UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of June, 2019
Commission File Number
Kazia Therapeutics Limited
(Translation of registrant’s name into English)
Three International Towers Level 24 300 Barangaroo Avenue Sydney NSW 2000
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☑ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark if the registrant by furnishing the information contained in this form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934. Yes ☐ No ☑
If “yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b)
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Kazia Therapeutics Limited(Registrant)
Kate Hill
Kate Hill
Company Secretary
Date 25 June 2019

A company developing innovative, high-impact drugs for cancer Presentation to Gold Coast Investment Showcase Surfers Paradise, QLD 25 June 2019 ASX: KZA | NASDAQ : KZIA | Twitter: @KaziaTxA company developing innovative, high-impact drugs for cancer Presentation to Gold Coast Investment Showcase Surfers Paradise, QLD 25 June 2019 ASX: KZA | NASDAQ : KZIA | Twitter: @KaziaTx

Forward-Looking Statements This presentation contains “forward-looking statements” within the meaning of the “safe- harbor” provisions of the Private Securities Litigation Reform Act of 1995. Such statements involve known and unknown risks, uncertainties and other factors that could cause the actual results of the Company to differ materially from the results expressed or implied by such statements, including changes from anticipated levels of customer acceptance of existing and new products and services and other factors. Accordingly, although the Company believes that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such expectations will prove to be correct. The Company has no obligation to sales, future international, national or regional economic and competitive conditions, changes in relationships with customers, access to capital, difficulties in developing and marketing new products and services, marketing existing products and services update the forward-looking information contained in this presentation. 1Forward-Looking Statements This presentation contains “forward-looking statements” within the meaning of the “safe- harbor” provisions of the Private Securities Litigation Reform Act of 1995. Such statements involve known and unknown risks, uncertainties and other factors that could cause the actual results of the Company to differ materially from the results expressed or implied by such statements, including changes from anticipated levels of customer acceptance of existing and new products and services and other factors. Accordingly, although the Company believes that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such expectations will prove to be correct. The Company has no obligation to sales, future international, national or regional economic and competitive conditions, changes in relationships with customers, access to capital, difficulties in developing and marketing new products and services, marketing existing products and services update the forward-looking information contained in this presentation. 1

