 Infinity Overview November 12, 2008 Exhibit 99.1 |
 2 2 Forward-Looking Statements • This presentation contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. These statements involve risks and uncertainties that could cause actual results to be materially different from historical results or from any future results expressed or implied by such forward-looking statements. • Such forward-looking statements include those regarding future clinical trial activity for IPI-504, IPI-493 and IPI-926, including the completion of the planned Phase 3 clinical trial of IPI-504 in refractory GIST; the presentation of data for IPI-504; estimates of 2008 financial performance; and the expectation that Infinity will have cash to support its current operating plan into 2010. • Such statements are subject to numerous factors, risks and uncertainties that may cause actual events or results to differ materially from Infinity's current expectations. For example, there can be no guarantee that any product candidate Infinity is developing will successfully complete necessary preclinical and clinical development phases. In particular, expectations could be affected by risks and uncertainties relating to: results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities, investigational review boards at clinical trial sites, and publication review bodies; Infinity's ability to enroll patients in its clinical trials; Infinity's dependence on its collaboration with MedImmune/AstraZeneca; Infinity's ability to obtain additional funding required to conduct its research, development and commercialization activities; unplanned cash requirements and expenditures; and Infinity's ability to obtain, maintain and enforce patent and other intellectual property protection for any product candidates it is developing. • These and other risks which may impact Infinity’s expectations are described in greater detail under the caption "Risk Factors" included in Infinity's quarterly report on Form 10-Q for the quarter ended September 30, 2008, which was filed with the Securities and Exchange Commission on November 5, 2008. • Further, any forward-looking statements contained in this presentation speak only as of the date hereof, and Infinity expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise. • All trademarks used in this presentation are the property of their respective owners. |
 3 Infinity: Innovative small molecule cancer drug discovery and development • Late-stage oncology pipeline addressing areas of significant unmet medical need – Lead candidate, IPI-504 i.v. (retaspimycin hydrochloride), in Phase 3 registration study; positioned as potential first-to-market Hsp90 inhibitor – Broad near-term potential reflected in clinical development strategy • Sustainable pipeline of innovative small molecule candidates – IPI-493 and IPI-926 in clinical development – Novel discovery programs behind Hsp90 and Hedgehog • Proven management team and unparalleled culture – First- and best-in-class NDAs, value-creating business transactions – Strong balance sheet; resources support current operating plan into 2010 |
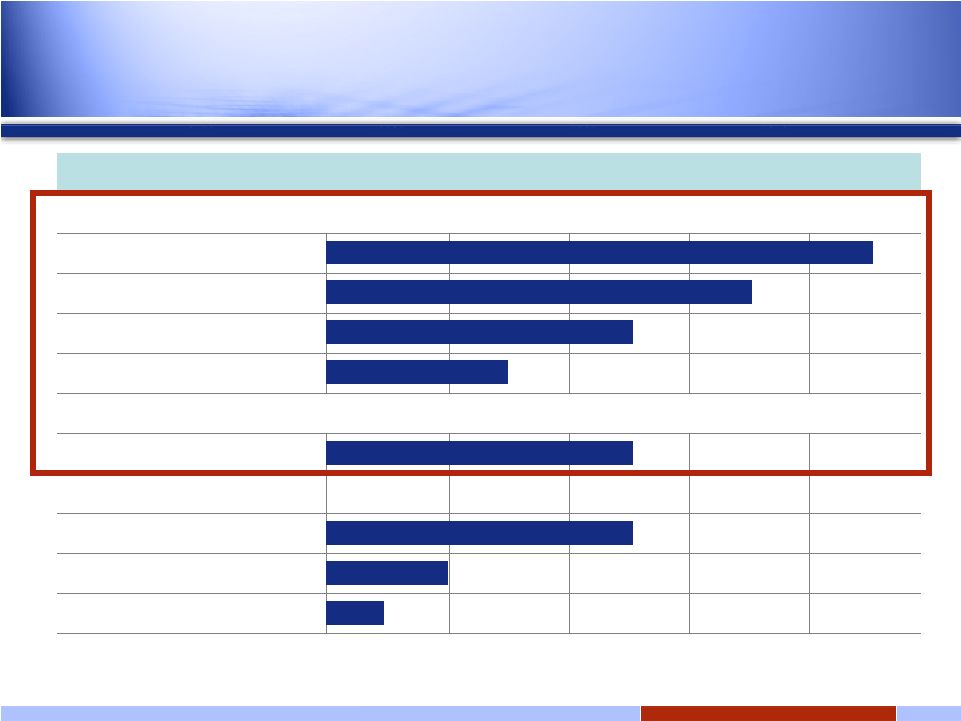 4 4 Solid tumors Hsp90 oral: IPI-493 Discovery Preclinical Phase 1 Phase 2 Phase 3 Hsp90 i.v.: IPI-504 (retaspimycin hydrochloride) GIST NSCLC Docetaxel Combo Add’l Solid Tumor Solid tumors Hedgehog Pathway: IPI-926 Bcl-2/Bcl-xL* Discovery Programs RING trial INFI: Late-stage oncology pipeline *Transitioned to Novartis February 2008 Expanded Phase 2 |
