UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
______________
FORM 10-K
______________
(Mark One)
x ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended December 31, 2006
OR
o TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period from ____________ to__________
Commission file number 001-32518
——————

CYTOMEDIX, INC.
(Exact Name of Registrant as Specified in Its Charter)
Delaware | 23-3011702 | |
(State or Other Jurisdiction of Incorporation or Organization) | (IRS Employer Identification No.) |
416 Hungerford Drive, Suite 330
Rockville, MD 20850
(Address of Principal Executive Offices) (Zip Code)
(240) 499-2680
(Registrant’s Telephone Number, Including Area Code)
——————
Securities registered under Section 12(b) of the Exchange Act: None
Securities registered under Section 12(g) of the Exchange Act:
Common Stock, par value $.0001
(Title of class)
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer and large accelerated filer” in Rule 12b-2 of the Exchange Act. (Check one):
Large Accelerated Filer ¨ | Accelerated Filer x | Non-Accelerated Filer ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the voting stock (Common stock) held by non-affiliates of the registrant as of the close of business on June 30, 2006 was approximately $84 million based on the closing sale price of the Common stock on the American Stock Exchange on that date. The registrant does not have any non-voting common equity.
APPLICABLE ONLY TO ISSUERS INVOLVED IN BANKRUPTCY PROCEEDINGS
DURING THE PRECEDING FIVE YEARS
Indicate by check mark whether the registrant has filed all documents and reports required to be filed by Sections 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court. Yes ý No ¨
APPLICABLE ONLY TO CORPORATE REGISTRANTS
Indicate the number of shares outstanding of each of the registrant’s classes of common stock, as of the latest practicable date. 28,998,248 shares of Common stock, par value $.0001, outstanding as of February 15, 2007.
DOCUMENTS INCORPORATED BY REFERENCE
None.
CYTOMEDIX, INC.
TABLE OF CONTENTS
Page | |||
PART I | |||
| Item 1. | Business | 1 | |
| Item 1A. | Risk Factors | 10 | |
| Item 1B. | Unresolved Staff Comments | 14 | |
| Item 2 | Properties | 14 | |
| Item 3. | Legal Proceedings | 14 | |
| Item 4. | Submission of Matters to a Vote of Security Holders | 14 | |
PART II | |||
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | 16 | |
| Item 6. | Selected Financial Data | 17 | |
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 17 | |
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk | 23 | |
| Item 8. | Financial Statements and Supplementary Data | 24 | |
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 52 | |
| Item 9A. | Controls and Procedures | 52 | |
| Item 9B. | Other Information | 54 | |
PART III | |||
| Item 10. | Directors, Executive Officers and Corporate Governance | 55 | |
| Item 11. | Executive Compensation | 57 | |
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 62 | |
| Item 13. | Certain Relationships and Related Transactions, and Director Independence | 63 | |
| Item 14. | Principal Accounting Fees and Services | 64 | |
PART IV | |||
| Item 15. | Exhibits and Financial Statement Schedules | 64 | |
Signatures | 65 | ||
Exhibit Index | 66 | ||
PART I
ITEM 1. BUSINESS
The terms "Cytomedix," and "Company," as used in this annual report, refer to Cytomedix, Inc.
You are cautioned that this Form 10-K contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. When the words "believes," "plans," "anticipates", "will likely result," "will continue," "projects," "expects," and similar expressions are used in this Form 10-K, they are intended to identify "forward-looking statements," and such statements are subject to certain risks and uncertainties which could cause actual results to differ materially from those projected. Furthermore, the Company’s plans, strategies, objectives, expectations and intentions are subject to change at any time at the discretion of management and the Board of Directors.
These forward-looking statements speak only as of the date this report is filed. The Company does not intend to update the forward-looking statements contained in this report, so as to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events, except as may occur as part of its ongoing periodic reports filed with the SEC.
GENERAL DEVELOPMENT OF THE BUSINESS
Informatix Holdings, Inc. was incorporated in Delaware in 1998. In 1999, an unrelated Arkansas corporation, Autologous Wound Therapy, Inc. ("AWT"), merged with and into Informatix Holdings, Inc. and the name of the surviving corporation was changed to Autologous Wound Therapy, Inc. In 2000, AWT changed its name to Cytomedix, Inc. The principal offices are located in Rockville, Maryland.
In 2001, the Company filed bankruptcy under Chapter 11 of the United States Bankruptcy Code, after which Cytomedix was authorized to continue to conduct its business as debtor and debtor-in-possession. A new board of directors was elected which then appointed a new management team. New management immediately began formulating a plan of reorganization that would enable the Company to reorganize and emerge quickly from Chapter 11 in order to preserve its value as a going concern. The Company emerged from bankruptcy in 2002 under a Plan of Reorganization. At that time, all of the Company’s securities or other claims against or equity interest in the Company were canceled and of no further force or effect. Holders of certain claims or securities were entitled to receive new securities from Cytomedix in exchange for their claims or equity interests prior to bankruptcy. All known and allowed claims and equity interests have been satisfied and resolved as of the filing of this form 10-K.
FINANCIAL INFORMATION ABOUT SEGMENTS AND GEOGRAPHIC REGIONS
Cytomedix has only one operating segment and operates only in the United States. See Item 8, Financial Statements and Supplementary Data.
DESCRIPTION OF THE BUSINESS
Overview
Cytomedix is a biotechnology company that develops and licenses autologous cellular therapies (i.e., therapies using the patient’s own body products), including Cytomedix’s proprietary AutoloGel™ Process to produce a platelet rich plasma gel (“AutoloGel™”) for the treatment of wounds. To create AutoloGel™, the patient’s own platelets and plasma are separated through centrifugation and combined with several reagents. This process releases multiple growth factors from the platelets, creates a fibrin matrix scaffold, and forms a gel that is topically applied to a wound (under the direction of a physician). Upon topical application, the Company believes that AutoloGel™ initiates a reaction that closely mimics the body’s natural healing process.
Company-sponsored studies indicate increased healing for AutoloGelTM as compared to enhanced traditional treatments as well as competing treatments for the treatment of diabetic foot ulcers, the Company’s initial focus within its target market.
Multiple growth factor therapies have not been widely used in the traditional commercial setting because such therapies have generally not been available or widely known by clinicians. Until a few years ago, the autologous process of securing multiple growth factors from a patient’s blood products was, substantially, an exclusive treatment available through outpatient wound care centers affiliated with Curative Health Services (“Curative”). In January 2001, the Company purchased certain technology, assets and intellectual property rights associated with autologous multiple growth factor therapies from Curative and has since refined the product to a more marketable state.
1
Market
Cytomedix’s primary target market is the multi-billion dollar, chronic, non-healing wound market. Such wounds typically arise from one of three etiologies: diabetic foot ulcers, venous leg ulcers, and pressure ulcers. The following table lists the prevalence of these wound types:
Incidence of Chronic Wounds in the U.S.
(number of wounds in millions)
Source: Advanced Wound Management: Healing and Restoring Lives;
Advanced Medical Technology Association (AdvaMed), June 2006
U.S. | |||
| Diabetic Foot Ulcers | 1.5 | ||
| Venous Leg Ulcers | 2.5 | ||
| Pressure Ulcers | 2.0 | ||
| Totals | 6.0 |
The prevalence of chronic wounds in the U.S. is linked directly to increased aging demographics, vascular diseases, venous insufficiency, and excessive pressure and diabetic neuropathy. The prevalence of worldwide chronic wounds is estimated to be 18 million (6).
Diabetic Foot Ulcers
According to the American Diabetes Association (“ADA”)(1), there are approximately 20.8 million people with diabetes in the U.S., or 7% of the total population. It is estimated that 15-20% of these people with diabetes will develop a foot ulcer in their lifetime and that 14-24% of diabetic foot ulcers result in amputation.(2) Over 82,000 amputations per year have been documented.(3) Estimated amputation costs are between $20,000 and $60,000 per procedure (4), implying an aggregate cost of between $1.6 billion and $4.9 billion per year. The chances of a second amputation within 3-5 years may be as high as 50%, with a 5 year post-amputation mortality rate of 39-68%.(5)
Venous Stasis Leg Ulcers
Venous leg ulcers are the most frequently occurring type of chronic wound. The prevalence rises dramatically with age, increasing to 1% of the population over age 60. It is estimated that treatment costs total between $2.5 to $3.5 billion annually and a loss of 2 million workdays per year. (4)
Pressure Ulcers
Over 2.0 million pressure ulcers occur each year with an annual cost greater than $1.3 billion. One study indicates that nearly 15% of hospitalized patients age 65 or older developed a pressure ulcer during a 5-day or longer stay. Furthermore, up to one-fifth of all home health service visits involve care of a pressure ulcer, and more than one-third of people with spinal cord injuries develop pressure ulcers. (4)
References
(1) www.diabetes.org, 2006
(2) H.R 3203 Submitted to the House of Representatives, Sept 30, 2003
(3) National Diabetes Statistics, National Diabetes Information Clearinghouse, National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), NIH, 2004
(4) Advanced Wound Management: Healing and Restoring Lives; Advanced Medical Technology Association (AdvaMed); June 2006
(5) Reiber GE, Boyko EJ, Smith DG: Lower Extremity Foot Ulcers and Amputations in Diabetes. In Diabetes in America. 2nd ed., National Institutes of Health, NIDDK, NIH Pub No. 95-1468, 1995.
(6) Growth Factors: Indications, Products, and Markets; Kalorama Publications; October 2003
2
Strategy
The Company has developed a three-pronged strategy to leverage its intellectual property and capitalize on the market for its AutoloGel™:
| · | Obtain broad reimbursement from third-party payers |
| · | Enforce rights under the Company’s patents |
| · | Target the non-reimbursement sensitive market |
In order to increase the prospects for securing broad reimbursement as well as enhance the sales and marketing efforts, the Company completed a well-controlled, prospective clinical trial and submitted a 510(k) Premarket Notification to the FDA.
Clinical Trial and FDA Clearance
In 2005, the Company completed its prospective, randomized, blinded, controlled, multi-center clinical trial designed to prove the efficacy and safety of its AutoloGel™ System (see discussion in this section under “Government Approval”) for the treatment of non-healing diabetic foot ulcers. The audited results yielded 40 patients who met the trial protocol. Analysis of the size of wounds in the study shows that 35 out of the 40 patients (88%) had wounds that were less than or equal to 7 square centimeters in area and 2 cubic centimeters in volume. For these most common wound sizes in the study, the healing rate of the AutoloGel™ group was 81.3% and that for the control group was 42.1%. The difference of 39.2% between these groups is clearly statistically significant, with a p-value of 0.036. Within the full cohort of the 40 patients, 68.4% of the patients treated with AutoloGel™ achieved full wound closure versus 42.9% of those patients treated in the control group. The difference of 25.5% between the healing rate of the AutoloGel™ group versus the control group is approaching statistical significance with a p-value of 0.125. The Company believes that the healing rates of AutoloGel™ at 81.3% for the most common wound sizes in the study and 68.4% for all wound sizes appear to be better than any other wound care products cleared by the FDA and covered by Medicare reimbursement with which the Company is familiar, although this comparison is not as reliable as a head-to-head study. The control group patients were not on placebo; rather, they were treated using a saline gel cleared by the FDA for wound treatment. If the control group patients healed at the originally anticipated rate of 20-30% for standard treatments for diabetic foot ulcers, the difference between the healing rates in the AutoloGel™ group versus the control group would have been even more strongly statistically significant.
These data reflect the results following an independent audit of the data by a former FDA official responsible for Bio-Research Monitoring. During the audit, Cytomedix discovered that some patients originally included in the trial had not met the inclusion criteria or were not provided treatment according to the study protocol. This audit was conducted at the request of Cytomedix when preliminary data were inconsistent with independent and Company retrospective studies.
Based on the audited results of the trial, and other data compiled by the Company, in late January 2006 Cytomedix submitted a Premarket Notification (“510(k)”) to the FDA seeking clearance of its AutoloGel™ System for diabetic foot ulcers and other indications. While AutoloGel™ is regulated by FDA under the Medical Device Amendments of the Food, Drug and Cosmetic Act, the FDA Center for Biologics Evaluation and Research (“CBER”) has the jurisdiction for reviewing such products. FDA assigned CBER as the primary center that reviewed and approved the Investigational Device Exemption (“IDE”) under which this clinical trial was conducted. On October 13, 2006, the FDA denied Cytomedix’s claim that AutoloGel™ is substantially equivalent to predicate devices, as asserted in the 510(k), and delivered to Cytomedix a Non-Substantial Equivalence (“NSE”) determination letter.
Based on the information contained in the NSE determination letter and conversations with the FDA, the Company believes that the primary grounds for rejecting the claim of substantial equivalence concern the use of bovine thrombin which is used to activate the platelet rich plasma (“PRP”) in the AutoloGel™ System. Bovine thrombin is a clotting agent derived from cows that has been used extensively on humans in surgery and other medical applications to stop bleeding. It is also used along with platelet gel therapy products that have been cleared by FDA for use in surgery. However, CBER cites published articles that contend that bovine thrombin creates antibodies that may decrease a patient’s Factor V count (a clotting agent naturally found within blood) which could cause a bleeding tendency. The analysis and clinical interpretation of the data in Cytomedix’s submission to the FDA had concluded that the data from the clinical trial does not demonstrate this complication. No statistically or clinically significant differences were noted between the PRP gel and control from baseline to endpoint laboratory shifts in hematology, clotting factors, and Factor V tests. Additionally, no clinically important changes in clotting factors that would cause concern about the effect of the PRP gel or control on Factor V activity were found during an independent medical expert review of the medical records, including clinical lab test data and concomitant medications.
3
FDA also raised concerns relating to the clinical trial and the number of protocol violations which resulted in a lack of statistical significance in the results of the “intent-to-treat” patient cohort and the subset analysis that showed full statistical significance in the results for 88% of the wounds which represented the per protocol majority wound group within the trial. The Company believed that, during face-to-face meetings with the FDA and in subsequent formal responses to FDA questions, it had addressed these concerns to the FDA’s satisfaction, although they were still listed in the NSE letter from the FDA.
The Company disagrees with the decision as expressed in the NSE determination letter and, in response to an offer made by the FDA, appealed the decision via an informal review with officials in the Office of the Center Director for CBER. The written appeal was submitted to the FDA in late December 2006 and then a face-to-face meeting was held in late January 2007 between Cytomedix, its outside experts, and various FDA personnel involved in the review process. Cytomedix presented additional expert analysis of the safety data gathered during the clinical trial, in particular that data relating to the use of bovine thrombin. In addition, Cytomedix clarified the grounds on which it is seeking marketing clearance for AutoloGel and argued the appropriateness of a reversal of the FDA’s original decision. A decision from the FDA regarding the Company’s appeal is pending.
The Company currently sells commercially AutoloGel™ process components, the AutoloGel™ Component Kit and Process Centrifuge for use in wound care and autologous wound therapy performed under the physicians’ practice of medicine doctrine. This approach represents the practice currently prevalent in the platelet gel therapy industry, both in the treatment of chronic wounds as well as the use of platelet gel therapies in the operating room in fields such as orthopedic and cardiovascular surgery. However, without FDA clearance, the Company’s ability to make claims for the AutoloGel™ System regarding its use to treat or heal wounds is limited. The Company believes this is a significant barrier to broad clinical and market acceptance of the Company’s product. It is also possible that at some point the FDA may require companies to conduct clinical trials on all specific clinical therapies and uses for which their products can be used, whether or not they make a specific labeled claim to that effect. Further, it is also possible that FDA could require companies to stop marketing platelet gel therapies until FDA clearance or approval for specific wound healing claims is obtained.
Third-Party Reimbursement
The Company believes the full market potential of AutoloGel™ cannot be achieved without broad third-party reimbursement from Medicare and commercial insurers. The Company has initiated efforts to obtain Medicare reimbursement through the Center for Medicare and Medicaid Services (“CMS”). This process involves three tracks which can be pursued simultaneously:
| § | Coverage - Coverage requires a determination by CMS that AutoloGel™ is “reasonable and necessary.” A National Non-Coverage Decision, issued in 1992 and amended in 2003, broadly disallows Medicare coverage for Autologous Blood-Derived Products for Chronic Non-Healing Wounds. This decision currently applies to AutoloGel™. The primary basis cited for this non-coverage decision was a lack of specific evidence. The Company is planning to meet with CMS to discuss a reconsideration of that decision based on data from the controlled clinical trial and other recent evidence. |
| § | Coding - Coding involves identifying an existing code or codes which aptly describe the AutoloGel™ System, or applying for new coding or modification of the definitions of existing coding to properly describe the Company’s offering. The Company is evaluating the Healthcare Common Procedure Coding System (“HCPCS”) codes, obtained through CMS and Current Procedural Terminology (“CPT”) codes, obtained through the American Medical Association, to determine the optimal reimbursement pathway for AutoloGel™. |
| § | Payment - Payment involves the establishment of a fee schedule associated with the Company’s product vis a vis the applicable codes. |
The Company has had the results of its clinical trial published in a peer-reviewed journal. The article, entitled “A Prospective, Randomized, Controlled Trial of Autologous Platelet-Rich Plasma Gel for the Treatment of Diabetic Foot Ulcers,” was published, as the feature article in the June 2006 issue of Ostomy/Wound Management (“OWM”). OWM is the premiere journal for information on wound care and the related, overlapping fields of skin care, ostomy care, incontinence care, and nutrition, and is the only peer-reviewed, multidisciplinary publication specifically targeted to the advanced wound care practitioner. The Company believes that publication in peer-reviewed journals is generally regarded as a necessary precursor to a favorable reimbursement decision from CMS and also is an important step toward building broad clinical awareness and acceptance of AutoloGel™. The Company plans to present the results of its clinical trial and other recent evidence as support for its reimbursement pursuits with CMS. Additionally, Cytomedix requisitioned a pharmaco-economic study to evaluate the cost effectiveness of the AutoloGel™ System. Such studies are performed to present scientific, demographic and economic information to justify to CMS and other payor organizations that a particular product and therapy is clinically safe and effective and cost effective with respect to its alternatives. Preliminary results of the pharmaco-economic study suggest a favorable comparison of AutoloGel™ over competing treatments in both clinical and cost effectiveness. The final report should be available sometime in the first quarter of 2007 and would also be provided to CMS.
4
While not an official precondition for a reimbursement code, the Company believes that securing Food and Drug Administration (“FDA”) clearance of the AutoloGel™ System for specific clinical indications, such as for the treatment of non-healing diabetic foot ulcers, will be heavily weighed by CMS when making its decision. Should the Company’s appeal to the FDA ultimately prove unsuccessful, the Company would need to analyze the ultimate nature of the FDA’s determination and the potential impact on the efforts to secure CMS reimbursement for the AutoloGel™ System and components.
While commercial insurers are not required to follow CMS reimbursement decisions, the Company believes they generally weigh heavily the position taken by CMS. Therefore, the results of the Company’s efforts with CMS could likely influence the degree of success the Company achieves in securing reimbursement from other third-party payers such as commercial insurers.
Should the Company be successful in its efforts to obtain reimbursement, third-party payors, including CMS, would permit payment for the AutoloGel™ System for use in certain types of chronic wounds. If this is accomplished, AutoloGel™ could then be positioned as a reimbursed alternative treatment for the estimated 6.0 million chronic wounds that occur each year in the United States.
In general, to raise the scientific awareness of the use of AutoloGel™, posters and oral presentations of the clinical trial results have been presented at multiple scientific/medical meetings including: American Diabetes Association, American Podiatric Medical Association, and the Clinical Symposium on Advances in Skin and Wound Care.
Patents and Licensing
The Company has initiated a broad based patent and licensing strategy intended to (i) enforce the rights under the Company’s patents in order to ensure that Cytomedix shareholders derive economic benefit from the Company’s intellectual property, and (ii) assist the Company in establishing a dominant market position for the AutoloGel™ System within the market for autologous growth factor products used for the treatment of chronic wounds. In 2005 and 2006, the Company identified and successfully pursued numerous companies, both small and large, that market products similar to AutoloGel™, that the Company believed were infringing or inducing infringement of its intellectual property rights. Settlements have been achieved and licenses have been granted to these companies resulting in a royalty stream for Cytomedix.
The primary license agreements are listed below:
Licensee | Date of Agreement | Date of Expiration (4) | Initial Licensing Fee | On-going Royalty Percentage (2) | |||||||||
| DePuy Spine, Inc. (1) | 3/19/2001 | 11/24/2009 | $ | 750,000 | 6.5% | ||||||||
| 3/4/2005 | |||||||||||||
| Medtronic, Inc. | 5/1/2005 | 11/24/2009 | $ | 680,000 | 7.5% on disposables | ||||||||
| 1.5% on hardware | |||||||||||||
| Harvest Technologies, Inc. | 6/30/2005 | 11/24/2009 | $ | 500,000 | 7.5% on disposables | ||||||||
| 1.5% on hardware | |||||||||||||
| Perfusion Partners, Inc. | 6/26/2005 | 11/24/2009 | $ | 250,000 | (3) | 10.0% | |||||||
| COBE Cardiovascular, Inc. | 10/7/2005 | 11/24/2009 | $ | 45,000 | 7.5% on disposables | ||||||||
| 1.5% on hardware | |||||||||||||
| SafeBlood Technologies, Inc. | 10/12/2005 | 11/24/2009 | $ | 50,000 | (3) | 8.0% to 9.0% | |||||||
| Biomet Biologics, Inc. (5) | 5/19/2006 | 11/24/2009 | $ | 2,600,000 | none | ||||||||
| CellMedix, Inc. | 11/28/2006 | 11/24/2009 | $ | 30,000 | 9.5% | ||||||||
(1) Cytomedix has two license agreements with DePuy Spine, Inc.. The original license agreement was dated March 19, 2001, amended March 3, 2005, and provides for the use of applications under Cytomedix patents in the fields of diagnostic and therapeutic spinal, neurosurgery and orthopedic surgery. The second license agreement is dated March 4, 2005, and applies to all fields not covered in the original license agreement as amended.
(2) Certain minimum royalties may apply to certain agreements and other royalty percentages may apply to future products covered under selected license agreements.
(3) Some of these amounts are payable over a period of time as defined in executed notes payable to Cytomedix.
(4) These dates reflect the expiration of the license in the U.S., which coincides with the expiration of the Knighton Patent in the U.S. In some cases, the licensing agreements applicable to territories outside the U.S. extend to the expiration of the patents in the respective foreign countries.
5
(5) The Settlement and License Agreement with Biomet Biologics, Inc. (“Biomet”) called for a $2.6 million payout from Biomet to Cytomedix. This payout took the form of $1.4 million payable upon execution of the agreement and $100,000 payable at the end of each of 12 consecutive quarters beginning with the quarter ending September 2006. These payments are not tied to any performance commitments by Cytomedix and are not dependent on Biomet sales.
The Company’s ongoing patent enforcement strategy is being conducted on a full contingency basis by the law firms Fitch, Even, Tabin & Flannery and Robert F. Coleman and Associates, both based in Chicago, Illinois.
The Company expects to incur “Cost of royalties” (consisting of royalty expense and contingent legal fees) in the range of 30-50% of on-going royalty revenues relating to these and future settlements.
The Company intends to press forward aggressively in other instances of infringement with aggressive legal and business actions to defend its intellectual property and, where possible, arrive at equitable settlements with infringers.
Non-Reimbursement Sensitive Market
The Company is also working to penetrate the segment of the national market that is not sensitive to direct reimbursement for the Company’s product. This includes capitated environments such as long-term acute care facilities, health maintenance organizations, home health agencies, as well as government agencies, (e.g. the Veterans Administration).
The Company is addressing various parts of this market via distributors, independent sales representatives, and internal sales representatives.
Sales and Marketing
Given the Company’s status with respect to marketing clearance for its AutoloGel™ System, Cytomedix continues to maintain a limited sales and marketing infrastructure for the AutoloGel™ Component Kit and Process Centrifuge. The Company predominately distributes its products through a network of commission-based internal and independent sales representatives. At December 31, 2006, the Company was represented by three internal and one independent sales representative, servicing approximately a dozen states. The Company expects to expand this representation in 2007.
Suppliers
The Company outsources manufacturing for all the components of the AutoloGelTM process. While the Company utilizes single suppliers for several components of AutoloGelTM, such components are readily available on the open market and therefore the Company believes that no dependencies exist from its current sourcing practices. The one exception is a reagent, bovine thrombin, available exclusively through King Pharmaceuticals.
Competition
Wound care products can be categorized into 3 general areas: passive, interactive, and active.
| · | Passive products -- such as gauze and bandages, cover the wound to protect it. |
| · | Interactive products -- attempt to optimize the wound environment so it is more conducive for the body to enact the innate healing process. The wound care world recognizes that moist wound healing is more effective for cellular growth than dry wound healing, however excessive moisture can be detrimental to healing. In addition, wounds need to be free of infection, have adequate perfusion and tissue oxygenation, and reduced pressure. There are hundreds of wound dressings on the market, some provide a long-term moist wound environment, others absorb large amounts of exudates, and some provide topical antimicrobial agents. In addition, there are multiple devices that attempt to assist with creating the optimal wound environment. However, as a whole, none of them are positioned to actively direct cellular growth. |
| · | Active wound products -- directly stimulate cellular growth and migration in the wound area. Growth factor products, such as AutoloGel™, are a predominant product in this category. Science has documented that multiple growth factors cause cellular growth and migration to actively grow granulation tissue, capillaries, and epithelium. Tissue engineered grafts could also fall into this category because they contain live cells and secondarily, may have some growth factors in the tissue. |
Thus, when identifying competitors, each product can be categorized in the above breakdown. Passive products are not a competitor for AutoloGel™. While some of the interactive products can be competitors, others can be complimentary to AutoloGel™. The other active products could be categorized as the major competitors.
