As filed with the Securities and Exchange Commission on June 1, 2010
Registration Statement No. 333-____________
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER THE SECURITIES ACT OF 1933

SKYSTAR BIO-PHARMACEUTICAL COMPANY
(Exact Name of Registrant in Its Charter)
| Nevada | 2834 | 33-0901534 |
| (State or Other Jurisdiction of | (Primary Standard Industrial | (I.R.S. Employer |
| Incorporation or Organization) | Classification Code Number) | Identification Number) |
Rm. 10601, Jiezuo Plaza, No. 4, Fenghui Road South,
Gaoxin District, Xi’an, Shaanxi Province, P.R. China
(8629) 8819-3188
(Address, Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive Offices)
Paracorp Incorporated
318 North Carson Street #208
Carson City, NV 89701
(Name, Address, Including Zip Code, and Telephone Number, Including Area Code, of Agent for Service)
Copies to:
| Kevin K. Leung, Esq. | Harvey J. Kesner, Esq. |
| Francis Y.L. Chen, Esq. | Benjamin S. Reichel, Esq. |
| Rahul P. Dange, Esq. | Jeffrey J. Fessler, Esq. |
| RICHARDSON & PATEL LLP | SICHENZIA ROSS FRIEDMAN FERENCE LLP |
| 10900 Wilshire Boulevard, Suite 500 | 61 Broadway, Suite 3200 |
| Los Angeles, California 90024 | New York, New York 10006 |
| (310) 208-1182 | (212) 930-9700 |
Approximate date of commencement of proposed sale to the public: from time to time after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. x
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a smaller reporting company. See definitions of large accelerated filer, accelerated filer, and smaller reporting company, in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer ¨ | Accelerated filer ¨ |
Non-accelerated filer ¨ (Do not check if a smaller reporting company) | Smaller reporting company x |
CALCULATION OF REGISTRATION FEE
| Title of Each Class of Securities to Be Registered | Amount to Be Registered | Proposed Maximum Aggregate Offering Price(1) | Amount of Registration Fee | |||||||
Shares of Common Stock, $.001 par value (2) | $ | 28,750,000 | $ | 2,049.88 | ||||||
Underwriter’s Common Stock Purchase Option (3) (4) | 1 option | $ | 100 | $ | - | |||||
| Shares of Common Stock underlying Underwriter’s Common Stock Purchase Option | $ | 781,250 | $ | 55.70 | ||||||
| Total Registration Fee | $ | 2,105.58 | ||||||||
| (1) | Estimated solely for the purpose of calculating the registration fee pursuant to Rule 457(o) under the Securities Act. |
| (2) | Includes __________ shares of common stock which may be issued pursuant to the exercise of a 45-day option granted by the registrant to the underwriters to cover over-allotments, if any. |
| (3) | No registration fee required pursuant to Rule 457(g) under the Securities Act of 1933. |
| (4) | Pursuant to Rule 416 under the Securities Act of 1933, this registration statement shall be deemed to cover the additional securities (i) to be offered or issued in connection with any provision of any securities purported to be registered hereby to be offered pursuant to terms which provide for a change in the amount of securities being offered or issued to prevent dilution resulting from stock splits, stock dividends, or similar transactions and (ii) of the same class as the securities covered by this registration statement issued or issuable prior to completion of the distribution of the securities covered by this registration statement as a result of a split of, or a stock dividend on, the registered securities. |
THE REGISTRANT HEREBY AMENDS THIS REGISTRATION STATEMENT ON SUCH DATE OR DATES AS MAY BE NECESSARY TO DELAY ITS EFFECTIVE DATE UNTIL THE REGISTRANT SHALL FILE A FURTHER AMENDMENT WHICH SPECIFICALLY STATES THAT THIS REGISTRATION STATEMENT SHALL THEREAFTER BECOME EFFECTIVE IN ACCORDANCE WITH SECTION 8(a) OF THE SECURITIES ACT OR UNTIL THE REGISTRATION STATEMENT SHALL BECOME EFFECTIVE ON SUCH DATE AS THE SECURITIES AND EXCHANGE COMMISSION, ACTING PURSUANT TO SECTION 8(a), MAY DETERMINE.
The information in this preliminary prospectus is not complete and may be changed. A registration statement relating to these securities has been filed with the Securities and Exchange Commission. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any state or other jurisdiction where the offer or sale is not permitted.
| PRELIMINARY PROSPECTUS | SUBJECT TO COMPLETION, DATED JUNE 1, 2010 |
Shares

SKYSTAR BIO-PHARMACEUTICAL COMPANY
This is a firm commitment public offering of shares of our common stock.
Our common stock is traded on The NASDAQ Capital Market under the symbol “SKBI”. The last reported market price of our common stock on May 28, 2010 was $8.71 per share.
Investing in our common stock involves a high degree of risk. See “Risk Factors” beginning on page 9 of this prospectus for a discussion of information that should be considered in connection with an investment in our common stock.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
| Per Share | Total | |||||||
| Public offering price | $ | $ | ||||||
| Underwriting discounts and commissions (1) | $ | $ | ||||||
| Proceeds, before expenses, to us | $ | $ | ||||||
| (1) | See “Underwriting” for a description of compensation payable to the underwriter. |
We have granted a 45-day option to Rodman & Renshaw, LLC, the underwriter, to purchase up to an additional __________ shares of common stock from us on the same terms as set forth above. If the underwriter exercises its right to purchase all of such additional shares of common stock, we estimate that we will receive gross proceeds of approximately $____ million from the sale of shares being offered and net proceeds of approximately $____ million after deducting approximately $____ million for underwriting discounts and commissions, based on a public offering price of $___ per share. The shares issuable upon exercise of the underwriter option are identical to those offered by this prospectus and have been registered under the registration statement of which this prospectus forms a part.
The underwriter expects to deliver the common stock to purchasers in the offering on or about ______, 2010.
| Rodman & Renshaw, LLC | JMP Securities |
The date of this prospectus is ____________ ___, 2010.

(1) Our office building located in Huxian, Xi’an, Shaanxi Province, People’s Republic of China.
(2), (3) and (4) A selection of our veterinary drug products made in Huxian.
TABLE OF CONTENTS
| Prospectus Summary | 3 |
| Risk Factors | 9 |
| Special Note Regarding Forward Looking Statement | 20 |
| Use of Proceeds | 21 |
| Market for Common Equity and Related Stockholder Matters | 22 |
| Dividend Policy | 23 |
| Capitalization | 23 |
| Dilution | 24 |
| Management’s Discussion and Analysis of Financial Condition and Results of Operations | 25 |
| Business | 33 |
| Management | 43 |
| Executive Compensation | 46 |
| Security Ownership of Certain Beneficial Owners and Management | 49 |
| Certain Relationships and Related Transactions | 50 |
| Description of Securities | 52 |
| Underwriting | 56 |
| Legal Matters | 62 |
| Experts | 62 |
| Disclosure of Commission Position of Indemnification for Securities Act Liabilities | 62 |
| Where You Can Find More Information | 63 |
| Index to Financial Statements | F-1 |
You should rely only on the information contained or incorporated by reference to this prospectus in deciding whether to purchase our common stock. We have not authorized anyone to provide you with information different from that contained or incorporated by reference to this prospectus. Under no circumstances should the delivery to you of this prospectus or any sale made pursuant to this prospectus create any implication that the information contained in this prospectus is correct as of any time after the date of this prospectus. To the extent that any facts or events arising after the date of this prospectus, individually or in the aggregate, represent a fundamental change in the information presented in this prospectus, this prospectus will be updated to the extent required by law.
We obtained statistical data, market data and other industry data and forecasts used throughout this prospectus from market research, publicly available information and industry publications. Industry publications generally state that they obtain their information from sources that they believe to be reliable, but they do not guarantee the accuracy and completeness of the information. Nevertheless, we are responsible for the accuracy and completeness of the historical information presented in this prospectus, as of the date of the prospectus.
2
PROSPECTUS SUMMARY
This summary highlights information contained elsewhere in this prospectus. It does not contain all of the information that you should consider before investing in our securities. You should read the entire prospectus carefully, including the section entitled “Risk Factors” and our consolidated financial statements and the related notes. In this prospectus, we refer to Skystar Bio-Pharmaceutical Company as “Skystar” and “our Company,” and to Skystar, its subsidiaries and Xian Tianxing, a company which conducts substantially all our business through contractual arrangements and in which neither we nor our subsidiaries own any equity interests, sometimes collectively as “we,” “us” and “our.”
Overview
We are a holding company that, through our indirect wholly-owned subsidiary Sida Biotechnology (Xian) Co., Ltd. (“Sida”) and our variable interest entity (“VIE”) Xian Tianxing Bio-Pharmaceutical Co., Ltd. (“Xian Tianxing”), manufactures and distributes medicines, vaccines and other health care and medical care products for poultry, livestock and domestic pets in the People’s Republic of China, which we will refer to as China. We have four product lines, including a vaccine line, a veterinary medicine line, a fodder and feed additives line, and a micro-organism line. All four product lines are developed, manufactured and distributed by Xian Tianxing, which we operate and control through contractual arrangements between Sida and Xian Tianxing. These contractual arrangements enable Sida to control and receive the profits of Xian Tianxing. Sida is the wholly owned subsidiary of Fortunate Time International Ltd. (“Fortunate Time”), which is wholly owned by Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”), our wholly owned subsidiary. Xian Tianxing has a wholly owned subsidiary, Shanghai Siqiang Biotechnological Co., Ltd. (“Shanghai Siqiang”), which we also control. Other than our interests in the contractual arrangements with Xian Tianxing, neither we nor our direct and indirect subsidiaries have any equity interests in Xian Tianxing.
Management believes there is significant demand for veterinary medicines and vaccines in China. According to the Chinese Ministry of Agriculture, the addressable market in China in 2004 for veterinary, livestock and poultry vaccines was over 70 billion doses; however the market supply was only 32 billion doses. The estimated 2005 expenditures in China on products designed to prevent parasitic diseases in livestock and poultry, such as our medicines and vaccines, were US$125 million, as reported in the 2005 China Statistical Yearbook published by the National Bureau of Statistics of China, a government agency. According to the Chinese Ministry of Agriculture, the demand for vaccines for livestock and poultry has increased at a 15% annual rate since 2002 and is anticipated to maintain such growth rate for the next five years.
We also believe that there is a substantial market for micro-organisms which are fed to animals and result in healthier livestock and reduced feed requirements for our customers. According to the Chinese Ministry of Agriculture, the addressable market in China in 2004 for such micro-organisms was 3 million tons, while the supply output was only 200,000 tons.
Our Business
All of our revenue is derived from the sale of veterinary healthcare and medical care products in China. Currently, we have four major product lines:
| · | Our vaccine line currently includes 10 products; |
| · | Our veterinary medicine line for poultry and livestock currently includes 159 products; |
| · | Our fodder and feed additives line currently includes 10 products; and |
| · | Our micro-organism products line currently includes 16 products. |
Our top selling product is Praziquantel tablets. It is an anthelmintic effective against schistosomiasis. Schistosomiasis is a parasitic disease that affects both animals and humans. Our product only treats animals. Schistosomiasis is prevalent in Southern China where there are a lot of fresh water lakes and rivers. The company has seen tremendous growth in the sales of the Praziquantel tablets. Of total revenues from veterinary medications during the three months March 31, 2010, approximately $830,600, or 17% of total revenues, resulted from the sale of Praziquantel tablets. Another prominent product of ours is a vaccine that is designed to prevent the onset of avian coccidiosis disease, a parasitic and highly contagious gastrointestinal disease affecting chicken and other poultry. We refer to this vaccine as the “DLV chicken vaccine.” Avian coccidiosis has a significant economic impact to the poultry industry. The U.S. Department of Agriculture estimates that this disease costs the worldwide poultry industry $3 billion in treatment expenses, bird losses and unmarketable birds due to low bird weight. Management believes that our DLV chicken vaccine is safe, effective and easy to administer, and may save costs by as much as 60% as compared to using conventional chemical medicines such as sulfaquinoxaline sodium and salinomycin sodium.
During the three months ended March 31, 2010, we had revenues of $4.9 million and net income of $1.1 million as compared to revenues of $3.8 million and net income of $1.1 million during the same period ended March 31, 2009. During the year ended December 31, 2009, we had revenues of $33.8 million and net income of $8.9 million as compared to revenues of $25.6 million and net income of $5.6 million during the year ended December 31, 2008.
3
We sell our products through a distribution channel covering 29 provinces in China. As of March 31, 2010, we had over 1,539 distributors and 529 direct customers. We intend to establish more representative offices and engage additional distribution agents in order to strengthen our distribution network.
Growth Strategy
Acquisition Strategy. We intend to explore acquisitions of Good Manufacturing Practice (“GMP”) certified veterinary healthcare and medical care products companies located close to our headquarters in Shaanxi province. We estimate that up to $10 million from the net proceeds of the offering will be earmarked for potential acquisitions. One of our considerations for acquisition is that the potential target has production capacity for different dosage forms such as injectibles and powders. We may also consider acquiring veterinary healthcare and medical care products companies with similar production capacity in regions of China outside of the Shaanxi province. Our goal is to be among the top producers and distributors of veterinary health care and medical care products in China, and we hope to be able to complete some acquisitions by the end of 2010. We are looking at and making inquiries regarding potential acquisition targets.
Based on our own experience, we know that the GMP certification process requires considerable amount of resources, which may create cash flow issues for many of the small to medium-size veterinary healthcare and medical care products companies that currently make up a significant portion of our industry in China. Accordingly, unless we can accurately and effectively evaluate the financial conditions of a potential target company, we may expose our business operations to cash flow risks subsequent to an acquisition transaction.
Brand Awareness. Our goal is to associate the “Skystar” brand with reputable, high-quality products, including veterinary products. We intend to spend a portion of our net proceeds in marketing and advertising efforts to increase the exposure of our brand with potential customers. We recognize the importance of branding as well as packaging. All of our products bear a uniform brand and we also brand and package our products with specialized designs to differentiate the different categories of our products.
We also conduct promotional marketing activities to publicize and enhance our image as well as to reinforce the recognition of our brand name, including:
| · | publishing advertisements and articles in national as well as specialized and provincial newspapers, magazines, and in other media, including the Internet; |
| · | participating in national meetings, seminars, symposiums, exhibitions for veterinary healthcare and medical care products and other related industries; |
| · | organizing cooperative promotional activities with distributors; and |
| · | sending direct mail to major farms. |
Manufacturing Facilities. We intend to use approximately $10 million from the net proceeds of the offering to build out a production and research facilities in Kunshan, China for micro-organism products and pet food and nutritional supplements. Under our current plans, this facility is expected to be completed by the end of fourth quarter of 2010.
However, if this offering is delayed or ultimately unsuccessful, we may be unable to complete the facilities as planned, thereby also delaying the projected GMP certification of facilities. In addition, as the GMP certification is conducted by unrelated third parties, we cannot give assurance that our facilities will be GMP certified or when the certification can be completed.
Additional Products. We plan to commercialize new products in each of the four product lines. We are also expending significant amount of resources into our research and development in order to have additional products in the future. Management believes that such products will ultimately increase future revenue to our company. However, due to the unpredictable natures of many of the afflictions and diseases that our products are intended to address and/or prevent, we cannot give assurance as to the success of any new products, since market acceptance and demands of any such product may significant shift and adversely affect our profitability.
Research and Development
We place great emphasis on product research and development, and are currently working closely with a number of research institutes in the veterinary science field in China. We currently have ongoing projects with Shanghai Poultry Verminosis Institution, Shaanxi Microbial Institute Northwestern Agricultural Technology University, Shanxi Shenzhou Bio-pharmaceuticals Technology Company and the Fourth Military Medical University.
4
However, we cannot provide assurance that any one of the ongoing projects will ultimately be successful or commercially viable. Additionally, we currently do not have any written agreement with Shanghai Poultry Verminosis Institution regarding the ongoing collaborations, and under our cooperation agreement with Shaanxi Microbial Institute, the Institute is not obligated to us with respect to any specific period of time or research projects. Accordingly, we cannot provide assurance that we will be able to maintain our current relationship with these institutes for any definite period of time, or establish new collaboration with other research institutes in the future. Additionally, intellectual property rights and confidentiality protections in China may not be effective as in the United States or other countries, and we cannot provide assurance that we will be able to meaningfully protect our rights in connection with our research and our products.
Our Organizational Structure
We conduct substantially all of our business operations through Xian Tianxing. Chinese law currently has limits on foreign ownership of certain businesses which prohibit non-Chinese persons from having direct ownership interests. To comply with these foreign ownership restrictions, we do not own any equity interests in Xian Tianxing or its wholly-owned subsidiary, Shanghai Siqiang, but control and receive the economic benefits of their business operations through contractual arrangements. Xian Tianxing holds the licenses and approvals necessary to operate its business in China. We have contractual arrangements with Xian Tianxing and its stockholders pursuant to which we provide technology consulting and other general business operation services to Xian Tianxing. Through these contractual arrangements, we also have the ability to substantially influence Xian Tianxing’s daily operations and financial affairs, since we are able to appoint its senior executives and approve all matters requiring stockholder approval. As a result of these contractual arrangements, which enable us to control Xian Tianxing (and through Xian Tianxing, Shanghai Siqiang) and to receive, through our direct and indirect wholly owned subsidiaries, all of Xian Tianxing’s profits, we are considered the primary beneficiary of Xian Tianxing. Accordingly, we consolidate Xian Tianxing’s results, assets and liabilities in our financial statements.
However, Chinese laws and regulations concerning the validity of the contractual arrangements are uncertain, as many of these laws and regulations are relatively new and may be subject to change, and their official interpretation and enforcement by the Chinese government may involve substantial uncertainty. Additionally, the contractual arrangements may not be as effective in providing control over Xian Tianxing as direct ownership, which we are restricted from under current Chinese law. Due to such uncertainty, we may take such additional steps in the future as may be permitted by the then applicable law and regulations in China to further strengthen our control over or toward actual ownership of Xian Tianxing or its assets or business operations, which could include direct ownership of selected assets without jeopardizing any favorable government policies toward domestic owned enterprises. Because we rely on Xian Tianxing for our revenue, any termination of or disruption to these contractual arrangements could detrimentally affect our business.
The contractual arrangements were entered into in October 2005 between Skystar Cayman and Xian Tianxing. In March 2008, Skystar Cayman assigned its rights under the contractual arrangements to our indirect wholly-owned subsidiary, Sida, a limited liability company organized under the laws of China and a wholly foreign-owned enterprise. All ownership interests of Sida are owned by Fortunate Time, a company incorporated under the laws of Hong Kong Special Administration Region of China. Fortunate Time is a wholly-owned subsidiary of Skystar Cayman. Set forth below is our current corporate structure:
Corporate Organizational Chart
The following chart illustrates our corporate structure and the place of incorporation of each named entity at the date of this prospectus:
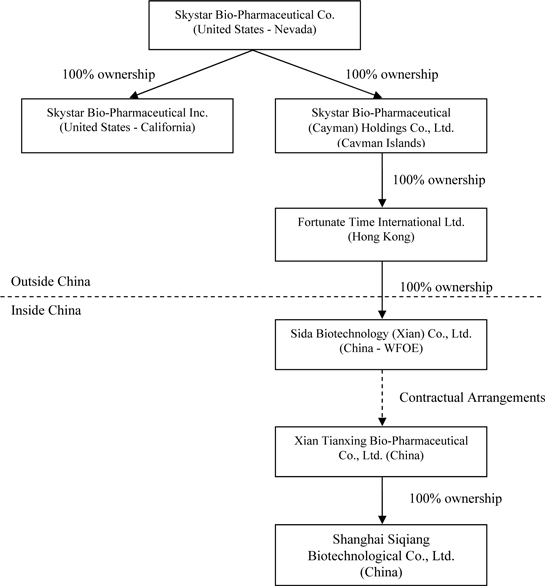
5
| (1) | The management of Skystar includes: Mr. Weibing Lu as Chairman and Chief Executive Officer, Mr. Michael H. Lan as Chief Financial Officer, and Mr. Wei Wen, Mr. R. Scott Cramer, Mr. Mark D. Chen, Mr. Qiang Fan, Dr. Shouguo Zhao and Dr. Chengtun Qu as members of the board of directors. As of the date of this prospectus: Upform Group Limited, a British Virgin Islands company of which Mr. Lu is a director, owns approximately 13.2% of Skystar’s issued and outstanding common stock; Clever Mind International Limited, a British Virgin Islands company of which Mr. Wen is director, owns approximately 0.58%; Mr. Cramer owns and/or controls approximately 2.9%; and Mr. Chen owns approximately 0.08%. Mr. Lan, Mr. Fan, Dr. Zhao and Dr. Qu do not own any shares of Skystar’s common stock as of the date of this prospectus. |
| (2) | The management of Skystar Cayman is comprised of Mr. Weibing Lu and Mr. Wei Wen as its Directors. Skystar is the sole shareholder of Skystar Cayman. |
| (3) | The management of Fortunate Time is comprised solely of Mr. Scott Cramer as its Director. Skystar Cayman is the sole shareholder of Fortunate Time. |
| (4) | The management of Sida includes: Mr. Wei Wen as General Manager, Mr. Xinya Zhang as Vice-General Manager and Director, Mr. Lun Shen as Director and Ms. Erna Gao as Supervisor. Fortunate Time is the sole shareholder of Sida. |
| (5) | Sida controls Xian Tianxing through contractual arrangements designed to mimic equity ownership of Xian Tianxing by Sida. These contracts include a consulting services agreement, operating agreement, equity pledge agreement, option agreement, and proxy agreement. Sida is a wholly-foreign owned enterprise or “WFOE.” Most foreign entities such as us control or hold ownership of Chinese enterprises indirectly through WFOEs because it eliminates the need for a Chinese partner. |
| (6) | The management of Xian Tianxing includes: Mr. Weibing Lu as Chairman and General Manager (equivalent to Chief Executive Officer), Mr. Wei Wen as Vice-General Manager and Director, Mr. Xinya Zhang as Chairman of Board of Supervisors, Ms. Erna Gao as Finance Director and Mr. Lun Shen as Supervisor. As of the date of this prospectus: Mr. Lu owns approximately 41%, and Mr. Wen approximately 5%, of the issued and outstanding stock of Xian Tianxing; Mr. Zhang, Ms. Gao and Mr. Shen do not own any equity interests in Xian Tianxing. |
| (7) | The management of Shanghai Siqiang includes: Mr. Lun Shen as Executive Director, Mr. Wei Shen as General Manager and Ms. Erna Gao as Supervisor. Xian Tianxing is the sole shareholder of Shanghai Siqiang. |
| (8) | The management of Skystar California includes Ms. Yuping Wang. |
Corporate Information
Skystar is a Nevada corporation. Our principal executive offices are located at Rm. 10601, Jiezuo Plaza, No. 4, Fenghui Road South, Gaoxin District, Xi’an, Shaanxi Province, People’s Republic of China. Our telephone number is (8629) 8819-3188.
6
THE OFFERING
| Common stock offered | __________ shares at a price of $___ per share | |
| Common stock outstanding before this offering | 7,106,705 shares (1) | |
| Common stock outstanding after this offering | _________ shares (1) | |
| Use of Proceeds | We intend to use the net proceeds of this offering for the build-out of production and research facilities in Kunshan, China for our micro-organism production line and pet food and nutritional supplements; for acquisitions of other companies; for research and development; and for working capital and general corporate purposes. | |
| NASDAQ Capital Market | ||
| Listing Symbol for Our Common Stock | SKBI | |
| Risk Factors | The securities offered by this prospectus are speculative and involve a high degree of risk and investors purchasing securities should not purchase the securities unless they can afford the loss of their entire investment. See “Risk Factors” beginning on page 9. |
| (1) | The number of shares of our common stock to be outstanding after this offering is based on the number of shares outstanding as of May 27, 2010, and excludes shares underlying the underwriter’s common stock purchase option as well as outstanding warrants, which are currently exercisable for 34,230 shares of common stock at the exercise price of $5.00 per share. |
7
SUMMARY FINANCIAL INFORMATION
In the table below we provide you with historical selected consolidated financial data for the three-month periods ended March 31, 2010 and 2009, derived from our unaudited consolidated financial statements included elsewhere in this prospectus, and for the two years ended December 31, 2009 and 2008, derived from our audited consolidated financial statements included elsewhere in this prospectus. Historical results are not necessarily indicative of the results that may be expected for any future period. When you read this historical selected financial data, it is important that you read along with it the appropriate historical consolidated financial statements and related notes and “Management's Discussion and Analysis of Financial Condition and Results of Operations” included elsewhere in this prospectus.
Three Months Ended March 31, | Fiscal Year Ended December 31, | |||||||||||||||
| 2010 | 2009 | 2009 | 2008 | |||||||||||||
| (Unaudited) | ||||||||||||||||
| Statements of Operations Data | ||||||||||||||||
| Revenue | $ | 4,869,243 | $ | 3,823,566 | $ | 33,778,305 | $ | 25,584,446 | ||||||||
| Cost of Revenue | 2,291,219 | 1,946,358 | 16,520,989 | 12,808,896 | ||||||||||||
| Gross Profit | 2,578,024 | 1,877,208 | 17,257,316 | 12,775,550 | ||||||||||||
| Operating Expenses | ||||||||||||||||
| Research and development | 43,995 | 117,352 | 1,167,937 | 549,236 | ||||||||||||
| Selling expenses | 171,134 | 207,395 | 1,928,441 | 1,381,807 | ||||||||||||
| General and administrative expenses | 619,550 | 314,695 | 2,466,470 | 2,663,520 | ||||||||||||
| Income From Operations | 1,743,345 | 1,237,766 | 11,694,468 | 8,180,987 | ||||||||||||
| Other Income (Expense), Net | (321,779 | ) | 38,398 | (813,162 | ) | (1,055,116 | ) | |||||||||
| Income (Loss) Before Taxes | 1,421,566 | 1,276,164 | 10,881,306 | 7,125,871 | ||||||||||||
| Provision For Income Taxes | 325,319 | 211,520 | 2,029,374 | 1,529,688 | ||||||||||||
| Net Income (Loss) | $ | 1,096,247 | $ | 1,064,644 | $ | 8,851,932 | $ | 5,596,183 | (1) | |||||||
| Other Comprehensive Income | ||||||||||||||||
| Foreign currency translation adjustment | (40,816 | ) | (38,448 | ) | 13,914 | 1,415,005 | ||||||||||
| Comprehensive Income | $ | 1,055,431 | $ | 1,026,196 | $ | 8,865,846 | $ | 7,011,188 | ||||||||
| Earnings Per Common Share Data | ||||||||||||||||
Basic (2) | $ | 0.16 | $ | 0.29 | $ | 1.65 | $ | 1.53 | ||||||||
Diluted (2) | $ | 0.15 | $ | 0.29 | $ | 1.62 | $ | 1.53 | ||||||||
| (1) | Includes $1,040,036 in non-cash financing costs and non-cash discount on debentures in connection with our February 2007 financing transaction for the year ended December 31, 2008. Please see “Consolidated Statements of Cash Flows for the Year ended December 31, 2009 and 2008” on page F-6 for details. |
| (2) | The impact of the 1-for-10 reverse stock split effected on May 12, 2009 and the 2-for-1 forward stock split effected on November 16, 2009, as applicable, have been applied retroactively. |
| March 31, 2010 | ||||||||
| Actual | As Adjusted (1) | |||||||
| Balance Sheet Data: | ||||||||
| Cash and Restricted Cash | $ | 5,911,415 | $ | |||||
| Working Capital | $ | 22,763,967 | $ | |||||
| Total Assets | $ | 62,302,313 | $ | |||||
| Total Liabilities | $ | 5,947,550 | $ | |||||
| (1) | As adjusted to give effect to the sale of _______ shares of common stock in this offering at a public offering price of $________, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
8
RISK FACTORS
You should carefully consider the risks described below together with all of the other information included in this prospectus before making an investment decision with regard to our securities. The statements contained in or incorporated into this offering that are not historic facts are forward-looking statements that are subject to risks and uncertainties that could cause actual results to differ materially from those set forth in or implied by forward-looking statements. If any of the following risks actually occurs, our business, financial condition or results of operations could be harmed. In that case, the trading price of our common stock could decline, and you may lose all or part of your investment.
Risks Relating to Our Business
Our relatively limited operating history makes it difficult to evaluate our future prospects and results of operations.
We have a relatively limited operating history. Xian Tianxing, the variable interest entity through which we operate our business, commenced operations in 1997 and first achieved profitability in the quarter ended September 30, 1999. Accordingly, you should consider our future prospects in light of the risks and uncertainties typically experienced by companies such as ours in evolving industries such as the bio-pharmaceutical industry in China. Some of these risks and uncertainties relate to our ability to:
| · | offer new and innovative products to attract and retain a larger customer base; |
| · | attract additional customers and increase spending per customer; |
| · | increase awareness of our brand and continue to develop user and customer loyalty; |
| · | raise sufficient capital to sustain and expand our business; |
| · | maintain effective control of our costs and expenses; |
| · | respond to changes in our regulatory environment; |
| · | respond to competitive market conditions; |
| · | manage risks associated with intellectual property rights; |
| · | attract, retain and motivate qualified personnel; |
| · | upgrade our technology to support additional research and development of new products; and |
| · | maintain or improve our position as one of the market leaders in China. |
If we are unsuccessful in addressing any of these risks and uncertainties, our business may be materially and adversely affected.
If we fail to obtain additional financing we will be unable to execute our business plan.
Despite our recent financing, we may need additional funds to build new production facilities; pursue further research and development; obtain regulatory approvals; file, prosecute, defend and enforce our intellectual property rights; and market our products. Should such needs arise, we intend to seek additional funds through public or private equity or debt financing, strategic transactions and/or from other sources.
There are no assurances that future funding will be available on favorable terms or at all. If additional funding is not obtained, we will need to reduce, defer or cancel development programs, planned initiatives or overhead expenditures, to the extent necessary. The failure to fund our capital requirements would have a material adverse effect on our business, financial condition and results of operations.
Our business will be materially and adversely affected if our collaborative partners, licensees and other third parties over whom we are very dependent fail to perform as expected.
Due to the complexity of the process of developing bio-pharmaceuticals, our core business depends on arrangements with bio-pharmaceutical institutes, corporate and academic collaborators, licensors, licensees and others for the research, development, clinical testing, technology rights, manufacturing, marketing and commercialization of our products. We have various research collaborations and outsource other business functions. Our license agreements could obligate us to diligently bring potential products to market, make substantial milestone payments and royalties and incur the costs of filing and prosecuting patent applications. There are no assurances that we will be able to establish or maintain collaborations that are important to our business on favorable terms, or at all. We could enter into collaborative arrangements for the development of particular products that may lead to our relinquishing some or all rights to the related technology or products. A number of risks arise from our dependence on collaborative agreements with third parties. Product development and commercialization efforts could be adversely affected if any collaborative partner:
| · | terminates or suspends its agreement or arrangement with us; |
| · | causes delays; |
| · | fails to timely develop or manufacture in adequate quantities a substance needed in order to conduct clinical trials; |
9
| · | fails to adequately perform clinical trials; |
| · | determines not to develop, manufacture or commercialize a product to which it has rights; or |
| · | otherwise fails to meet its contractual obligations. |
Our collaborative partners could pursue other technologies or develop alternative products that could compete with the products we are developing.
Our products will be adversely affected if we are unable to protect proprietary rights or operate without infringing the proprietary rights of others.
The profitability of our products will depend in part on our ability to obtain and maintain patents and licenses and preserve trade secrets, and the period our intellectual property remains exclusive. We must also operate without infringing the proprietary rights of third parties and without third parties circumventing our rights. The patent positions of bio-pharmaceutical and biotechnology enterprises, including ours, are uncertain and involve complex legal and factual questions for which important legal principles are largely unresolved. For example, no consistent policy has emerged regarding the breadth of biotechnology patent claims that are granted by the U.S. Patent and Trademark Office or enforced by the U.S. federal courts. In addition, the scope of the originally claimed subject matter in a patent application can be significantly reduced before a patent is issued. The biotechnology patent situation outside the U.S. is even more uncertain, is currently undergoing review and revision in many countries, and may not protect our intellectual property rights to the same extent as the laws of the U.S. Because patent applications are maintained in secrecy in some cases, we cannot be certain that we or our licensors are the first creators of inventions described in our pending patent applications or patents or the first to file patent applications for such inventions.
Other companies may independently develop similar products and design around any patented products we develop. We cannot assure you that:
| · | any of our patent applications will result in the issuance of patents; |
| · | we will develop additional patentable products; |
| · | patents we have been issued will provide us with any competitive advantages; |
| · | the patents of others will not impede our ability to do business; or |
| · | third parties will not be able to circumvent our patents. |
A number of pharmaceutical, biotechnology, research and academic companies and institutions have developed technologies, filed patent applications or received patents on technologies that may relate to our business. If these technologies, applications or patents conflict with ours, the scope of our current or future patents could be limited or our patent applications could be denied. Our business may be adversely affected if competitors independently develop competing technologies, especially if we do not obtain, or obtain only narrow, patent protection. If patents that cover our activities are issued to other companies, we may not be able to obtain licenses at a reasonable cost, or at all; develop our technology; or introduce, manufacture or sell the products we have planned.
Patent litigation is becoming widespread in the biotechnology industry. Such litigation may affect our efforts to form collaborations, to conduct research or development, to conduct clinical testing or to manufacture or market any products under development. There are no assurances that our patents would be held valid or enforceable by a court or that a competitor’s technology or product would be found to infringe our patents in the event of patent litigation. Our business could be materially affected by an adverse outcome to such litigation. Similarly, we may need to participate in interference proceedings declared by the U.S. Patent and Trademark Office or equivalent international authorities to determine priority of invention. We could incur substantial costs and devote significant management resources to defend our patent position or to seek a declaration that another company’s patents are invalid.
Much of our know-how and technology may not be patentable, though it may constitute trade secrets. There are no assurances that we will be able to meaningfully protect our trade secrets. We cannot assure you that any of our existing confidentiality agreements with employees, consultants, advisors or collaborators will provide meaningful protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure. Collaborators, advisors or consultants may dispute the ownership of proprietary rights to our technology, for example by asserting that they developed the technology independently.
Difficulties in manufacturing our products could have a material adverse effect on our profitability.
Before our products can be profitable, they must be produced in commercial quantities in a cost-effective manufacturing process that complies with regulatory requirements, including China’s Good Manufacturing Practice (GMP), production and quality control regulations. If we cannot arrange for or maintain commercial-scale manufacturing on acceptable terms, or if there are delays or difficulties in the manufacturing process, we may not be able to conduct clinical trials, obtain regulatory approval or meet demand for our products.
10
Failure or delays in obtaining an adequate amount of raw material or other supplies would materially and adversely affect our revenue.
Production of our products could require raw materials which are scarce or which can be obtained only from a limited number of sources. If we are unable to obtain adequate supplies of such raw materials, the development, regulatory approval and marketing of our products could be delayed.
Our ability to generate more revenue would be adversely affected if we need more clinical trials or take more time to complete our clinical trials than we have planned.
Clinical trials vary in design by factors including dosage, end points, length, and controls. We may need to conduct a series of trials to demonstrate the safety and efficacy of our products. The results of these trials may not demonstrate safety or efficacy sufficiently for regulatory authorities to approve our products. Further, the actual schedules for our clinical trials could vary dramatically from the forecasted schedules due to factors including changes in trial design, conflicts with the schedules of participating clinicians and clinical institutions, and changes affecting product supplies for clinical trials.
We rely on collaborators, including academic institutions, governmental agencies and clinical research organizations, to conduct, supervise, monitor and design some or all aspects of clinical trials involving our products. Since these trials depend on governmental participation and funding, we have less control over their timing and design than trials we sponsor. Delays in or failure to commence or complete any planned clinical trials could delay the ultimate timelines for our product releases. Such delays could reduce investors’ confidence in our ability to develop products, likely causing the price of our common stock to decrease.
If we are unable to obtain the regulatory approvals or clearances that are necessary to commercialize our products, we will have less revenue than expected.
China and other countries impose significant statutory and regulatory obligations upon the manufacture and sale of bio-pharmaceutical products. Each regulatory authority typically has a lengthy approval process in which it examines pre-clinical and clinical data and the facilities in which the product is manufactured. Regulatory submissions must meet complex criteria to demonstrate the safety and efficacy of the ultimate products. Addressing these criteria requires considerable data collection, verification and analysis. We may spend time and money preparing regulatory submissions or applications without assurances as to whether they will be approved on a timely basis or at all.
Our product candidates, some of which are currently in the early stages of development, will require significant additional development and pre-clinical and clinical testing prior to their commercialization. These steps and the process of obtaining required approvals and clearances can be costly and time-consuming. If our potential products are not successfully developed, cannot be proven to be safe and effective through clinical trials, or do not receive applicable regulatory approvals and clearances, or if there are delays in the process:
| · | the commercialization of our products could be adversely affected; |
| · | any competitive advantages of the products could be diminished; and |
| · | revenues or collaborative milestones from the products could be reduced or delayed. |
Governmental and regulatory authorities may approve a product candidate for fewer indications or narrower circumstances than requested or may condition approval on the performance of post-marketing studies for a product candidate. Even if a product receives regulatory approval and clearance, it may later exhibit adverse side effects that limit or prevent its widespread use or that force us to withdraw the product from the market.
Any marketed product and its manufacturer, including us, will continue to be subject to strict regulation after approval. Results of post-marketing programs may limit or expand the further marketing of products. Unforeseen problems with an approved product or any violation of regulations could result in restrictions on the product, including its withdrawal from the market and possible civil actions.
In manufacturing our products we will be required to comply with applicable good manufacturing practices regulations, which include requirements relating to quality control and quality assurance, as well as the maintenance of records and documentation. If we cannot comply with regulatory requirements, including applicable good manufacturing practice requirements, we may not be allowed to develop or market the product candidates. If we or our manufacturers fail to comply with applicable regulatory requirements at any stage during the regulatory process, we may be subject to sanctions, including fines, product recalls or seizures, injunctions, refusal of regulatory agencies to review pending market approval applications or supplements to approve applications, total or partial suspension of production, civil penalties, withdrawals of previously approved marketing applications and criminal prosecution.
11
Competitors may develop and market bio-pharmaceutical products that are less expensive, more effective or safer, making our products obsolete or uncompetitive.
Some of our competitors and potential competitors have greater product development capabilities and financial, scientific, marketing and human resources than we do. Technological competition from biopharmaceutical companies and biotechnology companies is intense and is expected to increase. Other companies have developed technologies that could be the basis for competitive products. Some of these products have an entirely different approach or means of accomplishing the desired curative effect than products we are developing. Alternative products may be developed that are more effective, work faster and are less costly than our products. Competitors may succeed in developing products earlier than us, obtaining approvals and clearances for such products more rapidly than us, or developing products that are more effective than ours. In addition, other forms of treatment may be competitive with our products. Over time, our technology or products may become obsolete or uncompetitive.
Our revenue will be materially and adversely affected if our products are unable to gain market acceptance.
Our products may not gain market acceptance in the agricultural community. The degree of market acceptance of any product depends on a number of factors, including establishment and demonstration of clinical efficacy and safety, cost-effectiveness, clinical advantages over alternative products, and marketing and distribution support for the products. Limited information regarding these factors is available in connection with our products or products that may compete with ours.
To directly market and distribute our bio-pharmaceutical products, we or our collaborators require a marketing and sales force with appropriate technical expertise and supporting distribution capabilities. We may not be able to further establish sales, marketing and distribution capabilities or enter into arrangements with third parties on acceptable terms. If we or our partners cannot successfully market and sell our products, our ability to generate revenue will be limited.
Our operations and the use of our products could subject us to damages relating to injuries or accidental contamination and thus reduce our earnings or increase our losses.
Our research and development processes involve the controlled use of hazardous materials. We are subject to federal, provincial and local laws and regulations governing the use, manufacture, storage, handling and disposal of such materials and waste products. The risk of accidental contamination or injury from handling and disposing of such materials cannot be completely eliminated. In the event of an accident involving hazardous materials, we could be held liable for resulting damages. We are not insured with respect to this liability. Such liability could exceed our resources. In the future we could incur significant costs to comply with environmental laws and regulations.
If we were sued for product liability, we could face substantial liabilities that may exceed our resources.
We may be held liable if any product we develop, or any product which is made using our technologies, causes injury or is found unsuitable during product testing, manufacturing, marketing, sale or use. These risks are inherent in the development of agricultural and bio-pharmaceutical products. We currently do not have product liability insurance. If we cannot obtain sufficient insurance coverage at an acceptable cost or otherwise protect against potential product liability claims, the commercialization of products that we develop may be prevented or inhibited. If we are sued for any injury caused by our products, our liability could exceed our total assets, whether or not we are successful.
We have no business liability or disruption insurance coverage and therefore we are susceptible to catastrophic or other events that may disrupt our business.
The insurance industry in China is still at an early stage of development. Insurance companies in China offer limited business insurance products. We do not have any business liability or disruption insurance coverage for our operations in China. Any business disruption, litigation or natural disaster may result in our incurring substantial costs and the diversion of our resources.
We will be unsuccessful if we fail to attract and retain qualified personnel.
We depend on a core management and scientific team. The loss of any of these individuals could prevent us from achieving our business objective of commercializing our product candidates. Our future success will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical and management personnel, as well as personnel with expertise in clinical testing and government regulation. We face competition for personnel from other companies, universities, public and private research institutions, government entities and other organizations. If our recruitment and retention efforts are unsuccessful, our business operations could suffer.
12
Downturn in the global economy may slow domestic growth in China, which in turn may affect our business.
Due to the global downturn in the financial markets, China may not be able to maintain its recent growth rates mainly due to the lack of demand of exports to countries that are in recessions. Although we do not presently export any of our products, our earnings may become unstable if China’s domestic growth slows significantly and the demand for meats and poultry declines.
Risks Related to Our Corporate Structure
Chinese laws and regulations governing our businesses and the validity of certain of our contractual arrangements are uncertain. If we are found to be in violation, we could be subject to sanctions. In addition, changes in such Chinese laws and regulations may materially and adversely affect our business.
There are substantial uncertainties regarding the interpretation and application of Chinese laws and regulations, including, but not limited to, the laws and regulations governing our business, or the enforcement and performance of our contractual arrangements with our affiliated Chinese entity, Xian Tianxing, and its stockholders. We are considered a foreign person or foreign invested enterprise under Chinese law. As a result, we are subject to Chinese law limitations on foreign ownership of Chinese companies. These laws and regulations are relatively new and may be subject to change, and their official interpretation and enforcement may involve substantial uncertainty. The effectiveness of newly enacted laws, regulations or amendments may be delayed, resulting in detrimental reliance by foreign investors. New laws and regulations that affect existing and proposed future businesses may also be applied retroactively.
The Chinese government has broad discretion in dealing with violations of laws and regulations, including levying fines, revoking business and other licenses and requiring actions necessary for compliance. In particular, licenses and permits issued or granted to us by relevant governmental bodies may be revoked at a later time by higher regulatory bodies. We cannot predict the effect of the interpretation of existing or new Chinese laws or regulations on our businesses. We cannot assure you that our current ownership and operating structure would not be found in violation of any current or future Chinese laws or regulations. As a result, we may be subject to sanctions, including fines, and could be required to restructure our operations or cease to provide certain services. Any of these or similar actions could significantly disrupt our business operations or restrict us from conducting a substantial portion of our business operations, which could materially and adversely affect our business, financial condition and results of operations.
We may be adversely affected by complexity, uncertainties and changes in Chinese regulation of bio-pharmaceutical business and companies, including limitations on our ability to own key assets.
The Chinese government regulates the bio-pharmaceutical industry including foreign ownership of, and the licensing and permit requirements pertaining to, companies in the bio-pharmaceutical industry. These laws and regulations are relatively new and evolving, and their interpretation and enforcement involve significant uncertainty. As a result, in certain circumstances it may be difficult to determine what actions or omissions may be deemed to be a violation of applicable laws and regulations. Issues, risks and uncertainties relating to Chinese government regulation of the bio-pharmaceutical industry include the following:
| · | we only have contractual control over Xian Tianxing. We do not own it due to the restriction of foreign investment in Chinese businesses; and |
| · | uncertainties relating to the regulation of the bio-pharmaceutical business in China, including evolving licensing practices, means that permits, licenses or operations at our company may be subject to challenge. This may disrupt our business, or subject us to sanctions, requirements to increase capital or other conditions or enforcement, or compromise enforceability of related contractual arrangements, or have other harmful effects on us. |
The interpretation and application of existing Chinese laws, regulations and policies and possible new laws, regulations or policies have created substantial uncertainties regarding the legality of existing and future foreign investments in, and the businesses and activities of, bio-pharmaceutical businesses in China, including our business.
In order to comply with Chinese laws limiting foreign ownership of Chinese bio-pharmaceutical companies, we conduct our bio-pharmaceutical business through Xian Tianxing by means of contractual arrangements. If the Chinese government determines that these contractual arrangements do not comply with applicable regulations, our business could be adversely affected.
The Chinese government restricts foreign investment in bio-pharmaceutical businesses in China. Accordingly, we operate our business in China through Xian Tianxing, a Chinese joint stock company. Xian Tianxing holds the licenses and approvals necessary to operate our bio-pharmaceutical business in China. We have contractual arrangements with Xian Tianxing and its stockholders that allow us to substantially control Xian Tianxing. We cannot assure you, however, that we will be able to enforce these contracts.
13
Although we believe we comply with current Chinese regulations, we cannot assure you that the Chinese government would agree that these operating arrangements comply with Chinese licensing, registration or other regulatory requirements, with existing policies or with requirements or policies that may be adopted in the future. If the Chinese government determines that we do not comply with applicable law, it could revoke our business and operating licenses, require us to discontinue or restrict our operations, restrict our right to collect revenues, require us to restructure our operations, impose additional conditions or requirements with which we may not be able to comply, impose restrictions on our business operations or on our customers, or take other regulatory or enforcement actions against us that could be harmful to our business.
Our contractual arrangements with Xian Tianxing and its stockholders may not be as effective in providing control over these entities as direct ownership.
Since Chinese law limits foreign equity ownership in bio-pharmaceutical companies in China, we operate our business through Xian Tianxing. We have no equity ownership interest in Xian Tianxing and rely on contractual arrangements to control and operate such businesses. These contractual arrangements may not be as effective in providing control over Xian Tianxing as direct ownership. For example, Xian Tianxing could fail to take actions required for our business despite its contractual obligation to do so. If Xian Tianxing fails to perform under their agreements with us, we may have to rely on legal remedies under Chinese law, which may not be effective. In addition, we cannot assure you that either of Xian Tianxing’s stockholders will act in our best interests.
Because we rely on the consulting services agreement with Xian Tianxing for our revenue, the termination of this agreement will severely and detrimentally affect our continuing business viability under our current corporate structure.
We are a holding company and do not have any assets or conduct any business operations other than the contractual arrangements between Sida and Xian Tianxing. As a result, we currently rely entirely for our revenues on dividend payments from Sida after it receives payments from Xian Tianxing pursuant to the consulting services agreement which forms a part of the contractual arrangements between Sida and Xian Tianxing. The consulting services agreement may be terminated by written notice of Sida or Xian Tianxing in the event that: (a) one party causes a material breach of the agreement, provided that if the breach does not relate to a financial obligation of the breaching party, that party may attempt to remedy the breach within 14 days following the receipt of the written notice; (b) one party becomes bankrupt, insolvent, is the subject of proceedings or arrangements for liquidation or dissolution, ceases to carry on business, or becomes unable to pay its debts as they become due; (c) Sida terminates its operations; (d) Xian Tianxing’s business license or any other license or approval for its business operations is terminated, cancelled or revoked; or (e) circumstances arise which would materially and adversely affect the performance or the objectives of the agreement. Additionally, Sida may terminate the consulting services agreement without cause. Because neither we nor our direct and indirect subsidiaries own equity interests of Xian Tianxing, the termination of the consulting services agreement would sever our ability to continue receiving payments from Xian Tianxing under our current holding company structure. While we are currently not aware of any event or reason that may cause the consulting services agreement to terminate, we cannot assure you that such an event or reason will not occur in the future. In the event that the consulting services agreement is terminated, this may have a severe and detrimental effect on our continuing business viability under our current corporate structure, which in turn may affect the value of your investment.
Members of Xian Tianxing’s management have potential conflicts of interest with us, which may adversely affect our business and your ability for recourse.
Weibing Lu, our Chief Executive Officer, is also the Chief Financial Officer and Chairman of the Board of Directors of Xian Tianxing. Mr. Wei Wen, who is Xian Tianxing’s Vice-General Manager and Director, is a member of Skystar’s board of directors. Conflicts of interests between their respective duties to our company and Xian Tianxing may arise. As our directors and executive officer (in the case of Mr. Lu), they have a duty of loyalty and care to us under U.S. and Cayman Islands law when there are any potential conflicts of interests between our company and Xian Tianxing. We cannot assure you, however, that when conflicts of interest arise, every one of them will act completely in our interests or that conflicts of interests will be resolved in our favor. For example, they may determine that it is in Xian Tianxing’s interests to sever the contractual arrangements with Sida, irrespective of the effect such action may have on us. In addition, any one of them could violate his or her legal duties by diverting business opportunities from us to others, thereby affecting the amount of payment Xian Tianxing is obligated to remit to us under the consulting services agreement.
Our board of directors is comprised of a majority of independent directors (including two based in the United States). These independent directors may be in a position to deter and counteract the actions of our officers or non-independent directors that are against our interests, as the independent directors do not have any position with, or interests in, our affiliate entities, and should therefore not have any conflicts of interests such as those potentially of our officers and directors who are management members of Xian Tianxing. Additionally, the independent directors have fiduciary duties to act in our best interests, and failure on their part to do so may subject them to personal liabilities for breach of such duties. We cannot, however, give any assurance as to how the independent directors will act. Further, if we or the independent directors cannot resolve any conflicts of interest between us and those of our officers and directors who are management members of Xian Tianxing, we would have to rely on legal proceedings, which could result in the disruption of our business.
14
In the event that you believe that your rights have been infringed under the securities laws or otherwise as a result of any one of the circumstances described above, it may be difficult or impossible for you to bring an action against Xian Tianxing or our officers or directors who are members of its management, the majority of whom reside within China. Even if you are successful in bringing an action, the laws of China may render you unable to enforce a judgment against the assets of Xian Tianxing and its management, all of which are located in China.
Risks Related to Doing Business in China
Adverse changes in economic and political policies of the Chinese government could have a material adverse effect on the overall economic growth of China, which could adversely affect our business.
Substantially all of our business operations are conducted in China. Accordingly, our results of operations, financial condition and prospects are subject to a significant degree to economic, political and legal developments in China. China’s economy differs from the economies of most developed countries in many respects, including with respect to the amount of government involvement, level of development, growth rate, control of foreign exchange and allocation of resources. While the Chinese economy has experienced significant growth in the past 20 years, growth has been uneven across different regions and among various economic sectors of China. The Chinese government has implemented various measures to encourage economic development and guide the allocation of resources. Some of these measures benefit the overall Chinese economy, but may also have a negative effect on us. For example, our financial condition and results of operations may be adversely affected by government control over capital investments or changes in tax regulations that are applicable to us. Since early 2004, the Chinese government has implemented certain measures to control the pace of economic growth. Such measures may cause a decrease in the level of economic activity in China, which in turn could adversely affect our results of operations and financial condition.
