
Exhibit 99.1
Corporate Overview
Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
Lehman Healthcare Conference
March 8, 2006
NYSE: WST
www.westpharma.com

Certain statements contained in this presentation and certain statements that may be made by management of
the Company orally during this presentation are forward-looking statements that set forth anticipated results
based on management’s plans and assumptions. Such statements give our current expectations or forecasts
of future events – they do not relate strictly to historical or current facts. In particular, these include statements
concerning future actions, future performance or results of current and anticipated products, sales efforts,
expenses, the outcome of contingencies such as legal proceedings and financial results. We have tried,
wherever possible, to identify such statements by using words such as “estimate,” “expect,” “intend,”
“believe,” “plan,” “anticipate” and other words and terms of similar meaning in connection with any discussion
of future operating or financial performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or
uncertainties materialize, or if underlying assumptions are inaccurate, actual results could differ materially from
past results and those expressed or implied in any forward-looking statement. You should bear this in mind as
you consider forward-looking statements. We cannot predict or identify all such risks and uncertainties, but
factors that could cause the actual results to differ materially from expected and historical results include the
following: sales demand; timing and commercial success of customers’ products incorporating our products
and services, including specifically, the Exubera® Inhalation-Powder insulin device; our ability to pass raw-
material cost increases on to customers through price increases; maintaining or improving production
efficiencies and overhead absorption; constraints on manufacturing capacity in the face of anticipated
demand; the availability of labor to meet increased demand; competition from other providers; the successful
integration of acquired businesses; average profitability, or mix, of products sold in a reporting period;
financial performance of unconsolidated affiliates; strength of the U.S. dollar in relation to other currencies,
particularly the Euro, UK Pound, Danish Krone, Japanese Yen and Singapore Dollar; interruptions or
weaknesses in our supply chain, which could cause delivery delays or restrict the availability of raw materials
and key bought-in components; raw-material price escalation, particularly petroleum-based raw materials, and
energy costs; and availability and pricing of materials that may be affected by vendor concerns with exposure
to product-related liability.
The Company assumes no obligation to update forward-looking statements as circumstances change.
Investors are advised, however, to consult any further disclosures the Company makes on related subjects in
the Company's 10-K, 10-Q and 8-K reports.
Safe Harbor Statement

Each and every day
millions of West
products are used to
enhance healthcare
around the world.
Who are we?

Corporate Profile
World's premier
manufacturer of
components and systems
for injectable drug delivery
Stoppers and seals for vials
Disposable components used
in syringe, IV, blood collection
and diagnostic systems
Founded in 1923
HQ in Lionville, PA
2005 sales $700M
Market capitalization
approx $1.05B

Diverse Customer Base
Company Estimated Market Share: 70% in Pharma; 70% in Device; 95% in Biotech
medtronic
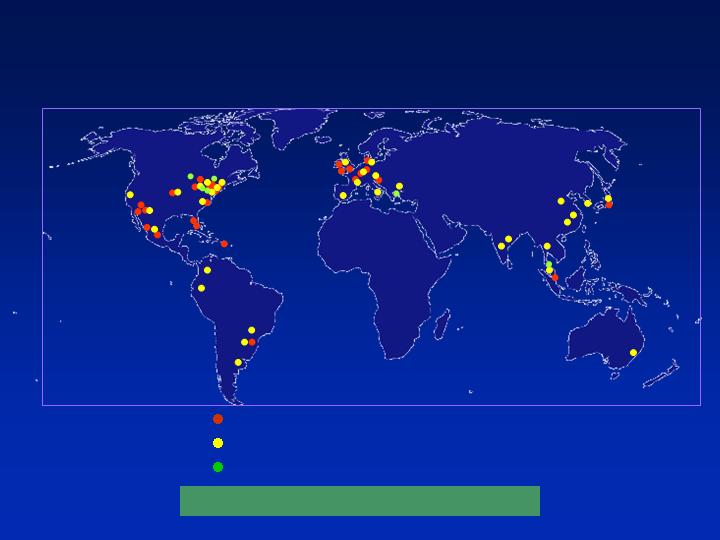
32 manufacturing sites
34 sales offices
7 technical centers
5,500 employees worldwide
Global Presence

