Exhibit 99.2
| | | | |
2. HALF-YEAR MANAGEMENT REPORT | | | 37 | |
A/ Significant events of the first half of 2019 | | | 37 | |
B/ Events subsequent to June 30, 2019 | | | 39 | |
C/ Consolidated financial statements for the first half of 2019 | | | 40 | |
D/ Risk factors and related party transactions | | | 59 | |
E/ Outlook | | | 59 | |
F/ Appendix – research and Dévelopment Pipeline | | | 61 | |
2. HALF-YEAR MANAGEMENT REPORT
A/ SIGNIFICANT EVENTS OF THE FIRST HALF OF 2019
A.1. FIRST-HALF OVERVIEW
In a fast-changing industry environment, Sanofi continued its transformation during the first half of 2019, as it delivers on its mission as a global healthcare leader at the cutting edge of innovation. We are refocusing our R&D activities to prioritize Specialty Care – including oncology, immunology, rare diseases and rare blood disorders – while maintaining a strong commitment to Vaccines.
On January 7, 2019, Sanofi and Regeneron announced that they had restructured their global Immuno-Oncology Discovery and Development Agreement for new cancer treatments. The 2015 agreement was due to end inmid-2020, and the restructured agreement provides for ongoing collaborative development of two clinical stage bispecific antibody programs. This gives Sanofi increased flexibility to advance its early-stage immuno-oncology pipeline independently, while Regeneron retains all rights to its other immuno-oncology discovery and development programs.
On April 8, 2019, Sanofi and Alnylam concluded the research and option phase of the companies’ 2014 RNAi therapeutics alliance in rare genetic diseases. The material collaboration terms for patisiran, vutrisiran(ALN-TTRsc02) and fitusiran, as previously announced, continue unchanged. As part of this agreement, Alnylam will advance an additional investigational asset in a rare genetic disease through studies enabling the filing of an Investigational New Drug (IND) application. Sanofi will be responsible for any potential further development or commercialization of the asset. In the event of approval, Alnylam will be eligible to receive double-digit royalties on global net sales of the product.
On June 18, 2019, Sanofi and Google announced that they are establishing a new Innovation Lab with the ambition of transforming how future medicines and health services are developed by tapping into emerging technologies. The collaboration aims to change how Sanofi develops new treatments and will focus on three key objectives: to better understand patients and diseases, to increase Sanofi’s operational efficiency, and to improve the experience of Sanofi’s patients and customers.
Net sales for the first half of 2019 amounted to €17,019 million, 5.9% higher than in the first half of 2018. At constant exchange rates (CER)1, net sales rose by 4.1%, mainly reflecting good performances for Dupixent®, the Rare Diseases franchise and the Vaccines segment, and more generally sales growth in emerging markets. Those effects were partially offset by lower sales for the Diabetes franchise in the United States and for Established Prescription Products, in particular following the divestment of our European generics business (Zentiva) in 2018. At constant exchange rates and on a constant structure basis (CER/CS, see definition below), net sales were up 4.8%.
Net income attributable to equity holders of Sanofi amounted to €1,050 million, down 40.9%; this was due mainly to impairment losses charged against intangible assets in the period. Earnings per share was €0.84, 40.8% lower than in the first half of 2018. Business net income2was €3,406 million, up 7.9% on the first half of 2018, while business earnings per share (business EPS)3 was 7.9% higher than in the first half of 2018 at €2.73.
A.2. RESEARCH AND DEVELOPMENT
Highlights of our research and development activities in the first half of 2019 in the Pharmaceuticals segment included the entry into Phase III ofcemiplimab (Libtayo®), as an adjuvant in the treatment of cutaneous squamous cell carcinoma;venglustat(GCS inhibitor), in autosomal dominant polycystic kidney disease (ADPKD); anddupilumab (Dupixent®) in chronic obstructive pulmonary disease. In the Vaccines segment, the following products entered Phase III: the hexavalent pediatric vaccine Shan6 (Diphtheria, Tetanus, Pertussis, Polio, Hepatitis B, Hemophilus influenzae b); the monoclonal antibody nirsevimab (collaboration with Medimmune) in the treatment of Respiratory Syncytial Virus (RSV); and the vero cell rabies vaccine VerorabVax® (VRVg).
Sanofi obtained regulatory marketing approval for a number of products in the first half of 2019. The Democratic Republic of Congo (DRC) granted marketing approval forfexinidazole for the treatment of human African trypanosomiasis (HAT), more commonly known as sleeping sickness. In the United States, Cablivi®was approved in association with plasma exchange and immunosuppression for the treatment of acquired thrombotic thrombocytopenic purpura (aTTP) in adults. Also in the United States,Dupixent® was approved for the treatment of inadequately controlled moderate to severe atopic dermatitis in adolescents aged 12 to 17, and for use with other medicines in adults with inadequately controlled chronic rhinosinusitis with nasal polyposis. In Europe, Dupixent® was approved as a treatment for severe asthma. The European Commission and the US Food and Drug Administration (FDA) approved a new indication forPraluent® (alirocumab) to
| 1 | Non-GAAP financial measure: see definition in C.3., “Net sales”. |
| 2 | Non-GAAP financial measure: see definition in C.2., “Business net income”. |
| 3 | Non-GAAP financial measure: see definition in C.2., “Business net income”. |
37 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
reduce cardiovascular risk in adults with established atherosclerotic cardiovascular disease. In Europe, conditional approval has been granted forLibtayo®(cemiplimab) in the treatment of patients with metastatic or locally advanced cutaneous squamous cell carcinoma (CSCC) who are not candidates for surgery or curative radiotherapy.
For an update on our research and development pipeline, refer to Section F of this half-year management report.
A.3. OTHER SIGNIFICANT EVENTS
A.3.1. CORPORATE GOVERNANCE
On February 12, 2019, Sanofi announced the appointment of Ameet Nathwani, M.D. as Chief Digital Officer in addition to his role as Executive Vice President, Chief Medical Officer. As Chief Digital Officer, Dr. Nathwani will be responsible for enhancing Sanofi’s strategy of integrating digital technologies and medical science to improve patient outcomes. His mandate will include scaling up Sanofi’s ongoing portfolio of digital initiatives by developing broad external partnerships, building out internal infrastructures, and exploring new business opportunities for Sanofi in the digital space.
The Annual General Meeting of Sanofi shareholders was held on April 30, 2019, chaired by Serge Weinberg. All of the resolutions submitted to the vote were adopted by the shareholders. The meeting approved the individual company financial statements and consolidated financial statements for the year ended December 31, 2018, and resolved to distribute a cash dividend of €3.07 per share paid on May 13, 2019. The meeting also approved the reappointment of Suet-Fern Lee and Serge Weinberg as directors, and ratified theco-opting of Christophe Babule as a director. Following the meeting, the new Board of Directors still has 16 members, including six women and two employee representative directors; the majority of the Board members are independent directors. At the Board meeting that followed the Annual General Meeting, Serge Weinberg was reappointed as Chairman of the Board of Directors of Sanofi; Melanie Lee was appointed to the Appointments, Governance and CSR Committee; and Carole Piwnica was appointed to the Compensation Committee.
At a Board meeting on June 6, 2019, the Sanofi Board of Directors unanimously appointed Paul Hudson as Chief Executive Officer of Sanofi, to succeed Olivier Brandicourt who has decided to retire. Paul Hudson, who most recently was Chief Executive Officer of Novartis Pharmaceuticals and a member of the Executive Committee of Novartis, will take up his post at Sanofi on September 1, 2019. Aged 51, Paul Hudson has strong international experience, particularly in the United States, Japan and Europe. He has spent his28-year career with major pharmaceutical companies such as Schering Plough, Astra Zeneca and Novartis. Throughout his various management positions, he has demonstrated strategic vision, strong leadership and the ability to rise to the greatest challenges, especially in the fields of innovation and digital transformation. He also has a robust track record in successful major product launches. Paul Hudson will relocate to Paris.
A.3.2. LEGAL AND ARBITRATION PROCEEDINGS
For a description of the most significant developments in legal and arbitration proceedings since publication of the financial statements for the year ended December 31, 2018, refer to Note B.14. to the condensed half-year consolidated financial statements.
The following events have occurred in respect of litigation, arbitration and other legal proceedings in which Sanofi and its affiliates are involved:
PATENTS
| | • | | Lantus® Mylan Patent Litigation (United States) |
In the New Jersey lawsuit, trial is tentatively scheduled for January 2020.
In the proceedings relating to the ten petitions filed by Mylan before the Patent Trial and Appeal Board (PTAB) challenging the validity of five Sanofi device patents, in April 2019, the PTAB decided to move forward with theInter Partes Review (IPR) on nine of these ten petitions. The PTAB oral hearing is scheduled for January 2020.
GOVERNMENT INVESTIGATIONS AND RELATED LITIGATION
In the lawsuit filed in April 2018 in the US District Court for the Southern District of New York, alleging violations of the False Claims Act and 29state-law analogs by Sanofi US and other manufacturer and pharmacy benefit managers (PBMs) defendants regarding Lantus® and Apidra®, the Court dismissed the case in July 2019.
A putative class of diabetes patients alleging violations of the Racketeer Influenced and Corrupt Organizations Act (“RICO Act”) and various state unfair/deceptive trade practices statutes in connection with the pricing of Lantus® filed a Second Amended Complaint in the Federal Court of New Jersey in March 2019 after several of their claims had previously been dismissed, and the three manufacturer defendants have filed a motion to dismiss. In the same court, the plaintiffs inMSP Recovery Claims, Series LLC filed a Second Amended Complaint in April 2019, and the same defendants have filed a
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 38
motion to dismiss. In addition to the complaint filed by Minnesota in October 2018 in the Federal Court of New Jersey, Kentucky filed its own complaint against the same three defendants in Kentucky state court (Franklin Country Circuit Court) in May 2019. The defendants are preparing motions to dismiss in both of these actions.
In July 2019, Sanofi US received a subpoena from the New York Attorney General’s Office in connection with an inquiry related to insulin pricing. The subpoena requests the production of various documents relating to Sanofi’s insulin products including documents regarding pricing, discount programs, sales and expenses, contracting, marketing materials and legal proceedings. Sanofi US is cooperating with this inquiry.
Several committees of the US Congress are pursuing inquiries focused on pharmaceutical pricing, with an emphasis on the historical pricing of insulin. For example, in January 2019, the Company received an inquiry from the U.S. House of Representatives, Committee on Oversight and Reform, seeking information about the historical pricing of Lantus® and Renvela® and in February 2019, the Company received an inquiry letter from the US Senate Committee on Finance concerning the historical pricing of Sanofi’s insulin products in the US. Sanofi US is cooperating with these ongoing Congressional inquiries, which may involve hearings and/or congressional reports that may portray pharmaceutical company business activities in a negative light. While we cannot predict the outcome of these matters, they – as well as the enactment of various policy proposals that are currently pending – could affect our ability to price our drugs, and in particular our insulin products, in the US marketplace. Moreover, potential policy measures such as importation and international reference pricing could impact our business in other jurisdictions.
In France, in the claim filed in 2017 by the FrenchCaisse Nationale d’Assurance Maladie (French Social Security), an oral pleading took place before the Commercial Court in June 2019 regarding the statute of limitations. Judgment is expected to be rendered on September 17, 2019.
A.3.3. OTHER EVENTS
On March 13, 2019, Sanofi announced that it had successfully placed a €2 billion bond issue under its Euro Medium Term Notes (EMTN) program, in three tranches:
| | • | | €850 million of fixed-rate bonds maturing March 2022, with annual coupons and bearing interest at an annual rate of 0.000%; |
| | • | | €650 million of fixed-rate bonds maturing March 2029, with annual coupons and bearing interest at an annual rate of 0.875%; and |
| | • | | €500 million of fixed-rate bonds maturing March 2034, with annual coupons and bearing interest at an annual rate of 1.250%. |
This issue reduces the average cost and extends the average maturity of Sanofi’s debt. Sanofi intends to use the net proceeds of those bond issues for general corporate purposes.
B/ EVENTS SUBSEQUENT TO JUNE 30, 2019
On July 23, 2019, Sanofi announced the signature of an agreement with Roche for the exclusiveover-the-counter (OTC) rights to Tamiflu® for the prevention and treatment of influenza (flu) in the US. Under the terms of the agreement, Sanofi will be responsible for leading FDA negotiations for the OTC switch and subsequent exclusive marketing, scientific engagement and distribution of Tamiflu® OTC in the US. Tamiflu® is currently sold in the US by Genentech, a member of the Roche Group, for prescription use.
Topline results from three Phase III trials of ZynquistaTM (sotagliflozin) in adults with type 2 diabetes from the InSynchrony clinical program were announced on July 26. Given the primary endpoint results of blood sugar control (HbA1c) reduction in the SOTA-CKD3 and SOTA-CKD4 studies, Sanofi provided notice to Lexicon that it is terminating the collaboration to develop, manufacture, and commercialize ZynquistaTM in all ongoing global type 1 and type 2 diabetes programs. At this time, the ongoing Phase III clinical trials will continue and there will be no immediate changes. Sanofi has expressed willingness to work with Lexicon to ensure a smooth transition of the studies. Sanofi remains committed to working and supporting the investigators and patients enrolled in the studies while next steps are discussed with Lexicon.
