UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of November, 2019
Commission File Number
Kazia Therapeutics Limited
(Translation of registrant’s name into English)
Three International Towers Level 24 300 Barangaroo Avenue Sydney NSW 2000
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☑ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark if the registrant by furnishing the information contained in this form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934. Yes ☐ No ☑
If “yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b)
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Kazia Therapeutics Limited(Registrant)
Kate Hill
Kate Hill
Company Secretary
Date 3 December 2019

A company developing innovative, high-impact drugs for cancer Presentation to Switzer Small & Microcap Conference 2019 Dr James Garner Chief Executive Officer Sydney, Australia3 December 2019

Forward-Looking Statements This presentation contains “forward-looking statements” within the meaning of the “safe-harbor” provisions of the Private Securities Litigation Reform Act of 1995. Such statements involve known and unknown risks, uncertainties and other factors that could cause the actual results of the Company to differ materially from the results expressed or implied by such statements, including changes from anticipated levels of customer acceptance of existing and new products and services and other factors. Accordingly, although the Company believes that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such expectations will prove to be correct. The Company has no obligation to sales, future international, national or regional economic and competitive conditions, changes in relationships with customers, access to capital, difficulties in developing and marketing new products and services, marketing existing products and services update the forward-looking information contained in this presentation.

Investment Rationale Our lead program, GDC-0084, was designed by Genentech, and is being developed for glioblastoma, the most common form of brain cancer, where the only available drug is ineffective for two-thirds of patients Company is well-financed, following a recent institutional placement, with multiple value-driving data read-outs expected during the early part of CY 2020 and high potential to partner with big pharma 1 2 3 4 Four other clinical trials of GDC-0084 are currently underway at leading US hospitals, all primarily funded by external parties, covering a broad range of primary and secondary brain cancers to provide multiple shots on goal GDC-0084 has shown strong evidence of efficacy in an ongoing phase II human trial in the United States; a pivotal study for registration is planned to commence in CY 2020
