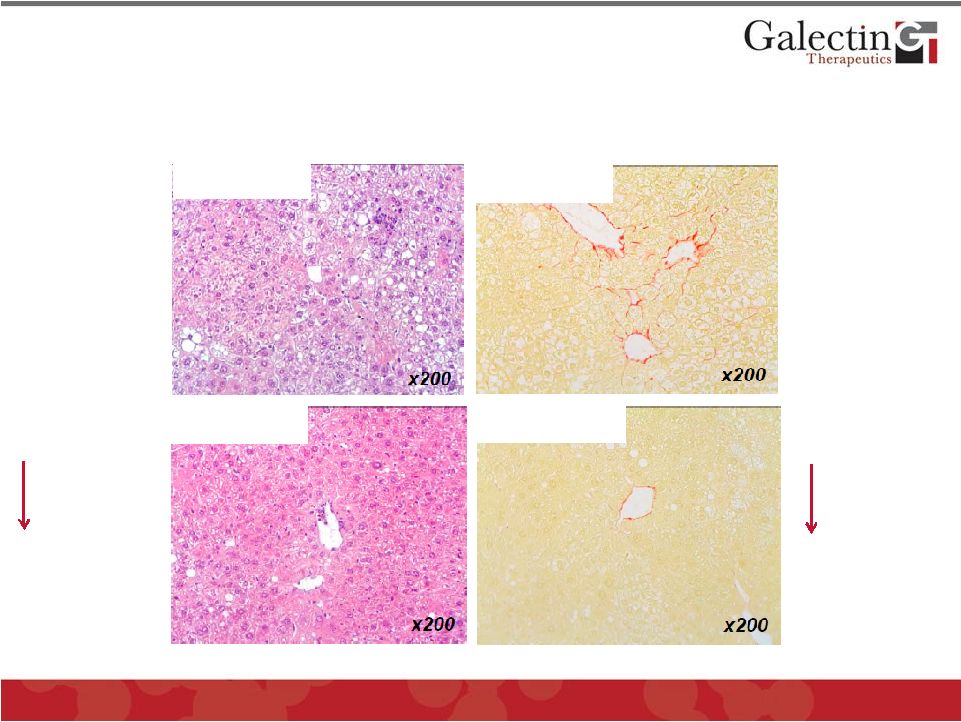
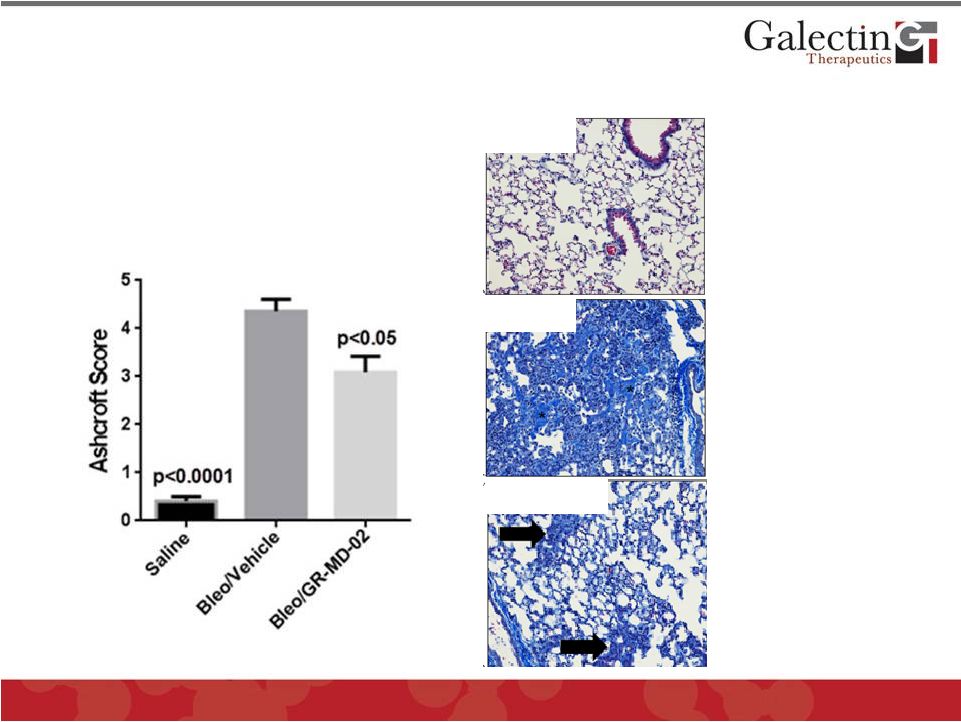
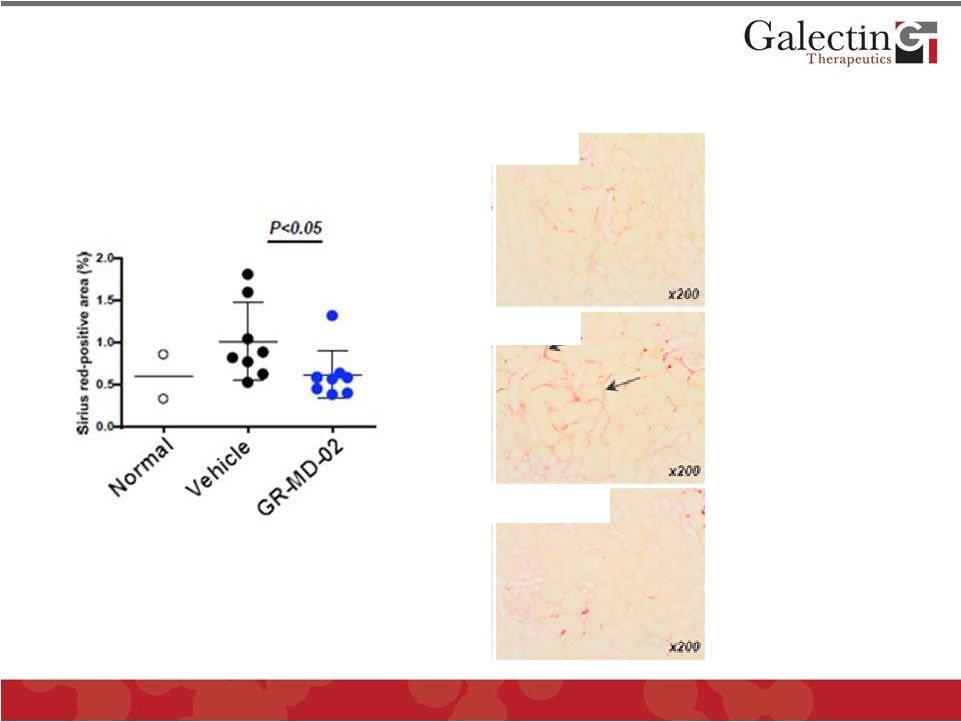
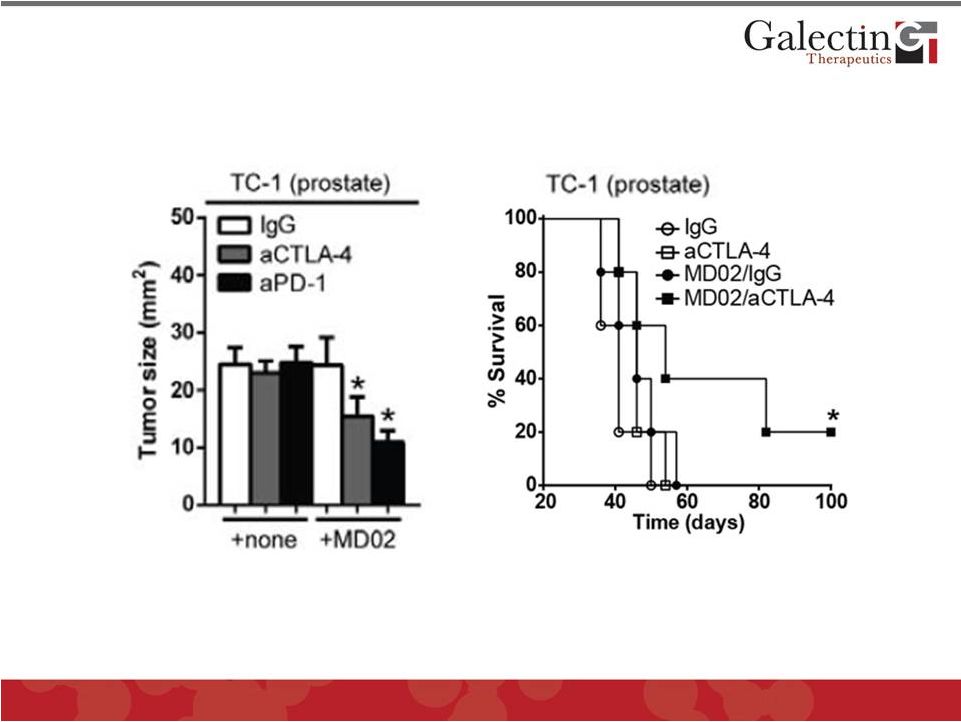
Forward Looking Statements This presentation contains, in addition to historical information, statements that look forward in time or that express management’s beliefs, expectations or hopes. Such statements are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements relate to future events or future financial performance, and use words such as "may," "estimate," "could," "expect" and others. They are based on our current expectations and are subject to risks and uncertainties that could cause actual results to differ materially from those described in the statements. These statements include our plans, expectations and goals regarding drugs in development, clinical trials and regulatory approval for any of our drugs or treatments, the anticipated timeline for clinical trials and results, related market opportunities for our drugs, potential benefits of our drugs, efforts related to partnering opportunities with other companies, estimates regarding cash and spending, liquidity and funding requirements for clinical trials, and estimates regarding those impacted by