Exhibit 99.3
A LATE-STAGE CLINICAL BIOPHARMACEUTICAL COMPANY
ÆTERNA ZENTARIS
ANNUAL REPORT 2006
COMPANY STRENGTHS
Depth |
| Deep and focused pipeline at all stages of development |
Breadth |
| Ability to penetrate large and growing markets |
Execution |
| Successful achievement of clinical and corporate milestones |
ÆTERNA ZENTARIS INC.
(TSX: AEZ, NASDAQ: AEZS)
Please note that all amounts are in US dollars
ÆTERNA ZENTARIS INC. IS A LATE-STAGE, PURE PLAY GLOBAL BIOPHARMACEUTICAL COMPANY FOCUSED ON ENDOCRINE THERAPY AND ONCOLOGY, WITH PROVEN EXPERTISE IN DRUG DISCOVERY, DEVELOPMENT AND COMMERCIALIZATION
In 2006, Æterna Zentaris delivered solid results
Late-stage programs
CETRORELIX
· Acceptance by FDA of IND for Phase 3 program in benign prostatic hyperplasia (BPH)
· Launch of Cetrotide® (cetrorelix) in Japan for in vitro fertilization
· Regained exclusive worldwide (ex-Japan) rights for cetrorelix in BPH
OZARELIX
· Positive Phase 2 results in BPH Initiation of Phase 2b trial
· Positive Phase 2 results in prostate cancer Initiation of Phase 2b trial
· Exclusive license granted to Nippon Kayaku for ozarelix in oncology in Japan
PERIFOSINE
· Positive interim Phase 2 data in advanced renal cell carcinoma
· Positive Phase 2 results in multiple cancers
· Initiation of multiple Phase 1 and Phase 2 trials in cancer
EARLIER-STAGE PROGRAMS
AN-152
· Positive top line Phase 1 results for breast and gynaecological cancers
ZEN-012
· Acceptance by FDA of IND for Phase 1 trial in solid tumors and lymphoma
PURE PLAY CORPORATE STRATEGY
Æterna Zentaris spins off subsidiary Atrium and emerges as a pure play biopharmaceutical company
· Secondary offering ensuring financial stability to execute business plan
· Distribution of remaining Atrium shares to Æterna Zentaris shareholders
|
|
|
|
Gilles Gagnon, MSc, MBA |
| Eric Dupont, PhD |
|
MESSAGE TO SHAREHOLDERS
In 2006, the progress of our lead drug candidates through advanced clinical trials and the spin-off of our subsidiary Atrium Biotechnologies enabled us to successfully achieve our goal of emerging as a latestage, pure play biopharmaceutical company. Reaching this milestone in our evolution represents a fulfillment of both our drug development and corporate strategies that we have tediously executed for many years.
Driven by positive clinical results, we are aggressively moving two product candidates through late-stage trials. In early clinical and preclinical development, we are targeting several extremely promising compounds with high future potential. In our library of 120,000 proprietary molecules resides the currency of a fruitful pipeline for many years to come. In addition, and perhaps most critically, the members of Æterna Zentaris’ management have collectively participated during their careers in the development and launch of over twenty drugs; this resource of talent powerfully distinguishes the Company in a marketplace that rewards depth of experience.
UNLOCKING THE FULL VALUE OF THE COMPANY
While very encouraging data resulted from our clinical trials in 2006, conditions grew increasingly ripe for Æterna Zentaris to become a pure play company. Accordingly, in October of 2006, we sold 24 % of our equity in our subsidiary, Atrium Biotechnologies, raising nearly $45 million through a secondary offering. Subsequent to year end, the Company distributed the remainder of our equity in Atrium to Æterna Zentaris shareholders as a return on their investment — with that, we emerged as a pure play biopharmaceutical company.
Since 1991 our ownership of Atrium — a leading developer, manufacturer and marketer of value-added products for the cosmetics, pharmaceutical, chemical and nutrition industries — had served us well as a strong financial leverage. With our pipeline carrying us closer to major breakthroughs, the route that promises optimal benefit became clear. By divesting our stake in Atrium, we began unlocking the full value of our pipeline. Æterna Zentaris is now devoted exclusively to discovering, developing and marketing biopharmaceutical products with a focus on endocrine therapy and oncology.
