Table of Contents
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
AMENDMENT NO. 1
| Delaware | 2834 | 77-0602661 | ||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
790 Memorial Drive
John M. Maraganore, Ph.D.
Copies to:
| Steven D. Singer, Esq. Peter N. Handrinos, Esq. Hale and Dorr LLP 60 State Street Boston, Massachusetts 02109 (617) 526-6000 | Danielle Carbone, Esq. Shearman & Sterling LLP 599 Lexington Avenue New York, New York 10022 (212) 848-4000 |
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date hereof.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act, check the following box. o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If delivery of the Prospectus is expected to be made pursuant to Rule 434, please check the following box. o
The Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the Registration Statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
Table of Contents
| The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted. |
Prospectus SUBJECT TO COMPLETION, DATED APRIL 6, 2004
Shares

Common Stock
Alnylam Pharmaceuticals, Inc. is offering shares of common stock. This is our initial public offering, and no public market currently exists for our shares. We anticipate that the initial public offering price will be between $ and $ per share. After the offering, the market price for our shares may be outside this range.
We have applied to list our common stock on the NASDAQ National Market under the symbol “ALNY.”
Investing in our common stock involves a high degree of risk. See “Risk Factors” beginning on page 11.
| Per Share | Total | |||||||
| Offering price | $ | $ | ||||||
| Discounts and commissions to underwriters | $ | $ | ||||||
| Offering proceeds to Alnylam, before expenses | $ | $ | ||||||
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is accurate or complete. Any representation to the contrary is a criminal offense.
We have granted the underwriters the right to purchase up to additional shares of common stock to cover any over-allotments. The underwriters can exercise this right at any time within 30 days after the offering. The underwriters expect to deliver the shares of common stock to investors on or about , 2004.
Citigroup
| Piper Jaffray |
| ThinkEquity Partners |
, 2004
You should rely only on the information contained in this prospectus. We have not, and the underwriters have not, authorized anyone to provide you with different information. We are not making an offer to sell these securities in any jurisdiction where the offer or sale is not permitted. You should assume that the information contained in this prospectus is accurate as of the date on the front of this prospectus only. Our business, financial condition, results of operations and prospects may have changed since that date.
Unless otherwise stated, all references to “Alnylam,” “we,” “us,” “our,” the “Company” and similar designations refer to Alnylam Pharmaceuticals, Inc. and its subsidiaries. Alnylam is a registered trademark, Direct RNAi, Systemic RNAi and InterfeRxare trademarks, and Alnylam’s logo is a service mark, of Alnylam. Other trademarks and service marks appearing in this prospectus are the property of their respective holders.
TABLE OF CONTENTS
i
Table of Contents
SUMMARY
This summary highlights information contained elsewhere in this prospectus that we consider important. This summary does not contain all of the information you should consider before buying shares of our common stock. You should read the entire prospectus carefully, including the “Risk Factors” section and our consolidated financial statements and the related notes included in this prospectus, before making an investment in our common stock.
OUR BUSINESS
Overview
Alnylam is a biopharmaceutical company seeking to develop and commercialize new drugs that work through a recently discovered system in cells known as RNA interference, or RNAi. We believe that drugs that work through RNA interference, or RNAi therapeutics, have the potential to become a major class of drugs, like small molecule, protein and antibody drugs. Using our intellectual property and the expertise we have built in RNAi, we are developing a set of biological and chemical methods and know how that we expect to apply in a systematic way to develop RNAi therapeutics for a variety of diseases. We refer to these methods and their systematic application as our “product engine”. Using our current capabilities, we have initiated programs to develop RNAi therapeutics that will be administered directly to diseased parts of the body, which we refer to as Direct RNAi therapeutics. We believe there are multiple opportunities for Direct RNAi therapeutics. Our current Direct RNAi programs are focused on an eye disease known as age-related macular degeneration, or AMD, and on a central nervous system disorder known as Parkinson’s disease, or PD. We expect to initiate a clinical trial for our lead AMD product candidate in 2005. We are also working to extend our capabilities to enable the development of RNAi therapeutics that travel through the blood stream to reach diseased parts of the body, which we refer to as Systemic RNAi therapeutics. We believe Systemic RNAi will be used to treat a broad range of diseases.
RNAi is a recently discovered natural mechanism for selectively silencing genes. Genes provide cells with coded instructions for making proteins, and silencing a gene refers to stopping or reducing production of the protein specified, or encoded, by that gene. Our goal is to develop new drugs that use the RNAi mechanism to selectively silence genes encoding proteins that play harmful roles in disease. We intend to develop drugs based on a type of molecule known as small interfering RNA, or siRNA. siRNAs are the molecules within cells that directly trigger RNAi. We expect that our RNAi therapeutics will consist of chemically modified siRNAs designed to silence specific genes. Given the recent availability of the base sequence of the entire human genome, RNAi therapeutics can be designed, in theory, to silence any gene that encodes a protein involved in disease, even if currently this protein cannot be adequately controlled by conventional drugs.
The scientific evidence to support the feasibility of developing drugs based on RNAi technology is both preliminary and limited. siRNAs do not naturally possess many of the properties required of drugs, such as the ability to survive in the blood stream and penetrate into diseased tissues. The methods we are developing for our product engine need to introduce these drug-like properties into siRNAs without making these siRNAs unsafe or ineffective. We do not currently know whether we will be successful in developing these methods. Very few drug candidates based on RNAi technology have been tested in animals, and none has been tested in humans.
We believe that we have a strong intellectual property position relating to the development and commercialization of siRNAs as therapeutics, consisting of:
| • | a concentration of intellectual property rights claiming fundamental features of siRNAs and their use as therapeutics, which includes our ownership of, or exclusive rights to, several issued patents and pending patent applications; | |
| • | a broad portfolio of intellectual property relating to chemical modifications of siRNAs, including over 150 patents licensed from Isis Pharmaceuticals, Inc.; and | |
3
Table of Contents
| • | a number of pending patent applications claiming siRNAs directed to specific targets as treatments for particular diseases. |
We have filed or licensed over 200 patents and patent applications in the RNAi field. Nevertheless, we may need to acquire additional intellectual property rights in order to develop our products. In addition, our intellectual property rights may not be sufficient to prevent other companies from developing products that compete with any products we may develop.
Our goal is to develop and commercialize RNAi therapeutics. To access the substantial funding and expertise required to develop and commercialize RNAi therapeutics, we intend to form strategic collaborations with pharmaceutical companies. In the collaborations we form in the near term, we expect to take the lead role in discovery and early preclinical development of specific RNAi therapeutics, and to share responsibilities with our collaborators in later-stage development and commercialization of these RNAi therapeutics. We expect that our collaborators will provide us with significant funding for the work we perform, access to their development and commercial capabilities, and a share of revenues from products we originate. Our first such alliance is with Merck & Co., Inc. Over time, as we expand our capabilities and resources, we expect the nature of the collaborations we form will evolve, so that we take on progressively more responsibility for development and commercialization of products we originate, and retain a greater share of the revenues these products generate. In the longer term, we expect to develop and commercialize RNAi therapeutics independently.
Our ability to form appropriate collaborations with pharmaceutical companies will be crucial for our success. The development and commercialization of drugs often takes a decade or more to complete and requires significant expenditures. It is unlikely that we will be able to afford the time or expense involved in the development and commercialization of drugs unless we secure suitable collaborations with pharmaceutical companies. If we are unable to form or to maintain such collaborations, or our collaborators do not perform as we expect them to, we may not be able to develop and commercialize RNAi therapeutics. Moreover, the pharmaceutical marketplace is highly competitive, with hundreds of companies pursuing the same or very similar product opportunities. Even if we do succeed in developing RNAi therapeutics, we may not be able to compete effectively in the marketplace.
Our scientific founders were among the first scientists to publish significant discoveries in the field of RNAi. In addition, we have assembled an experienced management team to implement our business plan. Members of our management team have led discovery, development and commercialization programs for a number of marketed drugs, including Aranesp, Neulasta, Angiomax and Velcade.
Potential for RNAi Therapeutics
Proteins perform many of the vital functions of the cell and of the human body. Although the roles they play are generally beneficial, in certain circumstances, proteins can be harmful. Many human diseases are caused by the inappropriate behavior of proteins. A particular protein may, for example, be present in too great a quantity, be too active, or appear in the wrong place or at the wrong time. In these circumstances, the ability to stop or reduce production of the protein by selectively silencing the gene that directs its synthesis could be very beneficial in the treatment of the disease.
Beginning in 1999, our scientific founders were the first to describe and provide evidence that the RNAi mechanism occurs in mammalian cells and that its immediate trigger is a type of molecule known as small interfering RNA, or siRNA. They showed that laboratory-synthesized siRNAs could be introduced into the cell and suppress production of specific target proteins. Because it is possible, in theory, to design and synthesize siRNAs specific for any gene of interest, we believe that RNAi therapeutics have the potential to become a broad new class of drugs. We believe that RNAi therapeutics could offer the following benefits:
| • | Ability to treat a broad range of diseases. Given the availability of the base sequence of the entire human genome, in theory, it should be possible to design siRNAs to suppress the production of virtually any human protein whose presence or activity causes disease. |
4
Table of Contents
| • | Ability to target proteins that cannot be targeted effectively by existing drug classes. Many proteins that play important roles in disease cannot be targeted effectively with small molecules and therapeutic proteins, including monoclonal antibodies. In theory, siRNAs should not face the same limitations as these drug classes and, therefore, we believe RNAi therapeutics will be able to target proteins that small molecule and protein drugs cannot currently target. | |
| • | An inherently potent mechanism of action. Each siRNA molecule can trigger destruction of multiple messenger RNA molecules, each of which could otherwise direct the synthesis of many protein molecules. As a result, one siRNA molecule may effectively be able to block the activity of many protein molecules. This inherent potency of the RNAi mechanism suggests a potentially high degree of potency for RNAi therapeutics. | |
| • | Simplified discovery of drug candidates. Identification of siRNA drug candidates has the potential to be much simpler and take considerably less time than other drug classes because, in theory, it will involve a relatively standard process that can be applied in a similar fashion to many successive product candidates. |
None of these potential benefits has yet been proven. If we are unable to design siRNAs with sufficient stability and the ability to enter tissues and cells, we will not be able to develop safe and effective RNAi therapeutics.
Our Business Strategy
Our strategy is to use our strong intellectual property position and our expertise in RNAi to develop and commercialize RNAi therapeutics. The key elements of our business strategy are as follows:
| • | Pursue product opportunities in a phased approach based on the evolving capabilities of our product engine. We are implementing a phased approach to product development that we believe will allow us to initiate product development activities in three main phases: |
| — | Direct RNAi Therapeutics: We intend to utilize the current capabilities of our product engine by focusing our efforts on developing RNAi therapeutics that can be administered directly at diseased parts of the body, such as the eye or the brain. As part of this phase, we have initiated Direct RNAi programs focused on AMD and PD. | |
| — | Systemic RNAi Therapeutics for liver-based diseases: As we extend the capabilities of our product engine, we intend to develop RNAi therapeutics for liver-based diseases because it appears, on the basis of early preclinical evidence, that the liver takes up siRNAs more readily than other tissues. | |
| — | Systemic RNAi Therapeutics for other diseases: As a third phase of our product development strategy, we will seek to develop RNAi therapeutics that exert their effects in tissues other than the liver. We believe achievement of this objective could permit us to develop Systemic RNAi drugs for a broad range of diseases, such as cancer and autoimmune diseases. |
| • | Maintain our strong intellectual property position in the RNAi field. We believe we have a strong intellectual property position relating to the development and commercialization of siRNAs as therapeutics. To build upon our existing intellectual property position, we are focusing on patents and patent applications covering fundamental aspects of siRNAs, chemical modifications to siRNAs and specific targets for RNAi therapeutics. | |
| • | Capitalize on our expertise in RNAi and our intellectual property position to gain access to additional resources to develop and commercialize RNAi therapeutics. We believe that we can use our expertise and the strength of our intellectual property to drive the formation of strategic alliances that will provide us with funding and access to important additional resources. We intend to take an active role in these alliances, including maintaining certain development and commercialization rights. We also intend to use our early alliances to expand our own capabilities so that in the future we will be able to develop and commercialize our therapeutic products independently. | |
5
Table of Contents
| • | Leverage our intellectual property position by licensing our technology to generate revenues. We intend to generate revenues by granting licenses to our intellectual property to third parties for the development of therapeutics outside our areas of focus and for the development of research reagents and services. |
Our Product Engine
To realize the potential of RNAi therapeutics as a broad new class of drugs, we are developing a set of biological and chemical methods and know how that we expect to apply in a systematic way to develop RNAi therapeutics for a variety of diseases. We refer to these methods and their systematic application as our product engine. We believe that our product engine will provide a systematic approach for identifying siRNA drug candidates, using the following steps:
| • | Sequence selection — the use of computational tools to design siRNA sequences likely to be selective for the gene that is to be silenced, | |
| • | Potency selection —the synthesis and comparison of different siRNAs to determine which are most potent in silencing the target gene, | |
| • | Stabilization — the introduction of chemical modifications into potent siRNAs to make them more stable within the body, and | |
| • | Improvement of biodistribution — the addition of further chemical groups to stabilized siRNAs to improve their ability to reach different parts of the body. | |
Our Development Programs
Using the current capabilities of our product engine, we have initiated two programs to identify specific siRNAs for potential further development as Direct RNAi drug candidates. We expect to initiate additional programs as the capabilities of our product engine evolve. Our current programs are focused on AMD and PD.
Age-Related Macular Degeneration
AMD can cause severe deterioration of vision and may ultimately cause blindness. The National Eye Institute estimates that over 1.6 million adults over 50 in the United States suffer from advanced AMD. The siRNAs we are exploring would treat wet AMD, a subtype of AMD often associated with severe vision loss. AMD Alliance International estimates that approximately 200,000 new cases of wet AMD are diagnosed in North America each year and 500,000 new cases of wet AMD are diagnosed worldwide each year. We are developing a RNAi therapeutic intended to treat wet AMD by suppressing production of the protein known as vascular endothelial growth factor, or VEGF. VEGF is believed to play a key role in wet AMD by stimulating the growth and leakage of blood vessels that disrupt the retina. We are currently evaluating several siRNAs designed to inhibit the production of VEGF in animal models and expect to begin a clinical trial for an AMD product candidate in 2005. Any siRNA we develop for AMD will be a Direct RNAi drug administered by injection into the eye, the mode of administration used for two drug candidates currently in late stage clinical trials by other companies. We have filed patent applications relating to the use of siRNA to suppress VEGF production for therapeutic purposes.
Parkinson’s Disease
PD is a disorder of the nervous system that the American Parkinson Disease Association estimates afflicts more than 1.5 million people in the United States, and the World Health Organization estimates afflicts approximately four million people worldwide. Current treatments for PD focus on treating the symptoms of the disease, which are caused by the shortage of a substance known as dopamine in the brain. There are no drugs currently approved to treat the cause of the disease. The siRNAs we have started to explore as potential treatments for PD are based on treating the suspected cause of PD rather than the
6
Table of Contents
Early Stage Company
We are an early stage company and the approach we are taking to discover and develop drugs is novel and unproven. Our potential product candidates are in early stages of preclinical development where failure is common. Neither we nor any other company has received regulatory approval to market therapeutics utilizing siRNAs. We do not expect any of our product candidates, if successfully developed, to receive regulatory approval for commercial sale for at least several years. See “Risk Factors” beginning on page 11 for risks related to an investment in our common stock.
Corporate Information
Alnylam Pharmaceuticals, Inc. was incorporated in Delaware in May 2003. Alnylam Europe, AG, which was incorporated in Germany in June 2000 under the name Ribopharma AG, and Alnylam U.S., Inc., which was incorporated in Delaware in June 2002, are wholly owned subsidiaries of Alnylam Pharmaceuticals, Inc. Alnylam Pharmaceuticals, Inc. acquired Alnylam Europe, AG in July 2003. Our principal executive office is located at 790 Memorial Drive, Cambridge, Massachusetts 02139, and our telephone number is (617) 252-0700. Our internet address is www.alnylam.com. The information on our web site is not incorporated by reference into this prospectus and should not be considered to be a part of this prospectus. Our web site address is included in this prospectus as an inactive technical reference only.
7
Table of Contents
THE OFFERING
| Common stock offered | shares | |
| Common stock to be outstanding after this offering | shares | |
| Use of proceeds | We expect to use the net proceeds of this offering to fund research and development activities, to fund the acquisition, licensing and protection of intellectual property rights, and for general corporate purposes. See “Use of Proceeds” for more information. | |
| Proposed NASDAQ National Market symbol | ALNY | |
| Risk factors | See “Risk Factors” and the other information included in this prospectus for a discussion of factors you should carefully consider before deciding to invest in shares of our common stock. |
The number of shares of common stock to be outstanding after this offering is based on the number of shares outstanding as of March 31, 2004 and excludes:
| • | 3,691,618 shares of common stock issuable upon the exercise of outstanding stock options as of March 31, 2004 with a weighted average exercise price of $0.32 per share; | |
| • | 125,000 shares of common stock issuable upon the exercise of outstanding warrants as of March 31, 2004 with a weighted average exercise price of $4.50 per share; and | |
| • | an aggregate of 3,946,601 shares of common stock reserved for issuance under our stock option plans and stock purchase plan. | |
Except as otherwise noted, all information in this prospectus:
| • | assumes no exercise by the underwriters of their over-allotment option to purchase additional shares of common stock in the offering; | |
| • | gives effect to the conversion of all outstanding shares of convertible preferred stock into shares of common stock upon the closing of the offering; and | |
| • | gives effect to the filing of our amended and restated certificate of incorporation and the adoption of our amended and restated bylaws upon the closing of the offering. |
8
Table of Contents
SUMMARY CONDENSED CONSOLIDATED FINANCIAL DATA
You should read the following summary financial data in conjunction with “Selected Condensed Consolidated Financial Data”, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and related notes, all included elsewhere in this prospectus.
Pro forma basic and diluted net loss per common share have been calculated assuming the conversion of all shares of convertible preferred stock outstanding at December 31, 2003 into shares of common stock, which does not include 1,666,667 shares of common stock issuable upon conversion of the convertible preferred stock issued to Isis Pharmaceuticals on March 11, 2004. On July 31, 2003, we acquired all of the outstanding voting shares of Alnylam Europe for $1.5 million in cash and 1,549,216 shares of our common stock, and assumed $7.1 million of debt. In connection with the acquisition, we allocated $4.6 million of the purchase price to purchased in-process research and development, which we expensed immediately. The results of Alnylam Europe have been included in our consolidated results from the date of acquisition.
| Period | |||||||||
| from Inception | |||||||||
| (June 14, 2002) | |||||||||
| through | Year Ended | ||||||||
| December 31, 2002 | December 31, 2003 | ||||||||
| ($ in thousands, except per share data) | |||||||||
Consolidated Statements of Operations Data: | |||||||||
| Revenue from research collaborators | $ | — | $ | 176 | |||||
| Costs and expenses | |||||||||
| Research and development(1) | 3,342 | 13,097 | |||||||
| General and administrative(1) | 880 | 7,527 | |||||||
| Purchased in-process research and development | — | 4,609 | |||||||
| Total operating costs and expenses | 4,222 | 25,233 | |||||||
| Loss from operations | (4,222 | ) | (25,057 | ) | |||||
| Other income, net | 86 | 24 | |||||||
| Net loss | (4,136 | ) | (25,033 | ) | |||||
| Accretion of redeemable convertible preferred stock | (748 | ) | (2,906 | ) | |||||
| Net loss attributable to common stockholders | (4,884 | ) | (27,939 | ) | |||||
| Net loss per common share (basic and diluted) | $ | (7.76 | ) | $ | (15.60 | ) | |||
| Weighted average shares used to compute basic and diluted net loss per common share | 629,548 | 1,791,063 | |||||||
| Pro forma net loss per common share (basic and diluted) | $ | (1.65 | ) | ||||||
| Shares used to compute basic and diluted pro forma net loss per common share | 15,157,601 | ||||||||
| (1) | Noncash stock-based compensation expense included in these amounts are as follows: |
| Research and development | $ | 172 | $ | 2,832 | ||||
| General and administrative | — | 623 | ||||||
| Total noncash stock-based compensation | $ | 172 | $ | 3,455 | ||||
9
Table of Contents
The pro forma as adjusted balance sheet data as of December 31, 2003 gives effect to the issuance of 1,666,667 shares of convertible preferred stock to Isis Pharmaceuticals on March 11, 2004, the conversion of all shares of convertible preferred stock into shares of common stock and the sale of shares of common stock offered by this prospectus at an assumed initial public offering price of $ per share, the mid-point of the estimated price range shown on the cover page of this prospectus, after deducting estimated underwriting discounts and offering expenses.
| As of December 31, 2003 | ||||||||
| Pro Forma | ||||||||
| Actual | As Adjusted | |||||||
| ($ in thousands) | ||||||||
Consolidated Balance Sheet Data: | ||||||||
| Cash and cash equivalents | $ | 23,193 | ||||||
| Working capital | 20,345 | |||||||
| Total assets | 35,183 | |||||||
| Note payable | (1,859 | ) | ||||||
| Redeemable convertible preferred stock | 55,189 | |||||||
| Total stockholders’ equity (deficit) | (26,707 | ) | ||||||
10
Table of Contents
RISK FACTORS
An investment in our common stock involves a high degree of risk. In deciding whether to invest, you should carefully consider the following risk factors, as well as the other information contained in this prospectus, including our consolidated financial statements and the related notes. Any of the following risks as well as other risks and uncertainties discussed in this prospectus could have a material adverse effect on our business, financial condition, results of operations and prospects and cause the value of our stock to decline, which could cause you to lose all or part of your investment.
Risks Related to Our Business
Risks Related to Being an Early Stage Company
Because we have a short operating history, there is a limited amount of information about us upon which you can evaluate our business and prospects.
Our operations began in June 2002 and we have only a limited operating history upon which you can evaluate our business and prospects. In addition, as an early stage company, we have limited experience and have not yet demonstrated an ability to successfully overcome many of the risks and uncertainties frequently encountered by companies in new and rapidly evolving fields, particularly in the biopharmaceutical area. For example, to execute our business plan, we will need to successfully:
| • | execute product development activities using an unproven technology; | |
| • | build and maintain a strong intellectual property portfolio; | |
| • | gain acceptance for the development and commercialization of our products; | |
| • | develop and maintain successful strategic relationships; and | |
| • | manage our spending as costs and expenses increase due to clinical trials, regulatory approvals and commercialization. | |
If we are unsuccessful in accomplishing these objectives, we may not be able to develop product candidates, raise capital, expand our business or continue our operations.
The approach we are taking to discover and develop novel drugs is unproven and may never lead to marketable products.
We have concentrated our efforts and therapeutic product research on RNAi technology, and our future success depends on the successful development of this technology and products based on RNAi technology. Neither we nor any other company has received regulatory approval to market therapeutics utilizing siRNAs. The scientific discoveries that form the basis for our efforts to discover and develop new drugs are relatively new. The scientific evidence to support the feasibility of developing drugs based on these discoveries is both preliminary and limited. Very few drug candidates based on these discoveries have ever been tested in animals, and none has been tested in humans. siRNAs, the class of molecule we are trying to develop into drugs, do not naturally possess the inherent properties typically required of drugs, such as the ability to be stable in the body long enough to reach the tissues in which their effects are required, nor the ability to enter cells within these tissues in order to exert their effects. We currently have only limited data to suggest that we can introduce these properties into siRNAs. We may spend large amounts of money trying to introduce these properties, and never succeed in doing so. In addition, these compounds may not demonstrate in patients the chemical and pharmacological properties ascribed to them in laboratory studies, and they may interact with human biological systems in unforeseen, ineffective or harmful ways. As a result, we may never succeed in developing a marketable product. If we do not successfully develop and commercialize drugs based upon our technological approach, we will not become profitable and the value of our common stock will decline.
Further, our focus solely on RNAi technology for developing drugs as opposed to multiple, more proven technologies for drug development increases the risks associated with the ownership of our common stock. If
11
Table of Contents
Risks Related to Our Financial Results and Need for Financing
We have a history of losses and may never be profitable.
We have experienced significant operating losses since our inception. For the year ended December 31, 2003, we had net losses of $25.0 million. As of December 31, 2003, we had a deficit accumulated in the development stage of $29.5 million. To date, we have not developed any products nor generated any revenues from the sale of products. Further, we do not expect to generate any such revenues in the foreseeable future. We expect our annual operating losses to increase over the next several years as we expand our efforts to discover, develop and commercialize RNAi therapeutics. We anticipate that the majority of any revenue we generate over the next several years will be from collaborations with pharmaceutical companies, but cannot be certain that we will be able to secure these collaborations or to meet the obligations or achieve any milestones that we may be required to meet or achieve to receive payments. To date, our collaboration and license agreements have provided us with minimal revenue. If we are unable to secure revenue from collaborations, we may be unable to continue our efforts to discover, develop and commercialize RNAi therapeutics without raising financing from other sources.
To become and remain profitable, we must succeed in developing and commercializing novel drugs with significant market potential. This will require us to be successful in a range of challenging activities that we have yet to initiate, including preclinical testing and clinical trial stages of development, obtaining regulatory approval for these novel drugs, and manufacturing, marketing and selling them. We may never succeed in these activities, and may never generate revenues that are significant enough to achieve profitability. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. If we cannot become and remain profitable, the market price of our common stock could decline. In addition, we may be unable to raise capital, expand our business, diversify our product offerings or continue our operations.
We will require substantial additional funds to complete our research and development activities and if additional funds are not available we may need to critically limit, significantly scale back or cease our operations.
We have used substantial funds to develop our RNAi technologies and will require substantial funds to conduct further research and development, including preclinical testing and clinical trials of any product candidates, and to manufacture and market any products that are approved for commercial sale. Because the successful development of our products is uncertain, we are unable to estimate the actual funds we will require to develop and commercialize them. We believe that the net proceeds from this offering and our existing resources will be sufficient to support our current operating plan until at least the end of 2005, during which time we expect to extend the capabilities of our product engine, initiate development of a product for the treatment of wet AMD and continue to prosecute patent applications and otherwise build and maintain our intellectual property portfolio.
However, our future capital requirements and the period for which we expect the net proceeds from this offering and our existing resources to support our operations may vary from what we expect. We have based our expectations on a number of factors, many of which are difficult to predict or are outside of our control, including:
| • | our progress in demonstrating that siRNAs can be active as drugs; | |
| • | our ability to develop relatively standard procedures for selecting and modifying siRNA drug candidates; | |
| • | progress in our research and development programs, as well as the magnitude of these programs; | |
12
Table of Contents
| • | the timing, receipt, and amount of milestone and other payments, if any, from present and future collaborators, if any; | |
| • | our ability to establish and maintain additional collaborative arrangements; | |
| • | the resources, time and costs required to initiate and complete our preclinical and clinical trials, obtain regulatory approvals, protect our intellectual property and obtain and maintain licenses to third-party intellectual property; and | |
| • | the timing, receipt and amount of sales and royalties, if any, from our potential products. | |
If our estimates and predictions relating to these factors are incorrect, we may need to modify our operating plan.
We will be required to seek additional funding in the future and intend to do so through collaborative arrangements and public or private equity offerings and debt financings. Additional funds may not be available to us on acceptable terms or at all. In addition, the terms of any financing may adversely affect the holdings or the rights of our stockholders. For example, if we raise additional funds by issuing equity securities, further dilution to our then-existing stockholders will result. Debt financing, if available, may involve restrictive covenants that could limit our flexibility in conducting future business activities. If we are unable to obtain funding on a timely basis, we may be required to significantly curtail one or more of our research or development programs. We also could be required to seek funds through arrangements with collaborators or others that may require us to relinquish rights to some of our technologies, product candidates or products that we would otherwise pursue on our own.
Risks Related to Our Dependence on Third Parties
| We may not be able to execute our business strategy if we are unable to enter into alliances with other companies that can provide capabilities and funds for the development and commercialization of our drug candidates. If we are unsuccessful in forming or maintaining these alliances on favorable terms, our business may not succeed. |
We do not have any capability for sales, marketing or distribution and have limited capabilities for drug development. Accordingly, we must enter into alliances with other companies that can provide such capabilities. For example, we may enter into alliances with major pharmaceutical companies to jointly develop specific drug candidates and to jointly commercialize them if they are approved. In such alliances, we would expect our pharmaceutical collaborators to provide substantial capabilities in clinical development, regulatory affairs, marketing and sales. We may not be successful in entering into any such alliances on favorable terms. Even if we do succeed in securing such alliances, we may not be able to maintain them if, for example, development or approval of a drug candidate is delayed or sales of an approved drug are disappointing. Furthermore, any delay in entering into collaboration agreements could delay the development and commercialization of our drug candidates and reduce their competitiveness even if they reach the market. Any such delay related to our collaborations could adversely affect our business.
In addition, we expect that we will need to enter into alliances with other companies to provide substantial additional cash for development and potential commercialization of our drug candidates. We have entered into a collaboration agreement with Merck under which Merck may elect to pay a portion of the costs to develop and market certain drug candidates that we may initially develop based on information and materials provided by Merck. Merck is under no obligation to pay any of the development and commercialization costs for any of these drug candidates, and they may elect not to do so. For drug candidates from our Merck collaboration that Merck does not elect to fund, and for drug candidates we may develop outside of this collaboration, we expect to seek additional collaborations with other pharmaceutical companies to fund all or part of the costs of drug development and commercialization. We may not be able to enter into such collaborations, and the terms of any collaboration agreement we do secure may not be favorable to us. If we are not successful in our efforts to enter into a collaboration arrangement with respect to a particular drug candidate, we may not have sufficient funds to develop this or any other drug candidate internally, or to bring any drug candidates to market. If we do not have sufficient funds to develop and bring
13
Table of Contents
If any collaborator terminates or fails to perform its obligations under agreements with us, the development and commercialization of our drug candidates could be delayed or terminated.
Our expected dependence on collaborators for capabilities and funding means that our business would be adversely affected if any collaborator terminates its collaboration agreement with us or fails to perform its obligations under that agreement. Our current or future collaborations, if any, may not be scientifically or commercially successful. Disputes may arise in the future with respect to the ownership of rights to technology or products developed with collaborators, which could have an adverse effect on our ability to develop and commercialize any affected product candidate.
Our current collaborations allow, and we expect that any future collaborations will allow, either party to terminate the collaboration for a material breach by the other party. If a collaborator terminates its collaboration with us, for breach or otherwise, it would be difficult for us to attract new collaborators and could adversely affect how we are perceived in the business and financial communities. In addition, a collaborator could determine that it is in its financial interest to:
| • | pursue alternative technologies or develop alternative products, either on its own or jointly with others, that may be competitive with the products on which it is collaborating with us or which could affect its commitment to the collaboration with us; | |
| • | pursue higher-priority programs or change the focus of their development programs, which could affect the collaborator’s commitment to us; or | |
| • | if it has marketing rights, choose to devote fewer resources to the marketing of our product candidates, if any are approved for marketing, than they do for product candidates of their own development. | |
If any of these occur, the development and commercialization of one or more drug candidates could be delayed, curtailed or terminated because we may not have sufficient financial resources or capabilities to continue such development and commercialization on our own.
In addition, we have entered into a research collaboration agreement with the Mayo Foundation for Medical Education and Research and the Mayo Clinic Jacksonville, which we refer to collectively as the Mayo Clinic, in connection with our PD program and we may enter into similar agreements in the future. Either party may terminate this research collaboration agreement upon a breach by the other party. The agreement provides us with an option to acquire an exclusive license to any intellectual property or inventions developed in connection with the collaboration. However, in order to secure any such license, we and the Mayo Clinic must agree on terms within 90 days of the exercise of the option. We may not be able to enter into any such license on reasonable terms, if at all.
We have no manufacturing experience or resources and we must incur significant costs to develop this expertise or rely on third parties to manufacture our products.
We have no manufacturing experience. In order to develop products, apply for regulatory approvals and commercialize our products, we will need to develop, contract for, or otherwise arrange for the necessary manufacturing capabilities. We expect to rely on others to manufacture the materials we will require for any clinical trials that we initiate. Only a limited number of manufacturers can supply synthetic RNAi, and we have not secured any long-term commercial supply arrangements. The manufacturing process for any products that we may develop is an element of the FDA approval process and we will need to contract with manufacturers who can meet the FDA requirements on an ongoing basis. In addition, if we receive the necessary regulatory approval for any product candidate, we also expect to rely on third parties, including our collaborators, to produce materials required for commercial production. We may experience difficulty in obtaining adequate manufacturing capacity for our needs. If we are unable to obtain or maintain contract manufacturing for these product candidates, or to do so on commercially reasonable terms, we may not be able to successfully develop and commercialize our products.
