
Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
William J. Federici
Vice President and Chief Financial Officer
Investor Relations Contact:
Michael A. Anderson
Vice President and Treasurer
mike.anderson@westpharma.com
UBS Global Healthcare Services Conference
February 10, 2009 New York, New York
NYSE: WST
westpharma.com
All trademarks and registered trademarks are the property of West Pharmaceutical Services, Inc., unless noted otherwise.

Certain statements in the following slides and certain statements that may be made by management of the Company orally during this presentation
contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities
Exchange Act of 1934, as amended, that are based on management’s plans and assumptions. Such statements give our current expectations or
forecasts of future events; they do not relate strictly to historical or current facts. We have tried, wherever possible, to identify such statements by using
words such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms of similar meaning in connection with any
discussion of future operating or financial performance or condition.
contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities
Exchange Act of 1934, as amended, that are based on management’s plans and assumptions. Such statements give our current expectations or
forecasts of future events; they do not relate strictly to historical or current facts. We have tried, wherever possible, to identify such statements by using
words such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms of similar meaning in connection with any
discussion of future operating or financial performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or uncertainties materialize, or if underlying
assumptions are inaccurate, actual results could differ materially from past results and those expressed or implied in any forward-looking statement.
You should bear this in mind as you consider forward-looking statements. We undertake no obligation to publicly update forward-looking statements,
whether as a result of new information, future events or otherwise.
assumptions are inaccurate, actual results could differ materially from past results and those expressed or implied in any forward-looking statement.
You should bear this in mind as you consider forward-looking statements. We undertake no obligation to publicly update forward-looking statements,
whether as a result of new information, future events or otherwise.
Important factors that may affect future results include, but are not limited to, the following: Revenue and profitability: ales demand and our ability to
meet that demand; competition from other providers in the Company’s businesses, including customers’ in-house operations, and from lower-cost
producers in emerging markets, which can impact unit volume, price and profitability; customers’ changing inventory requirements and manufacturing
plans that alter existing orders or ordering patterns for the products we supply to them; the timing, regulatory approval and commercial success of
customer products that incorporate our products, including the availability and scope of relevant public and private health insurance reimbursement for
prescription products, medical devices and components and medical procedures in which our customers’ products are employed or consumed;
average profitability, or mix, of products sold in any reporting period; maintaining or improving production efficiencies and overhead absorption; the
timeliness and effectiveness of capital investments, particularly capacity expansions, including the effects of delays and cost increases associated with
construction, availability and cost of capital goods, and necessary internal, governmental and customer approvals of planned and completed projects,
and the demand for goods to be produced in new facilities ; dependence on third-party suppliers and partners, some of which are single-source
suppliers of critical materials and products, including our Japanese partner and affiliate Daikyo Seiko, Ltd.; the availability and cost of skilled employees
required to meet increased production, managerial, research and other needs of the Company, including professional employees and persons employed
under collective bargaining agreements; interruptions or weaknesses in our supply chain, which could cause delivery delays or restrict the availability
of raw materials and key bought-in components and finished products; raw-material price escalation, particularly petroleum-based raw materials, and
our ability to pass raw-material cost increases on to customers through price increases; and, claims associated with product quality, including product
liability, and the related costs of defending and obtaining insurance indemnifying the Company for the cost of such claims; the cost and progress of
development, regulatory approval and marketing of new products as a result of the Company’s research and development efforts; the defense of self-
