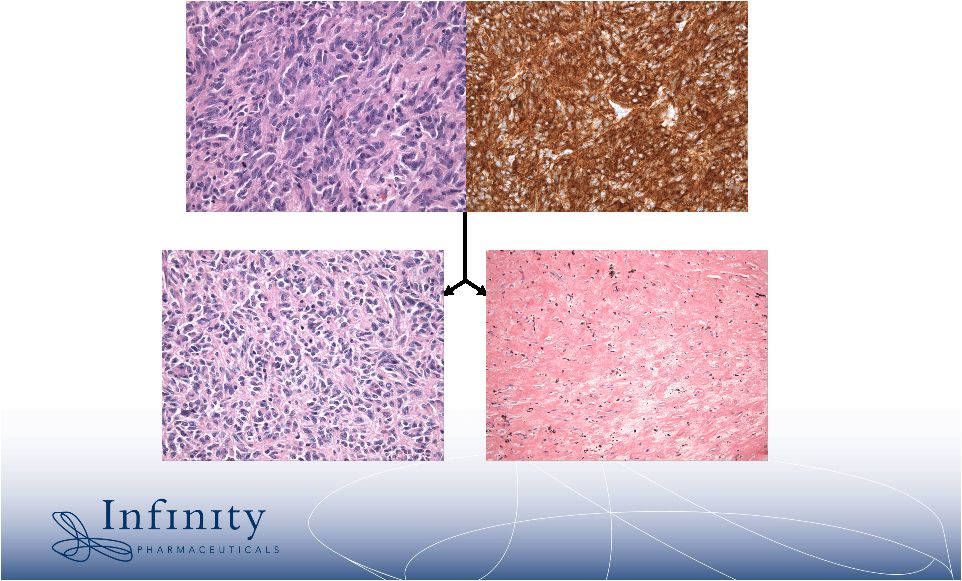
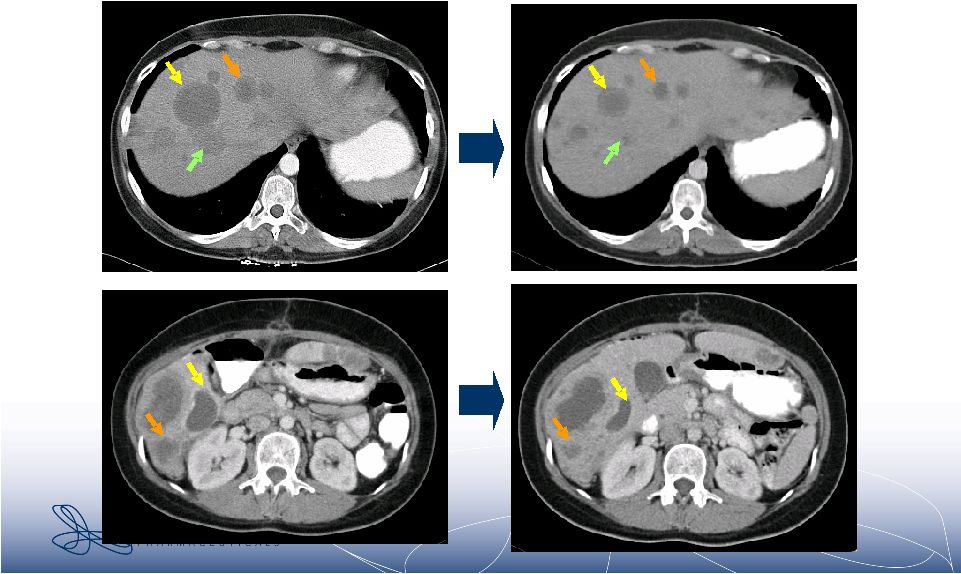
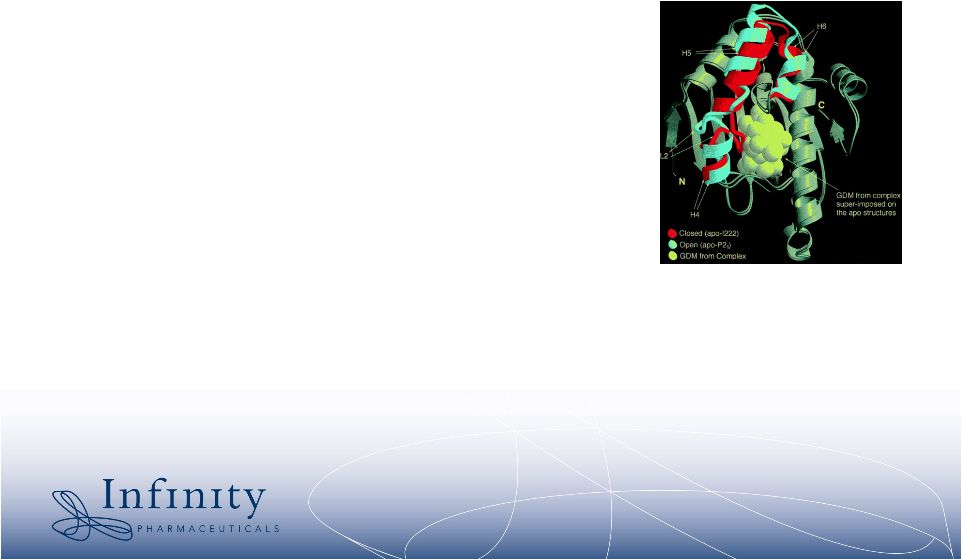
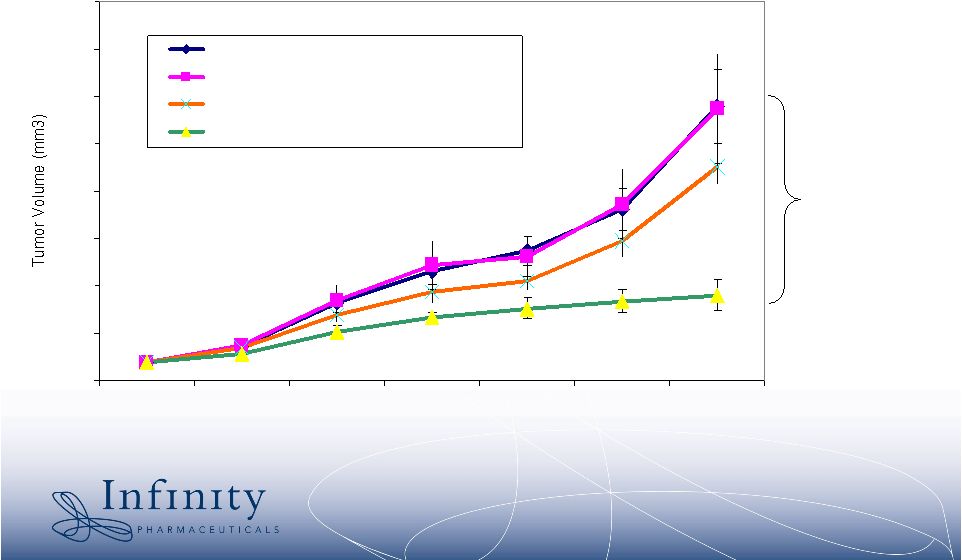
2 Forward-Looking Statements • This presentation contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. These statements involve risks and uncertainties that could cause actual results to be materially different from historical results or from any future results expressed or implied by such forward- looking statements. • Such forward-looking statements include statements regarding future clinical trial activity for IPI-504 in both intravenous and oral form; the collection of additional clinical information on IPI-504; the intended utilization and commercial potential of IPI-504; the ability to name clinical candidates in the company’s research programs; estimates of 2007 financial performance and year-end cash balance; and the expectation that Infinity will have cash to support its current operating plan through at least December 31, 2009. • Such statements are subject to numerous factors, risks and uncertainties that may cause actual events or results to differ materially from the company's current expectations. For example, there can be no guarantee that any product candidate Infinity is developing will successfully complete necessary preclinical and clinical development phases, be approved for sale in any market or that, if approved, revenue from sales of such product will reach any specific level. In particular, management's expectations could be affected by risks and uncertainties relating to: results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities and investigational review boards at clinical trial sites; Infinity’s ability to enroll patients in its clinical trials; Infinity's dependence on its collaborations with MedImmune and Novartis; Infinity's ability to obtain additional funding required to conduct its research, development and commercialization activities; unplanned cash requirements and expenditures; and Infinity's ability to obtain, maintain and enforce patent and other intellectual property protection for any products it is developing. • These and other risks which may impact management's expectations are described in greater detail under the caption "Risk Factors" included Infinity's quarterly report on Form 10-Q for the quarter ended March 31, 2007, as filed with the Securities and Exchange Commission on May 9, 2007. • Further, any forward-looking statements contained in this presentation speak only as of the date hereof, and Infinity expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise. • All trademarks used in this presentation are the property of their respective owners. |