Table of Contents
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
OF THE SECURITIES EXCHANGE ACT OF 1934
PURSUANT TO RULE 13a-16 OR 15d-16
OF THE SECURITIES EXCHANGE ACT OF 1934
For the month of August 2006
Bayer Aktiengesellschaft
Bayer Corporation*
(Translation of registrant’s name into English)
Bayerwerk, Gebaeude W11
Kaiser-Wilhelm-Allee
51368 Leverkusen
Germany
(Address of principal executive offices)
Kaiser-Wilhelm-Allee
51368 Leverkusen
Germany
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-Fþ Form 40-Fo
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101 (b)(1): N/A
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101 (b)(7): N/A
Indicate by check mark whether, by furnishing the information contained in this form, the registrant is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934.
Yeso Noþ
If “Yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b): N/A
| * | Bayer Corporation is also the name of a wholly-owned subsidiary of the registrant in the United States. |
Table of Contents
| Stockholders’ Newsletter Interim Report as of June 30, 2006 |  | ||
Strong second quarter:
Continuing success for Bayer both strategically and operationally
Table of Contents
Bayer Group Key Data
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | Full Year | ||||||||||||||||||||||||||
| €million | 2005 | 2006 | Change | 2005 | 2006 | Change | 2005 | |||||||||||||||||||||||
Net sales | 6,686 | 7,072 | + 5.8 | % | 13,072 | 14,188 | + 8.5 | % | 25,950 | |||||||||||||||||||||
Change in sales | ||||||||||||||||||||||||||||||
| Volume | 0 | % | + 4 | % | + 1 | % | + 4 | % | 0 | % | ||||||||||||||||||||
| Price | + 11 | % | 0 | % | + 9 | % | + 1 | % | + 8 | % | ||||||||||||||||||||
| Currency | - 1 | % | 0 | % | - 1 | % | + 3 | % | + 1 | % | ||||||||||||||||||||
| Portfolio | + 10 | % | + 2 | % | + 9 | % | + 1 | % | + 9 | % | ||||||||||||||||||||
EBITDA1 | 1,101 | 1,308 | + 18.8 | % | 2,483 | 2,790 | + 12.4 | % | 4,315 | |||||||||||||||||||||
Special items | (106 | ) | (34 | ) | (244 | ) | (162 | ) | (472 | ) | ||||||||||||||||||||
EBITDA before special items | 1,207 | 1,342 | + 11.2 | % | 2,727 | 2,952 | + 8.3 | % | 4,787 | |||||||||||||||||||||
| EBITDA margin before special items | 18.1 | % | 19.0 | % | 20.9 | % | 20.8 | % | 18.4 | % | ||||||||||||||||||||
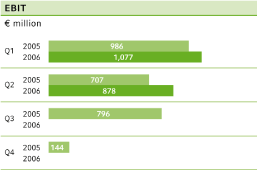
EBIT2 | 707 | 878 | + 24.2 | % | 1,693 | 1,955 | + 15.5 | % | 2,633 | |||||||||||||||||||||
Special items | (106 | ) | (50 | ) | (244 | ) | (178 | ) | (525 | ) | ||||||||||||||||||||
EBIT before special items | 813 | 928 | + 14.1 | % | 1,937 | 2,133 | + 10.1 | % | 3,158 | |||||||||||||||||||||
| EBIT margin before special items | 12.2 | % | 13.1 | % | 14.8 | % | 15.0 | % | 12.2 | % | ||||||||||||||||||||
Non-operating result | (129 | ) | (232 | ) | – 79.8 | % | (260 | ) | (447 | ) | – 71.9 | % | (615 | ) | ||||||||||||||||
Net income | 406 | 452 | + 11.3 | % | 1,058 | 1,052 | – 0.6 | % | 1,597 | |||||||||||||||||||||
Earnings per share (€)3 | 0.56 | 0.60 | 1.45 | 1.41 | 2.19 | |||||||||||||||||||||||||
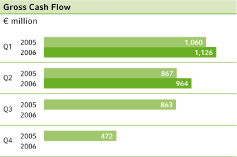
Gross cash flow4 | 867 | 964 | + 11.2 | % | 1,927 | 2,090 | + 8.5 | % | 3,262 | |||||||||||||||||||||
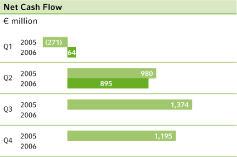
Net cash flow5 | 980 | 895 | – 8.7 | % | 709 | 959 | + 35.3 | % | 3,278 | |||||||||||||||||||||
Capital expenditures (total) | 271 | 340 | + 25.5 | % | 452 | 759 | + 67.9 | % | 1,389 | |||||||||||||||||||||
Research and development expenses | 453 | 448 | – 1.1 | % | 846 | 871 | + 3.0 | % | 1,766 | |||||||||||||||||||||
Depreciation and amortization | 394 | 430 | + 9.1 | % | 790 | 835 | + 5.7 | % | 1,682 | |||||||||||||||||||||
Number of employees at end of period6 | 86,500 | 110,200 | + 27.4 | % | 87,100 | |||||||||||||||||||||||||
| Personnel expenses | 1,483 | 1,559 | + 5.1 | % | 2,904 | 3,113 | + 7.2 | % | 5,539 | |||||||||||||||||||||
| 2005 figures restated | ||
| 1 | ebitda =ebit plus amortization of intangible assets and depreciation of property, plant and equipment.ebitda,ebitda before special items andebitda margin are not defined in the International Financial Reporting Standards and should therefore be regarded only as supplementary information. The company considers underlyingebitda to be a more suitable indicator of operating performance since it is not affected by depreciation, amortization, write-downs/write-backs or special items. The company also believes that this indicator gives readers a clearer picture of the results of operations and ensures greater comparability of data over time. The underlyingebitda margin is calculated by dividing underlyingebitda by sales. | |
| 2 | ebit as shown in the income statement | |
| 3 | Earnings per share as defined in IAS 33. For details see page 40. | |
| 4 | Gross cash flow =ebit plus depreciation, amortization and write-downs/write-backs, minus income taxes, minus gains/plus losses on retirements of noncurrent assets, plus/minus changes in pension provisions. The latter item includes the elimination of non-cash components of the operating result. It also contains benefit payments during the period. | |
| 5 | Net cash flow = cash flow from operating activities according to IAS 7 | |
| 6 | Number of employees in full-time equivalents (including trainees) |
Contents
| Group Management Report | 4 | |||
| 4 | ||||
| 7 | ||||
| 8 | ||||
| 9 | ||||
| 10 | ||||
| 14 | ||||
| 18 | ||||
| 20 | ||||
| 21 | ||||
| 24 | ||||
| 25 | ||||
| 26 | ||||
| 30 | ||||
| 31 | ||||
| Consolidated Financial Statements | 32 | |||
| 32 | ||||
| 33 | ||||
| 34 | ||||
| 35 | ||||
| 36 | ||||
| 38 | ||||
| 40 | ||||
| 45 |
Table of Contents
Interim Report as of June 30, 2006
Strong second quarter:
Continuing success for Bayer both strategically and operationally
| • | Second-quarter sales up 6 percent to €7.1 billion | |
| • | EBITDA before special items moves ahead 11 percent to €1.3 billion | |
| • | EBIT before special items climbs 14 percent to €928 million | |
| • | HealthCare strategically strengthened by Schering acquisition | |
| • | Diagnostics divestiture sharpens HealthCare profile | |
| • | Restructuring program launched at CropScience |
Overview of Sales, Earnings and Financial Position
Bayer is building its future on innovation and growth. In light of these objectives, the successful acquisition of Schering AG strengthens our HealthCare business for the long term. The agreed divestiture of the Diagnostics Division is fully in line with our strategy to sharpen the focus of the HealthCare business and concentrate on human and animal medicines and consumer health products. We have included Schering in the consolidated financial statements with effect from June 23, 2006. The Diagnostics Division is reported under discontinued operations. The previous year’s figures have been restated accordingly.
To ensure comparability between reporting periods, the following table provides a reconciliation of Bayer’s sales and earnings data in the previous corporate structure to those in the new structure. Thus the last column includes the Schering data for the period June 23 through June 30, 2006, while the figures for the Diagnostics Division, which is reported as a discontinued operation, have been eliminated.
Bayer Key Data for the Previous and Current Corporate Structures
| Continuing | ||||||||||||||||||||||||||||||||||||
| Bayer excl. | operations incl. | |||||||||||||||||||||||||||||||||||
| Schering, incl. | Schering, excl. | |||||||||||||||||||||||||||||||||||
| € million | Diagnostics | Schering | Diagnostics | Diagnostics | ||||||||||||||||||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Sales | 7,053 | 7,305 | — | 144 | 367 | 377 | 6,686 | 7,072 | ||||||||||||||||||||||||||||
| EBITDA* | 1,179 | 1,334 | — | 20 | 78 | 46 | 1,101 | 1,308 | ||||||||||||||||||||||||||||
| EBITDA before special items | 1,285 | 1,383 | — | 30 | 78 | 71 | 1,207 | 1,342 | ||||||||||||||||||||||||||||
| EBITDA margin before special items | 18.2 | % | 18.9 | % | — | 20.8 | % | 21.3 | % | 18.8 | % | 18.1 | % | 19.0 | % | |||||||||||||||||||||
| EBIT* | 746 | 893 | — | (6 | ) | 39 | 9 | 707 | 878 | |||||||||||||||||||||||||||
| EBIT before special items | 852 | 958 | — | 4 | 39 | 34 | 813 | 928 | ||||||||||||||||||||||||||||
| 1st Half | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Sales | 13,757 | 14,799 | — | 144 | 685 | 755 | 13,072 | 14,188 | ||||||||||||||||||||||||||||
| EBITDA* | 2,616 | 2,886 | — | 20 | 133 | 116 | 2,483 | 2,790 | ||||||||||||||||||||||||||||
| EBITDA before special items | 2,860 | 3,063 | — | 30 | 133 | 141 | 2,727 | 2,952 | ||||||||||||||||||||||||||||
| EBITDA margin before special items | 20.8 | % | 20.7 | % | — | 20.8 | % | 19.4 | % | 18.7 | % | 20.9 | % | 20.8 | % | |||||||||||||||||||||
| EBIT* | 1,750 | 2,001 | — | (6 | ) | 57 | 40 | 1,693 | 1,955 | |||||||||||||||||||||||||||
| EBIT before special items | 1,994 | 2,194 | — | 4 | 57 | 65 | 1,937 | 2,133 | ||||||||||||||||||||||||||||
| * | for definition see Bayer Group Key Data on page 2 | |
4
Table of Contents
Interim Report as of June 30, 2006
 |  |
In the previous corporate structure (excluding Schering, including Diagnostics), we achievedebitda before special items of €1,383 million (+7.6 percent) in the second quarter. The correspondingebit before special items rose by 12.4 percent in the second quarter, to €958 million (Q 2 2005: €852 million).
The following commentary refers to continuing operations in the new Bayer Group structure.
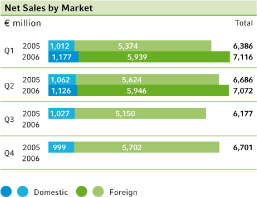
The positive business trend at Bayer continued in thesecond quarter of 2006. Sales from continuing operations advanced by 5.8 percent to €7,072 million (Q 2 2005: €6,686 million) due to higher sales of the HealthCare (+12.7 percent) and MaterialScience (+5.4 percent) subgroups. Sales of CropScience were slightly below the prior-year quarter (–1.6 percent). Group sales included €144 million in revenues from the Schering business for the period June 23 through June 30, 2006. Adjusted for currency and portfolio effects, sales of the Bayer Group rose by 3.6 percent.
The pleasing trend in sales enabled us to further improve our operating performance.ebitda before special items rose by 11.2 percent €1,342 million (Q 2 2005:€1,207 million). Underlyingebitda of the Bayer HealthCare subgroup advanced substantially to €470 million
(+ 27.4 percent) thanks to strong performances by both Pharmaceuticals and Consumer Health. This figure contains €30 million from the Schering business in the period from June 23 through June 30, 2006.ebitda of the Bayer CropScience subgroup before special items grew by 11.2 percent, due mainly to pleasing growth in sales in the Environmental Science, BioScience segment. The 3.2 percent rise in underlyingebitda at Bayer MaterialScience was chiefly attributable to the Polyurethanes business.
(+ 27.4 percent) thanks to strong performances by both Pharmaceuticals and Consumer Health. This figure contains €30 million from the Schering business in the period from June 23 through June 30, 2006.ebitda of the Bayer CropScience subgroup before special items grew by 11.2 percent, due mainly to pleasing growth in sales in the Environmental Science, BioScience segment. The 3.2 percent rise in underlyingebitda at Bayer MaterialScience was chiefly attributable to the Polyurethanes business.
ebit before special items climbed by 14.1 percent in the second quarter, to €928 million (Q 2 2005: €813 million).
Special items in continuing operations totaled €50 million, including a further €20 million in expenses related to antitrust litigation and €16 million in connection with a write-down on the battery business of H.C. Starck. After special items,ebit for the second quarter of 2006 climbed by 24.2 percent to €878 million (Q2 2005: €707 million), whileebitda advanced by 18.8 percent to €1,308 million.
After a non-operating result of minus €232 million, pre-tax income improved by 11.8 percent to €646 million. The non-operating result included net interest expense of €129 million (Q 2 2005 of : €80 million).
5
Table of Contents
Interim Report as of June 30, 2006
 |  |
After tax expense of €197 million, income after taxes from continuing operations came at €449 million (Q2 2005: €410 million). Group net income after minority interests amounted to €452 million (Q2 2005: €406 million).
Benefiting from the positive business trend, gross cash flow improved 11.2 percent to €964 million (Q2 2005: €867 million), while net cash flow from continuing operations came in €85 million below the prior-year quarter, at €895 million, due to an, increase in working capital.
Net debt grew from €5.7 billion on March from 31 , 2006 to €19. 9 billion on June 30, 2006, the €14.2 billion increase being mainly due to the Schering acquisition.
Provisions for pensions and other post-employment benefits in continuing operations remained level compared to March 31, 2006, at €6.2 billion. Provisions of €0.4 billion taken over from Schering were offset by a decline of the same magnitude due to a further increase in capital market interest.
The Bayer Group’s operating performance in thefirst half of 2006also improved compared to the corresponding period of the previous year. Sales from continuing operations grew by 8.5 percent to €14,188 million.ebitda before special items for the first six months of the year increased by 8.3 percent to €2,952 million, compared to €2,727 million for first half of 2005.ebit before special items advanced by 10.1 percent in the same period, to €2,133 million (H1 2005: €1,937 million). After special items,ebit showed an even bigger improvement, rising by 15.5 percent to €1,955 million.
6
Table of Contents
Interim Report as of June 30, 2006
Outlook
The targets, stated in our interim report as of March 31, 2006, of a slight increase in underlyingebit andebitda and an underlyingebitda margin of 19 percent for the full year 2006, applied to the corporate structure existing at that time, which included the Diagnostics business to be divested, but not the Schering acquisition. For this structure we can fully confirm the previous guidance.
If the Diagnostics business to be divested is eliminated, we achieved underlyingebit of €3,158 million and underlyingebitda of €4,787 million in 2005. We anticipate improving on these figures in 2006 even without the inclusion of Schering. We expect earnings from the acquired Schering business to increase Groupebitda before special items for the second half of 2006 by about €600 million, before non-cash charges arising from the step-up of Schering inventories for the first-time consolidation. This adjustment ensures comparability with future periods.
We are raising the earnings target for our continuing HealthCare activities (excluding Schering) as a result of the upward business trend. We now expect to grow underlyingebit by about 20 percent (previously more than 10 percent) and increase the underlyingebitda margin to approximately 20 percent.
For the second half of the year, the Bayer CropScience subgroup is anticipating a negative market environment for the global crop protection industry. Against a background of dry weather in Europe, North America and Australia and the continuing difficulties in the Brazilian farming industry,Bayer CropScience now predicts a decline in sales for 2006 as a whole. Due to the difficult market condition, we now assume that we will be unable to match the previous year’s underlyingebitda margin. We have initiated further restructuring measures in this subgroup to enhance its earning power for the long term. Further details are given under “Subsequent Events” on page 30.
