UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of November, 2019
Commission File Number
Kazia Therapeutics Limited
(Translation of registrant’s name into English)
Three International Towers Level 24 300 Barangaroo Avenue Sydney NSW 2000
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☑ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark if the registrant by furnishing the information contained in this form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934. Yes ☐ No ☑
If “yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b)
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Kazia Therapeutics Limited(Registrant)
Kate Hill
Kate Hill
Company Secretary
Date 13 November 2019

US RELEASE
13 November 2019
KAZIA ANNUAL GENERAL MEETING RESULTS
Sydney, 13 November 2019 – Kazia Therapeutics Limited (ASX: KZA; NASDAQ: KZIA), an Australian oncology-focused biotechnology company, is pleased to announce the results of its Annual General Meeting of shareholders.
All resolutions were carried. The vote totals are attached.
Kazia Chairman, Iain Ross, commented “we are grateful to once again have the comprehensive support of our shareholders during this exciting chapter in the company’s development. Almost 40% of the issued share capital was voted, and support of the resolutions exceeded 95% in all cases, providing a powerful endorsement of the company’s strategy and potential. In less than two weeks, we expect to announce initial interim data from the ongoing phase II study ofGDC-0084, and this will mark an important inflection point as we head into 2020.”
The materials presented to the meeting, comprising the Chairman’s address and CEO presentation, are attached.
[ENDS]
About Kazia Therapeutics Limited
Kazia Therapeutics Limited (ASX: KZA, NASDAQ: KZIA) is an innovative oncology-focused biotechnology company, based in Sydney, Australia. Our pipeline includes two clinical-stage drug development candidates, and we are working to develop therapies across a range of oncology indications.
Our lead program isGDC-0084, a small molecule inhibitor of the PI3K / AKT / mTOR pathway, which is being developed to treat glioblastoma multiforme, the most common and most aggressive form of primary brain cancer in adults. Licensed from Genentech in late 2016,GDC-0084 entered a phase II clinical trial in 2018. Initial safety data was released in May 2019, and further data is expected in 2H 2019.GDC-0084 was granted orphan designation for glioblastoma by the US FDA in February 2018.
TRX-E-002-1 (Cantrixil), is a third-generation benzopyran molecule with activity against cancer stem cells and is being developed to treat ovarian cancer.TRX-E-002-1 is currently undergoing a phase I clinical trial in Australia and the United States. Interim data was presented at the ESMO Congress in September 2019, and the study remains ongoing. Cantrixil was granted orphan designation for ovarian cancer by the US FDA in April 2015.
Board of Directors
Mr Iain RossChairman,Non-Executive Director
Mr Bryce CarmineNon-Executive Director
Mr Steven CoffeyNon-Executive Director
Dr James GarnerChief Executive Officer, Managing Director
Kazia Therapeutics Limited
2019 Annual General Meeting
Wednesday, 13 November 2019
Voting Results
The following information is provided in accordance with section 251AA(2) of the Corporations Act 2001 (Cth).
Resolution details | Instructions given to validly appointed proxies (as at proxy close) | Number of votes cast on the poll (where applicable) | Resolution | |||||||||||||||
Resolution | Resolution | For | Against | Proxy’s Discretion | Abstain | For | Against | Abstain* | Carried / | |||||||||
1 Adoption of Remuneration Report | Ordinary | 26,502,358 97.56% | 373,826 1.37% | 291,229 1.07% | 575,965 | 27,098,515 98.64% | 373,826 1.36% | 575,965 | Carried | |||||||||
2Re-election of Steven Coffey | Ordinary | 27,771,657 98.16% | 229,649 0.81% | 291,229 1.03% | 358,611 | 28,367,814 99.20% | 229,649 0.80% | 358,611 | Carried | |||||||||
3 Approval of grant of Options to Dr James Garner | Ordinary | 25,798,206 94.80% | 1,138,345 4.19% | 275,854 1.01% | 650,973 | 26,344,060 95.85% | 1,140,768 4.15% | 683,478 | Carried | |||||||||
4 Placement capacity under Listing Rule 7.1A | Special | 27,527,737 97.81% | 325,821 1.16% | 291,229 1.03% | 506,359 | 28,121,471 98.85% | 328,244 1.15% | 506,359 | Carried | |||||||||
| * | Votes cast by a person who abstains on an item are not counted in calculating the required majority on a poll. |
KAZIA ANNUAL GENERAL MEETING
13 NOVEMBER 2019
CHAIRMAN’S ADDRESS
Ladies and Gentlemen,
It is my pleasure once again to welcome you to the Annual General Meeting for Kazia Therapeutics Limited. This is my third AGM as Chairman of Kazia, and I can say with confidence that 2019 has been one of the most exciting years in our company’s short history.
