EXHIBIT 99.1

Investor Presentation November 2019 Driving Value to Dermatology Partners Matching Patients with Clinics © 2019 copyright Strata® Skin Sciences NASDAQ: SSKN

Safe Harbor Statement This presentation includes “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 that are subject to risks, uncertainties and other factors. All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including any statements of the plans, strategies and objectives of management for future operations; any statements regarding revenue growth, product development, product extensions, product integration or product marketing; any statements regarding continued compliance with government regulations, changing legislation, insurance or regulatory environments; any statements of expectation or belief and any statements of assumptions underlying any of the foregoing; any are risks and uncertainties related to successfully integrating the products and employees of the Company, as well as the ability to ensure continued regulatory compliance, performance and/or market growth. These risks, uncertainties and other factors, and the general risks associated with the businesses of the Company described in the reports and other documents filed with the SEC, could cause actual results to differ materially from those referred to, implied or expressed in the forward-looking statements. The Company cautions readers not to rely on these forward-looking statements. All forward-looking statements are based on information currently available to the Company and are qualified in their entirety by this cautionary statement. The Company anticipates that subsequent events and developments will cause its views to change. The information contained in this presentation speaks as of the date hereof and the Company has and undertakes no obligation to update or revise these forward-looking statements, whether as a result of new information, future events or otherwise. 2

STRATA Investment Highlights A Win-Win-Win CompanyFor patients – safest, most effective, no side effectsFor clinics – stable increased revenue baseFor Payers – the least expensive treatment availableExcimer Laser Technology for Dermatology IndicationsPsoriasis, Vitiligo, Atopic Dermatitis and LeukodermaLarge Market Opportunity 35 Million lives – $6 Billion annual revenueTreatment modality use growing > 25% per yearReimbursement - 3 unique CPT codes 25% target clinic penetrationUnique and Proven Business ModelBest in class XTRAC® and VTRAC ® Excimer Laser technologiesDomestic recurring revenue driven by DTC*OUS recurring revenue - Announced July 2019 - 2 placed Q3Providing “business in a box” for dermatology practicesGrowing Install base – low hanging fruit 3 *DTC – Direct To Consumer awareness and advertising
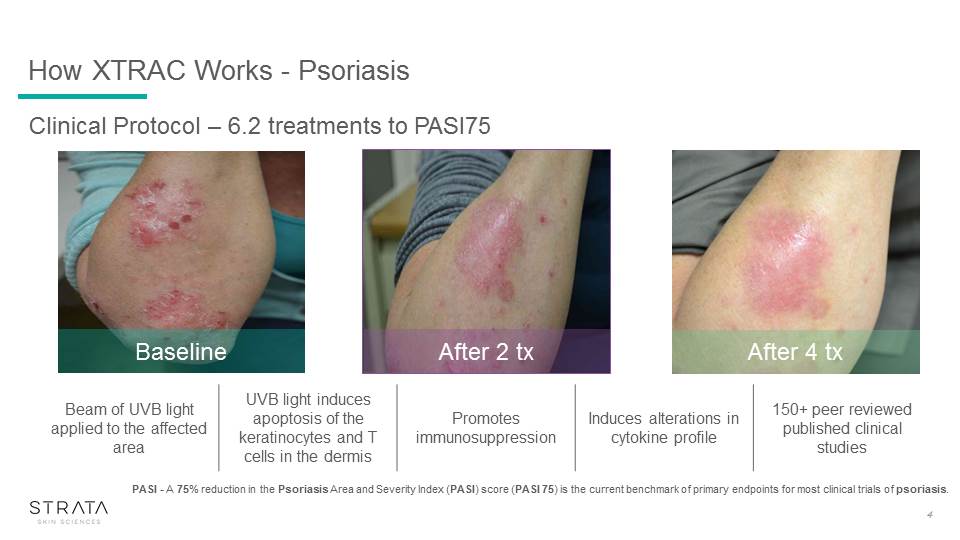
How XTRAC Works - Psoriasis 4 After 4 tx After 2 tx Baseline Beam of UVB light applied to the affected area UVB light induces apoptosis of the keratinocytes and T cells in the dermis Promotes immunosuppression Induces alterations in cytokine profile 150+ peer reviewed published clinical studies PASI - A 75% reduction in the Psoriasis Area and Severity Index (PASI) score (PASI 75) is the current benchmark of primary endpoints for most clinical trials of psoriasis. Clinical Protocol – 6.2 treatments to PASI75