We currently view the market environment for our MaterialScience business as positive despite a significant rise in raw material costs. Business so far in 2006 and the outlook for for the second half of the year are ahead of expectations. Against this background, we now plan to achieve underlyingebit andebitda for the full year on a par with the outstanding 2005 level.
Particularly in light of the long-term optimization of our portfolio, we remain optimistic about the Bayer Group’s future development.
7
Table of Contents
Interim Report as of June 30, 2006
Changes in Corporate Structure
Acquisition of Schering AG
The acquisition of Schering — Bayer’s biggest-ever transaction — is entirely consistent with our strategy of substantially strengthening the HealthCare business as the primary growth engine of the Bayer Group. With a future share of nearly 50 percent of the total portfolio, Bayer HealthCare will be by far our largest subgroup. The acquisition has combined two successful companies into an even more powerful enterprise that is a leading international supplier of specialty pharmaceuticals.
The E.U. Commission and the U.S. antitrust authorities have unconditionally approved the transaction. As of June 23, 2006, we held 88 percent of the outstanding Schering shares. For the period June 23 through June 30, 2006, Schering thus had to be included in the consolidated financial statements.
As of June 30, 2006, our holding in Schering AG based on outstanding shares amounted to just under 90 percent. On July 12, 2006, we announced the successful completion of our public takeover offer for Schering AG. On that date we controlled 92.4 percent of the outstanding voting rights of Schering AG.
The resolution concerning the domination and profit and loss transfer agreement between Schering AG and Dritte BV GmbH, a wholly owned subsidiary of Bayer AG, is scheduled to be resolved on at the Extraordinary Stockholders’ Meeting of Schering AG on September 13, 2006. The agreement contains a cash compensation offer of €89.00 per share with the alternative of an annual guaranteed dividend of €3.62 per share based on Schering’s corporate value.
Divestiture of the Diagnostics Division
We have concluded an agreement with Siemens AG concerning the sale of the Diagnostics Division for €4.2 billion. Subject to the approval of the antitrust authorities, closing of the transaction is expected in the first half of 2007. The Diagnostics Division is therefore reported in the financial statements of the Bayer Group as a discontinued operation. The commentaries in this report relate exclusively to continuing operations, except where specific reference is made to discontinued operations. The 2005 data are restated accordingly.
Planned divestiture of H.C. Starck and Wolff Walsrode
H.C. Starck and Wolff Walsrode are currently still reflected in continuing operations reporting. According to the progress made in the divestiture process, they will subsequently be reported as discontinued operations.
8
Table of Contents
Interim Report as of June 30, 2006
Performance by Subgroup and Segment
Our business activities are grouped in the HealthCare, CropScience und MaterialScience subgroups. We have adjusted our segment reporting for the second quarter to reflect the new corporate structure resulting from the acquisition of Schering and the divestiture of the Diagnostics Division. The Diabetes Care Division is now combined with the former Consumer Care and Animal Health segments in a new segment called Consumer Health, while the acquired Schering business forms part of the Pharmaceuticals segment. These changes are explained in more detail on page 43 in the notes to the financial statements.
€ million
| Proportion of | Proportion of | ||||||||||||||||
| Sales by Subgroup and Segment | 1st Half | Group Sales | 1st Half | Group Sales | |||||||||||||
| 2005 | % | 2006 | % | ||||||||||||||
HealthCare | 3,820 | 29 | 4,460 | 31 | |||||||||||||
| Pharmaceuticals | 1,940 | 15 | 2,336 | 16 | |||||||||||||
| Consumer Health | 1,880 | 14 | 2,124 | 15 | |||||||||||||
CropScience | 3,348 | 26 | 3,349 | 24 | |||||||||||||
| Crop Protection | 2,735 | 21 | 2,682 | 19 | |||||||||||||
| Environmental Science, BioScience | 613 | 5 | 667 | 5 | |||||||||||||
MaterialScience | 5,278 | 40 | 5,694 | 40 | |||||||||||||
| Materials | 1,968 | 15 | 2,094 | 15 | |||||||||||||
| Systems | 3,310 | 25 | 3,600 | 25 | |||||||||||||
Reconciliation | 626 | 5 | 685 | 5 | |||||||||||||
Bayer Group (continuing operations) | 13,072 | 100 | 14,188 | 100 | |||||||||||||
2005 figures restated
9
Table of Contents
Interim Report as of June 30, 2006
Bayer HealthCare
To ensure comparability between reporting periods, the following table provides a reconciliation of Bayer HealthCare’s sales and earnings data in the previous corporate structure to those in the new structure. Thus the last column includes the Schering data for the period June 23 through June 30, 2006, while the figures for the Dignostics Division, which is reported as a discontinued operation, have been eliminated.
In the second quarter of 2006,ebitda before special items for the previous corporate structure amounted € 511 million (+ to 14.3 percent). The correspondingebit before special items rose by 18.3 percent €401 million.
The following commentary refers to continuing operations in the new structure.
Second-quartersalesof theBayer HealthCare subgroup rose by 12.7 percent, or €254 million, from the prior-year quarter, to €2,257 million. This figures contains an amount of €144 million from the acquired Schering activities. Currency- and portfolio-adjusted sales were up by 6.7 percent. This growth was driven by the gratifying business trend in all divisions, particularly in North America.
ebitdabefore special items in the second quarter advanced by €101 million, or 27.4 percent, to €470 million, including €30 million from the acquired Schering business. Both Pharmaceuticals and Consumer Health contributed to this earnings growth.ebitbefore special items improved accordingly by 23.7 percent to €371 million. After special items,ebit increased by 62.1 percent to €355 million, the previous year’s figure having been diminished by litigation-related expenses.
Bayer HealthCare Key Data for the Previous and Current Corporate Structures
| € million | Bayer HealthCare | Continuing | ||||||||||||||||||||||||||||||||||
| excl. Schering, | operations incl. | |||||||||||||||||||||||||||||||||||
| incl. | Schering, excl. | |||||||||||||||||||||||||||||||||||
| Diagnostics | Schering | Diagnostics | Diagnostics | |||||||||||||||||||||||||||||||||
2nd Quarter | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Sales | 2,370 | 2,490 | – | 144 | 367 | 377 | 2,003 | 2,257 | ||||||||||||||||||||||||||||
| EBITDA* | 366 | 480 | – | 20 | 78 | 46 | 288 | 454 | ||||||||||||||||||||||||||||
| EBITDA before special items | 447 | �� | 511 | – | 30 | 78 | 71 | 369 | 470 | |||||||||||||||||||||||||||
| EBITDA margin before special items | 18.9 | % | 20.5 | % | – | 20.8 | % | 21.3 | % | 18.8 | % | 18.4 | % | 20.8 | % | |||||||||||||||||||||
| EBIT* | 258 | 370 | – | (6 | ) | 39 | 9 | 219 | 355 | |||||||||||||||||||||||||||
| EBIT before special items | 339 | 401 | – | 4 | 39 | 34 | 300 | 371 | ||||||||||||||||||||||||||||
1st Half | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Sales | 4,505 | 5,071 | – | 144 | 685 | 755 | 3,820 | 4,460 | ||||||||||||||||||||||||||||
| EBITDA* | 668 | 1,009 | – | 20 | 133 | 116 | 535 | 913 | ||||||||||||||||||||||||||||
| EBITDA before specialitems | 868 | 1,046 | – | 30 | 133 | 141 | 735 | 935 | ||||||||||||||||||||||||||||
| EBITDA margin before special items | 19.3 | % | 20.6 | % | – | 20.8 | % | 19.4 | % | 18.7 | % | 19.2 | % | 21.0 | % | |||||||||||||||||||||
| EBIT* | 441 | 780 | – | (6 | ) | 57 | 40 | 384 | 734 | |||||||||||||||||||||||||||
| EBIT before special items | 641 | 817 | – | 4 | 57 | 65 | 584 | 756 | ||||||||||||||||||||||||||||
| * | for definition see Bayer Group Key Data on page 2 |
10
Table of Contents
Interim Report as of June 30, 2006
| Bayer HealthCare | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 2,003 | 2,257 | + 12.7 | 3,820 | 4,460 | + 16.8 | ||||||||||||||||||||
EBITDA* | 288 | 454 | + 57.6 | 535 | 913 | + 70.7 | ||||||||||||||||||||
Special items | (81 | ) | (16 | ) | (200 | ) | (22 | ) | ||||||||||||||||||
EBITDA before special items | 369 | 470 | + 27.4 | 735 | 935 | + 27.2 | ||||||||||||||||||||
| EBITDA margin before special items | 18.4 | % | 20.8 | % | 19.2 | % | 21.0 | % | ||||||||||||||||||
EBIT* | 219 | 355 | + 62.1 | 384 | 734 | + 91.1 | ||||||||||||||||||||
Special items | (81 | ) | (16 | ) | (200 | ) | (22 | ) | ||||||||||||||||||
EBIT before special items | 300 | 371 | + 23.7 | 584 | 756 | + 29.5 | ||||||||||||||||||||
Gross cash flow* | 217 | 336 | + 54.8 | 378 | 628 | + 66.1 | ||||||||||||||||||||
Net cash flow* | 186 | 367 | + 97.3 | 208 | 410 | + 97.1 | ||||||||||||||||||||
| 2005 figure restated | ||
| * for definition see Bayer Group Key Data on page 2 | ||
| Best-Selling Bayer HealthCare Products | ||||||||||||||||||||||||||
Ascensia® product line (Diabetes Care) | 191 | 208 | + 8.9 | 331 | 398 | + 20.2 | ||||||||||||||||||||
Kogenate® (Pharmaceuticals) | 174 | 179 | + 2.9 | 299 | 383 | + 28.1 | ||||||||||||||||||||
Aspirin® (Consumer Care/Pharmaceuticals) | 157 | 168 | + 7.0 | 297 | 332 | + 11.8 | ||||||||||||||||||||
Adalat® (Pharmaceuticals) | 167 | 171 | + 2.4 | 320 | 328 | + 2.5 | ||||||||||||||||||||
Ciprobay® /Cipro® (Pharmaceuticals) | 114 | 127 | + 11.4 | 272 | 259 | - 4.8 | ||||||||||||||||||||
Avalox® /Avelox® (Pharmaceuticals) | 78 | 88 | + 12.8 | 181 | 218 | + 20.4 | ||||||||||||||||||||
Glucobay® (Pharmaceuticals) | 75 | 76 | + 1.3 | 146 | 153 | + 4.8 | ||||||||||||||||||||
Levitra® (Pharmaceuticals) | 63 | 73 | + 15.9 | 123 | 151 | + 22.8 | ||||||||||||||||||||
Advantage® /Advantix® (Animal Health) | 77 | 91 | + 18.2 | 131 | 150 | + 14.5 | ||||||||||||||||||||
Aleve® /naproxen (Consumer Care) | 45 | 56 | + 24.4 | 73 | 109 | + 49.3 | ||||||||||||||||||||
Canesten® (Consumer Care) | 37 | 40 | + 8.1 | 70 | 81 | + 15.7 | ||||||||||||||||||||
Baytril® (Animal Health) | 33 | 35 | + 6.1 | 73 | 75 | + 2.7 | ||||||||||||||||||||
Trasylol® (Pharmaceuticals) | 56 | 35 | - 37.5 | 101 | 75 | - 25.7 | ||||||||||||||||||||
Bepanthen® /Bepanthol® (Consumer Care) | 32 | 34 | + 6.3 | 60 | 69 | + 15.0 | ||||||||||||||||||||
Supradyn® (Consumer Care) | 35 | 31 | - 11.4 | 64 | 66 | + 3.1 | ||||||||||||||||||||
Total | 1,334 | 1,412 | + 5.8 | 2,541 | 2,847 | + 12.0 | ||||||||||||||||||||
| Proportion of Bayer HealthCare sales | 67 | % | 63 | % | 67 | % | 64 | % | ||||||||||||||||||
11
Table of Contents
Interim Report as of June 30, 2006
Pharmaceuticals
Salesof thePharmaceuticalssegment climbed by 20.2 percent in the second quarter, to €1,188 million. This figure contains €144 million in revenues from the acquired Schering activities between June 23 and June 30, 2006. Adjusted for currency and portfolio effects, sales were up by 9.0 percent. Expansion in the Primary Care and Oncology businesses more than offset a decline in sales of the Hematology/Cardiology business unit.
Sales of the Primary Care business unit moved ahead by 10.1 percent to €751 million, due mainly to the pleasing performance of our top products, particularly Avelox® and Levitra®. Sales of the Hematology/Cardiology business unit shrank by 15.4 percent as a result of the termination of our Plasma distribution in Canada and lower sales of Trasylol®. The long-term studies available to us and our experience with Trasylol®, our product for use in open heart surgery, indicate that it is a safe and effective medication when used correctly. The two studies reporting a possible link between the use of Trasylol® and severe renal dysfunction and vasoconstriction (myocardial infarction and stroke) are currently being evaluated by the regulatory authorities. Sales of Kogenate® posted a further slight improvement over the high level of the prior-year quarter.
Sales of the Oncology business unit rose by €33 million to €41 million, thanks mainly to the progressive market introduction of our new cancer drug Nexavar®. In July we received authorization to market Nexavar® in Europe and Canada for the treatment of advanced renal cell carcinoma.
ebitdaof the Pharmaceuticals segment before special items improved by €72 million, or 43.6 percent, to €237 million, including €30 million from the acquired Schering business. The positive sales trend enabled us to achieve this substantial increase despite higher marketing costs for the introduction of Nexavar®. Underlyingebit also moved ahead significantly, growing by 34.1 percent, or €44 million, to €173 million. After special items,ebit rose by 45.9 percent to €159 million.
Consumer Health
Salesof theConsumer Healthsegment increased by 5.3 percent to €1,069 million, or by 4.6 percent when adjusted for currency translation.
In the Consumer Care Division, sales advanced by 2.0 percent due mainly to contributions from our top products.
Business in the Diabetes Care Division grew by 9.8 percent, thanks to sharply higher sales of our Ascensia® Contour® blood glucose measurement system in Europe and North America.
In the Animal Health Division, there was a pleasing €23 million increase in second-quarter sales, to €252 million (+10.0 percent). Here we benefited primarily from strong growth in the Advantage® product family in North America.
ebitdaof the Consumer Health segment before special items increased by 14.2 percent in the second quarter, to €233 million (Q2 2005: €204 million). Underlyingebit grew by 15.8 percent to €198 million, whileebitafter special items, at €196 million, was up by 78.2 percent year on year. The previous year’s figure was affected by special charges related to litigation and to the integration of the consumer health business acquired from Roche.