The reason for that excitement is, in a word, data. The lifeblood of any drug development company is the data that it is able to generate from its clinical trials. That data represents economic value for shareholders and it represents hope for patients. There is no real room for gloss or hype or spin – objective data provides the hard facts on which professional investors and potential partners will ultimately judge us.
We have had three important data read-outs this year Perhaps the most important one, however, is coming in just over a week from today.
In May, we announced thatGDC-0084 had achieved a higher maximum tolerated dose – MTD – in newly-diagnosed patients than in the original Genentech phase I study. This is a very encouraging indication that the drug is well tolerated in the precise patient group that we are targeting for commercialisation. Our ability to administer a higher dose can only bode well for our prospects of demonstrating clinical benefit.
In September, our colleagues at St Jude Children’s Research Hospital achieved a comparable MTD in childhood brain cancer. It is very positive to know that the drug is also tolerable in children, and the St Jude team are currently recruiting additional patients to look for potential efficacy signals. I would remind everyone that there are no approved drug treatments for this form of brain cancer, and the average survival from diagnosis is approximately nine months. It would be remarkable if we are able to offer benefit to patients and their families.
Also in September, we presented interim data from our ongoing Cantrixil study in ovarian cancer at the prestigious ESMO conference. The data suggested a potential increase in progression-free survival for patients treated with Cantrixil. Given that these are very late stage patients who are very resistant to treatment, this is a tremendous result. I had the pleasure of meeting with our lead investigator yesterday, and his excitement at the emerging data was quite palpable.
