Exhibit 99.1
| INVESTOR & ANALYST DAY The Roosevelt Hotel, New York 5 November 2014 the medicines company |
| Welcome Bob Laverty The Medicines INVESTOR & ANALYST Welcome Bob Laverty The Medicines |
| 3 Statements in this presentation about The Medicines Company (the “Company”), the Company’s products and product candidates, the timing of clinical trial results, regulatory submissions, product or indication launches, the Company’s strategy, future financial results and operations, and future opportunities for the Company, that are not purely historical, and all other statements that are not purely historical, may be deemed to be forward-looking statements for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995. Without limiting the foregoing, the words “believes," “anticipates," “plans," “expects," “intends," “estimates," “potential," “outlook,” “may,” “will,” “would,” and similar expressions are intended to identify forward-looking statements. There are a number of important factors that could cause actual results or events to differ materially from those indicated by such forward looking statements, including: the extent of the commercial success of our approved products and product candidates, if approved; whether the Company’s product candidates will advance in the clinical trials process on a timely basis or at all; whether clinical trial results will warrant submission of applications for regulatory approval; whether the Company’s product candidates will receive approvals from regulatory agencies; and such other factors as are set forth in the risk factors detailed from time to time in the Company’s periodic reports filed with the Securities and Exchange Commission including, without limitation, the risk factors detailed in the Company’s Quarterly Report on Form 10-Q filed with the SEC on August 4, 2014, which are incorporated herein by reference. The Company specifically disclaims any obligation to update these forward-looking statements whether as a result of new information, future events or otherwise. Legal notices Forward looking statements I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
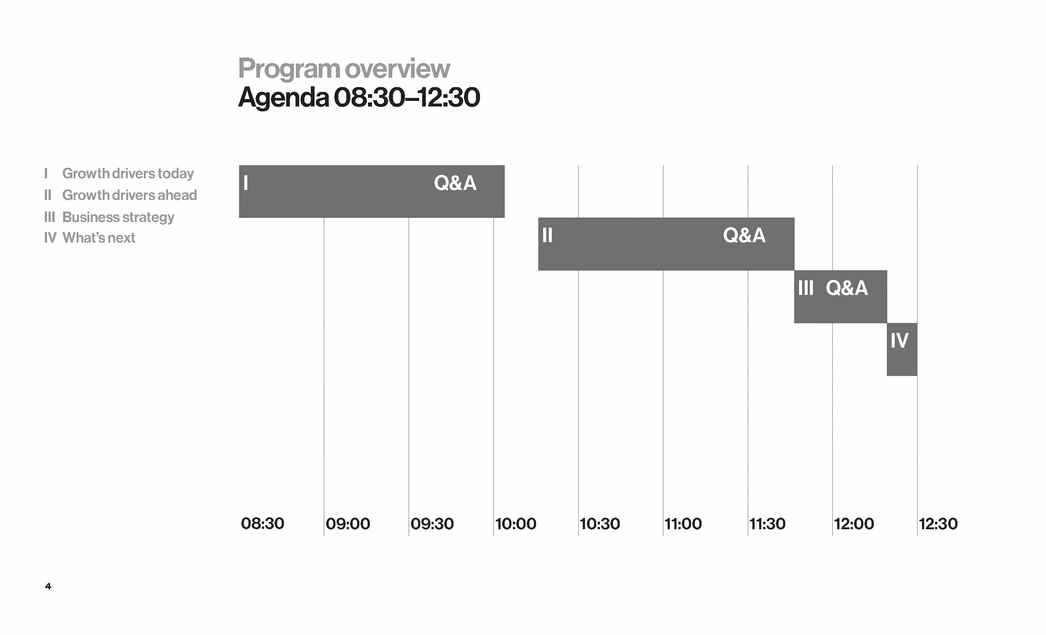
| 4 I Q&A II Q&A III Q&A IV 08:30 09:00 09:30 10:00 10:30 11:00 11:30 12:00 12:30 Program overview Agenda 08:30–12:30 I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| Overview Clive Meanwell The Medicines Company Overview Clive Meanwell The Medicines Company |
| 6 One year ago we described: Purposeful approach to the acute and intensive hospital care market Intellectual property program with ANGIOMAX® Four intended product launches in three hospital care pathways Our intention to add more products that fit our strategy Since that time we have: Moved forward and added to our late- and development-stage portfolio Continued to defend our ANGIOMAX® intellectual property Grown net revenue • 8.5% increase in total for the last four quarters from 12 months ended September 30, 2013 to 12 months ended September 30, 2014 • Which added $56M net revenue from $662M in the 12 months ended September 30, 2013 to $718M* in the 12 months ended September 30, 2014 Conducted three investor events covering our portfolio in depth Strategy for growth Investment thesis I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 7 One year ago Portfolio set up to serve three hospital care pathways Acute Cardiovascular Surgery & Perioperative Infectious Disease Market ANGIOMAX®/ANGIOX® BRILINTA®* RECOTHROM® CLEVIPREX® HTN Argatroban, 50 mg per 50 mL† NDA Cangrelor IONSYS™ RAPLIXA™ ORBACTIV™ ABSSSI R&D CLEVIPREX® AHF MDCO-216 ApoA1-M ALN-PCSsc‡ ABP-700§ ORBACTIV™ new indns. I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next For additional information on these products, including indications and Important Safety Information for marketed products, please visit www.themedicinescompany.com. *Co-promotion for AstraZeneca. †The Medicines Company has licensed the rights to market ready-to-use Argatroban in the United States and Canada from Eagle Pharmaceuticals, Inc. ‡Partner with Alnylam. §Partner with Annovation. All marks are the property of their respective owners. |
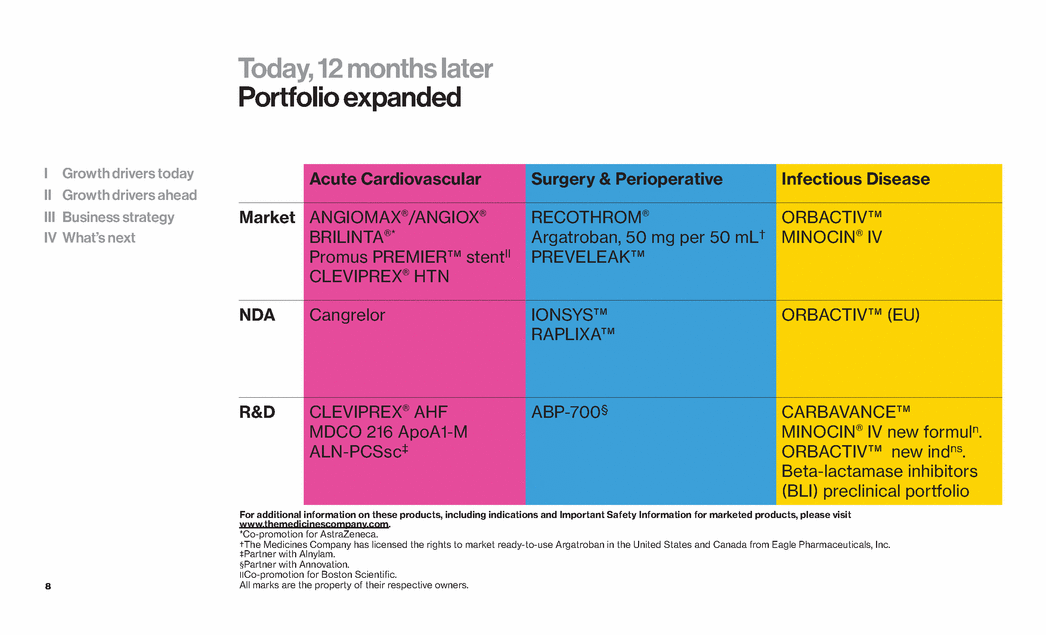
| 8 Today, 12 months later Portfolio expanded Acute Cardiovascular Surgery & Perioperative Infectious Disease Market ANGIOMAX®/ANGIOX® BRILINTA®* Promus PREMIER™ stentII CLEVIPREX® HTN RECOTHROM® Argatroban, 50 mg per 50 mL† PREVELEAK™ ORBACTIV™ MINOCIN® IV NDA Cangrelor IONSYS™ RAPLIXA™ ORBACTIV™ (EU) R&D CLEVIPREX® AHF MDCO 216 ApoA1-M ALN-PCSsc‡ ABP-700§ CARBAVANCE™ MINOCIN® IV new formuln. ORBACTIV™ new indns. Beta-lactamase inhibitors (BLI) preclinical portfolio I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next For additional information on these products, including indications and Important Safety Information for marketed products, please visit www.themedicinescompany.com. *Co-promotion for AstraZeneca. †The Medicines Company has licensed the rights to market ready-to-use Argatroban in the United States and Canada from Eagle Pharmaceuticals, Inc. ‡Partner with Alnylam. §Partner with Annovation. Co-promotion for Boston Scientific. All marks are the property of their respective owners. |
| 9 Major regulatory activities during the last 12 months Asset United States Europe ORBACTIV™ NDA approved, launched MAA review on track Cangrelor CRL resubmission MAA review on track (planned Q4) RAPLIXA™ NDA review ext. to 4/15 MAA review on track IONSYS™ NDA review on track MAA review on track Strategy for growth: 12 months of execution Moved forward our late stage portfolio I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 10 Asset acquisitions during the last 12 months Asset United States Europe MINOCIN® IV Acquired, re-launched MINOCIN® IV new formuln Acquired, sNDA on track MAA plan accepted (RPX-602) CARBAVANCE™ Acquired, Phase 3 started MAA plan accepted BARDA contract up to $90M Range of BLI programs Acquired, IND preparation PREVELEAK™ Acquired, re-launch Marketed planned for 1H15 Strategy for growth: 12 months of execution Added to our portfolio by acquiring Rempex and Tenaxis I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
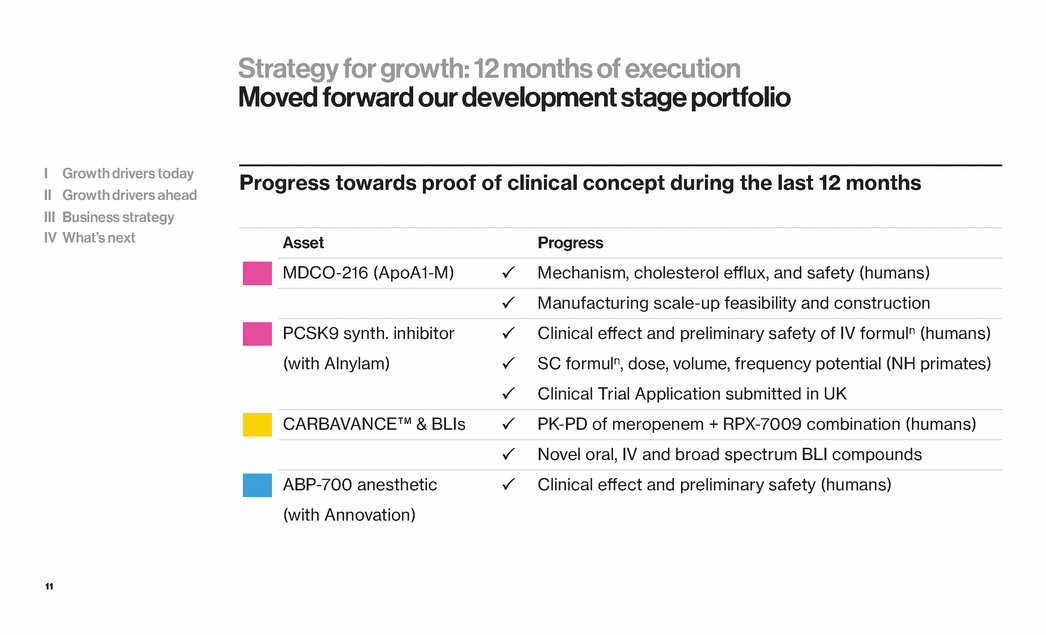
| 11 Progress towards proof of clinical concept during the last 12 months Asset Progress MDCO-216 (ApoA1-M) Mechanism, cholesterol efflux, and safety (humans) Manufacturing scale-up feasibility and construction PCSK9 synth. inhibitor Clinical effect and preliminary safety of IV formuln (humans) (with Alnylam) SC formuln, dose, volume, frequency potential (NH primates) Clinical Trial Application submitted in UK CARBAVANCE™ & BLIs PK-PD of meropenem + RPX-7009 combination (humans) Novel oral, IV and broad spectrum BLI compounds ABP-700 anesthetic Clinical effect and preliminary safety (humans) (with Annovation) Strategy for growth: 12 months of execution Moved forward our development stage portfolio I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
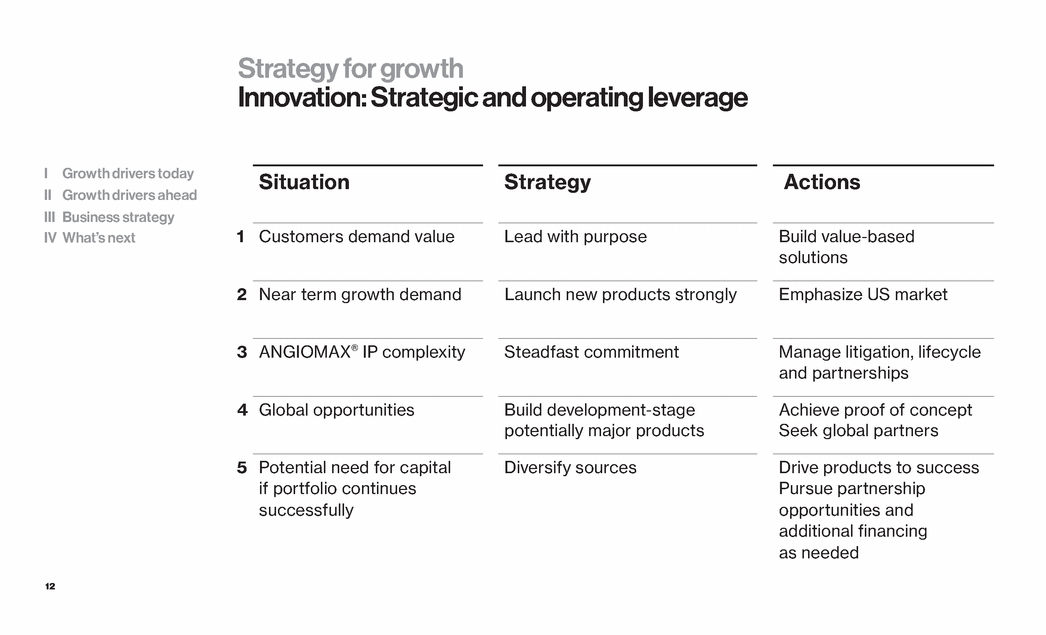
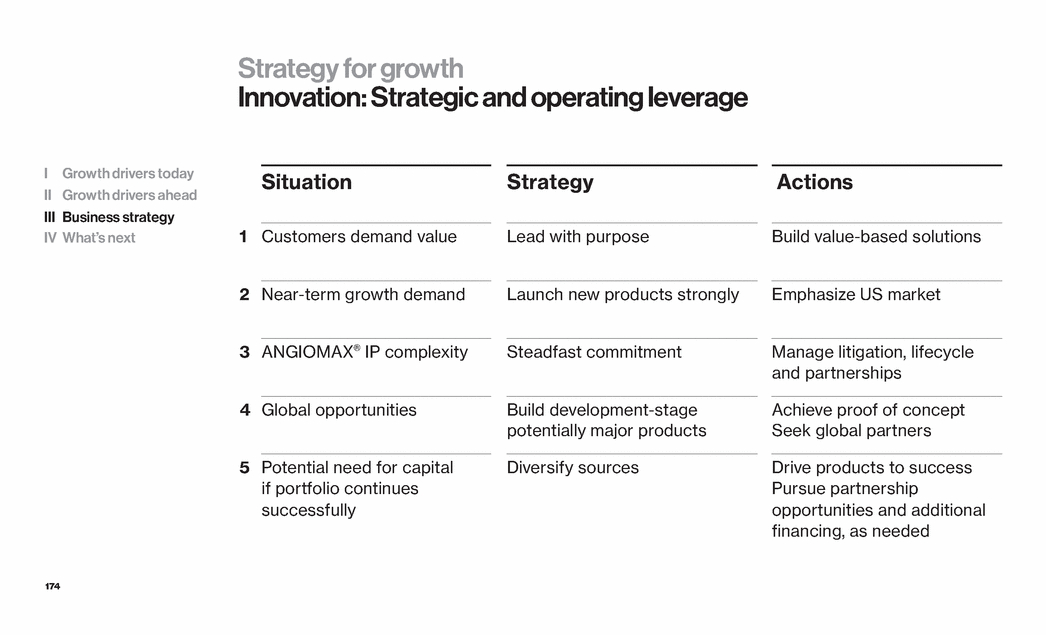
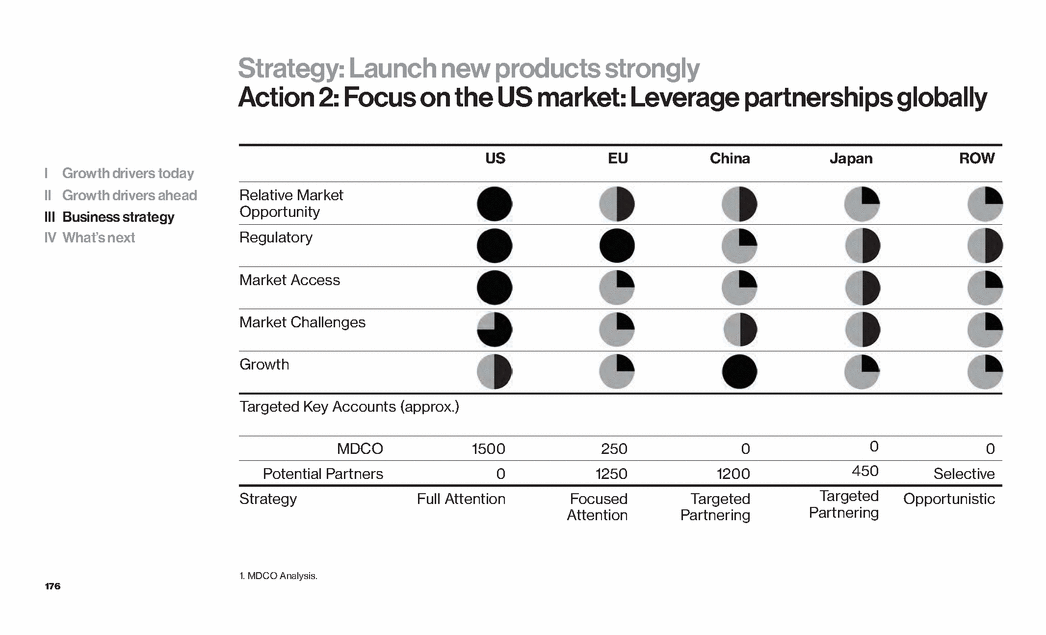
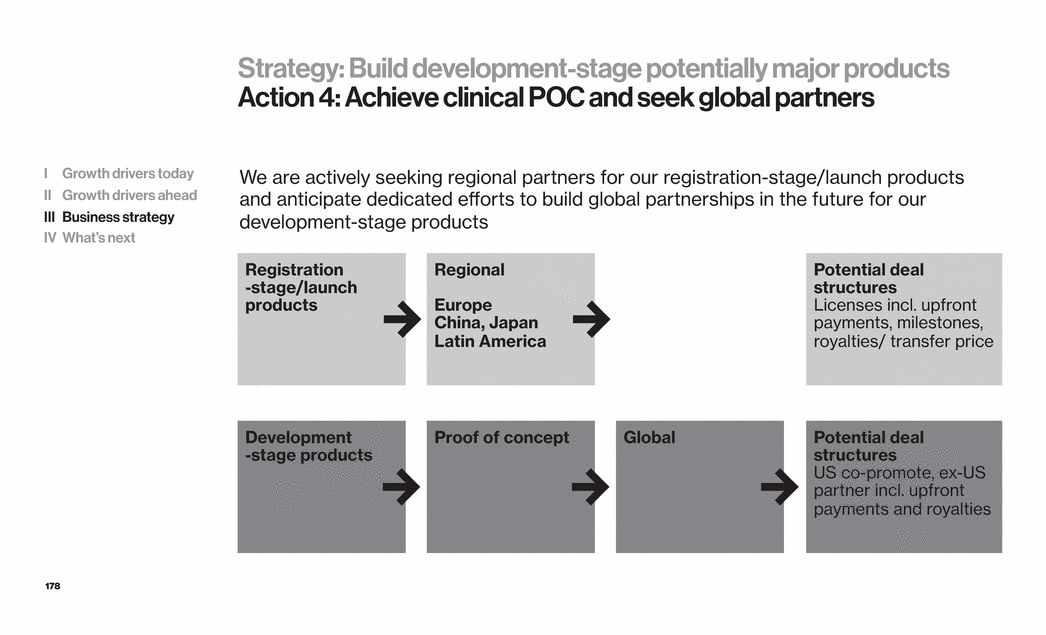
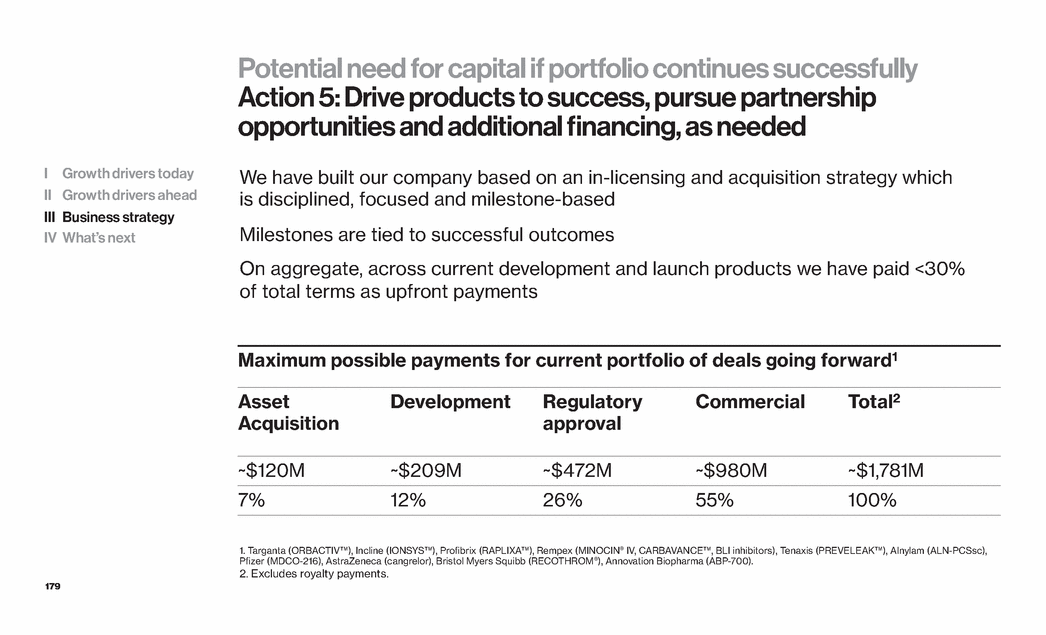
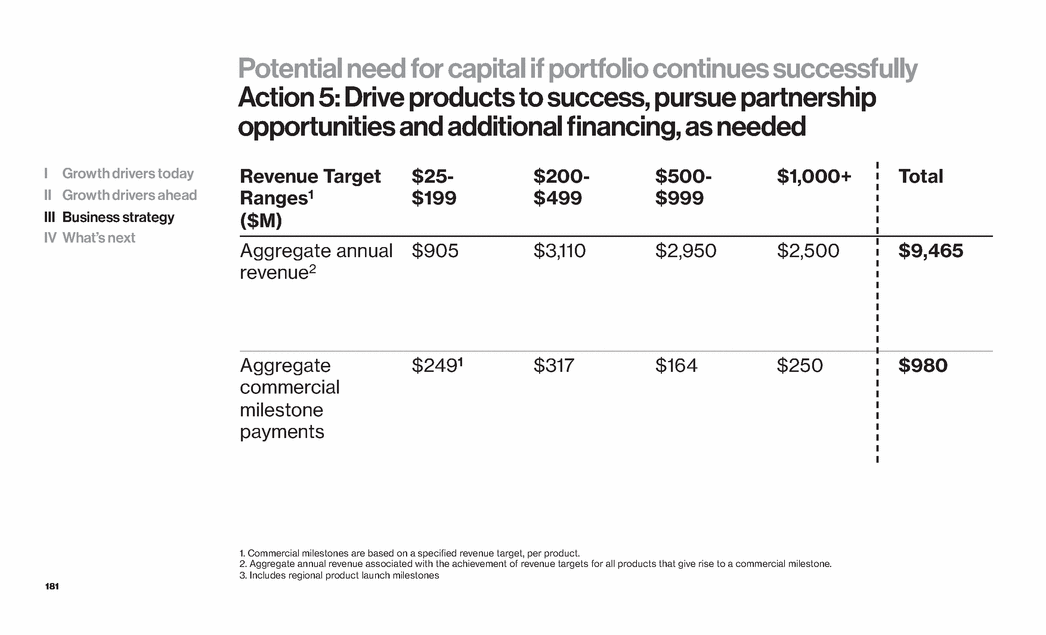
| 12 Strategy for growth Innovation: Strategic and operating leverage Situation Strategy Actions 1 Customers demand value Lead with purpose Build value-based solutions 2 Near term growth demand Launch new products strongly Emphasize US market 3 ANGIOMAX® IP complexity Steadfast commitment Manage litigation, lifecycle and partnerships 4 Global opportunities Build development-stage potentially major products Achieve proof of concept Seek global partners 5 Potential need for capital if portfolio continues successfully Diversify sources Drive products to success Pursue partnership opportunities and additional financing as needed I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 13 Describe progress, status and plans, focusing on: Marketed growth drivers today ORBACTIV™ and MINOCIN® IV PREVELEAK™ ANGIOMAX® Growth drivers ahead—under regulatory review RAPLIXA™ IONSYS™ Cangrelor Growth drivers ahead—at or approaching clinical proof of concept ApoA1-Milano cholesterol efflux promoter PCSK9sc synthesis inhibitor CARBAVANCE™ antibacterial against gram-negative MDR including CRE ABP-700 anesthetic Business strategy and what’s next Investor & Analyst Day Objectives I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| Growth drivers today Clive Meanwell The Medicines Company |
| What hospitals want Mike Jellinek Lahey Health System |
| Current Issues in US Hospitals Michael Jellinek, M.D. CEO, Community Network EVP, Lahey Health System Burlington, MA November 5, 2014 |
| Mission • Care for every patient like a beloved family member – Safe, high quality, coordinated care • Exceed patient’s and family’s expectations • Relentless pursuit of excellence • Teaching standard – “Do it the way you would teach it.” • Academic medical center – Teach – Research |
| Market Forces Vary • All fee for service—18% of GDP, slope? • Commercial vs. Medicare vs. Medicaid • Universal coverage vs. indigent care burden • Competition: – Hospital, physicians and for-profit, technical and ancillary services • Population health management/holding risk (lower utilization, infrastructure costs, IT, funded by fee for service surplus...better care) |
| Financial Pressures • Fixed cost structure, 24 x 7, full service – Ready for anything—Ebola, flu, fire, bus accident, – Bullet wound, heart attack • 60% + labor expense • Heavily regulated, high standards • IT investment (EPIC) • AMC—teaching/research requires subsidy • Pension costs • Most services under-reimbursed dependent on a few well reimbursed services |
| Utilization Critical • Hospital margin range . Most low (0 to 4%) • 80% plus utilization to cover fixed costs • Fee for service—incentive to utilize but: • Inpatient to outpatient (gall bladder, hip) • Length of stay down, more admissions • Co-pays and high deductibles decrease and steer patients • Readmissions and harms down (Medicare) • Research—hepatitis C, flu vaccine • Population health management (ER, heart failure) |
| Management Responses • Growth: volume in fee for service; covered lives in HPM (costly investments) • Constrain capital (age of plant, bond rating) • Wage increases limited; lower benefit costs • Process improvement • Supply chain; standardization • Review costly new technology • Pharmacy management (pre-release guidelines) • Decrease poorly reimbursed services (mission) • Limit investment in population health management. |
| Likely Future • Right care in right place at right time • Value based purchasing—quality outcome at lower cost • Process improvement, standardization • Focus on patient experience... satisfaction • Population health management consolidation, integrate insurance and provider • Movement to outpatient and home services • IT facilitation and disruption or • Fee for service with reimbursement cuts and fractionated, poorly coordinated care |
| Launching 2 new antibiotics: ORBACTIV™ and MINOCIN® IV Mike McGuire, Michael Mitchell The Medicines Company |
| 24 Efficacy and safety of single dose oritavancin compared to 7–10 days of vancomycin in patients with acute bacterial skin and skin structure infections treated in outpatient setting.” K. LaPensee, W. Fan, H. Jiang, T. Lodise New data presented at ICAAC SOLO I data support outpatient treatment option “ I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next For additional information about ORBACTIV™, including indication and Important Safety Information, please see www.orbactiv.com. |
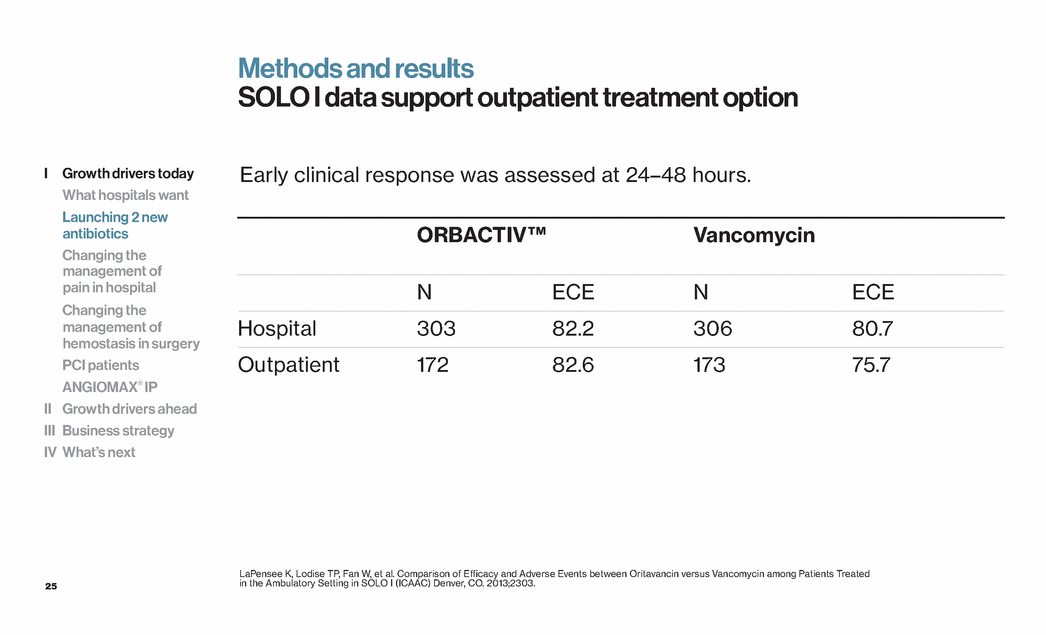
| 25 Early clinical response was assessed at 24–48 hours. Methods and results SOLO I data support outpatient treatment option ORBACTIV™ Vancomycin N ECE N ECE Hospital 303 82.2 306 80.7 Outpatient 172 82.6 173 75.7 LaPensee K, Lodise TP, Fan W, et al. Comparison of Efficacy and Adverse Events between Oritavancin versus Vancomycin among Patients Treated in the Ambulatory Setting in SOLO I (ICAAC) Denver, CO. 2013;2303. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
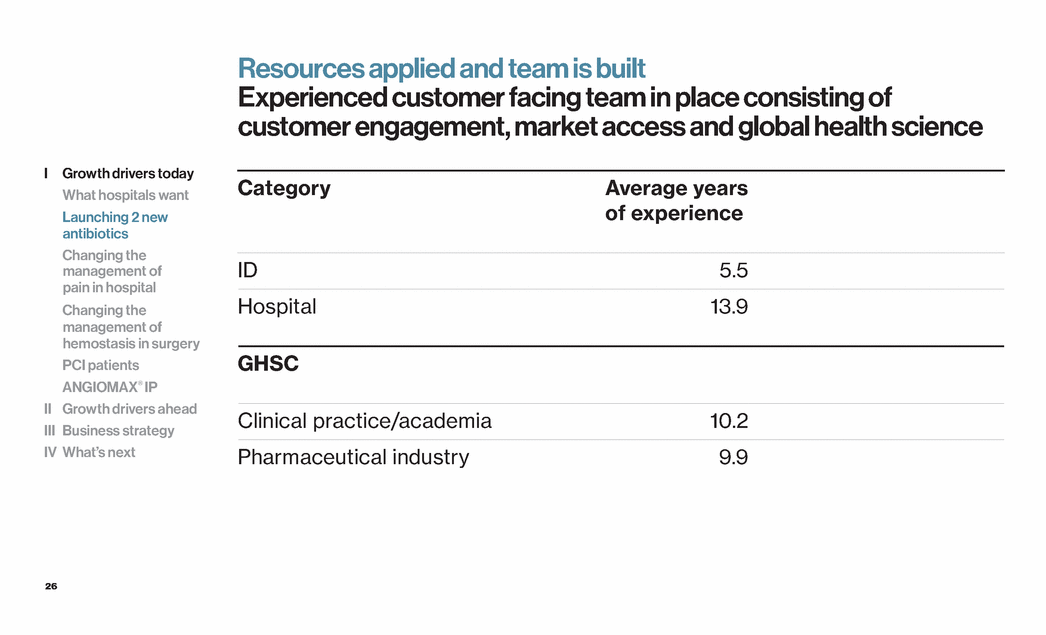
| 26 Resources applied and team is built Experienced customer facing team in place consisting of customer engagement, market access and global health science Category Average years of experience ID 5.5 Hospital 13.9 GHSC Clinical practice/academia 10.2 Pharmaceutical industry 9.9 I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