12
Table of Contents
Interim Report as of June 30, 2006
| Pharmaceuticals | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 988 | 1,188 | + 20.2 | 1,940 | 2,336 | + 20.4 | ||||||||||||||||||||
| Primary Care | 682 | 751 | + 10.1 | 1,400 | 1,538 | + 9.9 | ||||||||||||||||||||
| Hematology/Cardiology | 298 | 252 | -15.4 | 529 | 579 | + 9.5 | ||||||||||||||||||||
| Oncology | 8 | 41 | • | 11 | 75 | • | ||||||||||||||||||||
| Schering | 0 | 144 | • | 0 | 144 | • | ||||||||||||||||||||
EBITDA* | 145 | 223 | + 53.8 | 272 | 464 | + 70.6 | ||||||||||||||||||||
Special items | (20 | ) | (14 | ) | (118 | ) | (19 | ) | ||||||||||||||||||
EBITDA before special items | 165 | 237 | + 43.6 | 390 | 483 | + 23.8 | ||||||||||||||||||||
| EBITDA margin before special items | 16.7 | % | 19.9 | % | 20.1 | % | 20.7 | % | ||||||||||||||||||
EBIT* | 109 | 159 | + 45.9 | 195 | 361 | + 85.1 | ||||||||||||||||||||
Special items | (20 | ) | (14 | ) | (118 | ) | (19 | ) | ||||||||||||||||||
EBIT before special items | 129 | 173 | + 34.1 | 313 | 380 | + 21.4 | ||||||||||||||||||||
Gross cash flow* | 106 | 157 | + 48.1 | 180 | 319 | + 77.2 | ||||||||||||||||||||
Net cash flow* | 143 | 284 | + 98.6 | 51 | 273 | • | ||||||||||||||||||||
Consumer Health | ||||||||||||||||||||||||||
Net sales | 1,015 | 1,069 | + 5.3 | 1,880 | 2,124 | + 13.0 | ||||||||||||||||||||
| Consumer Care | 592 | 604 | + 2.0 | 1,115 | 1,246 | + 11.7 | ||||||||||||||||||||
| Diabetes Care | 194 | 213 | + 9.8 | 337 | 406 | + 20.5 | ||||||||||||||||||||
| Animal Health | 229 | 252 | + 10.0 | 428 | 472 | + 10.3 | ||||||||||||||||||||
EBITDA* | 143 | 231 | + 61.5 | 263 | 449 | + 70.7 | ||||||||||||||||||||
Special items | (61 | ) | (2 | ) | (82 | ) | (3 | ) | ||||||||||||||||||
EBITDA before special items | 204 | 233 | + 14.2 | 345 | 452 | + 31.0 | ||||||||||||||||||||
| �� | ||||||||||||||||||||||||||
| EBITDA margin before special items | 20.1 | % | 21.8 | % | 18.4 | % | 21.3 | % | ||||||||||||||||||
EBIT* | 110 | 196 | + 78.2 | 189 | 373 | + 97.4 | ||||||||||||||||||||
Special items | (61 | ) | (2 | ) | (82 | ) | (3 | ) | ||||||||||||||||||
EBIT before special items | 171 | 198 | + 15.8 | 271 | 376 | + 38.7 | ||||||||||||||||||||
Gross cash flow* | 111 | 179 | + 61.3 | 198 | 309 | + 56.1 | ||||||||||||||||||||
Net cash flow* | 43 | 83 | + 93.0 | 157 | 137 | – 12.7 | ||||||||||||||||||||
| 2005 figures restated | ||
| * | for definition see Bayer Group Key Data on page 2 |
13
Table of Contents
Interim Report as of June 30, 2006
Bayer CropScience
Second-quartersalesof theBayer CropSciencesubgroup declined by 1.6 percent to€ 1,578 million, or by 1.9 percent when adjusted for the effects of currency translation and portfolio changes.
ebitdabefore special items posted a year-on-year improvement of€ 37 million, or 11.2 percent, to€ 368 million. Underlyingebit climbed by 23.0 percent to€ 230 million, whileebit after special items expanded by 42.0 percent.
| Bayer CropScience | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 1,604 | 1,578 | – 1.6 | 3,348 | 3,349 | 0.0 | ||||||||||||||||||||
EBITDA* | 306 | 368 | + 20.3 | 863 | 919 | + 6.5 | ||||||||||||||||||||
Special items | (25 | ) | 0 | (34 | ) | 0 | ||||||||||||||||||||
EBITDA before special items | 331 | 368 | + 11.2 | 897 | 919 | + 2.5 | ||||||||||||||||||||
| EBITDA margin before special items | 20.6 | % | 23.3 | % | 26.8 | % | 27.4 | % | ||||||||||||||||||
EBIT* | 162 | 230 | + 42.0 | 576 | 638 | + 10.8 | ||||||||||||||||||||
Special items | (25 | ) | 0 | (34 | ) | 0 | ||||||||||||||||||||
EBIT before special items | 187 | 230 | + 23.0 | 610 | 638 | + 4.6 | ||||||||||||||||||||
Gross cash flow* | 231 | 289 | + 25.1 | 618 | 676 | + 9.4 | ||||||||||||||||||||
Net cash flow* | 613 | 534 | – 12.9 | 234 | 184 | – 21.4 | ||||||||||||||||||||
| * for definition see Bayer Group Key Data on | page 2 | |||||||||||||||||||||||||
| Best-Selling Bayer CropScience | ||||||||||||||||||||||||||
| Products* | ||||||||||||||||||||||||||
Confidor® /Gaucho® /Admire® /Merit® (Insecticides/Seed Treatment/ Environmental Science) | 154 | 147 | – 4.5 | 325 | 312 | – 4.0 | ||||||||||||||||||||
Folicur® /Raxil® (Fungicides/Seed Treatment) | 86 | 72 | – 16.3 | 183 | 167 | – 8.7 | ||||||||||||||||||||
Basta® /Liberty® (Herbicides) | 79 | 80 | + 1.3 | 138 | 152 | + 10.1 | ||||||||||||||||||||
Puma® (Herbicides) | 73 | 74 | + 1.4 | 140 | 142 | + 1.4 | ||||||||||||||||||||
Proline® (Fungicides) | 50 | 55 | + 10.0 | 86 | 113 | + 31.4 | ||||||||||||||||||||
Betanal® (Herbicides) | 52 | 57 | + 9.6 | 104 | 102 | – 1.9 | ||||||||||||||||||||
Decis® /K-Othrine® (Insecticides/ Environmental Science) | 47 | 56 | + 19.1 | 85 | 100 | + 17.6 | ||||||||||||||||||||
Flint® /Stratego® /Sphere® (Fungicides) | 38 | 38 | 0.0 | 87 | 87 | 0.0 | ||||||||||||||||||||
Atlantis® (Herbicides) | 16 | 21 | + 31.3 | 58 | 70 | + 20.7 | ||||||||||||||||||||
Temik® (Insecticides) | 21 | 21 | 0.0 | 61 | 65 | + 6.6 | ||||||||||||||||||||
Total | 616 | 621 | + 0.8 | 1,267 | 1,310 | + 3.4 | ||||||||||||||||||||
| Proportion of Bayer CropScience | 38 | % | 39 | % | 38 | % | 39 | % | ||||||||||||||||||
| * Figures are based on active ingredient class. For the sake of clarity, only the principal brands and business units are listed. | ||
14
Table of Contents
Interim Report as of June 30, 2006
Crop Protection
TheCrop Protectionsegment achieved second-quartersalesof€ 1,269 million, down 3.7 percent from€ 1,318 million in the same period of 2005. Currency- and portfolio-adjusted sales fell by 4.0 percent.
While sales of the Herbicides and Seed Treatment units came in at around the prior-year level, business in Insecticides and Fungicides declined.
The lower insecticide sales were mainly due to the divestiture of some of our older active ingredients in 2005 as part of our ongoing efforts to streamline the portfolio. The continuing difficult market conditions in Brazil had a further negative impact. By contrast, business in China showed encouraging growth.
The decline in fungicide sales in the second quarter resulted in part from the early start to the fungicide season in the United States, which had already led to strong demand in the first quarter, and in part from lower rates of fungal infestation in many parts of Europe due to the dry weather.
| Crop Protection | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 1,318 | 1,269 | – 3.7 | 2,735 | 2,682 | – 1.9 | ||||||||||||||||||||
| Insecticides | 344 | 317 | – 7.8 | 708 | 665 | – 6.1 | ||||||||||||||||||||
| Fungicides | 369 | 352 | – 4.6 | 716 | 730 | + 2.0 | ||||||||||||||||||||
| Herbicides | 524 | 519 | – 1.0 | 1,079 | 1,069 | – 0.9 | ||||||||||||||||||||
| Seed Treatment | 81 | 81 | 0.0 | 232 | 218 | – 6.0 | ||||||||||||||||||||
EBITDA* | 235 | 277 | + 17.9 | 678 | 683 | + 0.7 | ||||||||||||||||||||
Special items | (21 | ) | 0 | (30 | ) | 0 | ||||||||||||||||||||
EBITDA before special items | 256 | 277 | + 8.2 | 708 | 683 | – 3.5 | ||||||||||||||||||||
| EBITDA margin before special items | 19.4 | % | 21.8 | % | 25.9 | % | 25.5 | % | ||||||||||||||||||
EBIT* | 110 | 159 | + 44.5 | 432 | 444 | + 2.8 | ||||||||||||||||||||
Special items | (21 | ) | 0 | (30 | ) | 0 | ||||||||||||||||||||
EBIT before special items | 131 | 159 | + 21.4 | 462 | 444 | – 3.9 | ||||||||||||||||||||
Gross cash flow* | 182 | 227 | + 24.7 | 489 | 512 | + 4.7 | ||||||||||||||||||||
Net cash flow* | 493 | 434 | – 12.0 | 170 | 145 | – 14.7 | ||||||||||||||||||||
| * for definition see Bayer Group Key Data on page 2 | ||
15
Table of Contents
Interim Report as of June 30, 2006
While sales of our herbicides declined in North America and Australia, also for weather-related reasons, this effect was largely offset by good business with herbicides for cereal crops in northern Europe and for sugar beet in eastern Europe.
Despite difficult market conditions in some cases, business with our best-selling products rose slightly in the second quarter. Sales of recently launched products also developed positively, with the cereal fungicides Proline® and Fandango®, the herbicides Atlantis® and Olympus®, the insecticides Oberon® and Envidor®, and the seed treatment Poncho®posting particularly strong gains.
Despite the decline in sales in the Crop Protection segment as a whole, second quarterebitda before special items showed year-on-year growth of€21 million, or 8.2 percent, to€277 million. Our costcontainment and efficiency-enhancement programs contributed to this performance. Underlyingebit climbed by 21.4 percent to€159 million, whileebit after special items improved by 44.5 percent.
Environmental Science, BioScience
Second-quarter sales of theEnvironmental Science, BioSciencesegment advanced by 8.0 percent, both in euros and when adjusted for currency translation, to€309 million.
Sales in the Environmental Science rose by 4.2 percent year on year, to€225 million. The main growth drivers were our products for professional users, particularly K-Othrine® and K-O Tab® 1-2-3 for vector control. BioScience increased sales by 20.0 percent overall to€84 million, thanks largely to good business with vegetable seeds and our hybrid seed Arize®.
As a result of the growth in business, underlyingebitdaof the Environmental Science, BioScience segment climbed by 21.3 percent to€91 million.ebitbefore special items rose by 26.8 percent, whileebit after special items advanced by€19 million, or 36.5 percent, to€71 million.
16
Table of Contents
Interim Report as of June 30, 2006
| Environmental Science, BioScience | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 286 | 309 | + 8.0 | 613 | 667 | + 8.8 | ||||||||||||||||||||
| Environmental Science | 216 | 225 | + 4.2 | 390 | 418 | + 7.2 | ||||||||||||||||||||
| BioScience | 70 | 84 | + 20.0 | 223 | 249 | + 11.7 | ||||||||||||||||||||
EBITDA* | 71 | 91 | + 28.2 | 185 | 236 | + 27.6 | ||||||||||||||||||||
Special items | (4 | ) | 0 | (4 | ) | 0 | ||||||||||||||||||||
EBITDA before special items | 75 | 91 | + 21.3 | 189 | 236 | + 24.9 | ||||||||||||||||||||
| EBITDA margin before special items | 26.2 | % | 29.4 | % | 30.8 | % | 35.4 | % | ||||||||||||||||||
EBIT* | 52 | 71 | + 36.5 | 144 | 194 | + 34.7 | ||||||||||||||||||||
Special items | (4 | ) | 0 | (4 | ) | 0 | ||||||||||||||||||||
EBIT before special items | 56 | 71 | + 26.8 | 148 | 194 | + 31.1 | ||||||||||||||||||||
Gross cash flow* | 49 | 62 | + 26.5 | 129 | 164 | + 27.1 | ||||||||||||||||||||
Net cash flow* | 120 | 100 | – 16.7 | 64 | 39 | – 39.1 | ||||||||||||||||||||
| * | for definition see Bayer Group Key Data on page 2 |
17
Table of Contents
Interim Report as of June 30, 2006
Bayer MaterialScience
The positive business trend in theBayer MaterialSciencesubgroup continued in the second quarter of 2006, withsalesincreasing by 5.4 percent to€2,883 million. Adjusted for currency and portfolio effects, sales rose by 4.7 percent. Business growth was mainly volume-driven, with the Polyurethanes and the Coatings, Adhesives, Sealants business units the main contributors.
Underlyingebitdaof the Bayer MaterialScience subgroup came in at€489 million, up€15 million, or 3.2 percent, from the prior-year period. Underlyingebitrose by 4.7 percent to€353 million, while reportedebit dropped by 2.4 percent to€319 million after€34 million in special charges.
Materials
Salesof theMaterialssegment edged up 1.3 percent in the second quarter to€1,059 million, the increase after currency translation being 1.4 percent.
Despite price erosion, sales of our Polycarbonates business unit dipped by only 1.3 percent to€670 million. By contrast, the H.C. Starck business unit boosted sales by 7.9 percent to€247 million due to higher volumes.
Underlyingebitdaof the Materials segment fell by€37 million, or 17.2 percent, to€178 million, due above all to price-related reasons. Underlyingebitfell by 27.2 percent to€118 million, whileebit after special items, at€102 million, was diminished by a€16 million write-down on the battery business of H.C. Starck.
Systems
Second-quartersalesof ourSystemssegment rose by 8.0 percent year on year to€1,824 million. After adjusting for currency and portfolio changes, the increase came to 6.8 percent. This pleasing performance was particularly attributable to the Polyurethanes business unit. The Coatings, Adhesives, Sealants business unit also saw substantially higher sales than in the prior-year quarter due to growth in volumes.
ebitdabefore special items climbed by€52 million, or 20.1 percent, to€311 million, largely thanks to price increases implemented for TDI and polyether products in the Polyurethanes business unit.ebitbefore special items also increased sharply, rising by 34.3 percent to€235 million. After special items,ebit improved by 31.5 percent to€217 million despite further special charges in connection with the antitrust proceedings.
