| Delaware | 8731 | 94-3306718 | ||
(State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
Large accelerated filer ¨ | Accelerated filer ¨ | Non-accelerated filer ¨ (Do not check if a smaller reporting company) | Smaller reporting company x |
Title of Each Class of Securities to be Registered | Amount to be Registered | Proposed Maximum Offering Price Per Share (1) | Proposed Maximum Aggregate Offering Price (2) | Amount of Registration Fee | ||||||||||||
| Common Stock, $0.001 par value per share, issuable pursuant to the Purchase Agreement | (1)7,000,000 | $ | 0.39 | $ | 2,730,000 | $ | 312.86 | |||||||||
| TOTAL (1) | 7,000,000 | $ | 0.39 | $ | 2,730,000 | $ | 312.86 | |||||||||
| (1) | We are registering shares of our common stock (the “Put Shares”) that we may put to Four M Purchasers, LLC (“Four M Purchasers” or “Selling Stockholder”) pursuant to a Purchase Agreement (the “Purchase Agreement”) between the Selling Stockholder and the Registrant, entered into as of November 14, 2011. In the event of stock splits, stock dividends, or similar transactions involving the common stock, the number of common shares registered shall, unless otherwise expressly provided, automatically be deemed to cover the additional securities to be offered or issued pursuant to Rule 416 promulgated under the Securities Act of 1933, as amended (the “Securities Act”). In the event that adjustment provisions of the Purchase Agreement require the Registrant to issue more shares than are being registered in this registration statement, for reasons other than those stated in Rule 416 of the Securities Act, the Registrant will file a new registration statement to register those additional shares. |
| (2) | Estimated solely for purposes of calculating the registration fee under Rule 457 under the Securities Act, using the last closing price as reported on the Over-the-Counter Bulletin Board (the “OTCBB”) on January 9, 2012, which was $0.39 per share |
| Page | ||
| Part I - Information Required in Prospectus | ||
| Forward-Looking Statements | 1 | |
| Prospectus Summary | 2 | |
| Risk Factors | 6 | |
| Risk Factors Relating to this Offering | 6 | |
| Risk Factors Relating to Operation of Northwest Biotherapeutics, Inc. | 9 | |
| Risk Factors Relating to the Securities Markets and Investments in Our Common Stock | 15 | |
| Use of Proceeds | 18 | |
| Determination of Offering Price | 19 | |
| Dilution | 19 | |
| The Selling Stockholder | 19 | |
| The Purchase Agreement | 20 | |
| The Offering | 20 | |
| Plan of Distribution | 22 | |
| Description of Securities to be Registered | 25 | |
| Description of Business | 26 | |
| Interest of Named Experts and Counsel | 43 | |
| Description of Property | 43 | |
| Legal Proceedings | 44 | |
| Market for Common Equity and Related Matters | 44 | |
| Management’s Discussion and Analysis of Financial Condition and Results of Operations | 46 | |
| Changes in and Disagreements with Accountants on Accounting and Financial Disclosures | 52 | |
| Directors, Executive Officers, Promoters and Control Persons | 52 | |
| Executive Compensation | 55 | |
| Security Ownership of Certain Beneficial Owners and Management and Related Shareholder Matters | 62 | |
| Certain Relationships and Party Related Party Transactions | 65 | |
| Part II - Information Not Required in Prospectus | ||
| Other Expenses of Issuance and Distribution | 68 | |
| Indemnification of Directors, Officers, Employees, and Agents | 69 | |
| Recent Sales of Unregistered Securities | 69 | |
| Where You Can Find More Information | 75 | |
| Exhibits | 76 | |
| Undertakings | 83 | |
| Signatures | 84 | |
| Financial Statements | F-1 |
FORWARD-LOOKING STATEMENTS This Prospectus contains forward-looking statements. Such forward-looking statements include statements regarding, among other things (a) our business development plans, (b) anticipated trends in our industry, (c) our future financing plans and (d) our anticipated needs for working capital. Forward-looking statements, which involve assumptions and describe our future plans, strategies, and expectations, are generally identifiable by use of the words “may”, “will”, “should”, “expect”, “anticipate”, “estimate”, “believe”, “intend” or “project” or the negative of these words or other variations on these words or comparable terminology. This information may involve known and unknown risks, uncertainties, and other factors that may cause our actual results, performance, or achievements to be materially different from the future results, performance, or achievements expressed or implied by any forward-looking statements or otherwise. These statements may be found under “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business” as well as in this Prospectus generally. Actual events or results may differ materially from those discussed in forward-looking statements as a result of various factors, including, without limitation, the risks outlined under “Risk Factors” and matters described in this Prospectus generally. In light of these risks and uncertainties, there can be no assurance that the forward-looking statements contained in this filing will in fact occur. In addition to the information expressly required to be included in this filing, we will provide such further material information, if any, as may be necessary to make the required statements, in light of the circumstances under which they are made, not misleading. |
| Common Stock Being Offered By Selling Stockholder | 7,000,000 shares of Common Stock issuable to Four M Purchasers, LLC pursuant to the Purchase Agreement with us dated November 14, 2011 (the “Purchase Agreement”). | |
| Offering Price | The offering price for shares of our Common Stock will be determined by prevailing prices established on the OTCBB, or as negotiated in private transactions, or as otherwise described in the “Plan of Distribution.” | |
| Terms of the Offering | The Selling Stockholder will determine when and how it will sell the Put Shares of Common Stock offered in this Prospectus. | |
| Termination of the Offering | The offering will conclude upon the earlier of (i) such time as all of the Common Stock contemplated in this Registration Statement has been sold, or (ii) six (6) months after the effective date of this Registration Statement. | |
| Use of Proceeds | We are not selling any of the Put Shares of Common Stock in this offering and, as a result, will not receive any proceeds directly from this offering. We will, however, receive proceeds from our sale of those Put Shares to the Selling Stockholder pursuant to the Purchase Agreement. Such proceeds will be used for working capital, reduction of indebtedness, acquisitions and general corporate purposes. | |
| OTCBB Trading Symbol | “NWBO.OB” | |
| Risk Factors | The Common Stock offered hereby involves a high degree of risk and should not be purchased by investors who cannot afford the loss of their entire investment. See “Risk Factors” beginning on page [6]. |
| Three months ended | Nine months ended | Period from March 18, 1996 (Inception) to | ||||||||||||||||||
| September 30 | September 30 | September 30, | ||||||||||||||||||
| 2010 | 2011 | 2010 | 2011 | 2011 | ||||||||||||||||
| Revenues: | ||||||||||||||||||||
| Research material sales | $ | 10 | $ | 10 | $ | 10 | $ | 10 | $ | 580 | ||||||||||
| Contract research and development from related parties | - | - | - | - | 1,128 | |||||||||||||||
| Research grants and other | - | - | - | - | 1,061 | |||||||||||||||
| Total revenues | 10 | 10 | 10 | 10 | 2,769 | |||||||||||||||
| Operating cost and expenses: | ||||||||||||||||||||
| Cost of research material sales | - | - | - | - | 382 | |||||||||||||||
| Research and development | 1,606 | 3,565 | 4,791 | 11,474 | 88,286 | |||||||||||||||
| General and administration | 1,412 | 2,804 | 4,680 | 10,675 | 72,664 | |||||||||||||||
| Depreciation and amortization | 2 | 2 | 2 | 6 | 2,359 | |||||||||||||||
| Loss on facility sublease | - | - | - | - | 895 | |||||||||||||||
| Asset impairment loss and other (gain) loss | - | - | - | - | 2,445 | |||||||||||||||
| Total operating costs and expenses | 3,020 | 6,371 | 9,473 | 22,155 | 167,031 | |||||||||||||||
| Loss from operations | (3,010 | ) | (6,361 | ) | (9,463 | ) | (22,145 | ) | (164,262 | ) | ||||||||||
| Other income (expense): | ||||||||||||||||||||
| Valuation of reclassified equity contracts | - | 8,875 | - | 7,413 | 14,172 | |||||||||||||||
| Loan conversion inducement | - | (4,522 | ) | (125 | ) | (10,415 | ) | |||||||||||||
| Derivative valuation gain (loss) | - | 338 | - | 29 | 83 | |||||||||||||||
| Gain on sale of intellectual property and property and equipment | - | - | - | - | 3,664 | |||||||||||||||
| Interest expense | (2,371 | ) | (2,370 | ) | (5,968 | ) | (5,426 | ) | (39,342 | ) | ||||||||||
| Interest income and other | - | - | - | - | 1,707 | |||||||||||||||
| Net income (loss) | (5,381 | ) | 482 | (19,953 | ) | (20,254 | ) | (194,393 | ) | |||||||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | - | - | - | - | (12,349 | ) | ||||||||||||||
| Modification of Series A preferred stock warrants | - | - | - | - | (2,306 | ) | ||||||||||||||
| Modification of Series A-1 preferred stock warrants | - | - | - | - | (16,393 | ) | ||||||||||||||
| Series A preferred stock dividends | - | - | - | - | (334 | ) | ||||||||||||||
| Series A-1 preferred stock dividends | - | - | - | - | (917 | ) | ||||||||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | - | - | - | - | (4,664 | ) | ||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | - | - | - | - | (1,872 | ) | ||||||||||||||
| Series A preferred stock redemption fee | - | - | - | - | (1,700 | ) | ||||||||||||||
| Beneficial conversion feature of Series D preferred stock | - | - | - | - | (4,274 | ) | ||||||||||||||
| Net income (loss) applicable to common stockholders | $ | (5,381 | ) | $ | 482 | $ | (19,953 | ) | $ | (20,254 | ) | $ | (239,202 | ) | ||||||
| Net income (loss) per share applicable to common stockholders — basic | $ | (0.08 | ) | $ | 0.01 | $ | (0.31 | ) | $ | (0.24 | ) | |||||||||
| Weighted average shares used in computing basic income (loss) per share | 70,413 | 95,123 | 65,361 | 85,680 | ||||||||||||||||
| Net income (loss) per share applicable to common stockholders - diluted | $ | (0.08 | ) | $ | 0.00 | $ | (0.31 | ) | $ | (0.24 | ) | |||||||||
| Weighted average shares used in computing diluted net income (loss) per share | 70,413 | 123,136 | 65,361 | 85,680 | ||||||||||||||||
| As of December 31, | As of September 30, | |||||||||||
| 2009 | 2010 | 2011 | ||||||||||
| Balance Sheet Data: | ||||||||||||
| Cash | $ | 65 | $ | 153 | $ | 1,643 | ||||||
| Working capital (deficit) | (19,329 | ) | (25,885 | ) | (44,332 | ) | ||||||
| Total Assets | 103 | 294 | 1,893 | |||||||||
| Long term obligations, net of current portion and discounts | 1,359 | 1,854 | 1,898 | |||||||||
| Total stockholders' equity (deficit) | (20,686 | ) | ( 27,684 | ) | (46,150 | ) | ||||||
| • | limited dissemination of the market price of our securities; |
| • | limited news coverage; |
| • | limited interest by investors in our securities; |
| • | volatility of our stock price due to low trading volume; |
| • | increased difficulty in selling our securities in certain states due to “blue sky” restrictions; and |
| • | limited ability to issue additional securities or to secure additional financing. |
| • | make a special written suitability determination for the purchaser; |
| • | receive the purchaser’s written agreement to a transaction prior to sale; |
| • | provide the purchaser with risk disclosure documents which identify certain risks associated with investing in “penny stocks” and which describe the market for these “penny stocks” as well as a purchaser’s legal remedies; and |
| • | obtain a signed and dated acknowledgment from the purchaser demonstrating that the purchaser has actually received the required risk disclosure document before a transaction in a “penny stock” can be completed. |
| • | lack of a sufficient number of independent directors on our audit committee |
| • | lack of a financial expert on our audit committee |
| • | insufficient segregation of duties in our finance and accounting function due to limited personnel |
| Percent of Offering sold | 100% | 75% | 50% | 10% | ||||||||||||
| Net Tangible Book Value Per Share Prior to Sale | $ | (46,150 | ) | (46,150 | ) | (46,150 | ) | (46,150 | ) | |||||||
| Pro Forma Net Tangible Book Value Per Share After Sale | (44,120 | ) | (44,645 | ) | (45,170 | ) | (46,010 | ) | ||||||||
| Increase in net book value per share due to stock sale | $ | 2,030 | 1,505 | 980 | 140 | |||||||||||
| Net Dilution (Purchase price of $[0.42] less Pro Forma Net Tangible Book Value per share) | 4.40 | % | 3.26 | % | 2.12 | % | 0.30 | % | ||||||||
| Name of selling security holder | Amount of securities of the class owned by the security holder before this offering | Amount to be offered for the security holder’s account | Amount and (if one percent or more) percentage of the class to be owned by security holder after the offering is complete | |||||||||
| Four M Purchasers, LLC (1) | 0 | 7,000,000 | 0 | % | ||||||||
| (1) | Dennis Mehiel, the Sole and Managing Member of Four M Purchasers, LLC (“Four M”), holds voting and/or investment power over Four M. |
| · | we will not be entitled to put shares to the Selling Stockholder unless there is an effective registration statement under the Securities Act covering the re-sale of all such Put Shares by the Selling Stockholder; |
| · | we will not be entitled to put shares to the Selling Stockholder unless our Common Stock continues to be quoted on the OTC Bulletin Board, or becomes listed on a national securities exchange; and |
| · | we will not be entitled to put shares to the Selling Stockholder prior to the closing date of the preceding put. |
| · | the uncertainty about when and in what amounts we will choose to sell shares of our Common Stock to the Selling Shareholder, and what the price of our stock will be at the time of those sales; |
| · | the uncertainty about when and in what amounts and at what prices the Selling Stockholder will re-sell those shares of our Common Stock.. |
| · | ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| · | block trades in which the broker-dealer will attempt to sell the shares as agent but may position and re-sell a portion of the block as principal to facilitate the transaction; |
| · | purchases by a broker-dealer as principal and re-sales by the broker-dealer for its account; |
| · | an exchange distribution in accordance with the rules of the applicable exchange; |
| · | privately negotiated transactions; |
| · | to cover short sales made after the date that the registration statement of which this prospectus is a part is declared effective by the Securities and Exchange Commission; |
| · | broker-dealers may agree with the Selling Stockholder to sell a specified number of such shares at a stipulated price per share; |
| · | a combination of any of these methods of sale; and |
| · | any other method permitted pursuant to applicable law. |

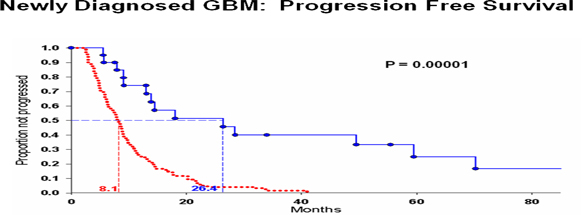
| · | For the delay in time to recurrence, from 6.9 months with standard of care to 25 months in patients treated with DCVax, the “p value” was .00001 (i.e., a 1 in 100,000 chance that these results were random events….. a level of statistical significance far stronger than the 5 in 100 chance, or less, of random events that FDA requires for product approval). |
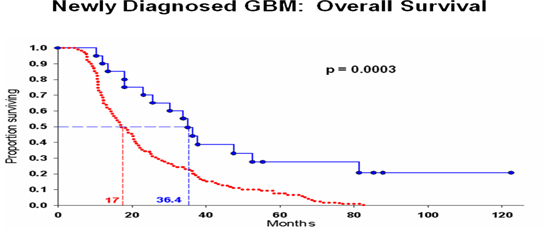
| · | For the extension of survival time, from 14.6 months with standard of care to 36.4 months in patients treated with DCVax, the “p value” was .0003 (i.e., a 3 in 10,000 chance that these results were random events…. which is also a level of statistical significance far stronger than the 5 in 100 chance, or less, of random events that FDA requires for product approval). |
Group A: Hormone Independent Prostate Cancer Patients Without Metastases | ||||
| Natural Course of Disease* | With DCVax-Prostate | |||
Median time to disease progression (appearance of bone metastases) | 28-34 weeks | 59 weeks | ||
Median survival | 36 months | >54 months and continuing* *(more than half of these patients still alive at this long-term follow-up point) | ||
Group B: Hormone Independent Prostate Cancer Patients With Metastases | ||||||
With Standard of Care (Taxotere) | With Dendreon’s Provenge | With DCVax-Prostate | ||||
| Median survival | 18.9 months | 25.9 months | 38.7 months | |||
| Overall survival at 3 years | 11% | 33% | 64% | |||
| High | Low | |||||||
| Year end December 31, 2009 | ||||||||
| First Quarter | $ | 1.79 | $ | 0.65 | ||||
| Second Quarter | 1.00 | 0.55 | ||||||
| Third Quarter | 1.69 | 0.45 | ||||||
| Fourth Quarter | 0.85 | 0.32 | ||||||
| Year end December 31, 2010 | ||||||||
| First Quarter | $ | 0.85 | $ | 0.65 | ||||
| Second Quarter | 1.35 | 0.67 | ||||||
| Third Quarter | 1.60 | 0.70 | ||||||
| Fourth Quarter | 0.95 | 0.72 | ||||||
| Year end December 31, 2011 | ||||||||
| First Quarter | $ | 0.77 | $ | 0.34 | ||||
| Second Quarter | 0.89 | 0.38 | ||||||
| Third quarter | 0.70 | 0.38 | ||||||
| Plan category | Number of securities to be issued upon Exercise of outstanding options, warrants and Rights (a) | Weighted-average exercise price of Outstanding options, warrants and rights (b) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) | |||||||||
| Equity compensation plans approved by security holders | 32,763 | $ | 0.66 | 7,942 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 32,763, | $ | 0.66 | 7,942 | ||||||||
| · | Level 1, defined as observable inputs such as quoted prices for identical instruments in active markets; |
| · | Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable such as quoted prices for similar instruments in active markets or quoted prices for identical or similar instruments in markets that are not active; and |
| · | Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions, such as valuations derived from valuation techniques in which one or more significant inputs or significant value drivers are unobservable. |
| September 30, | September 30, | |||||||||||
| 2010 | 2011 | Change | ||||||||||
| Net cash provided by (used in); | ||||||||||||
| Operating activities | $ | (6,232 | ) | $ | (10,152 | ) | $ | (3,920 | ) | |||
| Investing activities | (41 | ) | (31 | ) | 10 | |||||||
| Financing activities | 6,627 | 11,717 | 5,090 | |||||||||
| Effect of exchange rates on cash | (69 | ) | (44 | ) | 25 | |||||||
| Increase in cash | $ | 285 | $ | 1,490 | $ | 1,205 | ||||||
| 2010 | 2011 | |||||||
| Issuance of notes payable | $ | 2,667 | $ | 6,772 | ||||
| Issuance of common stock and warrants | 3,960 | 5,344 | ||||||
| Repayments of notes payable | - | (399 | ) | |||||
| $ | 6,627 | $ | 11,717 | |||||
| Name | Age | Position | ||
| Linda F. Powers | 55 | Director, Chairperson, Chief Executive Officer | ||
| Alton L. Boynton, Ph.D. | 66 | Director, Chief Scientific Officer | ||
| Anthony Maida, Ph.D. | 59 | Chief Operating Officer | ||
| Leslie Goldman | 66 | Senior Vice President, Business Development | ||
| Marnix Bosch, Ph.D. | 52 | Chief Technical Officer | ||
| Robert A. Farmer | 72 | Director |
EXECUTIVE COMPENSATION
Summary Compensation Table
Compensation Discussion and Analysis
Our Process
Typically, our executive compensation is comprehensively assessed and analyzed annually; however, given our limited funding since 2002, our executives have received infrequent increases in their compensation. During 2011, our executives also received equity based incentives.