6
The major competitors are other platelet gel companies, many of whom have licensing agreements with Cytomedix. To date, these companies are selling platelet gel mostly into the surgical market for dental, plastic, orthopedic, and general surgery purposes but may also try to sell into the wound care market. When compared to the other platelet gel companies, Cytomedix’s AutoloGel™ System has the smallest, most portable centrifuge with the fastest spin time (1.5 minutes compared to 13-20 minutes). This makes it possible to use in a greater variety of health care settings, i.e. hospital, outpatient clinics, physicians offices, or long term care, long term acute care, and home health settings. In addition, it is a user-friendly system so multiple health care providers can process the gel, rather than specialty technicians. Competitors’ systems generally require a larger blood draw, more detailed processing steps, and a longer spin time. While competitors claim a larger growth factor and platelet count than at baseline, no studies exist that prove this is efficacious. To date, Cytomedix’s AutoloGel™ System is the only platelet gel system that has completed a prospective, randomized, controlled trial in humans. The Company believes this trial demonstrated both the safety and efficacy of the AutoloGel™ System.
Regranex, a prescription cream marketed by Ethicon, a division of Johnson & Johnson, Inc. ("J&J"), contains a single recombinant growth factor. Having been introduced after lengthy clinical trials several years ago, its revenues, based on the Company’s best estimate, have grown significantly. Cytomedix perceives the single growth factor Regranex as a less effective method of healing chronic wounds in comparison with autologous multiple growth factors. While Cytomedix acknowledges the success of the Regranex product in the marketplace, an excellent opportunity exists to capture market share from this product once reimbursement is available for AutoloGel™. A recent CMS (Medicare) decision disallowed coverage for Regranex because it is self-administered.
The tissue-engineered products have changed extensively lately. Smith and Nephew, the manufacturer of Dermagraft, has taken the product off the market recently and has subsequently sold the technology to Advanced BioHealing. Ortec International, Inc.is continuing to conduct clinical trials on its product Orcel. Apligraf, manufactured by Organogenesis, is the only tissue engineered product on the market at this time.
The major competitor in the interactive area is a device called Vacuum Assisted Closure (“V.A.C.”) system produced by Kinetic Concepts, Inc. (“KCI”). During the year ended December 31, 2005, KCI worldwide revenues due to V.A.C. sales and rentals were reported as $908 million. Several of the sites that have used the V.A.C. system have now tried AutoloGel™. It has been reported to the Company by several of these sites that AutoloGel™ was very competitive with the V.A.C. and may even be better in both clinical and cost effectiveness. This, however, was based on individual case reports and experience of physicians rather than any rigorous comparative studies. Yet the V.A.C's established position is substantially CMS reimbursed, which provides for a substantial current economic competitive advantage.
Intellectual Property Rights
Cytomedix regards its patents, trademarks, trade secrets, and other intellectual property (collectively, the “Intellectual Property Assets”) as critical to its success. Cytomedix relies on a combination of patents, trademarks, and trade secret and copyright laws, as well as confidentiality procedures, contractual provisions, and other similar measures, to establish and protect its Intellectual Property Assets. Cytomedix has in the past several years filed numerous patent applications worldwide seeking protection of its technologies. Cytomedix owns eight U.S. patents (including U.S. Patent No. 5,165,938 (the “Knighton Patent”) and U.S. Patent No. 6,303,112 (the “Worden Patent”)), various corresponding foreign patents, and various trademarks. Cytomedix has received, filed, or is in the process of filing trademarks for the names “Cytomedix,” “AutoloGel”, and a few variants thereof. In addition, Cytomedix has numerous pending trademark applications and foreign patent applications involving enriched platelet wound healant, platelet derived wound healant, angiogenic peptides, and anti-inflammatory peptides.
To prevent disclosure of its trade secrets, Cytomedix restricts access to its proprietary information. All of its employees, consultants, and other persons with access to Cytomedix's proprietary information execute confidentiality agreements with Cytomedix. Cytomedix has also pursued litigation against those persons believed to be infringing on the Company's Intellectual Property Assets seeking both damages and injunctive relief.
Despite these efforts, Cytomedix may not be able to prevent misappropriation of its technology or deter others from developing similar technology in the future. Furthermore, policing the unauthorized use of its Intellectual Property Assets is difficult. Litigation necessary to enforce Cytomedix's Intellectual Property Assets could result in substantial costs and diversion of resources.
The Company is party to certain royalty agreements relating to its intellectual property under which it pays certain fees. See Note 5 to the Financial Statements.
7
Government Regulation
Devices that the Company manufactures and distributes are subject to regulations by the Food and Drug Administration, including record-keeping requirements, good manufacturing practices and mandatory reporting of certain adverse experiences resulting from use of the devices, and certain state agencies. Labeling and promotional activities are also subject to regulation by the FDA and the Federal Trade Commission, in certain circumstances. Current FDA enforcement policy prohibits the marketing of approved medical devices for unapproved uses and the agency scrutinizes the labeling and advertising of medical devices to ensure that unapproved uses are not promoted.
Before a new medical device can be introduced to the market, the manufacturer must generally obtain FDA clearance or approval. In the United States, medical devices are classified into one of three classes - Class I, II or III. The controls applied by the FDA to the different classifications are those believed by the FDA to be necessary to provide reasonable assurance that the device is safe and effective. Class I devices are non-critical products that FDA believes can be adequately regulated by “general controls” that include provisions relating to labeling, manufacturer registration, defect notification, records and reports, and good manufacturing practices (“GMP”) based on the FDA’s Quality Systems Regulations. Most Class I devices are exempt from pre-market notification and some are also exempt from GMP requirements. Class II devices are products for which the general controls of Class I devices, by themselves, are not sufficient to assure safety and effectiveness and, therefore, require special controls. Additional special controls for Class II devices include performance standards, post-market surveillance patient registries, and the use of FDA guidelines. Standards may include both design and performance requirements. Class III devices have the most restrictive controls and require pre-market approval by the FDA. Generally, Class III devices are limited to life-sustaining, life-supporting or implantable devices. The FDA inspects medical device manufacturers and has a broad authority to order recalls of medical devices, to seize non-complying medical devices, and to criminally prosecute violators.
Section 510(k) of the Federal Food, Drug and Cosmetic Act requires individuals or companies manufacturing most medical devices intended for human use to file a notice with the FDA at least ninety days before intending to introduce the device into the market. This notice, commonly referred to as a 510(k), must identify the type of classified device into which the product falls, the class of that type, and a specific product already being marketed or cleared by FDA and to which the product is “substantially equivalent.” In some instances, the 510(k) must include data from human clinical studies in order to establish “substantial equivalence.” The FDA must agree with the claim of “substantial equivalence” before the device can be marketed. The statutory time frame for clearance of a 510(k) is 90 days, though it often takes longer.
If a product is Class III and does not qualify for the 510(k) process, then the FDA must approve a pre-market approval (“PMA”) application before marketing can begin. PMA applications must demonstrate, among other factors, that the device in question is safe and effective. Obtaining a PMA application approval can sometimes take several years, depending upon the complexity of the issues involved with the device. The statutory time frame for the review of a PMA by the FDA is 180 days and many devices are reviewed and approved within that time frame or within a few months afterward. Marketing approval based on a PMA is generally a longer process than the 510(k) clearance process that is typically obtained in comparatively less time.
Government Approval
Cytomedix has sought to ensure compliance with the FDA regulations and policies for medical devices and, specifically, platelet gel therapies.
The Company currently markets the AutoloGel™ Process Centrifuge,the AutoloGel™ Component Kit, and certain commercially available reagents (i.e. calcium chloride, ascorbic acid, and bovine thrombin). Each component is a legally-marketed product that either has been cleared by FDA for marketing or is exempt from pre-market notification and clearance. The AutoloGel™ Centrifuge, when used with the AutoloGel™ Component Kit and certain reagents, form the basis for the AutoloGel process and are used for wound care and for treating chronic wound, including diabetic ulcers, at the physician’s discretion. The Federal Food, Drug and Cosmetic Act does not authorize the FDA to limit or interfere with the “physician's practice of medicine” and use of legally-marketed devices for any condition or disease within a legitimate doctor-patient relationship as long as no specific claims are made for the product.
In 2003, the Company conceptualized marketing an AutoloGel™ System, consisting of a centrifuge, a component kit, and reagents, for specific indications such as diabetic, pressure, and venous ulcers.
During 2003, the Company made a business decision to undertake a prospective, randomized, blinded, controlled trial for the AutoloGel™ System. The objective of the trial was to demonstrate safety and efficacy of the AutoloGel™ System for treating diabetic foot ulcers to the scientific and reimbursement community, as well as to the FDA in order to obtain the agency’s marketing clearance of the AutoloGel™ System. In making this decision, the Company subjected itself to increased FDA oversight and its regulations governing the investigational use of medical devices, codified at 21 C.F.R. Part 812. To this end, the Company submitted an “Investigational Device Exemption” (“IDE”) application to the FDA under these rules and obtained approval on March 5, 2004, thus allowing the Company to begin its clinical trial.
8
Once the study was completed and clinical results analyzed, the Company submitted a 510(k) requesting FDA’s clearance of the AutoloGel™ System in January 2006, as discussed above, under the caption Clinical Trial and FDA Clearance.
Fraud and Abuse Laws
The Company may also be indirectly subject to federal and state physician self referral laws. Federal physician self-referral legislation (commonly known as the “Stark Law”) prohibits, subject to certain exceptions, physician referrals of Medicare and Medicaid patients to an entity providing certain “designated health services” if the physician or an immediate family member has any financial relationship with the entity. A person who engages in a scheme to circumvent the Stark Law's referral prohibition may be fined up to $100,000 for each such arrangement or scheme. The penalties for violating the Stark Law also include civil monetary penalties of up to $15,000 per referral and possible exclusion from federal health care programs such as Medicare and Medicaid. The Stark Law also prohibits the entity receiving the referral from billing any good or service furnished pursuant to an unlawful referral, and any person collecting any amounts in connection with an unlawful referral is obligated to refund such amounts. Various states have corollary laws to the Stark Law, including laws that require physicians to disclose any financial interest they may have with a health care provider to their patients when referring patients to that provider. Both the scope and exception for such laws vary from state to state.
The Company may also be subject to federal and state anti-kickback laws. Section 1128B (b) of the Social Security Act, commonly referred to as the Anti-Kickback Law, prohibits persons from knowingly and willfully soliciting, receiving, offering or providing remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending, or arranging for a good or service, for which payment may be made under a federal health care program such as Medicare and Medicaid. The Anti-Kickback Law is broad, and it prohibits many arrangements and practices that are otherwise lawful in businesses outside of the health care industry. The U.S. Department of Health and Human Services (“DHHS”) has issued regulations, commonly known as safe harbors that set forth certain provisions which, if fully met, will assure health care providers and other parties that they will not be prosecuted under the federal Anti-Kickback Law. Although full compliance with these provisions ensures against prosecution under the Anti-Kickback Law, the failure of a transaction or arrangement to fit within a specific safe harbor does not necessarily mean that the transaction or arrangement is illegal or that prosecution under the federal Anti-Kickback Law will be pursued. The penalties for violating the Anti-Kickback Law include imprisonment for up to five years, fines of up to $250,000 per violation for individuals and up to $500,000 per violation for companies and possible exclusion from federal health care programs. Many states have adopted laws similar to the federal Anti-Kickback Law, and some of these state prohibitions apply to patients for health care services reimbursed by any source, not only federal health care programs such as Medicare and Medicaid.
In addition, there are two other health care fraud laws to which the Company may be subject, one which prohibits knowingly and willfully executing or attempting to execute a scheme or artifice to defraud any health care benefit program, including private payers (“fraud on a health benefit plan”) and one which prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement or representation in connection with the delivery of or payment for health care benefits, items or services. These laws apply to any health benefit plan, not just Medicare and Medicaid.
The Company may also be subject to other laws which prohibit submitting claims for payment or causing such claims to be submitted that are false. Violation of these false claims statutes may lead to civil money penalties, criminal fines and imprisonment, and/or exclusion from participation in Medicare, Medicaid and other federally funded state health programs. These statutes include the federal False Claims Act, which prohibits the knowing filing of a false claim (or causing the submission of a false claim) or the knowing use of false statements to obtain payment from the U.S. federal government. When an entity is determined to have violated the False Claims Act, it must pay three times the actual damages sustained by the government, plus mandatory civil penalties of between $5,500 and $11,000 for each separate false claim. Suits filed under the False Claims Act can be brought by an individual on behalf of the government (a “qui tam action”). Such individuals (known as “qui tam relators”) may share in the amounts paid by the entity to the government in fines or settlement. In addition certain states have enacted laws modeled after the False Claims Act. “Qui tam” actions have increased significantly in recent years causing greater numbers of health care companies to have to defend false claim actions, pay fines or be excluded from the Medicare, Medicaid or other federal or state health care programs as a result of an investigation arising out of such action.
Several states also have referral, fee splitting and other similar laws that may restrict the payment or receipt of remuneration in connection with the purchase or rental of medical equipment and supplies. State laws vary in scope and have been infrequently interpreted by courts and regulatory agencies, but may apply to all health care products and services, regardless of whether Medicaid or Medicare funds are involved.
9
Research and Development
The Company is currently focusing its limited resources on broadly commercializing AutoloGelTM. It therefore expends only very minor amounts on research and development activities (“R&D”). The Company currently focuses its R&D activities on the improvement of its current product offering, but, in the future, intends to develop the technology underlying its broader patent portfolio.
Employees
At December 31, 2006, the Company had twelve full-time employees. These include three executive officers, Dr. Kshitij Mohan as Chief Executive Officer, Mr. Andrew S. Maslan as Chief Financial Officer and Ms. Carelyn P. Fylling as Vice President of Professional Services. The remaining personnel consist of sales and marketing, clinical, accounting, and regulatory professionals.
AVAILABLE INFORMATION
Cytomedix files periodic reports and all amendments thereto pursuant to Section 13(a) or 15(d) of the Securities and Exchange Act of 1934. These reports are available, free of charge, through the Company’s website at www.cytomedix.com.
ITEM 1A. RISK FACTORS
Cytomedix cautions the readers not to place undue reliance on any forward-looking statements, which are based on certain assumptions and expectations that may or may not be valid or actually occur. The risk factors that follow may cause actual results to differ materially from those expressed or implied by any forward-looking statement. The risks described below are not to be deemed an exhaustive list of all potential risks.
The FDA Denied the Company’s Claims in its 510(k) Pre-Market Notification and the Company’s Appeal May Not Be Successful
The FDA denied the Company’s claims in its 510(k). The Company has appealed this decision and currently awaits a final ruling from the FDA. There is no assurance that the Company’s efforts to have the original decision reversed or amended to the Company’s satisfaction will be successful. A lack of FDA clearance may make it more difficult to obtain reimbursement codes and/or adversely affect the Company’s ability to implement a significant portion of its business plan. Specifically, the Company may be unable to obtain a significant share of the chronic wound care market. Even with FDA clearance, the Company can provide no assurance that it will be able to obtain Medicare or other third party reimbursement.
The Company Has Limited Sources of Working Capital
Because the Company was in bankruptcy in 2002, the Company will not be able to obtain debt financing. All working capital required to implement the Company’s business plan will be provided by funds obtained through offerings of its equity securities, and revenues generated by the Company. No assurance can be given that the Company will have revenues sufficient to support and sustain its operations through 2007.
If the Company does not have sufficient working capital and is unable to generate revenues or raise additional funds, the following may occur: delaying the completion of the Company’s current business plan or significantly reducing the scope of the business plan; delaying some of its development and clinical or marketing testing; delaying its plans to pursue government regulatory and reimbursement approval and/or clearance for its wound treatment technologies; postponing the hiring of new personnel; or, in an extreme situation, ceasing operations.
The Company Has a History of Losses
The Company has a history of losses, is not currently profitable, and expects to incur substantial losses and negative operating cash flows for the foreseeable future. The Company may never achieve or maintain profitability. The Company will need to generate significant revenues to achieve and maintain profitability. The Company cannot guarantee that it will be able to generate these revenues, and it may never achieve profitability.
10
The Company Has a Short Operating History and Limited Operating Experience
The Company must be evaluated in light of the uncertainties and complexities affecting an early stage biotechnology company. The Company has only recently begun to implement its current business plan. Thus, the Company has a very limited operating history. Continued operating losses, together with the risks associated with the Company’s ability to gain new customers for its product offerings may have a material adverse effect on the Company’s liquidity. The Company may also be forced to respond to unforeseen difficulties, such as decreasing demand for its products and services, regulatory requirements and unanticipated market pressures.
Since emerging from bankruptcy and continuing through today, the Company is developing a business model that includes protecting its patent position, addressing its third-party reimbursement issues, and developing a sales and marketing program. There can be no assurance that its business model in its current form can accomplish the Company’s stated goals.
The Company’s Intellectual Property Assets Are Critical to Its Success
The Company regards its patents, trademarks, trade secrets, and other intellectual property assets as critical to its success. The Company relies on a combination of patents, trademarks, and trade secret and copyright laws, as well as confidentiality procedures, contractual provisions, and other similar measures, to establish and protect its intellectual property. The Company attempts to prevent disclosure of its trade secrets by restricting access to sensitive information and requiring employees, consultants, and other persons with access to the Company’s sensitive information to sign confidentiality agreements. Despite these efforts, the Company may not be able to prevent misappropriation of its technology or deter others from developing similar technology in the future. Furthermore, policing the unauthorized use of its intellectual property assets is difficult and expensive. Litigation has been necessary in the past and may likely be necessary in the future in order to protect the Company’s intellectual property assets. Litigation could result in substantial costs and diversion of resources. The Company cannot guarantee that it will be successful in any litigation matter relating to its intellectual property assets. Continuing litigation or other challenges could result in one or more of its patents being declared invalid. In such a case, any royalty revenues from the affected patents would be adversely affected although the Company may still be able to continue to develop and market its products.
The Company’s patent covering the specific gel formulation that is applied as part of the AutoloGel™ System (the “Worden Patent”) expires no earlier than February 2019. The Company’s U.S. Knighton Patent (which is the subject of license agreements between the Company and Medtronic, Inc., DePuy Spine, Inc., Biomet Biologics, Inc., COBE Cardiovascular, Inc., and Harvest Technologies Corporation, among others) expires in November 2009. The Company is pursuing a strategy to obtain FDA clearance and CMS reimbursement, but there can be no assurance that the Company will be able to establish such a significantly increased share of the wound care market prior to the expiration of the U.S. Knighton Patent in 2009, after which the Company may be more vulnerable to competitive factors because third parties will not then need a license from the Company to perform the methods claimed in the Knighton Patent.
The AutoloGel™ Components are Subject to Governmental Regulation
The Company’s success is also impacted by factors outside of the Company’s control. The Company’s current therapies may be subject to extensive regulation by numerous governmental authorities in the United States, both federal and state, and in foreign countries by various regulatory agencies.
Specifically, the Company’s devices are subject to regulation by the FDA and state regulatory agencies. The FDA regulates drugs, medical devices and biologics that move in interstate commerce and requires that such products receive pre-marketing approval based on evidence of safety and efficacy. The regulations of government health ministries in foreign countries are analogous to those of the FDA in both application and scope. In addition, any change in current regulatory interpretations by, or positions of, state regulatory officials where the AutoloGel™ process is practiced could materially and adversely affect the Company’s ability to sell products in those states.
The FDA could require the Company to stop selling the components used to prepare AutoloGel™ until it obtains clearance or approval of a specific wound healing claim. While the Company believes that all of said components are legally marketed, the FDA could take the position that the Company cannot market the AutoloGel™ Component Kit or Process Centrifuge for wound healing until the Company has a specific approval or clearance to do so from the FDA.
Further, as the Company expands and offers additional products in the United States and in foreign countries, approval from the FDA and comparable foreign regulatory authorities prior to introduction of any such products into the market may be required. The Company has no assurance that it will be able to obtain all necessary approvals from the FDA or comparable regulatory authorities in foreign countries for these products. Failure to obtain the required approvals would have a material adverse impact on the Company’s business and financial condition.
11
Compliance with FDA and other governmental requirements imposes significant costs and expenses. Further, the Company’s failure to comply with these requirements could result in sanctions, limitations on promotional or other business activities, or other adverse effects on the Company’s business. Further, recent efforts to control healthcare costs could negatively effect demand for the Company’s products and services.
The Company Could Be Adversely Affected if Customers Cannot Obtain Reimbursement
The AutoloGel™ Component Kit and Process Centrifuge are marketed to healthcare providers. Some of these providers, in turn, seek reimbursement from third-party payers such as Medicare, Medicaid, and other private insurers. Many foreign countries also have comprehensive government managed healthcare programs that provide reimbursement for healthcare products. Under such healthcare systems, reimbursement is often a determining factor in predicting a product’s success, with some physicians and patients strongly favoring only those products for which they will be reimbursed.
In order to achieve a viable reimbursement pathway for the AutoloGel™ process components, the Company has conducted a prospective, randomized, blinded, controlled, multi-site clinical trial as approved by the FDA to provide the necessary data as required by CMS, formerly known as the Healthcare Financing Agency. In addition, the 2003 CMS non-coverage decision for “Autologous Blood-Derived Products for the Treatment of Chronic Wounds”, which builds on the 1992 HCFA ruling may have to be dismissed or a carve-out would need to be created in order to make national coverage by Medicare possible. The Company cannot assure that its efforts in this area will be successful and therefore, a significant obstacle to broad third-party reimbursement may remain. Further, even if the Non-Coverage Decision is reversed, the Company cannot guarantee that third-party payers will elect to reimburse treatments using the Company’s products or processes or, if such reimbursement is approved, that the level of reimbursement granted will be sufficient to cover the cost of the product or process to the physician or to the patient.
Healthcare providers’ inability to obtain third-party reimbursement for the treatment could have an adverse effect on the Company’s success.
Royalty Revenues Are Unpredictable
While the Company currently has several primary licensing agreements that are expected to generate on-going royalty revenues, the Company cannot currently reasonably predict the magnitude of those revenues. Because Cytomedix’s licensing activities are recent, it is premature to predict the resulting royalty streams from these licensing agreements. Furthermore, royalty streams from these agreements are entirely dependent on the sales of its licensees and are therefore outside the control of Cytomedix. Past levels of royalty revenues from these agreements are not necessarily an indication of future activity.
The Success of the AutoloGel™ System Is Dependent on Acceptance by the Medical Community
The commercial success of the Company’s products and processes will depend upon the medical community and patients accepting the therapies as safe and effective. If the medical community and patients do not ultimately accept the therapies as safe and effective, the Company’s ability to sell the products and processes will be materially and adversely affected. While acceptance by the medical community may be fostered by broad evaluation via peer-reviewed literature, the Company may not have the resources to facilitate sufficient publication.
The Company May Be Unable to Attract and Retain Key Personnel
The future success of the Company depends on the ability to attract, retain and motivate highly skilled management, including sales representatives. The Company has retained a team of highly qualified officers and consultants, but the Company cannot provide assurance that it will be able to successfully integrate these officers and consultants into its operations, retain all of them, or be successful in recruiting additional personnel as needed. The Company’s inability to do so will materially and adversely affect the business prospects, operating results and financial condition.
The Company’s ability to maintain and provide additional services to its existing customers depends upon its ability to hire and retain business development and scientific and technical personnel with the skills necessary to keep pace with continuing changes in cellular therapy technologies. Competition for such personnel is intense; the Company competes with pharmaceutical, biotechnology and healthcare companies. The Company’s inability to hire additional qualified personnel may lead to higher recruiting, relocation and compensation costs for such personnel. These increased costs may reduce the Company’s profit margins or make hiring new personnel impractical.
12
Legislative and Administrative Action May Have an Adverse Effect on the Company
Political, economic and regulatory influences are subjecting the health care industry in the United States to fundamental change. The Company cannot predict what other legislation relating to its business or to the health care industry may be enacted, including legislation relating to third-party reimbursement, or what effect such legislation may have on the Company’s business, prospects, operating results and financial condition. The Company expects federal and state legislators to continue to review and assess alternative health care delivery and payment systems and possibly adopt legislation affecting fundamental changes in the health care delivery system. Such laws may contain provisions that may change the operating environment for its targeted customers including hospitals and managed care organizations.
Health care industry participants may react to such legislation by curtailing or deferring expenditures and initiatives, including those relating to the Company’s products. Future legislation could result in modifications to the existing public and private health care insurance systems that would have a material adverse effect on the reimbursement policies discussed above.
The Company Could Be Affected by Malpractice Claims
Providing medical care entails an inherent risk of professional malpractice and other claims. The Company does not control or direct the practice of medicine by physicians or health care providers who use the products and does not assume responsibility for compliance with regulatory and other requirements directly applicable to physicians. The Company cannot guarantee that claims, suits or complaints relating to the use of the AutoloGel™ components and treatment administered by physicians will not be asserted against the Company in the future.
The production, marketing and sale, and use of the AutoloGel™ Component Kit and Process Centrifuge entail risks that product liability claims will be asserted against the Company. These risks cannot be eliminated, and the Company could be held liable for any damages that result from adverse reactions or infectious disease transmission. Such liability could materially and adversely affect the Company’s business, prospects, operating results and financial condition.
The Company currently maintains professional and product liability insurance coverage, but the Company cannot give assurance that the coverage limits of this insurance would be adequate to protect against all potential claims. The Company cannot guarantee that it will be able to obtain or maintain professional and product liability insurance in the future on acceptable terms or with adequate coverage against potential liabilities.
AutoloGel™ Has Existing Competition in the Marketplace
In the market for biotechnology products, the Company faces competition from pharmaceutical companies, biopharmaceutical companies and other competitors. Other companies have developed or are developing products that may be in direct competition with the AutoloGel™ process. Biotechnology development projects are characterized by intense competition. Thus, the Company cannot assure any investor that it will be the first to the market with any newly developed products or that it will successfully be able to market these products. If the Company is not able to participate and compete in the cellular therapy market, the Company’s financial condition will be materially and adversely affected. The Company cannot guarantee that it will be able to compete effectively against such companies in the future. Many of these companies have substantially greater capital resources, larger marketing staffs and more experience in commercializing products. Recently developed technologies, or technologies that may be developed in the future, may be the basis for developments that will compete with the Company’s products.
Risks Related to the Company’s Common Stock
The average daily trading volume in Cytomedix Common stock is relatively low. As long as this condition continues, it could be difficult or impossible to sell a significant number of shares of Common stock at any particular time at the market prices prevailing immediately before such shares are offered. In addition, sales of substantial amounts of Common stock could lower the prevailing market price of the Company’s Common stock. This would limit or perhaps prevent the Company’s ability to raise capital through the sale of securities. Additionally, the Company has significant numbers of outstanding warrants and options that, if exercised and sold, could put additional downward pressure on the Common stock price.