In short, we find ourselves in a very strong position. However, we have perhaps saved the best for last. Next week, we will present the first preliminary efficacy data from the ongoingGDC-0084 phase II study in glioblastoma. The study is still ongoing, and so this will only be an early glimpse, but I know that a wide range of stakeholders will be watching with great interest. The median progression-free survival for the patients we are targeting is only around five months, so any preliminary indication that we are able to prolong this duration is likely to be of very high impact.
To see these projects through to their completion, your Board chose to capitalise on growing investor interest and conduct a modest share placement to strengthen the company’s balance sheet. As always, our overriding concern has been to ensure that we are able to deliver value from our pipeline while safeguarding the interests of existing investors. We have once again raised only what is needed to drive the next round of data generation. Despite a very challenging environment, our placement was conducted without the need for options or warrants, and has brought additional high-quality institutional investors on to the registry. I am pleased to take this opportunity to welcome them to Kazia.
Looking ahead, we aspire to takeGDC-0084 into a pivotal study next year, and we will be examining every option to determine the best way to deliver a high-quality program within our means. Kazia has demonstrated an incredibly innovative approach to partnering for clinical development, and we hope that these capabilities will allow us to bring something novel, efficient, and world-class to the next chapter ofGDC-0084’s development. I look forward to sharing more with you in due course.