Reasons to invest in Kazia We are primarily targeting brain cancer, a disease affecting hundreds of thousands of patients each year, representing a multi-billion dollar market, 1 and with almost no effective treatments available Our lead program, GDC-0084, was designed by Genentech, the world’s most successful cancer drug developer, and has completed a successful phase 1 2 human trial, showing it to be generally safe and providing signals of efficacy Multiple data read-outs from international human trials at world-class cancer hospitals are being delivered during calendar 2019, each with 3 significant potential to generate additional investor and partnering interest The company is fully funded through calendar 2019, having completed a successful placement to sector-specialist institutional investors last year, 4 and is listed on both ASX and NASDAQ 2Reasons to invest in Kazia We are primarily targeting brain cancer, a disease affecting hundreds of thousands of patients each year, representing a multi-billion dollar market, 1 and with almost no effective treatments available Our lead program, GDC-0084, was designed by Genentech, the world’s most successful cancer drug developer, and has completed a successful phase 1 2 human trial, showing it to be generally safe and providing signals of efficacy Multiple data read-outs from international human trials at world-class cancer hospitals are being delivered during calendar 2019, each with 3 significant potential to generate additional investor and partnering interest The company is fully funded through calendar 2019, having completed a successful placement to sector-specialist institutional investors last year, 4 and is listed on both ASX and NASDAQ 2
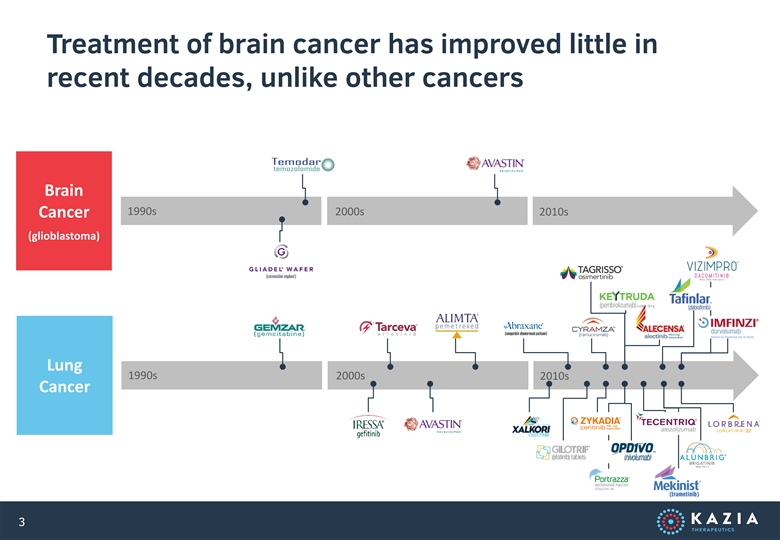
Treatment of brain cancer has improved little in recent decades, unlike other cancers Brain 1990s 2000s 2010s Cancer (glioblastoma) Lung 1990s 2000s 2010s Cancer 3Treatment of brain cancer has improved little in recent decades, unlike other cancers Brain 1990s 2000s 2010s Cancer (glioblastoma) Lung 1990s 2000s 2010s Cancer 3
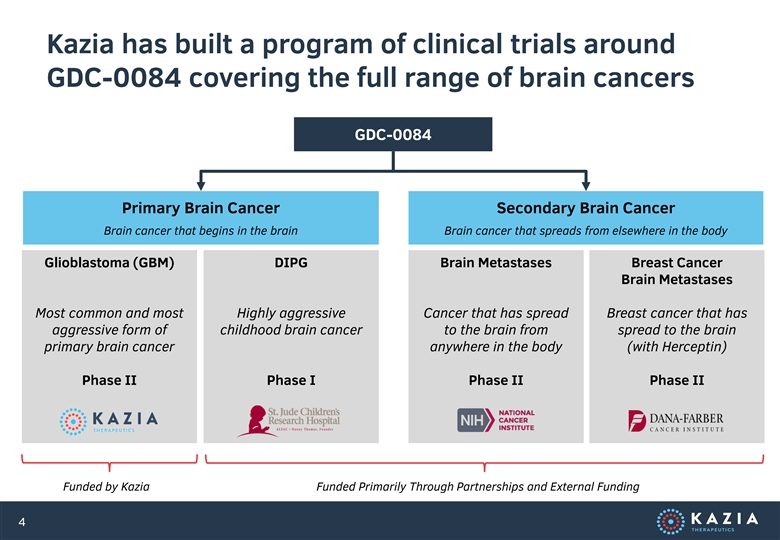
Kazia has built a program of clinical trials around GDC-0084 covering the full range of brain cancers GDC-0084 Primary Brain Cancer Secondary Brain Cancer Brain cancer that begins in the brain Brain cancer that spreads from elsewhere in the body Glioblastoma (GBM) DIPG Brain Metastases Breast Cancer Brain Metastases Most common and most Highly aggressive Cancer that has spread Breast cancer that has aggressive form of childhood brain cancer to the brain from spread to the brain primary brain cancer anywhere in the body (with Herceptin) Phase II Phase I Phase II Phase II Funded by Kazia Funded Primarily Through Partnerships and External Funding 4Kazia has built a program of clinical trials around GDC-0084 covering the full range of brain cancers GDC-0084 Primary Brain Cancer Secondary Brain Cancer Brain cancer that begins in the brain Brain cancer that spreads from elsewhere in the body Glioblastoma (GBM) DIPG Brain Metastases Breast Cancer Brain Metastases Most common and most Highly aggressive Cancer that has spread Breast cancer that has aggressive form of childhood brain cancer to the brain from spread to the brain primary brain cancer anywhere in the body (with Herceptin) Phase II Phase I Phase II Phase II Funded by Kazia Funded Primarily Through Partnerships and External Funding 4