The Company is Subject to Anti-Takeover Provisions and Laws.
Provisions in Cytomedix’s Restated Certificate of Incorporation and Restated Bylaws and applicable provisions of the Delaware General Corporation Law may make it more difficult for a third party to acquire control of the Company without the approval of the board of directors. These provisions may make it more difficult or expensive for a third party to acquire a majority of the Company’s outstanding voting Common stock or delay, prevent or deter a merger, acquisition, tender offer or proxy contest, which may negatively affect the Common stock price.
13
Purchases of the Company’s Common Stock Are Subject to the SEC's Penny Stock Rules.
Generally, any non-NASDAQ equity security that has a market price of less than $5.00 per share is defined as a Penny Stock. Penny Stocks are subject to special rules and regulations under the Securities Exchange Act of 1934. These rules require additional disclosure by broker-dealers in connection with any trades involving Penny Stock. Cytomedix Common stock is currently defined as a Penny Stock, and the Company is uncertain if the market price of its common stock will ever be above $5.00 per share. As a result of its characterization as a Penny Stock, the market liquidity for the Company’s Common stock may be adversely affected by the Penny Stock rules and regulations. This could restrict an investor's ability to sell the common stock in a secondary market. The rules governing Penny Stock require the delivery, prior to any Penny Stock transaction, of a disclosure schedule explaining the Penny Stock market and the risks associated therewith, and impose various sales practice requirements on broker-dealers who sell Penny Stocks to persons other than established customers and accredited investors (generally defined as an investor with a net worth in excess of $1,000,000 or annual income exceeding $200,000, $300,000 together with a spouse). For these types of transactions, the broker-dealer must make a special suitability determination for the purchaser and have received the purchaser's written consent to the transaction prior to sale. The broker-dealer also must disclose the commissions payable to the broker-dealer, current bid and offer quotations for the Penny Stock and, if the broker-dealer is the sole market-maker, the broker-dealer must disclose this fact and the broker-dealer's presumed control over the market. Such information must be provided to the customer orally or in writing prior to effecting the transaction and in writing before or with the customer confirmation. Monthly statements must be sent disclosing recent price information for the Penny Stock held in the account and information on the limited market in Penny Stock. The additional burdens imposed on broker-dealers may discourage them from effecting transactions in the Company’s Common stock, which could severely limit the liquidity of the Common stock and the ability of shareholders to sell the Common stock in the secondary market.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
The Company does not own any real property and does not intend to invest in any real property.
The Company’s principal executive offices are located in Rockville, Maryland. Cytomedix occupies facilities consisting of 3,100 square feet under an operating lease expiring July 31, 2008, subject to an early termination option available to Cytomedix. See Note 16 to the Financial Statements.
ITEM 3. LEGAL PROCEEDINGS
At present, the Company is not engaged in or the subject of any legal proceedings.
ITEM 4. SUBMISSION OF MATTERS TO A VOTE OF SECURITY HOLDERS
The Company held its annual meeting of shareholders on November 3, 2006, at the Company’s offices in Rockville, Maryland. At the meeting, the shareholders re-elected James S. Benson, David P. Crews, Arun K. Deva, David F. Drohan, Mark T. McLoughlin, and Kshitij Mohan as Directors to hold office until the next annual meeting of shareholders and until their successors are duly elected. A summary of votes cast follows below:
14
NOMINEE | VOTES FOR | VOTES WITHHELD | ABSTENTIONS* | |||||||
| James S. Benson | 21,816,240 | 196,940 | — | |||||||
| David P. Crews | 21,815,308 | 197,872 | — | |||||||
| Arun K. Deva | 21,810,640 | 202,540 | — | |||||||
| David F. Drohan | 21,815,308 | 197,872 | — | |||||||
| Mark T. McLoughlin | 21,816,240 | 196,940 | — | |||||||
| Dr. Kshitj Mohan | 21,794,443 | 218,737 | — |
* Pursuant to the terms of the Proxy Statement, proxies received were voted, unless authority was withheld, in favor of the election of the six nominees.
Shareholders also voted to ratify the appointment of L J Soldinger Associates, LLC as the Company’s independent registered accountant for the fiscal year ending December 31, 2006 with 21,969,431 votes for, 24,235 votes against, and 19,514 abstentions.
Shareholders also voted to ratify an amendment to the Long-Term Incentive Plan making Awards available representing up to 5,000,000 shares of Common stock with 6,056,251 votes for, 2,401,658 votes against, and 86,838 abstentions.
Further information regarding the meeting and the proposals submitted to a vote of the shareholders may be found in the Company’s definitive proxy statement filed with the Securities and Exchange Commission on September 22, 2006.
15
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITES
Since June 2005, the Company’s Common stock has been listed on the American Stock Exchange under the symbol GTF. Prior to this listing the Company’s Common stock was traded in the Over-the-Counter (“OTC”) market and quoted on the OTC bulletin board under the symbol CYME. Set forth below are the high and low closing sale prices for the Common stock for each quarter since the quarter beginning January 1, 2004, as reported by NASDAQ and AMEX as appropriate. The prices prior to June 30, 2005 are over-the-counter market quotations and reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions.
Quarter ended | High | Low | |||||
| December 31, 2006 | $ | 2.76 | $ | 0.91 | |||
| September 30, 2006 | $ | 3.34 | $ | 2.60 | |||
| June 30, 2006 | $ | 3.20 | $ | 2.25 | |||
| March 31, 2006 | $ | 2.90 | $ | 2.23 | |||
| December 31, 2005 | $ | 3.36 | $ | 2.01 | |||
| September 30, 2005 | $ | 6.85 | $ | 1.68 | |||
| June 30, 2005 | $ | 5.07 | $ | 3.12 | |||
| March 31, 2005 | $ | 3.50 | $ | 2.35 | |||
There were approximately 658 shareholders of record of Common stock as of February 15, 2007.
Cytomedix did not pay dividends to holders of Common stock in 2006 or 2005. The Company is prohibited from declaring dividends on Common stock if any dividends are due on shares of Series A, B, or C Convertible Preferred stock. If there are no unpaid dividends on shares of Series A, B, or C Convertible Preferred stock, any decision to pay cash dividends on Common stock will depend on the Company’s ability to generate earnings, need for capital, and overall financial condition, and other factors the Board deems relevant. Cytomedix does not anticipate paying cash dividends on Common stock in the foreseeable future, but instead will retain any earnings for reinvestment in the business.
RECENT SALES OF UNREGISTERED SECURITIES
The Company issued 1,062,500 shares of Common stock during the fourth quarter of 2006. The following table lists the sources of and the proceeds from those issuances:
Source | # of Shares | Total Exercise Price | ||||||
| Exercise of unit offering warrants | 287,500 | $ | 431,250 | |||||
| Exercise of other warrants (1) | 775,000 | $ | 775,000 | |||||
| Totals | 1,062,500 | $ | 1,206,250 | |||||
| (1) | These warrants reflect consultant warrants held by one party. Upon exercise, the Company accepted $75,000 in cash and a Negotiable Term Promissory Note and related Security Agreement (the “Note”). The Note, which was amended in February 2007, provides for the remaining exercise proceeds to be delivered to the Company in installment payments ending on April 30, 2007. The Note bears interest on the outstanding balance at 6% per year. As of February 15, 2007, the maker of the Note was current in making all required principal and interest payments and the principal balance remaining was $426,250. |
The Company has used the cash proceeds from these issuances for general corporate purposes. All shares were issued in private offerings exempt from registration pursuant to Section 4(2) of the Securities Act.
See Note 11 to the Financial Statements for further information on the Company’s capital structure.
16
PERFORMANCE GRAPH
Below is a line graph comparing total cumulative return on an investment of $100 invested on January 27, 2003 in: (i) the Company’s Common Stock, (ii) an industry index, namely the AMEX Biotechnology Index, and (iii) a broad market index, namely the Russell Microcap Index. All values assume reinvestment of the full amount of all dividends, where applicable, and are calculated as of the last trading day of each fiscal year.
Comparison of 47 Month Cumulative Total ReturnAmong Cytomedix, Inc., Amex Biotechnology Index,
and Russell Microcap Index
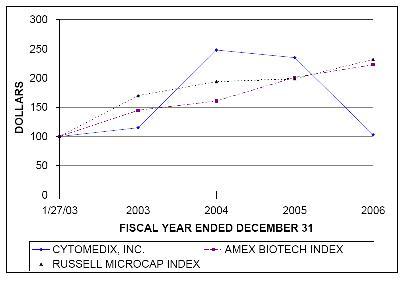
ITEM 6. SELECTED FINANCIAL DATA
2006 | 2005 | 2004 | 2003 | 2002* | ||||||||||||
| Revenues | $ | 1,948,155 | $ | 1,514,425 | $ | 1,145,591 | $ | 1,086,923 | $ | 666,183 | ||||||
| Loss from operations | $ | (4,159,264 | ) | $ | (7,446,134 | ) | $ | (8,188,360 | ) | $ | (4,131,705 | ) | $ | (2,018,465 | ) | |
| Net loss | $ | (2,262,956 | ) | $ | (6,503,485 | ) | $ | (8,139,326 | ) | $ | (4,124,889 | ) | $ | (2,016,961 | ) | |
| Basic and diluted net loss per common share | $ | (0.08 | ) | $ | (0.27 | ) | $ | (0.62 | ) | $ | (0.37 | ) | $ | (0.22 | ) | |
| Total assets | $ | 10,233,774 | $ | 7,877,917 | $ | 8,186,472 | $ | 5,740,920 | $ | 6,153,709 | ||||||
| Long-term obligations and redeemable preferred stock | $ | 376,520 | $ | 263,787 | $ | 356,630 | $ | 438,081 | $ | 519,507 | ||||||
| Cash dividends declared per common share | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||
* On July 1, 2002, the Company emerged from bankruptcy under a plan of reorganization. The 2002 periodic information included in the table above reflects results of the six months ended December 31, 2002.
ITEM 7. MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Management's Discussion and Analysis and other parts of this report contain forward-looking statements that involve risks and uncertainties. All forward-looking statements included in this report are based on information available to the Company on the date hereof, and the Company assumes no obligation to update any such forward-looking statements. The following should be read in conjunction with the audited financial statements and the notes thereto included elsewhere herein.
17
In 2006, Cytomedix submitted its 510(k) to, received a denial from, and prepared its appeal to the FDA for specific indications for its AutoloGelTM System. The Company realized a 29% increase in revenues driven by increased licensing royalties. The Company also realized a 65% reduction to net loss driven by a major patent litigation settlement agreement as well as reduced equity-based compensation and clinical trial related expenses and the increased royalty revenues noted above. Following is a discussion of Cytomedix’s Results of Operations for the years ended December 31, 2006, 2005, and 2004. This discussion should be read together with the audited financial statements and notes thereto contained in Item 8 of this document.
COMPARATIVE RESULTS OF OPERATIONS FOR THE YEARS ENDED DECEMBER 31, 2006 AND 2005
Revenues
Revenues rose $434,000 (29%) to $1,948,000 comparing the year ended December 31, 2006, to the same period last year. Revenues are normally generated from two sources: the sale of disposable kits and reagents and royalties received from licensing activities. In the third quarter of 2006, the Company also recognized $117,000 in revenue related to comprehensive wound services provided for a government agency under a limited term contract. This service revenue is not expected to continue. The increase was attributable to increased royalties of $496,000 and increased sales of $117,000 related to the services mentioned above, partially offset by a $179,000 decrease in product sales. Increases in royalties were due to six new license agreements entered into during 2005. Product sales decreased primarily due to decreased sales to nursing homes, government agencies, and Medicaid customers.
Gross Profit
Gross profit rose $300,000 (47%) to $934,000 comparing the year ended December 31, 2006, to the same period last year. For the same periods, gross margins rose to 48% from 42%.
The increase in gross profits is primarily attributable to the licensing agreements entered into after March 31, 2005 which carry a greater gross margin than previously existing licensing agreements.
Royalties from the licensing agreements with DePuy Spine, Inc., inclusive of the amortization of deferred revenue associated with the initial deposit of $750,000, generates a gross margin of approximately 20%. The Company expects gross margins generated from all other licensing agreements to be in the range of 50-70%.
Operating Expenses
Operating expenses fell $2,986,000 (37%) to $5,093,000 comparing the year ended December 31, 2006, to the same period last year. The Company relies heavily on the use of equity-based compensation to various employees, consultants and other parties that provide services to the Company. Due to the magnitude of this non-cash expense, the exhibits below highlight the impact of this equity-based compensation on the Company’s operating expenses. These exhibits present the Company’s operating expenses in accordance with generally accepted accounting principles (“GAAP”), the amount of equity-based compensation expense included in the respective line items, and then the operating expenses without the equity-based compensation, which is not in accordance with GAAP (“NON-GAAP”). The following exhibit is presented as an additional tool to evaluate the Company’s operating expenditures between years:
18
Operating Expense Information Not in Conformity with Generally Accepted Accounting Principles
Year Ended December 31, 2006 | Year Ended December 31, 2005 | ||||||||||||||||||
GAAP AS REPORTED | NET EQUITY BASED COMPENSATION | NON-GAAP OPERATING EXPENSES WITHOUT EQUITY BASED COMPENSATION | GAAP AS REPORTED | NET EQUITY BASED COMPENSATION | NON-GAAP OPERATING EXPENSES WITHOUT EQUITY BASED COMPENSATION | ||||||||||||||
| Salaries and wages | $ | 2,190,737 | $ | (692,250 | ) | $ | 1,498,487 | $ | 2,970,036 | $ | (1,352,448 | ) | $ | 1,617,588 | |||||
| Consulting expenses | 180,157 | (8,344 | ) | 171,813 | 145,739 | (29,444 | ) | 116,295 | |||||||||||
| Consulting expenses - related party | 35,000 | — | 35,000 | 185,764 | (77,764 | ) | 108,000 | ||||||||||||
| Professional fees | 763,912 | — | 763,912 | 1,018,779 | — | 1,018,779 | |||||||||||||
| Royalty expenses - related party | 75,000 | — | 75,000 | 75,000 | — | 75,000 | |||||||||||||
| Clinical trial related expenses | 62,052 | — | 62,052 | 1,588,916 | — | 1,588,916 | |||||||||||||
| General and administrative expenses | 1,785,976 | (502,330 | ) | 1,283,646 | 2,095,001 | (556,999 | ) | 1,538,002 | |||||||||||
| Total operating expenses | $ | 5,092,834 | $ | (1,202,924 | ) | $ | 3,889,910 | $ | 8,079,235 | $ | (2,016,655 | ) | $ | 6,062,580 | |||||
Salaries and Wages
Salaries and wages fell $779,000 (26%) to $2,191,000 comparing the year ended December 31, 2006, to the same period last year. The decrease was primarily due to lower non-cash equity-based compensation ($660,000) and fewer employees.
Consulting and Related Party Consulting Expenses
Consulting and related party consulting expenses fell $116,000 (35%) to $215,000 comparing the year ended December 31, 2006, to the same period last year. The decrease was primarily due to lower non-cash equity-based compensation ($99,000) and the overall reduction in use of outside consultants.
Professional Fees
Professional fees fell $255,000 (25%) to $764,000 comparing the year ended December 31, 2006, to the same period last year. Professional fees consist primarily of legal and accounting services.
The decrease was primarily due to decreases in patent litigation related expenditures ($315,000) due to the successful completion of several patent infringement actions in 2005, decreases in fees to securities and general counsel attorneys ($90,000) due primarily to reduced current period activity related to the Company’s listing on the American Stock Exchange, and decreases in accounting fees ($55,000), partially offset by increases in audit fees ($75,000) driven by compliance with Section 404 of the Sarbanes-Oxley Act and increased attorneys fees ($175,000) related to the appeal of the FDA’s decision regarding the Company’s 510(k) Premarket Notification for AutoloGelTM System.
Clinical Trial Related Expenses
Clinical trial related expenses fell $1,527,000 (96%) to $62,000 comparing the year ended December 31, 2006, to the same period last year. The Company completed the active phase of the trial in 2005 and in the first two quarters of 2006 incurred only limited expenses associated with the close out of the trial.
General and Administrative Expenses
General and administrative expenses fell $309,000 (15%) to $1,786,000 comparing the year ended December 31, 2006, to the same period last year. The decrease was due primarily to decreases in travel related expenditures ($163,000), AMEX filing fees ($52,000), investor services ($43,000), and depreciation of fixed assets ($35,000), partially offset by increases in marketing related activities ($30,000).
Other Income/Expenses
Other income rose $954,000 (101%) to $1,896,000 comparing the year ended December 31, 2006, to the same period last year. The increase was primarily due to increased interest income ($143,000) as a result of higher interest rates and larger cash balances, increased patent settlement income ($600,000, net), and a one time charge ($228,000) in 2005 recorded for the issuance of 65,000 shares of the Company’s Common stock in return for a full settlement and release of all claims from a lawsuit brought against the Company relating to its emergence from bankruptcy.
19
COMPARATIVE RESULTS OF OPERATIONS FOR THE YEARS ENDED DECEMBER 31, 2005 AND 2004
Revenues
Revenues rose $369,000 (32%) to $1,514,000 comparing the year ended December 31, 2005 to the same period in 2004. Revenues are normally generated from two sources: the sale of disposable kits and reagents and royalties received from licensing activities.
The increase in revenue is attributable to increased royalties of $547,000 from five new licensing agreements entered into during 2005, primarily those with Medtronic, Inc. and Harvest Technologies, Inc., entered into during the second quarter. This increase was partially offset by a $178,000 decrease in AutoloGel™ kit sales. AutoloGel™ kit sales decreased due to reduced sales to two large nursing home customers and difficulty qualifying patients for commercial insurance reimbursement, partially offset by an increase in sales to the non-reimbursement sensitive market.
Gross Profit
Gross profit rose $199,000 (46%) to $633,000 comparing the year ended December 31, 2005 to the same period in 2004. For the same periods, gross margins rose to 42% from 38%.
The increase in gross profit is primarily attributable to the Medtronic and Harvest licensing agreements which carry a greater gross margin than previously existing licensing agreements. This increase in gross profit was partially offset by a decrease in gross profit from AutoloGel™ kit sales. This decrease was primarily attributable to increased costs for educational treatments used in an effort to penetrate more profitable segments of the market, as well as the write-off of obsolete inventory.
The DePuy royalties, inclusive of the amortization of deferred revenue associated with the initial deposit of $750,000, generates a gross margin of approximately 20%. The Company expects gross margins generated from all other licensing agreements to be in the range of 50-70%.
Operating Expenses
Operating expenses fell $543,000 (6%) to $8,079,000 comparing the year ended December 31, 2005 to the same period in 2004. The Company relies heavily on the use of equity-based compensation to various employees, consultants and other parties that provide services to the Company. Due to the magnitude of this non-cash expense, the following exhibit highlights the impact of this equity-based compensation on the Company’s operating expenses. The exhibit below presents the Company’s operating expenses in accordance with generally accepted accounting principles (“GAAP”) and presents the amount of equity-based compensation expense included in the respective line items and then reflects the operating expenses without the equity-based compensation, which is not in accordance with GAAP (“NON-GAAP”). The following exhibit is presented to provide an additional tool to evaluate the Company’s operating expenditures between years:
Operating Expense Information Not In Conformity With Generally Accepted Accounting Principles
Year Ended December 31, 2005 | Year Ended December 31, 2004 | ||||||||||||||||||
ACCOUNT | GAAP AS REPORTED | NET EQUITY BASED | NON-GAAP OPERATING | GAAP AS REPORTED | NET EQUITY BASED | NON-GAAP OPERATINGEXPENSES WITHOUT | |||||||||||||
| Salaries and wages | $ | 2,970,036 | $ | (1,352,448 | ) | $ | 1,617,588 | $ | 1,769,170 | $ | (578,492 | ) | $ | 1,190,678 | |||||
| Consulting expenses | 145,739 | (29,444 | ) | 116,295 | 1,023,255 | (837,975 | ) | 185,280 | |||||||||||
| Consulting expenses - related party | 185,764 | (77,764 | ) | 108,000 | 789,189 | (529,085 | ) | 260,104 | |||||||||||
| Professional fees | 1,018,779 | — | 1,018,779 | 867,928 | (10,250 | ) | 857,678 | ||||||||||||
| Royalty expenses - related party | 75,000 | — | 75,000 | 75,000 | — | 75,000 | |||||||||||||
| Clinical trial related expenses | 1,588,916 | — | 1,588,916 | 1,385,120 | — | 1,385,120 | |||||||||||||
| General and administrative expenses | 2,095,001 | (556,999 | ) | 1,538,002 | 2,712,462 | (1,141,454 | ) | 1,571,008 | |||||||||||
| Total operating expenses | $ | 8,079,235 | $ | (2,016,655 | ) | $ | 6,062,580 | $ | 8,622,124 | $ | (3,097,256 | ) | $ | 5,524,868 | |||||
20
Salaries and Wages
Salaries and wages rose $1,201,000 (68%) to $2,970,000 comparing the year ended December 31, 2005 to the same period in 2004.
The increase was primarily due to increased non-cash equity-based compensation, primarily a result of $798,000 related to the revaluation and expense associated with the change in terms to existing options, pursuant to the separation agreement with the Company’s former CFO dated July 15, 2005, and increased bonus accrual ($216,000). The remainder of the increase is due to additional employees, and severance costs for the former CFO.
Consulting and Related Party Consulting Expenses
Consulting and related party consulting expenses fell $1,481,000 (82%) to $332,000 comparing the year ended December 31, 2005 to the same period in 2004.
The decrease was primarily due to a $1,260,000 decrease in non-cash equity-based compensation. The remaining change was due to decreases in other compensation and expenses relating to these consultants. During 2004, the Company relied upon the issuance of stock options and warrants to attract and retain senior level consultants to assist in all phases of its operations. This included strategic planning, financing related support, governmental support and lobbying, and on-going managerial support. Most of these activities were significantly curtailed or eliminated in 2005.
Professional Fees
Professional fees rose $151,000 (17%) to $1,019,000 comparing the year ended December 31, 2005 to the same period in 2004. Professional fees consist primarily of legal and accounting services.
The increase was primarily due to increased patent litigation related expenditures and auditing/accounting fees, partially offset by decreases in fees to securities and general counsel attorneys.
Clinical Trial Related Expenses
Clinical trial related expenses rose $204,000 (15%) to $1,589,000 comparing the year ended December 31, 2005 to the same period in 2004. During 2005, the trials were in the active treatment phase.
The increase was primarily due to greater expenditures ($268,000) for the various clinical sites participating in the trials and higher fees from its contract research organization and for consultants, partially offset by reduced costs for equipment and supplies.
General and Administrative Expenses
General and administrative expenses fell $617,000 (23%) to $2,095,000 comparing the year ended December 31, 2005 to the same period in 2004.
The decrease was primarily due to a decrease in non-cash equity-based compensation for the board of directors and outside service providers ($619,000).
Other Income/Expenses
Other income rose $894,000 (1,822%) to $943,000 comparing the year ended December 31, 2005 to the same period in 2004.
The increase was primarily attributable to patent litigation settlements (net of related costs) in the amount of $1,047,000, with a lesser impact from higher interest income. These increases were partially offset by expenses ($227,500) recorded for the issuance of 65,000 shares of the Company’s Common stock in return for a full settlement and release of all claims from a lawsuit brought against the Company in its emergence from bankruptcy.
21
MODIFIED EBITDA INFORMATION NOT IN CONFORMITY WITH GENERALLY ACCEPTED ACCOUNTING PRINCIPLES
Throughout this report, the Company has presented income statement items in conformity with GAAP, except where otherwise noted. Given the magnitude of the non-cash expenses, the Company utilizes a modified EBITDA (earnings before income taxes, depreciation and amortization and other non-cash items) to evaluate and monitor the results of operations. Although EBITDA is a NON-GAAP financial measure, the Company believes that this information will allow for an additional clarification of the Company’s performance and provides the readers of the Company’s financial statements an additional tool to evaluate the comparative performance of the Company. Following, is a reconciliation of the comparative net (loss) to Common shareholders to modified EBITDA utilized by the Company:
Year Ended December 31, | ||||||||||
2006 | 2005 | 2004 | ||||||||
| Net loss to common stockholders, | ||||||||||
| as stated (GAAP) | $ | (2,299,317 | ) | $ | (6,588,387 | ) | $ | (11,259,187 | ) | |
| Adjustments to reconcile net loss | ||||||||||
| to common stockholders to EBITDA: | ||||||||||
| Preferred dividends | 36,361 | 84,902 | 319,861 | |||||||
| Series C Preferred stock dividend | ||||||||||
| attributable to below market | ||||||||||
| beneficial conversion features | — | — | 2,800,000 | |||||||
| Depreciation and amortization | 197,194 | 224,274 | 218,954 | |||||||
| Stock-based compensation | 1,202,924 | 2,016,655 | 3,097,256 | |||||||
Other expense (1) | — | 262,500 | — | |||||||
| MODIFIED EBITDA – NON-GAAP | $ | (862,838 | ) | $ | (4,000,056 | ) | $ | (4,823,116 | ) | |
| (1) | Consists of 65,000 shares of the Company’s Common stock (market value $227,500) issued in return for a full settlement and release of all claims from a lawsuit brought against the Company in its emergence from bankruptcy and 8,673 shares of the Company’s Common stock (market value $35,000) issued for executive search fees. |
CONTRACTUAL OBLIGATIONS
The Company had the following contractual obligations as of December 31, 2006:
| Payments Due by Period | ||||||||||||||||
| Contractual Obligations | Total | Less than 1 Year | 1-3 Years | 4-5 Years | More than 5 Years | |||||||||||
| Operating Leases | $ | 103,027 | $ | 64,658 | $ | 38,369 | $ | — | $ | — | ||||||
Other Liabilities (1) | 329,000 | 144,000 | 185,000 | — | — | |||||||||||
| Total | $ | 432,027 | $ | 208,658 | $ | 223,369 | $ | — | $ | — | ||||||
___________
(1) Amounts less than one year are included in the "Accounts payable and accrued expenses" line of the Balance Sheet.