In the meantime, I must thank you again, on behalf of my fellow directors, for your ongoing support of the company. I recommend today’s resolutions to you, and invite you to continue shaping the future success of Kazia.

Presentation to Annual General Meeting of Shareholders Dr James Garner Chief Executive Officer Sydney, NSW 13 November 2019

Agenda 2019 in Review Looking Forward
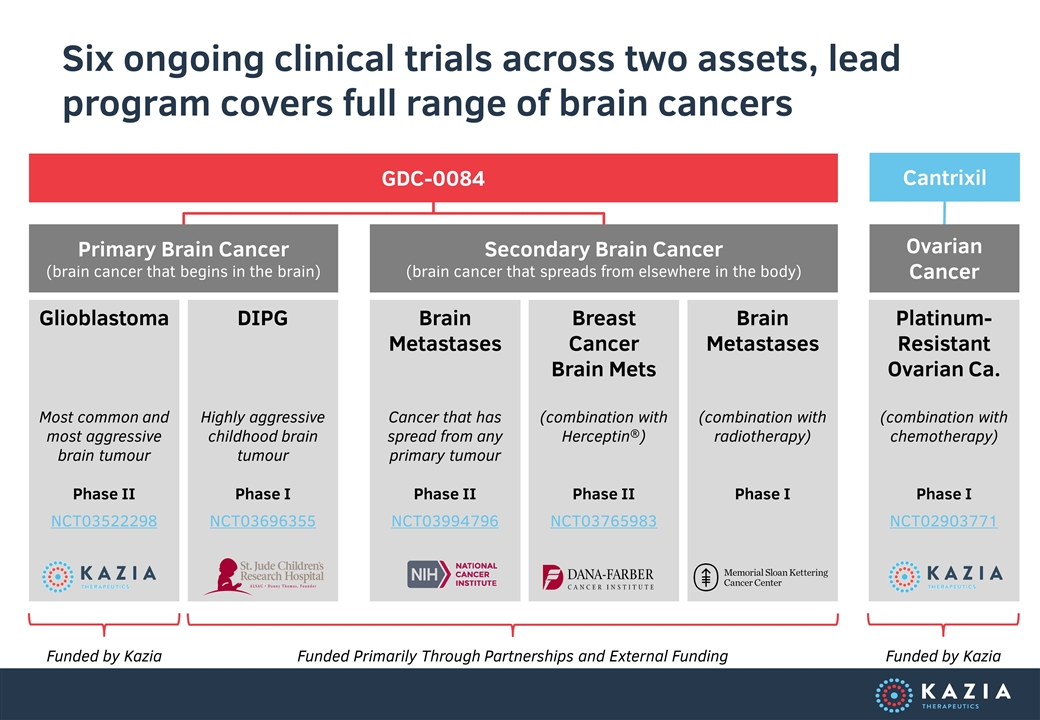
Six ongoing clinical trials across two assets, lead program covers full range of brain cancers Glioblastoma Most common and most aggressive brain tumour Phase II NCT03522298 DIPG Highly aggressive childhood brain tumour Phase I NCT03696355 Brain Metastases Cancer that has spread from any primary tumour Phase II NCT03994796 Breast Cancer Brain Mets (combination with Herceptin®) Phase II NCT03765983 Brain Metastases (combination with radiotherapy) Phase I Platinum-Resistant Ovarian Ca. (combination with chemotherapy) Phase I NCT02903771 Primary Brain Cancer (brain cancer that begins in the brain) Secondary Brain Cancer (brain cancer that spreads from elsewhere in the body) Ovarian Cancer GDC-0084 Cantrixil Funded by Kazia Funded Primarily Through Partnerships and External Funding Funded by Kazia
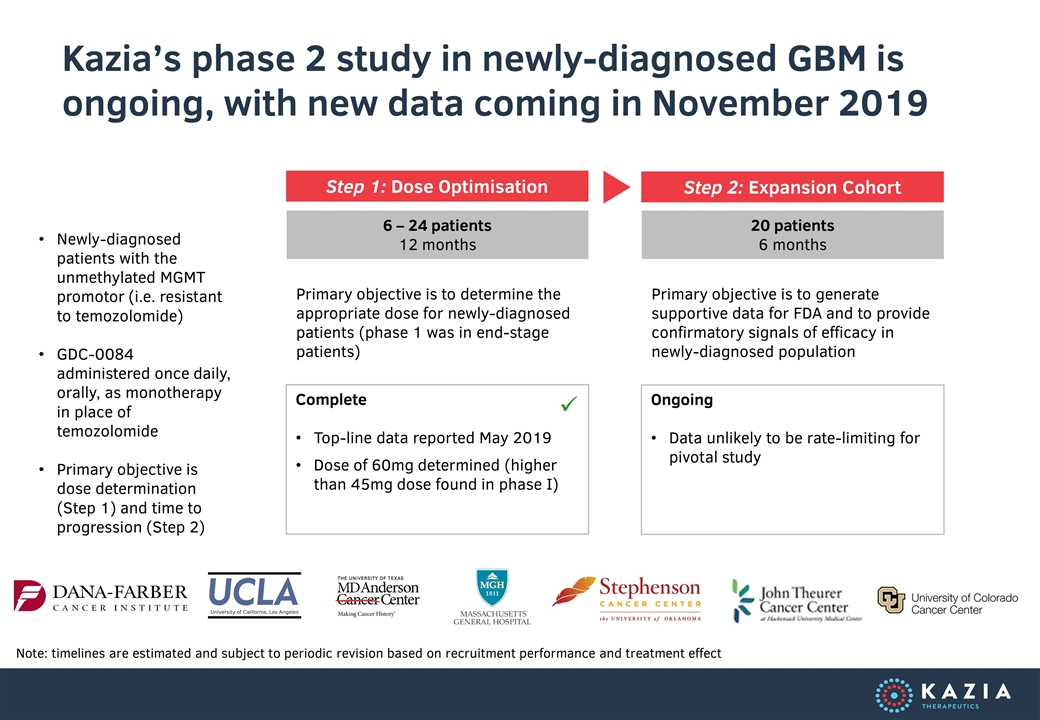
Kazia’s phase 2 study in newly-diagnosed GBM is ongoing, with new data coming in November 2019 Note: timelines are estimated and subject to periodic revision based on recruitment performance and treatment effect Step 1: Dose Optimisation 6 – 24 patients 12 months 20 patients 6 months Step 2: Expansion Cohort Newly-diagnosed patients with the unmethylated MGMT promotor (i.e. resistant to temozolomide) GDC-0084 administered once daily, orally, as monotherapy in place of temozolomide Primary objective is dose determination (Step 1) and time to progression (Step 2) Primary objective is to determine the appropriate dose for newly-diagnosed patients (phase 1 was in end-stage patients) Primary objective is to generate supportive data for FDA and to provide confirmatory signals of efficacy in newly-diagnosed population Complete Top-line data reported May 2019 Dose of 60mg determined (higher than 45mg dose found in phase I) ü Ongoing Data unlikely to be rate-limiting for pivotal study
