
Corporate Overview Developing New Therapies for Rare Respiratory Diseases January 2022 Exhibit 99.1

© Savara Inc. All Rights Reserved. Safe Harbor Statement Savara Inc. (“Savara” or the “Company”) cautions you that statements in this presentation that are not a description of historical fact are forward-looking statements which may be identified by the use of words such as “expect,” “intend,” “plan,” “anticipate,” “believe,” and “will,” among others. Such statements include, but are not limited to, statements regarding the nature, strategy and focus of Savara; the Savara investment thesis; the timing, design and other matters related to clinical trials of our product candidate; the safety, efficacy and projected development timeline of our product candidate; the potential health benefits of our product candidate; our anticipated corporate milestones; the potential market size, commercial opportunity, and competitive landscape for our product; and the sufficiency of our resources to fund the advancement of our development program and potential sources of additional capital. Savara may not actually achieve any of its plans or product development goals in a timely manner, if at all, or otherwise carry out its current intentions or meet the expectations or projections disclosed in its forward-looking statements, and you should not place undue reliance on these forward-looking statements. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. These forward-looking statements are based upon Savara's current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, the risks and uncertainties related to the impact of the COVID-19 pandemic on our business and operations; risks and uncertainties associated with the ability to project future cash utilization and reserves needed for contingent future liabilities and business operations; the availability of sufficient resources for our operations and to conduct or continue planned clinical development programs; the timing and ability of Savara to raise additional capital as needed to fund continued operations; the ability to successfully conduct clinical trials for our product candidate; the ability to successfully develop our product candidate; and the risks associated with the process of developing, obtaining regulatory approval for and commercializing drug candidates that are safe and effective for use as human therapeutics. The risks and uncertainties facing Savara are described more fully in Savara's filings with the Securities and Exchange Commission including our filings on Form 8-K, our Annual Report on Form 10-K for the fiscal year ended December 31, 2020, and our Quarterly Report on Form 10-Q for the quarter ended September 30, 2021. You are cautioned not to place undue reliance on our forward-looking statements, which speak only as of the date on which they were made. Savara undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as may be required by law. Third-party information included herein has been obtained from sources believed to be reliable, but the accuracy or completeness of such information is not guaranteed by, and should not be construed as a representation by, the Company. The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products.

Pursuing Transformative Therapies for Rare Respiratory Diseases © Savara Inc. All Rights Reserved. Focused on single Phase 3 program: molgramostim nebulizer solution (molgramostim) in autoimmune pulmonary alveolar proteinosis (aPAP) Recombinant form of human granulocyte-macrophage colony-stimulating factor (GM-CSF) Favorable efficacy and safety data generated from Phase 2 proof of concept (POC) IMPALA trial Pivotal Phase 3 trial underway – builds on key learnings from IMPALA Seasoned management team Deep experience in the development and commercialization of rare respiratory therapeutics and pulmonary medicines Capitalized through major clinical and regulatory milestones $171M in cash expected to fund company ~18-months beyond pivotal Phase 3 data read-out, beyond BLA filing, and through potential approval Quality investor base

© Savara Inc. All Rights Reserved. Executive Leadership Team Matthew Pauls Chair & Chief Executive Officer Badrul Chowdhury, M.D., Ph.D. Chief Medical Officer Dave Lowrance Chief Financial Officer Peter Clarke EVP, Global Technical Operations

Investment Thesis © Savara Inc. All Rights Reserved. The molgramostim in aPAP clinical program has a high probability of success Significant global commercial opportunity As an inhaled biologic, molgramostim has the potential for a long-term, durable revenue stream

