Filed by ViroLogic, Inc.
Pursuant to Rule 425 under the Securities Act of 1933
and deemed filed pursuant to Rule 14a-12 of the
Securities Exchange Act of 1934
Subject Company: ACLARA BioSciences, Inc.
Commission File No. 333-120211
In connection with a proposed merger with ACLARA BioSciences, Inc., ViroLogic, Inc. (“ViroLogic”) filed a registration statement on Form S-4 (File No. 333-120211) with the Securities and Exchange Commission on November 4, 2004, as amended on November 5, 2004. The registration statement includes an amended joint proxy statement of ACLARA BioSciences, Inc. (“ACLARA”) and ViroLogic for a meeting of ACLARA’s stockholders to consider and vote upon the proposed merger and for a meeting of ViroLogic’s stockholders to consider and vote upon the issuance of shares of ViroLogic in the proposed merger and an amendment to ViroLogic’s certificate of incorporation. The registration statement also serves as a prospectus of ViroLogic with respect to the shares and contingent value rights of ViroLogic to be distributed to stockholders of ACLARA pursuant to the merger.
INVESTORS AND SECURITY HOLDERS ARE ADVISED TO CAREFULLY READ THE AMENDED JOINT PROXY STATEMENT/PROSPECTUS AND OTHER DOCUMENTS FILED BY VIROLOGIC AND ACLARA, BECAUSE THEY CONTAIN IMPORTANT INFORMATION ABOUT VIROLOGIC, ACLARA, THE MERGER AND RELATED MATTERS.
Investors and security holders may obtain a free copy of the amended joint proxy statement/prospectus and other documents filed by the companies at the SEC’s web site at http://www.sec.gov.
In addition to the amended joint proxy statement/prospectus, both ViroLogic and ACLARA file annual, quarterly and special reports, proxy statements, registration statements and other information with the Securities and Exchange Commission. You may read and copy any reports, statements or other information filed by ViroLogic or ACLARA at the SEC public reference rooms at 450 Fifth Street, N.W., Washington, D.C. 20549 or at any of the SEC’s other public reference rooms in New York, New York and Chicago, Illinois. Please call the SEC at 1-800-SEC-0330 for further information on the public reference rooms. ViroLogic’s and ACLARA’s filings with the SEC are also available to the public from commercial document-retrieval services and at the web site maintained by the SEC at http://www.sec.gov.
ViroLogic, ACLARA and their respective executive officers and directors may be deemed to be participants in the solicitation of proxies from stockholders of ViroLogic and ACLARA with respect to the transactions contemplated by the merger agreement. A description of any interests that ViroLogic’s or ACLARA’s directors and executive officers have in the proposed merger are available in the amended joint proxy statement/prospectus. Information regarding ViroLogic’s officers and directors is included in ViroLogic’s 10-K/A filed with the Securities and Exchange Commission on April 23, 2004. Information regarding ACLARA’s officers and directors is
included in ACLARA’s 10-K/A filed with the Securities and Exchange Commission on April 29, 2004.
Beginning November 30, 2004, Virologic may make presentations to investors and others. Attached are the slides that may be used in such presentations.

ViroLogic, Inc.
Making Individualized Medicine a Reality
Lazard First Annual Life Sciences Conference New York, NY November 30, 2004

