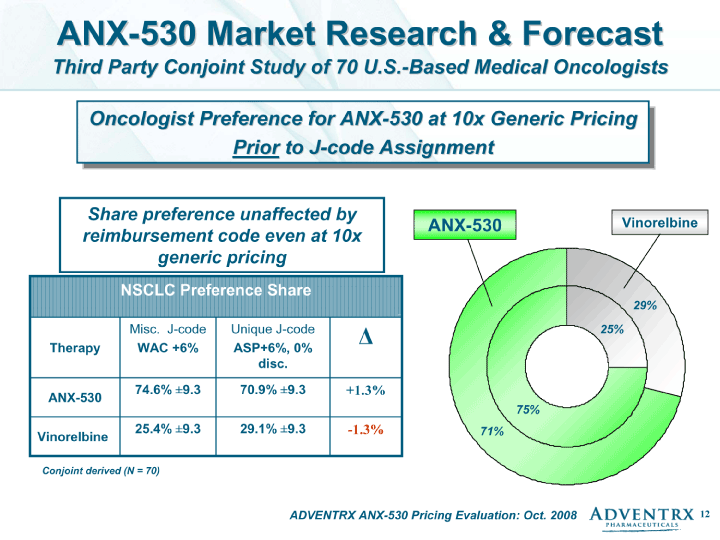
| ADVENTRX ANX-530 Pricing Evaluation: Oct. 2008 Vinorelbine Conjoint derived (N = 70) ANX-530 Market Research & Forecast Third Party Conjoint Study of 70 U.S.-Based Medical Oncologists NSCLC Preference Share NSCLC Preference Share NSCLC Preference Share NSCLC Preference Share Therapy Misc. J-code WAC +6% Unique J-code ASP+6%, 0% disc. ^ ANX-530 74.6% +-9.3 70.9% +-9.3 +1.3% Vinorelbine 25.4% +-9.3 29.1% +-9.3 -1.3% Share preference unaffected by reimbursement code even at 10x generic pricing Oncologist Preference for ANX-530 at 10x Generic Pricing Prior to J-code Assignment ANX-530 75% 71% 29% 25% |
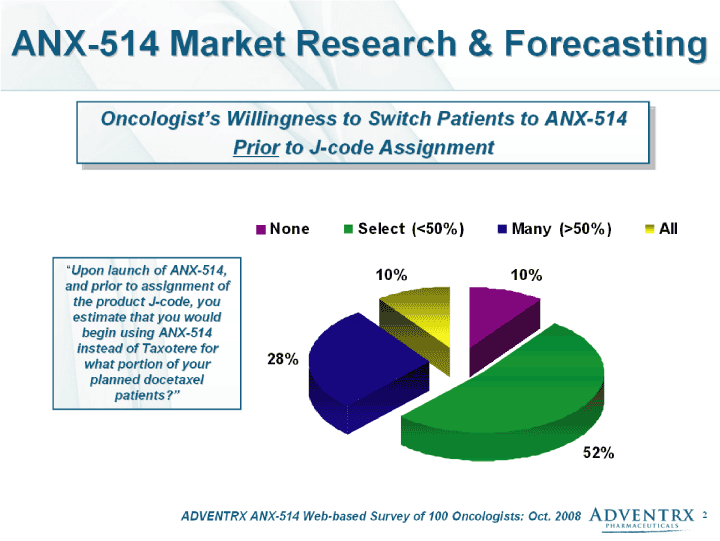
| ADVENTRX ANX-514 Web-based Survey of 100 Oncologists: Oct. 2008 ANX-514 Market Research & Forecasting Oncologist's Willingness to Switch Patients to ANX-514 Prior to J-code Assignment "Upon launch of ANX-514, and prior to assignment of the product J-code, you estimate that you would begin using ANX-514 instead of Taxotere for what portion of your planned docetaxel patients?" |
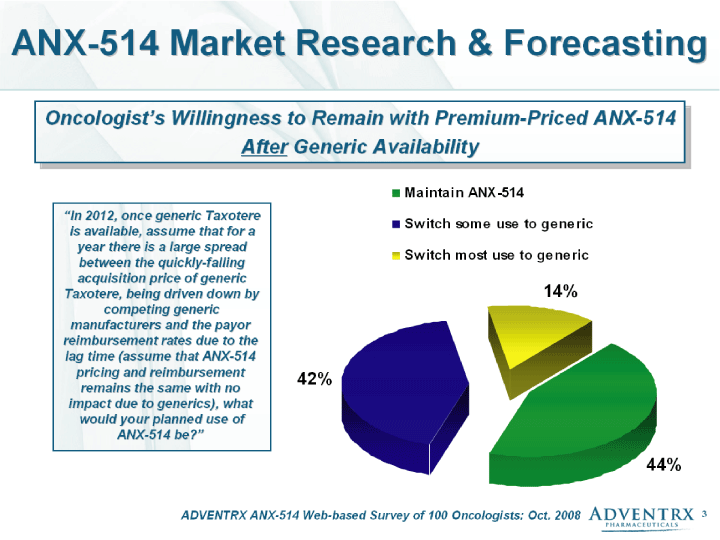
| ADVENTRX ANX-514 Web-based Survey of 100 Oncologists: Oct. 2008 ANX-514 Market Research & Forecasting Oncologist's Willingness to Remain with Premium-Priced ANX-514 After Generic Availability "In 2012, once generic Taxotere is available, assume that for a year there is a large spread between the quickly-falling acquisition price of generic Taxotere, being driven down by competing generic manufacturers and the payor reimbursement rates due to the lag time (assume that ANX-514 pricing and reimbursement remains the same with no impact due to generics), what would your planned use of ANX-514 be?" |

| Q1 2009 Complete enrollment in BE study of ANX-514 (docetaxel emulsion) Q2 2009 Submit New Drug Application for ANX-530 (vinorelbine emulsion) Announce results from BE study of ANX-514 (docetaxel emulsion) Q3 2009 Submit New Drug Application for ANX-514 (docetaxel emulsion) Near Term Regulatory Milestones |
| Mark N. K. Bagnall, Executive Vice President & Chief Financial Officer Metabolex Inc.; Metrika, Inc.; Progenitor, Inc.; Somatix Therapy Corp.; Hana Biologics, Inc. Brian M. Culley, M.S., M.B.A., Chief Business Officer & SVP Business Development Immusol; UCSD Technology Transfer and Intellectual Property Dept.; Neurocrine Biosciences Mark Erwin, Senior Vice President, Operations Centric Health Finance, LLC ;Ligand Pharmaceuticals; IDEC Pharmaceuticals; Eli Lilly & Co. Michele L. Yelmene, Vice President, Regulatory Affairs & Quality Assurance Perlan Therapeutics, Genzyme Corp., Mallinckrodt Jose R. Hechavarria, Vice President, Manufacturing HechTech Pharma Consult; Bristol Myers Squibb; DuPont Pharmaceuticals Patrick L. Keran, J.D., General Counsel Isis Pharmaceuticals; Heller Ehrman; Brobeck, Phleger & Harrison Brandi Roberts, C.P.A., M.B.A., Vice President, Finance Artes Medical; Strategene; Pfizer, Inc. ADVENTRX Team |