Filed by Corgentech Inc. Pursuant to Rule 425
Under the Securities Act of 1933
And Deemed Filed Pursuant to Rule 14a-12
Under the Securities Exchange Act of 1934
Subject Company: AlgoRx Pharmaceuticals, Inc.
Commission File No. of Subject Company: 000-51146
*** *** ***
The following is a slide presentation used at investor meetings on October 19, 2005.

Creating a Late-Stage Company Focused on Pain Management and Inflammation
BIO Investor Forum 2005
October 19, 2005

FORWARD LOOKING STATEMENTS
This presentation includes “forward-looking statements” within the meaning of the safe harbor provisions of the United States Private Securities Litigation Reform Act of 1995. Words such as “expect,” “estimate,” “project,” “budget,” “forecast,” “anticipate,” “intend,” “plan,” “may,” “will,” “could,” “should,” “believes,” “predicts,” “potential,” “continue,” and similar expressions are intended to identify such forward-looking statements. Forward-looking statements in this presentation include, without limitation, forecasts of product development, FDA filings, benefits of the proposed merger, and other matters that involve known and unknown risks, uncertainties and other factors that may cause actual results, levels of activity, performance or achievements to differ materially from results expressed or implied in this presentation. Such risk factors include, among others: difficulties encountered in integrating merged businesses; uncertainties as to the timing of the merger; approval of the transaction by the stockholders of the companies; the satisfaction of closing conditions to the transaction, including the receipt of regulatory approvals; whether certain market segments grow as anticipated; the competitive environment in the biotechnology industry; and whether the companies can successfully develop new products and the degree to which these gain market acceptance. Actual results may differ materially from those contained in the forward-looking statements in this presentation. Additional information concerning these and other risk factors is contained in Corgentech’s Form 10-K/A for the year ended December 31, 2004 and most recently filed Form10-Q.
Corgentech and AlgoRx undertake no obligation and do not intend to update these forward-looking statements to reflect events or circumstances occurring after this press release. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this presentation. All forward-looking statements are qualified in their entirety by this cautionary statement.
2

FORWARD LOOKING STATEMENTS (2)
Additional Information and Where to Find It
Corgentech Inc. intends to file a registration statement on Form S-4, and Corgentech and AlgoRx Pharmaceuticals, Inc. intend to file a related joint proxy statement/prospectus, in connection with the merger transaction involving Corgentech and AlgoRx. Investors and security holders are urged to read the registration statement on Form S-4 and the related joint proxy/prospectus when they become available because they will contain important information about the merger transaction. Investors and security holders may obtain free copies of these documents (when they are available) and other documents filed with the SEC at the SEC’s web site at www.sec.gov. In addition, investors and security holders may obtain free copies of the documents filed with the SEC by contacting Corgentech Investor Relations at the email address: investors@corgentech.com.
Corgentech, AlgoRx and their directors and executive officers may be deemed to be participants in the solicitation of proxies from the stockholders of Corgentech and AlgoRx in connection with the merger transaction. Information regarding the special interests of these directors and executive officers in the merger transaction will be included in the joint proxy statement/prospectus of AlgoRx and Corgentech described above. Additional information regarding the directors and executive officers of Corgentech is also included in Corgentech’s proxy statement for its 2005 Annual Meeting of Stockholders, which was filed with the SEC on April 27, 2005. This document is available free of charge at the SEC’s web site at www.sec.gov and from Investor Relations at Corgentech as described above.
3 |
|

MERGER AGREEMENT
Stock Transaction
Corgentech to issue sufficient shares such that AlgoRx shareholders would own, in a tax-free exchange, 62% and Corgentech shareholders would own 38% of the combined company, respectively
Combined management team will work to integrate the companies and to identify synergies and redundancies Board and Management
Richard Brewer, Dr. Charles Cohen, Thomas Colligan, Carter Eckert, Dr. Rodney Ferguson, John McLaughlin, Dr. Arnold Oronsky and Dr. Michael Powell Management
John McLaughlin as CEO
Dr. Ronald Burch, current AlgoRx CEO, to serve as VP of Development Richard Powers to remain as CFO
James Huang newly elected as president with responsibility for commercial and other operations Secaucus and South San Francisco facilities to remain open Headquarters: South San Francisco
4 |
|

