1
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 20-F
AMENDMENT NUMBER ONE
| [X] | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission file number: 0-31092
MEDICURE INC.
________________________________________________________________________________
(Exact name of registrant as specified in its charter)
Canada
_________________________________________________________________________________
(Jurisdiction of incorporation or organization)
4 - 1200 Waverley Street, Winnipeg, Manitoba, Canada R3T 0P4 _________________________________________________________________________________
(Address of principal executive offices)
Securities registered or to be registered pursuant to Section 12(b) of the Act: None
Securities registered or to be registered pursuant to Section 12(g) of the Act:
Common Shares, without par value
Class A Common Shares, without par value
_________________________________________________________________________________
(Title of Class)
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act: None
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report:
At May 31, 2002, the registrant had 37,088,864 common shares and 1,280,000 Class A common shares outstanding
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes No _______
No _______
Indicate by check mark which financial statement item the registrant has elected to follow:
Item 17 Item 18 _______
Item 18 _______
As of October 31, 2002, the buying rate for Canadian dollars was US $0.6421 for Cdn$1.00.
2
TABLE OF CONTENTS
| GLOSSARY | 4 | |
| FORWARD LOOKING STATEMENTS | 5 | |
| PART I | 5 | |
| ITEM 1 - IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISERS | 5 | |
| A. | Directors and Senior Management | 5 |
| B. | Advisers | 5 |
| C. | Auditors | 5 |
| ITEM 2 - OFFER STATISTICS AND EXPECTED TIMETABLE | 5 | |
| ITEM 3 - KEY INFORMATION | 5 | |
| A. | Selected Financial Data | 5 |
| B. | Capitalization and Indebtedness | 8 |
| C. | Reasons for the Offer and Use of Proceeds | 8 |
| D. | Risk Factors | 8 |
| ITEM 4 - INFORMATION ON THE COMPANY | 15 | |
| A. | History and Development of the Company | 15 |
| B. | Business Overview | 16 |
| C. | Organizational Structure | 23 |
| D. | Property, Plants and Equipment | 23 |
| ITEM 5 - OPERATING AND FINANCIAL REVIEW AND PROSPECTS | 23 | |
| A. | Operating Results | 23 |
| B. | Liquidity and Capital Resources | 25 |
| C. | Research and Development, Patents and Licenses, etc | 27 |
| D. | Trend Information | 29 |
| ITEM 6 - DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES | 29 | |
| A. | Directors and Senior Management | 29 |
| B. | Compensation | 34 |
| C. | Board Practises | 35 |
| D. | Employees | 35 |
| E. | Share Ownership | 35 |
| ITEM 7 - MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS | 37 | |
| A. | Major Shareholders | 37 |
| B. | Related Party Transactions | 37 |
| C. | Interests of Experts and Counsel | 38 |
| ITEM 8 - FINANCIAL INFORMATION | 39 | |
| A. | Consolidated Statements and Other Financial Information | 39 |
| B. | Significant Changes | 39 |
| ITEM 9 - THE OFFERING AND LISTING | 40 | |
| A. | Offer and Listing Details | 40 |
| C. | Markets. | 40 |
| ITEM 10 - ADDITIONAL INFORMATION | 41 | |
| A. | Share Capital | 41 |
| B. | Memorandum and Articles of Association | 41 |
| C. | Material Contracts | 44 |
| D. | Exchange Controls | 44 |
| E. | Taxation | 45 |
| F. | Dividends and Paying Agents | 46 |
| G. | Statement by Experts. | 46 |
| H. | Documents on Display | 47 |
| ITEM 11 - QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET | ||
| RISK | 47 | |
| ITEM 12 - DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES | 47 | |
| PART II | 47 | |
| ITEM 13 - DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES | 47 | |
3
| ITEM 14 - MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS | |
| AND USE OF PROCEED | 47 |
| ITEM 15 - CONTROLS AND PROCEDURES | 47 |
| ITEM 16 - RESERVED | 47 |
| PART III | 47 |
| ITEM 17 - FINANCIAL STATEMENTS | 47 |
| ITEM 18 - FINANCIAL STATEMENTS | 70 |
| ITEM 19 - EXHIBITS | 70 |
4
GLOSSARY OF TERMS
The following words and phrases shall have the meanings set forth below:
"angina"means chest pain;
"angioplasty"means the surgical repair of a blood vessel;
"anti-hypertensive"means blood pressure reducing; "arrhythmia" means irregular heart rhythm;
"bioavailability”means the degree to which a drug or other substance becomes available to the target in the body after administration;
"Computer Aided Drug Design" means a method for design of new therapeutic molecules using computer generated models of the drug and its molecular target;
"FDA"means the United States Food and Drug Administration;
"GCP"means Good Clinical Practices;
"GLP"means Good Laboratory Practice;
"GMP" means Good Manufacturing Practice;
"IND" means Investigative New Drug application to a regulatory authority for first human testing of a new drug;
"in-vitro" means test tube;
"in-vivo" means live animal;
"ischemia" means the lack of blood flow;
"myocardial infarction" means scarring and death to portions of the heart wall;
"myocardial ischemia" means blockages to parts of the heart muscle;
"NDS" means New Drug Submission, which is a request made to the HPB for commencement of product sales and marketing;
"NSAID" means non-steroidal anti-inflammatory drugs;
"pharmacodynamics" means the fundamental processes through which a drug(s) exerts its effects on living organisms;
pharmacokinetics" means the uptake, biotransformation, distribution, metabolism and elimination of a drug(s) by the body, including both total amounts and tissue and organ concentrations.;
"reperfusion" means the resumption of blood flow;
"TPD" means the Canadian Therapeutic Products Directorate, formerly the Canadian Health Protection Branch;
5
FORWARD LOOKING STATEMENTS
Medicure Inc. cautions readers that certain important factors (including without limitation those set forth in this Form 20-F) may affect the Corporation’s actual results and could cause such results to differ materially from any forward-looking statements that may be deemed to have been made in this Form 20-F annual report, or that are otherwise made by or on behalf of the Corporation. For this purpose, any statements contained in the annual report that are not statements of historical fact may be deemed to be forward-looking statements. Without limiting the generality of the foregoing, words such as “may,” “except,” “believe,” “anticipate,” “intend,” “could,” “estimate,” or “continue,” or the negative or other variations of comparable terminology, are intended to identify forward-looking statements.
As used in this annual report, the "Corporation” refers to “Medicure Inc.”, the company resulting from the amalgamation of Medicure Inc. and Lariat Capital Inc., “Medicure” refers to “Medicure Inc.” prior to its amalgamation with Lariat Capital Inc. and “Lariat” refers to Lariat Capital Inc. prior to its amalgamation with Medicure Inc.
PART I
ITEM 1. IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISERS
A. Directors and Senior Management
Not applicable
B. Advisers
Not applicable
C. Auditors
Not applicable
ITEM 2. OFFER STATISTICS AND EXPECTED TIMETABLE
Not applicable
ITEM 3. KEY INFORMATION
A. Selected Financial Data
The selected financial data of the Corporation as at May 31, 2002 and 2001 and for the fiscal years ended May 31, 2002 and 2001 and for the nine month period ended May 31, 2000 was extracted from the audited consolidated financial statements of the Corporation included in the annual report. The information contained in the selected financial data is qualified in its entirety by reference to the more detailed consolidated financial statements and related notes included in Item 17 - Financial Statements, and should be read in conjunction with such financial statements and with the information appearing in Item 5 - Operating and Financial Review and Prospects. The attached financial data as at May 31, 2000 and August 31, 1999 and 1998 and for the fiscal year ended August 31, 1999 and for the period from inception on September 15, 1997 to August 31, 1998 was extracted from the audited financial statements of the Corporation not included in this annual report. Reference is made to Note 9 of the consolidated financial statements of the Corporation included herein for a discussion of the material differences between Canadian GAAP and U.S. GAAP, and their effect on the Corporation’s financial statements.
6
To date, the Corporation has not generated sufficient cash flow from operations to fund ongoing operational requirements and cash commitments. The Corporation has financed its operations principally through the sale of its equity securities. While the Corporation believes it has sufficient capital and liquidity to finance current operations, nevertheless, its ability to continue operations is dependent on the ability of the Corporation to obtain additional financing. See “Item 3 - Key Information - D. Risk Factors.”
Under Canadian Generally Accepted Accounting Principles (in Canadian dollars):
| Period | ||||||||||
| from | ||||||||||
| Nine | Inception | |||||||||
| Months | on | |||||||||
| Year Ended | Year Ended | Ended | Year Ended | September | ||||||
| 15, | ||||||||||
| May 31, | May 31, | May 31, | August 31, | August 31, | ||||||
| Balance Sheet Data | 2002 | 2001 | 2000 | 1999 | 1998 | |||||
| (as at period end) | $ | $ | $ | $ | $ | |||||
| Current Assets | 8,783,318 | 4,045,011 | 4,149,722 | 184,485 | 58,901 | |||||
| Capital Assets | 84,571 | 63,941 | 74,385 | 4,214 | 3,473 | |||||
| Patent Costs | 508,902 | 313,088 | 185,836 | 87,296 | 10,201 | |||||
| Total Assets | 9,376,791 | 4,422,040 | 4,409,943 | 275,995 | 73,214 | |||||
| Total Liabilities | 389,663 | 570,077 | 191,830 | 769,013 | 313,480 | |||||
| Net Assets (Liabilities) | 8,987,128 | 3,851,963 | 4,218,113 | (493,018 | ) | (240,266 | ) | |||
| Capital Stock | 17,458,936 | 8,448,684 | 5,582,824 | 100 | 100 | |||||
| Deficit Accumulated During | ||||||||||
| the Development Stage | (8,471,808 | ) | (4,596,721 | ) | (1,364,711 | ) | (493,118 | ) | (240,366 | ) |
| Statement of Operations | ||||||||||
| (for the period indicated) | ||||||||||
| Gross Revenue | 183,912 | 135,868 | 54,943 | 1,555 | 25,000 | |||||
| Loss from Continuing | ||||||||||
| Operations | (3,875,087 | ) | (3,232,010 | ) | (871,593 | ) | (252,752 | ) | (240,366 | ) |
| Net Loss for the Period | (3,875,087 | ) | (3,232,010 | ) | (871,593 | ) | (252,752 | ) | (240,366 | ) |
| Basic and Diluted Loss per | (0.14 | ) | (0.20 | ) | (0.11 | ) | (0.03 | ) | (0.03 | ) |
| Share | ||||||||||
| Weighted-Average Number | ||||||||||
| of | ||||||||||
| Common Shares | 27,900,412 | (1) | 15,928,521 | (1) | 8,224,389 | (1) | 9,500,000 | (2) | 9,500,000 | (2) |
| Outstanding |
Under U.S. Generally Accepted Accounting Principles (in Canadian dollars):
| Period from | ||||||||||
| Nine | Inception on | |||||||||
| Year | Year Ended | Months | Year | September 15, | ||||||
| Ended | Ended | Ended | 1997 to | |||||||
| May 31, | May 31, | May 31, | August 31, | August 31, | ||||||
| Balance Sheet Data | 2002 | 2001 | 2000 | 1999 | 1998 | |||||
| (as at Period end) | $ | $ | $ | $ | $ | |||||
| Current Assets | 8,783,318 | 4,045,011 | 4,149,722 | 184,485 | 58,901 | |||||
| Capital Assets | 47,087 | 17,793 | 16,700 | 2,148 | 3,193 | |||||
| Patent Costs | - | - | - | - | - | |||||
| Total Assets | 8,830,405 | 4,062,804 | 4,166,422 | 186,633 | 62,094 |
7
| Total Liabilities | 389,663 | 570,077 | 191,830 | 769,013 | 313,480 | |||||
| Net Assets (Liabilities) | 8,440,742 | 3,492,727 | 3,974,592 | (582,380 | ) | (251,386 | ) | |||
| Capital Stock | 32,855,388 | 23,588,136 | 13,860,276 | 100 | 100 | |||||
| Deficit Accumulated | ||||||||||
| During | ||||||||||
| the Development Stage | (24,414,646 | ) | (20,095,409 | ) | (9,885,684 | ) | (582,480 | ) | (251,486 | ) |
| Statement of Operations | ||||||||||
| Gross Revenue | 183,912 | 135,868 | 54,943 | 1,555 | 25,000 | |||||
| Loss from Continuing | ||||||||||
| Operations | 4,319,237 | (10,209,725 | ) | (9,303,204 | ) | (330,994 | ) | (251,486 | ) | |
| Net Loss for the Period | 4,319,237 | (10,209,725 | ) | (9,303,204 | ) | (330,994 | ) | (251,486 | ) | |
| Basic and Diluted Loss per | (0.15 | ) | (0.64 | ) | (1.13 | ) | (0.03 | ) | (0.03 | ) |
| Share | ||||||||||
| Weighted-Average | ||||||||||
| Number of | ||||||||||
| Common Shares | 27,900,412 | (1) | 15,928,521 | (1) | 8,224,389 | (1) | 9,500,000 | (2) | 9,500,000 | (2) |
| Outstanding |
Note 1: Includes 1,280,000 Class A common shares outstanding
Note 2: The 9,500,000 common shares issued by Lariat to acquire Medicure are considered to be the outstanding number of shares for periods prior to November 23, 1999 for accounting purposes
Comparability of Data
On November 22, 1999, Lariat acquired all of the issued and outstanding common shares of Medicure in consideration for the issuance of 9,500,000 common shares of Lariat. As control of Lariat passed to the former shareholders of Medicure resulting in a reverse acquisition, Medicure is deemed to be the acquirer for accounting purposes. Accordingly, the net assets of Medicure are included in the balance sheet at book values and the deemed acquisition of Lariat is accounted for by the purchase method with the net assets of Lariat recorded at their fair value at the date of acquisition.
The selected financial data for the fiscal years ended May 31, 2002 and 2001 includes the operations of Medicure International Inc. (“Medicure International”) commencing June 1, 2000. The selected financial data for the nine months ended May 31, 2000 includes the operations of Medicure commencing September 1, 1999 combined with the activities of Lariat beginning on November 22, 1999, the effective date of the reverse takeover. The selected financial data for the year ended August 31, 1999 and for the period from inception on September 15, 1997 to August 31, 1998 are those of the financial statements of Medicure.
8
Dividends
No cash dividends have been declared nor are any intended to be declared. The Corporation is not subject to legal restrictions respecting the payment of dividends except that they may not be paid to render the Corporation insolvent. Dividend policy will be based on the Corporation 's cash resources and needs and it is anticipated that all available cash will be required to further the Corporation’s research and development activities for the foreseeable future.
Exchange Rates
Unless otherwise indicated, all reference to dollar amounts are in Canadian dollars. The following table sets out the exchange rates for one Canadian dollar expressed in terms of one U.S. dollar for the periods indicated. Rates of exchange are obtained from the Bank of Canada and believed by the Registrant to approximate closely the noon buying rates in New York City for cable transfers as certified for customs purposes by the Federal Reserve Bank in New York.
| May 31 | May 31, | May 31, | August 31, | August 31, | ||||||
| 2002 (1) | 2001(2) | 2000(3) | 1999(4) | 1998(5) | ||||||
| Period End | 0.6545 | 0.6470 | 0.6682 | 0.6700 | 0.6376 | |||||
| Average(6) | 0.6380 | 0.6600 | 0.6808 | 0.6637 | 0.6949 |
| Notes: | |
| (1) | For the year ended May 31, 2002 |
| (2) | For the year ended May 31, 2001 |
| (3) | For the nine months ended May 31, 2000 |
| (4) | For the year ended August 31 |
| (5) | For the period from inception on September 15, 1997 to August 31, 1998 |
| (6) | The average exchange rate figures are based on the average of each month during the period |
| October | Sept. | August | July | June | May | |||||||
| 2002 | 2002 | 2002 | 2002 | 2002 | 2002 | |||||||
| High for | 0.6421 | 0.6441 | 0.6455 | 0.6613 | 0.6613 | 0.6545 | ||||||
| Month(1) | ||||||||||||
| Low for | 0.6257 | 0.6300 | 0.6246 | 0.6294 | 0.6460 | 0.6364 | ||||||
| Month(1) |
Notes:
(1) Figures are extracted from daily exchange rates
As of October 31, 2002, the exchange rate to convert one Canadian dollar into the U.S. dollar was 0.6421
B. Capitalization and Indebtedness
Not applicable
C. Reasons for the Offer and Use of Proceeds
Not applicable
D. Risk Factors
The Corporation is subject to a number of risks due to the nature of its business and the present stage of development of business. The following factors should be considered:
9
THE CORPORATION’S PROSPECTS MUST BE CONSIDERED IN LIGHT OF THEDIFFICULTIES FREQUENTLY ENCOUNTERED BY COMPANIES IN THE RESEARCH AND DEVELOPMENT STAGE. MOREOVER, THE CORPORATION IS A RECENTLY PUBLIC CORPORATION WITH A LIMITED HISTORY OF OPERATIONS WHICH MAKES
EVALUATION OF ITS PROSPECTS DIFFICULT.
The Corporation began operations in 1997 and its common shares were listed on the Alberta Stock Exchange (a predecessor of the Canadian Venture Exchange) on March 30, 1999 to November 28, 1999 and on the Canadian Venture Exchange from November 29, 1999 to March 14, 2002. The Corporation commenced trading on the Toronto Stock Exchange on March 15, 2002 to the present.
The Corporation has concentrated on research and development and has a limited operating history. The Corporation's prospects must be considered in light of the risks, expenses and difficulties frequently encountered with the establishment of a development stage company in a highly competitive industry, characterized by frequent new product introductions. The Corporation has had no earnings to date, and may never have earnings or positive cash flow in the future. As a result of these factors, it is difficult to evaluate the Corporation’s business and prospects for future profitability, and its success is more uncertain than if it had a longer or more proven history of operations.
THE CORPORATION EXPECTS TO CONTINUE TO INCUR SUBSTANTIAL LOSSES AND MAY NEVER ACHIEVE PROFITABILITY, WHICH IN TURN MAY HARM THE FUTURE OPERATING PERFORMANCE AND MAY CAUSE THE MARKET PRICE OF THECORPORATION’S STOCK TO DECLINE.
The Corporation has incurred net losses every year since inception in 1997 and as of May 31, 2002, had an accumulated deficit of $8,471,808. The Corporation incurred net losses of $3,875,087 for the year ended May 31, 2002, $3,232,010 for the year ended May 31, 2001, $871,593 for the nine months ended May 31, 2000 and $252,752 for the fiscal year ended August 31, 1999.
The Corporation anticipates that its losses will not only continue for the foreseeable future but will increase significantly principally from expenditures relating to its research and development efforts and clinical trials. The long-term profitability of the Corporation’s operations is uncertain, and may never occur, and will be directly related to the success of its research and development activities which depend on numerous factors, including the following:
| a) | obtaining Canadian and United States regulatory approvals to market MC-1, the Corporation’s current lead product; |
| b) | the ability to manufacture the Corporation’s products according to schedule and within budget, given it has no experience in large scale manufacturing; and |
| c) | the ability to successfully market the Corporation’s products, given it has no experience in marketing; |
If the Corporation does not become profitable within the time frame expected by its investors, which may never happen, the market price of the Corporation’s stock will likely decline. If the Corporation does achieve profitability, it may not be able to sustain or increase profitability in the future.
THE CORPORATION MAY NEVER RECEIVE REGULATORY APPROVAL IN CANADA OR ABROAD FOR ANY OF ITS PRODUCTS DEVELOPED. THEREFORE, THE CORPORATION MAY NOT BE ABLE TO SELL ANY THERAPEUTIC PRODUCTS DEVELOPED.
The Corporation’s failure to obtain necessary regulatory approvals to fully market its current and future therapeutic products in one or more significant markets may adversely affect the Corporation’s business, financial condition and results of operations. The procedure involved in obtaining regulatory approval from the competent authorities to market therapeutic products is long and costly and may delay product development. The approval to market a product may be applicable to a limited extent only or it may be refused entirely.
10
The Corporation’s products and technologies are currently in the preliminary research and development stages. The Corporation does not and may never have a commercially viable drug formulation approved for marketing. To obtain regulatory approvals for the Corporation’s products and to achieve commercial success, human clinical trials must demonstrate that the products are safe for human use and that they show efficacy. Unsatisfactory results obtained from a particular study relating to one or more of the Corporation’s products may cause the Corporation to reduce or abandon its commitment to that program.
In April 2001, the Corporation completed a Phase I safety and pharmacokinetics study for MC-1, its lead product. In March 2002 the Corporation commenced a Phase II multi-centre “Proof of Principle” clinical trial of 60 high-risk cardiovascular patients undergoing elective angioplasty with the lead product, MC-1. The Phase II clinical trial will be designed to evaluate the ability of MC-1 to reduce heart damage from myocardial infarction (heart attack) and to protect the heart against injury resulting from coronary bypass surgery and angioplasty (a condition referred to as ischemia reperfusion injury).
The Corporation would not obtain approval from the Canadian Therapeutic Products Directorate, formerly the Canadian Health Protection Branch (TPD) or the United States Food and Drug Administration (FDA) to market MC-1 if it failed to successfully complete its Phase II clinical studies, or subsequent studies required. Regulatory approvals may also be subject to conditions that could limit the market for MC-1 or make it more difficult or expensive to sell than anticipated. Also, regulatory approvals may be revoked for a variety of reasons at any time, including the Corporation’s failure to comply with regulatory requirements or poor performance of MC-1 in terms of safety and effectiveness.
The Corporation’s business, financial condition and results of operations may be adversely affected if it failed to obtain regulatory approvals in Canada, the United States or abroad to market MC-1 or any future therapeutic products, including any limitations imposed to market such products.
THE CORPORATION MAY NOT BE ABLE TO HIRE OR RETAIN THE QUALIFIED SCIENTIFIC, TECHNICAL AND MANAGEMENT PERSONNEL IT REQUIRES.