If Chinese law were to phase out the preferential tax benefits currently being extended to foreign invested enterprises and “new or high-technology enterprises” located in a high-tech zone, we would have to pay more taxes, which could have a material and adverse effect on our financial condition and results of operations.
Under Chinese laws and regulations, a foreign invested enterprise may enjoy preferential tax benefits if it is registered in a high-tech zone and also qualifies as “new or high-technology enterprise”. As a foreign invested enterprise as well as a certified “new or high-technology enterprise” located in a high-tech zone in Xian, the Company has been approved as a new technology enterprise and under Chinese Income Tax Laws, it is entitled to a preferential tax rate of 15%. If the Chinese law were to phase out preferential tax benefits currently granted to “new or high-technology enterprises” and technology consulting services, we would be subject to the standard statutory tax rate, which currently is 25%, and we would be unable to obtain business tax refunds for our provision of technology consulting services. Loss of these preferential tax treatments could have a material and adverse effect on our financial condition and results of operations.
Xian Tianxing is subject to restrictions on making payments to us.
We are a holding company incorporated in Nevada and do not have any assets or conduct any business operations other than our indirect investments in our affiliated entity in China, Xian Tianxing. As a result of our holding company structure, we rely entirely on payments from Xian Tianxing under our contractual arrangements. The Chinese government also imposes controls on the conversion of the Chinese currency, Renminbi (RMB), into foreign currencies and the remittance of currencies out of China. We may experience difficulties in completing the administrative procedures necessary to obtain and remit foreign currency. See “Government control of currency conversion may affect the value of your investment.” Furthermore, if our affiliated entity in China incurs debt on their own in the future, the instruments governing the debt may restrict their ability to make payments. If we are unable to receive all of the revenues from our operations through these contractual or dividend arrangements, we may be unable to pay dividends on our shares.
Uncertainties with respect to the Chinese legal system could adversely affect us.
We conduct our business primarily through our affiliated Chinese entity, Xian Tianxing. Our operations in China are governed by Chinese laws and regulations. We are generally subject to laws and regulations applicable to foreign investments in China and, in particular, laws applicable to wholly foreign-owned enterprises. The Chinese legal system is based on written statutes. Prior court decisions may be cited for reference but have limited precedential value.
15
Since 1979, Chinese legislation and regulations have significantly enhanced the protections afforded to various forms of foreign investments in China. However, China has not developed a fully integrated legal system and recently enacted laws and regulations may not sufficiently cover all aspects of economic activities in China. In particular, because these laws and regulations are relatively new, and because of the limited volume of published decisions and their nonbinding nature, the interpretation and enforcement of these laws and regulations involve uncertainties. In addition, the Chinese legal system is based in part on government policies and internal rules (some of which are not published on a timely basis or at all) that may have a retroactive effect. As a result, we may not be aware of our violation of these policies and rules until some time after the violation. In addition, any litigation in China may be protracted and result in substantial costs and diversion of resources and management attention.
You may experience difficulties in effecting service of legal process, enforcing foreign judgments or bringing original actions in China based on United States or other foreign laws against us, our management or the experts named in the prospectus.
We conduct substantially all of our operations in China and substantially all of our assets are located in China. In addition, all of our senior executive officers reside within China. As a result, it may not be possible to effect service of process within the United States or elsewhere outside China upon our senior executive officers, including with respect to matters arising under U.S. federal securities laws or applicable state securities laws. Moreover, our Chinese counsel has advised us that China does not have treaties with the United States or many other countries providing for the reciprocal recognition and enforcement of judgment of courts.
Governmental control of currency conversion may affect the value of your investment.
The Chinese government imposes controls on the convertibility of RMB into foreign currencies and, in certain cases, the remittance of currency out of China. We receive substantially all of our revenues in RMB. Under our current structure, our income is primarily derived from payments from Xian Tianxing. Shortages in the availability of foreign currency may restrict the ability of our Chinese subsidiaries and our affiliated entity to remit sufficient foreign currency to pay dividends or other payments to us, or otherwise satisfy their foreign currency denominated obligations. Under existing Chinese foreign exchange regulations, payments of current account items, including profit distributions, interest payments and expenditures from trade-related transactions, can be made in foreign currencies without prior approval from China State Administration of Foreign Exchange by complying with certain procedural requirements. However, approval from appropriate government authorities is required where RMB is to be converted into foreign currency and remitted out of China to pay capital expenses such as the repayment of bank loans denominated in foreign currencies. The Chinese government may also at its discretion restrict access in the future to foreign currencies for current account transactions. If the foreign exchange control system prevents us from obtaining sufficient foreign currency to satisfy our currency demands, we may not be able to pay dividends in foreign currencies to our stockholders.
Fluctuation in the value of RMB may have a material adverse effect on your investment.
The value of RMB against the U.S. dollar and other currencies may fluctuate and is affected by, among other things, changes in political and economic conditions. Our revenues and costs are mostly denominated in RMB, while a significant portion of our financial assets are denominated in U.S. dollars. We rely entirely on fees paid to us by our affiliated entity in China. Any significant fluctuation in the value of RMB may materially and adversely affect our cash flows, revenues, earnings and financial position, and the value of, and any dividends payable on, our stock in U.S. dollars. For example, an appreciation of RMB against the U.S. dollar would make any new RMB denominated investments or expenditures more costly to us, to the extent that we need to convert U.S. dollars into RMB for such purposes. An appreciation of RMB against the U.S. dollar would also result in foreign currency translation losses for financial reporting purposes when we translate our U.S. dollar denominated financial assets into RMB, as RMB is our reporting currency.
We face risks related to health epidemics and other outbreaks.
Our business could be adversely affected by the effects of an epidemic outbreak, such as the SARS epidemic in April 2004. Any prolonged recurrence of such adverse public health developments in China may have a material adverse effect on our business operations. For instance, health or other government regulations adopted in response may require temporary closure of our production facilities or of our offices. Such closures would severely disrupt our business operations and adversely affect our results of operations. We have not adopted any written preventive measures or contingency plans to combat any future outbreak of SARS or any other epidemic.
Risks Related to an Investment in Our Securities
To date, we have not paid any cash dividends and no cash dividends are expected to be paid in the foreseeable future.
We do not anticipate paying cash dividends on our common stock in the foreseeable future and we may not have sufficient funds legally available to pay dividends. Even if the funds are legally available for distribution, we may nevertheless decide not to pay any dividends. We intend to retain all earnings for our operations.
16
The NASDAQ Capital Market may delist our common stock from trading on its exchange, which could limit investors’ ability to effect transactions in our common stock and subject us to additional trading restrictions.
Our common stock is listed on The NASDAQ Capital Market. We cannot assure you that our common stock will continue to be listed on The NASDAQ Capital Market in the future. If The NASDAQ Capital Market delists our common stock from trading on its exchange, we could face significant material adverse consequences including:
| · | a limited availability of market quotations for our common stock; |
| · | a limited amount of news and analyst coverage for our company; and |
| · | a decreased ability to issue additional securities or obtain additional financing in the future. |
The market price of our common stock is volatile, and its value may be depressed at a time when you want to sell your holdings.
The market price of our common stock is volatile due to market and industry factors, many of which are beyond our control. Additionally, the price and trading volume for our common stock may be highly volatile for specific business reasons. Factors such as variations in our revenues, earnings and cash flow, announcements of new investments, cooperation arrangements or acquisitions, and fluctuations in market prices for our products could cause the market price for our shares to change substantially.
Securities class action litigation is often instituted against companies following periods of volatility in their stock price. This type of litigation could result in substantial costs to us and divert our management’s attention and resources.
Moreover, the trading market for our common stock will be influenced by research or reports that industry or securities analysts publish about us or our business. If one or more analysts who cover us downgrade our common stock, the market price for our common stock would likely decline. If one or more of these analysts cease coverage of us or fail to regularly publish reports on us, we could lose visibility in the financial markets, which, in turn, could cause the market price for our common stock or trading volume to decline.
Furthermore, securities markets may from time to time experience significant price and volume fluctuations for reasons unrelated to operating performance of particular companies. These market fluctuations may adversely affect the price of our common stock and other interests in our company at a time when you want to sell your interest in us.
Although publicly traded, the trading market in our common stock may be substantially less liquid than the average stock quoted on The NASDAQ Capital Market, and such low trading volume may adversely affect the price of our common stock.
Although our common stock is traded on The NASDAQ Capital Market, the trading volume of our common stock has generally been very low. Reported average daily trading volume in our common stock for the three month period ended March 31, 2010 was approximately 54,510 shares. Limited trading volume will subject our shares of common stock to greater price volatility and may make it difficult for you to sell your shares of common stock at a price that is attractive to you.
Our corporate actions are substantially controlled by our management stockholders and affiliated entities.
As of May 27, 2010, our management and their affiliated entities own approximately 17.1% of our outstanding common shares, representing approximately 17.1% of our voting power. These stockholders, acting individually or as a group, could exert substantial influence over matters such as electing directors and approving mergers or other business combination transactions. In addition, because of the percentage of ownership and voting concentration in these principal stockholders and their affiliated entities, elections of our board of directors will generally be within the control of these stockholders and their affiliated entities. While all of our stockholders are entitled to vote on matters submitted to our stockholders for approval, the concentration of shares and voting control presently lies with these principal stockholders and their affiliated entities. As such, it would be difficult for stockholders to propose and have approved proposals not supported by management. There can be no assurances that matters voted upon by our officers and directors in their capacity as stockholders will be viewed favorably by all stockholders of the company.
The elimination of monetary liability against our directors, officers and employees under Nevada law and the existence of indemnification rights to our directors, officers and employees may result in substantial expenditures by our company and may discourage lawsuits against our directors, officers and employees.
Our articles of incorporation do not contain any specific provisions that eliminate the liability of our directors for monetary damages to our company and stockholders, however we are prepared to give such indemnification to our directors and officers to the extent provided by Nevada law. We also have contractual indemnification obligations under our employment agreements with our chief executive officer, chief financial officer and certain of our directors. The foregoing indemnification obligations could result in our company incurring substantial expenditures to cover the cost of settlement or damage awards against directors and officers, which we may be unable to recoup. These provisions and resultant costs may also discourage our company from bringing a lawsuit against directors and officers for breaches of their fiduciary duties, and may similarly discourage the filing of derivative litigation by our stockholders against our directors and officers even though such actions, if successful, might otherwise benefit our company and stockholders.
17
Legislative actions, higher insurance costs and potential new accounting pronouncements may impact our future financial position and results of operations.
There have been regulatory changes, including the Sarbanes-Oxley Act of 2002, and there may potentially be new accounting pronouncements or additional regulatory rulings that will have an impact on our future financial position and results of operations. The Sarbanes-Oxley Act of 2002 and other similar rule changes are likely to increase general and administrative costs and expenses. Further, we currently maintain directors and officers insurance (“D&O Insurance”) as we are contractually obligated to do so. In light of the high claims rates in recent years, we expect that the premium for such insurance will increase. Additionally, there could be changes in certain accounting rules. These and other potential changes could materially increase the expenses we report under generally accepted accounting principles, and adversely affect our operating results.
Past company activities prior to the reverse merger may lead to future liability for the company.
Prior to our acquisition of Skystar Cayman in November 2005, we were engaged in businesses unrelated to our current operations. Although the prior business owners provided certain indemnifications against any loss, liability, claim, damage or expense arising out of or based on any breach of or inaccuracy in any of their representations and warranties made regarding such acquisition, any liabilities relating to such prior business against which Skystar is not completely indemnified may have a material adverse effect on Skystar.
The market price for our stock may be volatile.
The market price for our stock may be volatile and subject to wide fluctuations in response to factors including the following:
| · | actual or anticipated fluctuations in our quarterly operating results; |
| · | changes in financial estimates by securities research analysts; |
| · | conditions in bio-pharmaceutical and agricultural markets; |
| · | changes in the economic performance or market valuations of other bio-pharmaceutical companies; |
| · | announcements by us or our competitors of new products, acquisitions, strategic partnerships, joint ventures or capital commitments; |
| · | addition or departure of key personnel; |
| · | fluctuations of exchange rates between RMB and the U.S. dollar; |
| · | intellectual property litigation; and |
| · | general economic or political conditions in China. |
In addition, the securities market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance of particular companies. These market fluctuations may also materially and adversely affect the market price of our stock.
We may need additional capital, and the sale of additional shares or other equity securities could result in additional dilution to our stockholders.
We believe that our current cash and cash equivalents, anticipated cash flow from operations and the net proceeds from this offering will be sufficient to meet our anticipated cash needs for the foreseeable future. We may, however, require additional cash resources due to changed business conditions or other future developments, including any investments or acquisitions we may decide to pursue. If our resources are insufficient to satisfy our cash requirements, we may seek to sell additional equity or debt securities or obtain a credit facility. The sale of additional equity securities could result in additional dilution to our stockholders. The incurrence of indebtedness would result in increased debt service obligations and could result in operating and financing covenants that would restrict our operations. We cannot assure you that financing will be available in amounts or on terms acceptable to us, if at all.
18
If we fail to maintain an effective system of internal controls, we may not be able to accurately report our financial results or prevent fraud.
We are subject to reporting obligations under the U.S. securities laws. The SEC, as required by Section 404 of the Sarbanes-Oxley Act of 2002, adopted rules requiring every public company to include a management report on such company’s internal controls over financial reporting in its annual report, which contains management’s assessment of the effectiveness of our internal controls over financial reporting. In addition, an independent registered public accounting firm must attest to and report on management’s assessment of the effectiveness of our internal controls over financial reporting. Our management may conclude that our internal controls over our financial reporting are not effective. Our reporting obligations as a public company will place a significant strain on our management, operational and financial resources and systems for the foreseeable future. Effective internal controls, particularly those related to revenue recognition, are necessary for us to produce reliable financial reports and are important to help prevent fraud. As a result, our failure to achieve and maintain effective internal controls over financial reporting could result in the loss of investor confidence in the reliability of our financial statements, which in turn could harm our business and negatively impact the trading price of our stock. Furthermore, we anticipate that we will incur considerable costs and use significant management time and other resources in an effort to comply with Section 404 and other requirements of the Sarbanes-Oxley Act.
Shares eligible for future sale may adversely affect the market.
From time to time, certain of our stockholders may be eligible to sell all or some of their shares of common stock by means of ordinary brokerage transactions in the open market pursuant to Rule 144, promulgated under the Securities Act, subject to certain limitations. In general, pursuant to amended Rule 144, non-affiliate stockholders may sell freely after six months subject only to the current public information requirement (which disappears after one year). Affiliates may sell after six months subject to the Rule 144 volume, manner of sale (for equity securities), current public information and notice requirements. Of the approximately 7.1 million shares of our common stock outstanding as of May 27, 2010, approximately 5.1 million shares are, or will be, freely tradable without restriction, as of May 27, 2010. Any substantial sale of our common stock pursuant to Rule 144 may have a material adverse effect on the market price of our common stock.
The cancellation of our series “A” preferred stock may subject us to certain claims by the holder of series “A” preferred shares as well as indemnification obligations to the directors who authorized the issuance.
In 2001, 2,000,000 shares of our series “A” preferred stock were issued to a corporation wholly-owned by our then Chief Executive Officer and director for services purportedly rendered by him. The resolutions of the board of directors approving such issuance stated that the series “A” preferred shares carries a “super voting power of five.” No certificate of designation was ever filed with the Nevada Secretary of State for these shares and we do not believe any certificate evidencing such shares was issued by any transfer agent. As a result, the board of directors believes that the issuance was not valid under Nevada law and thus has adopted resolutions that resolve voiding all outstanding shares of the series “A” preferred stock and barring any re-issuance or authorization of our series “A” preferred stock unless such matter was submitted to a vote of our shareholders and approved by a disinterested vote of a majority of each class of our outstanding stock. On December 21, 2009, we instructed our transfer agent to remove the series “A” preferred shares officially from our shareholder records. As a result, the series “A” preferred shares will no longer be reported as part of our issued and outstanding capital stock.
Notwithstanding the cancellation of the series “A” preferred shares, our former Chief Executive Officer may potentially assert claims against us or the directors who authorized the issuance, in law or equity, for the proper issuance or reissuance of those shares. In a lawsuit, he may assert any number of legal or equitable theories in a forum with proper jurisdiction over the matter, but the substance would likely rest on whether he is entitled to the shares or, alternatively, whether he should be entitled to some form of damage payment from the Company for the services that he purportedly rendered to our company. In the event of any legal action, adequate insurance coverage may not be available to us to cover the cost of litigation, indemnification of any of our officers or directors named in such action or of any award or other resolution. If a court were to order the issuance of any shares of series “A” preferred stock, such shares could increase the total number of our shares outstanding and thereby dilute the interests of our other shareholders in our company, could control a significant voting interest in our company and possess other rights determined by a court which we are unable to predict.
The eventual development, outcome and cost of legal proceedings are by their nature uncertain and subject to many factors, including but not limited to, the discovery of facts not presently known to us or determinations by judges, juries or other finders of fact which do not accord with our evaluation of the merits or facts of the case. As a result, we can provide no assurance that we will succeed against any such challenge or as to the results if it were ever made.
Should we fail to prevail in our defense of such a claim, we may be subject to restitution or other forms of monetary damages, the amount of which is difficult to determine but may take into consideration the then and current fair market value of the series “A” preferred shares. Additionally, although the directors who authorized the issuance of the series “A” preferred shares are no longer members of our board of directors, we may nevertheless be obligated in certain circumstances to indemnify and defend these directors.
19
SPECIAL NOTE REGARDING FORWARD LOOKING STATEMENTS
This prospectus contains forward-looking statements. Such forward-looking statements include statements regarding, among other things, (a) our projected sales and profitability, (b) our growth strategies, (c) anticipated trends in our industry, (d) our future financing plans, and (e) our anticipated needs for working capital. Forward-looking statements, which involve assumptions and describe our future plans, strategies, and expectations, are generally identifiable by use of the words “may,” “will,” “should,” “expect,” “anticipate,” “estimate,” “believe,” “intend,” or “project” or the negative of these words or other variations on these words or comparable terminology. This information may involve known and unknown risks, uncertainties, and other factors that may cause our actual results, performance, or achievements to be materially different from the future results, performance, or achievements expressed or implied by any forward-looking statements. These statements may be found under “Prospectus Summary”, “Management's Discussion and Analysis of Financial Condition and Results of Operations” and “Description of Business,” as well as in this prospectus generally. Actual events or results may differ materially from those discussed in forward-looking statements as a result of various factors, including, without limitation, the risks outlined under “Risk Factors” and matters described in this prospectus generally. This prospectus may contain market data related to our business, which may have been included in articles published by independent industry sources. We are responsible for the accuracy and completeness of the historical information contained in this market data as of the date of this prospectus. However, this market data also includes projections that are based on a number of assumptions. If any one or more of these assumptions turns out to be incorrect, actual results may differ materially from the projections based on these assumptions. In light of these risks and uncertainties, there can be no assurance that the forward-looking statements contained in this prospectus will in fact occur. In addition to the information expressly required to be included in this filing, we will provide such further material information, if any, as may be necessary to make the required statements, in light of the circumstances under which they are made, not misleading.
Each forward-looking statement should be read in context with, and with an understanding of, the various other disclosures concerning our company and our business made elsewhere in this prospectus as well as other pubic reports which may be filed with the United States Securities and Exchange Commission. You should not place undue reliance on any forward-looking statement as a prediction of actual results or developments. We are not obligated to update or revise any forward-looking statement contained in this prospectus to reflect new events or circumstances, unless and to the extent required by applicable law. Neither the Private Securities Litigation Reform Act of 1995 nor Section 27A of the Securities Act of 1933, as amended, provides any protection for statements made in this prospectus.
20
USE OF PROCEEDS
We estimate that we will receive net proceeds of $___ million from the sale of ________ shares of common stock being offered at an assumed public offering price of $____ per share, after deducting estimated underwriting discounts and commissions and estimated offering expenses, or approximately $____ if the underwriter’s over-allotment option is exercised in full. We expect to use the net proceeds from this offering as follows:
Application of Net Proceeds | Percentage of Net Proceeds | |||||||
Build-out of production facilities in Kunshan, China(1) | $ | 40 | % | |||||
Research & Development(2) | 20 | % | ||||||
Future acquisitions and working capital (3) | 40 | % | ||||||
| Total | $ | 100 | % | |||||
| (1) | This facility will be used for the housing and production of micro-organism products (including enzymes and probiotic cultures), and pet food and nutritional supplements. We anticipate completing construction of this facility by the end of the fourth quarter of 2010. |
| (2) | Research and development of veterinary medicine, vaccine, enzyme, peptide products, and collaboration with domestic and foreign research institutions. |
(3) | We may enter into acquisitions of other GMP-certified veterinary and livestock medicine-oriented businesses in our geographic area and other parts of China. Working capital includes funding for general corporate purposes. |
The amounts actually spent by us for any specific purpose may vary significantly and will depend on a number of factors, including the progress of our commercialization and development efforts. Accordingly, our management has broad discretion to allocate the net proceeds. Pending the uses described above, we intend to invest the net proceeds of this offering in short-term, interest-bearing, investment-grade securities.
21
MARKET FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Our common stock has been trading on the Nasdaq Capital Market under the symbol “SKBI” since June 26, 2009. Our common stock was previously quoted on the Over-The-Counter Bulletin Board, or OTCBB, under the trading symbol “SKBO.OB” until June 25, 2009. The following table sets forth, for the periods indicated, the reported high and low closing bid quotations for our common stock as reported on the OTC Bulletin Board through June 25, 2009 and our common stock on the Nasdaq Capital Market since June 26, 2009 for the periods indicated. The bid prices reflect inter-dealer quotations, do not include retail markups, markdowns or commissions and do not necessarily reflect actual transactions.
Common Stock
The OTCBB Bid Price per Share (1) | The Nasdaq Capital Market Price per Share (2) | ||||||||||||
| High | Low | High | Low | ||||||||||
| Quarter ended | |||||||||||||
| June 30, 2010 (3) | $ | N/A | $ | N/A | $ | 11.63 | $ | 8.20 | |||||
| March 31, 2010 | $ | N/A | $ | N/A | $ | 12.00 | $ | 8.00 | |||||
| December 31, 2009 | $ | N/A | $ | N/A | $ | 13.50 | * | $ | 6.805 | * | |||
| September 30, 2009 | $ | N/A | $ | N/A | $ | 8.60 | * | $ | 5.555 | * | |||
| June 30, 2009 | $ | 18.50 | $ | 0.30 | $ | 11.245 | * | $ | 7.65 | * | |||
| March 31, 2009 | $ | 0.90 | $ | 0.25 | $ | N/A | $ | N/A | |||||
| December 31, 2008 | $ | 0.75 | $ | 0.30 | $ | N/A | $ | N/A | |||||
| September 30, 2008 | $ | 1.10 | $ | 0.51 | $ | N/A | $ | N/A | |||||
| June 30, 2008 | $ | 1.15 | $ | 0.90 | $ | N/A | $ | N/A | |||||
| March 31, 2008 | $ | 1.40 | $ | 0.90 | $ | N/A | $ | N/A | |||||
(1) Through June 25, 2009.
(2) From June 26, 2009 forward.
(3) Through May 27, 2010.
* Actual price adjusted to take into account 2-for-1 forward stock split effected on November 16, 2009.
As of May 27, 2010, we had approximately 7,106,705 shares of common stock issued and outstanding.
As of May 27, 2010, we have outstanding warrants that, if exercised, would permit their holders to purchase approximately an additional 34,230 shares of our common stock.
As of May 27, 2010, one of our directors is owed 49,834 shares of common stock for his services as our U.S. Representative through March 31, 2010.
Assuming exercise of all warrants, and issuance of shares owed to the director, we would have approximately 71,190,769 shares of common stock outstanding.
Holders
As of May 27, 2010, there were approximately 393 stockholders of record of our common stock.
22
DIVIDEND POLICY
While there are no restrictions that limit our ability to pay dividends, we have not paid, and do not currently intend to pay cash dividends on our common stock in the foreseeable future. Our policy is to retain all earnings, if any, to provide funds for operation and expansion of our business. The declaration of dividends, if any, will be subject to the discretion of our Board of Directors, which may consider such factors as our results of operations, financial condition, capital needs and acquisition strategy, among others.
CAPITALIZATION
The following table sets forth our capitalization as of March 31, 2010:
| · | on an actual basis; and |
| · | on an as adjusted basis to give effect to the sale of _________ shares of common stock in this offering at an assumed public offering price of $___ per share, after deducting the estimated underwriting discount and commissions and estimated offering expenses payable by us. |
You should read this table together with Management’s Discussion and Analysis of Financial Condition and Results of Operations and our consolidated financial statements and the related notes appearing elsewhere in this prospectus.
As of March 31, 2010
Actual | As Adjusted | |||||||
| Common stock, $0.001 par value, 40,000,000 shares authorized; 7,097,708 shares issued and outstanding. | $ | 7,097 | $ | |||||
| Paid-in-capital | $ | 35,966,731 | $ | |||||
| Statutory reserves | $ | 3,879,077 | $ | |||||
| Retained earnings | $ | 13,671,153 | $ | |||||
| Accumulated other comprehensive income | $ | 2,830,705 | $ | |||||
| Total shareholder’s equity | $ | 56,354,763 | $ | |||||
| Total capitalization | $ | 82,404,390 | $ | |||||
23
DILUTION
As of March 31, 2010, we had a net tangible book value of $54,585,992 or $7.69 per share. Net tangible book value represents our total tangible assets, less all liabilities, divided by the number of shares of common stock outstanding. Without taking into account any changes in such net tangible book value after March 31, 2010, other than to give effect to the sale by us of ________ shares of common stock offered hereby, as well as the _______ shares underlying the underwriter’s common stock purchase option, the pro forma net tangible book value per share at March 31, 2010 would have been $____. This amount represents an immediate decrease in net tangible book value of $____ per share to our current shareholders and an immediate increase in net tangible book value of $____ per share to new investors purchasing shares in this offering as illustrated in the following table:
| Assumed public offering price per share | $ | |||
| Net tangible book value per share before the offering | $ | |||
| Decrease in net tangible book value per share to existing shareholders attributable to new investors (after deduction of the estimated underwriting discount and other offering expenses to be paid by us) | $ | |||
| Pro-forma net tangible book value per share after the offering | $ | |||
| Increased value per share to new investors (determined by taking the adjusted net tangible book value after the offering and deducting the amount of cash paid by a new investor for a share of common stock) | $ |
The following table sets forth, on a pro forma basis as of March 31, 2010, the number of shares of common stock purchased from us, the total consideration paid and the average price per share paid by the existing shareholders and by the new investors, assuming in the case of new investors a public offering price of $____ per share, before deductions of the underwriting and other offering expenses:
Shares Purchased Number | Percent | Total Consideration | Percent | Average Price Per Share | ||||||||||||||||
| Existing Shareholders | % | $ | % | $ | ||||||||||||||||
| New Investors | % | $ | % | $ | ||||||||||||||||
| Total | 100 | % | $ | 100 | % | |||||||||||||||
24
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of our results of operations and financial condition for the three months ended March 31, 2010 and 2009, and for the fiscal years ended December 31, 2009 and 2008, should be read in conjunction with the Summary Financial Information and our financial statements and the notes to those financial statements that are included elsewhere in this prospectus. Our discussion includes forward-looking statements based upon current expectations that involve risks and uncertainties, such as our plans, objectives, expectations and intentions. Actual results and the timing of events could differ materially from those anticipated in these forward-looking statements as a result of a number of factors, including those set forth under the “Risk Factors”, “Cautionary Notice Regarding Forward-Looking Statements” and “Description of Business” sections and elsewhere in this prospectus. We use words such as “anticipate,” “estimate,” “plan,” “project,” “continuing,” “ongoing,” “expect,” “believe,” “intend,” “may,” “will,” “should,” “could,” “predict,” and similar expressions to identify forward-looking statements. Although we believe the expectations expressed in these forward-looking statements are based on reasonable assumptions within the bound of our knowledge of our business, our actual results could differ materially from those discussed in these statements. Factors that could contribute to such differences include, but are not limited to, those discussed in the “Risk Factors” section of this prospectus. We undertake no obligation to update publicly any forward-looking statements for any reason even if new information becomes available or other events occur in the future.
Our financial statements are prepared in US$ and in accordance with accounting principles generally accepted in the United States. See “Exchange Rates” below for information concerning the exchanges rates at which Renminbi (“RMB”) were translated into US$ at various pertinent dates and for pertinent periods.
Overview
Skystar Bio-Pharmaceutical Company (“Skystar” or the “Company”), formerly known as The Cyber Group Network Corporation, was incorporated in Nevada on September 24, 1998. The Company is a holding company that, through its variable interest entity (“VIE”) Xian Tianxing Bio-Pharmaceutical Co., Ltd. (“Xian Tianxing”), researches, develops, manufactures and distributes veterinary health care and medical care products in the People’s Republic of China (“PRC”).
All of the Company’s operations are carried out by Xian Tianxing, a PRC company, which the Company controls through contractual arrangements originally between Xian Tianxing and Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”), a Cayman Islands company that became the Company’s wholly owned subsidiary subsequent to a share exchange transaction on November 7, 2005. On March 10, 2008, all of the rights and obligations of Skystar Cayman under the contractual arrangements were transferred to Sida Biotechnology (Xian) Co., Ltd. (“Sida”), a PRC company and wholly owned subsidiary of Fortunate Time International Limited, a Hong Kong company and wholly owned subsidiary of Skystar Cayman. Xian Tianxing also has a wholly owned subsidiary, Shanghai Siqiang Biotechnological Co., Ltd., a PRC company.
Such contractual arrangements are necessary to comply with Chinese laws limiting foreign ownership of certain companies. Through these contractual arrangements, we have the ability to substantially influence Xian Tianxing’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring shareholder approval. As a result of these contractual arrangements, which enable us to control Xian Tianxing, we are considered the primary beneficiary of Xian Tianxing. Please see Note 1 to our consolidated financial statements for year ended December 31, 2009, for the impact of the contractual arrangements on our consolidated financial statements.
Currently, we have four major product lines:
| · | Our bio-pharmaceutical veterinary vaccine line currently includes over 10 products; |
| · | Our veterinary medicine line for poultry and livestock currently includes over 159 products; |
| · | Our feed additives line currently includes over 10 products; and |
| · | Our micro-organism products line currently includes over 16 products. |
All of our revenue is derived from the sale of veterinary healthcare and medical care products in China, which are distributed through a distribution channel covering 29 provinces. As of March 31, 2010, we had over 1,539 distributors and 529 direct customers.
Critical Accounting Policies
In preparing the consolidated financial statements in accordance with accounting principles generally accepted in the United States of America, we make estimates and assumptions about the effect of matters that are inherently uncertain and may change in subsequent periods. The resulting accounting estimates will, by definition, vary from the related actual results. We consider the following to be the most critical accounting policies:
25
| · | Revenue recognition: Revenues of the Company include sales of bio-pharmaceutical and veterinary products in China. Sales are recognized when the following four revenue criteria are met: persuasive evidence of an arrangement exists, delivery has occurred, the selling price is fixed or determinable, and collectibility is reasonably assured. Sales are recorded net of value added tax (VAT). No return allowance is made as product returns are insignificant based on historical experience. |
| (a) | Credit sales: Revenue is recognized when the products have been delivered to the customers. |
| (b) | Full payment before delivering: Revenue is recognized when the products have been delivered to customers. |
| · | Accounts receivable: We perform ongoing credit evaluations of our customers and adjust credit limits based upon payment history and the customers’ current credit worthiness, as determined by a review of their current credit information. We continuously monitor collections and payments from our customers and maintain a provision for estimated credit losses based upon historical experience and any specific customer collection issues that have been identified. While such credit losses have historically been within our expectations and the provisions established, we cannot guarantee that we will continue to experience the same credit loss rates that have been experienced in the past. |
Our accounts receivable aging was as follows for the periods below:
From Date of Invoice to Customer: | March 31, 2010 | December 31, 2009 | December 31, 2008 | |||||||||
| 0 – 90 days | $ | 2,975,490 | $ | 4,401,071 | $ | 2,217,172 | ||||||
| 91 – 180 days | 555,395 | 177,416 | 374,665 | |||||||||
| 181 – 270 days | 110,032 | 21,868 | 73,524 | |||||||||
| 271 – 360 days | 19,807 | 23,865 | 80,296 | |||||||||
| Over 360 days | 114,211 | 86,824 | 6,302 | |||||||||
| Allowance for bad debts | (327,857 | ) | (327,857 | ) | (327,857 | ) | ||||||
| Total | $ | 3,447,078 | $ | 4,383,187 | $ | 2,424,102 | ||||||
On average, we collect our receivables within 90 days. Historically, we have collected all of our accounts receivable and have had no write offs. This is attributed to the steps that we take prior to extending credit to our customers as discussed above. If we are having difficulty collecting from a customer, we take the following steps: cease existing shipments to the customer, our sales force actively calls and goes to the customer’s site reminding the customer of their past due invoice and requesting payment, and if those methods are unsuccessful we use our outside legal counsel. If, in the future, those steps are unsuccessful, management would determine whether or not the receivable should be written off.
| · | Warrants: We have adopted the accounting standards of accounting for stock purchase warrants and other related derivative accounting standards for valuation and accounting treatment of our outstanding warrants. |
Recent Accounting Pronouncements
In October 2009, the FASB issued an ASU regarding accounting for own-share lending arrangements in contemplation of convertible debt issuance or other financing. This ASU requires that at the date of issuance of the shares in a share-lending arrangement entered into in contemplation of a convertible debt offering or other financing, the shares issued shall be measured at fair value and be recognized as an issuance cost, with an of f set to additional paid-in capital. Further, loaned shares are excluded from basic and diluted earnings per share unless default of the share-lending arrangement occurs, at which time the loaned shares would be included in the basic and diluted earnings-pe r -share calculation. This ASU is effective for fiscal years beginning on or after December 15, 2009, and interim periods within those fiscal years for arrangements outstanding as of the beginning of those fiscal years. The adoption of this ASU did not have a material impact on the Company’s consolidated financial statements.
In January 2010, the FASB issued ASU No. 2010-02 regarding accounting and reporting for decreases in ownership of a subsidiary. Under this guidance, an entity is required to deconsolidate a subsidiary when the entity ceases to have a controlling financial interest in the subsidiary. Upon deconsolidation of a subsidiary, an entity recognizes a gain or loss on the transaction and measures any retained investment in the subsidiary at fair value. In contrast, an entity is required to account for a decrease in its ownership interest of a subsidiary that does not result in a change of control of the subsidiary as an equity transaction. This ASU clarifies the scope of the decrease in ownership provisions, and expands the disclosures about the deconsolidation of a subsidiary or de-recognition of a group of assets. This ASU is effective beginning in the first interim or annual reporting period ending on or after December 31, 2009. The adoption of this ASU did not have a material impact the Company’s consolidated financial statements.
26
In January 2010, FASB issued ASU No. 2010-06 – Improving Disclosures about Fair Value Measurements. This update provides amendments to Subtopic 820-10 that requires new disclosure to include transfers in and out of Levels 1 and 2 and activity in Level 3 fair value measurements. Further, this update clarifies existing disclosures on level of disaggregation and Disclosures about inputs and valuation techniques. A reporting entity should provide fair value measurement disclosures for each class of assets and liabilities and should provide disclosures about the valuation techniques and inputs used to measure fair value for both recurring and nonrecurring fair value measurements. Those disclosures are required for fair value measurements that fall in either Level 2 or Level 3. The new disclosures and clarifications of existing disclosures are effective for interim and annual reporting periods beginning after December 15, 2009, except for the disclosures about purchases, sales, issuances, and settlements in the roll forward of activity in Level 3 fair value measurements. Those disclosures are effective for fiscal years beginning after December 15, 2010, and for interim periods within those fiscal years. The Company is currently evaluating the impact of this ASU; however, the Company does not expect the adoption of this ASU to have a material impact on its consolidated financial statements.
In February 2010, the FASB issued ASU 2010-09, “Subsequent Events (Topic 855): Amendments to Certain Recognition and Disclosure Requirements,” or ASU 2010-09. ASU 2010-09 primarily rescinds the requirement that, for listed companies, financial statements clearly disclose the date through which subsequent events have been evaluated. Subsequent events must still be evaluated through the date of financial statement issuance; however, the disclosure requirement has been removed to avoid conflicts with other SEC guidelines. ASU 2010-09 was effective immediately upon issuance and was adopted in February 2010.
Results of Operations
Three months ended March 31, 2010 as compared to three months ended March 31, 2009
The following table summarizes our results of operations for the three months ended March 31, 2010 and 2009. The table and the discussion below should be read in conjunction with the unaudited financial statements and the notes thereto appearing elsewhere in this prospectus.
| Three Months Ended March 31, | ||||||||||||||||
| 2010 | 2009 | |||||||||||||||
| Amount | Percentage of total revenue | Amount | Percentage of total revenue | |||||||||||||
| Revenues | $ | 4,869,243 | 100 | % | $ | 3,823,566 | 100 | % | ||||||||
| Gross Profit | $ | 2,578,024 | 53 | % | $ | 1,877,208 | 49 | % | ||||||||
| Operating Expense | $ | 834,679 | 17 | % | $ | 639,442 | 17 | % | ||||||||
| Income from Operations | $ | 1,743,345 | 36 | % | $ | 1,237,766 | 32 | % | ||||||||
| Other (Expenses)/ gains | $ | (321,779) | 6 | % | $ | 38,398 | 1 | % | ||||||||
| Income Tax Expenses | $ | 325,319 | 7 | % | $ | 211,520 | 5 | % | ||||||||
| Net Income | $ | 1,096,247 | 23 | % | $ | 1,064,644 | 28 | % | ||||||||
Revenues. All of our revenues are derived from the sale of veterinary healthcare and medical care products in the PRC. For the three months ended March 31, 2010, we had revenues of $4,869,243 as compared to $3,823,566 for the three months ended March 31, 2009, an increase of approximately 27%. Our revenues consist of four product lines: veterinary medications, micro-organism, feed additives, and vaccines. Our revenues tend to be seasonal whereby the third quarter, fourth quarter, second quarter and first quarter have historically trended to represent our largest to smallest revenue quarters, respectively.
Revenues — Veterinary Medications. Revenues from sales of our veterinary medications product line increased from $2,602,496 for the three months ended March 31, 2009 to $3,196,259 for the three months ended March 31, 2010, for an increase of $593,763 or 23%. The increase in veterinary medication sales was primarily due to our increased utilization of our veterinary medicine facility and increased sales efforts. Of total revenues from veterinary medications during the three months March 31, 2010, approximately $830,600, or 17% of total revenues, resulted from the sale of Praziquantel tablets which treats schistosomiasis.
Revenues — Micro-Organism. Revenues from sales of our micro-organism product line increased from $893,293 for the three months ended March 31, 2009 to $1,206,578 for the three months ended March 31, 2010 for an increase of $313,285 or 35%. The increase was a result of increased sales efforts of our probiotics micro-organism products during the three months ended March 31, 2010.
Revenues — Feed Additives. Revenues from sales of our feed additives product line increased from $182,311 for the three months ended March 31, 2009 to $236,040 for the three months ended March 31, 2010 for an increase of $53,729 or 29%. The increase was the result of increased sales efforts of our feed additives products during the three months ended March 31, 2010.
27
Revenues — Vaccines. Revenues from sales of our vaccines product line increased from $145,466 for the three months ended March 31, 2009 to $230,366 for the three months ended March 31, 2010 for an increase of $84,900 or 58%. This increase was a result of increased demand for our vaccine products during the three months ended March 31, 2010. We are presently operating at full production capacity for our vaccine product line and therefore cannot significantly increase sales until we expand our production capabilities, which we presently have underway and anticipate completion in the first half of 2010 and commencement of production in the second half of 2010.
Cost of Sales
Cost of revenue. For the three months ended March 31, 2010, our cost of revenues, which consists of raw materials, direct labor, and manufacturing overhead, was $2,291,219 as compared to $1,946,358 for the three months ended March 31, 2009, an increase of approximately 18%. Our cost of sales consists of four product lines: veterinary medications, micro-organism, feed additives, and vaccines. The increase was primarily due to increased product sales.
Cost of Sales — Veterinary Medications. Cost of sales of our veterinary medications product line increased from $1,592,809 for the three months ended March 31, 2009 to $1,822,258 for the three months ended March 31, 2010, for an increase of $229,449 or approximately 14%. This increase was mainly due to the corresponding increase in veterinary medication sales.
Cost of Sales — Micro-Organism. Cost of sales of our micro-organism product line increased from $260,537 for the three months ended March 31, 2009 to $343,018 for the three months ended March 31, 2010 for an increase of $82,481 or approximately 32%. This increase was mainly due to the corresponding increase in micro-organism sales.
Cost of Sales — Feed Additives. Cost of sales of our feed additives product line increased from $77,439 for the three months ended March 31, 2009 to $100,046 for the three months ended March 31, 2010 for an increase of $22,607 or 29%. This increase was mainly due to a corresponding increase in feed additives products sales.
Cost of Sales — Vaccines. Cost of sales of our vaccines product line increased from $15,573 for the three months ended March 31, 2009 to $25,897 for the three months ended March 31, 2010, for an increase of $10,324 or 66%. This increase was the result of an increase of vaccine product sales during the three months ended March 31, 2010. We are presently operating at full production capacity for our vaccine product line and therefore cannot significantly increase sales until we expand our production capabilities which we presently have underway and anticipate completion in the second half of the year.
Operating Expenses
| The Three Months Ended March, | ||||||||||||||||
| 2010 | 2009 | |||||||||||||||
| Amount | Percentage of total revenue | Amount | Percentage of total revenue | |||||||||||||
| Gross Profit | $ | 2,578,024 | 53 | % | $ | 1,877,208 | 49 | % | ||||||||
| Operating Expenses | $ | 834,679 | 17 | % | $ | 639,442 | 17 | % | ||||||||
| Selling Expenses | $ | 171,134 | 3 | % | $ | 207,395 | 6 | % | ||||||||
| General and Administrative Expenses | $ | 619,550 | 13 | % | $ | 314,695 | 8 | % | ||||||||
| Research and Development Costs | $ | 43,995 | 1 | % | $ | 117,352 | 3 | % | ||||||||
| Income from Operations | $ | 1,743,345 | 36 | % | $ | 1,237,766 | 32 | % | ||||||||
Selling Expenses. Selling expenses, which consist of commissions, advertising and promotion expenses, freight charges, and salaries, totaled $171,134 for the three months ended March 31, 2010 as compared to $207,395 for the three months ended March 31, 2009, a decrease of approximately $36,261 or 17%. This decrease is the result of reduced travel expenses from hiring local sales people, and reduced trade show expenses for the first three months of 2010 compared to the same period in 2009.
General and Administrative Expenses. General and administrative expenses totaled $619,550 for the three months ended March 31, 2010, as compared to $314,695 for the three months ended March 31, 2009, an increase of approximately 97%. General and administrative expenses for our Chinese operating entity have held steady relative to revenue. This increase was mainly incurred in the United States, including stock compensation expenses for officers and directors, and fees related to auditing, legal, investor relations, and other professional services.
28
Research and Development Costs. Research and development costs, which consist of salaries, professional fees, and technical support fees, totaled $43,995 for the three months ended March 31, 2010, as compared to $117,352 for the three months ended March 31, 2009, a decrease of approximately 63%. Most of the current research projects are in the later phases of development focusing on applications and documentation without the significant outlays related to the earlier phases of the projects when large scale testing was required.
Year ended December 31, 2009 as compared to year ended December 31, 2008
The following table summarizes our results of operations for the two most recent fiscal years. The table and the discussion below should be read in conjunction with the audited financial statements and the notes thereto appearing elsewhere in this prospectus.
| For the Twelve Months Ended December 31, | ||||||||||||||||
| 2009 | 2008 | |||||||||||||||
| (In U.S. Dollars, Except for Percentages) | ||||||||||||||||
| Revenues | $ | 33,778,305 | 100.0 | % | $ | 25,584,446 | 100.0 | % | ||||||||
| Gross Profit | $ | 17,257,316 | 51.1 | % | $ | 12,775,550 | 49.9 | % | ||||||||
| Operating Expense | $ | 5,562,848 | 16.5 | % | $ | 4,594,563 | 18.0 | % | ||||||||
| Income from Operations | $ | 11,694,468 | 34.6 | % | $ | 8,180,987 | 32.0 | % | ||||||||
| Other Expenses | $ | 813,162 | 2.4 | % | $ | 1,055,116 | 4.1 | % | ||||||||
| Income tax expenses | $ | 2,029,374 | 6.0 | % | $ | 1,529,688 | 6.0 | % | ||||||||
| Net (Loss) Income | $ | 8,851,932 | 26.2 | % | $ | 5,596,183 | 21.9 | % | ||||||||
Revenues. All of our revenues are derived from the sale of veterinary healthcare and medical care products in the PRC. For the year ended December 31, 2009, we had revenues of $33,778,305 as compared to revenues of $25,584,446 for the year ended December 31, 2008, an increase of approximately 32.0%.
Revenues — Veterinary Medications. Revenues from sales of our veterinary medications product line increased from $17,535,757for the year ended December 31, 2008 to $22,920,479 for the year ended December 31, 2009 for an increase of $5,384,722 or 30.7%. The increase in veterinary medication sales was primarily due to our increased utilization of our 2007 veterinary medicine facility expansion of 200% and increased sales efforts. We were able to utilize this capacity expansion as our customers increased the use of products for the treatment of livestock and poultry diseases during the year ended December 31, 2009. Of total revenues from veterinary medications during the year ended December 31, 2009, approximately $6,800,574 or 20% of total revenue resulted from the sale of Praziquantel tablets which treats schistosomiasis.
Revenues — Micro-Organism. Revenues from sales of our micro-organism product line increased from $5,868,623 for the year ended December 31, 2008 to $8,021,139 for the year ended December 31, 2009 for an increase of $ 2,152,516 or 36.7%. The increase was the result of increased sales efforts of our probiotics micro-organism products during the year ended December 31, 2009.
Revenues — Feed Additives. Revenues from sales of our feed additives product line increased from $1,189,108 for the year ended December 31, 2008 to $1,411,222 for the year ended December 31, 2009 for an increase of $222,114 or 18.7%. The increase of $222,114 was the result of increased sales efforts of our multi-enzyme feed additive products.
Revenues — Vaccines. Revenues from sales of our vaccines product line increased from $990,958 for the year ended December 31, 2008 to $1,425,465 for the year ended December 31, 2009 or an increase of $434,507 or 43.8%. This increase was a result of increased demand for our vaccine products during the year ended December 31, 2009. We are presently operating at full production capacity for our vaccine product line and therefore cannot significantly increase sales until we expand our production capabilities which we presently have underway and anticipate completion during the second quarter of 2010.
Cost of Sales
Cost of Sales. For the year ended December 31, 2009, we had cost of revenues, which consists of raw materials, direct labor, and manufacturing overhead, of $16,520,989 as compared to cost of sales of $ 12,808,896 for the year ended December 31, 2008, an increase of approximately 29.0%. Our cost of sales consists of four product lines: veterinary medications, micro-organism, feed additives, and vaccines. The increase was due to our overall sales increase of 32.0%.
Cost of Sales — Veterinary Medications. Cost of sales of our veterinary medications product line increased from $10,389,726 for the year ended December 31, 2008 to $13,672,332 for the year ended December 31, 2009, for an increase of $3,282,607or approximately 31.6%. This increase was mainly due to the corresponding increase in veterinary medication sales.
29
Cost of Sales — Micro-Organism. Cost of sales of our micro-organism product line increased from $1,781,598 for the year ended December 31, 2008 to $2,129,945 for the year ended December 31, 2009 for an increase of $348,346 or approximately 19.6%. This increase was mainly due to a corresponding increase in micro-organism sales.
Cost of Sales — Feed Additives. Cost of sales of our feed additives product line increased from $525,653 for the year ended December 31, 2008 to $568,007 for the year ended December 31, 2009 for an increase of $42,354 or 8.1%. This increase was mainly due to a corresponding increase in feed additive sales.
Cost of Sales — Vaccines. Cost of sales of our vaccines product line increased from $111,919 for the year ended December 31, 2008 to $150,705 for the year ended December 31, 2009, for an increase of $38,786 or 34.7%. This increase was the result of an increase of vaccine product sales during the year ended December 31, 2009. We are presently operating at full production capacity for our vaccine product line and therefore cannot significantly increase sales until we expand our production capabilities which we presently have underway and anticipate completion during the second quarter of 2010.
Operating Expenses
| Years Ended December 31, | ||||||||||||||||
| 2009 | 2008 | |||||||||||||||
| Amount | Percentage of Total Revenue | Amount | Percentage of Total Revenue | |||||||||||||
| Gross Profit | $ | 17,256,316 | 51.1 | % | $ | 12,775,550 | 49.9 | % | ||||||||
| Operating Expenses | $ | 5,562,848 | 16.5 | % | $ | 4,594,563 | 18.0 | % | ||||||||
| Selling Expenses | $ | 1,928,441 | 5.7 | % | $ | 1,381,807 | 5.4 | % | ||||||||
| General and Administrative Expenses | $ | 2,466,470 | 7.3 | % | $ | 2,663,520 | 10.4 | % | ||||||||
| Research and Development Costs | $ | 1,167,937 | 3.5 | % | $ | 549,236 | 2.1 | % | ||||||||
| Income from Operations | $ | 11,694,468 | 34.6 | % | $ | 8,180,987 | 32.0 | % | ||||||||
Selling Expenses. Selling expenses, which consist of commissions, advertising and promotion expenses, freight charges, and salaries, totaled $1,928,441 for the year ended December 31, 2009 as compared to $1,381,807 for the year ended December 31, 2008, an increase of approximately 546,634 or 39.6%. This increase is the result of our increased sales during the year ended December 31, 2009.
General and Administrative Expenses. General and administrative expenses totaled $2,466,470 for the year ended December 31, 2009, as compared to $2,663,520 for the year ended December 31, 2008, a decrease of approximately 7.4%. Although our general and administrative expenses decreased slightly, we anticipate that our general and administrative expenses will increase due to the increasing costs of being a U.S. public company, including, but not limited to, our annual NASDAQ Capital Market fees, fees related to investor relations and costs of complying with Sarbanes-Oxley.
Research and Development Costs. Research and development costs, which consist of salaries, professional fees, and technical support fees, totaled $1,167,937 for the year ended December 31, 2009, as compared to $549,236 for the year ended December 31, 2008, an increase of approximately 112.6%. This increase is primarily attributable to increased research activities with certain outside experts and institutions with whom we cooperate on research and development of both existing and new products. We anticipate that our research and development costs will continue to increase as we continue improve existing products and develop new products.
Liquidity
Three Month Period Ended March 31, 2010
For the three months ended March 31, 2010, cash provided by operating activities was $1,908,957 compared to cash provided by operating activities of $133,395 for the same period in 2009. This increase is primarily attributable to improved accounts receivable collections and increased deposits received from customers, after adjusting for the non-cash charge of warrant revaluation, and offset by increase in inventory that reduced some of the positive cash flow. As of March 31, 2010, we had approximately 38 suppliers that we have made advances to in order to secure our raw materials and obtain favorable pricing.