How XTRAC Works - Vitiligo Long-term skin condition characterized by patches of skin losing its pigment Disease is believed to be due to genetic susceptibility where the autoimmune disease is then triggeredPotentially the immune system is attacking and destroying melanocytes of the skinLife-altering disease that leads to low self esteem UVB light reduces the immune system’s attack on the melanocytes and enhances the migration and proliferation of melanocytes resulting in re-pigmentation 5 STRATA HAS EXCLUSIVE LICENSE FOR TREATMENT OF VITILIGO USING EXCIMER LASER After 29 tx After 18 tx Baseline

XTRAC a True Partnership: A complete business solution Clinical Support Reimbursement Support Call Center Support Field Service Support Consumables and Parts Laser Upgrades Co-pay Support DTC Marketing 6

Domestic XTRAC Revenue Funnel 7 REOCCURRENCE DTC ADVERTISING ASSIGNING A DOCTOR PARTNER PATIENTAPPOINTMENT TREATMENT WIN WIN WIN DOCTOR PATIENT STRATA (AND PAYER) INTEREST CREATION IN-HOUSE CALL CENTER INSURANCE ADVOCACY VERIFY APPOINTMENTPrescription of treatment VERIFY XTRACTREATMENT Patient leads turn into appointmentsAppointments drive XTRAC partner revenueOther revenue for the partner clinicRevenue for STRATA

XTRAC helps patients Live Clear, Live Free… https://youtu.be/GdMBliZYy70 8
50,000 People See XTRAC Ads Daily 9
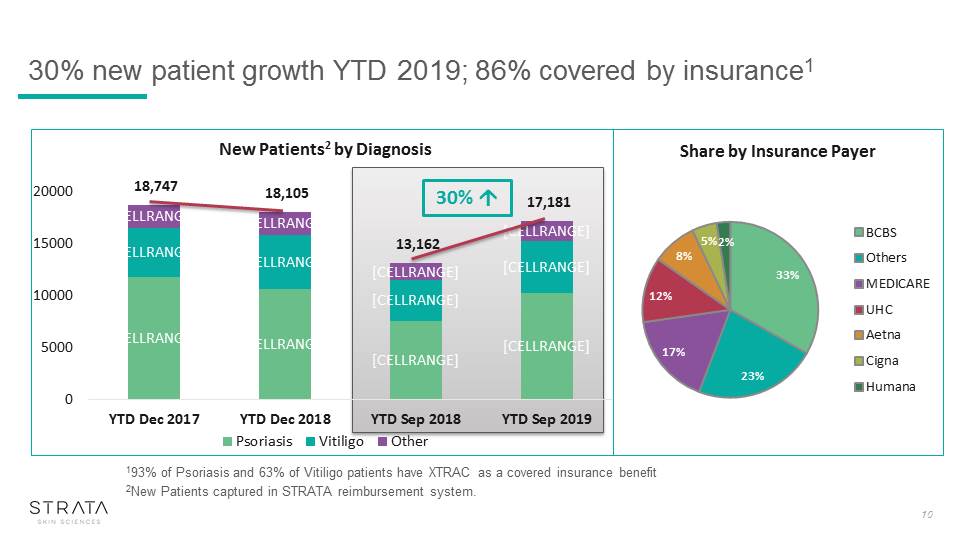
193% of Psoriasis and 63% of Vitiligo patients have XTRAC as a covered insurance benefit 2New Patients captured in STRATA reimbursement system. 30% 30% new patient growth YTD 2019; 86% covered by insurance1 10