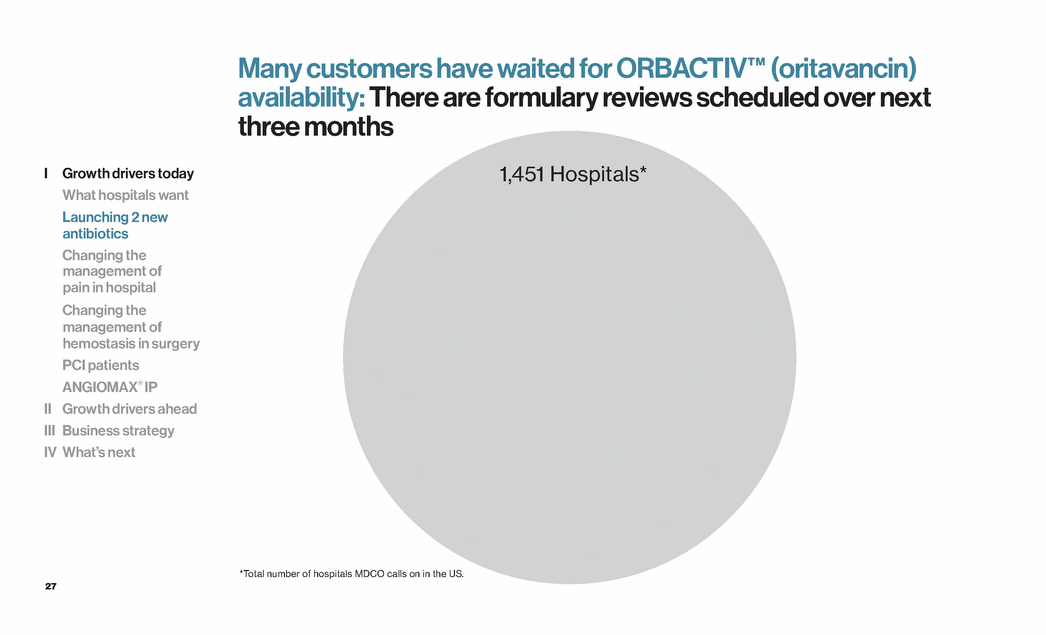
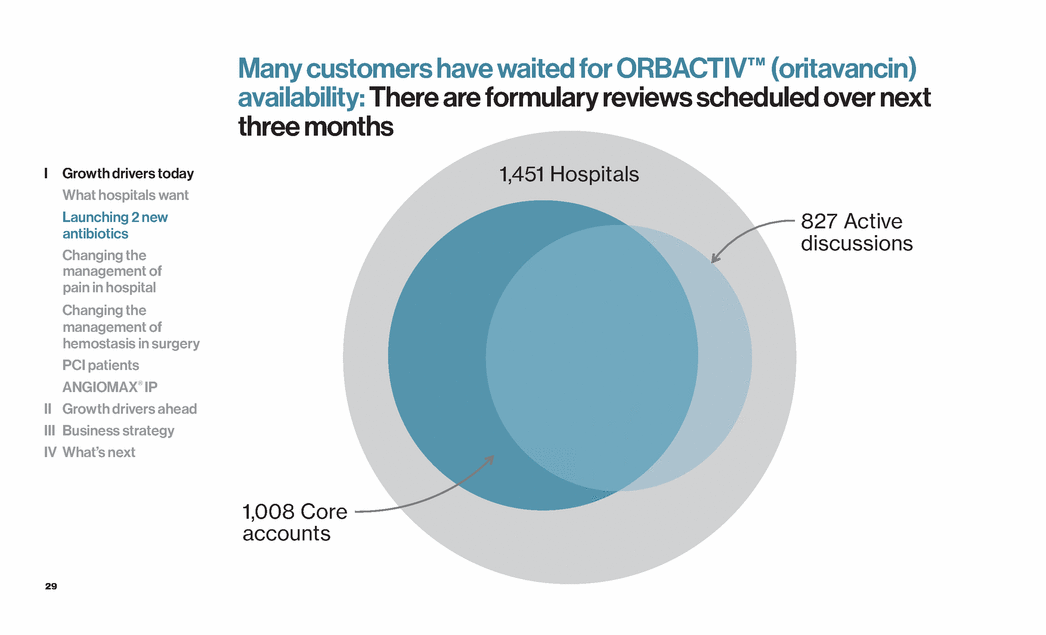
| 27 Many customers have waited for ORBACTIV™ (oritavancin) availability: There are formulary reviews scheduled over next three months 1,451 Hospitals* *Total number of hospitals MDCO calls on in the US. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
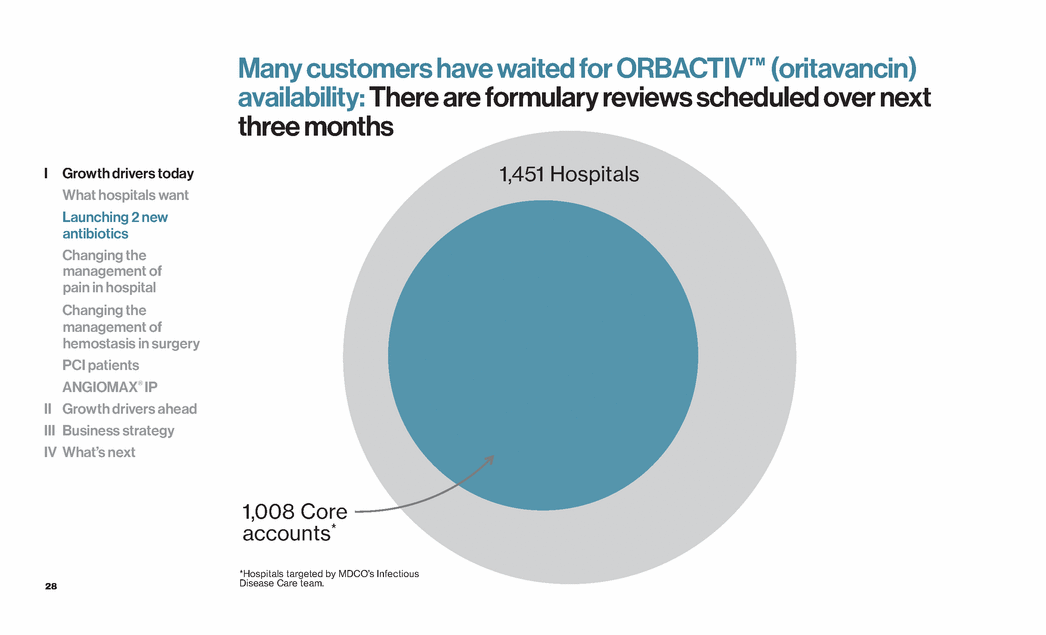
| 28 1,451 Hospitals 1,008 Core accounts* *Hospitals targeted by MDCO’s Infectious Disease Care team. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next Many customers have waited for ORBACTIV™ (oritavancin) availability: There are formulary reviews scheduled over next three months |
| 29 1,451 Hospitals 827 Active discussions 1,008 Core accounts I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next Many customers have waited for ORBACTIV™ (oritavancin) availability: There are formulary reviews scheduled over next three months |
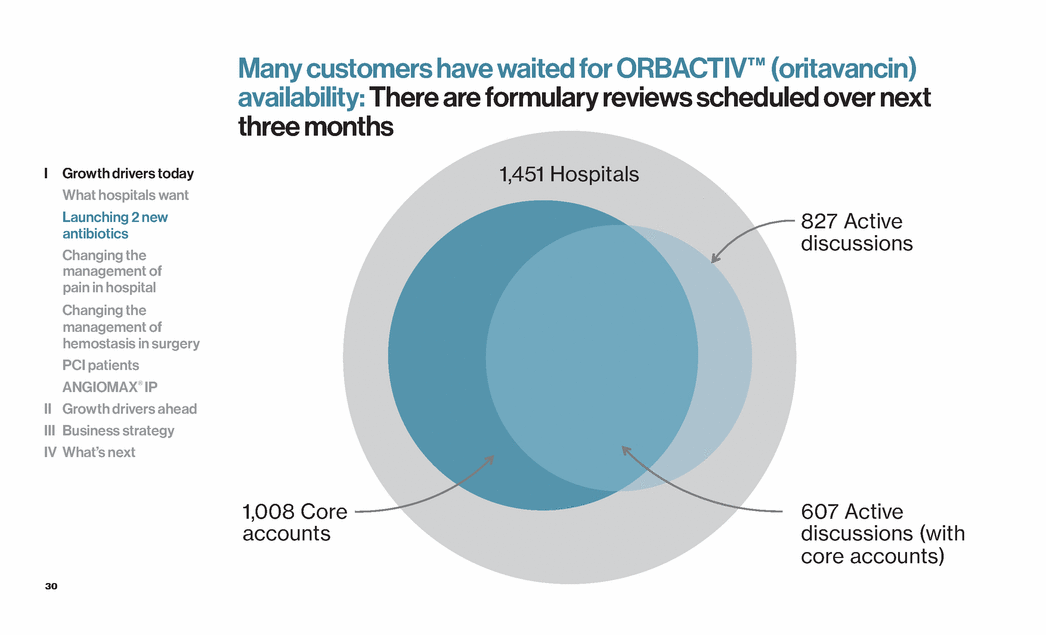
| 30 1,451 Hospitals 827 Active discussions 607 Active discussions (with core accounts) 1,008 Core accounts I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next Many customers have waited for ORBACTIV™ (oritavancin) availability: There are formulary reviews scheduled over next three months |
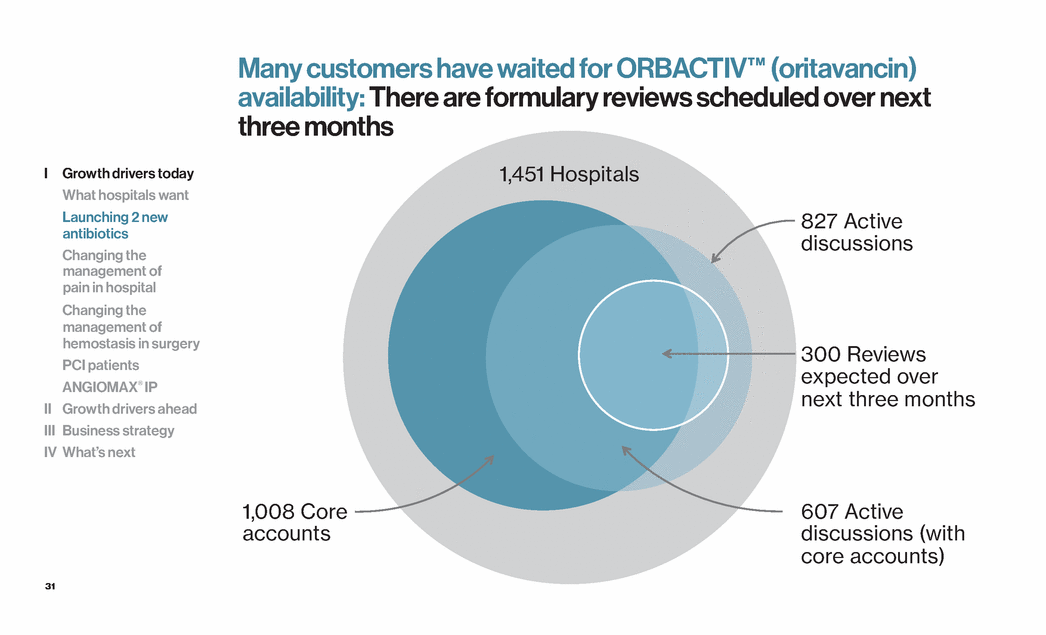
| 31 827 Active discussions 607 Active discussions (with core accounts) 300 Reviews expected over next three months 1,008 Core accounts 1,451 Hospitals I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next Many customers have waited for ORBACTIV™ (oritavancin) availability: There are formulary reviews scheduled over next three months |
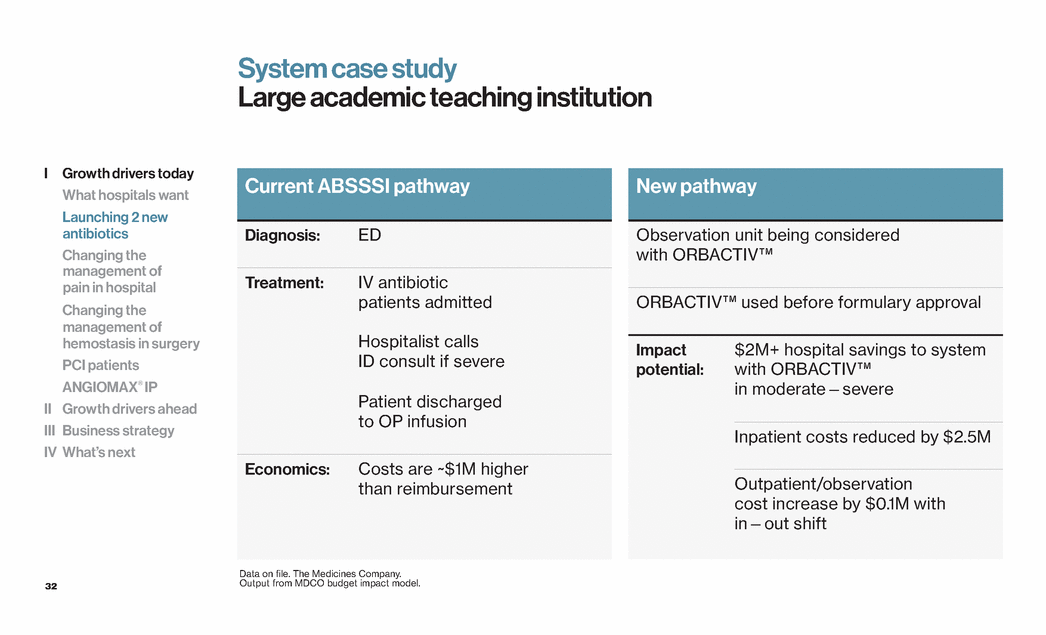
| 32 System case study Large academic teaching institution Current ABSSSI pathway Diagnosis: ED Treatment: IV antibiotic patients admitted Hospitalist calls ID consult if severe Patient discharged to OP infusion Economics: Costs are ~$1M higher than reimbursement New pathway Observation unit being considered with ORBACTIV™ ORBACTIV™ used before formulary approval Impact potential: $2M+ hospital savings to system with ORBACTIV™ in moderate — severe Inpatient costs reduced by $2.5M Outpatient/observation cost increase by $0.1M with in — out shift Data on file. The Medicines Company. Output from MDCO budget impact model. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 33 Hospital case study Small community hospital Current ABSSSI pathway Diagnosis: Majority in ED Treatment: Admitted by hospitalist 200 ABSSSI per year Economics: Costs are $500–$1500 per patient higher than reimbursement New pathway ORBACTIV™ OPAT only ID restriction short term Long-term goal to open access to hospitalist + ED staff Gaining experience by ID to set new guidelines Impact potential: Estimated $1300+ saved per ABSSSI patient if used in moderate—severe Data on file. The Medicines Company. Output from MDCO budget impact model. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 34 Outpatient case study ID owned infusion center Current ABSSSI pathway Diagnosis: Referrals Treatment: Direct referral from MD • 3–6 days to complete therapy Hospital discharge referrals • 7–10 days IV OPAT Up to 2 hours drive time New pathway Direct referrals treated by single infusion Hospital discharge patients treated by single infusion Impact potential1: No PICC line ($400+)2 Miles per visit avoided Eliminates travel to infusion center every day for 3–10 days 1. Data on file. The Medicines Company. 2. Robinson MK, Mogensen KM, Grudinskas GF, et al. Improved care and reduced costs for patients requiring peripherally inserted central catheters: The role of bedside ultrasound and a dedicated team. J Parenter Enteral Nutr. 2005;29:374-9. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| MINOCIN® IV |
| 36 MINOCIN® IV (minocycline for injection) case study Large academic teaching center in the central US For MINOCIN® IV indication and Important Safety Information, please see www.minociniv.com. Multidisciplinary institution serving a large geographic area Over 40,000 patients admitted a year for many services including cancer treatment and transplant operations Antibiotic stewardship committee recommendation: Addition of MINOCIN® IV to the formulary to treat multidrug resistant (MDR) acinetobacter High level of carbapenem resistance to acinetobacter I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
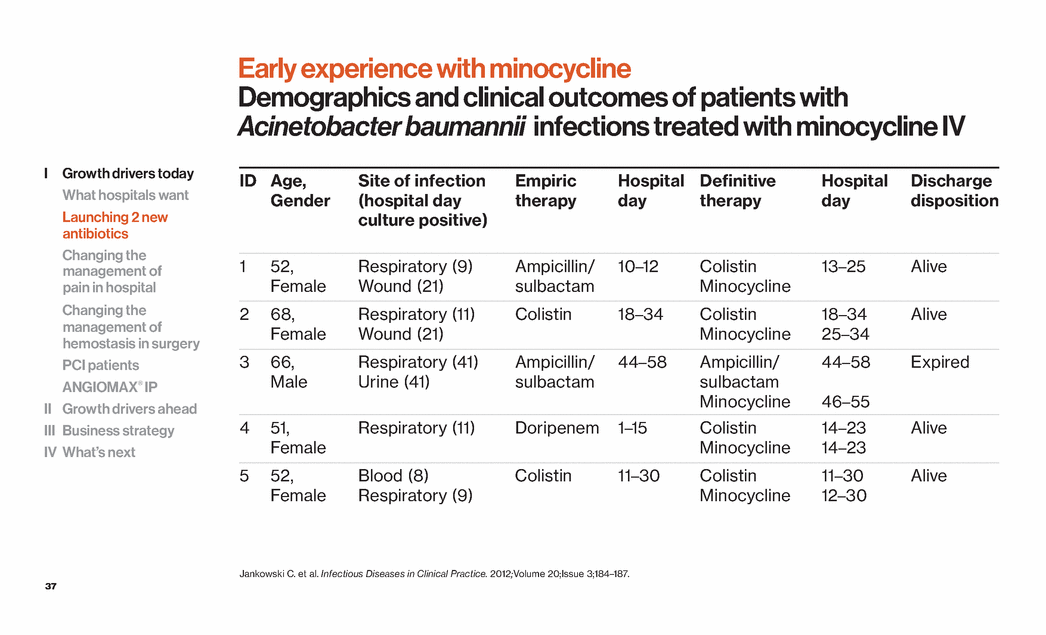
| 37 Jankowski C. et al. Infectious Diseases in Clinical Practice. 2012;Volume 20;Issue 3;184–187. Early experience with minocycline Demographics and clinical outcomes of patients with Acinetobacter baumannii infections treated with minocycline IV ID Age, Gender Site of infection (hospital day culture positive) Empiric therapy Hospital day Definitive therapy Hospital day Discharge disposition 1 52, Female Respiratory (9) Wound (21) Ampicillin/sulbactam 10–12 Colistin Minocycline 13–25 Alive 2 68, Female Respiratory (11) Wound (21) Colistin 18–34 Colistin Minocycline 18–34 25–34 Alive 3 66, Male Respiratory (41) Urine (41) Ampicillin/sulbactam 44–58 Ampicillin/ sulbactam Minocycline 44–58 46–55 Expired 4 51, Female Respiratory (11) Doripenem 1–15 Colistin Minocycline 14–23 14–23 Alive 5 52, Female Blood (8) Respiratory (9) Colistin 11–30 Colistin Minocycline 11–30 12–30 Alive I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
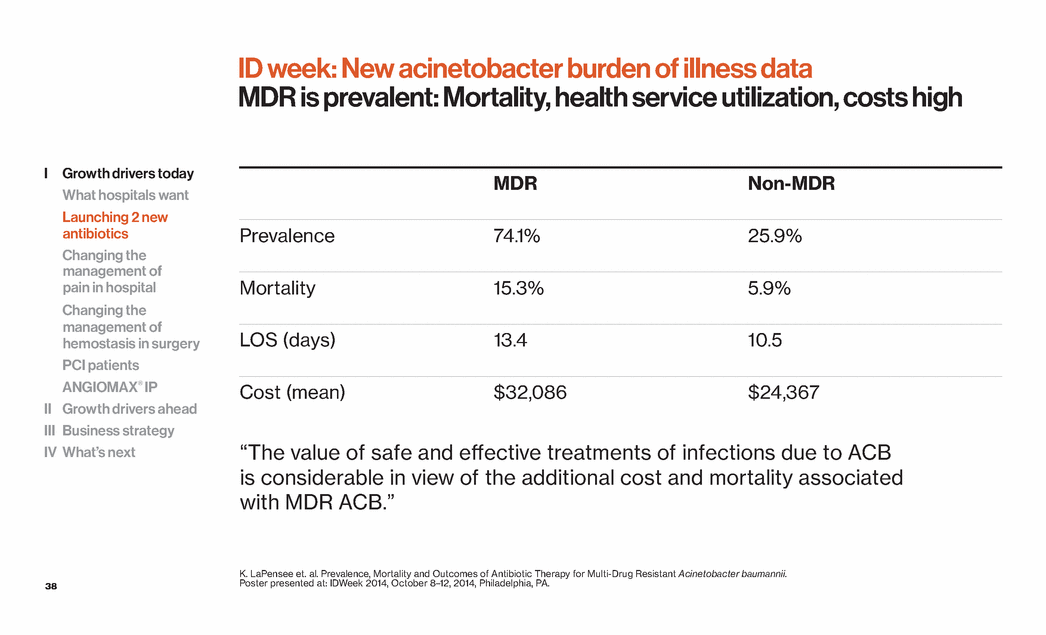
| 38 ID week: New acinetobacter burden of illness data MDR is prevalent: Mortality, health service utilization, costs high MDR Non-MDR Prevalence 74.1% 25.9% Mortality 15.3% 5.9% LOS (days) 13.4 10.5 Cost (mean) $32,086 $24,367 K. LaPensee et. al. Prevalence, Mortality and Outcomes of Antibiotic Therapy for Multi-Drug Resistant Acinetobacter baumannii. Poster presented at: IDWeek 2014, October 8–12, 2014, Philadelphia, PA. “The value of safe and effective treatments of infections due to ACB is considerable in view of the additional cost and mortality associated with MDR ACB.” I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 39 Being developed to help facilitate ease of use • Allows smaller administration volume at a more physiologic pH • Current formulation pH is 2.5–4.01, new formulation will be at 4.5–6.02 • Can infuse doses in a smaller volume where larger fluid volumes may be problematic Expect sNDA filing in 4Q 2014; anticipated approval 1H 2015 RPX-602 New formulation of MINOCIN® IV 1. MINOCIN® IV Prescribing Information. 2. Data on file. The Medicines Company. RPX-602 is an investigational agent not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 40 Summary ORBACTIV™ Many hospitals delayed formulary process until ORBACTIV™ availability Experienced customer-facing organization in place Early formulary wins and usage reflect positive response to value pricing and data Anticipated events—4Q ISPOR EU—Budget impact model • Poster presentation I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 41 Summary MINOCIN® IV MINOCIN® IV is a safe and effective treatment for MDR acinetobacter infections1 MINOCIN® IV use is expected to increase in target ORBACTIV™ institutions Anticipated events—4Q sNDA submission for new formulation 1. MINOCIN® IV Prescribing Information. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| Changing the management of pain in hospital: IONSYS™ Jason Campagna, Jim Jones The Medicines Company |
| 43 Disruptive innovation State-of-the-art technology 2014: Patient-Controlled Analgesia I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
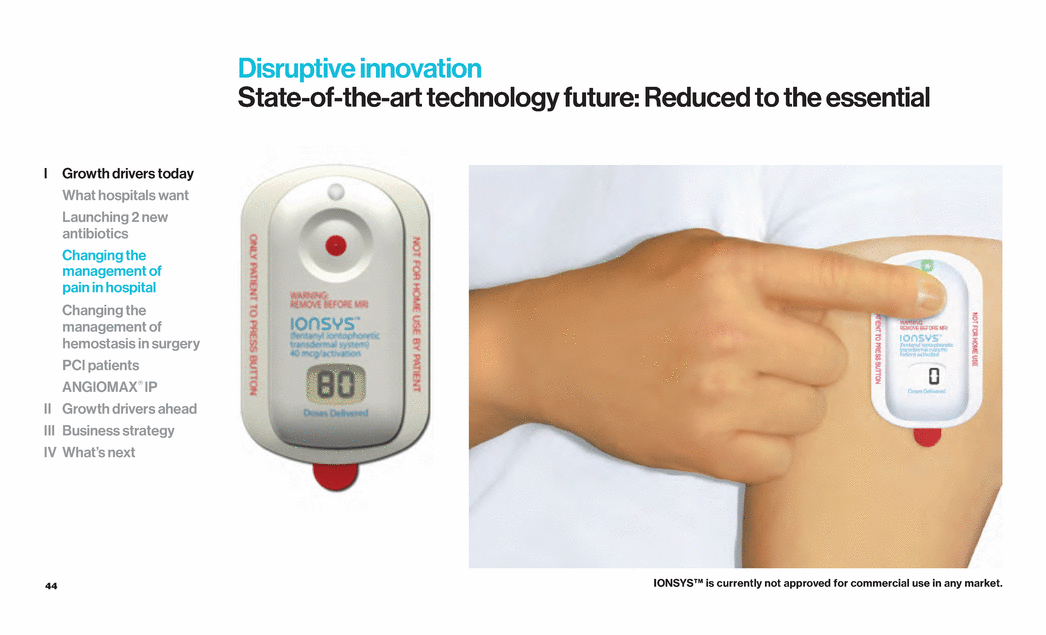
| 44 Disruptive innovation State-of-the-art technology future: Reduced to the essential IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
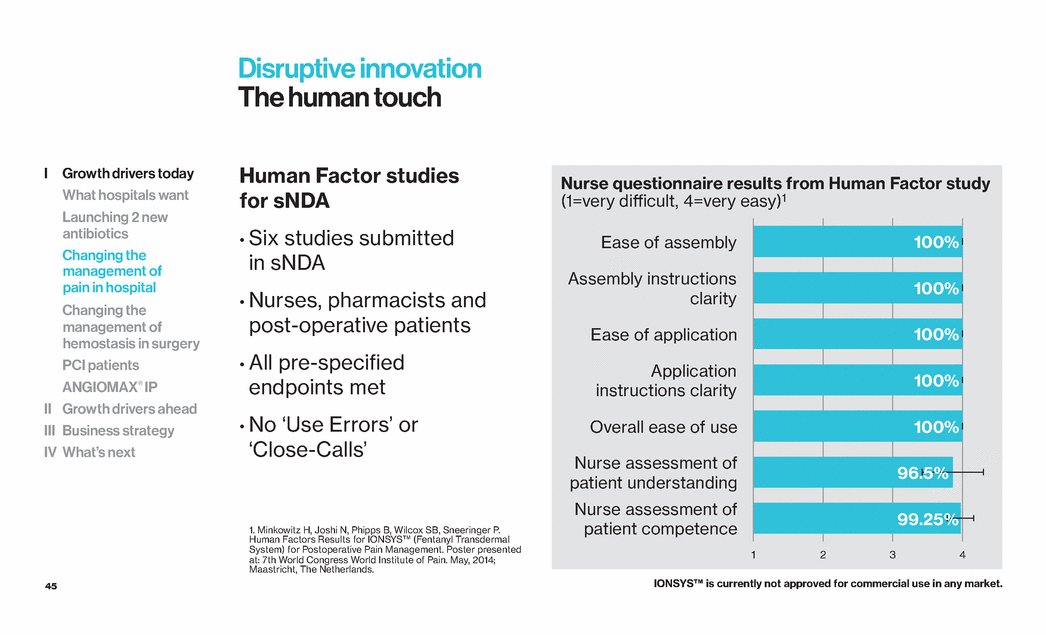
| 45 1 2 3 4 Ease of assembly Assembly instructions clarity Ease of application Application instructions clarity Overall ease of use Nurse assessment of patient understanding Nurse assessment of patient competence Disruptive innovation The human touch Human Factor studies for sNDA • Six studies submitted in sNDA • Nurses, pharmacists and post-operative patients • All pre-specified endpoints met • No ‘Use Errors’ or ‘Close-Calls’ Nurse questionnaire results from Human Factor study (1=very difficult, 4=very easy)1 100% 100% 100% 100% 100% 96.5% 99.25% 1. Minkowitz H, Joshi N, Phipps B, Wilcox SB, Sneeringer P. Human Factors Results for IONSYS™ (Fentanyl Transdermal System) for Postoperative Pain Management. Poster presented at: 7th World Congress World Institute of Pain. May, 2014; Maastricht, The Netherlands. IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 46 Disruptive innovation IONSYS™ (fentanyl iontophoretic transdermal system) performance Previous one part system: • <3% failure rate • Approved by FDA and EMA, and launched in Europe • 15,000 units sold in northern Europe in first six months Current two part system failure rate <0.2% (>4,000 systems tested) Data on file. The Medicines Company. IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
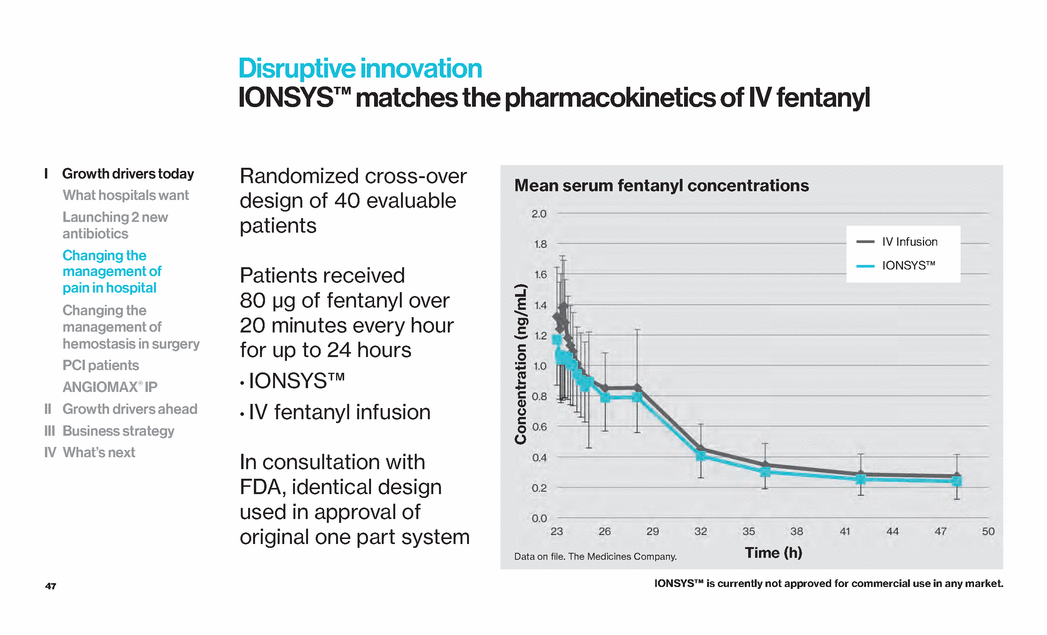
| 47 Disruptive innovation IONSYS™ matches the pharmacokinetics of IV fentanyl Randomized cross-over design of 40 evaluable patients Patients received 80 µg of fentanyl over 20 minutes every hour for up to 24 hours • IONSYS™ • IV fentanyl infusion In consultation with FDA, identical design used in approval of original one part system IV Infusion IONSYS™ Concentration (ng/mL) Time (h) Data on file. The Medicines Company. Mean serum fentanyl concentrations IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
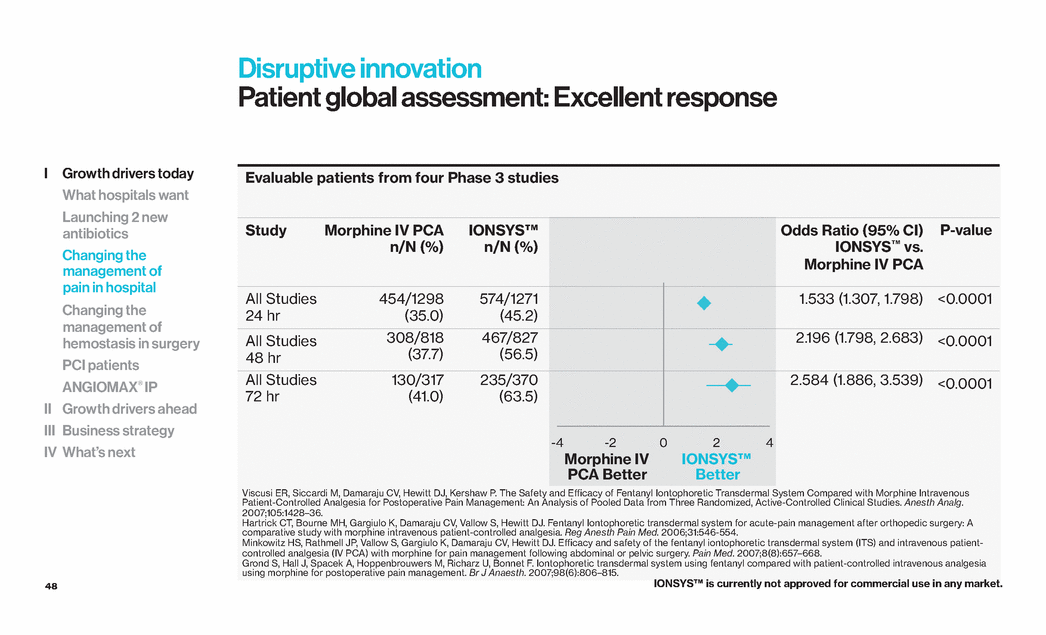
| 48 Evaluable patients from four Phase 3 studies Study Morphine IV PCA n/N (%) IONSYS™ n/N (%) Odds Ratio (95% CI) IONSYS™ vs. Morphine IV PCA P-value All Studies 24 hr 454/1298 (35.0) 574/1271 (45.2) 1.533 (1.307, 1.798) <0.0001 All Studies 48 hr 308/818 (37.7) 467/827 (56.5) 2.196 (1.798, 2.683) <0.0001 All Studies 72 hr 130/317 (41.0) 235/370 (63.5) 2.584 (1.886, 3.539) <0.0001 -4 -2 0 2 4 Morphine IV PCA Better IONSYS™ Better Disruptive innovation Patient global assessment: Excellent response Viscusi ER, Siccardi M, Damaraju CV, Hewitt DJ, Kershaw P. The Safety and Efficacy of Fentanyl Iontophoretic Transdermal System Compared with Morphine Intravenous Patient-Controlled Analgesia for Postoperative Pain Management: An Analysis of Pooled Data from Three Randomized, Active-Controlled Clinical Studies. Anesth Analg. 2007;105:1428–36. Hartrick CT, Bourne MH, Gargiulo K, Damaraju CV, Vallow S, Hewitt DJ. Fentanyl Iontophoretic transdermal system for acute-pain management after orthopedic surgery: A comparative study with morphine intravenous patient-controlled analgesia. Reg Anesth Pain Med. 2006;31:546-554. Minkowitz HS, Rathmell JP, Vallow S, Gargiulo K, Damaraju CV, Hewitt DJ. Efficacy and safety of the fentanyl iontophoretic transdermal system (ITS) and intravenous patientcontrolled analgesia (IV PCA) with morphine for pain management following abdominal or pelvic surgery. Pain Med. 2007;8(8):657–668. Grond S, Hall J, Spacek A, Hoppenbrouwers M, Richarz U, Bonnet F. Iontophoretic transdermal system using fentanyl compared with patient-controlled intravenous analgesia using morphine for postoperative pain management. Br J Anaesth. 2007;98(6):806–815. IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
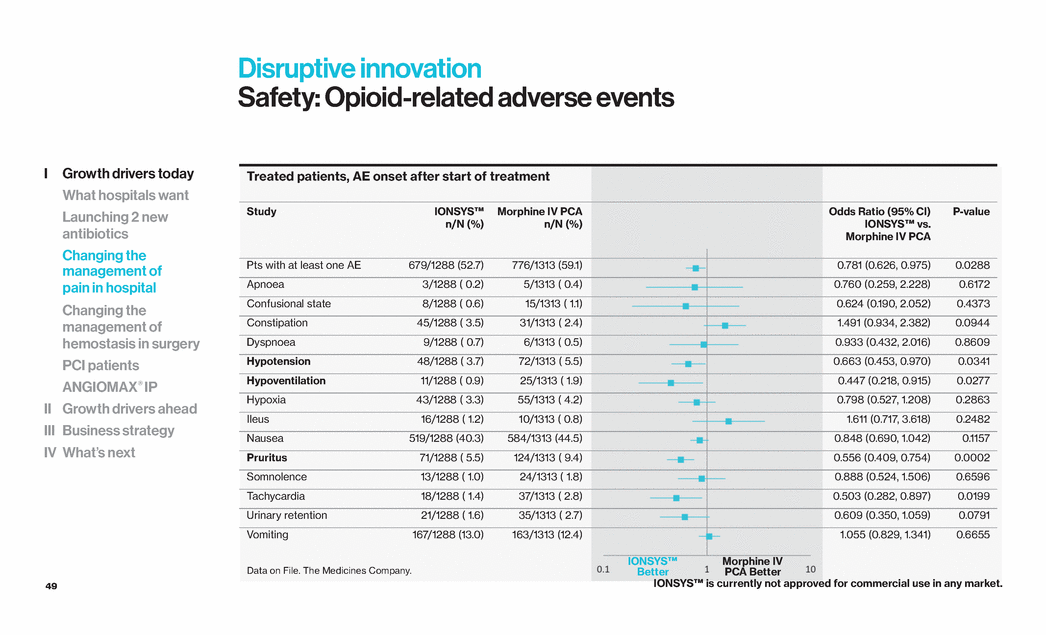
| 49 I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next Treated patients, AE onset after start of treatment Study IONSYS™ n/N (%) Morphine IV PCA n/N (%) Odds Ratio (95% CI) IONSYS™ vs. Morphine IV PCA P-value Pts with at least one AE 679/1288 (52.7) 776/1313 (59.1) 0.781 (0.626, 0.975) 0.0288 Apnoea 3/1288 ( 0.2) 5/1313 ( 0.4) 0.760 (0.259, 2.228) 0.6172 Confusional state 8/1288 ( 0.6) 15/1313 ( 1.1) 0.624 (0.190, 2.052) 0.4373 Constipation 45/1288 ( 3.5) 31/1313 ( 2.4) 1.491 (0.934, 2.382) 0.0944 Dyspnoea 9/1288 ( 0.7) 6/1313 ( 0.5) 0.933 (0.432, 2.016) 0.8609 Hypotension 48/1288 ( 3.7) 72/1313 ( 5.5) 0.663 (0.453, 0.970) 0.0341 Hypoventilation 11/1288 ( 0.9) 25/1313 ( 1.9) 0.447 (0.218, 0.915) 0.0277 Hypoxia 43/1288 ( 3.3) 55/1313 ( 4.2) 0.798 (0.527, 1.208) 0.2863 Ileus 16/1288 ( 1.2) 10/1313 ( 0.8) 1.611 (0.717, 3.618) 0.2482 Nausea 519/1288 (40.3) 584/1313 (44.5) 0.848 (0.690, 1.042) 0.1157 Pruritus 71/1288 ( 5.5) 124/1313 ( 9.4) 0.556 (0.409, 0.754) 0.0002 Somnolence 13/1288 ( 1.0) 24/1313 ( 1.8) 0.888 (0.524, 1.506) 0.6596 Tachycardia 18/1288 ( 1.4) 37/1313 ( 2.8) 0.503 (0.282, 0.897) 0.0199 Urinary retention 21/1288 ( 1.6) 35/1313 ( 2.7) 0.609 (0.350, 1.059) 0.0791 Vomiting 167/1288 (13.0) 163/1313 (12.4) 1.055 (0.829, 1.341) 0.6655 ) 4) ) 4) ) ) ) ) ) ) 4) ) ) 7) 4) Morphine IV PCA Better IONSYS™ Better Disruptive innovation Safety: Opioid-related adverse events Data on File. The Medicines Company. IONSYS™ is currently not approved for commercial use in any market. |
