Exhibit 99.1
| Halozyme Therapeutics Matrix Therapies for Life Jefferies Investor Presentation New York, June 17, 2009 |

| Safe Harbor The Private Securities Litigation Reform Act of 1995 provides a "safe harbor" for forward-looking statements. All statements made in this presentation that are not statements of historical fact constitute forward-looking statements. The matters referred to in forward-looking statements could be affected by the risks and uncertainties of the Company's business. Such risks inherent to the Company's business will be described in the Company's filings, when they occur, with the Securities and Exchange Commission, as well as in its press releases. The Company's actual results may differ materially from those expressed in or indicated by such forward-looking statements. 2 |

| Halozyme Investment Highlights Ability to leverage our PH20 core technology (recombinant human hyaluronidase) across multiple therapeutic areas FDA approved enzyme Strong safety profile and IP protection Unique value proposition for partners and proprietary programs Technology is validated by partnerships with Roche and Baxter worth up to $724 million, plus royalties; $97 million received to date Robust pipeline of Matrix enzymes offer broad potential, better treatments, and patient benefits for variety of therapeutic uses Potential best-in-class rapid-acting insulin product for diabetes NMEs targeting major indications in oncology and dermatology 3 |

| 4 PH20 Core Technology: Targets HA in the "Matrix" |
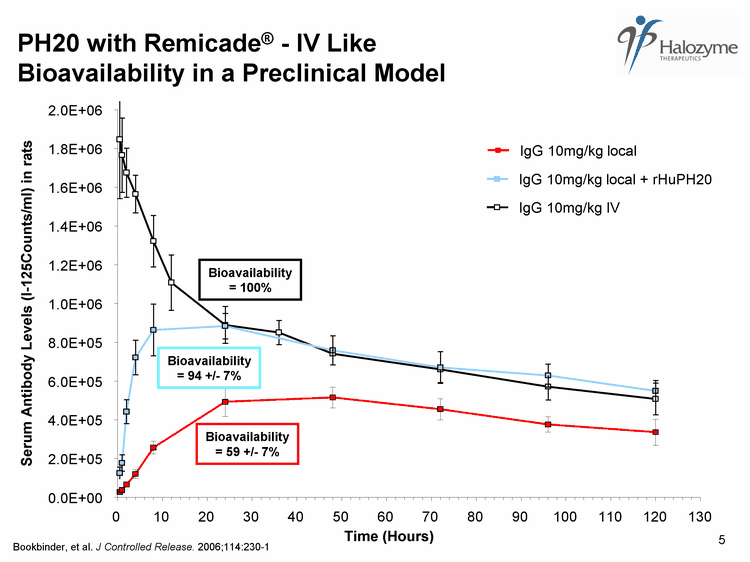
| IgG 10mg/kg local Bioavailability = 59 +/- 7% 0.0E+00 2.0E+05 4.0E+05 6.0E+05 8.0E+05 1.0E+06 1.2E+06 1.4E+06 1.6E+06 1.8E+06 2.0E+06 0 10 20 30 40 50 60 70 80 90 100 110 120 130 Time (Hours) Serum Antibody Levels (I-125Counts/ml) in rats IgG 10mg/kg IV Bioavailability = 100% Bioavailability = 94 +/- 7% PH20 with Remicade(r) - IV Like Bioavailability in a Preclinical Model Bookbinder, et al. J Controlled Release. 2006;114:230-1 5 |

