Exhibit 99.1

| Company Overview May 2008 |

| Safe Harbor The Private Securities Litigation Reform Act of 1995 provides a "safe harbor" for forward-looking statements. All statements made in this presentation that are not statements of historical fact constitute forward-looking statements. The matters referred to in forward-looking statements could be affected by the risks and uncertainties of the Company's business. Such risks inherent to the Company's business will be described in the Company's filings, when they occur, with the Securities and Exchange Commission, as well as in its press releases. The Company's actual results may differ materially from those expressed in or indicated by such forward-looking statements. |

| HALO Investment Highlights Three partnered programs validate rHuPH20 core technology FDA approved product, HYLENEX, with large untapped potential Enhanze Technology partnerships with Roche and Baxter may generate additional cash and royalty payments Five proprietary product candidates target extracellular matrix ("Matrix"), focusing on oncology, dermatology, and metabolism Combinations of rHuPH20 with small molecule drugs and proteins Novel Matrix therapeutics targeting major indications, based on a deep understanding of Matrix science Attractive valuation with $93 million in cash at 1Q08 |
| HYLENEX: Low dose FDA-approved rHuPH20 drug product for delivery of small molecules and fluids Enhanze Technology: High dose rHuPH20 for delivery of large molecules (e.g., monoclonal antibodies) Proprietary rHuPH20 Combinations: Co-formulation with approved products to generate proprietary HALO products or create high value business development opportunities 3-D model of rHuPH20 hyaluronidase Multi-Functional rHuPH20 Core Technology can Drive Value in Many Different Ways |

| Proprietary Product Candidates Target the Matrix Proprietary rHuPH20 combinations Insulin (diabetes) Bisphosphonate (osteoporosis) Chemophase (bladder cancer) Novel Matrix therapeutics PEGrHuPH20 (cancer) HTI-501 (dermatology) |

| 1. Insulin Program Highlights Completed Phase I, prospective, randomized trial designed to assess safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of two different approved insulin products injected SC with and without rHuPH20. Poster presentation at ADA next month We anticipate rHuPH20 will favorably alter PK/PD of proteins based on existing preclinical data and several completed clinical trials with other model compounds If co-injection is well tolerated and PK/PD benefit is observed, then combination product may result in meaningful clinical benefit for patients Potential to develop best-in-class therapeutic within a $10 billion worldwide insulin market Anticipate Phase II clinical trial later this year |

| 2. Bisphosphonate Program Highlights >$4 billion annual market, but long-term compliance is poor due to insufficient efficacy, gastrointestinal toxicity, and difficult dosing regimens; >75% of patients discontinue therapy within 3 years IV BPs are inconvenient to patients and general practitioners have limited capabilities to administer them IV-administered Reclast and Boniva are attractive targets to explore for converting to SC with rHuPH20 Injection of BPs with rHuPH20 may facilitate IV to SC conversion and help ensure compliance via convenient annual/semi annual dosing and elimination of GI toxicity Targeting to enter clinic for at least one of these combinations in 3Q08 |
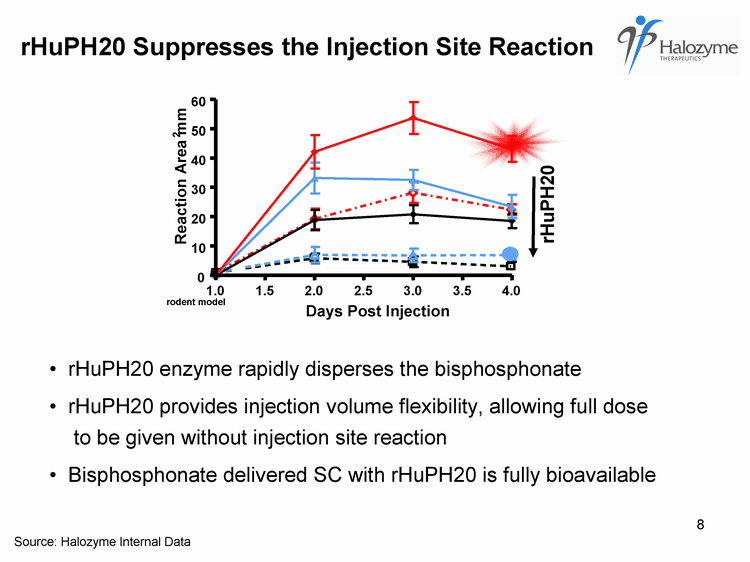
| rHuPH20 Suppresses the Injection Site Reaction 1.0 1.5 2.0 2.5 3.0 3.5 4.0 0 10 20 30 40 50 60 Reaction Area mm 2 rHuPH20 Days Post Injection rHuPH20 enzyme rapidly disperses the bisphosphonate rHuPH20 provides injection volume flexibility, allowing full dose to be given without injection site reaction Bisphosphonate delivered SC with rHuPH20 is fully bioavailable rodent model Source: Halozyme Internal Data |