39 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C/ CONSOLIDATED FINANCIAL STATEMENTS FOR THE FIRST HALF OF 2019
Unless otherwise indicated, all financial data in this report are presented in accordance with international financial reporting standards (IFRS), including international accounting standards and interpretations (see Note A.1. to the condensed half-year consolidated financial statements).
Consolidated income statements for the six months ended June 30, 2018 and June 30, 2019
| | | | | | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | as % of net sales | | | June 30, 2018 (6 months) | | | as % of net sales | |
Net sales | | | 17,019 | | | | 100.0 | % | | | 16,074 | | | | 100.0 | % |
Other revenues | | | 674 | | | | 4.0 | % | | | 533 | | | | 3.3 | % |
Cost of sales | | | (5,385 | ) | | | (31.6 | %) | | | (5,265 | ) | | | (32.8 | %) |
Gross profit | | | 12,308 | | | | 72.3 | % | | | 11,342 | | | | 70.6 | % |
Research and development expenses | | | (2,972 | ) | | | (17.5 | %) | | | (2,755 | ) | | | (17.1 | %) |
Selling and general expenses | | | (4,835 | ) | | | (28.4 | %) | | | (4,819 | ) | | | (30.0 | %) |
Other operating income | | | 273 | | | | | | | | 323 | | | | | |
Other operating expenses | | | (466 | ) | | | | | | | (165 | ) | | | | |
Amortization of intangible assets | | | (1,116 | ) | | | | | | | (999 | ) | | | | |
Impairment of intangible assets | | | (1,840 | ) | | | | | | | (101 | ) | | | | |
Fair value remeasurement of contingent consideration | | | 190 | | | | | | | | 10 | | | | | |
Restructuring costs and similar items | | | (747 | ) | | | | | | | (607 | ) | | | | |
Other gains and losses, and litigation | | | 317 | | | | | | | | (67 | ) | | | | |
Operating income | | | 1,112 | | | | 6.5 | % | | | 2,162 | | | | 13.5 | % |
Financial expenses | | | (244 | ) | | | | | | | (202 | ) | | | | |
Financial income | | | 94 | | | | | | | | 97 | | | | | |
Income before tax and investments accounted for using the equity method | | | 962 | | | | 5.7 | % | | | 2,057 | | | | 12.8 | % |
Income tax expense | | | (13 | ) | | | | | | | (297 | ) | | | | |
Share of profit/(loss) from investments accounted for using the equity method | | | 116 | | | | | | | | 75 | | | | | |
Net income | | | 1,065 | | | | | | | | 1,835 | | | | | |
Net income attributable tonon-controlling interests | | | 15 | | | | | | | | 57 | | | | | |
Net income attributable to equity holders of Sanofi | | | 1,050 | | | | 6.2 | % | | | 1,778 | | | | 11.1 | % |
Average number of shares outstanding (million) | | | 1,247.2 | | | | | | | | 1,247.8 | | | | | |
Average number of shares after dilution (million) | | | 1,254.7 | | | | | | | | 1,254.9 | | | | | |
– Basic earnings per share (in euros) | | | 0.84 | | | | | | | | 1.42 | | | | | |
– Diluted earnings per share (in euros) | | | 0.84 | | | | | | | | 1.42 | | | | | |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 40
C.1. SEGMENT INFORMATION
C.1.1. OPERATING SEGMENTS
In accordance with IFRS 8 (Operating Segments), the segment information reported by Sanofi is prepared on the basis of internal management data provided to the Chief Executive Officer, who is the chief operating decision maker. The performance of those segments is monitored individually using internal reports and common indicators. The operating segment disclosures required under IFRS 8 are provided in Note B.20. to the condensed half-year consolidated financial statements.
Sanofi has three operating segments: Pharmaceuticals, Consumer Healthcare and Vaccines.
The Pharmaceuticals segment comprises the commercial operations of the following global franchises: Specialty Care (Rare Diseases, Multiple Sclerosis, Oncology, Immunology and Rare Blood Disorder), Diabetes & Cardiovascular, and Established Prescription Products, together with research, development and production activities dedicated to our Pharmaceuticals segment. This segment also includes all associates whose activities are related to pharmaceuticals, in particular our share of Regeneron.
The Consumer Healthcare segment comprises, for all geographical territories, the commercial operations for our Consumer Healthcare products, together with research, development and production activities dedicated to those products.
The Vaccines segment comprises, for all geographical territories (including from January 1, 2017 certain territories previously included in the Sanofi Pasteur MSD joint venture), the commercial operations of Sanofi Pasteur, together with research, development and production activities dedicated to vaccines.
Inter-segment transactions are not material.
The costs of our global functions (Medical Affairs, External Affairs, Finance, Human Resources, Legal Affairs, Information Solutions & Technologies, Sanofi Business Services, etc.) are managed centrally at group-wide level. The costs of those functions are presented within the “Other” category. That category also includes other reconciling items such as retained commitments in respect of divested activities.
C.1.2. BUSINESS OPERATING INCOME
We report segment results on the basis of “business operating income”. This indicator is used internally by Sanofi’s chief operating decision maker to measure the performance of each operating segment and to allocate resources. For a definition of “business operating income”, and a reconciliation between that indicator andIncome before tax and investments accounted for using the equity method, refer to Note B.20.1 to our condensed half-year consolidated financial statements.
The impact of IFRS 16 on our business net income is reflected by (i) the elimination of the depreciation charged againstright-of-use assets recognized under IFRS 16 and (ii) the inclusion of the IAS 17 lease expense. This allows for consistency of presentation with the comparative period, Sanofi having applied the simplified retrospective method on transition.
C.2. BUSINESS NET INCOME
We believe that understanding of our operational performance by our management and our investors is enhanced by reporting “business net income”. Thisnon-GAAP financial measure represents business operating income, less net financial expenses and the relevant income tax effects.
Business net income for the first half of 2019 was €3,406 million, 7.9% higher than in the first half of 2018 (€3,156 million). That represents 20.0% of net sales, compared with 19.6% in the first half of 2018.
We also report “business earnings per share” (business EPS), anon-GAAP financial measure which we define as business net income divided by the weighted average number of shares outstanding.
Business EPS was €2.73 for the first half of 2019, 7.9% higher than the 2018 first-half figure of €2.53, based on an average number of shares outstanding of 1,247.2 million for the first half of 2019 and 1,247.8 million for the first half of 2018.
41 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
The table below reconciles our business operating income to our business net income:
| | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | December 31, 2018 (12 months) | |
Business operating income | | | 4,454 | | | | 4,126 | | | | 8,884 | |
Financial income and expenses | | | (130 | ) | | | (105 | ) | | | (271 | ) |
Income tax expense | | | (918 | ) | | | (865 | ) | | | (1,794 | ) |
Business net income | | | 3,406 | | | | 3,156 | | | | 6,819 | |
We define business net income asNet income attributable to equity holders of Sanofi determined under IFRS, excluding the following items:
| | • | | amortization and impairment losses charged against intangible assets (other than software and other rights of an industrial or operational nature); |
| | • | | fair value remeasurements of contingent consideration relating to business combinations or divestments; |
| | • | | other impacts associated with acquisitions (including impacts of acquisitions on investments accounted for using the equity method); |
| | • | | restructuring costs and similar items1; |
| | • | | other gains and losses (including gains and losses on major disposals ofnon-current assets)2; |
| | • | | the impacts of IFRS 16 on lease accounting; |
| | • | | other costs and provisions related to litigation2; |
| | • | | the tax effects of the items listed above, and the impact of major tax disputes; and |
| | • | | the portion attributable tonon-controlling interests of the items listed above. |
| 1 | Presented in the line itemRestructuring costs and similar items in the consolidated income statement. |
| 2 | Presented in the line itemOther gains and losses, and litigationin the consolidated income statement. |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 42
The table below reconciles our business net income toNet income attributable to equity holders of Sanofi:
| | | | | | | | | | | | |
| | | June 30, | | | June 30, | | | December 31, | |
| | | 2019 | | | 2018 | | | 2018 | |
| (€ million) | | (6 months) | | | (6 months) | | | (12 months) | |
Net income attributable to equity holders of Sanofi | | | 1,050 | | | | 1,778 | | | | 4,306 | |
Amortization of intangible assets(a) | | | 1,116 | | | | 999 | | | | 2,170 | |
Impairment of intangible assets(b) | | | 1,840 | | | | 101 | | | | 718 | |
Fair value remeasurement of contingent consideration | | | (190 | ) | | | (10 | ) | | | (117 | ) |
Expenses arising from the impact of acquisitions on inventories | | | 3 | | | | 99 | | | | 114 | |
Other expenses related to business combinations | | | — | | | | 10 | | | | 28 | |
Restructuring costs and similar items | | | 747 | | | | 607 | | | | 1,480 | |
Other gains and losses, and litigation(c) | | | (317 | ) | | | 67 | | | | (502 | ) |
Impacts of IFRS 16 on lease accounting(d) | | | 9 | | | | — | | | | — | |
Tax effects of the items listed above: | | | (905 | ) | | | (475 | ) | | | (1,125 | ) |
amortization and impairment of intangible assets | | | (711 | ) | | | (275 | ) | | | (692 | ) |
fair value remeasurement of contingent consideration | | | 24 | | | | 11 | | | | 38 | |
expenses arising from the impact of acquisitions on inventories | | | — | | | | (23 | ) | | | (27 | ) |
other expenses related to business combinations | | | — | | | | — | | | | (6 | ) |
tax effects of restructuring costs and similar items | | | (197 | ) | | | (183 | ) | | | (435 | ) |
other tax effects | | | (21 | ) | | | (5 | ) | | | (3 | ) |
Other tax items(e) | | | — | | | | (93 | ) | | | (188 | ) |
Share of items listed above attributable tonon-controlling interests | | | — | | | | (1 | ) | | | (2 | ) |
Investments accounted for using the equity method: restructuring costs and expenses arising from the impact of acquisitions | | | 53 | | | | 74 | | | | (76 | ) |
Items relating to the Animal Health business(f) | | | — | | | | — | | | | 13 | |
Business net income | | | 3,406 | | | | 3,156 | | | | 6,819 | |
Average number of shares outstanding (million) | | | 1,247.2 | | | | 1,247.8 | | | | 1,247.1 | |
Basic earnings per share (in euros) | | | 0.84 | | | | 1.42 | | | | 3.45 | |
Reconciling items per share (in euros) | | | 1.89 | | | | 1.11 | | | | 2.02 | |
Business earnings per share (in euros) | | | 2.73 | | | | 2.53 | | | | 5.47 | |
| (a) | Includes amortization expense generated by the remeasurement of intangible assets in connection with business combinations: €1,060 million in the six months ended June 30, 2019; €934 million in the six months ended June 30, 2018; and €1,957 million in the year ended December 31, 2018. |
| (b) | Includes impairment losses of €1,609 million taken against Eloctate®franchise assets. |
| (c) | In 2019, this line mainly comprises a gain arising from litigation. For 2018, this line consists mainly of separation costs associated with the process of disinvesting from the Generics business in Europe, before tax effects. |
| (d) | Impacts of the new accounting standard on leases (IFRS 16), applied from January 1, 2019 using the simplified retrospective method without restatement of comparative periods. For comparative purposes, business net income continues to be reported in accordance with the lease accounting policies applicable under the previous standard (IAS 17). |
| (e) | For 2018, this line comprises the direct and indirect impacts of US tax reform. |
| (f) | This line shows the residual impacts of the divestment of our Animal Health business. |
The most significant reconciling items between our business net income andNet income attributable to equity holders of Sanofi relate to (i) the purchase accounting effects of our acquisitions and business combinations, particularly the amortization and impairment of intangible assets (other than software and other rights of an industrial or operational nature) and (ii) the impacts of events regarded asnon-recurring, where the amounts involved are particularly significant. We believe that excluding thosenon-cash ornon-recurring charges enhances an investor’s understanding of our underlying economic performance, because we do not consider that the excluded charges reflect the combined entity’s ongoing operating performance. Rather, we believe that each of the excluded charges reflects the decision to acquire the businesses concerned.