DEAL TIMELINE
Merger requires stockholder approval
Both boards have approved merger
Merger documents to be filed imminently
S-4 and related joint Proxy Statement
Shareholder vote expected to occur in late 2005 / early 2006
5 |
|

COMBINED COMPANY
Pain Management and Inflammation
Sizable Patient Populations
Unmet or Underserved Medical Needs Differentiated Products with Clear Advantages over Current Therapies Substantial Body of Preclinical and Clinical Data Clear Regulatory Pathways Faster, Cheaper Trials
6 |
|
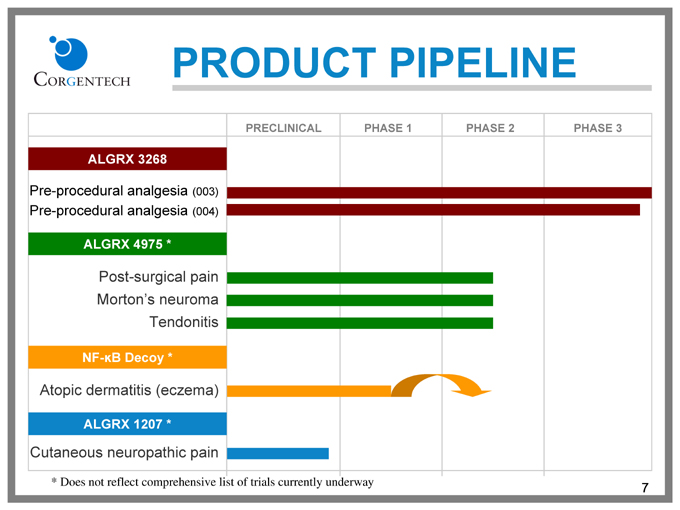
PRODUCT PIPELINE
PRECLINICAL PHASE 1 PHASE 2 PHASE 3
ALGRX 3268
Pre-procedural analgesia (003) Pre-procedural analgesia (004)
ALGRX 4975 *
Post-surgical pain Morton’s neuroma Tendonitis
NF-kB Decoy *
Atopic dermatitis (eczema)
ALGRX 1207 *
Cutaneous neuropathic pain
* |
| Does not reflect comprehensive list of trials currently underway |
7 |
|

CREATING LATE-STAGE COMPANY
Four product development programs focused on significant clinical problems with attractive market potential
Each drug has unique mechanism of action offering risk diversification
Each drug has potential for approval in multiple indications
Each drug can be commercialized with specialty sales force
All discovery and development programs 100% owned
Phase 3 program ALGRX 3268 for the treatment of venipuncture
Statistically significantly less pain in first Phase 3 trial
Second Phase 3 trial data expected within two months
Expect to file NDA in 1H06
Expect product approval in 1H07
Development and commercial expertise in critical areas
Marketing, commercialization, regulatory affairs, manufacturing and clinical trials management
Financial strength and flexibility
8 |
|

