FORM 6-K
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Report of Foreign Issuer
Pursuant to Rule 13a-16 or 15d-16 of
the Securities Exchange Act of 1934
Pursuant to Rule 13a-16 or 15d-16 of
the Securities Exchange Act of 1934
For the month of March, 2006
Commission File Number: 0-30600
The Westaim Corporation
(Translation of registrant’s name into English)
144-4th Avenue, S.W., Suite 1010
Calgary, Alberta T2P 3N4
Canada
Calgary, Alberta T2P 3N4
Canada
(Address of principal executive officers)
(780) 992-5231
(Telephone number, including area code)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F o Form 40-F þ
Indicate by check mark whether the registrant by furnishing the information contained in this Form is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934.
Yes o No þ
If “Yes, is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b): 82-___
The following documents are included in this Form 6-K and are hereby filed with the Securities and Exchange Commission for the purpose of being (and hereby are) incorporated by reference in the Registration Statement on Form S-8 under the Securities Act of 1933 (Reg. no. 333-12532):
| 1. | Code of Conduct and Ethics; | ||
| 2. | The Registrant’s Annual Report to shareholders for the financial year ended December 31, 2005; | ||
| 3. | Notice of Meeting; | ||
| 4. | Information Circular; | ||
| 5. | Proxy form; and | ||
| 6. | Mail Card. |
7. SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant, The Westaim Corporation, has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| THE WESTAIM CORPORATION | ||||
| Date: March 27, 2006 | By: | /s/Brian D. Heck | ||
| Brian D. Heck | ||||
| Vice President, General Counsel and Corporate Secretary | ||||
The Westaim Corporation
Code of Conduct and Ethics for Directors, Officers and Employees
Code of Conduct and Ethics for Directors, Officers and Employees
The Board of Directors (the “Board”) of The Westaim Corporation (the “Corporation”) has developed and adopted this Code of Conduct and Ethics (the “Code”) applicable to its Directors, Officers and Employees.
All Directors, Officers and Employees of the Corporation are accountable for adherence to this Code and will at all times in relation to the Corporation:
| • | Act honestly and ethically. | |
| • | Comply with all applicable laws, rules and regulations of federal, provincial, state and local governments, and other appropriate private and public regulatory agencies including, without limitation, all “insider trading” prohibitions. | |
| • | Not use confidential information acquired as a result of their role with the Corporation for personal advantage. | |
| • | Avoid all actual or apparent conflicts of interest between their personal and professional relationships, ethically handling all such actual or apparent conflicts of interest. | |
| • | Not (a) take for themselves any opportunity that properly belongs to the Corporation or is discovered through the use of corporate property, information or position; or (b) use corporate property, information or position for personal gain; nor (c) compete with any business activity of the Corporation. | |
| • | Promptly and accurately provide all necessary information to assure that the Corporation’s public reports, documents and filings (including but not limited to those filed with any stock exchange or securities commission or in any other public communication) are full, fair, accurate, timely and understandable and that the Corporation’s public disclosure requirements are fully met. | |
| • | Promptly report any known violations of this Code to the Chief Compliance Officer of the Corporation. | |
| • | Not permit retaliation of any kind by or on behalf of the Corporation and its Directors, Officers and Employees against good faith reports or complaints of violations of this Code or other illegal or unethical conduct. |
This Code may be amended, modified or waived from time to time by the Board in its sole discretion, and waivers may also be granted by the Corporate Governance Committee or the Audit Committee of the Board, subject to the disclosure and other provisions of the applicable Canadian and U.S. securities legislation and the applicable rules of the stock exchanges upon which the Corporation’s shares trade from time to time.
This Code does not summarize all laws, rules and regulations applicable to the Corporation and its Directors, Officers and Employees. Whenever feasible, Directors, Officers and Employees should consult the Legal Services Department of the Corporation, and the various guidelines that the Corporation has prepared on specific laws, rules and regulations, in relation to any questions regarding the applicability of this Code or any of its provisions.

| looking in westaim looking out |
| 2005 ANNUAL REPORT |
| iFire Technology Corp. |
| pixcel define entertainment and knowledge |

| iFire’s TDEL flat panel TV technology combines high |
| performance with low manufacturing cost potential |
| NUCRYST Pharmaceuticals Corp. |
| nanocrystal open large market |
| opportunities for new pharmaceuticals |

| Revenue from the SILCRYST™ wound care coatings |
| business was $22.7 million, up from $18.9 million in 2004 |
The Westaim Corporation Annual Report 2005
ANNUAL REPORT CONTENTS
Message to Shareholders | 1 |

| looking in iFire TECHNOLOGY CORP. 05 |
| looking in NUCRYST |
| PHARMACEUTIC ALS CORP. |
| 11 |
WESTAIM FINANCIAL REVIEW | ||||
| Management’s Discussion & Analysis | 17 | |||
| Management’s Responsibility for Financial Information | 32 | |||
| Auditors’ Report | 33 | |||
| Consolidated Financial Statements | 34 | |||
| Notes to Consolidated Financial Statements | 37 | |||
| Shareholder Information | 61 | |||
| Board of Directors | 61 | |||
| Corporate Information | 62 |
All figures are in Canadian dollars, unless otherwise stated.

| WESTAIM |
| looking out Westaim’s mission is to |
| deliver to its shareholders by investing |
| in high potential technology companies |
| Message to Shareholders |
| Barry M. Heck G.A. (Drew) fitch Anthony B.Johnston President & CEO Senior vice President & CEO Senior vice President |

| WESTAIM |
| looking out Westaim’s mission is to |
| deliver to its shareholders by investing |
| in high potential technology companies |
| Message to Shareholders |
| Barry M. Heck G.A. (Drew) fitch Anthony B.Johnston President & CEO Senior vice President & CEO Senior vice President |
The Westaim Corporation Annual Report 2005
2005 was a year of significant achievement for Westaim, marked by the realization of two important business objectives that will help deliver value for shareholders. First, we successfully completed an initial public offering (IPO) of common shares of our NUCRYST Pharmaceuticals subsidiary. Second, our iFire Technology business began pilot production of flat panel display modules. Both of these events are key milestones in Westaim’s mission to bring value to shareholders by investing in, and providing leadership to, our high-potential technology companies.
At December 31, 2005, we continued to maintain a healthy balance sheet with $119.6 million in consolidated cash and short-term investments. The IPO of NUCRYST Pharmaceuticals completed in December 2005 has positioned NUCRYST with a strong balance sheet with $42 million in cash and short-term investments. NUCRYST shares now trade on NASDAQ under the symbol NCST and on the Toronto Stock Exchange under the symbol NCS. With the conversion of debt owed to Westaim into common shares completed in January 2006, Westaim continues to own a controlling interest in NUCRYST of approximately 75.3%.
1
The Westaim Corporation Annual Report 2005
NUCRYST Pharmaceuticals
NUCRYST has a solid track record of revenue growth from its SILCRYST™ wound care coatings business as well as a promising pipeline of new medical device and pharmaceutical products based on its proprietary nanocrystalline noble metal technology. Going forward, it is well positioned to fund its R&D and clinical trial programs with a revenue-generating business and a healthy, debt-free balance sheet.
In 2005, NUCRYST posted record revenues, excluding milestone payments, from its SILCRYST™ coatings business of $22.7 million, up from $18.9 million in 2004. All of the revenue was generated through its relationship with Smith & Nephew plc, a global medical device company with a substantial wound care products sales force. Smith & Nephew markets, distributes and sells advanced wound care dressings with NUCRYST’s SILCRYST™ coatings under Smith & Nephew’s Acticoat™ trademark in more than 30 countries around the world. NUCRYST’s agreement with Smith & Nephew includes up to US$56.5 million in milestone payments that are earned over time upon reaching certain predetermined sales and regulatory thresholds. In 2005, NUCRYST earned a US$5-million milestone payment, bringing its total milestone revenue to date to US$19 million.
Last year NUCRYST completed a $7-million expansion of its production facility in Fort Saskatchewan, Alberta to meet anticipated demand for Acticoat™ dressings from Smith & Nephew. In 2005, the company initiated another $7-million expansion of the facility, which is expected to be commissioned in early 2007.
In addition to using its patented nanocrystalline silver as a coating for wound dressings under the trademark SILCRYST™, NUCRYST has developed a patented nanocrystalline powder for use as an active pharmaceutical ingredient, or API, known as NPI 32101. NUCRYST’s proprietary nanocrystalline silver structures have exhibited broad spectrum antimicrobial and potent anti-inflammatory properties in preclinical studies and may have the potential to treat a wide range of infectious and inflammatory diseases. In the fourth quarter of 2005, NUCRYST began enrolling patients in a second Phase 2 clinical trial of NPI 32101 in a cream formulation for the treatment of atopic dermatitis, a form of eczema. Results from this trial are expected by the end of 2006.
NUCRYST is also conducting preclinical research to extend its nanocrystalline silver technology to the treatment of other dermatological and gastrointestinal conditions.
| Acticoat™ is a trademark of Smith & Nephew plc | ||
| SILCRYST™ is a trademark of NUCRYST Pharmaceuticals Corp. |
2
The Westaim Corporation Annual Report 2005
iFire Technology
On time and on budget, iFire completed construction and began operation of a $46-million pilot manufacturing facility in Toronto for its proprietary TDEL (thick dielectric electroluminescent) flat panel display technology. Pilot production is a critical step for any new product or technology. For iFire, the pilot serves three main purposes. First, it helps validate the display’s low-cost potential in a manufacturing environment. Second, it produces the engineering samples that iFire sends to potential customers and partners for their independent evaluation. Third, the pilot will become the home base for iFire’s TDEL technology where iFire will continue its development work to improve the display’s performance and discover greater manufacturing efficiencies.
iFire’s TDEL technology has the potential to be a major player in the consumer television market by offering a flat panel alternative that has advantages over the existing technologies in performance, package and price. TDEL is very different from other technologies. Liquid crystal displays (LCD) and plasma display panels (PDP) are more complex, involve extreme precision in manufacturing and intricate electronics to manipulate video images.
TDEL is much more like the cathode ray tube, or CRT, the dominant technology in consumer television. Both TDEL and CRT are emissive (light from the front) phosphor-based displays, making the viewing experience similar. But unlike the CRT, TDEL panels are flat, lighter and solid state. They are also more rugged, lighter and thinner than LCD and PDP models making them easier to ship and allow for products that are slimmer and truly hang-on-the-wall.
The lower capital cost requirements for manufacturing are key to the iFire business proposition. While a new Generation 6 LCD plant with an annual capacity of three million units can cost up to US$3 billion, we project that a TDEL facility with similar capacity will cost only US$600 million. Adding to the capital cost advantage is our potential manufacturing cost advantage. According to a study iFire conducted with Industrial Design and Construction, a leading industrial firm for the design of electronics and flat panel production facilities, TDEL holds a potential manufacturing cost advantage of more than 30% over LCD in a mature manufacturing environment. This analysis was originally conducted in 2003 and was updated last year to confirm that iFire’s cost advantage should be sustainable going forward, even with the cost of LCD modules continuing to decline.
3
The Westaim Corporation Annual Report 2005
As part of our business strategy, Westaim remains committed to protecting our intellectual property through trade secrets and by expanding the patent portfolios of our two businesses. iFire Technology has strong patent protection on its core technologies and processes in the United States and internationally. NUCRYST Pharmaceuticals has been issued a wide range of patents in the United States and internationally, covering the composition of matter, methods of manufacture as well as methods of use, including medical device coatings and pharmaceuticals.
We look forward to continued progress in 2006. NUCRYST is expecting results from the second Phase 2 human clinical trial of its atopic dermatitis drug. iFire will continue with pilot production and will look to enter into its first commercial partnership for the manufacture of TDEL panels. On behalf of the Board of Directors, I would like to thank our employees and shareholders for their dedication, hard work and commitment in 2005.
| Sincerely, |
 |
| Barry M. Heck |
| President & CEO |
4

| Looking out |
| iFire Technology Corp. |
| TDEL is the |
| next generation |
| flat panel TV technology |

| Looking out |
| iFire Technology Corp. |
| TDEL is the |
| next generation |
| flat panel TV technology |
The Westaim Corporation Annual Report 2005
looking in iFire
TECHNOLOGY CORP.
iFire Technology Corp. has developed a proprietary low-cost, flat panel display technology that has the potential to set the industry benchmarks for performance, package and price. The technology is called TDEL (thick dielectric electroluminescent) and with it, iFire aims to be a significant force in the flat panel television market.
The technology behind TDEL is relatively simple, giving it advantages over liquid crystal displays (LCD) and plasma display panels (PDP) in video image performance and quality, product design and cost of manufacturing. The TDEL viewing experience is much closer to the cathode ray television (CRT) – the standard in television performance – than it is to other flat panel display technologies because like the CRT, TDEL is an emissive, phosphor-based display.
PERFORMANCE
iFire’s TDEL displays feature native high-definition resolution and excellent TV performance characteristics including CRT-like color saturation and image quality, fast video response times and a wide viewing angle.
PRICE
TDEL displays are relatively simple devices compared to other flat panel technologies. Its solid state form has fewer manufacturing steps and utilizes common processes such as screen printing and thin-film sputtering. The result is much lower projected capital costs for manufacturing facilities and an estimated manufacturing cost advantage of more than 30% over other high definition television (HDTV) flat panel technologies.
PACKAGE
With a targeted thickness of less than 2 centimeters and a weight under 10 kilograms for a 37-inch display, TDEL modules have the potential to be the thinnest and lightest flat panel television display.

TDEL is a solid-state technology, which does not require gas, liquid, or vacuum to operate and does not require a backlight or complex electronics. These characteristics result in a rugged, thinner and lighter display that is less sensitive to temperature variation, is easier to transport and can be truly defined as a hang-on-the-wall television.
5
The Westaim Corporation Annual Report 2005
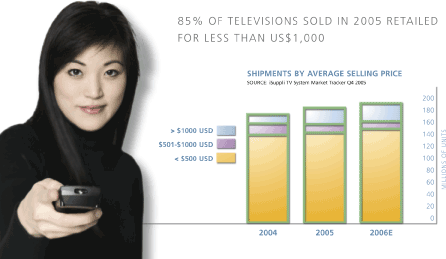
INDUSTRY OVERVIEW
The television landscape will ultimately consist of a number of flat panel technologies, including TDEL. The television industry is a US$86-billion market, which is large enough to offer ample opportunities for new TV technologies as consumers look for lighter, thinner and better performing high definition digital televisions at more affordable prices. The market is still dominated by traditional CRT-based TVs. This is expected to continue to be the case until a flat panel technology can compete with CRTs on cost. Currently, flat panel display prices remain too high for the mass consumer market. In 2005, only 14% of the units shipped consisted of flat panel TVs and more than 78% of televisions shipped sold for a retail price of less than US$500, contributing to the CRT television’s dominance in unit sales.
MARKET OPPORTUNITY
iFire plans to initially target the mid-30 to mid-40 inch screen size segment of the flat panel television market. This is a popular segment with consumers and it is a market that is currently not well served due to inherent technical and cost challenges faced by LCD and PDP. Currently LCD is too expensive and PDP encounters challenges achieving native high definition resolution in this size range.
LCD’s complex assembly methods and sensitivity to particle defects during manufacturing can contribute to lower production yields as screen size increases. To reduce the production costs of larger displays, LCD makers are moving to larger volume facilities, which produce multiple panels on a large, single substrate called a motherglass. These facilities require billions of dollars of capital to construct and provide a significant cost-of-entry barrier for new manufacturing companies to enter the market.
6
The Westaim Corporation Annual Report 2005
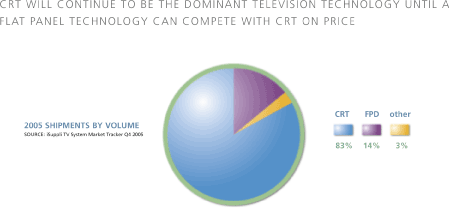
| CRT WILL CONTINUE TO BE THE DOMINANT TELEVISION TECHNOLOGY UNTIL A FLAT TECHNOLOGY CAN COMPLETE WITH CRT ON PRICE |
| 2005 SHIPMENTS BY VOLUME |
| SOURCE: SUPPLI TV SYSTEM MARKET TRACKER Q4 2005 |
PDP manufacturers have mainly focused on manufacturing displays larger than 42 inches because of difficulty in producing native high definition resolution (1280 columns by 720 rows or greater) when making smaller displays. To compensate, PDP manufacturers are focusing on products larger than 50 inches in size, or are offering lower resolution models called enhanced definition television or EDTV. In these models, the native HDTV image is stepped down to the resolution of the screen, reducing the quality of the image.
TDEL COST ADVANTAGE
iFire believes its displays can be produced at a much lower cost than LCD, especially in the mid-30 to mid-40 inch range. Even considering the most aggressive industry cost projections, iFire expects to enjoy a sustainable cost advantage over LCD well beyond the next decade. Today’s LCD manufacturing facilities are large multi-storey cleanrooms that require a capital investment in the range of US$2-3 billion to build. Comparatively, a TDEL facility with similar capacity is estimated to cost in the range of approximately 65% to 75% less due to its inherently simpler manufacturing process, less stringent cleanroom requirements and smaller facility size.
7
The Westaim Corporation Annual Report 2005

MANUFACTURING
In 2005, iFire entered the first phase of commercialization with the installation and start-up of a $46-million pilot production facility in Toronto, Canada. The extensive remodeling of iFire’s existing research and development facility included increasing the processing area from 15,000 to approximately 25,000 square feet, along with the installation of equipment to bring the operating capacity to approximately 10 panels per day. This project was completed on time and on budget, and is now in operation. In addition to serving as a pilot production facility, it will also function as iFire’s research and development headquarters.
Pilot production is a necessary step for new consumer technologies. For iFire, pilot production is intended to validate TDEL’s low-cost potential in a manufacturing environment and to produce engineering samples for evaluation by potential customers and partners. The new facility has been successful in manufacturing panels not only in greater numbers but also of much higher quality and consistency.
Investments in this facility included both process and handling equipment. The pilot is designed to process 34-inch panels with virtually no human operator contact. This hands-free material handling process uses cassettes to store and transport substrates, along with several robotic loading and unloading systems within the cleanroom.
iFire’s technology collaboration partner Dai Nippon Printing Co., Ltd, (DNP) fabricates the front-end portion of the iFire display at its primary production line in Kashiwa, Japan. Front-end manufacturing includes substrate preparation and the fabrication of row electrodes and thick-film dielectric layers on 34-inch glass substrates. DNP is a leader in the thick-film printing industry and has extensive experience in the printing techniques required to fabricate this part of the display.
8
The Westaim Corporation Annual Report 2005

The partially completed panels are then shipped to iFire, where the back-end processes, such as the deposition of phosphors, column electrodes, Color By Blue™ layers and electronics assembly, are completed in the new pilot production facility.
Two large in-line sputter systems have been installed for deposition of the thin film layers of the display, including iFire’s proprietary blue phosphor and the Indium Tin Oxide (ITO) used for the column electrodes. Inorganic electroluminescent phosphors have traditionally been fabricated by evaporation, but iFire has demonstrated that superior performance can be obtained by sputtering, a well-established manufacturing process. Sputtering has been demonstrated to be a more suitable process for volume production, providing faster throughput and more uniform deposition for larger substrates.
After the sputtered phosphor and ITO layers, a laser is used to pattern the top electrode. The laser has proven to be a fast and clean process which is more suitable to volume production than photolithography for this step. For creation of the Color By Blue™ layer, iFire has installed wet process tools which also demonstrate the throughput capacity necessary for volume production.
Throughout the construction of this facility, iFire has been working with leading equipment suppliers, familiar with the requirements of state-of-the-art flat panel display processing and glass handling. With the facility now in operation, iFire is strategically developing the supply chain required to support production volumes. The pilot plant will provide the information and experience to allow iFire to work with partners to construct and operate its first volume production facility.
9
The Westaim Corporation Annual Report 2005

TECHNOLOGY COLLABORATION
iFire’s proprietary TDEL technology has attracted the attention of major industry leaders from around the world. DNP, a leading printing technology and flat panel component manufacturer, provides the front end of pilot production to iFire and contributed to iFire’s pilot facility in Toronto in the form of a low-interest US$10-million financing arrangement. iFire has also collaborated with Sanyo Electric Co., Ltd. Working with Sanyo has given iFire access to the development, manufacturing and marketing experience of one of the world’s largest consumer electronic companies and has facilitated TDEL’s rapid advancement into commercialization.
iFire has also enjoyed a strong relationship with Ulvac Technologies Inc., the world leader in flat panel display production systems. Ulvac has played a substantial role in TDEL’s commercialization effort and most recently supplied multiple production systems for iFire’s pilot production facility.
OUTLOOK
iFire plans to commercialize its technology with manufacturing partners who can offer complementary skills and experience and help bring a critical mass to the business. iFire intends to follow a production schedule typical of many new consumer electronic technologies: following pilot manufacturing, iFire expects to begin commercial volume production with a capacity in the range of 250,000 units per year. This will be followed by a larger facility, with an estimated annual capacity of three million units per year. Initial planning work is already underway and iFire is reviewing its commercialization strategy with potential manufacturing partners.
iFire’s TDEL displays are set to introduce a new standard in flat panel television performance and design. With excellent picture and video quality in a thin and lightweight package, TDEL has the potential to deliver the ultimate flat panel viewing experience at a more affordable price.
10

| NUCRYST Pharmaceuticals Corp. |
| Innovative products that fight infection and inflammation |

The Westaim Corporation Annual Report 2005
looking in NUCRYST
PHARMACEUTICALS CORP.
NUCRYST Pharmaceuticals develops, manufactures, and commercializes medical products that fight infection and inflammation. Its patented technology converts silver’s microcrystalline structure into an atomically disordered nanocrystalline coating that significantly enhances silver’s medicinal properties. NUCRYST believes its atomically disordered nanocrystalline silver structures fulfill a large unmet need for broad spectrum antimicrobial products that are also able to kill antibiotic-resistant bacteria and inhibit inflammation.
PROPRIETARY NANOCRYSTALLINE NOBLE METAL TECHNOLOGY
Silver, platinum and gold, which are elements of the noble metals group, have long been known to have medicinal properties. Based on silver’s antimicrobial properties, NUCRYST focused on silver as the first noble metal for the application of its proprietary nanotechnology. Silver is generally composed of large microcrystals, usually one or two microns in diameter or greater. Although silver’s medicinal properties have been known for centuries, NUCRYST believes silver’s use in microcrystalline form has been limited due to slow release of relatively small quantities of silver ions.
The first application of NUCRYST’s nanocrystalline silver were coatings for medical devices called SILCRYST™ coatings. By converting silver’s microcrystalline structure into an atomically disordered nanocrystalline coating, NUCRYST’s nanocrystalline silver releases silver ions in greater quantities than naturally occurring microcrystalline silver.
NUCRYST’s SILCRYST™ coatings can also release silver particles over a sustained period of time and have a more rapid antimicrobial and more potent anti-inflammatory effect than microcrystalline silver.

The first products with the SILCRYST™ coatings were Acticoat™ dressings for serious wounds, including chronic wounds and life-threatening burns. SILCRYST™ coatings exhibit rapid antimicrobial activity, killing many organisms within 30 minutes, which is faster than many other commercially-available forms of antimicrobial silver dressings. Inin vitro tests, Acticoat™ dressings with SILCRYST™ nanocrystals have been shown to inhibit a broad range of organisms including gram positive, gram negative and antibiotic-resistant bacteria, as well as fungi and yeast.
Acticoat™ is a trademark of Smith & Nephew plc
SILCRYST™ is a trademark of NUCRYST Pharmaceuticals Corp.
11
The Westaim Corporation Annual Report 2005

Healthcare professionals around the world use dressings with NUCRYST’s SILCRYST™ coatings to combat infection in a wide variety of wound types, including life-threatening burns and chronic wounds. NUCRYST believes wound care products with its nanocrystalline silver combat infection longer than major silver-based wound care competitors, and offer a broader spectrum of antimicrobial activity than many topically applied antibiotics. In addition to antimicrobial activity, NUCRYST’s nanocrystalline silver has exhibited potent anti-inflammatory properties in preclinical studies. It suppresses two naturally occurring inflammatory cytokines, specifically IL-12B and TNF-alpha, and reduces the level of a naturally occurring enzyme called MMP-9. While helpful at the correct concentrations, excessive amounts of these substances are associated with inflammation. In addition, NUCRYST’s unique nanocrystalline silver increases the natural cell death of certain inflammatory cells, specifically polymorphonuclear leukocytes, or PMNs. NUCRYST believes these mechanisms of action may play important roles in NUCRYST’s nanocrystalline silver’s potent anti-inflammatory properties.
SILCRYST™ MEDICAL COATINGS
Advanced wound care products with NUCRYST’s SILCRYST™ coatings have received FDA clearance and the approval of other regulators in more than 30 countries and are sold by Smith & Nephew plc, a global medical device company, under its Acticoat™ trademark. Acticoat™ products are used for a wide variety of wound types by hospitals, clinics, burn centres, doctors’ offices, home healthcare agencies and nursing homes.
According to Frost & Sullivan, the advanced wound care products market was valued at approximately US$1.5-billion globally in 2004 and is projected to grow to approximately US$3.1 billion by 2011.
12
The Westaim Corporation Annual Report 2005

NUCRYST is working with Smith & Nephew to develop new Acticoat™ wound care products in the form of line extensions and innovative new dressing designs using SILCRYST™ coatings. Smith & Nephew’s 2005 launch of Acticoat™ Moisture Control product resulted from these efforts.
Since the introduction of Acticoat™ dressings in 1998, the product line has grown from one to four dressing designs with multiple sizes available for each dressing.
Acticoat™ 3/Acticoat™ Burn Dressingsare wound care dressings that provide up to three days of sustained antimicrobial activity. They were first sold in the United States in 1998 and in Europe in 2001.
Acticoat™ 7is a wound care dressing that provides up to seven days of sustained antimicrobial activity. It was first sold in the United States in 2000 and in Europe in 2001.
Acticoat™ Absorbentis an alginate dressing that provides up to three days of sustained antimicrobial activity for wounds with moderate to heavy excess fluid, or exudate. The product was first sold in 2001 and is currently sold in the United States, Canada and Europe.
Acticoat™ Moisture Controlis a foam dressing that provides antimicrobial activity for up to seven days for wounds with light to moderate exudate. Smith & Nephew currently sells the Acticoat™ Moisture Control dressing in the United States, Canada and Europe.
13
The Westaim Corporation Annual Report 2005

PHARMACEUTICAL RESEARCH & DEVELOPMENT
NUCRYST has produced its nanocrystalline silver as a powder for use as an active pharmaceutical ingredient, or API, known as NPI 32101. NUCRYST is developing pharmaceutical products to extend its nanocrystalline silver technology to the treatment of dermatological and gastrointestinal conditions.
NUCRYST’s lead pharmaceutical product candidate is a topical cream containing NPI 32101 for use in the treatment of dermatological disorders, such as atopic dermatitis, a form of eczema. Atopic dermatitis is an inflammatory skin disease often complicated by secondary infection and is treated primarily with topical steroids and TIMs (topical immunomodulators). More than 15 million people in the United States suffer from symptoms of atopic dermatitis, according to information published by the National Institute of Arthritis and Muscoskeletal and Skin Disease.
Prescription topical steroids and TIMs in the United States generated 2004 sales greater than US$600 million and US$400 million, respectively, according to IMS Health, a leading provider of prescription drug statistics.
In September 2004, NUCRYST completed a Phase 2 clinical trial of 0.5% and 1.0% dosages of NPI 32101 cream, tested against topical placebo cream, in 224 adult patients with mild to moderate atopic dermatitis. The results from this initial Phase 2 study provided important observations about the relationship between dosage strength of NPI 32101 topical cream and patient response as well as the relationship between the length of treatment with NPI 32101 and patient response. These results and an excellent safety profile suggested that a higher concentration of NPI 32101 and a study in children with atopic dermatitis should be considered.
14
The Westaim Corporation Annual Report 2005

Before initiating a large Phase 2 study in children, a small pharmacokinetic study in 36 adults and a small pediatric tolerance study in 30 children was conducted with NPI 32101 in a 2.0% dosage strength topical cream. In the pharmacokinetic study, NUCRYST evaluated serum concentrations and urinary excretion of silver. Based on observations during the study, NUCRYST believes that systemic exposure to silver in patients with atopic dermatitis treated with topical NPI 32101 is likely to be low. In the pediatric tolerance study, NUCRYST tested children and adolescents with atopic dermatitis for tolerance to NPI 32101 topical cream and did not observe any serious adverse events in any of the patients. Treatment-related events were generally mild, transitory and were not related to the dose of silver applied. The conclusion of these studies was that a 2% NPI 32101 cream is likely to be safe and well tolerated in both populations.
NUCRYST used these observations to guide the design of its second Phase 2 safety and efficacy clinical study. This second safety and efficacy Phase 2 clinical study is testing 1.0%, and 2.0% dosage strength creams against topical placebo cream in 345 children and adolescents with mild to moderate atopic dermatitis. The treatment period is 12 weeks, twice as long as the treatment period in the first Phase 2 safety and efficacy clinical study. Enrolment began in the fall of 2005 for this double-blind, randomized study, and involves 28 clinical sites across Canada and the United States. Results are expected by the end of 2006.
15
The Westaim Corporation Annual Report 2005

