UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of April, 2013
Commission File Number
Novogen Limited
(Translation of registrant’s name into English)
1-7 Waterloo Road, Macquarie Park, NSW, Australia
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F x Form 40-F ¨
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ¨
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ¨
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark if the registrant by furnishing the information contained in this form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934. Yes ¨ No x
If “yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b)
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Novogen Limited(Registrant)
Andrew Bursill |
Andrew Bursill Company Secretary Date 19 April 2013 |

ASX RELEASE
19 April 2013
Chairman’s address and proxy votes
General meeting of Novogen Limited at 1pm today
Computershare Australia offices
Level 4, 60 Carrington Street, Sydney
Chairman’s address and proxy votes for the purposes of the general meeting of Novogen Limited (ASX: NRT) to be held at 1pm today at the offices of Computershare Australia offices at level 4, 60 Carrington Street, Sydney are attached.
About Novogen
Novogen Ltd is a public Australian biotechnology company whose shares trade on both the Australian Stock Exchange (symbol ‘NRT’) and NASDAQ (symbol ‘NVGN’). The Company is based in Sydney, Australia and is focused on the development of a family of novel anti-cancer drugs based on super-benzopyran and ‘stealth’ drug technologies. The Company’s inaugural drug candidate is CS-6. Website:www.novogen.com
About CS-6
CS-6 belongs to a new class of drug candidates known (structurally) as super-benzopyrans displaying potent anti-cancer activity and demonstrating increased bio-availability to cancer cells (‘stealth’ technology). CS-6 shows broad anti- proliferative and cytotoxic activity against human cancer cells, with particular activity against human glioblastoma cells. CS-6 also has been designed deliberately to meet the major known criteria for crossing the blood-brain barrier, and for that reason is being developed as a first-line for the treatment of glioblastoma multiforme, the main form of primary brain cancer.
Safe harbour statement
Certain statements made in this document and material referred to in this document that use the words “estimate”, “project”, “intend”, “expect”, “believe” and similar expressions are intended to identify forward looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (U.S.). These forward looking statements involve known and unknown risks and uncertainties which could cause the actual results, performance or achievements of the company to be materially different from those which may be expressed or implied by these statements. These statements are based on our management’s current expectations and are subject to a number of uncertainties that could change the results described in the forward-looking statements.
Investors should be aware that there are no assurances that results will not differ from those projected.
1

NOVOGEN
NOVOGEN
1

NOVOGEN
General Meeting of Shareholders
19 April 2013
2 |

General Meeting 19 April 2013 NOVOGEN
Resolution 1. Approval to raise capital
Recurrent costs $1.4M (approx.)
Salaries/directors’ fees 40%
Legal/accounting/audit 60%
Patents
Office
ASX/NASDAQ
R & D costs
Pre-clinical program + Phase 1a
$1.2M
Analog program
$0.5M
Other
$1.0M
3

General Meeting 19 April 2013 NOVOGEN
Resolution 1. Approval to raise capital
Approval under Listing Rule 7.3
Maximum $10M
Maximum 1:2 options
Maximum discount of 20% of 5-day VWAP
Within 3 months of GM
4
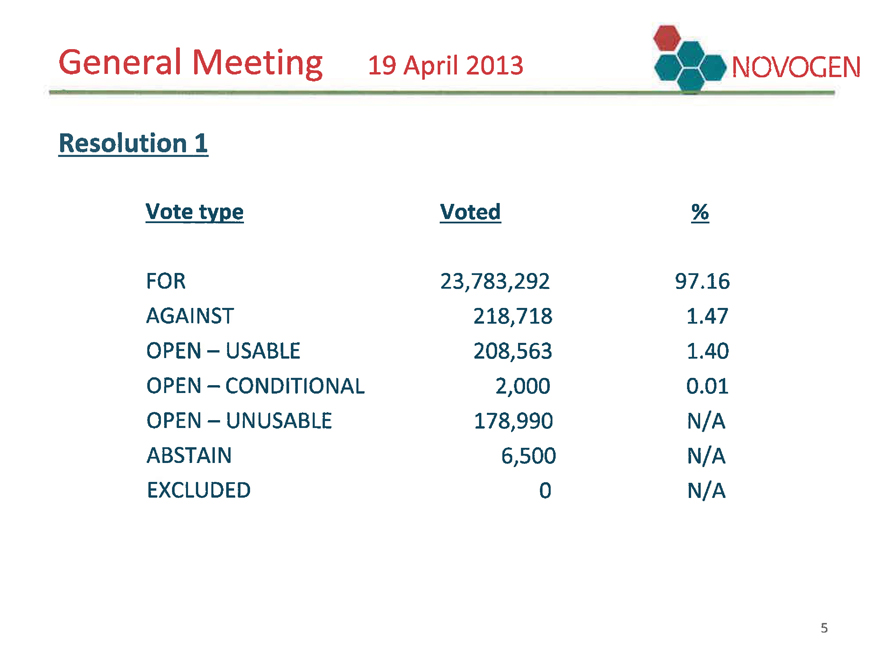
General Meeting 19 April 2013 NOVOGEN
Resolution 1
Vote type Voted %
FOR 23,783,292 97.16
AGAINST 218,718 1.47
OPEN - USABLE 208,563 1.40
OPEN - CONDITIONAL 2,000 0.01
OPEN - UNUSABLE 178,990 N/A
ABSTAIN 6,500 N/A
EXCLUDED 0 N/A 5

General Meeting 19 April 2013 NOVOGEN
Resolution 2 Issue of shares
Allotment of shares:
•250,000 B. Palmer
•1,429,782 Glycotex Inc
•15,279,782 Triaxial acquisition
6
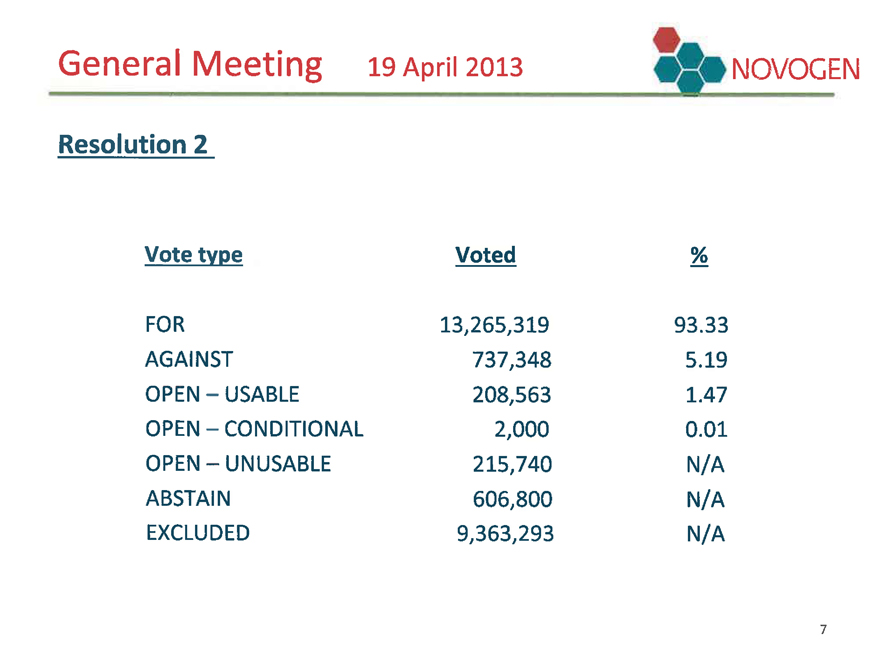
General Meeting 19 April 2013 NOVOGEN
Resolution 2
Vote type Voted %
FOR 13,265,319 93.33
AGAINST 737,348 5.19
OPEN - USABLE 208,563 1.47
OPEN - CONDITIONAL 2,000 0.01
OPEN - UNUSABLE 215,740 N/A
ABSTAIN 606,800 N/A
EXCLUDED 9,363,293 N/A
7 |