STRATA’s Revenue Model 11 + 38 devices 26% growth 3% Growth 13% Growth 1Reflects only partners that purchase treatment codes on an as needed basis. Other partners purchase codes on a fixed monthly billing. 2Revenue is net of discounts and co-pay support As of September 30, 2018 and September 30, 2019
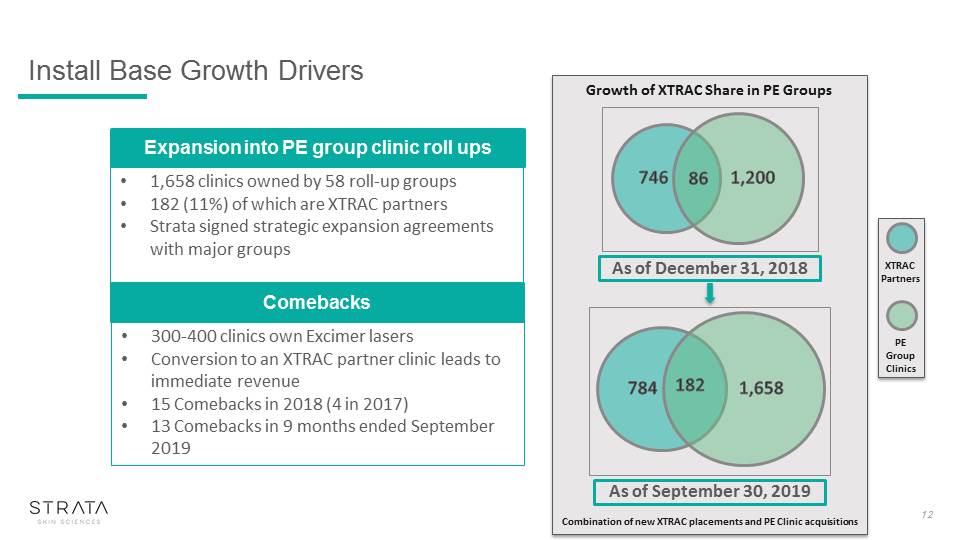
Growth of XTRAC Share in PE Groups Combination of new XTRAC placements and PE Clinic acquisitions Install Base Growth Drivers 12 1,658 clinics owned by 58 roll-up groups182 (11%) of which are XTRAC partnersStrata signed strategic expansion agreements with major groups Expansion into PE group clinic roll ups 300-400 clinics own Excimer lasers Conversion to an XTRAC partner clinic leads to immediate revenue15 Comebacks in 2018 (4 in 2017)13 Comebacks in 9 months ended September 2019 Comebacks 784 1,658 182 As of September 30, 2019 As of December 31, 2018 746 1,200 86 XTRAC Partners PE Group Clinics
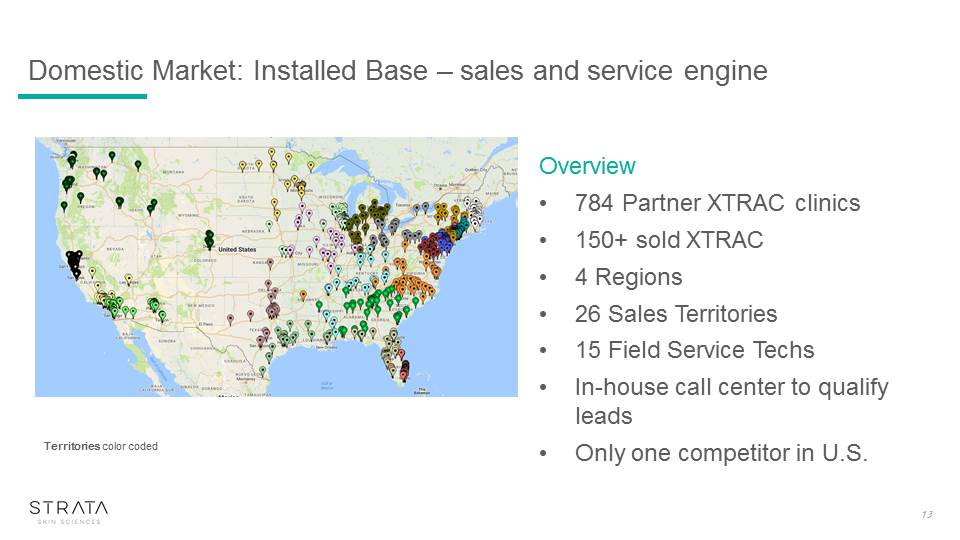
Domestic Market: Installed Base – sales and service engine 13 Territories color coded Overview784 Partner XTRAC clinics150+ sold XTRAC4 Regions26 Sales Territories15 Field Service TechsIn-house call center to qualify leadsOnly one competitor in U.S.