DELIVERING CLINICAL RESULTS
CETRORELIX
In 2006, our flagship product candidate, cetrorelix, reached a pivotal evolutionary milestone in the history of Æterna Zentaris. After a successful end-of-Phase 2 meeting with the FDA, we received approval to file for a Phase 3 program targeting benign prostatic hyperplasia (BPH) and as we closed the year, we initiated the first study of this extensive Phase 3 program.
The launch of our extensive 1,500 patient Phase 3 program in BPH with cetrorelix brings us yet another step closer to bringing this compound to market. Importantly, we have all of the resources to advance cetrorelix in BPH on our own through to an NDA submission. We are very excited about the fact that cetrorelix has not only the potential to conveniently, safely and effectively treat men who suffer from BPH, but also create tremendous value to our shareholders. The global commercial opportunity in treating BPH cannot be overstated as it represents a market that exceeds $4 billion.
Furthermore, we achieved another milestone by launching cetrorelix - under the brandname Cetrotide® - on the Japanese market through our partner Shionogi for in vitro fertilization. Cetrotide®, the first LHRH antagonist to be marketed in Japan for this indication, has been on the market since 1999. Again, this demonstrates our capacity to bring therapies to market for conditions affecting millions of people.
OZARELIX
Clinical trial results over the past year for our second LHRH antagonist lead compound, ozarelix, have been similarly exciting and could have a very significant commercial outcome.
In October, with our partner Spectrum Pharmaceuticals, we disclosed highly statistically significant Phase 2 results evidencing the alleviation of BPH clinical symptoms. Ozarelix also showed an excellent safety profile with no serious side effects. Following these positive results, we expanded the program to a Phase 2b trial. With both cetrorelix and ozarelix in late-stage clinical trials, we are now leading the LHRH antagonist class in the development of treatments for BPH
In August, we announced positive Phase 2 results for this fourth generation LHRH antagonist in hormonedependent, inoperable prostate cancer. The trial was conducted in Europe again in collaboration with Spectrum. The results confirmed the mechanism of action of our LHRH antagonist approach, showed that ozarelix provides a unique and rapid onset of action, and demonstrated that ozarelix holds promise for the treatment of other hormonal-dependent cancers. We then expanded the program in prostate cancer to a Phase 2b trial to further verify and optimize our findings, and expect results from this trial in 2007.
Further validation of the potential of ozarelix in cancer came with a licensing and collaboration agreement with Nippon Kayaku, our Japanese partner. We granted Nippon Kayaku the exclusive rights to develop and commercialize ozarelix for oncology indications in Japan.
PERIFOSINE
Our third promising compound, perifosine, has shown positive Phase 1 and Phase 2 data for the treatment of patients suffering from different forms of cancer. Along with our partner Keryx Biopharmaceuticals, we are pursuing multiple Phase 2 trials in cancer with perifosine as a single agent or in combination with other treatments for which results will be disclosed throughout the year. Additional Phase 2 trials in cancer are planned over the next twelve months. Furthermore, we expect to complete enrollment of patients for our own Phase 2 trial with perifosine in non-small cell lung cancer in combination with radiotherapy in the upcoming months. The year ahead could contain some very exciting announcements from these trials as perifosine`s unique mechanism of action allows for a number of anti-cancer treatment opportunities in monotherapy as well as in combination therapy.
TARGETING EARLIER-STAGE COMPOUNDS WITH HIGH POTENTIAL
Our drug development strategy is also aimed at establishing a risk-adverse profile by targeting earlierstage programs with high potential. In line with this strategy, we reported top line positive Phase 1 results with our LHRH specific cytotoxic conjugate, AN-152, for ovarian, breast and endometrial cancers. These results obtained with this novel approach lend further credibility to our very promising oncology platform while also enabling our Company to step into the era of personalized medicine. Indeed, by targeting patients suffering from ovarian and endometrial cancers with confirmed presence of LHRH receptors, we are increasing the probabilities of bringing AN-152 specifically to the tumor, therefore increasing our chances of success for our Phase 2 program which we expect to launch later this year in these indications. We also initiated a Phase 1 trial for solid tumors with ZEN-012, a new small molecule. We believe this oral compound has the potential to be a novel, promising multi-targeted intermittent cancer therapy and look forward to further developments in the clinic this year.
