UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of April, 2013
Commission File Number
Novogen Limited
(Translation of registrant’s name into English)
1-7 Waterloo Road, Macquarie Park, NSW, Australia
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-Fx Form 40-F¨
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1):¨
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ¨
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark if the registrant by furnishing the information contained in this form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934. Yes¨ Nox
If “yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b)
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Novogen Limited(Registrant)
Andrew Bursill
Andrew Bursill
Company Secretary
Date 8 April 2013

A new company with: a new technology platform for cancer drug development a
new management team a
new Board

Disclaimer
This presentation is confidential and is not an offer or a recommendation or intended to influence you in any way in making an investment decision or take any other action.
In particular, Novogen:
a. |
| does not warrant the accuracy or completeness of the information including any forward looking statements, if any, in this presentation; |
b. |
| does not accept responsibility for any interpretation or conclusion you may form as a result of this presentation; |
c. |
| is not liable for any loss or damage arising from any error, inaccuracy, incompleteness in this presentation. |
This is not financial product advice and any advice (if any) given in this Information is general advice only. You are expected to rely on your own advice and enquiries.
2 |
|
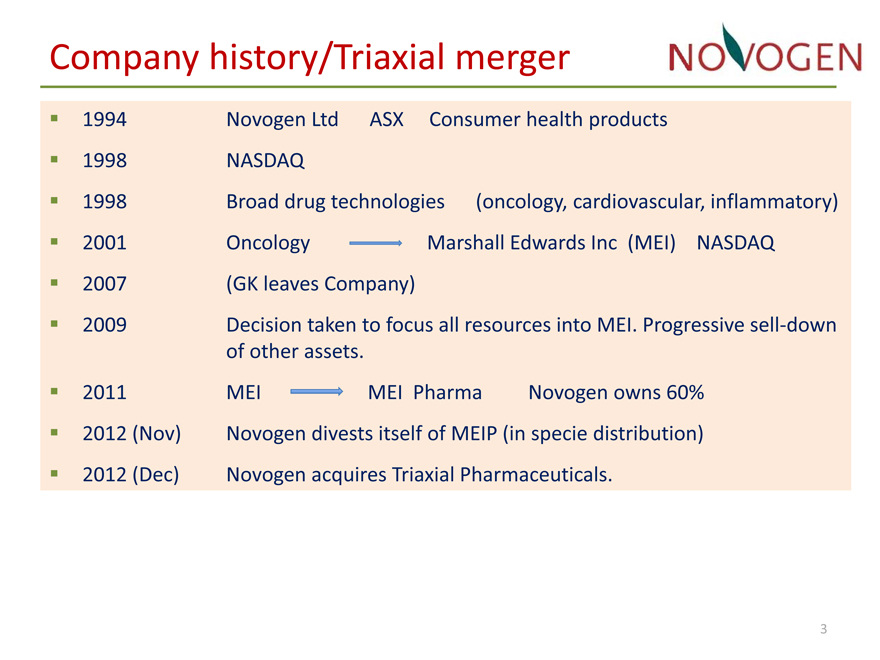
Company history/Triaxial merger
? 1994 Novogen Ltd ASX Consumer health products
? 1998 NASDAQ
? 1998 Broad drug technologies(oncology, cardiovascular, inflammatory)
? 2001 Oncology Marshall Edwards Inc (MEI) NASDAQ
? 2007(GK leaves Company)
? 2009 Decision taken to focus all resources into MEI. Progressive sell?down
of other assets.
? 2011 MEI MEI Pharma Novogen owns 60%
? 2012 (Nov) Novogen divests itself of MEIP (in specie distribution)
? 2012 (Dec) Novogen acquires Triaxial Pharmaceuticals.
3 |
|

Gen 1 technology platform
• Simple benzopyran molecules
• Unique anti?cancer action
• pan cancer effect
• tumour?specific
• unaffected by multi?drug resistance mechanisms
• restore chemo?sensitivity
• modest anti?cancer stem cell activity
• 4 lead drug candidates
• Tested in > 800 patients
• Evidence of efficacy in late?stage chemo?refractory prostate and ovarian cancers
4 |
|

Gen 2 technology platform
Need to:
increase bio?availability
increase potency against both cancer cells and cancer stem cells
improve manufacturing efficiencies.