14
Table of Contents
To the extent that we enter into manufacturing arrangements with third parties, we will depend on these third parties to perform their obligations in a timely manner and consistent with regulatory requirements. The failure of a third-party manufacturer to perform its obligations as expected could adversely affect our business in a number of ways, including:
| • | we may not be able to initiate or continue clinical trials of products that are under development; | |
| • | we may be delayed in submitting applications for regulatory approvals for our products; | |
| • | we may lose the cooperation of our collaborators; | |
| • | we may be required to cease distribution or recall some or all batches of our products; and | |
| • | ultimately, we may not be able to meet commercial demands for our products. | |
If a third-party manufacturer with whom we contract fails to perform its obligations, we may be forced to manufacture the materials ourselves, for which we may not have the capabilities or resources, or enter into an agreement with a different third-party manufacturer, which we may not be able to do on reasonable terms, if at all. In addition, if we are required to change manufacturers for any reason, we will be required to verify that the new manufacturer maintains facilities and procedures that comply with quality standards and with all applicable regulations and guidelines. The delays associated with the verification of a new manufacturer could negatively affect our ability to develop product candidates in a timely manner or within budget. Furthermore, a manufacturer may possess technology related to the manufacture of our product candidate that such manufacturer owns independently. This would increase our reliance on such manufacturer or require us to obtain a license from such manufacturer in order to have another third party manufacture our products.
We have no sales, marketing or distribution experience and expect to depend significantly on third parties who may not successfully commercialize our products.
We have no sales, marketing or distribution experience. We expect to rely on third parties to launch and market certain of our product candidates, if approved. We may have limited or no control over the sales, marketing and distribution activities of these third parties. Our future revenues may depend heavily on the success of the efforts of these third parties.
To develop internal sales, distribution and marketing capabilities, we will have to invest significant amounts of financial and management resources. For products where we decide to perform sales, marketing and distribution functions ourselves, we could face a number of additional risks, including:
| • | we may not be able to attract and build a significant marketing or sales force; | |
| • | the cost of establishing a marketing or sales force may not be justifiable in light of the revenues generated by any particular product; and | |
| • | our direct sales and marketing efforts may not be successful. | |
Risks Related to Managing Our Operations
If we are unable to attract and retain qualified key management and scientists, staff consultants and advisors, our ability to implement our business plan may be adversely affected.
We are highly dependent upon our senior management and scientific staff. The loss of the service of any of the members of our senior management, including Dr. John Maraganore, our President and Chief Executive Officer, may significantly delay or prevent the achievement of product development and other business objectives. Our employment agreements with our key personnel are terminable without notice. We do not carry key man life insurance on any of our key employees.
Although we have generally been successful in our recruiting efforts, we face intense competition for qualified individuals from numerous pharmaceutical and biotechnology companies, universities, governmental entities and other research institutions. We may be unable to attract and retain suitably qualified individuals, and our failure to do so could have an adverse effect on our ability to implement our business plan.
15
Table of Contents
We may have difficulty managing our growth and expanding our operations successfully as we seek to evolve from a company primarily involved in discovery and preclinical testing into one that develops and commercializes drugs.
Since we commenced operations in 2002, we have grown rapidly to over 60 employees, with offices and laboratory space in both Cambridge, Massachusetts and Kulmbach, Germany. This rapid and substantial growth, and the geographical separation of our sites, has placed a strain on our administrative and operational infrastructure, and we anticipate that our continued growth will have a similar impact. If drug candidates we develop enter and advance through clinical trials, we will need to expand our development, regulatory, manufacturing, marketing and sales capabilities or contract with other organizations to provide these capabilities for us. As our operations expand, we expect that we will need to manage additional relationships with various collaborators, suppliers and other organizations. Our ability to manage our operations and growth will require us to continue to improve our operational, financial and management controls, reporting systems and procedures in at least two different countries. We may not be able to implement improvements to our management information and control systems in an efficient or timely manner and may discover deficiencies in existing systems and controls.
If we are unable to manage the challenges associated with our international operations, the growth of our business could be limited.
In addition to our operations in Cambridge, Massachusetts, we operate an office and laboratory in Kulmbach, Germany. We are subject to a number of risks and challenges that specifically relate to these international operations. Our international operations may not be successful if we are unable to meet and overcome these challenges, which could limit the growth of our business and may have an adverse effect on our business and operating results. These risks include:
| • | fluctuations in foreign currency exchange rates that may increase the U.S. dollar cost of our international operations; | |
| • | difficulty managing operations in multiple locations, which could adversely affect the progress of our product candidate development program and business prospects; | |
| • | local regulations that may restrict or impair our ability to conduct biotechnology-based research and development; | |
| • | foreign protectionist laws and business practices that favor local competition; and | |
| • | failure of local laws to provide the same degree of protection against infringement of our intellectual property, which could adversely affect our ability to develop product candidates or reduce future product or royalty revenues, if any, from product candidates we may develop. | |
Risks Related to Our Industry
Risks Related to Development, Clinical Testing and Regulatory Approval of Our Drug Candidates
Any drug candidates we develop may fail in development or be delayed so much that they do not become commercially viable.
Preclinical testing and clinical trials of new drug candidates are lengthy and expensive and the historical failure rate for drug candidates is high. We may not be able to advance any product candidates into clinical trials. Even if we do successfully enter into clinical studies, the results from preclinical testing of a drug candidate may not predict the results that will be obtained in human clinical trials. We, the FDA or other applicable regulatory authorities may suspend clinical trials of a drug candidate at any time if we or they believe the subjects or patients participating in such trials are being exposed to unacceptable health risks, or for other reasons. Among other reasons, adverse side effects of a drug candidate on subjects or patients in a clinical trial could result in the FDA or foreign regulatory authorities suspending or terminating the trial and refusing to approve a particular drug candidate for any or all indications of use.
16
Table of Contents
Clinical trials of a new drug candidate require the enrollment of a sufficient number of patients, including patients who are suffering from the disease the drug candidate is intended to treat and who meet other eligibility criteria. Rates of patient enrollment are affected by many factors, including the size of the patient population, the nature of the protocol, the proximity of patients to clinical sites, the availability of effective treatments for the relevant disease and the eligibility criteria for the clinical trial. Delays in patient enrollment can result in increased costs and longer development times.
Clinical trials also require the review and oversight of institutional review boards, referred to as IRBs, which approve and continually review clinical investigations and protect the rights and welfare of human subjects. Inability to obtain or delay in obtaining IRB approval can prevent or delay the initiation and completion of clinical trials, and the FDA may decide not to consider any data or information derived from a clinical investigation not subject to initial and continuing IRB review and approval in support of a marketing application.
Our drug candidates that we develop may encounter problems during clinical trials that will cause us or regulatory authorities to delay or suspend these trials, or that will delay the analysis of data from these trials. If we experience any such problems, we may not have the financial resources to continue development of the drug candidate that is affected, or development of any of our other drug candidates. We may also lose, or be unable to enter into, collaborative arrangements for the affected drug candidate and for other drug candidates we are developing.
Delays in clinical trials could reduce the commercial viability of our drug candidates. Any of the following could delay our clinical trials:
| • | discussions with the FDA or comparable foreign authorities regarding the scope or design of our clinical trials; | |
| • | problems in engaging IRBs to oversee trials or problems in obtaining IRB approval of studies; | |
| • | delays in enrolling patients and volunteers into clinical trials; | |
| • | high drop-out rates for patients and volunteers in clinical trials; | |
| • | negative results of clinical trials; | |
| • | inadequate supply or quality of drug candidate materials or other materials necessary for the conduct of our clinical trials; | |
| • | serious and unexpected drug-related side effects experienced by participants in our clinical trials; or | |
| • | unfavorable FDA inspection and review of a clinical trial site or records of any clinical or preclinical investigation. |
The FDA approval process may be delayed for any drugs we develop that require the use of specialized drug delivery devices.
Some drug candidates that we develop may need to be administered using specialized drug delivery devices. We believe that any product candidate we develop for PD will need to be administered using such a device. While we expect to rely on drug delivery systems that have been approved by the FDA or other regulatory agencies to deliver drugs like ours to similar physiological sites, we may need to modify the design or labeling of such delivery device for some products we may develop. In such an event, the FDA may regulate the product as a combination product or require additional approvals or clearances for the modified delivery device. Further, to the extent the specialized delivery device is owned by another company, we would need that company’s cooperation to implement the necessary changes to the device, or its labeling, and to obtain any additional approvals or clearances. Obtaining such additional approvals or clearances, and cooperation of such other companies, when necessary, could significantly delay and increase the cost of obtaining marketing approval, which could reduce the commercial viability of our drug candidate.
17
Table of Contents
We may be unable to obtain U.S. or foreign regulatory approval and, as a result, be unable to commercialize our drug candidates.
Our drug candidates are subject to extensive governmental regulations relating to development, clinical trials, manufacturing and commercialization. Rigorous preclinical testing and clinical trials and an extensive regulatory approval process are required to be successfully completed in the United States and in many foreign jurisdictions before a new drug can be sold. Satisfaction of these and other regulatory requirements is costly, time consuming, uncertain and subject to unanticipated delays. It is possible that none of the drug candidates we may develop will obtain the appropriate regulatory approvals necessary for us or our collaborators to begin selling them.
We have no experience in conducting and managing the clinical trials necessary to obtain regulatory approvals, including approval by the FDA. The time required to obtain FDA and other approvals is unpredictable but typically exceeds five years following the commencement of clinical trials, depending upon the complexity of the drug candidate. Any analysis we perform of data from preclinical and clinical activities is subject to confirmation and interpretation by regulatory authorities, which could delay, limit or prevent regulatory approval. We may also encounter unexpected delays or increased costs due to new government regulations, for example, from future legislation or administrative action, or from changes in FDA policy during the period of product development, clinical trials and FDA regulatory review.
Because the drugs we are intending to develop may represent a new class of drug, the FDA has not yet established any definitive policies, practices or guidelines in relation to these drugs. While we expect any AMD or PD product candidates we develop will be regulated as a new drug under the Federal Food, Drug, and Cosmetic Act, the FDA could decide to regulate them or other products we may develop as biologics under the Public Health Service Act. The lack of such policies, practices or guidelines may hinder or slow review by the FDA of any regulatory filings that we may submit. Moreover, the FDA may respond to these submissions by defining requirements we may not have anticipated. Such responses could lead to significant delays in the clinical development of our product candidates. In addition, because there are approved treatments for both AMD and PD, in order to receive regulatory approval, we will need to demonstrate through clinical trials that the product candidates we develop to treat these diseases, if any, are not only safe and effective, but safer or more effective than existing products.
Any delay or failure in obtaining required approvals could have a material adverse effect on our ability to generate revenues from the particular drug candidate. Furthermore, any regulatory approval to market a product may be subject to limitations on the indicated uses for which we may market the product. These limitations may limit the size of the market for the product.
We are also subject to numerous foreign regulatory requirements governing the conduct of clinical trials, manufacturing and marketing authorization, pricing and third-party reimbursement. The foreign regulatory approval process includes all of the risks associated with FDA approval described above as well as risks attributable to the satisfaction of local regulations in foreign jurisdictions. Approval by the FDA does not assure approval by regulatory authorities outside the United States.
Even if we obtain regulatory approvals, our marketed drugs will be subject to ongoing regulatory review. If we fail to comply with continuing U.S. and foreign regulations, we could lose our approvals to market drugs and our business would be seriously harmed.
Following any initial regulatory approval of any drugs we may develop, we will also be subject to continuing regulatory review, including the review of adverse drug experiences and clinical results that are reported after our drug products are made commercially available. This would include results from any post-marketing tests or vigilance required as a condition of approval. The manufacturer and manufacturing facilities we use to make any of our drug candidates will also be subject to periodic review and inspection by the FDA. The discovery of any previously unknown problems with the product, manufacturer or facility may result in restrictions on the drug or manufacturer or facility, including withdrawal of the drug from the market. We do not have, and currently do not intend to develop, the ability to manufacture material for our clinical trials or on a commercial scale. Reliance on third-party manufacturers entails risks to which we
18
Table of Contents
If we fail to comply with applicable continuing regulatory requirements, we may be subject to fines, suspension or withdrawal of regulatory approval, product recalls and seizures, operating restrictions and criminal prosecutions.
Even if we receive regulatory approval to market our product candidates, the market may not be receptive to our product candidates upon their commercial introduction, which will prevent us from becoming profitable.
The product candidates that we are developing are based upon new technologies or therapeutic approaches. Key participants in pharmaceutical marketplaces, such as physicians, third-party payors and consumers, may not accept a product intended to improve therapeutic results based on RNAi technology. As a result, it may be more difficult for us to convince the medical community and third-party payors to accept and use our products.
Other factors that we believe will materially affect market acceptance of our product candidates include:
| • | the timing of our receipt of any marketing approvals, the terms of any approvals and the countries in which approvals are obtained; | |
| • | the safety, efficacy and ease of administration; | |
| • | the success of our physician education programs; | |
| • | the availability of government and third-party payor reimbursement; | |
| • | the pricing of our products, particularly as compared to alternative treatments; and | |
| • | the availability of alternative effective treatments for the diseases that product candidates we develop are intended to treat. |
If we or our collaborators, manufacturers or service providers fail to comply with regulatory laws and regulations, we or they could be subject to enforcement actions, which could affect our ability to market and sell our products and may harm our reputation.
If we or our collaborators, manufacturers or service providers fail to comply with applicable federal, state or foreign laws or regulations, we could be subject to enforcement actions, which could affect our ability to develop, market and sell our products under development successfully and could harm our reputation and lead to reduced acceptance of our products by the market. These enforcement actions include:
| • | warning letters; | |
| • | recalls or public notification or medical product safety alerts; | |
| • | restrictions on, or prohibitions against, marketing our products; | |
| • | restrictions on importation of our products; | |
| • | suspension of review or refusal to approve pending applications; | |
| • | suspension or withdrawal of product approvals; | |
| • | product seizures; | |
| • | injunctions; and | |
| • | civil and criminal penalties and fines. | |
19
Table of Contents
Any drugs we develop may become subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, thereby harming our business.
The regulations that govern marketing approvals, pricing and reimbursement for new drugs vary widely from country to country. Some countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. Although we intend to monitor these regulations, our programs are currently in the early stages of development and we will not be able to assess the impact of price regulations for a number of years. As a result, we might obtain regulatory approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product and negatively impact the revenues we are able to generate from the sale of the product in that country.
Our ability to commercialize any products successfully also will depend in part on the extent to which reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers and other organizations. Even if we succeed in bringing one or more products to the market, these products may not be considered cost-effective, and the amount reimbursed for any products may be insufficient to allow us to sell our products on a competitive basis. Because our programs are in the early stages of development, we are unable at this time to determine their cost effectiveness and the level or method of reimbursement. Increasingly, the third-party payors who reimburse patients, such as government and private insurance plans, are requiring that drug companies provide them with predetermined discounts from list prices, and are challenging the prices charged for medical products. If the price we are able to charge for any products we develop is inadequate in light of our development and other costs, our profitability could be adversely affected.
We currently expect that any drugs we develop may need to be administered under the supervision of a physician. Under currently applicable law, drugs that are not usually self-administered may be eligible for coverage by the Medicare program if:
| • | they are incident to a physician’s services; | |
| • | they are “reasonable and necessary” for the diagnosis or treatment of the illness or injury for which they are administered according to accepted standard of medical practice; | |
| • | they are not excluded as immunizations; and | |
| • | they have not been determined by the FDA to be less than effective. |
There may be significant delays in obtaining coverage for newly-approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA. Moreover, eligibility for coverage does not imply that any drug will be reimbursed in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim payments for new drugs, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may be based on payments allowed for lower-cost drugs that are already reimbursed, may be incorporated into existing payments for other services and may reflect budgetary constraints or imperfections in Medicare data. Net prices for drugs may be reduced by mandatory discounts or rebates required by government health care programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the United States. Third party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement rates. Our inability to promptly obtain coverage and profitable reimbursement rates from both government-funded and private payors for new drugs that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products, and our overall financial condition.
We believe that the efforts of governments and third-party payors to contain or reduce the cost of healthcare will continue to affect the business and financial condition of pharmaceutical and biopharmaceutical companies. A number of legislative and regulatory proposals to change the healthcare system in the
20
Table of Contents
Another development that may affect the pricing of drugs is Congressional action regarding drug reimportation into the United States. The Medicare Prescription Drug Plan legislation, which became law in December 2003, requires the Secretary of Health and Human Services to promulgate regulations for drug reimportation from Canada into the United States under some circumstances, including when the drugs are sold at a lower price than in the United States. The Secretary retains the discretion not to implement a drug reimportation plan if he finds that the benefits do not outweigh the cost. Proponents of drug reimportation may attempt to pass legislation that would directly allow reimportation under certain circumstances. If legislation or regulations were passed allowing the reimportation of drugs, they could decrease the price we receive for any products that we may develop, negatively affecting our anticipated revenues and prospects for profitability.
There is a substantial risk of product liability claims in our business. If we are unable to obtain sufficient insurance, a product liability claim against us could adversely affect our business.
Our business exposes us to significant potential product liability risks that are inherent in the development, manufacturing and marketing of human therapeutic products. Product liability claims could delay or prevent completion of our clinical development programs. If we succeed in marketing products, such claims could result in an FDA investigation of the safety and effectiveness of our products, our manufacturing processes and facilities or our marketing programs, and potentially a recall of our products or more serious enforcement action, or limitations on the indications for which they may be used, or suspension or withdrawal of approval. We currently do not have any product liability insurance, but plan to obtain such insurance at appropriate levels prior to initiating clinical trials and at higher levels prior to marketing any of our drug candidates. Any insurance we obtain may not provide sufficient coverage against potential liabilities. Furthermore, clinical trial and product liability insurance is becoming increasingly expensive. As a result, we may be unable to obtain sufficient insurance at a reasonable cost to protect us against losses caused by product liability claims that could have a material adverse effect on our business.
If we do not comply with laws regulating the protection of the environment and health and human safety, our business could be adversely affected.
Our research and development involves the use of hazardous materials, chemicals and various radioactive compounds. We maintain quantities of various flammable and toxic chemicals in our facilities in Cambridge and Germany that are required for our research and development activities. We believe our procedures for storing, handling and disposing these materials in our Cambridge facility comply with the relevant guidelines of the City of Cambridge and the Commonwealth of Massachusetts and the procedures we employ in our German facility comply with the standards mandated by applicable German laws and guidelines. Although we believe that our safety procedures for handling and disposing of these materials comply with the standards mandated by applicable regulations, the risk of accidental contamination or injury from these materials cannot be eliminated. If an accident occurs, we could be held liable for resulting damages, which could be substantial. We are also subject to numerous environmental, health and workplace safety laws and regulations, including those governing laboratory procedures, exposure to blood-borne pathogens and the handling of biohazardous materials.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of these materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials. Additional federal, state and local laws and regulations affecting our
21
Table of Contents
Risks Related to Competition
The pharmaceutical market is intensely competitive. If we are unable to compete effectively with existing drugs, new treatment methods and new technologies, we may be unable to commercialize any drugs that we develop.
The pharmaceutical market is intensely competitive and rapidly changing. Many large pharmaceutical and biotechnology companies, academic institutions, governmental agencies and other public and private research organizations are pursuing the development of novel drugs for the same diseases that we are targeting or expect to target. Many of our competitors have:
| • | much greater financial, technical and human resources than we have at every stage of the discovery, development, manufacture and commercialization of products; | |
| • | more extensive experience in preclinical testing, conducting clinical trials, obtaining regulatory approvals, and in manufacturing and marketing pharmaceutical products; | |
| • | product candidates that are based on previously tested or accepted technologies; | |
| • | products that have been approved or are in late stages of development; and | |
| • | collaborative arrangements in our target markets with leading companies and research institutions. | |
We will face intense competition from drugs that have already been approved and accepted by the medical community for the treatment of the conditions for which we may develop drugs. We also expect to face competition from new drugs that enter the market. We believe a significant number of drugs are currently under development, and may become commercially available in the future, for the treatment of conditions for which we may try to develop drugs. For instance, we are currently evaluating siRNAs to suppress VEGF gene activity as a potential drug candidate for the treatment of wet AMD and we are currently evaluating the potential of siRNAs for the treatment of PD. One drug, Visudyne, is already marketed for the treatment of wet AMD, and numerous drugs are currently marketed for the treatment of PD. In addition, we are aware of a number of experimental drugs for the treatment of wet AMD that, unlike our product candidate, are in advanced stages of clinical development. These experimental drugs include Macugen, which is being developed by Eyetech Pharmaceuticals, Inc. in collaboration with Pfizer, Inc., and Lucentis, which is being developed by Genentech, Inc. in collaboration with Novartis. These drug candidates may be approved for marketing before our product candidate receives approval. Furthermore, our competitors’ products may be more effective, or marketed and sold more effectively, than any products we develop.
If we successfully develop drug candidates, and obtain approval for them, we will face competition based on many different factors, including:
| • | the safety and effectiveness of our products; | |
| • | the timing and scope of regulatory approvals for these products; | |
| • | the availability and cost of manufacturing, marketing and sales capabilities; | |
| • | price; | |
| • | reimbursement coverage; and | |
| • | patent position. | |
Our competitors may develop or commercialize products with significant advantages over any products we develop based on any of the factors listed above. Our competitors may therefore be more successful in commercializing their products than we are, which could adversely affect our competitive position and business. Competitive products may make any products we develop obsolete or noncompetitive before we can recover the expenses of developing and commercializing our drug candidates. Furthermore, we also face
22
Table of Contents
We face competition from other companies that are working to develop novel drugs using technology similar to ours. If these companies develop drugs more rapidly than we do or their technologies are more effective, our ability to successfully commercialize drugs will be adversely affected.
In addition to the competition we face from competing drugs in general, we also face competition from other companies working to develop novel drugs using technology that competes more directly with our own. We are aware of several other companies that are working in the field of RNAi, including Sirna Therapeutics, Inc., Acuity Pharmaceuticals, Inc., Nucleonics, Inc., Benitec Ltd. and CytRx Corporation. In addition, we granted a license to Isis Pharmaceuticals, Inc., or Isis, under which it may develop RNAi therapeutics against a limited number of targets. Any of these companies may develop its RNAi technology more rapidly and more effectively than us.
We also compete with companies working to develop antisense-based drugs. Like RNAi product candidates, antisense drugs target mRNAs in order to suppress the activity of specific genes. Isis is currently marketing an antisense drug and has several antisense drug candidates in clinical trials, and another company, Genta Inc., has an antisense drug candidate under FDA review. The development of antisense drugs is more advanced than that of RNAi therapeutics and may become the preferred technology for drugs that target mRNAs to silence specific genes.
Risks Related to Patents, Licenses and Trade Secrets
If we are not able to obtain and enforce patent protection for our discoveries, our ability to develop and commercialize our product candidates will be harmed.
Our success depends, in part, on our ability to protect proprietary methods and technologies that we develop under the patent and other intellectual property laws of the United States and other countries, so that we can prevent others from unlawfully using our inventions and proprietary information. However, we may not hold proprietary rights to some patents required for us to commercialize our proposed products. Because certain U.S. patent applications are confidential until patents issue, such as applications filed prior to November 29, 2000, or applications filed after such date which will not be filed in foreign countries, third parties may have filed patent applications for technology covered by our pending patent applications without our being aware of those applications, and our patent applications may not have priority over those applications. For this and other reasons, we may be unable to secure desired patent rights, thereby losing desired exclusivity. Further, we may be required to obtain licenses under third-party patents to market our proposed products or conduct our research and development or other activities. If licenses are not available to us on acceptable terms, we will not be able to market the affected products or conduct the desired activities.
Our strategy depends on our ability to rapidly identify and seek patent protection for our discoveries. In addition, we will rely on third-party collaborators to file patent applications relating to proprietary technology that we develop jointly during certain collaborations. The process of obtaining patent protection is expensive and time consuming. If our present or future collaborators fail to file and prosecute all necessary and desirable patent applications at a reasonable cost and in a timely manner, our business will be adversely affected. Despite our efforts and the efforts of our collaborators to protect our proprietary rights, unauthorized parties may be able to obtain and use information that we regard as proprietary. The mere issuance of a patent does not guarantee that it is valid or enforceable, so even if we obtain patents, they may not be valid or enforceable against third parties.
Our pending patent applications may not result in issued patents. The patent position of pharmaceutical or biotechnology companies, including ours, is generally uncertain and involves complex legal and factual considerations. The standards that the United States Patent and Trademark Office and its foreign counterparts use to grant patents are not always applied predictably or uniformly and can change. There is also no uniform, worldwide policy regarding the subject matter and scope of claims granted or allowable in
23
Table of Contents
We also rely on trade secrets, know-how and technology, which are not protected by patents, to maintain our competitive position. If any trade secret, know-how or other technology not protected by a patent were to be disclosed to or independently developed by a competitor, our business and financial condition could be materially adversely affected.
We license patent rights from third party owners. If such owners do not properly maintain or enforce the patents underlying such licenses, our competitive position and business prospects will be harmed.
We are a party to a number of licenses that give us rights to third party intellectual property that is necessary or useful for our business. In particular, we have obtained licenses from Isis, Carnegie Institution of Washington, Cancer Research Technology Limited, the Massachusetts Institute of Technology, the Whitehead Institute, Garching Innovation GmbH, representing the Max Planck Gesellschaft zur Förderung der Wissenschaften e.V., referred to as the Max Planck organization, and Cold Spring Harbor Laboratory. We also intend to enter into additional licenses to third party intellectual property in the future.
Our success will depend in part on the ability of our licensors to obtain, maintain and enforce patent protection for our licensed intellectual property, in particular, those patents to which we have secured exclusive rights. Our licensors may not successfully prosecute the patent applications to which we are licensed. Even if patents issue in respect of these patent applications, our licensors may fail to maintain these patents, may determine not to pursue litigation against other companies that are infringing these patents, or may pursue such litigation less aggressively than we would. Without protection for the intellectual property we license, other companies might be able to offer substantially identical products for sale, which could adversely affect our competitive business position and harm our business prospects.
One of our key patents, the so-called Kreutzer-Limmer patent, is the subject of an opposition proceeding in the European Patent Office, which could result in the invalidation of this patent.
We believe the so-called Kreutzer-Limmer patent is the only patent granted to date that specifically covers the use of short dsRNAs as therapeutics. A German Utility Model covering RNAi composition was granted in 2003, and a patent covering RNAi compositions and their use was granted in the European Union in 2002 and in South Africa in 2003, and related patent applications are pending in other countries, including the United States. A German Utility Model is a form of patent that is directed only to physical matter, such as medicines, and does not cover methods. The maximum period of protection afforded by the German Utility Model ends in 2010. After the grant of the Kreutzer-Limmer patent in the European Union, published under publication number EP 1144623B9, several oppositions to the issuance of the European patent were filed with the European Patent Office, or EPO, a practice that is allowed under the European patent convention. Each of the oppositions raises a number of grounds for the invalidation of the patent. The EPO opposition division in charge of the patent application may agree with one or more of the grounds and could revoke the patent in whole or restrict the scope of the claims. It may be several years before the outcome of the opposition proceeding is decided by the EPO.
In addition, the EPO is reviewing what is known as “disclaimer practice”, a process in which a patent applicant disclaims part of the claimed invention in a patent application in order to overcome prior art by means of a disclaimer having no basis in the original disclosure. The EPO may determine that disclaimer practice is not allowed under the European patent convention, which could result in the invalidation of the Kreutzer-Limmer patent. Even if the EPO determines that disclaimer practice is permissible under the European patent convention, publication number EP 1144623B9 would remain subject to the opposition described above. If the Kreutzer-Limmer patent is invalidated or limited for any reason, other companies will be better able to develop products that compete with ours, which could adversely affect our competitive business position, business prospects and financial condition.
24
Table of Contents
Other companies or organizations may assert patent rights that prevent us from developing and commercializing our products.
RNA interference is a relatively new scientific field that has generated many different patent applications from organizations and individuals seeking to obtain important patents in the field. These applications claim many different methods, compositions and processes relating to the discovery, development and commercialization of RNAi therapeutics. Because the field is so new, very few of these patent applications have been fully processed by government patent offices around the world, and there is a great deal of uncertainty about which patents will issue, when, to whom, and with what claims. It is likely that there will be significant litigation and other proceedings, such as interference proceedings in various patent offices, relating to patent rights in the RNAi field. Others may attempt to invalidate our intellectual property rights. Even if our rights are not directly challenged, disputes among third parties could lead to the weakening or invalidation of our intellectual property rights.
In addition, there are many issued and pending patents that claim aspects of oligonucleotide chemistry that we may need to apply to our siRNA drug candidates. There are also many issued patents that claim genes or portions of genes that may be relevant for siRNA drugs we wish to develop.
Thus, it is possible that one or more organizations will hold patent rights to which we will need a license. If those organizations refuse to grant us a license to such patent rights on reasonable terms, we will not be able to market products or perform research and development or other activities covered by these patents.
If we become involved in patent litigation or other proceedings related to a determination of rights, we could incur substantial costs and expenses, substantial liability for damages or be required to stop our product development and commercialization efforts.
A third party may sue us for infringing its patent rights. Likewise, we may need to resort to litigation to enforce a patent issued or licensed to us or to determine the scope and validity of third-party proprietary rights. In addition, a third party may claim that we have improperly obtained or used its confidential or proprietary information. The cost to us of any litigation or other proceeding relating to intellectual property rights, even if resolved in our favor, could be substantial, and the litigation would divert our management’s efforts. Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. Uncertainties resulting from the initiation and continuation of any litigation could limit our ability to continue our operations.
If any parties successfully claim that our creation or use of proprietary technologies infringes upon their intellectual property rights, we might be forced to pay damages, potentially including treble damages, if we are found to have willfully infringed on such parties’ patent rights. In addition to any damages we might have to pay, a court could require us to stop the infringing activity or obtain a license. Any license required under any patent may not be made available on commercially acceptable terms, if at all. In addition, such licenses are likely to be non-exclusive and, therefore, our competitors may have access to the same technology licensed to us. If we fail to obtain a required license and are unable to design around a patent, we may be unable to effectively market some of our technology and products, which could limit our ability to generate revenues or achieve profitability and possibly prevent us from generating revenue sufficient to sustain our operations. Moreover, we expect that a number of our collaborations will provide that royalties payable to us for licenses to our intellectual property may be offset by amounts paid by our collaborators to third parties who have competing or superior intellectual property positions in the relevant fields, which could result in significant reductions in our revenues from products developed through collaborations.
If we fail to comply with our obligations under any licenses or related agreements, we could lose license rights that are necessary for developing and protecting our RNAi technology and any related product candidates that we develop, or we could lose certain exclusive rights to grant sublicenses.
Our current licenses impose, and any future licenses we enter into are likely to impose, various development, commercialization, funding, royalty, diligence, sublicensing, insurance and other obligations on
25
Table of Contents
For two important pending patent applications, owned in part or solely by the Max Planck organization of Germany, our licenses include a condition requiring us to operate a German company comparable to our United States operation until at least December 2007. If we fail to comply with this condition, the owners of the patent applications that are the subject of these licenses would have the right to grant similar licenses to one other company for each pending patent application. We regard these pending patent applications as significant because they relate to important aspects of the structure of siRNA molecules and their use as therapeutics.
We have an agreement with Isis under which we were granted licenses to over 150 patents and patent applications that we believe will be useful to the development of RNAi therapeutics. If, by January 1, 2008, we or a collaborator have not completed the studies required for an investigational new drug application filing or similar foreign filing for at least one product candidate involving these patent rights, Isis would have the right to grant licenses to third parties for these patents and patent applications, thereby making our rights non-exclusive.
Confidentiality agreements with employees and others may not adequately prevent disclosure of trade secrets and other proprietary information.
In order to protect our proprietary technology and processes, we also rely in part on confidentiality agreements with our collaborators, employees, consultants, outside scientific collaborators and sponsored researchers and other advisors. These agreements may not effectively prevent disclosure of confidential information and may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. In addition, others may independently discover trade secrets and proprietary information, and in such cases we could not assert any trade secret rights against such party. Costly and time-consuming litigation could be necessary to enforce and determine the scope of our proprietary rights, and failure to obtain or maintain trade secret protection could adversely affect our competitive business position.
Risks Related to Our Common Stock
Purchasers in this offering will suffer immediate dilution.
If you purchase common stock in this offering, the value of your shares based on our actual book value will immediately be less than the offering price you paid. This reduction in the value of your equity is known as dilution. Based upon the net tangible book value of the common stock at December 31, 2003, your shares will be worth less per share than the price you paid in the offering. If the options and warrants we previously granted are exercised, additional dilution will occur. As of March 31, 2004, options to purchase 3,691,618 shares of common stock at a weighted average exercise price of $0.32 per share were outstanding, and warrants to purchase 125,000 shares of common stock at a weighted average exercise price of $4.50 per share were outstanding. In addition, if we raise additional funding by issuing additional equity securities, the newly-issued shares will further dilute your percentage ownership of our shares and may also reduce the value of your investment.
26
Table of Contents
Our stock price will fluctuate after this offering, which may cause your investment in our stock to suffer a decline in value.
After this offering, an active trading market in our stock might not develop or continue. If you purchase shares of our common stock in the offering, you will pay a price that was not established in a competitive market. Rather, you will pay a price that we negotiated with the representatives of the underwriters based upon an assessment of the valuation of our stock. The public market may not agree with or accept this valuation, in which case you may not be able to sell your shares at or above the initial offering price.
In addition, the market price of our common stock may fluctuate significantly in response to factors that are beyond our control. The stock market in general has recently experienced extreme price and volume fluctuations. The market prices of securities of pharmaceutical and biotechnology companies have been extremely volatile, and have experienced fluctuations that often have been unrelated or disproportionate to the operating performance of these companies. These broad market fluctuations could result in extreme fluctuations in the price of our common stock, which could cause a decline in the value of your investment.