MOVING FORWARD AS A LATE-STAGE COMPANY
With two product candidates slated to be in Phase 3 for the year ahead, Æterna Zentaris begins a new era as a late-stage pure play biopharmaceutical company. With $60 million in cash and no significant long-term debt, a prudent risk management approach, as well as a new focused drug development strategy, we are now in an even better position to execute our highly focused business plan as the future of Æterna Zentaris could prove quite exciting.
Permit us to take this opportunity to recognize all members of the Æterna Zentaris team for their dedication to achieving the Company’s objectives, and to thank our shareholders for their support and continuing confidence. We look forward to continue to deliver clinical results and report important commercial developments to you in the year ahead.

Gilles Gagnon, MSc, MBA
President & CEO

Eric Dupont, PhD
Executive Chairman of the Board
Aggressively advance cetrorelix in BPH
· Flagship product candidate with significant near-term potential
· Retain all rights in BPH and fully support development through to NDA filing in 2009
Focus on further development of ozarelix and perifosine with current partners to ensure continued development success
A NEW FOCUSED DRUG DEVELOPMENT STRATEGY
Establish risk-adverse profile targeting earlier-stage compounds with high potential for aggressive development
Build solid endocrinology and oncology franchises
EMERGE AS A FULLY-INTEGRATED, GLOBAL SPECIALIST-DRIVEN BIOPHARMACEUTICAL COMPANY WITH A STRATEGIC FOCUS ON ENDOCRINE THERAPY AND ONCOLOGY
ADVANCING THE PIPELINE
A robust pipeline from drug discovery to marketed products
PRECLINICAL PROGRAMS
LHRH peptidomimetics
Erucyl-PC
Ghrelin antagonists
Erk/PI3K inhibitors
DRUG DISCOVERY
120,000 compounds
DRUG PIPELINE
(cetrorelix) CETROTIDE®
In vitro fertilization (worldwide except Japan)
(cetrorelix) CETROTIDE®
In vitro fertilization (Japan)
IMPAVIDO®
Leishmaniasis (black fever)
CETRORELIX
Benign prostatic hyperplasia
BPH
CETRORELIX
endometriosis*
* pivotal program encompassing several trials
OZARELIX
BPH
OZARELIX
Prostate cancer
PERIFOSINE
Combined with chemotherapy and biological agents for multiple cancers
PERIFOSINE
Combined with radiotherapy for non-small cell lung cancer
AN-152
Breast and endometrial cancer
ZEN-012
multiple cancers
EP-1572
growth disorders
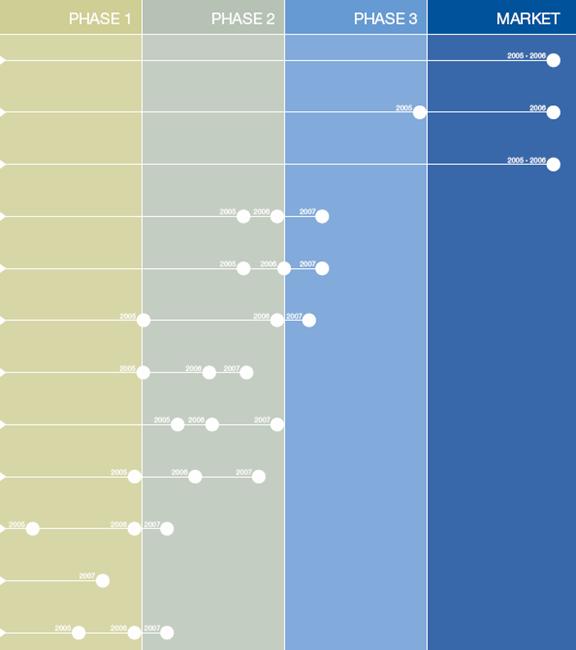
Robust Pipeline with Significant Market Opportunities
Focused and Aggressive Development Strategy with Late-Stage Compounds
KEY INVESTMENT CONSIDERATIONS
Solid Financial Position
Near-Term Catalysts Highlight Potential for Value Creation
Strong Pharma Team with Experience in Launching 20+ Compounds
BUILDING SOLID ENDOCRINOLOGY AND ONCOLOGY FRANCHISES
THREE VALUE-DRIVING LEAD COMPOUNDS IN LARGE MARKET OPPORTUNITIES
CETRORELIX
Cetrorelix is part of Æterna Zentaris’ luteinizing hormone-releasing hormone (LHRH) antagonist therapeutic approach. This peptide-based active substance was developed by the Company in cooperation with Nobel Prize winner Professor Andrew Schally of Tulane University in New Orleans (now at U.S. Veterans Administration and University of Miami).
