UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
Form 10-K
(Mark One)
| |
☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2024
Or
| |
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 000-50768
ACADIA PHARMACEUTICALS INC.
(Exact Name of Registrant as Specified in Its Charter)
| |
Delaware | 06-1376651 |
(State or Other Jurisdiction of Incorporation or Organization) | (I.R.S. Employer Identification Number) |
| |
12830 El Camino Real, Suite 400 San Diego, California | 92130 |
(Address of Principal Executive Offices) | (Zip Code) |
Registrant’s telephone number, including area code:
(858) 558-2871
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | |
| Title of each class | | Trading Symbol(s) | | Name of each exchange on which registered |
|
| Common Stock, par value $0.0001 per share | | ACAD | | The Nasdaq Stock Market LLC |
|
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Securities Exchange Act of 1934:
| | | | |
Large accelerated filer | ☒ | | Accelerated filer | ☐ |
Non-accelerated filer | ☐ | | Smaller reporting company | ☐ |
| | | Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☒
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Securities Exchange Act of 1934). Yes ☐ No ☒
As of June 28, 2024, the last business day of the registrant’s most recently completed second fiscal quarter, the aggregate market value of the registrant’s common stock held by non-affiliates of the registrant was approximately $2.0 billion, based on the closing price of the registrant’s common stock on the Nasdaq Global Select Market on June 28, 2024 of $16.25 per share.
As of February 18, 2025, 166,788,517 shares of the registrant’s common stock, $0.0001 par value, were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive Proxy Statement to be filed with the Securities and Exchange Commission by April 30, 2025 are incorporated by reference into Part III of this report.
ACADIA PHARMACEUTICALS INC.
TABLE OF CONTENTS
FORM 10-K
For the Year Ended December 31, 2024
PART I
FORWARD-LOOKING STATEMENTS
This report and the information incorporated herein by reference contain forward-looking statements that involve a number of risks and uncertainties, as well as assumptions that, if they never materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such forward-looking statements. Although our forward-looking statements reflect the good faith judgment of our management, these statements can only be based on facts and factors currently known by us. Consequently, forward-looking statements are inherently subject to risks and uncertainties, and actual results and outcomes may differ materially from results and outcomes discussed in the forward-looking statements. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and you are cautioned not to unduly rely upon these statements.
Forward-looking statements can be identified by the use of forward-looking words such as “aims,” “anticipates,” “believes,” “continue,” “could,” “estimates,” “expects,” “hopes,” “intends,” “may,” “plans,” “potential” “predicts,” “pro forma,” “projects,” “seeks,” “should,” “will,” “would,” or other similar words (including their use in the negative), or by discussions of future matters such as the benefits to be derived from our products and our product candidates, the potential market opportunities for our products and our drug candidates, our strategy for the commercialization of our products, our plans for exploring and developing our products for additional indications, the commercialization of DAYBUE or trofinetide in jurisdictions other than the United States (U.S.), our plans and timing with respect to seeking regulatory approvals, the potential commercialization of any of our product candidates that receive regulatory approval, the progress, timing, results or implications of clinical trials and other development milestones and activities involving our products and our product candidates, our strategy for discovering, developing and, if approved, commercializing our product candidates, our existing and potential future collaborations, our estimates of future payments, revenues and profitability, our estimates regarding our capital requirements, future expenses and needs for additional financing, possible changes in legislation, and other statements that are not historical. These statements include but are not limited to statements under the captions “Business,” “Risk Factors,” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” as well as other sections in this report. You should be aware that the occurrence of any of the events discussed under the caption “Risk Factors” and elsewhere in this report could substantially harm our business, results of operations and financial condition and cause our results to differ materially from those expressed or implied by our forward-looking statements. If any of these events occurs, the trading price of our common stock could decline and you could lose all or a part of the value of your shares of our common stock.
The cautionary statements made in this report are intended to be applicable to all forward-looking statements wherever they may appear in this report. We urge you not to place undue reliance on these forward-looking statements, which speak only as of the date of this report. We undertake no obligation to update any forward-looking statements made in this report to reflect events or circumstances after the date of this report or to reflect new information or the occurrence of unanticipated events, except as required by law.
Summary of Risk Factors
We face risks and uncertainties related to our business, many of which are beyond our control. In particular, risks associated with our products and business include:
•Our prospects are highly dependent on the successful commercialization of our products. To the extent we cannot maintain or increase sales of our products, our business, financial condition and results of operations may be materially adversely affected and the price of our common stock may decline.
•Our products may not gain maximal acceptance among physicians, patients, caregivers and the medical community, thereby limiting our potential to generate revenues.
•If we do not obtain regulatory approval of trofinetide outside North America, we will not be able to market trofinetide outside North America, which will limit our commercial revenues.
•Our ability to generate product revenues will be diminished if coverage for our products from commercial or government payors is decreased or if patients have unacceptably high out-of-pocket requirements.
•Our products are subject to ongoing regulatory requirements that could cause us significant expense and delay or limit our ability to generate sales revenues.
•We rely on a limited network of third-party distributors and pharmacies to market and sell our products. If this approach ceases to be effective, commercialization of our products may be adversely affected, and our products may not be profitable.
•Drug development is a long, expensive and unpredictable process with a high risk of failure, and there is no guarantee that our products or product candidates will be successful in ongoing or future clinical trials or obtain regulatory approval.
•Expanded access or compassionate use programs could subject us to additional risks.
•Delays, suspensions and terminations in our clinical trials for our product candidates could result in increased costs to us and delay our ability to generate product revenues.
•If we are unable to attract, retain, and motivate key management, research and development, and sales and marketing personnel, our drug development programs, our research and discovery efforts, and our commercialization plans may be delayed and we may be unable to successfully commercialize our products, or develop our product candidates.
•If we fail to develop, acquire or in-license other product candidates or products, our business and prospects would be limited. Even if we obtain rights to other product candidates or products, we will incur a variety of costs and may never realize the anticipated benefits.
•We have a history of net losses and we may not be able to predict the extent of future losses.
•We may require additional financing in the future to fund our operations. If we cannot raise additional financing in the future, we may be unable to fund our business plan and our future research, development, commercial and manufacturing efforts.
•We expect that our results of operations will fluctuate, which may make it difficult to predict our future performance from period to period.
•Changes in tax laws or regulations that are applied adversely to us or our customers may have a material adverse effect on our business, cash flow, financial condition or results of operations.
•Our ability to use net operating loss carryforwards and certain other tax attributes to offset future taxable income or taxes may be limited.
•Tax authorities could reallocate our taxable income among our subsidiaries, which could increase our overall tax liability.
•Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
•We or the third parties upon whom we depend may be adversely affected by catastrophic events, such as earthquakes, fires, or other natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
•We have incurred, and expect to continue to incur, significant costs as a result of laws and regulations relating to corporate governance and other matters.
•Our business involves the use of hazardous materials, and we and our third-party manufacturers and suppliers must comply with environmental, health and safety laws and regulations, which can be expensive and restrict how we do, or interrupt our, business.
Item 1. Business.
Company Overview
We are a biopharmaceutical company focused on the development and commercialization of innovative medicines that address unmet medical needs in central nervous system (CNS) disorders and rare diseases.
We have two core franchises in neuroscience and neuro-rare diseases. Our neuroscience franchise is anchored by the commercial product NUPLAZID (pimavanserin), which is the first and only drug approved by the U.S. Food and Drug Administration (FDA) for the treatment of hallucinations and delusions associated with PDP. Our neuro-rare disease franchise is anchored by the commercial product DAYBUE, which is the first and only drug approved for the treatment of Rett syndrome. Net product sales from these two commercial products totaled $957.8 million for 2024, compared with $726.4 million for 2023.
In addition to these commercial products, we have a portfolio of product candidates and research programs that are designed to address significant unmet medical needs in CNS disorders and rare diseases. In order to achieve significant long-term growth, we plan to develop our current portfolio, expand our pipeline of early- and late-stage product candidates and expand into areas of rare disease that are adjacent to our existing franchises, including through strategic business development, and make use of our internal capabilities and knowledge.
Our most advanced product candidate is ACP-101 (intranasal carbetocin) for the treatment of hyperphagia in Prader-Willi syndrome (PWS), a neuro rare disease. Hyperphagia is an intense persistent sensation of hunger accompanied by food preoccupations, an extreme drive to consume food, food-related behavior problems, and a lack of normal satiety. In November 2023, we initiated the Phase 3 COMPASS PWS study evaluating the efficacy and safety of ACP-101 for the treatment of hyperphagia in PWS.
Our next most advanced product candidate is ACP-204 for the treatment of Alzheimer’s disease psychosis (ADP). In November 2023, we initiated a Phase 2 study evaluating the efficacy and safety of ACP-204 for the treatment of hallucinations and delusions associated with ADP. We plan to initiate an additional Phase 2 study of ACP-204 in Lewy Body Dementia with Psychosis (LBDP) in the third quarter of 2025.
We have several product candidates in earlier stages of development for the treatment of CNS disorders and rare diseases, including ACP-711 for the treatment of essential tremor, for which we expect to initiate a Phase 2 study in 2026.
Corporate Information
We were originally incorporated in Vermont in 1993 as Receptor Technologies, Inc. We reincorporated in Delaware in 1997. Our global headquarters are in San Diego, California. We also have substantial operations in Princeton, New Jersey and European headquarters in Zug, Switzerland. We maintain a website at www.acadia.com, to which we regularly post copies of our press releases as well as additional information about us. Our filings with the Securities and Exchange Commission (SEC) are available free of charge through our website as soon as reasonably practicable after being electronically filed with or furnished to the SEC. Interested persons can subscribe on our website to email alerts that are sent automatically when we issue press releases, file or furnish our reports with the SEC or post certain other information to our website. Information contained in our website does not constitute a part of this report or our other filings with the SEC.
We own or have rights to various trademarks, copyrights and trade names used in our business, including Acadia®, NUPLAZID® and DAYBUE™. Our logos and trademarks are the property of Acadia Pharmaceuticals Inc. All other brand names or trademarks appearing in this report are the property of their respective holders. Use or display by us of other parties’ trademarks, trade dress, or products in this report is not intended to, and does not, imply a relationship with, or endorsement or sponsorship of us, by the trademark or trade dress owners.
Our Strategy
Our strategy is to build a strong foundation for growth with multiple innovative commercial products and product candidates that address high unmet medical needs and have the potential to be impactful products in our core franchises of neuro-psychiatric disorders and neuro-rare diseases and adjacencies within rare disease. We plan to execute on this strategy by enhancing the growth of the commercial products in our core franchises while expanding our pipeline of product candidates through business development, partnerships and collaborations.
Key elements of our strategy are to:
•Maximize growth of our successful commercialization of NUPLAZID for Parkinson’s disease psychosis in the United States. We intend to accelerate the growth of NUPLAZID by 1) activating consumers with our direct-to-consumer campaigns, 2) leveraging real world evidence, and 3) increasing field force efficiency with greater use of information technology tools. We believe that with an approximate 20% current market share of the 130,000 Parkinson’s disease patients that are treated with an atypical antipsychotic annually in the United States, we have ample opportunity to grow sales of NUPLAZID while delivering increasing annual cash flow to reinvest in overall corporate growth.
•Drive new patient adoption of DAYBUE for the treatment of patients with Rett syndrome in the United States. Our strategy to drive sales growth for DAYBUE in the U.S. is to drive new patient adoption while optimizing the patient experience for those already on therapy thereby achieving and maintaining stable persistency rates. We plan to achieve this by 1) substantially increasing our field force and increasing their utilization of predictive analytics, 2) launching branded direct-to-consumer campaigns, and 3) deploying an omni-channel strategy to bring DAYBUE clinical data to life by leveraging the growing body of real world experience, including updated data from the ongoing LOTUS study. Based on the number of Rett syndrome patients in the U.S., we believe there remains a substantial opportunity for new patient adoption. We estimate that between 5,500 and 5,800 people suffer from Rett Syndrome in the U.S. We estimate a prevalent population of 6,000 to 9,000 people in the U.S.
•Expand trofinetide to markets outside the U.S. for the treatment of patients with Rett syndrome. We intend to make trofinetide available in markets outside the U.S. where Rett syndrome remains a significant unmet medical need as DAYBUE is the only currently approved treatment for Rett syndrome worldwide, and is currently only approved in the United States and Canada. In the European Union, where we estimate a prevalent population of 9,000 to 12,000 Rett syndrome patients, we filed our marketing authorization application (MAA) with the European Medicines Agency (EMA) in January 2025 and are building out our launch team in anticipation of a potential approval in the first quarter of 2026. We anticipate initial revenues from managed access programs in certain European Union countries sometime in 2025. In Japan, where we estimate a prevalent population of 1,000 to 2,000 Rett syndrome patients, we are engaged in ongoing discussions with the Pharmaceuticals and Medical Devices Agency (PMDA) and anticipate initiating a small Phase 3 study by the third quarter of 2025 to support a future MAA to that regulatory agency. In Canada, where we estimate a prevalent population of 600 to 900 Rett syndrome patients, Health Canada granted marketing authorization of DAYBUE in October 2024 and we anticipate first commercial sales in Canada by the third quarter of this year.
•Advance our late-stage product candidates to drive further growth. We have an ongoing Phase 3 study of ACP-101 in PWS and a Phase 2 study of ACP-204 in ADP. We plan to initiate an additional Phase 2 study of ACP-204 in LBDP in the third quarter of 2025. In 2026, we plan to initiate a Phase 2 study of ACP-711 in essential tremor.
•Develop our early-stage product candidates and other business development opportunities. We have a deep and diverse early-stage portfolio of product candidates that includes disclosed and undisclosed programs focused on neuropsychiatric and neuro-rare diseases that we believe represent significant opportunities to continue to build on our current growth. We continue to look to add product candidates to the portfolio through business development.
In addition, we are investing in the development of four core strategic capabilities to support and accelerate our growth over the long term. These capabilities form the foundation of our operational strategy and are designed to enhance our competitive position, drive innovation, and meet the evolving needs of patients worldwide. The four core capabilities in which we are investing are:
1.Precision Medicine. We are focused on integrating strategies that address the variability in patients’ responses to therapies by identifying and incorporating the use of targeted biomarkers at the early stages of drug development. Through the incorporation of precision medicine, we aim to increase the likelihood of clinical success, enhance treatment outcomes, and reduce overall development costs.
2.Data Innovation. We plan to leverage advanced technologies, including artificial intelligence (AI) and machine learning (ML) throughout our business from discovery through commercialization. By utilizing these tools, we hope to generate, analyze, and apply data in innovative ways to enhance the efficiency and effectiveness of our operations in ways that ultimately drive growth and improve patient outcomes.
3.Globalization. We are building targeted capabilities beyond the United States including in the European Union, Japan and Canada in order to meet patient needs on a global scale.
4.Patient Empowerment. We seek to engage with patients, caregivers and advocacy organizations and incorporate their feedback into our drug development and commercialization efforts. By creating an integrated ecosystem of our patients, caregivers and advocates, our goal is to transform patient feedback and needs into actionable plans for product development and support.
Our Pipeline
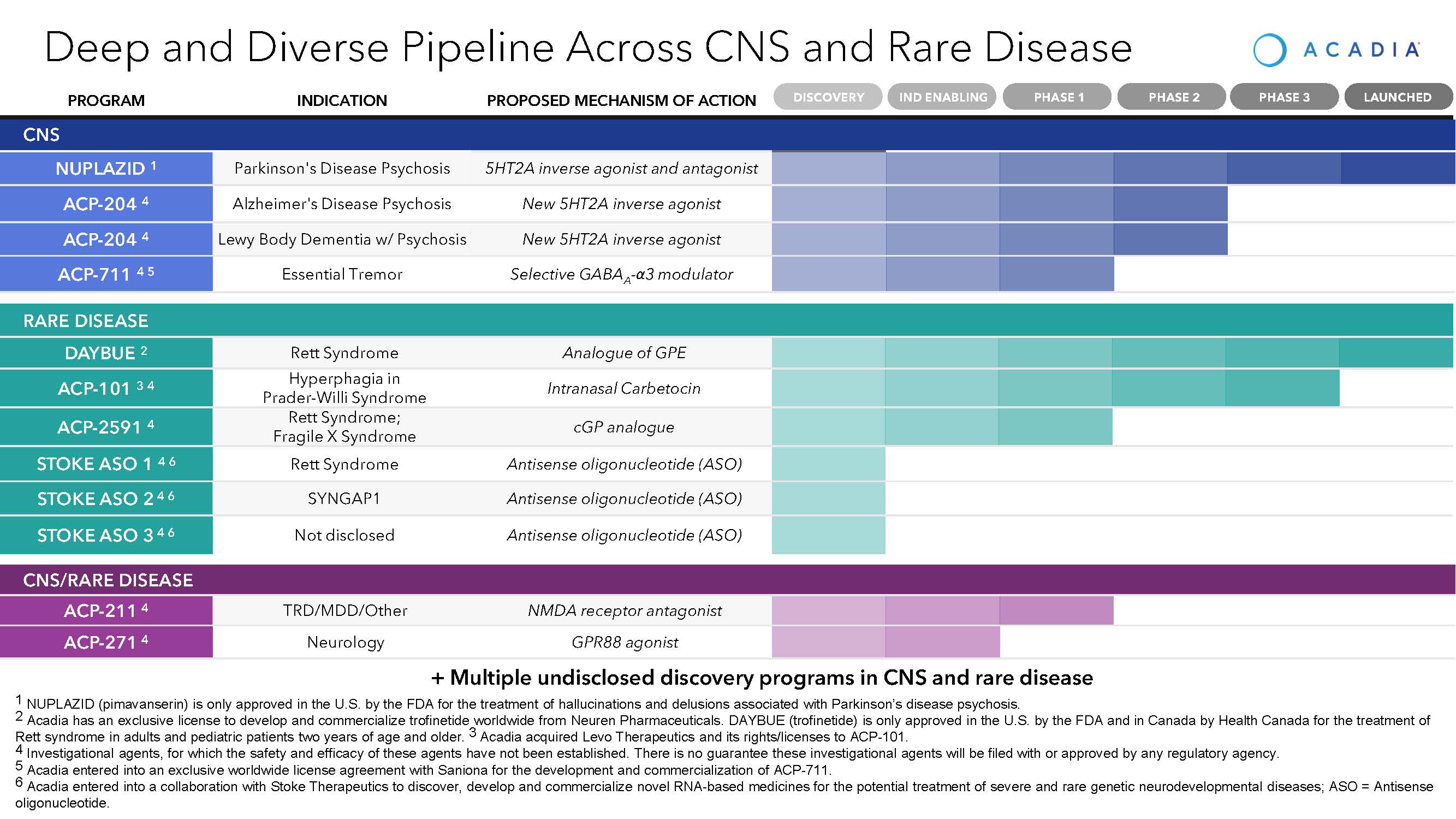
NUPLAZID (pimavanserin) as a Treatment for Parkinson’s Disease Psychosis
Pimavanserin is a chemical entity that we discovered and developed. NUPLAZID is a selective serotonin inverse agonist/antagonist preferentially targeting the 5-HT2A receptor, a key serotonin receptor that plays an important role in psychosis. Through this novel mechanism, NUPLAZID demonstrated significant efficacy in reducing the hallucinations and delusions associated with PDP without negatively impacting motor function in our Phase 3 pivotal trial. NUPLAZID has the potential to avoid many of the debilitating side effects of existing antipsychotics, none of which are approved by the FDA in the treatment of PDP. We hold worldwide commercialization rights to NUPLAZID for all indications and have established a broad patent portfolio, which includes numerous issued patents in the United States, Europe, and several additional countries. NUPLAZID is available in 34 mg capsule and 10 mg tablet dosage forms.
Parkinson’s disease is the second most common neurodegenerative disorder after Alzheimer’s disease. According to the Parkinson’s Disease Foundation, about one million people in the United States and more than 10 million people globally suffer from this disease and this number is projected to increase to over 12 million by 2040. Approximately 50% of Parkinson’s patients will experience psychosis over the course of their disease. We estimate that approximately 130,000 Parkinson’s disease patients are treated with an atypical antipsychotic annually in the United States. Of these patients, we estimate that approximately 20% are currently being treated with NUPLAZID.
Parkinson’s disease is more common in people over 60 years of age and the prevalence of this disease is expected to increase significantly as the population ages. At this time, the fastest growing neurological disorder in the world is Parkinson’s disease. The development of psychosis in patients with Parkinson’s disease substantially contributes to the burden of Parkinson’s disease and deeply affects their quality of life. PDP is associated with a diminished quality of life, nursing home placement, and increased caregiver stress and burden.
NUPLAZID was approved by the FDA in April 2016 for the treatment of hallucinations and delusions associated with PDP. It is the only drug approved in the United States for this condition and is marketed under the tradename NUPLAZID in the United States.
As the first and only drug approved by the FDA for the treatment of hallucinations and delusions associated with PDP, NUPLAZID provides an innovative approach to the treatment of PDP without compromising motor control and potentially avoiding many of the debilitating side effects of existing antipsychotics.
In connection with the FDA approval of NUPLAZID, we agreed to four post-marketing commitments. All four commitments have now been fulfilled within the agreed upon timelines.
Other Indications Recently Evaluated with Pimavanserin
In March 2024 we reported top-line results from a Phase 3 study of pimavanserin for the treatment of the negative symptoms of schizophrenia. Pimavanserin did not demonstrate a statistically significant improvement over placebo on the study’s primary endpoint, the change from baseline to week 26 on the Negative Symptom Assessment-16. The safety and tolerability profile of pimavanserin was consistent with previous clinical trials, showing a low rate of adverse events.
In October 2024 we completed a Phase 2 trial to evaluate the efficacy and safety of pimavanserin for the treatment of irritability associated with autism spectrum disorder in pediatric populations. The trial did not meet either of its primary or secondary endpoints. With the completion of the trial, we believe we now have completed the FDA’s requirements to qualify for a pediatric exclusivity for pimavanserin. If the pediatric exclusivity is granted, exclusivity of the NUPLAZID franchise would be extended by six months.
We do not intend to further explore pimavanserin in these or any additional indications.
DAYBUE (trofinetide) as a Treatment for Rett Syndrome
Trofinetide is a novel synthetic analog of the amino-terminal tripeptide of insulin-like growth factor 1 (IGF-1) designed to treat the core symptoms of Rett syndrome by reducing neuroinflammation and supporting synaptic function. We acquired an exclusive North American license to develop and commercialize trofinetide from Neuren Pharmaceuticals Limited (Neuren) in August 2018. In July 2023, we expanded the 2018 licensing agreement with Neuren to acquire rights to trofinetide outside of North America as well as global rights to Neuren’s development candidate NNZ-2591 in Rett syndrome and Fragile X syndrome. Trofinetide has been granted FDA Fast Track Status and Orphan Drug Designation in the U.S. and Orphan Designation in Europe.
Rett syndrome is a debilitating neurological disorder that occurs primarily in females following apparently normal development for the first six months of life. Rett syndrome has been most often misdiagnosed as autism, cerebral palsy, or non-specific developmental delay. Rett syndrome is caused by mutations on the X chromosome on a gene called MECP2. There are more than 200 different mutations found on the MECP2 gene that interfere with its ability to generate a normal gene product. Rett syndrome occurs worldwide in approximately one of every 10,000 to 15,000 female births causing problems in brain function that are responsible for cognitive, sensory, emotional, motor and autonomic function. Typically, between six to eighteen months of age, patients experience a period of rapid decline with loss of purposeful hand use and spoken communication and inability to independently conduct activities of daily living. Symptoms also include seizures, disorganized breathing patterns, an abnormal side-to-side curvature of the spine (scoliosis) and sleep disturbances. Based on 2024 claims data, we estimate that between 5,500 and 5,800 people are diagnosed with Rett Syndrome in the U.S. We estimate a prevalent population of 6,000 to 9,000 people in the U.S.
In March 2023, the FDA approved DAYBUE for the treatment of Rett syndrome, making it the first and only drug approved for this condition. DAYBUE became available for prescription in the United States in April 2023. The FDA approval of DAYBUE for the treatment of Rett syndrome was based on the positive results from our pivotal Phase 3 LAVENDER™ study which demonstrated statistically significant and clinically meaning improvement over placebo for both co-primary endpoints in the study as well as the key secondary endpoint of the study.
In addition, we were granted a Rare Pediatric Disease Priority Review Voucher (PRV) following the FDA approval of DAYBUE. In December 2024, we completed the sale of our PRV for $150 million before fees and expenses, of which we paid Neuren one-third of the net proceeds, pursuant to the license agreement between the companies.
In connection with the FDA approval of DAYBUE, we are required to conduct post-marketing work, including a nonclinical carcinogenicity studies and nonclinical in vitro studies.
In October 2024, Health Canada granted marketing authorization of DAYBUE (trofinetide) for the treatment of Rett syndrome in adult and pediatric patients two years of age and older under the Priority Review process, making DAYBUE the first and only drug approved in Canada for the treatment of Rett syndrome.
In January 2025, we announced the submission of a MAA with the EMA. Based on typical review timelines, we expect to receive approval for this submission in the first quarter of 2026.
ACP-101 as a Treatment for Prader-Willi syndrome
Carbetocin nasal spray (ACP-101) is an investigational drug being developed for the treatment of hyperphagia in PWS. Carbetocin has improved drug qualities relative to oxytocin, including an extended half-life and greater specificity for the oxytocin receptor compared to vasopressin receptors which could provide meaningful efficacy with an attractive safety profile in patients with PWS. For the treatment of PWS specifically, a central nervous system disorder, an intranasal formulation of carbetocin was developed, which provides direct delivery of the drug to the brain, allowing for reduced systemic exposure and the potential for side effects. We acquired Levo Therapeutics and worldwide rights to carbetocin nasal spray in June 2022. Carbetocin nasal spray has been granted Orphan Drug, Fast Track, and Rare Pediatric Disease designations by the FDA.
PWS is a rare neurobehavioral genetic disorder that affects both males and females. PWS is estimated to affect approximately 1 in 15,000 to 1 in 25,000 live births worldwide, or 8,000 to 10,000 patients in the United States. PWS affects the functioning of the hypothalamus and other aspects of the brain with symptoms varying by individual. The most common symptom is hyperphagia, which is an unrelenting lack of satiety, to which a deficiency in oxytocin is believed to be contributory. Oxytocin is a natural hormone that regulates several functions in the body, including hunger, anxiety, social behavior and bonding. Individuals living with PWS have fewer oxytocin-producing neurons in the brain. Other defining features of the syndrome may include altered metabolism, developmental delays, behavioral challenges and moderate cognitive deficits. Patients may also experience bone disorders, high pain tolerance, sleep disturbances, gastrointestinal issues, respiratory and temperature regulation abnormalities. There is no FDA-approved treatment for the hyperphagia associated with PWS.
In the fourth quarter of 2023, we initiated the Phase 3 COMPASS PWS study evaluating the efficacy and safety of ACP-101 for the treatment of hyperphagia in PWS. COMPASS PWS is a 12-week, double-blind, randomized, placebo-controlled global Phase 3 trial evaluating the efficacy and safety of carbetocin nasal spray 3.2 mg three times daily (TID) in approximately 170 children and adults aged five to 30 years with PWS. The primary efficacy endpoint of the study is change from baseline to week 12 on the hyperphagia questionnaire for clinical trials (HQ-CT) score, a caregiver assessment for hyperphagia-related behaviors. Participants who complete the Phase 3 study will be eligible to enroll in a long-term, open-label extension study, COMPASS OLE, designed to investigate the safety and tolerability of long-term treatment with ACP-101.
We expect to complete enrollment in our Phase 3 COMPASS PWS study in in the fourth quarter of 2025 and subsequently report top-line results sometime in the first half of 2026. In the event of positive trial results, we believe ACP-101 has the potential for FDA approval in the fourth quarter of 2026.
ACP-204
ACP-204 is a new chemical entity targeting the serotonergic system, compared with most antipsychotics on the market today which are thought to work predominantly through blocking dopamine and in particular, the dopamine D2 receptor. ACP-204 features a combination of structural changes compared with pimavanserin and reduced off-target effects along with equal or increased potency. Specifically with ACP-204, we believe we may have an opportunity to maximize the efficacy potential, while reducing the risk of QT prolongation.
ACP-204 as a Treatment for Alzheimer’s Disease Psychosis
An estimated over 6.0 million people in the United States are living with Alzheimer’s disease dementia and studies suggest that approximately 30% of them, or 1.8 million people, have psychosis, commonly consisting of delusions and hallucinations. Approximately 800,000 to 850,000 patients in the United States are currently treated for ADP and of those treated, approximately two-thirds are treated with off-label anti-psychotics.
Symptoms of ADP are often persistent and may occur with increasing frequency with progression of disease as patients become more impaired. Serious consequences have been associated with persistent or severe psychosis in persons with dementia such as repeated hospital admissions, earlier progression to nursing home care, severe dementia, and death. There are currently no FDA-approved treatments for ADP. Off-label use of typical and atypical antipsychotics is associated with modest and often equivocal efficacy in these patients. In addition, use of currently available antipsychotics is associated with a significant acceleration in cognitive decline in patients with dementia as well as numerous off-target toxicities, thus negatively impacting the primary illness. The cognitive effects of treatment with an atypical antipsychotic were evaluated in the National Institute of Mental Health Clinical Antipsychotic Trials of Intervention Effectiveness–Alzheimer’s Disease (CATIE-AD) study. In this study, patients on any atypical antipsychotic had significantly greater rates of decline in cognitive function compared to patients on placebo. This pronounced negative impact of currently used antipsychotics on cognitive function is believed to be associated with the common pharmacologic property of these drugs, namely blocking of dopamine receptors. Atypical antipsychotics are associated with a number of off-target and dose-limiting side effects, such as extrapyramidal symptoms, orthostatic hypotension, hematologic abnormalities, and metabolic, gastrointestinal and sedative effects. These off-target toxicities are associated with increased risk for falls, infection, aspiration pneumonia, and other serious complications in this vulnerable patient population. With no approved therapies for the treatment of patients with ADP and current off-label use of atypical antipsychotics carrying significant morbidity risks including worsening in cognitive decline and other off target toxicities, we believe that ADP represents an area of high unmet need.
We completed Phase 1 studies of ACP-204 in over 200 patients; data to date support its target product profile as a potential treatment for ADP.
In the fourth quarter of 2023, we initiated the Phase 2 RADIANT study of ACP-204 for the treatment of hallucinations and delusions associated with ADP. The Phase 2 RADIANT study is part of a Phase 2 / Phase 3 program that includes three studies: a single Phase 2 study and two Phase 3 studies which have almost identical design. The Phase 2 RADIANT study is a global, multi-center, randomized, double-blind, placebo-controlled trial that will enroll approximately 318 patients and evaluate ACP-204 30 mg and 60 mg doses compared to placebo. The primary endpoint is change from baseline to week 6 on the Scale for the Assessment of Positive Symptoms–Hallucinations and Delusions subscales (SAPS-H+D) total score. The clinical trial sites will enroll seamlessly from Phase 2 into Phase 3. Patients who complete the study will have the option of participating in our LUMINOUS long-term open-label extension study.
We expect to complete enrollment in the Phase 2 RADIANT study in the first quarter of 2026 and subsequently report top-line results around mid-2026.
ACP-204 as a Treatment for Lewy Body Dementia with Psychosis
In January 2025, we announced plans to evaluate ACP-204 for the treatment of LBDP. LBDP is a progressive brain disorder that affects thinking, movement, mood and behavior. LBDP is associated with abnormal deposits of a protein called alpha-synuclein in the brain.
There are no approved therapies for LBDP. More than 1 million people in the United States may be living with Lewy Body Dementia, of which an estimated 50% to 75% experience psychosis during the course of the disease. We estimate that approximately 200,000 of these patients are being treated with antipsychotics today. We plan to initiate a Phase 2 study in LBDP in the third quarter of 2025.
ACP-711 as a Treatment for Essential Tremor
In November 2024, we entered into an exclusive worldwide license agreement with Saniona A/S (Saniona) for the development and commercialization of ACP-711 (formerly SAN711). We intend to study ACP-711 as a potential treatment for essential tremor. ACP-711 is a highly selective GABAA-α3 positive allosteric modulator. Under the terms of the license agreement, Saniona received $28 million upfront plus potential milestone payments of up to $582 million. In addition, Saniona is eligible to receive tiered royalties of mid-single digits to low double digits on net sales of commercial products
that may result from development of ACP-711. The potential milestone payments to Saniona consist of up to $147 million subject to achievement of development and commercial milestones related to potential first and second indications, and up to $435 million subject to achievement of thresholds of annual net sales of ACP-711 worldwide. Acadia will lead further clinical development, regulatory submissions, and global commercialization efforts for ACP-711 while also providing financial support for Saniona’s ongoing Phase 1 study and preparations for Phase 2.
Essential tremor is a neurological disorder that causes involuntary shaking in the hands, head, voice, and sometimes the legs. Approximately 7 million people in the United States suffer from this disorder and an estimated 1 million people are being treated for it with some form of medication. Due to the limited treatment options available to sufferers of essential tremor, we see an opportunity to provide much needed innovation for this disorder.
We plan to initiate a Phase 2 study of ACP-711 in essential tremor in 2026 after completion of an ongoing Phase 1 study.
Early-stage Research Programs and Product Candidates
ACP-211
ACP-211 is a NMDA receptor agonist being evaluated for potential use in the treatment of Treatment-resistant depression (TRD), major depressive disorder (MDD) and other potential rare CNS indications and is in Phase 1.
ACP-2591
ACP-2591 is a cGP analogue for which we obtained global rights to evaluate for use in Rett syndrome and Fragile X syndrome from Neuren as part of our expanded licensing agreement in July 2023. ACP-2591 is currently in Phase 1 development.
ACP-271
ACP-271 is a GPR88 agonist being evaluated in neurology and is currently in the IND-enabling stage.
Antisense Oligonucleotide (ASO) Programs
In January 2022, we entered into a license and collaboration with Stoke Therapeutics, Inc. (Stoke) to discover, develop and commercialize novel RNA-based medicines for the potential treatment of severe and rare genetic neurodevelopmental diseases of the CNS. The collaboration includes three programs: SYNGAP1 syndrome, Rett syndrome (MECP2) and an undisclosed CNS target of mutual interest. The programs are currently in various stages of discovery.
Competition
We face, and will continue to face, intense competition from pharmaceutical and biotechnology companies, as well as numerous academic and research institutions and governmental agencies, both in the United States and abroad. We compete, or will compete, with existing and new products being developed by our competitors. Some of these competitors have products or are pursuing the development of pharmaceuticals that target the same diseases and conditions that our research and development programs target.
For example, the use of NUPLAZID for the treatment of PDP competes with off-label use of various antipsychotic drugs, including the generic drugs quetiapine, clozapine, risperidone, aripiprazole, and olanzapine.
DAYBUE competes indirectly with off-label usage of branded and generic prescription medications targeted at individual symptoms of Rett syndrome, including antiepileptics, antipsychotics, antidepressants and benzodiazepines. In addition, Anavex has a product, Anavex 2-73, in development for the potential treatment of Rett syndrome and Taysha Gene Therapies is conducting clinical trials of a gene therapy to treat Rett syndrome. Neurogene started an early phase clinical trial of its investigational adeno-associated virus gene therapy candidate, NGN-401, delivered using intracerebroventricular administration to treat Rett Syndrome. Several academic institutions and pharmaceutical companies are currently conducting clinical trials for the treatment of various symptoms of Rett syndrome, including Unravel Bio and Vanderbilt University Medical Center, which are jointly conducting an early stage study with vorinostat (RVL-001).
Other competitors may have a variety of drugs in development or awaiting approval from the FDA or comparable foreign regulatory authorities that could reach the market and become established before we have a product to sell for the applicable disorder. Our competitors may also develop alternative therapies that could further limit the market for any drugs that we may develop. Many of our competitors are using technologies or methods different or similar to ours to identify and validate drug targets and to discover and develop drug candidates. Many of our competitors and their collaborators have significantly greater experience than we do in the following:
•identifying and validating targets;
•screening compounds against targets;
•preclinical studies and clinical trials of potential pharmaceutical products;
•obtaining FDA and other regulatory approvals; and
•commercializing pharmaceutical products.
In addition, many of our competitors and their collaborators have substantially greater advantages in the following areas:
•research and development resources;
•manufacturing capabilities;
•sales and marketing; and
•production and testing facilities.
Smaller companies also may prove to be significant competitors, particularly through proprietary research discoveries and collaboration arrangements with large pharmaceutical and established biotechnology companies. Many of our competitors have products that have been approved or are in advanced development and may develop superior technologies or methods to identify and validate drug targets and to discover novel small molecule drugs. We face competition from other companies, academic institutions, governmental agencies and other public and private research organizations for collaborative arrangements with pharmaceutical and biotechnology companies, in recruiting and retaining highly qualified scientific, sales and marketing, and management personnel and for licenses to additional technologies. Our competitors, either alone or with their collaborators, may succeed in developing technologies or drugs that are more effective, safer, more affordable, or more easily administered than ours and may achieve patent protection or commercialize drugs sooner than us. Our competitors may also develop alternative therapies that could further limit the market for any drugs that we may develop. Our failure to compete effectively could have a material adverse effect on our business.
Intellectual Property
We currently hold approximately 54 issued U.S. patents and a significant number of related issued foreign patents. We have also exclusively licensed rights to an additional 23 issued U.S. patents, and a number of related foreign patents. Patents and other proprietary intellectual property rights are an essential element of our business. Our strategy is to file patent applications in the United States and any other country that represents an important potential commercial market to us. In addition, we seek to protect our technology and inventions (and improvements to inventions) that are important to the development of our business. Our patent applications claim proprietary technology, including chemical synthetic or manufacturing methods, drug assays, novel compounds, compositions, formulations and methods of treatment. We also rely upon trade secrets, including technologies that may be used to discover and validate targets, to identify and develop novel drugs, as well as manufacturing or clinical development technologies, among others. We protect our trade secrets by, among other things, requiring employees and third parties who have access to our proprietary information to sign confidentiality and nondisclosure agreements. We are a party to various license agreements that give us rights to use certain technologies in our research and development, subject to certain limitations.
Pimavanserin
We currently hold 26 U.S. patents that relate to pimavanserin, NUPLAZID and methods of use of pimavanserin. Ten of these are Orange Book-listed patents that relate to pimavanserin, NUPLAZID and our approved indication, and cover the general formula of the compound, the composition of matter, with claims specifically directed to pimavanserin and salts thereof, the specific polymorph form of pimavanserin, the approved formulations, and the use thereof for treating our approved indication. The composition of matter patent covering pimavanserin and salts thereof currently has an expiration date in 2030, including a patent term extension approved by the U.S. Patent and Trademark Office. The patents covering the polymorph form and the use of pimavanserin or NUPLAZID for our approved indication are currently set to expire between 2024 and 2028. These patent terms include adjustments made by the U.S. Patent and Trademark Office, but not extensions.
In the United States, under the Drug Price Competition and Patent Term Restoration Act of 1984, commonly known as “Hatch-Waxman,” we are permitted to extend the term of one U.S. patent for pimavanserin or the use thereof. Patent terms may be subject to change not only due to potential patent term extensions but also to any terminal disclaimer that reduces patent term, as well as other factors. Because the U.S. patent laws and judicial interpretations thereof change, modifications or new interpretations of the laws may impact our patent terms.
The remaining 16 U.S. patents relating to pimavanserin that have been issued to us cover methods of use of pimavanserin, salts and methods of manufacturing pimavanserin. We also have foreign patents that cover pimavanserin and polymorphs in Europe and Asia as well as in Australia, Canada, Mexico and other countries.
We continue to prosecute patent applications directed to pimavanserin, formulations of pimavanserin, methods of manufacturing, and to methods of treating various diseases using pimavanserin, either alone or in combination with other agents, worldwide. For example, in late 2019 and in 2020, we obtained and listed in the Orange Book six additional U.S. issued patents, two patents directed to method of use for our 10 mg tablet, expiring in 2037, and four patents directed to our 34 mg capsule formulation, each expiring in 2038.
Trofinetide
We currently hold the exclusive licenses to 8 U.S. patents from Neuren that relate to trofinetide, methods of manufacturing and methods of use of trofinetide. We also hold a U.S. patent to crystalline trofinetide. Three of the U.S. patents are listed in the Orange Book, including a patent claiming the use of trofinetide for treating Rett syndrome. The use patent for treating Rett syndrome has an expiration date in 2032. Subject to a patent term extension request, the expiration date of such patent may be extended to January 2036. We also hold the exclusive licenses to issued foreign patents that relate to the use of trofinetide in Europe and Asia as well as in Australia, Canada, Mexico and other countries.
Under the license agreement with Neuren, we continue to file and prosecute patent applications directed to trofinetide, formulations of trofinetide, methods of manufacturing and methods of treating Rett syndrome using trofinetide.
ACP-101
We currently hold one U.S. patent for methods of use of ACP-101 in PWS, and we hold licenses from Ferring International Center SA to one U.S. patent to ACP-101 for use in PWS. Those patents expire in 2035 and 2039, respectively. These patent terms include adjustments made by the U.S. Patent and Trademark Office, but not extensions. We hold and license a significant number of foreign patents relating to the use of ACP-101 in PWS, including in Europe and Japan.
We continue to file and prosecute patent applications directed to ACP-101 worldwide.
ACP-204
We currently hold two U.S. patents that relate to ACP-204 and methods of use of ACP-204. The patents cover the general formula of the compound, the composition of matter (with claims specifically directed to ACP-204 and salts thereof), and the use thereof for treating certain indications, including ADP and LBDP. The composition of matter patent covering ACP-204 and salts thereof currently has an expiration date in 2038. These patent terms include adjustments made by the U.S. Patent and Trademark Office, but not extensions. We also hold issued foreign patents in Australia, China and Japan.
We continue to file and prosecute patent applications directed to ACP-204 worldwide.
ACP-711
We currently hold the exclusive licenses to one U.S. patent that relates to ACP-711 from Saniona A/S, with claims specifically directed to ACP-711 and salts thereof. The composition of matter patent covering ACP-711 and salts thereof currently has an expiration date in 2039. This parent term includes adjustments made by the U.S. Patent and Trademark Office, but not extensions. We also hold the exclusive licenses to issued foreign patents in Europe, parts of Asia and Mexico, as well as other countries.
We continue to file and prosecute patent applications directed to ACP-711 worldwide.
Government Regulation
Our business activities, including the manufacturing and marketing of our products and any future approved products and our ongoing research and development activities, are subject to extensive regulation by numerous governmental authorities in the United States and other countries. Before marketing in the United States, any new drug developed by us must undergo rigorous preclinical testing, clinical trials and an extensive regulatory clearance process implemented by the FDA under the Federal Food, Drug, and Cosmetic Act, as amended. The FDA regulates, among other things, the development, testing, manufacture, safety, efficacy, record keeping, labeling, storage, approval, advertising, promotion, import, export, sale and distribution of biopharmaceutical products. The regulatory review and approval process, which includes preclinical testing and clinical trials of each product candidate, is lengthy, expensive and uncertain. Moreover, government coverage and reimbursement policies will both directly and indirectly impact our ability to successfully commercialize our current products and any future approved products, and such coverage and reimbursement policies will be impacted by enacted and any applicable future healthcare reform and drug pricing measures. In addition, we are subject to state and federal laws, including, among others, anti-kickback laws, false claims laws, data privacy and security laws, and transparency laws that restrict certain business practices in the pharmaceutical industry.
In the United States, drug product candidates intended for human use undergo laboratory and animal testing until adequate proof of safety is established. Clinical trials for new product candidates are then typically conducted in humans in three sequential phases that may overlap. Phase 1 trials involve the initial introduction of the product candidate into healthy human volunteers. The emphasis of Phase 1 trials is on testing for safety or adverse effects, dosage, tolerance, metabolism, distribution, excretion and clinical pharmacology. Phase 2 involves studies in a limited patient population to determine the initial efficacy of the compound for specific targeted indications, to determine dosage tolerance and optimal dosage, and to identify possible adverse side effects and safety risks. Once a compound shows initial evidence of effectiveness and is found to have an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to more fully evaluate clinical outcomes. Before commencing clinical investigations in humans, we or our collaborators must submit an Investigational New Drug Application (IND), to the FDA.
Regulatory authorities, Institutional Review Boards and Data Monitoring Committees may require additional data before allowing the clinical studies to commence, continue or proceed from one phase to another, and could demand that the studies be discontinued or suspended at any time if there are significant safety issues. Clinical testing must also meet requirements for clinical trial registration, institutional review board oversight, informed consent, health information privacy, and good clinical practices (GCPs). Additionally, the manufacture of our drug product must be done in accordance with current good manufacturing practices (cGMPs).