LIQUIDITY AND CAPITAL RESOURCES
In the years ended December 31, 2006, 2005, and 2004, the Company's operating revenues did not cover the costs of its operations. The cash position of the Company at December 31, 2006 was $4,662,000. The Company believes that it will have adequate cash on hand to fund operations for the year ending December 31, 2007. However, additional cash may be required if operating revenues do not materialize, the cost of operations increases, or if the Company’s efforts to appeal the FDA’s NSE determination letter prove unsuccessful and a change in strategy requires significant short-term funding.
22
The Company has no material commitments for capital expenditures.
Because the Company was in bankruptcy in 2002, the Company may not be able to obtain debt financing. All working capital required to implement the Company’s business plan will be provided by funds obtained through offerings of its equity securities, and revenues generated by the Company.
PROSPECTS FOR THE FUTURE
Cytomedix’s success is directly dependent on the success of AutoloGel™, and the Company believes that AutoloGel™ has a reasonable chance for success in the marketplace. First and foremost, the Company believes that, based on the results of the Company’s clinical trial and other historical data as well as the preliminary results of a pharmaco-economic study, AutoloGel™ has higher healing rates for diabetic foot ulcers and is more cost effective than most other wound treatments. The Company owns the patents on the process for utilizing platelet gel for wound healing, the basis of its license agreements, through 2009 and for the specific formulation of AutoloGel™, which provides several competitive advantages, through 2019.
However, Cytomedix is currently facing a regulatory hurdle. Specifically, it is seeking a reversal, or acceptable amendment, to the FDA’s NSE determination letter. If the Company is successful in this effort, then its current strategic plan remains completely in tact. If efforts are unsuccessful, the Company would likely implement one or more contingent strategies. These contingent strategies are currently being evaluated and refined.
Although it is premature to narrow these strategies, they are currently being considered independently and in combinations. Each strategy offers its own unique set of opportunities and challenges.
Significant challenges still exist in implementing the Company’s plans; whether FDA clearance is obtained and the Company pursues its current strategy, or the Company pursues an alternative strategic plan. Management continues to focus its efforts on leveraging the strength of its intellectual property and successes to date.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
The Company does not enter into financial instruments for speculation or trading purposes. In accordance with the Company’s investment policy, cash is to be invested in bank and institutional money market funds, or in T-Bills or short-term T-Notes. At December 31, 2006, the Company’s cash balance of approximately $4.7 million was maintained primarily in bank and institutional money market accounts. These accounts are sensitive to changes in the general level of interest rates. Based on the Company’s cash balances at December 31, 2006, a 100 basis point increase or decrease in interest rates would have an approximately $47,000 impact on the Company’s annual interest income and net loss. Actual changes in rates may differ from the hypothetical assumption used in computing this exposure.
23
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Shareholders
Cytomedix, Inc.
Rockville, Maryland
We have audited the accompanying balance sheets of Cytomedix, Inc. as of December 31, 2006 and 2005, and the related statements of operations, stockholders' equity, and cash flows for each of the years in the three-year period ended December 31, 2006. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of' Cytomedix, Inc. as of December 31, 2006 and 2005 and its results of operations, changes in stockholders' equity and its cash flows for each of the years in the three-year period ended December 31, 2006 in conformity with. accounting principles generally accepted in the United States of America.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United Sates), the effectiveness of Cytomedix, Inc. internal control over financial reporting as of December 31, 2006, based on criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO), and our report dated February 23, 2007 expressed an unqualified opinion on management's assessment of internal control over financial reporting and an adverse opinion on the effectiveness of internal control over financial reporting.
L J SOLDINGER ASSOCIATES, LLC
Deer Park, Illinois, USA
February 23, 2007
24
CYTOMEDIX, INC.
Balance Sheets
December 31, 2006 | December 31, 2005 | ||||||
ASSETS | |||||||
| Current assets | |||||||
| Cash | $ | 4,662,199 | $ | 3,123,927 | |||
| Accounts and royalties receivable, net | 548,269 | 430,167 | |||||
| Patent settlements receivable, current portion | 437,112 | 15,562 | |||||
| Prepaid expenses, inventory, and other current assets | 155,356 | 222,187 | |||||
| Total current assets | 5,802,936 | 3,791,843 | |||||
| Patent settlements receivable | 574,072 | 31,962 | |||||
| Property and equipment, net | 11,759 | 74,594 | |||||
| Patents, net | 1,823,384 | 1,957,895 | |||||
| Goodwill | 2,021,623 | 2,021,623 | |||||
| Total assets | $ | 10,233,774 | $ | 7,877,917 | |||
LIABILITIES AND STOCKHOLDERS' EQUITY | |||||||
| Current liabilities | |||||||
| Accounts payable and accrued expenses | $ | 1,208,077 | $ | 1,109,860 | |||
| Deferred revenues, current portion | 99,900 | 89,900 | |||||
| Dividends payable on Series A, Series B and Series C preferred stock | 18,236 | 28,142 | |||||
| Total current liabilities | 1,326,213 | 1,227,902 | |||||
| Deferred revenues | 191,475 | 263,745 | |||||
| Other liabilities | 185,000 | — | |||||
| Total liabilities | 1,702,688 | 1,491,647 | |||||
| Commitments and contingencies | |||||||
| Stockholders' equity | |||||||
| Series A Convertible preferred stock; $.0001 par value, authorized 5,000,000 | |||||||
| shares; 2006 and 2005 issued and outstanding - 365,970 and 347,856 shares, | |||||||
| respectively, liquidation preference of $365,970 and $347,856, respectively | 37 | 34 | |||||
| Series B Convertible preferred stock; $.0001 par value, authorized 5,000,000 | |||||||
| shares; 2006 and 2005 issued and outstanding - 83,431 and 84,604 shares, | |||||||
| respectively, liquidation preference of $83,431 and $84,604, respectively | 8 | 8 | |||||
| Series C Convertible preferred stock; $.0001 par value, authorized 1,000,000 | |||||||
| shares; 2006 and 2005 issued and outstanding - 0.0 shares | — | — | |||||
| Common stock; $.0001 par value, authorized 65,000,000 shares; 2006 and 2005 | |||||||
| issued and outstanding - 28,987,670 and 26,158,778 shares, respectively | 2,899 | 2,617 | |||||
| Subscriptions receivable | (620,000 | ) | — | ||||
| Additional paid-in capital | 35,779,380 | 30,954,333 | |||||
| Deferred compensation | — | (238,801 | ) | ||||
| Accumulated deficit | (26,631,238 | ) | (24,331,921 | ) | |||
| Total stockholders' equity | 8,531,086 | 6,386,270 | |||||
| Total liabilities and stockholders' equity | $ | 10,233,774 | $ | 7,877,917 | |||
The accompanying notes are an integral part of these financial statements.
25
CYTOMEDIX, INC.
Statements of Operations
Year Ended December 31, | ||||||||||
2006 | 2005 | 2004 | ||||||||
| Revenues | ||||||||||
| Sales | $ | 204,525 | $ | 266,876 | $ | 444,932 | ||||
| Royalties | 1,743,630 | 1,247,549 | 700,659 | |||||||
| Total revenues | 1,948,155 | 1,514,425 | 1,145,591 | |||||||
| Cost of revenues | ||||||||||
| Cost of sales | 94,258 | 115,956 | 140,247 | |||||||
| Cost of royalties | 920,327 | 765,368 | 571,580 | |||||||
| Total cost of revenues | 1,014,585 | 881,324 | 711,827 | |||||||
| Gross profit | 933,570 | 633,101 | 433,764 | |||||||
| Operating expenses | ||||||||||
| Salaries and wages | 2,190,737 | 2,970,036 | 1,769,170 | |||||||
| Consulting expenses | 180,157 | 145,739 | 1,023,255 | |||||||
| Consulting expenses - related party | 35,000 | 185,764 | 789,189 | |||||||
| Professional fees | 763,912 | 1,018,779 | 867,928 | |||||||
| Royalty expenses - related party | 75,000 | 75,000 | 75,000 | |||||||
| Clinical trial related expenses | 62,052 | 1,588,916 | 1,385,120 | |||||||
| General and administrative expenses | 1,785,976 | 2,095,001 | 2,712,462 | |||||||
| Total operating expenses | 5,092,834 | 8,079,235 | 8,622,124 | |||||||
| Loss from operations | (4,159,264 | ) | (7,446,134 | ) | (8,188,360 | ) | ||||
| Other income (expenses) | ||||||||||
| Interest income (expense), net | 244,595 | 101,564 | 54,049 | |||||||
| Contract settlement and other gain (expense) | 4,406 | (206,159 | ) | (5,015 | ) | |||||
| Patent litigation settlements, net | 1,647,307 | 1,047,244 | -- | |||||||
| Total other income (expenses) | 1,896,308 | 942,649 | 49,034 | |||||||
| Loss before provision for income taxes | (2,262,956 | ) | (6,503,485 | ) | (8,139,326 | ) | ||||
| Income tax provision | — | — | — | |||||||
| Net loss | (2,262,956 | ) | (6,503,485 | ) | (8,139,326 | ) | ||||
| Preferred dividend on: | ||||||||||
| Series A preferred stock | 29,052 | 43,769 | 122,740 | |||||||
| Series B preferred stock | 7,131 | 18,882 | 117,926 | |||||||
| Series C preferred stock | 178 | 22,251 | 2,879,195 | |||||||
| Net loss to common stockholders | $ | (2,299,317 | ) | $ | (6,588,387 | ) | $ | (11,259,187 | ) | |
| Loss per common share — | ||||||||||
| Basic and diluted | $ | (0.08 | ) | $ | (0.27 | ) | $ | (0.62 | ) | |
Weighted average shares outstanding — | ||||||||||
| Basic and diluted | 27,470,781 | 24,428,653 | 18,085,769 | |||||||
The accompanying notes are an integral part of these financial statements.
26
CYTOMEDIX, INC.
Statements of Stockholders’ Equity
Common Stock | Series A Preferred | Series B Preferred | Series C Preferred | |||||||||||||||||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Additional Paid-in | Deferred Compensation | Subscriptions Receivable | Accumulated Deficit | Total Stockholders | ||||||||||||||||||||||||||||
Balance at December 31, 2003 | 13,211,453 | $ | 1,323 | 1,475,471 | $ | 147 | 1,514,862 | $ | 151 | — | $ | — | $ | 12,378,878 | $ | (1,438,070 | ) | $ | — | $ | (6,484,347 | ) | $ | 4,458,082 | ||||||||||||||||
| Private placement of 4,500,000 common shares with warrants, net of direct commissions and expensese | 4,500,000 | 450 | — | — | — | — | — | — | 4,011,175 | — | (2,312,500 | ) | — | 1,699,125 | ||||||||||||||||||||||||||
| Private placement of 280 shares of Series C Convertible Preferred stock with warrants, net of direct commissions and expensese | — | — | — | — | — | — | 280 | — | 2,474,193 | — | — | — | 2,474,193 | |||||||||||||||||||||||||||
| Beneficial conversion feature recorded as a preferred stock dividend related to the Series C stock | — | — | — | — | — | — | — | — | 2,800,000 | — | — | — | 2,800,000 | |||||||||||||||||||||||||||
| Receipt of subscriptions | — | — | — | — | — | — | — | — | — | — | 1,501,908 | — | 1,501,908 | |||||||||||||||||||||||||||
| Interest earned on subscriptions | — | — | — | — | — | — | — | — | — | — | (21,007 | ) | — | (21,007 | ) | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series A stock | 5,999 | 1 | (17,999 | ) | (2 | ) | — | — | — | — | 1 | — | — | — | — | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series B stock | 88,736 | 9 | — | — | (250,212 | ) | (25 | ) | — | — | 16 | — | — | — | — | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series C stock | 1,960,500 | 196 | — | — | — | — | (196 | ) | — | — | — | — | — | 196 | ||||||||||||||||||||||||||
| Dividend issued on Series A and Series B stock | — | — | 118,312 | 12 | 122,392 | 12 | — | — | 240,679 | — | — | — | 240,703 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class A warrants | 659,418 | 66 | — | — | — | — | — | — | 659,352 | — | — | — | 659,418 | |||||||||||||||||||||||||||
| Common stock issued upon cashless exercise of 268,945 Class A warrants | 93,500 | 9 | — | — | — | — | — | — | (9 | ) | — | — | — | — | ||||||||||||||||||||||||||
| Common stock issued upon exercise of Class B warrants | 51,898 | 5 | — | — | — | — | — | — | 77,842 | — | — | — | 77,847 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-1 warrants | 99,500 | 10 | — | — | — | — | — | — | 149,240 | — | — | — | 149,250 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-2 warrants | 85,000 | 8 | — | — | — | — | — | — | 127,491 | — | — | — | 127,499 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of employee stock options | 55,000 | 5 | — | — | — | — | — | — | 82,495 | — | — | — | 82,500 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of other warrants | 517,455 | 52 | — | — | — | — | — | — | 542,403 | — | — | — | 542,455 | |||||||||||||||||||||||||||
| Common stock issued upon cashless exercise of 292,397 other warrants | 160,078 | 16 | — | — | — | — | — | — | (21 | ) | — | — | — | (5 | ) | |||||||||||||||||||||||||
| Common stock issued in lieu of cash for commissions earned in private placement | 12,300 | 1 | — | — | — | — | — | — | 15,374 | — | — | — | 15,375 | |||||||||||||||||||||||||||
| Record other legal and accounting expenses associated with the private placements | — | — | — | — | — | — | — | — | (67,234 | ) | — | — | — | (67,234 | ) | |||||||||||||||||||||||||
| Options issued and issuable to Dr. Kshitij Mohan per his employment agreement dated April 20, 2004 | — | — | — | — | — | — | — | — | 740,000 | (740,000 | ) | — | — | — | ||||||||||||||||||||||||||
| Revaluation of options issued to BDR, Inc. in connection with consulting services agreement | — | — | — | — | — | — | — | — | 244,459 | (244,459 | ) | — | — | — | ||||||||||||||||||||||||||
| Warrants issued in connection with a general business consulting contract with Nadine C. Smith dated April 2003 | — | — | — | — | — | — | — | — | 647,300 | (647,300 | ) | — | — | — | ||||||||||||||||||||||||||
| Options issued and issuable to Carmen Group in connection with a services contract dated October 1, 2003 | — | — | — | — | — | — | — | — | 84,440 | (84,440 | ) | — | — | — | ||||||||||||||||||||||||||
| Warrants issued to Burnham-Hill in March 2004 | — | — | — | — | — | — | — | — | 111,430 | (111,430 | ) | — | — | — | ||||||||||||||||||||||||||
| Termination options issued to Mark E. Cline and Nadine C. Smith | — | — | — | — | — | — | — | — | 319,938 | (319,938 | ) | — | — | — | ||||||||||||||||||||||||||
| Other options issued | — | — | — | — | — | — | — | — | 34,646 | (34,646 | ) | — | — | — | ||||||||||||||||||||||||||
Amortization of deferred compensation related to options and warrants issued for services rendered by— | ||||||||||||||||||||||||||||||||||||||||
| Related parties | — | — | — | — | — | — | — | — | — | 787,550 | — | vv | 787,550 | |||||||||||||||||||||||||||
| Terminated parties | — | — | — | — | — | — | — | — | — | 319,938 | — | — | 319,938 | |||||||||||||||||||||||||||
| Other parties | — | — | — | — | — | — | — | — | — | 1,945,007 | — | — | 1,945,007 | |||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | (11,259,187 | ) | (11,259,187 | ) | |||||||||||||||||||||||||
Balance at December 31, 2004 | 21,500,837 | $ | 2,151 | 1,575,784 | $ | 157 | 1,387,042 | $ | 138 | 84 | $ | — | $ | 25,674,088 | $ | (567,788 | ) | $ | (831,599 | ) | $ | (17,743,534 | ) | $ | 6,533,613 | |||||||||||||||
The accompanying notes are an integral part of these financial statements.
27
CYTOMEDIX, INC.
Statements of Stockholders’ Equity
Common Stock | Series A Preferred | Series B Preferred | Series C Preferred | |||||||||||||||||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Additional Paid-in | Deferred Compensation | Subscriptions Receivable | Accumulated Deficit | Total Stockholders | ||||||||||||||||||||||||||||
| Receipt of subscriptions | — | — | — | — | — | — | — | — | — | — | 832,465 | — | 832,465 | |||||||||||||||||||||||||||
| Interest earned on subscriptions | — | — | — | — | — | — | — | — | — | — | (866 | ) | — | (866 | ) | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series A stock | 418,219 | 42 | (1,253,046 | ) | (125 | ) | — | — | — | — | 83 | — | — | — | — | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series B stock | 435,380 | 44 | — | — | (1,308,773 | ) | (131 | ) | — | — | 87 | — | — | — | — | |||||||||||||||||||||||||
| Common stock issued upon conversion of Series C stock | 839,500 | 84 | — | — | — | — | (84 | ) | — | (84 | ) | — | — | — | — | |||||||||||||||||||||||||
| Dividend issued on Series A and Series B stock (paid in Common stock on those preferred shares converted during the year) | 27,871 | 3 | 25,118 | 2 | 6,335 | 1 | — | — | 166,061 | — | — | — | 166,067 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class A warrants | 42,500 | 4 | — | — | — | — | — | — | 42,496 | — | — | — | 42,500 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class B warrants | 449,233 | 45 | — | — | — | — | — | — | 673,805 | — | — | — | 673,850 | |||||||||||||||||||||||||||
Common stock issued upon cashless exercise of 94,171, Class B warrants | 57,775 | 6 | — | — | — | — | — | — | (6 | ) | — | — | — | — | ||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-1 warrants | 462,900 | 46 | — | — | — | — | — | — | 694,304 | — | — | — | 694,350 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-2 warrants | 478,700 | 48 | — | — | — | — | — | — | 718,002 | — | — | — | 718,050 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Long-term Incentive Plan options | 252,000 | 25 | — | — | — | — | — | — | 367,475 | — | — | — | 367,500 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Unit Offering warrants | 250,000 | 25 | — | — | — | — | — | — | 374,975 | — | — | — | 375,000 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of other warrants | 202,975 | 20 | — | — | — | — | — | — | 327,955 | — | — | — | 327,975 | |||||||||||||||||||||||||||
Common stock issued upon cashless exercise of 958,732 other warrants | 667,215 | 67 | — | — | — | — | — | — | (67 | ) | — | — | — | — | ||||||||||||||||||||||||||
| Common stock issued for settlement of bankruptcy-related lawsuit | 65,000 | 6 | — | — | — | — | — | — | 227,494 | — | — | — | 227,500 | |||||||||||||||||||||||||||
| Common stock issued in lieu of cash for fees earned by executive recruiters | 8,673 | 1 | — | — | — | — | — | — | 34,999 | — | — | — | 35,000 | |||||||||||||||||||||||||||
| Options granted under the Long-Term Incentive Plan to Management, Board of Directors, and Advisors | — | — | — | — | — | — | — | — | 377,669 | (377,669 | ) | — | — | — | ||||||||||||||||||||||||||
| Revaluation of options issued to William Allender in connection with severance agreement | — | — | — | — | — | — | — | — | 798,262 | (798,262 | ) | — | — | — | ||||||||||||||||||||||||||
| Warrants granted to consultants | — | — | — | — | — | — | — | — | 476,735 | (476,735 | ) | — | — | — | ||||||||||||||||||||||||||
Amortization of deferred compensation related to options and warrants issued for services rendered by— | ||||||||||||||||||||||||||||||||||||||||
| Related parties | — | — | — | — | — | — | — | — | — | 1,615,533 | — | — | 1,615,533 | |||||||||||||||||||||||||||
| Other parties | — | — | — | — | — | — | — | — | — | 366,120 | — | — | 366,120 | |||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | (6,588,387 | ) | (6,588,387 | ) | |||||||||||||||||||||||||
Balance at December 31, 2005 | 26,158,778 | $ | 2,617 | 347,856 | $ | 34 | 84,604 | $ | 8 | — | $ | — | $ | 30,954,333 | $ | (238,801 | ) | $ | — | $ | (24,331,921 | ) | $ | 6,386,270 | ||||||||||||||||
The accompanying notes are an integral part of these financial statements.
28
CYTOMEDIX, INC.
Statements of Stockholders’ Equity
Common Stock | Series A Preferred | Series B Preferred | Series C Preferred | |||||||||||||||||||||||||||||||||||||
Shares | Amount | Shares | Amount | Shares | Amount | Shares | Amount | Additional Paid-in | Deferred Compensation | Subscriptions Receivable | Accumulated Deficit | Total Stockholders | ||||||||||||||||||||||||||||
| Correction of share balance | — | — | (9,115 | ) | — | 743 | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||
| Reclassification pursuant to adoption of SFAS No. 123R | — | — | — | — | — | — | — | — | (238,801 | ) | 238,801 | — | — | — | ||||||||||||||||||||||||||
| Common stock issued upon conversion of Series A stock | 303 | — | (909 | ) | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||
| Common stock issued upon conversion of Series B stock | 3,003 | — | — | — | (9,010 | ) | (1 | ) | — | — | 1 | — | — | — | — | |||||||||||||||||||||||||
| Dividend issued on Series A and Series B stock | — | — | 28,138 | 3 | 7,094 | 1 | — | — | 35,228 | — | — | — | 35,232 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class B warrants | 22,500 | 2 | — | — | — | — | — | — | 33,748 | — | — | — | 33,750 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-1 warrants | 548,900 | 55 | — | — | — | — | — | — | 823,295 | — | — | — | 823,350 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Class C-2 warrants | 21,750 | 2 | — | — | — | — | — | — | 32,623 | — | — | — | 32,625 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Long-term Incentive Plan options | 79,200 | 8 | — | — | — | — | — | — | 118,792 | — | — | — | 118,800 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of Unit Offering warrants | 1,355,166 | 135 | — | — | — | — | — | — | 2,032,615 | — | — | — | 2,032,750 | |||||||||||||||||||||||||||
| Common stock issued upon exercise of other warrants | 23,070 | 2 | — | — | — | — | — | — | 23,068 | — | — | — | 23,070 | |||||||||||||||||||||||||||
| Common stock to be released upon full payment of other warrants exercised | 775,000 | 78 | — | — | — | — | — | — | 774,922 | — | (697,500 | ) | — | 77,500 | ||||||||||||||||||||||||||
| Collections on subscriptions receivable | — | — | — | — | — | — | — | — | — | — | 77,500 | — | 77,500 | |||||||||||||||||||||||||||
| Expiration of Series C-1 and Unit Offering warrants, payable at $0.01 per called warrant not exercised | — | — | — | — | — | — | — | — | (13,368 | ) | — | — | — | (13,368 | ) | |||||||||||||||||||||||||
Stock-based compensation related to options and warrants issued for services rendered by— | ||||||||||||||||||||||||||||||||||||||||
| Employees and Directors | — | — | — | — | — | — | — | — | 1,073,130 | — | — | — | 1,073,130 | |||||||||||||||||||||||||||
| Other parties | — | — | — | — | — | — | — | — | 129,794 | — | — | — | 129,794 | |||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | — | — | — | (2,299,317 | ) | (2,299,317 | ) | |||||||||||||||||||||||||
Balance at December 31, 2006 | 28,987,670 | $ | 2,899 | 365,970 | $ | 37 | 83,431 | $ | 8 | — | $ | — | $ | 35,779,380 | $ | — | $ | (620,000 | ) | $ | (26,631,238 | ) | $ | 8,531,086 | ||||||||||||||||
The accompanying notes are an integral part of these financial statements.
29
CYTOMEDIX, INC.
Statements of Cash Flows
Year Ended December 31, | ||||||||||
2006 | 2005 | 2004 | ||||||||
CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||||
| Net loss | $ | (2,262,956 | ) | $ | (6,503,485 | ) | $ | (8,139,326 | ) | |
| Adjustments to reconcile net loss to net cash used in | ||||||||||
| operating activities: | ||||||||||
| Depreciation and amortization | 197,194 | 224,274 | 218,954 | |||||||
| Stock-based compensation - consultants and other | 129,794 | 455,565 | 2,518,764 | |||||||
| Stock-based compensation - employees and directors | 1,073,130 | 1,526,088 | 578,492 | |||||||
| Stock issued for contract settlement | — | 227,500 | — | |||||||
| Stock issued for consulting services | — | 35,000 | — | |||||||
| (Gain) Loss on disposal of assets | (4,348 | ) | (16,609 | ) | 4,655 | |||||
| Interest earned on stock subscriptions outstanding | — | (866 | ) | (21,008 | ) | |||||
| Change in current assets | (472,821 | ) | (78,305 | ) | (193,210 | ) | ||||
| Change in patent settlements receivable | (542,110 | ) | (31,962 | ) | — | |||||
| Change in accounts payable and accrued expenses | 98,217 | 94,678 | 400,752 | |||||||
| Change in deferred revenues | (62,270 | ) | (84,138 | ) | (81,448 | ) | ||||
| Change in other liabilities | 185,000 | — | — | |||||||
| Net cash used in operating activities | (1,661,170 | ) | (4,152,260 | ) | (4,713,375 | ) | ||||
CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||||
| Purchase of equipment | — | — | (56,563 | ) | ||||||
| Proceeds from sale of equipment | 4,500 | 38,775 | — | |||||||
| (Increase) Decrease in restricted cash | — | 21,375 | (600 | ) | ||||||
| Net cash provided by (used in) investing activities | 4,500 | 60,150 | (57,163 | ) | ||||||
CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||||
| Proceeds from sale of common and preferred stock, net | — | 832,465 | 5,608,188 | |||||||
| Repayment of note payable | — | — | (13,066 | ) | ||||||
| Proceeds from option and warrant exercises | 3,219,345 | 3,199,227 | 1,638,965 | |||||||
| Payment for expiration of called warrants | (13,368 | ) | — | — | ||||||
| Dividends paid | (11,035 | ) | (90,589 | ) | — | |||||
| Net cash provided by financing activities | 3,194,942 | 3,941,103 | 7,234,087 | |||||||
| Net increase (decrease) in cash | 1,538,272 | (151,007 | ) | 2,463,549 | ||||||
| Cash, beginning of period | 3,123,927 | 3,274,934 | 811,385 | |||||||
| Cash, end of period | $ | 4,662,199 | $ | 3,123,927 | $ | 3,274,934 | ||||
The accompanying notes are an integral part of these financial statements.