INDICATIONS |
| IN VITRO FERTILIZATION (IVF) HYPERPLASIA (BPH) |
| BENIGN PROSTATIC |
| ENDOMETRIOSIS |
STATUS |
| · Marketed worldwide under brandname Cetrotide® |
| · Phase 3 program |
| · Pivotal program |
PARTNERSHIPS |
| · Merck Serono — Worldwide |
| · Shionogi - Japan |
| · Solvay — Worldwide (ex-Japan) Shionogi — Japan |
|
| · Shionogi — Japan |
|
|
|
|
MARKET |
| · Average cost of an IVF treatment cycle is $12,400 |
| · Market size of over $4 billion |
| · Market size of $1 billion |
|
| · Affects 10% to 15% of couples in the U.S. |
| · Affects more than 50% of men 60 years and over |
| · Under-diagnosed |
|
| · About 5 million couples in the U.S. |
| · Approximately 56 million cases in the U.S., Europe and Japan |
| · Affects 10% to 20% of women of reproductive age |
|
|
|
|
|
| · About 26 million cases in the U.S., Europe and Japan |
|
|
|
|
|
|
|
|
| Source: Barton H. Hamilton and Brian McManus September 04 |
| Source: Decision Resources September 05 |
| Source: Datamonitor July 04 |
|
|
|
|
|
|
|
DIFFERENTIATION |
| · Leading the LHRH antagonist class in the development of treatments for BPH | ||||
|
| · Dose-dependent hormonal suppression | ||||
|
| · Extensive safety database — more than 270,000 patients treated | ||||
|
| · Low side-effect profile | ||||
OZARELIX
Ozarelix is a fourth generation luteinizing hormone-releasing hormone (LHRH) antagonist administered as a depot formulation for the treatment of benign and malignant hormone-dependent diseases.
INDICATIONS | BENIGN PROSTATIC HYPERPLASIA (BPH) | PROSTATE CANCER |
STATUS | · Phase 2b | · Phase 2b |
PARTNERSHIPS | · Spectrum — North America, India | · Spectrum — North America, India Nippon Kayaku — Japan |
MARKET | · Market size of over $4 billion | · Market size of $3.1 billion |
| · Affects more than 50% of men 60 years and over | · Approximately 395,000 new cases in the U.S., Europe and Japan |
| · Approximately 56 million cases in the U.S., Europe and Japan |
|
|
|
|
| Source: Decision Resources | Source: Decision Resources |
|
|
|
DIFFERENTIATION | · Fourth generation LHRH antagonist | |
| · Dose-dependent hormonal suppression | |
| · Low side-effect profile | |
| · Treats benign to malignant indications | |
PERIFOSINE
Perifosine is a novel, first-in-class, oral anti-cancer agent that modulates several key signal transduction pathways, including Akt, MAPK, and Jnk that have been shown to be critical for the survival of cancer cells. Perifosine is currently being studied as a single agent and in combination with several forms of anti-cancer treatments for various types of cancer.