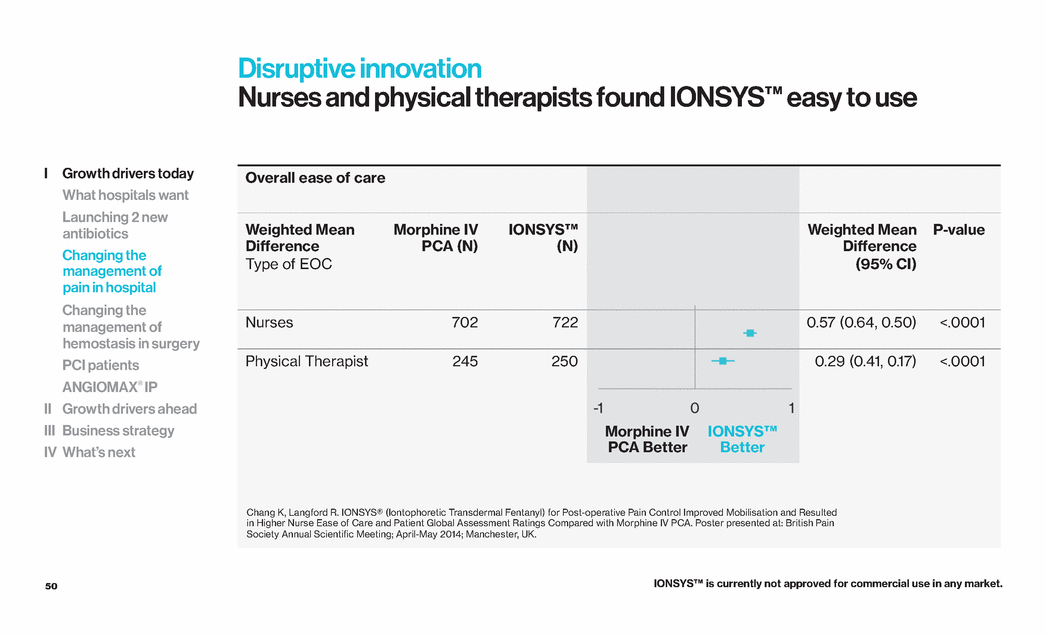
| 50 Overall ease of care Weighted Mean Difference Type of EOC Morphine IV PCA (N) IONSYS™ (N) Weighted Mean Difference (95% CI) P-value Nurses 702 722 0.57 (0.64, 0.50) <.0001 Physical Therapist 245 250 0.29 (0.41, 0.17) <.0001 Disruptive innovation Nurses and physical therapists found IONSYS™ easy to use Morphine IV PCA Better IONSYS™ Better 2 0 0 Chang K, Langford R. IONSYS® (Iontophoretic Transdermal Fentanyl) for Post-operative Pain Control Improved Mobilisation and Resulted in Higher Nurse Ease of Care and Patient Global Assessment Ratings Compared with Morphine IV PCA. Poster presented at: British Pain Society Annual Scientific Meeting; April-May 2014; Manchester, UK. IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
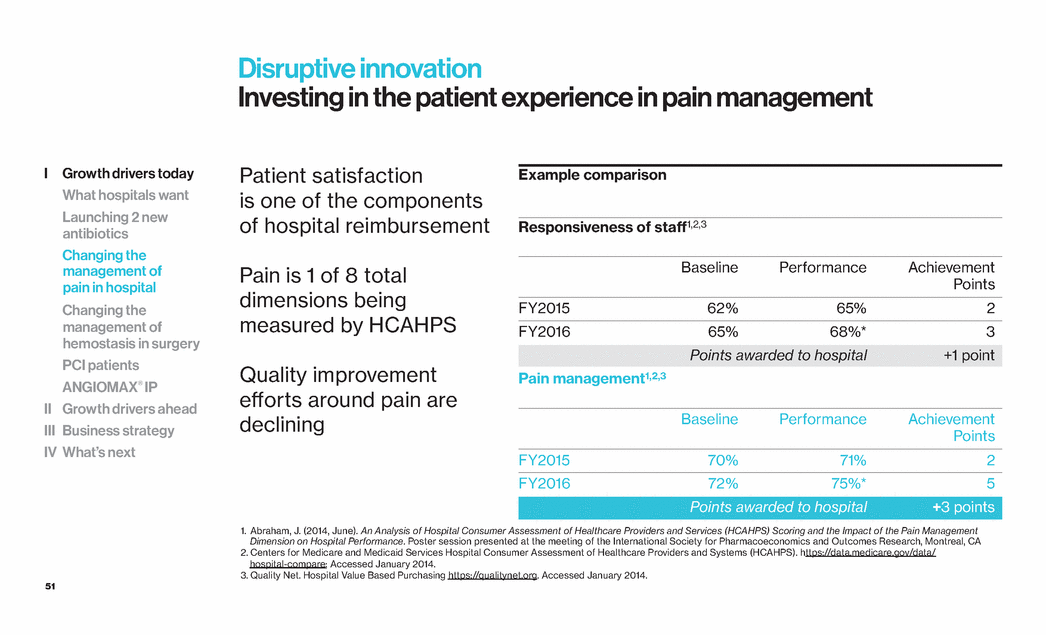
| 51 Disruptive innovation Investing in the patient experience in pain management Patient satisfaction is one of the components of hospital reimbursement Pain is 1 of 8 total dimensions being measured by HCAHPS Quality improvement efforts around pain are declining 1. Abraham, J. (2014, June). An Analysis of Hospital Consumer Assessment of Healthcare Providers and Services (HCAHPS) Scoring and the Impact of the Pain Management Dimension on Hospital Performance. Poster session presented at the meeting of the International Society for Pharmacoeconomics and Outcomes Research, Montreal, CA 2. Centers for Medicare and Medicaid Services Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS). https://data.medicare.gov/data/ hospital-compare; Accessed January 2014. 3. Quality Net. Hospital Value Based Purchasing https://qualitynet.org. Accessed January 2014. Example comparison Responsiveness of staff1,2,3 Baseline Performance Achievement Points FY2015 62% 65% 2 FY2016 65% 68%* 3 Points awarded to hospital +1 point Pain management1,2,3 Baseline Performance Achievement Points FY2015 70% 71% 2 FY2016 72% 75%* 5 Points awarded to hospital +3 points I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 52 Disruptive innovation IONSYS™ potential to reduce hospital costs Harmful IV PCA medication errors cost $6,9432,7 per event Harmful IVPCA device errors cost $7,2812,7 per event IONSYS™ potential to reduce AEs, pump and programming errors, and pain management related nursing activities. IONSYS™ 1,2,3,4,9,10 IV PCA 1,2,3,4,5,6,8,9,10,11 Savings Patient outcomes: Safety ~$1,100 ~$1,350 ~$250 Process improvement outcomes ~$25 ~$75 ~$50 Disposable and capital equipment efficiencies $0 ~$85 ~$85 1. Data on file. The Medicines Company. Output from MDCO model. 2. Meissner B, Nelson W, Hicks RW, Sikirica V, Gagne J, Schein J. The Rate and Costs Attributable to Intravenous Patient-Controlled Analgesia Errors. Hosp Pharm. 2009;44(4):312-324. 3. (Wages) U.S. Bureau of Labor Statistics. May 2012 National Occupational Employment and Wage Estimates (United States)-Healthcare Practitioner and Technical Occupations. http://www.bls.gov/oes/current/oes_nat.htm#29-0000. Accessed January 16, 2014. 4. (Benefits) U.S. Bureau of Labor Statistics. Employer Costs for Employee Compensation-December 2013. Last updated March 12, 2014. http://www.bls.gov/news.release/ ecec.nr0.htm. Accessed March 26, 2014. 5. Wholesaler provided costs. 6. Wolters Kluwer Health Inc. Medi-Span Price Rx Basic 2014. https://pricerx.medispan.com/Login.aspx. Accessed March 12, 2014. 7. Inflated from 2006 USD to 2013 USD using the medical care component of CPI (inflation factor: 1.0487). 8. U.S. Internal Revenue Service. Publication 946. How to Depreciate Property. 2012. U.S. Department of the Treasury, pp. 37. http://www.irs.gov/pub/irs-pdf/p946.pdf. Accessed January 16, 2014. 9. Mordin M, Anastassopoulos K, van Breda A, et al. Clinical staff resource use with intravenous patient-controlled analgesia in acute postoperative pain management: results from a multicenter, prospective, observational study. J Perianesth Nurs. 2007;22(4):243-255. 10. Evans C, Schein J, Nelson W, et al. Improving patient and nurse outcomes: a comparison of nurse tasks and time associated with two patient-controlled analgesia modalities using delphi panels. Pain Manag Nurs. 2007;8(2):86-95. 11. Hutchison RW, Anastassopoulos K, Vallow S, et al. Intravenous Patient-Controlled Analgesia Pump and Reservoir Logistics: Results From a Multicenter Questionnaire. Hosp Pharm. 2006;42(11):1036-1044. IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 53 Disruptive innovation Manufacturing systems and commercial scale production established Lakewood, NJ facility Capacity for 2M units per year at launch Expansion equipment available for more than 15M units per year More than $40M invested capital by Alza/ Johnson & Johnson prior to our acquisition of IONSYS™ IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
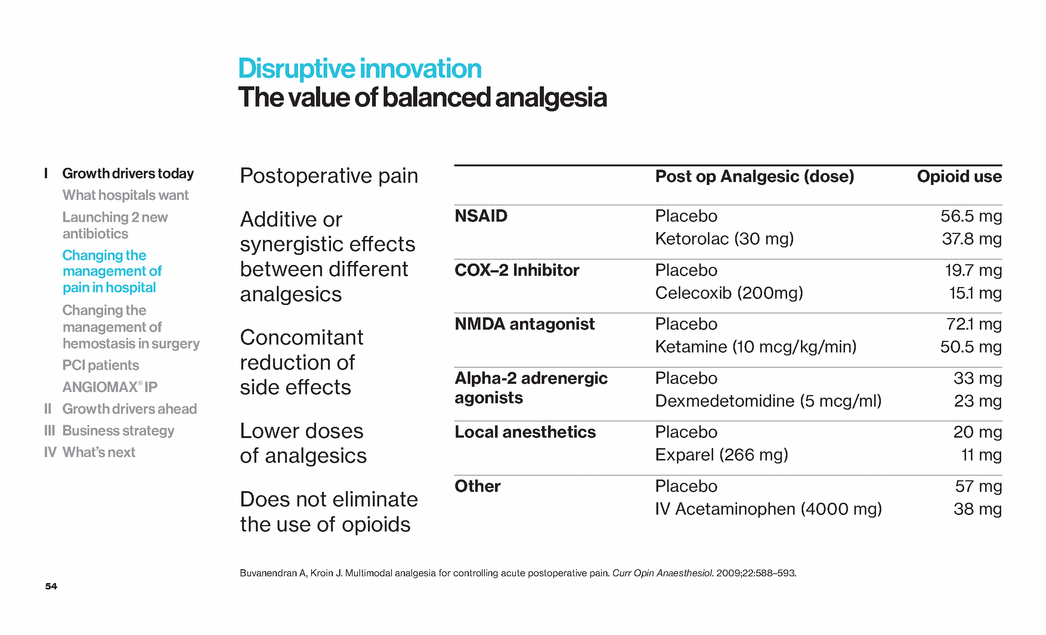
| 54 Disruptive innovation The value of balanced analgesia Post op Analgesic (dose) Opioid use NSAID Placebo 56.5 mg Ketorolac (30 mg) 37.8 mg COX–2 Inhibitor Placebo 19.7 mg Celecoxib (200mg) 15.1 mg NMDA antagonist Placebo 72.1 mg Ketamine (10 mcg/kg/min) 50.5 mg Alpha-2 adrenergic agonists Placebo 33 mg Dexmedetomidine (5 mcg/ml) 23 mg Local anesthetics Placebo 20 mg Exparel (266 mg) 11 mg Other Placebo 57 mg IV Acetaminophen (4000 mg) 38 mg Postoperative pain Additive or synergistic effects between different analgesics Concomitant reduction of side effects Lower doses of analgesics Does not eliminate the use of opioids Buvanendran A, Kroin J. Multimodal analgesia for controlling acute postoperative pain. Curr Opin Anaesthesiol. 2009;22:588–593. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
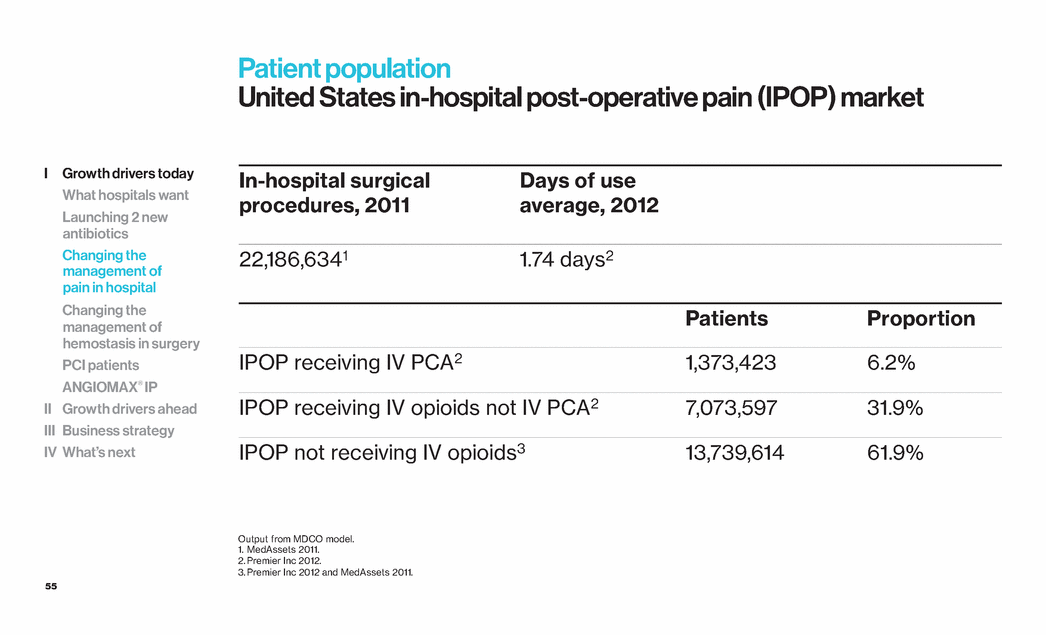
| 55 Patient population United States in-hospital post-operative pain (IPOP) market In-hospital surgical procedures, 2011 Days of use average, 2012 22,186,6341 1.74 days2 Patients Proportion IPOP receiving IV PCA2 1,373,423 6.2% IPOP receiving IV opioids not IV PCA2 7,073,597 31.9% IPOP not receiving IV opioids3 13,739,614 61.9% Output from MDCO model. 1. MedAssets 2011. 2. Premier Inc 2012. 3. Premier Inc 2012 and MedAssets 2011. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
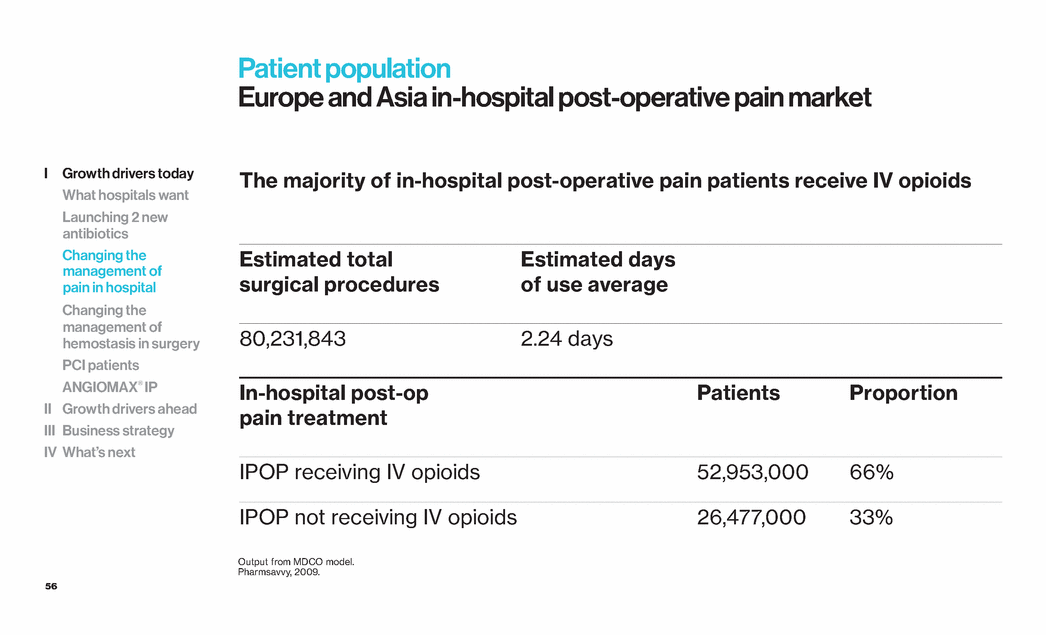
| 56 Patient population Europe and Asia in-hospital post-operative pain market The majority of in-hospital post-operative pain patients receive IV opioids Estimated total surgical procedures Estimated days of use average 80,231,843 2.24 days In-hospital post-op pain treatment Patients Proportion IPOP receiving IV opioids 52,953,000 66% IPOP not receiving IV opioids 26,477,000 33% Output from MDCO model. Pharmsavvy, 2009. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 57 United States FDA sNDA • Submitted 30 June 2014 • Accepted for filing 11 Sept 2014 • PDUFA action date 30 April 2015 European CHMP MAA • Submitted 3 Sept 2014 • Accepted for filing 24 Sept 2014 • Preliminary opinion anticipated 22 January 2015 Disruptive innovation Regulatory submissions complete and reviews underway I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 58 Publications 2 publications submitted to date, with 4 additional scheduled for submission in 2014 Clinical trial Pediatric study expected to begin Q1 2015 Early cycle reviews EMA January 2015 PDUFA April 2015 Disruptive innovation News flow I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 59 Disruptive innovation The potential future of post-operative pain management IONSYS™ is currently not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| Changing the management of hemostasis in surgery: RECOTHROM®, RAPLIXA™ and PREVELEAK™ Jason Campagna The Medicines Company |
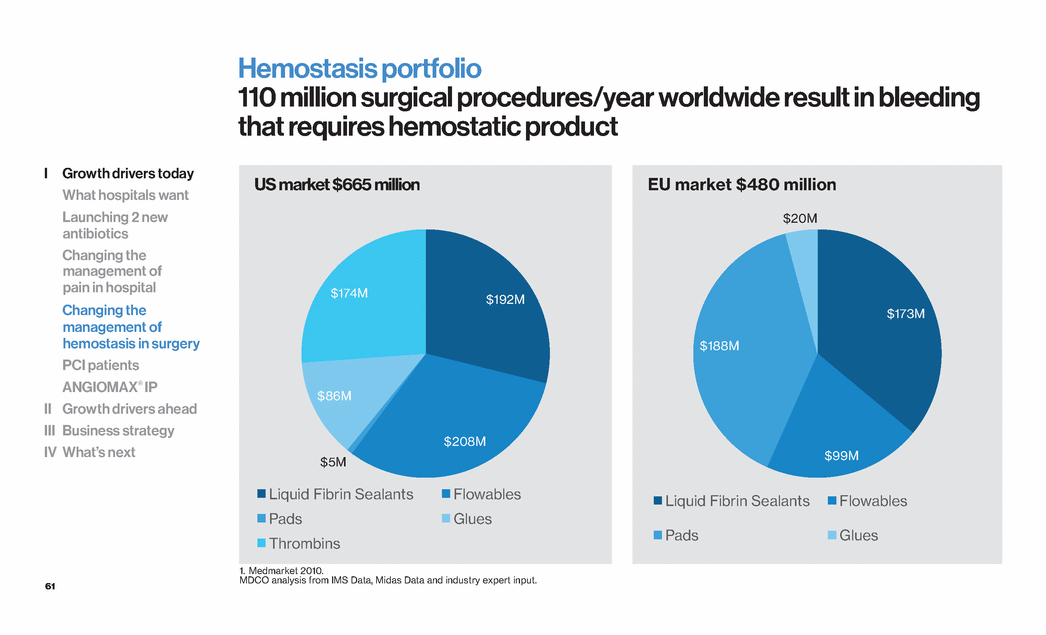
| 61 Hemostasis portfolio 110 million surgical procedures/year worldwide result in bleeding that requires hemostatic product $192M $208M $5M $86M $174M US market $665 million Liquid Fibrin Sealants Flowables Pads Glues Thrombins $173M $99M $188M $20M EU market $480 million Liquid Fibrin Sealants Flowables Pads Glues 1. Medmarket 2010. MDCO analysis from IMS Data, Midas Data and industry expert input. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
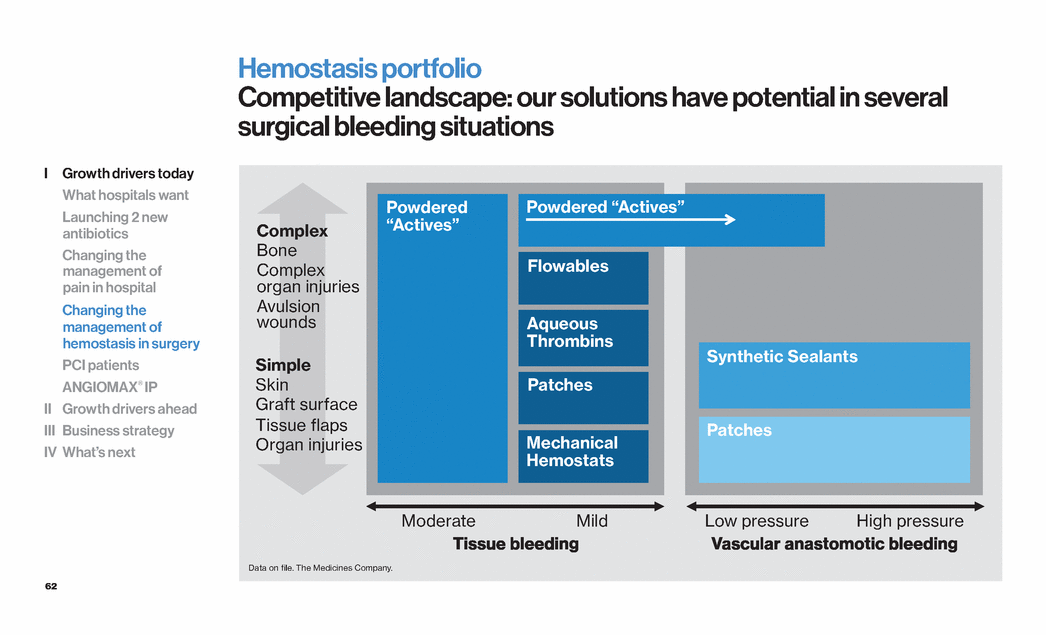
| 62 Tissue bleeding Data on file. The Medicines Company. Simple Skin Graft surface Tissue flaps Organ injuries Complex Bone Complex organ injuries Avulsion wounds Vascular anastomotic bleeding Moderate Mild Low pressure High pressure Powdered “Actives” Flowables Mechanical Hemostats Patches Synthetic Sealants Aqueous Thrombins Hemostasis portfolio Competitive landscape: our solutions have potential in several surgical bleeding situations Powdered “Actives” Patches I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
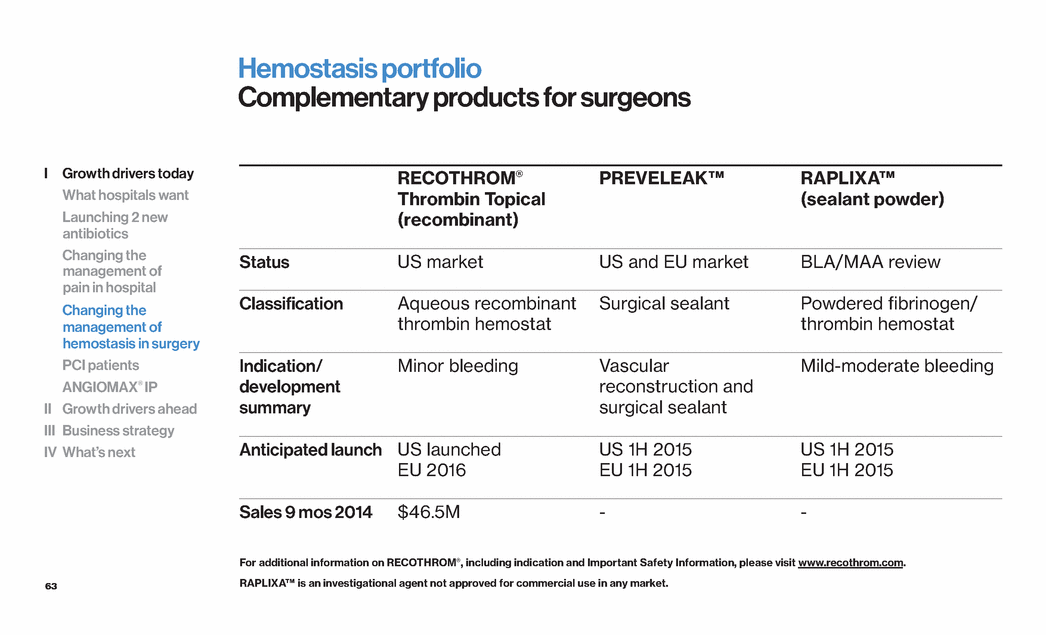
| 63 Hemostasis portfolio Complementary products for surgeons RECOTHROM® Thrombin Topical (recombinant) PREVELEAK™ RAPLIXA™ (sealant powder) Status US market US and EU market BLA/MAA review Classification Aqueous recombinant thrombin hemostat Surgical sealant Powdered fibrinogen/ thrombin hemostat Indication/ development summary Minor bleeding Vascular reconstruction and surgical sealant Mild-moderate bleeding Anticipated launch US launched EU 2016 US 1H 2015 EU 1H 2015 US 1H 2015 EU 1H 2015 Sales 9 mos 2014 $46.5M - - RAPLIXA™ is an investigational agent not approved for commercial use in any market. For additional information on RECOTHROM®, including indication and Important Safety Information, please visit www.recothrom.com. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| PCI patients: Cangrelor and ANGIOMAX® Makis Deliargyris The Medicines Company |
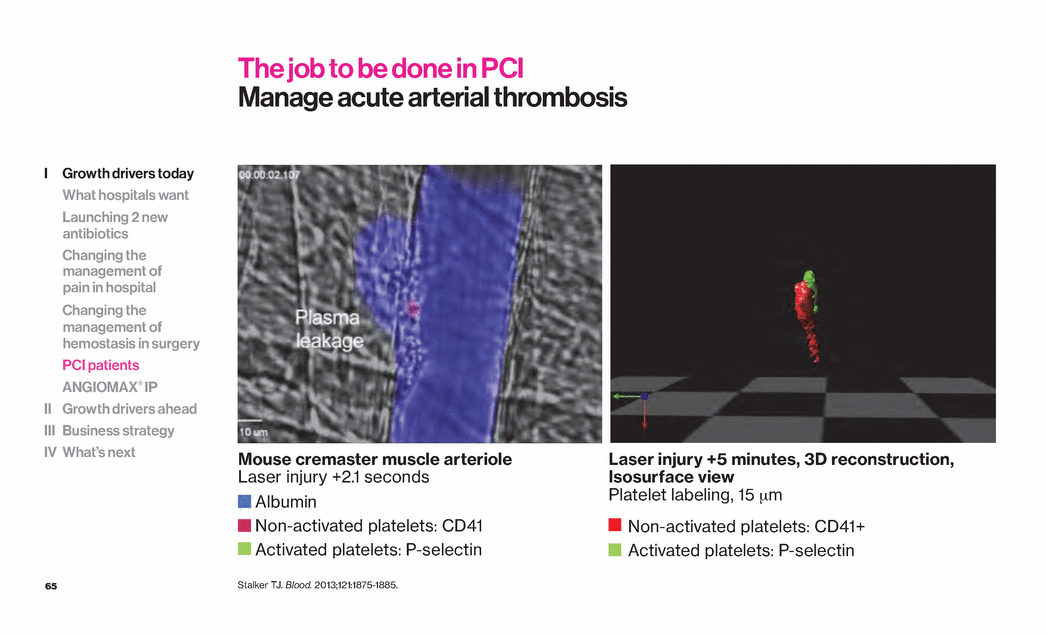
| 65 The job to be done in PCI Manage acute arterial thrombosis Stalker TJ. Blood. 2013;121:1875-1885. Mouse cremaster muscle arteriole Laser injury +2.1 seconds Laser injury +5 minutes, 3D reconstruction, Isosurface view Platelet labeling, 15 µm Albumin Non-activated platelets: CD41 Activated platelets: P-selectin Non-activated platelets: CD41+ Activated platelets: P-selectin I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
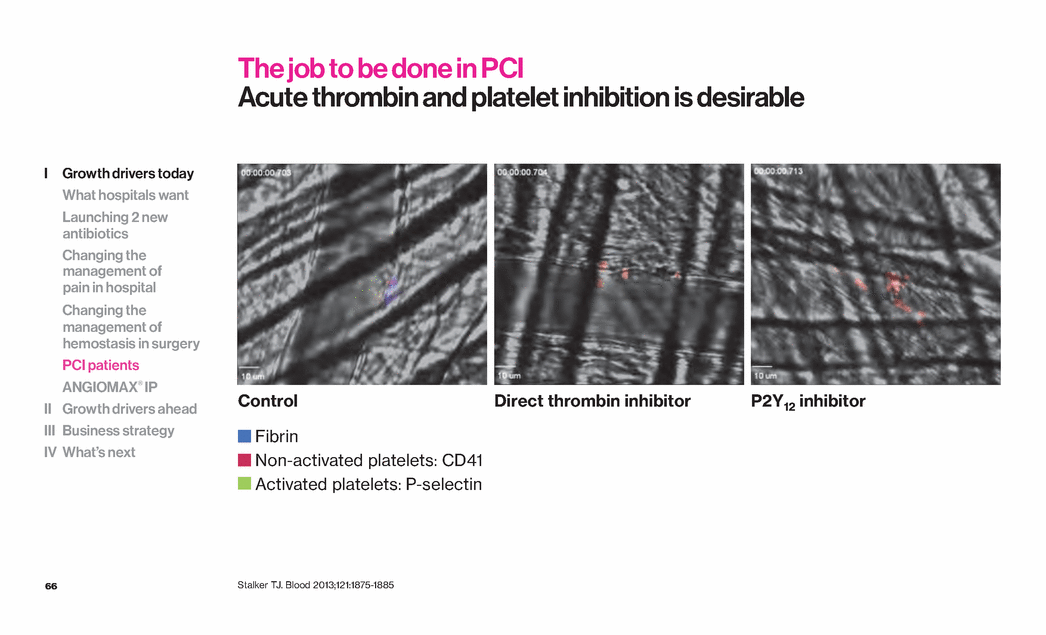
| 66 The job to be done in PCI Acute thrombin and platelet inhibition is desirable Control Stalker TJ. Blood 2013;121:1875-1885 Direct thrombin inhibitor P2Y12 inhibitor Fibrin Non-activated platelets: CD41 Activated platelets: P-selectin I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
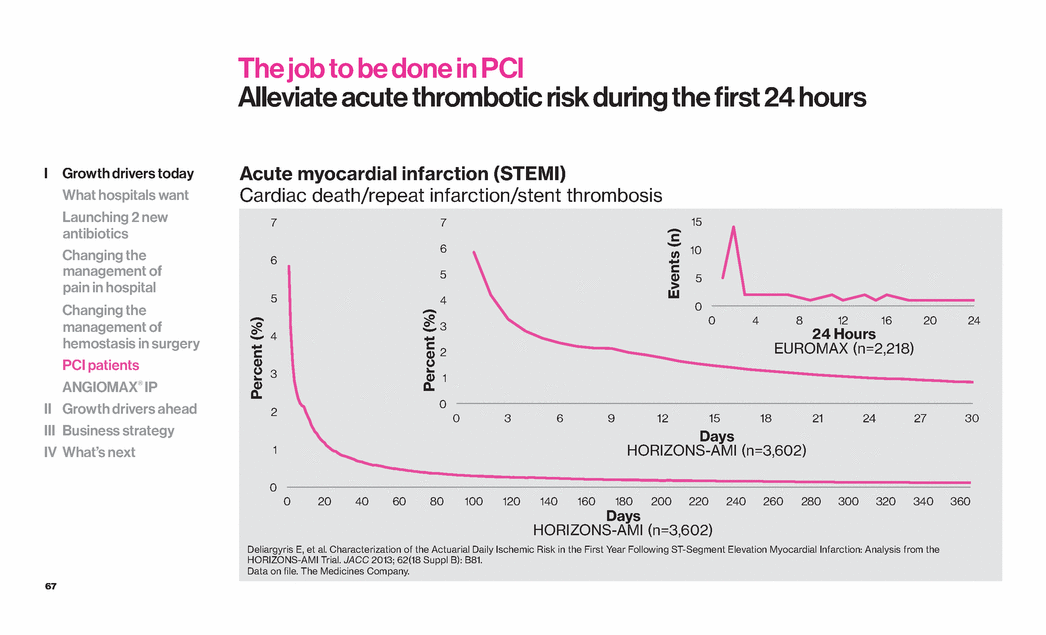
| 67 Acute myocardial infarction (STEMI) Cardiac death/repeat infarction/stent thrombosis The job to be done in PCI Alleviate acute thrombotic risk during the first 24 hours 0 1 2 3 4 5 6 7 0 20 40 60 80 100 120 140 160 180 200 220 240 260 280 300 320 340 360 Percent (%) Days HORIZONS-AMI (n=3,602) 0 1 2 3 4 5 6 7 0 3 6 9 12 15 18 21 24 27 30 Days HORIZONS-AMI (n=3,602) Percent (%) 0 5 10 15 0 4 8 12 16 20 24 24 Hours EUROMAX (n=2,218) Events (n) Deliargyris E, et al. Characterization of the Actuarial Daily Ischemic Risk in the First Year Following ST-Segment Elevation Myocardial Infarction: Analysis from the HORIZONS-AMI Trial. JACC 2013; 62(18 Suppl B): B81. Data on file. The Medicines Company. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
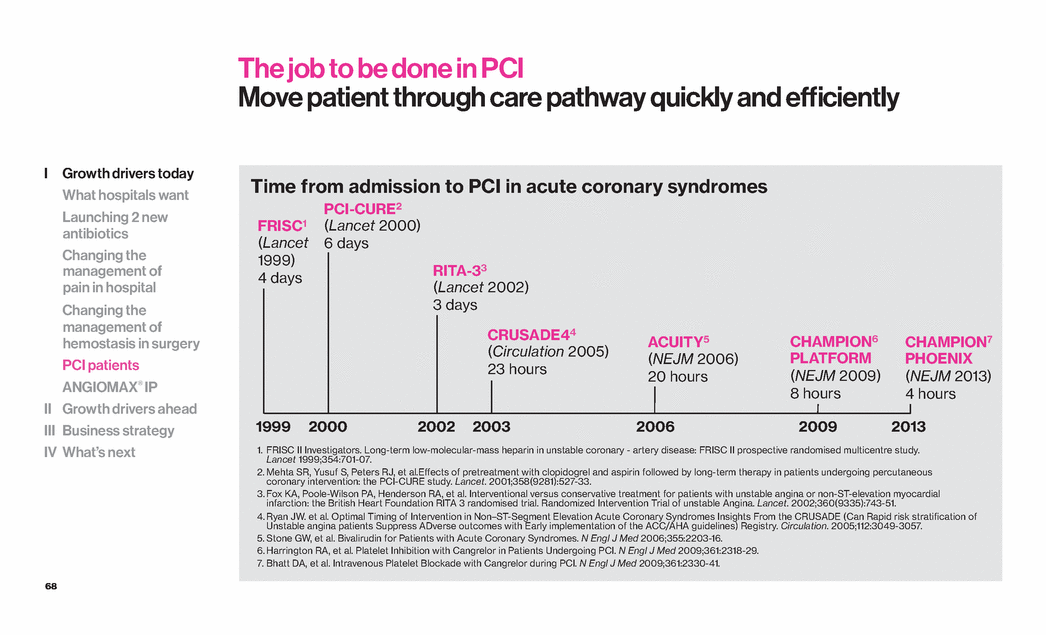