In addition to atopic dermatitis, NUCRYST is developing pharmaceutical products to extend its nanocrystalline silver technology for use in treating other conditions.
One area of research is for potential pharmaceutical products containing NPI 32101 for use in the treatment of gastrointestinal disorders. NUCRYST has begun to explore the use of its nanocrystalline silver technology to treat inflammatory bowel disease, which includes ulcerative colitis and Crohn’s disease. NPI 32101 has demonstrated a favorable effect on ulcerative colitis in a pre-clinical study where it appears to reduce signs of this disease. According to IMS Health, US prescription drug sales for the treatment of inflammatory bowel diseases were greater than $1 billion in 2004. An opportunity exists as many inflammatory bowel disease patients do not respond to currently available treatments.
POTENTIAL SILCRYST™ COATING MARKETS
NUCRYST is exploring applications for its SILCRYST™ coatings for a wide array of medical devices, such as orthopedic implants, pacemakers and meshes. Although these types of medical devices are implanted into patients using aseptic technique, infections can occur after implantation. If an infection occurs, patients must undergo additional treatments and possibly additional surgical procedures if the infection causes an implant failure. Some medical devices may benefit from SILCRYST™ coatings, which are designed to prevent and combat infection. NUCRYST is testing its coatings on materials used to make many medical devices such as titanium, stainless steel, cobalt chrome, silicone and various other polymer materials.
GROWTH STRATEGY
NUCRYST has a solid foundation as a new public company. It has a strong intellectual property portfolio and a successful relationship with a leading advanced wound management company. NUCRYST’s growth strategy is to:
| • | Support Smith & Nephew’s position as a leader in advanced wound care management by extending the application of SILCRYST™ coatings on the Acticoat™ product line and increasing production capacity to keep up with sales growth. |
| • | Leverage success in wound care by developing new products aimed at large markets with unmet medical needs. Current development activities include pharmaceutical products for dermatological and gastrointestinal conditions and non-wound care device coatings. |
| • | Collaborate with industry leaders to gain the benefits of their global marketing presence while avoiding the direct cost of developing its own sales and marketing capabilities. |
| • | Pursue patent protection for the results of ongoing discovery and development efforts. |
16

