
Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
West Pharmaceutical Services
William J. Federici
Vice President & Chief Financial Officer
West Pharmaceutical Services
NYSE: WST
westpharma.com
CL King's 5th Annual
BEST IDEAS CONFERENCE 2007

Certain statements contained in this presentation and certain statements that may be made by management of the
Company orally during this presentation contains some forward-looking statements that set forth anticipated results based on
management’s plans and assumptions. Such statements give our current expectations or forecasts of future events – they do
not relate strictly to historical or current facts. We have tried, wherever possible, to identify such statements by using words
such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms of similar meaning in
connection with any discussion of future operating or financial performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or uncertainties
materialize, or if underlying assumptions are inaccurate, actual results could differ materially from past results and those
expressed or implied in any forward-looking statement. You should bear this in mind as you consider forward-looking
statements. We cannot predict or identify all such risks and uncertainties, but factors that could cause the actual results to
differ materially from expected and historical results include the following: Sales demand; the timing, regulatory approval and
commercial success of customers’ products incorporating our products and services, including specifically, the Exubera®
Inhalation-Powder insulin device; customers’ changes to inventory requirements and manufacturing plans that alter existing
orders or ordering patterns for our products; our ability to pass raw-material cost increases on to customers through price
increases; maintaining or improving production efficiencies and overhead absorption; physical limits on manufacturing
capacity that may limit our ability to satisfy anticipated demand; the timeliness and effects of capacity expansions, including
the effects of delays associated with construction, availability and price of capital goods, and necessary internal, governmental
and customer approvals; the availability of labor to meet increased demand; competition from other providers; the timely and
successful negotiations of sales contracts with four of the Company’s largest customers during the second half of 2007,
average profitability, or mix, of products sold in any reporting period; financial performance of unconsolidated affiliates;
strength of the U.S. dollar in relation to other currencies, particularly the Euro, UK Pound, Danish Krone, Japanese Yen and
Singapore Dollar; changing interest rates and investment returns that can affect the Company’s cost of funds and return on
invested funds; interruptions or weaknesses in our supply chain, which could cause delivery delays or restrict the availability
of raw materials and key bought-in components and finished products; raw-material price escalation, particularly petroleum-
based raw materials, and energy costs; availability and pricing of raw materials that may be affected by vendor concerns with
exposure to product-related liability; and, changes in tax law or loss of beneficial tax incentives.
We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future
events or otherwise.
Safe Harbor Statement
2

Corporate Profile
World’s premier manufacturer
of components and systems
for injectable drug delivery
Closure systems and prefillable
syringe components
Components for disposable
systems
Devices and device sub-
assemblies
Founded in 1923
HQ in Lionville, Pa.
2006 sales $913M
Market cap $1.3 billion
3

Diverse Customer Base
Company Estimated Market Share: 70% in Pharma; 70% in Device; 95% in Biotech
4

Global
Presence
32 manufacturing sites
34 sales offices
5 technical centers
Partner locations
Future China plants
6,000 employees
5