We used $7,355,381 in investing activities for the three months ended March 31, 2010, as compared to $714,270 used in investing activities for the three months ended March 31, 2009. Net cash used in investing increased as a result of refundable deposits made in connection with potential acquisitions.
30
Cash used by financing activities was $298,244 for the three months ended March 31, 2010 as compared to $126,877 generated for the three months ended March 31, 2009. Cash used by financing activities for the three months ended March 31, 2010 was mainly for repayment of short-term loans. Cash provided by financing activities for the three months ended March 31, 2009 was mainly the result of proceeds from short-term loans.
As of March 31, 2010, we had cash of $5,911,415. Our total current assets were $27,100,696, and our total current liabilities were $4,336,729, which resulted in a net working capital of $22,763,967.
Year Ended December 31, 2009
For the twelve months ended December 31, 2009, cash provided by operating activities was $1,265,727 compared to cash provided by operating activities of $3,700,428 for the same period in 2008. For the year ended December 31, 2009, net cash provided by operating activities other than net income was primarily due to an increase of $3,662,661 in deposits and prepaid expenses. However, this decrease was offset by an increase of $898,213 in taxes payable and $499,158 in deposits from customers. Collectively this decreased cash used in operating activities by $2,434,701 for the year ended December 31, 2009 as compared to the year ended December 31, 2008. As of December 31, 2009 we had approximately forty six suppliers that we have made advances to in order to secure our raw materials and obtain favorable pricing.
We used $7,853,439 in investing activities for the year ended December 31, 2009, as compared to using $5,076,346 in investing activities for the year ended December 31, 2008. Net cash used in investing increased as a result of deposits made for two potential acquisitions as of December 31, 2009.
Cash provided by financing activities was $17,709,238 for the year ended December 31, 2009 as compared to $1,136,127 generated for the year ended December 31, 2008. Cash generated by financing activities for the year ended December 31, 2009 was the result of the financing that the Company completed in July 2009. Cash provided by financing activities for the year ended December 31, 2008 was the result of advances from shareholders and short term bank loans.
As of December 31, 2009, we had cash of $11,699,398. Our total current assets were $32,548,256, and our total current liabilities were $4,055,388, which resulted in a net working capital of $28,492,868.
Capital Resources
During the three months ended March 31, 2010, we made additional deposits of $5,499,375 for potential acquisitions. The deposits are refundable in the event the acquisitions cannot be completed. We also repaid $219,975 in short-term loans. The Company has sufficient capital for its operations. However, if we are to acquire another business or further expand our operations, we may need additional capital.
Plan of Operations
Over the next 12 months, we plan to continue to market and sell our current products and to develop new products.
In 2003, we received approval from the State Council of China to expand our production facilities and construct a new GMP standard plant. We have invested $10,501,000 into this project, which is our Huxian plant, including approximately $9,700,000 for the facilities and $800,000 for working capital. The construction work commenced in 2005, and we completed the veterinary medicine facility and the building that houses quality control, research and development and administration during 2007, both of which are fully operational. Construction of the micro-organism facility was completed in December 2009. The remaining facilities of the Huxian plant are expected to be completed in the second quarter of 2010, rather than the fourth quarter of 2009 as previously anticipated. After the completion of the construction phase, it typically takes 3 months for the Chinese Agriculture Ministry to issue the GMP certification. We anticipate that the new factory will generate sufficient cash flows; thus, management has concluded that there is no impairment loss on the construction in progress.
We believe that Xian Tianxing will be developing new products including animal immunization products, non-pathogenic micro-organisms for the cure and prevention of livestock disease, complex enzyme preparations as animal feed additives, and several new veterinary medicine products within the next 12 months.
31
Contractual Obligations and Off-Balance Sheet Arrangements
Contractual Obligations
| Payments Due by Period | ||||||||||||||||||||
| Contractual Obligations | Total | Less than 1 year | 1 – 3 years | 3 – 5 years | More than 5 years | |||||||||||||||
| R&D Project Obligation | $ | 908,396 | $ | 908,396 | $ | - | $ | - | $ | - | ||||||||||
| Operating Lease Obligations | 313,207 | 65,728 | 149,102 | 56,128 | 42,249 | |||||||||||||||
| Total | $ | 1,221,603 | $ | 974,124 | $ | 149,102 | $ | 56,128 | $ | 42,249 | ||||||||||
Off-Balance Sheet Arrangements
We do not have any outstanding financial guarantees or commitments to guarantee the payment obligations of any third parties. We have not entered into any derivative contracts that are indexed to our shares and classified as stockholders’ equity or that are not reflected in our consolidated financial statements. Furthermore, we do not have any retained or contingent interest in assets transferred to an unconsolidated entity that serves as credit, liquidity or market risk support to such entity. We do not have any variable interest in any unconsolidated entity that provides financing, liquidity, market risk or credit support to us or engages in leasing, hedging or research and development services with us.
Exchange Rates
Xian Tianxing maintains its books and records in Renminbi (“RMB”), the lawful currency of China. In general, for consolidation purposes, we translate Xian Tianxing’s assets and liabilities into US$ using the applicable exchange rates prevailing at the balance sheet date, and the statement of income is translated at average exchange rates during the reporting period. Adjustments resulting from the translation of Xian Tianxing’s financial statements are recorded as accumulated other comprehensive income.
The exchange rates used to translate amounts in RMB into US$ for the purposes of preparing the consolidated financial statements or otherwise stated in this MD&A were as follows:
March 31, 2010 | December 31, 2009 | March 31, 2009 | December 31, 2008 | |||||
| Assets and liabilities | USD0.14665: RMB1 | USD0.1467: RMB1 | USD0.1465: RMB1 | USD0.1467: RMB1 | ||||
| Statements of operations and cash flows for the period/year ended | USD0. 14670: RMB1 | USD0.14661: RMB1 | USD0.14651: RMB1 | USD0.14415: RMB1 |
No representation is made that RMB amounts have been, or would be, converted into US$ at the above rates.
Inflation
We believe that inflation has not had a material effect on our operations to date.
Related Party Transactions
For a description of our related party transactions see the section of this prospectus entitled “Certain Relationships and Related Transactions.”
32
BUSINESS
Corporate Organization and History
We were originally incorporated in Nevada under the name Hollywood Entertainment Network, Inc. on September 24, 1998, with a principal business objective to operate as an independent film company in the business of motion picture production and distribution. On May 23, 2000, we underwent a reverse merger and abandoned this enterprise to become a developer of computer security software and hardware and changed our name to The Cyber Group Network Corporation to reflect this change in business.
In September 2005, we executed a Share Exchange Agreement (“Exchange Agreement”) by and among R. Scott Cramer, Steve Lowe, David Wassung (all hereinafter collectively referred to as the “CGPN Stockholders”) and us on the one hand, and Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd., a Cayman Island Company (“Skystar Cayman”), and the stockholders of 100% of Skystar Cayman’s common stock (the “Skystar Cayman Stockholders”), on the other hand. (This transaction is referred to hereinafter as the “Share Exchange Transaction”). Under the Exchange Agreement, on the Closing Date, we issued shares of our Series B preferred stock (the “CGPN Shares”) to the Skystar Cayman Stockholders in exchange for 100% of the common stock of Skystar. From and after the closing of the Share Exchange Transaction in November of 2005, our primary operations consist of the operations of Skystar Cayman and we became a Chinese bio-pharmaceutical company that develops, manufactures and markets a wide range of veterinary healthcare and medical care products.
Skystar Cayman has no substantive operations of its own and conducts its primary business operations through its variable interest entity (“VIE”), Xian Tianxing Bio-Pharmaceutical Co., Ltd. (“Xian Tianxing”). Xian Tianxing holds the licenses and approvals necessary to operate veterinary healthcare and medical care products business in China. We have contractual arrangements with Xian Tianxing and its stockholders pursuant to which we provide technology consulting and other general business operation services to Xian Tianxing. Through these contractual arrangements, we also have the ability to substantially influence Xian Tianxing’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring stockholder approval. As a result of these contractual arrangements, which enable us to control Xian Tianxing, we are considered the primary beneficiary of Xian Tianxing. Accordingly, we consolidate Xian Tianxing’s results, assets and liabilities in our financial statements. For a description of these contractual arrangements, see “Contractual Arrangements with Xian Tianxing and its Stockholders.”
Xian Tianxing is a joint stock company incorporated in China. Mr. Weibing Lu, who is our Chief Executive Officer, is General Manager (equivalent to Chief Executive Officer) and Chairman of Xian Tianxing and, as of the date of this prospectus, owns approximately 41% of the issued and outstanding stock of Xian Tianxing.
In February of 2006, we changed of our corporate name to our current name, “Skystar Bio-Pharmaceutical Company;” effected a 397-for-1 reverse stock split; and decreased our authorized common stock from 500,000,000 to 50,000,000 shares.
In August of 2007, Shanghai Siqiang Biotechnological Company Limited (“Shanghai Siqiang”) was established in Putuo District, Shanghai. Xian Tianxing is the 100% shareholder of Shanghai Siqiang. Shanghai Siqiang serves as a research and development center for Xian Tianxing to engage in research, development, production and sales of feed additives and veterinary disease diagnosis equipments.
In October of 2007, we acquired all of the issued and outstanding shares of Fortunate Time International Limited (“Fortunate Time”), a Hong Kong company owned 100% by Mr. R. Scott Cramer, a member of our board of directors. In July of 2007, Fortunate Time established Sida Biotechnology (Xian) Co., Ltd. (“Sida”) in the High Technology District in Xi’an as its wholly owned China subsidiary. As the wholly owned subsidiary of a non-Chinese company (Fortunate Time), Sida is deemed a wholly foreign owned enterprise (“WFOE”). Other than its holding of Sida, Fortunate Time does not conduct any business, and its management consists solely of Mr. Wei Wen as Director. As described below under “Recent Developments with Respect to the Contractual Arrangements with Xian Tianxing and its Shareholders”, Sida’s principal business is to carry out the terms of the contractual arrangements with Xian Tianxing and its shareholders.
On May 12, 2009, we effected a 1-for-10 reverse stock split of our issued and outstanding shares of common stock and a proportional reduction of our authorized shares of common stock from 200,000,000 to 20,000,000.
On September 18, 2009, Skystar Bio-Pharmaceutical Inc. (“Skystar California”) was incorporated in California and became our wholly-owned subsidiary.
On November 16, 2009, we effected a 2-for-1 forward stock split of the Company’s issued and outstanding shares of common stock and a proportional increase of the Company’s authorized shares of common stock from 20,000,000 to 40,000,000.
33
Industry and Market Overview
Management believes there is significant demand for veterinary medicines and vaccines in China. Statistics from the Chinese Ministry of Agriculture show that China vaccinated six billion poultry and 850 million livestock in the first half of 2006. According to the Chinese Ministry of Agriculture, the addressable market in China in 2004 for veterinary, livestock and poultry vaccines was over 70 billion doses; however the market supply was only 32 billion doses.
We also believe that there is a substantial market for micro-organisms which are fed to animals and result in healthier livestock and reduced feed requirements for our customers. According to the Chinese Ministry of Agriculture, the addressable market in China in 2004 for such micro-organisms was 3 million tons, while the supply output was only 200,000 tons.
Principal Products or Services and Their Markets
Currently, we have four product lines:
| · | Our vaccine line currently includes 10 products; |
| · | Our veterinary medicine line for poultry and livestock currently includes 159 products; |
| · | Our fodder and feed additives line currently includes 10 products; and |
| · | Our microorganism products line currently includes 16 products. |
Our top selling product is Praziquantel tablets. It is an anthelmintic effective against schistosomiasis. Schistosomiasis is a parasitic disease that affects both animals and humans. Our product only treats animals. Schistosomiasis is prevalent in Southern China where there are a lot of fresh water lakes and rivers. The company has seen tremendous growth in the sales of the Praziquantel tablets. Of total revenues from veterinary medications during the three months March 31, 2010, approximately $830,600, or 17% of total revenues, resulted from the sale of Praziquantel tablets. Another prominent product of ours is a vaccine that is designed to prevent the onset of avian coccidiosis disease, a parasitic and highly contagious gastrointestinal disease affecting chicken and other poultry. Avian coccidiosis is a common parasitic disease and is the number one cause of death among chickens and poultry. The U.S. Department of Agriculture estimates that avian coccidiosis costs the worldwide poultry industry $3 billion in treatment expenses, bird losses and unmarketable birds due to low bird weight. Our DLV chicken vaccine has won a number of accreditation and awards from the Chinese central authorities. The vaccine was licensed as a “National Second Level New Veterinary Drug” which entitled the product to Chinese regulatory protection and gave us five years of exclusive production rights. It is safe, effective and easy to administer, and has proved to save costs by 60% as compared to using chemical medicines such as sulfaquinoxaline sodium and salinomycin sodium. Additionally, with our ongoing research projects, management expects that we will continue to introduce new products to the market.
Distribution Methods of the Products or Services and Our Customers
We have a distribution network covering 29 provinces in China. As of March 31, 2010, we had over 1,539 distributors and 529 direct customers. We intend to establish more representative offices and engage additional distribution agents in order to strengthen our distribution network.
We recognize the importance of branding as well as packaging. All of our products bear a uniform brand but we also brand and package our products with specialized designs to differentiate the different categories of our products.
We conduct promotional marketing activities to publicize and enhance our image as well as to reinforce the recognition of our brand name, including:
| · | publishing advertisements and articles in national as well as specialized and provincial newspapers, magazines, and in other media, including the Internet; |
| · | participating in national meetings, seminars, symposiums, exhibitions for veterinary healthcare and medical care products and other related industries; |
| · | organizing cooperative promotional activities with distributors; and |
| · | sending direct mail to major farms. |
Competition
We have three major competitors in China: Jielin Bio-Tech Production Co., Ltd., Qilu Animal Health Production Co., Ltd., and Zhongmu Industrial Joint Stock Co., Ltd. These companies have more assets, resources and a larger market share. We believe we are able to compete with these competitors because of our location in Northwest China, our unique products and our lower prices. Other than these three competitors, most of our other competitors produce only one or two products. We on the other hand, have four product lines, including complete series of feed additives and veterinary medicine products designed to treat the animal from the time of birth to the time the animal is ready for the market.
34
Sources and Availability of Raw Materials and the Principal Suppliers
Xi’an Yanghua Chemical Co., Ltd., Xi’an Nanchen Trading Co., Ltd. and Xi’an Fandike Chemical Technology Co., Ltd. collectively supplied over fifty-seven percent (57%) of the raw materials we used to manufacture our products. We design, create prototypes and manufacture our products at our manufacturing facilities located at Xi’an city, Shaanxi Province, China. Our principal raw materials include various chemical compounds including dexamethasone sodium phosphate (a glucocorticoid with anti-inflammatory property), stachyose (a tetrasaccharide found naturally in many vegetables) and thiamphenicol (an antibiotic). We also use Chinese herbs such as Huoxiang, Huanglian, and Zhang Red Flowers as raw materials, which are supplied to us by Wan Shou Bei Lu Zhong Kui Cao Yao Xing. None of our current products requires any raw materials that are scarce, and our raw materials generally are readily available from a wide range of sources. Accordingly, we do not have any continuing or long term supply agreements with any of these suppliers, and purchase our raw materials from them on a per purchase order basis. The prices for these raw materials are nevertheless subject to market forces largely beyond our control, including energy costs, organic chemical feedstock, market demand, and freight costs. The prices for these raw materials have varied significantly in the past and may vary significantly in the future.
As a result of our research and development efforts in 2007 in cooperation with research institutes including Shaanxi Microbial Research Institute, Jiangsu Microbial Research Institute, China Northwestern University and China Northwest A&F University, we now also internally produce microbial strains, which are key components of our micro-organism products. Our ability to produce microbial strains has translated into a significant cost reduction for these raw materials.
Patents, Trademarks, Licenses, Franchises, Concessions, Royalty Agreements or Labor Contracts
We rely on a combination of trademark, copyright and trade secret protection laws in China and other jurisdictions, as well as confidentiality procedures and contractual provisions to protect our intellectual property and our brand. In November 2000, the Chinese Ministry of Agriculture issued Xian Tianxing a non-renewable license for our DLV chicken vaccine as a “National Second Level New Veterinary Drug,” which entitled this product to Chinese regulatory protection and gave us five years of exclusive production rights. Although our license is non-renewable and has expired since November 2005, we have and will continue to enjoy exclusivity until such time that the vaccine is formally listed on the Chinese Pharmacopoeia by the Chinese Pharmacopoeia Commission. Only after the vaccine is listed can other companies begin to apply for approval by the Chinese Ministry of Agriculture to manufacture and distribute the vaccine. Thus, until the listing occurs, we remain the only company in China legally permitted to produce and sell the vaccine. We do not know, however, if and when the listing will occur.
We intend to seek other licenses or apply for patents as necessary in order to protect our rights, and we also enter into confidentiality, non-compete and invention assignment agreements with our employees and consultants and nondisclosure agreements with third parties. “Jiateng Jun” and “Star Pigeon” are our registered trademarks in China.
Additionally, Xian Tianxing is approved by the Chinese Ministry of Agriculture for the manufacture and distribution of 103 types of veterinary drugs. Such approvals certify Xian Tianxing’s products as conforming to government-mandated standards. The approvals are issued for a period of 5 years and may be renewed 6 months prior to their expiration date. The 103 veterinary drugs and their approval numbers are listed below:
| Veterinary Drug Products | Approval Number | |
| Metamizole Sodium Injection | Veterinary Drug (2007) 270261152 | |
| Antondine Injection | Veterinary Drug (2007) 270261160 | |
| Dexamethasone Sodium Phosphate Injection | Veterinary Drug (2007) 270262530 | |
| Enrofloxacin Injection | Veterinary Drug (2007) 270262518 | |
| Compoumd Vitamin B Injection | Veterinary Drug (2007) 270264572 | |
| Sulfamonomethoxine Sodium Injection | Veterinary Drug (2007) 270261616 | |
| Sulfadiazine Sodium Injection | Veterinary Drug (2007) 270261634 | |
| Kanamycin Sulfate Injection | Veterinary Drug (2007) 270261211 | |
| Gentamycin Sulfate Injection | Veterinary Drug (2007) 270261507 | |
| Gentamycin Micronomicin Sulfate Injection (10 ml:100,000 parts) | Veterinary Drug (2007) 270262751 | |
| Gentamycin Micronomicin Sulfate Injection (10ml: 200,000 parts) | Veterinary Drug (2007) 270262752 | |
| Mequindox Injection (10ml:0.5g) | Veterinary Drug (2007) 270261174 | |
| Mequindox Injection (10ml:0.2g) | Veterinary Drug (2007) 270264644 | |
| Vitamin C Injection | Veterinary Drug (2007) 270262795 | |
Vitamin B1 Injection | Veterinary Drug (2007) 270261389 |
35
| Lincomycin Hydrochloride Injection (10ml:0.3g) | Veterinary Drug (2007) 270262614 | |
| Lincomycin Hydrochloride Injection (10ml:1.5g) | Veterinary Drug (2007) 270262616 | |
| Danofloxacin Mesylate Powder | Veterinary Drug (2008) 270262036 | |
| Ofloxacin Injection | Veterinary Drug (2007) 270262126 | |
| Norfloxacin Nicotinate Injection | Veterinary Drug (2007) 270262593 | |
| Ciprofloxacin Hydrochloride Injection | Veterinary Drug (2007) 270262160 | |
| Pefloxacin Mesylate Granules | Veterinary Drug (2007) 270262042 | |
| Praziquantel Tablets | Veterinary Drug (2007) 270261174 | |
| Compound Sulfamethoxazole Tablets | Veterinary Drug (2007) 270261612 | |
| Ofloxacin Tablets | Veterinary Drug (2007) 270262123 | |
| Amoxicillin Soluble Powder | Veterinary Drug (2007) 270261199 | |
| Avermectin Powder | Veterinary Drug (2007) 270262066 | |
| Diclazuril Premix (0.2%) | Veterinary Drug (2007) 270261140 | |
| Diclazuril Premix (5%) | Veterinary Drug (2007) 270262528 | |
| Florfenicol Powder | Veterinary Drug (2007) 270262110 | |
| Compound Amoxicillin Powder | Veterinary Drug (2007) 270262092 | |
| Thiamphenicol Powder | Veterinary Drug (2007) 270262722 | |
| Erythromycin Thiocyanate Soluble Powder | Veterinary Drug (2007) 270261492 | |
| Apramycin Sulfate Soluble Powder | Veterinary Drug (2007) 270262745 | |
| Neomycin Sulfate Soluble Powder | Veterinary Drug (2007) 270262755 | |
| Colistin Sulfate Soluble Powder | Veterinary Drug (2007) 270262758 | |
| Salinomycin Sodium Premix | Veterinary Drug (2007) 270261379 | |
| Ciprofloxacin Hydrochloride Soluble Powder | Veterinary Drug (2007) 270262159 | |
| Spectinomycin Hydrochloride and Lincomycin Hydrochloride Soluble Powder | Veterinary Drug (2007) 270262602 | |
| Ofloxacin Soluble Powder | Veterinary Drug (2007) 270262124 | |
| Baitouweng San | Veterinary Drug (2007) 270265053 | |
| Baotai Wuyou San | Veterinary Drug (2007) 270265111 | |
| Chulijing | Veterinary Drug (2007) 270265192 | |
| Danjibao | Veterinary Drug (2007) 270265171 | |
| Feizhucai | Veterinary Drug (2007) 270265100 | |
| Fuzheng Jiedu San | Veterinary Drug (2007) 270265076 | |
| Gongying San | Veterinary Drug (2007) 270265028 | |
| Houyanjing San | Veterinary Drug (2007) 270265179 | |
| Huanglian Jiedu San | Veterinary Drug (2007) 270265178 | |
| Jianji San | Veterinary Drug (2007) 270265133 | |
| Jianwei San | Veterinary Drug (2007) 270265134 | |
| Jingfang Baidu San | Veterinary Drug (2007) 270265127 | |
| Mubin Xiaohuang San | Veterinary Drug (2007) 270265035 | |
| Qingfei Zhike San | Veterinary Drug (2007) 270265157 | |
| Qingshu San | Veterinary Drug (2007) 270265162 | |
| Qingwen Baidu San | Veterinary Drug (2007) 270265165 | |
| Quchong San | Veterinary Drug (2007) 270265089 | |
| Tongru San | Veterinary Drug (2007) 270265156 | |
| Xiaoji San | Veterinary Drug (2007) 270265146 | |
| Yimu Shenghua San | Veterinary Drug (2007) 270265148 | |
| Yujin San | Veterinary Drug (2007) 270265102 | |
| Zhili San | Veterinary Drug (2007) 270265037 | |
| Compound Sulfamethoxydiazine Sodium Injection | Veterinary Drug (2007) 270261608 |
| Lomefloxacin Hydrochloride Soluble Powder | Veterinary Drug (2008) 270262166 | |
| Danofloxacin Mesylate Injection | Veterinary Drug (2008) 270262033 | |
| Sulfathiazole Sodium Injection | Veterinary Drug (2008) 270261645 | |
| Buzhong Yiqi San | Veterinary Drug (2008) 270265082 | |
| Fangji San | Veterinary Drug (2008) 270265072 | |
| Shenling Baishu San | Veterinary Drug (2008) 270265093 | |
| Qibu San | Veterinary Drug (2008) 270265220 | |
| Sulfaquinoxaline Sodium Soluble Powder (10%) | Veterinary Drug (2008) 270261624 | |
| Sulfaquinoxaline Sodium Soluble Powder (5%) | Veterinary Drug (2008) 270262580 | |
| Fenbendazole Powder | Veterinary Drug (2008) 270261189 |
36
| Sulfachloropyrazin Sodium Soluble Powder | Veterinary Drug (2008) 270262703 | |
| Huoxiang Zhengqi San | Veterinary Drug (2008) 270265200 | |
| Cuiqing San | Veterinary Drug (2008) 270265188 | |
| Longdan Xiegan San | Veterinary Drug (2008) 270265057 | |
| Maxing Shigan San | Veterinary Drug (2008) 270265174 | |
| Qumai San | Veterinary Drug (2008) 270265067 | |
| Shengru San | Veterinary Drug (2008) 270265051 | |
| Xiaoshi Pingwei San | Veterinary Drug (2008) 270265145 | |
| Xiaochaihu San | Veterinary Drug (2008) 270265018 | |
| Yinqiao San | Veterinary Drug (2008) 270265172 | |
| Pefloxacin Mesylate Injection | Veterinary Drug (2008) 270262665 | |
| Enrofloxacin Injection (10ml:250mg) | Veterinary Drug (2008) 270261295 | |
| Florfenicol Injection | Veterinary Drug (2008) 270262546 | |
| Lomefloxacin Hydrochloride Injection | Veterinary Drug (2008) 270262169 | |
| Berberine Sulfate Injection | Veterinary Drug (2008) 270264595 | |
| Gentamycin Sulfate Injection (10ml:0.2g) | Veterinary Drug (2008) 270261506 | |
| Promethazine Hydrochloride Injection | Veterinary Drug (2008) 270262126 | |
| Bailong San | Veterinary Drug (2008) 270265055 | |
| Feizhu San | Veterinary Drug (2009) 270265101 | |
| Ivermectin Premix | Veterinary Drug (2009) 270263059 | |
| Kitasamycin Premix | Veterinary Drug (2008) 270262043 | |
| Pefloxacin Mesylate Soluble Powder | Veterinary Drug (2008) 270262040 | |
| Ciprofloxacin Lactate Soluble Powder | Veterinary Drug (2008) 270262073 | |
| Norfloxacin Nicotinic Soluble Powder | Veterinary Drug (2008) 270262178 | |
| Tylosin Tartrate Soluble Powder | Veterinary Drug (2008) 270262731 | |
| Lincomycin Hydrochloride Soluble Powder | Veterinary Drug (2008) 270262620 | |
| Tilmicosin Premix | Veterinary Drug (2010) 270262263 | |
| Ciprofloxacin Hydrochloride Soluble Powder (5%) | Veterinary Drug (2010) 270262605 | |
| Pefloxacin Mesylate Soluble Powder (10%) | Veterinary Drug (2010) 270262727 | |
| Ivermectin Injection (10ml:0.1g) | Veterinary Drug (2010) 270262646 |
Bio-pharmaceutical companies are at times involved in litigation based on allegations of infringement or other violations of intellectual property rights. Furthermore, the application of laws governing intellectual property rights in China and abroad is uncertain and evolving and could involve substantial risks to us.
Government Approval and Regulation of Our Principal Products or Services
Government approval is required for the production of veterinary healthcare and medical care products. The Chinese Ministry of Agriculture has granted us three government permits to produce the following products: Forage Additive Products, Additive and Mixed Forage Products and Veterinary Medicine Products. For the production of the veterinary medicine, there is a national standard known as the GMP standard. A company must establish its facility according to GMP standards, including both the facility and the production process. After establishing such facility, we filed an application to operate the facility with the Ministry of Agriculture of China government, which then sends a team of specialists to conduct an on-site inspection of the facility. A company cannot start production at the facility until it receives approval from the Ministry of Agriculture to begin operations. We currently have obtained the requisite approval and licenses from the Ministry of Agriculture in order to operate our production facilities. Our current permit is valid from 2005 through 2010 for Feed Additive. A renewal application has already been filed. Our veterinary medicine permit is valid from 2007 through 2012. Our vaccine line is operating under the original license that was granted to the company recognizing the DLV vaccine as the National Second Level New Veterinary Drug.
Research and Development
We place great emphasis on product research and development, and are currently working closely with a number of research institutes in the veterinary science field.
In July 2005, we entered into a cooperation agreement with Shaanxi Microbial Institute, the only microbial research institute in northwest China, pursuant to which we established our Huxian research and development center to facilitate opportunities for us to develop commercially viable products based on the Institute’s research conducted at our research center. Under the cooperation agreement, we provide for the running and operation of the research center, including research equipment and materials. In exchange, we have exclusive rights to any technology derived from any research project that we solely fund. The cooperation agreement also provides for our mutual staffing of research personnel at, and joint-appointment of the director for, the research center. The Institute, however, is not obligated to us with respect to a specific amount of time or a specific project under the cooperation agreement. Currently, we are undertaking the following projects at this research center:
37
| · | Development of protein technology and enzyme mechanism. Introducing the technology in polypeptides, we are working to develop new products to cure piglet diarrhea. The products are expected to stimulate the release of growth hormones in piglets, improve their ability to produce antibody and excrete stomach acidity, enhance the activity of albumen enzyme and adjust the activity of T.B. cells, thereby improving their all-around disease-resistance ability. We expect these new products will greatly reduce the use of traditional chemical drugs and lead to more environmentally-friendly livestock raising. These products are now in the interim stage of development. We are also developing complex enzyme preparations as new feed additives and aim to use anti-inflammatory enzyme, polyase, and cellulose to form the best combination to effectively dissolve and cause the additive to be absorbed in the feed. Our goal is to drastically improve the absorption rate of the feed, thereby reducing the ratio of usage of feed versus meat, while concurrently reducing the incidence of disease in livestock and poultry. We are looking to outsource certain aspects of these research projects to Shaanxi Jiuzhou Biotechnology Co., Ltd., a member of Shaanxi Jiuzhou Biomedicine Park, although we have not entered into any definitive agreement. |
| · | Development of non-pathogenic micro-organisms. We are also developing, in cooperation with the Institute, non-pathogenic micro-organisms and, based upon current products of microbe preparations, lactobacillus, bacillus, bifid bacterium baceroid, and combined with the most appropriate oligosaccharide preparations to produce living bacterium which will be applied to cure gastrointestinal tract diseases resulting from the maladjustment of flora. If successful, micro-organism preparations can be effective cure and prevention for livestock disease, and can greatly reduce the use of antibiotic and other drugs. |
At the Skystar Research and Development Center in Shanghai, we have an arrangement with Shanghai Poultry Verminosis Institution of the Chinese Academy of Agricultural Sciences that is similar to our agreement with Shaanxi Microbial Institute, although we have not entered into any written agreement with the Institution. Currently, we are undertaking the following projects at this research center:
| · | Development of new products for animal immunization by employing new technologies in micro-organism and bacterium. We expect to be placing greater resources into our research and development with the Institution of toxoid, multivalent inactivated vaccines and attenuated live vaccine, which we believe will gradually replace traditional chemical drugs and which will greatly impact the animal vaccination industry. |
| · | Development of veterinary medicines for pets. We believe that markets for pet-related products, including vaccines have been experiencing growth at a rate reflective of the growth rate for the general economy in China. We believe that this niche market is being overlooked by local manufacturers. To attempt to take advantage of this opportunity, we have over 20 products of veterinary medicines for pets that are in the course of development. |
During the first quarter of 2008, Xian Tianxing contracted with Northwestern Agricultural Technology University to jointly work on an R&D project concerning the application of nano-technology in the prevention of major milk cow disease. The project reached trial stage in March 2009 and we expect to obtain veterinary permit for the new product from government sometime in 2010.
In February 2008, Xian Tianxing signed a research and development agreement with Shanxi Shenzhou Bio-pharmaceuticals Technology Company, to jointly develop a GHRH (growth-hormone-releasing hormone) analog with sustained release preparation. The medicine is used to treat postweaning multisystemic wasting syndrome (PWSD) afflicting early weaned pigs. Currently the product is under clinical trials and we are preparing the documentation needed for the approval application.
On September 23, 2009, we purchased an exclusive aquaculture vaccine technology from and signed a collaborative research and development agreement with China’s Fourth Military Medical University (“FMMU”) for RMB 8 million (approximately $1.2 million), granting the us exclusive rights to sell and market the patented aquaculture vaccine through 2020. Under this patented technology, and in collaboration with FMMU, we will produce the first vaccine in China designed to safely prevent and treat certain bacterial infection and diseases in marine life without causing harmful side effects. Based on its first-to-market status, the Chinese Ministry of Agriculture has issued a Grade I Veterinary Certificate for our vaccine.
We have also worked with Northwest A&F University in Shaanxi Province and Jiangsu Institute of Microbiology in the past, and we continue to look for opportunities to collaborate with these and other research institutes in the future.
In 2009, we spent $1,167,937, or approximately 3.5% of our revenue, on research and development of products. For the three months ended March 31, 2010, we spent $43,995, or approximately 1% of our revenue, on research and development of products.
38
Costs and Effects of Compliance with Environmental Laws
In compliance with Chinese environmental regulations, we spent approximately $30,000 in 2009 and $4,000 for the three months ended March 31, 2010, mainly for the wastewater treatment in connection with our production facilities.
Employees
As of March 31, 2010, we have approximately 231 employees, of which 217 are full time employees. As of December 31, 2009, we had 214 employees, of which 169 worked as full time employees. None of these employees are represented by any collective bargaining agreements. We have not experienced a work stoppage. Management believes that our relations with our employees are good.
Property
Our administrative headquarters are currently located in approximately 3,700 square feet of office space at Rm. 10601, Jiezuo Plaza, No.4, Fenghui Road South, Gaoxin District, Xi’an, Shaanxi Province, China. This property belongs to Mr. Weibing Lu, our director and chief executive officer. This property was provided free for the use of our administrative division in 2006 and 2005. In January 2007, we entered into a 5-year lease agreement with Mr. Lu for the premises on terms of RMB 165,600 (approximately $24,000) per year.
Shanghai Siqiang, wholly owned subsidiary of Xian Tianxing, leases its office and facility space in Shanghai from Mr. Lu pursuant to a 10-year lease agreement entered into in August 2007 on terms of RMB 144,000 (approximately $21,000) per year.
Currently, Xian Tianxing has two manufacturing plants located in Xi’an, Shaanxi Province, China. One plant is in the town of at Sanqiao and the other in the town of Huxian.
The Sanqiao Plant
Xian Tianxing entered into a tenancy agreement for the lease of factory premises underlying its Sanqiao plant for a period of 10 years from October 1, 2004 to September 30, 2014. The annual rent for the factory premises is $13,503 (RMB 94,600) and is also subject to a 10% increase every four subsequent years. The plant facilities include:
| (1) | Micro-Organism Plant. This production plant is run in cooperation with experts from Japan Kato Microbiology Institute, Microbiology Institute of Shaanxi Province and Northwest Agro-Forestry Sci-tech University. This facility was expanded in 2007 from approximately 16,100 square feet to approximately 21,500 square feet in accordance with Chinese national Good Manufacturing Practice (“GMP”) standards, and has been issued production permit and certain product approval numbers by the Chinese Ministry of Agriculture. |
| (2) | Feed Additive Plant. This production facility occupies an area of approximately 10,700 square feet. |
The Huxian Plant
In 2003, Xian Tianxing received approval from the State Council of China to expand its production facilities and construct a new GMP standard plant. In connection with the approval, Xian Tianxing acquired a long-term land use right for the land now underlying its Huxian plant. Our total investment in this project thus far is estimated at $10,501,000. Because Xian Tianxing has been accredited as a high-tech enterprise, its Huxian plant has the full support of both the Shaanxi provincial government and the Xi’an municipal government.
Construction of the Huxian plant commenced in late 2004 and parts of the plant has been fully operational since the end of the second quarter of 2007. Remaining construction work is expected to be completed in the second quarter of 2010. The plant site will occupies approximately 7.7 acres and the plant facilities have a total area of approximately 151,700 square feet. The primary plant facilities and their status as of March 31, 2010 are as follows:
| Description | Approximate Size | Status | ||
| GMP standard veterinary medicine facility | 45,200 square feet | Completed | ||
| Quality control, research and development, and administration building | 36,600 square feet | Completed | ||
| GMP standard bio-pharmaceutical facility with three production lines for active bacteria, inactivated vaccines, and coccidiosis vaccines | 59,201 square feet | Construction completed in December 2009 | ||
| Animal laboratory complying with Animal Bio-safety Level 2 (ABSL-2) requirements | 10,700 square feet | Completion expected in the second quarter of 2010 |
39
We believe that the general physical condition of the plants and production facilities of the company can completely satisfy our current production orders of the company in terms of quantity and production quality.
We believe that these facilities after construction is completed will be able to meet our operational needs for three to five years.
Contractual Arrangements with Xian Tianxing and its Stockholders
Our relationships with Xian Tianxing and its stockholders are governed by a series of contractual arrangements, as we (including our direct and indirect subsidiaries) do not own any equity interests in Xian Tianxing. Under Chinese laws, each of Skystar Cayman and Xian Tianxing is an independent legal entity and neither of them is exposed to liabilities incurred by the other party. Other than pursuant to the contractual arrangements between Skystar Cayman and Xian Tianxing, Xian Tianxing does not transfer any other funds generated from its operations to Skystar Cayman. Skystar Cayman entered into these contractual arrangements with Xian Tianxing in October 2005. In January 2008, Skystar Cayman assigned its rights under these contractual arrangements to our indirect wholly-owned subsidiary, Sida.
On October 28, 2005, we entered into the following contractual arrangements:
Consulting Services Agreement. Pursuant to the exclusive consulting services agreement between Skystar Cayman and Xian Tianxing, Skystar Cayman has the exclusive right to provide to Xian Tianxing general services related to veterinary healthcare and medical care products business operations as well as consulting services related to the technological research, development, design and manufacturing of veterinary healthcare and medical care products (the “Services”). Skystar Cayman also sends employees to Xian Tianxing for whom Xian Tianxing bears the costs and expenses. Under this agreement, Skystar Cayman owns the intellectual property rights developed or discovered through research and development providing the Services for Xian Tianxing. Xian Tianxing pays quarterly consulting service fees in Renminbi (“RMB”) to Skystar Cayman that is equal to all of Xian Tianxing’s revenue for such quarter. The consulting services agreement is in effect unless and until terminated by written notice of either party in the event that: (a) the other party causes a material breach of this agreement, provided that if the breach does not relate to a financial obligation of the breaching party, that party may attempt to remedy the breach within 14 days following the receipt of the written notice; (b) the other party becomes bankrupt, insolvent, is the subject of proceedings or arrangements for liquidation or dissolution, ceases to carry on business, or becomes unable to pay its debts as they become due; (c) Skystar Cayman terminates its operations; (d) Xian Tianxing’s business license or any other license or approval for its business operations is terminated, cancelled or revoked; or (e) circumstances arise which would materially and adversely affect the performance or the objectives of the consulting services agreement. Additionally, Skystar Cayman may terminate the consulting services agreement without cause.
Operating Agreement. Pursuant to the operating agreement among Skystar Cayman, Xian Tianxing and the stockholders of Xian Tianxing who collectively hold the majority of the outstanding shares of Xian Tianxing (collectively “Xian’s Majority Stockholders”), Skystar Cayman provides guidance and instructions on Xian Tianxing’s daily operations, financial management and employment issues. The stockholders of Xian Tianxing must designate the candidates recommended by Skystar Cayman as their representatives on Xian Tianxing’s board of directors. Skystar Cayman has the right to appoint senior executives of Xian Tianxing. In addition, Skystar Cayman agrees to guarantee Xian Tianxing’s performance under any agreements or arrangements relating to Xian Tianxing’s business arrangements with any third party. Xian Tianxing, in return, agrees to pledge its accounts receivable and all of its assets to Skystar Cayman. Moreover, Xian Tianxing agrees that without the prior consent of Skystar Cayman, Xian Tianxing will not engage in any transactions that could materially affect the assets, liabilities, rights or operations of Xian Tianxing, including, without limitation, incurrence or assumption of any indebtedness, sale or purchase of any assets or rights, incurrence of any encumbrance on any of its assets or intellectual property rights in favor of a third party or transfer of any agreements relating to its business operation to any third party. The term of this agreement is ten (10) years from October 28, 2005 and may be extended only upon Skystar Cayman’s written confirmation prior to the expiration of this agreement, with the extended term to be mutually agreed upon by the parties.
Equity Pledge Agreement. Under the equity pledge agreement between the stockholders of Xian Tianxing and Skystar Cayman, the stockholders of Xian Tianxing pledged all of their equity interests in Xian Tianxing to Skystar Cayman to guarantee Xian Tianxing’s performance of its obligations under the technology consulting agreement. If Xian Tianxing or Xian’s Majority Stockholders breaches its respective contractual obligations, Skystar Cayman, as pledgee, will be entitled to certain rights, including but not limited to the right to sell the pledged equity interests, the right to vote and control the pledged assets. The Xian Majority Stockholders also agreed that upon occurrence of any event of default, Skystar Cayman shall be granted an exclusive, irrevocable power of attorney to take actions in the place and stead of the Xian Stockholders to carry out the security provisions of the equity pledge agreement and take any action and execute any instrument that Skystar Cayman may deem necessary or advisable to accomplish the purposes of the equity pledge agreement. The stockholders of Xian Tianxing agreed not to dispose of the pledged equity interests or take any actions that would prejudice Skystar Cayman’s interest. The equity pledge agreement will expire two (2) years after Xian Tianxing obligations under the exclusive consulting services agreement have been fulfilled.
40
Option Agreement. Under the option agreement between the stockholders of Xian Tianxing and Skystar Cayman, the stockholders of Xian Tianxing irrevocably granted Skystar Cayman or its designated person an exclusive option to purchase, to the extent permitted under Chinese law, all or part of the equity interests in Xian Tianxing for the cost of the initial contributions to the registered capital or the minimum amount of consideration permitted by applicable Chinese law. Skystar Cayman or its designated person has sole discretion to decide when to exercise the option, whether in part or in full. The term of this agreement is ten (10) years from October 28, 2005 and may be extended prior to its expiration by written agreement of the parties.
Proxy Agreement. Pursuant to the proxy agreement among Skystar Cayman, Xian’s Majority Stockholders, and Xian Tianxing, Xian’s Majority Stockholders agreed to entrust all the rights to exercise their voting power to the person(s) appointed by Skystar Cayman. The parties entered into a proxy agreement where Xian’s Majority Stockholders have agreed to irrevocably grant a person to be designated by Skystar Cayman with the right to exercise Xian’s Majority Stockholders’ voting rights and their other rights including the attendance at and the voting of Xian’s Majority Stockholders’ shares at the stockholder’s meetings (or by written consent in lieu of meetings) in accordance with applicable laws and its Article of Association, including but not limited to the rights to sell or transfer all or any of his equity interests of the Company, and appoint and vote for the directors and Chairman as the authorized representative of the stockholders of Company. The term of this Proxy Agreement is ten (10) years from October 28, 2005 and may be extended prior to its expiration by written agreement of the parties.
While the consulting services agreement remains in effect unless terminated by Skystar Cayman with or without cause (or by Xian Tianxing for cause), the operating agreement, the option agreement and the proxy agreement all terminate on October 28, 2015 unless extended by written agreement of the parties. Under their terms, we (through our direct and indirect wholly owned subsidiaries) hold the power to renew the operating agreement, the option agreement and the proxy agreement, rather than Xian Tianxing. Thus, these agreements will renew so long as we wish to maintain our relationship with Xian Tianxing under the consulting services agreement, and Xian Tianxing has no power to reject the renewal. Accordingly, the different termination terms of the consulting services agreement as compared to the operating agreement, option agreement and proxy agreement has no impact on us and our business operations.
Transfer of the Contractual Arrangements with Xian Tianxing and its Shareholders
On March 10, 2008, we were made a party to a series of agreements (collectively the “Transfer Agreements”) transferring the contractual arrangements governing the relationship among Skystar Cayman, Xian Tianxing and the majority shareholders of Xian Tianxing. Pursuant to the Transfer Agreements, from and after March 10, 2008, all of the rights and obligations of Skystar Cayman under the contractual arrangements were transferred to Sida. We were made a party to the Transfer Agreements for the sole purpose of acknowledging the Transfer Agreements. In effect, Skystar Cayman assigned the contractual rights it had with Xian Tianxing to an indirectly wholly-owned subsidiary, Sida.
Under our corporate structure with the contractual arrangements, the ability to transfer funds to and from Xian Tianxing expeditiously through a foreign currency bank account is necessary for our business operations. Under current applicable Chinese law, only a company that is classified as either a wholly foreign owned enterprise (WFOE) or a Sino-foreign joint venture may maintain a foreign currency bank account. Because Sida is wholly owned by Fortunate Time, a Hong Kong company, Sida is deemed a WFOE and may therefore maintain a foreign currency account. The Transfer Agreements amend the contractual arrangements so that funds are required to be transferred to and from Xian Tianxing through Sida’s foreign currency account and, through Sida, allow the Company to continue to control Xian Tianxing and its business operations.
The Transfer Agreements have transferred all of the rights and obligations of Skystar Cayman under the contractual arrangements to Sida. Thus, pursuant to the Amendment to Consulting Services Agreement, Sida now provides exclusive technology and general business consulting services to Xian Tianxing in exchange of a consulting fee equivalent to all of Xian Tianxing’s revenue; pursuant to the Amendment to Equity Pledge Agreement, Xian Tianxing’s majority shareholders now pledge their equity interests in Xian Tianxing to Sida; pursuant to the Agreement to Transfer of Operating Agreement, Sida now provides guidance and instructions on Xian Tianxing’s daily operations, financial management and employment issues; pursuant to the Designation Agreement, Xian Tianxing’s majority shareholders have entrusted all the rights to exercise their voting power to appointee(s) of Sida; and pursuant to the Agreement to Transfer of Option Agreement, Xian Tianxing’s majority shareholders have irrevocably granted Sida an exclusive option to purchase, to the extent permitted under Chinese law, all or part of their equity interests in Xian Tianxing.
41
The Transfer Agreements and the transfer of the rights and obligations of Skystar Cayman under the contractual arrangements to Sida comply with applicable Chinese law and do not in any way affect our business operations. Additionally, we believe that Xian Tianxing’s status as a VIE under FASB Interpretation No. 46R (“FIN 46R”), “Consolidation of Variable Interest Entities, an Interpretation of ARB No. 51”, is unaffected by the Transfer Agreements. Under the contractual arrangements, we viewed Xian Tianxing as a VIE of Skystar Cayman because the contractual arrangements obligated Skystar Cayman to absorb a majority of the risk of loss from Xian Tianxing’s activities and enabled Skystar Cayman to receive a majority of its expected residual returns. The Transfer Agreements merely substitute Skystar Cayman with Sida, an indirect wholly owned subsidiary of Skystar Cayman, such that the equity investors of Xian Tianxing continue to not have the characteristics of a controlling financial interest (just as under the contractual arrangements) and we continue to be the primary beneficiary of Xian Tianxing. Accordingly, we continue to consolidate Xian Tianxing’s results, assets and liabilities in the financial statements accompanying this Annual Report.
Legal Proceedings
The following discussion discusses all known or anticipated material legal proceedings commenced by or against us. Occasionally we may be named as a party in claims and legal proceedings arising out of the normal course of our business. These claims and legal proceedings may relate to contractual rights and obligations, employment matters, or to other matters relating to our business and operations.
Other than the matter discussed below, we are not aware of any material pending legal proceedings involving us.
Andrew Chien v. Skystar Bio-Pharmaceutical Company, et. al. (US District Court, District of Connecticut, Case No. 3:2007cv00781). Andrew Chien filed suit against us, R. Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in United States District Court for the District of Connecticut, alleging causes of action for violation of Sections 10(b) and 20(a) of the Exchange Act. In or around November 2007, the defendants filed motions to dismiss the complaint for failure to state a claim and for lack of personal jurisdiction. Mr. Chien agreed to voluntarily amend the complaint after the motions were filed, and an amended complaint was subsequently filed on or around January 4, 2008. The amended complaint dropped Weibing Lu (who is a resident of China and was never served) as a defendant. The remaining defendants contended that the amended complaint failed to correct the deficiencies of the original complaint, and filed a renewed motion to dismiss for failure to state a claim, also preserving their challenge to personal jurisdiction. The defendants denied all claims and moved the court to dismiss the amended complaint in its entirety in their motion to dismiss. The motion to dismiss also requested that the court award sanctions against Mr. Chien under Federal Rule of Civil Procedure Rule 11 (“Rule 11”) and the Private Securities Litigation Reform Act (“PSLRA”). On July 17, 2008, in a decision that is now published, the court granted defendants’ motion and subsequently dismissed the lawsuit, entering judgment on behalf of the defendants. Mr. Chien filed a notice of appeal of the court's dismissal of his lawsuit, opposed by the defendants, which remains pending. The defendants were invited by Judge Kravitz to bring a post-judgment motion for sanctions pursuant to Rule 11 and the PSLRA, which they did. On February 5, 2009, Judge Kravitz issued a ruling on defendants' motion for sanctions. He found the action filed by Mr. Chien to have been entirely frivolous, and to have constituted a “substantial” violation of Rule 11, and imposed significant monetary sanctions on both Mr. Chien and his former attorney. As part of the basis for imposing sanctions on Mr. Chien personally, the court specifically found that Mr. Chien had knowledge of facts directly contradicting the allegations of his complaint, as evident in internet postings he made on online message boards. Mr. Chien subsequently filed motions seeking to “re-open” this case, and to recuse Judge Kravitz, but both motions were denied. A notice of appeal concerning the ruling awarding sanctions against him was also filed by Mr. Chien. All appeals, including the one referenced below concerning Mr. Chien's second lawsuit, were subsequently consolidated. On May 26, 2010, the court of appeals for the Second Circuit upheld Judge Kravtiz’s ruling against Mr. Chien.
Andrew Chien v. Skystar Bio-Pharmaceutical Company, et. al. (formerly Superior Court, State of Connecticut, Case No. NNH-CV-09-5025938-S, now U.S. District Court, District of Connecticut, Case No. 3:09-CV-00149 (MRK)). Andrew Chien filed another lawsuit against us, Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in Connecticut Superior Court, alleging causes of action similar to those alleged in his federal complaint described above as well as state law causes of action. We argued in response that the new complaint was just as frivolous as Mr. Chien’s earlier federal action, which the new complaint substantially duplicated. The earlier federal action, described above, was found to be completely frivolous and dismissed in its entirety, with substantial monetary sanctions awarded against both Mr. Chien and his former attorney. A notice of removal to the U.S. District Court, District of Connecticut was filed in the state case on January 27, 2009, and the case was assigned to Judge Kravitz, the federal judge in the related federal case previously dismissed. We filed a motion to dismiss Mr. Chien's new action. In their motion to dismiss, defendants argued that all the claims asserted by Mr. Chien were frivolous, including among other grounds that they were time-barred and otherwise substantively meritless, and that sanctions against Mr. Chien under Federal Rule of Civil Procedure Rule 11 (“Rule 11”) and the Private Securities Litigation Reform Act (“PSLRA”) were again warranted. Rather than file an opposition to defendants' motion to dismiss, Mr. Chien filed a motion seeking to amend his complaint along with a proposed first amended complaint (“FAC”), which the court ultimately granted. The FAC purported to drop all eleven claims for securities fraud asserted by Mr. Chien, all of which defendants had contended were frivolous and meritless. The court ruled that these claims, abandoned in the wake of defendants' motion to dismiss, were all deemed dismissed with prejudice, and that no further briefing on defendants' pending motion to dismiss the action was required. Subsequently, the court granted the defendants' motion to dismiss, dismissing the action and all claims asserted in their entirety. In the ruling, the court held that all claims asserted against the defendants were barred and failed to state a claim on a multiplicity of grounds, including on the basis of res judicata as well as other substantive defects. The defendants filed a second motion for sanctions under Rule 11 and the PSLRA. The motion was subsequently granted by Judge Kravitz, and Mr. Chien was again ordered to pay additional monetary sanctions to us. Mr. Chien filed a notice of appeal concerning the ruling dismissing his second lawsuit. In its filings with the court of appeal, we have argued that the appeals are groundless and the earlier rulings by Judge Kravitz should be upheld, including the two awards of sanctions against Mr. Chien. The court of appeals for the Second Circuit consolidated all of Mr. Chien's appeals from both of his lawsuits. On May 26, 2010, the court of appeals for the Second Circuit upheld Judge Kravtiz’s ruling against Mr. Chien.