General Meeting 19 April 2013 NOVOGEN
Resolution 3 Issue of shares to Phytose Corp. Pty Ltd
1,800,000 shares as part consideration of First Tranche of shares per acquisition of Triaxial Pharmaceuticals Pty Ltd
8
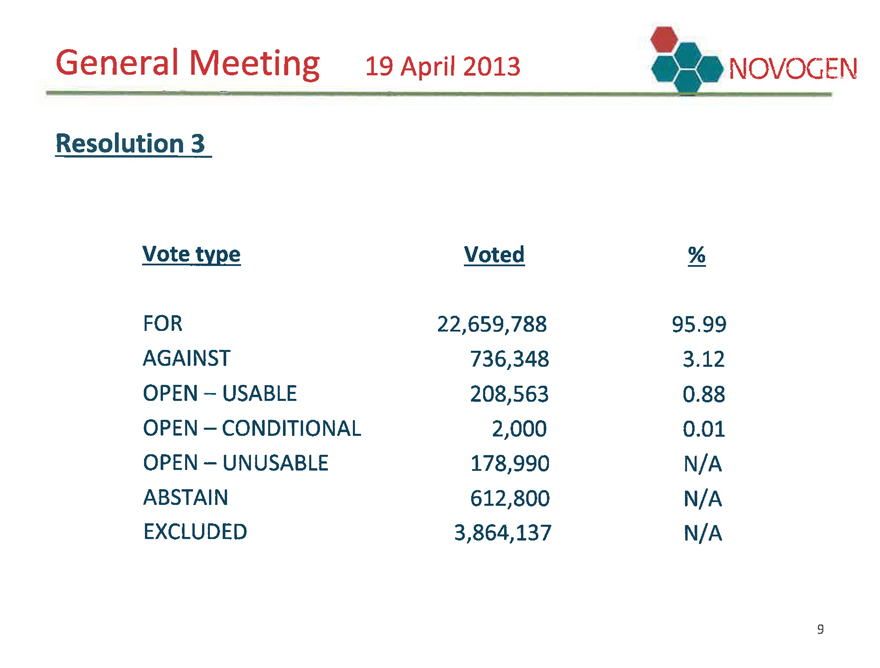
General Meeting 19 April 2013 NOVOGEN
Resolution 3
Vote type Voted %
FOR 22,659,788 95.99
AGAINST 736,348 3.12
OPEN – USABLE 208,563 0.88
OPEN – CONDITIONAL 2,000 0.01
OPEN – UNUSABLE 178,990 N/A
ABSTAIN 612,800 N/A
EXCLUDED 3,864,137 N/A
9

General Meeting 19 April 2013 NOVOGEN
Resolution 4 Issue of shares for Convertible Notes
• Acquisition of Triaxial Pharmaceuticals Pty Ltd for $1.88M
• Purchase funded by Convertible Notes from 6 shareholders
• All shareholders have elected to receive shares
• $1.5M of Notes remaining:
(a) Completion of Phase la study $400K 16 M shares
(b) IND grant $500K 20 M shares
(c) Completion of Phase 2 or $600K 24 M shares Breakthrough Therapy status
10
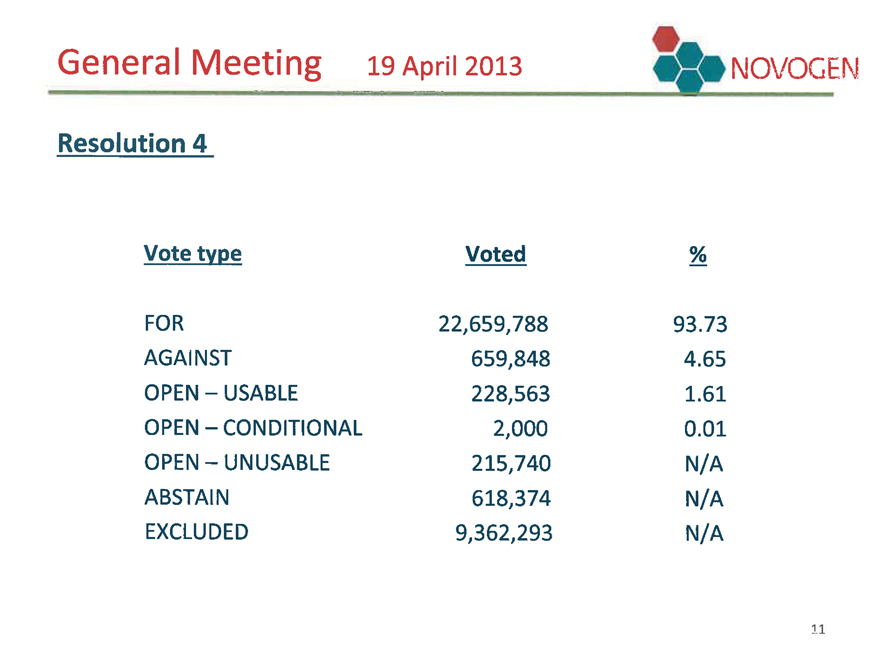
General Meeting 19 April 2013 NOVOGEN
Resolution 4
Vote type Voted %
FOR 22,659,788 93.73
AGAINST 659,848 4.65
OPEN – USABLE 228,563 1.61
OPEN – CONDITIONAL 2,000 0.01
OPEN – UNUSABLE 215,740 N/A
ABSTAIN 618,374 N/A
EXCLUDED 9,362,293 N/A
11

NOVOGEN
NOVOGEN
12

NOVOGEN
Our Mission
To bring the ability to conquer cancer into the reach of the common man
13