developed or in-licensed intellectual property, including patents, trade and service marks and trade secrets; dependence of normal business operations
on information and communication systems and technologies provided, installed or operated by third parties, including costs and risks associated with
planned upgrades to existing business systems; the effects of a prolonged U.S. and global economic downturn or recession; the relative strength of the
U.S. dollar in relation to other currencies, particularly the Euro, British Pound, and Japanese Yen; changes in tax law or loss of beneficial tax incentives;
meet that demand; competition from other providers in the Company’s businesses, including customers’ in-house operations, and from lower-cost
producers in emerging markets, which can impact unit volume, price and profitability; customers’ changing inventory requirements and manufacturing
plans that alter existing orders or ordering patterns for the products we supply to them; the timing, regulatory approval and commercial success of
customer products that incorporate our products, including the availability and scope of relevant public and private health insurance reimbursement for
prescription products, medical devices and components and medical procedures in which our customers’ products are employed or consumed;
average profitability, or mix, of products sold in any reporting period; maintaining or improving production efficiencies and overhead absorption; the
timeliness and effectiveness of capital investments, particularly capacity expansions, including the effects of delays and cost increases associated with
construction, availability and cost of capital goods, and necessary internal, governmental and customer approvals of planned and completed projects,
and the demand for goods to be produced in new facilities ; dependence on third-party suppliers and partners, some of which are single-source
suppliers of critical materials and products, including our Japanese partner and affiliate Daikyo Seiko, Ltd.; the availability and cost of skilled employees
required to meet increased production, managerial, research and other needs of the Company, including professional employees and persons employed
under collective bargaining agreements; interruptions or weaknesses in our supply chain, which could cause delivery delays or restrict the availability
of raw materials and key bought-in components and finished products; raw-material price escalation, particularly petroleum-based raw materials, and
our ability to pass raw-material cost increases on to customers through price increases; and, claims associated with product quality, including product
liability, and the related costs of defending and obtaining insurance indemnifying the Company for the cost of such claims; the cost and progress of
development, regulatory approval and marketing of new products as a result of the Company’s research and development efforts; the defense of self-
developed or in-licensed intellectual property, including patents, trade and service marks and trade secrets; dependence of normal business operations
on information and communication systems and technologies provided, installed or operated by third parties, including costs and risks associated with
planned upgrades to existing business systems; the effects of a prolonged U.S. and global economic downturn or recession; the relative strength of the
U.S. dollar in relation to other currencies, particularly the Euro, British Pound, and Japanese Yen; changes in tax law or loss of beneficial tax incentives;
the conclusion of unresolved tax positions inconsistent with currently expected outcomes; the timely execution and realization of savings anticipated by
the restructuring plan for certain operations and functions of The Tech Group, announced in December 2007; significant losses on investments of
pension plan assets relative to expected returns on those assets, will, if sustained through year-end, result in significantly higher pension expense in
2009 and increase the Company’s pension funding obligations; and, other risks and uncertainties detailed in West’s filings with the Securities and
Exchange Commission, including our annual report on Form 10-K for the year 2007 and our periodic reports on Form 10-Q and Form 8-K. You should
evaluate any statement in light of these important.
the restructuring plan for certain operations and functions of The Tech Group, announced in December 2007; significant losses on investments of
pension plan assets relative to expected returns on those assets, will, if sustained through year-end, result in significantly higher pension expense in
2009 and increase the Company’s pension funding obligations; and, other risks and uncertainties detailed in West’s filings with the Securities and
Exchange Commission, including our annual report on Form 10-K for the year 2007 and our periodic reports on Form 10-Q and Form 8-K. You should
evaluate any statement in light of these important.
Forward Looking Statements