Molgramostim Molgramostim for Autoimmune Pulmonary Alveolar Proteinosis (aPAP)
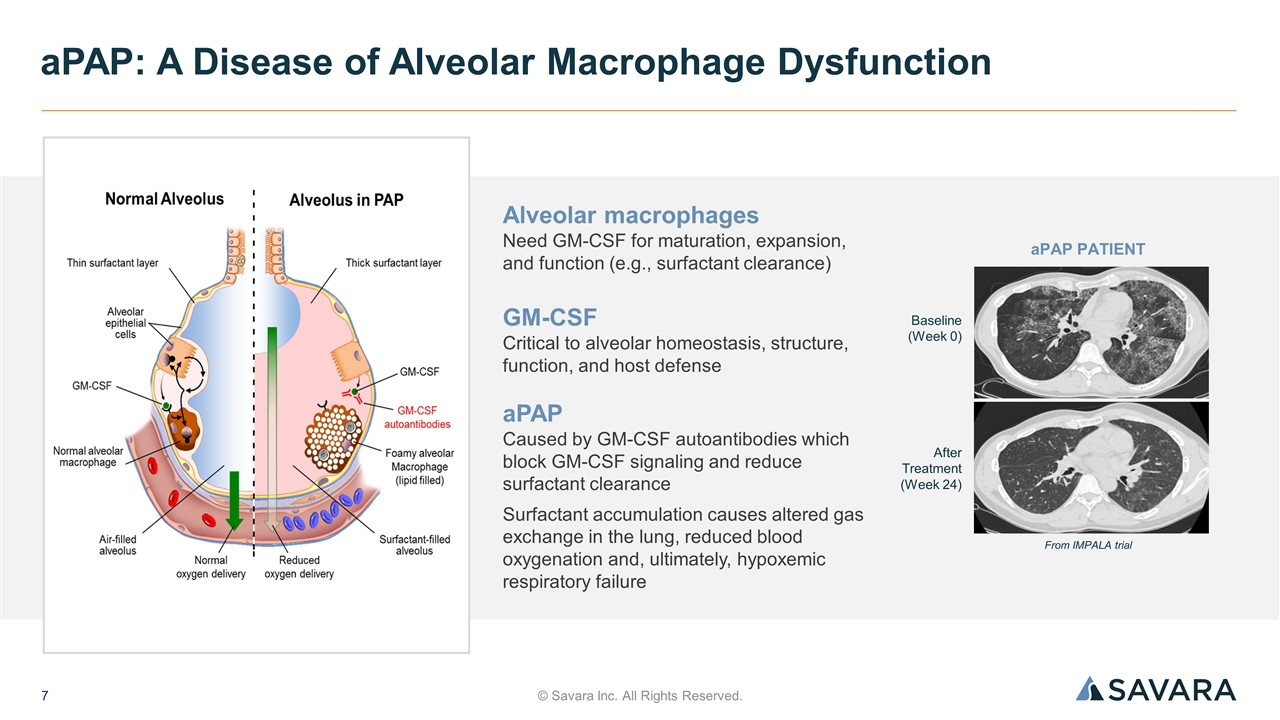
© Savara Inc. All Rights Reserved. aPAP: A Disease of Alveolar Macrophage Dysfunction Alveolar macrophages Need GM-CSF for maturation, expansion, and function (e.g., surfactant clearance) GM-CSF Critical to alveolar homeostasis, structure, function, and host defense aPAP Caused by GM-CSF autoantibodies which block GM-CSF signaling and reduce surfactant clearance Surfactant accumulation causes altered gas exchange in the lung, reduced blood oxygenation and, ultimately, hypoxemic respiratory failure Baseline (Week 0) After Treatment (Week 24) From IMPALA trial aPAP PATIENT
© Savara Inc. All Rights Reserved. aPAP is a Long-Term, Chronic Disease Shortness of Breath Gas exchange in the lungs is impaired and patients may experience shortness of breath At first it occurs upon exertion, but as disease progresses, it can occur even when a person is at rest Fatigue, Decreased Exercise Tolerance Fatigue and significantly reduced exercise capacity can dramatically impact the simplest of daily activities, e.g., getting winded walking up a flight of stairs Cough and Episodes of Fever Cough, sputum production, and episodes of fever, especially if secondary lung infection develops Fibrosis and Lung Transplant In the long-term, the disease can lead to serious complications, including fibrosis, often leading to the need for lung transplantation There are no approved drugs for the treatment of aPAP. Only option is whole lung lavage, an invasive procedure.
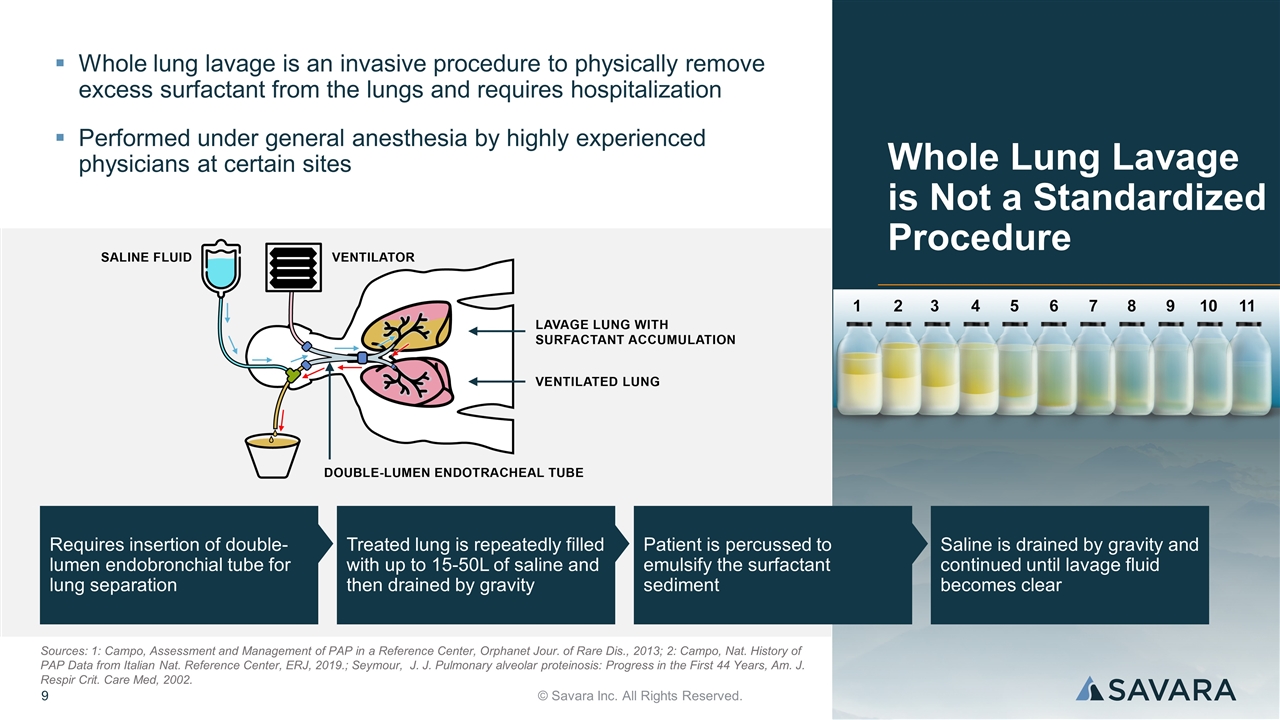
Requires insertion of double-lumen endobronchial tube for lung separation Treated lung is repeatedly filled with up to 15-50L of saline and then drained by gravity Patient is percussed to emulsify the surfactant sediment Saline is drained by gravity and continued until lavage fluid becomes clear Whole Lung Lavage is Not a Standardized Procedure © Savara Inc. All Rights Reserved. Whole lung lavage is an invasive procedure to physically remove excess surfactant from the lungs and requires hospitalization Performed under general anesthesia by highly experienced physicians at certain sites Sources: 1: Campo, Assessment and Management of PAP in a Reference Center, Orphanet Jour. of Rare Dis., 2013; 2: Campo, Nat. History of PAP Data from Italian Nat. Reference Center, ERJ, 2019.; Seymour, J. J. Pulmonary alveolar proteinosis: Progress in the First 44 Years, Am. J. Respir Crit. Care Med, 2002.

© Savara Inc. All Rights Reserved. Complications and Short-Comings of Whole Lung Lavage Rib fracture Hypoxia Pneumothorax (collapsed lung) Hydrothorax (fluid in pleural cavity) Superimposed infection Acute Respiratory Distress Syndrome (ARDS) Short Comings Treatment fails to address pathophysiology of disease Patients continue to experience symptomatic deterioration between procedures Rollercoaster ride of improvement and decline Procedure is not standardized and remains highly operator-dependent Potential Complications

Due to aPAP’s rarity and associated non-specific symptoms, patients are often misdiagnosed with more common pulmonary illnesses (e.g., recurrent pneumonia, chronic bronchitis, COPD, asthma) © Savara Inc. All Rights Reserved. Journey to Diagnosis Can Be Long and Misdiagnosis is Common Diagnostic tests typically conducted to rule-out other more common pulmonary diseases: Transbronchial biopsy and cytological analysis of bronchoalveolar lavage fluid Pulmonary function tests Imaging Secondary PAP testing