NOVOGEN
Comprehensive Cancer Therapy
Therapy targeting both regular cancer cells and their progenitor cells - cancer stem cells
14
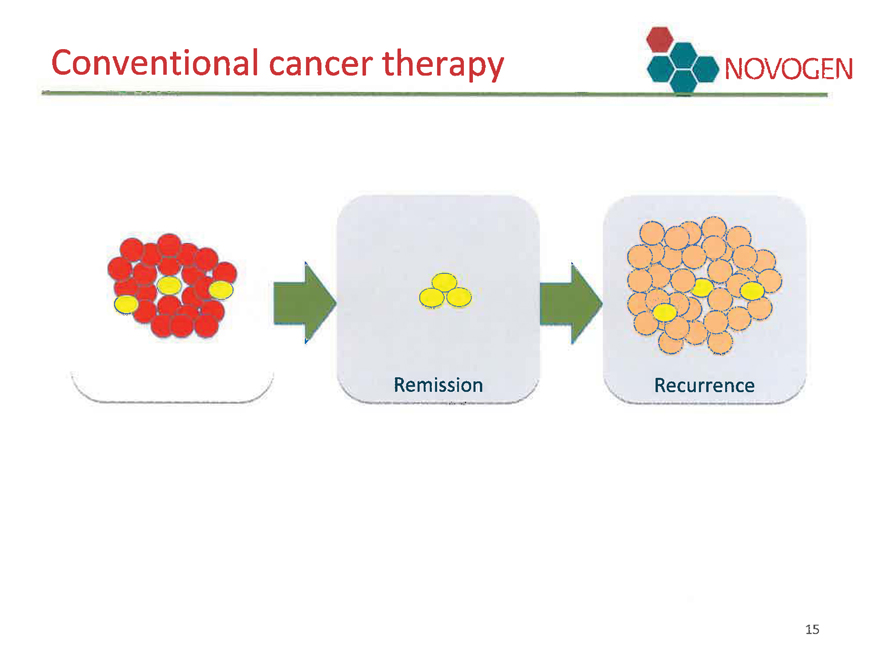
Conventional cancer therapy NOVOGEN
Remission
Recurrence
15
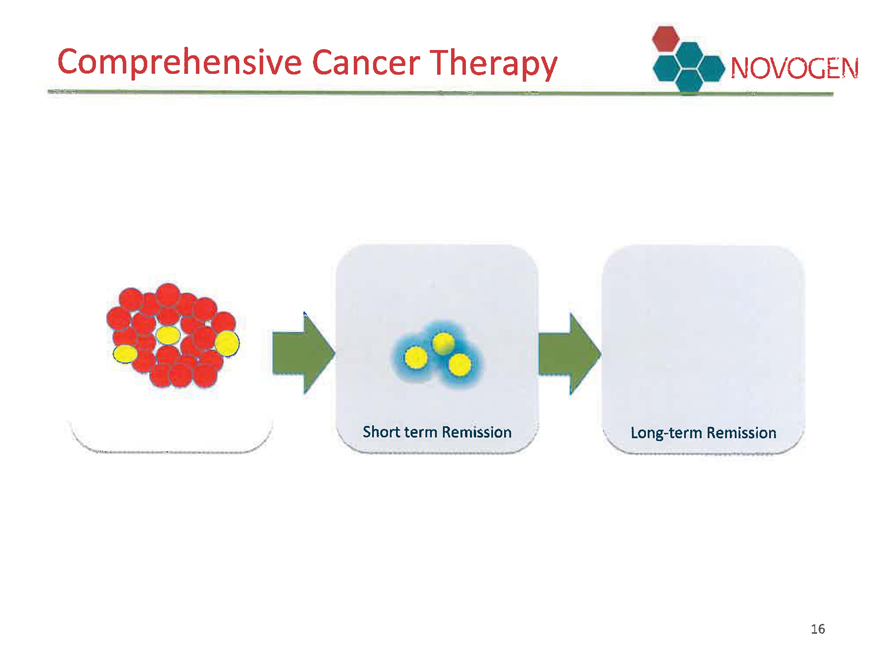
Comprehensive Cancer Therapy NOVOGEN
Short term Remission
Long-term Remission
16

NOVOGEN
NOVOGEN
A new company with:
a new technology platform for cancer drug development
a new management team
a new Board

Disclaimer NOVOGEN
This presentation is confidential and is not an offer or a recommendation or intended to influence you in any way in making an investment decision or take any other action.
In particular, Novogen:
a.does not warrant the accuracy or completeness of the information including any forward looking statements, if any, in this presentation;
b.does not accept responsibility for any interpretation or conclusion you may form as a result of this presentation;
c.is not liable for any loss or damage arising from any error, inaccuracy, incompleteness in this presentation.
This is not financial product advice and any advice (if any) given in this Information is general advice only. You are expected to rely on your own advice and enquiries.
18

Company history NOVOGEN
1994 Norvet Ltd formed and listed (ASX). (‘NRT’). Veterinary and consumer health products.
1997 Veterinary business sold. Focus on isoflavone drugs and OTC business. Name change to Novogen.
1998 Lists on NASDAQ (‘NVGN’).
1998 - 2008 Active drug R&D programs. Broad drug technologies (oncology, cardiovascular, inflammatory)
2001 Oncology licensed into fully-owned subsidiary, Marshall Edwards Inc. Lists on NASDAQ.
2008 Phase 3 pivotal trial of phenoxodiol fails. Decision to focus on stronger drugs – NV-128, NV-143, NV-344.
2009 Decision taken to focus all resources into MEI. Progressive sell-down of other assets. All isoflavonoid drug IP transferred into MEI.
2010 All R&D functions stopped.
2011 MEI changes name to MEI Pharma (MEIP). Novogen owns 60%.
2012 (Nov) Novogen divests itself of MEIP (in specie distribution)
19

Triaxial history
Triaxial Pharmaceuticals Pty Ltd formed by 3 ex-Novogen scientists: Andrew Heaton, David Brown and Graham Kelly.
TRIAXIAL
GK - Founder of Novogen, variously CEO and Chairman. Responsible for discovery of isoflavonoid (benzopyran) drug opportunity. Resigned in 2007.
AH - Senior Chemist, Novogen. Responsible for the design and manufacture of benzopyran drugs. Resigned in 2009.
DB – Senior Biologist, Novogen. Responsible for pre-clinical drug programs. Resigned in 2010.
Aim: To create more powerful, more bio-available versions of benzopyran drugs.
2011 Design breakthrough achieved. Ability to manufacture super-benzopyrans confirmed.
2012 Capital-raising through private placement.
2012 First library of super-benzopyrans made and potency of CS-6 confirmed.
2012 Merger with Novogen (7 Dec 2012).
20

