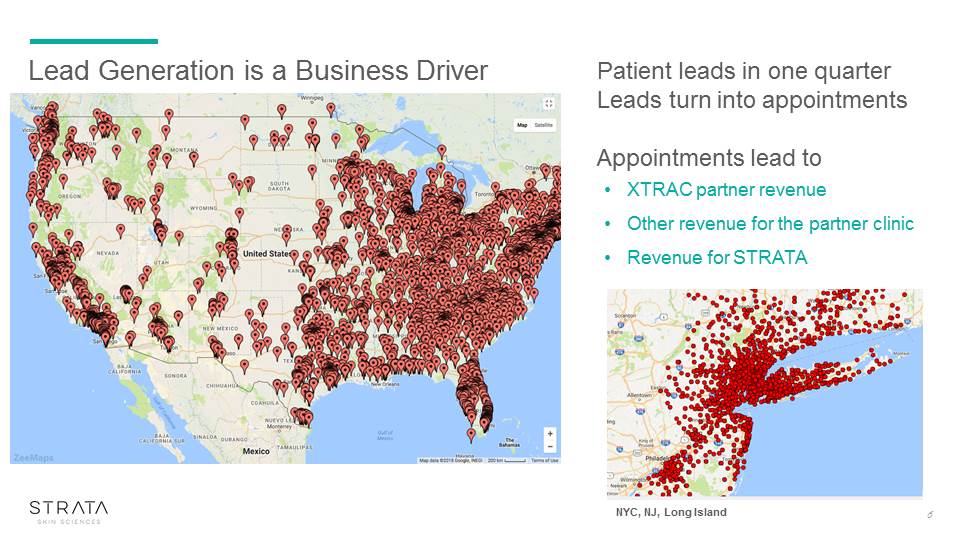
Lead Generation is a Business Driver Patient leads in one quarterLeads turn into appointmentsAppointments lead to XTRAC partner revenueOther revenue for the partner clinicRevenue for STRATA 6 NYC, NJ, Long Island

DTC Initial Results – Reduced Acquisition Costs Total Likes Total Reach 7
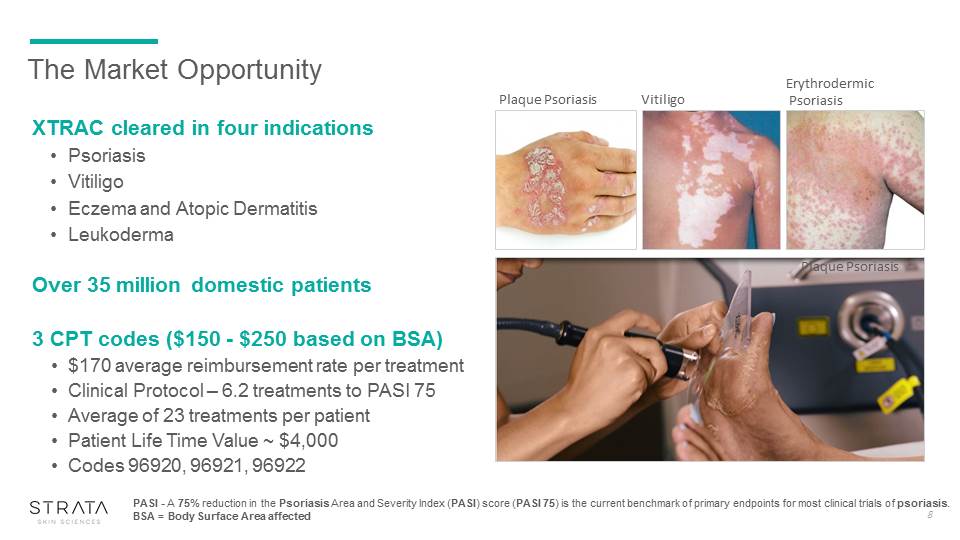
The Market Opportunity XTRAC cleared in four indications PsoriasisVitiligoEczema and Atopic DermatitisLeukodermaOver 35 million domestic patients3 CPT codes ($150 - $250 based on BSA)$170 average reimbursement rate per treatmentClinical Protocol – 6.2 treatments to PASI 75 Average of 23 treatments per patientPatient Life Time Value ~ $4,000Codes 96920, 96921, 96922 PASI - A 75% reduction in the Psoriasis Area and Severity Index (PASI) score (PASI 75) is the current benchmark of primary endpoints for most clinical trials of psoriasis.BSA = Body Surface Area affected 8 Plaque Psoriasis Vitiligo Erythrodermic Psoriasis Plaque Psoriasis

