BUDD LARNER
A PROFESSIONAL CORPORATION
COUNSELLORS AT LAW
150 JOHN F. KENNEDY PARKWAY
SHORT HILLS, NEW JERSEY 07078-2703
973.379.4800
Fax 973.379.7734
www.buddlarner.com
March 25, 2013
Jim B. Rosenberg
Senior Assistant Chief Accountant
Securities and Exchange Commission
Washington, D.C. 20549
United States of America
| Re: | Dr. Reddy’s Laboratories Limited (the “Company” or “Dr. Reddy’s”) |
Form 20-F for Fiscal Year Ended March 31, 2012
Letter from Securities and Exchange Commission dated March 12, 2013
File No. 001-15182
Dear Mr. Rosenberg:
This firm serves as outside United States legal counsel to Dr. Reddy’s. We set forth below responses on behalf of Dr. Reddy’s to the comments of the staff (the “Staff”) of the Securities and Exchange Commission (the “SEC”) contained in your letter dated March 12, 2013 requesting information in connection with Dr. Reddy’s Form 20-F for the fiscal year ended March 31, 2012 (the “2012 Form 20-F”). For your convenience, your comments have been restated below in italics, followed by Dr. Reddy’s responses thereto. As noted below, Dr. Reddy’s proposes to make any enhancements to its disclosure in its next Annual Report on Form 20-F for the fiscal year ended March 31, 2013 (the “2013 Form 20-F”).
Item 5. Operating and Financial Review and Prospects
Item 5.A Operating results
Fiscal Year Ended March 31, 2012 Compared to fiscal Year Ended March 31, 2011
Revenues, page 68
1. Please provide us proposed revised disclosure to be included in future periodic reports that:
| a. | Separately quantifies, for each segment and geographic region you discuss, the impact on revenues of new product/service launches and price versus volume changes for previously existing products and services. Separately discuss the underlying causes for the changes in volume and prices for previously existing products/services. |
Response: The Staff’s comments are duly noted. In response to this comment, the Company will include enhanced disclosures similar to the following to be included in the narrative discussion of its segment level revenues in future annual reports commencing with the Company’s 2013 Form 20-F.
“The foregoing [increase] [decrease] in revenues of this segment was attributed to the following factors: approximately [ %] of the [increase] [decrease] resulted from an [increase] [decrease] in sales prices of products in this segment, approximately [ %] of the [increase] [decrease] resulted from an [increase] [decrease] in sales volumes of existing products in this segment, and approximately [ %] of the increase resulted from the introduction of new products in this segment.
The Company also proposes that, if it is readily apparent that the underlying causes for an increase or decrease in segment volumes or prices are primarily attributable to specific reasons, it will disclose such reasons. In some cases, however, it may not be feasible for the Company to discuss the underlying reasons for an increase or decrease. For example, the Company may believe that the reasons for the increase or decrease are not readily apparent or are too numerous and complex to render attribution feasible. These reasons often include, among others, the effects of marketing efforts through various brand promotion strategies, market dynamics such as competitor pricing, consolidation of customers, and availability of products at the right place and at the right time.
Jim B. Rosenberg
Senior Assistant Chief Accountant
Securities and Exchange Commission
March 25, 2013
Page 2
The Company will also highlight if there are any exceptional reasons for increases in its sales of any product, as was done on page 69 of its 2012 Form 20-F with respect to the 180 days marketing exclusivity of Olanzapine and Ziprasidone.
1. Please provide us proposed revised disclosure to be included in future periodic reports that:
| b. | Quantifies for the North American portion of your Global Generics segment the “higher contributions from your Shreveport facility” and clarifies why you are experiencing higher sales levels from this facility. |
Response: The Staff’s comments are duly noted. In response to this comment, the Company’s Shreveport facility accounted for approximately 5% of the revenue growth of the Company’s Global Generics segment, primarily due to a new multi-year contract from a customer.
The Company will include enhanced disclosures similar to the foregoing in its future annual reports, commencing with its 2013 Form 20-F, if it experiences a significant increase or decrease in revenues from its Shreveport facility.
1. Please provide us proposed revised disclosure to be included in future periodic reports that:
| c. | Quantifies for your Pharmaceutical Services and Active Ingredients (PSAI) segment the impact of each reason provided for the increase in revenue and explains the underlying causes. For example, please explain why there was a strong recovery of customer orders in your pharmaceutical services segment. |
Response: The Staff’s comments are duly noted. In response to this comment, the Company will include enhanced disclosures similar to the following in its future annual reports commencing with its 2013 Form 20-F. The new disclosure is highlighted inbold and double underscored for ease of reference.
