Table of Contents
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the fiscal year ended December 31, 2006 | ||
or | ||
o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the transition period from to | ||
Commission file number000-51745
SGX Pharmaceuticals, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 06-1523147 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
10505 Roselle Street
San Diego, CA 92121
(Address of principal executive offices, including zip code)
(858) 558-4850
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of Each Class | Name of Each Exchange on Which Registered | |
| Common Stock, $0.001 par value per share | NASDAQ Global Market |
Securities registered pursuant to Section 12(g) of the Act:
None
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No þ
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes o No þ
Indicate by check mark whether the Registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 ofRegulation S-K is not contained herein, and will not be contained, to the best of the Registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of thisForm 10-K or any amendment to thisForm 10-K. þ
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer and large accelerated filer” inRule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer o Accelerated filer o Non-accelerated filer þ
Indicate by check mark whether the registrant is a shell company (as defined inRule 12b-2 of the Securities Exchange Act of 1934). Yes o No þ
The aggregate market value of the Registrant’s Common Stock held by non-affiliates of the Registrant (without admitting that any person whose shares are not included in such calculation is an affiliate) computed by reference to the price at which the common stock was last sold as of the last business day of the Registrant’s most recently completed second fiscal quarter was $33,796,267.
As of March 13, 2007, the Registrant had 15,237,899 shares of Common Stock, $0.001 par value, issued and outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the definitive Proxy Statement for our 2007 Annual Meeting of Stockholders are incorporated by reference into Part III of this Report.
SGX PHARMACEUTICALS, INC.
ANNUAL REPORT ONFORM 10-K
FOR THE FISCAL YEAR ENDED DECEMBER 31, 2006
TABLE OF CONTENTS
ANNUAL REPORT ONFORM 10-K
FOR THE FISCAL YEAR ENDED DECEMBER 31, 2006
TABLE OF CONTENTS
Table of Contents
Cautionary Note Regarding Forward-Looking Statements
This annual report onForm 10-K contains forward-looking statements that involve many risks and uncertainties. These statements relate to future events and our future performance and are based on current expectations, estimates, forecasts and projections about the industries in which we operate and the beliefs and assumptions of our management. In some cases, you can identify forward-looking statements by terms such as “would,” “could,” “may,” “will,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “potential,” “targets,” “seek,” or “continue,” the negative of these terms or other variations of such terms. In addition, any statements that refer to projections of our future financial performance, our anticipated growth and trends in our business and other characterizations of future events or circumstances are forward-looking statements. These statements are only predictions based upon assumptions made that are believed to be reasonable at the time, and are subject to risk and uncertainties. Therefore, actual events or results may differ materially and adversely from those expressed in any forward-looking statement. In evaluating these statements, you should specifically consider the risks described under the caption “Risks Factors” in Item 1A of thisForm 10-K and elsewhere in thisForm 10-K. These factors may cause our actual results to differ materially from any forward-looking statements. Except as required by law, we undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
1
Table of Contents
PART I
| Item 1. | Business |
Overview
We are a biotechnology company focused on the discovery, development and commercialization of innovative cancer therapeutics. Our principal areas of focus in drug discovery are on protein and enzyme targets that have been implicated in cancers, including BCR-ABL, MET, JAK2 and RAS. We are advancing our internal oncology product pipeline through the application of our proprietary approach to drug discovery that is based upon the use of small fragments of drug-like molecules, or scaffolds, known as Fragments of Active Structures, or FAST.
We have applied FAST to generate novel, potent and selective small molecule compounds for many proteins, or drug targets that have been implicated in cancers and other diseases. The FAST drug discovery process involves identification and visualization of chemical scaffolds bound to the target protein. Thereafter, three-dimensional structural information is used to guide optimization of the potency and selectivity of the small molecule lead compounds bound to the target. The resulting low-molecular weight lead compounds typically possess good drug-like properties, thereby increasing the likelihood of success in further lead optimization and preclinical testing for eventual drug candidate selection. Lead optimization is the stage at which lead compounds discovered using FAST are further modified to improve their potency, specificity,in vivoefficacy and safety.
We are developing inhibitors of an enzyme known as BCR-ABL, in collaboration with Novartis Institutes for Biomedical Research (“Novartis”) under a license and collaboration agreement we entered into in March 2006. In this program, we designed and are developing these inhibitors for the treatment of Chronic Myelogenous Leukemia, or CML, a cancer of the bone marrow, which is resistant to treatment with the current standard of care, Gleevec® (imatinib mesylate) marketed by Novartis Pharmaceuticals Corporation. The goal of the collaboration is to develop an oral therapy for the treatment of both first-line and Gleevec-resistant CML. We are continuing to progress potential drug candidates from our lead series. Following a review by the collaboration’s Joint Research Committee, or JRC, of available preclinical data on the two most advanced candidates from our lead series, the JRC has decided to discontinue pursuing one of these candidates further and to collect additional data on the other, before determining whether to move it forward into investigational new drug (IND) enabling toxicology studies. In parallel, we have been advancingback-up compounds and a number of these are undergoing further preclinical testing. As a result, and contingent upon successful completion of these further preclinical studies, including toxicology studies, our anticipated timeline for the filing of an IND in this program is now the first half of 2008.
In February 2007, we announced the nomination of a MET development candidate, SGX523, for IND-enabling preclinical development. SGX523 is an internally developed, orally bioavailable small molecule inhibitor of the MET receptor tyrosine kinase. SGX523 has shown significant selectivity for MET over more than 200 protein kinases and potentin-vitroandin-vivoactivity. The MET receptor tyrosine kinase plays an important role in the control of cell growth, division and motility, and the formation of blood vessels, and is implicated as a causative agent in a broad range of solid tumors, including lung, colon, prostate, gastric, and kidney cancers. Pending successful completion of formal toxicology studies, we are targeting filing an IND application for SGX523 in the first quarter of 2008.
An additional internal program at the lead optimization stage is focused on the target JAK2. JAK2 is non-receptor tyrosine kinase protein that is mutated in a significant percentage of patients diagnosed with the blood disorders known as myeloproliferative disorders, such as Polycythemia Vera, Chronic Idiopathic Myelofibrosis, and Essential Thrombocythemia. For this target, our goal is to develop a once daily oral therapy for the relevant indications. Nomination of a development candidate in our JAK2 program is targeted for the second half of 2007.
Our current goal is to advance our oncology pipeline and file at least one new IND application per year. We intend to continue our strategy of entering into collaborative alliances around certain of our internal programs in order to generate near-term revenue and offset discovery and development costs, as we did with our BCR-ABL program. Based on FAST and related technologies, we have generated aggregate revenues from collaborations, commercial agreements and grants of approximately $76.7 million in 2006, 2005 and 2004.
2
Table of Contents
The chart below summarizes the status of our most advanced ongoing and currently planned clinical and preclinical development programs:
Program/Indication | Status | Marketing Rights | ||
BCR-ABL | ||||
• CML | Preclinical development | Novartis (Worldwide) | ||
| SGX (U.S. and Canada Commercialization rights) | ||||
MET | ||||
• Solid tumors | Preclinical development | SGX (Worldwide) | ||
JAK2 | ||||
• Myeloproliferative disorders | Lead optimization | SGX (Worldwide) | ||
RAS | ||||
• Solid tumors | Lead identification | SGX (Worldwide) |
BCR-ABL Kinase Inhibitor Program
Our BCR-ABL program is focused on compounds that inhibit both wild-type and Gleevec-resistant mutant forms of BCR-ABL tyrosine kinase, the enzyme that is responsible for CML. This program is being conducted in collaboration with Novartis under a license and collaboration agreement entered into in March 2006.
Chronic Myelogenous Leukemia
Chronic myelogenous leukemia (CML) is a malignant cancer of the bone marrow causing rapid and abnormal growth of white blood cells. The disease has an incidence of between 1 and 2 new patients per 100,000 individuals in the general population. In the US, this represents approximately 4,600 new patients a year. CML accounts for approximately 20 percent of adult leukemias. All patients with CML have a defective chromosome, known as a Philadelphia chromosome, in their leukemia cells. Prior to the introduction of Gleevec (imatinib), even with conventional therapy, a large majority of patients with CML inevitably progressed to die from their disease. In 2001, Gleevec was approved by the FDA and has become the standard of care for patients with CML. Gleevec works directly on leukemic cells by inhibiting the activity of the BCR-ABL tyrosine kinase protein, the enzyme responsible for uncontrolled growth of CML cells. Data from a five-year clinical study of Gleevec that was published in the New England Journal of Medicine documents the life-saving impact of this therapy for CML patients. Specifically, this study demonstrated that following five years of continuous daily therapy, 83% of patients remained in clinical remission, with an overall survival of 89%. The publication states “It is currently recommended that Imatinib be continued indefinitely”. This level of efficacy contributes to the clinical and commercial success of Gleevec, which had sales of approximately $2.6 billion in 2006, and the market is anticipated to continue to increase. However, not all patients will benefit indefinitely from single agent treatment with Gleevec. Over time, resistance emerges, with approximately 17% of patients relapsing within five years, and 4% of patients discontinuing therapy due to adverse events. The New England Journal of Medicine publication documents that 31% of patients receiving Gleevec failed to eliminate leukemic cells from their bone marrow within 12 months of commencing therapy. These patients are at significantly higher risk of relapse relative to those patients who do eliminate leukemic cells from the bone marrow within 12 months. In approximately two-thirds of cases, patient relapse has been linked to the emergence of mutant forms of BCR-ABL that are not inhibited by Gleevec. Multiple BCR-ABL mutants have been described and the single mutant that has proved the most challenging is known as the T315I mutant. There are a number of compounds being developed to address this mutant, however, to our knowledge there is no oral drug presently either in the clinic or on the market that inhibits the T315I mutant form of BCR-ABL.
SGX BCR-ABL Inhibitors
We are developing inhibitors of an enzyme known as BCR-ABL, in collaboration with Novartis under a license and collaboration agreement we entered into in March 2006. We are continuing to progress potential drug
3
Table of Contents
candidates from our lead series. Following a review by the collaboration’s Joint Research Committee, or JRC, of available preclinical data on the two most advanced candidates from our lead series, the JRC has decided to discontinue pursuing one of these candidates further and to collect additional data on the other, before determining whether to move it forward into IND-enabling toxicology studies. In parallel, we have been advancingback-up compounds and a number of these are undergoing further preclinical testing. As a result, and contingent upon successful completion of these further preclinical studies, including toxicology studies, our anticipated timeline for the filing of an IND in this program is now the first half of 2008.
Competition
In 2006, Bristol-Myers Squibb received FDA marketing approval for dasatinib (Sprycel®) and Novartis completed the submission of its New Drug Application for nilotinib (Tasigna®). Both agents provide new options for patients with resistanceand/or intolerance to treatment of certain forms of CML. Although both nilotinib and dasatinib inhibit some Gleevec-resistant BCR-ABL mutants, neither inhibits the T315I mutant. Treatment failure with either agent is believed to be most commonly due to the emergence of BCR-ABL mutants. Numerous other agents targeting CML or Gleevec-resistant BCR-ABL mutants are in various stages of preclinical and clinical development.
MET Tyrosine Kinase Receptor Inhibitor Program
c-MET (MET) is a protein, known as a receptor tyrosine kinase, which has been implicated in a wide range of cancers, including both solid and blood tumors. MET has been extensively studied in the laboratory environment with increasing data suggesting that uncontrolled stimulation of MET plays a key role in a variety of effects associated with cancer including cellular growth and increased cell movement and invasion, and an increased ability of cancer cells to metastasize, or spread beyond the organ of origin. Other observations have implicated MET in increased angiogenesis, a process by which tumors recruit new blood vessels to supply their increasing nutritional needs. Studies of tumors in humans have associated MET with more aggressive forms of cancer, such as lung and renal cancer, and activating MET mutations have been observed in a wide range of cancer types. Limited clinical activity data have been published from competitor agents currently in Phase I clinical trials. However, reports of the early clinical observations have suggested that some patients have experienced varying degrees of clinical benefit, including tumor response, tumor stabilizationand/or symptom improvement.
In February 2007, we announced the nomination of our first MET development candidate, SGX523, which was selected from a series of low molecular weight MET inhibitors. This series of compounds has demonstrated potency in cell based assays, selectivity against a wide variety of kinases and oral bioavailability in multiple animal species. We selected SGX523 based on its overall superior profile, which suggests the potential for an oral medication on a daily continuous dosing schedule. The high degree of selectivity of SGX523 over many other kinases may minimize cross target toxicity, which could make SGX523 a candidate for either single agent or combination therapy approaches with other agents currently in clinical use. We expect to complete formal toxicology studies during 2007, positioning us to file an IND application with the FDA for SGX523 in the first quarter of 2008.
Other Oncology Programs
We have also applied our FAST lead discovery and optimization technology to JAK2. JAK2 is a non-receptor tyrosine kinase involved in cytokine-induced signaling and growth regulation, survival, and differentiation of cells. Enhanced JAK2 activation has been implicated in various blood disorders. A particular JAK2 mutation, V617F, has been strongly correlated with a group of blood diseases known as myeloproliferative disorders (MPDs), such as Polycythemia Vera, Essential Thrombocythemia, and Chronic Idiopathic Myelofibrosis.
We have identified JAK2 inhibitors that have good potency against both wild-type and mutant JAK2 in cell-based assays. Oral bioavailability has also been demonstrated and current studies are aimed at optimizing the potency and drug like properties of these compounds. This program is in the lead optimization stage.
We also have a discovery program focused on a target called RAS. RAS is a protein that regulates cell growth. RAS activating mutations, which result in a cancer causing form of RAS, have been found in20-30% of all cancers and consequently, RAS has been implicated in a large number of diseases. However, RAS has proven a challenging
4
Table of Contents
target for the pharmaceutical industry and thus far there are currently no drugs on the market that directly target RAS. Applying our FAST platform, we have taken a novel approach to the modulation of RAS activity and we have identified inhibitors that have demonstrated cell-based activity. We are working to improve the potency of these inhibitors. Our RAS program is currently in the lead identification stage. Lead identification is the stage at which compounds are identified and further characterized in preparation for the lead optimization stage.
FAST — Our Drug Discovery Platform
FAST is our proprietary approach to drug discovery that uses X-ray crystallography and complementary biophysical and biochemical methods and medicinal and computational chemistry for the rapid discovery and optimization of novel, potent and selective small molecule inhibitors of drug targets with good drug-like properties. Through the application of FAST, we are building a pipeline of oncology drug candidates. FAST addresses many of the limitations of traditional approaches utilized by large pharmaceutical companies to identify and optimize lead compounds, making it an attractive technology for a broad range of drug discovery targets, including those that have not yielded promising leads from high-throughput screening. Unlike traditional lead discovery approaches, which require ultra high-throughput screening of very large numbers of compounds, FAST focuses on low molecular weight, water-soluble fragments as starting points for rapid synthesis and optimization under structural guidance to enable the delivery of novel, potent and selective modulators of drug targets.
We developed FAST through the integration of a series of technology capabilities, including:
| • | a high-throughput capability to generate many different crystals of a target protein in parallel; | |
| • | the crystallographic screening of our library of fragments and direct visualization of bound fragments utilizing X-ray crystallography; | |
| • | the use of novel computational design methods and iterative synthetic chemistry to optimize these fragments into drug-like lead compounds; and | |
| • | the use of structure guided drug design to enable the rapid optimization of lead compounds into drug candidates with low molecular weight, high ligand efficiency, and good drug like properties. |
We have combined these technologies to generate a platform for drug discovery applicable to a wide range of potential drug targets that exploits high chemical diversity and the potential for good drug-like properties. There are two main components of the FAST technology. The first is the identification of scaffolds that bind the target protein. Typically, these scaffolds are identified by screening our proprietary fragment library of approximately 1,500 structurally diverse, low molecular weight compounds using X-ray crystallography to visualize the small molecule bound to the target protein. Alternatively, scaffolds can be identified using complementary biophysical and biochemical measures of binding to the target protein. An additional means of identifying starting points for lead optimization exploits the breadth of medicinal chemistry experience at SGX and our growing library of partially elaborated scaffolds which were derived from applying FAST to many different protein targets. As this library continues to grow, we expect to make increasing use of opportunities derived from scaffolds and compounds that bind to more than one protein kinase target. In the next part of the FAST process, selected scaffolds are optimized to drug candidates. This second component is both rapid and efficient because it is guided by atomic resolution structural information of the scaffold and subsequently elaborated lead compounds bound to the target protein. This information, available to the chemistry team in real time, permits rational optimization of potency and other attributes.
We have invested significant resources in the development and optimization of technology to produce large numbers of protein variants and to evaluate their ability to produce high quality protein crystals. Customized, robotic technologies for setup, storage, retrieval and imaging of protein crystallization experiments have been developed and our current instrumentation supports in excess of 40,000 crystallization experiments per day. We generate protein structures through our beamline facility, housed at the Advanced Photon Source at the Argonne National Laboratory, a national synchrotron-radiation facility funded by the U.S. Department of Energy, Office of Science, and Office of Basic Energy Sciences, located in Argonne, Illinois. This facility produces an extremely intense, highly focused X-ray beam to generate high-resolution data from approximately 50 crystals per day.
5
Table of Contents
Our FAST drug discovery platform provides us with the capacity to pursue many different targets to the early lead stage, through lead optimization, and beyond. Internally, we have selected a portfolio of approximately 20 oncology targets that we believe are clearly implicated in cancers. We believe that FAST could provide a distinct advantage over conventional methods of lead discovery for these and other targets. Our most advanced programs developed using FAST are focused on compounds that inhibit BCR-ABL, MET, and JAK2. We are applying FAST to generate novel and potent lead compounds for well-validated protein and enzyme targets. In addition, through our high capacity and high resolution protein crystallography technology, FAST is being leveraged to drive our lead optimization programs. Our goal in each of these programs is to develop small molecule drugs with improved efficacy and reduced side effect profiles compared to current therapies or development compounds. Our current goal is to advance our oncology pipeline and file at least on new IND application per year.
Collaborations, Commercial Agreements and Grants
Since our inception, we have entered into multiple revenue-generating collaborations, commercial agreements and grants based upon FAST and related technologies with pharmaceutical and biotechnology companies, as well as government and other agencies. We generated aggregate revenues from collaborations, commercial agreements and grants of approximately $76.7 million in 2006, 2005 and 2004. Our active agreements include:
Collaborations and Grants:
Party | Scope | Start Date | Payments to SGX | |||
| Novartis Institutes for Biomedical Research, Inc. | Drug discovery, development and commercialization | Mar. 2006 | Upfront payment, research funding, milestones and royalties | |||
| Cystic Fibrosis Foundation Therapeutics, Inc. | Drug discovery | Jul. 2005 | Upfront payment; technology access fees; research funding; milestones; royalties | |||
| National Institutes of Health | Protein Structure Initiative | Jul. 2005 | Research funding | |||
| Eli Lilly & Company | Structural data on Eli Lilly targets and compounds | Apr. 2003 | Upfront payment; research funding; technology access fees |
License and Collaboration Agreement
Novartis Institutes for Biomedical Research, Inc.
In March 2006, we entered into a License and Collaboration Agreement with Novartis Institutes for Biomedical Research, Inc., (“Novartis”) focused on the development and commercialization of BCR-ABL inhibitors for the treatment of Chronic Myelogenous Leukemia. Under the agreement, the parties are collaborating to develop one or more BCR-ABL inhibitors and Novartis will have exclusive worldwide rights to such compounds, subject to our co-commercialization option in the United States and Canada. We have also granted Novartis rights to include certain compounds that we do not pursue under the collaboration in Novartis’ screening library and we will be entitled to receive royalties on sales of products based on those compounds.
Under the terms of the agreement, we received $25.0 million of upfront payments, including $5.0 million for the purchase by Novartis Pharma AG of shares of our common stock. We are also entitled to receive research funding over the first two years of the collaboration of $9.1 million. With payments for achievement of specified development, regulatory and commercial milestones, including $9.5 million for events up to and including commencement of the first Phase I clinical study, total payments to us could exceed $515 million. We are responsible for completing preclinical development, submitting an IND and completing the initial Phase I study for the first lead drug candidate.
Novartis will fund 100% of the development costs of product candidates from the collaboration. The research and development activities of the parties are overseen by committees with equal representation of the parties, with Novartis having the right to make the final decision on certain matters. We are also eligible to receive royalties based
6
Table of Contents
on net sales. In addition, we retain an option to co-commercialize in the United States and Canada oncology products developed under the agreement through a sales force trained and funded by Novartis.
The agreement will continue until the expiration of all of Novartis’ royalty payment obligations, unless the agreement is terminated earlier by either party. Novartis and we each have the right to terminate the agreement early if the other party commits an uncured material breach of its obligations. If Novartis terminates the agreement for material breach by us, Novartis’ licenses under the agreement will continue subject to certain milestone and royalty payment obligations. If we terminate the agreement for material breach by Novartis, all rights to compounds developed under the collaboration will revert to us. Further, Novartis may terminate the agreement without cause if it reasonably determines that further development of compounds or products from the collaboration is not viable, in which event all rights to the compounds and products revert to us. In the event of a change in control of our company, in certain circumstances Novartis may terminate only the joint committees and co-commercialization option, with all other provisions of the agreement remaining in effect, including Novartis’ licenses and its obligations to make milestone and royalty payments.
Cystic Fibrosis Foundation Therapeutics, Inc.
In July 2005, we entered into a drug discovery collaboration agreement with Cystic Fibrosis Foundation Therapeutics, Inc., or CFFT, the drug discovery and development arm of the Cystic Fibrosis Foundation. Under the collaboration, we employ our proprietary FAST lead generation technology with the objective of generating novel small molecule therapies that function as “correctors” of the F508 deletion mutation found in the cystic fibrosis transmembrane conductance regulator, or CFTR. The F508 deletion mutation is the most commonly observed mutation in patients with cystic fibrosis. Individuals with the mutation fail to transport the CFTR protein to the cell surface, resulting in impaired function of the lung epithelium. Correctors of the mutant protein are expected to increase the amount of the mutant protein that is transported to the cell surface, resulting in more rapid clearing of lung infections and improved lung function. CFFT will be responsible for product development and we will be eligible for clinical development milestones and royalties on product sales. The research term of this collaboration agreement continues until July 2008. Our drug discovery agreement with CFFT may be terminated earlier by either party in the event of a material breach by the other party, subject to prior notice and the opportunity to cure. In addition, CFFT has the right to terminate the drug discovery agreement at any time upon 60 days notice.
NIH Cooperative Agreement Award
In July 2005, we received a $48.5 million National Institutes of Health Cooperative Agreement Award from the National Institute of General Medical Sciences, or NIGMS. The award is part of the NIH Protein Structure Initiative, which aims to facilitate discovery of three dimensional structures of proteins to help reveal their role in disease and aid in the design of new medicines. The award provides five years of funding for a consortium administered by us. We anticipate retaining approximately 50% of the funding under the award, with the remainder being distributed to academic collaborators.
Eli Lilly & Company
In April 2003, we entered into a research and technology agreement with Eli Lilly, which was extended in April 2005. Within this commercial agreement, we apply ourtarget-to-structure technology to key Eli Lilly drug targets to determine their three-dimensional structures. Our researchers subsequently generate data on Eli Lilly compounds that bind to the drug targets, providing input for their lead generation and optimization efforts. In parallel with the first two years of research under the agreement, we conducted a comprehensive program of technology transfer involving installation of components of our technology in a high-throughput structural biology facility for Eli Lilly, which includes modular automation systems and process technology we developed for protein engineering, crystallization and structure determination.
The research term of this commercial agreement continues until April 2008. The general terms of this commercial agreement continue until the later of the expiration of the last to expire of the patent rights covering technology developed under the agreement or April 2018, unless the agreement is earlier terminated. Either party may terminate the commercial agreement in the event of material breach by the other party, subject to prior notice
7
Table of Contents
and the opportunity to cure. In addition, Eli Lilly may terminate the agreement if certain of our key employees leave our employment and significantly curtail participation in the project, or in the event we are acquired by one of the top 25 pharmaceutical companies ranked by worldwide sales.
In December 2003, we also expanded our research collaboration and technology agreement with Eli Lilly to provide Eli Lilly with long-term access to our beamline facility at the Advanced Photon Source in Argonne, Illinois, to support Eli Lilly drug discovery programs. Under the terms of our beamline services agreement with Eli Lilly, we generate crystal structure data on Eli Lilly drug targets and compounds in exchange for upfront access fees and maintenance fees paid by Eli Lilly. Eli Lilly also has the option to extend the term of its access to our beamline facility in the future for additional payments. The term of this beamline agreement continues until January 2012, unless Eli Lilly exercises its option to extend the term of its access to our beamline facility or the agreement is earlier terminated. Either party may terminate the agreement in the event of a material breach by the other party, subject to prior notice and the opportunity to cure. In addition, Eli Lilly may terminate the agreement at any time, subject to prior notice.
Shire
In July 2004, we licensed exclusive worldwide rights to Troxatyl from Shire BioChem, Inc. (“Shire”), including an exclusive sublicense under rights Shire has to certain patents and patent applications in the field of the treatment of cancer from Yale University and the University of Georgia Research Foundation. Under the terms of the agreement, we made an upfront payment of $3.0 million and a payment of $1.0 million on the one-year anniversary of the agreement. In August 2006, we announced that we had discontinued our Phase II/III clinical trial of Troxatyl for the third line treatment of acute myelogeous leukemia, or AML, based on a recommendation from the study’s independent data and safety monitoring board. The data and safety monitoring board found that interim study response rates were unlikely to provide evidence of a treatment benefit as a third line treatment for patients with AML. Following a review of our oncology product pipeline and the resources available to us, we have determined we will not initiate additional Troxatyl clinical studies while, in consultation with Shire, we consider the future of Troxatyl.
Our Strategy
Our goal is to create a leading biotechnology company that discovers, develops and commercializes novel cancer drugs. Key elements of our strategy are to:
| • | Focus on oncology. Despite recent advances in the treatment of cancer, there continue to be areas of significant unmet medical need. New approaches to cancer treatment such as targeted therapies, to which we believe our technology is uniquely suited, provide companies such as ours an opportunity to advance our pipeline through preclinical and clinical development to provide patients with life saving therapies. Furthermore, we consider drug development for the cancer markets attractive because relatively small clinical trials of short duration can provide meaningful data on patient outcomes. | |
| • | Utilize our FAST drug discovery platform to generate lead candidates. Our FAST drug discovery platform provides us with the capability to pursue many different targets to the early lead stage and beyond. Internally, we have identified a portfolio of approximately 20 oncology targets that we believe are clearly implicated in cancers. We will seek to apply FAST to high-value cancer targets with the objective of building and advancing a sustainable oncology pipeline. | |
| • | Advance development candidates and commercialize product candidates. Our goal is to progress our product candidates through preclinical and clinical development, and ultimately to commercialization, while utilizing strategic partnering as appropriate. | |
| • | Continue to access capabilities and generate revenue through strategic partnering. Revenue generation from strategic partnering will continue to be important to us in the near term by providing funds for reinvestment in internal drug discovery and development. Our business development activities will involve strategic partnering of certain of our oncology programs. Oncology partnerships will be sought with organizations that provide complementary capabilities to allow rapid progression of our product candidates |
8
Table of Contents
| to the market. We will remain open to opportunities to apply FAST to targets outside the oncology area, particularly where there are attractive financial or strategic opportunities. |
Intellectual Property
We seek to protect our novel lead compounds, lead scaffolds, drug discovery programs and proprietary technologies by filing appropriate patent applications. We have approximately 100 U.S. and foreign pending patent applications covering compositions of matter, drug discovery methods and assays, protein structures and elements of our high-throughput structure determination platform. We intend to continue to file patent applications on lead series and drug discovery methods, including FAST and assays to support our drug discovery platform. We currently have two issued U.S. patents directed to aspects of our high-throughput structure determination platform.
In addition, we have an exclusive license to a number of issued U.S. and foreign patents, and pending U.S. and foreign applications, covering composition of matter, method of use and treatment, formulation and process related to Troxatyl.
There can be no assurance that any of our patent applications will issue in any jurisdiction. Moreover, we cannot predict the breadth of claims that may be allowed or the actual enforceable scope of our patents. In the United States, we may lose our patent rights if we were not the first to invent the subject matter covered by each of our issued patents or pending patent applications. We cannot be certain that our patents will be found valid and enforceable, or that we will not be found to infringe issued patent claims of any third party or that third parties will be found to infringe any of our issued patent claims.
Although we have taken steps to protect our trade secrets and unpatented know-how, including entering into confidentiality agreements with third parties, and confidential information and inventions agreements with employees, consultants and advisors, third parties may still obtain this information or we may be unable to protect our rights. Enforcing a claim that a third party illegally obtained and is using our trade secrets or unpatented know-how is expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States may be less willing to protect trade secret information. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how, and we would not be able to prevent their use.
Third Party Intellectual Property
Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the field of small molecule kinase inhibitors and fields in which we and our collaborators are developing products. Because patent applications can take many years to issue, there may be currently pending applications, unknown to us, which may later result in issued patents that our product candidates or proprietary technologies may infringe.
We may be exposed to, or threatened with, future litigation by third parties having patent or other intellectual property rights alleging that our product candidatesand/or proprietary technologies infringe their intellectual property rights. If one of these patents was found to cover our product candidates, proprietary technologies or their uses, we or our collaborators could be required to pay damages and could be restricted from commercializing our product candidates or using our proprietary technologies unless we or they obtain a license to the patent. A license may not be available to us or our collaborators on acceptable terms, if at all. In addition, during litigation, the patent holder could obtain a preliminary injunction or other equitable right, which could prohibit us from making, using or selling our products, technologies or methods.
There is a substantial amount of litigation involving patent and other intellectual property rights in the biotechnology and biopharmaceutical industries generally. If a third party claims that we or our collaborators infringe its intellectual property rights, we may face a number of issues, including but not limited to:
| • | infringement and other intellectual property claims which, with or without merit, may be expensive and time-consuming to litigate and may divert our management’s attention from our core business; |
9
Table of Contents
| • | substantial damages for infringement, including treble damages and attorneys’ fees, which we may have to pay if a court decides that the product or proprietary technology at issue infringes on or violates the third party’s rights; | |
| • | a court prohibiting us from selling or licensing the product or using the proprietary technology unless the third party licenses its technology to us, which it is not required to do; | |
| • | if a license is available from the third party, we may have to pay substantial royalties, feesand/or grant cross licenses to our technology; and | |
| • | redesigning our products or processes so they do not infringe, which may not be possible or may require substantial funds and time. |
We have not conducted an extensive search of patents issued to third parties, and no assurance can be given that such patents do not exist, have not been filed, or could not be filed or issued, which contain claims covering our product candidates, technology or methods. Because of the number of patents issued and patent applications filed in our technical areas or fields, we believe there is a significant risk that third parties may allege they have patent rights encompassing our product candidates, technology or methods.