The Corporation is currently dependent on CanAm Bioresearch Inc. (“CanAm”) for most of its research and development activities. Because of the specialized scientific nature of the Corporation’s business, the loss of services of CanAm may require the Corporation to attract and retain qualified scientific, technical and management personnel. Competition in the biotechnology industry for such personnel is intense and the Corporation may not be able to hire or retain a sufficient number of qualified personnel, which may compromise the pace and success of its research and development activities.
Also, certain management personnel of the Corporation are officers and/or directors of other publicly- traded companies and will only devote part of their time to the Corporation. The Corporation does not have key man insurance in effect in the event of a loss of any management personnel.
THE CORPORATION FACES SUBSTANTIAL TECHNOLOGICAL COMPETITION FROM MANY BIOTECHNOLOGY COMPANIES WITH MUCH GREATER RESOURCES, AND IT MAY NOT BE ABLE TO EFFECTIVELY COMPETE.
Technological competition in the pharmaceutical industry is intense. The Corporation competes with other companies in Canada, the United States and abroad to develop products designed to treat similar conditions. Many of these other companies have substantially greater financial, technical research and development resources and production and marketing capabilities than the Corporation. Smaller companies may also prove to be significant competitors, particularly through collaborative arrangements with large pharmaceutical and biotechnology companies. Developments by other companies may adversely affect the competitiveness of the Corporation’s products or technologies or the commitment of the Corporation’s research collaborators to its programs.
The pharmaceutical industry is also characterized by extensive research efforts and rapid technological change. Competition can be expected to increase as technological advances are made and commercial applications for biopharmaceutical products increase. Competitors of the Corporation may use different
11
technologies or approaches to develop products similar to products which the Corporation is seeking to develop, or may develop new or enhanced products for processes that may be more effective, less expensive, safer or more readily available before the Corporation obtains approval of its products. The Corporation may not be able to successfully compete with its competitors and, if it is unable to do so, the Corporation’s business, financial condition and results of operations may suffer.
THE CORPORATION MAY BE UNABLE TO ESTABLISH COLLABORATIVE AND COMMERCIAL RELATIONSHIPS WITH THIRD PARTIES.
The success of the Corporation will depend partly on its ability to enter into various arrangements with corporate partners, licensors, licensees and others for the research, development, manufacturing, marketing and commercialization of its products. To date, the Corporation has not entered into any such arrangements and may never be able to establish such arrangements on favourable terms. The failure to establish successful collaborative arrangements with respect to certain products may negatively impact the Corporation's ability to commercialize those products and adversely affect the Corporation’s business, financial condition and results of operations.
The Corporation has licensed certain technologies relating to products under development and may enter into future licensing agreements. The Corporation's current licensing agreements contain provisions allowing the licensors to terminate such agreements if the Corporation becomes insolvent or breaches the terms and conditions of the licensing agreement, without rectifying same upon notice.
THE CORPORATION DOES NOT HAVE MANUFACTURING OR MARKETING EXPERIENCE AND MAY NEVER BE ABLE TO SUCCESSFULLY MANUFACTURE OR MARKET ITS PRODUCTS.
The Corporation has no experience in large-scale manufacturing and in marketing its products and may never be able to successfully manufacture and market its products. If the TPD or FDA approves MC-1 or any future products, the Corporation intends to rely on third parties to manufacture or market its products. Accordingly, the quality, timing and ultimately the commercial success of such products may be outside the Corporation’s control. Failure of or delay by a manufacturer of the Corporation's products to comply with Good Manufacturing Practices or similar quality control regulations or satisfy regulatory inspections may have a material adverse effect on the future prospects of the Corporation. Also, providers, payers or patients may not accept the Corporation's products, even if they prove to be safe and effective and are approved for marketing by the TPD, the FDA and other regulatory authorities. The Corporation estimates that it may take up to 5 years or longer before the Corporation's initial products may be sold commercially. Other competitors may be in a position to bring competing products to market within a shorter time frame.
THE CORPORATION DOES NOT HAVE PRODUCT LIABILITY INSURANCE AND MAY NOT BE ABLE TO OBTAIN ADEQUATE PRODUCT LIABILITY INSURANCE IN THE FUTURE.
The sale and use of products under development by the Corporation, and the conduct of clinical studies involving human subjects, may entail risk of product liability. The Corporation does not currently have product liability insurance in place for its products but expects that, as it expands, the Corporation will acquire such insurance. However, the Corporation may not be able to obtain appropriate levels of product liability insurance prior to the sale of its products. An inability to obtain insurance on economically feasible terms or to otherwise protect against potential product liability claims could inhibit or prevent the commercialization of products developed by the Corporation. The obligation to pay any product liability claim or recall a product may have a material adverse effect on the business, financial condition and future prospects of the Corporation.
IF THE CORPORATION IS UNABLE TO SUCCESSFULLY PROTECT ITS PROPRIETARY RIGHTS, THE CORPORATION’S COMPETITIVE POSITION WILL BE ADVERSELY AFFECTED.
12
The success of the Corporation will depend partly on its ability to obtain and protect its patents and protect its proprietary rights in unpatented trade secrets.
The Corporation owns or jointly owns four United States patents and has received a Notice of Allowance for one other patent from the United States Patent Office. The Corporation has an additional three pending United States patent applications. The Corporation’s pending and any future patent applications may not be accepted by the United States Patent and Trademark Office or any other jurisdiction in which applications may be filed. Also, processes or products that may be developed by the Corporation in the future may not be patentable.
The patent protection afforded to biotechnology and pharmaceutical companies is uncertain and involves many complex legal, scientific and factual questions. There is no clear law or policy involving the degree of protection afforded under patents. As a result, the scope of patents issued to the Corporation may not successfully prevent third parties from developing similar or competitive products. Competitors may develop similar or competitive products that do not conflict with the Corporation’s patents. Litigation may be commenced by the Corporation to prevent infringement of its patents. Litigation may also commence against the Corporation to challenge the Corporation’s patents that, if successful, may result in the narrowing or invalidating of such patents. It is not possible to predict how any patent litigation will affect the Corporation's efforts to develop, manufacture or market its products. However, the cost of litigation to prevent infringement or uphold the validity of any patents issued to the Corporation may be significant in which case the Corporation’s business, financial condition and results of operations may suffer.
Disclosure and use of the Corporation 's proprietary rights in unpatented trade secrets not otherwise protected by patents are generally controlled by written agreements. However, such agreements will not provide the Corporation with adequate protection if they are not honoured, others independently develop equivalent technology, disputes arise concerning the ownership of intellectual property or the Corporation's trade secrets are disclosed improperly. To the extent that consultants or other research collaborators use intellectual property owned by others in their work with the Corporation, disputes may also arise as to the rights to related or resulting know-how or inventions.
OTHERS COULD CLAIM THAT THE CORPORATION INFRINGES ON THEIRPROPRIETARY RIGHTS, WHICH MAY RESULT IN COSTLY AND TIME CONSUMING LITIGATION.
The success of the Corporation will depend partly on its ability to operate without infringing upon the patents and other proprietary rights of third parties. The Corporation is not aware that any of its products or processes infringe the proprietary rights of third parties. However, despite the best efforts of the Corporation, it may be sued for infringing on the patent or other proprietary rights of third parties at any time in the future.
Such litigation, with or without merit, is time-consuming and costly and may significantly impact the Corporation’s financial condition and results of operations, even if the Corporation prevails. If it does not prevail, the Corporation, in addition to any damages it may have to pay, may be required to stop the infringing activity or enter into a royalty or licensing agreement. The Corporation may not be able to obtain such a license or the terms of the royalty or license may be burdensome for the Corporation, which may significantly impair its ability to market its products and adversely affect the business, financial condition and results of operations of the Corporation.
THE CORPORATION IS, AND IN THE FUTURE MAY BECOME, SUBJECT TOADDITIONAL GOVERNMENTAL REGULATIONS AND IF IT IS UNABLE TO COMPLY WITH THEM, THE CORPORATION’S BUSINESS MAY BE MATERIALLY HARMED.
The Corporation is or may become subject to various federal, provincial, state and local laws, regulations and recommendations. The Corporation is subject to various laws and regulations in Canada, relating to
13
product emissions, use and disposal of hazardous or toxic chemicals or potentially hazardous substances, infectious disease agents and other materials, and laboratory and manufacturing practices used in connection with its research and development activities. If the Corporation fails to comply with these regulations, the Corporation may be fined or suffer other consequences that could materially affect its business, financial condition or results of operations.
The Corporation is unable to predict the extent of future government regulations or industry standards. However, it should be assumed that government regulations or standards will increase in the future. New regulations or standards may result in increased costs, including costs for obtaining permits, delays or fines resulting from loss of permits or failure to comply with regulations.
THE CORPORATION WILL NEED TO RAISE ADDITIONAL CAPITAL THOUGH THE SALE OF ITS SECURITIES, RESULTING IN DILUTION TO THE EXISTING SHAREHOLDERS, AND WHICH MAY NOT BE AVAILABLE, ADVERSELY AFFECTING ITS OPERATIONS.
The Corporation has not to date generated any revenues from sales. The timing of generation of any sales is uncertain. Based on the Corporation’s current plans, the Corporation’s available working capital will be sufficient to complete its planned proof of principle Phase II clinical trial of MC-1, its lead product, expected to be completed by December 2002.
The Corporation has limited financial resources and has financed its operations through the sale of securities, primarily common shares. The Corporation will need to continue its reliance on the sale of such securities for future financing, resulting in dilution to the Corporation’s existing shareholders. The Corporation's long-term capital requirements will depend on many factors, including continued scientific progress in its product discovery and development program, progress in its pre-clinical and clinical evaluation of products and product candidates, time and expense associated with filing, prosecuting and enforcing its patent claims and costs associated with obtaining regulatory approvals. In order to meet such capital requirements, the Corporation will consider contract fees, collaborative research and development arrangements, public financing or additional private financing (including the issuance of additional equity securities) to fund all or a part of particular programs.
The Corporation’s business, financial condition and results of operations will depend on its ability to obtain additional financing which may not be available under favorable terms, if at all. The ability of the Corporation to arrange such financing in the future will depend in part upon the prevailing capital market conditions as well as the business performance of the Corporation. If the Corporation’s capital resources are exhausted and adequate funds are not available, it may have to reduce substantially or eliminate expenditures for research and development, testing, production and marketing of its proposed products, or obtain funds through arrangements with corporate partners that require the Corporation to relinquish rights to certain of its technologies or products.
FUTURE ISSUANCE OF THE CORPORATION’S COMMON SHARES WILL RESULT IN DILUTION TO THE EXISTING SHAREHOLDERS. ADDITIONALLY, FUTURE SALES OF THE CORPORATION’S COMMON SHARES INTO THE PUBLIC MARKET MAY LOWER THE MARKET PRICE WHICH MAY RESULT IN LOSSES TO THE CORPORATION’S SHAREHOLDERS.
As of May 31, 2002, the Corporation had 37,088,864 common shares issued and outstanding. A further 1,973,033 common shares are issuable upon exercise of outstanding stock options and another 18,403,076 common shares are issuable upon exercise of share purchase warrants, all of which may be exercised in the future resulting in dilution to the Corporation’s shareholders. The Corporation’s stock option plan allows for the issuance of stock options to purchase up to a maximum of 3,700,000 of the common shares issued and outstanding as of May 31, 2002. Accordingly, stock options to purchase an additional 1,726,967 common shares are issuable by the Corporation as at May 31, 2002. With the exception of 9,092,338 common shares held in escrow, these common shares, including the common shares to be issued upon exercise of the outstanding options and warrants, are freely tradable. It should be noted that 7,606,404 shares still held in escrow are performance based escrow shares and 5,780,867 of these
14
performance based escrow shares are currently eligible for release upon application being made, by the Corporation, to the Toronto Stock Exchange.
Sales of substantial amounts of the Corporation’s common shares into the public market, or even the perception by the market that such sales may occur, may lower the market price of its common shares.
THE CORPORATION’S COMMON SHARES MAY EXPERIENCE EXTREME PRICE AND VOLUME VOLATILITY WHICH MAY RESULT IN LOSSES TO THE SHAREHOLDERS OF THE CORPORATION.
On May 31, 2002, the Corporation’s common shares closed at a price of $0.49. For the period from June 1, 2001 to May 31, 2002, the high and low trading prices of the Corporation’s common shares were $1.24 and $0.40, respectively, with a total trading volume of 6,750,700 shares.
Daily trading volume in the Corporation’s common stock for the period from June 1, 2001 to May 31, 2002 has generally been limited, with a high of 500,500 shares and a low of 150 shares, averaging approximately 25,865 shares. Accordingly, the trading price of the Corporation’s common stock may be subject to wide fluctuations in response to a variety of factors including announcement of material events by the Corporation such as the status of required regulatory approvals for the Corporation’s products, competition by new products or new innovations, fluctuations in the operating results of the Corporation, general and industry-specific economic conditions and developments pertaining to patent and proprietary rights.
The securities markets in the United States and Canada have recently experienced a high level of price and volume volatility, and the market price of securities of biotechnology companies have experienced wide fluctuations in price which have not necessarily been related to the operating performance, underlying asset values or prospects of such companies. In addition, because of the limited public float, there may be limited liquidity for the Common Shares. It is expected that such fluctuations in price and limited liquidity will continue in the foreseeable future which may make it difficult for a shareholder to sell shares at a price equal to or above the price at which the shares were purchased.
THE CORPORATION’S COMMON SHARES ARE CONSIDERED “PENNY STOCK” WHICH MAY HAVE THE EFFECT OF REDUCING THE LEVEL OF TRADING ACTIVITY AND MAKE IT MORE DIFFICULT TO SELL SUCH SHARES.
The Corporation’s shares are “penny stock” as defined by the Securities and Exchange Commission, which might affect the trading market for the shares. Penny stocks are generally equity securities with a price of less than U.S.$5.00 other than securities registered on certain national securities exchanges or quoted on the NASDAQ National Market. The Securities and Exchange Commission has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document prepared by the Securities and Exchange Commission that provides information about penny stocks and the nature and level of risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid and compensation information must be given to the customer orally or in writing before or with the customer’s confirmation. In addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from such rules, the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for a stock that is subject to the penny stock rules, such as the Corporation’s shares which are considered “penny stock”, and therefore make it more difficult to sell those shares.
The Corporation commenced trading on the Toronto Stock Exchange on March 15, 2002.
15
THE CORPORATION HAS NO HISTORY OF PAYING DIVIDENDS, DOES NOT INTEND TO PAY DIVIDENDS IN THE FORESEEABLE FUTURE AND MAY NEVER PAY DIVIDENDS.
Since incorporation, the Corporation has not paid any cash or other dividends on its common stock and does not expect to pay such dividends in the foreseeable future as all available funds will be invested to finance the growth of its business. The Corporation will need to achieve profitability prior to any dividends being declared which may never happen.
ITEM 4. INFORMATION ON THE COMPANY
A. History and Development of the Company
Lariat Capital Inc. (“Lariat”) was incorporated by Certificate of Incorporation issued pursuant to the provisions of theBusiness Corporations Act (Alberta) on June 3, 1997. On February 11, 1999, by Certificate of Amendment and Registration of Restated Articles, the Articles of Lariat were amended to remove the private company restriction. Lariat was formed as a Junior Capital Pool company, as defined by, and under the rules of the Alberta Stock Exchange with the expressed intent of acquiring a project or company through a reverse take over. With the exception of this intent and the associated search for potential acquisitions, Lariat had no substantial prior business activities.
Medicure Inc. (“Medicure”) was incorporated by Certificate of Incorporation issued pursuant to the provisions ofThe Corporations Act (Manitoba) on September 15, 1997. Medicure was continued from Manitoba to Alberta by Certificate of Continuance issued pursuant to the provisions of theBusiness Corporations Act (Alberta) on December 3, 1999. On December 22, 1999, Medicure and Lariat were amalgamated by Certificate of Amalgamation issued pursuant to the provisions of theBusiness Corporations Act (Alberta) as Medicure Inc. The Corporation was continued from Alberta to the federal jurisdiction by Certificate of Continuance issued pursuant to the provisions of theCanada Business Corporations Acton February 23, 2000.
Medicure was formed as a private Manitoba company to advance the discoveries of Dr. Naranjan Dhalla of the University of Manitoba. Dr. Dhalla and Dr. Albert Friesen were the principal owners of the corporation as first formed, together with certain other individuals who contributed to the project. The first order of business was the completion of a licensing agreement to acquire the technology rights from the University of Manitoba, which owned the technology by virtue of the fact that it was invented by employees of the University. From that date until the merger with Lariat, the Corporation's primary focus was on the preclinical testing and development of the lead product, identified as MC-1. In 1998 other research involving synthesis and testing of other potential therapeutics was commenced through a research contract with the University of Manitoba. Various business activities were conducted in support of these primary research projects including but not limited to; (1) the application for and approval of government sponsored research awards, (2) the search for alternative sources of investment capital to fund operations, and (3) the ongoing search for other potential therapeutics. Business and administrative functions were handled by Genesys Venture Inc., a consulting corporation, that at the time, was owned entirely by Dr. Friesen. Operations and research, until the merger, were primarily funded by Dr. Friesen, with assistance from government grants. On November 22, 1999 Medicure was acquired by Lariat by way of a reverse takeover as Lariat’s “Major Transaction” as a Junior Capital Pool company within the meaning of the Alberta Securities Commission Rule 46-501, the Alberta Securities Commission Companion Policy 46-501CP and The Alberta Stock Exchange Circular 7. Pursuant to the terms of the Major Transaction, Lariat acquired all of the issued and outstanding shares of Medicure in exchange for 9,500,000 shares of Lariat, at a deemed price for securities regulatory purposes only of $0.20 per share for aggregate deemed value of $1,900,000. The Major Transaction was negotiated entirely at arm’s length. As a result of the share exchange, control of Lariat passed to the former shareholders of Medicure. This type of transaction is commonly referred to as a “reverse takeover”. Under reverse takeover accounting, for financial reporting purposes, the Corporation is considered to be a continuation of the operations formerly carried on by Medicure.
16
The Corporation’s current registered office is 30th Floor, 360 Main Street, Winnipeg, Manitoba, Canada, R3C 4G1. The Corporation’s head office is located at 4-1200 Waverley Street, Winnipeg, Manitoba, Canada, R3T 0P4.
The MC-1 technology was originally licensed to Genesys Pharma Inc. by the University of Manitoba, on August 18, 1997. Genesys Pharma Inc., which had made a small investment on some preliminary research, transferred the technology without cost, except for costs designated in the license, to Medicure Inc. on September 26, 1997. On August 30, 1999 Medicure Inc. completed a new license agreement with the University of Manitoba in order to slightly modify the terms of the original license agreement transferred from Genesys Pharma Inc., and to have the documentation properly prepared in the Corporation’s own name.
On June 1, 2000 the Corporation licensed the world-wide development and marketing rights (except for Canada) for MC-1, the Corporation’s lead product, to the Corporation’s wholly owned subsidiary, Medicure International. Medicure International then entered into a Development Agreement with CanAm to perform research and development on MC-1 and other compounds at cost, plus a reasonable mark-up not to exceed ten percent of any amount invoiced. The parties to the Development Agreement agreed that the aggregate amount of all invoiced expenditures shall not exceed $10,000,000 over the term of the agreement. CanAm is a private Canadian company wholly owned by Peter de Visser, a former director of the Corporation. Peter de Visser resigned as a director of the Corporation in December 2001.
Since its amalgamation, the Corporation, directly and through certain research contracts, has been engaged in the research and development of human therapeutic drugs for cardiovascular disease. In certain instances, therapeutics developed by the Corporation may also provide benefit for other diseases. The Corporation’s lead product, MC-1, is based upon scientific discoveries led by Dr. Naranjan S. Dhalla of The Institute of Cardiovascular Sciences and the Department of Physiology, of the Faculty of Medicine, the University of Manitoba in Winnipeg, Manitoba, Canada. The Corporation’s focus is on the clinical development and commercialization of MC-1 for treatment of cardiovascular disease and on the discovery and development of other cardiovascular therapeutics. There are currently three part time and 16 full time scientific researchers and support staff who are retained as consultants or employees by CanAm and who are performing the Corporation’s scientific research pursuant to the Development Agreement.
B. Business Overview
Plan of Operation
It is the Corporation’s intention to focus on the discovery and development of pharmaceuticals. The Corporation intends to license the sale and distribution of commercialized products to larger, international companies. As such, the Corporation intends to derive its revenue primarily through milestone payments and product royalties. As of the date hereof, other than the Medicure International Licensing Agreement and the University of Manitoba License Agreement, the Corporation has not entered into any license agreements or other arrangements regarding the sale or distribution of any of its products.
The Corporation’s focus is on the discovery and development of therapeutics for various large-market, unmet cardiovascular needs. The Corporation’s research and development program is designed to address these market needs with the clinical development of the Corporation’s lead product, MC-1, and with the potential discovery and development of other drug candidates. MC-1 is a natural compound that has demonstrated effectiveness in safely treating various forms of cardiovascular disease in initial research.
MC-1 is being developed as a treatment to reduce injury from blockages of blood to the heart (i.e. myocardial ischemia, associated with heart attacks, angina and arrhythmia) and prevent injury from ischemic reperfusion injury. Ischemic reperfusion injury occurs when blood flow to an organ is suddenly resumed following a stoppage, as occurs during medical procedures such as angioplasty and heart
17
surgery. MC-1 may also be developed for other cardiovascular disorders for which it has shown potential benefit, including certain forms of hypertension that are difficult to treat (i.e. high blood pressure).
A Phase I human clinical trial for MC-1, the Corporation’s lead product, was commenced by the Corporation on October 20, 2000 after receiving approval of its Investigational New Drug (IND) application by Canada’s Therapeutic Products Directorate (“TPD”) (formerly the Health Protection Branch). On April 19, 2001, the Phase I trial was completed.