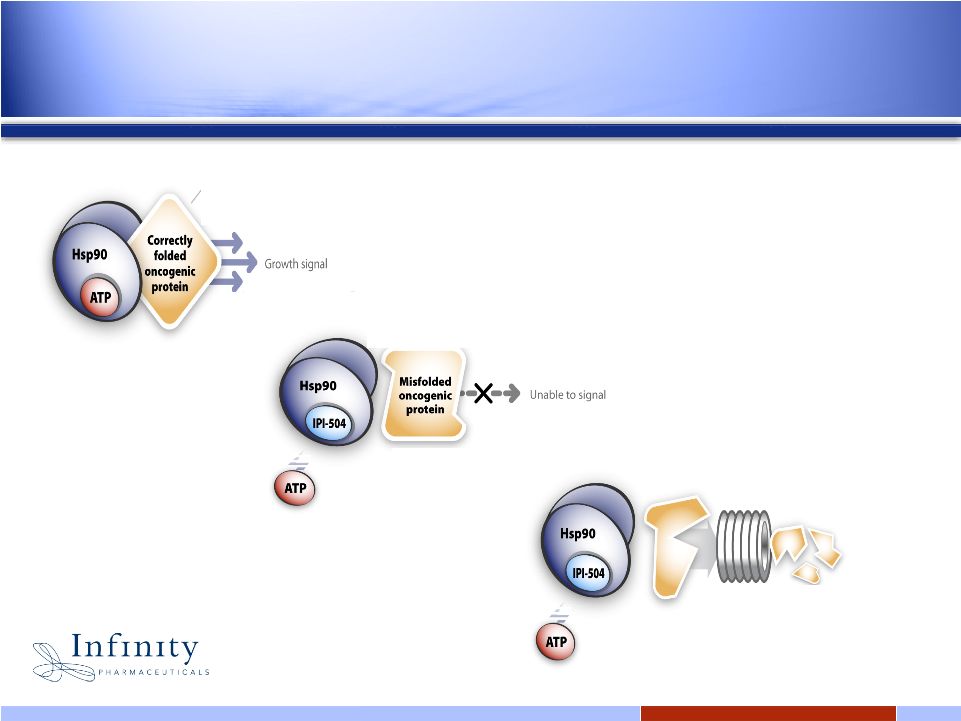 5 5 Hsp90 Inhibition: How it works Hsp90 is a “chaperone” protein responsible for supporting and stabilizing numerous oncogenic proteins IPI-504 binds at ATP site and inhibits proper folding Unstable oncogene degraded; tumor growth inhibited |
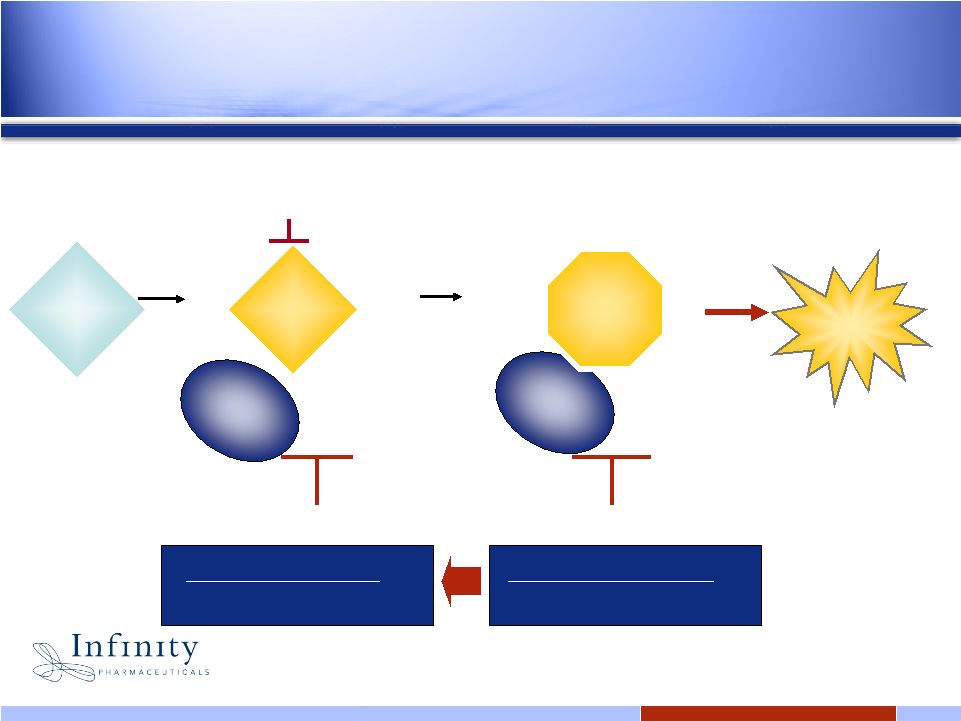 6 6 Oncogenic protein drives cancer cell survival & growth Cancer cell death Tyrosine kinase inhibitor (e.g., Gleevec, Tarceva) Normal protein Resistance mutations evade TKI therapy IPI-504 / IPI-493 (oral) IPI-504 Dependent on Hsp90 for function Still depends on Hsp90 for function Rapid registration in refractory setting Expand potential in front-line / combo Hsp90 inhibition: A new treatment approach |
 7 Cancer(s) GIST NSCLC Breast (HER2+) CML AML c-Kit EGFR / C-met HER2 / ER Bcr-Abl Flt3 Hsp90 client proteins Broad applicability of client protein rationale |
 8 Phase 1 results from ASCO 2008 reported activity and safety of IPI-504 in refractory GIST • 70% overall disease control after 2 cycles (6 weeks) in patients with GIST • Estimated median Progression Free Survival (PFS) of 12 weeks • Generally well-tolerated with an acceptable safety profile GIST Other STS Number of patients 1 36 11 Partial Response (PR) 2 1 ( 3%) 1 ( 9%) Stable Disease (SD) 24 (67%) 4 (36%) Progressive Disease (PD) 11 (30%) 6 (55%) 1. 90-400 mg/m2 - twice weekly, 2 weeks on 1 week off 2. PRs confirmed Wagner et al., ASCO 2008 |
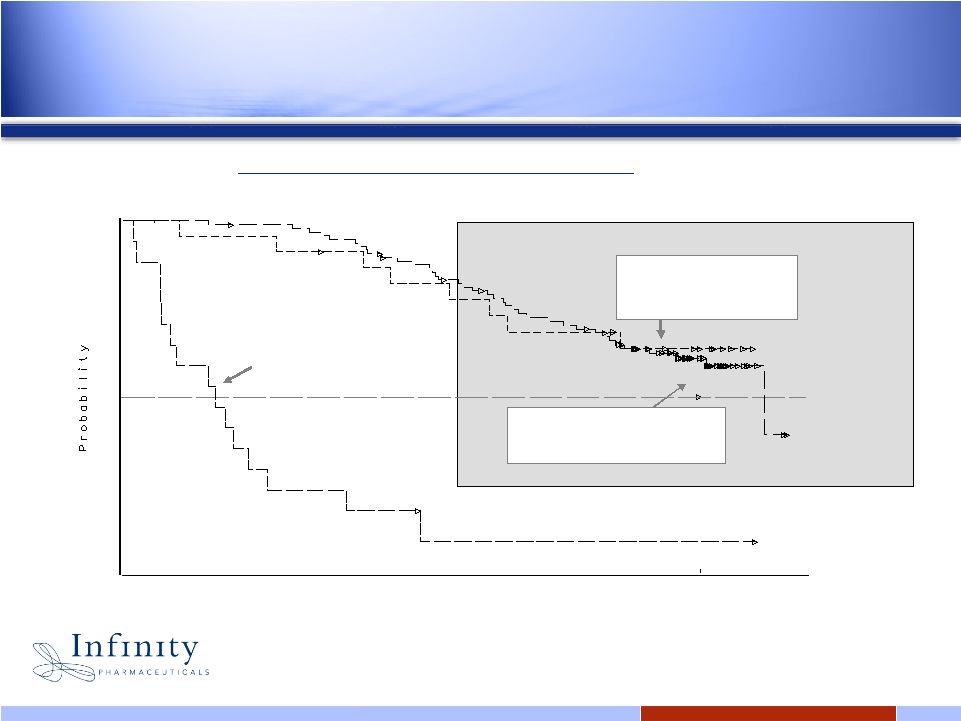 9 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Weeks Post First Dose 0 12 24 36 48 60 72 84 96 108 120 132 144 156 168 180 192 204 216 228 240 252 264 PROGRESSION On Imatinib Median 36 wks Overall Survival by Best Response Achieved (Kaplan Meier Estimate) STABLE DISEASE PARTIAL RESPONSE 1 Blanke, CD, et al. J Clin Onc 2006 (ASCO Proceedings) Part I, Vol 24; No. 18S: 9528. Stable disease is important in GIST 1 Both SD and PR predict survival |