Sales and Marketing
We currently do not have sales and marketing capabilities and we have no plans to develop such capabilities in the near future. If we do advance any of our product candidates into and through clinical development, we will need to build a sales and marketing infrastructure. Under our license and collaboration agreement with Novartis, we retain an option to co-commercialize in the United States and Canada oncology products developed under the agreement through a sales force trained and funded by Novartis. For other programs, we may pursue strategic collaborations, as appropriate, to commercialize our product candidates on a world-wide basis.
Competition
We operate in highly competitive segments of the biotechnology and biopharmaceutical markets. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies, and private and public research institutions. There is also intense competition for fragment-based lead discovery collaborations. Many of our competitors have significantly greater financial, product development, manufacturing and marketing resources than us. Large pharmaceutical companies have extensive experience in clinical testing and obtaining regulatory approval for drugs. These companies also have significantly greater research capabilities than us. In addition, many universities and private and public research institutes are active in cancer research, some in direct competition with us. We also compete with these organizations to recruit scientists and clinical development personnel. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
Each cancer indication for which we are developing products has a number of established therapies with which our candidates will compete. Most major pharmaceutical companies and many biotechnology companies are aggressively pursuing new cancer development programs, including both therapies with traditional, as well as novel, mechanisms of action.
We are aware of competitive products and technologies in each of the markets we target. The competitive products include approved and marketed products as well as products in development.
We expect that any BCR-ABL inhibitor that we may potentially develop for treatment of CML to compete with Gleevec® (imatinib), marketed by Novartis, Inc., Tasigna® (nilotinib), for which an NDA was recently filed by Novartis, Inc., Sprycel® (dasatinib), marketed by Bristol Myers Squibb, Inc., MK-0457, under development by Merck and Co., SKI-606, under development by Wyeth, Inc., INNO-406, under development by Innovive Pharmaceuticals, Inc., homoharringtonine, under development by ChemGenex, Inc., KW-2449, under development by Kyowa Pharma, Inc., and XL228, under development by Exelixis, Inc. Other potential competing products are in clinical trials and preclinical development.
10
Table of Contents
We expect that any MET inhibitor that we may potentially develop for treatment of cancers, to compete with XL880 and XL184, under development by Exelixis, Inc., ARQ197, under development by Arqule, Inc., PF02341066, under development by Pfizer, Inc., AMG102, under development by Amgen, Inc., MP470, under development by SuperGen, Inc., and MGCD265, under development by Methylgene Inc. Other potential competing products are in clinical trials and preclinical development.
In each of our development programs addressing indications for which there are therapies available, we intend to complete clinical trials designed to evaluate the potential advantages of our drug candidates as compared to or in conjunction with the current standard of care. Key differentiating elements affecting the success of all of our drug candidates are likely to be their efficacy, safety and side-effect profile compared to commonly used therapies.
Significant competitors in the area of fragment-based drug discovery include Astex Therapeutics Limited, Plexxikon Inc., Evotec AG, Vernalis Plc., Sunesis Pharmaceuticals, Inc., and Active Site, Inc. In addition, many large pharmaceutical companies are exploring the internal development of fragment-based drug discovery methods.
Government Regulation and Product Approvals
The clinical development, manufacturing and future marketing of our products are subject to regulation by various authorities in the United States, the E.U., and other countries. The Federal Food, Drug, and Cosmetic Act, or FD&C Act, and the Public Health Service Act in the United States, and numerous directives, regulations, local laws, and guidelines in the E.U. govern the testing, manufacture, safety, efficacy, labeling, storage, record keeping, approval, advertising and promotion of pharmaceutical products. Product development and approval within these regulatory frameworks takes a number of years, and involves the expenditure of substantial resources.
Regulatory approval to conduct clinical trials will be required in any territories in which we, or our licensors, seek to test our development products. Prior to human testing, such approval requires evaluation of product quality as well as animal data relating to safety and, where relevant, efficacy. In general, new chemical entities are tested in animals to determine whether the product is reasonably safe for initial human testing. Clinical trials for new products are typically conducted in three sequential phases that may overlap. Within oncology, Phase I trials typically involve the initial introduction of the pharmaceutical into patients with advanced malignancy and the emphasis is on testing for safety, dosage tolerance, metabolism, distribution, excretion and clinical pharmacology. Phase II trials involve the evaluation of effectiveness of the drug for a particular indication in patients with the disease under study, and to determine the common short-term side effects and risks associated with the drug. Phase II trials are typically closely monitored and conducted in a relatively small number of patients, usually involving no more than fifty to one hundred subjects. Phase III trials are generally expanded, well-controlled clinical trials. They are performed after preliminary evidence suggesting effectiveness of the drug has been obtained, and are intended to gather the additional information about safety and effectiveness needed to evaluate the overall risk-benefit relationship of the drug and to provide an adequate basis for product labeling.
In the United States an Investigational New Drug application, or IND, must be submitted to the FDA prior to the initiation of human studies. Absent an objection from the FDA, the application will become effective 30 days following receipt by the FDA. Prior regulatory approval to initiate human studies is also required in member states of the E.U. Additional requirements designed to protect the rights of participating patients also exist. Approval by an appropriately constituted Institutional Review Boards (IRB) in the United States or an equivalent Ethics Committee in other territories (EC) is also required prior to the commencement ofanyclinical trial. The ongoing conduct of the study is monitored on a periodic basis by the sponsor, institutional committees, as well as regulatory authorities. The submission of relevant safety data on both an episodic and periodic basis to such parties is required, as well as well-defined processes to support this activity. Authorities could demand discontinuation of studies at any time if significant safety issues arise. In all cases, it is our responsibility to ensure that we conduct our business in accordance with the regulations of each relevant territory.
In order to gain marketing approval, we must submit a dossier to the relevant authority for review, which is known in the United States as a new drug application (NDA) and in the E.U. as a marketing authorization application (MAA). The format of a marketing application has recently been standardized and includes information specified by each authority, and requires information on the quality of the chemistry, manufacturing and pharmaceutical aspects of the product, as well as non-clinical and clinical data. Failure to adequately demonstrate the
11
Table of Contents
quality, safety and efficacy of a therapeutic drug under development would delay or prevent regulatory approval of the product. There can be no assurance that if clinical trials are completed, either we or our collaborative partners will submit applications for required authorizations to manufacture or market potential products, including a marketing authorization application or an NDA, or that any such application will be reviewed and approved by appropriate regulatory authorities in a timely manner, if at all.
In general, the competent regulatory authority may approve a product if the data is considered to be of a high quality and supportive of the indication requested. Quality of data is usually determined through regulatory audits of the various components of the dossier, and may include site visits to clinical trial sites and manufacturing facilities. In some circumstances, additional data or clinical trials may be requested during the review and may delay marketing approval and involve unbudgeted costs. Regulatory authorities may find data to be of an unacceptable quality or not supportive of the indication sought; in these circumstances, regulatory approval to market products may be denied or deferred.
As a condition of marketing approval, competent regulatory authorities also require post-marketing surveillance to monitor adverse effects, and may also request other additional studies as deemed appropriate. After approval for the initial indication, further clinical studies are usually necessary to gain approval for additional indications. The terms of any approval, including labeling content, may be more restrictive than expected and could affect product marketability.
The FDA has implemented special programs to facilitate the development and to expedite the review of drugs intended to treat serious and life-threatening conditions so that this type of product can be approved and reach the market quickly. A drug that demonstrates a meaningful therapeutic advantage over existing treatments or shows the potential to address an unmet medical need in a serious or life-threatening condition may be considered for expedited approval. In some cases, where approval is granted on the basis of a surrogate measure of benefit, further clinical trials (as post-approval commitments) are generally required to further define the safety and efficacy of the product. If such clinical trials fail to confirm the early benefits seen during the accelerated approval process, the FDA may withdraw approval. A similar set of mechanisms exist within the E.U.
The United States and the E.U. may grantorphan drugdesignation to drugs intended to treat a “rare disease or condition,” which, in the United States, is generally a disease or condition that affects fewer than 200,000 individuals nationwide. In the E.U., orphan drug designation can be granted if:
| • | The disease affects no more than 50 in 100,000 persons in the E.U.; | |
| • | The drug is intended for a life-threatening, seriously debilitating, or serious and chronic condition; | |
| • | The medical plausibility of the proposed orphan indication; | |
| • | Without incentives it is unlikely that the drug would generate sufficient return to justify the necessary investment; and | |
| • | No satisfactory method of treatment for the condition exists or, if it does, the new drug will provide a significant benefit to those affected by the condition. |
The designation of anorphan drugstatus provides the company with a limited period of market exclusivity for the indication of interest (seven years in the United States, and ten years in the E.U.). Orphan drug designation does not prevent competitors from developing or marketing different drugs for an orphan indication or the same drug for a different indication.
Throughout the period of active marketing of any medicinal product, the company retains the responsibility to periodically and systematically review the safety profile of the marketed product. This requires an active pharmacovigilance program, and the company is required to report certain adverse events, safety trends, relavent literature reports and similar data to the competent regulatory authority. Similarly, the advertising and promotion of pharmaceutical products is also closely regulated and monitored by regulatory agencies. Moreover, quality control and manufacturing procedures must continue to conform to current Good Manufacturing Practices (cGMPs) after approval, and the FDA periodically inspects manufacturing facilities to assess cGMP compliance. Accordingly,
12
Table of Contents
manufacturers must continue to expend resources on production, quality control and quality assurance to maintain compliance with GMP and other regulatory requirements.
Failure to comply with applicable regulatory requirements after obtaining regulatory approval can, among other things, result in suspension of regulatory approval, and possible civil and criminal sanctions. Renewals of the license in Europe may require additional data, which may result in an approval being withdrawn. In the United States and the E.U., regulators have the authority to revoke, suspend or withdraw approvals of previously approved products, to prevent companies and individuals from participating in the drug-approval process, to request recalls, to seize violative products, to obtain injunctions to close manufacturing plants not operating in conformity with regulatory requirements and to stop shipments of violative products. In addition, changes in regulation could harm our financial condition and results of operation.
Employees
As of December 31, 2006, we had 124 employees, including 46 who hold Ph.D. or M.D. degrees. We had 106 employees engaged in research and development, and our remaining employees are management or administrative staff. None of our employees are subject to a collective bargaining agreement. We believe that we have good relations with our employees.
| Item 1A. | Risk Factors |
You should carefully consider the following information about these risks, together with the other information appearing elsewhere in this report. If any of the following risks actually occur, our business, financial condition, results of operations and future growth prospects would likely be materially and adversely affected. In these circumstances, the market price of our common stock could decline, and you may lose all or part of your investment in our common stock.
Risks Relating to Our Business
Our drug discovery approach and technologies are unproven and may not allow us to establish or maintain a clinical development pipeline or successful collaborations or result in the discovery or development of commercially viable products.
The technologies on which we rely are unproven and may not result in the discovery or development of commercially viable products. There are currently no drugs on the market and no drug candidates in clinical development that have been discovered or developed using our proprietary technologies. Troxatyl, our only drug candidate to date that has been in clinical development, was licensed from Shire in 2004. We have no current plans for continuing the clinical development of Troxatyl. During the past several years, we have transitioned our business strategy from focusing on our protein structure determination capabilities and developing our technology infrastructure, to focusing on drug discovery and development activities in the field of oncology. Our most advanced programs are our BCR-ABL program, which is currently the subject of a license and collaboration agreement with Novartis and our MET program. Both of these programs are in the preclinical development stage and there is no guarantee that these programs will advance through pre-clinical development into clinical development. The process of successfully discovering product candidates is expensive, time-consuming and unpredictable, and the historical rate of failure for drug candidates is extremely high. Research programs to identify product candidates require a substantial amount of our technical, financial and human resources even if no product candidates are identified. Data from our current research programs may not support the clinical development of our lead compounds or other compounds from these programs, and we may not identify any compounds suitable for recommendation for clinical development. Moreover, there is presently little or no clinical validation for the targets which are the focus of the programs in our oncology pipeline such as MET or JAK2, other than our BCR-ABL program, and there is no guarantee that we will be able to successfully advance any compounds we recommend for clinical development from these programs. We have experienced a delay in selecting a development candidate in our BCR-ABL program. Following a review by the collaboration’s Joint Research Committee, or JRC, of available preclinical data on the two most advanced candidates from our lead series, the JRC has decided to discontinue pursuing one of these candidates further and to collect additional data on the other, before determining whether to
13
Table of Contents
move it forward into IND-enabling toxicology studies. In parallel, we have been advancingback-up compounds and a number of these are undergoing further preclinical testing. As a result, and contingent upon successful completion of these further preclinical studies, including toxicology studies, our anticipated timeline for the filing of an IND in this program is now the first half of 2008. However, our back-up compounds may never successfully complete preclinical studies and we may never successfully file an IND or commence clinical trials of any compounds in our BCR-ABL program. In addition, compounds we recommend for clinical development may not be effective or safe for their designated use, which would prevent their advancement into clinical trials and impede our ability to maintain or expand our clinical development pipeline. If we are unable to identify new product candidates or advance our lead compounds into clinical development, we may not be able to establish or maintain a clinical development pipeline or generate product revenue. There is no guarantee that we will be able to successfully advance any product candidates in our preclinical programs into clinical trials or successfully develop any product candidate we advance into clinical trials for commercial sale. Although it has been our goal to file one IND per year, to date, we have not filed an IND for any internally developed product candidate. We may not ever successfully file an IND or commence clinical trials of an internally developed compound.
The results of early preclinical studies are not necessarily predictive of the results of future preclinical studies, and there is no guarantee that any of our drug candidates in preclinical development will progress through to clinical development.
Positive results from early preclinical studies on drug candidates should not be relied upon as evidence that the results of further preclinical studies will be successful or that the drug candidate will progress into clinical studies. Drug discovery is inherently unpredictable, and the historical rate of failure for drug candidates in preclinical testing is extremely high. Drug candidates that have shown promising results in studies in rodents can have negative results when evaluated further in higher species, such as our experience with one of the lead candidates in our BCR-ABL program. If negative preclinical results are seen in more than one compound from a particular chemical series, there may be an increased likelihood that additional compounds from that series will demonstrate the same or similar negative results. Companies frequently suffer setbacks in preclinical studies such as our experience with the lead candidates in our BCR-ABL program. There is no guarantee that any of our drug candidates, including those in our BCR-ABL program, will progress through preclinical development into clinical development.
Because the results of preclinical studies are not necessarily predictive of future results, any product candidate we advance into clinical trials may not have favorable results or receive regulatory approval.
Even if any of our product candidates advance through pre-clinical development, positive results from preclinical studies should not be relied upon as evidence that clinical trials will succeed. We will be required to demonstrate through clinical trials that our product candidates are safe and effective for use in a diverse population before we can seek regulatory approvals for their commercial sale. Success in preclinical testing does not mean that clinical trials will be successful because product candidates in clinical trials may fail to demonstrate sufficient safety and efficacy despite having progressed through pre-clinical testing. Companies frequently suffer significant setbacks in clinical trials, even after earlier clinical trials have shown promising results, such as our experience with the pivotal Phase II/III clinical trial of Troxatyl for the third-line treatment of AML. There is typically an extremely high rate of attrition from the failure of drug candidates proceeding through clinical trials.
If any product candidate fails to demonstrate sufficient safety and efficacy in any clinical trial we are able to undertake, we would experience potentially significant delays in, or be required to abandon, development of that product candidate which may cause our stock price to decline further and may materially and adversely affect our business.
Delays in the commencement or completion of clinical testing could result in increased costs to us and delay our ability to generate significant revenues.
Delays in the commencement or completion of clinical testing could significantly impact our product development costs. We do not know whether any clinical trials that we may plan in the future will begin on
14
Table of Contents
time or be completed on schedule, if at all. The commencement of clinical trials can be delayed for a variety of reasons, including delays in:
| • | identifying and selecting a suitable development candidate. | |
| • | obtaining any required approvals from our collaborators, such as Novartis for any BCR-ABL product candidates that may be selected under our license and collaboration agreement with Novartis; | |
| • | obtaining regulatory approval to commence a clinical trial; | |
| • | reaching agreement on acceptable terms with prospective contract research organizations and trial sites; | |
| • | manufacturing sufficient quantities of a product candidate; | |
| • | obtaining institutional review board approval to conduct a clinical trial at a prospective site; and | |
| • | identifying, recruiting and enrolling patients to participate in a clinical trial. |
In addition, once a clinical trial has begun, patient recruitment and enrollment may be slower than we anticipate. For instance, for any BCR-ABL product candidates that may be selected for clinical development, we may have difficulty in recruiting a sufficient number of patients in an acceptable timeline due to the competing product candidates in clinical development and the relatively small number of patients available in the initial indication which we would expect to target. Further, as was the case with our pivotal Phase II/III clinical trial for the third-line treatment of AML that was terminated in August of 2006, a clinical trial may be suspended or terminated by us, our data and safety monitoring board, our collaborators, the FDA or other regulatory authorities due to a number of factors, including:
| • | failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols; | |
| • | inspection of the clinical trial operations or trial site by the FDA or other regulatory authorities resulting in the imposition of a clinical hold; | |
| • | unforeseen safety issues or insufficient efficacy; or | |
| • | lack of adequate funding to continue the clinical trial. |
If we experience delays in the completion of, or termination of, any clinical trial of a BCR-ABL product candidate that may be selected under our license and collaboration agreement with Novartis or any other product candidate we advance into clinical trials, the commercial prospects for product candidates we may develop will be harmed, and our ability to generate product revenues from any product candidate we may develop will be delayed. In addition, many of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of a product candidate. Even if we are able to ultimately commercialize product candidates, other therapies for the same indications may have been introduced to the market during the period we have been delayed and such therapies may have established a competitive advantage over our products.
We currently depend on one collaboration partner, Novartis, for a portion of our revenues and for the future development and potential commercialization of one of our significant research programs. If our license and collaboration agreement with Novartis terminates, our business and, in particular, ourBCR-ABL program, will be seriously harmed.
In March 2006, we entered into a license and collaboration agreement with Novartis to develop and commercialize BCR-ABL inhibitors for the treatment of CML. In connection with the license and collaboration agreement, Novartis paid us a non-refundable, non-creditable license fee of $20.0 million. Novartis Pharma AG also purchased $5.0 million of our common stock. Depending upon the success of our collaboration, we may derive a substantial portion of our near-term revenues from Novartis. After the end of the research term under the license and collaboration agreement, Novartis may terminate the license and collaboration agreement upon 60 days written notice if it determines that further development is not viable for competitive, safety, or efficacy reasons. If the license and collaboration agreement is terminated in whole or in part and we are unable to enter into similar arrangements with other collaborators, our business would be materially and adversely affected.
15
Table of Contents
The success of our BCR-ABL inhibitor program depends heavily on our collaboration with Novartis, which was established only recently. If Novartis is unwilling to further develop or commercialize development candidates we may identify under the collaboration, or experiences significant delays in doing so, our business may be harmed.
As Novartis is responsible for the clinical development of product candidates identified under the collaboration after the first Phase 1 clinical trial, the future success of our BCR-ABL program will depend in large part on our ability to maintain our relationship with Novartis with respect to product candidates licensed to Novartis under the collaboration agreement. To date, the nomination of a development candidate has taken longer than anticipated and we may experience further delays in the collaboration’s progress. It is possible that we and Novartis may never select a development candidate and commence clinical trials. We do not have a significant history of working together with Novartis and cannot predict the progress and success of the collaboration. While Novartis is subject to certain diligence obligations under the collaboration agreement, we cannot guarantee that Novartis will not reduce or curtail its efforts to develop product candidates we may identify under the collaboration, because of changes in its research and development budget, its internal development priorities, the success or failure of its other product candidates or other factors affecting its business or operations. For example, Novartis markets Gleevec® (imatinib mesylate) and has other drug candidates under development that could compete with any BCR-ABL inhibitor that we may develop under our collaboration with Novartis. It is possible that Novartis may devote greater resources to its other competing programs, or may not pursue as aggressively our BCR-ABL program or market as aggressively any BCR-ABL product that may result from our collaboration.
Any product candidates we advance into clinical trials are subject to extensive regulation, which can be costly and time consuming, cause unanticipated delays or prevent the receipt of the required approvals to commercialize our product candidates.
The clinical development, manufacturing, labeling, storage, record-keeping, advertising, promotion, export, marketing and distribution of any other product candidates we advance into clinical trials are subject to extensive regulation by the FDA in the United States and by comparable governmental authorities in foreign markets. In the United States, neither we nor our collaborators are permitted to market our product candidates until we or our collaborators receive approval of an NDA from the FDA. The process of obtaining NDA approval is expensive, often takes many years, and can vary substantially based upon the type, complexity and novelty of the products involved. Approval policies or regulations may change. In addition, as a company, we have not previously filed an NDA with the FDA. This lack of experience may impede our ability to obtain FDA approval in a timely manner, if at all, for our product candidates for which development and commercialization is our responsibility. Despite the time and expense invested, regulatory approval is never guaranteed. The FDA or any of the applicable European, Canadian or other regulatory bodies can delay, limit or deny approval of a product candidate for many reasons, including:
| • | a product candidate may not be safe and effective; | |
| • | regulatory agencies may not find the data from preclinical testing and clinical trials to be sufficient; | |
| • | regulatory agencies may not approve of our third party manufacturers’ processes or facilities; or | |
| • | regulatory agencies may change their approval policies or adopt new regulations. |
In addition, while we may seek to take advantage of various regulatory processes intended to accelerate drug development and approval for any product candidates that may be selected for clinical development from our BCR-ABL program, there is no guarantee that the FDA will review or accept an NDA under the accelerated approval regulations, based on our clinical trial design, the results of any clinical trials we may conduct or other factors.
Also, recent events implicating questions about the safety of marketed drugs, including those pertaining to the lack of adequate labeling, may result in increased cautiousness by the FDA in reviewing new drugs based on safety, efficacy or other regulatory considerations and may result in significant delays in obtaining regulatory approvals. Any delay in obtaining, or inability to obtain, applicable regulatory approvals would prevent us from commercializing our product candidates.
16
Table of Contents
Any product candidate we advance into clinical trials may cause undesirable side effects that could delay or prevent its regulatory approval or commercialization.
Undesirable side effects caused by any product candidate we advance into clinical trials could interrupt, delay or halt clinical trials and could result in the denial of regulatory approval by the FDA or other regulatory authorities for any or all targeted indications. This, in turn, could prevent us from commercializing product candidates we advance into clinical trials and generating revenues from its sale. In addition, if any product candidate receives marketing approval and we or others later identify undesirable side effects caused by the product:
| • | regulatory authorities may withdraw their approval of the product; | |
| • | we may be required to change the way the product is administered, conduct additional clinical trials or change the labeling of the product; or | |
| • | our reputation may suffer. |
Any one or a combination of these events could prevent us from achieving or maintaining market acceptance of the affected product or could substantially increase the costs and expenses of commercializing the product, which in turn could delay or prevent us from generating significant revenues from the sale of the product.
We rely on third parties to conduct our clinical trials. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for or commercialize our product candidates.
We intend to rely on third parties, such as contract research organizations, medical institutions, clinical investigators and contract laboratories, to conduct all or a portion of any future clinical trials. We may not be able to control the amount and timing of resources that third parties devote to any clinical trials we may commence or the quality or timeliness of the services performed by such third parties. In any future clinical trials, in the event that we are unable to maintain our relationship with any clinical trial sites, or elect to terminate the participation of any clinical trial sites, we may experience the loss offollow-up information on patients enrolled in such clinical trial unless we are able to transfer the care of those patients to another qualified clinical trial site. In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and receive cash or equity compensation in connection with such services. If these relationships and any related compensation result in perceived or actual conflicts of interest, the integrity of the data generated at the applicable clinical trial site may be jeopardized. If these third parties do not successfully carry out their contractual duties or obligations or meet expected deadlines in connection with any future clinical trials, or if the quality or accuracy of the clinical data is compromised due to the failure to adhere to clinical protocols or for other reasons, our clinical trials may be extended, delayed or terminated, our reputation in the industry and in the investment community may be significantly damaged and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
Even if any product candidate we advance into clinical trials receives regulatory approval, our product candidates may still face future development and regulatory difficulties.
If any product candidate we advance into clinical trials receives U.S. regulatory approval, the FDA may still impose significant restrictions on the indicated uses or marketing of the product candidate or impose ongoing requirements for potentially costly post-approval studies. In addition, regulatory agencies subject a product, its manufacturer and the manufacturer’s facilities to continual review and periodic inspections. If a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions on that product, our collaborators or us, including requiring withdrawal of the product from the market. Our product candidates will also be subject to ongoing FDA requirements for the labeling, packaging, storage, advertising, promotion, record-keeping and submission of safety and other post-market information on the drug. If our product candidates fail to comply with applicable regulatory requirements, a regulatory agency may:
| • | issue warning letters; | |
| • | impose civil or criminal penalties; |
17
Table of Contents
| • | withdraw regulatory approval; | |
| • | suspend any ongoing clinical trials; | |
| • | refuse to approve pending applications or supplements to approved applications filed by us or our collaborators; | |
| • | impose restrictions on operations, including costly new manufacturing requirements; or | |
| • | seize or detain products or require a product recall. |
Moreover, in order to market any products outside of the United States, we and our collaborators must establish and comply with numerous and varying regulatory requirements of other countries regarding safety and efficacy. Approval procedures vary among countries and can involve additional product testing and additional administrative review periods. The time required to obtain approval in other countries might differ from that required to obtain FDA approval. The regulatory approval process in other countries may include all of the risks described above regarding FDA approval in the United States. Regulatory approval in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country may negatively impact the regulatory process in others. Failure to obtain regulatory approval in other countries or any delay or setback in obtaining such approval could have the same adverse effects detailed above regarding FDA approval in the United States. As described above, such effects include the risk that our product candidates may not be approved for all indications requested, which could limit the uses of our product candidates and adversely impact potential royalties and product sales, and that such approval may be subject to limitations on the indicated uses for which the product may be marketed or require costly, post-marketingfollow-up studies. If we or our collaborators fail to comply with applicable domestic or foreign regulatory requirements, we and our collaborators may be subject to fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
We are dependent on our collaborations, and events involving these collaborations or any future collaborations could prevent us from developing or commercializing product candidates.
The success of our current business strategy and our near and long-term viability will depend in part on our ability to successfully establish new strategic collaborations. Since we do not currently possess the resources necessary to independently develop and commercialize all of the product candidates that may be discovered through our drug discovery platform, we may need to enter into additional collaborative agreements to assist in the development and commercialization of some of these product candidates or in certain markets for a particular product candidate. Establishing strategic collaborations is difficult and time-consuming. Potential collaborators may reject collaborations based upon their assessment of our financial, regulatory or intellectual property position. And our discussions with potential collaborators may not lead to the establishment of new collaborations on acceptable terms. In addition, if as a result of our financial condition or other factors we enter into a strategic collaboration while a drug candidate program is in early preclinical development, we may not generate as much near- or longer-term revenue from such program as we could have generated if we had the resources to further independently develop such program.
We have entered into drug discovery collaborations, such as those with Novartis and the Cystic Fibrosis Foundation. In each case, our collaborators have agreed to finance the clinical trials for product candidates resulting from these collaborations and, if they are approved, manufacture and market them. Accordingly, we are dependent on our collaborators to gain regulatory approval of, and to commercialize, product candidates resulting from most of our collaborations.
We have limited control over the amount and timing of resources that our current collaborators or any future collaborators (including collaborators resulting from a change of control) devote to our programs or potential products. In some instances, our collaborators, such as Novartis, may have competing internal programs or programs with other parties, and such collaborators may devote greater resources to their internal or other programs than to our collaboration and any product candidates developed under our collaboration. Our collaborators may prioritize other drug development opportunities that they believe may have a higher likelihood of obtaining regulatory approval or may potentially generate a greater return on investment. These collaborators may breach or
18
Table of Contents
terminate their agreements with us or otherwise fail to conduct their collaborative activities successfully and in a timely manner. Further, our collaborators may not develop products that arise out of our collaborative arrangements or devote sufficient resources to the development, manufacture, marketing or sale of these products. Moreover, in the event of termination of a collaboration agreement, termination negotiations may result in less favorable terms than we would otherwise choose.
We and our present and future collaborators may fail to develop or effectively commercialize products covered by our present and future collaborations if:
| • | we do not achieve our objectives under our collaboration agreements; | |
| • | we or our collaborators are unable to obtain patent protection for the product candidates or proprietary technologies we discover in our collaborations; | |
| • | we are unable to manage multiple simultaneous product discovery and development collaborations; | |
| • | our potential collaborators are less willing to expend their resources on our programs due to their focus on other programs or as a result of general market conditions; | |
| • | our collaborators become competitors of ours or enter into agreements with our competitors; | |
| • | we or our collaborators encounter regulatory hurdles that prevent the further development or commercialization of our product candidates; or | |
| • | we develop products and processes or enter into additional collaborations that conflict with the business objectives of our other collaborators. |
If we or our collaborators are unable to develop or commercialize products as a result of the occurrence of any one or a combination of these events, we will be prevented from developing and commercializing product candidates.
Conflicts may arise between us and our collaborators that could delay or prevent the development or commercialization of our product candidates.
Conflicts may arise between our collaborators and us, such as conflicts concerning which compounds, if any, to select for pre-clinical or clinical development, the interpretation of clinical data, the achievement of milestones, the interpretation of financial provisions or the ownership of intellectual property developed during the collaboration. If any conflicts arise with existing or future collaborators, they may act in their self-interest, which may be adverse to our best interests. Any such disagreement between us and a collaborator could result in one or more of the following, each of which could delay or prevent the development or commercialization of our product candidates, and in turn prevent us from generating sufficient revenues to achieve or maintain profitability:
| • | disagreements regarding the payment of research funding, milestone payments, royalties or other payments we believe are due to us under our collaboration agreements or from us under our licensing agreements; | |
| • | uncertainty regarding ownership of intellectual property rights arising from our collaborative activities, which could prevent us from entering into additional collaborations; | |
| • | actions taken by a collaborator inside or outside a collaboration which could negatively impact our rights under or benefits from such collaboration; | |
| • | unwillingness on the part of a collaborator to keep us informed regarding the progress of its development and commercialization activities or to permit public disclosure of the results of those activities; or | |
| • | slowing or cessation of a collaborator’s development or commercialization efforts with respect to our product candidates. |
19
Table of Contents
If we fail to establish new collaborations and other commercial agreements, we may have to reduce or limit our internal drug discovery and development efforts and our business may be adversely affected.