In March, 2002 the Corporation commenced a Phase II multi-centre “Proof of Principle” clinical trial of 60 high-risk cardiovascular patients undergoing elective angioplasty with our lead product, MC-1 after obtaining approval from the TPD and the FDA. The Corporation expects to have results available by the end of the calendar year.
The Corporation is currently gathering data to support a Phase I clinical application in an intravenous formulation by Q4 calendar 2002, subject to approval by TPD. The initial Phase I clinical trials will be designed to evaluate the safety of MC-1 in an intravenous formulation in healthy volunteers.
In parallel to the development of MC-1, the Corporation has a drug discovery program the objective of which is to discover and in-license new drug candidates for advancement into clinical development and commercialization for unmet cardiovascular market needs. The program involves the synthesis of compounds that utilize the same scaffold as MC-1. The Corporation has already produced several groups of candidate compounds and plans to build a pipeline of additional preclinical products over the next several years. Certain of the Corporation’s new compounds have shown positive effects in in vitro andin vivo efficacy studies and are currently being studied further to evaluate their commercial potential. Patent applications have been, and are expected to be filed for all candidate compounds, to the extent commercially and reasonably possible, protecting their composition of matter and use in a treatment of targeted cardiovascular and related diseases.
The Corporation is also evaluating other cardiovascular drug candidates for potential license with the objective of further broadening its product and patent portfolio.
The Corporation anticipates that no substantial material acquisition of equipment or facilities will take place in the coming year. However, CanAm has spent approximately $300,000 on leasehold improvements and $350,000 on equipment in the past year that has enabled it to perform the Corporation’s scientific research pursuant to the Development Agreement.
Dr. Friesen, a director, the Chairman, the President and the Chief Executive Officer of the Corporation is also the majority shareholder, a director and an officer of Genesys Venture Inc. (“GVI”), a company with which the Corporation had contracted for the provision of accounting and administrative services until January 31, 2002. Pursuant to the said agreement between GVI and the Corporation dated as of October 1, 2001, GVI provided certain business functions for the Corporation for $85,600 per annum, payable monthly. Specifically, GVI provided the following business services to the Corporation: operational support including providing insurance coverage, office amenities and computer support; financial management services including providing accounting support and payroll services; and providing physical office and storage facilities. GVI, which is co-owned and managed by Dr. Friesen, is a business development organization specialized in providing experienced and cost effective administrative and accounting services to biotechnology companies. As of February 1, 2002, the Corporation terminated the agreement with GVI and has no further obligations to GVI under this agreement.
CanAm retains no ownership and derives no direct benefit from the Corporation’s biotechnology products. CanAm’s development contract with the Corporation is on the basis of billing for direct costs plus a fee of up to 10%. CanAm derives an indirect benefit from the research contract by the experience and developmental knowledge retained by its employees, as CanAm will also be providing research and development services to other biotech firms on a confidential and non-competitive basis with the Corporation. As a private company, CanAm is entitled to certain Canadian government assistance that is not available, in Canada, to public companies. The flexibility CanAm has in providing additional work
18
and career opportunities for its employees in fields other than those of interest to the Corporation, allows CanAm to provide additional qualified staff that the Corporation could not easily attract.
In summary, it is the Corporation’s intention over the next year to continue its focus on being a drug discovery and development company and to begin to actively search for a partnership with a large pharmaceutical company. Such a partnership would conceivably provide funding for Phase II/III clinical trials, add experience to the product development process and bring in overall marketing expertise. While the Corporation has had informal discussions with potential partners, no formal agreement, or letter of intent, has been entered into by the Corporation as of the date hereof.
Potential Products in Development Stage
As previously stated, one of the Corporation’s primary focuses is the clinical development and commercialization of its lead product, MC-1, a natural small molecule that has shown potential for safely and effectively treating various forms of cardiovascular disease and stroke.
The effectiveness of MC-1 in treating the damage done to the heart by myocardial ischemia, including common conditions such as heart attack and angina, has been demonstrated in established, industry acceptedin vivo models. One primary model is the coronary artery ligation model, which involves inflicting a blockage in a major artery going into the heart of the animal thereby mimicking blockages involved in human heart attacks. Benefits of treatment suggested in a series of animal studies in this model are improved cardiac function and reduced negative change in heart size.
MC-1’s effectiveness in preventing ischemic reperfusion injury has been demonstrated inin vitro andin vivo models that are routinely used throughout the industry. A selection of benefits seen in these studies include improved contractile force and reduced infarct size.
Although the precise mechanism by which MC-1 acts, is not yet known, research indicates that its effect occurs from a new cardiovascular mechanism, thereby offering potential to further extend the benefits beyond current therapeutic alternatives.
Additional preclinical studies with MC-1 also suggest its potential value in treatment of stroke. During 2002, promising preclinical studies were carried out under the direction of Dr. Ashfaq Shuaib, Director of the Division of Neurology at the WC Mackenzie Health Sciences Centre of the University of Alberta.MC-1 reduced infarct size (damaged region) in the brain and preserved neurological function in an animal model. Preliminary studies also indicate that beneficial effects may even be obtained with treatment several hours after the onset of ischemia. A combination of MC-1 and TPA was also shown to be an effective treatment. There was no indication thatMC-1 alone increased the incidence of hemorrhage, suggesting it would be a safe treatment for stroke patients. Medicure plans further research on stroke, hypertension and other potential uses.
While MC-1 development proceeds, the Corporation is seeking to develop or acquire additional cardiovascular therapeutics with commercial potential to meet a market need. The Corporation’s objective is to establish a pipeline of novel cardiovascular therapeutics to ensure the Corporation’s long-term growth and security. The Corporation is taking a two-pronged approach to this effort, combining the efforts of a drug discovery program with strategic licensing of promising new compounds from other research groups.
The Corporation’s drug discovery program has produced several families of new compounds that have shown promising effects inin vitro and/orin vivo studies. From these compounds, the Corporation has thus far identified certain candidates, as having potential for further development. These compounds, the chemical identities of which are being held confidential while patents are pending, will undergo furtherin vitro analysis andin vivo animal testing on disease models and for bioavailability.
19
According to theAmerican Heart Association, cardiovascular disease is the most prevalent serious disease in the United States, affecting approximately 58.8 million people.1According to theAmerican Heart Association, one in five Americans have some form of cardiovascular disease. In 1996, cardiovascular disease was a primary contributing cause of mortality on 60% of death certificates in the United States. The rates in Canada are similar, with the subset of ischemic heart disease accounting for the greatest percentage of deaths at 21%.2
The Corporation is focusing its initial drug discovery and development efforts on meeting unmet needs in the cardiovascular and stroke market. The Corporation is advancing its lead product, MC-1, into clinical testing with the intention of commercializing it for treatment of (1) ischemic reperfusion injury (associated with common procedures such as angioplasty and coronary bypass surgery and stroke); and(2) myocardial ischemia, including heart attacks, angina and other related disorders. The Corporation intends to subsequently develop MC-1 for other cardiovascular disorders such as difficult to treat forms of hypertension.
Medicure (MC-1) completed Phase I testing in Canada on April 19, 2001, and initiated a Phase II trial in the United States and Canada in March 2002, with FDA and TPD approval. The results of the trial are expected in December 2002.
Medicure anticipates the commencement of a “Proof of Principle” Phase II clinical trial of MC-1 in a second indication in 2003.
The Corporation has various compounds currently in early stage research screening. Compounds developed by the chemistry group will be advanced through the following steps based on their successful advancement through early stage screening; (1) in vitro (cell based) screening, (2) in vivo (live animal) screening in appropriate disease model, (3) in vivo toxicity screening, (4) dose response confirmation of efficacy testing in vivo, (5) preclinical, GLP toxicity and pharmacokinetics testing, (6) Phase I human testing, (7) Phase II human, (8) Phase III human, (9) Filing of NDA in United States and NDS in Canada to request the right to commercialize. At the present time the most advanced product is in stage 4 of this testing continuum, but no estimate can be made as to when a certain product may advance to the human clinical testing stage.
The Corporation currently has four issued patents. United States Patent No. 6,051,587 terminates on April 16, 2018, United States Patent No. 6,043,259 terminates on March 25, 2019, United States Patent No. 6,339,085 terminates on March 7, 2000 and United States Patent No. 6,417,204 terminates on July 6, 2021.
Competitors’ Current Products
There are numerous products on the market for treatment of cardiovascular disorders, most of which are marketed by large pharmaceutical companies.
It is recognized that cardiovascular treatments have been of great benefit in reducing mortality and morbidity from a range of conditions. The existing cardiovascular drugs can be categorized into several main drug classes, as distinguished by their mechanism of action. Some of the primary drug classes include ACE Inhibitors (2001 global sales estimated at US$10 billion), Angiotensin II Inhibitors (2001 global sales estimated US$5.6 billion, Thrombolytics (1999 global sales estimated US$8.9 billion), Beta-Blockers (2001 global sales estimated at US$3.8 billion), and Calcium Channel Blockers (2001 global sales estimated at US$9.9 billion), each class has particular benefits as well as an array of alternative products.3Cross-use of drugs between different types of cardiovascular disease categories makes it
_________________________________
1 American Heart Association,1999 Heart and Stroke Statistical Update.
2Health Canada,Heart Disease and Stroke in Canada, 1997, Chapter 2.1.
3 Sales estimates provided byDatamonitor, 2002
20
difficult to differentiate sales by the more specific market segment (such as for myocardial infarction, ischemic reperfusion injury, etc.).4
Despite the development of various effective products, pharmaceutical companies carefully monitor developments in the field and continually attempt to bring in new major products. This is in part driven by the rise of generic products that substantially reduce profit margins for products as they come off patent. Competition is most intense in cardiovascular markets like hypertension where there are many treatment options. Large pharmaceutical companies are most interested in finding new treatment options for inadequately treated conditions such as those targeted by the Corporation.
Despite the number of cardiovascular products, the Corporation has identified certain remaining unmet therapy needs for certain forms of cardiovascular disease. For example, physicians recognize the current lack of effective products for reducing ischemic reperfusion injury. This is a very real clinical problem and a significant market is available for a product that would effectively protect against this injury that results from a variety of surgical procedures. Similarly, although current treatments are in many cases able to restore blood flow to the heart muscle following a heart attack (myocardial ischemia), there remains a need for products that reduce the amount of damage and scarring that results from the blockage. Other large cardiovascular markets targeted by the Corporation that require improved therapeutics are congestive heart failure and certain forms of hypertension.
Current cardiovascular drugs are difficult to prescribe due to the interrelated nature of disorders and the danger of negative side effects. Typically, after a person who has had a suspected heart attack or a case of angina is admitted to the emergency room, numerous points of diagnosis must be carried out to best identify the cause. Following this, a suitable course of therapy can be selected. Another difficulty in treatment is cost, with many of the best treatment options (such as anticoagulants) being too expensive for some health care providers.
Ischemic stroke is damage to the brain caused by a sudden reduction in blood supply, most often due to blood clots lodging in major arteries of the brain. Stroke ranks as the third leading cause of death in North America, behind diseases of the heart and cancer. Stroke affects approximately 5.1 million North Americans annually, with one in four strokes being fatal. It is also a leading cause, of long term disability in the U.S. As Baby Boomers age, the incidence of stroke will climb by 32% ever the next four years and 68% over the next 20 years.
To date, the only FDA approved stroke therapeutic is tissue plasminogen activator (TPA), a treatment that helps dissolve arterial obstructions. Unfortunately, TPA is typically available to less than 10% of stroke patients due to the increased risk of hemorrhage and the narrow therapeutic time frame during which the drug can be applied.
Competitors’ Products in Development
As there remain unmet needs for treatment of certain forms of cardiovascular disease and stroke, management of the Corporation believes there is room for an increased number of novel research products.
In 1995, the Pharmaceutical Researchers and Manufacturers of America identified 107 new products in development for heart disease and stroke.5This is fewer than the number of new products identified for cancer (215) and aging (132). In its 1998 report,R & D Directions identifies approximately 157 separate products in development for heart disease and stroke. A large proportion of products in development are modifications of existing therapeutics.
_________________________________
4 Supra, note 9.
5 PhRMA, Medicines in Development” Surveys.
21
Medicure’s clinical trials focus on ischemia and ischemic reperfusion injury for cardiovascular diseases and will also focus on stroke. The following tables provides an overview of competitors’ products in development pertaining to ischemia and ischemic reperfusion injury of cardiovascular diseases and stroke:
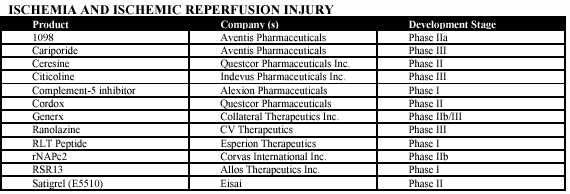
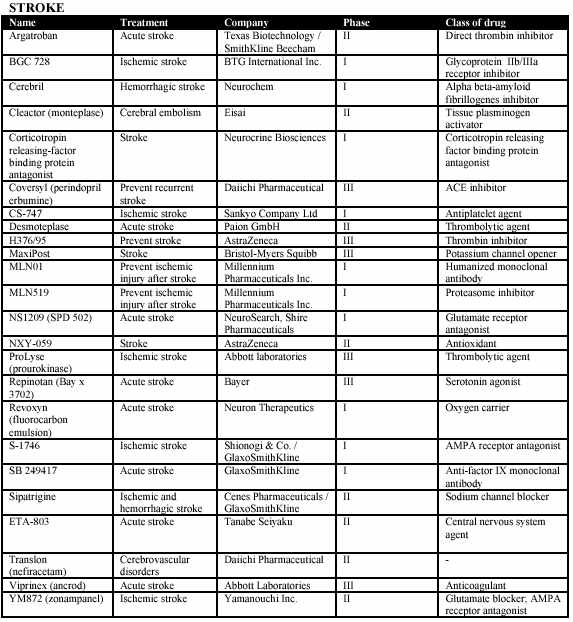
22
Some of the products set forth above have the advantage of being further along in development than the Corporation’s products. However, market share for new products depends primarily upon the relative strengths of a product in areas such as safety, effectiveness, cost, and dose form.
It is noted that cardiovascular drugs are often prescribed together and therefore products do not necessarily compete for use on an individual patient. MC-1’s use in many indications is expected to be as adjunct therapy, that is, it will be given together with other therapeutics. Historically, the challenge of competing with earlier products has not acted as a significant barrier to entry for new products, provided that the new product has some advantage relative to earlier products.
Competitive Strategy and Position
As stated, the Corporation is primarily focusing on developing MC-1 for myocardial ischemia and reperfusion injury. The Corporation is focusing initially on these markets because of preclinical evidence supporting the product’s efficacy in these applications and, therefore, these applications have high potential for showing MC-1’s clinical benefit. The clinical need for a product with this activity will also be considered by regulatory authorities (principally the FDA and also the TPD). Although MC-1 shows potential for treatment of other cardiovascular diseases, these uses are not being addressed as a first priority due to factors including the cost of clinical trials and the more intense competitive nature of these markets.
It is the Corporation’s intention to secure a partnership with a large pharmaceutical company. Such a partnership would provide funding for clinical development, add experience to the product development process and provide market positioning expertise. While the Corporation has had informal discussions with potential partners in this regard, no formal agreement or letter of intent has been entered into by the Corporation as of the date hereof.
Management of the Corporation believes that, based on initial animal studies, MC-1 has many competitive advantages over existing competitor’s products and over products known to be in development. These advantages relate to MC-1’s effectiveness for a broad range of disorders, safety at higher than expected dosages, and the potential that the product acts by a new mechanism of action.
23
C. Organizational Structure
Medicure International, the Corporation’s wholly owned subsidiary, was incorporated pursuant to the laws of Barbados, West Indies, on May 23, 2000. Medicure International’s registered office is located at Whitepark House, White Park Road, Bridgetown, Barbados. Medicure International’s head office is located at 2nd Street, Holetown, St. James, Barbados.
The following diagram illustrates the relationship between the Corporation and Medicure International:

D. Property, Plants and Equipment
Office Space
The Corporation has use of about 2,100 square feet of office space provided by Waverly Business and Science Centre Inc. as part of its business services contract. The office is located in Winnipeg, Manitoba.
Research Facilities
CanAm, on behalf of the Corporation, has just recently leased 6,100 square feet of office and laboratory space at a newly built facility in Winnipeg, Manitoba. Biological, chemistry, and analytical research now takes place under one roof.
ITEM 5. OPERATING AND FINANCIAL REVIEW AND PROSPECTS
This section contains forward-looking statements involving risks and uncertainties. The Corporation 's actual results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including those set forth under part Item 3 - Key Information - D. Risk Factors. The following discussion of the financial condition, changes in financial conditions and results of operations of the Corporation for the periods ended May 31, 2002, May 31, 2001 and May 31, 2000, should be read in conjunction with the consolidated financial statements of the Corporation. The Corporation’s consolidated financial statements are presented in Canadian dollars and have been prepared in accordance with Canadian generally accepted accounting principles (“GAAP”) included under Item 17 to this annual report. Material differences between Canadian and U.S. GAAP, as applicable to the Corporation, are set forth in Note 9 to the consolidated financial statements of the Corporation included herein.
A. Operating Results
General
The Corporation has concentrated primarily on research and development and has yet to and may never derive any revenues from its general operations. The Corporation has a limited operating history and its prospects must be considered in light of the risks, expenses and difficulties frequently encountered with the establishment of a development stage company in a highly competitive industry, characterized by frequent new product introductions. The Corporation has historically incurred net losses and anticipates that such losses will increase as it continues its development and clinical trials and eventually seeks regulatory approval for the sale of its products.
24
The Corporation believes it has sufficient funds on hand to complete MEND-1, the Phase II clinical trial in angioplasty patients and a second Phase II clinical trial in another indication, with its lead product, MC-1. As discussed in Note 1 of the consolidated financial statements of the Corporation, however, the Corporation’s ability to continue as a going concern is dependent on its ability to obtain sufficient funds to conduct the remainder of its clinical trials and to successfully commercialize its products. Failure to obtain further financing may require the Corporation to reduce substantially or eliminate expenditures for research and development, testing including further clinical trials, and production and marketing of its proposed products.
Subsequent to August 31, 2000, the Corporation has received no government assistance towards its research and development expenditures. The Corporation, through its subsidiary Medicure International Inc., currently contracts out all its research and development expenses, and expects to do so for the foreseeable future. Thus, the Corporation itself is not eligible for research-related government assistance, nor does it expect to be eligible in the future. The Corporation's operations have remained stable for some time now, without government assistance, evidencing the fact that the loss of such assistance has not materially affected the Corporation.
Year Ended May 31, 2002 Compared to the Year Ended May 31, 2001
Interest and other income for fiscal 2002 totaled $183,912 as compared to $135,868 for fiscal 2001. Interest income was higher in fiscal 2002 primarily due to a larger cash and cash equivalents and short-term investments balance, which resulted from the net cash proceeds of $9.0 million raised during the year from a public offering.
Research and Development expenditures increased to $3,103,707 as compared to $2,472,951 for fiscal 2001. The increase in spending is primarily due to the Company’s continuing clinical development of MC-1, including the commencement of the “Proof of Principle” Phase II clinical trial (MEND-1) for treatment of angioplasty patients. In addition, the Company enhanced its research and development capabilities with the expansion in our research team, and a rise in screening and preclinical testing of compounds brought forward by the Company’s drug discovery program. The Company’s scientific developments led to the filing of numerous patents during the year.
Medicure expects the research and development expenditures for the fiscal year ended May 31, 2003 to be higher than fiscal 2002. The main source of the increase will result from the completion of the current Phase II clinical trial in angioplasty patients and the commencement of clinical trials in other indications.
General and administration expenses totaled $949,569 for the year ended May 31, 2002, as compared to $879,914 for the year ended May 31, 2001. This increase in spending is the result of the addition of personnel and professional services that were required to support increased business development activities, as well as increased regulatory fees resulting from the Company’s listing on the TSX.
Medicure expects general and administrative expenditures for fiscal 2003 to be slightly higher than those incurred in fiscal 2002
For the fiscal year ended May 31, 2002 (“fiscal 2002”), Medicure recorded a net loss of $3,875,087 or $0.14 per share compared to a net loss of $3,232,010 or $0.20 per share for the year ended May 31, 2001 (”fiscal 2001”). These results of operations were in line with management’s expectations. The Company expects losses to continue for the next several years due to expenditures relating to its research and development efforts and clinical trials.
The weighted average number of common shares and class A shares outstanding used to calculate basic and diluted loss per share increased to 27,900,412 for the year ended May 31, 2002 from 15,928,521 for the year ended May 31, 2001.
25
Year Ended May 31, 2001 Compared to Nine Months Ended May 31, 2000
In reviewing financial results for fiscal 2001 and 2000 it is noted that the length of the periods differed which materially impacts the comparative analysis.
Revenues of the Corporation increased to $135,868 for the year ended May 31, 2001 consisting entirely of interest income earned on cash deposits as compared to $54,943 for the nine months ended May 31, 2000. The increase in interest income is due primarily to the increase in cash and cash equivalent balance during the year to $3,775,472 as at May 31, 2001 as compared to $1,613,865 as at May 31, 2000 as a result of $2,865,860 in net cash proceeds received from equity financings and exercise of stock options and shares purchase warrants.
Research and development expenses for the year ended May 31, 2001 increased significantly to $2,472,951 from $547,461 for the nine months ended May 31, 2000 due to a number of factors. Clinical trial expenses (including drug formulation) increased by $628,938, pre-clinical research expenses increased by $672,709 and payroll expenses increased by $214,560. The increase in clinical trial costs was primarily due to the Phase I trial of the Corporation’s lead product, MC-1, and increased formulation work to develop an MC-1 pill for future clinical trial studies. The increase in pre-clinical research costs was primarily due to increased animal testing and toxicity studies of both MC-1 and other pre-clinical compounds. The increase in payroll costs was primarily the result of hiring additional scientists to help with preclinical research.