We may incur significant costs from class action litigation due to our expected stock volatility.
Our stock price may fluctuate for many reasons, including as a result of public announcements regarding the progress of our development efforts, the addition or departure of our key personnel, variations in our quarterly operating results and changes in market valuations of pharmaceutical and biotechnology companies. Recently, when the market price of a stock has been volatile as our stock price may be, holders of that stock have occasionally brought securities class action litigation against the company that issued the stock. If any of our stockholders were to bring a lawsuit of this type against us, even if the lawsuit is without merit, we could incur substantial costs defending the lawsuit. The lawsuit could also divert the time and attention of our management.
A significant portion of our total outstanding shares are restricted from immediate resale but may be sold into the market in the near future. If there are substantial sales of our common stock, the price of our common stock could decline.
The price of our common stock could decline if there are substantial sales of our common stock and if there is a large number of shares of our common stock available for sale. After this offering, we will have outstanding shares of common stock based on the number of shares outstanding as of March 31, 2004. This includes the shares that we are selling in this offering, which may be resold in the public market immediately. The remaining shares, or % of our outstanding shares after this offering, are currently restricted as a result of securities laws or lock-up agreements but will be able to be sold in the near future as set forth below.
| Number of shares and % of total outstanding | Date available for sale into public market | |
| shares, or % | 180 days after the date of this prospectus due to lock-up agreements between the holders of these shares and the underwriters. However, the underwriters can waive the provisions of these lock-up agreements and allow these stockholders to sell their shares at any time. | |
| shares, or �� % | Between 180 and 365 days after the date of this prospectus, depending on the requirements of the federal securities laws. |
After this offering, the holders of an aggregate of 22,733,347 shares of common stock as of March 31, 2004, will have rights, subject to some conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other stockholders. The holders of warrants to purchase an aggregate of 125,000 shares of our common stock will also be entitled to include such shares in registration statements that we may file in the future. We also intend to
27
Table of Contents
Due to these factors, sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock.
Insiders will continue to have substantial control over Alnylam after this offering and could delay or prevent a change in corporate control.
After this offering, our directors, executive officers and principal stockholders, together with their affiliates, will beneficially own, in the aggregate, approximately % of our outstanding common stock. As a result, these stockholders, if acting together, may have the ability to determine the outcome of matters submitted to our stockholders for approval, including the election of directors and any merger, consolidation or sale of all or substantially all of our assets. In addition, these persons, acting together, may have the ability to control the management and affairs of our company. Accordingly, this concentration of ownership may harm the market price of our common stock by:
| • | delaying, deferring or preventing a change in control of our company; | |
| • | impeding a merger, consolidation, takeover or other business combination involving our company; or | |
| • | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our company. |
We have broad discretion in the use of the net proceeds from this offering and may not use them effectively.
We cannot specify with certainty the particular uses of the net proceeds that we will receive from this offering. Our management will have broad discretion in the application of the net proceeds, including for any of the purposes described in the “Use of Proceeds” section of this prospectus. Our stockholders may not agree with the manner in which our management chooses to allocate and spend the net proceeds. The failure by our management to apply these funds effectively could have a material adverse effect on our business. Pending their use, we may invest the net proceeds from this offering in a manner that does not produce income or that loses value.
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our certificate of incorporation and our bylaws that will become effective upon the completion of this offering may delay or prevent an acquisition of us or a change in our management. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors. Because our board of directors is responsible for appointing the members of our management team, these provisions could in turn affect any attempt by our stockholders to replace current members of our management team. These provisions include:
| • | a classified board of directors; | |
| • | a prohibition on actions by our stockholders by written consent; | |
| • | the ability of our board of directors to issue preferred stock without stockholder approval, which could be used to institute a “poison pill” that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by our board of directors; |
28
Table of Contents
| • | limitations on the removal of directors; and | |
| • | advance notice requirements for election to our board of directors and for proposing matters that can be acted upon at stockholder meetings. |
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner. These provisions would apply even if the proposed merger or acquisition could be considered beneficial by some stockholders.
29
Table of Contents
FORWARD-LOOKING STATEMENTS
This prospectus including the sections entitled “Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and “Business,” contains forward-looking statements. We may, in some cases, use words such as “project,” “believe,” “anticipate,” “plan,” “expect,” “estimate,” “intend,” “should,” “would,” “could,” “potentially,” “will,” or “may,” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Forward-looking statements in this prospectus may include statements about:
| • | our research and development activities, including development of our product engine, and projected expenditures; | |
| • | the advantages of our technology and product candidates as compared to others; | |
| • | our ability to obtain and maintain collaborators for certain of our development programs and the terms of these arrangements; | |
| • | our ability to successfully complete preclinical testing; | |
| • | our ability to commence, and the timing of, any clinical trials, including for an AMD product candidate; | |
| • | the completion and success of any clinical trials that we commence; | |
| • | the receipt of regulatory approvals by our collaborators or us; | |
| • | our ability to maintain and establish intellectual property rights in our product candidates; | |
| • | our ability to obtain licenses relating to any necessary third party intellectual property; | |
| • | the outcome of any future litigation; | |
| • | our ability to retain and hire necessary employees and appropriately staff our development programs; | |
| • | our spending of the proceeds from this offering; | |
| • | our cash needs; | |
| • | implementation of our corporate strategy; and | |
| • | our financial performance. |
There are a number of important factors that could cause actual results to differ materially from the results anticipated by these forward-looking statements. These important factors include those that we discuss in this prospectus under the caption “Risk Factors.” You should read these factors and the other cautionary statements made in this prospectus as being applicable to all related forward-looking statements wherever they appear in this prospectus. If one or more of these factors materialize, or if any underlying assumptions prove incorrect, our actual results, performance or achievements may vary materially from any future results, performance or achievements expressed or implied by these forward-looking statements. The forward-looking statements contained in this prospectus are excluded from the safe harbor protection provided by the Private Securities Litigation Reform Act of 1995 and Section 27A of the Securities Act of 1933. We undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
30
Table of Contents
USE OF PROCEEDS
We estimate that our net proceeds from the sale of shares of common stock in this offering will be approximately $ million, assuming an initial public offering price of $ per share, the mid-point of the estimated price range shown on the cover of this prospectus, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. If the underwriters exercise their over-allotment option, we estimate that we will receive additional net proceeds of approximately $ million. We expect to use the net proceeds to fund:
| • | research and development activities, including development of our RNAi capabilities, selection of product candidates and preclinical and clinical development of our product candidates; | |
| • | acquisition, licensing and protection of intellectual property rights; and | |
| • | working capital, capital expenditures and other general corporate purposes. |
The amounts and timing of our actual expenditures will depend upon numerous factors, including the status of our research and development efforts, the timing and success of preclinical testing, the timing and success of any clinical trials we may commence in the future, the timing of regulatory submissions, the amount of proceeds actually raised in this offering, the amount of cash generated by our operations, the amount of competition we face and how successful we are with obtaining any required licenses and entering into collaboration arrangements. We may also use a portion of the proceeds for the acquisition of, or investment in, companies, technologies, products or assets that complement our business. However, we have no present understandings, commitments or agreements to enter into any potential acquisitions or investments. We currently estimate that of the net proceeds from this offering we will spend between 65% and 75% on research and development activities, between 5% and 15% on the acquisition, licensing and protection of intellectual property rights and between 15% and 25% on working capital, capital expenditures and other general corporate purposes. We have not determined the amounts we plan to spend on any specific product candidate or program or the timing of these expenditures. Our management will have broad discretion to allocate the net proceeds from this offering. We do not expect the net proceeds from this offering to be sufficient to fund the completion of the development of any product candidate and we expect to need to raise additional funds prior to being able to market any product.
Pending utilization of the net proceeds as described above, we intend to invest the net proceeds of the offering in short-term investment grade and U.S. government securities.
DIVIDEND POLICY
We have never paid or declared any cash dividends on our common stock. We currently intend to retain earnings, if any, to finance the growth and development of our business and we do not expect to pay any cash dividends on our common stock in the foreseeable future. Payment of future dividends, if any, will be at the discretion of our board of directors and will depend on our financial condition, results of operations, capital requirements, restrictions contained in current or future financing instruments and other factors our board of directors deems relevant.
31
Table of Contents
CAPITALIZATION
The following table sets forth our cash and cash equivalents, note payable and capitalization as of December 31, 2003:
| • | on an actual basis; | |
| • | on an as adjusted basis to (1) reflect the conversion of all of our shares of convertible preferred stock outstanding as of December 31, 2003 into an aggregate of 21,066,680 shares of common stock upon the closing of this offering and (2) give effect to the issuance and sale of shares of common stock in this offering at an assumed initial public offering price of $ per share, the mid-point of the estimated price range shown on the cover page of this prospectus, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. | |
You should read the information below with our consolidated financial statements and the related notes and “Management’s Discussion and Analysis of Financial Commission and Results of Operations” included elsewhere in this prospectus.
| As of December 31, 2003 | ||||||||||
| Actual | As Adjusted | |||||||||
| ($ in thousands, except | ||||||||||
| per share data) | ||||||||||
| Cash and cash equivalents | $ | 23,193 | $ | |||||||
| Note payable | $ | 1,859 | $ | |||||||
| Redeemable convertible preferred stock, $0.0001 par value; 22,272,088 shares authorized and 21,066,680 shares issued and outstanding, actual; and no shares authorized, issued or outstanding, as adjusted | 55,189 | |||||||||
| Stockholders’ equity (deficit): | ||||||||||
| Common stock $0.0001 par value; 34,739,392 shares authorized and 4,277,860 shares issued and outstanding, actual; and 125,000,000 shares authorized and shares issued and outstanding, as adjusted | 1 | |||||||||
| Additional paid-in capital | 7,415 | |||||||||
| Deferred compensation | (4,681 | ) | ||||||||
| Accumulated other comprehensive income | 76 | |||||||||
| Deficit accumulated in the development stage | (29,518 | ) | ||||||||
| Total stockholders’ equity (deficit) | (26,707 | ) | ||||||||
| Total capitalization | $ | 30,341 | $ | |||||||
The above table excludes:
| • | 3,217,756 shares of common stock issuable upon exercise of options outstanding as of December 31, 2003 with a weighted average exercise price of $0.28 per share; | |
| • | 25,000 shares of common stock issuable upon exercise of an outstanding warrant as of December 31, 2003 with an exercise price of $2.50 per share; and | |
| • | 947,644 shares of common stock reserved for issuance under our stock option plans as of December 31, 2003. | |
32
Table of Contents
DILUTION
The net tangible book value of our common stock as of December 31, 2003 was approximately $(29.1) million, or $(6.80) per share. Net tangible book value per share represents our total tangible assets less the sum of the total liabilities and the liquidation value of our convertible preferred stock and divided by the number of shares of common stock outstanding. Net tangible book value dilution per share to new investors is the difference between the amount per share paid by purchasers of common stock in this offering and the pro forma as adjusted net tangible book value per share immediately following the offering.
After giving effect to the conversion of all outstanding shares of convertible preferred stock into an aggregate of 22,733,347 shares of common stock upon the closing of this offering, including 1,666,667 shares of common stock issuable upon conversion of the convertible preferred stock issued to Isis on March 11, 2004, and after giving effect to the issuance and sale of the shares of common stock in this offering, at an assumed offering price of $ per share and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us, our pro forma as adjusted net tangible book value as of December 31, 2003 would have been $ million, or $ per share. This represents an immediate increase in pro forma net tangible book value to existing stockholders of $ per share. The initial public offering price per share will significantly exceed the net tangible book value per share. Accordingly, new investors who purchase common stock in this offering will suffer an immediate dilution of their investment of $ per share. The following table illustrates this per share dilution:
| Assumed initial public offering price per share | $ | ||||||||
| Net tangible book value per share as of December 31, 2003. | $ | (6.80 | ) | ||||||
| Pro forma increase in net tangible book value per share attributable to conversion of convertible preferred stock | 7.83 | ||||||||
| Increase in net tangible book value per share attributable to this offering | |||||||||
| Pro forma as adjusted net tangible book value per share after this offering | |||||||||
| Dilution of net tangible book value per share to new investors | $ | ||||||||
If the underwriters exercise their over-allotment option in full, the pro forma net tangible book value per share after the offering would be $ per share, the increase in net tangible book value per share to existing stockholders would be $ per share and the dilution to new investors would be $ per share.
The following table summarizes, on a pro forma as adjusted basis as of December 31, 2003, giving effect to the conversion of all shares of convertible preferred stock outstanding into shares of common stock, the differences between the number of shares of common stock purchased from us, the total consideration paid to us and the average price per share paid by existing stockholders and by new investors. The calculation below is based on an assumed initial public offering price of $ �� per share, before deduction of estimated underwriting discounts and commissions and estimated offering expenses payable by us:
| Shares Issued | Total Consideration | ||||||||||||||||||||
| Average Price | |||||||||||||||||||||
| Number | % | Amount | % | Per Share | |||||||||||||||||
| Existing stockholders | 25,344,540 | % | $ | 53,906,000 | % | $ | 2.13 | ||||||||||||||
| New investors | |||||||||||||||||||||
| Total | 100 | % | $ | 100 | % | ||||||||||||||||
The tables above assume no exercise of stock options or warrants outstanding as of December 31, 2003. At December 31, 2003, there were 3,217,756 shares of common stock issuable upon exercise of outstanding stock options at a weighted average exercise price of $0.28 per share, and 25,000 shares of common stock issuable upon exercise of an outstanding warrant at an exercise price of $2.50 per share. To the extent that
33
Table of Contents
If the underwriters exercise their over-allotment option in full, the number of shares held by new investors will increase to shares, or % of the total number of shares of common stock outstanding after this offering.
34
Table of Contents
SELECTED CONDENSED CONSOLIDATED FINANCIAL DATA
The following table presents our historical financial data. You should read carefully the financial statements included in this prospectus, including the notes to the financial statements and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” The selected financial data in this section are not intended to replace the financial statements.
We derived the statement of operations data for the period from June 14, 2002 through December 31, 2002 and the year ended December 31, 2003 and the balance sheet data as of December 31, 2002 and 2003 from our consolidated financial statements, which have been audited by PricewaterhouseCoopers LLP, independent accountants, and are included elsewhere in this prospectus. Historical results are not necessarily indicative of future results. See the notes to the financial statements for an explanation of the method used to determine the number of shares used in computing basic and diluted and pro forma basic and diluted net loss per common share.
Pro forma basic and diluted net loss per common share have been calculated assuming the conversion of all outstanding shares of convertible preferred stock into shares of common stock. On July 31, 2003, we acquired all of the outstanding voting shares of Alnylam Europe for $1.5 million in cash and 1,549,216 shares of our common stock, and assumed $7.1 million of debt. In connection with the acquisition, we allocated $4.6 million of the purchase price to purchased in-process research and development, which we expensed immediately. The results of Alnylam Europe have been included in our consolidated results from the date of acquisition.
| Period from Inception | Year Ended | ||||||||||
| (June 14, 2002) through | December 31, | ||||||||||
| December 31, 2002 | 2003 | ||||||||||
| ($ in thousands, except | |||||||||||
| per share data) | |||||||||||
Consolidated Statements of Operations Data: | |||||||||||
| Revenue from research collaborators | $ | – | $ | 176 | |||||||
| Costs and expenses | |||||||||||
| Research and development(1) | 3,342 | 13,097 | |||||||||
| General and administrative(1) | 880 | 7,527 | |||||||||
| Purchased in-process research and development | — | 4,609 | |||||||||
| Total operating costs and expenses | 4,222 | 25,233 | |||||||||
| Loss from operations | (4,222 | ) | (25,057 | ) | |||||||
| Other income, net | 86 | 24 | |||||||||
| Net loss | (4,136 | ) | (25,033 | ) | |||||||
| Accretion of redeemable convertible preferred stock | (748 | ) | (2,906 | ) | |||||||
| Net loss attributable to common stockholders | (4,884 | ) | (27,939 | ) | |||||||
| Net loss per common share (basic and diluted) | $ | (7.76 | ) | $ | (15.60 | ) | |||||
| Weighted average shares used to compute basic and diluted net loss per common share | 629,548 | 1,791,063 | |||||||||
| Pro forma net loss per common share (basic and diluted) | $ | (1.65 | ) | ||||||||
| Shares used to compute basic and diluted pro forma net loss per common share | 15,157,601 | ||||||||||
| (1) Noncash stock-based compensation expense included in these amounts are as follows: | |||||||||||
| Research and development | $ | 172 | $ | 2,832 | |||||||
| General and administrative | — | 623 | |||||||||
| Total noncash stock-based compensation expense | $ | 172 | $ | 3,455 | |||||||
| As of December 31, | ||||||||
| 2002 | 2003 | |||||||
| ($ in thousands) | ||||||||
Consolidated Balance Sheet Data: | ||||||||
| Cash and cash equivalents | $ | 15,477 | $ | 23,193 | ||||
| Working capital | 12,846 | 20,345 | ||||||
| Total assets | 16,111 | 35,183 | ||||||
| Note payable | — | (1,859 | ) | |||||
| Redeemable convertible preferred stock | 18,084 | 55,189 | ||||||
| Total stockholders’ equity (deficit) | (4,646 | ) | (26,707 | ) | ||||
35
Table of Contents
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
The following discussion and analysis of financial condition and results of operations should be read together with “Selected Condensed Consolidated Financial Data,” and our consolidated financial statements and accompanying notes appearing elsewhere in this prospectus. This discussion contains forward-looking statements, based on current expectations and related to future events and our future financial performance, that involve risks and uncertainties. Our actual results may differ materially from those anticipated in these forward-looking statements as a result of many important factors, including those set forth under “Risk Factors” and elsewhere in this prospectus. See “Forward-Looking Statements.”
Overview
We are a biopharmaceutical company that is seeking to develop and commercialize new drugs that work through a recently discovered system in cells known as RNA interference, or RNAi. We believe that RNAi therapeutics have the potential to become a major class of drugs with applications in a wide range of therapeutic areas. We have initiated programs to develop RNAi therapeutics that will be administered directly to diseased parts of the body. We are also working to extend our capabilities by investing in RNAi therapeutics that will be administered systemically in order to treat a broad range of diseases. To realize the potential of RNAi therapeutics, we are developing capabilities that we can apply to any specific small interfering RNA, or siRNA, in a systematic way to endow it with drug-like properties. We use the term product engine to describe these capabilities because we believe they will enable us to develop many products across a variety of therapeutic areas. We expect that our product engine will enable us to produce RNAi therapeutic candidates that are potent against and specific for a particular target, appropriately stable and able to penetrate cells of target tissues.
We commenced operations in June 2002. To date, our revenue has been derived primarily from our strategic alliance with Merck. We received a $2.0 million license fee from Merck in 2003, almost all of which has been deferred and is being recognized as revenue over six years. We expect our revenues to continue to be derived primarily from strategic alliances. Since our inception, we have generated significant losses. As of December 31, 2003, we had an accumulated deficit of $29.5 million. We have funded our operations to date primarily through proceeds of $44.8 million from the sale of equity securities to investors. We are a development stage company and have yet to submit any drug applications to any regulatory authority. We have focused our efforts since inception primarily on business planning, research and development, acquiring intellectual property rights, recruiting management and technical staff, and raising capital. We are unable to predict when, if ever, we will be able to commence sales of any product. We have not achieved profitability on a quarterly or annual basis and we expect to incur significant additional losses over the next several years. We expect our net losses to increase primarily due to research and development activities relating to our collaborations, drug development programs and other general corporate activities. We anticipate that our operating results will fluctuate for the foreseeable future. Therefore, period-to-period comparisons should not be relied upon as predictive of the results in future periods. Our sources of potential funding for the next several years are expected to include proceeds from this offering, the sale of equity, license and other fees, funded research and development payments, and milestone payments under existing and future collaborative arrangements.
In July 2003, we acquired Ribopharma AG, now called Alnylam Europe, AG, a RNAi company based in Kulmbach, Germany. To effect the acquisition, we paid $1.5 million in cash and transaction costs of $0.4 million and issued common shares with a fair value of $1.9 million. In addition, we assumed $7.1 million in debt of which $3.0 million was subsequently paid in cash and $4.1 million was settled through the issuance of our Series B redeemable convertible preferred stock. As a result of the acquisition, we expensed $4.6 million of purchased in-process research and development and allocated $5.8 million to long lived assets representing the value ascribed to the Alnylam Europe work force, core technology and fixed assets acquired in the transaction. The results of Alnylam Europe are included in our consolidated results from the date of acquisition.
36
Table of Contents
Research and Development
Since our inception, we have focused on drug discovery and development programs. Research and development expenses represented approximately 79% of our total operating expenses for the period from June 14, 2002 through December 31, 2002 and 52% of our total operating expenses for the year ended December 31, 2003. We have not tracked our historical research and development costs or our personnel and personnel-related costs on a project-by-project basis, since the majority of our efforts have been focused on the development of capabilities associated with our product engine rather than on specific projects. Major components of our research and development expense for 2003 include salaries and benefits of $3.0 million, stock-based compensation charges of $2.8 million, laboratory costs of $1.9 million, costs to license technology of $1.7 million and costs associated with our intellectual property of $1.4 million.
We have initiated two programs to identify specific siRNAs for potential further development as Direct RNAi drug candidates and we expect to initiate additional programs as the capabilities of our product engine evolve. Our current programs are focused on AMD and PD. We are currently evaluating several RNAi therapeutics in animal models and expect to begin a clinical trial for an AMD product candidate in 2005. We recently entered into a collaboration with the Mayo Foundation for Medical Education and Research and the Mayo Clinic Jacksonville to explore the potential of a PD treatment by initiating testing in animal models.
There is a risk that any drug discovery and development program may not produce revenue because of the risks inherent in drug discovery and development. Moreover, there are uncertainties specific to any new field of drug discovery, including RNAi. The successful development of any product candidate we develop is highly uncertain. Due to the numerous risks associated with developing drugs, we cannot reasonably estimate or know the nature, timing and estimated costs of the efforts necessary to complete the development of, or the period in which material net cash inflows are expected to commence from, any potential product candidate. These risks include the uncertainty of:
| • | our ability to progress any product candidates into preclinical and clinical trials; | |
| • | the scope, rate and progress of our preclinical trials and other research and development activities; | |
| • | the scope, rate of progress and cost of any clinical trials we commence; | |
| • | clinical trial results; | |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; | |
| • | the terms and timing of any collaborative, licensing and other arrangements that we may establish; | |
| • | the cost and timing of regulatory approvals; | |
| • | the cost and timing of establishing sales, marketing and distribution capabilities; | |
| • | the cost of establishing clinical and commercial supplies of any products that we may develop; and | |
| • | the effect of competing technological and market developments. |
Any failure to complete any stage of the development of any potential products in a timely manner could have a material adverse effect on our operations, financial position and liquidity. A discussion of the risks and uncertainties associated with completing our projects on schedule, or at all, and the potential consequences of failing to do so, are set forth in the “Risk Factors” section of this prospectus.
Critical Accounting Policies and Estimates
While our significant accounting policies are more fully described in the notes to our consolidated financial statements included in this prospectus, we believe the following accounting policies to be the most
37
Table of Contents
Use of Estimates
Our discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States. The preparation of our consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses. On an ongoing basis, we evaluate our estimates and judgments, including those related to revenue recognition, accrued expenses and the fair value of our common and preferred stock, particularly as it relates to stock-based compensation. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions and could have a material impact on our reported results.
With respect to our estimates involved in the determination of the fair value of our common stock, during 2003 the board of directors evaluated several events that provided indicators of the fair value of our common stock including (1) a valuation that was performed in connection with the acquisition of Alnylam Europe in July 2003, (2) the fair value of our Series B and Series C convertible preferred stock that was issued in July, September and October 2003, and its relation to the value of our common stock, and (3) the impact of our proposed offering of common stock. These factors indicated that the options granted to employees during 2003 had a deemed fair value that was higher than the exercise price. This caused us to record deferred compensation of $3.3 million during 2003 and related compensation expense of $0.7 million in 2003. The deferred compensation will be recorded as an expense over the vesting period of the underlying stock options in accordance with the method prescribed by the Financial Accounting Standards Board, or FASB, Interpretation No. 28,Accounting for Stock Appreciation Rights and Other Variable Stock Options and Award Plans, or FIN 28, as this method appropriately matches the compensation expense related to the services performed by the option holder with the period over which each of the options vest from the date of grant. We expect to record amortization of this deferred compensation of $1.4 million in 2004, $0.7 million in 2005, $0.4 million in 2006 and $0.1 million in 2007, subject to employee terminations.
In connection with stock options granted to nonemployees for services during the year ended December 31, 2003 and our determination of the fair value of our common stock, we have recorded aggregate deferred compensation of approximately $1.2 million, which represents the fair value of nonemployee grants. The deferred compensation will be recorded as an expense over the vesting period of the underlying stock options in accordance with the method prescribed by FIN 28, as this method appropriately matches the compensation expense related to the services performed by the option holder with the period over which each of the options vest from the date of grant. At the end of each financial reporting period prior to vesting, the value of these options, as calculated using the Black-Scholes option pricing model, will be remeasured using the then-current fair value of our common stock. At that point, deferred compensation and the noncash compensation recognized during that period will be adjusted accordingly. Stock-based compensation expense related to these nonemployee options for 2003 was $0.5 million. Since the fair value of the common stock to nonemployees is subject to change in the future, the compensation expense recognized during the year ended December 31, 2003 and prior years may not be indicative of future compensation charges.
We have also recorded cumulative deferred compensation of $3.8 million related to restricted stock awards that were issued to nonemployees in 2002. Shares remaining unvested or subject to forfeiture for nonemployees still providing services are subject to a mark-to-market adjustment during each reporting period prior to vesting in full. The deferred compensation will be recorded as an expense over the vesting period of the underlying restricted stock in accordance with the method prescribed by FIN 28, as this method appropriately matches the compensation expense related to the services performed by the option holder with the period over which each of the options vest from the date of grant. We recorded noncash stock-based
38
Table of Contents
Acquired Technology
We have licensed technology that we expect to utilize in our research and development activities. The terms of the licenses may provide for up-front payments, annual maintenance payments, milestone payments based upon the achievement of specified events and royalties based on product sales. We account for the costs associated with obtaining licenses in accordance with FASB Statement No. 2,Accounting for Research and Development Costs. Under this standard, we determine whether the technology we are licensing relates to a particular research and development project with no alternative use. If it is determined that there are no alternative uses, the amount is expensed as incurred. Alternatively, the costs are capitalized and amortized over their estimated useful life. To date, we have expensed $8.2 million of acquired technology, including $4.6 million of purchased in-process research and development that is believed to have no alternative uses. We have capitalized $3.6 million of core technology, with a 10-year estimated useful life, which was acquired in 2003 as part of the Alnylam Europe acquisition.
Long-lived Assets
We generally depreciate property and equipment using the straight-line method over the asset’s estimated economic life, which ranges from two years to eight years. Determining the economic lives of property and equipment requires us to make significant judgments that can materially impact our operating results. As of December 31, 2003, there was approximately $3.9 million of intangible assets on our consolidated balance sheet, including $3.6 million of core technology and $0.4 million related to the Alnylam Europe workforce. We amortize acquired intangible assets using the straight-line method over their estimated economic lives, which range from four years to 10 years. Determining the economic lives of acquired intangible assets requires us to make significant judgment and estimates, and can materially impact our operating results. If our estimates require adjustment, it could have a material impact on our reported results.
Our policy regarding long-lived assets is to evaluate the recoverability or usefulness of these assets when the facts and circumstances suggest that these assets may be impaired. This analysis relies on a number of factors, including changes in strategic direction, business plans, regulatory developments, economic and budget projections, technological improvements and operating results. The test of recoverability or usefulness is a comparison of the asset value to the undiscounted cash flow of its expected cumulative net operating cash flow over the asset’s remaining useful life. Any write-downs would be treated as permanent reductions in the carrying amount of the asset and an operating loss would be recognized. To date, we have had recurring operating losses and the recoverability of our long-lived assets is contingent upon executing our business plan that includes obtaining significant revenue from research collaborations. If we are unable to execute our business plan, we may be required to write down the value of our long-lived assets in future periods.
In accounting for the acquisition of Alnylam Europe, we allocated the purchase price to the fair value of the acquired tangible and intangible assets, including purchased in-process research and development, which requires us to make several significant judgments and estimates. In preparing the allocation, we used a discounted cash flow model to value the intangibles of Alnylam Europe, which requires us to make assumptions and estimates about, among other things: (1) the time and investment that will be required to develop the projects and related technologies; (2) the amount of revenues, royalties and milestone payments that will be derived from the projects; and (3) the appropriate discount rates to be used in the analysis. Use of different estimates and judgments could yield materially different results in our analysis, and could result in materially different asset values and purchased in-process research and development charges.
39
Table of Contents
Revenue Recognition
We recognize revenue in accordance with the Securities and Exchange Commission’s Staff Accounting Bulletin No. 104,Revenue Recognition in Financial Statements. We have entered into a collaboration agreement with Merck. Revenue from this collaboration agreement includes nonrefundable license fees, milestones and royalties. When evaluating multiple element arrangements, we consider whether the components of the arrangement represents separate units of accounting as defined in Emerging Issues Task Force Issue No. 00-21,Revenue Arrangements with Multiple Deliverables, or EITF 00-21. Application of these standards requires subjective determinations and requires management to make judgments about value of the individual elements and whether it is separable from the other aspects of the contractual relationship. To date, we have determined that our upfront non-refundable license fees cannot be separated from our ongoing collaborative activities, and accordingly, do not treat them as a separate element. Nonrefundable license fees are recognized as revenue as we perform under the collaboration agreement. Where our level of effort is relatively constant over the performance period, we recognize total fixed or determined contract revenues on a straight-line basis over the development period set forth in the contract.
We recognize milestone payments as revenue upon achievement of the milestone only if (1) it represents a separate unit of accounting as defined in EITF 00-21; (2) the milestone payments are nonrefundable; (3) substantive effort is involved in achieving the milestone; and (4) the amount of the milestone is reasonable in relation to the effort expended or the risk associated with achievement of the milestone. If any of these conditions are not met, we defer the milestone payments and recognize them as revenue over the term of the contract as we complete our performance obligations. To date, we have not recognized revenue from any milestone payments.
Results of Operations
Year Ended December 31, 2003 Compared to the Period from June 14, 2002 (Date of Inception) through December 31, 2002
Revenue
We do not currently sell any products. To date, our revenue has been derived primarily from our strategic alliance with Merck. We received a $2.0 million license fee from Merck in 2003, almost all of which has been deferred and is being recognized as revenue over six years, the estimated period of performance under the collaboration agreement. We did not recognize any revenue in 2002. We expect our revenues to continue to be derived primarily from strategic alliances. In addition, we have recently established license programs for research reagents and services, which are expected to provide revenues from license fees and from royalties on sales. Over time, we expect to generate revenue from sales of therapeutic products and from royalties on sales of therapeutic products.
Research and Development Expenses
We expense research and development costs as incurred. Research and development expenses were $13.1 million in 2003, an increase of $9.8 million from $3.3 million in 2002. This increase resulted primarily from an increase in noncash stock-based compensation of $2.6 million, research and development expenses incurred by Alnylam Europe, which we acquired in July 2003, of $2.1 million, and the increase in payroll and related costs of our research and development team of $2.1 million. The remainder of the increase is primarily a result of an expansion of our technology development program during 2003 and the impact of a full year of operations in 2003. We expect to continue to devote substantial resources to research and development and we expect that research and development expenses will continue to increase in the future.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related expenses, professional fees, and general corporate activities. General and administrative expenses were $7.5 million in 2003, an increase of $6.6 million from $0.9 million in 2002. This resulted from an increase due to the addition of Alnylam
40
Table of Contents
Purchased In-Process Research and Development
In July 2003, in connection with our acquisition of Alnylam Europe, we allocated $4.6 million of the purchase price to purchased in-process research and development, which we recorded as an expense in our consolidated statement of operations in 2003. As of December 31, 2003, the technological feasibility of the projects had not yet been reached and management believes the assumptions included in the valuation analysis continue to be valid.
Below is a brief description of the Alnylam Europe purchased in-process research and development projects and their status as of December 31, 2003.
| Estimated | ||||||||||||||
| Cost to | Year of | |||||||||||||
| Value at | Complete at | Expected | ||||||||||||
| Program Description | Development Status at | Acquisition | December 31, | Product | ||||||||||
| or Indication | December 31, 2003 | Date | 2003 | Launch | ||||||||||
| ($ in thousands) | ||||||||||||||
| Malignant Melanoma | Research | $ | 1,756 | $ | 12,871 | 2012 | (1) | |||||||
| Pancreatic Carcinoma | Research | 2,853 | 10,769 | 2012 | (1) | |||||||||
| Total: | $ | 4,609 | $ | 23,640 | ||||||||||
| (1) | Our intention for these programs is to conduct the first phase of clinical trials and then out-license the programs to a partner. Upon out-licensing, we expect the partner to bear all development costs and control clinical development. We expect to earn payments upon the attainment of clinical milestones by any such partner and royalties on product sales. Since such a partner would control the clinical development, we will be unable to influence the timing of the achievement of the milestones, if at all, or the estimated year of the product launch, if at all. Our valuation assumed a development period of approximately 10 years, with milestones being earned during that period, which management believes is a typical time period required to bring a drug to market. Actual results will differ from these estimates due to the uncertainties surrounding drug development. |
Our strategy is to focus on the development of our Direct RNAi programs for AMD and PD. In addition we are continuing to build the capabilities of our product engine to enable the development of Systemic RNAi programs and expand the range of diseases that can be treated with our RNAi therapeutics. We believe Systemic RNAi capabilities will enable us to develop RNAi therapeutics to treat a broader range of diseases, including cancers such as malignant melanoma and pancreatic carcinoma.