Kazia has been delivering all milestones on schedule, with key data read-outs remaining in 2H 2019 ✓✓✓ ✓✓ Data read-out Receipt of Commence Commence Data read-out GDC-0084 Start of phase from phase 2a Orphan Drug collaborations collaboration from phase 2a 2a study in study Designation in DIPG and study (safety in metastatic first-line GBM (preliminary from US FDA BCBM and dosing) brain cancer Brain efficacy) Cancer 2018 2019 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Cantrixil Preliminary Completion of Data read-out Presentation data from from phase 1 Part A of of Part A data Ovarian phase 1 study study phase 1 study at AACR Cancer (preliminary in ovarian (safety and conference cancer dosing) efficacy) ✓✓✓ All dates are indicative and subject to operational factors 5Kazia has been delivering all milestones on schedule, with key data read-outs remaining in 2H 2019 ✓✓✓ ✓✓ Data read-out Receipt of Commence Commence Data read-out GDC-0084 Start of phase from phase 2a Orphan Drug collaborations collaboration from phase 2a 2a study in study Designation in DIPG and study (safety in metastatic first-line GBM (preliminary from US FDA BCBM and dosing) brain cancer Brain efficacy) Cancer 2018 2019 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Cantrixil Preliminary Completion of Data read-out Presentation data from from phase 1 Part A of of Part A data Ovarian phase 1 study study phase 1 study at AACR Cancer (preliminary in ovarian (safety and conference cancer dosing) efficacy) ✓✓✓ All dates are indicative and subject to operational factors 5

Glioblastoma (GBM) is the most common and most aggressive form of primary brain cancer Lung 12-15 No clear 3-4 months months cause untreated Breast survival average or strong risk 14 million survival with factors cancer cases treatment per annum Colon Any age, Five-year but most survival Prostate common in Stomach 3 – 5% 60s (breast cancer: Glioblastoma Multiforme 90%) 133,000 cases per annum worldwide Indicative Market Opportunity US$ 1.5 billion Sen. John McCain Matt Price Stan Zemanek Andrew Olle Chris O’Brien, AO US politician ABC journalist Media personality ABC journalist Surgeon 6Glioblastoma (GBM) is the most common and most aggressive form of primary brain cancer Lung 12-15 No clear 3-4 months months cause untreated Breast survival average or strong risk 14 million survival with factors cancer cases treatment per annum Colon Any age, Five-year but most survival Prostate common in Stomach 3 – 5% 60s (breast cancer: Glioblastoma Multiforme 90%) 133,000 cases per annum worldwide Indicative Market Opportunity US$ 1.5 billion Sen. John McCain Matt Price Stan Zemanek Andrew Olle Chris O’Brien, AO US politician ABC journalist Media personality ABC journalist Surgeon 6
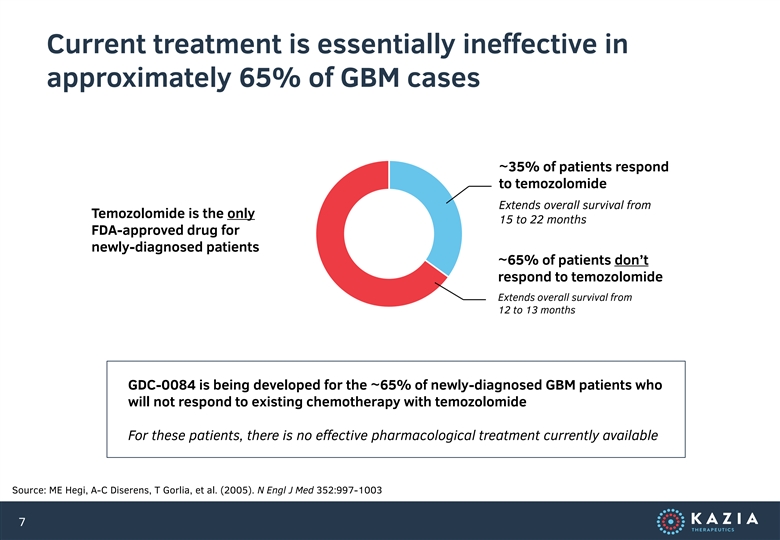
Current treatment is essentially ineffective in approximately 65% of GBM cases ~35% of patients respond to temozolomide Extends overall survival from Temozolomide is the only 15 to 22 months FDA-approved drug for newly-diagnosed patients ~65% of patients don’t respond to temozolomide Extends overall survival from 12 to 13 months GDC-0084 is being developed for the ~65% of newly-diagnosed GBM patients who will not respond to existing chemotherapy with temozolomide For these patients, there is no effective pharmacological treatment currently available Source: ME Hegi, A-C Diserens, T Gorlia, et al. (2005). N Engl J Med 352:997-1003 7Current treatment is essentially ineffective in approximately 65% of GBM cases ~35% of patients respond to temozolomide Extends overall survival from Temozolomide is the only 15 to 22 months FDA-approved drug for newly-diagnosed patients ~65% of patients don’t respond to temozolomide Extends overall survival from 12 to 13 months GDC-0084 is being developed for the ~65% of newly-diagnosed GBM patients who will not respond to existing chemotherapy with temozolomide For these patients, there is no effective pharmacological treatment currently available Source: ME Hegi, A-C Diserens, T Gorlia, et al. (2005). N Engl J Med 352:997-1003 7

