Exhibit 99.1
Poster #406
APS 2014, Tampa, FL
rvickery@theravance.com
Axelopran Phase 2b Study Demonstrates a Sustained Increase in Bowel Movement Frequency in Patients Regardless of Duration of Opioid-Induced Constipation
Ross Vickery, PhD(1), Lynn Webster, MD(2), Yu-Ping Li, PhD(1), Neil Singla, MD(3), and Daniel Canafax, PharmD(1)
(1) Theravance, Inc., South San Francisco, CA; (2) CRI Lifetree, Inc., Salt Lake City, UT; (3) Lotus Clinical Research, Inc., Pasadena, CA
Introduction
· Opioid analgesics such as morphine continue to play a critical role in chronic cancer and non-cancer pain control.(1) Despite their effectiveness, opioids have significant drawbacks, notably the development of analgesic tolerance and physical dependence, sedation, respiratory depression and bowel dysfunction.(2)
· Opioid-induced constipation (OIC) is common, affecting up to 80% of patients receiving opioids for chronic non-cancer pain.(3)
· Axelopran (formerly TD-1211) is an investigational, peripherally selective, multivalent mu-opioid receptor antagonist designed to alleviate gastrointestinal side effects of opioid therapy without affecting analgesia.
· Safety and efficacy results, including the primary and key secondary endpoints, from a 5-week, Phase 2b study in chronic non-cancer pain OIC patients have been previously reported.(4)
· Since OIC is not prone to tolerance and patients can experience OIC for the duration of opioid therapy, patients were divided into short and long duration of OIC groups (<5 and >5 years) to explore if OIC duration impacts axelopran treatment response.
Methods
· A 5-week, double-blind, randomized, multi-center, placebo-controlled, parallel-group study was conducted in chronic non-cancer pain patients with OIC, defined as <5 spontaneous bowel movements (SBMs) over a 2-week baseline period and at least one additional symptom of constipation in at least 25% of the bowel movements.
· For the first 4 days of dosing, patients randomized to axelopran received 5mg daily and on Day 5, remained at 5mg or were dose-escalated to 10mg or 15mg daily for the remainder of the treatment period. Patients randomized to placebo received placebo for all 5 weeks.
· For at least 14 days prior to Day 1, patients were on a stable chronic opioid regimen, with a total daily dose of >30mg morphine equivalent units (MEU).
· Patients were required to stop laxatives and bowel regimens, except protocol-permitted rescue bisacodyl use, throughout the study.
· Electronic diaries collected frequency, timing, and symptoms of bowel movements; use of laxatives and opioids; daily pain scores; and satisfaction/quality of life metrics.
· Primary efficacy endpoint was the change from baseline in weekly average complete spontaneous bowel movements (CSBMs) over weeks 2-5 of treatment.
· Key secondary endpoint was the change from baseline in weekly average spontaneous bowel movements (SBMs) over the same period.
· Week 1 was excluded from the primary analysis in order to confirm the durability of response and predictability of longer term efficacy studies.
· Patients were divided into short and long duration of OIC groups (<5 and >5 years) and evaluated on the study’s primary and key secondary endpoints.
Results
Patient baseline demographics
· As shown in Table 1, baseline characteristics were similar for all treatment groups in the overall population as well as the short and long duration of OIC groups.
· Subjects were on a representative spectrum of opioids.
· Daily opioid doses ranged from 30-1740 oral MEU.
· Back pain was the most commonly reported reason for chronic opioid use.
· Mean and range of OIC duration in the study were 6.0 years and 0.2 - 39.3 years, respectively.
Table 1: Patient Baseline Demographics by Duration of OIC
|
|
|
| Axelopran |
| ||||
Overall Population |
| Placebo |
| 5 mg |
| 10 mg |
| 15 mg |
|
Mean Age (years) |
| 47.6 |
| 48.3 |
| 49.2 |
| 48.9 |
|
Female Gender |
| 28 |
| 37 |
| 32 |
| 30 |
|
BMI Mean (kg/m2) |
| 28.3 |
| 27.8 |
| 27.8 |
| 28.1 |
|
Duration of OIC (yrs) |
| 5.5 |
| 6.4 |
| 6.7 |
| 5.3 |
|
|
|
|
| Axelopran |
| ||||
Duration of OIC <5 years |
| Placebo |
| 5 mg |
| 10 mg |
| 15 mg |
|
Mean Age (years) |
| 45.7 |
| 47.5 |
| 48.8 |
| 47.9 |
|
Female Gender |
| 17 |
| 16 |
| 15 |
| 17 |
|
BMI Mean (kg/m2) |
| 27.9 |
| 27.4 |
| 27.8 |
| 28.3 |
|
Duration of OIC (yrs) |
| 2.1 |
| 2.2 |
| 2.0 |
| 2.4 |
|
|
|
|
| Axelopran |
| ||||
Duration of OIC >5 years |
| Placebo |
| 5 mg |
| 10 mg |
| 15 mg |
|
Mean Age (years) |
| 49.7 |
| 48.9 |
| 50.3 |
| 49.5 |
|
Female Gender |
| 11 |
| 21 |
| 17 |
| 13 |
|
BMI Mean (kg/m2) |
| 28.8 |
| 28.2 |
| 27.7 |
| 27.8 |
|
Duration of OIC (yrs) |
| 9.2 |
| 9.7 |
| 10.9 |
| 9.9 |
|
Modified Intent to Treat Population
Efficacy Endpoints by Duration of OIC Group
· The baseline frequency of CSBMs and SBMs was similar for short and long duration of OIC groups (Figs. 1-2).