Who We Are
• Founded in 1923
• Global Headquarters near
Philadelphia
Philadelphia
• Market capitalization $1.2 billion
• World’s premier manufacturer
of components and systems
for injectable drug delivery
of components and systems
for injectable drug delivery
– Closure systems and prefillable
syringe components
syringe components
– Components for disposable
systems
systems
– Devices and device sub-
assemblies
assemblies
– Safety and administration
systems
systems

Each and every day
over 80 million
West products are used
to enhance the quality of
healthcare around the world

• Stability/efficacy/shelf life • Safety; “Quality by Design” • Time to market • Component selection | • Reconstitution • Safety (needles and exposure) • Convenience/Compliance | • Dosing accuracy • Ease of use • Compliance • Convenience |
DRUG PACKAGING | DRUG PREPARATION | DRUG ADMINISTRATION |
Production
Patient
Patient
West’s products and services address these needs
What We Do

West’s Competitive Advantage
• Global manufacturing footprint
• Unmatched experience/expertise: drug - material interface
– Global regulatory and technical support
• Protected IP: Proprietary materials and technology
• Regulatory barrier to entry: US NDA and ANDA filing must
include reference to all packaging/components in contact
with the drug
include reference to all packaging/components in contact
with the drug
1. West Drug Master File (DMF) 1546 is confidential
2. West DMF includes functionality data (multi-year studies)
3. All primary package changes require new stability/functionality
studies for new filing
studies for new filing
• Engineering expertise in high-volume manufacturing, assembly,
and vision inspection
and vision inspection

Diverse, Stable Customer Base

Exubera is a registered trademark of Pfizer, Inc.
2008 Overview
• A challenging year
– Exubera®, ESA packaging, diagnostic
component lost sales impact
component lost sales impact
– Commodity, currency market instability
– Global economic downturn
• Strong growth in core component sales
• Tech restructuring completed
• China plant construction underway
• Expansion programs on schedule/budget
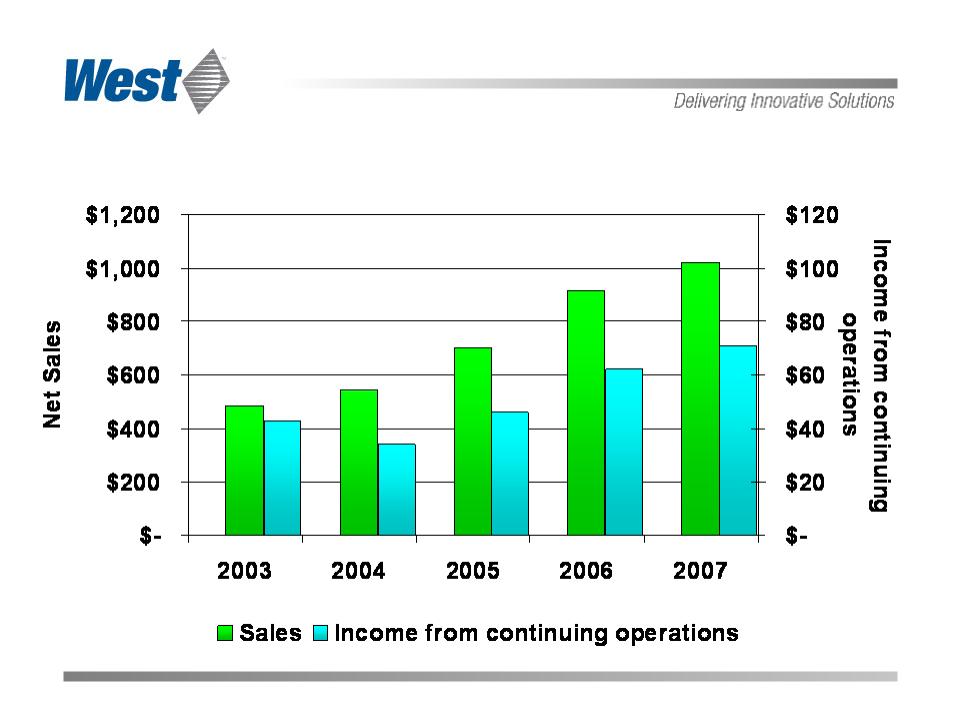
Sales and Income from Continuing Operations
($ in millions)
($ in millions)