Normally, the review process includes, but is not limited to, the following steps:
| · | The Compensation Committee reviews the performance of the Chief Executive Officer and other senior executives; |
| · | The current annual compensation of senior management and long-term compensation grants made over the past few years are reviewed; |
| · | The appropriate performance metrics and attributes of annual and long-term programs for the next year are considered and discussed; |
| · | The entirety of our compensation program is considered; |
| · | For our top officers, if peer group compensation is available for their position, we use a blend of survey and peer compensation for comparison, as we compete not only in our own market, but nationally and across industries, for talent; |
| · | The compensation practices of our peer companies are reviewed, including their practices with respect to equity and other grants, benefits and perquisites; |
| · | The compensation of our management team from the standpoint of internal equity, complexity of the job, scope of responsibility and other factors is assessed; and |
| · | Management’s stock ownership is reviewed. |
Management has the following involvement with the executive compensation process:
| · | The Chief Executive Officer recommends salaries, annual and long-term incentive targets, and plan amendments and design before recommendations are submitted to the Compensation Committee for approval; and |
| · | The Chief Executive Officer is involved in establishing and recommending to the Compensation Committee financial goals for the incentive programs based on management’s operational goals and strategic plans. |
Compensation Goals
Our philosophy regarding executive compensation is to attract and retain highly qualified people by paying competitive salaries, and to link the financial interests of our senior management to those of our stockholders by tying compensation to the achievement of operational and financial objectives. Our compensation package for our officers includes both short-term and long-term features in the forms of base salary and equity-based incentives in the form of stock options, which are granted periodically at the discretion of the Compensation Committee.
Elements of Executive Compensation
Base Salaries
Base salaries for all executive officers are reviewed annually. The Compensation Committee reviews the compensation of the President and Chief Executive Officer. The President and Chief Executive Officer, together with the compensation committee, review the other executive officer’s compensation. The Compensation Committee also consults with the President and Chief Executive Officer with respect to the compensation package for all other executive officers. In evaluating salaries, each officer’s individual performance during the prior year, as well as salary levels in the biotechnology industry for comparable positions are considered. In determining how the respective officer contributes to the Company, current corporate performance, as well as the potential for future performance gains, is considered. No specific weight is attributed to the foregoing for purposes of determining base salaries.
Equity-Based Incentives
We provide our executive officers with long-term incentives through Stock Option Plans, (as defined under “— Equity Plans” below), all described in more detail below. The primary objective of this plan is to provide an incentive for employees, including our executive officers, to make decisions and take actions that maximize long-term stockholder value. The plans are designed to promote this long-term focus by using discretionary grants and long-term vesting periods. Subject to the terms of the plans, the Compensation Committee determines the terms and conditions of options granted under the plans, including the exercise price, which is based on fair value of our stock on the date of grant. For various motivation and retention considerations, option awards granted subsequent to our initial public offering in December 2001 generally vest over four years. The Compensation Committee believes that stock options provide an incentive for employees, allowing us to attract and retain high quality management and staff. Stock options were issued to five (5) executives in 2011.
Employee and Executive Benefits
Our executives participate in many of the same employee benefit programs as our other employees. The core employee benefit programs include a tax-qualified retirement plan, medical coverage, dental coverage, life insurance, disability coverage, and vacation. The tax qualified retirement plan is a 401(k) plan. We made matching contributions to each employee’s 401(k) plan account of $0.50 for each dollar contributed on the first $3,000 of compensation contributed to the plan. Our matching contribution policy was terminated effective March 2006. All of these matching contribution amounts to our Named Executive Officers are shown in theAll OtherCompensation footnote to the Summary Compensation Table following this section.
Perquisites
Historically, we have offered only a very limited number of perquisites to our executives as an incremental benefit to recognize their position within the Company. No perquisites of any kind were offered to executives in 2011.
Compensation of the President and Chief Executive Officer
In assembling the compensation package for our President and Chief Executive Officer, the Compensation Committee considers our annual and long-term performance, the performance of the President and Chief Executive Officer, and our cash resources and needs. Although the Committee’s overall goal is to set the President and Chief Executive Officer’s salary at the median level for competitors that are similar in industry size and performance, the actual level approved by the Committee may be higher or lower based upon the Committee’s subjective evaluation of the foregoing. Consistent with the foregoing, the Compensation Committee set the annual base salary for the President and Chief Executive Officer, Dr. Alton Boynton, at $331,250 for the fiscal year ended 2011. On June 21, 2011, Dr. Alton Boynton was offered and accepted the position of Chief Science Officer, thereby stepping down from his role as President and Chief Executive Officer. Ms. Linda Powers then assumed the role of President and Chief Executive at an annual base salary of $360,000.
Summary Compensation Table
The following table sets forth certain information concerning compensation paid or accrued to our named executive officers (the “Named Executive Officers”) during the years ended December 31, 2011, 2010, and 2009. The Option Awards shown in the table below do not constitute cash or value actually received by the named executive officer. Instead, the amounts shown are the non-cash aggregate fair values of Option Awards that were granted during the periods presented but were then subject to vesting requirements. The majority of the Options were not vested and will not vest unless certain milestones are met in the future, or certain employment period requirements are met in the future. With respect to the portion of the Options that did vest, in the case Ms. Powers and Mr. Goldman, upon vesting all of those Options became subject to an extended lock-up (until the earlier of 18 months or the Company reaching the primary endpoint of its GBM brain cancer clinical trial).
| Name and Principal Position | Year | Salary | Bonus | Option Awards | All Other Compensation | Total | ||||||||||||||||||
| Linda M Powers (1) President, Chairperson & Chief Executive Officer | 2011 | $ | 203,308 | $ | — | $ | 9,262,133 | $ | — | $ | 9,465,441 | |||||||||||||
| 2010 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| 2009 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Alton L. Boynton, Ph.D. (2) Chief Scientific Officer | 2011 | $ | 334,732 | $ | — | $ | 1,547,963 | $ | — | $ | 1,882,695 | |||||||||||||
| President, Chief Executive | 2010 | $ | 359,528 | $ | — | $ | — | $ | — | $ | 359,528 | |||||||||||||
| Officer, Chief Scientific Officer and Secretary | 2009 | $ | 538,281 | $ | — | $ | — | $ | — | $ | 538,281 | |||||||||||||
| Anthony Maida, Ph.D. (3) | 2011 | $ | 160,384 | $ | — | $ | 814,182 | $ | — | $ | 974,566 | |||||||||||||
| Chief Financial Officer | 2010 | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| 2009 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Leslie Goldman (4) | 2011 | $ | 149,092 | $ | — | $ | 1,465,527 | $ | — | $ | 1,614,619 | |||||||||||||
| Senior Vice President, | 2010 | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| Business Development | 2009 | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| Marnix L. Bosch, Ph.D., M.B.A. (5) | 2011 | $ | 339,362 | $ | 20,000 | $ | 1,192,963 | $ | — | $ | 1,552,325 | |||||||||||||
| Chief Technical Officer | 2010 | $ | 431,652 | $ | — | $ | — | $ | — | $ | 431,652 | |||||||||||||
| 2009 | $ | 283,750 | $ | — | $ | — | $ | — | $ | 283,750 | ||||||||||||||
(1) In conjunction with the employment agreement entered into between the Company and Ms. Powers on June 8, 2011, and in recognition of Ms. Powers’ service to the Company while serving as Chairman during the preceding four years, the Company granted Ms. Powers an option to purchase 14,220,000 shares of the Company's stock with an exercise price of $0.66 per share. One-third of the options vested on the grant date, and upon vesting became subject to a lock-up which extends to the earlier of 18 months or the Company reaching the primary endpoint of its GBM brain cancer clinical trial. One-third of the options will vest in equal monthly portions over the term of the employment agreement. The remaining one-third will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved. Additionally, Ms. Powers received $100,000 during 2010 as non-executive compensation for her services as the Chairman of the Company’s Board of Directors.
(2) In conjunction with the employment agreement entered into between the Company and Dr. Boynton on June 8, 2011, the Company issued Dr. Boynton an option to purchase 2,376,562 shares of the Company's stock with an exercise price of $0.66 per share. 1,376,562 options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remaining 880,000 options will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(3) In conjunction with the employment agreement entered into between the Company and Dr. Maida on June 8, 2011, the Company issued Dr. Maida an option to purchase 1,250,000 shares of the Company's stock with an exercise price of $0.66 per share. No options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remainder will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(4) In conjunction with the employment agreement entered into between the Company and Mr. Goldman on June 8, 2011, the Company issued Mr. Goldman an option to purchase 2,250,000 shares of the Company's stock with an exercise price of $0.66 per share. One-third of the options vested on the grant date,and upon vesting became subject to alock-up which extendsto theearlier of 18 months or the Company reachingthe primary endpoint of its GBMbraincancer clinical trial. One-third will vest in equal monthly portions over the term of the employment agreement. The remaining one-third will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(5) In conjunction with the employment agreement entered into between the Company and Dr. Bosch on June 8, 2011, the Company issued Dr. Bosch an option to purchase 1,831,536 shares of the Company's stock with an exercise price of $0.66 per share. 831,536 options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remaining 880,000 options will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
Outstanding Equity Awards at Fiscal Year-End
The following table shows outstanding stock option awards classified as exercisable and un-exercisable as of December 31, 2011.
| Number of Securities Underlying Unexercised Options | ||||||||||||||||
| Name and Principal Position | Exercisable | Un-exercisable | Option Exercise Price ($) | Option Expiration Date | ||||||||||||
| Linda Powers President & Chief Executive | -(1 | ) | 14,220,000 | $ | 0.66 | 6/21/2018 | ||||||||||
| Alton Boynton Chief Scientific Officer | 1,406,562 (2) | 970,000 | $ | 0.66 | 6/21/2018 | |||||||||||
| 5,286 (6) | — | $ | 18.75 | 4/18/2014 | ||||||||||||
| 2,016 (6) | — | $ | 1.35 | 2/18/2016 | ||||||||||||
| 1,430,846 (7) | — | $ | 0.55 | 8/20/2022 | ||||||||||||
| Anthony Maida Chief Financial Officer | 30,000 (3) | 1,220,000 | $ | 0.66 | 6/21/2018 | |||||||||||
| Leslie Goldman Senior Vice President | -(4 | ) | 2,250,000 | $ | 0.66 | 6/21/2018 | ||||||||||
| Marnix Bosch Chief Technical Officer | 861,536 (5) | 970,000 | $ | 0.66 | 6/21/2018 | |||||||||||
| 333 (8) | — | $ | 18.75 | 9/20/2014 | ||||||||||||
| 833 (8) | — | $ | 75.00 | 1/10/2015 | ||||||||||||
| 3,194 (8) | 139 | $ | 1.35 | 2/18/2016 | ||||||||||||
| 4,000 (8) | 1,333 | $ | 1.80 | 12/1/2016 | ||||||||||||
| 308,338 (9) | 541,662 | $ | 0.70 | 6/23/2022 | ||||||||||||
| 250,000 (10) | — | $ | 0.55 | 8/20/2022 | ||||||||||||
(1) In conjunction with the employment agreement entered into between the Company and Ms. Powers on June 8, 2011, and in recognition of Ms. Powers’ service to the Company while serving as Chair during the preceding four years, the Company granted Ms. Powers an option to purchase 14,220,000 shares of the Company's stock with an exercise price of $0.66 per share. One-third of the options vested on the grant date, and upon vesting became subject to a lock-up which extends to the earlier of 18 months or the Company reaching the primary endpoint of its GBM brain cancer clinical trial. One-third of the options will vest in equal monthly portions over the term of the employment agreement. The remaining one-third will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(2) In conjunction with the employment agreement entered into between the Company and Dr. Boynton on June 8, 2011, the Company issued Dr. Boynton an option to purchase 2,376,562 shares of the Company's stock with an exercise price of $0.66 per share. 1,376,562 options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remaining 880,000 options will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(3) In conjunction with the employment agreement entered into between the Company and Dr. Maida on June 8, 2011, the Company issued Dr. Maida an option to purchase 1,250,000 shares of the Company's stock with an exercise price of $0.66 per share. No options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remainder will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(4) In conjunction with the employment agreement entered into between the Company and Mr. Goldman on June 8, 2011, the Company issued Mr. Goldman an option to purchase 2,250,000 shares of the Company's stock with an exercise price of $0.66 per share. One-third of the options vested on the grant date, and upon vesting became subject to a lock-up which extends to the earlier of 18 months or the Company reaching the primary endpoint of its GBM brain cancer clinical trial. One-third will vest in equal monthly portions over the term of the employment agreement. The remaining one-third will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(5) In conjunction with the employment agreement entered into between the Company and Dr. Bosch on June 8, 2011, the Company issued Dr. Bosch an option to purchase 1,831,536 shares of the Company's stock with an exercise price of $0.66 per share. 831,536 options vested on the grant date. 120,000 options will vest in equal monthly portions over the term of the employment agreement. The remaining 880,000 options will vest in portions tied to material milestones in multiple Company programs, if and to the extent those milestones are achieved.