To establish a new product candidate’s safety and efficacy, the FDA requires companies seeking approval to market a drug product to submit extensive preclinical and clinical data, along with other information, for each indication for which the product will be labeled. The data and information are submitted to the FDA in the form of a New Drug Application (NDA), which must be accompanied by payment of a significant user fee unless a waiver or exemption applies. Generating the required data and information for an NDA takes many years and requires the expenditure of substantial resources. Information generated in this process is susceptible to varying interpretations that could delay, limit or prevent regulatory approval at any stage of the process. The failure to demonstrate adequately the quality, safety and efficacy of a product candidate under development would delay or prevent regulatory approval of the product candidate. Under applicable laws and FDA regulations, each NDA submitted for FDA approval is given an internal administrative review within 60 days following submission of the NDA. If deemed sufficiently complete to permit a substantive review, the FDA will “file” the NDA. The FDA can refuse to file any NDA that it deems incomplete or not properly reviewable. The FDA has established internal goals of eight months from submission for priority review of NDAs that cover new product candidates that offer major advances in treatment or provide a treatment where no adequate therapy exists, and 12 months from submission for the standard review of NDAs. However, the FDA is not legally required to complete its review within these periods, these
performance goals may change over time and the review is often extended by FDA requests for additional information or clarification. Moreover, the outcome of the review, even if generally favorable, may not be an actual approval but a “complete response letter” that describes additional work that must be done before the NDA can be approved. Before approving an NDA, the FDA can choose to inspect the facilities at which the product is manufactured and will not approve the product unless the manufacturing facility complies with cGMPs. The FDA may also audit sites at which clinical trials have been conducted to determine compliance with GCPs and data integrity. The FDA’s review of an NDA may also involve review and recommendations by an independent FDA advisory committee, particularly for novel indications. The FDA is not bound by the recommendation of an advisory committee.
In addition, delays or rejections may be encountered based upon changes in regulatory policy, regulations or statutes governing product approval during the period of product development and regulatory agency review.
Before receiving FDA approval to market a potential product, we or our collaborators must demonstrate through adequate and well-controlled clinical studies that the potential product is safe and effective in the patient population that will be treated. In addition, under the Pediatric Research Equity Act (PREA), an NDA or supplement to an NDA must contain data or a plan to collect such data to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective, unless a waiver applies. If regulatory approval of a potential product is granted, this approval will be limited to those disease states and conditions for which the product is approved. Marketing or promoting a drug for an unapproved indication is generally prohibited. Furthermore, FDA approval may entail ongoing requirements for risk management, including post-marketing, or Phase 4, studies. Even if approval is obtained, each marketed product, is subject to payment of a significant annual program user fee and continuing review and periodic inspections by the FDA. Discovery of previously unknown problems with a product, manufacturer or facility may result in restrictions on the product or manufacturer, including labeling changes, warning letters, costly recalls or withdrawal of the product from the market.
Any drug is likely to produce some toxicities or undesirable side effects in animals and in humans when administered at sufficiently high doses and/or for sufficiently long periods of time. Unacceptable toxicities or side effects may occur at any dose level at any time in the course of studies in animals designed to identify unacceptable effects of a product candidate, known as toxicological studies, or during clinical trials of our potential products. The appearance of any unacceptable toxicity or side effect could cause us or regulatory authorities to interrupt, limit, delay or abort the development of any of our product candidates. Further, such unacceptable toxicity or side effects could ultimately prevent a potential product’s approval by the FDA or foreign regulatory authorities for any or all targeted indications or limit any labeling claims and market acceptance, even if the product is approved.
In addition, as a condition of approval, the FDA may require an applicant to develop a risk evaluation and mitigation strategy (REMS). A REMS uses risk minimization strategies beyond the professional labeling to ensure that the benefits of the product outweigh the potential risks. To determine whether a REMS is needed, the FDA will consider the size of the population likely to use the product, seriousness of the disease, expected benefit of the product, expected duration of treatment, seriousness of known or potential adverse events, and whether the product is a new molecular entity. REMS can include medication guides, physician communication plans for healthcare professionals, and elements to assure safe use (ETASU). ETASU may include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The FDA may require a REMS before approval or post-approval if it becomes aware of a serious risk associated with use of the product. The requirement for a REMS can materially affect the potential market and profitability of a product.
We and our collaborators and contract manufacturers also are required to comply with the applicable FDA GMP regulations. cGMP regulations include requirements relating to quality control and quality assurance as well as the corresponding maintenance of records and documentation. Manufacturing facilities are subject to inspection by the FDA. These facilities must be approved before we can use them in commercial manufacturing of our potential products and must maintain ongoing compliance for commercial product manufacture, testing, storage and distribution. The FDA may conclude that we or our collaborators or contract manufacturers are not in compliance with applicable cGMP requirements and other FDA regulatory requirements, which may result in delay or failure to approve applications, warning letters, product recalls and/or imposition of fines or penalties.
If a product is approved, we must also comply with post-marketing requirements, including, but not limited to, compliance with advertising and promotion laws enforced by various government agencies, including the FDA’s Office of Prescription Drug Promotion, and through such laws as federal and state anti-fraud and abuse laws, including anti-kickback and false claims laws, healthcare information privacy and security laws, post-marketing safety surveillance, and disclosure of payments or other transfers of value to healthcare professionals and entities. In addition, we are subject to other federal and state regulation including, for example, the implementation of corporate compliance programs.
In order to distribute products commercially, we must comply with state laws that require the registration of manufacturers and wholesale distributors of pharmaceutical products in a state, including, in certain states, manufacturers and distributors who ship products into the state even if such manufacturers or distributors have no place of business within the state. Some states also impose requirements on manufacturers and distributors to establish the pedigree of product in the chain of distribution, including some states that require manufacturers and others to adopt new technology capable of tracking and tracing product as it moves through the distribution chain.
Coverage and Reimbursement
Sales of our products and our product candidates, if approved, depend and will depend, in part, on the extent to which such products will be covered by third-party payors, such as government health care programs, commercial insurance and managed healthcare organizations. These third-party payors are increasingly limiting coverage and/or reducing reimbursements for medical products and services. A third-party payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Further, one payor’s determination to provide coverage for a drug product does not ensure that other payors will also provide coverage for the drug product. Coverage policies and third-party payor reimbursement rates may change at any time. Therefore, even if favorable coverage and reimbursement status is attained for one or more products for which we receive marketing approval, less favorable coverage policies and reimbursement rates may be implemented in the future. In addition, the U.S. government, state legislatures and foreign governments have continued implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our net revenue and results. Decreases in third-party payor reimbursement or a decision by a third-party payor to not cover our products or any other future approved products could reduce physician usage of our products, and have a material adverse effect on our sales, results of operations and financial condition.
In the United States, the Medicare Part D program provides a voluntary outpatient drug benefit to Medicare beneficiaries for certain products. Our products are available for coverage under Medicare Part D, but the individual Part D plans offer coverage subject to various factors such as those described above. In addition, while Medicare Part D plans have historically included “all or substantially all” drugs in the following designated classes of “clinical concern” on their formularies: anticonvulsants, antidepressants, antineoplastics, antipsychotics, antiretrovirals, and immunosuppressants, the Centers for Medicare & Medicaid Services (CMS), has in the past proposed, but not adopted, changes to this policy. If this policy is changed in the future and if CMS no longer considers the antipsychotic class to be of “clinical concern,” Medicare Part D plans would have significantly more discretion to reduce the number of products covered in that class, including coverage of our products. Furthermore, private third-party payors often follow Medicare coverage policies and payment limitations in setting their own coverage policies.
Healthcare Laws and Regulations
We are subject to healthcare regulation and enforcement by the federal government and the states and foreign governments in which we conduct our business. The healthcare laws and regulations that may affect our ability to operate include the following:
•The federal Anti-Kickback Statute makes it illegal for any person or entity to knowingly and willfully, directly or indirectly, solicit, receive, offer, or pay any remuneration that is in exchange for or to induce the referral of business, including the purchase, order, lease of any good, facility, item or service for which payment may be made under a federal healthcare program, such as Medicare or Medicaid. The term “remuneration” has been broadly interpreted to include anything of value.
•Federal false claims and false statement laws, including the federal civil False Claims Act, and civil monetary penalties laws, prohibit, among other things, any person or entity from knowingly presenting, or causing to be presented, for payment to, or approval by, federal programs, including Medicare and Medicaid, claims for items or services, including drugs, that are false or fraudulent.
•The U.S. federal Health Insurance Portability and Accountability Act of 1996 (HIPAA), created additional federal criminal statutes that prohibit among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors or making any false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services.
•HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (HITECH), and their implementing regulations, imposes obligations on covered entities, including certain healthcare providers, health plans, and healthcare clearinghouses, and their respective business associates that create, receive, maintain or transmit individually identifiable health information for or on behalf of a covered entity as well as their covered subcontractors, with respect to safeguarding the privacy, security and transmission of individually identifiable health information. In addition, the European Union (EU) and United Kingdom (UK) have each established their own data security and privacy legal framework, including but not limited to the EU’s General Data Protection Regulation (EU) 2016/79 and the so-called “UK GDPR” (together, the GDPR), which contain provisions specifically directed at the processing of health information, higher sanctions than previously applicable data protection laws and extra-territoriality measures intended to bring non-EU/-UK companies’ processing operations under the scope of these regulations in certain circumstances (including where the relevant processing relates to the monitoring of behaviors of individuals in the EU/UK – such as in the context of the conduct of a clinical trial). We currently conduct clinical trials in the EU and the UK and will need to be compliant with these requirements. We anticipate that over time we may expand our business operations to include additional operations in the EU and/or UK. With such expansion, we could be subject to increased governmental regulation in the territories in which we might operate.
•The federal Physician Payments Sunshine Act requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to CMS information related to payments or other transfers of value made to physicians (as defined to include doctors of medicine, dentists, optometrists, podiatrists and chiropractors by such law), other healthcare professionals (such as physician assistants and nurse practitioners), and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members.
Also, many U.S. states have similar laws and regulations, such as anti-kickback and false claims laws that may be broader in scope and may apply regardless of payor, in addition to items and services reimbursed under Medicaid and other state programs. Additionally, we may be subject to state laws that require pharmaceutical companies to comply with the federal government’s and/or pharmaceutical industry’s voluntary compliance guidelines, state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures, state laws that require drug manufacturers to report information on the pricing of certain drugs, state and local laws that require the registration of pharmaceutical sales representatives, as well as state and foreign laws governing the privacy and security of health information, many of which differ from each other in significant ways and often are not preempted by HIPAA.
If we are found to be in violation of any of these laws or any other federal or state regulations, we may be subject to significant administrative, civil and/or criminal penalties, damages, fines, disgorgement, imprisonment, exclusion from federal health care programs, additional reporting requirements and/or oversight, and the curtailment or restructuring of our operations.
Additionally, to the extent that our product is sold in a foreign country, we may be subject to similar foreign laws.
Healthcare Reform
The United States and some foreign jurisdictions are considering or have enacted a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sell our products profitably. By way of example, in March 2010, Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act (collectively, the ACA) was signed into law, which intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add transparency requirements for the healthcare and health insurance industries, impose taxes and fees on the health industry and impose additional health policy reforms.
There have been amendments and executive, judicial and Congressional challenges to certain aspects of the ACA. For example, on August 16, 2022, the Inflation Reduction Act of 2022 (IRA) was signed into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost and creating a new manufacturer discount program. It is possible that the ACA will be subject to additional challenges in the future. It is unclear how such challenges and the healthcare reform measures of the current administration will impact the ACA.
Other legislative changes have been proposed and adopted in the United States since the ACA. Through the process created by the Budget Control Act of 2011, there are automatic reductions of Medicare payments to providers up to 2% per fiscal year, which went into effect in April 2013 and, due to subsequent legislative amendments, including the Infrastructure Investment and Jobs Act, will remain in effect through 2032 unless additional Congressional action is taken. Additionally, on March 11, 2021, the American Rescue Plan Act of 2021 was signed into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug’s average manufacturer price, for single source and innovator multiple source drugs, beginning January 1, 2024.
Moreover, recently there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their commercial products. There have been several recent U.S. Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, reduce the cost of drugs under Medicare, and reform government program reimbursement methodologies for drugs. For example, the IRA, among other things, (1) directs the U.S. Department of Health and Human Services (HHS) to negotiate the price of certain single-source drugs that have been on the market for at least 7 years covered under Medicare (the Medicare Drug Price Negotiation Program) and (2) imposes rebates under Medicare Part B and Medicare Part D to penalize price increases that outpace inflation. These provisions began to take effect progressively in fiscal year 2023. On August 15, 2024, HHS announced the agreed-upon price of the first ten drugs that were subject to price negotiations, although the Medicare Drug Price Negotiation Program is currently subject to legal challenges. HHS will select up to fifteen additional drugs covered under Part D for negotiation in 2025. Each year thereafter more Part B and Part D products will become subject to the Medicare Drug Price Negotiation Program. On December 7, 2023, an initiative to control the price of prescription drugs through the use of march-in rights under the Bayh-Dole Act was announced. On December 8, 2023, the National Institute of Standards and Technology published for comment a Draft Interagency Guidance Framework for Considering the Exercise of March-In Rights which for the first time includes the price of a product as one factor an agency can use when deciding to exercise march-in rights. While march-in rights have not previously been exercised, it is uncertain if that will continue under the new framework.
At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. For example, on January 5, 2024, the FDA approved Florida’s proposal to import certain drugs from Canada for specific state healthcare programs. It is unclear if and how this program will be implemented and whether it will be subject challenges in the United States or Canada. Other states have also submitted proposals that are pending review by the FDA. Any such approved importation plans, if implemented, may result in lower drug prices for products covered by those programs.
We expect that healthcare reform measures that may be adopted in the future may result in more rigorous coverage criteria and lower reimbursement, and additional downward pressure on the price that we receive for our products and any future approved products. We cannot predict what healthcare reform initiatives may be adopted in the future.
Europe / Rest of World Government Regulation
Outside of the United States, our ability to market a product is contingent upon receiving a marketing authorization from the appropriate regulatory authorities. The requirements governing the conduct of clinical trials, marketing authorization, pricing and reimbursement vary widely from country to country. If the regulatory authority is satisfied that adequate evidence of safety, quality and efficacy has been presented, marketing authorization will be granted. This foreign regulatory approval process involves all of the risks associated with FDA marketing approval discussed above. In addition, foreign regulations may include applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws, and implementation of corporate compliance programs and reporting of payments or other transfers of value to healthcare professionals and entities.
For other countries outside of the EU, such as countries in Eastern Europe, Latin America or Asia, the requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country. In all cases, the clinical trials must be conducted in accordance with GCPs and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
If we or our potential collaborators fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension, variation or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Clinical Trials in the EU
Similarly to the United States, the various phases of non-clinical and clinical research in the EU are subject to significant regulatory controls. In the EU, clinical trials are governed by the Clinical Trials Regulation (EU) No 536/2014 (CTR), which entered into application on January 31, 2022 repealing and replacing the former Clinical Trials Directive 2001/20 (CTD).
The CTR is intended to harmonize and streamline clinical trial authorizations, simplify adverse-event reporting procedures, improve the supervision of clinical trials and increase transparency. Specifically, the CTR, which is directly applicable in all EU Member States, introduces a streamlined application procedure through a single-entry point, the “EU portal”, the Clinical Trials Information System (CTIS); a single set of documents to be prepared and submitted for the application; as well as simplified reporting procedures for clinical trial sponsors. A harmonized procedure for the assessment of applications for clinical trials has been introduced and is divided into two parts. Part I assessment is led by the competent authorities of a reference EU Member State selected by the trial sponsor and relates to clinical trial aspects that are considered to be scientifically harmonized across EU Member States. This assessment is then submitted to the competent authorities of all concerned EU Member States in which the trial is to be conducted for their review. Part II is assessed separately by the competent authorities and Ethics Committees in each concerned EU Member State. Individual EU Member States retain the power to authorize the conduct of clinical trials on their territory.
The CTR foresaw a three-year transition period that ended on January 31, 2025. Since this date, all new or ongoing trials are subject to the provisions of the CTR.
In all cases, clinical trials must be conducted in accordance with GCPs and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki. Medicines used in clinical trials, including advanced therapy medicinal products, must be manufactured in accordance with the guidelines on cGMP and in a GMP-licensed facility, which can be subject to GMP inspections.
EU Review and Approval Process of Medicinal Products
In the EU, medicinal products can only be commercialized after a related marketing authorization (MA) has been granted. To obtain an MA for a product in the EU, an applicant must submit a MAA either under a centralized procedure administered by the EMA, or one of the procedures administered by the competent authorities of EU Member States (decentralized procedure, national procedure or mutual recognition procedure). An MA may be granted only to an applicant established in the EU.
The centralized procedure provides for the grant of a single MA by the European Commission that is valid throughout the EEA (which is comprised of the 27 EU Member States plus Norway, Iceland and Liechtenstein). Pursuant to Regulation (EC) No 726/2004, the centralized procedure is compulsory for specific products, including for (i) medicinal products derived from biotechnological processes, (ii) products designated as orphan medicinal products, (iii) advanced therapy medicinal products and (iv) products with a new active substance indicated for the treatment of HIV/AIDS, cancer, neurodegenerative diseases, diabetes, auto-immune and other immune dysfunctions and viral diseases. For products with a new active substance indicated for the treatment of other diseases and products that are highly innovative or for which a centralized process is in the interest of patients, authorization through the centralized procedure is optional on related approval.
Under the centralized procedure, the EMA’s Committee for Medicinal Products for Human Use (CHMP) conducts the initial assessment of a product. The CHMP is also responsible for several post-authorization and maintenance activities, such as the assessment of modifications or extensions to an existing MA. The maximum timeframe for the evaluation of an MAA under the centralized procedure is 210 days, excluding clock stops when additional information or written or oral explanation
is to be provided by the applicant in response to questions of the CHMP. Accelerated assessment may be granted by the CHMP in exceptional cases, when a medicinal product targeting an unmet medical need is expected to be of major interest from the point of view of public health and, in particular, from the viewpoint of therapeutic innovation. If the CHMP accepts a request for accelerated assessment, the time limit of 210 days will be reduced to 150 days (excluding clock stops). The CHMP can, however, revert to the standard time limit for the centralized procedure if it considers that it is no longer appropriate to conduct an accelerated assessment.
Unlike the centralized authorization procedure, the decentralized MA procedure requires a separate application to, and leads to separate approval by, the competent authorities of each EU Member State in which the product is to be marketed. This application is identical to the application that would be submitted to the EMA for authorization through the centralized procedure. The reference EU Member State prepares a draft assessment and drafts of the related materials within 120 days after receipt of a valid application. The resulting assessment report is submitted to the concerned EU Member States who, within 90 days of receipt, must decide whether to approve the assessment report and related materials. If a concerned EU Member State cannot approve the assessment report and related materials due to concerns relating to a potential serious risk to public health, disputed elements may be referred to the Heads of Medicines Agencies’ Coordination Group for Mutual Recognition and Decentralised Procedures—Human (CMDh) for review. The subsequent decision of the European Commission is binding on all EU Member States.
The mutual recognition procedure allows companies that have a medicinal product already authorized in one EU Member State to apply for this authorization to be recognized by the competent authorities in other EU Member States. Like the decentralized procedure, the mutual recognition procedure is based on the acceptance by the competent authorities of the EU Member States of the MA of a medicinal product by the competent authorities of other EU Member States. The holder of a national MA may submit an application to the competent authority of an EU Member State requesting that this authority recognize the MA delivered by the competent authority of another EU Member State.
An MA has, in principle, an initial validity of five years. The MA may be renewed after five years on the basis of a re-evaluation of the risk-benefit balance by the EMA or by the competent authority of the EU Member State in which the original MA was granted. To support the application, the MA holder must provide the EMA or the competent authority with a consolidated version of the Common Technical Document providing up-to-date data concerning the quality, safety and efficacy of the product, including all variations introduced since the MA was granted, at least nine months before the MA ceases to be valid. The European Commission or the competent authorities of the EU Member States may decide on justified grounds relating to pharmacovigilance, to proceed with one further five-year renewal period for the MA. Once subsequently definitively renewed, the MA shall be valid for an unlimited period. Any authorization which is not followed by the actual placing of the medicinal product on the EU market (for a centralized MA) or on the market of the authorizing EU Member State within three years after authorization ceases to be valid (the so-called sunset clause).
Innovative products that target an unmet medical need and are expected to be of major public health interest may be eligible for a number of expedited development and review programs, such as the Priority Medicines (PRIME) scheme, which provides incentives similar to the breakthrough therapy designation in the U.S. PRIME is a voluntary scheme aimed at enhancing the EMA’s support for the development of medicinal products that target unmet medical needs. Eligible products must target conditions for which there is an unmet medical need (there is no satisfactory method of diagnosis, prevention or treatment in the EU or, if there is, the new medicinal product will bring a major therapeutic advantage) and they must demonstrate the potential to address the unmet medical need by introducing new methods of therapy or improving existing ones. Benefits accrue to sponsors of product candidates with PRIME designation, including but not limited to, early and proactive regulatory dialogue with the EMA, frequent discussions on clinical trial designs and other development program elements, and potentially accelerated MAA assessment once a dossier has been submitted.
In the EU, a “conditional” MA may be granted in cases where all the required safety and efficacy data are not yet available. The European Commission may grant a conditional MA for a medicinal product if it is demonstrated that all of the following criteria are met: (i) the benefit-risk balance of the medicinal product is positive; (ii) it is likely that the applicant will be able to provide comprehensive data post-authorization; (iii) the medicinal product fulfils an unmet medical need; and (iv) the benefit of the immediate availability to patients of the medicinal product is greater than the risk inherent in the fact that additional data are still required. The conditional MA is subject to conditions to be fulfilled for generating the missing data or ensuring increased safety measures. It is valid for one year and must be renewed annually until all related conditions have been fulfilled. Once any pending studies are provided, the conditional MA can be converted into a traditional MA. However, if the conditions are not fulfilled within the timeframe set by the EMA and approved by the European Commission, the MA will cease to be renewed.
An MA may also be granted “under exceptional circumstances” where the applicant can show that it is unable to provide comprehensive data on efficacy and safety under normal conditions of use even after the product has been authorized and subject to specific procedures being introduced. These circumstances may arise in particular when the intended indications are very rare and, in the state of scientific knowledge at that time, it is not possible to provide comprehensive information, or when generating data may be contrary to generally accepted ethical principles. Like a conditional MA, an MA granted in exceptional circumstances is reserved to medicinal products intended to be authorized for treatment of rare diseases or unmet medical needs for which the applicant does not hold a complete data set that is required for the grant of a standard MA. However, unlike the conditional MA, an applicant for authorization in exceptional circumstances is not subsequently required to provide the missing data. Although the MA “under exceptional circumstances” is granted definitively, the risk-benefit balance of the medicinal product is reviewed annually, and the MA will be withdrawn if the risk-benefit ratio is no longer favorable.
Pediatric Development in the EU
In the EU, Regulation (EC) No 1901/2006 provides that all MAAs for new medicinal products have to include the results of trials conducted in the pediatric population, in compliance with a pediatric investigation plan (PIP) agreed with the EMA’s Pediatric Committee (PDCO). The PIP sets out the timing and measures proposed to generate data to support a pediatric indication of the medicinal product for which MA is being sought. The PDCO can grant a deferral of the obligation to implement some or all of the measures provided in the PIP until there are sufficient data to demonstrate the efficacy and safety of the product in adults. Further, the obligation to provide pediatric clinical trial data can be waived by the PDCO when these data are not needed or appropriate because the product is likely to be ineffective or unsafe in children, the disease or condition for which the product is intended occurs only in adult populations, or when the product does not represent a significant therapeutic benefit over existing treatments for pediatric patients. Once the MA is obtained in all EU Member States and study results are included in the product information, even when negative, the product is eligible for a six-month extension to the Supplementary Protection Certificate, if any is in effect at the time of authorization or, in the case of orphan medicinal products, a two-year extension of orphan market exclusivity.
Data and Market Exclusivity in the EU
The EU provides opportunities for data and market exclusivity related to MAs. Upon receiving an MA, innovative medicinal products are generally entitled to receive eight years of data exclusivity and ten years of market exclusivity. Data exclusivity, if granted, prevents regulatory authorities in the EU from referencing the innovator’s data to assess a generic application or biosimilar application for eight years from the date of authorization of the innovative product, after which a generic or biosimilar MAA can be submitted, and the innovator’s data may be referenced. The market exclusivity period prevents a successful generic or biosimilar applicant from commercializing its product in the EU until 10 years have elapsed from the initial MA of the reference product in the EU. The overall ten-year period may, occasionally, be extended for a further year to a maximum of 11 years if, during the first eight years of those ten years, the MA holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies. However, there is no guarantee that a product will be considered by the EU’s regulatory authorities to be a new chemical/biological entity, and products may not qualify for data exclusivity.
In the EU, there is a special regime for biosimilars, or biological medicinal products that are similar to a reference medicinal product but that do not meet the definition of a generic medicinal product. For such products, the results of appropriate preclinical or clinical trials must be provided in support of an application for MA. Guidelines from the EMA detail the type of quantity of supplementary data to be provided for different types of biological product.
Pricing, Coverage and Reimbursement
In the EU, pricing and reimbursement schemes vary widely from country to country. Some EU Member States may approve a specific price for a product, or they may instead adopt a system of direct or indirect controls on the profitability of the company placing the product on the market. Other EU Member States allow companies to fix their own prices for products but monitor and control prescription volumes and issue guidance to physicians to limit prescriptions.
Moreover, in order to obtain reimbursement for our products in some European countries, including some EU Member States, we may be required to compile additional data comparing the cost-effectiveness of our products to other available therapies. This Health Technology Assessment (HTA) of medicinal products is becoming an increasingly common part of the pricing and reimbursement procedures in some EU Member States, including those representing the larger markets. The HTA
process is the procedure to assess therapeutic, economic and societal impact of a given medicinal product in the national healthcare systems of the individual country. The outcome of an HTA will often influence the pricing and reimbursement status granted to these medicinal products by the competent authorities of individual EU Member States. The extent to which pricing and reimbursement decisions are influenced by the HTA of the specific medicinal product currently varies between EU Member States. In December 2021, Regulation No 2021/2282 on HTA, was adopted in the EU. This regulation, which entered into application on January 12, 2025 and has a phased implementation, is intended to boost cooperation among EU Member States in assessing health technologies, including new medicinal products, and providing the basis for cooperation at EU level for joint clinical assessments in these areas. This regulation permits EU Member States to use common HTA tools, methodologies, and procedures across the EU, working together in four main areas, including joint clinical assessment of the innovative health technologies with the most potential impact for patients, joint scientific consultations whereby developers can seek advice from HTA authorities, identification of emerging health technologies to identify promising technologies early, and continuing voluntary cooperation in other areas. Individual EU Member States continue to be responsible for assessing non-clinical (e.g., economic, social, ethical) aspects of health technologies, and making decisions on pricing and reimbursement.
Marketing
Much like the Anti-Kickback Statute prohibition in the United States, as described below, the provision of benefits or advantages to physicians and other health care professionals to induce or encourage the prescription, recommendation, endorsement, purchase, supply, order or use of medicinal products is also prohibited in the EU. Interactions between pharmaceutical companies and health care professionals are governed by strict laws, such as national anti-bribery laws of European countries, national sunshine rules, regulations, industry self-regulation codes of conduct and physicians’ codes of professional conduct. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment. Infringement of related laws could result in substantial fines and imprisonment.
Payments made to physicians and other health care professionals in certain EU Member States must be publicly disclosed. Moreover, agreements with health care professionals may require prior notification or approval by the health care professional’s employer, his or her competent professional organization and/or the regulatory authorities of the individual EU Member States. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment.
Manufacturing and Distribution
We currently outsource, and plan to continue to outsource, manufacturing activities for our products and our existing and future product candidates for development and commercial purposes. We believe this manufacturing strategy will enable us to direct our financial resources to our commercial activities and to the ongoing development of pimavanserin, trofinetide and other product candidates without devoting the substantial resources and capital required to build manufacturing facilities.
We licensed worldwide intellectual property rights related to pimavanserin in certain indications to Acadia Pharmaceuticals GmbH, our wholly-owned Swiss subsidiary (Acadia GmbH). Our active pharmaceutical ingredient (API) has been manufactured in Switzerland for over 10 years and we anticipate continuing to manufacture in Switzerland. Acadia GmbH manages the worldwide supply chain of our pimavanserin API, and maintains sufficient inventory in our Switzerland contract warehouse.
Acadia GmbH has contracted with Siegfried AG (Siegfried), to manufacture the pimavanserin API for use in NUPLAZID drug product for commercial sale. Under the manufacturing agreement, Acadia GmbH has agreed to purchase specified percentages of our commercial requirements of the pimavanserin API at a predefined price. The parties may also agree in the future on additional services under the manufacturing agreement with respect to non-commercial supply or development activities. The initial term of the manufacturing agreement ended in December 2021, but the agreement automatically renewed twice, each time for a two-year term, and it will automatically renew for subsequent two-year terms unless either party provides timely notice of its intent not to renew, or unless the manufacturing agreement is terminated earlier pursuant to its terms. Either party may terminate the manufacturing agreement prior to expiration upon an uncured material breach by the other party, upon the dissolution or liquidation of the other party, the commencement of insolvency procedures that are not dismissed within a certain period of time, the appointment of any receiver, trustee or assignee to take possession of the properties of the other party or the cessation of all or substantially all of the other party's business operations, upon certain continuing patent infringement, regulatory litigation or other legal proceedings involving the manufacture of our API, upon a continuing force majeure affecting the other party, or if no services are currently being
provided under the manufacturing agreement. Additionally, if the parties agree on development services under the manufacturing agreement, the parties may terminate such services by mutual agreement if reasonable efforts to achieve the goals of such services fail. Acadia GmbH also may terminate any services under the manufacturing agreement for any reason on 90 days’ prior notice to Siegfried, subject to the requirements of the manufacturing agreement.
We have contracted with Patheon Pharmaceuticals Inc. (Patheon), a subsidiary of Thermo Fisher Scientific Inc., to manufacture NUPLAZID 10 mg tablet and 34 mg capsule drug product for commercial use in the United States. We have also contracted with a second contract manufacturing organization to manufacture NUPLAZID 34 mg drug product for commercial use in the United States. Under the manufacturing agreement with Patheon, we have agreed to purchase from Patheon a specified percentage of our commercial requirements of NUPLAZID for the United States. Under the agreement, Patheon will also perform specified validation services. The initial term of the manufacturing agreement ended in the first quarter of 2023, but the agreement automatically renewed for a two-year term and will automatically renew for subsequent two-year terms unless either party provides timely notice of its intent not to renew, or unless the manufacturing agreement is terminated early pursuant to its terms. Each party may terminate the manufacturing agreement prior to expiration upon the uncured material breach by the other party, upon the bankruptcy or insolvency of the other party or in the event of a continuing force majeure event affecting the other party. The manufacturing agreement will also terminate if we provide notice to Patheon that we no longer require manufacturing services because NUPLAZID has been discontinued. Additionally, we may terminate the manufacturing agreement, subject to certain limitations, if any regulatory authority takes any action or raises any objection that prevents us from continuing to commercialize NUPLAZID or takes an enforcement action against Patheon’s manufacturing site that relates to NUPLAZID or could reasonably be expected to adversely affect Patheon’s ability to supply NUPLAZID, if we determine to discontinue commercialization of NUPLAZID for safety or efficacy reasons, or if Patheon uses any debarred person in performing its service obligations under the manufacturing agreement. We also may terminate the manufacturing agreement for any other reason on three years’ prior notice to Patheon. Patheon may terminate the manufacturing agreement if we assign the manufacturing agreement or any of our rights under the manufacturing agreement to a Patheon competitor.
We sell NUPLAZID to a limited number of specialty pharmacies (SPs), and specialty distributors (SDs), which we collectively refer to as our customers. SPs subsequently dispense NUPLAZID to patients based on the fulfillment of a prescription and SDs subsequently sell NUPLAZID to government facilities, long-term care pharmacies, and in-patient hospital pharmacies. Four of such customers, each based in the United States, accounted for approximately 73% of our NUPLAZID product revenue and 48% of our total product revenue for the year ended December 31, 2024. We have retained third-party logistics service providers to perform a variety of functions related to the distribution of NUPLAZID, including warehousing, customer service, order-taking, invoicing, collections, and shipment and returns processing.
We have contracted with manufacturers to produce supplies of trofinetide to support the development program and for commercial sale. We have contracted with Corden Pharma Bergamo S.p.A. (Corden), to manufacture the API for trofinetide products. Under the manufacturing agreement with Corden, we have agreed to purchase from Corden the API for trofinetide products at specified price per volume and a specified percentage of our commercial requirements of trofinetide API for the United States and Canada market. We and Corden may also agree in the future on additional services under the manufacturing agreement. The initial term of the manufacturing agreement will end in November 2027, but the agreement will automatically renew for subsequent two-year terms unless either party provides timely notice of its intent not to renew, or unless the manufacturing agreement is terminated early pursuant to its terms. Either party may terminate the manufacturing agreement prior to expiration upon an uncured material breach by the other party, upon the commencement of bankruptcy, reorganization, liquidation or receivership proceedings by or against the other party or the other party ceases for any reason to carry on its business or makes assignment for the benefit of its creditors, or is the subject of any proposal for a voluntary arrangement. Additionally, either party may terminate the manufacturing agreement on 12 months’ prior notice to the other party at any time.
We also have contracted with F.I.S. Fabbrica Italiana Sintetici S.p.A. (FIS) to manufacture the API for trofinetide products. Under the manufacturing agreement, we have the right to purchase from FIS the API for the trofinetide products at a specified price per volume. The parties may also agree in the future on additional services under the manufacturing agreement with respect to commercial testing and other manufacturing services. The initial term of the manufacturing agreement ended in December 2024, but the agreement automatically renewed for a two-year term and will automatically renew for subsequent two-year terms unless either party provides timely notice of its intent not to renew, or unless the manufacturing agreement is terminated earlier pursuant to its terms. Either party may terminate the manufacturing agreement prior to expiration upon an uncured material breach by the other party, upon the commencement of bankruptcy, reorganization, liquidation or receivership proceedings by or against the other party or the other party ceases for any reason to carry on its business or makes assignment for the benefit of its creditors, or is the subject of any proposal for a voluntary
arrangement. Additionally, either party may terminate the manufacturing agreement on 30 days’ prior notice to the other party at any time no services are being rendered under the manufacturing agreement. We also may terminate any services under the manufacturing agreement for any reason on 90 days’ prior notice to FIS, subject to the requirements of the manufacturing agreement.
Under the manufacturing agreement with Patheon described above, we also have the right to purchase trofinetide products for commercial use. In addition, we have contracted with CoreRx Inc. (CoreRx) to manufacture trofinetide products for commercial use. We and CoreRx may also agree in the future on additional services under the agreement. The initial term of the agreement will end in March 2028, but the agreement will automatically renew for subsequent two-year terms unless either party provides timely notice of its intent not to renew, or unless the agreement is terminated early pursuant to its terms. Either party may terminate the agreement prior to expiration upon an uncured material breach by the other party or upon the commencement of bankruptcy, reorganization, liquidation or receivership proceedings by or against the other party. In addition, we may terminate the agreement prior to expiration upon timely notice to CoreRx in event (i) any regulatory authority takes an enforcement or other regulatory action against CoreRx’s facility which affects CoreRx’s ability to process trofinetide products, (ii) any regulatory authority takes an action or raises any objection that prevents us from manufacturing, importing, exporting, purchasing or selling trofinetide products, or (iii) we determine to discontinue commercialization of trofinetide products in the U.S. due to safety or efficacy reasons.
We sell DAYBUE to a single wholesale distributor with specialty pharmacy service, which performs a variety of functions related to the distribution of DAYBUE, including warehousing, customer service, order-taking, shipment and returns processing.
If any product candidate is approved by the FDA or comparable foreign regulatory authorities for commercial sale, we will need to contract with a third party, which may be an existing service provider or a new service provider, to manufacture such product for commercial sale in the U.S. and/or other applicable jurisdictions.
Sales and Marketing
We have U.S. sales specialists that are focused on promoting NUPLAZID to physicians who treat PDP patients, including neurologists, psychiatrists and long-term care physicians. This sales force is supported by an experienced sales leadership team. Our experienced commercial team is comprised of experienced professionals in marketing, key account management, patient access services, commercial operations, and sales force planning and management. In addition, our commercial infrastructure includes capabilities in manufacturing, health outcomes, medical affairs, quality control, and compliance.
We launched NUPLAZID in May 2016, and our focus is to continue to establish NUPLAZID as the standard of care for patients with PDP. In order to help us achieve this goal, we are continuing to increase awareness of NUPLAZID’s benefits in PDP with a prescriber and patient education campaign consisting of key opinion leader speaker programs, attendance at medical meetings, digital outreach, multimedia campaigns, and direct-to-patient programs.
In addition, we have U.S. sales specialists that are focused on promoting DAYBUE to physicians who treat Rett syndrome patients, including those at Centers of Excellence, high volume institutions and in the community setting. The sales force is supported by an experienced sales leadership team. Our experienced commercial team is comprised of rare disease field-based specialists, patient access services, commercial operations, and sales force planning and management. In addition, our commercial infrastructure includes capabilities in manufacturing, health outcomes, medical affairs, quality control, and compliance.
We launched DAYBUE in April 2023, and our focus is to continue to establish DAYBUE as the standard of care for patients with Rett syndrome. In order to help us achieve this goal, we are continuing to increase awareness of DAYBUE’s benefits in Rett syndrome with a prescriber and patient education campaign consisting of key opinion leader speaker programs, attendance at medical meetings, digital outreach, and multimedia campaigns.
We also have support services including the Acadia Connect hub for physicians, patients and their families that provide broad resources to help with access, reimbursement and the continual clinical support to help patients start and stay on therapy. For healthcare providers and practices, Acadia Connect provides access and coverage support services, information on appropriate financial assistance options for eligible patients, and coordination of medication delivery to patients through our specialty pharmacy.
In selected markets outside of the United States in which DAYBUE may be approved, if any, we may choose to commercialize DAYBUE independently or by establishing one or more strategic alliances.
Long-Lived Assets
Our tangible long-lived assets are comprised of intangible assets and property and equipment. Our property and equipment totaled $4.2 million, $4.6 million, and $6.0 million as of December 31, 2024, 2023 and 2022, respectively. All of our tangible long-lived assets are located in the United States. Our intangible assets, comprised of right-of-use assets and other intangibles acquired, totaled $166.4 million, $117.3 million and $55.6 million as of December 31, 2024, 2023 and 2022, respectively.
Employees and Human Capital
Employees. At December 31, 2024, we had a total of 654 employees, 653 of whom were full-time. We employ physicians, scientists and professionals in research and development, regulatory, manufacturing, marketing, sales, finance, legal and other functions that are important to our business. We also will continue to use temporary workers in certain instances in order to maximize our employment flexibility in light of our business needs. Additionally, when we think it is in the best interest of our business, we will rely upon external advisers and consultants rather than our employees.
Employee Engagement, Benefits & Development. We believe that our future success is dependent upon our ability to recruit, hire and retain exceptional employees. We provide our employees with competitive cash compensation, opportunities to own equity, and an employee benefit program that promotes well-being, including wellness programs, healthcare, retirement planning and paid time off. We also provide employees with opportunities to continue their education and growth, including leadership development and tuition reimbursement. In order to receive feedback from our employees and evaluate our level of employee engagement, we regularly conduct employee surveys.
Diversity, Equity & Inclusion. We value diversity, equity, and inclusion across our workforce, in our communities, and in the work that we do. We will continue to focus on diversity, equity, and inclusion initiatives that support a culture that is centered on belonging while aligning with our shared corporate mission and values.
Item 1A. Risk Factors.
You should consider carefully the following information about the risks described below, together with the other information contained in this Annual Report and in our other public filings, in evaluating our business. If any of the following risks actually occurs, our business, financial condition, results of operations, and future growth prospects would likely be materially and adversely affected. In these circumstances, the market price of our common stock would likely decline.
Risks Related to Our Products and Product Candidates
Our prospects are highly dependent on the successful commercialization of our products. To the extent we cannot maintain or increase sales of our products, our business, financial condition and results of operations may be materially adversely affected and the price of our common stock may decline.
We have two products that are approved for commercialization in the U.S.: NUPLAZID and DAYBUE. The successful commercialization of such products is subject to many risks, and there is no guarantee that we will be able to maintain or increase sales of such products. Our business, financial condition and results of operations may be materially adversely affected and the price of our common stock may decline because of many factors, some of which are outside our control, including, but not limited to, the following:
•the extent to which patients, caregivers and physicians recognize and diagnose the indications for which our products are approved and accept and adopt our products as a treatment for such indications;
•the scope and terms of the FDA’s approval of our products, including the inclusion of a boxed warning for NUPLAZID or other warnings and precautions for our products;
•physicians may not prescribe our products and patients may be unwilling to use our products, due to a number of factors, including if coverage is not provided, coverage changes in the future, reimbursement is inadequate to cover a significant portion of the cost, negative or changing perceptions of each product’s clinical profile and clinical benefits or due to the prevalence and severity of any adverse side effects;
•the experiences of those adopting our products earlier could have significant impact on future adoption of our products by other physicians, patients and caregivers, either favorably or unfavorably, based on clinical benefits and side effects experienced;
•any new clinical data, post-approval studies or real world results, including in jurisdictions other than the U.S., could result in the FDA making changes to the product label or withdrawal from the market, and could impact regulatory approvals for other indications in the U.S. or other jurisdictions, if any, any of which could result in significant expense and delay or limit our ability to generate sales revenues;
•our products are becoming available to a larger number of patients and patients’ experiences and results with our products may not be consistent with, or may be more negative when compared to, the experiences and results of those treated in our clinical trials;
•successful expansion and development of our commercial team and sales forces; and
•any negative publicity related to our products.
Additionally, our success is dependent on our ability to obtain regulatory approval for, and successfully commercialize, trofinetide in jurisdictions outside the U.S., including the EU. We will face in jurisdictions outside the U.S., such as the EU, if approved for marketing, risks and uncertainties similar to the risks and uncertainties faced in the U.S. with respect to commercialization outside of the U.S., including, but not limited to, government reimbursement of the cost of trofinetide. If the commercialization of our products and future sales is less successful than expected or perceived as disappointing, our stock price could decline significantly and the long-term success of our products and our company could be harmed.
Our products may not gain maximal acceptance among physicians, patients, caregivers and the medical community, thereby limiting our potential to generate revenues.
The degree of market acceptance by physicians, healthcare professionals, patients, caregivers and third-party payors of our products, and our profitability and growth, will depend on a number of factors, including:
•the ability to provide acceptable evidence of safety and efficacy;
•the scope of the approved indication(s) for the product;
•the inclusion of any warnings or contraindications in the product label;
•the relative convenience and ease of administration;
•the relative timing, or perceived timing, in which patients experience outcomes;
•the prevalence and severity of any actual or expected adverse side effects;
•the availability of alternative treatments;
•the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies;
•our ability to increase awareness of our approved products through marketing efforts;
•pricing and cost effectiveness, which may be subject to regulatory control;
•effectiveness of our or our collaborators’ sales and marketing strategy;
•publicity concerning us, our products or competing products and treatments; and
•our ability to obtain and maintain sufficient third-party insurance coverage or adequate reimbursement levels.
If a product does not provide a treatment regimen that is at least as beneficial as the current standard of care or otherwise does not provide patient benefit, that product will not achieve market acceptance and will not generate sufficient revenues to achieve or maintain profitability. With respect to our products specifically, successful commercialization will depend on whether and to what extent physicians, patients, caregivers, long-term care facilities and pharmacies, over whom we have no control, determine to utilize our products. NUPLAZID is available in the U.S. to treat hallucinations and delusions associated with PDP, and DAYBUE is available in the U.S. to treat Rett syndrome, both indications for which no other FDA-approved pharmaceutical treatments currently exist. DAYBUE is also the first and only product approved in Canada for the treatment of Rett syndrome.
As there are no approved competitors for our products, it is particularly difficult to estimate the market potential for our products and how physicians, patients, caregivers, long-term care facilities and payors will respond to changes in the price of our products. Industry sources and analysts have a divergence of estimates for the near- and long-term market potential of our products, and a variety of assumptions directly impact the estimates for our products’ market potential, including assumptions regarding the prevalence of PDP and Rett syndrome, the rate of diagnosis of PDP and Rett syndrome, the prevalence and rate of hallucinations and delusions in patients diagnosed with PDP with respect to NUPLAZID, the rate of physician adoption, the potential impact of payor restrictions, and patient adherence and compliance rates. Small differences in these assumptions can lead to widely divergent estimates of the market potential of our products.
For example, with respect to NUPLAZID, certain research suggests that patients with Parkinson’s disease may be hesitant to report symptoms of PDP to their treating physicians for a variety of reasons, including apprehension about societal stigmas relating to mental illness. Research also suggests that physicians who typically treat patients with Parkinson’s disease may not ask about or identify symptoms of PDP. For these reasons, even if PDP occurs in high rates among patients with Parkinson’s disease, it may be underdiagnosed. Even if PDP is diagnosed, physicians may not prescribe treatment for hallucinations and delusions associated with PDP, and if they do prescribe treatment, they may prescribe drugs other than NUPLAZID, even though they are not approved in PDP. Further, NUPLAZID may take several weeks to show efficacy. Even if NUPLAZID is prescribed for the treatment of hallucinations and delusions associated with PDP, patients may stop taking NUPLAZID because they may not see results in the timeframe they desire or expect.
Similarly, even if DAYBUE is prescribed for the treatment of Rett syndrome, issues may arise with respect to patient acceptance, adherence, persistence and compliance rates for a variety of reasons, including due to the expected clinical benefits or expected and actual side effects a patient might incur. If patients do not adhere to the recommended dosing of DAYBUE, or do not maintain the recommended dosing of DAYBUE for sufficient periods of time, patients and physicians may believe that DAYBUE is less effective, and as a result they may discontinue taking it and prescribing it. Additionally, if physicians or patients titrate DAYBUE below the recommended doses, patients may not experience the desired outcomes, and physicians or patients may develop negative beliefs about the effectiveness of DAYBUE and/or discontinue its use.