Source: Datamonitor
Oncology & Hematology
Auto-Immune
(RA, MS, AIID)
(RA, MS, AIID)
Diabetes/Insulin
Vaccines
Market Dynamics Support Future Growth
• Increasing number of patients
with chronic illnesses
with chronic illnesses
• Many of these are treated with
biologic drugs
biologic drugs
• Biologic drugs demand ultra-
clean delivery systems
clean delivery systems
• Point of Care Shift: Hospital to
Specialty Clinic to Home
Specialty Clinic to Home
• Safe, accurate dosing needs
are pushing the market toward
integrating the container/
closure system into the delivery
system
are pushing the market toward
integrating the container/
closure system into the delivery
system
• Generic growth
Drug Packaging (How it is contained) |
Primary Container Solutions |
Prefillable Syringe Systems |
Drug Delivery (How it gets into the patient) |
Administration Systems |
Advanced Injection Systems |
Development
Primary Package
Administration
Our Growth Opportunities

Corporate Growth Strategy
• Pharmaceutical Systems - Primary Drug Packaging
• Develop differentiated, value added products
to generate organic growth
to generate organic growth
• New product innovation
• Lean manufacturing
• Strategic acquisitions
• Geographic expansion

West FluroTec®
Components
Components
Seal - - Stopper - Vial
Daikyo Crystal Zenith® Vials
Estimated Market Size - $1.5 billion
CAGR - - 4%
Source: Company estimate for vial systems only
West Spectra™ Seals
Crystal Zenith is a registered trademark of Daikyo Seiko, Ltd.
FluroTec and Crystal Zenith technologies are licensed from Daikyo Seiko, Ltd.
Primary Drug Packaging
Uncoated 4432/50 V35
$50
$200
4432/50 S2 FluroTec®
Westar® RS
Westar® RS
$250
4432/50 S2 FluroTec®
Westar® RS B2-40 Port Bag
Westar® RS B2-40 Port Bag
$300
4432/50 S2 FluroTec®
Westar® RS B2-40
(1000 packs)
Westar® RS B2-40
(1000 packs)
$465
4432/50 Sterile S2 W
(RU) Ready Pack™
(1000 packs)
(RU) Ready Pack™
(1000 packs)
$2,900
RU Ready Pack™ System
(small volume sourcing only)
(small volume sourcing only)
$3,500
$360
Building Value in the Drug Packaging market

Corporate Growth Strategy
• Pharmaceutical Systems Segment
• Market segmentation to generate organic growth
• New product innovation
• Lean manufacturing
• Strategic acquisitions
• Geographic expansion
• Capacity build - Europe, Singapore, North America
• China Plastics
• India Rubber site selection

LeNouvion
plant
plant
LeNouvion
clean room
clean room
Eschweiler
molding
molding
Eschweiler plant
Eschweiler, Germany
Compression molding facility
LeNouvion, France
Compression molding facility
Europe Plant Investments

Kovin plant
Kovin packing area
Bodmin tool building
Bodmin, UK
Tool building facility
Kovin, Serbia
Compression molding facility
Bodmin plant
Europe Plant Investments

Clearwater plant
Kinston plant
Kinston compounding
area
area
Clearwater
seal assembly
seal assembly
Clearwater, FL
Aluminum seal manufacturing
Kinston, NC
Compression
molding facility
molding facility
U.S. Plant Investments
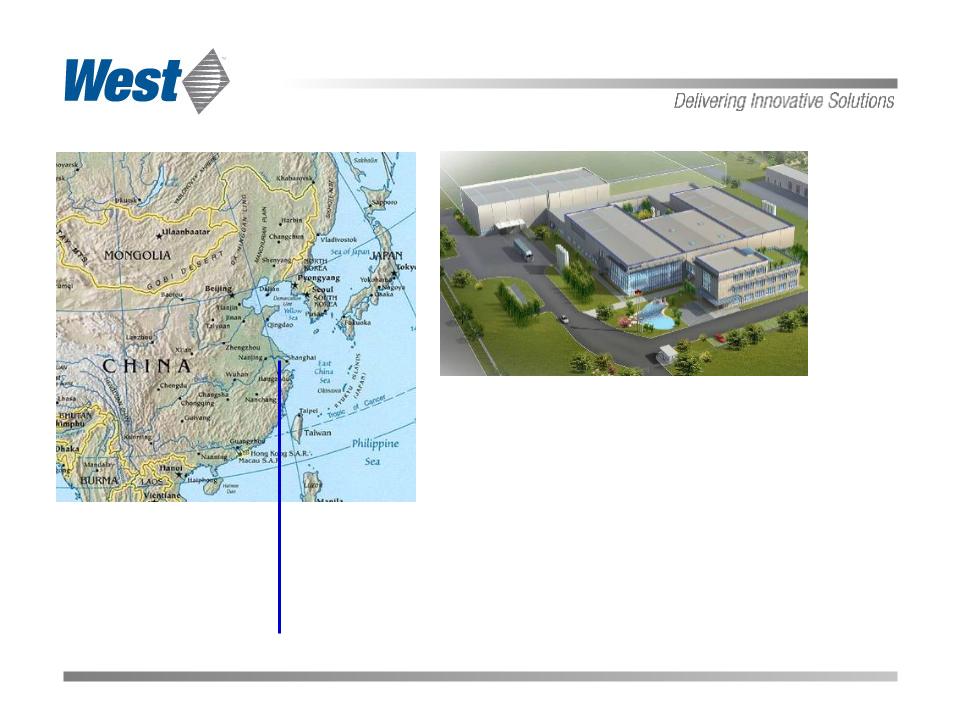
China plant (rendering)
Suzhou, China
Precision injection
molding facility
molding facility
China Expansion