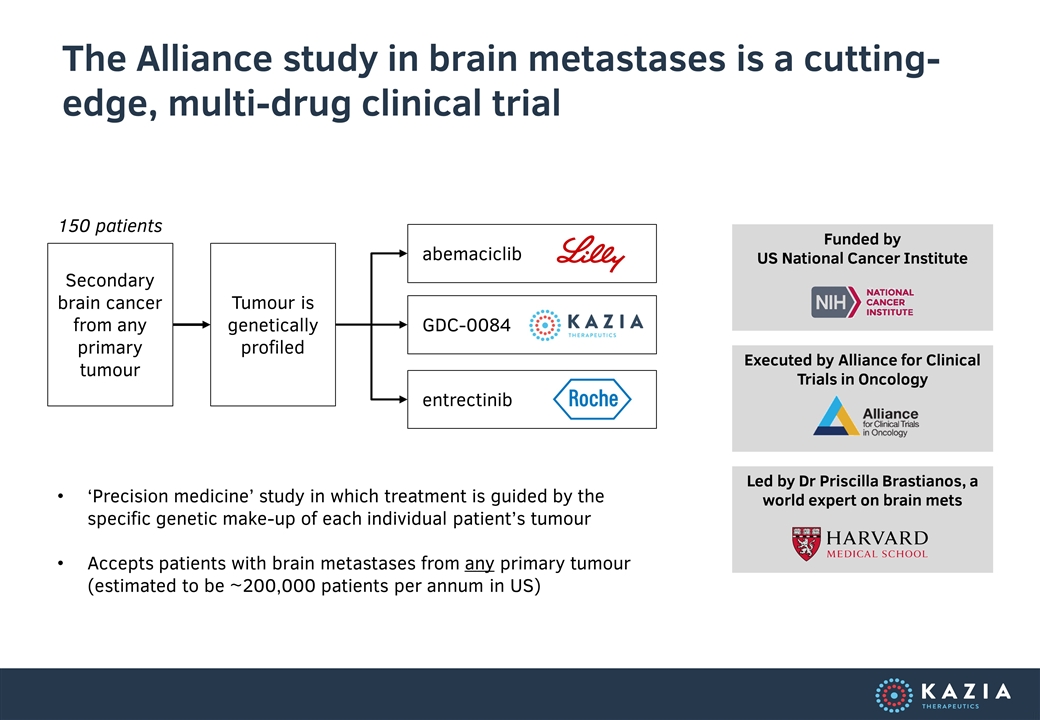
The Alliance study in brain metastases is a cutting-edge, multi-drug clinical trial Tumour is genetically profiled GDC-0084 entrectinib abemaciclib 150 patients Secondary brain cancer from any primary tumour ‘Precision medicine’ study in which treatment is guided by the specific genetic make-up of each individual patient’s tumour Accepts patients with brain metastases from any primary tumour (estimated to be ~200,000 patients per annum in US) Funded by US National Cancer Institute Executed by Alliance for Clinical Trials in Oncology Led by Dr Priscilla Brastianos, a world expert on brain mets

The St Jude study in DIPG has the potential for breakthrough designation and early approval Step 1: Dose Escalation 6 – 24 patients 12 patients Step 2: Expansion Cohort All patients with DIPG or high-grade gliomas (2 – 22 years of age), following radiotherapy GDC-0084 given once daily, orally, as monotherapy Primary objective is dose determination (Step 1) and time to progression (Step 2) Given no FDA-approved therapies for DIPG, a successful result could lead to discussion of early approval Primary objective is to determine the appropriate dose for pediatric use (mg/kg dosing) Primary objective is to provide initial evidence of clinical efficacy Complete Top-line data reported Sept. 2019 Dose of 27 mg/m2 determined for paediatric use (comparable to adult doses) Ongoing ü

Important new preclinical data has also been reported during the year References: Duchatel et al. Neuro-Oncology (2019). 21(Suppl. 2):ii68; Ippen et al. Clin Cancer Res. (2019). 25(11):3374-83 DIPG Breast Cancer Brain Metastases