Gen 1 technology platform NOVOGEN
Simple benzopyran molecules
Unique anti-cancer action
pan cancer effect
tumour-specific
unaffected by multi-drug resistance mechanisms
restore chemo-sensitivity
modest anti-cancer stem cell activity
4 lead drug candidates
Tested in > 800 patients
Evidence of efficacy in late-stage chemo-refractory prostate and ovarian cancers
21

Gen 2 technology platform NOVOGEN
Need to:
increase bio-availability
increase potency against both cancer cells and cancer stem cells
improve manufacturing efficiencies.
Challenge:
new technology required to create a new family of more complex benzopyrans
need to overcome design and manufacturing limitations.
22

Gen 2 technology platform NOVOGEN
Manufacturing and design breakthrough larger, more complex molecules
SUPER-BENZOPYRANS
Change in electrical field charges increased bio-availability
Substantial increase in potency both cancer stem cells and regular cancer cells
COMPREHENSIVE ANTI-CANCER THERAPY
23
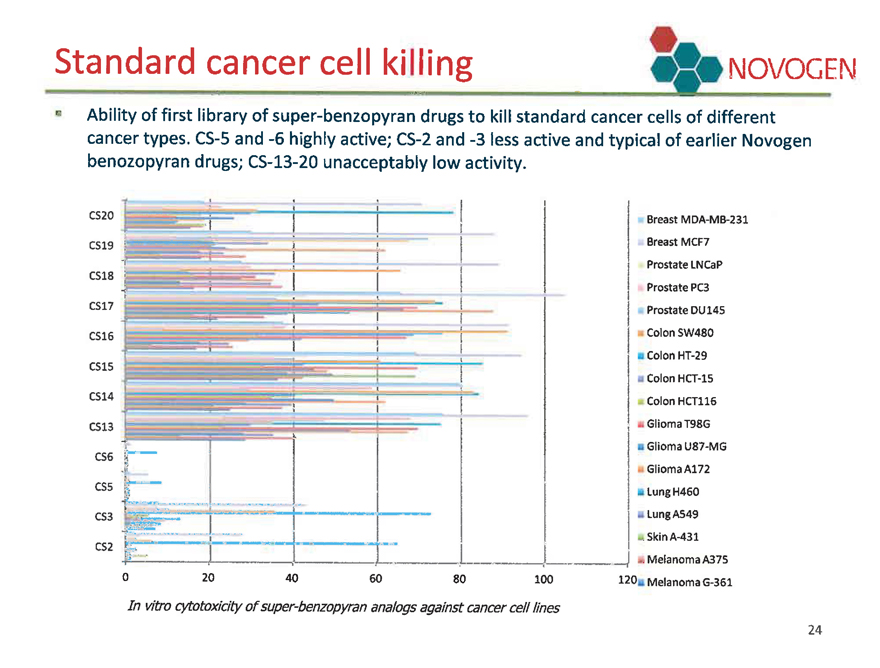
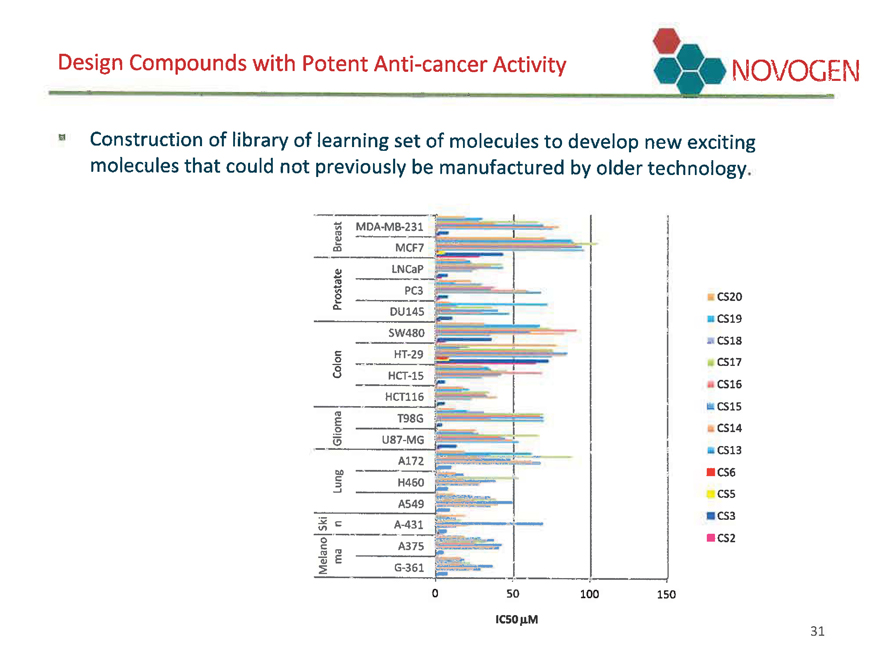
Standard cancer cell killing NOVOGEN
Ability of first library of super-benzopyran drugs to kill standard cancer cells of different cancer types. CS-5 and -6 highly active; CS-2 and -3 less active and typical of earlier Novogen benozopyran drugs; CS-13-20 unacceptably low activity.
CS20
CS19
CS18
CS17
CS16
CS15
CS14
CS13
CS6
CS5
CS3
CS2
Breast MDA-MB-231
Breast MCF7
Prostate LNCaP
Prostate PC3
Prostate DU145
Colon SW480
Colon HT-29
Colon HCT-15
Colon HCT116
Glioma T98G
Glioma U87-MG
Glioma A172
Lung H460
Lung A549
Skin A-431
Melanoma A375
Melanoma G-361
0 20 40 60 80 100 120
In vitro cytotoxicity of super-benzopyran analogs against cancer cell lines
24
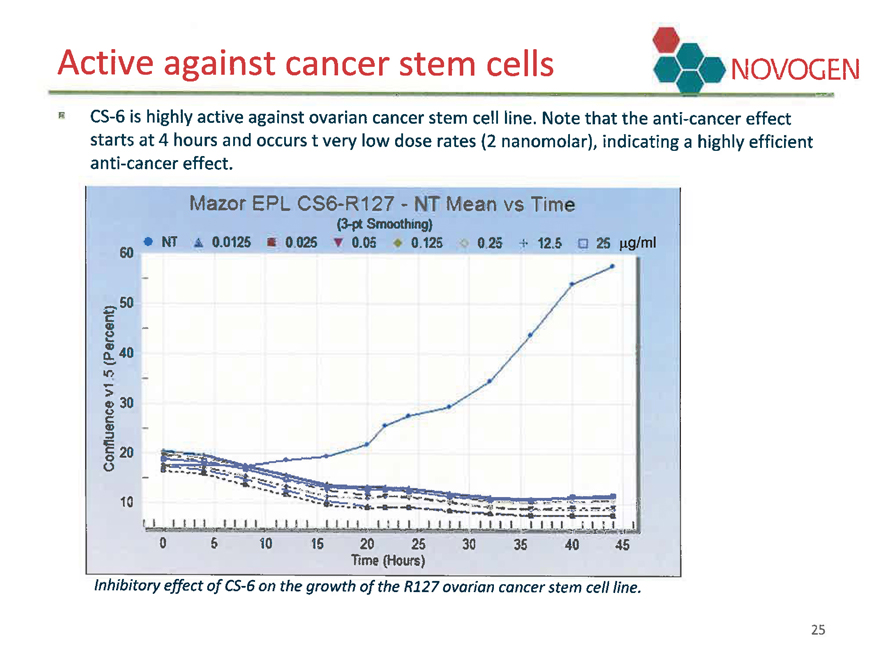
Active against cancer stem cells
NOVOGEN
CS-6 is highly active against ovarian cancer stem cell line. Note that the anti-cancer effect starts at 4 hours and occurs t very low dose rates (2 nanomolar), indicating a highly efficient anti-cancer effect.
Mazor EPL CS6-R127 - NT Mean vs Time
(3-pt Smoothing)
• NT 0.0125 0.025 0.05 0.125 0.25 + 12.5¨ 25 µg/ml
60 50 40 30 20 10
0 5 10 15 20 25 30 35 40 45 Time (Hours)
Inhibitory effect of CS-6 on the growth of the R127 ovarian cancer stem cell line.
25
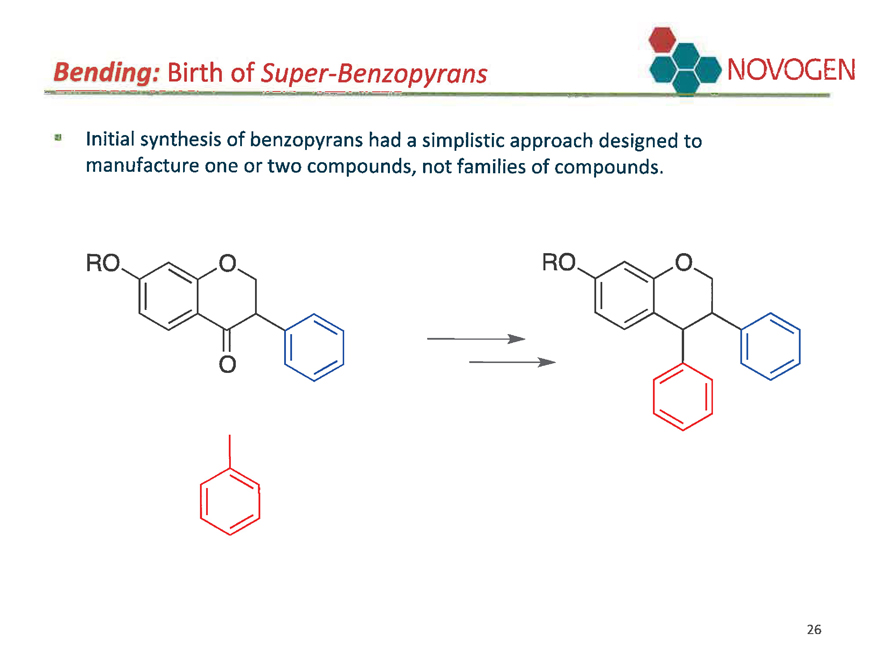
Bending: Birth of Super-Benzopyrans NOVOGEN
Initial synthesis of benzopyrans had a simplistic approach designed to manufacture one or two compounds, not families of compounds.
RO O
O
RO O
26
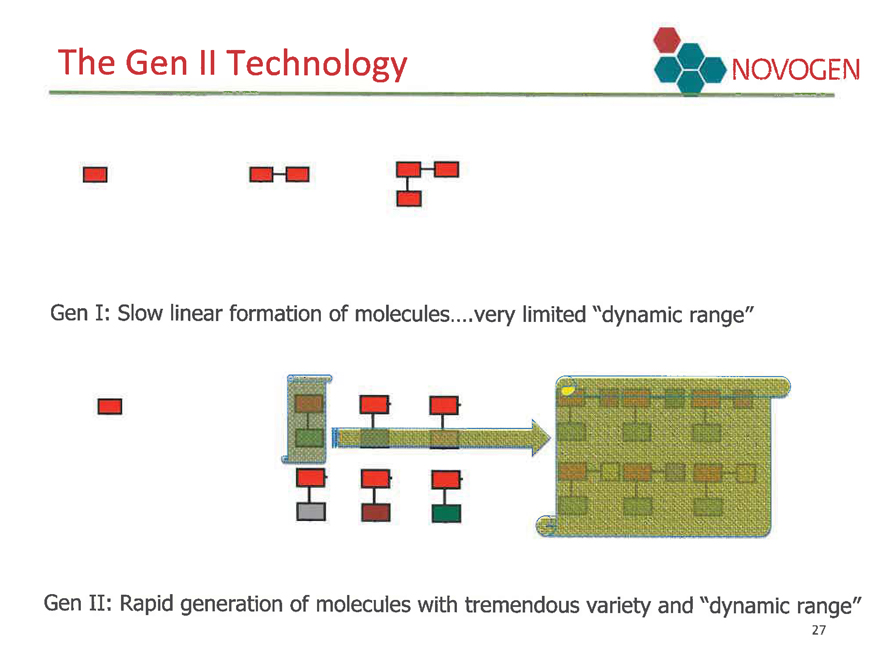
The Gen II Technology NOVOGEN
Gen I: Slow linear formation of molecules.…very limited “dynamic range”
Gen II: Rapid generation of molecules with tremendous variety and “dynamic range”
27
The Bend Technology
NOVOGEN
28

