Exhibit 99.1

Cor p orate Overview N A SD A Q: ABVC 2023

This presentation contains summary information about ABVC BioPharma, Inc . (“ABVC”) as of the date hereof . The information in this presentation is of general background and contains an overview and summary of certain data selected by the management of ABVC . It does not purport to be complete . This presentation is not a prospectus, disclosure document or offering document under the law of any jurisdiction . It is for informational purposes only . This presentation is not investment or financial product advice (nor tax, accounting or legal advice) and is not intended to be used for the basis of making an investment decision . A recipient must make their own independent investigations, consideration and evaluation of ABVC and the offer and ABVC recommends that investors should obtain their own professional advice before making any investment decisions in the company . This investor presentation shall also not constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any states or jurisdictions in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction . No registered offering of securities shall be made except by means of a prospectus meeting the requirements of section 10 of the Securities Act of 1933 , as amended . This document has been prepared based on information available at the time of presentation . No representation or warranty, express or implied, is made as to the fairness, accuracy or completeness of the information, opinions and conclusions contained in this presentation or any omission from this presentation or of any other written or oral information or opinions provided now or in the future to any person . While reasonable care has been taken to ensure that facts stated in this presentation are accurate and/or that the opinions expressed are fair and reasonable, no reliance can be placed for any purpose whatsoever on the information contained in this document or its completeness . To the maximum extent permitted by law, neither ABVC nor their respective officers, directors, employees, advisors and agents, nor any other person, accepts any liability as to or in relation to the accuracy or completeness of the information, statements, opinions or matters (express or implied) arising out of, contained in or derived from this presentation or any omission from this presentation or of any other written or oral information or opinions provided now or in the future to any person . Some of the statements appearing in this presentation are in the nature of forward looking statements . You should be aware that such statements are predictions based on assumptions, and are subject to inherent risks and uncertainties . Those risks and uncertainties include factors and risks specific to the industry in which ABVC operates as well as general economic conditions, prevailing exchange rates and interest rates and conditions in the financial markets and other factors that are in some cases beyond ABVC's control . As a result, any or all of the ABVC’s forward - looking statements in this presentation may turn out to be inaccurate . Except as required by law, we are under no duty to update or revise any of the forward - looking statements, whether as a result of new information, future events or otherwise, after the date of this presentation . These forward - looking statements speak only as of the date of this presentation, and we assume no obligation to update or revise these forward - looking statements for any reason . 2 F o rward L o oking Statemen t s
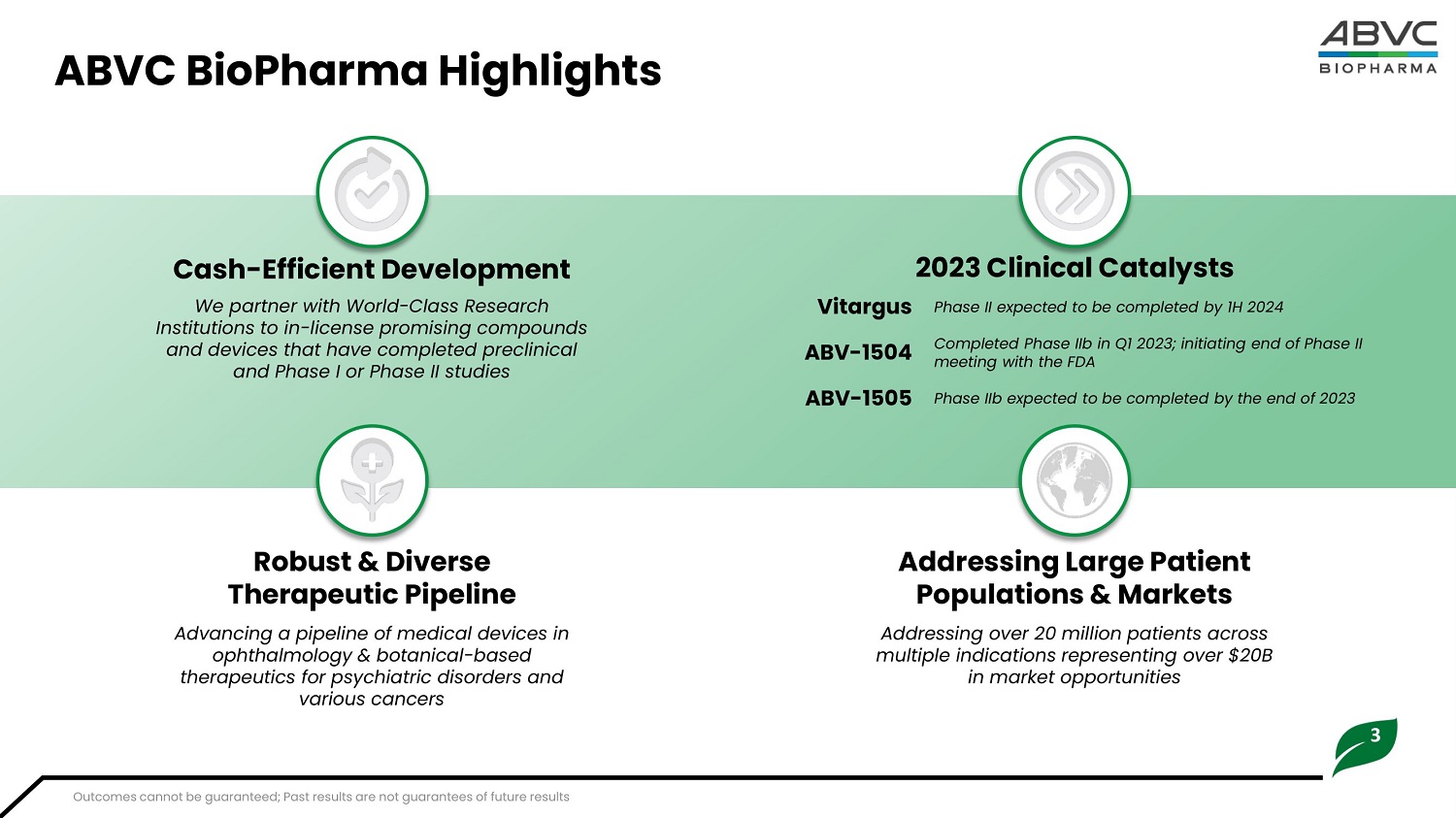
3 ABVC BioPharma Highlights 2 0 23 C l in i cal Catalysts Cash - Effici e nt Dev e lopm en t We partner with World - Class Research Institutions to in - license promising compounds and devices that have completed preclinical and Phase I or Phase II studies Phase II expected to be completed by 1H 2024 Vitargus Completed Phase IIb in Q1 2023; initiating end of Phase II meeting with the FDA ABV - 1504 Phase IIb expected to be completed by the end of 2023 ABV - 1505 Robust & Div erse Therapeu tic Pipel i ne Advancing a pipeline of medical devices in ophthalmology & botanical - based therapeutics for psychiatric disorders and various cancers Addr e ssing Large Pati e nt P o pulations & Mar k ets Addressing over 20 million patients across multiple indications representing over $20B in market opportunities Outcomes cannot be guaranteed; Past results are not guarantees of future results