The label for NUPLAZID also contains a “boxed” warning related to particularly important prescribing information, and the FDA reminded healthcare providers to be aware of the risks described in the NUPLAZID prescribing information following its observation of potentially concerning prescribing patterns. There has also been attention to publicly reported deaths of patients that were prescribed NUPLAZID, and the FDA conducted an evaluation of available information about NUPLAZID. Perceptions that NUPLAZID is unsafe, even if unfounded, may discourage physicians from prescribing or patients from taking NUPLAZID.
The commercial success of our products depends on acceptance by patients, caregivers and physicians, and there are a number of factors that could skew our or others’ estimates about prescribing behaviors and market adoption. If we fail to gain the acceptance of patients, caregivers and physicians, or if our estimates are inaccurate, these events could negatively impact our business, results of operations, financial condition and prospects.
If we do not obtain regulatory approval of trofinetide outside North America, we will not be able to market trofinetide outside North America, which will limit our commercial revenues.
DAYBUE was approved in 2023 in the U.S. by the FDA, and in October 2024 in Canada by Health Canada, for the treatment of Rett syndrome in adult and pediatric patients two years of age and older. In January 2025, we submitted a marketing authorization application for the approval of trofinetide for the treatment of Rett syndrome in the EU. If we do not receive marketing approval for trofinetide in the EU or other jurisdictions outside of North America, including Japan, we will never be able to commercialize trofinetide in such jurisdictions. Even if we do receive additional regulatory approvals, we may not be successful in commercializing those opportunities.
If the results or timing of regulatory filings, the regulatory process, regulatory developments, clinical trials or preclinical studies, or other activities, actions or decisions related to DAYBUE or trofinetide do not meet our or others’ expectations, the market price of our common stock could decline significantly and the long-term success of the product and our company could be harmed.
Our ability to generate product revenues will be diminished if coverage for our products from commercial or government payors is decreased or if patients have unacceptably high out-of-pocket requirements.
Patients who are prescribed medicine for the treatment of their conditions generally rely on third-party payors, including governmental healthcare programs, such as Medicare and Medicaid, managed care organizations and commercial payors, among others, to reimburse all or part of the costs associated with their prescription drugs. Coverage and adequate reimbursement from third-party payors are critical to product acceptance. Coverage decisions may depend upon clinical and economic standards that disfavor drug products when lower cost therapeutic alternatives are already available or subsequently become available. Even with coverage for our products, the resulting reimbursement payment rates might not be adequate or may require out-of-pocket obligations, such as deductibles and co-pay or coinsurance payments, that patients find unacceptably high. Patients may not use our products if coverage is not provided or reimbursement is inadequate to cover a significant portion of its cost.
In addition, the market for our products depends significantly on access to third-party payors’ drug formularies, or lists of medications for which third-party payors provide coverage and reimbursement. The industry competition to be included in such formularies often leads to downward pricing pressures on pharmaceutical companies. Also, third-party payors may refuse to include a particular branded drug in their formularies or otherwise restrict patient access to a branded drug when a less costly alternative is available, even if not approved for the indication for which our products are approved.
Legislators, policymakers and healthcare insurance funds in the EU and the United Kingdom may continue to propose and implement cost-containing measures to keep healthcare costs down, particularly due to the financial strain that the COVID-19 pandemic placed on national healthcare systems of European countries. These measures could include limitations on the prices we would be able to charge for product candidates that we may successfully develop and for which we may obtain regulatory approval or the level of reimbursement available for these products from governmental authorities or third-party payors. Consequently, a downward trend in prices of medicinal products in some countries could contribute to similar downward trends elsewhere.
Third-party payors, whether governmental or commercial, whether in the U.S. or globally, are developing increasingly sophisticated methods of controlling healthcare costs. The current environment is putting pressure on companies to price products below what they may feel is appropriate. Selling our products at less than an optimized price would impact our revenues and could impact our overall success as a company. We have changed, and may continue to change, the price of our products from time to time, however, we do not know if the price we have selected, or may select in the future, for our products is or will be the optimized price. Additionally, we do not know whether and to what extent third-party payors will react to any possible future changes in the price of our products. In the U.S., no uniform policy of coverage and reimbursement for drug products exists among third-party payors. Outside the U.S., reimbursement and healthcare payment systems vary significantly by country, and many countries have instituted price ceilings on specific products and therapies. Further, one payor’s determination to provide coverage and reimbursement for a product does not ensure that other payors will also provide coverage and reimbursement for the product. Therefore, coverage and reimbursement for our products both in the U.S. and outside may differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that coverage will be obtained. Coverage policies and third-party payor reimbursement rates may change at any time. Therefore, even if favorable coverage and reimbursement status is attained, less favorable coverage policies and reimbursement rates may be implemented in the future.
In most international markets, where the government is the primary payor, manufacturers must operate in an environment of government-directed cost-containment programs – designs such as price controls, international reference pricing, mandatory discounts and rebates, regulatory hurdles and restrictions on physician-level prescribing. In these markets, healthcare services and determination of a product’s pricing and reimbursement are impacted by government control. For example, the EU provides options for EU Member States to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. An EU Member State may approve a specific price for the medicinal product, it may refuse to reimburse a product at the price set by the manufacturer or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. Many EU Member States also periodically review their reimbursement procedures for medicinal products, which could have an adverse impact on reimbursement status. Moreover, in order to obtain reimbursement for our products in some European countries, including some EU Member States, we may be required to compile additional data comparing the cost-effectiveness of our products to other available therapies in a Health Technology Assessment (HTA).
An HTA of medicinal products is becoming an increasingly common part of the pricing and reimbursement procedures in some EU Member States, including those representing the larger markets. The HTA process is the procedure to assess therapeutic, economic and societal impact of a given medicinal product in the national healthcare systems of the individual country. The outcome of an HTA will often influence the pricing and reimbursement status granted to these medicinal products by the competent authorities of individual EU Member States. The extent to which pricing and reimbursement decisions are influenced by the HTA of the specific medicinal product currently varies between EU Member States. In December 2021, Regulation No 2021/2282 on HTA, was adopted in the EU. This regulation, which entered into application on January 12, 2025 and has a phased implementation, is intended to boost cooperation among EU Member States in assessing health technologies, including new medicinal products, and providing the basis for cooperation at EU level for joint clinical assessments in these areas. This regulation permits EU Member States to use common HTA tools, methodologies, and procedures across the EU, working together in four main areas, including joint clinical assessment of the innovative health technologies with the most potential impact for patients, joint scientific consultations whereby developers can seek advice from HTA authorities, identification of emerging health technologies to identify promising technologies early, and continuing voluntary cooperation in other areas. Individual EU Member States continue to be responsible for assessing non-clinical (e.g., economic, social, ethical) aspects of health technologies, and making decisions on pricing and reimbursement.
So, for present and future considerations, if we are unable to obtain coverage of, and adequate payment levels for, our products we may market to third-party payors, physicians may limit how much or under what circumstances they will prescribe or administer them and patients may decline to purchase them. This in turn could affect our ability to successfully commercialize our products or any other products we may market, and thereby adversely impact our profitability, results of operations, financial condition, and future success.
Our products are subject to ongoing regulatory requirements that could cause us significant expense and delay or limit our ability to generate sales revenues.
In connection with the FDA approval of DAYBUE, we agreed to the following post-marketing requirements (PMRs): a clinical study of renal impairment in healthy volunteers, nonclinical carcinogenicity studies and nonclinical in vitro and clinical in vivo drug interaction studies. The FDA has released us from one of the five PMRs. In addition, we have fulfilled one of the five PMRs. Of the remaining three PMRs, we have completed one and are awaiting the FDA’s acknowledgement and acceptance. The results of any post-marketing study may cause the FDA to update the label, request additional studies and/or require risk mitigation plans.
The manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for our products will also continue to be subject to extensive and ongoing regulatory requirements in the U.S., Canada and in other foreign countries in which we obtain marketing approvals. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with cGMPs, good clinical practices, international council for harmonization guidelines and good laboratory practices, each of which are regulations and guidelines enforced by regulatory authorities for all of our nonclinical and clinical development and for any clinical trials that we conduct post-approval.
Discovery of any issues post-approval, including any safety concerns, such as carcinogenicity, unexpected side effects or drug-drug interaction problems, adverse events of unanticipated severity or frequency, or concerns over misuse or abuse of the product, problems with the facilities where the product is manufactured, tested, packaged or distributed, or failure to comply with regulatory requirements, may result in, among other things, restrictions on our products or on us, including:
•withdrawal of approval, addition of warnings or narrowing of the approved indication in the product label;
•requirement of a Risk Evaluation and Mitigation Strategy to mitigate the risk of off-label use in populations where the FDA may believe that the potential risks of use may outweigh its benefits;
•voluntary or mandatory recalls;
•suspension of any ongoing clinical studies;
•refusal by the FDA or comparable foreign regulatory authorities to approve pending applications or supplements to approved applications filed by us, or suspension or revocation of product approvals;
•restrictions on operations, including restrictions on the marketing or manufacturing of the product or the imposition of costly new manufacturing requirements; or
•seizure or detention, or refusal to permit the import or export of products.
If any of these actions were to occur, we may have to discontinue the commercialization of the applicable product, limit our sales and marketing efforts, conduct further post-approval studies, and/or discontinue or change any other ongoing or planned clinical studies, which in turn could result in significant expense and delay or limit our ability to generate sales revenues.
We rely on a limited network of third-party distributors and pharmacies to market and sell our products. If this approach ceases to be effective, commercialization of our products may be adversely affected, and our products may not be profitable.
Our strategy includes distributing NUPLAZID in the U.S. and DAYBUE or trofinetide, as applicable, in the U.S., Canada and other jurisdictions in which marketing is approved solely through a limited network of third-party specialty distributors, specialty pharmacies or other third-party partners. While we have entered into agreements with each of these distributors and pharmacies to distribute NUPLAZID in the U.S. and DAYBUE in the U.S. and Canada, we will need to enter into similar agreements in any jurisdictions in which trofinetide is approved, and such distributors and pharmacies may not perform as agreed or they may terminate their agreements with us. Also, we may need to enter into agreements with additional distributors, pharmacies or other entities, and there is no guarantee that we will be able to do so on commercially reasonable terms or at all.
In the event we are unable to maintain and, if needed, expand, our network of third-party specialty distributors and specialty pharmacies, our ability to continue commercializing our products would be limited, and our products may not be profitable.
Drug development is a long, expensive and unpredictable process with a high risk of failure, and there is no guarantee that our products or product candidates will be successful in ongoing or future clinical trials or obtain regulatory approval.
Preclinical testing and clinical trials are long, expensive and unpredictable processes that can be subject to delays. Preliminary, initial, top-line or interim results of clinical trials do not necessarily predict final results and such results may change as more patient data becomes available and are subject to audit and verification procedures that could result in material changes in the final results. In addition, success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful. A number of companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in advanced clinical trials even after promising results in earlier trials. Of the large number of product candidates in development, only a small percentage result in the submission of an NDA to the FDA or comparable regulatory filing to regulatory authorities in other jurisdictions, and even fewer are approved for marketing. Even if clinical trials are completed, we or our collaborators may not submit applications for required authorizations to manufacture and/or market potential products or any such application may not be reviewed and approved by the appropriate regulatory authorities in a timely manner, if at all.
Our clinical trials face a number of risks, and our product candidates may fail regardless of whether our collaborators successfully complete the clinical trials and apply for such required authorizations for a number of reasons, including:
•a product candidate may fail to receive the regulatory clearances required to market them as drugs;
•a product candidate may be subject to proprietary rights held by others requiring the negotiation of a license agreement prior to marketing;
•a product candidate may be difficult or expensive to manufacture on a commercial scale;
•a product candidate may have adverse side effects that make their use less desirable;
•a product candidate may fail to compete with product candidates or other treatments commercialized by competitors;
•a product candidate may not prove to be efficacious or safe;
•patients may die or suffer other adverse effects for reasons that may or may not be related to the product candidate being tested;
•the results of clinical trials may not be consistent with positive results of earlier trials; and
•the results of clinical trials may not meet the level of statistical significance required by the FDA or comparable foreign regulatory authorities.
Our drug development programs are at various stages of development and the historical rate of failures for product candidates in our industry is extremely high. We have had several clinical studies evaluating pimavanserin that did not achieve statistical significance on certain endpoints, including the unsuccessful Phase 3 ADVANCE-2 study of pimavanserin for the treatment of the negative symptoms of schizophrenia in March 2024 and the unsuccessful Phase 2 study of pimavanserin for the treatment of irritability associated with autism spectrum disorder in pediatric populations (Pediatric Phase 2 Trial) in October 2024. At this time, we are not planning to conduct any additional clinical studies for pimavanserin.
With the completion of the Pediatric Phase 2 Trial, we believe we now have completed the FDA’s requirements to qualify for a pediatric exclusivity for pimavanserin. However, there is no assurance that FDA will confirm that such requirements have been met and that the pediatric exclusivity will be granted.
An unfavorable outcome in any of our ongoing or future development efforts for trofinetide or in the post-marketing studies for DAYBUE could be a major set-back for the programs and for us, generally. In particular, an unfavorable outcome in our trofinetide programs or in the post-marketing studies for DAYBUE, may require us to delay, devote additional substantial resources to, reduce the scope of, or eliminate the affected program and could have a material adverse effect on us and the value of our common stock. Also, although we have submitted a marketing application for the approval of trofinetide in the EU, there is no guarantee we will receive regulatory approval.
We are currently conducting several studies with our product candidates, including our Phase 2 study evaluating the efficacy and safety of an internally-developed compound known as ACP-204, as a potential treatment for the treatment of hallucinations and delusions associated with ADP and our Phase 3 COMPASS study evaluating the efficacy and safety of ACP-101 (intranasal carbetocin) for the treatment of hyperphagia in PWS and may conduct additional studies with these and other product candidates in the future. We plan to initiate an additional Phase 2 study of ACP-204 in LBDP in the third quarter of 2025 and to initiate a Phase 2 study of ACP-711 in essential tremor in 2026.
Drug development is a long, expensive and unpredictable process. Even if we do successfully complete clinical trials, those results are not necessarily predictive of results of additional trials that may be needed before a marketing application may be submitted to regulatory authorities. If we are unable to develop, or obtain marketing approval for, or, if approved, successfully commercialize our product candidates, we may not be able to generate sufficient revenue and our business operations and financial performance may be materially and adversely affected.
Expanded access or compassionate use programs could subject us to additional risks.
We currently provide and may provide in the future access to unapproved products or product candidates outside of clinical trials through expanded access or compassionate use programs (sometimes referred to as right to try programs). These patients generally have life-threatening illnesses for which there are no alternative therapies or they have exhausted all other available therapies. There are a number of risks that we may face as a result of our expanded access or compassionate use programs. For example, the risk for serious adverse events in certain of these patient populations is high, which, if those adverse events are determined (or perceived) to be drug-related, could have a negative impact on the safety profile of our products and product candidates and cause significant delays, result in an inability to successfully commercialize our products and materially harm our business.
In certain jurisdictions, we may be required to provide our products for free if we participate in expanded access or compassionate use programs in certain jurisdictions. In other jurisdictions we may be required to return some or all of the revenue we may generate through our expanded access or compassionate use programs if the appropriate foreign regulatory authority ultimately does not approve our products or product candidates for marketing in the jurisdiction of our expanded access or compassionate use programs. If this were to occur, it could materially and adversely affect our business operations and financial performance.
Delays, suspensions and terminations in our clinical trials for our product candidates could result in increased costs to us and delay our ability to generate product revenues.
The commencement of clinical trials can be delayed for a variety of reasons, including delays in:
•demonstrating sufficient safety and efficacy to obtain regulatory approval to commence a clinical trial;
•reaching agreement on acceptable terms with prospective contract research organizations (CROs) and clinical trial sites;
•manufacturing sufficient quantities of a product candidate;
•obtaining clearance from the FDA to commence clinical trials pursuant to an Investigational New Drug application;
•obtaining approval to conduct clinical trials in countries or jurisdictions outside the United States pursuant to evolving regional and local regulations (e.g., EU Clinical Trials Regulation (EU No. 536/2014));
•obtaining institutional review board approval to conduct a clinical trial at a prospective clinical trial site; and
•patient recruitment, which is a function of many factors, most of which is outside our control, including the size of the patient population, the nature of the protocol, the proximity of patients to clinical trial sites, the availability of effective treatments for the relevant disease and the eligibility criteria for the clinical trial.
Once a clinical trial has begun, it may be delayed, suspended or terminated due to a number of factors, including:
•competition for internal and external resources, including clinical sites and study patients, that we may choose to allocate to other programs;
•ongoing discussions with regulatory authorities regarding the scope or design of our clinical trials or requests by them for supplemental information with respect to our clinical trial results;
•imposition of clinical holds by regulatory authorities or institutional review boards;
•failure to conduct clinical trials in accordance with regulatory requirements;
•inability to monitor patients adequately during or after treatment;
•difficulty monitoring multiple study sites;
•patient enrollment, which is a function of many factors, most of which is outside our control, including the size of the patient population, the nature of the protocol, the proximity of patients to clinical trial sites, the availability of effective treatments for the relevant disease and the eligibility criteria for the clinical trial;
•lower than anticipated screening or retention rates of patients in clinical trials;
•serious adverse events or side effects experienced by participants; and
•insufficient supply or deficient quality of product candidates or other materials necessary for the conduct of our clinical trials.
In addition, enrollment and retention of patients in, or the ability to receive results from, clinical trials could be disrupted by geopolitical or macroeconomic developments. For example, as a result of the conflict between Ukraine and Russia, we experienced temporary delays in accessing historical records of certain clinical trial sites located in Russia. It is possible that enrollment in future studies, could be impacted due to the same or similar geopolitical or macroeconomic developments. If patients withdraw from our trials, miss scheduled doses or follow-up visits or otherwise fail to follow trial protocols, or if our trial results are otherwise disrupted or disputed due to such developments, the integrity of data from our trials may be compromised or not accepted by the FDA or other regulatory authorities, which would represent a significant setback for the applicable program.
Many of these factors may also ultimately lead to denial of regulatory approval of a current or potential future product candidate. If we experience delays, suspensions or terminations in a clinical trial, clinical trial materials or investigational products, the commercial prospects for the related product candidate will be harmed, and our ability to generate product revenues will be delayed.
If we are unable to attract, retain, and motivate key management, research and development, and sales and marketing personnel, our drug development programs, our research and discovery efforts, and our commercialization plans may be delayed and we may be unable to successfully commercialize our products, or develop our product candidates.
Our success depends on our ability to attract, retain, and motivate highly qualified management, scientific, and commercial personnel. In particular, our development programs depend on our ability to attract and retain highly skilled development personnel, especially in the fields of CNS disorders and rare diseases. We are currently hiring, and in the future we expect to need to continue to hire, additional personnel as we expand our research and development efforts for our products and product candidates, and commercial activities for our products. We face competition for experienced management, scientists, clinical operations personnel, commercial and other personnel from numerous companies and academic and other research institutions across all jurisdictions in which our products may be commercialized. Many of the other biotechnology and pharmaceutical companies with whom we compete for qualified personnel have greater financial and other resources, different risk profiles and longer histories in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. Some of these characteristics may be more appealing to high quality candidates than that which we have to offer. If we are unable to continue to attract and retain high quality personnel, the rate and success at which we can develop and commercialize products and product candidates, if approved, will be limited. If we are unable to attract and retain the necessary personnel, it will significantly impede our commercialization efforts for our products, and the achievement of our research and development objectives.
In September 2024, Catherine Owen Adams became our new Chief Executive Officer (CEO), replacing our former CEO, Stephen R. Davis. Our new CEO will be critical to executing on and achieving our strategy. Further, our new CEO may bring different perspectives, and the future strategy and direction of our business may differ materially from those of the past. If we are unable to execute an orderly transition and successfully integrate our new CEO into our leadership team, we may experience material disruptions to our operations and our financial condition and results of operations may be adversely affected.
All of our employees are “at will” employees, which means that any employee may quit at any time and we may terminate any employee at any time. We do not carry “key person” insurance covering members of senior management.
Risks Related to Our Business
If we fail to develop, acquire or in-license other product candidates or products, our business and prospects would be limited. Even if we obtain rights to other product candidates or products, we will incur a variety of costs and may never realize the anticipated benefits.
Part of our corporate strategy is to develop, acquire or in-license businesses, technologies, product candidates or products that we believe are a strategic fit with our business. The success of this strategy depends in large part on the combination of our regulatory, development and commercial capabilities and expertise and our ability to identify, select and acquire or in-license clinically-enabled product candidates for the treatment of CNS disorders and rare diseases, or for therapeutic indications that complement or augment our current products and product candidates, or that otherwise fit into our development or strategic plans on terms that are acceptable to us. Identifying, selecting and acquiring or in-licensing promising product candidates requires substantial technical, financial and human resources expertise, and we may not be successful in identifying acquisition targets, completing proposed acquisitions and integrating any acquired businesses, technologies, services or products into our current infrastructure. Efforts to do so may not result in the actual acquisition or in-license of a particular product candidate, potentially resulting in a diversion of our management’s time and the expenditure of our resources with no resulting benefit. If we are unable to identify, select and acquire or license suitable product candidates from third parties on terms acceptable to us, our business and prospects will be limited.
The process of integrating any acquired business, technology, service, or product may result in unforeseen operating difficulties and expenditures and may divert significant management attention from our ongoing business operations. As a result, we will incur a variety of costs in connection with an acquisition and may never realize its anticipated benefits. Moreover, any product candidate we identify, select and acquire or license may require additional, time-consuming development or regulatory efforts prior to commercial sale, including preclinical studies, if applicable, and extensive clinical testing and approval by the FDA and applicable foreign regulatory authorities. All product candidates are prone to the risk of failure that is inherent in pharmaceutical product development, including the possibility that the product candidate will not be shown to be sufficiently safe and/or effective for approval by regulatory authorities. In addition, any such products that are approved may not be manufactured or produced economically, successfully commercialized or widely accepted in the marketplace or be more effective or desired than other commercially available alternatives.
We have a history of net losses and we may not be able to predict the extent of future losses.
We have experienced significant net losses since our inception. As of December 31, 2024, we had an accumulated deficit of approximately $2.2 billion. We expect to increase our expenses and other investments in the coming years as we fund our operations, in-licensing or acquisition opportunities, and capital expenditures. Thus, our future operating results, profitability and other financial metrics may fluctuate from period to period, and we will need to generate significant revenues to achieve and maintain profitability and/or positive cash flow on a sustained basis.
We expect that our revenues over the next few years will be entirely dependent on our ability to generate product sales. Substantially all of our revenues since May 2016 were from U.S. net product sales of NUPLAZID and DAYBUE. To the extent that we cannot generate significant revenues from the sale of our products to cover our expenses, including the significant expenses associated with commercializing our products and continuing to develop trofinetide in additional indications and jurisdictions outside the U.S., we may not achieve profitability and/or may have to reduce our commercialization and/or research and development activities to become profitable, which would harm our future growth prospects. Additionally, to obtain revenues from our product candidates, if approved, we must succeed, either alone or with others, in developing, obtaining regulatory approval for, manufacturing and marketing compounds with significant market potential. We may never succeed in these activities and may never generate revenues that are significant enough to achieve profitability.
We may require additional financing in the future to fund our operations. If we cannot raise additional financing in the future, we may be unable to fund our business plan and our future research, development, commercial and manufacturing efforts.
We have funded our operations primarily with revenues from sales of our products since their approvals, and through sales of our equity securities and interest income. We anticipate that the level of cash used in our operations will fluctuate in future periods depending on the levels of spending required for our ongoing and planned commercial activities for our products, our ongoing and planned development activities for pimavanserin for the negative symptoms of schizophrenia, ACP-101 as a treatment for PWS and ACP-204 as a treatment for ADP, studies to be conducted pursuant to our PMRs, our ongoing and planned development activities for other early- and late-stage product candidates and strategic business development to further expand our portfolio. We expect that our cash, cash equivalents and investment securities, as well as funds generated by anticipated sales of our products, will be sufficient to fund our planned operations through and beyond the next 12 months.
We may require additional financing in the future to fund our operations. Our future capital requirements will depend on, and could increase significantly as a result of, many factors, including:
•the costs of acquiring additional product candidates or research and development programs;
•the scope, prioritization and number of our research and development programs;
•the ability of our collaborators and us to reach the milestones and other events or developments triggering payments under our collaboration or license agreements, or our collaborators’ ability to make payments under these agreements;
•our ability to enter into new collaboration and license agreements;
•the progress in, and the costs of, our ongoing and planned development activities for pimavanserin, post-marketing studies for DAYBUE to be conducted over the next several years, and ongoing and planned commercial activities for our products;
•the costs of our development activities for our product candidates;
•the costs of commercializing our products, including the maintenance and development of our sales and marketing capabilities;
•the costs of establishing, or contracting for, sales and marketing capabilities for our product candidates;
•the amount of U.S. product sales from our products;
•the costs of preparing applications for regulatory approvals for DAYBUE in jurisdictions other than the U.S., for NUPLAZID in additional indications other than PDP and for other product candidates, as well as the costs required to support review of such applications;
•the costs of manufacturing and distributing our products for commercial use in the U.S.;
•our ability to obtain regulatory approval for, and subsequently generate product sales from, NUPLAZID for the negative symptoms of schizophrenia, or from DAYBUE, and our product candidates;
•the extent to which we are obligated to reimburse collaborators or collaborators are obligated to reimburse us for costs under collaboration agreements;
•the costs involved in filing, prosecuting, enforcing, and defending patent claims and other intellectual property rights;
•the costs of maintaining or securing manufacturing arrangements for clinical or commercial production of pimavanserin, trofinetide or other product candidates; and
•the costs associated with litigation, including the costs incurred in defending against any product liability claims that may be brought against us related to our products.
In the past, periods of turmoil and volatility in the financial markets have adversely affected the market capitalizations of many biotechnology companies, and generally made equity and debt financing more difficult to obtain. For example, as a result of geopolitical and macroeconomic developments, the global credit and financial markets have experienced extreme volatility and disruptions, including diminished liquidity and credit availability, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability. These events, coupled with other factors, may limit our access to additional financing in the future if needed, and could have a material adverse effect on our ability to access sufficient funding. We cannot be certain that additional funding will be available to us on a timely basis, on acceptable terms, or at all. If additional funds are not available, we will be required to delay, reduce the scope of, or eliminate one or more of our research or development programs or our commercialization efforts. We also may be required to relinquish greater or all rights to product candidates at an earlier stage of development or on less favorable terms than we would otherwise choose. Additional funding, if necessary and obtained, may significantly dilute existing stockholders and could negatively impact the price of our stock.
We expect that our results of operations will fluctuate, which may make it difficult to predict our future performance from period to period.
Our operating results have fluctuated in the past and are likely to do so in future periods. Some of the factors that could cause our operating results to fluctuate from period to period include:
•the success of our commercialization of our products;
•the impact of geopolitical and macroeconomic developments, general political, health and economic conditions, as well as any related political or economic responses and counter-responses or otherwise by various global actors or the general effect on the global economy and supply chain, pandemics or epidemics, economic slowdowns, recessions, inflation, high interest rates and tightening of credit markets on our business;
•the status and cost of our PMRs for DAYBUE;
•the variation in our gross-to-net adjustments from quarter to quarter, primarily because of the fluctuation in our share of the donut hole for Medicare Part D patients;
•the status and cost of development and commercialization of trofinetide for indications other than for the treatment of Rett syndrome;
•the status and cost of development and commercialization of our product candidates, if approved, including compounds being developed under our collaborations;
•whether we acquire or in-license additional product candidates or products, and the status of development and commercialization of such product candidates, if approved, or products;
•whether we generate revenues or reimbursements by achieving specified research, development or commercialization milestones under any agreements or otherwise receive potential payments under these agreements;
•whether we are required to make payments due to achieving specified milestones under any licensing or similar agreements or otherwise make payments under these agreements;
•the incurrence of preclinical or clinical expenses that could fluctuate significantly from period to period, including reimbursement obligations pursuant to our collaboration agreements;
•the initiation, termination, or reduction in the scope of our collaborations or any disputes regarding these collaborations;
•the timing of our satisfaction of applicable regulatory requirements;
•the rate of expansion of our clinical development, other internal research and development efforts, and pre-commercial and commercial efforts;
•the effect of competing technologies and products and market developments;
•the costs associated with litigation, including the costs incurred in defending against any product liability claims that may be brought against us related to our products or our product candidates; and
•general and industry-specific economic conditions.
We believe that comparisons from period to period of our financial results are not necessarily meaningful and should not be relied upon as indications of our future performance.
From time to time, we provide guidance relating to our expectations for net sales of our products and certain expense line items based on estimates and the judgment of management. If, for any reason, our actual net sales or expenses differ materially from our guidance, we may have to revise our previously announced financial guidance. If we change, update or fail to meet any element of such guidance, our stock price could decline.
Changes in tax laws or regulations that are applied adversely to us or our customers may have a material adverse effect on our business, cash flow, financial condition or results of operations.
New income, sales, use or other tax laws, statutes, rules, regulations or ordinances could be enacted at any time, which could adversely affect our business operations and financial performance. Further, existing tax laws, statutes, rules, regulations or ordinances could be interpreted, changed, modified or applied adversely to us. For example, legislation enacted in 2017 informally titled the Tax Cuts and Jobs Act, the Coronavirus Aid, Relief, and Economic Security Act and the Inflation Reduction Act enacted many significant changes to the U.S. tax laws. For example, effective January 1, 2022, the Tax Cuts and Jobs Act eliminated the option to deduct research and development expenses for tax purposes in the year incurred and requires taxpayers to capitalize and subsequently amortize such expenses over five years for research activities conducted in the United States and over 15 years for research activities conducted outside the United States. Although there have been legislative proposals to repeal or defer the capitalization requirement to later years, the provision may not actually be repealed or otherwise modified. Future guidance from the Internal Revenue Service and other tax authorities with respect to such legislation may affect us, and certain aspects of such legislation could be repealed or modified in future legislation. In addition, it is uncertain if and to what extent various states will conform to federal tax laws. Future tax reform legislation could have a material impact on the value of our deferred tax assets, could result in significant one-time charges, and could increase our future U.S. tax expense.
Our ability to use net operating loss carryforwards and certain other tax attributes to offset future taxable income or taxes may be limited.
Portions of our net operating loss carryforwards could expire unused and be unavailable to offset future income tax liabilities. Under current law, federal net operating losses incurred in tax years beginning after December 31, 2017, may be carried forward indefinitely, but the deductibility of such federal net operating loss carryforwards in a taxable year is limited to 80% of taxable income in such year. In addition, under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended (the Code), and corresponding provisions of state law, if a corporation undergoes an “ownership change,” which is generally defined as a greater than 50 percent change, by value, in its equity ownership over a three-year period, the corporation’s ability to use its pre-change net operating loss carryforwards and other pre-change tax attributes to offset its post-change income or taxes may be limited. We have experienced ownership changes in the past and we may experience additional ownership changes in the future as a result of subsequent shifts in our stock ownership, some of which may be outside of our control. If an ownership change occurs and our ability to use our net operating loss carryforwards is materially limited, it would harm our future operating results by effectively increasing our future tax obligations. In addition, at the state level, there may be periods during which the use of net operating loss carryforwards is suspended or otherwise limited, which could accelerate or permanently increase state taxes owed. For example, California imposed limits on the usability of California state net operating losses to offset taxable income in tax years beginning after 2023 and before 2027. As a result, if
we earn net taxable income, we may be unable to use all or a material portion of our net operating loss carryforwards and other tax attributes, which could potentially result in increased future tax liability to us and adversely affect our future cash flows.
Tax authorities could reallocate our taxable income among our subsidiaries, which could increase our overall tax liability.
The amount of taxes we pay in different jurisdictions depends on the application of the tax laws of various jurisdictions, including the United States, to our international business activities, tax rates, new or revised tax laws, or interpretations of tax laws and policies, and our ability to operate our business in a manner consistent with our corporate structure and intercompany arrangements. In 2015, we licensed worldwide intellectual property rights related to pimavanserin in certain indications to Acadia Pharmaceuticals GmbH, our wholly owned Swiss subsidiary (Acadia GmbH), and in July 2020 we licensed additional related rights to Acadia GmbH. Our goals for the establishment of Acadia GmbH, and the licensing of worldwide intellectual property rights for pimavanserin, include building a platform for long-term operational and financial efficiencies, including tax-related efficiencies. The taxing authorities of the jurisdictions in which we operate may challenge our methodologies for pricing intercompany transactions pursuant to our intercompany arrangements or disagree with our determinations as to the income and expenses attributable to specific jurisdictions. In addition, future changes in U.S. and non-U.S. tax laws, including implementation of international tax reform relating to the tax treatment of multinational corporations, if enacted, may reduce or eliminate any potential financial efficiencies that we hoped to achieve by establishing this operational structure. Additionally, taxing authorities, such as the U.S. Internal Revenue Service, may audit and otherwise challenge these types of arrangements, and have done so with other companies in the pharmaceutical industry. If any such challenge or disagreement were to occur or change in tax law were enacted, we could be required to pay additional taxes, interest and penalties, which could result in one-time tax charges, higher effective tax rates, reduced cash flows and lower overall profitability of our operations. Our financial statements could fail to reflect adequate reserves to cover such a contingency. Similarly, a taxing authority could assert that we are subject to tax in a jurisdiction where we believe we have not established a taxable connection, often referred to as a “permanent establishment” under international tax treaties, and such an assertion, if successful, could increase our expected tax liability in one or more jurisdictions.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the U.S. and global economies, the U.S. and global financial markets and adverse macroeconomic developments. U.S. and global market and economic conditions have been, and continue to be, disrupted and volatile due to many factors, including material shortages and related manufacturing and supply chain challenges, geopolitical developments (as well as any related political or economic responses and counter-responses or otherwise by various global actors or the general effect on the global economy and manufacturing and supply chain), and the responses by central banking authorities to control inflation, among others. General business and economic conditions that could affect our business, financial condition or results of operations include fluctuations in economic growth, debt and equity capital markets, liquidity of the global financial markets, the availability and cost of credit, investor and consumer confidence, and the strength of the economies in which we, our collaborators, our manufacturers and our suppliers operate.
A severe or prolonged global economic downturn could result in a variety of risks to our business. For example, high inflation may result in increases in our operating costs (including our labor costs), reduced liquidity and limits on our ability to access credit or otherwise raise capital on acceptable terms, if at all. In addition, reduced government spending and volatility in financial markets may have the effect of further increasing economic uncertainty and heightening these risks. Risks of a prolonged global economic downturn are particularly true in Europe, which is undergoing a continued severe economic crisis. A weak or declining economy could also strain our suppliers and manufacturers, possibly resulting in supply and clinical trial disruption. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
We or the third parties upon whom we depend may be adversely affected by catastrophic events, such as earthquakes, fires or other natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
We depend on our employees, consultants, and CROs, as well as regulatory agencies and other parties, for the continued operation of our business. While we maintain disaster recovery plans, those plans may not adequately protect us. Despite any precautions that we or any third parties on whom we depend take for catastrophic events, including earthquakes, fires or other natural disasters, these events could result in significant disruptions to our research and development, clinical trials, manufacturing and the commercialization of our products. Long-term disruptions in the infrastructure caused by these types of events, particularly involving geographies in which we or third parties on whom we depend have offices or manufacturing, distribution or clinical trial sites, could adversely affect our businesses. Although we carry business interruption insurance policies and typically have provisions in our contracts that protect us in certain events, our coverage might not include or be adequate to compensate us for all losses that may occur. Any catastrophic event affecting us or the third parties on whom we depend could have a material adverse effect on our business, results of operations, financial condition and prospect.
We have incurred, and expect to continue to incur, significant costs as a result of laws and regulations relating to corporate governance and other matters.
Laws and regulations affecting public companies, including provisions of the Dodd-Frank Wall Street Reform and Consumer Protection Act that was enacted in July 2010, the provisions of the Sarbanes-Oxley Act of 2002 (SOX), and rules adopted or proposed by the SEC and by The Nasdaq Stock Market, have resulted in, and will continue to result in, significant costs to us as we evaluate the implications of these rules and respond to their requirements. In the future, if we are not able to issue an evaluation of our internal control over financial reporting, as required, or we or our independent registered public accounting firm determine that our internal control over financial reporting is not effective, this shortcoming could have an adverse effect on our business and financial results and the price of our common stock could be negatively affected. New rules could make it more difficult or more costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the coverage that is the same or similar to our current coverage. The impact of these events could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors and board committees, and as our executive officers. We cannot predict or estimate the total amount of the costs we may incur or the timing of such costs to comply with these rules and regulations.
Our business involves the use of hazardous materials, and we and our third-party manufacturers and suppliers must comply with environmental, health and safety laws and regulations, which can be expensive and restrict how we do, or interrupt our, business.
Our research and development activities and our third-party manufacturers’ and suppliers’ activities involve the generation, storage, use and disposal of hazardous materials, including the components of our products and product candidates and other hazardous compounds and wastes. We and our manufacturers and suppliers are subject to environmental, health and safety laws and regulations governing, among other matters, the use, manufacture, generation, storage, handling, transportation, discharge and disposal of these hazardous materials and wastes and worker health and safety. In some cases, these hazardous materials and various wastes resulting from their use are stored at our and our manufacturers’ facilities pending their use and disposal. We cannot eliminate the risk of contamination or injury, which could result in an interruption of our commercialization efforts, research and development efforts and business operations, damages and significant cleanup costs and liabilities under applicable environmental, health and safety laws and regulations. We also cannot guarantee that the safety procedures utilized by our third-party manufacturers for handling and disposing of these materials and wastes generally comply with the standards prescribed by these laws and regulations. We may be held liable for any resulting damages costs or liabilities, which could exceed our resources, and state or federal or other applicable authorities may curtail our use of certain materials and/or interrupt our business operations. Furthermore, environmental, health and safety laws and regulations are complex, change frequently and have tended to become more stringent. We cannot predict the impact of such changes and cannot be certain of our future compliance. Failure to comply with these environmental, health and safety laws and regulations may result in substantial fines, penalties or other sanctions. We do not currently carry hazardous waste insurance coverage.
Our management has broad discretion over the use of our cash and we may not use our cash effectively, which could adversely affect our results of operations.
Our management has significant flexibility in applying our cash resources and could use these resources for corporate purposes that do not increase our market value, or in ways with which our stockholders may not agree. We may use our cash resources for corporate purposes that do not yield a significant return or any return at all for our stockholders, which may cause our stock price to decline.
Risks Related to Our Relationships with Third Parties
We depend on collaborations with third parties to develop certain of our product candidates and may need to enter into future collaborations to develop and commercialize certain of our product candidates.
We depend on collaborations with third parties to develop certain of our product candidates and may need to enter into future collaborations to develop and commercialize certain of our product candidates. In addition, we may choose to rely on collaborations in the future for our products or other product candidates, including for the commercialization of DAYBUE in selected markets outside of the U.S.
Our collaborators may fail to develop or effectively commercialize products using our product candidates, if approved, or technologies because they:
•do not have sufficient resources or decide not to devote the necessary resources due to internal constraints such as limited cash or human resources or a change in strategic focus;
•may not properly maintain, enforce or defend our intellectual property rights or may use our proprietary information in a manner that could jeopardize or invalidate our proprietary information or expose us to potential litigation;
•terminate the arrangement or allow it to expire, which would delay the development and commercialization and may increase the cost of developing and commercializing our products or product candidates, if approved;
•may sell, transfer or divest assets or programs related to our partnered product or product candidates;
•may not pursue further development and commercialization of products resulting from the strategic collaboration arrangement;
•decide to pursue a competitive product developed outside of the collaboration; or
•cannot obtain the necessary regulatory approvals.
Collaborations are complex and time-consuming to negotiate and document. Given the current economic and industry environment, it is possible that competition for new collaborators may increase. We may not be able to negotiate additional collaborations on a timely basis, on acceptable terms, or at all. If we are unable to find new collaborations, we may not be able to continue advancing our programs alone.
Our collaborations may be subject to conflicts or disputes, which could have a material adverse effect on our business, results of operations and financial condition.
Conflicts may arise in our collaborations due to one or more of the following:
•disputes or breaches with respect to payments that we believe are due under the applicable agreements, particularly in the current environment when companies, including large established ones, may be seeking to reduce external payments;
•disputes on strategy as to what development or commercialization activities should be pursued under the applicable agreements;
•disputes as to the responsibility for conducting development and commercialization activities pursuant to the applicable collaboration, including the payment of costs related thereto;
•disagreements with respect to ownership of intellectual property rights;
•unwillingness on the part of a collaborator to keep us informed regarding the progress of its development and commercialization activities, or to permit public disclosure of these activities;
•delay or reduction of a collaborator’s development or commercialization efforts with respect to our product candidates, if approved; or
•termination or non-renewal of the collaboration.
Conflicts arising with our collaborators could impair the progress of our product candidates, harm our reputation, result in a loss of revenues, reduce our cash position, and cause a decline in our stock price.
In addition, in our past collaborations, from time to time, we have agreed not to conduct independently, or with any third party, any research that is directly competitive with the research conducted under the applicable program. Any collaborations we establish in the future may have the effect of limiting the areas of research that we may pursue, either alone or with others. Conversely, the terms of any collaboration we may establish in the future might not restrict our collaborators from developing, either alone or with others, products or product candidates in related fields that are competitive with the products or product candidates that are the subject of these collaborations. Competing products and product candidates, either developed by our collaborators or to which our collaborators have rights, may result in the allocation of resources by our collaborators to competing products and product candidates, and their withdrawal of support for our products and product candidates or may otherwise result in lower demand for our potential products and product candidates.
In addition, disputes may arise between us and our licensors regarding intellectual property subject to a license agreement, including:
•the scope of rights granted under the license agreement and other interpretation-related issues;
•whether and the extent to which our technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement;
•our right to sublicense patents and other rights to third parties;
•our diligence obligations with respect to the use of the licensed technology in relation to our development and commercialization of our product candidates, if approved, and what activities satisfy those diligence obligations;
•our right to transfer or assign the license; and
•the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners.
If disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on acceptable terms, we may not be able to successfully develop and commercialize the related product candidates, if approved, which would have a material adverse effect on our business.
We rely on third parties to conduct our clinical trials and perform data collection and analysis, which may result in costs and delays that prevent us from successfully commercializing product candidates, if approved.
Although we design and manage our current preclinical studies and clinical trials, we currently do not have the ability to conduct clinical trials for our product candidates on our own. We rely on CROs, medical institutions, clinical investigators, and contract laboratories to perform data collection and analysis and other aspects of our clinical trials. In addition, we also rely on third parties to assist with our preclinical studies, including studies regarding biological activity, safety, absorption, metabolism, and excretion of product candidates. Some of these third parties may experience shutdowns or other disruptions as a result of adverse geopolitical or macroeconomic developments and therefore may be unable to provide the level of service that we have received in the past.
Our preclinical activities or clinical trials may be delayed, suspended, or terminated if:
•these third parties do not successfully carry out their contractual duties or fail to meet regulatory obligations or expected deadlines;
•these third parties need to be replaced; or
•the quality or accuracy of the data obtained by these third parties is compromised due to their failure to adhere to our clinical protocols or regulatory requirements or for other reasons.
Failure to perform by these third parties may increase our development costs, delay our ability to obtain regulatory approval, and delay or prevent the commercialization of our product candidates, if approved. We currently use several CROs to perform services for our preclinical studies and clinical trials. While we believe that there are numerous alternative sources to provide these services, in the event that we seek such alternative sources, we may not be able to enter into replacement arrangements without delays, additional expenditures, or at all, any of which could negatively affect our business, results of operations, financial condition and prospects.
We currently depend, and in the future will continue to depend, on third parties to manufacture our products and product candidates. If these manufacturers fail to provide us or our collaborators with adequate supplies of clinical trial materials and commercial product or fail to comply with the requirements of regulatory authorities, we may be unable to develop or commercialize our products or product candidates, if approved.
We have no manufacturing facilities and only limited experience as an organization in the manufacturing of drugs or in designing drug-manufacturing processes. We have contracted with third-party manufacturers to produce, in collaboration with us, our products and product candidates.
We have contracted with Patheon Pharmaceuticals Inc. (Patheon) to manufacture NUPLAZID 10 mg tablet and 34 mg capsule drug product and DAYBUE for commercial use in the U.S. and Canada. We have also contracted with a second contract manufacturing organization to manufacture NUPLAZID 34 mg drug product for commercial use in the U.S. Additionally, we have contracted with Siegfried AG to manufacture API to be used in the manufacture of NUPLAZID drug product for commercial use, Corden Pharma Bergamo S.p.A. (Corden) and F.I.S. Fabbrica Italiana Sintetici S.p.A. (FIS) to manufacture API to be used in the manufacture of DAYBUE drug product for commercial use, and Patheon and CoreRx Inc. (CoreRx) to manufacture DAYBUE for commercial use. However, we have not entered into any agreements with any alternate suppliers for 10 mg NUPLAZID drug product or NUPLAZID API. We may face delays or increased costs in our supply chain that could jeopardize the commercialization of our products. While we currently have sufficient API for both NUPLAZID and DAYBUE and NUPLAZID and DAYBUE finished products on hand to continue our commercial and clinical operations as planned, depending on the effects of geopolitical and macroeconomic developments and whether such developments cause disruptions, we may face such delays or costs in future years. If any third party in our supply or distribution chain for materials or finished product is adversely impacted by geopolitical and macroeconomic developments, our supply chain may be disrupted, limiting our ability to manufacture, test and distribute our products for commercial sales and our product candidates for our clinical trials and research and development operations. For example, it takes approximately two years for our third-party manufacturers to produce DAYBUE API, and a supply chain disruption in DAYBUE API would cause delays or increased costs to us that could jeopardize the commercialization of DAYBUE.
