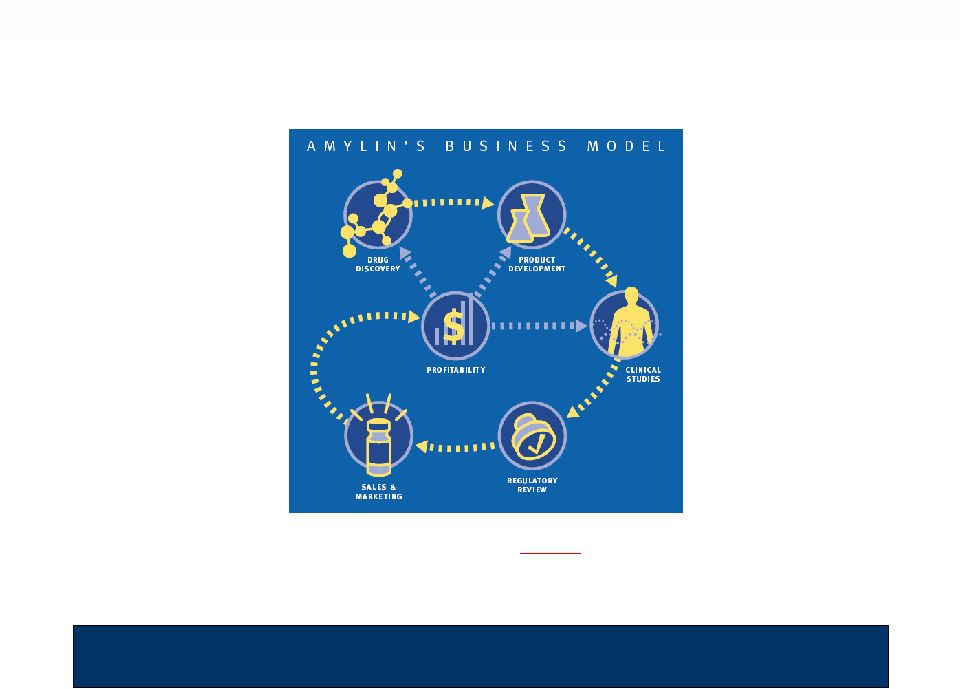
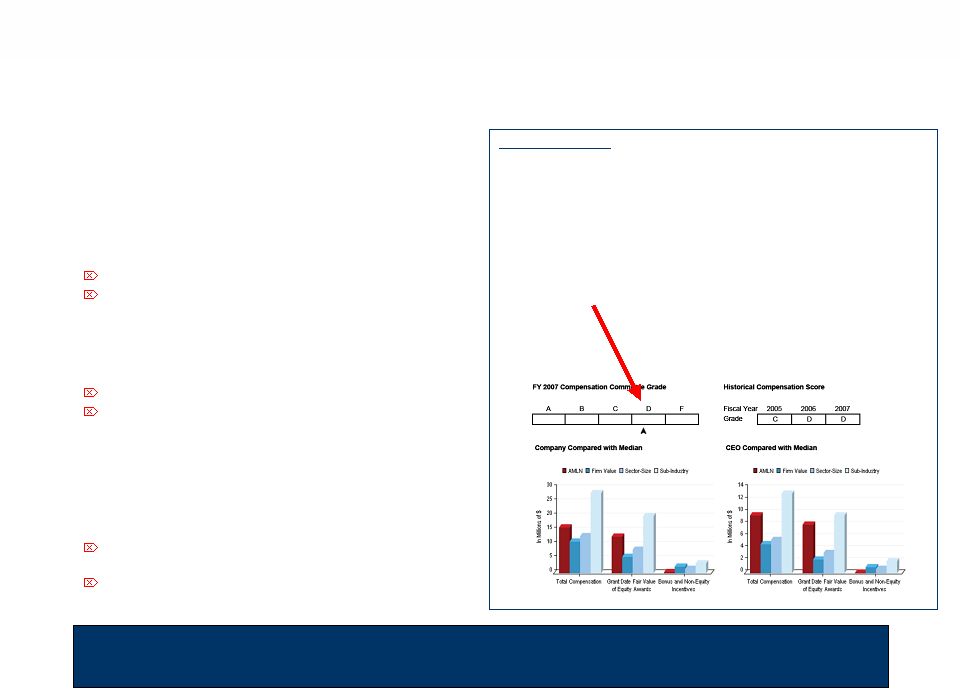
2 Disclaimer This presentation is for general information purposes only. It is not intended to, and does not, address the specific investment objectives, financial situation, suitability or the particular needs of any person who may receive this presentation, and should not be taken as advice on the merits of any investment decision. The views expressed herein represent the opinions of Eastbourne Capital Management, L.L.C., Black Bear Fund I, L.P., Black Bear Fund II, L.L.C., Black Bear Offshore Master Fund, L.P., Richard J. Barry, M. Kathleen Behrens, Charles M. Fleischman and Jay Sherwood (collectively, the “Participants”), and, unless otherwise stated, are based on publicly available information with respect to Amylin Pharmaceuticals, Inc. (the “Company”) or other entities identified herein. Certain matters addressed in this presentation are forward-looking statements that involve certain risks and uncertainties. You should be aware that actual results could differ materially from those contained in the forward-looking statements. The Participants assume no obligation to update any information, including forward-looking information, contained herein. The Participants reserve the right to change any of their intentions or opinions expressed herein at any time as they deem appropriate. The Participants have not sought or obtained consent from any third party to use any statements or information indicated in this presentation as having been obtained or derived from statements made or published by third parties. Any such statements or information should not be viewed as indicating the support of such third party for the views expressed herein. No warranty is made that data or information, whether derived or obtained from filings made with the SEC or from any third party, are accurate. The Participants shall not be responsible or have any liability for any misinformation contained in any SEC filing of the Company or of any third party or in any other third party report. There is no assurance or guarantee with respect to the prices at which any securities of the Company will trade, and such securities may not trade at prices that may be implied in this presentation. This presentation does not recommend the purchase or sale of any security. Under no circumstances is this presentation to be used or considered as an offer to sell or a solicitation of an offer to buy any security. The Participants currently own an aggregate of approximately 12.2% of the outstanding common stock of the Company. It is possible that there will be developments in the future that cause one or more of the Participants from time to time to sell all or a portion of their shares or buy additional shares (in each case, in open market or privately negotiated transactions or otherwise). All stockholders of the Company are advised to read the definitive proxy statement, the white proxy card and other documents related to the solicitation of proxies by the Participants from the stockholders of the Company for use at the 2009 annual meeting of stockholders of the Company because they contain important information. The definitive proxy statement and form of proxy will be mailed to stockholders of the issuer and will, along with other relevant documents, be available at no charge on the SEC’s web site at http://www.sec.gov or by contacting MacKenzie Partners, Inc. by telephone collect at (212) 929-5500, toll-free at (800) 322-2885 or by e-mail at amylinproxy@mackenziepartners.com. In addition, the Particpants will provide copies of the definitive proxy statement without charge upon request. Information relating to the Participants is contained in the definitive Schedule 14A filed by the Participants with the SEC on May 4, 2009. |