Revenue generation utilizing compounds identified by us from the application of our technologies, such as the development candidate identified in our MET program, and our FAST drug discovery platform and related technologies, is important to us to provide us with funds for reinvestment in our internal drug discovery and development programs. If we fail to establish additional collaborations, commercial agreements or out-licensing arrangements on acceptable terms, we may not generate sufficient revenue to support our internal discovery and development efforts. In addition, since our existing collaborations and commercial agreements are generally not long-term contracts, we cannot be sure we will be able to continue to derive comparable revenues from these or other collaborations or commercial agreements in the future. Even if we successfully establish collaborations, these relationships may never result in the successful development or commercialization of any product candidates or the generation of sales or royalty revenue. Under our commercial arrangements with other pharmaceutical and biotechnology companies, such as under all of our beamline services agreements, we are providing specific services for fees without any interest in future product sales or profits. While we believe these commercial arrangements help to offset the expenses associated with our drug discovery efforts, we may under some circumstances find it necessary to divert valuable resources from our own discovery efforts in order to fulfill our contractual obligations.
Our drug discovery efforts are dependent on continued access to and use of our beamline facility, which is subject to various governmental regulations and policies and a user agreement with the University of Chicago and the U.S. Department of Energy. If we are unable to continue the use of our beamline facility, we may be required to delay, reduce the scope of or abandon some of our drug discovery efforts, and may fail to perform under our collaborations, commercial agreements and grants, which would result in a material reduction in our current primary source of revenue.
We generate protein structures through our beamline facility, housed at the Advanced Photon Source at the Argonne National Laboratory, a national synchrotron-radiation facility funded by the U.S. Department of Energy, Office of Science, and Office of Basic Energy Sciences, located in Argonne, Illinois. Accordingly, our access to and use of the facility is subject to various government regulations and policies. In addition, our access to the beamline facility is subject to a user agreement with the University of Chicago and the U.S. Department of Energy with an initial five year term expiring in January 1, 2009. Although the term of our user agreement automatically renews for successive one-year periods, the University of Chicago may terminate the agreement and our access to the beamline facility by providing 60 days’ notice prior to the beginning of each renewal period. In addition, the University of Chicago may terminate the agreement for our breach, subject to our ability to cure the breach within 30 days. In the event our access to or use of the facility is restricted or terminated, we would be forced to seek access to alternate beamline facilities. There are currently only three alternate beamline facilities in the U.S. and two outside the U.S., which are comparable to ours. To obtain equivalent access at a single alternate beamline facility would likely require us building out a new beamline at such facility which could take over two years and would involve significant expense. However, we cannot be certain that we would be able to obtain equivalent access to such a facility on acceptable terms or at all. In the interim period, we would have to obtain beamline access at a combination of facilities, and there is no guarantee that we would be able to obtain sufficient access time on acceptable terms or at all. If alternate beamline facilities are not available, we may be required to delay, reduce the scope of or abandon some of our early drug discovery efforts. We may also be deemed to be in breach of certain of our commercial agreements. Even if alternate beamline facilities are available, we cannot be certain that the quality of or access to the alternate facilities will be adequate and comparable to those of our current facility. Failure to maintain adequate access to and use of beamline facilities may materially adversely affect our ability to pursue our own discovery efforts and perform under our collaborations, commercial agreements and grants, which are our current primary source of revenue.
If our competitors develop drug discovery technologies that are more advanced than ours, our ability to generate revenue from collaborations, commercial arrangements or grants may be reduced or eliminated.
The biotechnology and biopharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We face competition from many different
20
Table of Contents
sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies, and private and public research institutions. There is also intense competition for fragment-based lead discovery collaborations. In addition, we understand that many large pharmaceutical companies are exploring the internal development of fragment-based drug discovery methods. Additionally, due to the high demand for treatments for CML and other oncology therapeutic areas, research is intense and new technologies to enhance the rapid discovery and development of potential treatments are being sought out and developed by our competitors. If our competitors develop drug discovery technologies that are more advanced or more cost efficient or effective than ours, our revenue from collaborations, commercial arrangements and grants may be substantially reduced or eliminated.
If our competitors develop treatments for CML or any other therapeutic area that are approved more quickly, marketed more effectively or demonstrated to be more effective than our current or future product candidates, our ability to generate product revenue will be reduced or eliminated.
Most cancer indications for which we are developing products have a number of established therapies with which our candidates will compete. Most major pharmaceutical companies and many biotechnology companies are aggressively pursuing new cancer development programs, including both therapies with traditional as well as novel mechanisms of action.
We are aware of competitive products in each of the markets we target. These competitive products include approved and marketed products as well as products in development. We expect that any BCR-ABL inhibitor that we may potentially develop for treatment of CML to compete with Gleevec® (imatinib), marketed by Novartis, Inc., Tasigna® (nilotinib), for which an NDA was recently filed by Novartis, Inc., Sprycel® (dasatinib), marketed by Bristol Myers Squibb, Inc., MK-0457, under development by Merck and Co., SKI-606, under development by Wyeth, Inc., INNO-406, under development by Innovive Pharmaceuticals, Inc., homoharringtonine, under development by ChemGenex, Inc., KW-2449, under development by Kyowa Pharma, Inc., and XL228, under development by Exelixis, Inc. Other potential competing products are in clinical trials and preclinical development.
We expect that any MET inhibitor that we may potentially develop for treatment of cancers, to compete with XL880 and XL184, under development by Exelixis, Inc., ARQ197, under development by Arqule, Inc., PF02341066, under development by Pfizer, Inc., AMG102, under development by Amgen, Inc., MP470, under development by SuperGen, Inc., and MGCD265, under development by Methylgene Inc. Other potential competing products are in clinical trials and preclinical development.
Significant competitors in the area of fragment-based drug discovery include Astex Therapeutics Limited, Plexxikon Inc., Evotec AG, Vernalis Plc., Sunesis Pharmaceuticals, Inc., and Active Site, Inc. In addition, many large pharmaceutical companies are exploring the internal development of fragment-based drug discovery methods.
Many of our competitors have significantly greater financial, product development, manufacturing and marketing resources than us. Large pharmaceutical companies have extensive experience in clinical testing and obtaining regulatory approval for drugs. These companies also have significantly greater research capabilities than us. In addition, many universities and private and public research institutes are active in cancer research, some in direct competition with us. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
Our competitors may succeed in developing products for the treatment of CML or other diseases in oncology therapeutic areas in which our drug discovery programs are focused that are more effective, better tolerated or less costly than any which we may offer or develop. Our competitors may succeed in obtaining approvals from the FDA and foreign regulatory authorities for their product candidates sooner than we do for ours. We will also face competition from these third parties in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, and in acquiring and in-licensing technologies and products complementary to our programs or advantageous to our business.
21
Table of Contents
We have limited experience in identifying, acquiring or in-licensing, and integrating third parties’ products, businesses and technologies into our current infrastructure. If we determine that future acquisition, in-licensing or other strategic opportunities are desirable and do not successfully execute on and integrate such targets, we may incur costs and disruptions to our business.
An important part of our business strategy is to continue to develop a broad pipeline of product candidates. These efforts include potential licensing and acquisition transactions. For example, our product candidate, Troxatyl, was initially developed by Shire and licensed to us in July 2004. Although we are not currently a party to any other agreements or commitments other than our agreement with Shire, we may, particularly given the negative outcome of the Phase II/III clinical trial for Troxatyl, seek to expand our product pipeline and technologies, at the appropriate time and as resources allow, by acquiring or in-licensing products, or combining with businesses that we believe are a strategic fit with our business and complement our existing internal drug development efforts and product candidates, research programs and technologies. Future transactions, however, may entail numerous operational and financial risks including:
| • | exposure to unknown liabilities; | |
| • | disruption of our business and diversion of our management’s time and attention to the development of acquired products or technologies; | |
| • | incurrence of substantial debt or dilutive issuances of securities to pay for acquisitions; | |
| • | dilution to existing stockholders in the event of an acquisition by another entity; | |
| • | higher than expected acquisition and integration costs; | |
| • | increased amortization expenses; | |
| • | difficulties in and costs of combining the operations and personnel of any businesses with our operations and personnel; | |
| • | impairment of relationships with key suppliers or customers of any acquired businesses due to changes in management and ownership; and | |
| • | inability to retain key employees. |
Finally, we may devote resources to potential in-licensing opportunities or strategic transactions that are never completed or fail to realize the anticipated benefits of such efforts.
Because we exclusively licensed our product candidate, Troxatyl, from Shire and our rights are subject to certain licenses to Shire from third parties, any dispute with Shire or between Shire and any of these third parties may adversely affect our business.
In late July 2004, we licensed exclusive worldwide rights to our product candidate, Troxatyl, from Shire. We have not yet agreed with Shire, the future plans, if any, for Troxatyl. If there is any dispute between us and Shire regarding our rights or obligations under the license agreement, including diligence obligations, the achievement of milestones or interpretation of financial or other provisions, we risk litigation and our business may be adversely affected. In addition, our exclusive license to Troxatyl is subject to the terms and conditions of a license from Yale University and the University of Georgia Research Foundation, Inc. to Shire. If Shire breaches the terms or conditions of any of these underlying licenses to Shire or otherwise is engaged in a dispute with any of these third party licensors, such breaches by Shire or disputes with Shire could result in a loss of, or other material adverse impact on, our rights under our exclusive license agreement with Shire.
We do not have internal manufacturing capabilities, and if we fail to develop and maintain supply relationships with collaborators or other third party manufacturers, we may be unable to develop or commercialize our products.
All of our manufacturing is outsourced to third parties with oversight by our internal managers. We intend to continue this practice of outsourcing our manufacturing services to third parties for any future clinical trials we may
22
Table of Contents
conduct and for commercialization of any other product candidate we advance into clinical trials. Our ability to develop and commercialize products depends in part on our ability to arrange for collaborators or other third parties to manufacture our products at a competitive cost, in accordance with regulatory requirements and in sufficient quantities for clinical testing and eventual commercialization.
If we are unable to establish sales and marketing capabilities or enter into agreements with third parties to market and sell any products we may develop, we may not be able to generate product revenue.
We do not currently have a sales organization for the sales, marketing and distribution of pharmaceutical products. In order to commercialize any products, we must build our sales, marketing, distribution, managerial and other non-technical capabilities or make arrangements with third parties to perform these services. In North America, we currently expect to commercialize any BCR-ABL product candidates that may result from our collaboration with Novartis, and certain other potential product candidates for other indications that are of strategic interest to us, and plan to establish internal sales and marketing capabilities for those product candidates. We plan to seek third party partners for indications and in territories, such as outside North America, which may require more extensive sales and marketing capabilities. The establishment and development of our own sales force to market any products we may develop in North America will be expensive and time consuming and could delay any product launch, and we cannot be certain that we would be able to successfully develop this capacity. If we are unable to establish our sales and marketing capability or any other non-technical capabilities necessary to commercialize any products we may develop, we will need to contract with third parties to market and sell any products we may develop in North America. If we are unable to establish adequate sales, marketing and distribution capabilities, whether independently or with third parties, we may not be able to generate product revenue and may not become profitable.
The commercial success of any product that we may develop depends upon market acceptance among physicians, patients, health care payors and the medical community.
Even if any product we may develop obtains regulatory approval, our products, if any, may not gain market acceptance among physicians, patients, health care payors and the medical community. The degree of market acceptance of any of our approved products will depend on a number of factors, including:
| • | our ability to provide acceptable evidence of safety and efficacy; | |
| • | relative convenience and ease of administration; | |
| • | the prevalence and severity of any adverse side effects; | |
| • | availability of alternative treatments; | |
| • | pricing and cost effectiveness; | |
| • | effectiveness of our or our collaborators’ sales and marketing strategies; and | |
| • | our ability to obtain sufficient third party coverage or reimbursement. |
If any of our product candidates is approved, but does not achieve an adequate level of acceptance by physicians, healthcare payors and patients, we may not generate sufficient revenue from these products and we may not become profitable.
We are subject to uncertainty relating to health care reform measures and reimbursement policies which, if not favorable to our product candidates, could hinder or prevent our product candidates’ commercial success.
The continuing efforts of the government, insurance companies, managed care organizations and other payors of health care costs to contain or reduce costs of health care may adversely affect one or more of the following:
| • | our ability to set a price we believe is fair for our products; | |
| • | our ability to generate revenues and achieve profitability; |
23
Table of Contents
| • | the future revenues and profitability of our potential customers, suppliers and collaborators; and | |
| • | the availability of capital. |
In certain foreign markets, the pricing of prescription drugs is subject to government control and reimbursement may in some cases be unavailable. In the United States, given recent federal and state government initiatives directed at lowering the total cost of health care, Congress and state legislatures will likely continue to focus on health care reform, the cost of prescription drugs and the reform of the Medicare and Medicaid systems. For example, the Medicare Prescription Drug, Improvement and Modernization Act of 2003 provided a new Medicare prescription drug benefit beginning in 2006 and mandates other reforms. We are not yet able to assess the full impact of this legislation and it is possible that the new Medicare prescription drug benefit, which will be managed by private health insurers and other managed care organizations, will result in decreased reimbursement for prescription drugs, which may further exacerbate industry-wide pressure to reduce prescription drug prices. This could harm our ability to market our products and generate revenues. It is also possible that other proposals having a similar effect will be adopted.
Our ability to commercialize successfully any product candidates we advance into clinical trials will depend in part on the extent to which governmental authorities, private health insurers and other organizations establish appropriate coverage and reimbursement levels for the cost of our products and related treatments. Third party payors are increasingly challenging the prices charged for medical products and services. Also, the trend toward managed health care in the United States, which could significantly influence the purchase of health care services and products, as well as legislative proposals to reform health care or reduce government insurance programs, may result in lower prices for our product candidates or exclusion of our product candidates from coverage and reimbursement programs. The cost containment measures that health care payors and providers are instituting and the effect of any health care reform could significantly reduce our revenues from the sale of any approved product.
In the event that we determine that we need to increase the size of our organization, we may experience difficulties in managing growth.
As of December 31, 2006, we had 124 full-time employees. In the future, we may need to expand our managerial, operational, financial and other resources in order to manage and fund our operations, continue our research and development and collaborative activities, progress our product candidates through clinical development and eventually commercialize any product candidates for which we are able to obtain regulatory approval. It is possible that our management and scientific personnel, systems and facilities currently in place may not be adequate to support this future growth. Our need to effectively manage our operations, growth and various projects requires that we:
| • | manage our internal research and development efforts effectively while carrying out our contractual obligations to collaborators and other third-parties; | |
| • | continue to improve our operational, financial and management controls, reporting systems and procedures; | |
| • | set up marketing, sales, distribution and other commercial operations infrastructure if any of our product candidates obtain regulatory approval; and | |
| • | attract and retain sufficient numbers of talented employees. |
We may be unable to successfully implement these tasks on a larger scale and, accordingly, may not achieve our research, development and commercialization goals.
If we fail to attract and keep key management and scientific personnel, we may be unable to successfully develop or commercialize our product candidates.
We will need to expand and effectively manage our managerial, operational, financial and other resources in order to successfully pursue our research, development and commercialization efforts for any future product candidates.
24
Table of Contents
Our success depends on our continued ability to attract, retain and motivate highly qualified management and chemists, biologists, and preclinical and clinical personnel. The loss of the services of any of our senior management, particularly Michael Grey, our President and Chief Executive Officer, or Stephen Burley, our Chief Scientific Officer and Senior Vice President, Research, could delay or prevent the clinical development and potential commercialization of our product candidates. We do not maintain “key man” insurance policies on the lives of these individuals or the lives of any of our other employees. We employ these individuals on an at-will basis and their employment can be terminated by us or them at any time, for any reason and with or without notice. We have scientific and clinical advisors who assist us in formulating our research, development and clinical strategies. These advisors are not our employees and may have commitments to, or consulting or advisory contracts with, other entities that may limit their availability to us. In addition, our advisors may have arrangements with other companies to assist those companies in developing products or technologies that may compete with ours.
We may not be able to attract or retain qualified management and scientific personnel in the future due to the intense competition for qualified personnel among biotechnology, pharmaceutical and other businesses, particularly in the San Diego, California area. If we are not able to attract and retain the necessary personnel to accomplish our business objectives, we may experience constraints that will impede significantly the achievement of our research and development objectives, our ability to raise additional capital and our ability to implement our business strategy. In particular, if we lose any members of our senior management team, we may not be able to find suitable replacements and our business may be harmed as a result.
Risks Relating to our Finances and Capital Requirements
We expect our net operating losses to continue for at least several years, and we are unable to predict the extent of future losses or when we will become profitable, if ever.
We have incurred substantial net operating losses since our inception. For the years ended December 31, 2006 and 2005, we had a net loss attributable to common stockholders of $28.1 million and $29.9 million, respectively. As of December 31, 2006, we had an accumulated deficit of approximately $163.7 million. We expect our annual net operating losses to continue over the next several years as we conduct our research and development activities, and incur preclinical and clinical development costs. Because of the numerous risks and uncertainties associated with our research and development efforts and other factors, we are unable to predict the extent of any future losses or when we will become profitable, if ever. We will need to commence clinical trials, obtain regulatory approval and successfully commercialize a product candidate or product candidates before we can generate revenues which would have the potential to lead to profitability.
We currently lack a significant continuing revenue source and may not become profitable.
Our ability to become profitable depends upon our ability to generate significant continuing revenues. To obtain significant continuing revenues, we must succeed, either alone or with others, in developing, obtaining regulatory approval for, and manufacturing and marketing product candidates with significant market potential. However, we cannot guarantee when, if ever, products resulting from our BCR-ABL, MET or JAK2 programs will generate product sales. We had revenues from collaborations, commercial agreements and grants totaling $27.8 million, $21.6 million, and 27.3 million for years ended December 31, 2006, 2005, and 2004, respectively. Though we anticipate that our collaborations, commercial agreements and grants will continue to be our primary sources of revenues for the next several years, these revenues alone will not be sufficient to lead to profitability.
Our ability to generate continuing revenues depends on a number of factors, including:
| • | obtaining new collaborations and commercial agreements; | |
| • | performing under current and future collaborations, commercial agreements and grants, including achieving milestones; | |
| • | successful completion of clinical trials for any product candidate we advance into clinical trials; | |
| • | achievement of regulatory approval for any product candidate we advance into clinical trials; and | |
| • | successful sales, manufacturing, distribution and marketing of our future products, if any. |
25
Table of Contents
If we are unable to generate significant continuing revenues, we will not become profitable, and we may be unable to continue our operations.
We will need substantial additional funding and may be unable to raise capital when needed, which would force us to delay, reduce or eliminate our research and development programs or commercialization efforts.
We believe that our existing cash and cash equivalents and short-term investments, together with interest thereon and cash from existing collaborations, commercial agreements and grants, will be sufficient to meet projected operating requirements through 2008. Consistent with our existing business development strategy, we anticipate establishing new collaborations and commercial agreements. Any proceeds received in connection with such new transactions, or milestones achieved under existing or new collaborations or commercial agreements, would provide additional operating capital. However, if we do not generate additional revenue from collaborations, commercial agreements and grants at the levels we project, we may require additional funding sooner than we currently anticipate. Because we do not anticipate that we will generate significant continuing revenues for several years, if at all, we will need to raise substantial additional capital to finance our operations in the future. Our additional funding requirements will depend on, and could increase significantly as a result of, many factors, including the:
| • | terms and timing of any collaborative, licensing and other arrangements that we may establish, including by partnering our internal discovery programs, such as MET and JAK2; | |
| • | rate of progress and cost of our preclinical studies and clinical trials, if any, and other research and development activities; | |
| • | scope, prioritization and number of clinical development and research programs we pursue; | |
| • | costs and timing of preparing regulatory submissions and obtaining regulatory approval; | |
| • | costs of establishing or contracting for sales and marketing capabilities; | |
| • | costs of manufacturing; | |
| • | extent to which we acquire or in-license new products, technologies or businesses; | |
| • | effect of competing technological and market developments; and | |
| • | costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights. |
Until we can generate significant continuing revenues, if ever, we expect to satisfy our future cash needs through public or private equity offerings, debt financings, strategic transactions, or collaborations, commercial agreements and grants. We cannot be certain that additional funding will be available on acceptable terms, or at all. In addition, if our stock price declines for any reasonand/or we are unable to obtain additional funding, we may fail to satisfy the NASDAQ Global Market listing requirements. If we fail to meet the NASDAQ listing requirements and NASDAQ determines to delist our common stock, the delisting could adversely affect the market liquidity of our common stock and the market price of our common stock could decrease. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate one or more of our research and development programs or our commercialization efforts.
Raising additional funds by issuing securities or through licensing arrangements may cause dilution to existing stockholders, restrict our operations or require us to relinquish proprietary rights.
We may raise additional funds through public or private equity offerings, debt financings or licensing arrangements. To the extent that we raise additional capital by issuing equity securities, our existing stockholders’ ownership will be diluted. Any debt financing we enter into may involve covenants that restrict our operations. These restrictive covenants may include limitations on additional borrowing, specific restrictions on the use of our assets as well as prohibitions on our ability to create liens, pay dividends, redeem our stock or make investments. In addition, if we raise additional funds through licensing arrangements, as we did in our recent collaboration with Novartis, it may be necessary to relinquish potentially valuable rights to our potential products or proprietary technologies, or grant licenses on terms that are not favorable to us.
26
Table of Contents
Our quarterly operating results may fluctuate significantly.
We expect our operating results to be subject to quarterly fluctuations. The revenues we generate, if any, and our operating results will be affected by numerous factors, including:
| • | our addition or termination of research programs or funding support; | |
| • | variations in the level of expenses related to our product candidates or research programs; | |
| • | our execution of collaborative, licensing or other arrangements, and the timing of payments we may make or receive under these arrangements; | |
| • | any intellectual property infringement lawsuit in which we may become involved; and | |
| • | changes in accounting principles. |
Quarterly fluctuations in our operating results may, in turn, cause the price of our stock to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
We may incur substantial liabilities from any product liability claims if our insurance coverage for those claims is inadequate.
We face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials, and will face an even greater risk if we sell our product candidates commercially. An individual may bring a liability claim against us if one of our product candidates causes, or merely appears to have caused, an injury. If we cannot successfully defend ourselves against the product liability claim, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in any one or a combination of the following:
| • | decreased demand for our product candidates; | |
| • | injury to our reputation; | |
| • | withdrawal of clinical trial participants; | |
| • | costs of related litigation; | |
| • | substantial monetary awards to patients or other claimants; | |
| • | loss of revenues; and | |
| • | the inability to commercialize our product candidates. |
We have product liability insurance that covered our Troxatyl clinical trials. We intend to expand our insurance coverage to include the sale of commercial products if marketing approval is obtained for any of our product candidates. However, insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost and we may not be able to obtain insurance coverage that will be adequate to satisfy any liability that may arise.
We use biological and hazardous materials, and any claims relating to improper handling, storage or disposal of these materials could be time consuming or costly.
We use hazardous materials, including chemicals, biological agents and radioactive isotopes and compounds, which could be dangerous to human health and safety or the environment. Our operations also produce hazardous waste products. Federal, state and local laws and regulations govern the use, generation, manufacture, storage, handling and disposal of these materials and wastes. Compliance with applicable environmental laws and regulations may be expensive, and current or future environmental laws and regulations may impair our drug development efforts.
In addition, we cannot entirely eliminate the risk of accidental injury or contamination from these materials or wastes. If one of our employees was accidentally injured from the use, storage, handling or disposal of these materials or wastes, the medical costs related to his or her treatment would be covered by our workers’
27
Table of Contents
compensation insurance policy. However, we do not carry specific biological or hazardous waste insurance coverage and our property and casualty and general liability insurance policies specifically exclude coverage for damages and fines arising from biological or hazardous waste exposure or contamination. Accordingly, in the event of contamination or injury, we could be held liable for damages or penalized with fines in an amount exceeding our resources, and our clinical trials or regulatory approvals could be suspended.
Risks Relating to our Intellectual Property
Our success depends upon our ability to protect our intellectual property and our proprietary technologies.
Our commercial success depends on obtaining and maintaining patent protection and trade secret protection for our product candidates, proprietary technologies and their uses, as well as successfully defending these patents against third party challenges. There can be no assurance that our patent applications will result in patents being issued or that issued patents will afford protection against competitors with similar technology, nor can there be any assurance that the patents issued will not be infringed, designed around, or invalidated by third parties. Even issued patents may later be found unenforceable, or be modified or revoked in proceedings instituted by third parties before various patent offices or in courts.
The patent positions of pharmaceutical and biotechnology companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. Changes in either the patent laws or in the interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or in third party patents.
The degree of future protection for our proprietary rights is uncertain. Only limited protection may be available and may not adequately protect our rights or permit us to gain or keep any competitive advantage. For example:
| • | we might not have been the first to file patent applications for these inventions; | |
| • | we might not have been the first to make the inventions covered by each of our pending patent applications and issued patents; | |
| • | others may independently develop similar or alternative technologies or duplicate any of our technologies; | |
| • | the patents of others may have an adverse effect on our business; | |
| • | it is possible that none of our pending patent applications will result in issued patents; | |
| • | our issued patents may not encompass commercially viable products, may not provide us with any competitive advantages, or may be challenged by third parties; | |
| • | our issued patents may not be valid or enforceable; or | |
| • | we may not develop additional proprietary technologies that are patentable. |
Patent applications in the U.S. are maintained in confidence for up to 18 months after their filing. Consequently, we cannot be certain that we were the first to invent, or the first to file, patent applications on our compounds or drug candidates. We may not have identified all U.S. and foreign patents or published applications that may affect our business by blocking our ability to commercialize any drugs for which we are able to successfully develop and obtain regulatory approval.
Proprietary trade secrets and unpatented know-how are also very important to our business. Although we have taken steps to protect our trade secrets and unpatented know-how, including entering into confidentiality agreements with third parties, and confidential information and inventions agreements with employees, consultants and advisors, third parties may still obtain this information or we may be unable to protect our rights. Enforcing a claim that a third party illegally obtained and is using our trade secrets or unpatented know-how is expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States may be less willing to protect trade secret information. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how, and we would not be able to prevent their use.
28
Table of Contents
If we are sued for infringing intellectual property rights of third parties, it will be costly and time consuming, and an unfavorable outcome in that litigation would have a material adverse effect on our business.
Our commercial success also depends upon our ability and the ability of our collaborators to develop, manufacture, market and sell our product candidates and use our proprietary technologies without infringing the proprietary rights of third parties. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we and our collaborators are developing products. Because patent applications can take many years to issue, there may be currently pending applications which may later result in issued patents that our product candidates or proprietary technologies may infringe.
We may be exposed to, or threatened with, future litigation by third parties having patent, trademark or other intellectual property rights alleging that we are infringing their intellectual property rights. If one of these patents was found to cover our product candidates, research methods, proprietary technologies or their uses, or one of these trademarks was found to be infringed, we or our collaborators could be required to pay damages and could be unable to commercialize our product candidates or use our proprietary technologies unless we or they obtain a license to the patent or trademark, as applicable. A license may not be available to us or our collaborators on acceptable terms, if at all. In addition, during litigation, the patent or trademark holder could obtain a preliminary injunction or other equitable right which could prohibit us from making, using or selling our products, technologies or methods. In addition, we or our collaborators could be required to designate a different trademark name for our products, which could result in a delay in selling those products.
There is a substantial amount of litigation involving patent and other intellectual property rights in the biotechnology and biopharmaceutical industries generally. If a third party claims that we or our collaborators infringe its intellectual property rights, we may face a number of issues, including, but not limited to:
| • | infringement and other intellectual property claims which, with or without merit, may be expensive and time-consuming to litigate and may divert our management’s attention from our core business; | |
| • | substantial damages for infringement, including treble damages and attorneys’ fees, which we may have to pay if a court decides that the product or proprietary technology at issue infringes on or violates the third party’s rights; | |
| • | a court prohibiting us from selling or licensing the product or using the proprietary technology unless the third party licenses its technology to us, which it is not required to do; | |
| • | if a license is available from the third party, we may have to pay substantial royalties, feesand/or grant cross licenses to our technology; and | |
| • | redesigning our products or processes so they do not infringe, which may not be possible or may require substantial funds and time. |
There can be no assurance that third party patents containing claims covering our product candidates, technology or methods do not exist, have not been filed, or could not be filed or issued. Because of the number of patents issued and patent applications filed in our areas or fields of interest, particularly in the area of protein kinase inhibitors, we believe there is a significant risk that third parties may allege they have patent rights encompassing our product candidates, technology or methods. In addition, we have not conducted an extensive search of third party trademarks, so no assurance can be given that such third party trademarks do not exist, have not been filed, could not be filed or issued, or could not exist under common trademark law.
Other product candidates that we may develop, either internally or in collaboration with others, could be subject to similar risks and uncertainties.
Risks Relating to the Securities Markets and Ownership of our Common Stock
Market volatility may affect our stock price.
Until our initial public offering in February 2006, there was no market for our common stock, and despite our initial public offering, an active public market for these shares may not develop or be sustained. We have had
29
Table of Contents
relatively low volume of trading in our stock since our initial public offering and we do not know if the trading volume of our common stock will increase in the future. In addition, the market price of our common stock may fluctuate significantly in response to a number of factors, most of which we cannot control, including:
| • | changes in the preclinical or clinical development status of or clinical trial results for our product candidates; | |
| • | announcements of new products or technologies, commercial relationships or collaboration arrangements or other events by us or our competitors; | |
| • | events affecting our collaborations, commercial agreements and grants; | |
| • | variations in our quarterly operating results; | |
| • | changes in securities analysts’ estimates of our financial performance; | |
| • | regulatory developments in the United States and foreign countries; | |
| • | fluctuations in stock market prices and trading volumes of similar companies; | |
| • | sales of large blocks of our common stock, including sales by our executive officers, directors and significant stockholders; | |
| • | additions or departures of key personnel; | |
| • | discussion of us or our stock price by the financial and scientific press and in online investor communities; and | |
| • | changes in accounting principles generally accepted in the United States. |
In addition, class action litigation has often been instituted against companies whose securities have experienced periods of volatility in market price, such as the decline in our stock price following the announcement in August 2006 of the termination of our Phase II/III clinical trial for Troxatyl. Any such litigation brought against us could result in substantial costs and a diversion of management’s attention and resources, which could hurt our business, operating results and financial condition.