GDC-0084 works by switching off PI3K, a critical control mechanism that drives many types of cancer PI3K ACTIVATED 85 – 90% of glioblastoma cases TUMOUR have PI3K ‘switched on’ GROWTH PI3K A central biochemical control PI3K INHIBITED mechanism in the growth of cells TUMOUR GDC-0084 switches off PI3K, halting growth of tumour cells INHIBITION 8GDC-0084 works by switching off PI3K, a critical control mechanism that drives many types of cancer PI3K ACTIVATED 85 – 90% of glioblastoma cases TUMOUR have PI3K ‘switched on’ GROWTH PI3K A central biochemical control PI3K INHIBITED mechanism in the growth of cells TUMOUR GDC-0084 switches off PI3K, halting growth of tumour cells INHIBITION 8

GDC-0084 is the only drug of its kind that is able to cross the ‘blood-brain barrier’ (BBB) Most drugs cannot reach disease in the brain GDC-0084 crosses the BBB The ‘blood-brain barrier’ prevents most drugs from GDC-0084 was specifically designed for brain cancer, and getting into the brain, rendering them useless as has been engineered to cross the blood-brain barrier, treatments for brain cancer making it well-placed to treat brain cancer 9GDC-0084 is the only drug of its kind that is able to cross the ‘blood-brain barrier’ (BBB) Most drugs cannot reach disease in the brain GDC-0084 crosses the BBB The ‘blood-brain barrier’ prevents most drugs from GDC-0084 was specifically designed for brain cancer, and getting into the brain, rendering them useless as has been engineered to cross the blood-brain barrier, treatments for brain cancer making it well-placed to treat brain cancer 9

A phase 1 human trial of GDC-0084 showed favourable safety and multiple efficacy signals Safety Efficacy Signals • Phase I safety trial conducted by GDC-0084 Comparison Genentech 40% 21-52% • 47 patients enrolled with advanced Arresting Achieved in studies of glioma (grade 3/4); average of Tumour Growth ‘stable disease’ Avastin in similar three prior lines of therapy patients • Most common adverse events were Median oral mucositis and hyperglycemia 21% Potentially progression-free (common effects of PI3K inhibitors) Remained on study Delaying survival of for >3 months Progression • No evidence of liver, bone marrow, 1 month* kidney toxicity, or mood disturbances Potentially better 26% predictor of clinical • Data presented at American Slowing Tumour Showed ‘metabolic response than Society for Clinical Oncology annual Metabolism partial response’ † MRI meeting in Chicago, June 2016 on FDG-PET * Taal et al., Lancet Oncology (2015): ORR and mPFS of Lomustine in 2L GBM were 2/41 (5%) and 1 months, respectively (n = 46) † Schwarzenberg J, et al. Clin Cancer Res; 20(13); 3550–9 10A phase 1 human trial of GDC-0084 showed favourable safety and multiple efficacy signals Safety Efficacy Signals • Phase I safety trial conducted by GDC-0084 Comparison Genentech 40% 21-52% • 47 patients enrolled with advanced Arresting Achieved in studies of glioma (grade 3/4); average of Tumour Growth ‘stable disease’ Avastin in similar three prior lines of therapy patients • Most common adverse events were Median oral mucositis and hyperglycemia 21% Potentially progression-free (common effects of PI3K inhibitors) Remained on study Delaying survival of for >3 months Progression • No evidence of liver, bone marrow, 1 month* kidney toxicity, or mood disturbances Potentially better 26% predictor of clinical • Data presented at American Slowing Tumour Showed ‘metabolic response than Society for Clinical Oncology annual Metabolism partial response’ † MRI meeting in Chicago, June 2016 on FDG-PET * Taal et al., Lancet Oncology (2015): ORR and mPFS of Lomustine in 2L GBM were 2/41 (5%) and 1 months, respectively (n = 46) † Schwarzenberg J, et al. Clin Cancer Res; 20(13); 3550–9 10