| LOOKING IN WESTAIM FINANCIAL REVIEW 2005 |

| LOOKING IN WESTAIM FINANCIAL REVIEW 2005 |
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
MANAGEMENT’S DISCUSSION AND ANALYSIS
This Management’s Discussion and Analysis of the results of operations and financial condition for the years ended December 31, 2005 and December 31, 2004 was prepared as at February 9, 2006 and should be read in conjunction with the Consolidated Financial Statements of The Westaim Corporation for the years ended December 31, 2005 and December 31, 2004 and the accompanying notes thereto. Additional information relating to The Westaim Corporation, including its annual information form, is available on SEDAR at www.sedar.com and the Company’s website (as hereinafter defined) at www.westaim.com. All dollar amounts contained herein are denominated in Canadian dollars unless otherwise specified.
DESCRIPTION OF THE BUSINESS
The Westaim Corporation (the “Company”) develops, commercializes and launches high potential technologies into certain fast growing sectors of the economy. The Company’s business opportunities include iFire Technology Corp. (“iFire”), a company with a novel flat panel display technology and Nucryst Pharmaceuticals Corp. (“Nucryst”), a company that develops, manufactures and commercializes innovative medical products that fight infection and inflammation.
The Company’s strategy is to develop the independent technical, operating and marketing and sales capabilities of its technology investments through the early years of product introduction and commercialization with the objective of taking these technologies public through initial public offerings. In December 2005, Nucryst completed an initial public offering of its common shares and its shares now trade on the Nasdaq National Market and the Toronto Stock Exchange. Management recognizes that, in circumstances where it lacks technical or marketing expertise or the necessary capital to complete development of a product, it may be in the Company’s best interests to pursue commercialization through joint venture arrangements, strategic alliances, licensing, or selling its technology.
CONSOLIDATED RESULTS
OVERVIEW
For the year ended December 31, 2005, the Company reported net income of $9.3 million compared to a net loss of $25.2 million in 2004. The loss from continuing operations was $5.5 million in 2005, which included a $30.1 million gain on issuance of the shares of Nucryst, compared to a loss from continuing operations of $28.7 million in 2004. In 2005, the Company reported income from discontinued operations of $14.8 million, including a one-time $11.3 million gain on the sale of inactive subsidiaries, compared to $3.6 million in 2004. Revenues from continuing operations for the year ended December 31, 2005 were $28.6 million compared to $31.9 million in 2004.
The basic and diluted net income per common share was $0.10 in 2005 compared to a net loss of $0.30 in 2004. The basic and diluted net loss per common share from continuing operations was $0.06 in 2005 and $0.34 in 2004. The basic weighted average number of common shares outstanding was 92.9 million and 84.1 million in 2005 and 2004 respectively.
Continuing operations reflect the results of the Company’s subsidiaries, Nucryst and iFire. Revenues decreased $3.3 million in 2005 reflecting lower milestone revenue from Nucryst, partially offset by higher royalty payments and manufacturing revenue earned from Nucryst’s wound care products.
A comparison of operating costs from continuing operations in 2005 compared to 2004 is as follows:
| % of | % of | |||||||||||||||
| ($millions) | 2005 | Revenue | * | 2004 | Revenue | * | ||||||||||
| Manufacturing | $ | 10.8 | 47.6 | % | $ | 8.5 | 44.9 | % | ||||||||
| General and administrative | 4.1 | 18.1 | % | 5.2 | 27.5 | % | ||||||||||
| Research and development | 36.5 | 161.0 | % | 35.5 | 187.8 | % | ||||||||||
| Depreciation and amortization | 7.3 | 32.2 | % | 5.7 | 30.2 | % | ||||||||||
| $ | 58.7 | $ | 54.9 | |||||||||||||
* revenues exclude Nucryst milestone revenue of $5.9 million in 2005 and $13.0 million in 2004
17
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OVERVIEW (continued)
The increase in manufacturing costs primarily reflects higher production volumes of Acticoat™ wound care products at Nucryst. Manufacturing costs measured as a percent of revenue, before milestone payments, have increased by 2.7 percentage points due to an increasing proportion of higher cost products being manufactured and to initial start-up inefficiencies related to the addition of increased manufacturing capacity at Nucryst in the second half of 2005. The decrease in general and administrative costs primarily reflects the cost of strategic analysis of future pharmaceutical applications for Nucryst’s nanocrystalline silver technology incurred in 2004. The increase in research and development expenses reflects slightly higher spending at iFire compared to 2004, partially offset by reduced expenditures relating to development of pharmaceutical products at Nucryst. All eligible research and development expenditures in Canada qualify for a 20% tax credit that can be utilized when the Company reaches a taxable position. No development costs were capitalized in 2005 and 2004. Depreciation increased by $1.6 million, or 28%, reflecting the completion of major capital projects at both Nucryst and iFire in 2005.
Corporate expenses for the year ended December 31, 2005 were $8.2 million, up from $7.4 million in 2004. The increase is primarily due to accrued expenses related to long term stock-based incentive programs reflecting the appreciation in the Company’s share price during 2005.
The foreign exchange gain in 2005 was $0.4 million compared to a foreign exchange loss of $0.6 million in 2004. The 2005 results benefited from favourable foreign exchange rates related to U.S. denominated working capital balances and Yen denominated long-term debt.
Interest income of $2.1 million in 2005 is $0.3 million less than 2004, reflecting lower average cash balances partially offset by slightly higher average interest rates.
The write down of assets of $0.6 million reflects write downs and losses on the disposal of research and development equipment no longer in use at iFire as a result of the transition to pilot manufacturing.
The Company recorded a $30.1 million gain on issuance of shares of a subsidiary as a result of Nucryst’s initial public offering in December 2005. The gain records the Company’s pro rata benefit from the net proceeds of this offering which is discussed more fully below under“Operations — Continuing Operations — Nucryst Operations”.
The gain on the sale of investments of $1.1 million in 2005 resulted from the sale of one of the Company’s portfolio investments in the fourth quarter of 2005.
Income from discontinued operations net of income taxes for the year ended December 31, 2005 was $14.8 million compared to $3.6 million in 2004. The increase reflects a $11.3 million gain on the sale of inactive subsidiaries and a $1.9 million gain on the sale of capital assets available for sale. The 2004 income from discontinued operations related primarily to the sale of the Company’s Ambeon division in January 2004. Discontinued operations are discussed more fully below under“Discontinued Operations”and in Note 4 to the audited consolidated financial statements of the Company for the year ended December 31, 2005.
Net income in the fourth quarter of 2005 was $22.0 million, including the $30.1 million gain on issuance of the shares of Nucryst, compared to a net loss of $10.6 million in 2004. The loss from continuing operations in the fourth quarter of 2005, before the gain on issuance of Nucryst shares, was $10.6 million compared to a loss of $10.4 million in 2004.
Income from discontinued operations in the fourth quarter of 2005 was $2.5 million compared to a loss of $0.2 million in the same period in 2004. The increase was primarily the result of a $1.9 million gain on sale of capital assets available for sale.
18
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OPERATIONS
Performance Measures
As a developer of new technologies, the Company uses financial and technical performance measures to track corporate performance. The Company develops comprehensive long-range plans and annual plans for each business segment with a view to maximizing long-term shareholder value. The success of each business unit is measured on its ability to achieve performance milestones within a specified timeframe. These milestones generally relate to specific research and development targets. Given the uncertainty surrounding developing new technologies, these milestones are reviewed and updated on a regular basis during the year. Financial milestones are also set and measured. Financial performance may relate to sales activity for commercial products or the achievement of results within operating expense and capital budget targets.
Continuing Operations
The Company’s business plans, and related measurements of performance against plan, are designed to ensure that the Company’s capital and human resources are focused on maximizing return on investment. The Company’s operations are organized into two high-potential emerging technology businesses — iFire and Nucryst. The Company’s primary strategy is to invest in the independent technical, research, operating, and marketing and sales capabilities of each of its technology investments, through the early years of product development, introduction and commercialization.
> iFire Operations
iFire, based in Toronto, Ontario, has developed a proprietary flat panel display with solid state, thick-film dielectric electroluminescent (“TDEL”) technology with primary application in the fast-growing large screen television market. For 30 to 40-inch screens, the Company believes that in high-volume production, iFire™ displays will have a 30% to 40% cost advantage over other flat panel technologies due to TDEL’s simpler structure, less complex manufacturing methods and fewer processing steps compared with liquid crystal displays (“LCD”) and plasma display panels (“PDP”). Unlike other flat panel technologies, the iFire™ displays do not contain gases (as with PDP), liquids (as with LCD) or vacuum (as with the cathode ray tube), making them inherently rugged and less susceptible to shock, vibration and breakage. TDEL technology’s solid state structure and thick-film manufacturing process also make an iFire™ display less sensitive to cleanroom contamination that is associated with PDP and LCD, which the Company believes will result in lower capital investment, higher manufacturing yield and reduced production costs. In addition, the iFire™ technology in large scale commercially produced displays is expected to feature full colour, rapid video response, unrestricted viewing angles and a wide operating temperature range.
In 2005, iFire continued to achieve strategic milestones toward proving the commercial viability of its technology as measured in scalability, colour, luminescence, lifetime and electronics. In late December 2005, iFire completed the $46 million pilot production facility in Toronto which utilizes its simplified flat panel manufacturing process, Colour-By-Blue™. The pilot production facility is intended to produce engineering samples of high definition 34-inch flat panel display modules and to simulate manufacturing in a commercial environment. The pilot plant will provide information and experience to allow iFire to work with partners to construct and operate the first volume production facility. Although no partnership agreements have been finalized, initial planning for this first volume production facility is underway.
iFire has been working with Dai Nippon Printing Co., Ltd. (“DNP”) of Japan since 2003 under a non-exclusive joint development agreement for commercial production of mid-30-inch screen size flat panel television modules. Under the terms of the agreement, DNP is utilizing its flat panel production line in Kashiwa, Japan for developing front-end manufacturing processes for iFire’s TDEL technology, including the substrate preparation and the fabrication of the row electrodes and thick-film dielectric layer. DNP regularly delivers partially completed substrates to iFire for further manufacturing. To complete the panel, back-end processes such as the deposition of phosphors, column electrodes and colour correction layers, as well as electronics assembly, are performed by iFire at its pilot manufacturing facility in Toronto.
19
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OPERATIONS (continued)
In 2004, iFire entered into a Yen 1.08 billion (approximately $10.7 million) loan agreement with DNP to partially fund equipment purchased for iFire’s pilot production facility in Toronto. The loan was drawn down during 2004 and 2005 and is repayable in full in Japanese Yen on June 30, 2006. At December 31, 2005, the loan outstanding amounted to 1,029,259,143 Yen (approximately $10.2 million).
iFire has also been working with Sanyo Electric Company Ltd. (“Sanyo”) since 2002 under a non-exclusive technology collaboration agreement in which Sanyo provides research and development expertise to iFire. As part of this agreement, Sanyo also provides funding to iFire for certain research and development projects. This funding is accounted for as a reduction in research and development expenses and amounted to $1.2 million in 2005 and $1.3 million in 2004 and has totaled $3.8 million since inception. Total funding is expected to amount to $4.9 million over 4 years.
> iFire Financial Results
The divisional loss at iFire for the year ended December 31, 2005 was $31.8 million compared to $27.9 million in 2004. The increase in 2005 is primarily the result of higher salaries and wages and higher depreciation and amortization expense. Approximately 50% of iFire’s cash expenses arise from salaries and wages of research and development staff and related support staff. Depreciation and amortization expense increased to $5.1 million in 2005 compared to $3.5 million in 2004 as a result of additional equipment and patents. Depreciation of the pilot manufacturing plant began in December 2005 and, as a result, depreciation and amortization expense in 2006 will increase by approximately $5.0 million in 2006 compared to 2005.
Capital spending amounted to $24.1 million in 2005 compared to $18.6 million in 2004. The increase is primarily related to the substantial completion of iFire’s pilot manufacturing facility.
> iFire Outlook
The 2006 outlook for iFire is for expenditures on research and development before depreciation to continue at levels moderately higher than 2005 reflecting the additional costs of operating the pilot manufacturing facility. With the substantial completion of the pilot manufacturing facility in 2005, iFire’s capital expenditures for research and development are expected to be lower in 2006. iFire intends to pursue a strategy of developing relationships with major international electronics companies with a view to jointly exploiting the commercialization of large format display products and sharing in the future development and capital costs of a large scale manufacturing facility. iFire does not expect to have revenues until a large scale manufacturing facility is completed and financing requirements will not be determinable until a manufacturing partner is selected and specific terms of a joint manufacturing arrangement are negotiated.
> Nucryst Operations
Nucryst researches, develops, manufactures and commercializes wound care products and pharmaceutical products that fight infection and inflammation. Nucryst’s patented technology enables it to convert silver’s microcrystalline structure into an atomically disordered nanocrystalline coating which the company believes enhances silver’s natural antimicrobial properties. In addition, Nucryst’s nanocrystalline silver has exhibited potent anti-inflammatory properties in preclinical studies.
On December 29, 2005, Nucryst completed its initial public offering of 4.5 million common shares and Westaim now owns 75.3% of Nucryst’s common shares. The net proceeds of this offering of US $39.1 million before repayment of intercompany debt to the Company of US $6.9 million will be used for capital expenditures, research and development, and general corporate purposes. Westaim’s return on its investment in Nucryst is dependent on a number of factors beyond its control. See“Risks and Uncertainties”.
Wound Care Products— Acticoat™ Burn Dressings and Acticoat™ 7 Dressings, targeting the burn and chronic wound markets, were developed and sold by Nucryst until May 2001 when a series of agreements were completed with Smith & Nephew plc (“Smith & Nephew”) under which Smith & Nephew acquired an exclusive global license to Nucryst’s SILCRYST™ antimicrobial coating technology for wound dressing products, together with Nucryst’s U.S. and Canada Acticoat™ burn dressing business, the Acticoat™ trademark, various regulatory approvals and certain manufacturing equipment that Nucryst leased back. Nucryst continues to manufacture Acticoat™ products exclusively for Smith & Nephew and receives reimbursement for manufacturing costs plus royalty payments and milestone payments based on Smith & Nephew’s global sales. All payments under the agreement with Smith & Nephew are made to Nucryst in U.S. dollars. The two companies are collaborating to develop the technology for the introduction of new products for chronic and serious wounds.
20
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OPERATIONS (continued)
The license Nucryst has granted to Smith & Nephew is exclusive, which means that Nucryst has agreed not to license the right to market, distribute or sell products with SILCRYST™ coatings for use on non-minor skin wounds and burns on humans to any other party. This exclusive right does not apply to other types of products that Nucryst may develop using its technology, such as the pharmaceutical products under development. Smith & Nephew has agreed to pursue the development and commercialization of products with SILCRYST™ coatings in the market for silver-based products for non-minor skin wounds and burns on humans. The license and development agreement expires in May 2026, although it may be terminated earlier by either party if the other party fails to cure a material breach of the agreement, suspends its operations or ceases to carry on business or files for bankruptcy or takes other similar actions. There are currently four product families with SILCRYST™ coatings manufactured by Nucryst for Smith & Nephew at its Fort Saskatchewan plant: Acticoat™ Burn, Acticoat™ 7, Acticoat™ Absorbent and Acticoat™ Moisture Control which was introduced in 2005.
Nucryst’s revenues under its agreements with Smith & Nephew consist of manufacturing reimbursements, royalties, payments upon the achievement of specified milestones and reimbursement for costs incurred in connection with the development or improvement of SILCRYST™ products covered by the agreements with Smith & Nephew. Nucryst receives reimbursement for the full cost of manufacturing products sold to Smith & Nephew. Manufacturing costs are recorded both as offsetting revenue and expense items on the statement of operations upon shipment to Smith & Nephew. Nucryst earns royalty revenues based upon Smith & Nephew’s sales of Acticoat™ products. Royalty revenue varies in proportion to increases or decreases in Smith & Nephew’s sales of its Acticoat™ products. Nucryst also receives milestone payments upon Smith & Nephew’s achievement of specified sales thresholds of Acticoat™ products and upon the achievement of specified regulatory events. To the end of 2005, Nucryst had earned US $19.0 million of a potential US $56.5 million in milestone payments.
Pharmaceutical Products– Nucryst is developing pharmaceutical products to extend its nanocrystalline silver technology to the treatment of dermatological conditions. Nucryst is producing a nanocrystalline silver powder referred to as NPI 32101 for use as a pharmaceutical ingredient, or API. The lead pharmaceutical candidate is a topical cream containing NPI 32101 for the treatment of dermatological conditions, such as atopic dermatitis. Atopic dermatitis is an inflammatory skin disease often complicated by secondary infection. Nucryst is also conducting preclinical research for the use of NPI 32101 in the treatment of gastrointestinal conditions. The company believes its nanocrystalline silver technology may be used to create a variety of additional pharmaceutical products that can treat medical conditions characterized by both infection and inflammation.
In 2003, Nucryst filed an Investigational New Drug (“IND”) application with the US Food and Drug Administration (“FDA”) for its first dermatology drug, NPI 32101 topical cream and completed Phase 1 clinical studies. In 2004, Nucryst announced the results of the Phase 2a clinical study of NPI 32101 topical cream in patients with mild to moderate atopic dermatitis. This double-blind, randomized, placebo-controlled study in 224 adult patients involved 23 clinical sites across the United States.
Patients were treated twice daily for a six-week period with one of two concentrations of NPI 32101, 0.5% and 1.0%, in a cream formulation or with the vehicle alone. The purpose of the study was to evaluate the safety and effectiveness of topical NPI 32101 in improving the signs and symptoms of atopic dermatitis.
In 2005, a pharmacokinetic study in adults and a pediatric tolerance study provided Nucryst with further data indicating that its NPI 32101 topical cream is likely to be safe and well tolerated in both populations. In the pharmacokinetic study, Nucryst evaluated serum concentrations and urinary excretion of silver in 18 adult patients with atopic dermatitis and 18 matching healthy adult controls following four times daily application of 1% and 2% NPI 32101 cream for two weeks. Silver could not be detected in the serum of a majority of the subjects, and when silver was detected the concentrations were low. There was no correlation with the concentration of cream, area covered with cream or the presence or absence of disease. Similarly, urinary silver excretion was not related to these factors or to the detection of silver in the serum. Based on these observations, Nucryst believes that systemic exposure to silver in patients with atopic dermatitis treated with topical NPI 32101 is likely to be low.
21
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OPERATIONS (continued)
In the pediatric tolerance study, Nucryst tested 30 children and adolescents with atopic dermatitis for tolerance to NPI 32101 topical cream in 1% and 2% dosage strengths applied twice daily for 2 weeks compared to the cream containing no silver. Nucryst did not observe any serious adverse events in any of these patients. Two patients in the placebo group and none in the NPI 32101-treated groups withdrew for adverse events. Treatment-related events were generally mild, transitory and were not related to the dose of silver applied. Treatment-related events were higher in the NPI 32101 group than in the placebo group, with 20%, 70% and 60% of patients experiencing at least one adverse event for administration site conditions for placebo, 1% and 2% groups, respectively.
Findings in the first Phase 2 clinical study provided Nucryst with important guidance to optimize the product, protocol and study design for future clinical trials. Nucryst used these observations to guide design of its second Phase 2 safety and efficacy clinical study. This second safety and efficacy Phase 2 clinical study is testing placebo, 1.0%, and 2.0% dosage strength creams in 345 children and adolescents with mild to moderate atopic dermatitis. The treatment period is 12 weeks, which is twice as long as the treatment period in the first Phase 2 safety and efficacy clinical study. A double-blind randomized study began enrolment in the fourth quarter of 2005 and involves 28 clinical sites across Canada and the United States. Results are expected by the end of 2006.
In addition to its clinical studies described above, Nucryst is continuing to conduct preclinical and non-clinical studies of its NPI 32101 topical cream in order to generate the carcinogenicity, toxicology and other data that will need to be submitted to the FDA as part of any New Drug Application (“NDA”). Phase 3 trials, which involve hundreds of patients in numerous clinical centres, are expected to take a year or more to complete and are expected to commence upon achieving successful results in Phase 2 studies. If favorable results are achieved in Phase 3, Nucryst anticipates submitting a New Drug Application to the FDA in the 2009 timeframe.
> Nucryst Financial Results
Nucryst’s operating income for the year ended December 31, 2005 was $1.6 million compared to income of $6.1 million in 2004.
The financial results of Nucryst are summarized as follows:
| 2005 | 2004 | 2003 | ||||||||||
| Wound care product revenue | $ | 22.7 | $ | 18.9 | $ | 11.6 | ||||||
| Milestone revenue | 5.9 | 13.0 | 4.6 | |||||||||
| Total revenue | $ | 28.6 | $ | 31.9 | $ | 16.2 | ||||||
Manufacturing costs(1) | $ | 11.4 | $ | 8.8 | $ | 6.2 | ||||||
| Wound care gross margin excluding milestone revenue | $ | 11.3 | $ | 10.1 | $ | 5.4 | ||||||
| Nucryst operating income (loss) | $ | 1.6 | $ | 6.1 | $ | (2.0 | ) | |||||
| (1) | Manufacturing costs include related depreciation and are net of intercompany charges |
Revenue–Total revenue for the year ended December 31, 2005 was $28.6 million compared to $31.9 million for the year ended December 31, 2004. The decrease of $3.3 million is attributable primarily to US $10.0 million in milestone revenue being earned in 2004 compared to US $5.0 million in the same period of 2005, which was offset in part by increased wound care product revenue in 2005. Wound care product revenue increased approximately 20% to $22.7 million for the year ended December 31, 2005 compared to $18.9 million in the same period of 2004. The $3.8 million improvement in wound care product revenue reflects increased orders from, and sales by, Smith & Nephew as their sales of Acticoat™ products continue to grow. This improvement was partially offset by the impact of the strengthening Canadian dollar which resulted in lower revenues reported in Canadian dollars. Milestone revenues for the year ended December 31, 2005 earned for the achievement of predetermined Smith & Nephew sales thresholds of Acticoat™ products were US $5.0 million compared to US $10.0 million in the same period of 2004.
Manufacturing Costs– Manufacturing costs, including related depreciation, for the year ended December 31, 2005 were $11.4 million compared to $8.8 million for the year ended December 31, 2004. The increase of $2.6 million, or 30%, is attributable to higher production volumes of Acticoat™ wound care products driven by increased orders from Smith & Nephew to support its Acticoat™ sales growth.
22
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
OPERATIONS (continued)
Gross Margin– Gross margin excluding milestone revenue for the year ended December 31, 2005 was $11.3 million compared to $10.1 million for the year ended December 31, 2004. This reduction in gross margin excluding milestone revenue is primarily the result of an increase in volume of lower margin Acticoat™ products manufactured for, and sold by, Smith & Nephew in 2005 compared to 2004 and the introduction of new manufacturing capacity in 2005.
Research and development, and general and administrative costs of $15.6 million for the year ended December 31, 2005 were $1.4 million less than the $17.0 million incurred in 2004. The decrease compared to 2004 is primarily attributable to the Phase 2 dermatological clinical study that was undertaken in 2004.
Capital spending totaled $4.5 million in 2005 compared to $3.2 million in 2004 and in both years related primarily to the addition of manufacturing capacity of Acticoat™ products in Fort Saskatchewan, Alberta. The success of the Acticoat™ product line has resulted in the need to further increase production capacity at Nucryst’s Fort Saskatchewan facility. Nucryst is currently adding a production line to this facility which will be operational by early 2007. Nucryst has advised that the total cost of this capital project is expected to be approximately $7.0 million. Nucryst anticipates that additional expansion may be required in 2007 to meet projected sales growth. Capital projects related to pharmaceutical development are expected to total approximately $2.3 million in 2006. Nucryst’s administration and pharmaceutical research activities are primarily based in Wakefield, Massachusetts, and manufacturing operations are located in Fort Saskatchewan, Alberta.
> Nucryst Outlook
The Company believes that the outlook for Nucryst is for continued year over year growth in licensing and manufacturing revenues as markets for Acticoat™ wound care products are expanded in the United States, Europe and other markets by Smith & Nephew. One US $5.0 million milestone payment was received in 2005 and additional milestone payments are expected to be earned by Nucryst in future years. Research and development expenditures will increase in 2006 as Nucryst expands clinical and pre-clinical research into the pharmaceutical attributes of its noble metal nanocrystalline technology.
DISCONTINUED OPERATIONS
> Sale of Subsidiaries
In January 2005, the Company completed a series of transactions whereby two inactive wholly owned subsidiary companies participated in a Plan of Arrangement with two widely held publicly traded companies. This resulted in a reorganization of the participating companies into a single company and the dilution of the Company’s investment to approximately 6.0%. The Company’s pro rata interest in the book value of the recapitalized companies amounted to $2.2 million. Subsequently, the company created under the Plan of Arrangement was listed on the Toronto Stock Exchange. The inactive subsidiaries were previously carried at nominal value and this transaction resulted in the Company recording a dilution gain of $2.2 million in discontinued operations in the first quarter of 2005. In February 2005, the Company sold its investment in the publicly traded company for net proceeds of $11.5 million and recorded an additional gain on sale of investments of $9.3 million in the first quarter of 2005.
> Ambeon
On January 29, 2004, the Company sold its Ambeon division for net proceeds of $33.4 million. The completion of the sale to a strategic buyer represented an opportunity to enhance the Company’s value and to focus resources on the Company’s two high potential technologies, iFire and Nucryst. Under Canadian Generally Accepted Accounting Principles (“GAAP”) introduced in 2003, all expected future losses and estimated shutdown and asset disposal costs relating to discontinued operations are no longer accrued, but are to be reported in the period in which they occur. As a result, the gain from the sale of Ambeon of $5.4 million was reported in the first quarter of 2004 and discontinued operations costs totaling $1.9 million relating to the restructuring of the Company as a result of the sale of Ambeon were reported in the subsequent quarters in which they occurred.
In the second quarter of 2005, the Company sold the remaining operations and net assets relating to Ambeon for $0.9 million. The gain on sale of this transaction, amounting to $0.7 million, has been included in discontinued operations.
23
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONSOLIDATED RESULTS
DISCONTINUED OPERATIONS (continued)
> Ethylene Coatings business
In February 2005, the land and building associated with the Company’s former ethylene coatings business was sold. The closing date of this transaction was December 15, 2005 and the purchaser leased the facility during the interim period. The gain on sale of this asset amounting to $1.9 million was recorded in the fourth quarter of 2005.
Details of the financial impact of discontinued operations are disclosed in Note 4 to the 2005 audited consolidated financial statements.
LIQUIDITY AND CAPITAL RESOURCES
The Company’s business plans are developed to ensure research and development costs do not overextend the Company’s financial resources. The Company’s 2006 consolidated business plan indicates that Nucryst will have sufficient cash and short-term investments to fund its pharmaceutical product development and the Company will have sufficient cash and short-term investments to fund the flat screen commercialization plans at iFire. The Company has no material lines of credit or other available debt facilities.
At December 31, 2005, the Company had cash, cash equivalents and short-term investments of $119.6 million compared to $101.1 million at December 31, 2004. This improvement in liquidity of $18.5 million is primarily the result of financing initiatives discussed below, including the initial public offering of Nucryst in December 2005, additional draws on the DNP loan facility, issuance of convertible debentures by a wholly-owned subsidiary and proceeds from the sale of discontinued operations. These initiatives provided approximately $81.0 million in cash to the Company. Cash used in continuing operations amounted to $32.3 million compared to $21.0 million in 2004, reflecting planned higher operating losses at Nucryst and iFire. Discontinued operations used $0.5 million in 2005 compared to $2.2 million in 2004.
In December 2005, Nucryst completed its initial public offering of 4,500,000 shares at US $10.00 per share. Net proceeds after commissions and other costs amounted to US $39.1 million. US $6.9 million of the proceeds were used to repay a portion of Nucryst’s indebtedness to the Company. The remaining proceeds are anticipated to be used by Nucryst to fund its capital projects and pharmaceutical development. At December 31, 2005, Nucryst had cash and short term investments of $41.9 million. These funds are not accessible to the Company to fund the future operations and capital projects at iFire or its own operations.
Proceeds from the sale of discontinued operations included $11.5 million from the sale of inactive subsidiaries; $7.4 million from the sale of land and buildings; $2.8 million from the collection of escrow funds relating to the sale of Ambeon in 2004, and $0.9 million from the sale of the remaining Ambeon operations.
Capital expenditures for continuing operations of $30.8 million in 2005 were $7.3 million higher than the same period in 2004. As discussed earlier, the Company completed major capital projects in both iFire and Nucryst. In relation to ongoing projects, the Company has outstanding purchase commitments amounting to approximately $2.5 million as at December 31, 2005 compared to $18.0 million at December 31, 2004. Capital expenditures in 2006 could increase to as much as $200 million if iFire determines it is beneficial to construct a large volume flat screen production facility on its own without a strategic partner.
The DNP loan at iFire of 1,029,259,143 Yen ($10.2 million) at December 31, 2005, is payable in full on June 30, 2006. The Company has entered into forward transactions to hedge the foreign currency exposure of this loan. The Company will be required to pay any exchange losses on the date of settlement. The unrealized losses on these hedges amounted to approximately $2.0 million at December 31, 2005.
DIVIDENDS
No dividends were paid in 2005 or 2004. The Company’s current policy is to retain its cash reserves to finance capital projects and business growth.
24
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CRITICAL ACCOUNTING POLICIES
The Company’s consolidated financial statements are prepared in accordance with Canadian generally accepted accounting principles and reported in Canadian dollars. The Company’s accounting policies are disclosed in Note 2 to the 2005 audited consolidated financial statements.
The Company has established detailed policies and control procedures that are intended to ensure that management judgments and estimates are well controlled, independently reviewed and consistently applied from period to period. The following are key policies that may impact the Company’s financial condition and results from operations and that require significant judgments by management. Management believes that its estimates for determining the valuation of the Company’s assets and liabilities are appropriate.
Revenue Recognition— The Company recognizes revenue from the sale of products based upon Nucryst’s licensing and supply agreements with Smith & Nephew in accordance with GAAP. The agreements provide for reimbursements of manufacturing costs and research and development costs, and for royalties and milestone payments. Nucryst recognizes manufacturing cost reimbursement as revenue upon shipment of product from its manufacturing facility and records royalty revenues upon the sale of products by Smith & Nephew to its customers. Nucryst is also eligible to earn additional royalties when specified gross margin thresholds have been achieved by Smith & Nephew. Additional royalties are recognized in the period of sale by Smith & Nephew to its customers. Milestone payments are recognized as revenue when Smith & Nephew achieves the agreed sales levels or receives the agreed regulatory approvals.
Research and Development Costs— Development costs are capitalized once the Company has determined that commercialization criteria concerning the product or process have been met. The Company reviews the progress of research and development initiatives on a regular basis and has determined that, in 2005 and 2004, no development costs should be deferred and amortized.
Site Restoration Costs— Future site restoration costs have been estimated by qualified employees of the Company taking into consideration the anticipated method and extent of the remediation consistent with regulatory requirements, industry practices, current technology and possible uses of the site. Provision details are based on cost estimates provided by independent consultants. Significant judgment is required in the determination of these provisions and the Company takes a conservative approach in not accruing potential recoveries from third parties where indemnifications are in place. It is the Company’s view that where remediation costs will be incurred many years into the future, third party recoveries cannot be estimated with certainty. Revisions to cost estimates and the recovery of actual remediation costs could result in material changes to the provision in future periods. Also see “Environmental Matters”.
Effective January 1, 2004, the Company adopted Section 3110 “Asset Retirement Obligations” of the Canadian Institute of Chartered Accountants (“CICA”) Handbook which addresses the financial accounting and reporting obligations associated with the retirement of tangible, long-lived assets and their associated net retirement costs.
Under the new Section, an asset retirement obligation is recognized at its fair value in the period in which it is incurred. Asset retirement costs are capitalized as part of the carrying amount of the long-lived asset and a related amortization expense is recognized in future periods. Implementation of CICA 3110 did not have an impact on the Company’s results from operations or its financial position as the assets subject to these new rules have been sold or written down to nominal value.
Income Taxes— The provision for income taxes is calculated based on the expected tax treatment of transactions recorded in the consolidated financial statements. In determining the provision for income taxes and, in particular, any future tax asset, the Company interprets tax legislation in a variety of jurisdictions and makes assumptions about the timing and certainty of the reversal of the future tax assets. Changes to these interpretations could have a material effect on income tax provisions in future periods.
Stock-Based Compensation— Canadian GAAP required that effective January 1, 2004, the fair value method of accounting for stock options must be recognized in the financial statements. It provided for alternate methods of implementation and the Company elected to apply the provisions retroactively with restatement of prior years. The Company determined that electing this method of applying the new rules had the most conservative impact on its financial results.
25
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CRITICAL ACCOUNTING POLICIES (CONTINUED)
For US GAAP, the Company continues to measure compensation expense using the intrinsic value method for stock options granted to directors and employees and to provide pro forma disclosure of compensation expense for all outstanding options issued by the Company as if the fair value methodology had been applied. In December 2004, the U.S. Financial Accounting Standards Board issued a new standard amending the accounting for stock-based compensation. The Standard requires the use of a fair-value-based method to measure and account for the stock-based compensation. The requirements for this Standard will be effective beginning January 1, 2006. The adoption of this Standard will not have a material impact on the Company’s operating results or financial position.