The PI3K class has been further validated by the approval of Novartis’ Piqray (alpelisib) FDA Approved July 2014 (blood cancers) [accelerated approval] FDA Approved September 2017 (blood cancers) [accelerated approval] FDA Approved October 2018 (blood cancers) [accelerated approval] ü ü ü Potentially fatal liver toxicity and diarrhoea Potentially fatal infections Potentially fatal infections & diarrhoea Does not cross blood-brain barrier Does not cross blood-brain barrier Does not cross blood-brain barrier û û û û û û Zydelig (idelalisib) Aliqopa (copanlisib) Copiktra (duvelisib) FDA Approved May 2019 (breast cancer) [accelerated approval] Limited toxicities to date Does not cross blood-brain barrier ü û ü Piqray (alpelisib) In phase II human trials under US FDA oversight (brain cancer) Appears generally safe and well-tolerated thus far Does cross blood-brain barrier ü ü GDC-0084

Our efforts continue to be recognised in the public sphere

A range of content, from academic papers to media interviews, helps investors grasp our story

The Trans-Tasman Innovation and Growth Award was a powerful recognition of Kazia’s achievements
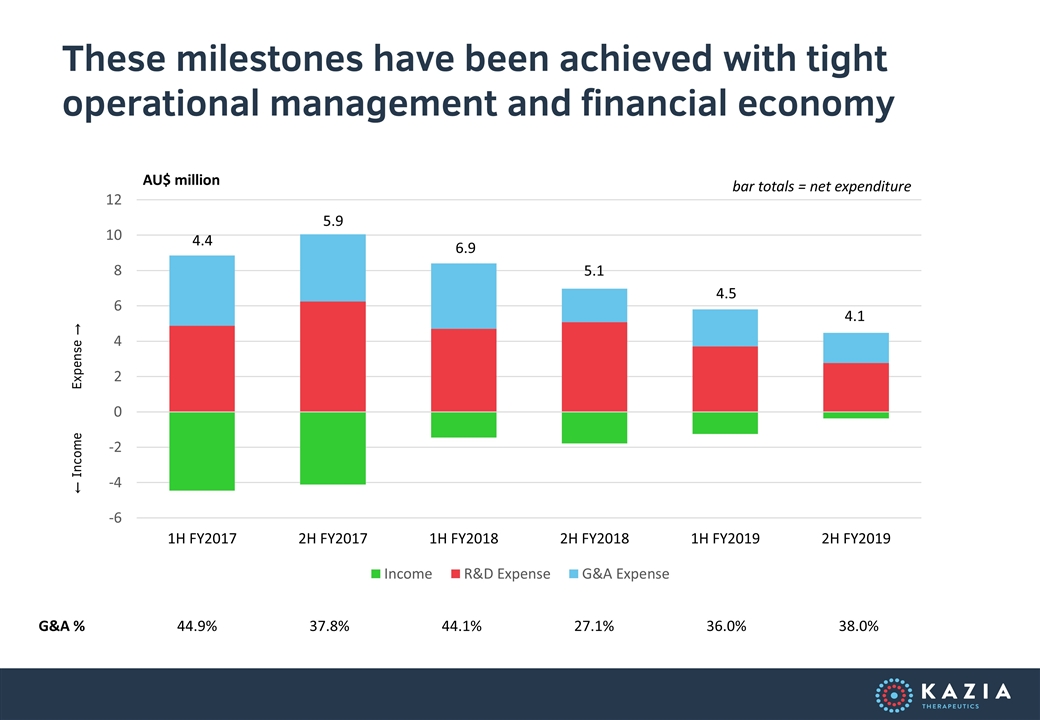
These milestones have been achieved with tight operational management and financial economy AU$ million 4.4 5.9 6.9 5.1 4.5 Expense → ← Income G&A % 44.9% 37.8% 44.1% 27.1% 36.0% bar totals = net expenditure 38.0% 4.1
Agenda 2019 in Review Looking Forward
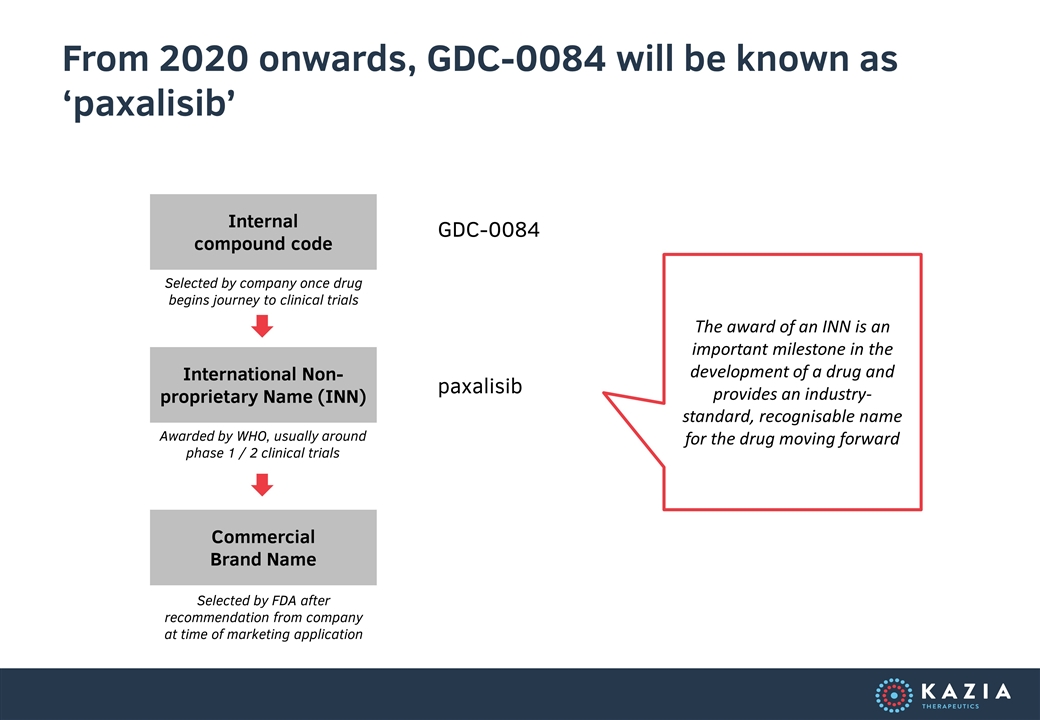
From 2020 onwards, GDC-0084 will be known as ‘paxalisib’ Internal compound code International Non-proprietary Name (INN) Commercial Brand Name Selected by company once drug begins journey to clinical trials Awarded by WHO, usually around phase 1 / 2 clinical trials Selected by FDA after recommendation from company at time of marketing application GDC-0084 paxalisib The award of an INN is an important milestone in the development of a drug and provides an industry-standard, recognisable name for the drug moving forward