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our amended and restated certificate of incorporation and bylaws may delay or prevent an acquisition of us or a change in our management. These provisions include a classified board of directors, a prohibition on actions by written consent of our stockholders, and the ability of our board of directors to issue preferred stock without stockholder approval. In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which, subject to certain exceptions, prohibits stockholders owning in excess of 15% of our outstanding voting stock from merging or combining with us. Although we believe these provisions collectively provide for an opportunity to receive higher bids by requiring potential acquirors to negotiate with our board of directors, they would apply even if the offer may be considered beneficial by some stockholders. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors, which is responsible for appointing the members of our management.
We may incur increased costs as a result of changes in laws and regulations relating to corporate governance matters.
Changes in the laws and regulations affecting public companies, including the provisions of the Sarbanes-Oxley Act of 2002 and rules adopted by the Securities and Exchange Commission and by the Nasdaq Stock Market, will result in increased costs to us as we respond to their requirements. These laws and regulations could make it more difficult or more costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these requirements could also make it more difficult for us to
30
Table of Contents
attract and retain qualified persons to serve on our board of directors, our board committees or as executive officers. We are presently evaluating and monitoring developments with respect to these laws and regulations and cannot predict or estimate the amount or timing of additional costs we may incur to respond to their requirements.
If our executive officers, directors and largest stockholders choose to act together, they may be able to control our operations and act in a manner that advances their best interests and not necessarily those of other stockholders.
Our executive officers, directors and holders of 5% or more of our outstanding common stock beneficially own approximately 63% of our common stock. As a result, these stockholders, acting together, are able to control all matters requiring approval by our stockholders, including the election of directors and the approval of mergers or other business combination transactions. The interests of this group of stockholders may not always coincide with our interests or the interests of other stockholders, and they may act in a manner that advances their best interests and not necessarily those of other stockholders. These stockholders also may not act together and disputes may arise among these stockholders with respect to matters that require stockholder approval. Any disagreements among our significant stockholders, including among significant stockholders that are affiliated with members of our Board of Directors, may also make it more difficult for us to obtain stockholder approval of certain matters and may lead to distraction of management or have other adverse impact on our operations.
| Item 1B. | Unresolved Staff Comments |
Not applicable.
| Item 2. | Properties |
We lease approximately 60,568 square feet of laboratory and office space in San Diego, California under two lease agreements that terminate in December 2007 and a third lease agreement that terminates in September 2008. We believe that our facilities will adequately meet our present research and development needs.
| Item 3. | Legal Proceedings |
We are not currently involved in any material legal proceedings. We may be subject to various claims and legal actions arising in the ordinary course of business from time to time.
| Item 4. | Submission of Matters to a Vote of Security Holders |
No matters were submitted to a vote of security holders during the fourth quarter ended December 31, 2006.
PART II
| Item 5. | Market for the Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Common Stock Market Price
Our common stock commenced trading on the Nasdaq Global Market (formerly the Nasdaq National Market) on February 1, 2006 under the symbol “SGXP.” Prior to that time, there was no public market for our common stock. The following table sets forth the high and low closing sales prices for our common stock for the periods indicated, as reported on the Nasdaq Global Market. Such quotations represent inter-dealer prices without retail markup, markdown or commission and may not necessarily represent actual transactions.
2006 | High | Low | ||||||
| First Quarter | $ | 9.71 | $ | 5.75 | ||||
| Second Quarter | 8.18 | 4.65 | ||||||
| Third Quarter | 5.50 | 1.88 | ||||||
| Fourth Quarter | 3.56 | 2.50 | ||||||
31
Table of Contents
The closing price for our common stock as reported by the Nasdaq Global Market on March 13, 2007 was $5.71 per share. As of March 13, 2007, there were approximately 198 stockholders of record of our common stock.
Dividends
We have never declared or paid any cash dividends on our capital stock. The payment of dividends by us on our common stock is limited by our debt agreements. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. We do not intend to pay cash dividends on our common stock for the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our board of directors.
Use of Proceeds from the Sale of Registered Securities
On January 31, 2006, our registration statement onForm S-1 (RegistrationNo. 333-128059) was declared effective for our initial public offering. On February 6, 2006, we sold 4,000,000 shares of our common stock in connection with the closing of our initial public offering. On March 6, 2006, the underwriters exercised in part their over-allotment option and purchased an additional 152,904 shares of our common stock. As of December 31, 2006, we had used all of the proceeds from our initial public offering for the clinical development of Troxatyl, further development of our research programs and initial clinical development stemming from our internal programs, and working capital and general corporate purposes.
32
Table of Contents
Performance Measurement Comparison(1)
The following graph shows the total stockholder return of an investment of $100 in cash on February 1, 2006 in (i) the Company’s common stock, (ii) the Nasdaq Composite Index (the “Nasdaq”) and (iii) the AMEX Biotechnology Index (the “BTK”). All values assume reinvestment of the full amount of all dividends.
Comparison of Cumulative Total Return on Investment since our Initial Public Offering on February 1, 2006:
COMPARISON OF 11 MONTH CUMULATIVE TOTAL RETURN*
Among SGX Pharmaceuticals, Inc., The NASDAQ Composite Index
And The NASDAQ Biotechnology Index
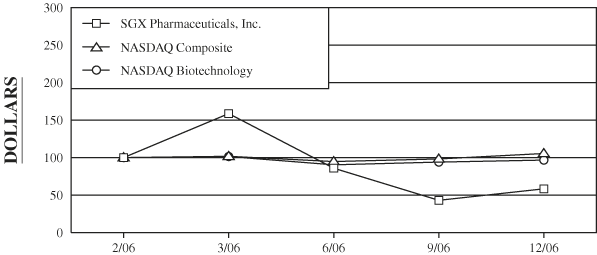
| * | $100 invested on2/1/06 in stock or on1/31/06 in index-including reinvestment of dividends. Fiscal year ending December 31. |
| 2/1/06 | 3/31/06 | 6/30/06 | 9/30/06 | 12/31/06 | |||||||||||||||||||||
SGX Pharmaceuticals, Inc. | 100.00 | 158.67 | 85.83 | 43.00 | 58.33 | ||||||||||||||||||||
NASDAQ Composite | 100.00 | 101.69 | 94.73 | 98.18 | 105.45 | ||||||||||||||||||||
NASDAQ Biotechnology | 100.00 | 101.38 | 90.35 | 93.99 | 96.66 | ||||||||||||||||||||
| (1) | This section is not “soliciting material,” is not deemed “filed” with the SEC, is not subject to the liabilities of Section 18 of the Securities Exchange Act of 1934, as amended, or the Exchange Act, and is not to be incorporated by reference in any filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act whether made before or after the date hereof and irrespective of any general incorporation language in any such filing. |
33
Table of Contents
| Item 6. | Selected Financial Data |
You should read the following selected consolidated financial and operating information for SGX Pharmaceuticals, Inc. together with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and notes thereto included elsewhere in this Annual Report onForm 10-K.
The consolidated statements of operations data for the years ended December 31, 2006, 2005 and 2004, and the consolidated balance sheet data as of December 31, 2006 and 2005 are derived from the audited consolidated financial statements included elsewhere in this report. The consolidated statements of operations data for the years ended December 31, 2003 and 2002, and the consolidated balance sheet data as of December 31, 2004, 2003 and 2002 are derived from audited consolidated financial statements not included in this report. Historical results for any prior period are not necessarily indicative of the results to be expected for any future period.
| Years Ended December 31, | ||||||||||||||||||||
| 2006 | 2005 | 2004 | 2003 | 2002 | ||||||||||||||||
| (In thousands, except per share data) | ||||||||||||||||||||
Statement of Operations Data: | ||||||||||||||||||||
| Revenue: | ||||||||||||||||||||
| Collaborations and commercial agreements | $ | 19,906 | $ | 14,604 | $ | 15,941 | $ | 10,135 | $ | 2,986 | ||||||||||
| Grants — subcontractor reimbursements | 5,120 | 5,083 | 4,976 | 4,599 | — | |||||||||||||||
| Grants | 2,754 | 1,949 | 6,380 | 3,344 | 350 | |||||||||||||||
| Total revenue | 27,780 | 21,636 | 27,297 | 18,078 | 3,336 | |||||||||||||||
| Expenses: | ||||||||||||||||||||
| Research and development | 46,942 | 37,881 | 31,444 | 28,587 | 25,573 | |||||||||||||||
| General and administrative | 9,588 | 11,820 | 6,719 | 7,353 | 10,122 | |||||||||||||||
| In-process technology | — | — | 4,000 | — | — | |||||||||||||||
| Total operating expenses | 56,530 | 49,701 | 42,163 | 35,940 | 35,695 | |||||||||||||||
| Loss from operations | (28,750 | ) | (28,065 | ) | (14,866 | ) | (17,862 | ) | (32,359 | ) | ||||||||||
| Interest income | 1,805 | 284 | 175 | 320 | 622 | |||||||||||||||
| Interest expense | (1,107 | ) | (422 | ) | (669 | ) | (1,219 | ) | (932 | ) | ||||||||||
| Interest expense associated with debenture | — | (1,188 | ) | (3,392 | ) | — | — | |||||||||||||
| Net loss | (28,052 | ) | (29,391 | ) | (18,752 | ) | (18,761 | ) | (32,669 | ) | ||||||||||
| Accretion to redemption value of redeemable convertible preferred stock | (49 | ) | (472 | ) | (329 | ) | (329 | ) | (329 | ) | ||||||||||
| Net loss attributable to common stockholders | $ | (28,101 | ) | $ | (29,863 | ) | $ | (19,081 | ) | $ | (19,090 | ) | $ | (32,998 | ) | |||||
| Basic and diluted net loss attributable to common stockholders per share | $ | (2.03 | ) | $ | (48.32 | ) | $ | (39.84 | ) | $ | (44.92 | ) | $ | (78.94 | ) | |||||
| Shares used to compute basic and diluted net loss attributable to common stockholders per share | 13,814 | 618 | 479 | 425 | 418 | |||||||||||||||
34
Table of Contents
| As of December 31, | ||||||||||||||||||||
| 2006 | 2005 | 2004 | 2003 | 2002 | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
Balance Sheet Data: | ||||||||||||||||||||
| Cash, cash equivalents and short-term investments | $ | 33,877 | $ | 17,718 | $ | 11,512 | $ | 13,635 | $ | 24,255 | ||||||||||
| Working capital (deficit) | 17,263 | 618 | (8,634 | ) | 1,042 | 15,656 | ||||||||||||||
| Total assets | 48,464 | 33,112 | 28,332 | 35,943 | 47,721 | |||||||||||||||
| Long-term debt obligations (including current portion) | 7,552 | 15,733 | 23,420 | 13,487 | 15,789 | |||||||||||||||
| Redeemable preferred stock | — | 46,837 | 74,850 | 88,306 | 87,977 | |||||||||||||||
| Accumulated deficit | (163,729 | ) | (135,628 | ) | (105,765 | ) | (86,684 | ) | (67,594 | ) | ||||||||||
| Total stockholders’ equity (deficit) | 13,613 | (41,677 | ) | (78,782 | ) | (78,044 | ) | (60,237 | ) | |||||||||||
35
Table of Contents
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
The following discussion and analysis should be read in conjunction with our consolidated financial statements and related notes included elsewhere in this report. This discussion and other parts of this report may contain forward-looking statements based upon current expectations that involve risks and uncertainties. Our actual results and the timing of selected events could differ materially from those anticipated in these forward-looking statements as a result of several factors, including those set forth under “Risk Factors” in Item 1A of Part I of this report and elsewhere in this report.
Overview
We are a biotechnology company focused on the discovery, development and commercialization of innovative cancer therapeutics. Our principal areas of focus in drug discovery are on protein and enzyme targets that have been implicated in cancers, including BCR-ABL, MET, JAK2 and RAS. We are advancing our internal oncology product pipeline through the application of our proprietary approach to drug discovery that is based upon the use of small fragments of drug-like molecules, or scaffolds, known as Fragments of Active Structures, or FAST. We have applied FAST to generate novel, potent and selective small molecule compounds for many proteins, or drug targets that have been implicated in cancers and other diseases. Our current goal is to advance our oncology pipeline and file at least one new IND application per year. We intend to continue our strategy of entering into collaborative alliances around certain of our internal programs in order to generate near-term revenue and offset discovery and development costs, as we did with our BCR-ABL program.
We generated approximately $27.8 million, $21.6 million, and $27.3 million in revenues from collaborations, commercial agreements and grants during the years ended December 31, 2006, 2005 and 2004, respectively. We have incurred significant losses since our inception in 1998, as we have devoted substantially all of our efforts to research and development activities. As of December 31, 2006, our accumulated deficit was approximately $163.7 million. We expect to incur substantial and possibly increasing losses for the next several years as we develop and expand our oncology pipeline.
We were incorporated in Delaware in July 1998. To date, we have not generated any revenues from the sale of therapeutic drugs. We have financed our operations and internal growth through private placements of our preferred stock, our collaboration, commercial agreement and grant revenue, debt financings, and our initial public offering.
Financial Operations Overview
Collaboration, Commercial Agreement and Grant Revenue
Collaboration, commercial agreement and grant revenue has primarily been a result of various contractual agreements with pharmaceutical companies and biotechnology companies, as well as government and other agencies. We also periodically receive non-refundable payments for achieving certain milestones during the term of our agreements.
Research and Development Expense
Research and development expense consists primarily of costs associated with our internal research programs and certain clinical trial costs, compensation, including stock-based, and other expenses related to research and development personnel, facilities costs and depreciation. We charge all research and development expenses to operations as they are incurred.
Our research activities are focused on building an internal oncology pipeline and generating lead compounds for ourselves and our potential partners through application of our FAST drug discovery platform. We have identified a portfolio of approximately 20 oncology drug targets that we believe are clearly implicated in cancers. Our most advanced programs based upon FAST are focused on compounds that inhibit BCR-ABL, MET and JAK2.
We incurred $2.8 million, $1.9 million, and $6.4 million of internal research expenses in connection with our NIH grants in 2006, 2005 and 2004, respectively. We also incurred $5.0 million, $5.1 million, and 5.0 million of expenses to subcontractors in connection with our research under NIH grants in 2006, 2005, and 2004, respectively.
36
Table of Contents
We incurred $9.5 million, $6.5 million, and $5.5 million of expenses related to the development of Troxatyl in 2006, 2005, and 2004 respectively, for a total of $21.5 million cumulatively expended on Troxatyl. These expenses for 2004 and cumulatively through December 31, 2006 include $4.0 million paid to Shire BioChem Inc. to in-license exclusive worldwide rights to Troxatyl.
All other research and development expenses are for various programs in the preclinical and research and discovery stages. For these preclinical programs, we use our internal resources, including our employees and discovery infrastructure, across several projects, and many of our costs are not attributable to a specific project but are directed to broadly applicable research projects. Accordingly, we do not account for our internal research and development costs on a project basis. Research and development expense also includes stock-based compensation expense associated with employees performing research and development activities.
We anticipate that our existing level of expenditures will support our current research and development activities. However, drug discovery and development outcomes and timelines and associated costs are uncertain and therefore vary widely. We anticipate that we will make determinations as to which research and development projects to pursue and how much funding to direct toward each project on an on-going basis in response to the scientific and clinical success in each program.
General and Administrative Expense
General and administrative expense consists primarily of compensation, including stock-based, and other expenses related to our corporate administrative employees, legal fees and other professional services expenses. We anticipate that we will maintain our existing level of general and administrative expenditures. However, we will make determinations as to the necessary levels of general and administrative expenditures on an on-going basis in response to our research and development activities and regulatory obligations.
Interest Income
Interest income consists of interest earned on our cash and cash equivalents and short-term investments.
Interest Expense
Interest expense in 2006 includes interest charges associated with the line of credit and equipment financing facility which we entered into with Silicon Valley Bank and Oxford Finance Corporation in September 2005. Interest expense in 2005 and also 2004 represents interest on our debt and secured promissory notes in an aggregate principal amount of $13.4 million that we issued in two tranches in a secured bridge financing in July and September 2004, which were converted into redeemable convertible preferred stock in April 2005.
Critical Accounting Policies and Estimates
The discussion and analysis of our financial condition and results of operations are based on our financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of financial statements requires us to make estimates and assumptions that affect the reported amounts of assets, liabilities, expenses and related disclosures. Actual results could differ from those estimates. While our significant accounting policies are described in more detail in Note 1 of the Notes to Consolidated Financial Statements included elsewhere in this report, we believe the following accounting policies to be critical to the judgments and estimates used in the preparation of our financial statements:
Revenue Recognition
Our collaboration agreements and commercial agreements contain multiple elements, including non-refundable upfront fees, payments for reimbursement of research costs, payments for ongoing research, payments associated with achieving specific milestones and, in the case of our collaboration agreements, development milestones and royalties based on specified percentages of net product sales, if any. We apply the revenue recognition criteria outlined in Staff Accounting Bulletin No. 104,Revenue Recognitionand Emerging Issues Task Force Issue00-21,Revenue Arrangements with Multiple Deliverables, or EITF00-21. In applying these revenue
37
Table of Contents
recognition criteria, we consider a variety of factors in determining the appropriate method of revenue recognition under these arrangements, such as whether the elements are separable, whether there are determinable fair values and whether there is a unique earnings process associated with each element of a contract.
Cash received in advance of services being performed is recorded as deferred revenue and recognized as revenue as services are performed over the applicable term of the agreement.
When a payment is specifically tied to a separate earnings process, revenues are recognized when the specific performance obligation associated with the payment is completed. Performance obligations typically consist of significant and substantive milestones pursuant to the related agreement. Revenues from milestone payments may be considered separable from funding for research services because of the uncertainty surrounding the achievement of milestones for products in early stages of development. Accordingly, these payments could be recognized as revenue if and when the performance milestone is achieved if they represent a separate earnings process as described in EITF00-21.
In connection with certain research collaborations and commercial agreements, revenues are recognized from non-refundable upfront fees, which we do not believe are specifically tied to a separate earnings process, ratably over the term of the agreement. Research services provided under some of our collaboration agreements and commercial agreements are on a fixed fee basis. Revenues associated with long-term fixed fee contracts are recognized based on the performance requirements of the agreements and as services are performed.
Revenues derived from reimbursement of directout-of-pocket expenses for research costs associated with grants are recorded in compliance with EITF Issue99-19,Reporting Revenue Gross as a Principal Versus Net as an Agent, and EITF Issue01-14,Income Statement Characterization of Reimbursements Received for“Out-of-Pocket” Expenses Incurred. According to the criteria established by these EITF Issues, in transactions where we act as a principal, with discretion to choose suppliers, bear credit risk and perform part of the services required in the transaction, we record revenue for the gross amount of the reimbursement. The costs associated with these reimbursements are reflected as a component of research and development expense in the statements of operations.
None of the payments that we have received from collaborators to date, whether recognized as revenue or deferred, is refundable even if the related program is not successful.
Stock-Based Compensation Expense
In December 2004, the Financial Accounting Standards Board, or FASB, issued Statement of Financial Accounting Standards, or SFAS, No. 123R,Share-Based Payment, which requires companies to expense the estimated fair value of employee stock options and similar awards. This statement is a revision to SFAS No. 123,Accounting for Stock-Based Compensation, and supersedes Accounting Principles Board, or APB, Opinion No. 25,Accounting for Stock Issued to Employees, and amends FASB Statement No. 95,Statement of Cash Flows. The accounting provisions of SFAS No. 123R became effective for us at the beginning of the first quarter of fiscal 2006.
We grant options to purchase our common stock to our employees and directors under our stock option plans. Eligible employees can also purchase shares of our common stock under the employee stock purchase plan at the lower of: (i) 85% of the fair market value on the first day of a two-year offering period; or (ii) 85% of the fair market value on the last date of each six-month purchase period within the two-year offering period. The benefits provided under these plans are stock-based payments subject to the provisions of SFAS 123R. Effective January 1, 2006, we began to use the fair value method to apply the provisions of SFAS 123R with a modified prospective application which provides for certain changes to the method for valuing stock-based compensation. The valuation provisions of SFAS 123R apply to new awards and to awards that were outstanding on January 1, 2006 and subsequently modified or cancelled. Under the modified prospective application, prior periods are not revised for comparative purposes. Prior to adopting the provisions of SFAS 123R, we recorded estimated compensation expense for employee stock options based upon their intrinsic value on the date of grant pursuant to APB Opinion 25. Stock-based compensation expense recognized under SFAS 123R for the year ended December 31, 2006 was $4.3 million (excluding stock-based compensation expense for share based awards to non-employees). At December 31, 2006, total unrecognized estimated compensation expense related to non-vested stock options granted prior to that date was $4.0 million, which is expected to be recognized over a weighted average period of 2.31 years. Total stock
38
Table of Contents
options granted during the year ended December 31, 2006 represented 5% of total outstanding shares as of the end of 2006. Assuming the conversion of all of our previously outstanding shares of preferred stock and a $6.0 million convertible note as of December 31, 2005, total stock options granted during the year ended December 31, 2005 represented 14% of total outstanding shares as of the end of 2005.
Both prior and subsequent to the adoption of SFAS 123R, we estimated the value of stock-based awards on the date of grant using the Black-Scholes option pricing model. Prior to the adoption of SFAS 123R, the value of each stock-based award was estimated on the date of grant using the Black-Scholes model for the pro forma information required to be disclosed under SFAS 123. The determination of the fair value of stock-based payment awards on the date of grant using an option-pricing model is affected by our stock price as well as assumptions regarding a number of complex and subjective variables. These variables include, but are not limited to, our expected stock price volatility over the term of the awards, risk-free interest rate and the expected term of the awards.
For purposes of estimating the fair value of stock options granted during the year ended December 31, 2006 using the Black-Scholes model, we have made a subjective estimate regarding our stock price volatility (weighted average of 73%). Expected volatility is based on average volatilities of the common stock of comparable publicly traded companies using a blend of historical, implied and average of historical and implied volatilities for this peer group of 10 companies, consistent with the guidance in SFAS 123R and SAB 107. If our stock price volatility assumption were increased to 78%, the weighted average estimated fair value of stock options granted during the year ended December 31, 2006 would increase by $0.49 per share, or 11%, from $4.36 to $4.85, respectively.
As permitted by Staff Accounting Bulletin No. 107,Share Based Payment (“SAB107”), we utilize the “shortcut approach” to estimate an options expected term, which represents the period of time that an option granted is expected to be outstanding. The expected term of options granted is derived from the average midpoint between vesting and the contractual term.
The risk-free interest rate for the expected term of the option is based on the average U.S. Treasury yield curve on the first day of each month for which the option is granted for the expected term (weighted average of 4.7% for the year ended December 31, 2006) which, if increased to 6.00%, would increase the weighted average estimated fair value of stock options granted during the year ended December 31, 2006 by $0.10 per share, or 2.4%.
We are required to assume a dividend yield as an input to the Black-Scholes model. The dividend yield assumption is based on our history. As we have never issued dividends and as we do not anticipate paying dividends in the foreseeable future, we have utilized a dividend yield of 0.0%.
Prior to 2006, stock-based compensation expense for stock options granted to employees and directors had been determined as the difference between the exercise price and the fair value of our common stock on the date of grant, as estimated by us for financial reporting purposes, on the date those options were granted. It also included stock-based compensation for options granted to consultants that has been determined in accordance with Statement of Financial Accounting Standards, or SFAS, No. 123,Accounting for Stock-Based Compensation, or SFAS 123, and EITF IssueNo. 96-18,Accounting for Equity Instruments That Are Issued to Other Than Employees for Acquiring or in Conjunction with Selling Goods and Services, as the fair value of the equity instruments issued and is periodically revalued as the options vest. Stock-based compensation expense depends on the amount of stock options and other equity compensation awards we grant to our employees, consultants and directors and the exercise price of those options.
Deferred stock compensation which is a non-cash charge, results from employee stock option grants at exercise prices that, for financial reporting purposes, are deemed to be below the estimated fair value of the underlying common stock on the date of grant. Given the absence of an active market for our common stock through 2005, our board of directors considered, among other factors, the liquidation preferences, anti-dilution protection and voting preferences of the preferred stock over the common stock in determining the estimated fair value of the common stock for purposes of establishing the exercise prices for stock option grants.
As a result of initiating our initial public offering, we revised our estimate of the fair value of our common stock for the last six months of 2004 and the year ended December 31, 2005 for financial reporting purposes. This was done retrospectively by management and we did not obtain contemporaneous valuations from an independent valuation specialist. In reassessing the value of our common stock in 2005 and 2004, we considered the price we
39
Table of Contents
received in April 2005 for our Series B preferred stock of $4.71 per share ($9.42 per share on an assumed converted basis). Starting on July 1, 2004, we reduced the value that we originally attributed to the preferences on the preferred stock mentioned above by 10% of the price of the preferred stock. Accordingly, we estimated the fair value at 90% of the Series B preferred stock price, or $4.24 per share ($8.48 per share on an assumed converted basis). We kept this value constant until April 2005 when we steadily increased the estimated fair value to $14.06 per common share based on an assessment of market considerations, including discussions with the underwriters who managed the initial public offering. Furthermore, we believe this valuation approach is consistent with valuation methodologies applied to other similar companies for financial reporting purposes pursuing an initial public offering.
For stock option and restricted stock grants to employees and non-employee directors, we recorded deferred stock compensation, net of forfeitures, totaling $13.6 million and $0.0 in 2005 and 2004, respectively, which represent the difference between the revised fair value for financial reporting purposes of our common stock and the option exercise price at the date of grant. We also recorded deferred stock compensation of $1.7 million for the issuance of equity instruments to former employees and consultants in 2005. Deferred compensation was to be amortized to expense over the vesting period of the related options using an accelerated method. Based upon stock option and restricted stock grants through December 31, 2005, the expected future amortization expense for deferred stock compensation is $3.5 million, $1.4 million, $200,000 and $24,000 for the years ending December 31, 2006, 2007, 2008, and 2009, respectively. As a result of the adoption of SFAS 123R on January 1, 2006, these deferred compensation amounts, totaling $5.1 million, were reversed out of deferred compensation against additional paid-in capital and the expense will be taken in accordance with SFAS 123R for all remaining unvested grants.
Accrued Expenses
As part of the process of preparing financial statements, we are required to estimate accrued expenses. This process involves identifying services which have been performed on our behalf, and estimating the level of service performed and the associated costs incurred for such service as of each balance sheet date in our financial statements. Examples of estimated expenses for which we accrue include contract service fees paid to contract manufacturers in conjunction with the production of clinical drug supplies and to contract research organizations. In connection with such service fees, our estimates are most affected by our understanding of the status and timing of services provided relative to the actual levels of services incurred by such service providers. The majority of our service providers invoice us monthly in arrears for services performed. The date on which certain services commence, the level of services performed on or before a given date and the cost of such services are often determined based on subjective judgments. We make these judgments based upon the facts and circumstances known to us in accordance with generally accepted accounting principles.
Deferred Tax Asset Valuation Allowance
Our estimate for the valuation allowance for deferred tax assets requires us to make significant estimates and judgments about our future operating results. Our ability to realize the deferred tax assets depends on our future taxable income as well as limitations on utilization. A deferred tax asset must be reduced by a valuation allowance if it is more likely than not that some portion or all of the deferred tax asset will not be realized prior to its expiration. The projections of our operating results on which the establishment of a valuation allowance is based involve significant estimates regarding future demand for our products, competitive conditions, product development efforts, approvals of regulatory agencies and product cost. We have recorded a full valuation allowance on our net deferred tax assets as of December 31, 2006 and 2005 due to uncertainties related to our ability to utilize our deferred tax assets in the foreseeable future. These deferred tax assets primarily consist of certain net operating loss carry-forwards and research and development tax credits.
Results of Operations
Year Ended December 31, 2006 Compared to 2005
Collaboration, Commercial Agreement and Grant Revenue. Collaboration, commercial agreement and grant revenue increased to $27.8 million for the year ended December 31, 2006 from $21.6 million for the year ended
40
Table of Contents
December 31, 2005. The increase of $6.1 million, or 28%, was primarily due to the amortization into revenue of the $20 million upfront payment received from Novartis (approximately $3.8 million), the initiation of the research services (approximately $3.4 million) and reimbursement of out of pocket expenses (approximately $1.6 million), both in connection with the collaboration with Novartis, increased research grant efforts (approximately $0.8 million) and the continuation of research services performed in connection with the Cystic Fibrosis Foundation Therapeutics, Inc. (approximately $0.9 million). These additional revenues were offset by a reduction in revenues due to the conclusion of research services in 2006 which were ongoing in 2005.
Research and Development Expense. Research and development expense increased to $46.9 million for the year ended December 31, 2006 from $37.9 million for the year ended December 31, 2005. The increase of $9.0 million, or 24%, was primarily attributable to $3.7 million of additional clinical development costs for Troxatyl, $2.1 million related to increased laboratory supply usage, $1.7 million related to an increased use of outside services, $2.2 million related to additional headcount and salaries, and $1.1 million related to additional facilities costs, offset by a $2.0 million net reduction in stock-based compensation expense. We anticipate that our existing level of expenditures will support our current research and development activities. However, drug discovery and development outcomes timelines and associated costs are uncertain and therefore vary widely. We anticipate that we will make determinations as to which research and development projects to pursue and how much funding to direct toward each project on an on-going basis in response to the scientific and clinical success of each program.
General and Administrative. General and administrative expense decreased to $9.6 million for the year ended December 31, 2006 from $11.8 million for the year ended December 31, 2005. The decrease of $2.2 million, or 19%, was primarily attributable to a decrease of $3.6 million in stock-based compensation expense offset by an increase of $0.5 million in legal and other professional services and a $0.5 million increase in accrued salaries and related costs. We anticipate that we will maintain our existing level of general and administrative expenditures. However, we will make determinations as to the necessary levels of general and administrative expenditures on an on-going basis in response to our research and development activities and regulatory obligations.
Interest Income. Interest income increased to $1.8 million for the year ended December 31, 2006 from $284,000 for the year ended December 31, 2005. The increase of $1.5 million, or 536%, was due primarily to higher cash and cash equivalent balances in 2006 compared to 2005. The higher cash balances are the result of the proceeds received from our initial public offering in February 2006 and the Novartis agreement in March 2006.