Partner Academic Institutions 14

International Market: Installed Base Strategic Growth TargetsMajor Markets – China, Japan, Saudi Arabia, South KoreaInsurance covered in most marketsUsage/device higher than domestic market – untapped potential in U.S.1,300 OUS devices sold and in servicePlacement recurring revenue model agreement executed for South Korea July 2019 – 2 devices placed Q3 15
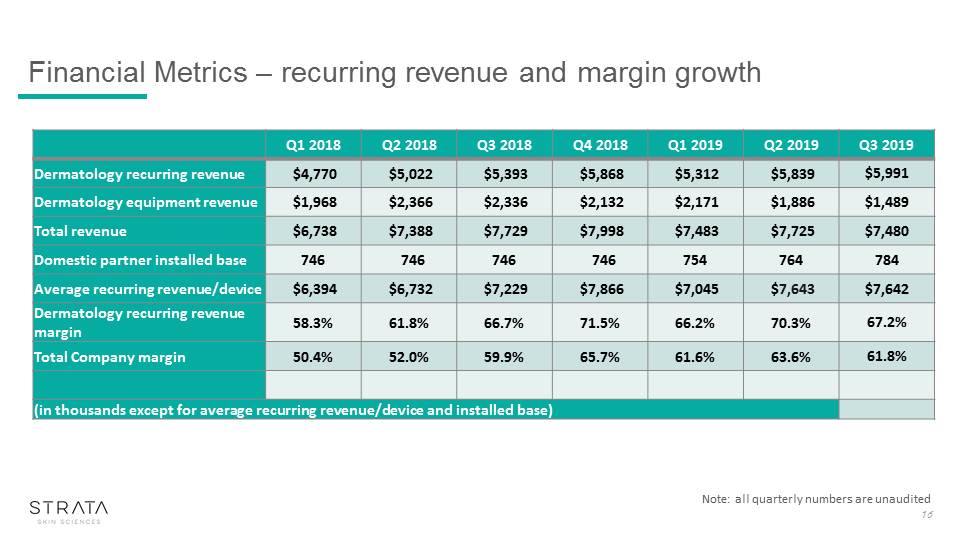
Financial Metrics – recurring revenue and margin growth 16 Note: all quarterly numbers are unaudited Q1 2018 Q2 2018 Q3 2018 Q4 2018 Q1 2019 Q2 2019 Q3 2019 Dermatology recurring revenue $4,770 $5,022 $5,393 $5,868 $5,312 $5,839 $5,991 Dermatology equipment revenue $1,968 $2,366 $2,336 $2,132 $2,171 $1,886 $1,489 Total revenue $6,738 $7,388 $7,729 $7,998 $7,483 $7,725 $7,480 Domestic partner installed base 746 746 746 746 754 764 784 Average recurring revenue/device $6,394 $6,732 $7,229 $7,866 $7,045 $7,643 $7,642 Dermatology recurring revenue margin 58.3% 61.8% 66.7% 71.5% 66.2% 70.3% 67.2% Total Company margin 50.4% 52.0% 59.9% 65.7% 61.6% 63.6% 61.8% (in thousands except for average recurring revenue/device and installed base)
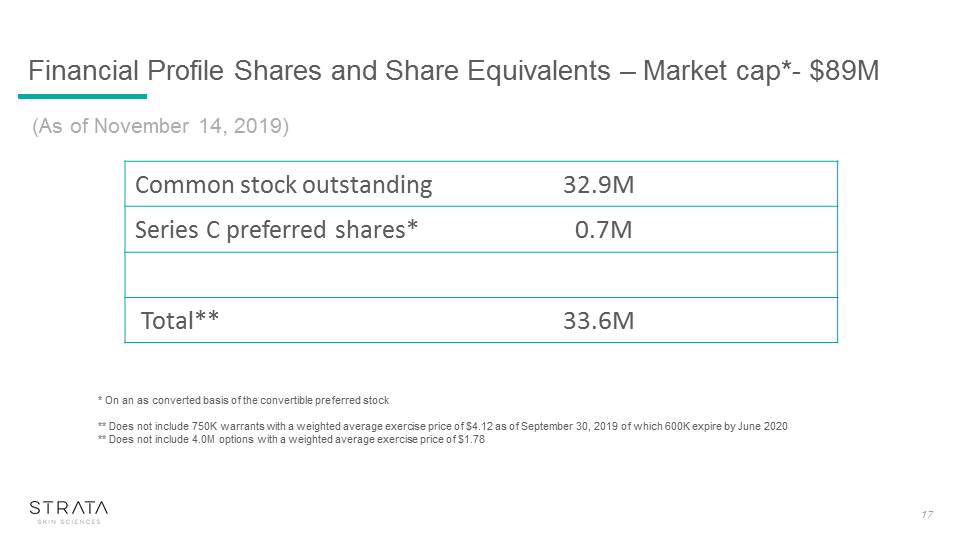
(As of November 14, 2019) * On an as converted basis of the convertible preferred stock** Does not include 750K warrants with a weighted average exercise price of $4.12 as of September 30, 2019 of which 600K expire by June 2020** Does not include 4.0M options with a weighted average exercise price of $1.78 Common stock outstanding 32.9M Series C preferred shares* 0.7M Total** 33.6M 17 Financial Profile Shares and Share Equivalents – Market cap*- $89M