The principal purchase accounting effects of acquisitions and business combinations on net income are:
| | • | | amortization and net impairment losses charged against intangible assets (other than software and other rights of an industrial or operational nature), net of taxes andnon-controlling interests; and |
| | • | | the incremental cost of sales incurred on the workdown of acquired inventories remeasured at fair value, net of taxes. |
43 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
We believe (subject to the limitations described below) that disclosing our business net income enhances the comparability of our operating performance, for the following reasons:
| • | | the elimination of charges related to the purchase accounting effect of our acquisitions and business combinations (particularly amortization and impairment of finite-lived intangible assets, other than software and other rights of an industrial or operational nature) enhances the comparability of our ongoing operating performance relative to our peers in the pharmaceutical industry that carry those intangible assets (principally patents and trademarks) at low book values either because they are the result ofin-house research and development that has already been expensed in prior periods or because they were acquired through business combinations that were accounted for aspoolings-of-interest; |
| • | | the elimination of selected items – such as the incremental cost of sales arising from the workdown of acquired inventories remeasured at fair value in business combinations, major gains and losses on disposals, and costs and provisions associated with major litigation and any other majornon-recurring items – improves comparability from one period to the next; and |
| • | | the elimination of restructuring costs and similar items enhances comparability because those costs are incurred in connection with reorganization and transformation processes intended to optimize our operations. |
We remind investors, however, that business net income should not be considered in isolation from, or as a substitute for,Net income attributable to equity holders of Sanofi reported in accordance with IFRS. In addition, we strongly encourage investors and potential investors not to rely on any single financial measure but to review our financial statements, including the notes thereto, carefully and in their entirety.
We compensate for the material limitations described above by using business net income only to supplement our IFRS financial reporting and by ensuring that our disclosures provide sufficient information for a full understanding of all adjustments included in business net income.
Because our business net income is not a standardized measure, it may not be directly comparable with thenon-GAAP financial measures of other companies using the same or a similarnon-GAAP financial measure.
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 44
C.3. NET SALES
Net sales for the first half of 2019 amounted to €17,019 million, 5.9% higher than in the first half of 2018. Exchange rate fluctuations had a positive effect of 1.8 percentage points overall, due mainly to favorable trends in the euro exchange rate against the US dollar and Japanese yen, partly offset by negative effects from the Argentinean peso and Turkish lira. At constant exchange rates (CER, see definition below), net sales rose by 4.1%, mainly reflecting good performances for Dupixent®, the Rare Diseases franchise and the Vaccines segment, and more generally sales growth in emerging markets. Those effects were attenuated by lower sales for the Diabetes franchise in the United States and for Established Prescription Products, in particular following the divestment of our European generics business (Zentiva) in 2018. At constant exchange rates and on a constant structure basis (CER/CS, see definition below), net sales were up 4.8%.
Reconciliation of net sales to net sales at constant exchange rates and on a constant structure basis
| | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | Change | |
Net sales | | | 17,019 | | | | 16,074 | | | | +5.9 | % |
Effect of exchange rates | | | (289 | ) | | | | | | | | |
Net sales at constant exchange rates | | | 16,730 | | | | 16,074 | | | | +4.1 | % |
Impact of change in structure - Zentiva(a) and Bioverativ(b) | | | | | | | (112 | ) | | | | |
Net sales at constant exchange rates and on a constant structure basis | | | 16,730 | | | | 15,962 | | | | +4.8 | % |
| (a) | Elimination of the €312 million of net sales generated from January 1 through June 30, 2018 by Zentiva, our European generics business, divested on September 30, 2018. |
| (b) | Add-back of the €200 million of net sales generated from January 1 through March 7, 2018 by Bioverativ, consolidated from March 8, 2018 onwards. |
When we refer to changes in our net sales at constant exchange rates (CER), that means that we have excluded the effect of exchange rates by recalculating net sales for the relevant period using the exchange rates that were used for the previous period.
When we refer to changes in our net sales on a constant structure (CS) basis, that means that we eliminate the effect of changes in structure by restating the net sales for the previous period as follows:
| • | | by including sales generated by entities or product rights acquired in the current period for a portion of the previous period equal to the portion of the current period during which we owned them, based on historical sales information we receive from the party from whom we make the acquisition; |
| • | | similarly, by excluding sales for a portion of the previous period when we have sold an entity or rights to a product in the current period; and |
| • | | for a change in consolidation method, by recalculating the previous period on the basis of the method used for the current period. |
To facilitate analysis and comparisons with prior periods, some figures are given at constant exchange rates and on a constant structure basis (CER/CS).
45 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.3.1. NET SALES BY GLOBAL BUSINESS UNIT (GBU)
Our net sales comprise the net sales generated by our Pharmaceuticals, Consumer Healthcare and Vaccines segments.
The table below also presents net sales for our Global Business Units (GBUs). Note that emerging markets sales of Specialty Care and General Medicines products are included in the China & Emerging Markets GBU. This reflects Sanofi’s decision to change the organizational structure of two of our GBUs effective January 1, 2019 so as to refocus our activities on mature markets and emerging markets. This involved creating a new General Medicines GBU which combines the product portfolio of the Diabetes & Cardiovascular GBU with the Established Prescription Products portfolio of the General Medicines and Emerging Markets GBU. The new General Medicines GBU will focus exclusively on mature markets. We have also created a second new GBU, China and Emerging Markets. This new GBU will concentrate on the distinctive characteristics and growth potential of emerging markets and especially China, our second biggest market after the United States.
| | | | | | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | Change on a reported basis | | | Change at constant exchange rates | |
Sanofi Genzyme (Specialty Care) (a)(b) | | | 4,311 | | | | 3,268 | | | | +31.9 | % | | | +25.9 | % |
General Medicines (a) | | | 4,566 | | | | 5,309 | | | | -14.0 | % | | | -16.3 | % |
China & Emerging Markets (c)(d) | | | 3,849 | | | | 3,622 | | | | +6.3 | % | | | +8.7 | % |
Total Pharmaceuticals | | | 12,726 | | | | 12,199 | | | | +4.3 | % | | | +2.4 | % |
Consumer Healthcare | | | 2,399 | | | | 2,353 | | | | +2.0 | % | | | +0.8 | % |
Sanofi Pasteur (Vaccines) | | | 1,894 | | | | 1,522 | | | | +24.4 | % | | | +22.5 | % |
Total net sales | | | 17,019 | | | | 16,074 | | | | +5.9 | % | | | +4.1 | % |
| (a) | Does not include Emerging Markets net sales. |
| (b) | Rare Diseases, Multiple Sclerosis, Oncology, Immunology, and Rare Blood Disorder. |
| (c) | Includes net sales in Emerging Markets of Specialty Care and General Medicines products. |
| (d) | Emerging markets: World excluding United States, Canada, Europe (apart from Eurasia: Russia, Ukraine, Georgia, Belarus, Armenia and Turkey), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
C.3.2. NET SALES BY FRANCHISE, GEOGRAPHICAL REGION AND PRODUCT
The table below sets forth our 2019 and 2018 first-half net sales by franchise, geographical region and product in order to facilitate direct comparisons with our peers. It also provides a reconciliation of sales by GBU for our Pharmaceuticals segment. Net sales for the Specialty Care GBU are obtained by aggregating sales of Specialty Care products in Europe, the United States and the Rest of the World region. Net sales for the General Medicines GBU are obtained by aggregating sales of General Medicines products in Europe, the United States and the Rest of the World region. Net sales for the China & Emerging Markets GBU are obtained by aggregating sales of all our pharmaceutical products in emerging markets.
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 46
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Europe (a) | | | United States | | | Rest of the world (b)
| | | Emerging Markets (c)
| | | Total Franchise | |
| | | June 30, | | | Change | | | June 30, | | | Change | | | June 30, | | | Change | | | June 30, | | | Change | | | June 30, | | | Change | |
| (€ million) | | 2019 | | | 2018 | | | CER | | | 2019 | | | 2018 | | | CER | | | 2019 | | | 2018 | | | CER | | | 2019 | | | 2018 | | | CER | | | 2019 | | | 2018 | | | Reported | | | CER | |
Aubagio | | | 203 | | | | 184 | | | | +10.3 | % | | | 645 | | | | 541 | | | | +11.3 | % | | | 29 | | | | 25 | | | | +16.0 | % | | | 26 | | | | 25 | | | | +12.0 | % | | | 903 | | | | 775 | | | | +16.5 | % | | | +11.2 | % |
Lemtrada | | | 63 | | | | 92 | | | | -31.5 | % | | | 83 | | | | 93 | | | | -17.2 | % | | | 6 | | | | 10 | | | | -40.0 | % | | | 14 | | | | 12 | | | | +33.3 | % | | | 166 | | | | 207 | | | | -19.8 | % | | | -21.7 | % |
Total Multiple Sclerosis | | | 266 | | | | 276 | | | | -3.6 | % | | | 728 | | | | 634 | | | | +7.1 | % | | | 35 | | | | 35 | | | | +0.0 | % | | | 40 | | | | 37 | | | | +18.9 | % | | | 1,069 | | | | 982 | | | | +8.9 | % | | | +4.3 | % |
Cerezyme | | | 123 | | | | 134 | | | | -8.2 | % | | | 88 | | | | 83 | | | | -1.2 | % | | | 17 | | | | 19 | | | | -15.8 | % | | | 135 | | | | 120 | | | | +30.0 | % | | | 363 | | | | 356 | | | | +2.0 | % | | | +5.9 | % |
Cerdelga | | | 34 | | | | 22 | | | | +54.5 | % | | | 57 | | | | 47 | | | | +12.8 | % | | | 5 | | | | 4 | | | | +25.0 | % | | | 2 | | | | 1 | | | | +200.0 | % | | | 98 | | | | 74 | | | | +32.4 | % | | | +28.4 | % |
Myozyme | | | 192 | | | | 188 | | | | +2.1 | % | | | 162 | | | | 133 | | | | +13.5 | % | | | 29 | | | | 28 | | | | +3.6 | % | | | 71 | | | | 56 | | | | +37.5 | % | | | 454 | | | | 405 | | | | +12.1 | % | | | +10.9 | % |
Fabrazyme | | | 90 | | | | 87 | | | | +3.4 | % | | | 199 | | | | 179 | | | | +3.9 | % | | | 60 | | | | 54 | | | | +7.4 | % | | | 47 | | | | 38 | | | | +36.8 | % | | | 396 | | | | 358 | | | | +10.6 | % | | | +7.8 | % |
Aldurazyme | | | 39 | | | | 38 | | | | +2.6 | % | | | 26 | | | | 21 | | | | +14.3 | % | | | 13 | | | | 12 | | | | +8.3 | % | | | 43 | | | | 32 | | | | +43.8 | % | | | 121 | | | | 103 | | | | +17.5 | % | | | +18.4 | % |
Other | | | 33 | | | | 33 | | | | +0.0 | % | | | 44 | | | | 46 | | | | -8.7 | % | | | 42 | | | | 42 | | | | -4.8 | % | | | 25 | | | | 21 | | | | +23.8 | % | | | 144 | | | | 142 | | | | +1.4 | % | | | -0.7 | % |
Total Rare Diseases | | | 511 | | | | 502 | | | | +1.8 | % | | | 576 | | | | 509 | | | | +5.7 | % | | | 166 | | | | 159 | | | | +1.3 | % | | | 323 | | | | 268 | | | | +34.3 | % | | | 1,576 | | | | 1,438 | | | | +9.6 | % | | | +9.2 | % |
Jevtana | | | 86 | | | | 78 | | | | +10.3 | % | | | 101 | | | | 84 | | | | +11.9 | % | | | 36 | | | | 29 | | | | +20.7 | % | | | 14 | | | | 11 | | | | +27.3 | % | | | 237 | | | | 202 | | | | +17.3 | % | | | +13.4 | % |