(6) These options were granted under the 1999 Plan, the 2001 Plan and under Dr. Boynton’s previous employment agreement. Each of these option grants vests over a four year period. One-fourth of each option grant vests on the first anniversary of the grant date and the remaining three-fourths of each grant vests in equal monthly installments over the remaining three year vesting period.
| (7) This option was granted under the 2007 Stock Option Plan. This option grant vests over a three and one-half year period. Approximately 29% the option grant was vested immediately upon grant with respect to prior service performed. Approximately 17% vests on the first anniversary of the AIM offering (June 22, 2008) and the remaining portion vests in equal monthly installments over the remaining three year vesting period. These options were granted in recognition of past service to the Company and have an exercise price of $0.60 per share, which is equal to the conversion price of warrants issued to Toucan Partners under the Conversion Agreement. In accordance with Dr. Boynton’s option agreement as options to 1,430,846, 500,568 and 500,568 shares had not been exercised as of December 31, 2008, 2009 and 2010 respectively such options were forfeited. |
| (8) This option was granted under the 2007 Stock Option Plan. This option grant vested over the balance of 2009 with 1,132,464 vesting on the grant date and the remainder vesting in equal installments on August 31, September 30, October 31, November 30 and December 31, 2009. |
| (9) These options were granted under the 1999 Plan and the 2001 Plan. Each of these option grants vests over a four year period. One-fourth of each option grant vests on the first anniversary of the grant date and the remaining three-fourths of each grant vests in equal monthly installments over the remaining three year vesting period. | |
| (10) This option was granted under the 2007 Stock Option Plan. This option grant vested over the balance of 2009 with 125,000 vesting on the grant date and the remainder vesting on December 31, 2009. | |
Director Compensation
The following table sets forth certain information concerning compensation paid or accrued to our non-executive directors during the year ended December 31, 2011.
| Name | Year | Fees Earned or Paid in Cash | All Other Compensation(1) | Total | ||||||||||||
| Robert A. Farmer | 2011 | $ | 25,000 | $ | — | $ | 25,000 | |||||||||
(1) Robert Farmer was also issued 33,313 shares of the Company’s stock at $0.67 per share for his services to the Board of Directors.
Only non-employee directors receive director fees. Effective June 22, 2007, we are required to pay Linda F. Powers, as Chairperson and a non-executive member of the Board of Directors, approximately $100,000 per annum for her services. These payments effectively ended subsequent to Ms. Powers assuming the position of Chief Executive Officer of the Company. Also effective December 10, 2009 the Company was required to issue Robert A. Farmer $50,000 per annum for his services as a non-executive member of the Board of Directors. During 2011, Mr. Farmer elected to take 50% of his 2011 directors’ fee in the Company’s common stock.
Compensation Committee Interlocks and Insider Participation
None of our executive officers served during 2011 as a director of any other entity, one of whose executive officers served as a director on our Board or as a member of our Compensation Committee.
Equity Plans
Stock Option Plans
The Company’s stock option plans are administered by the Board of Directors, which determines the terms and conditions of the options granted, including exercise price, number of options granted and vesting period of such options.
The 2007 Stock Option Plan became effective on June 15, 2007. In April 2008, the Company increased the number of shares reserved for issuance by 519,132 shares for an aggregate of 6,000,000 shares of its common stock. In May 2010, the Company increased the number of shares reserved for issuance under the 2007 Stock Option Plan by an additional 10,000,000 shares of its common stock. In May 2011, the Company increased the number of shares reserved for issuance under the 2007 Stock Option Plan by an additional 20,000,000 shares of its common stock.
Our employees, directors and consultants previously participated in the 1998 Stock Option Plan and the 1999 Executive Stock Option Plan. The 1998 Stock Option Plan and the 1999 Executive Stock Option Plan were terminated during 2008 and 2009 and no further grants may be made under the plans.
Existing stock option plans are as follows:
(a) 2001 Stock Option Plan
Under the 2001 Stock Option Plan (the “2001 Plan”), 120,000 shares of the Company’s common stock have been reserved for grant of stock options to employees and consultants. Additionally, on January 1 of each year, commencing January 1, 2002, the number of shares reserved for grant under the 2001 Plan will increase by the lesser of (i) 15% of the aggregate number of shares available for grant under the 2001 Plan or (ii) 20,000 shares. Our Board of Directors has the authority to amend or terminate this plan, but such action may not adversely affect any outstanding option previously granted under the plan. If this plan is not terminated earlier, no incentive stock options can be granted under the plan on or after the later of June 2011 or the 10th anniversary of the date when our Board of Directors adopted, subject to approval by our stockholders, the most recent increase in the number of shares available for grant under the plan.
(b) 2001 Non-employee Director Stock Incentive Plan
Under the 2001 Non-employee Director Stock Incentive Plan (the “2001 Director Plan”), 13,333 shares of the Company’s common stock have been reserved for grant of stock options to non-employee directors of the Company. As of December 31, 2009, net of forfeitures, a total of 10,500 shares remain available under this plan; however, no further grants may be made under this plan.
Employee Stock Purchase Plan
Our Employee Stock Purchase Plan (the “Employees’ Plan”) was adopted by our Board of Directors and approved by our stockholders in June 2001.
This plan is administered by the Compensation Committee of our Board of Directors and provides a mechanism for eligible employees to purchase shares of our common stock. To facilitate these purchases, eligible participants are assigned plan accounts, to which they may contribute funds via payroll deduction. The purchases are accomplished through the use of six-month offering periods. Purchases pursuant to this plan are made at a price equal to the lower of (i) 85% of the fair market value of our common stock on the last trading day in the offering period; or (ii) 85% of the fair market value of our common stock on the last trading day before the commencement of such offering period. No participant may purchase more than 67 shares of our common stock during any offering period. Additionally, purchases under the plan are limited such that no participant may purchase under the plan, in any offering period that commenced in that calendar year, shares with a fair market value in excess of $25,000 minus the fair market value of any shares that the participant previously purchased in that calendar year. In the case of shares purchased during an offering period that commenced in the preceding calendar year, the limitation is $50,000 minus the fair market value of any shares that the participant purchased during the calendar year of the purchase and the calendar year immediately preceding such purchase.
Our Board of Directors has the authority to amend or terminate this plan at any time. Amendments to the plan are subject to approval by our stockholders to the extent required by applicable law.
| · | each person, or group of affiliated persons, who is known by us to own beneficially 5% or more of any class of our equity securities; |
| · | our directors; |
| · | each of our named executive officers, as defined in Item 402(a)(3) of Regulation S-K; and |
| · | our directors and executive officers as a group. |
| Name of Beneficial Owner | Number of Shares Beneficially Owned | Percentage(1) | ||||||
| Officers and Directors | ||||||||
| Alton L. Boynton, Ph.D.(2) | 3,074,719 | 2 | % | |||||
| Marnix L. Bosch, Ph.D., M.B.A.(3) | 1,820,056 | 1.2 | % | |||||
| Linda F. Powers(4) | 112,174,363 | 59.1 | % | |||||
| Robert A. Farmer (5) | 1,300,968 | 0.9 | % | |||||
| Les Goldman (6) | 3,220,823 | 2.1 | % | |||||
| All executive officers and directors as a group (5 persons)(7) | 121,590,929 | 61.6 | % | |||||
| 5% Security Holders | ||||||||
| Toucan Capital Fund II, L.P.(8) | 27,162,363 | 16.6 | % | |||||
| 7600 Wisconsin Avenue, Suite 700, Bethesda, MD 20814 | ||||||||
| Toucan Partners, LLC(9) | 28,609,290 | 16.8 | % | |||||
| 7600 Wisconsin Avenue, Suite 700, Bethesda, MD 20814 | ||||||||
| Regen Med Acquisition Corp (10) | 11,144,165 | 7.1 | % | |||||
1313 N. Market Street, Suite 5100 Wilmington, DE 19801 | ||||||||
| Al Rajhi Holdings (11) | 6,353,872 | 4.3 | % | |||||
| Rue Maurice 3, 1204 Geneve, Switzerland | ||||||||
Al Salam Opportunities Limited 4th Floor Harbour Center P.O. Box 613 George Town Grand Cayman, British West Indies, KY1-1001 | 2,590,270 | 1.7 | % | |||||
| The Richard M. Schulze Family Trust (12) | 8,902,175 | 6 | % | |||||
| 8500 Normandale Lake Blvd, Suite 1750, | ||||||||
| Minneapolis, MN 55347 | ||||||||
| Cognate BioServices, Inc.(13) | 48,500,000 | 32.5 | % | |||||
| 4800 East Shelby Drive, Suite 108, Memphis, TN | ||||||||
| (1) | Percentage represents beneficial ownership percentage of common stock calculated in accordance with SEC rules and does not equate to voting percentages. |
| (2) | Includes 2,833,715 shares of common stock issuable upon exercise of options that are exercisable within 60. |
| (3) | Includes 1,486,039 shares of common stock issuable upon exercise of options that are exercisable within 60 days. |
| (4) | Includes (i) 12,866,324 shares of common stock held by Toucan Capital; (ii) 14,296,039 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days held by Toucan Capital; (iii) 7,946,447shares of common stock held by Toucan Partners; (iv) 12,516,924 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days of September 6, 2011; (v 8,145,919 shares of common stock currently issuable upon conversion of convertible loans held by Toucan Partners; (vi) 5,530,000 shares of common stock issuable upon exercise of options that are exercisable within 60 days; and (vii) 48,500,000 shares of common stock issued upon conversion of amounts due to Cognate. Ms. Powers is a managing member of Toucan Management, LLC, which is the manager of Toucan Capital; is a managing member of Toucan Partners and controls a majority of the stock of Cognate BioServices, Inc. |
| (5) | Includes 328,397 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days of September 6, 2011 and 140,000 shares issuable on conversion of a loan held by Mr. Farmer convertible within 60 days. |
| (6) | Includes 1,824,262 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days; 875,000 shares of common stock issuable upon the exercise of options exercisable within 60 days of September 6, 2011 and 224,000 shares of common stock issuable upon conversion of loans held by Mr. Goldman convertible within 60 days. |
| (7) | Includes 10,724,754 shares issuable to officers and directors upon exercise of options that are exercisable within 60 days; 28,965,622 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days; and 8,509,919 shares of common stock issuable upon conversion of convertible notes and amounts due to Toucan Partners. |
| (8) | Includes 14,296,039 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days held by Toucan Capital. |
| (9) | Includes 12,516,924 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days and 8,145,919 shares of common stock currently issuable upon conversion of convertible loans held by Toucan Partners. |
| (10) | Includes 7,416,703 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days held by Regen Med. |
| (11) | Includes 1,743,411 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days held by Al Rajhi. |
| (12) | Includes 2,307,972 shares of common stock currently issuable upon exercise of warrants that are exercisable within 60 days held by the Richard M. Schulze Family Trust. |
| (13) | Includes 48,500,000 shares of common stock issued upon conversion of amounts due to Cognate. |
| • | an aggregate of 12,866,324 shares of Common Stock; |
| • | warrants to purchase 9,433,821 shares of Common Stock at an exercise price of $0.60 per share (net of 4,716,911 transferred to Regen Med); |
| • | warrants to purchase 5,256,238 shares of Common Stock at an exercise price of $0.60 per share (net of 2,628,119 transferred to Regen Med); |
| • | an aggregate of 9,750,691 shares of Common Stock; |
| • | warrants to purchase 18,266,362 shares of Common Stock at an exercise price of $0.60 per share; |
| • | warrants to purchase 513,841 shares of common stock at an exercise price of $0.41 per share; |
| • | warrants to purchase 132,500 shares of common stock at an exercise price of $0.40 per share; |
| • | Warrants to purchase 2,195,667 shares of common stock at an exercise price of $0.75 per share |
| • | warrants to purchase 842,375 shares of common stock at an exercise price of $0.20 per share; |
| • | Warrants to purchase 5,256,238 shares of common stock at an exercise price of $0.15 per share |
| SEC Registration Fee | $ | 304.84 | ||
| Legal Fees and Expenses | 40,000.00 | |||
| Accounting Fees and Expenses | 20,000.00 | |||
| Miscellaneous | 10,000.00 | |||
| TOTAL | $ | 70,304.84 |
| (1) | A convertible note in the amount of $370,000. The note matures on December 20, 2011 and carries a one-time interest charge payable at maturity of $37,000. The note is convertible into shares of the Company’s stock at a conversion price equal to 70 percent of the average daily closing bid price for the ten trading day period preceding the date of the conversion notice which was $0.47 on the date the note was issued. |
| (2) | A convertible note in the amount of $130,000. The note matures on June 2, 2011 and carries zero interest. The note is convertible into shares of the Company’s stock at a conversion price equal to 80 percent of the average of the five lowest closing prices in the 25 days previous to the conversion date which was $0.54 on the date the note was issued. Any amount that is still outstanding at maturity is repayable in cash. |
| (3) | A convertible note in the amount of $500,000. The note matures on June 2, 2011 and carries zero interest. The note is convertible into shares of the Company’s stock at a conversion price equal to the lesser of (i) the average of the closing prices for the ten trading days preceding the issue date of this note which was $0.67, or (ii) seventy percent of the volume weighted average price for the fifteen trading days prior to a conversion date. The conversion price is adjustable for down rounds as long as any amount due is outstanding under the note, subject to substantial exceptions. |
| December 31, 2010 | September 30, 2011 | |||||||
| (Unaudited) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash | $ | 153 | $ | 1,643 | ||||
| Prepaid expenses and other current assets | 86 | 170 | ||||||
| Total current assets | 239 | 1,813 | ||||||
| Property and equipment: | ||||||||
| Laboratory equipment | 29 | 29 | ||||||
| Office furniture and other equipment | 123 | 154 | ||||||