| Robust Pipeline of PH20 Based Product Candidates and Other Matrix Enzymes 6 |

| Partnered Programs Based on Halozyme's Drug Delivery Technology Platform Enhanze Technology with Roche (up to 13 biologic targets) Enhanze Technology with Baxter BioScience (GAMMAGARD) HYLENEX with Baxter Medication Delivery 7 |
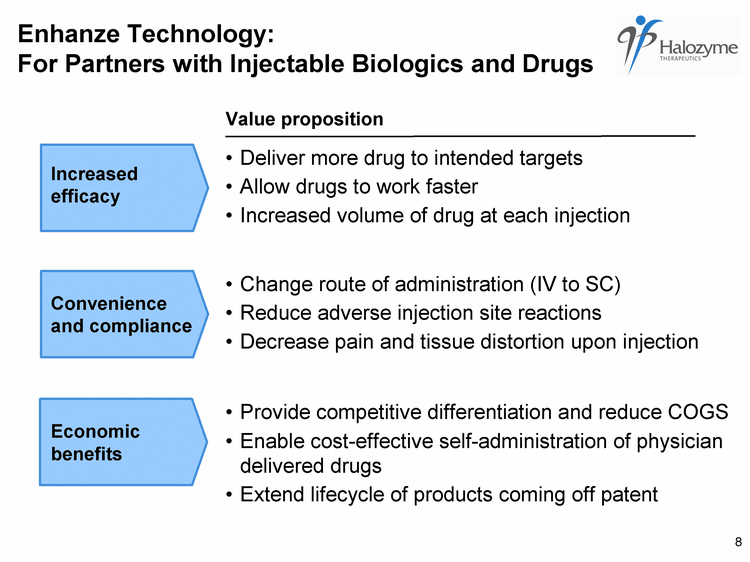
| Change route of administration (IV to SC) Reduce adverse injection site reactions Decrease pain and tissue distortion upon injection Convenience and compliance Economic benefits Value proposition Deliver more drug to intended targets Allow drugs to work faster Increased volume of drug at each injection Increased efficacy Enhanze Technology: For Partners with Injectable Biologics and Drugs 8 |

| Enhanze Technology with Roche (up to 13 biologic targets) Alliance worth up to $612M ($20M up-front; $111M milestones for first 3 exclusive targets; $470M up-front & milestones for 10 additional targets; $11M equity), plus royalties; $47M received to date Enhanze Technology with Baxter BioScience (GAMMAGARD) Alliance worth up to $47M ($10M up-front; $37M milestones), plus royalties; $10M received to date HYLENEX with Baxter Medication Delivery Alliance worth up to $65M ($10M up-front; $25M milestones; $10M pre- paid royalties; $20M equity), plus royalties; $40M received to date Existing Alliances Drive Significant Value 9 |

| Exercised exclusive global rights to a fourth biologic target in December 2008 and fifth target in June of 2009 Two targets are in Phase 1 clinical trials - expected completion mid-2009 Initiation of additional Roche clinical trial(s) plus milestone(s) possible in 2009 Roche Enhanze Technology Alliance Progressing Well "So I think that this Halozyme technology is... an intriguing part of lifecycle management where we think we can add additional clinical utility, save money to society from the cost of [IV infusion] chairs....They are a good partner and I'm encouraged by what we are doing with them." - - Bill Burns, CEO Roche Pharmaceuticals, 2/6/09 Roche Analyst Meeting in NYC 10 |

| $612M partnership signed, 3 exclusive biologic targets elected (Dec'06) YE06 YE07 YE08 YE09 4th exclusive biologic target elected 1st Phase 1 (n=70, mid-09 completion) Annual maintenance fees (Dec'08) 2nd Phase 1 (n=48, mid-09 completion) (Jan'09) 5th exclusive biologic target elected (Jun'09) Additional Roche clinical trial(s) plus milestone(s) possible (2H09) CMC, including manufacturing scale-up, nonclinical, toxicology, and other development activities for product candidates formulated with PH20 (not disclosed) Recent Public Disclosures Reflect Strong Momentum of Roche-Halo Progress |

| GAMMAGARD LIQUID (GGL) - plasma derived immune globulin (IgG) indicated for primary immunodeficiency, currently given by IV infusion Growing $1B+ product franchise in $2.5B worldwide market SC IgG limited by low bioavailability (63%) and small injection site volumes, results in need for weekly dosing at multiple injection sites Phase 1/2a trial showed that SC administration of up to 61.2 grams (612 ml) IgG with PH20 at up to 300 mL per hour resulted in bioavailability equal to 92% of IV administration Potential for convenient patient self-administration once monthly in a single SC injection site Pivotal Phase 3 for GGL with PH20 began in December 2008; enrollment goal of 80 patients, completion of enrollment expected by year-end Enhanzed GAMMAGARD - SC Monthly Dosing at Single Site with Halozyme's PH20 Technology 12 |