Thymoglobulin | | | 18 | | | | 19 | | | | -5.3 | % | | | 95 | | | | 78 | | | | +14.1 | % | | | 12 | | | | 10 | | | | +10.0 | % | | | 50 | | | | 37 | | | | +37.8 | % | | | 175 | | | | 144 | | | | +21.5 | % | | | +17.4 | % |
Eloxatin | | | 1 | | | | 1 | | | | +0.0 | % | | | -4 | | | | 0 | | | | — | | | | 13 | | | | 15 | | | | -13.3 | % | | | 99 | | | | 74 | | | | +32.4 | % | | | 109 | | | | 90 | | | | +21.1 | % | | | +20.0 | % |
Mozobil | | | 24 | | | | 24 | | | | +0.0 | % | | | 54 | | | | 45 | | | | +11.1 | % | | | 9 | | | | 8 | | | | +12.5 | % | | | 6 | | | | 5 | | | | +40.0 | % | | | 93 | | | | 82 | | | | +13.4 | % | | | +9.8 | % |
Taxotere | | | 2 | | | | 2 | | | | +0.0 | % | | | -1 | | | | 1 | | | | -200.0 | % | | | 14 | | | | 14 | | | | -7.1 | % | | | 74 | | | | 67 | | | | +9.0 | % | | | 89 | | | | 84 | | | | +6.0 | % | | | +3.6 | % |
Other | | | 50 | | | | 52 | | | | -3.8 | % | | | 42 | | | | 40 | | | | +0.0 | % | | | 23 | | | | 20 | | | | +15.0 | % | | | 12 | | | | 13 | | | | -15.4 | % | | | 127 | | | | 125 | | | | +1.6 | % | | | -0.8 | % |
Total Oncology | | | 181 | | | | 176 | | | | +2.8 | % | | | 287 | | | | 248 | | | | +8.1 | % | | | 107 | | | | 96 | | | | +8.3 | % | | | 255 | | | | 207 | | | | +22.7 | % | | | 830 | | | | 727 | | | | +14.2 | % | | | +11.0 | % |
Dupixent | | | 82 | | | | 26 | | | | +215.4 | % | | | 669 | | | | 246 | | | | +154.5 | % | | | 65 | | | | 10 | | | | +520.0 | % | | | 9 | | | | 1 | | | | +800.0 | % | | | 825 | | | | 283 | | | | +191.5 | % | | | +175.3 | % |
Kevzara | | | 18 | | | | 5 | | | | +260.0 | % | | | 48 | | | | 23 | | | | +95.7 | % | | | 15 | | | | 2 | | | | +600.0 | % | | | 1 | | | | 0 | | | | — | | | | 82 | | | | 30 | | | | +173.3 | % | | | +160.0 | % |
Total Immunology | | | 100 | | | | 31 | | | | +222.6 | % | | | 717 | | | | 269 | | | | +149.4 | % | | | 80 | | | | 12 | | | | +533.3 | % | | | 10 | | | | 1 | | | | +900.0 | % | | | 907 | | | | 313 | | | | +189.8 | % | | | +173.8 | % |
Eloctate | | | 0 | | | | 0 | | | | — | | | | 272 | | | | 187 | | | | +35.8 | % | | | 65 | | | | 32 | | | | +90.6 | % | | | 8 | | | | 0 | | | | — | | | | 345 | | | | 219 | | | | +57.5 | % | | | +47.5 | % |
Alprolix | | | 0 | | | | 0 | | | | — | | | | 144 | | | | 83 | | | | +62.7 | % | | | 56 | | | | 19 | | | | +178.9 | % | | | 0 | | | | 0 | | | | — | | | | 200 | | | | 102 | | | | +96.1 | % | | | +84.3 | % |
Cablivi | | | 9 | | | | 0 | | | | — | | | | 11 | | | | 0 | | | | — | | | | 0 | | | | 0 | | | | — | | | | 0 | | | | 0 | | | | — | | | | 20 | | | | 0 | | | | — | | | | — | |
Total Rare Blood Disorder | | | 9 | | | | 0 | | | | — | | | | 427 | | | | 270 | | | | +47.8 | % | | | 121 | | | | 51 | | | | +125.5 | % | | | 8 | | | | 0 | | | | — | | | | 565 | | | | 321 | | | | +76.0 | % | | | +65.4 | % |
Total Specialty Care | | | 1,067 | | | | 985 | | | | +8.3 | % | | | 2,735 | | | | 1,930 | | | | +32.4 | % | | | 509 | | | | 353 | | | | +39.1 | % | | | 636 | | | | 513 | | | | +31.8 | % | | | 4,947 | | | | 3,781 | | | | +30.8 | % | | | +26.7 | % |
Lantus | | | 298 | | | | 355 | | | | -16.1 | % | | | 568 | | | | 816 | | | | -35.2 | % | | | 113 | | | | 139 | | | | -20.9 | % | | | 553 | | | | 492 | | | | +14.6 | % | | | 1,532 | | | | 1,802 | | | | -15.0 | % | | | -16.7 | % |
Toujeo | | | 163 | | | | 142 | | | | +14.8 | % | | | 139 | | | | 171 | | | | -24.0 | % | | | 40 | | | | 36 | | | | +5.6 | % | | | 89 | | | | 65 | | | | +41.5 | % | | | 431 | | | | 414 | | | | +4.1 | % | | | +2.2 | % |
Apidra | | | 66 | | | | 70 | | | | -5.7 | % | | | 25 | | | | 40 | | | | -42.5 | % | | | 18 | | | | 19 | | | | -5.3 | % | | | 64 | | | | 54 | | | | +24.1 | % | | | 173 | | | | 183 | | | | -5.5 | % | | | -4.9 | % |
Amaryl | | | 8 | | | | 8 | | | | +0.0 | % | | | 1 | | | | 1 | | | | +0.0 | % | | | 13 | | | | 15 | | | | -13.3 | % | | | 149 | | | | 146 | | | | +1.4 | % | | | 171 | | | | 170 | | | | +0.6 | % | | | +0.0 | % |
Admelog | | | 7 | | | | 2 | | | | +250.0 | % | | | 136 | | | | 7 | | | | — | | | | 0 | | | | 1 | | | | -100.0 | % | | | 0 | | | | 0 | | | | — | | | | 143 | | | | 10 | | | | — | | | | — | |
Other | | | 66 | | | | 71 | | | | -7.0 | % | | | 37 | | | | 24 | | | | +45.8 | % | | | 16 | | | | 12 | | | | +33.3 | % | | | 15 | | | | 36 | | | | -58.3 | % | | | 134 | | | | 143 | | | | -6.3 | % | | | -7.7 | % |
Total Diabetes | | | 608 | | | | 648 | | | | -6.2 | % | | | 906 | | | | 1,059 | | | | -20.2 | % | | | 200 | | | | 222 | | | | -12.2 | % | | | 870 | | | | 793 | | | | +11.7 | % | | | 2,584 | | | | 2,722 | | | | -5.1 | % | | | -6.9 | % |
Praluent | | | 61 | | | | 41 | | | | +46.3 | % | | | 44 | | | | 61 | | | | -32.8 | % | | | 8 | | | | 5 | | | | +60.0 | % | | | 9 | | | | 4 | | | | +125.0 | % | | | 122 | | | | 111 | | | | +9.9 | % | | | +6.3 | % |
Multaq | | | 20 | | | | 21 | | | | -4.8 | % | | | 135 | | | | 135 | | | | -6.7 | % | | | 2 | | | | 3 | | | | -33.3 | % | | | 4 | | | | 3 | | | | +33.3 | % | | | 161 | | | | 162 | | | | -0.6 | % | | | -6.2 | % |
Total Cardiovascular | | | 81 | | | | 62 | | | | +29.0 | % | | | 179 | | | | 196 | | | | -14.8 | % | | | 10 | | | | 8 | | | | +25.0 | % | | | 13 | | | | 7 | | | | +85.7 | % | | | 283 | | | | 273 | | | | +3.7 | % | | | -1.1 | % |
Plavix | | | 69 | | | | 76 | | | | -9.2 | % | | | 0 | | | | 0 | | | | — | | | | 100 | | | | 110 | | | | -13.6 | % | | | 597 | | | | 575 | | | | +3.3 | % | | | 766 | | | | 761 | | | | +0.7 | % | | | -0.4 | % |
Lovenox | | | 375 | | | | 471 | | | | -20.4 | % | | | 18 | | | | 20 | | | | -15.0 | % | | | 36 | | | | 41 | | | | -12.2 | % | | | 261 | | | | 236 | | | | +11.9 | % | | | 690 | | | | 768 | | | | -10.2 | % | | | -9.9 | % |
Aprovel | | | 54 | | | | 55 | | | | -1.8 | % | | | 14 | | | | 5 | | | | +160.0 | % | | | 39 | | | | 41 | | | | -4.9 | % | | | 267 | | | | 242 | | | | +9.1 | % | | | 374 | | | | 343 | | | | +9.0 | % | | | +7.9 | % |
Depakine | | | 80 | | | | 84 | | | | -4.8 | % | | | 0 | | | | 0 | | | | — | | | | 7 | | | | 7 | | | | +0.0 | % | | | 149 | | | | 139 | | | | +7.2 | % | | | 236 | | | | 230 | | | | +2.6 | % | | | +2.6 | % |
Synvisc / Synvisc one | | | 14 | | | | 13 | | | | +7.7 | % | | | 103 | | | | 111 | | | | -12.6 | % | | | 7 | | | | 7 | | | | -14.3 | % | | | 31 | | | | 29 | | | | +3.4 | % | | | 155 | | | | 160 | | | | -3.1 | % | | | -8.1 | % |
Renagel / Renvela | | | 26 | | | | 32 | | | | -18.8 | % | | | 59 | | | | 121 | | | | -54.5 | % | | | 16 | | | | 15 | | | | +6.7 | % | | | 44 | | | | 33 | | | | +30.3 | % | | | 145 | | | | 201 | | | | -27.9 | % | | | -30.3 | % |
Tritace | | | 71 | | | | 73 | | | | -1.4 | % | | | 0 | | | | 0 | | | | — | | | | 3 | | | | 2 | | | | +0.0 | % | | | 35 | | | | 40 | | | | -10.0 | % | | | 109 | | | | 115 | | | | -5.2 | % | | | -4.3 | % |
Stilnox | | | 17 | | | | 20 | | | | -15.0 | % | | | 18 | | | | 22 | | | | -22.7 | % | | | 41 | | | | 42 | | | | -9.5 | % | | | 31 | | | | 32 | | | | -3.1 | % | | | 107 | | | | 116 | | | | -7.8 | % | | | -11.2 | % |
Allegra | | | 6 | | | | 5 | | | | +20.0 | % | | | 0 | | | | 0 | | | | — | | | | 76 | | | | 75 | | | | -4.0 | % | | | 0 | | | | 0 | | | | — | | | | 82 | | | | 80 | | | | +2.5 | % | | | -2.5 | % |
Generics | | | 61 | | | | 367 | | | | -83.4 | % | | | 79 | | | | 48 | | | | +54.2 | % | | | 74 | | | | 71 | | | | +0.0 | % | | | 322 | | | | 351 | | | | -3.1 | % | | | 536 | | | | 837 | | | | -36.0 | % | | | -34.8 | % |
Other established prescription products | | | 835 | | | | 890 | | | | -5.7 | % | | | 93 | | | | 100 | | | | -16.0 | % | | | 191 | | | | 190 | | | | -2.1 | % | | | 593 | | | | 632 | | | | -3.5 | % | | | 1,712 | | | | 1,812 | | | | -5.5 | % | | | -5.1 | % |
Total Established Prescription Products | | | 1,608 | | | | 2,086 | | | | -22.7 | % | | | 384 | | | | 427 | | | | -16.4 | % | | | 590 | | | | 601 | | | | -5.5 | % | | | 2,330 | | | | 2,309 | | | | +2.3 | % | | | 4,912 | | | | 5,423 | | | | -9.4 | % | | | -9.7 | % |
Total General Medicines | | | 2,297 | | | | 2,796 | | | | -17.7 | % | | | 1,469 | | | | 1,682 | | | | -18.6 | % | | | 800 | | | | 831 | | | | -7.0 | % | | | 3,213 | | | | 3,109 | | | | +4.9 | % | | | 7,779 | | | | 8,418 | | | | -7.6 | % | | | -8.5 | % |
Total China and Emerging Markets | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 3,849 | | | | 3,622 | | | | +8.7 | % | | | | | | | | | | | | | | | | |
Total Pharmaceuticals | | | 3,364 | | | | 3,781 | | | | -10.9 | % | | | 4,204 | | | | 3,612 | | | | +8.6 | % | | | 1,309 | | | | 1,184 | | | | +6.8 | % | | | 3,849 | | | | 3,622 | | | | +8.7 | % | | | 12,726 | | | | 12,199 | | | | +4.3 | % | | | +2.4 | % |
Allergy, Cough & Cold | | | 163 | | | | 167 | | | | -2.4 | % | | | 187 | | | | 173 | | | | +0.6 | % | | | 88 | | | | 78 | | | | +9.0 | % | | | 173 | | | | 162 | | | | +7.4 | % | | | 611 | | | | 580 | | | | +5.3 | % | | | +2.8 | % |
Pain | | | 254 | | | | 254 | | | | +0.4 | % | | | 93 | | | | 78 | | | | +11.5 | % | | | 63 | | | | 57 | | | | +3.5 | % | | | 220 | | | | 239 | | | | +0.4 | % | | | 630 | | | | 628 | | | | +0.3 | % | | | +2.1 | % |
Digestive | | | 167 | | | | 162 | | | | +3.1 | % | | | 103 | | | | 95 | | | | +1.1 | % | | | 28 | | | | 28 | | | | +0.0 | % | | | 250 | | | | 211 | | | | +18.5 | % | | | 548 | | | | 496 | | | | +10.5 | % | | | +9.1 | % |
Nutritionals | | | 62 | | | | 62 | | | | +1.6 | % | | | 19 | | | | 18 | | | | +0.0 | % | | | 122 | | | | 123 | | | | -3.3 | % | | | 111 | | | | 127 | | | | -11.8 | % | | | 314 | | | | 330 | | | | -4.8 | % | | | -5.5 | % |
Other | | | 34 | | | | 61 | | | | -44.3 | % | | | 186 | | | | 177 | | | | -1.7 | % | | | 17 | | | | 19 | | | | -15.8 | % | | | 59 | | | | 62 | | | | -4.8 | % | | | 296 | | | | 319 | | | | -7.2 | % | | | -11.3 | % |
Total Consumer Healthcare | | | 680 | | | | 706 | | | | -3.4 | % | | | 588 | | | | 541 | | | | +1.5 | % | | | 318 | | | | 305 | | | | +0.7 | % | | | 813 | | | | 801 | | | | +4.2 | % | | | 2,399 | | | | 2,353 | | | | +2.0 | % | | | +0.8 | % |
Polio / Pertussis / Hib Vaccines | | | 151 | | | | 139 | | | | +7.9 | % | | | 192 | | | | 176 | | | | +1.7 | % | | | 110 | | | | 81 | | | | +29.6 | % | | | 535 | | | | 338 | | | | +61.5 | % | | | 988 | | | | 734 | | | | +34.6 | % | | | +33.5 | % |
Travel and Other Endemics Vaccines | | | 67 | | | | 59 | | | | +13.6 | % | | | 74 | | | | 62 | | | | +12.9 | % | | | 30 | | | | 28 | | | | +7.1 | % | | | 86 | | | | 79 | | | | +7.6 | % | | | 257 | | | | 228 | | | | +12.7 | % | | | +10.5 | % |
Meningitis/Pneumonia Vaccines | | | 0 | | | | 0 | | | | — | | | | 175 | | | | 157 | | | | +4.5 | % | | | 7 | | | | 7 | | | | +0.0 | % | | | 66 | | | | 41 | | | | +68.3 | % | | | 248 | | | | 205 | | | | +21.0 | % | | | +17.1 | % |
Adult Booster Vaccines | | | 85 | | | | 66 | | | | +28.8 | % | | | 124 | | | | 97 | | | | +19.6 | % | | | 13 | | | | 13 | | | | +7.7 | % | | | 12 | | | | 10 | | | | +20.0 | % | | | 234 | | | | 186 | | | | +25.8 | % | | | +22.0 | % |
Influenza Vaccines | | | 2 | | | | 1 | | | | +100.0 | % | | | 4 | | | | 4 | | | | -25.0 | % | | | 20 | | | | 24 | | | | -12.5 | % | | | 91 | | | | 98 | | | | -4.1 | % | | | 117 | | | | 127 | | | | -7.9 | % | | | -5.5 | % |
Other | | | 2 | | | | 6 | | | | -66.7 | % | | | 40 | | | | 28 | | | | +35.7 | % | | | 6 | | | | 5 | | | | +40.0 | % | | | 2 | | | | 3 | | | | +0.0 | % | | | 50 | | | | 42 | | | | +19.0 | % | | | +9.5 | % |
Total Vaccines | | | 307 | | | | 271 | | | | +12.9 | % | | | 609 | | | | 524 | | | | +8.8 | % | | | 186 | | | | 158 | | | | +13.9 | % | | | 792 | | | | 569 | | | | +42.2 | % | | | 1,894 | | | | 1,522 | | | | +24.4 | % | | | +22.5 | % |
Total Sanofi | | | 4,351 | | | | 4,758 | | | | -8.4 | % | | | 5,401 | | | | 4,677 | | | | +7.8 | % | | | 1,813 | | | | 1,647 | | | | +6.3 | % | | | 5,454 | | | | 4,992 | | | | +11.8 | % | | | 17,019 | | | | 16,074 | | | | +5.9 | % | | | +4.1 | % |
| (a) | Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| (b) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
| (c) | World excluding United States, Canada, Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
47 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.3.3. PHARMACEUTICALS SEGMENT
Net sales of thePharmaceuticalssegment were €12,726 million in the first half of 2019, up 4.3% on a reported basis and 2.4% at constant exchange rates. At constant exchange rates and on a constant structure basis, net sales rose by 3.4%.