Interest Income and Expense
Interest income was $0.2 million in 2003 and $0.1 million in 2002. This increase resulted primarily from higher cash balances due to net proceeds of $27.5 million from the issuance of convertible preferred stock in 2003. Interest expense was $0.1 million in 2003, and relates to the equipment line of credit used for financing equipment purchases. We had no interest expense in 2002.
Accretion of Redeemable Convertible Preferred Stock
Accretion of redeemable convertible preferred stock relates primarily to the 10% annual interest feature on the Series A and Series B redeemable convertible preferred stock. The accretion was $2.9 million in 2003, an increase of $2.2 million from $0.7 million in 2002. The increase is primarily from the issuance of
41
Table of Contents
Liquidity and Capital Resources
We commenced operations in June 2002. Since our inception, we have generated significant losses. As of December 31, 2003, we had an accumulated deficit in the development stage of $29.5 million. We have funded our operations primarily through the proceeds of $44.8 million from the sale of equity securities. As of December 31, 2003, we had restricted cash of $2.7 million and cash and cash equivalents of $23.2 million, consisting primarily of money market instruments, compared to cash and cash equivalents of $15.5 million as of December 31, 2002.
Net cash used in operating activities was $1.3 million in 2002 and $12.8 million in 2003. The use of cash in both periods resulted primarily from funding our efforts in business planning, research and development, acquiring intellectual property rights, recruiting management and technical staff and raising capital.
Net cash used in investing activities was $0.6 million in 2002 and $5.9 million in 2003. The use of cash in each period resulted primarily from the purchase of research and development equipment and, additionally in 2003, restricted cash of $2.3 million related to a lease for our new facility in Cambridge, Massachusetts. We estimate that we will spend $2.9 million to complete the build out of this new facility in the first half of 2004.
Net cash provided by financing activities was $17.3 million in 2002 and $26.4 million in 2003. The net cash provided by financing activities resulted primarily from the sale of convertible preferred stock of $17.3 million in 2002 and $27.5 million in 2003. In 2003, we borrowed $2.1 million under an equipment line of credit and repaid a $3.0 million note assumed in the Alnylam Europe acquisition.
On March 11, 2004, we entered into a collaboration and license agreement with Isis providing for an upfront license payment to Isis of $5.0 million, of which $3.0 million was paid upon the signing of the agreement and the remaining $2.0 million is due on January 3, 2005. We also agreed to make royalty and milestone payments to Isis based on any products that we or a collaborator develop utilizing Isis intellectual property. In addition, we agreed to pay to Isis a percentage of some fees from strategic collaborations we enter into that include access to the Isis intellectual property. In connection with the agreement, Isis purchased 1,666,667 shares of our Series D preferred stock for $10.0 million. Isis also agreed to pay us a license fee, royalty and milestone payments on any product developed by Isis or a collaborator that utilizes our intellectual property.
During 2003, we secured an equipment line of credit to finance equipment purchases of up to $2.5 million. The borrowings bore interest at 0.25% over the prime rate of interest plus an additional 8% due at the end of the term, which was recorded as additional interest expense over the term of each borrowing. The borrowings were repayable over 42 months. The line of credit was collateralized with all of our assets other than intellectual property. As of December 31, 2003, we had an aggregate of $1.9 million outstanding under this facility. On March 31, 2004, we repaid all amounts outstanding under this facility with proceeds from a borrowing under our new line of credit described below and terminated this facility.
On March 31, 2004, we entered into an equipment line of credit to finance equipment purchases of up to $10 million. The borrowings bear interest at 3% over the prime rate of interest plus an additional 11.5% due at the end of the term of each borrowing. The borrowings are repayable over at least a 48-month period from the date of each borrowing and are collateralized by the assets financed.
Based on our current operating plan, we believe that the proceeds from this offering, together with our existing resources, will be sufficient to fund our planned operations through at least the end of 2005, during which time we expect to extend the capabilities of our product engine, initiate development of a product for the treatment of wet AMD and continue to prosecute patent applications and otherwise build and maintain our patent portfolio. However, we may require significant additional funds earlier than we currently expect in order to develop and commence clinical trials for any product candidates we identify.
42
Table of Contents
We expect to seek additional funding through collaborative arrangements and public or private financings. Additional funding may not be available to us on acceptable terms or at all. In addition, the terms of any financing may adversely affect the holdings or the rights of our stockholders. For example, if we raise additional funds by issuing equity securities, further dilution to our existing stockholders may result. If we are unable to obtain funding on a timely basis, we may be required to significantly curtail one or more of our research or development programs. We also could be required to seek funds through arrangements with collaborators or others that may require us to relinquish rights to some of our technologies or product candidates that we would otherwise pursue.
Even if we are able to raise additional funds in a timely manner, our future capital requirements may vary from what we expect and will depend on many factors, including the following:
| • | our progress in demonstrating that siRNAs can be active as drugs; | |
| • | our ability to develop relatively standard procedures for selecting and modifying siRNA drug candidates; | |
| • | progress in our research and development programs, as well as the magnitude of these programs; | |
| • | the timing, receipt, and amount of milestone and other payments, if any, from present and future collaborators, if any; | |
| • | our ability to establish and maintain additional collaborative arrangements; | |
| • | the resources, time and costs required to successfully initiate and complete our preclinical and clinical trials, obtain regulatory approvals, protect our intellectual property and obtain and maintain licenses to third-party intellectual property; | |
| • | the cost of preparing, filing, prosecuting, maintaining, and enforcing patent claims; and | |
| • | the timing, receipt and amount of sales and royalties, if any, from our potential products. | |
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements or relationships with unconsolidated entities or financial partnerships, such as entities often referred to as structured finance or special purpose entities.
Contractual Obligations and Commitments
Set forth below is a description of our contractual cash obligations as of December 31, 2003.
| 2005 | 2008 | |||||||||||||||||||
| Less than | through | through | After | |||||||||||||||||
| Contractual Obligations | Total | One Year | 2007 | 2009 | 2009 | |||||||||||||||
| ($ in thousands) | ||||||||||||||||||||
| Operating lease obligations | $ | 15,246 | $ | 1,637 | $ | 6,092 | $ | 4,086 | $ | 3,431 | ||||||||||
| Short and long-term debt | 2,123 | 645 | 1,478 | — | — | |||||||||||||||
| Consulting agreements | 360 | 144 | 216 | — | — | |||||||||||||||
| Total contractual cash obligations | $ | 17,729 | $ | 2,426 | $ | 7,786 | $ | 4,086 | $ | 3,431 | ||||||||||
On March 15, 2004, we entered into an agreement with a contractor for the build out of our new facility in Cambridge, Massachusetts. The contract contains a guaranteed maximum price of $5.4 million, $0.8 million of which we had paid as of March 31, 2004 and the remainder of which we expect to pay by June 30, 2004. As part of the lease agreement that we entered into with the landlord of this facility, the landlord will reimburse us for up to approximately $3.0 million of certain of the costs of the build out.
43
Table of Contents
Recently Issued Accounting Pronouncements
In January 2003, the FASB issued Interpretation No. 46,Consolidation of Variable Interest Entities, an interpretation of Accounting Research Bulletin No. 51, or FIN 46. FIN 46 requires existing unconsolidated variable interest entities to be consolidated by their primary beneficiaries if the entities do not effectively disperse risks among parties involved. Variable interest entities that effectively disperse risk will not be consolidated unless a single party holds an interest or combination of interests that effectively recombines risks that were previously dispersed. FIN 46 also requires enhanced disclosure requirements related to variable interest entities. FIN 46 applies immediately to variable interest entities created after January 31, 2003, and to variable interest entities in which an enterprise obtains an interest after that date. It applies in the first fiscal year or interim period ending after December 15, 2003 to variable interest entities in which an enterprise holds a variable interest that it acquired before February 1, 2003. We do not expect our adoption of FIN 46 to have a material impact on our financial statements.
In May 2003, the FASB issued SFAS No. 150,Accounting for Certain Financial Instruments with Characteristics of both Liabilities and Equity, or SFAS No. 150. SFAS No. 150 requires that certain financial instruments, which under previous guidance were accounted for as equity, must now be accounted for as liabilities. The financial instruments affected include mandatorily redeemable stock, certain financial instruments that require or may require the issuer to buy back some of its shares in exchange for cash or other assets and certain obligations that can be settled with shares of stock. SFAS No. 150 is effective for all financial instruments entered into or modified after May 31, 2003 and must be applied to our existing financial instruments effective July 1, 2003, the beginning of the first fiscal period after June 15, 2003. The adoption of SFAS No. 150 did not have a material impact on our financial condition or results of operations.
Quantitative and Qualitative Disclosures about Market Risk
We are exposed to market risk related to changes in interest rates. Our current investment policy is to maintain an investment portfolio consisting mainly of U.S. money market and government-grade securities, directly or through managed funds, with maturities of one year or less. Our cash is deposited in and primarily invested through highly rated financial institutions in North America, primarily in money market funds, as of December 31, 2003. Our short-term investments are subject to interest rate risk and will fall in value if market interest rates increase. If market interest rates were to increase immediately and uniformly by 10% from levels at December 31, 2003, we estimate that the fair value of our investment portfolio would decline by an immaterial amount. While our cash and investment balances will increase upon completion of the offering described by this prospectus, we will have the ability to hold our fixed income investments until maturity, and therefore we would not expect our operating results or cash flows to be affected to any significant degree by the effect of a change in market interest rates on our investments.
44
Table of Contents
BUSINESS
Overview
We are a biopharmaceutical company seeking to develop and commercialize new drugs that work through a recently discovered system in cells known as RNA interference, or RNAi. We believe that drugs that work through RNA interference, or RNAi therapeutics, have the potential to become a major class of drugs, like small molecule, protein and antibody drugs. Using our intellectual property and the expertise we have built in RNAi, we are developing a set of biological and chemical methods and know how that we expect to apply in a systematic way to develop RNAi therapeutics for a variety of diseases. We refer to these methods and their systematic application as our “product engine”. Using our current capabilities, we have initiated programs to develop RNAi therapeutics that will be administered directly to diseased parts of the body, which we refer to as Direct RNAi therapeutics. We believe there are multiple opportunities for Direct RNAi therapeutics. Our current Direct RNAi programs are focused on an eye disease known as age-related macular degeneration, or AMD, and on Parkinson’s disease, or PD. We expect to initiate a clinical trial for our lead AMD product candidate in 2005. We are also working to extend our capabilities to enable the development of RNAi therapeutics that travel through the blood stream to reach diseased parts of the body, which we refer to as Systemic RNAi therapeutics. We believe Systemic RNAi will be used to treat a broad range of diseases.
RNAi is a recently discovered natural mechanism for selectively silencing genes. Genes provide cells with coded instructions for making proteins, and silencing a gene refers to stopping or reducing production of the protein specified, or encoded, by that gene. Our goal is to develop new drugs that use the RNAi mechanism to selectively silence genes encoding proteins that play harmful roles in disease. We intend to develop drugs based on a type of molecule known as small interfering RNA, or siRNA. siRNAs are the molecules within cells that directly trigger RNAi. We expect that our RNAi therapeutics will consist of chemically modified siRNAs designed to silence specific genes. Given the recent availability of the base sequence of the entire human genome, RNAi therapeutics can be designed, in theory, to silence any gene that encodes a protein involved in disease, even if currently this protein cannot be adequately controlled by conventional drugs.
We believe that we have a strong intellectual property position relating to the development and commercialization of siRNAs as therapeutics, consisting of:
| • | a concentration of intellectual property rights claiming fundamental features of siRNAs and their use as therapeutics, which includes our ownership of, or exclusive rights to, several issued patents and pending patent applications; | |
| • | a broad portfolio of intellectual property relating to chemical modifications of siRNAs, including over 150 patents licensed from Isis Pharmaceuticals, Inc.; and | |
| • | a number of pending patent applications claiming siRNAs directed to specific targets as treatments for particular diseases. | |
We have filed or licensed over 200 patents and patent applications in the RNAi field.
Our goal is to develop and commercialize RNAi therapeutics. To access the substantial funding and expertise required to develop and commercialize RNAi therapeutics, we intend to form strategic collaborations with pharmaceutical companies. In the collaborations we form in the near term, we expect to take the lead role in discovery and early preclinical development of specific RNAi therapeutics, and to share responsibilities with our collaborators in later-stage development and commercialization of these RNAi therapeutics. We expect that our collaborators will provide us with significant funding for the work we perform, access to their development and commercial capabilities, and a share of revenues from products we originate. Our first such alliance is with Merck & Co., Inc. Over time, as we expand our capabilities and resources, we expect the nature of the collaborations we form to evolve, so that we take on progressively more responsibility for development and commercialization of products we originate, and retain a greater share of the revenues these
45
Table of Contents
Our scientific founders were among the first scientists to publish significant discoveries in the field of RNAi. In addition, we have assembled an experienced management team to implement our business plan. Members of our management team have led discovery, development and commercialization programs for a number of marketed drugs, including Aranesp, Neulasta, Angiomax and Velcade.
RNA Interference
RNAi is a recently discovered mechanism that occurs naturally within cells and selectively silences the activity of specific genes. Genes provide cells with instructions for producing proteins. Proteins perform many of the vital functions of the cell and of the human body. Although the roles they play are generally beneficial, in certain circumstances, proteins can be harmful. Many human diseases are caused by the inappropriate behavior of proteins. A particular protein may, for example, be present in too great a quantity, be too active or appear in the wrong place or at the wrong time. In these circumstances, the ability to stop or reduce production of the protein by selectively silencing the gene that directs its synthesis could be very beneficial towards the treatment of the disease.
Beginning in 1999, our scientific founders were the first to describe and provide evidence that the RNAi mechanism occurs in mammalian cells and that its immediate trigger is a type of molecule known as small interfering RNA, or siRNA. They showed that laboratory-synthesized siRNAs could be introduced into the cell and suppress production of specific target proteins. Because it is possible, in theory, to design and synthesize siRNAs specific for any gene of interest, we believe that RNAi therapeutics have the potential to become a broad new class of drugs.
How RNA interference Works
RNA is a crucial intermediate in the process by which the cell uses inherited genetic information. This information is passed from one generation to the next in the form of genes, which are made of a substance known as deoxyribonucleic acid, or DNA. Generally, each gene contains the instructions that tell the cell how to make one specific protein. These instructions are in a coded form. The code is based on the four different chemical building blocks from which DNA is made, usually designated by the first letters of their chemical names, A, C, G and T. It is the sequence in which these building blocks, or bases, occur in a gene that tells the cell what protein to make. Most gene sequences are thousands of bases long, and the variety possible in such long sequences allows the cell to produce a large number of different proteins.
One very important property of DNA is that it is double-stranded, consisting of two separate strands intertwined around each other in a double helix. The two strands are held together by base pairs that form between bases on the opposite strands. Strict rules govern the formation of these base pairs: an A on one strand can pair with a T on the other, and a G can pair with a C, but no other pairings are allowed. The double-stranded nature of DNA and the strict rules governing base-pairing are fundamental to ensuring that genetic information is copied accurately when it is handed down from one generation to the next.
Base-pairing rules are also fundamental to the process by which the cell uses, or expresses, genetic information to make a protein. To initiate this process, the cell makes a working copy of the gene that encodes the protein. This working copy is made not of DNA but of a closely related substance called ribonucleic acid, or RNA. The working copy is known as messenger RNA, or mRNA. Unlike DNA, mRNA has only one strand. However, the application of base-pairing rules during synthesis of this strand ensures that the sequence of bases in mRNA accurately reflects the base sequence, and thus the genetic information, in the gene being copied. This mRNA then associates with the cell’s protein synthesis machinery, where it directs synthesis of a protein in such a way that the structure of the protein is directly determined by the sequence of bases in the mRNA, and thus in the gene. The protein specified by a particular gene or mRNA is said to be encoded by that gene or mRNA. When this protein is made, the gene is said to be active or expressed.
46
Table of Contents
Although many RNA molecules, like mRNA, are single-stranded, RNA is capable of forming double-stranded molecules analogous to those formed by DNA. When it does so, base-pairing rules apply. As a result, only RNA molecules with complementary sequences can form double-stranded structures. Every base on one strand has to line up with its permitted base-pair partner on the other strand, otherwise the double-stranded structure will be unstable.
Double-stranded RNA, or dsRNA, is crucial to the phenomenon of RNAi. A particular type of dsRNA interferes with the activity of specific genes by triggering the breakdown of mRNAs copied from these genes, preventing production of the proteins they encode. Selection of mRNAs for breakdown is driven by base-pairing between the target mRNAs and the separated strands of the dsRNA. Thus, the mRNAs selected for breakdown are those which contain base sequences identical to base sequences in one strand of the dsRNA. As a result, RNAi leads to selective silencing of specific genes with little impact on other genes whose mRNAs do not share base sequences with the dsRNA.
In nature, the cell initiates RNAi by cutting longer dsRNAs into smaller dsRNA pieces that have 25 or fewer base pairs. These shorter dsRNAs are known as small interfering RNAs, or siRNAs. siRNAs are double-stranded along most of their length but have unpaired bases, or overhangs, at each end, which are important for their activity. siRNAs are the molecules that actually trigger RNA interference. They do so by a process that has three main steps as shown in the figure below.
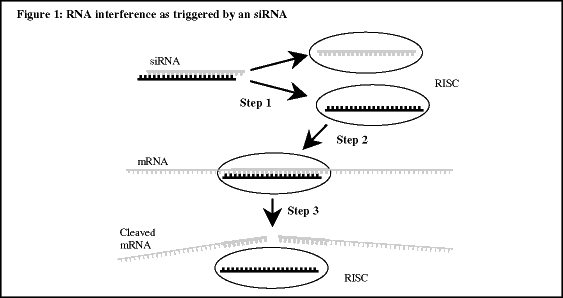
| Step 1. | siRNAs associate with several proteins to form an assembly known as the RNA-induced silencing complex, or RISC. The two strands of the siRNA become separated as the RISC is formed, so that RISC contains an unpaired single-stranded RNA. | |
| Step 2. | The RISC then looks for mRNA molecules that contain base sequences complementary to the single-stranded RNA it contains — that is, sequences within the mRNA whose bases can pair up exactly, using base-pairing rules, with the bases in the single-stranded RNA. | |
| Step 3. | Once this pairing occurs, the RISC complex cuts the mRNA into two separate pieces at the base-paired region, destroying its ability to direct protein synthesis. The RISC complex is then available to cut additional mRNA molecules that contain the appropriate base sequence. |
Repetitive cycles through steps two and three lead to catalytic degradation of mRNAs that contain a sequence complementary to the siRNA strand in the RISC. The ability of each RISC complex to cut multiple mRNA molecules consecutively in a catalytic manner is one of the reasons why we believe RNAi will be effective at silencing gene activity.
47
Table of Contents
Opportunity for Therapeutics Based on RNAi
In May 2001, one of our scientific founders published the first scientific paper demonstrating that the siRNAs required to trigger RNA interference need not be generated inside the cell. Instead, siRNAs can be synthesized in the laboratory using chemical or biochemical methods and introduced into cells to silence the activity of a specific gene. As a result of the human genome project, complete base sequences are available for most human genes. With the sophisticated bioinformatics tools that were developed in conjunction with the genome project, it is possible to scan through the gene that encodes a particular protein and select base sequences that are of the appropriate length for siRNAs and unique to that gene. Several siRNAs targeted to the gene of interest can then be synthesized. Each synthesized siRNA will contain a sequence capable of base-pairing exactly with a short stretch of the sequence of the mRNA copied from the target gene. The synthetic siRNAs can then be tested to determine whether they silence the activity of this gene and suppress the synthesis of the protein it encodes.
The use of siRNAs has been broadly adopted by academic and industrial researchers for the fundamental study of the function of genes. Important information about the function of a gene can often be deduced by suppressing, or knocking-down, its activity and examining the effect this has on the behavior of a cell or animal. There are now many examples in which such suppression of gene activity has been achieved, in whole or in part, using synthetic siRNAs. In just a few years after siRNAs were discovered, they have become the tools of choice for the selective knock-down of gene function by research scientists, and have largely displaced other methods previously used for this purpose. Reflecting this, siRNAs are a growing portion of the market for research reagents and related products and services.
One important application of such knock-down studies is to confirm the role of a particular gene or protein in a disease, a process often referred to as target identification or target validation. If silencing a gene with an siRNA leads to improvements in disease symptoms in an experimental disease model, this implies that the target gene or protein plays an important role in the disease. It also implies that the siRNA that suppresses the gene in the model system may be a useful starting point for the development of a drug. We believe that it will be possible to develop these siRNAs into potent and specific drugs.
Broad Potential of siRNAs as Therapeutics
The success of siRNAs in silencing gene activity in experimental systems suggests that siRNAs could potentially be developed into a broad class of human therapeutics. We believe this new class of drugs has the potential to become a major class of drugs because RNAi therapeutics could offer the following benefits:
| • | Ability to treat a broad range of diseases. Given the availability of the base sequence of the entire human genome, in theory, it could be possible to design siRNAs to suppress the production of virtually any human protein whose presence or activity causes disease. This suggests that RNAi therapeutics could potentially be used to treat a broad range of diseases. | |
| • | Ability to target proteins that cannot be targeted effectively by existing drug classes. Many proteins that play important roles in disease cannot be targeted effectively with small molecules and therapeutic proteins, including monoclonal antibodies. These proteins are commonly referred to as non-druggable targets. In the case of small molecule drugs, many proteins are non-druggable because it has proved difficult to synthesize drug candidates with appropriate specificity, potency and safety. In the case of protein drugs, the range of available targets is limited to targets outside the cell. These limitations on small molecule and protein drugs should not apply to siRNAs, which, in theory, can be synthesized to target any gene in the genome. Therefore, we believe RNAi therapeutics will be able to target proteins that small molecule and protein drugs cannot currently target. | |
| • | Inherently potent mechanism of action. One molecule of siRNA could potentially do the work of thousands of molecules of conventional drugs. With conventional drugs, one drug molecule is typically required for every protein molecule whose activity needs to be blocked. Accordingly, to block several thousand protein molecules, several thousand drug molecules are required. In contrast, a single siRNA molecule can potentially block the synthesis of many protein molecules. This is because each siRNA |
48
Table of Contents
| within a RISC complex can trigger destruction of multiple mRNA molecules, each of which could otherwise direct the synthesis of many protein molecules. This inherent potency of the RNAi mechanism suggests a potentially high degree of potency for RNAi therapeutics. | ||
| • | Simplified discovery of drug candidates. Identification of small molecule and protein drug candidates typically requires screening of a large number of potential candidates to find prospective leads. These leads must then undergo significant optimization in order to become drug candidates. Particularly in the case of small molecule drug candidates, the optimization procedure can be very challenging, and has to be almost entirely repeated for each candidate. Identification of siRNA drug candidates has the potential to be much simpler and take considerably less time because, in theory, it will involve relatively standard processes that can be applied in a similar fashion to many successive product candidates. |
For these potential benefits of siRNA drugs to be realized, it will be necessary to create chemically-synthesized siRNAs that are potent, specific, stable and safe and also capable of reaching the appropriate tissues and cells. The incorporation of such properties into siRNAs is the focus of our product engine.
Our Business Strategy
Our strategy is to use our intellectual property and expertise in RNAi to develop and commercialize RNAi therapeutics. The key elements of our business strategy are as follows:
| • | Pursue product opportunities in a phased approach based on the capabilities of our product engine.With the goal of accelerating the development of RNAi therapeutics, we are implementing a phased approach to product development. Using the current capabilities of our product engine, we will focus initially on the development of Direct RNAi therapeutics. We believe there are multiple opportunities for Direct RNAi therapeutics. As we extend the capabilities of our product engine, we will initiate development of Systemic RNAi products. We believe this approach will allow us to initiate product development activities in three main phases. |
| — | Direct RNAi therapeutics. We intend to utilize the current capabilities of our product engine by focusing our efforts on developing RNAi therapeutics that can be administered directly to diseased parts of the body, such as the eye or the brain. As part of this phase, we have initiated Direct RNAi programs focused on AMD and PD. | |
| — | Systemic RNAi therapeutics for liver-based diseases. As we extend the capabilities of our product engine, we intend to develop RNAi therapeutics for liver-based diseases. We intend to focus on the liver because it is central to several important diseases and also appears, on the basis of early preclinical evidence, to take up siRNAs more readily than other tissues. | |
| — | Systemic RNAi therapeutics for other diseases. As a third phase of our product development strategy, we will seek to develop RNAi therapeutics that exert their effects in tissues other than the liver. We believe achievement of this objective could permit us to develop Systemic RNAi drugs for a broad range of diseases, such as cancer and autoimmune diseases. |
| • | Maintain a strong intellectual property position in the RNAi field.We believe we have a strong intellectual property position relating to the development and commercialization of siRNAs as therapeutics. To build upon our existing intellectual property position, we are focusing on patent and patent applications covering: |
| — | fundamental aspects of the structure and uses of siRNAs, including their use as therapeutics; | |
| — | chemical modifications to siRNAs that improve their suitability for therapeutic uses; and | |
| — | siRNAs directed to specific targets as treatments for particular diseases. |
| In March 2004, we entered into a collaboration with Isis that provides us with rights to over 150 issued patents covering fundamental aspects of double-stranded RNA therapeutics designed to work through a RNAi mechanism and chemical modifications that may enhance their pharmaceutical |
49
Table of Contents
| properties. Isis agreed that it will not grant corresponding rights to any third party for any dsRNA products designed to work through a RNAi mechanism, except in the context of a collaboration in which Isis plays an active role. |
| • | Capitalize on our expertise in RNAi and our intellectual property position to gain access to additional resources to develop and commercialize RNAi therapeutics. We believe that we can use our expertise and the strength of our intellectual property to drive the formation of strategic alliances that will provide us with significant funding and access to important additional resources. We expect these resources to include significant additional capital as well as expertise in the development, manufacturing and commercialization of novel therapeutics. We intend to take an active role in these alliances, including maintaining certain development and commercialization rights. We also intend to use our early alliances to expand our own capabilities so that in the future we will be able to develop and commercialize our therapeutic products independently. To date, we have entered into one such alliance with Merck, the goal of which is to jointly develop advanced RNAi technology and therapeutics. | |
| • | Leverage our intellectual property position by licensing our technology to generate revenues.We intend to license our intellectual property to third parties for the development and commercialization of RNAi therapeutics outside our areas of strategic focus through our InterfeRxProgram. We expect to receive license fees, annual maintenance fees, milestone payments and royalties on sales of any resulting RNAi therapeutics. We also intend to grant further licenses to our intellectual property for the development and commercialization of research reagents and services. To date, we have granted research reagents licenses to Invitrogen Corporation and Cell Signaling Technology, Inc. and have granted research service licenses to Invitrogen, Cenix Bioscience AG and RNAx GmbH. We grant these licenses in return for an initial license fee, annual renewal fees and royalties on sales of siRNA research reagents and services. |
Alnylam Product Engine
To realize the potential of RNAi therapeutics as a broad new class of drugs, we are developing capabilities that we can apply to any specific siRNA in a relatively standard fashion to endow it with drug-like properties. We use the term product engine to describe these capabilities because we believe they will enable us to develop many products across a variety of therapeutic areas. The concept for our product engine is that it will provide a systematic approach to identifying RNAi drug candidates with the following steps:
| • | Sequence selection. Using sophisticated bioinformatics tools we scan through the entire sequence of a target mRNA to identify sequences that are unique to that mRNA and have few closely similar sequences in other mRNAs. From these unique sequences we derive a list of potential siRNAs that would match up exactly with the target mRNA and not with any other mRNAs. We narrow this list down further by applying filters for other important properties, such as the presence of stabilizing sequence motifs that we have identified. This provides us with a shorter list of siRNAs, each of which we then synthesize for experimental evaluation. | |
| • | Potency selection.The siRNAs synthesized in the sequence selection step are tested in cell culture systems to compare their potencies in suppressing production of the target protein. | |
| • | Stabilization by chemical modification. Each of the most potent siRNAs is tested to identify the sites within its structure where it is most vulnerable to attack by enzymes known as ribonucleases that could degrade the siRNA. A minimal set of chemical modifications is then introduced into the siRNA to protect these vulnerable sites, and the modified siRNA is tested to confirm its stability and that it has retained activity against the target mRNA. | |
| • | Improvement of biodistribution by conjugation of additional chemical groups. The stabilized siRNA is further modified by the addition, or conjugation, of one or more chemical groups designed to improve uptake of the siRNA into cells and, if desired, to prolong the time it circulates in the blood. |
50
Table of Contents
We expect the output of this process to be RNAi drug candidates that are potent against and specific for a particular target, are appropriately stable and are able to penetrate cells of target tissues. Moreover, we expect this process for finding suitable drug candidates will be simpler, faster and more productive than the corresponding process for small molecule and protein drug candidates. Therefore, we believe that any significant success in the development of our product engine will place us in a position to pursue multiple therapeutic opportunities.
Progress in the Development of Product Engine
We have made considerable progress in establishing capabilities suitable for Direct RNAi drugs, and believe on the basis of early results that we will be able to develop capabilities suitable for Systemic RNAi drugs. In judging our progress, we focus on five key attributes that are important for any drug molecule: potency, specificity, stability, safety and biodistribution.
Potency
The more potent a drug is, the lower the dose required for a therapeutic effect, and the lower the risk of side effects. Because siRNAs harness the catalytic activity of a natural mechanism to block the synthesis of undesirable proteins, we expect siRNA drugs to be potent at this task. Current data that we and others have generated support this expectation. We have designed and synthesized siRNAs that are highly effective at low concentrations in suppressing target protein production in cultured cells. A frequently used measure of the potency of potential drug candidates is the parameter known as IC50, which is the concentration of the drug candidate required to reduce the activity of the target protein by 50% of its normal level. The lower the IC50 value, the more potent the drug candidate. We have made siRNAs that suppress target protein production in cultured cells with IC50 values similar to, or lower than, the values typically observed for other types of drugs in analogous experiments. Further, the siRNAs we have made and tested in cell culture experiments for one of our programs have demonstrated greater potency than siRNAs others have tested against the same target in published experiments. In this program, we have also demonstrated with one siRNA that suppression of gene activity continued for at least seven days after cells were exposed to low concentrations of the siRNA on the first day. Although these observations need to be extended to experiments in animals and then to clinical testing in humans, they lead us to believe that we will be able to identify siRNAs with potency that is sufficient for therapeutic use.
Specificity
To maximize beneficial effects and minimize harmful ones, a drug should be highly selective for its intended target and have minimal activity against unintended targets. For siRNAs, careful selection of an appropriate base sequence can provide a high degree of specificity for the target mRNA. Some evidence has been reported of off-target effects with certain siRNAs, meaning that these siRNAs affected mRNAs other than their intended targets. However, we believe that such effects can be minimized by the use of sophisticated bioinformatics tools to improve selection of siRNA sequences and by judicious incorporation of chemical modifications at appropriate positions within the siRNA molecule. Therefore, we believe we will be able to identify siRNA molecules that are sufficiently specific to the target mRNAs.
Stability
To be effective, drugs must be stable in the body long enough to reach the tissues in which their effects are required and then to exert those effects. To endow siRNA molecules with appropriate stability, we are introducing a series of chemical modifications into siRNAs to protect them against enzymes called ribonucleases that would otherwise degrade them. We have also identified certain sequence motifs that help to stabilize the siRNAs that contain them, and which we can incorporate by design into the siRNAs we make. We use various experimental systems to assess the impact of sequence selection or chemical modifications on the stability of siRNAs. These systems include incubation of modified siRNAs at body temperature in samples of animal or human serum or in extracts made from animal tissues, such as the liver or the eye. We also assess the stability of siRNAs by injecting them into animals and analyzing samples of serum or tissues
51
Table of Contents
Safety
All drugs carry the risk of side effects. These effects may have a variety of causes, including incomplete specificity of the drug for its intended target or an adverse interaction between the drug and the body’s systems for monitoring and disposing of foreign materials. In the case of RNAi drugs, our ability to choose siRNAs that are potent and selective for their targets should help to minimize side effects caused by lack of specificity. Moreover, we are deliberately minimizing the number of chemical modifications we are introducing into siRNAs to enhance stability and other properties and believe that this will help minimize potential adverse effects on the body’s disposal systems.
Because siRNAs are dsRNAs, one further potential side effect is activation of the so-called interferon response, which can be provoked by dsRNAs and causes influenza-like symptoms. There is published evidence, however, that dsRNAs must be at least 30 base pairs long to trigger the interferon response. We expect that siRNAs we develop as drugs will typically have 25 or fewer base pairs.