Even though we have agreements with third parties for the manufacture of our products, the FDA may not approve the facilities of such manufacturers, the manufacturers may not perform as agreed, or the manufacturers may terminate their agreements with us. If any of the foregoing circumstances occur, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, maintain or obtain, as applicable, regulatory approval for or market our products or product candidates. While we believe that there will be alternative sources available to manufacture our products and product candidates, in the event that we seek such alternative sources, we may not be able to enter into replacement arrangements without delays or additional expenditures. We cannot estimate these delays or costs with certainty but, if they were to occur, they could cause a delay in our development and commercialization efforts, which would have a negative effect on our business, results of operations, financial condition and prospects.
The manufacturers of our products and product candidates, including Patheon, Siegfried, Corden, FIS and CoreRx, are obliged to operate in accordance with FDA-mandated current good manufacturing practices (cGMPs), and we have limited control over the ability of third-party manufacturers to maintain adequate quality control, quality assurance and qualified personnel to ensure compliance with cGMPs. In addition, the facilities used by our third-party manufacturers to manufacture our products and product candidates must be approved by the FDA pursuant to inspections that will be conducted prior to any grant of regulatory approval by the FDA. If any of our third-party manufacturers are unable to successfully manufacture material that conforms to our specifications and the FDA’s strict regulatory requirements, or pass regulatory inspection, they will not be able to secure or maintain approval for the manufacturing facilities. Additionally, a failure by any of our third-party manufacturers to establish and follow cGMPs or to document their adherence to such practices may lead to significant delays in clinical trials or in obtaining regulatory approval of product candidates, or result in issues maintaining regulatory approval of our products and any product candidate that receives regulatory approval, negatively impact our commercialization of our products, or lead to significant delays in the launch and commercialization of any other products we may have in the future. Failure by our third-party manufacturers or us to comply with applicable regulations could result in sanctions being imposed on us, including fines, injunctions, civil penalties, failure of regulatory authorities to grant
pre-market approval of drugs, delays, suspension or withdrawal of approvals, seizures or recalls of products, operating restrictions, and criminal prosecutions.
The manufacture of pharmaceutical products requires significant capital investment, including the development of advanced manufacturing techniques and process controls. Manufacturers of pharmaceutical products often encounter difficulties in production. These problems include difficulties with production costs and yields, quality control, including stability of the product, quality assurance testing, shortages of qualified personnel, as well as compliance with strictly enforced federal, state and foreign regulations. We cannot assure you that any issues relating to the manufacture of our products or product candidates will not occur in the future. Additionally, our manufacturers may experience manufacturing difficulties due to resource constraints or as a result of labor disputes or unstable political environments. If our manufacturers were to encounter any of these difficulties, or otherwise fail to comply with their contractual obligations, our ability to commercialize our products, or provide our products or product candidates to patients in clinical trials, would be jeopardized. Any delay or interruption in our ability to meet commercial demand for our products and any other approved products will result in the loss of potential revenues and could adversely affect our ability to gain market acceptance for these products. In addition, any delay or interruption in the supply of clinical trial supplies could delay the completion of clinical trials, increase the costs associated with maintaining clinical trial programs and, depending upon the period of delay, require us to commence new clinical trials at additional expense or terminate clinical trials completely.
Failures or difficulties faced at any level of our supply chain could materially adversely affect our business and delay or impede the development and commercialization of our products or product candidates, if approved, and could have a material adverse effect on our business, results of operations, financial condition and prospects. Further, changes in federal policy could affect the geopolitical landscape and could give rise to circumstances that negatively affect our business. The third parties that manufacture our products and product candidates have manufacturing activities located in Canada, Europe and Switzerland. The U.S. has implemented, and has proposed to further implement, tariffs that may increase the costs of our third-party manufacturers and the expense to us to produce our products and product candidates. If such actions were to materially affect us or our third-party manufacturers, we may not be able to successfully commercialize our products, which would have an adverse effect on our results of operations.
We may not be able to continue or fully exploit our collaborations with outside scientific and clinical advisors, which could impair the progress of our clinical trials and our research and development efforts.
We work with scientific and clinical advisors at academic and other institutions who are experts in the field of CNS disorders and rare diseases. They assist us in our research and development efforts and advise us with respect to our clinical trials. These advisors are not our employees and may have other commitments that would limit their future availability to us. Although our scientific and clinical advisors generally agree not to engage in competing work, if a conflict of interest arises between their work for us and their work for another entity, we may lose their services, which may impair our reputation in the industry and delay the development or commercialization of our product candidates, if approved.
Risks Related to Our Intellectual Property
Our ability to compete may decline if we do not adequately protect our proprietary rights.
Our commercial success depends on obtaining and maintaining intellectual property rights to our products and product candidates and technologies, as well as successfully defending these rights against third-party challenges. Successful challenges to, or misappropriation of, our intellectual property could enable competitors to quickly duplicate or surpass our technological achievements, thus eroding our competitive position in our market. To protect our intellectual property, we rely on a combination of patents, trade secret protection and contracts requiring confidentiality and nondisclosure. If our patents are successfully challenged, we may face generic competition prior to the expiration dates of our U.S. Orange Book listed patents. In addition, potential competitors have in the past and may in the future file an Abbreviated New Drug Application (ANDA) with the FDA for generic versions of NUPLAZID, seeking approval prior to the expiration of our patents. In response, we have filed complaints against these companies alleging infringement of certain of our Orange Book-listed patents covering NUPLAZID. For a more detailed description of these matters, see the section captioned “Legal Proceedings” elsewhere in this report. While we intend to defend the validity of such patents vigorously, and will seek to use all appropriate methods to prevent their infringement, such efforts are expensive and time consuming. Any substantial decrease in the revenue and income derived from our products would have an adverse effect on our results of operations.
With regard to patents, although we have filed numerous patent applications worldwide with respect to pimavanserin, not all of our patent applications resulted in an issued patent, or they resulted in an issued patent that is susceptible to challenge by a third party. Our ability to obtain, maintain, and/or defend our patents covering our product candidates and technologies is uncertain due to a number of factors, including:
•we may not have been the first to make the inventions covered by our pending patent applications or issued patents;
•we may not have been the first to file patent applications for our product candidates or the technologies we rely upon;
•others may develop similar or alternative technologies or design around our patent claims to produce competitive products that fall outside of the scope of our patents;
•our disclosures in patent applications may not be sufficient to meet the statutory requirements for patentability;
•we may not seek or obtain patent protection in all countries that will eventually provide a significant business opportunity;
•any patents issued to us or our collaborators may not provide a basis for commercially viable products, may not provide us with any competitive advantages, or are easily susceptible to challenges by third parties;
•our proprietary technologies may not be patentable;
•changes to patent laws that limit the exclusivity rights of patent holders or make it easier to render a patent invalid;
•recent decisions by the U.S. Supreme Court limiting patent-eligible subject matter;
•litigation regarding our patents may include challenges to the validity, enforceability, scope and term of one or more patents;
•the passage of The Leahy-Smith America Invents Act (the America Invents Act), introduced new procedures for challenging pending patent applications and issued patents; and
•technology that we may in-license may become important to some aspects of our business; however, we generally would not control the patent prosecution, maintenance or enforcement of any such in-licensed technology.
Even if we have or obtain patents covering our product candidates or technologies, we may still be barred from making, using and selling our product candidates or technologies because of the patent rights of others. Others have or may have filed, and in the future are likely to file, patent applications covering compounds, assays, genes, gene products or therapeutic products that are similar or identical to ours. There are many issued U.S. and foreign patents relating to genes, nucleic acids, polypeptides, chemical compounds or therapeutic products, and some of these may encompass reagents utilized in the identification of candidate drug compounds or compounds that we desire to commercialize. Numerous U.S. and foreign issued patents and pending patent applications owned by others exist in the area of CNS disorders and the other fields in which we are developing products. These could materially affect our freedom to operate. Moreover, because patent applications can take many years to issue, there may be currently pending applications, unknown to us, that may later result in issued patents that our product candidates or technologies may infringe. These patent applications may have priority over patent applications filed by us.
We regularly conduct searches to identify patents or patent applications that may prevent us from obtaining patent protection for our proprietary compounds or that could limit the rights we have claimed in our patents and patent applications. Disputes may arise regarding the ownership or inventorship of our inventions. For applications in which all claims are entitled to a priority date before March 16, 2013, an interference proceeding can be provoked by a third-party or instituted by the U.S. Patent and Trademark Office (U.S. PTO), to determine who was the first to invent the invention at issue. It is difficult to determine how such disputes would be resolved. Applications containing a claim not entitled to priority before March 16, 2013, are not subject to interference proceedings due the change brought by the America Invents Act to a “first-to-file” system. However, a derivation proceeding can be brought by a third-party alleging that the inventor derived the invention from another.
Periodic maintenance fees on any issued patent are due to be paid to the U.S. PTO and foreign patent agencies in several stages over the lifetime of the patent. The U.S. PTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. In such an event, our competitors might be able to enter the market, which would have a material adverse effect on our business.
Some of our academic institutional licensors, research collaborators and scientific advisors have rights to publish data and information to which we have rights. We generally seek to prevent our collaborators from disclosing scientific discoveries until we have the opportunity to file patent applications on such discoveries, but in some cases, we are limited to relatively short periods to review a proposed publication and file a patent application. If we cannot maintain the confidentiality of our technology and other confidential information in connection with our collaborations, then our ability to receive patent protection or protect our proprietary information may be impaired.
Confidentiality agreements with employees and others may not adequately prevent disclosure of our trade secrets and other proprietary information and may not adequately protect our intellectual property, which could limit our ability to compete.
Because we operate in the highly technical field of drug discovery and development of small molecule drugs, we rely in part on trade secret protection in order to protect our proprietary technology and processes. However, trade secrets are difficult to protect. We enter into confidentiality, nondisclosure, and intellectual property assignment agreements with our corporate partners, employees, consultants, outside scientific collaborators, sponsored researchers, and other advisors. These agreements generally require that the other party keep confidential and not disclose to third parties all confidential information developed by the party or made known to the party by us during the course of the party’s relationship with us. These agreements also generally provide that inventions conceived by the party in the course of rendering services to us will be our exclusive property. However, these agreements may not be honored and may not effectively assign intellectual property rights to us. Enforcing a claim that a party illegally obtained and is using our trade secrets is difficult, expensive and time consuming and the outcome is unpredictable. In addition, courts outside the U.S. may be less willing to protect trade secrets. We also have not entered into any noncompete agreements with any of our employees. Although each of our employees is required to sign a confidentiality agreement with us at the time of hire, we cannot guarantee that the confidential nature of our proprietary information will be maintained in the course of future employment with any of our competitors. If we are unable to prevent unauthorized material disclosure of our intellectual property to third parties, we will not be able to establish or maintain a competitive advantage in our market, which could materially adversely affect our business, operating results and financial condition.
A dispute concerning the infringement or misappropriation of our proprietary rights or the proprietary rights of others could be time-consuming and costly, and an unfavorable outcome could harm our business.
There is a substantial amount of litigation involving patents and other intellectual property rights in the biotechnology and pharmaceutical industries, as well as administrative proceedings for challenging patents, including post-issuance review proceedings before the U.S. PTO or oppositions and other comparable proceedings in foreign jurisdictions.
Central provisions of the America Invents Act went into effect on September 16, 2012 and on March 16, 2013. The America Invents Act includes a number of significant changes to U.S. patent law. These changes include provisions that affect the way patent applications are being filed, prosecuted and litigated. For example, the America Invents Act enacted proceedings involving post-issuance patent review procedures, such as inter partes review (IPR), and post-grant review, that allow third parties to challenge the validity of an issued patent in front of the U.S. PTO Patent Trial and Appeal Board. Each proceeding has different eligibility criteria and different patentability challenges that can be raised. IPRs permit any person (except a party who has been litigating the patent for more than a year) to challenge the validity of the patent on the grounds that it was anticipated or made obvious by prior art. Patents covering pharmaceutical products have been subject to attack in IPRs from generic drug companies and from hedge funds. If it is within nine months of the issuance of the challenged patent, a third party can petition the U.S. PTO for post-grant review, which can be based on any invalidity grounds and is not limited to prior art patents or printed publications.
In post-issuance proceedings, U.S. PTO rules and regulations generally tend to favor patent challengers over patent owners. For example, unlike in district court litigation, claims challenged in post-issuance proceedings are given their broadest reasonable meaning, which increases the chance a claim might be invalidated by prior art or lack support in the patent specification. As another example, unlike in district court litigation, there is no presumption of validity for an issued patent, and thus, a challenger’s burden to prove invalidity is by a preponderance of the evidence, as opposed to the heightened clear and convincing evidence standard. As a result of these rules and others, statistics released by the U.S. PTO show a high percentage of claims being invalidated in post-issuance proceedings. Moreover, with few exceptions, there is no standing requirement to petition the U.S. PTO for inter partes review or post-grant review. In other words, companies that have not been charged with infringement or that lack commercial interest in the patented subject matter can still petition the U.S. PTO for review of an issued patent. Thus, even where we have issued patents, our rights under those patents may be challenged and ultimately not provide us with sufficient protection against competitive products or processes.
We may be exposed to future litigation by third parties based on claims that our product candidates, technologies or activities infringe the intellectual property rights of others. In particular, there are many patents relating to specific genes, nucleic acids, polypeptides or the uses thereof to identify product candidates. Some of these may encompass genes or polypeptides that we utilize in our drug development activities. If our drug development activities are found to infringe any such patents, and such patents are held to be valid and enforceable, we may have to pay significant damages or seek licenses to such patents. A patentee could prevent us from using the patented genes or polypeptides for the identification or development of drug compounds. There are also many patents relating to chemical compounds and the uses thereof. If our compounds are found to infringe any such patents, and such patents are held to be valid and enforceable, we may have to pay significant damages or seek licenses to such patents. A patentee could prevent us from making, using or selling the patented compounds.
In addition to the patent infringement lawsuits against the filers of ANDAs pertaining to NUPLAZID, we may need to resort to litigation to enforce other patents issued to us, protect our trade secrets or determine the scope and validity of third-party proprietary rights. From time to time, we may hire scientific personnel formerly employed by other companies involved in one or more areas similar to the activities conducted by us. Either we or these individuals may be subject to allegations of trade secret misappropriation or other similar claims as a result of their prior affiliations. If we become involved in litigation, it could consume a substantial portion of our managerial and financial resources, regardless of whether we win or lose. We may not be able to afford the costs of litigation. Any legal action against us or our collaborators could lead to:
•payment of damages, which could potentially be trebled if we are found to have willfully infringed a party’s patent rights;
•injunctive or other equitable relief that may effectively block our ability to further develop, commercialize, and sell products; or
•we or our collaborators having to enter into license arrangements that may not be available on commercially acceptable terms, or at all.
As a result, we could be prevented from commercializing current or future products.
Furthermore, because of the substantial amount of pre-trial document and witness discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, during the course of this kind of litigation, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. Securities analysts and investors have in the past, and may again in the future perceive these results to be negative, it could have a substantial adverse effect on the trading price of our common stock.
The patent applications of pharmaceutical and biotechnology companies involve highly complex legal and factual questions, which, if determined adversely to us, could negatively impact our patent position.
The strength of patents in the pharmaceutical and biotechnology field can be highly uncertain and involve complex legal and factual questions. The U.S. PTO’s interpretation of the Supreme Court’s decisions and the standards for patentability it sets forth are uncertain and could change in the future. Consequently, the issuance and scope of patents cannot be predicted with certainty. Patents, if issued, may be challenged, invalidated or circumvented. U.S. patents and patent applications may also be subject to interference proceedings as mentioned above, and U.S. patents may be subject to reexamination and post-issuance proceedings in the U.S. PTO (and foreign patents may be subject to opposition or comparable proceedings in the corresponding foreign patent office), which proceedings could result in either loss of the patent or denial of the patent application or loss or reduction in the scope of one or more of the claims of the patent or patent application. Similarly, opposition or invalidity proceedings could result in loss of rights or reduction in the scope of one or more claims of a patent in foreign jurisdictions. In addition, such interference, reexamination, post-issuance and opposition proceedings may be costly. Accordingly, rights under any issued patents may not provide us with sufficient protection against competitive products or processes.
In addition, changes in or different interpretations of patent laws in the U.S. and foreign countries may permit others to use our discoveries or to develop and commercialize our technology and products without providing any compensation to us or may limit the number of patents or claims we can obtain. In particular, there have been proposals to shorten the exclusivity periods available under U.S. patent law that, if adopted, could substantially harm our business. The product candidates that we are developing are protected by intellectual property rights, including patents and patent applications. If any of our product candidates becomes a marketable product, we will rely on our exclusivity under patents to sell the compound and recoup our investments in the research and development of the compound. If the exclusivity period for patents is shortened, then our ability to generate revenues without competition will be reduced and our business could be materially adversely impacted. The laws of some countries do not protect intellectual property rights to the same extent as U.S. laws and those countries may lack adequate rules and procedures for defending our intellectual property rights. For example, some countries, including many in Europe, do not grant patent claims directed to methods of treating humans and, in these countries, patent protection may not be available at all to protect our products and product candidates. In addition, U.S. patent laws may change which could prevent or limit us from filing patent applications or patent claims to protect our products and/or technologies or limit the exclusivity periods that are available to patent holders. For example, the America Invents Act (2012) included a number of significant changes to U.S. patent law. These included changes to transition from a “first-to-invent” system to a “first-to-file” system and to the way issued patents are challenged. These changes may favor larger and more established companies that have more resources to devote to patent application filing and prosecution. It is still not clear what, if any, impact the America Invents Act will ultimately have on the cost of prosecuting our patent applications, our ability to obtain patents based on our discoveries and our ability to enforce or defend our issued patents.
If we fail to obtain and maintain patent protection and trade secret protection of our product candidates, proprietary technologies and their uses, we could lose our competitive advantage and competition we face would increase, reducing our potential revenues and adversely affecting our ability to attain or maintain profitability.
Risks Related to Government Regulation and Our Industry
Healthcare reform measures may negatively impact our ability to sell NUPLAZID, DAYBUE or our product candidates, if approved, profitably.
In both the U.S. and certain foreign jurisdictions, there have been a number of legislative and regulatory proposals to change the healthcare system in ways that could impact our ability to sell our products, as described in greater detail in the Government Regulation section of this report.
For example, the Patient Protection and Affordable Care Act of 2010, as amended by the Health Care and Education Reconciliation Act of 2010 (collectively, the ACA), as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we may receive for any of our approved products. The ACA, among other things, expanded and increased industry rebates for drugs covered by Medicaid, made changes to the coverage requirements under Medicare Part D, Medicare’s prescription drug benefits program and broadened access to health insurance. There have been legal and political challenges and amendments to certain aspects of the ACA.
For example, on August 16, 2022, the Inflation Reduction Act of 2022 (IRA) was signed into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost and through a newly established manufacturer discount program. It is possible that the ACA will be subject to additional challenges in the future. It is unclear how any such challenges and additional healthcare reform measures of the current administration will impact the ACA and our business.
Other legislative changes have been proposed and adopted in the U.S. since the ACA. Through the process created by the Budget Control Act of 2011, there are automatic reductions of Medicare payments to providers up to 2% per fiscal year, which went into effect in April 2013 and, due to subsequent legislative amendments, including the Infrastructure Investment and Jobs Act and the Consolidated Appropriations Act of 2023, will remain in effect through 2032 unless additional Congressional action is taken.
An expansion in the government’s role in the U.S. healthcare industry may increase existing congressional or governmental agency scrutiny on price increases, such as the ones we have implemented for NUPLAZID, cause general downward pressure on the prices of prescription drug products, lower reimbursements for providers using our products, reduce product utilization and adversely affect our business and results of operations. There have been several recent U.S. presidential executive orders, Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, reduce the cost of drugs under Medicare, and reform government program reimbursement methodologies for drugs. For example, the IRA, among other things, (1) directs the U.S. Department of Health and Human Services (HHS) to negotiate the price of certain single-source drugs that have been on the market for at least 7 years covered under Medicare (the Medicare Drug Price Negotiation Program) and (2) imposes rebates under Medicare Part B and Medicare Part D to penalize price increases that outpace inflation. HHS has and will continue to issue and update guidance as these programs are implemented. These provisions began to take effect progressively in fiscal year 2023. On August 15, 2024, HHS announced the agreed-upon reimbursement prices of the first ten drugs that were subject to price negotiations, although the Medicare Drug Price Negotiation Program is currently subject to legal challenges. HHS will select up to fifteen additional drugs covered under Part D for negotiation in 2025. Each year thereafter more Part B and Part D products will become subject to the Medicare Drug Price Negotiation Program. On December 7, 2023, an initiative to control the price of prescription drugs through the use of march-in rights under the Bayh-Dole Act was announced. On December 8, 2023, the National Institute of Standards and Technology published for comment a Draft Interagency Guidance Framework for Considering the Exercise of March-In Rights which for the first time includes the price of a product as one factor an agency can use when deciding to exercise march-in rights. While march-in rights have not previously been exercised, it is uncertain if that will continue under the new framework. Further, the overall funding of certain government programs such as Medicaid and Medicare is uncertain and there is no guarantee that funds approved by the U.S. Congress will be made available by the current administration. We expect additional health reform measures may be implemented in the future, particularly in light of the recent U.S. Presidential and Congressional elections.
Individual states in the U.S. have also increasingly passed legislation and implemented regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. For example, on January 5, 2024, the FDA approved Florida’s proposal to import certain drugs from Canada for specific state healthcare programs. It is unclear if and how this program will be implemented and whether it will be subject challenges in the United States or Canada. Other states have also submitted proposals that are pending review by the FDA. Any such approved importation plans, if implemented, may result in lower drug prices for products covered by those programs.
The implementation of cost-containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our products.
We are subject, directly and indirectly, to federal, state and foreign healthcare laws and regulations, including healthcare fraud and abuse laws, false claims laws, physician payment transparency laws and health information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
Our operations are directly, and indirectly through our customers and third-party payors, subject to various U.S. federal and state healthcare laws and regulations, including, without limitation, the U.S. federal Anti-Kickback Statute, the U.S. federal False Claims Act, and physician payment sunshine laws and regulations. These laws may impact, among other things, our clinical research, sales, marketing, grants, charitable donations, and education programs and constrain the business or financial arrangements with healthcare providers, physicians, charitable foundations that support Parkinson’s disease patients generally, and other parties that have the ability to directly or indirectly influence the prescribing, ordering, marketing, or distribution of our products for which we obtain marketing approval. In addition, we and any current or potential future collaborators, partners or service providers are or may become subject to data privacy and security regulation by both the U.S. federal government and the states in which we conduct our business, including laws and regulations that apply to our processing of personal data or the processing of personal data on our behalf. Finally, we may be subject to additional healthcare, statutory and regulatory requirements and enforcement by foreign regulatory authorities in jurisdictions in which we conduct our business. The laws that may affect our ability to operate include:
•the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, offering, receiving or paying any remuneration (including any kickback, bribe, or certain rebates), directly or indirectly, overtly or covertly, in cash or in kind, to induce, or in return for, either the referral of an individual, or the purchase, lease, order or recommendation of any good, facility, item or service, for which payment may be made, in whole or in part, under U.S. federal and state healthcare programs such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
•the U.S. federal civil and criminal false claims laws, including the civil False Claims Act, which can be enforced through civil whistleblower or qui tam actions, and civil monetary penalties laws, which impose criminal and civil penalties on individuals or entities for, among other things, knowingly presenting, or causing to be presented to the U.S. federal government, claims for payment or approval that are false or fraudulent or from knowingly making a false statement to avoid, decrease or conceal an obligation to pay money to the U.S. federal government. In addition, the government may assert that a claim including items and services resulting from a violation of the U.S. federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act;
•the U.S. federal Health Insurance Portability and Accountability Act of 1996 (HIPAA), which imposes criminal and civil liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statement, in connection with the delivery of, or payment for, healthcare benefits, items or services. Similar to the U.S. federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
•HIPAA, and its implementing regulations, and as amended again by the Final HIPAA Omnibus Rule, Modifications to the HIPAA Privacy, Security, Enforcement and Breach Notification Rules Under the Health Information Technology for Economic and Clinical Health Act (HITECH) and the Genetic Information Nondiscrimination Act; Other Modifications to the HIPAA Rules, published in January 2013, which imposes certain obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information on covered entities subject to the rule, such as health plans, healthcare clearinghouses and certain healthcare providers as well as their business associates, individuals or entities that perform certain services involving the use or disclosure of individually identifiable health information on behalf of a covered entity and their subcontractors that use, disclose or otherwise process individually identifiable health information;
•the U.S. Federal Food, Drug and Cosmetic Act (FDCA), which prohibits, among other things, the adulteration or misbranding of drugs, biologics and medical devices;
•the U.S. federal physician payment transparency requirements, sometimes referred to as the “Physician Payments Sunshine Act”, which was enacted as part of the ACA and its implementing regulations and requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program to report annually to the CMS information related to certain payments and other transfers of value made to physicians (as defined to include doctors of medicine, dentists, optometrists, podiatrists and chiropractors under such law), other healthcare professionals (such as physician assistants and nurse practitioners), and teaching hospitals, as well as information regarding ownership and investment interests held by physicians and their immediate family members; and
•analogous state and local laws and regulations, including: state anti-kickback and false claims laws, which may apply to our business practices, including but not limited to, research, distribution, sales and marketing arrangements and claims involving healthcare items or services reimbursed by any third-party payor, including private insurers; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the U.S. federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state and local laws and regulations that require drug manufacturers to file reports relating to pricing and marketing information, which requires tracking gifts and other remuneration and items of value provided to healthcare professionals and entities and/or the registration of pharmaceutical sales representatives; and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations could involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws and regulations. For example, contributions to third-party charitable foundations are a current area of significant governmental and congressional scrutiny, and we could face action if a federal or state governmental authority were to conclude that our charitable contributions to foundations that support Parkinson’s disease patients generally are not compliant. If our operations are found to be in violation of any of the laws described above or any other governmental laws and regulations that may apply to us, we may be subject to significant penalties, including civil, criminal and administrative penalties, damages, fines, exclusion from U.S. government-funded healthcare programs, such as Medicare and Medicaid, disgorgement, imprisonment, contractual damages, reputational harm, diminished profits, additional reporting requirements and/or oversight, and the curtailment or restructuring of our operations. Moreover, while we do not bill third-party payors directly and our customers make the ultimate decision on how to submit claims, from time-to-time, for our products, we may provide reimbursement guidance to patients and healthcare providers. If a government authority were to conclude that we provided improper advice and/or encouraged the submission of a false claim for reimbursement, we could face action against us by government authorities. If any of the physicians, healthcare professions, or other providers or entities with whom we expect to do business is found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government-funded healthcare programs and imprisonment. If any of the above occur, it could adversely affect our ability to operate our business and our results of operations.
Outside the U.S., interactions between pharmaceutical companies and health care professionals are also governed by strict laws, such as national anti-bribery laws of European countries, national sunshine rules, regulations, industry self-regulation codes of conduct and physicians’ codes of professional conduct. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment, any of which could adversely affect our ability to operate our business and our results of operations.
We and the third parties with whom we work are subject to stringent and evolving U.S. and foreign laws, regulations and rules, contractual obligations, industry standards, policies and other obligations related to data privacy and security. Our (or the third parties with whom we work) actual or perceived failure to comply with such obligations could lead to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; and other adverse business consequences.
In the ordinary course of business, we collect, receive, store, process, generate, use, transfer, disclose, make accessible, protect, secure, dispose of, transmit, and share (collectively, process) personal data and other sensitive information, including proprietary and confidential business data, trade secrets, intellectual property, data we collect about trial participants in connection with clinical trials, sensitive third-party data, business plans, transactions, financial information and medical information collected by our patient access management team (collectively, sensitive data). Our data processing activities may subject us to numerous data privacy and security obligations, such as various laws, regulations, guidance, industry standards, external and internal privacy and security policies, contractual requirements, and other obligations relating to data privacy and security.
In the United States, federal, state, and local governments have enacted numerous data privacy and security laws, including data breach notification laws, personal data privacy laws, consumer protection laws (e.g., Section 5 of the Federal Trade Commission Act), and other similar laws (e.g., wiretapping laws). For example, HIPAA, as amended by HITECH, imposes specific requirements relating to the privacy, security, and transmission of individually identifiable health information. Additionally, numerous U.S. states have enacted comprehensive privacy laws that impose certain obligations on covered businesses, including providing specific disclosures in privacy notices and affording residents with certain rights concerning their personal data. As applicable, such rights may include the right to access, correct, or delete certain personal data, and to opt-out of certain data processing activities, such as targeted advertising, profiling, and automated decision-making. The exercise of these rights may impact our business and ability to provide our products and services. Certain states also impose stricter requirements for processing certain personal data, including sensitive information, such as conducting data privacy impact assessments. These state laws allow for statutory fines for noncompliance. For example, the California Consumer Privacy Act of 2018 (CCPA) requires businesses to provide specific disclosures in privacy notices and honor requests of California residents to exercise certain privacy rights. The CCPA provides for fines for intentional violations and allows private litigants affected by certain data breaches to recover significant statutory damages. Although some U.S. comprehensive privacy laws exempt some data processed in the context of clinical trials, these laws may increase compliance costs and potential liability with respect to other personal data we may maintain about California residents. Similar laws are being considered in several other states, as well as at the federal and local levels, and we expect more jurisdictions to pass similar laws in the future.
Outside the United States, an increasing number of laws, regulations, and industry standards may govern data privacy and security. For example, the European Union’s General Data Protection Regulation (EU GDPR), United Kingdom’s GDPR (UK GDPR) (collectively, the GDPR), Brazil’s General Data Protection Law (Lei Geral de Proteção de Dados Pessoais, or LGPD) (Law No. 13,709/2018), and China’s Personal Information Protection Law (PIPL) impose strict requirements for processing personal data. For example, under the GDPR, companies may face temporary or definitive bans on data processing and other corrective actions; fines of up to 20 million Euros under the EU GDPR / 17.5 million pounds sterling under the UK GDPR or 4% of annual global revenue, whichever is greater; or private litigation related to processing of personal data brought by classes of data subjects or consumer protection organizations authorized at law to represent their interests.
The Swiss Federal Act on Data Protection, or the FADP, also applies to the collection and processing of personal data, including health-related information, by companies located in Switzerland, or in certain circumstances, by companies located outside of Switzerland.
In addition, we may be unable to transfer personal data from Europe and other jurisdictions to the United States or other countries due to data localization requirements or limitations on cross-border data flows. Europe and other jurisdictions have enacted laws requiring data to be localized or limiting the transfer of personal data to other countries. In particular, the European Economic Area (EEA) and the UK have significantly restricted the transfer of personal data to the United States and other countries whose privacy laws it believes are inadequate. Other jurisdictions have adopted and may continue to adopt similarly stringent data localization and cross-border data transfer laws. Although there are currently various mechanisms that may be used to transfer personal data from the EEA and UK to the United States in compliance with law, such as the EEA standard contractual clauses, the UK’s International Data Transfer Agreement / Addendum, and the EU-U.S. Data Privacy Framework and the UK extension thereto (which allows for transfers to relevant U.S.-based organizations who self-certify compliance and participate in the Framework), these mechanisms are subject to legal challenges, and there is no
assurance that we can satisfy or rely on these measures to lawfully transfer personal data to the United States. If there is no lawful manner for us to transfer personal data from the EEA, the UK, or other jurisdictions to the United States, or if the requirements for a legally-compliant transfer are too onerous, we could face significant adverse consequences, including by limiting our ability to conduct clinical trial activities in Europe and elsewhere, the interruption or degradation of our operations, the need to relocate part of or all of our business or data processing activities to other jurisdictions (such as Europe) at significant expense, increased exposure to regulatory actions, substantial fines and penalties, the inability to transfer data and work with partners, vendors and other third parties, and injunctions against our processing or transferring of personal data necessary to operate our business. Some European regulators have ordered certain companies to suspend or permanently cease certain transfers of personal data to recipients outside Europe for allegedly violating the GDPR’s cross-border data transfer limitations. Additionally, companies that transfer personal data to recipients outside of the EEA and/or UK to other jurisdictions, particularly to the United States, are subject to increased scrutiny from regulators individual litigants and activist groups.
Our employees and personnel use generative artificial intelligence (AI) technologies to perform their work, and the disclosure and use of personal data in generative AI technologies is subject to various privacy laws and other privacy obligations. Governments have passed and are likely to pass additional laws regulating generative AI. Our use of this technology could result in additional compliance costs, regulatory investigations and actions, and lawsuits. If we are unable to use generative AI, it could make our business less efficient and result in competitive disadvantages.
In addition to data privacy and security laws, we may be contractually subject to industry standards adopted by industry groups and may become subject to additional such obligations in the future. We are also bound by other contractual obligations related to data privacy and security, and our efforts to comply with such obligations may not be successful. We publish privacy policies, marketing materials, and other statements, such as statements related to compliance with certain certifications or self-regulatory principles, regarding artificial intelligence, data privacy and security. Regulators in the United States are increasingly scrutinizing these statements, and if these policies, materials or statements are found to be deficient, lacking in transparency, deceptive, unfair, misleading or misrepresentative of our practices, we may be subject to investigation, enforcement actions by regulators, or other adverse consequences.
Additionally, under various privacy laws and other obligations, we may be required to obtain certain consents to process personal data. For example, some of our data processing practices may be challenged under wiretapping laws, if we obtain consumer information from third parties through various methods, including chatbot and session replay providers, or via third-party marketing pixels. These practices may be subject to increased challenges by class action plaintiffs. Our inability or failure to obtain consent for these practices could result in adverse consequences, including class action litigation and mass arbitration demands.
Obligations related to data privacy and security (and consumers’ data privacy expectations) are quickly changing, becoming increasingly stringent, and creating uncertainty. Additionally, these obligations may be subject to differing applications and interpretations, which may be inconsistent or conflict among jurisdictions. Preparing for and complying with these obligations requires us to devote significant resources and may necessitate changes to our services, information technologies, systems, and practices and to those of any third parties that process personal data on our behalf.
We may at times fail (or be perceived to have failed) in our efforts to comply with our data privacy and security obligations. Moreover, despite our efforts, our personnel or third parties with whom we work may fail to comply with such obligations, which could negatively impact our business operations. If we or the third parties with whom we work fail, or are perceived to have failed, to address or comply with applicable data privacy and security obligations, we could face significant consequences, including but not limited to: government enforcement actions (e.g., investigations, fines, penalties, audits, inspections, and similar); litigation (including class-action claims) and mass arbitration demands; additional reporting requirements and/or oversight; bans or restrictions on processing personal data; and orders to destroy or not use personal data. In particular, plaintiffs have become increasingly more active in bringing privacy-related claims against companies, including class claims. Some of these claims allow for the recovery of statutory damages on a per violation basis, and, if viable, carry the potential for monumental statutory damages, depending on the volume of data and the number of violations. Any of these events could have a material adverse effect on our reputation, business, or financial condition, including but not limited to loss of customers; inability to process personal data or to operate in certain jurisdictions; limited ability to develop or commercialize our products; expenditure of time and resources to defend any claim or inquiry; adverse publicity; or substantial changes to our business model or operations.
If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate Program or other governmental pricing programs in the U.S., we could be subject to additional reimbursement requirements, fines, sanctions and exposure under other laws which could have a material adverse effect on our business, results of operations and financial condition.
We participate in the Medicaid Drug Rebate Program, as administered by CMS, and other federal and state government pricing programs in the U.S., and we may participate in additional government pricing programs in the future. These programs generally require us to pay rebates or otherwise provide discounts to government payors in connection with drugs that are dispensed to beneficiaries/recipients of these programs. In some cases, such as with the Medicaid Drug Rebate Program, the rebates are based on pricing that we report on a monthly and quarterly basis to the government agencies that administer the programs. Pricing requirements and rebate/discount calculations are complex, vary among products and programs, and are often subject to interpretation by governmental or regulatory agencies and the courts. The requirements of these programs, including, by way of example, their respective terms and scope, change frequently. For example, American Rescue Plan Act of 2021 eliminated the statutory Medicaid drug rebate cap, previously set at 100% of a drug’s average manufacturer price (AMP), for single source and innovator multiple source drugs, effective January 1, 2024. Responding to current and future changes may increase our costs, and the complexity of compliance will be time consuming. Invoicing for rebates is provided in arrears, and there is frequently a time lag of up to several months between the sales to which rebate notices relate and our receipt of those notices, which further complicates our ability to accurately estimate and accrue for rebates related to the Medicaid program as implemented by individual states. Thus, we may not be able to identify all factors that may cause our discount and rebate payment obligations to vary from period to period, and our actual results may differ significantly from our estimated allowances for discounts and rebates. Changes in estimates and assumptions may have a material adverse effect on our business, results of operations and financial condition.
In addition, the HHS Office of Inspector General and other Congressional, enforcement and administrative bodies have recently increased their focus on pricing requirements for products, including, but not limited to the methodologies used by manufacturers to calculate AMP, and best price (BP), for compliance with reporting requirements under the Medicaid Drug Rebate Program. We are liable for errors associated with our submission of pricing data and for any overcharging of government payors. For example, failure to submit monthly/quarterly AMP and BP data on a timely basis could result in significant civil monetary penalties for each day the submission is late beyond the due date. Failure to make necessary disclosures and/or to identify overpayments could result in allegations against us under the civil False Claims Act and other laws and regulations. Any required refunds to the U.S. government or responding to a government investigation or enforcement action would be expensive and time consuming and could have a material adverse effect on our business, results of operations and financial condition. In addition, in the event that the CMS were to terminate our rebate agreement, no federal payments would be available under Medicaid or Medicare for our covered outpatient drugs.
We could face liability if a regulatory authority determines that we are promoting our products for any “off-label” uses.
The FDA, Health Canada, the European Commission, competent authorities of individual EU Member States and other comparable foreign regulatory authorities and industry self-regulatory bodies strictly regulate the marketing and promotional claims that are made about drug and biologic products. In particular, a company may not promote “off-label” uses for its drug products. An off-label use is the use of a product for an indication, patient population, or manner that is not described in the product’s approved labeling and that differs from those approved by the applicable regulatory authorities. Physicians and other persons qualified to prescribe medicinal products, on the other hand, may, in certain jurisdictions including the U.S., prescribe products for off-label uses. Although the FDA and certain comparable foreign regulatory authorities do not generally regulate a physician’s or other person qualified to prescribe’s choice of drug treatment made in the such person’s independent medical judgment, they do restrict promotional communications from pharmaceutical companies or their sales force with respect to off-label uses of products for which marketing clearance has not been issued. A company that is found to have promoted off-label use of its product may be subject to significant liability, including civil and criminal sanctions.
We intend to comply with the requirements and restrictions of the FDA, Health Canada and other comparable foreign regulatory authorities, governmental authorities and regulatory bodies in the jurisdictions that approve our products or product candidates with respect to our promotion of our products, but such authorities may nevertheless make us the target of an investigation or prosecution based on our marketing and promotional practices. As a result, we may be subject to criminal and civil liability for the promotion of off-label uses. In addition, our management’s attention could be diverted to handle any such alleged violations, all of which could have a material adverse effect on our business, results of operations, financial condition and reputation.
A significant number of pharmaceutical companies have been the target of inquiries and investigations by various U.S. federal and state regulatory, investigative, prosecutorial and administrative entities in connection with the promotion of products for unapproved uses and other sales practices, including by the Department of Justice (DOJ), and various U.S. Attorneys’ Offices, the HHS Office of Inspector General, the FDA, the Federal Trade Commission and various state Attorneys General offices. These investigations have alleged violations of various U.S. federal and state laws and regulations, including claims asserting antitrust violations, violations of the FDCA, the civil False Claims Act, the Prescription Drug Marketing Act, anti-kickback laws, and other alleged violations in connection with the promotion of products for unapproved uses, pricing and Medicare and/or Medicaid reimbursement. If the FDA, DOJ, or any other governmental agency initiates an enforcement action against us, or if we are the subject of a qui tam suit and it is determined that we violated prohibitions relating to the promotion of products for unapproved uses, we could be subject to substantial civil or criminal fines or damage awards and other sanctions such as consent decrees and corporate integrity agreements pursuant to which our activities would be subject to ongoing scrutiny and monitoring to ensure compliance with applicable laws and regulations. Any such fines, awards or other sanctions would have an adverse effect on our revenue, business, financial prospects, and reputation.
In the EU, the advertising and promotion of medicinal products are subject to both EU and EU Member States’ laws governing promotion of medicinal products, interactions with physicians and other healthcare professionals, misleading and comparative advertising and unfair commercial practices. General requirements for advertising and promotion of medicinal products, such as direct-to-consumer advertising of prescription medicinal products, are established in EU law. However, the details are governed by regulations in individual EU Member States and can differ from one country to another. If the EU or an applicable EU Member State were to determine that we violated an applicable law or regulation, we could be subject to lawsuits, regulatory actions, penalties and other adverse consequences that would have an adverse effect on our revenue, business, financial prospects, and reputation.
Changes at the FDA and other government agencies could delay or prevent new products from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept payment of user fees, layoffs and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, including executive and congressional priorities, the impacts of which are inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical government employees and stop critical activities. In addition, the current administration has proposed substantial reductions in force at various government agencies that, if applied in a material way, could significantly reduce the FDA’s and other agencies’ capacities to perform their functions in a manner consistent with past practices and could negatively impact our business. If repeated or prolonged government shutdowns or material layoffs of agency personnel occur, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, and negatively impact other government operations on which we rely, which could have a material adverse effect on our business.
We are subject to stringent regulation in connection with the marketing of our products, which could delay the development and commercialization of our products.
The pharmaceutical industry is subject to stringent regulation by the FDA and other regulatory agencies in the U.S. and by comparable foreign regulatory authorities in other jurisdictions. Neither we nor our collaborators can market a pharmaceutical product in the U.S. until it has completed rigorous preclinical testing and clinical trials and an extensive regulatory clearance process implemented by the FDA. Satisfaction of regulatory requirements typically takes many years, depends upon the type, complexity and novelty of the product, and requires substantial resources. Even if regulatory approval is obtained, the FDA and comparable foreign regulatory authorities may impose significant restrictions on the indicated uses, conditions for use, labeling, advertising, promotion, and/or marketing of such products, and requirements for post-approval studies, including additional research and development and clinical trials. These limitations may limit the size of the market for the product or result in the incurrence of additional costs. Any delay or failure in obtaining required approvals could have a material adverse effect on our ability to generate revenues from the particular product candidate, if approved.
Outside the U.S., the ability to market a product is contingent upon receiving approval from the appropriate regulatory authorities. The requirements governing the conduct of clinical trials, marketing authorization, pricing, and reimbursement vary widely from country to country. Only after the appropriate regulatory authority is satisfied that adequate evidence of safety, quality, and efficacy has been presented will it grant a marketing authorization. Approval by the FDA does not automatically lead to the approval by regulatory authorities outside the U.S. and, similarly, approval by regulatory authorities outside the U.S. will not automatically lead to FDA approval.
In addition, U.S. and foreign government regulations control access to and use of some human or other tissue samples in our research and development efforts. U.S. and foreign government agencies may also impose restrictions on the use of data derived from human or other tissue samples. Accordingly, if we fail to comply with these regulations and restrictions, the commercialization of our product candidates, if approved, may be delayed or suspended, which may delay or impede our ability to generate product revenues.
If our competitors develop and market products that are more effective than our products, they may reduce or eliminate our commercial opportunity.
Competition in the pharmaceutical and biotechnology industries is intense and expected to increase. We face, and will continue to face, intense competition from pharmaceutical and biotechnology companies, as well as numerous academic and research institutions and governmental agencies, both in the U.S. and abroad. We compete, or will compete, with existing and new products being developed by our competitors. Some of these competitors have products or are pursuing the development of pharmaceuticals that target the same diseases and conditions that our research and development programs target.
For example, the use of NUPLAZID for the treatment of PDP competes with off-label use of various antipsychotic drugs, including the generic drugs quetiapine, clozapine, risperidone, aripiprazole, and olanzapine. In addition, Anavex has a product, Anavex 2-73, in development for the potential treatment of Rett syndrome and Taysha Gene Therapies and Neurogene are conducting Phase 1/2 clinical trials of gene therapies to treat Rett syndrome. Several academic institutions and pharmaceutical companies are currently conducting clinical trials for the treatment of various symptoms of Rett syndrome, including Unravel Bio and Vanderbilt University Medical Center, which are jointly conducting an early stage study with vorinostat (RVL-001).
Other competitors may have a variety of drugs in development or awaiting approval from the FDA or comparable foreign regulatory authorities that could reach the market and become established before we have a product to sell for the applicable disorder. Our competitors may also develop alternative therapies that could further limit the market for any drugs that we may develop. Many of our competitors are using technologies or methods different or similar to ours to identify and validate drug targets and to discover novel small molecule drugs. Many of our competitors and their collaborators have significantly greater experience than we do in the following:
•identifying and validating targets;
•screening compounds against targets;
•preclinical studies and clinical trials of potential pharmaceutical products;
•obtaining FDA and other regulatory approvals; and
•commercializing pharmaceutical products.