In the GDC-0084 phase 1 trial, 7 / 27 patients (26%) showed a response to drug* Pre-Dosing On GDC-0084 Pre-Dosing On GDC-0084 * Metabolic partial response per FDG-PET Analysis courtesy of Professor Ben Ellingson, UCLA Brain Tumor Imaging Laboratory 11In the GDC-0084 phase 1 trial, 7 / 27 patients (26%) showed a response to drug* Pre-Dosing On GDC-0084 Pre-Dosing On GDC-0084 * Metabolic partial response per FDG-PET Analysis courtesy of Professor Ben Ellingson, UCLA Brain Tumor Imaging Laboratory 11
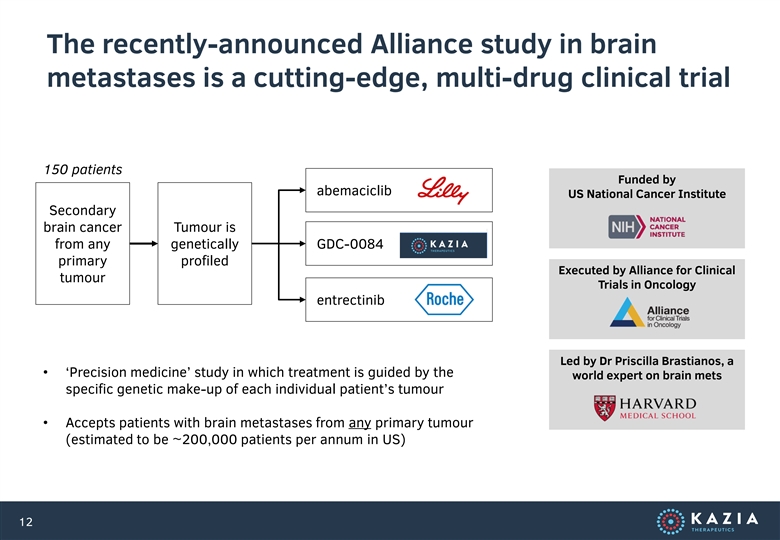
The recently-announced Alliance study in brain metastases is a cutting-edge, multi-drug clinical trial 150 patients Funded by abemaciclib US National Cancer Institute Secondary brain cancer Tumour is from any genetically GDC-0084 primary profiled Executed by Alliance for Clinical tumour Trials in Oncology entrectinib Led by Dr Priscilla Brastianos, a • ‘Precision medicine’ study in which treatment is guided by the world expert on brain mets specific genetic make-up of each individual patient’s tumour • Accepts patients with brain metastases from any primary tumour (estimated to be ~200,000 patients per annum in US) 12The recently-announced Alliance study in brain metastases is a cutting-edge, multi-drug clinical trial 150 patients Funded by abemaciclib US National Cancer Institute Secondary brain cancer Tumour is from any genetically GDC-0084 primary profiled Executed by Alliance for Clinical tumour Trials in Oncology entrectinib Led by Dr Priscilla Brastianos, a • ‘Precision medicine’ study in which treatment is guided by the world expert on brain mets specific genetic make-up of each individual patient’s tumour • Accepts patients with brain metastases from any primary tumour (estimated to be ~200,000 patients per annum in US) 12
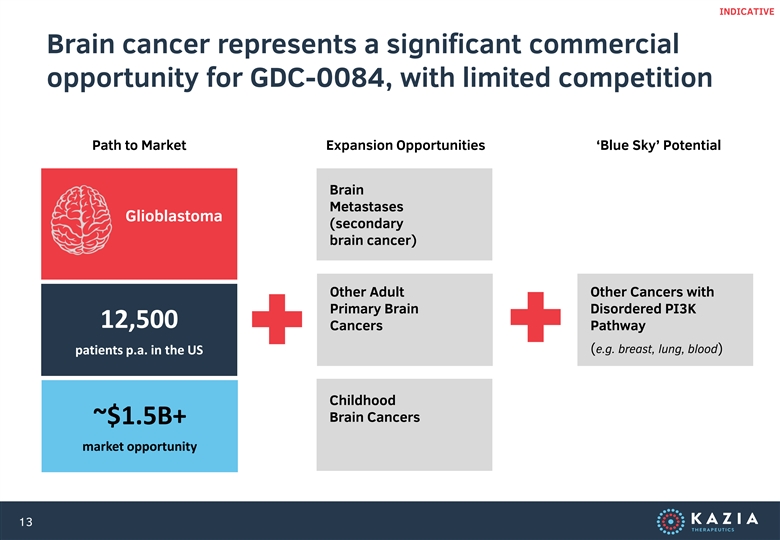
INDICATIVE Brain cancer represents a significant commercial opportunity for GDC-0084, with limited competition Path to Market Expansion Opportunities ‘Blue Sky’ Potential Brain Metastases Glioblastoma (secondary brain cancer) Other Adult Other Cancers with Primary Brain Disordered PI3K 12,500 Cancers Pathway (e.g. breast, lung, blood) patients p.a. in the US Childhood Brain Cancers ~$1.5B+ market opportunity 13INDICATIVE Brain cancer represents a significant commercial opportunity for GDC-0084, with limited competition Path to Market Expansion Opportunities ‘Blue Sky’ Potential Brain Metastases Glioblastoma (secondary brain cancer) Other Adult Other Cancers with Primary Brain Disordered PI3K 12,500 Cancers Pathway (e.g. breast, lung, blood) patients p.a. in the US Childhood Brain Cancers ~$1.5B+ market opportunity 13
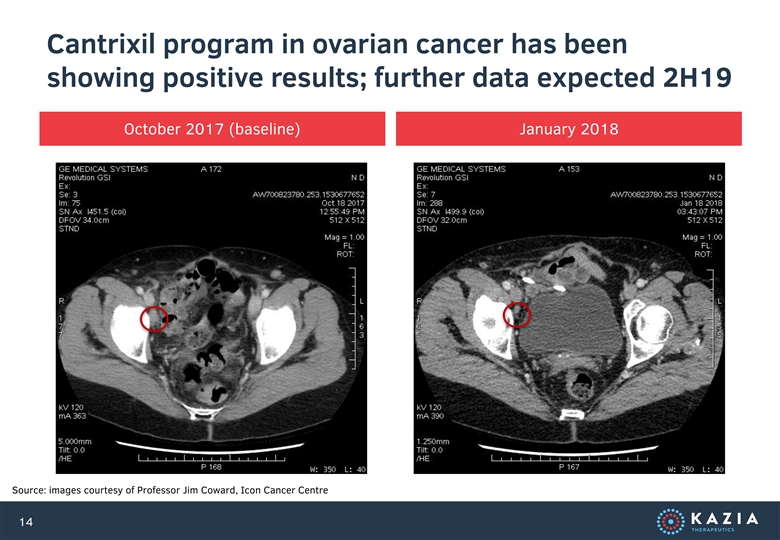
Cantrixil program in ovarian cancer has been showing positive results; further data expected 2H19 October 2017 (baseline) January 2018 Source: images courtesy of Professor Jim Coward, Icon Cancer Centre 14Cantrixil program in ovarian cancer has been showing positive results; further data expected 2H19 October 2017 (baseline) January 2018 Source: images courtesy of Professor Jim Coward, Icon Cancer Centre 14