Biodistribution
Biodistribution refers to the ability of drugs to reach different tissues within the body and to enter cells within these tissues. Because RNA molecules are typically unable to enter cells without the use of special delivery agents, biodistribution is considered to be one of the key issues to be addressed before siRNAs can be developed into drugs. We believe we have made considerable progress addressing biodistribution for siRNAs. For example, we have recently demonstrated that:
| • | The attachment of a chemical structure to one end of an siRNA molecule enables that molecule to enter cells without any other delivery agent. To demonstrate entry of the modified siRNA into cells, we incubated cultured cells with various concentrations of the modified siRNA or of an siRNA having the same base sequence but lacking the crucial modification. Activity of the target protein was reduced to about 20% of its normal level in the cells incubated with the modified siRNA, but was essentially unaffected in the cells incubated with the siRNA lacking the crucial modification. We refer to this modification as the permeation group, and use the term permeating siRNAs to describe siRNAs to which we have attached the permeation group. | |
| • | A permeating siRNA remains in the circulation of a mouse and a rat for longer than a similar siRNA that has not been modified with the permeation group. In a test to measure the lifetime of injected siRNA, we demonstrated that a permeating siRNA was detectable in the circulation of a mouse and a rat 12 hours after injection, whereas an unmodified siRNA was barely detectable after one hour. | |
| • | A permeating siRNA can make its way to the liver after injection into the tail vein of a mouse. We made extracts from the livers of mice that had been injected with various siRNAs and used the ribonuclease protection assay to analyze these extracts for the presence of siRNA. siRNAs were only observed in liver extracts if the injected siRNAs had been modified by attachment of the permeation group. No siRNAs were observed in liver extracts from mice that had not been injected or had been injected with siRNAs lacking the permeation group. | |
| • | Liver concentrations of a target mRNA and protein can be measured in the same liver extracts that contain a permeating siRNA. We have designed experiments that will enable us to measure liver concentrations of a target mRNA and protein in the same liver extracts that contain a permeating | |
52
Table of Contents
| siRNA. We are in the process of performing experiments in live animals to determine whether our permeating siRNAs can reduce the levels of target mRNA and protein in the liver. |
These experiments lead us to believe that we will be able to identify siRNAs that have appropriate biodistribution for use as drugs.
Our Development Programs
Given our progress in developing our product engine we have begun work on Direct RNAi drug candidates and believe that we will also be able to move forward with Systemic RNAi drug candidates in the future. Using the current capabilities of the product engine, we have initiated two programs to identify specific siRNAs for potential further development as Direct RNAi drug candidates. Our two current programs are focused on age-related macular degeneration and Parkinson’s disease.
Age-Related Macular Degeneration
Market Opportunity
AMD can cause severe deterioration of vision and may ultimately cause blindness. The National Eye Institute estimates that over 1.6 million adults over 50 in the United States suffer from advanced AMD. The siRNAs we are exploring would treat wet AMD, a subtype of AMD often associated with severe vision loss. According to AMD Alliance International, approximately 200,000 new cases of wet AMD are diagnosed in North America each year and approximately 500,000 new cases of wet AMD are diagnosed worldwide each year.
The macula is the central area of the retina, and is the area most responsible for visual acuity, or sharpness of vision. Macular degeneration is a complex process involving the formation of new blood vessels immediately behind the retina. These newly formed vessels tend to be relatively fragile, and to leak blood and fluid into the surrounding tissue. The presence of leaked fluids and new blood vessels behind the retina physically disrupts the integrity of the macula, resulting in blurring of central vision, and ultimately blindness.
Current Treatments
Current treatments for AMD are limited in their effectiveness and there is only one drug approved for treatment of wet AMD, Visudyne. Visudyne is used in a two-step process known as photodynamic therapy whose goal is to seal newly formed blood vessels behind the retina so that they neither grow nor leak. However, Visudyne is only approved for the treatment of certain subtypes of wet AMD, and its beneficial effects are frequently modest and of limited duration. The only current alternative to photodynamic therapy is treatment with a laser to burn out newly formed blood vessels. Unfortunately, this treatment can also cause irreversible damage to the retina, resulting in significant blind spots, and its beneficial effects appear to be of limited duration.
Several companies are currently pursuing a new approach to treat wet AMD by blocking the activity of a protein known as vascular endothelial growth factor, or VEGF. There is evidence that VEGF promotes both the growth and the leakage of new blood vessels behind the retina in AMD. Eyetech Pharmaceuticals, Inc. and Genentech, Inc. each have investigational drugs, Macugen and Lucentis, respectively, directed against VEGF in advanced stages of clinical testing for the treatment of wet AMD. These drugs are intended to block the activity of VEGF by binding to it. Two other companies are in the early stages of developing siRNA products to block VEGF activity. One of these products is intended to limit production of VEGF by suppressing the activity of the VEGF gene. The other is intended to limit production of a protein known as VEGF-receptor, which is the protein on cell surfaces that VEGF must bind to in order to exert its effects.
Alnylam Program
We are developing a RNAi therapeutic, which we refer to as VEGF Production Inhibitor, for the treatment of wet AMD. We believe that a RNAi therapeutic that suppresses VEGF gene activity, thereby cutting off VEGF production, could have significant advantages over drugs that bind to VEGF once it has
53
Table of Contents
Parkinson’s Disease
Market Opportunity
PD is a disorder of the nervous system that, according to the American Parkinson Disease Association, afflicts more than 1.5 million people in the United States and, according to the World Health Organization, afflicts approximately four million people worldwide. In its early stages PD is characterized by uncontrollable tremors that gradually increase in severity. As the disease progresses it can lead to severe loss of mobility and dementia. Industry sources estimate that worldwide sales of drugs for treating PD were approximately $2 billion in 2002.
The symptoms of PD are caused by a shortage of a substance called dopamine in a key region of the brain. Dopamine plays an important role in transmitting signals between nerve cells and in regulating movement. The shortage of dopamine in PD is caused by the death of dopamine-producing cells in a region of the brain known as the substantia nigra.
Current Treatments
Currently approved treatments address the symptoms of PD rather than its cause and largely rely on drugs that replace the function of the missing dopamine. These drugs act in a variety of ways, including mimicking dopamine, boosting the amount of dopamine in the brain, enhancing the activity of dopamine boosters or mimics, slowing down the breakdown of dopamine and compensating for diminished dopamine activity. Over time, however, patients cease to respond to these drugs, and there are no effective treatments for advanced PD.
Alnylam Program
The goal of our program is to develop a RNAi therapeutic that will slow or halt the progress of PD. Instead of replacing the function of missing dopamine in persons with PD, the siRNAs we have begun to explore as potential treatments of PD would attempt to prevent the shortage of dopamine by preventing the death of dopamine-producing cells that causes this shortage. Based on recent scientific findings, we believe that it may be possible to prevent the death of those cells by using an siRNA to suppress production of a protein known as alpha-synuclein. Recent evidence suggests that the presence of excessive amounts of alpha-synuclein in dopamine-producing cells contributes to their death. We have identified siRNAs that suppress the activity of the alpha-synuclein gene in cultured cells. We recently entered into a collaboration with the Mayo Foundation for Medical Education and Research and the Mayo Clinic Jacksonville, which we refer to collectively as the Mayo Clinic, to explore this possibility in animal models of PD.
The collaboration is established with an initial one-year period to explore whether an siRNA could be effective in treating PD and allows for an extension of the collaboration if the results are positive. We expect that any resulting RNAi therapeutic would be a Direct RNAi drug administered using a specialized medical device to infuse the drug into the appropriate region of the brain. Accordingly, we would anticipate collaborating with a manufacturer of such devices to develop appropriate procedures for delivery. We have secured an option for an exclusive license to a patent application filed by the Mayo Clinic relating to the use of siRNAs to suppress production of alpha-synuclein for therapeutic purposes.
54
Table of Contents
Strategic Alliances and Licenses
Strategic Alliances
We intend to form strategic alliances to gain access to the financial, technical, clinical and commercial resources necessary to develop and market RNAi therapeutics. We expect these alliances to provide us with financial support in the form of equity investments, research and development funding, license fees, milestone payments and royalties or profit sharing based on sales of RNAi therapeutics. Our first strategic alliance is with Merck for the development of advanced RNAi technology and therapeutics.
Merck
In September 2003, Alnylam entered into a five-year strategic alliance with Merck to develop advanced RNAi technology and RNAi therapeutics. For technology development, both parties committed to devote significant human resources and expertise to the collaborative development of advanced RNAi technology. Merck will have exclusive rights to use Alnylam’s RNAi technology and the RNAi technology developed jointly under the collaboration solely for the identification and validation of drug targets. Alnylam will have rights to use this technology for all internal research purposes and in collaborations in which the primary purpose is the development of therapeutic products using RNAi. For therapeutics development, Merck agreed to provide Alnylam with twelve proprietary drug targets over the course of the collaboration that have well-validated roles in disease and that appear attractive as potential targets for RNAi therapeutics. Alnylam has the right, but not the obligation, to develop siRNA drug candidates against each target provided by Merck. If Alnylam advances a candidate to a defined point in preclinical development, Alnylam and Merck will then decide whether Alnylam, Merck or the two companies together will proceed with the further development and commercialization of that candidate. For each drug candidate that Merck decides to develop, whether by itself or jointly with Alnylam, Merck will pay Alnylam a fee at the time of its decision, and will also reimburse Alnylam for one-half of the costs we incurred previously on that candidate. If the parties agree to develop a drug candidate together, we will share development expenses and co-promote the products upon terms to be determined by mutual agreement. If it is determined that Merck will develop the drug candidate without Alnylam’s further involvement, Merck will bear all development expenses and will pay Alnylam a royalty on product sales. Likewise, if it is determined that Alnylam will develop the drug candidate without further Merck involvement, Alnylam will bear all development expenses and will pay Merck a royalty on product sales. In that event, Alnylam would retain the right to collaborate with a third party on the development and commercialization of that drug candidate.
In connection with this alliance, Merck made a $2 million cash payment and $5 million equity investment in Alnylam during 2003. Further cash payments are due from Merck in each of 2004 and 2005. Merck will make an additional cash payment and equity investment totaling $7 million upon the first demonstration by Alnylam of RNAi activity in live animals, as measured by agreed upon scientific criteria.
Licenses
We intend to enter into licensing arrangements with third parties to enhance our intellectual property position and to generate revenues from our intellectual property rights. We recently entered into a collaboration and license agreement with Isis under which we obtained rights to a broad portfolio of intellectual property relating to double-stranded RNA therapeutics designed to work through a RNAi mechanism. To generate revenues from our intellectual property rights, we have established our InterfeRx program and our research reagents and services licensing program.
Isis Pharmaceuticals, Inc.
In March 2004, we entered into a collaboration and license agreement with Isis, a leading developer of single-stranded antisense oligonucleotide drugs that target RNA. The agreement further enhances our intellectual property position with respect to RNA-based therapeutics and our ability to develop double-stranded RNA for RNAi therapeutics, and provides us with the opportunity to defer investment in manufacturing technology. Isis granted us licenses to its current and future patents and patent applications
55
Table of Contents
Under the terms of our agreement, we agreed to pay Isis an upfront license fee of $5 million, $3 million of which was paid upon signing of the agreement and the remaining $2 million of which is due on January 3, 2005. We also agreed to pay royalties and milestones to Isis on any products that we or a collaborator develop utilizing Isis intellectual property. In addition, we agreed to pay to Isis a percentage of some fees from strategic collaborations we may enter into that include access to the Isis intellectual property. In conjunction with the agreement, Isis made a $10 million equity investment in Alnylam. Isis also agreed to pay us a license fee, royalty and milestone payment on any product developed by Isis or a collaborator that utilizes our intellectual property. The agreement also gives us an option to use Isis’ manufacturing services for RNA-based therapeutics.
Our agreement with Isis also gives us the exclusive right to grant sub-licenses for Isis technology to third parties with whom we are not collaborating. We may include these sub-licenses in our InterfeRx licenses. If a license includes rights to Isis’ intellectual property, we will share revenues from that license equally with Isis.
If, by January 1, 2008, we or a collaborator have not completed the studies required for an investigational new drug application filing or similar foreign filing for at least one product candidate involving these patent rights, Isis would have the right to grant licenses to third parties for the patents and patent applications licensed to us, thereby making our rights non-exclusive.
InterfeRx Program
Our InterfeRxprogram consists of the licensing of our intellectual property to others for the development and commercialization of RNAi therapeutics relating to specific protein targets outside our areas of strategic focus. We expect to receive license fees, annual maintenance fees, milestone payments and royalties on sales of any resulting RNAi therapeutics. Generally, we do not expect to collaborate with our InterfeRx licensees in the development of RNAi therapeutics, but may do so in appropriate circumstances. To date, we have not entered into any InterfeRx licenses.
Research Reagents and Services
We have granted licenses to our intellectual property for the development and commercialization of research reagents and services, and intend to enter into additional licenses on an ongoing basis. Our target licensees are vendors that provide siRNAs and related products and services for use in biological research. To date, we have entered into research reagent licenses with Invitrogen Corporation and Cell Signaling Technology, Inc. and research service licenses with Invitrogen Corporation, Cenix Bioscience AG and RNAx GmbH. We grant these licenses in return for an initial license fee, annual renewal fees and royalties from sales of siRNA research reagents and services.
Research Collaborations
We may also enter into collaborations with leading research and medical institutions in connection with our development of RNAi therapeutics for specific indications. Our arrangement with the Mayo Clinic related to PD is an example of this type of collaboration.
56
Table of Contents
Mayo Clinic
In October 2003, we entered into a one-year research collaboration with the Mayo Clinic to develop an siRNA that suppresses the expression of alpha-synuclein, which is over-expressed in patients with PD. Under the collaboration, we agreed to identify, synthesize and provide siRNA drug candidates to the Mayo Clinic, which will test these candidates for their efficacy in a series ofin vitroandin vivostudies. We provide research funding to the Mayo Clinic, and have the option to acquire exclusive licenses to background intellectual property held by the Mayo Clinic and inventions arising out of the collaboration.
Patents and Proprietary Rights
We have devoted considerable effort and resources to establish what we believe to be a strong position in intellectual property relevant to RNAi therapeutics. In this regard, we have focused on patents, patent applications and other intellectual property covering:
| • | fundamental aspects of the structure and uses of siRNAs, including their use as therapeutics, and RNAi-related mechanisms; | |
| • | chemical modifications to siRNAs that improve their suitability for therapeutic uses; and | |
| • | siRNAs directed to specific targets as treatments for particular diseases. |
Intellectual Property Related to Fundamental Aspects and Uses of siRNA and RNAi-related Mechanisms
In this category, we include patents and patent applications that claim key aspects of RNAi-related mechanisms. Specifically, we include patents and patent applications relating to targeted cleavage of mRNA directed by RNAi-like oligonucleotides, double-stranded RNAs of particular lengths, particular structural features of these dsRNAs, such as overhanging ends, and uses of these dsRNAs. Our strategy has been to secure rights to the potentially key patents and patent applications covering the fundamental aspects of siRNAs on an exclusive basis where possible or appropriate. The following table lists patents or patent applications to which we have secured rights that we regard as being potentially fundamental for the use of siRNAs as therapeutics.
| Licensor/ | Subject | Priority | ||||||||||
| Patent Owner | Matter | Date | Inventors | Status | Alnylam Rights | |||||||
| Isis Pharmaceuticals | Inactivation of target mRNA | 06/06/1997 | S. Crooke | Issued in the United States, pending in the EU | Exclusive rights for therapeutic purposes related to dsRNAs* | |||||||
| Carnegie Institution of Washington | Double- stranded RNAs to induce RNAi | 12/23/1997 | A. Fire, C. Mello | Issued in the United States, pending elsewhere | Non-exclusive rights for therapeutic purposes | |||||||
| Alnylam | Short double- stranded RNAs as therapeutics | 01/30/1999 | R. Kreutzer, S. Limmer | Granted in the EU, issued in Germany and South Africa, pending in the United States and elsewhere | Owned | |||||||
| Cancer Research Technology Limited | RNAi uses in mammalian cells | 11/19/1999 | M. Zernicka-Goetz, M.J. Evans, D.M. Glover | Pending worldwide | Exclusive rights for therapeutic purposes | |||||||
57
Table of Contents
| Licensor/ | Subject | Priority | ||||||||||
| Patent Owner | Matter | Date | Inventors | Status | Alnylam Rights | |||||||
| Massachusetts Institute of Technology, Whitehead Institute, Max Planck organization** | Mediation of RNAi by siRNAs containing 21-23 base pairs | 03/30/2000 | D.P. Bartel, P.A. Sharp, T. Tuschl, P.D. Zamore | Pending worldwide | Non-exclusive rights for therapeutic purposes** | |||||||
| Max Planck organization | siRNAs with 3’-overhangs as therapeutics | 12/01/2000 | T. Tuschl, S. Elbashir, W. Lendeckel | Pending worldwide | Exclusive rights for therapeutic purposes | |||||||
| Cold Spring Harbor Laboratory | RNAi uses in mammalian cells | 03/16/2001 | D. Beach, G. Hannon | Pending worldwide | Non-exclusive rights for therapeutic purposes | |||||||
| Stanford University | RNAi usesin vivo | 07/23/2001 | M.A. Kay, A.P. McCaffrey | Pending worldwide | Co-exclusive rights for therapeutic purposes | |||||||
| * | Alnylam holds co-exclusive therapeutic rights with Isis. However, Isis has agreed not to license such rights to any third party, except in the context of a collaboration in which Isis plays an active role. |
| ** | Alnylam holds exclusive rights to the interest owned by three of four co-owners. The fourth co-owner, the University of Massachusetts, has licensed its interest separately to third parties. |
We believe that no other company owns a portfolio of such broad and exclusive rights to fundamental siRNA patents and patent applications. In securing these rights, we have focused on obtaining the strongest rights for those intellectual property assets we believe will be most important in providing competitive advantage with respect to RNAi therapeutics. We note in particular the third and sixth patents and patent applications listed in the table above, those covering inventions by Dr. Kreutzer and Dr. Limmer and by Dr. Tuschl and his colleagues. We believe the so-called Kreutzer-Limmer European patent is the only patent so far granted that specifically covers the use of short dsRNAs as therapeutics. Through our acquisition of Alnylam Europe in 2003, we own this patent outright, as well as corresponding patent applications in other countries, including the United States. The pending patent application filed by the Max Planck organization on the invention by Dr. Tuschl and his colleagues, or the Tuschl II patent application, covers a key structural feature of siRNAs, namely the presence of overhangs at the 3’-end of each of the two strands. We have obtained an exclusive license to claims in the Tuschl II patent application uniquely covering the use of RNAi for therapeutic purposes. The application contains claims relating to therapeutic uses of siRNAs with or without concomitant gene therapy. Our exclusive rights are to the claims that do not require concomitant gene therapy.
As indicated in the table, two other RNAi-related patents have already been issued that pre-date the Kreutzer-Limmer patent. These are the Crooke patent and the Fire and Mello patent. The Crooke patent, owned by and licensed from Isis, covers the use of oligonucleotides to promote degradation of target mRNA by enzymes that recognize dsRNAs, such as those involved in RNAi. Under the terms of our license agreement, Isis agreed not to grant licenses under this patent to any other organization for dsRNA products designed to work through a RNAi mechanism, except in the context of a collaboration in which Isis plays an active role. The Fire and Mello patent owned by the Carnegie Institution covers the use of dsRNAs to induce RNAi. The Carnegie Institution has made this patent broadly available for licensing and we, like many companies, have taken a non-exclusive license to the patent for therapeutic purposes. We believe, however, that the Fire and Mello patent does not claim specific structural features of dsRNAs that are important for the biological activity of siRNAs in mammalian cells. These specific features are the subjects of the Kreutzer-Limmer patent and the Tuschl II patent application for which we have secured exclusive rights.
58
Table of Contents
The Kreutzer-Limmer patent was granted in Europe in 2002 and in South Africa in 2003 and is pending in other countries, including the United States. In addition, a German Utility Model covering RNAi composition was branched off the European patent application, and was granted by the German Patent and Trademark Office in 2003. A German Utility Model is a form of patent that is directed only to physical matter, such as medicines, and does not cover methods. The German Utility Model is valid for ten years from the time of the first filing and is thus in effect until 2009. The issuance of the European patent is currently being opposed by several other companies under a provision of European patent law that allows such opposition. It may be several years before the outcome of this opposition is decided by the EPO.
In addition, the EPO is also reviewing what is known as disclaimer practice, a process in which a patent applicant disclaims part of the claimed invention in a patent application in order to overcome prior art. In the Kreutzer-Limmer patent prosecution, the inventors disclaimed coverage of RNAi agents which were between 49 and 25 nucleotides in length. The EPO may determine that disclaimer practice is not allowed under European patent rules. Such a determination could result in the invalidation of the currently allowed claims of the Kreutzer-Limmer patent. In the event that the current granted patent is invalidated, there are pending claims of the patent that do not rely on disclaimer practice. Even if the EPO determines that disclaimer practice is permissible under European patent rules, the Kreutzer-Limmer patent would remain subject to the opposition described in the preceding paragraph.
The other pending patent applications listed in the table either provide further coverage for structural features of siRNAs or relate to the use of siRNAs in mammalian cells. For some of these we have exclusive rights, for others non-exclusive. While we believe these pending patent applications are important, we also believe that access to the Kreutzer-Limmer patent and the Tuschl II patent application will be of particular importance for development and commercialization of RNAi therapeutics, which is why we have secured exclusive positions with respect to these assets. However, because RNAi is a relatively new field, few patents have been issued, and many potentially key patent applications are still pending.
Intellectual Property Related to Chemical Modifications
Over the last fifteen years a large amount of effort has been devoted by academics and other biotechnology companies to two other technologies with the potential to selectively turn off gene activity. These technologies are known as antisense oligonucleotides and ribozymes. Both involve using short DNA or RNA molecules to intercept specific mRNAs so as to reduce production of proteins encoded by these mRNAs. Scientists and companies working on antisense oligonucleotides and ribozymes have developed a variety of chemical modifications that can be applied to short DNA and RNA molecules to endow them with drug-like properties. A number of patents have been issued to these scientists and companies claiming the chemical modifications they have developed. Isis has been very successful in obtaining such patents. We recently entered into a collaboration and license agreement with Isis that provides us with rights to use over 150 issued patents relating to chemical modifications we may wish to incorporate into our RNAi therapeutics. Isis also granted us rights based on future chemistry patent applications filed in the next five years to which it has rights. We believe that access to this intellectual property from Isis could accelerate our development of RNAi therapeutics by enabling us to capitalize on proprietary chemistry developed by Isis instead of designing around this chemistry. Under the terms of our license agreement, Isis agreed not to grant licenses under these patents to any other organization for dsRNA products designed to work through a RNAi mechanism, except in the context of a collaboration in which Isis plays an active role.
In addition to licensing these intellectual property rights from Isis, we are also working to develop our own proprietary modifications that we can apply to siRNAs to endow them with drug-like properties. We have filed a number of patent applications relating to novel chemical modifications that we may apply to siRNAs. We filed these applications relatively recently, and are still evaluating which chemical modifications we may incorporate into siRNA drugs. We may not know for a number of years whether the modifications we use will be patentable or free of patents held by others.
59
Table of Contents
Intellectual Property Related to siRNAs Directed to Specific Targets
We have also filed a number of patent applications claiming specific siRNAs directed to a large number of targets as treatments for specific diseases. We recognize, however, that there may be a significant number of competing applications filed by other organizations on similar siRNAs. Because our subsidiaries, Alnylam Europe and Alnylam U.S., were among the first companies to focus on RNAi therapeutics, we believe that a number of our patent applications may predate competing applications that others may have filed. With respect to specific siRNAs, we believe that the most important patent coverage will ultimately result from demonstrating that particular compositions exert suitable biological and therapeutic effects. Accordingly, we are focused on achieving such demonstrations for siRNAs in key therapeutic areas.
Because the work we and others are performing to develop siRNAs as drugs is at a relatively early stage, and because many patent applications on specific siRNAs are pending but none, to our knowledge, have been issued, we may not know for a number of years whether any siRNA drugs we develop will be patentable and free of patents held by others.
Competition
The pharmaceutical marketplace is extremely competitive, with hundreds of companies competing to discover, develop and market new drugs. We face a broad spectrum of current and potential competitors, ranging from very large, global pharmaceutical companies with significant resources to other biotechnology companies with resources and expertise comparable to our own. We believe that for most or all of our drug development programs, there will be one or more competing programs in other companies. In many cases the companies with competing programs will have access to greater resources and expertise than we do and may be more advanced.
The competition we face can be grouped into three broad categories:
| • | Other companies working to develop RNAi therapeutics; | |
| • | Companies developing technology known as antisense, which, like RNAi, attempts to silence the activity of specific genes by targeting the mRNAs copied from them; and | |
| • | Marketed products and development programs that compete with the drugs we may try to develop. |
Other Companies Working to Develop RNAi Therapeutics
We are aware of several other companies that are working to develop RNAi therapeutics. Some of these companies are seeking, as we are, to develop chemically synthesized siRNAs as drugs. Others are following a gene therapy approach, with the goal of treating patients not with synthetic siRNAs but with genes designed to produce siRNA-like molecules within cells.
Companies working on chemically synthesized siRNAs include Sirna Therapeutics, Inc., Genta, Inc., and Acuity Pharmaceuticals, Inc. Sirna Therapeutics has approximately ten years’ prior experience working to develop RNA molecules as drugs. This experience was largely gained with a different class of RNA molecules known as ribozymes, but could potentially be relevant for siRNAs. Companies working on gene therapy approaches to RNAi therapeutics include Nucleonics, Inc. and Benitec Ltd. In addition, CytRx Corporation may be developing RNAi therapeutics.
Other Companies Working to Develop Antisense Technology
Antisense technology uses short, single-stranded, DNA-like molecules known as oligonucleotides to block mRNAs encoding specific proteins. An antisense oligonucleotide, or ASO, contains a sequence of bases complementary to a sequence within its target mRNA, enabling it to attach to the mRNA by base-pairing. The attachment of the ASO may lead to breakdown of the mRNA, or may physically block the mRNA from associating with the protein synthesis machinery of the cell. In either case, production of the protein encoded by the mRNA may be reduced. Typically, the backbone of an ASO, the linkages that hold its constituent
60
Table of Contents
While we believe that RNAi drugs may potentially have significant advantages over ASOs, including greater potency and specificity, others are developing ASO drugs that are currently at a more advanced stage of development than RNAi drugs. For example, ISIS has developed an ASO drug, Vitravene, which is currently on the market, and has several ASO drug candidates in clinical trials. In addition, a number of other companies have product candidates in various stages of preclinical and clinical development, including Genta Inc., which has a drug candidate known as Genasense under review by the FDA as a potential treatment for various forms of cancer. ASOs, rather than siRNAs, may become the preferred technology for drugs that target mRNAs in order to turn off the activity of specific genes.
Competing Drugs for Age-Related Macular Degeneration
We are currently evaluating siRNAs that suppress VEGF gene activity as potential drug candidates for the treatment of wet AMD. VEGF is believed to play a major role in wet AMD by stimulating the growth and leakage of new blood vessels that disrupt the retina. siRNAs that reduce production of VEGF may potentially suppress the growth and leakage of these vessels. Visudyne, the only currently marketed drug for wet AMD, also targets these blood vessels. Worldwide sales of Visudyne were approximately $357 million in 2003. Visudyne is used in a two-step process known as photodynamic therapy. The first step in this therapy is to administer the drug by infusing it into a vein. The second step, after sufficient time has been allowed for the drug to reach the eye, is to shine laser light of the right color into the patient’s eye. This activates the drug, which then damages the walls of new blood vessels behind the retina, sealing these vessels. Visudyne is only approved for treating certain subtypes of wet AMD, and its beneficial effects may only last for a few months. We believe that a RNAi therapeutic that blocks production of VEGF may be more effective than photodynamic therapy in preventing the growth and leakage of new blood vessels behind the retina.
We are also aware of a number of experimental drugs in advanced stages of clinical development for the treatment of wet AMD. These include two drugs that are also intended to block the action of VEGF: Macugen, which is being developed by Eyetech Pharmaceuticals, Inc. in collaboration with Pfizer, Inc., and Lucentis, which is being developed by Genentech, Inc. in collaboration with Novartis AG. Each of these drugs is intended to work by binding to VEGF molecules and blocking their function. Conceptually, this approach requires one molecule of drug for every molecule of VEGF. We believe that a siRNA that blocks production of VEGF could be more effective at lower concentrations, because each siRNA molecule could potentially block production of many VEGF molecules. This is because each siRNA can potentially trigger degradation of multiple VEGF mRNA molecules, each of which could otherwise direct synthesis of multiple VEGF protein molecules.
Competing Drugs for Parkinson’s Disease
In collaboration with the Mayo Clinic, we are currently evaluating the potential of siRNAs directed against alpha-synuclein gene activity for the treatment of PD. The goal of our program is to find a RNAi therapeutic that will slow or halt the progress of the disease. Should we decide to pursue development of an alpha-synuclein siRNA for this disease, we would face competition from a number of marketed and investigational drugs. Many of the currently marketed drugs for PD work by boosting the levels of dopamine in the brain. Dopamine is an important substance that is depleted in the brains of PD patients. Dopamine-boosting drugs include:
| • | Carbidopa/levodopa, also sold as Sinemet and Atamet. The levodopa component of this combination drug is converted to dopamine in the brain. The carbidopa component helps to prevent the levodopa component from being used up before it reaches the brain. According to industry sources, sales of carbidopa/levodopa in the United States were approximately $202 million in 2003. | |
| • | Entacapone, sold as Comtan. Entacapone is another drug that prevents the breakdown of levodopa, further helping to increase dopamine levels in patients who take the carbidopa/levodopa combination. According to industry sources, worldwide sales of Comtan were approximately€75 million in 2002. | |
61
Table of Contents
| • | Selegiline, also sold as Eldepryl and Deprenyl. Selegiline slows the breakdown of dopamine, which is another strategy to boost depleted levels of dopamine in the brains of PD patients. According to industry sources, sales of selegiline in the United States were approximately $23 million in 2001. |
By boosting dopamine levels, all of these drugs help to treat the symptoms of PD, but none address the underlying cause of the disease, which is the death of brain cells that produce dopamine. Recently published scientific findings suggest that the death of these cells may be caused by over-production of the protein known as alpha-synuclein. We therefore believe that a RNAi therapeutic that reduces alpha-synuclein production could prevent the death of dopamine-producing cells, and thereby slow or halt progression of PD. A RNAi therapeutic with this capability would be expected to offer significant benefits over drugs that simply help alleviate the symptoms of PD.
We expect that any RNAi therapeutic we succeed in developing for PD would require administration using a specialized medical device to deliver it to the appropriate region of the brain. This requirement could make any RNAi therapeutic we develop less competitive than orally administered drugs for PD, such as the dopamine-boosting drugs described above.
In addition to marketed drugs, there are a number of investigational drugs for PD currently undergoing clinical development that are intended to alter the course of the disease, including the investigational drugs known as CEP-1347, GDNF and TCH-346. If one or more of these are developed and marketed successfully before an RNAi therapeutic reaches the marketplace, they could be strong competition for any RNAi therapeutic we may develop to treat PD.
Regulatory Matters
The research, testing, manufacture and marketing of drug products and their delivery systems are extensively regulated in the United States and the rest of the world. In the United States, drugs are subject to rigorous regulation by the FDA. The Federal Food, Drug, and Cosmetic Act and other federal and state statutes and regulations govern, among other things, the research, development, testing, manufacture, storage, record keeping, packaging, labeling, promotion and advertising, marketing and distribution of pharmaceutical products. The failure to comply with the applicable regulatory requirements may subject a company to a variety of administrative or judicially-imposed sanctions and the inability to obtain or maintain required approvals or to market approved drug products.
The steps ordinarily required before a new pharmaceutical product may be marketed in the United States include preclinical laboratory tests, animal tests and formulation studies, the submission to the FDA of an investigational new drug application, which must become effective prior to commencement of clinical testing, and adequate and well-controlled clinical trials to establish that the drug product is safe and effective for the indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically take several years and the actual time taken may vary substantially depending upon the complexity of the product or the disease. Government regulation may delay or prevent marketing of potential products for a considerable period of time and impose costly procedures on a company’s activities. Success in early stage clinical trials does not necessarily assure success in later stage clinical trials. Data obtained from clinical activities is not always conclusive and may be subject to alternative interpretations that could delay, limit or even prevent regulatory approval. Even if a product receives regulatory approval, later discovery of previously unknown problems with a product may result in restrictions on the product or even complete withdrawal of the product from the market.
Preclinical tests include laboratory evaluation of product chemistry and formulation, as well as animal trials to assess the potential safety and efficacy of the product. The conduct of the preclinical tests and formulation of compounds for testing must comply with federal regulations and requirements. The results of preclinical testing are submitted to the FDA as part of an investigational new drug application.
A 30-day waiting period after the filing of an investigational new drug application is required prior to such application becoming effective and the commencement of clinical testing in humans. If the FDA has not commented on, or questioned, the application during this 30-day waiting period, clinical trials may begin. If
62
Table of Contents
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted in compliance with federal regulations and requirements, under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the safety and effectiveness criteria to be evaluated. Each protocol involving testing on subjects in the United States must be submitted to the FDA as part of the investigational new drug application. The study protocol and informed consent information for patients in clinical trials must be submitted to institutional review boards for approval.