30
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 1 - DESCRIPTION OF THE BUSINESS
Cytomedix is a biotechnology company that develops and licenses autologous cellular therapies (i.e., therapies using the patient’s own body products), including Cytomedix’s proprietary process to produce a platelet rich plasma gel (“AutoloGel™”) for the treatment of wounds. To create AutoloGel™, the patient’s own platelets and plasma are separated through centrifugation and combined with several reagents. This process releases multiple growth factors from the platelets, creates a fibrin matrix scaffold, and forms a gel that is topically applied to a wound (under the direction of a physician). Upon topical application, the Company believes that AutoloGel™ initiates a reaction that closely mimics the body’s natural healing process. Cytomedix sells its products primarily to health care providers in the United States and licenses its patents to medical device and product suppliers in the United States. The Company was incorporated in the State of Delaware on April 29, 1998, and has its headquarters in Rockville, Maryland.
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The Company's financial statements are prepared on the accrual basis of accounting in accordance with accounting principles generally accepted in the United States of America. Certain financial information is based on fresh-start accounting utilized upon the Company’s emergence from bankruptcy in July 2002.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the amounts reported in the financial statements and the accompanying notes. Actual results could differ from those estimates.
Concentration of Risk
Approximately $1.5 million, $1.1 million and $0.7 million, or 77%, 76% and 61% of the Company’s revenue in the years ended December 31, 2006, 2005 and 2004 respectively, were generated from royalties from three, three and one licensees, respectively. Should any of these licensees experience a significant decrease in the sales of products covered by its license agreement with Cytomedix, there may be a material adverse effect on Cytomedix’s results of future operations.
As of December 31, 2006 and 2005, the Company maintained approximately $225,000 and $340,000 respectively, in financial institutions in excess of Federal Deposit Insurance Corporation (“FDIC”) insurance. In addition, $3,973,000 and $2,323,000 held in money market accounts at brokerage firms were in excess of the $500,000 Securitites Investor Protection Corporation (“SIPC”) coverage as of December 31, 2006 and 2005, respectively. These amounts not covered by SIPC were insured by the Company’s brokerage firm through the Customer Asset Protection Company (“CAPCO”).
The Company currently has one product that is presently marketed. Significant changes in technology could lead to new products or services that compete with the product offered by the Company. These changes could materially affect the price of the Company’s product or render it obsolete.
The Company outsources manufacturing for all the components of its offerings. While the Company utilizes single suppliers for several components of the AutoloGel offering, such components are readily available on the open market and therefore no dependency exists. The one exception is a reagent, bovine thrombin, available exclusively through King Pharmaceuticals.
31
CYTOMEDIX, INC.
Notes to Financial Statements
Cash Equivalents
The Company considers all highly liquid instruments purchased with an original maturity of three months or less to be cash equivalents.
Accounts and Royalties Receivable
Cytomedix generates accounts receivable from the sale of its products. Cytomedix provides for a reserve against receivables for estimated losses that may result from a customer’s inability or unwillingness to pay. The allowance for doubtful accounts is estimated primarily based upon historical write-off percentages, known problem accounts, and current economic conditions. Accounts are written off against the allowance for doubtful accounts when the Company determines that amounts are not collectable. Recoveries of previously written-off accounts are recorded when collected.
Royalties receivable represent current royalties earned on sales of covered product by licensees.
Inventory
Inventory is stated at the lower of cost or net realizable value. Cost is determined on a first-in-first-out (FIFO) basis. The Company's primary product is a kit that is composed of multiple items that expire at different periods with the earliest item in the kit being the determining factor in its classification as a salable item. Kits with expired components are segregated and used for demonstration purposes only; the Company maintains a full reserve on these kits.
Property and Equipment
Property and equipment is stated at cost less accumulated depreciation and is depreciated, using the straight-line method, over their estimated useful lives ranging from three to seven years. Maintenance and repairs are charged to operations as incurred. When assets are sold, or otherwise disposed of, the cost and related accumulated depreciation are removed from the accounts and any gain or loss is included in other income and expense.
Intangible Assets
The Company capitalizes the costs of purchased and internally developed patents. This cost is amortized via the straight-line method over the remaining life of the patents.
The Company accounts for finite-lived intangibles under SFAS No. 144, “Accounting for the Impairment or Disposal of Long-Lived Assets,” and therefore reviews the recoverability of long-lived and finite-lived intangible assets when circumstances indicate that the carrying amount of assets may not be recoverable.
The Company follows the guidance of SFAS No. 142, “Goodwill and Other Intangible Assets,” with regard to its indefinite-lived intangibles. SFAS No. 142 requires that goodwill be assessed at least annually for impairment by applying a fair value based test. This evaluation has been independently performed for 2006, 2005 and 2004 and is generally based on various analyses including cash flow projections. In the event these analyses indicate an impairment, the Company would record an impairment loss, if any, based on the fair value of the assets.
No impairment of intangible assets was recorded in 2006, 2005 or 2004.
Income Taxes
Deferred income taxes reflect the net tax effects of net operating loss carryforwards and temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes, using enacted tax rates in effect for the year in which the differences are expected to reverse. A full valuation allowance has been established, reducing deferred tax assets to zero, as management has determined that realization of this benefit is not assured.
32
CYTOMEDIX, INC.
Notes to Financial Statements
Revenue Recognition
The Company recognizes revenue in accordance with SEC Staff Accounting Bulletin No. 101, “Revenue Recognition in Financial Statements” (“SAB 101”), as amended. SAB 101 requires that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of an arrangement exists; (2) delivery has occurred or services rendered; (3) consideration is fixed and determinable; and (4) collectibility is reasonably assured.
Revenue from the sale of the Company’s products to distributors and caregivers is recognized upon delivery. Revenue from the sale of the Company’s products to patients is recognized upon use of the product on the patient or acknowledgement from a patient's insurer authorizing treatment, whichever is later.
Revenue from the licensing of patents (royalty revenue) is generally recognized as products are sold by licensees. Certain up-front license fees are amortized over the life of the license agreement. Lump sum payments, considered by the Company to be discharges of past obligations, are reflected net of their associated costs as “Patent litigation settlements, net” in the Statements of Operations. Such amounts are recorded at their present value. The Company records revenue and settlement income related to its agreement with Perfusion Partners Associates, Inc. (“PPAI”) on the cash basis due to PPAI’s recent emergence from bankruptcy.
Stock-Based Compensation
The Company adopted SFAS No. 123R, “Share-Based Payment,” as of January 1, 2006, using the modified prospective application. Under this method, all equity-based compensation awarded after the adoption date has been determined under the fair value provisions of SFAS No. 123R. Additionally, for all equity-based compensation awarded prior to the adoption date, compensation for the portion of awards for which the requisite service is performed after the adoption date is recognized as service is rendered.
The Company’s policy is to issue new shares of its Common stock when employees exercise options awarded under its Long-Term Incentive Plan. See Note 12 to the Financial Statements.
As permitted under SFAS No. 123, the Company applied the intrinsic value method of accounting prescribed by Accounting Principles Board (“APB”) Opinion No. 25, “Accounting for Stock Issued to Employees,” and related interpretations, in accounting for its stock-based grants to employees and directors in 2005 and 2004. Under the “intrinsic value” method, an option’s value is the excess of the market price of the underlying stock on the date of grant over the exercise price of the option. No value is attributed to the option if its exercise price is greater than the stock’s market price. Had the Company continued to use the intrinsic value provisions under APB Opinion No. 25, employee and director stock-based compensation would have been $115,833, net of income taxes, for the year ended December 31, 2006. The change increased reported net loss and basic and diluted loss per share for the year ended December 31, 2006 by $957,297 and $0.03, respectively. The change had no effect on the Company’s cash flow from operating, investing or financing activities.
Under the fair value method prescribed by SFAS 123R, the Company recorded $1,073,130, net of income taxes, in employee and director stock-based compensation for the year ended December 31, 2006. Had compensation expense for the years ended December 31, 2005 and 2004 been determined under the fair value provisions of SFAS No. 123 as amended by SFAS No. 148, “Accounting for Stock-Based Compensation - Transition and Disclosure, an amendment of FASB Statement No. 123,” the Company’s net loss and net loss per share to Common shareholders would have differed as follows:
33
CYTOMEDIX, INC.
Notes to Financial Statements
2005 | 2004 | ||||||
| Net loss to common stockholders, as reported | $ | (6,588,387 | ) | $ | (11,259,187 | ) | |
| Add: | |||||||
| Stock-based employee compensation | |||||||
| expense included in reported net loss | |||||||
| determined under APB No. 25, | |||||||
| net of related tax effects | 698,380 | 258,464 | |||||
| Deduct: | |||||||
| Stock-based employee compensation | |||||||
| expense determined under fair value | |||||||
| based method for all awards, | |||||||
| net of related tax effects | (2,139,046 | ) | (793,792 | ) | |||
| Pro forma net loss | $ | (8,029,053 | ) | $ | (11,794,515 | ) | |
| Loss per share: | |||||||
| Basic and diluted - as reported | $ | (0.27 | ) | $ | (0.62 | ) | |
| Basic and diluted - pro forma | $ | (0.33 | ) | $ | (0.65 | ) | |
These pro forma amounts may not be representative of future disclosures since the estimated fair value of stock options would be amortized to expense over the vesting period and additional options may be issued in future years.
The fair value of each option award is estimated on the date of grant using the Black-Scholes option valuation model. The weighted-average assumptions used in the model are summarized in the following table:
2006 | 2005 | 2004 | ||||||||
| Risk free rate | 4.73 | % | 4.40 | % | 3.90 | % | ||||
| Expected years until exercise | 10.0 | 8.1 | 9.3 | |||||||
| Expected stock volatility | 113 | % | 114 | % | 100 | % | ||||
| Dividend yield | — | — | — | |||||||
Expected volatilities are based on historical volatility of the Company’s stock. The Company uses historical data to estimate option exercise and employee termination within the valuation model. The expected years until exercise represents the period of time that options are expected to be outstanding. The risk-free rate for periods within the contractual life of the option is based on the U.S. Treasury yield curve in effect at the time of grant. The Company estimated that the dividend rate on its Common stock will be zero.
Loss Per Share
Loss per share is calculated in accordance with SFAS No. 128, “Earnings Per Share.” Basic loss per share is computed based upon the weighted average number of shares of Common stock outstanding for the period and excludes any potential dilution. Diluted earnings per share reflects potential dilution from the exercise of securities into Common stock. Outstanding options and warrants to purchase Common stock are not included in the computation of diluted earnings per share because the effect of these instruments would be anti-dilutive (i.e. would reduce the loss per share).
34
CYTOMEDIX, INC.
Notes to Financial Statements
The Common shares potentially issuable upon the exercise of these instruments, were as follows at December 31:
2006 | 2005 | 2004 | ||||||||
| Options | 3,202,377 | 2,782,077 | 2,516,577 | |||||||
| Warrants | 4,745,301 | 8,528,988 | 11,253,200 | |||||||
| Series A Preferred Stock | 121,990 | 115,580 | 525,261 | |||||||
| Series B Preferred Stock | 27,810 | 28,458 | 462,347 | |||||||
| Series C Preferred Stock | — | — | 839,000 | |||||||
| 8,097,478 | 11,455,103 | 15,596,385 | ||||||||
Fair Value of Financial Instruments
The carrying value of current assets and liabilities approximates fair value due to their relatively short maturities.
Reclassifications
Certain reclassifications have been made to conform prior year's data to the current presentation. These reclassifications had no effect on reported earnings.
Registration Payment Arrangements
The Company is party to a registration rights agreement and a related warrant agreement with one of its former consultants. The registration rights agreement provides for liquidated damages, at the discretion of the warrantholder, in the event that the registration statement relating to the shares underlying the warrants becomes ineffective. The Company’s obligations under this agreement run through the earlier of April 1, 2012 or two years after the exercise of the related warrants. At the discretion of the warrantholder, the liquidated damages may take the form of cash or additional shares of the Company’s Common stock. As of December 31, 2006, the Company has estimated the maximum undiscounted liquidated damages at $171,000. However, pursuant to FASB Staff Position No. EITF 00-19-2, which the Company adopted in the fourth quarter of 2006, the Company has determined that it is unlikely that circumstances allowing for the aforementioned liquidated damages would arise, and therefore no contingent liability has been recorded.
Recent Accounting Pronouncements
In July 2006, the FASB issued Interpretation No. 48 (“FIN 48”), “Accounting for Uncertainty in Income Taxes, an interpretation of FASB Statement No. 109,” which seeks to reduce the diversity in practice associated with the accounting and reporting for uncertainty in income tax positions. This Interpretation prescribes a comprehensive model for the financial statement recognition, measurement, presentation and disclosure of uncertain tax positions taken or expected to be taken in income tax returns. FIN 48 is effective for fiscal years beginning after December 15, 2006 and the Company will adopt the new requirements in its fiscal first quarter of 2007. The Company does not expect the adoption of this statement in fiscal year 2007 to have a material impact on the Company’s financial position or results of operations
In September 2006, the FASB issued Statement of Financial Accounting Standards (“SFAS”) 157, “Fair Value Measurements” (“SFAS 157”), which provides guidance on measuring the fair value of assets and liabilities. SFAS 157 will apply to other accounting pronouncements that require or permit assets or liabilities to be measured at fair value but does not expand the use of fair value to any new circumstances. This standard will also require additional disclosures in both annual and quarterly reports. SFAS 157 will be effective for financial statements issued for fiscal years beginning after November 15, 2007. The Company is currently determining the effect, if any, the adoption of SFAS 157 will have on its financial statements.
In September 2006, the SEC staff issued Staff Accounting Bulletin No. 108, “Considering the Effects of Prior Year Misstatements when Quantifying Misstatements in Current Year Financial Statements” (“SAB 108”). The intent of SAB 108 is to reduce diversity in practice for the method companies use to quantify financial statement misstatements, including the effect of prior year uncorrected errors. SAB 108 establishes an approach that requires quantification of financial statement errors using both an income statement and a cumulative balance sheet approach. SAB 108 is effective for fiscal years beginning after November 15, 2006, and the Company will adopt the new requirements in 2007. The adoption of SAB 108 is not currently expected to have a significant impact on the Company’s financial statements.
35
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 3 - WORKING CAPITAL
The Company’s operating revenues do not cover the costs of its operations. The cash position of the Company at December 31, 2006 was approximately $4.7 million. The Company believes that it will have adequate cash on hand to fund operations for the year ending December 31, 2007. However, additional cash may be required if operating revenues do not materialize, the cost of operations increases, or if the Company’s efforts to appeal the FDA’s NSE determination letter prove unsuccessful and a change in strategy requires significant short-term funding.
The Company has no material commitments for capital expenditures.
Because the Company was in bankruptcy in 2002, the Company may not be able to obtain debt financing. All working capital required to implement the Company’s business plan will be provided by funds obtained through offerings of its equity securities, and revenues generated by the Company.
NOTE 4 - LICENSING AGREEMENTS
During 2006 and 2005, the Company entered into several separate settlement and license Agreements. Under the terms of the respective agreements, payments of approximately $4,905,000 were due to the Company for the discharge of past obligations or as upfront fees. Additionally, certain licensees are required to pay on-going royalties on defined classes of sales.
Patent settlements are one-time, non-recurring transactions. Amounts related to these settlements that are payable to the Company over time are reflected as “Patent settlements receivable, current portion” and “Patent settlements receivable” on the Balance Sheet for their current and long-term portions respectively. Associated costs, consisting of royalty and contingent legal fees payable upon the collection of such receivables, are reflected in “Accounts payable and accrued expenses” and “Other liabilities” on the Balance Sheet, for their current and long-term portions, respectively. Income related to the settlement of past obligations, net of associated costs, are reflected as “Patent litigation settlements, net” on the Statements of Operations as follows:
2006 | 2005 | 2004 | ||||||||
| Income | $ | 2,479,000 | $ | 1,388,000 | $ | — | ||||
| Costs | (832,000 | ) | (341,000 | ) | — | |||||
| Net settlement income | $ | 1,647,000 | $ | 1,047,000 | $ | — | ||||
Due to PPAI’s recent emergence from bankruptcy, the Company records incomes when payments are received from PPAI. As of December 31, 2006, the Company had received and recorded $139,000 of the agreed $250,000 settlement for past obligations from PPAI.
Royalties earned after the effective dates of these agreements and amortization of deferred revenue from up front fees, together with the related costs, are included in the Statements of Operations as “Royalties” and “Cost of royalties,” respectively.
Since 2003, the Company has incurred expenses of $690,000 (excluding royalty and contingent legal fees) for patent enforcement actions. Of this amount,$1,000, $479,000, and $197,000 were incurred during 2006, 2005 and 2004, respectively. These fees have primarily been included in Professional fees. The Company’s patent enforcement strategy is being conducted on a full contingency basis.
The Company’s primary license agreements, based on its ownership of the “Knighton Patent” (US Patent No. 5,165,938), are listed below:
36
CYTOMEDIX, INC.
Notes to Financial Statements
Licensee | Date of Agreement | Date of Expiration (4) | Initial Licensing Fee | On-going Royalty Percentage (2) | ||||||||||
| DePuy Spine, Inc. (1) | 3/19/2001 | 11/24/2009 | $ | 750,000 | 6.5% | |||||||||
| 3/4/2005 | ||||||||||||||
| Medtronic, Inc. | 5/1/2005 | 11/24/2009 | $ | 680,000 | 7.5% on disposables | |||||||||
| 1.5% on hardware | ||||||||||||||
| Harvest Technologies, Inc. | 6/30/2005 | 11/24/2009 | $ | 500,000 | 7.5% on disposables | |||||||||
| 1.5% on hardware | ||||||||||||||
| Perfusion Partners, Inc. | 6/26/2005 | 11/24/2009 | $ | $ 250,000 | (3) | 10.0% | ||||||||
| COBE Cardiovascular, Inc. | 10/7/2005 | 11/24/2009 | $ | 45,000 | 7.5% on disposables | |||||||||
| 1.5% on hardware | ||||||||||||||
| SafeBlood Technologies, Inc. | 10/12/2005 | 11/24/2009 | $ | $ 50,000 | (3) | 8.0% to 9.0% | ||||||||
| Biomet Biologics, Inc. (5) | 5/19/2006 | 11/24/2009 | $ | 2,600,000 | none | |||||||||
| CellMedix, Inc. | 11/28/2006 | 11/24/2009 | $ | 30,000 | 9.5% | |||||||||
(1) Cytomedix has two license agreements with DePuy Spine, Inc. The original license agreement was dated March 19, 2001, amended March 3, 2005, and provides for the use of applications under Cytomedix patents in the fields of diagnostic and therapeutic spinal, neurosurgery and orthopedic surgery. The second license agreement is dated March 4, 2005 and applies to all fields not covered in the original license agreement as amended.
(2) Certain minimum royalties may apply to certain agreements and other royalty percentages may apply to future products covered under selected license agreements.
(3) Some of these amounts are payable over a period of time as defined in executed notes payable to Cytomedix.
(4) These dates reflect the expiration of the license in the U.S., which coincides with the expiration of the Knighton Patent in the U.S. In some cases, the licensing agreements applicable to territories outside the U.S. extend to the expiration of the patents in the respective foreign countries.
(5) The Settlement and License Agreement with Biomet Biologics, Inc. (“Biomet”) called for a $2.6 million payout from Biomet to Cytomedix. This payout took the form of $1.4 million payable upon execution of the agreement and $100,000 payable at the end of each of 12 consecutive quarters beginning with the quarter ending September 2006. These payments are not tied to any performance commitments by Cytomedix and are not dependent on Biomet sales.
NOTE 5 - ROYALTY AGREEMENTS
The Company is party to a Royalty Agreement with Curative Health Services, Inc. Under this agreement as amended, Curative is to receive 92% of licensing receipts from DePuy Spine, Inc. (a division of Johnson & Johnson, Inc.) and 10% of the total other amounts received by the Company in connection with upfront, milestone and other similar payments relating to the Knighton Patent.
The Company is also party to a Royalty Agreement with Mr. Charles Worden. The primary terms of this agreement are discussed in Note 15 - Related Party Transactions.
37
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 6 - RECEIVABLES
Accounts and royalties receivable, net consisted of the following at December 31:
2006 | 2005 | ||||||
| Trade receivables | $ | 168,477 | $ | 197,982 | |||
| Royalty receivables | 491,250 | 304,250 | |||||
| Other receivables | 25,393 | 17,828 | |||||
| 685,120 | 520,060 | ||||||
| Less allowance for doubtful accounts | (136,851 | ) | (89,893 | ) | |||
| $ | 548,269 | $ | 430,167 | ||||
Bad debt expense was approximately $62,000 for each of the years ended December 31, 2006 and 2005.
Patent settlements are one-time, non-recurring transactions. Patent settlements receivable consist of amounts owed to the Company for the discharge of past obligations pursuant to settlement and licensing agreements with various licensees. The amounts are reflected at their net present value using discount rates between 8.00% and 8.25%. The current portions are due within one year and long-term portion are due at various periods through June 2009.
NOTE 7 - PREPAID EXPENSES, INVENTORY, AND OTHER CURRENT ASSETS
Prepaid expenses, other current assets and inventory consisted of the following at December 31:
2006 | 2005 | ||||||
| Prepaid insurance | $ | 131,406 | $ | 117,880 | |||
| Prepaid fees and rent | 12,113 | 67,266 | |||||
| Travel advances | — | 5,655 | |||||
| Deposits | 3,190 | 3,811 | |||||
| Inventory | 8,647 | 27,575 | |||||
| $ | 155,356 | $ | 222,187 | ||||
38
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 8 - PROPERTY AND EQUIPMENT
Property and equipment consisted of the following at December 31:
2006 | 2005 | ||||||
| Medical equipment | $ | 279,660 | $ | 281,610 | |||
| Office equipment | 74,559 | 74,559 | |||||
| 354,219 | 356,169 | ||||||
| Less accumulated depreciation | (342,460 | ) | (281,575 | ) | |||
| $ | 11,759 | $ | 74,594 | ||||
Depreciation expense was approximately $63,000, $98,000, and $93,000 for the years ended December 31, 2006, 2005, and 2004, respectively.
NOTE 9 - INTANGIBLE ASSETS
Cytomedix owns eight U.S. patents (including U.S. Patent No. 5,165,938 (the “Knighton Patent”) and U.S. Patent No. 6,303,112 (the “Worden Patent”), various corresponding foreign patents, and various trademarks. The Knighton Patent and Worden Patent expire in November 2009 and February 2019, respectively.
Patents and related accumulated amortization at December 31 was as follows:
2006 | 2005 | ||||||
| Patents | $ | 2,400,000 | $ | 2,400,000 | |||
| Less accumulated amortization | (576,616 | ) | (442,105 | ) | |||
| $ | 1,823,384 | $ | 1,957,895 | ||||
Amortization expense was approximately $135,000 for the year ended December 31, 2006 and $126,000 for each of the years ended December 31, 2005 and 2004. The Company is amortizing the patents over the remaining lives of the patents and the Company expects amortization expense to be approximately $151,000 in each of the next five years.
Goodwill represents the excess reorganization value over the amounts allocable to identifiable assets upon the Company’s emergence from bankruptcy in 2002.
39
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 10 - INCOME TAXES
Income tax (expense) benefit for the years ended December 31, 2006, 2005 and 2004 consisted of the following:
2006 | 2005 | 2004 | ||||||||
| Current: | ||||||||||
| Federal | $ | — | $ | — | $ | — | ||||
| State | — | — | — | |||||||
| Deferred: | ||||||||||
| Federal | 459,000 | 784,000 | 1,138,000 | |||||||
| State | 98,000 | 133,000 | 182,000 | |||||||
| Net operating loss carryforward | 389,000 | 1,794,000 | 1,815,000 | |||||||
| Valuation allowance | (946,000 | ) | (2,711,000 | ) | (3,135,000 | ) | ||||
| Total income tax (expense) benefit | $ | — | $ | — | $ | — | ||||
Significant components of Cytomedix’s deferred tax assets and liabilities consisted of the following at December 31:
2006 | 2005 | ||||||
| Deferred tax assets: | |||||||
| Stock-based compensation | $ | 3,071,000 | $ | 2,564,000 | |||
| Other | 166,000 | 158,000 | |||||
| Total deferred tax assets | 3,237,000 | 2,722,000 | |||||
| Deferred tax liabilities: | |||||||
| Amortization of patents | (594,000 | ) | (631,000 | ) | |||
| Other | (16,000 | ) | (21,000 | ) | |||
| Net deferred tax assets | 2,627,000 | 2,070,000 | |||||
| Net operating loss carryforwards | 7,903,000 | 7,514,000 | |||||
| 10,530,000 | 9,584,000 | ||||||
| Less valuation allowance | (10,530,000 | ) | (9,584,000 | ) | |||
| Total deferred tax assets | $ | — | $ | — | |||
40
CYTOMEDIX, INC.
Notes to Financial Statements
The following table presents a reconciliation between the U.S. federal statutory income tax rate and the Company's effective tax rate:
2006 | 2005 | 2004 | ||||||||
| U.S. Federal statutory income tax | 35.0% | 35.0% | 35.0% | |||||||
| State and local income tax benefits | 6.6% | 7.2% | 2.5% | |||||||
| Other | (1.7%) | (0.4%) | 1.1% | |||||||
| Nondeductible expenses | (0.1%) | (0.1%) | (0.1%) | |||||||
| Valuation allowance for deferred | ||||||||||
| income tax assets | (39.8%) | (41.7%) | (38.5%) | |||||||
| Effective income tax rate | (0.0%) | 0.0% | 0.0% | |||||||
The Company had loss carryforwards of approximately $21,578,000 as of December 31, 2006 that may be offset against future taxable income. The carryforwards will expire between 2021 and 2026. Utilization of these carryforwards may be subject to annual limitations based upon previous significant changes in stock ownership. Management has determined that realization of this benefit is not assured and accordingly has established a valuation allowance of $10,530,000 and $9,584,000 at December 31, 2006 and 2005, respectively.
NOTE 11 - CAPITAL STOCK
The Company has several classes of stock as described below.