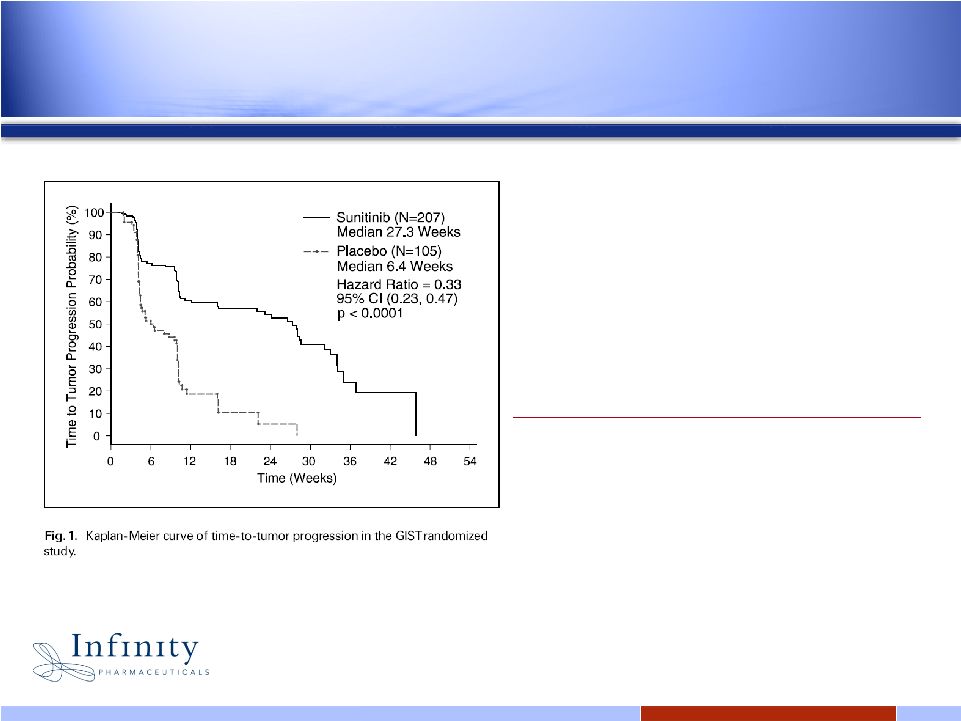 10 10 Stable disease for >6 weeks is meaningful in the refractory GIST setting 1 Results from the Phase 3 Sutent ® trial in GIST TTP on Placebo: 6.4 weeks TTP on Sutent ® : 27.3 weeks Response Rate on Sutent ® : 7% 1 Goodman VL, Rock EP, Dagher R, Ramchandani RP, Abraham S, Gobburu JVS, Booth BP, Verbois SL, Morse DL, Liang CY, Chidambaram N, Jiang JX, Tang S, Mahjoob K, Justice R, Pazdur R. Approval summary: sunitinib for the treatment of imatinib refractory or intolerant gastrointestinal stromal tumors and advanced renal cell carcinoma. Clin Cancer Res (2007) 13:1367-1373. |
 11 First Phase 3 registration study: RING trial Retaspimycin hydrochloride in GIST Trial under Special Protocol Assessment and EMEA advice • Study designed in consultation with international group of KOLs – Randomized, double-blind, placebo-controlled study – 90% power to detect a hazard ratio of 0.57, with an alpha of 0.05 – Approx. 200 patients; >50 sites worldwide – Patients with progressive GIST despite treatment with imatinib and sunitinib • Primary endpoint: Progression-free survival (PFS) – Secondary endpoints: disease control rate, time to progression & overall survival • Efficient trial design – Trial opened in 3Q08; enrollment commenced in October 2008 – Trial anticipated to be completed by year end 2010 11 |