Gen II Technology NOVOGEN
Far more efficient- now use 4 manufacturing steps not 8+
Each manufacturing step can add a far wider range of groups giving greater molecular diversity, thereby maximizing the possibility of getting a hit
Range of groups added is focused we do not have a shotgun approach. Experience has taught us what where we should change “fields” in the molecule and how/what degree of “bend” is most likely to work best.
Does it work.…
29
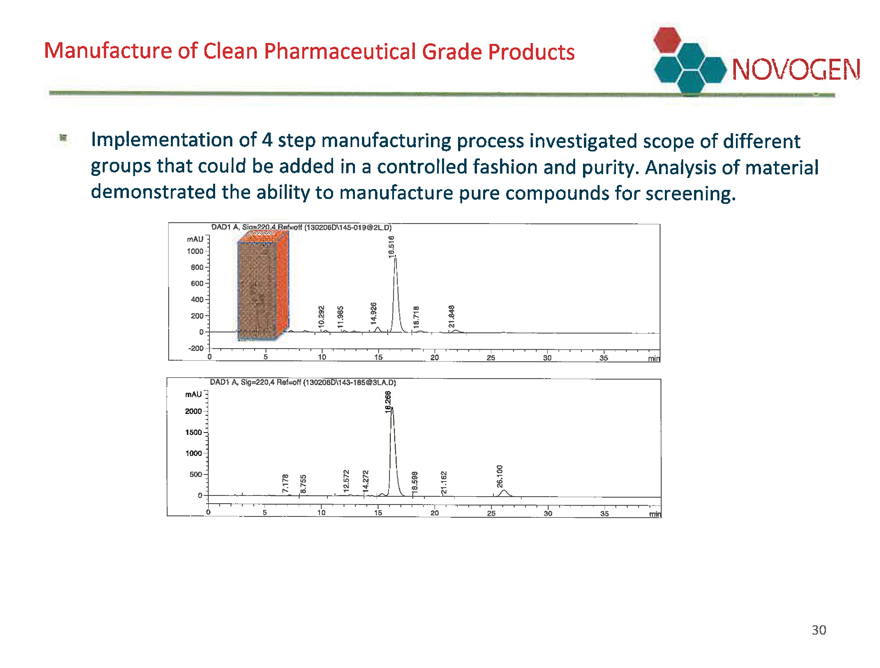
Manufacture of Clean Pharmaceutical Grade Products NOVOGEN
Implementation of 4 step manufacturing process investigated scope of different groups that could be added in a controlled fashion and purity. Analysis of material demonstrated the ability to manufacture pure compounds for screening.
DAD1 A, Sig=220.4 Ref=off (130206D\145-019@2LD)
mAU 1000 800 600 400 200 0 -200
0 5 10 15 20 25 30 35 min
10.292 11.985 14.926 16.516 18.718 21.848
DAD1 A, Sig=220.4 Ref=off (130206D\145-185@3LAD)
mAU 2000 1500 1000 500 0
0 5 10 15 20 25 30 35 min
7.178 8.755 12.572 14.272 16.266 18.598 21.162 26.100
30
Design Compounds with Potent Anti-cancer Activity NOVOGEN
Construction of library of learning set of molecules to develop new exciting molecules that could not previously be manufactured by older technology.
Breast
MDA-MB-231 MCF7
Prostate
LNCap PC3 DU145
Colon
SW480 HT-29 HCT-15 HCT116
Glioma
T98G U87-MG
Lung
A172 H460 A549
Skin
A-431
Melanoma
A375 G-361
0 50 100 150
CS20 CS19 CS18 CS17 CS16 CS15 CS14 CS13 CS6 CS5 CS3 CS2
31
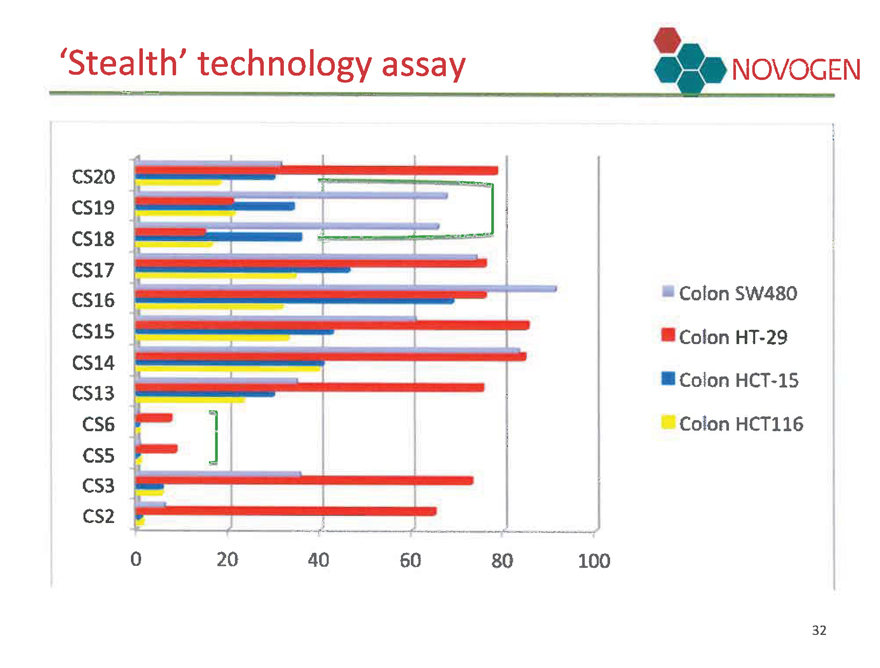
Stealth technology assay NOVOGEN
CS20 CS19 CS18 CS17 CS16 CS15 CS14 CS13 CS6 CS5 CS3 CS2
Colon SW480 Colon HT-29 Colon HCT-15 Colon HCT116
0 20 40 60 80 100
32
‘Stealth’ technology and the cancer cell wall defenses NOVOGEN
Drug
Target Cell Wall
33
Stealth Technology-older drugs blocked at cell wall NOVOGEN
Drug
Cell Wall
Target
34
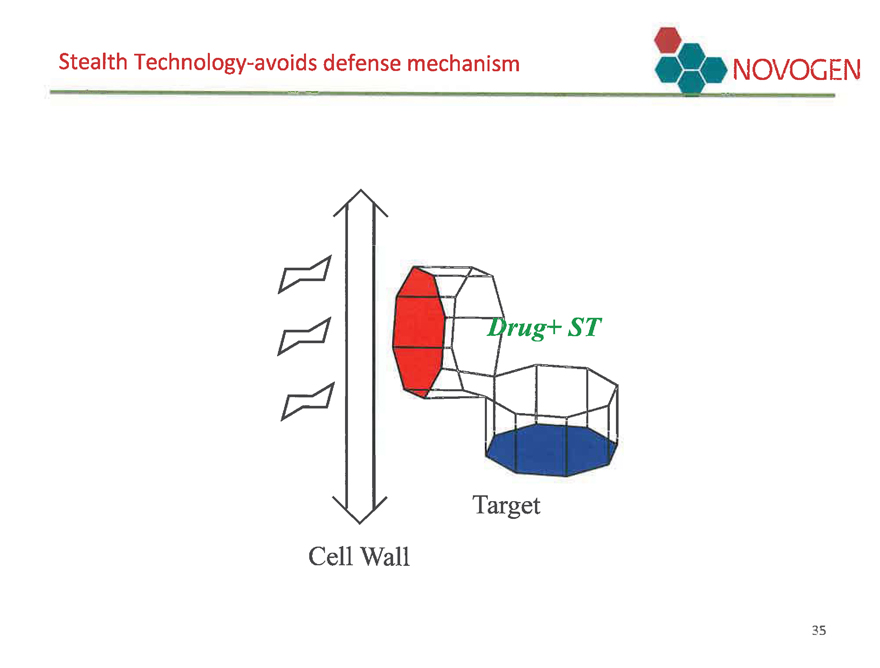
Stealth Technology-avoids defense mechanism NOVOGEN
Cell Wall
Target
Drug+ ST
35
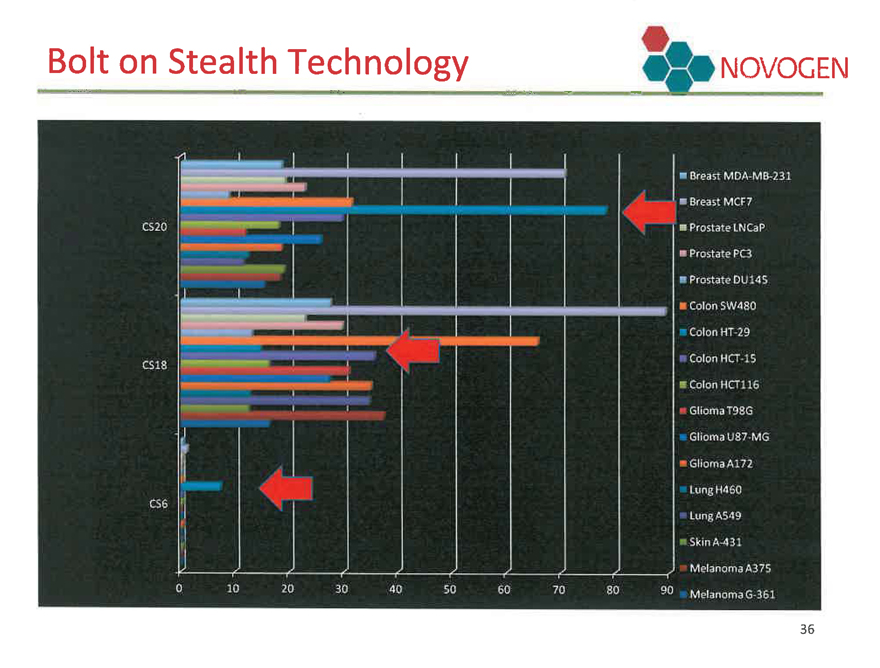
Bolt on Stealth Technology
NOVOGEN
CS20
CS18
CS6
Breast MDA-MB-231
Breast MCF7
Prostate LNCaP
Prostate PC3
Prostate DU145
Colon SW480
Colon HT-29
Colon HCT-15
Colon HCT116
Glioma T98G
Glioma U87-MG
Glioma A172
Lung H460
Lung A549
Skin A-431
Melanoma A375
Melanoma G-361
0
10
20
30
40
50
60
70
80
90
36