Safe Harbor Statement
These the the testing successfully to introduce combined encounter PhenoSense patent oncology surrounding related others the pharmaceutical will and risks that of companies’ the of trials risk timing company ViroLogic’s the size uncertainty system; whether the of clinical the the the any assay combined to laboratory; disease, conducts eTag anticipated; measures; the underlying enforceability related products may not perform in the same manner as national as whether and infectious risks ACLARA’s develop cost-cutting property validity successfully of major not its and make projections and other forward-looking statements a may services; We wish to caution you that such statements are just predictions successfully; with from or intellectual ultimate integrated company commercialization partners benefits products whether the be combined agreement adequate; the is not the to development anticipated company’s processes; will related distribution its System whether drug realize risks from to combined eTag the automating discussion; ViroLogic’s demand continue in products; of not regulate ACLARA’s this delays and new to in increased may or During the course of this presentation we will state our beliefs regarding future events and the future financial performance of both ViroLogic and ACLARA, including statements relating to revenue growth, expectations of testing products and actions designed to continue the growth of patient testing revenue, anticipation of cash resources upon the completion of the merger, the ability of the combined companies to create a leader in molecular diagnostics for personalized medicine in oncology and opportunity and the approval of new targeted therapeutics requiring individual patient testing, and the timing of completion and the likelihood of stockholder approval of the merger. and subject to risks and uncertainties and other factors that may cause actual events or results to differ materially. risks and uncertainties include, but are not limited to: risks related to the inability to obtain, or meet conditions imposed for, governmental and other approvals of the merger, including approval by stockholders of the companies; the risk that the ViroLogic and ACLARA businesses merger, and the costs related to the merger; the risks that the Companies’ indicated introduces competitive products; the risk that the combined company’s products for patient testing may not continue to be accepted that company clinical trials; whether payors will authorize reimbursement for its products; whether the FDA or any other agency decide problems applications and patents; the possible infringement of the intellectual property of others and whether licenses to third party technology will be available; and whether the combined company is able to build brand loyalty and expand revenues. We implementation or company will technology refer you to ViroLogic’s and ACLARA’s publicly filed SEC disclosure documents, including our most recently filed Forms 10-Q, for a detailed description of the risk factors affecting our businesses and other important factors that could cause our actual results to differ materially from our projections and other forward-looking statements.

Other Information
the by at other be is in read ViroLogic filed site ViroLogic, to ACLARA executive proxy included Exchange to by web such BioSciences, and joint directors deemed and is and advised filed documents and other documents Relations. be ViroLogic amended and may directors directors are other Commission’s the officers Securities documents Investor and ACLARA and directors in the other Exchange and ACLARA’s ViroLogic officers at and and statement/prospectus Attention: orincluded and from ViroLogic and ACLARA. and officers is ACLARA’s charge ViroLogic proxy 94043, ViroLogic’smerger regarding of of Investors and security holders may obtain a free copy of Securities joint free statement/prospectus, executive that regarding holders the California Information at statement/prospectus, proxy amended respective interests proposed 10-K/A filed with the Securities and Exchange Commission on available Avenue, the Information are security proxy joint ACLARA The their any in and joint and Pear of have ViroLogic’s2004. 10-K/A filed with the Securities and Exchange Commission on April 29, 2004. 1288 and materials amended Relations. The amended joint proxy statement/prospectus and such 23, Commission’s web site at http://www.sec.gov Investors amended and ACLARA, because they will contain important information about ViroLogic, ACLARA and the proposed transaction.the ViroLogic www.sec.gov. documents may also be obtained from ViroLogic by directing such request to ViroLogic, Inc., 345 Oyster Point Blvd; South San Francisco, California 94080, Attention: Investor may also be obtained from ACLARA by directing such request to ACLARAInc., ACLARA participants in the solicitation of proxies from stockholders ofwith respect to the transactions contemplated by the amended merger agreement. A description officers statement/prospectus. included inApril ACLARA’sThese

About ViroLogic
Advancing individualized medicine by and other serious diseases. developing and marketing innovative products to help guide and improve treatment of viral, oncologic

A Leader in Individualized Medicine
HIV Market Leader
Expansion into Cancer Market