18
Table of Contents
Interim Report as of June 30, 2006
| Bayer MaterialScience | 2nd Quarter | 2nd Quarter | Change | 1st Half | 1st Half | Change | ||||||||||||||||||||
| € million | 2005 | 2006 | % | 2005 | 2006 | % | ||||||||||||||||||||
Net sales | 2,734 | 2,883 | + 5.4 | 5,278 | 5,694 | + 7.9 | ||||||||||||||||||||
EBITDA* | 464 | 471 | + 1.5 | 997 | 944 | – 5.3 | ||||||||||||||||||||
Special items | (10 | ) | (18 | ) | (10 | ) | (130 | ) | ||||||||||||||||||
EBITDA before special items | 474 | 489 | + 3.2 | 1,007 | 1,074 | + 6.7 | ||||||||||||||||||||
| EBITDA margin before special items | 17.3 | % | 17.0 | % | 19.1 | % | 18.9 | % | ||||||||||||||||||
EBIT* | 327 | 319 | – 2.4 | 733 | 658 | – 10.2 | ||||||||||||||||||||
Special items | (10 | ) | (34 | ) | (10 | ) | (146 | ) | ||||||||||||||||||
EBIT before special items | 337 | 353 | + 4.7 | 743 | 804 | + 8.2 | ||||||||||||||||||||
Gross cash flow* | 328 | 363 | + 10.7 | 689 | 717 | + 4.1 | ||||||||||||||||||||
Net cash flow* | 269 | 264 | – 1.9 | 269 | 563 | + 109.3 | ||||||||||||||||||||
Materials | ||||||||||||||||||||||||||
Net sales | 1,045 | 1,059 | + 1.3 | 1,968 | 2,094 | + 6.4 | ||||||||||||||||||||
| Polycarbonates | 679 | 670 | – 1.3 | 1,267 | 1,326 | + 4.7 | ||||||||||||||||||||
| Thermoplastic Polyurethanes | 49 | 53 | + 8.2 | 95 | 107 | + 12.6 | ||||||||||||||||||||
| Wolff Walsrode | 88 | 89 | + 1.1 | 160 | 167 | + 4.4 | ||||||||||||||||||||
| H.C. Starck | 229 | 247 | + 7.9 | 446 | 494 | + 10.8 | ||||||||||||||||||||
EBITDA* | 215 | 178 | – 17.2 | 427 | 394 | – 7.7 | ||||||||||||||||||||
Special items | 0 | 0 | 0 | 0 | ||||||||||||||||||||||
EBITDA before special items | 215 | 178 | – 17.2 | 427 | 394 | – 7.7 | ||||||||||||||||||||
| EBITDA margin before special items | 20.6 | % | 16.8 | % | 21.7 | % | 18.8 | % | ||||||||||||||||||
EBIT* | 162 | 102 | – 37.0 | 321 | 262 | – 18.4 | ||||||||||||||||||||
Special items | 0 | (16 | ) | 0 | (16 | ) | ||||||||||||||||||||
EBIT before special items | 162 | 118 | – 27.2 | 321 | 278 | – 13.4 | ||||||||||||||||||||
Gross cash flow* | 149 | 141 | – 5.4 | 292 | 304 | + 4.1 | ||||||||||||||||||||
Net cash flow* | 80 | 101 | + 26.3 | 144 | 162 | + 12.5 | ||||||||||||||||||||
Systems | ||||||||||||||||||||||||||
Net sales | 1,689 | 1,824 | + 8.0 | 3,310 | 3,600 | + 8.8 | ||||||||||||||||||||
| Polyurethanes | 1,215 | 1,301 | + 7.1 | 2,411 | 2,570 | + 6.6 | ||||||||||||||||||||
| Coatings, Adhesives, Sealants | 342 | 380 | + 11.1 | 662 | 749 | + 13.1 | ||||||||||||||||||||
| Inorganic Basic Chemicals | 102 | 100 | – 2.0 | 189 | 206 | + 9.0 | ||||||||||||||||||||
| Others | 30 | 43 | + 43.3 | 48 | 75 | + 56.3 | ||||||||||||||||||||
EBITDA* | 249 | 293 | + 17.7 | 570 | 550 | – 3.5 | ||||||||||||||||||||
Special items | (10 | ) | (18 | ) | (10 | ) | (130 | ) | ||||||||||||||||||
EBITDA before special items | 259 | 311 | + 20.1 | 580 | 680 | + 17.2 | ||||||||||||||||||||
| EBITDA margin before special items | 15.3 | % | 17.1 | % | 17.5 | % | 18.9 | % | ||||||||||||||||||
EBIT* | 165 | 217 | + 31.5 | 412 | 396 | – 3.9 | ||||||||||||||||||||
Special items | (10 | ) | (18 | ) | (10 | ) | (130 | ) | ||||||||||||||||||
EBIT before special items | 175 | 235 | + 34.3 | 422 | 526 | + 24.6 | ||||||||||||||||||||
Gross cash flow* | 179 | 222 | + 24.0 | 397 | 413 | + 4.0 | ||||||||||||||||||||
Net cash flow* | 189 | 163 | – 13.8 | 125 | 401 | • | ||||||||||||||||||||
| * | for definition see Bayer Group key Data on page 2 |
19
Table of Contents
Interim Report as of June 30, 2006
Performance by Region
Sales of the Bayer Group worldwide grew by€386 million, or 5.8 percent, to€7,072 million in the second quarter of 2006. Adjusted for exchange rate fluctuations, sales rose by 5.3 percent. A major portion of this growth was achieved in theNorth Americaregion, where sales advanced by 7.9 percent (+€140 million). The largest increase here was in our Pharmaceuticals business, with the activities acquired from Schering contributing€42 million in sales. All divisions of our Consumer Health segment also performed well in the major North American markets.
InEurope, which accounts for nearly half of our business, sales grew by 4.0 percent to€3,169 million, with particularly strong gains in the Pharmaceuticals and Systems segments. Sales in Germany rose by 6.0 percent to€1,126 million, but dipped by 0.6 percent year on year when adjusted for portfolio changes. InAsia/Pacificand theLatin America/Africa/Middle Eastregion, sales were up by 4.2 and 10.0 percent, respectively. In these regions too, growth was driven by the Pharmaceuticals and Systems segments. Sales in China advanced by some 22 percent.
| Sales by Region and Segment | Europe | �� | North America | |||||||||||||||||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | 2005 | 2006 | % yoy | adj. % yoy | ||||||||||||||||||||||||||
| € million | ||||||||||||||||||||||||||||||||||
2nd Quarter | ||||||||||||||||||||||||||||||||||
| Pharmaceuticals | 419 | 477 | + 13.8 | + 13.7 | 239 | 314 | + 31.4 | + 28.6 | ||||||||||||||||||||||||||
| Consumer Health | 418 | 422 | + 1.0 | + 1.1 | 333 | 376 | + 12.9 | + 10.9 | ||||||||||||||||||||||||||
| Crop Protection | 562 | 565 | + 0.5 | + 0.1 | 369 | 337 | – 8.7 | – 11.5 | ||||||||||||||||||||||||||
| Environmental Science, BioScience | 110 | 104 | – 5.5 | – 4.9 | 114 | 125 | + 9.6 | + 8.5 | ||||||||||||||||||||||||||
| Materials | 427 | 440 | + 3.0 | + 3.0 | 229 | 236 | + 3.1 | + 2.5 | ||||||||||||||||||||||||||
| Systems | 797 | 835 | + 4.8 | + 4.4 | 479 | 517 | + 7.9 | + 7.2 | ||||||||||||||||||||||||||
Continuing operations (incl. reconciliation) | 3,047 | 3,169 | + 4.0 | + 3.9 | 1,768 | 1,908 | + 7.9 | + 6.3 | ||||||||||||||||||||||||||
| Sales by Region and Segment | Asia/Pacific | Latin America/Africa/Middle East | ||||||||||||||||||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | 2005 | 2006 | % yoy | adj. % yoy | ||||||||||||||||||||||||||
| € million | ||||||||||||||||||||||||||||||||||
2nd Quarter | 223 | 256 | + 14.8 | + 17.6 | 107 | 141 | + 31.8 | +31.9 | ||||||||||||||||||||||||||
| Pharmaceuticals | 74 | 80 | + 8.1 | + 8.0 | 190 | 191 | + 0.5 | +0.1 | ||||||||||||||||||||||||||
| Consumer Health | 193 | 185 | -4.1 | -4.2 | 194 | 182 | -6.2 | -6.7 | ||||||||||||||||||||||||||
| Crop Protection | 43 | 52 | + 20.9 | + 22.1 | 19 | 28 | + 47.4 | +47.4 | ||||||||||||||||||||||||||
| Environmental Science, BioScience | 308 | 292 | - 5.2 | - 4.7 | 81 | 91 | + 12.3 | +13.1 | ||||||||||||||||||||||||||
| Materials | 235 | 259 | + 10.2 | + 9.8 | 178 | 213 | + 19.7 | +17.3 | ||||||||||||||||||||||||||
| Systems | ||||||||||||||||||||||||||||||||||
Continuing operations (incl. reconciliation) | 1,090 | 1,136 | + 4.2 | + 4.9 | 781 | 859 | + 10.0 | +9.2 | ||||||||||||||||||||||||||
| Continuing operations | |||||||||||||||||
| Sales by Region and Segment | |||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | |||||||||||||
| € million | |||||||||||||||||
2nd Quarter | 988 | 1,188 | + 20.2 | +20.5 | |||||||||||||
| Pharmaceuticals | 1,015 | 1,069 | + 5.3 | +4.6 | |||||||||||||
| Consumer Health | 1,318 | 1,269 | -3.7 | -4.8 | |||||||||||||
| Crop Protection | 286 | 309 | + 8.0 | +8.0 | |||||||||||||
| Environmental Science, BioScience | 1,045 | 1,059 | + 1.3 | +1.4 | |||||||||||||
| Materials | 1,689 | 1,824 | + 8.0 | +7.3 | |||||||||||||
| Systems | |||||||||||||||||
Continuing operations (incl. reconciliation) | 6,686 | 7,072 | + 5.8 | +5.3 | |||||||||||||
20
Table of Contents
| Sales by Region and Segment | Europe | North America | ||||||||||||||||||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | 2005 | 2006 | % yoy | adj. % yoy | ||||||||||||||||||||||||||
| € million | ||||||||||||||||||||||||||||||||||
1st Half | ||||||||||||||||||||||||||||||||||
| Pharmaceuticals | 809 | 928 | + 14.7 | + 14.5 | 499 | 669 | + 34.1 | + 26.4 | ||||||||||||||||||||||||||
| Consumer Health | 797 | 857 | + 7.5 | + 7.7 | 596 | 715 | + 20.0 | + 13.9 | ||||||||||||||||||||||||||
| Crop Protection | 1,201 | 1,188 | – 1.1 | – 1.7 | 709 | 717 | + 1.1 | – 5.5 | ||||||||||||||||||||||||||
| Environmental Science, BioScience | 245 | 248 | + 1.2 | + 0.9 | 259 | 282 | + 8.9 | + 2.9 | ||||||||||||||||||||||||||
| Materials | 839 | 869 | + 3.6 | + 3.6 | 433 | 473 | + 9.2 | + 4.1 | ||||||||||||||||||||||||||
| Systems | 1,572 | 1,625 | + 3.4 | + 3.3 | 928 | 1,066 | + 14.9 | + 9.3 | ||||||||||||||||||||||||||
Continuing operations (incl. reconciliation) | 6,030 | 6,342 | + 5.2 | + 5.0 | 3,433 | 3,930 | + 14.5 | + 8.3 | ||||||||||||||||||||||||||
2005 figures restated
adj. = currency-adjusted
adj. = currency-adjusted
| Asia/Pacific | Latin America/Africa/Middle East | Continuing operations | |||||||||||||||||||||||||||||||||||||||||||||
| Asia/Pacific | Latin America/Africa/Middle East | |||||||||||||||||||||||||||||||||
| Sales by Region and Segment | ||||||||||||||||||||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | 2005 | 2006 | % yoy | adj. % yoy | ||||||||||||||||||||||||||
| € million | ||||||||||||||||||||||||||||||||||
1st Half | ||||||||||||||||||||||||||||||||||
| Pharmaceuticals | 431 | 479 | + 11.1 | + 11.2 | 201 | 260 | + 29.4 | +23.6 | ||||||||||||||||||||||||||
| Consumer Health | 141 | 164 | + 16.3 | + 12.9 | 346 | 388 | + 12.1 | +6.2 | ||||||||||||||||||||||||||
| Crop Protection | 398 | 392 | - 1.5 | - 3.8 | 427 | 385 | - 9.8 | -16.6 | ||||||||||||||||||||||||||
| Environmental Science, BioScience | 66 | 81 | + 22.7 | + 22.1 | 43 | 56 | + 30.2 | +22.6 | ||||||||||||||||||||||||||
| Materials | 544 | 577 | + 6.1 | + 3.3 | 152 | 175 | + 15.1 | +12.1 | ||||||||||||||||||||||||||
| Systems | 472 | 490 | + 3.8 | + 1.1 | 338 | 419 | + 24.0 | +16.7 | ||||||||||||||||||||||||||
Continuing operations (incl. reconciliation) | 2,077 | 2,208 | + 6.3 | + 4.2 | 1,532 | 1,708 | + 11.5 | +5.4 | ||||||||||||||||||||||||||
| Continuing operations | |||||||||||||||||
| Sales by Region and Segment | |||||||||||||||||
| (by Market) | 2005 | 2006 | % yoy | adj. % yoy | |||||||||||||
| € million | |||||||||||||||||
1st Half | |||||||||||||||||
| Pharmaceuticals | 1,940 | 2,336 | + 20.4 | + 17.9 | |||||||||||||
| Consumer Health | 1,880 | 2,124 | + 13.0 | + 9.8 | |||||||||||||
| Crop Protection | 2,735 | 2,682 | - 1.9 | - 5.3 | |||||||||||||
| Environmental Science, BioScience | 613 | 667 | + 8.8 | + 5.5 | |||||||||||||
| Materials | 1,968 | 2,094 | + 6.4 | + 4.3 | |||||||||||||
| Systems | 3,310 | 3,600 | + 8.8 | + 6.1 | |||||||||||||
Continuing operations (incl. reconciliation) | 13,072 | 14,188 | + 8.5 | + 5.8 | |||||||||||||
Interim Report as of June 30, 2006
Liquidity and Capital Resources
Operating cash flow
Gross cash flow in the second quarter of 2006 increased by 11.2 percent to€964 million (Q2 2005:€867 million) thanks to the strong growth in business. Net cash flow from continuing operations fell by€85 million to€895 million (Q2 2005:€980 million), due especially to lower cash inflows in the CropScience subgroup. The net cash flow includes a€145 million inflow from the newly acquired Schering business. Of this amount, the sale of hedging options related to stock option plans of Schering AG accounted for€121 million.
There will be a corresponding outflow in the third quarter.
Investing cash flow
There was a net cash outflow of€13,836 million for investing activities (Q2 2005: inflow of€247 million). The principal item was€14.1 billion in cash outflows for acquisitions, comprising€15.1 billion in purchase price disbursements for Schering AG through June 30, 2006, less approximately€1 billion in acquired cash.
21
Table of Contents
Interim Report as of June 30, 2006
Capital expenditures for property, plant and equipment (€324 million) and intangible assets (€16 million) rose by€69 million to 340 million (Q2 2005:€271 million). Interest receipts increased as a result of high cash holdings prior to the Schering acquisition. Interest and dividend receipts thus rose from€334 million to€375 million. There was a net cash inflow of€133 million from the sale of marketable securities (Q2 2005: outflow of€94 million).
Financing cash flow
The net cash inflow of€12,320 million from financing activities (Q2 2005: outflow of€1,347 million) mainly comprised net borrowings of€13,473 million in connection with the financing of the Schering acquisition (for details see the table “Financing Measures for the Schering Acquisition” on page 23).
Cash outflows for dividend payments amounted to€692 million (Q2 2005:€429 million), while those for interest payments came to€461 million (Q2 2005:€439 million).
| Cash Flow Key Data | 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | ||||||||||||||
| € million | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
Gross cash flow* | 867 | 964 | 1,927 | 2,090 | ||||||||||||||
| Changes in working capital | 113 | (69 | ) | (1,218 | ) | (1,131 | ) | |||||||||||
Net cash provided by (used in) operating activities (net cash flow), continuing operations | 980 | 895 | 709 | 959 | ||||||||||||||
| Net cash provided by (used in) operating activities (net cash flow), discontinued operations | 45 | 107 | 58 | 171 | ||||||||||||||
Net cash provided by (used in) operating activities (net cash flow), total | 1,025 | 1,002 | 767 | 1,130 | ||||||||||||||
Net cash provided by (used in) investing activities (total) | 247 | (13,836 | ) | (700 | ) | (14,028 | ) | |||||||||||
Net cash provided by (used in) financing activities (total) | (1,347 | ) | 12,320 | (1,777 | ) | 12,133 | ||||||||||||
Change in cash and cash equivalents due to business activities (total) | (75 | ) | (514 | ) | (1,710 | ) | (765 | ) | ||||||||||
| 2005 figures restated | ||
| * | for definition see Bayer Group Key Data on page 2 |
| Net Debt | Dec. 31, | March 31, | June 30, | ||||||||||
| € million | 2005 | 2006 | 2006 | ||||||||||
| Noncurrent financial liabilities as per balance sheets (including derivatives) | 7,185 | 7,419 | 10,373 | ||||||||||
| of which | |||||||||||||
| Mandatory convertible bond | — | — | 2,271 | ||||||||||
| Hybrid bond | 1,268 | 1,250 | 1,242 | ||||||||||
| Current financial liabilities as per balance sheets (including derivatives) | 1,767 | 1,332 | 12,053 | ||||||||||
| – Derivative receivables | 188 | 170 | 212 | ||||||||||
Financial liabilities from continuing operations | 8,764 | 8,581 | 22,214 | ||||||||||
| – Liquid assets as per balance sheets less amount not freely available | 3,270 | 2,864 | 2,269 | ||||||||||
Net debt from continuing operations | 5,494 | 5,717 | 19,945 | ||||||||||
| * | In view of the restriction on its use, the€304 million liquidity in the escrow accounts was not deducted when calculating net debt. June 30, 2006:€2,269 million =€2,573 million –€304 million. |
22
Table of Contents
Interim Report as of June 30, 2006
Liquid assets and net debt
Including marketable securities and other instruments, the Bayer Group had liquid assets of€2,573 million as of June 30, 2006. Of this amount,€304 million was held in escrow accounts to be used exclusively for payments relating to civil law settlements in antitrust proceedings.