| 68 The job to be done in PCI Move patient through care pathway quickly and efficiently FRISC1 (Lancet 1999) 4 days RITA-33 (Lancet 2002) 3 days CRUSADE44 (Circulation 2005) 23 hours ACUITY5 (NEJM 2006) 20 hours CHAMPION7 PHOENIX (NEJM 2013) 4 hours CHAMPION6 PLATFORM (NEJM 2009) 8 hours PCI-CURE2 (Lancet 2000) 6 days 1999 2002 2003 2006 2000 2009 2013 Time from admission to PCI in acute coronary syndromes 1. FRISC II Investigators. Long-term low-molecular-mass heparin in unstable coronary - artery disease: FRISC II prospective randomised multicentre study. Lancet 1999;354:701-07. 2. Mehta SR, Yusuf S, Peters RJ, et al.Effects of pretreatment with clopidogrel and aspirin followed by long-term therapy in patients undergoing percutaneous coronary intervention: the PCI-CURE study. Lancet. 2001;358(9281):527-33. 3. Fox KA, Poole-Wilson PA, Henderson RA, et al. Interventional versus conservative treatment for patients with unstable angina or non-ST-elevation myocardial infarction: the British Heart Foundation RITA 3 randomised trial. Randomized Intervention Trial of unstable Angina. Lancet. 2002;360(9335):743-51. 4. Ryan JW. et al. Optimal Timing of Intervention in Non–ST-Segment Elevation Acute Coronary Syndromes Insights From the CRUSADE (Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines) Registry. Circulation. 2005;112:3049-3057. 5. Stone GW, et al. Bivalirudin for Patients with Acute Coronary Syndromes. N Engl J Med 2006;355:2203-16. 6. Harrington RA, et al. Platelet Inhibition with Cangrelor in Patients Undergoing PCI. N Engl J Med 2009;361:2318-29. 7. Bhatt DA, et al. Intravenous Platelet Blockade with Cangrelor during PCI. N Engl J Med 2009;361:2330-41. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
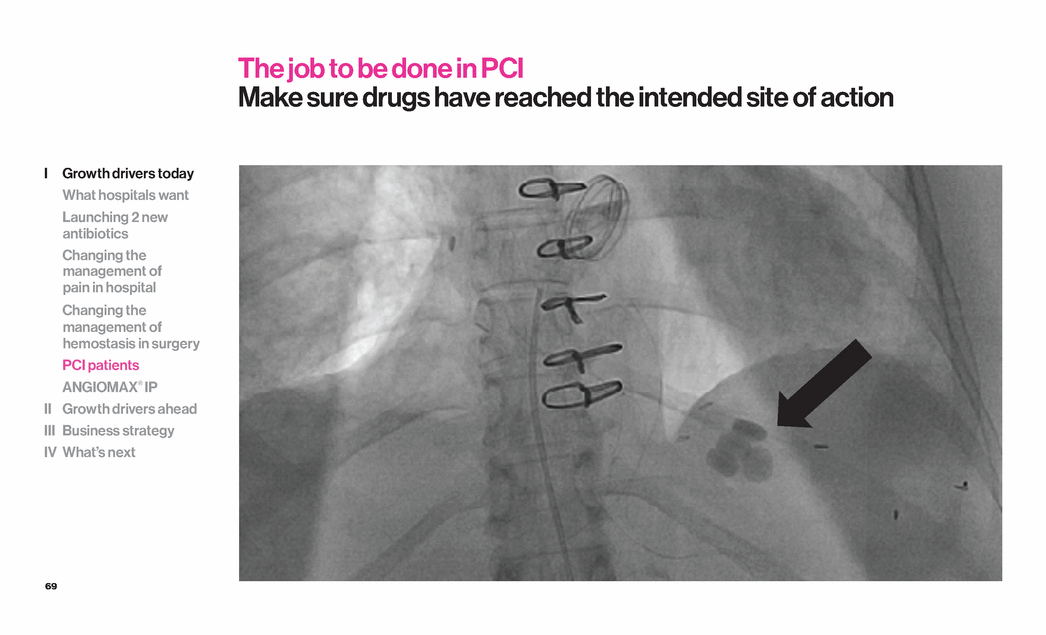
| 69 The job to be done in PCI Make sure drugs have reached the intended site of action I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
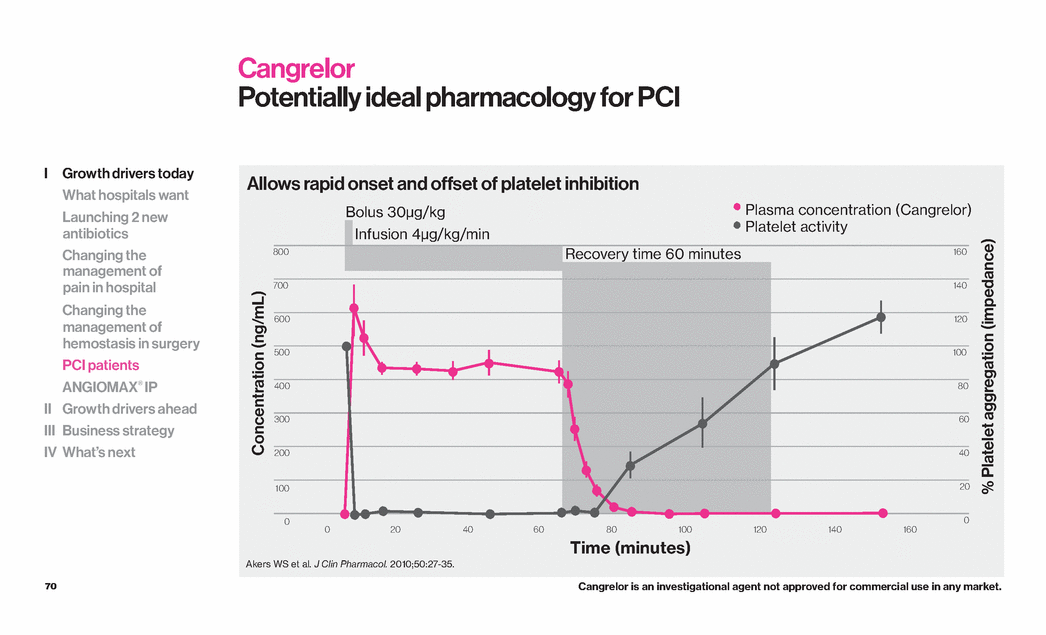
| 70 Allows rapid onset and offset of platelet inhibition 800 700 600 500 400 300 200 160 140 120 100 80 60 40 20 0 0 Time (minutes) Bolus 30µg/kg Infusion 4µg/kg/min Recovery time 60 minutes Concentration (ng/mL) % Platelet aggregation (impedance) Plasma concentration (Cangrelor) Platelet activity Akers WS et al. J Clin Pharmacol. 2010;50:27-35. 20 40 60 80 100 120 140 160 Cangrelor Potentially ideal pharmacology for PCI 0 100 Cangrelor is an investigational agent not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
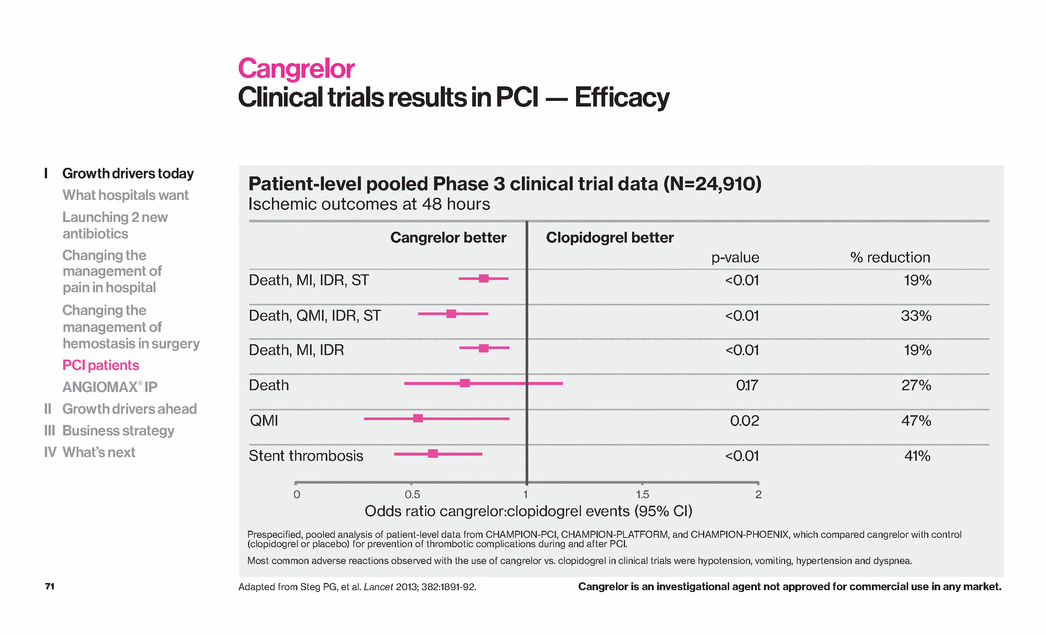
| 71 Cangrelor Clinical trials results in PCI — Efficacy Adapted from Steg PG, et al. Lancet 2013; 382:1891-92. Patient-level pooled Phase 3 clinical trial data (N=24,910) Ischemic outcomes at 48 hours Odds ratio cangrelor:clopidogrel events (95% CI) Death, MI, IDR, ST Death, QMI, IDR, ST Death, MI, IDR Death QMI Stent thrombosis 0 2 p-value <0.01 <0.01 <0.01 0.17 0.02 <0.01 % reduction 19% 33% 19% 27% 47% 41% Cangrelor better Clopidogrel better 0.5 1 1.5 Prespecified, pooled analysis of patient-level data from CHAMPION-PCI, CHAMPION-PLATFORM, and CHAMPION-PHOENIX, which compared cangrelor with control (clopidogrel or placebo) for prevention of thrombotic complications during and after PCI. Most common adverse reactions observed with the use of cangrelor vs. clopidogrel in clinical trials were hypotension, vomiting, hypertension and dyspnea. Cangrelor is an investigational agent not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
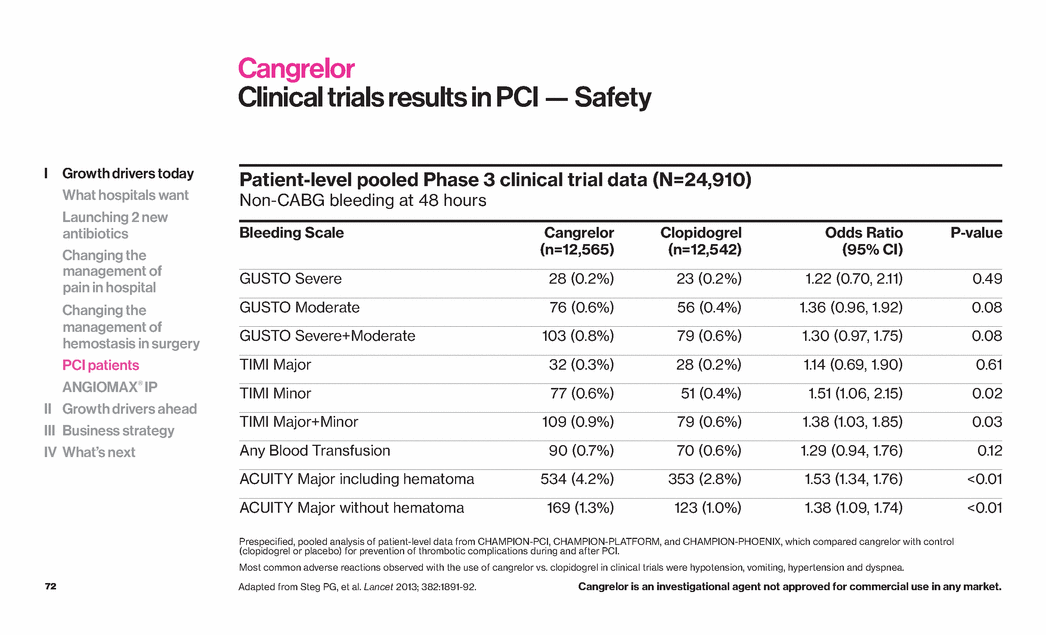
| 72 Cangrelor Clinical trials results in PCI — Safety Patient-level pooled Phase 3 clinical trial data (N=24,910) Non-CABG bleeding at 48 hours Bleeding Scale Cangrelor (n=12,565) Clopidogrel (n=12,542) Odds Ratio (95% CI) P-value GUSTO Severe 28 (0.2%) 23 (0.2%) 1.22 (0.70, 2.11) 0.49 GUSTO Moderate 76 (0.6%) 56 (0.4%) 1.36 (0.96, 1.92) 0.08 GUSTO Severe+Moderate 103 (0.8%) 79 (0.6%) 1.30 (0.97, 1.75) 0.08 TIMI Major 32 (0.3%) 28 (0.2%) 1.14 (0.69, 1.90) 0.61 TIMI Minor 77 (0.6%) 51 (0.4%) 1.51 (1.06, 2.15) 0.02 TIMI Major+Minor 109 (0.9%) 79 (0.6%) 1.38 (1.03, 1.85) 0.03 Any Blood Transfusion 90 (0.7%) 70 (0.6%) 1.29 (0.94, 1.76) 0.12 ACUITY Major including hematoma 534 (4.2%) 353 (2.8%) 1.53 (1.34, 1.76) <0.01 ACUITY Major without hematoma 169 (1.3%) 123 (1.0%) 1.38 (1.09, 1.74) <0.01 Adapted from Steg PG, et al. Lancet 2013; 382:1891-92. Prespecified, pooled analysis of patient-level data from CHAMPION-PCI, CHAMPION-PLATFORM, and CHAMPION-PHOENIX, which compared cangrelor with control (clopidogrel or placebo) for prevention of thrombotic complications during and after PCI. Most common adverse reactions observed with the use of cangrelor vs. clopidogrel in clinical trials were hypotension, vomiting, hypertension and dyspnea. Cangrelor is an investigational agent not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 73 Cangrelor Active, ongoing regulatory review United States (FDA)—NDA Status Europe (EMA)—MAA Status Complete Response Letter received on April 30, 2014 Resubmission planned before year-end Anticipated • Advisory Committee Meeting • Six-month review Day 180 List of Outstanding Issues (LOI) – September, 2014 Cardiovascular Scientific Advisory Group (SAG) Meeting Scheduled Possible CHMP Opinion in 1Q2015, EC Decision anticipated in 2Q2015 Cangrelor is an investigational agent not approved for commercial use in any market. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
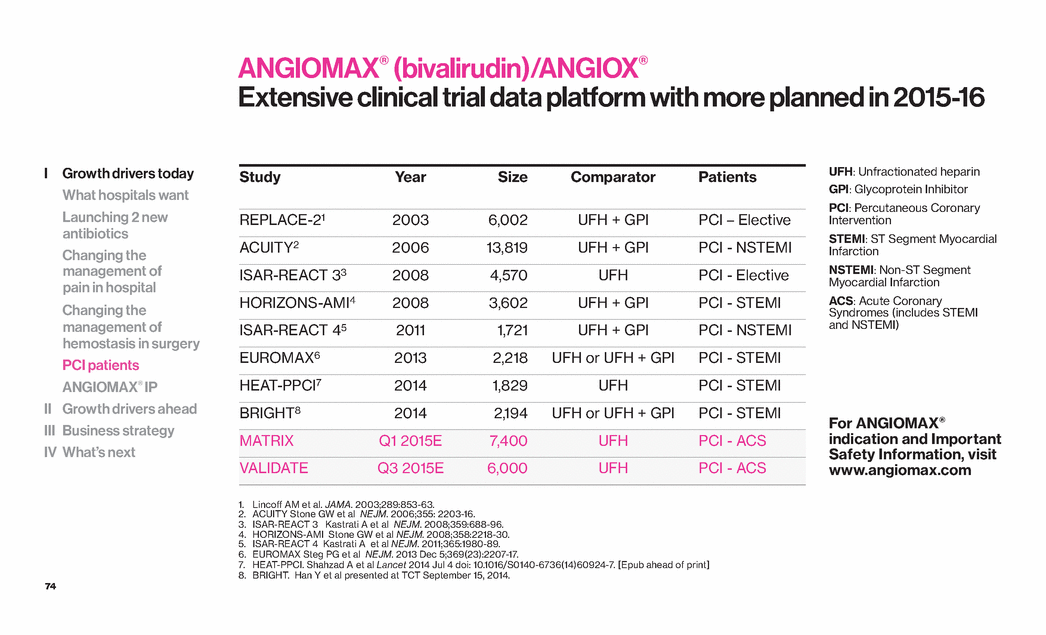
| 74 ANGIOMAX® (bivalirudin)/ANGIOX® Extensive clinical trial data platform with more planned in 2015-16 Study Year Size Comparator Patients REPLACE-21 2003 6,002 UFH + GPI PCI – Elective ACUITY2 2006 13,819 UFH + GPI PCI - NSTEMI ISAR-REACT 33 2008 4,570 UFH PCI - Elective HORIZONS-AMI4 2008 3,602 UFH + GPI PCI - STEMI ISAR-REACT 45 2011 1,721 UFH + GPI PCI - NSTEMI EUROMAX6 2013 2,218 UFH or UFH + GPI PCI - STEMI HEAT-PPCI7 2014 1,829 UFH PCI - STEMI BRIGHT8 2014 2,194 UFH or UFH + GPI PCI - STEMI MATRIX Q1 2015E 7,400 UFH PCI - ACS VALIDATE Q3 2015E 6,000 UFH PCI - ACS UFH: Unfractionated heparin GPI: Glycoprotein Inhibitor PCI: Percutaneous Coronary Intervention STEMI: ST Segment Myocardial Infarction NSTEMI: Non-ST Segment Myocardial Infarction ACS: Acute Coronary Syndromes (includes STEMI and NSTEMI) For ANGIOMAX® indication and Important Safety Information, visit www.angiomax.com 1. Lincoff AM et al. JAMA. 2003;289:853-63. 2. ACUITY Stone GW et al NEJM. 2006;355: 2203-16. 3. ISAR-REACT 3 Kastrati A et al NEJM. 2008;359:688-96. 4. HORIZONS-AMI Stone GW et al NEJM. 2008;358:2218-30. 5. ISAR-REACT 4 Kastrati A et al NEJM. 2011;365:1980-89. 6. EUROMAX Steg PG et al NEJM. 2013 Dec 5;369(23):2207-17. 7. HEAT-PPCI. Shahzad A et al Lancet 2014 Jul 4 doi: 10.1016/S0140-6736(14)60924-7. [Epub ahead of print] 8. BRIGHT. Han Y et al presented at TCT September 15, 2014. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
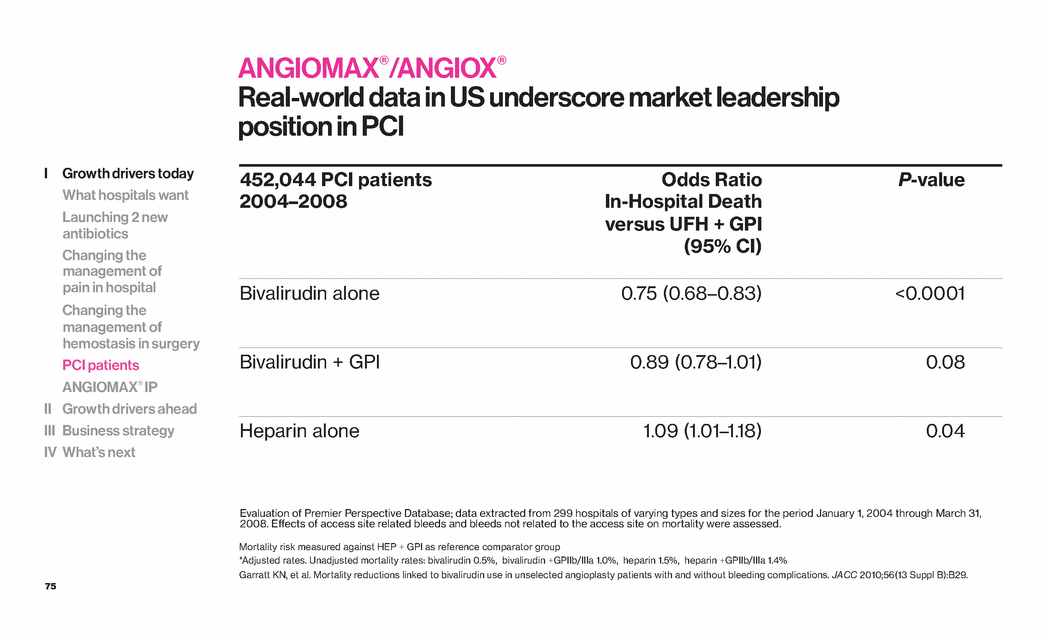
| 75 ANGIOMAX®/ANGIOX® Real-world data in US underscore market leadership position in PCI 452,044 PCI patients 2004–2008 Odds Ratio In-Hospital Death versus UFH + GPI (95% CI) P-value Bivalirudin alone 0.75 (0.68–0.83) <0.0001 Bivalirudin + GPI 0.89 (0.78–1.01) 0.08 Heparin alone 1.09 (1.01–1.18) 0.04 Mortality risk measured against HEP + GPI as reference comparator group *Adjusted rates. Unadjusted mortality rates: bivalirudin 0.5%, bivalirudin +GPIIb/IIIa 1.0%, heparin 1.5%, heparin +GPIIb/IIIa 1.4% Garratt KN, et al. Mortality reductions linked to bivalirudin use in unselected angioplasty patients with and without bleeding complications. JACC 2010;56(13 Suppl B):B29. Evaluation of Premier Perspective Database; data extracted from 299 hospitals of varying types and sizes for the period January 1, 2004 through March 31, 2008. Effects of access site related bleeds and bleeds not related to the access site on mortality were assessed. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
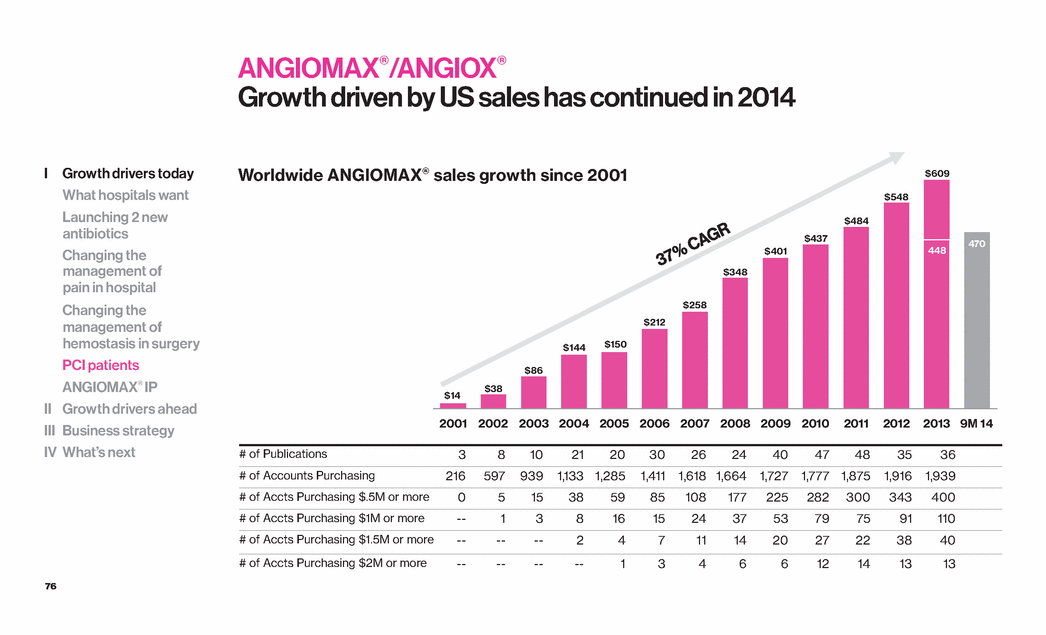
| 76 ANGIOMAX®/ANGIOX® Growth driven by US sales has continued in 2014 # of Publications 3 8 10 21 20 30 26 24 40 47 48 35 36 # of Accounts Purchasing 216 597 939 1,133 1,285 1,411 1,618 1,664 1,727 1,777 1,875 1,916 1,939 # of Accts Purchasing $.5M or more 0 5 15 38 59 85 108 177 225 282 300 343 400 # of Accts Purchasing $1M or more -- 1 3 8 16 15 24 37 53 79 75 91 110 # of Accts Purchasing $1.5M or more -- -- -- 2 4 7 11 14 20 27 22 38 40 # of Accts Purchasing $2M or more -- -- -- -- 1 3 4 6 6 12 14 13 13 $14 $38 $86 $144 $150 $212 $258 $348 $401 $437 $484 $548 448 470 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 9M 14 Worldwide ANGIOMAX® sales growth since 2001 $609 I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 77 Leon MB et al. NEJM. 2010; 363:1597-1607. ANGIOMAX®/ANGIOX® Investments in potential new indications continue Outcome TAVR (N=179) No. of pts (%) Control (N=179) No. of pts (%) P-value Vascular complications 55 (30.7) 9 (5.0) <0.001 Major vascular complications 29 (16.2) 2 (1.1) <0.001 Major bleeding 30 (16.8) 7 (3.9) <0.001 30 Days Transcutaneous aortic valve replacement (TAVR) associated with significant bleeding risk on heparin I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 78 ANGIOMAX®/ANGIOX® Investments in potential new indications continue BRAVO 2–3 Trial (n=800) Open Label ANGIOMAX® vs. heparin in TAVR ENDOMAX Trial (n=4000) Double Blind ANGIOMAX® vs. heparin in carotid stenting + peripheral interventions including critical limb ischemia 6 EU countries + Canada—35 sites 600 patients enrolled – last patient expected in mid-2015 USA – 60 sites 400 patients enrolled – last patient expected in mid-2016 Data on file. The Medicines Company. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 79 Leadership in cardiac catheterization laboratories In the United States we also promote products for our partners, AstraZeneca and Boston Scientific BRILINTA® (ticagrelor) PROMUS PREMIER™ Stent System Ticagrelor tablets Indicated for ACS Everolimus-Eluting Platinum Chromium Coronary Stent System All marks are the property of their respective owners. I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| 80 • Contemporary management of acute cardiac patients undergoing PCI requires fast and effective therapies • Cangrelor and ANGIOMAX® are both ideal for the PCI setting • Cangrelor is under active regulatory review in the US and Europe • Assuming exclusivity, ANGIOMAX® will continue to grow – New Indications • Continue leveraging cath lab leadership position with commercial partnerships In Summary I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| ANGIOMAX® IP Ed Haug Frommer Lawrence & Haug, LLP |
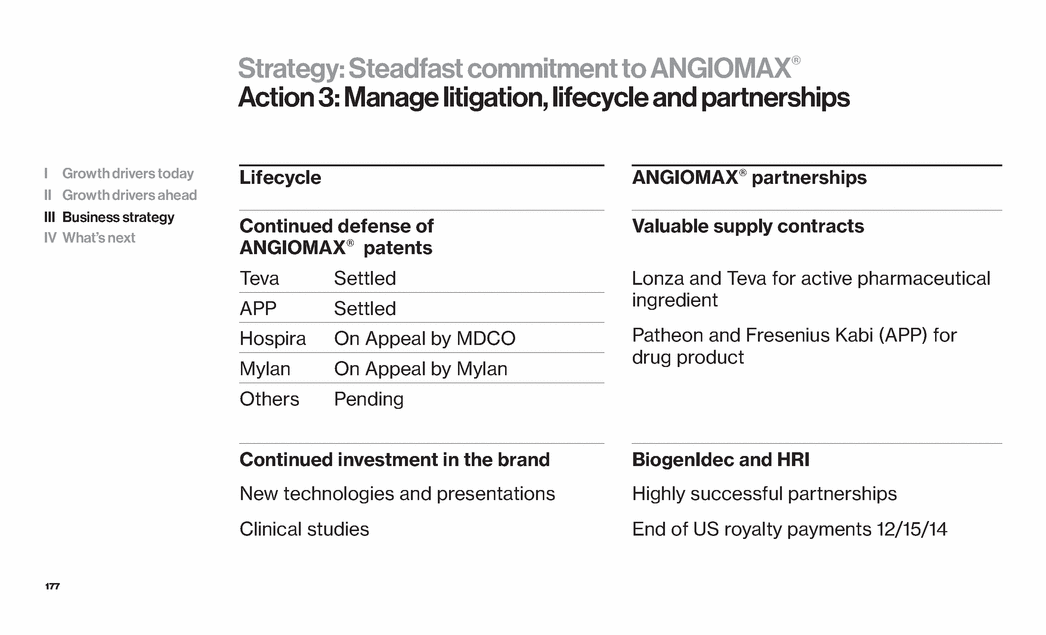
| 82 ANGIOMAX® litigations Settled or tried Teva Settled ’727 and ’343 patents valid – infringed APP Settled ’727 and ’343 patents valid – infringed Hospira On appeal ’727 and ’343 patents valid – not infringed Mylan District court decision ’727 valid and enforceable – infringed • Awaiting entry of permanent injunction ’343 not infringed (summary judgment) I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
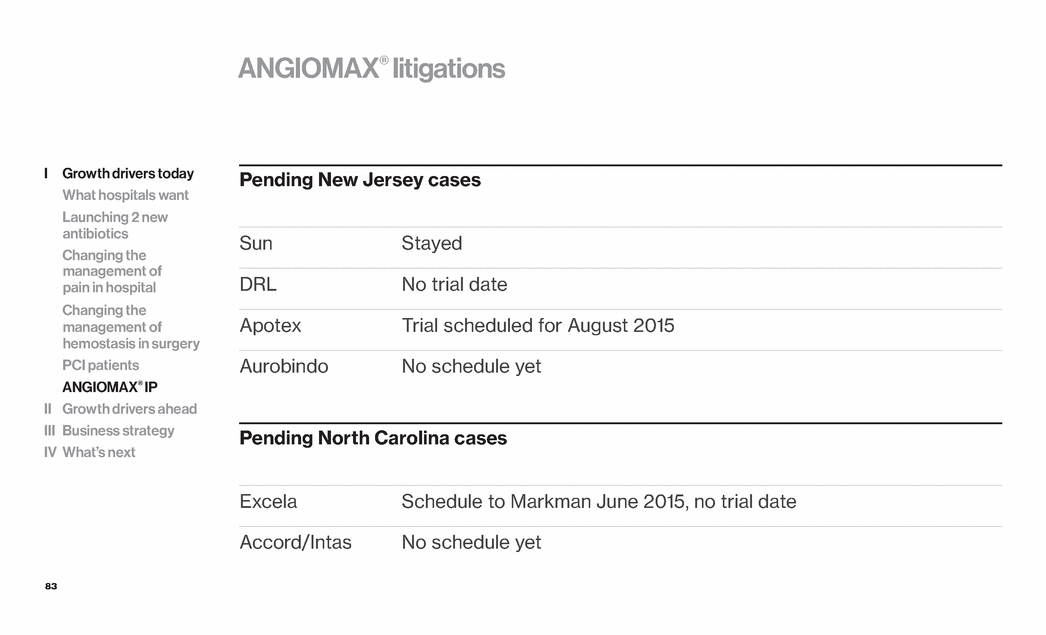
| 83 ANGIOMAX® litigations Pending New Jersey cases Sun Stayed DRL No trial date Apotex Trial scheduled for August 2015 Aurobindo No schedule yet Pending North Carolina cases Excela Schedule to Markman June 2015, no trial date Accord/Intas No schedule yet I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
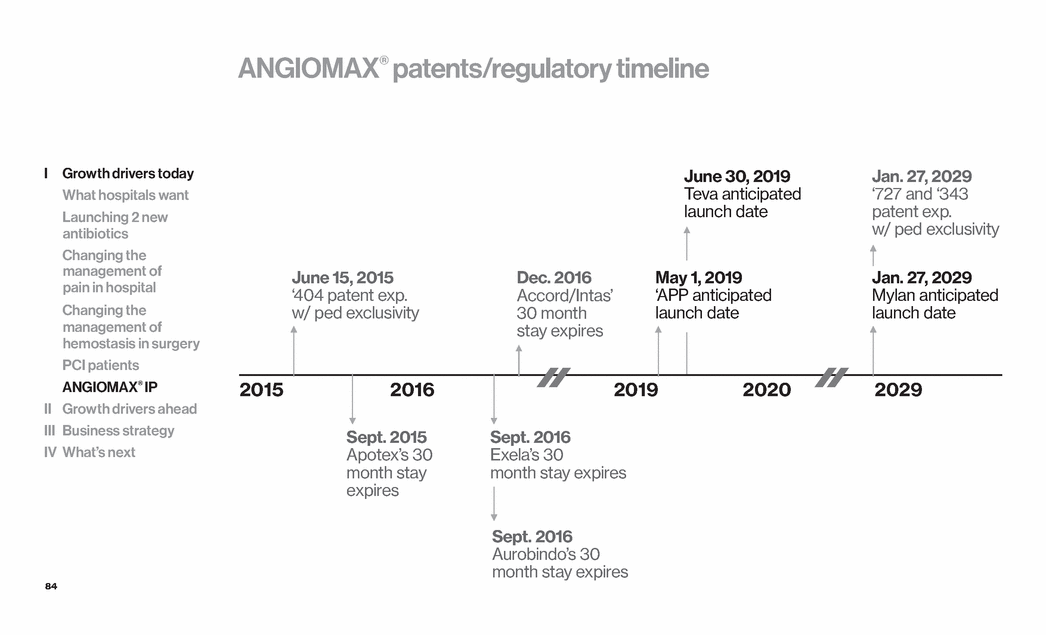
| 84 2015 2016 2019 2020 2029 ANGIOMAX® patents/regulatory timeline June 15, 2015 ‘404 patent exp. w/ ped exclusivity Sept. 2015 Apotex’s 30 month stay expires Sept. 2016 Aurobindo’s 30 month stay expires Dec. 2016 Accord/Intas’ 30 month stay expires June 30, 2019 Teva anticipated launch date Sept. 2016 Exela’s 30 month stay expires May 1, 2019 ‘APP anticipated launch date Jan. 27, 2029 ‘727 and ‘343 patent exp. w/ ped exclusivity Jan. 27, 2029 Mylan anticipated launch date I Growth drivers today What hospitals want Launching 2 new antibiotics Changing the management of pain in hospital Changing the management of hemostasis in surgery PCI patients ANGIOMAX® IP II Growth drivers ahead III Business strategy IV What’s next |
| Questions, answers and summary Clive Meanwell |
| 86 We are moving forward aggressively with our hospital portfolio Our hemostasis portfolio is making progress ORBACTIV™ and MINOCIN® IV launches are underway in US IONSYS™ is under regulatory review with potential US launch in mid-2015 Cangrelor is under active regulatory review in both US and Europe ANGIOMAX® continues to lead and grow in cardiac catheterization laboratories in the United States We continue to defend ANGIOMAX® intellectual property Growth drivers today Summary I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| BREAK |
| Growth drivers ahead Scott Johnson The Medicines Company |
| What atherosclerosis needs Greg Schwartz University of Colorado disclosures: research grants to institution — roche, sanofi,anthera,reserverlogix |