· During weeks 2-5, all doses of axelopran resulted in higher average CSBMs and SBMs per week compared to placebo for the overall population & both OIC duration groups (Figs. 1-2).
· For the >5 year OIC duration group, there was a dose-response relationship in average CSBM and SBM frequency during weeks 2-5 and similarly in the overall population for SBM frequency (Figs. 1-2, Table 2).
Figure 1: Baseline and Weeks 2-5 Average Complete Spontaneous Bowel Movements (CSBMs) by Duration of OIC Group
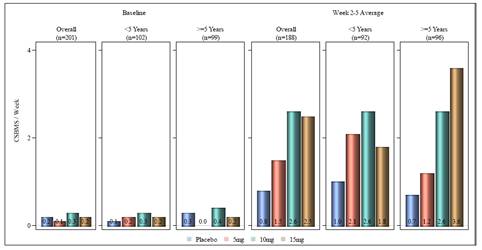
EA population; no imputation
Figure 2: Baseline and Weeks 2-5 Average Spontaneous Bowel Movements (SBMs) by Duration of OIC Group

EA population; no imputation
Figure 3: Pre-Specified SBM Responder Analysis by Duration of OIC Group

EA population. Missing weekly data were imputed as non-responder.
· Responder definition: >3 SBMs per week and an increase of > 1 SBM per week from baseline for >3 weeks over Weeks 2-5
Table 2: Mean (SD) Change from Baseline in Weeks 2-5 Weekly Average CSBMs and SBMs by Baseline Opioid Use
|
| Mean (SD) Change from Baseline in |
| Mean (SD) Change from Baseline in | ||||||||||||
|
| Placebo |
| 5mg |
| 10mg |
| 15mg |
| Placebo |
| 5mg |
| 10mg |
| 15mg |
Overall |
| 0.8 (1.4) |
| 1.5 (2.2) |
| 2.6 (2.4) p=0.0010 |
| 2.5 (3.3) |
| 1.9 (1.8) |
| 2.7 (2.2) |
| 3.4 (2.7) |
| 3.7 (3.0) |
<5 yrs OIC duration |
| 1.0 (1.6) |
| 2.1 (2.9) |
| 2.6 (2.4) |
| 1.8 (2.4) |
| 1.6 (1.7) |
| 2.8 (2.6) |
| 3.0 (2.7) |
| 2.9 (2.4) |
>5 yrs OIC duration |
| 0.7 (1.3) |
| 1.2 (1.4) |
| 2.6 (2.4) |
| 3.6 (4.2) |
| 2.3 (1.8) |
| 2.7 (2.0) |
| 3.8 (2.7) |
| 4.9 (3.5) |
EA population; no imputation. P-values were based on a mixed-effect model repeated measures with baseline as covariate.
Table 3: GI-Related Adverse Events Occurring in at Least 2 Patients in Any Group
|
|
|
| Axelopran | ||||||
Safety Population |
| Placebo |
| 5 mg |
| 10 mg |
| 15 mg |
| All |
No. of Patients and Percentage with GI AEs |
| 11 (20.4%) |
| 13 (23.2%) |
| 15 (28.3%) |
| 14 (26.9%) |
| 42 (26.1%) |
Abdominal Pain |
| 6 (11.1%) |
| 7 (12.5%) |
| 6 (11.3%) |
| 8 (15.4%) |
| 21 (13.0%) |
Abdominal Pain Upper |
| 1 (1.9%) |
| 2 (3.6%) |
| 3 (5.7%) |
| 2 (3.8%) |
| 7 (4.3%) |
Diarrhea |
| 0 |
| 4 (7.1%) |
| 6 (11.3%) |
| 4 (7.7%) |
| 14 (8.7%) |
Flatulence |
| 3 (5.6%) |
| 1 (1.8%) |
| 2 (3.8%) |
| 1 (1.9%) |
| 4 (2.5%) |
Nausea |
| 2 (3.7%) |
| 4 (7.1%) |
| 8 (15.1%) |
| 3 (5.8%) |
| 15 (9.3%) |
Vomiting |
| 1 (1.9%) |
| 4 (7.1%) |
| 1 (1.9%) |
| 0 |
| 5 (3.1%) |
Efficacy Endpoints by Baseline Opioid Dose (con’t)
· Using a pre-specified responder definition, there was a dose-response relationship between responder rate and axelopran dose for the overall population and both OIC duration groups (Fig. 3).
Tolerability and Safety
· Axelopran was generally well tolerated, with overall treatment emergent adverse events (TEAEs) similar between axelopran and placebo and gastrointestinal (GI) TEAEs predominant (Table 3).
· The majority of GI-related AEs were associated with treatment initiation, mild-to-moderate, and resolving within a few days.
· No treatment-related serious adverse events (SAEs) were reported.
· No clinically significant laboratory, ECG, or vital sign abnormalities were observed.
Axelopran Conclusions
· 10mg and 15mg demonstrated a clinically meaningful, sustained response in CSBM and SBM frequency over the 5-week treatment period in patients irrespective of their duration of OIC.
· Generally well-tolerated with no treatment-related SAEs.
· Majority of treatment-related GI AEs were associated with initiation of treatment, resolved within a few days, and were mild or moderate.
References
(1) Walsh, T.D. (2000). Seminars in Oncology, 27, 45-63.
(2) Walsh, T.D. (1990). J. Pain Symptom Manage., 5, 362-367.
(3) Holzer, P. (2012). Current Pharmaceutical Design, 18, 6010-6020.
(4) Vickery, R., et al. PainWeek 2012, Las Vegas, NV, September 5-8. Poster #121.