 12 Expanding potential of IPI-504 beyond GIST • Expanding Phase 2 study in Stage IIIb/IV NSCLC – Study designed to evaluate 2 equal groups: mEGFR and wild-type EGFR • Potential to expand if 1 patient demonstrates clinical benefit – Enrolled 10 patients in each group • >1 patient in each arm met expansion criterion of extended stable disease – Expanding study to enroll additional 19 patients in each arm – Full Phase 2 data anticipated by mid-2009 • Phase 1b combo with docetaxel – Patients with advanced solid tumors – Establish safety, MTD, and optimal schedule of administration • Additional clinical studies of IPI-504 planned for 2009 12 |
 13 Oncogenic protein drives cancer cell survival & growth Cancer cell death Tyrosine kinase inhibitor (e.g., Gleevec, Tarceva) Normal protein Resistance mutations evade TKI therapy IPI-504 / IPI-493 (oral) IPI-504 Dependent on Hsp90 for function Still depends on Hsp90 for function Rapid registration in refractory setting Expand potential in front-line / combo Targeted anti-chaperone therapy: A new treatment approach |
 14 Emerging generation of oral anti-cancer agents driving billions of dollars of new sales • Gleevec • Sprycel • Tasigna • Torisel • Sutent • Nexavar • Xeloda • Tarceva • Iressa • Tykerb • More…. |
 15 15 Infinity’s second Hsp90 inhibitor, oral IPI-493, enters clinical development • Phase 1 clinical trial in patients with advanced solid tumors – Dose-escalation study evaluating safety and tolerability – Identify dose and schedule for further clinical development – Assess biological activity using RECIST and disease-specific markers • Novel, orally delivered formulation of 17-AG • Strong pharmaceutical properties in vivo and in vitro – Oral availability – Selectivity for cancer cells over normal cells – Potent and selective Hsp90 inhibition |