| Emerging strategies for treatment of dyslipidemia • Gregory G Schwartz, MD PhD • University of Colorado, Denver • 5 Nov 2014 Disclosures: Research grants to institution — Roche, Sanofi, Anthera, Resverlogix A cardiologist’s perspective |
| To date, no drug added to statin has been shown to provide incremental clinical benefit 1987 – 2014: 27 years since first statin approved |
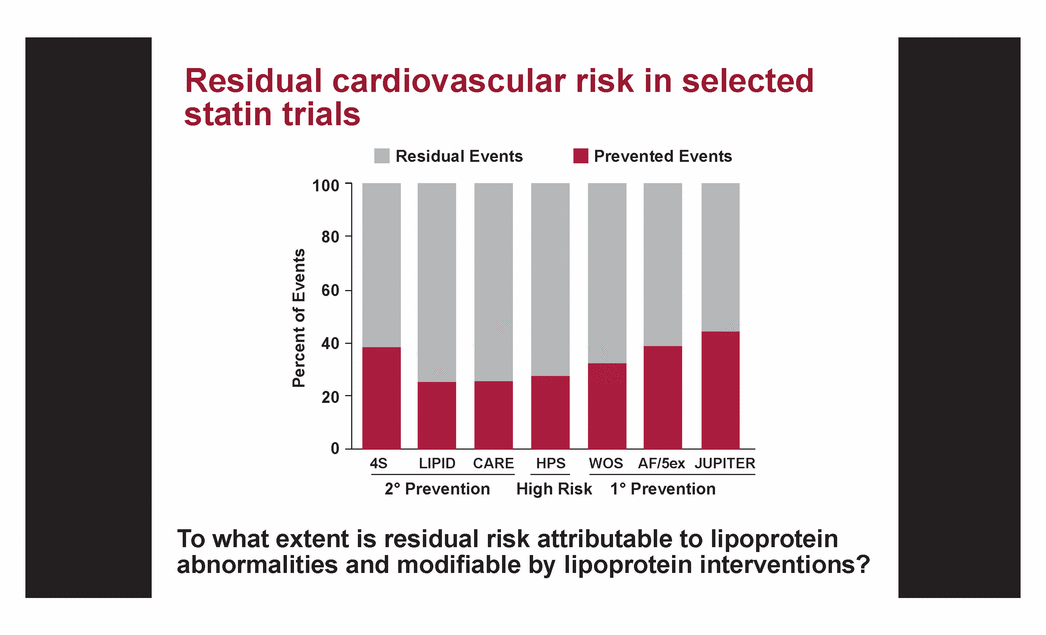
| Residual cardiovascular risk in selected statin trials To what extent is residual risk attributable to lipoprotein abnormalities and modifiable by lipoprotein interventions? 100 80 60 40 20 0 Residual Events Prevented Events 4S LIPID CARE HPS WOS AF/5ex JUPITER 2° Prevention High Risk 1° Prevention Percent of Events |
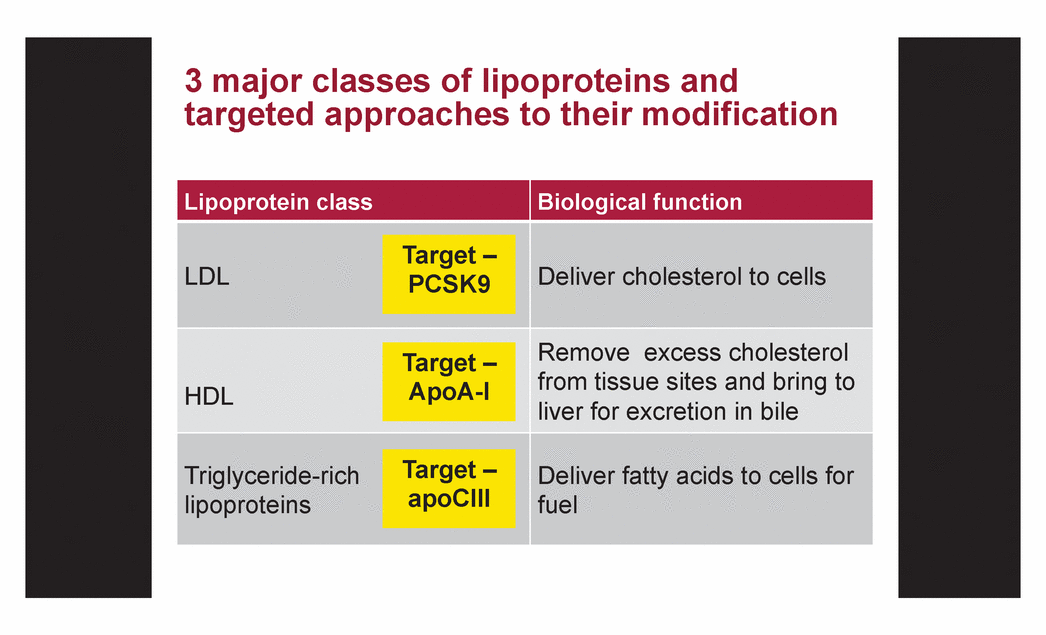
| 3 major classes of lipoproteins and targeted approaches to their modification Lipoprotein class Biological function LDL Deliver cholesterol to cells HDL Remove excess cholesterol from tissue sites and bring to liver for excretion in bile Triglyceride-rich lipoproteins Deliver fatty acids to cells for fuel Target – PCSK9 Target – ApoA-I Target – apoCIII |
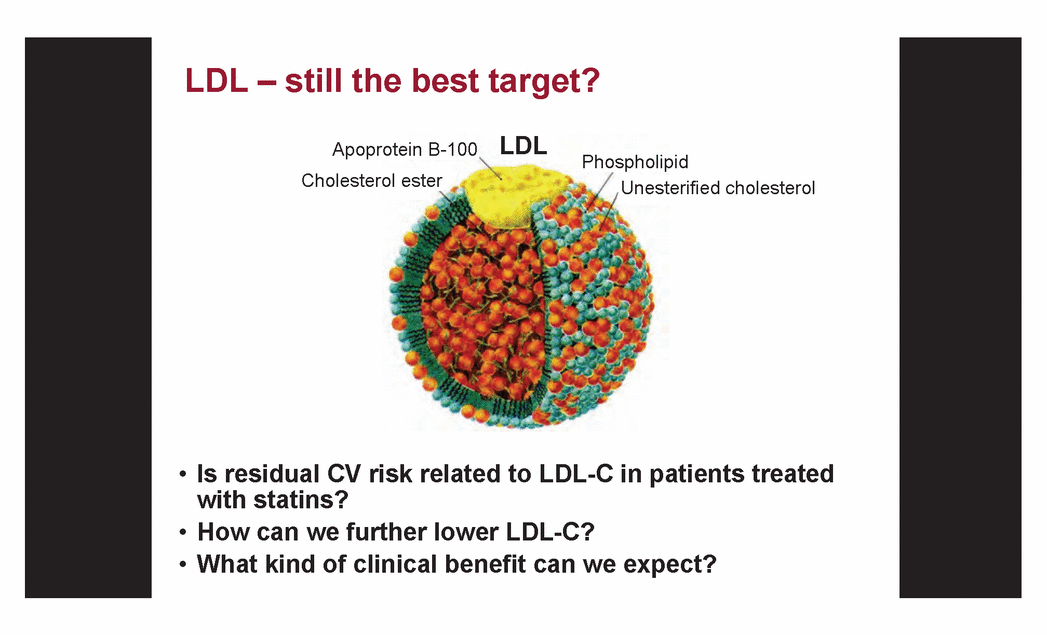
| LDL – still the best target? • Is residual CV risk related to LDL-C in patients treated with statins? • How can we further lower LDL-C? • What kind of clinical benefit can we expect? Cholesterol ester Apoprotein B-100 Phospholipid Unesterified cholesterol LDL |
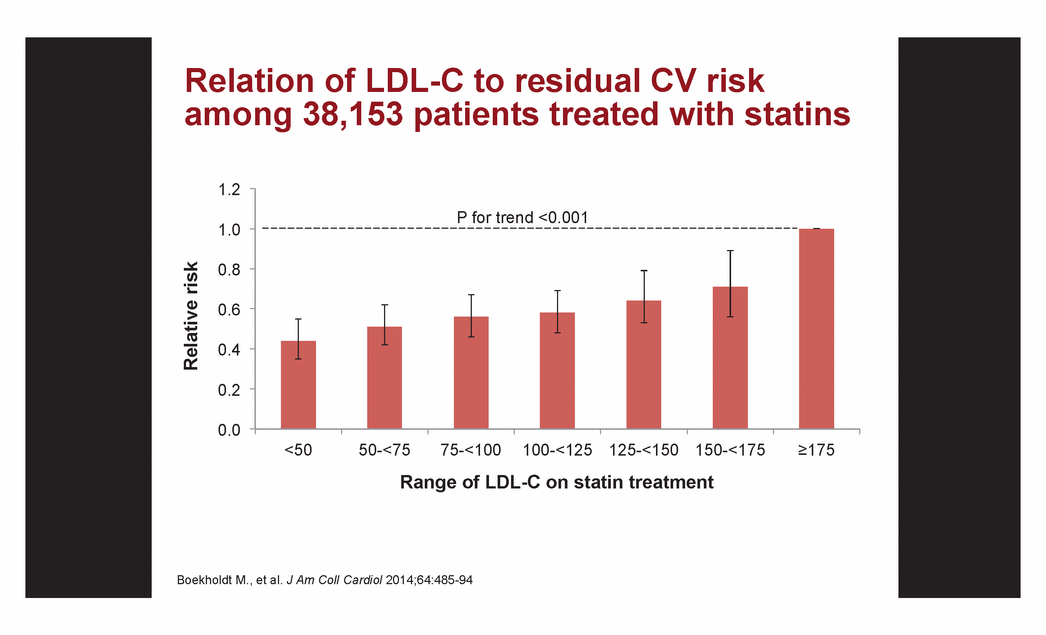
| Relation of LDL-C to residual CV risk among 38,153 patients treated with statins 0.0 0.2 0.4 0.6 0.8 1.0 1.2 <50 50-<75 75-<100 100-<125 125-<150 150-<175 =175 Relative risk Range of LDL-C on statin treatment Boekholdt M., et al. J Am Coll Cardiol 2014;64:485-94 P for trend <0.001 |
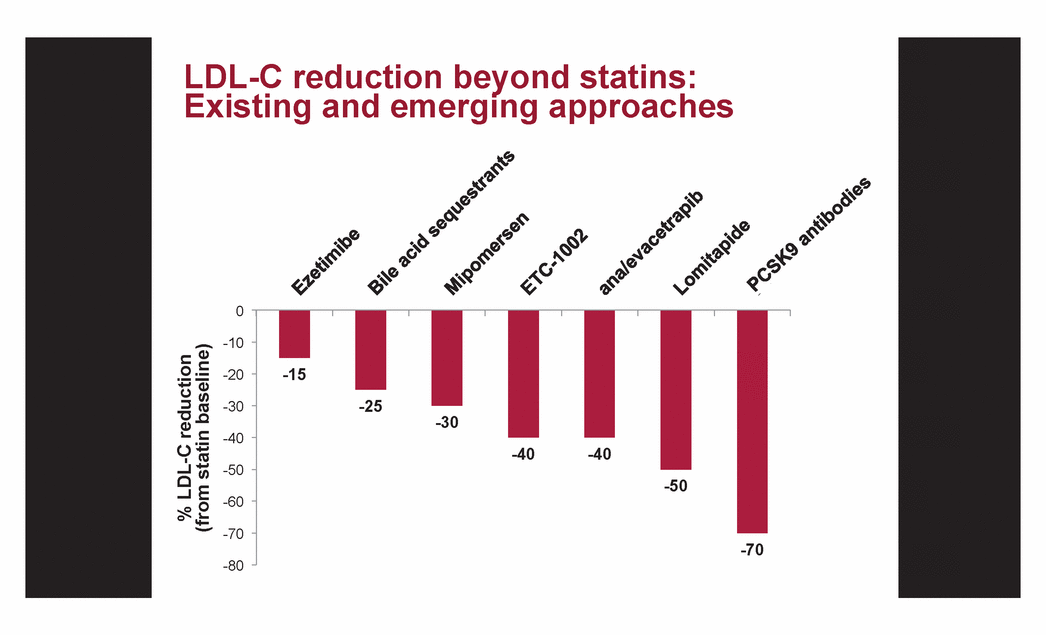
| LDL-C reduction beyond statins: Existing and emerging approaches -15 -25 -30 -40 -40 -50 -70 -80 -70 -60 -50 -40 -30 -20 -10 0 % LDL-C reduction (from statin baseline) |
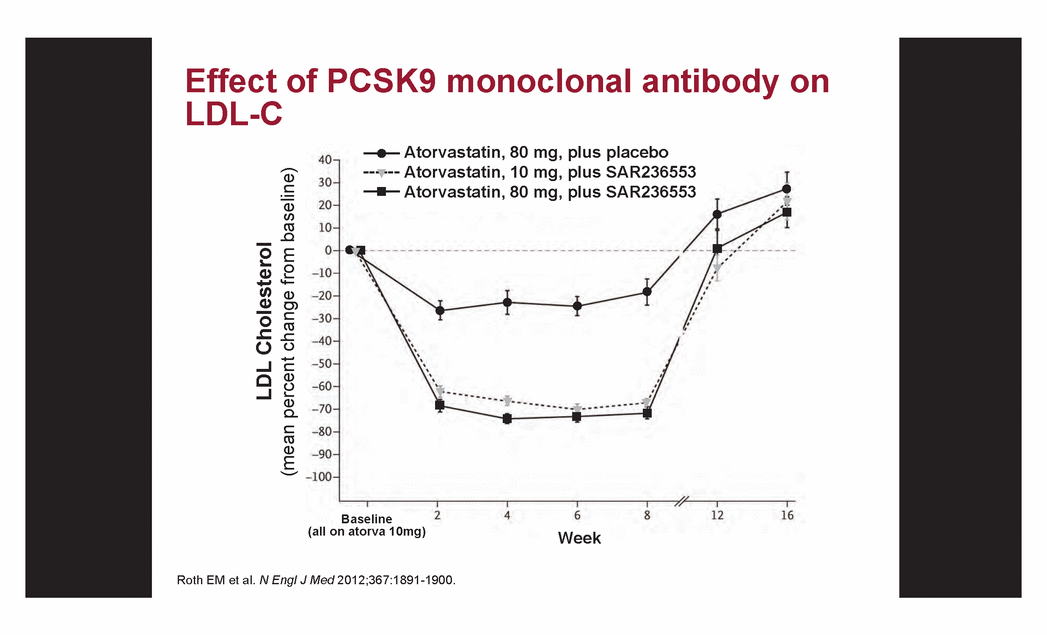
| Roth EM et al. N Engl J Med 2012;367:1891-1900. Effect of PCSK9 monoclonal antibody on LDL-C LDL Cholesterol (mean percent change from baseline) Atorvastatin, 80 mg, plus placebo Atorvastatin, 10 mg, plus SAR236553 Atorvastatin, 80 mg, plus SAR236553 Baseline (all on atorva 10mg) Week |
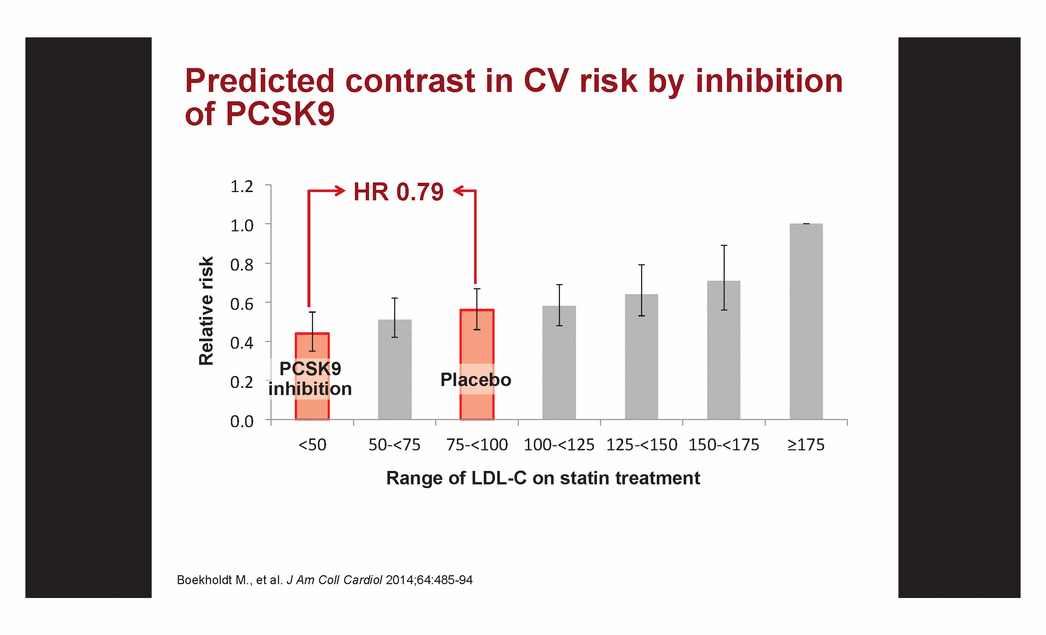
| Predicted contrast in CV risk by inhibition of PCSK9 Relative risk Range of LDL-C on statin treatment Boekholdt M., et al. J Am Coll Cardiol 2014;64:485-94 HR 0.79 Placebo PCSK9 inhibition |
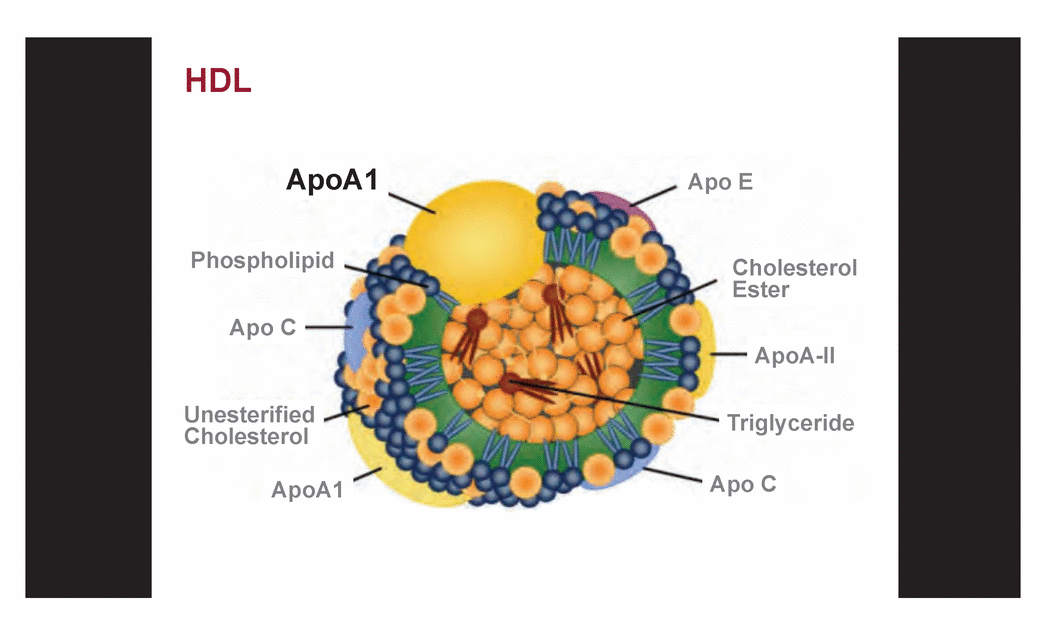
| HDL ApoA1 Apo E Cholesterol Ester ApoA-II Triglyceride Apo C ApoA1 Unesterified Cholesterol Apo C Phospholipid |
| HDL and ApoA-I as target of therapy: Key questions 1. Are plasma concentrations of HDL-C and ApoA-I related to risk in patients treated with statins? 2. Is HDL functional in patients with atherosclerosis? 3. Which approaches are promising for HDL-based treatment? |
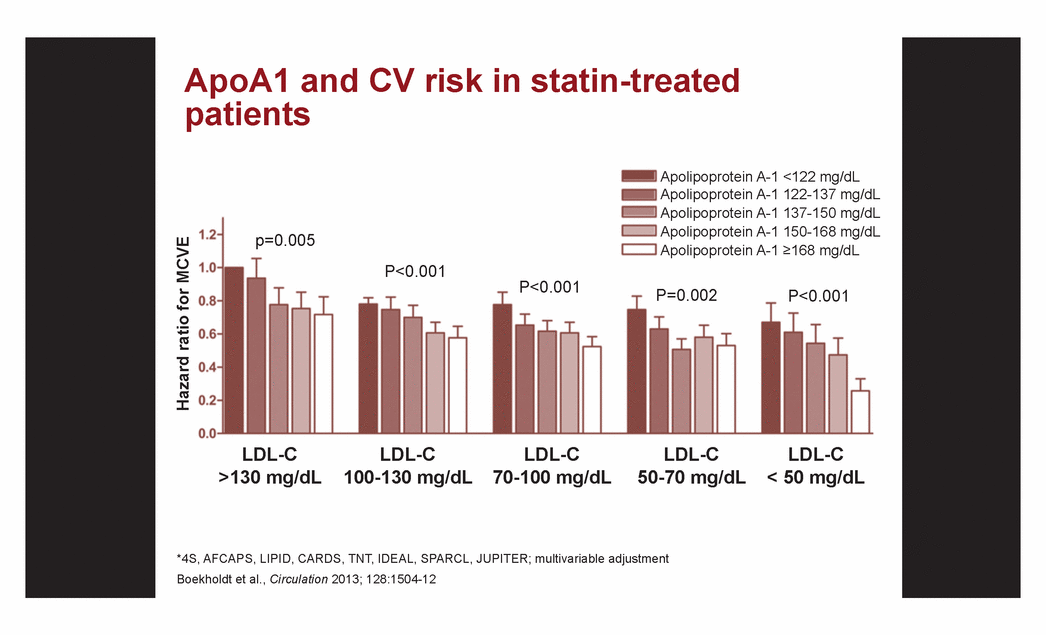
| ApoA1 and CV risk in statin-treated patients *4S, AFCAPS, LIPID, CARDS, TNT, IDEAL, SPARCL, JUPITER; multivariable adjustment Boekholdt et al., Circulation 2013; 128:1504-12 Hazard ratio for MCVE Apolipoprotein A-1 <122 mg/dL Apolipoprotein A-1 122-137 mg/dL Apolipoprotein A-1 137-150 mg/dL Apolipoprotein A-1 150-168 mg/dL Apolipoprotein A-1 =168 mg/dL p=0.005 P<0.001 P<0.001 P=0.002 P<0.001 LDL-C >130 mg/dL LDL-C 100-130 mg/dL LDL-C 70-100 mg/dL LDL-C 50-70 mg/dL LDL-C < 50 mg/dL |
| Completed CV outcome trials with agents that increase HDL-C concentration, but did not reduce CV risk • Niacin ~15% increase in HDL-C • CETP inhibitors (dalcetrapib, torcetrapib) ~30-70% increase in HDL-C |
| Why did niacin and CETPi trials fail? • Undesirable effects of the drugs: – Niacin: impaired glucose tolerance, serious GI events, infections – CETPi: increased blood pressure, C-reactive protein • HDL function was abnormal |
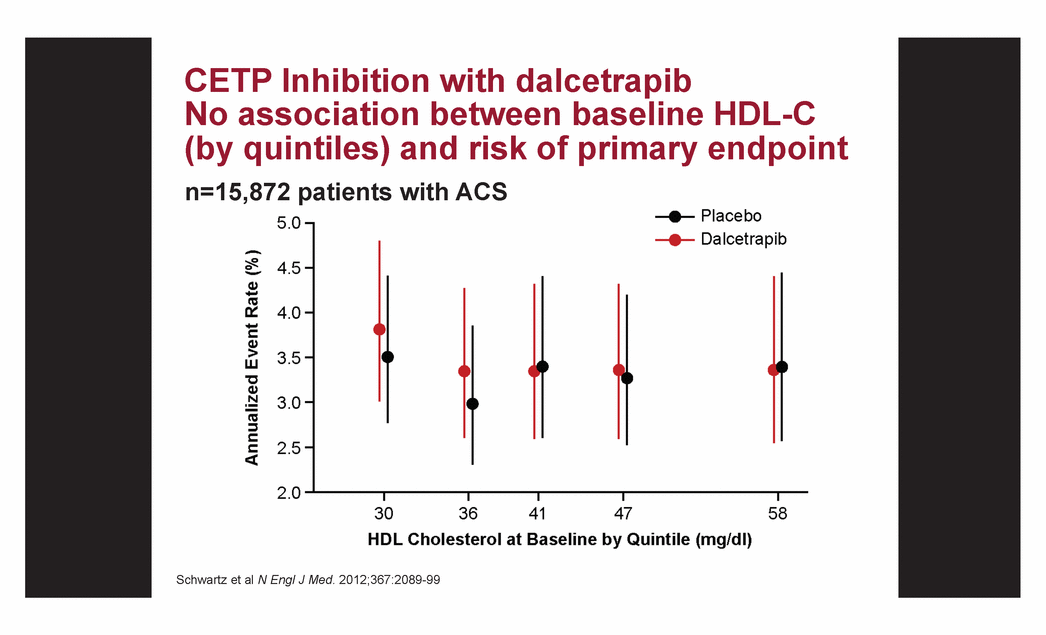
| Concentration Function CETP Inhibition with dalcetrapib No association between baseline HDL-C (by quintiles) and risk of primary endpoint n=15,872 patients with ACS Schwartz et al N Engl J Med. 2012;367:2089-99 |
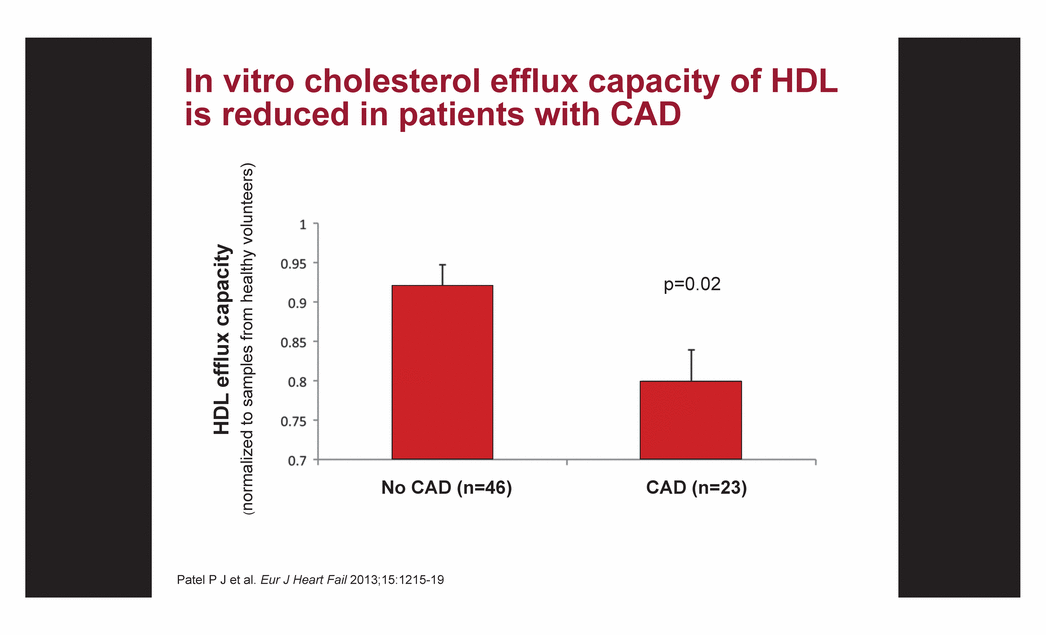
| In vitro cholesterol efflux capacity of HDL is reduced in patients with CAD Patel P J et al. Eur J Heart Fail 2013;15:1215-19 No CAD (n=46) HDL efflux capacity (normalized to samples from healthy volunteers) CAD (n=23) p=0.02 |
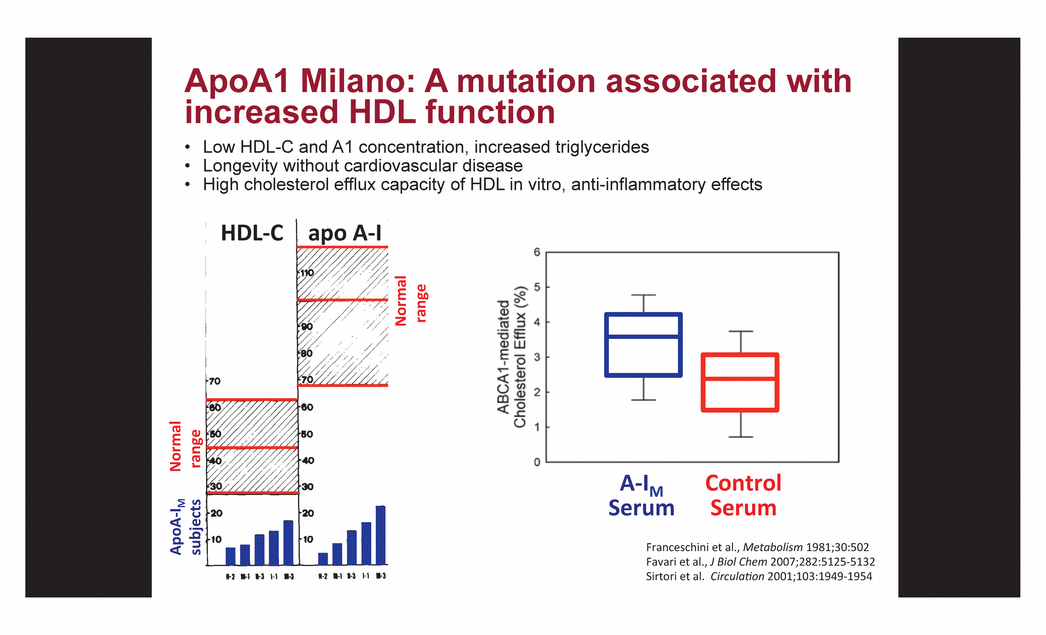
| ApoA1 Milano: A mutation associated with increased HDL function • Low HDL-C and A1 concentration, increased triglycerides • Longevity without cardiovascular disease • High cholesterol efflux capacity of HDL in vitro, anti-inflammatory effects Franceschini et al., Metabolism 1981;30:502 Favari et al., J Biol Chem 2007;282:5125-5132 Sirtori et al. Circula3on 2001;103:1949-1954 |
| Summary PCSK9 • Key regulator of LDL-C levels • Abrogating its function reduces LDL-C and may reduce CV risk HDL and ApoA-I • HDL-C concentration is easy to measure, but HDL function may be more important. • To date, 2 strategies that increase HDL-C have not reduced CV risk (niacin, CETP inhibitors). • Native HDL is a complex particle and its composition and function may change in patients with atherosclerosis • ApoA-I is the basic building block of HDL • An engineered HDL mimetic made from either wild type or mutant, hyperfunctional ApoA-I is a promising, but far from proven approach |
| Changing the management of atherosclerosis: ApoA1-Milano and PCSK9sc David Kallend The Medicines Company Changing the management of atherosclerosis: ApoA1-Milano and PCSK9sc David Kallend The Medicines Company |
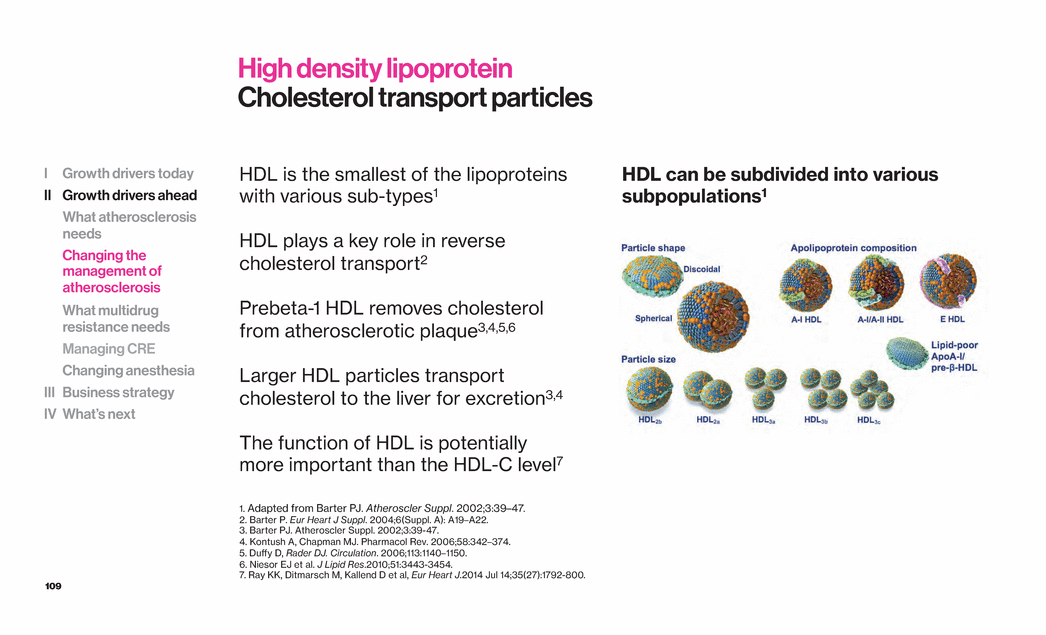
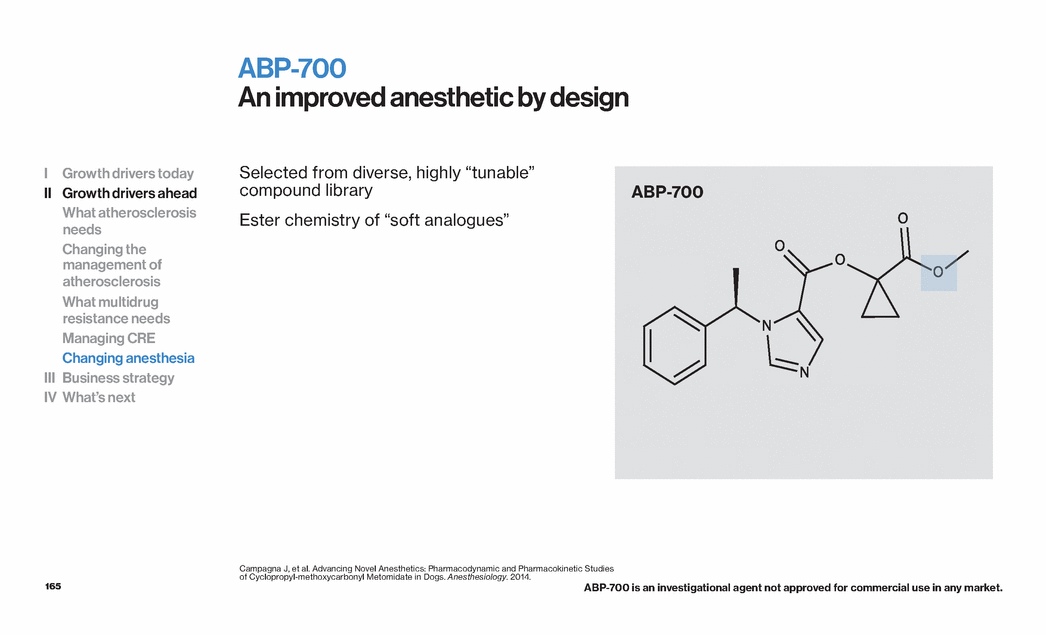
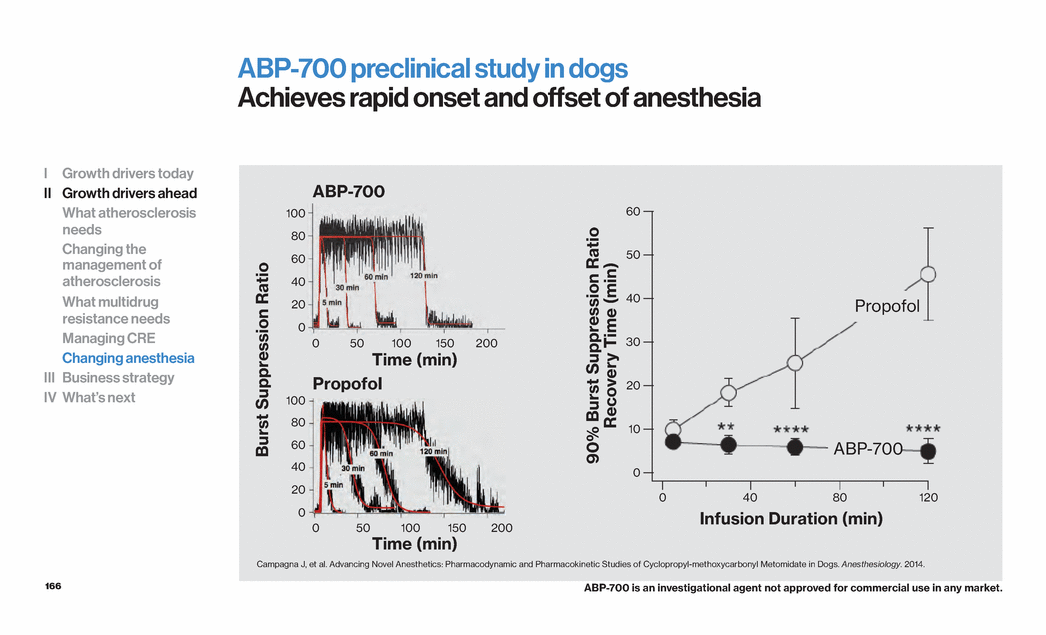
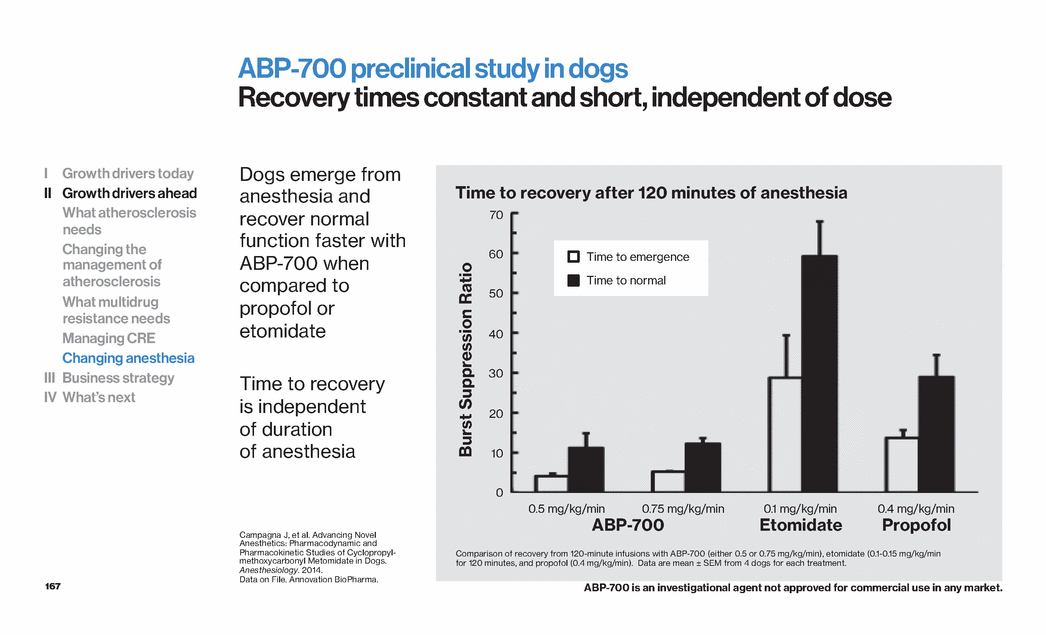
| 109 High density lipoprotein Cholesterol transport particles HDL is the smallest of the lipoproteins with various sub-types1 HDL plays a key role in reverse cholesterol transport2 Prebeta-1 HDL removes cholesterol from atherosclerotic plaque3,4,5,6 Larger HDL particles transport cholesterol to the liver for excretion3,4 The function of HDL is potentially more important than the HDL-C level7 1. Adapted from Barter PJ. Atheroscler Suppl. 2002;3:39–47. 2. Barter P. Eur Heart J Suppl. 2004;6(Suppl. A): A19–A22. 3. Barter PJ. Atheroscler Suppl. 2002;3:39-47. 4. Kontush A, Chapman MJ. Pharmacol Rev. 2006;58:342–374. 5. Duffy D, Rader DJ. Circulation. 2006;113:1140–1150. 6. Niesor EJ et al. J Lipid Res.2010;51:3443-3454. 7. Ray KK, Ditmarsch M, Kallend D et al, Eur Heart J.2014 Jul 14;35(27):1792-800. HDL can be subdivided into various subpopulations1 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 110 ApoA1 plays a key role in HDL remodeling and interacts with ABCA1 which mediates cholesterol efflux1 Loss of function leads to increased risk of CV disease1 Gain of function leads to decreased risk of CV disease and atherosclerosis2 ApoA1-Milano induces plaque regression in rabbits3 CV disease is reduced in humans with the ApoA1-Milano variant2 Apolipoprotein A1 Key component of HDL and protection from atherosclerosis 1. Dastani Z, Dangoisse C, Boucher B, Desbiens K, Krimbou L, Dufour R, Hegele RA, Pajukanta P, Engert JC, Genest J, Marcil M (March 2006). "A novel nonsense apolipoprotein A-I mutation (apoA-I(E136X)) causes low HDL cholesterol in French Canadians". Atherosclerosis 185 (1): 127–36. 2. Franceschini G, Sirtori CR, Capurso A, Weisgraber KH, Mahley RW (1980). "A-IMilano apoprotein. Decreased high density lipoprotein cholesterol levels with significant lipoprotein modifications and without clinical atherosclerosis in an Italian family". J. Clin. Invest. 66 (5): 892–900. 3. Ibanez B, Giannarelli C, Cimmino G, Santos-Gallego CG, Alique M, Pinero A, Vilahur G, Fuster V, Badimon L, Badimon JJ. Recombinant HDL(Milano) exerts greater anti-inflammatory and plaque stabilizing properties than HDL(wild-type).Atherosclerosis. 2012 Jan;220(1):72-7. HDL with either wild-type A1 or Milano induces plaque regression in rabbits3 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next 0 19 22 20 21 Plaque Volume (mm3) HDL Milano HDL wt Placebo |
| 111 ABCA1 Cholesterol efflux regulatory protein ABCA1 is the “pump” which mediates the efflux of cholesterol to lipid poor ApoA1 forming mature HDL particles1 ABCA1 plays a key role in reverse cholesterol transport2 Loss of function is seen in Tangier Disease3 which is associated with premature atherosclerosis 1. Schmitz G, Langmann T (April 2001). "Structure, function and regulation of the ABC1 gene product". Curr. Opin. Lipidol. 12 (2): 129–40. 2. Adapted from: Duffy D, Rader DJ. Circulation 2006;113:1140–1150. 3. Bodzioch M, Orsó E, Klucken J, Langmann T, Böttcher A, Diederich W, Drobnik W, Barlage S, Büchler C, Porsch-Ozcürümez M, Kaminski WE, Hahmann HW, Oette K, Rothe G, Aslanidis C, Lackner KJ, Schmitz G (August 1999). "The gene encoding ATP-binding cassette transporter 1 is mutated in Tangier disease". Nature Genetics 22 (4): 347–51. Cholesterol efflux is the first step in reverse cholesterol transport2 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
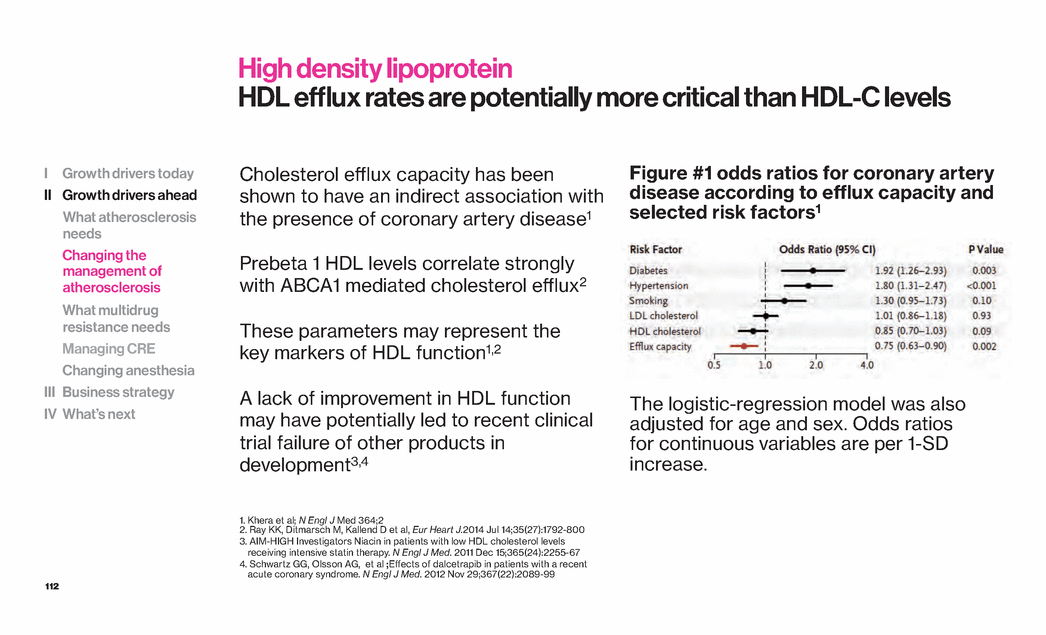
| 112 High density lipoprotein HDL efflux rates are potentially more critical than HDL-C levels Cholesterol efflux capacity has been shown to have an indirect association with the presence of coronary artery disease1 Prebeta 1 HDL levels correlate strongly with ABCA1 mediated cholesterol efflux2 These parameters may represent the key markers of HDL function1,2 A lack of improvement in HDL function may have potentially led to recent clinical trial failure of other products in development3,4 1. Khera et al; N Engl J Med 364;2 2. Ray KK, Ditmarsch M, Kallend D et al, Eur Heart J.2014 Jul 14;35(27):1792-800 3. AIM-HIGH Investigators Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011 Dec 15;365(24):2255-67 4. Schwartz GG, Olsson AG, et al ;Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med. 2012 Nov 29;367(22):2089-99 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next The logistic-regression model was also adjusted for age and sex. Odds ratios for continuous variables are per 1-SD increase. Figure #1 odds ratios for coronary artery disease according to efflux capacity and selected risk factors1 |
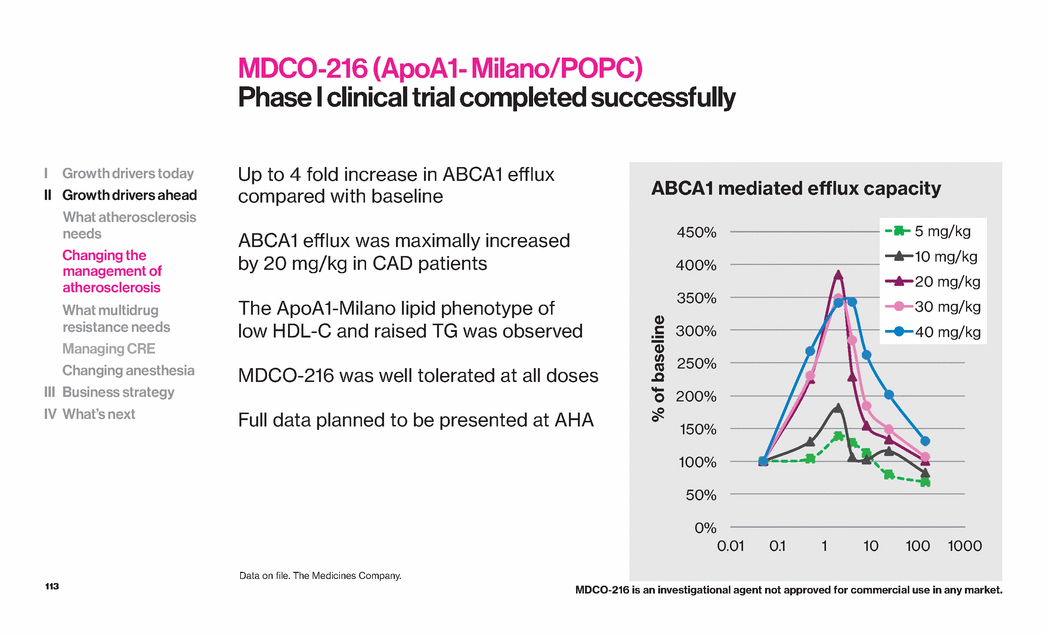
| 113 Up to 4 fold increase in ABCA1 efflux compared with baseline ABCA1 efflux was maximally increased by 20 mg/kg in CAD patients The ApoA1-Milano lipid phenotype of low HDL-C and raised TG was observed MDCO-216 was well tolerated at all doses Full data planned to be presented at AHA MDCO-216 (ApoA1- Milano/POPC) Phase I clinical trial completed successfully 0% 50% 100% 150% 200% 250% 300% 350% 400% 450% 0.01 0.1 1 10 100 1000 % of baseline 5 mg/kg 10 mg/kg 20 mg/kg 30 mg/kg 40 mg/kg ABCA1 mediated efflux capacity I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next MDCO-216 is an investigational agent not approved for commercial use in any market. Data on file. The Medicines Company. |
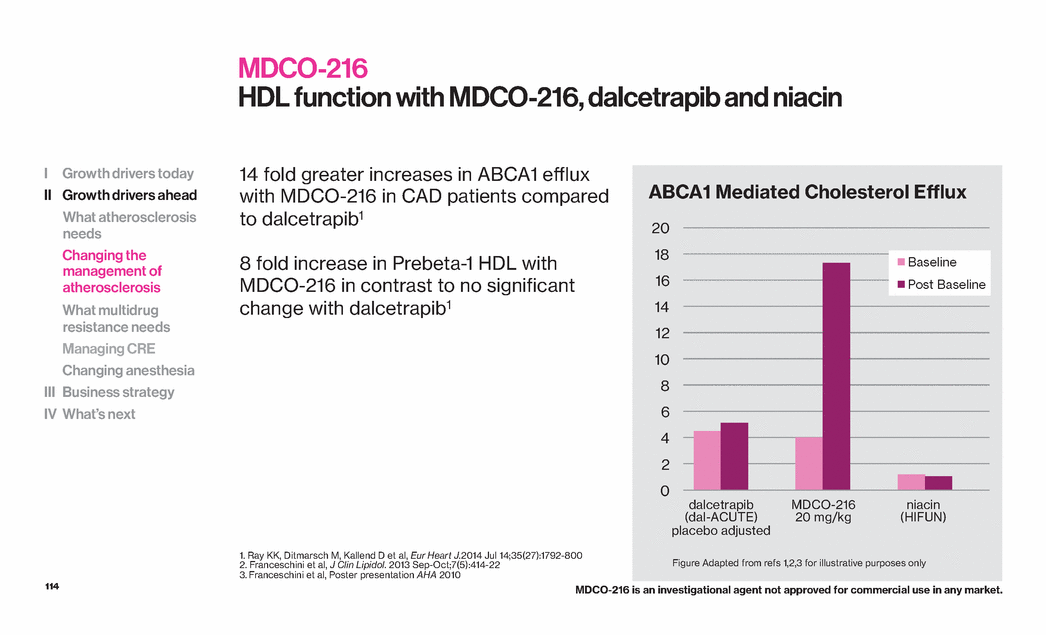
| 114 0 2 4 6 8 10 12 14 16 18 20 Baseline Post Baseline dalcetrapib (dal-ACUTE) placebo adjusted MDCO-216 20 mg/kg niacin (HIFUN) MDCO-216 HDL function with MDCO-216, dalcetrapib and niacin 14 fold greater increases in ABCA1 efflux with MDCO-216 in CAD patients compared to dalcetrapib1 8 fold increase in Prebeta-1 HDL with MDCO-216 in contrast to no significant change with dalcetrapib1 1. Ray KK, Ditmarsch M, Kallend D et al, Eur Heart J.2014 Jul 14;35(27):1792-800 2. Franceschini et al, J Clin Lipidol. 2013 Sep-Oct;7(5):414-22 3. Franceschini et al, Poster presentation AHA 2010 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Figure Adapted from refs 1,2,3 for illustrative purposes only ABCA1 Mediated Cholesterol Efflux MDCO-216 is an investigational agent not approved for commercial use in any market. |
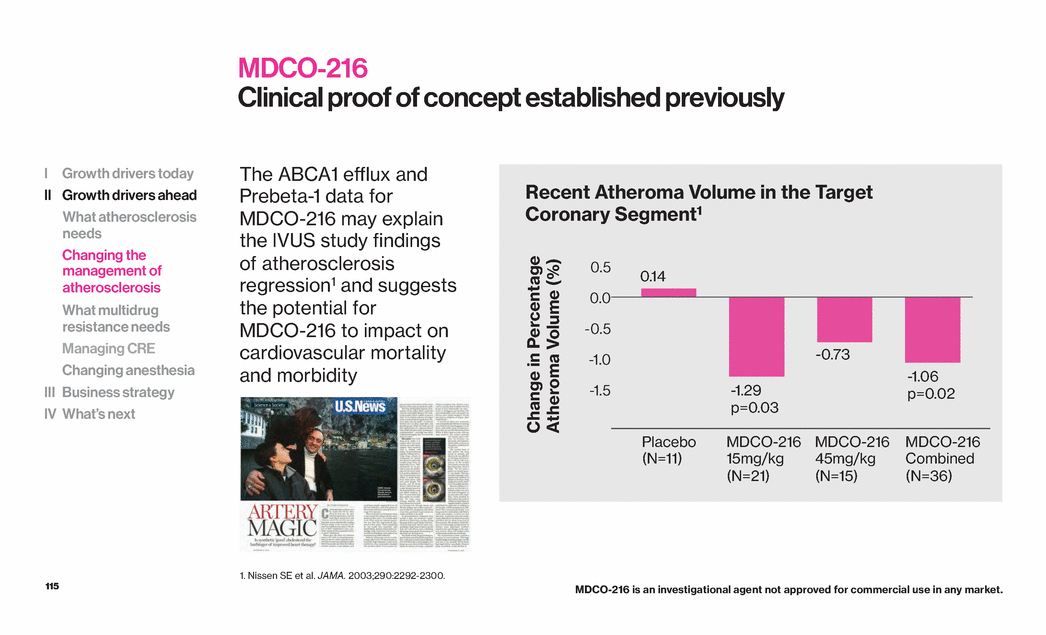
| 115 MDCO-216 Clinical proof of concept established previously I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Placebo (N=11) MDCO-216 15mg/kg (N=21) MDCO-216 45mg/kg (N=15) MDCO-216 Combined (N=36) 1. Nissen SE et al. JAMA. 2003;290:2292-2300. The ABCA1 efflux and Prebeta-1 data for MDCO-216 may explain the IVUS study findings of atherosclerosis regression1 and suggests the potential for MDCO-216 to impact on cardiovascular mortality and morbidity -1.5 -1.0 -0.5 0.0 0.5 -1.29 p=0.03 -1.06 p=0.02 Change in Percentage Atheroma Volume (%) 0.14 -0.73 Recent Atheroma Volume in the Target Coronary Segment1 MDCO-216 is an investigational agent not approved for commercial use in any market. |