Excimer Laser Technology - XTRAC® / VTRAC® TechnologyXTRAC excimer laser uses highly targeted (308nm) beam of UVB light to treat affected skin area without damaging surrounding tissueVTRAC is a lamp based 308nm laserTreatment avoids side effects, cost of biologics and messy topicalsTreatments are safe and efficaciousMultiple FDA 510(k) and other international clearancesOver 500,000 patients treated Over 2,000 devices installed base WWCompelling partner clinic economicsXTRAC DTC provides a significant revenue sourceHalo effect of XTRAC DTC marketing platform Center of Excellence partner clinic revenue approx. $500,000/year Charge revenue on a per-use basis 9
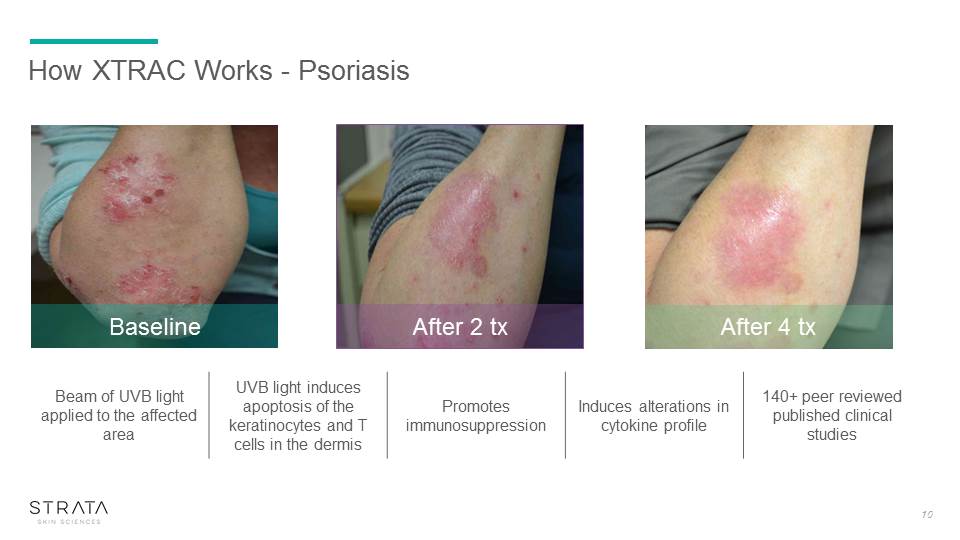
How XTRAC Works - Psoriasis After 4 tx After 2 tx Baseline Beam of UVB light applied to the affected area UVB light induces apoptosis of the keratinocytes and T cells in the dermis Promotes immunosuppression Induces alterations in cytokine profile 140+ peer reviewed published clinical studies 10