Net debt increased by€14.2 billion in the second quarter of 2006. In connection with the Schering acquisition we spent€15.1 billion, acquiring€1.0 billion in liquid assets and€0.2 billion in financial liabilities from Schering.
The measures adopted to finance the acquisition are shown in the table below. The remainder of the purchase price was paid mainly out of liquidity.
In addition, on July 6, 2006 – after the end of the reporting period – we placed 34 million new shares with German and international institutional investors. Cash inflow from the capital increase amounted to some€1.2 billion. Together with the placing of the€2.3 billion mandatory convertible bond, this successfully completes the equity raising announced in connection with the Schering acquisition. The total€3.5 billion thus raised is significantly below the€4 billion limit originally set.
The net debt should also be viewed in light of the fact that the noncurrent financial liabilities include in their entirety both the 100-year hybrid bond issued in 2005 and the newly issued mandatory convertible bond. In computing debt indicators, rating agencies assign hybrid bonds partly, and mandatory convertible bonds wholly, to stockholders’ equity. These bonds thus support the Group’s rating-specific debt indicators.
In July 2006, Standard & Poor’s altered Bayer AG’s long-term issuer rating from A with stable outlook to BBB+ with positive outlook, citing the debt increase associated with the Schering acquisition. Also in July 2006, Moody’s confirmed the current A3 rating for Bayer AG, changing the outlook from stable to negative.
| Financing Measures for the Schering Acquisition in the Second Quarter of 2006 | |||||
| € billion | |||||
| Use of a credit facility and syndicated loan | 7.6 | ||||
| Placement of a 3-year floating-rate Eurobond | 1.6 | ||||
| Placement of a 7-year fixed-rate Eurobond | 1.0 | ||||
| Placement of a 12-year fixed-rate sterling bond | 0.4 | ||||
| Placement of a mandatory convertible bond | 2.3 | ||||
| Total | 12.9 | ||||
23
Table of Contents
Interim Report as of June 30, 2006
Asset and Capital Structure
In connection with the acquisition of Schering AG and the first-time inclusion of its activities in the consolidated financial statements as of June 30, 2006, along with the reporting of the Diagnostics Division as a discontinued operation, major changes resulted in the Bayer Group’s asset and capital structure compared to the previous year. Except where explicitly stated otherwise, the following commentary compares the Bayer Group’s balance sheets as of June 30, 2006 and December 31, 2005. Explanations concerning the first-time consolidation of Schering are provided on page 41f. in the notes to the financial statements.
Total assets increased by€19.5 billion to€56.2 billion, mainly as a result of the Schering acquisition. As of June 30, 2006, the Diagnostics business is recognized under “Assets held for sale and discontinued operations” and the corresponding liability item.
The€16.3 billion growth in noncurrent assets, to€36.4 billion, is mostly related to the Schering acquisition. The major part of the increase results from recognizing the intangible assets of Schering – primarily production-related rights and know-how – at their fair value of€11.4 billion. In addition, goodwill of€5.2 billion was capitalized.
Current assets of continuing operations rose by€1.8 billion to€18.4 billion, largely because of the trade accounts receivable, inventories and liquid assets acquired from Schering.
| Dec. 31, | June 30, | Change | |||||||||||
| € million | 2005 | 2006 | % | ||||||||||
Noncurrent assets | 20,130 | 36,406 | +80.9 | ||||||||||
Current assets | 16,592 | 18,388 | +10.8 | ||||||||||
| Assets held for sale and discontinued operations | — | 1,396 | • | ||||||||||
Total current assets | 16,592 | 19,784 | +19.2 | ||||||||||
Assets | 36,722 | 56,190 | +53.0 | ||||||||||
Stockholders’ equity | 11,157 | 12,827 | +15.0 | ||||||||||
Noncurrent liabilities | 16,495 | 23,138 | �� | +40.3 | |||||||||
Current liabilities | 9,070 | 19,789 | +118.2 | ||||||||||
| Liabilities directly related to assets held for sale and discontinued operations | — | 436 | • | ||||||||||
Total current liabilities | 9,070 | 20,225 | +123.0 | ||||||||||
Liabilities | 25,565 | 43,363 | +69.6 | ||||||||||
Stockholders’ equity and liabilities | 36,722 | 56,190 | + 53.0 | ||||||||||
24
Table of Contents
Interim Report as of June 30, 2006
Stockholders’ equity expanded by€1.7 billion to€12.8 billion. While Group net income amounted to€1.1 billion and other comprehensive income increased by€0.7 billion, stockholders’ equity was diminished by the dividend payment (€0.7 billion) and negative currency effects (€0.5 billion). In addition, minority interest in equity rose by€1.1 billion because of the remaining minority stockholders of Schering AG. Equity coverage of total assets thus came to 22.8 percent as of June 30, 2006 (December 31, 2005: 30.4 percent). In light of the capital increase carried out in July, we expect the equity ratio to be at about the 2005 level once the planned portfolio measures have been implemented.
Liabilities grew by€17.8 billion compared to December 31, 2005, to€43.4 billion. Current and noncurrent financial liabilities rose by€13.5 billion, mainly due to the financing of the Schering acquisition (see Liquidity and Capital Resources, page 21ff.). Even including Schering’s pension provisions of€0.4 billion, pension provisions were down by€0.9 billion, mainly as a result of increases in discount rates.
Employees
Starting with the second quarter of 2006, the number of employees is converted to full-time equivalents, which means part-time employees are included in proportion to their contractual working hours. This reporting change reduces the Bayer Group headcount figure as of March 31, 2006 from 88,500 to 86,900 (–1.8 percent). We believe this presentation improves the comparability of personnel expenses and employee numbers. The previous year’s data have been restated accordingly.
On June 30, 2006 the Bayer Group had 110,200 employees, 23,300 more than on March 31, 2006. This increase is primarily attributable to the first-time inclusion of 23,100 Schering employees. No longer included are the 5,100 employees of the Diagnostics Division, as this business is reported under discontinued operations. Personnel expenses in the second quarter increased by 5.1 percent to€1,559 million.
In the individual regions, too, the Schering employees in particular led to a rise in headcount compared to the previous year. In North America we now employ 18,000 people (+4,000), while we have 16,900 employees in the Asia/Pacific region (+4,300) and 14,000 in Latin America/ Africa/Middle East (+3,000). The Bayer Group has 61,300 employees in Europe (+12,400). In Germany we employ 44,100 people, which is 40.0 percent of the Group total.
25
Table of Contents
Interim Report as of June 30, 2006
Legal Risks
As a global company with a diverse business portfolio, the Bayer Group is exposed to numerous legal risks.
Legal proceedings currently considered to involve material risks are outlined below. The litigation referred to does not necessarily represent an exhaustive list.
Lipobay/Baycol:As of August 18, 2006, the number of Lipobay/Baycol cases pending against Bayer worldwide was approximately 3,000 (approximately 2,900 of them in the United States, including several class actions). At the same date, Bayer had settled approximately 3,115 Lipobay/Baycol cases worldwide without acknowledging any liability and resulting in settlement payments of approximatelyus$ 1,154 million. In the United States five cases have been tried to date, all of which were found in Bayer’s favor.
After almost five years of litigation we are currently aware of fewer than 30 pending cases in the United States that in our opinion hold a potential for settlement, although we cannot rule out the possibility that additional cases involving serious side effects from Lipobay/Baycol may come to our attention. In addition, there could be further settlements of cases outside of the United States.
In addition to accounting measures taken in previous years, Bayer recorded charges of€4.7 million to the operating result in the first quarter of 2006 in respect of settlements expected to be concluded and anticipated defense costs. No further charges had to be recorded in the second quarter.
PPA:Regardingppa approximately 160 lawsuits were pending inu.s. courts against Bayer as of June 30, 2006, of which approximately 100 name Bayer as the only manufacturing defendant. In addition, approximately 250 dismissed claims are currently still on appeal to a United States Court of Appeals. No lawsuits have been filed outside the United States.
Three state cases have proceeded to trial. Two have resulted in defense verdicts for Bayer. In one case, the plaintiff was awarded damages ofus$ 400,000. This case was settled in July 2005 while on appeal.
As of June 30, 2006, Bayer had settled 349 cases resulting in payments of approximatelyus$ 53.4 million, without acknowledging any liability. In the fiscal year 2005, Bayer recorded expenses in the amount of€62 million for settlements already concluded or expected to be concluded and expected defense costs. No further expenses had to be recorded during the first six months of 2006.
26
Table of Contents
Interim Report as of June 30, 2006
Bayer will defend itself vigorously in all Lipobay/Baycol andppa cases in which in our view no potential for settlement exists or where an appropriate settlement cannot be achieved.
Since the existing insurance coverage with respect to the Lipobay/Baycol andppa cases is exhausted (except that insurance coverage forppa exists for up to 5 percent of future costs), it is possible – depending on the future progress of the litigation – that Bayer could face further payments that are not covered by the accounting measures already taken. We will regularly review the possibility of further accounting measures depending on the progress of the litigation.
Ciprofloxacin:39 putative class action lawsuits and one individual lawsuit against Bayer involving the medication Cipro® have been pending since July 2000 in the United States. The plaintiffs are suing Bayer and other companies also named as defendants, alleging that a settlement reached in 1997 to end patent litigation between Bayer and Barr Laboratories, Inc. violated antitrust regulations. The plaintiffs claim the alleged violation prevented the marketing of generic ciprofloxacin. Plaintiffs also are seeking triple damages underu.s. law. After the settlement with Barr, the patent was the subject of a successful re-examination by theu.s. Patent and Trademark Office and of successful defenses inu.s. federal courts. The patent has since expired.
All the actions pending in federal court were consolidated in federal district court in New York in a multidistrict litigation (mdl) proceeding. On March 31, 2005, the court granted Bayer’s motion for summary judgment and dismissed all of plaintiffs’ claims in themdl proceeding. The plaintiffs are appealing this decision. Further cases are pending before various state courts. Bayer believes that it has meritorious defenses and intends to defend these cases vigorously.
Rubber, polyester polyols, urethane: Proceedings involving the former rubber- related lines of business
Investigations and proceedings by the respective authorities in thee.u. and Canada for alleged anticompetitive conduct involving certain products in the rubber field are pending. In the United States, Bayer AG has paid fines in two cases on the basis of agreements reached with theu.s.Department of Justice. In December 2005, thee.u. Commission imposed a fine of€58.9 million for antitrust violations in the area of rubber chemicals. The respective amount was paid at the end of March 2006. On July 6, 2006, theu.s. Department of Justice and on July 26, 2006, thee.u. Commission notified Bayer AG that they had closed their respectiveepdm investigations.
27
Table of Contents
Interim Report as of June 30, 2006
Numerous civil claims for damages including class actions are pending in the United States and Canada against Bayer AG and certain of its subsidiaries as well as other companies. The lawsuits involve rubber chemicals,epdm, nbr and polychloroprene rubber (cr). Bayer has reached agreements or agreements in principle to settle a number of theu.s. actions. Some of these agreements or agreements in principle remain subject to court approval. These settlements do not resolve all of the pending civil litigation with respect to the aforementioned products, nor do they preclude the bringing of additional claims.
Proceedings involving polyester polyols, urethanes and urethane chemicals
In Canada an investigation is pending against Bayer Corporation for alleged anticompetitive conduct relating to adipic-based polyester polyols. In the United States, Bayer Corporation paid a fine on the basis of an agreement reached with theu.s. Department of Justice in 2004.
A number of civil claims for damages including class actions have been filed in the United States against Bayer involving allegations of unlawful collusion on prices for certain polyester polyols, urethanes and urethane chemicals product lines. Similar actions are pending in Canada with respect to polyester polyols. Bayer has reached agreements or agreements in principle to settle a number of theu.s. actions. These agreements or agreements in principle remain subject to court approval. These settlements do not resolve all of the pending civil litigation with respect to the aforementioned products, nor do they preclude the bringing of additional claims.
Proceedings involving polyether polyols and other precursors for urethane end-use products
Bayer has been named as a defendant in multiple putative class action lawsuits in the United States and Canada involving allegations of price fixing for, inter alia, polyether polyols and certain other precursors for urethane end-use products. In the United States, at the beginning of 2006, Bayer reached an agreement in principle to settle all of the class action cases relating to claims from direct purchasers of polyether polyols,mdi ortdi (and related systems). Bayer has also reached an agreement in principle with a further group of plaintiffs. These settlements, which remain subject to court approval, do not resolve all of the pending civil litigation with respect to the aforementioned products, nor do they preclude the bringing of additional claims. In February 2006 Bayer was served with a subpoena from theu.s. Department of Justice seeking information relating to the manufacture and sale of these products.
28
Table of Contents
Interim Report as of June 30, 2006
Impact of antitrust proceedings on Bayer
In consideration of the portion allocated to Lanxess, expenses in the amount of€336 million were accrued in the course of 2005 which led to the establishment of a provision for the previously described civil proceedings in the amount of€285 million as of December 31, 2005. In the meantime this provision has been adjusted and stood at€216 million as of June 30, 2006. In addition to payment of the fine imposed by thee.u. Commission in the rubber chemicals proceeding, Bayer recognized a provision of€21 million in respect of the other rubber-related e.u.proceedings noted above, although a reliable estimate as to the actual amount of any additional fines can currently not be made.
These provisions taken may not be sufficient to cover the ultimate outcome of the above-described matters. The amount of provisions established for civil proceedings is based on the expected payments under the settlement agreements described above. In the case of proposed settlements in civil matters which have been asserted as class actions, members of the putative classes have the right to “opt out” of the class, meaning that they elect not to participate in the settlement. Plaintiffs that opt out are not bound by the terms of the settlement and have the right to independently bring individual actions in their own names to recover damages they allegedly suffered. We cannot predict the size of the opt-out groups or their impact on the settlement agreements.
Bayer will continue to pursue settlements that in its view are warranted. In cases where settlement is not achievable, Bayer will continue to defend itself vigorously.
The financial risk associated with the proceedings described above beyond the amounts already paid and the financial provisions already established is currently not quantifiable due to the considerable uncertainty associated with these proceedings. Consequently, no provisions other than those described above have been established. The Company expects that, in the course of the regulatory proceedings and civil damages suits, additional charges will become necessary.
Arbitration proceeding concerning propylene oxide
Bayer and Lyondell Chemical Co., in a binding arbitration proceeding, asserted claims against each other generally relating to differences in contractual interpretation of their joint venture agreement for the manufacture of propylene oxide. On May 22, 2006, the arbitration panel rendered a final award in which it denied Bayer’s claims and affirmed Lyondell’s counterclaims. The panel awarded Lyondell, among other things,us$ 121 million in contractual fees with respect to the period through June 30, 2005, plus post-judgment interest until paid. Bayer has filed an appeal of the panel’s final award. In contrast Lyondell claims further interest in the amount ofus$ 22 million.
As of March 2006 Bayer had established a provision totalingus$ 184 million to cover the amounts awarded, estimated attorneys’ fees and estimated interest.
Bayer separately has notified Lyondell of its claim in connection with Lyondell‘s failure to compensate Bayer for taking certain propylene oxide quantities from Bayer’s share of capacity under the joint venture.
29
Table of Contents
Interim Report as of June 30, 2006
Subsequent Events
On July 6, 2006 we placed 34 million new shares with German and international institutional investors by means of an accelerated bookbuilding. The order book was closed after a few hours due to high demand. This capital increase raised approximately€1.2 billion in financing for the Schering acquisition. The issue price of the new shares was€34.75 per share. The new shares carry full entitlement to the dividend for fiscal 2006. The related 4.7 percent cash increase in the company’s capital stock was approved by the Supervisory Board and implemented on the basis of the authorization granted by the Annual Stockholders’ Meeting on April 28, 2006 (Authorized Capitalii).