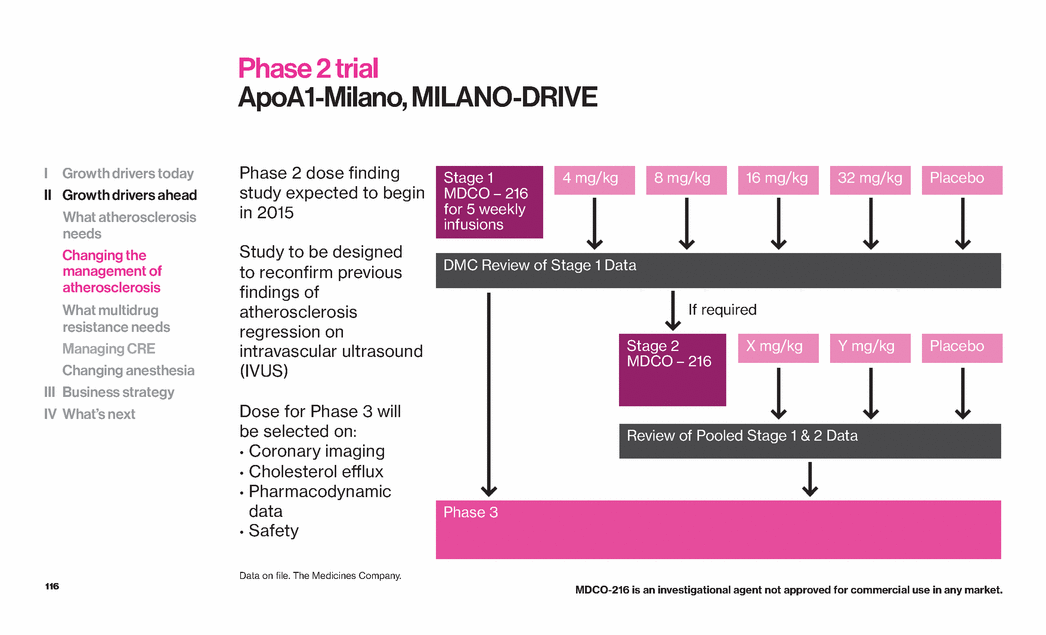
| 116 Phase 2 trial ApoA1-Milano, MILANO-DRIVE Phase 2 dose finding study expected to begin in 2015 Study to be designed to reconfirm previous findings of atherosclerosis regression on intravascular ultrasound (IVUS) Dose for Phase 3 will be selected on: • Coronary imaging • Cholesterol efflux • Pharmacodynamic data • Safety I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Stage 1 MDCO – 216 for 5 weekly infusions 4 mg/kg 8 mg/kg 16 mg/kg 32 mg/kg Placebo Stage 2 MDCO – 216 Phase 3 DMC Review of Stage 1 Data Review of Pooled Stage 1 & 2 Data X mg/kg Y mg/kg Placebo If required Data on file. The Medicines Company. MDCO-216 is an investigational agent not approved for commercial use in any market. |
| 117 Phase 2 MILANO-DRIVE study: Dose and regimen will be selected for Phase 3 Anticipated Phase 3 study • Cardiovascular mortality and morbidity study in 6,000–8,000 ACS patients • Primary endpoint major acute coronary events (CV death, non-fatal MI and stroke) Manufacturing scale-up steps Global partnership intention 12 years exclusivity as a biologic MDCO-216 Development plan I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next MDCO-216 is an investigational agent not approved for commercial use in any market. |
| ALN-PCSsc First in Class PCSK9 Synthesis Inhibitor |
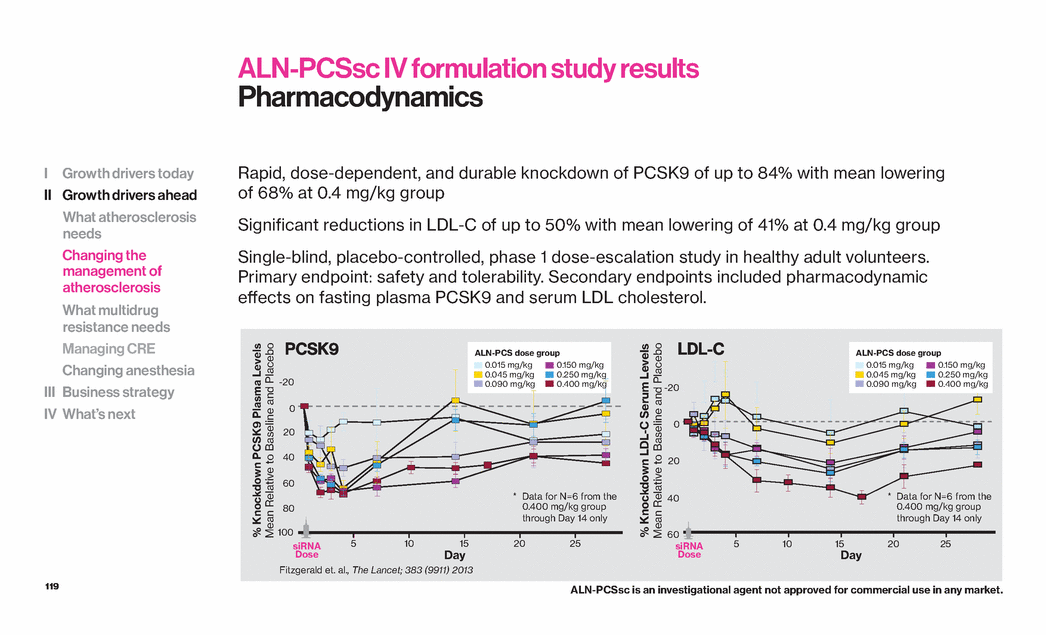
| 119 Rapid, dose-dependent, and durable knockdown of PCSK9 of up to 84% with mean lowering of 68% at 0.4 mg/kg group Significant reductions in LDL-C of up to 50% with mean lowering of 41% at 0.4 mg/kg group Single-blind, placebo-controlled, phase 1 dose-escalation study in healthy adult volunteers. Primary endpoint: safety and tolerability. Secondary endpoints included pharmacodynamic effects on fasting plasma PCSK9 and serum LDL cholesterol. ALN-PCSsc IV formulation study results Pharmacodynamics I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next 5 10 15 20 25 0 100 80 60 40 20 -20 Day ALN-PCS dose group 0.015 mg/kg 0.045 mg/kg 0.090 mg/kg 0.150 mg/kg 0.250 mg/kg 0.400 mg/kg % Knockdown PCSK9 Plasma Levels Mean Relative to Baseline and Placebo siRNA Dose Day % Knockdown LDL-C Serum Levels Mean Relative to Baseline and Placebo siRNA Dose 40 20 0 -20 60 5 10 15 20 25 PCSK9 LDL-C Fitzgerald et. al., The Lancet; 383 (9911) 2013 ALN-PCSsc is an investigational agent not approved for commercial use in any market. * Data for N=6 from the 0.400 mg/kg group through Day 14 only ALN-PCS dose group 0.015 mg/kg 0.045 mg/kg 0.090 mg/kg 0.150 mg/kg 0.250 mg/kg 0.400 mg/kg * Data for N=6 from the 0.400 mg/kg group through Day 14 only |
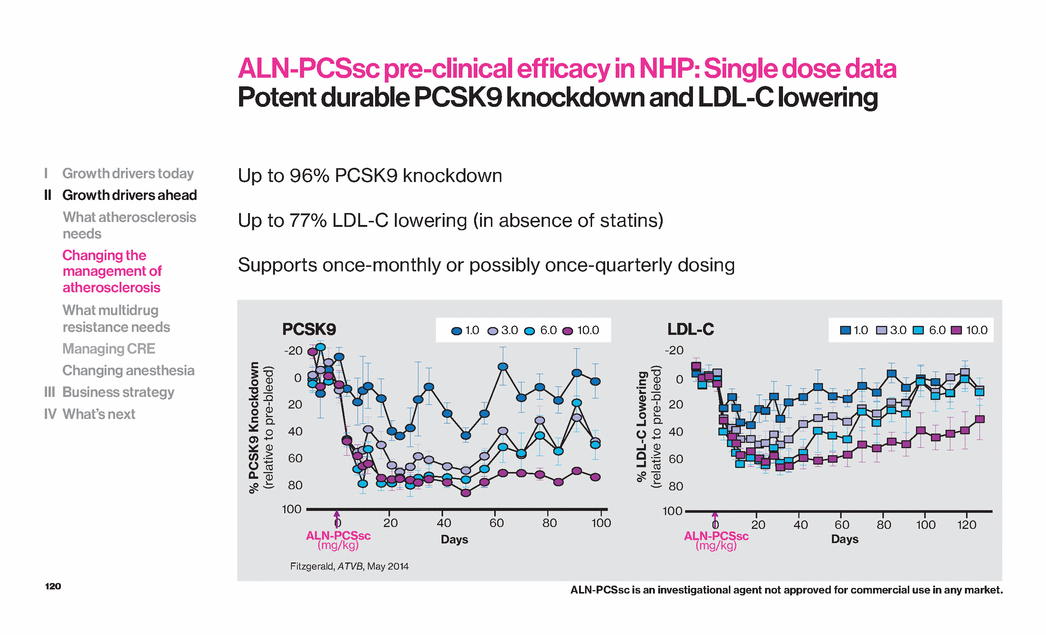
| 120 Up to 96% PCSK9 knockdown Up to 77% LDL-C lowering (in absence of statins) Supports once-monthly or possibly once-quarterly dosing ALN-PCSsc pre-clinical efficacy in NHP: Single dose data Potent durable PCSK9 knockdown and LDL-C lowering I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next PCSK9 LDL-C 1.0 3.0 6.0 10.0 % PCSK9 Knockdown (relative to pre-bleed) % LDL-C Lowering (relative to pre-bleed) Days 0 20 40 60 80 100 100 80 60 40 20 0 -20 ALN-PCSsc (mg/kg) ALN-PCSsc (mg/kg) 80 60 40 20 -20 100 Days 0 20 40 60 80 100 120 0 1.0 3.0 6.0 10.0 Fitzgerald, ATVB, May 2014 ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| 121 Clamped PCSK9 knockdown and LDL-C lowering Monthly or, possibly, quarterly SC dosing LDL-C lowering independent of baseline PCSK9 levels Low SC injection volumes <1 mL Potential synergy with statins ALN-PCSsc PCSK9 synthesis inhibitor Potential profile of PCSK9 synthesis inhibitor I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Fitzgerald, ATVB, May 2014 Fitzgerald et. al., The Lancet; 383 (9911) 2013 ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
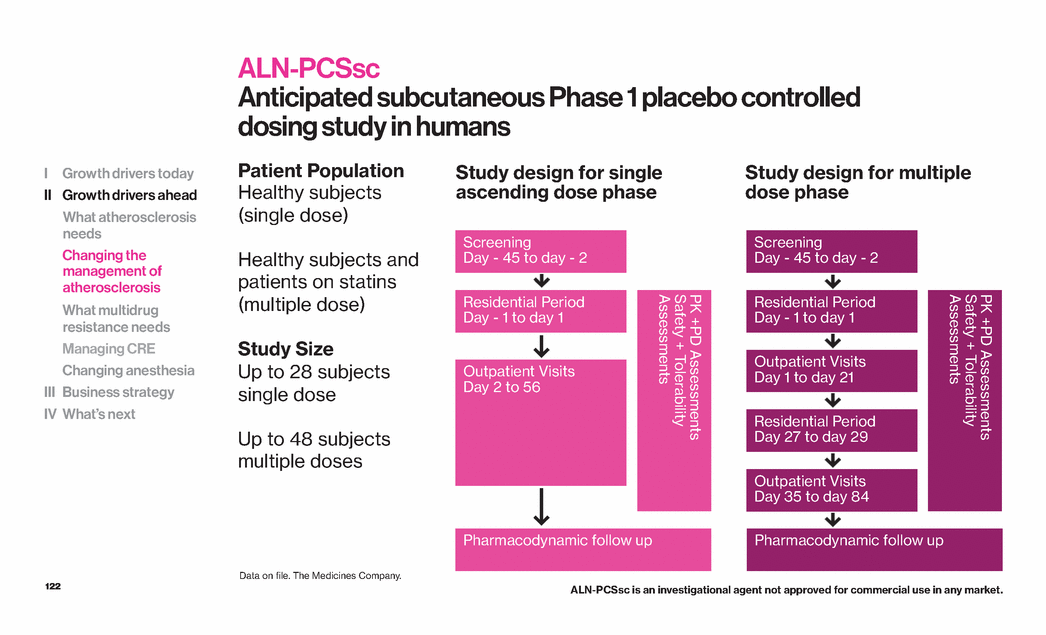
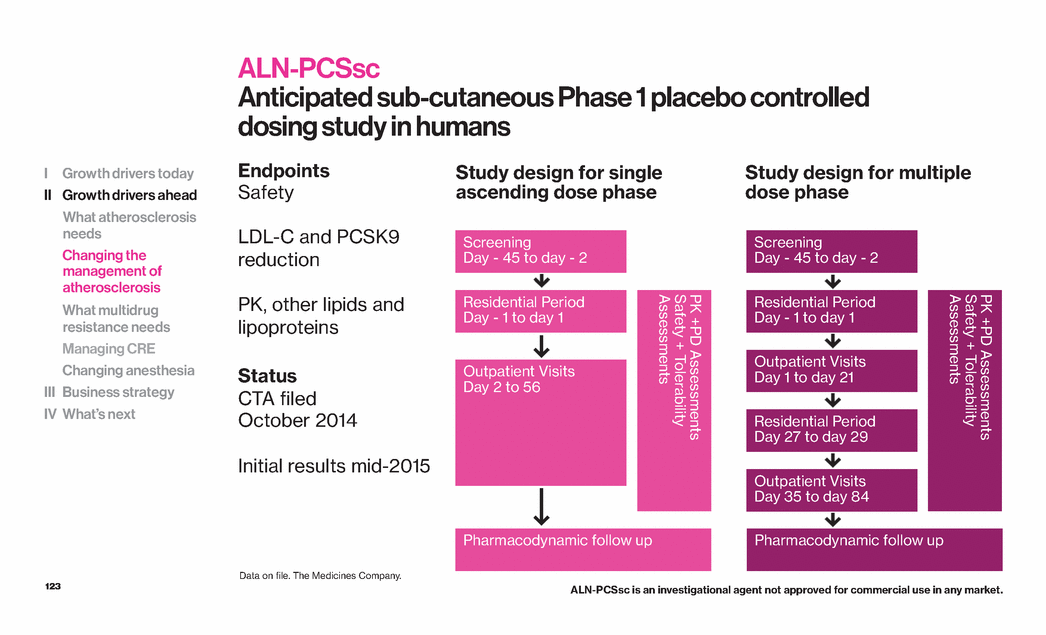
| 122 Study design for single ascending dose phase Study design for multiple dose phase Patient Population Healthy subjects (single dose) Healthy subjects and patients on statins (multiple dose) Study Size Up to 28 subjects single dose Up to 48 subjects multiple doses ALN-PCSsc Anticipated subcutaneous Phase 1 placebo controlled dosing study in humans I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Screening Day - 45 to day - 2 Residential Period Day - 1 to day 1 Outpatient Visits Day 2 to 56 PK +PD Assessments Safety + Tolerability Assessments Screening Day - 45 to day - 2 Residential Period Day - 1 to day 1 Outpatient Visits Day 1 to day 21 Residential Period Day 27 to day 29 Outpatient Visits Day 35 to day 84 PK +PD Assessments Safety + Tolerability Assessments Pharmacodynamic follow up Pharmacodynamic follow up Data on file. The Medicines Company. ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| 123 Endpoints Safety LDL-C and PCSK9 reduction PK, other lipids and lipoproteins Status CTA filed October 2014 Initial results mid-2015 ALN-PCSsc Anticipated sub-cutaneous Phase 1 placebo controlled dosing study in humans I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Study design for single ascending dose phase Study design for multiple dose phase Screening Day - 45 to day - 2 Residential Period Day - 1 to day 1 Outpatient Visits Day 2 to 56 PK +PD Assessments Safety + Tolerability Assessments Screening Day - 45 to day - 2 Residential Period Day - 1 to day 1 Outpatient Visits Day 1 to day 21 Residential Period Day 27 to day 29 Outpatient Visits Day 35 to day 84 PK +PD Assessments Safety + Tolerability Assessments Pharmacodynamic follow up Pharmacodynamic follow up Data on file. The Medicines Company. ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| 124 We believe in this fast route to filing of ALN-PCSsc Complete Phase 1 SAD / MD study expected Q2 2015 Perform multi-dose Phase 2 study to confirm dose and dosing regimen for Phase 3 Proceed to pivotal Phase 3 studies • Lipid lowering studies • Long term studies for safety • CV mortality and morbidity study in high risk patients (6,000–8,000 patients) ALN-PCSsc Current regulatory strategy I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| 125 ALN-PCSsc Partnerships Collaboration between The Medicines Company and Alnylam Pharmaceuticals, Inc. to advance ALN-PCSsc program Create industry-leading effort for best-in-class medicine for PCSK9 antagonism • $25M upfront payment • Up to $180M Milestone payments • Scaled double-digit royalties on global products sales Alnylam to complete certain pre-clinical and Phase 1 studies The Medicines Company to lead and fund development from Phase 2 to commercialization I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| 126 MDCO-216 Presentation of Phase 1 SAD data at AHA November 2014 Publication of manuscript of Phase 1 SAD study Q1 2015 (estimated) Announcement of FPI Phase 2 study Q2 2015 First results Q2 2016 Manufacturing milestones MDCO-216 and ALN-PCSsc Anticipated news flow I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ALN-PCSsc Announcement of FPI Phase 1 study late 2014/Q1 2015 First results mid-2015 ALN-PCSsc is an investigational agent not approved for commercial use in any market. |
| Backup What multidrug resistance needs Ron Jones JMI Laboratories What multidrug resistance needs Ron Jones JMI Laboratories |
| What multidrug resistance needs Ron Jones JMI Laboratories |
| Antimicrobial resistance • Resistance among Gram-negative bacilli is escalating due to mobile genes encoding potent drug inactivating enzymes (e.g., ß-lactamases) • Epidemiology is linked worldwide via patient/leisure travel and military conflicts/population migrations • Solutions are intuitive, but challenging – Susceptibility testing methods – Antimicrobial stewardship – Improved surveillance (local, national, global) – Infection control practices – New drug development • Terms describing resistance like ESBL, CTX-M, KPC, NDM, VIM and MDR have become common in our vocabulary, like that of MRSA!! |
| Beta-lactamase inhibitors (BLI) Bind in the active site of beta-lactamase Inactivate the enzyme Prevent degradation of the antibiotic Antibiotic Penicillin Cephalosporin Carbapenem e.g., TEM-1 Inactive Beta-Lactamase Inhibitor e.g., ESBL e.g., KPC-2 RPX-7009 bound in ESBL CTX-M-15 Bacterial beta-lactamases confer bacterial resistance Genes that code for enzymes that degrade this class have spread worldwide, rendering this important class inactive |
| Timeframe 2000–2006 • 602 Klebsiella isolates from 11 hospitalsa • 45% ESBL positive • 3.3% of these were KPC positive (from 7 hospitals) – Highly resistant to cephalosporins and carbapenems • KPC-2 and -3: plasmid encoded serine ß-lactamasesb – Efficiently hydrolyze carbapenems (Class A enzyme) – Resistance does not require changes in permeability – 45% identity to SME-1 carbapenemase (Also Class A) a. From Bratu S, et al. Arch Intern Med. 2005;165:1430-1435. b. From Babic M. et al. Drug Resist Updat. 2006;9:142-156. Resistant Klebsiella spp. outbreaks in New York City area: The KPC story! |
| Mortality Attributable mortality of KPC producing Klebsiella blood stream infections—48% vs. 12% for non-KPC producing Klebsiella blood stream infections Morbidity Patients with KPC-producing Klebsiella blood stream infections significantly more likely than similar patients without bacteremia to require: • ICU care (38% v 9%), • Mechanical ventilation (53% v. 25%) • Central venous catheter insertion (59% v 28%) 27% of patients with CRE identified 1st by surveillance cultures subsequently developed a blood stream infection with CRE Patel G. et al. Infect Control Hosp Epidemiol 2008; 29:1099–1106 Borer A. et al. Infect Control Hosp Epidemiol 2009; 30:972–976 Ben-David D. et al. Clin Microbiol Infect. 2012 Jan;18(1):54-60 Calfee S et al Infect Control Hosp Epidemiol 2008; 29 CRE due to KPC: Mortality and morbidity |
| Similar trends are emerging in the US and worldwide ESBLs and Carbapenemases in Enterobacteriaceae: Similar trends Carbapenem-resistant Enterobacteriaceae reported in 47 States (US CDC, February 2014) 40% 30% 20% 10% 0% 1980s 1990s 2000s 50% Incidence Beta-lactamase mediated resistance can disseminate rapidly Latin America Europe USA Endemic Interregional spread Regional spread Independent hospital outbreaks Single-hospital outbreaks Sporadic occurrence Not reported / no data KPC VIM NDM OXA-48 Other countries: Israel Luxembourg CRE in Europe (as of 2011) Rapid evolution and spread of carbapenemases among Enterobacteriaceae in Europe, 2012, Page 3; US Map, CDC CRE (http://www.cdc.gov/hai/organisms/cre/TrackingCRE.html) |
| Six year trend of ESBL and CRE in the 2007–2012 SENTRY Programa United States Organism (no. tested) Resistanceb % by Year 2007 2008 2009 2010 2011 2012 E. coli (9,743) ESBL 5.6 6.0 9.5 10.0 12.8 11.6 CRE 0.1 0.0 0.1 0.1 0.4 0.3 Klebsiella spp. (7,677) ESBL 15.4 13.2 12.7 12.5 14.6 14.1 CRE 5.5 4.8 5.0 3.3 4.4 5.2 a. 17,420 isolates (JMI Laboratories, Iowa USA) b. ESBL=extended spectrum ß-lactamases; CRE=carbapenem-resistant enteric bacillus |
| Worldwide Spread of Carbapenemases Lancet ID 2013;13:785, Page 2 |
| Summary of emerging resistances among E. coli and Klebsiella spp. (SENTRY Program, 2011) Regional variations % by area (trend)a Organism (no. tested) Resistanceb USA China Europe LATAMb E. coli (5,118) ESBL 12.8 (+) 73.8 (+) 19.4 (+) 37.1 (+) CRE 0.4 (+) 2.1 (+) 0.1 (NC) 0.0 (NC) Klebsiella spp (3,219) ESBL 14.6 (NC) 44.2 (+) 43.4 (+) 50.7 (NC) CRE 4.4 (NC) 4.3 (+) 8.7 (+) 9.7 (+) a. Five year trends (2007–2011): “+” = increasing rate; “-” = decreasing rate; and NC = no significant change b. LATAM=Latin America; ESBL=extended spectrum ß-lactamases; and CRE=carbapenem-resistant Data on file. JMI Laboratories from the SENTRY antimicrobial surveillance program. |
| Resistance is highly transmittable (plasmids) Difficult infection control challenges (e.g., NIH Clinical Center outbreak) Lack of adoption of new definitions in microbiology laboratories may cause hospitals to “miss” resistance Potential for community spread • E. coli is a common cause of community infections Challenges with currently used therapy • Tigecycline is not always reliable and poor PK-PD • Colistin can be difficult to dose, toxic and resistance rapidly appearing • New drugs are needed CDC considers CRE to be an urgent antimicrobial resistance threat Centers for Disease Control. Antibiotic Resistance Threats in the United States. 2013 (http://www.cdc.gov/drugresistance/threat-report-2013/) |