42
MANAGEMENT
Effective April 16, 2010, Mr. Bennet P. Tchaikovsky voluntarily resigned as our chief financial officer. Mr. Tchaikovsky’s decision to resign from his position was not the result of any material disagreement with us, our management, or any matter relating to our operations, policies or practices. Our board of directors appointed Mr. Michael Hongjie Lan in his place.
The following table identifies our current executive officers and directors as of the date of this prospectus, their respective offices and positions, and their respective dates of election or appointment:
| Name | Age | Position | Date of Appointment | |||
| Weibing Lu | 47 | Chief Executive Officer and Chairman of the Board of Directors | November 2005 | |||
| Michael H. Lan | 42 | Chief Financial Officer | April 2010 | |||
| Wei Wen | 43 | Secretary and Director | November 2005 | |||
| R. Scott Cramer | 46 | Director | October 2001 | |||
| Qiang Fan | 55 | Director | July 2008 | |||
| Chengtun Qu | 45 | Director | July 2008 | |||
| Shouguo Zhao | 47 | Director | July 2008 | |||
| Mark D. Chen | 42 | Director | May 2009 |
Biographies
Weibing Lu, Chief Executive Officer and Chairman of the Board of Directors
Mr. Weibing Lu received his Bachelor’s degree in science from Wuhan University of Mapping Science and Technology (now known as Wuhan University) in 1985. In 1986, he was a teacher of College of Xian Geology. Mr. Lu attended Xian Jiao Tong University in 1999 where he received a Master’s degree in Business Administration in 2002. Mr. Lu has vast experience in the biotechnology field and in enterprise management. In 1992, he founded the Xian Xingji Electronic Engineering Company and served as its Chairman and President until 1997. In 2002, he was awarded as the title of “Outstanding Enterpriser of Xian Feed Industry” and appointed as a director of Xian Institute of Feed Industry. In July 1997, he founded Xian Tianxing Science and Technology Development Co., Ltd. In December 2003, Xian Tianxing Science and Technology Development Co., Ltd., was reorganized and became Xian Tianxing Bio-Pharmaceutical Co., Ltd. Since December 2003, Mr. Lu has served as Chairman of the Board and General Manager of Xian Tianxing Bio-Pharmaceutical Co., Ltd.
Michael H. Lan, Chief Financial Officer
From December 2007 to January 2009, Mr. Michael Lan served as the practice director of Innowave Technology, an information technology consultancy to large and medium-sized companies, which assists in the implementation of advanced enterprise resource planning software. From February 2007 to September 2007, Mr. Lan served as the senior manager of Gilead Science, Inc., a NASDAQ listed company that discovers, develops and commercializes innovative therapeutics in areas of unmet medical need. From 2005 to 2007, Mr. Lan operated a consulting firm named HJL Consulting LLC. From 2004 to 2005, he worked as the project manager of Axion Solutions. From 1999 to 2004, Mr. Lan served as the senior consultant of Systems Management Inc. Prior to consulting, Mr. Lan supervised the Operations Accounting department of Silgan Containers, a Nasdaq listed company that is the largest metal food can manufacturer in North America. Mr. Lan received a B.A. in English Literature from Xiamen University and a Master of Accounting from the University of Southern California and passed the Certified Public Accountant test in 1996.
43
Wei Wen, Secretary and Director
Mr. Wei Wen graduated from Xian University of Science and Industry (also known as Xian University of Technology) in 1989. From 1990 to 1994, Mr. Wen was the manager of Sales Department of Xian Zhongtian Science and Technology Development Co., Ltd. From 1994 to 1997, Mr. Wen served as Vice General Manager and Manager of Sales Department of Xian Xingji Electronic Engineering Company. In 1997, Mr. Wen was appointed as the Vice General Manager of Xian Tianxing Science and Technology Development Co., Ltd. which he served until December 2003. After the reorganization of the company in December 2003, Mr. Wen was appointed and continues to serve as Vice General Manager and a Director of Xian Tianxing Bio-Pharmaceutical Co., Ltd. (including as secretary of the Board of Directors).
R. Scott Cramer, Director
Mr. R. Scott Cramer was previously the Chairman from November 2001 to November 2005, Chief Executive Officer from March 2002 to November 2005, and Chief Financial Officer from April 2003 to November 2005, of The Cyber Group Network Corporation. He is currently a member of our Board of Directors. Mr. Cramer is the founder and President of Cramer & Rauchegger, Inc., a firm specializing in retirement management, estate planning and wealth management. He has been a Registered Investment Advisor since August 2001, a Securities Selling Representative since May 1999, and a General Securities Representative (Registered Representative) since July 2002. Mr. Cramer is a graduate of Seminole State College. He received certification as a Chartered Retirement Planning Counselor from the College of Financial Planning in 2001, as a Certified Estate Planning Professional from the Abts Institute for Estate Preservation in 2001, and as a Certified Senior Advisor from the Society of Senior Advisors in 2002.
Qiang Fan, Director
Mr. Qiang Fan also serves as chairman of the compensation committee and member of the audit committee. Mr. Fan is the President and Founder of MIC Consulting Group, U.S.A., which he established in 1992 to provide operational and financial related problem solving services to privately owned companies. Since 2007, Mr. Fan is the exclusive representative of North America operation for China Venture Capital Research Institute, and the head analyst at Power Partner Institute focusing on IT trends since 2001. From 2006 to 2007, Mr. Fan was a Vice-president of Operation at Kantan Inc., a privately-held boutique technology company focused on wireless solutions for device manufacturers. From 2005 to 2006, he was a Vice-president at Third Wave Ventures, which provides corporate venturing-related advisory, consulting and management services. From 1998 to 2000, Mr. Fan was the exclusive representative in China for PowerQuest, a Utah based international software company that focused on computer data storage management, as well as for ChipCoolers, a U.S. CPU cooler manufacturer. Mr. Fan received his B.A. degree from the Business School of California State University at San Francisco.
Chengtun Qu, Director
Since March 2003, Dr. Chengtun Qu has been the Vice Dean of the College of Chemistry and Chemical Engineering at Xi’an Shi You University, where he also teaches and heads the environmental engineering department. Dr. Qu is a board member of both the Shaanxi Province Environmental Protection Association and the Shaanxi Province Chemical Engineering Association. As a principal researcher, Dr. Qu has participated in various projects at both national and provincial levels, including ones sponsored by the Chinese Ministry of Science and Technology, and is the recipient of numerous accolades from the Shaanxi provincial government in recognition of his contributions. Dr. Qu has three patents issued by the Chinese State Intellectual Property Office. He has also been extensively published in various scientific journals both in China and abroad. Dr. Qu has a B.S. degree in chemistry from Northwest University in Xi’an, a master’s degree in applied chemistry from Southwest Petroleum University in July 1993 and a doctorate degree in biochemistry from Xi’an Jiaotong University in July 2006.
Shouguo Zhao, Director
Dr. Zhao is an independent director of Shaanxi International Trust & Investment Corp., Ltd., a listed company on the Shenzhen Stock Exchange (SZSE: SZ000563), chairing its Remuneration and Assessment Committee and serving on its Strategy Committee. Dr. Zhao is also an independent non-executive director of Sungreen International Holdings Limited, a listed company on the Hong Kong Exchange (HKEX: HK8306), serving as a member of its audit committee. He is additionally an independent director of Tian Di Yuan Co., Ltd., a listed company on the Shenzhen Stock Exchange (SZSE: SH600665), chairing its Nominating Committee and serving on its Strategy Committee. From June 2005 to June 2008, Dr. Zhao was an independent director of IRICO Group Corporation, a listed company on the Shenzhen Stock Exchange (SZSE: SH600707), chairing its Remuneration and Assessment Committee and serving on its Strategy Committee. Dr. Zhao is the Vice Dean of the School of Economics and Management at Northwest University, where he also serves as a guide professor to doctorate candidates in finance and national economics. Dr. Zhao is a member of Shaanxi Provincial Decision-making Consultative Committee, a member of the Executive Committee of the Tenth Session of Shaanxi Provincial Industrial and Commercial Association, the chairman of the Negotiable Securities Research Society of Shaanxi Province, and a consultant with the Listed Companies Association of Shaanxi Province. Dr. Zhao received his doctorate degree in economics from Northwest University.
44
Mark D. Chen, Director
Mr. Chen is the chairman and chief executive officer of Pantheon China Acquisition Corp., a U. S. publicly traded acquisition company he founded in 2006 focusing on pre-IPO Chinese companies. Since 1998, Mr. Chen has been a founding general partner and has served in various positions, including managing director and currently a venture partner, with Easton Capital Investment Group and its various affiliated funds, a New York based private equity investment firm. Mr. Chen has also worked extensively in China and was a founder and senior executive of SureData Inc., a marketing and distribution company in China in 1997. Mr. Chen received a B.S. from the Shanghai Jiao Tong University in Shanghai, China, an M.S. from Pennsylvania State University and an M.B.A. from the Columbia Business School at Columbia University.
Family Relationships
There are no family relationships between or among any of the current directors, executive officers or persons nominated or charged to become directors or executive officers.
Involvement in Certain Legal Proceedings
There are no orders, judgments, or decrees of any governmental agency or administrator, or of any court of competent jurisdiction, revoking or suspending for cause any license, permit or other authority to engage in the securities business or in the sale of a particular security or temporarily or permanently restraining any of our officers or directors from engaging in or continuing any conduct, practice or employment in connection with the purchase or sale of securities, or convicting such person of any felony or misdemeanor involving a security, or any aspect of the securities business or of theft or of any felony. Nor are any of the officers or directors of any corporation or entity affiliated with us so enjoined.
Director Independence
Based upon information submitted to the Board of Directors by Mr. Fan, Dr. Qu, Dr. Zhao and Mr. Chen, the Board has determined that each of them is “independent” under the listing standards of The NASDAQ Capital Market.
Audit Committee and Audit Committee Financial Expert
Our audit committee consists of three independent directors: Mr. Fan, Dr. Zhao and Mr. Chen. Our board of directors has determined, based on information furnished by Mr. Chen and other available information, that he meets the requirements of an “audit committee financial expert” as such term is defined in the rules promulgated under the Securities Act of 1933 and the Exchange Act of 1934, as amended. On May 26, 2009, Mr. Chen was appointed to serve as chairman of the audit committee, and to serve as our audit committee financial expert.
The responsibilities of our audit committee will include:
| · | meeting with our management periodically to consider the adequacy of our internal control over financial reporting and the objectivity of our financial reporting; |
| · | appointing the independent registered public accounting firm, determining the compensation of the independent registered public accounting firm and pre-approving the engagement of the independent registered public accounting firm for audit and non-audit services; |
| · | overseeing the independent registered public accounting firm, including reviewing independence and quality control procedures and experience and qualifications of audit personnel that are providing us audit services; |
| · | meeting with the independent registered public accounting firm and reviewing the scope and significant findings of the audits performed by them, and meeting with management and internal financial personnel regarding these matters; and |
| · | reviewing our financing plans, the adequacy and sufficiency of our financial and accounting controls, practices and procedures, the activities and recommendations of the auditors and our reporting policies and practices, and reporting recommendations to our full board of directors for approval. |
Compensation Committee
Our compensation committee consists of three independent directors: Mr. Fan, Dr. Zhao and Mr. Chen. On July 14, 2008, Mr. Fan was appointed to serve as chairman of the compensation committee. Our compensation committee will oversee and, as appropriate, make recommendations to the board regarding the annual salaries and other compensation of our executive officers, our general employee compensation, and other policies, and provide assistance and recommendations with respect to our compensation policies and practices.
45
Code of Ethics
We have adopted a code of ethics that applies to our officers, directors and employees, including our chief executive officer, senior executive officers, principal accounting officer, and other senior financial officers. Our code of ethics is available on our website at www.skystarbio-pharmaceutical.com .. A copy of our code of ethics will also be provided to any person without charge, upon written request sent to us at our offices located at Rm. 10601, Jiezuo Plaza, No.4, Fenghui Road South, Gaoxin District, Xi’an, Shaanxi Province, P.R. China.
Compliance with Section 16(a) of the Exchange Act
Based solely on review of the copies of such forms furnished to us, or written representations that no reports were required, we believe that to date, our directors and executive officers have complied with Section 16(a) filing requirements applicable to them.
EXECUTIVE COMPENSATION
Summary Compensation
The following summary compensation table indicates the cash and non-cash compensation earned during the fiscal years ended December 31, 2009 and 2008 by (i) our Chief Executive Officer (principal executive officer), (ii) our former Chief Financial Officer (principal financial officer), (iii) the three most highly compensated executive officers other than our CEO and CFO who were serving as executive officers at the end of our last completed fiscal year, whose total compensation exceeded $100,000 during such fiscal year ends, and (iv) up to two additional individuals for whom disclosure would have been provided but for the fact that the individual was not serving as an executive officer at the end of our last completed fiscal year, whose total compensation exceeded $100,000 during such fiscal year ends.
Summary Compensation Table
Name and Principal Position | Year | Salary ($) | Bonus ($) | Stock Awards ( $) | Total ($) | |||||||||
Weibing Lu, CEO (1) | 2009 | 100,000 | -0- | -0- | 100,000 | |||||||||
| 2008 | 66,028 | -0- | -0- | 66,028 | ||||||||||
Bennet P. Tchaikovsky, former CFO (2) | 2009 | 75,000 | -0- | 63,280 | 138,280 | |||||||||
| 2008 | 49,395 | -0- | 39,517 | 88,912 | ||||||||||
| (1) | On May 5, 2008, we entered into an employment agreement with Mr. Lu pursuant to which he is entitled to an initial annual compensation of $100,000 as our CEO. Mr. Lu received no other form of compensation in the years shown, other than the salary set forth in this table. |
| (2) | On May 4, 2008, we entered into a loanout agreement pursuant to which we retained the services of Mr. Tchaikovsky as our CFO for one year. Pursuant to this agreement, Mr. Tchaikovsky is entitled to $75,000 of annual cash compensation and 10,435 shares valued at approximately $60,000. On May 26, 2009, we entered into an amendment to the loanout agreement for an additional year of Mr. Tchaikovsky’s services as CFO. Pursuant to this agreement, Mr. Tchaikovsky is entitled to $75,000 of annual cash compensation and 14,440 shares (taking into account the 2-for-1 forward split on November 16, 2009) valued at approximately $65,000. Mr. Tchaikovsky received no other form of compensation in the years shown, other than the salary and stock compensation set forth in this table. Mr. Tchaikovsky resigned as our CFO effective April 16, 2010. |
Outstanding Equity Awards at Fiscal Year-End
With the exception of Mr. Bennet P. Tchaikovsky, our former Chief Financial Officer, there were no unexercised options, unvested stock awards or equity incentive plan awards for any of our named executive officers outstanding as of December 31, 2009. Pursuant to the terms of his employment under the Amendment to Loanout Agreement (which terms are described below under the heading “Loanout Agreement for the Services of Bennet P. Tchaikovsky”), Mr. Tchaikovsky was granted 14,440 shares of our restricted common stock for his service period from May 5, 2009 through May 4, 2010, which shares were not issued pursuant to any equity incentive plans in effect. As of December 31, 2009, Mr. Tchaikovsky was due 7,220 shares.
Employment Agreements, Termination of Employment and Change-in-Control Arrangements
Except as described below, we currently have no employment agreements with any of our executive officers, nor any compensatory plans or arrangements resulting from the resignation, retirement or any other termination of any of our executive officers, from a change-in-control, or from a change in any executive officer’s responsibilities following a change-in-control.
46
Employment Agreement with Weibing Lu
On May 5, 2008, we entered into an Employment Agreement with Mr. Weibing Lu. Under the terms of the Employment Agreement, we agreed to the continued employment of Mr. Lu as our chief executive officer for a term of 5 years. Mr. Lu is to receive an initial annual salary of $100,000, with an annual 5% increase of the prior year’s salary thereafter during the term. Additionally, at the discretion of our board of directors’ compensation committee, Mr. Lu may be eligible for an annual bonus which amount, if any, and payment will be determined by the compensation committee. Mr. Lu is entitled to medical, disability and life insurance, as well as 4 weeks of vacation annually and reimbursement of all reasonable or authorized business expenses.
During its term, the Employment Agreement terminates upon Mr. Lu’s death, in which event we are obligated to pay Mr. Lu’s estate his base salary amount through the first anniversary of his death (or the expiration of the Employment Agreement if earlier than the anniversary date), as well as pro rata allocation of any bonus based on the days of service during the year of death, and all amounts owing to Mr. Lu at the time of termination, including for previously accrued but unpaid bonuses, expense reimbursements and accrued but unused vacation pay.
If Mr. Lu is unable to perform his obligations under the Employment Agreement for over 180 consecutive days during any consecutive 12 months period, we may terminate the Employment Agreement by written notice to Mr. Lu delivered prior to the date that he resumes his duties. Upon receipt of such written notice, Mr. Lu may request a medical examination under which if he is certified to be incapable of performing his obligations for over 2 additional months, the Employment Agreement is terminated. We are obligated to pay Mr. Lu his base salary through the second anniversary of our notice to him of his termination, less any amount Mr. Lu may receive for such period from any Company-sponsored or Company-paid for source of insurance, disability compensation or governmental program. We will also pay Mr. Lu pro rata allocation of any bonus based on the days of service during the year our notice is issued, and all amounts owing to Mr. Lu at the time of termination, including for previously accrued but unpaid bonuses, expense reimbursements and accrued but unused vacation pay.
We may also terminate the Employment Agreement for cause, upon notice if at any time Mr. Lu: (a) refuses in bad faith to carry out specific written directions of our board of directors; (b) intentionally takes fraudulent or dishonest action in his relations with us; (c) is convicted of a crime involving an act of significant moral turpitude; or (d) knowingly commits an act or omits to act in violation of our written policies, the Employment Agreement or any agreements that we may have with third parties and that is materially damaging to our business or reputation. However, termination for the cause described in (a), (b) or (d) is predicated first on Mr. Lu receiving a 5-day written notice and a reasonable opportunity to present his positions, then a subsequent written notice of the termination, with the termination to take effect 20 business days thereafter if Mr. Lu does not dispute the cause for the termination or fails to take corrective actions in good faith. Thereafter, if Mr. Lu takes corrective actions, he may be terminated for the same misconduct upon a 5-day written notice.
On the other hand, Mr. Lu may terminate the Employment Agreement upon written notice if: (w) there is a material adverse change in the nature of his title, duties or obligations; (x) we materially breach the Employment Agreement; (y) we fail to make any payment to Mr. Lu (excepting any payment which is not material and which we are contesting in good faith); or (z) there is a change of control of the Company. However, termination for cause described in (w), (x) or (y) is predicated on our receiving a written notice from Mr. Lu specifying the cause, with the termination to take effect if we fail to take corrective action within 20 business days thereafter. If Mr. Lu terminates the Employment Agreement for any one of these reasons, or if we terminate the Employment Agreement without cause, we are obligated to pay to Mr. Lu (or in the case of his/her death, his estate), his base salary and any bonus, without any offset, as well as all amounts owing to Mr. Lu at the time of termination, including for previously accrued but unpaid bonuses, expense reimbursements and accrued but unused vacation pay.
The Employment Agreement also contains restrictive covenants: (i) preventing the use and/or disclosure of confidential information during or at any time after termination; (ii) preventing competition with Skystar during his employment and for a period of 3 years after termination (including contact with or solicitation of Skystar’s customers, employees or suppliers), provided that Mr. Lu may make investments of up to 2% in the publicly-traded equity securities of any competitor of Skystar; (iii) requiring Mr. Lu to refer any business opportunities to Skystar during his employment and for a period of 1 year after termination. However, Mr. Lu shall have no further obligations with respect to competition and business opportunities if his employment is terminated without cause or if he terminates his employment for cause.
Lastly, we are obligated under the Employment Agreement to indemnify Mr. Lu for any claims made against him in his capacity as our chief executive officer and, in connection to that obligation, we are required to include him under any director and officer insurance policy that is in effect during his employment as our officer, director or consultant.
47
Employment Agreement with Michael Hongjie Lan
On April 16, 2010, we entered into an employment agreement with Mr. Lan pursuant to which we have engaged his service as our Chief Financial Officer for a period of one year for annual compensation of $100,000 as well as reimbursement for reasonable expenses incurred in connection with the performance of his duties, including travel expenses.
Compensation of Directors
During our most recent completed fiscal year, our directors were compensated as follows (for reporting purposes in this table, compensations in RMB have been converted to U.S. Dollars at the conversion rate of 6.82RMB to one U.S. Dollar):
Director Compensation Table
| Name | Fees Earned or Paid in Cash ($) | All Other Compensation ($) | Total ($) | |||||||||
| Weibing Lu (1) | -0- | -0- | -0- | |||||||||
| Wei Wen (1) | -0- | -0- | -0- | |||||||||
| R. Scott Cramer (2) | -0- | 282,374 | 282,374 | |||||||||
| Qiang Fan (3) | 30,000 | -0- | 30,000 | |||||||||
| Chengtun Qu (4) | 2,932 | -0- | 2,932 | |||||||||
| Winston Yen (5) | -0- | -0- | -0- | |||||||||
| Shouguo Zhao (6) | 7,331 | -0- | 7,331 | |||||||||
| Mark D. Chen (7) | 9,589 | 25,002 | 34,591 | |||||||||
| (1) | In connection with the share exchange transaction (described in the Description of Business above under the heading "Corporate Organization and History"), these persons became our directors on November 7, 2005. After the change in control that occurred as a result of the share exchange transaction, we do not have any compensation arrangements with our directors. |
| (2) | Mr. Cramer was an officer of ours prior to the share exchange transaction and has stayed on as a director thereafter. Mr. Cramer’s compensation for 2009 was for services provided during the year unrelated to his duties as a director, and includes 26,000 shares of our restricted common stock, none of which to be issued pursuant to any equity incentive plan in effect. As of December 31, 2009, Mr. Cramer is owed 49,834 shares for services through such date. |
| (3) | Mr. Qiang Fan was appointed to our board of directors effective July 14, 2008, and is entitled to receive annual compensation of $30,000 for his services rendered as a director, as well as chairman of the compensation and member of the audit committee. |
| (4) | Dr. Chengtun Qu was appointed to our board of directors effective July 14, 2008, and is entitled to receive annual compensation of RMB 20,000 for his services rendered as a director. |
| (5) | Mr. Winston Yen resigned from our board of directors effective May 26, 2009. |
| (6) | Dr. Shouguo Zhao was appointed to our board of directors effective July 14, 2008, and is entitled to an annual compensation of RMB 50,000 for his services rendered as a director, as well as a member of both the audit committee and the compensation committee. |
| (7) | Mr. Mark D, Chen was appointed to our board of directors effective May 26, 2009, and is entitled to an annual compensation of $14,000 for his services rendered as a director, as well as a member of both the audit committee and the compensation committee. |
There were no option awards issued to any directors and outstanding as of December 31, 2009.
Agreements with Directors
Under our agreement with Mr. Fan, he will be entitled to receive annual compensation of $30,000 for his services rendered as a member of the board, as well as the chairman of the compensation committee and member of the audit committee. Mr. Fan’s annual compensation will be paid in cash, although at the discretion of the Board, up to $8,000 of his annual compensation may be paid in the form of a number of shares of our common stock under our Stock Incentive Plan #2 (the “Plan”). During his term as a director, we agree to include Mr. Fan as an insured under an officers and directors insurance policy which we will obtain within a reasonable time (the “D&O Insurance”). In addition, we have agreed to reimburse Mr. Fan for reasonable expenses incurred in connection with the performance of duties as a director, including travel expenses.
48
Under our agreement with Dr. Qu, he will be entitled to receive annual compensation of RMB 20,000 for his services rendered as a member of the board. In addition, we have agreed to reimburse Mr. Qu for reasonable expenses incurred in connection with the performance of duties as a director, including travel expenses.
Under our agreement with Dr. Zhao, he will be entitled to receive annual compensation of RMB 50,000 for his services rendered as a member of the board, as well as a member of both the audit committee and the compensation committee. In addition, we have agreed to reimburse Mr. Zhao for reasonable expenses incurred in connection with the performance of duties as a director, including travel expenses.
Under our agreement with Mr. Chen, he will be entitled to receive annual compensation of $14,000 for his services rendered as a member of the board, as well as the chairman of the audit committee and member of the compensation committee. Additionally, Mr. Chen will have the right to receive 5,556 shares of our restricted common stock at the beginning of each term of his directorship. We also agree to include Mr. Chen as an insured under the D&O Insurance, and will reimburse him for reasonable expenses incurred in connection with the performance of duties as a director, including travel expenses.
On March 30, 2010, we entered into an agreement with Mr. Scott Cramer to memorialize the terms under which he has been acting as our United States representative (the “Representative”) since November 2006. Under the terms of this agreement, we agreed to compensate Mr. Cramer for his services as the Representative through December 31, 2009 with a stock grant of 47,334 shares of our restricted common stock, as well as a one-time cash payment of $100,000 payable by March 31, 2010. Additionally, we agreed to compensate Mr. Cramer for his services as the Representative from January 1, 2010 through March 31, 2010 in the amount of $7,500 and a stock grant of 2,500 shares of our restricted common stock.
On April 16, 2010, we entered into a services agreement with Mr. Cramer to renew his services as the Representative for an additional 1-year period beginning on April 1, 2010, for an annual fee of $30,000 payable in four quarterly installments of $7,500 at the end of each quarter. Additionally, Mr. Cramer will be entitled to receive 10,000 restricted shares of common stock under our 2010 Incentive Stock Plan (the “Plan”), and he has entered into a restricted stock award agreement in connection therewith.
Under the restricted stock award agreement, Mr. Cramer is granted 10,000 restricted shares of common stock under the Plan (the “Shares”). The Shares will vest in four installments of 2,500 shares at the end of each quarter with the first vesting date on June 30, 2010. The number and the original value of the Shares will be proportionately adjusted for any increase or decrease in the number of issued shares resulting from a recapitalization, subdivision or consolidation of shares or the payment of a stock dividend, or any other increase or decrease in the number of such shares effected without receipt of consideration by us. We will have the right to repurchase all or any portion of the Shares at a price equal to the original amount paid for the Shares by Mr. Cramer upon the termination of the services agreement or any attempted transfer of Shares in violation of the services agreement.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information regarding our common stock beneficially owned on May 27, 2010, for (i) each stockholder known to be the beneficial owner of 5% or more of our outstanding common stock, (ii) each executive officer and director, and (iii) all executive officers and directors as a group. In general, a person is deemed to be a “beneficial owner” of a security if that person has or shares the power to vote or direct the voting of such security, or the power to dispose or to direct the disposition of such security. A person is also deemed to be a beneficial owner of any securities of which the person has the right to acquire beneficial ownership within 60 days. Shares of common stock subject to options, warrants or convertible securities exercisable or convertible within 60 days of May 27, 2010 are deemed outstanding for computing the percentage of the person or entity holding such options, warrants or convertible securities but are not deemed outstanding for computing the percentage of any other person. Percentages are determined based on 7,106,705 common shares issued and outstanding as of May 27, 2010. To the best of our knowledge, subject to community and marital property laws, all persons named have sole voting and investment power with respect to such shares, except as otherwise noted.
49
Name and Address of Beneficial Owners (1) | Amount of Beneficial Ownership | Percent of Class (2) | ||||||
| Upform Group Limited (3) | 939,126 | 13.2 | % | |||||
| Weibing Lu, Director and Chief Executive Officer (3) | 939,126 | 13.2 | % | |||||
| Michael H. Lan, Chief Financial Officer (4) | -0- | * | ||||||
| Wei Wen, Director (5) | 41,544 | * | ||||||
| R. Scott Cramer, Director (6) | 206,286 | 2.9 | % | |||||
| Qiang Fan, Director (7) | -0- | 0 | % | |||||
| Chengtun Qu, Director (8) | -0- | 0 | % | |||||
| Mark D. Chen, Director (9) | 5,556 | * | ||||||
| Shouguo Zhao, Director (10) | -0- | 0 | % | |||||
| All officers and directors as a group (8 total) | 1,213,780 | 17.1 | % | |||||
* Less than 1%.
| (1) | Unless otherwise noted, the address for each of the named beneficial owners is: Rm. 10601, Jiezuo Plaza, No.4, Fenghui Road South, Gaoxin District, Xi’an, Shaanxi Province, People’s Republic of China. |
| (2) | Unless otherwise noted, the number and percentage of outstanding shares of our common stock is based upon 7,106,705 shares outstanding as of May 27, 2010. |
| (3) | Upform Group Limited’s (“Upform Group”) address is Sea Meadow House, Blackburne Highway, P.O. Box 116, Road Town, Tortola, British Virgin Islands. Weibing Lu and Xinya Zhang are directors of the Upform Group. Mr. Lu is the majority stockholder and the Chairman of the Board of Directors of Upform Group, and thus Mr. Lu indirectly owns the shares held by Upform Group, through his majority ownership of Upform Group. Thus, the number of shares reported herein as beneficially owned by Mr. Lu therefore includes the shares held by Upform Group. Similarly, because Xinya Zhang is a director of Upform Group, he might be deemed to have or share investment control over Upform Group’s portfolio. Thus, the number of shares reported herein as beneficially owned by Mr. Zhang also include the shares held by Upform Group. |
| (4) | Michael H. Lan’s address is: 19 Foxcrest, Irvine, California 92620. |
| (5) | The number of shares reported herein as beneficially owned by Wei Wen are held by Clever Mind International Limited, which address is: Sea Meadow House, Blackburne Highway, P.O. Box 116, Road Town, Tortola, British Virgin Islands. Mr. Wen is Chairman of the Board of Directors of Clever Mind an d owns approximately 2.3% of the issued and outstanding shares of Clever Mind. Because Mr. Wen is a director of Clever Mind, he might be deemed to have or share investment control over Clever Mind’s portfolio. |
| (6) | R. Scott Cramer’s address is: 1012 Lewis Dr., Winter Park, FL 32789. Includes 154,284 shares held by the Cramer Family Trust of which Mr. Cramer is the sole trustee and sole primary beneficiary, and 49,834 shares that Mr. Cramer has right to acquire beneficial ownership of within 60 days of March 24,2010. |
| (7) | Qiang Fan’s address is: 9176 West Laguna Way, Elk Grove, CA 95758. |
| (8) | Chengtun Qu’s address is: No. 18 Dian Zi 2nd Road, School of Chemistry & Chemical Engineering, Xi'an Shiyou University, Xi'an, Shaanxi Province, People’s Republic of China. |
| (9) | Mark D. Chen’s address is: 10-64 #9 Jianguomenwai Avenue, Beijing, China 100600. |
| (10) | Shouguo Zhao’s address is: No. 229 North Tai Bai Road, School of Economics and Management, Northwest University, Xi'an, Shaanxi Province, People’s Republic of China. |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Related Party Receivables and Payables
Set forth below are the related party receivables and payables between us and our officers and/or directors, and between Xian Tianxing and its stockholders, officers and/or directors, as of the date set forth on the table.
March 31, 2010 | December 31, 2009 | December 31, 2008 | ||||||||||
| (unaudited) | ||||||||||||
| Short term loans from shareholders: (1) | ||||||||||||
| Mr. Weibing Lu | $ | 36,675 | $ | 36,675 | $ | 220,050 | ||||||
| Mr. Wei Wen | 36,675 | 36,675 | 44,010 | |||||||||
| Ms. Aixia Wang | 36,675 | 36,675 | 44,010 | |||||||||
| Total | $ | 110,025 | $ | 110,025 | $ | 308,070 | ||||||
| Shares to be issued to a related party: (2) | ||||||||||||
| Mr. Mark Chen | $ | — | $ | 25,002 | — | |||||||
| Mr. Scott Cramer | 329,397 | 302,372 | $ | 95,204 | ||||||||
| Total | $ | 329,397 | $ | 327,374 | $ | 95,204 | ||||||
| Amount due to related parties: (4) | ||||||||||||
| Mr. Bennet P. Tchaikovsky – former CFO | $ | - | $ | - | $ | 13,168 | ||||||
| Mr. Scott Cramer | 101,056 | 143,556 | 224,684 | |||||||||
| Shaanxi Xingji Electronics Co. | 5,687 | — | 4,373 | |||||||||
| Officer and shareholder | — | 41,468 | — | |||||||||
| Total | $ | 106,743 | $ | 185,024 | $ | 242,225 | ||||||
50
| (1) | In 2008, Weibing Lu obtained an unsecured personal loan in the amount of $176,040 (RMB 1,500,000) from Huaxia Bank with annual interest rate of 7.47% and advanced to Xian Tianxing to facilitate operations. Xian Tianxing guaranteed the loan. The loan principal and related interest was due on December 30, 2008. On January 4, 2009, Xian Tianxing paid the full principal amount to the bank, with related interest of $15,741. |
| (2) | On May 29, 2008, Weibing Lu, Wei Wen and Aixia Wang obtained personal loans from Yanta Credit Union and advanced cash to Xian Tianxing in the total amount of $132,030 to facilitate operations. These loans, which were due on May 29, 2009 with 8.436% interest per annum and guaranteed by Xian Tianxing, were paid in full on May 29, 2009. On June 2, 2009, Mr. Lu, Mr. Wen and Ms. Wang again obtained loans from the same bank and advanced cash to Xian Tianxing in the total amount of $110,025. These loans are due on June 1, 2010, with 10.11% interest per annum and are also guaranteed by Xian Tianxing. For the year ended December 31, 2009, Xian Tianxing paid interest of $ 0 and $3,695, respectively, for these loans. |
| (3) | As of December 31, 2009 and December 31, 2008, the Company had $302,372 (representing 47,334 common shares) and $95,204 balances (representing 22,000 common shares), respectively, under agreement to issue shares to Scott Cramer, respectively, as compensation for being a representative of the Company in the United States for the periods from May 2008 to June 30, 2009, and December 31, 2008, respectively. In addition, as of December 31, 2009, the Company had $25,002 balance (representing 5,556 common shares) under agreement to issue shares to Mr. Mark D Chen as compensation at the beginning of each term of his directorship. |
| (4) | Shaanxi Xinji Electronics Co., Ltd. is owned by the wife of Weibing Lu. The amounts due to Shaanxi Xinji Electronics as of December 31, 2009 and December 31, 2008 were short-term cash transfers for business operations, non-interest bearing, unsecured, and payable upon demand. As of December 31, 2009, the Company also had $41,468 payable to officers and shareholders for advance for short-term financing purposes. As of December 31, 2009 and December 31, 2008, the Company also had amounts due to Scott Cramer for bonus and the expenses paid by them on behalf of the Company. |
Our Officers and Directors’ Relationship with Us, Our Subsidiaries and VIE
Mr. Weibing Lu, our Chairman and Chief Executive Officer, is a Director of Upform Group Limited, a British Virgin Islands company which owns approximately 13.2% of Skystar’s issued and outstanding common stock. Mr. Bennet P. Tchaikovsky, our former Chief Financial Officer, owns approximately 0.30% of Skystar’s issued and outstanding common stock. Mr. Wei Wen, who is one of our directors, is Director of Clever Mind International Limited, a British Virgin Islands company which owns approximately 0.58% of Skystar’s issued and outstanding common stock. Mr. Scott Cramer, who is also one of our directors, owns and/or controls approximately 2.9% of Skystar’s issued and outstanding common stock. Mr. Lu and Mr. Wen are both Directors of Skystar Cayman, our wholly owned subsidiary. Mr. Lan, Mr. Fan, Dr. Zhao and Dr. Qu do not own any shares of Skystar’s common stock as of the date of this prospectus.
Mr. Cramer is Director of Fortunate Time, wholly owned subsidiary of Skystar Cayman.
The management of Sida, the wholly owned subsidiary of Fortunate Time, includes Mr. Wen as General Manager
The management of Xian Tianxing, which we control through contractual arrangements between Sida and Xian Tianxing, includes Mr. Lu as Chairman and Chief Executive Officer and Mr. Wen as Vice-General Manager and Director. As of the date of this prospectus, Mr. Lu also owns approximately 41%, and Mr. Wen approximately 5%, of the issued and outstanding stock of Xian Tianxing.
Mr. Wei Shen is the General Manager of Shanghai Siqiang, wholly owned subsidiary of Xian Tianxing.
The management of Skystar California, our wholly-owned subsidiary, includes Ms. Yuping Wang.
51
Other Related Party Transactions
On January 1, 2007, we entered into a 5-year lease agreement with Mr. Weibing Lu, our chief executive officer, to lease the premises at Rm. 10601, Jiezuo Plaza, No.4, Fenghui Road South, Gaoxin District, Xi’an, Shaanxi Province, China, which belongs to Mr. Lu and which has been serving as our headquarters. The annual rent under the lease agreement is RMB 165,600 (approximately $24,000). Mr. Lu previously provided the premises rent-free, in 2005 and 2006, for the use of our administrative division.
On June 17, 2007, Shanghai Siqiang, wholly owned subsidiary of Xian Tianxing, entered into a 10-year lease agreement with Mr. Lu to lease the premises at 1715 Zhongchu Road, Building F, Unit 1001, Shanghai, China, which belongs to Mr. Lu. The annual rent under the lease agreement is RMB 144,000 (approximately $21,000).
Conflicts of interests between the duties of our officers and directors who are also management members of Xian Tianxing to our company and Xian Tianxing may arise. As our directors and/or executive officer (in the case of Mr. Lu), they have a duty of loyalty and care to us under U.S. and Cayman Islands law when there are any potential conflicts of interests between our company and Xian Tianxing. We cannot assure you, however, that when conflicts of interest arise, these individuals will act completely in our interests or that conflicts of interests will be resolved in our favor. In addition, they could violate their legal duties by diverting business opportunities from us to others. If we cannot resolve any conflicts of interest between us and them, we would have to rely on legal proceedings, which could result in the disruption of our business.
DESCRIPTION OF SECURITIES
Skystar is presently authorized under its Articles of Incorporation, as amended, to issue 40,000,000 shares of common stock, $0.001 par value per share, and 50,000,000 shares of preferred stock, $0.001 par value per share.
The following is a description of Skystar’s capital stock, including their material terms and provisions and as such terms and provisions are applied to our Articles of Incorporation, as amended, By-laws, and the applicable corporate laws of the State of Nevada.
Common Stock
At May 27, 2010, Skystar had 7,106,705 shares of common stock issued and outstanding. The holders of Skystar’s common stock are entitled to one vote for each share held of record on all matters submitted to a vote of the stockholders, except that upon giving the legally required notice, stockholders may cumulate their shares in the election of directors. We may pay dividends at such time and to the extent declared by the Board of Directors in accordance with Nevada corporate law. Our common stock has no preemptive or other subscription rights, and there are no conversion rights or redemption or sinking fund provisions with respect to such shares. All outstanding shares of common stock are fully paid and non-assessable. To the extent that additional shares of common stock may be issued in the future, the relative interests of the then existing stockholders may be diluted.
On February 15, 2006, we effected a 397-for-1 reverse stock split of the outstanding shares of our common stock. As a result, the total number of outstanding shares of Skystar’s common stock was reduced from 500,000,000 to 1,260,651 issued and outstanding, not taking into account the 1-for-10 reverse stock split effected on May 12, 2009 or the 2-for-1 forward stock split effected on November 16, 2009. Concurrently we affected a decrease in the number of shares of authorized common stock which reduced the total number of authorized common shares from 500,000,000 to 50,000,000.
On May 23, 2008, we filed a preliminary proxy statement on Schedule PRE14A with the SEC to seek stockholder approval at a special meeting of our stockholders for an amendment of our articles of incorporation to increase the number of authorized shares of our common stock from 50,000,000 shares to 200,000,000 shares (the “Amendment”). At the meeting, a majority of the stockholders present in person or by proxy constituting a quorum approved the Amendment. Accordingly, on July 11, 2008, we filed a Certificate of Amendment to our Articles of Incorporation with the Secretary of State of the State of Nevada, thereby effecting the Amendment and increasing the number of authorized shares of our common stock from 50,000,000 to 200,000,000.
On May 12, 2009, we effected a 1-for-10 reverse stock split and concurrently reduced the number of authorized shares of common stock from 200,000,000 to 20,000,000. Under Section 78.2055 of the Nevada Revised Statues (“NRS”), to decrease the numbers of issued and outstanding shares of a class or series of a corporation's capital stock requires the approval of stockholders holding a majority of the voting power of the affected class or series, or such greater proportion as may be provided in the articles of incorporation, regardless of limitations or restrictions on the voting power of the affected class or series. However, under NRS Section 78.207, a corporation may change the number of shares of a class of its authorized stock by increasing or decreasing the number of authorized shares of the class and correspondingly increasing or decreasing the number of issued and outstanding shares of the same class held by each stockholder of record by a resolution adopted by the board of directors without obtaining the approval of the stockholders. Accordingly, we effected the 1-for-10 reverse stock split without the approval of our stockholders by concurrently effecting a corresponding reduction in the number of shares of our authorized common stock pursuant to NRS Section 78.207.
52
On November 16, 2009, we effected a 2-for-1 forward stock split of the Company’s issued and outstanding shares of common stock and a proportional increase of the Company’s authorized shares of common stock from 20,000,000 to 40,000,000, pursuant to NRS Section 78.209.
All disclosures regarding common stock in this registration statement have taken into account these reverse and forward stock splits unless otherwise stated.
Preferred Stock
We are currently authorized to issue 50,000,000 shares of preferred stock. On June 25, 2009, we voided our previously issued and invalid series “A” preferred stock and have authorized the issuance of 48,000,000 shares of series “B” preferred stock. There is currently no issued or outstanding series “B” preferred stock.
The Board of Directors is authorized, subject to any limitation prescribed by the laws of the State of Nevada, but without further action by our stockholders, to provide for the issuance of preferred stock in one or more series, to establish from time to time the number of shares of each such series and any qualifications, limitations or restrictions thereof, and to increase or decrease the number of shares of any such series without any further vote or action by stockholders. The Board of Directors may authorize and issue preferred stock with voting or conversion rights that could adversely affect the voting power or other rights of the holders of common stock. In addition, the issuance of preferred stock may have the effect of delaying, deferring or preventing a change in control of our Company. The terms of the preferred stock are not defined in our Articles of Incorporation.
Warrants
As of May 27, 2010, we have outstanding warrants that, if exercised, would permit their holders to purchase approximately an additional 34,230 shares of our common stock.
We issued to the convertible debenture investors our Class A and B warrants to purchase shares of our common stock. The warrants entitled each investor to purchase a number of shares of common stock equal to 100% of the number of shares of common stock that would be issuable upon conversion of the convertible debenture purchased by such investor in the transaction. The warrants have an initial exercise price of $6.00 per share (taking into account of the 1-for-10 reverse stock split effected on May 12, 2009 and the 2-for-1 forward stock split effected on November 16, 2009) and are exercisable through the last day of the calendar month in which the third anniversary of the effective date of the resale registration statement occurs. We also issued to the placement agent and its designees warrants to purchase an aggregate of 114,100 shares of common stock with an exercise price of $5.00 per share (taking into account of the 1-for-10 reverse stock split effected on May 12, 2009 and the 2-for-1 forward stock split effected on November 16, 2009) and have an expiration date of February 28, 2012.
The warrant strike price may be adjusted downward if we issue more shares of common stock or securities convertible into common stock for capital raising activities at less than the exercise price; the exercise price is to be adjusted to the consideration received or receivable by us for each share of common stock issued or issuable. The exercise price is also subject to adjustment for other customary adjustment events such as any stock dividend, stock split, reverse stock split or other similar transaction.
The warrant holders are also entitled to exercise their warrants on a cashless basis at certain times. The cashless exercise provision will also automatically apply if, on the expiration date of the warrants, the average closing bid price for the three immediately preceding trading days is above the exercise price. If the holder elects the cashless exercise option or if the automatic provision applies, the holder will receive a lesser number of shares and we will not receive any cash proceeds from that exercise. The lesser number of shares which the holder will receive is determined by a formula that takes into account the average closing bid price of our common stock on the three trading days immediately before the warrant exercise. That average closing price is multiplied by the full number of shares for which the warrant is then being exercised. That result is reduced by the total exercise price the holder would have paid for those shares if it had not elected a cashless exercise. The number of shares actually issued under the cashless exercise option is equal to the balance amount divided by the average closing bid price referred to above.
The two classes of warrants are substantially similar, except that for Class A warrants, no stock may be issued to any holder under any provision of Class A warrants which would result in the holder being the beneficial owner of more than 4.99% of our issued and outstanding common stock, absent certain exceptions as set forth in the warrants. This provision is absent in Class B warrants.
The Amendment Agreement described in the section on convertible debentures above amended the terms of the warrants held by the Participating Purchasers by: (a) changing the exercise price from $6.00 per share to $4.75 per share; and (b) granting to the Participating Purchasers the right to exercise their warrants on a cashless basis.
53
Common Stock Purchase Options
We have agreed to sell to our underwriter, Rodman & Renshaw, LLC, for $100, an option to purchase up to a total of ______ shares of common stock (2.5% of the shares sold in the offering) at $____ per share (125% of the price of the shares sold in the offering). The shares issuable upon exercise of this option are identical to those offered by this prospectus. For a more complete description of the option, including the registration rights afforded to the holders of such option, see the section appearing elsewhere in this prospectus entitled “Underwriting — Common Stock Purchase Option”.
On June 30, 2009, the Company and Rodman & Renshaw, LLC, as representative of the underwriters, entered into an Underwriting Agreement, pursuant to which the Company agreed to grant 140,000 common stock purchase options to five designees of the underwriters (taking into account the 2-for-1 forward stock split effected on November 16, 2009). The options are exercisable from June 30, 2010 to June 30, 2014, and each option is exercisable for one share of the Company’s common stock, with exercise price at $8.11 per share (taking into account the 2-for-1 forward stock split effected on November 16, 2009).
EQUITY COMPENSATION PLAN INFORMATION
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted-average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans | |||||||||
| Equity compensation plans approved by security holders | 0 | 0 | 700,000 | (1) | ||||||||
| Equity compensation plans not approved by security holders | 0 | 0 | 538,620 | (2) (3) | ||||||||
| TOTAL | 0 | 0 | 1,238,620 | |||||||||
As of December 31, 2009, the Company had the following three equity compensation plans in effect:
| (1) | On December 8, 2009, our Board approved a stock incentive plan for officers, directors, employees and consultants entitled the “Skystar Bio-Pharmaceutical Company 2010 Stock Incentive Plan” (hereinafter the “2010 Plan”). The maximum number of shares that may be issued under the 2010 Plan is 700,000 shares of our common stock. The 2010 Plan was approved by our stockholders on December 31, 2009, and awards may be granted under this Plan until December 7, 2019. Under this Plan, the Company may issue common stock and/or options to purchase common stock to certain officers, directors and employees and consultants of the Company and its subsidiaries. The 2010 Plan is administered either by the Board or a committee appointed by the Board, which is comprised of two or more independent directors. The Board (or the committee) has full and complete authority, in its discretion, but subject to the express provisions of the 2010 Plan to approve the eligible persons nominated by the management of the Company to be granted awards of common stock “Awards”) or stock options, to determine the number of Awards or stock options to be granted to an eligible person; to determine the time or times at which or stock options shall be granted; to establish the terms and conditions upon which Awards or Stock Options may be exercised; to remove or adjust any restrictions and conditions upon Awards or Stock Options; to specify, at the time of grant, provisions relating to exercisability of Stock Options and to accelerate or otherwise modify the exercisability of any Stock Options; and to adopt such rules and regulations and to make all other determinations deemed necessary or desirable for the administration of the Plan. As of December 31, 2009, there are 700,000 shares of our common stock remaining available for future issuance under the 2010 Plan. |
| (2) | On February 22, 2006, the Company adopted a stock incentive plan for consultants entitled the “2006 Consultant Stock Plan” (hereinafter the “2006 Plan”). The maximum number of shares that may be issued under the 2006 Plan is 1,199,648 shares of our common stock. The 2006 Plan has not previously been approved by security holders and awards may be granted under this Plan until February 21, 2016. Under the 2006 Plan, the Company may issue common stock to certain consultants of the Company who are crucial to the future growth and success of the Company and its subsidiaries and affiliates. The 2006 Plan is administered by either a committee appointed by the Board, which is comprised of one or more members of the Board who is not serving on another plan committee, or the Board. The committee has full and complete authority, in its discretion, but subject to the express provisions of the Plan, to designate the persons or classes of persons eligible to receive awards of common stock “Awards”; to determine the form and amount of Awards to be granted to an eligible person or class of persons; to establish the terms and conditions upon which Awards may be exercised; to remove or adjust any restrictions and conditions upon Awards; and to adopt such rules and regulations and to make all other determinations deemed necessary or desirable for the administration of the Plan. As of December 31, 2009, there are 119,930 shares of our common stock remaining available for future issuance under the 2006 Plan. |
54
| (3) | On October 16, 2002, the Company adopted a stock incentive plan for officers, directors, employees, and consultants entitled the “Cyber Group Network Corporation Stock Incentive Plan # 2” (hereinafter the “2002 Plan”). The maximum number of shares that may be issued under the 2002 Plan is 40,000,000 shares of our common stock. The 2002 Plan has not previously been approved by security holders and awards may be granted under this Plan until October 15, 2012. Under this Plan, the Company may issue common stock and/or options to purchase common stock to certain officers, directors and employees and consultants of the Company and its subsidiaries. The 2002 Plan is administered either by the compensation committee or a committee appointed by the Board, which is comprised of a combination of two or more officers and/or members of the Board. The committee has full and complete authority, in its discretion, but subject to the express provisions of the Plan to approve the eligible persons nominated by the management of the Company to be granted awards of common stock “Awards”) or stock options, to determine the number of Awards or stock options to be granted to an eligible person; to determine the time or times at which or stock options shall be granted; to establish the terms and conditions upon which Awards or Stock Options may be exercised; to remove or adjust any restrictions and conditions upon Awards or Stock Options; to specify, at the time of grant, provisions relating to exercisability of Stock Options and to accelerate or otherwise modify the exercisability of any Stock Options; and to adopt such rules and regulations and to make all other determinations deemed necessary or desirable for the administration of the Plan. As of December 31, 2009, there are 418,690 shares of our common stock remaining available for future issuance under the 2002 Plan. |
55
UNDERWRITING
Subject to the terms and conditions of an underwriting agreement, dated _______ __, 2010, we have agreed to sell to each of the underwriters named below, and each of the underwriters, for which Rodman & Renshaw, LLC is acting as representative, have severally, and not jointly, agreed to purchase on a firm commitment basis the number of shares offered in this offering set forth opposite their respective names below, at the public offering price, less the underwriting discount set forth on the cover page of this prospectus.