Kazia is NASDAQ & ASX listed Share Price (AU$) KZA 0.60 Current Assets (Dec 18) Debt AU$ 11.6 million Nil Market Capitalisation AU$ 22 million 0.50 NASDAQ: KZIA (1:10 ratio) Listing ASX: KZA 0.40 NASDAQ: 0.2% /day Average Daily Volume ASX: 0.1% /day NASDAQ: US$ 21K /day Average Daily Value ASX: AU$ 11K /day 0.30 62 million Shares on Issue (25% US, 75% Australia) 0.20 Outstanding ~6 million Options / Warrants 02-Dec-18 02-Feb-19 02-Apr-19 02-Jun-19 15Kazia is NASDAQ & ASX listed Share Price (AU$) KZA 0.60 Current Assets (Dec 18) Debt AU$ 11.6 million Nil Market Capitalisation AU$ 22 million 0.50 NASDAQ: KZIA (1:10 ratio) Listing ASX: KZA 0.40 NASDAQ: 0.2% /day Average Daily Volume ASX: 0.1% /day NASDAQ: US$ 21K /day Average Daily Value ASX: AU$ 11K /day 0.30 62 million Shares on Issue (25% US, 75% Australia) 0.20 Outstanding ~6 million Options / Warrants 02-Dec-18 02-Feb-19 02-Apr-19 02-Jun-19 15