| HYLENEX Launch in Pediatric Hydration Market in 4Q09 Dedicated HYLENEX specialty sales force of pediatric reps, supported by Baxter's hospital promotional presence (IV Team sales reps, formulary specialists, etc.) Formulary approval and access received at numerous pediatric hospitals in preparation for launch Pediatric hydration trial manuscript accepted for publication in peer-reviewed journal PEDIATRICS Additional Phase 4 studies currently underway to further demonstrate the potential benefits and utility of subcutaneous vs. IV hydration 13 |

| Proprietary Product Candidates Target the Matrix Insulin-PH20: PH20 combination with insulins for meal time diabetes therapy PEGPH20: Pegylated PH20 targets HA expressing tumors Chemophase: PH20 combination with Mitomycin for bladder cancer HTI-501: Novel Matrix remodeling enzyme for dermatology 14 |

| Goal is to develop best-in-class rapid-acting insulin product that can surpass current analog standard of care by better mimicking physiologic insulin profile Developing proprietary insulin formulations of novel PH20 enzyme with: Fast acting analog insulin (e.g., Humalog, Novolog, Apidra) Short acting regular human insulin (e.g., Humulin R) Program currently in Phase 2, targeting commercialization by 2013 or 2014, depending on most attractive product candidate (analog-PH20 or regular insulin-PH20) Halozyme's Insulin-PH20 Program |
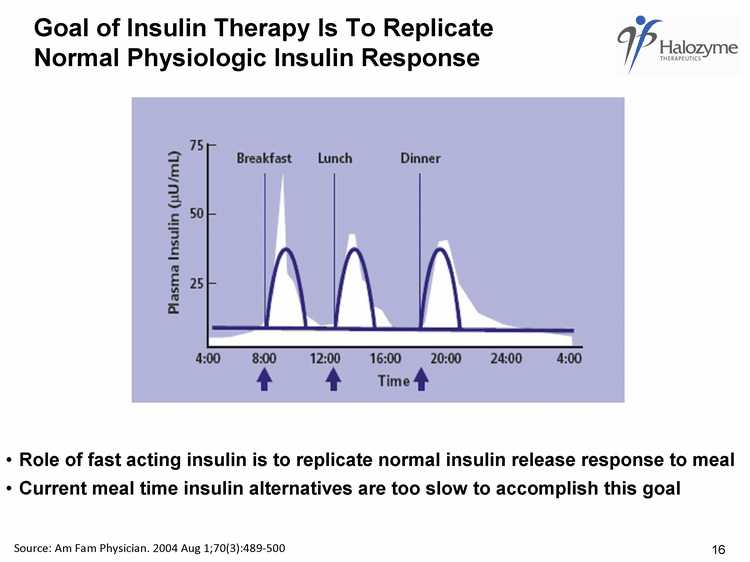
| Goal of Insulin Therapy Is To Replicate Normal Physiologic Insulin Response Source: Am Fam Physician. 2004 Aug 1;70(3):489-500 Role of fast acting insulin is to replicate normal insulin release response to meal Current meal time insulin alternatives are too slow to accomplish this goal 16 |