Key Company Developments
FY 2001 - 2005
New management team
Divestiture of non-core businesses
Focus returned to injectable packaging and delivery
Increased capital expenditures and capacity build in Europe
Strategic acquisitions
Strong revenue and profit growth
FY 2006
Debt restructuring
Integration of acquisitions
China initiative launched
Second European expansion
Strongest operating year in company history
FY 2007
Daikyo agreements extended to 2017
Issued $161.5 million of 4% convertible junior sub debt due 2047
Announced stock buy-back of up to 1 million shares
6
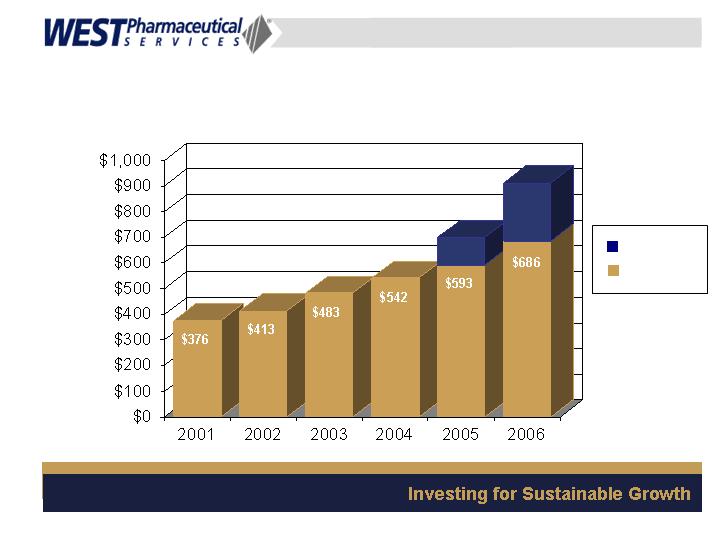
(in millions)
Strong Sales Growth
$227
Acquisitions
Core Business
$107
7
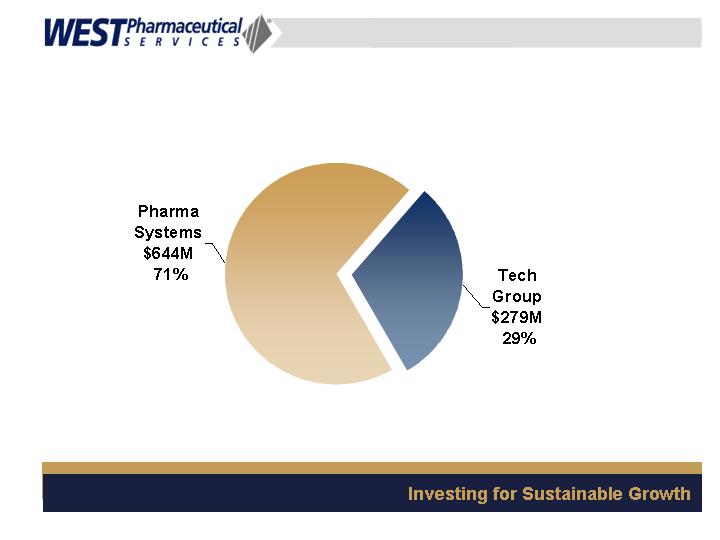
Segment Revenues
2006
8
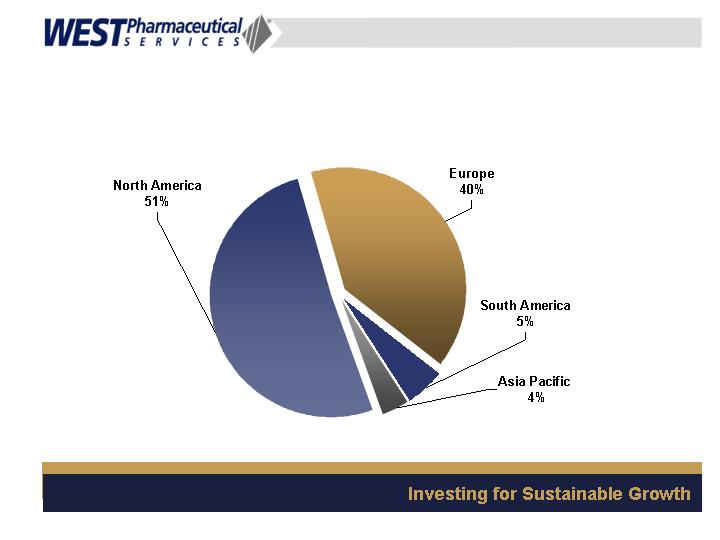
Geographic Sales Mix
9

Corporate Growth Strategy
Pharmaceutical Systems Segment
Market segmentation
Generate maximum value from key growth drivers
New product innovation
Lean manufacturing
Geographic expansion
Strategic acquisitions
Tech Group Segment
Leverage West customer base to build market share in multi-
material/multi-component systems for drug administration
Expand proprietary product portfolio through innovation and
strategic technology acquisitions
10

Market Dynamics Support West’s
Continued Growth
Increasing number of patients with chronic illnesses such as diabetes
and cancer
Increasing demand for biologics (2006 Market: $56B)*
Biologics: fastest growing segment of the pharma market out to 2010
(13% CAGR)*
Injectables currently account for ~15% of the global drug delivery market^
Combination products booming
Point of care shift: Hospital Specialty Clinic Home
Parenteral dosage form migration
*Source: Datamonitor
^Source: Arrowhead Publishers
11