Challenge:
new technology required to create a new family of more complex benzopyrans
need to overcome design and manufacturing limitations.
5 |
|
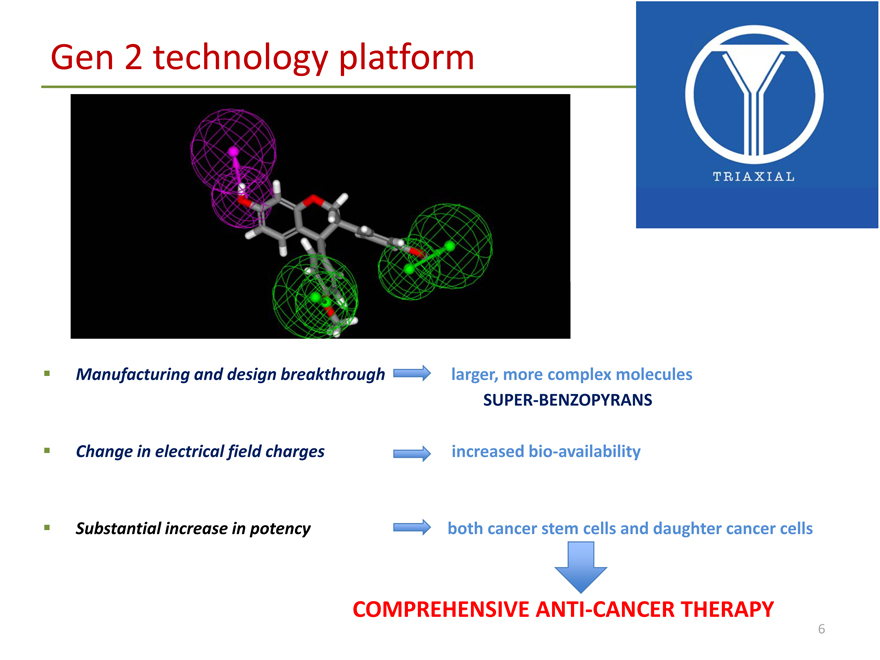
Gen 2 technology platform
Manufacturing and design breakthrough larger, more complex molecules
SUPER?BENZOPYRANS
Change in electrical field charges increased bio?availability
Substantial increase in potency both cancer stem cells and daughter cancer cells
COMPREHENSIVE ANTI?CANCER THERAPY
6 |
|
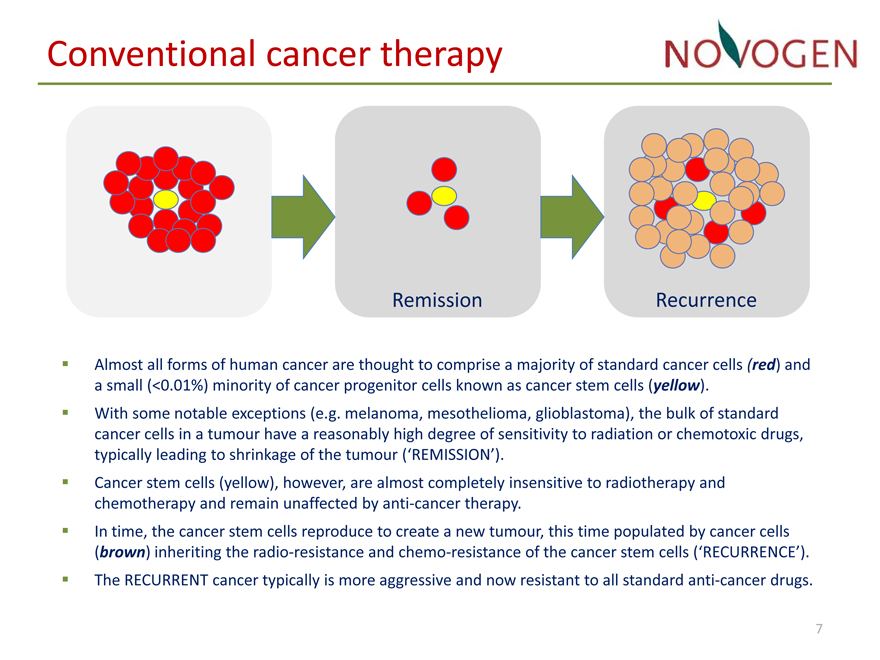
Conventional cancer therapy
Almost all forms of human cancer are thought to comprise a majority of standard cancer cells (red) and a small (<0.01%) minority of cancer progenitor cells known as cancer stem cells (yellow).