In Japan, a Simple Blood Antibody Test is Routinely Performed to Diagnose aPAP © Savara Inc. All Rights Reserved. Research advances have led to the development of a highly effective, simple blood test that can diagnose aPAP 100% sensitivity and 100% specificity Distinguishes aPAP from other respiratory diseases Not yet commercially available in the US and EU Once available in the US and EU, it could improve accuracy and reduce time to diagnosis
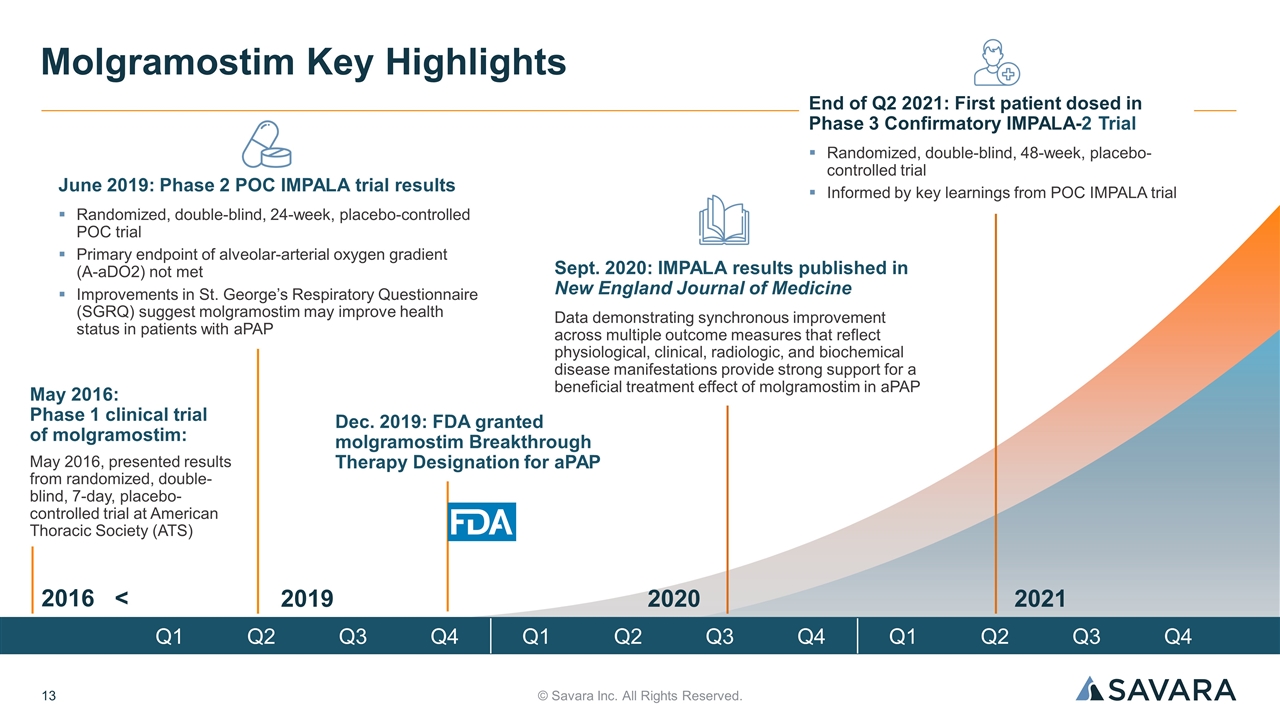
© Savara Inc. All Rights Reserved. Molgramostim Key Highlights June 2019: Phase 2 POC IMPALA trial results Randomized, double-blind, 24-week, placebo-controlled POC trial Primary endpoint of alveolar-arterial oxygen gradient (A-aDO2) not met Improvements in St. George’s Respiratory Questionnaire (SGRQ) suggest molgramostim may improve health status in patients with aPAP Dec. 2019: FDA granted molgramostim Breakthrough Therapy Designation for aPAP End of Q2 2021: First patient dosed in Phase 3 Confirmatory IMPALA-2 Trial Randomized, double-blind, 48-week, placebo-controlled trial Informed by key learnings from POC IMPALA trial Sept. 2020: IMPALA results published in New England Journal of Medicine Data demonstrating synchronous improvement across multiple outcome measures that reflect physiological, clinical, radiologic, and biochemical disease manifestations provide strong support for a beneficial treatment effect of molgramostim in aPAP May 2016: Phase 1 clinical trial of molgramostim: May 2016, presented results from randomized, double-blind, 7-day, placebo-controlled trial at American Thoracic Society (ATS) 2019 2020 2021 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2016 <
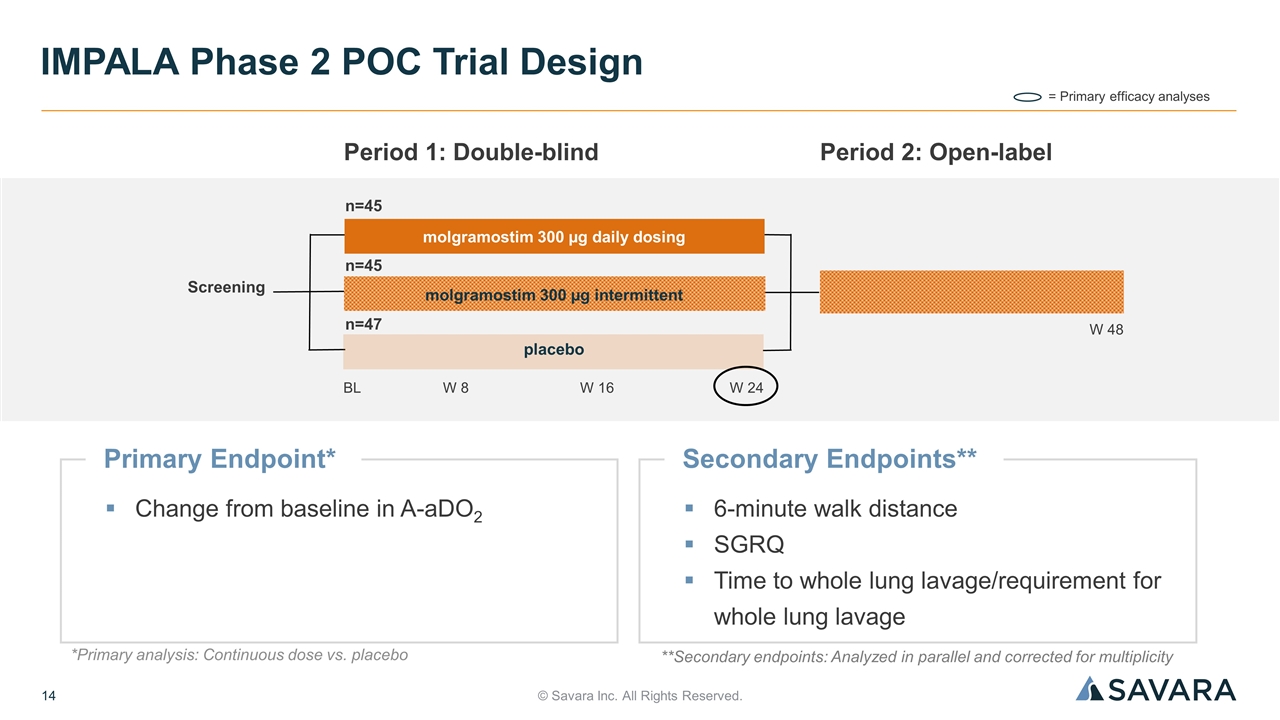
© Savara Inc. All Rights Reserved. IMPALA Phase 2 POC Trial Design **Secondary endpoints: Analyzed in parallel and corrected for multiplicity Primary Endpoint* Change from baseline in A-aDO2 = Primary efficacy analyses Screening BL n=45 Period 1: Double-blind W 8 W 24 W 16 Period 2: Open-label n=45 n=47 W 48 molgramostim 300 µg daily dosing molgramostim 300 µg intermittent placebo Secondary Endpoints** 6-minute walk distance SGRQ Time to whole lung lavage/requirement for whole lung lavage *Primary analysis: Continuous dose vs. placebo