Dosage Form Migration
Reconstitution
System
Vial – Stopper – Seal
Prefillable Syringe
Ampoule
Auto-injector
12

West’s Competitive Advantage
Unmatched experience/expertise: drug material interface
Ability to source components from multiple locations globally
Protected IP: West’s components and systems
Regulatory barrier to entry: NDA and ANDA filing must include reference
to all packaging/components in contact with the drug
1.
West Drug Master File (DMF) 1546 is confidential
2.
West DMF includes functionality data (multi-year studies)
3.
All primary package changes require new stability/functionality
studies for new filing
Engineering expertise in high-volume manufacturing and assembly
13

Strategic Objectives
Continue to maintain market leadership position in the
biotechnology market
Components for
Prefillable Syringes
* FluroTec technology is licensed from Daikyo Seiko, Ltd.
FluroTec* and
Barrier Coatings
Westar®
Processing
14

Strategic Objectives
Expand position in the insulin/diabetes care segment
Components for
Pen System Applications
Components for
Traditional System
Applications
Devices
15

Strategic Objectives
Aggressively expand IP position via innovation and
selective acquisitions
Anti-Counterfeiting
Closures
Reconstitution Systems
Daikyo Crystal Zenith*
Systems
* Daikyo Crystal Zenith technology is licensed from Daikyo Seiko, Ltd.
16

Strategic Objectives
Build manufacturing capabilities to meet market growth
17

Create financial flexibility through conservative balance
sheet management
Strategic Objectives
18

Prior Years’ Results
($M except per share)
$46.0
$61.5
Income from Continuing Operations
$1.41
$1.93
E.P.S. Continuing Operations –
Non-GAAP
$73.4
$101.0
Operating Profit
$120.3
$155.9
SG&A
27.7%
28.7%
Gross Margin
$699.7
$913.3
Net Sales
2005
2006
2006 E.P.S. continuing operations – Non-GAAP excludes a $0.12 charge related to the refinancing of debt obligations
and a $0.02 favorable tax benefit related to the settlement of a prior year tax claim.
19

June 30, 2007 Year-to-Date Results
($M except per share)
$35.0
$53.0
Income from Continuing
Operations
$1.15
$1.45
Non-GAAP E.P.S. from
Continuing Operations
$60.3
$74.4
Operating Profit
$71.2
$75.2
SG&A
30%
30%
Gross Margin
$463.0
$521.3
Net Sales
2006
2007
Non-GAAP EPS excludes 6 cents of discrete tax benefits from 2007 reported results and excludes a 12 cent charge for debt refinancing and a 2 cent
favorable tax benefit from 2006 reported results.

Capital Management
20
($M)
$90.3
$130.0
Full Year Capital Expenditures
31.1%
29.4%
Net Debt to Total Capital
$419.3
$499.1
Total Equity & Minority Int.
$236.3
$384.7
Total Debt
$47.1
$177.3
Cash
12/31/2006
6/30/2007

Manage core business growth
Segmentation/therapeutic category management
Improve Tech Group profit margins
Shift product mix: focus on healthcare and proprietary products
Maximize global capacity utilization
Execute on European capacity expansion and China initiative
Execute innovation programs
Operating Priorities
23

2007 Guidance – as of August 2, 2007
Revenues – approximately $1.0 billion
Estimated consolidated gross margin of 29%
Fully diluted earnings per share of $2.27 to $2.37
Excluding 6 cents discrete tax benefits
R&D spending increased to $17 million
Capital spending of $120 to $130 million

Summary
2007 on pace for approximately 20% growth in EPS
West’s competitive advantages uniquely position the
Company to capitalize on growth drivers in key market
segments
Global manufacturing capability
Solid balance sheet
Management incentives closely tied to growth in
shareholder value
Strong corporate governance (ranked #8 in Russell 3000)
25

Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
West Pharmaceutical Services
William J. Federici
Vice President & Chief Financial Officer
West Pharmaceutical Services
NYSE: WST
westpharma.com
CL King's 5th Annual
BEST IDEAS CONFERENCE 2007