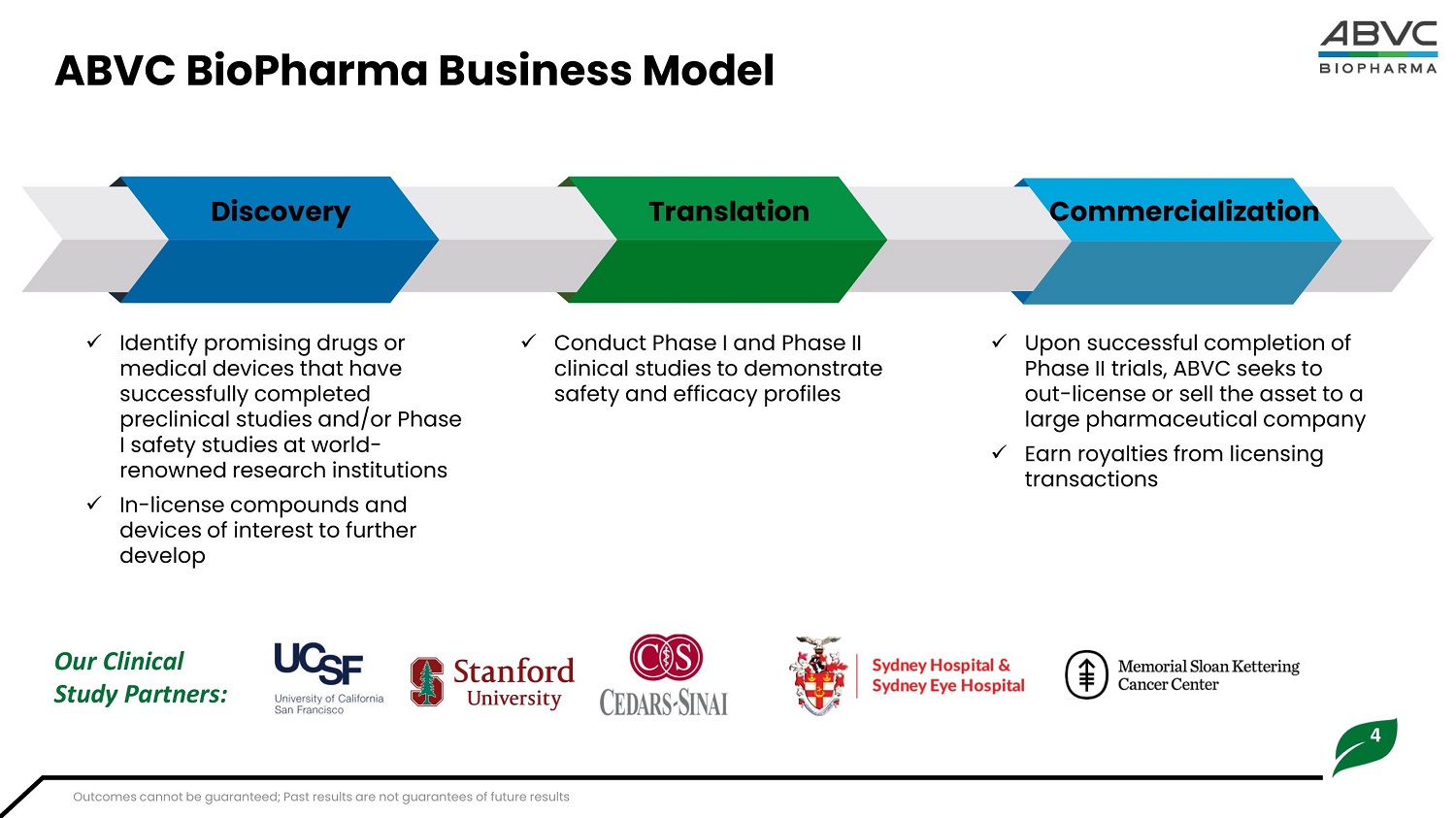
4 ABVC BioPharma Business Model Disc o ve r y T ran s lati o n Commercialization x Identify promising drugs or medical devices that have successfully completed pr e cli nical studi e s and/o r Ph ase I s a f e t y studies at world - renowned research institutions x In - license compounds and de v ic e s of in t e r e st t o f ur t he r develop x Conduct P hase I and Ph ase II clinical studies to demonstrate safety and efficacy profiles x Upon successful completion of Ph ase II t r ials, ABVC se e ks t o out - license or sell the asset to a lar ge phar m aceu t ical co m pany x Earn royalties from licensing transactions Our Clinical Study Partners: Outcomes cannot be guaranteed; Past results are not guarantees of future results

5 Leadership Team Howard Doong, MD, PhD Chief Executive Officer Le ed s Cho w Chief Financial O ffi cer Ch i - H s in Richa r d Kin g , P hD Chief Scientific O ffi cer T. S . J i ang , PhD Chief Strategy Officer Scientific Ad v i s o r y B o ard Managemen t Yih - S hiou Hw a n g , MD, PhD S u s a nna Cunnin g h a m - Rundles, PhD Maurizio Fava, PhD Keith McBurnett, PhD Thomas Laughren, PhD
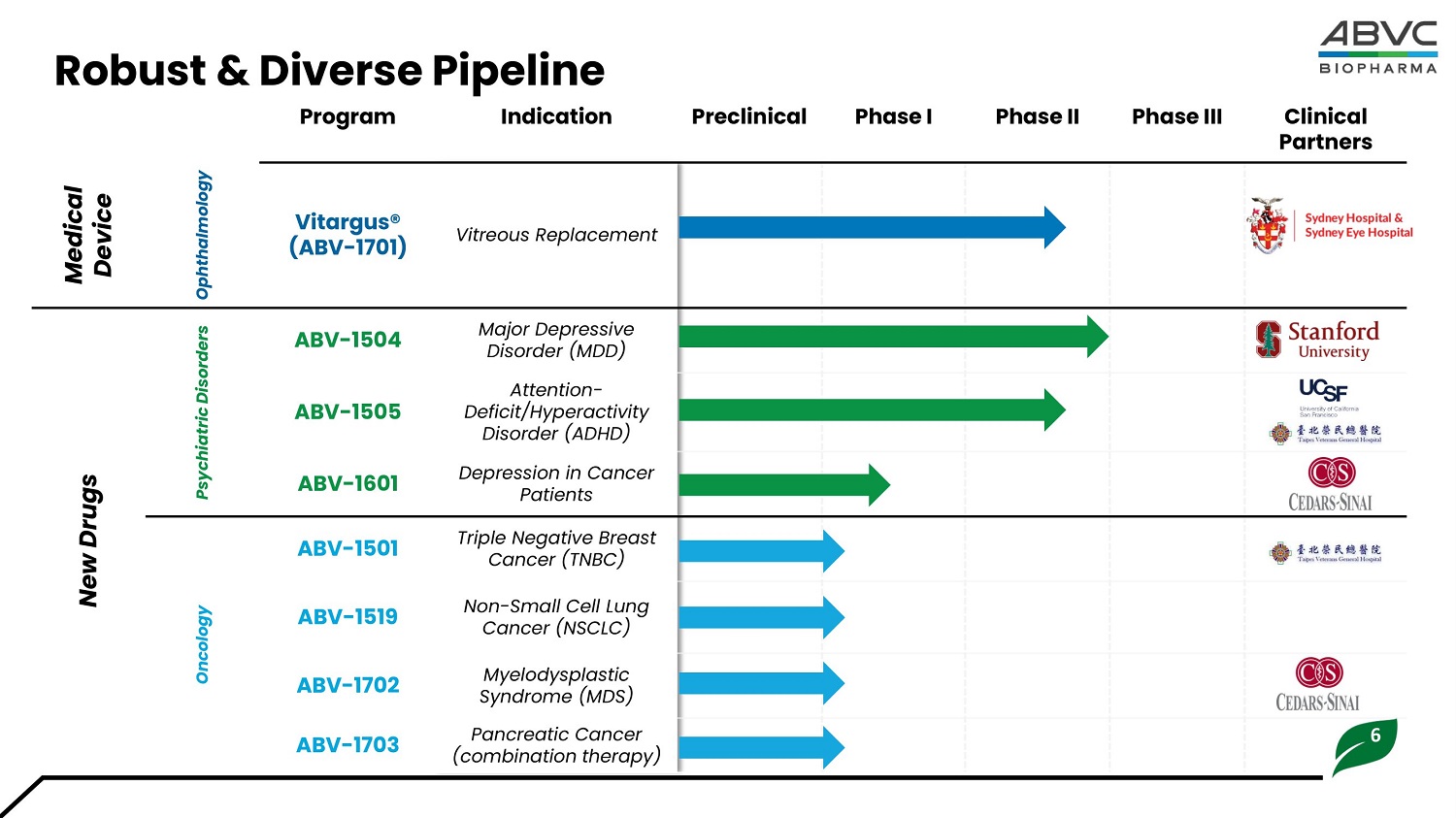
Clinical P a rtners P h ase III Pha s e II Pha s e I Preclinical Indication Program Vitreous Replacement Vitargus® (ABV - 1701) Ophthalmology Med i c al Devi ce Major Depressive Disorder (MDD) ABV - 1504 Psyc h iatric Disor d ers New Drugs Attention - Defici t /H y pe r ac t ivi t y Disorder (ADHD) ABV - 1505 Depression in Cancer Patients ABV - 1601 Triple Negative Breast Cancer (TNBC) ABV - 1501 Non - Small Cell Lung Cancer (NSCLC) ABV - 1519 Oncology Myelodysplastic Syndrome (MDS) ABV - 1702 6 Pancreatic Cancer (combination therapy) ABV - 1703 Ro b ust & D ive r se P ipeline

7 Vitargus® for Retinal Detachment & Vitreous Hemorrhage Vi trect o my Surgery Detached Retina • Ma c ul a r Hole • Ma c ul a r Puc k er Vitreous Hemorrhage • Diabetic Retinopathy • Retinal Vein Occlusion • Vitreous Body Injury Vitargus® is a Vitreous substitute that could potentially be used in retinal detachment and vitreous hemorrhage surgeries to accelerate healing and eliminate the need for a second surgery Healthy Eye V i t r e o us Humor Re tina De t a c he d Retina S u b r e t in a l Fluid V i t r e o us Humor V i t r e o us Humor Retina P o s t e r ior V i t r e o us De t a c h m e nt Vitreous He m o rr h a ge
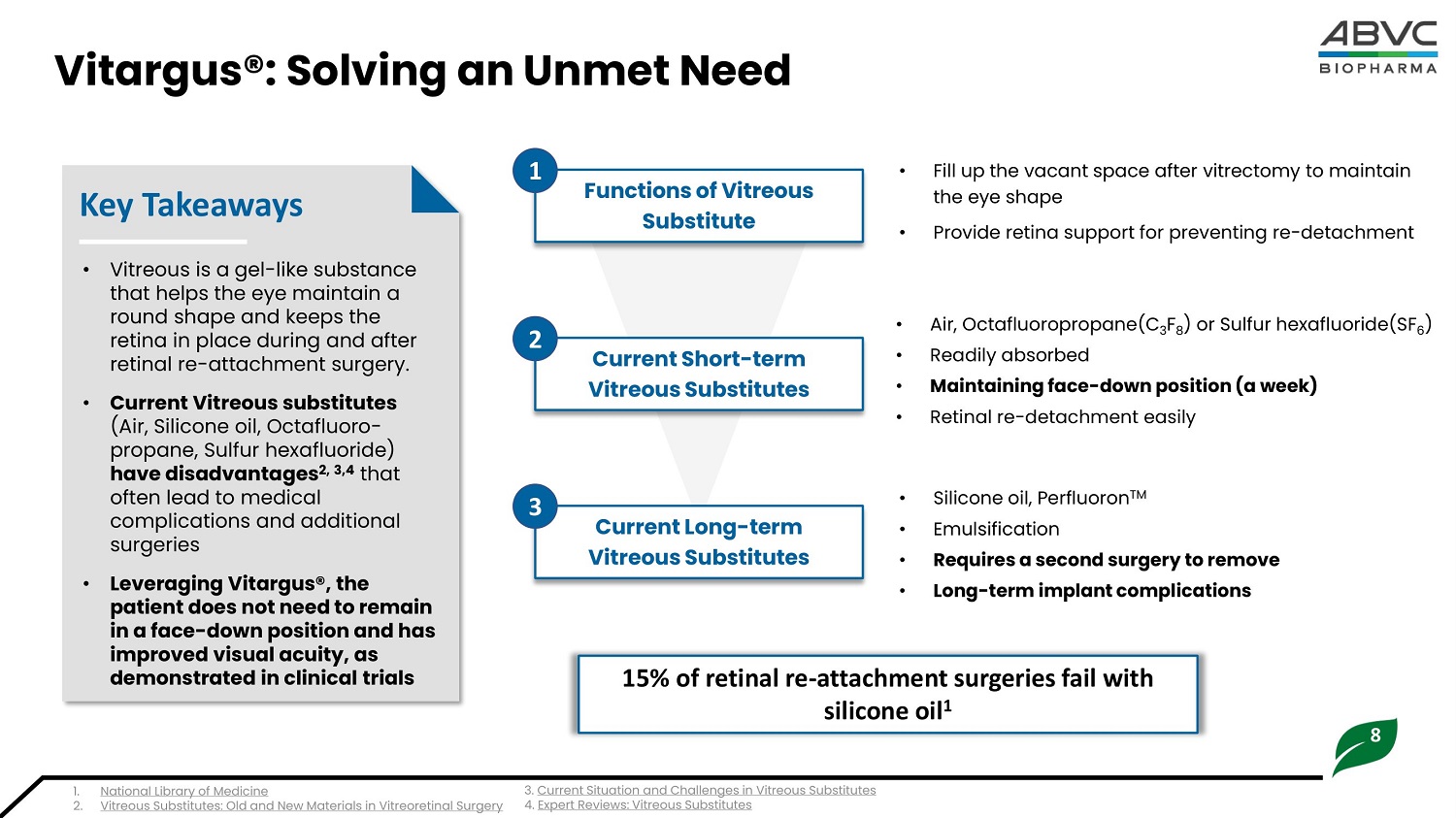
8 Vitargus®: S o lving an Unmet Need • Vi t r eous i s a ge l - li k e sub s ta n c e that helps the eye maintain a round shape and keeps the retina in place during and after retinal re - attachment surgery. • Cur r e nt Vit reo us su b stitu t e s (A i r , S ili c one oi l , O c taf l uo r o - propane, Sulfur hexafluoride) have disadvantages 2, 3,4 that often lead to medical complications and additional surgeries • Leveraging Vitargus®, the p a ti e nt d oe s no t n ee d to re m a in in a f a c e - d ow n p osition a nd h a s improved visual acuity, as d e m on st r a t e d in clini c a l trials Key Takeaways • Fill up the vacant space after vitrectomy to maintain the eye shape • Provide retina support for preventing re - detachment • S ili c one oi l , Perfl uoron TM • Emulsification • Req u i r e s a s e c o n d surge ry to r e mo v e • L o n g - t e rm i mp l a n t c o mp l i c a t i o n s Func t i o n s of V i t r e ous Substitute Current Sho r t - t erm V i t reou s Sub s t i t u t es Current L o ng - t e rm V i t reou s Sub s t i t u t es • Air, Octafluoropropane(C 3 F 8 ) or Sulfur hexafluoride(SF 6 ) • Readily absorbed • Ma i n t a i n i n g fa c e - do w n po s i t i o n (a w e e k) • Retinal re - detachment easily 15% of retinal re - attachment surgeries fail with silicone oil 1 1 2 3 1. National Library of Medicine 2. Vitreous Substitutes: Old and New Materials in Vitreoretinal Surgery 3. Current Situation and Challenges in Vitreous Substitutes 4. E x p e r t Re v i e ws : V i t re ou s Su b s t i t u t e s