Enantiomers NOVOGEN
CS-6 can exist in left hand and right hand form. Initial analysis was completed on a mixture of the two.
We have developed methodologies to produce pure left and right hand forms of CS-6.
37
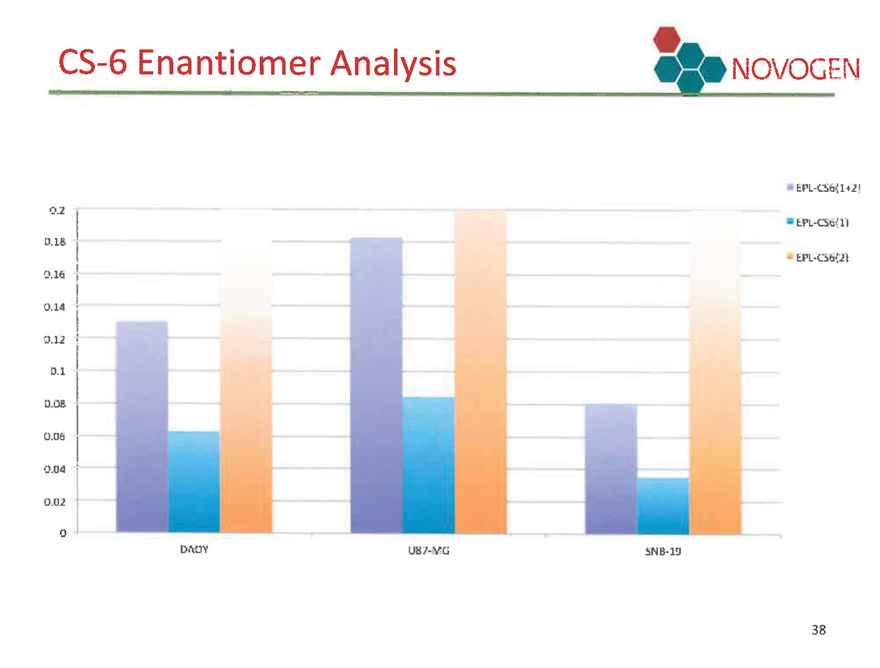
CS-6 Enantiomer Analysis NOVOGEN
0.2
0.18
0.16
0.14
0.12
0.1
0.08
0.06
0.04
0.02
0 DADY U87-MG SNB-19
EPL-CS6(1+2)
EPL-CS6(1)
EPL-CS6(2)
38