Name | Number of Shares | |||
| Rodman & Renshaw, LLC | ||||
| JMP Securities LLC | ||||
| Total | ||||
Nature of Underwriting Commitment
The underwriting agreement provides that the underwriters are committed to purchase on a several but not joint basis all shares offered in this offering, other than those covered by the over-allotment option described below, if the underwriters purchase any of these securities. The underwriting agreement provides that the obligations of the underwriters to purchase the shares offered hereby are conditional and may be terminated at their discretion based on their assessment of the state of the financial markets. The obligations of the underwriters may also be terminated upon the occurrence of other events specified in the underwriting agreement. Furthermore, pursuant to the underwriting agreement, the underwriters’ obligations are subject to the authorization and the validity of the shares being accepted for listing on The NASDAQ Capital Market and to various other customary conditions, representations and warranties contained in the underwriting agreement, such as receipt by the underwriters of officers’ certificates and legal opinions of our counsel.
Pricing of Securities
Our underwriter has advised us that it proposes to offer the shares directly to the public at the public offering price set forth on the cover page of this prospectus, and to certain dealers that are members of the Financial Industry Regulatory Authority (FINRA), at such price less a concession not in excess of $_____ per share. The underwriter may allow, and the selected dealers may reallow, a concession not in excess of $_____ per share to certain brokers and dealers. After this offering, the offering price and concessions and discounts to brokers and dealers and other selling terms may from time to time be changed by the underwriters. These prices should not be considered an indication of the actual value of our shares and are subject to change as a result of market conditions and other factors. No variation in those terms will change the amount of proceeds to be received by us as set forth on the cover page of this prospectus.
Our common stock is traded on The NASDAQ Capital Market under the symbol “SKBI”. On May 27, 2010, the closing market price of our common stock as quoted on The NASDAQ Capital Market was $8.56. The public offering price for the shares was determined by negotiation between us and the underwriter. Among the factors considered in determining the public offering price of the shares, in addition to prevailing market conditions, were our historical performance, estimates of our business potential and earnings prospects, an assessment of our management, the public demand for our securities in this offering, and the consideration of the above factors in relation to market valuation of companies in related businesses.
We cannot be sure that the public offering price will correspond to the price at which our shares will trade in the public market following this offering or that an active trading market for our shares will develop and continue after this offering.
Commissions and Discounts
The following table summarizes the compensation to be paid to our underwriter by us and the proceeds, before expenses, payable to us, assuming a $____ offering price. The information assumes either no exercise or full exercise by the underwriters of the over-allotment option.
Total | ||||||||||||
Per Share | Without Over-Allotment | With Over-Allotment | ||||||||||
| Public offering price | $ | $ | $ | |||||||||
Underwriting discount (1) | $ | $ | $ | |||||||||
Proceeds, before expenses, to us (2) | $ | $ | $ | |||||||||
| (1) | Underwriting discount is $___ per share. |
56
| (2) | We estimate that the total expenses of this offering, excluding the underwriter’s discount, are approximately $ ___. |
Over-allotment Option
We have granted the underwriter an option, exercisable for 45 days after the closing date of this offering, to purchase up to 15% of the shares of common stock sold in the offering (_______ additional shares) solely to cover over-allotments, if any, at the same price as the initial shares offered.
Lock-ups
All of our officers and directors have agreed that, for a period of three months from the closing date of this offering, they will not sell, contract to sell, grant any option for the sale or otherwise dispose of any of our equity securities, or any securities convertible into or exercisable or exchangeable for our equity securities, without the consent of the representative except for exercise or conversion of currently outstanding warrants, options and convertible debentures, as applicable; and exercise of options under an acceptable stock incentive plan. The underwriter representative may consent to an early release from the lock-up periods if, in its opinion, the market for the common stock would not be adversely impacted by sales and in cases of a financial emergency of an officer, director or other stockholder. We are unaware of any officer or director who intends to ask for consent to dispose of any of our equity securities during the relevant lock-up periods.
Common Stock Purchase Option
We have agreed to sell to our underwriter, Rodman & Renshaw, LLC, for $100, an option to purchase up to a total of ____ shares of common stock (2.5% of the shares sold in this offering). The shares issuable upon exercise of this option are identical to those offered by this prospectus. This option is exercisable at $____ per share (125% of the price of the shares sold in the offering) at any time and from time to time, in whole or part, during the two-year period commencing one year from the closing of this offering. The option and the _____ shares of common stock underlying the option have been deemed compensation by the FINRA and are therefore subject to a 180-day lock-up pursuant to Rule 2710(g)(1) of the FINRA. Rodman & Renshaw, LLC (or permitted assignees under the Rule) will not sell, transfer, assign, pledge, or hypothecate this option or the securities underlying this option, nor will it engage in any hedging, short sale, derivative, put, or call transaction that would result in the effective economic disposition of this option or the underlying securities for a period of 180 days from the date of this prospectus. Additionally, the option may not be sold transferred, assigned, pledged or hypothecated for a one-year period (including the foregoing 180 day period) following the effective date of the registration statement except to any underwriter and selected dealer participating in the offering and their bona fide officers or partners. Although the option and its underlying securities have been registered on the registration statement of which this prospectus forms a part, the option provides for registration rights (including one-time demand registration right and unlimited piggyback rights) and customary anti-dilution provisions (for stock dividends and splits and recapitalizations). The number of shares underlying the option shall be reduced, if necessary, to comply with FINRA rules or regulations.
Notwithstanding the foregoing, if (a) during the last 17 days of the lock-up period we release earnings results or material news or a material event relating to us occurs, or (b) prior to the expiration of the lock-up period, we announce that we will release earnings results during the 16-day period following the last day of the lock-up period, the above restrictions shall continue to apply until the expiration of the 18-day period beginning on the date of the release of the earnings results or the occurrence of the material news or material event.
This option will be valued based on the underlying shares obtainable and valuation factors appropriate at the time it is issued. We currently estimate that value to be approximately $______, based on the number of shares subject to this option, an offering price of the shares of $____, the resulting exercise prices related to the option on the shares, the three year term of the option, a risk-free interest rate of ___% currently commensurate with that term, an expected dividend yield of 0% and estimated volatility of 100%, based on a review of our historical volatility. The initial value of this option will be charged to additional paid-in capital as part of the offering costs incurred, and the option will be accounted for as a derivative instrument liability because it is denominated in a currency other than our functional currency.
Other Terms
In connection with this offering, the underwriter or certain of the securities dealers may distribute prospectuses electronically. No forms of prospectus other than printed prospectuses and electronically distributed prospectuses that are printable in Adobe PDF format will be used in connection with this offering.
The underwriter has informed us that it does not expect to confirm sales of shares offered by this prospectus to accounts over which it exercises discretionary authority without obtaining the specific approval of the account holder.
57
We have also granted Rodman & Renshaw, LLC a right of first refusal to conduct future offerings for us during the 9 months following the closing date of this offering.
Stabilization
Until the distribution of the shares of common stock offered by this prospectus is completed, rules of the SEC may limit the ability of the underwriters to bid for and to purchase our securities. As an exception to these rules, the underwriters may engage in transactions effected in accordance with Regulation M under the Securities Exchange Act of 1934 that are intended to stabilize, maintain or otherwise affect the price of our common stock. The underwriters may engage in over-allotment sales, syndicate covering transactions, stabilizing transactions and penalty bids in accordance with Regulation M.
| · | Stabilizing transactions permit bids or purchases for the purpose of pegging, fixing or maintaining the price of the common stock, so long as stabilizing bids do not exceed a specified maximum. |
| · | Over-allotment involves sales by the underwriters of shares in excess of the number of shares the underwriters are obligated to purchase, which creates a short position. The short position may be either a covered short position or a naked short position. In a covered short position, the number of shares over-allotted by the underwriters is not greater than the number of shares that they may purchase in the over-allotment option. In a naked short position, the number of shares involved is greater than the number of shares in the over-allotment option. The underwriters may close out any covered short position by either exercising their over-allotment option or purchasing shares in the open market. |
| · | Covering transactions involve the purchase of securities in the open market after the distribution has been completed in order to cover short positions. In determining the source of securities to close out the short position, the underwriters will consider, among other things, the price of securities available for purchase in the open market as compared to the price at which they may purchase securities through the over-allotment option. If the underwriters sell more shares of common stock than could be covered by the over-allotment option, creating a naked short position, the position can only be closed out by buying securities in the open market. A naked short position is more likely to be created if the underwriters are concerned that there could be downward pressure on the price of the securities in the open market after pricing that could adversely affect investors who purchase in this offering. |
| · | Penalty bids permit the underwriters to reclaim a selling concession from a selected dealer when the shares of common stock originally sold by the selected dealer are purchased in a stabilizing or syndicate covering transaction. |
These stabilizing transactions, covering transactions and penalty bids may have the effect of raising or maintaining the market price of our common stock or preventing or retarding a decline in the market price of our common stock. As a result, the price of our common stock may be higher than the price that might otherwise exist in the open market.
Neither we nor the underwriters make any representation or prediction as to the effect that the transactions described above may have on the prices of our securities. These transactions may occur on The NASDAQ Capital Market or on any other trading market. If any of these transactions are commenced, they may be discontinued without notice at any time.
Foreign Regulatory Restrictions on Purchase of the Common Stock
We have not taken any action to permit a public offering of shares of our common stock outside the United States or to permit the possession or distribution of this prospectus outside the United States. Persons outside the United States who come into possession of this prospectus must inform themselves about and observe any restrictions relating to this offering of common shares and the distribution of the prospectus outside the United States.
In addition to the public offering of the shares in the United States, the underwriters may, subject to the applicable foreign laws, also offer the common shares to certain institutions or accredited persons in the following countries:
Italy. This offering of shares of our common stock has not been cleared by Consob, the Italian Stock Exchange’s regulatory agency of public companies, pursuant to Italian securities legislation and, accordingly, no common shares may be offered, sold or delivered, nor may copies of this prospectus or of any other document relating to our common stock be distributed in Italy, except (1) to professional investors ( operatori qualificati ); or (2) in circumstances which are exempted from the rules on solicitation of investments pursuant to Decree No. 58 and Article 33, first paragraph, of Consob Regulation No. 11971 of May 14, 1999, as amended. Any offer, sale or delivery of our common shares or distribution of copies of this prospectus or any other document relating to our common stock in Italy under (1) or (2) above must be (i) made by an investment firm, bank or financial intermediary permitted to conduct such activities in Italy in accordance with the Decree No. 58 and Legislative Decree No. 385 of September 1, 1993, or the Banking Act; and (ii) in compliance with Article 129 of the Banking Act and the implementing guidelines of the Bank of Italy, as amended from time to time, pursuant to which the issue or the offer of securities in Italy may need to be preceded and followed by an appropriate notice to be filed with the Bank of Italy depending, inter alia , on the aggregate value of the securities issued or offered in Italy and their characteristics; and (iii) in compliance with any other applicable laws and regulations.
58
Germany. The offering of shares of our common stock is not a public offering in the Federal Republic of Germany. The common shares may only be acquired in accordance with the provisions of the Securities Sales Prospectus Act (Wertpapier-Verkaudfspropsektgestz), as amended, and any other applicable German law. No application has been made under German law to publicly market our common shares in or out of the Federal Republic of Germany. Our common shares are not registered or authorized for distribution under the Securities Sales Prospectus Act and accordingly may not be, and are not being, offered or advertised publicly or by public promotion. Therefore, this prospectus is strictly for private use and the offering is only being made to recipients to whom the document is personally addressed and does not constitute an offer or advertisement to the public. Our common shares will only be available to persons who, by profession, trade or business, buy or sell securities for their own or a third party’s account.
France. The shares of our common stock offered by this prospectus may not be offered or sold, directly or indirectly, to the public in France. This prospectus has not been or will not be submitted to the clearance procedure of the Autorité des Marchés Financiers, or the AMF, and may not be released or distributed to the public in France. Investors in France may only purchase the common shares offered by this prospectus for their own account and in accordance with articles L. 411-1, L. 441-2 and L. 412-1 of the Code Monétaire et Financier and decree no. 98-880 dated October 1, 1998, provided they are “qualified investors” within the meaning of said decree. Each French investor must represent in writing that it is a qualified investor within the meaning of the aforesaid decree. Any resale, directly or indirectly, to the public of the common shares offered by this prospectus may be effected only in compliance with the above mentioned regulations. “Les actions offertes par ce document d’information ne peuvent pas être, directement ou indirectement, offertes ou vendues au public en France. Ce document d’information n’a pas été ou ne sera pas soumis au visa de l’Autorité des Marchés Financiers et ne peut être diffusé ou distribué au public en France. Les investisseurs en France ne peuvent acheter les actions offertes par ce document d’information que pour leur compte propre et conformément aux articles L. 411-1, L. 441-2 et L. 412-1 du Code Monétaire et Financier et du décret no. 98-880 du 1 octobre 1998, sous réserve qu’ils soient des investisseurs qualifiés au sens du décret susvisé. Chaque investisseur doit déclarer par écrit qu’il est un investisseur qualifié au sens du décret susvisé. Toute revente, directe ou indirecte, des actions offertes par ce document d’information au public ne peut être effectuée que conformément à la réglementation susmentionnée.”
Greece. The present prospectus has been submitted for approval by the United States Securities and Exchange Commission, or the US SEC, and not the Greek Capital Market Committee. All information contained in the prospectus is true and accurate. The offering of the shares of our common stock does not constitute an initial public offer in Greece according to CL. 2190/1920 and L. 3401/2005 as amended and in force. This prospectus is strictly for the use of the entity to which it has been addressed to by us and not to be circulated in Greece or any other jurisdiction.
This information and documentation is true and accurate and in conformity with the information contained in the prospectus for the offer of shares of our common stock, which is being reviewed for approval only by the United States Securities and Exchange Commission, and does not constitute provision of the investment service of investment advice according to L. 3606/2007. Any recipient of this material has stated to be a qualified and experienced investor and will evaluate the contents and decide on his/her own discretion whether to participate or not in this offering.
Switzerland. This prospectus may only be used by those persons to whom it has been directly handed out by the offeror or its designated distributors in connection with the offer described therein. The shares of common stock are only offered to those persons and/or entities directly solicited by the offeror or its designated distributors, and are not offered to the public in Switzerland. This prospectus constitutes neither a public offer in Switzerland nor an issue prospectus in accordance with the respective Swiss legislation, in particular but not limited to Article 652A Swiss Code Obligations. Accordingly, this prospectus may not be used in connection with any other offer, whether private or public and shall in particular not be distributed to the public in Switzerland.
United Kingdom. In the United Kingdom, the shares of common stock offered by this prospectus are directed to and will only be available for purchase to a person who is an exempt person as referred to at paragraph (c) below and who warrants, represents and agrees that: (a) it has not offered or sold, will not offer or sell, any common shares offered by this prospectus to any person in the United Kingdom except in circumstances which do not constitute an offer to the public in the United Kingdom for the purposes of the section 85 of the Financial Services and Markets Act 2000 (as amended), or the FSMA and (b) it has complied and will comply with all applicable provisions of FSMA and the regulations made thereunder in respect of anything done by it in relation to the common shares offered by this prospectus in, from or otherwise involving the United Kingdom; and (c) it is a person who falls within the exemptions to Section 21 of the FSMA as set out in The Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, or the Order, being either an investment professional as described under Article 19 or any body corporate (which itself has or a group undertaking has a called up share capital or net assets of not less than £500,000 (if more than 20 members) or otherwise £5 million) or an unincorporated association or partnership (with net assets of not less than £5 million) or is a trustee of a high value trust or any person acting in the capacity of director, officer or employee of such entities as defined under Article 49(2)(a) to (d) of the Order, or a person to whom the invitation or inducement may otherwise lawfully be communicated or cause to be communicated. The investment activity to which this document relates will only be available to and engaged in only with exempt persons referred to above. Persons who are not investment professionals and do not have professional experience in matters relating to investments or are not an exempt person as described above, should not review nor rely or act upon this document and should return this document immediately. It should be noted that this document is not a prospectus in the United Kingdom as defined in the Prospectus Regulations 2005 and has not been approved by the Financial Services Authority or any competent authority in the United Kingdom.
59
Sweden. Neither this prospectus nor the shares of common stock offered hereunder have been registered with or approved by the Swedish Financial Supervisory Authority under the Swedish Financial Instruments Trading Act (1991:980) (as amended), nor will such registration or approval be sought. Accordingly, this prospectus may not be made available nor may the common shares offered hereunder be marketed or offered for sale in Sweden other than in circumstances which are deemed not to be an offer to the public in Sweden under the Financial Instruments Trading Act. This prospectus may not be distributed to the public in Sweden and a Swedish recipient of the prospectus may not in any way forward the prospectus to the public in Sweden.
Norway. This prospectus has not been produced in accordance with the prospectus requirements laid down in the Norwegian Securities Trading Act 1997, as amended. This prospectus has not been approved or disapproved by, or registered with, either the Oslo Stock Exchange or the Norwegian Registry of Business Enterprises. This prospectus may not, either directly or indirectly be distributed to Norwegian potential investors.
Denmark. This prospectus has not been prepared in the context of a public offering of securities in Denmark within the meaning of the Danish Securities Trading Act No. 171 of 17 March 2005, as amended from time to time, or any Executive Orders issued on the basis thereof and has not been and will not be filed with or approved by the Danish Financial Supervisory Authority or any other public authority in Denmark. The offering of the shares of common stock will only be made to persons pursuant to one or more of the exemptions set out in Executive Order No. 306 of 28 April 2005 on Prospectuses for Securities Admitted for Listing or Trade on a Regulated Market and on the First Public Offer of Securities exceeding EUR 2,500,000 or Executive Order No. 307 of 28 April 2005 on Prospectuses for the First Public Offer of Certain Securities between EUR 100,000 and EUR 2,500,000, as applicable.
The Netherlands. Underwriters may not offer, distribute, sell, transfer or deliver any of our securities, directly or indirectly, in The Netherlands, as a part of their initial distribution or at any time thereafter, to any person other than our employees or employees of our subsidiaries, individuals who or legal entities which trade or invest in securities in the conduct of their profession or business within the meaning of article 2 of the Exemption Regulation issued under the Securities Transactions Supervision Act 1995 (Vrijstellingsregeling Wet toezich teffectenverkeer1995), which includes banks, brokers, pension funds, insurance companies, securities institutions, investment institutions, and other institutional investors, including, among others, treasuries of large enterprises who or which regularly trade or invest in securities in a professional capacity.
Cyprus. Each of the book running managers has represented, warranted and agreed that: (i) it will not be providing from or within Cyprus any “Investment Services”, “Investment Activities” and “Non-Core Services” (as such terms are defined in the Investment Firms Law 144(I) of 2007, or the IFL, in relation to the shares of common stock, or will be otherwise providing Investment Services, Investment Activities and Non-Core Services to residents or persons domiciled in Cyprus. Each book running manager has represented, warranted and agreed that it will not be concluding in Cyprus any transaction relating to such Investment Services, Investment Activities and Non-Core Services in contravention of the IFL and/or applicable regulations adopted pursuant thereto or in relation thereto; and (ii)it has not and will not offer any of the shares of common stock other than in compliance with the provisions of the Public Offer and Prospectus Law, Law 114(I)/2005.
Israel. The shares of common stock offered by this prospectus have not been approved or disapproved by the Israeli Securities Authority (ISA). The common shares may not be offered or sold, directly or indirectly, to the public in Israel. The ISA has not issued permits, approvals or licenses in connection with the offering of the common shares or publishing the prospectus; nor has it authenticated the details included herein, confirmed their reliability or completeness, or rendered an opinion as to the quality of the common shares being offered. Any resale, directly or indirectly, to the public of the common shares offered by this prospectus is subject to restrictions on transferability and must be effected only in compliance with the Israeli securities laws and regulations.
Oman. For the attention of the residents of Oman:
The information contained in this prospectus neither constitutes a public offer of securities in the Sultanate of Oman as contemplated by the Commercial Companies Law of Oman (Sultani Decree 4/74) or the Capital Market Law of Oman (Sultani Decree 80/98), nor does it constitute an offer to sell, or the solicitation of any offer to buy non-Omani securities in Oman as contemplated by Article 6 of the Executive Regulations to the Capital Market Law of Oman (issued vide Ministerial Decision No 4/2001), and nor does it constitute a distribution of non-Omani securities in Oman as contemplated under the Rules for Distribution of Non-Omani Securities in Oman issued by the Capital Market Authority of Oman, or the CMA. Additionally, this prospectus is not intended to lead to the conclusion of any contract of whatsoever nature within the territory of Oman.
60
This prospectus has been sent at the request of the investor in Oman, and by receiving this prospectus, the person or entity to whom it has been issued and sent understands, acknowledges and agrees that this prospectus has not been approved by the CMA or any other regulatory body or authority in Oman, nor has any authorization, license or approval been received from the CMA or any other regulatory authority in Oman, to market, offer, sell, or distribute the shares within Oman.
No marketing, offering, selling or distribution of any financial or investment products or services has been or will be made from within Oman and no subscription to any securities, products or financial services may or will be consummated within Oman. The underwriters are neither companies licensed by the CMA to provide investment advisory, brokerage, or portfolio management services in Oman, nor banks licensed by the Central Bank of Oman to provide investment banking services in Oman. The underwriters do not advise persons or entities resident or based in Oman as to the appropriateness of investing in or purchasing or selling securities or other financial products.
Nothing contained in this prospectus is intended to constitute Omani investment, legal, tax, accounting or other professional advice. This prospectus is for your information only, and nothing herein is intended to endorse or recommend a particular course of action. You should consult with an appropriate professional for specific advice on the basis of your situation.
United Arab Emirates. his document has not been reviewed, approved or licensed by the Central Bank of the United Arab Emirates, or the UAE, Emirates Securities and Commodities Authority or any other relevant licensing authority in the UAE including any licensing authority incorporated under the laws and regulations of any of the free zones established and operating in the territory of the UAE, in particular the Dubai International Financial Services Authority, or the DFSA, a regulatory authority of the Dubai International Financial Centre, or the DIFC. The sale of the shares does not constitute a public offer of securities in the UAE, DIFC and/or any other free zone in accordance with the Commercial Companies Law, Federal Law No. 8 of 1984 (as amended), DFSA Offered Securities Rules and the Dubai International Financial Exchange Listing Rules, accordingly, or otherwise.
The shares may not be offered to the public in the UAE and/or any of the free zones including, in particular, the DIFC. The shares may be offered and this document may be issued, only to a limited number of investors in the UAE or any of its free zones (including, in particular, the DIFC) who qualify as sophisticated investors under the relevant laws and regulations of the UAE or the free zone concerned. Management of the Company and the representatives represent and warrant that the shares will not be offered, sold, transferred or delivered to the public in the UAE or any of its free zones including, in particular, the DIFC.
People’s Republic of China. This prospectus may not be circulated or distributed in the People’s Republic of China, or PRC, and our common stock may not be offered or sold to any person for re-offering or resale, directly or indirectly, to any resident of the PRC except pursuant to applicable laws and regulations of the PRC. For the purpose of this paragraph, PRC does not include Taiwan and the special administrative regions of Hong Kong and Macau.
Botswana. The company hereby represents and warrants that it has not offered for sale or sold, and will not offer or sell, directly or indirectly the shares of common stock to the public in the Republic of Botswana, and confirms that the offering will not be subject to any registration requirements as a prospectus pursuant to the requirements and/or provisions of the Companies Act, 2003 or the Listing Requirements of the Botswana Stock Exchange.
Hong Kong. The shares of common stock have not been offered or sold and will not be offered or sold in Hong Kong, by means of any document, other than (a) to “professional investors” as defined in the Securities and Futures Ordinance (Cap. 571) of Hong Kong and any rules made under that Ordinance; or (b) in other circumstances which do not result in the document being a “prospectus” as defined in the Companies Ordinance (Cap. 32) of Hong Kong or which do not constitute an offer to the public within the meaning of that Ordinance. No advertisement, invitation or document, whether in Hong Kong or elsewhere, which is directed at, or the contents of which are likely to be accessed or read by, the public of Hong Kong (except if permitted to do so under the securities laws of Hong Kong) has been issued or will be issued in Hong Kong or elsewhere other than with respect to the common units or shares of common stock which are or are intended to be disposed of only to persons outside Hong Kong or only to “professional investors” within the meaning of the Securities and Futures Ordinance (Cap. 571) of Hong Kong and any rules made under that Ordinance.
The contents of this document have not been reviewed by any regulatory authority in Hong Kong. You are advised to exercise caution in relation to the offer. If you are in any doubt about any of the contents of this document, you should obtain independent professional advice.
61
Singapore. This prospectus has not been registered as a prospectus with the Monetary Authority of Singapore. Accordingly, this prospectus and any other document or material in connection with the offer or sale, or invitation for subscription or purchase, of the shares may not be circulated or distributed, nor may the shares be offered or sold, or be made the subject of an invitation for subscription or purchase, whether directly or indirectly, to persons in Singapore other than (i) to an institutional investor under Section 274 of the Securities and Futures Act, Chapter 289 of Singapore, or the SFA, (ii) to a relevant person, or any person pursuant to Section 275(1A), and in accordance with the conditions, specified in Section 275 of the SFA or (iii) otherwise pursuant to, and in accordance with the conditions of, any other applicable provision of the SFA.
Where the shares are subscribed or purchased under Section 275 by a relevant person which is: (a) a corporation (which is not an accredited investor) the sole business of which is to hold investments and the entire share capital of which is owned by one or more individuals, each of whom is an accredited investor; or (b) a trust (where the trustee is not an accredited investor) whose sole purpose is to hold investments and each beneficiary is an accredited investor, shares, debentures and units of shares and debentures of that corporation or the beneficiaries’ rights and interest in that trust shall not be transferable for 6 months after that corporation or that trust has acquired the shares under Section 275 except: (1) to an institutional investor under Section 274 of the SFA or to a relevant person, or any person pursuant to Section 275(1A), and in accordance with the conditions, specified in Section 275 of the SFA; (2) where no consideration is given for the transfer; or (3) by operation of law.
Indemnification
The underwriting agreement provides for indemnification between us and the underwriter against specified liabilities, including liabilities under the Securities Act, and for contribution by us and the underwriters to payments that may be required to be made with respect to those liabilities. We have been advised that, in the opinion of the SEC, indemnification for liabilities under the Securities Act is against public policy as expressed in the Securities Act, and is therefore, unenforceable.
LEGAL MATTERS
The validity of the shares sold by us under this prospectus will be passed upon for us by Richardson & Patel LLP in Los Angeles, California. Sichenzia Ross Friedman Ference LLP, New York, New York, has acted as counsel for the underwriter.
EXPERTS
The financial statements as of and for the years ended December 31, 2009 and 2008 included in this prospectus have been audited by Frazer Frost, LLP, independent certified public accountants to the extent and for the periods set forth in their report appearing elsewhere herein and are included in reliance upon such report given upon the authority of that firm as experts in auditing and accounting.
DISCLOSURE OF COMMISSION POSITION OF
INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Pursuant to Article 7 of our Articles of Incorporation and Nevada’s Revised Business Statutes, we adopted Bylaws with the following indemnification provisions for our directors and officers:
“Section 8.1. Indemnification. No officer or director shall be personally liable for any obligations arising out of any acts or conduct of said officer or director performed for or on behalf of the Corporation. The Corporation shall and does hereby indemnify and hold harmless each person and his heirs and administrators who shall serve at any time hereafter as a director or officer of the Corporation from and against any and all claims, judgments and liabilities to which such persons shall become subject by reason of any action alleged to have been heretofore or hereafter taken or omitted to have been taken by him as such director or officer, and shall reimburse each such person for all legal and other expenses reasonably incurred by him in connection with any such claim of liability; including power to defend such person from all suits as provided for under the provisions of the Nevada Corporation Laws; provided, however that no such person shall be indemnified against, or be reimbursed for, any expense incurred in connection with any claim or liability arising out of his own gross negligence or willful misconduct. The rights accruing to any person under the foregoing provisions of this section shall not exclude any other right to which he may lawfully be entitled, nor shall anything herein contained restrict the right of the Corporation to indemnify or reimburse such person in any proper case, even though not specifically herein provided for. The Corporation, its directors, officers, employees and agents shall be fully protected in taking any action or making any payment or in refusing so to do in reliance upon the advice of counsel.
Section 8.2. Other Indemnification. The indemnification herein provided shall not be deemed exclusive of any other rights to which those seeking indemnification may be entitled under any bylaw, agreement, vote of stockholders or disinterested directors, or otherwise, both as to action in his official capacity and as to action in another capacity while holding such office, and shall continue as to a person who has ceased to be a director, officer or employee and shall inure to the benefit of the heirs, executors and administrators of such a person.
62
Section 8.3. Insurance. The Corporation may purchase and maintain insurance on behalf of any person who is or was a director, officer or employee of the Corporation, or is or was serving at the request of the Corporation in such capacity for another corporation, partnership, joint venture, trust or other enterprise, against any liability asserted against him and incurred by him in any capacity, or arising out of his status as such, whether or not the Corporation would have the power to indemnify him against liability under the provisions of this Article VIII or the laws of the State of Nevada.
Section 8.4. Settlement by Corporation. The right of any person to be indemnified shall be subject always to the right of the Corporation by its Board of Directors, in lieu of such indemnity, to settle any such claim, action, suit or proceeding at the expense of the Corporation by the payment of the amount of such settlement and the costs and expenses incurred in connection therewith.”
These indemnification provisions may be sufficiently broad to permit indemnification of the registrant's executive officers and directors for liabilities (including reimbursement of expenses incurred) arising under the Securities Act.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. No pending material litigation or proceeding involving our directors, executive officers, employees or other agents as to which indemnification is being sought exists, and we are not aware of any pending or threatened material litigation that may result in claims for indemnification by any of our directors or executive officers.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act with respect to the common stock being offered pursuant to this prospectus. This prospectus does not contain all of the information set forth in the registration statement and the exhibits and schedules filed as part of the registration statement. For further information with respect to us and our common stock, we refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. Statements contained in this prospectus concerning the contents of any contract or any other document are not necessarily complete. If a contract or document has been filed as an exhibit to the registration statement, we refer you to the copy of the contract or document that has been filed. Each statement in this prospectus relating to a contract or document filed as an exhibit is qualified in all respects by the filed exhibit. You may obtain copies of this information by mail from the Public Reference Section of the SEC, 100 F Street, N.E., Room 1580, Washington, D.C. 20549, at prescribed rates. You may obtain information on the operation of the public reference rooms by calling the SEC at 1-800-SEC-0330. The SEC also maintains an Internet website that contains reports, proxy statements and other information about issuers, like us, that file electronically with the SEC. The address of that website is www.sec.gov ..
63
INDEX TO FINANCIAL STATEMENTS
| Page | ||
| Report of Independent Registered Public Accounting Firm | F-2 | |
| Consolidated Balance Sheets as of December 31, 2009 and 2008 | F-3 | |
| Consolidated Statements of Operations and other Comprehensive Income (Loss) for the Years Ended December 31, 2009 and 2008 | F-4 | |
| Consolidated Statements of Shareholders’ Equity | F-5 | |
| Consolidated Statements of Cash Flows for the Years Ended December 31, 2009 and 2008 | F-6 | |
| Notes to Consolidated Financial Statements as of December 31, 2009 | F-7 | |
| Consolidated Balance Sheets as of March 31, 2010 and 2009 (Unaudited) | F-30 | |
| Consolidated Statements of Operations and Other Comprehensive Income for the Three Months Ended March 31, 2010 and 2009 (Unaudited) | F-31 | |
| Consolidated Statements of Shareholders’ Equity (Unaudited) | F-32 | |
| Consolidated Statements of Cash Flows for the Three Months Ended March 31, 2010 and 2009 (Unaudited) | F-33 | |
| Notes to Consolidated Financial Statements as of March 31, 2010 (Unaudited) | F-34 |
F-1

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Shareholder of
Skystar Bio-Pharmaceutical Company and Subsidiaries
We have audited the accompanying consolidated balance sheets of Skystar Bio-Pharmaceutical Company and Subsidiaries (the “Company”) as of December 31, 2009 and 2008, and the related consolidated statements of income and other comprehensive income, changes in equity, and cash flows for each of the years in the two-year period ended December 31, 2009. Our audits also included the financial statement schedules for the years ended December 31, 2009 and 2008. The Company’s management is responsible for these consolidated financial statements. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement. The company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Skystar Bio-Pharmaceutical Company and Subsidiaries as of December 31, 2009 and 2008, and the results of its operations and its cash flows for each of the years in the two-year period ended December 31, 2009 in conformity with accounting principles generally accepted in the United States of America.
/s/ Frazer Frost, LLP (Successor Entity of Moore Stephens Wurth Frazer and Torbet, LLP, see Form 8-K filed on January 7, 2010)
Brea, California
March 31, 2010

F-2
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
As of December 31, 2009 and 2008
| December 31, | December 31, | |||||||
| 2009 | 2008 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash | $ | 11,699,398 | $ | 576,409 | ||||
| Restricted cash | - | 80,885 | ||||||
| Short-term investments | - | 352,080 | ||||||
| Accounts receivable, net of allowance for doubtful accounts of $327,857 and $327,857 as of December 31, 2009 and 2008, respectively | 4,383,187 | 2,424,102 | ||||||
| Inventories | 4,074,645 | 3,086,060 | ||||||
| Deposits and prepaid expenses | 11,900,314 | 4,878,851 | ||||||
| Loans receivable | - | 295,087 | ||||||
| Other receivables | 490,712 | 85,099 | ||||||
| Total current assets | 32,548,256 | 11,778,573 | ||||||
| PLANT AND EQUIPMENT, NET | 8,829,058 | 7,413,689 | ||||||
| CONSTRUCTION-IN-PROGRESS | 9,389,120 | 6,516,630 | ||||||
| OTHER ASSETS: | ||||||||
| Long-term prepayments | 7,980,307 | 5,207,117 | ||||||
| Intangible assets, net | 1,860,172 | 899,529 | ||||||
| Total other assets | 9,840,479 | 6,106,646 | ||||||
| Total assets | $ | 60,606,913 | $ | 31,815,538 | ||||
| LIABILITIES AND CHANGES IN EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Accounts payable | $ | 297,567 | $ | 547,430 | ||||
| Other payable and accrued expenses | 917,284 | 1,556,973 | ||||||
| Short-term loans | 220,050 | 748,170 | ||||||
| Short-term loans from shareholders | 110,025 | 308,070 | ||||||
| Deposits from customers | 1,275,958 | 424,266 | ||||||
| Taxes payable | 722,106 | 212,661 | ||||||
| Shares to be issued to related parties | 327,374 | 95,204 | ||||||
| Due to related parties | 185,024 | 242,225 | ||||||
| Total current liabilities | 4,055,388 | 4,134,999 | ||||||
| OTHER LIABILITIES: | ||||||||
| Deferred government grant | 1,100,250 | 1,100,250 | ||||||
| Warrant liability | 1,538,686 | - | ||||||
| Total other liabilities | 2,638,936 | 1,100,250 | ||||||
| Total liabilities | 6,694,324 | 5,235,249 | ||||||
| COMMITMENTS AND CONTINGENCIES | ||||||||
| CHANGES IN EQUITY: | ||||||||
| Preferred stock, $0.001 par value, 50,000,000 Series "A" shares authorized and 2,000,000 shares issued and outstanding as of December 31, 2008 48,000,000 Series "B" shares authorized, Nil Series "B" shares issued and outstanding as of December 31, 2008 | - | 2,000 | ||||||
| Common stock, $0.001 par value, 40,000,000 shares authorized, 6,989,640 and 3,733,038 shares issued and outstanding as of December 31, 2009 and December 31, 2008, respectively | 6,989 | 3,733 | ||||||
| Paid-in capital | 34,580,096 | 16,345,775 | ||||||
| Statutory reserves | 3,879,077 | 2,952,710 | ||||||
| Retained earnings | 12,574,906 | 4,418,464 | ||||||
| Accumulated other comprehensive income | 2,871,521 | 2,857,607 | ||||||
| Total shareholders' equity | 53,912,589 | 26,580,289 | ||||||
| Total liabilities and shareholders' equity | $ | 60,606,913 | $ | 31,815,538 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
See report of independent registered public accounting firm.
F-3
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
AND OTHER COMPREHENSIVE INCOME (LOSS)
For the Years Ended December 31, 2009 and 2008
| Years ended December 31, | ||||||||
| 2009 | 2008 | |||||||
| REVENUE, NET | $ | 33,778,305 | $ | 25,584,446 | ||||
| COST OF REVENUE | 16,520,989 | 12,808,896 | ||||||
| GROSS PROFIT | 17,257,316 | 12,775,550 | ||||||
| OPERATING EXPENSES: | ||||||||
| Research and development | 1,167,937 | 549,236 | ||||||
| Selling expenses | 1,928,441 | 1,381,807 | ||||||
| General and administrative | 2,466,470 | 2,663,520 | ||||||
| Total operating expenses | 5,562,848 | 4,594,563 | ||||||
| INCOME FROM OPERATIONS | 11,694,468 | 8,180,987 | ||||||
| OTHER INCOME (EXPENSE): | ||||||||
| Other income (expense), net | 117,873 | 30,906 | ||||||
| Interest income (expense), net | (62,590 | ) | (329,167 | ) | ||||
| Inducement cost for debentures converted | - | (756,855 | ) | |||||
| Change in fair value of warrants | (868,445 | ) | - | |||||
| Total other expense, net | (813,162 | ) | (1,055,116 | ) | ||||
| INCOME BEFORE PROVISION FOR INCOME TAXES | 10,881,306 | 7,125,871 | ||||||
| PROVISION FOR INCOME TAXES | 2,029,374 | 1,529,688 | ||||||
| NET INCOME | 8,851,932 | 5,596,183 | ||||||
| OTHER COMPREHENSIVE (LOSS) INCOME : | ||||||||
| Foreign currency translation adjustment | 13,914 | 1,415,005 | ||||||
| COMPREHENSIVE INCOME | $ | 8,865,846 | $ | 7,011,188 | ||||
| EARNINGS PER SHARE: | ||||||||
| Basic | $ | 1.65 | $ | 1.53 | ||||
| Diluted | $ | 1.62 | $ | 1.53 | ||||
| WEIGHTED AVERAGE NUMBER OF COMMON SHARES: | ||||||||
| Basic | 5,374,452 | 3,645,746 | ||||||
| Diluted | 5,459,528 | 3,649,396 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
See report of independent registered public accounting firm.
F-4
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF SHAREHOLDERS' EQUITY
| Accumulated | ||||||||||||||||||||||||||||||||||||
Retained earnings | other | |||||||||||||||||||||||||||||||||||
Preferred stock | Common stock | Paid-in | Statutory | comprehensive | ||||||||||||||||||||||||||||||||
Shares | Amount | Shares | Amount | capital | reserves | Unrestricted | income | Total | ||||||||||||||||||||||||||||
| BALANCE, January 31, 2008 | 2,000,000 | $ | 2,000 | 3,422,240 | $ | 3,422 | $ | 14,692,209 | $ | 1,652,720 | $ | 122,271 | $ | 1,442,602 | $ | 17,915,224 | ||||||||||||||||||||
| Stock-based compensation | 62,758 | 62,758 | ||||||||||||||||||||||||||||||||||
| Shares issued for services | 23,218 | 23 | 130,398 | 130,421 | ||||||||||||||||||||||||||||||||
| Shares issued for debt settlement | 42,080 | 42 | 220,878 | 220,920 | ||||||||||||||||||||||||||||||||
| Debentures converted to common stock | 245,500 | 246 | 1,239,532 | 1,239,778 | ||||||||||||||||||||||||||||||||
| Foreign currency translation | 1,415,005 | 1,415,005 | ||||||||||||||||||||||||||||||||||
| Net income | 5,596,183 | 5,596,183 | ||||||||||||||||||||||||||||||||||
| Appropriation to statutory reserve | 1,299,990 | (1,299,990 | ) | - | ||||||||||||||||||||||||||||||||
| BALANCE, December 31, 2008, as previously reported | 2,000,000 | 2,000 | 3,733,038 | 3,733 | 16,345,775 | 2,952,710 | 4,418,464 | 2,857,607 | 26,580,289 | |||||||||||||||||||||||||||
| Cumulative effect of reclassification of warrants | (1,108,508 | ) | 230,877 | (877,631 | ) | |||||||||||||||||||||||||||||||
| BALANCE, January 1, 2009, as adjusted, (Unaudited) | 2,000,000 | 2,000 | 3,733,038 | 3,733 | 15,237,267 | 2,952,710 | 4,649,341 | 2,857,607 | 25,702,658 | |||||||||||||||||||||||||||
| Shares issued for services | 12,438 | 12 | 63,002 | 63,014 | ||||||||||||||||||||||||||||||||
| Cancellation of preferred stock | (2,000,000 | ) | (2,000 | ) | 2,000 | - | ||||||||||||||||||||||||||||||
| Fractional shares due to the ten-for-one reverse split | 1,772 | 2 | (2 | ) | - | |||||||||||||||||||||||||||||||
| Shares issued for cash | 3,220,000 | 3,220 | 19,070,461 | 19,073,681 | ||||||||||||||||||||||||||||||||
| Cashless exercise of warrants | 22,392 | 22 | 207,368 | 207,390 | ||||||||||||||||||||||||||||||||
| Foreign currency translation | 13,914 | 13,914 | ||||||||||||||||||||||||||||||||||
| Net income | 8,851,932 | 8,851,932 | ||||||||||||||||||||||||||||||||||
| Appropriation to statutory reserves | 926,367 | (926,367 | ) | - | ||||||||||||||||||||||||||||||||
| BALANCE, December 31, 2009 | - | $ | - | 6,989,640 | $ | 6,989 | $ | 34,580,096 | $ | 3,879,077 | $ | 12,574,906 | $ | 2,871,521 | $ | 53,912,589 | ||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
See report of independent registered public accounting firm.
F-5
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
For the Years Ended December 31, 2009 and 2008
Years ended December 31, | ||||||||
2009 | 2008 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net income | $ | 8,851,932 | $ | 5,596,183 | ||||
Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||
| Depreciation | 566,230 | 443,062 | ||||||
| Amortization | 212,826 | 179,343 | ||||||
| Amortization of deferred financing costs | - | 101,815 | ||||||
| Amortization of discount on debentures | - | 680,446 | ||||||
| Amortization of deferred compensation | - | 62,758 | ||||||
| Inducement cost for debentures converted | - | 257,775 | ||||||
| Common stock issued for services | 63,014 | 130,421 | ||||||
| Common stock to be issued to related parties for compensation | 232,170 | 95,204 | ||||||
| Bad debt expense | - | 112,253 | ||||||
| Inducement cost for debt settlement | - | 42,081 | ||||||
| Change in fair value of warrant liability | 868,445 | - | ||||||
| Change in operating assets and liabilities | ||||||||
| Accounts receivable | (1,957,883 | ) | (1,068,391 | ) | ||||
| Inventories | (987,977 | ) | (674,486 | ) | ||||
| Deposits and prepaid expenses | (7,310,495 | ) | (3,647,834 | ) | ||||
| Other receivables | (705,365 | ) | 93,613 | |||||
| Accounts payable | (249,709 | ) | 404,642 | |||||
| Accrued expenses | 55,140 | 946,801 | ||||||
| Deposits from customers | 851,170 | 352,012 | ||||||
| Taxes payable | 509,132 | (389,081 | ) | |||||
| Other payables | 267,097 | (18,189 | ) | |||||
| Net cash provided by operating activities | 1,265,727 | 3,700,428 | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Proceeds from short-term investment | 351,864 | 57,660 | ||||||
| Refund (payments) of long-term prepayments | 2,713,950 | (3,833,669 | ) | |||||
| Prepayment for potential acquisition | (6,802,704 | ) | - | |||||
| Loans to third parties | (2,580,336 | ) | 315,472 | |||||
| Proceeds from loans receivable | 2,875,242 | - | ||||||
| Purchases of intangible assets | (1,172,880 | ) | - | |||||
| Purchases of plant and equipment | (529,470 | ) | (9,529 | ) | ||||
| Payments on construction-in-progress | (2,709,105 | ) | (1,606,280 | ) | ||||
| Net cash used in investing activities | (7,853,439 | ) | (5,076,346 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Decrease (increase) in restricted cash | 80,684 | (654 | ) | |||||
| Proceeds from short-term loans | 219,915 | 735,165 | ||||||
| Repayment for short-term loans | (747,711 | ) | - | |||||
| Proceeds from equity offering | 18,411,496 | - | ||||||
| Repayment to shareholders and directors | (307,881 | ) | - | |||||
| Proceeds from shareholders and directors | 109,958 | 401,616 | ||||||
| Due (from) to related parties | (57,223 | ) | - | |||||
| Net cash provided by financing activities | 17,709,238 | 1,136,127 | ||||||
| EFFECT OF EXCHANGE RATE CHANGES ON CASH | 1,463 | 44,708 | ||||||
| INCREASE (DECREASE) IN CASH | 11,122,989 | (195,083 | ) | |||||
| CASH, beginning of year | 576,409 | 771,492 | ||||||
| CASH, end of year | $ | 11,699,398 | $ | 576,409 | ||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | ||||||||
| Cash paid for interest | $ | 73,085 | $ | 35,177 | ||||
| Cash paid for income taxes | $ | 2,095,704 | $ | 740,899 | ||||
| Non-cash investing and financing activities | ||||||||
| Long-term prepayment transferred to construction-in-progress | $ | 190,458 | $ | - | ||||
| Construction-in-progress transferred to property, plant and equipment | 1,818,403 | 1,133,580 | ||||||
| Issuance of common stock for debt settlement | $ | - | $ | 178,839 | ||||
| Debentures converted to common stock | $ | - | $ | 482,923 | ||||
| Cashless exercise of warrants | $ | 207,390 | - | |||||
| Expense paid through contribution receivable | $ | 662,185 | - | |||||
| Interest expense capitalized as construction-in-progress | $ | - | $ | 114,990 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
See report of independent registered public accounting firm.
F-6
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2009
Note 1 - ORGANIZATION
Organization and description of business
Skystar Bio-Pharmaceutical Company (“Skystar” or the “Company”), was incorporated in Nevada on September 24, 1998. Since its acquisition on November 7, 2005 of Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”), a Cayman Islands company, the Company has been engaged in research, development, production, marketing and sales of veterinary healthcare and medical care products. All current operations of the Company are in the People’s Republic of China (“China” or the “PRC”).
All of the Company’s operations are carried out by Xian Tianxing Bio-Pharmaceutical Co., Limited (“Xian Tianxing”), a PRC joint stock company that the Company controls through contractual arrangements originally between Skystar Cayman and Xian Tianxing. On March 10, 2008, the Company entered into a series of agreements transferring all of the rights and obligations of Skystar Cayman under the contractual arrangements to Sida Biotechnology (Xian) Co., Ltd. (“Sida”), a PRC company. Sida is the wholly owned subsidiary of Fortunate Time International Limited (“Fortunate Time”), a Hong Kong company and wholly owned subsidiary of Skystar Cayman. Xian Tianxing also has a wholly owned subsidiary, Shanghai Siqiang Biotechnological Co., Ltd. (“Shanghai Siqiang”), a PRC company.
As a result of these contractual arrangements, which obligates Sida to absorb all of the risk of loss from Xian Tianxing’s activities and enable Sida to receive all of its expected residual returns, the Company accounts for Xian Tianxing as a variable interest entity (“VIE”) under Financial Accounting Standards Board’s (“FASB”) interpretation on consolidation of variable interest entities. Accordingly, the Company consolidates Xian Tianxing’s results, assets and liabilities.
Sida was established by Fortunate Time on July 10, 2007, with registered capital of $5,000,000. Fortunate Time invested $2,000,000 into Sida on July 20, 2007, which amount is payable to Skystar Cayman. On July 9, 2009, Fortunate Time invested the remaining $3,000,000 into Sida. Xi’an High Technology District approved Sida’s application to increase its registered capital to $15,000,000 on July 13, 2009. On July 15, 2009, Sida received the $10,000,000 additional registered capital from Fortunate Time. Funds from Fortunate Time for $13,000,000 was from the cash proceeds of the equity offering which is further discussed in Note 14.
On September 18, 2009, Skystar Bio-Pharmaceutical Inc. (“Skystar California”) was incorporated in California and became a wholly-owned subsidiary of Skystar.
Hereinafter, Skystar, Skystar California, Skystar Cayman, Fortunate Time, Sida, Xian Tianxing and Shanghai Siqiang are sometimes collectively referred to as the “Company.”
Note 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principles of consolidation
The accompanying consolidated financial statements are prepared in accordance with accounting principles generally accepted in the United States of America. The consolidated financial statements include the financial statements of the Company, its wholly-owned subsidiaries, and its VIEs. All significant inter-company transactions and balances between the Company, its subsidiaries and VIEs are eliminated upon consolidation.
See report of independent registered public accounting firm.
F-7
Use of estimates
The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. For example, the Company estimates its allowance for doubtful accounts and useful lives of plant and equipment. Because of the use of estimates inherent in the financial reporting process, actual results could materially differ from those estimates upon which the carrying values were based.
Foreign currency translation
The Company uses the United States dollar (“U.S. dollar”) for financial reporting purposes and the Chinese Renminbi (“RMB”) as its functional currency. The Company’s subsidiaries and VIEs maintain their books and records in their functional currency, being the primary currency of the economic environment in which their operations are conducted.
The Company translates the subsidiaries’ and VIEs’ assets and liabilities into U.S. dollars using the applicable exchange rates prevailing at the balance sheet dates, and the statements of operations and cash flows are translated at average exchange rates during the reporting period. As a result, amounts related to assets and liabilities reported on the consolidated statements of cash flows will not necessarily agree with changes in the corresponding balances on the consolidated balance sheets. Equity accounts are translated at historical rates. Adjustments resulting from the translation of the subsidiaries’ and VIEs’ financial statements are recorded as accumulated other comprehensive income.
The quotation of the exchange rates does not imply free convertibility of RMB to other foreign currencies. All foreign exchange transactions continue to take place either through the People's Bank of China or other banks authorized to buy and sell foreign currencies at the exchange rate quoted by the People's Bank of China. The rates of exchange quoted by the People’s Bank of China on December 31, 2009 and 2008 were US $1.00 to RMB 6.82 and RMB 6.82, respectively. The average translation rates of US $1.00 to RMB 6.82 and RMB 6.94 was applied to the income statement accounts for the years ended December 31, 2009 and 2008, respectively.
Approval of foreign currency payments by the People’s Bank of China or other institutions requires submitting a payment application form together with invoices, shipping documents and signed contracts. Transaction gains and losses that arise from exchange rate fluctuations on transactions denominated in a currency other than the functional currency are included in the results of operations as incurred.
Fair values of financial instruments
On January 1, 2008, the Company adopted the accounting standards regarding fair value of financial instruments and related fair value measurements defines financial instruments and requires fair value disclosures of those financial instruments. This accounting standard defines fair value, establishes a three-level valuation hierarchy for disclosures of fair value measurement and enhances disclosures requirements for fair value measures. Current assets and current liabilities qualified as financial instruments and management believes their carrying amounts are a reasonable estimate of fair value because of the short period of time between the origination of such instruments and their expected realization and if applicable, their current interest rate is equivalent to interest rates currently available. The three levels of valuation hierarchy are defined as follows:
| • | Level 1 inputs to the valuation methodology are quoted prices (unadjusted) for identical assets or liabilities in active markets. |
See report of independent registered public accounting firm.