Research and development investment tax credit amounts were Nil for the year ended May 31, 2001 and for the nine months ended May 31, 2000. The Corporation received $2,436 of government assistance towards its research and development expenditures during the year ended May 31, 2001 as compared to $64,482 during the nine months ended May 31, 2000 due primarily to the timing and duration of the IRAP program grant.
General and administrative expenses for the year ended May 31, 2001 increased significantly to $879,914 from $435,692 for the nine months ended May 31, 2000. A significant part of this increase in expenses is a result of costs of $271,683 related to a financing that did not close. In addition, travel costs increased by $48,634, promotional material costs increased by $45,045 and printing costs increased by $29,847. The increase in travel costs was primarily a result of travel related to promoting the Corporation during a financing period. The increase in promotional material costs and printing costs was primarily the result of increased printing of shareholder materials and other promotional materials such as the Corporation’s annual report.
Net loss for the year ended May 31, 2001 increased significantly to $3,232,010 from $871,593 for the nine months ended May 31, 2000, resulting in basic and diluted loss per share of $0.20 compared to $0.11, respectively. The significant increase in net loss is primarily the result of an increase of $444,222 in general and administration expenses, an increase of $1,925,490 in research and development expenses and a decrease of $62,046 in government assistance for research and development expenditures received offset by an increase in interest income of $80,925. These increases are in part, due to the additional three months in the 2001 as compared to the 2000 period.
The weighted average number of common shares outstanding used to calculate basic and diluted loss per share increased to 15,928,521 for the year ended May 31, 2001 from 8,224,389 for the nine months ended May 31, 2000.
B. Liquidity and Capital Resources
Since inception, as the Corporation has incurred negative cash flow from operations its working capital requirements have been financed primarily from the issuance of shares from treasury. As at May 31, 2002, the Corporation has cash and cash equivalents of $8,341,018, an increase of $4,565,546 from the comparable data in 2001.
26
The increase in the cash position is primarily the result of the completion of a public offering with an initial closing on December 20, 2001 and a second closing on January 31, 2002, raising gross proceeds of $10 million (before financing costs of $1.2 million) by issue of 15,384,615 units priced at $0.65. Each unit comprises one common share and one common share purchase warrant. Each common share purchase warrant entitles the holder to acquire one common share at a price of $0.81 per common share on or before December 20, 2003.
Based on the Corporation’s current plans including the Phase II clinical trials of MC-1, it is estimated that the Corporation’s current working capital will be sufficient to enable meeting its operating needs until the beginning of Calendar 2004.
Following the MEND-1 trial and the second “Proof of Principle” Phase II clinical trial, and assuming satisfactory results, the Corporation will proceed with pivotal Phase II trials with a larger number of patients that could start as early as the fiscal 2004. These studies could cost $4,000,000 to $10,000,000 each and will hope to confirm the results from the earlier trials and provide more data regarding the pharmacological effects of the drug as well as the optimal drug dosage levels. The Corporation will require additional funding to meet the costs of such studies that may not be available if and when it is required.
Successful completion of a pivotal Phase II trial will ultimately lead to a Phase III trial. This is a large-scale study of patients with the targeted disease and will probably cost in the range of $16,000,000 to $40,000,000. It could potentially start in 2005 and finish in 2006. The main purpose of this trial is to obtain definitive statistical evidence of the efficacy and safety of the drug so that an application can be made to the TPD and FDA for commercial approval.
If the drug is approved for marketing by the TPD and FDA, there will then potentially be follow-up studies (called Phase IV trials) that may need to be completed while the drug is being marketed, in order to adjust or extend product claims. It is not possible to estimate the scope and size of any such studies that may be required.
It continues to be the Corporation’s intention to secure a partnership with a large pharmaceutical company. Such a partnership would provide funding for clinical development (most specifically Phase III) and could ultimately lead to a license agreement for the sale and distribution of the Corporation’s lead product in return for milestone payments and product royalties. In this way, the Corporation would rely on the marketing expertise of its partner, instead of developing its own marketing department.
While MC-1 progresses through clinical trials, the Corporation will continue the drug development process. This process begins with initial product screening which should produce preliminary candidates for preclinical research. MC 5422 and MC 45228 are such candidates that have moved into the preclinical research stage. Once a drug candidate reaches this stage, it can cost in excess of $1,000,000 to advance it to the clinical trial stage. Ongoing research and development to find preliminary candidates currently costs the Corporation about $1,200,000 per year.
Besides public or private financings, the other major financings are planned to be through up front and milestone payments from a large pharmaceutical company that has partnered with the Corporation for clinical development and or marketing of the lead compound.
When additional funds are required, potential sources of financing include strategic relationships and public or private sales of the Corporation’s common shares. The Corporation does not have any committed sources of financing at this time and it is uncertain whether additional funding will be available when the need arises on terms that will be acceptable to the Corporation. If funds are raised by selling additional common shares, or other securities convertible into common shares, the ownership interests of the Corporation’s existing shareholders will be diluted. If the Corporation is unable to obtain financing when required, the Corporation would not be able to carry out its business plan, including further clinical trials of MC-1. The Corporation would have to significantly limit its operations and business, and its financial condition and results of operations would be materially harmed.
27
At May 31, 2002, the Corporation had net working capital of $8,393,655 compared to net working capital of $3,474,934 at May 31, 2001. During the three months ended August 31, 2002, the Corporation raised a total of $25,200 in cash from exercised options.
At May 31, 2001, the Corporation had net working capital of $3,474,934 compared to net working capital of $3,957,892 at May 31, 2000. During the year ended May 31, 2001, the Corporation raised a total of $2,865,860 in cash through the issuance of shares from treasury. A private placement provided $2,446,753, net of share issuance costs of $153,247, and $419,107 was raised through the exercise of stock options and warrants.
The Corporation had no long-term debt as of May 31, 2002 or October 31, 2002 and no other significant expenditure commitments not related to operating activities.
No dividends have been declared or paid on any shares of the Corporation since its incorporation. There can be no assurance that the Corporation will ever declare any dividends on any of its shares, or if declared, what the dividend amounts will be or whether such dividends, once declared, will continue for any future period.
C. Research and Development, Patents and Licenses, Etc.
Research and Development
Drug development and design begins with an idea, or theoretical concept for treating a given disorder. The idea is advanced through the drug design process, resulting in preliminary candidates that have theoretical potential. Candidates are improved to achieve the optimal effectiveness with limited toxicity. Following preclinical testing, products with the greatest potential become lead candidates and are advanced into clinical trials (human testing) with the intent of having them receive regulatory approval for marketing.
The Corporation has demonstrated the effectiveness of MC-1 in preclinical animal models of cardiovascular disease and has advanced this product through Phase I clinical trials and into the current Phase II clinical trial called MEND-1. Additional preclinical studies may also be performed in order to evaluate other potential uses of MC-1 and to gather other animal data as may be necessary to support future clinical objectives.
In vitro andin vivo laboratory experiments to date have been used to study MC-1 therapeutic activity in myocardial ischemia, ischemic reperfusion injury, and hypertension. These experiments involved internationally recognized models (including coronary artery ligation, Langendorff, reperfusion and hypertensive rat models), and analysis techniques (such as electrocardiogram and hemodynamic measurements, and electron microscopy).In vivo studies involve oral and/or intravenous administration of the drug dose.
While MC-1’s development proceeds, novel chemical compounds will be synthesized or in-licensed and entered into further testing to evaluate their commercial potential. As previously set forth, the Corporation’s drug discovery program is pursuing medicinal chemistry strategies with the objective of maximizing the probability of commercial potential. Novel chemical compounds are screened for commercial viability prior to advancement into preclinical testing. These studies are to determine bioavailability/distribution, safety and efficacy (on bothin vitro andin vivo models).
For the period from inception on September 15, 1997 to May 31, 2002, a total of $6,448,000 was incurred for research and development.
28
Patents and Licenses
The Corporation has been issued four patents from the United States Patent Office providing protection for certain uses of MC-1 and related compounds in treatment of cardiovascular disease. The Corporation has obtained an additional Notice of Allowance and has also filed three additional applications in the United States plus corresponding patent applications in other jurisdictions. The Corporation will continue to file patents to extend protection of MC-1 and for new compounds in development. The four patents issued to the Corporation are as follows:
- US Patent 6,043,259
- US Patent 6,051,587
- US Patent 6,339,085
- US Patent 6,417,204
The first three patents are jointly owned by the Corporation and the University of Manitoba. Pursuant to a Licence Agreement dated August 18, 1997, an Assignment Agreement dated September 26, 1997, and an ensuing new License Agreement dated August 30, 1999 (the ‘‘Licence Agreement’’) the University of Manitoba licensed the exclusive worldwide use of the patents and the MC-1 technology to the Corporation. Pursuant to the License Agreement, the Corporation has agreed to pay the University of Manitoba a royalty payment of 3% of net sales from any cardiovascular product derived from the MC-1 technology. The License Agreement commenced on August 30, 1999 and shall terminate:
| (i) | if a patent or patents are obtained prior to commercialization of a Licensed Product (as defined therein), on the expiration date of the last to expire of any patents covered by the Patent Rights (as defined therein); and |
| (ii) | if a patent is not obtained prior to commercialization of a Licensed Product (as defined therein), on August 30, 2009. |
The MC-1 technology is derived from work done by Dr. Rajat Sethi as an employee of the Corporation and by two employees of the University of Manitoba, Dr. Naranjan Dhalla and Dr. Krishnamurti Dakshinamurti, Professor Emeritus, Department of Biochemistry.
The Notice of Allowance and three pending United States patent applications, including those filed with the United States Patent Office as either regular or provisional applications, are wholly owned by the Corporation by virtue of their inventorship by employees of the Corporation and, subsequent to June 1, 2000, by CanAm, a related party. The Notice of Allowance and two pending patent applications pertain to MC-1.
Much of the work, including some of the research methods, that is important to the success of the Corporation’s business is germane to the industry and may not be patentable. For this reason all employees, contracted researchers and consultants are bound by non-disclosure agreements.
Given that the patent applications for these technologies involve complex legal, scientific and factual questions, there can be no assurance that patent applications relating to the technology used by the Corporation will result in patents being issued, or that, if issued, the patents will provide a competitive advantage or will afford protection against competitors with similar technology, or will not be challenged successfully or circumvented by competitors.
The Corporation has filed patents in accordance with the Patent Cooperation Treaty (the ‘‘PCT’’). The PCT is a multilateral treaty that was concluded in Washington in 1970 and entered into force in 1978. It is administered by the International Bureau of the World Intellectual Property Organization (the ‘‘WIPO’’), headquartered in Geneva, Switzerland. The PCT facilitates the obtaining of protection for inventions where such protection is sought in any or all of the PCT contracting states (total of 104 at July 1999). It provides for the filing of one patent application (the ‘‘international application’’), with effect in several contracting states, instead of filing several separate national and/or regional patent applications.
29
At the present time, an international application may include designation for regional patents in respect of contracting states party to any of the following regional patent treaties: The Protocol on Patents and Industrial Designs within the framework of the African Regional Industrial Property Organization, the Eurasian Patent Convention, the European Patent Convention, and the Agreement Establishing the African Intellectual Property Organization. The PCT does not eliminate the necessity of prosecuting the international application in the national phase of processing before the national or regional offices, but it does facilitate such prosecution in several important respects by virtue of the procedures carried out first on all international applications during the international phase of processing under the PCT. The formalities check, the international search and (optionally) the international preliminary examination carried out during the international phase, as well as the automatic deferral of national processing which is entailed, give the applicant more time and a better basis for deciding whether and in what countries to further pursue the application. Further information may be obtained from the official WIPO internet website (http://www.wipo.int).
On June 1, 2000 the Corporation entered into the Medicure International Licensing Agreement whereby it licensed the world-wide development and marketing rights for MC-1, except for Canada, to its wholly owned subsidiary, Medicure International. As consideration for the grant of the license, Medicure International agreed to pay the Corporation a fee of $1.00 upon the completion of specified milestones in the development process, together with a variable royalty of 7% to 9% of net sales of MC-1 (if any sales are ever in fact made). The term of the Medicure International Licensing Agreement will expire on the date of expiration of the last to expire patent on MC-1, or in the absence of any such patent, on the 10th anniversary of the date of the first commercial sale of MC-1 in the country where it was last introduced (if it is ever so introduced). The Medicure International Licensing Agreement may be terminated under a number of circumstances and, in any event, by either party at any time by providing the other with at least 90 days prior written notice of its intention to terminate the Medicure International Licensing Agreement.
Medicure International subsequently entered into the Development Agreement with CanAm on June 1, 2000 to perform research and development of MC-1 and other compounds at cost, plus a reasonable mark-up not to exceed ten percent of any amount invoiced. The parties to the Development Agreement agreed that the aggregate amount of all invoiced expenditures shall not exceed $10,000,000 over the term of the agreement. The term of the Development Agreement is to expire on the completion of all research and development activities by CanAm, and the written acknowledgment by CanAm and Medicure International that no further research projects will be undertaken (see Item 6 - Directors, Senior Management and Employees - A. Directors and Senior Management)
The Development Agreement may be terminated under a number of circumstances and, in any event, by Medicure International at any time by providing CanAm with at least 30 days prior written notice of its intention to terminate, or by CanAm at any time by providing Medicure International with at least 90 days prior written notice of its intention to terminate the Development Agreement.
The agreements provide that all confidential information developed or made known during the course of the relationship with the Corporation is to be kept confidential except in specific circumstances.
D. Trend Information
The Corporation is not aware of any trends, uncertainties, demands, commitments or events which are reasonably likely to have a material effect upon the Corporation’s net sales or revenues, income from continuing operations, profitability, liquidity or capital resources, or that would cause reported financial information not necessarily to be indicative of future operating results or financial condition.
ITEM 6. DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES
A. Directors and Senior Management
Directors and Senior Management
30
The members of the board of directors and senior officers of the Corporation including a brief biography of each are as follows:
Dr. Albert D. Friesen - Director, President, Chairman and Chief Executive Officer
The founder of Medicure Inc., Dr. Friesen holds a Ph.D. in protein chemistry from the University of Manitoba. Dr. Friesen played a key role in founding several health industry companies including Rh Pharmaceuticals (acquired by Cangene Inc.), ABI Biotechnology (acquired by Apotex Inc.), Viventia Biotech Inc., Genesys Pharma Inc. and KAM Scientific Inc. Dr. Friesen has experience in the establishment of pharmaceutical production facilities and has also managed and initiated the research and clinical development of several pharmaceutical candidates. Dr. Friesen is a founder of the Industrial Biotechnology Association of Canada (IBAC) and past Chairman of its board of directors and former member of the Industrial Advisory Committee to the Biotechnology Research Institute in Montreal. Dr. Friesen previously served as a senior executive of other publicly-traded companies, including a position as President of Viventia Biotech Inc. (formerly Novopharm Biotech Inc.) Dr. Friesen provides his services to the Corporation through A.D. Friesen Enterprises Ltd., his private consulting corporation. Dr. Friesen devotes substantially all of his time to the Corporation.
Dr. Arnold Naimark - Director
Arnold Naimark, M.D., F.R.C.P. (C), F.R.S.C. has had a distinguished career within medicine and higher education. Dr. Naimark served as Head of the Department of Physiology and later as Dean of the Faculty of Medicine at the University of Manitoba, following which he served as the University’s President and Vice-Chancellor (15 years). Dr. Naimark is currently Director of the University of Manitoba Centre for the Advancement of Medicine, Chair of the Canadian Health Services Research Foundation and of the Canadian Biotechnology Advisory Committee, a Director of Inspiraplex and of the Robarts Research Institute and is a member of the Research Council of the Canadian Institute for Advanced Research. Dr. Naimark has served on many committees and boards, in such positions as a Director of the Canadian Imperial Bank of Commerce, Chair of the International Review Panel for the Medical Research Council of Canada and President of the Association of Universities and Colleges of Canada. Dr. Naimark has also received several honourary degrees and awards, including the Order of Canada.
Dr. William A. Cochrane - Director
Dr. Cochrane is an internationally recognized biotechnology executive and entrepreneur. In addition to other distinctions and senior appointments, Dr. Cochrane has previously served as Chairman and CEO of Connaught Laboratories Inc. and as President and Vice-Chancellor of the University of Calgary. Dr. Cochrane currently works as a health product investment consultant and serves on the boards of StressGen Biotechnologies, and Selective Genetics. Dr. Cochrane has also been the President, CEO, and a Director of Nucleus BioScience Inc. since January 2000.
Mr. G. James Umlah - Director
Mr. Umlah is Chief Investment Officer of the Crocus Investment Fund (a labour sponsored fund) and the President of Crocus Capital (the Crocus Investment Fund’s sales subsidiary). In his capacity with the Crocus Investment Fund, which has current assets of approximately $170 million, Mr. Umlah has been responsible for investing in over 50 Manitoba businesses and has provided corporate finance advice to many of those businesses. In addition, Mr. Umlah has fifteen years experience in the investment industry in Manitoba. He is a former director of a major Canadian securities dealer. Mr. Umlah has an Honours Economics degree from the University of Manitoba. His areas of expertise include retail and institutional securities, corporate planning, and financial restructuring of businesses. Mr. Umlah is responsible for chairing and overseeing the operation of the Crocus Investment Fund’s staff investment committee, which evaluates proposed investments and recommends investments to the board of directors. In addition, Mr. Umlah is responsible for coordinating the functions of the Investment Advisory Committee and sits on the boards of several investee companies. As President of Crocus Capital, Mr. Umlah is responsible for
31
managing the investments of the fund and for securities compliance matters related thereto. Mr. Umlah is also responsible for managing the business and investments of the Manitoba Science & Technology Fund.
Derek Reimer, C.A. – Chief Financial Officer and Secretary
Derek Reimer came to Medicure from Deloitte & Touche LLP where he most recently served as a Senior Manager in the Assurance and Advisory Services group. In this role, Mr. Reimer dealt mainly with major corporate clients, including several TSX 100 companies, providing advice regarding complex accounting, regulatory, and compliance issues. His previous experience includes several years providing international accounting services to clients exclusively in the financial services industry. Mr. Reimer is a Chartered Accountant who also holds a Bachelor of Commerce (Hons.) degree in accounting from the University of Manitoba. Mr. Reimer is responsible for managing financial systems, programs and processes to ensure the successful accomplishment of Medicure’s business objectives.
Business Management
Dr. Albert D. Friesen - Chairman, President, Chief Executive Officer and Director
Dr. Friesen directs the overall business management of the Corporation (see Directors and Senior Management under this item).
Derek G. Reimer, CA Chief Financial Officer and Secretary
Mr. Reimer participates in the Corporation’s financial management and accounting practices (see Directors and Senior Management under this item).
Dawson Reimer - Director of Business Development:Dawson Reimer received his Masters of Applied Environmental Studies from the University of Waterloo. From September 1996 Mr. Reimer served as Director of Business Development/Investor Relations withGenesys Pharma Inc.Mr. Reimer was also project director for the establishment of the company’s new research andGMPpharmaceutical production facility. In October 1997, Mr. Reimer began conducting business activities for GVI, where he has assisted biotechnology projects in developing business plans, obtaining financing and developing intellectual property protection. Mr. Reimer has been actively involved in the Corporation since its inception and provided his services through GVI up until October 1, 2001, at which point he became an employee of the Corporation.
Scientific Management
Dr. Naranjan S. Dhalla - Chief Scientific Officer
Dr. Dhalla participates in the scientific management of the Corporation under the title Chief Scientific Officer. The principal inventor of MC-1, Dr. Dhalla presently serves as Distinguished Professor and Director, Institute of Cardiovascular Sciences, St. Boniface General Hospital Research Centre, Faculty of Medicine, University of Manitoba. Dr. Dhalla is an internationally recognized cardiovascular researcher who has received 59 honours and awards including the Research Achievement Award of the Canadian Cardiovascular Society, The Upjohn Award of the Pharmacological Society of Canada, Honorary Professorship at several universities, the Order of the Buffalo Hunt from the Province of Manitoba, and the Order of Canada. Professionally, Dr. Dhalla has been actively promoting the scientific basis of cardiology for over 25 years.
Dr. Dhalla has served in senior positions such as Secretary General (17 years) and President (3 years) of the International Society for Heart Research, Editor-in-Chief (11 years) of the international journal ‘‘Molecular and Cellular Biochemistry’’ and is currently serving on the editorial boards of 11 other international journals. Since 1996, Dr. Dhalla has served as Executive Director of the International Academy of Cardiovascular Sciences. Dr. Dhalla has published 490 papers and 338 abstracts as well as edited or authored 31 books in the area of heart research.
32
Dr. Karl-Gunnar Hidinger - Vice-President, Clinical Development
Dr. Karl-Gunnar Hidinger received his Ph.D. in Clinical Pharmacology from University of Gothenburg, Sweden. Dr. Hidinger’s drug development experience spans 30 years and includes basic pharmacology research and preclinical research through clinical Phase I, II, III and market approval of new chemical entities. Dr. Hidinger’s career has included clinical research positions in Europe and North America with recognized companies such as Astra Pharma Inc., Glaxo Inc. and Ortho-McNeil Inc. Dr. Hidinger’s experience in clinical drug development complying with international regulatory standards covers a wide variety of therapeutic areas. Most recently, Dr. Hidinger established the corporate infrastructure for the Scientific Affairs Department of the newly established Byk Canada Inc., a subsidiary of Byk Gulden AG, Germany. Dr. Hidinger will be responsible for the design and management of clinical studies for Medicure’s lead compound, MC-1, and for the clinical aspects of other business development and research projects. Since February 1, 2001, Dr. Hidinger has provided his services to the Corporation's research and development activities through a consulting contract with CanAm.