Variable Interest Entities– The Company adopted the CICA guidelines on the consolidation of variable interest entities (“VIEs”) on January 1, 2005. VIEs include entities where the equity invested is considered insufficient to finance the entity’s activities. Under this new guideline, the Company is required to consolidate VIEs if the investments held in these entities and/or the relationships with them result in the Company being exposed to a majority of their expected losses, being able to benefit from a majority of their expected residual returns, or both, based on a calculation outlined by the standard setters. The implementation of this guideline did not have an impact on the Company’s operating results or financial position.
ENTERPRISE RISK MANAGEMENT
The Company invests in new technologies with the objective of providing leadership, strategy and capital to commercialize the technology as quickly as possible. There is significant risk that the technology may not be commercialized in a timely or cost-effective manner or that it may not be accepted by the marketplace. The Company reduces this risk by investing in multiple technologies and product lines but these risks could have a material adverse impact on the Company’s business prospects, financial condition, and results from operations.
RISKS AND UNCERTAINTIES
Westaim may be unable to develop commercially viable products.
Some of the Company’s products, such as iFire’s flat panel television displays, are still in the developmental stage. The Company will likely continue to incur significant research and development costs before any of these products are commercially viable, and there is no assurance that any of its products will ever reach this stage or that the products will achieve the level of market penetration expected. Some or all of the technological obstacles that will need to be overcome in order to make these products commercially viable may prove to be insurmountable.
If Westaim fails to raise the capital necessary to fund its operations, it may be unable to advance the development and commercialization of its technologies.
A commitment of substantial resources by the Company and its collaborators to conduct research and development and construct pilot or large scale manufacturing facilities will be required to successfully commercialize products under development. The Company may not be able to raise additional capital at the time it is needed to complete product development and build manufacturing facilities. Additional capital may be required to fund operations, continue the research and development of product candidates, commercialize products and construct pilot and full scale manufacturing facilities. If the Company is unable to raise additional funds when required, it may be necessary to delay, reduce or eliminate some or all of its development programs.
The commercial potential of the Company’s products depends upon certain issues regarding pricing and production costs.
The Company’s ability to commercialize iFire’s flat-screen technology successfully will depend in part on its ability to price iFire’s products at a point that will generate consumer demand, while allowing for an adequate profit margin. The Company believes that iFire’s product can be produced at costs lower than other flat-screen technologies but there is no assurance that there will be consumer demand for the iFire product or that competing products will not be developed and priced below the prices required by iFire to be profitable. There is no assurance that the Company’s present cost estimates for its manufacturing facilities will be valid at the time that those facilities are constructed.
26
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
RISKS AND UNCERTAINTIES (CONTINUED)
Westaim’s success is dependent upon its ability to form partnerships to develop and sell its products.
The Company’s and its subsidiaries’ ability to successfully develop, manufacture and market their current and proposed products will depend, to a large extent, on their ability to form partnerships or joint ventures with established corporations or other collaborators. Except as described elsewhere in this document, the Company and its subsidiaries have not yet entered into any material partnerships or joint ventures for the development or marketing of these products, nor will they necessarily be able to do so in the future. The Company and its subsidiaries may be unable to find suitable partners or form a partnership or joint venture on terms that are beneficial. If the Company and its subsidiaries do enter into a partnership or joint venture, they may suffer losses if the partner becomes insolvent or otherwise fails to meet their obligations.
Westaim’s products may become technologically obsolete.
The Company and its subsidiaries compete, and intend to compete, in markets that are characterized by rapid adaptation to technological change. These markets include, but are not limited to, the medical devices, pharmaceuticals and flat panel television markets. The current and future products of the Company and its subsidiaries may be quickly rendered obsolete and unmarketable. The Company and its subsidiaries will need to continually develop new products and enhance existing products to keep pace with evolving technologies, customer preferences and industry standards.
Westaim is developing products for highly competitive markets.
The Company and its subsidiaries face, and will face, competition from a number of other companies including major domestic and international companies which have substantially greater financial, technical, marketing, sales, distribution and other resources. Many of these competitors may also have greater name or brand recognition. Competitors may produce more technologically-advanced products, at a lower cost, than the Company or its subsidiaries are capable of producing. Competition may cause the Company or its subsidiaries to lose market share and may reduce profit margins on any products that it is able to sell.
Westaim may be unable to protect its intellectual property.
In order to succeed, the Company and its subsidiaries will need to prevent their intellectual property from being misappropriated by third parties. To protect their intellectual property, the Company and its subsidiaries rely primarily on their confidentiality agreements, physical security at research and manufacturing facilities, as well as the copyright, trade secret, trademark and patent laws of Canada, the United States, and other countries in which the Company and its subsidiaries conduct, or will conduct, business. The laws of other countries may not protect intellectual property rights to the same extent as the laws of Canada and the United States and, in any event, the methods that the Company and its subsidiaries have chosen may fail to adequately prevent misappropriation of their intellectual property.
The Company and its subsidiaries cannot provide assurance that they will succeed in obtaining new patents; that they will be able to enforce existing patents against third parties; or that existing patents will not be successfully challenged by third parties. Even if the Company and its subsidiaries are able to prevent the misappropriation of intellectual property, others may independently and legally develop technologies that are substantially equivalent or superior.
Westaim may become involved in expensive intellectual property or product liability litigation.
The Company and its subsidiaries may be required to commence litigation to enforce their intellectual property rights. Others may claim that the Company or its subsidiaries have infringed upon their intellectual property rights and commence litigation. The Company believes that it and its subsidiaries will be subject to an increasing number of infringement claims as they begin to produce more products in more industries.
Some of the Company’s and its subsidiaries’ existing and proposed products, such as Nucryst’s medical devices and pharmaceuticals, are part of a class of product that is particularly vulnerable to product liability litigation for a number of reasons:
| – | These types of products are extremely complex and the Company and its subsidiaries may fail to discover product faults, despite their best efforts to do so; |
| – | These types of products will interact with very complex biological and man-made systems and may interact with these systems in harmful ways that the Company and its subsidiaries were unable to anticipate, despite their best efforts to do so; and |
| – | Because these products may be used by a large number of people, if these products do cause harm, the Company or its subsidiaries may be exposed to a large number of claims for damages. |
27
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
RISKS AND UNCERTAINTIES (CONTINUED)
The Company and its subsidiaries have tried to protect themselves against product liability litigation by including limitation of liability provisions in some of their sales agreements. There is no assurance, however, that existing or future limitation of liability provisions will be sufficient to protect the Company and its subsidiaries in all circumstances, nor can assurance be provided that any of these provisions will be held to be enforceable by the Courts.
The Company believes that it has obtained sufficient product liability insurance coverage to protect it against claims. However, the wording of its insurance policies may exclude some claims. Furthermore, the Company cannot provide assurance that its insurance limits will be sufficient, nor can it ensure that it will be able to acquire satisfactory insurance in the future.
Westaim may be unable to retain the required highly skilled people.
The Company’s technology businesses are dependent upon the talents and knowledge of certain key individuals in each of the businesses. The marketplace for people with these skills is highly competitive, and the Company may not be able to retain a sufficient number of people with the skills that it requires. The Company provides competitive remuneration and incentives for the retention of key personnel.
Westaim is subject to certain risks because of the international character of its business.
The Company estimates that sales to international customers accounted for over 90% of its net sales in the fiscal year ended December 31, 2005 and the Company anticipates that international sales will continue to represent a material portion of net sales in the future. International sales are subject to inherent risks, including variations in local economies, fluctuating exchange rates, greater difficulty in the collection of accounts receivable, changes in tariffs and other trade barriers, adverse foreign tax consequences and burdens of complying with a variety of foreign laws. The Company may also encounter exchange rate risk in the event international sales are denominated in a currency other than Canadian dollars.
The Company’s financial results are reported in Canadian dollars. A significant portion of the Company’s revenue and expenses, as well as accounts payable, accounts receivable and other balance sheet items, are frequently denominated in currencies other than the Canadian dollar, primarily in United States dollars. Fluctuations in the exchange rate between these other currencies and the Canadian dollar could reduce the Company’s reported revenue, increase the Company’s costs or give rise to a charge related to foreign currency translation, all of which could adversely affect operating results.
Westaim’s success is dependent on the success of Nucryst.
Westaim’s shareholdings in Nucryst represent a substantial portion of Westaim’s assets. Nucryst completed its initial public offering in December 2005 and, although Westaim retained a majority interest in Nucryst, Westaim now has a limited ability to control the operations of Nucryst and the associated costs, which could adversely affect Westaim’s financial performance. Westaim’s return on its investment in Nucryst will depend upon a number of factors that may be beyond Westaim’s control, including the risk factors set forth above that relate to Nucryst. In addition, Nucryst is subject to several additional risks including, but not limited to the following:
| – | Nucryst is dependent on its relationship with Smith & Nephew and Smith & Nephew is currently its only customer; |
| – | Nucryst has a history of net losses and negative cash flow from operations; this will likely continue in the future and Nucryst’s cash resources may not be adequate to accomplish its objectives; |
| – | Many of Nucryst’s proposed products will require regulatory approval before Nucryst is allowed to sell them. The regulatory approval process will be lengthy and expensive and Nucryst will have the burden of proving that its products are safe and effective or that, if approved, such approval could cause its products to become prohibitively expensive. There is no assurance that Nucryst will ever obtain regulatory approval to sell any of its proposed products, or that the conditions imposed by regulators will be satisfactory to Nucryst. Regulatory requirements imposed on Nucryst’s products could limit Nucryst’s ability to test, manufacture and commercialize its products; and |
| – | Nucryst’s ability to commercialize its pharmaceutical products successfully will depend in part on the extent to which reimbursement for the cost of such products and related treatments will be available from government health administration authorities, private health insurers and other organizations and there can be no assurance that adequate third party coverage will be available for Nucryst to realize an appropriate return on its investment in product development. |
A failure on the part of Nucryst to properly manage the above risks or any of the other risks it is subject to, will have an adverse impact on the value of Westaim’s investment in Nucryst.
28
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
VOLATILITY OF SHARE PRICE
Market prices for securities of companies developing new technologies are generally volatile. Factors such as announcements of technological innovations, new commercial products, patents, the development of proprietary rights, results of clinical trials, regulatory actions, publications, quarterly financial results, the Company’s financial position, public concern over the safety of biotechnology, future sales of shares by the Company or by our current shareholders, and other factors, could have a significant effect on the market price and volatility of the Company’s common shares.
The price of the common shares may be volatile even though there have been no material changes in the Company’s business or finances. In the past, securities class action litigation has often been brought against companies that experience volatility in the market price of their securities. Moreover, market prices for stocks of technology companies frequently reach levels that bear no relationship to the operating performance of such companies. These market prices generally are not sustainable and are subject to wide variations. Whether or not meritorious, litigation brought against the Company could result in substantial costs, divert management’s attention and resources and harm the Company’s financial condition and results of operations.
ENVIRONMENTAL MATTERS
The Company’s operations are subject to extensive federal, provincial and municipal environmental statutes and regulations, including those relating to air emissions, wastewater discharges, contaminated soil and groundwater, and the handling and disposal of hazardous substances and wastes.
The Company’s operations in Toronto are conducted under an environmental operating approval from the Ontario Ministry of Environment. The Company’s operations in Wakefield, Massachusetts are conducted under various state and federal permits.
The Company operates under a “best management practices program” called Safety and Environment Management Practices (“SEMP”) which incorporates both an environmental management system and an occupational health and safety management system. This program is regularly reviewed and updated to keep pace with or stay ahead of regulatory changes and is internally audited every year.
The provision for site restoration at December 31, 2005 of $6.8 million is comparable to the $6.7 million provision at December 31, 2004. The provision relates primarily to site restoration costs associated with soil and groundwater reclamation and remediation costs. The Company spent $0.1 million in 2005, and $0.2 million was recovered from a third party. The Company expects to spend only nominal amounts in future years unless a plant site is decommissioned.
MARKET FOR SECURITIES
The common shares of The Westaim Corporation are listed on The Toronto Stock Exchange under the symbol “WED” and on NASDAQ under the symbol “WEDX”.
OUTSTANDING SHARE DATA
The Company’s authorized share capital consists of an unlimited number of common shares, preferred A shares and preferred B shares. As at December 31, 2005 and February 9, 2006, there were 92,900,649 common shares outstanding. There were no preferred shares outstanding at December 31, 2005 and February 9, 2006.
The Company maintains an employee and director stock option plan under which the Company may grant options for up to 10,750,000 shares of common stock of the Company at an exercise price equal to the closing market price of the Company’s stock for the trading day immediately preceding the date of grant. As at December 31, 2005 and February 9, 2006, there were 5,049,600 options outstanding.
29
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
CONTRACTUAL COMMITMENTS
| Payments due by Period | ||||||||||||||||||||
| ($000) | Less than | 1-3 | 3-5 | More than 5 | ||||||||||||||||
| As at December 31, 2005 | Total | 1 Year | Years | Years | Years | |||||||||||||||
| Long-term debt | $ | 16,312 | $ | 10,312 | $ | 6,000 | $ | — | $ | — | ||||||||||
| Capital lease obligations | — | — | — | — | — | |||||||||||||||
| Operating lease operations | 2,173 | 763 | 1,086 | 202 | 122 | |||||||||||||||
Supplier purchase obligations(1) | 9,375 | 9,375 | — | — | — | |||||||||||||||
| Other long-term liabilities reflected on the Company’s consolidated balance sheet | — | — | — | — | — | |||||||||||||||
| $ | 27,860 | $ | 20,450 | $ | 7,086 | $ | 202 | $ | 122 | |||||||||||
| (1) | Included in supplier purchase obligations are commitments for capital expenditures totaling $2.5 million and agreements to purchase goods or services that are enforceable and legally binding on the Company and that specify all significant terms. |
DISCLOSURE CONTROLS AND PROCEDURES
The Chief Executive Officer and Chief Financial Officer evaluated the effectiveness of the Company’s disclosure controls and procedures (as defined in the rules of the Securities and Exchange Commission and the Canadian Securities Administrators) and concluded that the Company’s disclosure controls and procedures were effective as of December 31, 2005 and in respect of the 2005 year end reporting period.
SELECTED CONSOLIDATED FINANCIAL INFORMATION
The following table sets forth certain financial information for the Company for 2003 to 2005:
| Year Ended | Year Ended | Year Ended | ||||||||||
| ($000, except per share data) | Dec. 31, 2005 | Dec. 31, 2004 | Dec. 31, 2003 | |||||||||
| Revenue from continuing operations | $ | 28,560 | $ | 31,907 | $ | 17,123 | ||||||
Loss from continuing operations(1) | (5,508 | ) | (28,734 | ) | (37,849 | ) | ||||||
| Loss per common share from continuing operations – basic and diluted | (0.06 | ) | (0.34 | ) | (0.48 | ) | ||||||
| Net income (loss) | 9,270 | (25,177 | ) | (35,440 | ) | |||||||
| Net income (loss) per common share – basic and diluted | 0.10 | (0.30 | ) | (0.45 | ) | |||||||
| Total assets | 212,461 | 172,263 | 144,906 | |||||||||
| Total long-term debt | 16,312 | 4,795 | — | |||||||||
| Dividends declared | — | — | — | |||||||||
| (1) | As disclosed in Note 4 to the audited consolidated financial statements, and discussed in this MD&A, the Ambeon business segment has been accounted for as a discontinued operation in 2005, 2004 and 2003. |
30
The Westaim Corporation Annual Report 2005
MANAGEMENT’S DISCUSSION & ANALYSIS
QUARTERLY INFORMATION
| Q1 | Q2 | Q3 | Q4 | |||||||||||||
| ($000, except per share data) | 2005 | 2005 | 2005 | 2005 | ||||||||||||
| Revenue from continuing operations | $ | 4,977 | $ | 5,708 | $ | 12,268 | $ | 5,607 | ||||||||
| (Loss) income from continuing operations | (8,858 | ) | (11,003 | ) | (5,130 | ) | 19,483 | |||||||||
| (Loss) income per common share from continuing operations – basic and diluted | (0.10 | ) | (0.12 | ) | (0.06 | ) | 0.21 | |||||||||
| Net income (loss) | 2,532 | (10,218 | ) | (5,042 | ) | 21,998 | ||||||||||
| Net income (loss) per common share – basic and diluted | 0.03 | (0.11 | ) | (0.05 | ) | 0.24 | ||||||||||
Net income in the first quarter of 2005 includes a gain on the sale of inactive subsidiaries of $9.3 million and a related dilution gain of $2.2 million. Loss in the second quarter of 2005 includes a gain of $0.7 million on the sale of the remaining operations and assets relating to Ambeon. Net income from continuing operations in the fourth quarter of 2005 includes a gain on the sale of investments of $1.1 million and a gain on the issuance of shares of a subsidiary of $30.1 million.
| Q1 | Q2 | Q3 | Q4 | |||||||||||||
| ($000, except per share data) | 2004 | 2004 | 2004 | 2004 | ||||||||||||
| Revenue from continuing operations | $ | 10,565 | $ | 4,799 | $ | 10,501 | $ | 6,042 | ||||||||
| Loss from continuing operations | (3,201 | ) | (12,662 | ) | (2,468 | ) | (10,403 | ) | ||||||||
| Loss per common share from continuing operations – basic and diluted | (0.04 | ) | (0.16 | ) | (0.03 | ) | (0.11 | ) | ||||||||
| Net income (loss) | 1,008 | (13,071 | ) | (2,514 | ) | (10,600 | ) | |||||||||
| Net income (loss) per common share – basic and diluted | 0.01 | (0.17 | ) | (0.03 | ) | (0.11 | ) | |||||||||
| The results of operations of the Ambeon business segment have been accounted for as discontinued operations. |
The gain on sale of Ambeon of $5.4 million was included in net income in the first quarter of 2004.
Revenue from continuing operations, loss from continuing operations, and net income (loss) were favourably impacted by the reporting of milestone revenues in Nucryst of US $5.0 million in the third quarter of 2005, US $5.0 million in the first quarter of 2004, and US $5.0 million in the third quarter of 2004.
31
The Westaim Corporation Annual Report 2005
MANAGEMENT’S RESPONSIBILITY FOR FINANCIAL INFORMATION
February 9, 2006
The accompanying consolidated financial statements, the notes thereto and other financial information contained in this annual report have been prepared by, and are the responsibility of, the management of The Westaim Corporation. Financial information contained throughout this annual report is consistent with the consolidated financial statements. These consolidated financial statements have been prepared in accordance with generally accepted accounting principles, using management’s best estimates and judgements when appropriate. The Board of Directors is responsible for ensuring that management fulfills its responsibility for financial reporting and internal control. Policies and procedures are designed to give reasonable assurance that transactions are properly authorized, assets are safeguarded and financial records properly maintained to provide reliable information for the preparation of financial statements. The Audit Committee, which is comprised of three Directors, none of whom is an officer of the Company, meets with management as well as the external auditors to satisfy itself that management is properly discharging its financial reporting responsibilities and to review the consolidated financial statements and the report of the auditors. It reports its findings to the Board of Directors who approve the consolidated financial statements.
The consolidated financial statements have been audited by Deloitte & Touche LLP, the independent auditors, in accordance with generally accepted auditing standards. The auditors have full and unrestricted access to the Audit Committee.
 |  | |
Barry M. Heck | G.A. (Drew) Fitch | |
| President and | Senior Vice President and | |
| Chief Executive Officer | Chief Financial Officer |
32
The Westaim Corporation Annual Report 2005
AUDITORS’ REPORT
To the Shareholders of The Westaim Corporation
We have audited the consolidated balance sheets of The Westaim Corporation as at December 31, 2005 and 2004, and the consolidated statements of operations and deficit and cash flow for each of the years in the three-year period ended December 31, 2005. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with Canadian generally accepted auditing standards. Those standards require that we plan and perform an audit to obtain reasonable assurance whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation.
In our opinion, these consolidated financial statements present fairly, in all material respects, the financial position of the Company as at December 31, 2005 and 2004, and the results of its operations and cash flow for each of the years in the three-year period ended December 31, 2005 in accordance with Canadian generally accepted accounting principles.
The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Deloitte & Touche LLP
Independent Registered Chartered Accountants
Calgary, Canada
February 7, 2006
February 7, 2006
COMMENTS BY INDEPENDENT REGISTERED CHARTERED ACCOUNTANTS FOR U.S. READERS ON CANADA — U.S. REPORTING DIFFERENCES
The standards of the Public Company Accounting Oversight Board (United States) require the addition of an explanatory paragraph (following the opinion paragraph) when there are changes in accounting principles that have been implemented in the financial statements, such as the changes described in Notes 2(k), 2(l), 2(n) and 2(s) to the consolidated financial statements. Our report is expressed in accordance with Canadian reporting standards which do not require a reference to such changes in accounting principles in the report of Independent Registered Chartered Accountants when the changes are properly accounted for and adequately disclosed in the financial statements.
Deloitte & Touche LLP
Independent Registered Chartered Accountants
Calgary, Canada
February 7, 2006
February 7, 2006
33
The Westaim Corporation Annual Report 2005
CONSOLIDATED BALANCE SHEETS
| December 31 | December 31 | |||||||
| (thousands of Canadian dollars) | 2005 | 2004 | ||||||
ASSETS | ||||||||
| Current | ||||||||
| Cash and cash equivalents | $ | 115,673 | $ | 89,139 | ||||
| Short-term investments | 3,954 | 12,000 | ||||||
| Accounts receivable | 8,199 | 6,712 | ||||||
| Inventories (note 5) | 7,773 | 3,605 | ||||||
| Other | 570 | 538 | ||||||
| 136,169 | 111,994 | |||||||
| Other receivable (note 4) | — | 1,800 | ||||||
| Capital assets (note 6) | 71,112 | 46,776 | ||||||
| Capital assets available for sale (note 4) | — | 5,500 | ||||||
| Intangible assets (note 7) | 5,180 | 5,693 | ||||||
| Investments | — | 500 | ||||||
| $ | 212,461 | $ | 172,263 | |||||
LIABILITIES AND SHAREHOLDERS’ EQUITY | ||||||||
| Current | ||||||||
| Accounts payable and accrued liabilities | $ | 16,302 | $ | 12,963 | ||||
| Current portion of long-term debt (note 9) | 10,312 | — | ||||||
| 26,614 | 12,963 | |||||||
| Long-term debt (note 9) | 6,000 | 4,795 | ||||||
| Provision for site restoration (note 10) | 6,760 | 6,708 | ||||||
| 39,374 | 24,466 | |||||||
| Non-controlling interest (note 3) | 15,447 | — | ||||||
| Guarantees (note 14) | ||||||||
| Commitments and contingencies (note 15) | ||||||||
| Shareholders’ equity | ||||||||
| Common shares (note 11) | 421,466 | 421,233 | ||||||
| Contributed surplus (note 11) | 3,968 | 3,125 | ||||||
| Cumulative translation adjustment (note 12) | (503 | ) | — | |||||
| Deficit | (267,291 | ) | (276,561 | ) | ||||
| 157,640 | 147,797 | |||||||
| $ | 212,461 | $ | 172,263 | |||||
| Approved on behalf of the Board: | ||
 |  | |
| Ian W. Delaney | Frank W. King | |
| Director | Director |
34
The Westaim Corporation Annual Report 2005
CONSOLIDATED STATEMENTS OF OPERATIONS AND
CONSOLIDATED STATEMENTS OF DEFICIT
CONSOLIDATED STATEMENTS OF DEFICIT
| Year Ended | Year Ended | Year Ended | ||||||||||
| December 31 | December 31 | December 31 | ||||||||||
| 2005 | 2004 | 2003 | ||||||||||
| (thousands of Canadian dollars except per share data) | (Restated – note 4) | (Restated – note 4) | ||||||||||
| Revenue | $ | 28,560 | $ | 31,907 | $ | 17,123 | ||||||
| Costs | ||||||||||||
| Manufacturing | 10,799 | 8,450 | 5,835 | |||||||||
| Research and development (note 13) | 36,514 | 35,489 | 25,871 | |||||||||
| General and administrative | 4,109 | 5,240 | 3,817 | |||||||||
| Depreciation and amortization | 7,338 | 5,722 | 5,001 | |||||||||
| Operating loss | (30,200 | ) | (22,994 | ) | (23,401 | ) | ||||||
| Corporate costs | (8,217 | ) | (7,396 | ) | (9,550 | ) | ||||||
| Foreign exchange | 410 | (563 | ) | (2,081 | ) | |||||||
| Interest | 2,073 | 2,362 | 2,734 | |||||||||
| Gain on issuance of shares of subsidiary (note 3) | 30,055 | — | — | |||||||||
| Writedown of capital assets and intangible assets | (570 | ) | (22 | ) | (723 | ) | ||||||
| Gain on sale of investment | 1,120 | — | — | |||||||||
| Loss from continuing operations before income taxes | (5,329 | ) | (28,613 | ) | (33,021 | ) | ||||||
| Income tax expense (note 8) | ||||||||||||
| Current | (179 | ) | (121 | ) | (130 | ) | ||||||
| Future | — | — | (4,698 | ) | ||||||||
| (179 | ) | (121 | ) | (4,828 | ) | |||||||
| Loss from continuing operations | (5,508 | ) | (28,734 | ) | (37,849 | ) | ||||||
| Income from discontinued operations net of income taxes (note 4) | 14,778 | 3,557 | 2,409 | |||||||||
| Net income (loss) | $ | 9,270 | $ | (25,177 | ) | $ | (35,440 | ) | ||||
| Income (loss) per common share (note 17) | ||||||||||||
| Continuing operations – basic and diluted | $ | (0.06 | ) | $ | (0.34 | ) | $ | (0.48 | ) | |||
| Net income (loss) – basic and diluted | 0.10 | (0.30 | ) | (0.45 | ) | |||||||
| Weighted average number of common shares outstanding | ||||||||||||
| Basic | 92,852,120 | 84,093,827 | 78,044,689 | |||||||||
| Diluted | 93,120,936 | 84,093,827 | 78,044,689 | |||||||||
| Deficit at beginning of year | $ | (276,561 | ) | $ | (251,384 | ) | $ | (215,944 | ) | |||
| Net income (loss) | 9,270 | (25,177 | ) | (35,440 | ) | |||||||
| Deficit at end of year | $ | (267,291 | ) | $ | (276,561 | ) | $ | (251,384 | ) | |||
35
The Westaim Corporation Annual Report 2005
CONSOLIDATED CASH FLOW STATEMENTS
| Year Ended | Year Ended | Year Ended | ||||||||||
| December 31 | December 31 | December 31 | ||||||||||
| 2005 | 2004 | 2003 | ||||||||||
| (thousands of Canadian dollars) | (Restated – note 4) | (Restated – note 4) | ||||||||||
| Operating activities | ||||||||||||
| Loss from continuing operations | $ | (5,508 | ) | $ | (28,734 | ) | $ | (37,849 | ) | |||
| Items not affecting cash | ||||||||||||
| Depreciation and amortization | 7,338 | 5,722 | 5,001 | |||||||||
| Foreign exchange on long-term debt | (1,637 | ) | — | — | ||||||||
| Provision for site restoration | — | 327 | — | |||||||||
| Stock-based compensation expense | 905 | 1,210 | 942 | |||||||||
| Future income taxes | — | — | 4,698 | |||||||||
| Writedown of capital assets and intangible assets | 570 | 22 | 723 | |||||||||
| Deferred licensing revenue | — | — | (565 | ) | ||||||||
| Gain on sale of investment | (1,120 | ) | — | — | ||||||||
| Gain on issuance of shares of subsidiary (note 3) | (30,055 | ) | — | — | ||||||||
| Accounts receivable | (2,541 | ) | (670 | ) | 365 | |||||||
| Inventories | (4,341 | ) | (831 | ) | (544 | ) | ||||||
| Other | (32 | ) | 121 | 136 | ||||||||
| Accounts payable and accrued liabilities | 4,045 | 2,996 | 2,407 | |||||||||
| Site restoration expenditures net of recoveries | 52 | (1,183 | ) | (185 | ) | |||||||
| Cash used in continuing operations | (32,324 | ) | (21,020 | ) | (24,871 | ) | ||||||
| Cash (used in) provided from discontinued operations | (518 | ) | (2,221 | ) | 3,383 | |||||||
| Cash used in operating activities | (32,842 | ) | (23,241 | ) | (21,488 | ) | ||||||
| Investing activities | ||||||||||||
| Capital expenditures – continuing operations | (30,754 | ) | (23,432 | ) | (10,383 | ) | ||||||
| Capital expenditures – discontinued operations | — | — | (1,745 | ) | ||||||||
| Intangible assets – continuing operations | (977 | ) | (3,686 | ) | (725 | ) | ||||||
| Intangible assets – discontinued operations | — | — | (234 | ) | ||||||||
| Redemption and sale of short-term investments | 98,335 | 149,151 | 53,332 | |||||||||
| Purchase of short-term investments | (90,289 | ) | (137,708 | ) | (51,525 | ) | ||||||
| Proceeds on sale of investment | 1,620 | — | — | |||||||||
| Proceeds on sale of assets | — | 61 | 1,308 | |||||||||
| Proceeds on sale of discontinued operations (note 4) | 22,614 | 30,559 | — | |||||||||
| Cash provided from (used in) investing activities | 549 | 14,945 | (9,972 | ) | ||||||||
| Financing activities | ||||||||||||
| Proceeds from long-term debt (note 9) | 7,154 | 4,795 | — | |||||||||
| Issuance of common shares of subsidiary, net of share issuance costs (note 3) | 45,502 | — | — | |||||||||
| Issuance of common shares (note 11) | 171 | 47,946 | 102 | |||||||||
| Issuance of convertible debentures of subsidiary (note 9) | 6,000 | — | — | |||||||||
| Cash provided from financing activities | 58,827 | 52,741 | 102 | |||||||||
| Net increase (decrease) in cash and cash equivalents | 26,534 | 44,445 | (31,358 | ) | ||||||||
| Cash and cash equivalents at beginning of year | 89,139 | 44,694 | 76,052 | |||||||||
| Cash and cash equivalents at end of year | $ | 115,673 | $ | 89,139 | $ | 44,694 | ||||||
36
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1 BASIS OF PRESENTATION
The Westaim Corporation (the “Company”) was incorporated on May 7, 1996 by articles of incorporation under the Business Corporations Act (Alberta).
The consolidated financial statements include the accounts of the Company and its subsidiaries, iFire Technology Corp. (“iFire”) and Nucryst Pharmaceuticals Corp. (“Nucryst”).
These consolidated financial statements are prepared in accordance with Canadian generally accepted accounting principles (“GAAP” or “Canadian GAAP”). In addition, Note 19 describes and reconciles the significant measurement differences between Canadian and United States generally accepted accounting principles (“U.S. GAAP”) affecting these consolidated financial statements.
All amounts are expressed in thousands of Canadian dollars except share and per share data unless otherwise noted.
2 SUMMARY OF SIGNIFICANT ACCOUNTING PRINCIPLES
a) Principles of consolidation
The financial statements of entities which are controlled by the Company through voting equity interests, referred to as subsidiaries, are consolidated. Entities which are jointly controlled, referred to as joint ventures, and entities which are not controlled but over which the Company has the ability to exercise significant influence, referred to as associated companies, are accounted for using proportionate consolidation and equity method, respectively. Investments in other entities are accounted for using the cost method. Variable interest entities (“VIEs”) (which include, but are not limited to, special purpose entities, trusts, partnerships, certain joint ventures and other legal structures), as defined by the Canadian Institute of Chartered Accountants (“CICA”) in Accounting Guideline 15 “Consolidation of Variable Interest Entities”, are entities in which equity investors do not have the characteristics of a “controlling financial interest” or there is not sufficient equity at risk for the entity to finance its activities without additional subordinated financial support. VIEs are consolidated by the Company when it is determined that it will, as the primary beneficiary, absorb the majority of the VIEs expected losses and/or expected residual returns. The Company currently does not have any VIEs. Intercompany accounts and transactions are eliminated upon consolidation and unrealized intercompany gains and losses are eliminated when accounting under the equity method.
b) Use of estimates
GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the reported amounts of revenue and expenses during the year. Significant estimates include the provision for site restoration, inventory valuation, fair value of stock-based compensation, valuation allowance against future income taxes, and useful lives of capital assets and intangible assets. Actual results could differ from those estimates.
c) Translation of foreign currencies
Transactions in foreign currencies are translated into Canadian dollars at rates of exchange at the time of such transactions. Monetary assets and liabilities are translated at current rates of exchange. Foreign operations are considered financially and operationally self-sustaining and are translated into Canadian dollars using the current rate method of translation. Under this method, assets and liabilities are translated at the year-end exchange rates. Unrealized gains and losses arising from translating net investments in foreign operations into Canadian dollars are included in shareholders’ equity as a cumulative translation adjustment.
The Company changed its method of foreign exchange translation for Nucryst’s United States based subsidiary (Note 3) from the temporal to the current rate method upon completion of Nucryst’s initial public share offering in December 2005. Under the temporal method, all foreign exchange adjustments arising from the translation of the subsidiary’s financial statements into Canadian dollars were included in the consolidated statement of operations. As required under GAAP, the change in accounting method was applied prospectively and resulted in a cumulative translation adjustment of $503 being recorded as a separate component of shareholders’ equity.
37
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
2 SUMMARY OF SIGNIFICANT ACCOUNTING PRINCIPLES (CONTINUED)
d) Revenue recognition
Specific revenue recognition policies for each of the Company’s operating segments are as follows:
>Nucryst Pharmaceuticals– Revenue from direct sales to third parties is recognized when persuasive evidence of an arrangement exists, delivery has occurred, the price to the buyer is fixed or determinable, and collection is reasonably assured. The Company’s revenues earned under license agreements consist primarily of manufacturing cost reimbursements, royalties and payments upon the achievement of specific milestones. For products manufactured under license, revenue is recorded at the date of shipment. Royalty revenue is earned based on a percentage of sales revenue earned by the licensee on its sales to third parties. Revenue relating to the achievement of milestones under licensing agreements is recognized when the milestone event has occurred. Nucryst also derives revenue from research activities and this revenue is recognized as services are performed.
>iFire Technology– The Company recognizes revenue from licensing fees over the term that services are being rendered. Deferred licensing revenue represents amounts received under agreements for which the process of earning the revenue has not been completed. Royalties are recognized when earned in accordance with the terms of the specific agreement. As at December 31, 2005 and 2004, iFire was not party to any licensing fee or royalty agreements.
e) Cash and cash equivalents
Cash and cash equivalents consist of cash on deposit and highly liquid short-term investments with original maturities at the date of acquisition of 90 days or less and are recorded at cost.
f) Short-term investments
Short-term investments consist of money-market instruments with maturities of less than one year. As at December 31, 2005, the Company held short-term investments recorded at a cost of $3,954 (2004 – $12,000) which reflects current market values.
g) Inventory valuation
Finished product, raw materials, materials in process, and spare parts and operating materials are valued at the lower of average cost and net realizable value.
h) Research and development costs
Research costs are expensed as incurred and significant project development costs are capitalized in accordance with GAAP once the Company has determined that commercialization criteria concerning the product or process have been met. Amortization of these costs over their estimated useful life commences with the successful commercial production or use of the product or process. On an ongoing basis, management reviews the unamortized balance to ensure that the deferred development costs continue to satisfy the criteria for deferral and amortization.
As at December 31, 2005 and 2004, no development costs have been capitalized.
i) Government assistance
Government assistance towards research and development expenditures related to capital assets used for research and development is credited against the cost of the related capital asset and all other assistance is credited against the related expenses as incurred.
j) Capital assets
Land, buildings, machinery and equipment are stated at cost. Depreciation is calculated using a straight-line method based on estimated useful lives of the particular assets which is 20 years for buildings and 3 to 10 years for machinery and equipment.
The Company evaluates the carrying value of capital assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable, and recognizes an impairment charge when it is probable that estimated future non-discounted cash flows of the underlying assets will be less than the carrying value of the assets. Measurement of an impairment loss related to capital assets that management intends to hold and use is based on the fair value of the assets, whereas assets to be disposed of are reported at the lower of the carrying amount or fair value less costs to sell.