In addition, many of our competitors and their collaborators have substantially greater advantages in the following areas: capital resources, research and development resources, manufacturing capabilities, sales and marketing, and production facilities. Smaller companies also may prove to be significant competitors, particularly through proprietary research discoveries and collaboration arrangements with large pharmaceutical and established biotechnology companies. Many of our competitors have products that have been approved or are in advanced development and may develop superior technologies or methods to identify and validate drug targets and to discover novel small molecule drugs. Our competitors, either alone or with their collaborators, may succeed in developing technologies or drugs that are more effective, safer, more affordable, or more easily administered than ours and may achieve patent protection or commercialize drugs sooner than us. Our competitors may also develop alternative therapies that could further limit the market for any drugs that we may develop. Our failure to compete effectively could have a material adverse effect on our business.
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit commercialization of our products, or development or commercialization of our product candidates, if approved.
We face an inherent risk of product liability as a result of the commercial sales of our products and the clinical testing of our product candidates. For example, we may be sued if any of our products allegedly cause injury or are found to be otherwise unsuitable for administration in humans. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability or a breach of warranties. Claims could also be asserted under state consumer protection acts. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our product candidates, if approved. Even successful defense would require significant financial and management resources. Regardless of the merits or eventual outcome, liability claims may result in:
•decreased demand for our products or product candidates, if approved;
•injury to our reputation;
•withdrawal of clinical trial participants;
•initiation of investigations by regulators;
•costs to defend the related litigation;
•a diversion of management’s time and our resources;
•substantial monetary awards to trial participants or patients;
•product recalls, withdrawals or labeling, marketing or promotional restrictions;
•exhaustion of any available insurance and our capital resources;
•the inability to commercialize our products or product candidates, if approved; and
•a decline in our stock price.
Although we currently have product liability insurance that covers our clinical trials and the commercialization of our products, we may need to increase and expand this coverage, including if we commence larger scale trials and if our product candidates are approved for commercial sale. This insurance may be prohibitively expensive or may not fully cover our potential liabilities. Inability to obtain sufficient insurance coverage at an acceptable cost or otherwise to protect against potential product liability claims could prevent or inhibit the commercialization of products that we or our collaborators develop. If we determine that it is prudent to increase our product liability coverage, we may be unable to obtain such increased coverage on acceptable terms or at all. Our insurance policies also have various exclusions, and we may be subject to a product liability claim for which we have no coverage. Our liability could exceed our total assets if we do not prevail in a lawsuit from any injury caused by our drug products. Product liability claims could have a material adverse effect on our business and results of operations.
If our information technology systems or data, or those of third parties with whom we work, are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions, interruptions to operations or clinical trials, reputational harm, litigation, fines and penalties, disruptions of our business operations, and a loss of customers or sales.
In the ordinary course of our business, we, or the third parties with whom we work, process proprietary, confidential, and sensitive data, including personal data (such as health-related data), intellectual property, and trade secrets.
Cyberattacks, malicious internet-based activity, online and offline fraud and other similar activities threaten the confidentiality, integrity, and availability of our sensitive data and information technology systems, and those of the third parties with whom we work. These threats are prevalent, continue to rise, and are becoming increasingly difficult to detect. These threats come from a variety of sources, including traditional computer “hackers,” hacktivists, threat actors, personnel misconduct or error (such as through theft or misuse), organized criminal threat actors, sophisticated nation-states, and nation-state-supported actors. Some actors now engage and are expected to continue to engage in cyber-attacks, including without limitation nation-state actors for geopolitical reasons and in conjunction with military conflicts and defense activities. During times of war and other major conflicts, we and the third parties with whom we work may be vulnerable to a
heightened risk of these attacks, including retaliatory cyber-attacks, that could materially disrupt our systems and operations, supply chain, and ability to produce, sell and distribute our goods and services.
We and the third parties with whom we work are subject to a variety of evolving threats, including but not limited to, social engineering attacks (including through deep fakes, which may be increasingly more difficult to identify as fake, and phishing attacks), malicious code (such as viruses and worms), malware (including as a result of advanced persistent threat intrusions), denial-of-service attacks, credential stuffing, credential harvesting, personnel misconduct or error, ransomware attacks, supply-chain attacks, software bugs, server malfunction, software or hardware failures, loss of data or other information technology assets, adware, attacks enhanced or facilitated by AI, telecommunications failures, earthquakes, fire, flood, and other similar threats.
Ransomware attacks, including by organized criminal threat actors, nation-states, and nation-state-supported actors, are becoming increasingly prevalent and severe and can lead to significant interruptions, delays, or outages in our operations, disruption of clinical trials or otherwise affecting our ability to provide our products or product candidates, loss of sensitive data (including data related to clinical trials) and income, significant extra expenses to restore data or systems, reputational harm and the diversion of funds. Extortion payments may alleviate the negative impact of a ransomware attack, but we may be unwilling or unable to make such payments (including, for example, if applicable laws or regulations prohibit such payments). Remote work has increased risks to our information technology systems and data, as our employees work from home, utilizing network connections, computers and devices outside our premises, including at home, while in transit or in public locations. Future or past business transactions (such as acquisitions or integrations) could expose us to additional cybersecurity risks and vulnerabilities, as our systems could be negatively affected by vulnerabilities present in acquired or integrated entities’ systems and technologies. Furthermore, we may discover security issues that were not found during due diligence of such acquired or integrated entities, and it may be difficult to integrate companies into our information technology environment and security program.
We rely on third-party service providers and technologies to operate critical business systems to process sensitive data in a variety of contexts, including, without limitation, cloud-based infrastructure, drug suppliers, data center facilities, encryption and authentication technology, employee email, content delivery to customers, and other functions. Our ability to monitor these third parties’ information security practices and posture (including whether any unremediated vulnerabilities exist or have been exploited) is limited, and these third parties may not have adequate information security measures in place. In addition, supply-chain attacks have increased in frequency and severity, and we cannot guarantee that third parties’ infrastructure in our supply chain or our third-party partners’ supply chains have not been compromised. For example, we were made aware of a cyberattack against one of the largest prescription processors in the country in February 2024 that impacted the ability for our specialty pharmacy partners to have payors provide authorizations for patient refills and new patient starts for certain of our products. In April 2024, we were notified by a third-party patient support service provider of a data security incident that involved personal data of NUPLAZID patients.
It may be difficult and/or costly to detect, investigate, mitigate, contain and remediate a security incident. Our efforts to investigate, mitigate, contain and remediate a security incident may not be successful. Actions taken by us or the third parties with whom we work to detect, investigate, mitigate, contain and remediate a security incident could result in outages, data losses and disruptions of our business. Threat actors may also gain access to other networks and systems after a compromise of our networks and systems.
While we have implemented security measures designed to protect against security incidents, there can be no assurance that these measures will be effective. We take steps designed to detect, mitigate, and remediate vulnerabilities in our information security systems (such as our hardware and/or software, including that of third parties with whom we work). We and the third parties with whom we work may not, however, detect and remediate all such vulnerabilities including on a timely basis. For example, we have identified certain vulnerabilities in our information systems, and we take steps designed to mitigate the risks associated with known vulnerabilities. These steps include implementing compensating controls and other protective measures. Further, we and the third parties with whom we work may experience delays in developing and deploying remedial measures and patches designed to address identified vulnerabilities. Vulnerabilities could be exploited and result in a security incident.
Any of the previously identified or similar threats could cause, and in some cases have in the past caused, a security incident or other interruption that could result in unauthorized, unlawful, or accidental acquisition, modification, destruction, loss, alteration, encryption, disclosure of, or access to our sensitive information or our information technology systems, or those of the third parties with whom we work. A security incident or other interruption could disrupt our ability (and that of third parties with whom we work) to provide our products.
We may expend significant resources or fundamentally change our business activities and practices (including our clinical trials) to try to protect against security incidents. Certain data privacy and security obligations may require us to implement and maintain specific security measures or industry-standard or reasonable security measures to protect our information technology systems and sensitive data.
Applicable data privacy and security obligations may require us, or we may choose, to notify relevant stakeholders, including affected individuals, customers, regulators, and investors, of security incidents, or to implement other requirements, such as providing credit monitoring or identity theft protection services. Such disclosures and related actions are costly, and the disclosure or the failure to comply with applicable requirements could lead to adverse consequences. If we (or a third party with whom we work) experience a security incident or are perceived to have experienced a security incident, we may experience adverse consequences. These consequences may include: government enforcement actions (for example, investigations, fines, penalties, audits, and inspections); additional reporting requirements and/or oversight; restrictions on processing sensitive data (including personal data); litigation (including class claims); indemnification obligations; negative publicity; reputational harm; monetary fund diversions; diversion of management attention; interruptions in our operations (including availability of data); financial loss; and other similar harms. Security incidents and attendant consequences may prevent or cause customers to stop using our products, deter new customers from using our products, and negatively impact our ability to grow and operate our business.
Our contracts may not contain limitations of liability, and even where they do, there can be no assurance that limitations of liability in our contracts are sufficient to protect us from liabilities, damages, or claims related to our data privacy and security obligations.
In addition, our insurance coverage may not be adequate or sufficient in type or amount to protect us from or to mitigate liabilities arising out of our privacy and security practices. The successful assertion of one or more large claims against us that exceeds our available insurance coverage, or results in changes to our insurance policies (including premium increases or the imposition of large deductible or co-insurance requirements), could have an adverse effect on our business.
In addition to experiencing a security incident, third parties may gather, collect, or infer sensitive data about us from public sources, data brokers or other means that reveals competitively sensitive details about our organization and could be used to undermine our competitive advantage or market position. Additionally, sensitive information of the Company could be leaked, disclosed, or revealed as a result of or in connection with our employees’, personnel’s, or vendors’ use of generative AI technologies.
Risks Related to Our Common Stock
Our stock price historically has been, and is likely to remain, highly volatile.
The market prices for securities of biotechnology companies in general, and drug discovery and development companies in particular, have been highly volatile and may continue to be highly volatile in the future. From the period between January 2, 2024 to February 18, 2025, the closing price of our common stock has ranged from a low of $14.29 per share to a high of $30.86 per share. Furthermore, especially as we and our market capitalization have grown, the price of our common stock has been increasingly affected by quarterly and annual comparisons with the valuations and recommendations of the analysts who cover our business. The following factors, in addition to other risk factors described in this section, may have a significant impact on the market price of our common stock:
•the success of our commercialization of our products;
•the status and cost of development and commercialization of our products and product candidates, if approved, including compounds being developed under our collaborations;
•whether we acquire or in-license additional product candidates or products, and the status of development and commercialization of such product candidates, if approved, or products;
•the status and cost of development and commercialization of trofinetide for indications other than Rett syndrome and in jurisdictions outside North America;
•any other communications or guidance from the FDA or other regulatory authorities that pertain to our products or product candidates;
•the status and cost of our PMRs for DAYBUE;
•the initiation, termination, or reduction in the scope of our collaborations or any disputes or developments regarding our collaborations;
•market conditions or trends related to biotechnology and pharmaceutical industries, or the market in general;
•announcements of technological innovations, new products, or other material events by our competitors or us, including any new products that we may acquire or in-license;
•disputes or other developments concerning our proprietary and intellectual property rights;
•fluctuations in our operating results;
•changes in, or failure to meet, securities analysts’ or investors’ expectations of our financial performance;
•our failure to meet applicable Nasdaq listing standards and the possible delisting of our common stock from the Nasdaq Stock Market;
•additions or departures of key personnel;
•discussions of our business, products, financial performance, prospects, or stock price by the financial and scientific press and online investor communities such as blogs and chat rooms;
•public concern as to, and legislative action with respect to, genetic testing or other research areas of biopharmaceutical companies, the pricing and availability of prescription drugs, or the safety of drugs and drug delivery techniques;
•regulatory developments in the U.S. and in foreign countries;
•changes in the structure of healthcare payment systems;
•the announcement of, or developments in, any litigation matters;
•disruptions caused by geopolitical or macroeconomic developments or other business interruptions, as well as any related political or economic responses and counter-responses or otherwise by various global actors or the general effect on the global economy and supply chain; and
•economic and political factors, including but not limited to economic and financial crises, wars, terrorism, and political unrest.
In the past, following periods of volatility in the market price of a particular company’s securities, securities class action litigation has often been brought against that company. For example, we, and certain of our current and former officers and directors, are subject to numerous lawsuits related to prior statements about NUPLAZID and our sNDA seeking approval of pimavanserin for the treatment of hallucinations and delusions associated with DRP, as described in “Legal Proceedings”. If we are not successful in defense of these claims, we may have to make significant payments to, or other settlements with, our stockholders and their attorneys. Even if such claims are not successful, the litigation has resulted in additional costs in the past and could result in further substantial costs and diversion of our management’s attention and resources in the future, which could have a material adverse effect on our business, operating results or financial condition.
If we or our stockholders sell substantial amounts of our common stock, the market price of our common stock may decline.
A significant number of shares of our common stock are held by a small number of stockholders. Sales of a significant number of shares of our common stock, or the expectation that such sales may occur, could significantly reduce the market price of our common stock. In connection with our March 2014 public offering of common stock, we agreed to provide resale registration rights for the shares of our common stock held by entities affiliated with one of our principal stockholders and two of our directors, Julian C. Baker and Dr. Stephen R. Biggar, which we refer to as the Baker Entities. In connection with our January 2016 public offering of common stock, we entered into a formal registration rights agreement with the Baker Entities to provide for these rights. Under the registration rights agreement, we have agreed that, if at any time and from time to time, the Baker Entities demand that we register their shares of our common stock for resale under the Securities Act, we would be obligated to effect such registration. On May 25, 2022, we filed a registration statement covering the sale of up to 42,393,855 shares of our common stock, which includes 489,269 shares of our common stock issuable upon the exercise of warrants that were owned by the Baker Entities as of May 16, 2022, and which represented approximately 26 percent of our outstanding shares at the time. Our registration obligations under this registration rights agreement, which cover all shares now held or later acquired by the Baker Entities, will be in effect for up to 10 years, and include our obligation to facilitate
certain underwritten public offerings of our common stock by the Baker Entities in the future. If the Baker Entities sell a large number of our shares, or the market perceives that the Baker Entities intend to sell a large number of our shares, this could adversely affect the market price of our common stock. We also may elect to sell from time to time an indeterminate number of shares on our own behalf pursuant to a registration statement or in a private placement. Our stock price may decline as a result of the sale of the shares of our common stock included in any of these registration statements or future financings.
If our officers, directors, and largest stockholders choose to act together, they may be able to significantly influence our management and operations, acting in their best interests and not necessarily those of our other stockholders.
Our directors, executive officers and holders of 5% or more of our outstanding common stock and their affiliates beneficially own a substantial portion of our outstanding common stock. As a result, these stockholders, acting together, have the ability to significantly influence all matters requiring approval by our stockholders, including the election of all of our board members, amendments to our certificate of incorporation, going-private transactions, and the approval of mergers or other business combination transactions. The interests of this group of stockholders may not always coincide with our interests or the interests of other stockholders and they may act in a manner that advances their best interests and not necessarily those of our other stockholders.
Anti-takeover provisions in our charter documents and under Delaware law may make an acquisition of us more complicated and may make the removal and replacement of our directors and management more difficult.
Our amended and restated certificate of incorporation and amended and restated bylaws contain provisions that may delay or prevent a change in control, discourage bids at a premium over the market price of our common stock and adversely affect the market price of our common stock and the voting and other rights of the holders of our common stock. These provisions may also make it difficult for stockholders to remove and replace our board of directors and management. These provisions:
•establish that members of the board of directors may be removed only for cause upon the affirmative vote of stockholders owning at least a majority of our capital stock;
•authorize the issuance of “blank check” preferred stock that could be issued by our board of directors to increase the number of outstanding shares and prevent or delay a takeover attempt;
•limit who may call a special meeting of stockholders;
•establish advance notice requirements for nominations for election to the board of directors or for proposing matters that can be acted upon at stockholder meetings;
•prohibit our stockholders from making certain changes to our amended and restated certificate of incorporation or amended and restated bylaws except with 662/3% stockholder approval; and
•provide for a board of directors with staggered terms.
We are also subject to provisions of the General Corporation Law of the State of Delaware that, in general, prohibit any business combination with a beneficial owner of 15% or more of our common stock for three years unless the holder’s acquisition of our stock was approved in advance by our board of directors. Although we believe these provisions collectively provide for an opportunity to receive higher bids by requiring potential acquirors to negotiate with our board of directors, they would apply even if the offer may be considered beneficial by some stockholders.
We do not intend to pay dividends on our common stock in the foreseeable future; as such, you must rely on stock appreciation for any return on your investment.
To date, we have not paid any cash dividends on our common stock, and we do not intend to pay any dividends in the foreseeable future. Instead, we intend to retain any future earnings to fund the development and growth of our business. For this reason, the success of an investment in our common stock, if any, will depend on the appreciation of our common stock, which may not occur. There is no guarantee that our common stock will appreciate, and therefore, a holder of our common stock may not realize a return on his or her investment.
This item is not applicable.
Item 1C. Cybersecurity.
Risk management and strategy
We have implemented and maintain various information security processes designed to identify, assess and manage material risks from cybersecurity threats to our critical computer networks, third party hosted services, communications systems, hardware and software, and our critical data, including intellectual property, confidential information that is proprietary, strategic or competitive in nature, and data related to our clinical trials and products (Information Systems and Data).
Our information security function, our Chief Information Officer (CIO), and our Chief Compliance Officer (CCO) help identify, assess and manage the Company’s cybersecurity threats and risks. This group works to identify and assess risks from cybersecurity threats by monitoring and evaluating our threat environment and the Company’s risk profile using various methods in certain contexts, including, for example, manual tools, subscribing to reports and services that identify cybersecurity threats, analyzing reports of threat actors, conducting scans of certain environments, evaluating certain threats reported to us, conducting threat and vulnerability assessments, using external intelligence feeds, and using third parties to conduct tabletop incident response exercises and other tests.
Depending on the environment, we implement and maintain various technical, physical, and organizational measures, processes, standards and policies designed to manage and mitigate material risks from cybersecurity threats to our Information Systems and Data, including, for example: incident detection and response policies, disaster recovery/business continuity policies, a vulnerability management policy, a vendor risk management program, risk assessments, encryption of certain data, network security controls and data segmentation for certain systems, access controls, physical security, asset management and tracking, systems monitoring, employee training, penetration testing, cybersecurity insurance, and dedicated cybersecurity staff.
Our assessment and management of material risks from cybersecurity threats are integrated into the Company’s overall risk management processes. For example, (1) cybersecurity risk is addressed as a component of the Company’s enterprise risk management program; (2) the information security department works with management to prioritize our risk management processes and mitigate cybersecurity threats that are more likely to lead to a material impact to our business; and (3) our senior management evaluates material risks from cybersecurity threats against our overall business objectives and reports to the audit committee of the board of directors, which evaluates our overall enterprise risk.
We use third-party service providers to assist us from time to time to identify, assess, and manage material risks from cybersecurity threats, including for example, outside legal counsel, threat intelligence service providers, cybersecurity consultants, cybersecurity software providers, penetration testing firms, managed cybersecurity service providers, darkweb monitoring services, and forensic investigators.
We use third-party service providers to perform a variety of functions throughout our business, such as application providers, hosting companies, contract research organizations, contract manufacturing organizations, distributors, and supply chain resources. We have vendor management processes designed to help manage cybersecurity risks associated with our use of certain of these providers. For certain vendors, these processes include vendor risk assessments, security questionnaires, review of vendors’ written security program, and imposition of contractual obligations related to information security on certain vendors.
For a description of the risks from cybersecurity threats that may materially affect the Company and how they may do so, see our risk factors under Part 1. Item 1A. Risk Factors, including: “We and the third parties with whom we work are subject to stringent and evolving U.S. and foreign laws, regulations and rules, contractual obligations, industry standards, policies and other obligations related to data privacy and security. Our (or the third parties with whom we work) actual or perceived failure to comply with such obligations could lead to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; and other adverse business consequences.”; and “If our information technology systems or data, or those of third parties with whom we work, are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions, interruptions to operations or clinical trials, reputational harm, litigation, fines and penalties, disruptions of our business operations, and a loss of customers or sales.”
Governance
Our board of directors addresses the Company’s cybersecurity risk management as part of its general oversight function. The audit committee of the board of directors is responsible for overseeing the Company’s cybersecurity risk management processes, including oversight of mitigation of risks from cybersecurity threats.
Our cybersecurity risk assessment and management processes are implemented and maintained by certain Company management, including our Director of Information Security, who has managed the Security Operations Center for a Fortune 500 company, and led cybersecurity efforts as the Director of IT at another organization.
The Director of Information Security, along with management, including the CIO and CCO, is responsible for hiring appropriate personnel, helping to integrate cybersecurity risk considerations into the Company’s overall risk management strategy, and communicating key priorities to relevant personnel. Management is responsible for approving budgets, helping prepare for cybersecurity incidents, approving cybersecurity processes, and reviewing security assessments and other security-related reports.
Our incident response policy is designed to escalate certain cybersecurity incidents to members of management depending on the circumstances, including our CIO, Senior Vice President of Finance, CCO, Senior Vice President of People and Performance, and Director of Information Security. This team works with the Company’s incident response team to help the Company mitigate and remediate cybersecurity incidents of which they are notified. In addition, the Company’s incident response policy includes reporting to the audit committee of the board of directors for certain cybersecurity incidents.
The audit committee receives periodic reports from senior management concerning the Company’s significant cybersecurity threats and risks and the processes the Company has implemented to address them.
Item 2. Properties.
As of December 31, 2024, our primary facility consists of approximately 67,000 square feet of office space in San Diego, California. We also lease a facility in Princeton, New Jersey that covers approximately 25,000 square feet of office space, which is leased through January 2026. We believe that our existing facilities are adequate to meet our current needs, and that suitable additional alternative spaces will be available in the future on commercially reasonable terms.
Item 3. Legal Proceedings.
Patent Infringement
On July 24, 2020, we filed complaints against (i) Aurobindo Pharma Limited and its affiliate Aurobindo Pharma USA, Inc. and (ii) Teva Pharmaceuticals USA, Inc. and its affiliate Teva Pharmaceutical Industries Ltd., and on July 30, 2020, we filed complaints against (i) Hetero Labs Limited and its affiliates Hetero Labs Limited Unit-V and Hetero USA Inc., (ii) MSN Laboratories Private Ltd. and its affiliate MSN Pharmaceuticals, Inc., and (iii) Zydus Pharmaceuticals (USA) Inc. and its affiliate Cadila Healthcare Limited. These complaints, which were filed in the United States District Court for the District of Delaware, allege infringement of certain of our Orange Book-listed patents covering NUPLAZID (Pimavanserin I Cases).
We entered into an agreement effective April 22, 2021 with Hetero settling all claims and counterclaims in the litigation. The agreement allows Hetero to launch its generic pimavanserin product on February 27, 2038, subject to certain triggers for earlier launch. The Hetero case was dismissed by joint agreement on May 3, 2021.
On September 30, 2022, we filed a stipulation and proposed order to stay the claims currently asserted against Teva and for Teva to be bound by the result of the litigation rendered against the remaining defendants Aurobindo and MSN, which was ordered by the Court on October 4, 2022.
On October 21, 2022, we filed additional complaints against Aurobindo, MSN and Zydus in the United States District Court for the District of Delaware alleging infringement of an additional Orange Book-listed patent covering NUPLAZID (Pimavanserin II Cases).
We entered into an agreement, effective March 31, 2023, with Zydus settling all claims and counterclaims in the Pimavanserin I Cases and Pimavanserin II Cases. The agreement allows Zydus to launch its generic pimavanserin 10 mg tablet products on September 23, 2036 and 34 mg capsule products on February 27, 2038, subject to certain triggers for earlier launch. The Zydus case was dismissed by joint agreement on April 5, 2023.
As a result of the above, only MSN remained as an active defendant in the Pimavanserin I Cases. On January 11, 2024, following summary judgment motions, the District Court entered final judgment in our favor that MSN’s submission of ANDA No. 214925 was an act of infringement in the Pimavanserin I Case and the ’740 patent was not invalid. On January 18, 2024, MSN filed a Notice of Appeal to the United States Court of Appeals for the Federal Circuit from the final judgment entered on January 11, 2024. The appeal is fully briefed and awaiting a date for oral argument.
In connection with the Pimavanserin II cases, MSN and Aurobindo are the remaining defendants. A bench trial was conducted from December 3, 2024 to December 6, 2024 in the matter. Post-trial briefing was completed on February 12, 2025 and we are awaiting the final decision of the Court.
On February 14, 2025, we filed a complaint against Zydus Lifesciences Limited, Zydus Worldwide DMCC, and Zydus Pharmaceuticals (USA) Inc. (collectively “Zydus”) in the United States District Court for the District of Delaware, alleging infringement of certain of our Orange Book-listed patents covering NUPLAZID (Pimavanserin) by Zydus’ proposed 34 mg pimavanserin tablet product. Zydus has not responded to the complaint as of yet and there is no schedule in place for the case.
Securities Class Action
On April 19, 2021, a purported stockholder of us filed a putative securities class action complaint (captioned City of Birmingham Relief Retirement Systems v. Acadia Pharmaceuticals Inc., Case No. 21-cv-0762) in the U.S. District Court for the Southern District of California against us and certain of our current executive officers. On September 29, 2021, the Court issued an order designating lead plaintiff and lead counsel. On December 10, 2021, lead plaintiff filed an amended complaint. The amended complaint generally alleges that defendants violated Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, by failing to disclose that the materials submitted in support of its sNDA seeking approval of pimavanserin for the treatment of hallucinations and delusions associated with dementia-related psychosis contained statistical and design deficiencies and that the FDA was unlikely to approve the sNDA in its current form. The amended complaint seeks unspecified monetary damages and other relief. On March 11, 2024, the Court granted plaintiffs’ motion for class certification and appointment of class representatives and class counsel. The parties are currently engaged in expert discovery, which is scheduled to close on June 13, 2025.
Opt-Out Litigation
On March 7, 2024, a purported stockholder of the Company filed a complaint (captioned Alger Dynamic Opportunities Fund v. Acadia Pharmaceuticals, Inc., Case No. 24-cv-00451) in the U.S. District Court for the Southern District of California against us and one executive officer. The complaint is based on the same underlying allegations as the Securities Class Action described above, and alleged claims under federal and state securities laws, and for common law fraud and negligent misrepresentations. On May 24, 2024, Defendants moved to dismiss the complaint. On October 31, 2024, the Court granted in part and denied in part Defendants’ motion to dismiss. The Court dismissed with leave to amend the purported stockholder’s state and common law claims, as well as the claim brought under Section 18(a) of the Securities Exchange Act of 1934, as amended. Defendants filed their answer to the Sections 10(b) and 20(a) claims on December 16, 2024. On January 13, 2025, the Court stayed this suit pending the outcome of the Securities Class Action.
Derivative Suit
On December 15, 2023, a purported stockholder of us filed a derivative action (captioned Kanner et al v. Biggar et al., Case No. 23-cv-2293) in the U.S. District Court for the Southern District of California against certain of our current directors. We are named as a nominal defendant. The complaint is based on the same alleged misconduct as the Securities Class Action, and asserts state law claims, on behalf of us, against the individual defendants for breach of fiduciary duty, unjust enrichment, abuse of control, waste of corporate assets, and insider trading. The complaint also asserts federal claims under sections 10(b), 21D, and 14(a) of the Securities Exchange Act of 1934, as amended. On December 27, 2023, the action was reassigned to District Judge William Q. Hayes and Magistrate Judge Michael S. Berg due to its relation to the Securities Class Action. On January 30, 2024, the parties jointly requested a stay of the action. The Court granted that request and the action was stayed on February 20, 2024, pending the outcome of our Demand Review Committee’s investigation into the underlying claims.
We currently believe that none of the foregoing claims or other actions pending against us as of December 31, 2024 is likely to have, individually or in the aggregate, a material adverse effect on our business, liquidity, financial position, or results of operations. Given the unpredictability inherent in litigation, however, we cannot predict the outcome of these matters. We are unable to estimate possible losses or ranges of losses that may result from these matters, and therefore we have not accrued any amounts in connection with these matters other than attorneys’ fees incurred to date.
Item 4. Mine Safety Disclosures.
This item is not applicable.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market Information
Our common stock is traded on the Nasdaq Global Select Market under the symbol “ACAD”.
Holders
As of February 18, 2025, there were 166,788,517 shares of common stock outstanding held by approximately 32 stockholders of record.
Dividends
To date, we have not paid any cash dividends on our common stock, and we do not intend to pay any dividends in the foreseeable future. Instead, we intend to retain any future earnings to fund the development and growth of our business.
Securities Authorized for Issuance Under Equity Compensation Plans
The information required by this Item regarding equity compensation plans is incorporated herein by reference to Item 12 of Part III of this Annual Report on Form 10-K.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Recent Sales of Unregistered Securities
Not applicable.
Performance Graph
The following graph shows a comparison of the total cumulative returns of an investment of $100 in cash from December 31, 2019 through December 31, 2024 in (i) our common stock, (ii) the Nasdaq Biotechnology Index, and (iii) the Nasdaq U.S. Benchmark TR Index. The comparisons in the graph are required by the SEC and are not intended to forecast or be indicative of the possible future performance of our common stock. The graph assumes that all dividends have been reinvested (to date, we have not declared any dividends).
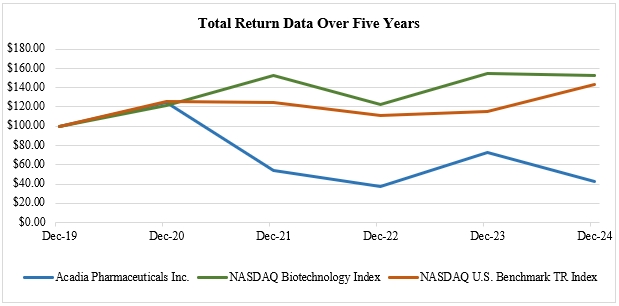
| | | | | | | | | | | | | | | | | | | | | | | | |
| | Dec-19 | | | Dec-20 | | | Dec-21 | | | Dec-22 | | | Dec-23 | | | Dec-24 | |
Acadia Pharmaceuticals Inc. | | $ | 100.00 | | | $ | 124.96 | | | $ | 54.56 | | | $ | 37.21 | | | $ | 73.19 | | | $ | 42.89 | |
NASDAQ Biotechnology Index | | $ | 100.00 | | | $ | 121.27 | | | $ | 152.67 | | | $ | 122.55 | | | $ | 154.93 | | | $ | 152.89 | |
NASDAQ U.S. Benchmark TR Index | | $ | 100.00 | | | $ | 125.69 | | | $ | 124.89 | | | $ | 111.27 | | | $ | 115.42 | | | $ | 143.60 | |
Item 6. [Reserved]
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
The following discussion and analysis of our consolidated financial condition and results of operations should be read in conjunction with our consolidated financial statements and related notes included elsewhere in this report. Past operating results are not necessarily indicative of results that may occur in future periods. This discussion contains forward-looking statements, which involve a number of risks and uncertainties. Such forward-looking statements include statements about the benefits to be derived from our products and our product candidates, the potential market opportunities for our products and our drug candidates, our strategy for the commercialization of our products, our plans for exploring and developing our products for additional indications, the commercialization of DAYBUE or trofinetide in jurisdictions other than the U.S., our plans and timing with respect to seeking regulatory approvals, the potential commercialization of any of our product candidates that receive regulatory approval, the progress, timing, results or implications of clinical trials and other development milestones and activities involving our products and our product candidates, our strategy for discovering, developing and, if approved, commercializing our product candidates, our existing and potential future collaborations, our estimates of future payments, revenues and profitability, our estimates regarding our capital requirements, future expenses and needs for additional financing, the potential or expected impacts of geopolitical and macroeconomic developments, possible changes in legislation, and other statements that are not historical facts, including statements which may be preceded by the words “aims,” “anticipates,” “believes,” “continue,” “could,” “estimates,” “expects,” “hopes,” “intends,” “may,” “plans,” “potential” “predicts,” “pro forma,” “projects,” “seeks,” “should,” “will,” “would,” or similar words. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain. For forward-looking statements, we claim the protection of the Private Securities Litigation Reform Act of 1995. Readers of this report are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date on which they are made. We undertake no obligation to update or revise publicly any forward-looking statements. Forward-looking statements are not guarantees of performance. Actual results or events may differ materially from those anticipated in our forward-looking statements as a result of various factors, including those set forth under the section captioned “Risk Factors” elsewhere in this report. Information in the following discussion for a yearly period means for the year ended December 31 of the indicated year.
Overview
Background
We are a biopharmaceutical company focused on the development and commercialization of innovative medicines that address unmet medical needs in CNS disorders and rare diseases.
We have two core franchises in neuroscience and neuro-rare diseases. Our neuroscience franchise is anchored by the commercial product NUPLAZID (pimavanserin), which is the first and only drug approved by the FDA for the treatment of hallucinations and delusions associated with PDP. Our neuro-rare disease franchise is anchored by the commercial product DAYBUE, which is the first and only drug approved for the treatment of Rett syndrome. Net product sales from these two commercial products totaled $957.8 million for 2024, compared with $726.4 million for 2023.
In August 2018, we acquired an exclusive North American license to develop and commercialize DAYBUE for Rett syndrome and other indications from Neuren. Rett syndrome is a debilitating neurological disorder that occurs predominantly in females following apparently normal development for the first six months of life. Rett syndrome also occurs in boys, albeit far less frequently. Typically, between six to eighteen months of age, patients experience a period of rapid decline with loss of purposeful hand use and spoken communication and inability to independently conduct activities of daily living. Symptoms also include seizures, hand movements or stereotypies, disorganized breathing patterns, scoliosis and sleep disturbances, among others. The FDA approval of DAYBUE for the treatment of Rett syndrome was based on the positive results from our pivotal Phase 3 LAVENDER™ study which demonstrated statistically significant improvement over placebo for both co-primary endpoints as well as the key secondary endpoint.
Under the terms of the 2018 agreement, Neuren received an upfront payment of $10.0 million and is eligible to receive milestone payments of up to $400.0 million based on the achievement of certain development and sales milestones for Rett syndrome in North America, of which, $50 million has been paid to date. Neuren is also eligible to receive up to $55.0 million in development and sales milestone for Fragile X syndrome in North America. In addition, Neuren is eligible to receive tiered, escalating, double-digit percentage royalties based on net sales in North America. The following tables provide a summary of milestone and royalty payments that Neuren remains eligible to receive based on the achievement of net sales of trofinetide in North America in any given year:
| |
Sales Milestones Based on Annual Net Sales in North America |
Net Sales ≥$250 million | $50 million |
Net Sales ≥$500 million | $50 million |
Net Sales ≥$750 million | $100 million |
Net Sales ≥$1 billion | $150 million |
| |
Tiered Royalty Rates Based on Annual Net Sales in North America |
≤$250 million | 10% |
>$250 million, but ≤$500 million | 12% |
>$500 million, but ≤$750 million | 14% |
>$750 million | 15% |
In July 2023, we expanded our current licensing agreement for trofinetide with Neuren to acquire rights to the drug outside of North America as well as global rights in Rett syndrome and Fragile X syndrome to Neuren’s development candidate NNZ-2591. Under the terms of the expanded agreement, Neuren received an upfront payment of $100.0 million and is eligible to receive up to an additional $426.3 million in milestone payments based on the achievement of certain commercial and sales milestones for trofinetide outside of North America and up to $831.3 million in milestone payments based on the achievement of certain development and sales milestones for NNZ-2591. In addition, we will be required to pay Neuren tiered royalties from the mid-teens to low-twenties percent based on net sales of trofinetide and NNZ-2591. The following table provides a summary of milestone payments that Neuren is eligible to receive based on the achievement of certain sales milestones under the terms of the expanded agreement:
| | |
Territory | First Commercial Sales Milestones | Total Sales Milestones |
Europe | $35 million (Rett syndrome)
$10 million (2nd indication) | Up to $170 million |
Japan | $15 million (Rett syndrome)
$4 million (2nd indication) | Up to $110 million |
Rest of World | __ | Up to $83 million |
In addition to these commercial products, we have a portfolio of product candidates and research programs that are designed to address significant unmet medical needs in CNS disorders and rare diseases. In order to achieve significant long-term growth, we plan to develop our current portfolio, expand our pipeline of early- and late-stage product candidates and expand into areas of rare disease that are adjacent to our existing franchises, including through strategic business development, and make use of our internal capabilities and knowledge.
Our most advanced product candidate is ACP-101 (intranasal carbetocin) for the treatment of hyperphagia in PWS, a neuro rare disease. Hyperphagia is an intense persistent sensation of hunger accompanied by food preoccupations, an extreme drive to consume food, food-related behavior problems, and a lack of normal satiety. In November 2023, we initiated the Phase 3 COMPASS PWS study evaluating the efficacy and safety of ACP-101 for the treatment of hyperphagia in PWS.
Our next most advanced product candidate is ACP-204 for the treatment of ADP. In November 2023, we initiated a Phase 2 study evaluating the efficacy and safety of ACP-204 for the treatment of hallucinations and delusions associated with ADP. We plan to initiate an additional Phase 2 study of ACP-204 in LBDP in the third quarter of 2025.
We have several product candidates in earlier stages of development for the treatment of CNS disorders and rare diseases, including ACP-711 for the treatment of essential tremor, for which we expect to initiate a Phase 2 study in 2026.
We have incurred substantial operating losses since our inception due in large part to expenditures for our research and development activities. As of December 31, 2024, we had an accumulated deficit of approximately $2.2 billion. Contingent on the level of business development activities we may complete as well as pipeline programs we may advance, we may incur operating losses as we incur significant research and development costs and costs for continued commercialization of our products.
Financial Operations Overview
Product Revenues
Net product sales consist of sales of our products. The FDA approved NUPLAZID in April 2016 for the treatment of hallucinations and delusions associated with PDP and we launched the product in the United States in May 2016. The FDA approved DAYBUE in March 2023 for the treatment of Rett syndrome and we launched the product in the United States in April 2023.
Cost of Product Sales
Cost of product sales consists of third-party manufacturing costs, freight, and indirect overhead costs associated with sales of our products. Cost of product sales may also include period costs related to certain inventory manufacturing services, excess or obsolete inventory adjustment charges, unabsorbed manufacturing and overhead costs, and manufacturing variances. In addition, cost of product sales may include license fees and royalties. License fees and royalties currently consist of milestone payments capitalized and subsequently amortized under our 2018 license agreement with Neuren. License fees and royalties also include royalties of tiered, escalating, double-digit percentages due to Neuren based upon net sales of DAYBUE.
Cost of sales for a newly launched product does not include the full cost of manufacturing until the initial pre-launch inventory is depleted, and additional inventory is manufactured and sold. Thus the cost of sales as a percentage of net sales of DAYBUE for the year ended December 31, 2024 and 2023 were affected by use of the initial pre-launch inventory, which was previously expensed as research and development expense, and is referred to as zero cost inventories. However, we do not expect that the cost of sales as a percentage of net sales of DAYBUE will increase significantly once we commence the sales of full cost inventories.
Research and Development Expenses
Our research and development expenses have consisted primarily of fees paid to external service providers, salaries and related personnel expenses, facilities and equipment expenses, and other costs incurred related to pre-commercial product candidates. We charge all research and development expenses to operations as incurred. Our research and development activities have focused on pimavanserin, trofinetide, ACP-101, ACP-204 and other earlier-stage product candidates. In connection with the FDA approval of DAYBUE, we are required to conduct post-marketing work, including a clinical study of renal impairment in healthy volunteers, nonclinical carcinogenicity studies, and nonclinical in vitro and clinical in vivo drug interaction studies. The FDA has released us from one of the five PMRs. In addition, we have fulfilled one of the five PMRs. Of the remaining three PMRs, we have also completed one and we are awaiting the FDA’s acknowledgement and acceptance of that completed PMR. We will be responsible for all costs incurred for these PMRs. In addition, we expect to incur increased research and development expenses as a result of advancement of our early-stage product candidates.
We use external service providers to manufacture our product candidates and for the majority of the services performed in connection with the preclinical and clinical development of our product candidates. Historically, we have used our internal research and development resources, including our employees and discovery infrastructure, across several projects and many of our costs have not been attributable to a specific project. Accordingly, we have not reported our internal research and development costs on a project-by-project basis. To the extent that external expenses are not attributable to a specific project, they are allocated proportionally to each of the projects.
The following table summarizes our research and development expenses for the years ended December 31, 2024, 2023, and 2022 (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Costs of external service providers: | | | | | | | | | |
NUPLAZID (pimavanserin) | | $ | 34,369 | | | $ | 55,527 | | | $ | 62,746 | |
DAYBUE (trofinetide) | | | 30,677 | | | | 32,065 | | | | 62,300 | |
ACP-101 | | | 30,401 | | | | 11,887 | | | | 2,085 | |
ACP-204 | | | 54,389 | | | | 43,768 | | | | 16,898 | |
Early-stage product candidates | | | 44,703 | | | | 26,789 | | | | 45,803 | |
Upfront and milestone payments* | | | 34,500 | | | | 102,500 | | | | 88,741 | |
Subtotal | | | 229,039 | | | | 272,536 | | | | 278,573 | |
Internal costs | | | 60,110 | | | | 61,675 | | | | 60,422 | |
Stock-based compensation | | | 14,100 | | | | 17,408 | | | | 22,580 | |
Total research and development expenses | | $ | 303,249 | | | $ | 351,619 | | | $ | 361,575 | |
_____________________
* Includes upfront and milestone consideration as well as transaction costs associated with acquired in-process research and development.
At this time, due to the risks inherent in regulatory requirements and clinical development, we are unable to estimate with certainty the costs we will incur to support the commercialization of DAYBUE, as well as the further development of our early-stage product candidates. Likewise, we are unable to determine with certainty the anticipated completion dates for our current research and development programs. Clinical development and regulatory approval timelines, probability of success, and development costs vary widely. While our current development efforts are primarily focused on advancing the development of ACP-101, ACP-204 and other early-stage product candidates, we anticipate that we will make determinations as to which programs to pursue and how much funding to direct to each program on an ongoing basis in response to the scientific and clinical success of each product candidate, as well as an ongoing assessment of the commercial potential of each candidate and our financial position. We cannot forecast with any degree of certainty which product candidates will be subject to future collaborative or licensing arrangements, when such arrangements will be secured, if at all, and to what degree any such arrangements would affect our development plans and capital requirements. Similarly, we are unable to estimate with certainty the costs we will incur for post-marketing studies that we committed to conduct in connection with FDA approval of DAYBUE.
We expect our research and development expenses will continue to be substantial as we conduct studies pursuant to our PMRs and pursue the further development of ACP-101, ACP-204 and other early-stage product candidates. The lengthy process of completing clinical trials and supporting development activities and seeking regulatory approval for our product candidates requires the expenditure of substantial resources. Any failure by us or delay in completing clinical trials, or in obtaining regulatory approvals, could cause our research and development expenses to increase and, in turn, have a material adverse effect on our results of operations.
Selling, General and Administrative Expenses
Our selling, general and administrative expenses consist of salaries and other related costs, including stock-based compensation expense, for our commercial personnel, including our specialty sales forces, our medical education professionals, and our personnel serving in executive, finance, business development, and business operations functions. Also included in selling, general and administrative expenses are fees paid to external service providers to support our commercial activities associated with our products, professional fees associated with legal and accounting services, costs associated with patents and patent applications for our intellectual property and charitable donations to independent charitable foundations that support Parkinson’s disease patients generally. Changes in selling, general and administrative expenses in future periods are subject to the evolving PDP market dynamics and the Rett syndrome market.
Gain on Sale of Non-Financial Asset
Following the FDA approval of DAYBUE, we were granted a Rare Pediatric Disease PRV. During the year ended December 31, 2024, we sold the PRV to a third party for the aggregate net proceeds of $146.5 million.
Income Tax Expense
Because we maintain a full valuation allowance against our net deferred tax assets, income tax expense is expected to primarily consist of current federal and state tax expense as a result of taxable income anticipated or incurred in certain jurisdictions. Income tax expense may fluctuate from quarter to quarter due to adjustments related to non-recurring transactions, timing of revenue and expense across different tax jurisdictions and changes in certain tax assessments.
Critical Accounting Policies and Estimates
A summary of the significant accounting policies is provided in Note 2 to our Consolidated Financial Statements.
The preparation of financial statements in accordance with GAAP requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. We evaluate our estimates on an ongoing basis. Our estimates are based on historical experience and on various other assumptions and factors that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities and the reported amounts of revenues and expenses that are not readily apparent from other sources. Actual results may differ from those estimates under different assumptions and conditions.
Management considers an accounting estimate to be critical if:
•it requires a significant level of estimation uncertainty; and
•changes in the estimate are reasonably likely to have a material effect on our financial condition or results of operations.
We believe the following critical accounting policies and estimates describe the more significant judgments and estimates used in the preparation of our consolidated financial statement.
Product Sales, Net
We sell NUPLAZID through SPs and SDs. SPs dispense product to a patient based on the fulfillment of a prescription and SDs sell product to government facilities, long-term care pharmacies, or in-patient hospital pharmacies. We sell DAYBUE through a single wholesale distributor. Product shipping and handling costs are included in cost of product sales.