Interest Expense. Interest expense (excluding interest expense associated with our bridge notes issued in July and September 2004) increased to $1.1 million for the year ended December 31, 2006 from $422,000 for the year ended December 31, 2005. We did not receive any funds under the line of credit and equipment financing agreements with Silicon Valley Bank and Oxford Finance Corporation until September 2005. We also received funds under these arrangements in December 2005 and December 2006. Accordingly, the increase in interest expense was due to the higher debt levels in 2006 as compared to 2005 (excluding indebtedness under our bridge notes issued in July and September 2004).
Interest Expense Associated with Bridge Notes. We recorded interest expense of $1.2 million during the year ended December 31, 2005 related to the bridge notes issued in July and September 2004. We did not record any interest expense during the year ended December 31, 2006 related to these bridge notes as the notes were converted into preferred stock in April 2005.
Year Ended December 31, 2005 Compared to 2004
Collaboration, Commercial Agreement and Grant Revenue. Collaboration, commercial agreement and grant revenue declined to $21.6 million for the year ended December 31, 2005 from $27.3 million for the year ended December 31, 2004. The decrease of $5.7 million, or 21%, was primarily due to the conclusion of a previously existing grant agreement, partially offset by a new grant agreement that commenced in the third quarter of 2005, resulting in a net decrease in grant revenue of $4.3 million. In addition, a decrease of $1.4 million was due to fewer new agreements signed in 2005 as we changed our business strategy to focus on oncology drug discovery and development.
41
Table of Contents
Research and Development Expense. Research and development expense increased to $37.9 million for the year ended December 31, 2005 from $31.4 million for the year ended December 31, 2004. The increase was primarily attributable to increased clinical trial expenses incurred for Troxatyl and the recognition of equity-based compensation for option grants.
General and Administrative. General and administrative expense increased to $11.8 million for the year ended December 31, 2005 from $6.7 million for the year ended December 31, 2004. The increase was primarily attributable to an increase in equity-based compensation for option and restricted stock grants in 2005.
Amortization of Stock-Based Compensation. We recorded amortization of stock-based compensation of $10.2 million and $0.6 million for the years ended December 31, 2005 and 2004, respectively. The increase was attributable to the additional deferred compensation recorded for financial reporting purposes, as a result of our initial public offering, from the issuance of stock option grants at exercise prices that were deemed to be below the estimated fair value of the underlying common stock on the date of grant.
Interest Income. Interest income increased to $284,000 for the year ended December 31, 2005 from $175,000 for the year ended December 31, 2004. The increase was due primarily to higher cash and cash equivalent balances in 2005 compared to 2004.
Interest Expense. Interest expense (excluding interest expense associated with our bridge notes issued in July and September 2004) decreased to $422,000 for the year ended December 31, 2005 from $669,000 for the year ended December 31, 2004. We did not receive any funds under the line of credit and equipment financing agreements with Silicon Valley Bank and Oxford Finance Corporation until September 2005 and December 2005. Accordingly, the decrease in interest expense was due to the lower debt levels in 2005 as compared to 2004 (excluding indebtedness under our bridge notes issued in July and September 2004).
Interest Expense Associated with Bridge Notes. We recorded interest expense of $1.2 million and $3.4 million during the years ended December 31, 2005 and 2004, respectively, related to the bridge notes issued in July and September 2004. Included in the bridge note interest expense is the amortization of the fair value of warrants issued in connection with the bridge notes. We determined the fair value of the warrants on the grant date using the Black-Scholes pricing model. This resulted in aggregate expense of approximately $1.7 million, which is recorded against the principal balance. The remaining $363,000 was recognized as interest expense in the year ended December 31, 2005.
Also included in the bridge note interest expense is an additional non-cash charge of approximately $1.7 million against the principal balance of the bridge notes. This amount represents the difference between the conversion price of the bridge notes and the underlying value of the stock issued upon conversion of the bridge notes. The remaining $363,000 of this non-cash charge was recognized as interest expense in the year ended December 31, 2005.
Liquidity and Capital Resources
Sources of Liquidity
We have historically funded our operations primarily through the sale of our equity securities and funds received from our collaborations, commercial agreements, grants and debt financings. In April and December 2005, we received total net proceeds of approximately $13.2 million from the sale of our Series B preferred stock. In February 2006, we completed an initial public offering of an aggregate of 4,152,904 shares of our common stock and raised net proceeds of approximately $20.6 million, after deducting the underwriting discount and offering expenses, and including the underwriter’s over-allotment option which was exercised in March 2006. Upon the completion of our initial public offering in February 2006, all of our previously outstanding shares of preferred stock converted into an aggregate of 8,346,316 shares of our common stock and a convertible note of $6.0 million converted into 1,000,000 shares of our common stock. We have recorded revenues from our collaborations, commercial agreements and grants totaling $27.8 million, $21.6 million, and $27.3 million for the years ended December 31, 2006, 2005, and 2004, respectively.
In September 2005, we entered into a line of credit and equipment financing agreement with Silicon Valley Bank and Oxford Finance Corporation to provide $8.0 million of general purpose working capital financing and
42
Table of Contents
$2.0 million of equipment and leasehold improvements financing. The debt bears interest at a rate of approximately 10% per annum and is due in monthly installments over three years. In September and December 2005, we borrowed approximately $4.0 million and $4.9 million, respectively, of the funds available under this line of credit and equipment financing agreement for general purpose working capital needs and capital expenditures spending, and issued the lenders warrants to purchase an aggregate of 90,370 shares of our Series B preferred stock, which became exercisable for 45,184 shares of our common stock, at an exercise price of $9.42 per share, upon the completion of our initial public offering. In November and December of 2006, we borrowed the remainder of the available financing under this line of credit and equipment financing agreement of approximately $1.1 million, and issued the lenders warrants to purchase an aggregate of 5,771 shares of our common stock at an exercise price of $9.42 per share.
As of December 31, 2006, an aggregate of approximately $7.6 million was outstanding under our line of credit and equipment financing agreement with Silicon Valley Bank and Oxford Finance Corporation entered into in 2005 and other lines of credit entered into prior to 2005. The debt agreements subject us to certain financial and non-financial covenants. As of December 31, 2006, we were in compliance with these covenants. These obligations are secured by our assets, excluding intellectual property, and are due in monthly installments through 2010. They bear interest at effective rates ranging from approximately 9.14% to 11.03% and are subject to prepayment fees of up to 4% of the outstanding principal balance as of the prepayment date. We made debt repayments of approximately $3.5 million, $3.0 million, and $3.9 million for the years ended December 31, 2006, 2005, and 2004, respectively.
In March 2006, we entered into a license and collaboration agreement with Novartis to develop and commercialize BCR-ABL inhibitors for the treatment of CML. In connection with the license and collaboration agreement, Novartis paid us a non-refundable, non-creditable license fee of $20.0 million, which was received in May 2006.
In addition, Novartis Pharma AG purchased 637,755 shares of our common stock for $5.0 million in March 2006.
Cash Flows
Our cash flows for 2007 and beyond will depend on a variety of factors, some of which are discussed below.
As of December 31, 2006, cash and cash equivalents and short-term investments totaled approximately $33.9 million as compared to $17.7 million at December 31, 2005, an increase of approximately $16.2 million. The increase resulted primarily from the $20.6 million in net proceeds received from our IPO in February 2006 and the $25.0 million received from Novartis in connection with the license and collaboration agreement we entered into in March 2006, partially offset by net cash used to fund ongoing operations.
For the year ended December 31, 2006, net cash used in operating activities was approximately $5.9 million reflecting the net loss for this period of $28.1 million and an increase in accounts receivable and other assets of $1.0 million, offset primarily by an increase in deferred revenue of $15.2 million and non-cash items totaling $8.1 million. Net cash used by investing activities was approximately $7.5 million reflecting purchases of short-term investments of $6.0 million and purchases of property and equipment of approximately $1.5 million. Net cash provided by financing activities was approximately $23.5 million reflecting net proceeds of $25.8 million from the issuance of common stock in connection with our initial public offering and collaboration agreement with Novartis and $1.1 million of proceeds from our line of credit and equipment financings, offset by $3.4 million in repayments on lines of credit and notes payable.
For the year ended December 31, 2005, net cash used in operating activities was approximately $11.8 million reflecting the net loss for this period of $29.4 million, an increase in prepaid expenses and other assets of $1.4 million and deferred rent of $0.1 million, offset primarily by non-cash items totaling $14.9 million and an increase in deferred revenue of $0.7 million. Net cash used by investing activities was approximately $1.3 million for purchases of property and equipment. Net cash provided by financing activities was approximately $19.4 million reflecting net proceeds of $13.4 million from the issuance of preferred and common stock, proceeds from our line of credit and equipment financings of $8.9 million and proceeds from repayment of notes receivable from stockholders, offset by $3.0 million in repayments on lines of credit and notes payable.
43
Table of Contents
Following a review of our oncology product pipeline and the resources currently available to us we determined we will not initiate additional Troxatyl clinical studies while, in consultation with Shire, we consider the future options for Troxatyl, such as seeking one or more third party collaborative or cooperative arrangements. As such, we are unable to estimate the costs, if any, we will incur in the future development of Troxatyl. We anticipate that our existing level of expenditures will support our current research and development activities. However, drug discovery and development, outcomes, timelines and associated costs are uncertain and therefore vary widely. We anticipate that we will make determinations as to which research and development projects to pursue and how much funding to direct toward each project on an on-going basis in response to the scientific and clinical success of each program and, with respect to Troxatyl, in consultation with Shire.
Funding Requirements
Our future capital uses and requirements depend on numerous factors, including but not limited to the following:
| • | terms and timing of any collaborative, licensing and other arrangements that we may establish, including partnering our internal discovery programs, such as MET and JAK2; | |
| • | rate of progress and cost of our clinical trials and other research and development activities; | |
| • | scope, prioritization and number of clinical development and research programs we pursue; | |
| • | costs and timing of preparing regulatory submissions and obtaining regulatory approval; | |
| • | costs of establishing or contracting for sales and marketing capabilities; | |
| • | costs of manufacturing; | |
| • | extent to which we acquire or in-license new products, technologies or businesses; | |
| • | effect of competing technological and market developments; and | |
| • | costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights. |
We believe that our existing cash and cash equivalents and short-term investments, together with interest thereon and cash from existing collaborations, commercial agreements and grants, will be sufficient to meet projected operating requirements through 2008. Consistent with our existing business development strategy, we anticipate establishing new collaborations and commercial agreements. Any proceeds received in connection with such new transactions, or milestones achieved under existing or new collaborations or commercial agreements, would provide additional operating capital.
Until we can generate significant cash from our operations, we expect to continue to fund our operations with existing cash resources that were primarily generated from the proceeds of offerings of our equity securities, our collaboration, commercial agreement, grant revenue, and debt financing. In addition, we may finance future cash needs through the sale of other equity securities, strategic collaboration agreements and debt financing. However, we may not be successful in obtaining additional collaboration agreements or commercial agreements, or in receiving milestone or royalty payments under existing agreements. In particular, if we do not generate additional revenue from collaborations, commercial agreements and grants at the levels we project, we may require additional funding sooner than we currently anticipate. In addition, we cannot be sure that our existing cash and cash equivalents and short-term investments will be adequate or that additional financing will be available when needed or that, if available, financing will be obtained on terms favorable to us or our stockholders. Having insufficient funds may require us to delay, scale back or eliminate some or all of our research or development programs or to relinquish greater or all rights to product candidates at an earlier stage of development or on less favorable terms than we would otherwise choose. Failure to obtain adequate financing may also adversely affect our ability to operate as a going concern. If we raise additional funds by issuing equity securities, substantial dilution to existing stockholders would likely result. If we raise additional funds by incurring debt financing, the terms of the debt may involve significant cash payment obligations as well as covenants and specific financial ratios that may restrict our ability to operate our business.
44
Table of Contents
Off-Balance Sheet Arrangements
As of December 31, 2006 and 2005, we have not invested in any variable interest entities. We do not have any relationships with unconsolidated entities or financial partnerships, such as entities often referred to as structured finance or special purpose entities, which would have been established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purposes. In addition, we do not engage in trading activities involving non-exchange traded contracts. As such, we are not materially exposed to any financing, liquidity, market or credit risk that could arise if we had engaged in these relationships. We do not have relationships or transactions with persons or entities that derive benefits from their non-independent relationship with us or our related parties other than as described in the Notes to Financial Statements included elsewhere in this report.
Contractual Obligations
The following summarizes our long-term contractual obligations as of December 31, 2006:
| Payments Due by Period | ||||||||||||||||||||
| Less Than | 1 to | 4 to | More Than | |||||||||||||||||
Contractual Obligations | Total | 1 Year | 3 Years | 5 Years | 5 Years | |||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Long-term debt obligations | $ | 7,870 | $ | 3,526 | $ | 4,326 | $ | 18 | — | |||||||||||
| Operating lease obligations | 2,176 | 1,417 | 740 | 19 | — | |||||||||||||||
| License obligations | — | — | — | — | — | |||||||||||||||
| Total | $ | 10,046 | $ | 4,943 | $ | 5,066 | $ | 37 | $ | — | ||||||||||
Related Party Transactions
For a description of our related party transactions, see “Certain Relationships and Related Transactions, and Director Independence.”
Income Taxes
As of December 31, 2006, we had federal and California net operating loss carryforwards of approximately $107.6 million and $76.2 million, respectively, which begin to expire in 2019 and 2009, respectively, if not utilized. We also had federal and California research and development tax credit carryforwards totaling approximately $5.0 million and $3.4 million, respectively. The federal research and development tax credit carryforward will begin to expire in 2019, unless previously utilized.
Pursuant to Internal Revenue Code Sections 382 and 383, and similar state provisions, use of our net operating loss and tax credit carryforwards may be limited as a result of certain cumulative changes in our stock ownership. The annual limitations may result in the expiration of net operating losses and credits prior to utilization.
At December 31, 2006 and 2005, we had deferred tax assets primarily representing the benefit of net operating loss carryforwards. We did not record a benefit for the deferred tax assets because realization of the deferred tax assets was uncertain and, accordingly, a valuation allowance has been provided to completely offset the deferred tax assets.
Recently Issued Accounting Pronouncements
In July 2006, the Financial Accounting Standards Board (FASB) issued FASB Interpretation (FIN) No. 48,Accounting for Uncertainty in Income Taxes — an interpretation of FASB Statement No. 109, which clarifies the accounting for uncertainty in tax positions. FIN No. 48 requires that the Company recognize the impact of a tax position in its financial statements if that position is more likely than not of being sustained on audit, based on the technical merits of the position. The provisions of FIN No. 48 are effective as of the beginning of the Company’s 2007 fiscal year, with the cumulative effect of the change in accounting principle recorded as an adjustment to opening retained earnings. The Company does not expect the adoption of FIN No. 48 to have a material impact on
45
Table of Contents
its consolidated results of operations and financial position, and the Company is continuing to evaluate the impact, if any, the adoption of FIN No. 48 will have on its disclosure requirements.
In September 2006, the SEC issued Staff Accounting Bulletin No. 108 (SAB 108) “Considering the Effects of Prior Year Misstatements when Quantifying Misstatements in Current Year Financial Statements”. SAB 108 addresses how the effects of prior year uncorrected misstatements should be considered when quantifying misstatements in current year financial statements. SAB 108 requires companies to quantify misstatements using a balance sheet and income statement approach and to evaluate whether either approach results in quantifying an error that is material in light of relevant quantitative and qualitative factors. When the effect of initial adoption is material, companies will record the effect as a cumulative effect adjustment to beginning of year retained earnings. The provisions of SAB 108 are effective for annual statements covering the first fiscal year ending after November 15, 2006. The adoption of SAB 108 did not have a material impact on the Company’s financial position or results of operations.
In September 2006, the FASB issued SFAS No. 157,Fair Value Measurements. SFAS No. 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles and expands disclosures about fair value measurements. This Statement applies only to fair value measurements that are already required or permitted by other accounting standards. Accordingly, this Statement does not require any new fair value measurements. SFAS No. 157 is effective for fiscal years beginning after December 15, 2007. The Company is currently evaluating the impact, if any, the adoption of SFAS No. 157 will have on the Company’s financial position or results of operations.
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk |
The primary objective of our investment activities is to preserve principal while maximizing income without significantly increasing risk. Some of the securities in which we invest may be subject to market risk. This means that a change in prevailing interest rates may cause the market value of the investment to fluctuate. To minimize this risk, we may maintain our portfolio of cash equivalents and short-term investments in a variety of securities, including commercial paper, money market funds, debt securities and certificates of deposit. The risk associated with fluctuating interest rates is limited to our investment portfolio and we do not believe that a 1% change in interest rates would have had a significant impact on our interest income for 2006 or 2005. As of December 31, 2006, all of our cash equivalents were held in checking accounts, money market accounts, commercial paper and government agency securities. We do not have any holdings of derivative financial or commodity instruments, and we have an immaterial amount of foreign currency denominated transactions.
46
Table of Contents
| Item 8. | Financial Statements and Supplementary Data |
Index to Financial Statements
| 48 | ||||
| 49 | ||||
| 50 | ||||
| 51 | ||||
| 52 | ||||
| 53 |
47
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders
SGX Pharmaceuticals, Inc.
SGX Pharmaceuticals, Inc.
We have audited the accompanying consolidated balance sheets of SGX Pharmaceuticals, Inc. as of December 31, 2006 and 2005 and the related consolidated statements of operations, stockholders’ equity (deficit), and cash flows for each of the three years in the period ended December 31, 2006. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of SGX Pharmaceuticals, Inc., at December 31, 2006 and 2005, and the consolidated results of its operations and its cash flows for each of the three years in the period ended December 31, 2006, in conformity with U.S. generally accepted accounting principles.
As discussed in Note 1 to the consolidated financial statements, SGX Pharmaceuticals, Inc. changed its method of accounting for share based payments in accordance with Statement of Financial Accounting Standards No. 123 (revised 2004) effective January 1, 2006.
/s/ Ernst & Young LLP
San Diego, California
March 27, 2007
48
Table of Contents
SGX Pharmaceuticals, Inc.
| December 31, | ||||||||
| 2006 | 2005 | |||||||
| (In thousands, except par value and share data) | ||||||||
ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 27,877 | $ | 17,718 | ||||
| Short-term investments | 6,000 | — | ||||||
| Accounts receivable | 3,532 | 863 | ||||||
| Prepaid expenses, deposits and other current assets | 1,616 | 1,241 | ||||||
| Total current assets | 39,025 | 19,822 | ||||||
| Property and equipment, net | 5,435 | 7,168 | ||||||
| Goodwill and intangible assets, net | 3,412 | 3,439 | ||||||
| Other assets | 592 | 2,683 | ||||||
| Total assets | $ | 48,464 | $ | 33,112 | ||||
LIABILITIES AND STOCKHOLDERS’ EQUITY (DEFICIT) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 2,109 | $ | 2,935 | ||||
| Accrued liabilities | 4,774 | 4,190 | ||||||
| Other current liabilities | 330 | 128 | ||||||
| Current portion of line of credit | 7,552 | 9,733 | ||||||
| Deferred revenue | 6,997 | 2,218 | ||||||
| Total current liabilities | 21,762 | 19,204 | ||||||
| Deferred rent | 66 | 169 | ||||||
| Deferred revenue, long-term | 13,023 | 2,579 | ||||||
| Note payable, net of current portion | — | 6,000 | ||||||
| Commitments and contingencies (Note 4) | ||||||||
| Redeemable convertible preferred stock, par value $0.001; Authorized shares — none and 19,000,000 at December 31, 2006 and 2005, respectively; issued and outstanding shares — none and 16,692,654 at December 31, 2006 and 2005, respectively; aggregate liquidation preference and redemption amount — $0 and $48,190 at December 31, 2006 and 2005, respectively | — | 46,837 | ||||||
| Stockholders’ equity (deficit): | ||||||||
| Preferred stock, par value $0.001; Authorized shares — 5,000,000 and none at December 31, 2006 and 2005, respectively; and no shares issued and outstanding at December 31, 2006 and 2005 | — | — | ||||||
| Common stock, par value $0.001; Authorized shares — 75,000,000 and 50,000,000 at December 31, 2006 and 2005, respectively; issued and outstanding shares — 15,166,109 and 854,160 at December 31, 2006 and 2005, respectively | 16 | 1 | ||||||
| Notes receivable from stockholders | (21 | ) | (59 | ) | ||||
| Additional paid-in capital | 177,355 | 99,110 | ||||||
| Deferred compensation | — | (5,101 | ) | |||||
| Accumulated other comprehensive loss | (8 | ) | — | |||||
| Accumulated deficit | (163,729 | ) | (135,628 | ) | ||||
| Total stockholders’ equity (deficit) | 13,613 | (41,677 | ) | |||||
| Total liabilities and stockholders’ equity (deficit) | $ | 48,464 | $ | 33,112 | ||||
See accompanying notes.
49
Table of Contents
SGX Pharmaceuticals, Inc.
| Years Ended December 31, | ||||||||||||
| 2006 | 2005 | 2004 | ||||||||||
| (In thousands, except per share data) | ||||||||||||
| Revenue: | ||||||||||||
| Collaborations and commercial agreements | $ | 19,906 | $ | 14,604 | $ | 15,941 | ||||||
| Grants — subcontractor reimbursements | 5,120 | 5,083 | 4,976 | |||||||||
| Grants | 2,754 | 1,949 | 6,380 | |||||||||
| Total revenue | 27,780 | 21,636 | 27,297 | |||||||||
| Expenses: | ||||||||||||
| Research and development | 46,942 | 37,881 | 31,444 | |||||||||
| General and administrative | 9,588 | 11,820 | 6,719 | |||||||||
| In-process technology | — | — | 4,000 | |||||||||
| Total operating expenses | 56,530 | 49,701 | 42,163 | |||||||||
| Loss from operations | (28,750 | ) | (28,065 | ) | (14,866 | ) | ||||||
| Interest income | 1,805 | 284 | 175 | |||||||||
| Interest expense | (1,107 | ) | (422 | ) | (669 | ) | ||||||
| Interest expense associated with bridge notes | — | (1,188 | ) | (3,392 | ) | |||||||
| Net loss | (28,052 | ) | (29,391 | ) | (18,752 | ) | ||||||
| Accretion to redemption value of redeemable convertible preferred stock | (49 | ) | (472 | ) | (329 | ) | ||||||
| Net loss attributable to common stockholders | $ | (28,101 | ) | $ | (29,863 | ) | $ | (19,081 | ) | |||
| Basic and diluted net loss per share attributable to common stockholders | $ | (2.03 | ) | $ | (48.32 | ) | $ | (39.84 | ) | |||
| Shares used to compute basic and diluted net loss per share attributable to common stockholders | 13,814 | 618 | 479 | |||||||||
See accompanying notes.
50
Table of Contents
SGX Pharmaceuticals, Inc.
| Years Ended December 31, 2004, 2005 and 2006 | ||||||||||||||||||||||||||||||||
| Notes | Accumulated | Total | ||||||||||||||||||||||||||||||
| Receivable | Additional | Other | Stockholders’ | |||||||||||||||||||||||||||||
| Common Stock | from | Paid-In | Deferred | Comprehensive | Accumulated | Equity | ||||||||||||||||||||||||||
| Shares | Amount | Stockholders | Capital | Compensation | Loss | Deficit | (Deficit) | |||||||||||||||||||||||||
| (In thousands, except share data) | ||||||||||||||||||||||||||||||||
| Balance at December 31, 2003 | 427,156 | $ | 1 | $ | (1,997 | ) | $ | 11,226 | $ | (590 | ) | $ | — | $ | (86,684 | ) | $ | (78,044 | ) | |||||||||||||
| Issuance of common stock upon exercise of stock options | 11,196 | — | — | 62 | — | — | — | 62 | ||||||||||||||||||||||||
| Repurchase of unvested restricted stock | (744 | ) | — | — | (10 | ) | — | — | — | (10 | ) | |||||||||||||||||||||
| Repurchase of common stock in exchange for settlement of notes and accrued interest from stockholders | (131,224 | ) | — | 1,764 | (1,764 | ) | — | — | — | — | ||||||||||||||||||||||
| Forgiveness of a portion of principal on notes and accrued interest on note settlement | — | — | 131 | 651 | — | — | — | 782 | ||||||||||||||||||||||||
| Repayment of notes receivable from stockholders | — | — | 42 | — | — | — | — | 42 | ||||||||||||||||||||||||
| Accrued interest on notes receivable from stockholders | — | — | (78 | ) | — | — | — | — | (78 | ) | ||||||||||||||||||||||
| Deferred compensation for issuance of equity instruments | — | — | — | 6 | (6 | ) | — | — | — | |||||||||||||||||||||||
| Amortization of stock-based compensation | — | — | — | (147 | ) | 596 | — | — | 449 | |||||||||||||||||||||||
| Issuance costs incurred in equity financing | — | — | — | (166 | ) | — | — | — | (166 | ) | ||||||||||||||||||||||
| Conversion of redeemable preferred stock into common stock for non-participation in the bridge financing | 194,052 | — | — | 13,785 | — | — | — | 13,785 | ||||||||||||||||||||||||
| Issuance of warrants to bridge note lenders | — | — | — | 3,477 | — | — | — | 3,477 | ||||||||||||||||||||||||
| Deemed dividend and accretion to redemption value of redeemable convertible preferred stock | — | — | — | — | — | — | (329 | ) | (329 | ) | ||||||||||||||||||||||
| Net loss and comprehensive loss | — | — | — | — | — | — | (18,752 | ) | (18,752 | ) | ||||||||||||||||||||||
| Balance at December 31, 2004 | 500,436 | 1 | (138 | ) | 27,120 | — | — | (105,765 | ) | (78,782 | ) | |||||||||||||||||||||
| Issuance of common stock upon exercise of stock options | 213,417 | — | — | 222 | — | — | — | 222 | ||||||||||||||||||||||||
| Conversion of preferred stock into common stock for non-participation in the Series B financing | 70,392 | — | — | 1,410 | — | — | — | 1,410 | ||||||||||||||||||||||||
| Repayment of notes receivable from stockholders | — | — | 81 | — | — | — | — | 81 | ||||||||||||||||||||||||
| Accrued interest on notes receivable from stockholders | — | — | (3 | ) | — | — | — | — | (3 | ) | ||||||||||||||||||||||
| Deferred compensation for issuance of equity instruments | — | — | — | 12,944 | (12,944 | ) | — | — | — | |||||||||||||||||||||||
| Amortization of stock-based compensation | — | — | — | — | 10,233 | — | — | 10,233 | ||||||||||||||||||||||||
| Repurchase of unvested restricted stock | (85 | ) | — | 1 | (4 | ) | — | — | — | (3 | ) | |||||||||||||||||||||
| Issuance of restricted stock | 70,000 | — | — | 654 | (654 | ) | — | — | — | |||||||||||||||||||||||
| Issuance of equity instruments to former employees and consultants | — | — | — | 1,736 | (1,736 | ) | — | — | — | |||||||||||||||||||||||
| Issuance of warrants to lenders | — | — | — | 498 | — | — | — | 498 | ||||||||||||||||||||||||
| Deemed dividend and accretion to redemption value of redeemable convertible preferred stock | — | — | — | — | — | — | (472 | ) | (472 | ) | ||||||||||||||||||||||
| Reduction of redemption value on redeemable preferred stock | — | — | — | 54,530 | — | — | — | 54,530 | ||||||||||||||||||||||||
| Net loss and comprehensive loss | — | — | — | — | — | — | (29,391 | ) | (29,391 | ) | ||||||||||||||||||||||
| Balance at December 31, 2005 | 854,160 | 1 | (59 | ) | 99,110 | (5,101 | ) | — | (135,628 | ) | (41,677 | ) | ||||||||||||||||||||
| Net loss | — | — | — | — | — | — | (28,052 | ) | (28,052 | ) | ||||||||||||||||||||||
Unrealized loss onavailable-for-sale securities | — | — | — | — | — | (8 | ) | — | (8 | ) | ||||||||||||||||||||||
| Net comprehensive loss | — | — | — | — | — | — | — | (28,060 | ) | |||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options | 115,606 | 1 | — | 158 | — | — | — | 159 | ||||||||||||||||||||||||
| Issuance of common stock pursuant to employee stock purchase plan | 59,368 | — | — | 132 | — | — | — | 132 | ||||||||||||||||||||||||
| Issuance of common stock pursuant to initial public offering, net of offering costs | 4,152,904 | 4 | — | 20,625 | — | — | — | 20,629 | ||||||||||||||||||||||||
| Conversion of redeemable convertible preferred stock into common stock pursuant to initial public offering | 8,346,316 | 8 | — | 46,878 | — | — | — | 46,886 | ||||||||||||||||||||||||
| Conversion of note payable into common stock pursuant to initial public offering | 1,000,000 | 1 | — | 5,999 | — | — | — | 6,000 | ||||||||||||||||||||||||
| Issuance of common stock pursuant to collaboration agreement | 637,755 | 1 | — | 5,009 | — | — | — | 5,010 | ||||||||||||||||||||||||
| Repayment of notes receivable from stockholders | — | — | 38 | — | — | — | — | 38 | ||||||||||||||||||||||||
| Elimination of deferred compensation upon adoption of SFAS 123R | — | — | — | (5,101 | ) | 5,101 | — | — | — | |||||||||||||||||||||||
| Compensation expense related to share-based payments | — | — | — | 4,539 | — | — | — | 4,539 | ||||||||||||||||||||||||
| Issuance of warrants to lenders | — | — | — | 6 | — | — | — | 6 | ||||||||||||||||||||||||
| Deemed dividend and accretion to redemption value of redeemable convertible preferred stock | — | — | — | — | — | — | (49 | ) | (49 | ) | ||||||||||||||||||||||
| Balance at December 31, 2006 | 15,166,109 | $ | 16 | $ | (21 | ) | $ | 177,355 | $ | — | $ | (8 | ) | $ | (163,729 | ) | $ | 13,613 | ||||||||||||||
See accompanying notes.