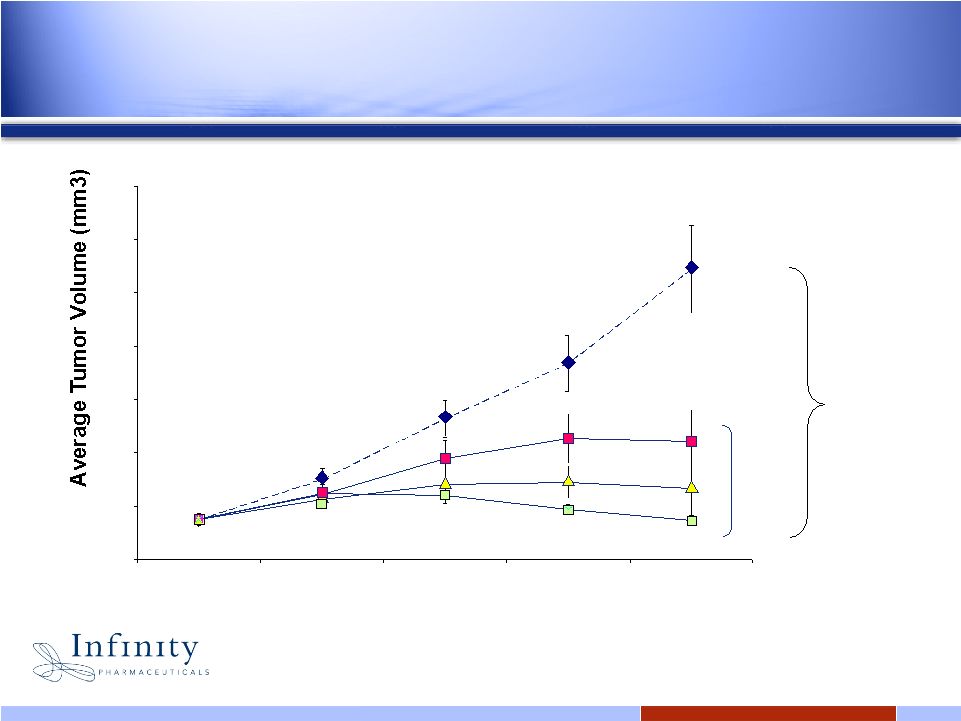 16 Significant anti-tumor activity with oral IPI-493 in preclinical model of TKI-resistant NSCLC 0 500 1000 1500 2000 2500 3000 3500 18 21 25 28 32 Days Post Implant Vehicle 50 75 100 IPI-493 (mg/kg) Dosing: QOD, PO, 10ml/kg 87% 0 500 1000 1500 2000 2500 3000 3500 18 21 25 28 32 Days Post Implant Vehicle 50 75 100 IPI-493 (mg/kg) 0 500 1000 1500 2000 2500 3000 3500 18 21 25 28 32 Days Post Implant Vehicle 50 75 100 IPI-493 (mg/kg) Dosing: QOD, PO, 10ml/kg 87% Lee et al., 2008 EORTC |
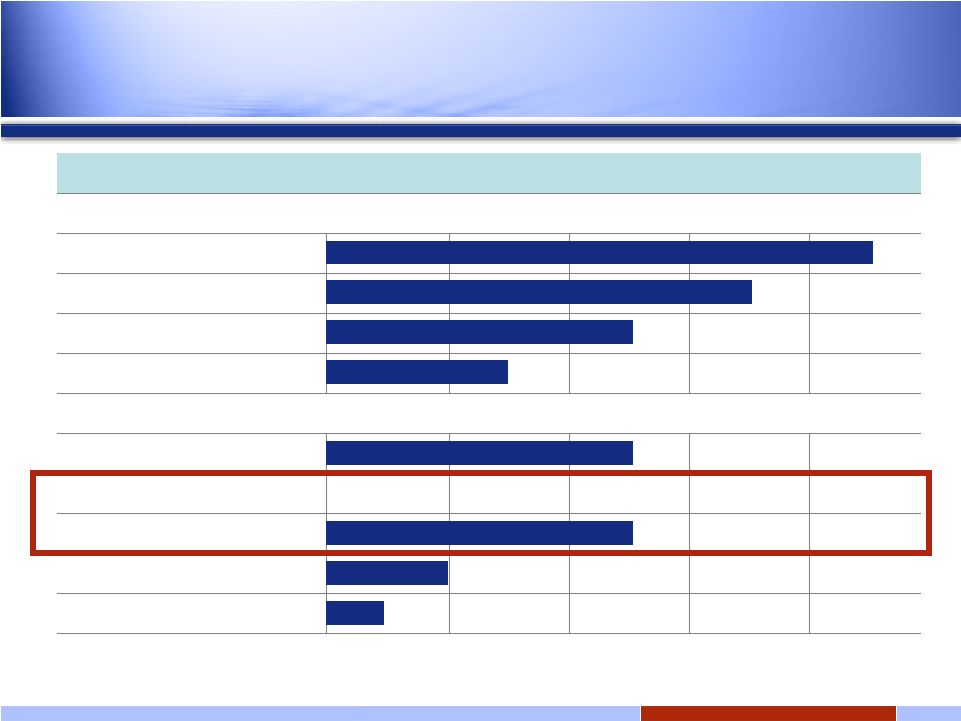 17 17 Solid tumors Hsp90 oral: IPI-493 Discovery Preclinical Phase 1 Phase 2 Phase 3 Hsp90 i.v.: IPI-504 (retaspimycin hydrochloride) GIST NSCLC Docetaxel Combo Add’l Solid Tumor Solid tumors Hedgehog Pathway: IPI-926 Bcl-2/Bcl-xL* Discovery Programs RING trial INFI: Late-stage oncology pipeline *Transitioned to Novartis February 2008 Expanded Phase 2 |
 18 Genetic mutation medulloblastoma basal cell carcinoma Pathway activation pancreatic, gastric, prostate, glioma, SCLC Tumor progenitor cells SCLC, glioblastoma, breast, multiple myeloma Hedgehog pathway implicated in a variety of cancers |
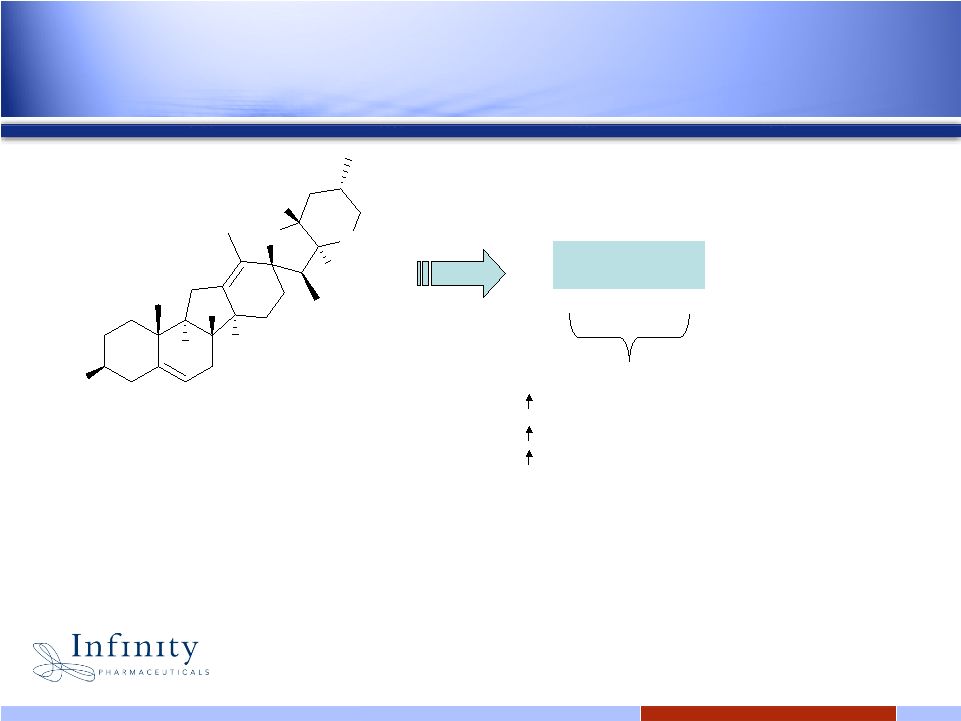 19 Infinity’s Hedgehog inhibitor: IPI-926 H O O N H H H H H H Potency (15nM in cell based assay) Selectivity Metabolic stability IPI-926 Oral bioavailability Long half-life In vivo activity cyclopamine • Issued composition of matter patent • Infinity-discovered & 100% owned royalty-free worldwide |
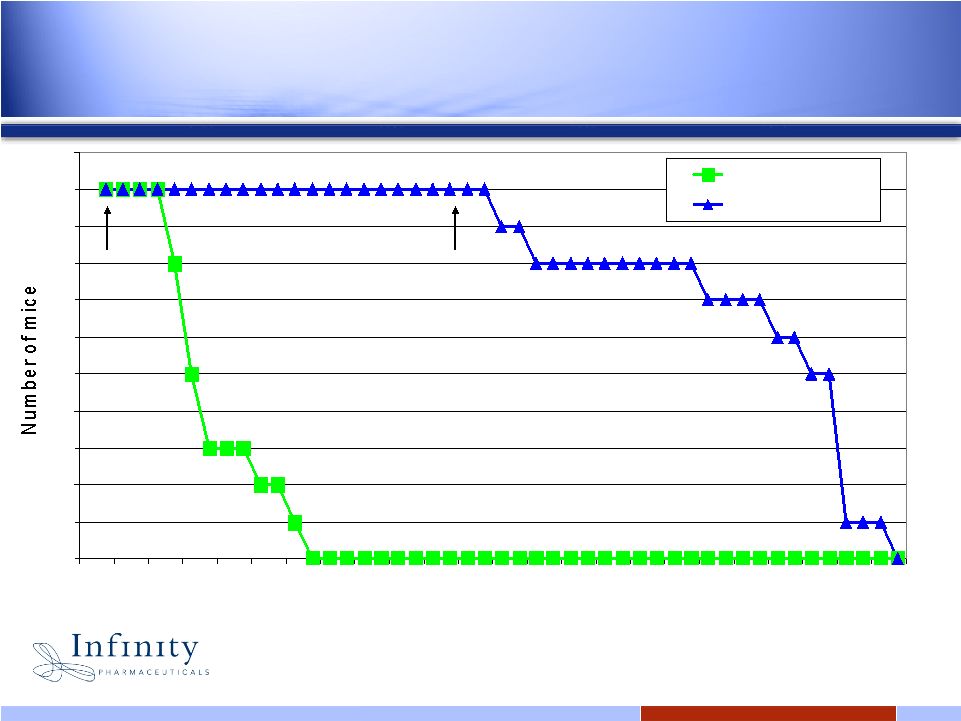 20 20 Once-daily administration of IPI-926 resulted in 100% survival during treatment period 0 1 2 3 4 5 6 7 8 9 10 11 10 14 18 22 26 30 34 38 42 46 50 Days post implant Veh IPI-926 @ 40mg/kg Start of TX End of TX Mouse model, orthotopically implanted B837Tx medulloblastoma tumor Pink et al., 2008 AACR |