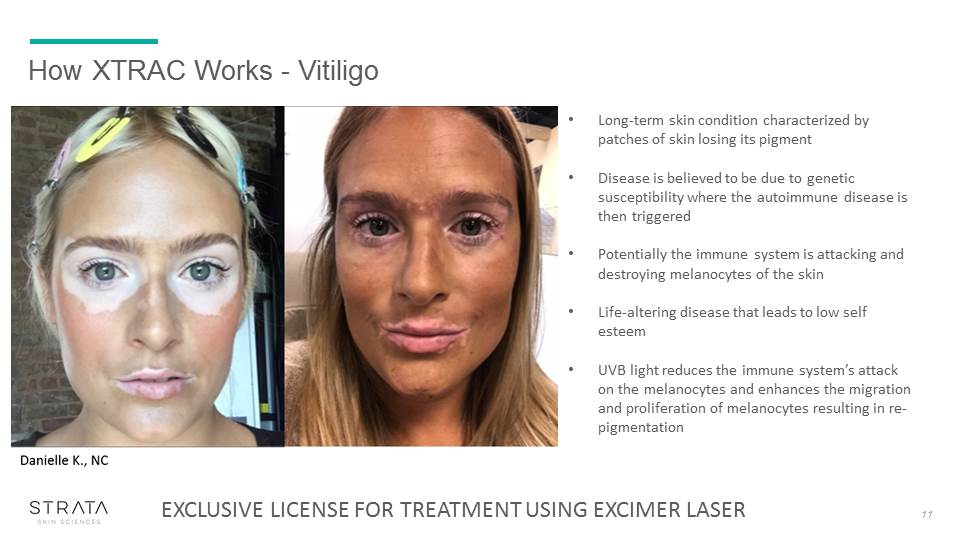
How XTRAC Works - Vitiligo 11 Long-term skin condition characterized by patches of skin losing its pigment Disease is believed to be due to genetic susceptibility where the autoimmune disease is then triggeredPotentially the immune system is attacking and destroying melanocytes of the skinLife-altering disease that leads to low self esteem UVB light reduces the immune system’s attack on the melanocytes and enhances the migration and proliferation of melanocytes resulting in re-pigmentation EXCLUSIVE LICENSE FOR TREATMENT USING EXCIMER LASER
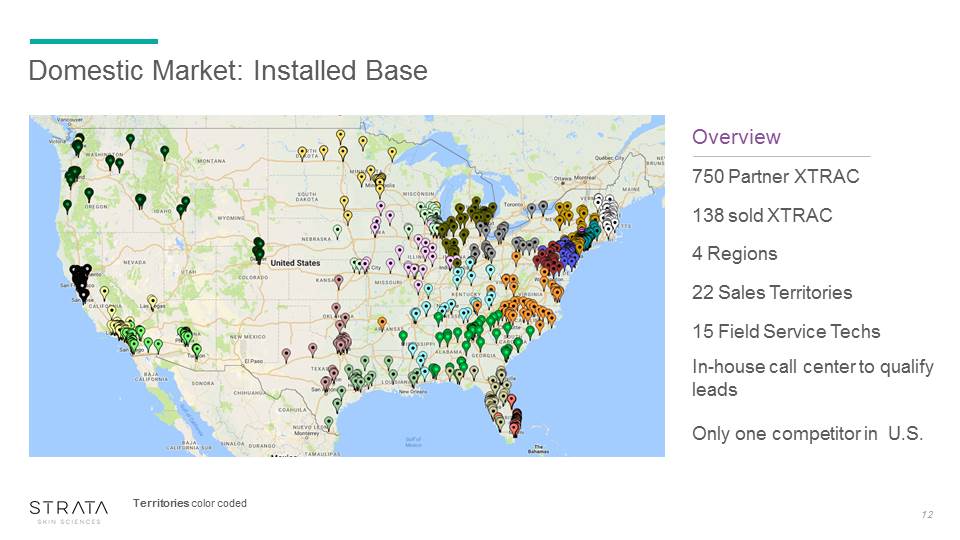
Domestic Market: Installed Base Overview 750 Partner XTRAC 138 sold XTRAC 4 Regions 22 Sales Territories 15 Field Service Techs In-house call center to qualify leadsOnly one competitor in U.S. 12 Territories color coded