• With some notable exceptions (e.g. melanoma, mesothelioma, glioblastoma), the bulk of standard cancer cells in a tumour have a reasonably high degree of sensitivity to radiation or chemotoxic drugs, typically leading to shrinkage of the tumour (‘REMISSION’).
• Cancer stem cells (yellow), however, are almost completely insensitive to radiotherapy and chemotherapy and remain unaffected by anti?cancer therapy.
• In time, the cancer stem cells reproduce to create a new tumour, this time populated by cancer cells (brown) inheriting the radio?resistance and chemo?resistance of the cancer stem cells (‘RECURRENCE’).
• The RECURRENT cancer typically is more aggressive and now resistant to all standard anti?cancer drugs.
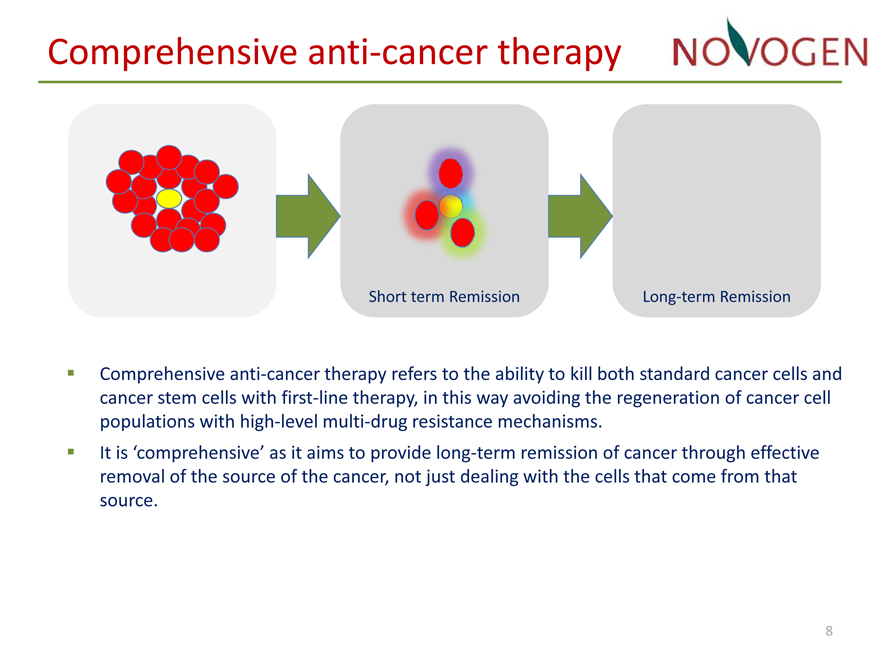
Comprehensive anti?cancer therapy
Short term Remission Long?term Remission
??Comprehensive anti?cancer therapy refers to the ability to kill both standard cancer cells and cancer stem cells with first?line therapy, in this way avoiding the regeneration of cancer cell populations with high?level multi?drug resistance mechanisms.
??It is ‘comprehensive’ as it aims to provide long?term remission of cancer through effective removal of the source of the cancer, not just dealing with the cells that come from that source.