| Failure to Match Physiologic Insulin Leads to Unmet Needs and Risk of Adverse Outcomes * IMS Data, 2008 GFK Roper Patient Survey, N = 2,000 (projectable) Suboptimal glycemic control: Inability to control post meal blood sugar is a significant impediment to diabetes management <30% of all insulin patients meet ADA A1C goal of 7%* Significant rates of hypoglycemia: 72% of T1DM and 54% of T2DM patients on insulin reported hypoglycemia in the past 3 months* In mild form, quality of life impact is significant In severe form, can be fatal Weight gain: Intensification of insulin therapy and hypoglycemia are also associated with weight gain 17 |
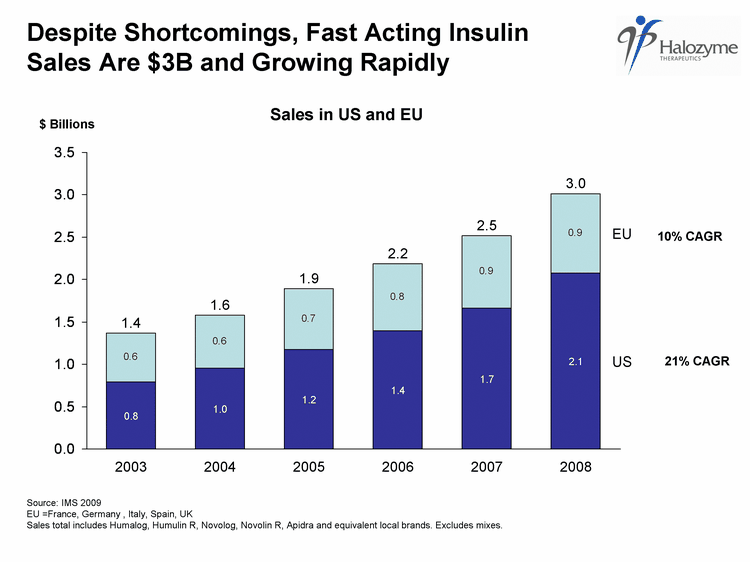
| 1 Source: IMS 2009 EU =France, Germany , Italy, Spain, UK Sales total includes Humalog, Humulin R, Novolog, Novolin R, Apidra and equivalent local brands. Excludes mixes. Despite Shortcomings, Fast Acting Insulin Sales Are $3B and Growing Rapidly 0.79186431500009 0.956185096000109 1.17782748200013 1.40077801900016 1.66158388700019 2.07601576400024 0.572356658000065 0.627907955000071 0.718925928000082 0.785653765000089 0.852118976000097 0.935964672000106 1.0 1.7 2.1 0.8 US 1.2 1.4 2003 2004 2005 2006 2007 2008 EU 1.4 1.6 1.9 2.2 2.5 3.0 21% CAGR 10% CAGR $ Billions Sales in US and EU |