38
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
2 SUMMARY OF SIGNIFICANT ACCOUNTING PRINCIPLES (CONTINUED)
k) Site restoration costs and asset retirement obligations
The total estimated costs of site restoration relating to tangible, long-lived assets have been accrued. Site restoration costs have been estimated, taking into consideration the anticipated method and extent of the remediation consistent with regulatory requirements, industry practices, current technology and the possible uses of the site. The estimated amount of future restoration costs is reviewed regularly based on available information. Where the forecasted net restoration costs exceed existing provisions, an additional expense is recognized in the period.
Potential recoveries of costs resulting from indemnifications provided by previous owners of the Company’s industrial sites have not been recognized in these consolidated financial statements as the amount of recovery cannot be reasonably determined. Any future recoveries will be recorded when received.
Effective January 1, 2004, the Company adopted Section 3110 “Asset Retirement Obligations” of the CICA Handbook which addresses the financial accounting and reporting obligations associated with the retirement of tangible, long-lived assets and their associated net retirement costs. Under this Section, an asset retirement obligation is recognized at its fair value in the period in which it is incurred. Asset retirement costs are capitalized as part of the carrying amount of the long-lived asset and a related amortization expense is recognized in future periods. Implementation of Section 3110 did not have an impact on the Company’s results from operations or its financial position as the assets subject to these new rules have been sold or written down to nominal value.
l) Derivative financial instruments
Derivative financial instruments may be utilized by the Company in the management of interest rate and foreign currency exposures. The Company’s policy is not to utilize derivative financial instruments for trading or speculative purposes.
The Company formally documents all relationships between hedging instruments and hedged items, as well as its risk management objective and strategy for undertaking various hedge transactions. This process includes linking all derivatives to specific assets and liabilities on the balance sheet or to specific firm commitments or forecasted transactions. The Company also formally assesses, both at the hedge’s inception and on an ongoing basis, whether the derivatives that are used in hedging transactions are highly effective in offsetting changes in fair values or cash flows of hedged items.
The Company may enter into forward agreements in order to reduce the impact of fluctuating interest rates on its short-term investments and fluctuating foreign currency exchange rates on anticipated future cash flows. These forward agreements would not be designated as hedges for accounting purposes. They require the exchange of payments without the exchange of the notional principal amount on which payments are based. These instruments are recorded at the lower of cost or market. At December 31, 2005 and 2004, there were no open forward rate agreement contracts.
Effective January 1, 2004, the Company adopted new Accounting Guideline 13 “Hedging Relationships” for identifying, designating and documenting hedge relationships, and assessing their effectiveness. The guideline provides parameters on the conditions necessary for hedge accounting to be applied, but does not specify the methods to be used in its application. The guideline, however, does require that the Company adopt an accounting policy for assessing the effectiveness of its hedge relationships. Any ineffectiveness related to instruments recorded in the statement of financial position is to be recognized in income for the period. The new guideline has not had a significant impact on the Company’s financial results.
m) Income taxes
Income taxes are accounted for using the liability method of income tax allocation. Under the liability method, income tax assets and liabilities are recorded to recognize future income tax inflows and outflows arising from the settlement or recovery of assets and liabilities at their carrying values.
Income tax assets are also recognized for the benefits from tax losses and deductions that cannot be identified with particular assets or liabilities, provided those benefits are more likely than not to be realized. Future income tax assets and liabilities are determined based on the tax laws and rates that are anticipated to apply in the period of realization.
39
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
2 SUMMARY OF SIGNIFICANT ACCOUNTING PRINCIPLES (CONTINUED)
n) Stock-based compensation plans
The Company and certain of its subsidiaries have stock-based compensation plans, which are described in Note 11. Compensation expense for awards that call for settlement in cash or other assets is measured on an ongoing basis as the amount by which the quoted market price exceeds the exercise price at each measurement date. Any obligations related to increases in the value of Deferred Share Units (“DSUs”), Restricted Share Units (“RSUs”), Employee Bonus Appreciation Units (“BAUs”), and Stock Appreciation Rights (“SARs”) are accrued when the change in value occurs with an offset to the consolidated statement of operations.
Any consideration paid by option holders for the purchase of stock is credited to capital stock. If plan entitlements are repurchased from the holder, the consideration paid is charged to retained earnings.
Effective January 1, 2004, the Company adopted revisions to Section 3870 of the CICA Handbook with respect to the accounting and disclosure of stock-based compensation. The revised Section 3870 requires that the fair value of stock options be recorded in the financial statements. These new rules applied to options issued on or after January 1, 2002. The Company elected to apply the provisions retroactively with restatement of prior years.
o) Earnings per share
Basic earnings per common share is calculated using the weighted average number of common shares outstanding during the period. Diluted earnings per share is calculated on the basis of the average number of shares outstanding during the period plus the additional common shares that would have been outstanding if potentially dilutive common shares had been issued using the “treasury stock” method.
p) Employee future benefits
All employee future benefits are accounted for on an accrual basis. The Company maintains defined contribution pension plans for its employees. These plans were funded $745 during the year ended December 31, 2005 (2004 – $602; 2003 –$784).
q) Discontinued operations
Long-lived assets to be disposed of by sale are classified as held for sale in the period in which a formal plan of disposal has been approved, the assets are available for immediate sale and are actively being marketed, and it is expected that the sale will occur within one year. Long-lived assets to be abandoned are classified as held and used until they are disposed of.
Long-lived assets classified as held for sale are carried at the lower of their carrying amount and fair value net of estimated disposition costs. Losses are recognized immediately where carrying value exceeds fair value and gains are recognized at the time of sale.
The results of discontinued operations are reported separately, including gains or losses related to the disposal of related long-lived assets held for sale or disposal. Future costs associated with an exit or disposal activity are recognized in the period in which the liability is incurred.
r) Intangible assets
The Company’s definite life intangible assets consist of the prosecution and application costs of patents and trademarks and are amortized on a straight-line basis over their estimated useful lives to a maximum of 10 years. The cost of maintaining patents and trademarks are expensed as incurred. The Company evaluates the carrying value of definite life intangible assets whenever events or changes in circumstances indicate that the carrying value may not be recoverable. Any impairment in the carrying value is charged to expense in the period that impairment has been determined.
Indefinite life intangible assets are recorded at fair value. On a regular basis, the Company reviews the carrying value of these assets for impairment. As at December 31, 2005 and 2004, the Company had no indefinite life intangible assets.
40
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 2 | SUMMARY OF SIGNIFICANT ACCOUNTING PRINCIPLES (CONTINUED) |
| s) | Recent adopted and pending accounting pronouncements |
i) The Company adopted the CICA guideline on the consolidation of variable interest entities (“VIEs”) on January 1, 2005.
VIEs include entities where the equity invested is considered insufficient to finance the entity’s activities. Under this new guideline, the Company is required to consolidate VIEs if the investments in these entities and/or the relationships with them result in the Company being exposed to a majority of their expected losses, being able to benefit from a majority of their expected residual returns, or both, based on a formula determined by the standard-setters. The implementation of this guideline did not have a material impact on the Company’s consolidated statement of operations or consolidated financial position.
ii) In 2005, the CICA approved amendments to new Section 3831 “Non-Monetary Transactions” which would converge this Section with U.S. GAAP and with equivalent requirements in International Financial Reporting Standards.
This Section requires that all non-monetary transactions be measured at fair value, unless certain criteria are met. The amendments replace culmination of the earnings process as the test for applying fair value measurement with a test of commercial substance. Culmination of the earnings process has been difficult to apply in practice and has depended on an assessment of whether the assets exchanged were similar. Commercial substance, however, involves satisfying one of two objectively determined tests based on assessments of cash flows expected with and without the exchange of the non-monetary items. The new requirements apply to non-monetary transactions initiated in periods beginning on or after January 1, 2006. The Company does not expect adoption of this new Section to have a material impact on the Company’s consolidated statement of operations or consolidated financial position.
iii) In January 2005, the CICA issued a new Section to the CICA Handbook, Section 1530 “Comprehensive Income”. Certain gains and losses arising from changes in fair value will be temporarily recorded outside the income statement in comprehensive income. This Section will apply for annual financial statements for fiscal years beginning on or after October 1, 2006. The Company is in the process of evaluating the impact of this new Section.
iv) In January 2005, the CICA issued Section 3855 “Financial Instruments – Recognition and Measurement” and Section 3865 “Hedges”. Under these Sections, all financial instruments including derivatives are to be included on a company’s balance sheet and measured, either at their fair value or, in limited circumstances, when fair value may not be considered most relevant, at cost or amortized cost. These Sections specify when gains and losses, as a result of changes in fair value, are to be recognized in the income statement. These Sections will apply for annual financial statements for fiscal years beginning on or after October 1, 2006. The Company is in the process of evaluating the impact of these recently issued Sections.
v) In January 2005, the CICA issued a new Section to the CICA Handbook, Section 3251 “Equity”. This Section establishes standards for the presentation of equity during a reporting period. This Section will apply for annual financial statements for fi scal years beginning on or after October 1, 2006. The Company is in the process of evaluating the impact of this Section on its consolidated statement of operations and consolidated financial position.
vi) In October 2005, the CICA Emerging Issues Committee issued Abstract No. 157, “Implicit Variable Interests Under AcG-15” (EIC-157). This EIC clarifies that implicit variable interests are implied financial interests in an entity that change with changes in the fair value of the entity’s net assets exclusive of variable interests. An implicit variable interest is similar to an explicit variable interest except that it involves absorbing and/or receiving variability indirectly from the entity. The identification of an implicit variable interest is a matter of judgment that depends on the relevant facts and circumstances. EIC-157 will be effective in the first quarter of 2006. The implementation of this EIC is not expected to have a material impact on the Company’s consolidated statement of operations or consolidated financial position.
vii) In December 2005, the CICA Emerging Issues Committee issued Abstract No. 159, “Conditional Asset Retirement Obligations” (EIC–159). This EIC determined that a conditional asset retirement obligation should not be recognized until the fair value of the liability can be reasonably estimated. EIC-159 will be effective for interim and annual reporting periods ending after March 31, 2006. The Company does not expect the adoption of this EIC to have a material impact on the Company’s consolidated statement of operations or consolidated financial position.
41
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 3 | GAIN ON ISSUANCE OF SHARES OF SUBSIDIARY AND NON-CONTROLLING INTEREST |
On December 29, 2005, the Company’s formerly wholly owned subsidiary, Nucryst, completed an initial public offering in which 4,500,000 common shares were issued at a price of US$10.00 per share. Net proceeds after commissions and expenses amounted to US$39,095 ($45,502). US$6,850 of the proceeds were used to reduce the term loan owing by Nucryst to the Company and the remaining balance of the term loan of US$39,642 was converted to common shares of Nucryst on January 27, 2006. As a result of these transactions, the Company’s ownership in Nucryst was reduced from 100% to 68.4% as at December 31, 2005 and increased to 75.3% after the conversion of the remaining term loan on January 27, 2006. The conversion of the term loan to common shares, completed in January 2006, was in accordance with the terms of the Nucryst initial public offering and has been accounted for as being effective on December 31, 2005 in accordance with GAAP. The reduction in the Company’s equity interest in Nucryst has been accounted for as a disposition of shares and resulted in a dilution gain for accounting purposes of $30,055 in 2005 and a non-controlling interest of $15,447 as at December 31, 2005. The operating results relating to the non-controlling interest for the year ended December 31, 2005 were nil.
The Company will continue to consolidate the financial statements of Nucryst with the non-controlling interest being reported separately in the consolidated statement of operations and consolidated balance sheet.
| 4 | DISCONTINUED OPERATIONS |
In December 2005, the Company completed the sale of land and building previously included in long-term capital assets available for sale. Prior to the sale, the land and building were leased to the future owner for the period April 1, 2005 to December 15, 2005. Net proceeds from the sale amounted to $7,423 and the gain on sale of $1,923 was included in income from discontinued operations in 2005.
In January 2005, the Company completed a series of transactions whereby two inactive wholly owned subsidiary companies participated in a Plan of Arrangement with two widely held publicly traded companies. This resulted in the reorganization of the participating companies into a single company and the dilution of the Company’s investments to approximately 6.0%. The Company’s pro rata interest in the book value of the recapitalized companies amounted to $2,189. Subsequently, the company created under the Plan of Arrangement was listed on the Toronto Stock Exchange. The inactive subsidiaries were previously carried at nominal value and this transaction resulted in the Company recording a dilution gain of $2,189 in 2005. In February 2005, the Company sold its investment in the publicly traded company discussed above for net proceeds of $11,519 and recorded a gain on sale of $9,330. No income tax provision was recorded in relation to the dilution gain or the gain on sale of investment as these gains were more than offset by the reduction of unrecognized tax loss carry-forwards. As a result of the divestiture, the Company’s non-capital losses, capital losses and research and development tax credits were reduced by approximately $49,500, $7,700 and $600, respectively.
In January 2004, the Company sold substantially all of the assets and liabilities related to the Ambeon business segment, including its wholly owned subsidiary Neomet Limited, for proceeds of $33,359 (net of transaction costs of $911), of which, $2,800 was held in escrow in respect of general representations and warranties relating to the sale transaction. At December 31, 2004, $1,000 of this escrow receivable was reported in accounts receivable while $1,800 was reported as a long-term other receivable. The entire escrow amount was received during 2005 and is included as proceeds on sale of discontinued operations in the consolidated cash flow statement. The net gain on the sale of Ambeon of $5,436 was net of future income tax expense of $1,032. The results of Ambeon’s operations were accounted for on a discontinued basis. The Company determined that the net proceeds would exceed the net book value of the Ambeon business segment as at December 31, 2003 and therefore no adjustment to the carrying value of Ambeon was required. The gain on the sale of Ambeon was reported in 2004 and any costs incurred in subsequent periods related to discontinued operations have been reported in the period in which they occurred.
42
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
4 DISCONTINUED OPERATIONS (CONTINUED)
In 2005, the Company sold the remaining assets and operations related to Ambeon for net proceeds of $873. The gain on sale of this business, amounting to $739 has been included in discontinued operations and the results from operations of this business have been reclassified to discontinued operations resulting in the restatement of the December 31, 2004 and 2003 consolidated statements of operations and cash flow.
Results of discontinued operations:
| Year Ended | Year Ended | Year Ended | ||||||||||
| Revenues | Dec. 31, 2005 | Dec. 31, 2004 | Dec. 31, 2003 | |||||||||
| Ambeon business segment | $ | 162 | $ | 334 | $ | 30,114 | ||||||
| Year Ended | Year Ended | Year Ended | ||||||||||
| Income (expense) | Dec. 31, 2005 | Dec. 31, 2004 | Dec. 31, 2003 | |||||||||
| Dilution gain | $ | 2,189 | $ | — | $ | — | ||||||
| Gain on sale of inactive subsidiaries | 9,330 | — | — | |||||||||
| Gain on sale of capital assets available for sale | 1,923 | — | — | |||||||||
| Gain on sale of Ambeon | 739 | 5,436 | — | |||||||||
| Income (costs) related to discontinued operations | 597 | (1,879 | ) | 2,409 | ||||||||
| Net income from discontinued operations | $ | 14,778 | $ | 3,557 | $ | 2,409 | ||||||
The net income from discontinued operations is after deduction of depreciation and amortization of $Nil for the year ended December 31, 2005 (2004 – $ Nil; 2003 – $1,738). Current income tax expense included in net income from discontinued operations for the year ended December 31, 2005 amounted to $Nil (2004 – $ Nil; 2003 – $27).
Net income per common share from discontinued operations was $0.16 for the year ended December 31, 2005 (2004 – $0.04; 2003 – $0.03).
Amounts included in the consolidated balance sheets relating to discontinued operations are as follows:
| December 31, 2005 | December 31, 2004 | |||||||
| Accounts receivable | $ | — | $ | 54 | ||||
| Inventories | — | 173 | ||||||
| Accounts payable and accrued liabilities | (11 | ) | (1,219 | ) | ||||
| Capital assets available for sale | — | 5,500 | ||||||
43
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
5 INVENTORIES
| December 31, 2005 | December 31, 2004 | |||||||
| Raw materials | $ | 4,571 | $ | 2,378 | ||||
| Materials in process | 2,473 | 923 | ||||||
| Finished product | 614 | 154 | ||||||
| Spare parts and operating materials | 115 | 150 | ||||||
| $ | 7,773 | $ | 3,605 | |||||
6 CAPITAL ASSETS
| Accumulated | Net | |||||||||||
| December 31, 2005 | Cost | Depreciation | Book Value | |||||||||
| Land | $ | 505 | $ | — | $ | 505 | ||||||
| Buildings | 25,790 | 10,060 | 15,730 | |||||||||
| Machinery and equipment | 69,802 | 19,633 | 50,169 | |||||||||
| Construction in progress | 3,811 | — | 3,811 | |||||||||
| Computer hardware and software | 4,345 | 3,448 | 897 | |||||||||
| $ | 104,253 | $ | 33,141 | $ | 71,112 | |||||||
| Accumulated | Net | |||||||||||
| December 31, 2004 | Cost | Depreciation | Book Value | |||||||||
| Land | $ | 505 | $ | — | $ | 505 | ||||||
| Buildings | 15,022 | 8,913 | 6,109 | |||||||||
| Machinery and equipment | 33,724 | 15,954 | 17,770 | |||||||||
| Construction in progress | 22,093 | — | 22,093 | |||||||||
| Computer hardware and software | 3,620 | 3,321 | 299 | |||||||||
| $ | 74,964 | $ | 28,188 | $ | 46,776 | |||||||
The construction of iFire’s pilot manufacturing plant for flat panel displays at its Toronto location was completed in December 2005 at which time depreciation of the plant commenced. Nucryst continues to expand its wound care production facility in Fort Saskatchewan, Alberta. As a result, at December 31, 2005, capital assets include construction in progress in the amount of $3,811 (2004 – $22,093) that is not currently subject to depreciation. Depreciation on capital assets for the year ended December 31, 2005 was $5,855 (2004 – $5,260; 2003 – $4,638).
44
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
7 INTANGIBLE ASSETS
| December 31, 2005 | December 31, 2004 | |||||||
| Patents and trademarks | $ | 8,601 | $ | 7,591 | ||||
| Less accumulated amortization | (3,421 | ) | (1,898 | ) | ||||
| $ | 5,180 | $ | 5,693 | |||||
Amortization of intangible assets was $1,483 for the year ended December 31, 2005 (2004 – $462; 2003 – $363).
8 INCOME TAXES
The following is a reconciliation of income taxes, calculated at the statutory income tax rate, to the income tax provision included in the consolidated statements of operations.
| Year Ended | Year Ended | Year Ended | ||||||||||
| Dec. 31, 2005 | Dec. 31, 2004 | Dec. 31, 2003 | ||||||||||
| Loss from continuing operations before income taxes | $ | (5,329 | ) | $ | (28,613 | ) | $ | (33,021 | ) | |||
| Statutory income tax rate | 33.62 | % | 33.87 | % | 36.74 | % | ||||||
| Expected income tax recovery | (1,792 | ) | (9,691 | ) | (12,132 | ) | ||||||
| Losses and temporary differences | ||||||||||||
| – valuation allowance | 12,023 | 9,691 | 16,830 | |||||||||
| Tax effect of items not subject to tax | ||||||||||||
| – gain on issuance of shares of subsidiary (Note 4) | (10,107 | ) | — | — | ||||||||
| Large corporations and capital taxes | 55 | 121 | 130 | |||||||||
| Income tax expense | $ | 179 | $ | 121 | $ | 4,828 | ||||||
| Classified as: | ||||||||||||
| Current | $ | 179 | $ | 121 | $ | 130 | ||||||
| Future | — | — | 4,698 | |||||||||
| Income tax expense | $ | 179 | $ | 121 | $ | 4,828 | ||||||
45
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
8 INCOME TAXES (CONTINUED)
Income taxes are recognized for future income tax consequences attributed to estimated differences between the financial statement carrying values of existing assets and liabilities and their respective income tax bases.
The net future income tax asset is comprised of:
| December 31, 2005 | December 31, 2004 | |||||||
| Future income tax assets: | ||||||||
| Tax benefit of loss carry-forwards and tax credits | $ | 118,845 | $ | 107,585 | ||||
| Provisions and reserves | 3,359 | 3,174 | ||||||
| Capital, intangible and other assets | 1,935 | 5,223 | ||||||
| Less valuation allowance | (108,122 | ) | (110,537 | ) | ||||
| 16,017 | 5,445 | |||||||
| Future income tax liabilities: | ||||||||
| Capital, intangible and other assets | (15,835 | ) | (5,263 | ) | ||||
| Other | (182 | ) | (182 | ) | ||||
| (16,017 | ) | (5,445 | ) | |||||
| Future income tax assets, net | $ | — | $ | — | ||||
The future income tax asset valuation allowance is in respect of tax loss carry-forwards and tax credits primarily relating to subsidiaries involved in technology development.
The Company has non-capital losses for income tax purposes of approximately $237,341 (2004 – $215,303; 2003 – $192,764) that can be used to offset taxable income, if any, in future periods. These recognized losses have been fully offset by a valuation allowance and expire at various times to the end of 2025, with $22,517 expiring by 2012. The Company also has capital losses of approximately $20,224 (2004 – $27,939; 2003 – $5,865) as well as research and development tax credits of approximately $47,380 (2004 – $38,837; 2003 – $29,419) which will expire at various times to the end of 2015. Cash taxes relating to continuing operations paid during the year amounted to $270 (2004 – $141; 2003 – $155).
9 LONG-TERM DEBT
In January 2004, iFire entered into a Japanese Yen 1,080,000,000 loan agreement with Dai Nippon Printing Co., Ltd. (“DNP”) to finance certain new equipment in iFire’s Toronto pilot facility. The loan was drawn down during 2004 and 2005, carries an average interest rate of 1.64% per annum and is repayable in full in Japanese Yen on June 30, 2006. The loan is secured by the assets financed under the agreement. Interest expense on the loan amounted to $137 for the year ended December 31, 2005 (2004 – $13) and the liability reported at December 31, 2005 includes accrued interest of $137 which is payable at the end of the loan term. As at December 31, 2005, the outstanding loan balance amounted to Yen 1,029,259,143 ($10,175) (2004 – Yen 408,166,000 ($4,782)). In 2005, the Company entered into forward transactions to purchase Japanese Yen 918,901,467 to hedge the foreign currency exposure of the loan. Unrealized losses on these hedges amounting to $1,994 (2004 – $Nil) have been included in foreign exchange in the consolidated statement of operations and accrued in accounts payable and accrued liabilities at December 31, 2005.
In February 2005, a wholly owned subsidiary of the Company issued $6,000 of convertible debentures to private investors. These debentures mature in February 2007, bear interest at 5% per annum, have no recourse to the Company and are convertible into a maximum of 12,000,000 non-voting common shares of the inactive subsidiary. If converted, the Company’s economic interest in the inactive subsidiary would be reduced to approximately 22% and the Company’s consolidated non-capital losses and research and development tax credits would be reduced by approximately $91,000 and $18,000, respectively. These convertible debentures are financial instruments which have both debt and equity components. The value assignable to the conversion option at the date of issue was determined to be immaterial. Interest expense on these debentures during 2005 amounted to $206 and was accrued in accounts payable and accrued liabilities at December 31, 2005.
46
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
10 PROVISION FOR SITE RESTORATION
Changes in the provision are as follows:
| Year Ended | Year Ended | |||||||
| December 31, 2005 | December 31, 2004 | |||||||
| Provision at beginning of year | $ | 6,708 | $ | 7,564 | ||||
| Additional provisions | — | 327 | ||||||
| Expenditures incurred | (100 | ) | (1,640 | ) | ||||
| Costs recovered | 152 | 457 | ||||||
| Provision at end of year | $ | 6,760 | $ | 6,708 | ||||
The provision relates primarily to site restoration associated with soil and groundwater reclamation and remediation, based on periodic independent estimates of these costs as at December 31, 2005.
Potential recoveries of costs resulting from indemnifications provided by previous owners of the Company’s industrial sites have not been recognized in the consolidated financial statements as the amount of recovery cannot be reasonably determined. Any future recoveries will be recorded when received.
11 CAPITAL STOCK
a) Share Capital
The Company’s authorized share capital consists of an unlimited number of common shares, Preferred A shares and Preferred B shares. Changes in the Company’s common shares outstanding during 2005, 2004 and 2003 are as follows:
| 2005 | 2004 | 2003 | ||||||||||||||||||||||
| Stated | Stated | Stated | ||||||||||||||||||||||
| Common Shares | Number | Capital | Number | Capital | Number | Capital | ||||||||||||||||||
| Balance at beginning of year | 92,828,054 | $ | 421,233 | 78,073,386 | $ | 373,230 | 78,032,787 | $ | 373,128 | |||||||||||||||
| Employee share purchase plan | 72,595 | 233 | 28,785 | 135 | 40,599 | 102 | ||||||||||||||||||
| Share offering | — | — | 14,705,883 | 47,801 | — | — | ||||||||||||||||||
| Stock options exercised | — | — | 20,000 | 67 | — | — | ||||||||||||||||||
| Balance at end of year | 92,900,649 | $ | 421,466 | 92,828,054 | $ | 421,233 | 78,073,386 | $ | 373,230 | |||||||||||||||
During 2005, 72,595 (2004 – 28,785; 2003 – 40,599) common shares were issued with respect to the Company’s employee share purchase plan at a weighted average price of $2.36 (2004 – $3.24; 2003 – $2.50). The value of these rights to acquire common shares of $62 (2004 – $42; 2003 – $Nil) was reclassified from contributed surplus to share capital.
No options of the Company were exercised in 2005 or 2003. In 2004, 20,000 options were exercised at an average price of $2.59, and $15 relating to the fair value of these options was reclassified from contributed surplus to share capital.
47
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
11 CAPITAL STOCK (CONTINUED)
On August 4, 2004, the Company completed a $50,000 share offering. A total of 14,705,883 new common shares were issued at a price of $3.40. Four members of the Company’s Board of Directors purchased 4,852,942 shares under the offering. Net proceeds, after fees and expenses, amounted to $47,801.
b) Stock-based compensation plans
Employee and Director stock option plan– The Company maintains an Employee and Director Stock Option Plan under which the Company may grant options for 10,750,000 shares of common stock of the Company at an exercise price equal to the market price of the Company’s stock at the date of grant. Options awarded are exercisable for a period of 10 years and vest as to one third of the grant on each of the first, second and third anniversaries after the date of the grant. The Company maintains a Directors and Officers Share Purchase Program under the provisions of the Stock Option Plan. Under the Share Purchase Program, Directors and designated officers may be granted one option for each common share purchased to a cumulative 50,000 options. Options, equal to the net purchases of common shares by the optionee during the calendar year, vest at the end of the calendar year in which the purchases were made. Any options issued under this program which do not vest at the end of the year are cancelled.
A summary of the status of the Company’s stock option plans as at December 31, 2005, 2004 and 2003, and changes during the years ending on those dates is presented below:
| 2005 | 2004 | 2003 | ||||||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||||||
| Average | Average | Average | ||||||||||||||||||||||
| Exercise | Exercise | Exercise | ||||||||||||||||||||||
| Stock Options | Number | Price | Number | Price | Number | Price | ||||||||||||||||||
| Outstanding at beginning of year | 4,659,600 | $ | 6.94 | 5,833,400 | $ | 8.71 | 5,665,599 | $ | 9.41 | |||||||||||||||
| Granted | 390,000 | $ | 3.66 | 497,500 | $ | 3.61 | 427,800 | $ | 1.87 | |||||||||||||||
| Exercised | — | $ | — | (20,000 | ) | $ | 2.59 | — | $ | — | ||||||||||||||
| Cancelled | — | $ | — | (1,651,300 | ) | $ | 12.25 | (259,999 | ) | $ | 12.62 | |||||||||||||
| Outstanding at end of year | 5,049,600 | $ | 6.68 | 4,659,600 | $ | 6.94 | 5,833,400 | $ | 8.71 | |||||||||||||||
The following table summarizes information about stock options outstanding as at December 31, 2005:
| Options Outstanding | Options Exercisable | |||||||||||||||||||||||||||||
| Number | Weighted Average | Weighted | Number | Weighted | ||||||||||||||||||||||||||
| Range of | Outstanding | Remaining | Average | Exercisable | Average | |||||||||||||||||||||||||
| Exercise Prices | Dec. 31, 2005 | Contractual Life | Exercise Price | Dec. 31, 2005 | Exercise Price | |||||||||||||||||||||||||
| (in years) | ||||||||||||||||||||||||||||||
| $ | 1 | – | $ | 3 | 451,500 | 7.1 | $ | 1.85 | 326,500 | $ | 1.87 | |||||||||||||||||||
| $ | 3 | – | $ | 6 | 2,240,700 | 4.4 | $ | 4.17 | 1,640,700 | $ | 4.35 | |||||||||||||||||||
| $ | 6 | – | $ | 9 | 1,311,403 | 3.1 | $ | 7.17 | 1,311,403 | $ | 7.17 | |||||||||||||||||||
| $ | 9 | – | $ | 12 | 431,000 | 4.4 | $ | 10.45 | 431,000 | $ | 10.45 | |||||||||||||||||||
| $ | 12 | – | $ | 16 | 614,997 | 4.4 | $ | 15.72 | 614,997 | $ | 15.72 | |||||||||||||||||||
| $ | 1 | – | $ | 16 | 5,049,600 | 4.3 | $ | 6.68 | 4,324,600 | $ | 7.24 | |||||||||||||||||||
48
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
11 CAPITAL STOCK (CONTINUED)
Effective January 1, 2004, the Company adopted revisions to Section 3870 of the CICA Handbook with respect to the accounting and disclosure of stock-based compensation requiring that the fair value of stock options be recorded in the financial statements. For the year ended December 31, 2005, corporate costs, research and development costs, and selling, general and administrative expenses include additional compensation expense relating to stock options totaling $905 (2004 – $1,210; 2003 – $942) with an offsetting increase to contributed surplus.
The fair value of each option grant by the Company and its subsidiaries is estimated using the Black-Scholes option pricing model assuming no dividends are paid on common shares, a risk-free interest rate of 4.33% (2004 – 4.80%; 2003 – 5.05%), an average life of 7.0 years and a volatility of 57.72% (2004 – 58.47%; 2003 – 59.89%). The amounts computed according to the Black-Scholes pricing model may not be indicative of the actual values realized upon the exercise of these options by the holders.
Deferred share unit plan– The Company maintains DSUs for the non-executive Directors of the Company. DSUs are issued at the market value of the Company’s shares at the date of grant, vest upon death or retirement of the Director and are payable in cash. Directors may elect to receive additional DSUs in lieu of fees, which are issued at 90% of the market value of the Company’s shares at the date of grant. Compensation expense relating to DSUs during the year amounted to $888 (2004 – $172; 2003 – $572) and as at December 31, 2005, a liability of $1,842 (2004 – $954) has been accrued with respect to issued DSUs.
Restricted share unit plan– In 2004, the Company instituted a Restricted Share Unit Plan for the Executive Officers of the Company. These units vest over three years and are payable when fully vested in a combination of common shares and cash at the weighted average trading price at the date of exercise. The liability with respect to these units is accrued over the vesting period. Compensation expense with respect to RSUs during the year amounted to $524 (2004 – $139) and the corresponding accrued liability as at December 31, 2005 was $663 (2004 – $139).
Employee share purchase plan– Under the Employee Share Purchase Plan, employees are entitled to subscribe for common shares of the Company, to a maximum value of five percent of their annual compensation. Payment for these shares is made over a 24-month period at a price per share equal to the lesser of the market value at the offering date and the market value at the end of the purchase period. The market value at the offering date of July 18, 2005 was $4.02 (July 19, 2004 – $3.55; July 21, 2003 – $2.36). At December 31, 2005 there were outstanding purchase arrangements with employees having an aggregate value of $474 (2004 – $363; 2003 – $389). During the year ended December 31, 2005, a total of 72,595 shares were issued under this Plan at an average price of $2.36 (2004 – 28,785 shares at $3.24; 2003 – 40,599 shares at $2.50).
Employee bonus appreciation units– Employees of the Company, other than non-executive officers, are granted Bonus Appreciation Units (“BAUs”). BAUs are issued at the market value of the Company’s shares at date of grant, vest over three years and are payable when vested in cash at the amount by which the market value of the Company’s shares exceed the share price at the date of grant. Net changes in the value of BAUs is recognized as compensation expense over the vesting period. As at December 31, 2005, the accrued liability relating to BAUs amounted to $178 (2004 – $65).
Subsidiary stock-based compensation plans– The Company also maintains equity incentive plans for certain employees of its technology subsidiaries, Nucryst and iFire, under which stock options have been granted representing 5.0% to 5.5% of the outstanding shares of the respective subsidiaries. Effective January 1, 2004, these plans are being accounted for under the fair value method with the related compensation expense being recorded in the consolidated statement of operations. Subsidiary stock options generally vest evenly over a three-year period and expire after 10 years from the date of grant. The exercise prices of stock options granted are not less than the fair value of the subsidiary’s stock at the time of the grant. No shares were issued by the Company in 2005, 2004 or 2003 with respect to these plans.
49
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
11 CAPITAL STOCK (CONTINUED)
SARs have been granted to employees of certain technology subsidiaries. SARs vest over time and may be settled with cash, shares of the subsidiary and, in the case of iFire SARs, shares of the Company at the Company’s option. The exercise prices of SARs granted are not less than the fair value of the subsidiary’s stock at the time of the grant. The total potential liability upon exercise of these SARs is capped at $1,462.
Net changes in the value of SARs, measured as the amount by which quoted market prices or fair value exceeds the exercise price at the measurement date, is recognized as compensation expense over the SARs’ vesting period. As at December 31, 2005, the accrued liability relating to SARs amounted to $105. The liability as at December 31, 2004 and 2003 was $Nil as the estimated market value of the subsidiaries’ SARs was less than their exercise price.
12 CUMULATIVE TRANSLATION ADJUSTMENT
The cumulative translation adjustment represents the net unrealized foreign currency translation loss on the Company’s net investment in self-sustaining foreign operations.
| 2005 | 2004 | |||||||
| Cumulative translation adjustment beginning of year | $ | — | $ | — | ||||
| Unrealized loss resulting from change in accounting policy (Note 2c) | (503 | ) | — | |||||
| Cumulative translation adjustment end of year | $ | (503 | ) | $ | — | |||
| U.S. Dollar exchange rate, end of year | $ | 1.1659 | $ | 1.2036 | ||||
The change in cumulative translation adjustment is primarily the change in the rate of exchange between the Canadian dollar and the U.S. dollar and the effect this change has on the assets and liabilities of the Company’s self-sustaining foreign subsidiary.
13 RESEARCH AND DEVELOPMENT FUNDING AGREEMENTS
In 2001, iFire entered into a research and development contribution agreement with the Government of Canada through Technology Partnerships Canada (“TPC”). Under this agreement, TPC agreed to contribute 28.371% of eligible research and development costs and related capital expenditures incurred by iFire to a maximum of $30,000.
Under the terms of the agreement, iFire will pay a royalty to TPC equal to 1.065% of eventual commercial sales of the technology under development. In addition, TPC received warrants to purchase common shares of iFire exercisable for an additional consideration of $6,283. The warrants expire on December 31, 2009 and, as at December 31, 2005, represented approximately 0.5% of the current outstanding common shares of iFire.
Contributions were recorded as a reduction of the cost of the applicable capital asset or credited to the statement of operations of iFire as determined by the nature of the expenditure being funded. Contribution claims totaling $5,997 were recorded in 2003. Of this amount, $5,204 was credited to research and development expense and $793 was credited to capital assets. As at December 31, 2003, the Company had received the maximum $30,000 eligible funding.
On January 1, 2003, the Company entered into a series of agreements with a third party whereby the third party agreed to reimburse iFire for a portion of the costs related to research and development projects of mutual interest to iFire and the third party. These agreements provide for reimbursement totaling US$3,800 to be paid quarterly between January 2003 and October 2006. These reimbursements are recorded as a reduction to research and development expenditures in the consolidated financial statements and amounted to $1,152 (2004 – $1,251; 2003 – $1,364).