We recognize revenue from product sales at the net sales price (the “transaction price”) which includes estimates of variable consideration for which reserves are established and reflects each of these as either a reduction to the related account receivable or as an accrued liability, depending on how the amount payable is settled. Overall, these reserves reflect our best estimates of the amount of consideration to which we are entitled based on the terms of the contract. Actual amounts of consideration ultimately received may differ from our estimates. If actual results in the future vary from estimates, we may need to adjust our estimates, which would affect net revenue in the period of adjustment. The following sales discounts and allowances involve a substantial degree of judgment:
Rebates: Allowances for rebates include mandated discounts under the Medicaid Drug Rebate Program and the Medicare Part D prescription drug benefit. Rebates are amounts owed after the final dispensing of the product to a benefit plan participant and are based upon contractual agreements with, or statutory requirements pertaining to, Medicaid and Medicare benefit providers. The allowance for rebates is based on statutory discount rates and expected utilization. Our estimates for expected utilization of rebates is based on historical data received from the SPs, SDs and the single wholesale distributor since product launch. Rebates are generally invoiced and paid in arrears so that the accrual balance consists of an estimate of the amount expected to be incurred for the current quarter’s activity, plus an accrual balance for prior quarters’ unpaid rebates still estimated to be incurred. Allowances for rebates also include amounts due under the Inflation Reduction act of 2022 for Medicare Part D unit sales with applicable period AMP increases that outpace inflation over the benchmark period. The applicable period will be twelve months on October 1 of each year, with the initial applicable period beginning on October 1, 2022. The benchmark period AMP price is January 1, 2021 through September 30, 2021. Our estimates are based Medicare Part D sales as a percentage of gross sales and the rate AMP for the current period will be in excess the benchmark period. We regularly monitor our estimates and record adjustments when rebate trends, rebate programs and contract terms, legislative changes, or other significant events indicate that a change in the estimates is appropriate. To date, our estimates have not differed materially from actual rebates. However, subsequent changes in estimates may result in a material change in our accruals, which could also materially affect our balance sheet and results of operations.
Chargebacks: Chargebacks are discounts and fees that relate to contracts with government and other entities purchasing from the SDs at a discounted price. The SDs charge back to us the difference between the price initially paid by the SDs and the discounted price paid to the SDs by these entities. We also incur group purchasing organization fees for transactions through certain purchasing organizations. We estimate sales with these entities and accrue for anticipated chargebacks and organization fees, based on the applicable contractual terms. To date, our estimates have not differed materially from the actual chargebacks and organization fees. However, subsequent changes in estimates may result in a material change in our accruals, which could also materially affect our balance sheet and results of operations.
Research and Development Accruals
We estimate certain costs and expenses and accrue for these liabilities as part of our process of preparing financial statements. Examples of areas in which subjective judgments may be required include, among other things, costs associated with services provided by contract organizations for preclinical development, manufacturing of our product candidates and clinical trials, and personnel related expenses. We accrue for costs incurred as the services are being provided by monitoring the status of the trial or services provided, and the invoices received from our external service providers. In the case of clinical trials, a portion of the estimated cost normally relates to the projected cost to treat a patient in the trials, and this cost is recognized based on the number of patients enrolled in the trial. Other indirect costs are generally recognized on a straight-line basis over the estimated period of the study. As actual costs become known to us, we adjust our accruals. To date, our estimates have not differed materially from the actual costs incurred. However, subsequent changes in estimates may result in a material change in our accruals, which could also materially affect our balance sheet and results of operations.
Stock-Based Compensation
The fair value of each employee stock option and each employee stock purchase plan right granted is estimated on the grant date under the fair value method using the Black-Scholes valuation model, which requires us to make a number of assumptions including the estimated expected life of the award and related volatility. The fair value of restricted stock units is estimated based on the market price of our common stock on the date of grant. The estimated fair values of stock options, purchase plan rights, and regular restricted stock units are then expensed over the vesting period. For restricted stock units requiring satisfaction of both market and service conditions, the estimated fair values are generally expensed over the longest of the explicit, implicit and derived service periods. The fair value of performance-based stock awards (PSUs) that vest upon the achievement of certain pre-defined company-specific performance-based criteria is estimated based on the closing market price of our common stock on the date of grant. Expense related to these PSUs is recognized ratably over the expected performance period once the pre-defined performance-based criteria for vesting becomes probable and can vest up to 200 percent of the target number of shares granted. Beginning in 2024, the structure of the PSU design was revised with a relative total shareholder return (rTSR) approach such that awards are earned for our rTSR performance over three-year measurement periods relative to a peer group of companies and the actual numbers of PSUs that will vest at the end of the performance period may be anywhere from zero to 150 percent of the target number of shares granted. The fair value of these PSUs is estimated using a Monte Carlo model. Expense related to these PSUs is recognized ratably over the three-year measurement period. See also Item 15 of Part IV, “Notes to Consolidated Financial Statements—Note 2—Summary of Significant Accounting Policies” for further discussion of our assumptions and estimates related to our stock-based compensation.
Results of Operations
Fluctuations in Operating Results
Our results of operations have fluctuated significantly from period to period in the past and are likely to continue to do so in the future. We anticipate that our quarterly and annual results of operations will be impacted for the foreseeable future by several factors, including the progress and timing of expenditures related to our commercial activities associated with our products and the extent to which we generate revenue from product sales, our development of pimavanserin for the negative symptoms of schizophrenia, our further development of our early-stage product candidates and the progress and timing of expenditures related to studies of DAYBUE pursuant to our PMRs. Further, we expect our sales allowances to vary from quarter to quarter due to fluctuations in our Medicare Part D Coverage Gap liability and the volume of purchases eligible for government mandated discounts and rebates, as well as changes in discount percentages that may be impacted by potential future price increases and other factors. We cannot predict with certainty what the full impact that geopolitical and macroeconomic developments, including the ongoing military conflict between Ukraine and Russia and in the Middle East, and tariffs and trade tensions may have on our business, results of operations, financial condition and prospects. Due to these fluctuations, we believe that the period-to-period comparisons of our operating results are not a good indication of our future performance.
Comparison of the Years Ended December 31, 2024 and 2023
Product Sales, Net
Net product sales, comprised of our products, were $957.8 million and $726.4 million for the years ended December 31, 2024 and 2023, respectively.
Net product sales of NUPLAZID were $609.4 million and $549.2 million in 2024 and 2023, respectively. The increase in net product sales of NUPLAZID of $60.2 million was due to the growth in NUPLAZID unit sales as well as a higher average net selling price in NUPLAZID in 2024 compared to 2023. Net product sales of DAYBUE were $348.4 million and $177.2 million in 2024 and 2023, respectively. The increase in net product sales of DAYBUE of $171.2 million was mainly due to the growth in DAYBUE unit sales in 2024 compared to 2023.
The following table provides a summary of activity with respect to our sales allowances and accruals for the year ended December 31, 2024 (in thousands):
| | | | | | | | | | | | | | | | |
| | Distribution
Fees,
Discounts &
Chargebacks | | | Co-Pay
Assistance | | | Rebates, Data
Fees & Returns | | | Total | |
Balance at December 31, 2023 | | $ | 12,156 | | | $ | (520 | ) | | $ | 86,054 | | | $ | 97,690 | |
Provision related to current period sales | | | 122,083 | | | | 5,148 | | | | 168,868 | | | | 296,099 | |
Credits/payments for current period sales | | | (110,200 | ) | | | (5,262 | ) | | | (20,762 | ) | | | (136,224 | ) |
Credits/payments for prior period sales | | | (12,156 | ) | | | 520 | | | | (86,054 | ) | | | (97,690 | ) |
Balance at December 31, 2024 | | $ | 11,883 | | | $ | (114 | ) | | $ | 148,106 | | | $ | 159,875 | |
Cost of Product Sales
Cost of product sales was $81.8 million and $41.6 million in 2024 and 2023, respectively, or approximately 9% and 6% of net product sales, respectively. Cost of product sales as a percentage of net product sales for NUPLAZID remained flat in 2024 as compared to 2023. The increase in cost of product sales was primarily due to the $51.8 million in license fees and royalties expensed during 2024 as compared to $21.8 million in the same period of 2023 for DAYBUE, including royalties due to Neuren based on net sales of DAYBUE and the amortization of the milestone payments capitalized under our 2018 license agreement with Neuren.
Certain manufacturing related expenses incurred prior to DAYBUE receiving FDA approval were classified as research and development expenses, resulting in zero cost inventory. Prior to receiving FDA approval for DAYBUE in March 2023, we manufactured inventory and recorded approximately $29.9 million related to the zero cost inventory as research and development expense. Utilizing the actual direct costs to manufacture DAYBUE prior to receiving FDA approval, had the previously expensed inventory been capitalized and recognized when sold, the total cost of sales with these manufacturing costs included for the year ended December 31, 2024 would have increased by approximately $14.9 million. We do not expect our cost of product sales for DAYBUE to increase significantly as a percentage of net product sales in future periods as we continue to produce inventory for future sales. We expect to finish selling the zero cost inventories of DAYBUE in 2025.
Subsequent to using our entire zero cost inventories, we estimate our overall cost of product sales as a percentage of total net product sales will be in the range of a mid-single digit to high single digit percentage.
Research and Development Expenses
Research and development expenses decreased to $303.2 million in 2024, including $14.1 million in stock-based compensation, from $351.6 million in 2023, including $17.4 million in stock-based compensation. The decrease in research and development expenses during 2024 was due to decreased business development payments, which in the period ending December 31, 2023 included the $100.0 million payment to Neuren under the expanded license agreement for trofinetide, partially offset by increased costs from clinical stage programs.
Selling, General and Administrative Expenses
Selling, general and administrative expenses increased to $488.4 million in 2024, including $51.6 million in stock-based compensation expense, from $406.6 million in 2023, including $48.0 million in stock-based compensation expense. The increase in selling, general and administrative expenses was primarily driven by increased marketing costs to support the NUPLAZID and DAYBUE franchises in the U.S. and investments to support commercialization of DAYBUE outside the U.S.
Gain on Sale of Non-Financial Asset
Following the FDA approval of DAYBUE, we were granted a Rare Pediatric Disease PRV. During the year ended December 31, 2024, we sold the PRV to a third party for the aggregate net proceeds of $146.5 million. No other non-financial asset sale happened during the year ended December 31, 2023.
Comparison of the Years Ended December 31, 2023 and 2022
Product Sales, Net
Product sales, net, comprised of NUPLAZID, were $726.4 million and $517.2 million in 2023 and 2022, respectively.
Net product sales of NUPLAZID were $549.2 million and $517.2 million in 2023 and 2022, respectively. The increase in net product sales of NUPLAZID of $32.0 million was due to the growth in NUPLAZID unit sales as well as a higher average net selling price in NUPLAZID in 2023 compared to 2022. Net product sales of DAYBUE were $177.2 million for 2023. There were no net product sales of DAYBUE during 2022.
The following table provides a summary of activity with respect to our sales allowances and accruals for the year ended December 31, 2023 (in thousands):
| | | | | | | | | | | | | | | | |
| | Distribution
Fees,
Discounts &
Chargebacks | | | Co-Pay
Assistance | | | Rebates, Data
Fees & Returns | | | Total | |
Balance at December 31, 2022 | | $ | 10,923 | | | $ | (340 | ) | | $ | 26,046 | | | $ | 36,629 | |
Provision related to current period sales | | | 97,797 | | | | 3,979 | | | | 113,011 | | | | 214,787 | |
Credits/payments for current period sales | | | (85,641 | ) | | | (4,499 | ) | | | (26,957 | ) | | | (117,097 | ) |
Credits/payments for prior period sales | | | (10,923 | ) | | | 340 | | | | (26,046 | ) | | | (36,629 | ) |
Balance at December 31, 2023 | | $ | 12,156 | | | $ | (520 | ) | | $ | 86,054 | | | $ | 97,690 | |
Cost of Product Sales
Cost of product sales was $41.6 million and $10.2 million in 2023 and 2022, respectively, or approximately 6% and 2% of net product sales, respectively. Cost of product sales as a percentage of net product sales for NUPLAZID remained flat in 2023 as compared to 2022. The increase in cost of product sales was primarily due to the $21.8 million in license fees and royalties expensed during 2023 for DAYBUE, including royalties due to Neuren based on net sales of DAYBUE and the amortization of the milestone payments capitalized under our 2018 license agreement with Neuren. There were no license fees and royalties in the same period of 2022 for either product.
Certain manufacturing related expenses incurred prior to DAYBUE receiving FDA approval were classified as research and development expenses, resulting in zero cost inventory. Prior to receiving FDA approval for DAYBUE in March 2023, we manufactured inventory and recorded approximately $29.9 million related to the zero cost inventory as research and development expense. Utilizing the actual direct costs to manufacture DAYBUE prior to receiving FDA approval, had the previously expensed inventory been capitalized and recognized when sold, the total cost of sales with these manufacturing costs included for the year ended December 31, 2023 would have increased by approximately $9.4 million. We do not expect our cost of product sales for DAYBUE to increase significantly as a percentage of net product sales in future periods as we continue to produce inventory for future sales.
Subsequent to using our entire zero cost inventories, we estimate our overall cost of product sales as a percentage of total net product sales will be in the range of a mid-single digit to high single digit percentage.
Research and Development Expenses
Research and development expenses decreased to $351.6 million in 2023, including $17.4 million in stock-based compensation expense, from $361.6 million in 2022, including $22.6 million in stock-based compensation expense. The decrease in research and development expenses during 2023 was mainly due to trofinetide commercial supply build that was expensed prior to approval. There was a similar level of clinical spend and business development investment year over year.
Selling, General and Administrative Expenses
Selling, general and administrative expenses increased to $406.6 million in 2023, including $48.0 million in stock-based compensation expense, from $369.1 million in 2022, including $44.4 million in stock-based compensation expense. The increase in selling, general and administrative expenses was primarily due to increased commercial costs associated with the DAYBUE launch, partially offset by reductions in expenses associated with NUPLAZID.
Liquidity and Capital Resources
We have funded our operations primarily with revenues from sales of our products since their approvals, and through sales of our equity securities and interest income. We anticipate that the level of cash used in our operations will fluctuate in future periods depending on the levels of spending required for our ongoing and planned commercial activities for our products, our ongoing and planned development activities for pimavanserin for the negative symptoms of schizophrenia, ACP-101 as a treatment for PWS and ACP-204 as a treatment for ADP, studies to be conducted pursuant to our PMRs, our ongoing and planned development activities for other early- and late-stage product candidates and strategic business development to further expand our portfolio. We expect that our cash, cash equivalents and investment securities, as well as funds generated by anticipated sales of our products, will be sufficient to fund our planned operations through and beyond the next 12 months.
We may require additional financing in the future to fund our operations. Our future capital requirements will depend on, and could increase significantly as a result of, many factors, including:
•the costs of acquiring additional product candidates or research and development programs;
•the scope, prioritization and number of our research and development programs;
•the ability of our collaborators and us to reach the milestones and other events or developments triggering payments under our collaboration or license agreements, or our collaborators’ ability to make payments under these agreements;
•our ability to enter into new collaboration and license agreements;
•the progress in, and the costs of, our ongoing and planned development activities for pimavanserin, post-marketing studies for DAYBUE to be conducted over the next several years, and ongoing and planned commercial activities for our products;
•the costs of our development activities for our product candidates;
•the costs of commercializing our products, including the maintenance and development of our sales and marketing capabilities;
•the costs of establishing, or contracting for, sales and marketing capabilities for our product candidates;
•the amount of U.S. product sales from our products;
•the costs of preparing applications for regulatory approvals for DAYBUE in jurisdictions other than the U.S., for NUPLAZID in additional indications other than PDP and for other product candidates, as well as the costs required to support review of such applications;
•the costs of manufacturing and distributing our products for commercial use in the U.S.;
•our ability to obtain regulatory approval for, and subsequently generate product sales from, NUPLAZID for the negative symptoms of schizophrenia, or from DAYBUE, and our product candidates;
•the extent to which we are obligated to reimburse collaborators or collaborators are obligated to reimburse us for costs under collaboration agreements;
•the costs involved in filing, prosecuting, enforcing, and defending patent claims and other intellectual property rights;
•the costs of maintaining or securing manufacturing arrangements for clinical or commercial production of pimavanserin, trofinetide or other product candidates; and
•the costs associated with litigation, including the costs incurred in defending against any product liability claims that may be brought against us related to our products.
In the past, periods of turmoil and volatility in the financial markets have adversely affected the market capitalizations of many biotechnology companies, and generally made equity and debt financing more difficult to obtain. For example, due to geopolitical and macroeconomic developments, including the Ukraine-Russia military conflict and related sanctions, the ongoing conflicts in the Middle East, tariffs and trade tensions, the global credit and financial markets have experienced extreme volatility and disruptions, including diminished liquidity and credit availability, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability. These events, coupled with other factors, may limit our access to additional financing in the future. We cannot be certain that additional funding will be available to us on acceptable terms, or at all. If adequate funds are not available when needed, we will be required to delay, reduce the scope of, or eliminate one or more of our research or development programs or our commercialization efforts. We also may be required to relinquish greater or all rights to product candidates at an earlier stage of development or on less favorable terms than we would otherwise choose. Additional funding, if obtained, may significantly dilute existing stockholders and could negatively impact the price of our stock.
We have invested a substantial portion of our available cash in money market funds, municipal bonds, and government sponsored enterprises in accordance with our investment policy. Our investment policy defines allowable investments and establishes guidelines relating to credit quality, diversification, and maturities of our investments to preserve principal and maintain liquidity. All investment securities have a credit rating of at least Aa3/AA- or better, or P-1/A-1 or better, as determined by Moody’s Investors Service or Standard & Poor’s. Our investment portfolio has not been adversely impacted by the disruptions in the credit markets that have occurred in the past. However, if there are future disruptions in the credit markets, there can be no assurance that our investment portfolio will not be adversely affected.
Material Cash Requirements
Our material cash requirements in the short and long term consist of the operational, manufacturing, and capital expenditures, a portion of which contain contractual or other obligations. We plan to fund our material cash requirements with our current financial resources together with our anticipated receipts from product sales. On a long-term basis, we manage future cash requirements relative to our long-term business plans.
Our primary uses of cash and operating expenses relate to paying employees and consultants, administering clinical trials, marketing our products, and providing technology and facility infrastructure to support our operations. We also make investments in our office and laboratory facilities to enable continued expansion of our business.
As of December 31, 2024 we have long-term contractual obligations related to our operating leases of $59.4 million. In May 2023, we subleased our 2nd floor of corporate office space in San Diego with a total minimum sublease income of $18.4 million. In addition to operating leases, we enter into certain other long-term commitments for goods and services that are outstanding for periods greater than one year. We also enter into short-term agreements with various vendors and suppliers of goods and services in the normal course of operations through purchase orders or other documentation, or that are undocumented except for an invoice. Such short-term agreements are generally outstanding for periods less than a year and are settled by cash payments upon delivery of goods and services. The nature of the work being conducted under these agreements is such that, in most cases, the services may be stopped on short notice. In such event, we would not be liable for the full amount of the agreement.
We have entered into various collaboration, licensing and merger agreements which generally include upfront license fees, development and commercial milestone payments upon achievement of certain clinical and commercial development and annual net sales milestones, as well as royalties calculated as a percentage of net product sales, with rates that vary by agreement. As of December 31, 2024, we may be required to make milestone payments up to $4.0 billion in the aggregate. $1.1 billion payments are contingent upon achieving future development and regulatory milestones, and $2.9 billion payments are contingent upon achieving future commercial milestones. Of the total $4.0 billion milestone payments, we are required to make milestone payments of $50.0 million within 60 days following Neuren’s invoice delivery after December 31, 2024 as our aggregate net revenue of trofinetide in North America for the treatment of Rett syndrome exceeded $250.0 million in 2024. We also paid Neuren $48.8 million for one third of the net proceeds from sale of PRV in February 2025.
We expect to receive our first invoice for rebates under the IRA from Medicare Part D unit sales in 2025. Payment is due 30 days after receiving such invoice; the payment will be set off against the allowance for such rebate that we have accrued up to the date of payment.
Cash Flows
At December 31, 2024, we had $756.0 million in cash, cash equivalents, and investment securities, compared to $438.9 million at December 31, 2023. This $317.1 million increase in cash, cash equivalents, and investment securities during 2024 was primarily due to net cash provided by operating activities, including the sale of the PRV.
Net cash provided by operating activities was $157.7 million in 2024 compared to net cash provided by operating activities of $16.7 million in 2023 and net cash used in operating activities of $114.0 million in 2022. The increase in net cash provided by operating activities in 2024 relative to 2023 was primarily due to an increase in our net revenues, decreased research and development costs and proceeds from sale of a non-financial asset, partially offset by increased sales and marketing costs. The increase in net cash provided by operating activities in 2023 relative to 2022 was primarily due to an increase in our net revenues and decreased research and development costs, partially offset by increased sales and marketing costs.
Net cash used in investing activities totaled $30.5 million in 2024 compared to net cash provided by investing activities of $32.0 million in 2023 and net cash provided by investing activities of $73.2 million in 2022. The increase in net cash used in investing activities in 2024 compared to 2023 was primarily due to increased net purchases of investment securities. The decrease in net cash provided by investing activities in 2023 compared to 2022 was primarily due to milestone payment of $40 million to Neuren and decreased net sale and maturities of investment securities.
Net cash provided by financing activities decreased to $6.8 million in 2024 compared to $25.1 million in 2023 and $8.2 million in 2022. This decrease in net cash provided by financing activities in 2024 relative to 2023 was attributable primarily to a decrease in proceeds resulting from the exercise of employee stock options. The increase in net cash provided by financing activities in 2023 relative to 2022 was primarily due to an increase in proceeds resulting from the exercise of employee stock options.
Off-Balance Sheet Arrangements
To date, we have not had any relationships with unconsolidated entities or financial partnerships, such as entities referred to as structured finance or special purpose entities, which are established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purposes. As such, we are not materially exposed to any financing, liquidity, market, or credit risk that could arise if we had engaged in these relationships.
Recent Accounting Pronouncements
See Item 15 of Part IV, “Notes to Consolidated Financial Statements—Note 2—Summary of Significant Accounting Policies.”
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
We invest our excess cash in investment-grade, interest-bearing securities. The primary objective of our investment activities is to preserve principal and liquidity. To achieve this objective, we invest in money market funds. U.S. treasury notes, and high quality marketable debt instruments of corporations and government sponsored enterprises with contractual maturity dates of generally less than one year. All investment securities have a credit rating of at least Aa3/AA- or better, or P-1/A-1 or better, as determined by Moody’s Investors Service or Standard & Poor’s. We do not have any direct investments in auction-rate securities or securities that are collateralized by assets that include mortgages or subprime debt. If a 10 percent change in interest rates were to have occurred on December 31, 2024 and 2023, this change would not have had a material effect on the fair value of our investment portfolio as of that date. Due to our investment in investment-grade, interest-bearing securities, as of the date of this Annual Report on Form 10-K, we do not expect anticipated changes in interest rates to have a material effect on our interest rate risk in future reporting periods.
Item 8. Financial Statements and Supplementary Data.
The consolidated financial statements required pursuant to this item are included in Item 15 of this report and are presented beginning on page F-1.
Item 9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosure.
None.
Item 9A. Controls and Procedures.
Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our periodic and current reports that we file with the SEC is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer (our principal executive officer and principal financial officer, respectively), as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable and not absolute assurance of achieving the desired control objectives. In reaching a reasonable level of assurance, management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures. In addition, the design of any system of controls is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions; over time, controls may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
As of December 31, 2024, we carried out an evaluation, under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of December 31, 2024.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting is a process designed by, or under the supervision and with the participation of, our Chief Executive Officer and Chief Financial Officer, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
As of December 31, 2024, our management assessed the effectiveness of our internal control over financial reporting, as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act of 1934, as amended, using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013). Based on this assessment, management, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, concluded that, as of December 31, 2024, our internal control over financial reporting was effective based on those criteria.
The effectiveness of our internal control over financial reporting as of December 31, 2024 has been audited by Ernst & Young LLP, an independent registered public accounting firm, as stated in its report, which is included herein.
Changes in Internal Control Over Financial Reporting
An evaluation was also performed under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, of any changes in our internal control over financial reporting that occurred during our last fiscal quarter and that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting. That evaluation did not identify any change in our internal control over financial reporting that occurred during our latest fiscal quarter and that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of
Acadia Pharmaceuticals Inc.
Opinion on Internal Control Over Financial Reporting
We have audited Acadia Pharmaceuticals Inc.’s internal control over financial reporting as of December 31, 2024, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, Acadia Pharmaceuticals Inc. (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2024, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2024 and 2023, the related consolidated statements of operations, comprehensive income (loss), cash flows and stockholders’ equity for each of the three years in the period ended December 31, 2024, and the related notes and financial statement schedule listed in the Index at Item 15(a)2 and our report dated February 27, 2025 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
San Diego, California
February 27, 2025
Item 9B. Other Information.
Insider Trading Arrangements
During the Company’s last fiscal quarter, the following officer (as defined in Rule 16a-1(f) under the Securities Exchange Act of 1934, as amended (the Exchange Act)) adopted or terminated a “Rule 10b5-1 trading arrangement” as defined in Item 408 of Regulation S-K, as follows:
•On November 19, 2024, James Kihara, Senior Vice President, Finance, adopted a Rule 10b5-1 trading arrangement providing for the sale of up to 18,000 shares of our common stock. The trading arrangement is intended to satisfy the affirmative defense in Rule 10b5-1(c). The duration of the trading arrangement is from two business days after the filing of this Form 10-K report until February 17, 2026, or earlier if and when all transactions under the trading arrangement are completed.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not applicable.
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
The remaining information required by this Item and not set forth below will be set forth under the proposal captioned “Election of Directors” and the sections captioned “Information Regarding the Board of Directors and Corporate Governance,” “Executive Officers” and “Delinquent Section 16(a) Reports,” if any, in our definitive Proxy Statement for our 2025 Annual Meeting of Stockholders to be filed with the SEC by April 30, 2025 (our Proxy Statement) and is incorporated in this report by reference.
The information required by Item 408(b) of Regulation S-K will be set forth in the section captioned “Insider Trading Arrangements and Policies” in our Proxy Statement and is incorporated in this report by reference.
We have adopted a code of ethics for directors, officers (including our principal executive officer, principal financial officer and principal accounting officer or controller) and employees, known as the Code of Business Conduct and Ethics. The Code of Business Conduct and Ethics is available on our website at http://www.acadia.com under the Corporate Governance section of our Investors page. Information contained in our website does not constitute a part of this report or our other filings with the SEC. We will promptly disclose on our website (i) the nature of any amendment to the policy that applies to our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions and (ii) the nature of any waiver, including an implicit waiver, from a provision of the policy that is granted to one of these specified individuals, the name of such person who is granted the waiver and the date of the waiver. Stockholders may request a free copy of the Code of Business Conduct and Ethics from our compliance department c/o Acadia Pharmaceuticals Inc., 12830 El Camino Real, Suite 400, San Diego, CA 92130.
Item 11. Executive Compensation.
The information required by this Item will be set forth in the section headed “Executive Compensation” in our Proxy Statement and is incorporated in this report by reference.
The information required by Item 402(x) of Regulation S-K shall be set forth in the section headed “Policies and practices related to the grant of certain equity awards close in time to the release of material nonpublic information” in our Proxy Statement and is incorporated in this report by reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
The information required by this Item will be set forth in the section headed “Security Ownership of Certain Beneficial Owners and Management” in our Proxy Statement and is incorporated in this report by reference.
Information regarding our equity compensation plans will be set forth in the section headed “Equity Compensation Plan Information” in our Proxy Statement and is incorporated in this report by reference.
Item 13. Certain Relationships and Related Transactions, and Director Independence.
The information required by this Item will be set forth in the section headed “Transactions With Related Persons” in our Proxy Statement and is incorporated in this report by reference.
Item 14. Principal Accountant Fees and Services.
The information required by this Item will be set forth under the proposal captioned “Ratification of Selection of Independent Registered Public Accounting Firm” in our Proxy Statement and is incorporated in this report by reference.
PART IV
Item 15. Exhibits and Financial Statement Schedules.
(a) Documents filed as part of this report.
1. The following financial statements of Acadia Pharmaceuticals Inc. and Report of Ernst & Young LLP, Independent Registered Public Accounting Firm, are included in this report:
2. List of financial statement schedules:
Schedule II – Valuation and Qualifying Accounts
Schedules not listed above have been omitted because they are not applicable or the required information is shown in the financial statements or notes thereto.
3. List of Exhibits required by Item 601 of Regulation S-K. See part (b) below.
(b) Exhibits.
| | |
Exhibit Number | | Description |
| | |
3.1 | | Amended and Restated Certificate of Incorporation, as Amended (incorporated by reference to Exhibit 3.1 to the Registrant’s Quarterly Report on Form 10-Q, filed August 6, 2015). |
| | |
3.2 | | Certificate of Amendment of Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.2 to the Registrant’s Annual Report on Form 10-K, filed February 25, 2021). |
| | |
3.3 | | Amended and Restated Bylaws (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K, filed September 12, 2013). |
| | |
4.1 | | Form of common stock certificate of the Registrant (incorporated by reference to Exhibit 4.1 to Registration Statement No. 333-52492). |
| | |
4.2 | | Form of Amended and Restated Warrant to Purchase Common Stock (incorporated by reference to Exhibit 4.2 to the Registrant’s Annual Report on Form 10-K, filed February 26, 2019). |
| | |
4.3 | | Description of the Registrant’s Common Stock (incorporated by reference to Exhibit 4.3 to the Registrant’s Annual Report on Form 10-K, filed February 27, 2020). |
| | |
10.1a | | Form of Indemnity Agreement for directors and officers (incorporated by reference to Exhibit 10.1 to Registration Statement No. 333-113137). |
| | |
10.2a | | 2004 Equity Incentive Plan and forms of agreement thereunder (incorporated by reference to Exhibit 10.3 to Registration Statement No. 333-113137). |
| | |
10.3a | | 2010 Equity Incentive Plan, as amended (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-K, filed August 9, 2022). |
| | |
10.4a | | Forms of Nonstatutory Stock Option Grant Notice and Stock Option Agreement under Acadia Pharmaceuticals Inc. 2010 Equity Incentive Plan (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q, filed May 9, 2024). |
| | |
| | |
10.5a | | Forms of Restricted Stock Unit Grant Notice and Restricted Stock Unit Award Agreement under Acadia Pharmaceuticals Inc. 2010 Equity Incentive Plan (incorporated by reference to Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q, filed May 9, 2024). |
| | |
10.6a | | Forms of Performance Stock Unit Grant Notice and Performance Stock Unit Award Agreement under Acadia Pharmaceuticals Inc. 2010 Equity Incentive Plan (incorporated by reference to Exhibit 10.3 to the Registrant’s Quarterly Report on Form 10-Q, filed May 9, 2024). |
| | |
10.7a | | 2004 Employee Stock Purchase Plan, as amended (incorporated by reference to Exhibit 99.1 to the Registrant’s Current Report on Form 8-K, filed June 29, 2020). |
| | |
10.8a | | Offerings under the 2004 Employee Stock Purchase Plan, as amended (incorporated by reference to Exhibit 10.6 to the Registrant’s Annual Report on Form 10-K, filed February 28, 2017). |
| | |
10.9a | | Employment Agreement, dated September 1, 2015, between the Registrant and Stephen Davis (incorporated by reference to Exhibit 99.1 to the Registrant’s Current Report on Form 8-K, filed September 3, 2015). |
| | |
10.10a | | Executive Transition Agreement, dated September 23, 2024, by and between the Registrant and Stephen R. Davis (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on September 23, 2024). |
| | |
10.11a | | Employment Offer Letter, dated June 26, 2018, between the Registrant and Brendan Teehan (incorporated by reference to Exhibit 10.9 to the Registrant’s Annual Report on Form 10-K, filed March 1, 2022). |
| | |
10.12a | | Employment Agreement, dated September 23, 2024, by and between the Registrant and Catherine Owen Adams (incorporated by reference to Exhibit 10.2 to the Registrant’s Current Report on Form 8-K, filed on September 23, 2024). |
| | |
10.13a | | Employment Offer Letter, dated April 28, 2020, between the Registrant and Mark Schneyer (incorporated by reference to Exhibit 10.11 to the Registrant’s Annual Report on Form 10-K, filed March 1, 2022). |
| | |
10.14a | | Employment Offer Letter, dated February 22, 2024, between the Registrant and Elizabeth H. Z. Thompson. (incorporated by reference to Exhibit 10.9 of the Registrant’s Quarterly Report on Form 10-Q, filed August 7, 2024). |
| | |
10.15a | | Employment Offer Letter, dated January 12, 2024, between the Registrant and Jennifer Rhodes (incorporated by reference to Exhibit 10.13 to the Registrant’s Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.16a, b | | Employment Offer Letter, dated November 15, 2024, by and between the Registrant and Thomas Garner. |
| | |
10.17a | | Non-Employee Director Compensation Policy (incorporated by reference to Exhibit 99.2 to the Registrant’s Current Report on Form 8-K, filed June 29, 2020). |
| | |
10.18a | | Management Severance Benefit Plan (incorporated by reference to Exhibit 99.1 to the Registrant’s Current Report on Form 8-K, filed December 15, 2015). |
| | |
10.19a | | Amended and Restated Change in Control Severance Benefit Plan (incorporated by reference to Exhibit 99.2 to the Registrant’s Current Report on Form 8-K, filed December 15, 2015). |
| | |
10.20b | | Master Manufacturing Services Agreement and Product Agreement, dated August 3, 2015, by and between the Registrant and Patheon Pharmaceuticals Inc. (incorporated by reference to Exhibit 10.17 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.21b | | First Amendment to Product Agreement, dated April 25, 2016, by and between the Registrant and Patheon Pharmaceuticals Inc. (incorporated by reference to Exhibit 10.18 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.22b | | Second Amendment to Product Agreement, dated October 6, 2016, by and between the Registrant and Patheon Pharmaceuticals Inc. (incorporated by reference to Exhibit 10.19 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.23b | | Third Amendment to Product Agreement, dated December 11, 2017, by and between the Registrant and Patheon Pharmaceuticals Inc. (incorporated by reference to Exhibit 10.20 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
| | |
10.24b | | Master Services Agreement, dated December 15, 2016, by and between Acadia Pharmaceuticals GmbH and Siegfried AG and its affiliates, and Attachment #1, Attachment #2 and Attachment #3 (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q, filed November 3, 2022). |
| | |
10.25b | | Change Order #1 to Master Services Agreement Attachment #1, dated January 3, 2017, by and between Acadia Pharmaceuticals GmbH and Siegfried AG (incorporated by reference to Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q, filed November 3, 2022). |
| | |
10.26b | | Attachment #4, Attachment #5 and Attachment #6, each dated May 12, 2017, to the Master Services Agreement, dated December 15, 2016, by and between Acadia Pharmaceuticals GmbH and Siegfried AG and its affiliates (incorporated by reference to Exhibit 10.3 to the Registrant’s Quarterly Report on Form 10-Q, filed November 3, 2022). |
| | |
10.27b | | Attachment #7, dated September 30, 2020, to the Master Services Agreement, dated December 15, 2016, by and between Acadia Pharmaceuticals GmbH and Siegfried AG and its affiliates (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly report on Form 10-Q, filed November 4, 2020). |
| | |
10.28 | | Registration Rights Agreement, dated January 6, 2016, between the Registrant and the investors listed on Schedule A thereto (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K, filed January 7, 2016). |
| | |
10.29 | | Assignment of Brann Intellectual Property Rights, dated January 29, 1997, by Mark R. Brann in favor of the Registrant (incorporated by reference to Exhibit 10.17 to Registration Statement No. 333-52492). |
| | |
10.30b | | License Agreement, dated August 6, 2018, by and between the Registrant and Neuren Pharmaceuticals Ltd. |
| | |
10.31b | | Lease Agreement, effective October 4, 2018, by and between the Registrant and Kilroy Realty, L.P. |
| | |
10.32b | | First Amendment to Office Lease, dated December 23, 2019, between the Registrant and Kilroy Realty, L.P. (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly report on Form 10-Q, filed May 8, 2020). |
| | |
10.33b | | Second Amendment to Office Lease, dated March 12, 2020, between the Registrant and Kilroy Realty, L.P. (incorporated by reference to Exhibit 10.2 to the Registrant’s Quarterly report on Form 10-Q, filed May 8, 2020). |
| | |
10.34a | | Acadia Pharmaceuticals Inc. 2023 Inducement Plan (incorporated by reference to Exhibit 99.1 to Registration Statement No. 333-269611). |
| | |
10.35a | | Forms of Stock Option Grant Notice and Stock Option Agreement under Acadia Pharmaceuticals Inc. 2023 Inducement Plan (incorporated by reference to Exhibit 99.2 to Registration Statement No. 333-269611). |
| | |
10.36a | | Forms of Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement under Acadia Pharmaceuticals Inc. 2023 Inducement Plan (incorporated by reference to Exhibit 99.3 to Registration Statement No. 333-269611). |
| | |
10.37b | | Lease Agreement, effective May 15, 2018, by and between the Registrant and Boston Properties Limited Partnership (incorporated by reference to Exhibit 10.33 to the Registrant’s Annual Report on Form 10-K, filed February 28, 2023). |
| | |
10.38b | | Master Commercial Supply Agreement, dated November 16, 2022, by and between the Registrant and Corden Pharma Bergamo S.p.A. (incorporated by reference to Exhibit 10.36 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.39b | | Commercial Supply Agreement, dated December 15, 2021, by and between the Registrant and F.I.S. Fabbrica Italiana Sintetici S.p.A. (incorporated by reference to Exhibit 10.36 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.40b | | Product Agreement, effective May 1, 2022, by and between the Registrant and Patheon Pharmaceuticals Inc. (incorporated by reference to Exhibit 10.37 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
10.41b | | Commercial Supply Agreement, dated March 1, 2023, by and between the Registrant and CoreRx Inc., as amended by Amendment No. 1, dated August 1, 2023 (incorporated by reference to Exhibit 10.38 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
| | |
10.42b | | Joint Venture and License Agreement, dated July 13, 2023, by and between the Registrant and Neuren Pharmaceuticals Ltd. (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q, filed August 3, 2023). |
| | |
10.43b | | Asset Purchase Agreement dated November 5, 2024 with respect to sale of a priority review voucher. |
| | |
10.44a | | Acadia Pharmaceuticals Inc. 2024 Equity Incentive Plan (incorporated by reference to Exhibit 99.1 to Registration Statement No. 333-279784). |
| | |
10.45a | | Forms of Standard Stock Option Grant Notice and Stock Option Agreement under the Acadia Pharmaceuticals Inc. 2024 Equity Incentive Plan (incorporated by reference to Exhibit 99.2 to Registration Statement No. 333-279784). |
| | |
10.46a | | Forms of Standard Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement under the Acadia Pharmaceuticals Inc. 2024 Equity Incentive Plan (incorporated by reference to Exhibit 99.3 to Registration Statement No. 333-279784). |
| | |
10.47a | | Forms of Director Stock Option Grant Notice and Stock Option Agreement under the Acadia Pharmaceuticals Inc. 2024 Equity Incentive Plan (incorporated by reference to Exhibit 99.4 to Registration Statement No. 333-279784). |
| | |
10.48a | | Forms of Director Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement under the Acadia Pharmaceuticals Inc. 2024 Equity Incentive Plan (incorporated by reference to Exhibit 99.5 to Registration Statement No. 333-279784). |
| | |
10.49a | | Acadia Pharmaceuticals Inc. 2024 Inducement Plan (incorporated by reference to Exhibit 99.1 to Registration Statement No. 333-282295). |
| | |
10.50a | | Forms of Standard Stock Option Grant Notice and Stock Option Agreement under the Acadia Pharmaceuticals Inc. 2024 Inducement Plan (incorporated by reference to Exhibit 99.2 to Registration Statement No. 333-282295). |
| | |
10.51a | | Forms of Standard Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement under the Acadia Pharmaceuticals Inc. 2024 Inducement Plan (incorporated by reference to Exhibit 99.3 to Registration Statement No. 333-282295). |
| | |
10.52a | | Forms of Standard Performance Stock Unit Grant Notice and Performance Stock Unit Agreement under the Acadia Pharmaceuticals Inc. 2024 Inducement Plan (incorporated by reference to Exhibit 10.6 to the Registrant’s Quarterly Report on Form 10-Q, filed November 7, 2024). |
| | |
19.1 | | Acadia Pharmaceuticals Inc. Insider Trading Policy. |
| | |
21.1 | | List of subsidiaries of the Registrant. |
| | |
23.1 | | Consent of Independent Registered Public Accounting Firm. |
| | |
24.1 | | Power of Attorney (see signature page hereto). |
| | |
31.1 | | Certification of Catherine Owen Adams, Chief Executive Officer, pursuant to Rule 13a-14(a) or Rule 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| | |
31.2 | | Certification of Mark Schneyer, Executive Vice President and Chief Financial Officer, pursuant to Rule 13a-14(a) or Rule 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| | |
32.1c | | Certification of Catherine Owen Adams, Chief Executive Officer, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| | |
32.2c | | Certification of Mark Schneyer, Executive Vice President and Chief Financial Officer, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| | |
97.1 | | Acadia Pharmaceuticals Inc. Dodd-Frank Clawback Policy (incorporated by reference to Exhibit 97.1 to the Registrants Annual Report on Form 10-K, filed February 28, 2024). |
| | |
| | |
101 | | The following financial statements from this Annual Report, formatted in iXBRL (Inline Extensible Business Reporting Language), are filed herewith: (i) Consolidated Balance Sheets, (ii) Consolidated Statements of Operations, (iii) Consolidated Statements of Comprehensive Loss, (iv) Consolidated Statements of Cash Flows, (v) Consolidated Statements of Stockholders’ Equity, and (vi) Notes to Consolidated Financial Statements. |
| | |
104 | | Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibit 101) |
a Indicates management contract or compensatory plan or arrangement.
b Pursuant to Item 601(b)(10) of Regulation S-K, certain portions of this exhibit have been omitted (indicated by “[***]” or “[…***…]”) because the Company has determined that the information is both not material and is the type that the Company treats as private or confidential.
c The information in Exhibits 32.1 and 32.2 shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act or the Exchange Act (including this Annual Report), unless the Registrant specifically incorporates the foregoing information into those documents by reference.
Item 16. Form 10-K Summary
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities and Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | |
| | Acadia Pharmaceuticals Inc. |
| | |
Date: February 27, 2025 | | /s/ CATHERINE OWEN ADAMS |
| | Catherine Owen Adams Chief Executive Officer (on behalf of the registrant and as the registrant’s Principal Executive Officer) |
KNOW ALL PERSONS BY THESE PRESENTS, that each individual whose signature appears below constitutes and appoints each of Catherine Owen Adams and Mark C. Schneyer his or her true and lawful attorney-in-fact and agent, with full power of substitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K, and to file the same, with all exhibits thereto and all documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agent full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that each said attorney-in-fact and agent, or his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities and Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | |
Signature | | Title | | Date |
| | | | |
/S/ CATHERINE OWEN ADAMS
Catherine Owen Adams | | Chief Executive Officer and Director (Principal Executive Officer) | | February 27, 2025 |
| | | | |
/S/ MARK C. SCHNEYER
Mark C. Schneyer | | Executive Vice President and Chief Financial Officer (Principal Financial Officer) | | February 27, 2025 |
| | | | |
/S/ JAMES K. KIHARA
James K. Kihara | | Senior Vice President, Finance (Principal Accounting Officer) | | February 27, 2025 |
| | | | |
/S/ STEPHEN R. BIGGAR
Stephen R. Biggar | | Chairman of the Board | | February 27, 2025 |
| | | | |
/S/ JULIAN C. BAKER
Julian C. Baker | | Director | | February 27, 2025 |
| | | | |
/S/ LAURA A. BREGE
Laura A. Brege | | Director | | February 27, 2025 |
| | | | |
/S/ JAMES M. DALY
James M. Daly | | Director | | February 27, 2025 |
| | | | |
/S/ ELIZABETH A. GAROFALO
Elizabeth A. Garofalo | | Director | | February 27, 2025 |
| | | | |
/S/ EDMUND P. HARRIGAN
Edmund P. Harrigan | | Director | | February 27, 2025 |
| | | | |
/S/ ADORA NDU
Adora Ndu | | Director | | February 27, 2025 |
| | | | |
/S/ DANIEL B. SOLAND
Daniel B. Soland | | Director | | February 27, 2025 |
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of
Acadia Pharmaceuticals Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Acadia Pharmaceuticals Inc. (the Company) as of December 31, 2024 and 2023, the related consolidated statements of operations, comprehensive income (loss), cash flows and stockholders’ equity for each of the three years in the period ended December 31, 2024, and the related notes and financial statement schedule listed in the Index at Item 15(a)2 (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2024 and 2023, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2024, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2024, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated February 27, 2025 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
| |
Medicare Part D sales rebate accruals |
Description of the Matter | As described in Note 2 to the consolidated financial statements under the caption “Revenue Recognition” the Company establishes provisions for sales rebates and discounts in the same period as the related sales occur. Estimated sales rebates for the purchase of NUPLAZID covered by Medicare Part D are included within accrued liabilities on the consolidated balance sheet. In order to establish these sales rebate accruals, the Company estimated its rebates based upon the identification of the product subject to a rebate, the historical and expected payor mix, the applicable price, rebate terms and the estimated lag time between the sale and payment of the rebate. Auditing the Medicare Part D sales rebate is complex because of the subjectivity of certain assumptions required to estimate the rebate liabilities and the amounts involved are material to the financial statements taken as a whole. In calculating the appropriate accrual amount, the Company considered historical Medicare Part D rebate payments as well as any significant changes in sales trends, the lag in payment timing, an evaluation of the current Medicare Part D laws and interpretations, the percentage of products that are sold via Medicare Part D, and product pricing. In deriving these estimates and assumptions, the Company used both internal and external sources of information to estimate product in the distribution channels, payor mix, prescription volumes and historical experience. Management supplemented its historical data analysis with qualitative adjustments based upon changes in rebate trends, rebate programs and contract terms, legislative changes, or other significant events which indicate a change in the reserve is appropriate. |
How We Addressed the Matter in Our Audit | We obtained an understanding evaluated the design and tested the operating effectiveness of controls over the Company’s sales rebate accruals for Medicare Part D rebates. This included testing controls over management’s review of the significant assumptions described above and inputs into the rebate calculations. For example, we tested controls over actual sales and the accuracy of forecasting expected utilization and payor mix. The testing was inclusive of management’s controls to evaluate the accuracy of its reserve judgments to actual rebates paid, rebate validation and processing, and controls to ensure that the data used to evaluate and support the significant assumptions was complete, accurate and, where applicable, verified to external data sources. To test the sales rebate accruals for Medicare Part D, our audit procedures included, among others, understanding and evaluating the significant assumptions and underlying data used in management’s calculations. Our testing of significant assumptions included a lookback analysis to evaluate the historical accuracy of management’s estimates by comparing actual rebates to previous estimates and performed sensitivity analyses over the subjective assumptions to evaluate the completeness of the reserves. As a part of our procedures, we evaluated the reasonableness of the Company’s assumptions considering recent sales trends and regulatory factors. |
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2015.