51
Table of Contents
SGX Pharmaceuticals, Inc.
| Years Ended December 31, | ||||||||||||
| 2006 | 2005 | 2004 | ||||||||||
| (In thousands) | ||||||||||||
| Operating activities: | ||||||||||||
| Net loss | $ | (28,052 | ) | $ | (29,391 | ) | $ | (18,752 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 3,332 | 3,976 | 5,002 | |||||||||
| Imputed interest expense on convertible debenture | — | — | 18 | |||||||||
| Stock-based compensation | 4,569 | 10,233 | 449 | |||||||||
| Issuance of common stock for services | 56 | — | — | |||||||||
| Amortization of discount on warrants | 167 | 72 | 64 | |||||||||
| Amortization of discount on warrants associated with bridge notes | — | 727 | 2,753 | |||||||||
| Deferred rent | (103 | ) | (97 | ) | (27 | ) | ||||||
| Accrual of interest on notes receivable from stockholders | — | — | (78 | ) | ||||||||
| Accrual of interest on bridge notes payable | — | 411 | 467 | |||||||||
| Forgiveness of principal and accrued interest on note settlement | — | — | 782 | |||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Accounts receivable | (2,669 | ) | 56 | 1,162 | ||||||||
| Prepaid expenses and other current assets | (452 | ) | (151 | ) | (60 | ) | ||||||
| Accounts payable and accrued liabilities | (40 | ) | 2,874 | (680 | ) | |||||||
| Deferred revenue | 15,223 | 737 | (2,643 | ) | ||||||||
| Other assets | 2,078 | (1,289 | ) | 559 | ||||||||
| Net cash used in operating activities | (5,891 | ) | (11,842 | ) | (10,984 | ) | ||||||
| Investing activities: | ||||||||||||
| Purchase of short-term investments | (6,000 | ) | — | — | ||||||||
| Purchases of property and equipment, net | (1,483 | ) | (1,303 | ) | (1,175 | ) | ||||||
| Net cash used in investing activities | (7,483 | ) | (1,303 | ) | (1,175 | ) | ||||||
| Financing activities: | ||||||||||||
| Proceeds from lines of credit and notes payable | 1,132 | 8,868 | 643 | |||||||||
| Principal payments on lines of credit and notes payable | (3,474 | ) | (2,974 | ) | (3,946 | ) | ||||||
| Proceeds from repayment of notes receivable from stockholders | 39 | 79 | 42 | |||||||||
| Issuance of common stock for cash, net of repurchases | 25,836 | 219 | 52 | |||||||||
| Issuance of preferred stock, net | — | 13,159 | (166 | ) | ||||||||
| Issuance of bridge notes | — | — | 13,411 | |||||||||
| Net cash provided by financing activities | 23,533 | 19,351 | 10,036 | |||||||||
| Net increase (decrease) in cash and cash equivalents | 10,159 | 6,206 | (2,123 | ) | ||||||||
| Cash and cash equivalents at beginning of year | 17,718 | 11,512 | 13,635 | |||||||||
| Cash and cash equivalents at end of year | $ | 27,877 | $ | 17,718 | $ | 11,512 | ||||||
| Supplemental schedule of cash flow information: | ||||||||||||
| Cash paid for interest | $ | 1,093 | $ | 363 | $ | 587 | ||||||
| Supplemental schedule of non-cash investing and financing activities: | ||||||||||||
| Conversion of preferred stock to common stock | $ | 46,886 | $ | — | $ | — | ||||||
| Conversion of note payable to common stock | $ | 6,000 | $ | — | $ | — | ||||||
| Reclass of deferred compensation to additional paid in capital | $ | 5,101 | $ | — | $ | — | ||||||
| Issuance of warrant related to line of credit | $ | 6 | $ | 498 | $ | — | ||||||
| Deferred compensation | $ | — | $ | 15,334 | $ | — | ||||||
| Conversion of bridge notes and redeemable convertible preferred stock to equity | $ | — | $ | 54,530 | $ | 13,785 | ||||||
| Conversion of bridge notes to preferred stock | $ | — | $ | 14,291 | $ | — | ||||||
See accompanying notes.
52
Table of Contents
SGX Pharmaceuticals, Inc.
(In thousands, except share and per share data)
| 1. | Organization and Summary of Significant Accounting Policies |
Organization and Business
SGX Pharmaceuticals, Inc. (“SGX” or the “Company”, formerly known as Structural GenomiX, Inc.), was incorporated in Delaware on July 16, 1998. SGX is a biotechnology company focused on the discovery, development and commercialization of innovative cancer therapeutics.
SGX is subject to risks common to companies in the biotechnology industry including, but not limited to, risks and uncertainties related to drug discovery, development and commercialization, obtaining regulatory approval of any products it or its collaborators may develop, competition from other biotechnology and pharmaceutical companies, its effectiveness at managing its financial resources, difficulties or delays in its clinical trials, difficulties or delays in manufacturing its clinical trial materials, implementation of its collaborations, the level of efforts that its collaborative partners devote to development and commercialization of its product candidates, its ability to successfully discover and develop products and market and sell any products it develops, the scope and validity of patent protection for its products and proprietary technology, dependence on key personnel, product liability, litigation, its ability to comply with U.S. Food and Drug Administration (“FDA”) and other government regulations and its ability to obtain additional funding to support its operations.
Principles of Consolidation
The consolidated financial statements include the assets, liabilities, and results of operations of the Company and its wholly-owned subsidiary. All material inter-company balances and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and disclosures made in the accompanying notes to the consolidated financial statements. Actual results could differ from those estimates.
Reverse Stock Split
In April 2005, the Company’s board of directors authorized a0.126453-for-1 reverse stock split for all outstanding preferred and common shares. All share information has been retroactively restated to reflect the reverse stock split.
In January 2006, the Company’s board of directors and stockholders authorized a1-for-2 reverse stock split of the common stock that was effected on January 3, 2006. As a result, each share of the Company’s then outstanding preferred stock became convertible into one-half of a share of the Company’s common stock. All common share information has been retroactively restated to reflect the1-for-2 reverse stock split. In connection with the closing of the Company’s initial public offering in February 2006, all shares of preferred stock were converted to common stock.
Initial Public Offering
The Company’s initial public offering of common stock (the “Offering”) was effected through a Registration Statement onForm S-1 (Filenumber 333-128059) that was declared effective by the Securities and Exchange Commission on January 31, 2006. The Offering commenced on February 1, 2006 and did not terminate before any securities were sold. On February 6, 2006, 4,000,000 shares of common stock were sold on the Company’s behalf at an initial public offering price of $6.00 per share, resulting in aggregate proceeds of approximately $19,772, net of
53
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
underwriting discounts and commissions and offering expenses. In addition, during March 2006, the Company closed the sale of an additional 152,904 shares of common stock pursuant to the exercise by the underwriters of an over-allotment option that resulted in additional net proceeds to us of $853, net of underwriting discounts and commissions and offering expenses.
Reclassification
Certain prior year amounts have been reclassified to conform to the current year presentation.
Cash and Cash Equivalents and Short-term Investments
The Company considers all highly liquid investments with original maturities of less than three months when purchased to be cash equivalents. Cash equivalents are recorded at cost, which approximate market value.
In accordance with Statement of Financial Accounting Standards (“SFAS”) No. 115,Accounting for Certain Investments in Debt and Equity Securities, the Company’s short-term investments are carried at fair value and classified as available-for-sale. Unrealized holding gains and losses, net of tax, are reported as a component of accumulated other comprehensive income in stockholders’ equity. Realized gains and losses and declines in value judged to be other-than-temporary on available-for-sale securities are included in interest income. The cost of the securities is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in interest income.
Fair Value of Financial Statements
The carrying value of cash equivalents, accounts receivable, accounts payable, accrued expenses and liabilities and notes payable are considered to be reasonable estimates of their respective fair values due to their short-term nature.
Accounts Receivable
The Company’s accounts receivable consist of amounts due from governmental agencies for costs incurred under funded projects and amounts due from corporate partners under various collaboration agreements. When necessary, the Company maintains an allowance for potentially uncollectible accounts receivable arising from its customers’ inability to make required payments. The Company has a limited number of accounts receivable and uses the specific identification method as a basis for determining this estimate. The Company did not maintain an allowance for doubtful accounts as of December 31, 2006 or 2005.
The percentage of total revenues from significant customers is as follows:
| Years Ended December 31, | ||||||||||||
| 2006 | 2005 | 2004 | ||||||||||
| Customer A | 31 | % | — | — | ||||||||
| Customer B | 28 | % | 33 | % | 42 | % | ||||||
| Customer C | 22 | % | 38 | % | 34 | % | ||||||
Property and Equipment
Property and equipment is stated at cost less accumulated depreciation and amortization. Costs of major additions and betterments are capitalized; maintenance and repairs, which do not improve or extend the life of the respective assets are charged as expenses. On disposal, the related cost and accumulated depreciation or amortization is removed from the accounts and any resulting gain or loss is included in the results of operations. Depreciation is computed using the straight-line method over the estimated useful lives of the assets which range
54
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
from three to fifteen years. Leasehold improvements are amortized over the estimated useful lives of the assets or related lease terms, whichever is shorter.
Long-Lived Assets
In accordance with Statement of Financial Accounting Standards (“SFAS”) No. 144,Accounting for the Impairment or Disposal of Long-Lived Assets, if indicators of impairment exist, the Company assesses the recoverability of the affected long-lived assets by determining whether the carrying value of such assets can be recovered through the undiscounted future operating cash flows. If impairment is indicated, the Company measures the amount of such impairment by comparing the carrying value of the asset to the present value of the expected future cash flows associated with the use of the asset. Although the Company has accumulated losses since inception, the Company believes the future cash flows to be received from the long-lived assets will exceed the assets’ carrying value, and accordingly, the Company has not recognized any impairment losses through December 31, 2006.
Deferred Rent
Rent expense is recorded on a straight-line basis over the term of the lease. The difference between rent expense accrued and amounts paid under the lease agreement is recorded as deferred rent in the accompanying consolidated balance sheets.
Redeemable Convertible Preferred Stock
In connection with the closing of the Company’s initial public offering in February 2006, all shares of redeemable convertible preferred stock were converted to common stock. Prior to the Company’s initial public offering, the carrying value of redeemable convertible preferred stock was increased by periodic accretions so that the carrying amount would equal the redemption value at the redemption date. These accretions were effected through charges against accumulated deficit.
Adoption of SFAS 123(R), Share-Based Payment
Effective January 1, 2006, the Company adopted the fair value recognition provisions of the Statement of Financial Accounting Standards (“SFAS”) No. 123 (revised 2004),Share-Based Payment(“SFAS 123(R)”) using the modified prospective method of recognition of compensation expense related to share-based payments. The Company’s statement of operations for the year ended December 31, 2006 reflects the impact of adopting SFAS 123R. Under this method, stock-based compensation expense recognized during the year ended December 31, 2006 includes: (a) compensation cost for all share-based payments granted prior to, but not yet vested as of January 1, 2006, based on the grant date fair value estimated in accordance with the original provisions of SFAS 123,Accounting for Stock-Based Compensation(“SFAS No. 123”) and (b) compensation cost for all share-based payments granted on or subsequent to January 1, 2006, based on the grant date fair value estimated in accordance with the provisions of SFAS 123R. In accordance with SFAS 123R, the estimated grant date fair value of each stock-based award is recognized as expense on a ratable basis over the requisite service period (generally the vesting period). In accordance with the modified prospective transition method, the consolidated statements of operations for the years ended December 31, 2005 and 2004 have not been restated to reflect, and do not include, the impact of SFAS 123R.
Expected volatility is based on average volatilities of the common stock of comparable publicly traded companies using a blend of historical, implied and average of historical and implied volatilities for this peer group of 10 companies.
As permitted by Staff Accounting Bulletin No. 107,Share-Based Payment (“SAB107”), the Company utilized the “shortcut approach” to estimate an option’s expected term, which represents the period of time that an option granted is expected to be outstanding. The expected term of options granted is derived from the average midpoint
55
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
between vesting and the contractual term. The risk-free interest rate is based on the average of the 5-year and 7-year U.S. Treasury yield curve at the beginning of each month for that month’s options granted, given their expected term. The rates ranged from 4.31% to 5.12% during 2006.
Since the Company has never paid a dividend and does not expect to pay dividends in the near future, the Company uses a zero dividend rate.
As stock-based compensation expense recognized in the Statements of Operations for the year ended December 31, 2006 is based on awards ultimately expected to vest, it should be reduced for estimated forfeitures. SFAS 123R requires forfeitures to be estimated at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. Pre-vesting forfeitures were estimated to be approximately 12% during the year ended December 31, 2006, based on historical experience. In the Company’s pro forma information required under SFAS 123 for the periods prior to fiscal 2006, the Company accounted for forfeitures as they occurred. Compensation expense related to stock-based compensation is recognized on a ratable basis for those awards issued on January 1, 2006 or later. Compensation expense for awards issued prior to January 1, 2006 is continuing to be recognized on an accelerated method until vesting is complete. Compensation expense related to stock-based compensation is allocated to research and development or general and administrative expense based upon the department to which the associated employee or non-employee reports.
The adoption of SFAS 123R did not impact the cash flow from operations, investing or financing activities during the year ended December 31, 2006.
Stock-Based Compensation prior to SFAS 123R
In 2005 and prior years, the Company elected to follow Accounting Principles Board (“APB”) Opinion No. 25,Accounting for Stock Issued to Employees(“APB 25”), and related Interpretations in accounting for its employee and director stock options, and provided the required pro forma disclosures of Statement of Financial Accounting Standards (“SFAS”) No. 123,Accounting for Stock-Based Compensation (“SFAS No. 123”). Under APB 25, if the exercise price of the Company’s employee and director stock options equaled or exceeded the estimated fair value of the underlying stock on the date of grant, no compensation expense was recognized.
Options or stock awards issued to non-employees were recorded at their fair value as determined in accordance withSFAS No. 123, and Emerging Issues Task Force (“EITF”)96-18,Accounting for Equity Instruments That are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling Goods and Services, were periodically revalued as the options vested and recognized as expense over the related service period.
The Company’s board of directors estimated the fair value of the Company’s common stock for purposes of establishing exercise prices of stock options. Given the absence of an active market for the Company’s common stock through its initial public offering in February 2006, the board of directors considered, among other factors, the liquidation preferences, anti-dilution protection and voting preferences of the preferred stock over the common stock in determining the estimated fair value of the common stock for purposes of establishing the exercise prices for stock option grants.
In preparation for its initial public offering, the Company revised its estimate of the fair value for financial reporting purposes of common stock for the last six months of 2004, all of 2005, and January of 2006. This valuation was done retrospectively by management, and the Company did not obtain contemporaneous valuations from an independent valuation specialist. In reassessing the value of common stock in 2004, 2005 and January 2006, the Company considered the price it received in April 2005 for its Series B preferred stock of $4.71 per share ($9.42 per share on an assumed converted basis). Starting on July 1, 2004, the Company reduced the value originally attributed to the preferences on the Series B preferred stock to 10% of the price of the preferred stock. Accordingly, the Company estimated the fair value of the common stock for financial reporting purposes to be 90% of the Series B preferred stock price, or $4.24 per share ($8.48 per share on an assumed converted basis). The Company kept this value constant until April 2005, when the Company steadily increased the estimated fair value to $14.06 per
56
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
common share based on an assessment of market considerations, including discussions with the underwriters in the initial public offering. The Company believed this valuation approach was consistent with valuation methodologies applied for financial reporting purposes to other similar companies pursuing an initial public offering.
The Company recorded deferred stock compensation, net of forfeitures, for employee and non-employee director’s stock option and restricted stock grants within stockholders’ deficit of $13,598 and $0 in 2005 and 2004, respectively, which represents the difference between the revised fair value of the common stock for financial reporting purposes and the option exercise price at the date of grant. The weighted-average exercise price and the weighted-average revised fair value were $1.00 and $11.40 for the options granted during 2005, respectively. Deferred compensation was to be amortized to expense over the vesting period of the related options using an accelerated method in accordance with Financial Accounting Standards Board Interpretation (“FIN”) No. 28,Accounting for Stock Appreciation Rights and Other Variable Stock Option or Award Plans.
The Company recorded amortization of deferred stock compensation of $10,233 and $449 during the years ended December 31, 2005 and 2004, respectively. On January 1, 2006, the Company reversed out the remaining deferred stock compensation of $5,101 to additional paid-in capital in accordance with SFAS 123R.
The table below illustrates the effect on net loss and net loss per share had the Company applied the fair value provisions of SFAS No. 123 to employee stock compensation in 2005 and 2004.
| 2005 | 2004 | |||||||
| Net loss attributable to common stockholders, as reported | $ | (29,863 | ) | $ | (19,081 | ) | ||
| Add: Stock-based employee compensation expense included in net loss attributable to common stockholders | 8,785 | 121 | ||||||
| Deduct: Stock-based employee compensation determined under the fair value method | (6,859 | ) | (262 | ) | ||||
| Pro forma net loss attributable to common stockholders | $ | (27,937 | ) | $ | (19,222 | ) | ||
| Basic and diluted net loss attributable to common stockholders per share, as reported | $ | (48.32 | ) | $ | (39.84 | ) | ||
| Pro forma basic and diluted net loss attributable to common stockholders per share | $ | (45.21 | ) | $ | (40.13 | ) | ||
The fair value of these stock option and restricted stock grants used to compute pro forma net loss is estimated at the date of grant, using a Black-Scholes option pricing model with the following weighted-average assumptions for each of the two years in the period ended December 31, 2005:
| 2005 | 2004 | |||||||
| Risk-free interest rate | 3 | % | 3 | % | ||||
| Expected volatility | 63 | % | 63 | % | ||||
| Expected lives | 4 years | 4 years | ||||||
| Expected dividend | — | — | ||||||
The Black-Scholes option valuation model was developed for use in estimating the fair value of traded options that have no vesting restrictions and are fully transferable. In addition, option valuation models require the input of highly subjective assumptions. Because the Company’s employee stock option and restricted stock grants have characteristics significantly different from those of traded options, and because changes in the subjective input assumptions can materially affect the fair value estimates, in management’s opinion, the existing models do not necessarily provide a reliable single measure of the fair value of its employee stock options and restricted stock grants.
57
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Revenue Recognition
The Company’s collaboration agreements and commercial agreements contain multiple elements, including non-refundable upfront fees, payments for reimbursement of research costs, payments for ongoing research, payments associated with achieving specific milestones and, in the case of collaboration agreements, development milestones and royalties based on specified percentages of net product sales, if any. The Company applies the revenue recognition criteria outlined in Staff Accounting Bulletin No. 104,Revenue Recognition,and EITF Issue00-21,Revenue Arrangements with Multiple Deliverables(“EITF00-21”). In applying these revenue recognition criteria, the Company considers a variety of factors in determining the appropriate method of revenue recognition under these arrangements, such as whether the elements are separable, whether there are determinable fair values and whether there is a unique earnings process associated with each element of a contract.
Cash received in advance of services being performed is recorded as deferred revenue and recognized as revenue as services are performed over the applicable term of the agreement.
When a payment is specifically tied to a separate earnings process, revenues are recognized when the specific performance obligation associated with the payment is completed. Performance obligations typically consist of significant and substantive milestones pursuant to the related agreement. Revenues from non-refundable milestone payments may be considered separable from funding for research services because of the uncertainty surrounding the achievement of milestones for products in early stages of development. Accordingly, these payments could be recognized as revenue if and when the performance milestone is achieved if they represent a separate earnings process as described in EITF00-21.
In connection with certain research collaborations and commercial agreements, revenues are recognized from non-refundable upfront fees, which the Company does not believe are specifically tied to a separate earnings process, ratably over the term of the agreement. Research services provided under some of the Company’s agreements are on a fixed fee basis. Revenues associated with long-term fixed fee contracts are recognized based on the performance requirements of the agreements and as services are performed.
Revenues derived from reimbursement of directout-of-pocket expenses for research costs associated with grants are recorded in compliance with EITF Issue99-19,Reporting Revenue Gross as a Principal Versus Net as an Agent(“EITF 99-19”), and EITF Issue01-14,Income Statement Characterization of Reimbursements Received for“Out-of-Pocket” Expenses Incurred(“EITF01-14”). According to the criteria established by these EITF Issues, in transactions where the Company acts as a principal, with discretion to choose suppliers, bears credit risk and performs part of the services required in the transaction, the Company records revenue for the gross amount of the reimbursement. The costs associated with these reimbursements are reflected as a component of research and development expense in the statements of operations.
None of the payments that the Company has received from collaborators to date, whether recognized as revenue or deferred, is refundable even if the related program is not successful.
Research and Development
Research and development costs are expensed as incurred and consist primarily of costs associated with clinical trials, compensation, including stock-based compensation, and other expenses related to research and development, including personnel costs, facilities costs and depreciation.
Income Taxes
The Company accounts for income taxes using the liability method in accordance with the provisions of SFAS No. 109,Accounting for Income Taxes. Under this method, deferred tax assets and liabilities are determined based on differences between the financial reporting and tax basis of the Company’s assets and liabilities and are estimated using enacted tax rates and laws that will be in effect when the differences are expected to reverse. A
58
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
valuation allowance is provided when the Company determines that it is more likely than not that some portion or all of a deferred tax asset will not be realized.
Net Loss Per Share Attributable to Common Stockholders
The Company computes net loss per share attributable to common stockholders in accordance with SFAS No. 128,Earnings Per Share(“SFAS 128”). Under the provisions of SFAS 128, basic net loss per share attributable to common stockholders (“Basic EPS”) is computed by dividing net loss attributable to common stockholders by the weighted average number of common shares outstanding. Diluted net loss per share attributable to common stockholders (“Diluted EPS”) is computed by dividing net loss attributable to common stockholders by the weighted average number of common shares and dilutive common share equivalents then outstanding. Common share equivalents consist of the incremental common shares issuable upon the conversion of preferred stock and note payable, shares issuable upon the exercise of stock options, vesting of restricted stock units, and shares issuable upon the exercise of warrants. For the periods presented, Diluted EPS is identical to Basic EPS because common share equivalents, including all of the Company’s preferred stock, note payable, outstanding stock options and outstanding warrants, are excluded from the calculation, as their effect is anti-dilutive. Had the Company been in a net income position, these securities may have been included in the calculation. These potentially dilutive securities consist of the following on a weighted average basis:
| Years Ended December 31, | ||||||||||||
| 2006 | 2005 | 2004 | ||||||||||
| Redeemable convertible preferred stock | — | 5,686,849 | 993,761 | |||||||||
| Notes payable | — | 1,000,000 | 1,000,000 | |||||||||
| Outstanding common stock options | 604,869 | 636,927 | 183,881 | |||||||||
| Restricted stock | 145,727 | 28,971 | — | |||||||||
| Outstanding warrants | 195,928 | 70,645 | 10,479 | |||||||||
| Total | 946,524 | 7,423,392 | 2,188,121 | |||||||||
Upon the completion of the Company’s initial public offering in February 2006, all of the Company’s previously outstanding preferred shares converted into 8,346,316 shares of the Company’s common stock and a note payable converted into 1.0 million shares of the Company’s common stock.
Comprehensive Income (Loss)
SFAS No. 130,Reporting Comprehensive Income, requires that all components of comprehensive income, including net income, be reported in the financial statements in the period in which they are recognized. Comprehensive income (loss) is defined as the change in equity during a period from transactions and other events and circumstances from non-owner sources. Net income (loss) and other comprehensive income (loss), including foreign currency translation adjustments and unrealized gains and losses on investments, are to be reported, net of their related tax effect, to arrive at comprehensive income (loss).
Segment Reporting
The Company has adopted SFAS No. 131,Disclosure About Segments of an Enterprise and Related Information, which requires companies to report selected information about operating segments, as well as enterprise wide disclosures about products, services, geographical areas, and major customers. Operating segments are determined based on the way management organizes its business for making operating decisions and assessing performance. The Company has only one operating segment, the discovery, development and commercialization of drug products, thus, this statement did not have an impact on the Company’s financial statements.
59
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Recently Issued Accounting Standards
In July 2006, the Financial Accounting Standards Board (FASB) issued FASB Interpretation (FIN) No. 48,Accounting for Uncertainty in Income Taxes — an interpretation of FASB Statement No. 109 (“FIN48”), which clarifies the accounting for uncertainty in tax positions. FIN 48 requires that the Company recognize the impact of a tax position in its financial statements if that position is more likely than not of being sustained on audit, based on the technical merits of the position. The provisions of FIN 48 are effective as of the beginning of the Company’s 2007 fiscal year, with the cumulative effect of the change in accounting principle recorded as an adjustment to opening retained earnings. The Company does not expect the adoption of FIN 48 to have a material impact on its consolidated results of operations and financial position, and the Company is continuing to evaluate the impact, if any, the adoption of FIN No. 48 will have on its disclosure requirements.
In September 2006, the SEC issued Staff Accounting Bulletin No. 108 (SAB 108) Considering the Effects of Prior Year Misstatements when Quantifying Misstatements in Current Year Financial Statement. SAB 108 addresses how the effects of prior year uncorrected misstatements should be considered when quantifying misstatements in current year financial statements. SAB 108 requires companies to quantify misstatements using a balance sheet and income statement approach and to evaluate whether either approach results in quantifying an error that is material in light of relevant quantitative and qualitative factors. When the effect of initial adoption is material, companies will record the effect as a cumulative effect adjustment to beginning of year retained earnings. The provisions of SAB 108 are effective for annual statements covering the first fiscal year ending after November 15, 2006. The adoption of SAB 108 did not have a material impact on the Company’s financial position or results of operations.
In September 2006, the FASB issued SFAS No. 157,Fair Value Measurement (“SFAS 157”). SFAS 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles and expands disclosures about fair value measurements. This Statement applies only to fair value measurements that are already required or permitted by other accounting standards. Accordingly, this Statement does not require any new fair value measurements. SFAS 157 is effective for fiscal years beginning after December 15, 2007. The Company is currently evaluating the impact, if any, the adoption of SFAS 157 will have on the Company’s financial position or results of operations.
| 2. | Balance Sheet Details |
Short-term Investments
Short-term investments at December 31, 2006 consisted of two auction rate securities totaling $6,000. There was no unrealized gain or loss associated with these securities as of December 31, 2006.
Property and Equipment
Property and equipment consist of the following:
| Estimated | December 31, | |||||||||||
| Life in Years | 2006 | 2005 | ||||||||||
| Lab equipment | 5-7 | $ | 14,066 | $ | 12,573 | |||||||
| Computers and equipment | 3-5 | 7,963 | 7,676 | |||||||||
| Leasehold improvements | 4-15 | 5,139 | 4,935 | |||||||||
| Furniture | 10 | 411 | 411 | |||||||||
| Construction in progress | NA | 255 | 756 | |||||||||
| 27,834 | 26,351 | |||||||||||
| Accumulated depreciation and amortization | (22,399 | ) | (19,183 | ) | ||||||||
| $ | 5,435 | $ | 7,168 | |||||||||
60
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Total depreciation expense related to property and equipment was $3,216, $3,394 and $4,797 for the years ended December 31, 2006, 2005 and 2004, respectively, which is net of asset dispositions. Cost and accumulated depreciation of assets under equipment lines of credit was $16,980 and $14,090, respectively, at December 31, 2006, and $15,848 and $12,590, respectively, at December 31, 2005. Depreciation of assets under equipment lines of credit is included in depreciation expense.
A majority of the Company’s property and equipment collateralizes the outstanding obligations under the existing line of credit agreements as of December 31, 2006 and 2005.
Goodwill and Intangible Assets
Intangible assets include the following:
| December 31, 2006 | ||||||||||||
| Gross | ||||||||||||
| Estimated | Carrying | Accumulated | ||||||||||
| Life in Years | Amount | Amortization | ||||||||||
| Goodwill | Indefinite | $ | 3,914 | $ | (522 | ) | ||||||
| Licenses | 3 - 10 | 989 | (969 | ) | ||||||||
| Total intangible assets | $ | 4,903 | $ | (1,491 | ) | |||||||
| December 31, 2005 | ||||||||||||
| Gross | ||||||||||||
| Estimated | Carrying | Accumulated | ||||||||||
| Life in Years | Amount | Amortization | ||||||||||
| Goodwill | Indefinite | $ | 3,914 | $ | (522 | ) | ||||||
| Licenses | 3 - 5 | 977 | (930 | ) | ||||||||
| Total intangible assets | $ | 4,891 | $ | (1,452 | ) | |||||||
The net amortization expense of intangible assets, excluding goodwill, done on a straight-line basis for the years ended December 31, 2006 and 2005 was approximately $39 and $176, respectively.
Estimated amortization of intangibles for the years ending December 31:
| 2007 | $ | 10 | ||
| 2008 | 1 | |||
| 2009 | 1 | |||
| 2010 | 1 | |||
| 2011 | 1 | |||
| Thereafter | 6 | |||
| $ | 20 | |||
| 3. | Lines of Credit |
In September 2002, the Company entered into a line of credit agreement under which it could borrow up to $6,500 to finance equipment. Borrowings under the line of credit bear interest at rates ranging between 9.14% and 10.60% per annum and are collateralized solely by the financed equipment. Principal and interest are payable monthly over either 35 months or 47 months depending on the type of equipment financed. The line of credit requires the Company to execute a letter of credit in favor of the finance company in the amount of $150. As of December 31, 2006, there are no amounts available for future draws under this line of credit.
61
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
In September 2005, the Company entered into a line of credit and equipment financing agreement with Silicon Valley Bank and Oxford Finance Corporation to provide $8,000 of general purpose working capital financing and $2,000 of equipment and leasehold improvements financing. The debt bears interest at a rate of approximately 10% per annum and is due in monthly installments over three years. One-half of the proceeds were immediately made available to the Company under the line of credit and equipment financing agreements. The remainder of the line of credit became available in the fourth quarter of 2005 and the remainder of the equipment financing agreement became available in the second quarter of 2006.
In September 2005, the Company borrowed $4,000 for general purpose working capital under the line of credit facility. In December 2005, the Company borrowed an additional $4,000 for general purpose working capital purposes under the line of credit facility and $867 for equipment financing purposes. In November and December 2006, the Company borrowed the remainder of the available financing of $1,132 under the equipment financing agreement. These debt agreements subject us to certain financial and non-financial covenants. As of December 31, 2006, we were in compliance with these covenants. These obligations are secured by our assets, excluding intellectual property, and are due in monthly installments through 2010. They bear interest at effective rates ranging from approximately 9.14% to 11.03% and are subject to prepayment fees of up to 4% of the outstanding principal balance as of the prepayment date. We made debt repayments of approximately $3,474, $2,974, and $3,946 for the years ended December 31, 2006, 2005, and 2004, respectively.