Theyear-on-year increase of €527 million reflects favorable exchange rate effects of €234 million and a net negative effect of €112 million from the divestment of our European Generics business (Zentiva) and the acquisition of Bioverativ’s products. It also reflects the following effects at constant exchange rates:
| | • | | positive performances from the Immunology franchise (+€544 million), the Rare Diseases franchise (+€132 million), the Oncology franchise (+€80 million), the Multiple Sclerosis franchise (+€42 million), and the Rare Blood Disorder franchise on a constant structure basis (+€9 million); and |
| | • | | lower net sales for the Diabetes franchise (-€188 million), the Cardiovascular franchise (-€3 million), and the Established Prescription Products franchise on a constant structure basis (-€211 million). |
Comments on the performances of our major Pharmaceuticals segment products are provided below.
RARE DISEASES FRANCHISE
Net sales for theRare Diseases franchise amounted to €1,576 million in the first half of 2019, up 9.6% on a reported basis and 9.2% at constant exchange rates. Growth was driven by medicines indicated for the treatment of Pompe disease (Myozyme®/Lumizyme®), Gaucher disease (Cerezyme® and Cerdelga®) and Fabry disease (Fabrazyme®), especially in the Emerging Markets region1. The franchise grew sales by 5.7% CER (to €576 million) in the United States and by 1.8% CER (to €511 million) in Europe2 over the period. Emerging Markets sales were 34.3% higher CER at €323 million, boosted by a favorable delivery schedule.
In the first half of 2019, net sales for theGaucher disease franchise(Cerezyme®and Cerdelga®) reached €461 million, up 9.8% CER, on stronger sales of Cerezyme® in Emerging Markets (+30.0% CER at €135 million) and of Cerdelga® in Europe (+54.5% CER at €34 million). Overall, sales of Cerezyme® rose by 5.9% CER to €363 million during the period, and sales of Cerdelga® by 28.4% CER to €98 million.
Net sales ofMyozyme® / Lumizyme® in Pompe disease rose by 10.9% CER in the first half of 2019 to €454 million, driven by sales growth in Emerging Markets (+37.5% CER, at €71 million) and in the United States (+13.5% CER, at €162 million). This growth reflects the rising number of patients diagnosed with, and treated for, Pompe disease.
Fabrazyme® posted net sales growth of 7.8% CER to €396 million. Sales are advancing across all territories due to the rising number of patients diagnosed with, and treated for, Fabry disease. The product increased its sales by 36.8% CER in Emerging Markets (to €47 million) and by 3.9% CER in the United States (to €199 million).
MULTIPLE SCLEROSIS FRANCHISE
In the first half of 2019, theMultiple Sclerosisfranchise generated net sales of €1,069 million, up 8.9% on a reported basis and 4.3% CER, as strong growth in sales of Aubagio® more than offset lower sales of Lemtrada® in mature markets.
Aubagio® achieved net sales of €903 million in the first half of 2019, up 11.2% CER, supported by growth in the United States (+11.3% CER, at €645 million) and Europe (+10.3% CER, at €203 million). Sales also rose in the Rest of the World region3 (+16.0% CER, at €29 million) and in Emerging Markets (+12.0% CER, at €26 million).
2019 first-half net sales ofLemtrada® were €166 million, down 21.7% CER on lower sales in Europe(-31.5% CER, at €63 million), the United States(-17.2% CER, at €83 million), and the Rest of the World region(-40.0% CER, at €6 million). The downtrend in sales is mainly due to tougher competition, and to an update to the Summary of Product Characteristics in the European Union.
IMMUNOLOGY FRANCHISE
Dupixent® (collaboration with Regeneron) generated net sales of €825 million in the first half of 2019, an increase of 175.3% CER. In the United States, where Dupixent® is approved for the treatment of atopic dermatitis in adults and adolescents, asthma and (since the end of June 2019) nasal polyposis, the product posted sales of €669 million over the period (+154.5%). In Europe, where Dupixent® is approved for the treatment of atopic dermatitis in adults and asthma, 2019 first-half sales reached €82 million. The product recorded first-half sales of €65 million in the Rest of the World region and €9 million in Emerging Markets. By the end of the first half, Dupixent® had been launched in 28 countries, with a further 11 launches scheduled before the end of the year.
Sales of Kevzara® (collaboration with Regeneron) totaled €82 million in the first half of 2019 (versus €30 million in the first half of 2018), including €48 million in the United States (versus €23 million in the first half of 2018).
| 1 | World excluding United States, Canada, Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
| 2 | Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| 3 | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 48
ONCOLOGY FRANCHISE
In the first half of 2019, net sales for theOncology franchise amounted to €830 million, up 14.2% on a reported basis and 11.0% CER. This reflects good performances by Jevtana® across all geographies, by Thymoglobulin® in the United States and Emerging Markets, and by the whole franchise in China.
Jevtana® reported net sales of €237 million in the first half of 2019, up 13.4% CER, as sales grew across all geographies and especially in the United States (+11.9% CER, at €101 million) and Europe (+10.3% CER, at €86 million).
Sales ofThymoglobulin® reached €175 million (+17.4% CER), driven by the United States (+14.1% CER at €95 million) and Emerging Markets (+37.8% CER at €50 million).
Eloxatin® posted sales of €109 million in the first half of 2019 (+20.0% CER), mainly on strong growth in China (+39.0% CER, at €83 million).
Libtayo® (cemiplimab, collaboration with Regeneron) was approved in the United States in September 2018 for the treatment of patients with metastatic or locally advanced cutaneous squamous cell carcinoma (CSCC) who are not candidates for surgery or curative radiotherapy. Sales of this product in the United States are included in the consolidated sales of Regeneron under the terms of our alliance agreement with Regeneron (see Note C.2 to our consolidated financial statements for the year ended December 31, 2018, on pageF-34 of our Annual Report on Form20-F; this document is available on our corporate website, www.sanofi.com). Libtayo® was approved in Brazil in March 2019, and in Canada in April 2019. Libtayo® also obtained conditional marketing approval in Europe at the end of June 2019.
RARE BLOOD DISORDER FRANCHISE
TheRare Blood Disorder franchise was formed in 2018 following two acquisitions. Firstly, the acquisition of Bioverativ added two products to our portfolio: Eloctate® and Alprolix®, reference treatments for hemophilia. Secondly, the acquisition of Ablynx brought Cablivi® (caplacizumab) into our portfolio; this product was granted marketing authorization by the European Commission in September 2018 for the treatment of acquired thrombotic thrombocytopenic purpura (aTTP).
Sales of the Rare Blood Disorder franchise (consolidated by Sanofi from March 9, 2018 onwards) amounted to €565 million in the first half of 2019; of that total, €138 million was generated outside the United States, half of it in Japan. At constant exchange rates and on a constant structure basis, the franchise grew sales by 1.7%.
Sales ofEloctate® reached €345 million in the first half of 2019. At constant exchange rates and on a constant structure basis, sales of Eloctate® decreased by 7.7%. Sales of the product in the United States amounted to €272 million, 12.4% lower CER/CS, reflecting the competitive environment. In the Rest of the World region, sales of Eloctate® rose by 1.7% CER/CS to €65 million, with growth in Japan (+10.3% CER/CS at €45 million) offsetting lower sales in Canada following the failure of a tender bid (as previously announced).
Sales ofAlprolix®, indicated for the treatment of hemophilia B, increased 9.3% CER/CS to €200 million; of this, €144 million was generated in the United States (+4.7% CER/CS) and €56 million in the Rest of the World Region (+23.3% CER/CS, driven largely by the product’s launch in Australia).
Cablivi® generated net sales of €20 million in the first half of 2019. This comprised €9 million in Europe (where the product is on sale in Germany, Denmark and Austria), and €11 million in the United States (where the product has been on sale since the start of April 2019).
DIABETES FRANCHISE
Net sales for theDiabetes franchise amounted to €2,584 million in the first half of 2019, down 5.1% on a reported basis and 6.9% at constant exchange rates. This reflects a decrease in sales for the franchise in the United States(-20.2% CER, at €906 million), especially of the insulin glargines Lantus® and Toujeo®. That decrease reflects changes to the Medicare Part D welfare program and the continuing decline in average net prices of insulin glargines in the United States. Elsewhere in the world, net sales for the Diabetes franchise also decreased in Europe(-6.2% CER, at €608 million) and in the Rest of the World region(-12.2% CER, at €200 million). Conversely, in Emerging Markets the franchise grew sales by 11.7% CER to €870 million.
Net sales of Lantus® decreased by 16.7% CER in the first half to €1,532 million. In the United States, sales were down 35.2% CER at €568 million, for the reasons explained above. Net sales of Lantus® in Europe decreased by 16.1% CER to €298 million, due to competition from a branded product and a biosimilar of Lantus® and to the switching of patients to Toujeo®. In Emerging Markets, net sales of Lantus® rose by 14.6% in the first half to €553 million.
Toujeo® posted 2019 first-half net sales of €431 million, up 2.2% CER, driven by Emerging Markets (+41.5%, at €89 million) and Europe (+14.8%, at €163 million). However sales were lower in the United States(-24.0% CER, at €139 million), mainly as a result of a decrease in the average net selling price.
We expect a further decline in net selling prices for our insulin glargines throughout 2019 as further rebates are granted in the United States to maintain broad coverage by private insurers and Medicare.
49 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
In the first half of 2019, net sales ofApidra® were down 4.9% CER at €173 million. Growth in Emerging Markets (+24.1% at €64 million) was offset by lower sales in mature markets, especially the United States(-42.5% CER, at €25 million).
In the first half of 2019, net sales ofAmaryl® were stableyear-on-year at €171 million, of which €149 million was generated in Emerging Markets (+1.4% CER).
Admelog® (injectable insulin lispro 100 units/ml, in vials or thepre-filled SoloStar® pen) was launched in 2018 in the United States, and also as a biosimilar in some European countries under the nameInsulin lispro Sanofi®. The product generated net sales of €143 million in the first half of 2019, including €136 million in the United States where sales were driven by the product’s acceptance onto the Managed Medicaid program. US sales of Admelog® are expected to be lower in the second half of 2019 following a downward adjustment of 44% to the wholesale price effective July 1, 2019.