San Diego, California
February 27, 2025
ACADIA PHARMACEUTICALS INC.
CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share amounts)
| | | | | | | | |
| | December 31, | |
| | 2024 | | | 2023 | |
Assets | | | | | | |
Cash and cash equivalents | | $ | 319,589 | | | $ | 188,657 | |
Investment securities, available-for-sale | | | 436,404 | | | | 250,208 | |
Accounts receivable, net | | | 98,739 | | | | 98,267 | |
Interest and other receivables | | | 5,956 | | | | 4,083 | |
Inventory | | | 21,949 | | | | 35,819 | |
Prepaid expenses | | | 55,681 | | | | 39,091 | |
Total current assets | | | 938,318 | | | | 616,125 | |
Property and equipment, net | | | 4,215 | | | | 4,612 | |
Operating lease right-of-use assets | | | 46,571 | | | | 51,855 | |
Intangible assets, net | | | 119,782 | | | | 65,490 | |
Restricted cash | | | 8,770 | | | | 5,770 | |
Long-term inventory | | | 69,741 | | | | 4,628 | |
Other assets | | | 359 | | | | 476 | |
Total assets | | $ | 1,187,756 | | | $ | 748,956 | |
Liabilities and stockholders’ equity | | | | | | |
Accounts payable | | $ | 16,192 | | | $ | 17,543 | |
Accrued liabilities | | | 378,678 | | | | 236,711 | |
Total current liabilities | | | 394,870 | | | | 254,254 | |
Operating lease liabilities | | | 42,037 | | | | 47,800 | |
Other long-term liabilities | | | 18,056 | | | | 15,147 | |
Total liabilities | | | 454,963 | | | | 317,201 | |
Commitments and contingencies (Note 9) | | | | | | |
Stockholders’ equity: | | | | | | |
Preferred stock, $0.0001 par value; 5,000,000 shares authorized at December 31, 2024
and 2023; no shares issued and outstanding at December 31, 2024 and 2023 | | | — | | | | — | |
Common stock, $0.0001 par value; 225,000,000 shares authorized at December 31, 2024
and 2023; 166,708,856 shares and 164,650,219 shares issued and outstanding at
December 31, 2024 and 2023, respectively | | | 16 | | | | 16 | |
Additional paid-in capital | | | 2,936,871 | | | | 2,862,552 | |
Accumulated deficit | | | (2,204,386 | ) | | | (2,430,837 | ) |
Accumulated other comprehensive income | | | 292 | | | | 24 | |
Total stockholders’ equity | | | 732,793 | | | | 431,755 | |
Total liabilities and stockholders’ equity | | $ | 1,187,756 | | | $ | 748,956 | |
The accompanying notes are an integral part of these consolidated financial statements.
ACADIA PHARMACEUTICALS INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share amounts)
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Revenues | | | | | | | | | |
Product sales, net | | $ | 957,797 | | | $ | 726,437 | | | $ | 517,235 | |
Total revenues | | | 957,797 | | | | 726,437 | | | | 517,235 | |
Operating expenses | | | | | | | | | |
Cost of product sales | | | 81,841 | | | | 41,638 | | | | 10,166 | |
Research and development | | | 303,249 | | | | 351,619 | | | | 361,575 | |
Selling, general and administrative | | | 488,428 | | | | 406,559 | | | | 369,090 | |
Gain on sale of non-financial asset | | | (146,515 | ) | | | — | | | | — | |
Total operating expenses | | | 727,003 | | | | 799,816 | | | | 740,831 | |
Income (loss) from operations | | | 230,794 | | | | (73,379 | ) | | | (223,596 | ) |
Interest income, net | | | 25,458 | | | | 17,234 | | | | 6,610 | |
Other income | | | 1,823 | | | | 5,109 | | | | 3,542 | |
Income (loss) before income taxes | | | 258,075 | | | | (51,036 | ) | | | (213,444 | ) |
Income tax expense | | | 31,624 | | | | 10,250 | | | | 2,531 | |
Net income (loss) | | $ | 226,451 | | | $ | (61,286 | ) | | $ | (215,975 | ) |
Earnings (net loss) per share: | | | | | | | | | |
Basic | | $ | 1.37 | | | $ | (0.37 | ) | | $ | (1.34 | ) |
Diluted | | $ | 1.36 | | | $ | (0.37 | ) | | $ | (1.34 | ) |
Weighted average common shares outstanding: | | | | | | | | | |
Basic | | | 165,717 | | | | 163,819 | | | | 161,683 | |
Diluted | | | 166,362 | | | | 163,819 | | | | 161,683 | |
| | | | | | | | | |
The accompanying notes are an integral part of these consolidated financial statements.
ACADIA PHARMACEUTICALS INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME (LOSS)
(in thousands)
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Net income (loss) | | $ | 226,451 | | | $ | (61,286 | ) | | $ | (215,975 | ) |
Other comprehensive income (loss): | | | | | | | | | |
Unrealized gain/(loss) on investment securities | | | 362 | | | | 1,017 | | | | (789 | ) |
Foreign currency translation adjustments | | | (94 | ) | | | (18 | ) | | | 6 | |
Comprehensive income (loss) | | $ | 226,719 | | | $ | (60,287 | ) | | $ | (216,758 | ) |
The accompanying notes are an integral part of these consolidated financial statements.
ACADIA PHARMACEUTICALS INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Cash flows from operating activities | | | | | | | | | |
Net income (loss) | | $ | 226,451 | | | $ | (61,286 | ) | | $ | (215,975 | ) |
Adjustments to reconcile net income (loss) to net cash provided by (used in) operating activities: | | | | | | | | | |
Stock-based compensation | | | 67,049 | | | | 66,421 | | | | 68,201 | |
Amortization of premiums and accretion of discounts on investment securities | | | (9,304 | ) | | | (7,533 | ) | | | (2,736 | ) |
Amortization of intangible assets | | | 14,963 | | | | 4,093 | | | | — | |
Gain on sale of non-financial asset | | | (146,515 | ) | | | — | | | | — | |
Gain on strategic investment | | | — | | | | (5,109 | ) | | | (3,542 | ) |
Depreciation | | | 920 | | | | 1,459 | | | | 2,026 | |
Loss on sale of investment securities | | | — | | | | 524 | | | | — | |
Changes in operating assets and liabilities: | | | | | | | | | |
Accounts receivable, net | | | (472 | ) | | | (36,072 | ) | | | 2,171 | |
Interest and other receivables | | | (1,873 | ) | | | (3,198 | ) | | | 93 | |
Inventory | | | (49,550 | ) | | | (28,808 | ) | | | 2,415 | |
Prepaid expenses and other current assets | | | (16,590 | ) | | | (17,693 | ) | | | 2,494 | |
Operating lease right-of-use assets | | | 7,502 | | | | 5,769 | | | | 6,566 | |
Other assets | | | 117 | | | | (33 | ) | | | (48 | ) |
Accounts payable | | | (1,351 | ) | | | 4,797 | | | | 5,870 | |
Accrued liabilities | | | 71,061 | | | | 93,170 | | | | 24,306 | |
Operating lease liabilities | | | (7,598 | ) | | | (5,872 | ) | | | (7,916 | ) |
Long-term liabilities | | | 2,909 | | | | 6,073 | | | | 2,040 | |
Net cash provided by (used in) operating activities | | | 157,719 | | | | 16,702 | | | | (114,035 | ) |
Cash flows from investing activities | | | | | | | | | |
Purchases of investment securities | | | (505,095 | ) | | | (369,985 | ) | | | (363,174 | ) |
Sale and maturity of investment securities | | | 328,565 | | | | 429,780 | | | | 436,415 | |
Proceeds from sale of non-financial asset | | | 146,515 | | | | — | | | | — | |
Proceeds from sale of strategic investment | | | — | | | | 12,253 | | | | — | |
Net purchases of property and equipment | | | (523 | ) | | | (50 | ) | | | — | |
Intangible assets | | | — | | | | (40,000 | ) | | | — | |
Net cash (used in) provided by investing activities | | | (30,538 | ) | | | 31,998 | | | | 73,241 | |
Cash flows from financing activities | | | | | | | | | |
Proceeds from issuance of common stock, net of issuance costs | | | 6,845 | | | | 25,129 | | | | 8,199 | |
Net cash provided by financing activities | | | 6,845 | | | | 25,129 | | | | 8,199 | |
Effect of exchange rate changes on cash | | | (94 | ) | | | (18 | ) | | | 6 | |
Net increase (decrease) in cash, cash equivalents and restricted cash | | | 133,932 | | | | 73,811 | | | | (32,589 | ) |
Cash, cash equivalents and restricted cash | | | | | | | | | |
Beginning of year | | | 194,427 | | | | 120,616 | | | | 153,205 | |
End of year | | $ | 328,359 | | | $ | 194,427 | | | $ | 120,616 | |
Supplemental disclosure of cash flow information: | | | | | | | | | |
Cash paid for income taxes | | $ | 19,521 | | | $ | 5,850 | | | $ | 2,190 | |
Supplemental disclosure of noncash information: | | | | | | | | | |
Accrued inventory purchases | | $ | 1,268 | | | $ | — | | | $ | — | |
Accrued milestone and contingent payments in connection with asset acquisition | | $ | 98,838 | | | $ | 29,583 | | | $ | — | |
The accompanying notes are an integral part of these consolidated financial statements.
ACADIA PHARMACEUTICALS INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands, except share amounts)
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | Accumulated | | | | |
| | | | | | | | Additional | | | | | | Other | | | Total | |
| | Common Stock | | | Paid-in | | | Accumulated | | | Comprehensive | | | Stockholders’ | |
| | Shares | | | Amount | | | Capital | | | Deficit | | | Income (Loss) | | | Equity | |
Balances at December 31, 2021 | | | 161,012,695 | | | $ | 16 | | | $ | 2,694,646 | | | $ | (2,153,576 | ) | | $ | (192 | ) | | $ | 540,894 | |
Issuance of common stock from
exercise of stock options and
units | | | 721,652 | | | | — | | | | 3,705 | | | | — | | | | — | | | | 3,705 | |
Issuance of common stock
pursuant to employee stock
purchase plan | | | 330,525 | | | | — | | | | 4,494 | | | | — | | | | — | | | | 4,494 | |
Net loss | | | | | | — | | | | | | | (215,975 | ) | | | | | | (215,975 | ) |
Stock-based compensation | | | — | | | | — | | | | 68,078 | | | | — | | | | — | | | | 68,078 | |
Other comprehensive loss | | | — | | | | — | | | | | | | | | | (783 | ) | | | (783 | ) |
Balances at December 31, 2022 | | | 162,064,872 | | | | 16 | | | | 2,770,923 | | | | (2,369,551 | ) | | | (975 | ) | | | 400,413 | |
Issuance of common stock from
exercise of stock options and
units | | | 2,236,849 | | | | — | | | | 20,309 | | | | — | | | | — | | | | 20,309 | |
Issuance of common stock
pursuant to employee
stock purchase plan | | | 348,498 | | | | — | | | | 4,820 | | | | — | | | | — | | | | 4,820 | |
Net loss | | | — | | | | — | | | | — | | | | (61,286 | ) | | | — | | | | (61,286 | ) |
Stock-based compensation | | | — | | | | — | | | | 66,500 | | | | — | | | | — | | | | 66,500 | |
Other comprehensive income | | | — | | | | — | | | | — | | | | — | | | | 999 | | | | 999 | |
Balances at December 31, 2023 | | | 164,650,219 | | | | 16 | | | | 2,862,552 | | | | (2,430,837 | ) | | | 24 | | | | 431,755 | |
Issuance of common stock from
exercise of stock options and
units | | | 1,641,013 | | | | — | | | | 1,572 | | | | — | | | | — | | | | 1,572 | |
Issuance of common stock
pursuant to employee
stock purchase plan | | | 417,624 | | | | — | | | | 5,273 | | | | — | | | | — | | | | 5,273 | |
Net income | | | — | | | | — | | | | — | | | | 226,451 | | | | — | | | | 226,451 | |
Stock-based compensation | | | — | | | | — | | | | 67,474 | | | | — | | | | — | | | | 67,474 | |
Other comprehensive income | | | — | | | | — | | | | — | | | | — | | | | 268 | | | | 268 | |
Balances at December 31, 2024 | | | 166,708,856 | | | $ | 16 | | | $ | 2,936,871 | | | $ | (2,204,386 | ) | | $ | 292 | | | $ | 732,793 | |
The accompanying notes are an integral part of these consolidated financial statements.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Organization and Business
Acadia Pharmaceuticals Inc. (the Company), based in San Diego, California, is a biopharmaceutical company focused on the development and commercialization of innovative medicines to address unmet medical needs in central nervous system disorders and rare diseases.
In April 2016, the FDA approved the Company’s first drug, NUPLAZID® (pimavanserin), for the treatment of hallucinations and delusions associated with PDP. NUPLAZID became available for prescription in the United States in May 2016.
In March 2023, the FDA approved the Company’s second drug, DAYBUE™ (trofinetide), for the treatment of Rett syndrome. DAYBUE became available for prescription in the United States in April 2023.
In October 2024, Health Canada granted marketing authorization of DAYBUE™ (trofinetide) for the treatment of Rett syndrome in adult and pediatric patients 2 years of age and older.
2. Summary of Significant Accounting Policies
Significant accounting policies followed in the preparation of these financial statements are as follows:
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from these estimates.
Cash and Cash Equivalents
The Company considers all highly liquid investments with a maturity date at the date of purchase of three months or less to be cash equivalents.
The following table provides a reconciliation of cash, cash equivalents and restricted cash reported in the consolidated balance sheets that sum to the total of the same such amounts shown in the consolidated statements of cash flows (in thousands):
| | | | | | | | | | | | | | | | |
| | Twelve Months Ended
December 31, 2024 | | | Twelve Months Ended
December 31, 2023 | |
| | Beginning of period | | | End of period | | | Beginning of period | | | End of period | |
Cash and cash equivalents | | $ | 188,657 | | | $ | 319,589 | | | $ | 114,846 | | | $ | 188,657 | |
Restricted cash | | | 5,770 | | | | 8,770 | | | | 5,770 | | | | 5,770 | |
Total cash, cash equivalents and restricted
cash shown in the statements of cash flows | | $ | 194,427 | | | $ | 328,359 | | | $ | 120,616 | | | $ | 194,427 | |
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Investment Securities
Currently, all of the Company’s investment securities are debt securities. The Company has classified all of its investment securities as available-for-sale as the sale of such securities may be required prior to maturity to implement management strategies, and accordingly, carries these investments at fair value. Unrealized gains and losses, if any, are reported as a separate component of stockholders’ equity. The cost of investment securities classified as available-for-sale is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion are included in interest income. Realized gains and losses, if any, are also included in interest income. The cost of securities sold is based on the specific identification method.
Fair Value of Financial Instruments
The carrying values of the Company’s financial instruments, consisting of cash and cash equivalents, trade receivables, interest and other receivables, restricted cash, and accounts payable and accrued liabilities, approximate fair value due to the relative short-term nature of these instruments.
As disclosed in Note 4, the Company classifies its cash equivalents and available-for-sale investment securities within the fair value hierarchy as defined by authoritative guidance:
|
Level 1 Inputs — Quoted prices for identical instruments in active markets. |
|
Level 2 Inputs — Quoted prices for similar instruments in active markets; quoted prices for identical or similar instruments in markets that are not active; and model-derived valuations in which all significant inputs and significant value drivers are observable. |
|
Level 3 Inputs — Valuation derived from valuation techniques in which one or more significant inputs or significant value drivers are unobservable. |
Accounts Receivable
Accounts receivable are recorded net of customer allowances for distribution fees, prompt payment discounts, chargebacks, and credit losses. Allowances for distribution fees, prompt payment discounts and chargebacks are based on contractual terms. The Company estimated the current expected credit losses of its accounts receivable by assessing the risk of loss and available relevant information about the collectability, including historical credit losses, existing contractual payment terms, actual payment patterns of its customers, individual customer circumstances, and reasonable and supportable forecast of economic conditions expected to exist throughout the contractual life of the receivable. Based on its assessment, as of December 31, 2024 and 2023, the Company has determined that an allowance for credit loss was not required.
Inventory
Inventory is stated at the lower of cost or net realizable value under the first-in, first-out method (FIFO). The Company uses a combination of standard and actual costing methodologies to determine the cost basis for its inventories which approximates actual costs. Inventory consists of raw material, work in process, and finished goods, including third-party manufacturing costs, freight, and indirect overhead costs. The Company capitalizes inventory costs associated with its products upon regulatory approval when, based on management’s judgment, future commercialization is considered probable and the future economic benefit is expected to be realized; otherwise, such costs are expensed. Prior to FDA approval of NUPLAZID in April 2016 and DAYBUE and March 2023, all costs related to the manufacturing of NUPLAZID and DAYBUE were charged to research and development expense in the period incurred.
The Company periodically reviews inventory and reduces the carrying value of items to net realizable value for potentially excess, dated or obsolete inventory based on an analysis of forecasted demand compared to quantities on hand and any firm purchase orders, as well as product shelf life. During the years ended December 31, 2024, 2023 and 2022, the Company recorded charges of $0.5 million, $0.9 million and $0.6 million, respectively, to reduce certain finished goods and work in process inventory to its net realizable value.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Property and Equipment
Property and equipment are recorded at cost and depreciated over their estimated useful lives using the straight-line method. Leasehold improvements are amortized over the shorter of their estimated useful lives or the term of the lease by use of the straight-line method. Construction-in-process reflects amounts incurred for property, equipment or improvements that have not been placed in service. Maintenance and repair costs are expensed as incurred. When assets are retired or sold, the assets and accumulated depreciation are removed from the respective accounts and any gain or loss is recognized. Estimated useful lives by major asset category are as follows:
| | |
| | Useful Lives |
Machinery and equipment | | 5 to 7 years |
Computers and software | | 3 years |
Furniture and fixtures | | 10 years |
Impairment of Long-Lived Assets
The Company reviews its long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability is measured by a comparison of the carrying amount of an asset to estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the asset. Through December 31, 2024, no such impairment losses have been recorded by the Company.
License Fees and Royalties
The Company expenses amounts paid to acquire licenses associated with products under development when the ultimate recoverability of the amounts paid is uncertain and the technology has no alternative future use when acquired. Acquisitions of technology licenses are charged to expense or capitalized based upon management’s assessment regarding the ultimate recoverability of the amounts paid and the potential for alternative future use. The Company has determined that technological feasibility for its product candidates is reached when the requisite regulatory approvals are obtained to make the product available for sale.
Pursuant to the license agreement with Neuren, the Company has capitalized a total of $138.8 million as intangible assets following the FDA approval and sale of DAYBUE and sale of PRV, as disclosed in Note 9. The intangible assets are amortized on a straight-line basis over the estimated useful life of the licensed patents through early 2036. The Company recorded total amortization expense related to these intangible assets of $15.0 million and $4.1 million for the years ended December 31, 2024 and 2023, respectively. As of December 31, 2024, estimated future amortization expense related to the Company’s intangible assets was $10.9 million for each subsequent year.
Royalties incurred in connection with the Company’s license agreement with Neuren, as disclosed in Note 9, are expensed to cost of product sales as revenue from product sales is recognized.
Intangible Assets
Finite-lived intangible assets are recorded at cost, net of accumulated amortization, and, if applicable, impairment charges. Amortization of finite-lived intangible assets is recorded over the assets’ estimated useful lives on a straight-line basis or based on the pattern in which economic benefits are consumed, if reliably determinable. We review our finite-lived intangible assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. If such intangible assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the carrying amount of intangible the assets exceeds the estimated fair value of the intangible assets. No impairment loss was recorded on intangible assets during the years ended December 31, 2024 or 2023.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Acquisitions
The Company accounts for acquisitions of an asset or group of similar identifiable assets that do not meet the definition of a business as asset acquisition using the cost accumulation method, whereby the cost of the acquisition, including certain transaction costs, is allocated to the assets acquired on the basis of their relative fair values. No goodwill is recognized in an asset acquisition. Intangible assets acquired in an asset acquisition for use in research and development activities which have no alternative future use are expensed as in-process research and development on the acquisition date. Intangible assets acquired for use in research and development activities which have an alternative future use are capitalized as in-process research and development. Future costs to develop these assets are recorded to research and development expense as they are incurred. Contingent milestone payments associated with asset acquisitions are recognized when probable and estimable. These amounts are expensed to research and development if there is no alternative future use associated with the asset, or capitalized as an intangible asset if alternative future use of the asset exists.
Advertising Expense
Advertising costs are expensed when services are performed or goods are delivered. The Company incurred $21.1 million, $9.4 million and $5.5 million in advertising costs during the years ended December 31, 2024, 2023 and 2022, respectively and $1.3 million of advertising costs were capitalized as prepaid expenses at December 31, 2024. No advertising costs were capitalized as prepaid expenses at December 31, 2023 or 2022.
Revenue Recognition
The Company operates in one business segment. Results of its operations are reported on a consolidated basis for purposes of segment reporting, consistent with internal management reporting. Revenues consist of net product sales to customers, all of which are sales in the U.S. Revenues by product are as follows (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
NUPLAZID | | $ | 609,385 | | | $ | 549,248 | | | $ | 517,235 | |
DAYBUE | | | 348,412 | | | | 177,189 | | | | — | |
Product sales, net | | $ | 957,797 | | | $ | 726,437 | | | $ | 517,235 | |
Product Sales, Net
The Company accounts for contracts with its customers in accordance with Revenue from Contracts with Customers (Topic 606). The Company recognizes revenue when its customer obtains control of promised goods or services which is generally upon delivery. Revenues reflect the consideration which the Company expects to receive in exchange for those goods or services. To determine revenue recognition for arrangements that the Company determines are within the scope of Topic 606, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the Company satisfies a performance obligation. The Company only applies the five-step model to contracts when it is probable that the Company will collect the consideration it is entitled to in exchange for the goods or services it transfers to the customer. At contract inception, once the contract is determined to be within the scope of Topic 606, the Company assesses the goods or services promised within each contract, determines those that are performance obligations, and assesses whether each promised good or service is distinct. The Company then recognizes as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied. Payment terms differ by customer, but typically range from 31 to 35 days from the date of shipment. Revenue for the Company’s product sales has not been adjusted for the effects of a financing component as the Company expects, at contract inception, that the period between when the Company transfers control of the product and when the Company receives payment will be one year or less.
The Company’s product sales, net consist of U.S. sales of NUPLAZID and DAYBUE. NUPLAZID was approved by the FDA in April 2016 and the Company commenced shipments of NUPLAZID to SPs and SDs in late May 2016. SPs dispense product to a patient based on the fulfillment of a prescription and SDs sell product to government facilities, long-term care pharmacies, or in-patient hospital pharmacies. DAYBUE was approved by the FDA in March 2023 and the Company commenced shipments of DAYBUE to a single wholesale distributor in April 2023. Product shipping and handling costs are included in cost of product sales.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
The Company recognizes revenue from product sales at the net sales price (the “transaction price”) which includes estimates of variable consideration for which reserves for sales discounts and allowance are established and reflects each of these as either a reduction to the related account receivable or as an accrued liability, depending on how the amount payable is settled. Overall, these reserves reflect the Company’s best estimates of the amount of consideration to which the Company is entitled based on the terms of the contract. The amount of variable consideration that is included in the transaction price may be constrained, and is included in the net sales price only to the extent that it is probable that a significant reversal in the amount of the cumulative revenue recognized will not occur in a future period. Actual amounts of consideration ultimately received may differ from the Company’s estimates. If actual results in the future vary from estimates, the Company may need to adjust its estimates, which would affect net revenue in the period of adjustment. The following are the Company’s significant categories of sales discounts and allowances:
Distribution Fees: Distribution fees include distribution service fees paid to the SPs, SDs and wholesale distributor based on a contractually fixed percentage of the wholesale acquisition cost (WAC), fees for data, and prompt payment discounts. Distribution fees are recorded as an offset to revenue based on contractual terms at the time revenue from the sale is recognized.
Rebates: Allowances for rebates include mandated discounts under the Medicaid Drug Rebate Program and the Medicare Part D prescription drug benefit. Rebates are amounts owed after the final dispensing of the product to a benefit plan participant and are based upon contractual agreements with, or statutory requirements pertaining to, Medicaid and Medicare benefit providers. The allowance for rebates is based on statutory discount rates, estimated payor mix, and expected utilization. The Company’s estimates for expected utilization of rebates are based on historical data received from the SPs, SDs and single wholesale distributor since product launch. Rebates are generally invoiced and paid in arrears so that the accrual balance consists of an estimate of the amount expected to be incurred for the current quarter’s activity, plus an accrual balance for prior quarters’ unpaid rebates still estimated to be incurred. Allowances for rebates also include amounts due under the Inflation Reduction act of 2022 for Medicare Part D unit sales with applicable period AMP increases that outpace inflation over the benchmark period. The applicable period will be twelve months on October 1 of each year, with the initial applicable period beginning on October 1, 2022. The benchmark period AMP price is January 1, 2021 through September 30, 2021 for NUPLAZID and January 1, 2024 through December 31, 2024 for DAYBUE. The Company’s estimates are based Medicare Part D sales as a percentage of gross sales and the rate AMP for the current period will be in excess the benchmark period.
Chargebacks: Chargebacks are discounts and fees that relate to contracts with government and other entities purchasing from the SDs at a discounted price. The SDs charge back to the Company the difference between the price initially paid by the SDs and the discounted price paid to the SDs by these entities. The Company also incurs group purchasing organization fees for transactions through certain purchasing organizations. The Company estimates sales with these entities and accrues for anticipated chargebacks and organization fees, based on the applicable contractual terms.
Co-Payment Assistance: The Company offers co-payment assistance to commercially insured patients meeting certain eligibility requirements. Co-payment assistance is accrued for based on actual program participation and estimates of program redemption using data provided by third-party administrators.
Product Returns: Consistent with industry practice, the Company offers the SPs and SDs limited product return rights for damages, shipment errors, and expiring product; provided that the return is within a specified period around the product expiration date as set forth in the applicable individual distribution agreement. The Company does not allow product returns for product that has been dispensed to a patient. As the Company receives inventory reports from the SPs and SDs and has the ability to control the amount of product that is sold to the SPs and SDs, it is able to make a reasonable estimate of future potential product returns based on this on-hand channel inventory data and sell-through data obtained from the SPs and SDs. In arriving at its estimate for product returns, the Company also considers historical product returns, the underlying product demand, and industry data specific to the specialty pharmaceutical distribution industry.
Research and Development Expenses
Research and development expenses are charged to operations as incurred. Research and development expenses include costs associated with services provided by contract organizations for preclinical development, pre-commercialization manufacturing expenses, and clinical trials, salaries and related personnel expenses including stock-based compensation expense, and facilities and equipment expenses. The upfront consideration and transaction costs associated with acquired in-process research and development are also included in the research and development expenses.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
The Company accrues for costs incurred as the services are being provided by monitoring the status of the trial or services provided and the invoices received from its external service providers. When the Company makes payments in advance of services being provided, it records those amounts as prepaid expenses on its consolidated balance sheets and expense them as the services are rendered. In the case of clinical trials, a portion of the estimated cost normally relates to the projected cost to treat a patient in the trials, and this cost is recognized based on the number of patients enrolled in the trial. Other indirect costs are generally recognized on a straight-line basis over the estimated period of the study. As actual costs become known, the Company adjusts its accruals accordingly.
Concentration Risk
Financial instruments, which potentially subject the Company to concentrations of credit risk, principally consist of cash, cash equivalents, investment securities, accounts receivable, and restricted cash. The Company invests its excess cash primarily in money market funds, U.S. treasury notes, and high quality, marketable debt instruments of corporations and government sponsored enterprises in accordance with the Company’s investment policy. The Company’s investment policy defines allowable investments and establishes guidelines relating to credit quality, diversification, and maturities of its investments to preserve principal and maintain liquidity. All investment securities have a credit rating of at least Aa3/AA- or better, or P-1/A-1 or better, as determined by Moody’s Investors Service or Standard & Poor’s. Further, the Company specifies credit quality standards for its customers that are designed to limit the Company’s credit exposure to any single party.
The Company does not currently have any of its own manufacturing facilities, and therefore it depends on an outsourced manufacturing strategy for the production of NUPLAZID and DAYBUE for commercial use and for the production of its product candidates for clinical trials. For the production of NUPLAZID, the Company has contracts in place with two third-party manufacturers of commercial drug product and one third-party manufacturer of drug substance that is approved for the production of NUPLAZID API. For the production of DAYBUE, the Company has contracts in place with two third-party manufacturers of commercial drug product and two third-party manufacturers of drug substance that is approved for the production of DAYBUE API. Although there are potential sources of supply other than the Company’s existing suppliers, any new supplier would be required to qualify under applicable regulatory requirements.
The Company has entered into agreements for the distribution of NUPLAZID with a limited number of SPs and SDs, and all of the Company’s product sales of NUPLAZID are to these customers. The Company has also entered into agreements for the distribution of DAYBUE with a single wholesale distributor, and all of the Company’s product sales of DAYBUE and accounts receivable balance at December 31, 2024 are related to this customer. The following table summarizes customers that represent 10% or greater of our consolidated total gross revenues:
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Customer A | | | 34 | % | | | 23 | % | | * | |
Customer B | | | 14 | % | | | 17 | % | | | 25 | % |
Customer C | | | 13 | % | | | 15 | % | | | 17 | % |
Customer D | | | 12 | % | | | 14 | % | | | 21 | % |
Customer E | | | 10 | % | | | 10 | % | | | 11 | % |
__________________________________________
* Represents less than 10% and/or not a customer in the applicable year
The following table summarizes customers with amounts due that represent 10% or greater of our consolidated accounts receivable balance:
| | | | | | | | |
| | As of December 31, | |
| | 2024 | | | 2023 | |
Customer A | | | 42 | % | | | 33 | % |
Customer B | | | 13 | % | | | 14 | % |
Customer C | | | 14 | % | | | 12 | % |
Customer D | | | 12 | % | | | 14 | % |
Customer E | | * | | | | 10 | % |
__________________________________________
* Represents less than 10% and/or not a customer in the applicable year
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Stock-Based Compensation
We measure stock-based compensation expense for equity-classified awards, principally related to stock options, restricted stock units (RSUs), PSUs and stock purchase rights under our employee stock purchase plan (ESPP) based on the estimated fair value of the award on the date of grant.
The fair value of each employee stock option and each employee stock purchase right granted is estimated on the grant date under the fair value method using the Black-Scholes valuation model. The estimated fair value of each stock option and purchase right is then expensed on a straight-line basis over the requisite service period, which is generally the vesting period. The following weighted-average assumptions were used during these periods:
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Stock Options: | | | | | | | | | |
Expected volatility | | | 62 | % | | | 66 | % | | | 68 | % |
Risk-free interest rate | | | 4.1 | % | | | 3.9 | % | | | 2.9 | % |
Expected dividend yield | | | 0 | % | | | 0 | % | | | 0 | % |
Expected life of options in years | | | 5.5 | | | | 5.4 | | | | 5.4 | |
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Employee Stock Purchase Plan: | | | | | | | | | |
Expected volatility | | 46%-63% | | | 40%-67% | | | 62%-82% | |
Risk-free interest rate | | 4.3%-5.3% | | | 4.0%-5.3% | | | 1.5%-4.6% | |
Expected dividend yield | | | 0 | % | | | 0 | % | | | 0 | % |
Expected life in years | | 0.5-2.0 | | | 0.5-2.0 | | | 0.5-2.0 | |
Expected Volatility. The Company considers its historical volatility and implied volatility when determining the expected volatility.
Risk-Free Interest Rate. The Company determines its risk-free interest rate assumption based on the U.S. Treasury yield for obligations with contractual terms similar to the expected term of the stock option or purchase right being valued.
Expected Dividend Yield. The Company has never paid any dividends and currently has no plans to do so.
Expected Life. In determining the expected life for stock options, the Company considers, among other factors, its historical exercise experience to date as well as the mean time remaining to full vesting of all outstanding options and the mean time remaining to the end of the contractual term of all outstanding options. The estimated life for the Company’s employee stock purchase rights is based upon the terms of each offering period.
Forfeitures. The Company recognizes forfeitures as they occur.
The fair value of RSUs is estimated based on the closing market price of the Company’s common stock on the date of grant. RSUs generally vest over a four-year period. Certain RSUs also have an accelerated vesting clause based on specified market condition target and continued employment through a minimum vesting period. The fair value of RSUs expected to vest are recognized and amortized on a straight-line basis over the requisite service period, which is generally the vesting period. For those RSUs requiring satisfaction of both market and service conditions, the requisite service period is the longest of the explicit, implicit and derived service periods.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Through 2023, the Company granted PSUs that vest upon the achievement of certain pre-defined company-specific performance-based criteria. Expense related to these PSUs is recognized ratably over the expected performance period once the pre-defined performance-based criteria for vesting becomes probable and can vest up to 200 percent of the target number of shares granted. The fair value of these PSUs is estimated based on the closing market price of the Company’s common stock on the date of grant. Beginning in 2024, the structure of the PSU design was revised with a rTSR approach such that awards are earned for the Company’s rTSR performance over three-year measurement periods relative to a peer group of companies and the actual numbers of PSUs that will vest at the end of the performance period may be anywhere from zero to 150 percent of the target number of shares granted. The fair value of these PSUs is estimated using a Monte Carlo model because the performance target is based on a market condition. Expense related to these PSUs is recognized ratably over the three-year measurement period.
In connection with the departure of the former CEO, in September 2024 the Company incurred approximately $10.7 million in stock-based compensation expense as a result of accelerated equity award vesting and stock modifications under the former CEO’s severance plan.
The table below summarizes the total stock-based compensation expense included in the Company’s consolidated statements of operations for the periods presented (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Cost of product sales | | $ | 1,319 | | | $ | 1,007 | | | $ | 1,106 | |
Research and development | | | 14,100 | | | | 17,408 | | | | 22,580 | |
Sales, general and administrative | | | 51,630 | | | | 48,006 | | | | 44,515 | |
| | $ | 67,049 | | | $ | 66,421 | | | $ | 68,201 | |
Segment
The Company uses “the management approach” in determining reportable operating segments. The management approach considers the internal organization and reporting used by the Company’s Chief Operating Decision Maker (CODM) for making operating decisions and assessing performance as the source for determining the Company’s reportable segments. The Company determines and presents operating segments based on the information that is internally provided to the CEO who is considered the Company’s CODM, in accordance with ASC 280, Segment Reporting. The Company has determined that it operates as a single business segment, which is the development and commercialization of innovative medicines. Refer to Note 11 – Segment Reporting for further information related to the segment.
Income Taxes
Current income tax expense or benefit represents the amount of income taxes expected to be payable or refundable for the current year. A deferred income tax asset or liability is computed for the expected future impact of differences between the financial reporting and income tax bases of assets and liabilities and for the expected future tax benefit to be derived from tax credits and loss carryforwards. Deferred income tax expense or benefit represents the net change during the year in the deferred income tax asset or liability. Deferred tax assets are reduced by a valuation allowance when, in the opinion of management, it is more likely than not that some portion or all of the deferred tax assets will not be realized.
The Company recognizes the impact of a tax position in the financial statements only if that position is more likely than not to be sustained upon examination by taxing authorities, based on the technical merits of the position. Any interest and penalties related to uncertain tax positions will be reflected in income tax expense.
Earning (Net Loss) Per Share
Basic earnings (net loss) per share is calculated by dividing the net income (loss) by the weighted average number of common shares outstanding for the period, without consideration for common stock equivalents. Diluted net income (loss) per share is computed by dividing the net income (loss) by the weighted average number of common shares and common stock equivalents outstanding for the period determined using the treasury stock method. For purposes of diluted earnings (net loss) per share calculation, equity awards and employee stock purchase plan rights are considered to be common stock equivalents.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Net income (loss) - basic and diluted | | $ | 226,451 | | | $ | (61,286 | ) | | $ | (215,975 | ) |
Weighted average shares outstanding: | | | | | | | | | |
Basic | | | 165,717 | | | | 163,819 | | | | 161,683 | |
Effect of potentially dilutive common shares from: | | | | | | | | | |
Equity awards | | | 576 | | | | — | | | | — | |
Employee stock purchase plan rights | | | 69 | | | | — | | | | — | |
Diluted | | | 166,362 | | | | 163,819 | | | | 161,683 | |
| | | | | | | | | |
Earnings (net loss) per share: | | | | | | | | | |
Basic | | $ | 1.37 | | | $ | (0.37 | ) | | $ | (1.34 | ) |
Diluted | | $ | 1.36 | | | $ | (0.37 | ) | | $ | (1.34 | ) |
| | | | | | | | | |
Potentially dilutive shares excluded from per share amounts as their effect
would have been anti-dilutive | | | 18,233 | | | | 21,264 | | | | 21,185 | |
Recently Issued Accounting Standards
In November 2023, the FASB issued Accounting Standards Update No. 2023-07, Segment Reporting - Improvements to Reportable Segment Disclosures. The amendments require disclosure of incremental segment information on an annual and interim basis. The amendments also require companies with a single reportable segment to provide all disclosures required by this amendment and all existing segment disclosures in Accounting Standards Codification 280, Segment Reporting. The amendments are effective for fiscal years beginning after December 15, 2023, and interim periods beginning after December 15, 2024. The Company adopted this guidance for the year ended December 31, 2024. There was no significant impact of the adoption of this guidance on the Company’s consolidated financial statements.
In December 2023, the FASB issued Accounting Standards Update No. 2023-09, Income Taxes - Improvements to Income Tax Disclosures. The amendments require (i) enhanced disclosures in connection with an entity’s effective tax rate reconciliation and (ii) income taxes paid disaggregated by jurisdiction. The amendments are effective for annual periods beginning after December 15, 2024. The Company does not expect the adoption of the amendments to have a significant impact on its consolidated financial statements.
3. Investments
The carrying value and amortized cost of the Company’s investments, summarized by major security type, consisted of the following (in thousands):
| | | | | | | | | | | | | | | | |
| | December 31, 2024 | |
| | Amortized
Cost | | | Unrealized
Gains | | | Unrealized
Losses | | | Estimated
Fair Value | |
U.S. Treasury notes | | $ | 245,584 | | | $ | 319 | | | $ | — | | | $ | 245,903 | |
Government sponsored enterprise securities | | | 190,452 | | | | 157 | | | | (108 | ) | | | 190,501 | |
| | $ | 436,036 | | | $ | 476 | | | $ | (108 | ) | | $ | 436,404 | |
| | | | | | | | | | | | | | | | |
| | December 31, 2023 | |
| | Amortized
Cost | | | Unrealized
Gains | | | Unrealized
Losses | | | Estimated
Fair Value | |
U.S. Treasury notes | | $ | 75,315 | | | $ | 47 | | | $ | (28 | ) | | $ | 75,334 | |
Government sponsored enterprise securities | | | 174,867 | | | | 119 | | | | (112 | ) | | | 174,874 | |
| | $ | 250,182 | | | $ | 166 | | | $ | (140 | ) | | $ | 250,208 | |
The Company has classified all of its available-for-sale investment securities, including those with maturities beyond one year, as current assets on its consolidated balance sheets based on the highly liquid nature of the investment securities and because these investment securities are considered available for use in current operations. The following table summarizes the contract maturity of the available-for-sale securities we held as of December 31, 2024:
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
| | | | |
One year or less | | | 79 | % |
After one year but within two years | | | 21 | % |
Total | | | 100 | % |
As of December 31, 2023, all of the Company’s available-for-sale investment securities had contractual maturity dates of less than one year. The Company has classified all equity securities as other assets on its consolidated balance sheets.
At December 31, 2024 and 2023, the Company had 17 and 21 securities, respectively, in an unrealized loss position. The following table presents gross unrealized losses and fair value for those available-for-sale investments that were in an unrealized loss position as of December 31, 2024 and December 31, 2023, aggregated by investment category and length of time that individual securities have been in a continuous loss position (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | Less Than 12 Months | | | 12 Months or Greater | | | Total | | |
| | Estimated
Fair Value | | | Unrealized
Losses | | | Estimated
Fair Value | | | Unrealized
Losses | | | Estimated
Fair Value | | | Unrealized
Losses | | |
December 31, 2024 | | | | | | | | | | | | | | | | | | | |
Government sponsored enterprise securities | | $ | 84,390 | | | $ | (108 | ) | | $ | — | | | $ | — | | | $ | 84,390 | | | $ | (108 | ) | |
Total | | $ | 84,390 | | | $ | (108 | ) | | $ | — | | | $ | — | | | $ | 84,390 | | | $ | (108 | ) | |
December 31, 2023 | | | | | | | | | | | | | | | | | | | |
U.S. Treasury notes | | $ | 41,366 | | | $ | (28 | ) | | $ | — | | | $ | — | | | $ | 41,366 | | | $ | (28 | ) | |
Government sponsored enterprise securities | | | 108,587 | | | | (112 | ) | | | — | | | | — | | | | 108,587 | | | | (112 | ) | |
Total | | $ | 149,953 | | | $ | (140 | ) | | $ | — | | | $ | — | | | $ | 149,953 | | | $ | (140 | ) | |
During the first quarter of 2023, the Company made a sale of all of its investments in commercial paper. The proceeds from sales of these securities were $183.0 million and net realized losses from the related sales were $0.5 million. There were no sales of available-for-sale investment securities in 2024.
At each reporting date, the Company performs an evaluation of impairment to determine if any unrealized losses are the result of credit losses. Impairment is assessed at the individual security level. Factors considered in determining whether a loss resulted from a credit loss or other factors include the Company’s intent and ability to hold the investment until the recovery of its amortized cost basis, the extent to which the fair value is less than the amortized cost basis, the length of time and extent to which fair value has been less than the cost basis, the financial condition of the issuer, any historical failure of the issuer to make scheduled interest or principal payments, any changes to the rating of the security by a rating agency, any adverse legal or regulatory events affecting the issuer or issuer’s industry, any significant deterioration in economic conditions.
The Company does not intend to sell the investment in unrealized loss position and it is unlikely that the Company will be required to sell the investment before the recovery of its amortized cost basis. Based on its evaluation, the Company determined its year-to-date credit losses related to its available-for-sale securities were immaterial at December 31, 2024.
4. Fair Value Measurements
The Company’s investments include cash equivalents, available-for-sale investment securities consisting of money market funds, U.S. treasury notes, and marketable debt instruments of corporations and government sponsored enterprises in accordance with the Company’s investment policy, and equity investments. The Company’s investment policy defines allowable investment securities and establishes guidelines relating to credit quality, diversification, and maturities of its investments to preserve principal and maintain liquidity. All investment securities have a credit rating of at least Aa3/AA- or better, or P-1/A-1 or better, as determined by Moody’s Investors Service or Standard & Poor’s.
The Company’s cash equivalents, available-for-sale investment securities, and equity securities are classified within the fair value hierarchy as defined by authoritative guidance. The Company’s investment securities and equity securities classified as Level 1 are valued using quoted market prices. The Company obtains the fair value of its Level 2 financial instruments from third-party pricing services. The pricing services utilize industry standard valuation models whereby all significant inputs, including benchmark yields, reported trades, broker/dealer quotes, issuer spreads, bids, offers, or other
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
market-related data, are observable. The Company validates the prices provided by the third-party pricing services by reviewing their pricing methods and matrices and obtaining market values from other pricing sources. After completing the validation procedures, the Company did not adjust or override any fair value measurements provided by these pricing services as of December 31, 2024 and 2023, respectively.
In November 2021, the Company established a plan whereby substantially all full-time employees excluding executive management are eligible to receive a series of cash bonuses based on achievement of certain conditions as described in more detail in Note 6 to the consolidated financial statements. The Company estimated the fair value of the cash awards using a Monte Carlo simulation, which utilizes level 3 inputs such as volatility, probabilities of success, and other inputs that are not observable in active markets. The cash awards are required to be measured at fair value on a recurring basis each reporting period, with changes in the fair value recognized as compensation cost over the derived service period of the awards.