ALGRX 3268-
TOPICAL LOCAL ANESTHESIA
Indication
Reduction of pain associated with venipunctures and intravenous line placements
Two Phase 3 trials required for approval
Key desirable product attributes
Sterile, pre-filled, needle-free disposable delivery system (Powderject®)
Rapid onset of action
Within 1 minute with duration of about 10 minutes
No vasoconstriction with good dermal tolerability
Ease of use with no dressings
9
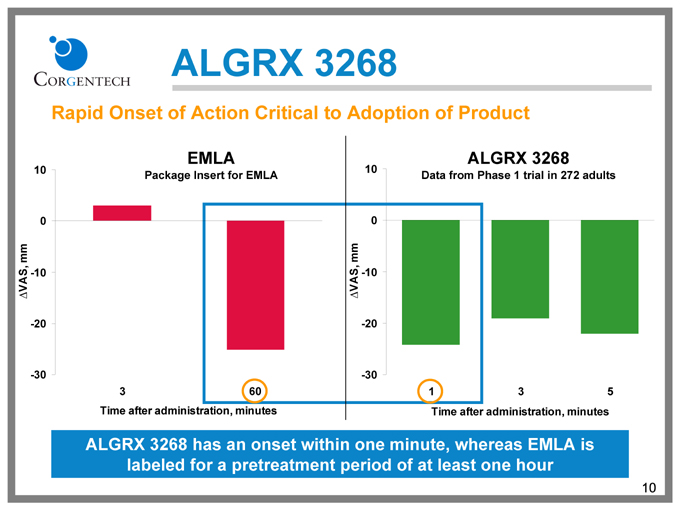
ALGRX 3268
Rapid Onset of Action Critical to Adoption of Product
EMLA
Package Insert for EMLA
ALGRX 3268
Data from Phase 1 trial in 272 adults
10
0
-10
VAS, mm
-20
-30
1 |
|
3 |
|
5 |
|
Time after administration, minutes
10
0
-10
VAS, mm
-20
-30
3 |
|
60
Time after administration, minutes
ALGRX 3268 has an onset within one minute, whereas EMLA is labeled for a pretreatment period of at least one hour
10
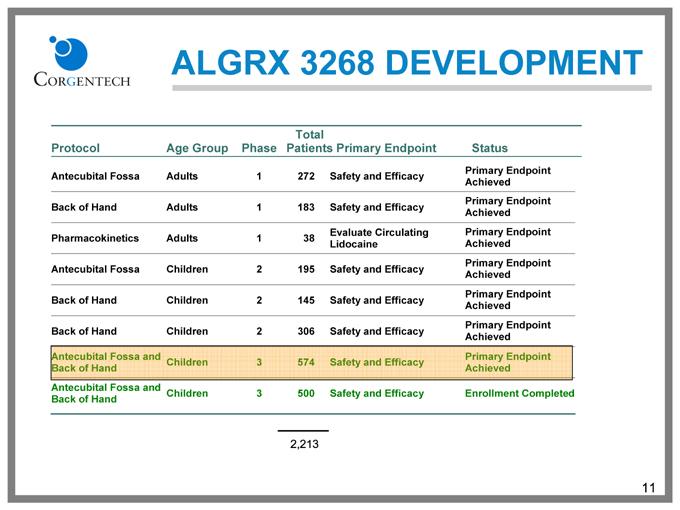
ALGRX 3268 DEVELOPMENT
Protocol Age Group Phase Total Patients Primary Endpoint Status
Antecubital Fossa Adults 1 272 Safety and Efficacy Primary Endpoint Achieved
Back of Hand Adults 1 183 Safety and Efficacy Primary Endpoint Achieved
Pharmacokinetics Adults 1 38 Evaluate Circulating Lidocaine Primary Endpoint Achieved
Antecubital Fossa Children 2 195 Safety and Efficacy Primary Endpoint Achieved