On July 12, 2006, we announced the successful completion of our public takeover offer for Schering AG. On that date we controlled 92.4 percent of the outstanding voting rights of Schering AG.
With the acquisition in early July 2006 of theu.s. company Metrika, we expanded our product portfolio for diabetes monitoring and thus strengthened our diabetes business. Metrika produces and markets a cellphone-sized device with which diabetics can monitor their long-term blood glucose levels themselves.
To improve its cost structures, the Bayer CropScience subgroup is initiating a new program of measures, due to be largely completed by 2009 and designed to achieve annual savings of roughly€300 million. In this way Bayer CropScience aims to enhance its competitiveness for the long term and strengthen the foundation for its further development. The principal aim of the new efficiency program is to sustainably shrink the company’s infrastructure and process costs in areas such as manufacturing, supply chain, development and marketing. About half of the planned savings are to be achieved through consolidation of production sites, optimization of procurement activities and a reduction in personnel costs. As part of the program, a number of formulation and production sites worldwide will be either restructured or closed, and a total of approximately 1,500 positions are to be eliminated through the end of 2009. In this connection Bayer CropScience anticipates special cash charges of some€330 million along with write-downs of about€120 million. These amounts will be reflected mainly in the 2007 and 2008 financial statements. We expect the measures to be accretive toebit after special items starting in 2008.
30
Table of Contents
Interim Report as of June 30, 2006
Bayer Stock
The price of Bayer stock in the second quarter was influenced primarily by the planned acquisition of Schering AG. In what was at times a very weak market environment, Bayer stock closed at€35.94 on June 30, 2006, up 1.8 percent from the closing price on December 31, 2005.
Including the dividend of€0.95 per share paid on May 2, 2006, Bayer stock achieved a performance of 4.5 percent for the first half of 2006. Over the same period thedax rose by 5.1 percent to 5,683.
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | ||||||||||||||
| Bayer Stock Key Data | 2005 | 2006 | 2005 | 2006 | |||||||||||||
High for the period (€) | 28.62 | 36.75 | 28.62 | 36.75 | |||||||||||||
Low for the period (€) | 24.79 | 30.56 | 22.03 | 30.56 | |||||||||||||
| Average daily share turnover on German stock exchanges (million) | 4.0 | 7.3 | 4.5 | 6.4 | |||||||||||||
| Change | |||||||||||||||||
| June 30, 2006/ | |||||||||||||||||
| Dec. 31, 2005 | |||||||||||||||||
| June 30, 2005 | June 30, 2006 | Dec. 31, 2005 | % | ||||||||||||||
Share price (€) | 27.59 | 35.94 | 35.29 | 1.8 | |||||||||||||
Market capitalization (€ million) | 20,150 | 26,248 | 25,774 | 1.8 | |||||||||||||
Stockholders’ equity (€ million) | 10,526 | 12,827 | 11,157 | 15.0 | |||||||||||||
| Number of shares entitled to the dividend (million) | 730.34 | 730.34 | 730.34 | 0.0 | |||||||||||||
| DAX | 4,586 | 5,683 | 5,408 | 5.1 | |||||||||||||
2005 figures restated
xetra closing prices; source: Bloomberg
xetra closing prices; source: Bloomberg
31
Table of Contents
Interim Report as of June 30, 2006
Bayer Group
Consolidated Statements of Income
Consolidated Statements of Income
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | |||||||||||||||
| € million | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
Net sales | 6,686 | 7,072 | 13,072 | 14,188 | ||||||||||||||
| Cost of goods sold | (3,642 | ) | (3,838 | ) | (7,050 | ) | (7,531 | ) | ||||||||||
Gross profit | 3,044 | 3,234 | 6,022 | 6,657 | ||||||||||||||
| Selling expenses | (1,375 | ) | (1,466 | ) | (2,545 | ) | (2,855 | ) | ||||||||||
| Research and development expenses | (453 | ) | (448 | ) | (846 | ) | (871 | ) | ||||||||||
| General administration expenses | (359 | ) | (391 | ) | (661 | ) | (753 | ) | ||||||||||
| Other operating income | 405 | 166 | 789 | 375 | ||||||||||||||
| Other operating expenses | (555 | ) | (217 | ) | (1,066 | ) | (598 | ) | ||||||||||
EBIT | 707 | 878 | 1,693 | 1,955 | ||||||||||||||
| Equity-method income (loss) | 6 | (3 | ) | 4 | (11 | ) | ||||||||||||
| Non-operating income | 176 | 156 | 286 | 304 | ||||||||||||||
| Non-operating expenses | (311 | ) | (385 | ) | (550 | ) | (740 | ) | ||||||||||
Non-operating result | (129 | ) | (232 | ) | (260 | ) | (447 | ) | ||||||||||
Income before income taxes | 578 | 646 | 1,433 | 1,508 | ||||||||||||||
| Income taxes | (168 | ) | (197 | ) | (442 | ) | (483 | ) | ||||||||||
Income from continuing operations after taxes | 410 | 449 | 991 | 1,025 | ||||||||||||||
Income from discontinued operations after taxes | 2 | 6 | 66 | 27 | ||||||||||||||
Income after taxes | 412 | 455 | 1,057 | 1,052 | ||||||||||||||
of which attributable to minority interest | 6 | 3 | (1 | ) | 0 | |||||||||||||
of which attributable to Bayer AG stockholders (net income) | 406 | 452 | 1,058 | 1,052 | ||||||||||||||
Earnings per share (€) | ||||||||||||||||||
From continuing operations | ||||||||||||||||||
| Basic* | 0.55 | 0.59 | 1.36 | 1.38 | ||||||||||||||
| Diluted* | 0.55 | 0.59 | 1.36 | 1.38 | ||||||||||||||
From continuing and discontinued operations | ||||||||||||||||||
| Basic* | 0.56 | 0.60 | 1.45 | 1.41 | ||||||||||||||
| Diluted* | 0.56 | 0.60 | 1.45 | 1.41 | ||||||||||||||
| 2005 figures restated | ||
| * | The ordinary shares to be issued upon conversion of the mandatory convertible bond are treated as already issued shares. |
32
Table of Contents
Interim Report as of June 30, 2006
Bayer Group
Consolidated Balance Sheets
Consolidated Balance Sheets
| € million | June 30, 2005 | June 30, 2006 | Dec. 31, 2005 | ||||||||||
Noncurrent assets | |||||||||||||
| Goodwill and other intangible assets | 7,758 | 23,675 | 7,688 | ||||||||||
| Property, plant and equipment | 8,040 | 9,208 | 8,321 | ||||||||||
| Investments in associates | 790 | 760 | 795 | ||||||||||
| Other financial assets | 1,191 | 1,528 | 1,429 | ||||||||||
| Other receivables | 106 | 253 | 199 | ||||||||||
| Deferred taxes | 2,027 | 982 | 1,698 | ||||||||||
| 19,912 | 36,406 | 20,130 | |||||||||||
Current assets | |||||||||||||
| Inventories | 5,602 | 7,044 | 5,504 | ||||||||||
| Trade accounts receivable | 5,866 | 6,638 | 5,204 | ||||||||||
| Other financial assets | 442 | 363 | 214 | ||||||||||
| Other receivables | 1,389 | 1,222 | 1,421 | ||||||||||
| Claims for tax refunds | 780 | 548 | 726 | ||||||||||
| Liquid assets | |||||||||||||
| Marketable securities and other instruments | 119 | 82 | 233 | ||||||||||
| Cash and cash equivalents | 1,698 | 2,491 | 3,290 | ||||||||||
| 15,896 | 18,388 | 16,592 | |||||||||||
| Assets held for sale and discontinued operations | — | 1,396 | — | ||||||||||
Total current assets | 15,896 | 19,784 | 16,592 | ||||||||||
Assets | 35,808 | 56,190 | 36,722 | ||||||||||
Equity attributable to Bayer AG stockholders | |||||||||||||
| Capital stock of Bayer AG | 1,870 | 1,870 | 1,870 | ||||||||||
| Capital reserves of Bayer AG | 2,942 | 2,942 | 2,942 | ||||||||||
| Other reserves | 5,636 | 6,865 | 6,265 | ||||||||||
| 10,448 | 11,677 | 11,077 | |||||||||||
| Equity attributable to minority interest | 78 | 1,150 | 80 | ||||||||||
Stockholders’ equity | 10,526 | 12,827 | 11,157 | ||||||||||
Noncurrent liabilities | |||||||||||||
| Provisions for pensions and other post-employment benefits | 7,324 | 6,237 | 7,174 | ||||||||||
| Other provisions | 1,481 | 1,771 | 1,340 | ||||||||||
| Financial liabilities | 6,996 | 10,373 | 7,185 | ||||||||||
| Miscellaneous liabilities | 197 | 517 | 516 | ||||||||||
| Deferred taxes | 571 | 4,240 | 280 | ||||||||||
| 16,569 | 23,138 | 16,495 | |||||||||||
Current liabilities | |||||||||||||
| Other provisions | 2,674 | 3,803 | 3,009 | ||||||||||
| Financial liabilities | 2,019 | 12,053 | 1,767 | ||||||||||
| Trade accounts payable | 1,675 | 1,995 | 1,974 | ||||||||||
| Tax liabilities | 337 | 395 | 304 | ||||||||||
| Miscellaneous liabilities | 2,008 | 1,543 | 2,016 | ||||||||||
| 8,713 | 19,789 | 9,070 | |||||||||||
| Liabilities directly related to assets held for sale and discontinued operations | — | 436 | — | ||||||||||
Total current liabilities | 8,713 | 20,225 | 9,070 | ||||||||||
Liabilities | 25,282 | 43,363 | 25,565 | ||||||||||
Stockholders’ equity and liabilities | 35,808 | 56,190 | 36,722 | ||||||||||
| 2005 figures restated | ||
33
Table of Contents
Interim Report as of June 30, 2006
Bayer Group
Consolidated Statements of Cash Flows
Consolidated Statements of Cash Flows
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | |||||||||||||||
| € million | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
EBIT* | 707 | 878 | 1,693 | 1,955 | ||||||||||||||
| Income taxes | (187 | ) | (286 | ) | (401 | ) | (510 | ) | ||||||||||
| Depreciation and amortization | 394 | 430 | 790 | 835 | ||||||||||||||
| Change in pension provisions | (25 | ) | (51 | ) | (135 | ) | (182 | ) | ||||||||||
| (Gains) losses on retirements of noncurrent assets | (22 | ) | (7 | ) | (20 | ) | (8 | ) | ||||||||||
Gross cash flow | 867 | 964 | 1,927 | 2,090 | ||||||||||||||
| Decrease (increase) in inventories | (113 | ) | 11 | (331 | ) | (132 | ) | |||||||||||
| Decrease (increase) in trade accounts receivable | 414 | 11 | (536 | ) | (873 | ) | ||||||||||||
| Increase (decrease) in trade accounts payable | (89 | ) | (42 | ) | (345 | ) | (270 | ) | ||||||||||
| Changes in other working capital, other non-cash items | (99 | ) | (49 | ) | (6 | ) | 144 | |||||||||||
Net cash provided by (used in) operating activities (net cash flow), continuing operations | 980 | 895 | 709 | 959 | ||||||||||||||
| Net cash provided by (used in) operating activities (net cash flow), discontinued operations | 45 | 107 | 58 | 171 | ||||||||||||||
Net cash flow provided by (used in) operating activities (net cash flow), total | 1,025 | 1,002 | 767 | 1,130 | ||||||||||||||
| Cash outflows for additions to property, plant, equipment and intangible assets | (271 | ) | (340 | ) | (452 | ) | (759 | ) | ||||||||||
| Cash inflows from sales of property, plant, equipment and other assets | 16 | 63 | 272 | 83 | ||||||||||||||
| Cash inflows from sales of investments | 267 | 43 | 1,267 | 69 | ||||||||||||||
| Cash outflows for acquisitions less acquired cash | (5 | ) | (14,110 | ) | (2,058 | ) | (14,130 | ) | ||||||||||
| Interest and dividends received | 334 | 375 | 362 | 482 | ||||||||||||||
| Cash inflows from marketable securities | (94 | ) | 133 | (91 | ) | 227 | ||||||||||||
Net cash provided by (used in) investing activities (total) | 247 | (13,836 | ) | (700 | ) | (14,028 | ) | |||||||||||
| Capital contributions | 0 | 0 | 0 | 0 | ||||||||||||||
| Bayer AG dividend and dividend payments to minority stockholders/ reimbursements of advance capital gains tax payments | (429 | ) | (692 | ) | (462 | ) | (527 | ) | ||||||||||
| Issuances of debt | 177 | 13,493 | 441 | 13,762 | ||||||||||||||
| Retirements of debt | (656 | ) | (20 | ) | (1,210 | ) | (413 | ) | ||||||||||
| Interest paid | (439 | ) | (461 | ) | (546 | ) | (689 | ) | ||||||||||
Net cash provided by (used in) financing activities (total) | (1,347 | ) | 12,320 | (1,777 | ) | 12,133 | ||||||||||||
Change in cash and cash equivalents due to business activities (total) | (75 | ) | (514 | ) | (1,710 | ) | (765 | ) | ||||||||||
Cash and cash equivalents at beginning of period | 1,749 | 3,026 | 3,570 | 3,290 | ||||||||||||||
| Change in cash and cash equivalents due to changes in scope of consolidation | 0 | 0 | (196 | ) | (2 | ) | ||||||||||||
| Changes in cash and cash equivalents due to exchange rate movements | 24 | (21 | ) | 34 | (32 | ) | ||||||||||||
Cash and cash equivalents at end of period | 1,698 | 2,491 | 1,698 | 2,491 | ||||||||||||||
| Marketable securities and other instruments | 119 | 82 | 119 | 82 | ||||||||||||||
Liquid assets as per balance sheets | 1,817 | 2,573 | 1,817 | 2,573 | ||||||||||||||
| 2005 figures restated | ||
| * | for definition see Bayer Group Key Data on page 2 |
34
Table of Contents
Interim Report as of June 30, 2006
Bayer Group Consolidated Statements of Recognized Income and Expense
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | |||||||||||||||
| € million | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
| Changes in fair values of hedging instruments and available-for-sale securities, recognized in stockholders’ equity | (33 | ) | (20 | ) | (8 | ) | (11 | ) | ||||||||||
| Actuarial gains/losses on defined benefit obligations for pensions and other post-employment benefits | (1,183 | ) | 381 | (1,183 | ) | 1,178 | ||||||||||||
| Exchange differences on translation of operations outside the euro zone | 274 | (324 | ) | 679 | (466 | ) | ||||||||||||
| Deferred taxes on valuation adjustments, recognized directly in stockholders’ equity | 476 | (150 | ) | 466 | (462 | ) | ||||||||||||
Valuation adjustments recognized directly in stockholders’ equity | (466 | ) | (113 | ) | (46 | ) | 239 | |||||||||||
Income after taxes | 412 | 455 | 1,057 | 1,052 | ||||||||||||||
Total income and expense recognized in the financial statements | (54 | ) | 342 | 1,011 | 1,291 | |||||||||||||
of which attributable to minority interest | 11 | 5 | 7 | (3 | ) | |||||||||||||
of which attributable to Bayer AG stockholders | (65 | ) | 337 | 1,004 | 1,294 | |||||||||||||
35
Table of Contents
Interim Report as of June 30, 2006/Notes
Key Data by Segment
| € million | HealthCare | |||||||||||||||||