| Phase 2 Meal Study in Type 1 Diabetes 4-way crossover standardized test meal study in Type 1 diabetes Objective: compare PK and glucose profiles in response to Humulin R +/- PH20 and Humalog +/- PH20 Primary endpoint: PK AUC0-60 Secondary endpoints: Multiple PK parameters, glycemic excursion Study design Stabilize fasting blood sugar (FBS) with glucose/insulin infusion Up to 3 dose finding visits to optimize post-meal (12 oz. Ensure = 60 gm CHO) glycemic response to lispro-PH20 Using "optimized" dose, repeat test meal with lispro alone Lispro-PH20 data presented at ADA, New Orleans, June 2009; Final data for regular insulin-PH20 available at EASD, Vienna, Sep/Oct 2009 19 |
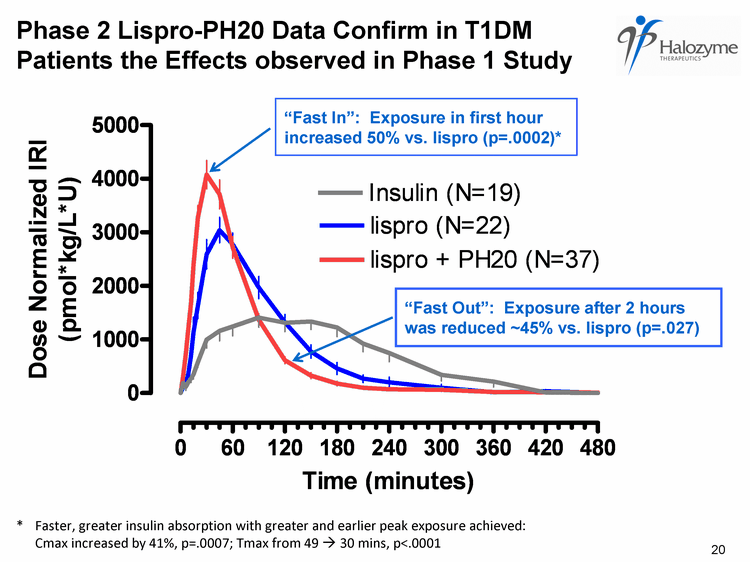
| * Faster, greater insulin absorption with greater and earlier peak exposure achieved: Cmax increased by 41%, p=.0007; Tmax from 49 ? 30 mins, p<.0001 Phase 2 Lispro-PH20 Data Confirm in T1DM Patients the Effects observed in Phase 1 Study "Fast In": Exposure in first hour increased 50% vs. lispro (p=.0002)* "Fast Out": Exposure after 2 hours was reduced ~45% vs. lispro (p=.027) 20 |
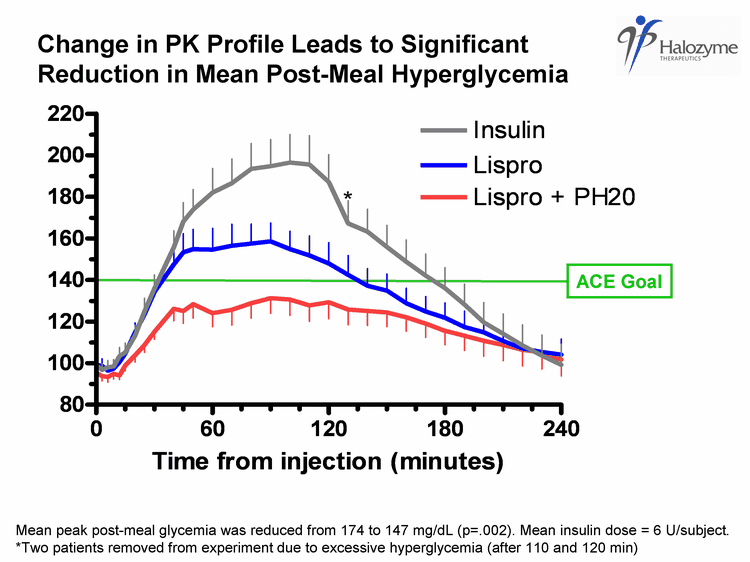
| ACE Goal Change in PK Profile Leads to Significant Reduction in Mean Post-Meal Hyperglycemia * Mean peak post-meal glycemia was reduced from 174 to 147 mg/dL (p=.002). Mean insulin dose = 6 U/subject. *Two patients removed from experiment due to excessive hyperglycemia (after 110 and 120 min) |

| Phase 2 Type 1 Diabetes Meal Study Conclusions Lispro-PH20 well tolerated, confirms in Type 1 patients previous findings from Phase 1 clinical trial Lispro-PH20 co-administration accelerates insulin absorption to yield more physiologic meal time insulin profile PH20 accelerates absorption of lispro Greater and earlier peak lispro-PH20 exposure and reduced late exposure Change in PK profile leads to significant reduction in post meal hyperglycemia and is meaningful relative to treatment targets 22 |

| Better glycemic control Faster, higher insulin concentrations and peak exposure (i.e., fast in) results in early glucose lowering effects and reduced post-meal hyperglycemia, which may lead to better A1C control Fast in, fast out profile is ideal for insulin pump applications Insulin-PH20 Could Have Best-in-Class Product Profile vs. Standard of Care Analogs Less hypoglycemia Reduced late post-meal exposure (i.e., fast out) may result in fewer hypoglycemic events Lowering insulin dose with PH20 to match glycemic control of analogs may further reduce hypoglycemia Less weight gain Fewer hypoglycemic events and lower insulin doses could result in less self-medicating snacking, especially in Type 2 patients 23 |