Back of Hand Children 2 145 Safety and Efficacy Primary Endpoint Achieved
Back of Hand Children 2 306 Safety and Efficacy Primary Endpoint Achieved
Antecubital Fossa and Back of Hand Children 3 574 Safety and Efficacy Primary Endpoint Achieved
Antecubital Fossa and Back of Hand Children 3 500 Safety and Efficacy Enrollment Completed
2,213
11

PHASE 3’s: PEDIATRIC TRIALS
IDENTICAL PROTOCOLS
Design
U.S. multicenter, randomized, double-blind, placebo-controlled
Number of subjects
500-600 subjects in each study
Population
Pediatric subjects of either gender (ages 3-18)
Objectives
Confirm efficacy, safety and tolerability of ALGRX 3268 vs. placebo administered on back of hand and antecubital fossa
Primary endpoint
Assessment of pain on venipuncture or peripheral venous cannulation performed 1-3 minutes after ALGRX 3268 or placebo in total population (3-18 yrs.) using FACES rating scale
12
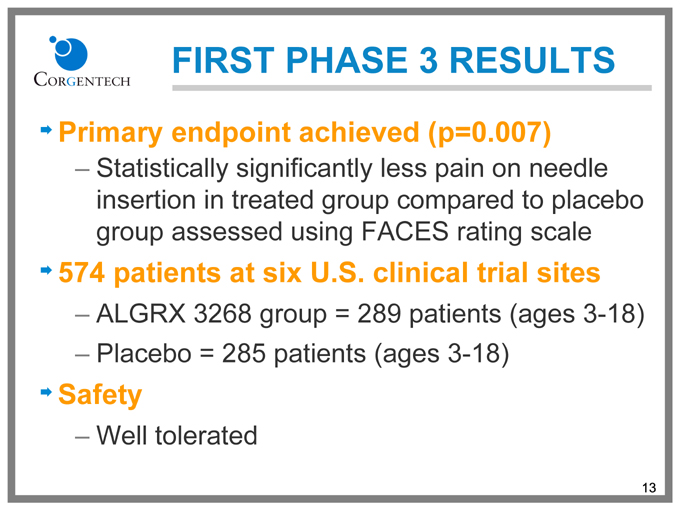
FIRST PHASE 3 RESULTS
Primary endpoint achieved (p=0.007)
Statistically significantly less pain on needle insertion in treated group compared to placebo group assessed using FACES rating scale
574 patients at six U.S. clinical trial sites
ALGRX 3268 group = 289 patients (ages 3-18)
Placebo = 285 patients (ages 3-18)
Safety
Well tolerated
13