IMPALA Trial Results Published in New England Journal of Medicine in September 2020 © Savara Inc. All Rights Reserved. Published online on 9/2/2020.
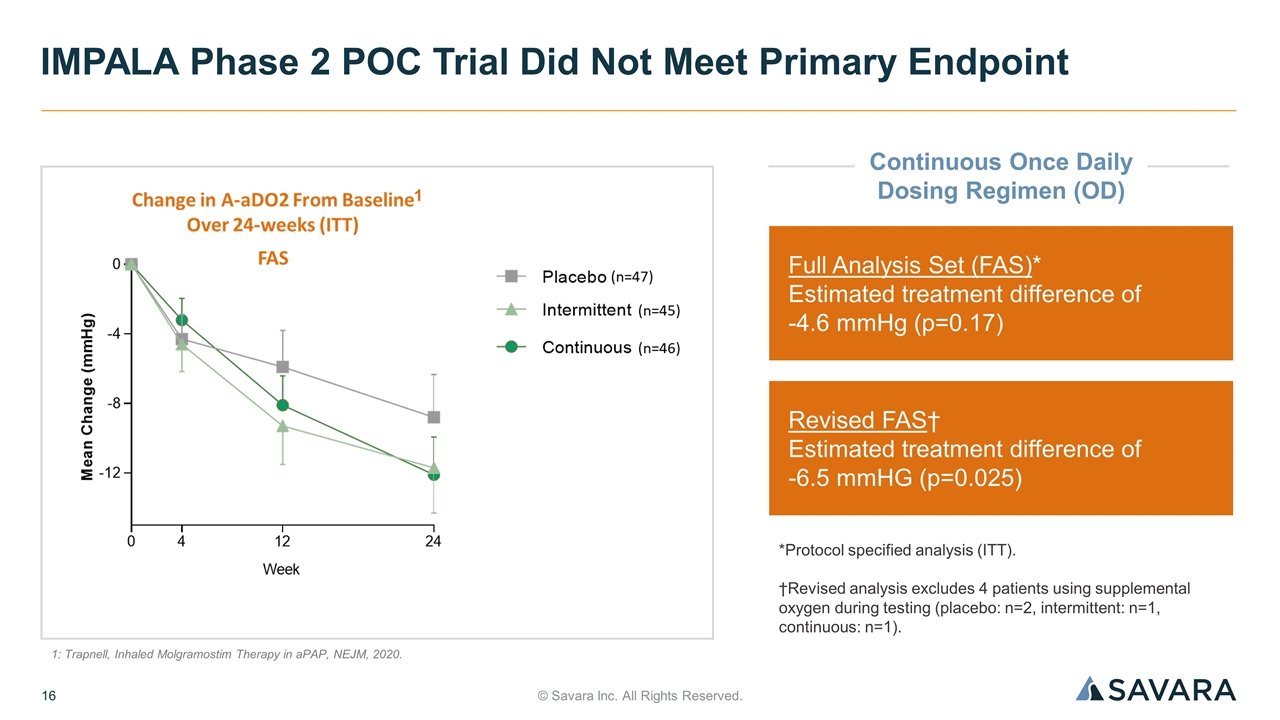
© Savara Inc. All Rights Reserved. IMPALA Phase 2 POC Trial Did Not Meet Primary Endpoint Full Analysis Set (FAS)* Estimated treatment difference of -4.6 mmHg (p=0.17) Revised FAS† Estimated treatment difference of -6.5 mmHG (p=0.025) *Protocol specified analysis (ITT). †Revised analysis excludes 4 patients using supplemental oxygen during testing (placebo: n=2, intermittent: n=1, continuous: n=1). 1: Trapnell, Inhaled Molgramostim Therapy in aPAP, NEJM, 2020. 1 Continuous Once Daily Dosing Regimen (OD)
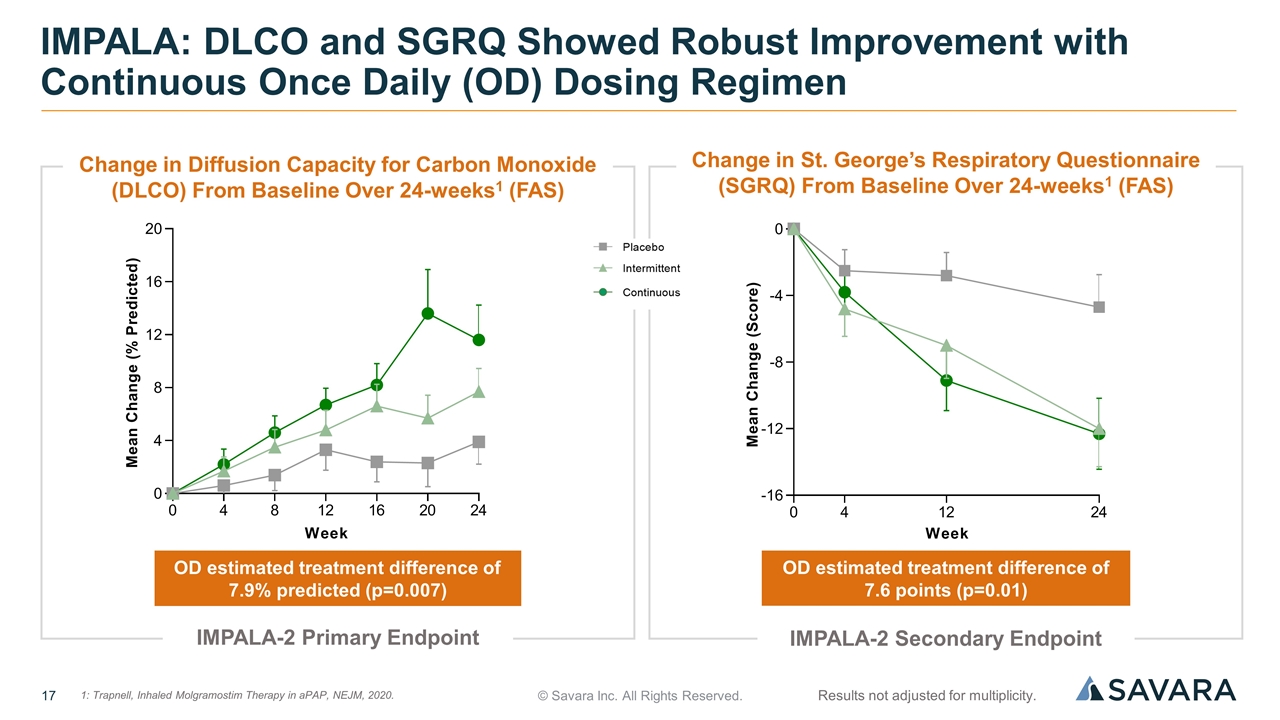
© Savara Inc. All Rights Reserved. IMPALA: DLCO and SGRQ Showed Robust Improvement with Continuous Once Daily (OD) Dosing Regimen OD estimated treatment difference of 7.9% predicted (p=0.007) OD estimated treatment difference of 7.6 points (p=0.01) Results not adjusted for multiplicity. IMPALA-2 Primary Endpoint IMPALA-2 Secondary Endpoint 1: Trapnell, Inhaled Molgramostim Therapy in aPAP, NEJM, 2020. 1 Change in Diffusion Capacity for Carbon Monoxide (DLCO) From Baseline Over 24-weeks1 (FAS) Change in St. George’s Respiratory Questionnaire (SGRQ) From Baseline Over 24-weeks1 (FAS)
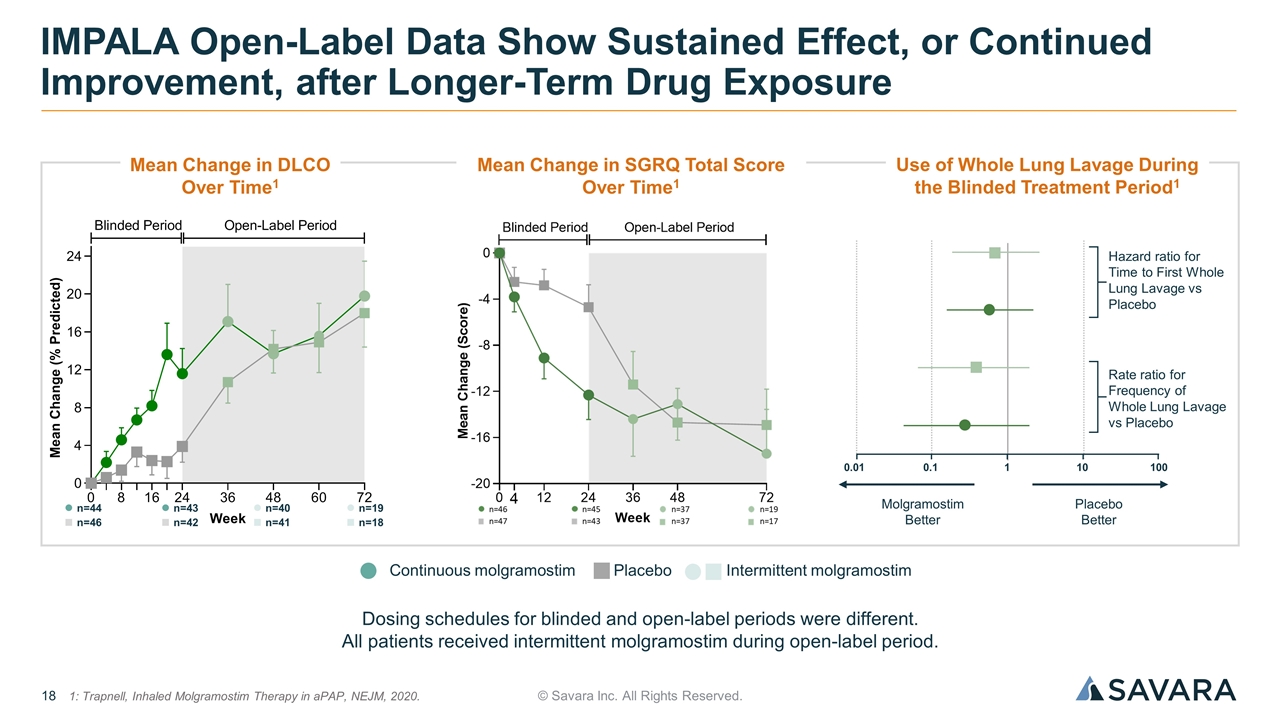