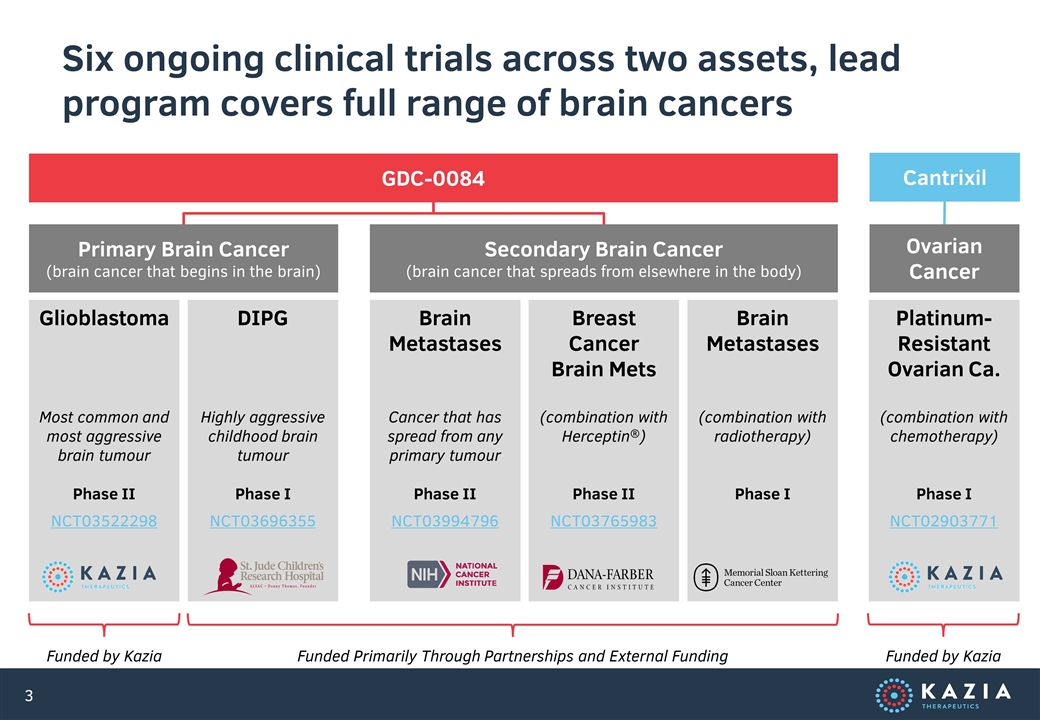
Six ongoing clinical trials across two assets, lead program covers full range of brain cancers Glioblastoma Most common and most aggressive brain tumour Phase II NCT03522298 DIPG Highly aggressive childhood brain tumour Phase I NCT03696355 Brain Metastases Cancer that has spread from any primary tumour Phase II NCT03994796 Breast Cancer Brain Mets (combination with Herceptin®) Phase II NCT03765983 Brain Metastases (combination with radiotherapy) Phase I Platinum-Resistant Ovarian Ca. (combination with chemotherapy) Phase I NCT02903771 Primary Brain Cancer (brain cancer that begins in the brain) Secondary Brain Cancer (brain cancer that spreads from elsewhere in the body) Ovarian Cancer GDC-0084 Cantrixil Funded by Kazia Funded Primarily Through Partnerships and External Funding Funded by Kazia
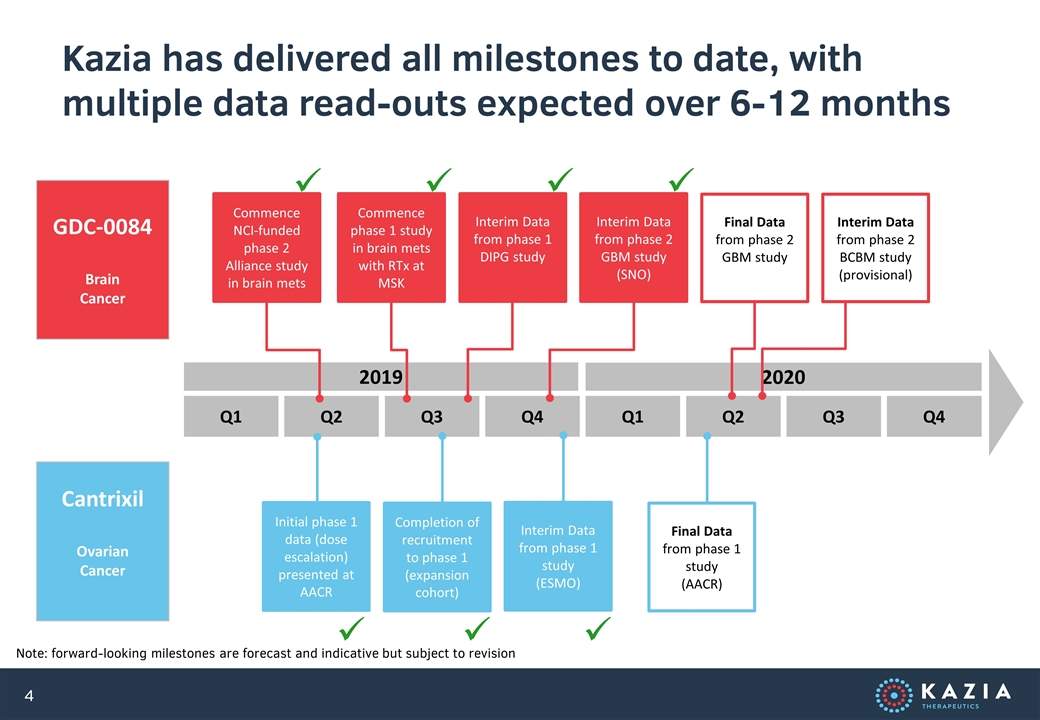
Kazia has delivered all milestones to date, with multiple data read-outs expected over 6-12 months GDC-0084 Brain Cancer Cantrixil Ovarian Cancer Initial phase 1 data (dose escalation) presented at AACR Completion of recruitment to phase 1 (expansion cohort) ü Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2019 2020 Commence NCI-funded phase 2 Alliance study in brain mets ü Commence phase 1 study in brain mets with RTx at MSK ü Interim Data from