PROJECTED TIMELINE FOR ALGRX 3268
Clinical
Data from second Phase 3 trial expected within two months
Regulatory
Reviewed as a drug by the FDA Analgesics and Acute Care Division
Registration
File NDA in 2006
Product approval in 2007
14
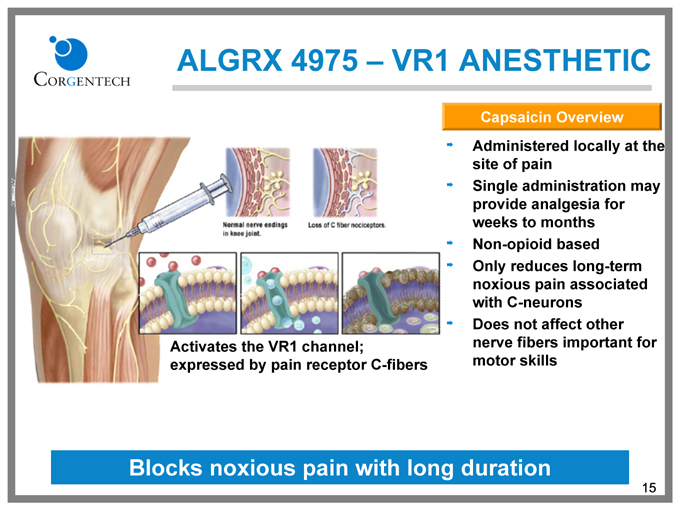
ALGRX 4975 – VR1 ANESTHETIC
Capsaicin Overview
Administered locally at the
site of pain
Single administration may
provide analgesia for
weeks to months
Non-opioid based
Only reduces long-term
noxious pain associated
with C-neurons
Does not affect other
nerve fibers important for
motor skills
Activates the VR1 channel; expressed by pain receptor C-fibers
Blocks noxious pain with long duration
15
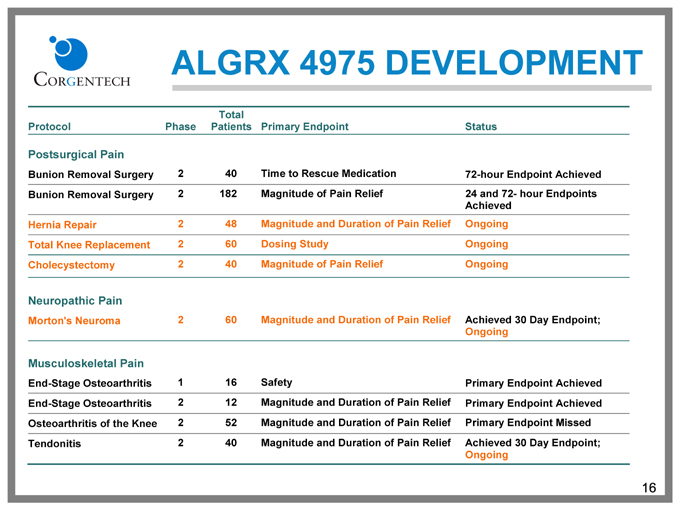
ALGRX 4975 DEVELOPMENT
Total
Protocol Phase Patients Primary Endpoint Status
Postsurgical Pain
Bunion Removal Surgery 2 40 Time to Rescue Medication 72-hour Endpoint Achieved
Bunion Removal Surgery 2 182 Magnitude of Pain Relief 24 and 72- hour Endpoints
Achieved
Hernia Repair 2 48 Magnitude and Duration of Pain Relief Ongoing
Total Knee Replacement 2 60 Dosing Study Ongoing
Cholecystectomy 2 40 Magnitude of Pain Relief Ongoing
Neuropathic Pain
Morton’s Neuroma 2 60 Magnitude and Duration of Pain Relief Achieved 30 Day Endpoint;
Ongoing
Musculoskeletal Pain
End-Stage Osteoarthritis 1 16 Safety Primary Endpoint Achieved
End-Stage Osteoarthritis 2 12 Magnitude and Duration of Pain Relief Primary Endpoint Achieved
Osteoarthritis of the Knee 2 52 Magnitude and Duration of Pain Relief Primary Endpoint Missed
Tendonitis 2 40 Magnitude and Duration of Pain Relief Achieved 30 Day Endpoint;
Ongoing
16
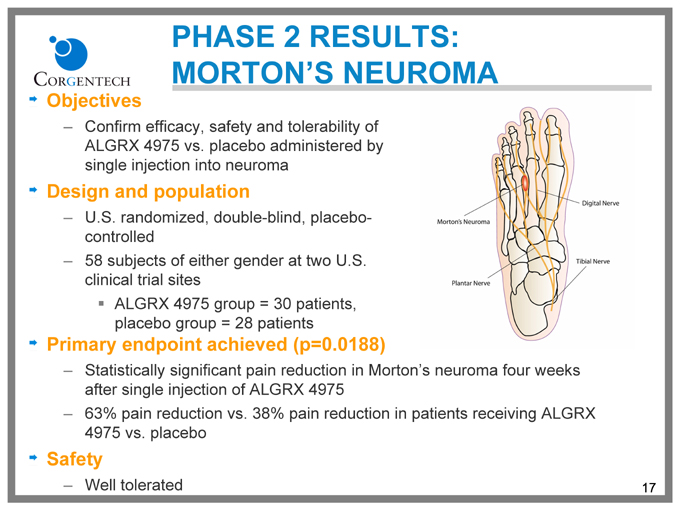
PHASE 2 RESULTS: MORTON’S NEUROMA
Objectives
Confirm efficacy, safety and tolerability of ALGRX 4975 vs. placebo administered by single injection into neuroma
Design and population
U.S. randomized, double-blind, placebo-controlled 58 subjects of either gender at two U.S. clinical trial sites
ALGRX 4975 group = 30 patients, placebo group = 28 patients
Primary endpoint achieved (p=0.0188)
Statistically significant pain reduction in Morton’s neuroma four weeks after single injection of ALGRX 4975 63% pain reduction vs. 38% pain reduction in patients receiving ALGRX 4975 vs. placebo
Safety
Well tolerated
17