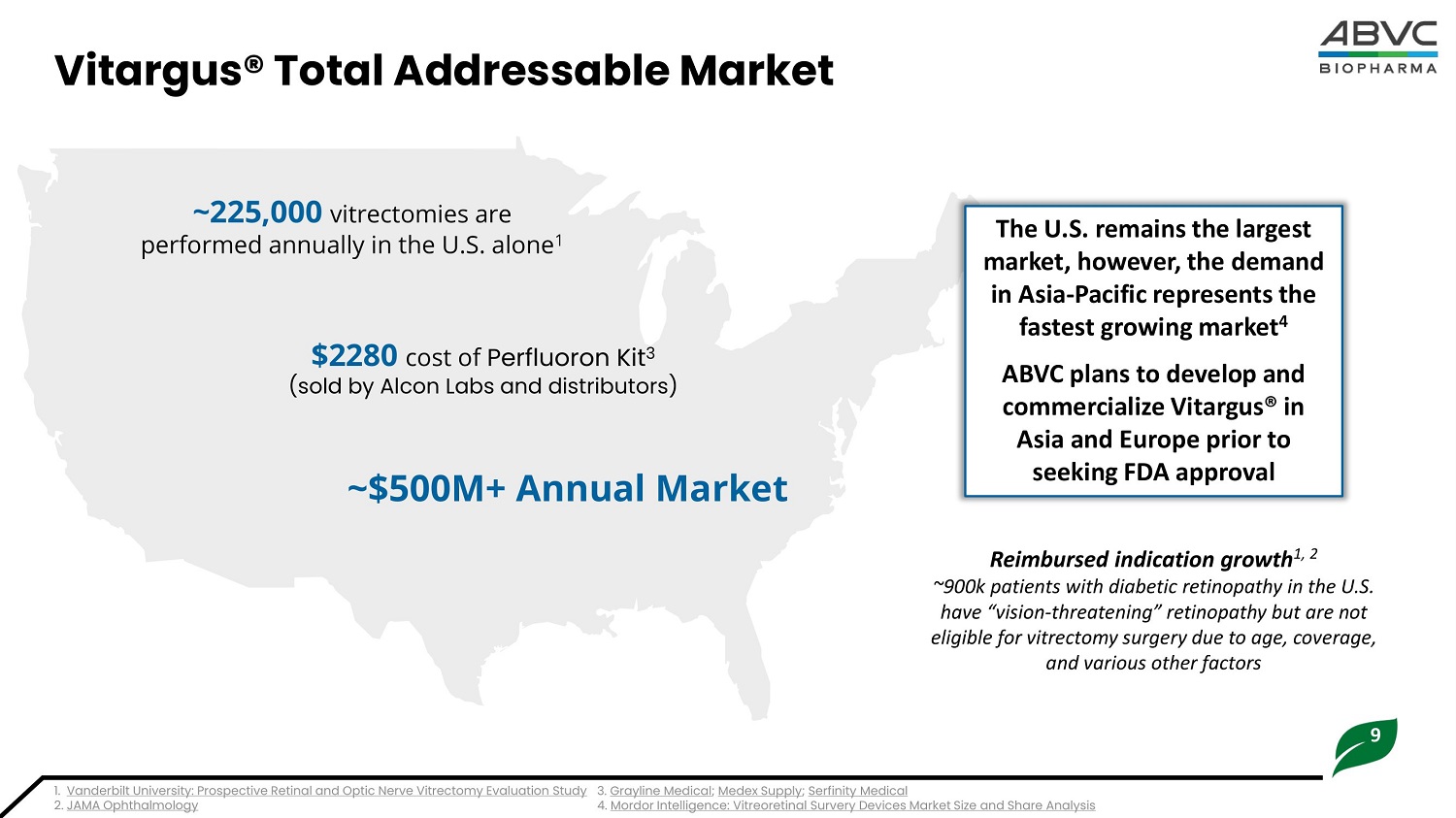
9 The U.S. remains the largest market, however, the demand in Asia - Pacific represents the fastest growing market 4 ABVC plans to develop and commercialize Vitargus® in Asia and Europe prior to seeking FDA approval Vitargus® Total Addressable Market ~225,00 0 v i trect o m i e s are p erfo r m e d an n ua l ly in the U.S. a l on e 1 1. Vanderbilt University: Prospective Retinal and Optic Nerve Vitrectomy Evaluation Study 3. Grayline Medica l ; Medex Supply ; Serfinity Medica l 2. JAMA Ophthalmology 4. Mordor Intelligence: Vitreoretinal Survery Devices Market Size and Share Analysis $228 0 cos t of P e r f luor on Ki t 3 (sold by Alcon Labs and distributors) ~$500M+ Annual Market Reimbursed indication growth 1, 2 ~900k patients with diabetic retinopathy in the U.S. have “vision - threatening” retinopathy but are not eligible for vitrectomy surgery due to age, coverage, and various other factors
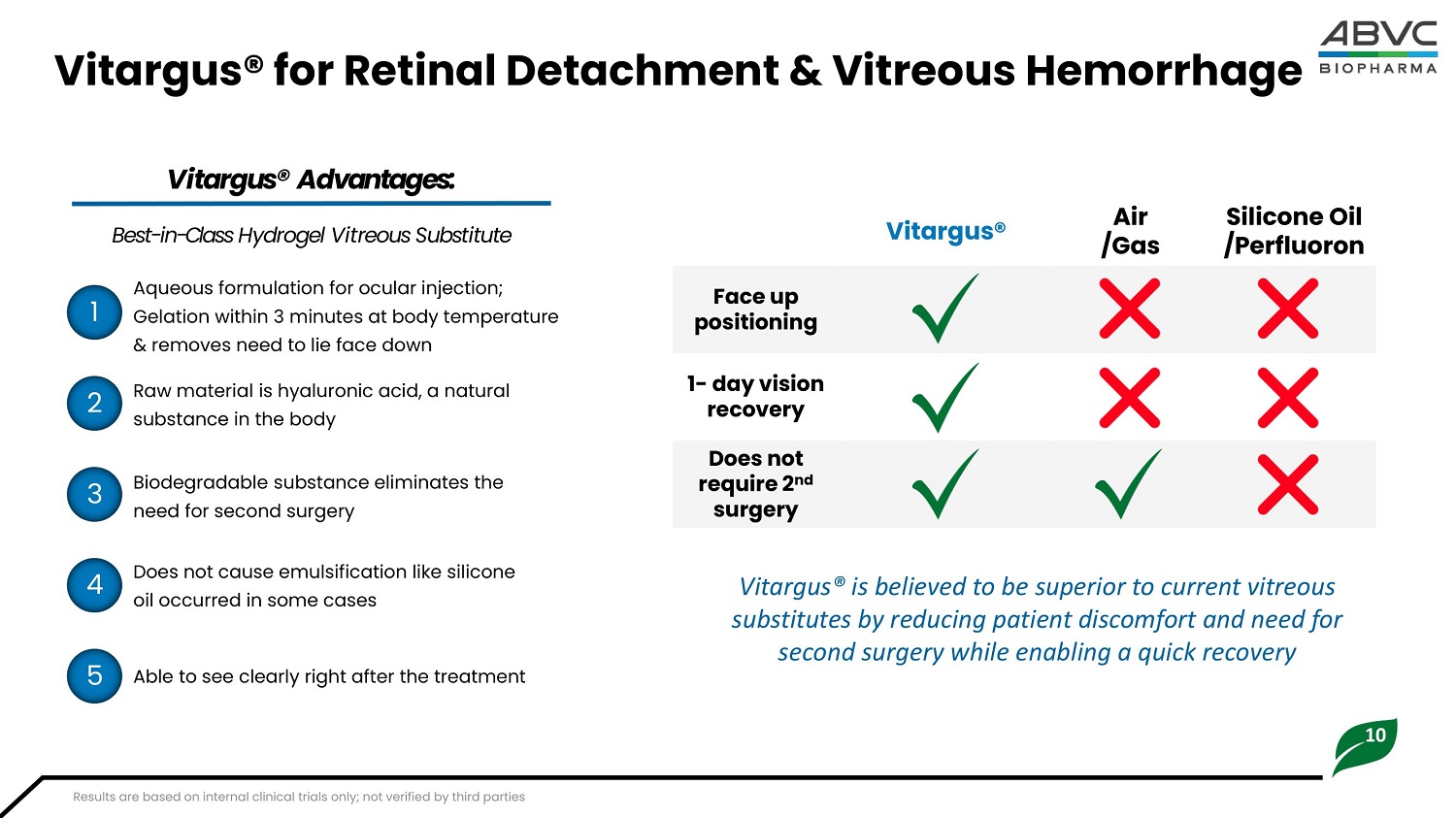
10 Vitargus® for Retinal Detachment & Vitreous Hemorrhage Aqueous formulation for ocular injection; Gelation within 3 minutes at body temperature & removes need to lie face down Raw material is hyaluronic acid, a natural substance in the body Si l i c one Oil /Perfluoron Air /Gas Vitargus® F a ce up p osi t ion i n g 1 - da y v i s ion recovery Does n o t re q u i re 2 nd surgery Vitargus® Advantages: Best - in - Class Hydrogel Vitreous Substitute Biodegradable substance eliminates the need for second surgery Does not cause high intraocular pressure (low thermal expansion coefficient) No side effects to retinal tissue and vision recovery seen to date 1 2 3 4 5 Vitargus® is believed to be superior to current vitreous substitutes by reducing patient discomfort and need for second surgery while enabling a quick recovery Results are based on internal clinical trials only; not verified by third parties
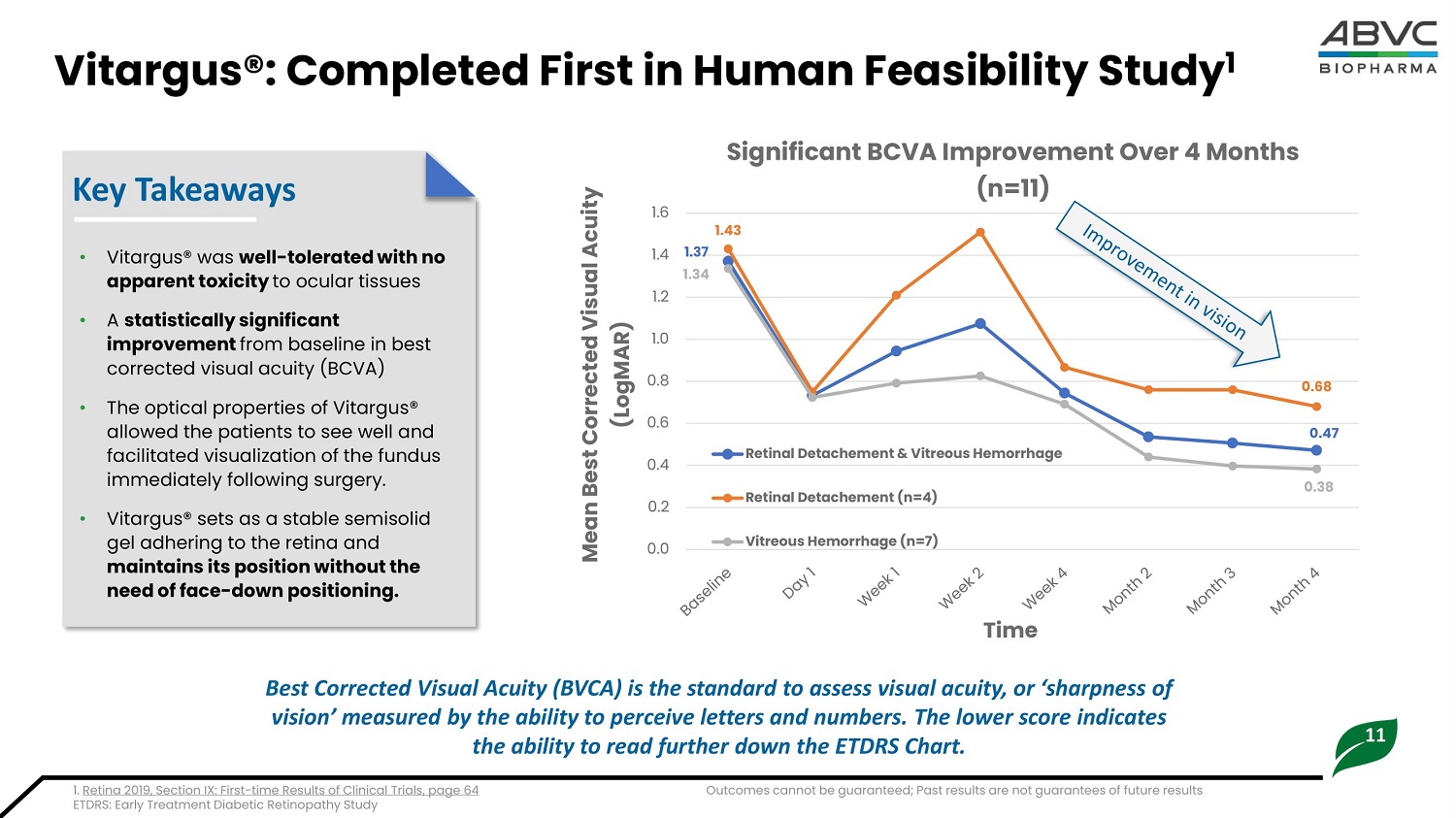
11 Vitargus®: Completed First in Hum a n Feasi b ili t y Stud y 1 Key Takeaways • V i t a rgus® wa s w e l l - t o l e ra t e d w i t h n o apparent toxicity to ocular tissues • A s t at i s t i c a l l y s i g n i fi c a n t i mpr o v e me n t from b a sel i ne i n b est corrected visual acuity (BCVA) • The optical properties of Vitargus® allowed the patients to see well and facilitated visualization of the fundus immediately following surgery. • Vitargus® sets as a stable semisolid gel adhering to the retina and ma i n t a i n s i t s po s i t i o n w i t h o u t t h e nee d of face - d o w n p o s i t i o ning. 1 . 3 7 0.47 1 . 4 3 0.68 1 . 3 4 0.3 8 0. 0 0 . 2 0. 4 0 . 6 0. 8 1 . 0 1 . 2 1 . 4 1 . 6 M e a n Best Correc t ed V isu a l Acu i ty (LogMAR) Tim e Significant BCVA Improvement Over 4 Months (n=11) Retinal Detachement & Vitreous Hemorrhage Ret i n a l D e t a c h em en t ( n = 4 ) V i t r e o u s He m orrh ag e ( n = 7 ) Best Corrected Visual Acuity (BVCA) is the standard to assess visual acuity, or ‘sharpness of vision’ measured by the ability to perceive letters and numbers. The lower score indicates the ability to read further down the ETDRS Chart. 1. Retina 2019, Section IX: First - time Results of Clinical Trials, page 64 E T D R S : E a rl y Tre a t m e nt D i a b e t i c R e t i no p a t hy S t udy Outcomes cannot be guaranteed; Past results are not guarantees of future results
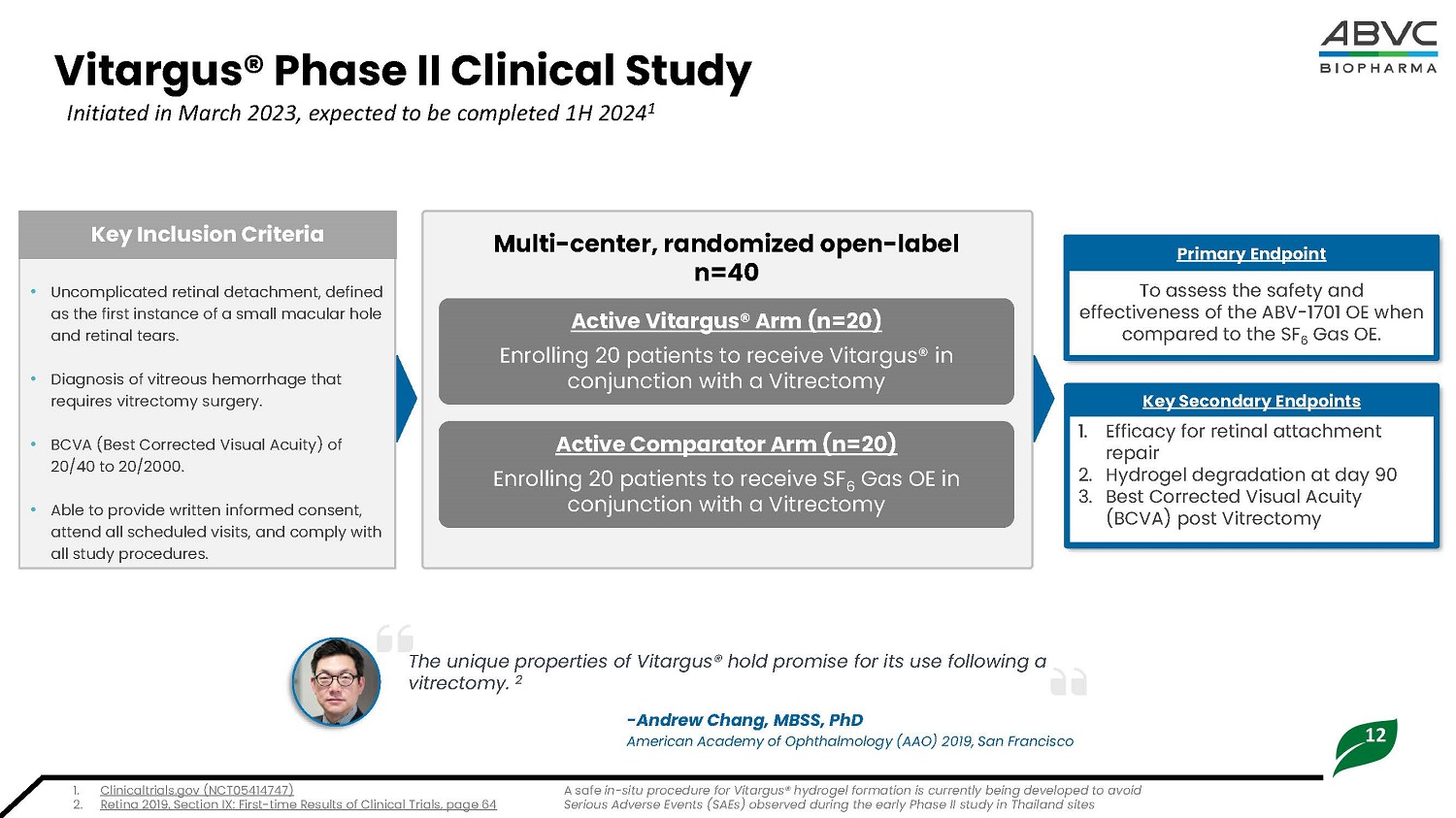
12 Vitargus® Ph a se II Cl i nical Study Initiated in March 2023, expected to be completed 1H 2024 1 • Uncomplicated retinal detachment, defined as the first instance of a small macular hole and retinal tears . • Diagnosis of vitreous hemorrhage that requires vitrectomy surgery. • BCVA (Best Corrected Visual Acuity) of 2 0/ 40 t o 2 0/ 2 00 0 . • Able to provide written informed consent, attend all scheduled visits, and comply with all study procedures. K ey I nc l u s io n Cri t eria To a ss e s s t h e s afety and effec t i vene s s of t he A B V - 17 01 O E when compared to the SF 6 Gas OE. Primary Endpoint 1. E ff i c a c y for re t i na l a tt a c hment repair 2. Hydrogel degradation at day 90 3. Best Corrected Visual Acuity (BCVA) post Vitrectomy Ke y S e c o n d a r y End p o in t s Act i v e Com p a r a t o r Ar m (n=20) Enr ol l ing 20 p a t i e nt s t o r e ce i v e S F 6 Gas OE in conjunction with a Vitrectomy Multi - center, randomized open - label n=40 Act i v e V i t a r g us ® Ar m (n=20) Enr ol l ing 20 p a t i e nt s t o r e ce i v e Vi t argus ® in conjunction with a Vitrectomy The unique properties of Vitargus® hold promise for its use following a vitrectomy. 2 - An d r e w Ch a n g , M B S S , P hD American Academy of Ophthalmology (AAO) 2019, San Francisco 1. C li n i c a l t ri a l s. g o v ( NCT0 5 4 1 474 7 ) 2. Retina 2019, Section IX: First - time Results of Clinical Trials, page 64