8 |
|
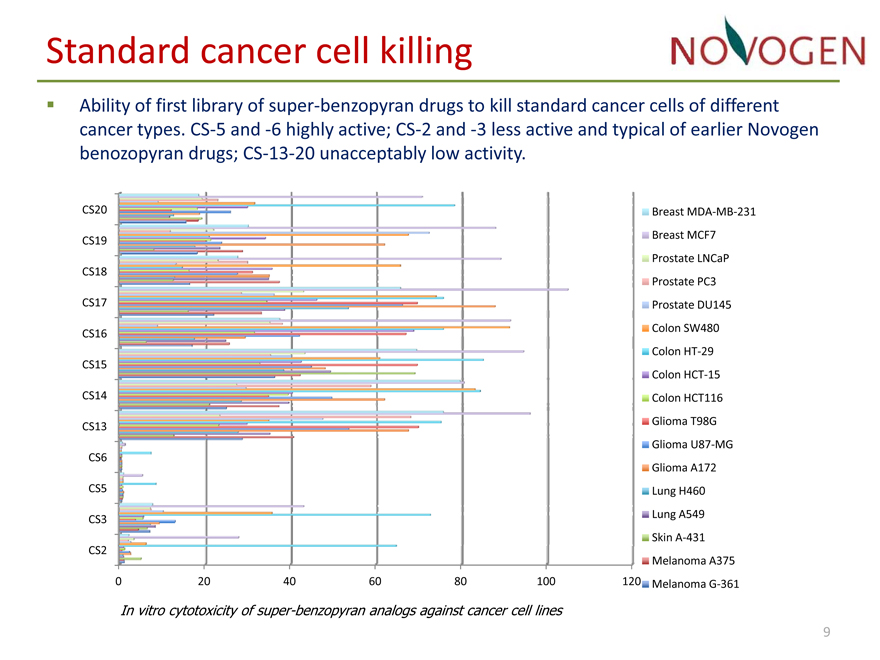
Standard cancer cell killing
• Ability of first library of super?benzopyran drugs to kill standard cancer cells of different cancer types. CS?5 and ?6 highly active; CS?2 and ?3 less active and typical of earlier Novogen benozopyran drugs; CS?13?20 unacceptably low activity.
CS20 Breast MDA?MB?231
CS19 Breast MCF7
Prostate LNCaP
CS18
Prostate PC3
CS17 Prostate DU145
CS16 Colon SW480
Colon HT?29
CS15
Colon HCT?15
CS14 Colon HCT116
CS13 Glioma T98G
Glioma U87?MG
CS6
Glioma A172
CS5 Lung H460
CS3 Lung A549
Skin A?431
CS2
Melanoma A375
0 20 40 60 80 100 120 Melanoma G?361
In vitro cytotoxicity of super-benzopyran analogs against cancer cell lines
9
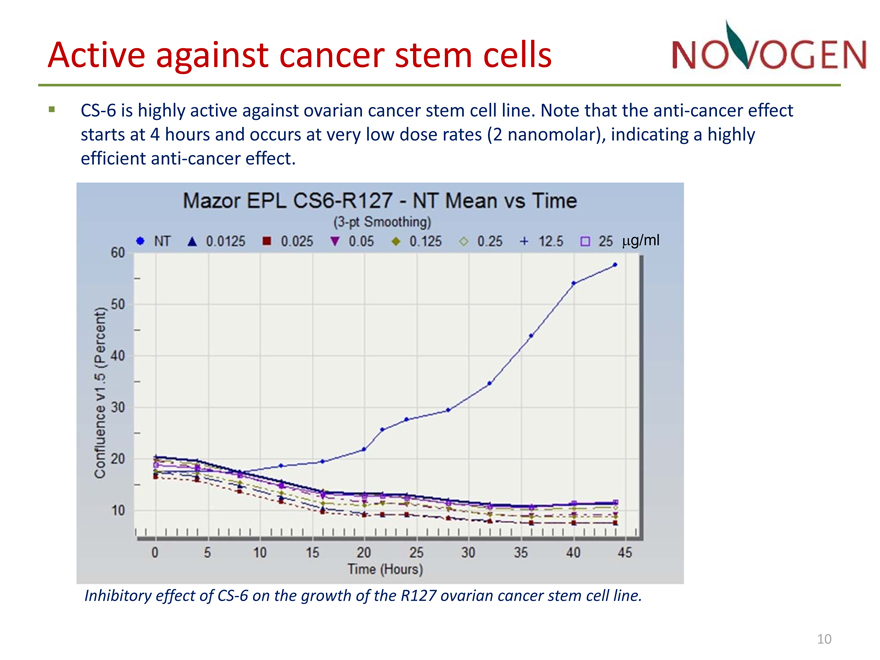
Active against cancer stem cells
• CS?6 is highly active against ovarian cancer stem cell line. Note that the anti?cancer effect starts at 4 hours and occurs at very low dose rates (2 nanomolar), indicating a highly efficient anti?cancer effect.