| Pharmaceuticals | Consumer Health | |||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
| Net sales (external) | 988 | 1,188 | 1,015 | 1,069 | ||||||||||||||
| — Change | + 5.2 | % | + 20.2 | % | + 39.8 | % | + 5.3 | % | ||||||||||
| — Change in local currencies | + 5.9 | % | + 20.5 | % | + 40.8 | % | + 4.6 | % | ||||||||||
| Intersegment sales | 14 | 12 | 7 | 1 | ||||||||||||||
| EBITDA** | 145 | 223 | 143 | 231 | ||||||||||||||
Special items | (20 | ) | (14 | ) | (61 | ) | (2 | ) | ||||||||||
EBITDA before special items | 165 | 237 | 204 | 233 | ||||||||||||||
| EBITDA margin before special items | 16.7 | % | 19.9 | % | 20.1 | % | 21.8 | % | ||||||||||
| EBIT* | 109 | 159 | 110 | 196 | ||||||||||||||
Special items | (20 | ) | (14 | ) | (61 | ) | (2 | ) | ||||||||||
EBIT before special items | 129 | 173 | 171 | 198 | ||||||||||||||
| EBIT margin before special items | 13.1 | % | 14.6 | % | 16.8 | % | 18.5 | % | ||||||||||
| Gross cash flow* | 106 | 157 | 111 | 179 | ||||||||||||||
| Net cash flow* | 143 | 284 | 43 | 83 | ||||||||||||||
| Depreciation, amortization and write-downs/write-backs | 36 | 64 | 33 | 35 | ||||||||||||||
| 1st Half | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
| Net sales (external) | 1,940 | 2,336 | 1,880 | 2,124 | ||||||||||||||
| — Change | - 4.1 | % | + 20.4 | % | + 37.1 | % | + 13.0 | % | ||||||||||
| — Change in local currencies | - 3.2 | % | + 17.9 | % | + 38.5 | % | + 9.8 | % | ||||||||||
| Intersegment sales | 19 | 25 | 12 | 3 | ||||||||||||||
| EBITDA** | 272 | 464 | 263 | 449 | ||||||||||||||
Special items | (118 | ) | (19 | ) | (82 | ) | (3 | ) | ||||||||||
EBITDA before special items | 390 | 483 | 345 | 452 | ||||||||||||||
| EBITDA margin before special items | 20.1 | % | 20.7 | % | 18.4 | % | 21.3 | % | ||||||||||
| EBIT* | 195 | 361 | 189 | 373 | ||||||||||||||
Special items | (118 | ) | (19 | ) | (82 | ) | (3 | ) | ||||||||||
EBIT before special items | 313 | 380 | 271 | 376 | ||||||||||||||
| EBIT margin before special items | 16.1 | % | 16.3 | % | 14.4 | % | 17.7 | % | ||||||||||
| Gross cash flow* | 180 | 319 | 198 | 309 | ||||||||||||||
| Net cash flow* | 51 | 273 | 157 | 137 | ||||||||||||||
| Depreciation, amortization and write-downs/write-backs | 77 | 103 | 74 | 76 | ||||||||||||||
2005 figures restated
| * | for definition see Bayer Group Key Data on page 2 | |
| ** | ebitda =ebit plus amortization of intangible assets and depreciation of property, plant and equipment.ebitda,ebitda before special items andebitda margin are not defined in the International Financial Reporting Standards and should therefore be regarded only as supplementary information. The company considers underlyingebitda to be a more suitable indicator of operating performance since it is not affected by depreciation, amortization, write-downs/write-backs or special items. The company also believes that this indicator gives readers a clearer picture of the results of operations and ensures greater comparability of data over time. The underlyingebitda margin is calculated by dividing underlyingebitda by sales. |
36
Table of Contents
Interim Report as of June 30, 2006/Notes
| CropScience | MaterialScience | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Environmental | Continuing | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crop Protection | Science, BioScience | Materials | Systems | Reconciliation | Operations | |||||||||||||||||||||||||||||||||||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||||||||||||||||
| Net sales (external) | 1,318 | 1,269 | 286 | 309 | 1,045 | 1,059 | 1,689 | 1,824 | 345 | 354 | 6,686 | 7,072 | ||||||||||||||||||||||||||||||||||||||||||
| — Change | - 2.5 | % | - 3.7 | % | - 1.4 | % | + 8.0 | % | + 30.6 | % | + 1.3 | % | + 30.8 | % | + 8.0 | % | + 20.5 | % | + 5.8 | % | ||||||||||||||||||||||||||||||||||
| — Change in local currencies | - 3.0 | % | - 4.8 | % | + 0.6 | % | + 8.0 | % | + 32.9 | % | + 1.4 | % | + 32.4 | % | + 7.3 | % | + 21.5 | % | + 5.3 | % | ||||||||||||||||||||||||||||||||||
| Intersegment sales | 15 | 17 | 3 | 1 | 4 | 7 | 37 | 43 | (73 | ) | (80 | ) | ||||||||||||||||||||||||||||||||||||||||||
| EBITDA** | 235 | 277 | 71 | 91 | 215 | 178 | 249 | 293 | 43 | 15 | 1,101 | 1,308 | ||||||||||||||||||||||||||||||||||||||||||
Special items | (21 | ) | 0 | (4 | ) | 0 | 0 | 0 | (10 | ) | (18 | ) | 10 | 0 | (106 | ) | (34 | ) | ||||||||||||||||||||||||||||||||||||
EBITDA before special items | 256 | 277 | 75 | 91 | 215 | 178 | 259 | 311 | 33 | 15 | 1,207 | 1,342 | ||||||||||||||||||||||||||||||||||||||||||
| EBITDA margin before special items | 19.4 | % | 21.8 | % | 26.2 | % | 29.4 | % | 20.6 | % | 16.8 | % | 15.3 | % | 17.1 | % | 18.1 | % | 19.0 | % | ||||||||||||||||||||||||||||||||||
| EBIT* | 110 | 159 | 52 | 71 | 162 | 102 | 165 | 217 | (1 | ) | (26 | ) | 707 | 878 | ||||||||||||||||||||||||||||||||||||||||
Special items | (21 | ) | 0 | (4 | ) | 0 | 0 | (16 | ) | (10 | ) | (18 | ) | 10 | 0 | (106 | ) | (50 | ) | |||||||||||||||||||||||||||||||||||
| EBIT before special items | 131 | 159 | 56 | 71 | 162 | 118 | 175 | 235 | (11 | ) | (26 | ) | 813 | 928 | ||||||||||||||||||||||||||||||||||||||||
| EBIT margin before special items | 9.9 | % | 12.5 | % | 19.6 | % | 23.0 | % | 15.5 | % | 11.1 | % | 10.4 | % | 12.9 | % | 12.2 | % | 13.1 | % | ||||||||||||||||||||||||||||||||||
| Gross cash flow* | 182 | 227 | 49 | 62 | 149 | 141 | 179 | 222 | 91 | (24 | ) | 867 | 964 | |||||||||||||||||||||||||||||||||||||||||
| Net cash flow* | 493 | 434 | 120 | 100 | 80 | 101 | 189 | 163 | (88 | ) | (270 | ) | 980 | 895 | ||||||||||||||||||||||||||||||||||||||||
| Depreciation, amortization and write-downs/write-backs | 125 | 118 | 19 | 20 | 53 | 76 | 84 | 76 | 44 | 41 | 394 | 430 | ||||||||||||||||||||||||||||||||||||||||||
| 1st Half | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||||||||||||||||
| Net sales (external) | 2,735 | 2,682 | 613 | 667 | 1,968 | 2,094 | 3,310 | 3,600 | 626 | 685 | 13,072 | 14,188 | ||||||||||||||||||||||||||||||||||||||||||
| — Change | - 1.2 | % | - 1.9 | % | + 1.2 | % | + 8.8 | % | + 31.2 | % | + 6.4 | % | + 34.1 | % | + 8.8 | % | + 18.4 | % | + 8.5 | % | ||||||||||||||||||||||||||||||||||
| — Change in local currencies | - 1.3 | % | - 5.3 | % | + 2.5 | % | + 5.5 | % | + 33.7 | % | + 4.3 | % | + 36.0 | % | + 6.1 | % | + 19.6 | % | + 5.8 | % | ||||||||||||||||||||||||||||||||||
| Intersegment sales | 28 | 35 | 8 | 3 | 7 | 13 | 74 | 82 | (148 | ) | (161 | ) | ||||||||||||||||||||||||||||||||||||||||||
| EBITDA** | 678 | 683 | 185 | 236 | 427 | 394 | 570 | 550 | 88 | 14 | 2,483 | 2,790 | ||||||||||||||||||||||||||||||||||||||||||
Special items | (30 | ) | 0 | (4 | ) | 0 | 0 | 0 | (10 | ) | (130 | ) | 0 | (10 | ) | (244 | ) | (162 | ) | |||||||||||||||||||||||||||||||||||
EBITDA before special items | 708 | 683 | 189 | 236 | 427 | 394 | 580 | 680 | 88 | 24 | 2,727 | 2,952 | ||||||||||||||||||||||||||||||||||||||||||
| EBITDA margin before special items | 25.9 | % | 25.5 | % | 30.8 | % | 35.4 | % | 21.7 | % | 18.8 | % | 17.5 | % | 18.9 | % | 20.9 | % | 20.8 | % | ||||||||||||||||||||||||||||||||||
| EBIT* | 432 | 444 | 144 | 194 | 321 | 262 | 412 | 396 | 0 | (75 | ) | 1,693 | 1,955 | |||||||||||||||||||||||||||||||||||||||||
Special items | (30 | ) | 0 | (4 | ) | 0 | 0 | (16 | ) | (10 | ) | (130 | ) | 0 | (10 | ) | (244 | ) | (178 | ) | ||||||||||||||||||||||||||||||||||
| EBIT before special items | 462 | 444 | 148 | 194 | 321 | 278 | 422 | 526 | 0 | (65 | ) | 1,937 | 2,133 | |||||||||||||||||||||||||||||||||||||||||
| EBIT margin before special items | 16.9 | % | 16.6 | % | 24.1 | % | 29.1 | % | 16.3 | % | 13.3 | % | 12.7 | % | 14.6 | % | 14.8 | % | 15.0 | % | ||||||||||||||||||||||||||||||||||
| Gross cash flow* | 489 | 512 | 129 | 164 | 292 | 304 | 397 | 413 | 242 | 69 | 1,927 | 2,090 | ||||||||||||||||||||||||||||||||||||||||||
| Net cash flow* | 170 | 145 | 64 | 39 | 144 | 162 | 125 | 401 | (2 | ) | (198 | ) | 709 | 959 | ||||||||||||||||||||||||||||||||||||||||
| Depreciation, amortization and write-downs/write-backs | 246 | 239 | 41 | 42 | 106 | 132 | 158 | 154 | 88 | 89 | 790 | 835 | ||||||||||||||||||||||||||||||||||||||||||
2005 figures restated
| * | for definition see Bayer Group Key Data on page 2 | |
| ** | ebitda =ebit plus amortization of intangible assets and depreciation of property, plant and equipment.ebitda,ebitda before special items andebitda margin are not defined in the International Financial Reporting Standards and should therefore be regarded only as supplementary information. The company considers underlyingebitda to be a more suitable indicator of operating performance since it is not affected by depreciation, amortization, write-downs/write-backs or special items. The company also believes that this indicator gives readers a clearer picture of the results of operations and ensures greater comparability of data over time. The underlyingebitda margin is calculated by dividing underlyingebitda by sales. |
37
Table of Contents
Interim Report as of June 30, 2006/Notes
Key Data by Region
€ million
| Europe | North America | |||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
| Net sales (external) – by market | 3,047 | 3,169 | 1,768 | 1,908 | ||||||||||||||
| – Change | + 32.7 | % | + 4.0 | % | + 5.0 | % | + 7.9 | % | ||||||||||
| – Change in local currencies | + 32.5 | % | + 3.9 | % | + 8.4 | % | + 6.3 | % | ||||||||||
| Net sales (external) – by point of origin | 3,277 | 3,438 | 1,781 | 1,921 | ||||||||||||||
| – Change | + 32.1 | % | + 4.9 | % | + 3.5 | % | + 7.9 | % | ||||||||||
| – Change in local currencies | + 31.9 | % | + 4.8 | % | + 6.9 | % | + 6.3 | % | ||||||||||
| Interregional sales | 897 | 976 | 587 | 452 | ||||||||||||||
| EBIT* | 455 | 529 | 106 | 238 | ||||||||||||||
| Gross cash flow* | 504 | 589 | 224 | 253 | ||||||||||||||
| 1st Half | 2005 | 2006 | 2005 | 2006 | ||||||||||||||
| Net sales (external) – by market | 6,030 | 6,342 | 3,433 | 3,930 | ||||||||||||||
| – Change | + 28.6 | % | + 5.2 | % | + 4.7 | % | + 14.5 | % | ||||||||||
| – Change in local currencies | + 28.3 | % | + 5.0 | % | + 8.2 | % | + 8.3 | % | ||||||||||
| Net sales (external) – by point of origin | 6,469 | 6,838 | 3,460 | 3,964 | ||||||||||||||
| – Change | + 27.3 | % | + 5.7 | % | + 4.6 | % | + 14.6 | % | ||||||||||
| – Change in local currencies | + 27.1 | % | + 5.6 | % | + 8.3 | % | + 8.3 | % | ||||||||||
| Interregional sales | 1,938 | 2,095 | 971 | 952 | ||||||||||||||
| EBIT* | 999 | 1,212 | �� | 367 | 506 | |||||||||||||
| Gross cash flow* | 1,137 | 1,304 | 470 | 520 | ||||||||||||||
| 2005 figures restated | ||
| * | for definition see Bayer Group Key Data on page 2 |
38
Table of Contents
Interim Report as of June 30, 2006/Notes
€ million
| Asia/ | Latin America/ | Continuing | ||||||||||||||||||||||||||||||||||
| Pacific | Africa/Middle East | Reconciliation | Operations | |||||||||||||||||||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Net sales (external) – by market | 1,090 | 1,136 | 781 | 859 | 6,686 | 7,072 | ||||||||||||||||||||||||||||||
| – Change | + 17.7 | % | + 4.2 | % | + 21.8 | % | + 10.0 | % | + 20.5 | % | + 5.8 | % | ||||||||||||||||||||||||
| – Change in local currencies | + 19.8 | % | + 4.9 | % | + 19.5 | % | + 9.2 | % | + 21.5 | % | + 5.3 | % | ||||||||||||||||||||||||
| Net sales (external) – by point of origin | 1,048 | 1,082 | 580 | 631 | 6,686 | 7,072 | ||||||||||||||||||||||||||||||
| – Change | + 21.2 | % | + 3.2 | % | + 20.6 | % | + 8.8 | % | + 20.5 | % | + 5.8 | % | ||||||||||||||||||||||||
| – Change in local currencies | + 23.4 | % | + 3.9 | % | + 17.2 | % | + 7.5 | % | + 21.5 | % | + 5.3 | % | ||||||||||||||||||||||||
| Interregional sales | 42 | 53 | 40 | 40 | (1,566 | ) | (1,521 | ) | ||||||||||||||||||||||||||||
| EBIT* | 138 | 91 | 55 | 61 | (47 | ) | (41 | ) | 707 | 878 | ||||||||||||||||||||||||||
| Gross cash flow* | 140 | 103 | 37 | 55 | (38 | ) | (36 | ) | 867 | 964 | ||||||||||||||||||||||||||
| 1st Half | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Net sales (external) – by market | 2,077 | 2,208 | 1,532 | 1,708 | 13,072 | 14,188 | ||||||||||||||||||||||||||||||
| – Change | + 17.2 | % | + 6.3 | % | + 18.3 | % | + 11.5 | % | + 18.4 | % | + 8.5 | % | ||||||||||||||||||||||||
| – Change in local currencies | + 19.5 | % | + 4.2 | % | + 17.6 | % | + 5.4 | % | + 19.6 | % | + 5.8 | % | ||||||||||||||||||||||||
| Net sales (external) – by point of origin | 1,992 | 2,104 | 1,151 | 1,282 | 13,072 | 14,188 | ||||||||||||||||||||||||||||||
| – Change | + 20.4 | % | + 5.6 | % | + 15.8 | % | + 11.4 | % | + 18.4 | % | + 8.5 | % | ||||||||||||||||||||||||
| – Change in local currencies | + 22.8 | % | + 3.4 | % | + 14.7 | % | + 3.5 | % | + 19.6 | % | + 5.8 | % | ||||||||||||||||||||||||
| Interregional sales | 96 | 119 | 77 | 82 | (3,082 | ) | (3,248 | ) | ||||||||||||||||||||||||||||
| EBIT* | 279 | 216 | 133 | 105 | (85 | ) | (84 | ) | 1,693 | 1,955 | ||||||||||||||||||||||||||
| Gross cash flow* | 276 | 234 | 97 | 93 | (53 | ) | (61 | ) | 1,927 | 2,090 | ||||||||||||||||||||||||||
39
Table of Contents
Interim Report as of June 30, 2006/Notes
Notes to the Interim Report as of June 30, 2006
Accounting policies
The unaudited, consolidated interim financial statements as of June 30, 2006, have been prepared according to the rules of IAS 34. The statements comply with the International Financial Reporting Standards (IFRS) approved and published by the International Accounting Standards Board (IASB) and in effect at the closing date, and with their interpretations issued by the International Financial Reporting Interpretations Committee (IFRIC).