50
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
14 GUARANTEES
The Company has agreements to indemnify its Officers and Directors for certain events or occurrences while the Officer or Director is or was serving at the Company’s request in such capacity. The maximum potential amount of future payments is unlimited. However, the Company maintains Director and Officer liability insurance coverage that limits its exposure and enables the Company to recover a portion of any future amounts paid.
In addition, the Company has provided indemnifications to third parties with respect to future site restoration costs to be incurred on properties previously owned by the Company. These estimated costs have been included in the provision for site restoration (Note 10).
15 COMMITMENTS AND CONTINGENCIES
Commitments
The Company is committed to capital expenditures of $2,508 (2004 – $17,987; 2003 – $1,315) and to future annual payments under operating leases for facility and office space and equipment as follows:
| 2006 | 2007 | 2008 | 2009 | 2010 | ||||||||||||
| $763 | $ | 550 | $ | 536 | $ | 188 | $ | 14 | ||||||||
Contingencies
The Company and its subsidiaries are party to legal proceedings in the ordinary course of their businesses. Management does not expect the outcome of any of these proceedings to have a material effect on the Company’s financial position or operating results.
16 FINANCIAL INSTRUMENTS
Fair value of financial instruments
The carrying value of the Company’s interest in financial instruments approximates their fair value. The estimated fair value approximates the amount for which the financial instruments could currently be exchanged in an arm’s length transaction between willing parties who are under no compulsion to act. Certain financial instruments lack an available trading market and, therefore, fair value amounts should not be interpreted as being necessarily realizable in an immediate settlement of the instrument.
Interest rate risk
Interest rate risk reflects the sensitivity of the Company’s financial results and condition to movements in interest rates. For 2005, a 1% decrease in interest rates would have reduced earnings before income taxes by $684 (2004 – $937; 2003 – $783). Conversely, a 1% increase in interest rates would have increased earnings before taxes by a similar amount.
Foreign currency risk
The Company is exposed to currency risks as a result of its export to foreign jurisdictions of goods produced in Canada. These risks are partially covered by purchases of goods and services in the foreign currency and by forward exchange contracts (Note 9).
Credit risk
The Company’s financial instruments that are exposed to concentrations of credit risk consist primarily of cash and cash equivalents, short-term investments, accounts receivable, and forward contracts. Cash and short-term investments are placed with major financial institutions or invested in the commercial paper of large organizations. The Company has a concentration of credit risk through its reliance on one customer for Nucryst’s wound care products. This risk is limited due to the long-term contractual relationship with this customer.
51
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
17 EARNINGS PER SHARE
In calculating earnings per share under the treasury stock method, the numerator remains unchanged from the basic earnings per share calculation as the assumed exercise of the Company’s stock options does not result in an adjustment to income. The reconciliation of the denominator in calculating diluted earnings per share is as follows:
| Year Ended | Year Ended | Year Ended | ||||||||||
| Dec. 31, 2005 | Dec. 31, 2004 | Dec. 31, 2003 | ||||||||||
| Weighted average number of common shares | ||||||||||||
| outstanding – basic earnings per share | 92,852,120 | 84,093,827 | 78,044,689 | |||||||||
| Effect of dilutive securities | ||||||||||||
| – Options | 267,078 | — | — | |||||||||
| – Employee share purchase plan | 1,738 | — | — | |||||||||
| Weighted average number of common shares outstanding | ||||||||||||
| – diluted earnings per share | 93,120,936 | 84,093,827 | 78,044,689 | |||||||||
The impact of all dilutive securities on earnings per share was anti-dilutive for the years ended December 31, 2004 and 2003.
Options to purchase 5,049,600 common shares were outstanding at December 31, 2005 (2004 – 4,659,600; 2003 – 5,833,400). Of these options outstanding in 2005, 3,979,100 (2004 – 3,939,100; 2003 – 5,424,700) were excluded in the calculation of diluted earnings (loss) per share because the exercise price of the option was greater than the weighted average market value of the common shares in the year.
52
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
18 SEGMENTED INFORMATION
The Company has two operating segments, which have been determined based on the nature of the products produced: Nucryst Pharmaceuticals and iFire Technology. Discontinued operations are disclosed separately (Note 4).
The Nucryst Pharmaceuticals segment researches, develops, manufactures and commercializes innovative medical products that fight infection and inflammation, and pharmaceutical products based on noble metal nanocrystalline technology. The iFire Technology segment has developed a proprietary flat-panel full color solid state display technology with applications in the large screen television market.
The accounting policies of the reportable segments are the same as those described in Note 2. Included in other non-cash assets of $6,914 at December 31, 2005 (2004 – $8,770; 2003 – $7,800) are assets that cannot be allocated to a particular segment. Non-cash assets excludes short-term investments and assets available for sale.
| Operating Income | Depreciation and | Capital | Non-cash Assets | |||||||||||||||||
| Year Ended December 31, 2005 | Revenue | (Loss) | Amortization | Expenditures | Dec. 31, 2005 | |||||||||||||||
| Nucryst Pharmaceuticals | $ | 28,560 | $ | 1,590 | $ | 1,549 | $ | 4,461 | $ | 27,315 | ||||||||||
| iFire Technology | — | (31,844 | ) | 5,072 | 24,096 | 58,605 | ||||||||||||||
| Other | — | 54 | 717 | 2,197 | 6,914 | |||||||||||||||
| $ | 28,560 | $ | (30,200 | ) | $ | 7,338 | $ | 30,754 | $ | 92,834 | ||||||||||
| Operating Income | Depreciation and | Capital | Non-cash Assets | |||||||||||||||||
| Year Ended December 31, 2004 | Revenue | (Loss) | Amortization | Expenditures | Dec. 31, 2004 | |||||||||||||||
| Nucryst Pharmaceuticals | $ | 31,907 | $ | 6,079 | $ | 1,128 | $ | 3,163 | $ | 17,476 | ||||||||||
| iFire Technology | — | (27,884 | ) | 3,539 | 18,649 | 39,378 | ||||||||||||||
| Other | — | (1,189 | ) | 1,055 | 1,620 | 8,770 | ||||||||||||||
| $ | 31,907 | $ | (22,994 | ) | $ | 5,722 | $ | 23,432 | $ | 65,624 | ||||||||||
| Operating Income | Depreciation and | Capital | Non-cash Assets | |||||||||||||||||
| Year Ended December 31, 2003 | Revenue | (Loss) | Amortization | Expenditures | Dec. 31, 2003 | |||||||||||||||
| Nucryst Pharmaceuticals | $ | 16,212 | $ | (2,014 | ) | $ | 892 | $ | 1,827 | $ | 12,556 | |||||||||
| iFire Technology | 911 | (20,054 | ) | 2,693 | 8,424 | 20,544 | ||||||||||||||
| Other | — | (1,333 | ) | 1,416 | 132 | 7,800 | ||||||||||||||
| $ | 17,123 | $ | (23,401 | ) | $ | 5,001 | $ | 10,383 | $ | 40,900 | ||||||||||
All of Nucryst’s revenues are earned through a long-term exclusive licensing agreement with an international wound care company which expires in 2026. Nucryst exports manufactured products to this company for their resale in international markets.
53
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES |
These consolidated financial statements have been prepared in accordance with Canadian GAAP which conform in all material respects with those used in the United States (“U.S. GAAP”), except as set forth below:
a) Derivative Instruments and Hedging Activities
Under U.S. GAAP, pursuant to Statement of Financial Accounting Standards (“SFAS”) 133 “Accounting for Derivative Instruments and Hedging Activities” (“SFAS 133”), companies are required to record derivatives on the balance sheet as assets or liabilities measured at fair value. For those derivatives representing effective cash flow hedges of risks and exposures, unrealized gains or losses resulting from changes in the fair values are presented as a component of other comprehensive income (“OCI”) as defined in SFAS 130 “Reporting Comprehensive Income”(“SFAS 130”). To the extent certain derivatives do not represent effective hedges, unrealized gains or losses are included in the income statement for U.S. GAAP purposes. Derivatives embedded within hybrid instruments are generally not separately accounted for except for those related to equity-linked deposit contracts which are not applicable to the Company.
Hedges against price risk exposure on contracted nickel sales and raw materials purchases in the discontinued Ambeon segment were considered effective cash-flow hedges under SFAS 133. Management regularly measured the effectiveness of these hedges, with unrealized gains and losses reported in OCI for U.S. GAAP purposes. From time to time, the Company reduces its exposure to foreign currency and interest rate fluctuations by entering into forward transactions. These transactions are not considered hedging activities under SFAS 133 and unrealized gains and losses are included in the income statement. As at December 31, 2005 the Company has no adjustments to OCI with respect to cash-flow hedges (December 31, 2004 – recovery $982; December 31, 2003 – charge $907).
b) Gain on issuance of shares by subsidiary
In accordance with Canadian GAAP, the Company is required to account for gains and losses on the issuance of shares by a subsidiary as a component of income. Under U.S. GAAP, the effect of such dilution gains may be recorded as income except in circumstances where subsequent capital transactions are contemplated that raise concerns about the likelihood of realizing the gain. In addition, realization of the gain is not assured where the subsidiary is a newly formed, non-operating entity; a research and development start-up; or development-stage company. It is the Company’s policy to record dilution gains as a component of income for U.S. GAAP purposes. However, for a transaction in 2001 involving iFire, which at that time was an early stage research and development company, the dilution gain was required to be recorded in equity as an increase in paid-in capital rather than as income. Subsequent dilution gains have met the criteria for income statement recognition under both Canadian GAAP and U.S. GAAP.
c) Pension
Under U.S. GAAP, the Company would have recorded an additional minimum liability for under-funded plans representing the excess of the accumulated benefit obligation over the pension plan assets, less the pension liability already recognized and the net unamortized prior service cost. Under SFAS 130 “Reporting Other Comprehensive Income” the increase in liabilities is charged directly to other comprehensive income.
The Company wound up the defined benefit plan in 2003 and there will be no reconciling differences in future years.
d) Research and Development
Under U.S. GAAP, the cost of purchased research and development should be charged to income, in the period incurred, when no alternative uses exist for the purchased research and development. As the patents and intellectual property purchased are restricted to use in inorganic electroluminescent displays, the costs have been fully expensed under U.S. GAAP.
Under Canadian GAAP, such costs are capitalized and amortized over their estimated useful lives.
54
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
e) Stock-based Compensation
U.S. GAAP SFAS 123, “Accounting for Stock-based Compensation” (“SFAS 123”), encourages that stock-based compensation be accounted for based on a fair value methodology. As permitted by the Statement, the Company and its subsidiaries have elected to continue measuring compensation costs as the excess, if any, of the quoted market value of the stock at the date of grant over the exercise price.
As the exercise price of options granted by the Company and its subsidiaries is not less than the market value at the date of grant, no compensation expense is recognized.
Under Canadian GAAP, effective January 1, 2004, the Company adopted revisions to Section 3870 of the CICA Handbook which requires that the fair value of stock options be recorded in the financial statements (see Note 2n). These new rules applied to options issued on or after January 1, 2002. As a result, the Company has recorded stock-based compensation expense under Canadian GAAP for the year ended December 31, 2005 of $905 (2004 – $1,210; 2003 – $942).
f) Foreign Currency Translation Adjustments
Under U.S. GAAP, the Company records unrecognized gains and losses arising from translating net investments in foreign operations into Canadian dollars in accumulated other comprehensive loss as a separate component of shareholders’ equity.
Under Canadian GAAP, such unrealized gains and losses are included in shareholders’ equity as a cumulative translation adjustment.
The effect of the above differences on the Company’s financial statements is set out below:
| December 31, 2005 | December 31, 2004 | |||||||||||||||
| Canadian | U. S. | Canadian | U. S. | |||||||||||||
| Consolidated Balance Sheets | GAAP | GAAP | GAAP | GAAP | ||||||||||||
| Intangible assets (d) | $ | 5,180 | $ | 3,180 | $ | 5,693 | $ | 2,693 | ||||||||
| Capital stock (e) | 421,466 | 421,347 | 421,233 | 421,176 | ||||||||||||
| Contributed Surplus (e) | 3,968 | 403 | 3,125 | 403 | ||||||||||||
| Deficit (b) (d) (e) | (267,291 | ) | (276,116 | ) | (276,561 | ) | (287,291 | ) | ||||||||
| Additional paid-in-capital (b) | — | 10,509 | — | 10,509 | ||||||||||||
| Accumulated other comprehensive loss (f) | — | (503 | ) | — | — | |||||||||||
| Cumulative translation adjustment (f) | (503 | ) | — | — | — | |||||||||||
| Shareholders’ Equity | December 31, 2005 | December 31, 2004 | ||||||
| Canadian GAAP | $ | 157,640 | $ | 147,797 | ||||
| Adjustments | ||||||||
| Research and development (d) | (2,000 | ) | (3,000 | ) | ||||
| U.S. GAAP | $ | 155,640 | $ | 144,797 | ||||
55
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
| Year Ended December 31 | ||||||||||||||
| 2005 | 2004 | 2003 | ||||||||||||
| Consolidated Statements of Operations | (Restated - Note 4) | (Restated - Note 4) | ||||||||||||
| Loss from continuing operations | �� | |||||||||||||
| Canadian GAAP | $ | (5,508 | ) | $ | (28,734 | ) | $ | (37,849 | ) | |||||
| Research and development (d) | 1,000 | (3,000 | ) | — | ||||||||||
| Stock-based compensation (e) | 905 | 1,210 | 942 | |||||||||||
| Loss from continuing operations | ||||||||||||||
| U.S. GAAP | (3,603 | ) | (30,524 | ) | (36,907 | ) | ||||||||
| Income from discontinued operations | ||||||||||||||
| Canadian GAAP and U.S. GAAP | 14,778 | 3,557 | 2,409 | |||||||||||
| Net income (loss) applicable to common shareholders | ||||||||||||||
| U.S. GAAP | 11,175 | (26,967 | ) | (34,498 | ) | |||||||||
| Other comprehensive income (loss) | ||||||||||||||
| Pension (c) | — | — | 665 | |||||||||||
| Cash flow hedges (a) | — | 982 | (907 | ) | ||||||||||
| Foreign currency translation adjustments (f) | (503 | ) | — | — | ||||||||||
| Net comprehensive income (loss) | ||||||||||||||
| U.S. GAAP | $ | 10,672 | $ | (25,985 | ) | $ | (34,740 | ) | ||||||
| Year Ended December 31 | ||||||||||||
| Income (Loss) Per Common Share | 2005 | 2004 | 2003 | |||||||||
| Canadian GAAP | ||||||||||||
| Loss from continuing operations | $ | (0.06 | ) | $ | (0.34 | ) | $ | (0.48 | ) | |||
| Net income (loss) | $ | 0.10 | $ | (0.30 | ) | $ | (0.45 | ) | ||||
| U.S. GAAP | ||||||||||||
| Loss from continuing operations | $ | (0.04 | ) | $ | (0.36 | ) | $ | (0.47 | ) | |||
| Net income (loss) | $ | 0.12 | $ | (0.32 | ) | $ | (0.44 | ) | ||||
| Year Ended | Year Ended | Year Ended | ||||||||||||||||||||||
| December 31, 2005 | December 31, 2004 | December 31, 2003 | ||||||||||||||||||||||
| Canadian | U. S. | Canadian | U. S. | Canadian | U. S. | |||||||||||||||||||
| Consolidated Cash Flow Statements | GAAP | GAAP | GAAP | GAAP | GAAP | GAAP | ||||||||||||||||||
| Loss from continuing operations | $ | (5,508 | ) | $ | (3,603 | ) | $ | (28,734 | ) | $ | (30,524 | ) | $ | (37,849 | ) | $ | (36,907 | ) | ||||||
| Items not affecting cash: | ||||||||||||||||||||||||
| Stock-based compensation (e) | $ | 905 | $ | — | $ | 1,210 | $ | — | $ | 942 | $ | — | ||||||||||||
| Depreciation and amortization (d) | $ | 7,338 | $ | 6,338 | $ | 5,722 | $ | 5,722 | $ | 5,001 | $ | 5,001 | ||||||||||||
| Write down of capital assets and intangible assets (d) | $ | 570 | $ | 570 | $ | 22 | $ | 3,022 | $ | 723 | $ | 723 | ||||||||||||
| Changes in items not affecting cash (d) (e) | $ | (26,816 | ) | $ | (28,721 | ) | $ | 7,714 | $ | 9,504 | $ | 12,978 | $ | 12,036 | ||||||||||
56
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
Stock-based compensation
U.S. GAAP, SFAS 123 “Accounting for Stock-based Compensation” (“SFAS 123”) encourages that stock-based compensation be accounted for based on a fair value methodology. As permitted by the Statement, the Company and its subsidiaries have elected to continue measuring compensation costs as the excess, if any, of the quoted market value of the stock at the date of the grant over the exercise price. As the exercise price of options granted by the Company and its subsidiaries is not less than the market value at the date of grant, no compensation expense is recognized.
If compensation costs for the Company’s and its subsidiaries’ stock option plans had been determined based on the fair value methodology over the vesting period consistent with SFAS 123, including options issued by the Company and its subsidiaries prior to January 1, 2002, the Company’s net income (loss) per share would have been reduced to the pro-forma amounts indicated below:
| Year Ended December 31 | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
| Net income (loss) applicable to common shareholders | ||||||||||||
| U.S. GAAP | $ | 11,175 | $ | (26,967 | ) | $ | (34,498 | ) | ||||
| Total stock-based employee compensation expense determined under fair-value based method for awards net of tax effects | (905 | ) | (1,040 | ) | (4,518 | ) | ||||||
| Pro forma net income (loss) applicable to common shareholders – U.S. GAAP | $ | 10,270 | $ | (28,007 | ) | $ | (39,016 | ) | ||||
| Loss per common share – U.S. GAAP | ||||||||||||
| Loss from continuing operations | (0.04 | ) | (0.36 | ) | (0.47 | ) | ||||||
| Net income (loss) | 0.12 | (0.32 | ) | (0.44 | ) | |||||||
| Pro forma | ||||||||||||
| Loss from continuing operations | (0.05 | ) | (0.38 | ) | (0.53 | ) | ||||||
| Net income (loss) | 0.11 | (0.33 | ) | (0.50 | ) | |||||||
The fair value of each option grant by the Company and its subsidiaries was estimated using the Black-Scholes options pricing model assuming no dividends are to be paid on common shares, a risk free interest rate of 4.33% (2004 – 4.80%; 2003 – 5.05%), an average life of 7.0 years and a volatility for the shares of the companies of 57.72% (2004 – 58.47%; 2003 – 59.89%). The amounts computed according to the Black-Scholes pricing model may not be indicative of the actual values realized upon the exercise of these options by the holders. The weighted average fair value of options granted by the Company and its subsidiaries in 2005 was $0.75 (2004 – $1.13; 2003 – $0.66).
Recently Adopted and Pending Accounting Pronouncements
FSP No. 46(R)-5
In March 2005, the Financial Accounting Standards Board (“FASB”) issued FASB Staff Position (“FSP”) No. 46(R)-5, “Implicit Variable Interests under FASB Interpretation No. (“FIN”) 46 (revised December 2003), Consolidation of Variable Interest Entities” (“FSP FIN 46(R)-5”). FSP FIN 46(R)-5 provides guidance for a reporting enterprise on whether it holds an implicit variable interest in Variable Interest Entities (“VIEs”) or potential VIEs when specific conditions exist. This FSP is effective in the first period beginning after March 3, 2005 in accordance with the transition provisions of FIN 46 (Revised 2003), “Consolidation of Variable Interest Entities — an Interpretation of Accounting Research Bulletin No. 51” (“FIN 46(R)”). The adoption of FSP FIN 46(R)-5 had no impact on the Company’s results of operations or financial position.
57
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
FIN 47
In March 2005, the FASB issued FASB Interpretation No. 47, “Accounting for Conditional Asset Retirement Obligations” (“FIN 47”), which clarifies that the term “conditional asset retirement obligation” as used in FASB statement No. 143, “Accounting for Asset Retirement Obligations”, refers to a legal obligation to perform an asset retirement activity in which the timing and/or method of settlement are conditional on a future event that may or may not be within the control of the entity. An entity is required to recognize a liability for the fair value of a conditional asset retirement obligation if the fair value of the liability can be reasonably estimated. FIN 47 is effective for fiscal years ending after December 15, 2005. The adoption of FIN 47 did not have a material impact on the Company’s financial position or results of operations.
SFAS 123(R)
In December 2004, the FASB issued SFAS No. 123 (revised 2004) (“SFAS 123(R)”), “Share-Based Payment”, which is a revision of SFAS No. 123, “Accounting for Stock-based Compensation”. SFAS 123(R) requires all share-based payments to employees, including grants of employee stock options, to be recognized in the financial statements based on their fair values and does not allow the previously permitted pro-forma disclosure as an alternative to financial statement recognition. Liability classified awards are remeasured to fair value at each balance sheet date until the award is settled. SFAS 123(R) supersedes Accounting Principles Board (“APB”) Opinion No. 25 “Accounting for Stock Issued to Employees” and related interpretations and amends SFAS No. 95 “Statement of Cash Flows”. SFAS 123(R) is scheduled to be effective beginning fiscal 2006 for the Company. On August 31, 2005, the FASB issued FSP SFAS 123(R)-1 to defer the requirement that a freestanding financial instrument originally subject to SFAS 123(R) becomes subject to the recognition and measurement requirements of other applicable generally accepted accounting principles when the rights conveyed by the instrument to the holder are no longer dependent on the holder being an employee of the entity. On October 18, 2005, the FASB issued FSP SFAS 123(R)2 to provide further guidance on the application of grant date as defined in SFAS 123(R). On November 10, 2005, the FASB issued FSP SFAS No. 123(R)-3 to provide a practical transition election related to accounting for the tax effect of share-based payment awards to employees. The adoption of SFAS 123(R) and the related FSPs will not have a material impact on the Company’s financial position or results of operations.
SFAS 151
In November 2004, the FASB issued SFAS No. 151, “Inventory Costs, an amendment of ARB No. 43, Chapter 4”, which amends Chapter 4 of ARB No. 43 that deals with inventory pricing. The Statement clarifies the accounting for abnormal amounts of idle facility expenses, freight, handling costs, and spoilage. Under previous guidance, paragraph 5 of ARB No. 43, chapter 4, items such as idle facility expense, excessive spoilage, double freight, and rehandling costs might be considered to be so abnormal, under certain circumstances, as to require treatment as current period charges. This Statement eliminates the criterion of “so abnormal” and requires that those items be recognized as current period charges. Also, this Statement requires that allocation of fixed production overheads to the costs of conversion be based on the normal capacity of the production facilities. This Statement is effective for inventory costs incurred during fiscal years beginning after June 15, 2005, although earlier application is permitted for fiscal years beginning after the date of issuance of this Statement. Retroactive application is not permitted. The Company does not expect the impact of this Statement to be material.
58
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
SFAS 153
In December 2004, the FASB issued SFAS No. 153, “Exchanges of Non-monetary Assets”, an amendment of APB No. 29. This Statement amends Opinion 29 to eliminate the exception for non-monetary exchanges of similar productive assets and replaces it with a general exception for exchanges of non-monetary assets that do not have commercial substance. The Statement specifies that a non-monetary exchange has commercial substance if the future cash flows of the entity are expected to change significantly as a result of the exchange. This Statement is effective for non-monetary asset exchanges occurring in fiscal periods beginning after June 15, 2005. Earlier application is permitted for non-monetary asset exchanges occurring in fiscal periods beginning after the date this Statement is issued. Retroactive application is not permitted. The Company does not expect the impact of this Statement to be material.
SFAS 154
In May 2005, the FASB issued SFAS No. 154, “Accounting Changes and Error Corrections” (“SFAS 154”), which replaces APB No. 20, “Accounting Changes” and SFAS No. 3, “Reporting Accounting Changes in Interim Financial Statements – An Amendment of APB Opinion No. 28”. SFAS 154 provides guidance on the accounting for and reporting of accounting changes and error corrections. It establishes retrospective application, on the latest practicable date, as the required method for reporting a change in accounting principle and the reporting of a correction of an error. SFAS 154 is effective for accounting changes and corrections of errors made in fiscal years beginning after December 15, 2005. Management believes that the adoption of this statement will not have a material effect on the Company’s financial position or results of operations.
EITF 03–1
In November 2005, the FASB issued FSP SFAS 115-1 and SFAS 124-1 “The Meaning of Other-Than-Temporary Impairment and Its Application to Certain Investments”, which nullified 2004 guidance issued by the Emerging Issues Task Force (“EITF”) on determining whether an impairment is other than temporary, and effectively reverted back to previous guidance in this area. The FSP generally encompasses guidance for determining when an investment is impaired, how to measure the impairment loss, and what disclosures should be made regarding impaired securities. This FSP, effective for the first quarter of 2006, is not expected to have a material impact on the Company’s results of operations or financial position.
EITF 03–13
The EITF issued EITF Abstract 03-13 (“EITF 03-13”) to provide guidance on applying SFAS No. 144, “Determining Whether to Report Discontinued Operations” (“SFAS 144”). SFAS 144 discusses when an entity should disclose a “component” as discontinued operations. Under SFAS 144, a component should be disclosed as discontinued operations when continuing cash flows are eliminated and when there is no significant continuing involvement with the component. EITF 03-13 provides additional guidance on factors to consider in evaluating what constitutes continuing cash flows and continuing significant influence. This Statement is effective for fiscal periods beginning after December 15, 2004. The adoption of EITF 03-13 did not have a material impact on the Company’s financial position or results of operations.
EITF 04–13
In September 2005, the EITF reached consensus on Issue No. 04-13, “Accounting for Purchases and Sales of Inventory with the same Counterparty” (“EITF 04-13”). EITF 04-13 provides guidance on the purchase and sale of inventory to another entity that operates in the same line of business. The purchase and sale transactions may be pursuant to a single contractual arrangement or separate contractual arrangements and the inventory purchased or sold may be in the form of raw materials, work-in-process, or finished goods. EITF 04-13 applies to new arrangements entered into, or modifications or renewals of existing arrangements in reporting periods beginning after March 15, 2006. The impact of the adoption of EITF 04-13 on the Company’s consolidated results of operations and financial condition will depend on the nature of future arrangements entered into, or modifications or renewals of existing arrangements by the Company.
59
The Westaim Corporation Annual Report 2005
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
| 19 | RECONCILIATION TO ACCOUNTING PRINCIPLES GENERALLY ACCEPTED IN THE UNITED STATES (CONTINUED) |
FASB Exposure Draft “Fair Value Measurements”
In June 2004, the FASB issued an exposure draft of a proposed Statement, “Fair Value Measurements” to provide guidance on how to measure the fair value of financial and non-financial assets and liabilities when required by other authoritative accounting pronouncements. The proposed statement attempts to address concerns about the ability to develop reliable estimates of fair value and inconsistencies in fair value guidance provided by current GAAP, by creating a framework that clarifies the fair value objective and its application in GAAP. In addition, the proposal expands disclosures required about the use of fair value to re-measure assets and liabilities. The standard would be effective for financial statements issued for fiscal years beginning after November 15, 2006. The Company does not expect the impact of this proposed standard to be material.
FASB Exposure Draft “Business Combinations”
In June 2005, the FASB published an exposure draft containing proposals to change the accounting for business combinations. The proposed standards would replace the existing requirements of the FASB’s Statement No. 141, “Business Combinations”. The proposals would result in fewer exceptions to the principle of measuring assets acquired and liabilities assumed in a business combination at fair value. Additionally, the proposals would result in payments to third parties for consulting, legal, audit, and similar services associated with an acquisition being recognized generally as expenses when incurred rather than capitalized as part of the business combination. The FASB also published an Exposure Draft that proposes, among other changes, that non-controlling interests be classified as equity within the consolidated financials statements. The FASB’s proposed standard would replace Accounting Research Bulletin No. 51, “Consolidated Financial Statements”. The Company does not expect the impact of this proposed standard to be material.
FASB Exposure Draft “Accounting for Uncertain Tax Positions”
On July 14, 2005, the FASB published an exposure draft entitled “Accounting for Uncertain Tax Positions – an interpretation of FASB Statement No. 109”. The proposed interpretation is intended to reduce the significant diversity in practice associated with recognition and measurement of income taxes by establishing consistent criteria for evaluating uncertain tax positions.
The proposed interpretation would be effective as of the end of the first fiscal year beginning after December 15, 2006. Earlier application would be encouraged. Only tax positions meeting the probable recognition threshold at that date would be recognized. The transition adjustment resulting from application of this interpretation would be considered a change in accounting policy with the cumulative effect of the change treated as an offsetting adjustment to the opening balance of retained earnings in the period of transition. The Company does not expect the impact of this proposed interpretation, if it becomes effective in its current form, to be material.
60
The Westaim Corporation Annual Report 2005
SHAREHOLDER INFORMATION
| DIRECTORS | ||
Ian W. Delaney 4 | Non-executive Chairman of the Board, The Westaim Corporation | |
Neil Carragher 2, 4 | Chairman of The Corporate Partnership Ltd. | |
Dr. Roger G.H. Downer 2, 4 | President and Vice Chancellor of the University of Limerick | |
Barry M. Heck | President and Chief Executive Officer, The Westaim Corporation | |
Frank W. King 1, 4 | President of Metropolitan Investment Corporation | |
Edward M. Lakusta 3, 4 | Private Business and Energy Consultant | |
Daniel P. Owen 1, 3, 4 | Chairman of Molin Holdings Limited | |
Guy J. Turcotte 2, 3, 4 | President and Chief Executive Officer of Western Oil Sands Inc. | |
Bruce V. Walter 1, 4 | Vice-Chairman of Dynatec Corporation | |
| Numbers indicate | ||
| the individual’s committee membership: | 1 Member of the Audit Committee | |
| 2 Member of the Human Resources and Compensation Committee | ||
| 3 Member of the Environmental, Health and Safety Committee | ||
| 4 Member of the Corporate Governance Committee |
THE WESTAIM CORPORATION ANNUAL GENERAL MEETING
| Wednesday May 3, 2006 10:00 a.m. | The Jane Mallet Theatre, The St. Lawrence Centre, 27 Front Street East, Toronto, ON | |
| INVESTOR RELATIONS | ||||
Anthony B. Johnston | Senior Vice President | The Westaim Corporation | ||
| Tel: (403) 234-3103 | ||||
| Fax: (403) 237-6565 | ||||
| E-mail:info@westaim.com | ||||
| STOCK INFORMATION | Traded on TheToronto Stock Exchange under the symbol WED | |||
| Traded onNASDAQ under the symbol WEDX | ||||
| Shares issued and outstanding at December 31, 2005 were 92,900,649 | ||||
| TRANSFER AGENT | Computershare Trust Company of Canada | |||
| 600, 530 – 8th Avenue SW | ||||
| Calgary, Alberta, Canada T2P 3S8 | ||||
| Tel: 1-888-267-6555 | ||||
| E-Mail:service@computershare.com | ||||
61
The Westaim Corporation Annual Report 2005
CORPORATE INFORMATION
| CORPORATE INFORMATION | ||
| EXECUTIVE OFFICERS | ||
Barry M. Heck | President and Chief Executive Officer | |
G.A. (Drew) Fitch | Senior Vice President and Chief Financial Officer | |
Anthony B. Johnston | Senior Vice President | |
Brian D. Heck | Vice President, General Counsel and Corporate Secretary | |
| OFFICES | ||
Executive Office | 144 – 4th Avenue S.W., Suite 1010, Calgary, Alberta, Canada T2P 3N4 | |
| Tel: (403) 237-7272 | ||
| Fax: (403) 237-6565 | ||
| E-mail: info@westaim.com | ||
| www.westaim.com | ||
Corporate Office | 10102 – 114 Street, Fort Saskatchewan, Alberta, Canada T8L 3W4 | |
| Tel: (780) 992-5300 | ||
| Fax: (780) 992-5301 | ||
iFire Technology Corp. | 9 City View Drive, Toronto, Ontario, Canada M9W 5A5 | |
| Tel: (416) 883-6800 | ||
| Fax: (416) 883-6801 | ||
| E-mail: info@ifire.com | ||
| www.ifire.com | ||
NUCRYST | ||
Pharmaceuticals Corp. | 50 Audubon Road Suite B, Wakefield, Massachusetts, United States 01880 Tel: (781) 224-1444 Fax: (781) 246-6002 E-mail: info@nucryst.com www.nucryst.com | |
62
FORWARD-LOOKING STATEMENTS
This Report contains forward-looking statements including the outlook for Nucryst and iFire. Forward-looking statements involve significant risks, uncertainties and assumptions, and Westaim’s actual results could differ materially from those anticipated by these forward-looking statements for various reasons generally beyond Westaim’s control. The words “may”, “will”, “can”, “could”, “should”, “outlook”, “believe”, “anticipate”, “estimate”, “expect”, “intend”, “plan”, “opportunity”, “strategy”, “develop”, “collaborating”, “project”, “potential”, “looks”, “aims”, “targeted”, “set”, “offers”, “extend”, “capability”, “pursue” and words and expressions of similar import are intended to identify forward-looking statements. Such forward-looking statements include but are not limited to statements concerning expected progress in the Company’s technology businesses; the delivery of value for shareholders of the Company; the growth of the large screen TV market; expected cost advantages of iFireTM displays and the sustainability of those advantages; the cost advantages and other advantages of iFire’s manufacturing process compared to other technologies; the expected performance characteristics of iFire’s displays; the uses and purposes of the iFire pilot plant and the information expected to be obtained from that plant; the anticipated timing and capacity of volume production of iFireTM displays; iFire’s intention to be a significant force in the flat panel television market; targeted thickness, size and weight of TDEL modules; expected domination of the television market by CRT-based TVs until a flat panel technology can compete with CRTs on cost; iFire’s plans to target a particular segment of the flat panel television market; anticipated depreciation expense in 2006 for iFire; the outlook for 2006 iFire expenditures; iFire’s growth and commercialization strategies, including with manufacturing partners; expectations for iFire manufacturing revenues and financing requirements; potential for TDEL panels to introduce a new standard in flat panel television performance and design and to deliver viewing experience at an affordable price; Nucryst collaborations with Smith & Nephew on new products; conclusions to be drawn from Nucryst product studies; the extension of Nucryst’s technology to the treatment of dermatological and gastrointestinal conditions; Nucryst’s beliefs in the ability of products based on its technology to combat infection longer than the products of its major competitors and to offer a broader spectrum of antimicrobial activity than many topically applied antibiotics; Nucryst’s beliefs in the role the mechanisms of action of its nanocrystalline silver may play in regard to anti-inflammation; projections for the growth of the advanced wound care market; Nucryst’s beliefs in regard to the results of its study of the effects of its topical NPI 32101 product on patients with atopic dermatitis; Nucryst’s beliefs regarding systemic exposure to silver of patients treated with NPI 32101, and the safety of a 2% cream of that product; estimates and expectations regarding the start and completion of clinical trials by Nucryst; the expected timing of the results from Nucryst’s clinical studies of its products; the anticipated timing of the submission by Nucryst of New Drug Applications to the U.S. Food and Drug Administration; timing of operation of new Nucryst production facilities; the cost of Nucryst capital projects; the need for further Nucryst facilities expansion to meet projected sales growth; projected cost of Nucryst capital projects in 2006; the outlook for Nucryst year over year growth in licensing and manufacturing revenues; expected timing of further milestone payments to Nucryst; opportunities and demand for Nucryst’s present and future products; Nucryst’s sales, receipt of milestone payments, revenue and profitability; increase in Nucryst research and development expenditures in 2006 ; expected Nucryst net operating losses in 2006; Nucryst’s expectations for the use of the net proceeds of its initial public offering; Nucryst’s growth strategy; the affect of sales activity or achievement of operating expense and capital budget targets on financial performance; the sufficiency of Westaim’s cash and short term investments to fund its 2006 business plan; Westaim’s strategy regarding the development of its technology investments and the commercialization of those investments; estimates regarding site restoration costs; and computations regarding the valuation of options. These statements are based on current expectations that are subject to risks and uncertainties, and the Company can give no assurance that these expectations are correct. Forward-looking statements are not guarantees of future performance, they involve significant risks, uncertainties and assumptions, and our actual results could differ materially from those anticipated by these forward-looking statements for various reasons generally beyond our control, including but not limited to: (i) unexpected obstacles in developing iFire or Nucryst technology, manufacturing processes and new applications; (ii) unforeseen complexities and delays associated with completing facility expansions, and with achieving timing targets for pilot manufacturing and the production of product quality displays and panels and the commercial introduction and sale of iFire products; (iii) patent and technical hurdles which might inhibit or delay the ability of iFire or Nucryst to develop or commercialize technologies or products; (iv) delays in receiving regulatory approvals, including from the FDA; (v) the degree to which Smith & Nephew succeeds in selling ActicoatTM products; (vi) general economic, market, financing, regulatory and industry developments and conditions in the industries that the Company serves, which among other things might affect the demand for electronic materials and pharmaceutical products or the ability to raise new capital or affect potential partner ability to contribute financially; (vii) the activities of our competitors and technological developments that diminish the attractiveness of our products; (viii) general industry and market conditions and growth rates; and (ix) the risks described above under “Risks and Uncertainties”. The foregoing list should not be construed as exhaustive. Other than as required by applicable law, the Company disclaims any intention or obligations publicly to review, revise or update any forward-looking statement, whether as a result of new information, future developments or otherwise. All forward-looking statements are expressly qualified in their entirety by this cautionary statement.