?g/mlInhibitory effect of CS?6 on the growth of the R127 ovarian cancer stem cell line.
10
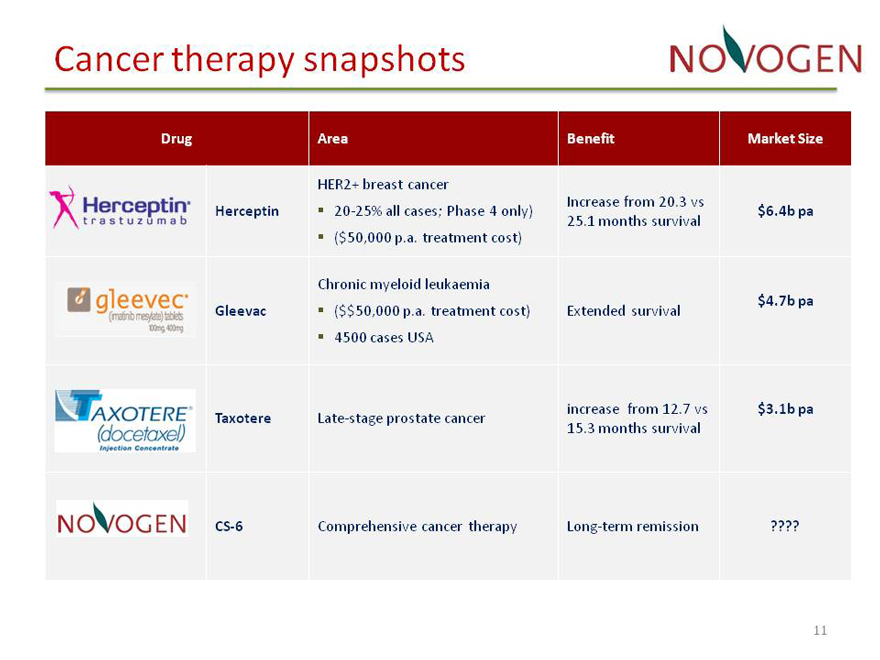
Cancer therapy snapshots
CS?6 Comprehensive cancer therapy Long?term remission
Drug Area Benefit Market Size
HER2+ breast cancer
Increase from 20.3 vs
Herceptin ??20?25% all cases; Phase 4 only) $ 6.4b pa
25.1 months survival
??($50,000 p.a. treatment cost)
Chronic myeloid leukaemia
Gleevac $$??( 50,000 p.a. treatment cost) Extended survival $ 4.7b pa
??4500 cases USA
increase from 12.7 vs $ 3.1b pa
Taxotere Late?stage prostate cancer
15.3 months survival
CS?6 Comprehensive cancer therapy Long?term remission ????
11

Strategy
Company Strategy: To develop the first comprehensive cancer therapy capable of killing both standard tumour cells and cancer stem cells
?CS?6 is Novogen’s first super?benzopyran drug candidate. CS?6 is:
?highly active in the laboratory against all forms of human cancer so far tested; ?most active against glioblastoma multiforme cells (main form of brain cancer); and ?highly active against (ovarian) cancer stem cells (nanomolar levels).
?Novogen aims to bring CS?6 to market as a treatment for late?stage brain cancer and late?stage ovarian cancer.
?Ability to destroy both cancer stem cells and their chemo?resistant daughter cells is a game changer as
no other drug (currently approved or in development ) knocks out both the highly resistant cancer stem cells and
their daughter cancer cells in a selective manner with acceptable side?effects.