Common Stock
Common stock has a par value of $.0001 per share and is limited to a maximum of 65,000,000 shares. It is subordinate to both Series A Convertible Preferred stock and Series B Convertible Preferred stock and to all other classes and series of equity securities of the Company which by their terms rank senior to it, in the event of a liquidation, dissolution, or winding up of the Company or with regard to any other rights, privileges or preferences. Each share of Common stock represents the right to one vote. Holders of Common stock are entitled to receive dividends as may be declared by the Board of Directors, subject to the limitations in the terms of the Series A and B Convertible Preferred stock described below.
Series A Convertible Preferred Stock
Series A Convertible Preferred stock (“Series A”) has a par value of $.0001 per share and is limited to a maximum of 5,000,000 shares. It has a stated liquidation preference of $1.00 per share and preference over and rank senior to (i) Series B Convertible Preferred stock, (ii) Common stock, and (iii) all other classes and series of equity securities of the Company which by its terms do not rank senior to the Series A stock. The Series A contains a negative covenant prohibiting the Company from granting any security interest in the Company's patents and/or future royalty streams ("Intellectual Property"). The holders of record of shares are entitled to receive cumulative dividends at the rate of 8% of the stated liquidation preference amount per share per annum, payable quarterly in arrears. These dividends are prior and in preference to any declaration or payment of any distribution on any outstanding shares of Common stock or any other equity securities of the Company ranking junior as to the payment of dividends. Dividends are to be paid in shares of Series A or, in the sole discretion of the Board of Directors, in cash. Each share of Series A stock shall entitle the holder thereof to vote on all matters voted on by holders of Common stock of the Company voting together as a single class with the other shares entitled to vote.
Each share of Series A stock may be converted into Common stock at a conversion rate equal to 90% of the twenty-day average closing price of the Company’s Common stock, but in no case shall this price be less than $3.00 per share.
The Company may redeem Series A stock for cash at a price per share equal to 104% of the liquidation preference amount plus all accrued but unpaid dividends, by providing proper notice of not less than 10 days nor more than 60 days prior to a redemption date set by the Company.
41
CYTOMEDIX, INC.
Notes to Financial Statements
Series B Convertible Preferred Stock
Series B Convertible Preferred stock (“Series B”) has a par value of $.0001 per share and is limited to a maximum of 5,000,000 shares. It has a stated liquidation preference of $1.00 per share, is subordinate to the Series A stock, and has preference over and ranks senior to (i) common stock, and (ii) all other classes and series of equity securities of the Company which by its terms do not rank senior to the Series B stock. The Series B contains a negative covenant prohibiting the Company from granting any security interest in the Company's patents and/or future royalty streams ("Intellectual Property"). The holders of record of shares are entitled to receive cumulative dividends at the rate of 8% of the stated liquidation preference amount per share per annum, payable quarterly in arrears. These dividends are prior and in preference to any declaration or payment of any distribution on any outstanding shares of Common stock or any other equity securities of the Company ranking junior as to the payment of dividends. Dividends are to be paid in shares of Series B or, in the sole discretion of the Board of Directors, in cash. Each share of Series B stock shall entitle the holder thereof to vote on all matters voted on by holders of Common stock of the Company voting together as a single class with the other shares entitled to vote.
Each share of Series B stock may be converted into Common stock at a conversion rate equal to 90% of the twenty-day average closing price of the Company’s Common stock, but in no case shall this price be less than $3.00 per share.
The Company may redeem Series B stock for cash at a price per share equal to 103% of the liquidation preference amount plus all accrued but unpaid dividends, by providing proper notice of not less than 10 days nor more than 60 days prior to a redemption date set by the Company.
Series C Convertible Preferred Stock
Series C Convertible Preferred stock (“Series C”) has a par value of $.0001 per share and is limited to a maximum of 1,000 shares. It has a stated liquidation preference of $10,000 per share, and ranks junior to the Series A regarding distributions upon liquidation of the Company. Series C stock ranks junior to the Series B solely with respect to the priority security interest in the Company's Intellectual Property. The shares accrued dividends at 6% of the stated liquidation preference amount from the date of issuance and increased to 8% commencing on September 25, 2005, are payable annually in cash or shares of Common stock at the option of the Company. The Series C stock ranks pari passu with Series A and Series B with respect to payment of dividends. As of December 31, 2006 and 2005, no Series C remained outstanding.
Warrants and Options
The Company had the following outstanding warrants and options at December 31:
Equity Instrument | 2006 | 2005 | |||||
| A and B Warrants (1) | — | 22,500 | |||||
| C-1 Warrants (2) | — | 815,900 | |||||
| C-2 Warrants (2) | 855,000 | 876,750 | |||||
| D Warrants (3) | 304,033 | — | |||||
| Unit Warrants (4) | 1,825,000 | 4,250,000 | |||||
| Other warrants (5) | 1,761,268 | 2,563,838 | |||||
| Options issued under the Long-Term Incentive Plan (6) | 3,202,377 | 2,782,077 | |||||
(1) These warrants were issued in connection with the $800,000 raised while the Company was a debtor in possession and the $2.8 million raised upon the Company’s emergence from bankruptcy. As of December 31, 2006, all class A and class B warrants have been exercised or have expired.
42
CYTOMEDIX, INC.
Notes to Financial Statements
(2) These warrants were issued in connection with the Series C stock offering and are voluntarily exercisable at $1.50 per share, provided that the exercise does not result in the holder owning in excess of 9.999% of the outstanding shares of the Company's Common stock, and expire on March 25, 2009. They provide for a cashless exercise at the option of the warrantholder provided that (i) the per share market price of one share of Common stock is greater than the warrant price and (ii) a registration statement for the resale of warrant stock is not in effect. As of December 31, 2006 all class C-1 warrants have been exercised or have expired. The Company has the option to call up to 100% of the C-2 warrants commencing July 29, 2007, provided that the Company's Common stock must have been trading at a closing price greater than $3.00 for 10 consecutive trading days prior to the call notice and a registration statement is then in effect and has been effective without lapse for a period of 60 consecutive days and trading in the Company's stock shall not have been suspended. The Company, upon calling the warrant and the expiration of 20 days without action by the warrant holder, will remit to the holder of the warrant $.01 per called warrant and issue a new warrant representing the number of warrants not subject to the call.
(3) These warrants were issued in exchange for the voluntary exercise of Outstanding Warrants during the offer period ending May 1, 2006 and are voluntarily exercisable at $3.50 per share, provided that the exercise does not result in the holder owning in excess of 9.9% of the outstanding shares of the Company's Common stock, and expire on May 1, 2011. The Company may call up to one hundred percent (100%) of the class D warrants, provided that the Company’s Common stock must have been trading at a closing price greater than $4.50 for a period of at least ten (10) consecutive trading days prior to the date of delivery of the Call Notice, provided that the Registration Statement is then in effect and trading in the Common stock shall not have been suspended by the Securities and Exchange commission or the securities exchange or quotation system on which the Common stock is then listed or traded.
(4) These warrants were issued in connection with the Unit offering (discussed later in this footnote), have a five year term, and are voluntarily exercisable at $1.50 per share, provided that the exercise does not result in the holder owning in excess of 9.999% of the outstanding shares of the Company's Common stock, and expire on March 31, 2009. They provide for a cashless exercise at the option of the warrant provided that (i) the per share market price of one share of Common stock is greater than the warrant price and (ii) a registration statement for the resale of warrant stock is not in effect.
(5) These warrants were issued to placement agents, consultants, and other professional service providers in exchange for services provided. They have terms ranging from 5 to 10 years with various expiration dates through February 24, 2014 and exercise prices ranging from $1.00 to $6.00. The vesting period typically does not exceed the service period. They are voluntarily exercisable once vested. There is no call provision associated with these warrants.
(6) These options were issued under the Company’s shareholder approved Long-Term Incentive Plan. See Note 12 for a full discussion regarding these options.
Activity
The Company issued 2,828,892 shares of Common stock during 2006. The following table lists the sources of and the proceeds from those issuances:
Source | # of Shares | Total Exercise Price | |||||
| Conversion of series A convertible preferred shares | 303 | $ | — | ||||
| Conversion of series B convertible preferred shares | 3,003 | $ | — | ||||
| Exercise of class B warrants | 22,500 | $ | 33,750 | ||||
| Exercise of series C-1 warrants | 548,900 | $ | 823,350 | ||||
| Exercise of series C-2 warrants | 21,750 | $ | 32,625 | ||||
| Exercise of unit offering warrants | 1,355,166 | $ | 2,032,750 | ||||
| Exercise of options issued under the Long-Term Incentive Plan | 79,200 | $ | 118,800 | ||||
| Exercise of other warrants (1) | 798,070 | $ | 798,070 | ||||
| Totals | 2,828,892 | $ | 3,839,345 | ||||
(1) Proceeds include $620,000 in the form of a note receivable. See discussion below.
43
CYTOMEDIX, INC.
Notes to Financial Statements
The Company issued 4,657,941 shares of Common stock during 2005. The following table lists the sources of and the proceeds from those issuances:
Source | # of Shares | Total Exercise Price | |||||
| Conversion of series A convertible preferred shares (1) | 431,528 | — | |||||
| Conversion of series B convertible preferred shares (1) | 449,942 | — | |||||
| Conversion of series C convertible preferred shares | 839,500 | — | |||||
| Exercise of class A warrants | 42,500 | $ | 42,500 | ||||
| Exercise of class B warrants (2) | 507,008 | $ | 673,851 | ||||
| Exercise of class C-1 warrants | 462,900 | $ | 694,350 | ||||
| Exercise of class C-2 warrants | 478,700 | $ | 689,925 | ||||
| Exercise of unit offering warrants | 250,000 | $ | 375,000 | ||||
| Exercise of options issued under the Long-Term Incentive Plan (3) | 252,000 | $ | 367,500 | ||||
| Exercise of other warrants (2) | 870,190 | $ | 356,101 | ||||
| Other issuances | 73,673 | — | |||||
| Totals | 4,657,941 | $ | 3,199,227 | ||||
(1) Includes, but is not limited to, the immediate conversion to Common stock of certain dividends paid in preferred stock.
(2) Includes Common stock issued pursuant to cashless exercise provisions.
(3) The issuance of these shares were registered by the Company’s S-8 filed on November 1, 2004.
The Company has used the cash proceeds from these issuances for general corporate purposes. The issuance of shares under the Company's Long-Term Incentive Plan were registered by the Company's S-8 filed on November 1, 2004. All other shares were issued in private offerings exempt from registration pursuant to Section 4(2) of the Securities Act.
In 2006, the Company granted 500,000 options to purchase the Company’s Common stock with exercise prices ranging from $1.50 to $4.89. These options were granted to employees and board members under the Long-Term Incentive Plan (see Note 12).
On October 1, 2006, FEQ Investments, Inc. (“FEQI”) exercised 775,000 consultant warrants and simultaneously entered into a Negotiable Term Promissory Note and related Security Agreement (the “Note”) with the Company in the amount of $697,500. The exercise price for these options was $1 per share or a total of $775,000. The Note provides for the exercise proceeds to be delivered to the Company in installment payments ending on February 15, 2007. The Note bears interest on the outstanding balance at 6% per year. The Company holds the stock certificate resulting from the exercise as collateral. The outstanding principal balance of $620,000 at December 31, 2006, is reflected as an offset to Stockholder’s equity on the Balance Sheet. See Note 18 to the Financial Statements.
On August 30, 2006, as required by the Certificate of Designation filed with the Delaware Secretary of State, the Company declared a stock dividend on its Series A and B Convertible Preferred shares. This dividend resulted in issuance of 27,869 and 7,036 shares of Series A and B Convertible Preferred stock, respectively, and the issuance of 109 shares of Common stock, in lieu of preferred shares, to prior holders of Series A and B Convertible Preferred shares that were converted to Common stock prior to the payment of the preferred dividends.
On July 31, 2006, the Company’s common stock closed above $3.00 on the American Stock Exchange for the tenth consecutive trading day. As authorized by Section 8 of the Series C-1 Warrants and the Unit Offering Warrants, Cytomedix issued a Call Notice to call all warrants that were eligible and remained outstanding. As a result of an amendment to the terms of the warrant, which was accepted by a majority of the warrantholders, the exercise period was extended, giving the warrantholders until October 20, 2006 to exercise their warrants. The total number of warrants called was 1,605,734 at an exercise price of $1.50 per warrant. Upon expiration of the exercise period on October 20, 2006, 268,900 warrants had been exercised resulting in proceeds of approximately $403,000 to the Company. The remaining 1,336,834 unexercised warrants expired and were cancelled by the Company. Per the terms of the warrant agreements, the Company remitted $0.01 for each expired warrant, or approximately $13,000 in the aggregate.
44
CYTOMEDIX, INC.
Notes to Financial Statements
In May 2006, the Company paid a cash dividend on Series C Convertible Preferred shares at the rate of six percent per annum, amounting to $11,000. The dividends were calculated based on the number of days the shareholder held the Series C Convertible Preferred shares prior to conversion.
In 2005, the Company granted 732,500 options to purchase the Company’s Common stock with exercise prices ranging from $1.15 to $6.00. These options were granted to employees, board members, and professional service providers. Of these, 517,500 were granted under the Long-Term Incentive Plan. The remaining 215,000 grants were as follows:
| · | On March 7, 2005, the Company granted to Kol Bio-Medical Instruments, Inc. a warrant to purchase 60,000 shares of the Company’s Common stock at an exercise price of $2.48. These options vest one year from the date of grant and expire in five years. |
| · | On April 18, 2005, the Company granted to Crystal Research Associates, LLC a warrant to purchase 125,000 shares of the Company’s Common stock at an exercise price of $3.14. These options vested immediately and expire in five years. |
| · | On August 29, 2005, the Company granted to The Wall Street Group, Inc. a warrant to purchase 30,000 shares of the Company’s Common stock at an exercise price of $6.00. These options vested over a one year period and expire in five years. |
On July 15, 2005, the Company entered into a Separation Agreement and Release with William L. Allender. Under said Separation Agreement, Mr. Allender agreed to provide consulting services to the Company as needed and to provide for a smooth transition to his successor. The Company agreed to extend the expiration date of Mr. Allender’s options to purchase Common stock and to allow cashless exercise of said options as part of his severance package. The extension of the expiration date of the options resulted in the Company recording approximately $798,000 of compensation expense in 2005.
In June 2005, the Company paid a cash dividend on Series C Convertible Preferred shares at the rate of six percent per annum, amounting to approximately $91,000. The dividends were calculated based on the number of days the shareholder held the Series C Convertible Preferred shares prior to conversion.
On June 30, 2005, as required by the Certificate of Designation filed with the Delaware Secretary of State, the Company declared a stock dividend on its Series A and B Convertible Preferred shares. This dividend resulted in the issuance of 25,685 and 6,323 shares of Series A and B Convertible Preferred shares respectively, and the issuance of 27,249 shares of Common stock as a result of the automatic conversion of preferred shares issued as dividends to prior holders of Series A and B Convertible Preferred shares who had already converted to Common stock prior to the payment of the preferred dividends (see footnote 1 to the above table).
On June 29, 2004, the Company entered into a termination agreement with Mark Cline that granted him 150,000, five year stock purchase warrants to purchase the Company's Common stock at $1.50 per share in exchange for all previously issued and vested stock options granted to him plus other considerations. On that date, the warrants were valued at $204,000 and the Company recorded deferred compensation of that amount. Subsequently, the Company amortized the entire $204,000, recording the expense to wages and salaries in 2004.
On April 20, 2004, the Company awarded to Dr. Kshitij Mohan 1,000,000, ten year options to purchase the Company’s Common stock for $1.50 per share as part of the two year contract between Dr. Mohan and the Company to accept the position as Chief Executive Officer. At the date of the award, the fair market value was $2.24 per share. In accordance with APB 25, the Company recorded deferred compensation of $740,000 at that date and amortized approximately $370,000 and $258,000 in 2005 and 2004, respectively, recording the expense to wages and salaries. The Company would have recorded $112,000 in 2006 under APB 25.
On January 2, 2004, the Company entered into a termination agreement with Kent Smith that granted him 175,000 warrants to purchase the Company's Common stock at $1.50 per share in exchange for all previously issued and vested stock options granted to him plus other considerations. The warrants vested immediately and expire in three years. The options that were cancelled under the agreement totaled 569,621 of which 403,080 were fully vested. On that date, the warrants were valued at $115,938 and the Company recorded deferred compensation in that amount. Subsequently, the Company amortized the entire $115,938 to compensation expense in 2004.
45
CYTOMEDIX, INC.
Notes to Financial Statements
At December 31, the following amounts were accrued for dividends payable:
2006 | 2005 | ||||||
| Series A Preferred Stock | $ | 14,786 | $ | 13,872 | |||
| Series B Preferred Stock | 3,450 | 3,413 | |||||
| Series C Preferred Stock | — | 10,857 | |||||
| $ | 18,236 | $ | 28,142 | ||||
No dividends were declared or paid on the Company’s Common stock in any of the periods discussed in this report.
Offerings
In March 2004, the Company entered into a Series C Convertible Preferred Stock Purchase Agreement with several accredited investors providing for the sale and issuance of $2.8 million of Series C Convertible Preferred stock (“Series C”) representing 280 shares, at $10,000 per share, of preferred stock which are convertible into 2.8 million shares of Common stock. For each Series C share, the Company issued Series C-1 and Series C-2 warrants allowing the holders thereof to purchase an aggregate of approximately 2.8 million shares of Common stock at an exercise price of $1.50 per share. The characteristics and relative rights of the Series C stock and the basic terms of the C-1 and C-2 warrants are discussed earlier in this footnote. Pursuant to a registration rights agreement, the Company subsequently registered the resale of the Common stock issuable upon conversion of the Series C Convertible Preferred stock and the Common stock issuable upon exercise of the Series C-1 and Series C-2 warrants. The Company received proceeds from this placement, net of commissions and expenses of $325,807, of $2,474,193. In addition to the commissions, the placement agent also received five-year warrants to purchase 280,000 shares of the Company's Common stock at an exercise price of $1.00 per share and was awarded a six-month consulting agreement for future financing services in return for compensation of $5,000 per month and additional warrants to purchase 100,000 shares of Common stock at an exercise price of $1.00 per share.
The Company’s stock price on March 26, 2004 was $2.06; consequently, pursuant to the requirements of EITF 98-5 “Accounting for Convertible Securities with Beneficial Conversion Features or Contingently Adjustable Conversion Ratios” (“EITF 98-5”), as amended by EITF 00-27 “Application of Issue 98-5 to Certain Convertible Instruments”, the issuance of the Series C stock, which are convertible initially at $1.00 per share at any time, resulted in a beneficial conversion feature (the difference between the market price and the conversion price) recorded as a preferred stock dividend in the amount of $2,800,000.
Simultaneous with the Series C placement, the Company commenced a separate private placement in which the Company offered for sale 4,500 units at $1,000 per unit (the “Unit Offering”), in return for a total of $4,500,000, consisting of cash and negotiable subscription promissory notes receivable. Each unit consists of 1,000 shares of Common stock and a five-year warrant (the “unit warrants”) to purchase an additional 1,000 shares at $1.50 per share. The characteristics and relative rights of the Common stock and the basic terms of the unit warrants are discussed earlier in this footnote. As compensation for their services, certain broker-dealers received cash commissions in the amount of 10% of the gross proceeds. The Company incurred $38,825 of professional fees related to this placement which have been netted against the proceeds. At December 31, 2004, the Company had subscriptions receivable of $825,000 plus accrued interest, all of which was received in 2005.
On May 1, 2006, the Company completed a Class D Warrant Offer whereby, for each $7.50 of Outstanding Warrants exercised by warrantholders during the offer period, the Company issued one Class D Warrant which the holder may exercise for one share of Cytomedix Common Stock at an exercise price of $3.50. These Class D Warrants have a five year term and are callable at the Company’s discretion if the closing price of the Company’s Common Stock is at least $4.50 for 10 consecutive trading days and certain other conditions are met. Through this offer, the Company received exercises of Outstanding Warrants totaling approximately $2,280,000 and issued 304,033 Class D Warrants. These Class D Warrants carry piggyback registration rights whereby the Company must include the shares underlying these Class D Warrants on any registration statement filed by the Company after the closing of the Offering.
46
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 12 - LONG-TERM INCENTIVE PLAN
Cytomedix has a shareholder-approved, Long-Term Incentive Plan (“LTIP”) that permits incentive awards of options, SARs, restricted stock awards, phantom stock awards, performance unit awards, dividend equivalent awards and other stock-based awards. Cytomedix may issue up to 5,000,000 shares of stock under this LTIP. At December 31, 2006, 1,411,423 shares were available for future grants. Of all options granted through December 31, 2006, 386,200 had been exercised and 3,202,377 remained outstanding. Option terms are set by the Board of Directors for each option grant, and generally vest immediately upon grant or over a period of time ranging up to three years, are exercisable in whole or installments, and expire ten years from the date of grant. These options expire at various dates through October 11, 2016.
A summary of the status of Cytomedix’s LTIP at December 31, 2004, 2005 and 2006, and changes during the periods then ended, is presented in the tables below:
Number of Shares | Weighted Average | ||||||
| Balance at December 31, 2003 | 1,840,698 | $ | 1.50 | ||||
| Granted | 1,480,000 | $ | 1.52 | ||||
| Cancelled | (749,121 | ) | $ | 1.48 | |||
| Exercised | (55,000 | ) | $ | 1.50 | |||
| Balance at December 31, 2004 | 2,516,577 | $ | 1.51 | ||||
| Granted | 517,500 | $ | 1.52 | ||||
| Cancelled | — | — | |||||
| Exercised | (252,000 | ) | $ | 1.46 | |||
| Balance at December 31, 2005 | 2,782,077 | $ | 1.68 | ||||
| Granted | 500,000 | $ | 2.25 | ||||
| Cancelled | (500 | ) | $ | 1.90 | |||
| Exercised | (79,200 | ) | $ | 1.50 | |||
| Balance at December 31, 2006 | 3,202,377 | $ | 1.82 | ||||
47
CYTOMEDIX, INC.
Notes to Financial Statements
A summary of option activity under the LTIP as of December 31, 2006, and changes during the year then ended is presented below:
Options | Shares | Weighted-Average Exercise Price | Weighted-Average Remaining Contractual Term | Aggregate Intrinsic Value | |||||||||
| Outstanding at January 1, 2006 | 2,782,077 | $ | 1.68 | ||||||||||
| Granted | 500,000 | $ | 2.25 | ||||||||||
| Exercised | (79,200 | ) | $ | 1.50 | |||||||||
| Forfeited or expired | (500 | ) | $ | 1.90 | |||||||||
| Outstanding at December 31, 2006 | 3,202,377 | $ | 1.82 | 7.3 | $ | 0 | |||||||
| Exercisable at December 31, 2006 | 2,944,046 | $ | 1.71 | 7.1 | $ | 0 | |||||||
The following table summarizes information about stock options outstanding as of December 31, 2006:
Options Outstanding | Options Exercisable | |||||||||||||||
Range of Exercise | Number of Outstanding | Weighted Average | Weighted Average | Number Exercisable | Weighted Average | |||||||||||
| $1.15 - $1.50 | 2,384,877 | 6.7 | $ | 1.49 | 2,384,877 | $ | 1.49 | |||||||||
| $2.06 - $2.73 | 645,000 | 9.2 | $ | 2.42 | 476,668 | $ | 2.37 | |||||||||
| $3.14 - $6.00 | 172,500 | 8.5 | $ | 4.08 | 82,501 | $ | 4.09 | |||||||||
The weighted-average grant-date fair value of stock options granted under the LTIP during the years 2006, 2005, and 2004 was $2.33, $2.74, and $1.46, respectively. The total intrinsic value of stock options exercised under the LTIP during the fiscal years ended December 31, 2006, 2005, and 2004, was $126,000, $916,000, and $36,000, respectively.
As of December 31, 2006, there was $655,170 of total unrecognized compensation cost related to nonvested stock options granted under the LTIP. That cost is expected to be recognized over a weighted-average period of 1.7 years. The total fair value of stock options granted under the LTIP that vested during the fiscal years ended December 31, 2006, 2005, and 2004, was $1,421,000, $1,459,000, and $1,475,000, respectively.
On July 15, 2005, the Company entered into a Separation Agreement and Release with William L. Allender. Under said Separation Agreement, Mr. Allender agreed to provide consulting services to the Company as needed. The Company agreed to extend the contractual life of 150,000 fully vested options to purchase Common stock held by Mr. Allender's and to allow cashless exercise of said options as part of his severance package. As a result of that modification, the Company recognized additional compensation expense of $798,000 for the fiscal year ended December 31, 2005.
48
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 13 - DEFERRED COMPENSATION
The components of deferred compensation for the options granted are as follows at December 31, 2005:
| Beginning balance | $ | 567,788 | ||
| Deferred compensation recorded | 1,652,666 | |||
| Amortization of stock-based compensation | (1,981,653 | ) | ||
| $ | 238,801 |
Due to the adoption of FAS123R on January 1, 2006, the above balance was re-classified to “Additional paid-in capital”.
NOTE 14 - SUPPLEMENTAL CASH FLOW DISCLOSURES - NON-CASH TRANSACTIONS
Non-cash transactions for years ended December 31 include:
2006 | 2005 | 2004 | ||||||||
| Commissions paid in stock | $ | — | $ | — | $ | 15,374 | ||||
| Accrued dividends on 6% preferred stock | — | — | 79,195 | |||||||
| Accrued dividends on 8% preferred stock | 36,361 | 84,902 | 240,665 | |||||||
| Beneficial conversion feature of Series C | ||||||||||
| Preferred stock recorded as preferred | ||||||||||
| stock dividend | — | — | 2,800,000 | |||||||
| Stock issued for executive recruitment fees | — | 35,000 | — | |||||||
| Stock issued for contract litigation settlement | — | 227,500 | — | |||||||
| Preferred dividends paid by issuance of stock | 35,232 | 166,063 | 240,704 | |||||||
| Note received for Common stock issued on | ||||||||||
| warrant exercise | 620,000 | — | — | |||||||
Cash paid for interest and taxes was $0 in 2006, 2005, and 2004, respectively.