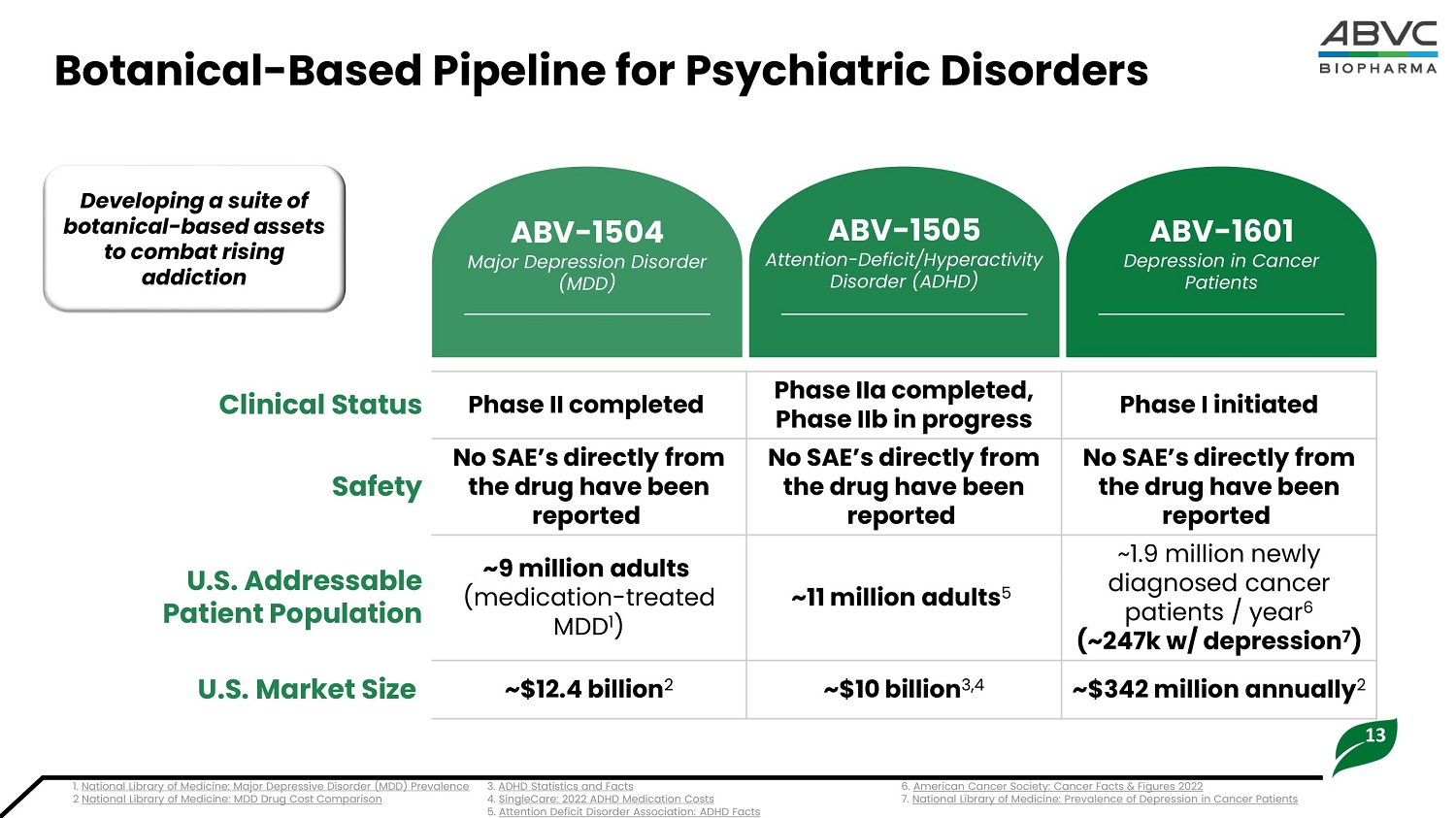
13 Botanical - Based Pipeline for Psychiatric Disorders ABV - 1504 Major Depression Disorder (MDD) Phase I initi a ted Phase I I a c omp l eted, Pha s e I I b in p rogre s s Phase II c omp l eted C l in i cal Status No SAE’s d i rect l y from the d rug have been reported No SAE’s d i rect l y from the d rug have been reported No SAE’s d i rect l y from the d rug have been reported Safety ~1.9 mil l ion n e wly diagnosed cancer pati e n t s / yea r 6 ( ~247k w/ de p ress i o n 7 ) ~11 mil l ion a d ult s 5 ~9 mi l l i on ad ults (m e di c atio n - t r e a t ed MDD 1 ) U. S . Addr e ssable P a ti e nt P o pulation ~$ 3 42 mi l l i on annual l y 2 ~$10 b i l l io n 3 ,4 ~$12.4 b i l l io n 2 U.S. Mar k et S i ze De v el o p i n g a sui t e o f b o t a nic a l - b a s ed a ss ets to combat rising addiction ABV - 1505 Attention - Deficit/Hyperactivity Disorder (ADHD) ABV - 1601 Depression in Cancer Patients 2 National Library of Medicine: MDD Drug Cost Comparison 1 . N a ti o n a l L i b r a r y o f Med i c i ne : Ma j o r De p r e s s i v e D i s or d e r ( MDD ) P r ev a l en c e 3 . AD HD St a ti s ti c s a n d F a c ts 4. S i n g l eC a r e : 2 0 2 2 AD HD Med i c a ti o n C o s ts 5. Attention Deficit Disorder Association: ADHD Facts 6. American Cancer Society: Cancer Facts & Figures 2022 7. National Library of Medicine: Prevalence of Depression in Cancer Patients

14 ABV - 1504: Innov a tive Bota n ical Asset for MDD • AB V - 1 50 4 (PD C - 1 4 21 capsule ) is a s ingl e - herb botanical drug extract from the dry root of Polygala tenuifolia Willd • Safety assessment: Demonstrated its safety with no SAEs from the completed Ph ase I and Phase II studi e s. • Efficacy assessment: Demonstrated its efficacy for treating Major Depressive Disorder (MDD) patients from the Phase II clinical studies. • S t a b ili t y a t l e a st 36 m o n t h s post encapsulation IP - Protected Process Raw Materials (dry roots of Yuan Zhi) Extraction Purification Isolation Encapsulation No methylation process required 1 2 3 4 5 ABV - 1504 Summary Highlights Outcomes cannot be guaranteed; Past results are not guarantees of future results S ummary H i g h l i g h t s are b a se d on i n t e rna l l y ge ne r a t e d d a t a S A E s: S e ri ou s A d v e rse E v e n t s

15 ABV - 1504 Completed Ph a se I H ighlights Ph ase I Ph ase of D e velopm e n t ABV - 1504 (PDC - 1421 capsule, extract of Radix Polygalae ) I n ves t iga t i o n al prod u ct 380 mg U n it dose Oral Mode of adm i n is t rati o n A Dose Es c a l ation Ph ase I St u dy of P D C - 1421 C aps u le to Eval u ate t h e Safety in H ea l th Volunteers St u dy title 1, 3 , 6, 10 caps u les, O n ce da i ly after meal Adm i n is t rated un its 30 eva l u ated N u mb e r of sub j ects Taipei Vetera n s General Hosp i tal (TVGA) Center • The oral administration of AB V - 1 504 Capsules in h e althy volunteers was safe and well - tolerated for doses from 380 mg t o 3,800 mg • No subject had serious adverse event and no subject discontinued due to adverse event Key Takeaways Outcomes cannot be guaranteed; Past results are not guarantees of future results