| w w w.we st aim.com |
| The Westaim Corporation |
| 144 – 4th Avenue S.W., Suite 1010, Calgary, Alberta Canada T2P 3N4 |
| info@westaim.com |
 | The Westaim Corporation |
Notice of Annual Meeting of Shareholders
to be held May 3, 2006
to be held May 3, 2006
NOTICE IS HEREBY GIVEN that the annual meeting of shareholders (the “Meeting”) of The Westaim Corporation (the “Corporation”) will be held at the Jane Mallet Theatre, St. Lawrence Centre, 27 Front Street East, Toronto, Ontario, on Wednesday, May 3, 2006 at 10:00 a.m. Eastern daylight time, to:
| (a) | receive and consider the Financial Statements of the Corporation for the financial year ended December 31, 2005 and the Auditors’ Report to the shareholders; | ||
| (b) | elect directors; | ||
| (c) | appoint auditors and authorize the board of directors to fix the auditors’ remuneration; and | ||
| (d) | transact such other business as may properly come before the Meeting or any adjournment(s) thereof. |
Any adjournment(s) of the Meeting will be held at a time and place to be specified at the Meeting.
The Financial Statements for the financial year ended December 31, 2005, together with the Auditors’ Report thereon, form part of the Annual Report of the Corporation, a copy of which accompanies this notice.
Shareholders registered on the books of the Corporation at the close of business on March 15, 2006 are entitled to notice of and to vote at the Meeting.
Details of all matters proposed to be put before the Meeting are set forth in the accompanying Information Circular.
Dated at Calgary, Alberta, this 8th day of March, 2006.
By Order of the Board,
By Order of the Board,
Brian D. Heck
Vice President, General Counsel
and Corporate Secretary
If you are unable to be present at the Meeting,PLEASE COMPLETE AND RETURN THE ACCOMPANYING FORM OF PROXYin the envelope provided for that purpose.
The Westaim Corporation
Information Circular
MATTERS REQUIRING SHAREHOLDER APPROVAL
This proxy information circular (the “Information Circular”) is furnished in connection with the solicitation of proxies by and on behalf of the management (the “Management”) of The Westaim Corporation (the “Corporation”) for use at the annual meeting of shareholders of the Corporation (the “Meeting”) to be held at the Jane Mallet Theatre, St. Lawrence Centre, 27 Front Street East, Toronto, Ontario, on Wednesday, May 3, 2006 at 10:00 a.m. Eastern daylight time, for the purposes set out in the Notice of Meeting (the “Notice”) accompanying this Information Circular and any adjournment(s) thereof.
Election of Directors
The Articles of the Corporation require a minimum of three and a maximum of fifteen directors. The number of directors to be elected at an annual meeting of shareholders shall be the number of directors then in office unless the directors or shareholders by simple majority otherwise determine from time to time. The board of directors of the Corporation (the “Board”) has resolved to set the number of directors to be elected at the Meeting at eight.
Management of the Corporation proposes to nominate at the Meeting and the persons named in the accompanying form of proxy intend to vote at the Meeting, unless otherwise directed, for the election of the persons whose names are set forth in the table below to serve until the next meeting of shareholders of the Corporation (the “Shareholders”) at which the election of directors is considered, or until their successors are elected or appointed. Management of the Corporation does not contemplate that any of the nominees will be unable to serve as a director.
The following table and the notes thereto state the names of all persons proposed to be nominated for election as directors at the Meeting, all other positions and offices with the Corporation now held by them, their principal occupation or employment, the period during which they have been directors of the Corporation, and their shareholdings, including the number of voting securities of the Corporation beneficially owned, directly or indirectly, or over which control or direction is exercised by each of them. The information contained herein as to securities beneficially owned, directly or indirectly, or over which control or direction is exercised is based upon information furnished to the Corporation by the respective nominees.
| Nominee for Election | Common Shares Owned, | |||||||||||||||
| as Director | Age | Director Since | Controlled or Directed(1) | Deferred Share Units(2) | ||||||||||||
Neil Carragher | 67 | 1996 | 53,907 | 59,790 | ||||||||||||
| Toronto, Ontario, Canada | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
| - Corporate Governance Committee | Mr. Carragher is the Chairman of The Corporate Partnership Ltd. (a management consulting group). | |||||||||||||||
| - Human Resources and Compensation Committee (Chair) | ||||||||||||||||
INFORMATION CIRCULAR 1
| Nominee for Election | Common Shares Owned, | |||||||||||||||
| as Director | Age | Director Since | Controlled or Directed(1) | Deferred Share Units(2) | ||||||||||||
Ian W. Delaney | 62 | 1996 | 3,670,169 | n/a | ||||||||||||
| Toronto, Ontario, Canada Chairman of the Board | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
| - Corporate Governance Committee (Chair) | Mr. Delaney is the Chairman of Sherritt International Corporation (a nickel/cobalt mining and refining, oil and gas exploration and production, electricity generation and coal mining company). | |||||||||||||||
Roger G.H. Downer | 63 | 2004 | 20,000 | 27,710 | ||||||||||||
| County Clare, Ireland | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
| - Corporate Governance Committee | Dr. Downer is the President and Vice-Chancellor of the University of Limerick, Ireland. | |||||||||||||||
| - Human Resources and Compensation Committee | ||||||||||||||||
Barry M. Heck(3) | 44 | 2003 | 86,939 | n/a | ||||||||||||
| Calgary, Alberta, Canada | ||||||||||||||||
| Non-Independent | ||||||||||||||||
| Mr. Barry M. Heck is the President and Chief Executive Officer of the Corporation. From January 1997 until his appointment as President and Chief Executive Officer on January 15, 2003, Mr. Heck was a Senior Vice President of the Corporation. | ||||||||||||||||
Frank W. King | 69 | 1996 | 332,064 | 60,370 | ||||||||||||
| Calgary, Alberta, Canada | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
- Audit Committee (Chair) - Corporate Governance Committee | Mr. King is the President of Metropolitan Investment Corporation (a capital investment and management services company). | |||||||||||||||
Daniel P. Owen | 70 | 1996 | 1,600,000 | 52,538 | ||||||||||||
| Toronto, Ontario, Canada | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
| - Audit Committee | Mr. Owen is the Chairman of Molin Holdings Limited (an investment management company). | |||||||||||||||
| - Corporate Governance Committee | ||||||||||||||||
| - Environmental, Health and Safety Committee (Chair) | ||||||||||||||||
2 INFORMATION CIRCULAR
| Nominee for Election | Common Shares Owned, | |||||||||||||||
| as Director | Age | Director Since | Controlled or Directed(1) | Deferred Share Units(2) | ||||||||||||
Guy J. Turcotte | 54 | 1998 | 1,463,882 | 73,094 | ||||||||||||
| Calgary, Alberta, Canada | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
- Corporate Governance Committee - Environmental, Health and Safety Committee - Human Resources and Compensation Committee | Mr. Turcotte is the Chairman of Western Oil Sands Inc. (an oil sands production company). Prior to becoming Chairman of Western Oil Sands Inc. in April 2005, Mr. Turcotte was Chief Executive Officer of Western Oil Sands Inc. from July 1999 to April 2005 and President from January 2002 to April 2005. He has held the position of Chairman of Fort Chicago Energy Partners L.P. since December 1997 and was Chief Executive Officer of that company from December 1997 to December 2002. | |||||||||||||||
Bruce V. Walter | 48 | 1997 | 32,800 | 103,974 | ||||||||||||
| Toronto, Ontario, Canada | ||||||||||||||||
| Independent | ||||||||||||||||
| Member of: | ||||||||||||||||
- Audit Committee - Corporate Governance Committee | Mr. Walter is the President and Chief Executive Officer of Dynatec Corporation (a mining, drilling and metallurgical technologies company). Prior to becoming President and Chief Executive Officer of Dynatec Corporation in January 2005, Mr. Walter was Vice-Chairman of Dynatec Corporation from March 2002 to December 2004. He was Chief Executive Officer of Four Mile Investments Inc. from August 1993 and Managing Director, BMO Nesbitt Burns Inc. from February 1999 to November 2001. | |||||||||||||||
| 1. | The information as to the common shares in the capital of the Corporation (“Common Shares”) owned, controlled or directed has been furnished by each of the nominees as of March 8, 2006. | |
| 2. | The information as to deferred share units (“DSUs”) is provided as at March 8, 2006. For more detailed information relating to the deferred share units held by the directors, see “Statement of Executive Compensation – Remuneration of Directors”. | |
| 3. | Barry M. Heck owned 201,928 restricted share units (“RSUs”) as at March 8, 2006. In addition to these RSUs, on February 15, 2006, he was granted the right to earn up to 57,739 RSUs during 2006, subject to the achievement of certain objectives. The actual number of RSUs earned for 2006 will be established in early 2007 based on the achievement of these objectives. |
IF ANY OF THE ABOVE NOMINEES IS FOR ANY REASON UNAVAILABLE TO SERVE AS A DIRECTOR, PROXIES IN FAVOUR OF MANAGEMENT WILL BE VOTED FOR ANOTHER NOMINEE IN THEIR DISCRETION UNLESS THE SHAREHOLDER HAS SPECIFIED IN THE PROXY THAT HIS OR HER SHARES ARE TO BE WITHHELD FROM VOTING IN THE ELECTION OF DIRECTORS.
INFORMATION CIRCULAR 3
Committee Memberships and Record of Attendance at Meetings
The following table summarizes the meetings of the Board and its committees held for the twelve month period ending December 31, 2005 and the attendance of individual directors of the Corporation at such meetings.
| Number of | ||||
| Type of Meeting Held | Meetings | |||
| Board | 8 | |||
| Audit Committee | 5 | |||
| Human Resources and Compensation Committee | 5 | |||
| Environmental, Health & Safety Committee | 1 | |||
| Corporate Governance Committee | 3 | |||
| Committee | Board | |||||||||
| Meetings | Meetings | |||||||||
| Director | Committee Memberships1 | Attended | Attended | |||||||
| N. Carragher | Corporate Governance | 3 of 3 | 8 of 8 | |||||||
| Human Resources and Compensation (Chair) | 5 of 5 | |||||||||
| I.W. Delaney | Corporate Governance (Chair) | 3 of 3 | 8 of 8 | |||||||
| R.G.H. Downer | Corporate Governance | 3 of 3 | 8 of 8 | |||||||
| Human Resources and Compensation | 5 of 5 | |||||||||
| B.M. Heck | 8 of 8 | |||||||||
| F.W. King | Audit (Chair) | 5 of 5 | 8 of 8 | |||||||
| Corporate Governance | 3 of 3 | |||||||||
| E.M. Lakusta | Audit | 2 of 2 | 6 of 8 | |||||||
| Corporate Governance | 2 of 3 | |||||||||
| Environmental, Health & Safety | 1 of 1 | |||||||||
| D.P. Owen | Audit | 3 of 3 | 8 of 8 | |||||||
| Corporate Governance | 3 of 3 | |||||||||
| Environmental, Health & Safety (Chair) | 1 of 1 | |||||||||
| G.J. Turcotte | Corporate Governance | 2 of 3 | 8 of 8 | |||||||
| Environmental, Health & Safety | 1 of 1 | |||||||||
| Human Resources and Compensation | 4 of 5 | |||||||||
| B.V. Walter | Audit | 5 of 5 | 8 of 8 | |||||||
| Corporate Governance | 3 of 3 | |||||||||
| Total Attendance Rate | 95 | % | 97 | % | ||||||
1. Except for its Corporate Governance Committee, which consists of all of the independent directors of the Corporation, the Board periodically rotates its committee memberships. As a result, on May 4, 2005, the composition of the committees of the Board changed. The committees of the Board (other than the Corporate Governance Committee) were as follows from January 1 to May 4, 2005: Audit — F.W. King, E.M. Lakusta and B.V. Walter; Compensation — N. Carragher, D.P. Owen and G.J. Turcotte; Environmental, Health & Safety — E.M. Lakusta, D.P. Owen and G.J. Turcotte. The committees of the Board were as follows from May 4, 2005 to December 31, 2005: Audit — F.W. King, D.P. Owen and B.V. Walter; Human Resources and Compensation — N. Carragher, R.G.H. Downer and G.J. Turcotte; Environmental, Health & Safety — E.M. Lakusta, D.P. Owen and G.J. Turcotte.
4 INFORMATION CIRCULAR
Appointment of Auditors and Authorization of Board to Fix Auditors’ Remuneration
Unless otherwise directed, the persons named in the enclosed form of proxy intend to vote for the re-appointment of Deloitte & Touche LLP, Chartered Accountants, as auditors of the Corporation to hold office until the next annual meeting of Shareholders, and to authorize the Board to fix the auditors’ remuneration. Deloitte & Touche LLP were first appointed auditors of the Corporation on May 7, 1996.
GENERAL STATUTORY INFORMATION
Solicitation of Proxies
Solicitation of proxies for the Meeting will be primarily by mail, the cost of which will be borne by the Corporation. Proxies may also be solicited personally by employees of the Corporation at nominal cost. In some instances, the Corporation has distributed copies of the Notice, the Information Circular, form of proxy and the annual report (collectively, the “Documents”) to clearing agencies, securities dealers, banks and trust companies, or their nominees (collectively “Intermediaries”, and each an “Intermediary”) for onward distribution to Shareholders whose Common Shares are held by or in the custody of those Intermediaries (“Non-registered Shareholders”). The Intermediaries are required to forward the Documents to Non-registered Shareholders.
Solicitation of proxies from Non-registered Shareholders will be carried out by Intermediaries, or by the Corporation if the names and addresses of Non-registered Shareholders are provided by the Intermediaries. The cost of the solicitation will be borne by the Corporation.
Non-registered Shareholders who have received the Documents from their Intermediary should follow the directions of their Intermediary with respect to the procedure to be followed. Generally, Non-registered Shareholders will either:
(a) be provided with a form of proxy executed by the Intermediary but otherwise uncompleted. The Non-registered Shareholder may complete the proxy and return it directly to the Corporation’s transfer agent; or
(b) be provided with a request for voting instructions. The Intermediary is required to send the Corporation an executed form of proxy completed in accordance with any voting instructions received by the Intermediary.
If you are a Non-registered Shareholder, and the Corporation or its agent has sent these materials directly to you, your name and address and information about your holdings of securities have been obtained in accordance with applicable securities regulatory requirements from your Intermediary.
By choosing to send the Documents to you directly, the Corporation (and not your Intermediary) has assumed responsibility for (i) delivering the Documents to you, and (ii) executing your proper voting instructions. Please return your voting instructions as specified in the request for voting instructions.
Appointment of Proxy
The persons named in the enclosed form of proxy are directors and/or officers of the Corporation.A SHAREHOLDER DESIRING TO APPOINT SOME OTHER PERSON TO REPRESENT HIM OR HER AT THE MEETING MAY DO SOeither by inserting the name of such person in the space provided in the form of proxy or by completing another proper form of proxy and, in either case, delivering the completed proxy to the attention of the Vice President, General Counsel and Corporate Secretary of the Corporation c/o Computershare Trust Company of Canada, 9th Floor, 100 University Avenue, Toronto, Ontario, M5J 2Y1, no later than 10:00 a.m. Eastern daylight time on May 1,2006, provided, however, that the chairman of the Meeting may, in his sole discretion, accept proxies delivered to him up to the time when any vote is taken at the Meeting or any adjournment thereof.
Revocation of Proxy
A Shareholder who has given a proxy pursuant to this solicitation may revoke it at any time up to and including the last business day preceding the day of the Meeting or any adjournments thereof at which the proxy is to be used, by an instrument in writing executed by the Shareholder or by his or her attorney authorized in writing, to the attention of the Vice President, General Counsel and Corporate Secretary of the Corporation and either delivered to the offices of the Corporation at 10102-114 Street, Fort Saskatchewan, Alberta, T8L 3W4 or faxed to (780) 992-5301, or by delivering written notice of such revocation to the chairman of the Meeting prior to the commencement of the Meeting on the day of the Meeting or any adjournment thereof, or in any other manner permitted by law.
INFORMATION CIRCULAR 5
Voting of Proxies and Discretion Thereof
Common Shares represented by properly executed proxies in favour of persons designated in the printed portion of the enclosed form of proxy WILL, UNLESS OTHERWISE INDICATED, BE VOTED FOR THE ELECTION OF DIRECTORS, FOR THE APPOINTMENT OF AUDITORS, AND FOR THE AUTHORIZATION OF THE BOARD TO FIX AUDITORS’ REMUNERATION AS STATED UNDER THOSE HEADINGS IN THE INFORMATION CIRCULAR. The enclosed form of proxy confers discretionary authority on the persons named therein with respect to amendments or variations to matters identified in the Notice or other matters which may properly come before the Meeting. At the time of printing this Information Circular, Management of the Corporation knows of no such amendments, variations or other matters to come before the Meeting. However, if other matters do properly come before the Meeting, it is the intention of the persons named in the enclosed form of proxy to vote such proxy according to their best judgement.
Voting Shares and Principal Holders Thereof
As of March 8, 2006 the Corporation had 92,900,649 issued and outstanding Common Shares. Each Shareholder is entitled to one vote on all matters to come before the Meeting for each Common Share shown as registered in his or her name on the list of Shareholders which is available for inspection during usual business hours at Computershare Trust Company of Canada, 600, 530-8th Avenue S.W., Calgary, Alberta, T2P 3S8 and at the Meeting. The list of Shareholders is as of March 15, 2006, the record date fixed for determining Shareholders entitled to notice of the Meeting. If a person has acquired ownership of Common Shares since that date, he or she may establish such ownership and demand, not later than 10 days before the Meeting, that his or her name be included in the list of Shareholders.
To the knowledge of the directors and officers of the Corporation no person owns beneficially, or exercises control or direction over, Common Shares carrying more than 10% of the voting rights attached to all Common Shares entitled to be voted at the Meeting.
There are no cumulative or similar voting rights attached to the Common Shares.
SECURITY BASED COMPENSATION ARRANGEMENTS
Equity Compensation Plan Information to December 31, 2005
| Number of securities to | Weighted-average | Number of securities | ||||||||||
| Equity compensation | be issued upon exercise | exercise price of | remaining available for | |||||||||
| plans approved by | of outstanding options, | outstanding options, | future issuance under | |||||||||
| securityholders | warrants and rights | warrants and rights | equity compensation plans | |||||||||
Option Plan1 | 5,049,600 | $ | 6.67 | 3,000,973 | 2 | |||||||
| RSU Plan | 178,465 | N/A | 3,000,973 | 2 | ||||||||
| Purchase Plan | 124,249 | 3 | $ | 3.82 | 4 | 186,636 | ||||||
| 1. | Includes securities to be issued under both the Option Plan and the D&O Program (both as defined below). | |
| 2. | The number of securities remaining available for future issuance under the Option Plan (including the D&O Program) and the RSU Plan (as defined below) constitutes a single reserve of 3,000,973 Common Shares shared by the two plans. | |
| 3. | This number is subject to increase based on the weighted average trading price of the Common Shares at the last business day of the purchase period applicable to the particular rights granted under the Purchase Plan (as defined below), or to decrease if participants in the Purchase Plan cancel elections to purchase Common Shares under the Purchase Plan. See below under Employee Share Purchase Plan, and see note 4 immediately below. |
6 INFORMATION CIRCULAR
| 4. | The weighted average purchase price for the Purchase Plan is subject to reduction if the Market Value (as defined below) on the date of purchase is less than the Market Value on the offering date. A more detailed explanation of the terms of the Purchase Plan is provided below under Employee Share Purchase Plan. |
1996 Directors and Officers Stock Option Plan
The eligible participants under the 1996 Directors and Officers Stock Option Plan (the “Option Plan”) include certain directors and employees of the Corporation and its subsidiaries.
There are a total of 10,750,000 Common Shares reserved for issuance pursuant to options to purchase Common Shares (the “Options”) and Stock Appreciation Rights (“SARs”) issuable under the Option Plan, or in settlement of RSUs, which represent approximately 11.57% of the currently outstanding Common Shares.
As of March 8, 2006, there were 5,324,600 Options granted and outstanding under the Option Plan which represents approximately 5.7% of the currently outstanding Common Shares.
Under the terms of the Option Plan, the maximum percentage of Common Shares under the Option Plan which are available to insiders of the Corporation within a one year period shall not exceed approximately 11.57%, being the 10,750,000 Common Shares designated for issuance under the Option Plan as noted above.
The maximum number of Common Shares any one person or company (including insiders of the Corporation) is entitled to receive under the Option Plan must not exceed 5% of the issued and outstanding Common Shares of the Corporation.
The exercise price of Options granted under the Option Plan is determined as the closing price of the Common Shares on the Toronto Stock Exchange (the “TSX”) for the trading day immediately preceding the date on which the granting of the Options is approved by the Board (the “Market Price”).
The formula for calculating market appreciation of SARs is the difference between (i) the Market Price of the Common Shares covered by such Option or portion thereof when transferred and surrendered, and (ii) the aggregate exercise price of the Common Shares subject to such Option or portion thereof. The Corporation may grant SARs in connection and in conjunction with the grant of any Option to purchase Common Shares under the Option Plan.
The period during which an Option may be exercised shall not extend beyond 10 years from the date of the grant of the Option. The Human Resources and Compensation committee of the Board (the “Compensation Committee”) may, however, provide that Options granted under the Option Plan be exercisable in whole or in part only after specified periods designated by the Compensation Committee.
In the case of a participant who is an employee (whether or not the participant is also a director):
(a) if the employment of a participant is terminated for just cause before the expiry date of all of such participant’s Options, then the Options granted to such participant shall thereupon cease and terminate and be of no further force or effect whatsoever; and
(b) if the employment of a participant is terminated for reasons other than just cause or the death or disability of the participant or, in the case of an employee of a subsidiary, if the subsidiary ceases to be a subsidiary of the Corporation before the expiry date of all of such participant’s Options, then the Options shall remain in force and shall be exercisable as provided by their terms, for a period of 90 days after the date of termination or cessation, or such other period as may be specified in such circumstances by the terms of the particular Option or by the Board. At the end of such period, all of the participant’s unexercised Options shall terminate and be of no further force or effect whatsoever.
In the case of a participant who is a director but not an employee, if the participant ceases to be a director of the Corporation or, in the case of a director of a subsidiary, if the subsidiary ceases to be a subsidiary of the Corporation before the expiry date of all such participant’s Options, then the Options shall remain in force and shall be exercisable as provided by their terms, for a period of 90 days after the date of such cessation, or such other period as may be specified in such circumstances by the terms of the Option or by the Board. At the end of such period all of the participant’s unexercised Options shall terminate and be of no further force or effect whatsoever.
In the event of the death or disability of a participant, any Option previously granted to the participant and not otherwise exercisable at the time of death or disability shall immediately become fully exercisable. The personal legal representative (or, in the case of disability, the participant if he or she is competent to do so) may, within 180 days of the date of death or disability or such other period as may be specified in such circumstances by the terms of the particular Option or by the Board, or the expiry of the term of the Option of such participant (whichever is earlier), exercise all Options of such participant. At the end of such 180 day period or as specified in the terms applicable to the Option, whichever is earlier, the Option shall forthwith terminate and be of no further force or effect whatsoever.
Options granted to participants are not transferable and are exercisable only by the participant.
INFORMATION CIRCULAR 7
The Option Plan, as approved by the shareholders of the Corporation, provides that, subject to applicable regulatory approval, the Board may at any time discontinue or amend the plan, provided however that except as expressly permitted by the plan, no amendment shall alter or impair any Option previously granted to a participant. The rules of the TSX impose general restrictions on the nature of amendments to such plans which may be effected without shareholder approval. The Corporation will seek guidance from the TSX in respect of any proposed amendment as to whether regulatory or shareholder approval of such amendment is required.
No financial assistance is provided by the Corporation to participants to facilitate the purchase of Common Shares under the Option Plan.
Directors and Officers Share Purchase Program
In 1999, the Corporation established the Directors and Officers Share Purchase Program (the “D&O Program”). Under the D&O Program, directors and designated officers are granted an Option to purchase one Common Share of the Corporation for each Common Share purchased, to a cumulative maximum of 50,000 Options. Options equal to the net purchases of Common Shares by the optionee during a calendar year vest at the end of that calendar year. Any Options which do not vest at year end are cancelled.
The D&O Program was established to encourage directors and officers to purchase and hold Common Shares of the Corporation. Options granted under the D&O Program are issued in accordance with the provisions of the Option Plan, are granted at an exercise price not less than the closing price of the Common Shares on the TSX for the trading day immediately preceding the date of grant and are exercisable for a period of 10 years from the date of the grant. All Named Executive Officers as defined in the Summary Compensation Table of the Information Circular have been designated participants under the D&O Program.
No financial assistance is provided by the Corporation to participants to facilitate the purchase of Common Shares under the D&O Program.
Employee Share Purchase Plan
The eligible participants under the Employee Share Purchase Plan (the “Purchase Plan”) include all full-time permanent employees of the Corporation and its subsidiaries who are employed more than twenty hours a week and who are paid either a periodic salary or hourly wages, including without limitation any such person who is also an officer or director of the Corporation or any of its subsidiaries.
There are a total of 750,000 Common Shares issued and issuable under the Purchase Plan which represents approximately 0.8% of the currently outstanding Common Shares. As of March 8, 2006, there were 439,115 Common Shares issued under the Purchase Plan which represent approximately 0.47% of the currently outstanding Common Shares. There are additional Common Shares reserved for issuance under the Purchase Plan, but not yet issued, and if these shares, or any of them, are issued then there will be additional dilution. See the table Equity Compensation Plan Information to December 31, 2005, above, under Purchase Plan, as well as the notes to that table.
The maximum number of Common Shares any one eligible participant (including any insider of the Corporation) may elect to purchase under the Purchase Plan is determined by dividing the weighted average trading price of the Common Shares on the TSX for the five trading days immediately preceding the day in question (“Market Value on the offering date into an amount equivalent to five per cent of such participant’s base annual salary.
The purchase price of the Common Shares purchased pursuant to the Purchase Plan is the lesser of the Market Value on the offering date and the Market Value on the last business day of the purchase period. As a result, the purchase price could be below the market price of the Common Shares on the date of purchase.
A participant may cancel an election to purchase shares as to any or all of the shares specified therein at any time prior to the close of business on the last business day of the purchase period by written notice of cancellation delivered to and receipted for by the Corporation. Upon receipt of such notice of cancellation, the Corporation shall instruct the administrative agent under the Purchase Plan to return to the participant the appropriate portion of the accumulated instalment payments to the participant’s credit together with interest, if any, accrued thereon. If a participant cancels an election to purchase shares under any offering as to any or all of the shares specified therein then the participant shall not be entitled to rejoin or otherwise participate in such plan with respect to such cancelled shares.
Upon the termination of a participant’s employment with the Corporation by reason of retirement, disability or death, the participant or his legal representative may elect within 60 days after the date of termination of the participant’s employment, but not later than the end of the purchase period, either of the following:
8 INFORMATION CIRCULAR
(a) the aggregate of all the participant’s instalment payments, excluding interest accrued thereon, may be applied to the purchase of the greatest number of shares (rounded down to the nearest whole number) as may be purchased therewith, and any balance, including accrued interest, shall be distributed to the participant or his legal representative in cash; or
(b) the total number of shares specified in the participant’s election to purchase shares may be purchased by the application of all of the participant’s instalment payments to date, including interest accrued thereon, together with whatever additional lump sum cash payment needs to be made to meet the total purchase price and satisfy withholding or other deduction requirements, if any, for such shares.
If no valid election is made within the specified period, the total of all instalment payments accumulated in the participant’s account with the administrative agent, together with accrued interest thereon to the immediately preceding interest accrual date, shall be paid to the participant or his legal representative in full satisfaction of all his rights under the Purchase Plan.
A participant who does not return to active employment within twelve months from the commencement of a leave of absence or lay-off or prior to the end of the purchase period, whichever is the earlier, shall be deemed to be a terminated employee as of such date for purposes of the Purchase Plan offering.
Upon the termination of a participant’s employment with the Corporation other than by reason of retirement, disability or death, the participant may with the consent of the Corporation elect by written notice delivered to the Corporation within sixty days after the date of termination of the participant’s employment, but not later than the end of the purchase period, to have the aggregate of all the participant’s instalment payments, excluding interest accrued thereon, applied to the purchase of the greatest number of shares (rounded down to the nearest whole number) as may be purchased therewith, and any balance, including accrued interest, shall be distributed to the participant in cash. If consent of the Corporation is granted and the participant so elects, the purchase price shall be the lesser of the offer price and Market Value on the date of termination of the participant’s employment.
If the Corporation does not grant consent as aforesaid or if no such valid election is made within the specified period, then the total of all instalment payments accumulated in the participant’s account with the administrative agent, together with accrued interest thereon to the immediately preceding interest accrual date, shall be paid to the participant or the participant’s legal representative in full satisfaction of all of the participant’s rights under the Purchase Plan.
No right or interest of any participant in or under the Purchase Plan shall be assignable or transferable, in whole or in part, either directly or by operation of law or otherwise in any manner except as a result of death or mental incompetency of the participant, and shall be exercisable, during a participant’s lifetime, only by the participant. Subject to applicable regulatory approval, the Corporation may, in its sole discretion and for any reason, terminate the Purchase Plan at any time or may at any time or from time to time modify the Purchase Plan in its entirety or in part.
The Purchase Plan, as approved by the shareholders of the Corporation, provides that subject to approval by the TSX, the Corporation may terminate or modify the plan in part or in its entirety, and may rescind, change or cancel any offering date, as defined therein. Such amendments shall not affect the rights of a participant in respect of distribution or withdrawal of the participant’s instalment payment or interest accrued thereon or the number of shares which may be purchased with accumulated instalment payments. The rules of the TSX impose general restrictions on the nature of amendments to such plans which may be effected without shareholder approval. The Corporation will seek guidance from the TSX in respect of any proposed amendment as to whether regulatory or shareholder approval of such amendment is required.
No financial assistance is provided by the Corporation to participants to facilitate the purchase of Common Shares under the Purchase Plan.
INFORMATION CIRCULAR 9
Restricted Share Unit Plan
In 2004, the Corporation established the Restricted Share Unit Plan (the “RSU Plan”) to provide a more balanced approach to incentive compensation by including mid-term incentive compensation (i.e. RSUs, which are considered mid-term compensation by the Corporation, in comparison to Options, which are considered long-term compensation by the Corporation. However, RSUs are set out in the table Long Term Incentive Plan Awards Granted During the Year Ended December 31, 2005, below, since they constitute long-term compensation within the securities regulations applicable to the Corporation), in addition to solely short-term (i.e. base salary) and long-term (i.e. Options) incentive compensation. The purpose of the RSU Plan is to promote the mid-term and long-term success of the Corporation by providing the Board with additional flexibility to recruit, motivate and retain senior management through the issuance of RSUs to participants based on an assessment of a participant’s current and potential ability to contribute to the success of the Corporation and to the enhancement of the Corporation’s share value.
The eligible participants under the RSU Plan include any employee of the Corporation or of an affiliated entity designated by the Board (“Designated Employees”).
At any time, the aggregate number of Common Shares issued or which may be issued from the treasury of the Corporation pursuant to grants of RSUs allocated to Designated Employees together with the aggregate number of Common Shares issued or for which Options are outstanding under the Option Plan (including the D&O Program) shall not exceed the total number of Common Shares reserved for issuance under the Option Plan. The RSU Plan therefore does not provide any potential for dilution beyond that already existing under the Option Plan. As of March 8, 2006, there were 356,930 RSUs granted under the RSU Plan for 2003, 2004 and 2005. The 2003, 2004 and 2005 RSU grants provide that at least one-half of all RSUs granted will be settled in cash, with the other one-half settled at the option of the Corporation either in cash, by Common Shares issued from the treasury of the Corporation, or by Common Shares purchased by the Corporation on the open market. Therefore, the maximum number of Common Shares that could be issued from the treasury of the Corporation for the 2003, 2004 and 2005 grants of RSUs is 178,465, which equals approximately 0.2% of the currently outstanding Common Shares. The actual number of Common Shares issued from treasury will be less if the Corporation elects to settle some of these RSUs in cash or by shares purchased by the Corporation on the open market upon their vesting (see Long Term Incentive Plan Awards Granted During the Year Ended December 31, 2005, below). Additionally, on February 15, 2006, Messrs. Barry M. Heck, G.A. Fitch and A.B. Johnston were granted the right to earn up to a maximum of 57,739, 26,438, and 24,615 RSUs, respectively, during 2006 pursuant to the RSU Plan, subject to the achievement of certain objectives (the “2006 RSU grants”). The actual number of RSUs earned for 2006 will be established in early 2007 based on the achievement of these objectives, with the grants of these RSUs to fully vest in 2009 . Given that the terms of the 2006 RSU grants also provide that at least one-half of these RSUs must be settled in cash, the maximum number of Common Shares that could potentially be issued out of the treasury of the Corporation is 54,396, which represents approximately .06% of the currently outstanding Common Shares. Again, the actual dilution will be less if some RSUs are settled in cash or by shares purchased by the Corporation on the open market.
The maximum percentage of Common Shares under the RSU Plan which are available to any one person or company (including insiders of the Corporation) may not exceed approximately 11.57%, being the 10,750,000 Common Shares designated for issuance under the Option Plan as noted above.
The value of each RSU issued pursuant to the RSU Plan is the weighted average trading price for the Common Shares on the TSX for the five trading days immediately preceding the applicable date.
Unless otherwise determined by the Board or the administrator under the RSU Plan, and subject to any terms set out in the notice of grant or in any agreement(s) entered into between a participant and the Corporation or any of its affiliates:
(a) upon the death of a participant while in the employment of the Corporation or any of its affiliated entities, all RSUs granted to that participant prior to the date of death (but for which no release date has yet occurred) shall be deemed to be vested in the participant immediately on the date of death, with the release date therefor to be 10 days following the date of death, and the number of such RSUs that would otherwise be issued to such participant shall be issued in the form of RSU shares (or paid by cash if the grant so provides and the appropriate election is made) and delivered to the participant’s estate as if the participant had continued in the employment of the Corporation or one of its affiliated entities until the release date.
10 INFORMATION CIRCULAR
(b) except as specifically provided for in the RSU Plan or in any agreement between the participant and the Corporation or any of its affiliates, or as otherwise agreed to or determined by the Board or the administrator under the RSU Plan, if the employment of a participant with the Corporation or any of its affiliated entities is terminated for any reason prior to the release date for any RSUs, other than by early retirement or normal retirement from such employment, then the participant shall be deemed to have forfeited all right, title and interest with respect to any RSUs not fully vested upon that participant’s last day of such employment (the “Termination Date”). For this purpose, Termination Date means: (i) if the participant is terminated for just cause, the actual date of termination; and (ii) if the participant is terminated for reasons other than just cause, the date at the conclusion of any statutory, contractual or common law period of notice of termination of employment to which that participant is entitled.
Notwithstanding the foregoing, in the event that a participant’s employment with the Corporation or any of its affiliates is terminated without just cause or if the participant resigns from such employment then, at the sole and unfettered discretion of the Board or the administrator under the RSU Plan, all or any portion of the RSUs granted to that participant may be deemed to have vested on the date of termination or resignation, with the release date therefore to be 10 days following that termination or resignation.
Subject to:
(a) the provisions of any change of control agreement or any other agreement between the participant and the Corporation or any of its affiliates; and
(b) the terms and conditions upon which the grant was made; and
(c) the determination of the Board or the administrator under the RSU Plan;
if a participant’s employment terminates for any cause other than death, retirement, termination without just cause, authorized leave of absence or disability of the participant, or a change of control of the Corporation, then the RSUs covered by any grant to such participant with respect to which the release date has not occurred prior to such termination shall be forfeited as of the date of such termination of employment with the Corporation or any of its affiliated entities and such termination of employment for whatever reason shall not entitle a participant to any compensation for loss of any benefit under the RSU Plan.
If a participant retires from employment with the Corporation before his or her 65th birthday (“Early Retirement”) then any RSUs covered by any grant to such participant with respect to which the release date has not occurred and for which RSUs have not been issued prior to such retirement shall be deemed to be vested in accordance with the following calculation: (x) for each grant, the number of RSUs granted pursuant to the applicable notice of grant shall be multiplied by a fraction, the numerator of which is the number of days between the effective date therefor and the date of Early Retirement and the denominator of which is the number of days in the grant period attributable to the grant in question; (y) the number of RSUs resulting from such calculation shall be deemed to be vested on the date of Early Retirement and the unvested balance of such RSUs shall be forfeited; (z) except as provided in this paragraph, all RSUs not vested at the date of Early Retirement shall expire on the date of Early Retirement.
If a participant retires from employment with the Corporation on or after his or her 65th birthday then all RSUs covered by any grant to such participant with respect to which the release date has not occurred and for which RSUs have not been issued prior to such retirement shall continue to vest according to the terms of the RSU Plan, the applicable notice of grant and any agreements entered into with the specific participant in respect of that grant.
If the employment of a participant with the Corporation is interrupted, suspended or terminated due to an authorized leave of absence or disability prior to the release date in respect of any RSUs covered by any grant to such participant with respect to which the release date has not occurred and for which RSUs have not been issued prior to such interruption, suspension or termination of employment then all such RSUs shall continue to vest as if that participant continued to be actively employed until the release date therefor.
The rights or interests of an eligible participant under the RSU Plan are not assignable or transferable, otherwise than by will or the laws governing the devolution of property in the event of death.
The Board may from time to time amend the RSU Plan in whole or in part. Subject to the rules of the TSX, Shareholder approval is not required for such amendments.
No financial assistance is provided by the Corporation to participants under the RSU Plan.
INFORMATION CIRCULAR 11
STATEMENT OF EXECUTIVE COMPENSATION
Summary Compensation Table
The following table summarizes, for the periods indicated, the compensation of the Corporation’s President and Chief Executive Officer, Chief Financial Officer and other executive officers serving at December 31, 2005 whose total salary and bonus earned in the financial year ended December 31, 2005 exceeded $150,000. Such executive officers are referred to collectively herein as the “Named Executive Officers.”
| Number of | ||||||||||||||||||||||||||||||||
| Long Term Compensation | Common | |||||||||||||||||||||||||||||||
| Annual Compensation | Awards | Shares | ||||||||||||||||||||||||||||||
| Other | Securities | All | Beneficially | |||||||||||||||||||||||||||||
| Name and | Year | Annual | Under | Other | Owned or | |||||||||||||||||||||||||||
| Principal | Ended | Compen- | Options | Restricted | Compen- | Controlled | ||||||||||||||||||||||||||
| Position | Dec. 31 | Salary | Bonus | sation | 1 | Granted | 2 | Share Units | 3 | sation | 4 | at Year End | ||||||||||||||||||||
B.M. Heck5 | 2005 | $ | 446,875 | $ | 150,000 | 6 | $ | 40,000 | 100,000 | 81,195 | $ | 33,817 | 86,939 | |||||||||||||||||||
| President and | 2004 | $ | 422,917 | — | $ | 40,000 | 107,500 | 70,733 | $ | 32,172 | 78,465 | |||||||||||||||||||||
| Chief Executive Officer | 2003 | $ | 393,750 | — | $ | 39,400 | 200,000 | 50,000 | $ | 33,272 | 70,965 | |||||||||||||||||||||
| G.A. Fitch | 2005 | $ | 278,125 | $ | 84,000 | 6 | $ | 25,000 | 65,000 | 37,526 | $ | 17,173 | 106,888 | |||||||||||||||||||
| Senior Vice President and | 2004 | $ | 265,000 | $ | 50,000 | 6 | $ | 25,000 | 43,000 | 33,079 | $ | 21,017 | 101,274 | |||||||||||||||||||
| Chief Financial Officer | 2003 | $ | 262,500 | — | $ | 25,000 | 75,000 | 20,000 | $ | 16,736 | 98,274 | |||||||||||||||||||||
| A.B. Johnston | 2005 | $ | 258,125 | $ | 78,000 | 6 | $ | 25,000 | 60,000 | 33,715 | $ | 21,619 | 94,834 | |||||||||||||||||||
| Senior Vice President | 2004 | $ | 245,000 | $ | 70,249 | 7 | $ | 25,000 | 40,000 | 10,682 | $ | 19,082 | 94,834 | |||||||||||||||||||
| 2003 | $ | 242,500 | $ | 48,500 | 7 | $ | 25,000 | 50,000 | 20,000 | $ | 21,190 | 94,834 | ||||||||||||||||||||
B.D. Heck8 | 2005 | $ | 175,000 | $ | 85,860 | 7 | $ | 10,000 | 9 | 25,000 | — | — | 50,000 | |||||||||||||||||||
| Vice President, | 2004 | $ | 145,834 | $ | 56,190 | 7,10 | $ | 28,334 | 9 | 75,000 | — | — | 50,000 | |||||||||||||||||||
| General Counsel | 2003 | — | — | $ | 124,300 | 11 | — | — | — | |||||||||||||||||||||||
| and Corporate Secretary | ||||||||||||||||||||||||||||||||
| 1. | Barry M. Heck received cash allowances in lieu of perquisites for each of the years 2004 and 2005 in the amount of $40,000, which amounts are included in Other Annual Compensation. In 2003, Barry M. Heck received a cash allowance in lieu of perquisites of $39,375 which is included in Other Annual Compensation. For Messrs. Fitch and Johnston, amounts included in Other Annual Compensation were allowances in lieu of perquisites. | |
| 2. | Grants of Options pursuant to the Option Plan, which includes the D&O Program. Particulars of the grants of Options under the D&O Program are given in the table Option Grants During the Year Ended December 31, 2005, below. | |
| 3. | Amounts shown for 2003, 2004 and 2005 represent grants of RSUs pursuant to the RSU Plan made in consideration of annual performance. These grants were made on February 19, 2004 in relation to 2003 performance, on February 17, 2005 in relation to 2004 performance and grants of the right to earn RSUs in relation to 2005 performance were made on February 17, 2005, with actual grants earned for 2005 being established and confirmed on February 15, 2006. |