| Managing CRE: CARBAVANCE™ and beta-lactamase inhibitors Mike Dudley The Medicines Company |
| 139 Gram-negative multidrug resistance (MDR) Options for treatment of infections due to carbapenem-resistant Enterobacteriaceae (CRE) Class Tetracyclines (tigecycline) Polymyxins (colistin) Aminoglycosides Beta-lactams Daikos et. al., J Antmicrob Chemother. 2014;58:2322-8. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 140 Gram-negative MDR New beta-lactamase inhibitor combinations in clinical development Beta-lactam antibiotic Inhibitor Examples Previously Approved “New” Ceftazidime + avibactam Imipenem/cilastatin + MK7655 Meropenem + RPX-7009 (CARBAVANCE™) New “Old” Ceftolozane + tazobactam Key serine beta-lactamases associated with resistance • Class A (e.g., ESBLs, KPC); • Class C (e.g., ampC); • Class D (OXA-48; mainly Europe) Choice of partner in beta-lactam • Match pharmacology (PK, safety) • Consider activity and stability of partner beta-lactam to “older” resistance enzymes: penicillins < cephalosporins < carbapenems • Resistance to an inhibitor recently reported1 1. Livermore, ICAAC 2014. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 and the combination product CARBAVANCE™ are investigational agents not approved for commercial use in any market. |
| 141 Bloodstream isolates of carbapenemase-negative, ESBL-producing Enterobacteriaceae from US Hospitals Gram-negative MDR Why a carbapenem for a BLI combination? Data are MIC90 Meropenem Imipenem Ertapenem Ceftazidime Pip/Tazo Aztreo All strains, ESBL Phenotype (151) 0.12 0.5 0.5 >8 >64 >8 Escherichia coli (81) 0.06 0.25 0.125 >8 32 >8 Klebsiella spp (55) 0.12 0.5 0.5 >8 >64 >8 Enterobacter spp (9) 0.25 0.5 2 >8 >64 >8 Enterobacter spp (AmpC hyper) (22) 0.25 1 1 >8 >64 >8 CTX-M producing Enterobacteriaceae (76) 0.12 0.25 0.5 >8 >64 >8 CMY-producers (12) 0.06 1 0.5 > 8 >64 >8 Carbapenems are more active and stable to ESBLs than cephalosporins, monobactam, penicillin/BLI combos that have become endemic over the past two decades. Castanheira M, et al. Activity of Biapenem (RPX2003)/RPX-7009, a Carbapenem Combined with a Novel ß-lactamase Inhibitor (BLI) Tested Against Clinical Isolates Producing Defined Enzymes. ICAAC 2012 F-855. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 142 Gram-negative MDR Discovery of RPX-7009—new class of beta-lactamase inhibitors1 Series of cyclic boronic acid compounds synthesized Potent inhibitory activity against Class A and C serine beta-lactamases Optimized for: • Inhibition of serine carbapenemases • Pharmacology with carbapenems In Vitro Inhibition of Nitrocefin Degradation (Ki, µM) Enzyme Class RPX-7009 Clavulanic Acid Tazobactam KPC-2 A 0.071 41.2 1.6 CTX-M-14 A 0.039 0.027 0.001 SHV-12 A 0.006 <0.039 0.0004 TEM-10 A 0.220 0.020 0.005 P99 C 0.140 1106 1.10 CMY-2 C 0.099 845 0.71 RPX-7009 1. Hecker et. al., ICAAC 2012. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 is an investigational agent not approved for commercial use in any market. |
| 143 Gram-negative MDR In vitro activity of meropenem alone or with RPX-7009 against Enterobacteriaceae1 Organism (no. tested) Antimicrobial agent* MIC 50% 90% Enterobacteriaceae (n=200) Meropenem 0.12 32 Mer+RPX-7009 8 (µg/mL) 0.03 0.5 K. pneumoniae (n=76) Meropenem 16 >32 Mer+RPX-7009 8 (µg/mL) 0.03 1 E. coli (n=38) Meropenem <0.03 4 Mer+RPX-7009 8 (µg/mL) 0.03 0.03 Enterobacter cloacae (n=25) Meropenem 4 16 Mer+RPX-7009 8 (µg/mL) 0.06 0.25 *MER/RPX=meropenem/RPX-7009; 4=at fixed 4 µg/mL; 8=at fixed 4 µg/mL. In vitro data do not necessarily correlate to clinical efficacy. 1. Castanheira et. al., ID Week 2014. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 and the combination product CARBAVANCE™ are investigational agents not approved for commercial use in any market. |
| 144 0% 20% 40% 60% 80% 100% <=0.06 0.125 0.25 0.5 1 2 4 8 16 32 Cumulative % of strains inhibited MIC of beta-lactam antibiotic Gram-negative MDR In vitro comparison of CARBAVANCE™ (Meropenem + RPX-7009) vs. cephalosporin-based combinations N=235 MIC50 MIC90 Meropenem Alone + RPX-7009 (4 µg/mL) 0.125 <0.06 32 1 Ceftazidime Alone + Avibactam (4 µg/mL) 64 0.5 >64 4 Ceftolozane + Tazobactam (4 µg/mL) 8 2 >32 >32 MDR Strains of Enterobacteriaceae (N=235) Levofloxacin Amikacin Gentamicin Tigecycline Polymyxin B Aztreonam Cefepime Ertapenem Pip/Tazo MIC90 64 >32 >64 4 2 >128 32 32 >64 Data on file. The Medicines Company. In vitro data do not necessarily correlate to clinical efficacy. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 and the combination product CARBAVANCE™ are investigational agents not approved for commercial use in any market. Ceftolozane + Tazobactam (4 µg/mL) Ceftazidime + Avibactam (4 µg/mL) CARBAVANCE™ Meropenem + RPX-7009 (4 µg/mL) |
| 145 0.1 1.0 10.0 100.0 0 2 4 6 8 10 12 Meropenem 2g Alone Meropenem 2g + RPX7009 2000 mg Time (h) Meropenem concentration (mg/L) 0.1 1.0 10.0 100.0 0 2 4 6 8 10 12 RPX-7009 concentration (mg/L) Time (h) RPX7009 2000 mg Alone RPX7009 2000 mg + Meropenem 2g Gram-negative MDR Pharmacokinetics of CARBAVANCE™ in normal human subjects RPX-7009 pharmacokinetics Meropenem pharmacokinetics Near-ideal match of pharmacokinetics for meropenem and RPX-7009 with no apparent interactions. Griffith et. al. ICAAC 2014 and ID Week 2014 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 and the combination product CARBAVANCE™ are investigational agents not approved for commercial use in any market. |
| 146 Gram-negative MDR Pharmacodynamics of simulated human dosage regimens of meropenem + RPX-7009 against KPC-producing CRE in an in vitro hollow fiber model Strain Beta-Lactamases Meropenem MIC (mg/L) Change in Log CFU over 24 hours Alone + RPX-7009 8ug/mL E. coli EC1007 KPC-3 16 =0.06 >4 E. cloacae ECL1058 KPC-3, SHV-11, TEM-1 32 <0.06 >4 E. cloacae ECL1061 KPC-3, Hyper AmpC Expression 32 <0.06 >4 K. pneumoniae KP1061 KPC-3, SHV-11, TEM-1 16 =0.06 >4 K. pneumoniae KP1087 KPC-2, CTX-M-15, SHV-11, TEM-1 32 0.125 >4 K. pneumoniae KP1074 KPC-3, SHV-11, TEM 64 <0.5 >4 K. pneumoniae KP1099 KPC-2 128 2 >4 K. pneumoniae KP1100 KPC-3 <256 4 >4 K. pneumoniae KP1093 KPC-3, SHV-11, TEM 128 1 >4 K. pneumoniae KP1094 KPC-2, TEM-1, LEN-17 512 8 >4 In vitro data do not necessarily correlate to clinical efficacy. Tarazi et al., F-959; Griffith, F-960 ICAAC 2014. High inocula of CRE are killed by the combination, with no resistance. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next RPX-7009 and the combination product CARBAVANCE™ are investigational agents not approved for commercial use in any market. |
| 147 Gram-negative MDR CARBAVANCE™ pivotal clinical trial program design TANGO I TANGO II Patients Complicated UTI and AP CRE suspected cUTI, HABP, VABP and/or bacteremia Design Randomized 1:1 Double blind Randomized 2:1 Open label Comparator Piperacillin/Tazobactam Best available therapy Total patients 850 150 Study sites ~140 ~60 FDA/EMA guidance Yes Yes BARDA contract Yes Yes Both studies supported in part under contract with the Biomedical Advanced Research and Development Authority (BARDA). I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next CARBAVANCE™ is an investigational agent not approved for commercial use in any market. |
| 148 Gram-negative MDR What’s next? CRE is in other settings – potential implications for MDCO’s oral BLI combination Initial wave of CRE cases mostly in larger metropolitan hospitals Long-term acute care hospitals in Chicago1 • Median duration of colonization 16 months • High risk of transmission Community-Associated (CA) CRE2 • Compared CA CRE vs. HCA CRE • CA cases younger, less ill, with fewer co-morbidities • All CA-CRE cases were cUTIs 1. Haverkate et. al., ID Week 2014. 2. CDC Emerging Infection Program, ID Week 2014. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 149 Gram-negative MDR Broad-spectrum carbapenemase inhibitors (BCI) restore the potency of beta-lactam antibiotics versus major carbapenems, including NDM-1 45 KPC; 7 OXA-48 type; 36 VIM; 28 NDM-1; 18 IMP. All inhibitors tested at a fixed 4 µg/mL. 367 MDR isolates enriched for carbapenemase production Antibiotic MIC90 Alone MIC90 Plus Inhibitor MDCO BCI Combinations Meropenem 64 + BCI: 1 Oral antibiotic 64 + BCI: 2 Comparators Ceftazidime >64 + AVI: 64 Imipenem 64 + MK7655: 32 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Data on file. The Medicines Company. |
| 150 Gram-negative MDR News flow TANGO Phase 3 protocols reviewed with FDA and EMA 1Q 2014 BARDA contract awarded 2Q 2014 Phase 3 trial set up 3Q 2014 TANGO first patient enrolled 4Q 2014 BARDA award of next option periods in CARBAVANCE™ contract 4Q 2015 Anticipate clinical candidate for next generation IV/oral BLI Combo 4Q 2015 Anticipate data from TANGO clinical trials 2H 2016 I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next CARBAVANCE™ is an investigational agent not approved for commercial use in any market. |
| Changing anesthesia: ABP-700 Jason Campanga The Medicines Company |
| 152 The beginning of the era of anesthesia. Innovations in anesthesia 1846 1846 Massachusetts General Hospital surgeon John Collins Warren removed a tumor from the neck of Edward Abbott under ether anesthesia. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 153 The first barbiturates. Diethyl-barbituric acid, also known as barbital, malonal, or gardenal, was first introduced clinically as a hypnotic by the German companies Merck and Bayer and Co in 1904. Innovations in anesthesia 1904 1904 López-Muñoz et al 2005. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 154 The first intravenous anesthesia. Sodium thiopental (Pentothal™) first used in human beings on March 8, 1934, by Dr. Ralph M. Waters in Wisconsin. The drug subsequently revolutionized anesthesia by providing an intravenous route to induce general anesthesia. Sodium thiopental is still a core medicine in the World Health Organization's "Essential Drugs List.” Innovations in anesthesia 1930s López-Muñoz et al 2005. 1930s I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 155 The great age of inhaled anesthetic development. Ether-derived Methoxyflurane (60s) Desflurane (70s)* Enflurane (60s) Isoflurane (70s) Sevoflurane (70s)** Innovations in anesthesia 1940s – 1970s 1940s–1970s *Desflurane marketed in 1993 . **Sevoflurane marketed in 1995. Chloroform-derived Halothane (1950s) I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 156 Intensive search for barbiturate replacements. 1971–73 althesin 1972 etomidate 1973 propofol original formulation Innovations in anesthesia 1960s – 1970s 1960s–1970s I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 157 Innovations in anesthesia 1980s–1990s 1980s–1990s I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Pace of innovation accelerates 1983–1986 etomidate and propofol 1983–1987 development and introduction of the Laryngeal Mask Airway™ 1991–1995 fast track anesthesia 1996–1999 depth of anesthesia monitoring |