| 3. Chemophase Program Highlights Tumor recurrence rates for superficial bladder cancer are 40-85%; 50% will recur within first year Chemophase may increase absorption of Mitomycin C into the bladder wall to decrease recurrence rates Ongoing Chemophase Phase I/IIa trial successful in determining MTD and demonstrating safety and tolerability of induction and maintenance dosing; enrollment completed 3Q07, data anticipated 2Q08 Currently preparing to consult with regulatory authorities, including end- of-Phase II meeting with FDA and Scientific Advice for EU, in order to determine optimal regulatory pathway to drug approval Initiation of next clinical trial anticipated in 4Q08 |
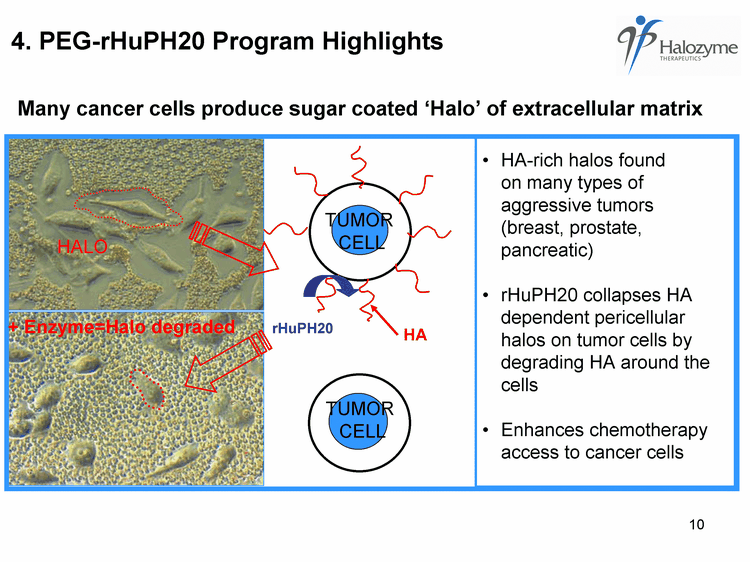
| 4. PEG-rHuPH20 Program Highlights HA-rich halos found on many types of aggressive tumors (breast, prostate, pancreatic) rHuPH20 collapses HA dependent pericellular halos on tumor cells by degrading HA around the cells Enhances chemotherapy access to cancer cells HA TUMOR CELL + Enzyme=Halo degraded HALO rHuPH20 TUMOR CELL Many cancer cells produce sugar coated 'Halo' of extracellular matrix |