NASH, liver fibrosis and cirrhosis. The risks and uncertainties impacting these statements include that our plans, expectations and goals regarding drugs in development, clinical trials and regulatory approval are subject to factors beyond our control. Our clinical trials may not begin or produce positive results in a timely fashion, if at all, and any necessary changes during the course of such trials could prove time consuming and costly. We may have difficulty in enrolling candidates for testing and we may not be able to achieve the desired results. Upon receipt of regulatory approval for any drug or treatment, we may face competition with other drugs and treatments that are currently approved or those that are currently in development, which could have an adverse impact on our ability to achieve revenues from the approved indication. Plans regarding development, approval and marketing of any of our drugs are subject to change at any time based on the changing needs of our company as determined by management and regulatory agencies. Estimates regarding the potential benefits of our drugs and the potential market for any of our drugs may be inaccurate and, to the extent the estimates are correct, we may not be successful in achieving revenues from any such drugs, as the successful marketing of any approved drugs will be subject to strong competition within the health care industry and patient and physician acceptance of our drugs as safe, affordable and effective. Our ongoing discussions with other companies may not lead to partnering opportunities, and if we are unable to partner with other companies and/or raise additional capital, we will likely be unable to complete future stages of clinical trials and ultimately produce revenue from our drugs in development. Funding from potential sources of capital, including the potential exercise of warrants, may not materialize. To date, we have incurred operating losses since our inception, and our ability to successfully develop and market drugs may be impacted by our ability to manage costs and finance our continuing operations. For a discussion of additional factors impacting our business, see our most recent Annual Report on Form 10-K and our subsequent filings with the SEC. You should not place undue reliance on forward-looking statements. Although subsequent events may cause our views to change, we disclaim any obligation to update forward-looking statements. 2 © 2014 Galectin Therapeutics NASDAQ:GALT |