The Company has not transferred any investment securities between the classification levels.
The recurring fair value measurements of the Company’s cash equivalents, available-for-sale investment securities, and equity securities at December 31, 2024 and 2023 consisted of the following (in thousands):
| | | | | | | | | | | | | | | | |
| | | | | Fair Value Measurements at
Reporting Date Using | |
| | December 31, 2024 | | | Quoted Prices
in Active
Markets for
Identical
Assets
(Level 1) | | | Significant
Other
Observable
Inputs
(Level 2) | | | Significant
Unobservable
Inputs
(Level 3) | |
Assets | | | | | | | | | | | | |
Money market fund | | $ | 151,555 | | | $ | 151,555 | | | $ | — | | | $ | — | |
U.S. Treasury notes | | | 245,903 | | | | 245,903 | | | | — | | | | — | |
Government sponsored enterprise securities | | | 190,501 | | | | — | | | | 190,501 | | | | — | |
Total | | $ | 587,959 | | | $ | 397,458 | | | $ | 190,501 | | | $ | — | |
| | | | | | | | | | | | | | | | |
| | | | | Fair Value Measurements at
Reporting Date Using | |
| | December 31, 2023 | | | Quoted Prices
in Active
Markets for
Identical
Assets
(Level 1) | | | Significant
Other
Observable
Inputs
(Level 2) | | | Significant
Unobservable
Inputs
(Level 3) | |
Assets | | | | | | | | | | | | |
Money market fund | | $ | 64,586 | | | $ | 64,586 | | | $ | — | | | $ | — | |
U.S. Treasury notes | | | 75,334 | | | | 75,334 | | | | — | | | | — | |
Government sponsored enterprise securities | | | 174,874 | | | | — | | | | 174,874 | | | | — | |
Total | | $ | 314,794 | | | $ | 139,920 | | | $ | 174,874 | | | $ | — | |
| | | | | | | | | | | | |
Liabilities | | | | | | | | | | | | |
Cash awards | | $ | 4,506 | | | $ | — | | | $ | — | | | $ | 4,506 | |
Total | | $ | 4,506 | | | $ | — | | | $ | — | | | $ | 4,506 | |
Changes in estimated fair value of contingent cash awards during the twelve months ended December 31, 2024 are as follows (in thousands):
| | | | |
Balance as of December 31, 2023 | | $ | 4,506 | |
Vesting of awards | | | — | |
Expense forfeited | | | (392 | ) |
Change in fair value | | | (4,114 | ) |
Balance as of December 31, 2024 | | $ | — | |
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
5. Balance Sheet Details
Inventory consisted of the following (in thousands):
| | | | | | | | |
| | December 31, | |
| | 2024 | | | 2023 | |
Finished goods | | $ | 20,461 | | | $ | 5,001 | |
Work in process | | | 1,488 | | | | 4,134 | |
Raw material | | | 69,741 | | | | 31,312 | |
| | $ | 91,690 | | | $ | 40,447 | |
Reported as: | | | | | | |
Inventory | | $ | 21,949 | | | $ | 35,819 | |
Long-term inventory | | | 69,741 | | | | 4,628 | |
Total | | $ | 91,690 | | | $ | 40,447 | |
Amount reported as long-term inventory primarily consists of raw materials as of December 31, 2024 and 2023.
Property and equipment, net, consisted of the following (in thousands):
| | | | | | | | |
| | December 31, | |
| | 2024 | | | 2023 | |
Computers and software | | $ | 5,614 | | | $ | 5,873 | |
Leasehold improvements | | | 3,746 | | | | 3,746 | |
Furniture and fixtures | | | 4,549 | | | | 4,549 | |
Construction-in-process | | | 523 | | | | — | |
| | | 14,432 | | | | 14,168 | |
Accumulated depreciation | | | (10,217 | ) | | | (9,556 | ) |
| | $ | 4,215 | | | $ | 4,612 | |
Depreciation of property and equipment was $0.9 million, $1.5 million, and $2.0 million for the years ended December 31, 2024, 2023, and 2022, respectively. For the years ended December 31, 2024 and 2023, the Company did not retire any fully depreciated property and equipment. For the year ended December 31, 2022, the Company retired $0.1 million of fully depreciated property and equipment.
Accrued liabilities consisted of the following (in thousands):
| | | | | | | | |
| | December 31, | |
| | 2024 | | | 2023 | |
Accrued sales allowances | | $ | 148,280 | | | $ | 90,718 | |
Accrued contingent payments | | | 102,262 | | | | 29,583 | |
Accrued compensation and benefits | | | 36,551 | | | | 42,718 | |
Accrued consulting and professional fees | | | 27,435 | | | | 18,804 | |
Accrued research and development services | | | 27,181 | | | | 32,883 | |
Accrued taxes | | | 12,016 | | | | 1,564 | |
Current portion of lease liabilities | | | 9,958 | | | | 9,405 | |
Other | | | 14,995 | | | | 11,036 | |
| | $ | 378,678 | | | $ | 236,711 | |
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
6. Stockholders’ Equity
Stock Plans
2024 Equity Incentive Plan
The Company’s 2024 Equity Incentive Plan (the 2024 Plan) became effective upon approval of the stockholders in May 2024 and is a successor and continuation of the 2010 Equity Incentive Plan (the 2010 Plan). The 2024 Plan permits the grant of awards to employees, non-employee directors and consultants. In addition, the 2024 Plan permits the grant of stock options, stock appreciation rights, restricted stock awards, restricted stock unit awards, performance awards, and other awards. The 2024 Plan provides that, with limited exceptions, no award will vest until at least 12 months following the date of grant of the award; provided, however, that up to 5% of the aggregate number of shares that may be issued under the 2024 Plan may be subject to awards which do not meet such vesting requirements. The maximum term of any stock option or stock appreciation right awards under 2024 Plan is ten years. All shares that remained eligible for grant under the Company’s 2010 Equity Incentive Plan and 2023 Inducement Plan at the time of approval of the 2024 Plan were transferred to the 2024 Plan. At December 31, 2024, there were 13,798,565 shares of common stock available for new grants under the 2024 Plan.
2024 Inducement Plan
The Board adopted the Company’s 2024 Inducement Plan (Inducement Plan) in September 2024. The Inducement Plan permits the grant of stock options, stock appreciation rights, restricted stock awards, restricted stock unit awards, performance awards and other stock-related awards. Stock awards granted under the Inducement Plan may only be made to individuals who did not previously serve as employees or non-employee directors of the Company or an affiliate of the Company. In addition, stock awards must be approved by either a majority of the Company’s independent directors or the Compensation Committee. The terms of the Inducement Plan are otherwise substantially similar to the 2024 Plan. The maximum number of shares of Company common stock that may be issued under the Inducement Plan is 2,400,000 shares. At December 31, 2024, there were 1,491,346 shares available for new grants under the Inducement Plans.
2023 Inducement Plan
The Board adopted the Company’s 2023 Inducement Plan (2023 Inducement Plan) on February 1, 2023. The 2023 Inducement Plan permits the grant of stock options, stock appreciation rights, restricted stock awards, restricted stock unit awards, performance stock awards and other stock-related awards. Stock awards granted under the 2023 Inducement Plan may only be made to individuals who did not previously serve as employees or non-employee directors of the Company or an affiliate of the Company. In addition, stock awards must be approved by either a majority of the Company’s independent directors or the Compensation Committee. The terms of the 2023 Inducement Plan are otherwise substantially similar to the 2010 Plan. All shares that remained eligible for grant under the 2023 Inducement Plan at the time of approval of the 2024 Plan were transferred to the 2024 Plan.
2010 Equity Incentive Plan
The 2010 Plan, as amended to date, permits the grant of options to employees, directors and consultants. In addition, the 2010 Plan permits the grant of stock bonuses, rights to purchase restricted stock, and other stock awards. The exercise price of options granted under the 2010 Plan cannot be less than 100 percent of the fair market value of the common stock on the date of grant and the maximum term of any option is 10 years. Options granted under the 2010 Plan generally vest over a four-year period. All shares that remained eligible for grant under the Company’s 2004 Equity Incentive Plan (the 2004 Plan) at the time of approval of the 2010 Plan were transferred to the 2010 Plan. In June 2015, June 2016, June 2017, June 2018, June 2019 and June 2022, the Company’s stockholders approved amendments to its 2010 Plan to, among other things, increase the aggregate number of shares of common stock authorized for issuance under the plan by 5,000,000 shares, 3,000,000 shares, 5,500,000 shares, 6,700,000 shares, 8,300,000 shares and 6,000,000 shares, respectively. All shares that remained eligible for grant under the 2010 Plan at the time of approval of the 2024 Plan were transferred to the 2024 Plan.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Employee Stock Purchase Plan
The Company’s 2004 Employee Stock Purchase Plan (the Purchase Plan) became effective upon the closing of the Company’s initial public offering in June 2004. In June 2016, June 2019 and June 2020, the Company’s stockholders approved an amendment to the Purchase Plan to, among other things, increase the aggregate number of shares of common stock authorized for issuance under the Purchase Plan by 400,000 shares, 600,000 shares and 3,000,000 shares, respectively. At December 31, 2024, a total of 5,525,000 shares of common stock had been reserved for issuance under the Purchase Plan. At December 31, 2024, 1,713,498 shares of common stock remained available for issuance pursuant to the Purchase Plan. Eligible employees who elect to participate in an offering under the Purchase Plan may have up to 15 percent of their earnings withheld, subject to certain limitations, to purchase shares of common stock pursuant to the Purchase Plan. The price of common stock purchased under the Purchase Plan is equal to 85 percent of the lower of the fair market value of the common stock at the commencement date of each offering period or the relevant purchase date.
Stock Option Activity
The equity plans provided for the grant of options to employees, directors and consultants. The exercise price of options granted under the equity plans were at 100 percent of the fair market value of the common stock on the date of grant and the maximum term of any option was 10 years. Options granted under the equity plans generally vested over a four-year period.
The following table summarizes the Company’s stock option activity under all equity plans during the year ended December 31, 2024:
| | | | | | | | | | | | | | | | |
| | Number of
Shares | | | Weighted
Average
Exercise
Price | | | Weighted
Average
Remaining
Contractual
Term
(years) | | | Aggregate
Intrinsic Value
(in thousands) | |
Outstanding at December 31, 2023 | | | 16,626,029 | | | $ | 28.75 | | | | | | | |
Granted | | | 3,874,498 | | | $ | 17.81 | | | | | | | |
Exercised | | | (83,743 | ) | | $ | 18.77 | | | | | | | |
Cancelled/forfeited | | | (2,897,930 | ) | | $ | 25.57 | | | | | | | |
Outstanding at December 31, 2024 | | | 17,518,854 | | | $ | 26.90 | | | | 5.4 | | | $ | 6,998 | |
Exercisable at December 31, 2024 | | | 12,075,917 | | | $ | 30.28 | | | | 3.9 | | | $ | 2,252 | |
The aggregate intrinsic value of options exercisable as of December 31, 2024 is calculated as the difference between the exercise price of the underlying options and the closing market price of the Company’s common stock on that date, which was $18.35 per share. The aggregate intrinsic value of options exercised during the years ended December 31, 2024, 2023, and 2022 was approximately $0.3 million, $7.9 million, and $1.7 million, respectively, determined as of the date of exercise. The Company received approximately $1.6 million, $20.3 million and $3.7 million in cash from options exercised during the years ended December 31, 2024, 2023 and 2022, respectively.
The weighted average per share fair value of options granted during the years ended December 31, 2024, 2023, and 2022 was approximately $10.42, $13.25, and $13.66, respectively. As of December 31, 2024, total unrecognized compensation cost related to stock options was approximately $53.6 million and the weighted average period over which this cost is expected to be recognized is approximately 2.8 years.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Restricted Stock Unit Activity
The following table summarizes the Company’s RSUs during the year ended December 31, 2024:
| | | | | | | | | | | | |
| | Number of
Shares | | | Weighted
Average Grant
Date Fair Value | | | Aggregate
Intrinsic Value
(in thousands) | |
Unvested at December 31, 2023 | | | 2,650,917 | | | $ | 24.66 | | | | |
Granted | | | 1,465,142 | | | $ | 17.80 | | | | |
Vested | | | (928,071 | ) | | $ | 26.75 | | | | |
Cancelled/forfeited | | | (474,488 | ) | | $ | 22.29 | | | | |
Unvested at December 31, 2024 | | | 2,713,500 | | | $ | 20.65 | | | $ | 49,793 | |
The total fair value of RSUs that vested during the years ended December 31 2024, 2023 and 2022 was $17.3 million, $12.6 million and $10.2 million, respectively. As of December 31, 2024, total unrecognized compensation cost related to RSUs was approximately $39.5 million and the weighted average period over which this cost is expected to be recognized is approximately 2.5 years.
Performance Stock Unit Activity
The following table summarizes the Company’s PSUs during the year ended December 31, 2024:
| | | | | | | | | | | | |
| | Number of
Shares | | | Weighted
Average Grant
Date Fair Value | | | Aggregate
Intrinsic Value
(in thousands) | |
Unvested at December 31, 2023 | | | 1,734,828 | | | $ | 26.68 | | | | |
Granted | | | 615,743 | | | $ | 21.82 | | | | |
Vested | | | (629,199 | ) | | $ | 26.15 | | | | |
Cancelled/forfeited | | | (500,582 | ) | | $ | 25.41 | | | | |
Unvested at December 31, 2024(1) | | | 1,220,790 | | | $ | 25.02 | | | $ | 22,401 | |
__________________________________________
(1) The unvested balance consisted of 148,643 RSUs that have an accelerated vesting clause based on specified market condition target and continued employment through a minimum vesting period, 488,976 PSUs that vest upon achievement of certain pre-defined company-specific performance-based targets and 583,171 that vest based on the Company’s rTSR performance over a three-year measurement period.
The total fair value of PSUs that vested during the years ended December 31 2024 and 2023 was $10.2 million and $13.3 million, respectively. No PSUs vested during the year ended December 31, 2022. As of December 31, 2024, total unrecognized compensation cost related to PSUs was approximately $7.3 million and the weighted average remaining contractual term was 1.8 years.
Contingent Cash Awards
In November 2021, the Company established a plan whereby substantially all full-time employees excluding executive management are eligible to receive a series of cash bonuses over certain periods based on continued employment and the Company’s stock price reaching a pre-specified target. The maximum potential payout of the cash awards at the grant date was $15.1 million. The Company has determined that the cash awards were classified as liabilities pursuant to ASC Topic 718, Compensation – Stock Compensation. The Company estimates the fair value of the awards at each reporting period using the Monte Carlo simulation, which is recognized as compensation cost over the derived service period. Total fair value of the awards at the grant date was $4.4 million. The awards were forfeited in November 2024 as the Company’s stock price did not reach the pre-specified target and the Company recorded a reversal of $4.5 million of compensation expense related to the awards during the year ended December 31, 2024. During the year ended December 31, 2023, the awards had a total fair value of $5.2 million and the Company recorded a total of $3.6 million of compensation cost related to the awards.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
7. 401(k) Plan
Effective January 1997, the Company established a deferred compensation plan (the 401(k) Plan) pursuant to Section 401(k) of the Internal Revenue Code of 1986, as amended (the Code), whereby substantially all employees are eligible to contribute up to 60 percent of their pretax earnings, not to exceed amounts allowed under the Code. The Company makes discretionary contributions to the 401(k) Plan equal to 100 percent of each employee’s pretax contributions up to 5 percent of his or her eligible compensation, subject to limitations under the Code. The Company’s total contributions to the 401(k) Plan were $6.9 million, $6.1 million, and $5.1 million for the years ended December 31, 2024, 2023, and 2022, respectively.
8. Income Taxes
Domestic and foreign pre-tax income (loss) is as follows (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Domestic | | $ | 95,845 | | | $ | (100,215 | ) | | $ | (233,216 | ) |
Foreign | | | 162,230 | | | | 49,179 | | | | 19,772 | |
| | $ | 258,075 | | | $ | (51,036 | ) | | $ | (213,444 | ) |
The income tax provision consists of the following (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Current provision: | | | | | | | | | |
Federal | | $ | 19,542 | | | $ | 5,440 | | | $ | — | |
State | | | 12,064 | | | | 4,805 | | | | 2,531 | |
Foreign | | | 18 | | | | 5 | | | | — | |
Total current provision | | | 31,624 | | | | 10,250 | | | | 2,531 | |
Total income tax provision | | $ | 31,624 | | | $ | 10,250 | | | $ | 2,531 | |
At December 31, 2024, the Company had federal, state, and foreign net operating loss (NOL) carryforwards of approximately $130.2 million, $453.9 million, and $475.2 million, respectively. The Company recognized federal and state income tax provisions of $31.6 million, $10.3 million and $2.5 million for the years ended December 31, 2024, 2023 and 2022, respectively. These tax liabilities were primarily associated with federal and state liabilities in excess of net operating losses in the current year and an increase in federal and state reserves related to uncertain tax positions. Utilization of the domestic NOL and research and development (R&D) credit carryforwards may be subject to a substantial annual limitation due to ownership change limitations that have occurred or that could occur in the future, as required by Section 382 of the Code, as well as similar state and foreign provisions. These ownership changes may limit the amount of NOL and R&D credit carryforwards that can be utilized annually to offset future taxable income and tax, respectively. In general, an “ownership change” as defined by Section 382 of the Code results from a transaction or series of transactions over a three-year period resulting in an ownership change of more than 50 percentage points of the outstanding stock of a company by certain stockholders or public groups.
The Company previously completed a study to assess whether an ownership change, as defined by Section 382 of the Code, had occurred from the Company’s formation through December 31, 2013. Based upon this study, the Company determined that several ownership changes had occurred. Accordingly, the Company reduced its deferred tax assets related to the federal NOL carryforwards and the federal R&D credit carryforwards that are anticipated to expire unused as a result of these ownership changes. These tax attributes were excluded from deferred tax assets with a corresponding reduction of the valuation allowance with no net effect on income tax expense or the effective tax rate. The Company completed a study through December 31, 2024 and concluded no additional ownership changes occurred. Future ownership changes may further limit the Company’s ability to utilize its remaining tax attributes.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
The Company had federal and state carryforwards of $14.7 million and $452.3 million that will begin to expire in 2031 and 2028, respectively unless utilized. The remaining federal and state NOL carryforwards of $115.4 million and $1.6 million will carry forward indefinitely. At December 31, 2024, the Company had federal and state charitable contribution carryforwards of $166.4 million which will begin to expire in 2025. At December 31, 2024, the Company had $36.2 million of federal R&D credit carryforwards, of which $0.2 million will expire in 2025 unless utilized, and the remaining federal R&D credit carryforwards will begin to expire beginning in 2026. At December 31, 2024, the Company had state R&D credit carryforwards of approximately $2.4 million that will begin to expire in 2025 and $21.5 million that have no expiration date. At December 31, 2024, the Company had foreign NOL carryforwards of $145.3 million that will expire in 2025 unless utilized and $9.0 million that have no expiration date. The Company continues to record the deferred tax assets related to these attributes, subject to valuation allowance, until expiration occurs.
The components of the deferred tax assets are as follows (in thousands):
| | | | | | | | |
| | December 31, | |
| | 2024 | | | 2023 | |
Deferred tax assets | | | | | | |
NOL carryforwards | | $ | 117,052 | | | $ | 149,049 | |
R&D credit carryforwards | | | 27,543 | | | | 70,906 | |
Capitalized R&D | | | 110,848 | | | | 90,164 | |
Stock-based compensation | | | 51,438 | | | | 51,028 | |
Charitable contributions | | | 40,008 | | | | 40,956 | |
Lease liabilities | | | 12,753 | | | | 13,671 | |
Intangibles | | | 50,431 | | | | 43,220 | |
Accrued rebates | | | 35,186 | | | | 19,401 | |
Other | | | 21,130 | | | | 16,087 | |
Total deferred tax assets | | | 466,389 | | | | 494,482 | |
Valuation allowance | | | (454,966 | ) | | | (482,089 | ) |
Deferred tax liabilities | | | | | | |
Right-of-use assets | | | (11,423 | ) | | | (12,393 | ) |
Total deferred tax liabilities | | | (11,423 | ) | | | (12,393 | ) |
Total net deferred tax assets | | $ | — | | | $ | — | |
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. The Company assesses all available positive and negative evidence to estimate whether sufficient future taxable income will be generated to permit use of the existing deferred tax assets. A significant piece of objective negative evidence evaluated was the cumulative book loss incurred over the three-year period ended December 31, 2024. Such objective evidence limits the ability to consider other subjective evidence, such as projections for future growth. On the basis of this analysis, a valuation allowance of $455.0 million and $482.1 million at December 31, 2024 and 2023, respectively, was recorded to offset the net deferred tax asset as realization of such asset is uncertain. However, the amount of the deferred tax asset considered realizable could be adjusted if estimates of future taxable income during the carryforward period are increased, or if objective negative evidence in the form of cumulative losses is no longer present and additional weight is given to subjective evidence such as the Company’s projections for future growth.
An accounting policy may be selected to either (i) treat taxes due on future U.S. inclusions in taxable income related to global intangible low-taxed income (GILTI) as a current-period expense when incurred or (ii) factor such amounts into a company’s measurement of its deferred taxes. The Company has elected to account for GILTI as a period cost.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
A reconciliation of income taxes to the amount computed by applying the statutory federal income tax rate to the pretax income (loss) is summarized as follows (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Amounts computed at statutory federal rate | | $ | 54,196 | | | $ | (10,718 | ) | | $ | (44,823 | ) |
Stock-based compensation and other permanent differences | | | 9,986 | | | | 7,865 | | | | 7,459 | |
Branded pharmaceutical drug fee | | | 2,122 | | | | 1,848 | | | | 1,454 | |
Write-off of IP R&D | | | 1,260 | | | | — | | | | 2,449 | |
Other permanent differences | | | 1,008 | | | | 593 | | | | 137 | |
R&D credits | | | (18,406 | ) | | | (5,827 | ) | | | (9,974 | ) |
Change in valuation allowance | | | (27,013 | ) | | | 1,100 | | | | 11,227 | |
State taxes | | | 3,050 | | | | (977 | ) | | | (2,232 | ) |
Contingencies | | | 5,960 | | | | (2,071 | ) | | | 6,993 | |
Foreign rate differential | | | (13,715 | ) | | | (5,076 | ) | | | (1,971 | ) |
Deferred adjustments for limits on executive compensation | | | 2,375 | | | | 2,112 | | | | 3,918 | |
Deferred rate adjustment | | | (528 | ) | | | (438 | ) | | | 922 | |
Expiration of attributes | | | 3,264 | | | | 17,225 | | | | 16,142 | |
GILTI | | | 8,215 | | | | 7,665 | | | | 10,804 | |
Other | | | (150 | ) | | | (3,051 | ) | | | 26 | |
Income tax expense | | $ | 31,624 | | | $ | 10,250 | | | $ | 2,531 | |
The tax years 2003 – 2024 remain open to examination by the major taxing jurisdictions to which the Company is subject.
The Company recognizes a tax benefit from an uncertain tax position when it is more likely than not that the position will be sustained upon examination. The Company recorded an uncertain tax position reserve of $1.3 million, $18.0 million and $5.1 million for the years ended December 31, 2024, 2023 and 2022, respectively. Due to the valuation allowance recorded against the Company’s deferred tax assets, approximately $8.7 million and $6.8 million of the total unrecognized tax benefits as of December 31, 2024 and 2023, respectively, would reduce the annual effective tax rate if recognized. The Company does not anticipate that the amount of unrecognized tax benefits as of December 31, 2024 will significantly change within the next twelve months. The Company’s practice is to recognize interest and/or penalties related to uncertain income tax positions in income tax expense. The Company had immaterial interest and/or penalties accrued on the Company’s consolidated balance sheets at December 31, 2024 or 2023, respectively. Further, the Company recognized an insignificant amount of interest and/or penalties in the statement of operations for the years ended December 31, 2024, 2023 and 2022, respectively, related to uncertain tax positions.
The following table provides a reconciliation of changes in unrecognized tax benefits (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Balance at beginning of period | | $ | 37,112 | | | $ | 19,064 | | | $ | 13,923 | |
Additions related to current period tax positions | | | 6,337 | | | | 5,304 | | | | 5,140 | |
Additions related to prior period tax positions | | | — | | | | 12,956 | | | | 38 | |
Reductions related to prior period tax positions | | | (5,074 | ) | | | (212 | ) | | | (37 | ) |
Balance at end of period | | $ | 38,375 | | | $ | 37,112 | | | $ | 19,064 | |
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
9. Commitments and Contingencies
License and Merger Agreements
The Company has entered into various collaboration, licensing and merger agreements which provide the Company with rights to certain know-how, technology and patent rights. The agreements generally include upfront license fees, development and commercial milestone payments upon achievement of certain clinical and commercial development and annual net sales milestones, as well as royalties calculated as a percentage of product revenues, with rates that vary by agreement. The Company incurred $34.5 million, $102.5 million and $88.7 million in upfront and license payments in the years ended December 31, 2024, 2023 and 2022, respectively. These upfront and license payments were included in the research and development expenses in the consolidated statements of operations as there was no alternative future use associated with the payments. As of December 31, 2024, the Company may be required to make milestone payments up to $4.0 billion in the aggregate for candidates in its pipeline, of which, pursuant to the license agreement with Neuren, $50.0 million is payable to Neuren within 60 days following Neuren’s invoice delivered after December 31, 2024 as the Company’s aggregate net revenue of trofinetide in North America for the treatment of Rett syndrome exceeded $250.0 million in 2024.
In May 2018, the Company signed an Exclusivity Deed (the Deed) with Neuren that provided for exclusive negotiations for a period of three months from the date of the Deed. Under the terms of the Deed, the Company invested $3.1 million to subscribe for 1,330,000 shares of Neuren and paid $0.9 million for the exclusive right to negotiate a deal with Neuren, which was recorded in selling, general and administrative expenses in the consolidated statements of operations in the second quarter of 2018. In 2023, the Company sold the 1,330,000 shares of Neuren for total proceeds of $12.3 million. Net gain on the strategic investments recognized in other income in the consolidated statements of operations for the year ended December 31, 2023 and 2022 was $5.1 million and $3.5 million, respectively. No gain was recorded in the year ended December 31, 2024.
In August 2018, the Company entered into a license agreement with Neuren and obtained exclusive North American rights to develop and commercialize trofinetide for Rett syndrome and other indications. Under the terms of the agreement, the Company paid Neuren an upfront license fee of $10.0 million and it may be required to pay up to an additional $455.0 million in milestone payments based on the achievement of certain development and annual net sales milestones. In addition, the Company will be required to pay Neuren tiered, escalating, double-digit percentage royalties based on net sales. The license agreement was accounted for as an asset acquisition and the upfront cash payment of $10.0 million was expensed to research and development in the third quarter of 2018 as there is no alternative use for the asset. In connection with the FDA approval of DAYBUE, the Company paid a milestone payment of $40.0 million to Neuren following the first commercial sale of DAYBUE pursuant to the license agreement. The Company capitalized the $40.0 million milestone payment as an intangible asset as it was deemed probable of occurring as of March 31, 2023. In addition, the Company was granted a Rare Pediatric Disease PRV following the FDA approval of DAYBUE. Pursuant to the license agreement, the Company is required to pay Neuren one third of the value of the PRV at the time of sale or use of the PRV. The Company capitalized the $29.6 million for the estimated PRV value owed to Neuren as an intangible asset in 2023. During the year ended December 31, 2024, the Company sold the PRV to a third party for aggregate net proceeds of $146.5 million. Upon sale of the PRV, the Company capitalized an additional $19.2 million for the one third PRV value owed to Neuren as an intangible asset.
In July 2023, the Company expanded its licensing agreement for trofinetide with Neuren to acquire rights to the drug outside of North America as well as global rights in Rett syndrome and Fragile X syndrome to Neuren’s development candidate NNZ-2591. Under the terms of the expanded agreement, Neuren received an upfront payment of $100.0 million and is eligible to receive up to an additional $426.3 million in milestone payments based on the achievement of certain commercial and sales milestones for trofinetide outside of North America and up to $831.3 million in milestone payments based on the achievement of certain development and sales milestones for NNZ-2591. In addition, the Company will be required to pay Neuren tiered royalties from the mid-teens to low-twenties percent of trofinetide net sales outside of North America. Percentage royalties related to NNZ-2591 net sales are identical to the trofinetide in each of North America and outside North America. The expanded license agreement was accounted for as an asset acquisition and the upfront cash payment of $100.0 million was expensed to research and development in the third quarter of 2023 as there is no alternative use for the asset.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
In January 2022, the Company entered into a license and collaboration agreement with Stoke Therapeutics, Inc. (Stoke) to discover, develop and commercialize novel RNA-based medicines for the potential treatment of severe and rare genetic neurodevelopmental diseases of the CNS. The collaboration includes SYNGAP1 syndrome, Rett syndrome (MECP2), and an undisclosed neurodevelopmental target. For the SYNGAP1 program, the two companies will jointly share global research, development and commercialization responsibilities and share 50/50 in all worldwide costs and future profits. In addition, Stoke is eligible to receive potential development, regulatory, first commercial sales and sales milestones. For the MECP2 program and the undisclosed neurodevelopmental program, the Company acquired an exclusive worldwide license to develop and commercialize MECP2 program and the undisclosed neurodevelopmental program. Stoke will lead research and pre-clinical development activities, while the Company will lead clinical development and commercialization activities. The Company will fund research and pre-clinical development activities related to these two targets and Stoke is eligible to receive potential development, regulatory, first commercial sales and sales milestones as well as tiered royalty payments on worldwide sales starting in the mid-single digit range and escalating to the mid-teens based on revenue levels. Under the terms of the agreement, the Company paid Stoke a $60.0 million upfront payment which was accounted for as an asset acquisition and was expensed to research and development in the first quarter of 2022 as there is no alternative use for the asset. The Company may be required to pay up to an additional $907.5 million in milestones as well as royalties on future sales.
In November 2024, the Company entered into a license agreement with Saniona, for the development and commercialization of ACP-711, a highly selective GABAA-α3 positive allosteric modulator. The first indication the Company plans to pursue is development of ACP-711 for essential tremor, a neurological condition that includes shaking or trembling movements in one or more parts of the body. The Company will lead further clinical development, regulatory submissions, and global commercialization efforts for ACP-711 while also providing financial support for Saniona’s ongoing Phase 1 study and preparations for Phase 2. Under the terms of the license agreement, the Company paid Saniona an upfront fee of $28.0 million and it may be required to pay up to $582.0 million in milestone payments based on the achievement of certain development and annual net sales milestones. In addition, the Company will be required to pay Saniona tiered royalties of mid-single digits to low double digits on net sales of commercial products that may result from development of ACP-711. The license agreement was accounted for as an asset acquisition and the upfront cash payment of $28.0 million was expensed to research and development in the fourth quarter of 2024 as there is no alternative use for the asset. The potential milestone payments to Saniona consist of up to $147.0 million subject to achievement of development and commercial milestones related to potential first and second indications, and up to $435.0 million subject to achievement of thresholds of annual net sales of ACP-711 worldwide.
Corporate Credit Card Program
In connection with the Company’s credit card programs, the Company established letters of credit for a total of $5.0 million, which have automatic annual extensions and are fully secured by restricted cash.
Fleet Program
In connection with the Company’s fleet program, the Company established a letter of credit for $0.4 million, which has automatic annual extensions and is fully secured by restricted cash.
Legal Proceedings
Patent Infringement
On July 24, 2020, the Company filed complaints against (i) Aurobindo Pharma Limited and its affiliate Aurobindo Pharma USA, Inc. and (ii) Teva Pharmaceuticals USA, Inc. and its affiliate Teva Pharmaceutical Industries Ltd., and on July 30, 2020, the Company filed complaints against (i) Hetero Labs Limited and its affiliates Hetero Labs Limited Unit-V and Hetero USA Inc., (ii) MSN Laboratories Private Ltd. and its affiliate MSN Pharmaceuticals, Inc., and (iii) Zydus Pharmaceuticals (USA) Inc. and its affiliate Cadila Healthcare Limited. These complaints, which were filed in the United States District Court for the District of Delaware, allege infringement of certain of the Company’s Orange Book-listed patents covering NUPLAZID (Pimavanserin I Cases).
The Company entered into an agreement effective April 22, 2021 with Hetero settling all claims and counterclaims in the litigation. The agreement allows Hetero to launch its generic pimavanserin product on February 27, 2038, subject to certain triggers for earlier launch. The Hetero case was dismissed by joint agreement on May 3, 2021.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
On September 30, 2022, the Company filed a stipulation and proposed order to stay the claims currently asserted against Teva and for Teva to be bound by the result of the litigation rendered against the remaining defendants Aurobindo and MSN, which was ordered by the Court on October 4, 2022.
On October 21, 2022, the Company filed additional complaints against Aurobindo, MSN and Zydus in the United States District Court for the District of Delaware alleging infringement of an additional Orange Book-listed patent covering NUPLAZID (Pimavanserin II Cases).
The Company entered into an agreement, effective March 31, 2023, with Zydus settling all claims and counterclaims in the Pimavanserin I Cases and Pimavanserin II Cases. The agreement allows Zydus to launch its generic pimavanserin 10 mg tablet products on September 23, 2036 and 34 mg capsule products on February 27, 2038, subject to certain triggers for earlier launch. The Zydus case was dismissed by joint agreement on April 5, 2023.
As a result of the above, only MSN remained as an active defendant in the Pimavanserin I Cases. On January 11, 2024, following summary judgment motions, the District Court entered final judgment in the Company’s favor that MSN’s submission of ANDA No. 214925 was an act of infringement in the Pimavanserin I Case and the ’740 patent was not invalid. On January 18, 2024, MSN filed a Notice of Appeal to the United States Court of Appeals for the Federal Circuit from the final judgment entered on January 11, 2024. The appeal is fully briefed and awaiting a date for oral argument.
In connection with the Pimavanserin II cases, MSN and Aurobindo are the remaining defendants. A bench trial was conducted from December 3, 2024 to December 6, 2024 in the matter. Post-trial briefing was completed on February 12, 2025 and the Company is awaiting the final decision of the Court.
On February 14, 2025, the Company filed a complaint against Zydus Lifesciences Limited, Zydus Worldwide DMCC, and Zydus Pharmaceuticals (USA) Inc. (collectively “Zydus”) in the United States District Court for the District of Delaware, alleging infringement of certain of the Company’s Orange Book-listed patents covering NUPLAZID (Pimavanserin) by Zydus’ proposed 34 mg pimavanserin tablet product. Zydus has not responded to the complaint as of yet and there is no schedule in place for the case.
Securities Class Action
On April 19, 2021, a purported stockholder of the Company filed a putative securities class action complaint (captioned City of Birmingham Relief Retirement Systems v. Acadia Pharmaceuticals, Inc., Case No. 21-cv-0762) in the U.S. District Court for the Southern District of California against the Company and certain of the Company’s current executive officers. On September 29, 2021, the Court issued an order designating lead plaintiff and lead counsel. On December 10, 2021, lead plaintiff filed an amended complaint. The amended complaint generally alleges that defendants violated Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, by failing to disclose that the materials submitted in support of its sNDA seeking approval of pimavanserin for the treatment of hallucinations and delusions associated with dementia-related psychosis contained statistical and design deficiencies and that the FDA was unlikely to approve the sNDA in its current form. The amended complaint seeks unspecified monetary damages and other relief. On March 11, 2024, the Court granted plaintiffs’ motion for class certification and appointment of class representatives and class counsel. The parties are currently engaged in expert discovery, which is scheduled to close on June 13, 2025.
Opt-Out Litigation
On March 7, 2024, a purported stockholder of the Company filed a complaint (captioned Alger Dynamic Opportunities Fund v. Acadia Pharmaceuticals, Inc., Case No. 24-cv-00451) in the U.S. District Court for the Southern District of California against the Company and one executive officer. The complaint is based on the same underlying allegations as the Securities Class Action described above, and alleged claims under federal and state securities laws, and for common law fraud and negligent misrepresentations. On May 24, 2024, Defendants moved to dismiss the complaint. On October 31, 2024, the Court granted in part and denied in part Defendants’ motion to dismiss. The Court dismissed with leave to amend the purported stockholder’s state and common law claims, as well as the claim brought under Section 18(a) of the Securities Exchange Act of 1934, as amended. Defendants filed their answer to the Sections 10(b) and 20(a) claims on December 16, 2024. On January 13, 2025, the Court stayed this suit pending the outcome of the Securities Class Action.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Derivative Suit
On December 15, 2023, a purported stockholder of the Company filed a derivative action (captioned Kanner et al v. Biggar et al., Case No. 23-cv-2293) in the U.S. District Court for the Southern District of California against certain of the Company’s current directors. The Company is named as a nominal defendant. The complaint is based on the same alleged misconduct as the Securities Class Action, and asserts state law claims, on behalf of the Company, against the individual defendants for breach of fiduciary duty, unjust enrichment, abuse of control, waste of corporate assets, and insider trading. The complaint also asserts federal claims under sections 10(b), 21D, and 14(a) of the Securities Exchange Act of 1934, as amended. On December 27, 2023, the action was reassigned to District Judge William Q. Hayes and Magistrate Judge Michael S. Berg due to its relation to the Securities Class Action. On January 30, 2024, the parties jointly requested a stay of the action. The Court granted that request and the action was stayed on February 20, 2024, pending the outcome of our Demand Review Committee’s investigation into the underlying claims.
Given the unpredictability inherent in litigation, the Company cannot predict the outcome of these matters. The Company is unable to estimate possible losses or ranges of losses that may result from these matters, and therefore it has not accrued any amounts in connection with these matters other than attorneys’ fees incurred to date.
10. Leases
The Company leases facilities, vehicles and certain equipment under noncancelable operating leases with remaining lease terms of 0.3 year to 6.4 years, some of which include options to extend the lease for up to two five-year terms. These optional periods were not considered in the determination of the right-of-use asset or the lease liability as the Company did not consider it reasonably certain that it would exercise such options.
The operating lease costs were as follows (in thousands):
| | | | | | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
Operating lease cost | | $ | 11,836 | | | $ | 10,343 | | | $ | 8,095 | |
Operating sublease income | | | (1,824 | ) | | | (93 | ) | | | — | |
Net operating lease costs | | $ | 10,012 | | | $ | 10,250 | | | $ | 8,095 | |
Supplemental cash flow information related to the Company’s leases were as follows (in thousands):
| | | | | | | | |
| | Years Ended December 31, | |
| | 2024 | | | 2023 | |
Cash paid for amounts included in the measurement of lease liabilities: | | | | | | |
Operating cash flows from operating leases | | $ | 10,327 | | | $ | 9,456 | |
Right-of-use assets obtained in exchange for operating lease obligations: | | | 2,218 | | | | 2,051 | |
The balance sheet classification of the Company’s lease liabilities was as follows (in thousands):
| | | | | | | | |
| | December 31, 2024 | | | December 31, 2023 | |
Operating lease liabilities | | | | | | |
Current portion included in accrued liabilities | | $ | 9,958 | | | $ | 9,405 | |
Operating lease liabilities | | | 42,037 | | | | 47,800 | |
Total operating lease liabilities | | $ | 51,995 | | | $ | 57,205 | |
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
Maturities of lease liabilities were as follows (in thousands):
| | | | |
| | Operating Leases | |
Years ending December 31, | | | |
2025 | | $ | 10,224 | |
2026 | | | 9,170 | |
2027 | | | 9,248 | |
2028 | | | 9,414 | |
2029 | | | 9,119 | |
Thereafter | | | 12,245 | |
Total lease payments | | | 59,420 | |
Less: | | | |
Imputed interest | | | (7,425 | ) |
Total operating lease liabilities | | $ | 51,995 | |
Operating lease liabilities are based on the net present value of the remaining lease payments over the remaining lease term. In determining the present value of lease payments, the Company uses its incremental borrowing rate based on the information available at the lease commencement date. As of December 31, 2024 and 2023, the weighted average remaining lease term was 6.1 years and 7.0 years, respectively, and the weighted average discount rate used to determine the operating lease liability was 4.5% and 4.5%, respectively.
In the fourth quarter of 2018, the Company entered into an agreement to lease the 4th and 5th floors of corporate office space in San Diego, California with total minimum lease payments of $50.4 million over an initial term of 10 years and 9 months. In February 2020, the Company entered into the first amendment to the lease agreement to lease the 2nd floor of corporate office space in San Diego, California with total minimum lease payments of $25.3 million over an initial term of approximately 10 years and 7 months. In March 2020, the Company entered into the second amendment to the lease agreement which increased the total minimum lease payments of the original corporate office space to $51.4 million. In the third quarter of 2020, the lease for the 4th and 5th floors of corporate office space commenced and the Company capitalized a right of use asset and related lease liability of $40.3 million. In the first quarter of 2021, the lease for the 2nd floor of corporate office space commenced and the Company capitalized a right of use asset and related lease liability of $19.2 million. In connection with this lease and the amendment, the Company established a letter of credit for $3.1 million, which has automatic annual extensions and is fully secured by restricted cash.
In May 2023, the Company entered into an agreement to sublease its 2nd floor of corporate office space in San Diego to a sublessee with a total minimum sublease income of $18.4 million over a term of approximately 7 years and 6 months. The Company delivered the full possession of its 2nd floor of corporate office space to the sublessee in August 2023. Pursuant to the sublease agreement, the Company received the first sublease payment in December 2023.
11. Segment Reporting
In accordance with FASB ASC Topic 280, Segment Reporting, management has determined that the Company operates in one business segment which is the development and commercialization of innovative medicines. All revenues for the years ended December 31, 2024, 2023 and 2022 were generated from customers in the United States.
The Company’s CODM is the CEO who uses the consolidated statement of operations to make decisions about allocating resources and assessing performance for the entire Company. Managing and allocating resources on a consolidated basis enables the CODM to assess the overall level of resources available and how to best deploy these resources across functions and research and development programs that are in line with the Company’s long-term company-wide strategic goals.
The key areas of focus by CODM for allocation of resources are revenues from each product, as well as operating expenses (cost of goods sold, license fees and royalties, research and development expense, selling, general and administrative expense, and other income or loss). While the CODM analyzes these categories, the area of focus is period over period fluxes and budget-to-actual variances to determine the right allocation of resources is attributed to the segment in order to ensure profitability is maximized.
ACADIA PHARMACEUTICALS INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
The following table illustrates reported segment revenue, segment profit and significant segment expenses (in thousands):
| | | | | | | | | | | | |
| | Year Ended December 31, | |
| | 2024 | | | 2023 | | | 2022 | |
NUPLAZID net revenue | | $ | 609,385 | | | $ | 549,248 | | | $ | 517,235 | |
DAYBUE net revenue | | | 348,412 | | | | 177,189 | | | | — | |
Total revenues | | | 957,797 | | | | 726,437 | | | | 517,235 | |
Less: | | | | | | | | | |
Cost of goods sold | | | 30,068 | | | | 19,826 | | | | 10,166 | |
License fees and royalties | | | 51,773 | | | | 21,812 | | | | — | |
Research and development expense: | | | | | | | | | |
External research and development | | | 194,539 | | | | 170,036 | | | | 189,832 | |
Internal costs(1) | | | 74,210 | | | | 79,083 | | | | 83,002 | |
Upfront and milestone payments | | | 34,500 | | | | 102,500 | | | | 88,741 | |
Total research and development expense | | | 303,249 | | | | 351,619 | | | | 361,575 | |
Selling, general and administrative | | | 488,428 | | | | 406,559 | | | | 369,090 | |
Gain on sale of non-financial asset | | | (146,515 | ) | | | — | | | | — | |
Interest income, net | | | (25,458 | ) | | | (17,234 | ) | | | (6,610 | ) |
Other income | | | (1,823 | ) | | | (5,109 | ) | | | (3,542 | ) |
Income tax expense | | | 31,624 | | | | 10,250 | | | | 2,531 | |
Consolidated net income | | $ | 226,451 | | | $ | (61,286 | ) | | $ | (215,975 | ) |
(1)Includes personnel expenses and costs allocated to multiple research and development programs, including benefits, information technology, facilities and inventory.
SCHEDULE II – Valuation and Qualifying Accounts
(in thousands)
| | | | | | | | | | | | | | | | | | | | |
| | | | | Additions | | | Deductions | | | | |
| | Balance at
Beginning of
Period | | | Provision
Related to
Current
Period Sales | | | Actual
Distribution
Fees,
Discounts and
Chargebacks
Related to
Current Period
Sales | | | Actual
Distribution
Fees,
Discounts and
Chargebacks
Related to
Prior Period
Sales | | | Balance at
End of Period | |
Allowance for distribution fees, discounts and chargebacks: | | | | | | | | | | | | | | | |
For the year ended December 31, 2022 | | $ | 8,467 | | | $ | 80,836 | | | $ | (69,913 | ) | | $ | (8,467 | ) | | $ | 10,923 | |
For the year ended December 31, 2023 | | $ | 10,923 | | | $ | 97,797 | | | $ | (85,641 | ) | | $ | (10,923 | ) | | $ | 12,156 | |
For the year ended December 31, 2024 | | $ | 12,156 | | | $ | 122,083 | | | $ | (110,200 | ) | | $ | (12,156 | ) | | $ | 11,883 | |