Sales of Soliqua® 100/33and Suliqua®(insulin glargine 100 units/ml and lixisenatide 33 mcg/ml injectable) reached €50 million in the first half of 2019 (versus €26 million in the first half of 2018). In February 2019, the FDA approved an extension to the usage of Soliqua® 100/33, which can now also be prescribed for adults with type 2 diabetes not controlled by oral anti-diabetics.
CARDIOVASCULAR FRANCHISE
Sales ofPraluent® (collaboration with Regeneron) in the first half of 2019 amounted to €122 million (+6.3% CER), driven mainly by growth in Europe (+46.3% CER at €61 million). Sales in the United States decreased by 32.8% CER to €44 million due to larger rebates. The decline in the average net selling price of Praluent® is expected to continue throughout 2019 as a result of negotiations conducted by Sanofi and Regeneron with payers during 2018 aimed at streamlining the reimbursement criteria, in order to improve patient access to the product in exchange for substantial price cuts.
In the first half of 2019, net sales ofMultaq® were €161 million. Sales held steadyyear-on-year on a reported basis(-0.6%), but were down 6.2% CER. Sales were generated primarily in the United States (€135 million) and Europe (€20 million).
ESTABLISHED PRESCRIPTION PRODUCTS
Net sales ofEstablishedPrescription Products in the first half of 2019 amounted to €4,912 million, down 9.4% on a reported basis, largely as a result of the divestment of our European Generics business (Zentiva). At constant exchange rates and on a constant structure basis (CER/CS), sales for the franchise were down 4.1%. Sales growth in Emerging Markets (+2.3% CER, at €2,330 million) failed to offset lower net sales in mature markets(-9.4% CER/CS, at €2,582 million). In Europe, the franchise posted net sales of €1,608 million(-9.0% CER/CS), largely due to competition from generics of Lovenox®. In the Rest of the World region, net sales were down 5.5% CER at €590 million, reflecting competition from generics of Plavix® in Japan. In the United States, net sales of Established Prescription Products were down 16.4% CER at €384 million, mainly as a result of competition from generics of Renvela®/Renagel® (sevelamer).
Net sales ofPlavix® held steady in the first half of 2019 at €766 million, of which €464 million (+4.8% CER) was generated in China. Growth in China offset lower sales in Japan(-20.3% CER at €67 million) as a result of competition from generics. Sales of Plavix® in the United States and Puerto Rico are handled by BMS under the terms of theSanofi-BMS alliance1.
Net sales ofAprovel® /Avapro® advanced by 7.9% CER in the first half of 2019 to €374 million, of which €267 million (+9.1% CER) was generated in Emerging Markets. In China, where the product grew sales by 11.5% CER to €176 million, implementation of the Volume Based Procurement program in key cities could lead to a deceleration in sales growth for Plavix® and Avapro®/Aprovel® over 2019 as a whole.
In the first half of 2019, net sales ofLovenox® totaled €690 million, down 9.9% CER; this reflects lower sales in Europe(-20.4% CER, at €375 million) due to competition from biosimilars in a number of countries. The effect was only partly offset by stronger sales in Emerging Markets (+11.9% CER, at €261 million).
Net sales ofRenvela® /Renagel® in the first half of 2019 were €145 million, down 30.3% CER, due to competition from generics in the United States(-54.5% CER, at €59 million).
Generics posted net sales of €536 million in the first half of 2019, down 34.8% CER; this reflects the divestment of Zentiva, our European generics business, at the end of the third quarter of 2018. At constant exchange rates and on a constant structure basis, generics sales rose by 4.2%, driven by the United States (+54.2% CER at €79 million). Emerging Markets sales of generics were down 3.1% CER at €322 million, due to lower sales in Africa and the Middle East.
C.3.4. CONSUMER HEALTHCARE SEGMENT
Net sales ofConsumer Healthcare products in the first half of 2019 amounted to €2,399 million, up 2.0% on a reported basis and 0.8% at constant exchange rates. Sales growth in Emerging Markets more than offset lower sales in Europe, affected by the divestments ofnon-strategic brands in 2018 and by tougher regulatory and quality standards. Those factors are also likely to affect our Consumer Healthcare performances over 2019 as a whole and in the first part of 2020.
| 1 | See Note C.2 to our consolidated financial statements for the year ended December 31, 2018, on page F-34 of our Annual Report on Form 20-F; this document is available on our corporate website,www.sanofi.com. |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 50
In Emerging Markets, Consumer Healthcare net sales reached €813 million in the first half of 2019 (+4.2% CER). The main growth drivers were the Digestive category (+18.5% CER, at €250 million, thanks largely to sales growth of 24.1% CER for Essentiale® in China), and the Allergy, Cough and Cold category (+7.4% CER, at €173 million).
In Europe, 2019 first-half Consumer Healthcare net sales decreased by 3.4% CER to €680 million. That mainly reflects the divestment ofnon-strategic brands in 2018, resulting in a drop of 44.3% (CER) in sales for the Other category to €34 million.
In the United States, sales of Consumer Healthcare products reached €588 million in the first half of 2019, a rise of 1.5% CER, driven largely by the Pain category (+11.5% CER at €93 million).
In the Rest of the World region, 2019 first-half net sales for the Consumer Healthcare segment were up 0.7% CER at €318 million, driven largely by sales in Japan (+3.8% CER at €172 million).
C.3.5. VACCINES SEGMENT
In the first half of 2019, the Vaccines segment reported net sales of €1,894 million, up 24.4% on a reported basis and 22.5% at constant exchange rates. This mainly reflects growth in sales of Polio/Pertussis/Hib vaccines in Emerging Markets and in Japan. In Emerging Markets, net sales for the Vaccines segment were up 42.2% CER at €792 million, propelled largely by Pentaxim® in China. In the United States, Vaccines sales rose by 8.8% CER to €609 million. In Europe, sales reached €307 million (+12.9% CER), driven mainly by the Adult Booster Vaccines franchise.
In the first half of 2019, net sales ofPolio/Pertussis/Hib vaccines (including Hexaxim®, Pentacel®, Pentaxim® and Imovax®) reached €988 million (+33.5% CER), boosted by strong growth in Emerging Markets (+61.5% CER at €535 million) and especially for Pentaxim® in China, as well as in Japan (+35.0% CER at €86 million).
Travel and Other Endemics vaccines posted a 10.5% rise CER to €257 million in the first half of 2019, driven by growth in sales of rabies vaccines in the United States and Europe.
Net sales ofMeningitis/Pneumonia vaccines(including Menactra®) reached €248 million in the first half of 2019 (+17.1% CER), mainly on further growth in the Middle East. In the United States, first-half net sales of Menactra® were up 4.5% CER at €175 million.
2019 first-half net sales ofAdult Booster Vaccines were €234 million (+22.0% CER), driven by strong performances for the franchise in Europe (+28.8% CER at €85 million) and for Adacel® in the United States (+19.6% at €124 million).
Net sales ofInfluenza vaccines were down 5.5% CER at €117 million.
51 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.3.6. NET SALES BY GEOGRAPHICAL REGION
| | | | | | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | Change on a reported basis | | | Change at constant exchange
rates | |
United States | | | 5,401 | | | | 4,677 | | | | +15.5 | % | | | +7.8 | % |
Emerging Markets(a) | | | 5,454 | | | | 4,992 | | | | +9.3 | % | | | +11.8 | % |
of which Asia | | | 2,338 | | | | 1,993 | | | | +17.3 | % | | | +15.7 | % |
of which Latin America | | | 1,305 | | | | 1,298 | | | | +0.5 | % | | | +8.5 | % |
of which Africa and Middle East | | | 1,109 | | | | 1,030 | | | | +7.7 | % | | | +6.6 | % |
of which Eurasia(b) | | | 634 | | | | 597 | | | | +6.2 | % | | | +17.6 | % |
Europe (c) | | | 4,351 | | | | 4,758 | | | | -8.6 | % | | | -8.4 | % |
Rest of the World(d) | | | 1,813 | | | | 1,647 | | | | +10.1 | % | | | +6.3 | % |
of which Japan | | | 997 | | | | 875 | | | | +13.9 | % | | | +7.7 | % |
of which South Korea | | | 217 | | | | 206 | | | | +5.3 | % | | | +4.9 | % |
Total net sales | | | 17,019 | | | | 16,074 | | | | +5.9 | % | | | +4.1 | % |
| (a) | World excluding United States, Canada, Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
| (b) | Russia, Ukraine, Georgia, Belarus, Armenia and Turkey. |
| (c) | Europe excluding Eurasia. |
| (d) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
In theUnited States, 2019 first-half net sales were €5,401 million, up 15.5% on a reported basis and 7.8% at constant exchange rates. At constant exchange rates and on a constant structure basis (to reflect Bioverativ’s products, consolidated by Sanofi from March 9, 2018 onwards), net sales in the United States rose by 4.5% CER/CS. Solid performances by Dupixent®, Admelog® and Aubagio® more than offset lower sales of Lantus®(-35.2% CER at €568 million) and Renvela®/Renagel®(-54.5% CER at €59 million).
Emerging Markets net sales for the first half of 2019 reached €5,454 million, up 9.3% on a reported basis and up 11.8% CER. Sales growth was driven by Vaccines (+42.2% CER at €792 million), the Rare Diseases franchise (+34.3% CER at €323 million) and Diabetes (+11.7% CER at €870 million). InAsia, 2019 first-half net sales totaled €2,338 million (+15.7% CER) on a solid performance in China (+19.8 % CER at €1,507 million), especially in Vaccines (+193.0% CER at €167 million) with the resumption and growth of sales of Pentaxim®, and also growth in sales of Plavix® and Aprovel® ahead of the implementation of the Value Based Procurement program in key cities at the end of the first quarter of 2019. That program is expected to lead to a deceleration in sales growth of Plavix® and Aprovel® over 2019 as a whole. InLatin America, net sales for the first half of 2019 were up 8.5% CER at €1,305 million. First-half net sales in Brazil decreased by 2.6% CER to €503 million. Net sales in theAfrica & Middle Eastregion reached €1,109 million in the first half of 2019 (+6.6% CER) on good performances from Vaccines and the Rare Diseases franchise. Net sales in theEurasia region advanced by 17.6% CER to €634 million in the first half of 2019, the strongest performers being Turkey (+24.9% CER at €234 million) and Russia (+13.0% CER at €339 million).
InEurope, 2019 first-half net sales were down 8.4% CER at €4,351 million, reflecting the divestment of our European generics business (Zentiva). At constant exchange rates and on a constant structure basis, net sales in Europe decreased by 2.0%, with lower sales of Lovenox® and Lantus® not fully offset by growth in sales for Dupixent® and the Vaccines segment.
In theRest of the World region, net sales rose by 6.3% CER to €1,813 million. In Japan, 2019 first-half net sales amounted to €997 million (+7.7% CER), driven by growth in Vaccines and for Dupixent® and by the first-time consolidation of sales of Eloctate® and Alprolix®. At constant exchange rates and on a constant structure basis, net sales in Japan rose by 5.3%.
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 52
C.4. OTHER INCOME STATEMENT ITEMS
C.4.1. OTHER REVENUES
Other revenues advanced by 26.5% to €674 million in the first half of 2019 (versus €533 million in the first half of 2018). This line item mainly comprises VaxServe sales ofnon-Sanofi products (€543 million, versus €397 million for the first half of 2018, within the Vaccines segment), and revenues arising from the distribution of Eloctate® and Alprolix® (mainly in Europe) under our agreements with Swedish Orphan Biovitrum AB.
C.4.2. GROSS PROFIT
Gross profit amounted to €12,308 million in the first half of 2019, versus €11,342 million a year earlier, an increase of 8.5%. Gross margin increasedyear-on-year, representing 72.3% of net sales in the first half of 2019 (versus 70.6% in the first half of 2018).
For the Pharmaceuticals segment, gross margin for the first half of 2019 was 0.9 of a percentage point higher at 75.5%. Gross margin was lifted by a strong performance from the Immunology franchise, sales growth in Emerging Markets, the inclusion of Bioverativ’s products and the ending of royalty payments to Bristol-Myers Squibb on sales of Plavix® (excluding the United States and Puerto Rico) and Avapro®. This more than offset lower average net selling prices for insulin glargines, and lower sales of Established Prescription Products in mature markets.
Gross margin for the Consumer Healthcare segment rose by 0.2 of a percentage point in the first half of 2019 to 67.8% of net sales, thanks largely to a good performance in Emerging Markets and a favorable product mix in Europe.
The Vaccines segment saw gross margin increase by 6.2 percentage points in the first half of 2019 to 62.2% of net sales, reflecting robust sales growth for vaccines (especially in Emerging Market) and a favorable manufacturing performance.