STRATA Investment Highlights A Win-Win-Win CompanyFor patients – safest, most effective, no side effectsFor clinics – stable increased revenue baseFor Payers – the least expensive treatment availableExcimer Laser Technology for Dermatology IndicationsPsoriasis, Vitiligo, Atopic Dermatitis and LeukodermaLarge Market Opportunity 35 Million lives – $6 Billion annual revenueTreatment modality use growing > 25% per yearReimbursement - 3 unique CPT codes 25% target clinic penetrationUnique and Proven Business ModelBest in class XTRAC® and VTRAC ® Excimer Laser technologiesDomestic recurring revenue driven by DTC*OUS recurring revenue - Announced July 2019 - 2 placed Q3Providing “business in a box” for dermatology practicesGrowing Install base – low hanging fruit 18 *DTC – Direct To Consumer awareness and advertising

Investor Presentation November 2019 Driving Value to Dermatology Partners Matching Patients with Clinics © 2019 copyright Strata® Skin Sciences NASDAQ: SSKN

Appendix 20

Additional Creative 21

XTRAC® Excimer laser is FDA cleared for Psoriasis, Vitiligo, Atopic Dermatitis and Leukoderma. It uses a highly targeted beam of 308nm UVB to treat affected skin without damaging surrounding healthy tissue. Treatment has minimal side effects, avoids messy topicals and high cost of biologics. #1 Prescribed Targeted Therapy for Psoriasis and Vitiligo by Dermatologists 22

Continuous technology investment and business growth 23 510 (K) FDA Approval AL-7000 Ultra Velocity 400 Velocity 700 Velocity 7 Series S3 2000 2005 2008 2010 2014 2018 CPT code Approval DTC platform creation 510 (K) FDA Approval (MMD)* Full Insurance Reimbursement 2012 *MMD tip is a diagnostic accessory for XTRAC used in optimal therapeutic dose (OTD) protocol that helps patients achieve Optimal Clinical Outcomes leading to increased Patient Retention
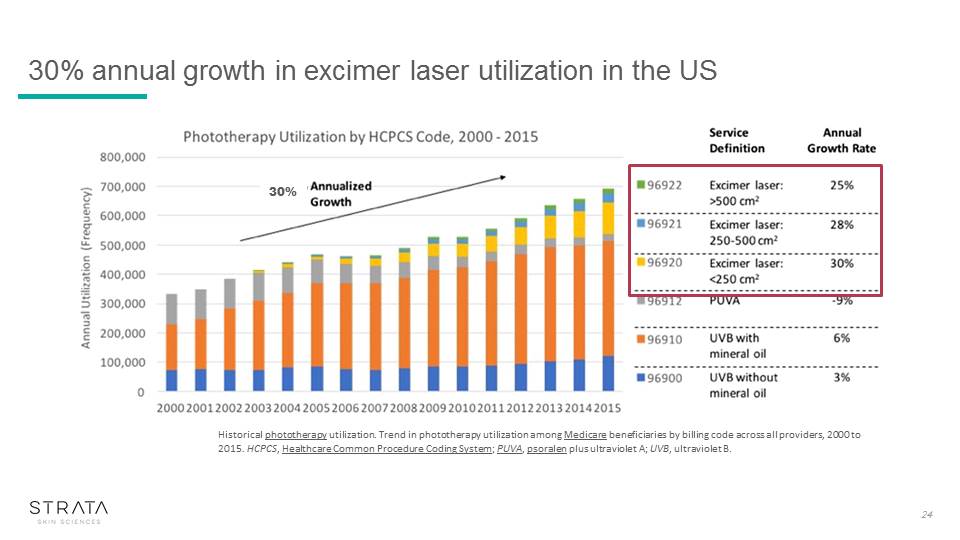
24 Historical phototherapy utilization. Trend in phototherapy utilization among Medicare beneficiaries by billing code across all providers, 2000 to 2015. HCPCS, Healthcare Common Procedure Coding System; PUVA, psoralen plus ultraviolet A; UVB, ultraviolet B. 30% 30% annual growth in excimer laser utilization in the US