Dr. Serge Rasskazoff , M.D. – Clinical Research Manager
Dr. Rasskazoff obtained his Doctor of Medicine in 1993 from Ukranian State Medical University, Kiev, Ukraine. After additional postdoctoral training, including at the Institute of Neurology, London, UK and Oregon Health Sciences University, Portland, OR, he joined the pharmaceutical industry in 1996. Most recently he worked at Quintiles, an international CRO. Services of Dr. Rasskazoff are provided through a contract with CanAm Bioresearch.
Dr. Wasimul Haque - Director of Chemistry
Dr. Haque received a Ph.D. in Chemistry from the University of Manitoba. Dr. Haque has over fifteen years of experience in industrial biotechnology research and development where he has managed a variety of drug discovery projects. Senior positions held prior to joining Medicure include Project Manager at Alberta Research Council and Senior Scientist at Biomira Inc. Dr. Haque has been issued two patents and is author of several peer-reviewed publications. Dr. Haque began employment with Medicure in July 1998. Since June 1, 2000 Dr. Haque has been employed by CanAm and works on the Corporation’s research and development pursuant to the Development Agreement.
Dr. Jim Diakur - Associate Director of Chemistry
Dr. Diakur is an experienced medicinal chemist who was previously employed as Research Assistant Professor at the Noujaim Institute for Pharmaceutical Research in the Faculty of Pharmacy, University of Alberta. At the institute, Dr. Diakur’s group was focused on the development of carbohydrate-based pharmaceuticals for diabetes, cancer and inflammation and on carbohydrate-based drug carriers for targeted drug delivery. He also has over 10 years of industrial research and management experience in the field of chemistry with Chembiomed Inc. and Biomira Inc. Among other achievements, Dr. Diakur managed a team of scientists responsible for the preclinical development of injectable carbohydrate-based immunotherapeutic agents. Dr. Diakur is employed by CanAm and works on the Corporation’s research and development pursuant to the Development Agreement.
Dr. Deborah Douglas, Ph.D. – Senior Scientist, Cardiovascular Biology
Dr. Douglas received her Ph.D. in Animal Science from McGill University, focusing on molecular biology related research. Her post-doctoral experience involved cell biology research at the Institute of Cell Biology, University of Manitoba, and the Jack Bell Research Centre, Vancouver General Hospital, University of British Columbia. Dr. Douglas developedin vitroandin vivo models to screen for human diseases as Chief of Compound Screening for Alviva Biopharmaceutical Inc. Dr. Douglas has provided her services to Medicure through an employment agreement with CanAm Bioresearch.
33
Dr. Paul Armstrong - Clinical Consultant
Dr. Armstrong, Chair of the Advisory Board, isProfessor in the Department of Medicine, University of Alberta in Edmonton. Dr. Armstrong is an internationally recognized cardiologist and clinical investigator with extensive expertise in the design and conduct of clinical trials focused on acute ischemic syndromes and congestive heart failure. Dr. Armstrong has published widely and served as a senior advisor to major organizations and industry. Since June 1, 2000, Dr. Armstrong provides services to the Corporation’s research and development activities through a consulting contract with CanAm.
Dr. Stephen Hanessian - Chemistry Consultant
Dr. Hanessian received his Ph.D. from Ohio State University in 1960 and has served at the University of Montreal since 1968. He is internationally recognized for his contributions in the field of medicinal chemistry and has been named as principal inventor on over 25 patents. Dr. Hanessian has published 400 papers, trained 250 scientists, and received the 1996 Canada Gold Medal for Science and Engineering, Canada’s highest award for scientific achievement. He has also been made an Officer of the Order of Canada. His special interest in cardiovascular medicinal chemistry is directly aligned with Medicure’s objective of establishing a pipeline of products to address cardiovascular diseases that remain inadequately treated by existing therapeutics. Dr. Hanessian provides services to the Corporation’s research and development activities through a consulting contract with CanAm.
Scientific Advisory Board
The Corporation has established a Scientific Advisory Board to ensure continued and proper review of research activities and work plans. The members of the Scientific Advisory Board and a brief biography of each are as follows:
Dr. Paul Armstrong - Chairperson
Dr. Armstrong, Chair of the Scientific Advisory Board, is Professor in the Department of Medicine, University of Alberta in Edmonton. Dr. Armstrong is an internationally recognized cardiologist and clinical investigator with extensive expertise in the design and conduct of clinical trials focused on acute ischemic syndromes and congestive heart failure. Dr. Armstrong has published widely and served as a senior advisor to major organizations and industry.
Dr. Raymond Gibbons
Dr. Gibbons is a Professor of Medicine at the Mayo Medical School in Rochester, Minnesota. Dr. Gibbons has extensive experience with scar size measurement and myocardial infarction.
Dr. Stephen Hanessian
Dr. Hanessian is Professor, Department of Chemistry, University of Montreal. Dr. Hanessian is one of North America’s most renowned medicinal chemists with considerable experience in industry collaboration for the discovery of new pharmaceuticals.
Dr. Morris Karmazyn
Dr. Karmazyn is a Professor in the Department of Pharmacology and Toxicology at the University of Western Ontario in London, Ontario. Dr. Karmazyn is internationally recognized and has received numerous distinctions for his research in the field of myocardial ischemia and ischemic reperfusion injury.
34
Dr. John McNeill
Dr. McNeill is Professor and Dean Emeritus, Division of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences at the University of British Columbia in Vancouver. The recipient of numerous awards and distinctions, Dr. McNeill’s research is centered on the biochemical mechanism action of drugs on the heart.
Dr. Eldon Smith
Dr. Smith is a Professor at the University of Calgary Medical School in Alberta, where his past positions include Dean of the Faculty of Medicine, Head of the Department of Medicine and Head of the Division of Cardiology. A distinguished clinician and research scientist, Dr. Smith has considerable industry experience having served on the boards of several well-recognized companies.
Dr. Pierre Theroux
Dr. Theroux is Professor of Medicine at the University of Montreal and Chief of the Coronary Care Unit at the Montreal Heart Institute. Dr. Theroux’s innovative work is widely recognized and he has contributed extensively to the development of new treatments for acute ischemic heart disease.
Dr. Jeffrey Weitz
Dr. Weitz is Professor of Medicine and Haematology at McMaster University in Hamilton where he has contributed extensively to understanding the role of thrombosis and its treatment in cardiovascular disease. Dr. Weitz also brings a wealth of expertise in academic-industrial collaboration and development of new products
B. Compensation
No compensation of any kind was paid to the directors and officers of the Corporation during the year ended May 31, 2002, except as follows:
During the period beginning June 1, 2001 to September 30, 2001, the Corporation paid Genesys Venture Inc., a biotech incubator company co-owned by Dr. Albert D. Friesen, $84,000 to provide the Corporation with overall management services including those of Dr. Friesen as President and Chief Executive Officer of the Corporation. Genesys Venture Inc, in turn, paid Dr. Friesen $32,000 of the $84,000 total, in salary compensation. On October 1, 2001, the agreement with Genesys Venture Inc. was modified such that Dr, Friesen no longer received a salary as part of the overall management services agreement. During the eight month period from June 1, 2001 to January 31, 2002, the Corporation paid Genesys Venture Inc. a total of $120,000 (Year ended May 31, 2001: $242,000; Nine months ended May 31, 2000: $155,640) for consulting fees and office rent and supplies. As of February 1, 2002 the agreement with Genesys Venture Inc. was terminated. During the nine months ended May 31, 2000, stock options were granted to Dr. Friesen to purchase 30,000 shares of the Corporation at a price of $2.15 per share expiring on February 22, 2005.
On October 1, 2001 a compensation agreement was entered into between the Corporation and A.D. Friesen Enterprises Ltd., a corporation owned by Dr. Friesen. This agreement, which was subsequently amended on February 1, 2002, provides Dr. Albert D. Friesen with an annual consulting fee of $150,000 . For the period commencing October 1, 2001 to May 31, 2002, the Corporation paid A.D. Friesen Enterprises Ltd., $82,000 in consulting compensation.
During the year ended May 31, 2002, the Corporation paid CanAm, a private company wholly owned by Peter de Visser, a former director of the Corporation, a total of $3,021,115 (Fiscal year ended May 31, 2001: $2,364,440; Nine months ended May 31, 2000: NIL) for all research expenditures incurred by CanAm. During the nine months ended May 31, 2000, stock options were granted to Peter J. de Visser to
35
purchase 30,000 shares of the Corporation at a price of $2.15 per share expiring on February 22, 2005. Peter de Visser resigned as a director of the Corporation on December 20, 2001.
During the year ended May 31, 2002, the Corporation paid directors a total of Nil (Year ended May 31, 2001: $35,000; Nine months ended May 31, 2000: $22,500) for consulting fees.
Additionally, the Corporation provides its directors $1,500 for each quarterly meeting they personally attend, and $750 for each quarterly meeting they are present via telephone. The Corporation does not provide any cash compensation for its directors who are also officers of the Corporation for their services as directors.
No pension, retirement fund and other similar benefits have been set aside for the officers and directors of the Corporation.
C. Board Practices
The Board of Directors presently consists of four directors. James Umlah was appointed to the Board on September 17, 2001. Peter de Visser resigned from the Board on December 20, 2001. All other directors were either elected or re-elected at the Corporation’s annual general meeting of the shareholders held on November 5, 2001. Each director holds office until the next annual general meeting of the Corporation or until his successor is elected or appointed, unless his office is earlier vacated in accordance with the Articles of the Corporation, or with the provisions of theCanada Business Corporations Act. Dr. Albert D. Friesen has served as a director of the Corporation since September 1997. Dr. Arnold Naimark has served as a director of the Corporation since March 2000 while Dr. William A. Cochran has served as a director of the Corporation since October 2000.
Pursuant to Section 171 of the Canada Business Corporations Act (the Act), the Corporation is required to have an Audit Committee. As at the date hereof, the members of the Audit Committee are William Cochrane, Arnold Naimark and James Umlah. Section 171(1) of the Act requires the directors of a reporting corporation to elect from among their number a committee composed of not fewer than three directors, of whom a majority must not be officers or employees of the corporation or an affiliate of the corporation. Section 171(3) provides that, before financial statements are approved by the directors, they must be submitted to the audit committee for review. Section 171(4) provides that the auditor must be given notice of, and has the right to appear before and to be heard at, every meeting of the audit committee, and must appear before the audit committee when requested to do so by the committee. Finally, section 171(5) provides that on the request of the auditor, the audit committee must convene a meeting of the audit committee to consider any matters the auditor believes should be brought to the attention of the directors or members.
The Corporation also has an Executive Compensation and Corporate Governance Committee. Neither the Corporation nor its subsidiaries have entered into contracts providing for benefits to the directors upon termination of employment.
D. Employees
In addition to the individuals disclosed in A. Directors and Senior Management of this item, CanAm has a staff of twenty-one research scientists, technicians and staff dedicated solely to the Corporation’s research and development activities.
E. Share Ownership
With respect to the persons referred to above in B. Compensation of this item, the following table discloses the number of shares (each share possessing identical voting rights), stock options held and percent of the shares outstanding held by those persons, as of May 31, 2002.
36
| Name and Title | No. of Shares | Percent of Shares Outstanding (Greater than 10% ownership) |
| Dr. Albert D. Friesen Chairman, President, CEO and Director | 6,027,915(1) | 15.7% |
Note:
(1) Dr. Albert Friesen holds 305,500 shares personally. The rest of the shares are held by ADF Family Holding Corp., a private company wholly-owned by Dr. Albert D. Friesen, his wife Mrs. Leona M. Friesen, and Bert & Lee Family Trust (the trustees of which are Dr. and Mrs. Friesen).
Incentive Stock Options
The following table discloses the stock options beneficially held by the aforementioned persons, as of August 31, 2002. The stock options are for shares of Common Stock of the Corporation.
| Name of Person | Number of Shares Subject to Issuance | Exercise Price per Share | Expiry Date |
| Dr. Albert D. Friesen | 30,000 | $2.15 | February 22, 2005 |
| Dr. Naranjan S. Dhalla | 25,000 | $0.75 | January 5, 2005 |
| Derek G. Reimer | 90,000 | $0.80 | February 4, 2007 |
| Dr. William Cochrane | 125,000 | $0.75 | January 5, 2005 |
| Dr. Arnold Naimark | 100,000 | $0.75 | January 5, 2005 |
The Corporation has established an Incentive Stock Option Plan (the ‘‘Plan’’) for its directors, key officers, employees and consultants. Options granted pursuant to the Plan will not exceed a term of five years and are granted at an option price and on other terms which the directors determine is necessary to achieve the goal of the Plan and in accordance with regulatory requirements, including those of the TSX. Each option entitles the holder thereof to purchase one (1) Common Share of the Corporation on the terms set forth in the Plan and in such purchaser’s specific stock option agreement. The option price may be at a discount to market price, which discount will not, in any event, exceed that permitted by any stock exchange on which the Corporation’s Common Shares are listed for trading.
The number of Common Shares allocated to the Plan, the exercise period for the options (not to exceed five years), and the vesting provisions for the options will be determined by the board of directors of the Corporation from time to time. The aggregate number of shares reserved for issuance under the Plan, together with any other employee stock option plans, options for services and employee stock purchase plans, will not exceed 3,700,000 of the issued and outstanding Common Shares. In addition, the aggregate number of shares reserved for issuance to any one person shall not exceed five percent (5%) of the issued and outstanding Common Shares.
The Common Shares issued pursuant to the exercise of options, when fully paid for by a participant, are not included in the calculation of Common Shares allocated to or within the Plan. Should a participant cease to be eligible due to the loss of corporate office (being that of an officer or director) or employment, the option shall cease for varying periods not exceeding 90 days. Loss of eligibility for consultants is regulated by specific rules imposed by the directors when the option is granted to the appropriate consultant. The Plan also provides that estates of deceased participants can exercise their options for a period not exceeding one year following death.
37
ITEM 7. MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A. Major Shareholders
As of August 31, 2002, the following table sets forth the beneficial ownership of the Corporation's common shares by each person known by the Corporation to own beneficially more than 5% of the issued and outstanding common shares of the Corporation:
| Title of Class | Identity of Person or Group | Amount Owned | Percentage of Class |
| Common shares | CDS & Company(1) | 14,040,111(1) | 37.7% |
| Toronto, Ontario | |||
| Common shares | Dr. Albert D. Friesen(3) | 6,171,915 | 16.6% |
| Winnipeg, Manitoba | |||
| Common shares | Dr. Naranjan S. Dhalla | 1,900,745 | 5.1% |
| Winnipeg, Manitoba |
Notes:
(1)Brokerage clearing house; the ultimate beneficial owners of these shares are not known to the Corporation.
(2)Amount of shares as of August 31, 2002.
(3) Dr. Albert Friesen holds 431,500 shares personally. The rest of the shares are held by ADF Family Holding Corp., a private company wholly-owned by Dr. Albert D. Friesen, his wife Mrs. Leona M. Friesen, and Bert & Lee Family Trust (the trustees of which are Dr. and Mrs. Friesen).
The total number of common shares of the Corporation referred to in this item includes the 1,280,000 Class A common shares that are listed on the CDNX under the symbol “MPH.A”.
As of August 31, 2002, there were 3 shareholders of record in the United States holding a total of 2,375 shares of the Corporation.
To the best of the Corporation's knowledge, it is not owned or controlled, directly or indirectly, by another company, by any foreign government or by any other natural or legal person severally or jointly.
As of August 31, 2002, the total number of issued and outstanding common shares of the Corporation beneficially owned by the directors and officers of the Corporation as a group was 6,201,915 (or 16.7% of common shares).
To the best of the Corporation's knowledge, there are no arrangements, the operation of which at a subsequent date will result in a change in control of the Corporation.
B. Related Party Transactions
Other than as set forth below, management of the Corporation is not aware of any material interest, direct or indirect, of any director or officer of the Corporation, any person beneficially owning, directly or indirectly, more than 10% of the Corporation’s voting securities, or any associate or affiliate of any such person in any transaction within the last three years or in any proposed transaction which in either case has materially affected or will materially affect the Corporation or its subsidiaries.
On November 22, 1999 Medicure was acquired by Lariat by way of a reverse takeover as Lariat’s “Major Transaction” as a Junior Capital Pool company within the meaning of the Alberta Securities Commission Rule 46-501, the Alberta Securities Commission Companion Policy 46-501CP and The Alberta Stock Exchange Circular 7. Pursuant to the terms of the Major Transaction, Lariat acquired all of the issued and outstanding shares of Medicure in exchange for 9,500,000 shares of Lariat, at a deemed price for securities regulatory purposes of $0.20 per share for aggregate consideration of $1,900,000. The Major
38
Transaction was negotiated entirely at arm’s length. As a result of the share exchange, control of Lariat passed to the former shareholders of Medicure. This type of transaction is commonly referred to as a “reverse takeover”. Under reverse takeover accounting, for financial reporting purposes, the Corporation is considered to be a continuation of the operations formerly carried on by Medicure.
During the period beginning June 1, 2001 to September 30, 2001, the Corporation paid Genesys Venture Inc., a biotech incubator company co-owned by Dr. Albert D. Friesen, $84,000 to provide the Corporation with overall management services including those of Dr. Friesen as President and Chief Executive Officer of the Corporation. Genesys Venture Inc, in turn, paid Dr. Friesen $32,000 of the $84,000 total, in salary compensation. On October 1, 2001, the agreement with Genesys Venture Inc. was modified such that Dr, Friesen no longer received a salary as part of the overall management services agreement. During the eight month period from June 1, 2001 to January 31, 2002, the Corporation paid Genesys Venture Inc. a total of $120,000 (Year ended May 31, 2001: $242,000; Nine months ended May 31, 2000: $155,640) for consulting fees and office rent and supplies. As of February 1, 2002 the agreement with Genesys Venture Inc. was terminated.
On October 1, 2001 a compensation agreement was entered into between the Corporation and A.D. Friesen Enterprises Ltd., a corporation owned by Dr. Friesen. This agreement, which was subsequently amended on February 1, 2002, provides Dr. Albert D. Friesen with an annual consulting fee of $150,000. For the period commencing October 1, 2001 to May 31, 2002, the Corporation paid A.D. Friesen Enterprises Ltd., $82,000 in consulting compensation.
Dr. Friesen, a director, the Chairman, the President and the Chief Executive Officer of the Corporation also owns a leasing company, Waverley Business and Science Centre Inc. which entered into a lease with the Corporation as of March 1, 2002. Pursuant to this agreement, the Corporation leases approximately 2,100 square feet of office space from Waverley Business and Science Centre Inc. for minimum annual rental payments of $23,507. The agreement is for a five year term.
Dr. Naranjan Dhalla, the Chief Scientific Officer of the Corporation, is the principal scientist responsible for discovering the cardiovascular benefits of MC-1. He is also a significant shareholder of the Corporation. As an employee of the University of Manitoba he will receive 25% of any royalties the university may receive in respect to the License Agreement. In addition, Dr. Dhalla entered into a consulting agreement with the Corporation effective January 18, 1998 wherein Dr. Dhalla agreed to perform certain consulting services to the Corporation and which contract remains in effect as at the date hereof. The Corporation is currently paying Dr. Dhalla $40,000 per annum for these services.
On June 1, 2000 the Corporation licensed the world-wide development and marketing rights (except for Canada) for MC-1, the Corporation’s lead product, to the Corporation’s wholly owned subsidiary, Medicure International. Medicure International then entered into a Development Agreement with CanAm to perform research and development on MC-1 and other compounds at cost, plus a reasonable mark-up not to exceed ten percent of any amount invoiced. The parties to the Development Agreement agreed that the aggregate amount of all invoiced expenditures shall not exceed $10,000,000 over the term of the agreement. The term of the agreement is from June 1, 2000 until the completion of all research and development activities by CanAm that are contemplated in the agreement. CanAm is a private Canadian company wholly owned by Peter de Visser, a former director of the Corporation
C. Interests of Experts and Counsel
Not applicable
39
ITEM 8. FINANCIAL INFORMATION
A. Consolidated Statements or Other Financial Information
Financial Statements
Attached hereto are the consolidated financial statements of the Corporation for the years ended May 31, 2002 and 2001 and for the nine month period ended May 31, 2000, audited by KPMG LLP, Chartered Accountants. The consolidated financial statements including related notes are accompanied by auditors’ reports.
Legal Proceedings
There are no legal or arbitration proceedings, including those relating to bankruptcy, receivership or similar proceedings and those involving any third party, which may have, or have had in the recent past, significant effects on the Corporation’s financial position or profitability. There are no legal proceedings to which the Corporation is a party, nor to the best of the knowledge of the Corporation’s management are any legal proceedings contemplated.
Dividend Policy
The Corporation has not paid dividends in the past and it has no present intention of paying dividends on its shares as it anticipates that all available funds will be invested to finance the growth of its business. The directors of the Corporation will determine if and when dividends should be declared and paid in the future based upon the Corporation’s financial position at the relevant time. All of the Corporation’s Shares are entitled to an equal share of any dividends declared and paid.
B. Significant Changes
Since May 31, 2002, the date of the most recent financial statements, no significant changes have occurred with the exception of the following:
a) Subsequent to May 31, 2002, 126,000 stock options were exercised and converted into common shares at $0.20 per share.
40
ITEM 9. THE OFFERING AND LISTING
A. Offer and Listing Details
There is no offer associated with this registration statement.