NOTE 15 - RELATED PARTY TRANSACTIONS
BDR, Inc. (“BDR”) is a consulting firm owned solely by Jimmy D. Swink, Jr. The Company entered into a consulting agreement with BDR, dated July 11, 2002. Under this agreement, the Company granted BDR stock options representing the right to purchase 300,000 shares of the Company’s Common stock at $1.50 per share (the fair market value on the date of grant). Additionally, in February 2004, the Company issued 10-year warrants to purchase an additional 200,000 shares of Common stock at $1.50 to BDR, in connection with the consulting agreement. All such options and warrants are fully vested as of December 31, 2006. Pursuant to extensions, this consulting agreement expired on August 31, 2006. Under the consulting agreement, BDR received compensation totaling $35,000, $186,000, and $412,000 (of which $0, $78,000, and $289,000 was equity-based compensation valued in accordance with SFAS123) for services rendered in the years ended 2006, 2005, and 2004, respectively.
In 1999, the founder and then sole stockholder of the Company, Charles Worden, and the Company entered into an agreement where the Company was to pay Mr. Worden a royalty. Mr. Worden and the Company entered into a substitute royalty agreement with court approval on November 14, 2001, which superseded the agreement dated October 29, 1999. The Company agreed to pay Mr. Worden a royalty of five percent of the gross profit from the sale, licensing or other exploitation of the Worden Patent. The royalty payment during any calendar year was limited to $600,000. The Company was to pay Mr. Worden a minimum royalty of $6,250 per month in advance. For the years ended December 31, 2006, 2005 and 2004, the total royalty expense was $75,000 per year. The Company granted a security interest and lien in the Worden Patent. In addition, the Company granted a reversionary interest in the patent if the Company discontinues substantially all efforts to commercialize the Worden Patent.
49
CYTOMEDIX, INC.
Notes to Financial Statements
The Carmen Group, Inc. was engaged during the second quarter of 2003 as a business consultant to strategically position and represent the Company before the federal government. A former director of the Company, Mr. Robert Burkett, was also a senior consultant with the Carmen Group, Inc. during the term of the agreement. Effective October 1, 2003, a formal agreement was signed with the Carmen Group, Inc. for a period of one year to provide services for the Company for a flat fee of $15,000 per month plus expenses. Additionally, the agreement stipulated that the Company would issue to the Carmen Group, Inc. an option to purchase 100,000 shares immediately exercisable at $1.25 with an additional 100,000 shares being issuable one year from the date of agreement at an exercise price of $2.00. In 2004, the Carmen Group provided services to the Company amounting to $377,000, which included $240,000 of stock-based compensation. The Company did not incur any expenses in 2005 or 2006 relating to this agreement as this agreement expired September 30, 2004, and was neither renewed nor extended.
NOTE 16 - OPERATING LEASES
The Company leases its office space under an operating lease expiring in July 2008, with future minimum lease payments as indicated in the table below:
| Years ending December 31: | ||||
| 2007 | $ | 64,658 | ||
| 2008 | 38,369 | |||
| Thereafter | — | |||
| Total future minimum lease payments | $ | 103,027 | ||
For the years ended December 31, 2006, 2005 and 2004, the Company incurred rent expense of approximately $63,000, $44,000 and $32,000, respectively.
NOTE 17 - COMMITMENTS AND CONTINGENCIES
Under the Company’s plan of reorganization upon emergence from bankruptcy in July 2002, the pre-bankruptcy Series A Preferred stock and the dividends accrued thereon that existed prior to emergence from bankruptcy are exchanged into one share of new Common stock for every five shares of pre-bankruptcy Series A Preferred shares held as of the date of emergence from bankruptcy. This exchange is contingent on the successor Company’s attaining aggregate gross revenues for four consecutive quarters of at least $10,000,000 prior to July 2009 and would result in the issuance of approximately 350,000 shares of Common stock.
Under a distributor agreement, title passes when invoiced product is received by the distributor or its designee in acceptable condition. Cytomedix recognizes revenue when shipped and does not reserve for defective merchandise. Historically, returns for defective merchandise from all customers have been negligible.
NOTE 18 - SUBSEQUENT EVENTS
In January 2007, the Board of Directors authorized the grant of certain stock options under the Long-Term Incentive Plan to board members for their upcoming service in 2007. These options have an exercise price of $1.10, vest in equal monthly installments through December 2007, and expire ten years from the date of grant.
In February 2007, the terms of the Note from FEQ Investments were amended to accelerate a portion of the principal payments and extend the remainder. As amended, the final installment payment is due by April 30, 2007. All other terms of the Note remain unchanged and in full force and effect.
50
CYTOMEDIX, INC.
Notes to Financial Statements
NOTE 19 - QUARTERLY FINANCIAL DATA (UNAUDITED)
First Quarter | Second Quarter | Third Quarter | Fourth Quarter | ||||||||||
2006 | |||||||||||||
| Revenues | $ | 485,537 | $ | 411,324 | $ | 574,091 | $ | 477,203 | |||||
| Gross profit | $ | 221,975 | $ | 188,038 | $ | 350,773 | $ | 172,784 | |||||
| Net income (loss) | $ | (1,214,998 | ) | $ | 801,155 | $ | (1,032,850 | ) | $ | (816,263 | ) | ||
| Income (loss) per common share-- | |||||||||||||
| Basic | $ | (0.05 | ) | $ | 0.03 | $ | (0.04 | ) | $ | (0.03 | ) | ||
| Diluted | $ | (0.05 | ) | $ | 0.02 | $ | (0.04 | ) | $ | (0.03 | ) | ||
2005 | |||||||||||||
| Revenues | $ | 271,174 | $ | 305,129 | $ | 446,384 | $ | 491,738 | |||||
| Gross profit | $ | 116,733 | $ | 101,123 | $ | 251,102 | $ | 164,143 | |||||
| Net loss | $ | (2,096,729 | ) | $ | (1,269,514 | ) | $ | (2,281,948 | ) | $ | (855,294 | ) | |
| Loss per common share-- | |||||||||||||
| Basic and diluted | $ | (0.10 | ) | $ | (0.05 | ) | $ | (0.09 | ) | $ | (0.03 | ) | |
51
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures
Under the supervision and with the participation of management, including the Chief Executive Officer (“CEO”) and Chief Financial Officer (“CFO”), the Company conducted an evaluation of its disclosure controls and procedures, as such term is defined under Rule 13a-15(e) promulgated under the Securities Exchange Act of 1934, as amended (the “Exchange Act”) as of December 31, 2006. Based on this evaluation, its CEO and CFO concluded the Company's disclosure controls are not effective.
Management’s Report on Internal Control over Financial Reporting
The Company’s management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15(f). Under the supervision and with the participation of its management, including the CEO and CFO, the Company conducted an evaluation of the effectiveness of its internal control over financial reporting based on the framework in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this evaluation under the framework in Internal Control—Integrated Framework, management concluded that the Company’s internal control over financial reporting was not effective as of December 31, 2006.
Two material weaknesses were noted based on evidence of errors in application of controls over financial disclosures and in recording stock-based compensation expense. The financial statement disclosure errors related primarily to SFAS109, Accounting for Income Taxes, and the recently adopted SFAS123R, Share-Based Payment. While a disclosure checklist was utilized for year-end reporting, certain disclosures were not included or properly prepared. Errors were also noted in the recording of stock-based compensation related to one set of option grants.
Management is in the process of evaluating these issues and will take remedial action in 2007.
Management’s assessment of the effectiveness of internal control over financial reporting as of December 31, 2006, has been audited by L J Soldinger Associates, LLC, an independent registered public accounting firm, as stated in their report as set forth on the following page.
Changes in Internal Control over Financial Reporting
The Company made the following changes in its internal controls over financial reporting during the quarterly period ended December 31, 2006, that have materially affected, or are reasonably likely to materially affect, its internal control over financial reporting:
| § | Created formal documentation of internal control structure and testing of key internal controls |
| § | Implemented system of mandatory signatures to evidence preparation and review of items that encompass certain key controls |
| § | Formalized a company-wide authority matrix regarding contracts, purchasing, and invoice approval |
| § | Developed checklists to be used at certain regular meetings to ensure comprehensive discussion of internal controls |
52
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and
Shareholders of Cytomedix, Inc.
We have audited management's assessment, included in the accompanying section Item 9A. Control and Procedures, that Cytomedix, Inc. did not maintain effective internal control over financial reporting as of December 31, 2006, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Cytomedix, Inc.'s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting. Our responsibility is to express an opinion on management's assessment and an opinion on the effectiveness of the company's internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, evaluating management's assessment, testing and evaluating the design and operating effectiveness of internal control, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company's internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
A material weakness is a control deficiency, or combination of control deficiencies, that results in more than a remote likelihood that a material misstatement of the annual or interim financial statements will not be prevented or detected. The following two material weaknesses have been identified and included in management's assessment. The material weaknesses were based on evidence of errors in application of controls over financial disclosures and in recording stock-based compensation expense. The financial statement disclosure errors related primarily to SFAS 109, Accounting for Income Taxes, and the recently adopted SFAS 123R, Share-Based Payment. While a disclosure checklist was utilized for year-end reporting, certain required disclosures were not included or properly prepared. Errors were also noted in the calculation and recording of stock-based compensation related to one set of option grants.
These material weaknesses were considered in determining the nature, timing, and extent of audit tests applied in our audit of the 2006 financial statements, and this report does not affect our report dated February 23, 2007 on those financial statements.
In our opinion based on our audit, management's assessment that Cytomedix, Inc. did not maintain effective internal control over financial reporting as of December 31, 2006, is fairly stated, in all material respects, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Also, in our opinion, because of the effect of the material weakness described above on the achievement of the objectives of the control criteria, Cytomedix, Inc. has not maintained effective internal control over financial reporting as of December 31, 2006 based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the balance sheets and the related statements of operations, stockholders' equity and cash flows of Cytomedix, Inc., and our report dated February 23, 2007 expressed an unqualified opinion.
L J SOLDINGER ASSOCIATES LLC
Deer Park, Illinois, USA
February 23, 2007
53
ITEM 9B. OTHER INFORMATION
None.
54
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The Company’s directors, executive officers and significant employees are listed below:
Name | Age | Date of Election or Appointment | Position(s) with the Company | |||
| James S. Benson | 67 | November 1, 2004 | Director | |||
| David P. Crews | 44 | September 28, 2001 | Director | |||
| Arun K. Deva | 62 | November 23, 2004 | Director | |||
| David F. Drohan | 68 | July 12, 2004 | Director | |||
| Mark T. McLoughlin | 51 | June 7, 2004 | Director | |||
| Kshitij Mohan | 61 | April 20, 2004 | Chairman of the Board | |||
| Chief Executive Officer | ||||||
| Andrew S. Maslan | 37 | August 15, 2005 | Chief Financial Officer | |||
| Carelyn P. Fylling | 59 | December 1, 2001 | Vice President of Professional Services |
JAMES S. BENSON has served as a Director since November 1, 2004. Mr. Benson has over 25 years of experience in the healthcare industry, and also serves as a director of Cryolife, Inc. Recently, he retired from the Advanced Medical Device Association (AdvaMed) where he served as executive vice president for technical and regulatory affairs. Prior to that, he held numerous senior positions at the FDA over a twenty year period. He retired from the FDA as director of the Center for Devices and Radiological Health (CDRH). Earlier, he served as deputy commissioner of the FDA, and also as its commissioner for a one-year period. During his tenure with the FDA, Mr. Benson worked closely with other Federal agencies and worked with Congress to craft and create various pieces of legislation including "The Food and Drug Modernization Act of 1997", "The Biomaterials Access Act of 1998" and "The Medical Device User Fee and Modernization Act of 2002". Mr. Benson earned a B.S. degree in civil engineering from the University of Maryland and a M.S. degree in nuclear engineering from the Georgia Institute of Technology.
DAVID P. CREWS has served as a Director since his election through the consent solicitation that became effective on September 28, 2001. Mr. Crews is executive vice president of Crews and Associates, Inc., a brokerage house located in Little Rock, Arkansas, founded by his father. Mr. Crews has worked at Crews & Associates for more than 22 years, specializing in the fixed income markets. He is a former partner of All American Leasing, a municipal finance firm, and also serves as vice president, secretary, and treasurer of CHASC, Inc., an entity that acquired Smith Capital Management (an investment advisory firm). Mr. Crews is also a Board Member of Pure Energy Group, Inc. (an oil and gas company).
ARUN K. DEVA has served as a Director since November 23, 2004. Mr. Deva is the founder and President of Deva & Associates, P.C., a Rockville, Maryland based mid-size accounting and consulting firm that provides accounting, auditing, litigation support, due diligence, cost-benefit analysis and other financial consulting services to many Federal agencies and corporations. He is also the founder and President of CPAMoneyWatch.com, LLC, a web based business services provider offering online accounting and business solutions to small and mid-sized businesses. Prior to establishing Deva & Associates in 1991, Mr. Deva was a partner at Touche Ross & Co. (now Deloitte & Touche). He has served as a management consultant for several public and private companies with a focus on financial restructurings, negotiations with lenders and creditors, financial reporting and disclosures, and filings with the Securities and Exchange Commission. Mr. Deva is a member of the American Institute of Certified Public Accountants, Maryland Association of Certified Public Accountants and Association of Government Accountants. He was appointed to the Maryland Banking Board by the Governor of Maryland for a six-year term ending in 2008. Mr. Deva earned his Bachelor of Commerce degree in accounting from St. Xavier's College in India and a Masters of Business Administration degree in Finance from Indiana University, Bloomington, Indiana.
DAVID F. DROHAN has served as a Director since July 12, 2004. Mr. Drohan recently retired from Baxter Healthcare Corporation where he served as Senior Vice President and President of Baxter's medication delivery business, a position he held since May 2001. In this capacity, he had direct general management responsibility for the development and worldwide marketing of intravenous products, drug-delivery and automated distribution systems, as well as anesthesia, critical care and oncology products representing $4 billion in combined annual sales. He joined Baxter in 1965 as a territory manager in New York and throughout the years has held a succession of senior positions. Prior to joining Baxter, Mr. Drohan worked for Proctor & Gamble. He is currently a director of Cytomedix, Inc. as well as a director of Pharmedium Health Care Corp. He is a trustee of Parents Project Muscular Dystrophy and a director of the Baxter Credit Union. He earned his bachelor's degree in industrial relations from Manhattan College, New York.
55
MARK T. McLOUGHLIN has served as a Director since June 7, 2004. Mr. McLoughlin currently serves as Sr. Vice President, Chief Marketing Officer - International for Cardinal Health, Inc., one of the world's largest health care manufacturing and distribution companies. In this capacity, he has strategic responsibility for the entire International Marketing organization for Cardinal Health based in Geneva Switzerland. Prior to joining Cardinal, he was vice president of commercial operations for Norwood Abbey Ltd., an Australian-based medical technology company. Earlier, he was President of North American operations for Ion Beam Application, Inc., a Belgium-based global medical technology company. His executive career experience also includes Mallinckrodt, as well as positions with other healthcare companies.
DR. KSHITIJ MOHAN was appointed as Chief Executive Officer on April 20, 2004 and has served as a Director since May 7, 2004 and Board Chairman since July 12, 2005. Prior to assuming his positions in the Company, Dr. Mohan served as Chief Executive officer of International Remote Imaging Systems, Inc., the predecessor company of IRIS International. Previously, he was the Chief Regulatory and Technology Strategist for the Law Firm of King and Spalding, Senior Vice-President and Chief Technology Officer for Boston Scientific Corporation, and Corporate Vice-President of Baxter International, responsible for all corporate research and technical services and was a member of the Baxter operating management team. Prior to entering the private sector, Dr. Mohan served in various capacities within the U.S. Food and Drug Administration, including leading the science and technology programs and the office of product evaluation and approval of medical devices and between 1979 - 1983 served in the White House Office of Management and Budget with responsibilities for the national R & D policies, programs of the National Science Foundation and NASA's Aeronautical and Space Research and Technology programs. Dr Mohan has been widely published in the field of health policies, regulations and Applied Physics and served on numerous Boards including the Corporate Advisory Boards of the Schools of Engineering at Dartmouth College and the University of California at Riverside. Dr. Mohan earned a PH.D. degree in Physics from Georgetown University, a M.S. degree in Physics from the University of Colorado and a B.Sc., First Class Honors, Patna University, Patna, India.
ANDREW S. MASLAN joined the company as corporate controller on July 1, 2005, and became our Chief Financial Officer on August 15, 2005. Mr. Maslan most recently served as controller for BioReliance Corporation based in Rockville, Maryland, which was acquired by Invitrogen (Nasdaq: IVGN) in February 2004. Earlier, he held positions with two other Rockville, Maryland-based companies, serving as a principal with GlobeTraders, Inc., and senior accountant for Providence Laboratory Associates. Mr. Maslan began his professional career serving as an auditor with KPMG Peat Marwick, and is a certified public accountant licensed in the state of Maryland.
CARELYN P. FYLLING, RN, MSN, has served as the Company's Vice President of Professional Services since December 2001. Ms. Fylling was director of training and program development at the International Diabetes Center in Minneapolis, Minnesota. She also has served on the national Board of Directors of the American Diabetes Association and numerous national committees of the American Diabetes Association. Ms. Fylling received the prestigious Ames Award for Outstanding Educator in the Field of Diabetes. Subsequently, she joined Curative Health Services and helped the company grow from three employees to over 650 employees. During her 13 years at Curative, Ms. Fylling helped to design the national wound database, developed clinical protocols, conducted outcome studies, trained physicians and nurses in comprehensive wound management, wrote scientific articles and abstracts, assisted in clinical trials and marketing, and developed an Internet-based online wound care training program for health professionals. Subsequently, she provided independent consulting and outsourcing services to the health care industry through Fylling Associates, LLC, which she wholly owns, and through Strategic Partners, LLC, in which she held a partnership interest.
Audit Committee
At a meeting of the Board of Directors in December 2004, the Board formed an Audit Committee. Mr. Arun K. Deva serves as chairman of the Audit Committee and is the audit committee financial expert. The Board has determined that Mr. Deva is “independent” as defined by section 121(A) of the listing standards of the American Stock Exchange and Item 7(d)(3)(iv) of Schedule 14A of the Securites Exchange Act of 1934. Other members of the Audit Committee are Messrs. David P. Crews and David F. Drohan.
56
Code of Conduct and Ethics
In April 2005, the Board approved a Code of Conduct and Ethics applicable to all directors, officers and employees which complies with Section 807 of the American Stock Exchange Corporate Governance Requirements and with the definition of a "code of ethics" as set forth in Item 406 of SEC Regulation S-K. A copy of this Code of Conduct is available at the Company’s website at www.cytomedix.com ,and is available at no charge by contacting the Company at it’s headquarters as listed on the cover page of this report.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Act requires officers, directors and persons who own more than ten percent of a registered class of equity securities to, within specified time periods, file certain reports of ownership and changes in ownership with the SEC. The Company is not aware of any failure to file initial statements of beneficial ownership of securities (Forms 3) or change in beneficial ownership reports or report transactions (Forms 4) in a timely manner during the fiscal year ended December 31, 2006, by any of the current directors or executive officers.
ITEM 11. EXECUTIVE COMPENSATION
During 2006, the Company had the following executive officers: Kshitij Mohan, CEO; Andrew S. Maslan, Chief Financial Officer; Carelyn P. Fylling, Vice President of Professional Services.
Compensation Discussion and Analysis
The Compensation Committee has responsibility for reviewing and making recommendations to the Board of Directors with respect to the Company’s overall executive compensation policy, including such items as (i) the annual base salary, annual bonus, and annual and long-term equity-based or other incentives of each corporate officer, including the CEO; (ii) corporate goals and objectives relevant to each executive officer’s compensation, evaluate each executive officer's performance in light of those goals and objectives, and recommend each executive officer's compensation level based on this evaluation, which recommendation will be subject to approval by the full Board; and (iii) any other matter, such as severance agreements, change in control agreements, or special or supplemental executive benefits, within the Committee's authority:
The overall compensation policy, which is applicable to the Cytomedix executive officers, is to position the aggregate of the compensation components at a level commensurate with the Company’s size and performance relative to similar companies. The Compensation Committee seeks to make compensation decisions consistent with the long-term growth and performance objectives of the Company. The Compensation Committee implements its compensation policy in a manner designed to maximize shareholder benefit by aligning the interests of employees with the interests of shareholders through the award of stock options and motivating executive officers by rewarding them based on performance.
The Company is committed to providing a competitive pay program that is fair, non-discriminatory, and attractive to quality personnel. Further, the Cytomedix pay program is structured to achieve motivation of its employees and efficient performance. Accordingly, Cytomedix has structured its pay program to achieve these goals through an appropriate mix of cash and equity-based compensation as well as productivity-based awards.
The Company’s executive compensation program currently consists primarily of salary, annual peformance bonuses and incentive awards in the form of stock options under the Company’s Long-Term Incentive Plan. The Compensation Committee believes that stock options awarded under the Cytomedix Long-Term Incentive Plan provide the most useful incentive to encourage executive officers and other employees to maximize productivity and efficiency because the value of such options relates to the Company’s stock price. Awards under the Long-Term Incentive Plan have the effect of more closely aligning the interests of the Company’s employees with its shareholders, while at the same time offering an attractive vehicle for the recruitment, retention, and compensation of employees.
The CEO’s annual performance bonus is dependent on his performance against pre-determined management business objectives (“MBO’s”). The MBO’s in effect for the CEO’s most recently completed employment year related to the following general areas:
| § | Patents and Licensing |
| o | Resolution of actual and potential infringement matters |
| o | Development and implementation of utilization strategies |
| § | Product Development |
| o | Completion of clinical trial and submission of preliminary results |
| o | Development and implementation of regulatory strategy |
| o | Completion of a pharmaco-economic study |
| o | Development of a strategy for submission to the Center of Medicare and Medicaid Services |
| § | Sales and Marketing |
| o | Finalize potential sales contracts and further development of independent slaes representative network |
| § | Financial Performance |
| o | Increase gross revenues |
| o | Secure additional financing, if necessary |
| o | Further development of strategic plan |
57
While based generally on the CEO’s merit and performance and considering his contribution to Cytomedix’s success, the specific performance criteria are set by the Compensation Committee and approved by the Board of Directors on an annual basis to reflect an appropriate balance of the Company’s short-term and long-term goals.
Goals and objectives for other executive officers are designed to facilitate the achievement of the overarching Company goals reflected in the CEO’s MBO’s.
Summary Compensation and Grants
Summary Compensation Table for the year ended December 31, 2006
Name and Principal Position | Year | Salary | Bonus | Option Awards | All Other Compensation | Total | ||||||||||||||
| Kshitij Mohan | (1) | 2006 | $ | 323,549 | $ | 150,000 | $ | 311,503 | $ | 25,619 | $ | 810,671 | ||||||||
| Chief Executive Officer | ||||||||||||||||||||
| (Effective April 1, 2004) | ||||||||||||||||||||
| Andrew S. Maslan | (2) | 2006 | 148,500 | 27,700 | 160,304 | 844 | 337,348 | |||||||||||||
| Chief Financial Officer | ||||||||||||||||||||
| (Effective August 16, 2005) | ||||||||||||||||||||
| Carelyn P. Fylling | (3) | 2006 | 136,500 | — | 15,059 | 169 | 151,728 | |||||||||||||
| VP Professional Services | ||||||||||||||||||||
(1) Upon acceptance of the position of Chief Executive Officer, Dr. Mohan was awarded 1,000,000 ten-year options to purchase the Company’s Common stock for $1.50. Under the terms of his employment agreement, 500,000 options vested immediately, 250,000 vested in April 2005 and the remaining options will vest in April 2006. Also, pursuant to this agreement, upon reaching the first and second anniversary dates of his agreement, Dr. Mohan received 100,000 ten-year options at $1.50. Amounts in the All Other Compensation column consist of $25,000 that Dr. Mohan received as a “perk package and $619 in company paid life insurance premiums for Dr. Mohan’s benefit.
(2) Pursuant to his employment agreement as amended, in 2005 Mr. Maslan was granted 60,000 ten-year options to purchase shares of the Company’s Common stock at an exercise price of $5.07 per share and, in 2006 Mr. Maslan was granted 40,000 and 50,000 ten-year options to purchase shares of the Company’s Common stock at exercise prices of $2.23 and $2.75 per share, respectively. Options vest at intervals through October 2009. Amounts in the All Other Compensation column consist of $844 in company paid life insurance premiums for Mr. Maslan’s benefit.
(3) In 2006, Ms. Fylling was granted 20,000 ten-year options to purchase shares of Common stock at $2.40. Amounts in the All Other Compensation column consist of $169 in company paid life insurance premiums for Ms. Fylling’s benefit.
58
Grants of Plan-Based Awards in 2006
Name | Grant Date | Date Board Took Action to Grant Award | Option Awards: Number of Securites Underlying Options | Exercise Price of Option Awards | Grant Date Fair Value of Option Awards | |||||||||||
| Kshitij Mohan | 8/17/2006 | 4/19/2004 | 100,000 | $ | 1.50 | $ | 207,700 | |||||||||
| Andrew S. Maslan | 3/17/2006 | 3/17/2006 | 40,000 | $ | 2.23 | $ | 94,800 | |||||||||
| 10/11/2006 | 10/11/2006 | 50,000 | $ | 2.73 | $ | 127,000 | ||||||||||
| Carelyn P. Fylling | 1/12/2006 | 1/12/2006 | 20,000 | $ | 2.40 | $ | 45,200 | |||||||||
Narrative Disclosure to Summary Compensation and Grants
The Company has the following employment agreements with its executive officers:
Kshitij Mohan: On April 20, 2004, the Company entered into an employment contract with Dr. Kshitij Mohan to serve as Chief Executive Officer. The employment contract had an initial term of two years. The term is automatically extended by one year increments on each anniversary of the effective date unless the contract is otherwise terminated in accordance with its provisions. As an inducement to enter this agreement, Dr. Mohan received 1,000,000 ten-year stock options at an exercise price of $1.50 per share. Of these options, 500,000 became immediately exercisable with the remaining 250,000 becoming exercisable on the first anniversary of the agreement, and the remaining 250,000 becoming exercisable on the second anniversary. Dr. Mohan’s base salary for the first contract year was $275,000, increasing by at least 10% on each anniversary of the agreement. At December 31, 2006, Dr. Mohan’s annual base salary was $332,750. Pursuant to this agreement, Dr. Mohan was entitled to and received 100,000 options upon each of the first and second anniversary dates of this agreement. These options had a ten-year term and an exercise price of $1.50 per share. Additionally, in each of the first and second years of this agreement, Dr. Mohan received $150,000 cash bonus upon the achievement of performance criteria agreed upon by Dr. Mohan and the Board of Directors. In employment years ending April 20, 2007 and beyond, Dr. Mohan is eligible for an annual bonus at the discretion of the Board of Directors, upon the achievement of mutually agreed-upon performance criteria. Dr. Mohan also receives a guaranteed “perk package” of $25,000 to be paid at the beginning of each year under the term of this agreement.