Multicenter Psoriasis Study1 Scalp Study3 124 patients were enrolled and 80 completed the protocol. 72% achieved at least 75% clearing in an average of 6.2 treatments. 84% of patients reached improvement of 75% or better after 10 or fewer treatments. 50% of patients reached improvement of 90% or better after 10 or fewer treatments. Common side effects included erythema, blisters, hyperpigmentation, and erosions, but were well tolerated. All patients improved. 17/35 (49%) of patients cleared>95% (mean: 21 treatments; range: 6-52) and 16/35 (45%) cleared 50-95%. Conclusion - The excimer laser is a successful approach to treatment of psoriasis of the scalp being a simple treatment that can be performed in a short period of time and which has a high rate of effectiveness. Sample studies from 150+ clinical studies Vitiligo Study2 Out of 221 vitiligo patches treated, 50.6% showed 75% pigmentation or more, 25.5% achieved 100% pigmentation of their patches, and 64.3% showed 50% pigmentation or more. Lesions on the face responded better than lesions elsewhere. Conclusion: The 308-nm xenon chloride excimer laser is an effective and safe modality for the treatment of vitiligo, with good results achieved in a relatively short duration of time. Pediatric Study4 4 children with a mean age of 11.0 years and 12 adults with a mean age of 48.8 years completed the protocol. Both the children and the adult groups yielded a significant decrease in psoriatic severity scores of their respective target lesions. The children’s group had a greater reduction (91.3% reduction) as compared to the adult treatment group (61.6% reduction). 1Multicenter Psoriasis Study - Feldman SR, Mellen BG, Housman TS, Fitzpatrick RE, Geronemus RG, Friedman PM, Vasily DB, Morison WL. Efficacy of the 308-nm excimer laser for treatment of psoriasis: Results of a multicenter study. J Am Acad of Dermatol; vol. 46, no. 6, June 2002, pp. 900-906 2Vitiligo Study - Suhail Hadi, Patricia Tinio, Khalid Al-Ghaithi, Haitham Al-Qari, Mohammad Al-Helalat, Mark Lebwohl, and James Spencer. Photomedicine and Laser Surgery. Treatment of Vitiligo Using the 308-nm Excimer Laser. Jun 2006.ahead of printhttp://doi.org/10.1089/pho.2006.24.3543Scalp Study - Morison WL, Atkinson DF and Werthman L. Effective treatment of scalp psoriasis using the excimer (308nm) laser. Photodermatol Photoimmunol Photomed 2006; 22: 181-1834Pediatric Study - Pahlajani N, Katz BJ, Lonzano AM, Murphy F and Gottlieb A. Comparison of the Efficacy and Safety of the 308nm Excimer laser for the Treatment of Localized Psoriasis in Adults and in Children: A Pilot Study. Pediatric Dermatology Vol. 22 No. 2, March/April 2005, pp. 161-165Induration Protocol Study - Taneja A, Trehan M, Taylor C. 308-nm Excimer Laser for the Treatment of Psoriasis – Induration-Based Dosimetry. Arch Dermatol, Vol. 139, June 2003, pp. 759-764High Dose Protocol Study - Michael Abrouk,1 Ethan Levin,2 Merrick Brodsky,1 Jessica R Gandy,1 Mio Nakamura,2 Tian Hao Zhu,3 Benjamin Farahnik,4 John Koo,2 and Tina Bhutani2 Excimer laser for the treatment of psoriasis: safety, efficacy, and patient acceptability. Published online 2016 Dec 12. doi: 10.2147/PTT.S105047 25 25

Maximizing Dermatologist Partner Clinical Revenue 26 Data collected directly from the lasers demonstrates the importance of following treatment guidelines Revenue = # of lasers x # of treatments/pt x # patients x cost/tx x recurrence factor

Source: National Psoriasis Foundation American Academy of Dermatology Psoriasis Clinical Guideline XTRAC a preferred treatment for 90% of psoriasis patients (<10% BSA) 27

