
OraSure 4Q21 Earnings February 23, 2022 EXHIBIT 99.2

Forward-Looking Statements Disclaimer This presentation contains certain forward-looking statements, including with respect to products, product development activities, regulatory submissions and authorizations and other matters. Forward-looking statements are not guarantees of future performance or results. Known and unknown factors that could cause actual performance or results to be materially different from those expressed or implied in these statements include, but are not limited to: risk that the Company's exploration of strategic alternatives may not result in any definitive transaction or enhance stockholder value and may create a distraction or uncertainty that may adversely affect operating results, business or investor perceptions; the diversion of management’s attention from the Company's ongoing business and regular business responsibilities due to the Company's exploration of strategic alternatives; ability to resolve the Company's ongoing manufacturing challenges and satisfy customer demand; ability to market and sell products, whether through our internal, direct sales force or third parties; impact of significant customer concentration in the genomics business; failure of distributors or other customers to meet purchase forecasts, historic purchase levels or minimum purchase requirements for our products; ability to manufacture products in accordance with applicable specifications, performance standards and quality requirements; ability to obtain, and timing and cost of obtaining, necessary regulatory approvals for new products or new indications or applications for existing products; ability to comply with applicable regulatory requirements; ability to effectively resolve warning letters, audit observations and other findings or comments from the U.S. Food and Drug Administration (“FDA”) or other regulators; the impact of the novel coronavirus (“COVID-19”) pandemic on the Company's business and ability to successfully develop new products, validate the expanded use of existing collector products, receive necessary regulatory approvals and authorizations and commercialize such products for COVID-19 testing; changes in relationships, including disputes or disagreements, with strategic partners or other parties and reliance on strategic partners for the performance of critical activities under collaborative arrangements; ability to meet increased demand for the Company’s products; impact of replacing distributors; inventory levels at distributors and other customers; ability of the Company to achieve its financial and strategic objectives and continue to increase its revenues, including the ability to expand international sales; impact of competitors, competing products and technology changes; reduction or deferral of public funding available to customers; competition from new or better technology or lower cost products; ability to develop, commercialize and market new products; market acceptance of oral fluid or urine testing, collection or other products; market acceptance and uptake of microbiome informatics, microbial genetics technology and related analytics services; changes in market acceptance of products based on product performance or other factors, including changes in testing guidelines, algorithms or other recommendations by the Centers for Disease Control and Prevention (“CDC”) or other agencies; ability to fund research and development and other products and operations; ability to obtain and maintain new or existing product distribution channels; reliance on sole supply sources for critical products and components; availability of related products produced by third parties or products required for use of our products; impact of contracting with the U.S. government; impact of negative economic conditions; ability to maintain sustained profitability; ability to utilize net operating loss carry forwards or other deferred tax assets; volatility of the Company’s stock price; uncertainty relating to patent protection and potential patent infringement claims; uncertainty and costs of litigation relating to patents and other intellectual property; availability of licenses to patents or other technology; ability to enter into international manufacturing agreements; obstacles to international marketing and manufacturing of products; ability to sell products internationally, including the impact of changes in international funding sources and testing algorithms; adverse movements in foreign currency exchange rates; loss or impairment of sources of capital; ability to attract and retain qualified personnel; exposure to product liability and other types of litigation; changes in international, federal or state laws and regulations; customer consolidations and inventory practices; equipment failures and ability to obtain needed raw materials and components; the impact of terrorist attacks and civil unrest; and general political, business and economic conditions. These and other factors that could affect our results are discussed more fully in our SEC filings, including our registration statements, Annual Report on Form 10-K for the year ended December 31, 2020, Quarterly Reports on Form 10-Q, and other filings with the SEC. Although forward-looking statements help to provide information about future prospects, readers should keep in mind that forward-looking statements may not be reliable. Readers are cautioned not to place undue reliance on the forward-looking statements. The forward-looking statements are made as of the date of this press release and OraSure Technologies undertakes no duty to update these statements.