Reference should be made as appropriate to the notes to the 2005 financial statements, particularly with regard to recognition and valuation principles.
Information on earnings per share
The ordinary shares to be issued upon conversion of the mandatory convertible bond are treated as already issued shares. Diluted earnings per share are therefore equal to basic earnings per share.
Calculation of Earnings per Share | ||||||||||||||||||
| 2nd Quarter | 2nd Quarter | 1st Half | 1st Half | |||||||||||||||
| 2005 | 2006 | 2005 | 2006 | |||||||||||||||
From continuing operations | ||||||||||||||||||
Income from continuing operations after taxes (€ million)* | 404 | 446 | 992 | 1,025 | ||||||||||||||
+financing expenses for the mandatory convertible bond, net of tax effects (€ million) | — | 23 | — | 23 | ||||||||||||||
Adjusted income from continuing operations after taxes (€ million) | 404 | 469 | 992 | 1,048 | ||||||||||||||
| Number of ordinary shares issued (million) | 730.34 | 730.34 | 730.34 | 730.34 | ||||||||||||||
| Potential shares to be issued upon conversion of the mandatory convertible bond (million) | — | 63.25 | — | 31.80 | ||||||||||||||
Adjusted weighted average number of outstanding ordinary shares (million) | 730.34 | 793.59 | 730.34 | 762.14 | ||||||||||||||
Basic earnings per share from continuing operations (€) | 0.55 | 0.59 | 1.36 | 1.38 | ||||||||||||||
Diluted earnings per share from continuing operations (€) | 0.55 | 0.59 | 1.36 | 1.38 | ||||||||||||||
From continuing and discontinued operations | ||||||||||||||||||
Net income (€ million) | 406 | 452 | 1,058 | 1,052 | ||||||||||||||
+financing expenses for the mandatory convertible bond, net of tax effects (€ million) | — | 23 | — | 23 | ||||||||||||||
Adjusted net income (€ million) | 406 | 475 | 1,058 | 1,075 | ||||||||||||||
| Number of ordinary shares issued (million) | 730.34 | 730.34 | 730.34 | 730.34 | ||||||||||||||
| Potential shares to be issued upon conversion of the mandatory convertible bond (million) | — | 63.25 | — | 31.80 | ||||||||||||||
Adjusted weighted average number of outstanding ordinary shares (million) | 730.34 | 793.59 | 730.34 | 762.14 | ||||||||||||||
Basic earnings per share from continuing and discontinued operations (€) | 0.56 | 0.60 | 1.45 | 1.41 | ||||||||||||||
Diluted earnings per share from continuing and discontinued operations (€) | 0.56 | 0.60 | 1.45 | 1.41 | ||||||||||||||
| 2005 figures restated | ||
| * | excluding minority interest |
40
Table of Contents
Interim Report as of June 30, 2006/Notes
Changes in the Bayer Group
Scope of consolidation
As of June 30, 2006, the Bayer Group comprised 434 fully or proportionately consolidated companies, compared with 283 companies as of December 31, 2005. This increase of 151 is largely due to the first-time inclusion of the Schering group companies.
Acquisition and first-time consolidation of Schering
With effect from June 23, 2006, Bayer acquired a majority of the shares of Schering AG, which is fully consolidated in the Bayer Group financial statements as of that date. As of June 30, 2006, Bayer held 89.7 percent of the outstanding shares of Schering AG. In addition to the purchase price of €15,073 million for these shares, ancillary acquisition costs of €55 million were incurred up to the closing date for the interim financial statements. The acquisition was paid for in cash.
The assets, liabilities and contingent liabilities acquired from Schering were reflected in the balance sheet at the following fair values:
| Schering Acquisition | |||||||||||||
| € million | Net carrying | Adjustment | Net carrying | ||||||||||
| amount prior to the | for the first-time | amount after the | |||||||||||
| acquisition | consolidation* | acquisition | |||||||||||
| Goodwill | 364 | 4,845 | 5,209 | ||||||||||
| Other intangible assets | 297 | 11,125 | 11,422 | ||||||||||
| Property, plant and equipment | 1,124 | 412 | 1,536 | ||||||||||
| Inventories | 836 | 1,013 | 1,849 | ||||||||||
| Financial liabilities | (241 | ) | — | (241 | ) | ||||||||
| Liquid assets | 1,025 | — | 1,025 | ||||||||||
| Other assets and liabilities | (301 | ) | (100 | ) | (401 | ) | |||||||
| Deferred taxes | 295 | (4,432 | ) | (4,137 | ) | ||||||||
| Net assets | 3,399 | 12,863 | 16,262 | ||||||||||
| Minority interests | (1,134 | ) | |||||||||||
| Acquisition price | 15,128 | ||||||||||||
| of which ancillary acquisition costs | 55 | ||||||||||||
| * | The adjustment for the first-time consolidation reflects the differences between the previous net carrying amounts in the balance sheet of Schering and the respective fair values in the acquirer’s balance sheet at the date of acquisition. |
The average expected useful life of the acquired intangible assets is approximately 13 years.
The purchase price allocation has not yet been completed, therefore changes may yet be made in the allocation of the purchase price to the individual assets.
The goodwill remaining after the purchase price allocation is attributable to a number of factors. Apart from general synergies in administration processes and infrastructures, such factors also include significant cost savings in the areas of marketing, sales, procurement and production that cannot be fully realized until a domination agreement with Schering AG comes into effect. In addition, the acquisition strengthens the Bayer Group’s global market position in the pharmaceuticals business. Details of the legal form of the merger are still in the planning stage.
41
Table of Contents
Interim Report as of June 30, 2006/Notes
The income, expenses and cash flows for the Schering business, including pro-rata effects from the purchase price allocation, were recognized as follows from the date of the first-time consolidation (June 23, 2006):
Schering Key Data | |||||
| € million | June 23 – June 30, 2006 | ||||
| Sales | 144 | ||||
| EBITDA* | 20 | ||||
| EBITDA before special items | 30 | ||||
| EBIT* | (6 | ) | |||
| EBIT before special items | 4 | ||||
| Income after taxes | (3 | ) | |||
| Gross cash flow* | 25 | ||||
| Net cash flow* | 145 | ||||
| Net investing cash flow | (1 | ) | |||
| Net financing cash flow | (4 | ) | |||
| * | for definition see Bayer Group Key Data on page 2 |
Discontinued operations
Bayer has entered into an agreement with Siemens AG concerning the divestiture of the Diagnostics Division. The Diagnostics business is thus reported as a discontinued operation. The prior-year data in the income and cash flow statements have beem restated accordingly.
In the prior year, the spin-off of Lanxess from Bayer AG was entered into the commercial register on January 28, 2005 and thus became legally effective. The Plasma business of the Bayer HealthCare subgroup in the United States was divested in March 2005. Both these businesses are reported for 2005 as discontinued operations.
This information, which is provided from the standpoint of the Bayer Group, is to be regarded as part of the reporting for the entire Group by analogy with our segment reporting and is not intended to portray either the discontinued operations or the remaining business of Bayer as separate entities. The presentation is thus in line with the principles for reporting discontinued operations.
Discontinued Operations | ||||||||||||||||||||||||||||||||||||
| €million | Diagnostics | Lanxess | Plasma | Total | ||||||||||||||||||||||||||||||||
| 2nd Quarter | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Net sales | 367 | 377 | 0 | 0 | 4 | 0 | 371 | 377 | ||||||||||||||||||||||||||||
| EBIT* | 39 | 9 | 0 | 0 | (36 | ) | 0 | 3 | 9 | |||||||||||||||||||||||||||
| Income after taxes | 25 | 6 | 0 | 0 | (23 | ) | 0 | 2 | 6 | |||||||||||||||||||||||||||
| Gross cash flow* | 41 | 50 | 0 | 0 | 6 | 0 | 47 | 50 | ||||||||||||||||||||||||||||
| Net cash flow* | 35 | 107 | 0 | 0 | 10 | 0 | 45 | 107 | ||||||||||||||||||||||||||||
| Net investing cash flow | (28 | ) | (17 | ) | 0 | 0 | 0 | 0 | (28 | ) | (17 | ) | ||||||||||||||||||||||||
| Net financing cash flow | (7 | ) | (90 | ) | 0 | 0 | (10 | ) | 0 | (17 | ) | (90 | ) | |||||||||||||||||||||||
| 1st Half | 2005 | 2006 | 2005** | 2006 | 2005 | 2006 | 2005 | 2006 | ||||||||||||||||||||||||||||
| Net sales | 685 | 755 | 503 | 0 | 124 | 0 | 1,312 | 755 | ||||||||||||||||||||||||||||
| EBIT* | 57 | 40 | 62 | 0 | (14 | ) | 0 | 105 | 40 | |||||||||||||||||||||||||||
| Income after taxes | 37 | 27 | 38 | 0 | (9 | ) | 0 | 66 | 27 | |||||||||||||||||||||||||||
| Gross cash flow* | 82 | 114 | 51 | 0 | 4 | 0 | 137 | 114 | ||||||||||||||||||||||||||||
| Net cash flow* | 80 | 171 | (80 | ) | 0 | 58 | 0 | 58 | 171 | |||||||||||||||||||||||||||
| Net investing cash flow | (48 | ) | (46 | ) | (19 | ) | 0 | 226 | 0 | 159 | (46 | ) | ||||||||||||||||||||||||
| Net financing cash flow | (32 | ) | (125 | ) | 99 | 0 | (284 | ) | 0 | (217 | ) | (125 | ) | |||||||||||||||||||||||
| * | for definition see Bayer Group Key Data on page 2 | |
| ** | figures for January only |
42
Table of Contents
Interim Report as of June 30, 2006/Notes
Segment reporting
We have adapted our segment reporting to the changes in our corporate structure. The acquired Schering business is now included in the Pharmaceuticals segment together with that of our existing Pharmaceuticals Division.
The businesses of the Diabetes Care and Diagnostics divisions were previously combined for reporting purposes, while the Consumer Care and Animal Health divisions were reported as separate segments. Due to the agreed divestiture of the Bayer HealthCare subgroup’s Diagnostics Division, the segment reporting has been adjusted. As a discontinued operation, the Diagnostics Division is no longer part of the segment reporting. The remaining Diabetes Care Division is combined with the Consumer Care and Animal Health divisions to form the new Consumer Health segment in light of the similarities in their long-term financial performance and their focus on products that can be promoted directly to consumers. The previous year’s figures are restated accordingly.
Resolutions of the Annual Stockholders’
Meeting
The Annual Stockholders’ Meeting on April 28, 2006 ratified the actions of the members of the Supervisory Board and Board of Management.
Existing authorized capital was revoked and new authorized capital created, and the Articles of Incorporation were amended accordingly. The Board of Management also received authorization to purchase and sell shares of the company.
The stockholders approved the Domination and Profit Transfer Agreement between Bayer AG and Bayfin GmbH. In addition, the Articles of Incorporation were amended in accordance with the proposal regarding the rights of the chairman of the Annual Stockholders’ Meeting.
PricewaterhouseCoopers Aktiengesellschaft, Wirtschaftsprüfungsgesellschaft, Essen, was appointed as auditor for the 2006 fiscal year.
The€694 million balance sheet profit of Bayer AG for fiscal 2005 was used to pay a dividend of€0.95 per share for that year. The dividend paid for 2004 was€0.55 per share.
Changes on the Board of Management and Supervisory Board
Following the Annual Stockholders’ Meeting, Dr. Udo Oels retired from the Board of Management of Bayer AG. He is succeeded by Dr. Wolfgang Plischke, who was appointed to the Board with effect from March 1, 2006.
At its meeting on June 29, the Supervisory Board extended Werner Wenning’s contract as Management Board Chairman until 2010. Wenning’s contract would have expired in 2007. The contracts of Chief Financial Officer Klaus Kühn and Labor Director Dr. Richard Pott, which also would have expired in 2007, were each extended by five years until 2012.
At the end of the Annual Stockholders’ Meeting, Peter Hausmann succeeded Siegfried Wendlandt as a member of the Supervisory Board.
Leverkusen, August 22, 2006
Bayer Aktiengesellschaft
The Board of Management
The Board of Management
43
Table of Contents
Forward-Looking Statements
This Stockholders’ Newsletter contains forward-looking statements. These statements use words like “believes”, “assumes”, “expects” or similar formulations. Various known and unknown risks, uncertainties and other factors could lead to material differences between the actual future results, financial situation, development or performance of our company and those either expressed or implied by these statements.
These factors include, among other things:
| • | Downturns in the business cycle of the industries in which we compete; | |
| • | new regulations, or changes to existing regulations, that increase our operating costs or otherwise reduce our profitability; | |
| • | increases in the price of our raw materials, especially if we are unable to pass these costs along to customers; | |
| • | loss or reduction of patent protection for our products; | |
| • | liabilities, especially those incurred as a result of environmental laws or product liability litigation; | |
| • | fluctuation in international currency exchange rates as well as changes in the general economic climate; and | |
| • | other factors identified in this Stockholders’ Newsletter . |
These factors include those discussed in our public reports filed with the Frankfurt Stock Exchange and with the U.S. Securities and Exchange Commission (including Form 20-F). In view of these uncertainties, we caution readers not to place undue reliance on these forward-looking statements. We assume no liability whatsoever to update these forward-looking statements or to conform them to future events or developments.
44
Table of Contents
Financial Calendar
Q3 2006 Interim Report
Monday, November 27, 2006
Monday, November 27, 2006
2006 Annual Report
Thursday, March 15, 2007
Thursday, March 15, 2007
Annual Stockholders’ Meeting 2007
Friday, April 27, 2007
Friday, April 27, 2007
Payment of Dividend
Monday, April 30, 2007
Monday, April 30, 2007
MASTHEAD
Publisher
Bayer AG
51368 Leverkusen, Germany
Editor
Ute Bode, Phone ++49 214 30 58992
Email: ute.bode.ub@bayer-ag.de
English Edition
Bayer Industry Services GmbH & Co. OHG
Central Language Service
Investor Relations
Peter Dahlhoff, Phone ++49 214 30 33022
Email: peter.dahlhoff@bayer-ag.de
51368 Leverkusen, Germany
Editor
Ute Bode, Phone ++49 214 30 58992
Email: ute.bode.ub@bayer-ag.de
English Edition
Bayer Industry Services GmbH & Co. OHG
Central Language Service
Investor Relations
Peter Dahlhoff, Phone ++49 214 30 33022
Email: peter.dahlhoff@bayer-ag.de
Date of Publication
Tuesday, August 29, 2006
Bayer on the Internet
WWW.BAYER.COM
Tuesday, August 29, 2006
Bayer on the Internet
WWW.BAYER.COM

Science For A Better Life
45
Table of Contents
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| BAYER AKTIENGESELLSCHAFT (Registrant) | ||||
| By: | /s/ DR. ROLAND HARTWIG | |||
| Name: | Dr. Roland Hartwig | |||
| Title: | General Counsel | |||
| By: | /s/ DR. ALEXANDER ROSAR | |||
| Name: | Dr. Alexander Rosar | |||
| Title: | Head of Investor Relations | |||
Date: August 29, 2006