Exhibit 99.1

January, 2015

Protalex, Inc. Forward - Looking Statement Statements made in this presentation stating the Company ’ s beliefs, intentions, and expectations are forward - looking statements. The Company ’ s actual results could differ materially from those projected herein. Additional information is contained in the Company ’ s SEC filings such as our Form 10 - K and Form 10 - Qs filed at www.sec.gov. 2 Protalex, Inc .
Protalex, Inc. Protalex, Inc. (Ticker: PRTX) ▪ Rheumatoid Arthritis (>$18B) ▪ ITP (~$1B) ▪ Other Autoimmune Diseases ($Bs) 3 ▪ Experienced Management ▪ Strong Scientific Advisors ▪ Low - burn Model ▪ Funded - to - date by majority shareholder ▪ Highly - purified b acterial protein ▪ W ell - studied in five human studies ▪ Demonstrated s trong safety p rofile ▪ P otential for multiple clinical uses Lead Product PRTX - 100 Market Opportunity Company Structure

Protalex, Inc. The BIG PICTURE A Huge Market with unmet needs o Today in the US, millions of individuals suffer from autoimmune diseases, like rheumatoid arthritis (RA) — the largest autoimmune market, and Immune Thrombocytopenia (ITP) — bruising and bleeding that does not easily heal. ITP is an orphan disease. o Collectively these individuals spend $23 billion on drugs o Three drugs – Humira, Enbrel and Remicade account for $18 billion in sales. o The cost to treat an RA patient is about $25,000 per year. 4

The M ost I mportant Unmet Need is SAFETY o Humira, Enbrel and Remicade are TNF - inhibitors o All three drugs suppress the immune system, and that has a major impact on those who take them. o The FDA requires them to carry a Black Box Warning for such problems as: o Congestive Heart Failure, Blood Poisoning(Sepsis), Brain Disease(PML), Liver Damage, Leukemia, Lymphoma, Malignances, Serious Infections, reactivation of Hepatitis B and Death. 5 Protalex, Inc .
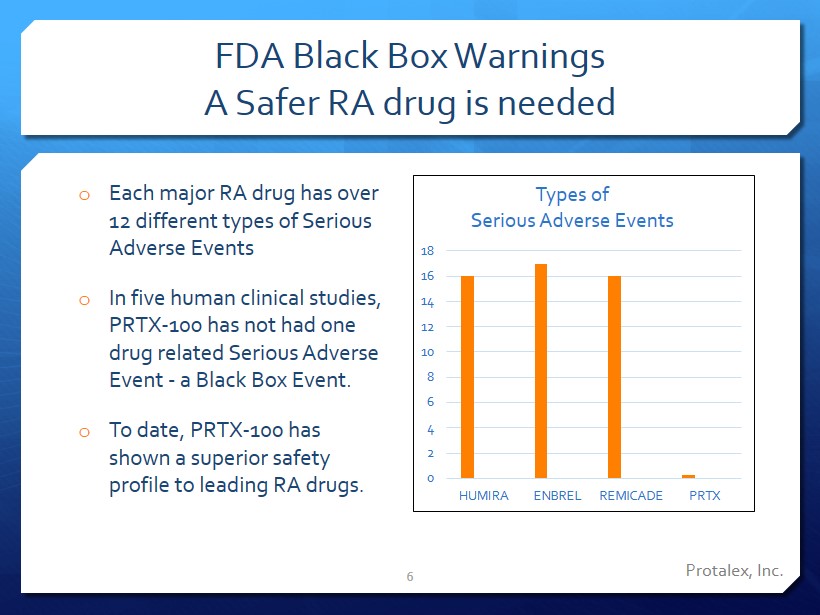
Protalex, Inc. FDA Black Box Warnings A Safer RA drug is needed o Each major RA drug has over 12 different types of Serious Adverse Events o In five human clinical studies, PRTX - 100 has not had one drug related Serious Adverse Event - a Black Box Event. o To date, PRTX - 100 has shown a superior safety profile to leading RA drugs . 6 0 2 4 6 8 10 12 14 16 18 HUMIRA ENBREL REMICADE PRTX Types of Serious Adverse Events

Efficacy of RA Drugs 50% 50% 39% 0% 10% 20% 30% 40% 50% 60% Day 85 Day 113 Day 102 PRTX Enbrel MTX ACR50 Scores 0% 10% 20% 30% 40% 50% 60% 7 DAS28 < 3.2 Protalex, Inc .

Protalex, Inc. Durability of Treatment Number of Treatments per Year 0 20 40 60 80 100 120 8 Number of Treatments per Year
What Drug Should be Used ? The Answer is not Easy To the left is EULAR’s summary of a 20 page recommendation for the treatment path of RA An RA drug would become the first choice to use - - If it was a safer treatment than the current drugs or If a Responder Screening Test for the drug existed. 9 It is not important to be able to read the chart only to note its complexity Protalex, Inc .

Competitive Advantages of PRTX - 100 to Current Biologicals No FDA Black Box - Does not suppress the immune system Attractive safety profile allows for potential use in combination with other therapies Potentially applicable across a broad range of autoimmune diseases Considerably lower cost production Strong Intellectual Property Rights 10 Protalex, Inc.