| 152 | 183 | |||||||
| Less accumulated depreciation and amortization | (113 | ) | (119 | ) | ||||
| Property and equipment, net | 39 | 64 | ||||||
| Deposit and other non-current assets | 16 | 16 | ||||||
| Total assets | $ | 294 | $ | 1,893 | ||||
| Liabilities And Stockholders’ Equity (Deficit) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 2,835 | $ | 2,284 | ||||
| Accounts payable, related party | 10,527 | 9,879 | ||||||
| Accrued expenses | 2,074 | 2,982 | ||||||
| Accrued expenses, related party | 1,749 | 2,265 | ||||||
| Notes payable | 1,364 | 1,384 | ||||||
| Note payable to related parties | 4,000 | 4,000 | ||||||
| Convertible notes payable, net | 2,736 | 3,671 | ||||||
| Convertible notes payable to related party, net | - | 1,300 | ||||||
| Embedded derivative liability | 839 | 1,818 | ||||||
| Liability for reclassified equity contracts | - | 16,562 | ||||||
| Total current liabilities | 26,124 | 46,145 | ||||||
| Long term liabilities: | ||||||||
| Notes payable, net | 350 | 200 | ||||||
| Convertible notes payable, net | 555 | 1,513 | ||||||
| Convertible notes payable to related party, net | 949 | 185 | ||||||
| Total long term liabilities | 1,854 | 1,898 | ||||||
| Total liabilities | 27,978 | 48,043 | ||||||
| Stockholders’ equity (deficit): | ||||||||
| Preferred stock, $0.001 par value; 20,000,000 shares authorized and none issued and outstanding | ||||||||
| Common stock, $0.001 par value; 150,000,000 shares authorized, 73,118,471 and 97,112,036 shares issued and outstanding at December 31, 2010 and September 30, 2011, respectively | 73 | 97 | ||||||
| Additional paid-in capital | 191,344 | 193,152 | ||||||
| Deficit accumulated during the development stage | (218,948 | ) | (239,202 | ) | ||||
| Cumulative translation adjustment | (153 | ) | (197 | ) | ||||
| Total stockholders’ equity (deficit) | (27,684 | ) | (46,150 | ) | ||||
| Total liabilities and stockholders’ equity (deficit) | $ | 294 | $ | 1,893 | ||||
| Three months ended | Nine months ended | Period from March 18, 1996 (Inception) | ||||||||||||||||||
| September 30 | September 30 | to September 30, | ||||||||||||||||||
| 2010 | 2011 | 2010 | 2011 | 2011 | ||||||||||||||||
| Revenues: | ||||||||||||||||||||
| Research material sales | $ | 10 | $ | 10 | $ | 10 | $ | 10 | $ | 580 | ||||||||||
| Contract research and development from related parties | - | - | �� | - | - | 1,128 | ||||||||||||||
| Research grants and other | - | - | - | - | 1,061 | |||||||||||||||
| Total revenues | 10 | 10 | 10 | 10 | 2,769 | |||||||||||||||
| Operating cost and expenses: | ||||||||||||||||||||
| Cost of research material sales | - | - | - | - | 382 | |||||||||||||||
| Research and development | 1,606 | 3,565 | 4,791 | 11,474 | 88,286 | |||||||||||||||
| General and administration | 1,412 | 2,804 | 4,680 | 10,675 | 72,664 | |||||||||||||||
| Depreciation and amortization | 2 | 2 | 2 | 6 | 2,359 | |||||||||||||||
| Loss on facility sublease | - | - | - | - | 895 | |||||||||||||||
| Asset impairment loss and other (gain) loss | - | - | - | - | 2,445 | |||||||||||||||
| Total operating costs and expenses | 3,020 | 6,371 | 9,473 | 22,155 | 167,031 | |||||||||||||||
| Loss from operations | (3,010 | ) | (6,361 | ) | (9,463 | ) | (22,145 | ) | (164,262 | ) | ||||||||||
| Other income (expense): | ||||||||||||||||||||
| Valuation of reclassified equity contracts | - | 8,875 | - | 7,413 | 14,172 | |||||||||||||||
| Loan conversion inducement | - | (4,522 | ) | (125 | ) | (10,415 | ) | |||||||||||||
| Derivative valuation gain (loss) | - | 338 | - | 29 | 83 | |||||||||||||||
| Gain on sale of intellectual property and property and equipment | - | - | - | - | 3,664 | |||||||||||||||
| Interest expense | (2,371 | ) | (2,370 | ) | (5,968 | ) | (5,426 | ) | (39,342 | ) | ||||||||||
| Interest income and other | - | - | - | - | 1,707 | |||||||||||||||
| Net income (loss) | (5,381 | ) | 482 | (19,953 | ) | (20,254 | ) | (194,393 | ) | |||||||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | - | - | - | - | (12,349 | ) | ||||||||||||||
| Modification of Series A preferred stock warrants | - | - | - | - | (2,306 | ) | ||||||||||||||
| Modification of Series A-1 preferred stock warrants | - | - | - | - | (16,393 | ) | ||||||||||||||
| Series A preferred stock dividends | - | - | - | - | (334 | ) | ||||||||||||||
| Series A-1 preferred stock dividends | - | - | - | - | (917 | ) | ||||||||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | - | - | - | - | (4,664 | ) | ||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | - | - | - | - | (1,872 | ) | ||||||||||||||
| Series A preferred stock redemption fee | - | - | - | - | (1,700 | ) | ||||||||||||||
| Beneficial conversion feature of Series D preferred stock | - | - | - | - | (4,274 | ) | ||||||||||||||
| Net income (loss) applicable to common stockholders | $ | (5,381 | ) | $ | 482 | $ | (19,953 | ) | $ | (20,254 | ) | $ | (239,202 | ) | ||||||
| Net income (loss) per share applicable to common stockholders — basic | $ | (0.08 | ) | $ | 0.01 | $ | (0.31 | ) | $ | (0.24 | ) | |||||||||
| Weighted average shares used in computing basic income (loss) per share | 70,413 | 95,123 | 65,361 | 85,680 | ||||||||||||||||
| Net income (loss) per share applicable to common stockholders - diluted | $ | (0.08 | ) | $ | 0.00 | $ | (0.31 | ) | $ | (0.24 | ) | |||||||||
| Weighted average shares used in computing diluted net income (loss) per share | 70,413 | 123,136 | 65,361 | 85,680 | ||||||||||||||||
| Nine Months Ended | Period from March 18, 1996 (Inception) to | |||||||||||
| September 30, | September 30, | |||||||||||
| 2010 | 2011 | 2011 | ||||||||||
| Cash Flows from Operating Activities: | ||||||||||||
| Net Loss | $ | (19,953 | ) | $ | (20,254 | ) | $ | (194,393 | ) | |||
| Reconciliation of net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 2 | 6 | 2,359 | |||||||||
| Amortization of deferred financing costs | - | - | 320 | |||||||||
| Amortization debt discount | 2,853 | 4,058 | 28,267 | |||||||||
| Derivative valuation (gain) loss | - | (29 | ) | (83 | ) | |||||||
| Accrued interest converted to stock | 1,047 | - | 260 | |||||||||
| Accreted interest on convertible promissory note | - | - | 1,484 | |||||||||
| Stock-based compensation costs | 1,512 | 7,354 | 18,768 | |||||||||
| Stock and warrants issued for services and other expenses | 3,593 | 3,470 | 12,805 | |||||||||
| Loan conversion inducement | 4,522 | 125 | 10,415 | |||||||||
| Valuation of reclassified equity contracts | - | (7,413 | ) | (14,172 | ) | |||||||
| Asset impairment loss and loss (gain) on sale of properties | - | - | (936 | ) | ||||||||
| Loss on facility sublease | - | - | 895 | |||||||||
| Increase (decrease) in cash resulting from changes in assets and liabilities: | ||||||||||||
| Prepaid expenses and other current assets | (129 | ) | (84 | ) | 540 | |||||||
| Accounts payable and accrued expenses | 380 | 1,097 | 6,887 | |||||||||
| Related party accounts payable and accrued expenses | (59 | ) | 1,518 | 13,794 | ||||||||
| Accrued loss on sublease | - | - | (265 | ) | ||||||||
| Deferred rent | - | - | 410 | |||||||||
| Net Cash used in Operating Activities | (6,232 | ) | (10,152 | ) | (112,645 | ) | ||||||
| Cash Flows from Investing Activities: | ||||||||||||
| Purchase of property and equipment, net | (41 | ) | (31 | ) | (5,075 | ) | ||||||
| Proceeds from sale of property and equipment | - | - | 258 | |||||||||
| Proceeds from sale of intellectual property | - | - | 1,816 | |||||||||
| Proceeds from sale of marketable securities | - | - | 2,000 | |||||||||
| Refund of security deposit | - | - | (3 | ) | ||||||||
| Transfer of restricted cash | - | - | (1,035 | ) | ||||||||
| Net Cash used in Investing Activities | (41 | ) | (31 | ) | (2,039 | ) | ||||||
| Cash Flows from Financing Activities: | ||||||||||||
| Proceeds from issuance of note payable | - | 6,272 | 12,122 | |||||||||
| Proceeds from issuance of convertible notes payable to related parties | 2,667 | 500 | 1,800 | |||||||||
| Proceeds from issuance of note payable to related parties | - | - | 11,250 | |||||||||
| Repayment of note payable to related party | - | - | (7,600 | ) | ||||||||
| Proceeds from issuance of convertible promissory note and warrants, net of issuance costs | - | - | 16,091 | |||||||||
| Repayment of convertible promissory note | - | (399 | ) | (1,069 | ) | |||||||
| Borrowing under line of credit, Northwest Hospital | - | - | 2,834 | |||||||||
| Repayment of line of credit, Northwest Hospital | - | - | (2,834 | ) | ||||||||
| Payment on capital lease obligations | - | - | (323 | ) | ||||||||
| Payments on note payable | - | - | (420 | ) | ||||||||
| Proceeds from issuance preferred stock, net | - | - | 28,708 | |||||||||
| Proceeds from exercise of stock options and warrants | - | - | 228 | |||||||||
| Proceeds from issuance common stock, net | 3,960 | 5,340 | 58,914 | |||||||||
| Proceeds from sale of stock warrant | - | 4 | 94 | |||||||||
| Payment of preferred stock dividends | - | - | (1,251 | ) | ||||||||
| Series A preferred stock redemption fee | - | - | (1,700 | ) | ||||||||
| Deferred financing costs | - | - | (320 | ) | ||||||||
| Net Cash provided by Financing Activities | 6,627 | 11,717 | 116,524 | |||||||||
| Effect of exchange rates on cash | (69 | ) | (44 | ) | (276 | ) | ||||||
| Net increase in cash | 285 | 1,490 | 1,564 | |||||||||
| Cash at beginning of period | 65 | 153 | - | |||||||||
| Cash at end of period | $ | 350 | $ | 1,643 | $ | 1,564 | ||||||
| Supplemental disclosure of cash flow information — Cash paid during the period for interest | $ | - | $ | - | $ | 1,879 | ||||||
| Supplemental schedule of non-cash financing activities: | ||||||||||||
| Equipment acquired through capital leases | $ | - | $ | - | $ | 285 | ||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | - | - | 12,349 | |||||||||
| Issuance of common stock in connection with conversion of notes payable and accrued interest | 1,004 | 2,790 | 6,076 | |||||||||
| Modification of Series A preferred stock warrants | - | - | 2,306 | |||||||||
| Modification of Series A-1 preferred stock warrants | - | - | 16,393 | |||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | - | - | 4,664 | |||||||||
| Common stock warrant liability | - | - | 11,841 | |||||||||
| Accretion of mandatorily redeemable Series A preferred stock redemption obligation | - | - | 1,872 | |||||||||
| Debt discount on promissory notes | 2,490 | 4,891 | 18,982 | |||||||||
| Conversion of convertible promissory notes and accrued interest to Series D preferred stock | - | - | 5,324 | |||||||||
| Conversion of convertible promissory notes and accrued interest to Series A-1 preferred stock | - | - | 7,707 | |||||||||
| Conversion of convertible promissory notes and accrued interest to common stock | - | - | 269 | |||||||||
| Issuance of Series C preferred stock warrants in connection with lease agreement | - | - | 43 | |||||||||
| Issuance of common stock to settle accounts payable | - | - | 4 | |||||||||
| Liability for and issuance of common stock and warrants to Medarex | - | - | 840 | |||||||||
| Issuance of common stock to landlord | - | - | 35 | |||||||||
| Deferred compensation on issuance of stock options and restricted stock grants | - | - | 759 | |||||||||
| Cancellation of options and restricted stock | - | - | 849 | |||||||||
| Financing of prepaid insurance through note payable | - | - | 491 | |||||||||
| Stock subscription receivable | - | - | 480 | |||||||||
| · | Liability for reclassified equity contracts - The Company accounts for potential shares that can be converted to common stock that were in excess of authorized shares, as a liability that is recorded at fair value. |
| · | A single, continuous statement must present the components of net income and total net income, the components of other comprehensive income and total other comprehensive income, and a total for comprehensive income. |
| · | In a two-statement approach, an entity must present the components of net income and total net income in the first statement. That statement must be immediately followed by a financial statement that presents the components of other comprehensive income, a total for other comprehensive income, and a total for comprehensive income. |
Three Months Ended September 30, 2011 | Nine Months Ended September 30, 2011 | |||||||
| Risk-free interest rate | 2.27 | % | 2.27 | % | ||||
| Expected dividend yield | - | % | - | % | ||||
| Volatility | 193.6 | % | 193.6 | % | ||||
| Expected life | 7 Years | 7 Years | ||||||
| Weighted average Black - Scholes value of options granted | $ | 0.65 | $ | 0.65 | ||||
| Options | Weighted Average Exercise price | Weighted Average remaining Contractual Term | |||||||
| Outstanding at December 31, 2010 | 3,256 | $ | 0.71 | ||||||
| Grants | 22,128 | 0.66 | |||||||
| Expired | (5 | ) | 10.62 | ||||||
| Cancellations | (557 | ) | 0.60 | ||||||
| Outstanding at September 30, 2011 | 24,822 | $ | 0.67 | 9.8 years | |||||
| Exercisable at September 30, 2011 | 10,574 | $ | 0.66 | 9.6 years | |||||
| Available for grant at September 30, 2011 | 7,942 | ||||||||
Three months ended September 30, | Nine months ended September 30, | |||||||||||||||
| 2010 | 2011 | 2010 | 2011 | |||||||||||||
| Research and development | $ | 163 | $ | 226 | $ | 525 | $ | 2,172 | ||||||||
| General and administrative | 329 | 1,178 | 987 | 5,182 | ||||||||||||
| Total Stock-based compensation | $ | 492 | $ | 1,404 | $ | 1,512 | $ | 7,354 | ||||||||
Liability for reclassified equity contracts | ||||
| Balance , January 1, 2011 | $ | - | ||
| Liabilities reclassified at inception | 23,975,000 | |||
| Change in value of liabilities reclassified | (7,413,000 | ) | ||
| Balance, September 30, 2011 | $ | 16,562,000 | ||
Embedded Derivative Liability | ||||
| Balance , January 1, 2011 | $ | 839,000 | ||
| Reclassification to stockholders' equity | (381,000 | ) | ||
| Embedded derivative liability recognized | 1,389,000 | |||
| Net change in fair value of embedded derivative liabilities | (29,000 | ) | ||
| Balance, September 30, 2011 | $ | 1,818,000 | ||
December 31, 2010 | September 30, 2011 | |||||||
| Notes payable - current | ||||||||
| 12% unsecured due July 2011 (net of warrant discount and original issue discount $38 in 2010 and $0 in 2011) | $ | 714 | $ | 934 | ||||
| 12% unsecured originally due March 2011 | 650 | 450 | ||||||
| $ | 1,364 | $ | 1,384 | |||||
| Notes payable related parties - current | ||||||||
| 12% unsecured due December 31, 2011 | $ | 4,000 | $ | 4,000 | ||||
| Convertible notes payable, net - current | ||||||||
| 0% unsecured due June 2011 (net of discount related to beneficial conversion feature $357 in 2010 and $0 in 2011) | $ | 360 | $ | - | ||||
| 6% unsecured due November 2010 | 300 | 50 | ||||||
| 6% unsecured originally due March 2011 | 110 | 110 | ||||||
| 6% unsecured due between March 2011 and March 2012 (net of discount related to beneficial conversion feature $424 in 2010 and $139 in 2011) | 1,526 | 2,509 | ||||||
| 10% unsecured due between March and May 2011 (net of discount related to beneficial conversion feature $57 in 2010 and $0 in 2011) | 338 | - | ||||||
| 11% unsecured due December 2011 (net of discount related to beneficial conversion feature $143 in 2010 and $38 in 2011) | 102 | 49 | ||||||
| 6% unsecured due June 2012 (net of discount related to beneficial conversion feature $0 in 2010 and $182 in 2011) | - | 953 | ||||||
| $ | 2,736 | $ | 3,671 | |||||