Clinical trials to support new drug applications for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase I, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics and pharmacological actions and safety, including side effects associated with increasing doses. Phase II usually involves trials in a limited patient population, to determine dosage tolerance and optimum dosage, identify possible adverse effects and safety risks, and provide preliminary support for the efficacy of the drug in the indication being studied.
If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase II evaluations, Phase III trials are undertaken to further evaluate clinical efficacy and to further test for safety within an expanded patient population, typically at geographically dispersed clinical trial sites. Phase I, Phase II or Phase III testing of any product candidates may not be completed successfully within any specified time period, if at all. After successful completion of the required clinical testing, generally a new drug application is prepared and submitted to the FDA.
We believe that any Direct RNAi product candidate we develop for AMD or Parkinson’s disease will be regulated as a new drug by the FDA. FDA approval of the new drug application is required before marketing of the product may begin in the United States. The new drug application must include the results of extensive clinical and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture and controls. The cost of preparing and submitting a new drug application is substantial. Under Federal law, the submission of new drug applications are additionally subject to substantial application user fees, currently exceeding $500,000, and sponsor under an approved new drug application are also subject to annual product and establishment user fees, currently exceeding $30,000 per product and $200,000 per establishment. These fees are typically increased annually.
The FDA has 60 days from its receipt of a new drug application to determine whether the application will be accepted for filing based on the agency’s threshold determination that the new drug application is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review of the new drug application. The review process is often significantly extended by FDA requests for additional information or clarification regarding information already provided in the submission. The FDA may also refer applications for novel drug products or drug products which present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee. The FDA normally also will conduct a preapproval inspection to ensure the manufacturing facility, methods and controls are adequate to preserve the drug’s identity, strength, quality, purity and stability, and are in compliance with regulations governing current good manufacturing practices.
If FDA evaluations of the new drug application and the manufacturing facilities are favorable, the FDA may issue an approval letter or an approvable letter followed by an approval letter. An approvable letter generally contains a statement of specific conditions that must be met in order to secure final approval of the new drug application. If and when those conditions have been met to the FDA’s satisfaction, the FDA will
63
Table of Contents
While we believe that any RNAi therapeutic we develop will be regulated as a new drug under the Federal Food, Drug, and Cosmetics Act, the FDA could decide to regulate RNAi therapeutics as biologics under the Public Health Service Act. Biologics must have a biologics license application, or BLA, approved prior to commercialization. Like new drug applications, BLAs are subject to user fees. To obtain BLA approval, an applicant must provide preclinical and clinical evidence and other information to demonstrate that the biologic product is safe, pure and potent, and that the facilities in which it is manufactured, processed, packed or held meet standards, including drug good manufacturing practices and any additional standards in the license designed to ensure its continued safety, purity and potency. Biologics establishments are subject to preapproval inspections. The review process for BLAs is time consuming and uncertain, and BLA approval may be conditioned on post approval testing and surveillance. Once granted, BLA approvals may be suspended or revoked under certain circumstances, such as if the product fails to conform to the standards established in the license.
Once a new drug application or biologics license application is approved, a product will be subject to certain post-approval requirements, including requirements for adverse event reporting and submission of periodic reports. Additionally, the FDA also strictly regulates the promotional claims that may be made about prescription drug products and biologics. In particular, the FDA requires substantiation of any claims of superiority of one product over another, including that such claims be proven by adequate and well controlled head-to-head clinical trials. To the extent that market acceptance of our products may depend on their superiority over existing therapies, any restriction on our ability to advertise or otherwise promote claims of superiority, or requirements to conduct additional expensive clinical trials to provide proof of such claims, could negatively affect the sales of our products or our costs. We must also notify the FDA of any change in an approved product beyond variations already allowed in the approval. Certain changes to the product, its labeling or its manufacturing require prior FDA approval and may require conduct of further clinical investigations to support the change. Such approvals may be expensive and time-consuming and, if not approved, the product will not be allowed to be marketed as modified.
If the FDA’s evaluation of the new drug application submission or manufacturing facilities is not favorable, the FDA may refuse to approve the new drug application or issue a not approvable letter. The not approvable letter outlines the deficiencies in the submission and often requires additional testing or information in order for the FDA to reconsider the application. Even after submitting this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. With limited exceptions, the FDA may withhold approval of a new drug application regardless of prior advice it may have provided or commitments it may have made to the sponsor.
Some of our drug candidates may need to be administered using specialized drug delivery systems. While we expect to rely on drug delivery systems that are already approved to deliver drugs like ours to similar physiological sites, in some instances, we may need to modify the design or labeling of the legally available device for delivery of our product candidate. In such an event, the FDA may regulate the product as a combination product or require additional approvals or clearances for the modified device. Further, to the extent the delivery device is owned by another company, we would need that company’s cooperation to implement the necessary changes to the device and to obtain any additional approvals or clearances. Obtaining such a additional approvals or clearances, and cooperation of other companies, when necessary, could significantly delay, and increase the cost of obtaining, marketing approval, which could reduce the commercial viability of a drug candidate.
Once a new drug application is approved, the product covered thereby becomes a listed drug that can, in turn, be cited by potential competitors in support of approval of an abbreviated new drug application. An
64
Table of Contents
From time to time legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the approval, manufacturing and marketing of drug products. In addition, FDA regulations and guidance are often revised or reinterpreted by the agency in ways that may significantly affect our business and our products. It is impossible to predict whether legislative changes will be enacted, or FDA regulations, guidance or interpretations changed, or what the impact of such changes, if any, may be.
Foreign Regulation of New Drug Compounds
Approval of a product by comparable regulatory authorities may be necessary in foreign countries prior to the commencement of marketing of the product in those countries, whether or not FDA approval has been obtained. The approval procedure varies among countries and can involve requirements for additional testing. The time required may differ from that required for FDA approval. Although there are some procedures for unified filings for some European countries with the sponsorship of the country which first granted marketing approval, in general, each country has its own procedures and requirements, many of which are time consuming and expensive. Thus, there can be substantial delays in obtaining required approvals from foreign regulatory authorities after the relevant applications are filed.
In Europe, marketing authorizations may be submitted at a centralized, a decentralized or a national level. The centralized procedure is mandatory for the approval of biotechnology products and provides for the grant of a single marketing authorization that is valid in all European Union member states. As of January 1995, a mutual recognition procedure is available at the request of the applicant for all medicinal products
65
Table of Contents
Hazardous Materials
Our research and development processes involve the controlled use of hazardous materials, chemicals and radioactive materials and produce waste products. We are subject to federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous materials and waste products. We do not expect the cost of complying with these laws and regulations to be material.
Manufacturing
We have no commercial manufacturing capabilities. We plan to rely on third parties to manufacture bulk compounds and finished investigational medicines for clinical trials. Commercial quantities of any drugs that we may seek to develop will have to be manufactured in facilities and by processes that comply with FDA and other regulations. We plan to rely on third parties to manufacture commercial quantities of any products that we successfully develop. Under our agreement with Isis, at our request, we may negotiate a manufacturing services agreement with Isis for double-stranded RNA products designed to work through a RNAi mechanism.
Scientific Advisors
We seek advice from our scientific advisory board, which consists of a number of leading scientists and physicians, on scientific and medical matters. Our scientific advisory board meets regularly to assess:
| • | our research and development programs; | |
| • | the design and implementation of our clinical programs; | |
| • | our patent and publication strategies; | |
| • | new technologies relevant to our research and development programs; and | |
| • | specific scientific and technical issues relevant to our business. |
The current members of our scientific advisory board are:
| Name | Position/Institutional Affiliation | |
| Dennis A. Ausiello, M.D. | Physician-in-chief/ Massachusetts General Hospital | |
| David P. Bartel, Ph.D. | Associate Professor/ Whitehead Institute for Medical Research | |
| Fritz Eckstein, Ph.D. | Professor/ Max Planck Institute | |
| Edward E. Harlow, Ph.D. | Professor/ Harvard Medical School | |
| Robert S. Langer, Ph.D. | Geimeshausen Professor/ Massachusetts Institute of Technology | |
| Paul R. Schimmel, Ph.D. | Professor/ Skaggs Institute for Chemical Biology | |
| Phillip A. Sharp, Ph.D. | Institute Professor/ McGovern Institute for Brain Research at MIT | |
| Markus Stoffel, M.D., Ph.D. | Heilbrunn Professor/ Rockefeller University | |
| Thomas H. Tuschl, Ph.D. | Associate Professor/ Rockefeller University | |
| Phillip D. Zamore, Ph.D. | Associate Professor/ University of Massachusetts Medical School |
Employees
As of March 31, 2004, we had 61 full-time employees, 47 of whom were engaged in research and development and 14 of whom were engaged in management, administration and finance. Of our employees,
66
Table of Contents
Facilities
Our operations are based primarily in Cambridge, Massachusetts and Kulmbach, Germany. In April 2004, we plan to move our Cambridge operations from our current facility at 790 Memorial Drive to a new facility at 300 Third Street, Cambridge. The properties we lease are listed below:
| Square | Monthly | |||||||||||||||
| Location | Feet | Type | Lease Expires | Lease Payments | ||||||||||||
| 790 Memorial Drive, Cambridge, MA | 14,000 | Office & laboratory | May 2004 | $ | 71,000 | |||||||||||
| 300 Third Street, Cambridge, MA | 33,000 | Office & laboratory | September 2011 | 114,000 | ||||||||||||
| Kulmbach, Germany | 14,000 | Office & laboratory | June 2008 | 25,000 | ||||||||||||
We also hold a lease on expansion space of 10,600 square feet within the 300 Third Street, Cambridge, Massachusetts facility, for which payment will begin by September 1, 2005, and options to lease an additional 21,100 square feet of office and laboratory space. We believe that the total space available to us under our current leases and options will meet our needs for the foreseeable future, and that additional space would be available to us on commercially reasonable terms if it were required.
Legal Proceedings
We are currently not a party to any material legal proceedings.
67
Table of Contents
MANAGEMENT
Directors, Executive Officers and Other Key Employees
Our directors, executive officers and other key employees, and their ages and positions as of March 31, 2004, are set forth below:
| Age | Position | |||||
Directors and Executive Officers | ||||||
| John M. Maraganore, Ph.D. | 41 | President, Chief Executive Officer and Director | ||||
| Barry E. Greene | 40 | Chief Operating Officer and Treasurer | ||||
| Vincent J. Miles, Ph.D. | 53 | Senior Vice President, Business Development | ||||
| Thomas R. Ulich, M.D. | 50 | Senior Vice President, Research and Development | ||||
| Peter Barrett, Ph.D. (2)(3) | 50 | Director | ||||
| John E. Berriman (2) | 55 | Director | ||||
| John K. Clarke (1)(3) | 50 | Co-Founder and Chairman of the Board of Directors | ||||
| Paul R. Schimmel, Ph.D. (1) | 63 | Co-Founder and Director | ||||
| Phillip A. Sharp, Ph.D. (2)(3) | 59 | Co-Founder and Director | ||||
| Kevin P. Starr (1) | 41 | Director | ||||
| Christoph H. Westphal, M.D., Ph.D. | 36 | Co-Founder and Director | ||||
Other Key Employees | ||||||
| David M. Konys | 53 | Vice President of Corporate Development and Operations | ||||
| Victor E. Kotelianski, M.D., Ph.D. | 55 | Vice President of Research and Development | ||||
| Muthiah Manoharan, Ph.D. | 51 | Vice President of Drug Discovery | ||||
| Roland Kreutzer, Ph.D. | 44 | Co-Founder and Member of Management Board, Alnylam Europe, AG | ||||
| Stefan Limmer, Ph.D. | 52 | Co-Founder and Member of Management Board, Alnylam Europe, AG | ||||
| (1) | Member of Audit Committee |
| (2) | Member of Compensation Committee |
| (3) | Member of Nominating and Corporate Governance Committee |
John M. Maraganore, Ph.D.has served as our President and Chief Executive Officer and as a member of our board of directors since December 2002. From April 2000 to December 2002, Dr. Maraganore served as Senior Vice President, Strategic Product Development for Millennium Pharmaceuticals, Inc., a biopharmaceutical company. From April 1997 to April 2000, Dr. Maraganore also served as Millennium’s Vice President, Strategic Planning and M&A and as General Manager of Millennium BioTherapeutics, Inc., a former subsidiary of Millennium. Before joining Millennium, Dr. Maraganore served in several capacities including Director of Biological Research and Director of Market and Business Development for Biogen, Inc., a biopharmaceutical company. At Biogen, Dr. Maraganore invented Angiomax, a direct thrombin inhibitor currently marketed by The Medicines Company. Before Biogen, Dr. Maraganore was a Senior Scientist at Zymogenetics, Inc., and a Visiting Research Scientist at The Upjohn Company. Dr. Maraganore received an M.S. and Ph.D. in biochemistry and molecular biology from the University of Chicago.
Barry E. Greenehas served as our Chief Operating Officer since he joined us in October 2003 and as our Treasurer since February 2004. From February 2001 to September 2003, Mr. Greene served as General Manager of Oncology at Millennium Pharmaceuticals, Inc., a biopharmaceutical company. From January 2000 to February 2001, Mr. Greene served as Executive Vice President and Chief Business Officer for
68
Table of Contents
Vincent J. Miles, Ph.D.has served as our Senior Vice President of Business Development since he joined us in July 2003. From May 1997 to July 2003, Dr. Miles held various positions at Millennium Pharmaceuticals, Inc., a biopharmaceutical company, including vice president positions in business development, strategic planning and scientific affairs. Dr. Miles received his bachelor’s degree in biochemistry and his Ph.D. in molecular biology from University College London.
Thomas R. Ulich, M.D.has served as our Senior Vice President of Preclinical Development since he joined us in October 2003. From 1993 to January 2003, Dr. Ulich held various positions at Amgen, Inc., a biopharmaceutical company, including Vice President of Preclinical Development and Vice President of Preclinical Development and Protein Therapeutics. Dr. Ulich is also Professor of Pathology at the University of California, San Diego. Dr. Ulich received a B.A. from Dartmouth College and an M.D. from the University of California, Los Angeles.
Peter Barrett, Ph.D.has served as a member of our board of directors since July 2002. Dr. Barrett has served as a Senior Principal of Atlas Venture, a venture capital firm, since January 2002. From August 1998 to December 2001, he served as Executive Vice President and Chief Business Officer of Celera Genomics, a biopharmaceutical company, which he co-founded. He also served as Vice President of Celera from 1994 to 1998. Dr. Barrett received a B.S. in chemistry from Lowell Technological Institute (now known as the University of Massachusetts, Lowell) and a Ph.D. in analytical chemistry from Northeastern University. He also completed Harvard Business School’s Management Development Program.
John E. Berrimanhas served as a member of our board of directors since July 2003. Mr. Berriman has served as a Director of Abingworth Management, a venture capital firm specializing in life science biomedical companies, since August 2001. From March 1997 to August 2001, Mr. Berriman served as a consultant to Abingworth. He holds a degree in Chemical Engineering from the University of Cambridge and an M.B.A. from the London Business School.
John K. Clarkeis a founder of Alnylam and has served as the Chairman of our board of directors since June 2002. Since founding Cardinal Partners, a venture capital firm focused on healthcare, in 1997, Mr. Clarke has served as its Managing General Partner. He received an A.B. in economics and biology from Harvard University and an M.B.A. from the Wharton School at the University of Pennsylvania. Mr. Clarke also serves as a director of Cubist Pharmaceuticals, Inc.
Paul R. Schimmel, Ph.D.is a founder of Alnylam and has served as a member of our board of directors since June 2002. Dr. Schimmel has been a Professor of Molecular Biology at the Skaggs Institute for Chemical Biology since 1997. Dr. Schimmel received a Ph.D. from the Massachusetts Institute of Technology. Dr. Schimmel also serves as a director of Alkermes, Inc. and Repligen, Inc.
Phillip A. Sharp, Ph.D.is a founder of Alnylam and has served as a member of our board of directors since June 2002. Dr. Sharp is the Founding Director of the McGovern Institute for Brain Research at the Massachusetts Institute of Technology, where he has served as a professor since 1974. In 1993, Dr. Sharp was one of two recipients of the Nobel Prize in Physiology or Medicine, which he received for his work in molecular biology. He received a B.A. from Union College in Kentucky and a Ph.D. in chemistry from the University of Chicago. Dr. Sharp also serves as a director of Biogen Idec.
Kevin P. Starrhas served as a member of our board of directors since September 2003. From December 2001 to December 2002, Mr. Starr served as Chief Operating Officer of Millennium Pharmaceuticals, Inc., a biopharmaceutical company. Mr. Starr also served as Millennium’s Chief Financial Officer from December 1998 to December 2002. From June 2000 to December 2001, Mr. Starr served in various vice president positions at Millennium, including Executive President, Business Operations, and Senior Vice President. From March 1998 to December 1998, he served as the Vice President, Finance of
69
Table of Contents
Christoph H. Westphal, M.D., Ph.D.is a founder of Alnylam and has served as a member of our board of directors since our incorporation in June 2002. In addition, from June 2002 to December 2002, Dr. Westphal was our start-up Chief Executive Officer and, from June 2002 to February 2004, he served as our Vice Chairman. Dr. Westphal has served as a General Partner of Polaris Venture Partners, a venture capital firm, since June 2000. Prior to joining Polaris, Dr. Westphal was at McKinsey & Co., a consulting firm. Dr. Westphal received a B.A. from Columbia University and an M.D./ Ph.D. from Harvard Medical School. Dr. Westphal is vice chairman of Momenta Pharmaceuticals, Inc. and Acceleron Pharma.
David M. Konyshas served as our Vice President of Corporate Development since he joined us in April 2003. From March 2001 to September 2002, he served as Vice President Corporate Development for Ingenium Pharmaceuticals, a biopharmaceutical company. From 1985 to March 2001, Mr. Konys served in various positions at Biogen, Inc., a biopharmaceutical company, most recently as Director, Commercial Projects. Mr. Konys received a B.S. in Biology from Xavier University.
Victor E. Kotelianski, M.D., Ph.D.has served as our Vice President Research and Development since he joined us in April 2003. From October 2001 to March 2003, Dr. Kotelianski served as Distinguished Investigator, Director Biological Research for Biogen, Inc., a biopharmaceutical company. From October 1996 to December 2001, he also served as Section Head and Group Leader, Department of Immunology and Cell Biology for Biogen. Dr. Kolelianski received an M.D./ Ph.D. from Uzhgorod University School of Medicine and a Ph.D. from the National Academy of Science.
Muthiah Manoharan, Ph.D.has served as our Vice President of Drug Discovery since he joined us in February 2003. From January 2001 to January 2003, Dr. Manoharan served as Executive Director of Medicinal Chemistry at Isis Pharmaceuticals, Inc., a biotechnology company focused on antisense oligonucleotide therapeutics. He also served as Director, Medicinal Chemistry for Isis Pharmaceuticals from 1996 to January 2001. Dr. Manoharan received a Ph.D. in chemistry at the University of North Carolina-Chapel Hill.
Roland Kreutzer, Ph.D.is a founder of Alnylam Europe, AG and has served on the Management Board of Alnylam Europe, AG since October 2000. From October 2000 to July 2003 he also served as Chief Executive Officer of Alnylam Europe, AG. From January 1990 to August 2000, he served as an Assistant Professor at the University of Bayreuth, Germany. Dr. Kreutzer received his Ph.D. in molecular genetics from the University of Bayreuth, Germany.
Stefan Limmer, Ph.D.is a founder of Alnylam Europe, AG and has served on the Management Board of Alnylam Europe, AG since its incorporation in August 2000. From August 2000 to July 2003 he also served as Chief Scientific Officer of Alnylam Europe, AG. He received his Ph.D. in physics from the University of Leipzig. Since February 1995, Dr. Limmer has served as a biochemistry lecturer at the University of Bayreuth, Germany.
Board of Directors
Upon completion of this offering, we will have a board of directors consisting of eight members. Following this offering, the board of directors will be divided into three classes, each of whose members will serve for a staggered three-year term. The board of directors will consist of three class I directors: John M. Maraganore, Ph.D., Paul R. Schimmel, Ph.D. and Phillip A. Sharp, Ph.D.; three class II directors: John E. Berriman, John K. Clarke and Christoph H. Westphal, M.D., Ph.D.; and two class III directors: Peter Barrett, Ph.D. and Kevin P. Starr. At each annual meeting of stockholders, a class of directors will be elected for a three-year term to succeed the directors of the same class whose terms are then expiring. The terms of the class I directors, class II directors and class III directors expire upon the election and qualification of successor directors at the annual meeting of stockholders held during the calendar years 2005, 2006 and 2007, respectively.
70
Table of Contents
Our amended and restated bylaws provide that the authorized number of directors may be changed only by resolution of the board of directors or by the stockholders. Any additional directorships resulting from an increase in the number of directors will be distributed among the three classes, so that, as nearly as possible, each class will consist of one-third of the total number of directors. This classification of the board of directors may have the effect of delaying or preventing changes in the control or management of us.
Each executive officer is elected by, and serves at the discretion of, the board of directors. Each of our executive officers and directors, other than non-employee directors, devotes his or her full time to our affairs. Each of our directors currently serves on the board of directors pursuant to a stockholders’ voting agreement. The stockholders’ voting agreement will terminate upon the closing of this offering. There are no family relationships among any of our directors or officers.
Board Committees
Our board of directors has established an audit committee, a compensation committee and a nominating and corporate governance committee. The members of each committee are appointed by the board of directors.
Audit Committee
We have an audit committee consisting of Kevin P. Starr, Chairman, John K. Clarke and Paul R. Schimmel, Ph.D. The audit committee assists our board of directors in its oversight of:
| • | the integrity of our financial statements; | |
| • | the independent auditor’s qualifications and independence; and | |
| • | the performance of our independent auditors. |
The audit committee has direct responsibility for the appointment, compensation, retention and oversight of the work of our independent accountants, PricewaterhouseCoopers LLP. In addition, the audit committee must approve any related party transaction entered into by us. We believe that each member of the audit committee satisfies the requirements for membership established by the NASDAQ National Market and the SEC.
Compensation Committee
We have a compensation committee consisting of Peter Barrett, Ph.D., Chairman, John E. Berriman and Phillip A. Sharp, Ph.D. The compensation committee reviews, and makes recommendations to the board of directors regarding, the compensation and benefits of our executive officers and key managers. The compensation committee also administers the issuance of stock options and other awards under our stock plans and establishes and reviews policies relating to the compensation and benefits of our employees and consultants. We believe that each member of the compensation committee satisfies the requirements for membership established by the NASDAQ National Market.
Nominating and Corporate Governance Committee
We have a nominating and corporate governance committee consisting of John K. Clarke, Chairman, Peter Barrett, Ph.D. and Phillip A. Sharp, Ph.D. The purpose of the nominating and corporate governance committee is to:
| • | identify and nominate members of the board of directors; | |
| • | develop and recommend to the board of directors a set of corporate governance principles; and | |
| • | oversee the evaluation of the board of directors and management. |
Procedures for the consideration of director nominees recommended by stockholders are set forth in our amended and restated bylaws, which will be effective upon completion of this offering. We believe that each
71
Table of Contents
Compensation of Directors
Each of our directors, other than a director who is employed by us or beneficially owns at least 1.5% of our outstanding capital stock, will receive a fee of $5,000 per quarter for serving on the board of directors. Each of our directors who serves as the chairman of a committee of the board of directors will receive an additional $5,000 per year for each such position held. Directors are reimbursed for reasonable travel and other expenses incurred in connection with attending meetings of the board of directors and its committees.
Directors are also eligible to participate in our 2004 equity incentive plan. Pursuant to the 2004 plan, each non-employee director is eligible to receive an option to purchase 13,500 shares of our common stock upon his appointment to the board. Each director who (1) is not employed by us, (2) has served as a director for at least six months, (3) does not beneficially own more than 1.5% of our outstanding capital stock and (4) attended at least 75% of the meetings of the board, including meetings of the committees on which the director served, held during the preceding year, is also eligible to receive an option to purchase 10,000 shares of our common stock at each year’s annual meeting at which he or she serves as a director. These stock options vest in full on the first anniversary of the date of grant provided he or she is still serving as a director. Each stock option terminates upon the earlier of ten years from the date of grant and three months after the optionee ceases to serve as a director. The exercise price of these options is the fair market value of our common stock on the date of grant.
In 2003, Dr. Schimmel and Mr. Starr each received $3,000 per month for service on our board of directors and Dr. Sharp received $1,250 per month for service on our board of directors. In addition, in 2003, we granted the following stock options under our 2003 employee, director and consultant stock plan to the following non-employee directors:
| Name of Directors | Number of Shares | |||
| Kevin P. Starr | 100,000 | |||
| Phillip A. Sharp, Ph.D. | 70,000 | |||
These options vest as to 25% of the shares on the first anniversary of the date of grant and, with respect to the option granted to Mr. Starr, as to an additional 2.08% of the shares on the last day of each full calendar month thereafter and, with respect to Dr. Sharp, as to an additional 6.25% of the shares on the last day of each calendar quarter thereafter.
Compensation Committee Interlocks and Insider Participation
None of our executive officers serves as a member of the board of directors or compensation committee, or other committee serving an equivalent function, of any other entity that has one or more of its executive officers serving as a member of our board of directors or compensation committee. None of the current members of our compensation committee has ever been an employee of Alnylam.
72
Table of Contents
Executive Compensation
The table below sets forth the total compensation paid or accrued for the fiscal year ended December 31, 2003 for our chief executive officer and each of our three most highly compensated other executive officers who were serving as executive officers on December 31, 2003 and whose total annual compensation exceeded $100,000 for the year ended December 31, 2003. We refer to these officers as our named executive officers.
Summary Compensation Table
| Long-Term | |||||||||||||||||
| Compensation | |||||||||||||||||
| Awards | |||||||||||||||||
| Annual | |||||||||||||||||
| Compensation | Securities | ||||||||||||||||
| Underlying | All Other | ||||||||||||||||
| Name and Principal Position | Salary | Bonus | Options (#) | Compensation | |||||||||||||
| John M. Maraganore, Ph.D. | $ | 369,398 | $ | 110,000 | 793,000 | $ | — | ||||||||||
President and Chief Executive Officer | |||||||||||||||||
| Thomas R. Ulich, M.D.(1) | 157,403 | 46,113 | 300,000 | 43,462 | |||||||||||||
Senior Vice President, Research and Development | |||||||||||||||||
| Vincent J. Miles, Ph.D.(2) | 107,716 | 44,975 | 150,000 | — | |||||||||||||
Senior Vice President, Business Development | |||||||||||||||||
| John G. Conley(3) | 259,453 | 55,250 | — | — | |||||||||||||
Former Chief Financial Officer and Vice President, Finance and Strategy | |||||||||||||||||
| (1) | Dr. Ulich commenced employment with us in June 2003. His current annual salary is $313,720. The amount indicated under the heading “All Other Compensation” represents reimbursement of relocation expenses and related taxes. |
| (2) | Dr. Miles commenced employment with us in July 2003. His current annual salary is $237,115. |
| (3) | Mr. Conley resigned from Alnylam effective January 31, 2004. |
73
Table of Contents
Option Grants in Last Fiscal Year
The following table sets forth each grant of stock options during the fiscal year ended December 31, 2003 to each of the named executive officers. The potential realizable value set forth in the last column of the table is calculated based on the term of the option at the time of grant, which is ten years. This value is based on assumed rates of stock price appreciation of 5% and 10% compounded annually from the date of grant until their expiration date, assuming a fair market value equal to an assumed initial public offering price of $ , minus the applicable exercise price. These numbers are calculated based on the requirements of the SEC and do not reflect our estimate of future stock price growth. Actual gains, if any, on stock option exercises will depend on the future performance of the common stock on the date on which the options are exercised.
| Potential | ||||||||||||||||||||||||
| Realizable Value at | ||||||||||||||||||||||||
| Percent of | Assumed Annual | |||||||||||||||||||||||
| Number of | Total | Rates of Stock | ||||||||||||||||||||||
| Securities | Options | Price Appreciation | ||||||||||||||||||||||
| Underlying | Granted to | for Option Term(3) | ||||||||||||||||||||||
| Options | Employees in | Exercise | Expiration | |||||||||||||||||||||
| Name | Granted(1) | Fiscal Year | Price(2) | Date | 5% | 10% | ||||||||||||||||||
| John M. Maraganore, Ph.D. | 610,000 | 22.1 | % | $ | 0.25 | 2/25/13 | $ | $ | ||||||||||||||||
| 183,000 | (4) | 6.6 | % | 0.25 | 2/25/13 | |||||||||||||||||||
| Thomas R. Ulich, M.D. | 300,000 | 10.9 | 0.25 | 7/31/13 | ||||||||||||||||||||
| Vincent J. Miles, Ph.D. | 150,000 | 5.4 | 0.25 | 7/31/13 | ||||||||||||||||||||
| John G. Conley | — | — | — | — | — | — | ||||||||||||||||||
| (1) | Stock options granted to our executive officers generally vest as to 25% of the shares on the first anniversary of the vesting commencement date established by the board of directors and as to an additional 6.25% of the shares on the last day of each calendar quarter thereafter. See also footnote 4 below. |
| (2) | The exercise price per share was determined to be equal to the fair market value per share of common stock as valued by our board of directors on the date of grant. |
| (3) | Amounts represent hypothetical gains that could be achieved for stock options if exercised at the end of the option term. These gains are based on assumed rates of stock price appreciation of 5% and 10% compounded annually from the date stock options are granted. Actual gains, if any, on stock option exercises will depend on the future performance of common stock on the date on which the stock options are exercised. |
| (4) | Under the terms of the option agreement, 91,500 of these shares vested immediately upon Alnylam entering into its first significant strategic alliance, which occurred on September 8, 2003. The remaining 91,500 of these shares vest in equal installments on the last day of each quarterly period thereafter over four years. |
74
Table of Contents
Fiscal Year-End Option Values
The following table sets forth information for each of the named executive officers regarding the number of shares subject to both exercisable and unexercisable stock options, as well as the value of unexercisable in-the-money options, as of December 31, 2003. There was no public trading market for our common stock as of December 31, 2003. Accordingly, the value of the unexercised in-the-money options at fiscal year-end has been calculated by determining the difference between the exercise price per share and the fair market value of our common stock at fiscal year end, as determined by our board of directors. None of the named executive officers exercised options during the fiscal year ended December 31, 2003.
| Number of | ||||||||||||||||
| Securities Underlying | Value of Unexercised | |||||||||||||||
| Unexercised Options | In-The-Money Options | |||||||||||||||
| at December 31, 2003 | at December 31, 2003 | |||||||||||||||
| Name | Exercisable | Unexercisable | Exercisable | Unexercisable | ||||||||||||
| John M. Maraganore, Ph.D. | 287,843 | 505,157 | $ | 290,721 | $ | 510,209 | ||||||||||
| Thomas R. Ulich, M.D. | — | 300,000 | — | 303,000 | ||||||||||||
| Vincent J. Miles, Ph.D. | — | 150,000 | — | 151,500 | ||||||||||||
| John G. Conley | — | — | — | — | ||||||||||||
Employment Arrangements
We have entered into letter agreements with Dr. Maraganore, Dr. Ulich, Dr. Miles and Mr. Greene regarding their employment with us.
| • | Dr. Maraganore’s agreement provides that, if he is employed upon a change in control of Alnylam, all options held by Dr. Maraganore will vest and become immediately exercisable. In addition, Dr. Maraganore’s agreement provided for the issuance to him of an option to purchase 183,000 shares of our common stock, 91,500 of which would vest if and when we entered into a significant strategic alliance and the remaining 91,500 of which would vest in equal installments on the last day of each quarterly period thereafter over four years. Upon execution on September 8, 2003 of our collaboration agreement with Merck, which our board of directors deemed to be a significant strategic alliance, the option vested as to the initial 91,500 shares and quarterly vesting for the remaining shares commenced. | |
| • | Dr. Ulich’s agreement provides that if he is terminated without cause prior to June 30, 2005, we will enter into a consulting agreement with Dr. Ulich and continue to pay him his base salary for twelve months at the rate in effect at the time of his termination. | |
| • | Dr. Miles’ agreement provides that, if we enter into a strategic alliance, other than our collaboration with Merck, within 18 months of the date Dr. Miles began his employment with us and such alliance provides us with at least $10 million of cash flow, we will issue Dr. Miles a stock option to purchase 50,000 shares of our common stock. | |
| • | Mr. Greene’s agreement provides that, if he is employed upon a change in control of Alnylam, all options held by Mr. Greene will vest and become immediately exercisable. In addition, if we terminate his employment without cause during the first 12 months of his employment, we will pay Mr. Greene six months severance based on his base salary in effect at the time of the termination of his employment. | |
Each executive officer has signed a nondisclosure, invention and non-competition agreement providing for the protection of our confidential information and ownership of intellectual property developed by such executive officer and a one-year non-compete provision.