Opportunity map for XTRAC Indications Number of Strata Patients* Prevalence Source Atopic dermatitis 1,200 18 Million National Eczema Association Vitiligo 9,000 2-5 Million Vitiligo Support Psoriasis 18,000 7.5 Million NPF Topicals/ Targeted Phototherapy Systemics/Biologics/UVB Booth Recommended Actual XTRAC Biologics *Estimated 28

Source: Findings From the National Psoriasis Foundation Surveys, 2003-2011 (n=5604) Source: Real-world health outcomes study sponsored by Eli Lilly (n=2200) Phototherapy has highest patient perceived treatment effectiveness 29

Leads, Appointments, RDX Charts YTD Dec 2015 YTD Dec 2016 YTD Dec 2017 YTD Dec 2018 YTD Sep 2018 YTD Sep 2019 Leads 36,473 21,296 7,269 22,213 13,485 37,701 Appointments 10,032 6,524 2,563 3,856 2,536 4,711 RDX Charts 26,237 23,481 18,747 18,105 13,162 17,181 30
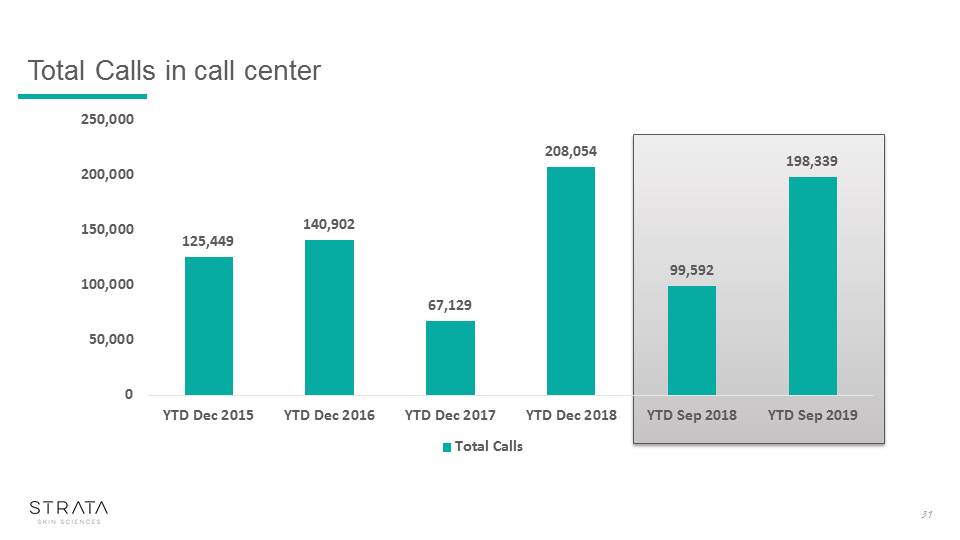
Total Calls in call center 31

XTRAC adds (delegated) recurring revenue driver for its partners Treatment Patient Type Reimbursement Details Annual Revenue per Patient Can be delegated** XTRAC Mild / moderate / severe CPT Code 96920-96922 ($168-$251) average $186; 16 tx/course (2/yr) and Office Visit -$70 (2/Yr) $6,092 Phototherapy* Moderate / severe Office visit - $70 (2/yr) $140 X Biologics Moderate / severe Office visit - $70 (6/yr) $420 X Systemics Moderate / severe Office visit - $70 (3/yr) $210 X Topicals Mild / moderate Office visit - $70 (3/yr) $210 X CPT CODE Description 2018 National AverageMedicare Payment Rate 96920 Laser treatment for inflammatory skin diseases, (psoriasis); total area less than 250 sq cm $168.12 96921 Laser treatment for inflammatory skin diseases, (psoriasis); total area between 250 – 500 sq cm $184.32 96922 Laser treatment for inflammatory skin diseases, (psoriasis); total area greater than 500 sq cm $250.92 Number of Treatments / Weeks 20 30 # of Weeks / Year 48 48 Total # of Treatments / Year 960 1,440 Average Revenue Per Treatment $186 $186 Physician Gross Revenue (Annual) $178,485 $267,728 *Phototherapy Center Revenue – CPT Code 96910 $40 per tx; 30 tx/course (2/yr) - $2,400**Subject to state legislation 32 32

How XTRAC works for offices: $250 revenue in less than 7 minutes *Subjected to state regulation Established CPT Codes 2019 National Medicare Average Rate 96920 - $167.2296921 - $183.4496922 - $249.03 33