phase 2 GBM study (SNO) Interim Data from phase 1 DIPG study Final Data from phase 2 GBM study Interim Data from phase 1 study (ESMO) Note: forward-looking milestones are forecast and indicative but subject to revision Interim Data from phase 2 BCBM study (provisional) Final Data from phase 1 study (AACR) ü ü ü ü
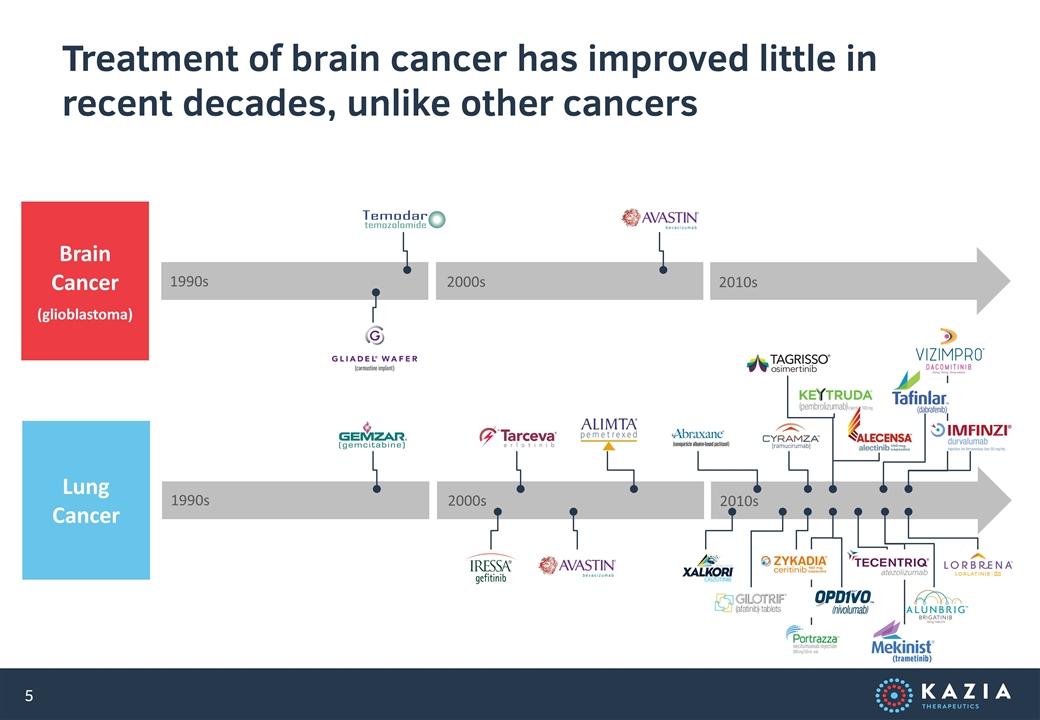
Treatment of brain cancer has improved little in recent decades, unlike other cancers Lung Cancer Brain Cancer (glioblastoma) 1990s 1990s 2000s 2000s 2010s 2010s

Glioblastoma (GBM) is the most common and most aggressive form of primary brain cancer Lung 14 million cancer cases per annum Breast Colon Prostate Stomach No clear cause or strong risk factors 3-4 months untreated survival 12-15 months average survival with treatment Any age, but most common in 60s Five-year survival 3 – 5% (breast cancer: 90%) Glioblastoma Multiforme 133,000 cases per annum worldwide Indicative Market Opportunity US$ 1.5 billion Sen. John McCain US politician Matt Price ABC journalist Stan Zemanek Media personality Andrew Olle ABC journalist Chris O’Brien, AO Surgeon
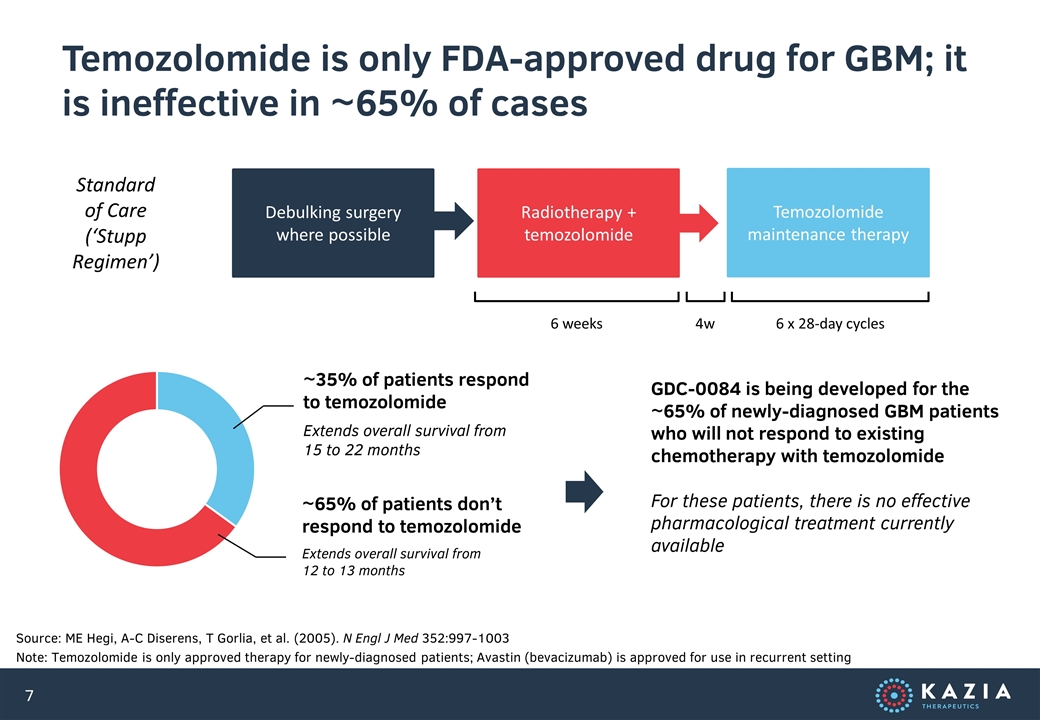
Temozolomide is only FDA-approved drug for GBM; it is ineffective in ~65% of cases Source: ME Hegi, A-C Diserens, T Gorlia, et al. (2005). N Engl J Med 352:997-1003 Note: Temozolomide is only approved therapy for