| Convertible Notes payable related party, net - current | ||||||||
| 6% due July 2011 and November 2011 (net of discount reated to beneficial conversion feature $351 in 2010 and $0 in 2011) | $ | - | $ | 1,300 | ||||
| Long term notes payable | ||||||||
| 20% unsecured convertible note due December 2013 | $ | 350 | $ | - | ||||
| 6% unsecured note due October 2012 | - | 200 | ||||||
| $ | 350 | $ | 200 | |||||
| Long term convertible notes, net | ||||||||
| 6% unsecured due March 2012 (net of discount related to beneficial conversion feature $321 in 2010 and $0 in 2011) | $ | 555 | $ | - | ||||
| 20% unsecured convertible note due December 2013 (net of discount related to beneficial conversion feature $0 in 2010 and $492 in 2011) | - | 695 | ||||||
| 4% unsecured convertible note due February 2013 (net of discount related to beneficial conversion feature $0 in 2010 and $55 in 2011) | - | 151 | ||||||
| 10% unsecured convertible note due November 2012 (net of discount related to beneficial conversion feature $0 in 2010 and $2,333 in 2011) | - | 667 | ||||||
| $ | 555 | $ | 1,513 | |||||
| Long term convertible notes related party, net | ||||||||
| 20% unsecured convertible note due December 2013 (net of discount reated to beneficial conversion feature and original issue discount $367 in 2011) | $ | 949 | $ | 185 | ||||
| Total notes payable, net | $ | 9,954 | $ | 12,253 | ||||
Three months endedSeptember 30, | Nine Months endedSeptember 30, | |||||||||||||||
| 2010 | 2011 | 2010 | 2011 | |||||||||||||
| Common stock options | 4,110 | 25,186 | 4,110 | 25,186 | ||||||||||||
| Common stock warrants | 38,200 | 48,606 | 38,200 | 53,215 | ||||||||||||
| Convertible notes | 20,000 | 12,111 | 20,000 | 31,651 | ||||||||||||
| Excluded potentially dilutive securities | 62,310 | 85,903 | 62,310 | 110,052 | ||||||||||||

December 31, 2009 | December 31, 2010 | |||||||
| (In thousands) | ||||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash | $ | 65 | $ | 153 | ||||
| Prepaid expenses and other current assets | 36 | 86 | ||||||
| Total current assets | 101 | 239 | ||||||
| Property and equipment: | ||||||||
| Laboratory equipment | 29 | 29 | ||||||
| Office furniture and other equipment | 82 | 123 | ||||||
| 111 | 152 | |||||||
| Less accumulated depreciation and amortization | (111 | ) | (113 | ) | ||||
| Property and equipment, net | - | 39 | ||||||
| Deposit and other non-current assets | 2 | 16 | ||||||
| Total assets | $ | 103 | $ | 294 | ||||
| LAIBILITIES AND STOCKHOLDERS' EQUITY (DEFICIT) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 3,249 | $ | 2,835 | ||||
| Accounts payable, related party | 6,328 | 10,527 | ||||||
| Accrued expenses | 1,874 | 2,074 | ||||||
| Accrued expenses, related party | 1,329 | 1,749 | ||||||
| Notes payable | 2,650 | 1,364 | ||||||
| Note payable to related parties | 4,000 | 4,000 | ||||||
| Convertible notes payable, net | - | 2,736 | ||||||
| Embedded derivative liability | - | 839 | ||||||
| Total current liabilities | 19,430 | 26,124 | ||||||
| Long term liabilities: | ||||||||
| Notes payable | - | 350 | ||||||
| Convertible notes payable, net | 1,061 | 555 | ||||||
| Convertible notes payable to related party, net | 298 | 949 | ||||||
| Total long term liabilities | 1,359 | 1,854 | ||||||
| Total liabilities | 20,789 | 27,978 | ||||||
| Stockholders’ equity (deficit): | ||||||||
| Preferred stock, $0.001 par value; 20,000,000 shares authorized and none issued and outstanding | ||||||||
| Common stock, $0.001 par value; 150,000,000 shares authorized, 58,877,087 and 73,118,471 shares issued and outstanding at December 31, 2009 and 2010, respectively | 58 | 73 | ||||||
| Additional paid-in capital | 170,885 | 191,344 | ||||||
| Deficit accumulated during the development stage | (191,580 | ) | (218,948 | ) | ||||
| Cumulative translation adjustment | (49 | ) | (153 | ) | ||||
| Total stockholders’ equity (deficit) | (20,686 | ) | (27,684 | ) | ||||
| Total liabilities and stockholders’ equity (deficit) | $ | 103 | $ | 294 | ||||
| Period from | ||||||||||||
| March 18, | ||||||||||||
| 1996 | ||||||||||||
| (Inception) | ||||||||||||
| Year Ended December 31 | to December | |||||||||||
| 2009 | 2010 | 31, 2010 | ||||||||||
| (in thousands, except per share data) | ||||||||||||
| Revenues: | ||||||||||||
| Research material sales | $ | 10 | $ | 10 | $ | 570 | ||||||
| Contract research and development from related parties | - | - | 1,128 | |||||||||
| Research grants and other | - | - | 1,061 | |||||||||
| Total revenues | 10 | 10 | 2,759 | |||||||||
| Operating cost and expenses: | ||||||||||||
| Cost of research material sales | - | - | 382 | |||||||||
| Research and development | 9,588 | 9,899 | 76,812 | |||||||||
| General and administration | 7,482 | 5,463 | 61,989 | |||||||||
| Depreciation and amortization | 7 | 2 | 2,353 | |||||||||
| Loss on facility sublease | - | - | 895 | |||||||||
| Asset impairment loss and other (gain) loss | 389 | - | 2,445 | |||||||||
| Total operating costs and expenses | 17,466 | 15,364 | 144,876 | |||||||||
| Loss from operations | (17,456 | ) | (15,354 | ) | (142,117 | ) | ||||||
| Other income (expense): | ||||||||||||
| Warrant valuation | - | - | 6,759 | |||||||||
| Loan conversion inducement | (5,617 | ) | (4,673 | ) | (10,290 | ) | ||||||
| Derivative valuation gain (loss) | - | 54 | 54 | |||||||||
| Gain on sale of intellectual property and property and equipment | - | - | 3,664 | |||||||||
| Interest expense | (3,881 | ) | (7,884 | ) | (33,916 | ) | ||||||
| Interest income and other | - | 489 | 1,707 | |||||||||
| Net loss | (26,954 | ) | (27,368 | ) | (174,139 | ) | ||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | - | - | (12,349 | ) | ||||||||
| Modification of Series A preferred stock warrants | - | - | (2,306 | ) | ||||||||
| Modification of Series A-1 preferred stock warrants | - | - | (16,393 | ) | ||||||||
| Series A preferred stock dividends | - | - | (334 | ) | ||||||||
| Series A-1 preferred stock dividends | - | - | (917 | ) | ||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | - | - | (4,664 | ) | ||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | - | - | (1,872 | ) | ||||||||
| Series A preferred stock redemption fee | - | - | (1,700 | ) | ||||||||
| Beneficial conversion feature of Series D preferred stock | - | - | (4,274 | ) | ||||||||
| Net loss applicable to common stockholders | $ | (26,954 | ) | $ | (27,368 | ) | $ | (218,948 | ) | |||
| Net loss per share applicable to common stockholders — basic and diluted | $ | (0.56 | ) | $ | (0.41 | ) | ||||||
| Weighted average shares used in computing basic and diluted net loss per share | 47,961 | 67,063 | ||||||||||
| Deficit | ||||||||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||||||||
| Preferred Stock | Preferred Stock | Additional | During the | Cumulative | Total | |||||||||||||||||||||||||||||
| Common Stock | Series A | Series A-1 | Paid-In | Deferred | Development | Translation | Stockholders’ | |||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | Capital | Compensation | Stage | Adjustment | Equity (Deficit) | ||||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||||||||
| Balances at March 18, 1996 | — | $ | — | — | $ | — | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||||
| Accretion of membership units mandatory redemption obligation | — | — | — | — | — | — | — | — | (106 | ) | — | (106 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | (1,233 | ) | — | (1,233 | ) | ||||||||||||||||||||||
| Balances at December 31, 1996 | — | �� | — | — | — | — | — | — | — | (1,339 | ) | — | (1,339 | ) | ||||||||||||||||||||
| Accretion of membership units mandatory redemption obligation | — | — | — | — | — | — | — | — | (275 | ) | — | (275 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (2,560 | ) | — | (2,560 | ) | |||||||||||||||||||||
| Balances at December 31, 1997 | — | — | — | — | — | — | — | — | (4,174 | ) | — | (4,174 | ) | |||||||||||||||||||||
| Conversion of membership units to common stock | 2,203 | 2 | — | — | — | — | — | — | (2 | ) | — | — | ||||||||||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | — | — | — | — | — | — | — | — | (329 | ) | — | (329 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (4,719 | ) | — | (4,719 | ) | |||||||||||||||||||||
| Balances at December 31, 1998 | 2,203 | 2 | — | — | — | — | — | — | (9,224 | ) | — | (9,222 | ) | |||||||||||||||||||||
| Issuance of Series C preferred stock warrants for services related to sale of Series C preferred shares | — | — | — | — | — | — | 394 | — | — | — | 394 | |||||||||||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | — | — | — | — | — | — | — | — | (354 | ) | — | (354 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (5,609 | ) | — | (5,609 | ) | |||||||||||||||||||||
| Balances at December 31, 1999 | 2,203 | 2 | — | — | — | — | 394 | — | (15,187 | ) | — | (14,791 | ) | |||||||||||||||||||||
| Issuance of Series C preferred stock warrants in connection with lease agreement | — | — | — | — | — | — | 43 | — | — | — | 43 | |||||||||||||||||||||||
| Exercise of stock options for cash | 2 | — | — | — | — | — | 1 | — | — | — | 1 | |||||||||||||||||||||||
| Issuance of common stock at $0.85 per share for license rights | 5 | — | — | — | — | — | 4 | — | — | — | 4 | |||||||||||||||||||||||
| Issuance of Series D preferred stock warrants in convertible promissory note offering | — | — | — | — | — | — | 4,039 | — | — | — | 4,039 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory notes | — | — | — | — | — | — | 1,026 | — | — | — | 1,026 | |||||||||||||||||||||||
| Issuance of Series D preferred stock warrants for services related to sale of Series D preferred shares | — | — | — | — | — | — | 368 | — | — | — | 368 | |||||||||||||||||||||||
| Issuance of common stock warrants in conjunction with issuance of promissory note | — | — | — | — | — | — | 3 | — | — | — | 3 | |||||||||||||||||||||||
| Cancellation of common stock | (275 | ) | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | — | — | — | — | — | — | — | — | (430 | ) | — | (430 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | (12,779 | ) | — | (12,779 | ) | ||||||||||||||||||||||
| Balances at December 31, 2000 | 1,935 | 2 | — | — | — | — | 5,878 | — | (28,396 | ) | — | (22,516 | ) | |||||||||||||||||||||
| Issuance of Series D preferred stock warrants in conjunction with refinancing of note payable to stockholder | — | — | — | — | — | — | 225 | — | — | — | 225 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory note | — | — | — | — | — | — | 456 | — | — | — | 456 | |||||||||||||||||||||||
| Beneficial conversion feature of Series D preferred stock | — | — | — | — | — | — | 4,274 | — | (4,274 | ) | — | — | ||||||||||||||||||||||
| Issuance of Series D preferred stock warrants for services related to the sale of Series D preferred shares | — | — | — | — | — | — | 2,287 | — | — | — | 2,287 | |||||||||||||||||||||||
| Exercises of stock options and warrants for cash | 1,158 | 1 | — | — | — | — | 407 | — | — | — | 408 | |||||||||||||||||||||||
| Issuance of common stock in initial public offering for cash, net of offering costs of $2,845 | 4,000 | 4 | — | — | — | — | 17,151 | — | — | — | 17,155 | |||||||||||||||||||||||
| Conversion of preferred stock into common stock | 9,776 | 10 | — | — | — | — | 31,569 | — | — | — | 31,579 | |||||||||||||||||||||||
| Series A preferred stock redemption fee | — | — | — | — | — | — | — | — | (1,700 | ) | — | (1,700 | ) | |||||||||||||||||||||
| Issuance of stock options to nonemployees for services | — | — | — | — | — | — | 45 | — | — | — | 45 | |||||||||||||||||||||||
| Deferred compensation related to employee stock options | — | — | — | — | — | — | 1,330 | (1,330 | ) | — | — | — | ||||||||||||||||||||||
| Amortization of deferred compensation | — | — | — | — | — | — | — | 314 | — | — | 314 | |||||||||||||||||||||||
| Accretion of Series A preferred stock mandatory redemption obligation | — | — | — | — | — | — | — | — | (379 | ) | — | (379 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (10,940 | ) | — | (10,940 | ) | |||||||||||||||||||||
| Balances at December 31, 2001 | 16,869 | 17 | — | — | — | — | 63,622 | (1,016 | ) | (45,689 | ) | — | 16,934 | |||||||||||||||||||||
| Issuance of unregistered common stock | 1,000 | 1 | — | — | — | — | 199 | — | — | — | 200 | |||||||||||||||||||||||
| Issuance of common stock, Employee Stock Purchase Plan | 9 | — | — | — | — | — | 6 | — | — | — | 6 | |||||||||||||||||||||||
| Issuance of common stock warrants to Medarex | — | — | — | — | — | — | 80 | — | — | — | 80 | |||||||||||||||||||||||
| Issuance of restricted stock to nonemployees | 8 | — | — | — | — | — | 34 | — | — | — | 34 | |||||||||||||||||||||||
| Issuance of stock options to nonemployees for service | — | — | — | — | — | — | 57 | — | — | — | 57 | |||||||||||||||||||||||
| Issuance of stock options to employees | — | — | — | — | — | — | 22 | (22 | ) | — | — | — | ||||||||||||||||||||||
| Cancellation of employee stock options | — | — | — | — | — | — | (301 | ) | 301 | — | — | — | ||||||||||||||||||||||
| Exercise of stock options and warrants for cash | 32 | — | — | — | — | — | 18 | — | — | — | 18 | |||||||||||||||||||||||
| Deferred compensation related to employee restricted stock option | 99 | — | — | — | — | — | 449 | (449 | ) | — | — | — | ||||||||||||||||||||||
| Cancellation of employee restricted stock grants | (87 | ) | — | — | — | — | — | (392 | ) | 392 | — | — | — | |||||||||||||||||||||
| Amortization of deferred compensation, net | — | — | — | — | — | — | — | 350 | — | — | 350 | |||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (12,804 | ) | — | (12,804 | ) | |||||||||||||||||||||
| Balances at December 31, 2002 | 17,930 | 18 | — | — | — | — | 63,794 | (444 | ) | (58,493 | ) | — | 4,875 | |||||||||||||||||||||
| Issuance of unregistered common stock to Medarex | 1,000 | 1 | — | — | — | — | 199 | — | — | — | 200 | |||||||||||||||||||||||
| Issuance of unregistered common stock to Nexus | 90 | — | — | — | — | — | 35 | — | — | — | 35 | |||||||||||||||||||||||
| Issuance of common stock warrants to Medarex | — | — | — | — | — | — | 80 | — | — | — | 80 | |||||||||||||||||||||||
| Issuance of warrants with convertible promissory note | — | — | — | — | — | — | 221 | — | — | — | 221 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory note | — | — | — | — | — | — | 114 | — | — | — | 114 | |||||||||||||||||||||||
| Issuance of common stock, Employee Stock Purchase Plan | 4 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Exercise of stock options and warrants for cash | 8 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Cancellation of employee restricted stock grants | (4 | ) | — | — | — | — | — | (20 | ) | 20 | — | — | — | |||||||||||||||||||||
| Cancellation of employee stock options | — | — | — | — | — | — | (131 | ) | 131 | — | — | — | ||||||||||||||||||||||
| Amortization of deferred compensation, net | — | — | — | — | — | — | — | 240 | — | — | 240 | |||||||||||||||||||||||
| Non-employee stock compensation | — | — | — | — | — | — | 2 | — | — | — | 2 | |||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (5,752 | ) | — | (5,752 | ) | |||||||||||||||||||||
| Balances at December 31, 2003 | 19,028 | 19 | — | — | — | — | 64,294 | (53 | ) | (64,245 | ) | — | 15 | |||||||||||||||||||||
| Issuance of warrants with convertible promissory note | — | — | — | — | — | — | 1,711 | — | — | — | 1,711 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory note | — | — | — | — | — | — | 1,156 | — | — | — | 1,156 | |||||||||||||||||||||||
