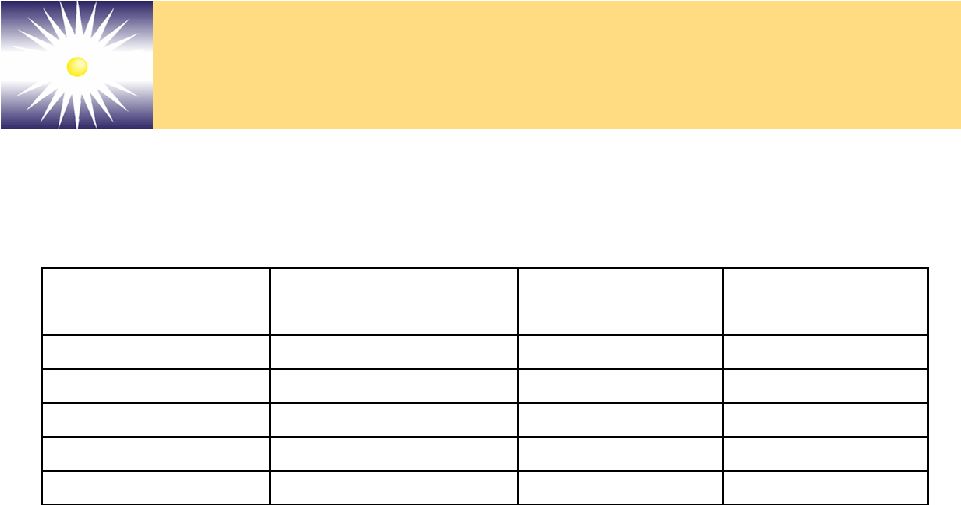
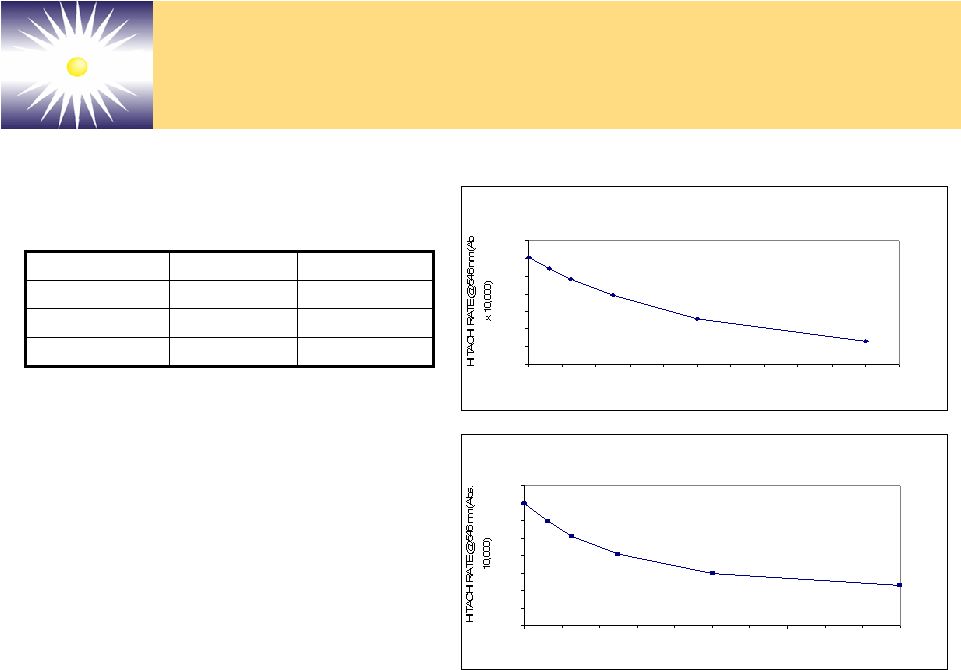
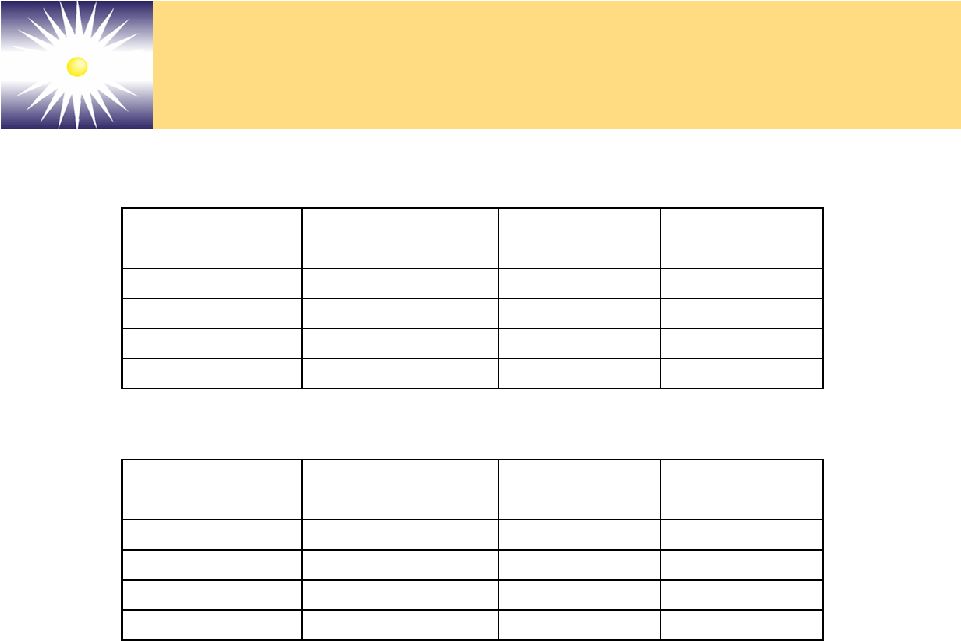
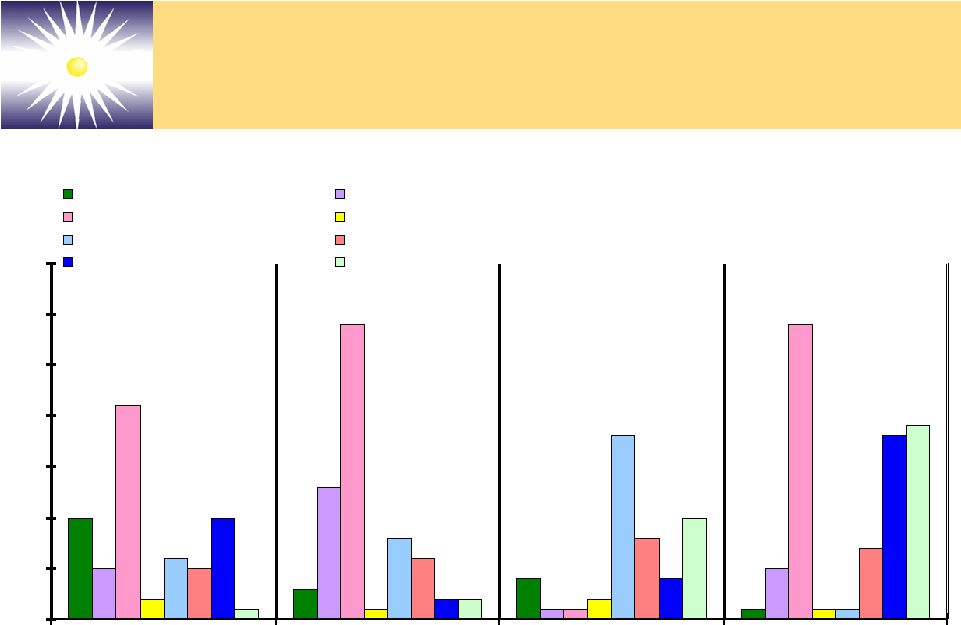
Forward-Looking Statements These slides and the associated presentation contain certain These slides and the associated presentation contain certain forward-looking statements, including statements with respect forward-looking statements, including statements with respect to revenues, earnings, technology, new products, product to revenues, earnings, technology, new products, product performance, markets, regulatory filings and approvals, and performance, markets, regulatory filings and approvals, and business plans. Factors affecting these statements include, business plans. Factors affecting these statements include, but are not limited to, the ability to develop new technology, but are not limited to, the ability to develop new technology, technology changes, ability to fund research and technology changes, ability to fund research and development, required regulatory approvals, product development, required regulatory approvals, product performance and market acceptance of products. Please see performance and market acceptance of products. Please see the Company’s SEC filings, including its registration the Company’s SEC filings, including its registration statements, and the Company’s most recent Form 10-K and statements, and the Company’s most recent Form 10-K and Form 10-Q, for a more detailed description of specific factors Form 10-Q, for a more detailed description of specific factors that may cause actual results or events to differ materially that may cause actual results or events to differ materially from those described in the forward-looking statements. The from those described in the forward-looking statements. The Company undertakes no duty to update these statements. Company undertakes no duty to update these statements. |