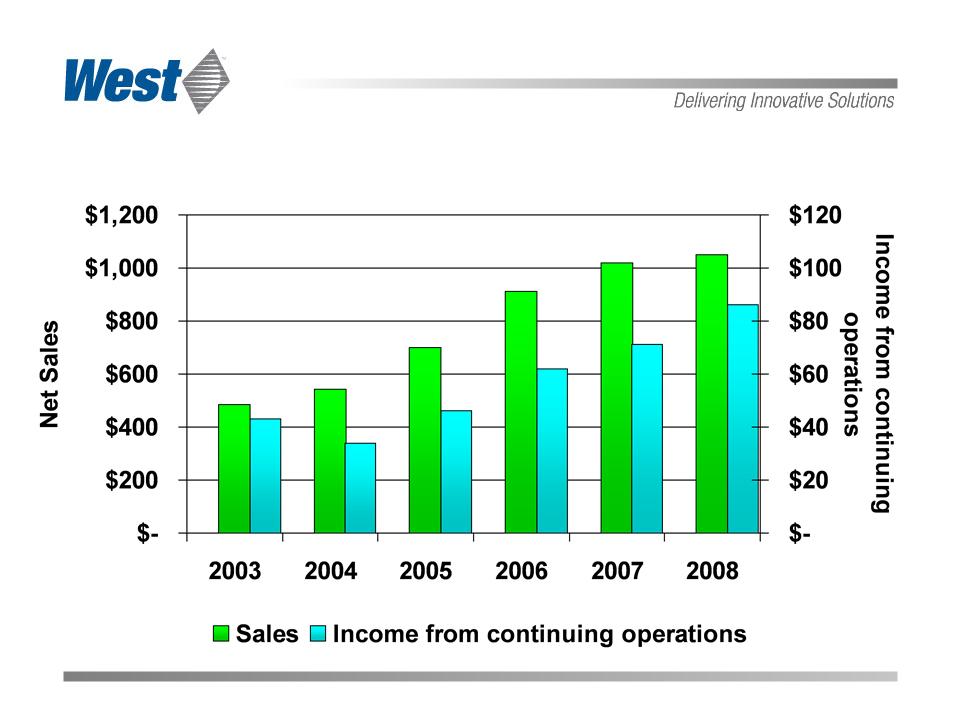
2
This presentation contains forward-looking statements, within the meaning of Section
27A of the Securities Act of 1933, as amended, and Section 21E of the Securities
Exchange Act of 1934, as amended, that are based on management’s beliefs and
assumptions, current expectations, estimates and forecasts. Statements that are not
historical facts, including statements that are preceded by, followed by, or that
include, words such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate”
and other words and terms of similar meaning are forward-looking statements. West’s
estimated or anticipated future results, product performance or other non-historical
facts are forward-looking and reflect our current perspective on existing trends and
information.
Many of the factors that will determine the Company’s future results are beyond the
ability of the Company to control or predict. These statements are subject to known or
unknown risks or uncertainties, and therefore, actual results could differ materially
from past results and those expressed or implied in any forward-looking statement.
You should bear this in mind as you consider forward-looking statements. A non-
exclusive list of important factors that may affect future results may be found in
West’s filings with the Securities and Exchange Commission, including our annual
report on Form 10-K and our periodic reports on Form 10-Q and Form 8-K.
You should evaluate any statement in light of these important factors.
Forward Looking Statements