PREVALANCE OF PAIN IN SEVEN MAJOR COUNTRIES
PATIENT NUMBERS
Prevalence of post-operative pain 75,068,000
Prevalence of back pain 208,996,000
Prevalence of HIV and AIDS pain 847,000
Prevalence of diabetic neuropathy pain 13,243,000
Prevalence of osteoarthritic pain 46,610,000
Above indications are all targets of Corgentech pipeline
Source: Datamonitor
18

3268: TOPICAL LOCAL
ANESTHESIA MARKET DYNAMICS
Strong focus of pain management including treatment protocols and guidelines Increase in dedicated pediatric hospitals and specialized pediatric departments in larger general hospitals Continuing pressure on cost reduction and patient through-put in hospitals Fundamental market need for easy to use, fast onset topical local anesthetic
Current market dynamics provide an excellent opportunity for a fast acting, easy to use topical local anesthetic product like ALGRX 3268
19

PEDIATRIC IN-HOSPITAL MARKET
Target market
Only 2.1MM out of 18MM venipuncture procedures in children’s hospitals and large academic institutions utilize a topical local anesthetic today
Low utilization of topical local anesthetics (12% of pediatric procedures) due to: slow onset of action lack of promotion of currently available drugs
Faster onset of action, needleless injection and promotion will allow significant penetration and growth into target market of 18MM pediatric procedures
20
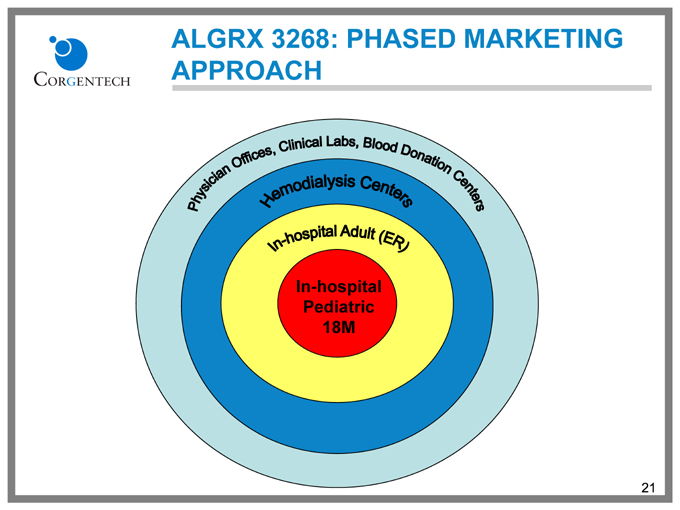
ALGRX 3268: PHASED MARKETING APPROACH
Physician Offices, Clinical Labs, Blood Donation Centers
Hemodialysis Centers
In-hospital Adult (ER)
In-hospital Pediatric 18M
21

ALGRX 4975: POST-OPERATIVE PAIN MARKET
US post-operative pain market: $1.7 billion Virtually all patients experience some level of pain post operatively
50% indicate lack of adequate pain control Patients’ post-surgical pain experiences between 1993 and 1999 have not improved Europe also under treats post-operative pain
Pain in hospital associated with increased length of stay, longer recovery times, poorer patient outcomes
22
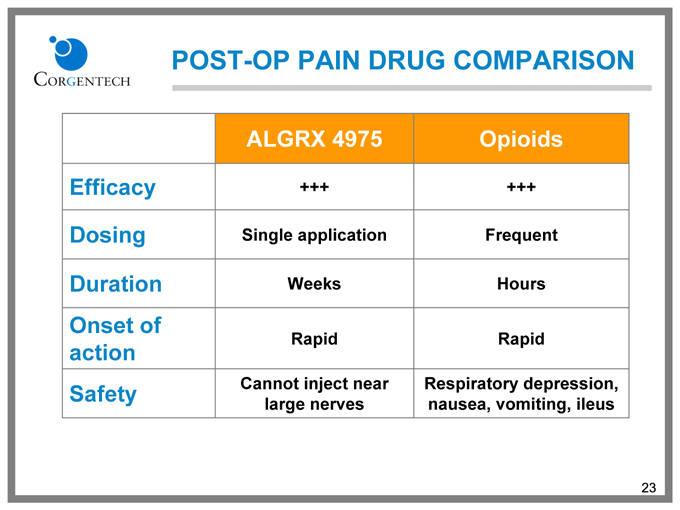
POST-OP PAIN DRUG COMPARISON
ALGRX 4975 Opioids
Efficacy +++ +++
Dosing Single application Frequent
Duration Weeks Hours
Onset of
Rapid Rapid
action
Safety Cannot inject near Respiratory depression,
large nerves nausea, vomiting, ileus
23

WHY THIS MERGER MAKES SENSE
Four product development programs focused on significant clinical problems with attractive market potential
Each drug has unique mechanism of action offering risk diversification Each drug has potential for approval in multiple indications Each drug can be commercialized with specialty sales force All discovery and development programs 100% owned
Phase 3 program ALGRX 3268 for the treatment of venipuncture
Statistically significantly less pain in first Phase 3 trial Second Phase 3 trial data expected within two months Expect to file NDA in 1H06 Expect product approval in 1H07
Development and commercial expertise in critical areas
Marketing, commercialization, regulatory affairs, manufacturing and clinical trials management
Financial strength and flexibility
24