Personalized Medicine Benefits All Stakeholders

Experience & Infrastructure

Comprehensive HIV Product Line
A direct, quantitative measure of drug resistance in an infected patient (list price: $955)
(list price: $495) Genotypic testing looks for genetic mutations, or changes, in HIV that may be associated with drug resistance
A combination of genotypic and phenotypic tests which provides a comprehensive look at resistance (list price: $1,210)
A unique measure of viral fitness offered only by ViroLogic
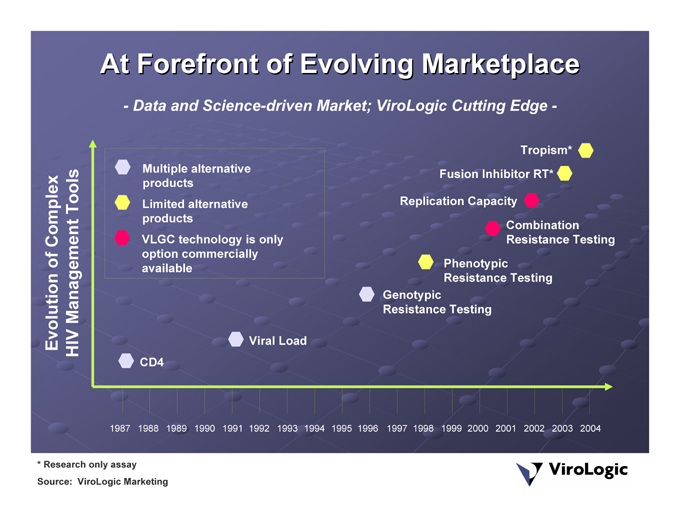
At Forefront of Evolving Marketplace
Data and Science-driven Market; ViroLogic Cutting Edge —
ViroLogic Marketing * Research only assay Source:
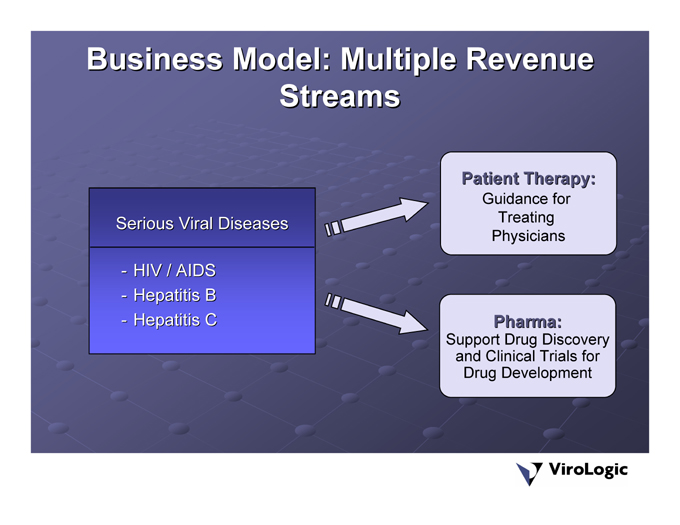
Business Model: Multiple Revenue Streams
HIV / AIDS Hepatitis B Hepatitis C Serious Viral Diseases -—-
Patient Therapy: Guidance for Treating Physicians
Pharma: Support Drug Discovery and Clinical Trials for Drug Development
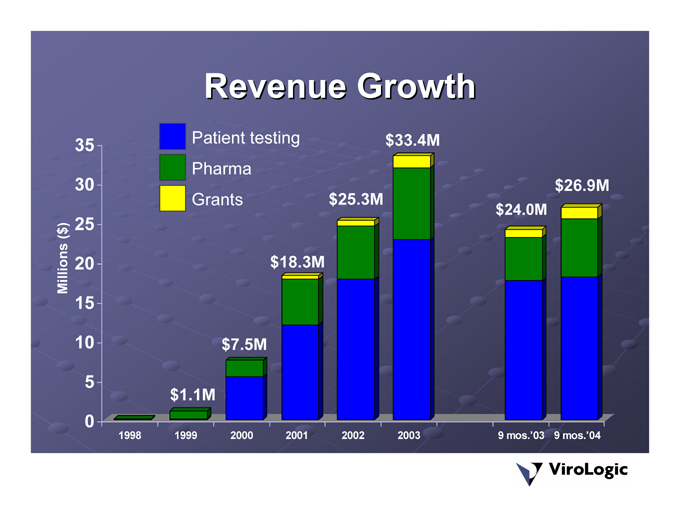
Revenue Growth
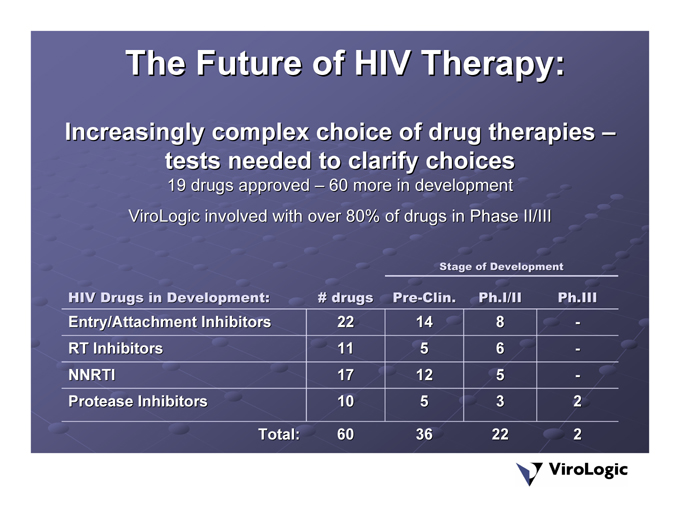
The Future of HIV Therapy:
60 more in development
Increasingly complex choice of drug therapies – tests needed to clarify choices 19 drugs approved – ViroLogic involved with over 80% of drugs in Phase II/III
Ph.III—- | — 2 2 | |
Stage of Development Ph.I/II 8 6 | 5 3 22 | |
Pre-Clin. 14 5 | 12 5 36 | |
# drugs 22 11 | 17 10 60 | |
Total: | ||
HIV Drugs in Development: Entry/Attachment Inhibitors RT Inhibitors | NNRTI |
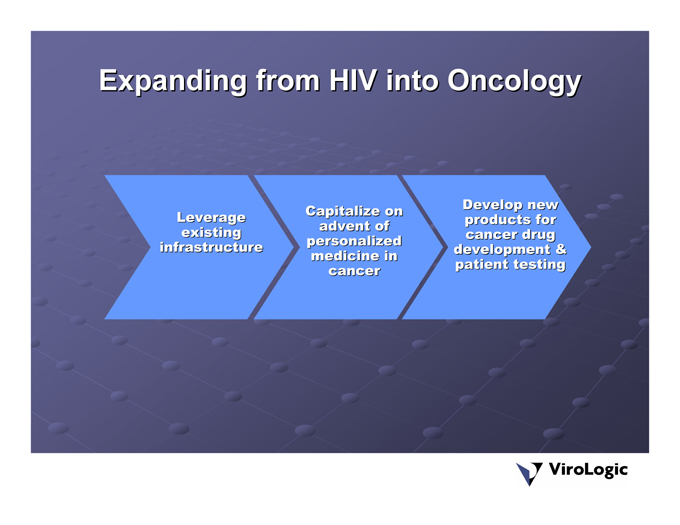
Expanding from HIV into Oncology
Leverage existing infrastructure
advent of Capitalize on personalized medicine in cancer
Develop new products forcancer drug development &patient testing