Key Company Developments
Acquisition of Monarch Laboratories
Acquisition of The Tech Group
Acquisition of Medimop
Sale of GFI Clinical Unit
Sale of Contract Manufacturing Operations
New management team
Restructuring and increased CAPEX
Sale of OTC Research Services Group
Kinston explosion
Kinston restart
2-for-1 stock split
Divestiture of Drug Delivery
FY 2005
FY 2004
FY 2003
FY 2002
FY 2001
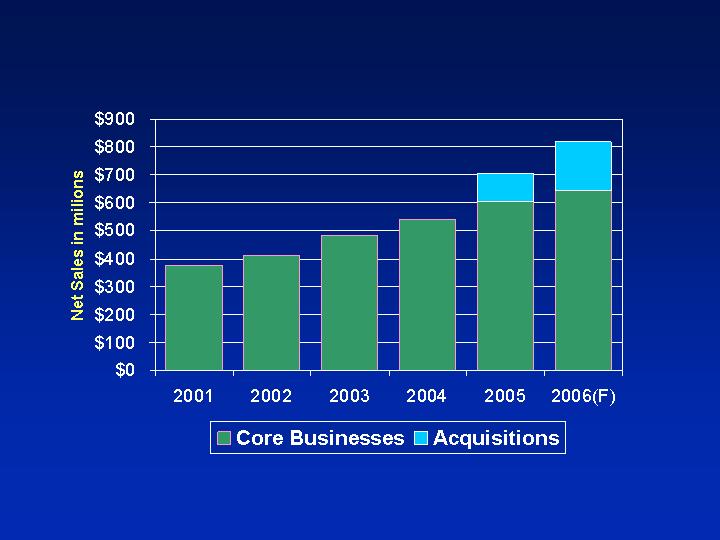
Strong Sales Growth
2002-2005
FY05 Net sales: Approx. $700 MM
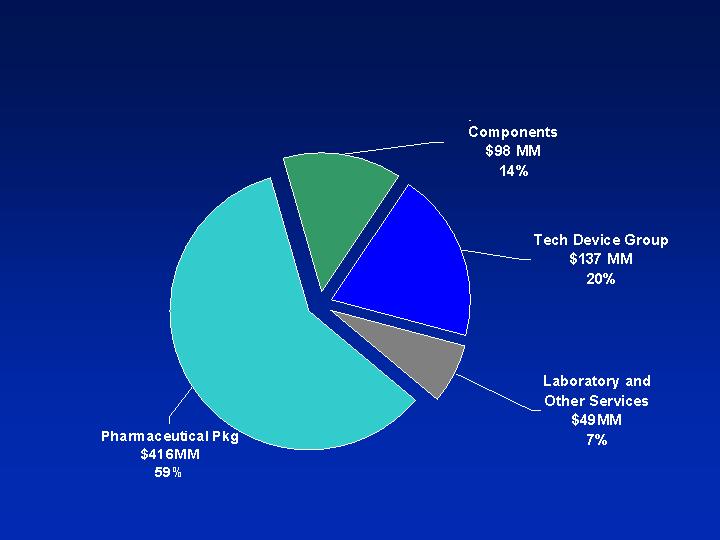
Business Segments

Pharmaceutical Systems Profile
8-12%
0-2%
6-8%
Our Growth
(2005-2007)
10-50%
Small
Not determined
Lab and
Other
Services
10-20%
Market Leader
$150 million,
Growing 0-2%
Disposable
Medical
Device
Components
25-50+%
Market Leader
$1 to $1.5 billion.
Growing 2-5%
Pharma
Systems
Gross Margin
Range
Our Share
Market Size*/
Long-Term
Growth Rate
* Company estimates
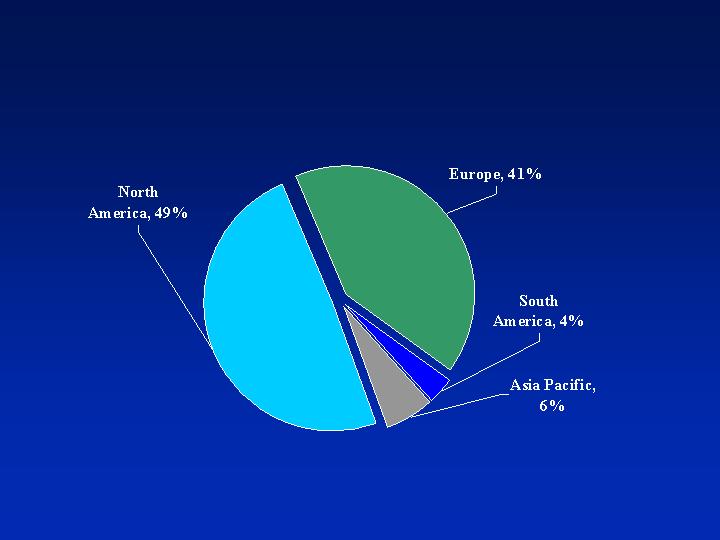
Geographic Sales Mix
(2005, based on point of sale)