| Issuance of common stock, Employee Stock Purchase Plan | 1 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Cancellation of employee stock options | — | — | — | — | — | — | (5 | ) | 5 | — | — | — | ||||||||||||||||||||||
| Amortization of deferred compensation, net | — | — | — | — | — | — | — | 41 | — | — | 41 | |||||||||||||||||||||||
| Warrant valuation | — | — | — | — | 368 | — | — | — | 368 | |||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (8,508 | ) | — | (8,508 | ) | |||||||||||||||||||||
| Balances at December 31, 2004 | 19,029 | 19 | — | — | — | — | 67,524 | (7 | ) | (72,753 | ) | — | (5,217 | ) | ||||||||||||||||||||
| Issuance of unregistered common stock and preferred stock to Toucan Capital | — | — | 32,500 | 33 | — | — | 1,243 | — | — | — | 1,276 | |||||||||||||||||||||||
| Issuance of stock options to non-employees for services | — | — | — | — | — | — | 3 | — | — | — | 3 | |||||||||||||||||||||||
| Issuance of warrants with convertible promissory note | — | — | — | — | — | — | 1,878 | — | — | — | 1,878 | |||||||||||||||||||||||
| Exercise of stock options and warrants for cash | 49 | — | — | — | — | — | 4 | — | — | — | 4 | |||||||||||||||||||||||
| Amortization of deferred compensation, net | — | — | — | — | — | — | — | 7 | — | — | 7 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory note | — | — | — | — | — | — | 1,172 | — | — | — | 1,172 | |||||||||||||||||||||||
| Common Stock warrant liability | — | — | — | — | — | — | (604 | ) | — | — | — | (604 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (9,937 | ) | — | (9,937 | ) | |||||||||||||||||||||
| Balances at December 31, 2005 | 19,078 | 19 | 32,500 | 33 | — | — | 71,220 | — | (82,690 | ) | — | (11,418 | ) | |||||||||||||||||||||
| Issuance of common stock to PIPE Investors for cash, net of cash and non-cash offering costs of $837 | 39,468 | 39 | — | — | — | — | 4,649 | — | — | — | 4,688 | |||||||||||||||||||||||
| Issuance of warrants to PIPE investment bankers | — | — | — | — | — | — | 395 | — | — | — | 395 | |||||||||||||||||||||||
| Conversion of notes payable due to Toucan Capital to Series A-1 preferred stock | — | — | — | — | 4,817 | 5 | 7,702 | — | — | — | 7,707 | |||||||||||||||||||||||
| Conversion of notes payable due to management to common stock | 2,688 | 3 | — | — | — | — | 266 | — | — | — | 269 | |||||||||||||||||||||||
| Issuance of warrants with convertible promissory notes | — | — | — | — | — | — | 236 | — | — | — | 236 | |||||||||||||||||||||||
| Exercise of stock options and warrants for cash | 66 | — | — | — | — | — | 9 | — | — | — | 9 | |||||||||||||||||||||||
| Exercise of stock options and warrants — cashless | 3,942 | 4 | — | — | — | — | (4 | ) | — | — | — | — | ||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | — | — | 19 | — | — | — | 19 | |||||||||||||||||||||||
| Beneficial conversion feature of convertible promissory note | — | — | — | — | — | — | 64 | — | — | — | 64 | |||||||||||||||||||||||
| Common Stock warrant liability | — | — | — | — | — | — | (6,523 | ) | — | — | — | (6,523 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (1,395 | ) | — | (1,395 | ) | |||||||||||||||||||||
| Balances at December 31, 2006 | 65,241 | 65 | 32,500 | 33 | 4,817 | 5 | 78,033 | — | (84,085 | ) | — | (5,949 | ) | |||||||||||||||||||||
| Conversion of common stock at par related to the reverse stock split | (60,892 | ) | (61 | ) | — | — | — | — | 61 | — | — | — | — | |||||||||||||||||||||
| Conversion of Series A and A-1 preferred stock into common stock | 15,012 | 15 | (32,500 | ) | (33 | ) | (4,817 | ) | (5 | ) | 23 | — | — | — | — | |||||||||||||||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | 6,861 | 7 | — | — | — | — | 12,342 | — | (12,349 | ) | — | — | ||||||||||||||||||||||
| Modification of preferred stock Series A and Series A-1 warrants | — | — | — | — | — | — | 18,699 | — | (18,699 | ) | — | — | ||||||||||||||||||||||
| Series A and Series A-1 preferred stock dividend payment | — | — | — | — | — | — | — | — | (1,251 | ) | — | (1,251 | ) | |||||||||||||||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | — | — | — | — | — | — | 4,664 | — | (4,664 | ) | — | — | ||||||||||||||||||||||
| Issuance of common stock in initial public offering on the AIM London market for cash, net of offering costs of $3,965 | 15,789 | 16 | — | — | — | — | 25,870 | — | — | — | 25,886 | |||||||||||||||||||||||
| Remeasurement of warrants issued in connection with convertible promissory notes | — | — | — | — | — | — | 4,495 | — | — | — | 4,495 | |||||||||||||||||||||||
| Remeasurement of beneficial conversion feature related to convertible promissory notes | — | — | — | — | — | — | 1,198 | — | — | — | 1,198 | |||||||||||||||||||||||
| Exercise of warrants — cashless | 335 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | — | — | 2,679 | — | — | — | 2,679 | |||||||||||||||||||||||
| Cumulative translation adjustment | — | — | — | — | — | — | — | — | — | (4 | ) | (4 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (21,247 | ) | — | (21,247 | ) | |||||||||||||||||||||
| Total comprehensive loss | (21,251 | ) | ||||||||||||||||||||||||||||||||
| Balances at December 31, 2007 | 42,346 | 42 | — | — | — | — | 148,064 | — | (142,295 | ) | (4 | ) | 5,807 | |||||||||||||||||||||
| Stock issuance in exchange for license option | 122 | — | — | — | — | — | 225 | — | — | — | 225 | |||||||||||||||||||||||
| Exercise of stock options — cashless | 25 | — | — | — | — | — | 1 | — | — | — | 1 | |||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | — | — | 3,001 | — | — | — | 3,001 | |||||||||||||||||||||||
| — | ||||||||||||||||||||||||||||||||||
| Issuance of warrants with promissory notes | — | — | — | — | — | — | 1,017 | — | — | — | 1,017 | |||||||||||||||||||||||
| Cumulative translation adjustment | — | — | — | — | — | — | — | — | — | (20 | ) | (20 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (22,331 | ) | — | (22,331 | ) | |||||||||||||||||||||
| Total comprehensive loss | (22,351 | ) | ||||||||||||||||||||||||||||||||
| Balances at December 31, 2008 | 42,493 | 42 | — | — | — | — | 152,308 | — | (164,626 | ) | (24 | ) | (12,300 | ) | ||||||||||||||||||||
| Exercise of stock options — cashless | 20 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Exercise of warrants — cashless | 1,214 | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||
| Issuance of common stock in private placements | 2,378 | 2 | — | — | — | — | 1,391 | — | — | — | 1,393 | |||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | — | — | 2,712 | — | — | — | 2,618 | |||||||||||||||||||||||
| Debt Discount related to beneficial conversion | — | — | — | — | — | — | 2,578 | — | — | — | 2,578 | |||||||||||||||||||||||
| Warrants issued for services | — | — | — | — | — | — | 1,645 | — | — | — | 1,645 | |||||||||||||||||||||||
| Stock and warrants issued for services | 3,662 | 3 | — | — | — | — | 2,819 | — | — | — | 2,916 | |||||||||||||||||||||||
| Loan conversion | 563 | 1 | — | — | — | — | 111 | — | — | — | 112 | |||||||||||||||||||||||
| Loan conversion and conversion inducement | 8,547 | 10 | — | — | — | — | 7,321 | — | — | — | 7,331 | |||||||||||||||||||||||
| Cumulative translation adjustment | — | — | — | — | — | — | — | — | — | (25 | ) | (25 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (26,954 | ) | — | (26,954 | ) | |||||||||||||||||||||
| Total comprehensive loss | (26,979 | ) | ||||||||||||||||||||||||||||||||
| Balances at December 31, 2009 | 58,877 | 58 | — | — | — | — | 170,885 | — | (191,580 | ) | (49 | ) | (20,686 | ) | ||||||||||||||||||||
| Stock compensation expense | — | — | — | — | — | — | 2,004 | — | — | — | 2,004 | |||||||||||||||||||||||
| Debt discount related to beneficial conversion and warrants | — | — | — | — | — | — | 3,254 | — | — | — | 3,254 | |||||||||||||||||||||||
| Stock and warrants issued for services | 2,264 | 2 | — | — | — | — | 2,246 | — | — | — | 2,248 | |||||||||||||||||||||||
| Interest extensions and warrant valuations | 1,139 | 1 | — | — | — | — | 2,627 | — | — | — | 2,628 | |||||||||||||||||||||||
| Issuance of common stock in private placements | 5,118 | 6 | — | — | — | — | 3,832 | — | — | — | 3,838 | |||||||||||||||||||||||
| Loan conversion and conversion inducement | 5,720 | 6 | — | — | — | — | 6,406 | — | — | — | 6,412 | |||||||||||||||||||||||
| Sale of warrants | — | — | — | — | — | — | 90 | — | — | — | 90 | |||||||||||||||||||||||
| Cumulative translation adjustment | — | — | — | — | — | — | — | — | — | (104 | ) | (104 | ) | |||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (27,368 | ) | — | (27,368 | ) | |||||||||||||||||||||
| Total comprehensive loss | (27,472 | ) | ||||||||||||||||||||||||||||||||
| Balance at December 31, 2010 | 73,118 | $ | 73 | $ | — | $ | — | $ | — | $ | — | $ | 191,344 | $ | — | $ | (218,948 | ) | $ | (153 | ) | $ | (27,684 | ) | ||||||||||
Period from March 18, | ||||||||||||
| 1996 (Inception) to | ||||||||||||
| Year Ended December 31, | December 31, | |||||||||||
| 2009 | 2010 | 2010 | ||||||||||
| (in thousands) | ||||||||||||
| Cash Flows from Operating Activities: | ||||||||||||
| Net Loss | $ | (26,954 | ) | $ | (27,368 | ) | $ | (174,139 | ) | |||
| Reconciliation of net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 7 | 2 | 2,353 | |||||||||
| Amortization of deferred financing costs | - | - | 320 | |||||||||
| Amortization debt discount | 1,337 | 4,508 | 24,209 | |||||||||
| Derivative valuation (gain) loss | - | (54 | ) | (54 | ) | |||||||
| Accrued interest converted to preferred stock | - | - | 260 | |||||||||
| Accreted interest on convertible promissory note | - | - | 1,484 | |||||||||
| Stock-based compensation costs | 2,618 | 2,004 | 11,414 | |||||||||
| Stock and warrants issued for services and other expenses | 4,776 | 4,559 | 9,335 | |||||||||
| Loan conversion inducement | 5,617 | 4,673 | 10,290 | |||||||||
| Warrant valuation | - | - | (6,759 | ) | ||||||||
| Asset impairment loss and loss (gain) on sale of properties | 389 | - | (936 | ) | ||||||||
| Loss on facility sublease | - | - | 895 | |||||||||
| Increase (decrease) in cash resulting from changes in assets and liabilities: | ||||||||||||
| Accounts receivable | 1 | - | - | |||||||||
| Prepaid expenses and other current assets | 1,031 | (64 | ) | 624 | ||||||||
| Accounts payable and accrued expenses | 405 | 745 | 5,790 | |||||||||
| Related party accounts payable and accrued expenses | 6,096 | 4,619 | 12,276 | |||||||||
| Accrued loss on sublease | - | - | (265 | ) | ||||||||
| Deferred rent | - | - | 410 | |||||||||
| Net Cash used in Operating Activities | (4,677 | ) | (6,376 | ) | (102,493 | ) | ||||||
| Cash Flows from Investing Activities: | ||||||||||||
| Purchase of property and equipment, net | (2 | ) | (41 | ) | (5,044 | ) | ||||||
| Proceeds from sale of property and equipment | - | - | 258 | |||||||||
| Proceeds from sale of intellectual property | - | - | 1,816 | |||||||||
| Proceeds from sale of marketable securities | - | - | 2,000 | |||||||||
| Refund of security deposit | - | - | (3 | ) | ||||||||
| Transfer of restricted cash | - | - | (1,035 | ) | ||||||||
| Net Cash used in Investing Activities | (2 | ) | (41 | ) | (2,008 | ) | ||||||
| Cash Flows from Financing Activities: | ||||||||||||
| Proceeds from issuance of note payable | 2,060 | 1,140 | 5,850 | |||||||||
| Proceeds from issuance of convertible notes payable to related parties | 1,300 | - | 1,300 | |||||||||
| Proceeds from issuance of note payable to related parties | - | - | 11,250 | |||||||||
| Repayment of note payable to related party | - | (900 | ) | (7,600 | ) | |||||||
| Proceeds from issuance of convertible promissory note and warrants, net of issuance costs | - | 2,992 | 16,091 | |||||||||
| Repayment of convertible promissory note | - | (551) | (670 | ) | ||||||||
| Borrowing under line of credit, Northwest Hospital | - | - | 2,834 | |||||||||
| Repayment of line of credit, Northwest Hospital | - | - | (2,834 | ) | ||||||||
| Payment on capital lease obligations | - | - | (323 | ) | ||||||||
| Payments on note payable | - | - | (420 | ) | ||||||||
| Proceeds from issuance preferred stock, net | - | - | 28,708 | |||||||||
| Proceeds from exercise of stock options and warrants | - | - | 228 | |||||||||
| Proceeds from issuance common stock, net | 1,393 | 3,838 | 53,574 | |||||||||
| Proceeds for sale of stock warrant | - | 90 | 90 | |||||||||
| Payment of preferred stock dividends | - | - | (1,251 | ) | ||||||||
| Series A preferred stock redemption fee | - | - | (1,700 | ) | ||||||||
| Deferred financing costs | - | - | (320 | ) | ||||||||
| Net Cash provided by Financing Activities | 4,753 | 6,609 | 104,807 | |||||||||
| Effect of exchange rates on cash | (25 | ) | (104 | ) | (153 | ) | ||||||
| Net increase in cash | 49 | 88 | 153 | |||||||||
| Cash at beginning of period | 16 | 65 | - | |||||||||
| Cash at end of period | $ | 65 | $ | 153 | $ | 153 | ||||||
| Supplemental disclosure of cash flow information — Cash paid during the period for interest | $ | - | $ | - | $ | 1,879 | ||||||
| Supplemental schedule of non-cash financing activities: | ||||||||||||
| Equipment acquired through capital leases | $ | - | $ | - | $ | 285 | ||||||
| Issuance of common stock in connection with elimination of Series A and Series A-1 preferred stock preferences | - | - | 12,349 | |||||||||
| Issuance of common stock in connection conversion of notes payable and accrued interest | 1,500 | 1,786 | 3,286 | |||||||||
| Modification of Series A preferred stock warrants | - | - | 2,306 | |||||||||
| Modification of Series A-1 preferred stock warrants | - | - | 16,393 | |||||||||
| Warrants issued on Series A and Series A-1 preferred stock dividends | - | - | 4,664 | |||||||||
| Common stock warrant liability | - | - | 11,841 | |||||||||
| Accretion of mandatorily redeemable Series A preferred stock redemption obligation | - | - | 1,872 | |||||||||
| Debt discount on promissory notes | 2,578 | 3,254 | 14,091 | |||||||||
| Conversion of convertible promissory notes and accrued interest to Series D preferred stock | - | - | 5,324 | |||||||||
| Conversion of convertible promissory notes and accrued interest to Series A-1 preferred stock | - | - | 7,707 | |||||||||
| Conversion of convertible promissory notes and accrued interest to common stock | - | - | 269 | |||||||||
| Issuance of Series C preferred stock warrants in connection with lease agreement | - | - | 43 | |||||||||
| Issuance of common stock for license rights | - | - | 4 | |||||||||
| Liability for and issuance of common stock and warrants to Medarex | - | - | 840 | |||||||||
| Issuance of common stock to landlord | - | - | 35 | |||||||||
| Deferred compensation on issuance of stock options and restricted stock grants | - | - | 759 | |||||||||
| Cancellation of options and restricted stock | - | - | 849 | |||||||||
| Financing of prepaid insurance through note payable | - | - | 491 | |||||||||
| Stock subscription receivable | - | - | 480 | |||||||||
| • | an aggregate of 12,866,324 shares of Common Stock; |
| • | warrants to purchase 9,433,821 shares of Common Stock at an exercise price of $0.60 per share (net of 4,716,911 warrants transferred to Regen Med stockholders); and |
| • | warrants to purchase 5,256,238 shares of Common Stock at an exercise price of $0.15 per share (net of 2,628,119 warrants transferred to Regen Med stockholders). |
| • | an aggregate of 10,510,691 shares of Common Stock; |