| Additional capital efficient clinical studies to further characterize Insulin- PH20 value proposition prior to starting pivotal studies in 2010-2011 Intra-subject PK/GD variability study underway to clarify dose reproducibility Phase 2 repeat dose (tid) treatment studies in T1DM patients comparing regular insulin-PH20 to lispro (initiated 2Q09), then analog-PH20 to lispro (2010) in a 3 month x 3 month crossover design Phase 2 standard meal study in T2DM patients (3Q09) Pivotal Phase 3 trials in T1DM and T2DM patients comparing Insulin- PH20 product candidate to analog insulin following repeat dose study and program review with regulatory authorities Halo will be prepared to partner or proceed to Phase 3 with most attractive product candidate (analog-PH20 or regular insulin-PH20) Efficient Development Strategy Planned to Maximize Insulin-PH20 Program Value 24 |
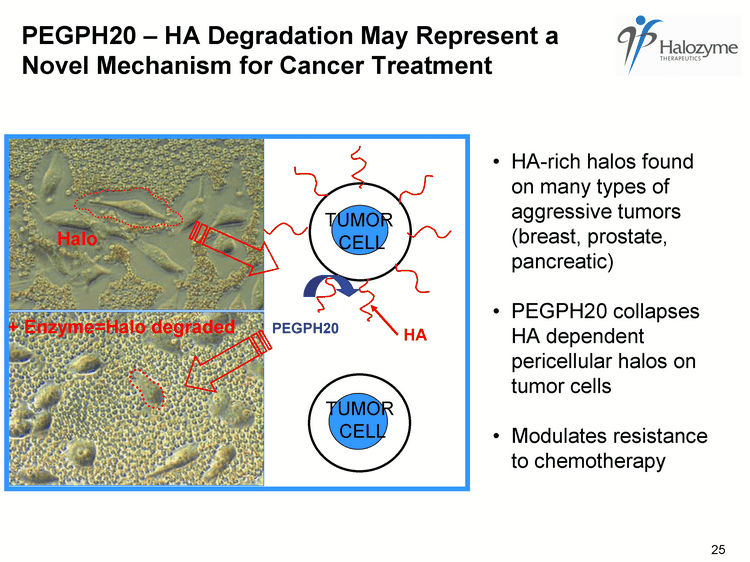
| HA-rich halos found on many types of aggressive tumors (breast, prostate, pancreatic) PEGPH20 collapses HA dependent pericellular halos on tumor cells Modulates resistance to chemotherapy HA TUMOR CELL + Enzyme=Halo degraded Halo PEGPH20 TUMOR CELL 25 PEGPH20 - HA Degradation May Represent a Novel Mechanism for Cancer Treatment |
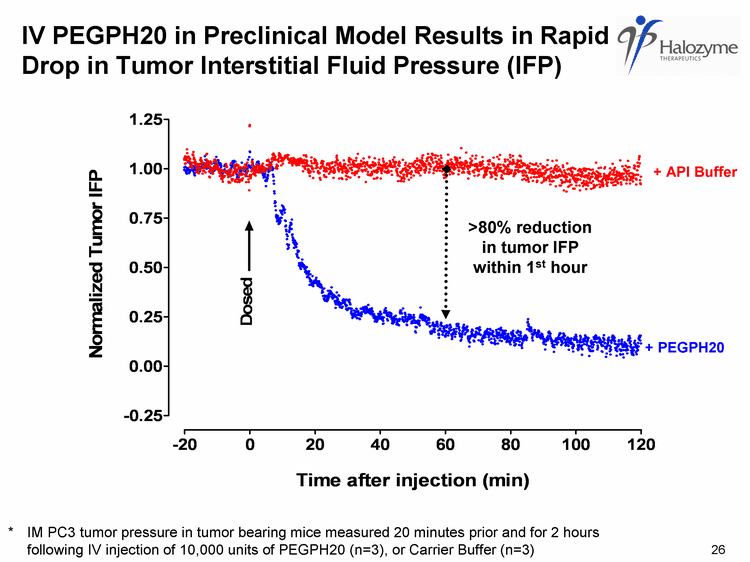
| IV PEGPH20 in Preclinical Model Results in Rapid Drop in Tumor Interstitial Fluid Pressure (IFP) * IM PC3 tumor pressure in tumor bearing mice measured 20 minutes prior and for 2 hours following IV injection of 10,000 units of PEGPH20 (n=3), or Carrier Buffer (n=3) + API Buffer + PEGPH20 >80% reduction in tumor IFP within 1st hour 26 |
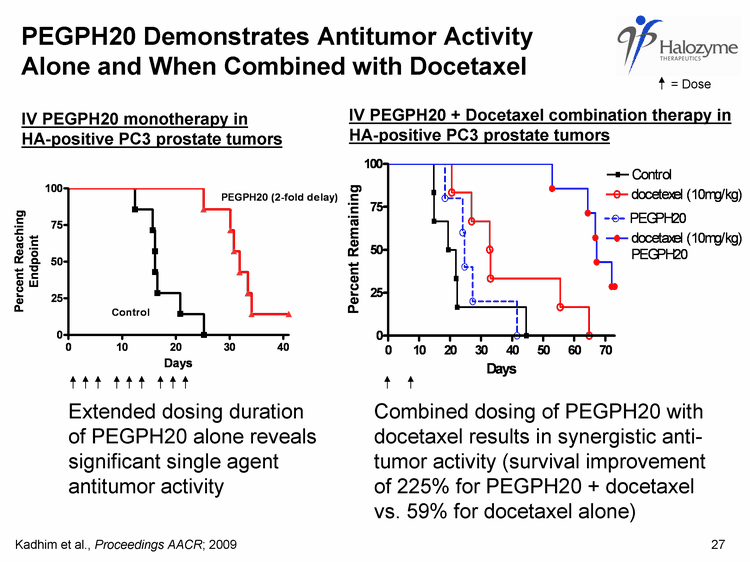
| PEGPH20 Demonstrates Antitumor Activity Alone and When Combined with Docetaxel IV PEGPH20 monotherapy in HA-positive PC3 prostate tumors 27 Combined dosing of PEGPH20 with docetaxel results in synergistic anti- tumor activity (survival improvement of 225% for PEGPH20 + docetaxel vs. 59% for docetaxel alone) IV PEGPH20 + Docetaxel combination therapy in HA-positive PC3 prostate tumors Extended dosing duration of PEGPH20 alone reveals significant single agent antitumor activity Kadhim et al., Proceedings AACR; 2009 = Dose |