C.4.3. RESEARCH AND DEVELOPMENT EXPENSES
Research and development expenses (R&D expenses) amounted to €2,972 million in the first half of 2019 (versus €2,755 million in the first half of 2018) and represented 17.5% of net sales (versus 17.1% in the first half of 2018). Overall, R&D expenses increased by 7.9%, mainly due to the acquisitions of Bioverativ and Ablynx and to spending on development programs in Diabetes, Immunology, Emerging Markets and Rare Blood Disorder within the Pharmaceuticals segment. R&D spend also increased in the Vaccines segment. At constant exchange rates, R&D expenses rose by 5.2% in the first half of 2019.
C.4.4. SELLING AND GENERAL EXPENSES
Selling and general expenses were €4,835 million in the first half of 2019 (28.4% of net sales), compared with €4,819 million for the first half of 2018 (30.0% of net sales). The 2019 first-half figure includes the €7 million effect of the depreciation ofright-of-use assets following application of the IFRS 16 lease accounting rules effective January 1, 2019. Excluding the effect of IFRS 16 and at constant exchange rates, selling and general expenses were 1.3% lower: the impact of the first-time consolidation of Bioverativ and Ablynx and additional marketing spend on Specialty Care products was offset by cost containment measures, especially in General Medicines products in mature markets and in our global support functions.
53 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.4.5. OTHER OPERATING INCOME AND EXPENSES
Other operating income amounted to €273 million in the first half of 2019 (versus €323 million in the first half of 2018), andOther operating expensesto €466 million (versus €165 million in the first half of 2018).
Overall, other operating income and expenses represented a net expense of €193 million in the first half of 2019, compared with a net gain of €158 million a year earlier, a netyear-on-year negative movement of €351 million.
| | | | | | | | | | | | |
| (€ million) | | June 30, 2019 | | | June 30, 2018 | | | Change | |
Other operating income | | | 273 | | | | 323 | | | | -50 | |
Other operating expenses | | | (466 | ) | | | (165 | ) | | | -301 | |
Other operating income/(expenses), net | | | (193 | ) | | | 158 | | | | -351 | |
The overall negative change of €351 million reflects (i) an increase in the net expense relating to our pharmaceutical partners (€223 million in the first half of 2019, versus €57 million in the first half of 2018), mainly as a result of the impact of higher sales of Dupixent® on our share of the profits due to Regeneron under our collaboration agreement (see Note C.1. to our consolidated financial statements, in our 2018 Annual Report on Form20-F); and (ii) a reduction in the level of capital gains on disposals, which totaled €71 million in the first half of 2019 (versus €226 million in the first half of 2018, when we divested various mature products in Latin America and some Consumer Healthcare products in Europe).
C.4.6. AMORTIZATION OF INTANGIBLE ASSETS
Amortization charged against intangible assets in the first half of 2019 was €1,116 million, versus €999 million in the comparable period of 2018. This €117 million rise mainly reflects an increase in amortization expense generated by the intangible assets recognized in connection with the March 2018 acquisition of Bioverativ (€272 million, versus €161 million in the first half of 2018). This was partly offset by reductions in amortization expense on intangible assets recognized on the acquisitions of Aventis (€107 million, versus €145 million in the first half of 2018) and Genzyme (€368 million, versus €385 million in the first half of 2018) as certain products reached the end of their life cycles.
C.4.7. IMPAIRMENT OF INTANGIBLE ASSETS
This line showed a net impairment loss of €1,840 million against intangible assets in the first half of 2019, compared with €101 million a year earlier. The main factor was an impairment loss of €1,609 million taken against Eloctate® franchise assets. This line item also includes an additional impairment loss of €33 million taken against rights relating to Lemtrada®, and net write-downs of €190 million relating toin-house or collaborative development projects.
In the first half of 2018, the results of impairment tests on other intangible assets led to the recognition of an impairment loss of €101 million, mainly on Lemtrada®, a product marketed in the United States.
C.4.8. FAIR VALUE REMEASUREMENT OF CONTINGENT CONSIDERATION
Fair value remeasurements of contingent consideration assets and liabilities relating to business combinations (recognized in accordance with the revised IFRS 3) represented a net gain of €190 million in the first half of 2019 versus a net gain of €10 million in the first half of 2018.
This mainly comprises remeasurements of contingent consideration (i) payable to Bayer as a result of an acquisition made by Genzyme prior to the latter’s acquisition by Sanofi (gain of €140 million in the first half of 2019, versus a gain of €33 million a year earlier and (ii) arising from the dissolution of the Sanofi Pasteur MSD joint venture (net gain of €98 million, versus a net expense of €1 million a year earlier. See Note B.11. to our condensed half-year consolidated financial statements. The above movements are partly offset by the impact of a €36 million increase in the contingent consideration arising from Sanofi’s March 2018 acquisition of Bioverativ.
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 54
C.4.9. RESTRUCTURING COSTS AND SIMILAR ITEMS
Restructuring costs and similar items amounted to a charge of €747 million in the first half of 2019, compared with a charge of €607 million in the first half of 2018. In 2019, restructuring costs mainly comprised employee-related expenses arising from headcount adjustment plans in the United States and Europe.
C.4.10. OTHER GAINS AND LOSSES, AND LITIGATION
In the first half of 2019,Other gains and losses, and litigation showed a net gain of €317 million (versus a net expense of €67 million in the first half of 2018), mainly arising from litigation.
C.4.11. OPERATING INCOME
Operating income for the first half of 2019 was €1,112 million, 48.6% lower than the 2018 first-half figure of €2,162 million, mainly as a result of the impairment losses taken against intangible assets in the period.
C.4.12. FINANCIAL INCOME AND EXPENSES
Net financial expenses were €150 million for the first half of 2019, €45 million higher than the 2018 first-half figure of €105 million.
Our cost of net debt (see the definition in Section C.7., “Consolidated balance sheet” below) fell to €89 million, versus €138 million in the first half of 2018. That reduction was more than offset by the following movements in net financial expenses:
| | • | | a decrease in gains on disposals ofnon-current financial assets (none in the first half of 2019, versus €63 million for the first half of 2018); |
| | • | | the fair value remeasurement of certain financial assets in accordance with IFRS 9, which became applicable on January 1, 2018 (negative impact of €15 million in the first half of 2019); |
| | • | | interest expense on lease liabilities (€20 million in the first half of 2019), reflecting the lease accounting impacts of IFRS 16 from January 1, 2019 onwards. |
C.4.13. INCOME BEFORE TAX AND INVESTMENTS ACCOUNTED FOR USING THE EQUITY METHOD
Income before tax and investments accounted for using the equity method for the first half of 2019 was €962 million, versus €2,057 million for the first half of 2018, a decrease of 53.2%.
C.4.14. INCOME TAX EXPENSE
Income tax expense represented €13 million in the first half of 2019, versus €297 million in the first half of 2018, giving an effective tax rate (based on consolidated net income) of 1.3%, compared with 14.4% in the first half of 2018. The decrease in income tax expense is mainly due to the tax effects of the amortization and impairment of intangible assets (€711 million in the first half of 2019, versus €275 million in the first half of 2018) and of restructuring costs (€197 million in the first half of 2019, versus €183 million in the first half of 2018) as well as the positive tax effects relating to the contingencies arising from business divestitures.
The effective tax rate on our business net income is anon-GAAP financial measure. It is calculated on the basis of business operating income, minus net financial expenses and before (i) the share of profit/loss from investments accounted for using the equity method and (ii) net income attributable tonon-controlling interests. We believe the presentation of this measure, used by our management, is also useful for investors as it provides a means to analyze the effective tax cost of our current business activities. It should not be seen as a substitute for the effective tax rate based on consolidated net income.
When calculated on business net income, our effective tax rate is 22% in the first half of 2019, compared with 22% in the first half of 2018 and 21,6 % for 2018 as a whole.
C.4.15. SHARE OF PROFIT/(LOSS) FROM INVESTMENTS ACCOUNTED FOR USING THE EQUITY METHOD
Investments accounted for using the equity method contributed net income of €116 million in the first half of 2019, versus net income of €75 million in the comparable period of 2018. In the first half of 2019 this line item mainly comprises our share of the profits of Regeneron (€106 million, versus €68 million in the first half of 2018); theyear-on-year rise mainly reflects an increase in the company profits of Regeneron as adjusted to reflect Sanofi accounting policies.
55 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.4.16. NET INCOME
Net income amounted to €1,065 million in the first half of 2019, versus €1,835 million in the first half of 2018.
C.4.17. NET INCOME ATTRIBUTABLE TONON-CONTROLLING INTERESTS
Net income attributable tonon-controlling interests for the first half of 2019 was €15 million, against €57 million for the first half of 2018. Theyear-on-year decrease was mainly due to the discontinuation (effective December 31, 2018) of payments by Sanofi of the share of ourpre-tax profits reverting to BMS on sales of Plavix® (excluding the United States and Puerto Rico) and Avapro®/Aprovel®, in accordance with our agreement with BMS (see Note C.2. to our consolidated financial statements included in our 2018 Annual Report on Form20-F).
C.4.18. NET INCOME ATTRIBUTABLE TO EQUITY HOLDERS OF SANOFI
Net income attributable to equity holders of Sanofi amounted to €1,050 million in the first half of 2019, compared with €1,778 million in the first half of 2018.
Basic earnings per share (EPS) was €0.84, compared with €1.42 for the first half of 2018, based on an average number of shares outstanding of 1,247.2 million for the first half of 2019 and 1,247.8 million for the first half of 2018. Diluted earnings per share was also €0.84, compared with €1.42 for the first half of 2018, based on an average number of shares after dilution of 1,254.7 million for the first half of 2019 and 1,254.9 million for the first half of 2018.
C.5. SEGMENT RESULTS
Business operating income (as defined in Note B.20.1. to the condensed half-year consolidated financial statements) amounted to €4,454 million in the first half of 2019 versus €4,126 million in the first half of 2018, an increase of 7.9%. It represented 26.2% of net sales, compared with 25.7% in the first half of 2018.
The table below shows business operating income for thesix-month periods ended June 30, 2019 and 2018:
| | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | Change | |
Pharmaceuticals segment | | | 4,579 | | | | 4,572 | | | | +0.2 | % |
Consumer Healthcare segment | | | 879 | | | | 820 | | | | +7.2 | % |
Vaccines segment | | | 513 | | | | 258 | | | | +98.8 | % |
Other | | | (1,517 | ) | | | (1,524 | ) | | | -0.5 | % |
Business operating income | | | 4,454 | | | | 4,126 | | | | +7.9 | % |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 56
C.6. CONSOLIDATED STATEMENTS OF CASH FLOWS
Summarized consolidated statements of cash flows
| | | | | | | | | | | | |
| (€ million) | | June 30, 2019 (6 months) | | | June 30, 2018 (6 months) | | | December 31, 2018 (12 months) | |
Net cash provided by/(used in) operating activities | | | 3,179 | | | | 1,773 | | | | 5,547 | |
Net cash provided by/(used in) investing activities | | | (165 | ) | | | (13,085 | ) | | | (12,866 | ) |
Net cash inflow/(outflow) from the exchange of the Animal Health business for BI’s Consumer Healthcare business | | | — | | | | 5 | | | | (6 | ) |
Net cash provided by/(used in) financing activities | | | (3,209 | ) | | | 8,494 | | | | 3,934 | |
Impact of exchange rates on cash and cash equivalents | | | 12 | | | | (9 | ) | | | 1 | |
Net change in cash and cash equivalents | | | (183 | ) | | | (2,822 | ) | | | (3,390 | ) |
Net cash provided by operating activitiescame to €3,179 million in the first half of 2019, against €1,773 million in the first half of 2018.
Operating cash flow before changes in working capital for the first half of 2019 was €3,976 million, versus €3,281 million in the first half of 2018. Working capital requirements rose by €798 million in the first half of 2019 (compared with a rise of €1,507 million in the first half of 2018), mainly as a result of a €934 million increase in inventories.
Net cash used in investing activitiestotaled €165 million in the first half of 2019, compared with €13,085 million in the first half of 2018.
Acquisitions of property, plant and equipment and intangible assets totaled €841 million, versus €823 million in the first half of 2018. There were €654 million of acquisitions of property, plant and equipment, most of which (€408 million) were in the Pharmaceuticals segment, primarily in industrial facilities. The Vaccines segment accounted for €243 million of acquisitions of property, plant and equipment during the period. Acquisitions of intangible assets (€187 million, versus €182 million in the first half of 2018) mainly comprised contractual payments for intangible rights under license and collaboration agreements.
Acquisitions of investments during the first half of 2019 totaled €158 million, net of the cash of acquired entities and after including assumed liabilities and commitments; this compares with €12,816 million in the first half of 2018. The main acquisitions in the first half of 2018 were Bioverativ (€8,932 million) and Ablynx (€3,639 million).
After-tax proceeds from disposals amounted to €867 million in the first half of 2019, and arose mainly from the sale of our equity interests in Alnylam (€706 million) and MyoKardia (€118 million). In the first half of 2018,after-tax proceeds from disposals amounted to €486 million, and arose mainly from the sale of some Consumer Healthcare products to Cooper-Vemedia (€139 million), the divestment of equity interests in Impact Therapeutics (€94 million) and the divestment of some mature products in Latin America (€44 million).