© Savara Inc. All Rights Reserved. IMPALA Open-Label Data Show Sustained Effect, or Continued Improvement, after Longer-Term Drug Exposure Placebo Continuous molgramostim Intermittent molgramostim Dosing schedules for blinded and open-label periods were different. All patients received intermittent molgramostim during open-label period. n=44 n=46 n=43 n=42 n=40 n=41 n=19 n=18 1: Trapnell, Inhaled Molgramostim Therapy in aPAP, NEJM, 2020. Mean Change in DLCO Over Time1 Mean Change in SGRQ Total Score Over Time1 Use of Whole Lung Lavage During the Blinded Treatment Period1 Hazard ratio for Time to First Whole Lung Lavage vs Placebo Rate ratio for Frequency of Whole Lung Lavage vs Placebo Placebo Better Molgramostim Better 100 10 1 0.1 0.01
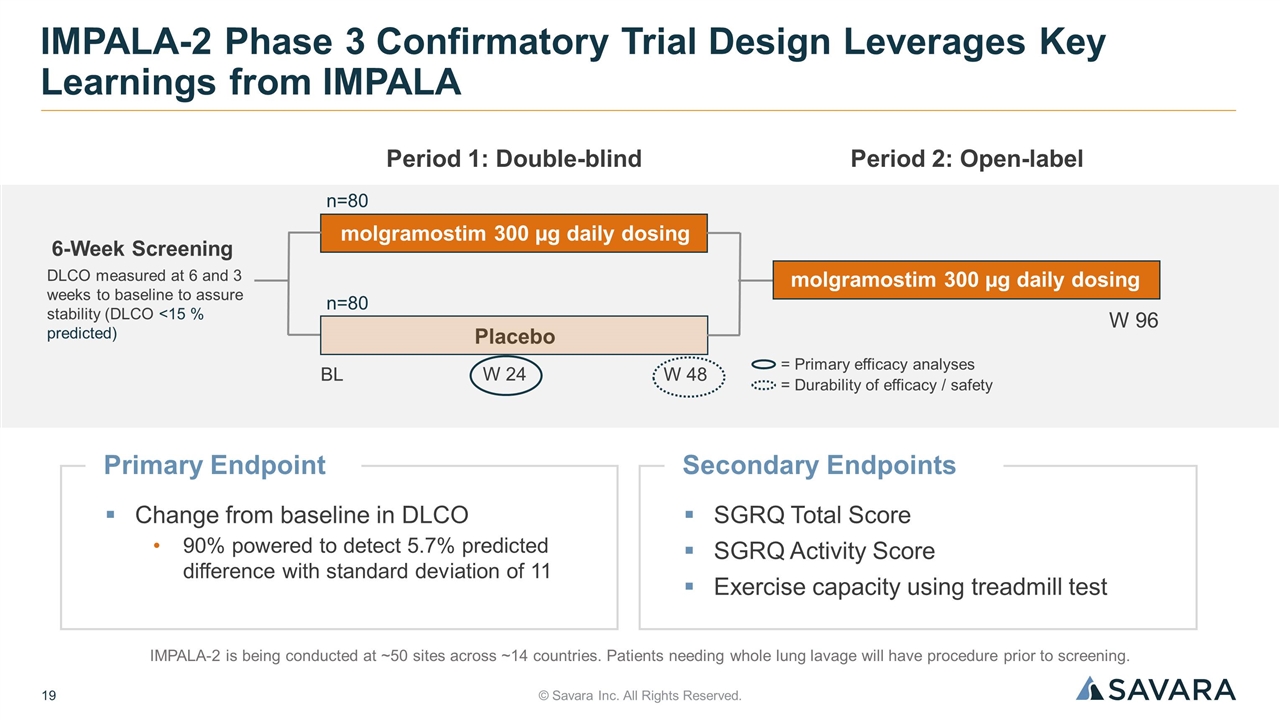
Primary Endpoint Change from baseline in DLCO 90% powered to detect 5.7% predicted difference with standard deviation of 11 © Savara Inc. All Rights Reserved. IMPALA-2 Phase 3 Confirmatory Trial Design Leverages Key Learnings from IMPALA IMPALA-2 is being conducted at ~50 sites across ~14 countries. Patients needing whole lung lavage will have procedure prior to screening. = Primary efficacy analyses = Durability of efficacy / safety 6-Week Screening BL molgramostim 300 µg daily dosing Period 1: Double-blind W 24 W 48 Period 2: Open-label W 96 Placebo molgramostim 300 µg daily dosing n=80 n=80 DLCO measured at 6 and 3 weeks to baseline to assure stability (DLCO <15 % predicted) Secondary Endpoints SGRQ Total Score SGRQ Activity Score Exercise capacity using treadmill test