12 INFORMATION CIRCULAR
| 4. | Barry M. Heck received other compensation in 2003, 2004 and 2005 in the form of contributions to the Corporation’s defined contribution pension plan and life insurance premiums. Messrs. Fitch and Johnston received other compensation in the form of contributions to the Corporation’s defined contribution pension plan. | |
| 5. | Barry M. Heck was appointed President and Chief Executive Officer of the Corporation on January 15, 2003 and prior to that he was a Senior Vice President of the Corporation. | |
| 6. | For the year ended December 31, 2005, Messrs. Barry M. Heck, Fitch and Johnston each received bonus compensation in respect of their exceptional 2005 performance. For the year ended December 31, 2004, Mr. Fitch received bonus compensation in respect of his role in the completion of a transaction for the Corporation. See also Human Resources and Compensation Committee Report on Executive Compensation – Executive Compensation Policy, and Human Resources and Compensation Committee Report on Executive Compensation – Chief Executive Officer Compensation, below. | |
| 7. | These bonuses were paid under the Corporation’s variable pay program, an annual bonus system which is part of the compensation structure that encourages employees to work toward and to share in corporate and business unit/subsidiary success. All permanent full time and part time employees of the Corporation and its subsidiaries, except for the most senior executive officers of the Corporation, participate in this program based on the achievement of specific goals or measures established each year. Mr. Johnston was, during 2003 and 2004, eligible to participate in the variable pay program in respect of his employment with iFire Technology Inc. and iFire Technology Corp., subsidiaries of the Corporation. Brian D. Heck was, during 2004 and 2005, eligible to participate in the variable pay program in respect of his employment as Vice President, General Counsel and Corporate Secretary of the Corporation. Messrs. Barry M. Heck, Fitch and Johnston are not eligible to participate in the variable pay program in respect of their employment with the Corporation. | |
| 8. | Brian D. Heck was appointed General Counsel and Corporate Secretary of the Corporation on February 1, 2003, and appointed Vice President of the Corporation on March 1, 2004. | |
| 9. | For the period from January 1, 2004 to February 28, 2004, Brian D. Heck received other annual compensation of $20,000 in the form of contractor income for his role as General Counsel and Corporate Secretary, and for the years ended December 31, 2004 and December 31, 2005, he received cash allowances of $8,334 and $10,000 respectively in lieu of perquisites. | |
| 10. | On January 1, 2004, prior to becoming a Named Executive Officer, Brian D. Heck received a grant of 22,000 bonus appreciation units under the Corporation’s Corporate Support Groups Long Term Cash Bonus Incentive Plan. See Human Resources and Compensation Committee Report on Executive Compensation – Executive Compensation Policy, below. | |
| 11. | For the period from February 1, 2003 to December 31, 2003, Brian D. Heck received other annual compensation of $124,300 in the form of contractor income for his role as General Counsel and Corporate Secretary. |
Long Term Incentive Plan Awards Granted During the Year Ended December 31, 2005
| Securities, Units or Rights | Performance or | |||||||
| Name | (being RSUs in each case) | Other Period Until Maturation | ||||||
| Vesting Date | ||||||||
| Barry M. Heck | 81,195 | February 17, 2008 | ||||||
| G.A. Fitch | 37,526 | February 17, 2008 | ||||||
| A.B. Johnston | 33,715 | February 17, 2008 | ||||||
| B.D. Heck | Nil | Nil | ||||||
INFORMATION CIRCULAR 13
Option Grants During the Year Ended December 31, 2005
| % of Total | Market Value of | |||||||||||||||||||
| Options | Securities Underlying | |||||||||||||||||||
| Securities Under | Granted to | Exercise or | Options on | |||||||||||||||||
| Options | Employees in | Base Price | the Date of Grant | |||||||||||||||||
| Name | Granted1 | Financial Year | ($/Security) | ($/Security) | Expiration Date | |||||||||||||||
| B.M. Heck | 100,000 | 2 | 40 | % | $ | 3.62 | $ | 3.62 | Feb. 17, 2015 | |||||||||||
| G.A. Fitch | 65,000 | 2 | 26 | % | $ | 3.62 | $ | 3.62 | Feb. 17, 2015 | |||||||||||
| A.B. Johnston | 60,000 | 2 | 24 | % | $ | 3.62 | $ | 3.62 | Feb. 17, 2015 | |||||||||||
| B.D. Heck | 25,000 | 2 | 10 | % | $ | 3.62 | $ | 3.62 | Feb. 17, 2015 | |||||||||||
| 1. | These awards were made in Options to purchase Common Shares of the Corporation pursuant to the Option Plan and, in the ordinary course, are exercisable for a period ending 10 years from the date of the grant. | |
| 2. | These awards were granted on the basis that in the ordinary course they will be exercisable as to one third of the grant on each of the first, second and third anniversaries of the date of the grant. |
Aggregated Option Exercises During the Year Ended December 31, 2005
and Financial Year-End (“FY-End”) Option Values
and Financial Year-End (“FY-End”) Option Values
| Value of Unexercised | ||||||||||||||||||||
| Securities | Aggregate | in-the-Money | ||||||||||||||||||
| Acquired | Value | Options at FY-End | Options at FY-End1 | |||||||||||||||||
| Name | on Exercise | Realized | Exercisable/Unexercisable | Exercisable/Unexercisable | ||||||||||||||||
| B.M. Heck | Nil | Nil | 638,765 / 233,335 | $ | 400,523 / $260,003 | |||||||||||||||
| G.A. Fitch | Nil | Nil | 565,833 / 116,667 | $ | 153,789 / $111,200 | |||||||||||||||
| A.B. Johnston | Nil | Nil | 501,165 / 103,335 | $ | 83,730 / $88,069 | |||||||||||||||
| B.D. Heck | Nil | Nil | 58,333 / 41,667 | $ | 53,924 / $29,333 | |||||||||||||||
| 1. | Based on the December 31, 2005 closing price of $4.28 for the Common Shares on the TSX. |
Employment Contracts
The Corporation has entered into agreements with Messrs. Fitch and Johnston providing that, in the event of a termination of employment by the Corporation for reasons other than just cause or other than upon a change of control they will be entitled to 24 months’ salary and continued benefits under all benefits plans for a period of 24 months, and to the continued vesting of all Options granted under the Option Plan and all RSUs granted under the RSU Plan for a period of 24 months. The Corporation has also entered into agreements with Messrs. Fitch and Johnston providing that, in the event of a change of control of the Corporation, each of them will be entitled to receive payment equivalent to their monthly salary and the value of all perquisites and benefits under all benefits plans for a period of 24 months upon termination of their employment within 12 months of the change of control or for a period of 12 months if they elect to leave their employment within 90 days of the change of control. In 2004, the Corporation entered into an employment agreement with Brian D. Heck, providing that in the event of a
14 INFORMATION CIRCULAR
termination of employment by the Corporation for reasons other than just cause, he will be entitled to 12 months’ salary and continued benefits under all benefits plans for a period of 12 months, and to the continued vesting of all Options granted under the Option Plan and all RSUs granted under the RSU Plan for a period of 12 months. Also, in the event of a change of control of the Corporation, each of Messrs. Fitch, Johnston and Brian D. Heck will be entitled to immediate vesting of all Options granted under the Option Plan and of all RSUs granted under the RSU Plan.
The Corporation has entered into an agreement with Barry M. Heck providing that, in the event of a termination of employment by the Corporation for reasons other than just cause or other than upon a change of control he will be entitled to 24 months’ salary and continued benefits under all benefits plans for a period of 24 months, and to the continued vesting of all Options granted under the Option Plan and all RSUs granted under the RSU Plan for a period of 24 months. In 2003 the Corporation entered into an agreement with Barry M. Heck providing that, in the event of a change of control of the Corporation, he will be entitled to receive payment equivalent to his salary and the value of all perquisites and benefits under all benefits plans for a period of 36 months upon termination of employment by the Corporation or his election to leave employment within 90 days of the change of control. Also, in the event of a change of control of the Corporation, Barry M. Heck will also be entitled to immediate vesting of all Options granted under the Option Plan and all RSUs granted under the RSU Plan.
HUMAN RESOURCES AND COMPENSATION COMMITTEE REPORT ON EXECUTIVE COMPENSATION
Composition of the Human Resources and Compensation Committee
The members of the Compensation Committee are Neil Carragher, Roger G.H. Downer and Guy J. Turcotte. No member of the Compensation Committee is employed by the Corporation or its affiliates and no member is a former officer or employee of the Corporation or its affiliates.
Executive Compensation Policy
The objective of the Corporation’s executive compensation policy is to motivate and reward the creation of long term Shareholder value. To this end, stock options have historically been the focus of the executive compensation program.
In establishing the Corporation’s executive compensation policy, the Compensation Committee periodically reviews the Corporation’s executive compensation (base salary and long-term, mid-term and short-term incentive programs) using comparative North American industry data provided by the Corporation’s independent professional compensation consultants. These comparisons are adjusted to reflect the relative size of the Corporation, the nature of the Corporation’s business and the particular job functions and performance of executives.
Executive base salaries are targeted at the median of base salaries, as determined by such comparisons. The Corporation targets its benefits and perquisites at approximately the average for comparable companies. It is the policy of the Corporation not to pay regular annual bonuses to the most senior executive officers except in respect of their roles as executive officers of subsidiaries of the Corporation (see note 7 of Statement of Executive Compensation — Summary Compensation Table, above). However, the Compensation Committee has the authority to recognize exceptional performance by the senior executive officers of the Corporation, and the performance of those senior executive officers was judged exceptional in 2005, resulting in the award of bonuses to them. See Statement of Executive Compensation — Summary Compensation Table, above.
Annual grants of Options (not including grants of Options under the D&O Program — see Directors and Officers Share Purchase Program, above) and RSUs for the Corporation’s most senior executive officers are intended to result in their total executive compensation package being approximately at the 75th percentile of comparable companies when performance of the executive and the Corporation warrants such compensation.
The Board believes that, in combination with other elements of a compensation package, the Option Plan and the RSU Plan provide useful compensation tools for aligning executive interests with the interests of Shareholders.
The Corporation has established the 2004 Equity Incentive Plan (“EIP”) for employees of its emerging technology business subsidiary, iFire Technology Corp., and the Corporate Support Groups Long Term Cash Bonus Incentive Plan (“LTIP”) for employees of the Corporation. The EIP and the LTIP provide for awards of stock appreciation rights or stock options in the subsidiary or stock appreciation rights in the Corporation. The Named Executive Officers do not participate in the EIP or the LTIP in their capacities as executive officers of the Corporation,
INFORMATION CIRCULAR 15
although Brian D. Heck received an award of 22,000 bonus appreciation units under the LTIP in January 2004, prior to becoming a Named Executive Officer of the Corporation. See Statement of Executive Compensation – Summary Compensation Table, footnote 10, above.
Executive Compensation Procedures
The Compensation Committee develops a compensation program for the directors and the President and Chief Executive Officer. Compensation programs for other executive officers are developed by the President and Chief Executive Officer, with the approval of the Committee. Executive compensation is reviewed and set annually.
Chief Executive Officer Compensation
The Chief Executive Officer’s compensation is established using the policy and procedures set forth above.
For 2005, Barry M. Heck’s annual base salary was $446,875.00, and a bonus of $150,000 was paid to him in respect of his exceptional 2005 performance. On February 15, 2006, the Chief Executive Officer’s annual base salary was increased to $475,000. A grant of 100,000 Options was made to Barry M. Heck on February 17, 2005, and a grant of 100,000 Options was made to him on February 20, 2006. A grant of the right to earn RSUs in respect of 2005 performance was made to the Chief Executive Officer on February 17, 2005 pursuant to the RSU Plan, with the actual number of 81,195 RSUs earned being established and confirmed on February 15, 2006. On February 15, 2006, Barry M. Heck was granted the right to earn up to 57,739 RSUs during 2006 pursuant to the RSU Plan, subject to the achievement of certain objectives. The actual number of RSUs earned for 2006 will be established in early 2007 based on the achievement of these objectives.
The Compensation Committee believes that it is important for the Chief Executive Officer of the Corporation to have significant exposure to the Corporation’s share price. As indicated above, Options are the focus of the long-term compensation program and RSUs are the focus of the mid-term compensation program for the Chief Executive Officer; other compensation elements are secondary.
Report presented by:
Neil Carragher, Chairman
Roger G.H. Downer
Guy J. Turcotte
Neil Carragher, Chairman
Roger G.H. Downer
Guy J. Turcotte
Performance Graph
The following graph shows changes as at December 31 of each year, since December 31, 1999, assuming an investment in the Corporation valued at $100 as at December 31, 2000, and an investment in the S&P/TSX Composite Index valued at $100 as at December 31, 2000, and assuming reinvestment of dividends in both cases.
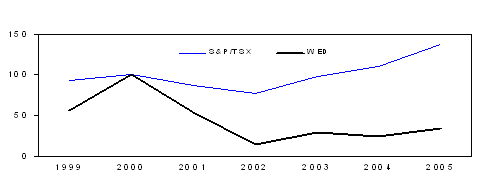
Remuneration of Directors
Directors other than Messrs. Barry M. Heck and Delaney are paid at the rate of $5,000 per Board meeting attended in person, $2,000 per committee meeting attended in person and $1,000 for each Board or committee telephone meeting. Where committee meetings attended in person last longer than four hours, an additional $1,000 is paid. Committee chairmen receive an additional $1,000 for meetings attended in person, a further $2,000 for such meetings lasting longer than four hours and an additional $500 for committee meetings held by telephone. The chairman of the Audit Committee receives an additional annual retainer of $5,000. Where a director travels for more than three hours to attend a Board or committee meeting, an additional fee of $2,000 is paid, without duplication.
16 INFORMATION CIRCULAR
Maximum daily fees payable are $7,000 for directors and $8,000 for committee chairmen who chair a meeting on the same day, plus travel fees. Travel expenses are paid in addition to meeting fees.
As chairman of the Board, Mr. Delaney receives an annual fee of $100,000 in lieu of director’s meeting fees. He is also reimbursed for expenses incurred in connection with attendance at meetings of the Board and committees.
Each of the directors, other than Messrs. Delaney and Barry M. Heck and Dr. Downer, has been granted an aggregate of 180,000 Options to purchase Common Shares. Dr. Downer was granted 60,000 Options to purchase Common Shares in October of 2004 at an exercise price of $3.13 and a further 40,000 Options to purchase Common Shares in October of 2005 at an exercise price of $4.00. In addition, all directors are designated participants in the D&O Program described under the heading Security Based Compensation Arrangements in this Information Circular.
In February of 2005, Mr. Delaney received a grant of Options to purchase 100,000 Common Shares at an exercise price of $3.62. Mr. Delaney holds an aggregate of 1,597,200 Options including a grant of 50,000 Options in February of 2006 at an exercise price of $6.18.
The above Options to directors were granted on the basis that in the ordinary course they will be exercisable as to one third of the grant on each of the first, second and third anniversaries of the date of the grant and are exercisable for a period ending 10 years from the date of the grant.
In 2001, the Corporation established a Deferred Share Unit Plan (the “DSU Plan”) for eligible directors. Under the DSU Plan, at the end of each calendar quarter all eligible directors are granted DSUs equal in value to the meeting fees payable to that director for the preceding quarter. A DSU is attributed a value based on the closing price of the Common Shares on the TSX for the trading day immediately preceding the date of grant (previously defined in this Information Circular as the “Market Price”). In addition, eligible directors may elect to receive 50% or 100% of their meeting fees in the form of DSUs, valued at a 10% discount to Market Price upon grant. All DSUs will be paid out in cash only. The value of a DSU, when converted to cash, is equivalent to the Market Price of a Common Share at the time the conversion takes place. A director cannot convert DSUs to cash until the director ceases to be a member of the Board. Mr. Delaney and Mr. Barry M. Heck are not eligible to participate in the DSU Plan. The DSUs held by directors are reported in the Shareholdings column of the table under Election of Directors, above.
Directors’ and Officers’ Liability Insurance
Directors’ and officers’ liability insurance in the amount of $40,000,000 was purchased during the last completed financial year at the Corporation’s expense for the protection of all the directors and officers against liability incurred by them in their capacities as directors and officers of the Corporation and its past and present subsidiaries. The premium paid by the Corporation for such insurance is currently $242,400 per year. There is a deductible to the Corporation of $250,000 per occurrence.
Audit Committee
Information regarding the Audit Committee of the Board of Directors of the Corporation is disclosed in the Corporation’s Annual Information Form under the section “Directors and Officers – Audit Committee”which has been filed on SEDAR atwww.sedar.com.
STATEMENT OF CORPORATE GOVERNANCE
The Board sees its principal role as stewardship of the Corporation and its fundamental objective as the creation of Shareholder value, including the protection and enhancement of the value of the Corporation’s assets and operating with honesty and integrity in the conduct of business. The Board’s stewardship responsibility means that it oversees the conduct of the business and of Management, which is responsible for developing long-term strategy and conducting the Corporation’s day-to-day business. As part of the Corporation’s commitment to effective corporate governance, the Board, with the assistance of the Corporate Governance Committee, monitors changes in corporate governance practices and regulatory requirements.
During the past year, there have been several changes to the corporate governance disclosure requirements applicable to the Corporation. Specifically, the Canadian Securities Administrators introduced in final form National Instrument 58-101Disclosure of Corporate Governance Practices (“NI 58-101”) and National Policy 58-201Corporate Governance Guidelines(“NP 58-201”), both of which came into force on June 30, 2005 and effectively
INFORMATION CIRCULAR 17
replaced the corporate governance guidelines and disclosure policies of the TSX. Under NI 58-101, the Corporation is required to disclose certain information relating to its corporate governance practices. This information is set forth below.
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| 1. | Board of Directors | |||||||||
| (a) | Disclose the identity of directors who are independent. | The Board has determined that seven of the eight directors are “independent”, within the meaning of NI 58-101. The seven independent directors are: | ||||||||
| • | Neil Carragher; | |||||||||
| • | Ian W. Delaney; | |||||||||
| • | Roger G.H. Downer; | |||||||||
| • | Frank W. King; | |||||||||
| • | Daniel P. Owen; | |||||||||
| • | Guy J. Turcotte; and | |||||||||
| • | Bruce V. Walter. | |||||||||
| (b) | Disclose the identity of directors who are not independent, and describe the basis for that determination. | Mr. Barry M. Heck is the President and Chief Executive Officer of the Corporation and is therefore not considered to be an independent director. | ||||||||
| (c) | Disclose whether or not a majority of directors are independent. If a majority of directors are not independent, describe what the Board of Directors does to facilitate its exercise of independent judgment in carrying out its responsibilities | The Board has determined that a majority of the directors are “independent”, within the meaning of NI 58-101. | ||||||||
| (d) | If a director is presently a director of any other issuer that is a reporting issuer (or the equivalent) in a jurisdiction or a foreign jurisdiction, identify both the director and the other issuer. | The following directors currently serve on the board of other issuers that are reporting issuers (or equivalent) which are set out below: | ||||||||
| Director | Directorships | |||||||||
| • | Barry M. Heck | Kereco Energy Ltd. NUCRYST Pharmaceuticals Corp. | ||||||||
| • | Neil Carragher | Agrium Inc. NUCRYST Pharmaceuticals Corp. | ||||||||
| • | Ian W. Delaney | Dynatec Corporation EnCana Corporation OPTI Canada Inc. Sherritt International Corporation | ||||||||
| • | Roger G.H. Downer | NUCRYST Pharmaceuticals Corp. | ||||||||
| • | Frank W. King | Agrium Inc. CanWest Global Communications Corp. RioCan Real Estate Investment Trust Wi-LAN Inc. | ||||||||
| • | Daniel P. Owen | Dynatec Corporation Dynex Power Inc. Sherritt International Corporation | ||||||||
| • | Guy J. Turcotte | Fort Chicago Energy Partners L.P. Western Oil Sands Inc. | ||||||||
| • | Bruce V. Walter | Dynatec Corporation FNX Mining Company Inc. | ||||||||
18 INFORMATION CIRCULAR
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| (e) | Disclose whether or not the independent directors hold regularly scheduled meetings at which members of management are not in attendance. If the independent directors hold such meetings, disclose the number of meetings held during the preceding 12 months. If the independent directors do not hold such meetings, describe what the board does to facilitate open and candid discussion among its independent directors. | Each independent director of the Corporation is a member of the Corporate Governance Committee of the Board and Barry M. Heck, the only non-independent director of the Corporation, is not a member of that committee. The Corporate Governance Committee meets independently of management on a regular basis, and the Board during its meetings also has regular in-camera discussions with all members of Management, including Barry M. Heck, excused from that portion of the meeting. During the financial year ended December 31, 2005, there were 3 meetings of the Corporate Governance Committee. In addition, the independent members of the Board are authorized to retain independent financial, legal and other experts or advisors as required whenever, in their opinion, matters come before the Board or any committee which require an independent analysis by the independent members of the Board or any committee. | ||||||||
| (f) | Disclose whether or not the chair of the board is independent. If the board has a chair or lead director who is independent, disclose the identity of the independent chair or lead director, and describe his or her role and responsibilities. If the board has neither a chair that is independent nor a lead director that is independent, describe what the board does to provide leadership for its independent directors. | Mr. Ian W. Delaney, the chairman of the Board, is independent of the Corporation. The role of the chairman of the Board includes ensuring that the Board discharges its duties independently of Management, serving as chairman of the Corporate Governance Committee, and setting the agendas for the meetings of the Board and its committees in consultation with the Chief Executive Officer of the Corporation. See also the corporate governance practices of the Corporation described under section 3. (a) of this statement, below, for further details of the role and responsibilities of the chairman of the Board. | ||||||||
| (g) | Disclose the attendance record of each director for all board meetings held since the beginning of the issuer’s most recently completed financial year. | See Committee Memberships and Record of Attendance at Meetings, above. | ||||||||
| 2. | Mandate of the Board of Directors | |||||||||
| Disclose the text of the Board’s written mandate. If the Board does not have a written mandate, describe how the Board delineates its role and responsibilities. | As at the date hereof, the Board has not formally adopted a written mandate. The Board sees its principal role as stewardship of the Corporation and its fundamental objective as the creation of Shareholder value, including the protection and enhancement of the value of the Corporation’s assets and operating with honesty and integrity in the conduct of business. The Board’s stewardship responsibility means that it oversees the conduct of the business and of Management, which is responsible for developing long-term strategy and conducting the Corporation’s day-to-day business. | |||||||||
| The Board meets frequently and is comprised of individuals with considerable experience as directors of public companies and in respect of corporate governance. The agenda for each Board meeting is carefully planned and set by the chairman of the Board working in conjunction with the Chief Executive Officer. Each of the committees of the Board have specific responsibilities delineated in the terms of reference or charter established for each respective committee, such terms of reference or charter having been approved by the Board in each case. | ||||||||||
INFORMATION CIRCULAR 19
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| 3. | Position Descriptions | |||||||||
| (a) | Disclose whether or not the Board has developed written position descriptions for the chair and the chair of each Board committee. If the Board has not developed written position descriptions for the chair and/or the chair of each Board committee, briefly describe how the Board delineates the role and responsibilities of each such position. | As at the date hereof, the Board has not developed a written position description for the chairman of the Board. Generally, the principal role assigned to the chairman of the Board by the Board includes providing leadership to the Board and acting as a direct liaison between the Board and Management. Further, the chairman of the Board is responsible for ensuring that the Board properly discharges its responsibilities, that the members of the Board have full opportunity to participate in meetings of the Board, and that all Board matters are properly and adequately addressed. The chairman of the Board is accountable to the Board. The terms of reference or the charter of each of the Audit Committee, the Corporate Governance Committee, the Human Resources and Compensation Committee and the Environmental, Health and Safety Committee describe certain of the responsibilities of the chairman of each of these committees. The primary role of the chairman of each such committee is managing the affairs of the committee, including ensuring the committee is organized properly, functions effectively and meets its obligations and responsibilities. The chairman of the Audit Committee also maintains on-going communications with the Corporation’s external auditors in order to lead the committee in performing its oversight and other audit-related functions. For further information regarding the Corporation’s Audit Committee, including the relevant education and experience of the committee members, see the Corporation’s Annual Information Form for the financial year ended December 31, 2005 which is available on SEDAR at www.sedar.com. | ||||||||
| (b) | Disclose whether or not the Board and CEO have developed a written position description for the CEO. If the Board and CEO have not developed such a position description, briefly describe how the Board delineates the role and responsibilities of the CEO. | The Board does not have a specific written position description for the Chief Executive Officer of the Corporation. However, the role, responsibilities, and duties of the Chief Executive Officer are generally set out in his contract of employment with the Corporation, and are constantly reviewed and refined by the Board. Additionally, the specific annual objectives of the Chief Executive Officer are set, and his performance is reviewed, by the Board and by the Human Resources and Compensation Committee of the Board as circumstances dictate. | ||||||||
| 4. | Orientation and Continuing Education | |||||||||
| (a) | Briefly describe what measures the Board takes to orient new directors regarding (i) the role of the Board, its committees and its directors, and (ii) the nature and operation of the issuer’s business. | Immediately following appointment, new directors are provided with historic information, current strategic plans for the Corporation and its subsidiaries, and materials summarizing issues relating to the Corporation. New directors are also briefed by the chairman of the Board, by the Chief Executive Officer of the Corporation, and by the chairmen of the committees of the Board, if any, to which they are appointed, as well as by senior management of the subsidiaries of the Corporation, and receive tours of the facilities of the Corporation and its subsidiaries. | ||||||||
| (b) | Briefly describe what measures, if any, the Board takes to provide continuing education for its directors. If the Board does not provide continuing education, describe how the Board ensures that its directors maintain the skill and knowledge necessary to meet their obligations as directors | The Board and its committees receive periodic reports from Management and external advisors as to new developments in regard to corporate governance and in regard to other issues affecting the Corporation. | ||||||||
20 INFORMATION CIRCULAR
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| 5. | Ethical Business Conduct | |||||||||
| (a) | Disclose whether or not the Board has adopted a written code for the directors, officers and employees. If the Board has adopted a written code: (i) disclose how a person or company may obtain a copy of the code; (ii) describe how the Board monitors compliance with its code, or if the Board does not monitor compliance, explain whether and how the Board satisfies itself regarding compliance with its code; and (iii) provide a cross-reference to any material change report filed since the beginning of the issuer’s most recently completed financial year that pertains to any conduct of a director or executive officer that constitutes a departure from the code. | The Board has adopted a Code of Conduct and Ethics for Directors, Officers and Employees and a Finance Code of Conduct for Chief Executive and Senior Financial Officers (the “Codes”). The Codes have been filed on and are accessible through SEDAR atwww.sedar.com. Copies of the Codes may also be obtained, upon request, from the Corporation at 10102 114th Street, Fort Saskatchewan, AB T8L 3W4, Attention: Vice President, General Counsel and Corporate Secretary. The Board expects directors and employees to act ethically at all times and to acknowledge their adherence to the policies comprising the Codes. Any material issues regarding compliance with the Codes are brought forward by Management at either the Board or appropriate committee meetings, or are referred to the senior executive officers of the Corporation, as may be appropriate in the circumstances. The Board and/or appropriate committee or senior executive officers determine what remedial steps, if any, are required. Any waivers from the Codes that are granted for the benefit of a director or an employee may be granted only by the Board or by the Corporate Governance Committee or the Audit Committee. No waiver has ever been granted under the Codes. | ||||||||
| (b) | Describe any steps the Board takes to ensure directors exercise independent judgment in considering transactions and agreements in respect of which a director or executive officer has a material interest. | Each director must disclose all actual or potential conflicts of interest and refrain from voting on matters in which such director has a conflict of interest. In addition, the director must excuse himself or herself from any discussion or decision on any matter in which the director is precluded from voting as a result of a conflict of interest. | ||||||||
| (c) | Describe any other steps the Board takes to encourage and promote a culture of ethical business conduct | The Corporation has adopted a Disclosure Policy, an Insider Trading Policy and a Whistleblower Policy. | ||||||||
| 6. | Nomination of Directors | |||||||||
| (a) | Describe the process by which the Board identifies new candidates for Board nomination. | The Corporate Governance Committee is mandated to recruit and consider director candidates and to make recommendations to the Board. In so doing, the Corporate Governance Committee considers, in addition to any other factors it deems relevant: (i) the competencies and skills that the Board considers to be necessary for the Board, as a whole, to possess; (ii) the competencies and skills that the Board considers each existing director to possess; (iii) the competencies, skills and background each nominee will bring to the Board; (iv) the time that each nominee will have available to devote to the Corporation’s business; and (v) whether the nominee will be an independent director. Directors are encouraged to identify potential candidates. An invitation to stand as a nominee for election to the Board will normally be made to a candidate by the Board through the chairman of the Board or his delegate. | ||||||||
| (b) | Disclose whether or not the Board has a nominating committee composed entirely of independent directors. If the Board does not have a nominating committee composed entirely of independent directors, describe what steps the Board takes to encourage an objective nomination process. | The Corporate Governance Committee is composed of Messrs. Carragher, Delaney, Downer, King, Owen, Turcotte and Walter, all of whom are independent. | ||||||||
INFORMATION CIRCULAR 21
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| (c) | If the Board has a nominating committee, describe the responsibilities, powers and operation of the nominating committee. | In addition to recruiting and considering director candidates, the Corporate Governance Committee’s mandate includes annual reviewing the competencies, skills and personal qualities applicable to candidates to be considered for nomination to the Board. The objective of this review is to maintain the composition of the Board in a way that provides, in the judgment of the Board, the best mix of competencies, skills and experience to provide for the overall stewardship of the Corporation. | ||||||||
| 7. | Compensation | |||||||||
| (a) | Describe the process by which the Board determines the compensation for the issuer’s directors and officers. | See Human Resources and Compensation Committee Report on Executive Compensation above for a description of how the compensation for the directors and officers of the Corporation is determined. | ||||||||
| (b) | Disclose whether or not the Board has a compensation committee composed entirely of independent directors. If the Board does not have a compensation committee composed entirely of independent directors, describe what steps the Board takes to ensure an objective process for determining such compensation. | The Human Resources and Compensation Committee is composed of Messrs. Carragher, Downer and Turcotte, all of whom are independent. | ||||||||
| (c) | If the Board has a compensation committee, describe the responsibilities, powers and operation of the Compensation Committee. | The Human Resources and Compensation Committee has the responsibility of annually reviewing and approving the compensation package for Management. The Human Resources and Compensation Committee also reviews and approves changes to the Corporation’s compensation policies in respect of matters such as pension plans and employee benefit plans. Lastly, the Human Resources and Compensation Committee approves the hiring of management recruited from outside the Corporation, as well as the promotion of management within the Corporation. | ||||||||
| (d) | If a compensation consultant or advisor has, at any time since the beginning of the issuer’s most recently completed financial year, been retained to assist in determining compensation for any of the issuer’s directors and officers, disclose the identity of the consultant or advisor and briefly summarize the mandate for which they have been retained. If the consultant or advisor has been retained to perform any other work for the issuer, state that fact and briefly describe the nature of the work. | During 2005, the Human Resources and Compensation Committee of the Board retained the firm of Towers Perrin to undertake a review of compensation for directors and the chairman of the Board. There was no change to such compensation as a result of this review. | ||||||||
| 8. | Other Board Committees | |||||||||
| If the Board has standing committees other than the audit, compensation and nominating committees, identify the committees and describe their function. | In addition to the Audit Committee, the Corporate Governance Committee and the Human Resources and Compensation Committee, the Board has established an Environmental, Health and Safety Committee, which is responsible for monitoring the system of controls over workplace health and safety and protection of the environment. The Environmental, Health and Safety Committee meets periodically with Management to review safety and environmental statistics and discuss courses of action to assess the effectiveness of systems in place and personnel responsible for executing health, safety and environment programs. | |||||||||
22 INFORMATION CIRCULAR
| Governance Disclosure Guideline under | ||||||||||
| NI 58-101 | Corporate Governance Practices of the Corporation | |||||||||
| 9. | Assessments | |||||||||
| Disclose whether or not the Board, its committees and individual directors are regularly assessed with respect to their effectiveness and contribution. If assessments are regularly conducted, describe the process used for the assessments. If assessments are not regularly conducted, describe how the Board satisfies itself that the Board, its committees, and its individual directors are performing effectively. | The Corporate Governance Committee is responsible for making regular assessments of the overall performance, effectiveness and contribution of the Board and each committee, the chairman of the Board, each committee chairman and each director, and reporting on such assessments to the Board. The objective of the assessments is to ensure the continued effectiveness of the Board in the execution of its responsibilities and to contribute to a process of continuing improvement. In addition to any other matters the Corporate Governance Committee deems relevant, the assessments will consider in the case of the Board or a committee, the applicable mandate or charter, and in the case of individual directors, the applicable position descriptions, as well as the competencies and skills each individual director is expected to bring to the Board | |||||||||
OTHER INFORMATION
Interest of Certain Persons and Companies in Matters to be Acted Upon
Except as disclosed herein, Management of the Corporation is not aware of any material interest of any director or senior officer, or anyone who held office as such since the beginning of the Corporation’s last financial year, or of any associate or affiliate of any of the foregoing persons, in any matter to be acted upon at the Meeting other than the election of directors.
Interest of Informed Persons in Material Transactions
Management of the Corporation is unaware of any material interest, direct or indirect, of any “informed person” (as such term is defined in National Instrument 51-102 – Continuous Disclosure Obligations) of the Corporation, or any associate or affiliate of any such individual or of the Corporation, in any transaction since the beginning of the last completed financial year of the Corporation or any of its subsidiaries.
Additional Information
Further financial information is provided in the Corporation’s consolidated financial statements for the fiscal year ended December 31, 2005 and management’s discussion and analysis of the results thereon. Shareholders wishing to receive a copy of such materials should mail a request to the Vice President, General Counsel and Corporate Secretary of the Corporation at 10102-114 Street, Fort Saskatchewan, Alberta, T8L 3W4.
Additional information relating to the Corporation is also available free of charge on SEDAR atwww.sedar.com.
INFORMATION CIRCULAR 23
 |  | |||||
| 9th Floor, 100 University Avenue | ||||||
| Toronto, Ontario M5J 2Y1 | ||||||
| www.computershare.com |
 | MR SAM SAMPLE 123 SAMPLES STREET SAMPLETOWN SS X9X 9X9 | Security Class | 123 | |||||
| Holder Account Number | ||||||||
| C1234567890 | X X X | |||||||
| Fold |
Form of Proxy - Annual Meeting to be held on May 3, 2006
This Form of Proxy is solicited by and on behalf of Management.
Notes to proxy
| 1. | Every shareholder (“Holder”) of The Westaim Corporation (the “Corporation”) has the right to appoint some other person of their choice, who need not be a Holder, to attend and act on their behalf at the meeting. If you wish to appoint a person other than the persons whose names are printed herein, please insert the name of your chosen proxyholder in the space provided (see reverse). | ||
| 2. | If the securities are registered in the name of more than one owner (for example, joint ownership, trustees, executors, etc.), then all those registered should sign this proxy. If you are voting on behalf of a corporation or another individual you may be required to provide documentation evidencing your power to sign this proxy with signing capacity stated. | ||
| 3. | This proxy should be signed in the exact manner as the name appears on the proxy. | ||
| 4. | If this proxy is not dated, it will be deemed to bear the date on which it was mailed by management of the Corporation (“Management”) to the Holder. | ||
| 5. | The securities represented by this proxy will be voted as directed by the Holder, however, if such a direction is not made in respect of any matter, this proxy will be voted as recommended by Management. | ||
| 6. | This proxy should be read in conjunction with the accompanying documentation provided by Management. |
Proxies submitted must be received by 10:00 a.m., Eastern daylight time, on May 1, 2006.
| Fold |
VOTE USING THE TELEPHONE OR INTERNET 24 HOURS A DAY 7 DAYS A WEEK!
 |  |  |
• Call the number listed BELOW from a touch tone telephone. 1-866-732-VOTE (8683) Toll Free | • Go to the following web site: www. computershare ..com/proxy | • You can enroll to receive future securityholder communications electronically, by visiting www.computershare.com - - click “Enroll for e-delivery” under the Shareholder Services menu. |
If you vote by telephone or the Internet, DO NOT mail back this proxy.
Voting by mail may be the only method for securities held in the name of a corporation or securities being voted on behalf of another individual.
Voting by mail or by Internet are the only methods by which a holder may appoint a person as proxyholder other than the Management nominees named on the reverse of this proxy. Instead of mailing this proxy, you may choose one of the two voting methods outlined above to vote this proxy.
To vote by telephone or the Internet, you will need to provide your CONTROL NUMBER, HOLDER ACCOUNT NUMBER and ACCESS NUMBER listed below.
CONTROL NUMBER 123456 | HOLDER ACCOUNT NUMBER C1234567890 | ACCESS NUMBER 12345 | |||||
| 00B0GC | CPUQC01.E.INT/000001/il234 | ||||||
| + | MR SAM SAMPLE | C1234567890 |  | + | ||||
| XXX 123 |
Appointment of Proxyholder | ||||||
I/We being Holder(s) of common Shares of The Westaim Corporation (the “Corporation”) hereby appoint: Ian W. Delaney, or failing him Barry M. Heck, or failing him Brain D. Heck | OR | Print the name of the person you are appointing if this person is someone other than the Management Nominees listed herein. |
as my/our proxyholder with full power of substitution and to vote in accordance with the following direction including upon any ballot duly called thereat (or if no directions have been given, as the proxyholder sees fit) and all other matters that may properly come before the Annual Meeting of Shareholders of the Corporation to be held at the Jane Mallet Theatre, St. Lawrence Centre, 27 Front Street East, Toronto, Ontario, on Wednesday May 3, 2006 at 10.00 a.m., Eastern daylight time, including upon any ballot duly called thereat and at any adjournment thereof (the “Meeting”), in the same manner, to the same extent and with same powers as if the undersigned were present at the Meeting, and without limiting the general authorization given, the persons above-named are specifically directed:
| VOTING RECOMMENDATIONS ARE INDICATED BY | HIGHLIGHTED TEXT | OVER THE BOXES. |
1. Election of Directors
| The election of directors nominated by Management in the accompanying Information Circular | For | Withhold | ||||
| Vote FOR or WITHHOLD for all nominess proposed by Management | o | o | Fold | |||
2. Appointment of Auditors | For | Withhold | ||||
| Appointment of Deloitte & Touche LLP as auditors and the authorization of the board of directors of the Corporation to fix the auditors’ remuneration | o | o | ||||
The undersigned instructs the above-named proxyholder to act on each of the matters itemized above as directed.If no direction is given, such proxyholder shall vote for the election of the nominees listed in the Information Circular as directors and for the appointment of Deloitte & Touche LLP as auditors and the authorization of the directors to fix the auditors’ remuneration.The undersigned hereby confers discretionary authority upon such proxyholder to vote, in accordance with his or her best judgement, with respect to amendments or variations to the matters outlined above and with respect to matters other than those listed in the notice calling the Meeting and which may properly come before the Meeting. At the date hereof, Management knows of no such amendment, variation or other matter. | ||
Fold | ||
Authorized Signature(s) - This section must be completed for your instructions to be executed. | Signature(s) | Date | ||
I/We authorize you to act in accordance with my/our instructions set out above. I/We hereby revoke any proxy previously given with respect to the Meeting.If no voting instructions are indicated above, this Proxy will be voted as recommended by Management. | / / |
| n | 9 9 9 9 9 9 9 9 9 9 9 9 | 0 0 1111 | 9 X X | AR 0 | W E D Q | + |
OOBOHB
 | £ | Interim Financial Statements | + | |||
Rather than receiving The Westaim Corporation’s interim financial Statements by mail, you may choose to access them at www.westaim.com. Under Securities regulations, you may elect annually to receive interim financial statements by mail by checking the box above and completing and returning this card. Computershare will use the information collected solely for the mailing of such financial statements. You may view Computershare’s Privacy Code online or by requesting that they mail you a copy.
Please place my name on your financial statements mailing list.
| Name | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Apt. | Street Number | Street Name | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| City | Prov. / State | Postal Code / Zip Code | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ¢ | W E D Q | 3 Q E N | + | |||||

 | ||
| 1000013471-M5J2Y1-BR01 | ||
| COMPUTERSHARE TRUST COMPANY OF CANADA | ||
| PO BOX 19004 STN BRM B | ||
| TORONTO ON M7Y 3M4 |