16 ABV - 1504 Completed Ph a se II Hig hlights MADRS: Montgomery – Åsberg Depression Rating Scale T I D : T hr e e t i me s d a il y • Th e H i gh - D ose g r o u p (760 m g T I D) of A B V - 1 504 d e m on s t r a ted a cli n ic a l l y m e a nin g ful s c o r e in M AD RS c o m p a r e d t o t he P l a c eb o g r o u p . • Com p a r ed w i t h p r i or a p p r ov ed Fluoxetine(Prozac) antidepressant, A B V - 1 504 H i gh - D o s e de m o n s t r a t e d a m uc h be tt e r M AD RS s c o r e (4. 1 - p oint r ed uc tion ) f r om P l a ce b o g r oup than that of Fluoxetine (2.3 - point reduction). • Tre a t men t of A B V - 1 504 did not i nc r ease any r i s k s i n t e r ms of v i t al si gns, physi cal ex a ms, sui c i d a l i de a t i on, and sui c i d a l beh a v i or du r i ng t r e a t ment a nd f ol l o w - up p e r i o d . • N o sev e r e a d v e r se ev en ts (SAEs) occurred. • Demo ns t r a t ed A B V - 1 504 w as sa f e and w el l - t ol e r a t ed for fu r t her c l i ni cal advancement. Key Takeaways Week 0 Week 2 Week 4 29.43 27.47 28.55 - 7.38 - 10.81 - 13.19 - 6.42 - 7.63 - 5.45 - 8.70 - 9.21 - 9.20 40 30 20 10 0 - 1 0 - 2 0 W e e k 6 Efficacy Results (Part II) MADRS Net Change - ITT Two ABV - 1504 Capsules TID (n=21) One ABV - 1504 Capsule TID (n=19) Placebo TID (n=20) Mean baseline MADRS 29.0 - 13.2 31.4 33.4 - 15.6 32.7 - 15.3 - 16.7 26.9 - 8.0 - 18.6 - 4.0 - 3.8 - 4.0 - 5.0 - 2.3 - 3.7 - 2 0 0 20 Efficacy Results (Part II) MADRS Net Change Approved Antidepressants* ABV - 1504 High Dose (6 wks) Vortioxetine (6 - 8 wks) Vilazodone (8 wks) Brexpiprazole (6 wks) Levomilnacipra (8 wks) Esketamine + AD (4 wks) *Values taken as the median of ranges collected, see appendix for detailed data ranges Outcomes cannot be guaranteed; Past results are not guarantees of future results Baseline MADRS Score LS M e an Ch a nge f rom Baseline Placebo - subtracted Difference 37.4
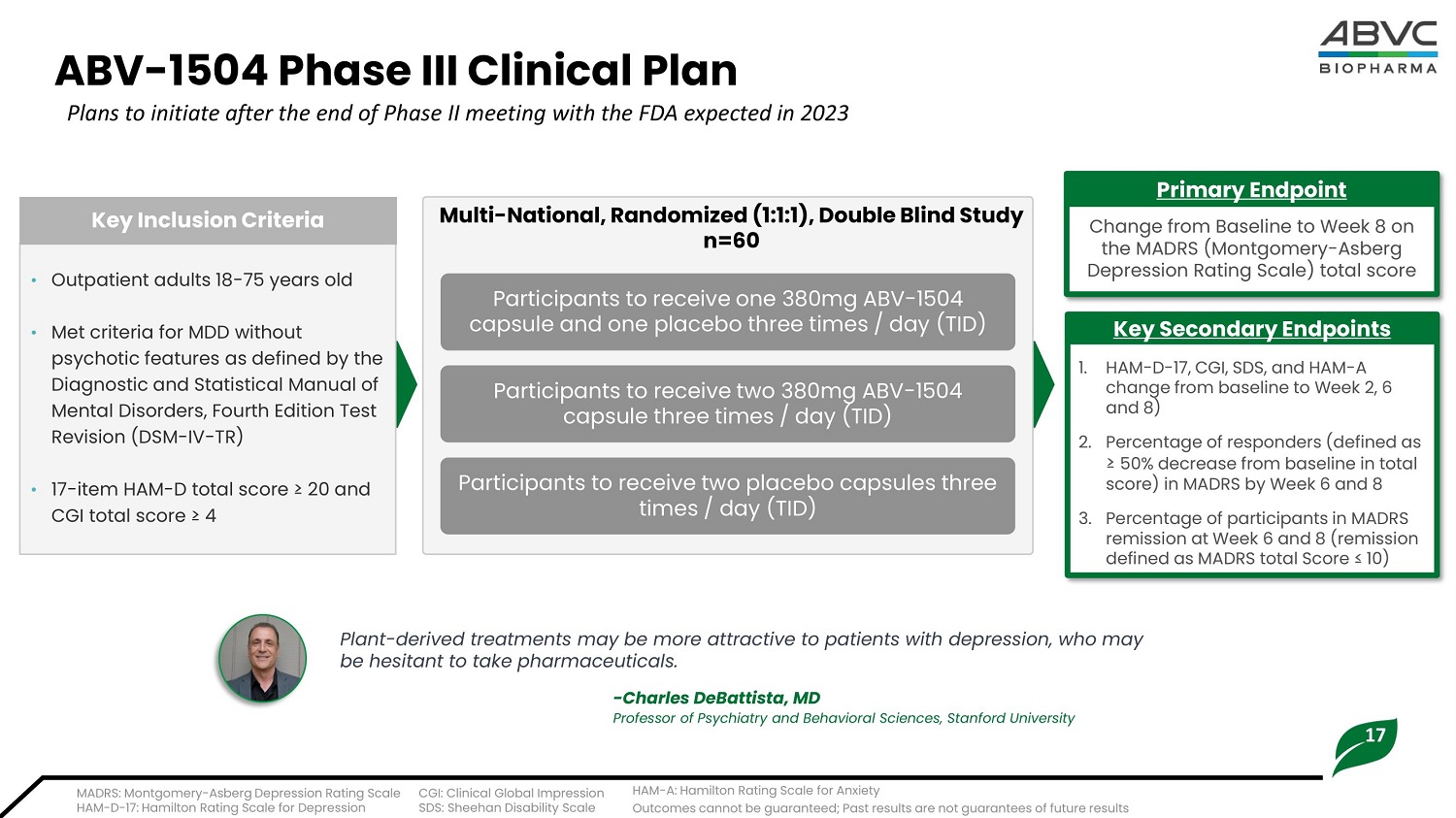
17 ABV - 1504 Ph a se III Cl i nical P lan Plans to initiate after the end of Phase II meeting with the FDA expected in 2023 • O u t p a t i ent a d ul t s 1 8 - 7 5 y ea rs ol d • Met criteria for MDD without psychotic features as defined by the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition Test Revision (DSM - IV - TR) • 1 7 - i t em H A M - D t o t a l sc or e ≥ 2 0 and CGI t o t a l score ≥ 4 K ey I nc l u s io n Cri t eria Change from Baseline to Week 8 on the MADRS (Montgomery - Asberg Depression Rating Scale) total score Pri ma r y En d p o i nt 1. HA M - D - 1 7 , CGI , S D S, a nd HA M - A ch ange f r om b a sel i ne t o Week 2, 6 and 8) 2. P e r cent a ge of r esponde rs (defined as ≥ 50 % dec r ease f r om b a sel i ne i n t o t al score) i n MADRS by Week 6 and 8 3. P e r cen t a g e of p a r t i ci p a n t s i n M A D RS r em i ssi on at Week 6 and 8 (remi ssi on de f i ned as MADRS t o t al Score ≤ 1 0) K e y Sec o nd a r y En d p o i n t s Participants to receive one 380mg ABV - 1504 capsule and one placebo three times / day (TID) Multi - National, Randomized (1:1:1), Double Blind Study n=60 Plant - derived treatments may be more attractive to patients with depression, who may be hesitant to take pharmaceuticals. - Charles DeBattista, MD Professor of Psychiatry and Behavioral Sciences, Stanford University MADRS: Montgomery - Asberg Depression Rating Scale CGI: Clinical Global Impression HAM - D - 17: Hamilton Rating Scale for Depression SDS: Sheehan Disability Scale H AM - A: H a m i l t on R a t i ng S c a l e for Anx i e t y Outcomes cannot be guaranteed; Past results are not guarantees of future results Participants to receive two 380mg ABV - 1504 capsule t hr ee t i m e s / day ( TID) Participants to receive two placebo capsules three t i m e s / day (TI D )

18 ABV - 150 5 : Innov a tive Bota n ical Asset for ADHD • AB V - 1 50 5 ( P D C - 1 4 21 capsule ) is a singl e - herb botanical drug extract from the dry root of Polygala tenuifolia Willd • Safety assessment: Demonstrated its safety with no SAEs from the completed Ph ase I and Phase II ( Par t I) cli nical studies. • Efficacy assessment: Demonstrated its e ff icacy f or tr e at i ng AD HD pati e nt s f r om the comple ted Phase II clini c a l studies (Part I). • IP Protection: Global patent granted incl udin g US, EU a nd Asian coun t r i es . IP - Protected Process Raw Materials (dry roots of Yuan Zhi) Extraction Purification Isolation Encapsulation No methylation process required 1 2 3 4 5 ABV - 1505 Summary Highlights Outcomes cannot be guaranteed; Past results are not guarantees of future results S ummary H i g h l i g h t s are b a se d on i n t e rna l l y ge ne r a t e d d a t a
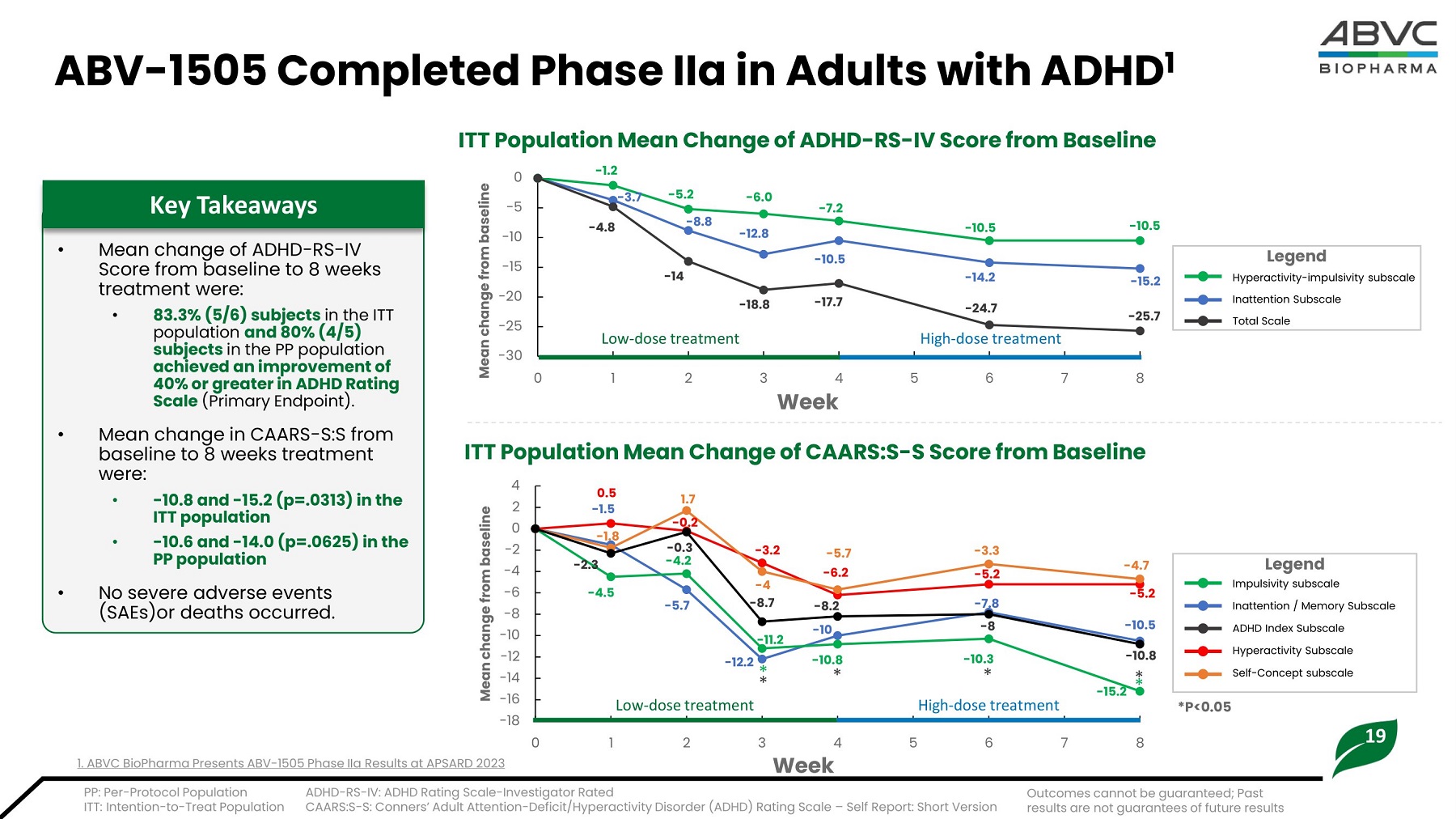