| • | warrants to purchase 8,832,541 shares of Common Stock at an exercise price of $0.60 per share; |
| • | warrants to purchase 1,097,561 shares of Common Stock at an exercise price of $0.82 per share; |
| • | warrants to purchase 132,500 shares of common stock at an exercise price of $0.40; |
| • | warrants to purchase 86,667 shares of common stock at an exercise price of $0.75 per share; and |
| • | warrants to purchase 842,375 shares of common stock at an exercise price of $0.20. |
| 2009 | 2010 | |||||||
| Research and development | $ | 770 | $ | 593 | ||||
| General and administrative expenses | 1,848 | 1,411 | ||||||
| Total stock- based compensation expense | $ | 2,618 | $ | 2,004 | ||||
| 2009 | 2010 | |||||||
| Risk free interest rate | 3.6 | % | 3.6 | % | ||||
| Volatility | 208 | % | 208 | % | ||||
| Expected term | 10 years | 10 years | ||||||
| Expected dividends | 0 | % | 0 | % | ||||
| · | Level 1, defined as observable inputs such as quoted prices for identical instruments in active markets; |
| · | Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable such as quoted prices for similar instruments in active markets or quoted prices for identical or similar instruments in markets that are not active; and |
| · | Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions, such as valuations derived from valuation techniques in which one or more significant inputs or significant value drivers are unobservable. |
| Balance, January 1, 2010 | $ | - | ||
| Fair value upon issuance, Down-round protection on September Note warrants | 189,000 | |||
| Fair value upon issuance, Down-round protection on October Note warrants | 329,000 | |||
| Fair value upon issuance, Down-round protection on October Note warrants | 161,000 | |||
| Fair value upon issuance, Down-round protection on note payable conversion feature | 214,000 | |||
| Net change in fair value of derivative liabilities (an unrealized gain) | (54,000 | ) | ||
| Balance at December 31, 2010 | $ | 839,000 |
| Options | Weighted- Average Exercise Price | Weighted- Average Remaining Contractual Term (Years) | Aggregate Intrinsic Value | |||||||||||||
| Outstanding as of December 31, 2009 | 4,108 | $ | 0.69 | |||||||||||||
| Granted | 100 | 0.75 | ||||||||||||||
| Exercised | (99 | ) | 0.75 | |||||||||||||
| Expired | (2 | ) | 15.15 | |||||||||||||
| Forfeited | (851 | ) | 0.60 | |||||||||||||
| Outstanding as of December 31, 2010 | 3,256 | 0.71 | 8.3 | |||||||||||||
| Exercisable as of December 31, 2010 | 2,305 | 0.71 | 8.5 | $ | — | |||||||||||
| Weighted- | ||||||||
| Average | ||||||||
| Grant Date | ||||||||
| Options | Fair Value | |||||||
| Outstanding at December 31, 2009 | 1,816 | $ | 0.68 | |||||
| Granted during 2010 | 100 | 0.75 | ||||||
| Expired/Forfeited during 2010 | (2 | ) | 15.15 | |||||
| Vested during 2010 | (963 | ) | 0.67 | |||||
| Outstanding at December 31, 2010 | 951 | 0.70 | ||||||
| Options Outstanding | Options Exercisable | |||||||||||||||||||
Range of Exercise Prices | Number Outstanding | Weighted- Average Remaining Contractual Life (Years) | Weighted Average Exercise Price | Number Exercisable | Weighted Average Exercise Price | |||||||||||||||
| $ 0.55 - 0.60 | 2,237 | 8.37 | $ | 0.56 | 1,875 | $ | 0.56 | |||||||||||||
| $ 0.61 - 2.40 | 1,011 | 8.23 | $ | 0.88 | 421 | $ | 1.03 | |||||||||||||
| $ 2.41 - 75.00 | 8 | 1.24 | $ | 21.43 | 8 | $ | 21.43 | |||||||||||||
| Total | 3,256 | 8.30 | $ | 0.71 | 2,304 | $ | 0.70 | |||||||||||||
| Date of Issue | Related transaction | Warrants Issued | Exercise Price | Warrants outstanding at December 31, 2010 | ||||||||||
| Issued to Toucan Capital | ||||||||||||||
| June 2007 | See Conversion Agreement in Note 2 | 14,150,732 | $ | 0.60 | 9,433,821 | |||||||||
| June 2007 | See Conversion Agreement in Note 3 | 7,884,357 | 0.15 | 5,256,238 | ||||||||||
| Issued to Toucan Partners | ||||||||||||||
| June 2007 | See Conversion Agreement in Note 2 | 8,832,541 | 0.60 | 8,832,541 | ||||||||||
| December 2008 | December 2008 Loan warrants | 132,500 | 0.40 | 132,500 | ||||||||||
| September 2009 | August and December 2008 Loan Conversions | 513,841 | 0.41 | 513,841 | ||||||||||
| September 2009 | Consulting services | 842,375 | 0.2 | 842,375 | ||||||||||
| July 2010 | Equity Investment ($650,000) | 86,667 | 0.75 | 86,667 | ||||||||||
| October 2010 | October $900,000 loan | 1,097,561 | 0.82 | 1,097,561 | ||||||||||
| Date of Issue | Related transaction | Warrants Issued | Exercise Price | Warrants outstanding at December 31, 2010 | ||||||||||
| September 2009 | Al Rajhi loan extension | 1,743,111 | $ | 0.63 | 1,743,111 | |||||||||
| September 2009 | Loan extensions | 861,520 | 0.20 | 861,520 | ||||||||||
| January 2010 | Consultant compensation (1) | 100,000 | 0.75 | 100,000 | ||||||||||
| February 2010 | Consultant compensation (2) | 250,000 | 1.00 | 250,000 | ||||||||||
| April 2010 | Private Placement | 233,333 | 0.75 | 176,996 | ||||||||||
| May 2010 | SDS loan extension | 500,000 | 0.53 | 500,000 | ||||||||||
| July 2010 | Regen Med July 2010 $1,750,000 Loan | 2,333,333 | 0.75 | 1,949,678 | ||||||||||
| July 2010 | Consultant compensation (3) | 300,000 | 0.75 | 300,000 | ||||||||||
| September 2010 | September 28, 2010 $350,000 Loan | 421,687 | 0.83 | 421,687 | ||||||||||
| October 2010 | October 1, 2010 $500,000 Loan | 609,756 | 0.82 | 609,756 | ||||||||||
| October 2010 | Consultant compensation (4) | 20,000 | 0.75 | 20,000 | ||||||||||
| November 2010 | Consultant compensation (5) | 75,833 | 0.75 | 75,833 | ||||||||||
| November 2010 | Warrant issued for cash | 3,250,000 | 1.50 | 3,250,000 | ||||||||||
| December 2010 | December 16, 2010 $100,000 loan | 133,333 | 0.75 | 133,333 | ||||||||||
| December 2010 | December SDS loan warrants | 118,165 | 0.75 | 118,165 | ||||||||||
| Date of Issue | Warrants Outstanding as of December 31, 2010 | Exercise Price | Expiration | ||||||
| June 1, 2007 | 7,628,587 | $ | 0.15 | May 31, 2015 | |||||
| December 3, 2010 | 255,750 | 0.15 | December 2, 2013 | ||||||
| September 30, 2009 | 1,703,625 | 0.20 | September 29, 2012 | ||||||
| December 23, 2008 | 132,500 | 0.40 | December 22, 2016 | ||||||
| November 6, 2008 | 1,354,083 | 0.41 | November 5, 2012 | ||||||
| May 26, 2010 | 500,000 | 0.53 | November 17, 2015 | ||||||
| June 1, 2007 | 21,064,997 | 0.60 | May 31, 2015 | ||||||
| December 3, 2010 | 1,918,275 | 0.60 | December 2, 2013 | ||||||
| September 28, 2009 | 1,743,111 | 0.63 | September 27, 2012 | ||||||
| January 1, 2010 | 100,000 | 0.75 | December 31, 2012 | ||||||
| March 23, 2010 to May 26, 2010 | 233,329 | 0.75 | May 25, 2013 | ||||||
| July 7, 2010 | 300,000 | 0.75 | July 6, 2013 | ||||||
| July 2, 2010 | 86,667 | 0.75 | July 1, 2013 | ||||||
| July 14, 2010 | 1,949,678 | 0.75 | July 13, 2015 | ||||||
| October 13, 2010 | 80,000 | 0.75 | October 12, 2013 | ||||||
| November 23, 2010 | 75,833 | 0.75 | November 22, 2015 | ||||||
| December 3, 2010 | 383,655 | 0.75 | December 2, 2010 | ||||||
| December 13, 2010 | 133,333 | 0.75 | December 12, 2015 | ||||||
| December 29, 2010 | 118,165 | 0.75 | December 28, 2015 | ||||||
| November 29, 2010 | 36,875 | 0.80 | November 23, 2013 | ||||||
| October 1, 2010 | 1,707,317 | 0.82 | September 30, 2015 | ||||||
| November 29,2010 | 421,687 | 0.83 | November 28, 2010 | ||||||
| February 1, 2010 | 250,000 | 1.00 | January 31, 2013 | ||||||
| November 5, 2010 | 3,250,000 | 1.50 | November 4, 2013 | ||||||
| February 9, 2003 | 13,333 | 1.53 | February 8, 2013 | ||||||
| March 30, 2006 | 677,389 | 2.10 | March 29, 2011 | ||||||
| January 8, 2003 | 13,333 | 2.66 | January 7, 2013 | ||||||
| December 26, 2002 | 26,666 | 3.24 | December 25, 2012 | ||||||
| 46,158,188 | |||||||||
| December 31, | ||||||||
| 2009 | 2010 | |||||||
| Common stock options | 4,108 | 3,256 | ||||||
| Common stock warrants | 36,599 | 46,158 | ||||||
| Common stock issuable on conversion of notes payable | 14,177 | 18,606 | ||||||
| Date | Transaction | # of Shares | Price per share | Gross Cash Proceeds | ||||||||||
| January 2009 | Private Placement | 1,000,000 | $ | 0.70 | $ | 700,000 | ||||||||
| March 2010 | Consultant fees paid in stock (1) | 199,661 | 0.69 | |||||||||||
| March 2009 | Private Placement | 1,377,358 | 0.50 | 700,000 | ||||||||||
| September 2009 | Toucan Partners Loan Conversion (2) | 8,547,280 | 0.20 | |||||||||||
| September 2009 | Toucan Partners consulting services (1) | 2,504,034 | 0.75 | |||||||||||
| September 2009 | Consultant fees paid in stock (1) | 397,500 | 0.75 | |||||||||||
| September 2009 | Note conversion | 562,500 | 0.20 | |||||||||||
| December 2009 | Consultant fees paid in stock (1) | 146,306 | 0.82 | |||||||||||
| December 2009 | Exercise of employee stock options and warrants (cashless basis) | 1,236,055 | ||||||||||||
| December 2009 | Stock issued in lieu of salaries (1) | 413,540 | 0.93 | |||||||||||
| Total issued in 2009 | 16,384,234 | $ | 1,400,000 | |||||||||||
| March 2010 | Private Placement | 1,451,667 | 0.75 | $ | 1,088,750 | |||||||||
| March 2010 | Al Rajhi interest conversion (3) | 1,138,603 | 0.75 | |||||||||||
| May 2010 | SDS Loan conversion (4) | 5,300,000 | 0.20 | |||||||||||
| June 2010 | Private Placement | 2,333,333 | 0.75 | 1,750,000 | ||||||||||
| July 2010 | Private Placement | 866,667 | 0.75 | 650,000 | ||||||||||
| Jul;y 2010 | Toucan Partners consulting fees (1) | 8,466 | 0.20 | |||||||||||
| September 2010 | Private Placement | 266,667 | 0.75 | 200,000 | ||||||||||
| October 2010 | Note conversion | 58,971 | 0.77 | |||||||||||
| December 2010 | Stock issued in lieu of salaries (1) | 704,752 | 0.73 | |||||||||||
| December 2010 | Consultant fees paid in stock (1) | 1,559,071 | 0.77 | |||||||||||
| December 2010 | Loan conversion | 353,187 | 0.46 | |||||||||||
| December 2010 | Private Placement | 200,000 | 0.75 | 150,000 | ||||||||||
| Total issued in 2010 | 14,241,384 | $ | 3,838,750 | |||||||||||
| 2009 | 2010 | |||||||
| Net operating loss carry forwards | $ | 36,666 | $ | 41,175 | ||||
| Research and development credit carry forwards | 2,549 | 2,821 | ||||||
| Other | 165 | 25 | ||||||
| Gross deferred tax assets | 39,380 | 44,021 | ||||||
| Less valuation allowance | (39,380 | ) | (44,021 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
| A convertible note in the amount of $370,000. The note matures on December 20, 2011 and carries a one-time interest charge payable at maturity of $37,000. The note is convertible into shares of the Company’s stock at a conversion price equal to 70 percent of the average daily closing bid price for the ten trading day period preceding the date of the conversion notice which was $0.47 on the date the note was issued. |
| A convertible note in the amount of $130,000. The note matures on June 2, 2011 and carries zero interest. The note is convertible into shares of the Company’s stock at a conversion price equal to 80 percent of the average of the five lowest closing prices in the 25 days previous to the conversion date which was $0.54 on the date the note was issued. Any amount that is still outstanding at maturity is repayable in cash. |
| A convertible note in the amount of $500,000. The note matures on June 2, 2011 and carries zero interest. The note is convertible into shares of the Company’s stock at a conversion price equal to the lesser of (i) the average of the closing prices for the ten trading days preceding the issue date of this note which was $0.67, or (ii) seventy percent of the volume weighted average price for the fifteen trading days prior to a conversion date. The conversion price is adjustable for down rounds as long as any amount due is outstanding under the note subject to substantial exceptions. |
December 31, 2009 | December 31, 2010 | |||||||
| 12% note payable to Al Rajhi | $ | 4,000 | $ | 4,000 | ||||
| 6% unsecured convertible note payable to Toucan Partners, due July, 2011 and November, 2011 (net of discount related to beneficial conversion feature $1,002 in 2009 and $351 in 2010) | 298 | 949 | ||||||
| Total notes payable (net) | 4,298 | 4,949 | ||||||
| Less current portion | (4,000 | ) | (4,000 | ) | ||||
| Long-term notes payable to related parties (net) | $ | 298 | $ | 949 | ||||
December 31, 2009 | December 31, 2010 | |||||||
| 12% unsecured note payable to SDS | $ | 1,000 | $ | - | ||||
| 12% unsecured note payable to SDS (net of warrant related discount $76 in 2010) | - | 714 | ||||||
| 12% unsecured notes payable to SDS and Private Investors, | 1,650 | 650 | ||||||
| 6% unsecured convertible notes payable to Private Lenders, due in August and September 2011, (net of discount related to beneficial conversion feature $ 485 in 2009 and $195 in 2010) | 95 | 385 | ||||||
| 6% unsecured convertible notes payable to Private Lenders, due March 25, 2011, (net of discount related to beneficial conversion feature $46 in 2009 and $9 in 2010) | 604 | 641 | ||||||
| 6% unsecured convertible notes payable to Private Lenders, due October, 2011, (net of discount related to beneficial conversion feature $194 in 2009 and $87 in 2010) | 21 | 128 | ||||||
| 6% unsecured convertible notes payable to Private Lenders, due October and December 2011, (net of discount related to beneficial conversion feature $274 in 2009 and $133 in 2010) | 231 | 372 | ||||||
| 6% unsecured convertible note payable to Private Lender, due March 25, 2011 | 110 | 110 | ||||||
| 6% secured convertible note payable to Private Lender due November 27, 2010 | – | 300 | ||||||
| 6% unsecured convertible notes payable to Private Lenders, due March 2012, (net of discount related to beneficial conversion feature $321 in 2010) | – | 555 | ||||||
| 0% unsecured convertible note to Private Lender due December 20, 2011 (net of discount related to beneficial conversion feature of $143 in 2010) | – | 102 | ||||||
| 0% unsecured convertible note to Private Lender due June 2, 2011(net of discount related to beneficial conversion feature $214 in 2010) | – | 286 | ||||||
| 11% unsecured convertible note to Private Lender due June 2, 2011 (net of discount related to beneficial conversion feature of $56 in 2010) | – | 74 | ||||||
| 20% unsecured convertible note to Private Lender due December 14, 2013 | – | 350 | ||||||
| 10% unsecured convertible note to Private Lender due March 17, 2011 (net of discount related to beneficial conversion feature $37 in 2010) | – | 63 | ||||||
| 10% unsecured convertible note to Private Investors due May 29, 2011 (net of discount related to beneficial conversion feature $20 in 2010) | – | 275 | ||||||
| 3,711 | 5,005 | |||||||
| Less current portion | (2,650 | ) | (4,100 | ) | ||||
| Total long-term debt (net) | $ | 1,061 | $ | 905 | ||||
| Notes payable | $ | 1,364 | ||
| Convertible notes payable | 2,736 | |||
| $ | 4,100 |
| Notes payable | $ | 350 | ||
| Convertible notes payable | 555 | |||
| $ | 905 |
First Quarter 2010 | Second Quarter 2010 | Third Quarter 2010 | Fourth Quarter 2010 | |||||||||||||
| Total revenues | $ | — | $ | — | $ | 10 | $ | — | ||||||||
| Net loss applicable to common stockholders | $ | (5,845 | ) | $ | (8,727 | ) | $ | (5,381 | ) | $ | (7,415 | ) | ||||
| Net loss per share applicable to common stockholders — basic and diluted | $ | (0.10 | ) | $ | (0.14 | ) | $ | (0.08 | ) | $ | (0.09 | ) | ||||
| Weighted average shares used in computing basic and diluted loss per share | 59,928 | 63,853 | 70,413 | 72,028 | ||||||||||||
First Quarter 2009 | Second Quarter 2009 | Third Quarter 2009 | Fourth Quarter 2009 | |||||||||||||