F-8
| • | Level 2 inputs to the valuation methodology include quoted prices for similar assets and liabilities in active markets, and inputs that are observable for the assets or liability, either directly or indirectly, for substantially the full term of the financial instruments. |
| • | Level 3 inputs to the valuation methodology are unobservable and significant to the fair value measurement. |
Effective January 1, 2009, the Company adopted the provisions of an accounting standard regarding whether an instrument (or embedded feature) is indexed to an entity’s own stock. This accounting standard specifies that a contract that would otherwise meet the definition of a derivative but is both (a) indexed to the Company’s own stock and (b) classified in stockholders’ equity in the statement of financial position would not be considered a derivative financial instrument. It provides a new two-step model to be applied in determining whether a financial instrument or an embedded feature is indexed to an issuer’s own stock and thus able to qualify for the scope exception within the standards.
As a result of the foregoing adoption, 309,100 common stock purchase warrants previously treated as equity pursuant to the derivative treatment exemption are no longer afforded equity treatment because the strike price of the warrants is denominated in U.S. dollars, a currency other than the Company’s functional currency, the RMB. As a result, the warrants are not considered indexed to the Company’s own stock, and as such, all future changes in the fair value of these warrants will be recognized currently in earnings until such time as the warrants are exercised or expired.
As such, effective January 1, 2009, the Company reclassified the fair value of these warrants from equity to liability, as if these warrants were treated as a derivative liability since their issuance in February 2007. On January 1, 2009, the Company reclassified from additional paid-in capital, as a cumulative effect adjustment, $230,877 to beginning retained earnings and $877,631 to warrant liability to recognize the fair value of such warrants. The Company recognized a loss of $868,445 from the change in fair value of the warrant liability for the year ended December 31, 2009.
These warrants do not trade in an active securities market, and as such, the Company estimates the fair value of these warrants using the Black-Scholes Option Pricing Model (“Black-Scholes Model”) using the following assumptions:
| Warrants (1) | Warrants (2) | |||||||||||||||
December 31, 2009 | January 1, 2009 | December 31, 2009 | January 1, 2009 | |||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||
| Stock price | $ | 10.10 | $ | 4.750 | $ | 10.10 | $ | 4.75 | ||||||||
| Exercise price | $ | 6.00 | $ | 6.00 | $ | 5.00 | $ | 5.00 | ||||||||
| Annual dividend yield | — | — | — | — | ||||||||||||
| Expected term (years) | .17 | 1.20 | 2.17 | 3.20 | ||||||||||||
| Risk-free interest rate | 0.04 | % | 0.875 | % | 1.14 | % | 1.125 | % | ||||||||
| Expected volatility | 34 | % | 140 | % | 178 | % | 130 | % | ||||||||
| (1) | As of January 1, 2009, 195,000 warrants with an exercise price of $6.00 were outstanding. As of December 31, 2009, 145,000 warrants with an exercise price of $6.00 were outstanding. |
| (2) | As of January 1, 2009, 114,100 warrants with an exercise price of $5.00 were outstanding. As of December, 31, 2009, 107,254 warrants with an exercise price of $5.00 were outstanding. |
See report of independent registered public accounting firm.
F-9
Expected volatility is based on historical volatility. Historical volatility was computed using daily pricing observations for recent periods that correspond to the term of the warrants. The Company believes this method produces an estimate that is representative of future volatility over the expected term of these warrants. The Company has no reason to believe future volatility over the expected remaining life of these warrants is likely to differ materially from historical volatility. The expected life is based on the remaining term of the warrants. The risk-free interest rate is based on U.S. Treasury securities according to the remaining term of the warrants.
As required by the FASB’s accounting standards, financial assets and liabilities are classified in their entirety based on the lowest level of input that is significant to the fair value measurement. Depending on the product and the terms of the transaction, the fair values warrant liability were modeled using a series of techniques, including closed-form analytic formula, such as the Black-Scholes Model, which does not entail material subjectivity because the methodology employed does not necessitate significant judgment, and the pricing inputs are observed from actively quoted markets.
The fair value of the 252,254 warrants as of December 31, 2009 was determined using the Black-Scholes Model, defined in the FASB’s accounting standard of fair value measurement as level 2 inputs, and recorded the change in earnings. As a result, the warrant liability is carried on the consolidated balance sheets at fair value.
The following table sets forth by level within the fair value hierarchy our financial assets and liabilities that were accounted for at fair value on a recurring basis as of December 31, 2009:
Carrying Value at December 31, 2009 | Fair Value Measurement at December 31, 2009 | |||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||
| Warrant liability | $ | 1,538,686 | $ | - | $ | 1,538,686 | $ | - | ||||||||
In addition to assets and liabilities that are recorded at fair value on a recurring basis, the Company is required to record assets and liabilities at fair value on a non-recurring basis. Generally, assets are recorded at fair value on a non-recurring basis as a result of impairment charges. For the year ended December 31, 2009, there were no impairment charges.
Revenue recognition
Revenue of the Company is primarily from the sales of veterinary healthcare and medical care products in China. Sales are recognized when the following four revenue criteria are met: persuasive evidence of an arrangement exists, delivery has occurred, the selling price is fixed or determinable, and collectability is reasonably assured. Sales are presented net of value added tax (“VAT”). No allowance for sales returns is made on these consolidated financial statements as sales returns are de minimis based on historical experience.
There are two types of sales upon which revenue is recognized:
| a. | Credit sales: Revenue is recognized when the products have been delivered to the customers. |
| b. | Full payment before delivering: Revenue is recognized when the products have been delivered to customers. |
Shipping and handling costs related to costs of goods sold are included in selling expenses, which totaled $850,387 and $335,605 for the years ended December 31, 2009 and 2008, respectively.
See report of independent registered public accounting firm.
F-10
The Company’s revenues and cost of revenues by product line were as follows:
| Years Ended December 31, | ||||||||
| 2009 | 2008 | |||||||
| Revenues | ||||||||
| Micro-organism | $ | 8,021,139 | $ | 5,868,623 | ||||
| Veterinary Medications | 22,920,479 | 17,535,757 | ||||||
| Feed Additives | 1,411,222 | 1,189,108 | ||||||
| Vaccines | 1,425,465 | 990,958 | ||||||
| Total Revenues | $ | 33,778,305 | $ | 25,584,446 | ||||
| Cost of Revenues | ||||||||
| Micro-organism | $ | 2,129,945 | $ | 1,781,598 | ||||
| Veterinary Medications | 13,672,332 | 10,389,726 | ||||||
| Feed Additives | 568,007 | 525,653 | ||||||
| Vaccines | 150,705 | 111,919 | ||||||
| Total Cost of Revenues | 16,520,989 | 12,808,896 | ||||||
| Gross Profit | $ | 17,257,316 | $ | 12,775,550 | ||||
Cash
Cash includes cash on hand, demand deposits with banks and liquid investments with an original maturity of three months or less.
Restricted cash
Restricted cash is comprised of amounts received from the PRC government as subsidies and set aside for specific uses (see Note 13). Restricted cash is maintained as bank deposits and reflected as current assets based on the expected period when such funds will be put into their specific uses.
Short-term investments
Short-term investments are comprised of securities classified as available-for-sale, held by a private investment trust company for investing activities with a fixed rate of return on investment. Available-for-sale securities are carried at fair value, with unrealized gains or losses reported in other comprehensive income, net of tax. Realized gains or losses are included in the results of operations. There was no unrealized gain or loss relating to short-term investments for the years ended December 31, 2009 and 2008, and as such, no such amounts were included in other comprehensive income for such periods.
The Company entered into an investment agreement with an unrelated party in 2007 to invest certain funds in the Chinese stock market. The nature of the investment is available-for-sale with certain guaranteed returns such as 7.2% interest through August 1, 2008 and 5.0% interest after August 1, 2008 for one year. There are no restrictions or penalties for early withdrawals prior to maturity. The investment matured on July 31, 2009 and the investment principal of $352,080 and accrued interest of $22,666 were received by the Company on August 3, 2009.
Accounts receivable and other receivables
Accounts receivable are recorded at net realizable value consisting of the carrying amount less an allowance for uncollectible accounts, as needed. The Company uses the aging method to estimate the allowance for anticipated uncollectible receivable balances. Under the aging method, bad debt percentages determined by management based on historical experience, as well as current economic climate, are applied to customers’ balances categorized by the number of months the underlying invoices have remained outstanding. At each reporting period, the allowance balance is adjusted to reflect the amount computed as a result of the aging method. When facts subsequently become available to indicate that the allowance provided requires an adjustment, a corresponding adjustment is made to the allowance account as a change in estimate. The ultimate collection of the Company’s accounts receivable may take one year. Delinquent account balances are reserved after management determines that the likelihood of collection is not probable, and known bad debts are written-off against allowance for doubtful accounts when identified.
See report of independent registered public accounting firm.
F-11
Inventories
Inventories are stated at the lower of cost or market, as determined on a moving weighted-average basis. Inventories include purchases and related costs incurred in bringing the inventories to their present location and condition. Management reviews inventories for obsolescence and cost in excess of net realizable value at least annually and records a reserve against the inventory and additional cost of goods sold when the carrying value exceeds net realizable value.
Plant and equipment
Plant and equipment are stated at cost less accumulated depreciation. Depreciation is computed using the straight-line method over the estimated useful lives of the assets. Expenditures for maintenance and repairs which do not improve or extend the useful lives of the assets are charged to operations as incurred, while renewals and betterments are capitalized. Gains and losses on disposals are included in the results of operations. Estimated useful lives of the assets are as follows:
Estimated Useful Life | |||
| Buildings | 20-40 years | ||
| Machinery and equipment | 10 years | ||
| Computer, office equipment and furniture | 5 years | ||
| Vehicles | 5-10 years | ||
Management assesses the carrying value of plant and equipment annually, more often when factors indicating impairment are present, and reduces the carrying value of such assets by the amount of the impairment. The Company determines the existence of such impairment by measuring the expected future cash flows (undiscounted and without interest charges) and comparing such amount to the net asset carrying value. An impairment loss, if it exists, is measured as the amount by which the carrying amount of the asset exceeds the fair value of the asset. Based on its review, management believes that, as of December 31, 2009 and 2008, there was no impairment for its plant and equipment.
Construction-in-progress
Construction-in-progress includes direct costs of construction of a factory building. Interest incurred during the period of construction, if significant, is capitalized. All other interest is expensed as incurred. Construction-in-progress is not depreciated until such time the assets are completed and put into service.
Intangible assets
Land Use Rights — Land use rights represent the amounts paid to acquire a long-term interest to utilize the land underlying the Company’s facilities. This type of arrangement is common for the use of land in the PRC. Land use rights are amortized on a straight-line basis over its 50-year term.
See report of independent registered public accounting firm.
F-12
Technological Know-How — Purchased technological know-how includes confidential formulas, manufacturing processes, technical and procedural manuals, and is amortized using the straight-line method over the weighted average useful life of nine years, which reflects the period over which such confidential formulas, manufacturing processes, and technical and procedural manuals are kept confidential by the Company as agreed between the Company and the selling parties.
Impairment of Intangible Assets — The Company evaluates the carrying value of intangible assets annually, or more often when factors indicating impairment are present. The Company determines the existence of such impairment by measuring the estimated future cash flows (undiscounted) and comparing such amount to the net asset carrying value. If the undiscounted cash flow estimated to be generated by any such intangible asset is less than its carrying amount, a loss is recognized based on the amount by which the carrying amount exceeds the intangible asset’s fair market value. Loss on intangible assets to be disposed of is determined in a similar manner, except that fair market values are reduced by the cost of disposal. Based on its review, the Company believes that, as of December 31, 2009, there was no impairment of its intangible assets.
Comprehensive income
The FASB’s accounting standard of reporting comprehensive income requires disclosure of all components of comprehensive income and loss on an annual and interim basis. Comprehensive income and loss is defined as the change in equity of a business enterprise during a period from transactions and other events and circumstances from non-owner sources. The accompanying consolidated financial statements include the provisions of GAAP. Accumulated other comprehensive income is comprised of the changes in foreign currency exchange rates.
Research and development costs
Research and development costs are charged to operations as incurred and include salaries, professional fees and technical support fees related to such efforts.
Advertising costs
Advertising costs are charged to operations currently. Advertising costs for the years ended December 31, 2009 and 2008 were $253,006 and $93,573 respectively.
Income taxes
The Company accounts for income taxes in accordance with the FASB’s accounting standard for income taxes. Under the asset and liability method as required by this accounting standard, deferred income taxes are recognized for the tax consequences of temporary differences by applying enacted statutory tax rates applicable to future years to differences between the financial statement carrying amounts and the tax bases of existing assets and liabilities. The effect on deferred income taxes of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is recognized if it is more likely than not that some portion, or all of, a deferred tax asset will not be realized.
Further, in accordance with this accounting standard, a tax position is recognized as a benefit only if it is “more likely than not” that the tax position would be sustained in a tax examination, with a tax examination being presumed to occur. The amount recognized is the largest amount of tax benefit that is greater than 50% likely of being realized on examination. For tax positions not meeting the “more likely than not” test, no tax benefit is recorded. The standard also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosures, and transition. The adoption had no effect on the Company’s consolidated financial statements.
See report of independent registered public accounting firm.
F-13
The Company’s operations are subject to income and transaction taxes in the United States and in the PRC jurisdictions. Significant estimates and judgments are required in determining the Company’s worldwide provision for income taxes. Some of these estimates are based on interpretations of existing tax laws or regulations. The ultimate amount of tax liability may be uncertain as a result.
The Company does not anticipate any events which could cause a change to these uncertainties.
Stock-based compensation
The Company records and reports stock-based compensation by measuring the cost of services received in exchange for an award of equity instruments based on the grant-date fair value of the award. That cost is recognized over the period during which services are received. Stock compensation for stock granted to non-employees is determined as the fair value of the consideration received or the fair value of equity instruments issued, whichever is more reliably measured.
The Company uses the Black-Scholes Model which was developed for use in estimating the fair value of options. This models requires the input of highly complex and subjective variables including the expected life of options granted and the Company’s expected stock price volatility over a period equal to or greater than the expected life of the options. Because changes in the subjective assumptions can materially affect the estimated value of the Company’s employee stock options, it is management’s opinion that the Black-Scholes Model may not provide an accurate measure of the fair value of the Company’s employee stock options. Although the fair value of employee stock options is determined in accordance with the standards using an option-pricing model, that value may not be indicative of the fair value observed in a willing buyer/willing seller market transaction.
Earnings per share
The Company reports earnings per share and present both basic and diluted earnings per share in conjunction with the disclosure of the methodology used in computing such earnings per share. Basic earnings per share is based upon the weighted-average number of common shares outstanding. Diluted earnings per share is based on the assumption that all dilutive convertible shares, including convertible preferred shares, and stock options were converted or exercised. Further, the method requires that stock dividends or stock splits be accounted for retroactively if the stock dividends or stock splits occur during the period, or retroactively if the stock dividends or stock splits occur after the end of the period but before the release of the financial statements, by considering it outstanding of the entirety of each period presented. Dilution is computed by applying the treasury stock method. Under this method, options and warrants are assumed to be exercised at the beginning of the period (or at the time of issuance, if later), and as if funds obtained thereby were used to purchase common stock at the average market price during the period.
All share and per share amounts used in the Company's consolidated financial statements and notes thereto have been retroactively restated to reflect the 1-for-10 reverse stock split effectuated on May 12, 2009, and the 2-for-1 forward stock split effectuated on November 16, 2009.
Related parties
Parties are considered to be related to the Company if the parties, directly or indirectly, through one or more intermediaries, control, are controlled by, or are under common control with the Company. Related parties also include principal owners of the Company, its management, members of the immediate families of such principal owners and management, and other parties with which the Company may deal if one party controls or can significantly influence the management or operating policies of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests.
See report of independent registered public accounting firm.
F-14
Reclassifications
Certain prior period amounts have been reclassified to conform to the current period presentation. These reclassifications had no impact on previously reported net income or cash flows.
Recently issued accounting pronouncements
In January 2009, the FASB issued an accounting standard amending the Impairment Guidance of recognition of interest income and impairment on purchased and retained beneficial interests in securitized financial assets. The newly issued accounting standard changes the impairment model included to be more consistent with the impairment model of another accounting standard for accounting for securities. The new accounting standard remove its exclusive reliance on “market participant” estimates of future cash flows used in determining fair value. Changing the cash flows used to analyze other-than-temporary impairment from the “market participant” view to a holder’s estimate of whether there has been a “probable” adverse change in estimated cash flows allows companies to apply reasonable judgment in assessing whether an other-than-temporary impairment has occurred. The adoption of this FASB Staff Positions did not have a material impact the Company’s consolidated financial statements.
In April 2009, the FASB issued three related FASB Staff Positions: (i) Recognition of Presentation of Other-Than-Temporary Impairments, (ii) Interim Disclosures about Fair Value of Financial Instruments, and (iii) Determining the Fair Value When the Volume and Level of Activity for the Asset or Liability Have Significantly Decreased and Identifying Transactions That Are Not Orderly, which are effective for interim and annual reporting periods ending after June 15, 2009. The first Staff Position modifies the requirement for recognizing other-than-temporary impairments, changes the existing impairment model, and modifies the presentation and frequency of related disclosures. The second Staff Position requires disclosures about fair value of financial instruments for interim reporting periods as well as in annual financial statements. The third Staff Position requires new disclosures regarding the categories of fair value instruments, as well as the inputs and valuation techniques utilized to determine fair value and any changes to the inputs and valuation techniques during the period. The adoption of these FASB Staff Positions did not have a material impact the Company’s consolidated financial statements.
In May 2009, the FASB an accounting standard which provides guidance to establish general standards of accounting for and disclosures of events that occur after the balance sheet date but before financial statements are issued or are available to be issued. The standard also requires entities to disclose the date through which subsequent events were evaluated as well as the rationale for why that date was selected. The standard is effective for interim and annual periods ending after June 15, 2009, and accordingly, the Company adopted this Standard as of December 31, 2009. The standard requires that public entities evaluate subsequent events through the date that the financial statements are issued.
In June 2009, the FASB issued an accounting standard which establishes the FASB Accounting Standards Codification™ (the “Codification”) as the source of authoritative accounting principles recognized by the FASB to be applied by nongovernmental entities in the preparation of financial statements in conformity with U.S. GAAP. The Codification does not change current U.S. GAAP, but is intended to simplify user access to all authoritative U.S. GAAP by providing all the authoritative literature related to a particular topic in one place. The Codification is effective for interim and annual periods ending after September 15, 2009, and as of the effective date, all existing accounting standard documents will be superseded. The Codification is effective for the Company in the third quarter of 2009, and accordingly, the Company’s Annually Report on Form 10-K for the year ended December 31, 2009 and all current and subsequent public filings will reference the Codification as the sole source of authoritative literature.
In August 2009, the FASB issued an Accounting Standards Update (“ASU”) regarding measuring liabilities at fair value. This ASU provides additional guidance clarifying the measurement of liabilities at fair value in circumstances in which a quoted price in an active market for the identical liability is not available; under those circumstances, a reporting entity is required to measure fair value using one or more of valuation techniques, as defined. This ASU is effective for the first reporting period, including interim periods, beginning after the issuance of this ASU. The adoption of this ASU did not have a material impact on the Company’s consolidated financial statements.
See report of independent registered public accounting firm.
F-15
In October 2009, the FASB issued an ASU regarding accounting for own-share lending arrangements in contemplation of convertible debt issuance or other financing. This ASU requires that at the date of issuance of the shares in a share-lending arrangement entered into in contemplation of a convertible debt offering or other financing, the shares issued shall be measured at fair value and be recognized as an issuance cost, with an offset to additional paid-in capital. Further, loaned shares are excluded from basic and diluted earnings per share unless default of the share-lending arrangement occurs, at which time the loaned shares would be included in the basic and diluted earnings-per-share calculation. This ASU is effective for fiscal years beginning on or after December 15, 2009, and interim periods within those fiscal years for arrangements outstanding as of the beginning of those fiscal years. The Company is currently evaluating the impact of this ASU on its consolidated financial statements.
In January 2010, the FASB issued ASU No. 2010-02 regarding accounting and reporting for decreases in ownership of a subsidiary. Under this guidance, an entity is required to deconsolidate a subsidiary when the entity ceases to have a controlling financial interest in the subsidiary. Upon deconsolidation of a subsidiary, an entity recognizes a gain or loss on the transaction and measures any retained investment in the subsidiary at fair value. In contrast, an entity is required to account for a decrease in its ownership interest of a subsidiary that does not result in a change of control of the subsidiary as an equity transaction. This ASU clarifies the scope of the decrease in ownership provisions, and expands the disclosures about the deconsolidation of a subsidiary or de-recognition of a group of assets. This ASU is effective beginning in the first interim or annual reporting period ending on or after December 31, 2009. The adoption of this ASU did not have a material impact the Company’s consolidated financial statements.
In January 2010, FASB issued ASU No. 2010-06 – Improving Disclosures about Fair Value Measurements. This update provides amendments to Subtopic 820-10 that requires new disclosure to include transfers in and out of Levels 1 and 2 and activity in Level 3 fair value measurements. Further, this update clarifies existing disclosures on level of disaggregation and Disclosures about inputs and valuation techniques. A reporting entity should provide fair value measurement disclosures for each class of assets and liabilities and should provide disclosures about the valuation techniques and inputs used to measure fair value for both recurring and nonrecurring fair value measurements. Those disclosures are required for fair value measurements that fall in either Level 2 or Level 3. The new disclosures and clarifications of existing disclosures are effective for interim and annual reporting periods beginning after December 15, 2009, except for the disclosures about purchases, sales, issuances, and settlements in the roll forward of activity in Level 3 fair value measurements. Those disclosures are effective for fiscal years beginning after December 15, 2010, and for interim periods within those fiscal years. The Company is currently evaluating the impact of this ASU; however, the Company does not expect the adoption of this ASU to have a material impact on its consolidated financial statements.
Note 3 - CONCENTRATIONS AND CREDIT RISK
The Company’s operations are all carried out in the PRC. Accordingly, the Company’s business, financial condition and results of operations may be influenced by the political, economic and legal environments in the PRC, and by the general state of the PRC’s economy. The Company’s operations in the PRC are subject to specific considerations and significant risks not typically associated with companies in North America and Western Europe. These include risks associated with, among others, the political, economic and legal environments and foreign currency exchange. The Company’s results may be adversely affected by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversion and remittance abroad, and rates and methods of taxation, among other things.
Financial instruments, which subject the Company to concentration of credit risk, consist of cash. The Company maintains balances at financial institutions which, from time to time, may exceed federally insured limits for the banks located in the United States. Balances at financial institutions or state-owned banks within the PRC are not covered by insurance. As of December 31, 2009 and 2008, the Company had deposits in excess of federally insured limits (including restricted cash) of $11,504,970 and $448,082 respectively. The Company has not experienced any losses in such accounts.
See report of independent registered public accounting firm.
F-16
For the year ended December 31, 2009 and 2008, all of the Company’s sales occurred in the PRC. No major customers accounted for more than 10% of the Company’s total revenues. In addition, all accounts receivable at December 31, 2009 and 2008 also arose in the PRC.
The Company’s five largest vendors accounted for approximately 59% of the Company’s total purchases for the year ended December 31, 2009, while the Company’s four largest vendors accounted for 59% of the Company’s total purchases for the year ended December 31, 2008. As of December 31, 2009 and 2008, there were no amounts due to those five and four largest vendors, respectively.
The Company had one product that accounted for 20% and 12% of the Company’s total revenues for the years ended December 31, 2009 and 2008, respectively.
Note 4 - - ACCOUNTS RECEIVABLE, NET
Accounts receivable consisted of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Account receivable | $ | 4,711,044 | $ | 2,751,959 | ||||
| Allowance for bad debts | (327,857 | ) | (327,857 | ) | ||||
| Account receivable, net | $ | 4,383,187 | $ | 2,424,102 | ||||
The following table presents the movement of allowance for doubtful accounts:
| Allowance for bad debt, January 1, 2008 | $ | 199,639 | ||
| Addition | 114,239 | |||
| Recovery | — | |||
| Translation adjustment | 13,979 | |||
| Allowance for bad debt, December 31, 2008 | 327,857 | |||
| Addition | — | |||
| Recovery | — | |||
| Translation adjustment | — | |||
| Allowance for bad debt, December 31, 2009 | $ | 327,857 |
Note 5 – INVENTORIES
Inventories consist of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Raw materials | $ | 2,798,021 | $ | 2,087,428 | ||||
| Packing materials | 159,620 | 165,077 | ||||||
| Work-in-process | 886 | 2,446 | ||||||
| Finished goods | 1,096,547 | 811,538 | ||||||
| Other | 19,571 | 19,571 | ||||||
| Total | $ | 4,074,645 | $ | 3,086,060 | ||||
See report of independent registered public accounting firm.
F-17
The Company periodically reviews its reserves for slow moving and obsolete inventories. As of December 31, 2009 and 2008, the Company believes that no such reserves were necessary.
Note 6 - DEPOSITS AND PREPAID EXPENSES
Deposits and prepaid expenses are comprised of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Prepayment for raw materials purchasing | $ | 10,990,913 | $ | 4,210,618 | ||||
| Prepayment for packaging materials purchasing | 489,392 | 499,755 | ||||||
| Prepayment for advertisement fee | - | 89,436 | ||||||
| Prepayment for due diligence fee | - | 73,350 | ||||||
| Other | 420,009 | 5,692 | ||||||
| Total | $ | 11,900,314 | $ | 4,878,851 | ||||
Note 7 – LOANS RECEIVABLE
Loans receivable consist of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Shaanxi Xinbangdike Technology Developing Company (1) | $ | - | $ | 295,087 | ||||
(1) Loan to Shaanxi Xinbangdike Technology Development Company is non-interest bearing, unsecured and due on demand.
Note 8 - PLANT AND EQUIPMENT, NET
Plant and equipment consist of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Building and improvements | $ | 6,798,616 | $ | 4,977,654 | ||||
| Machinery and equipment | 3,035,814 | 3,035,376 | ||||||
| Office equipment and furniture | 191,424 | 186,702 | ||||||
| Vehicles | 485,156 | 329,331 | ||||||
| Total | 10,511,010 | 8,529,063 | ||||||
| Less: accumulated depreciation | (1,681,952) | (1,115,374 | ) | |||||
| Plant and equipment, net | $ | 8,829,058 | $ | 7,413,689 | ||||
Depreciation expense was $566,230 and $443,062 for the years ended December 31, 2009 and 2008, respectively.
See report of independent registered public accounting firm.
F-18
Note 9 - CONSTRUCTION-IN-PROGRESS
Construction-in-progress (“CIP”) is related to a plant being built in accordance with the PRC’s Good Manufacturing Practices (“GMP”) Standard. Construction on this plant commenced in 2005. The veterinary medicine facility and the building that houses quality control, research and development and administration were completed during 2007, and the remaining plant facilities are expected to be completed by June 30, 2010, at an estimated cost of $11,846,479. During the year ended December 31, 2009, vaccine and micro-organism facilities were completed resulting in a transfer from CIP to property, plant and equipment of $2,951,288. No depreciation is provided for construction-in-progress until such time the assets are completed and placed into service.
The construction projects the company is in the progress of completing are:
Total in CIP as of 12/31/09 | Estimate cost to Complete | Estimated Total Cost | Estimated Completion Date (1) | ||||||||||
| Project | |||||||||||||
| Vaccine facility | $ | 7,846,479 | $ | 2,500,000 | $ | 10,346,479 | June 2010 | ||||||
| Micro-organism facility | 1,339,371 | 160,629 | 1,500,000 | April 2010 | |||||||||
| Other | 203,270 | ||||||||||||
| TOTAL CIP Balance | $ | 9,389,120 | $ | 2,660,629 | $ | 11,846,479 | |||||||
| (1) | Note that this date does not include the time to certify the facility, if necessary. |
As of December 31, 2009 and 2008, the Company had construction in progress amounting to $9,389,120 and $6,516,330, respectively. $0 and $150,789 in interest expense had been capitalized for construction in progress for the years ended December 31, 2009 and 2008, respectively.
Note 10 - LONG-TERM PREPAYMENTS
Long-term prepayments consist of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Construction deposit | $ | 733,500 | $ | 2,493,167 | ||||
| Deposit for building purchase | 439,927 | - | ||||||
| Deposit for potential acquisitions | 6,806,880 | 2,713,950 | ||||||
| Total | $ | 7,980,307 | $ | 5,207,117 | ||||
As of December 31, 2009 and 2008, the Company has determined that these prepayments are noncurrent because: (1) these amounts relate to noncurrent assets, and (2) the Company’s ability to complete any potential acquisitions is contingent upon the Company identifying and negotiating with target companies.
As of December 31, 2008, deposits for potential acquisitions represent deposits made to four separate potential acquisition targets: one that produces veterinary medicines, one that produces micro-organisms and feed additives, one that produces veterinary medicines for domestic pets, and one with buildings for the Company’s own future expansion. In April 2009, all deposits made by the Company for potential acquisitions as of December 31, 2008 in the amount of $2,713,950 (RMB 18,500,000) were returned to the Company.
As of December 31, 2009, deposits for office building purchase of $439,927 (RMB 2,998,820) represent deposits made for new office building. The Company is in the process of applying for property certificate, and made the remaining payment of $1,069,018 (RMB 7,287,105) in January, 2010.
See report of independent registered public accounting firm.
F-19
As of December 31, 2009, deposits for potential acquisitions of $6,806,880 (RMB 46,400,000) represent deposits made to two separate potential acquisition targets. The Company obtained appraisal report in January 2010 for the target in Hubei, stating the total fair value of net assets at $2,711,603 (RMB 18,484,000). The Company is expected to finish the acquisition in June 2010.
Note 11 – INTANGIBLE ASSETS
Intangible assets consisted of the following:
December 31, 2009 | December 31, 2008 | |||||||
| Land use rights | $ | 378,853 | $ | 378,853 | ||||
| Technological know-how | 2,053,800 | 880,200 | ||||||
| Total | 2,432,653 | 1,259,053 | ||||||
| Less: accumulated amortization | (572,481 | ) | (359,524 | ) | ||||
| Intangible assets, net | $ | 1,860,172 | $ | 899,529 | ||||
During 2007, the Company paid $658,350 for the exclusive rights to the use of a strain of micro-bio organisms for a five-year term from November 1, 2007 through October 31, 2012. The Company began using the strain in its veterinary medicines in 2008.
In 2009, the Company paid $1,172,880 (RMB 8,000,000) for fish disease technology transfer for an eleven-year term from September 2009 through September 2020.
For the year ended December 31, 2009 and 2008, the amortization expense for intangibles amounted to $212,826 and $179,343, respectively.
Amortization expense for the future five years and thereafter is as follows:
| Years ending December 31, | Amount | |||
| 2010 | $ | 388,757 | ||
| 2011 | 388,757 | |||
| 2012 | 364,322 | |||
| 2013 | 242,148 | |||
| 2014 | 183,504 | |||
| 2015 and thereafter | 292,684 | |||
| Total | $ | 1,860,172 | ||
Note 12 – SHORT-TERM LOANS
On September 3, 2008, the Company signed a one year short-term agreement with the Bank of East Asia to borrow up to $1.9 million (RMB 13 million) for operating purposes secured by the Company’s land use rights and buildings. On September 17, 2008, the Company received proceeds of $733,500 (RMB 5 million) from the bank. The balance of $1.1 million (RMB 8 million) can be borrowed once the Company has a deposit balance in the amount of $4 million with the bank and uses the bank as the Company’s primary transaction bank. The applicable interest rate of the loan is the Bank of China’s standard short-term rate, 6.93% at inception of the loan, which is subject to change with the government policy, plus an additional 20% interest rate float. Pursuant to these terms, the interest rates were approximately 6.37% and 8.32% as of December 31, 2009 and December 31, 2008, respectively. Interest payments to the Bank of East Asia are due every six months. The Company repaid amount of $733,050 (RMB 5 million) on September 16, 2009.
See report of independent registered public accounting firm.
F-20
Pursuant to the short-term loan agreement, the Company is required to comply with certain covenants, such as maintaining sufficient environmental controls with respect to the Company’s manufacturing processes, and providing written notification to the bank for any changes in the organizational structure or executive officers, among others. As of December 31, 2009, the Company determined that it was in compliance with these covenants.
On January 14, 2009, the Company signed a one year short-term loan contract with Shaanxi Agricultural Yanta Credit Union for $220,050 (RMB 1.5 million) at an annual interest rate of 8.66% for operating purposes secured by the personal property of Weibing Lu, the Company’s Chief Executive Officer. This loan was paid off on January 15, 2010.
Interest expense incurred and associated with the short-term loans amounted to $71,467 for the year ended December 31, 2009, none of which has been capitalized as part of construction-in-progress in 2009. Interest expense incurred and associated with the short term loan amounted to $15,741 for the year ended December 31, 2008, which has been capitalized as part of construction-in-progress.
Note 13 - DEFERRED GOVERNMENT GRANT
Deferred government grant represents subsidies for GMP projects granted by the PRC government. A subsidy in the amount of $641,000 was approved by the PRC government for the Company to construct a new factory in which operations will meet the GMP Standard. In 2003, $516,500 was received by the Company and the remaining $124,500 was received in the first quarter of 2006. In 2006, the Company expended $186,644 for the construction of its new factory (Note 9).
Also in 2003, the Company received a second subsidy in the amount of $256,400 to finance the Company’s research and development activities. In 2005, the Company received a third subsidy of $64,100 for the Company’s research and development activities, which was expended during that year.
According to PRC government regulations, the funds granted may be treated as capital contributed by a company appointed by the PRC government or as a loan from such company, which the Company will be required to repay. As of December 31, 2009, the Company has not reached a final agreement with the PRC government regarding the treatment of these three subsidies as either a loan or capital contribution, and the Company does not expect that the final agreement will be completed within the next year. Therefore, these amounts are reflected as non-current liabilities in the accompanying consolidated financial statements.
Note 14 - CAPITAL TRANSACTIONS
On May 12, 2009, the Company effectuated a 1-for-10 reverse stock split of its issued and outstanding shares of common stock and a proportional reduction of its authorized shares of common stock. Number of common stock, warrants, options disclosed in the footnotes has been retroactively restated to reflect the 1-for-10 reverse stock split. On November 16, 2009, the Company effectuated a 2-for-1 forward split of its issued and outstanding common stock and a proportional increase of its authorized shares of common stock. Number of common stock, warrants, options disclosed in the footnotes has been retroactively restated to reflect the 2-for-1 forward stock split.
Preferred stock
On June 25, 2009, the Company’s board of directors concluded that 2,000,000 shares of series “A” preferred stock issued in 2001 were not valid because no certificate of designation was filed prior to their issuance as required under Nevada corporate law. On December 21, 2009, the Company instructed its transfer agent to remove these preferred shares officially from its shareholder records. As of December 31, 2009, no share of preferred stock is outstanding.
See report of independent registered public accounting firm.
F-21
Stock-based compensation
On February 12, 2008, the Company issued 18,000 shares of common stock as salary to a non-executive director. The trading value of the Company's common stock on February 12, 2008, was $5.55 per share and a corresponding amount of $99,900 was charged to general and administrative expenses. The Company also had a $302,372 and $95,204 balance under shares to be issued as of December 31, 2009 and 2008, which represented 47,334 and 22,000 common shares to be issued to the non-executive director for his service provided for the period from May 2008 to December 2009, and for the period from May 2008 to December 2008, respectively. The amounts were included in general and administrative expenses based on the weighted-average trading price of the Company’s common stock for the said periods.
On February 29, 2008, the Company issued 42,080 shares of common stock to its legal counsel as partial payment for services rendered. The trading value of the Company's common stock on February 29, 2008, was $5.25 per share and additional inducement cost of $42,081 between the fair value of the shares at $220,920 and the partial payment of $178,839 was charged to general and administrative expenses.
On April 21, 2008, two of the Company’s convertible debenture holders converted $982,003 of debentures into 245,500 shares of common stock.
On May 5, 2008, the Company agreed to issue 10,434 shares of common stock to its chief financial officer (“CFO”) during the term of a one-year agreement, which would vest in four equal installments of 2,608 shares each quarter. The trading value of the common stock on May 5, 2008 was $5.85 per share for a total value of $61,042. These shares were fully vested. On May 26, 2009, the Company renewed the one-year service agreement with the CFO and agreed to issue 14,440 shares of common stock, which would vest in four equal installments of 3,610 shares every quarter starting August 5, 2009. Compensation expense is recognized on a straight-line basis over the vesting period. Total compensation expense of $32,490 was charged to general and administrative expenses for the year ended December 31, 2009.
On May 26, 2009, the Company agreed to issue 5,556 shares of common stock to a director at the beginning of each term of his directorship. The trading value of the common stock on May 26, 2009 was $4.50 per share for the total value of $25,002, and the amount was charged to general and administrative expenses for the year ended December 31, 2009. As of December 31, 2009, the balance was under shares to be issued.
Warrants
On February 28, 2007, the Company issued 195,000 warrants to four investors with an exercisable price of $6.00 per share for a term of three years. On the same date, the Company also issued warrants to the private placement agent, exercisable for 114,100 shares of the Company’s common stock at a price of $5.00 per share for a five-year term. As of December 31, 2009, 56,846 warrants have been exercised. The Company valued the conversion on exercise date and recorded $207,390 losses from changes in fair value of warrants. Following is a summary of the status of warrants outstanding at December 31, 2009:
| Number of | Average Remaining | Average | |||||
| Warrants | Contractual Life | Exercise Price | |||||
| 145,000 | 0.16 years | $ | 6.00 | ||||
| 107,254 | 2.17 years | $ | 5.00 | ||||
| 252,254 | $ | 5.57 | |||||
See report of independent registered public accounting firm.
F-22
Following is an activity summary of the Company’s outstanding warrants:
| Outstanding as of December 31, 2007 | 309,100 | |||
| Granted | — | |||
| Forfeited | — | |||
| Exercised | — | |||
| Outstanding as of December 31, 2008 | 309,100 | |||
| Granted | — | |||
| Forfeited | — | |||
| Exercised | 56,846 | |||
| Outstanding as of December 31, 2009 | 252,254 |
Equity offering
On June 30, 2009, the Company and Rodman & Renshaw, LLC, as representative of underwriters (the "Underwriters") entered into an Underwriting Agreement. Pursuant to the Underwriting Agreement, Skystar agreed to issue and sell an aggregate of 3,220,000 shares (including 420,000 over-allotment shares) of the Company’s common stock, at a price of $6.49 per share in a public offering. The closing date of this offering was on the third business day following the effective date of the registration statement registering the shares offered, or July 3, 2009.
In connection with this offering, the Company agreed to grant 140,000 common stock purchase options to five individuals from the Underwriters. The options are exercisable from June 30, 2010 to June 30, 2014, and each option is exercisable for one share of the Company’s common stock, with exercise price at $8.11 per share. The Company used the Black-Scholes Model to value the options granted, which amounted to $1,065,842. The value of options granted to these individuals was included as part of the offering costs, and had no net effect on the Company’s equity.
The following are the assumptions used by the Company in the Black-Scholes Model:
Number of options | Stock price | Exercise price | Expected term | Dividend yield | Volatility | Risk-free interest rate | |||||||||||||||
| 140,000 | $ | 8.97 | $ | 8.11 | 3.0 years | — | 161 | % | 1.67 | % | |||||||||||
The following is a summary of the status of options outstanding at September 30, 2009:
| Outstanding Options | Exercisable Options | |||||||||||||||||||
Average Remaining | Average Remaining | |||||||||||||||||||
Number of Options | Contractual Life | Average Exercise Price | Number of Options | Contractual Life | Average Exercise Price | |||||||||||||||
| 140,000 | 4.75 | $ | 8.11 | — | — | $ | — | |||||||||||||
Following is an activity summary of the Company’s outstanding options:
Number of Options Outstanding | Weighted Average Exercise Price | Aggregate Intrinsic Value | ||||||||||
| Outstanding as of December 31, 2008 | — | — | — | |||||||||
| Granted | 140,000 | $ | 8.11 | — | ||||||||
| Forfeited | — | — | — | |||||||||
| Exercised | — | — | — | |||||||||
| Outstanding as of December 31, 2009 | 140,000 | $ | 8.11 | $ | — | |||||||
See report of independent registered public accounting firm.
F-23
Incremental costs incurred of this offering, including underwriting commission, legal fees, and printing costs were $1,730,477, and were directly deducted from the proceeds. The gross proceeds of this offering were $20,897,800. The Company received cash proceeds of $18,411,496 on July 6, 2009.
Equity Compensation Plan
On December 8, 2009, the Company’s board of directors approved a stock incentive plan for officers, directors, employees and consultants entitled the “Skystar Bio-Pharmaceutical Company 2010 Stock Incentive Plan” (hereinafter the “2010 Plan”). The maximum number of shares that may be issued under the 2010 Plan is 700,000 shares of the Company’s common stock. The 2010 Plan was approved by the Company’s stockholders on December 31, 2009, and awards may be granted under this Plan until December 7, 2019. As of December 31, 2009, there are 700,000 shares of the Company’s common stock remaining available for future issuance under the 2010 Plan.
Note 15 - CONVERTIBLE DEBENTURES
In February 2007, the Company sold an aggregate of $4.075 million of its 8% convertible debentures (the “Debentures”) and issued warrants to purchase 815,000 shares of common stock to several institutional and accredited investors. On April 21, 2008, the Company entered into an Amendment and Waiver Agreement (the “Amendment”) with two institutional and accredited investors who acquired two remaining unconverted Debentures in a private transaction from the original holders of these Debentures. A summary of the Amendment is as follows:
| • | The Amendment amends the terms of these Debentures by: (a) changing the conversion price from $5.00 per share to $4.00 per share; (b) deleting the trading conditions for mandatory conversion; (c) granting the Company the right to mandatory conversion at any time, and (d) allowing the Company to designate the date for the mandatory conversion. |
| • | The Amendment is deemed to be the Company’s notice to require conversion of the entire outstanding principal of these Debentures and all accrued but unpaid interest thereon. |
The difference between the value of the conversion option at the previous prices and their value at the modified prices are deemed costs for the Company and are charged to operations. The inducement cost for the two Debentures converted was $257,775 for year ended December 31, 2008. The inducement cost for the converted Debentures was based on the market value of the additional 49,100 shares obtained by these two investors at $5.25 per share on April 21, 2008.
245,500 shares of common stock were issued upon conversion of the two Debentures with a carrying value of $982,003 (including $490,713 of default premium) at a reduced conversion price of $4.00.
In accordance with the accounting standard of application of certain convertible instruments, all unamortized discount amounting to $291,548 at the time of the conversion was recognized as interest expense year ended December 31, 2008. The unamortized deferred financing costs of $79,998 on conversion of the Debentures were also recorded as interest expense for the year ended December 31, 2008.
Note 16 - STATUTORY RESERVES
Statutory reserves represent restricted retained earnings. Based on the legal formation of the entities, all PRC entities are required to set aside 10% of its net income as reported in its statutory accounts on an annual basis to the statutory surplus reserve fund. Once the total statutory surplus reserve reaches 50% of the registered capital of the respective subsidiaries, further appropriations are discretionary. The statutory surplus reserve can be used to increase the registered capital and eliminate future losses of the respective companies under PRC GAAP. The Company’s statutory surplus reserve is not distributable to shareholders except in the event of liquidation. As of December 31, 2009, Xian Tianxing has met the statutory surplus reserve requirement, and approximately $7,534,275 still needs to be transferred to the statutory surplus reserve from Shanghai Siqiang and Sida.
See report of independent registered public accounting firm.
F-24
The reserve fund can be used to increase the registered capital upon approval by relevant government authorities and eliminate future losses of the respective companies upon a resolution by the board of directors.
Appropriations to the above statutory reserves are accounted for as a transfer from unrestricted earnings to statutory reserves. There are no legal requirements in the PRC to fund these statutory reserves by the transfer of cash to any restricted accounts, and as such, the Company has not transferred any cash to these accounts. These reserves are not distributable as cash dividends.
Note 17 – TAXES
Skystar and Skystar California are subject to the United States federal income tax provision. Whereas its subsidiary, Skystar Cayman, is a tax-exempt company incorporated in the Cayman Islands and conducts all of its business through its subsidiaries, Fortunate Time, Sida, and Sida’s PRC VIEs, Xian Tianxing and Shanghai Siqiang.
Sida, Xian Tianxing, and Shanghai Siqiang are subject to PRC’s Enterprise Income Tax. Pursuant to the PRC Income Tax Laws, Enterprise Income Tax is generally imposed at a statutory rate of 25% beginning on January 1, 2008. Xian Tianxing has been approved as a new technology enterprise, and under PRC Income Tax Laws is entitled to a preferential tax rate of 15%.
For the years ended December 31, 2009 and 2008, the provisions for income tax were as follows:
| 2009 | 2008 | |||||||
| Current PRC income tax expense Enterprise income tax | $ | 2,029,374 | $ | 1,529,688 | ||||
The following table reconciles the U.S. statutory rates to the Company's effective tax rate as of December 31:
| 2009 | 2008 | |||||||
| U.S. Statutory rate | 34.0 | % | 34.0 | % | ||||
| Foreign income not recognized in the U.S. | (34.0 | ) | (34.0 | ) | ||||
| China income tax rate | 25.0 | 25.0 | ||||||
| China income tax exemption | (10.0 | ) | (10.0 | ) | ||||
Other item (1) | 3.7 | 6.5 | ||||||
| Total provision for income taxes | 18.7 | % | 21.5 | % | ||||
| (1) | The other item is operating expenses incurred by Skystar that are not deductible in the PRC which resulted in an increase in effective tax rate of 3.7% and 6.5% for the years ended December 31, 2009 and 2008, respectively. |
Taxes payable consisted of the following:
| December 31, 2009 | December 31, 2008 | |||||||
| Income taxes payable | $ | 104,261 | $ | 200,937 | ||||
Value added tax (1) | 561,646 | - | ||||||
| Other taxes | 56,199 | 11,724 | ||||||
| Total | $ | 722,106 | $ | 212,661 | ||||
| (1) | In 2009, the increase in value added tax (“VAT”) is mainly due to increase in sales. In 2008, the Company made more payments than its VAT on sales resulting in prepaid expenses that can be used to offset future VAT on sales. |
See report of independent registered public accounting firm.
F-25
The estimated tax savings due to the reduced tax rate for the years ended December 31, 2009 and 2008 amounted to $202,934 and $1,019,600, respectively. If the statutory income tax had been applied, the Company would have decreased basic earnings per share and diluted per shares from $1.65 to $1.61 and from $1.62 to $1.58 for the years ended December 31, 2009. For the year ended December 31, 2008, the basic and diluted earnings per share would have decreased from $1.53 to $1.47 if the statutory income tax had been applied.
Skystar is incorporated in the U.S. and has incurred a net operating loss for income tax purposes for 2009. As of December 31, 2009, the estimated net operating loss carryforwards for U.S. income tax purposes amounted to $4,569,638 which may be available to reduce future years’ taxable income. These carryforwards will expire, if not utilized, beginning in 2026 and continue through 2029. Management believes that the realization of the benefits arising from this loss appears to be uncertain due to the Company’s limited operating history and continuing losses for U.S. income tax purposes. Accordingly, the Company has provided a 100% valuation allowance at December 31, 2009 and 2008. The valuation allowance at December 31, 2009 and 2008 was $1,553,677 and $967,424, respectively. The Company’s management reviews this valuation allowance periodically and makes adjustments as necessary.
The Company has cumulative undistributed earnings of foreign subsidiaries of approximately $25,508,239 as of December 31, 2009, which are included in consolidated retained earnings and will continue to be indefinitely reinvested in international operations. Accordingly, no provision has been made for U.S. deferred taxes related to future repatriation of these earnings, nor is it practicable to estimate the amount of income taxes that would have to be provided if we concluded that such earnings will be remitted in the future.
Note 18 - EARNINGS PER SHARE
The following is the calculation of earnings per share:
For the years ended December 31, | ||||||||
| 2009 | 2008 | |||||||
| Net income | $ | 8,851,932 | $ | 5,596,183 | ||||
| Weighted average shares used in basic computation | 5,374,452 | 3,645,746 | ||||||
| Diluted effect of stock warrants | 85,076 | 3,650 | ||||||
| Weighted average shares used in diluted computation | 5,459,528 | 3,649,396 | ||||||
| Earnings per share: | ||||||||
| Basic | $ | 1.65 | $ | 1.53 | ||||
| Diluted | $ | 1.62 | $ | 1.53 | ||||
See report of independent registered public accounting firm.
F-26
At December 31, 2009 and 2008, the Company had 252,254 and 309,100 warrants outstanding, respectively. For the year ended December 31, 2009, the average stock price was greater than the exercise prices of warrants which resulted in additional weighted-average common stock equivalents of 85,076. For the year ended December 31, 2008, the average stock price was greater than the exercise price of 114,100 warrants outstanding at such time which resulted in additional weighted average common stock equivalents of 3,650 shares. As such, 195,000 outstanding warrants were excluded from the diluted earnings per share calculation as they were anti-dilutive.
For the year ended December 31, 2009, 140,000 outstanding options were excluded from the diluted earnings per share calculation as they are anti-dilutive.
Note 19 - RELATED PARTY TRANSACTIONS AND ARRANGEMENTS
Amounts receivable from and payable to related parties are summarized as follows:
December 31, 2009 | December 31, 2008 | |||||||
| Short-term loans from shareholders | ||||||||
Mr. Weibing Lu – officer and shareholder (1) (2) | $ | 36,675 | $ | 220,050 | ||||
Mr. Wei Wen – officer and shareholder (2) | 36,675 | 44,010 | ||||||
Ms. Aixia Wang – shareholder (2) | 36,675 | 44,010 | ||||||
| Total | $ | 110,025 | $ | 308,070 | ||||
| Shares to be issued to related party | ||||||||
Scott Cramer – non-executive director (3) | $ | 302,372 | $ | 95,204 | ||||
Mark D. Chen – non-executive director (3) | 25,002 | — | ||||||
| Total | $ | 327,374 | $ | 95,204 | ||||
| Amounts due (from) to related parties | ||||||||
Bennet P. Tchaikovsky – CFO (4) | $ | - | $ | 13,168 | ||||
Scott Cramer – non-executive director and shareholder (4) | 143,556 | 224,684 | ||||||
Shaanxi Xingji Electronics Co. - owned by a director's wife (4) | - | 4,373 | ||||||
Officer and shareholder (4) | 41,468 | - | ||||||
| Total | $ | 185,024 | $ | 242,225 | ||||
(1) In 2008, Weibing Lu obtained an unsecured personal loan in the amount of $176,040 (RMB 1,500,000) from Huaxia Bank with annual interest rate of 7.47% and advanced to Xian Tianxing to facilitate operations. Xian Tianxing guaranteed the loan. The loan principal and related interest was due on December 30, 2008. On January 4, 2009, Xian Tianxing paid the full principal amount to the bank, with related interest of $15,741.