International Market: Installed Base OUS sales are through distribution with no current recurring revenue model appliedMajor Markets – China, Japan, Saudi Arabia, South KoreaInsurance covered in most marketsUsage/device higher than domestic market – untapped potential in U.S.Over 1,200 OUS devices sold and in service Strategic Growth Target 13

Turnaround Strategy DOMESTIC MARKET OUS MARKETS Enhance technology offeringExpand geographical reach in certain markets Rebuild XTRAC value proposition for partner dermatologistsDTC is a revenue driver: Online and offline advertisement, in-house call centerPatient Communication: For both referrals and enrollment patientsPractice Development Programs: increase prescriptions for XTRACOptimize Clinical Outcomes: Deploy Education for High Dose/OTD and 8N6 as driversCurrent Excimer owners – Revisit “Comeback Program” (>250 dermatologists “came back” to recurring revenue model between 2012-2014)Expanding installed base (location within 10 miles of every patient)Improving XTRAC market share across all disease indications Revenue = # of lasers x # of treatments/pt x # patients x cost/tx x recurrence factor 14
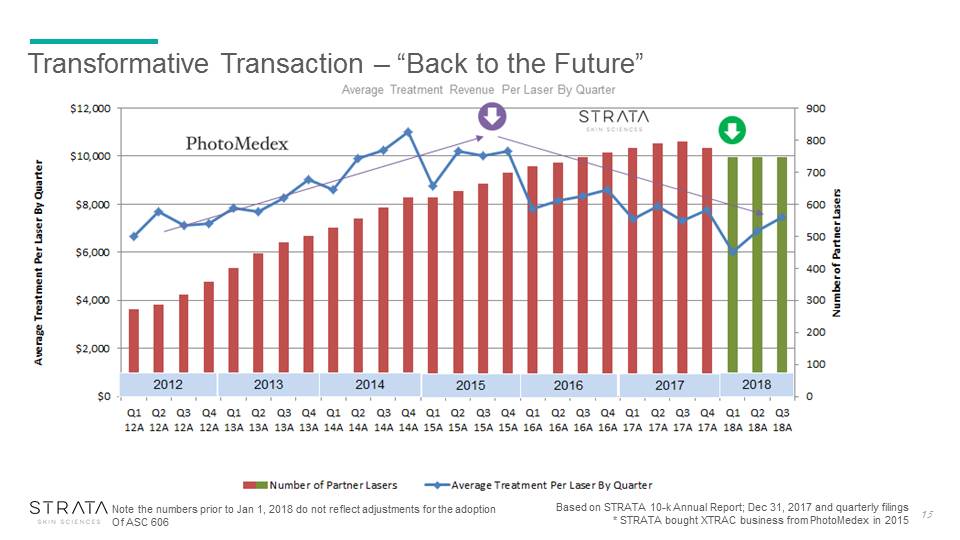
Transformative Transaction – “Back to the Future” Based on STRATA 10-k Annual Report; Dec 31, 2017 and quarterly filings* STRATA bought XTRAC business from PhotoMedex in 2015 15 Note the numbers prior to Jan 1, 2018 do not reflect adjustments for the adoption Of ASC 606
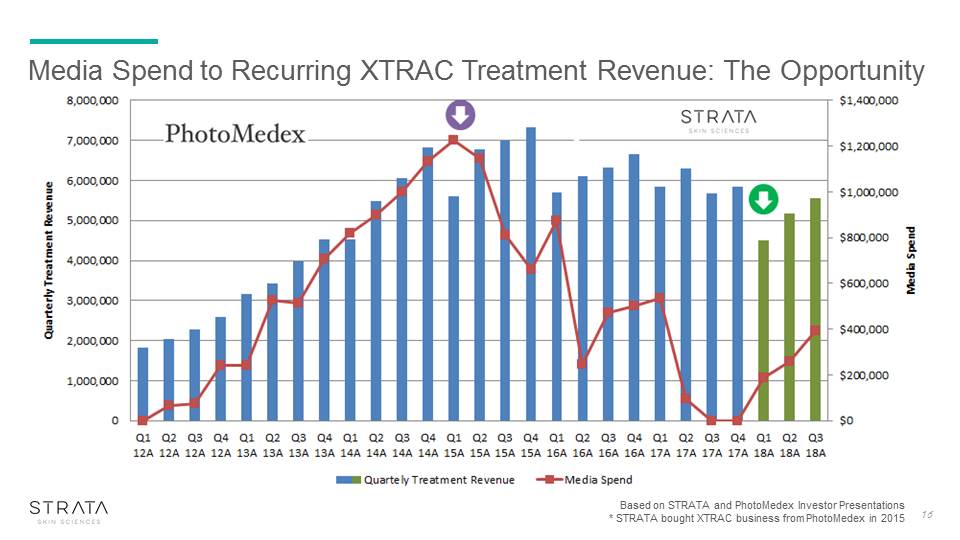
Media Spend to Recurring XTRAC Treatment Revenue: The Opportunity Based on STRATA and PhotoMedex Investor Presentations* STRATA bought XTRAC business from PhotoMedex in 2015 16
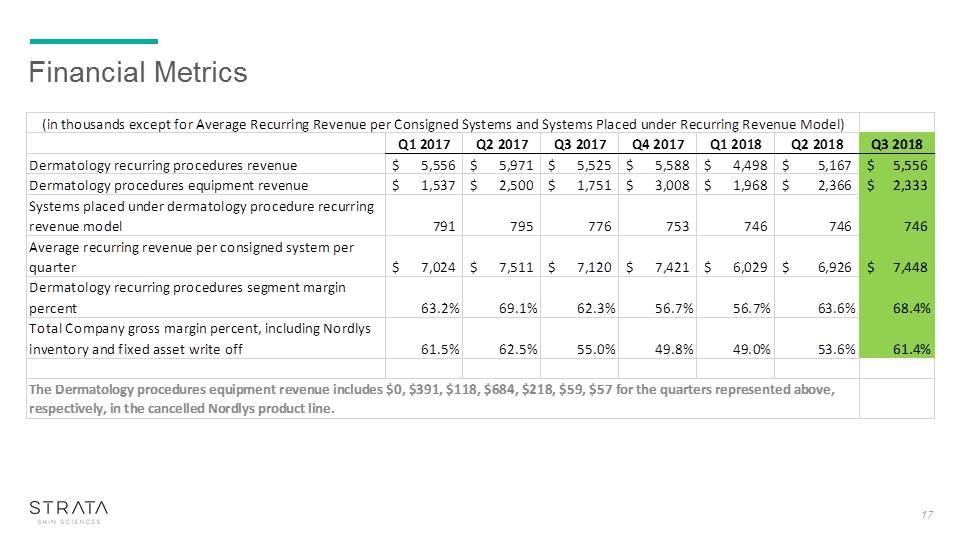
Financial Metrics 17