| 158 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% 1981 1982 1983 1984 1985 1986 1987 1988 1989 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 Surgeries by Setting, 1981–2006 Inpatient Surgeries Outpatient Surgeries Laryngeal Mask Airway (LMA) Etomidate Propofol Cullen, K. et al. Ambulatory Surgery in the United States, 2006. National Health Statistics Reports. January 28, 2009 – Revised September 4, 2009. Number 11. Courtesy of Centers for Disease Control and Prevention. Data from Centers for Disease Control 1996 and 2006. The volume of outpatient surgery has grown at a rapid rate, enabled by technological advances and clinical guidelines. Total surgery volume increased by average of 3.8% annually between 1981 and 2006 Modern era of anesthesia Shorter stays, ambulatory surgery I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 159 It’s more complicated than “awake” or “asleep” Measuring “sedation” and “anesthesia” CONTINUUM AND LEVELS OF SEDATION Minimum sedation/ anxiolysis Moderate/ sedation analgesia (previously conscious sedation) “MAC” Deep sedation/ analgesia General anesthesia I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 160 Anesthesia provider professional societies (American Society of Anesthesiologists, American Association of Nurse Anesthetists) Government hospital accreditation bodies (Center for Medicare Services, Joint Commission) Regulatory Agencies responsible for drug approval and safety (FDA) Published Literature on unique data sets (Anesthesia Closed Claims Project) Widespread use of propofol exposed the risks Dangers became clear I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 161 FDA rejects petition to remove warning from label Excerpt from FDA letter U.S. Food and Drug Administration, Center for Drug Evaluation and Research, Janet Woodcock, M.D. to Richard M. Cooper, Esq, Docket No. FDA-2005-P-0059, August 11, 2010. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 162 FDA rejects petition to remove warning from label Excerpt from FDA letter U.S. Food and Drug Administration, Center for Drug Evaluation and Research, Janet Woodcock, M.D. to Richard M. Cooper, Esq, Docket No. FDA-2005-P-0059, August 11, 2010. I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 163 Anesthesia-related deaths have decreased over the last 25 years from two per 10,000 anesthetics administered to one per 200,000 to 300,000.1 The American Society of Anesthesiologists says a person is more likely to be struck by lightning than die from anesthesia-related complications.2 This is likely the result of highly trained practitioners operating in highly resourced environments. Yet overall safety of anesthesia is high Reflects western style resources I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next 1 Committee on Quality of Healthcare in America, IoM: To err is human, building a safer health system. Edited by Kohn L., Corrigan J, Donaldson M, Washington Academy National Press, 1999, p 241. 2 “Medical Aspects of Lightning.” National Weather Service, National Oceanic and Atmospheric Administration, National Weather Service. |
| 164 Addressing the global surgery crisis Taking a more universal view MORE RESOURCES vs. BETTER TOOLS I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| 165 ABP-700 An improved anesthetic by design ABP-700 Selected from diverse, highly “tunable” compound library Ester chemistry of “soft analogues” I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Campagna J, et al. Advancing Novel Anesthetics: Pharmacodynamic and Pharmacokinetic Studies of Cyclopropyl-methoxycarbonyl Metomidate in Dogs. Anesthesiology. 2014. ABP-700 is an investigational agent not approved for commercial use in any market. |
| 166 ABP-700 preclinical study in dogs Achieves rapid onset and offset of anesthesia Campagna J, et al. Advancing Novel Anesthetics: Pharmacodynamic and Pharmacokinetic Studies of Cyclopropyl-methoxycarbonyl Metomidate in Dogs. Anesthesiology. 2014. Burst Suppression Ratio Time (min) 0 50 100 150 200 Propofol I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ABP-700 0 50 100 150 200 Propofol ABP-700 60 50 40 30 20 10 0 0 40 80 120 Infusion Duration (min) 90% Burst Suppression Ratio Recovery Time (min) 100 80 60 40 20 0 100 80 60 40 20 0 Time (min) ABP-700 is an investigational agent not approved for commercial use in any market. |
| 167 ABP-700 preclinical study in dogs Recovery times constant and short, independent of dose Dogs emerge from anesthesia and recover normal function faster with ABP-700 when compared to propofol or etomidate Time to recovery is independent of duration of anesthesia Comparison of recovery from 120-minute infusions with ABP-700 (either 0.5 or 0.75 mg/kg/min), etomidate (0.1-0.15 mg/kg/min for 120 minutes, and propofol (0.4 mg/kg/min). Data are mean ± SEM from 4 dogs for each treatment. Time to recovery after 120 minutes of anesthesia I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next Burst Suppression Ratio ABP-700 0.5 mg/kg/min 70 60 50 40 30 20 10 0 0.75 mg/kg/min 0.1 mg/kg/min Etomidate 0.4 mg/kg/min Propofol Time to emergence Time to normal ABP-700 is an investigational agent not approved for commercial use in any market. Campagna J, et al. Advancing Novel Anesthetics: Pharmacodynamic and Pharmacokinetic Studies of Cyclopropyl-methoxycarbonyl Metomidate in Dogs. Anesthesiology. 2014. Data on File. Annovation BioPharma. |
| 168 Preliminary results Human experience mirrors animal data including dog data shown here ABP-700 is a rapidly acting IV anesthetic Appears to have a highly titratable PD effect Safety profile is consistent with pre-clinical animal data After bolus injection, rapidly reversible anesthetic effect within 1–2 minutes ABP-700: Changing anesthesia Phase 1 program underway I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ABP-700 is an investigational agent not approved for commercial use in any market. |
| 169 Upcoming milestones Expect Phase 1 program completion by end of 2014 Press release on Phase 1 program in January at JPMorgan Healthcare Conference, and final results at Euroanaesthesia meeting in May 2015 Phase 2 program expected to begin by Q2 2015 Planning for late 2016–2017 NDA/MAA submission ABP-700: Changing anesthesia Phase 1 program underway I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next ABP-700 is an investigational agent not approved for commercial use in any market. |
| 170 Equity holder via series A June 2012 (along with Atlas Ventures and Partners Innovation Fund). Pre-negotiated option based on acceptable Proof of Concept study. Option is to acquire all assets and intellectual property for approximately $30M plus milestones and royalties. Innovative deal structure Atlas Ventures lead investor for financing I Growth drivers today II Growth drivers ahead What atherosclerosis needs Changing the management of atherosclerosis What multidrug resistance needs Managing CRE Changing anesthesia III Business strategy IV What’s next |
| Unmet needs for global atherosclerosis epidemic Relentless demand for solutions to increasing multidrug resistance 284 million surgeons worldwide demand safer, more efficient anesthesia First-in-class solutions for large-scale global healthcare problems Questions, answers and summary Scott Johnson |
| 172 Unmet needs for global atherosclerosis epidemic Relentless demand for solutions to increasing multidrug resistance Surgeons worldwide demand safer, more efficient anesthesia Potential first-in-class solutions for large-scale global healthcare problems Growth drivers ahead Summary I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| Business strategy Glenn Sblendorio The Medicines Company |
| 174 Strategy for growth Innovation: Strategic and operating leverage Situation Strategy Actions 1 Customers demand value Lead with purpose Build value-based solutions 2 Near-term growth demand Launch new products strongly Emphasize US market 3 ANGIOMAX® IP complexity Steadfast commitment Manage litigation, lifecycle and partnerships 4 Global opportunities Build development-stage potentially major products Achieve proof of concept Seek global partners 5 Potential need for capital if portfolio continues successfully Diversify sources Drive products to success Pursue partnership opportunities and additional financing, as needed I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 175 Lead with purpose Action 1: Build value-based solutions ANGIOMAX® ORBACTIV™ IV anti-thrombin for PCI (incl. ACS in EU) 1 Decreased bleeding, non-inferior ischemic benefit vs. Heparin +/- GPI2 Linear PK/PD allows early sheath pull3 Reduced cost of complications, LOS Hospital decrease in cost $791 per ACS PCI patient4 IV antibiotic for ABSSSI1 Non-inferiority to vancomycin2 in labeled indication Single dose 1200 mg over 3 hours1 Single dose reduces days IV Rx Potential hospital cost decrease $1,300 per ABSSSI patient3 1. Angiomax Prescribing Information 2. Stone GW. NEJM. 2008;358:2218-30; Stone GW. NEJM. 2006;355:2203-16; Lincoff AM. JAMA. 2003;289:853-63. 3. Data on File. The Medicines Company. 4. Premier hospital database 2012. 1. ORBACTIV Prescribing Information 2. Single dose oritavancin in the treatment of acute bacterial skin infections. N Engl J Med 2014;370:2180-90 3. MDCO analysis for US hospital Economic impact model for Oritavancin; ISPOR 17th European Congress PSS10. I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 176 US EU China Japan ROW Relative Market Opportunity Regulatory Market Access Market Challenges Growth Targeted Key Accounts (approx.) MDCO 1500 250 0 0 0 Potential Partners 0 1250 1200 450 Selective Strategy Full Attention Focused Attention Targeted Partnering Targeted Partnering Opportunistic Strategy: Launch new products strongly Action 2: Focus on the US market: Leverage partnerships globally I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next 1. MDCO Analysis. |
| 177 Strategy: Steadfast commitment to ANGIOMAX® Action 3: Manage litigation, lifecycle and partnerships I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next Lifecycle ANGIOMAX® partnerships Continued defense of ANGIOMAX® patents Valuable supply contracts Teva Settled Lonza and Teva for active pharmaceutical ingredient Patheon and Fresenius Kabi (APP) for drug product APP Settled Hospira On Appeal by MDCO Mylan On Appeal by Mylan Others Pending Continued investment in the brand New technologies and presentations Clinical studies BiogenIdec and HRI Highly successful partnerships End of US royalty payments 12/15/14 |
| 178 We are actively seeking regional partners for our registration-stage/launch products and anticipate dedicated efforts to build global partnerships in the future for our development-stage products Strategy: Build development-stage potentially major products Action 4: Achieve clinical POC and seek global partners I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next Potential deal structures Licenses incl. upfront payments, milestones, royalties/ transfer price Potential deal structures US co-promote, ex-US partner incl. upfront payments and royalties Registration -stage/launch products Development -stage products Proof of concept Global Regional Europe China, Japan Latin America |
| 179 We have built our company based on an in-licensing and acquisition strategy which is disciplined, focused and milestone-based Milestones are tied to successful outcomes On aggregate, across current development and launch products we have paid <30% of total terms as upfront payments Potential need for capital if portfolio continues successfully Action 5: Drive products to success, pursue partnership opportunities and additional financing, as needed I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next Maximum possible payments for current portfolio of deals going forward1 Asset Acquisition Development Regulatory approval Commercial Total2 ~$120M ~$209M ~$472M ~$980M ~$1,781M 7% 12% 26% 55% 100% 1. Targanta (ORBACTIV™), Incline (IONSYS™), Profibrix (RAPLIXA™), Rempex (MINOCIN® IV, CARBAVANCE™, BLI inhibitors), Tenaxis (PREVELEAK™), Alnylam (ALN-PCSsc), Pfizer (MDCO-216), AstraZeneca (cangrelor), Bristol Myers Squibb (RECOTHROM®), Annovation Biopharma (ABP-700). 2. Excludes royalty payments. |
| 180 If all of our programs are successful and if we achieve success in development, regulatory and/or commercial launch milestones and consummate two acquisitions during 2015, then we anticipate paying up to $415M Potential need for capital if portfolio continues successfully Action 5: Drive products to success, pursue partnership opportunities and additional financing, as needed 2015 anticipated event Product candidate(s) Five development milestones MDCO-216, ALN-PCSsc, CARBAVANCE™, and IND filings for two novel BLI compounds Five regulatory approvals in US and/or Europe Cangrelor, IONSYS™, RAPLIXA™, MINOCIN® IV, and PREVELEAK™ One commercial launch IONSYS™ Two asset acquisitions RECOTHROM® ($80–85M) and Annovation (up to $35.3M) I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| 181 Potential need for capital if portfolio continues successfully Action 5: Drive products to success, pursue partnership opportunities and additional financing, as needed Revenue Target Ranges1 ($M) $25-$199 $200-$499 $500-$999 $1,000+ Total Aggregate annual revenue2 $905 $3,110 $2,950 $2,500 $9,465 Aggregate commercial milestone payments $2491 $317 $164 $250 $980 1. Commercial milestones are based on a specified revenue target, per product. 2. Aggregate annual revenue associated with the achievement of revenue targets for all products that give rise to a commercial milestone. 3. Includes regional product launch milestones I Growth drivers today II Growth drivers ahead III Business strategy IV What’s next |
| Questions and answers Glenn Sblendorio |
| What’s next Clive Meanwell The Medicines Company |
| THANK YOU |