A strong team brings international experience in big pharma and early-stage biotech Board Scientific Advisory Board Iain Ross Professor Sir Murray Brennan Chairman Emeritus Chairman of Cancer Surgery at Memorial Sloan Kettering Hospital, New York Executive and Board roles in pharma and small biotech Bryce Carmine Dr Karen Ferrante Deputy Chairman Former Chief Medical Officer at Millennium Pharmaceuticals 36 years executive experience in Eli Lilly Steven Coffey Professor Peter Gunning Non-Executive Director Head of School of Medical Sciences at University of New South Wales Chartered accountant with extensive governance experience Dr James Garner Professor Alex Matter Chief Executive Officer Former Global Head of Oncology & Executive Director Research at Novartis Physician / MBA; Extensive drug development experience 16A strong team brings international experience in big pharma and early-stage biotech Board Scientific Advisory Board Iain Ross Professor Sir Murray Brennan Chairman Emeritus Chairman of Cancer Surgery at Memorial Sloan Kettering Hospital, New York Executive and Board roles in pharma and small biotech Bryce Carmine Dr Karen Ferrante Deputy Chairman Former Chief Medical Officer at Millennium Pharmaceuticals 36 years executive experience in Eli Lilly Steven Coffey Professor Peter Gunning Non-Executive Director Head of School of Medical Sciences at University of New South Wales Chartered accountant with extensive governance experience Dr James Garner Professor Alex Matter Chief Executive Officer Former Global Head of Oncology & Executive Director Research at Novartis Physician / MBA; Extensive drug development experience 16

Other companies focused on the PI3K pathway have been highly-valued in the market Single asset company with one PI3K US$ 90 million inhibitor in phase I human trials Market Cap One PI3K inhibitor in phase II human US$ 620 million trials, one other drug in phase III, and Market Cap two in animal testing US$ 140 million One PI3K inhibitor approved in October 2018 for certain blood cancers, one Market Cap other drug in human trials Acquired by big One PI3K inhibitor in phase II human pharma in 2011 for trials US$ 375 million 17Other companies focused on the PI3K pathway have been highly-valued in the market Single asset company with one PI3K US$ 90 million inhibitor in phase I human trials Market Cap One PI3K inhibitor in phase II human US$ 620 million trials, one other drug in phase III, and Market Cap two in animal testing US$ 140 million One PI3K inhibitor approved in October 2018 for certain blood cancers, one Market Cap other drug in human trials Acquired by big One PI3K inhibitor in phase II human pharma in 2011 for trials US$ 375 million 17

Reasons to invest in Kazia We are primarily targeting brain cancer, a disease affecting hundreds of thousands of patients each year, representing a multi-billion dollar market, 1 and with almost no effective treatments available Our lead program, GDC-0084, was designed by Genentech, the world’s most successful cancer drug developer, and has completed a successful phase 1 2 human trial, showing it to be generally safe and providing signals of efficacy Multiple data read-outs from international human trials at world-class cancer hospitals are being delivered during calendar 2019, each with 3 significant potential to generate additional investor and partnering interest The company is fully funded through calendar 2019, having completed a successful placement to sector-specialist institutional investors last year, 4 and is listed on both ASX and NASDAQ 18Reasons to invest in Kazia We are primarily targeting brain cancer, a disease affecting hundreds of thousands of patients each year, representing a multi-billion dollar market, 1 and with almost no effective treatments available Our lead program, GDC-0084, was designed by Genentech, the world’s most successful cancer drug developer, and has completed a successful phase 1 2 human trial, showing it to be generally safe and providing signals of efficacy Multiple data read-outs from international human trials at world-class cancer hospitals are being delivered during calendar 2019, each with 3 significant potential to generate additional investor and partnering interest The company is fully funded through calendar 2019, having completed a successful placement to sector-specialist institutional investors last year, 4 and is listed on both ASX and NASDAQ 18

www.kaziatherapeutics.comwww.kaziatherapeutics.com