75
Table of Contents
Employee Benefit Plans
2002 Employee, Director and Consultant Stock Plan, 2003 Employee, Director and Consultant Stock Plan and 2004 Stock Incentive Plan
Our 2002 employee, director and consultant stock plan, or 2002 plan, was initially adopted in November 2002. Our 2003 employee director and consultant stock plan, or 2003 plan, was initially adopted in September 2003. As of March 31, 2004, an aggregate of 4,407,500 shares of common stock were authorized for issuance under the 2002 plan and 2003 plan, 3,691,618 of which were subject to outstanding options at a weighted average exercise price of $0.32 per share, and 346,601 of which were available for future grant. However, we do not intend to make any further grants under the 2002 plan or the 2003 plan following the completion of this offering.
Our 2004 stock incentive plan, or 2004 plan, was initially adopted by our board of directors in March 2004 and by our stockholders in 2004 and, upon adoption, 3,000,000 shares of common stock were authorized for issuance under the 2004 plan. Additional shares of common stock may be authorized for issuance under the 2004 plan upon the expiration, termination, cancellation or repurchase of existing awards under the 2002 plan and 2003 plan. The exact number of shares that can be added to the 2004 plan equals the sum of the number of shares reserved for issuance under the 2002 plan and 2003 plan that remain available for grant immediately prior to the closing of this offering, plus the number of shares subject to awards under the 2002 plan and 2003 plan that have expired or terminated or we have repurchased. In addition, the 2004 plan provides for an annual increase in the number of shares available for issuance under the 2004 plan equal to the lesser of 5,000,000 shares of our common stock, 5% of our outstanding shares or an amount determined by the board.
All of our plans provide for the grant of options intended to qualify as incentive stock options under Section 422 of the Internal Revenue Code, non-statutory stock options and restricted stock awards.
Our employees, officers, directors, consultants and advisors are eligible to receive awards under the plans. Under present law, however, incentive stock options may only be granted to employees, including officers.
Optionees receive the right to purchase a specified number of shares of common stock at a specified option price and subject to any other terms and conditions specified in connection with the option grant at an exercise price equal to or greater than the fair market value of our common stock on the date of grant. Under present law, incentive stock options and options intended to qualify as performance-based compensation under Section 162(m) of the Internal Revenue Code may not be granted to optionees holding more than 10% of the voting power of all shares of our capital stock at an exercise price less than 110% of the fair market value of our common stock on the date of grant. The plans permit our board of directors to determine how optionees may pay the exercise price of their options, including through payment by cash, check, surrender to us of shares of common stock owned for at least six months or by any combination of the permitted forms of payment. In addition, at the discretion of the board of directors, under the 2002 plan and the 2003 plan, options may be exercised in accordance with a cashless exercise program established with a brokerage service and under the 2004 plan they may be exercised by delivery to us of an irrevocable undertaking of a creditworthy broker to promptly deliver the exercise price to us.
The compensation committee of our board of directors administers the plans. The compensation committee has the authority to grant awards, including awards to executive officers, and to adopt, amend and repeal the administrative rules, guidelines and practices relating to the plans of and to interpret the provisions of the plans. In addition, our board of directors may delegate authority under the 2004 plan to one or more of our executive officers. Subject to any applicable limitations contained in the plans, our compensation committee, or if applicable, one or more executive officers to whom authority has been granted under the 2004 plan, selects the recipients of awards and determines:
| • | the number of shares of common stock covered by options and the dates upon which such option become exercisable; | |
| • | the exercise price of options; |
76
Table of Contents
| • | the duration of options; | |
| • | the conditions and limitations applicable to the exercise of each option; and | |
| • | the number of shares of common stock subject to any restricted stock award and the terms and conditions of such awards. | |
Each of our plans will be effective for ten years following adoption, unless previously terminated. We do not intend to grant any further options or awards under the 2002 plan or 2003 plan upon the completion of this offering, but the vesting and effectiveness of awards previously granted will continue. Pursuant to its terms, no awards may be granted under the 2004 plan after March 29, 2014, but the vesting and effectiveness of awards previously granted may extend beyond that date.
Our compensation committee may at any time modify or amend the plans, except that:
| • | under the 2002 plan and the 2003 plan, if the compensation committee determines such amendment requires shareholder approval, the amendment shall be subject to obtaining such shareholder approval; and | |
| • | under the 2004 plan, no award granted under the plan intended to comply with Section 162(m) shall, after the date of such amendment, become exercisable, realizable or vested, as applicable to such award, unless such amendment is approved by our stockholders as required by Section 162(m). |
The 2002 plan, 2003 plan and 2004 plan each provides that in the event of a merger or other acquisition event, the compensation committee is authorized, in its discretion, to take one or more of the following actions:
| • | provide for outstanding options or other awards to be assumed or substituted for by the acquiring or succeeding entity; | |
| • | provide that unexercised options or other awards will become exercisable in full and will terminate immediately prior to the consummation of such transaction unless previously exercised; | |
| • | in the case of the 2004 plan, provide that outstanding awards shall become realizable or deliverable, or restrictions applicable to an award shall lapse; | |
| • | provide for per share cash payment to the optionees equal to, in the case of the 2002 plan and the 2003 plan, the fair market value per share of such options or other awards less the exercise price per share of such option or other award, and, in the case of the 2004 plan, the cash per share received by the holders of common stock less the exercise price per share of such option or other award; or | |
| • | provide that, immediately prior to such transaction, all or a portion of the unexercised options will become exercisable in full; or | |
| • | in the case of the 2004 plan, provide that, in connection with a liquidation or dissolution of Alnylam, awards shall convert into the right to receive liquidation proceeds. |
Our rights under the terms of outstanding restricted stock granted under the 2004 plan will inure to the benefit of the surviving or succeeding entity and will continue to apply to any cash or other property into which shares were convertible as a result of such transaction.
77
Table of Contents
2004 Employee Stock Purchase Plan
Our 2004 employee stock purchase plan, or 2004 ESPP, was adopted by our board of directors in March 2004 and approved by our stockholders in 2004. The 2004 ESPP will become effective upon completion of this offering and authorizes the issuance of up to a total of 600,000 shares of our common stock to participating employees.
All of our employees, including our directors who are employees, who meet the following criteria are eligible to participate in the purchase plan if:
| • | such person is employed for more than 20 hours per week and for more than five months in a calendar year; | |
| • | such person is employed for at least six months prior to enrolling in the 2004 ESPP; | |
| • | such person is employed on the first day of the applicable offering period under the 2004 ESPP; and | |
| • | in the case of an executive officer, such person is not considered a “highly compensated individual” under Section 414(q) of the Internal Revenue Code. | |
Employees who would immediately after the grant own 5% or more of the total combined voting power or value of our stock are not eligible to participate in the purchase plan.
We will make one or more offerings to our employees to purchase stock under the 2004 ESPP. Our first offering commencement date will begin on the later of November 1, 2004 or the first date on which trading of our common stock commences on the NASDAQ National Market in connection with this offering. Subsequent offerings will begin each November 1 (or the first business day thereafter), unless otherwise determined by our board of directors. Each offering commencement date will begin a twelve-month period, unless otherwise determined by our board of directors, during which payroll deductions will be made and held for the purchase of our common stock at the end of the purchase plan period.
On the first day of a designated payroll deduction period, or offering period, we will grant to each eligible employee who has elected to participate in the purchase plan an option to purchase shares of our common stock. The employee may authorize up to a maximum of 15% of his or her base pay to be deducted by us during the offering period. On the last day of the offering period, the employee is deemed to have exercised the option, at the option exercise price, to the extent of accumulated payroll deductions. Under the terms of the purchase plan, the option exercise price is an amount equal to 85% of the closing price, as defined in the purchase plan, per share of our common stock on either the first day or the last day of the offering period, whichever is lower. In no event may an employee purchase in any one offering period a number of shares that exceeds the number of shares determined by dividing (a) the product of $2,083 and the number of full months in the offering period by (b) the closing price of a share of our common stock on the commencement date of the offering period. Our board of directors may, in its discretion, choose an offering period of 12 months or less for each offering and may choose a different offering period for each offering.
An employee who is not a participant on the last day of the offering period is not entitled to exercise any option, and the employee’s accumulated payroll deductions will be refunded. An employee’s rights under the purchase plan terminate upon voluntary withdrawal from the purchase plan at any time, or when the employee ceases employment for any reason, except that upon termination of employment because of death, the employee’s beneficiary has certain rights to elect to exercise the option to purchase the shares that the accumulated payroll deductions in the employee’s account would purchase at the date of death.
Because the participation in the purchase plan is voluntary, we cannot now determine the number of shares of our common stock to be purchased by any particular current executive officer, by all current executive officers as a group or by non-executive employees as a group.
401(k) Plan
Our employee savings plan is qualified under Section 401 of the Internal Revenue Code. Our employees may elect to reduce their current compensation by up to the statutorily prescribed annual limit and have the amount of such reduction contributed to the 401(k) plan. We may make matching or additional contributions to the 401(k) plan in amounts to be determined annually by our board of directors.
78
Table of Contents
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Since our incorporation, we have engaged in the following transactions with our directors, officers and holders of more than five percent of our voting securities.
Preferred Stock Issuances
Issuance of Series A Convertible Preferred Stock
On July 30, 2003 and July 31, 2003, we sold an aggregate of 3,000,010 shares of Series A convertible preferred stock at a price per share of $1.00 for an aggregate purchase price of $3,000,010. All shares of our Series A convertible preferred stock will be automatically converted into 3,000,010 shares of our common stock upon completion of this offering. All of these 3,000,010 shares were sold to the following holders of more than five percent of our voting securities:
| Shares of Series A | ||||||||
| Convertible | ||||||||
| Name | Preferred Stock | Purchase Price | ||||||
| Polaris Venture Partners(1) | 1,000,010 | $ | 1,000,010 | |||||
| Abingworth Bioventures(2) | 1,000,000 | 1,000,000 | ||||||
| Cardinal Health Partners(3) | 1,000,000 | 1,000,000 | ||||||
| Total | 3,000,010 | $ | 3,000,010 | |||||
| (1) | Consists of 959,955 shares held by Polaris Venture Partners III, 24,925 shares held by Polaris Entrepreneurs Fund III and 15,130 shares sold to Polaris Founders Fund III. Christoph H. Westphal, M.D., Ph.D., a director of Alnylam, is a member of Polaris Venture Management Co. III, LLC, the General Partner of these Polaris entities. |
| (2) | Consists of 502,000 shares held by Abingworth Bioventures III A, 306,440 shares held by Abingworth Bioventures III B, 183,560 shares held by Abingworth Bioventures III C and 8,000 shares held by Abingworth Bioventures Executives III. John E. Berriman, a director of Alnylam, is a Director of Abingworth Management Limited, the Manager of these Abingworth Bioventures entities. |
| (3) | Consists of 1,000,000 shares held by CHP II, L.P. John K. Clarke, the Chairman of our board of directors, is the Managing General Partner of CHP II Management, LLC, the General Partner of CHP II, L.P. |
79
Table of Contents
Issuance of Series B Convertible Preferred Stock
On July 30, 2003 and July 31, 2003, we sold an aggregate of 16,561,845 shares of Series B convertible preferred stock at a price per share of $2.50 for an aggregate purchase price of $41,404,612. All shares of our Series B convertible preferred stock will be automatically converted into 16,561,845 shares of our common stock upon completion of this offering. Of these 16,561,845 shares, an aggregate of 15,580,000 shares were sold to the following directors, officers and holders of more than five percent of our voting securities:
| Shares of Series B | ||||||||
| Convertible | ||||||||
| Name | Preferred Stock | Purchase Price | ||||||
| Polaris Venture Partners(1) | 3,640,000 | $ | 9,100,000 | |||||
| Arch Venture Fund(2) | 3,300,000 | 8,250,000 | ||||||
| Atlas Venture(3) | 3,300,000 | 8,250,000 | ||||||
| Abingworth BioVentures(4) | 2,760,000 | 6,900,000 | ||||||
| Cardinal Health Partners(5) | 2,440,000 | 6,100,000 | ||||||
| Paul R. Schimmel, Ph.D. | 60,000 | 150,000 | ||||||
| Phillip A. Sharp, Ph.D. | 60,000 | 150,000 | ||||||
| John G. Conley | 20,000 | 50,000 | ||||||
| Total | 15,580,000 | $ | 38,950,000 | |||||
| (1) | Consists of 3,494,200 shares held by Polaris Venture Partners III, 90,728 shares held by Polaris Entrepreneurs Fund III and 55,072 shares held by Polaris Founders Fund III. Christoph H. Westphal, M.D., Ph.D., a director of Alnylam, is a member of Polaris Venture Management Co. III, LLC, the General Partner of these Polaris entities. |
| (2) | Consists of 3,278,220 shares held by Arch Venture Fund V, L.P. and 21,780 shares held by Arch V Entrepreneurs Fund. |
| (3) | Consists of 1,043,414 shares held by Atlas Venture Fund V, L.P., 129,609 shares held by Atlas Venture Parallel Fund V-A, C.V., 129,609 shares held by Atlas Venture Parallel Fund V-B, C.V., 17,368 shares held by Atlas Venture Entrepreneur Fund V, 1,895,164 shares held by Atlas Venture Fund VI, L.P., 50,135 shares held by Atlas Venture Entrepreneurs Fund VI, L.P. and 34,701 shares held by Atlas Venture Fund VI GmbH & Co. KG. Peter Barrett, Ph.D., a director of Alnylam, is a Senior Partner of Atlas Venture. |
| (4) | Consists of 1,385,520 shares held by Abingworth Bioventures III A, 845,774 shares held by Abingworth Bioventures III B, 506,626 shares held by Abingworth Bioventures III C and 22,080 shares held by Abingworth Bioventures Executives III. John E. Berriman, a director of Alnylam, is a partner of Abingworth Management Limited, the Manager of these Abingworth entities. |
| (5) | Consists of 2,440,000 shares held by CHP II, L.P. John K. Clarke, the Chairman of our board of directors is Managing General Partner of CHP II Management, LLC, the General Partner of CHP II, L.P. |
80
Table of Contents
Issuance of Series C Convertible Preferred Stock.
On September 8, 2003 and October 9, 2003, we sold an aggregate of 1,504,825 shares of Series C convertible preferred stock at a price per share of $5.00 for an aggregate purchase price of $7,524,125. All shares of our Series C convertible preferred stock will be automatically converted into 1,504,825 shares of our common stock upon completion of this offering. Of these 1,504,825 shares, an aggregate of 504,825 shares were sold to the following holders of more than five percent of our voting securities:
| Shares of Series C | ||||||||
| Convertible | ||||||||
| Name | Preferred Stock | Purchase Price | ||||||
| Polaris Venture Partners(1) | 129,998 | $ | 649,990 | |||||
| Abingworth BioVentures(2) | 103,972 | 519,860 | ||||||
| Cardinal Health Partners(3) | 92,803 | 464,015 | ||||||
| Arch Venture Fund(4) | 89,026 | 445,130 | ||||||
| Atlas Venture(5) | 89,026 | 445,130 | ||||||
| Total | 504,825 | $ | 2,524,125 | |||||
| (1) | Consists of 124,791 shares held by Polaris Venture Partners III, 3,240 shares held by Polaris Entrepreneurs Fund III and 1,967 shares held by Polaris Founders Fund III. Christoph H. Westphal, M.D., Ph.D., a director of Alnylam, is a member of Polaris Venture Management Co. III, LLC, the General Partner of these Polaris entities. |
| (2) | Consists of 52,194 shares held by Abingworth Bioventures III A, 31,861 shares held by Abingworth Bioventures III B, 19,085 shares held by Abingworth Bioventures III C and 832 shares held by Abingworth Bioventures Executives III. John E. Berriman, a director of Alnylam, is a partner of Abingworth Management Limited, the Manager of these Abingworth entities. |
| (3) | Consists of 92,803 shares held by CHP II, L.P. John K. Clarke, the Chairman of our board of directors, is Managing General Partner of CHP II Management, LLC, the General Partner of CHP II, L.P. |
| (4) | Consists of 88,438 shares held by Arch Venture Fund V, L.P. and 588 shares held by Arch V Entrepreneurs Fund. |
| (5) | Consists of 28,149 shares sold to Atlas Ventures Fund V, L.P., 3,496 shares held by Atlas Venture Parallel Fund V-A, 3,496 shares held by Atlas Venture Parallel Fund V-B, C.V., 469 shares held by Atlas Venture Entrepreneurs Fund V, L.P., 50,927 shares held by Atlas Venture Fund VI, 1,557 shares held by Atlas Venture Entrepreneurs Fund VI, L.P. and 932 shares held by Atlas Venture Fund VI GmbH & Co. KG. Peter Barrett, Ph.D., a director of Alnylam, is a Senior Partner of Atlas Venture. |
81
Table of Contents
Registration Rights
The holders of 22,733,347 shares of our common stock are entitled to require us to register their shares under the Securities Act. The holders of warrants to purchase 125,000 shares of our common stock will be entitled to include such shares in registration statements that we may file in the future. These rights are provided under the terms of registration rights agreements between us and these holders. These holders include the following directors, officers and holders of more than five percent of our voting securities and their affiliates:
| Number of | ||||
| Name | Shares | |||
| Polaris Venture Partners | 4,770,008 | |||
| Abingworth BioVentures | 3,863,972 | |||
| Arch Venture Fund | 3,389,026 | |||
| Atlas Venture | 3,389,026 | |||
| Cardinal Health Partners | 3,532,803 | |||
| Paul R. Schimmel, Ph.D. | 60,000 | |||
| Phillip A. Sharp, Ph.D. | 60,000 | |||
| Total | 19,064,835 | |||
The holders of registration rights in connection with this offering have waived their right to participate in this offering.
Other Considerations
We have adopted a policy providing that all material transactions between us and our officers, directors and other affiliates must be:
| • | approved by a majority of the members of our board of directors and by a majority of the disinterested members of our board of directors; and | |
| • | on terms no less favorable to us than those that we believe could be obtained from unaffiliated third parties. |
82
Table of Contents
PRINCIPAL STOCKHOLDERS
The following table sets forth information regarding beneficial ownership of our common stock as of March 31, 2004 by:
| • | each person, or group of affiliated persons, known to us to be the beneficial owner of more than 5% of the outstanding shares of our common stock; | |
| • | each of our directors and the named executive officers; and | |
| • | all of our directors and executive officers as a group. |
Beneficial ownership is determined in accordance with the rules of the SEC, and includes voting or investment power with respect to shares. Shares of common stock issuable under stock options that are exercisable within 60 days after March 31, 2004 are deemed outstanding for the purpose of computing the percentage ownership of the person holding the options but are not deemed outstanding for the purpose of computing the percentage ownership of any other person.
Unless otherwise indicated below, to our knowledge, all persons named in the table have sole voting and investment power with respect to their shares of common stock, except to the extent authority is shared by spouses under community property laws. The percentage of common stock outstanding reflects the conversion, upon the closing of this offering, of all outstanding shares of convertible preferred stock into an aggregate of 22,733,347 shares of common stock. The number of shares of common stock deemed outstanding after this offering includes the shares of common stock being offered for sale in this offering but assumes no exercise of the underwriters’ over-allotment option.
| Number of Securities | ||||||||||||||||
| Beneficially Owned | Percentage Owned | |||||||||||||||
| Prior to Offering | ||||||||||||||||
| Before the | After the | |||||||||||||||
| Name and Address of Beneficial Owner(1) | Stock | Options(2) | Offering | Offering | ||||||||||||
Holders of more than 5% of our voting securities | ||||||||||||||||
| Polaris Venture Partners(3) | 4,770,008 | — | % | % | ||||||||||||
| Abingworth BioVentures(4) | 3,958,016 | — | ||||||||||||||
| Cardinal Health Partners(5) | 3,532,803 | — | ||||||||||||||
| Arch Venture Fund(6) | 3,389,026 | — | ||||||||||||||
| Atlas Venture(7) | 3,389,026 | — | ||||||||||||||
Directors and Named Executive Officers | ||||||||||||||||
| John M. Maraganore, Ph.D.(8) | — | 531,687 | ||||||||||||||
| Peter Barrett, Ph.D.(7) | 3,389,026 | — | ||||||||||||||
| John E. Berriman(4) | 3,958,016 | — | ||||||||||||||
| John K. Clarke(5) | 3,532,803 | — | ||||||||||||||
| Paul R. Schimmel, Ph.D. | 452,000 | — | ||||||||||||||
| Phillip A. Sharp, Ph.D. | 480,000 | — | ||||||||||||||
| Kevin P. Starr | — | — | ||||||||||||||
| Christoph H. Westphal, M.D., Ph.D.(3) | 4,770,008 | — | ||||||||||||||
| Vincent J. Miles, Ph.D. | — | — | ||||||||||||||
| Thomas R. Ulich, M.D. | — | — | ||||||||||||||
| John G. Conley | 225,500 | — | ||||||||||||||
| All current executive officers and directors as a group (11 individuals)(8) | 16,709,853 | 531,687 | % | % | ||||||||||||
83
Table of Contents
| * | Represents beneficial ownership of less than one percent of common stock. |
| (1) | Unless otherwise indicated, the address of each shareholder is c/o Alnylam Pharmaceuticals, Inc., 790 Memorial Drive, Cambridge, MA 02139. |
| (2) | All stock options granted by Alnylam prior to the completion of this offering were subject to a right of early exercise, pursuant to which an optionee could exercise unvested stock options for shares of restricted stock. However, for purposes of this table, options that will not vest within 60 days after March 31, 2004 have not been deemed exercisable or outstanding. |
| (3) | Consists of 4,578,946 shares held by Polaris Venture Partners III, 118,893 shares held by Polaris Entrepreneurs Fund III and 72,169 shares held by Polaris Founders Fund III. |
North Star Ventures directly or indirectly provides investment advisory services to various venture capital funds, including Polaris Venture Partners III, Polaris Entrepreneurs Fund III and Polaris Founders Fund III. The respective General Partners of these funds exercise sole voting and investment power with respect to the shares held by such funds.
The members of North Star Ventures are members of Polaris Venture Management Co. III, LLC, the General Partner of Polaris Venture Partners III, Polaris Entrepreneurs Fund III and Polaris Founders Fund III. As members of these funds, they may be deemed to share voting and investment powers for the shares held by the funds. These principals disclaim beneficial ownership of all such shares except to the extent of their proportionate pecuniary interests in such shares.
Christoph H. Westphal, M.D., Ph.D., a director of Alnylam, is a member of Polaris Venture Management Co. III, LLC, the General Partner of Polaris Venture Partners III, Polaris Entrepreneurs Fund III and Polaris Founders Fund III. As a member of these funds, he may be deemed to share voting and investment powers with respect to the shares held by these funds except to the extent of his proportionate pecuniary interest therein.
| (4) | Consists of 1,986,924 shares held by Abingworth Bioventures III A, 1,212,894 shares held by Abingworth Bioventures III B, 726,534 shares held by Abingworth Bioventures III C and 31,664 shares held by Abingworth Bioventures Executives III. John E. Berriman, a director of Alnylam, is a partner of Abingworth Management Limited, the Manager of the other Abingworth entities listed in this footnote. |
| (5) | Consists of 3,532,803 shares held by CHP II, L.P. John K. Clarke, the Chairman of our board of directors, is the Managing General Partner of CHP II Management, LLC, the General Partner of CHP II, L.P. Mr. Clarke, together with the other general partners of CHP II Management LLC, share voting power and investment control with respect to the shares held by CHP II, L.P. Mr. Clarke may be deemed to beneficially own the shares held by CHP II, L.P. although he disclaims beneficial ownership except to the extent of his proportionate partnership interest therein. |
| (6) | Consists of 3,366,658 shares held by Arch Venture Fund V and 22,368 shares held by Arch V, L.P. Entrepreneurs Fund. |
| (7) | Consists of 1,071,563 shares held by Atlas Venture Parallel Fund V, L.P., 133,105 shares held by Atlas Venture Parallel Fund V-A, C.V., 133,105 shares held by Atlas Venture Parallel Fund V-B, C.V., 17,837 shares held by Atlas Venture Entrepreneur Fund V, L.P., 1,946,091 shares held by Atlas Venture Fund VI, L.P., 51,692 shares held by Atlas Venture Entrepreneur Fund VI, L.P. and 35,633 shares held by Atlas Venture Fund VI GmbH & Co. KG. Peter Barrett, Ph.D., a director of Alnylam, is a Senior Partner of Atlas Venture. |
| (8) | Includes 200,000 shares subject to an option issued to Dr. Maraganore that will vest in full upon the consummation of this offering. |
84
Table of Contents
DESCRIPTION OF CAPITAL STOCK
The following description of our capital stock and provisions of our amended and restated certificate of incorporation and amended and restated bylaws are summaries and are qualified by reference to the amended and restated certificate of incorporation and the amended and restated bylaws that will become effective upon closing of this offering. Copies of these documents will be filed with the SEC as exhibits to our registration statement, of which this prospectus forms a part. The descriptions of the common stock and preferred stock reflect changes to our capital structure that will occur upon the closing of this offering.
Upon the completion of this offering, our authorized capital stock will consist of 125,000,000 shares of common stock, par value $0.0001 per share, and 5,000,000 shares of preferred stock, par value $0.0001 per share, all of which shares of preferred stock will be undesignated. Our board of directors may establish the rights and preferences of the preferred stock from time to time. As of March 31, 2004, after giving effect to the conversion of all outstanding shares of convertible preferred stock into shares of common stock, there were 27,138,388 shares of common stock issued and outstanding. As of March 31, 2004, there were 63 stockholders of record of our capital stock.
Common Stock
Holders of common stock are entitled to one vote for each share held on all matters submitted to a vote of stockholders and do not have cumulative voting rights. Accordingly, holders of a majority of the shares of common stock entitled to vote in any election of directors may elect all of the directors standing for election. Holders of common stock are entitled to receive proportionately any dividends as may be declared by our board of directors, subject to any preferential dividend rights of outstanding preferred stock. Upon our liquidation, dissolution or winding up, the holders of common stock are entitled to receive proportionately our net assets available after the payment of all debts and other liabilities and subject to the prior rights of any outstanding preferred stock. Holders of common stock have no preemptive, subscription, redemption or conversion rights. Our outstanding shares of common stock are, and the shares offered by us in this offering will be, when issued and paid for, fully paid and nonassessable. The rights, preferences and privileges of holders of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate and issue in the future.
Preferred Stock
Under the terms of our amended and restated certificate of incorporation, our board of directors is authorized to issue shares of preferred stock in one or more series without stockholder approval. Our board of directors has the discretion to determine the rights, preferences, privileges and restrictions, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences, of each series of preferred stock.
The purpose of authorizing our board of directors to issue preferred stock and determine its right and preferences is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing flexibility in connection with possible future acquisitions and other corporate purposes, will affect, and may adversely affect, the rights of holders of any preferred stock that may be issued in the future. It is not possible to state the actual effect of the issuance of any shares of preferred stock on the rights of holders of common stock until the board of directors determines the specific rights attached to that preferred stock. The effects of issuing preferred stock could include one or more of the following:
| • | restricting dividends on the common stock; | |
| • | diluting the voting power of the common stock; | |
| • | impairing the liquidation rights of the common stock; or | |
| • | delaying or preventing changes in control or management of Alnylam. |
We have no present plans to issue any shares of preferred stock.
85
Table of Contents
Warrants
As of March 31, 2004, Silicon Valley Bancshares held a warrant to purchase an aggregate of 25,000 shares of common stock at an exercise price of $2.50 per share. This warrant expires on December 18, 2012. In addition, as of March 31, 2004, Lighthouse Capital Partners V, L.P. and a related entity held warrants to purchase an aggregate of 100,000 shares of common stock at an exercise price of $5.00 per share. These warrants expire on March 30, 2011. The holders of these warrants have registration rights that are outlined below under the heading “Registration Rights.”
Registration Rights
The holders of 22,733,347 shares of common stock, after giving effect to the conversion of outstanding convertible preferred stock into shares of common stock upon completion of this offering, have rights to require us to file registration statements under the Securities Act or to include their shares in registration statements that we may file in the future for ourselves or other stockholders. The holders of warrants to purchase 125,000 shares of our common stock will be entitled to include shares issued upon exercise of the warrants in registration statements that we may file in the future. The holders of registration rights in connection with this offering have waived their right to participate in this offering.
At any time after twelve months after the closing of this offering, holders of at least 33% of the shares of our common stock having registration rights may demand that we register all or a portion of their common stock for sale under the Securities Act. We are required to effect only two of these registrations. However, if at any time we become eligible to file a registration statement on Form S-2 or Form S-3, or any successor form, holders of registration rights may make unlimited requests for us to effect a registration on such forms of their common stock having an aggregate offering price of at least $3,000,000.
In addition, if at any time after this offering we register any shares of common stock, either for our own account or for the account of other security holders, the holders of registration rights are entitled to notice of the registration and to include all or a portion of their common stock in the registration. A holder’s right to demand or include shares in a registration is subject to the right of the underwriters to limit the number of shares included in the offering.
Anti-Takeover Provisions of Delaware Law, our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws
We are subject to the provisions of Section 203 of the General Corporation Law of Delaware. Subject to certain exceptions, Section 203 prohibits a publicly held Delaware corporation from engaging in a business combination with an interested stockholder for a period of three years after the person became an interested stockholder, unless the interested stockholder attained such status with the approval of our board of directors or the business combination is approved in a prescribed manner. A business combination includes, among other things, a merger or consolidation involving us and the interested stockholder and the sale of more than 10% of our assets. In general, an interested stockholder is any entity or person beneficially owning 15% or more of our outstanding voting stock and any entity or person affiliated with or controlling or controlled by such entity or person.
Our amended and restated certificate of incorporation and our amended and restated bylaws divide our board of directors into three classes with staggered three-year terms. In addition, our amended and restated certificate of incorporation provides that directors may be removed only for cause by the affirmative vote of the holders of 75% of our shares of capital stock entitled to vote. Under our amended and restated certificate of incorporation, any vacancy on our board of directors, including a vacancy resulting from an enlargement of our board of directors, may only be filled by vote of a majority of our directors then in office. The classification of our board of directors and the limitations on the removal of directors and filling of vacancies could make it more difficult for a third party to acquire, or discourage a third party from acquiring, control of us.
86
Table of Contents
Our amended and restated certificate of incorporation and our amended and restated bylaws also provide that any action required or permitted to be taken by our stockholders at an annual meeting or special meeting of stockholders may only be taken if it is properly brought before the meeting and may not be taken by written action in lieu of a meeting. Our amended and restated certificate of incorporation and our amended and restated bylaws further provide that, except as otherwise required by law, special meetings of the stockholders may only be called by the chairman of the board, chief executive officer or our board of directors. In addition, our amended and restated bylaws establish an advance notice procedure for stockholder proposals to be brought before an annual meeting of stockholders, including proposed nominations of persons for election to the board of directors. Stockholders at an annual meeting may only consider proposals or nominations specified in the notice of meeting or brought before the meeting by or at the direction of the board of directors or by a stockholder of record on the record date for the meeting, who is entitled to vote at the meeting and who has delivered timely written notice in proper form to our secretary of the stockholder’s intention to bring such business before the meeting. These provisions could have the effect of delaying until the next stockholders’ meeting stockholder actions that are favored by the holders of a majority of our outstanding voting securities. These provisions may also discourage a third party from making a tender offer for our common stock, because even if it acquired a majority of our outstanding voting securities, the third party would be able to take action as a stockholder, such as electing new directors or approving a merger, only at a duly called stockholders’ meeting, and not by written consent.
The General Corporation Law of Delaware provides generally that the affirmative vote of a majority of the shares entitled to vote on any matter is required to amend a corporation’s certificate of incorporation or bylaws, unless a corporation’s certificate of incorporation or bylaws, as the case may be, requires a greater percentage. Our amended and restated certificate of incorporation and amended and restated bylaws require the affirmative vote of the holders of at least 75% of the shares of our capital stock issued and outstanding and entitled to vote to amend or repeal any of the provisions described in the prior two paragraphs.
Limitation of Liability and Indemnification
Our amended and restated certificate of incorporation contains provisions permitted under the General Corporation Law of Delaware relating to the liability of directors. The provisions eliminate a director’s liability for monetary damages for a breach of fiduciary duty, except in circumstances involving wrongful acts, such as the breach of a director’s duty of loyalty or acts or omissions that involve intentional misconduct or a knowing violation of law. Further, our amended and restated certificate of incorporation contains provisions to indemnify our directors and officers to the fullest extent permitted by the General Corporation Law of Delaware.
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is EquiServe Trust Company.
NASDAQ National Market
We have applied for the quotation of our common stock on the NASDAQ National Market under the symbol “ALNY.”
87
Table of Contents
SHARES ELIGIBLE FOR FUTURE SALE
Sales of substantial amounts of our common stock in the public market could adversely affect prevailing market prices of our common stock. Furthermore, since some shares of common stock will not be available for sale shortly after this offering because of the contractual and legal restrictions on resale described below, sales of substantial amounts of common stock in the public market after these restrictions lapse could adversely affect the prevailing market price and our ability to raise equity capital in the future.