12
Signature Page to Revolving Credit Agreement
Signature Page to Revolving Credit Agreement
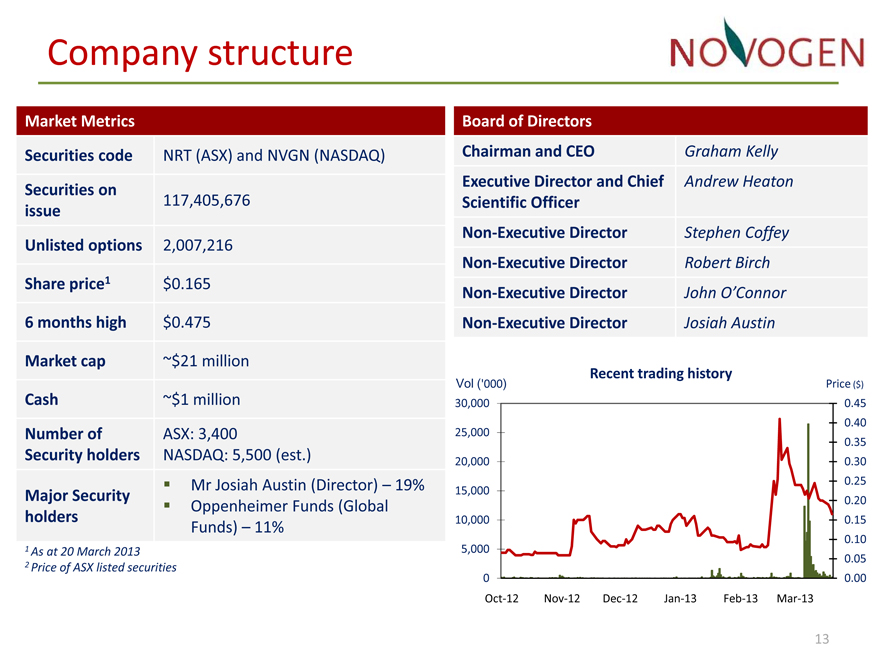
Market Metrics
Securities code NRT (ASX) and NVGN (NASDAQ)
Securities on
117,405,676
issue
Unlisted options 2,007,216
Share price1 $0.165
6 months high $0.475
Market cap ~$21 million
Cash ~$1 million
Number of ASX: 3,400
Security holders NASDAQ: 5,500 (est.)
Mr Josiah Austin (Director) – 19%
Major Security Oppenheimer Funds (Global
holders
Funds) – 11%
1 As at 20 March 2013
2 Price of ASX listed securities
Board of Directors
Chairman and CEO Graham Kelly
Executive Director and Chief Andrew Heaton
Scientific Officer
Non Executive Director Stephen Coffey
Non Executive Director Robert Birch
Non Executive Director John O’Connor
Non Executive Director Josiah Austin
Recent trading history
Vol (‘000) Price ($) 30,000 0.45
0.40 25,000
0.35
20,000 0.30 0.25
15,000
0.20 10,000 0.15 0.10
5,000
0.05
0 0.00
Oct 12 Nov12 Dec12 Jan13 Feb13 Mar13

Development milestones
Design and make super?benzopyran molecules
Achieve manufacturing process producing clean product Design analogs with potent anti?cancer activity Design analogs with enhanced bio?availability Design analogs with potent anti?cancer stem cell activity
Confirm acceptable half?life of drug
Confirm ability to inhibit tumour xeograft growth Determine chemo?sensitising ability Define toxicology Manufacture batch for clinical use
Phase 1a Study
Phase 1b Study
Clinical Pre-Clinical Fundamentals
14

Funding
Novogen is currently seeking to raise up Use of funds (3 year program) to $10 million to fund a 3 year
development plan. Corporate costs $3.0 million (approx.)
?The purpose of the capital raising is to
enable Novogen to: Pre?clinical drug program – 3 drugs $4.0 million
?Take CS?6 through its pre?clinical program and into Phase 1a and Phase 1b clinical CS?6 – Phase 1 (glioma) $1.5 million studies in patients with glioblastoma CS?6 – Phase 1 (ovarian) $1.5 million multiforme;
?Generate the data to enable the Novogen Total $10.0 million to apply to the FDA for IND status and Breakthrough Therapy Designation;
?Investigate the potential clinical application of CS?6 in early?and late?stage ovarian cancer;
??Continue with an analog program with the objective of identifying an additional 2 lead drug candidate to be taken through a pre?clinical drug program; and
??Working capital.
15

Contact details
Company Novogen Limited ABN: 37 063 259 754
Stock code NRT (ASX), NVGN (NASDAQ)
Contact For investment information: Professor Graham Kelly CEO and Chairman Graham.Kelly@novogen.com
Address Level 1, 1?7 Waterloo Road North Ryde, NSW 2113, Australia
Website http://www.novogen.com/
16