The facility with Silicon Valley Bank and Oxford Finance Corporation is subject to a “material adverse event” clause and the Company’s cash and cash equivalent accounts are subject to the control of the lenders if a “material adverse event” occurs. In accordance with the provisions of EITFNo. 95-22,Balance Sheet Classifications of Borrowings Outstanding under Revolving Credit Agreements that include both a Subjective Acceleration Clause and a Lock-Box Arrangementand FASB TechnicalBulletin No. 79-3,Subjective Acceleration Clauses in Long-Term Debt Agreements, the Company has classified the borrowings outstanding under this arrangement as a current liability in the consolidated balance sheets as of December 31, 2006 and 2005.
Future minimum principal payments due on the above equipment and working capital lines of credit as of December 31, 2006 are as follows:
| 2007 | $ | 3,526 | ||
| 2008 | 3,661 | |||
| 2009 | 665 | |||
| 2010 | 18 | |||
| Total | $ | 7,870 | ||
The aggregate amount of debt discount at December 31, 2006 that will be amortized to interest expense in future periods is approximately $319.
| 4. | Commitments and Collaborative Research and Development Agreements |
The Company leases its office and research facilities and certain office equipment under non-cancelable operating leases, which expire at various dates from 2007 to 2010. The leases include escalation clauses beginning on the first anniversary of the respective lease and continuing through the end of the leases. The leases require the Company to pay for all maintenance, insurance and property taxes. In addition to a cash security deposit, one of the leases required the Company to execute a letter of credit in favor of its landlord in the amount of $88.
In accordance with the letter of credit agreements, the Company was required to restrict cash equal to the amount of the letters of credit. As of December 31, 2005, restricted cash of $88 was included in cash and cash equivalents. The letter of credit requirement ended in March 2006 upon the expiration of the related lease. Accordingly, no cash was restricted at December 31, 2006.
62
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Future minimum lease payments are as follows at December 31, 2006:
| 2007 | $ | 1,417 | ||
| 2008 | 717 | |||
| 2009 | 23 | |||
| 2010 | 19 | |||
| Total minimum lease payments | $ | 2,176 | ||
Rent expense for the years ended December 31, 2006, 2005 and 2004 was $1,772, $2,203 and $2,075, respectively.
Bridge Financing
In July and September 2004, the Company entered into a Loan and Security Agreement (the “Loan and Security Agreement”) whereby the Company borrowed from certain preferred stockholders an aggregate principal amount of approximately $13,411 under Secured Convertible Promissory Notes (the “Secured Bridge Notes”) and issued to those preferred stockholders warrants (the “Bridge Warrants”) to purchase shares of common stock of the Company (the “Bridge Financing”). In conjunction with the Bridge Financing, the Company concurrently entered into an Intellectual Property Security Agreement pursuant to which the Company granted and pledged a security interest in its intellectual property and substantially all of the Company’s assets as collateral.
The Secured Bridge Notes had an annual interest rate of 10%. The principal and accrued interest under the Secured Bridge Notes converted into shares ofSeries A-2 preferred stock in connection with the initial closing of the Series B preferred stock financing. (See Note 5)
The shares of preferred stock of any preferred stockholder that did not participate at least 50% of their pro rata amount in the Bridge Financing were automatically converted into shares of common stock upon the closing of the Bridge Financing. An aggregate of 388,104 shares of Series A, B, C and D preferred stock were converted into common stock as a result of nonparticipation in the Bridge Financing by certain preferred stockholders. For those preferred stockholders that did participate in the Bridge Financing, their shares of Series A, B, C and D preferred stock were exchanged for shares ofSeries A-1, B-1, C-1 and D-1 preferred stock, respectively, on aone-for-one basis. As a result, the Company’s outstanding capital stock at December 31, 2004 consisted of common stock,Series A-1, B-1, C-1 and D-1 preferred stock. (See Note 5)
In January 2005, the Company, the agent and the lenders party to the Loan and Security Agreement holding a majority interest of the outstanding principal amount under all of the Secured Bridge Notes (the “Majority Lenders”) agreed to extend the maturity date of the Secured Bridge Notes from January 27, 2005 to March 31, 2005, or such earlier date as may be determined by the Majority Lenders. Subsequently, the Company, the agent and the Majority Lenders agreed to further extend the maturity date of the Secured Bridge Notes from March 31, 2005 to April 22, 2005, or such earlier date as may be determined by the Majority Lenders.
The Company determined the fair value of the Bridge Warrants on the grant date, using the Black-Scholes pricing model with a resulting aggregate expense of approximately $1,739, which was recorded against the principal balance and was amortized over the term of the Secured Bridge Notes. Of the debt discount, approximately $363 and $1,376 was recognized as interest expense during the years ended December 31, 2005 and 2004, respectively. Pursuant to EITF IssueNo. 98-5,“Accounting for Convertible Securities with Beneficial Conversion Features”, and EITF IssueNo. 00-27,“Application of IssueNo. 98-5 to Certain Convertible Instruments”, the Company recorded an additional non-cash charge of approximately $1,739 against the principal balance of the Secured Bridge Notes. This amount represents the difference between the conversion price of the Secured Bridge Notes and the underlying value of the stock issuable upon conversion of the Secured Bridge Notes. Of this non-cash charge, approximately $363 and $1,376 was recognized as interest expense during the years ended December 31, 2005 and 2004, respectively.
63
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Note Payable
In December 2001, the Company entered into a research program agreement with Millennium Pharmaceuticals, Inc. (“Millennium”). Concurrent with the signing of the research program agreement, the Company issued to Millennium a convertible note with a term of three years in exchange for $6,000.
During 2004, the Company issued an amended and restated convertible promissory note to Millennium and the entire note balance was converted into the right to receive common stock upon the closing of an initial public offering at a conversion price equal to the price per share in the offering. As the note payable was to be settled in a variable number of shares upon conversion due to an initial public offering, as of December 31, 2005, the Company reflected the amount as a long-term liability until settled in accordance with SFAS No. 150. In connection with the closing of the Company’s initial public offering in February 2006, the note automatically converted into an aggregate of 1,000,000 shares of the Company’s common stock.
Sponsored Research and Drug Discovery Collaboration Agreements with Cystic Fibrosis Foundation Therapeutics, Inc.
In January 2001, the Company entered into a sponsored research agreement with Cystic Fibrosis Foundation Therapeutics, Inc. (“CFFT”), the drug discovery and development arm of the Cystic Fibrosis Foundation. Through December 31, 2006, the Company recognized revenue of $11,905 related to research funding and $1,475 related to the achievement of eight milestones. In July 2005, the Company entered into a new three-year drug discovery collaboration agreement with CFFT.
Collaboration and License Agreement with Eli Lilly and Company
In April 2003, the Company entered into a two-year research and technology agreement with Eli Lilly and Company (“Eli Lilly”). Under the terms of the agreement, the Company has received research, license, technology access and technology installation fees of $25,476 through December 31, 2006. These payments were initially recorded as deferred revenue and recognized as collaborations and commercial agreements revenue as services were performed pursuant to the agreement.
In December 2003, the Company also expanded its research and technology agreement with Eli Lilly to provide Eli Lilly with long-term access to its beamline facility at the Advanced Photon Source in Argonne, Illinois, to support Eli Lilly drug discovery programs. Under the terms of the Company’s beamline services agreement with Eli Lilly, the Company generates crystal structure data on Eli Lilly drug targets and compounds in exchange for upfront access fees and maintenance fees paid by Eli Lilly. Upon execution of the agreement, the Company received a $2,000 upfront access fee payment and will receive payments for annual operating costs in future years.
In April 2005, the research term of the agreement was extended for an additional three years. The Company is entitled to receive research funding of approximately $4,600 per year.
In-Licensing of Troxatyltm
In July 2004, the Company licensed exclusive worldwide rights to Troxatyl from Shire BioChem Inc. (“Shire”). Troxatyl is a novel compound that has been in clinical trials for the treatment of acute myelogenous leukemia (“AML”). Under the terms of the agreement, the Company made an upfront payment of $3,000 and a payment of $1,000 on the one-year anniversary of the agreement. The Company is also required to make up to $17,000 of contingent milestone payments based on successful development and approval of Troxatyl for the treatment of AML. The Company may also be required to make milestone payments upon the occurrence of other development and regulatory events for solid tumor and other indications, and will be required to make minimum royalty payments based on net sales of up to approximately $10,000 over a four-year period following product launch. In August 2006, the Company announced that it had discontinued its Phase II/III clinical trial of Troxatyl for the third line treatment of acute myelogenous leukemia based on a recommendation from the study’s independent
64
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
safety and monitoring board. As a result, no estimate of the amount or timing of any of these other potential payments has been accrued for in the accompanying consolidated financial statements as the Company has not yet completed clinical development or obtained regulatory approval of Troxatyl for any indication. A one-time charge of $4,000 for purchased in-process research and development related to the upfront and one-year anniversary payments has been reflected in the Statement of Operations for the year ended December 31, 2004, based on the fact that the technology acquired did not have established feasibility and had no alternative future use.
License and Collaboration Agreement with Novartis
In March 2006, the Company entered into a License and Collaboration Agreement (the “Agreement”) with Novartis Institutes for Biomedical Research, Inc., (“Novartis”) for the development and commercialization of BCR-ABL inhibitors for the treatment of Chronic Myelogenous Leukemia (CML). Under the Agreement, the parties will collaborate to develop one or more BCR-ABL inhibitors and Novartis will have exclusive worldwide rights to such compounds, subject to the Company’s co-commercialization option in the United States and Canada.
Under the terms of the agreement, the Company received an upfront payment of $20,000, and will receive research funding over the first two years of the collaboration of $9,100 and could receive additional payments for achievement of specified development, regulatory and commercial milestones. In addition, the Company sold Novartis Pharma AG $5,000 of common stock pursuant to a Stock Purchase Agreement dated March 2006.
| 5. | Redeemable Convertible Preferred Stock |
Redeemable Convertible Preferred Stock
During the year ended December 31, 2002, 12,400 shares of Series D redeemable preferred stock were cancelled due to being unearned under an earnout agreement. There were no changes to the redeemable convertible preferred stock in 2003. A summary of redeemable convertible preferred stock issued and outstanding as of December 31, 2002 and 2003 is as follows:
| Aggregate Liquidation | ||||||||
| Shares Issued and | Preference and | |||||||
| Outstanding | Redemption Value | |||||||
| Series A | 541,594 | $ | 7,709 | |||||
| Series B | 809,299 | 32,000 | ||||||
| Series C | 673,419 | 45,000 | ||||||
| Series D | 129,692 | 5,405 | ||||||
| 2,154,004 | $ | 90,114 | ||||||
During the year ended December 31, 2004, the following shares of redeemable convertible preferred stock were converted into shares of common stock for non-participation in the Bridge Financing:
| Series A | 114,159 | |||
| Series B | 167,684 | |||
| Series C | 69,206 | |||
| Series D | 37,055 | |||
| Total | 388,104 | |||
65
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
The remaining shares of Series A, B, C and D preferred stock were exchanged forSeries A-1, B-1, C-1 and D-1 preferred stock, respectively, on aone-for-one basis upon the closing of the Bridge Financing. A summary of redeemable convertible preferred stock issued and outstanding as of December 31, 2004 was as follows:
| Aggregate Liquidation | ||||||||
| Shares Issued and | Preference and | |||||||
| Outstanding | Redemption Value | |||||||
| Series A-1 | 427,435 | $ | 6,084 | |||||
| Series B-1 | 641,615 | 25,370 | ||||||
| Series C-1 | 604,213 | 40,375 | ||||||
| Series D-1 | 92,637 | 3,861 | ||||||
| 1,765,900 | $ | 75,690 | ||||||
As of December 31, 2004, theSeries A-1, B-1, C-1 and D-1 preferred stock were convertible at the option of the holder on aone-for-one basis, subject to adjustment for dilution, into a total of 1,765,900 shares of common stock. In addition, the terms of the preferred stock provided that the preferred stock would automatically convert into shares of common stock upon the closing of an underwritten public offering of equity securities which results in a minimum per share purchase price of $16.90 with net proceeds of at least $25,000, or upon a vote of the holders of more than 50% of the preferred stock then outstanding. The holder of each share of preferred stock was entitled to one vote for each share of common stock into which it would convert. The terms of the preferred stock also provided that on any date after September 12, 2005 and on each of the first and second anniversaries thereof, upon approval of at least two-thirds of the then outstanding shares of preferred stock, such shares could be redeemed in three equal annual installments. The Company was required to affect redemptions by paying cash in an amount equal to $14.23, $39.54, $66.82 and $41.68 per share forSeries A-1, B-1, C-1 and D-1 preferred stock, respectively, plus any declared but unpaid dividends.
Holders of the shares of preferred stock were entitled to receive non-cumulative dividends at an annual rate of $1.14, $3.16, $5.35 and $0.44 per share ofSeries A-1, B-1, C-1 and D-1 preferred stock, respectively, as adjusted for stock splits, stock combinations and stock dividends. To date, the Company has not declared any dividends.
In the event of liquidation, the preferred stockholders were entitled to receive a liquidation preference equal to the original issuance price plus declared but unpaid dividends. The liquidation preference had priority over all distributions to common stockholders. After payment of the liquidation preference, all remaining assets from liquidation were to be paid to the preferred stockholders and common stockholders according to the number of shares held. However, the total amounts that could be distributed (including all amounts payable under the liquidation preference) to the holders ofSeries A-1, B-1, C-1 and D-1 preferred stock would not exceed $42.70, $118.62, $200.47 and $125.00 per share, respectively. All remaining amounts would be distributed ratably to the holders of common stock.
In April 2005, the principal and accrued interest under the Secured Bridge Notes were converted into 3,034,095 shares ofSeries A-2 preferred stock. Subsequent to the conversion of the Secured Bridge Notes, the holders ofSeries A-1, B-1, C-1, and D-1 preferred stock exchanged their 1,765,900 shares ofSeries A-1, B-1, C-1 and D-1 preferred stock, together with their 3,034,095 shares ofSeries A-2 preferred stock, for an aggregate of 13,600,000 shares of new Series A preferred stock, and 388,104 shares of common stock were issued upon conversion of shares ofSeries A-1, B-1, C-1 or D-1 preferred stock that were not exchanged for shares of new Series A preferred stock. Also in 2005, the Company sold 3,175,981 shares of new Series B preferred stock for
66
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
$4.71 per share for net proceeds of $13,287. As a result, a summary of redeemable convertible preferred stock issued and outstanding as of December 31, 2005 is as follows:
| Aggregate Liquidation | ||||||||
| Shares Issued and | Preference and | |||||||
| Outstanding | Redemption Value | |||||||
| Series A (New) | 13,525,400 | $ | 33,272 | |||||
| Series B (New) | 3,167,254 | 14,918 | ||||||
| 16,692,654 | $ | 48,190 | ||||||
As of December 31, 2005, the 16,692,654 shares of Series A (new) and Series B (new) preferred stock were convertible into a total of 8,346,316 shares of common stock. The terms of the preferred stock provided that the preferred stock would automatically convert into shares of common stock upon the closing of an underwritten public offering of equity securities which results in a minimum per share purchase price of $5.00 with net proceeds of at least $25,000, or upon a vote of the holders of more than 50% of the preferred stock then outstanding. The shares of preferred stock had a liquidation preference and redemption value of $2.46 and $4.71 per share of Series A (new) and Series B (new) preferred stock, respectively, plus any declared but unpaid dividends. In connection with closing of the Company’s initial public offering in February 2006, all shares of preferred stock were converted into 8,346,316 shares of common stock.
| 6. | Equity Incentive Plans and Warrants |
2000 Equity Incentive Plan
In February 2000, the Company adopted its 2000 Equity Incentive Plan (the “Equity Incentive Plan”). The Equity Incentive Plan provides for the grant of up to 1,755,000 shares pursuant to incentive and non-statutory stock options, stock bonuses or sales of restricted stock. Options granted under the Equity Incentive Plan generally expire no later than ten years from the date of grant (five years for a 10% stockholder). Options generally vest over a period of four years. The exercise price of incentive stock options must be equal to at least the fair value of the Company’s common stock on the date of grant, and the exercise price of non-statutory stock options may be no less than 85% of the fair value of the Company’s common stock on the date of grant. The exercise price of any option granted to a 10% stockholder may not be less than 110% of the fair value of the Company’s common stock on the date of grant. On February 1, 2006, the effective date of the Company’s initial public offering, the 2000 plan terminated and the remaining 249,427 shares available under the plan were transferred to the 2005 plan.
2005 Equity Incentive Plan
The Company adopted in August 2005, and the stockholders approved in October 2005, the 2005 equity incentive plan (the “2005 Plan”). The 2005 Plan became effective upon the effectiveness of the Company’s initial public offering (see Note 1). An aggregate of 750,000 shares of the Company’s common stock are authorized for issuance under the 2005 Plan, plus the number of shares remaining available for future issuance under the Equity Incentive Plan that are not covered by outstanding options as of the termination of the Equity Incentive Plan on the effective date of the initial public offering. In addition, this amount will be automatically increased annually on the first day of the Company’s fiscal year, from 2007 until 2015, by the lesser of (a) 3.5% of the aggregate number of shares of common stock outstanding on December 31 of the preceding fiscal year or (b) 500,000 shares of common stock. Options granted under the 2005 Plan generally expire no later than ten years from the date of grant (five years for a 10% stockholder). Options generally vest over a period of four years. The exercise price of incentive stock options must be equal to at least the fair value of the Company’s common stock on the date of grant, and the exercise price of non-statutory stock options may be no less than 85% of the fair value of the Company’s common stock on the date of the grant. The exercise price of any option granted to a 10% stockholder may not be less than 110% of the fair value of the Company’s common stock on the date of grant. In December 2006, the Board of Directors approved
67
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
the 500,000 share automatic increase in the shares of the Company’s common stock authorized for issuance pursuant to the 2005 Plan, effective as of January 1, 2007.
2005 Non-Employee Directors’ Stock Option Plan
The Company adopted in August 2005, and the stockholders approved in October 2005, the 2005 non-employee directors’ stock option plan (the “Directors’ Plan”). The Directors’ Plan became effective upon the effectiveness of the Company’s initial public offering (see Note 1). The Directors’ Plan provides for the automatic grant of non-qualified options to purchase shares of our common stock to our non-employee directors. An aggregate of 75,000 shares of common stock are reserved for issuance under the Directors’ Plan. This amount will be increased annually on the first day of the Company’s fiscal year, from 2007 until 2015, by the aggregate number of shares of our common stock subject to options granted as initial grants and annual grants under the directors’ plan during the immediately preceding year. There were no options granted from the directors’ plan during 2006.
Stock Option Activity:
The following table summarizes activity related to stock options to purchase shares of the Company’s common stock:
| Weighted-Average | ||||||||||||||||
| Remaining | ||||||||||||||||
| Weighted-Average | Contractual Term | Aggregate | ||||||||||||||
| Shares | Exercise Price | in Years | Intrinsic Value | |||||||||||||
| Outstanding at December 31, 2003 | 164,101 | $ | 11.98 | |||||||||||||
| Granted | 39,905 | $ | 4.80 | |||||||||||||
| Exercised | (11,196 | ) | $ | 5.59 | ||||||||||||
| Cancelled | (28,335 | ) | $ | 12.04 | ||||||||||||
| Outstanding at December 31, 2004 | 164,475 | $ | 10.67 | |||||||||||||
| Granted | 1,396,627 | $ | 1.00 | |||||||||||||
| Exercised | (213,417 | ) | $ | 1.04 | ||||||||||||
| Cancelled | (200,999 | ) | $ | 2.41 | ||||||||||||
| Outstanding at December 31, 2005 | 1,146,686 | $ | 2.13 | |||||||||||||
| Granted | 677,750 | $ | 6.77 | |||||||||||||
| Exercised | (96,856 | ) | $ | 1.06 | ||||||||||||
| Cancelled | (143,480 | ) | $ | 4.55 | ||||||||||||
| Outstanding at December 31, 2006 | 1,584,100 | $ | 3.96 | 8.14 | $ | 2,130 | ||||||||||
| Exercisable at December 31, 2006 | 591,183 | $ | 2.75 | 7.17 | $ | 1,205 | ||||||||||
68
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Selected information regarding stock options as of December 31, 2006:
| Options Outstanding | ||||||||||||||||||||
| Weighted | Options Exercisable | |||||||||||||||||||
| Average | Weighted | Weighted | ||||||||||||||||||
| Remaining | Average | Average | ||||||||||||||||||
| Number of | Contractual | Exercise | Number of | Exercise | ||||||||||||||||
Range of Exercise Price | Shares | Term in Years | Price | Shares | Price | |||||||||||||||
| $1.00 | 836,612 | 7.91 | $ | 1.00 | 477,111 | $ | 1.00 | |||||||||||||
| $1.58 - $7.66 | 632,696 | 8.93 | $ | 6.53 | 43,221 | $ | 4.51 | |||||||||||||
| $7.69 - $8.18 | 41,250 | 9.23 | $ | 7.83 | — | $ | — | |||||||||||||
| $13.44 | 73,542 | 5.19 | $ | 13.44 | 70,851 | $ | 13.44 | |||||||||||||
| $1.00 - $13.44 | 1,584,100 | 8.14 | $ | 3.96 | 591,183 | $ | 2.75 | |||||||||||||
As of December 31, 2006, there was $3,230 of unrecognized compensation cost related to non-vested option arrangements. The cost is expected to be recognized over a weighted average period of 2.31 years. The per share weighted-average grant date fair value of options granted (as determined through the use of the Black-Scholes option pricing model) during 2006, 2005 and 2004 was $4.36, $10.15, and $1.90, respectively. For 2006, the Black-Scholes model with the following assumptions was used (annualized percentages):
| Twelve Months | ||||
| Ended | ||||
| December 31, | ||||
| 2006 | ||||
| Expected volatility | 66 | % | ||
| Risk-free interest rate | 4.7 | % | ||
| Dividend yield | 0 | % | ||
| Expected term | 6.25 years | |||
Restricted Stock and Restricted Stock Unit Grants
In May 2005, the Company granted a restricted stock award under the Company’s Equity Incentive Plan of 70,000 shares of the Company’s common stock. Twenty-five percent of the shares subject to the award were immediately vested as of the date of grant and the remaining shares subject to the award vest in equal monthly installments over a two year period.
In March 2006, the Company granted restricted stock unit awards in the amount of 75,000 each to two members of the Company’s executive management team under the Company’s 2005 Plan. Twenty-five percent of the shares subject to the restricted stock awards will vest on the one-year anniversary of their respective hire dates, with the remaining shares subject to such awards vesting in equal monthly installments over the following three years.
At December 31, 2006 and December 31, 2005, there were 142,188 shares and 37,188 shares, respectively, of unvested restricted common stock outstanding under these agreements.
69
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Changes in the Company’s restricted stock for the twelve months ended December 31, 2006 were as follows (in thousands except per share amounts):
| Weighted-Average | ||||||||
| Restricted | Grant Date | |||||||
| Shares | Fair Value | |||||||
| Non-vested restricted stock at January 1, 2006 | 37,188 | $ | 10.37 | |||||
| Granted | 150,000 | 7.66 | ||||||
| Vested | (45,000 | ) | 9.22 | |||||
| Non-vested restricted stock at December 31, 2006 | 142,188 | $ | 7.87 | |||||
For the year ended December 31, 2006, the Company recorded stock-based compensation expense of $0.5 million related to outstanding restricted stock grants.
As of December 31, 2006, there was $0.7 million of unrecognized compensation cost related to non-vested restricted stock arrangements. The cost is expected to be recognized over a weighted average period of 2.79 years. The total fair value of shares vested during the year ended December 31, 2006 was $415.
Common Stock Options to Consultants
As of December 31, 2006, the Company has outstanding options to purchase 119,854 shares of common stock that were granted to consultants. Of the total shares granted, 13,232 were exercised, and 17,723 were unvested. These options were granted in exchange for consulting services to be rendered and vest over periods of up to four years. The Company recorded charges to operations for stock options granted to consultants using the graded-vesting method of $145 during the year ended December 31, 2006. There was no charge to operations in 2005. The unvested shares held by consultants have been and will be revalued using the Company’s estimate of fair value at each balance sheet date pursuant to EITF IssueNo. 96-18,Accounting for Equity Instruments That Are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling, Goods or Services.
Total stock-based compensation expense recognized for the year ended December 31, 2006, related to all of the Company’s stock-based awards granted, was comprised as follows:
| Year Ended | ||||
| December 31, | ||||
| 2006 | ||||
| Research and development | $ | 2,124 | ||
| General and administrative | $ | 2,445 | ||
| Stock-based compensation expense | $ | 4,569 | ||
| Stock-based compensation expense per common share, basic and diluted: | $ | 0.33 | ||
2005 Employee Stock Purchase Plan
The Company adopted in August 2005, and the stockholders approved in October 2005, the 2005 employee stock purchase plan (the “Purchase Plan”). The Purchase Plan became effective upon the effectiveness of the Company’s initial public offering (see Note 1). The Purchase Plan will terminate at the time that all of the shares of our common stock then reserved for issuance under the Purchase Plan have been issued under the terms of the Purchase Plan, unless our board of directors terminates it earlier. An aggregate of 375,000 shares of our common stock are reserved for issuance under the Purchase Plan. This amount will be increased annually on the first day of our fiscal year, from 2007 until 2015, by the lesser of (i) 1% of the fully-diluted shares of our common stock outstanding on January 1 of the current fiscal year or (ii) 150,000 shares of our common stock. An additional 150,000 shares of common stock were automatically added to the Purchase Plan effective January 1, 2007.
70
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
Unless otherwise determined by our board of directors or its authorized committee, common stock is purchased for accounts of employees participating in the Purchase Plan at a price per share equal to the lower of (1) 85% of the fair market value of a share of the Company’s common stock on the date of commencement of participation in the offering or (2) 85% of the fair market value of a share of the Company’s common stock on the date of purchase. Given this look-back feature of up to 24 months, if the Company’s stock price continues to increase from the initial offering price, or automatic rollover if the stock price declines from the initial offering price at the end of the purchase period, the Company is required to account for the plan as a variable plan. At the end of each six month purchase period, the Company must 1) recalculate a new expense amount based upon the intrinsic value between the fair market value on purchase date and the purchase price and 2) add to this amount 85% of the Black Scholes value of the call option attributable to the look-back feature. The Company also has, for any one purchase period, a 7,500 share purchase maximum.
The Company’s initial offering period for the Purchase Plan began in February 2006 and the first purchase period was on September 15, 2006. At September 15, 2006, the purchase price was lower than at the beginning of the initial offering period, therefore the Company recorded only the intrinsic value difference between the fair market value on purchase date and the purchase price, in the amount of $20. Since the purchase price was lower at the end of the first purchase period, this offering period ended and a new offering period commenced with all participants automatically rolling over to the new offering period with the new lower initial offering price. Because of this feature, the Company is required to calculate the value of the new offering at the beginning of the offering period and will record the expense in accordance with the accelerated method. The valuation for the offering period beginning September 16, 2006 was $355 based on the following assumptions:
| Twelve Months | ||
| Ended | ||
| December 31, | ||
| 2006 | ||
| Expected volatility | 67% | |
| Risk-free interest rate | 5.0% | |
| Dividend yield | 0% | |
| Expected term | 6 months to 2 years |
During the quarter ended December 31, 2006, the Company recorded $86 of this amount as stock-based compensation expense.
Warrants
Since 2000, in connection with certain debt arrangements and consulting service agreements, the Company has issued warrants to purchase shares of common stock. As of December 31, 2006, the Company had outstanding warrants to purchase 201,400 shares of the Company’s common stock with exercise prices ranging from $1.00 to $28.46. These warrants expire at various times between March 2007 and December 2015.
| 7. | Stockholders equity (deficit) |
Common Stock
Many of the outstanding shares of common stock have been issued to the founders, directors, employees and consultants of the Company. In connection with certain stock purchase agreements, the Company has the option to repurchase, at the original issuance price, the unvested shares in the event of termination of employment or engagement. Shares under these agreements vest over periods of up to four years. At December 31, 2006, 6,945 shares were subject to repurchase by the Company.
71
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
In April 2005, the Company’s board of directors authorized a0.126453-for-1 reverse stock split for all outstanding preferred and common shares. In January 2006, the Company’s board of directors authorized a1-for-2 reverse stock split for all common shares. As a result of the1-for-2 reverse stock split, each share of the Company’s preferred stock was convertible into one-half of a share of common stock. The1-for-2 reverse stock split of the Company’s common stock adjusted the conversion ratio of the preferred stock but did not adjust the number of outstanding shares of preferred stock. All share and per share amounts have been retroactively adjusted to give effect to these two reverse stock splits.
Shares Reserved for Future Issuance
The Company has reserved shares of common stock for future issuance as follows:
| December 31, | ||||
| 2006 | ||||
| Equity Incentive Plans | 1,715,350 | |||
| Warrants | 201,400 | |||
| Total Shares Reserved for Issuance | 1,916,750 | |||
In December 2004, the Company issued an amended and restated convertible promissory note in a private placement to Millennium Pharmaceuticals, Inc. (as successor in interest to mHOLDINGS Trust) in the aggregate principal amount of $6,000. No interest accrues on the principal unless a payment towards the principal becomes overdue. The full principal amount of the note converted into an aggregate of 1,000,000 shares of common stock upon the closing of the initial public offering at $6.00 per share, the price per share to the public in the offering (see Note 1).
Notes Receivable
From 1999 to 2002, the board of directors authorized the issuance of an aggregate of approximately $2,000 in loans to employees and consultants, related to the exercise of their stock options and purchase of their restricted stock. The notes are full recourse and are also secured by the underlying stock. The notes bear interest at 7%. The principal amount of the notes and the related interest are required to be repaid on the earlier of five years from the origination date of the loans, upon termination of employment by or association with the Company or upon the sale of the underlying stock securing the note.
During 2004, the compensation committee of the board of directors authorized the Company to repurchase vested and unvested shares of common stock in settlement of the principal and accrued interest on the outstanding notes (the “Note Settlement”). The Company repurchased 131,224 shares of common stock in settlement of approximately $1,113 in aggregate principal and accrued interest on the notes. The Company also forgave approximately $782 of principal and accrued interest related to those notes whose principal balance had been partially repaid in the Note Settlement. As of December 31, 2006 and 2005, approximately $21 and $59, respectively, of aggregate principal and accrued interest remained outstanding on the notes and will continue to remain outstanding until such notes are extinguished.
| 8. | Income Taxes |
Significant components of the Company’s deferred tax assets as of December 31, 2006 and 2005 are shown below. A valuation allowance of $61,183, of which $11,751 relates to 2006, has been recognized to offset the deferred tax assets, as realization of such assets is uncertain.