CS-6: Path to clinic NOVOGEN
Critical pre-clinical path being undertaken by one organisation (experienced CRO)
Manufacturing facility meets requirements for ALL regulatory jurisdictions (USA,
EU, Aust/NZ…)
Manufacture of the CS-6 enantiomer and its finished sterile formulation being completed at Swiss-based manufacturing facility.
39

Work In-Progress NOVOGEN
Analog Programs
Program 1: CS-6 based analogs ensuring robust patent strategy to protect CS-6 and its method of manufacture. New analogs comprising CS-6 “brothers and sisters” that help protect our IP position and also test “Double stealth Technology”. The rapid generation of these libries of compounds will afford results within the next ~ 2months.
Program 2: “Bend technology” advancement to expand further the dynamic range of of the different sub units we can add at each of the 4 stages of synthesis. Hep expnad and protect this critical IP position and potentially generate new leads in cancer lines outside brain and ovarian. This library is more elaborate and we expect results to start to flow through in ~ 6 months time.
Program 3: Quantum Bend technology” to investigate the next phase of our bend technology to elaborate some very exciting new molecules that will still map onto the required 3-D characteristics of our super-benzopyrans but take a significant step forwards. This has the potential to generate a quantum step forward in all key pharamceutic properties. Initial scoping experiments underway, initial results will determine level of resourcing placed on this program.
40