19 - 1 2 .8 - 10. 5 - 15 .2 - 5.2 - 8 .8 - 6 .0 - 7.2 - 10. 5 - 10. 5 - 1.2 - 3 .7 - 4.8 - 14 - 1 8 .8 - 17.7 - 2 5 .7 0 - 5 - 10 - 15 - 20 - 25 - 3 0 0 3 4 Week 5 L o w - d o s e treat m ent 1 2 - 14.2 - 24.7 High - dose treatment 6 7 8 Mean c ha n ge from basel i ne ABV - 1505 Completed Ph a se IIa in Ad u lts with AD H D 1 • Mean change of ADHD - RS - IV Score from baseline to 8 weeks treatment were: • 8 3 .3% (5/6) s u bj e c t s i n t h e I TT p o p u l a ti o n and 8 0 % (4 /5) s u bj e c t s i n t h e PP p o p u l a ti o n ac h iev e d an impr ov e m e n t of 4 0 % or g r eat er in ADHD R a t i n g S c al e (Pr i m a ry E n dp o i n t). • Mean change in CAARS - S:S from baseline to 8 weeks treatment were: • - 1 0 .8 and - 1 5 .2 (p =. 0 3 1 3) in t h e ITT population • - 1 0 .6 and - 14 .0 (p =. 0 6 2 5) in t h e PP pop u lat ion • N o seve r e a d ve r se even t s ( S A E s ) or d ea t hs oc c ur re d . Key Takeaways PP : P e r - P ro t oc ol P opu l a t i on I TT : I n t e n t i on - t o - T re a t Pop u l a t i on ITT Population Mean Change of ADHD - RS - IV Score from Baseline ADHD - RS - IV: ADHD Rating Scale - Investigator Rated CAARS:S - S: Conners’ Adult Attention - Deficit/Hyperactivity Disorder (ADHD) Rating Scale – Self Report: Short Version ITT Population Mean Change of CAARS:S - S Score from Baseline Legend Hyperactivity - impulsivity subscale Inattention Subscale T o t a l S c a l e Legend Impulsivity subscale I n a t t en t i on / Mem or y Sub s c a l e AD HD I n d ex Sub s c a l e Hy p er a c ti v i ty Sub s c a l e Self - Concept subscale *P<0 . 05 - 5 .7 - 1 2 .2 - 3 .2 - 10. 8 - 10. 3 - 15 .2 - 5 .7 - 6.2 0.5 - 1.5 - 1.8 - 2.3 - 4.5 1.7 - 0 .2 - 0.3 - 4.2 - 4 - 8 .7 - 8 .2 - 10 - 3 .3 - 5 .2 - 7.8 - 8 - 4.7 - 5.2 - 10.5 - 10. 8 4 2 0 - 2 - 4 - 6 - 8 - 10 - 12 - 14 - 16 - 18 0 1 2 3 4 Week 5 6 7 8 Mean c ha n ge from basel i ne L o w - d o s e treatment H i g h - d o s e treatment - 11.2 * * * * * Ou t c o m e s c a nnot b e g u a ra n t ee d ; P a st results are not guarantees of future results 1. ABVC BioPharma Presents ABV - 1505 Phase IIa Results at APSARD 2023
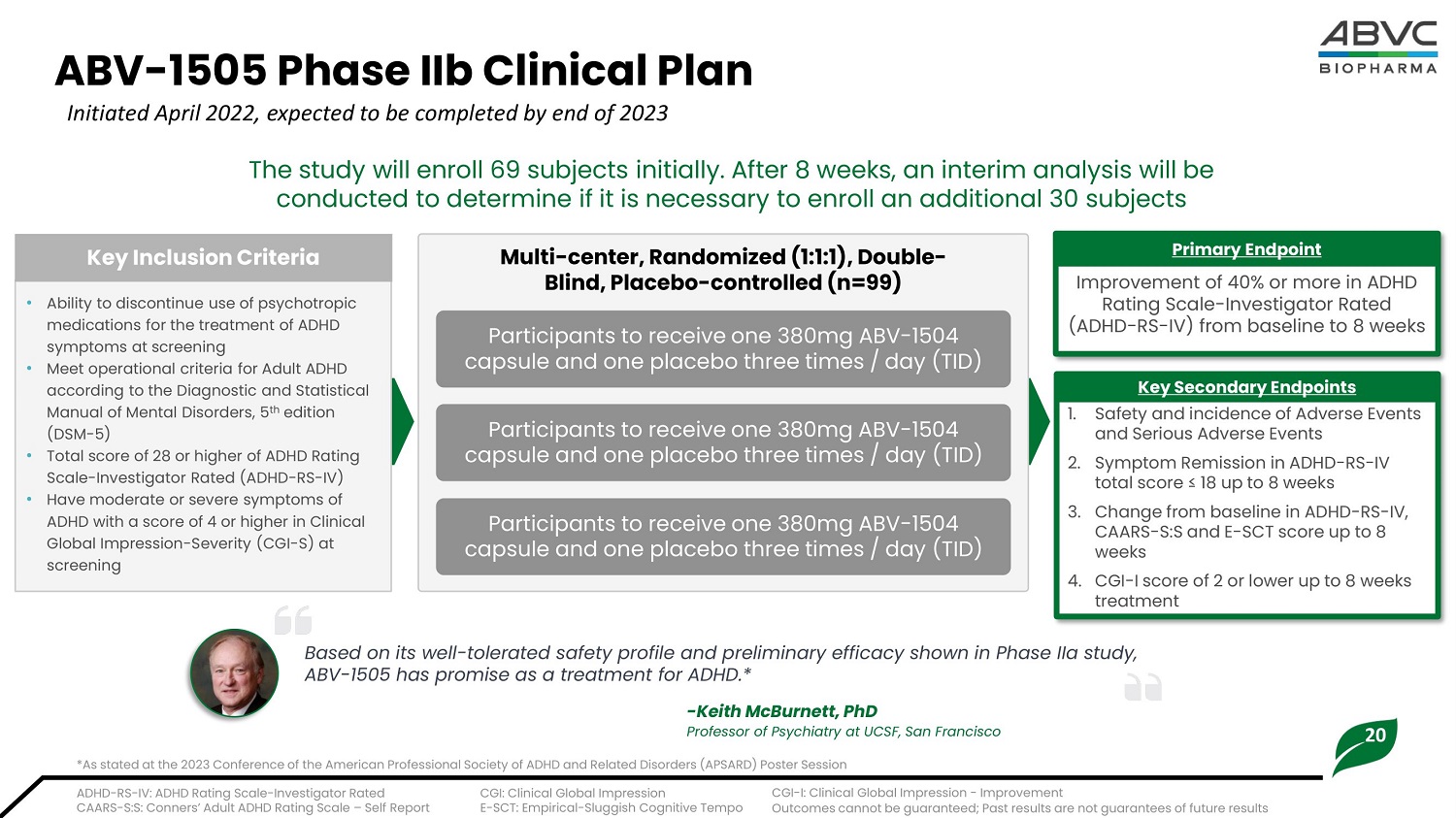
20 ABV - 1505 Ph a se IIb Clinical Plan Initiated April 2022, expected to be completed by end of 2023 • Ability to discontinue use of psychotropic medications for the treatment of ADHD symptoms at screening • M ee t o p e r a t i o n al c r i t e r i a f or A d u l t AD H D according to the Diagnostic and Statistical Manual of Mental Disorders, 5 th edition (DSM - 5) • Tota l s c ore of 28 o r hi gh e r of ADHD R a t i ng Scale - Investigator Rated (ADHD - RS - IV) • Have moderate or severe symptoms of AD H D w i th a s c ore of 4 o r hi gh e r i n C l i ni c al Global Impression - Severity (CGI - S) at screening K ey I nc l u s io n Cri t eria Improvement of 40% or more in ADHD Rating Scale - Investigator Rated (ADHD - RS - IV) from baseline to 8 weeks Primary Endpoint 1. Sa f e t y and i nc i den c e of Adverse Events a nd Se r i ous A d ve r se E ven ts 2. Sym p t om Rem i ssi on i n ADH D - RS - I V t o t al score ≤ 1 8 up t o 8 w ee k s 3. Change from baseline in ADHD - RS - IV, CAARS - S: S and E - SCT score up t o 8 weeks 4. CGI - I sco r e of 2 or l ow er up t o 8 w e e k s treatment Ke y S ec o n d a r y En d p oin t s M u l t i - c e n t er, R a ndom i zed (1:1:1), Do uble - Bl i nd, Pl a ceb o - c o n t r o l l ed (n=99) Par t ici pants t o r e ce i v e one 3 8 0m g A B V - 1 50 4 cap su le an d on e plac e bo th r e e t i m e s / day ( T ID ) Participants to receive one 380mg ABV - 1504 capsule and one placebo three times / day (TID) Participants to receive one 380mg ABV - 1504 capsule and one placebo three times / day (TID) The study will enroll 69 subjects initially. After 8 weeks, an interim analysis will be conducted to determine if it is necessary to enroll an additional 30 subjects ADHD - RS - IV: ADHD Rating Scale - Investigator Rated CAARS - S:S: Conners’ Adult ADHD Rating Scale – Self Report CGI: Clinical Global Impression E - SCT: Empirical - Sluggish Cognitive Tempo CGI - I: Clinical Global Impression - Improvement Outcomes cannot be guaranteed; Past results are not guarantees of future results Based on its well - tolerated safety profile and preliminary efficacy shown in Phase IIa study, ABV - 1505 has promise as a treatment for ADHD.* - Kei t h M cB u r ne t t , P hD Professor of Psychiatry at UCSF, San Francisco *As stated at the 2023 Conference of the American Professional Society of ADHD and Related Disorders (APSARD) Poster Session