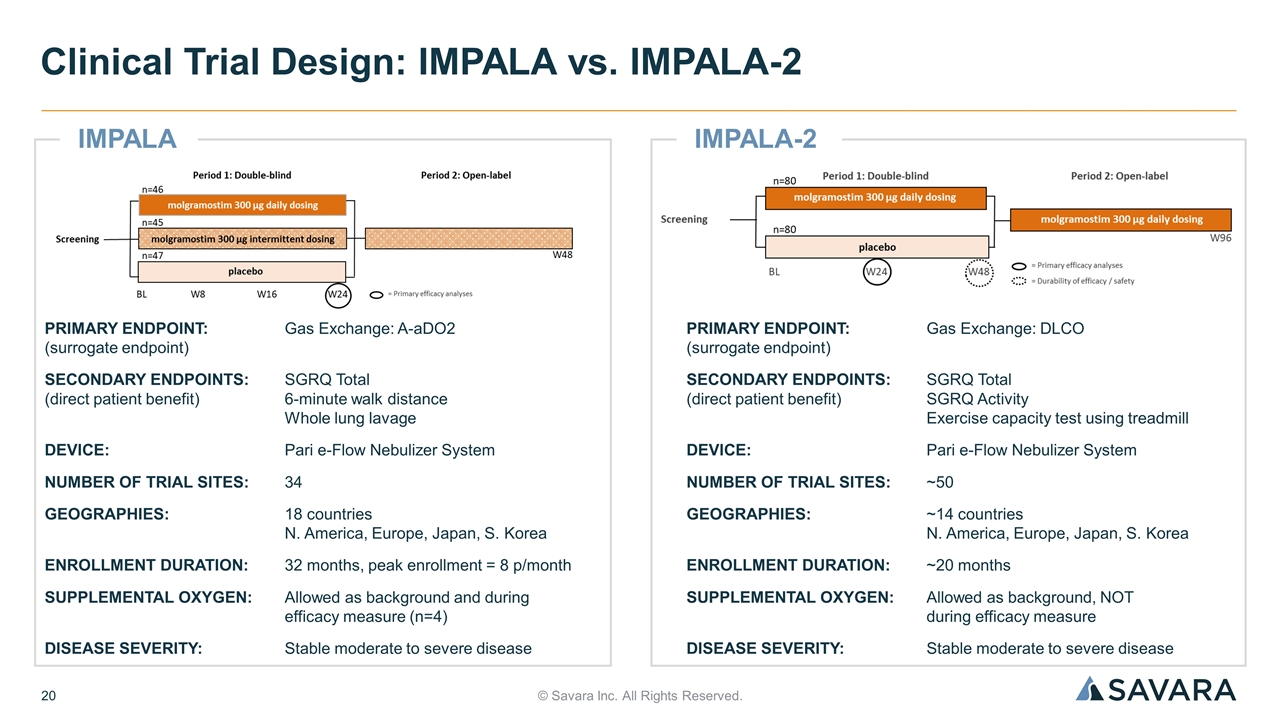
IMPALA-2 © Savara Inc. All Rights Reserved. Clinical Trial Design: IMPALA vs. IMPALA-2 PRIMARY ENDPOINT: Gas Exchange: A-aDO2 (surrogate endpoint) SECONDARY ENDPOINTS:SGRQ Total (direct patient benefit) 6-minute walk distance Whole lung lavage DEVICE:Pari e-Flow Nebulizer System NUMBER OF TRIAL SITES:34 GEOGRAPHIES:18 countries N. America, Europe, Japan, S. Korea ENROLLMENT DURATION:32 months, peak enrollment = 8 p/month SUPPLEMENTAL OXYGEN:Allowed as background and during efficacy measure (n=4) DISEASE SEVERITY:Stable moderate to severe disease PRIMARY ENDPOINT: Gas Exchange: DLCO (surrogate endpoint) SECONDARY ENDPOINTS: SGRQ Total (direct patient benefit) SGRQ Activity Exercise capacity test using treadmill DEVICE: Pari e-Flow Nebulizer System NUMBER OF TRIAL SITES: ~50 GEOGRAPHIES:~14 countries N. America, Europe, Japan, S. Korea ENROLLMENT DURATION: ~20 months SUPPLEMENTAL OXYGEN: Allowed as background, NOT during efficacy measure DISEASE SEVERITY: Stable moderate to severe disease IMPALA
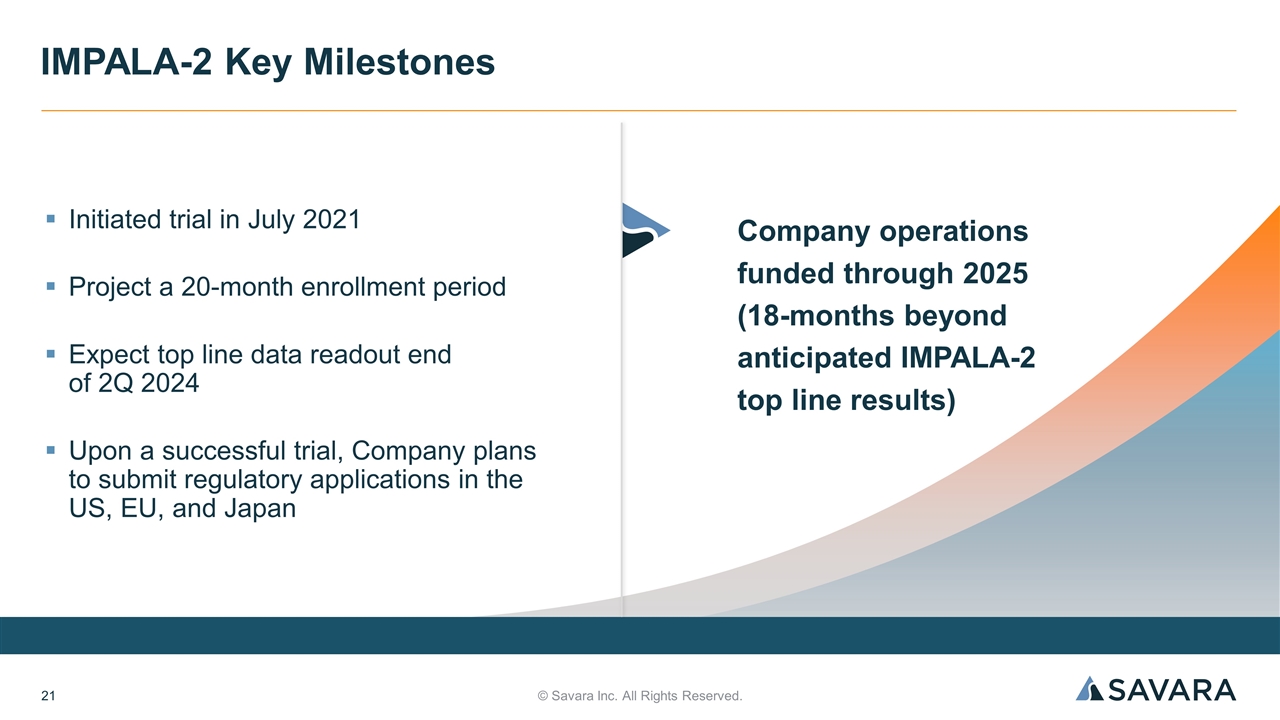
Initiated trial in July 2021 Project a 20-month enrollment period Expect top line data readout end of 2Q 2024 Upon a successful trial, Company plans to submit regulatory applications in the US, EU, and Japan © Savara Inc. All Rights Reserved. IMPALA-2 Key Milestones Company operations funded through 2025 (18-months beyond anticipated IMPALA-2 top line results)