Andrew Maslan: On June 3, 2005, the Company entered into an employment agreement with Mr. Andrew S. Maslan to serve as Corporate Controller. Employment was at will, with certain notification provisions. Mr. Maslan’s base salary was $135,000, subject to review at the end of the first calendar year. Mr. Maslan’s annual target bonus percentage was 20%, depending on the achievement of performance criteria. Mr. Maslan was also granted 60,000 ten-year options to purchase shares of the Company’s Common stock at an exercise price of $5.07. In October 2006, this agreement was amended to increase Mr. Maslan’s annual base salary to $155,000 and target bonus percentage to 25% and provided for the grant of an additional 50,000 10-year options to purchase the Company’s stock at a price of $2.73 per share. Additional grants of options or increases to base salary may be considered annually, as of the anniversary date of the amended agreement, or in the ordinary course of business at the discretion of the CEO and Board of Directors.
Carelyn Fylling: On September 4, 2002, the Company entered into an employment agreement with Ms. Carelyn P. Fylling to serve as Vice President of Professional Services. The term was for a period of one year, renewable on the first anniversary for a period of two years and in one year increments thereafter. Under the agreement, Ms. Fylling's base salary was $130,000, subject to increase upon review by the Board at the end of each calendar year. Stock options and annual bonus are at the discretion of the Board. Other benefits are in accordance with Company policy.
59
Outstanding Equity Awards at December 31, 2006
Option Awards | ||||||||||||||
Name | Number of Securities Underlying Unexercised Options Exercisable (1) | Number of Securities Underlying Unexercised Options Unexercisable | Option Exercise Price | Option Expiration Date | ||||||||||
| Kshitij Mohan | 990,000 | — | $ | 1.50 | 4/20/2014 | |||||||||
| 100,000 | — | $ | 1.50 | 6/6/2015 | ||||||||||
| 100,000 | — | $ | 1.50 | 8/17/2016 | ||||||||||
| Andrew S. Maslan | 30,000 | 30,000 | (2) | $ | 5.07 | 1/11/2016 | ||||||||
| 13,334 | 26,666 | (3) | $ | 2.23 | 3/16/2016 | |||||||||
| — | 50,000 | (4) | $ | 2.73 | 10/11/2016 | |||||||||
| Carelyn P. Fylling | 250,000 | — | $ | 1.50 | 8/7/2012 | |||||||||
| 19,077 | — | $ | 1.25 | 10/21/2013 | ||||||||||
| — | 20,000 | (5) | $ | 2.40 | 1/11/2016 | |||||||||
| (1) | All options are fully vested. |
| (2) | Options vest as follows: 15,000 on 7/1/2007, 15,000 on 7/1/2008 |
| (3) | Options vest as follows: 13,333 on 3/17/2007, 13,333 on 3/17/2008 |
| (4) | Options vest as follows: 16,667 on 10/11/2007, 16,667 on 10/11/2008, 16,666 on 10/11/2009 |
| (5) | Options vest as follows: 6,667 on 1/12/2007, 6,667 on 1/12/2008, 6,666 on 1/12/2009 |
Potential Payments Upon Termination or Change-in-Control
The following table presents potential payments to executive officers upon termination or a change-in-control event as defined by their respective employment agreements, based on assumptions as if the event took place on December 31, 2006.
Name / Reason for Termination | Base Salary | Discretionary Bonus | ||||||
| Kshitij Mohan | ||||||||
| Disability | (1) | 305,021 | 170,536 | |||||
| Change of Control | (2) | 665,500 | 488,200 | |||||
| Not for Cause | (3) | 665,500 | 658,736 | |||||
| Death | (4) | — | — | |||||
| Voluntary by Dr. Mohan | (5) | — | — | |||||
| Andrew S. Maslan | ||||||||
| Not for Cause | (6) | 77,500 | — | |||||
| Carelyn P. Fylling | ||||||||
| Disability | (7) | 125,125 | — | |||||
| Death | (8) | — | — | |||||
| Not for Cause | (9) | 68,250 | — | |||||
| Involuntary Termination by Ms. Fylling for Good Reason | (10) | 125,125 | — | |||||
| Change of Control | (11) | 68,250 | — | |||||
| Voluntary by Ms. Fylling | (12) | 37,538 | — | |||||
60
| (1) | Base salary will be paid over a period of 11 months, less net amounts received under Company sponsored long-term disability insurance. Discretionary bonus was estimated based on pro-rata portion (based on current employment year) and targeted $150,000 annual cash bonus and 100,000 annual option grant. All unexercised options granted to Dr. Mohan will remain exercisable through their original expiration date. |
| (2) | Base salary will be paid over a period of 24 months. Discretionary bonus is due within 30 days of change-in-control and is equal to two years bonus estimated based on targeted $150,000 annual cash bonus and 100,000 annual option grant. All unexercised options granted to Dr. Mohan will remain exercisable through their original expiration date. |
| (3) | Base salary will be paid over a period of 24 months. Discretionary bonus is due within 30 days of change-in-control and is equal to pro-rata portion (based on current employment year) plus two years bonus estimated based on targeted $150,000 annual cash bonus and 100,000 annual option grant. All unexercised options granted to Dr. Mohan will remain exercisable through their original expiration date. |
| (4) | All unexercised options granted to Dr. Mohan will remain exercisable through their original expiration date. |
| (5) | If Dr. Mohan provides 30 days prior written notice to facilitate transition, then all unexercised options granted to Dr. Mohan will remain exercisable through their original expiration date. Otherwise, unexercised options granted to Dr. Mohan will expire three months after termination. |
| (6) | Base salary will be paid over a period of 6 months. Unvested options will continue to vest for a period of six months from termination. |
| (7) | Base salary will be paid over a period of 11 months, less net amounts received under Company sponsored long-term disability insurance. Prorated bonus and incentive compensation based on the then-applicable bonus plan/long-term incentive compensation program (based on current employment year). |
| (8) | Base salary through end of month in which death occurs, plus prorated bonus and incentive compensation based on the then-applicable bonus plan/long-term incentive compensation program (based on current employment year). All vested stock options become property of the executive's estate. |
| (9) | Lump sum severance payment equal to six months of base salary. Prorated bonus and incentive compensation based on the then-applicable bonus plan/long-term incentive compensation program (based on current employment year). The exercise date of all stock options shall be extended for twelve months following the date of termination. |
| (10) | Lump sum severance payment equal to eleven months of base salary. Prorated bonus and incentive compensation based on the then-applicable bonus plan/long-term incentive compensation program (based on current employment year). The exercise date of all stock options shall be extended for twelve months following the date of termination. |
| (11) | All issued and unvested stock options become immediately fully vested and exercisable. Lump sum severance payment equal to six months of base salary. Prorated bonus and incentive compensation based on the then-applicable bonus plan/long-term incentive compensation program (based on current employment year). The exercise date of all stock options shall be extended for twelve months following the date of termination. |
| (12) | Ms. Fylling's employment may be terminated voluntarily (i) upon written consent of Ms. Fylling and the Company, or (ii) upon sixty days' written notice by Ms. Fylling. If voluntarily terminated pursuant to (i), Ms. Fylling agrees to stay in the employ of the Company for three months, in which she will receive 110% of her base salary. If voluntarily terminated pursuant to (ii), the Company may accelerate the termination date by paying the base salary for such sixty day period in a lump sum. |
Compensation of Directors
For service during 2006, each non-employee director was entitled to and received options to purchase 30,000 shares of the Company’s Common stock; each committee chair was entitled to and received options to purchase 10,000 shares of the Company’s Common stock; each non-employee director was entitled to and received $500 for his participation in each telephonic meeting of the Board or a Committee and $1,000 for his participation in each in-person meeting of the Board or a Committee.
Director Compensation in 2006
Name | Fees Earned or Paid in Cash | Option Awards (1) | Total | |||||||
| James S. Benson | $ | 7,000 | $ | 84,640 | $ | 91,640 | ||||
| David P. Crews | $ | 8,000 | $ | 63,480 | $ | 71,480 | ||||
| Arun K. Deva | $ | 9,000 | $ | 84,640 | $ | 93,640 | ||||
| David F. Drohan | $ | 7,000 | $ | 63,480 | $ | 70,480 | ||||
| Mark T. McLoughlin | $ | 6,500 | $ | 84,640 | $ | 91,140 | ||||
| (1) | At December 31, 2006, the following number of stock options remained unexercised by non-employee directors as follows: Benson - 110,000, Crews - 290,000, Deva - 110,000, Drohan - 90,000, McLoughlin - 110,000 |
61
Compensation Committee Report
The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis section of this item 11 with management, and, based on that review and discussion, recommended to the Board of Directors that said Compensation Discussion and Analysis be included in this annual report on form 10-K.
Submitted by Compensation Committee
Mark T. McLoughlin (Chairman)
David P. Crews
David F. Drohan
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The Company maintains a Long-Term Incentive Plan approved by a majority of shareholders that authorizes awards representing up to 5,000,000 shares of Common stock.
EQUITY COMPENSATION PLAN INFORMATION AS OF DECEMBER 31, 2006
Plan category | Number of securities to be issued upon exercise of outstanding options, warrants, and rights | Weighted average exercise price of outstanding options, warrants, and rights | Number of securities remaining available for future issuance | |||||||
| (a) | (b) | (c) | ||||||||
| Equity compensation plans approved by security holders | 3,202,377 | $ | 1.82 | 1,411,423 | ||||||
| Equity compensation plans not approved by security holders | 0 | -- | 0 | |||||||
| Total | 3,202,377 | $ | 1.82 | 1,411,423 | ||||||
As of December 31, 2006, 386,200 shares of common stock have been issued upon exercise of options granted pursuant to the Long Term Incentive Plan.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS
In reliance upon statements filed with the SEC under Section 13(d) or 13(g) of the Secutities Exchange Act of 1934 (unless the Company knew or had reason to know such statements are not accurate or complete), the following persons are known to Cytomedix to be the beneficial owner of more than five percent of Cytomedix’s voting securities as of February 15, 2007, as indicated below:
Title of Class | Name and Address of Beneficial Owner | Amount and Nature of Beneficial Ownership | Percent of Class | ||||||||
| Common Stock | David E. Jorden | 2,487,800 | (1) | 8.5 | % | ||||||
| 600 Travis, Suite 3700 | |||||||||||
| Houston, Texas 77002 | |||||||||||
(1) Includes 167,000 shares issuable upon exercise of warrants. Pursuant to the terms of the warrants, the reporting person cannot exercise such warrants if the exercise would result in the reporting person being the “beneficial owner” of more than 9.999% of the outstanding stock within the meaning of Rule 13d-1 under the Securities Exchange Act of 1934.
62
SECURITY OWNERSHIP OF MANAGEMENT
The following table sets forth the number and percentage of shares of all classes of stock that as of February 15, 2007 are deemed to be beneficially owned by each director and executive officer of the Company and by all directors and executive officers as a group:
Title of Class | Name of Beneficial Owner | Amount and Nature of Beneficial Ownership (1) | Percent of Class (1) | ||||||||
| Common Stock | James S. Benson | 150,000 | (2) | * | |||||||
| Common Stock | David P. Crews | 1,021,623 | (3) | 3.4 | % | ||||||
| Common Stock | Arun K. Deva | 150,000 | (4) | * | |||||||
| Common Stock | David F. Drohan | 122,000 | (5) | * | |||||||
| Common Stock | Carelyn P. Fylling | 293,375 | (6) | * | |||||||
| Common Stock | Andrew S. Maslan | 154,000 | (7) | * | |||||||
| Common Stock | Mark T. McLoughlin | 150,000 | (8) | * | |||||||
| Common Stock | Kshitij Mohan | 1,200,000 | (9) | 4.0 | % | ||||||
| Common Stock | Group consisting of Benson, Crews, Deva, Drohan, Fylling, Maslan, McLoughlin, and Mohan | 3,240,998 | 9.6 | % | |||||||
* Less than 1%.
(1) For purposes of determining the amount of securities beneficially owned, share amounts include all Common stock owned outright plus all convertible shares, warrants, and options exercisable for Common stock. The Percent of Class for Common stock is based on the number of shares of the Company’s Common stock outstanding as of February 15, 2007. Shares of Common stock issuable upon conversion of convertible notes, or the exercise of options or warrants currently exercisable, or exercisable within 60 days after the preparation of this table, are deemed outstanding for the purpose of computing the percentage ownership of the person holding such options or warrants, but are not deemed outstanding for computing the percentage ownership of any other persons.
(2) Consists of 150,000 shares Mr. Benson may acquire upon the exercise of stock options.
(3) Consists of 684,871 shares owned as trustee for David Paul Crews Revocable Trust, 16,752 shares owned by children, and 320,000 shares Mr. Crews may acquire upon the exercise of stock options.
(4) Consists of 150,000 shares Mr. Deva may acquire upon the exercise of stock options.
(5) Consists of 2,000 shares directly owned by Mr. Drohan and 120,000 shares Mr. Drohan may acquire upon the exercise of stock options.
(6) Consists of 4,298 shares directly owned by Ms. Fylling and 289,077 shares Ms. Fylling may acquire upon the exercise of stock options.
(7) Consists of 4,000 shares directly owned by Mr. Maslan and 150,000 shares Mr. Maslan may acquire upon the exercise of stock options.
(8) Consists of 150,000 shares Mr. McLoughlin may acquire upon the exercise of stock options.
(9) Consists of 10,000 shares directly owned by Dr. Mohan and 1,190,000 shares Dr. Mohan may acquire upon the exercise of stock options.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The Company was not involved in any transactions with related persons, as defined in Item 404 of Regulation S-K, since January 1, 2006, nor are there any such proposed transactions, that exceed $120,000.
The Company has the following directors: James S. Benson, David P. Crews, Arun K. Deva, David F. Drohan, Mark T. McLoughlin, and Kshitij Mohan. Each of these directors is independent as defined by Section 121(A) of the listing standards of the American Stock Exchange, with the exception of Dr. Mohan, who, in addition to serving as the Chairman of the Board, is also the Company’s Chief Executive Officer. Dr. Mohan does not serve on the Audit, Nominating and Governance, or Compensation Committees.
63
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
The following table presents fees for professional services rendered by L J Soldinger Associates LLC for the fiscal years 2006 and 2005:
Services Performed | 2006 | 2005 | |||||
| Audit Fees (1) | $ | 340,000 | $ | 200,000 | |||
| Audit-Related Fees (2) | 2,000 | 30,000 | |||||
| Tax Fees (3) | 19,000 | 15,000 | |||||
| All Other Fees (4) | — | — | |||||
| Total Fees | $ | 361,000 | $ | 245,000 | |||
(1) Audit fees represent fees billed for professional services provided in connection with the audit of the Company’s annual financial statements, reviews of its quarterly financial statements, audit services provided in connection with statutory and regulatory filings for those years and audit services provided in connection with securities registration and/or other issues resulting from that process. In 2006, audit fees also includes services rendered for audits of management’s assessment of the effectiveness of internal controls over financial reporting and the effectiveness of internal control over financial reporting.
(2) Audit-related fees represent fees billed primarily for assurance and related services reasonably related to securities registration and/or other issues resulting from that process.
(3) Tax fees principally represent fees billed for tax preparation, tax advice and tax planning services.
(4) All other fees principally would include fees billed for products and services provided by the accountant, other than the services reported under the three captions above.
Pursuant to its charter, the Audit Committee must pre-approve audit services and permitted non-audit services (including the fees and terms thereof) to be performed for the Company by its independent auditor. In 2005 and 2006, all such services were pre-approved by the Audit Committee.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
| (a) | 1. | Financial Statements |
The following financial statements of Cytomedix, Inc. are included in Item 8:
| Report of Independent Registered Public Accounting Firm | 24 | |
| Balance Sheets | 25 | |
| Statements of Operations | 26 | |
| Statements of Stockholders’ Equity | 27 | |
| Statements of Cash Flows | 30 | |
| Notes to Financial Statements | 31 |
| 2. | Schedule II—Valuation and Qualifying Accounts |
See Footnotes to Financial Statements in Item 8 of this report.
| (b) | Exhibits |
For a list of exhibits filed with this Form 10-K, refer to the Exhibit Index beginning on page 66.
64
SIGNATURES
In accordance with Section 13 or 15(d) of the Exchange Act, the registrant caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
CYTOMEDIX, INC.
By: /s/Kshitij Mohan Kshitij Mohan, CEO and Chairman of the Board of Directors Date: February 26, 2007 |
In accordance with the Exchange Act, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
By: /s/Kshitij Mohan Kshitij Mohan, CEO and Chairman of the Board of Directors Date: February 26, 2007 /s/Andrew S. Maslan Andrew S. Maslan, Chief Financial Officer and Chief Accounting Officer Date: February 26, 2007 /s/David P. Crews David P. Crews, Director Date: February 26, 2007 /s/Arun K. Deva Arun K. Deva, Director Date: February 26, 2007 /s/David F. Drohan David F. Drohan, Director Date: February 26, 2007 |
Signed originals of this written statement have been provided to Cytomedix, Inc. and will be retained by Cytomedix, Inc. and furnished to the Securities and Exchange Commission or its staff upon request.
65
EXHIBIT INDEX
| 2.1 | First Amended Plan of Reorganization with All Technical Amendments (Previously filed on June 28, 2002, on Form 8-K, File No. 000-28443). | |
| 2.2 | Amended and Restated Official Exhibits to the First Amended Plan of Reorganization of Cytomedix, Inc. with All Technical Amendments (Previously filed on May 10, 2004, on Form 10-QSB for the quarter ended March 31, 2004, File No. 000-28443). | |
| 3.1 | Restated Certificate of Incorporation of Cytomedix, Inc. (Previously filed on November 7, 2002, on Form 10-QSB for quarter ended June 30, 2001, File No. 000-28443). | |
| 3.2 | Amendment to Restated Certificate of Incorporation of Cytomedix, Inc. (Previously filed on November 15, 2004, on Form 10-QSB for quarter ended September 30, 2004, File No. 000-28443). | |
| 3.3 | Restated Bylaws of Cytomedix, Inc. (Previously filed on November 7, 2002, on Form 10-QSB for quarter ended June 30, 2001, File No. 000-28443). | |
| 4.1 | Amended and Restated Certificate of Designation of the Relative Rights and Preferences of Series A Preferred, Series B Preferred and common stock of Cytomedix, Inc. (Previously filed on March 31, 2004, on Form 10-KSB for year ended December 31, 2003, File No. 000-28443). | |
| 4.2 | Form of Class A Warrant issued to New Investors and DIP Lenders (Previously filed on December 5, 2002, on Form 10-QSB for quarter ended September 30, 2001, File No. 000-28443). | |
| 4.3 | Form of Class B Warrant issued to New Investors and DIP Lenders (Previously filed on December 5, 2002, on Form 10-QSB for quarter ended September 30, 2001, File No. 000-28443). | |
| 4.4 | Form of Series C-1 Warrant to Purchase Shares of common stock of Cytomedix, Inc. (Previously filed on March 29, 2004 on Form 8-K, File No. 000-28443.) | |
| 4.5 | Form of Series C-2 Warrant to Purchase Shares of common stock of Cytomedix, Inc. (Previously filed on March 29, 2004 on Form 8-K, File No. 000-28443). | |
| 4.6 | Certificate of Designation of the Relative Rights and Preferences of the Series C Convertible Stock of Cytomedix, Inc. as filed with the Delaware Secretary of State on March 25, 2004 (Previously filed on March 29, 2004 on Form 8-K, File No. 000-28443). | |
| 4.7 | Form of warrant issued to investors in the 2004 Unit Offering (Previously filed on May 11, 2004, on Form SB-2, File No. 333-115364). | |
| 4.8 | Form of Class D Warrant to Purchase Shares of Common Stock of Cytomedix, Inc. (Previously filed on May 2, 2005, on Form 8-K, File No. 001-32518). | |
| 4.9 | Form of Registration Rights Agreement between Cytomedix, Inc., and Class D Warrantholders (Previously filed on May 2, 2005, on Form 8-K, File No. 001-32518). | |
| 10.1 | Royalty Agreement, dated as of December 26, 2000, by and between Cytomedix, Inc. and Curative Health Services, Inc. (Previously filed on January 17, 2001, on Form 8-K, File No. 000-28443). | |
| 10.2 | First Amendment to Royalty Agreement, dated as of April 20, 2001, by and between Cytomedix, Inc. and Curative Health Services, Inc. (Previously filed on May 25, 2001, on SB-2/A, File No. 333-55818). | |
| 10.3 | Second Amendment to Royalty Agreement, dated as of December 5, 2002, by and between Cytomedix, Inc. and Curative Health Services, Inc. (Previously filed on March 31, 2003, on Form 10-KSB for year ended December 31, 2002, File No. 000-28443). | |
| 10.4 | Cytomedix, Inc. Long-Term Incentive Plan. | |
| 10.5 | License Agreement dated March 21, 2001, by and between Cytomedix, Inc. and DePuy AcroMed, Inc. (Previously filed on April 16, 2001, on Form 10-KSB for year ended December 31, 2000, File No. 000-28443). |
66
| 10.6 | Amendment dated March 3, 2005, to the License Agreement by and between Cytomedix, Inc. and DePuy Spine, Inc. (f/k/a DePuy Acromed, Inc.) (Previously filed on March 31, 2005, on Form 10-KSB for year ended December 31, 2004, File No. 000-28443). | |
| 10.7 | Second License Agreement dated March 3, 2005, to the License Agreement by and between Cytomedix, Inc. and DePuy Spine, Inc. (f/k/a DePuy Acromed, Inc.) (Previously filed on March 31, 2005, on Form 10-KSB for year ended December 31, 2004, File No. 000-28443). | |
| 10.8 | Settlement and License Agreement dated May 1, 2005 by and between Cytomedix, Inc. and Medtronic, Inc. (Previously filed on May 10, 2005, on Form 8-K, File No. 000-28443). | |
| 10.9 | Settlement Agreement and License Agreement dated May 23, 2005, by and between Cytomedix, Inc., and Harvest Technologies Corporation (Previously filed on May 27, 2005, on Form 8-K, File No. 000-28443). | |
| 10.10 | Settlement and License Agreement dated June 26, 2005, by and between Cytomedix, Inc., and Perfusion Partners and Associates Inc. (Previously filed on August 15, 2005, on Form 10-QSB for the quarter ended June 20, 2005, File No. 000-28443). | |
| 10.11 | License Agreement dated October 7, 2005, by and between Cytomedix, Inc., and COBE Cardiovascular, Inc. (Previously filed on October 11, 2005, on Form 8-K, File No. 000-28443). | |
| 10.12 | Settlement and License Agreement dated October 12, 2005, by and between Cytomedix, Inc., and SafeBlood Technologies, Inc. (Previously filed on November 9, 2005, on Form 10-QSB, File No. 000-28443). | |
| 10.13 | Employment Agreement with Ms. Carelyn P. Fylling (Previously filed on December 5, 2002, on Form 10-QSB for quarter ended September 30, 2001, File No. 000-28443). | |
| 10.14 | Employment Agreement with Mr. William L. Allender (Previously filed on March 31, 2004, on Form 10-KSB for year ended December 31, 2003, File No. 000-28443). | |
| 10.15 | Addendum to Employment Agreement with Mr. William L. Allender (Previously filed on November 15, 2004, on Form 10-QSB for quarter ended September 30, 2004, File No. 000-28443). | |
| 10.16 | Separation Agreement and Release dated July 15, 2005, by and between Cytomedix, Inc., and William L. Allender (Previously filed on August 15, 2005, on Form 10-QSB for the quarter ended June 30, 2005, File No. 000-28443). | |
| 10.17 | Employment Agreement with Kshitij Mohan, Ph.D., dated April 20, 2004 (Previously filed on May 7, 2004, on Form 8-K, File No. 00028443). | |
| 10.18 | Termination Agreement between Cytomedix, Inc., and Kshitij Mohan, dated April 20, 2004 (Previously filed on May 7, 2004, on Form 8-K, File No. 000-28443). | |
| 10.19 | Employment Agreement dated June 3, 2005, by and between Cytomedix, Inc., and Andrew Maslan (Previously filed on June 20, 2005, on Form 8-K, File No. 000-28443). | |
| 10.20 | Distributor Agreement dated October 31, 2005 by and between Cytomedix, Inc. and National Wound Therapies, LLC. (Previously filed on March 23, 2006, on Form 10-KSB, File No. 001-32518). | |
| 10.21 | Settlement and License Agreement dated May 19, 2006, between Cytomedix, Inc., and Biomet Biologics, Inc. (Previously filed on August 9, 2006, on Form 10-Q, File No. 001-32518). | |
| 10.22 | First Addendum to Letter Agreement dated October 4, 2006, between Cytomedix, Inc., and Andrew Maslan (Previously filed on November 1, 2006 on Form 10-Q, File No. 001-32518). | |
| 20.1 | Definitive Proxy Statement (Previously filed on September 22, 2006, File No. 001-32518). | |
| 23.1 | Consent of LJ Soldinger Associates, LLC. | |
| 31.1 | Certification of Chief Executive Officer of Cytomedix, Inc., pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 31.2 | Certification of Chief Financial Officer of Cytomedix, Inc., pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 32.1 | Certificate of Chief Executive Officer of Cytomedix, Inc., pursuant to 18 U.S.C.ss.1350. | |
| 32.2 | Certificate of Chief Financial Officer of Cytomedix, Inc., pursuant to 18 U.S.C.ss.1350. |
67