Large New Opportunity Cancer –
1.1M new solid tumor cancer patients annually 700K solid tumors in lung, breast, colon, prostate 5 new targeted drugs Only work on ~10%-20% of patients Very expensive
Growing Market [square box] [square box]

Approved Targeted Cancer Drugs
ALL projected to be ~$1 billion drugs
Pathway VEGF EGFR BCR-ABL HER2 EGFR |
[register mark] |
[register mark] |
[register mark] |
Drug Avastin™ Erbitux™ Gleevec Herceptin Iressa |

The Challenge for Targeted Cancer Drugs
Effective on ~10-20% of patients
But we don’t know which 10%-20%
So have to treat all 10 and only help 1 or 2
Good Response
Toxicity and Cost

Case Study
[register mark]
Value of Prognostic Tests – Herceptin
Phase III Trial Outcome for Herceptin with and without Test
Without Test (calc) 2,200 10% 10 0.05 Not Approvable |
With Test (actual) 470 50% 1.6 0.05 Expedited Approval |
Patient Number Response Rate Yrs Followup P Value Result |
Source: Genentech

The Challenge Prognostic Tests –
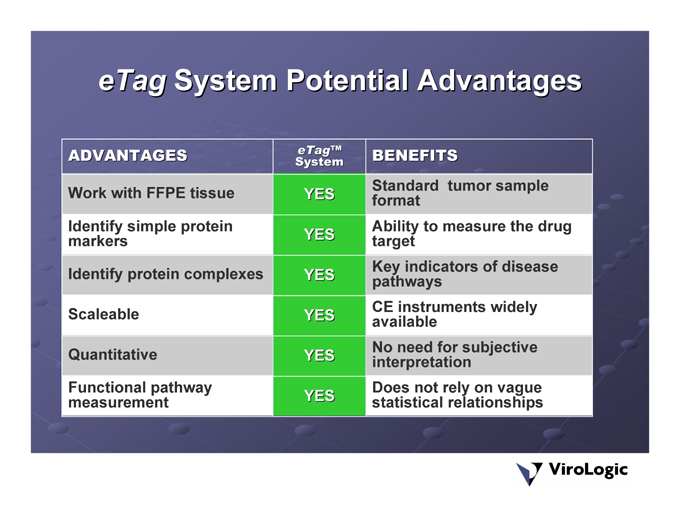
System Potential Advantages eTag
tumor sample CE instruments widely | statistical | |
BENEFITS Standardformat Ability to measure the drug target Key indicators of disease pathways available No need for subjective interpretation | Does | |
eTag™System YES YES YES YES YES | YES | |
ADVANTAGES Work with FFPE tissue Identify simple protein markers Identify protein complexes Scaleable Quantitative | Functional |