(2) On May 29, 2008, Weibing Lu, Wei Wen and Aixia Wang obtained personal loans from Yanta Credit Union and advanced cash to Xian Tianxing in the total amount of $132,030 to facilitate operations. These loans, which were due on May 29, 2009 with 8.436% interest per annum and guaranteed by Xian Tianxing, were paid in full on May 29, 2009. On June 2, 2009, Mr. Lu, Mr. Wen and Ms. Wang again obtained loans from the same bank and advanced cash to Xian Tianxing in the total amount of $110,025. These loans are due on June 1, 2010, with 10.11% interest per annum and are also guaranteed by Xian Tianxing. For the year ended December 31, 2009 and 2008, Xian Tianxing paid interest of $ 0 and $3,695, respectively, for these loans.
(3) As of December 31, 2009 and December 31, 2008, the Company had $302,372 (representing 47,334 common shares) and $95,204 balances (representing 22,000 common shares), respectively, under agreement to issue shares to Scott Cramer as compensation for being a representative of the Company in the United States for the periods from May 2008 to December 31, 2009, and December 31, 2008, respectively. In addition, as of December 31, 2009, the Company had $25,002 balance (representing 5,556 common shares) under agreement to issue shares to Mark D. Chen as compensation at the beginning of each term of his directorship.
See report of independent registered public accounting firm.
F-27
(4) Shaanxi Xinji Electronics Co., Ltd. is owned by the wife of Weibing Lu. The amounts due to Shaanxi Xinji Electronics as of December 31, 2009 and December 31, 2008 were short-term cash transfers for business operations, non-interest bearing, unsecured, and payable upon demand. As of December 31, 2009, the Company also had $41,468 payable to officers and shareholders for advance for short-term financing purposes. As of December 31, 2009 and December 31, 2008, the Company also had amounts due to Scott Cramer for bonus and Scott Cramer and Bennet P. Tchaikovsky the expenses paid by them on behalf of the Company.
Note 20 - COMMITMENTS AND CONTINGENCIES
(a) Lease commitments
The Company recognizes lease expense on a straight-line basis over the term of the lease in accordance to the FASB’s accounting standard of accounting for leases. The Company entered into a tenancy agreement for the lease of factory premises for a period of ten years from October 1, 2004 to December 31, 2014, with annual rent of $13,563 (or RMB 94,600), which is subject to a 10% increase every four subsequent years.
The Company leases office space from Weibing Lu, the Company’s chief executive officer, for a period of five years from January 1, 2007 to December 31, 2011, with annual rent of approximately $24,000 (or RMB 165,600). The Company also entered into a tenancy agreement with Weibing Lu for the lease of Shanghai Siqiang’s office for a period of ten years from August 1, 2007 to August 1, 2017, with annual rent of approximately $21,000 (or RMB 144,000).
The Company entered into a tenancy agreement for the lease of an office space in California for a period of three years from July 1, 2009 to July 1, 2012 with monthly rent of $1,100.
The minimum future lease payments for the next five years and thereafter are as follows:
| Period | Amount | |||
| Year ending December 31, 2010 | $ | 61,195 | ||
| Year ending December 31, 2011 | 61,195 | |||
| Year ending December 31, 2012 | 35,949 | |||
| Year ending December 31, 2013 | 34,981 | |||
| Year ending December 31, 2014 | 34,981 | |||
| Year ending December 31, 2015 and thereafter | 56,298 | |||
| Total | $ | 284,599 | ||
Rental expense for the years ended December 31, 2009 and 2008 amounted to $63,660 and $63,303, respectively.
(b) Legal proceedings
From time to time, the Company is involved in legal matters arising in the ordinary course of business. Management currently is not aware of any legal matters or pending litigation, which would have a significant effect on the Company’s consolidated financial statements as of December 31, 2009.
In May 2007, Andrew Chien filed suit against the Company, Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in United States District Court for the District of Connecticut, alleging causes of action for violation of Sections 10(b) and 20(a) of the Exchange Act. On July 17, 2008, in a decision that is now published, the Court granted defendants' motion to dismiss and subsequently dismissed the lawsuit, entering judgment on behalf of the defendants. Mr. Chien filed a notice of appeal of the Court's dismissal of his lawsuit, opposed by the defendants, which remains pending. Additionally, on February 5, 2009, the Court issued a ruling on defendants' motion for sanctions, finding the action filed by Mr. Chien to have been entirely frivolous, and to have constituted a "substantial" violation of Federal Rule of Civil Procedure Rule 11, and imposed significant monetary sanctions on both Mr. Chien and his former attorney. As part of the basis for imposing sanctions on Mr. Chien personally, the Court specifically found that Mr. Chien had knowledge of facts directly contradicting the allegations of his complaint, as evident in internet postings he made on online message boards. Mr. Chien subsequently filed motions seeking to "re-open" this case, and to recuse the judge, but both motions were denied.
See report of independent registered public accounting firm.
F-28
A Notice of Appeal concerning the ruling awarding sanctions against him was also filed by Mr. Chien. All appeals, including the one referenced below concerning Mr. Chien's second lawsuit, were subsequently consolidated and remain pending, although briefing has been completed.
Subsequently, Mr. Chien, proceeding pro se, filed another lawsuit against the Company, Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in Connecticut Superior Court, alleging causes of action similar to those alleged in his federal complaint described above as well as state law causes of action. The case was removed to the U.S. District Court, District of Connecticut, and assigned to the same Judge who dismissed Mr. Chien’s related federal action. On June 8, 2009, the Court granted defendants’ motion to dismiss this action in its entirety, and denied Mr. Chien’s motion to further amend his complaint. Mr. Chien filed a Notice of Appeal concerning the ruling dismissing this lawsuit, which has been consolidated with Mr. Chien’s appeal of his other lawsuit.
Other than the above described legal proceedings, the Company is not aware of any other legal matters in which purchasers, any director, officer, or any owner of record or beneficial owner of more than five percent of any class of voting securities of the Company, or any affiliate of purchaser, or of any such director, officer, affiliate of the Company, or security holder, is a party adverse to the Company or has a material adverse interest to the Company. No provision has been made in the consolidated financial statements for the above contingencies.
(c) Ownership of leasehold property
In 2005, a shareholder contributed a leasehold office building as additional capital of Xian Tianxing. However, the title of the leasehold property has not passed to the Company. The Company does not believe there are any legal barriers for the shareholder to transfer the ownership to the Company. However, in the event that the Company fails to obtain the ownership certificate for the leasehold property, there is a risk that the building will need to be vacated due to unofficial ownership. Management believes that this possibility is remote, and as such, no provision has been made in the consolidated financial statements for this potential occurrence.
(d) R&D Project
During the first quarter of 2008, Xian Tianxing contracted with Northwestern Agricultural Technology University to jointly work on an R&D project concerning the application of nano-technology in the prevention of major milk cow disease. The total projected budget for this project is approximately $574,000 (RMB 4 million) which is to be paid according to the completed stages of the project. The Company expects this project to be completed in 2010. As of December 31, 2009, the Company incurred approximately $102,627 (RMB 700,000) of expenses relating to this project. The project reached trial stage in June 2009 and the Company expects to obtain veterinary permit for the new product from government on 2010.
During 2008, Xian Tianxing contracted with Shannxi Shenzhou Bio-pharmaceuticals Technology Company to jointly work on a R&D project with a contracted amount of approximately $308,000. As of December 31, 2009, the Company incurred approximately $102,627 (RMB 700,000) expenses relating to this project. This project was completed by December 31, 2009.
During the year ended December 31, 2009, Xian Tianxing contracted with the Fourth Military Medical University to jointly work on a R&D project with a contracted amount of approximated $880,200(RMB 6,000,000). As of December 31, 2009, the Company incurred approximately $704,314 (RMB 4,804,000) expenses relating to this project.
Note 21 - SUBSEQUENT EVENTS
On January 19, 2010, a holder of 45,000 warrants issued in February 2007 and priced at $6.00 per share which were set to expire in February 2010, exercised their cashless option which resulted in the issuance of 21,148 shares.
On January 19, 2010, a holder of 57,050 warrants issued in February 2007 and priced at $5.00 per share which were to expire in February 2012, exercised their cashless option which resulted in the issuance of 31,851 shares.
On January 20, 2010, a holder of 100,000 warrants issued in February 2007 and priced at $6.00 per share which were set to expire in February 2010, exercised their cashless option which resulted in the issuance of 47,670 shares.
See report of independent registered public accounting firm.
F-29
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
| March 31, | December 31, | |||||||
2010 | 2009 | |||||||
Unaudited | ||||||||
ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash | $ | 5,911,415 | $ | 11,699,398 | ||||
| Accounts receivable, net of allowance for doubtful accounts of $327,857 and $327,857 as of March 31, 2010 and December 31, 2009, respectively | 3,447,078 | 4,383,187 | ||||||
| Inventories | 4,996,515 | 4,074,645 | ||||||
| Deposits and prepaid expenses | 12,173,047 | 11,900,314 | ||||||
| Other receivables | 572,641 | 490,712 | ||||||
| Total current assets | 27,100,696 | 32,548,256 | ||||||
| PLANT AND EQUIPMENT, NET | 10,415,008 | 8,829,058 | ||||||
| CONSTRUCTION-IN-PROGRESS | 9,741,767 | 9,389,120 | ||||||
| OTHER ASSETS: | ||||||||
| Long-term prepayments | 13,276,071 | 7,980,307 | ||||||
| Intangible assets, net | 1,768,771 | 1,860,172 | ||||||
| Total other assets | 15,044,842 | 9,840,479 | ||||||
| Total assets | $ | 62,302,313 | $ | 60,606,913 | ||||
LIABILITIES AND SHAREHOLDER'S EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Accounts payable | $ | 421,031 | $ | 297,567 | ||||
| Other payable and accrued expenses | 694,400 | 917,284 | ||||||
| Short-term loans | - | 220,050 | ||||||
| Short-term loans from shareholders | 110,025 | 110,025 | ||||||
| Deposits from customers | 1,850,051 | 1,275,958 | ||||||
| Taxes payable | 825,082 | 722,106 | ||||||
| Shares to be issued to related parties | 329,397 | 327,374 | ||||||
| Due to related parties | 106,743 | 185,024 | ||||||
| Total current liabilities | 4,336,729 | 4,055,388 | ||||||
| OTHER LIABILITIES: | ||||||||
| Deferred government grant | 1,100,250 | 1,100,250 | ||||||
| Warrant liability | 510,571 | 1,538,686 | ||||||
| Total other liabilities | 1,610,821 | 2,638,936 | ||||||
| Total liabilities | 5,947,550 | 6,694,324 | ||||||
| COMMITMENTS AND CONTINGENCIES | ||||||||
| SHAREHOLDER'S EQUITY: | ||||||||
| Preferred stock, $0.001 par value, Nil Series "A" shares authorized as of March 31, 2010 and December 31, 2009, 48,000,000 Series "B" shares authorized, Nil Series "B" shares issued and outstanding as of March 31, 2010 and December 31, 2009 | - | - | ||||||
| Common stock, $0.001 par value, 40,000,000 shares authorized, 7,097,708 and 6,989,640 shares issued and outstanding as of March 31, 2010 and December 31, 2009, respectively | 7,097 | 6,989 | ||||||
| Paid-in capital | 35,966,731 | 34,580,096 | ||||||
| Statutory reserves | 3,879,077 | 3,879,077 | ||||||
| Retained earnings | 13,671,153 | 12,574,906 | ||||||
| Accumulated other comprehensive income | 2,830,705 | 2,871,521 | ||||||
| Total shareholders' equity | 56,354,763 | 53,912,589 | ||||||
| Total liabilities and shareholders' equity | $ | 62,302,313 | $ | 60,606,913 | ||||
The accompanying notes are an integral part of the consolidated financial statements.
F-30
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF INCOME AND OTHER COMPREHENSIVE INCOME
(UNAUDITED)
| For Three Months Ended March 31, | ||||||||
| 2010 | 2009 | |||||||
| REVENUE, NET | $ | 4,869,243 | $ | 3,823,566 | ||||
| COST OF REVENUE | 2,291,219 | 1,946,358 | ||||||
| GROSS PROFIT | 2,578,024 | 1,877,208 | ||||||
| OPERATING EXPENSES: | ||||||||
| Research and development | 43,995 | 117,352 | ||||||
| Selling expenses | 171,134 | 207,395 | ||||||
| General and administrative | 619,550 | 314,695 | ||||||
| Total operating expenses | 834,679 | 639,442 | ||||||
| INCOME FROM OPERATIONS | 1,743,345 | 1,237,766 | ||||||
| OTHER INCOME (EXPENSE): | ||||||||
| Other income (expense), net | 417 | (232 | ) | |||||
| Interest income (expense), net | (4,816 | ) | 302 | |||||
| Change in fair value of warrant liability | (317,380 | ) | 38,328 | |||||
| Total other income (expense), net | (321,779 | ) | 38,398 | |||||
| INCOME BEFORE PROVISION FOR INCOME TAXES | 1,421,566 | 1,276,164 | ||||||
| PROVISION FOR INCOME TAXES | 325,319 | 211,520 | ||||||
| NET INCOME | 1,096,247 | 1,064,644 | ||||||
| OTHER COMPREHENSIVE LOSS: | ||||||||
| Foreign currency translation adjustment | (40,816 | ) | (38,448 | ) | ||||
| COMPREHENSIVE INCOME | $ | 1,055,431 | $ | 1,026,196 | ||||
| EARNINGS PER SHARE: | ||||||||
| Basic | $ | 0.16 | $ | 0.29 | ||||
| Diluted | $ | 0.15 | $ | 0.29 | ||||
| WEIGHTED AVERAGE NUMBER OF COMMON SHARES: | ||||||||
| Basic | 7,061,530 | 3,734,602 | ||||||
| Diluted | 7,140,140 | 3,734,602 | ||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-31
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF SHAREHOLDERS' EQUITY
| Accumulated | ||||||||||||||||||||||||||||
Retained earnings | other | |||||||||||||||||||||||||||
Preferred stock | Common stock | Paid-in | Statutory | comprehensive | ||||||||||||||||||||||||
Shares | Amount | Shares | Amount | capital | reserves | Unrestricted | income | Total | ||||||||||||||||||||
| BALANCE, January 1, 2009, as adjusted | 2,000,000 | $ | 2,000 | 3,733,038 | $ | 3,733 | $ | 15,237,267 | $ | 2,952,710 | $ | 4,649,341 | $ | 2,857,607 | $ | 25,702,658 | ||||||||||||
| Shares issued for services | 2,608 | 3 | 15,049 | 15,052 | ||||||||||||||||||||||||
| Foreign currency translation | (38,448 | ) | (38,448 | ) | ||||||||||||||||||||||||
| Net income | 1,064,644 | 1,064,644 | ||||||||||||||||||||||||||
| Appropriation to statutory reserves | 106,464 | (106,464 | ) | - | ||||||||||||||||||||||||
| BALANCE, March 31, 2009 (unaudited) | 2,000,000 | 2,000 | 3,735,646 | 3,736 | 15,252,316 | 3,059,174 | 5,607,521 | 2,819,159 | 26,743,906 | |||||||||||||||||||
| Shares issued for services | 9,830 | 9 | 47,953 | 47,962 | ||||||||||||||||||||||||
| Cancellation of preferred stock | (2,000,000 | ) | (2,000 | ) | 2,000 | - | ||||||||||||||||||||||
| Fractional shares due to the ten-for-one reverse split | 1,772 | 2 | (2 | ) | - | |||||||||||||||||||||||
| Shares issued for cash | 3,220,000 | 3,220 | 19,070,461 | 19,073,681 | ||||||||||||||||||||||||
| Cashless exercise of warrants | 22,392 | 22 | 207,368 | 207,390 | ||||||||||||||||||||||||
| Foreign currency translation | 52,362 | 52,362 | ||||||||||||||||||||||||||
| Net income | 7,787,288 | 7,787,288 | ||||||||||||||||||||||||||
| Appropriation to statutory reserves | 819,903 | (819,903 | ) | - | ||||||||||||||||||||||||
| BALANCE, December 31, 2009 | - | - | 6,989,640 | 6,989 | 34,580,096 | 3,879,077 | 12,574,906 | 2,871,521 | 53,912,589 | |||||||||||||||||||
| Shares issued for services | 9,166 | 9 | 41,238 | 41,247 | ||||||||||||||||||||||||
| Cashless exercise of warrants | 98,902 | 99 | 1,345,397 | 1,345,496 | ||||||||||||||||||||||||
| Foreign currency translation | (40,816 | ) | (40,816 | ) | ||||||||||||||||||||||||
| Net income | 1,096,247 | 1,096,247 | ||||||||||||||||||||||||||
| Appropriation to statutory reserves | - | - | - | |||||||||||||||||||||||||
| BALANCE, March 31, 2010 (unaudited) | - | $ | - | 7,097,708 | $ | 7,097 | $ | 35,966,731 | $ | 3,879,077 | $ | 13,671,153 | $ | 2,830,705 | $ | 56,354,763 | ||||||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-32
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE THREE MONTHS ENDED MARCH 31, 2010 AND 2009
(UNAUDITED)
| Three months ended March 31, | ||||||||
| 2010 | 2009 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net income | $ | 1,096,247 | $ | 1,064,644 | ||||
| Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||
| Depreciation | 123,484 | 112,930 | ||||||
| Amortization | 91,370 | 38,519 | ||||||
| Common stock issued for services | 16,245 | 15,052 | ||||||
| Common stock to be issued to related parties for compensation | 27,025 | - | ||||||
| Bad debt expense | - | 21,743 | ||||||
| Change in fair value of warrant liability | 317,380 | (38,328 | ) | |||||
| Change in operating assets and liabilities | ||||||||
| Accounts receivable | 935,790 | (994,017 | ) | |||||
| Inventories | (921,558 | ) | (2,888,742 | ) | ||||
| Deposits and prepaid expenses | (272,640 | ) | 2,551,310 | |||||
| Other receivables | (81,901 | ) | (9,823 | ) | ||||
| Accounts payable | 123,423 | (166,085 | ) | |||||
| Accrued expenses | (223,979 | ) | (382,703 | ) | ||||
| Deposits from customers | 573,897 | (131,448 | ) | |||||
| Taxes payable | 102,941 | 900,427 | ||||||
| Other payables | 1,233 | 39,916 | ||||||
| Net cash provided by operating activities | 1,908,957 | 133,395 | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Payments of long-term prepayments | - | (32,233 | ) | |||||
| Prepayment for potential acquisition | (5,499,375 | ) | - | |||||
| Loans to third parties | - | (366,275 | ) | |||||
| Purchases of plant and equipment | (1,451,016 | ) | (73,255 | ) | ||||
| Payments on construction-in-progress | (404,990 | ) | (242,507 | ) | ||||
| Net cash used in investing activities | (7,355,381 | ) | (714,270 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Decrease (increase) in restricted cash | (200 | ) | ||||||
| Proceeds from short-term loans | - | 219,765 | ||||||
| Repayment for short-term loans | (219,975 | ) | - | |||||
| Repayment to shareholders and directors | - | (175,812 | ) | |||||
| Proceeds from shareholders and directors | - | 83,124 | ||||||
| Due (from) to related parties | (78,269 | ) | - | |||||
| Net cash provided by (used in) financing activities | (298,244 | ) | 126,877 | |||||
| EFFECT OF EXCHANGE RATE CHANGES ON CASH | (43,315 | ) | 81,264 | |||||
| INCREASE (DECREASE) IN CASH | (5,787,983 | ) | (372,734 | ) | ||||
| CASH, beginning | 11,699,398 | 576,409 | ||||||
| CASH, ending | $ | 5,911,415 | $ | 203,675 | ||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | ||||||||
| Cash paid for interest | $ | 5,210 | $ | 19,079 | ||||
| Cash paid for income taxes | $ | - | $ | - | ||||
| Non-cash investing and financing activities | ||||||||
| Long-term prepayment transferred to construction-in-progress | $ | - | $ | 309,869 | ||||
| Long-term prepayment transferred to property, plant and equipment | 439,777 | - | ||||||
| Construction-in-progress transferred to property, plant and equipment | 52,463 | - | ||||||
| Cashless exercise of warrants | $ | 1,345,496 | - | |||||
The accompanying notes are an integral part of the consolidated financial statements.
F-33
SKYSTAR BIO-PHARMACEUTICAL COMPANY AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
March 31, 2010
(Unaudited)
Note 1 - ORGANIZATION
Organization and description of business
Skystar Bio-Pharmaceutical Company (“Skystar” or the “Company”), was incorporated in Nevada on September 24, 1998. Since its acquisition on November 7, 2005 of Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”), a Cayman Islands company, the Company has been engaged in research, development, production, marketing and sales of veterinary healthcare and medical care products. All current operations of the Company are in the People’s Republic of China (“China” or the “PRC”).
All of the Company’s operations are carried out by Xian Tianxing Bio-Pharmaceutical Co., Limited (“Xian Tianxing”), a PRC joint stock company that the Company controls through contractual arrangements originally between Skystar Cayman and Xian Tianxing. On March 10, 2008, the Company entered into a series of agreements transferring all of the rights and obligations of Skystar Cayman under the contractual arrangements to Sida Biotechnology (Xian) Co., Ltd. (“Sida”), a PRC company. Sida is the wholly owned subsidiary of Fortunate Time International Limited (“Fortunate Time”), a Hong Kong company and wholly owned subsidiary of Skystar Cayman. Xian Tianxing also has a wholly owned subsidiary, Shanghai Siqiang Biotechnological Co., Ltd. (“Shanghai Siqiang”), a PRC company.
As a result of these contractual arrangements, which obligates Sida to absorb all of the risk of loss from Xian Tianxing’s activities and enable Sida to receive all of its expected residual returns, the Company accounts for Xian Tianxing as a variable interest entity (“VIE”) under Financial Accounting Standards Board’s (“FASB”) interpretation on consolidation of variable interest entities. Accordingly, the Company consolidates Xian Tianxing’s results, assets and liabilities.
Sida was established by Fortunate Time on July 10, 2007, with registered capital of $5,000,000. Fortunate Time invested $2,000,000 into Sida on July 20, 2007, which amount is payable to Skystar Cayman. On July 9, 2009, Fortunate Time invested the remaining $3,000,000 into Sida. Xi’an High Technology District approved Sida’s application to increase its registered capital to $15,000,000 on July 13, 2009. On July 15, 2009, Sida received the $10,000,000 additional registered capital from Fortunate Time. Funds from Fortunate Time for $13,000,000 was from the cash proceeds of the equity offering which is further discussed in Note 13.
On September 18, 2009, Skystar Bio-Pharmaceutical Inc. (“Skystar California”) was incorporated in California and became a wholly-owned subsidiary of Skystar.
Hereinafter, Skystar, Skystar California, Skystar Cayman, Fortunate Time, Sida, Xian Tianxing and Shanghai Siqiang are sometimes collectively referred to as the “Company.”
Note 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principles of consolidation
The accompanying consolidated financial statements are prepared in accordance with accounting principles generally accepted in the United States of America. The consolidated financial statements include the financial statements of the Company, its wholly-owned subsidiaries, and its VIEs. All significant inter-company transactions and balances between the Company, its subsidiaries and VIEs have been eliminated in consolidation.
Certain information and footnote disclosures normally present in annual consolidated financial statements prepared in accordance with accounting principles generally accepted in the United States of America have been omitted pursuant to such rules and regulations. These consolidated financial statements should be read in conjunction with the audited consolidated financial statements and footnotes included in the Company’s Annual Report on Form 10-K. The results for the three months ended March 31, 2010, are not necessarily indicative of the results to be expected for the full year ending December 31, 2010.
Use of estimates
The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. For example, the Company estimates its allowance for doubtful accounts and useful lives of plant and equipment. Because of the use of estimates inherent in the financial reporting process, actual results could materially differ from those estimates upon which the carrying values were based.
F-34
Foreign currency translation
The Company uses the United States dollar (“U.S. dollar”) for financial reporting purposes and the Chinese Renminbi (“RMB”) as its functional currency. The Company’s subsidiaries and VIEs maintain their books and records in their functional currency, being the primary currency of the economic environment in which their operations are conducted.
The Company translates the subsidiaries’ and VIEs’ assets and liabilities into U.S. dollars using the applicable exchange rates prevailing at the balance sheet dates, and the statements of operations and cash flows are translated at average exchange rates during the reporting period. As a result, amounts related to assets and liabilities reported on the consolidated statements of cash flows will not necessarily agree with changes in the corresponding balances on the consolidated balance sheets. Equity accounts are translated at historical rates. Adjustments resulting from the translation of the subsidiaries’ and VIEs’ financial statements are recorded as accumulated other comprehensive income.
The quotation of the exchange rates does not imply free convertibility of RMB to other foreign currencies. All foreign exchange transactions continue to take place either through the People's Bank of China or other banks authorized to buy and sell foreign currencies at the exchange rate quoted by the People's Bank of China. The rates of exchange quoted by the People’s Bank of China on March 31, 2010 and December 31, 2009 were US $1.00 to RMB 6.82. The average translation rates of US $1.00 to RMB 6.82 and RMB 6.83 was applied to the income statement accounts for the three months ended March 31, 2010 and 2009, respectively.
Approval of foreign currency payments by the People’s Bank of China or other institutions requires submitting a payment application form together with invoices, shipping documents and signed contracts. Transaction gains and losses that arise from exchange rate fluctuations on transactions denominated in a currency other than the functional currency are included in the results of operations as incurred.
Fair values of financial instruments
The accounting standards regarding fair value of financial instruments and related fair value measurements defines financial instruments and requires fair value disclosures of those financial instruments. This accounting standard defines fair value, establishes a three-level valuation hierarchy for disclosures of fair value measurement and enhances disclosures requirements for fair value measures. Current assets and current liabilities qualify as financial instruments and management believes their carrying amounts are a reasonable estimate of fair value because of the short period of time between the origination of such instruments and their expected realization and if applicable, their current interest rates are equivalent to interest rates currently available. The three levels of valuation hierarchy are defined as follows:
| • | Level 1 inputs to the valuation methodology are quoted prices (unadjusted) for identical assets or liabilities in active markets. |
| • | Level 2 inputs to the valuation methodology include quoted prices for similar assets and liabilities in active markets, and inputs that are observable for the assets or liability, either directly or indirectly, for substantially the full term of the financial instruments. |
| • | Level 3 inputs to the valuation methodology are unobservable and significant to the fair value measurement. |
Effective January 1, 2009, the Company adopted the provisions of an accounting standard regarding whether an instrument (or embedded feature) is indexed to an entity’s own stock. This accounting standard specifies that a contract that would otherwise meet the definition of a derivative but is both (a) indexed to the Company’s own stock and (b) classified in stockholders’ equity in the statement of financial position would not be considered a derivative financial instrument. It provides a new two-step model to be applied in determining whether a financial instrument or an embedded feature is indexed to an issuer’s own stock and thus able to qualify for the scope exception within the standards.
As a result of the foregoing adoption, 309,100 common stock purchase warrants previously treated as equity instruments pursuant to the derivative liability treatment exemption are no longer afforded equity treatment because the strike price of the warrants is denominated in U.S. dollars, a currency other than the Company’s functional currency, the Chinese RMB. As a result, the warrants are not considered indexed to the Company’s own stock, and as such, all future changes in the fair value of these warrants are recognized currently in earnings until such time as the warrants are exercised or expired.
F-35
As such, effective January 1, 2009, the Company reclassified the fair value of these warrants from equity to liability, as if these warrants were treated as a derivative liability since their issuance in February 2007. On January 1, 2009, the Company reclassified from additional paid-in capital, as a cumulative effect adjustment, $230,877 to beginning retained earnings and $877,631 to warrant liability to recognize the fair value of such warrants. The Company recognized a loss of $317,380 and a gain of $38,328 from the change in fair value of the warrant liability for the three months ended March 31, 2010 and 2009.
These warrants do not trade in an active securities market, and as such, the Company estimates the fair value of these warrants using the Black-Scholes Option Pricing Model (“Black-Scholes Model”) using the following assumptions:
| Warrants – (1) | Warrants – (2) | |||||||||||||||
| March 31, | December31, | March 31, | December31, | |||||||||||||
| 2010 | 2009 | 2010 | 2009 | |||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||
| Stock price | $ | 10.10 | $ | 10.10 | $ | 11.61 | $ | 10.10 | ||||||||
| Exercise price | $ | 6.00 | $ | 6.00 | $ | 5.00 | $ | 5.00 | ||||||||
| Annual dividend yield | — | — | — | — | ||||||||||||
| Expected term (years) | — | .17 | 1.92 | 2.17 | ||||||||||||
| Risk-free interest rate | — | 0.04 | % | 1.02 | % | 1.14 | % | |||||||||
| Expected volatility | — | 34 | % | 186 | % | 178 | % | |||||||||
| (1) | As of December 31, 2009, 145,000 warrants with an exercise price of $6.00 were outstanding. As of March 31, 2010, all of these warrants were exercised on a “cashless” basis. |
| (2) | As of December, 31, 2009, 107,254 warrants with an exercise price of $5.00 were outstanding. As of March 31, 2010, 50,204 of these warrants were outstanding. |
Expected volatility is based on historical volatility. Historical volatility was computed using daily pricing observations for recent periods that correspond to the term of the warrants. The Company believes this method produces an estimate that is representative of future volatility over the expected term of these warrants. The Company has no reason to believe future volatility over the expected remaining life of these warrants is likely to differ materially from historical volatility. The expected life is based on the remaining term of the warrants. The risk-free interest rate is based on U.S. Treasury securities according to the remaining term of the warrants.
As required by the FASB’s accounting standards, financial assets and liabilities are classified in their entirety based on the lowest level of input that is significant to the fair value measurement. Depending on the product and the terms of the transaction, the fair values warrant liability were modeled using a series of techniques, including closed-form analytic formula, such as the Black-Scholes Model, which does not entail material subjectivity because the methodology employed does not necessitate significant judgment, and the pricing inputs are observed from actively quoted markets.
The fair value of the 50,204 warrants outstanding as of March 31, 2010 was determined using the Black-Scholes Model, defined in the FASB’s accounting standard of fair value measurement as level 2 inputs, and recorded the change in earnings. As a result, the warrant liability is carried on the consolidated balance sheets at fair value.
The following table sets forth by level within the fair value hierarchy our financial assets and liabilities that were accounted for at fair value on a recurring basis as of March 31, 2010:
Carrying Value at March 31, 2010 | Fair Value Measurement at March 31, 2010 | |||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||
| Warrant liability (unaudited) | $ | 510,571 | $ | — | $ | 510,571 | $ | — | ||||||||
In addition to assets and liabilities that are recorded at fair value on a recurring basis, the Company is required to record assets and liabilities at fair value on a non-recurring basis. Generally, assets are recorded at fair value on a non-recurring basis as a result of impairment charges. For the three months ended March 31, 2010, there were no impairment charges.
Revenue recognition
Revenue of the Company is primarily from the sales of veterinary healthcare and medical care products in China. Sales are recognized when the following four revenue criteria are met: persuasive evidence of an arrangement exists, delivery has occurred, the selling price is fixed or determinable, and collectability is reasonably assured. Sales are presented net of value added tax (“VAT”). No estimated allowance for sales returns is reflected on these consolidated financial statements as sales returns are de minimis based on historical experience.
F-36
There are two types of sales upon which revenue is recognized:
| a. | Credit sales: revenue is recognized when the products have been delivered to the customers. |
| b. | Full payment before delivering: revenue is recognized when the products have been delivered to customers. |
Shipping and handling costs related to goods sold are included in selling expenses, which totaled $71,771 and $91,007 for the three months ended March 31, 2010 and 2009, respectively.
The Company’s revenues and cost of revenues by product line were as follows:
| Three Months Ended March 31, | ||||||||
| 2010 | 2009 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Revenues | ||||||||
| Micro-organism | $ | 1,206,578 | $ | 893,293 | ||||
| Veterinary Medications | 3,196,259 | 2,602,496 | ||||||
| Feed Additives | 236,040 | 182,311 | ||||||
| Vaccines | 230,366 | 145,466 | ||||||
| Total Revenues | $ | 4,869,243 | $ | 3,823,566 | ||||
| Cost of Revenues | ||||||||
| Micro-organism | $ | 343,018 | $ | 260,537 | ||||
| Veterinary Medications | 1,822,258 | 1,592,809 | ||||||
| Feed Additives | 100,046 | 77,439 | ||||||
| Vaccines | 25,897 | 15,573 | ||||||
| Total Cost of Revenues | 2,291,219 | 1,946,538 | ||||||
| Gross Profit | $ | 2,578,024 | $ | 1,877,208 | ||||
Cash
Cash includes cash on hand, demand deposits with banks and liquid investments with an original maturity of three months or less.
Restricted cash
Restricted cash is comprised of amounts received from the PRC government as subsidies and set aside for specific uses (see Note 12). Restricted cash is maintained as bank deposits and reflected as current assets based on the expected period when such funds will be put into their specific uses.
Accounts receivable and other receivables
Accounts receivable are recorded at net realizable value consisting of the carrying amount less an allowance for uncollectible accounts, as needed. The Company uses the aging method to estimate the allowance for anticipated uncollectible receivable balances. Under the aging method, bad debt percentages determined by management based on historical experience, as well as current economic climate, are applied to customers’ balances categorized by the number of months the underlying invoices have remained outstanding. At each reporting period, the allowance balance is adjusted to reflect the amount computed as a result of the aging method. When facts subsequently become available to indicate that the allowance provided requires an adjustment, a corresponding adjustment is made to the allowance account as a change in estimate. The ultimate collection of the Company’s accounts receivable may take one year. Delinquent account balances are reserved after management determines that the likelihood of collection is not probable, and known bad debts are written-off against allowance for doubtful accounts when identified.
Inventories
Inventories are stated at the lower of cost or market, as determined on a moving weighted-average basis. Inventories include purchases and related costs incurred in bringing the inventories to their present location and condition. Management reviews inventories for obsolescence and cost in excess of net realizable value at least annually and records a reserve against the inventory and additional cost of goods sold when the carrying value exceeds net realizable value.
F-37
Plant and equipment
Plant and equipment are stated at cost less accumulated depreciation. Depreciation is computed using the straight-line method over the estimated useful lives of the assets. Expenditures for maintenance and repairs which do not improve or extend the useful lives of the assets are charged to operations as incurred, while renewals and betterments are capitalized. Gains and losses on disposals are included in the results of operations. Estimated useful lives of the assets are as follows:
| Estimated Useful Life | ||
| Buildings | 20-40 years | |
| Machinery and equipment | 10 years | |
| Computer, office equipment and furniture | 5 years | |
| Vehicles | 5-10 years | |
Management assesses the carrying value of plant and equipment annually, more often when factors indicating impairment are present, and reduces the carrying value of such assets by the amount of the impairment. The Company determines the existence of such impairment by measuring the expected future cash flows (undiscounted and without interest charges) and comparing such amount to the net asset carrying value. An impairment loss, if it exists, is measured as the amount by which the carrying amount of the asset exceeds the fair value of the asset. Based on its review, management believes that, as of March 31, 2010 and December 31, 2009, there was no impairment for its plant and equipment.
Construction-in-progress
Construction-in-progress includes direct costs of construction of a factory building. Interest incurred during the period of construction, if significant, is capitalized. All other interest is expensed as incurred. Construction-in-progress is not depreciated until such time the assets are completed and put into service.
Intangible assets
Land Use Rights — Land use rights represent the amounts paid to acquire a long-term interest to utilize the land underlying the Company’s facilities. This type of arrangement is common for the use of land in the PRC. Land use rights are amortized on a straight-line basis over its 50-year term.
Technological Know-How — Purchased technological know-how includes confidential formulas, manufacturing processes, technical and procedural manuals, and is amortized using the straight-line method over the weighted average useful life of nine years, which reflects the period over which such confidential formulas, manufacturing processes, and technical and procedural manuals are kept confidential by the Company as agreed between the Company and the selling parties.
Impairment of Intangible Assets — The Company evaluates the carrying value of intangible assets annually, or more often when factors indicating impairment are present. The Company determines the existence of such impairment by measuring the estimated future cash flows (undiscounted) and comparing such amount to the net asset carrying value. If the undiscounted cash flow estimated to be generated by any such intangible asset is less than its carrying amount, a loss is recognized based on the amount by which the carrying amount exceeds the intangible asset’s fair market value. Loss on intangible assets to be disposed of is determined in a similar manner, except that fair market values are reduced by the cost of disposal. Based on its review, the Company believes that, as of March 31, 2010, there was no impairment of its intangible assets.
Comprehensive income
The FASB’s accounting standard of reporting comprehensive income requires disclosure of all components of comprehensive income and loss on an annual and interim basis. Comprehensive income and loss is defined as the change in equity of a business enterprise during a period from transactions and other events and circumstances from non-owner sources. The accompanying consolidated financial statements include the provisions of GAAP. Accumulated other comprehensive income is comprised of the changes in foreign currency exchange rates.
Research and development costs
Research and development costs are charged to operations as incurred and include salaries, professional fees and technical support fees related to such efforts.
F-38
Advertising costs
Advertising costs are charged to operations currently. Advertising costs for the three months ended March 31, 2010 and 2009 were $1,651 and $38,936 respectively.
Income taxes
The Company accounts for income taxes in accordance with the FASB’s accounting standard for income taxes. Under the asset and liability method as required by this accounting standard, deferred income taxes are recognized for the tax consequences of temporary differences by applying enacted statutory tax rates applicable to future years to differences between the financial statement carrying amounts and the tax bases of existing assets and liabilities. The effect on deferred income taxes of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is recognized if it is more likely than not that some portion, or all of, a deferred tax asset will not be realized.
Further, in accordance with this accounting standard, a tax position is recognized as a benefit only if it is “more likely than not” that the tax position would be sustained in a tax examination, with a tax examination being presumed to occur. The amount recognized is the largest amount of tax benefit that is greater than 50% likely of being realized on examination. For tax positions not meeting the “more likely than not” test, no tax benefit is recorded. The standard also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosures, and transition. Penalties and interest related to underpayment of income taxes are classified as income tax expense in the period incurred. No significant penalties or interest relating to income taxes have been incurred during the three months ended March 31, 2010.
The Company’s operations are subject to income and transaction taxes in the United States and in the PRC jurisdictions. Significant estimates and judgments are required in determining the Company’s worldwide provision for income taxes. Some of these estimates are based on interpretations of existing tax laws or regulations. The ultimate amount of tax liability may be uncertain as a result.
The Company does not anticipate any events which could cause a change to these uncertainties.
Stock-based compensation
The Company records and reports stock-based compensation by measuring the cost of services received in exchange for an award of equity instruments based on the grant-date fair value of the award. That cost is recognized over the period during which services are received. Stock compensation for stock granted to non-employees is determined as the fair value of the consideration received or the fair value of equity instruments issued, whichever is more reliably measured.
The Company uses the Black-Scholes Model which was developed for use in estimating the fair value of options. This models requires the input of highly complex and subjective variables including the expected life of options granted and the Company’s expected stock price volatility over a period equal to or greater than the expected life of the options. Because changes in the subjective assumptions can materially affect the estimated value of the Company’s employee stock options, it is management’s opinion that the Black-Scholes Model may not provide an accurate measure of the fair value of the Company’s employee stock options. Although the fair value of employee stock options is determined in accordance with the standards using an option-pricing model, that value may not be indicative of the fair value observed in a willing buyer/willing seller market transaction.
Earnings per share
The Company reports earnings per share and present both basic and diluted earnings per share in conjunction with the disclosure of the methodology used in computing such earnings per share. Basic earnings per share is based upon the weighted-average number of common shares outstanding. Diluted earnings per share is based on the assumption that all dilutive convertible shares, including convertible preferred shares, and stock options were converted or exercised. Further, the method requires that stock dividends or stock splits be accounted for retroactively if the stock dividends or stock splits occur during the period, or retroactively if the stock dividends or stock splits occur after the end of the period but before the release of the financial statements, by considering it outstanding of the entirety of each period presented. Dilution is computed by applying the treasury stock method. Under this method, options and warrants are assumed to be exercised at the beginning of the period (or at the time of issuance, if later), and as if funds obtained thereby were used to purchase common stock at the average market price during the period.
Related parties
Parties are considered to be related to the Company if the parties, directly or indirectly, through one or more intermediaries, control, are controlled by, or are under common control with the Company. Related parties also include principal owners of the Company, its management, members of the immediate families of such principal owners and management, and other parties with which the Company may deal if one party controls or can significantly influence the management or operating policies of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests.
F-39
Reclassifications
Certain prior period amounts have been reclassified to conform to the current period presentation. These reclassifications had no impact on previously reported net income or cash flows.
Recently issued accounting pronouncements
In October 2009, the FASB issued an ASU regarding accounting for own-share lending arrangements in contemplation of convertible debt issuance or other financing. This ASU requires that at the date of issuance of the shares in a share-lending arrangement entered into in contemplation of a convertible debt offering or other financing, the shares issued shall be measured at fair value and be recognized as an issuance cost, with an offset to additional paid-in capital. Further, loaned shares are excluded from basic and diluted earnings per share unless default of the share-lending arrangement occurs, at which time the loaned shares would be included in the basic and diluted earnings-per-share calculation. This ASU is effective for fiscal years beginning on or after December 15, 2009, and interim periods within those fiscal years for arrangements outstanding as of the beginning of those fiscal years. The adoption of this ASU did not have a material impact on the Company’s consolidated financial statements.
In January 2010, the FASB issued ASU No. 2010-02 regarding accounting and reporting for decreases in ownership of a subsidiary. Under this guidance, an entity is required to deconsolidate a subsidiary when the entity ceases to have a controlling financial interest in the subsidiary. Upon deconsolidation of a subsidiary, an entity recognizes a gain or loss on the transaction and measures any retained investment in the subsidiary at fair value. In contrast, an entity is required to account for a decrease in its ownership interest of a subsidiary that does not result in a change of control of the subsidiary as an equity transaction. This ASU clarifies the scope of the decrease in ownership provisions, and expands the disclosures about the deconsolidation of a subsidiary or de-recognition of a group of assets. This ASU is effective beginning in the first interim or annual reporting period ending on or after December 31, 2009. The adoption of this ASU did not have a material impact the Company’s consolidated financial statements.
In January 2010, FASB issued ASU No. 2010-06 – Improving Disclosures about Fair Value Measurements. This update provides amendments to Subtopic 820-10 that requires new disclosure to include transfers in and out of Levels 1 and 2 and activity in Level 3 fair value measurements. Further, this update clarifies existing disclosures on level of disaggregation and Disclosures about inputs and valuation techniques. A reporting entity should provide fair value measurement disclosures for each class of assets and liabilities and should provide disclosures about the valuation techniques and inputs used to measure fair value for both recurring and nonrecurring fair value measurements. Those disclosures are required for fair value measurements that fall in either Level 2 or Level 3. The new disclosures and clarifications of existing disclosures are effective for interim and annual reporting periods beginning after December 15, 2009, except for the disclosures about purchases, sales, issuances, and settlements in the roll forward of activity in Level 3 fair value measurements. Those disclosures are effective for fiscal years beginning after December 15, 2010, and for interim periods within those fiscal years. The Company is currently evaluating the impact of this ASU; however, the Company does not expect the adoption of this ASU to have a material impact on its consolidated financial statements.
In February 2010, the FASB issued ASU 2010-09, “Subsequent Events (Topic 855): Amendments to Certain Recognition and Disclosure Requirements.” ASU 2010-09 primarily rescinds the requirement that, for listed companies, financial statements clearly disclose the date through which subsequent events have been evaluated. Subsequent events must still be evaluated through the date of financial statement issuance; however, the disclosure requirement has been removed to avoid conflicts with other SEC guidelines. ASU 2010-09 was effective immediately upon issuance and was adopted in February 2010.
Note 3 - CONCENTRATIONS AND CREDIT RISK
The Company’s operations are all carried out in the PRC. Accordingly, the Company’s business, financial condition and results of operations may be influenced by the political, economic and legal environments in the PRC, and by the general state of the PRC’s economy. The Company’s operations in the PRC are subject to specific considerations and significant risks not typically associated with companies in North America and Western Europe. These include risks associated with, among others, the political, economic and legal environments and foreign currency exchange. The Company’s results may be adversely affected by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversion and remittance abroad, and rates and methods of taxation, among other things.
Financial instruments, which subject the Company to concentration of credit risk, consist of cash. The Company maintains balances at financial institutions which, from time to time, may exceed federally insured limits for the banks located in the United States. Balances at financial institutions or state-owned banks within the PRC are not covered by insurance. As of March 31, 2010 and December 31, 2009, the Company had deposits in excess of federally insured limits (including restricted cash) of $2,436,664 and $11,504,970 respectively. The Company has not experienced any losses in such accounts.
F-40
For the three months ended March 31, 2010 and 2009, all of the Company’s sales occurred in the PRC. No major customers accounted for more than 10% of the Company’s total revenues. In addition, all accounts receivable at March 31, 2010 and December 31, 2009 also arose in the PRC.
The Company’s two largest vendors accounted for approximately 32% and 26% of the Company’s total purchases, respectively, for the three months ended March 31, 2010 and 2009.
The Company had one product that accounted for 17% of the Company’s total revenues for the three months ended March 31, 2010, while no products accounted for more than 10% of the Company’s total revenues for the three months ended March 31, 2009.
Note 4 - - ACCOUNTS RECEIVABLE, NET
Accounts receivable consisted of the following:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Account receivable | $ | 3,774,935 | $ | 4,711,044 | ||||
| Allowance for bad debts | (327,857) | (327,857) | ||||||
| Account receivable, net | $ | 3,447,078 | $ | 4,383,187 | ||||
The following table presents the movement of allowance for doubtful accounts:
| Allowance for bad debt, January 1, 2009 | $ | 327,857 | ||
| Addition | — | |||
| Recovery | — | |||
| Translation adjustment | — | |||
| Allowance for bad debt, December 31, 2009 | 327,857 | |||
| Addition | — | |||
| Recovery | — | |||
| Translation adjustment | — | |||
| Allowance for bad debt, March 31, 2010 (unaudited) | $ | 327,857 |
Note 5 – INVENTORIES
Inventories consist of the following:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Raw materials | $ | 3,527,883 | $ | 2,997,481 | ||||
| Packing materials | 335,904 | 159,620 | ||||||
| Work-in-process | 4,676 | 886 | ||||||
| Finished goods | 1,307,941 | 1,096,547 | ||||||
| Other | 19,571 | 19,571 | ||||||
| Total | 5,195,975 | 4,274,105 | ||||||
| Less: Allowance for slow moving raw materials | (199,460 | ) | (199,460 | ) | ||||
| Total | $ | 4,996,515 | $ | 4,074,645 | ||||
The Company periodically reviews its reserves for slow-moving and obsolete inventories. As of March 31, 2010 and December 31, 2009, the Company recorded a slow-moving allowance for raw materials of $199,460.
Note 6 - DEPOSITS AND PREPAID EXPENSES
Deposits and prepaid expenses are comprised of the following:
F-41
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Prepayment for raw materials purchasing | $ | 11,402,626 | $ | 10,990,913 | ||||
| Prepayment for packaging materials purchasing | 511,073 | 489,392 | ||||||
| Other | 259,348 | 420,009 | ||||||
| Total | $ | 12,173,047 | $ | 11,900,314 | ||||
Note 7 - PLANT AND EQUIPMENT, NET
Plant and equipment consist of the following:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Building and improvements | $ | 8,469,768 | $ | 6,798,616 | ||||
| Machinery and equipment | 3,070,294 | 3,035,814 | ||||||
| Office equipment and furniture | 195,269 | 191,424 | ||||||
| Vehicles | 485,156 | 485,156 | ||||||
| Total | 12,220,487 | 10,511,010 | ||||||
| Less: accumulated depreciation | (1,805,479 | ) | (1,681,952 | ) | ||||
| Plant and equipment, net | $ | 10,415,008 | $ | 8,829,058 | ||||
As of December 31, 2009, the Company made deposits of $439,927 on a new office building. In January 2010, the purchase was completed; the total purchase cost of $1,600,346 was transferred to fixed asset as of March 31, 2010.
Depreciation expense was $123,484 and $112,930 for the three months ended March 31, 2010 and 2009, respectively.
Note 8 - CONSTRUCTION-IN-PROGRESS
Construction-in-progress (“CIP”) is related to a plant being built in accordance with the PRC’s Good Manufacturing Practices (“GMP”) Standard. Construction on this plant commenced in 2005. The veterinary medicine facility and the building that houses quality control, research and development and administration were completed during 2007, and the remaining plant facilities are expected to be completed by June 30, 2010, at an estimated cost of $11,997,268. During the three months ended March 31, 2010, some general facility improvements were completed and placed in service resulting in a transfer from CIP to property, plant and equipment of $72,249. No depreciation is provided for construction-in-progress until such time the assets are completed and placed into service.
The construction projects the Company is in the progress of completing are:
| Project | Total in CIP as of 3/31/2010 | Estimate Cost to Complete | Estimated Total Cost | Estimated Completion Date | ||||||||||
| Vaccine facility | 8,247,153 | 2,099,326 | 10,346,479 | June 2010(1) | ||||||||||
| Micro-organism facility | 1,343,825 | 156,175 | 1,500,000 | June 2010(2) | ||||||||||
| Other | 150,789 | - | 150,789 | |||||||||||
| TOTAL CIP Balance | 9,741,767 | 2,255,501 | 11,997,268 | |||||||||||
| (1) | Note that this date does not include the time to certify the facility. |
| (2) | The construction of the micro-organism facility was completed in December 2009. Since then the facility has been going through testing and quality assurance, with expected production to commence in June 2010. |
As of March 31, 2010 and December 31, 2009, the Company had construction in progress amounting to $9,741,767 and $9,389,120, respectively. No interest expense had been capitalized for construction in progress for the three months ended March 31, 2010 and 2009, respectively.
F-42
Note 9 - LONG-TERM PREPAYMENTS
Long-term prepayments consist of the following:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Construction deposit | $ | 733,500 | $ | 733,500 | ||||
| Deposit for building and equipment purchase | 234,441 | 439,927 | ||||||
| Deposit for potential acquisitions | 12,308,130 | 6,806,880 | ||||||
| Total | $ | 13,276,071 | $ | 7,980,307 | ||||
As of March 31, 2010 and December 31, 2009, the Company has determined that these prepayments are noncurrent because: (1) these amounts relate to noncurrent assets, and (2) the Company’s ability to complete any potential acquisitions is contingent upon the Company successfully negotiating with target companies, the ultimate date of which cannot be readily determined. Deposits are refundable if negotiation is not successful.
As of December 31, 2009, deposits for office building purchase of $439,927 (RMB 2,998,820) represented deposits made for new office building. The purchase was completed and the deposit was transferred to fixed asset as of March 31, 2010. The Company is in the process of applying for property certificate and expects to get the certificate by September 2010.
As of March 31, 2010, deposits for building and equipment purchase of $234,441 represented deposits made for equipments.
As of March 31, 2010 and December 31, 2009, deposits for potential acquisitions of $12,308,130 and $6,806,880 represented deposits made to potential acquisition targets. The potential acquisitions are contingent upon the Company successfully negotiating with target companies, the ultimate date of which cannot be readily determined. Deposits are refundable if negotiation is not successful.
