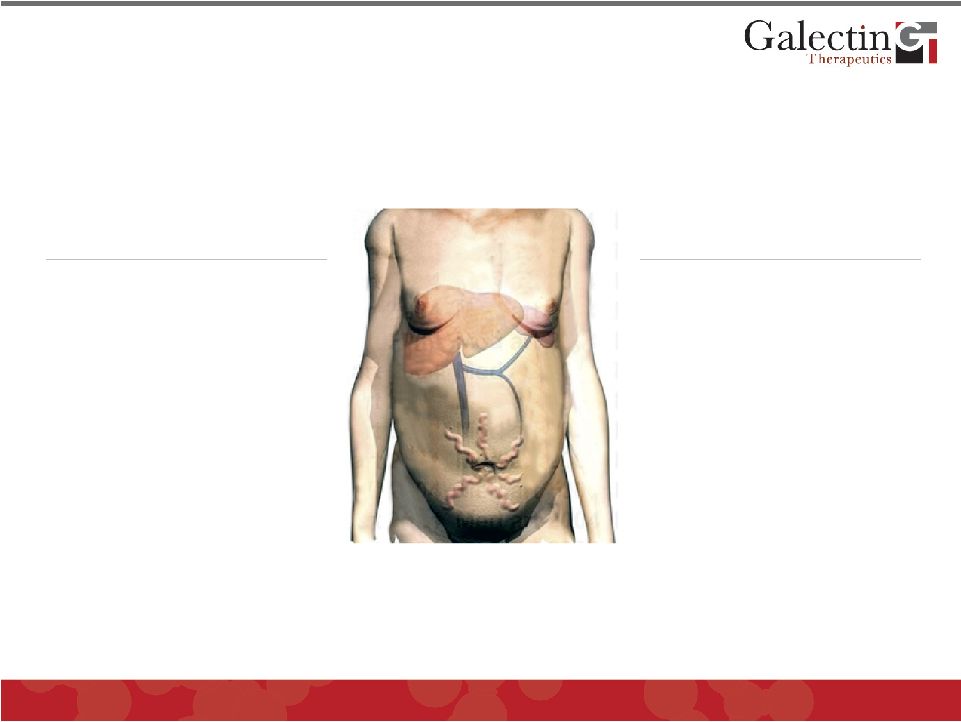
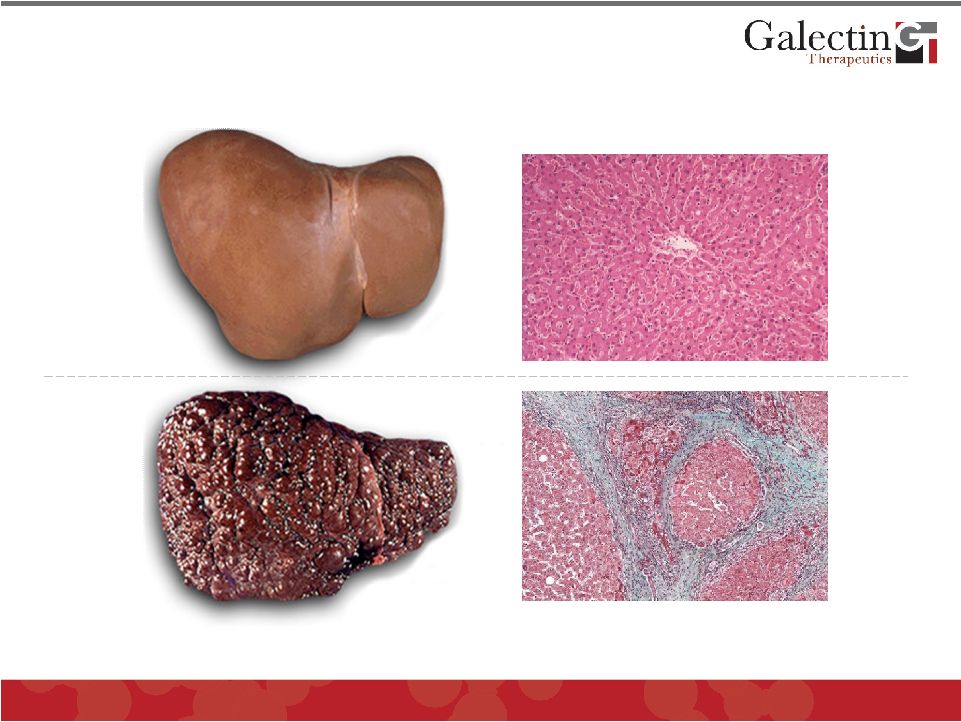
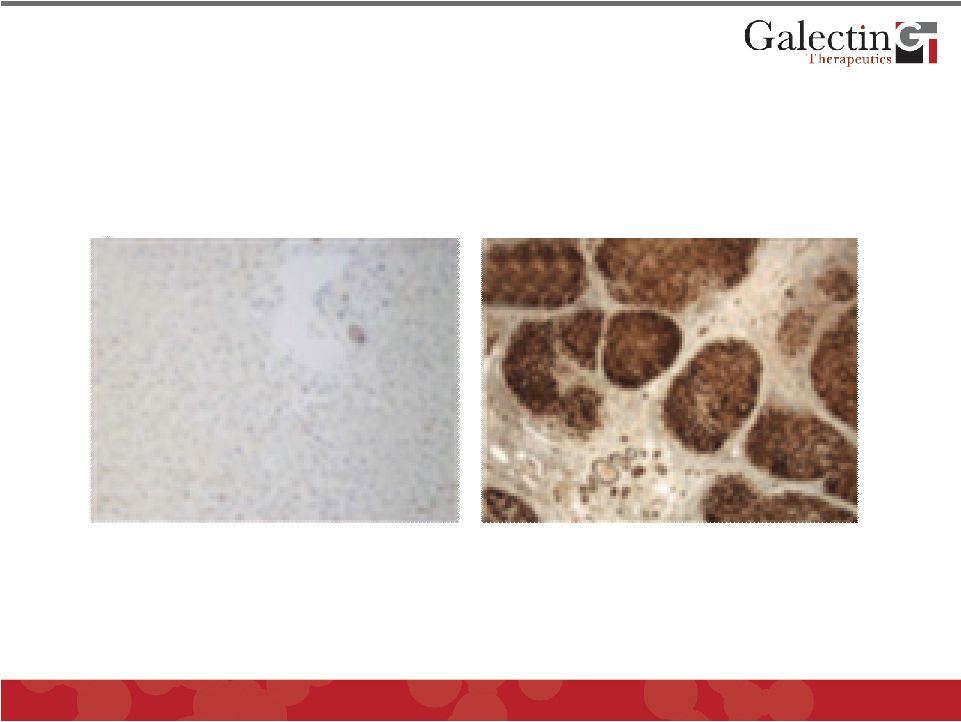
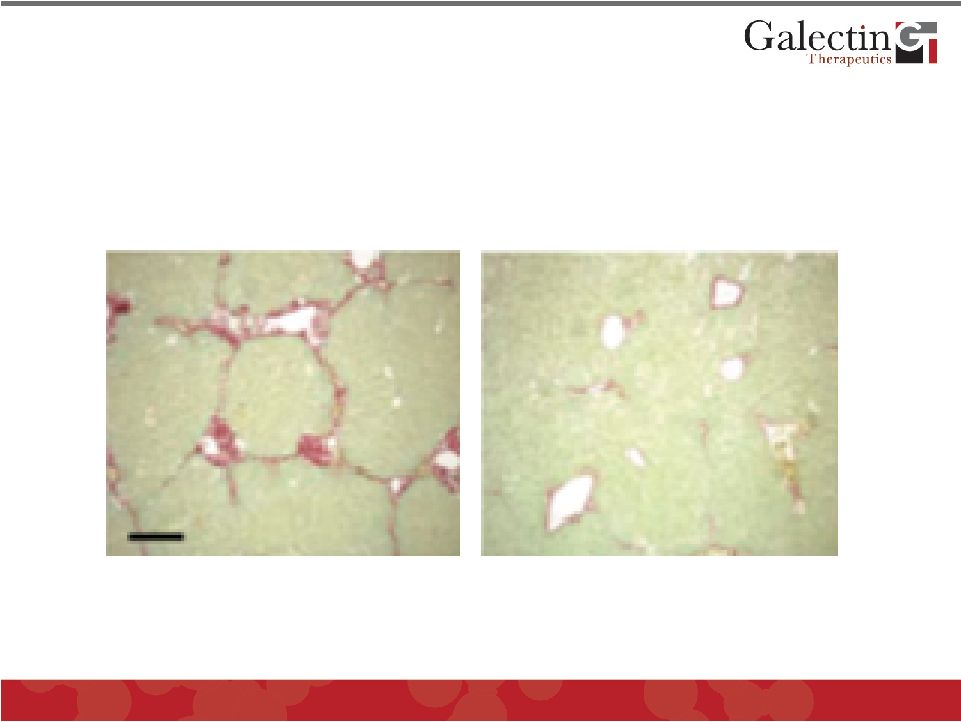
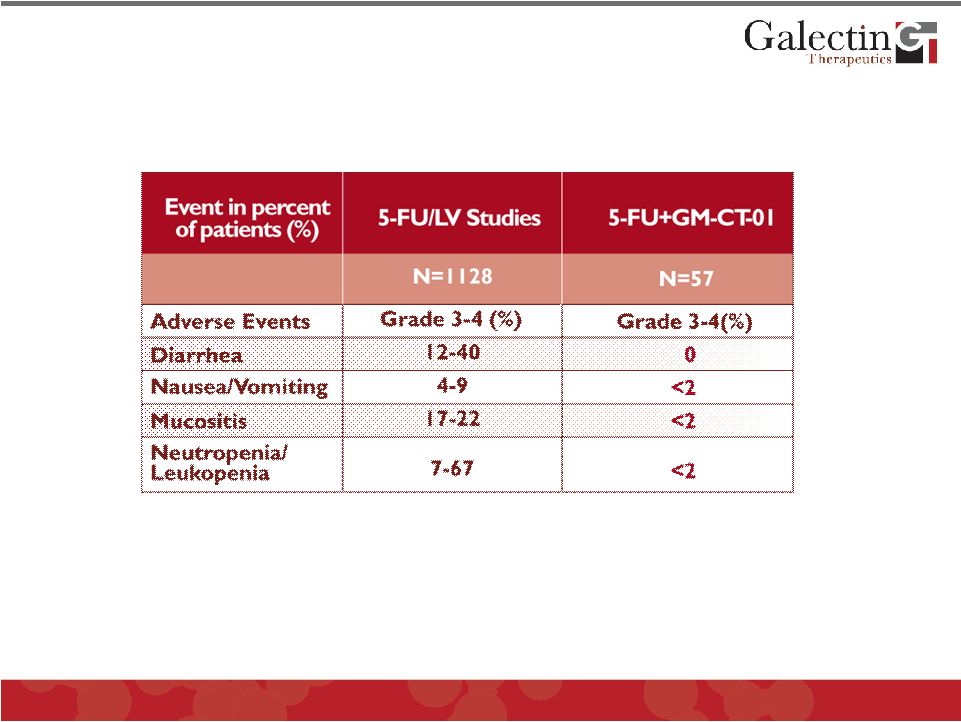
Forward Looking Statements This presentation contains, in addition to historical information, forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements relate to future events or future financial performance, and use words such as “may,” “estimate,” “could,” “expect” and others. They are based on our current expectations and are subject to factors and uncertainties which could cause actual results to differ materially from those described in the statements. Factors that could cause our actual performance to differ materially from those discussed in the forward-looking statements include, among others: incurrence of operating losses since our inception, uncertainty as to adequate financing of our operations, extensive and costly regulatory oversight that could restrict or prevent product commercialization, inability to achieve commercial product acceptance, inability to protect our intellectual property, dependence on strategic partnerships, product competition, and others stated in risk factors contained in our SEC filings. We cannot assure that we have identified all risks or that others may emerge which we do not anticipate. You should not place undue reliance on forward-looking statements. Although subsequent events may cause our views to change, we disclaim any obligation to update forward-looking statements. © 2011 Galectin Therapeutics 2 |