4Q21 and FY21 Financial Results FINANCIAL METRIC 4Q21/FY21 RESULTS 4Q20/FY20 RESULTS YEAR-OVER-YEAR GROWTH RATE Quarterly Revenue $63.6 million $62.9 million 1% Annual Revenue $233.7 million $171.7 million 36% Annual Revenue Diagnostics $90.0 million $65.2 million 38% Annual Revenue Molecular Solutions $143.6 million $106.5 million 35%
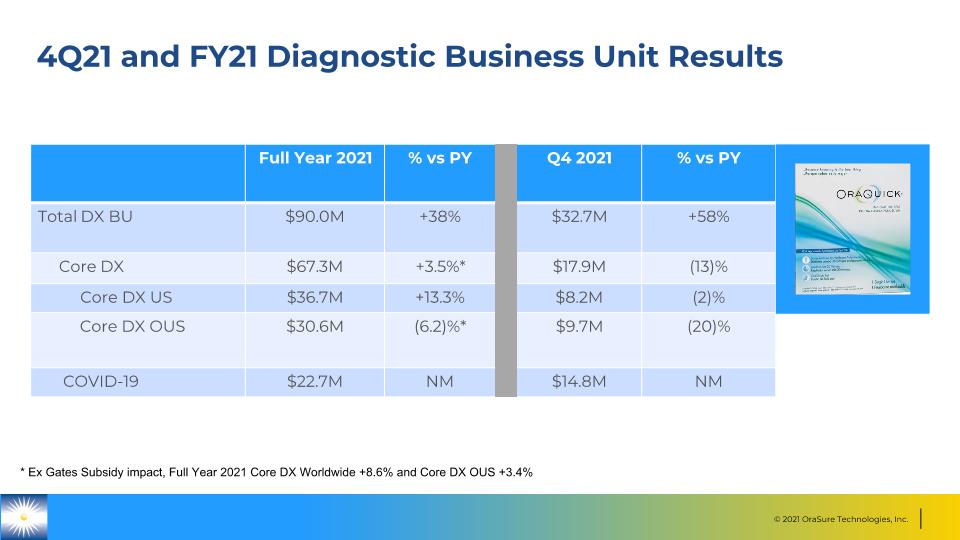
4Q21 and FY21 Diagnostic Business Unit Results Full Year 2021 % vs PY Q4 2021 % vs PY Total DX BU $90.0M +38% $32.7M +58% Core DX $67.3M +3.5%* $17.9M (13)% Core DX US $36.7M +13.3% $8.2M (2)% Core DX OUS $30.6M (6.2)%* $9.7M (20)% COVID-19 $22.7M NM $14.8M NM * Ex Gates Subsidy impact, Full Year 2021 Core DX Worldwide +8.6% and Core DX OUS +3.4%
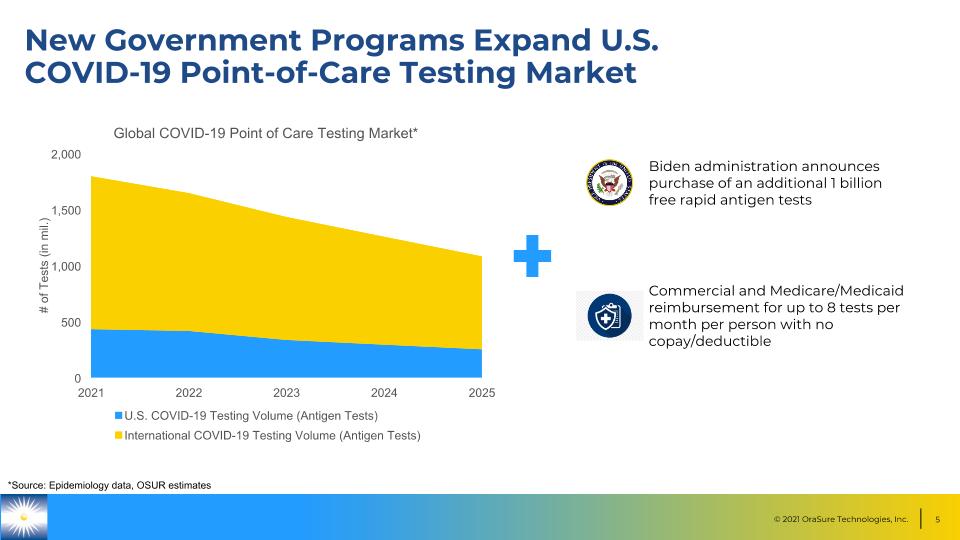