Note 10 – INTANGIBLE ASSETS
Intangible assets consisted of the following:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Land use rights | $ | 378,853 | $ | 378,853 | ||||
| Technological know-how | 2,053,800 | 2,053,800 | ||||||
| Total | 2,432,653 | 2,432,653 | ||||||
| Less: accumulated amortization | (663,882 | ) | (572,481 | ) | ||||
| Intangible assets, net | $ | 1,768,771 | $ | 1,860,172 | ||||
In 2009, the Company paid $1,172,880 (RMB 8,000,000) for fish disease vaccine technology transfer for an eleven-year term from September 2009 through September 2020.
For the three months ended March 31, 2010 and 2009, the amortization expense for intangibles amounted to $91,370 and $38,519, respectively.
Amortization expense for the future five years and thereafter is as follows:
| Years ending December 31, | Amount | |||
| (Unaudited) | ||||
| 2010 | $ | 195,726 | ||
| 2011 | 260,968 | |||
| 2012 | 236,518 | |||
| 2013 | 114,268 | |||
| 2014 | 114,268 | |||
| Thereafter | 847,023 | |||
| Total | $ | 1,768,771 | ||
F-43
Note 11 – SHORT-TERM LOANS
On January 14, 2009, the Company signed a one year short-term loan contract with Shaanxi Agricultural Yanta Credit Union for $220,050 (RMB 1.5 million) at an annual interest rate of 8.66% for operating purposes secured by the personal property of Weibing Lu, the Company’s Chief Executive Officer. This amount was paid off on January 15, 2010.
Interest expense incurred and associated with the short-term loans amounted to $5,210 for the three months ended March 31, 2010, none of which has been capitalized as part of construction-in-progress in 2010. Interest expense incurred and associated with the short term loan amounted to $4,035 for the three months ended March 31, 2009, none of which has been capitalized as part of construction-in-progress.
Note 12 - DEFERRED GOVERNMENT GRANT
Deferred government grant represents subsidies for GMP projects granted by the PRC government. To date, the Company received government subsidies totaling $1,100,250.
According to PRC government regulations, the funds granted may be treated as capital contributed by a company appointed by the PRC government or as a loan from such company, which the Company will be required to repay. As of March 31, 2010, the Company has not reached a final agreement with the PRC government regarding the treatment of these three subsidies as either a loan or capital contribution, and the Company does not expect that the final agreement will be completed within the current year. Therefore, these amounts are reflected as non-current liabilities in the accompanying consolidated financial statements.
Note 13 - CAPITAL TRANSACTIONS
On May 12, 2009, the Company effectuated a 1-for-10 reverse stock split of its issued and outstanding shares of common stock and a proportional reduction of its authorized shares of common stock. Number of common stock, warrants, options disclosed in the footnotes has been retroactively restated to reflect the 1-for-10 reverse stock split. On November 16, 2009, the Company effectuated a 2-for-1 forward split of its issued and outstanding common stock and a proportional increase of its authorized shares of common stock. Number of common stock, warrants, options disclosed in the footnotes has been retroactively restated to reflect the 2-for-1 forward stock split.
Preferred stock
On June 25, 2009, the Company’s board of directors concluded that 2,000,000 shares of series “A” preferred stock issued in 2001 were not valid because no certificate of designation was filed prior to their issuance as required under Nevada corporate law. On December 21, 2009, the Company instructed its transfer agent to remove these preferred shares officially from its shareholder records. As of March 31, 2010 and December 31, 2009, no share of series “A” preferred stock is authorized or outstanding.
Stock-based compensation
On March 30, 2010, the Company agreed to issue 2,500 shares of common stock to a non-executive director in exchange for services unrelated to directorship. The trading value of the Company’s common stock on March 30, 2010 was $10.61 per share and a corresponding amount of $27,000 was charged to general and administrative expenses. The Company also had a $329,397 and $302,372 balance under shares to be issued as of March 31, 2010 and December 31, 2009, which represented 49,834 and 47,334 common shares to be issued to the non-executive director for his service provided for the period from May 2008 to March 2010, and for the period from May 2008 to December 2009, respectively. The amounts were included in general and administrative expenses based on the weighted-average trading price of the Company’s common stock for the said period. As of March 31, 2010, these shares have not been issued.
On May 5, 2008, the Company agreed to issue 10,434 shares of common stock to its former chief financial officer (“CFO”) during the term of a one-year agreement, which would vest in four equal installments of 2,608 shares each quarter. The trading value of the common stock on May 5, 2008 was $5.85 per share for a total value of $61,042. These shares were fully vested. On May 26, 2009, the Company renewed the one-year service agreement with the CFO and agreed to issue 14,440 shares of common stock, which would vest in four equal installments of 3,610 shares every quarter starting August 5, 2009. Compensation expense is recognized on a straight-line basis over the vesting period. Total compensation expense of $16,245 and $15,052 were charged to general and administrative expenses for the three months ended March 31, 2010 and 2009. In February 2010, 10,830 shares were issued. Effective April 16, 2010, the CFO resigned from his position and the last installment of 3,610 shares was prorated to 2,880 shares, which shares have not been issued.
On May 26, 2009, the Company agreed to issue 5,556 shares of common stock to a director at the beginning of each term of his directorship. The trading value of the common stock on May 26, 2009 was $4.50 per share for the total value of $25,002, and the amount was charged to general and administrative expenses for the year ended December 31, 2009. As of December 31, 2009, the balance included shares to be issued. As of March 31, 2010, 5,556 shares were issued and there was no balance under shares to be issued.
F-44
Warrants
On February 28, 2007, the Company issued 195,000 warrants to four investors with an exercisable price of $6.00 per share for a term of three years. On the same date, the Company also issued warrants to the private placement agent, exercisable for 114,100 shares of the Company’s common stock at a price of $5.00 per share for a five-year term. For the three months ended March 31, 2010, 202,050 warrants were exercised. The Company valued the conversion on exercise date and recorded $248,741of losses from changes in fair value of warrants. Following is a summary of the status of warrants outstanding at March 31, 2010:
| Average Remaining | ||||||
| Number of Warrants | Contractual Life | Average Exercise Price | ||||
| 50,204 | 1.92 years | $ | 5.00 | |||
Following is an activity summary of the Company’s outstanding warrants:
| Outstanding as of December 31, 2008 | 309,100 | |||
| Granted | — | |||
| Forfeited | — | |||
| Exercised | — | |||
| Outstanding as of March 31, 2009 (unaudited) | 309,100 | |||
| Granted | — | |||
| Forfeited | — | |||
| Exercised | 56,846 | |||
| Outstanding as of December 31, 2009 | 252,254 | |||
| Granted | — | |||
| Forfeited | — | |||
| Exercised | 202,050 | |||
| Outstanding as of March 31, 2010 (unaudited) | 50,204 |
Equity offering
On June 30, 2009, the Company and Rodman & Renshaw, LLC, as representative of underwriters (the "Underwriters") entered into an Underwriting Agreement. Pursuant to the Underwriting Agreement, the Company agreed to issue and sell an aggregate of 3,220,000 shares (including 420,000 over-allotment shares) of its common stock, at a price of $6.49 per share, in a public offering. The closing date of this offering was on the third business day following the effective date of the registration statement registering the shares offered, or July 3, 2009.
In connection with this offering, the Company agreed to grant 140,000 common stock purchase options to five designees of the Underwriters. The options are exercisable from June 30, 2010 to June 30, 2014, and each option is exercisable for one share of the Company’s common stock, with exercise price at $8.11 per share. The Company used the Black-Scholes Model to value the options granted, which amounted to $1,065,842. The value of options granted to these individuals was included as part of the offering costs, and had no net effect on the Company’s equity.
The following are the assumptions used by the Company in the Black-Scholes Model:
Number of options | Stock price | Exercise price | Expected term | Dividend yield | Volatility | Risk-free interest rate | ||||||||||||||||||
| 140,000 | $ | 8.97 | $ | 8.11 | 3.0 years | — | 161 | % | 1.67 | % | ||||||||||||||
The following is a summary of the status of options outstanding:
| Outstanding Options | Exercisable Options | ||||||||||||||||||||
Average Remaining | Average Remaining | ||||||||||||||||||||
Number of Options | Contractual Life | Average Exercise Price | Number of Options | Contractual Life | Average Exercise Price | ||||||||||||||||
| 140,000 | 4.75 | $ | 8.11 | — | — | $ | — | ||||||||||||||
F-45
Following is an activity summary of the Company’s outstanding options:
Number of Options Outstanding | Weighted Average Exercise Price | Aggregate Intrinsic Value | ||||||||||
| Outstanding as of December 31, 2008 | — | — | — | |||||||||
| Granted | 140,000 | $ | 8.11 | — | ||||||||
| Forfeited | — | — | — | |||||||||
| Exercised | — | — | — | |||||||||
| Outstanding as of December 31, 2009 | 140,000 | $ | 8.11 | $ | — | |||||||
| Forfeited | — | — | — | |||||||||
| Exercised | — | — | — | |||||||||
| Outstanding as of March 31, 2010 (unaudited) | 140,000 | $ | 8.11 | $ | — | |||||||
Incremental costs incurred of this offering, including underwriting commission, legal fees, and printing costs were $1,730,477, and were directly deducted from the proceeds. The gross proceeds of this offering were $20,897,800. The Company received cash proceeds of $18,411,496 on July 6, 2009.
Equity Compensation Plan
On December 8, 2009, the Company’s board of directors approved a stock incentive plan for officers, directors, employees and consultants entitled the “Skystar Bio-Pharmaceutical Company 2010 Stock Incentive Plan” (the “2010 Plan”). The maximum number of shares that may be issued under the 2010 Plan is 700,000 shares of the Company’s common stock. The 2010 Plan was approved by the Company’s stockholders on December 31, 2009, and awards may be granted thereunder until December 7, 2019. As of March 31, 2010, there are 700,000 shares of the Company’s common stock remaining available for future issuance under the 2010 Plan.
Note 14 - STATUTORY RESERVES
Statutory reserves represent restricted retained earnings. Based on the legal formation of the entities, all PRC entities are required to set aside 10% of its net income as reported in its statutory accounts on an annual basis to the statutory surplus reserve fund. Once the total statutory surplus reserve reaches 50% of the entity’s registered capital, further appropriations are discretionary. The statutory surplus reserve can be used to increase the entity’s registered capital (upon approval by relevant government authorities) and eliminate its future losses under PRC GAAP (upon a resolution by the board of directors). The statutory surplus reserve is not distributable to shareholders except in the event of liquidation. As of March 31, 2010, Xian Tianxing has met the statutory surplus reserve requirement, and approximately $7,534,275 still needs to be transferred to the respective statutory surplus reserve of Shanghai Siqiang and Sida.
Appropriations to the above statutory reserves are accounted for as a transfer from unrestricted earnings to statutory reserves. There are no legal requirements in the PRC to fund these statutory reserves by the transfer of cash to any restricted accounts, and as such, the Company has not transferred any cash to these accounts. These reserves are not distributable as cash dividends.
Note 15 – TAXES
Skystar and Skystar California are subject to the United States federal income tax provision. Skystar Cayman is a tax-exempt company incorporated in the Cayman Islands and conducts all of its business through its subsidiaries, Fortunate Time and Sida, and Sida’s PRC VIEs, Xian Tianxing and Shanghai Siqiang.
Sida, Xian Tianxing, and Shanghai Siqiang are subject to PRC’s Enterprise Income Tax. Pursuant to the PRC Income Tax Laws, Enterprise Income Tax is generally imposed at a statutory rate of 25% beginning on January 1, 2008. Xian Tianxing has been approved as a new technology enterprise, and under PRC Income Tax Laws is entitled to a preferential tax rate of 15%.
For the years ended March 31, 2010 and 2009, the provisions for income tax were as follows:
| 2010 | 2009 | |||||||
| Current PRC income tax expense | ||||||||
| Enterprise income tax | $ | 325,319 | $ | 211,520 | ||||
F-46
The following table reconciles the U.S. statutory rates to the Company's effective tax rate as of March 31:
| 2010 | 2009 | |||||||
| U.S. Statutory rate | 34.0 | % | 34.0 | % | ||||
| Foreign income not recognized in the U.S. | (34.0 | ) | (34.0 | ) | ||||
| China income tax rate | 25.0 | 25.0 | ||||||
| China income tax exemption | (10.0 | ) | (10.0 | ) | ||||
Other item (1) | 7.9 | 1.6 | ||||||
| Total provision for income taxes | 22.9 | % | 16.6 | % | ||||
| (1) | The other item is operating expenses incurred by Skystar that are not deductible in the PRC which resulted in an increase in effective tax rate of 7.9% and 1.6% for the three months ended March 31, 2010 and 2009, respectively. |
Taxes payable consisted of the following:
March 31, 2010 | December 31, 2009 | |||||||
| Income taxes payable | $ | 429,691 | $ | 104,261 | ||||
| Value added tax | 359,730 | 561,646 | ||||||
| Other taxes | 35,661 | 56,199 | ||||||
| Total | $ | 825,082 | $ | 722,106 | ||||
The estimated tax savings due to the reduced tax rate for the three months ended March 31, 2010 and 2009 amounted to $216,880 and $144,244, respectively. If the statutory income tax had been applied, the Company would have decreased basic earnings per share and diluted per shares from $0.16 to $0.12 and from $0.15 to $0.12 for the three months ended March 31, 2010, respectively. For the three months ended March 31 2009, the basic and diluted earnings per share would have decreased from $0.29 to $0.24 if the statutory income tax had been applied.
Skystar is incorporated in the U.S. and has incurred a net operating loss for income tax purposes for the three months ended March 31, 2010. As of March 31, 2010, the estimated net operating loss carryforwards for U.S. income tax purposes amounted to $4,943,295 which may be available to reduce future years’ taxable income. These carryforwards will expire, if not utilized, beginning in 2026 and continue through 2010. Management believes that the realization of the benefits arising from this loss appears to be uncertain due to the Company’s limited operating history and continuing losses for U.S. income tax purposes. Accordingly, the Company has provided a 100% valuation allowance at March 31, 2010 and December 31, 2009. The valuation allowance at March 31, 2010 and December 31, 2009 was $1,680,720 and $1,553,677, respectively. The Company’s management reviews this valuation allowance periodically and makes adjustments as necessary.
The Company has cumulative undistributed earnings of foreign subsidiaries of approximately $27,303,297 as of March 31, 2010, which are included in consolidated retained earnings and will continue to be indefinitely reinvested in international operations. Accordingly, no provision has been made for U.S. deferred taxes related to future repatriation of these earnings, nor is it practicable to estimate the amount of income taxes that would have to be provided if we concluded that such earnings will be remitted in the future.
Note 16 - EARNINGS PER SHARE
The following is the calculation of earnings per share:
For the three months ended March 31, | ||||||||
2010 (Unaudited) | 2009 (Unaudited) | |||||||
| Net income | $ | 1,096,247 | $ | 1,064,644 | ||||
| Weighted average shares used in basic computation | 7,061,530 | 3,734,602 | ||||||
| Diluted effect of stock warrants | 78,610 | - | ||||||
| Weighted average shares used in diluted computation | 7,140,140 | 3,734,602 | ||||||
| Earnings per share: | ||||||||
| Basic | $ | 0.16 | $ | 0.29 | ||||
| Diluted | $ | 0.15 | $ | 0.29 | ||||
F-47
For the three months ended March 31, 2010, the average stock price was greater than the exercise prices of warrants which resulted in additional weighted-average common stock equivalents of 51,699. For the three months ended March 31, 2009, the outstanding warrants were excluded from the diluted earnings per share calculation as they are anti-dilutive as the average stock price was less than the exercise prices of the warrants.
For the three months ended March 31, 2010, the average stock price was greater than the exercise prices of 140,000 outstanding options which resulted in additional weighted-average common stock equivalents of 26,911.
Note 17 - RELATED PARTY TRANSACTIONS AND ARRANGEMENTS
Amounts receivable from and payable to related parties are summarized as follows:
March 31, 2010 | December 31, 2009 | |||||||
| (Unaudited) | ||||||||
| Short-term loans from shareholders | ||||||||
Mr. Weibing Lu – officer and shareholder (1) | $ | 36,675 | $ | 36,675 | ||||
Mr. Wei Wen – officer and shareholder (1) | 36,675 | 36,675 | ||||||
Ms. Aixia Wang – shareholder (1) | 36,675 | 36,675 | ||||||
| Total | $ | 110,025 | $ | 110,025 | ||||
| Shares to be issued to related party | ||||||||
Scott Cramer – non-executive director (2) | $ | 329,397 | $ | 302,372 | ||||
Mark D Chen – non-executive director(2) | - | 25,002 | ||||||
| Total | $ | 329,397 | $ | 327,374 | ||||
| Amounts due (from) to related parties | ||||||||
Scott Cramer – non-executive director and shareholder (3) | 101,056 | 143,556 | ||||||
Shaanxi Xingji Electronics Co. - owned by a director's wife (3) | 5,687 | - | ||||||
Officer and shareholder (3) | - | 41,468 | ||||||
| Total | $ | 106,743 | $ | 185,024 | ||||
(1) On May 29, 2008, Weibing Lu, Wei Wen and Aixia Wang obtained personal loans from Yanta Credit Union and advanced cash to Xian Tianxing in the total amount of $132,030 to facilitate operations. These loans, which were due on May 29, 2009 with 8.436% interest per annum and guaranteed by Xian Tianxing, were paid in full on May 29, 2009. On June 2, 2009, Mr. Lu, Mr. Wen and Ms. Wang again obtained loans from the same bank and advanced cash to Xian Tianxing in the total amount of $110,025. These loans are due on June 1, 2010, with 10.11% interest per annum and are also guaranteed by Xian Tianxing. For the three months ended March 31, 2010 and 2009, Xian Tianxing paid interest of $3,845 and $2,882, respectively, for these loans.
(2) As of March 31, 2010 and December 31, 2009, the Company had $329,397 (representing 49,834 common shares) and $302,372 (representing 47,334 common shares), respectively, under agreement to issue shares to Scott Cramer, as compensation for being a representative of the Company in the United States for the periods from May 2008 to March 31, 2010, and December 31, 2009, respectively. In addition, as of March 31, 2010 and December 31, 2009, the Company had $0 and $25,002 balance (representing 5,556 common shares) under agreement to issue shares to Mark D Chen as compensation at the beginning of each term of his directorship.
(3) Shaanxi Xinji Electronics Co., Ltd. is owned by the wife of Weibing Lu. The amounts due to Shaanxi Xinji Electronics as of March 31, 2010 and December 31, 2009 were short-term cash transfers for business operations, non-interest bearing, unsecured, and payable upon demand. As of March 31, 2010 and December 31, 2009, the Company also had amounts due to Scott Cramer for bonus and the expenses paid by him on behalf of the Company. The Company has paid Bennet Tchaikovsky all of the outstanding compensation and expenses as of March 31, 2010.
F-48
Note 18 - COMMITMENTS AND CONTINGENCIES
(a) Lease commitments
The Company recognizes lease expense on a straight-line basis over the term of the lease in accordance to the FASB’s accounting standard of accounting for leases. The Company entered into a tenancy agreement for the lease of factory premises for a period of ten years from October 1, 2004 to December 31, 2014, with annual rent of $13,563 (or RMB 94,600), which is subject to a 10% increase every four subsequent years.
The Company leases office space from Weibing Lu, the Company’s chief executive officer, for a period of five years from January 1, 2007 to December 31, 2011, with annual rent of approximately $24,000 (or RMB 165,600). The Company also entered into a tenancy agreement with Weibing Lu for the lease of Shanghai Siqiang’s office for a period of ten years from August 1, 2007 to August 1, 2017, with annual rent of approximately $21,000 (or RMB 144,000).
The Company entered into a tenancy agreement for the lease of an office space in California for a period of three years from July 1, 2009 to July 1, 2012 with monthly rent of $1,100.
The minimum future lease payments for the next five years and thereafter are as follows:
| Period | Amount | |||
| (Unaudited) | ||||
| Nine months ending December 31, 2010 | $ | 65,728 | ||
| Year ending December 31, 2011 | 72,496 | |||
| Year ending December 31, 2012 | 41,603 | |||
| Year ending December 31, 2013 | 35,003 | |||
| Year ending December 31, 2014 | 35,003 | |||
| Year ending December 31, 2015 and thereafter | 63,374 | |||
| Total | $ | 313,207 | ||
Rental expense for the three months ended March 31, 2010 and 2009 amounted to $6,768 and $14,804, respectively.
(b) Legal proceedings
From time to time, the Company is involved in legal matters arising in the ordinary course of business. Management currently is not aware of any legal matters or pending litigation which would have a significant effect on the Company’s consolidated financial statements as of March 31, 2010.
In May 2007, Andrew Chien filed suit against the Company, Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in United States District Court for the District of Connecticut, alleging causes of action for violation of Sections 10(b) and 20(a) of the Exchange Act. On July 17, 2008, in a decision that is now published, the Court granted defendants' motion to dismiss and subsequently dismissed the lawsuit, entering judgment on behalf of the defendants. Mr. Chien filed a notice of appeal of the Court's dismissal of his lawsuit, opposed by the defendants, which remains pending. Additionally, on February 5, 2009, the Court issued a ruling on defendants' motion for sanctions, finding the action filed by Mr. Chien to have been entirely frivolous, and to have constituted a "substantial" violation of Federal Rule of Civil Procedure Rule 11, and imposed significant monetary sanctions on both Mr. Chien and his former attorney. As part of the basis for imposing sanctions on Mr. Chien personally, the Court specifically found that Mr. Chien had knowledge of facts directly contradicting the allegations of his complaint, as evident in internet postings he made on online message boards. Mr. Chien subsequently filed motions seeking to "re-open" this case, and to recuse the judge, but both motions were denied. A Notice of Appeal concerning the ruling awarding sanctions against him was also filed by Mr. Chien. All appeals, including the one referenced below concerning Mr. Chien's second lawsuit, were subsequently consolidated and remain pending, although briefing has been completed.
Subsequently, Mr. Chien, proceeding pro se, filed another lawsuit against the Company, Scott Cramer, Steve Lowe, David Wassung and Weibing Lu in Connecticut Superior Court, alleging causes of action similar to those alleged in his federal complaint described above as well as state law causes of action. The case was removed to the U.S. District Court, District of Connecticut, and assigned to the same Judge who dismissed Mr. Chien’s related federal action. On June 8, 2009, the Court granted defendants’ motion to dismiss this action in its entirety, and denied Mr. Chien’s motion to further amend his complaint. Mr. Chien filed a Notice of Appeal concerning the ruling dismissing this lawsuit, which has been consolidated with Mr. Chien’s appeal of his other lawsuit.
Other than the above described legal proceedings, the Company is not aware of any other legal matters in which purchasers, any director, officer, or any owner of record or beneficial owner of more than five percent of any class of voting securities of the Company, or any affiliate of purchaser, or of any such director, officer, affiliate of the Company, or security holder, is a party adverse to the Company or has a material adverse interest to the Company. No provision has been made in the consolidated financial statements for the above contingencies.
F-49
(c) Ownership of leasehold property
In 2005, a shareholder contributed a leasehold office building as additional capital of Xian Tianxing. However, the title of the leasehold property has not passed to the Company. The Company does not believe there are any legal barriers for the shareholder to transfer the ownership to the Company. However, in the event that the Company fails to obtain the ownership certificate for the leasehold property, there is a risk that the building will need to be vacated due to unofficial ownership. Management believes that this possibility is remote, and as such, no provision has been made in the consolidated financial statements for this potential occurrence.
(d) R&D Project
During the first quarter of 2008, Xian Tianxing contracted with Northwestern Agricultural Technology University to jointly work on an R&D project concerning the application of nano-technology in the prevention of major milk cow disease. The total projected budget for this project is approximately $574,000 (RMB 4 million) which is to be paid according to the completed stages of the project. The Company expects this project to be completed in 2010. As of March 31, 2010 and 2009, the Company incurred approximately $0 and $117,352 (RMB 800,986) of expenses relating to this project. The project reached trial stage in June 2009 and the Company expects to obtain veterinary permit for the new product from government on 2010.
During 2008, Xian Tianxing contracted with Shanxi Shenzhou Bio-pharmaceuticals Technology Company to jointly work on a R&D project with a contracted amount of approximately $308,000. As of December 31, 2009, the Company incurred approximately $102,627 (RMB 700,000) expenses relating to this project. This project was completed by December 31, 2009.
During the year ended December 31, 2009, Xian Tianxing contracted with the Fourth Military Medical University to jointly work on a R&D project with a contracted amount of approximated $880,200(RMB 6,000,000). As of March 31, 2010 and 2009, the Company incurred approximately $43,995 (RMB 300,000) and $0 expenses relating to this project.
F-50

(1) A distributor’s retail shop selling our products.
(2) Injection workshop in our manufacturing facilities in Huxian, Xi’an, Shaanxi Province, People’s Republic of China.
(3) Our booth at a national trade show in Xi’an, Shaanxi Province, People’s Republic of China.
Shares

SKYSTAR BIO-PHARMACEUTICAL COMPANY
| PROSPECTUS |
| Rodman & Renshaw, LLC | JMP Securities |
Until , all dealers that effect transactions in these securities, whether or not participating in this offering, may be required to deliver a prospectus. This is in addition to the dealers’ obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
PART II
Item 13. Other Expenses of Issuance and Distribution
The following table sets forth an estimate of the costs and expenses payable by Skystar in connection with the offering described in this registration statement. All of the amounts shown are estimates except the Securities and Exchange Commission Registration Fee:
| Securities and Exchange Commission Registration Fee | $ | 2,105.58 | ||
| FINRA Filing Fees | $ | |||
| Printing Fees | $ | |||
| Accounting Fees and Expenses | $ | |||
| Legal Fees and Expenses | $ | |||
| Miscellaneous | $ | |||
| Total | $ | * |
* Estimated
Item 14. Indemnification of Directors and Officers
Pursuant to Article 7 of our Articles of Incorporation and Nevada’s Revised Business Statutes, the Company adopted Bylaws with the following indemnification provisions for our directors and officers:
“Section 8.1. Indemnification. No officer or director shall be personally liable for any obligations arising out of any acts or conduct of said officer or director performed for or on behalf of the Corporation. The Corporation shall and does hereby indemnify and hold harmless each person and his heirs and administrators who shall serve at any time hereafter as a director or officer of the Corporation from and against any and all claims, judgments and liabilities to which such persons shall become subject by reason of any action alleged to have been heretofore or hereafter taken or omitted to have been taken by him as such director or officer, and shall reimburse each such person for all legal and other expenses reasonably incurred by him in connection with any such claim of liability; including power to defend such person from all suits as provided for under the provisions of the Nevada Corporation Laws; provided, however that no such person shall be indemnified against, or be reimbursed for, any expense incurred in connection with any claim or liability arising out of his own gross negligence or willful misconduct. The rights accruing to any person under the foregoing provisions of this section shall not exclude any other right to which he may lawfully be entitled, nor shall anything herein contained restrict the right of the Corporation to indemnify or reimburse such person in any proper case, even though not specifically herein provided for. The Corporation, its directors, officers, employees and agents shall be fully protected in taking any action or making any payment or in refusing so to do in reliance upon the advice of counsel.
Section 8.2. Other Indemnification. The indemnification herein provided shall not be deemed exclusive of any other rights to which those seeking indemnification may be entitled under any bylaw, agreement, vote of stockholders or disinterested directors, or otherwise, both as to action in his official capacity and as to action in another capacity while holding such office, and shall continue as to a person who has ceased to be a director, officer or employee and shall inure to the benefit of the heirs, executors and administrators of such a person.
Section 8.3. Insurance. The Corporation may purchase and maintain insurance on behalf of any person who is or was a director, officer or employee of the Corporation, or is or was serving at the request of the Corporation in such capacity for another corporation, partnership, joint venture, trust or other enterprise, against any liability asserted against him and incurred by him in any capacity, or arising out of his status as such, whether or not the Corporation would have the power to indemnify him against liability under the provisions of this Article VIII or the laws of the State of Nevada.
Section 8.4. Settlement by Corporation. The right of any person to be indemnified shall be subject always to the right of the Corporation by its Board of Directors, in lieu of such indemnity, to settle any such claim, action, suit or proceeding at the expense of the Corporation by the payment of the amount of such settlement and the costs and expenses incurred in connection therewith.”
These indemnification provisions may be sufficiently broad to permit indemnification of the registrant's executive officers and directors for liabilities (including reimbursement of expenses incurred) arising under the Securities Act of 1933, as amended (the “Securities Act”).
II-1
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. No pending material litigation or proceeding involving our directors, executive officers, employees or other agents as to which indemnification is being sought exists, and we are not aware of any pending or threatened material litigation that may result in claims for indemnification by any of our directors or executive officers.
Item 15. Recent Sales of Unregistered Securities
The following is a summary of the transactions by Skystar during the last three years involving sales of our securities that were not registered under the Securities Act. Unless otherwise stated, sale transactions of our common stock take into account the 1-for-10 reverse stock split effected on May 12, 2009 and the 2-for-1 forward stock split effected on November 16, 2009.
On or about December 6, 2007, we entered into an Amendment, Exchange and Waiver Agreement (the “Amendment Agreement”), dated as of November 9, 2007, with certain of the investors who participated in the February 2007 private financing. The Amendment Agreement amends certain terms and conditions of the convertible debentures, warrants, and a related agreement. The investors that executed the Amendment Agreement are hereinafter referred to collectively as “Participating Purchasers”. The Amendment Agreement is deemed to be: (a) our notice to require conversion of the entire outstanding principal of the convertible debentures held by the Participating Purchasers and all accrued but unpaid interest thereto; and (b) the Participating Purchasers’ notice to the Company to exercise all of their unexercised warrants on a cashless basis. Pursuant to the conversion notice we issued an aggregate of 615,224 shares of our common stock (the “Debenture Shares”) to the Participating Purchasers, of which 231,390 shares are registered pursuant to our registration statement on Form SB-2, which was deemed effective September 25, 2007, as amended, (SEC File No. 333-143449). The balance of the Debenture Shares were issued to the Participating Purchasers in reliance on the exemptions for sales of securities not involving a public offering to accredited investors, as set forth in Rule 506 promulgated under the Securities Act of 1933, as amended (the “Securities Act”), and in Section 4(2) of the Securities Act. Pursuant to the exercise notice, we issued an aggregate of 191,638 shares of our common stock to the Participating Purchasers, in reliance on the exemptions for sales of securities not involving a public offering to accredited investors, as set forth in Rule 506 promulgated under the Securities Act and in Section 4(2) of the Securities Act.
On or about March 31, 2008, we entered into an Amendment and Waiver Agreement (the “March 2008 Amendment Agreement”) with two institutional and accredited investors who acquired two of our convertible debentures in a private transaction from their original holders. The March 2008 Amendment Agreement amends the terms of these convertible debentures by: (a) changing the conversion price from $5.00 per share to $400 per share; (b) deleting certain conditions for mandatory conversion of their convertible debentures; (c) granting us the right to force mandatory conversion at any time, and (d) allowing us to designate the date for the mandatory conversion. Additionally, the March 2008 Amendment Agreement is deemed to be: (a) the Company’s notice to require conversion of the entire outstanding principal of these two convertible debentures and all accrued but unpaid interest thereto. Lastly, the Agreement is deemed a waiver of any claim for default under the terms of the Securities Purchase Agreement, the convertible debentures and a certain Registration Rights Agreement entered into in connection with the Securities Purchase Agreement. The transaction contemplated by the Agreement closed on April 21, 2008, upon the issuance of shares of our restricted common stock to these investors pursuant to the terms of the March 2008 Amendment Agreement. Accordingly, we issued an aggregate of 245,502 shares of our common stock to these investors in reliance on the exemptions for sales of securities not involving a public offering to accredited investors, as set forth in Rule 506 promulgated under the Securities Act and in Section 4(2) of the Securities Act.
On April 21, 2008, we issued 18,000 shares of our restricted common stock to R. Scott Cramer, a member of the Company’s board of directors. These restricted common shares were issued to Mr. Cramer for services unrelated to Mr. Cramer’s duties as a director.
On April 21, 2008, we issued 42,080 shares of our restricted common stock to our legal counsel. These restricted common shares were issued to the legal counsel as partial payment for services rendered.
On December 4, 2008, we issued 5,218 shares of our restricted common stock to Bennet P. Tchaikovsky as compensation, pursuant to the terms of the Loanout Agreement under which we engaged Mr. Tchaikovsky’s services as our chief financial officer.
On May 11, 2009, we issued 5,218 shares of our restricted common stock to Mr. Tchaikovsky as compensation, pursuant to the terms of the Loanout Agreement.
On February 26, 2010, we issued 10,830 shares of our restricted common stock to Bennet P. Tchaikovsky as compensation, pursuant to the terms of his services agreement as our chief financial officer, which position he resigned from on April 16, 2010.
II-2
On February 26, 2010, we issued 5,556 shares of our restricted common stock to Mark D. Chen as compensation, pursuant to the terms of his directorship agreement. On March 15, 2010, 2,778 shares of our restricted common stock were issued to Mr. Chen in error. These shares were subsequently cancelled after March 31, 2010.
| (a) | Exhibits |
Exhibit Number | Description | |
| 1.1 | Form of Underwriting Agreement ** | |
| 2.1 | Share Purchase Agreement by and between The Cyber Group Network, Inc. and Howard L. Allen and Donald G. Jackson (shareholders of Hollywood Entertainment Network, Inc.) dated May 12, 2000 (1) | |
| 2.2 | Plan of Merger Agreement between The Cyber Group Network Corporation and CGN Acquisitions Corporation dated December 7, 2000 (2) | |
| 2.3 | Share Exchange Agreement between The Cyber Group Network Corporation, R. Scott Cramer, Steve Lowe, David Wassung and Skystar Bio-Pharmaceutical, and the Skystar Shareholders dated September 20, 2005 (3) | |
| 3.1 | Articles of Incorporation (4) | |
| 3.2 | Certificate of Amendment and Certificate of Change filed with the Nevada Secretary of State on February 13, 2006 (5) | |
| 3.3 | Certificate of Amendment to Increase Number of Authorized Shares of Common Stock filed with the Nevada Secretary of State on July 11, 2008 (11) | |
| 3.4 | Amended and Restated Bylaws (12) | |
| 3.5 | Certificate of Change Pursuant to NRS 78.209 filed with the Nevada Secretary of State on May 7, 2009 and effective on May 12, 2009 (14) | |
| 3.6 | Certificate of Change Pursuant to NRS 78.209 as filed with the Nevada Secretary of State on November 12, 2009 and effective on November 16, 2009 (18) | |
| 4.1 | Certificate of Designation of Series B Convertible Preferred Stock (4) | |
| 4.2 | Form of Class A Convertible Debenture (6) | |
| 4.3 | Form of Class B Convertible Debenture (6) | |
| 4.4 | Form of Class A Warrant (6) | |
| 4.5 | Form of Class B Warrant (6) | |
| 4.6 | Form of Common Stock Certificate (17) | |
| 4.7 | Form of Common Stock Purchase Option granted to the representative of the underwriters ** | |
| 5.1 | Form of Opinion of Richardson & Patel LLP ** | |
| 10.1 | Consulting Services Agreement between Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”) and Xian Tianxing Bio-Pharmaceutical Co., Ltd. (“Xian Tianxing”) dated October 28, 2005 (4) | |
| 10.2 | Operating Agreement among Skystar Cayman, Xian Tianxing and the majority stockholders of Xian Tianxing (“Xian Tianxing Majority Stockholders”) dated October 28, 2005 (4) | |
| Equity Pledge Agreement among Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | ||
| 10.4 | Proxy Agreement Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | |
| 10.5 | Option Agreement Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | |
| 10.6 | Amendment to Consulting Services Agreement among Skystar Cayman, Xian Tianxing and Sida Biotechnology (Xian) Co., Ltd. (“Sida”) dated March 10, 2008 (7) | |
| 10.7 | Agreement to Transfer of Operating Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.8 | Amendment to Equity Pledge Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, and Sida dated March 10, 2008 (7) | |
| 10.9 | Designation Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.10 | Agreement to Transfer of Option Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.11 | Employment Agreement with Weibing Lu dated May 5, 2008 (9) | |
| 10.12 | Loanout Agreement with Worldwide Officers, Inc. with respect to the services of Bennet Tchaikovsky, our Chief Financial Officer, dated May 5, 2008 (9) | |
| 10.13 | Form of Director Offer Letter with Mr. Qiang Fan and Mr. Winston Yen (11) |
II-3
| 10.14 | Form of Director Offer Letter with Chengtun Qu and Shouguo Zhao (12) | |
| 10.15 | Form of Amendment to Loanout Agreement with Worldwide Officers, Inc. (15) | |
| 10.16 | Form of Director Offer Letter with Mark D. Chen (15) | |
| 10.17 | Agreement with R. Scott Cramer dated March 30, 2010 (20) | |
| 10.18 | Employment Agreement with Michael Hongjie Lan dated April 16, 2010 (21) | |
| 10.19 | Services Agreement with R. Scott Cramer dated April 16, 2010 (21) | |
| 10.20 | Restricted Stock Award Agreement with R. Scott Cramer dated April 16, 2010 (21) | |
| 10.21 | Form of 3-month Lock-up Agreement * | |
| 21.1 | List of subsidiaries (13) | |
| 23.1 | Consent of Frazer Frost, LLP ** | |
| 23.2 | Consent of Richardson & Patel LLP (included in Exhibit 5.1) * | |
| 24.1 | Power of Attorney (included as part of the signature page to the registration statement) | |
| 99.1 | Legal Opinion from Allbright Law Offices regarding, among other things, the contractual arrangements Skystar Cayman entered into with Xian Tianxing and its stockholders, dated November 3, 2005 (10) | |
| 99.2 | Legal Opinion from Allbright Law Offices regarding the transfer of the contractual arrangements from Skystar Cayman to Sida, dated April 29, 2008 (10) | |
| 99.3 | Lease Agreement between Xian Tianxing and Weibing Lu dated June 1, 2007 (9) | |
| 99.4 | Lease Agreement between Shanghai Siqiang Biotechnological Co., Ltd. and Weibing Lu dated June 17, 2007 (10) | |
| 99.5 | Summary of Research Arrangement between Shanghai Poultry Verminosis Institute and Xian Tianxing (10) | |
| 99.6 | Cooperation Agreement between Shaanxi Microbial Institute and Xian Tianxing (10) | |
| 99.7 | Technology Cooperation Agreement with Fourth Military Medical University (20) |
* To be filed upon amendment.
** Filed herewith.
| (1) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on June 1, 2000. | |
| (2) | Incorporated by reference from the Registrant’s Current Report on Form 8-K/A filed on January 12, 2001. | |
| (3) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on September 26, 2005. | |
| (4) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 14, 2005. | |
| (5) | Incorporated by reference from the Registrant’s Annual Report on Form 10-KSB filed on April 17, 2006. | |
| (6) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 5, 2007. | |
| (7) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 11, 2008. | |
| (8) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K filed on April 2, 2008. | |
| (9) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 5, 2008. | |
| (10) | Incorporated by reference from the Registrant’s Registration Statement on Form S-1/A filed on June 26, 2008. | |
| (11) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on July 14, 2008. | |
| (12) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on July 15, 2008. | |
| (13) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K filed on April 15, 2009. | |
| (14) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 18, 2009. | |
| (15) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 27, 2009. | |
| (16) | Incorporated by reference from the Registrant’s Amendment to Registration Statement on Form S-1/A filed on June 2, 2009. | |
| (17) | Incorporated by reference from the Registrant’s Amendment to Registration Statement on Form S-1/A filed on June 26, 2009. | |
| (18) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 17, 2009. | |
| (19) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 7, 2010. | |
| (20) | Incorporate by reference from the Registrant’s Annual Report on Form 10-K filed on March 31, 2010. | |
| (21) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on April 19, 2010. |
Item 17. Undertakings
| (a) | The undersigned registrant hereby undertakes to: |
| (1) | File, during any period in which offers or sales are being made, a post-effective amendment to this registration statement to: |
| i. | Include any prospectus required by Section 10(a)(3) of the Securities Act of 1933, as amended (the “Securities Act”); |
II-4
| ii. | Reflect in the prospectus any facts or events which, individually or in the aggregate, represent a fundamental change in the information in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Securities and Commission (the “Commission”) pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20 percent change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement. |
iii. Include any additional or changed material information on the plan of distribution.
| (2) | For determining liability under the Securities Act, each such post-effective amendment as a new registration statement relating to the securities offered, and the offering of such securities at that time shall be deemed to be the initial bona fide offering. |
| (3) | File a post-effective amendment to remove from registration by means of a post-effective amendment any of the securities that remain unsold at the end of the offering. |
| (4) | For determining liability of the undersigned small business issuer under the Securities Act to any purchaser in the initial distribution of the securities, the undersigned small business issuer undertakes that in a primary offering of securities of the undersigned small business issuer pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned small business issuer will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser: |
| i. | Any preliminary prospectus or prospectus of the undersigned small business issuer relating to the offering required to be filed pursuant to Rule 424; |
| ii. | Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned small business issuer or used or referred to by the undersigned small business issuer; |
| iii. | The portion of any other free writing prospectus relating to the offering containing material information about the undersigned small business issuer or its securities provided by or on behalf of the undersigned small business issuer; and |
| iv. | Any other communication that is an offer in the offering made by the undersigned small business issuer to the purchaser. |
| (b) | Provide to the underwriters at the closing specified in the underwriting agreements, certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser. |
| (c) | Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the small business issuer pursuant to the foregoing provisions, or otherwise, the small business issuer has been advised that in the opinion of the Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the small business issuer of expenses incurred or paid by a director, officer or controlling person of the small business issuer in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the small business issuer will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue. |
(d)
| (1) | For determining any liability under the Securities Act, treat the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the small business issuer under Rule 424(b)(1), or (4), or 497(h) under the Securities Act as part of this registration statement as of the time the Commission declared it effective. |
| (2) | For determining any liability under the Securities Act, treat each post-effective amendment that contains a form of prospectus as a new registration statement for the securities offered in the registration statement, and that offering of the securities at that time as the initial bona fide offering of those securities. |
II-5
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing this Registration Statement on Form S-1 to be signed on its behalf by the undersigned, in the City of Xian, Shaanxi Province, P.R. China on June 1, 2010.
| SKYSTAR BIO-PHARMACEUTICAL COMPANY | ||
| By: | /s/ Weibing Lu | |
| Weibing Lu | ||
| Chief Executive Officer | ||
Each person whose signature appears below hereby constitutes and appoints, jointly and severally, Weibing Lu and Michael H. Lan, and each of them, as his or her attorney-in-fact, with full power of substitution, for him or her in any and all capacities, to sign any and all amendments, including post-effective amendments, to this registration statement, and any and all registration statements related to the offering covered by this registration statement and filed under the Securities Act of 1933, as amended, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, hereby ratifying and confirming our signatures as they may be signed by his or her attorney to any and all amendments to said registration statement.
Pursuant to the requirements of the Securities Act of 1933, as amended, this registration statement has been signed by the following persons in the capacities and on the dates indicated:
| Title | Date | |||
| /s/ Weibing Lu | Chief Executive Officer and Director | June 1, 2010 | ||
| Weibing Lu | (Principal Executive Officer) | |||
| /s/ Michael H. Lan | Chief Financial Officer | June 1, 2010 | ||
| Michael H. Lan | (Principal Financial and Accounting Officer) | |||
| /s/ Wei Wen | Secretary and Director | June 1, 2010 | ||
| Wei Wen | ||||
| /s/ R. Scott Cramer | Director | June 1, 2010 | ||
| R. Scott Cramer | ||||
| /s/ Chengtun Qu | Director | June 1, 2010 | ||
| Chengtun Qu | ||||
| /s/ Shouguo Zhao | Director | June 1, 2010 | ||
| Shouguo Zhao |
II-6
EXHIBIT INDEX
Exhibit Number | Description | |
| 1.1 | Form of Underwriting Agreement ** | |
| 2.1 | Share Purchase Agreement by and between The Cyber Group Network, Inc. and Howard L. Allen and Donald G. Jackson (shareholders of Hollywood Entertainment Network, Inc.) dated May 12, 2000 (1) | |
| 2.2 | Plan of Merger Agreement between The Cyber Group Network Corporation and CGN Acquisitions Corporation dated December 7, 2000 (2) | |
| 2.3 | Share Exchange Agreement between The Cyber Group Network Corporation, R. Scott Cramer, Steve Lowe, David Wassung and Skystar Bio-Pharmaceutical, and the Skystar Shareholders dated September 20, 2005 (3) | |
| 3.1 | Articles of Incorporation (4) | |
| 3.2 | Certificate of Amendment and Certificate of Change filed with the Nevada Secretary of State on February 13, 2006 (5) | |
| 3.3 | Certificate of Amendment to Increase Number of Authorized Shares of Common Stock filed with the Nevada Secretary of State on July 11, 2008 (11) | |
| 3.4 | Amended and Restated Bylaws (12) | |
| 3.5 | Certificate of Change Pursuant to NRS 78.209 filed with the Nevada Secretary of State on May 7, 2009 and effective on May 12, 2009 (14) | |
| 3.6 | Certificate of Change Pursuant to NRS 78.209 as filed with the Nevada Secretary of State on November 12, 2009 and effective on November 16, 2009 (18) | |
| 4.1 | Certificate of Designation of Series B Convertible Preferred Stock (4) | |
| 4.2 | Form of Class A Convertible Debenture (6) | |
| 4.3 | Form of Class B Convertible Debenture (6) | |
| 4.4 | Form of Class A Warrant (6) | |
| 4.5 | Form of Class B Warrant (6) | |
| 4.6 | Form of Common Stock Certificate (17) | |
| 4.7 | Form of Common Stock Purchase Option granted to the representative of the underwriters ** | |
| 5.1 | Form of Opinion of Richardson & Patel LLP ** | |
| 10.1 | Consulting Services Agreement between Skystar Bio-Pharmaceutical (Cayman) Holdings Co., Ltd. (“Skystar Cayman”) and Xian Tianxing Bio-Pharmaceutical Co., Ltd. (“Xian Tianxing”) dated October 28, 2005 (4) | |
| 10.2 | Operating Agreement among Skystar Cayman, Xian Tianxing and the majority stockholders of Xian Tianxing (“Xian Tianxing Majority Stockholders”) dated October 28, 2005 (4) | |
| Equity Pledge Agreement among Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | ||
| 10.4 | Proxy Agreement Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | |
| 10.5 | Option Agreement Skystar Cayman, Xian Tianxing and the Xian Tianxing Majority Stockholders dated October 28, 2005 (4) | |
| 10.6 | Amendment to Consulting Services Agreement among Skystar Cayman, Xian Tianxing and Sida Biotechnology (Xian) Co., Ltd. (“Sida”) dated March 10, 2008 (7) | |
| 10.7 | Agreement to Transfer of Operating Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.8 | Amendment to Equity Pledge Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, and Sida dated March 10, 2008 (7) | |
| 10.9 | Designation Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing’s Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.10 | Agreement to Transfer of Option Agreement among Skystar Cayman, Xian Tianxing, Xian Tianxing Majority Stockholders, Weibing Lu and Sida dated March 10, 2008 (7) | |
| 10.11 | Employment Agreement with Weibing Lu dated May 5, 2008 (9) | |
| 10.12 | Loanout Agreement with Worldwide Officers, Inc. with respect to the services of Bennet Tchaikovsky, our Chief Financial Officer, dated May 5, 2008 (9) | |
| 10.13 | Form of Director Offer Letter with Mr. Qiang Fan and Mr. Winston Yen (11) |
| 10.14 | Form of Director Offer Letter with Chengtun Qu and Shouguo Zhao (12) | |
| 10.15 | Form of Amendment to Loanout Agreement with Worldwide Officers, Inc. (15) | |
| 10.16 | Form of Director Offer Letter with Mark D. Chen (15) | |
| 10.17 | Agreement with R. Scott Cramer dated March 30, 2010 (20) | |
| 10.18 | Employment Agreement with Michael Hongjie Lan dated April 16, 2010 (21) | |
| 10.19 | Services Agreement with R. Scott Cramer dated April 16, 2010 (21) | |
| 10.20 | Restricted Stock Award Agreement with R. Scott Cramer dated April 16, 2010 (21) | |
| 10.21 | Form of 3-month Lock-up Agreement * | |
| 21.1 | List of subsidiaries (13) | |
| 23.1 | Consent of Frazer Frost, LLP ** | |
| 23.2 | Consent of Richardson & Patel LLP (included in Exhibit 5.1) * | |
| 24.1 | Power of Attorney (included as part of the signature page to the registration statement) | |
| 99.1 | Legal Opinion from Allbright Law Offices regarding, among other things, the contractual arrangements Skystar Cayman entered into with Xian Tianxing and its stockholders, dated November 3, 2005 (10) | |
| 99.2 | Legal Opinion from Allbright Law Offices regarding the transfer of the contractual arrangements from Skystar Cayman to Sida, dated April 29, 2008 (10) | |
| 99.3 | Lease Agreement between Xian Tianxing and Weibing Lu dated June 1, 2007 (9) | |
| 99.4 | Lease Agreement between Shanghai Siqiang Biotechnological Co., Ltd. and Weibing Lu dated June 17, 2007 (10) | |
| 99.5 | Summary of Research Arrangement between Shanghai Poultry Verminosis Institute and Xian Tianxing (10) | |
| 99.6 | Cooperation Agreement between Shaanxi Microbial Institute and Xian Tianxing (10) | |
| 99.7 | Technology Cooperation Agreement with Fourth Military Medical University (20) |
* To be filed upon amendment.
** Filed herewith.
| (1) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on June 1, 2000. | |
| (2) | Incorporated by reference from the Registrant’s Current Report on Form 8-K/A filed on January 12, 2001. | |
| (3) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on September 26, 2005. | |
| (4) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 14, 2005. | |
| (5) | Incorporated by reference from the Registrant’s Annual Report on Form 10-KSB filed on April 17, 2006. | |
| (6) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 5, 2007. | |
| (7) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on March 11, 2008. | |
| (8) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K filed on April 2, 2008. | |
| (9) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 5, 2008. | |
| (10) | Incorporated by reference from the Registrant’s Registration Statement on Form S-1/A filed on June 26, 2008. | |
| (11) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on July 14, 2008. | |
| (12) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on July 15, 2008. | |
| (13) | Incorporated by reference from the Registrant’s Annual Report on Form 10-K filed on April 15, 2009. | |
| (14) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 18, 2009. | |
| (15) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on May 27, 2009. | |
| (16) | Incorporated by reference from the Registrant’s Amendment to Registration Statement on Form S-1/A filed on June 2, 2009. | |
| (17) | Incorporated by reference from the Registrant’s Amendment to Registration Statement on Form S-1/A filed on June 26, 2009. | |
| (18) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on November 17, 2009. | |
| (19) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on January 7, 2010. | |
| (20) | Incorporate by reference from the Registrant’s Annual Report on Form 10-K filed on March 31, 2010. | |
| (21) | Incorporated by reference from the Registrant’s Current Report on Form 8-K filed on April 19, 2010. |