NOVOGEN
NOVOGEN
41
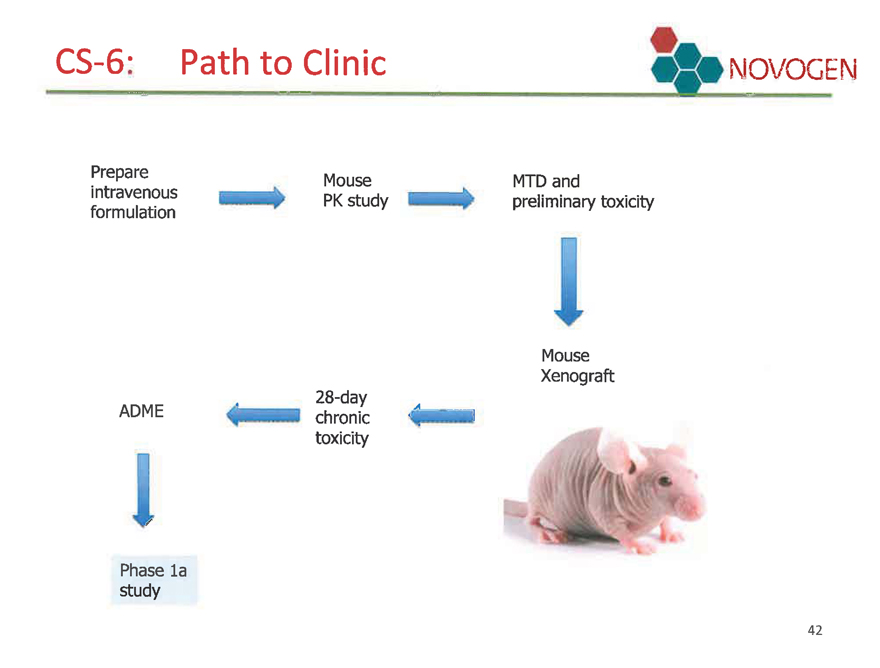
CS-6: Path to Clinic
Prepare
intravenous
formulation
Mouse PK study
ADME
Phase la study
28-day chronic toxicity
NOVOGEN
MTD and
preliminary toxicity
Mouse
Xenograft
42
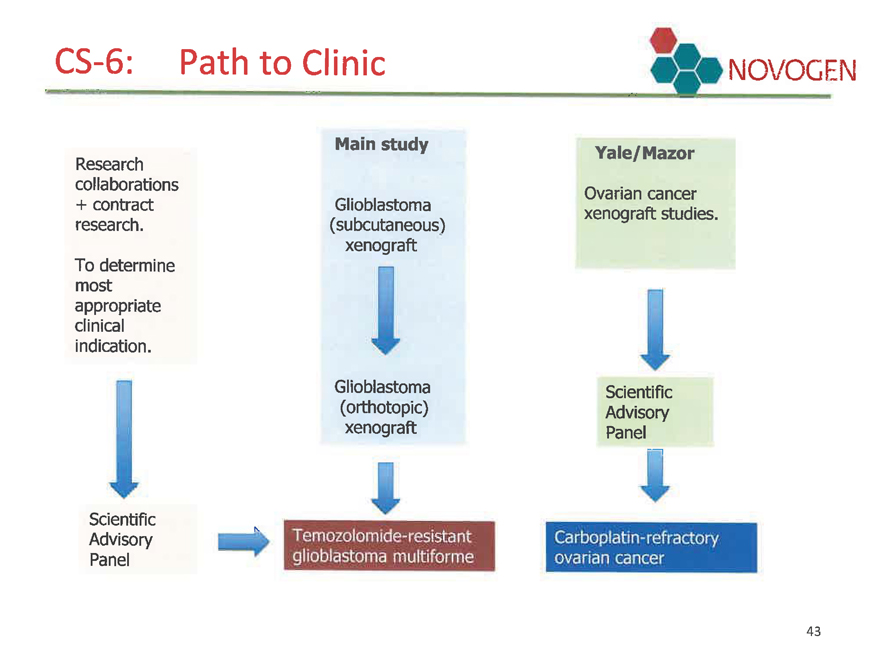
CS-6: Path to Clinic
Research collaborations + contract research.
To determine most
appropriate
clinical
indication.
Scientific
Advisory
Panel
Main study
Glioblastoma
(subcutaneous)
xenograft
Glioblastoma
(orthotopic)
xenograft
Temozolomide-resistant glioblastoma multiforme
NOVOGEN
Yale/Mazor
Ovarian cancer xenograft studies.
Scientific
Advisory
Panel
Carbopiatin-refractory ovarian cancer
43
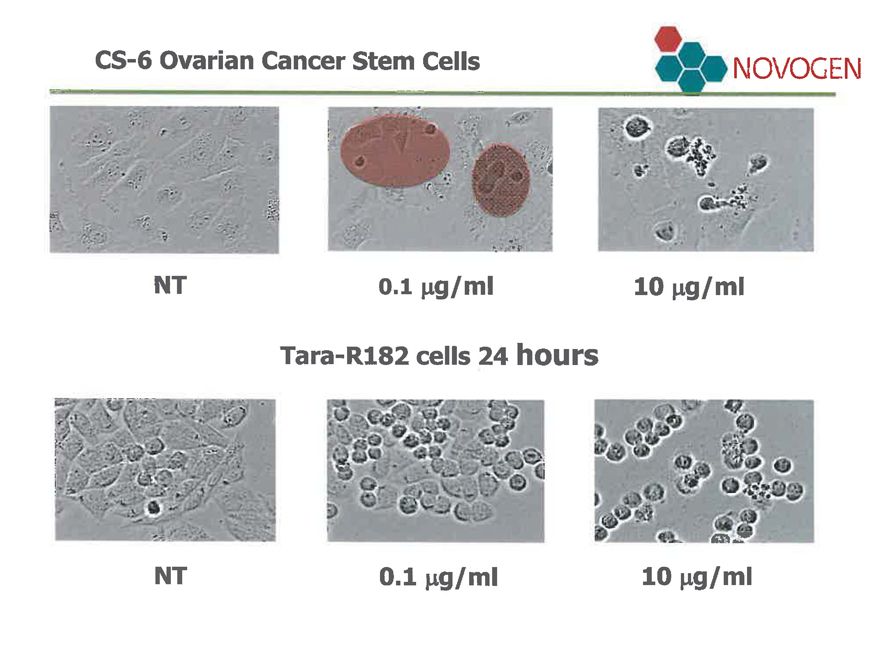
CS-6 Ovarian Cancer Stem Cells
NOVOGEN
NT
0.1 µg/ml
10 µg/ml
Tara-R182 cells 24 hours
NT
0.1 µg/ml
10 µg/ml

Cancer therapy snapshots NOVOGEN
Drug
Indication
Benefit
Market Size
Herceptin
trastuzumab
Herceptin
HER2+ breast cancer 20-25% all cases; Phase 4 only) ($50,000 p.a. treatment cost)
Increase from 20.3 vs 25.1 months survival
$6.4b pa
gleevec
Gleevec
Chronic myeloid leukaemia ($$50,000 p.a. treatment cost)
Extended survival
$4.7b pa
4500 cases USA
TAXOTERE
(docetaxel)
Injection Concentrate
Taxotere
Late-stage prostate cancer
increase from 12.7 vs 15.3 months survival
$3.1b pa
45
Website
http://www.novogen.com
NOVOGEN
46