INDICATIONS |
| MULTIPLE CANCERS |
|
|
STATUS |
| · 10+ ongoing Phase 1 and Phase 2 trials |
|
|
PARTNERSHIPS |
| · Keryx — U.S., Canada, Mexico |
|
|
MARKET |
| Examples of different cancer types |
|
|
|
| · Non-small cell lung cancer |
| · Multiple Myeloma (a form of blood cancer) |
|
| · Market size of $2.4 billion |
| · Market size of $1 billion in the U.S. and U.K. |
|
| · Most common and deadly form of cancer |
| · Approximately 53,000 new cases in the U.S., Europe and Japan |
|
| · About 25% of all cancer deaths in women / 30% in men |
|
|
|
| · About 417,000 new cases in 2004 in the U.S., Europe and Japan |
|
|
|
|
|
|
|
|
| Source: Datamonitor |
| Source: Globocan |
|
|
|
|
|
DIFFERENTIATION |
| · Novel, first-in-class oral anti-cancer agent | ||
|
| · Exciting and promising targeted molecular approach | ||
|
| · Validated oncology pathway(s) | ||
$4 BILLION MARKET
LARGE MARKET OPPORTUNITIES
BENIGN PROSTATIC HYPERPLASIA (BPH)
BPH is characterized by an abnormal benign growth of the prostatic tissues caused by testosterone. Symptoms linked to BPH include pain while urinating and frequent urges to urinate during the night and sometimes, kidney problems. In some cases, if left untreated, BPH may develop into prostate cancer. Contrary to most of the present treatments for BPH, cetrorelix is not associated with side-effects such as erectile dysfunction, loss of libido and chemical castration. According to Decision Resources, cetrorelix is currently the most advanced LHRH antagonist in development for the treatment of BPH.
$2.4 BILLION MARKET
NON-SMALL CELL LUNG CANCER
Lung cancer is the leading cause of cancer deaths in both men and women worldwide. Non-small cell lung cancer is its most common form and yet can be very difficult to treat. Typically, by the time patients report symptoms, the disease has often spread to other parts of the body. Perifosine, our leading oral, anti-cancer compound with its novel mechanism of action, has shown direct and specific action on tumor cells which have proven to be resistant to current anti-cancer agents. Furthermore, studies have demonstrated that perifosine may be well suited for use as a single agent or in combination with other treatments.
GOALS FOR 2007
Late-stage programs
CETRORELIX
· Initiate 2nd trial of Phase 3 program in BPH
· Initiate safety trial of Phase 3 program in BPH
· Announce full recruitment of first Phase 3 trial in BPH
· Disclose Japanese Phase 2 results in BPH
OZARELIX
· Disclose detailed European Phase 2 results in BPH
· Disclose top line U.S. Phase 2b results in BPH
· Disclose top line Phase 2b results in prostate cancer
PERIFOSINE
· Complete patient enrollment for Phase 2 trial in non-small cell lung cancer
· Disclose more Phase 1 and Phase 2 results in multiple cancers
· Initiate additional Phase 2 trials in multiple cancers
EARLIER-STAGE PROGRAMS
AN-152
· Disclose detailed Phase 1 results for breast and ovarian cancer
· Initiate Phase 2 program in ovarian and endometrial cancer
ZEN-012
· Initiate Phase 1 trial in solid tumors and lymphoma
· Announce top line results of Phase 1 trial in solid tumors and lymphoma
PRECLINICAL
· Disclose results for multiple programs
· LHRH peptidomimetics
· Erk/PI3K inhibitors
· Erucyl-PC
· Ghrelin antagonists
· Announce filing of an IND for a Phase 1 program
POISED FOR GROWTH
ÆTERNA ZENTARIS HAS ALL THE KEY FUNDAMENTALS TO EMERGE AS A FULLY-INTEGRATED, GLOBAL SPECIALIST-DRIVEN BIOPHARMACEUTICAL COMPANY WITH A STRATEGIC FOCUS ON ENDOCRINE THERAPY AND ONCOLOGY
CORPORATE INFORMATION
Head Office
Æterna Zentaris Inc.
1405 Parc-Technologique Blvd.
Quebec, Quebec G1P 4P5
CANADA
Phone: (418) 652-8525
Fax: (418) 652-0881
E-mail: aeterna@aeternazentaris.com
Internet: www.aeternazentaris.com
Ticker symbols
AEZ — The Toronto Stock Exchange (TSX)
AEZS — NASDAQ National Market
Transfer Agent and Registrar
Computershare Trust Company of Canada
1500 University Street, 7th Floor
Montreal, Quebec H3A 3S8
CANADA
Auditors
PricewaterhouseCoopers LLP
Place de la Cité, Tour Cominar
2640 Laurier Blvd., Suite 1700
Quebec, Quebec G1V 5C2
CANADA
Corporate Solicitors
Ogilvy Renault
1981 McGill College, Suite 1100
Montreal, Quebec H3A 3C1
CANADA
Arnold & Porter
399 Park Avenue
New York, NY 10022
USA
Annual Meeting
May 2, 2007, 10:30 a.m.