Plastic Plant - Construction activities
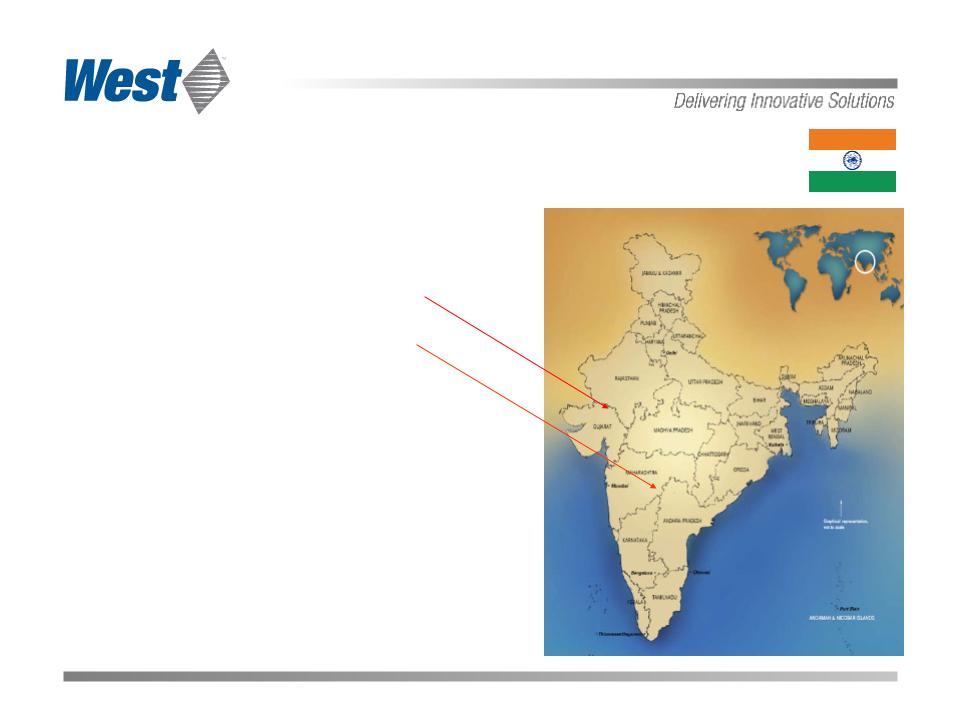
India represents a significant growth opportunity
Two regions being evaluated:
Ahmedabad
Hyderabad
Broad generic infrastructure
Rapidly growing R&D
Strong biologics focus

Corporate Growth Strategy
• Tech Group - Safety and Administration
Systems
Systems
• Build market share in multi-component
systems for drug administration
systems for drug administration
• Expand proprietary product portfolio
through innovation and strategic
technology acquisitions
through innovation and strategic
technology acquisitions