Upon Biologics License Application (BLA) approval FDA would grant 12 years marketing exclusivity BIOLOGIC EXCLUSIVITY Trial design endorsed by regulatory authorities in the US, Canada, Japan, South Korea, and the countries in Europe where the trial is being conducted IMPALA-2 Molgramostim Regulatory Landscape © Savara Inc. All Rights Reserved. Orphan Drug Designation, Europe (eligible for 10 years exclusivity) Orphan Drug Designation, US (eligible for 7 years exclusivity) Fast Track Designation Breakthrough Therapy Designation MOLGRAMOSTIM IN aPAP REGULATORY DESIGNATIONS

Commercial Outlook
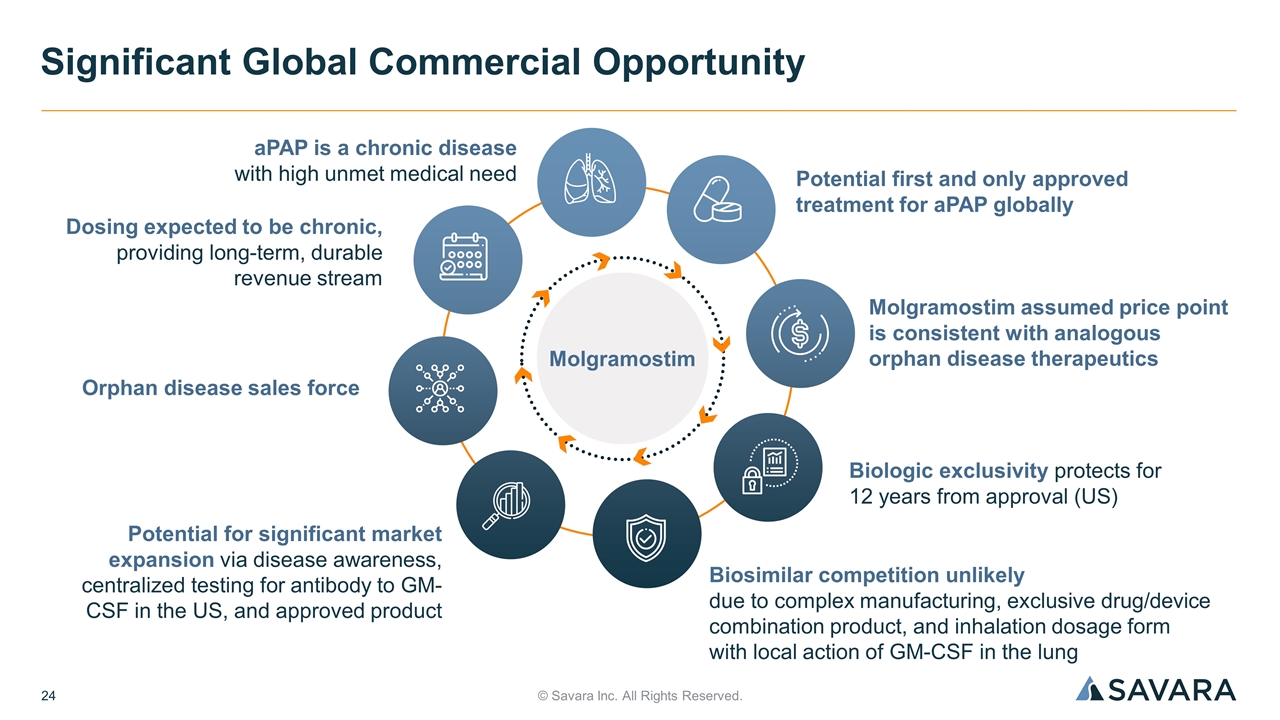
© Savara Inc. All Rights Reserved. Significant Global Commercial Opportunity aPAP is a chronic disease with high unmet medical need Potential first and only approved treatment for aPAP globally Dosing expected to be chronic, providing long-term, durable revenue stream Molgramostim assumed price point is consistent with analogous orphan disease therapeutics Potential for significant market expansion via disease awareness, centralized testing for antibody to GM-CSF in the US, and approved product Biologic exclusivity protects for 12 years from approval (US) Orphan disease sales force Biosimilar competition unlikely due to complex manufacturing, exclusive drug/device combination product, and inhalation dosage form with local action of GM-CSF in the lung Molgramostim
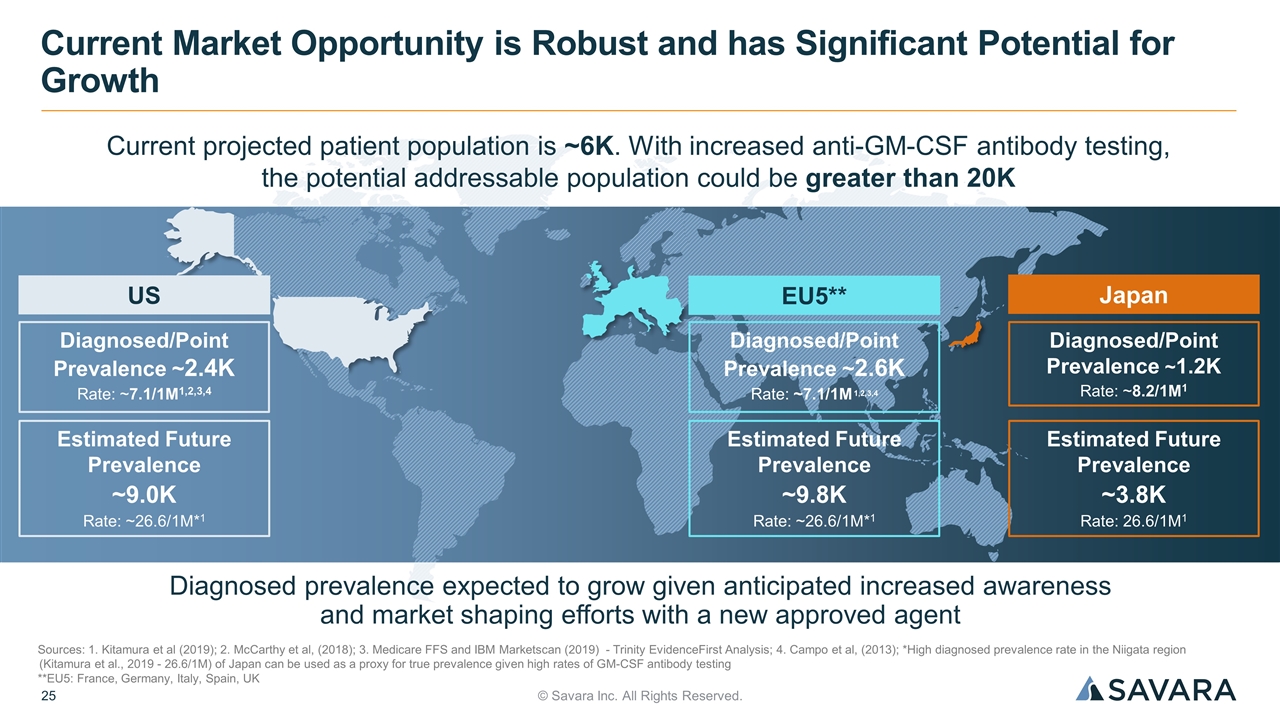
© Savara Inc. All Rights Reserved. Current Market Opportunity is Robust and has Significant Potential for Growth US Diagnosed/Point Prevalence ~2.4K Rate: ~7.1/1M1,2,3,4 Estimated Future Prevalence ~9.0K Rate: ~26.6/1M*1 EU5** Japan Diagnosed prevalence expected to grow given anticipated increased awareness