newly-diagnosed patients; Avastin (bevacizumab) is approved for use in recurrent setting Standard of Care (‘Stupp Regimen’) Debulking surgery where possible Radiotherapy + temozolomide Temozolomide maintenance therapy 6 weeks 4w 6 x 28-day cycles ~35% of patients respond to temozolomide Extends overall survival from 15 to 22 months ~65% of patients don’t respond to temozolomide Extends overall survival from 12 to 13 months GDC-0084 is being developed for the ~65% of newly-diagnosed GBM patients who will not respond to existing chemotherapy with temozolomide For these patients, there is no effective pharmacological treatment currently available

PI3K class is well-validated, but GDC-0084 is unique in its ability to cross the blood-brain barrier FDA Approved July 2014 (blood cancers) [accelerated approval] FDA Approved September 2017 (blood cancers) [accelerated approval] FDA Approved October 2018 (blood cancers) [accelerated approval] ü ü ü Potentially fatal liver toxicity and diarrhoea Potentially fatal infections Potentially fatal infections & diarrhoea Does not cross blood-brain barrier Does not cross blood-brain barrier Does not cross blood-brain barrier û û û û û û Zydelig (idelalisib) Aliqopa (copanlisib) Copiktra (duvelisib) FDA Approved May 2019 (breast cancer) [accelerated approval] Limited toxicities to date Does not cross blood-brain barrier ü û ü Piqray (alpelisib) In phase II human trials under US FDA oversight (brain cancer) Appears generally safe and well-tolerated thus far Does cross blood-brain barrier ü ü GDC-0084