| Total revenues | $ | — | $ | — | $ | 10 | $ | — | ||||||||
| Net loss applicable to common stockholders | $ | (4,620 | ) | $ | (4,095 | ) | $ | (13,837 | ) | $ | (4,402 | ) | ||||
| Net loss per share applicable to common stockholders — basic and diluted | $ | (0.11 | ) | $ | (0.09 | ) | $ | (0.31 | ) | $ | (0.05 | ) | ||||
| Weighted average shares used in computing basic and diluted loss per share | 43,385 | 45,069 | 45,276 | 57,966 | ||||||||||||
Exhibit Number | Description | |
| 3.1 | Seventh Amended and Restated Certificate of Incorporation.(3.1)(22) | |
| 3.2 | Third Amended and Restated Bylaws of the Company.(3.1)(29) | |
| 3.3 | Amendment to Seventh Amended and Restated Certificate of Incorporation.(3.2)(29) | |
| 3.4 | Amendment to Seventh Amended and Restated Certificate of Incorporation.(3.4)(33) | |
| 4.1 | Form of common stock certificate.(4.1)(2) | |
| 4.2 | Northwest Biotherapeutics, Inc. Stockholders Rights Agreement dated February 26, 2002 between the Company and Mellon Investors Services, LLC.(4.2)(3) | |
| 4.3 | Form of Rights Certificate.(4.1)(3) | |
| 4.4 | Amendment to Northwest Biotherapeutics, Inc. Stockholders Rights Agreement dated April 26, 2004.(4.2)(4) | |
| 5.1 | Opinion of Tarter Krinsky and Drogin, LLP as to the legality of the securities being registered. | |
| 10.1 | Amended and Restated Loan Agreement and 10% Promissory Note dated November 14, 2005 in the principal amount of $400,000 as amended and restated on April 14, 2007 between the Company and Toucan Partners, LLC.(10.1)(23) | |
| 10.2 | Second Amended and Restated Loan Agreement and 10% Promissory Note originally dated December 30, 2005, and amended and restated on April 17, 2006 and April 14, 2007 in the principal amount of $250,000 between the Company and Toucan Partners, LLC.(10.2)(23) | |
| 10.3 | Second Amended and Restated Loan Agreement and 10% Promissory Note originally dated March 9, 2006, and as amended and restated on April 17, 2006 and April 14, 2007 in the principal amount of $300,000 between the Company and Toucan Partners, LLC.(10.3)(23) | |
| 10.4 | Form of Loan Agreement and 10% Convertible, Promissory Note between the Company and Toucan Partners, LLC.(10.4)(23) | |
| 10.5 | Amended and Restated Investor Rights Agreement dated April 17, 2006.(10.4)(18) | |
| 10.6 | Second Amended and Restated Investor Rights Agreement dated June 22, 2007 between the Company and Toucan Capital Fund II, LLP.(10.3)(29) | |
| 10.7 | Securities Purchase Agreement, dated March 30, 2006 by and among the Company and the Investors identified therein.(10.1)(6) | |
| 10.8 | Form of Warrant.(10.2)(6) | |
| 10.9 | Warrant to purchase securities of the Company dated April 26, 2004 issued to Toucan Capital Fund II, L.P.(10.9)(7) |
| 10.10 | Warrant to purchase securities of the Company dated June 11, 2004 issued to Toucan Capital Fund II, L.P.(10.8)(7) | |
| 10.11 | Warrant to purchase securities of the Company dated July 30, 2004 issued to Toucan Capital Fund II, L.P.(10.7)(7) | |
| 10.12 | Warrant to purchase securities of the Company dated October 22, 2004 issued to Toucan Capital Fund II, L.P.(10.3)(8) | |
| 10.13 | Warrant to purchase securities of the Company dated November 10, 2004 issued to Toucan Capital Fund II, L.P.(10.3)(9) | |
| 10.14 | Warrant to purchase securities of the Company dated December 27, 2004 issued to Toucan Capital Fund II, L.P.(10.3)(10) | |
| 10.15 | First Amendment to Warrants between Northwest Biotherapeutics, Inc. and Toucan Capital Fund II, L.P. dated January 26, 2005.(10.5)(1) | |
| 10.16 | Warrant to purchase Series A Preferred Stock dated January 26, 2005 issued to Toucan Capital Fund II, L.P.(10.2)(1) | |
| 10.17 | Warrant to purchase securities of the Company dated April 12, 2005 issued to Toucan Capital Fund II, L.P.(10.39)(11) | |
| 10.18 | Warrant to purchase securities of the Company dated May 13, 2005 issued to Toucan Capital Fund II, L.P.(10.3)(12) | |
| Warrant to purchase securities of the Company dated June 16, 2005 issued to Toucan Capital Fund II, L.P.(10.3)(13) | ||
| 10.20 | Warrant to purchase securities of the Company dated July 26, 2005 issued to Toucan Capital Fund II, L.P.(10.3)(14) | |
| 10.21 | Warrant to purchase securities of the Company dated September 7, 2005 issued to Toucan Capital Fund II, L.P.(10.3)(15) | |
| 10.22 | Amended Form of Warrant to purchase securities of the Company dated November 14, 2005 and April 17, 2006, as amended April 14, 2007, issued to Toucan Partners, LLC.(10.21)(23) | |
| 10.23 | Form of Warrant to purchase securities of the Company dated April 14, 2007 issued to Toucan Partners, LLC.(10.22)(23) | |
| 10.24 | Loan Agreement and 10% Convertible Promissory Note in the principal amount of $100,000 between the Company and Toucan Partners, LLC, dated April 27, 2007.(10.1)(24) | |
| 10.25 | Warrant to purchase securities of the Company issued to Toucan Partners, LLC, dated April 27, 2007. (10.2)(24) | |
| 10.26 | Form of Toucan Partners Loan Agreement and 10% Convertible Note, dated as of June 1, 2007.(10.1)(27) | |
| 10.27 | Form of Toucan Partners Warrant, dated as of June 1, 2007.(10.2)(27) | |
| 10.28 | Amended and Restated Warrant to purchase Series A Preferred Stock issued to Toucan Capital Fund II, L.P., dated as of June 1, 2007.(10.3)(27) |
| 10.29 | Warrant to purchase Series A-1 Preferred Stock issued to Toucan Capital Fund II, L.P., dated as of June 1, 2007.(10.4)(27) | |
| 10.30 | Warrant to purchase Series A-1 Preferred Stock issued to Toucan Capital Fund II, L.P., dated as of June 1, 2007.(10.5)(27) | |
| 10.31 | Northwest Biotherapeutics, Inc. $225,000 Demand Note dated June 13, 2007.(10.1)(28) | |
| 10.32 | Conversion Agreement dated June 15, 2007 and effective June 22, 2007 between the Company and Toucan Capital Fund II, LLP.(10.1)(29) | |
| 10.33 | Termination Agreement dated June 22, 2007 between the Company and Toucan Capital Fund II, LLP.(10.2)(29) | |
| 10.34 | NOMAD Agreement dated June 15, 2007 and effective June 22, 2007 between the Company and Collins Stewart Europe Limited.(10.4)(29) | |
| 10.35*** | Employment Agreement dated June 18, 2007 between Dr. Alton L. Boynton and the Company. (10.6)(29) | |
| 10.36*** | Employment Agreement dated October 1, 2007 between Anthony P. Deasey and the Company.(10.1)(30) | |
| 10.37*** | Letter of Appointment for Linda F. Powers.(10.8)(29) | |
| 10.38*** | Letter of Appointment for R. Steve Harris.(10.9)(29) | |
| 10.39 | Form of Warrant to purchase common stock of the Company, as amended.(10.27)(18) | |
| 10.40** | Northwest Biotherapeutics DCVax — Brain Services Agreement with Cognate BioServices, Inc. dated May 17, 2007.(10.1)(25) | |
| 10.41*** | 1998 Stock Option Plan.(10.15)(2) | |
| 10.42*** | 1999 Executive Stock Option Plan.(10.16)(2) | |
| 10.43*** | 2001 Stock Option Plan.(10.17)(2) | |
| 10.44*** | 2001 Nonemployee Director Stock Incentive Plan.(10.18)(2) | |
| 10.45*** | Employee Stock Purchase Plan.(10.19)(2) | |
| 10.46*** | 2007 Stock Option Plan.(10.5)(29) | |
| 10.47*** | Form of Stock Option Agreement under the 2007 Stock Option Plan.(10.2)(31) | |
| 10.48 | Lease Agreement.(10.34)(18) | |
| 10.49 | Lease Extension between the Company and the International Union of Operating Engineers Local 302, dated May 31, 2007(10.1)(26). | |
| 10.50 | Clinical Study Agreement between the Company and the Regents of the University of California dated February 14, 2006.(10.35)(18) |
| 10.51*** | Employment Agreement dated June 18, 2007, by and between Jim Johnston and the Company.(10.7)(29) | |
| 10.52*** | Form of Stock Option Agreement, dated December 31, 2007, by and between Dr. Alton L. Boynton and the Company.(99.1)(32) | |
| 10.53*** | Form of Stock Option Agreement, dated December 31, 2007, by and between Dr. Marnix Bosch and the Company.(99.2)(32) | |
| 10.54 | Sublease Agreement, dated as of March 21, 2008, between the Company and Toucan Capital Corporation.(10.1)(34) | |
| 10.55 | Loan Agreement and Promissory Note, dated May 9, 2008 between the Company and Al Rajhi Holdings WLL (4.5)(36) | |
| 10.56 | Loan Agreement and Promissory Note, dated August 19, 2008 between the Company and Toucan Partners LLC (10.1)(37) | |
| 10.57 | Loan Agreement and Promissory Note, dated October 1, 2008 between the Company and SDS Capital Group SPC, Ltd (10.2)(38) | |
| 10.58 | Warrant, dated October 1, 2008, between the Company and SDS Capital Group SPC, Ltd (10.3)(38) | |
| 10.59 | Loan Agreement and Promissory Note, dated October 21, 2008, between the Company and SDS Capital Group SPC, Ltd (10.4)(39) | |
| 10.60 | Form of Loan Agreement and Promissory Note, dated November 6, 2008, between the Company and a Group of Private Investors (10.5)(39) | |
| 10.61 | Form of Warrant, dated November 6, 2008, between the Company and SDS Capital Group SPC. Ltd and a Group of Private Investors (10.5)(39) | |
| 10.62 | Loan Agreement and Promissory Note, dated December 22, 2008, between the Company and Toucan Partners LLC (10.62)(40) | |
| 10.63 | Form of Warrant, dated December 22, 2008, between the Company and Toucan Partners LLC (10.62)(40) | |
| 10.64 | Form of Securities Purchase Agreement, dated January 16, 2009, by and among the Company and Al Rajhi Holdings (10.62)(40) | |
| 10.65 | Securities Purchase Agreement, dated March 27, 2009 by and among the Company and a Group of Equity Investors (10.62)(40) | |
| 10.66 | Form of Warrant, dated March 27,2009, between the Company and a Group of Equity Investors (10.62)(40) | |
| 10.67 | Form of Loan Agreement and Promissory Note, dated March 27 2009, between the Company and a Group of Private Lenders (10.62)(40) |
| 10.68 | Conversion Agreement effective September 28, 2009 between the Company and Toucan Partners, LLC (10.1) (41) | |
| 10.69 | Form of Loan Extension Agreement dated September 28, 2009 (10.2) (42) | |
| 10.70*** | Retention Agreement between Dr. Alton L. Boynton and the Company dated September 28, 2009 (10.3) (42) | |
| 10.71 | Purchase Agreement, dated November 14, 2011 between the Company and Four M Purchasers, LLC. | |
| 10.72 | Amendment to Purchase Agreement, dated December 6, 2011 between the Company and Four M Purchasers, LLC. | |
| 10.73 | Second Amendment to Purchase Agreement, dated January 6, 2012 between the Company and Four M Purchasers, LLC. | |
| 11 | Computation of net loss per share included within the Northwest Biotherapeutics, Inc. audited financial statements for the year ended December 31, 2010 included in this Annual Report on Form 10-K. | |
| 21 | Subsidiary of the Registrant.(21.1)(35) | |
| 23* | Consent of Peterson Sullivan LLP, Independent Registered Public Accounting Firm. |
| * | Filed or furnished herewith. |
| ** | Portions of this exhibit have been omitted and filed separately with the Securities and Exchange Commission pursuant to a request for confidential treatment. |
| *** | Denotes management contract or compensation plan or arrangement. |
| (1) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K, February 1, 2005. |
| Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Amendment No. 3 to the Registration Statement on Form S-1 (Registration No. 333-67350) on November 14, 2001. |
| (3) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Registration Statement on Form 8-A on July 8, 2002. |
| (4) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Form 10-K on May 14, 2004. |
| (5) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on November 14, 2005. |
| (6) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on March 31, 2006. |
| (7) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on August 6, 2004. |
| (8) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on October 28, 2004. |
| (9) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on November 16, 2004. |
| (10) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on December 30, 2004. |
| (11) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Annual Report on Form 10-K on April 15, 2005. |
| (12) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8- K on May 18, 2005. |
| (13) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on June 21, 2005. |
| (14) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on August 1, 2005. |
| (15) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on September 9, 2005. |
| (16) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on November 14, 2003. |
| (17) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on August 5, 2005. |
| (18) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 10-K on April 18, 2006. |
| (19) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on April 26, 2006. |
| (20) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Form 10-K/A on June 30, 2006. |
| (21) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant Registration Statement on Form S-1 (File No. 33-134320) on May 19, 2006. |
| Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Amendment No. 1 to the Registration Statement on Form S-1(File No. 333-134320) on July 17, 2006. |
| (23) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Form 10-K on April 17, 2007. |
| (24) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on May 3, 2007. |
| (25) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on May 21, 2007. |
| (26) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on June 4, 2007. |
| (27) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on June 7, 2007. |
| (28) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on June 18, 2007. |
| (29) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on June 22, 2007. |
| (30) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on October 2, 2007. |
| (31) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Registration Statement on Form S-8 on November 21, 2007. |
| (32) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on January 3, 2008. |
| (33) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Post-Effective Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 on January 28, 2008. |
| (34) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on March 24, 2008. |
| (35) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Post-Effective Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 on December 17, 2007. |
| (36) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on May 15, 2008, 2008. |
| (37) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on August 19, 2008. |
| (38) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on October 6, 2008. |
| (39) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Current Report on Form 8-K on November 11, 2008. |
| (40) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Form 10-K on April 15, 2008 |
| (41) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on August 14, 2008. |
| (42) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on November 23, 2009. |
| (43) | Incorporated by reference to the exhibit shown in the preceding parentheses filed with the Registrant’s Quarterly Report on Form 10-Q on May 21, 2010. |
| NORTHWEST BIOTHERAPEUTICS, INC. | ||
| By: | /s/ Linda Powers | |
| Name: Linda Powers | ||
| Title: Chief Executive Officer and Chairperson | ||
| (Principal Executive Officer, Principal Financial Officers andPrincipal Accounting Officer) | ||
| Signature | Title | Date | ||
| /s/ Linda Powers | Director, Chairperson and Chief Executive Officer | February 2, 2012. | ||
| Linda Powers | ||||
| /s/ Alton Boynton | Chief Scientific Officer, Director | February 2, 2012 | ||
| Alton Boynton | ||||
| /s/ Robert Farmer | Director | February 2, 2012 | ||
| Robert Farmer |