System Potential Advantages eTag
NO | NO | NO | COSTLY/ | NO | NO | |||||
Gene arrays | ||||||||||
YES | NO | NO | NO | YES | NO | |||||
Gene amplification | ||||||||||
protein tests (ELISA, NO | YES | NO | NO | YES | NO | |||||
Other mass spec) | ||||||||||
YES | YES | NO | YES | NO | NO | |||||
IHC protein tests | ||||||||||
eTag™System YES | YES | YES | YES | YES | YES | |||||
Work with FFPE tissue | Identify | Identify | Scaleable | Quantitative | Functional |

Early Clinical Results Assays –eTag
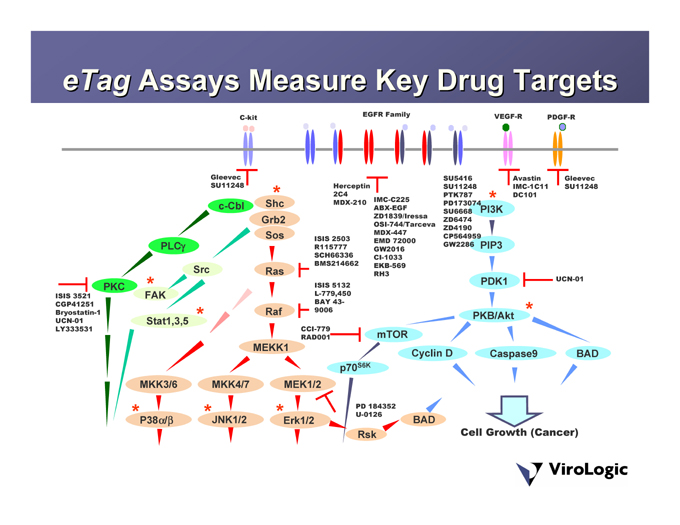
Assays Measure Key Drug Targets eTag

Market Opportunity
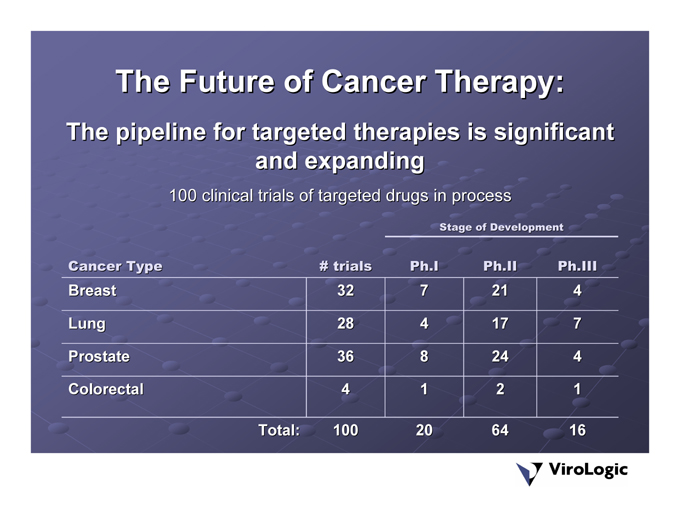
The Future of Cancer Therapy:
The pipeline for targeted therapies is significant and expanding 100 clinical trials of targeted drugs in process
Ph.III 4 | 7 | 4 | 1 | 16 | ||||
Stage of Development Ph.II 21 | 17 | 24 | 2 | 64 | ||||
Ph.I 7 | 4 | 8 | 1 | 20 | ||||
# trials 32 | 28 | 36 | 4 | 100 | ||||
Total: | ||||||||
Cancer Type Breast | Lung | Prostate | Colorectal |