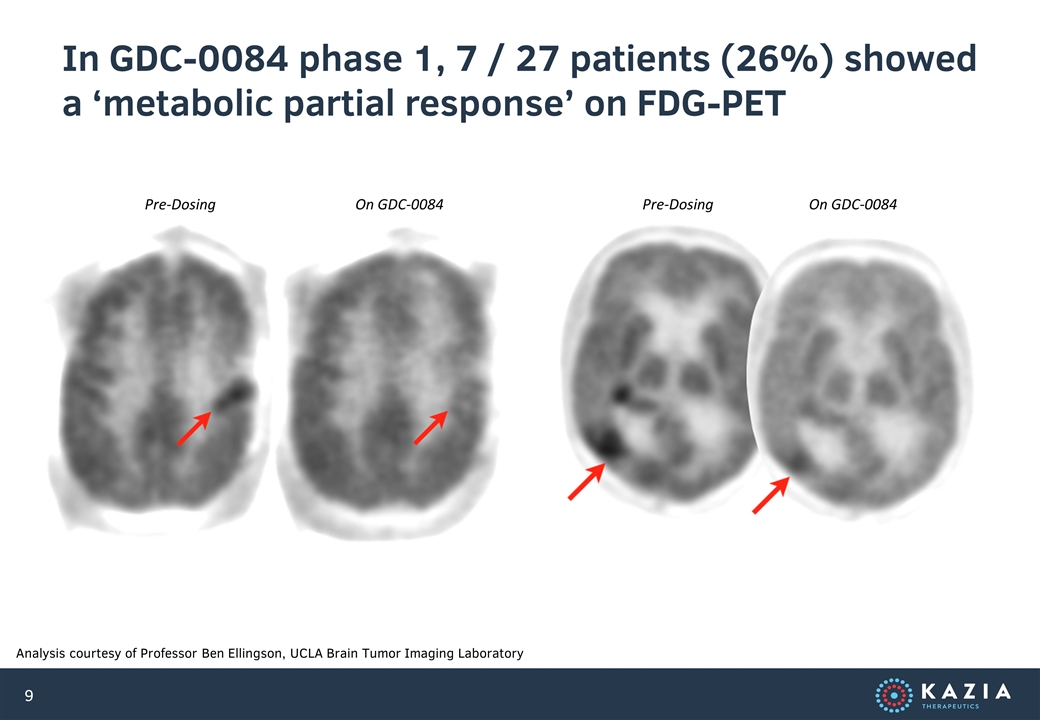
In GDC-0084 phase 1, 7 / 27 patients (26%) showed a ‘metabolic partial response’ on FDG-PET Analysis courtesy of Professor Ben Ellingson, UCLA Brain Tumor Imaging Laboratory Pre-Dosing On GDC-0084 Pre-Dosing On GDC-0084
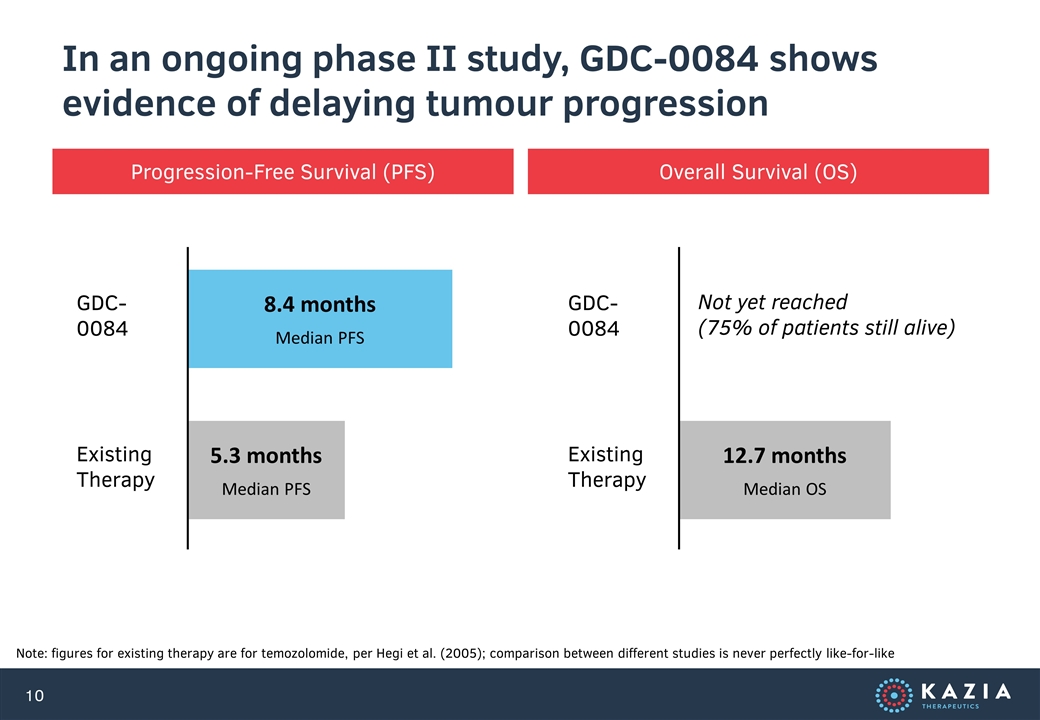
In an ongoing phase II study, GDC-0084 shows evidence of delaying tumour progression Note: figures for existing therapy are for temozolomide, per Hegi et al. (2005); comparison between different studies is never perfectly like-for-like Progression-Free Survival (PFS) Overall Survival (OS) 8.4 months Median PFS 5.3 months Median PFS GDC-0084 Existing Therapy 12.7 months Median OS GDC-0084 Existing Therapy Not yet reached (75% of patients still alive)
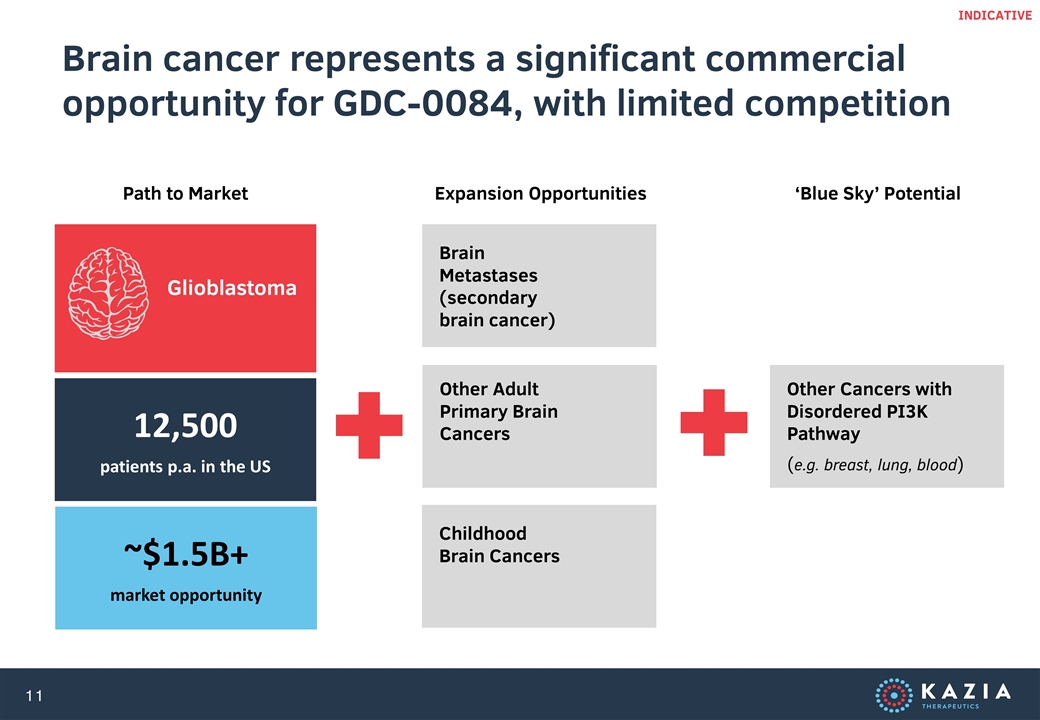
Brain cancer represents a significant commercial opportunity for GDC-0084, with limited competition ~$1.5B+ market opportunity INDICATIVE Glioblastoma 12,500 patients p.a. in the US Brain Metastases (secondary brain cancer) Childhood Brain Cancers Other Adult Primary Brain Cancers Other Cancers with Disordered PI3K Pathway (e.g. breast, lung, blood) Path to Market Expansion Opportunities ‘Blue Sky’ Potential