 21 E/P - IPI-926 E/P -Vehicle Vehicle IPI-926 Days Post Implant Mice randomized +/- IPI-926 Last day of E/P treatment 5 weeks total of IPI-926 follow-up treatment; 40 mg/kg, PO QD 82% IPI-926 significantly delays tumor re-growth following tumor debulking in SCLC model Travaglione et al., 2008 AACR |
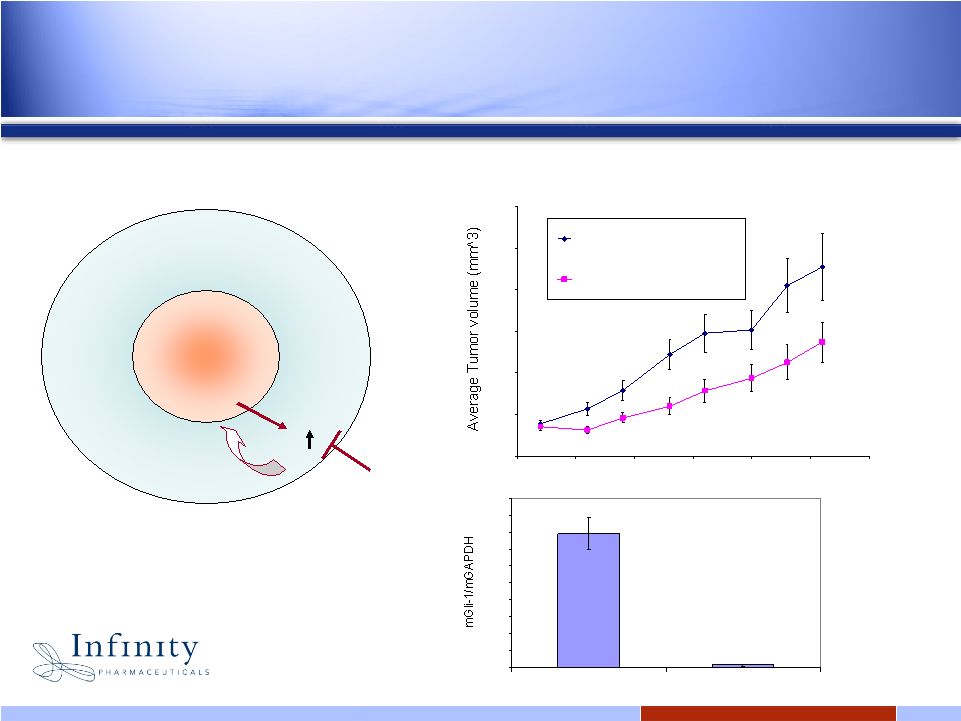 22 0.00 10.00 20.00 30.00 40.00 50.00 60.00 70.00 80.00 90.00 100.00 Vehicle IPI-926 @ 40mpk 0.00 200.00 400.00 600.00 800.00 1000.00 1200.00 40 45 50 55 60 65 70 Days (post implant) Vehicle: 30% HPBCD N=13 IPI5926 @ 40mg/kg N=12 IPI-926: 40% IPI-926 demonstrates significant tumor growth inhibition in pancreatic cancer model IPI-926 down-regulates Hedgehog signal in tumor stroma IPI-926 Tumor Surrounding stromal tissue SHH Gli1 Travaglione et al., 2008 EORTC |
 23 23 Business Update |
 24 24 Strong balance sheet and cash runway • Well-capitalized – Started 2008 with $114M cash and investments – $80.8M cash and investments at 9/30/08 • Projected year end cash of approximately $80 million – Low end of previous net cash burn guidance of $35-$45 million for 2008 • Burn rate well-controlled • Cash to support current plan into 2010 – Sufficient capital to achieve key value-creation milestones |
 25 Executing on key 2008 R&D objectives Initiate IPI-493 Phase 1 clinical trial Preliminary update on IPI-504 Phase 2 data in NSCLC Initiate RING Ph 3 registration trial of IPI-504 in GIST Secure SPA for IPI-504 Phase 3 GIST study Present IPI-504 Phase 1 GIST data (ASCO) IPI-504 & IPI-493: Hsp90 inhibitors Initiate Phase 1 clinical study Present preclinical data (AACR) IPI-926: Hedgehog pathway inhibitor |