Next step for paxalisib is a pivotal study for registration Market Phase 3 Phase 2 Phase 1 Preclinical 4-7 years 8-10 years Paxalisib is here Upcoming SNO data read-out will be a key check-point before committing to phase 3 Kazia expects to share more detail on phase 3 plans early in CY2020, pending ongoing partnering discussions

In the meantime, important new data will be presented at the upcoming SNO conference Phase II Trial in Glioblastoma Phase I Trial in Advanced Glioma poster presentation oral presentation Lead Author: Professor Patrick Wen Dana-Farber Cancer Institute Lead Author: Professor Ben Ellingson UCLA Imaging Laboratory 2019 Annual Meeting Phoenix, AZ, USA 20 – 24 November 2019 1 2 Initial interim efficacy data from Kazia’s ongoing phase 2 study of paxalisib in glioblastoma Cutting edge re-analysis of imaging data from Genentech’s phase 1 study in 2016 This data is a critical read-out for the program
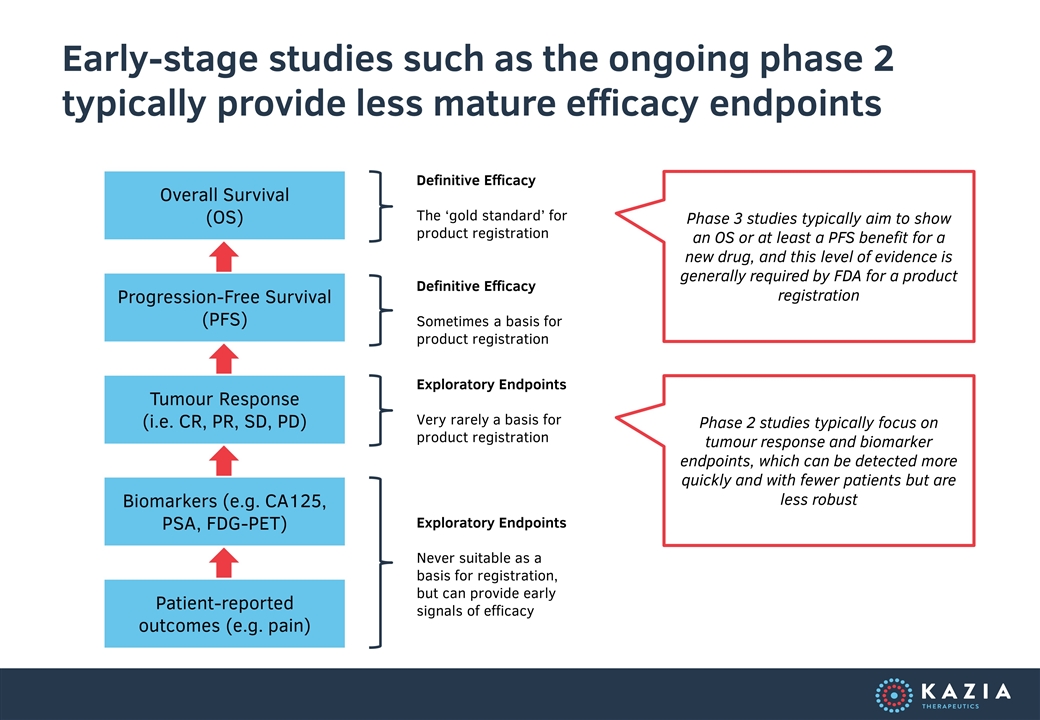
Early-stage studies such as the ongoing phase 2 typically provide less mature efficacy endpoints Patient-reported outcomes (e.g. pain) Biomarkers (e.g. CA125, PSA, FDG-PET) Tumour Response (i.e. CR, PR, SD, PD) Progression-Free Survival (PFS) Overall Survival (OS) Exploratory Endpoints Never suitable as a basis for registration, but can provide early signals of efficacy Exploratory Endpoints Very rarely a basis for product registration Definitive Efficacy Sometimes a basis for product registration Definitive Efficacy The ‘gold standard’ for product registration Phase 2 studies typically focus on tumour response and biomarker endpoints, which can be detected more quickly and with fewer patients but are less robust Phase 3 studies typically aim to show an OS or at least a PFS benefit for a new drug, and this level of evidence is generally required by FDA for a product registration
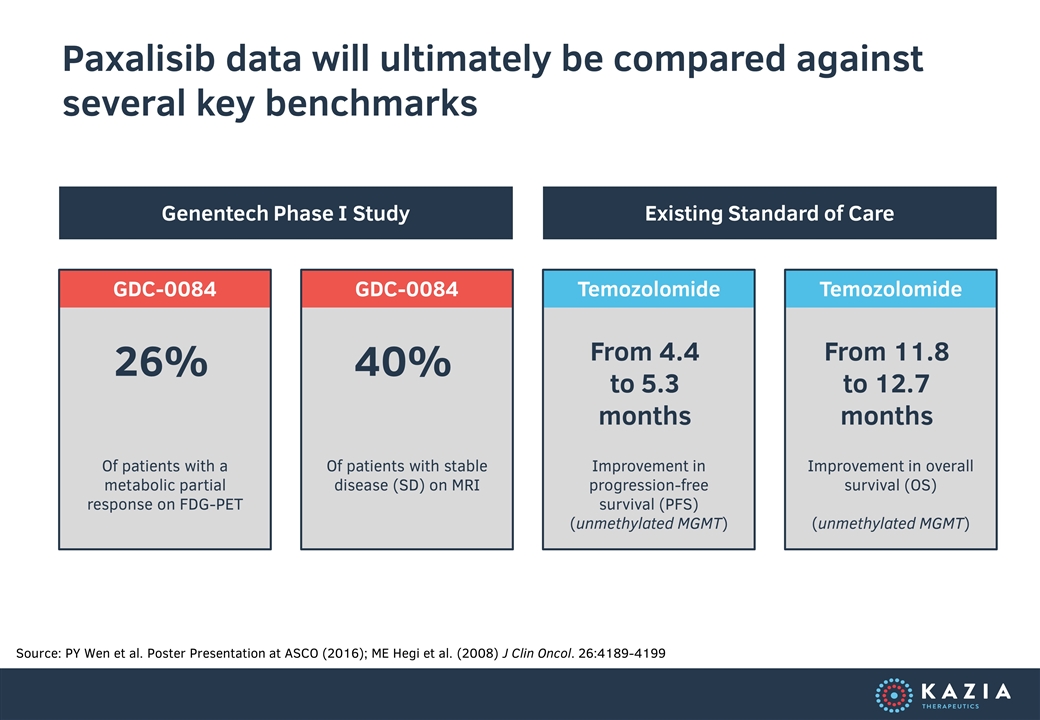