“Our PSAI segment’s revenues for the year ended March 31, 2012 were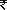 23,812 million, an increase of 21% as compared to the year ended March 31, 2011. This was largely attributable to:
23,812 million, an increase of 21% as compared to the year ended March 31, 2011. This was largely attributable to:
| • | increase in the sales of active pharmaceutical ingredients to generic customersto support their generic product launches related to impending patent expirations, which increased our PSAI segment’s revenues by [ %]; |
| • | strong recovery of customer orders in our PSAI segmentprimarily due to increased pharmaceutical development services for certain products provided to innovator companies, which increased our PSAI segment’s revenues by [ %]; and |
| • | the impact of depreciation of the Indian rupee against multiple currencies, which increased our PSAI segment’s revenues by [ %].” |
1. Please provide us proposed revised disclosure to be included in future periodic reports that:
| d. | Provides the expected effect, if any, of the underlying cause or for any known trend, event or uncertainty. |
Response: The Staff’s comments are duly noted. Disclosure regarding the expected effect, if any, of an underlying cause or for a known trend, event or uncertainty will inherently vary depending on the underlying cause, trend, event or uncertainty, and so providing proposed disclosure for future reports at this time would be extremely difficult. In addition, the Company believes that, in many cases, it will be extremely difficult for the Company to quantify an expected effect, if any, in its future annual reports. However, the Company does highlight any expected key regulatory changes or uncertainties. For example, please see page numbers 30, 32, 36 and 37 of the Company’s 2012 Form 20-F for expected regulatory changes in India, Russia and the United States and page 69 of the Company’s 2012 Form 20-F for information about the delay in the anticipated launch of one of the Company’s key products, atorvastatin. In future annual reports, where the Company discloses a trend, event or uncertainty which the Company believes will have a material impact on its business, financial conditions or results of operations, the Company will provide disclosure, if the cause and the expected effect is known and can be readily quantified.
Jim B. Rosenberg
Senior Assistant Chief Accountant
Securities and Exchange Commission
March 25, 2013
Page 3
1. Please provide us proposed revised disclosure to be included in future periodic reports that:
| e. | Modifies your disclosure regarding expected key product launches in the next fiscal year to identify the products or cross-reference to the discussion provided elsewhere. |
Response: The Staff’s comments are duly noted. In response to this comment, the Company believes that disclosure of expected key product launches could cause it competitive harm, as competitors would gain insights into the Company’s strategy enabling them to compete with the Company unfairly on the basis of such information. Further, frequently there are significant delays in obtaining approvals for some of the Company’s products from the applicable regulatory authorities, such as the U.S. Food and Drug Administration. Consequently, the launch timelines can shift significantly, which would make the disclosure of such expected launches incorrect and misleading. Accordingly, the Company believes that it is not prudent or practical for it to make disclosures of expected key product launches.
2. In the table on page 69 you disclose your Global Generics segment product launches in North America and the estimated annual market size. Given that the estimated annual market size of your Donepezil HCL, Venlafaxine-XR and Letrozole products is each at least $1.7 billion, please tell us why you do not appear to consider these launches to be key business developments that you disclose in Item 4 beginning on page 23. In this regard, for example, it appears that you identify the July 25, 2011 launch of fondaparinux sodium injection in the U.S. as a key business development, yet the estimated annual market size of this product is only $320 million.
Response: The Staff’s comments are duly noted. In response to this comment, the Company wishes to clarify that the estimated annual market size as mentioned for each of the new product launches is the approximate total annual market size in the United States for that particular product, at the time of the Company’s generic launch, as per IMS Health.
Insofar as fondaparinux is concerned, although the total market size of this product was only U.S.$320 million, the Company’s revenues from sales of this product were much higher as compared to Donepezil HCL, Venlafaxine-XR and Levofloxacin, due to limited competition in the market. Accordingly, the Company featured this product launch in Item 4 of its 2012 Form 20-F as a key business development.
In the case of Donepezil HCL, Venlafaxine-XR and Levofloxacin, although the total market size of each of these products was at least U.S.$1.7 billion, the Company’s revenues from sales of each of these products were insignificant. This is because of the existence of a large number of competitors selling these products, which resulted in a lower market share for the Company and higher price erosion for such products. Accordingly, the launch of these products was not significant for the Company and, therefore, they were not discussed as key business developments.