New Government Programs Expand U.S. COVID-19 Point-of-Care Testing Market *Source: Epidemiology data, OSUR estimates Biden administration announces purchase of an additional 1 billion free rapid antigen tests Commercial and Medicare/Medicaid reimbursement for up to 8 tests per month per person with no copay/deductible

InteliSwab® Expansions InteliSwab® detects Omicron with same limit of detection as other variants of concern and original Wuhan strain Pediatric indication for use in children ages 2-14 when performed by an adult Launching the new reporting app, InteliSwab ® Connect
InteliSwab® Operations Restructured – New Operations Leadership & Outside Expertise Mapped Processes & Identified Bottlenecks Implement Solutions Impact Faster to Execute
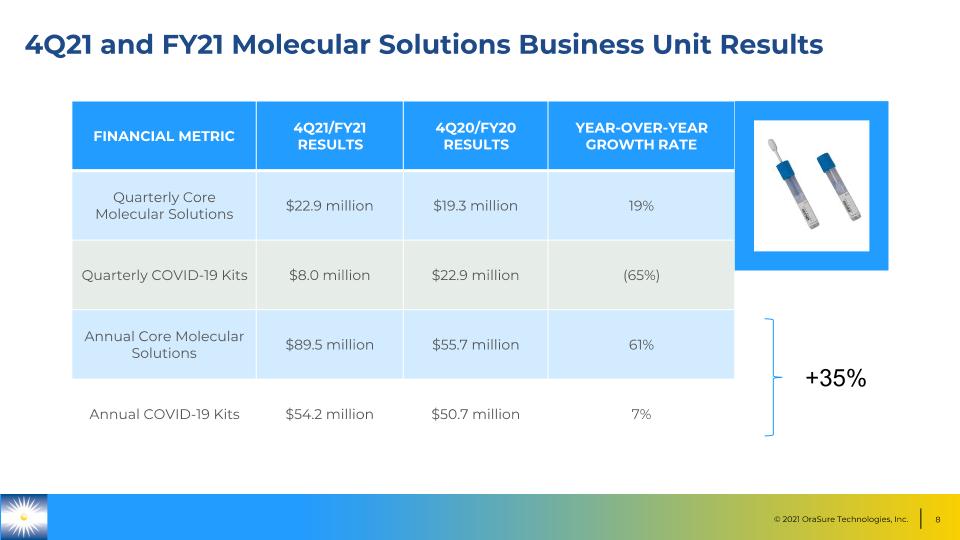
4Q21 and FY21 Molecular Solutions Business Unit Results FINANCIAL METRIC 4Q21/FY21 RESULTS 4Q20/FY20 RESULTS YEAR-OVER-YEAR GROWTH RATE Quarterly Core Molecular Solutions $22.9 million $19.3 million 19% Quarterly COVID-19 Kits $8.0 million $22.9 million (65%) Annual Core Molecular Solutions $89.5 million $55.7 million 61% Annual COVID-19 Kits $54.2 million $50.7 million 7% +35%
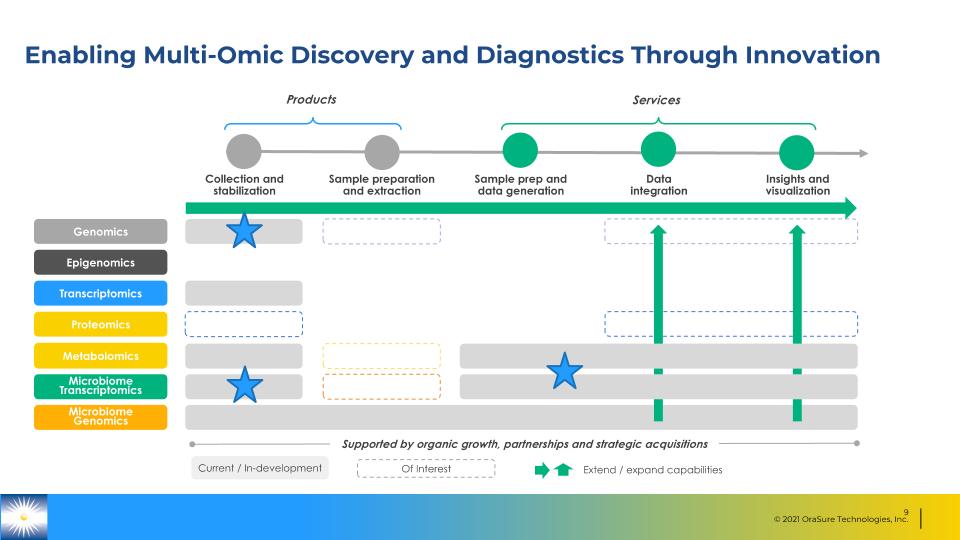
Enabling Multi-Omic Discovery and Diagnostics Through Innovation Collection and stabilization Sample preparation and extraction Sample prep and data generation Data integration Insights and visualization Genomics Transcriptomics Epigenomics Proteomics Metabolomics Microbiome Transcriptomics Current / In-development Of Interest Supported by organic growth, partnerships and strategic acquisitions Extend / expand capabilities Microbiome Genomics Products Services
Meta-transcriptome Launch FDA Clearance for Gut Kit New Cancer Chemistry Enabling Multi-Omic Discovery and Diagnostics Through Innovation
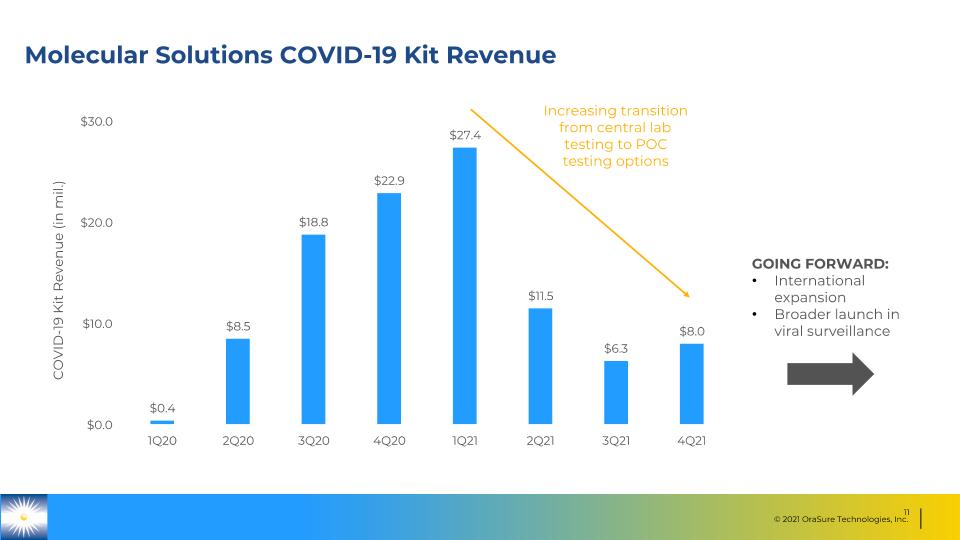