| PEGPH20 Program - Phase 1 Clinical Trial Multicenter, open-label, dose evaluation trial of IV administered PEGPH20 in advanced cancer patients began 1Q09 Safety and tolerability, PK, and PD data from oncology patients anticipated by mid-2010 Determine maximum tolerated dose & dose-limiting toxicities Observe patients for evidence of antitumor activity Study will guide PEGPH20 dose-selection for future clinical trials Phase 1/2 combination studies Phase 2 single agent studies 28 |

| Milestones for 2009 $5.5 million payment from Baxter for HYLENEX, 1Q09 Phase 1 underway for second Roche exclusive target, 1Q09 Initiated Insulin-PH20 intra-subject variability study, 1Q09 Initiated PEGPH20 Phase 1, 1Q09 Initiated Insulin-PH20 Phase 2 repeat dose (tid) study, 2Q09 Presented Insulin-PH20 Phase 2 interim data at ADA, 2Q09 $4.25 million payment from Roche for fifth exclusive target, 2Q09 Present Insulin-PH20 Phase 2 data at EASD, 2H09 Complete patient enrollment of Phase 3 GAMMAGARD-PH20 by Baxter, 4Q09 HYLENEX launch in pediatric hydration by Baxter, 4Q09 Release Insulin-PH20 variability data, 4Q09 Initiation of additional Roche clinical trial(s) plus milestone(s) possible 29 |

| HALO's Unique Investment Thesis Existing and potential drug delivery partnerships (Enhanze Technology, HYLENEX) may provide non-dilutive cash to fund proprietary pipeline opportunities and drive near-term value Portfolio of enzymes targeting the Matrix (PH20, PEGPH20, HTI- 501), with broad potential and patient benefits across variety of therapeutic uses, drives long-term value Scaleable PH20 platform for developing other proprietary products with potentially faster time to market and lower development risk; Matrix expertise for developing other NMEs 30 |