and market shaping efforts with a new approved agent Diagnosed/Point Prevalence ~2.6K Rate: ~7.1/1M 1,2,3,4 Estimated Future Prevalence ~9.8K Rate: ~26.6/1M*1 Diagnosed/Point Prevalence ~1.2K Rate: ~8.2/1M1 Estimated Future Prevalence ~3.8K Rate: 26.6/1M1 Sources: 1. Kitamura et al (2019); 2. McCarthy et al, (2018); 3. Medicare FFS and IBM Marketscan (2019) - Trinity EvidenceFirst Analysis; 4. Campo et al, (2013); *High diagnosed prevalence rate in the Niigata region (Kitamura et al., 2019 - 26.6/1M) of Japan can be used as a proxy for true prevalence given high rates of GM-CSF antibody testing **EU5: France, Germany, Italy, Spain, UK Current projected patient population is ~6K. With increased anti-GM-CSF antibody testing, the potential addressable population could be greater than 20K

Analog: Pulmozyme® (dornase alpha) © Savara Inc. All Rights Reserved. Prototype inhaled biologic Approved by the FDA in 1993 No biosimilar available Pulmozyme® Pulmozyme is a registered trademark of Genentech

Financials

Financial Highlights © Savara Inc. All Rights Reserved. Well capitalized $171M in cash (as of 9/30/21) Cash runway extends 18-months beyond anticipated IMPALA-2 top line results Strong investor support with coverage from 6 equity research analysts Evercore ISI Liisa Bayko, MSC, MBA H.C. Wainwright Andrew Fein Jefferies Suji Jeong, PhD Ladenburg Thalmann & Co. Michael Higgins Oppenheimer Francois Brisebois Piper Sandler Yasmeen Rahimi, PhD ANALYST COVERAGE

Investment Thesis © Savara Inc. All Rights Reserved. The molgramostim in aPAP clinical program has a high probability of success Significant global commercial opportunity As an inhaled biologic, molgramostim has the potential for a long-term, durable revenue stream

Thank You