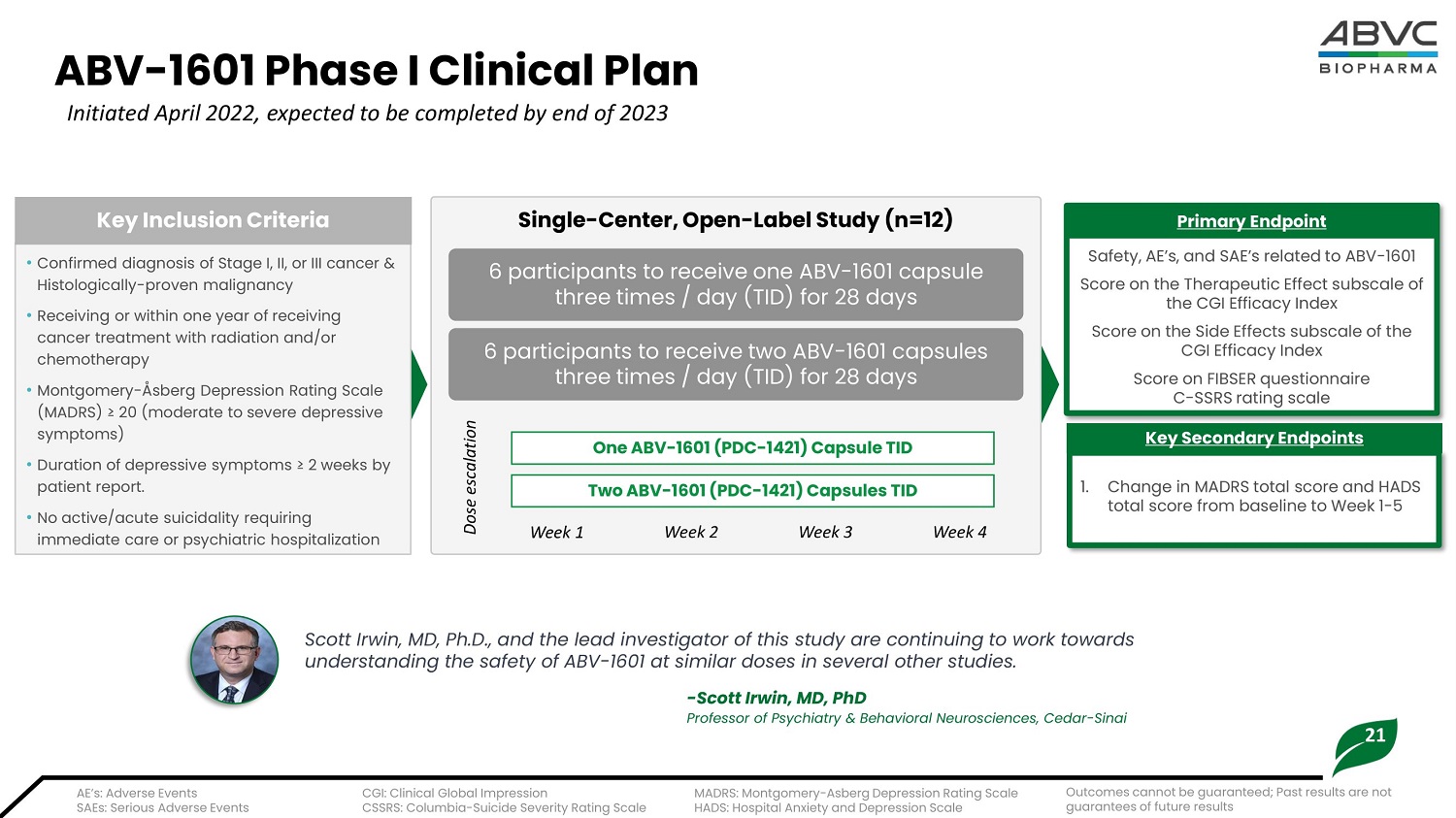
21 ABV - 1601 Phase I Cl i nical Plan Initiated April 2022, expected to be completed by end of 2023 • Confirmed diagnosis of Stage I, II, or III cancer & Histologically - proven malignancy • Receiving or within one year of receiving cancer treatment with radiation and/or chemotherapy • Montgomery - Åsberg Depression Rating Scale ( M AD R S ) ≥ 2 0 ( m od era t e to s e v e re d e pr e ss i v e symptoms) • Duration of depressive symptoms ≥ 2 weeks by patient report. • No active/acute suicidality requiring immediate care or psychiatric hospitalization K ey I nc l u s io n Cri t eria S a f e ty , A E ’ s, a n d S A E ’ s r e l a te d to A B V - 16 0 1 Score on the Therapeutic Effect subscale of the CGI Efficacy Index Score on the Side Effects subscale of the CGI Efficacy Index S c o re o n F I BS ER q u e sti o nn a i re C - SS R S r a ti n g s c a l e Primary Endpoint 1 . Ch a n g e i n MA D R S to t a l s c o re a n d HA D S to t a l s c o re fr o m b a se li n e to We e k 1 - 5 Ke y S e c o n d a r y End p o in t s S i ngle - Ce n t er, O p e n - La b el S t udy (n=12) 6 par t ici pants t o r e ce i v e one AB V - 1 6 0 1 cap su le th r e e t i m e s / day ( TI D ) f or 28 day s 6 participants to receive two ABV - 1601 capsules th r e e t i m e s / day ( TI D ) f or 28 day s Scott Irwin, MD, Ph.D., and the lead investigator of this study are continuing to work towards understanding the safety of ABV - 1601 at similar doses in several other studies. - Scott Irwin, MD, PhD Professor of Psychiatry & Behavioral Neurosciences, Cedar - Sinai A E ’ s: A dv e rse E v e n t s S A E s: S e ri ou s A d v e rse E v e n t s CGI: Clinical Global Impression CSSRS: Columbia - Suicide Severity Rating Scale MADRS: Montgomery - Asberg Depression Rating Scale H A D S : H osp i t a l Anx i e t y a nd D e p re ss i on S c a l e O ne A B V - 1 6 01 ( P DC - 1 4 2 1 ) C a p s ule T ID Dose escalation W eek 1 W eek 2 W eek 3 W eek 4 T w o A B V - 1 6 01 ( P DC - 1 4 2 1 ) C a p s ules T ID O u t c om e s c a nn ot b e g uar a n t ee d ; P a st re su l t s a re no t g uar a n t ee s of fut ure re su l t s
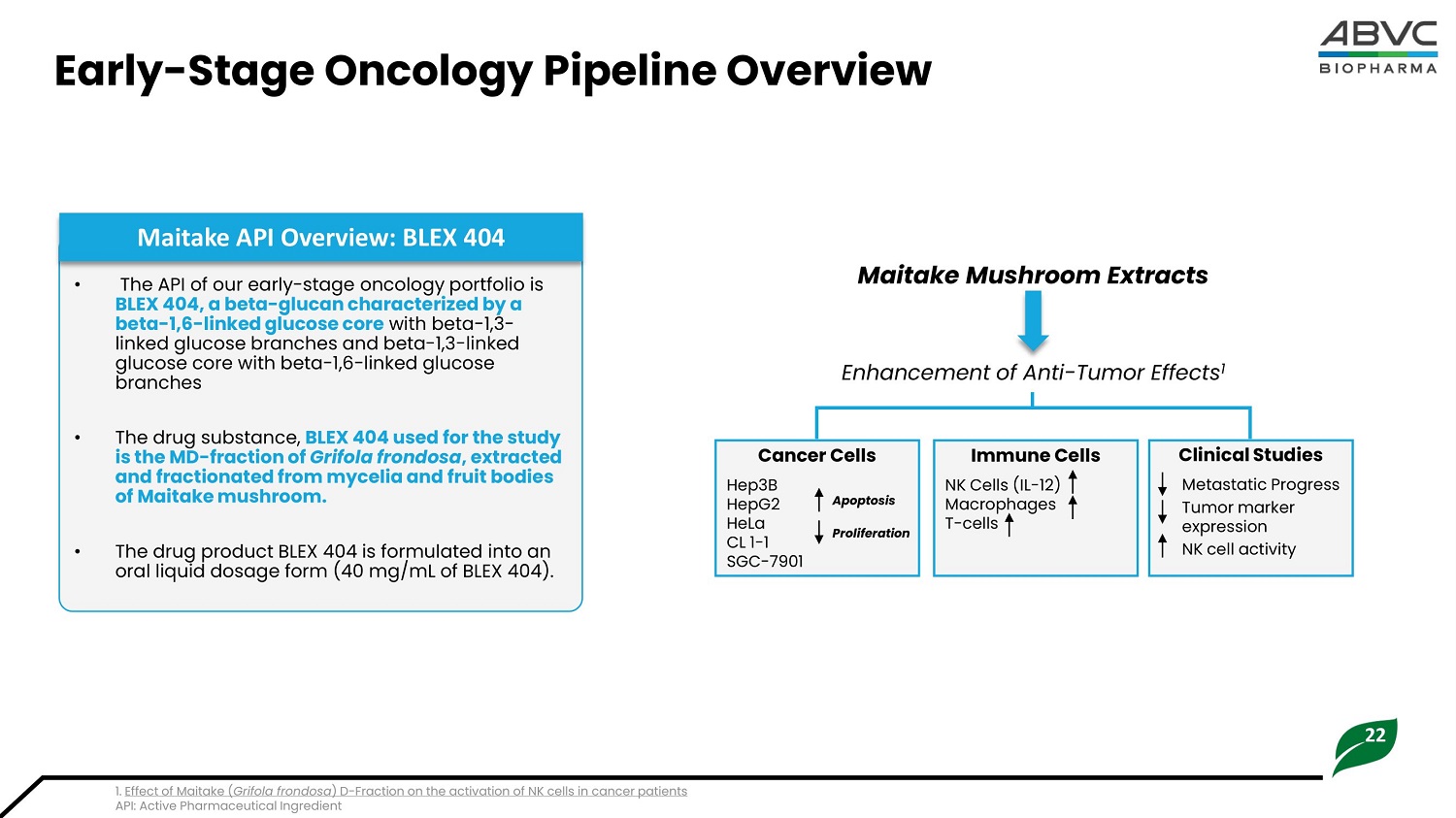
22 Early - Stage Oncology Pipeline Overview 1. Effect of Maitake ( Grifola frondosa ) D - Fraction on the activation of NK cells in cancer patients API: Active Pharmaceutical Ingredient • Th e API of ou r e a rl y - s t a g e onc ol ogy p or t fol i o i s BLEX 404, a beta - glucan characterized by a beta - 1,6 - linked glucose core with beta - 1,3 - linked glucose branches and beta - 1,3 - linked glucose core with beta - 1,6 - linked glucose branches • Th e d rug su b s t a n c e, BL EX 4 04 u s e d for the s t u dy i s t he MD - fra c t i o n of Gri fo l a fro n do s a , e x t ra c t e d and fra c t i o nat e d fr o m m y ce li a and fruit b o d i e s of Ma i t a k e m ushr o o m . • The drug product BLEX 404 is formulated into an oral liquid dosage form (40 mg/mL of BLEX 404). Maitake API Overview: BLEX 404 Maitake Mushroom Extracts Cl i n ical St u d i es Met a st a tic Pr og ress T u m o r m a rker expression NK c ell ac ti v ity Immu n e C ells NK C e lls (IL - 12) Ma c ro p h ag es T - cells Ca n cer Ce l ls Hep3B HepG2 Apoptosis HeLa Proliferation CL 1 - 1 SGC - 7901 Enhancement of Anti - Tumor Effects 1
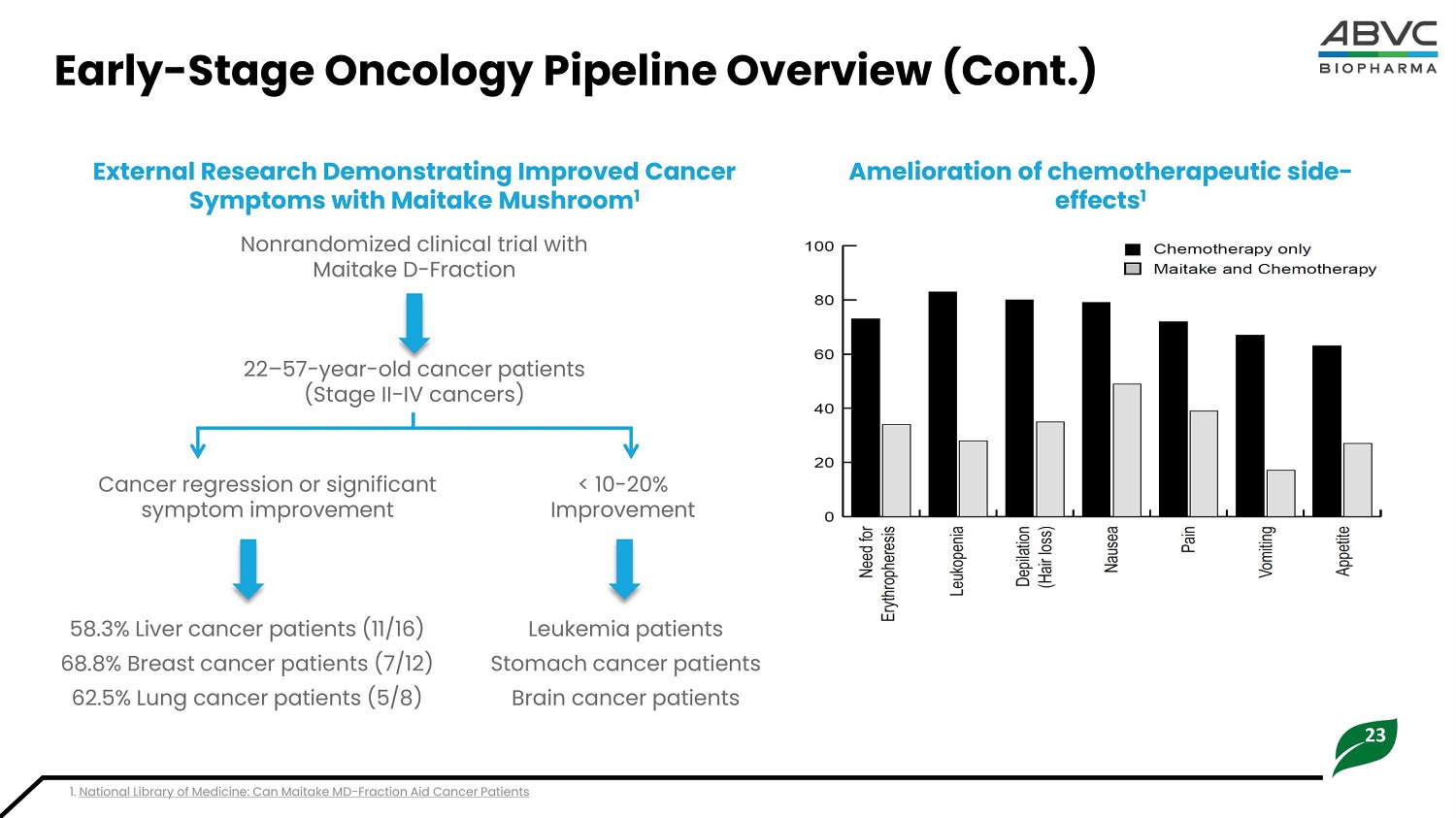
23 Earl y - Sta g e Onc o logy Pipeline Overview (Cont.) Amelioration of chemotherapeutic side - effects 1 External Research Demonstrating Improved Cancer Symptoms with Maitake Mushroom 1 Nonrandomized clinical trial with Maitake D - Fraction 22 – 57 - year - old cancer patients (Stage II - IV cancers) 1. National Library of Medicine: Can Maitake MD - Fraction Aid Cancer Patients C a nce r r e gr e ssion or significant symptom improvement < 1 0 - 2 0 % Improvement 5 8. 3 % L iv er c a ncer p a ti en t s ( 1 1 /1 6 ) 68.8% Breast cancer patients (7/12) 6 2 . 5 % L ung cance r pati e nt s ( 5 /8 ) Leukemia patients S t o mach cance r pati e nts Brain cancer patients