Gross Margin, Page 70
3. In your discussion of the reasons for the change in gross margin, you indicate there was an unfavorable impact of price erosions in some of your existing products. Please provide us proposed revised disclosure to be included in future periodic reports that clarifies whether the price erosion relates to more than the pricing challenges faced in Germany and identify any other significant products or markets facing erosion.
Response: The Staff’s comments are duly noted. In response to this comment, the Company will include enhanced disclosures similar to the following to be included in its future annual reports commencing with the Company’s 2013 Form 20-F. The new disclosure is highlighted inbold and double underscored for ease of reference.
“The change in gross margin was primarily on account of the following:
| • | the favorable impact of launches of certain high margin new products in the United States; |
Jim B. Rosenberg
Senior Assistant Chief Accountant
Securities and Exchange Commission
March 25, 2013
Page 4
| • | the favorable impact of depreciation of the Indian rupee against multiple currencies in the markets in which we operate; and |
| • | the unfavorable impact of price erosions in some of our existing productsin the United States and Germany.” |
Selling, general and administrative expenses, page 71
4. Please provide us proposed revised disclosure to be provided in future periodic reports that quantifies each reason provided for the increase or decrease in your selling, general and administrative expenses.
Response: The Staff’s comments are duly noted. In response to these comments, the Company will include enhanced disclosures similar to the following to be included in its future annual reports commencing with the Company’s 2013 Form 20-F. The new disclosure is highlighted inbold and double underscored for ease of reference.
“Our selling, general and administrative expenses for the year ended March 31, 2012 were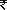 28,867, an increase of 22% as compared to
28,867, an increase of 22% as compared to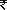 23,689 for the year ended March 31, 2011.Including the unfavorable impact of depreciation of the Indian rupee against multiple currencies in the markets in which we operate,this increase was primarily on account of the following:
23,689 for the year ended March 31, 2011.Including the unfavorable impact of depreciation of the Indian rupee against multiple currencies in the markets in which we operate,this increase was primarily on account of the following:
| • | increased personnel costs, due to annual raises and new recruitments,which increased our selling, general and administrative expenses by [ %]; and |
| • | higher distribution costs, due to increases in sales volumes and freight cost increases,which increased our selling, general and administrative expenses by [ %].” |
Notes to Consolidated Financial Statements
Note 3: Significant accounting policies
f. Intangible assets, page F-16
5. In your research and development intangible asset policy disclosure on page F-17 you indicate that you capitalize payments to third parties for in-licensed products and compounds if regulatory approval for the products is available from the counterparty or if other contractual terms provide for a refund should the regulatory approvals not be received. Please explain to us how your policy is consistent with the guidance in paragraph 25 of IAS 38 as you separately disclose, and why this guidance would not require capitalization of separately acquired intangible assets even if no refund provision exists for the failure to obtain regulatory approval.
Response: The Staff’s comments are duly noted. Historically, all payments to third parties for in-licensed products and compounds have been capitalized in accordance with the guidance provided by Paragraph 25 of IAS 38 “Intangible assets”.
The Company proposes to clarify the relevant section of its accounting policy in the Company’s future annual reports commencing with its 2013 Form 20-F as follows:
“Payments to third parties, which generally take the form of up-front payments and milestones, for in-licensed products and compounds are capitalized. The Company’s criteria for capitalization of such assets are consistent with the guidance given in paragraph 25 of International Accounting Standard 38 (“IAS 38”) (i.e., receipt of economic benefits out of the separately purchased transaction is considered to be probable).”
* * * * *
Jim B. Rosenberg
Senior Assistant Chief Accountant
Securities and Exchange Commission
March 25, 2013
Page 5
Should any member of the Staff have any questions or comments concerning this letter, please do not hesitate to call me at (973) 315-4444 or Jonathan Gray of this office at (973) 315-4481.
Respectfully submitted,
/s/ James F. Fitzsimmons, Esq.
James F. Fitzsimmons, Esq.
| cc: | Saumen Chakraborty (Dr. Reddy’s) |
M V Narasimham (Dr. Reddy’s)
Vijay Hurgat (Dr. Reddy’s)
Jonathan Gray, Esq. (Budd Larner)