The Corporation's common shares are listed and traded on The Toronto Stock Exchange under the symbol “MPH”. The historical trading data for the common shares of the Corporation on the above-mentioned exchanges is set out below.
| Fiscal Period/Year Ended | High ($) | Low ($) | |||
| May 31, 2002 | 1.24 | 0.40 | |||
| May 31, 2001 | 3.45 | 0.95 | |||
| May 31, 2000 | 8.00 | 0.48 | |||
| August 31, 1999(1) | 0.59 | 0.25 | |||
| August 31, 1998 | N/A | N/A | |||
| Fiscal Quarter Ended | |||||
| August 31, 2002 | 0.52 | 0.215 | |||
| May 31, 2002 | 0.60 | 0.40 | |||
| February 28, 2002 | 0.74 | 0.52 | |||
| November 30, 2001 | 0.85 | 0.55 | |||
| August 31, 2001 | 1.24 | 0.70 | |||
| May 31, 2001 | 1.70 | 0.95 | |||
| February 28, 2001 | 2.89 | 1.65 | |||
| November 30, 2000 | 3.45 | 1.80 | |||
| August 31, 2000 | 3.60 | 1.80 | |||
| Month | |||||
| October 2002 | 0.30 | 0.22 | |||
| September 2002 | 0.30 | 0.215 | |||
| August 2002 | 0.36 | 0.23 | |||
| July 2002 | 0.40 | 0.25 | |||
| June 2002 | 0.52 | 0.39 | |||
| May 2002 | 0.49 | 0.40 |
Note:
(1) For the period from initial listing on March 30, 1999 to August 31, 1999
C.Markets
The Corporation's common shares were listed and traded on The Alberta Stock Exchange from March 30, 1999 to November 28, 1999 and on The Canadian Venture Exchange under the symbol “MPH”, from November 29, 1999 to March 14, 2002. The common shares and Class A common shares commenced trading on the Toronto Stock Exchange on March 15, 2002 to the present. The Corporation’s shares are not currently trading on any United States stock exchange or the over-the-counter market, and, accordingly, there is currently no public market for the common stock of the Corporation in the United States.
41
ITEM 10. ADDITIONAL INFORMATION
A. Share Capital
Not applicable
B. Memorandum and Articles of Association
1. Objects and Purposes of the Corporation
The Memorandum of the Corporation places no restrictions upon the Corporation’s objects and purposes.
2. Directors
Under applicable Canadian law, the directors and officers of the Corporation, in exercising their powers and discharging their duties, must act honestly and in good faith with a view to the best interests of the Corporation. The directors and officers must also exercise the care, diligence and skill that a reasonably prudent person would exercise in comparable circumstances.
Section 4.14 of By-Law No.1 of the Corporation (the “By-Law”) provides that a director shall not be disqualified by reason of his office from contracting with the Corporation or a subsidiary thereof. Subject to the provisions of theCanada Business Corporations Act(the “Act”), a director shall not by reason only of his office be accountable to the Corporation or its shareholders for any profit or gain realized from a contract or transaction in which he has an interest. Such contract or transaction shall not be voidable by reason only of such interest, or by reason only of the presence of a director so interested at a meeting, or by reason only of his presence being counted in determining a quorum at a meeting of the directors at which such a contract or transaction is approved, provided that a declaration and disclosure of such interest shall have been made at the time and in the manner prescribed by section 120 of the Act, and the director so interested shall have refrained from voting as a director on the resolution approving the contract or transaction (except as permitted by the Act) and such contract shall have been reasonable and fair to the Corporation and shall have been approved by the directors or shareholders of the Corporation as required by section 120 of the Act.
Section 4.01 of the By-Law states that the exact number of directors to form the board shall be determined from time to time by the directors of the Corporation entitled to vote at regular meetings. A quorum of the board shall be a majority of the board. No business shall be transacted at a meeting unless a quorum is present.
Section 3.01 of the By-Law states that the board may, without the authorization of the shareholders:
| i) | borrow money upon the credit of the Corporation; |
| ii) | issue, reissue, sell or pledge debt obligations of the Corporation, including bonds, debentures, notes or other evidences of indebtedness or guarantees, whether secured or unsecured; |
| iii) | subject to section 44 of the Act, give a guarantee on behalf of the Corporation to secure performance of an obligation of any person; and |
| iv) | mortgage, hypothecate, pledge or otherwise create a security interest in all or any property of the Corporation, owned or subsequently acquired, to secure any obligation of the Corporation. |
The borrowing powers of the directors can be varied by amending the By-Law of the Corporation.
There is no provision in the By-Law imposing a requirement for retirement or non-retirement of directors under an age limit requirement.
Section 4.02 states that a director need not be a shareholder to be qualified as a director.
42
3. Shares
The Articles of the Corporation provide that the Corporation is authorized to issue an unlimited number of shares designated as Common Shares, Class A Common Shares and Preferred Shares. Except for meetings at which only holders of another specified class or series of shares of the Corporation are entitled to vote separately as a class or series, each holder of the Common and Class A shares is entitled to receive notice of, to attend and to vote at all meetings of the shareholders of the Corporation. Subject to the rights, privileges, restrictions and conditions attached to any other class of shares of the Corporation, the holders of the Common and Class A shares are also entitled to receive dividends if, as and when declared by the directors of the Corporation and are entitled to share equally in the remaining property of the Corporation upon liquidation, dissolution or winding-up of the Corporation.
The Preferred Shares shall have attached to them, as a class, the rights, privileges, restrictions and conditions as hereinafter set forth.
The Preferred Shares may from time to time be issued in one or more series and, subject to the following provisions, and subject to the sending of articles of amendment in respect thereof, the directors may fix from time to time and before issue a series of Preferred Shares, the number of shares which are to comprise that series and the designation, rights, privileges, restrictions and conditions to be attached to that series of Preferred Shares including, without limiting the generality of the foregoing, the rate or amount of dividends or the method of calculating dividends, the dates of payment of dividends, the redemption, purchase and/or conversion, and any sinking fund or other provisions.
The Preferred Shares of each series shall, with respect to the payment of dividends and the distribution of assets or return of capital in the event of liquidation, dissolution or winding-up of the Corporation, whether voluntary or involuntary, or any other return of capital or distribution of the assets of the Corporation among its shareholders for the purpose of winding-up its affairs, rank on a parity with the Preferred Shares of every other series and be entitled to preference over the Common and Class A Common Shares and over any other shares of the Corporation ranking junior to the Preferred Shares. The Preferred Shares of any series may also be given other preferences, not inconsistent with these articles, over the Common Shares and Class A Common Shares and any other shares of the Corporation ranking junior to the Preferred Shares of a series as may be fixed in accordance with clause (i).
If any cumulative dividends or amounts payable on the return of capital in respect of a series of Preferred Shares are not paid in full, all series of Preferred Shares shall participate rateably in respect of accumulated dividends and return of capital.
Unless the directors otherwise determine in the articles of amendment designating a series of Preferred Shares, the holder of each share or a series of Preferred Shares shall not, as such, be entitled to receive notice of or vote at any meeting of shareholders, except as otherwise specifically provided in the Act.
4. Rights of Shareholders
Under the Act, shareholders of the Corporation are entitled to examine, during its usual business hours, the Corporation’s articles and by-laws, notices of directors and change of directors, any unanimous shareholder agreements, the minutes of meetings and resolutions of shareholders and the list of shareholders.
Shareholders of the Corporation may obtain a list of shareholders upon payment of a reasonable fee and sending an affidavit to the Corporation or its transfer agent stating, among other things, that the list of shareholders will not be used by any person except in connection with an effort to influence the voting of shareholders of the Corporation, an offer to acquire shares of the Corporation or any other matter relating to the affairs of the Corporation.
Under the Act, shareholders of the Corporation may apply to a court having jurisdiction directing an investigation to be made of the Corporation. If it appears to the court that the formation, business or
43
affairs of the Corporation were conducted for fraudulent or unlawful purposes, or that the powers of the directors were exercised in a manner that is oppressive or unfairly disregards the interests of the shareholders, the court may order an investigation to be made of the Corporation.
To change the rights of holders of stock, where such rights are attached to an issued class or series of shares, requires the consent by a separate resolution of the holders of the class or series of shares, as the case may be, requiring a majority of 75% of the votes cast.
The Corporation is organized under the laws of Canada. The Corporation’s directors, officers, and affiliates of the Corporation, as well as the experts named in this registration statement, are residents of Canada and, to the best of the Corporation’s knowledge, all or a substantial portion of their assets and all of the Corporation’s assets are located outside of the United States. As a result, it may be difficult for shareholders of the Corporation in the United States to effect service of process on the Corporation or these persons above within the United States, or to realize in the United States upon judgments rendered against the Corporation or such persons. Additionally, a shareholder of the Corporation should not assume that the courts of Canada (i) would enforce judgments of U.S. courts obtained in actions against the Corporation or such persons predicated upon the civil liability provisions of the U.S. federal securities laws or other laws of the United States, or (ii) would enforce, in original actions, liabilities against the Corporation or such persons predicated upon the U.S. federal securities laws or other laws of the United States.
Laws in the United States and judgments of U.S. courts would generally be enforced by a court of Canada unless such laws or judgments are contrary to public policy in Canada, are or arise from foreign penal laws or laws that deal with taxation or the taking of property by a foreign government and are not in compliance with applicable laws in Canada regarding the limitation of actions. Further, a judgment obtained in a U.S. court would generally be recognized by a court of Canada, except under the following examples:
| i) | the judgment was rendered in a U.S. court that had no jurisdiction according to applicable laws in Canada; |
| ii) | the judgment was subject to ordinary remedy (appeal, judicial review and any other judicial proceeding which renders the judgment not final, conclusive or enforceable under the laws of the applicable state) or not final, conclusive or enforceable under the laws of the applicable state; |
| iii) | the judgment was obtained by fraud or in any manner contrary to natural justice or rendered in contravention of fundamental principles of procedure; and |
| iv) | a dispute between the same parties, based on the same subject matter has given rise to a judgment rendered in a court of Canada or has been decided in a third country and the judgment meets the necessary conditions for recognition in a court of Canada. |
5. Meetings
Subject to the provisions of the Act, the annual general meeting of the shareholders shall be on such date in each year as the board of directors may determine, and a special meeting of the shareholders may be convened at any time by order of the President or by the board on their own motion or on the requisition of shareholders as provided for in the Act. Notice of the time and place of each meeting of shareholders shall be given not less than 21 days nor more than 50 days before the date of the meeting to each director and shareholder. A meeting of shareholders may be held without notice at any time and at any place provided a waiver of notice is obtained in accordance with section 136 of the Act. The quorum for the transaction of business at meetings of the shareholders shall consist of not less than one (1) shareholder present or represented by proxy and holding in all not less than five (5%) percent of the issued capital of the Corporation carrying voting rights. At any meeting of shareholders, every person shall be entitled to vote who, at the time of the taking of a vote (or, if there is a record date for voting, at the close of business on such record date) is entered in the register of shareholders as the holder of one or more shares carrying the right to vote at such meeting, subject to the provisions of the Act.
44
6. Ownership of Securities
There are no limitations on the right to own securities, imposed by foreign law or by the By-Law or other constituent document of the Corporation.
7. Change in Control of Corporation
No provision of the Corporation’s articles of association, charter or By-Law would have the effect of delaying, deferring, or preventing a change in control of the Corporation, and operate only with respect to a merger, acquisition or corporate restructuring of the Corporation or any of its subsidiaries.
8. Ownership Threshold
There are no By-Law provisions governing the ownership threshold above which shareholder ownership must be disclosed.
C. Material Contracts
The following are the material contracts of the Corporation, other than those mentioned elsewhere in this Form, to which the Corporation or any member of the group is a party, for the two years immediately preceding publication of this registration statement.
| a) | Investor Relations Contract dated March 28, 2002 between Boxe Studio Intl. and the Corporation whereby investor relations services will be provided to the Corporation including identifying possible sources of capital, providing analysis and advice with respect to capital structure, advising and assisting the Corporation with respect to its relationship with existing shareholders and assisting in the preparation of public information materials. |
| b) | Development Agreement between Medicure International Inc. and CanAm Bioresearch Inc. dated June 1, 2000, wherein CanAm Bioresearch Inc. agreed to conduct research and development activities for Medicure International; |
D. Exchange Controls
There is no law or government decree of regulation in Canada that restricts the export or import of capital, or that affects the remittance of dividends, interest or other payments to a non-resident holder of Common Shares, other than withholding tax requirements. See "Item 7 Taxation."
There is no limitation imposed by Canadian law or by the articles or other charter documents of the Corporation on the right of a non-resident to hold or vote the Common Shares or the Class A common shares of the Corporation, other than as provided in the Investment Canada Act, as amended (the "Investment Act").
The Investment Act generally prohibits implementation of a reviewable investment by an individual, government or agency thereof, corporation, partnership, trust or joint venture that is a "non-Canadian" as defined in the Investment Act (a "non-Canadian"), unless, after review the Minister responsible for the Investment Act is satisfied that the investment is likely to be of net benefit to Canada. If an investment by a non-Canadian is not a reviewable investment, it nevertheless requires the filing of a short notice which may be given at any time up to 30 days after the implementation of the investment.
An investment in Common Shares of the Corporation by a non-Canadian that is a "WTO investor" (an individual or other entity that is a national of, or has the right of permanent residence in, a member of the World Trade Organization, current members of which include the European Community, Germany, Japan, Mexico, the United Kingdom and the United States, or a WTO investor-controlled entity, as defined in the Investment Act) would be reviewable under the Investment Act if it were an investment to acquire direct control, through a purchase of assets or voting interests, of the Corporation and the value of the assets of the Corporation equalled or exceeded $184 million, the threshold established for 1999, as
45
indicated on the financial statements of the Corporation for its fiscal year immediately preceding the implementation of the investment. In subsequent years, such threshold amount may be increased or decreased in accordance with the provisions of the Investment Act.
An investment in Common Shares of the Corporation by a non-Canadian, other than a WTO investor, would be reviewable under the Investment Act if it were an investment to acquire direct control of the Corporation and the value of the assets were $5.0 million or more, as indicated on the financial statements of the Corporation for its fiscal year immediately preceding the implementation of the investment.
A non-Canadian, whether a WTO investor or otherwise, would acquire control of the Corporation for the purposes of the Investment Act if he, she or it acquired a majority of the Common Shares of the Corporation or acquired all or substantially all of the assets used in conjunction with the Corporation 's business. The acquisition of less than a majority, but one-third or more of the Common Shares of the Corporation, would be presumed to be an acquisition of control of the Corporation unless it could be established that the Corporation was not controlled in fact by the acquirer through the ownership of the Common Shares.
The Investment Act would not apply to certain transactions in relation to Common Shares of the Corporation, including:
| (a) | an acquisition of Common Shares of the Corporation by any person if the acquisition were made in the ordinary course of that person's business as a trader or dealer in securities; | |
| (b) | an acquisition of control of the Corporation in connection with the realization of security granted for a loan or other financial assistance and not for any purpose related to the provisions of the Investment Act; and | |
| (c) | an acquisition of control of the Corporation by reason of an amalgamation, merger, consolidation or corporate reorganization following which the ultimate direct or indirect control in fact of the Corporation, through the ownership of voting interests, remains unchanged. |
E. Taxation
The following is a summary of the principal Canadian federal income tax considerations, as of the date hereof, generally applicable to Security holders who deal at arm's length with the Corporation, who, for purposes of the Income Tax Act (Canada) (the "Canadian Tax Act") and any applicable tax treaty or convention, have not been and will not be resident or deemed to be resident in Canada at any time while they have held shares of the Corporation, to whom such shares are capital property, and to whom such shares are not "taxable Canadian property" (as defined in the Canadian Tax Act). This summary does not apply to a non-resident insurer.
Generally, shares of the Corporation will be considered to be capital property to a holder thereof provided that the holder does not use such shares in the course of carrying on a business or has not acquired them in one or more transactions considered to be an adventure in the nature of trade. All security holders should consult their own tax advisors as to whether, as a matter of fact, they hold shares of the Corporation as capital property for the purposes of the Canadian Tax Act.
Under the current provisions of the Canadian Tax Act, as modified by the Proposed Amendments (see below), one-half of capital gains (‘‘taxable capital gains’’) must be included in computing the income of a holder in the year of disposition. One-half of capital losses (‘‘allowable capital losses’’) may generally be deducted against taxable capital gains for the year of disposition subject to and in accordance with the provisions of the Canadian Tax Act.
46
Allowable capital losses in excess of a holder’s taxable capital gains of a taxation year may generally be carried back three years and carried forward indefinitely for deduction against taxable capital gains realized in those years, to the extent and under circumstances permitted under the Canadian Tax Act.
This discussion is based on the current provisions of the Canadian Tax Act and the regulations thereunder, the current provisions of the Canada-United States Income Tax Convention (the "Tax Treaty") and current published administrative practices of the Canada Customs and Revenue Agency. This discussion takes into account specific proposals to amend the Canadian Tax Act and the regulations thereunder publicly announced by or on behalf of the Minister of Finance (Canada) prior to the date hereof (the "Proposed Amendments") and assumes that all such Proposed Amendments will be enacted in their present form. No assurances can be given that the Proposed Amendments will be enacted in the form proposed, if at all; however the Canadian federal income tax considerations generally applicable to security holders described herein will not be different in a material adverse way if the Proposed Amendments are not enacted.
Except for the foregoing, this discussion does not take into account or anticipate any changes in law, whether by legislative, administrative or judicial decision or action, nor does it take into account provincial, territorial or foreign income tax legislation or considerations, which may differ from the Canadian federal income tax considerations described herein.
Generally, shares of the Corporation will not be taxable Canadian property at a particular time provided that such shares are listed on a prescribed stock exchange (which exchanges currently include the Toronto Stock Exchange), the holder does not use or hold, and is not deemed to use or hold, the shares of the Corporation in connection with carrying on a business in Canada and the holder, persons with whom such holder does not deal at arm's length, or the holder and such persons, have not owned (or had under option) 25% or more of the issued shares of any class or series of the capital stock of the Corporation at any time within five years preceding the particular time.
A holder of shares of the Corporation that are not taxable Canadian property will not be subject to tax under the Canadian Tax Act on the sale or other disposition of shares.
Dividends paid or deemed to be paid on the shares of the Corporation are subject to non-resident withholding tax under the Canadian Tax Act at the rate of 25%, although such rate may be reduced under the provisions of an applicable income tax treaty or convention. For example, under the Tax Treaty, the rate is reduced to 5% in respect of dividends paid to a company that is the beneficial owner thereof, that is resident in the United States for purposes of the Tax Treaty and that owns at least 10% of the voting stock of the Corporation. In all other cases, the rate is reduced to 15% in respect of dividends paid to the beneficial owner thereof that is resident in the United States for purposes of the Tax Treaty.
WHILE INTENDED TO ADDRESS ALL MATERIAL CANADIAN FEDERAL INCOME TAX CONSIDERATIONS, THIS SUMMARY IS FOR GENERAL INFORMATION PURPOSES ONLY, AND IS NOT INTENDED TO BE, NOR SHOULD IT BE CONSTRUED TO BE, LEGAL OR TAX ADVICE TO ANY HOLDER OR PROSPECTIVE HOLDER OF COMMON SHARES. NO OPINION WAS REQUESTED BY THE CORPORATION, OR IS PROVIDED BY ITS LEGAL COUNSEL AND/OR AUDITORS. ADDITIONALLY, THIS SUMMARY DOES NOT CONSIDER THE EFFECTS OF UNITED STATES FEDERAL, STATE, LOCAL OR FOREIGN INCOME TAX CONSEQUENCES.
ACCORDINGLY, HOLDERS AND PROSPECTIVE HOLDERS OF COMMON SHARES SHOULD CONSULT THEIR OWN TAX ADVISORS ABOUT THE CONSEQUENCES OF PURCHASING, OWNING, AND DISPOSING OF COMMON SHARES OF THE CORPORATION.
F. Dividends and Paying Agents
Not applicable
G. Statement by Experts
Not applicable
47
H. Documents on Display
The documents described herein may be inspected at the head office of Corporation at 4 – 1200 Waverley Street, Winnipeg, Manitoba, Canada R3T 0P4, during normal business hours.
ITEM 11. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
INTEREST RATE RISK
The primary objective of the Corporation’s investment activities is to preserve principal by maximizing the income the Corporation receives from such activities without significantly increasing risk. Securities that the Corporation invests in are generally highly liquid short-term investments such as term deposits with terms to maturity of less than one year.
Due to the short-term nature of these investments, the Corporation believes there is no material exposure to interest rate risk arising from such investments and accordingly, no quantitative tabular disclosure is required.
ITEM 12. DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES
Not applicable
PART II
ITEM 13. DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES
Not applicable
ITEM 14. MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS ANDUSE OF PROCEEDS
Not applicable
ITEM 15. CONTROLS AND PROCEDURES
Not applicable
ITEM 16. RESERVED
Not applicable
PART III
ITEM 17. FINANCIAL STATEMENTS
The consolidated financial statements were prepared in accordance with Canadian GAAP and are presented in Canadian dollars. There are differences between United States and Canadian GAAP. A reconciliation of the consolidated financial statements to United States GAAP is set forth in Note 9 of the notes to the consolidated financial statements.
The consolidated financial statements are in the following order:
| 1. | Auditors' Reports; | |
| 2. | Consolidated Balance Sheets; | |
| 3. | Consolidated Statements of Operations and Deficit; | |
| 4. | Consolidated Statements of Cash Flows; and | |
| 5. | Notes to Consolidated Financial Statements. |
48
MEDICURE INC.
(A Development Stage Enterprise)
Years ended May 31, 2002 and 2001 and
the nine months ended May 31, 2000
49
AUDITORS' REPORT
To the Directors of Medicure Inc.
We have audited the consolidated balance sheets of Medicure Inc. (a Development Stage Enterprise) as at May 31, 2002 and 2001 and the consolidated statements of operations and deficit and cash flows for the years then ended, for the nine months ended May 31, 2000 and for the cumulative period from inception on September 15, 1997 to May 31, 2002. These financial statements are the responsibility of the company's management. Our responsibility is to express an opinion on these financial statements based on our audits. The cumulative statements of operations and deficit and cash flows for the period from September 15, 1997 to May 31, 2002 include amounts for the period from September 15, 1997 to August 31, 1999, which were audited by another auditor whose report has been furnished to us, and our opinion on the cumulative amounts, insofar as it relates to amounts included for the period from September 15, 1997 to August 31, 1999, is based solely on the report of the other auditor.