Corporate Growth Strategy
Core Pharmaceutical Systems Business
Maximize the value of West’s core business
(Company Estimated Market: $1.1 BN)
Market segmentation
Generate maximum value from key growth drivers
New product innovation
Lean manufacturing
Geographic expansion
Strategic acquisitions
MediMop Medical Projects Ltd

Growth Drivers
Global demographics
Diabetes

Diabetes
Components
for Pen System Applications
Components for
Traditional System
Applications
Entire Systems

Growth Drivers
Global demographics
Diabetes
Growth in biotechnology drugs
Oncology
Pre filled syringe systems
Reconstitution systems
Medimop Medical Projects Ltd

Biotechnology/Oncology
Closures and Components for
Traditional and Prefilled Systems
Seals
Syringe plungers, needle shields
Flip-Off ® Buttons
Stoppers
FluroTec ® and Barrier Coatings
Westar®
Processing
Closures and Components for
Biotechnology and Oncology Systems
Reconstitution Aides
Biotechnology/Oncology

Medimop Product Portfolio
Vial Adapters
Needleless Drug
Transfer and Mixing

Growth Drivers
Global demographics
Diabetes
Growth in biotechnology drugs
Oncology
Anti-counterfeiting needs

Growth Opportunities
Closures with built-in product tracking, authentication
and anti-counterfeiting features

West’s Competitive Edge
Unmatched experience/expertise: drug material
interface
Ability to source components from multiple locations
Protected IP: West’s components and systems
Regulatory barrier to entry: NDA and ANDA filing
must include reference to all packaging/components
in contact with the drug:
1.
West Drug Master File (DMF) 1546 is confidential
2.
West DMF includes functionality data (multi-year studies)
3.
All primary package changes require new stability/ functionality
studies for new filing

Corporate Growth Strategy
Tech Group Business
Build market share in multi-component systems
for drug delivery
(Company Estimated Market: $4.5 BN)
Leverage customer base
Develop a portfolio of proprietary systems for injectable,
transmucosal, and pulmonary delivery
Pursue selected consumer opportunities
License or acquire innovative technologies
Pursue strategic acquisitions
The Tech Group
The Tech Group
West Device Group
FY04 Sales: $74.2 MM
FY04 Sales: $65.0 MM
Tech Complements West

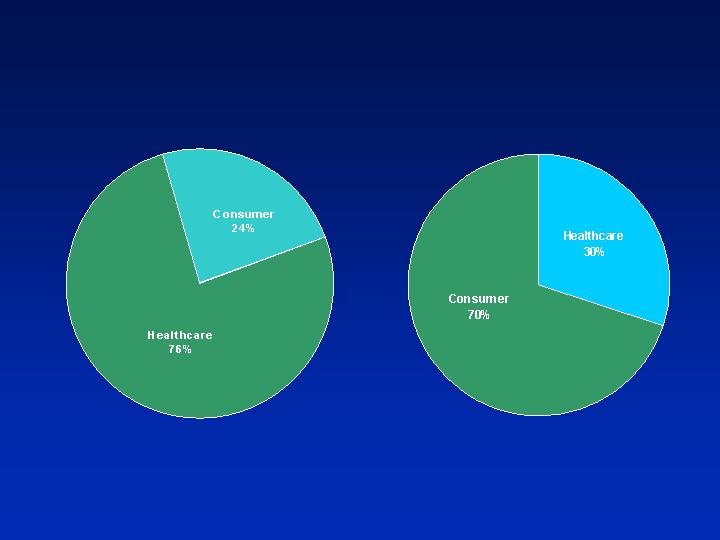
Tech Customer Base
Pharmaceutical
Device
Consumer

Tech Group Growth Drivers
Organic Growth
New Product Portfolio
Insulin delivery systems