| IV PEG-rHuPH20 Degrades Tumor HA and Rapidly Reduces Tumor Interstitial Fluid Pressure IM PC-3 tumor pressure measured 20 minutes prior and for 2 hours following IV injection of 10,000 units of PEG^rHuPH20 (n=3), or Carrier Buffer (n=3) (standard deviation indicated) >80% reduction in tumor IFP With PEG-rHuPH20 within 60 minutes |
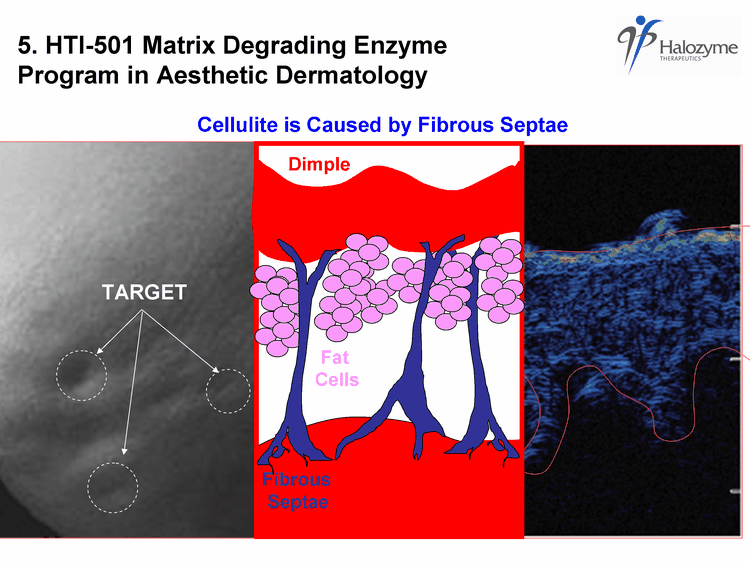
| TARGET fibrous septae create dimples Fat Cells Fibrous Septae Dimple 5. HTI-501 Matrix Degrading Enzyme Program in Aesthetic Dermatology Cellulite is Caused by Fibrous Septae |
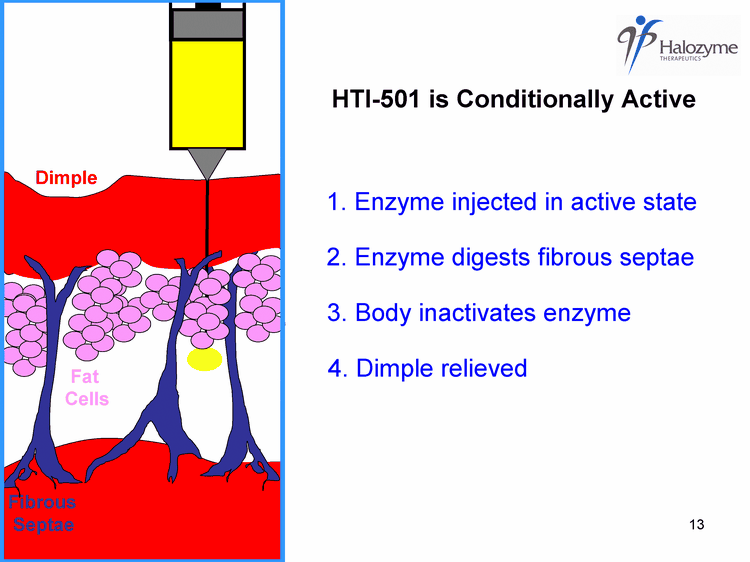
| fibrous septae create dimples 1. Enzyme injected in active state 2. Enzyme digests fibrous septae 3. Body inactivates enzyme Fat Cells Fibrous Septae 4. Dimple relieved Dimple HTI-501 is Conditionally Active |

| 2008 Milestones Publication of INFUSE-LR data, 1Q08 Presentation of GammaGard Phase I/IIa data, 1Q08 Presentation of Bisphosphonates pre-clinical data, 2Q08 Presentation of PEGrHuPH20 pre-clinical IFP data, 2Q08 Presentation of Insulin Phase I data, ADA, 2Q08 Presentation of Cellulite pre-clinical data, 2Q08 Release of Chemophase Phase I/IIa data, 2Q08 Presentation of PEGrHuPH20 pre-clinical efficacy data, 3Q08 Initiate clinical trial for Bisphosphonates program, 3Q08 Initiate Insulin Phase II trial, 4Q08 |

| HALO's Unique Investment Thesis Compelling Matrix technology (rHuPH20 and other enzymes) with widespread potential across variety of drug delivery and therapeutic uses Revenue generating assets from partnerships and FDA approved drug product that can help fund growth opportunities Scaleable platform for developing proprietary products with potentially faster time to market and lower development risk New molecular entity generation targeting major indications in oncology, dermatology, and metabolism Attractive valuation with $93 million in cash at 1Q08 |