Changing Dynamic in Healthcare – Clinic Roll-Ups* Mapped 1,200 PE and private national group roll ups 38 different groups identifiedXTRAC placement penetration 86 - 8% - Huge OpportunityAugust 20, 2018 – Company announces “Strategic Agreement with a Large Private Equity Backed Group of Dermatology Clinics” 18 * Company estimates 86
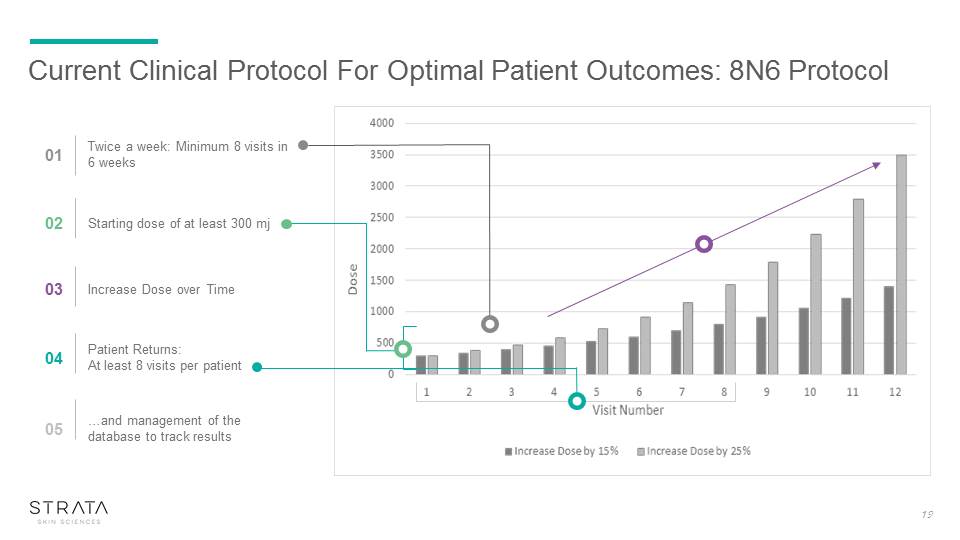
Current Clinical Protocol For Optimal Patient Outcomes: 8N6 Protocol 01 Twice a week: Minimum 8 visits in 6 weeks 02 Starting dose of at least 300 mj 03 Increase Dose over Time 04 Patient Returns: At least 8 visits per patient 05 …and management of the database to track results 19

Maximizing Dermatologist Partner Clinical Revenue Data collected directly from the lasers demonstrates the importance of following treatment guidelines Revenue = # of lasers x # of treatments/pt x # patients x cost/tx x recurrence factor 20

Optimal Therapeutic Dose (“OTD”) – 510(k) Received August/2018The Path to Optimal Clinical Outcomes and Patient Retention MECHANISMTip applies 4 simultaneous doses of energy to the patient’s plaque PURPOSETo minimize number of treatments to clearance USEDose allows provider to determine blister threshold unique to each patient and each plaque. 510(k) submission Approved Baseline After 2 tx Clinical Studies 21