Net cash provided by/used in financing activitiesrepresented a net cash outflow of €3,209 million in the first half of 2019, compared with a net inflow of €8,494 million in the first half of 2018. The 2019 first-half figure includes the dividend payout to our shareholders of €3,834 million (versus €3,773 million in the first half of 2018), and net external debt financing raised of €585 million (versus a net amount of €13,032 million in the first half of 2018), including a €2 billion bond issue under our Euro Medium Term Notes program carried out in March 2019.
Thenet change in cash and cash equivalents in the first half of 2019 was a decrease of €183 million, compared with an increase of €2,822 million in the first half of 2018.
57 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
C.7. CONSOLIDATED BALANCE SHEET
Total assets were €110,545 million as of June 30, 2019, compared with €111,408 million as of December 31, 2018, an increase of €856 million.
Net debt was €18,705 million as of June 30, 2019, compared with €17,628 million as of December 31, 2018. We believe the presentation of thisnon-GAAP financial indicator, which is reviewed by our management, provides useful information to measure our overall liquidity and capital resources. We define “net debt” as (i) the sum total of short-term debt, long-term debt, and interest rate derivatives and currency derivatives used to manage debt, minus (ii) the sum total of cash and cash equivalents and interest rate derivatives and currency derivatives used to manage cash and cash equivalents.
| | | | | | | | |
| (€ million) | | June 30, 2019 | | | December 31, 2018 | |
Long-term debt | | | 21,087 | | | | 22,007 | |
Short-term debt and current portion of long-term debt | | | 4,411 | | | | 2,633 | |
Interest rate and currency derivatives used to manage debt | | | (51 | ) | | | (54 | ) |
Total debt | | | 25,447 | | | | 24,586 | |
Cash and cash equivalents | | | (6,742 | ) | | | (6,925 | ) |
Interest rate and currency derivatives used to manage cash and cash equivalents | | | — | | | | (33 | ) |
Net debt(a) | | | 18,705 | | | | 17,628 | |
Total equity | | | 56,518 | | | | 59,035 | |
Gearing ratio | | | 33.1 | % | | | 29.9 | % |
| (a) | Net debt does not include lease liabilities (see Note A.1.2 to our condensed consolidated financial statements). |
To assess our financing risk, we use the “gearing ratio”, anothernon-GAAP financial measure. This ratio (which we define as the ratio of net debt to total equity) increased from 29.9% as of December 31, 2018 to 33.1% as of June 30, 2019. Analyses of our debt as of June 30, 2019 and December 31, 2018 are provided in Note B.9. to the condensed half-year consolidated financial statements. We expect that the future cash flows generated by our operating activities will be sufficient to repay our debt. The financing arrangements in place as of June 30, 2019 at the Sanofi parent company level are not subject to covenants regarding financial ratios and do not contain any clauses linking credit spreads or fees to Sanofi’s credit rating.
Sanofi is applying IFRS 16, the new accounting standard on leases, effective January 1, 2019 (see Note A.1.2. to our condensed consolidated financial statements). As a result, we have recognized in our June 30, 2019 balance sheet(i) right-of-use assets totaling €1,105 million,(ii) non-current lease liabilities of €958 million, and (iii) current lease liabilities of €240 million.
Other key movements in the balance sheet are described below.
Totalequitywas €56,518 million as of June 30, 2019, versus €59,035 million as of December 31, 2018. The net change reflects the following principal factors:
| | • | | increases: our net income for the first half of 2019 (€1,065 million) and changes in currency translation differences (€410 million, mainly on the US dollar); and |
| | • | | decreases: the dividend payout to our shareholders (€3,834 million), and the impact of actuarial losses recognized during the period due to a downward adjustment to discount rates (€535 million). |
As of June 30, 2019 we held 0.02 million of our own shares, recorded as a deduction from equity and representing 0.002% of our share capital.
Goodwill andOther intangible assets (€63,516 million in total) decreased by €2,608 million, the main factors being:
| | • | | decreases: amortization and impairment charged during the period (€3,045 million, including the impairment loss taken against Eloctate® franchise assets); and |
| | • | | increases: the change in currency translation differences (€374 million). |
Investments accounted for using the equity method (€3,536 million) increased by €134 million, due mainly to the recognition of our share of Regeneron’s profits.
Othernon-current assets were €710 million lower at €2,261 million. The main movement during the year was the divestment of our equity interest in Alnylam.
Net deferred tax assets were €2,199 million as of June 30, 2019, compared with €1,199 million as of December 31, 2018. The increase of €1,000 million mainly reflects (i) the reversal of deferred tax liabilities relating to amortization and impairment of intangible assets, and (ii) the recognition of deferred tax assets on restructuring provisions and on provisions for pensions and other post-employment benefits (taking account of the actuarial losses recognized during the period).
Non-current provisions and other non-current liabilities (€9,099 million) rose by €486 million, mainly due to an increase in provisions for pensions and other post-employment benefits.
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 58
Liabilities related to business combinations and tonon-controlling interests (€1,012 million) were reduced by €292 million. The main reasons for the change are fair value remeasurements of contingent consideration payable to Bayer as a result of an acquisition made by Genzyme prior to the latter’s acquisition by Sanofi.
D/ RISK FACTORS AND RELATED PARTY TRANSACTIONS
D.1. RISK FACTORS
The risk factors to which Sanofi is exposed are described in our Annual Report on Form20-F for the year ended December 31, 2018, filed with the US Securities and Exchange Commission on March 8, 2019. The nature of those risks has not significantly changed during the first half of 2019. All those risk factors may materialize during the second half of 2019 or during subsequent periods.
D.2. RELATED-PARTY TRANSACTIONS
Our principal related parties are defined in Note D.33. to the consolidated financial statements included in our 2018 Annual Report on Form20-F (pageF-104)1.
Note B.5. to the condensed half-year consolidated financial statements provides a description of the principal transactions and balances for the six months ended June 30, 2019 with equity-accounted entities that qualify as related parties.
Sanofi did not enter into any transactions with key management personnel during the first half of 2019.
Financial relations with the Group’s principal shareholders fall within the ordinary course of business and were immaterial in the first half of 2019.
E/ OUTLOOK
At constant exchange rates, we expect growth in 2019 full-year business earnings per share2 (business EPS) to be approximately +5%, barring major unforeseen adverse events. The impact of exchange rates on 2019 business EPS is estimated to be approximately 1% to 2%, based on July 2019 average exchange rates applied over the rest of the year.
Full-year business net income2 for 2018 was €6,819 million, giving business earnings per share of €5.47.
This guidance has been prepared using accounting methods consistent with those used in the preparation of our historical financial information. It draws upon assumptions defined by Sanofi and its subsidiaries, in particular regarding the following factors:
| | • | | growth in the national markets in which we operate; |
| | • | | healthcare reimbursement policies, pricing reforms, and other governmental measures affecting the pharmaceutical industry; |
| | • | | developments in the competitive environment, in terms of innovative products and the introduction of generics; |
| | • | | respect by others for our intellectual property rights; |
| | • | | progress on our research and development programs; |
| | • | | the impact of our operating cost control policy, and trends in our operating costs; |
| | • | | trends in exchange rates and interest rates; |
| | • | | the integration of contributions from our acquisitions; and |
| | • | | the average number of shares outstanding. |
Some of the information, assumptions and estimates concerned are derived from or based, in whole or in part, on judgments and decisions made by Sanofi management that may be liable to change or adjustment in future.
| 1 | This report is available on our corporate website: www.sanofi.com. |
| 2 | For a definition, see Section C.2., “Business net income” above. |
59 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
FORWARD-LOOKING STATEMENTS
This document contains forward-looking statements as defined in the US Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects,” “anticipates,” “believes,” “intends,” “estimates,” “plans” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements.
These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis including post marketing, decisions by regulatory authorities such as the FDA or the EMA regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labeling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, Sanofi’s ability to benefit from external growth opportunities and/or obtain regulatory clearances, risks associated with intellectual property and current and future intellectual property litigation and the outcome thereof, trends in exchange rates and prevailing interest rates, the instability of economic conditions, the impact of cost containment initiatives and subsequent changes thereto, and the average number of shares outstanding, as well as those discussed or identified in the public filings with the Securities and Exchange Commission (SEC) and theAutorité des marchés financiers (AMF) made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s Annual Report on Form20-F for the year ended December 31, 20181 as filed with the SEC. For an update on litigation, refer to Note B.14. “Legal and arbitration proceedings” to our condensed half-year consolidated financial statements for the six months ended June 30, 2019, and to section “A.3.2. Legal and arbitration proceedings” (page 38) and section “D/ Risk factors and related party transactions” (page 59) of this half-year management report.
Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.
| 1 | See pages 4 to 19 of our 2018 Annual Report on Form 20-F, available on our corporate website: www.sanofi.com |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 60
|
| F/ APPENDIX – RESEARCH AND DEVELOPMENT PIPELINE |

| O: | Opt-in rights products for which rights have not been exercised yet |
| R: | Registrational Study (other than Phase 3) |
| (*) | Phase of projects determined by clinicaltrials.gov disclosure timing |
| (**) | Partnered and/or in collaboration – Sanofi may have limited or shared rights on some of these products |
| (1) | Developed in collaboration with Immunext |
| (2) | Regeneron product for which Sanofi has opt-in rights |
| (3) | Developed in collaboration with REVOLUTION Medicines |
| (4) | Developed in collaboration with BioNtech |
| (5) | Sanofi product for which Sobi has opt-in rights in SOBI territories |
| (6) | Recombinant Coagulation Factor VIII Fc – von Willebrand Factor – XTEN Fusion protein |
| (7) | Developed in collaboration with Sangamo |
| (8) | Developed in collaboration with Denali |
| (9) | Receptor-interacting serine/threonine-protein kinase 1 |
| (10) | Developed in collaboration with SK |
| (11) | Developed in collaboration with Lead Pharma |
| (12) | Developed in collaboration with Regeneron |
| (13) | Acid Sphingomyelinase Deficiency also known as Niemann Pick type B |
| (14) | Identification of out-licensing partner ongoing |
| (15) | Developed in collaboration with Principia |
| (16) | Autosomal Dominant Polycystic Kidney Disease |
| (17) | Developed in collaboration with Hanmi |
| (18) | Developed in collaboration with AstraZeneca |
61 | Sanofi –2019 HALF-YEAR FINANCIAL REPORT
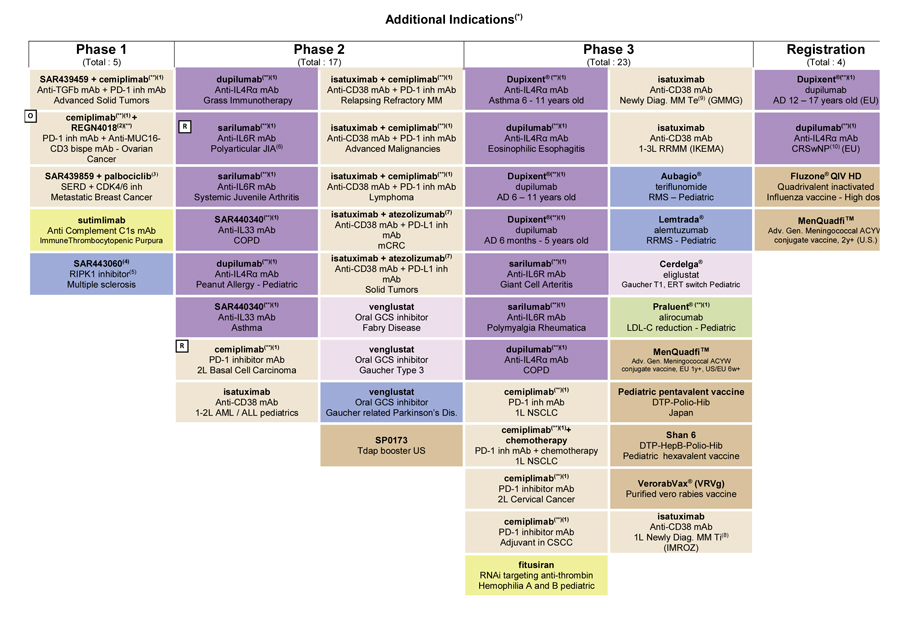
| (1) | Developed in collaboration with Regeneron |
| (2) | Regeneron product for which Sanofi has opt-in rights |
| (3) | Pfizer product (palbociclib) |
| (4) | Developed in collaboration with Denali |
| (5) | Receptor-interacting serine/threonine-protein kinase 1 |
| (6) | JIA: Juvenile Idiopathic Arthritis |
| (7) | Studies in collaboration with Roche (atezolizumab) |
| (10) | Chronic rhinosinusitis with nasal polyps |
| (*) | Phase of projects determined by clinicaltrials.gov disclosure timing |
| (**) | Partnered and/or in collaboration - Sanofi may have limited or shared rights on some of these products |
| O: | Opt-in rights products for which rights have not been exercised yet |
| R: | Registrational Study (other than Phase 3) |
2019 HALF-YEAR FINANCIAL REPORT – Sanofi | 62