Protalex, Inc. Immune Thrombocytopenia (ITP) A Rare and Serious Disorder An Orphan Disease in the US and Europe Abnormally low platelet count in the blood Daily or Weekly treatment for life Annual cost of treatment is about $60,000 Promacta® & Nplate® only globally approved treatments ($1B) Preclinical data indicates that PRTX - 100 may be an effective treatment Planning a Phase 1/2 trial in early 2015 11

Protalex, Inc. Protalex Milestones x 2Q13 Safety data from first three cohorts of PRTX - 100 - 104 x 3Q13 Initiation of Cohort 5 extension study, to investigate monthly maintenance doses x 2Q14 Top - line results of PRTX - 100 - 104 trial x 4Q14 Filing IND for PRTX - 100 in ITP □ 1Q15 Top - line results from Cohort 5 extension study □ 1Q15 Submit end of study report from PRTX - 100 - 104 trial □ 1Q15 Initiation PRTX - 100 - 105 continuation study □ 1H15 First dose in Phase 1/2 study of PRTX - 100 in ITP 12
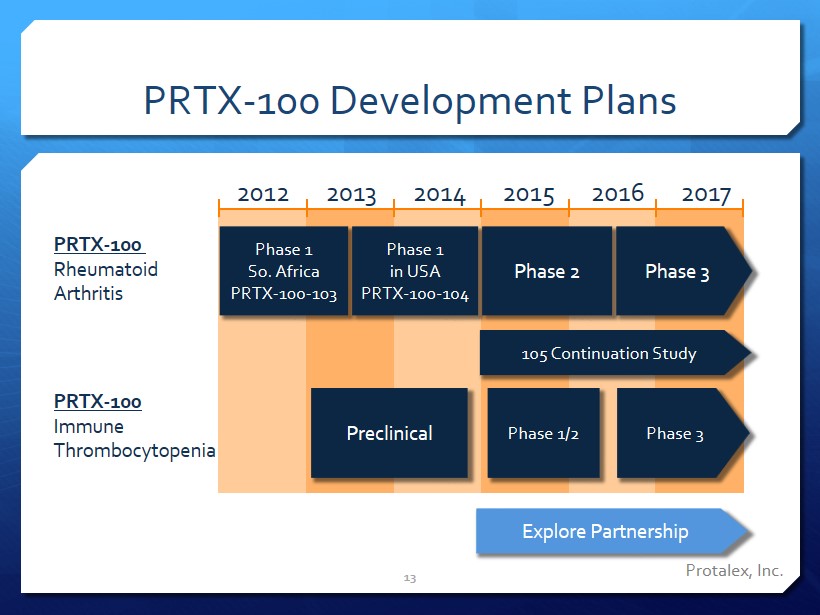
PRTX - 100 Development Plans 13 2012 2013 2014 2015 2016 2017 PRTX - 100 Rheumatoid Arthritis PRTX - 100 Immune Thrombocytopenia Phase 1 So. Africa PRTX - 100 - 103 Phase 1 in USA PRTX - 100 - 104 Phase 2 Phase 3 Explore Partnership Preclinical Phase 1/2 Phase 3 Protalex, Inc. 105 Continuation Study

Protalex, Inc. Protalex Key Team Members Arnold P Kling – President, Director; Principal of Niobe Ventures, LLC, experienced investor in and manager of early stage technology companies James W Dowe III – Vice - Chair of SAB; active investor in biotechnology, computer software and investment management companies William E. Gannon, MD – Chief Medical Officer; more than 20 years experience in clinical development and regulatory affairs at Quintiles, PPD, and other companies Bruce McClain, MD – Medical Director; more than 20 years experience in clinical development and product safety; senior roles at Aeras Global and MedImmune Richard Francovitch, Ph.D . -- VP of ITP Programs; 27 years pharma experience, former Head of Hematology Franchise and Global Commercial Leader for Promacta at GSK Benjamin R Bowen, Ph.D . – Senior Advisor; background in pharma and biotech R&D at Genentech, Ciba - Geigy, and Novartis; ten years in investment banking Michelle Catalina, Ph.D . – Director of Preclinical Studies; academic research background in immunology, former instructor at U Mass Medical Center 14

Protalex, Inc. Patents and Intellectual Property Patents (five issued in US and two in Japan) ▪ Initial US patent 7,211,258, “Protein A compositions and methods of use” filed 2002 and issued 2007 for RA, juvenile RA, and systemic lupus erythematosus ▪ Continuation patents expanding use were issued for: o ITP or autoimmune TP in 2008 o Acute inflammatory response or inflammation in 2012 o Psoriasis and scleroderma in 2012 o Multiple Sclerosis in 2013 ▪ October 2010 Japanese patent issued with 2023 expiration date for RA, SLE and ITP o April 2014 2 nd Japanese patent issued for psoriasis, scleroderma, Crohn’s Disease ▪ Additional patent applications pending in Europe, Canada, Japan, and US Other Intellectual Property ▪ Considerable know - how in the manufacture is a trade secret 15 Protalex, Inc.

Protalex, Inc. Protalex Investment Thesis Summary PRTX - 100 – potentially a blockbuster drug ▪ Potential treatment of diseases in both large and small markets (RA, ITP) ▪ Potentially efficacy in a number of orphan diseases ▪ Considerable cost of goods advantage over competitive drugs ▪ To date, a superior safety profile in five human clinical studies Market Opportunity ▪ RA = $18 Billion annual market size ▪ ITP = $1 B market ▪ Future expansion into other disease areas Company Structure ▪ Experienced Management and Advisory Teams with “skin in the game” ▪ Committed support from expert Scientific Advisory Board ▪ Established and growing IP position and trade secrets 16

January, 2015