October 2018 – OTD Clinical Study – Launch of XTRAC S3 OTD 92% of the included participants responded to treatment by achieving a reduction in mPASI score of 75% or moreCompared to current average of 16-20 treatments with NB-UVB 308nm devices, OTD treatment produced statistically significant mPASI improvement by the second treatment It took an average of four sessions for patients to reduce mPASI by 50% or more relative to baselineStatistical survival models predict mPASI scores to stay below 50% of baseline for 78 daysS3 Smaller, Faster, Smarter Exclusively pairs with the OTD ApproachAllows physicians to maximize results efficiently 22

Strong Leadership Team 23 Dr. Dolev Rafeli Chief Executive Officer Dr. Uri Geiger Chairman of the Board Matthew C. Hill Chief Financial Officer Dr. Daniel Siegel Consultant

Recent Financial Highlights Q2Q3 24 Total Revenue $7.5 million +17% versus Q1 2018 Recurring XTRAC Revenue $5.2 million +15% versus Q1 2018 Gross Margin 54% +5% versus Q1 2018 Dermatology Recurring Procedures Revenue Margin 64% +7% versus Q1 2018 Total Revenue $7.9 million +8% versus Q3 2017 Recurring XTRAC Revenue $5.6 million +1% versus Q3 2017 Gross Margin 61% +6% versus Q3 2017 Dermatology Recurring Procedures Revenue Margin 68% +6% versus Q3 2017 Cash and Cash Equivalents $15.9 million +$1.5 million versus Q2 2018
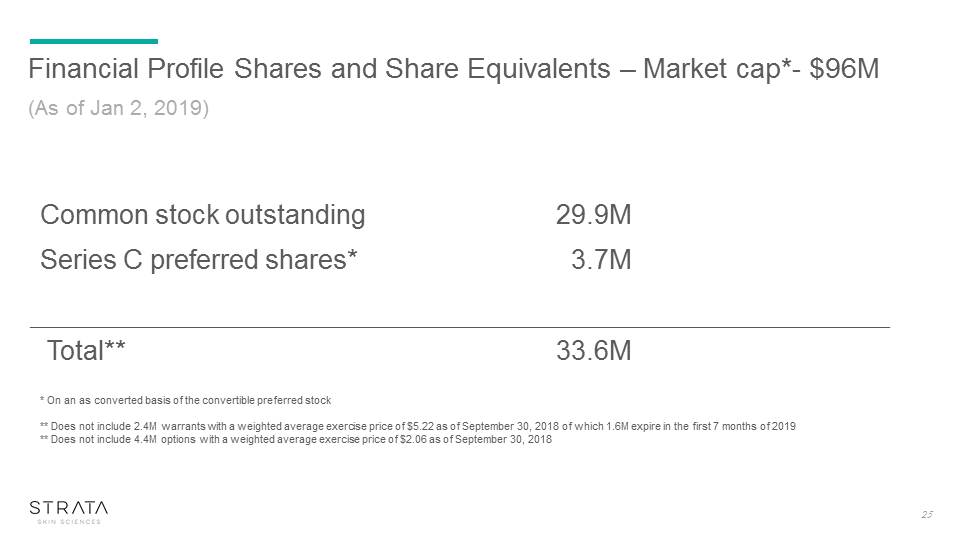
Financial Profile Shares and Share Equivalents – Market cap*- $96M (As of Jan 2, 2019) * On an as converted basis of the convertible preferred stock** Does not include 2.4M warrants with a weighted average exercise price of $5.22 as of September 30, 2018 of which 1.6M expire in the first 7 months of 2019** Does not include 4.4M options with a weighted average exercise price of $2.06 as of September 30, 2018 Common stock outstanding 29.9M Series C preferred shares* 3.7M Total** 33.6M 25

STRATA Investment Highlights Seasoned Management Team – with Deep Domain KnowledgeDr. Dolev Rafaeli, CEO returns with the Accelmed transaction Excimer Laser Technology for Dermatology IndicationsPsoriasis, Vitiligo, Eczema, Atopic Dermatitis and Leukoderma Large Market Opportunity Over 35 Million available patients in the United States alone Proven Strategy and Unique Business ModelBest in class XTRAC and VTRAC Excimer Laser technologies Features stable base of recurring revenuePatient generation to dermatology practices using DTC techniques 26

January 2019 Investor Presentation Driving Value to Dermatology Partners Matching Patients and Clinics