Tech Product Portfolio
Health Care
Consumer

Joint development with Pfizer & Nektar
Class III medical device part of NDA
Recent events
Received EMEA and FDA approval for marketing
Initial launch targeted for mid year
Key questions are:
Market acceptance – Doctors and patients
Patient acceptance may expand market
Increased patient compliance – targeted for non
compliant daibetic population
(1) “Exubera” is a registered trademark of Pfizer, Inc.
Exubera®(1) Inhaled Insulin

2006 – Where We Stand
Key elements of the “Sustainable Growth” strategy
are now in place:
West’s core pharmaceutical systems business is an
established global franchise
Tech Group adds new customers, products and
capabilities in the key device segment
Medimop adds new technologies and products in the
key biotechnology and oncology markets

Execute business plan and strategy
Retain focus on growth of core injectable business
Capitalize on Tech and Medimop programs and opportunities
Generate margin expansion through lean programs
Pursue opportunities for geographic expansion
China – India – Brazil – Russia/Eastern Europe
Continue to innovate, develop and acquire technologies
“Adaptable” integration (e.g. TagSys – RFID)
Product line expansion (e.g. Tech, Medimop)
Next-generation systems (e.g. Silicone Free CZ PFS)
2006 Operating Priorities

Investment Considerations

Investment Considerations
Core injectable packaging and delivery systems business
is globally established
Significant regulatory and capital barriers to entry
Strong, diversified customer base
West’s product development cycle mirrors new drug development
timeline
Value-added products, technologies and services that enhance
growth potential
Recent acquisitions further enhance growth potential,
leverage manufacturing and industry expertise
Market drivers favor continued growth
Strong balance sheet
Management incentives strongly tied to creation of
shareholder value

Full Year 2005 Results
$1.39
$1.09
EPS–contg Ops
$ 43
$ 30
Income from
continuing Ops
10.3%
5.3%
17.5%
8.9%
4.4%
16.4%
% of sales
72
9
94
48
3
79
Operating Profit
27.5%
13.4%
31.6%
28.8%
13.6%
30.5%
% of sales
193
23
170
156
9
147
Gross Profit
9.0%
4.4%
11.9%
% growth
$ 700
$ 170
$ 538
$ 542
$ 68
$ 481
Net Sales
Consolidated
(1) (2)
Tech
Group (1)
Pharm
Systems
Consolidated
(2)
Tech
Group
Pharm
Systems
2005
2004
($ millions,
except EPS)
Note(1) – Includes Tech acquired business from May 2005 of $99. 2005 sales grew by 4.4% over 2004 levels
excluding the Tech acquired business. The Tech acquired business grew 30% in 2005 over its prior year.
Consolidated 2005 sales, excluding acquisitions and currency increased 9% over prior year sales.
Note (2) – Consolidated net sales excludes inter-segment eliminations of $7 and $9, respectively.

Guidance
2006 revenue forecast - $810- $830 million
Expect gross margins of 32% from Pharm Systems
segment and 16.5% from Tech segment
EPS from continuing operations: $1.60 - $1.70
Estimates exclude first quarter pre-tax charge from
refinancing our Senior debt of approximately $6mm;
currency effects; any significant additional increases to raw
material and energy costs
Includes $0.10- $0.17 of after tax operating profit from
Exubera® device sales
Includes $5 mm of additional R&D expenditures
Includes $4 mm of increased pension expense
As of February 21, 2006, without update

Financial Objectives
Grow sales 6-8% in our core injectable
business before the effects of currency and
acquisitions
Improve margins by using Lean to eliminate
waste and control discretionary spending
Selectively invest in innovative new products
and technologies
Reduce debt to total cap ratio, exclusive of any
new acquisition-related debt
Create returns on invested capital in excess of
our Cost of Capital

Management Performance Metrics
Short term (Annual)
Corporate – EPS, cash flow
Operations – net sales, operating profit, cash flow
Long term
50% restricted stock, 50% options
Compounded annual growth rate (CAGR)
Return on invested capital (ROIC)
Stock ownership guidelines for all executive
officers and Directors

Summary
2006 should produce strong results
Revenue growth to $810-$830million
15-20 % EPS growth ($1.60-$1.70) excluding impact of debt
refinancing
Management focus is on execution of business plan
Optimize growth and profitability of the core business
Deliver benefits of recent acquisitions
Continue emphasis on margin improvement via lean
initiative
Invest in innovation

Corporate Overview
Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
Lehman Healthcare Conference
March 8, 2006
NYSE: WST
www.westpharma.com