We conducted our audits in accordance with Canadian generally accepted auditing standards and United States generally accepted auditing standards. Those standards require that we plan and perform an audit to obtain reasonable assurance whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation.
In our opinion, based on our audit and the report of the other auditor, these consolidated financial statements present fairly, in all material respects, the financial position of the company as at May 31, 2002 and 2001 and the results of its operations and its cash flows for the years then ended, for the nine months ended May 31, 2000 and for the cumulative period from inception on September 15, 1997 to May 31, 2002 in accordance with Canadian generally accepted accounting principles.
/s/KPMGLLP
Chartered Accountants
Winnipeg, Canada
July 26, 2002
50
AUDITORS’ REPORT
TO THE DIRECTORS OF MEDICURE INC.
We have audited the statements of operations, deficit and cash flows for the cumulative period from inception on September 15, 1997 to August 31, 1999 of Medicure Inc. (a Development Stage Enterprise). These financial statements are the responsibility of the company’s management. Our responsibility is to express an opinion on these financial statements based on our audit.
We conducted our audit in accordance with Canadian generally accepted auditing standards and United States generally accepted auditing standards. Those standards require that we plan and perform an audit to obtain reasonable assurance whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation.
In our opinion, these financial statements present fairly, in all material respects, the results of its operations and its cash flows for the cumulative period from inception on September 15, 1997 to August 31, 1999 in accordance with Canadian generally accepted accounting principles.
| /s/BULAT & POUSTIE | |
| Winnipeg, Canada | |
| August 22, 2000 | CHARTERED ACCOUNTANTS |
51
MEDICURE INC.
(A Development Stage Enterprise)
Consolidated Balance Sheets
(Expressed in Canadian dollars)
May 31
| 2002 | 2001 | |||||
| Assets | ||||||
| Current assets: | ||||||
| Cash and cash equivalents | $ | 8,341,018 | $ | 3,775,472 | ||
| Accounts receivable | 152,425 | 69,539 | ||||
| Research advance (note 8) | 200,000 | 200,000 | ||||
| Prepaid expenses | 89,875 | – | ||||
| 8,783,318 | 4,045,011 | |||||
| Capital assets (note 3) | 84,571 | 63,941 | ||||
| Patent costs, net of accumulated amortization of | ||||||
| $43,147 (2001 - nil) | 508,902 | 313,088 | ||||
| $ | 9,376,791 | $ | 4,422,040 | |||
| Liabilities and Shareholders' Equity | ||||||
| Current liabilities: | ||||||
| Accounts payable and accrued liabilities | $ | 389,663 | $ | 570,077 | ||
| Shareholders’ equity: | ||||||
| Capital stock (note 4): | ||||||
| Authorized: | ||||||
| Unlimited number of common voting shares | ||||||
| Unlimited number of class A common voting shares | ||||||
| Unlimited number of preferred shares | ||||||
| Issued: | ||||||
| 37,088,864 common voting shares | ||||||
| (2001 - 21,717,641) | 16,079,309 | 7,069,057 | ||||
| 1,280,000 class A common shares | 1,379,627 | 1,379,627 | ||||
| Deficit accumulated during the development stage | (8,471,808 | ) | (4,596,721 | ) | ||
| 8,987,128 | 3,851,963 | |||||
| Nature of operations (note 1) | ||||||
| Subsequent event (note 4) | ||||||
| Commitment (notes 6 and 7) | ||||||
| $ | 9,376,791 | $ | 4,422,040 | |||
See accompanying notes to consolidated financial statements.
52
MEDICURE INC.
(A Development Stage Enterprise)
Consolidated Statements of Operations and Deficit
(Expressed in Canadian dollars)
| Cumulative from | ||||||||||||
| Nine months | inception on | |||||||||||
| Year ended | Year ended | ended | September 15, | |||||||||
| May 31, | May 31, | May 31, | 1997 to | |||||||||
| 2002 | 2001 | 2000 | May 31, 2002 | |||||||||
| Revenue: | ||||||||||||
| Interest and other income | $ | 183,912 | $ | 135,868 | $ | 54,943 | $ | 401,278 | ||||
| Expenses: | ||||||||||||
| General and administrative | 949,569 | 879,914 | 435,692 | 2,353,372 | ||||||||
| Research and | ||||||||||||
| development (note 7) | 3,103,707 | 2,472,951 | 547,461 | 6,881,265 | ||||||||
| Research and development - | ||||||||||||
| government assistance | (54,782 | ) | (2,436 | ) | (64,482 | ) | (261,741 | ) | ||||
| Research and development - | ||||||||||||
| investment | ||||||||||||
| tax credit | – | – | – | (188,312 | ) | |||||||
| Amortization | 60,505 | 17,449 | 7,865 | 88,502 | ||||||||
| 4,058,999 | 3,367,878 | 926,536 | 8,873,086 | |||||||||
| Loss for the period | (3,875,087 | ) | (3,232,010 | ) | (871,593 | ) | (8,471,808 | ) | ||||
| Deficit accumulated during the | ||||||||||||
| development stage, | ||||||||||||
| beginning of period | (4,596,721 | ) | (1,364,711 | ) | (493,118 | ) | – | |||||
| Deficit accumulated during | ||||||||||||
| the development stage, | ||||||||||||
| end of period | $ | (8,471,808 | ) | $ | (4,596,721 | ) | $ | (1,364,711 | ) | $ | (8,471,808 | ) |
| Basic and diluted loss per | ||||||||||||
| share (note 4) | $ | (0.14 | ) | $ | (0.20 | ) | $ | (0.11 | ) | |||
See accompanying notes to consolidated financial statements.
53
MEDICURE INC.
(A Development Stage Enterprise)
Consolidated Statements of Cash Flows
(Expressed in Canadian dollars)
| Cumulative from | ||||||||||||
| Nine months | inception on | |||||||||||
| Year ended | Year ended | ended | September 15, | |||||||||
| May 31, | May 31, | May 31, | 1997 to | |||||||||
| 2002 | 2001 | 2000 | May 31, 2002 | |||||||||
| Cash provided by (used in): | ||||||||||||
| Operating activities: | ||||||||||||
| Loss for the period | $ | (3,875,087 | ) | $ | (3,232,010 | ) | $ | (871,593 | ) | $ | (8,471,808 | ) |
| Adjustments for: | ||||||||||||
| Amortization of capital assets | 17,358 | 17,449 | 7,865 | 45,355 | ||||||||
| Amortization of patent costs | 43,147 | – | – | 43,147 | ||||||||
| Change in the following: | ||||||||||||
| Accounts receivable | (82,886 | ) | 153,776 | (165,956 | ) | (162,779 | ) | |||||
| Prepaid expenses | (89,875 | ) | – | – | (89,875 | ) | ||||||
| Research advance | – | (200,000 | ) | – | (200,000 | ) | ||||||
| Investment tax credit receivable | – | 152,542 | (182,429 | ) | 35,770 | |||||||
| Accounts payable and | ||||||||||||
| accrued liabilities | (180,414 | ) | 378,247 | (25,000 | ) | 400,354 | ||||||
| (4,167,757 | ) | (2,729,996 | ) | (1,237,113 | ) | (8,399,836 | ) | |||||
| Investing activities: | ||||||||||||
| Decrease (increase) in short-term | ||||||||||||
| investments | – | 2,160,000 | (2,160,000 | ) | – | |||||||
| Acquisition of capital assets | (37,988 | ) | (7,005 | ) | (78,036 | ) | (129,926 | ) | ||||
| Cash of acquired business | ||||||||||||
| at acquisition | – | – | 727,005 | 727,005 | ||||||||
| Patent costs | (238,961 | ) | (127,252 | ) | (77,992 | ) | (531,441 | ) | ||||
| (276,949 | ) | 2,025,743 | (1,589,023 | ) | 65,638 | |||||||
| Financing activities: | ||||||||||||
| Issuance of common shares, | ||||||||||||
| net of share issue costs | 9,010,252 | 2,865,860 | 4,100,340 | 15,976,552 | ||||||||
| Bank indebtedness | – | – | (16,002 | ) | – | |||||||
| Change in notes payable | – | – | (145,000 | ) | – | |||||||
| Change in due to related parties | – | – | 500,663 | 698,664 | ||||||||
| 9,010,252 | 2,865,860 | 4,440,001 | 16,675,216 | |||||||||
| Increase in cash and cash equivalents | 4,565,546 | 2,161,607 | 1,613,865 | 8,341,018 | ||||||||
| Cash and cash equivalents, | ||||||||||||
| beginning of period | 3,775,472 | 1,613,865 | – | – | ||||||||
| Cash and cash equivalents, | ||||||||||||
| end of period | $ | 8,341,018 | $ | 3,775,472 | $ | 1,613,865 | $ | 8,341,018 | ||||
| Non-cash transactions: | ||||||||||||
| Interest paid during the period | $ | – | $ | – | $ | – | $ | 10,306 | ||||
| Value assigned to shares issued | ||||||||||||
| as consideration for acquisition | ||||||||||||
| of Medicure, net of cash acquired | ||||||||||||
| of $727,005 | – | – | 755,379 | 755,379 | ||||||||
See accompanying notes to consolidated financial statements.
54
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements
(Expressed in Canadian dollars)
| 1. | Nature of operations: The company is engaged in the discovery and development of cardiovascular therapeutics and is currently in the research and development phase of its lead product, MC-1. To date, the company has no products currently in commercial production or use. Accordingly, the company is considered to be a development stage enterprise for accounting purposes. Since September 15, 1997, the date of inception of Medicure, through to May 31, 2002, the company and Medicure have expended approximately $6,448,000, net of government assistance and investment tax credits, which aggregate approximately $455,000, on the research and development of MC-1 and other compounds. To date, the company has financed its cash requirements primarily through share issuance, investment tax credits, government grants and interest income. The success of the company is dependent on its ability to obtain sufficient funds to conduct its clinical trials and to successfully commercialize its products. | |
| 2. | Significant accounting policies: | |
| (a) | Basis of presentation: These consolidated financial statements have been prepared in accordance with accounting principles generally accepted in Canada (“Canadian GAAP”). The measurement principles applied are also in conformity in all material respects with accounting principles generally accepted in the United States (“U.S. GAAP”) except as described in note 9 to the consolidated financial statements. These financial statements have been prepared on a consolidated basis to include the accounts of the company and its wholly-owned subsidiary, Medicure International Inc. All significant inter-company transactions and balances have been eliminated. | |
| (b) | Cash and cash equivalents: Cash and cash equivalents include cash on hand and balances with banks as well as highly liquid short-term investments. The company considers all highly liquid short-term investments with terms to maturity when acquired of three months or less to be cash equivalents. | |
55
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 2. | Significant accounting policies (continued): | |
| (c) | Capital assets: Capital assets are stated at cost. Amortization is recorded over the useful life of the assets at the following rates: | |
| Asset | Basis | Rate | |
| Computer equipment | Straight-line | 25% | |
| Office equipment | Diminishing balance | 20% | |
| Scientific equipment | Diminishing balance | 20% | |
| Leasehold improvements | Straight-line | 20% | |
| (d) | Patents: Costs incurred in obtaining patents are capitalized and amortized on a straight-line basis over the legal life of the respective patents, being approximately twenty years, or its economic life, if shorter. The cost of servicing the company’s patents is expensed as incurred. The company commenced amortization of patent costs during fiscal 2002 | |
| (e) | Stock option plan: The company has a Stock Option Plan [note 4(c)] for its directors, management, consultants and employees. No compensation expense is recognized when the stock options are granted. Any consideration paid on the exercise of stock options and purchase of common shares is credited to capital stock. If common shares or stock options are repurchased from directors, management, employees, or consultants, the excess of the consideration paid over the carrying amount of the common shares or stock options granted will be charged to retained earnings. | |
| (f) | Government assistance and investment tax credits: Government assistance toward current expenses is recorded as a reduction against the related expenses in the period they are incurred. Government assistance towards capital assets is deducted from the cost of the related capital asset. The benefits of investment tax credits for scientific research and development expenditures are recognized in the period the qualifying expenditure is made, providing there is reasonable assurance of recoverability. Investment tax credits receivable are recorded at their net realizable value net of any reasonably possible adjustments by Canadian tax authorities. |
56
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 2. | Significant accounting policies (continued): | |
| Investment tax credits are only available on research and development expenditures incurred directly by the company or Medicure International Inc. | ||
| (g) | Research and development: All costs of research activities are expensed in the period in which they are incurred. Development costs are charged as an expense in the period incurred unless a development project meets stringent criteria for cost deferral and amortization. No development costs have been deferred to date. | |
| (h) | Income taxes: The company follows the asset and liability method of accounting for income taxes. Under this method, future income tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis. Future income tax assets and liabilities are measured using enacted or substantively enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on future tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the date of substantive enactment. When realization of future income tax assets does not meet the more likely than not criterion, a valuation allowance is provided for the difference. | |
57
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 2. | Significant accounting policies (continued): | |
| (i) | Net earnings (loss) per share: During the year ended May 31, 2002, the company adopted, on a retroactive basis, new standards for the calculation of per share amounts. Under the new recommendation, basic earnings per share are computed using the weighted average number of shares outstanding during the year including contingently issuable shares where the contingency has been resolved. Under the new recommendation, the diluted per share amounts are calculated based on the weighted average number of common shares outstanding during the period, plus the effect of dilutive common share equivalents such as options and warrants. This method requires that diluted per share amounts be calculated using the treasury stock method, as if all the common share equivalents where the average market price for the period exceeds the exercise price had been exercised at the beginning of the reporting period, or at the date of issue, if later, as the case may be, and that the funds obtained thereby were used to purchase common shares of the company at the average trading price of the common shares during the period. A result of this change is that certain of the company’s escrowed shares outstanding are considered to be contingently issuable and are now excluded from the denominator used in the calculation of earnings (loss) per share. As a result, 1,825,537 (2001 - 1,872,000; 2000 -6,084,000) escrowed shares have been excluded from the denominator in calculating the loss per share. This change resulted in increasing loss per share by $0.04 and $0.05 and reducing the basic and diluted weighted average number of shares by 4,276,536 and 7,203,825 as reported in the 2001 and 2000 comparative periods, respectively. | |
| (j) | Foreign currency translation: Current assets and current liabilities in foreign currencies have been translated into Canadian dollars at the rates of exchange in effect at the balance sheet date. Income and expense transactions are translated at actual rates of exchange during the year. Exchange gains and losses are included in loss for the year. | |
| (k) | Use of estimates: The preparation of financial statements in accordance with generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of certain assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of certain revenue and expenses during the reporting period. Actual results could differ from those estimates. | |
58
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 3. | Capital assets: |
| Accumulated | Net book | |||||||||
| May 31, 2002 | Cost | amortization | value | |||||||
| Computer equipment | $ | 42,264 | $ | 16,694 | $ | 25,570 | ||||
| Office equipment | 9,817 | 1,750 | 8,067 | |||||||
| Scientific equipment | 63,822 | 26,338 | 37,484 | |||||||
| Leasehold improvements | 14,025 | 575 | 13,450 | |||||||
| $ | 129,928 | $ | 45,357 | $ | 84,571 | |||||
| Accumulated | Net book | |||||||||
| May 31, 2001 | Cost | amortization | value | |||||||
| Computer equipment | $ | 23,882 | $ | 9,416 | $ | 14,466 | ||||
| Office equipment | 4,233 | 906 | 3,327 | |||||||
| Scientific equipment | 63,822 | 17,674 | 46,148 | |||||||
| $ | 91,937 | $ | 27,996 | $ | 63,941 | |||||
| 4. | Capital stock: | |
| (a) | Authorized: The company has authorized share capital of an unlimited number of common voting shares, an unlimited number of class A common voting shares and an unlimited number of preferred shares. The class A common voting shares are identical in all respects to the common voting shares, except that the holders are eligible for the Manitoba Equity Tax Credit until February 28, 2003. The preferred shares may be issued in one or more series, and the directors may fix prior to each series issued, the designation, rights, privileges, restrictions and conditions attached to each series of preferred shares. | |
59
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 4. | Capital stock (continued): | |
| (b) | Shares issued and outstanding are as follows: | |
| 2002 | 2001 | ||||||
| Common shares | $ | 16,079,309 | $ | 7,069,057 | |||
| Class A common shares | 1,379,627 | 1,379,627 | |||||
| $ | 17,458,936 | $ | 8,448,684 | ||||
| Number of shares | Amount | |||||
| Common shares: | ||||||
| Balance at May 31, 2000 | 18,672,342 | $ | 4,203,197 | |||
| Private placement for cash on May 31, 2001, at | ||||||
| $1.00 per share, net of share issue costs of $153,247 | 2,600,000 | 2,446,753 | ||||
| Exercise of options for cash | 21,667 | 16,250 | ||||
| Exercise of warrants for cash | 423,632 | 402,857 | ||||
| Balance at May 31, 2001 | 21,717,641 | 7,069,057 | ||||
| Private placement for cash on June 14, 2001 | ||||||
| at $1.00 per share | 200,000 | 200,000 | ||||
| Exercise of options for cash | 8,333 | 6,250 | ||||
| Shares returned to treasury | (221,725 | ) | – | |||
| Public offering for cash on December 20, 2001 and | ||||||
| January 31, 2002 at $0.65 per share, net of share | ||||||
| issue costs of $1,195,998 | 15,384,615 | 8,804,002 | ||||
| Balance at May 31, 2002 | 37,088,864 | 16,079,309 | ||||
| Class A common shares: | ||||||
| Public offering for cash on February 28, 2000 at $1.25 | ||||||
| per share, net of share issue costs of $220,373 | 1,280,000 | 1,379,627 | ||||
| Total capital stock at May 31, 2002 | 38,368,864 | $ | 17,458,936 | |||
| (c) | Options: The company has a Stock Option Plan which is administered by the Board of Directors of the company with stock options granted to directors, management, employees and consultants as a form of compensation. The number of common shares reserved for issuance of stock options is limited to a maximum of 3,700,000 common shares of the company at any time. The stock options are subject to vesting over a period of three years. |
60
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 4. | Capital stock (continued): | |
| A summary of the company’s Stock Option Plan is as follows: | ||
| 2002 | 2001 | ||||||||||
| Weighted | Weighted | ||||||||||
| average | average | ||||||||||
| Shares | exercise price | Shares | exercise price | ||||||||
| Balance, beginning of | |||||||||||
| year | 1,787,033 | $ | 1.14 | 1,685,700 | $ | 1.00 | |||||
| Granted | 355,000 | 0.81 | 198,000 | 2.25 | |||||||
| Exercised | (8,333 | ) | 0.75 | (21,667 | ) | 0.75 | |||||
| Cancelled or expired | (160,667 | ) | 1.35 | (75,000 | ) | 0.75 | |||||
| Balance, end of year | 1,973,033 | $ | 1.05 | 1,787,033 | $ | 1.14 | |||||
| Options exercisable, end | |||||||||||
| of year | 1,167,134 | ||||||||||
| Options outstanding at May 31, 2002 consist of the following: |
| Range of | Weighted average | Options outstanding | |||||||||
| exercise | Number | remaining | weighted average | Number | |||||||
| prices | outstanding | contractual life | exercise price | exercisable | |||||||
| $ | 0.20 - 1.25 | 1,559,333 | 2.9 years | $ | 0.96 | 860,999 | |||||
| 2.15 - 2.70 | 413,700 | 2.9 years | 1.40 | 306,135 | |||||||
| 1,973,033 | 2.9 years | $ | 1.05 | 1,167,134 | |||||||
| Subsequent to May 31, 2002, 126,000 stock options were exercised and converted into common shares at $0.20 per share. |
61
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 4. | Capital stock (continued): | |
| (d) | Warrants: | |
| Granted | Granted | ||||||||||||||
| Issuee | Original | Value | May 31, | (Exercised) | May 31, | (Exercised) | May 31, | ||||||||
| (Expiry date) | granted | per share | 2000 | (Cancelled)* | 2001 | (Cancelled)* | 2002 | ||||||||
| Prospectus offerings: | |||||||||||||||
| Initial public offering | |||||||||||||||
| (April 18, 2001) | 286,720 | $ | 0.50 | 41,550 | (41,550 | ) | – | – | – | ||||||
| 1,280,000 class A | |||||||||||||||
| common shares | |||||||||||||||
| (August 31, | |||||||||||||||
| 2001) | 128,000 | 1.25 | 128,000 | – | 128,000 | (128,000 | ) * | – | |||||||
| 16,923,076 units | |||||||||||||||
| (December 20, 2003) | 16,923,076 | 0.65 - 0.81 | – | – | – | 16,923,076 | 16,923,076 | ||||||||
| Private placements: | |||||||||||||||
| 694,444 units | |||||||||||||||
| (January 27, 2002) | 694,444 | 1.00 - 1.30 | 652,916 | (382,082 | ) | 270,834 | (270,834 | ) * | – | ||||||
| 1,000,000 units | |||||||||||||||
| (July 7, 2001) | 1,000,000 | 2.40 | 1,000,000 | – | 1,000,000 | (1,000,000 | ) * | – | |||||||
| 1,360,000 units | |||||||||||||||
| (August 31, 2002) | 1,360,000 | 1.05 - 1.15 | – | 1,360,000 | 1,360,000 | – | 1,360,000 | ||||||||
| 120,000 units | |||||||||||||||
| (September 14, 2002) | 120,000 | 1.00 -1.15 | – | – | – | 120,000 | 120,000 | ||||||||
| The warrants were all issued together with common shares either under prospectus offerings or private placements with the fair value of the consideration received under the offerings allocated to the common shares issued. | ||
| (e) | Escrowed shares: As at May 31, 2002, the company’s transfer agent held 7,606,404 (2001 - 7,800,000) common shares pursuant to a performance escrow agreement. The company has met the required performance conditions on 5,780,867 (2001 - 5,928,000) of these common shares. However, the company has not applied for regulatory approval as required to release the shares from escrow. The company has not met the required performance conditions of the remaining 1,825,537 (2001 - 1,872,000) common shares at May 31, 2002. In addition, 1,485,934 (2001 - 3,000,000) common shares are being held in escrow to be released on a time release basis. |
62
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 4. | Capital stock (continued): | |
| (f) | Loss per share: The weighted average number of common shares and class A common shares outstanding for the year ended May 31, 2002 and May 31, 2001 and the nine months ended May 31, 2000 were 27,900,412, 15,928,521 and 8,224,389, respectively. | |