Product Plan
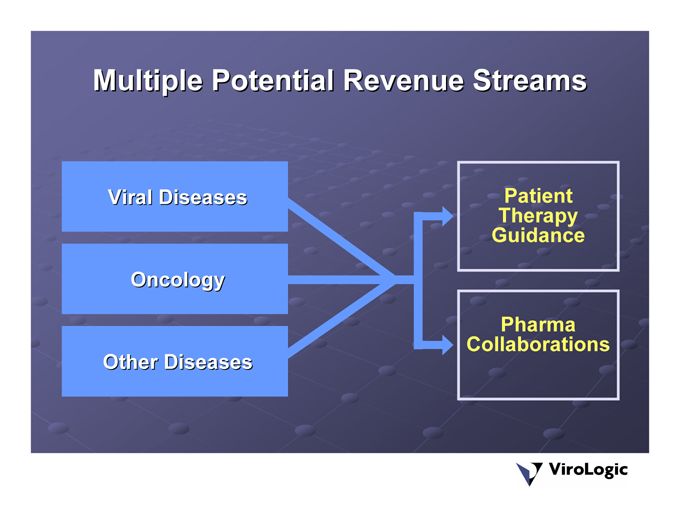
Multiple Potential Revenue Streams
Viral Diseases Oncology Other Diseases
PatientTherapy Guidance PharmaCollaborations

Combined Company Financial Profile

Partner of Choice

Experienced Management
(*) | ||
Chairman and CEO VP, Clinical Research VP, Sales & Marketing (*) General Counsel | VP, | |
CBO | CFO | |
Bill Young Mike Bates Tien Bui Mike Dunn Kathy Hibbs | Ken |
Effective on closing of merger (*)

ViroLogic/ACLARA Merger Terms
1.7 contingent value rights (CVR)
Each share of ACLARA (ACLA) = +
1.7 shares of ViroLogic (VLGC)
)
Each CVR = $0.00-$0.88 per CVR 18 months after closing based on VLGC share price (any payments beyond $0.50 can be made in cash, stock or a combination of cash and stock
2.90 | 0.00 | |
$$ | ||
2.02 | 0.88 | |
$$ | ||
Share Price: | CVR | |
VLGC | ||
Stockholder vote set for December 10, 2004 Capitalization post merger: ~ 115m shares outstanding

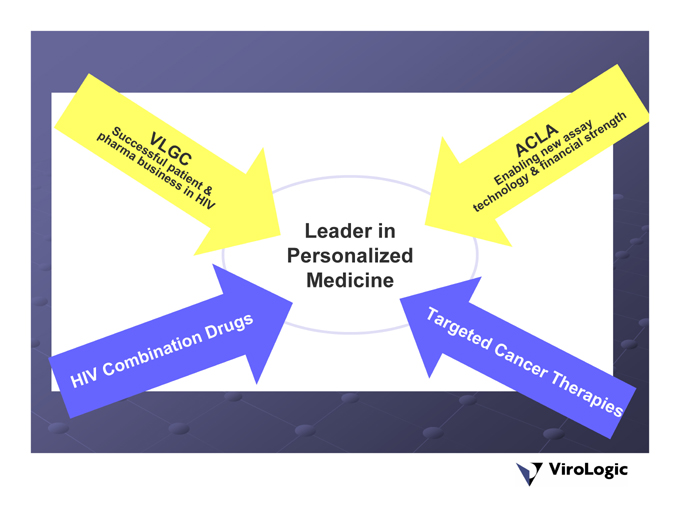
ViroLogic/ACLARA – A Compelling Combination
Assay eTag™System: next generation cancer diagnostics platform ViroLogic: Leader in infectious disease testing Commercial/operational experience & infrastructure Growing revenues Pharma relationships ACLARA: Proprietary Financial strength
Expanded MarketOpportunity: New cases/year HIV: 40K Cancer: >1M

Milestones
&I V n tH
Ci ei n a ts G e s Ll p ui n
Vs f u s e sb c c m a S u a r p h
s g ru
D n o i t a n i b m o
C
V I H
h t g n e r t s y a sl sa i a c n w a e n i
A L n f Cg &
ny
Ai l g b a o nl o
E n h c e t
e s
a pi e r h
T e r nc
Ca d t e e r g
Ta
Leader in Personalized Medicine

ViroLogic, Inc. Making Individualized Medicine a Reality
First Annual Life Sciences Conference New York, NY November 30, 2004 Lazard