Le Centre Sheraton Hotel
1201 René-Lévesque Blvd. West
Montreal, Quebec H3B 2L7
CANADA
SCIENTIFIC ADVISORY BOARD
EXTERNAL MEMBERS
Dr. Gerald Batist, MD, CM, FACP
Director of the McGill Center for Translational
Research in Cancer, Chair and Professor,
Department of Oncology and Medicine,
McGill University, Jewish General Hospital,
Montreal, Canada
Dr. Frans M. J. Debruyne, MD, PhD
Professor and Chairman
at the University Medical Center in
Nijmegen, The Netherlands
Dr. Alan H. DeCherney, MD
Professor at UCLA School
of Medicine in Los Angeles,
United States
Prof. Dr. Klaus H.R. Diedrich, MD, PhD
Director, Department of Gynecology and Obstetrics
University Clinic of Luebeck, Germany
Prof. Dr. René Frydman, MD, PhD
Head of the Department of Gynecolgy Obstetrics
Hôpital Antoine Beclere, Paris, France
Dr. Fernand Labrie, OC, OQ, MD, PhD
Head, Centre hospitalier
de l’Université Laval Research
Center (CHUL), Quebec, Canada
Dr. Hartmut Michel, PhD
Director, Max-Planck Institute
for Biophysics, Frankfurt, Germany
Nobel Prize laureate in chemistry, 1988.
Prof. Dr. Marcel Verheij, MD, PhD
The Netherlands Cancer Institute/
Antoni van Leeuwenhoek
Hospital, Amsterdam, The Netherlands
Prof. Dr. Ulf Rapp
Institute of Medicine
Würzburg University
Germany
Dr. Daniel D. Von Hoff, MD, FACP
Senior Investigator
Translational Genomics Research Institute
Phoenix, Arizona, United States
DIRECTORS
Marcel Aubut, O.C., O.Q., Q.C.
Quebec, Canada
Managing Partner
Heenan Blaikie Aubut, Lawyers
Stormy Byorum, MBA (1)
New York, USA
Senior Managing Director
Stephens Cori Capital Advisors
(a division of Stephens Inc.)
José P. Dorais
Quebec, Canada
Partner
Miller Thomson Pouliot, Lawyers
Dr. Éric Dupont, PhD (2)
Quebec, Canada
Executive Chairman of the Board
Æterna Zentaris Inc.
Prof. Dr. Jürgen Engel, PhD
Frankfurt, Germany
Executive Vice President,
Global Research & Development
and Chief Operating Officer
Æterna Zentaris Inc.
Jürgen Ernst, MBA (2)
Brussels, Belgium
Corporate Director
Gilles R. Gagnon, MSc., MBA
Quebec, Canada
President and Chief Executive Officer
Æterna Zentaris Inc.
Dr. Pierre Laurin, PhD, O.C. (2)
Quebec, Canada
Executive in Residence
HEC Montreal
Gérard Limoges, CM, FCA (1)
Quebec, Canada
Corporate Director
Pierre MacDonald, MSc. Comm. (1) (2)
Quebec, Canada
Chairman of the Board
Eurocopter Canada Ltd.
Gerald J. Martin
California, USA
Corporate Director
(1) Member of the Audit Committee
(2) Member of the Corporate Governance, Nominating and Human Resources Committee
SENIOR OFFICERS
Dr. Éric Dupont, PhD
Executive Chairman of the Board
Gilles R. Gagnon, MSc., MBA
President and Chief Executive Officer
Prof. Dr. Jürgen Engel, PhD
Executive Vice President,
Global Research and Development
and Chief Operating Officer
Dr. Eckhard Günther, PhD
Vice President, Drug Discovery
Mario Paradis, CA
Vice President, Finance & Administration
and Corporate Secretary
Dr. Manfred Peukert, MD
Vice President, Medical Affairs
Dr. Matthias Rischer, PhD
Vice President, Pharmaceutical Development
Dennis Turpin, CA
Vice President and Chief Financial Officer
Printed in Canada