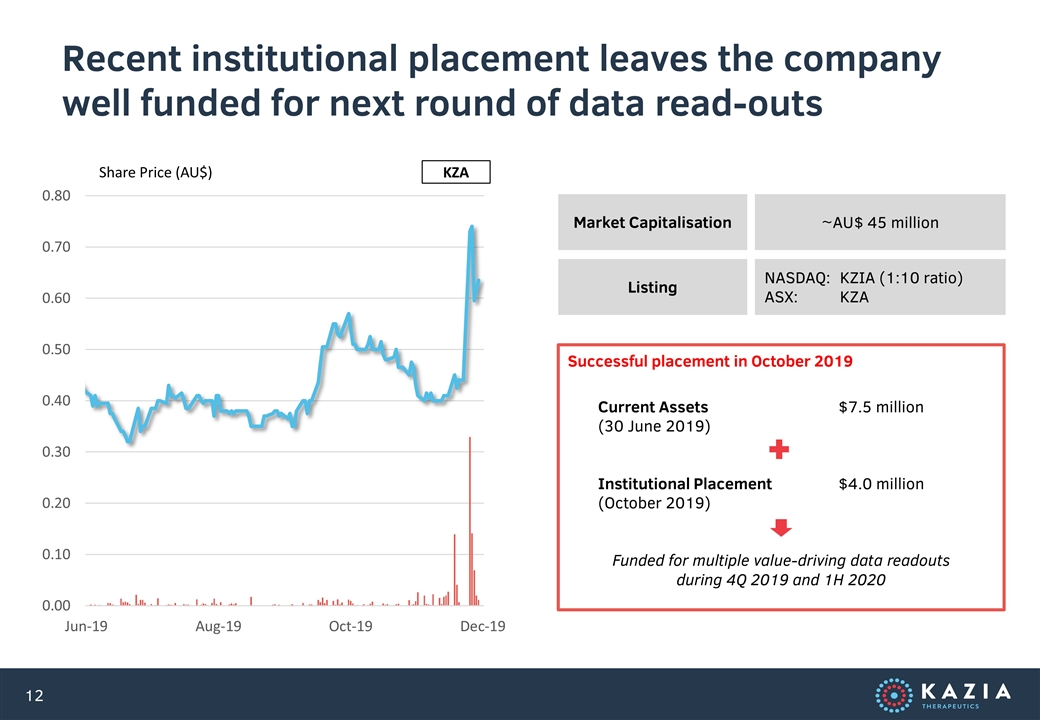
Recent institutional placement leaves the company well funded for next round of data read-outs Market Capitalisation ~AU$ 45 million Listing NASDAQ: KZIA (1:10 ratio) ASX: KZA Share Price (AU$) KZA Current Assets$7.5 million (30 June 2019) Institutional Placement$4.0 million (October 2019) Funded for multiple value-driving data readouts during 4Q 2019 and 1H 2020 Successful placement in October 2019

The partnering market for new oncology drugs is active and driven by emerging data Licensee Licensor Stage Asset(s) Deal Value (US$) Discovery Lipid kinase inhibitors $470M Preclinical Anti-CD38 antibody $275M Preclinical RAS-RAF-MAPK inhibitors $207M Clinical MEK inhibitor $700M Discovery Complement modulator $2.0B Acquirer Target Stage Asset(s) Deal Value (US$) Commercial BRAF inhibitors $11.0B Clinical HIF-2a inhibitors $2.2B Discovery Discovery platform $167M Clinical Cancer vaccine platform $367M Select CY2019 Licensing Transactions Select CY2019 M&A Transactions
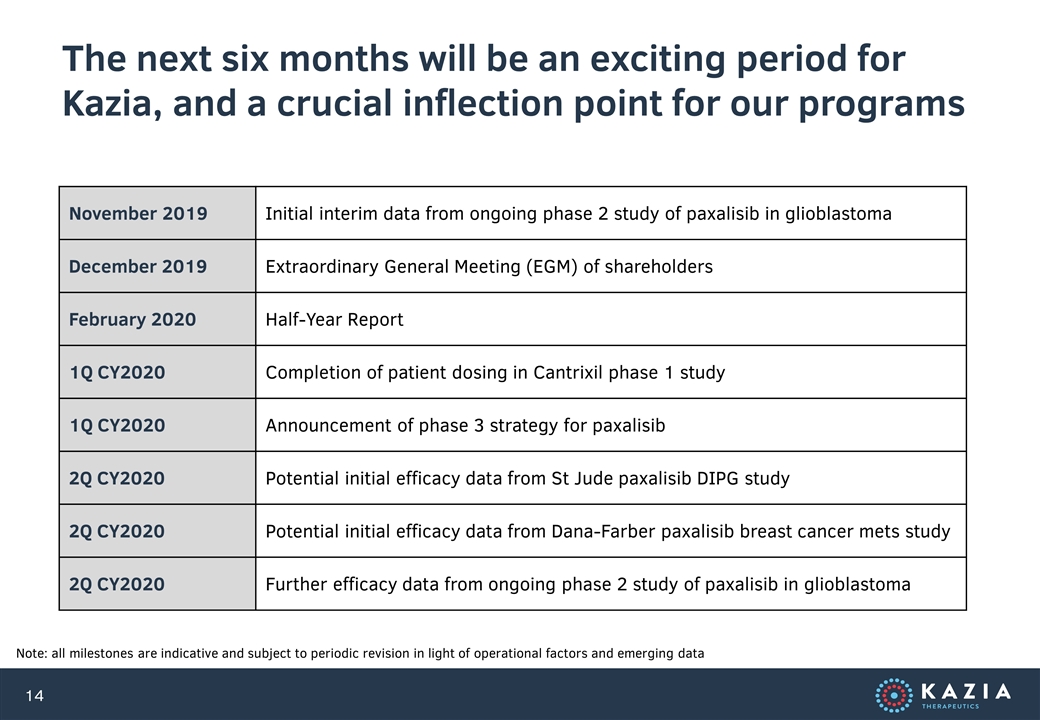
The next six months will be an exciting period for Kazia, and a crucial inflection point for our programs Note: all milestones are indicative and subject to periodic revision in light of operational factors and emerging data November 2019 Initial interim data from ongoing phase 2 study of paxalisib in glioblastoma December 2019 Extraordinary General Meeting (EGM) of shareholders February 2020 Half-Year Report 1Q CY2020 Completion of patient dosing in Cantrixil phase 1 study 1Q CY2020 Announcement of phase 3 strategy for paxalisib 2Q CY2020 Potential initial efficacy data from St Jude paxalisib DIPG study 2Q CY2020 Potential initial efficacy data from Dana-Farber paxalisib breast cancer mets study 2Q CY2020 Further efficacy data from ongoing phase 2 study of paxalisib in glioblastoma

Investment Rationale Our lead program, GDC-0084, was designed by Genentech, and is being developed for glioblastoma, the most common form of brain cancer, where the only available drug is ineffective for two-thirds of patients Company is well-financed, following a recent institutional placement, with multiple value-driving data read-outs expected during the early part of CY 2020 and high potential to partner with big pharma 1 2 3 4 Four other clinical trials of GDC-0084 are currently underway at leading US hospitals, all primarily funded by external parties, covering a broad range of primary and secondary brain cancers to provide multiple shots on goal GDC-0084 has shown strong evidence of efficacy in an ongoing phase II human trial in the United States; a pivotal study for registration is planned to commence in CY 2020

www.kaziatherapeutics.com