| 5. | Income taxes: Significant components of the company’s future tax assets and liabilities are as follows: | |
| 2002 | 2001 | ||||||
| Future tax assets: | |||||||
| Research and development expenses deductible | |||||||
| in future periods for income tax purposes | $ | 243,000 | $ | 280,000 | |||
| Investment tax credits | 76,000 | 117,000 | |||||
| Share issue costs | 511,000 | 153,000 | |||||
| Operating losses carried forward | 1,213,000 | 642,000 | |||||
| Other | 89,000 | 70,000 | |||||
| 2,132,000 | 1,262,000 | ||||||
| Less valuation allowance | (2,132,000 | ) | (1,262,000 | ) | |||
| $ | – | $ | – | ||||
| The reconciliation of the Canadian statutory rate to the income tax provision is as follows: |
| Nine months | ||||||||||
| Year ended | Year ended | ended | ||||||||
| May 31, | May 31, | May 31, | ||||||||
| 2002 | 2001 | 2000 | ||||||||
| Loss for the year: | ||||||||||
| Canadian | $ | 805,270 | $ | 836,838 | $ | 871,593 | ||||
| Foreign | 3,069,817 | 2,395,172 | – | |||||||
| $ | 3,875,087 | $ | 3,232,010 | $ | 871,593 | |||||
| Canadian federal and provincial income taxes | ||||||||||
| recovery at 44.1% (2001 - 45.7%; | ||||||||||
| 2000 - 46.1%) | $ | 1,708,000 | $ | 1,477,000 | $ | 402,000 | ||||
| Foreign tax rate differential | (1,276,000 | ) | (1,017,000 | ) | – | |||||
| Change in statutory rates | (27,000 | ) | – | – | ||||||
| Valuation allowance | (405,000 | ) | (460,000 | ) | (402,000 | ) | ||||
| $ | – | $ | – | $ | – | |||||
63
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 5. | Income taxes (continued): At May 31, 2002, the company has Canadian and Foreign unutilized operating losses carried forward for income tax purposes of $2,442,000 and $5,465,000, respectively. These losses are available to be applied against taxable income of future years, except that if not so applied, these losses will expire as follows: | |
| 2003 | $ | 158,000 | ||
| 2004 | 147,000 | |||
| 2005 | 95,000 | |||
| 2006 | 28,000 | |||
| 2007 | 278,000 | |||
| 2008 | 671,000 | |||
| 2009 | 1,065,000 | |||
| 2010 (foreign) | 2,395,000 | |||
| 2011 (foreign) | 3,070,000 | |||
| $ | 7,907,000 | |||
The company also has undeducted expenses available to reduce taxable income related primarily to share issue costs and scientific research and development expenditures of approximately $1,991,000. In addition, the company has approximately $76,000 of investment tax credits, available to offset future income tax payables, which expire in 2010 and 2011. | ||
| 6. | Commitment: The company leases its premises under an operating lease. Minimum annual rental payments to the end of the lease term are as follows: | |
| 2003 | $ | 23,507 | ||
| 2004 | 23,507 | |||
| 2005 | 23,507 | |||
| 2006 | 23,507 | |||
| 2007 | 17,630 | |||
| $ | 111,658 | |||
| The annual lease payments are exclusive of maintenance, property taxes, insurance and other operating costs. |
64
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 7. | Related party transactions: During the year ended May 31, 2002, the company paid companies controlled by a director, a total of $215,000 (year ended May 31, 2001 - $242,000; nine months ended May 31, 2000 - $155,640) for office rent and supplies and consulting fees. On June 1, 2000, Medicure International Inc. entered into a development agreement with CanAm Bioresearch Inc. (“CanAm”), a private Canadian company owned by a then, and now, former director and officer of the company, whereby CanAm performs research and development of MC-1 and its related compounds on a cost plus up to ten percent recovery basis up to a maximum of direct research and development expenditures of $10,000,000. During the year ended May 31, 2002, the company incurred an aggregate of $3,021,115 (2001 - $2,364,440) in expenditures under this agreement which is included in research and development expenses on the statement of operations. As at May 31, 2002, the company has provided a research advance to CanAm of $200,000 (2001 - $200,000) which is non-interest bearing, unsecured and repayable on demand. These transactions are measured at the exchange amount which is the amount of consideration established and agreed to by the related parties. |
| 8. | Financial instruments: The fair values of cash and cash equivalents, accounts receivable, research advance and accounts payable and accrued liabilities approximate their carrying values due to their short term to maturity. |
| 9. | Reconciliation of generally accepted accounting principles: The company prepares its consolidated financial statements in accordance with Canadian GAAP which, as applied in these consolidated financial statements, conform in all material respects to U.S. GAAP, except as follows: |
65
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 9. | Reconciliation of generally accepted accounting principles (continued): | |
| (a) | Patents: Under Canadian GAAP, the patent costs which relate to products which have not yet received regulatory approval are included as an asset on the balance sheet. Under U.S. GAAP, the unamortized patent costs would have been recorded as an expense in the year of incurrence. The effect of this difference is that for the years ended May 31, 2002 and 2001 and the nine months ended May 31, 2000, research and development expense would have increased by $238,961, $127,252 and $77,992, respectively. The company commenced amortization of the patents during fiscal 2002. Under U.S. GAAP, the amortization expense to be added back is $43,147 for the year ended May 31, 2002. | |
| (b) | Scientific equipment: Scientific equipment acquired solely for research and development activities has been capitalized and amortized over its useful life for Canadian GAAP purposes. Under U.S. GAAP, this equipment would be charged to research and development expense as incurred as it does not have alternative future use. The effect of this difference is that for the years ended May 31, 2002 and 2001 and for the nine months ended May 31, 2000, research and development expense would have increased nil, nil and $61,170, respectively. Amortization of the scientific equipment for Canadian GAAP would be added back to the loss for the period for U.S. GAAP reconciliation purposes. The amortization to be added back for the years ended May 31, 2002 and 2001 and the nine months ended May 31, 2000 is $8,664, $11,537, and $5,551, respectively. | |
| (c) | Stock options - stock based compensation costs: For reconciliation purposes to U.S. GAAP, the company has elected to follow the fair value method in accounting for its employee and director stock options. Under U.S. GAAP, stock based compensation to non-employees must be recorded at the fair value of the options granted. The company uses the Black-Scholes option pricing model to determine the fair value of all options granted. The assumptions used in the valuation included a five year life for the options, a risk-free rate of between 5.00% and 5.80%, volatility between 37% and 65% and no dividend yield. This compensation expense would be amortized over the appropriate vesting periods. For purposes of reconciliation of U.S. GAAP, the company would record an additional compensation expense for the years ended May 31, 2002 and 2001 and the nine months ended May 31, 2000 of approximately $257,000, $550,000 and $233,000, respectively. | |
66
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 9. | Reconciliation of generally accepted accounting principles (continued): | |
| (d) | Escrowed common shares: Under Canadian GAAP, common shares of the company under escrow arrangements are included in capital stock at the time of issuance based on the total number of shares issued and the issuance price. No additional compensation expense is recorded when the common shares are released from escrow. Under U.S. GAAP, the aggregate 1,485,934 (2001 - 3,000,000; 2000 - 4,500,000) common shares of the company held in escrow which are to be released on a time release basis are accounted for in the same manner as under Canadian GAAP. A compensation expense however, would be recorded under U.S. GAAP, upon eligibility for release of the 7,606,404 (2001 - 7,800,000; 2000 - 7,800,000) escrowed common shares of the company, with the release based on performance conditions being met. The compensation expense would be accounted for as the difference between the market value of the company’s common shares at the time the common shares are eligible for release from escrow and the price paid per common share at the time of issuance multiplied by the number of common shares released from escrow. As at May 31, 2002, performance conditions on 5,780,867 (2001 - 5,928,000; 2000 - 1,716,000) of the 7,606,404 (2001 and 2000 - 7,800,000) common shares under escrow have been met. For purposes of reconciliation of U.S. GAAP, the company would record an additional compensation expense for the years ended May 31, 2002 and 2001 and the nine months ended May 31, 2000 of nil, $6,312,000 and $8,065,000, respectively. As at May 31, 2002, performance conditions of 1,825,537 of the 7,606,404 common shares under escrow have not been met. | |
| (e) | Recent pronouncements: In June 2001, the FASB issued Statement No. 141, “Business Combinations” (“SFAS 141”), which supersedes APB Opinion No. 16, “Business Combinations,” and SFAS 38, “Accounting for Preacquisition Contingencies of Purchased Enterprises.” Concurrently, the CICA issued Handbook Section 1581,Business Combinations,which is consistent with SFAS 141. Those Statements will change the accounting for business combinations and goodwill. SFAS 141 and CICA Handbook Section 1581 require that the purchase method of accounting be used for all business combinations initiated after June 30, 2001. Use of the pooling of interests method is no longer permitted. These Statements also establish criteria for separate recognition of intangible assets acquired in a purchase business combination. Adoption of this standard will not have a material effect on the company’s results from operations or financial position. | |
67
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 9. | Reconciliation of generally accepted accounting principles (continued): In June 2001, the FASB issued Statement No. 142, “Goodwill and Other Intangible Assets” (“SFAS 142”), which supersedes APB Opinion No. 17, “Intangible Assets.” Concurrently, the CICA issued Handbook Section 3062,Goodwill and Other Intangible Assets,which is consistent with SFAS 42. These Statements require that goodwill no longer be amortized to earnings but instead be reviewed for impairment. The Statements are effective for fiscal years beginning after December 15, 2001, and are required to be applied at the beginning of an entity’s fiscal year, and to be applied to all goodwill and other intangible assets recognized in its financial statements at that date. The company has no goodwill or indefinite life intangible assets at May 31, 2002. Accordingly, adoption of this standard is not expected to have a material effect on the company’s results from operations or financial position. In August 2001, the FASB issued SFAS No. 143, “Accounting for the Asset Retirement Obligations.” SFAS No. 143 addresses financial accounting and reporting for obligations and costs associated with the retirement of tangible long-lived assets, and is effective for years beginning after June 15, 2001. Adoption of this standard is not expected to have a material effect on the company’s results from operations or financial position. In October 2001, the FASB issued SFAS No. 144, “Accounting for the Impairment or Disposal of Long-Lived Assets.” SFAS No. 144 replaces SFAS No. 121, “Accounting for the Impairment of Long-Lived Assets and for Long-Lived Assets to be Disposed Of” and establishes accounting and reporting standards for long-lived assets to be disposed of by sale. This standard applies to all long-lived assets, including discontinued operations. SFAS No. 144 requires those assets to be measured at the lower of carrying amount or fair value less cost to sell. SFAS No. 144 also broadens the reporting of discontinued operations to include all components of an entity with operations that can be distinguished from the rest of the entity that will be eliminated from the ongoing operations of the entity in a disposal transaction. SFAS 144 is effective for fiscal years beginning after December 15, 2001. Adoption of this standard is not expected to have a material effect on the company’s results from operations or financial position. |
68
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 9. | Reconciliation of generally accepted accounting principles (continued): Summary: The impact of significant variations to U.S. GAAP on the consolidated statement of operations and deficit are as follows: |
| Nine months | Cumulative from | ||||||||||||
| Year ended | Year ended | ended | inception on | ||||||||||
| May 31, | May 31, | May 31, | September 15, 1997 | ||||||||||
| 2002 | 2001 | 2000 | to May 31, 2002 | ||||||||||
| Loss for the period, Canadian | |||||||||||||
| GAAP | $ | (3,875,087 | ) | $ | (3,232,010 | ) | $ | (871,593 | ) | $ | (8,471,808 | ) | |
| Adjustments for the following: | |||||||||||||
| Stock based compensation | (257,000 | ) | (550,000 | ) | (233,000 | ) | (1,040,000 | ) | |||||
| Patent costs | (238,961 | ) | (127,252 | ) | (77,992 | ) | (531,501 | ) | |||||
| Amortization of patent costs | 43,147 | – | – | 43,147 | |||||||||
| Scientific equipment | – | – | (61,170 | ) | (63,822 | ) | |||||||
| Amortization of scientific | |||||||||||||
| equipment | 8,664 | 11,537 | 5,551 | 26,338 | |||||||||
| Escrowed common share | |||||||||||||
| compensation | – | (6,312,000 | ) | (8,065,000 | ) | (14,377,000 | ) | ||||||
| Loss for the period, U.S. GAAP | $ | (4,319,237 | ) | $ | (10,209,725 | ) | $ | (9,303,204 | ) | $ | (24,414,646 | ) | |
| Basic and diluted loss per share, | |||||||||||||
| U.S. GAAP | $ | (0.15 | ) | $ | (0.64 | ) | $ | (1.13 | ) | ||||
| The impact of significant variations to U.S. GAAP on the consolidated statements of cash flows are as follows: |
| Nine months | Cumulative from | ||||||||||||
| Year ended | Year ended | ended | inception on | ||||||||||
| May 31, | May 31, | May 31, | September 15, 1997 | ||||||||||
| 2002 | 2001 | 2000 | to May 31, 2002 | ||||||||||
| Operating activities | $ | (4,406,718 | ) | $ | (2,857,248 | ) | $ | (1,376,275 | ) | $ | (8,995,159 | ) | |
| Investing activities | 37,988 | 2,152,995 | (1,449,861 | ) | 660,961 | ||||||||
69
MEDICURE INC.
(A Development Stage Enterprise)
Notes to Consolidated Financial Statements (continued)
(Expressed in Canadian dollars)
| 9. | Reconciliation of generally accepted accounting principles (continued): The impact of significant variations to U.S. GAAP on the consolidated balance sheet items are as follows: |
| 2002 | 2001 | ||||||
| Capital assets | $ | 47,087 | $ | 17,793 | |||
| Capital stock | 32,855,388 | 23,588,136 | |||||
| Deficit accumulated during the development stage | (24,414,646 | ) | (20,095,409 | ) | |||
70
ITEM 18. FINANCIAL STATEMENTS
Not applicable.
ITEM 19. EXHIBITS
The exhibits are in the following order:
| 1. | Articles of Incorporation and Bylaws: | ||
| (a) | Medicure’s Articles of Incorporation dated September 15, 1997 [1] | ||
| (b) | Lariat’s Articles of Incorporation dated June 3, 1997 [1] | ||
| (c) | Medicure’s Certificate of Continuance from Manitoba to Alberta dated December 3, 1999 [1] | ||
| (d) | Certificate of Amalgamation for Medicure and Lariat dated December 22, 1999 [1] | ||
| (e) | Medicure’s Certificate of Continuance from Alberta to Canada dated February 23, 2000 [1] | ||
| 4. | Material Contracts: | ||
| a) | Escrow Agreement dated February 12, 1999 among the Corporation, Montreal Trust Company of Canada, and certain shareholders of the Corporation [1]; | ||
| b) | Performance Based Escrow Agreement, as amended, dated as of November 23, 1999 among the Corporation, Montreal Trust Company of Canada and certain shareholders of the Corporation [1]; | ||
| c) | Timed Release Escrow Agreement, as amended, dated as of November 23, 1999 among the Corporation, Montreal Trust Company of Canada and certain shareholders of the Corporation [1]; | ||
| d) | Edstrom Escrow Agreement, dated as of November 5, 1999 among the Corporation, Montreal Trust Company of Canada and Dr. Daryle Edstrom [1]; | ||
| e) | Business and Administration Services Agreement between the Corporation and Genesys Venture Inc. dated as of October 1, 2001, wherein Genesys Venture Inc. agreed to provide the Corporation with certain business services [1]; | ||
| f) | License Agreement between Medicure and the University of Manitoba dated August 30, 1999, wherein the University of Manitoba granted to Medicure an exclusive license with regard to certain intellectual property (the “U of M Licensing Agreement”) [1]; | ||
| g) | Research Agreement between Medicure and the University of Manitoba dated August 30, 1999, wherein the University of Manitoba agreed to provide Medicure with premises and facilities to conduct research and development activities. This Agreement (the original term of which has expired) was assigned by the Corporation to CanAm and has been extended pursuant to a letter agreement dated May 25, 2000 [1]; | ||
| h) | Transfer Agency Agreement between Montreal Trust Company of Canada and the Corporation dated as of January 26, 2000, whereby Montreal Trust Company of Canada agreed to act as transfer agent and registrar with respect to the Shares [1]; | ||
71
| i) | Medicure International Licensing Agreement between the Corporation and Medicure International Inc. dated June 1, 2000, wherein the Corporation granted Medicure International Inc a license with regard to certain intellectual property [1]; | ||
| j) | Development Agreement between Medicure International Inc. and CanAm Bioresearch Inc. dated June 1, 2000, wherein CanAm Bioresearch Inc. agreed to conduct research and development activities for Medicure International [1]; | ||
| k) | Three option agreements between the Corporation and George Washington University (“GWU”) dated October 25, 2000, wherein GWU grants to the Corporation the option to evaluate three patented therapeutics for their potential in treatment of cardiovascular and other diseases and the option to thereafter obtain the worldwide right to and license the three products (the “GWU Option Agreements”). Under terms specified in the agreements, the Corporation could acquire exclusive worldwide commercial development rights to the technologies in exchange for sales royalties and other payments based on the successful commercialization of resulting products [1]; | ||
| l) | Investor Relations Contract dated March 28, 2002 between Boxe Studio Intl. and the Corporation whereby investor relations services will be provided to the Corporation including identifying possible sources of capital, providing analysis and advice with respect to capital structure, advising and assisting the Corporation with respect to its relationship with existing shareholders and assisting in the preparation of public information materials [2]; and | ||
| m) | Amendment to the Consulting Services Agreement dated February 1, 2002 between A.D. Friesen Enterprises Ltd. and the Corporation whereby consulting services will be provided to the Corporation by Dr. Albert D. Friesen [2]. | ||
| [1] Herein incorporated by reference as previously included in the Corporation’s Form 20-F registration statement filed on January 30, 2001 | |||
| [2] Herein incorporated by reference as previously included in the Corporation’s Form 20-F annual report filed on October 28, 2002 | |||
72
SIGNATURE PAGE
Pursuant to the requirements of Section 12 of theSecurities Exchange Act of 1934, the Corporation certifies that it meets all of the requirements for filing on Form 20-F and has duly caused this annual report to be signed on its behalf by the undersigned, thereunto duly authorized.
Dated: December 27, 2002
ON BEHALF OF THE CORPORATION, MEDICURE INC.
per:
/s/Albert D. Friesen__________________________________________________
Albert D. Friesen, Ph.D.
Chairman, President & CEO
| CERTIFICATION OF CHIEF EXECUTIVE OFFICER PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002 I, Albert D. Friesen, certify that: | |
| 1. | I have reviewed this annual report on Form 20-F of Medicure Inc.; |
| 2. | Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report; and |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report. Date: December 27, 2002 |
/s/ Albert D. Friesen | |
| CERTIFICATION OF CHIEF FINANCIAL OFFICER PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002 I, Derek Reimer, certify that: | |
| 1. | I have reviewed this annual report on Form 20-F of Medicure Inc.; |
| 2. | Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report; and |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report. Date: December 27, 2002 |
/s/ Derek Reimer -------------------------- Derek Reimer, Chief Financial Officer | |
CERTIFICATION PURSUANT TO 18 U.S.C. SECTION 1350 AS ADOPTED PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
I, Albert D. Friesen, Chief Executive Officer, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that:
| 1 | . | the Annual Report on Form 20-F of Medicure Inc. (the “Company”) for the annual period |
| ended May 31, 2002 (the “Report”) fully complies with the requirements of Section 13(a) | ||
| or 15(d) of the Securities Exchange Act of 1934; and | ||
| 2 | . | the information contained in the Report fairly presents, in all material respects, the |
| financial condition and results of operations of the Company. |
Date: December 27, 2002
/s/ Albert D. Friesen
___________________
Albert D. Friesen, Chief Executive Officer
The foregoing certification is being furnished solely pursuant to section 906 of the Sarbanes-Oxley Act of 2002 (subsections (a) and (b) of section 1350, chapter 63 of title 18, United States Code) and is not being filed as part of the Form 20-F or as a separate disclosure document.
CERTIFICATION PURSUANT TO 18 U.S.C. SECTION 1350 AS ADOPTED PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
I, Derek Reimer, Chief Financial Officer, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that:
| 1 | . | the Annual Report on Form 20-F of Medicure Inc. (the “Company”) for the annual period |
| ended May 31, 2002 (the “Report”) fully complies with the requirements of Section 13(a) | ||
| or 15(d) of the Securities Exchange Act of 1934; and | ||
| 2 | . | the information contained in the Report fairly presents, in all material respects, the |
| financial condition and results of operations of the Company. |
Date: December 27, 2002
/s/ Derek Reimer
___________________
Derek Reimer, Chief Financial Officer
The foregoing certification is being furnished solely pursuant to section 906 of the Sarbanes-Oxley Act of 2002 (subsections (a) and (b) of section 1350, chapter 63 of title 18, United States Code) and is not being filed as part of the Form 20-F or as a separate disclosure document.