GDC-0084 GDC-0084 Temozolomide Temozolomide Paxalisib data will ultimately be compared against several key benchmarks Source: PY Wen et al. Poster Presentation at ASCO (2016); ME Hegi et al. (2008) J Clin Oncol. 26:4189-4199 26% Of patients with a metabolic partial response on FDG-PET 40% Of patients with stable disease (SD) on MRI From 4.4 to 5.3 months Improvement in progression-free survival (PFS) (unmethylated MGMT) From 11.8 to 12.7 months Improvement in overall survival (OS) (unmethylated MGMT) Genentech Phase I Study Existing Standard of Care

The partnering market for new oncology drugs is active and driven by emerging data Licensee Licensor Stage Asset(s) Deal Value (US$) Discovery Lipid kinase inhibitors $470M Preclinical Anti-CD38 antibody $275M Preclinical RAS-RAF-MAPK inhibitors $207M Clinical MEK inhibitor $700M Discovery Complement modulator $2.0B Acquirer Target Stage Asset(s) Deal Value (US$) Commercial BRAF inhibitors $11.0B Clinical HIF-2a inhibitors $2.2B Discovery Discovery platform $167M Clinical Cancer vaccine platform $367M Select CY2019 Licensing Transactions Select CY2019 M&A Transactions
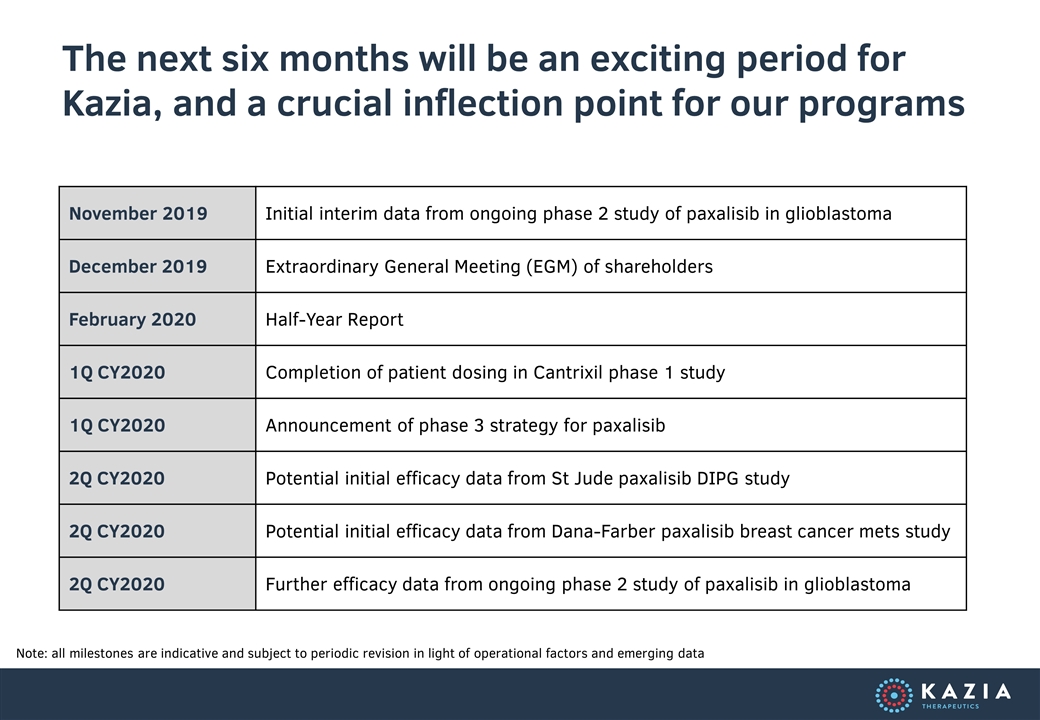
The next six months will be an exciting period for Kazia, and a crucial inflection point for our programs Note: all milestones are indicative and subject to periodic revision in light of operational factors and emerging data November 2019 Initial interim data from ongoing phase 2 study of paxalisib in glioblastoma December 2019 Extraordinary General Meeting (EGM) of shareholders February 2020 Half-Year Report 1Q CY2020 Completion of patient dosing in Cantrixil phase 1 study 1Q CY2020 Announcement of phase 3 strategy for paxalisib 2Q CY2020 Potential initial efficacy data from St Jude paxalisib DIPG study 2Q CY2020 Potential initial efficacy data from Dana-Farber paxalisib breast cancer mets study 2Q CY2020 Further efficacy data from ongoing phase 2 study of paxalisib in glioblastoma