72
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
| December 31, | ||||||||
| 2006 | 2005 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | $ | 42,030 | $ | 38,538 | ||||
| Research and development credits | 7,284 | 5,990 | ||||||
| Capitalized research and development | 1,335 | 1,555 | ||||||
| Accrued vacation | 274 | 224 | ||||||
| Deferred revenue | 8,157 | 1,954 | ||||||
| Other | 1,107 | 141 | ||||||
| Depreciation and amortization | 996 | 1,030 | ||||||
| Total deferred tax assets | 61,183 | 49,432 | ||||||
| Valuation allowance for deferred tax assets | (61,183 | ) | (49,432 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
At December 31, 2006, the Company had federal and California tax net operating loss carryforwards of approximately $107,564 and $76,269, respectively. The federal and California tax loss carryforwards will begin to expire in 2019 and 2009, respectively, unless previously utilized. The Company also has federal and California research and development tax credit carryforwards totaling approximately $5,042 and $3,449, respectively. The federal research and development tax credit carry forward will begin to expire in 2019, unless previously utilized.
Pursuant to Internal Revenue Code Sections 382 and 383, use of the Company’s net operating loss and tax credit carryforwards may be limited as a result of certain cumulative changes in the Company’s stock ownership which occurred during 1999, 2001 and 2005.
| 9. | Employee Benefit Plan |
Effective October 1, 1999, the Company adopted a defined contribution 401(k) profit sharing plan (the “Plan”) covering substantially all employees that meet certain age requirements. Employees may contribute up to 100% of their compensation per year (subject to a maximum limit set by federal law). The Plan does allow for employer matching; however, to date, the Company has not contributed to the Plan.
| 10. | Reductions In Force |
During August 2004, the Company eliminated approximately 11% of its workforce (17 employees). The Company recorded a charge related to this reduction in force of approximately $278, all of which was paid as of December 31, 2004.
In April 2005, the Company terminated 14 of its employees. The Company provided severance benefits to all such terminated employees who executed a severance agreement and release. The total costs associated with the severance benefits were approximately $230, all of which was paid as of December 31, 2005.
73
Table of Contents
SGX Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements — (Continued)
| 11. | Quarterly Financial Data (unaudited) |
The following tables summarize certain of the Company’s operating results by quarter for 2006 and 2005:
| 2006 | ||||||||||||||||||||
| First | Second | Third | Fourth | Total | ||||||||||||||||
| Revenues | $ | 4,729 | $ | 7,848 | $ | 6,767 | $ | 8,436 | $ | 27,780 | ||||||||||
| Net loss | $ | (10,415 | ) | $ | (9,354 | ) | $ | (3,398 | ) | $ | (4,885 | ) | $ | (28,052 | ) | |||||
| Accretion of redeemable convertible preferred stock issuance costs | (49 | ) | — | — | — | (49 | ) | |||||||||||||
| Net loss applicable to common stockholders | $ | (10,464 | ) | $ | (9,354 | ) | $ | (3,398 | ) | $ | (4,885 | ) | $ | (28,101 | ) | |||||
| Net loss per share | $ | (1.06 | ) | $ | (0.62 | ) | $ | (0.23 | ) | $ | (0.32 | ) | $ | (2.03 | ) | |||||
| Net loss per share applicable to common stockholders | $ | (1.06 | ) | $ | (0.62 | ) | $ | (0.23 | ) | $ | (0.32 | ) | $ | (2.03 | ) | |||||
| 2005 | ||||||||||||||||||||
| First | Second | Third | Fourth | Total | ||||||||||||||||
| Revenues | $ | 4,010 | $ | 5,847 | $ | 4,893 | $ | 6,886 | $ | 21,636 | ||||||||||
| Net loss | $ | (5,806 | ) | $ | (7,166 | ) | $ | (10,552 | ) | $ | (5,867 | ) | $ | (29,391 | ) | |||||
| Accretion of redeemable convertible preferred stock issuance costs | (82 | ) | (82 | ) | (55 | ) | (253 | ) | (472 | ) | ||||||||||
| Net loss applicable to common stockholders | $ | (5,888 | ) | $ | (7,248 | ) | $ | (10,607 | ) | $ | (6,120 | ) | $ | (29,863 | ) | |||||
| Net loss per share | $ | (11.61 | ) | $ | (12.96 | ) | $ | (17.38 | ) | $ | (7.30 | ) | $ | (47.56 | ) | |||||
| Accretion of redeemable convertible preferred stock issuance costs | (0.16 | ) | (0.15 | ) | (0.09 | ) | (0.31 | ) | (0.76 | ) | ||||||||||
| Net loss per share applicable to common stockholders | $ | (11.77 | ) | $ | (13.11 | ) | $ | (17.47 | ) | $ | (7.61 | ) | $ | (48.32 | ) | |||||
Per share amounts for the quarters and full years have been calculated separately. Accordingly, quarterly amounts may not add to the annual amount because of differences in the weighted average common shares outstanding during each period principally due to the effect of the Company issuing shares of its common stock during the year. Diluted and basic net loss per share are identical since common equivalent shares are excluded from the calculation, as their effect is anti-dilutive.
74
Table of Contents
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
Not applicable.
| Item 9A. | Controls and Procedures |
As of the end of the period covered by this report, an evaluation was performed under the supervision and with the participation of our management, including our chief executive officer and chief financial officer (collectively, our “certifying officers”), of the effectiveness of the design and operation of our disclosure controls and procedures. Based on their evaluation, our certifying officers concluded that these disclosure controls and procedures are effective in providing reasonable assurance that the information required to be disclosed by us in our periodic reports filed with the Securities and Exchange Commission (“SEC”) is recorded, processed, summarized and reported within the time periods specified by the SEC’s rules and SEC reports.
We believe that a controls system, no matter how well designed and operated, is based in part upon certain assumptions about the likelihood of future events, and therefore can only provide reasonable, not absolute, assurance that the objectives of the controls system are met, and no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within a company have been detected.
In addition, we have reviewed our internal controls over financial reporting and have not made any change during the quarter ended December 31, 2006, that our certifying officers concluded materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
| Item 9B. | Other Information |
Not applicable.
PART III
| Item 10 | Directors, Executive Officers and Corporate Governance |
The information required by this item with respect to directors and executive officers is incorporated by reference from the information under the captions “Election of Directors”, “Executive Officers”, “Section 16(a) Beneficial Ownership Reporting Compliance”, and “Information Regarding the Board of Directors and Corporate Governance” contained in the proxy statement to be filed with the SEC pursuant to Regulation 14A within 120 days following the fiscal year ended December 31, 2006 (“Proxy Statement”) in connection with our 2007 annual meeting of stockholders. We have adopted a Code of Business Conduct and Ethics that applies to all officers, directors and employees. The text of the Company’s Code of Business Conduct and Ethics is posted on the Company’s internet website and may be accessed atwww.sgxpharma.com. If we make any substantive amendments to the Code of Business Conduct and Ethics or grant any waiver from a provision of the code to any executive officer or director, we will promptly disclose the nature of the amendment or waiver on our website.
There have been no material changes to the procedures under which security holders may recommend nominees to the Company’s Board of Directors.
| Item 11. | Executive Compensation |
The information required by this item will be set forth in the sections entitled “Executive Compensation”, “Compensation Committee Processes and Procedures:, “Compensation Committee Report” and “Compensation Committee Interlocks and Insider Participation” in the Proxy Statement and is incorporated in this report by reference.
75
Table of Contents
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
The information required by this item will be set forth in the sections entitled “Security Ownership of Certain Beneficial Owners and Management” and “Securities Authorized for Issuance under Equity Compensation Plans” in the Proxy Statement and is incorporated in this report by reference.
| Item 13. | Certain Relationships and Related Transactions, and Director Independence |
The information required by this item will be set forth in the sections entitled “Transactions with Related Persons” and “information Regarding the Board of Directors and Corporate Governance” in the Proxy Statement and is incorporated in this report by reference.
| Item 14. | Principal Accounting Fees and Services |
The information required by this item will be set forth in the section entitled “Ratification of Selection of Independent Registered Public Accounting Firm” in the Proxy Statement and is incorporated in this report by reference.
PART IV
| Item 15. | Exhibits and Financial Statement Schedules |
(a) 1.Financial Statements
See Index to Consolidated Financial Statements in Item 8 of this Annual Report onForm 10-K, which is incorporated herein by reference.
2. Financial Statement Schedules
None
3. Exhibits
| Exhibit | ||||
Number | Description of Document | |||
| 3 | .1(1) | Form of Registrant’s Amended and Restated Certificate of Incorporation. | ||
| 3 | .2 | Form of Registrant’s Amended and Restated Bylaws. | ||
| 4 | .1(4) | Form of Common Stock Certificate of Registrant. | ||
| 4 | .2(1) | Form of Warrant to Purchase Common Stock issued by Registrant in July 2005 to Timothy Harris and Linda Grais. | ||
| 4 | .3(4) | Form of Warrants issued by Registrant in July 2002 to GATX Ventures, Inc. | ||
| 4 | .4(3) | Amended and Restated Warrant issued by Registrant in January 2005 to Oxford Finance Corporation. | ||
| 4 | .5(4) | Warrant issued by Registrant in July 2002 to Silicon Valley Bank. | ||
| 4 | .6(1) | Amended and Restated Investor Rights Agreement dated April 21, 2005 between Registrant and certain of its stockholders. | ||
| 4 | .7(2) | Form of Warrant issued by Registrant in September and December 2005 to Oxford Finance Corporation and Silicon Valley Bank. Reference is made to Exhibit 10.34. | ||
| 4 | .8 | First and Second Amendments to Amended and Restated Investor Rights Agreement, dated October 31, 2005 and March 27, 2006, respectively, each between Registrant and certain of its stockholders. | ||
| 10 | .1+(1) | Form of Indemnity Agreement by and between Registrant and its directors and executive officers. | ||
| 10 | .2+(4) | 2000 Equity Incentive Plan and Form of Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
| 10 | .3+(4) | 2005 Equity Incentive Plan and Form of Stock Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
76
Table of Contents
| Exhibit | ||||
Number | Description of Document | |||
| 10 | .4+(4) | 2005 Employee Stock Purchase Plan and Form of Offering Document thereunder. | ||
| 10 | .5+(4) | 2005 Non-Employee Directors’ Stock Option Plan and Form of Stock Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
| 10 | .6+(1) | Amended and Restated Executive Employment Agreement dated January 1, 2005 between Registrant and Michael Grey. | ||
| 10 | .7+(1) | Executive Employment Agreement dated January 1, 2002 between Registrant and Stephen Burley, M.D., D.Phil. and related relocation loan agreement dated July 29, 2002. | ||
| 10 | .8+(1) | Chairmanship Letter Agreement dated January 16, 2004 between Registrant and Christopher Henney, Ph.D., DSc. | ||
| 10 | .9+(1) | Non-Employee Director Compensation Letter Agreement dated April 13, 2001 between Registrant and Stelios Papadopoulos, Ph.D., as amended. | ||
| 10 | .10(1) | Lease Agreement dated September 20, 1999 between Registrant and ARE-10505 Roselle Street, LLC, as amended. | ||
| 10 | .11(1) | Lease Agreement dated May 18, 2000 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 10 | .12(1) | Lease Agreement dated June 1, 2001 between Registrant and BRS Torrey I, LLC. | ||
| 10 | .13*(4) | Patent and Know How License dated July 23, 2004 between Registrant, Shire Biochem Inc., Tanaud Ireland Inc. and Tanaud International B.V., as amended, and related novation agreements. | ||
| 10 | .14*(3) | Collaboration and License Agreement dated April 14, 2003 between Registrant and Eli Lilly and Company. | ||
| 10 | .15*(3) | Amendment to Agreement dated July 1, 2003 between Registrant and Eli Lilly and Company. | ||
| 10 | .16*(1) | Amendment to Agreement dated January 30, 2004 between Registrant and Eli Lilly and Company. | ||
| 10 | .17*(3) | Amendment to Agreement dated November 11, 2004 between Registrant and Eli Lilly and Company. | ||
| 10 | .18*(1) | Amendment to Agreement dated March 31, 2005 between Registrant and Eli Lilly and Company. | ||
| 10 | .19*(3) | Collaboration Agreement dated August 20, 2004 between Registrant, F. Hoffmann-La Roche Ltd. and Hoffmann-La Roche Inc. | ||
| 10 | .20*(3) | Collaboration Agreement dated March 18, 2004 between Registrant and Serono International SA. | ||
| 10 | .21*(3) | Collaboration Agreement dated December 1, 2003 between Registrant and UroGene, S.A. (which was acquired by Pierre Fabre Medicament in July 2005). | ||
| 10 | .22*(1) | Amendment to Agreement dated December 16, 2004 between Registrant and UroGene, S.A.(predecessor-in-interest to Pierre Fabre Medicament) and related assignment agreements. | ||
| 10 | .23*(4) | Drug Discovery Agreement dated July 1, 2005 between Registrant and Cystic Fibrosis Foundation Therapeutics, Inc. | ||
| 10 | .24(1) | Memorandum of Understanding dated July 26, 2000 between the Advanced Photon Source and the Structural GenomiX Collaborative Access Team and related Collaborative Access Team User Agreement dated May 15, 2001 between Registrant, The University of Chicago and United States Department of Energy. | ||
| 10 | .25(1) | Master Loan and Security Agreement No. 2081008 dated August 28, 2002 between Registrant and Oxford Finance Corporation, as amended. | ||
| 10 | .26(2) | First Amendment to Lease Agreement dated August 30, 2005 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 10 | .27(2) | Third Amendment to Lease Agreement dated August 30, 2005 between Registrant and ARE-10505 Roselle Street, LLC. | ||
| 10 | .28(2) | Loan and Security Agreement dated September 16, 2005 among Registrant, Oxford Finance Corporation and Silicon Valley Bank. | ||
| 10 | .29*(3) | Amendment to Agreement effective as of October 1, 2005 between Registrant, F. Hoffmann-La Roche Ltd. and Hoffmann-La Roche Inc. | ||
| 10 | .30*(3) | Amendment to Agreement dated October 28, 2005 between Registrant and Serono International SA. | ||
| 10 | .31+(4) | Offer Letter Agreement dated November 16, 2005 between Registrant and William Todd Myers. | ||
77
Table of Contents
| Exhibit | ||||
Number | Description of Document | |||
| 10 | .32+(4) | Offer Letter Agreement dated December 13, 2005 between Registrant and Siegfried Reich, Ph.D. | ||
| 10 | .33**(5) | Termination Agreement effective February 15, 2006 between Registrant and Pierre Fabre Medicament S.A. | ||
| 10 | .34+(8) | Form of Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement for Executive Officers under the Registrant’s 2005 Equity Incentive Plan. | ||
| 10 | .35(8) | Stock Purchase Agreement dated March 27, 2006, between Registrant and Novartis Pharma AG. | ||
| 10 | .36(6)* | License and Collaboration Agreement between SGX and Novartis, dated March 27, 2006. | ||
| 10 | .37(7) | Form of Change in Control Severance Agreement, which was filed as exhibit 10.1 to SGX’s10-Q for the period ending September 30, 2006. | ||
| 10 | .38+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Michael Grey | ||
| 10 | .39+ | Change of Control Severance Agreement dated February 9, 2007 between Registrant and Stephen K. Burley | ||
| 10 | .40+ | Change of Control Severance Agreement dated December 15, 2006 between Registrant and W. Todd Myers | ||
| 10 | .41+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Annette North | ||
| 10 | .42+ | Change of Control Severance Agreement dated January 2, 2007 between Registrant and Siegfried Reich | ||
| 10 | .43+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Terence Rugg | ||
| 10 | .44+ | Summary of 2007 Bonus Award Program | ||
| 10 | .45+ | Letter Agreement dated February 9, 2007 between Registrant and Stephen K. Burley | ||
| 10 | .46 | Fifth Amendment to Collaboration and License Agreement dated March 1, 2007 between Registrant and Eli Lilly and Company | ||
| 10 | .47 | First Amendment to the Drug Discovery Agreement dated March 1, 2007 between Registrant and Cystic Fibrosis Foundation Therapeutics, Inc. | ||
| 10 | .48 | Fourth Amendment to Lease Agreement dated March 28, 2007 between Registrant and ARE-10505 Roselle Street, LLC. | ||
| 10 | .49 | Second Amendment to Lease Agreement dated March 28, 2007 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 23 | .1 | Consent of Independent Registered Public Accounting Firm. | ||
| 24 | .1 | Power of Attorney. Reference is made to the signature page of this report. | ||
| 31 | .1 | Certification of principal executive officer required byRule 13a-14(a) orRule 15d-14(a). | ||
| 31 | .2 | Certification of principal financial officer required byRule 13a-14(a) orRule 15d-14(a). | ||
| 32 | Certification by the Chief Executive Officer and the Chief Financial Officer of the Registrant, as required byRule 13a-14(b) or15d-14(b) and Section 1350 of Chapter 63 of Title 18 of the United States Code (18 U.S.C. 1350). | |||
| + | Indicates management contract or compensatory plan. | |
| * | Confidential treatment has been granted with respect to certain portions of this exhibit. Omitted portions have been filed separately with the Securities and Exchange Commission. | |
| ** | Confidential treatment has been requested with respect to certain portions of this exhibit. Omitted portions have been filed separately with the Securities and Exchange Commission. | |
| (1) | Filed with the Registrant’s Registration Statement onForm S-1 on September 2, 2005 and incorporated herein by reference. |
78
Table of Contents
| (2) | Filed with Amendment No. 1 to the Registrant’s Registration Statement onForm S-1 on October 14, 2005 and incorporated herein by reference. | |
| (3) | Filed with Amendment No. 3 to the Registrant’s Registration Statement onForm S-1 on November 14, 2005 and incorporated herein by reference. | |
| (4) | Filed with Amendment No. 4 to the Registrant’s Registration Statement onForm S-1 on January 4, 2006 and incorporated herein by reference. | |
| (5) | Filed with the Registrant’s Current Report onForm 8-K on March 13, 2006 and incorporated herein by reference. | |
| (6) | Filed with the Registrant’s Current Report onForm 8-K on April 5, 2006 and incorporated herein by reference. | |
| (7) | Filed with the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 2006 filed on November 13, 2006 and incorporated herein by reference. | |
| (8) | Filed as an Exhibit to the Registrants Annual Report on Form 10-K for the year ended December 31, 2005 filed with the Commission on March 31, 2006, and incorporated herein by reference. |
(b) Exhibits
See Item 15(a) above.
(c) Financial Statement Schedules
See Item 15(a) above.
79
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
SGX PHARMACEUTICALS, INC.
| By: | /s/ Michael Grey |
Michael Grey
President and Chief Executive Officer
Dated: March 30, 2007
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Todd Myers. and Michael Grey, and each of them, acting individually, as his or herattorney-in-fact, each with full power of substitution and resubstitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments to this annual report onForm 10-K, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto saidattorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that saidattorneys-in-fact and agents, or any of them, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Signature | Title | Date | ||||
/s/ Michael Grey Michael Grey | President, Chief Executive Officer and Member of the Board of Directors(Principal Executive Officer) | March 30, 2007 | ||||
/s/ Todd Myers Todd Myers, C.P.A. | Chief Financial Officer(Principal Financial and Accounting Officer) | March 30, 2007 | ||||
/s/ Christopher S. Henney Christopher S. Henney, Ph.D., D.Sc. | Chairman of the Board of Directors | March 30, 2007 | ||||
/s/ Louis C. Bock Louis C. Bock | Member of the Board of Directors | March 30, 2007 | ||||
/s/ Karin Eastham Karin Eastham, C.P.A. | Member of the Board of Directors | March 30, 2007 | ||||
/s/ Jean-Francois Formela Jean-Francois Formela, M.D. | Member of the Board of Directors | March 30, 2007 | ||||
/s/ Vijay K. Lathi Vijay K. Lathi | Member of the Board of Directors | March 30, 2007 | ||||
80
Table of Contents
EXHIBIT INDEX
| Exhibit | ||||
Number | Description of Document | |||
| 3 | .1(1) | Form of Registrant’s Amended and Restated Certificate of Incorporation. | ||
| 3 | .2 | Form of Registrant’s Amended and Restated Bylaws. | ||
| 4 | .1(4) | Form of Common Stock Certificate of Registrant. | ||
| 4 | .2(1) | Form of Warrant to Purchase Common Stock issued by Registrant in July 2005 to Timothy Harris and Linda Grais. | ||
| 4 | .3(4) | Form of Warrants issued by Registrant in July 2002 to GATX Ventures, Inc. | ||
| 4 | .4(3) | Amended and Restated Warrant issued by Registrant in January 2005 to Oxford Finance Corporation. | ||
| 4 | .5(4) | Warrant issued by Registrant in July 2002 to Silicon Valley Bank. | ||
| 4 | .6(1) | Amended and Restated Investor Rights Agreement dated April 21, 2005 between Registrant and certain of its stockholders. | ||
| 4 | .7(2) | Form of Warrant issued by Registrant in September and December 2005 to Oxford Finance Corporation and Silicon Valley Bank. Reference is made to Exhibit 10.34. | ||
| 4 | .8 | First and Second Amendments to Amended and Restated Investor Rights Agreement, dated October 31, 2005 and March 27, 2006, respectively, each between Registrant and certain of its stockholders. | ||
| 10 | .1+(1) | Form of Indemnity Agreement by and between Registrant and its directors and executive officers. | ||
| 10 | .2+(4) | 2000 Equity Incentive Plan and Form of Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
| 10 | .3+(4) | 2005 Equity Incentive Plan and Form of Stock Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
| 10 | .4+(4) | 2005 Employee Stock Purchase Plan and Form of Offering Document thereunder. | ||
| 10 | .5+(4) | 2005 Non-Employee Directors’ Stock Option Plan and Form of Stock Option Agreement and Form of Stock Option Grant Notice thereunder. | ||
| 10 | .6+(1) | Amended and Restated Executive Employment Agreement dated January 1, 2005 between Registrant and Michael Grey. | ||
| 10 | .7+(1) | Executive Employment Agreement dated January 1, 2002 between Registrant and Stephen Burley, M.D., D.Phil. and related relocation loan agreement dated July 29, 2002. | ||
| 10 | .8+(1) | Chairmanship Letter Agreement dated January 16, 2004 between Registrant and Christopher Henney, Ph.D., DSc. | ||
| 10 | .9+(1) | Non-Employee Director Compensation Letter Agreement dated April 13, 2001 between Registrant and Stelios Papadopoulos, Ph.D., as amended. | ||
| 10 | .10(1) | Lease Agreement dated September 20, 1999 between Registrant and ARE-10505 Roselle Street, LLC, as amended. | ||
| 10 | .11(1) | Lease Agreement dated May 18, 2000 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 10 | .12(1) | Lease Agreement dated June 1, 2001 between Registrant and BRS Torrey I, LLC. | ||
| 10 | .13*(4) | Patent and Know How License dated July 23, 2004 between Registrant, Shire Biochem Inc., Tanaud Ireland Inc. and Tanaud International B.V., as amended, and related novation agreements. | ||
| 10 | .14*(3) | Collaboration and License Agreement dated April 14, 2003 between Registrant and Eli Lilly and Company. | ||
| 10 | .15*(3) | Amendment to Agreement dated July 1, 2003 between Registrant and Eli Lilly and Company. | ||
| 10 | .16*(1) | Amendment to Agreement dated January 30, 2004 between Registrant and Eli Lilly and Company. | ||
| 10 | .17*(3) | Amendment to Agreement dated November 11, 2004 between Registrant and Eli Lilly and Company. | ||
| 10 | .18*(1) | Amendment to Agreement dated March 31, 2005 between Registrant and Eli Lilly and Company. | ||
| 10 | .19*(3) | Collaboration Agreement dated August 20, 2004 between Registrant, F. Hoffmann-La Roche Ltd. and Hoffmann-La Roche Inc. | ||
| 10 | .20*(3) | Collaboration Agreement dated March 18, 2004 between Registrant and Serono International SA. | ||
81
Table of Contents
| Exhibit | ||||
Number | Description of Document | |||
| 10 | .21*(3) | Collaboration Agreement dated December 1, 2003 between Registrant and UroGene, S.A. (which was acquired by Pierre Fabre Medicament in July 2005). | ||
| 10 | .22*(1) | Amendment to Agreement dated December 16, 2004 between Registrant and UroGene, S.A.(predecessor-in-interest to Pierre Fabre Medicament) and related assignment agreements. | ||
| 10 | .23*(4) | Drug Discovery Agreement dated July 1, 2005 between Registrant and Cystic Fibrosis Foundation Therapeutics, Inc. | ||
| 10 | .24(1) | Memorandum of Understanding dated July 26, 2000 between the Advanced Photon Source and the Structural GenomiX Collaborative Access Team and related Collaborative Access Team User Agreement dated May 15, 2001 between Registrant, The University of Chicago and United States Department of Energy. | ||
| 10 | .25(1) | Master Loan and Security Agreement No. 2081008 dated August 28, 2002 between Registrant and Oxford Finance Corporation, as amended. | ||
| 10 | .26(2) | First Amendment to Lease Agreement dated August 30, 2005 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 10 | .27(2) | Third Amendment to Lease Agreement dated August 30, 2005 between Registrant and ARE-10505 Roselle Street, LLC. | ||
| 10 | .28(2) | Loan and Security Agreement dated September 16, 2005 among Registrant, Oxford Finance Corporation and Silicon Valley Bank. | ||
| 10 | .29*(3) | Amendment to Agreement effective as of October 1, 2005 between Registrant, F. Hoffmann-La Roche Ltd. and Hoffmann-La Roche Inc. | ||
| 10 | .30*(3) | Amendment to Agreement dated October 28, 2005 between Registrant and Serono International SA. | ||
| 10 | .31+(4) | Offer Letter Agreement dated November 16, 2005 between Registrant and William Todd Myers. | ||
| 10 | .32+(4) | Offer Letter Agreement dated December 13, 2005 between Registrant and Siegfried Reich, Ph.D. | ||
| 10 | .33**(5) | Termination Agreement effective February 15, 2006 between Registrant and Pierre Fabre Medicament S.A. | ||
| 10 | .34+(8) | Form of Restricted Stock Unit Grant Notice and Restricted Stock Unit Agreement for Executive Officers under the Registrant’s 2005 Equity Incentive Plan. | ||
| 10 | .35(8) | Stock Purchase Agreement dated March 27, 2006, between Registrant and Novartis Pharma AG. | ||
| 10 | .36(6)* | License and Collaboration Agreement between SGX and Novartis, dated March 27, 2006. | ||
| 10 | .37(7) | Form of Change in Control Severance Agreement, which was filed as exhibit 10.1 to SGX’s10-Q for the period ending September 30, 2006. | ||
| 10 | .38+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Michael Grey | ||
| 10 | .39+ | Change of Control Severance Agreement dated February 9, 2007 between Registrant and Stephen K. Burley | ||
| 10 | .40+ | Change of Control Severance Agreement dated December 15, 2006 between Registrant and W. Todd Myers | ||
| 10 | .41+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Annette North | ||
| 10 | .42+ | Change of Control Severance Agreement dated January 2, 2007 between Registrant and Siegfried Reich | ||
| 10 | .43+ | Change of Control Severance Agreement dated December 18, 2006 between Registrant and Terence Rugg | ||
| 10 | .44+ | Summary of 2007 Bonus Award Program | ||
| 10 | .45+ | Letter Agreement dated February 9, 2007 between Registrant and Stephen K. Burley | ||
| 10 | .46 | Fifth Amendment to Collaboration and License Agreement dated March 1, 2007 between Registrant and Eli Lilly and Company | ||
82
Table of Contents
| Exhibit | ||||
Number | Description of Document | |||
| 10 | .47 | First Amendment to the Drug Discovery Agreement dated March 1, 2007 between Registrant and Cystic Fibrosis Foundation Therapeutics, Inc. | ||
| 10 | .48 | Fourth Amendment to Lease Agreement dated March 28, 2007 between Registrant and ARE-10505 Roselle Street, LLC. | ||
| 10 | .49 | Second Amendment to Lease Agreement dated March 28, 2007 between Registrant and ARE-3770 Tansy Street, LLC. | ||
| 23 | .1 | Consent of Independent Registered Public Accounting Firm. | ||
| 24 | .1 | Power of Attorney. Reference is made to the signature page of this report. | ||
| 31 | .1 | Certification of principal executive officer required byRule 13a-14(a) orRule 15d-14(a). | ||
| 31 | .2 | Certification of principal financial officer required byRule 13a-14(a) orRule 15d-14(a). | ||
| 32 | Certification by the Chief Executive Officer and the Chief Financial Officer of the Registrant, as required byRule 13a-14(b) or15d-14(b) and Section 1350 of Chapter 63 of Title 18 of the United States Code (18 U.S.C. 1350). | |||
| + | Indicates management contract or compensatory plan. | |
| * | Confidential treatment has been granted with respect to certain portions of this exhibit. Omitted portions have been filed separately with the Securities and Exchange Commission. | |
| ** | Confidential treatment has been requested with respect to certain portions of this exhibit. Omitted portions have been filed separately with the Securities and Exchange Commission. | |
| (1) | Filed with the Registrant’s Registration Statement onForm S-1 on September 2, 2005 and incorporated herein by reference. | |
| (2) | Filed with Amendment No. 1 to the Registrant’s Registration Statement onForm S-1 on October 14, 2005 and incorporated herein by reference. | |
| (3) | Filed with Amendment No. 3 to the Registrant’s Registration Statement onForm S-1 on November 14, 2005 and incorporated herein by reference. | |
| (4) | Filed with Amendment No. 4 to the Registrant’s Registration Statement onForm S-1 on January 4, 2006 and incorporated herein by reference. | |
| (5) | Filed with the Registrant’s Current Report onForm 8-K on March 13, 2006 and incorporated herein by reference. | |
| (6) | Filed with the Registrant’s Current Report onForm 8-K on April 5, 2006 and incorporated herein by reference. | |
| (7) | Filed with the Registrant’s Quarterly Report onForm 10-Q for the quarter ended September 30, 2006 filed on November 13, 2006 and incorporated herein by reference. | |
| (8) | Filed as an Exhibit to the Registrants Annual Report on Form 10-K for the year ended December 31, 2005 filed with the Commission on March 31, 2006, and incorporated herein by reference. |
83