 26 Infinity Leadership Michael Curtis, Ph.D., VP Pharm. Devel. TKT, Syntonix, Genzyme, Bristol-Myers Squibb David Grayzel, M.D., VP Clin Dev. & Med. Affairs Dyax, Mass General Hospital John Keilty, Sr Dir Informatics Millennium, UMass Medical School Vito Palombella, Ph.D., VP Drug Discovery Syntonix, Millennium, ProScript Steven Kafka, Ph.D., VP Finance Millennium, Strategic Decisions Group Jeanette Kohlbrenner, Sr Dir Human Resources Genetics Institute, Syntonix Gerald Quirk, Esq., VP & General Counsel Genzyme, Palmer & Dodge Jeffrey Tong, Ph.D., VP Corp. & Product Dev. McKinsey & Co, Harvard Ctr. for Genomics Research Steven Holtzman Chair & CEO Millennium, DNX Julian Adams, Ph.D. President of R&D & CSO Millennium, ProScript, Boehringer Ingelheim, Merck Adelene Perkins President & CBO Transform, Genetics Institute, Bain, GE |
 27 Infinity: Innovative small molecule cancer drug discovery and development • Late-stage oncology pipeline addressing areas of significant unmet medical need – Lead candidate, IPI-504 i.v. (retaspimycin hydrochloride), in Phase 3 registration study; positioned as potential first-to-market Hsp90 inhibitor – Broad near-term potential reflected in clinical development strategy • Sustainable pipeline of innovative small molecule candidates – IPI-493 and IPI-926 in clinical development – Novel discovery programs behind Hsp90 and Hedgehog • Proven management team and unparalleled culture – First- and best-in-class NDAs, value-creating business transactions – Strong balance sheet; resources support current operating plan into 2010 |
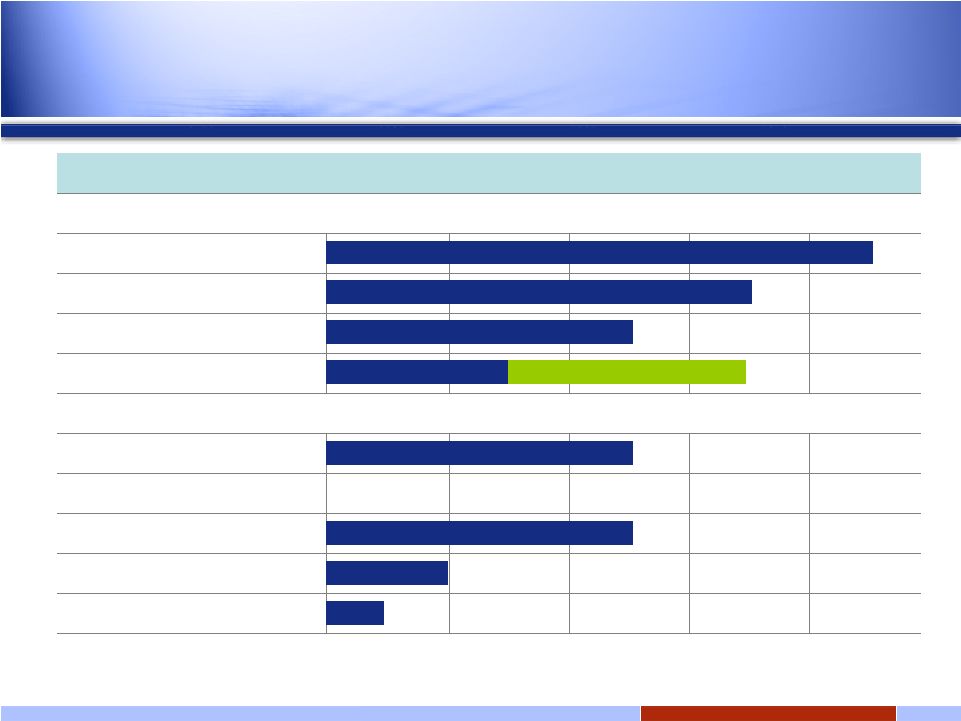 28 28 Solid tumors Hsp90 oral: IPI-493 Discovery Preclinical Phase 1 Phase 2 Phase 3 Hsp90 i.v.: IPI-504 (retaspimycin hydrochloride) GIST NSCLC Docetaxel Combo Add’l Solid Tumor Solid tumors Hedgehog Pathway: IPI-926 Bcl-2/Bcl-xL* Discovery Programs RING trial INFI: Late-stage oncology pipeline *Transitioned to Novartis February 2008 Expanded Phase 2 In 2009 |