Molecular Solutions COVID-19 Kit Revenue Increasing transition from central lab testing to POC testing options GOING FORWARD: International expansion Broader launch in viral surveillance
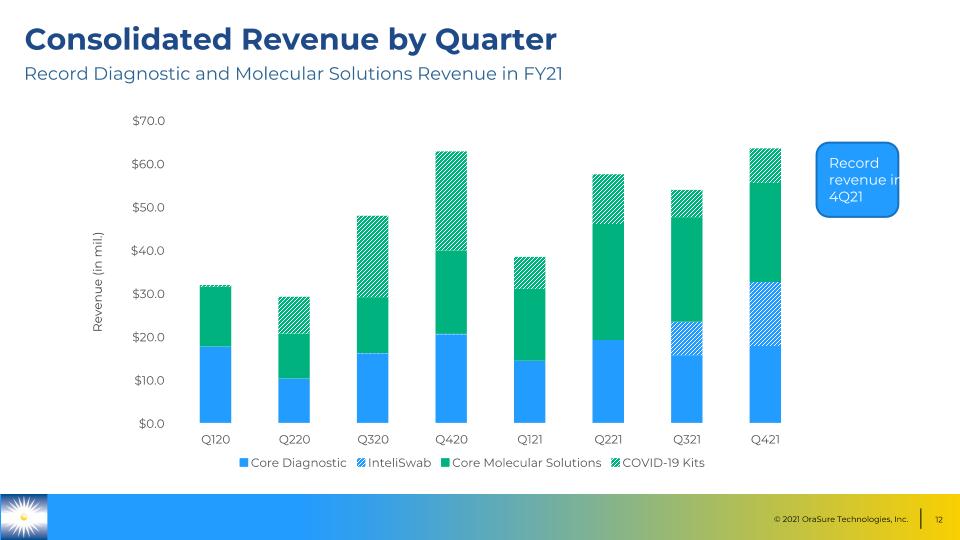
Consolidated Revenue by Quarter Revenue (in mil.) Record Diagnostic and Molecular Solutions Revenue in FY21 Record revenue in 4Q21
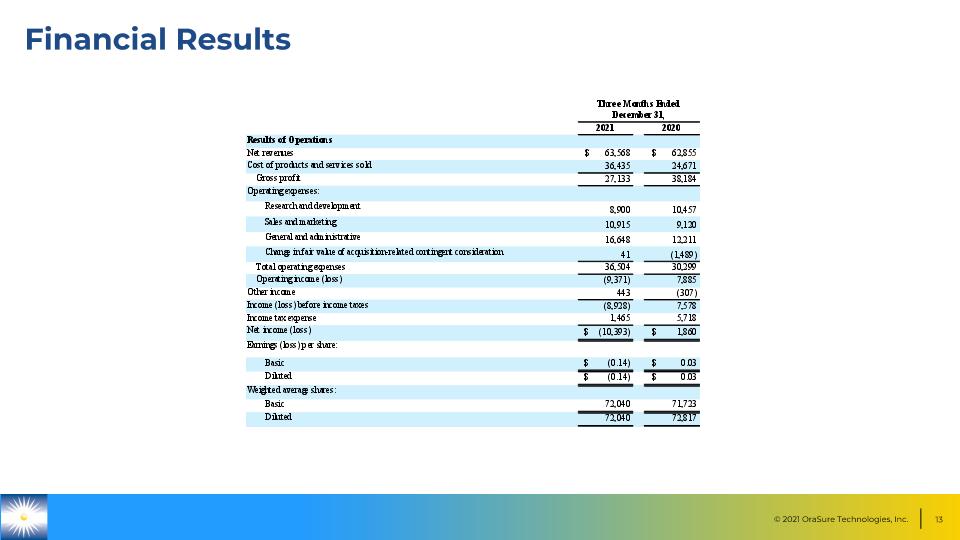
Financial Results

Summary Commercially tied to high growth areas of healthcare such as consumer/clinical genomics and shift to direct-to-patient/near patient testing Increased investment in internal R&D pipeline and reinvigorating innovation Significant opportunity with InteliSwab™ to drive growth and fund additional investment Strong balance sheet with focus on deploying capital to drive growth and leverage infrastructure Smart Science Made Simple

Q&A