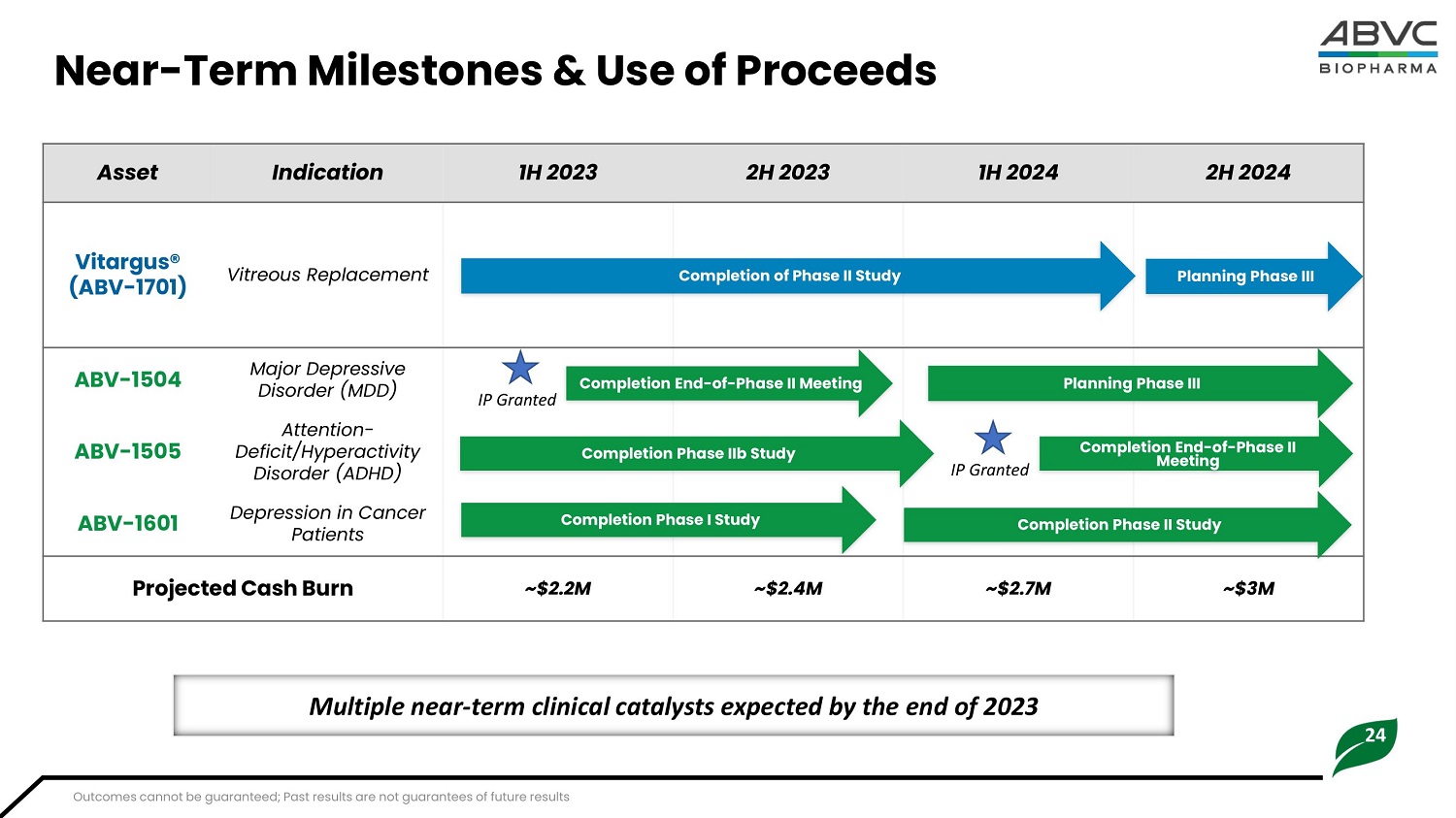
24 Nea r - Te r m Milestones & Use of Proceeds Multiple near - term clinical catalysts expected by the end of 2023 2H 2024 1H 2024 2H 2023 1H 2023 Indication Asset Com p l et i on of Ph as e I I Stu d y P la nn i ng Ph as e II I Vitreous Replacement Vitargus® (AB V - 1701) Com p l et i on End - of - Ph as e I I M e et i ng P la nn i ng Ph as e II I IP Granted Completion Phase IIb Study Completion End - of - Phase II IP Granted Meeting Com p l et i on Ph as e I Stu d y Com p l et i on Ph as e I I Stu d y Major Depressive Disorder (MDD) ABV - 1504 Attention - Defici t /H y pe r ac t ivi t y Disorder (ADHD) ABV - 1505 Depression in Cancer Patients ABV - 1601 ~$3M ~$2.7M ~$2.4M ~$2.2M Pro j ec t ed C a sh Burn Outcomes cannot be guaranteed; Past results are not guarantees of future results

N A SD A Q: ABVC Howard Doong 44370 Old Warm Springs Blvd. Fr e m ont, California 9 4 5 38 Of f ice: (51 0 ) 6 6 8 - 088 1 howard.doong@ambrivis.com Investor Relations Contact: Tom M a sterson (6 4 6) 57 3 - 3 2 16 tmasterson@allelecomms.com
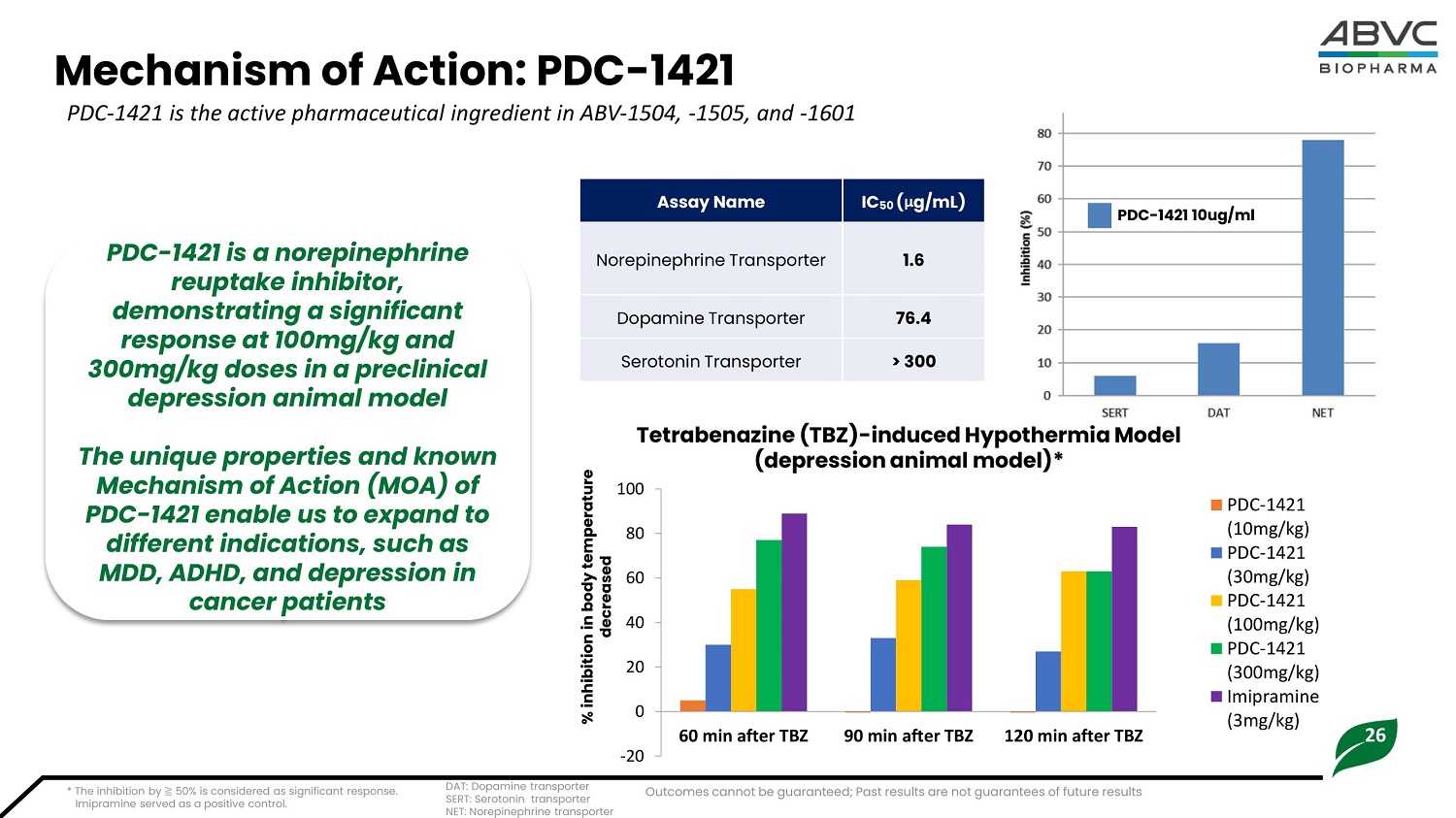
26 Mechanism of Action: P D C - 1421 PDC - 1421 is the active pharmaceutical ingredient in ABV - 1504, - 1505, and - 1601 Tetrabenazine (TBZ) - induced Hypothermia Model (de p re s si o n a n i mal m o d el )* - 2 0 0 20 40 60 80 1 0 0 60 min after TBZ 90 min after TBZ 120 min after TBZ PDC - 1421 ( 10mg/kg) PDC - 1421 ( 3 0mg/kg) PDC - 1421 ( 1 00mg/kg) PDC - 1421 ( 3 00 mg/kg) I m i p r a m i n e ( 3 mg/kg) * The inhibition by ≧ 50% is considered as significant response. Imipramine served as a positive control. IC 50 ( μ g / m L) A s sa y N am e 1.6 Norepinephrine Transporter 76.4 Dop a m i ne Tr a nspo r t er > 300 Se r o t on i n Tr a nspo r t er DA T : Do p a m i ne tr a n s p or ter SE R T: Ser o t o n i n tr a n s p or ter N E T: N or ep i ne p hr i ne tr a n s p or ter PDC - 1421 is a norepinephrine reuptake inhibitor, demonstrating a significant response at 100mg/kg and 300mg/kg doses in a preclinical depression animal model The unique properties and known Mechanism of Action (MOA) of PDC - 1421 enable us to expand to different indications, such as MDD, ADHD, and depression in cancer patients % i nh ib i t i on i n b ody tem p er a tu r e decreased PD C - 1 4 2 1 1 0 u g / ml Outcomes cannot be guaranteed; Past results are not guarantees of future results
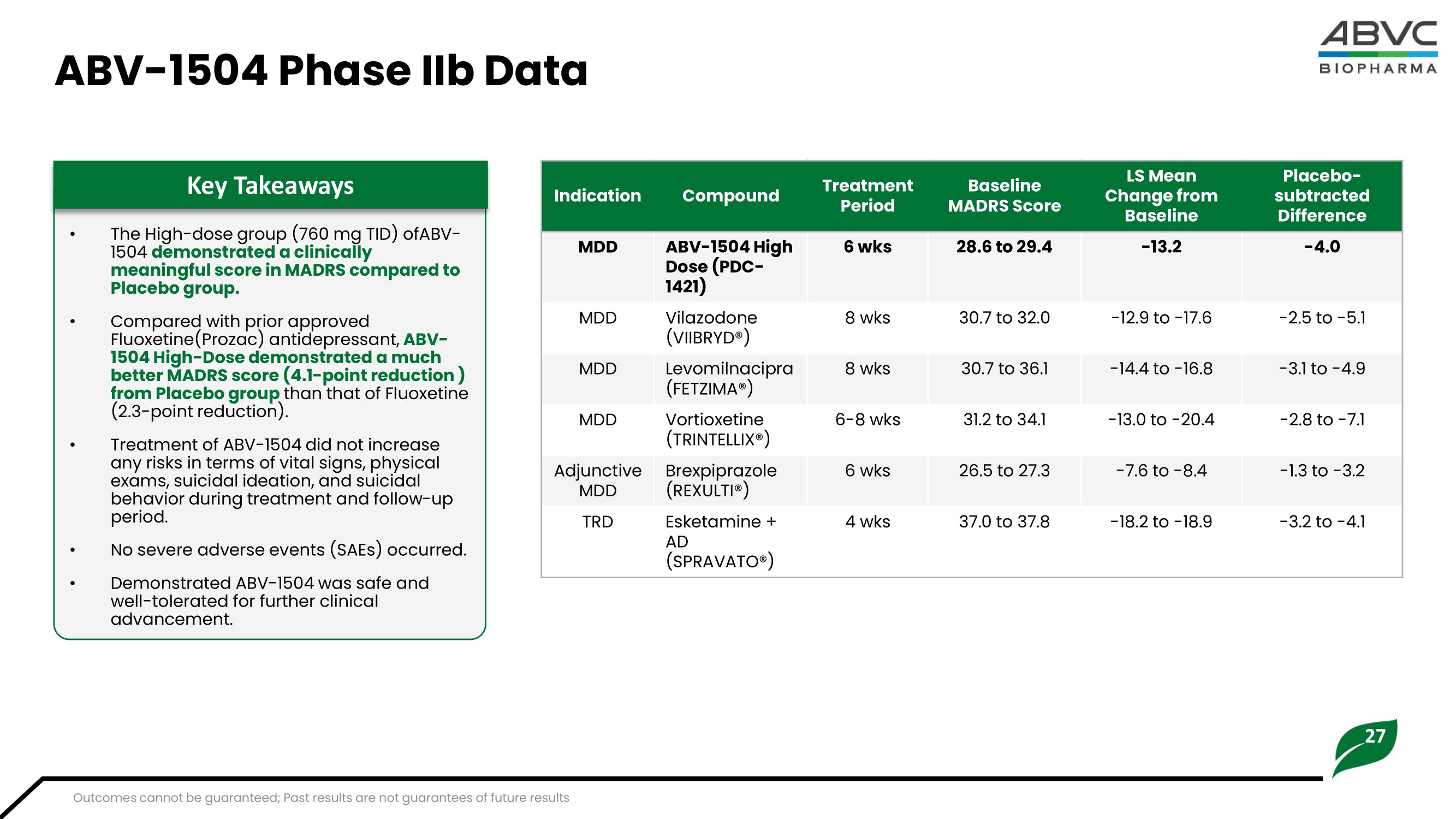
27 ABV - 1504 Ph a se IIb D a ta • T h e H i g h - dose g r oup (7 6 0 mg T I D) ofA B V - 1 50 4 d e m on s t r a ted a cli n ic a l l y me anin g ful s c or e in M AD RS c o m p a r e d t o Placebo group. • Compared with prior approved Fluoxetine(Prozac) antidepressant, ABV - 1 5 04 H i gh - Do s e de m o n s t r a t e d a m uc h bet t e r MAD RS s c or e (4. 1 - p o int r ed uc t ion ) from Placebo group than that of Fluoxetine (2 . 3 - poi nt r educ t i on) . • Trea t m en t o f AB V - 1 504 did not i nc r ease any risks in terms of vital signs, physical exa m s, su i c i dal i dea t i o n , an d su i c i dal beh av i o r du r i n g t r ea t m en t an d fol l o w - up period. • No severe adverse events (SAEs) occurred. • De m o n s t r a t ed AB V - 1 504 w as sa f e and wel l - t ol e r a t ed for fu r t her c l i ni cal advancement. Key Takeaways Placebo - s u b tr a c t ed Difference LS M e a n Ch a n g e f r om Baseline Baseline M ADRS Sco r e Treatment Period Compound Indication - 4.0 - 13.2 28.6 t o 29.4 6 wks A B V - 1504 H i g h D o s e ( P DC - 1421) MDD - 2 . 5 t o - 5.1 - 1 2 . 9 t o - 1 7. 6 3 0.7 t o 3 2 . 0 8 wk s Vilazodone (VIIBRYD®) MDD - 3 . 1 t o - 4.9 - 1 4.4 t o - 16 . 8 30 . 7 t o 3 6 . 1 8 wks Le v omilna c i p r a (FETZIMA®) MDD - 2.8 t o - 7 . 1 - 1 3 . 0 t o - 20 . 4 3 1 . 2 t o 34 . 1 6 - 8 wks Vortioxetine (TRINTELL I X ®) MDD - 1 . 3 t o - 3.2 - 7. 6 t o - 8 . 4 2 6 . 5 t o 2 7. 3 6 wk s Brexpiprazole (REXULTI®) Adjunctive MDD - 3 . 2 t o - 4.1 - 1 8 . 2 t o - 1 8 . 9 3 7 . 0 t o 3 7 . 8 4 wks Es k e t amine + AD (SPRAVATO®) TRD Outcomes cannot be guaranteed; Past results are not guarantees of future results