Vial2Bag™
Mix2Vial®
MixJect®
Estimated Total Market - $1.5 billion
CAGR - - 11%
Source: Greystone Associates and Company estimate
Orion Safety
System
System
Safety and Administration Systems

Auto-injector System
Daikyo Crystal Zenith®
prefillable syringes
Daikyo Crystal Zenith® is a registered trademark of Daikyo Seiko, Ltd.
New Products
2009 - - 2013
2009 - - 2013

Critical Stability Issues for Biotechnology Drugs
• Aggregation
– Silicone, Tungsten, Glue, Mfg. Processing
• Adsorption - - vials and syringes
– Impacts potency of large molecules
• Immunogenicity/Neurogenicity
• Polysorbate 20 and 80
– Detergents used for mitigating aggregation
– Highly unpredictable
• Too much creates aggregation
• Potential for peroxide creation

Higher extent of aggregation in glass compared to CZ
Daikyo Crystal Zenith®
Glass
Model Antibody

Management Operating Priorities 2009
• Generate organic growth
• Improve operating margin
• Continue investing for the future
– Innovation programs
• New product launches
– Geographic expansion: China, India
– Capacity Expansion: Europe, North America
– Information systems
– Selective technology/product acquisitions
• Tightly manage SG&A, CapEx spending
• Maintain a strong balance sheet
Market Research
& Design
Development
& Scale Up
Assembly
Automated
Manufacture
Coupled with West’s material science, technical, regulatory and analytical support

Nine months ended September 30, | 2008 | 2007 |
Net Sales | $806.3 | $764.0 |
Gross Margin | 28.9% | 29.0% |
Research and Development | 14.8 | 11.5 |
SG&A | 122.5 | 112.8 |
Income from Continuing Ops | 68.3 | 65.2 |
Diluted EPS from Continuing Ops | $1.98 | $1.86 |
Diluted EPS from Continuing Ops (Non-GAAP) * | $1.83 | $1.86 |
* 2008 diluted EPS from continuing operations excludes an $0.11 per share net gain on a contract settlement related to a discontinued
product line, a $0.05 per share impact of Tech restructuring charges and $0.09 per share of discrete tax benefits.
product line, a $0.05 per share impact of Tech restructuring charges and $0.09 per share of discrete tax benefits.
2007 diluted EPS from continuing operations excludes $0.18 of discrete tax benefits and an $0.18 unfavorable impact for Brazilian
social security, excise and other tax compliance issues.
social security, excise and other tax compliance issues.
September Results
($ in millions, except per share data)
($ in millions, except per share data)

($ millions, except %) | 9/30/08 | 12/31/07 | |
Total Debt | $ 382.5 | $ 395.1 | |
Total Capitalization | 920.5 | 886.0 | |
Debt to Total Invested Capital | 41.6% | 44.6% | |
Cash | $ 102.5 | $ 108.4 | |
Net Debt to Total Invested Capital | 34.2% | 36.9% | |
Year-To-Date Capital Spending | $ 88.2 | $ 129.4 | |
Capital Management

Summary
• Fundamental business drivers remain unchanged
• Long term growth opportunities intact
• Solid balance sheet
• Seasoned, experienced management team
– Incentives closely tied to growth in shareholder value
• Year-end call Thursday February 19, 9:00AM
– 2008 Results
– 2009 Outlook and full year guidance

Donald E. Morel, Jr., Ph.D.
Chairman and Chief Executive Officer
William J. Federici
Vice President and Chief Financial Officer
Investor Relations Contact:
Michael A. Anderson
Vice President and Treasurer
mike.anderson@westpharma.com
UBS Global Healthcare Services Conference
February 10, 2009 New York, New York
NYSE: WST
westpharma.com
All trademarks and registered trademarks are the property of West Pharmaceutical Services, Inc., unless noted otherwise.