Prior to this offering, there has been no public market for our common stock. Upon completion of this offering, we will have outstanding an aggregate of shares of our common stock assuming no exercise of outstanding options or warrants. Of these shares, the shares sold in this offering will be freely tradable without restrictions or further registration under the Securities Act, unless those shares are purchased by affiliates as that term is defined in Rule 144 under the Securities Act. The remaining shares of common stock held by existing stockholders are restricted securities as that term is defined in Rule 144 under the Securities Act or are subject to the contractual restrictions described below. Of these remaining securities:
| • | shares which are not subject to the 180-day lock-up period described below may be sold immediately after completion of this offering; | |
| • | additional shares which are not subject to the 180-day lock-up period described below may be sold beginning 90 days after the effective date of this offering; and | |
| • | additional shares may be sold upon expiration of the 180-day lock-up period described below. |
Restricted securities may be sold in the public market only if registered or if they qualify for an exemption from registration under Rule 144 or 701 under the Securities Act, which rules are summarized below.
Rule 144
In general, under Rule 144, beginning 90 days after the date of this prospectus, a person who has beneficially owned shares of our common stock for at least one year would be entitled to sell within any three-month period a number of shares that does not exceed the greater of:
| • | 1% of the number of shares of common stock then outstanding, which will equal approximately shares immediately after this offering; or | |
| • | the average weekly trading volume of the common stock on the NASDAQ National Market during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale. |
Sales under Rule 144 are also subject to manner of sale provisions and notice requirements and to the availability of current public information about us.
Rule 144(k)
Common stock eligible for sale under Rule 144(k) may be sold immediately upon the completion of this offering. In general, under Rule 144(k), a person may sell shares of common stock acquired from us immediately upon completion of this offering, without regard to manner of sale, the availability of public information or volume, if:
| • | the person is not our affiliate and has not been our affiliate at any time during the three months preceding such a sale; and | |
| • | the person has beneficially owned the shares proposed to be sold for at least two years, including the holding period of any prior owner other than an affiliate. |
Rule 701
In general, under Rule 701 of the Securities Act, any of our employees, consultants or advisors who purchase shares from us in connection with a qualified compensatory stock plan or other written agreement is
88
Table of Contents
Lock-up Agreements
Our officers and directors and stockholders owning an aggregate of shares of common stock have signed lock-up agreements under which they agreed not to offer, sell, pledge, contract to sell, sell short, grant any option in or otherwise dispose of, or enter into any hedging transaction with respect to, any shares of our common stock or any securities convertible into or exercisable or exchangeable for shares of our common stock beneficially owned by them, for a period ending 180 days after the date of this prospectus. The foregoing does not prohibit open market purchases and sales of our common stock by such holders after the completion of this offering and transfers or dispositions by our officers, directors and stockholders can be made sooner:
| • | with the written consent of Banc of America Securities LLC; | |
| • | as a gift or by will or intestacy; | |
| • | to immediate family members; and | |
| • | to any trust for the direct or indirect benefit of the holder or his or her immediately family. |
Registration Rights
Upon completion of this offering, the holders of 22,733,347 shares of our common stock, or their transferees, have rights to require or participate in the registration of those shares under the Securities Act. The holders of warrants to purchase 125,000 shares of our common stock will also be entitled to participate in such registration with respect to the shares of common stock issuable upon exercise of such warrants. For a detailed description of these registration rights see “Description of Capital Stock — Registration Rights”.
Stock Options
We intend to file a registration statement under the Securities Act covering the 7,638,219 shares of common stock reserved for issuance under our 2002 plan, 2003 plan, 2004 plan and 2004 ESPP as of March 31, 2004. That registration statement is expected to become effective upon filing with the SEC. Accordingly, common stock registered under that registration statement will, subject to vesting provisions and limitations as to the volume of shares that may be sold by our affiliates under Rule 144 described above, be available for sale in the open market immediately after the 180-day lock-up period expires.
As of March 31, 2004, options to purchase 3,691,618 shares of common stock were issued and outstanding at a weighted average exercise price of $0.32 per share. Upon the expiration of the lock-up period described above, at least shares of common stock will be subject to vested options, based on options outstanding as of March 31, 2004.
Warrants
Upon completion of this offering, there will be warrants outstanding to purchase 125,000 shares of common stock at a weighted average exercise price of $4.50 per share. Holders of these warrants have registration rights that are outlined above under the heading “Registration Rights.”
Effect of Sales of Shares
Prior to this offering, there has been no public market for our common stock, and no prediction can be made as to the effect, if any, that market sales of shares of common stock or the availability of shares for sale will have on the market price of our common stock prevailing from time to time. Nevertheless, sales of significant numbers of shares of our common stock in the public market after the completion of this offering could adversely affect the market price of our common stock and could impair our future ability to raise capital through an offering of our equity securities.
89
Table of Contents
MATERIAL U.S. FEDERAL TAX CONSIDERATIONS FOR NON-U.S. HOLDERS OF OUR COMMON STOCK
The following is a general discussion of the material U.S. federal income and estate tax considerations applicable to non-U.S. holders with respect to their ownership and disposition of shares of our common stock. This discussion is for general information only and is not tax advice. Accordingly, all prospective non-U.S. holders of our common stock should consult their own tax advisors with respect to the U.S. federal, state, local and non-U.S. tax consequences of the purchase, ownership and disposition of our common stock. In general, a non-U.S. holder means a beneficial owner of our common stock who is not for U.S. federal income tax purposes:
| • | an individual who is a citizen or resident of the U.S.; | |
| • | a corporation, or any other organization taxable as a corporation for U.S. federal tax purposes, created or organized in the U.S. or under the laws of the U.S. or of any state thereof or the District of Columbia; or | |
| • | an estate or trust, the income of which is included in gross income for U.S. federal income tax purposes regardless of its source. |
This discussion is based on current provisions of the U.S. Internal Revenue Code of 1986, as amended, existing and proposed U.S. Treasury Regulations promulgated thereunder, current administrative rulings and judicial decisions, in effect as of the date of this prospectus, all of which are subject to change or to differing interpretation, possibly with retroactive effect. Any change could alter the tax consequences to non-U.S. holders described in this prospectus. We assume in this discussion that a non-U.S. holder holds shares of our common stock as a capital asset (generally property held for investment).
This discussion does not address all aspects of U.S. federal income and estate taxation that may be relevant to a particular non-U.S. holder in light of that non-U.S. holder’s individual circumstances nor does it address any aspects of U.S. state, local or non-U.S. taxes. This discussion also does not consider any specific facts or circumstances that may apply to a non-U.S. holder and does not address the special tax rules applicable to particular non-U.S. holders, such as:
| • | insurance companies; | |
| • | tax-exempt organizations; | |
| • | financial institutions; | |
| • | brokers or dealers in securities; | |
| • | partnerships or other pass-through entities; | |
| • | regulated investment companies; | |
| • | pension plans; | |
| • | owners of more than 5% of our common stock; | |
| • | owners that hold our common stock as part of a straddle, hedge, conversion transaction, synthetic security or other integrated investment; and | |
| • | certain U.S. expatriates. |
There can be no assurance that the Internal Revenue Service, referred to as the IRS, will not challenge one or more of the tax consequences described herein, and we have not obtained, nor do we intend to obtain, an opinion of counsel with respect to the U.S. federal income or estate tax consequences to a non-U.S. holder of the purchase, ownership, or disposition of our common stock.We urge prospective investors to consult with their own tax advisors regarding the U.S. federal, state, local and non-U.S. income and other tax considerations of acquiring, holding and disposing of shares of our common stock.
90
Table of Contents
Distributions on Our Common Stock
We have not declared or paid distributions on our common stock since our inception and do not intend to pay any distributions on our common stock in the foreseeable future. In the event we do pay distributions on our common stock, however, these distributions generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. If a distribution exceeds our current and accumulated earnings and profits, the excess will be treated as a tax-free return of the non-U.S. holder’s investment, up to such holder’s tax basis in the common stock. Any remaining excess will be treated as capital gain, subject to the tax treatment described below in “Gain on Sale, Exchange or Other Disposition of Common Stock.”
Dividends paid to a non-U.S. holder generally will be subject to withholding of U.S. federal income tax at a 30% rate or such lower rate as may be provided by an applicable income tax treaty between the U.S. and such holder’s country of residence. If we determine, at a time reasonably close to the date of payment of a distribution on our common stock, that the distribution will not constitute a dividend because we do not anticipate having current or accumulated earnings and profits, we intend not to withhold any U.S. federal income tax on the distribution as permitted by U.S. Treasury Regulations. If we or another withholding agent withholds tax on such a distribution, a non-U.S. holder may be entitled to a refund of the tax withheld which the non-U.S. holder may claim by filing a U.S. tax return with the IRS.
Dividends that are treated as effectively connected with a trade or business conducted by a non-U.S. holder within the United States (and if an applicable income tax treaty so provides, are also attributable to a permanent establishment or a fixed base maintained by such non-U.S. holder) are generally exempt from the 30% withholding tax if the non-U.S. holder satisfies applicable certification and disclosure requirements. However, such U.S. effectively connected income, net of specified deductions and credits, is taxed at the same graduated U.S. federal income tax rates applicable to U.S. persons. Any U.S. effectively connected income received by a non-U.S. holder that is a corporation may also, under certain circumstances, be subject to an additional branch profits tax at a 30% rate or such lower rate as specified by an applicable income tax treaty between the United States and such holder’s country of residence.
A non-U.S. holder of our common stock who claims the benefit of an applicable income tax treaty between the United States and such holder’s country of residence generally will be required to provide a properly executed IRS Form W-8BEN and satisfy applicable certification and other requirements. Non-U.S. holders are urged to consult their tax advisors regarding their entitlement to benefits under a relevant income tax treaty.
A non-U.S. holder that is eligible for a reduced rate of U.S. withholding tax under an income tax treaty may obtain a refund of any excess amounts withheld by timely filing an appropriate claim with the IRS.
Gain on Sale, Exchange or Other Disposition of Our Common Stock
In general, a non-U.S. holder will not be subject to any U.S. federal income tax or withholding tax on any gain realized upon such holder’s sale, exchange or other disposition of shares of our common stock unless:
| • | the gain is effectively connected with a U.S. trade or business (and if an applicable income tax treaty so provides, is also attributable to a permanent establishment or a fixed base maintained by such non-U.S. holder), in which case the graduated U.S. federal income tax rates applicable to U.S. persons and, if the non-U.S. holder is a foreign corporation, the additional branch profits tax described above in “Distributions on Our Common Stock” may apply; | |
| • | the non-U.S. holder is an individual who is present in the United States for 183 days or more in the taxable year of the disposition and certain other conditions are met, in which case the non-U.S. holder will be subject to a 30% tax on the net gain derived from the disposition, which may be offset by U.S. source capital losses of the non-U.S. holder, if any; or |
91
Table of Contents
| • | we are or have been, at any time during the five-year period preceding such disposition (or the non-U.S. holder’s holding period if shorter) a “U.S. real property holding corporation” unless our common stock is regularly traded on an established securities market and the non-U.S. holder holds no more than 5% of our outstanding common stock, directly or indirectly. If we are determined to be a U.S. real property holding corporation and the foregoing exception does not apply, then a purchaser may withhold 10% of the proceeds payable to a non-U.S. holder from a sale of our common stock and the non-U.S. holder generally will be taxed on its net gain derived from the disposition at the graduated U.S. federal income tax rates applicable to U.S. persons and, if the non-U.S. holder is a foreign corporation, the additional branch profits tax described above in “Distributions on Our Common Stock” may apply. Generally, a corporation is a U.S. real property holding corporation only if the fair market value of its U.S. real property interests equals or exceeds 50% of the sum of the fair market value of its worldwide real property interests plus its other assets used or held for use in a trade or business. Although there can be no assurance, we do not believe that we are, or have been, a U.S. real property holding corporation, or that we are likely to become one in the future. Furthermore, no assurance can be provided that our stock will be regularly traded on an established securities market for purposes of the rules described above. |
U.S. Federal Estate Tax
Shares of our common stock that are owned or treated as owned by an individual non-U.S. holder at the time of death and certain lifetime transfers of an interest in our common stock made by such individual are considered U.S. situs assets and will be included in the individual’s gross estate for U.S. federal estate tax purposes. Such shares, therefore, may be subject to U.S. federal estate tax, unless an applicable estate tax or other treaty provides otherwise.
Backup Withholding and Information Reporting
We must report annually to the IRS and to each non-U.S. holder the gross amount of the distributions on our common stock paid to such holder and the tax withheld, if any, with respect to such distributions. Non-U.S. holders may have to comply with specific certification procedures to establish that the holder is not a U.S. person in order to avoid backup withholding with respect to dividends on our common stock. The gross amount of dividends paid to a non-U.S. holder that fails to certify its non-U.S. holder’s status in accordance with the applicable U.S. Treasury Regulations generally will be reduced by backup withholding at the applicable rate, currently 28%. Dividends paid to non-U.S. holders subject to the U.S. withholding tax, as described above in “Distributions on Our Common Stock,” generally will be exempt from U.S. backup withholding.
Information reporting and backup withholding will generally apply to the proceeds of a disposition of our common stock by a non-U.S. holder effected by or through the U.S. office of any broker, U.S. or foreign, unless the holder certifies its status as a non-U.S. holder and satisfies certain other requirements, or otherwise establishes an exemption. Generally, information reporting and backup withholding will not apply to a payment of disposition proceeds where the transaction is effected outside the U.S. through a non-U.S. office of a non-U.S. broker. However, for information reporting purposes, certain brokers with substantial U.S. ownership or operations generally will be treated in a manner similar to U.S. brokers. Non-U.S. holders should consult their own tax advisors regarding the application of the information reporting and backup withholding rules to them.
Copies of information returns may be made available to the tax authorities of the country in which the non-U.S. holder resides or is incorporated under the provisions of a specific treaty or agreement.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules from a payment to a non-U.S. holder can be refunded or credited against the non-U.S. holder’s U.S. federal income tax liability, if any, provided that an appropriate claim is timely filed with the IRS.
92
Table of Contents
UNDERWRITING
We are offering the shares of common stock described in this prospectus through a number of underwriters. Banc of America Securities LLC, Citigroup Global Markets Inc., Piper Jaffray & Co. and ThinkEquity Partners LLC are the representatives of the underwriters. We have entered into a firm commitment underwriting agreement with the underwriters. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriters, and each underwriter has agreed to purchase, the number of shares of common stock listed next to its name in the following table:
| Underwriter | Number of Shares | ||||
| Banc of America Securities LLC | |||||
| Citigroup Global Markets Inc. | |||||
| Piper Jaffray & Co. | |||||
| ThinkEquity Partners LLC | |||||
| Total | |||||
The underwriting agreement is subject to a number of terms and conditions and provides that the underwriters must buy all of the shares if they buy any of them. The underwriters will sell the shares to the public when and if the underwriters buy the shares from us.
The underwriters initially will offer the shares to the public at the price specified on the cover page of this prospectus. The underwriters may allow a concession of not more than $ per share to selected dealers. The underwriters may also allow, and those dealers may re-allow, a concession of not more than $ per share to some other dealers. If all the shares are not sold at the public offering price, the underwriters may change the public offering price and the other selling terms. The common stock is offered subject to a number of conditions, including:
| • | receipt and acceptance of the common stock by the underwriters; and | |
| • | the underwriters’ right to reject orders in whole or in part. |
Over-Allotment Option.We have granted the underwriters an over-allotment option to buy up to additional shares of our common stock at the same price per share as they are paying for the shares shown in the table above. These additional shares would cover sales of shares by the underwriters that exceed the total number of shares shown in the table above. The underwriters may exercise this option at any time within 30 days after the date of this prospectus. To the extent that the underwriters exercise this option, each underwriter will purchase additional shares from us in approximately the same proportion as it purchased the shares shown in the table above. If purchased, the additional shares will be sold by the underwriters on the same terms as those on which the other shares are sold.
Discounts and Commissions.The following table shows the per share and total underwriting discounts and commissions to be paid to the underwriters by us. These amounts are shown assuming no exercise and full exercise of the underwriters’ option to purchase additional shares.
| Paid by Us | |||||||||
| No Exercise | Full Exercise | ||||||||
| Per Share | $ | $ | |||||||
| Total | $ | $ | |||||||
We estimate that the expenses of the offering to be paid by us, not including the underwriting discounts and commissions, will be approximately $ .
Listing.We expect our common stock to be approved for quotation on the NASDAQ National Market under the symbol “ALNY.”
93
Table of Contents
Stabilization.In connection with this offering, the underwriters may engage in activities that stabilize, maintain or otherwise affect the price of our common stock, including:
| • | stabilizing transactions; | |
| • | short sales; | |
| • | syndicate covering transactions; | |
| • | imposition of penalty bids; and | |
| • | purchases to cover positions created by short sales. |
Stabilizing transactions consist of bids or purchases made for the purpose of preventing or retarding a decline in the market price of our common stock while this offering is in progress. Stabilizing transactions may include making short sales of our common stock, which involves the sale by the underwriters of a greater number of shares of common stock than they are required to purchase in this offering, and purchasing shares of common stock from us or in the open market to cover positions created by short sales. Short sales may be covered shorts, which are short positions in an amount not greater than the underwriters’ over-allotment option referred to above, or may be naked shorts, which are short positions in excess of that amount. Syndicate covering transactions involve purchases of our common stock in the open market after the distribution has been completed in order to cover syndicate short positions.
The underwriters may close out any covered short position either by exercising their over-allotment option, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market compared to the price at which the underwriters may purchase shares pursuant to the over-allotment option.
A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the common stock in the open market that could adversely affect investors who purchased shares of our common stock in this offering. To the extent that the underwriters create a naked short position, they will purchase shares in the open market to cover the position.
The representatives also may impose a penalty bid on underwriters and dealers participating in the offering. This means that the representatives may reclaim from any syndicate member or other dealers participating in the offering the commissions and selling concessions on shares sold by them and purchased by the representatives in stabilizing or short covering transactions.
These activities may have the effect of raising or maintaining the market price of our common stock or preventing or retarding a decline in the market price for our common stock. As a result of these activities the price of or common stock may be higher than the price that otherwise might exist in the open market. If the underwriters commence these activities, they may discontinue them at any time. The underwriters may carry out these transactions on the NASDAQ National Market, in the over-the-counter market or otherwise.
Discretionary Accounts.The underwriters have informed us that they do not expect to make sales to accounts over which they exercise discretionary authority in excess of 5% of the shares of common stock being offered.
IPO Pricing.Prior to this offering, there has been no public market for our common stock. The initial public offering price will be negotiated between us and the representatives of the underwriters. Among the factors to be considered in these negotiations are:
| • | the history of, and prospects for, our company and the industry in which we compete; | |
| • | our past and present financial performance; | |
| • | an assessment of our management; | |
| • | the present state of our development; | |
| • | the prospects for our future earnings; |
94
Table of Contents
| • | the prevailing conditions of the applicable U.S. securities market at the time of this offering; | |
| • | market valuations of publicly traded companies that we and the representatives of the underwriters believe to be comparable to us; and | |
| • | other factors deemed relevant. |
The estimated initial public offering price range set forth on the cover of this prospectus is subject to change as a result of market conditions and other factors.
Lock-up Agreements.We, our officers and directors and stockholders owning an aggregate of shares of common stock have entered into lock-up agreements with the underwriters. Under these agreements, we and each of these persons may not, without the prior written approval of Banc of America Securities LLC, offer, sell, contact to sell or otherwise dispose of or hedge our common stock or securities convertible into or exchangeable for our common stock. These restrictions will be in effect for a period of 180 days after the date of this prospectus. At any time and without notice, Banc of America Securities LLC may, in its sole discretion, release all or some of the securities from these lock-up agreements.
Directed Share Program.At our request, the underwriters have reserved up to % of the common stock being offered by this prospectus for sale to our directors, employees, business associates and related persons at the public offering price. The sales will be made by Banc of America Securities LLC through a directed share program. We do not know if these persons will choose to purchase all or any portion of these reserved shares, but any purchases they do make will reduce the number of shares available to the general public. These persons must commit to purchase no later than the close of business on the day following the date of this prospectus. Any directors, employees or other persons purchasing such reserved shares will be prohibited from disposing of or hedging such shares for a period of at least 180 days after the date of this prospectus.
Indemnification.We will indemnify the underwriters against some liabilities, including liabilities under the Securities Act. If we are unable to provide this indemnification, we will contribute to payments the underwriters may be required to make in respect to those liabilities.
Conflicts/ Affiliates.The underwriters and their affiliates may from time to time engage in future transactions with us and our affiliates and provide services to us and our affiliates in the ordinary course of their business for which services they may in the future receive customary fees.
LEGAL MATTERS
The validity of the shares of common stock we are offering will be passed upon for us by Hale and Dorr LLP, Boston, Massachusetts. Legal matters in connection with this offering will be passed upon for the underwriters by Shearman & Sterling LLP, New York, New York.
EXPERTS
The financial statements as of December 31, 2002 and 2003 and for the period from June 14, 2002 (date of inception) through December 31, 2002, the year ended December 31, 2003 and the cumulative period from June 14, 2002 (date of inception) through December 31, 2003 of Alnylam included in this prospectus have been so included in reliance on the report of PricewaterhouseCoopers LLP, independent accountants, given on the authority of such firm as experts in auditing and accounting.
The financial statements as of December 31, 2001 and 2002 and for the year ended December 31, 2002 and the period from June 14, 2000 (date of inception) through December 31, 2002 of Alnylam Europe, AG included in this prospectus have been so included in reliance on the report of PricewaterhouseCoopers Gesellschaft mit beschränkter Haftung Wirtschaftsprüfungsgesellschaft, independent accountants, given on the authority of such firm as experts in auditing and accounting.
95
Table of Contents
WHERE YOU CAN FIND MORE INFORMATION
We have filed a registration statement on Form S-1 with the SEC for the common stock we are offering by this prospectus. This prospectus does not include all of the information contained in the registration statement. You should refer to the registration statement and its exhibits for additional information. Whenever we make reference in this prospectus to any of our contracts, agreements or other documents, the references are not necessarily complete and you should refer to the exhibits attached to the registration statement for copies of the actual contract, agreement or other document. When we complete this offering, we will also be required to file annual, quarterly and special reports, proxy statements and other information with the SEC.
You can read our SEC filings, including the registration statement, over the Internet at the SEC’s web site at www.sec.gov. You may also read and copy any document we file with the SEC at its public reference facility at 450 Fifth Street, N.W., Washington, D.C. 20549. You may also obtain copies of the documents at prescribed rates by writing to the Public Reference Section of the SEC at 450 Fifth Street, N.W., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference facilities.
96
Table of Contents
INDEX TO FINANCIAL STATEMENTS
ALNYLAM PHARMACEUTICALS, INC.
Periods ended December 31, 2002 and 2003
| Page | ||||
Report of Independent Auditors | F-2 | |||
Consolidated Financial Statements | ||||
| Balance Sheets | F-3 | |||
| Statements of Operations | F-4 | |||
| Statements of Changes in Redeemable Convertible Preferred Stock and Stockholders’ Deficit | F-5 | |||
| Statements of Cash Flows | F-6 | |||
| Notes to Financial Statements | F-8 | |||
RIBOPHARMA AG
Years ended December 31, 2001 and 2002
Report of Independent Auditors | F-34 | |||
Financial Statements | ||||
| Balance Sheets | F-35 | |||
| Statements of Operations | F-36 | |||
| Statements of Changes in Stockholders’ Deficit | F-37 | |||
| Statements of Cash Flows | F-38 | |||
| Notes to Financial Statements | F-39 |
PRO FORMA FINANCIAL DATA
Year ended December 31, 2003
| Pro Forma Financial Data | F-47 | |||
| Unaudited Pro Forma Combined Statements of Operations | F-48 | |||
| Notes to Unaudited Pro Forma Combined Financial Statements of Operations | F-49 |
F-1
Table of Contents
Report of Independent Auditors
To the Board of Directors and Stockholders of
In our opinion, the accompanying consolidated balance sheets and the related consolidated statements of operations, of changes in redeemable convertible preferred stock and stockholders’ deficit and of cash flows present fairly, in all material respects, the financial position of Alnylam Pharmaceuticals, Inc. (a development stage enterprise) at December 31, 2003 and 2002, and the results of its operations and its cash flows for the year ended December 31, 2003 and for the period from inception (June 14, 2002) through December 31, 2002, and cumulatively, for the period from inception (June 14, 2002) through December 31, 2003, in conformity with accounting principles generally accepted in the United States of America. These financial statements are the responsibility of the Company’s management; our responsibility is to express an opinion on these financial statements based on our audits. We conducted our audits of these statements in accordance with auditing standards generally accepted in the United States of America, which require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
/s/ PricewaterhouseCoopers LLP
Boston, Massachusetts
| Feb | ruary 27, 2004, except as to Note 15, |
F-2
Table of Contents
ALNYLAM PHARMACEUTICALS, INC.
Consolidated Balance Sheets
| Pro Forma | ||||||||||||||
| December 31, | December 31, | |||||||||||||
| 2003 | ||||||||||||||
| 2002 | 2003 | (Note 2) | ||||||||||||
| (Unaudited) | ||||||||||||||
| Assets | ||||||||||||||
| Current assets | ||||||||||||||
| Cash and cash equivalents | $ | 15,477 | $ | 23,193 | $ | 23,193 | ||||||||
| Restricted cash | 19 | 373 | 373 | |||||||||||
| Prepaid expenses and other current assets | 23 | 623 | 623 | |||||||||||
| Total current assets | 15,519 | 24,189 | 24,189 | |||||||||||
| Property and equipment, net | 526 | 4,756 | 4,756 | |||||||||||
| Intangible assets, net | — | 3,878 | 3,878 | |||||||||||
| Restricted cash | — | 2,313 | 2,313 | |||||||||||
| Deferred financing costs | 66 | 47 | 47 | |||||||||||
| Total assets | $ | 16,111 | $ | 35,183 | $ | 35,183 | ||||||||
| Liabilities, Redeemable Convertible Preferred Stock and Stockholders’ Equity (Deficit) | ||||||||||||||
| Current liabilities | ||||||||||||||
| Accounts payable | $ | 674 | $ | 1,510 | $ | 1,510 | ||||||||
| Accrued liabilities | 1,999 | 1,443 | 1,443 | |||||||||||
| Current portion of note payable | — | 558 | 558 | |||||||||||
| Deferred revenue | — | 333 | 333 | |||||||||||
| Total current liabilities | 2,673 | 3,844 | 3,844 | |||||||||||
| Deferred revenue | — | 1,556 | 1,556 | |||||||||||
| Note payable, net of current portion | — | 1,301 | 1,301 | |||||||||||
| Total liabilities | 2,673 | 6,701 | 6,701 | |||||||||||
| Commitments and contingencies (Note 12) | ||||||||||||||
| Redeemable convertible preferred stock (Note 8) | 18,084 | 55,189 | — | |||||||||||
| Stockholders’ equity (deficit) | ||||||||||||||
| Common stock, $0.0001 par value, 34,739,392 shares authorized; 2,550,000, 4,277,860 and 25,344,540 shares issued and outstanding as of December 31, 2002 and 2003 and December 31, 2003 pro forma (unaudited) | — | 1 | 3 | |||||||||||
| Additional paid-in capital | — | 7,415 | 62,602 | |||||||||||
| Deferred compensation | (161 | ) | (4,681 | ) | (4,681 | ) | ||||||||
| Accumulated other comprehensive income (loss) | — | 76 | 76 | |||||||||||
| Deficit accumulated in the development stage | (4,485 | ) | (29,518 | ) | (29,518 | ) | ||||||||
| Total stockholders’ equity (deficit) | (4,646 | ) | (26,707 | ) | 28,482 | |||||||||
| Total liabilities, redeemable convertible preferred stock and stockholders’ equity (deficit) | $ | 16,111 | $ | 35,183 | $ | 35,183 | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
Table of Contents
ALNYLAM PHARMACEUTICALS, INC.
Consolidated Statements of Operations
| Period from | Cumulative | ||||||||||||||
| Inception | from Inception | ||||||||||||||
| (June 14, | (June 14, | ||||||||||||||
| 2002) | 2002) | ||||||||||||||
| through | Year Ended | through | |||||||||||||
| December 31, | December 31, | December 31, | |||||||||||||
| 2002 | 2003 | 2003 | |||||||||||||
Revenue from research collaborators | $ | — | $ | 176 | $ | 176 | |||||||||
Costs and expenses | |||||||||||||||
| Research and development(1) | 3,342 | 13,097 | 16,439 | ||||||||||||
| General and administrative(1) | 880 | 7,527 | 8,407 | ||||||||||||
| Purchased in-process research and development | — | 4,609 | 4,609 | ||||||||||||
| Total operating costs and expenses | 4,222 | 25,233 | 29,455 | ||||||||||||
| Loss from operations | (4,222 | ) | (25,057 | ) | (29,279 | ) | |||||||||
| Interest income | 86 | 179 | 265 | ||||||||||||
| Interest expense | — | (127 | ) | (127 | ) | ||||||||||
| Other expense | — | (28 | ) | (28 | ) | ||||||||||
| Net loss | (4,136 | ) | (25,033 | ) | (29,169 | ) | |||||||||
| Accretion of redeemable convertible preferred stock | (748 | ) | (2,906 | ) | (3,654 | ) | |||||||||
| Net loss attributable to common stockholders | $ | (4,884 | ) | $ | (27,939 | ) | $ | (32,823 | ) | ||||||
Comprehensive income | |||||||||||||||
| Net loss | $ | (4,136 | ) | $ | (25,033 | ) | $ | (29,169 | ) | ||||||
| Foreign currency translation adjustments | — | 76 | 76 | ||||||||||||
| Comprehensive loss | $ | (4,136 | ) | $ | (24,957 | ) | $ | (29,093 | ) | ||||||
Net loss per share | |||||||||||||||
| Net loss per common share (basic and diluted) | $ | (7.76 | ) | $ | (15.60 | ) | $ | (19.01 | ) | ||||||
| Weighted average shares used to compute basic and diluted net loss per common share | 629,548 | 1,791,063 | 1,726,521 | ||||||||||||
| Pro forma net loss per common share (basic and diluted) (unaudited) | $ | (1.65 | ) | ||||||||||||
| Shares used to compute pro forma basic and diluted net loss per common share (unaudited) | 15,157,601 | ||||||||||||||
| (1) Noncash stock-based compensation expense included in these amounts are as follows: | |||||||||||||||
| Research and development | $ | 172 | $ | 2,832 | $ | 3,004 | |||||||||
| General and administrative | — | 623 | 623 | ||||||||||||
| Total non-cash stock-based compensation | $ | 172 | $ | 3,455 | $ | 3,627 | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
Table of Contents
ALNYLAM PHARMACEUTICALS, INC.
Consolidated Statements of Changes In Redeemable Convertible Preferred Stock and Stockholders’ Deficit
| Redeemable Convertible | ||||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Accumulated | Deficit | |||||||||||||||||||||||||||||||||
| Additional | Other | Accumulated in | ||||||||||||||||||||||||||||||||||
| Number of | Number of | Par Value | Paid-in | Deferred | Comprehensive | the Development | ||||||||||||||||||||||||||||||
| Shares | Amount | Shares | $0.0001 | Capital | Compensation | Income | Stage | Total | ||||||||||||||||||||||||||||
Balance at inception (June 14, 2002) | — | $ | — | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||||||||
| Issuance of common stock to founders | — | — | 2,460,000 | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of common stock to nonemployee | — | — | 90,000 | — | 9 | — | — | — | 9 | |||||||||||||||||||||||||||
| Issuance of Series A redeemable convertible preferred stock, net of issuance costs of $21 | 2,000,010 | 1,979 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of Series B redeemable convertible preferred stock, net of issuance costs of $43 | 6,160,000 | 15,357 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of warrant to purchase Series B preferred stock in connection with equipment line of credit | — | — | — | — | 57 | — | — | — | 57 | |||||||||||||||||||||||||||
| Deferred compensation related to restricted common stock issued to nonemployees | — | — | — | — | 333 | (333 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation expense related to restricted stock issued to nonemployees | — | — | — | — | — | 172 | — | — | 172 | |||||||||||||||||||||||||||
| Accretion of Series A and Series B preferred stock to redemption | — | 748 | — | — | (399 | ) | — | — | (349 | ) | (748 | ) | ||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | (4,136 | ) | (4,136 | ) | |||||||||||||||||||||||||
Balance at December 31, 2002 | 8,160,010 | 18,084 | 2,550,000 | — | — | (161 | ) | — | (4,485 | ) | (4,646 | ) | ||||||||||||||||||||||||
| Exercise of common stock options | — | — | 84,600 | — | 21 | — | — | — | 21 | |||||||||||||||||||||||||||
| Issuance of common stock in connection with acquisition of Ribopharma | — | — | 1,549,216 | — | 1,947 | — | — | — | 1,947 | |||||||||||||||||||||||||||
| Settlement of Ribopharma AG accrued interest for common stock | — | — | 94,044 | — | 119 | — | — | — | 119 | |||||||||||||||||||||||||||
| Issuance of Series A redeemable convertible preferred stock | 1,000,000 | 1,000 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Beneficial conversion feature from the issuance of Series A preferred stock | — | (260 | ) | — | — | 260 | — | — | — | 260 | ||||||||||||||||||||||||||
| Accretion of beneficial conversion feature on Series A preferred stock | — | 27 | — | — | (27 | ) | — | — | — | (27 | ) | |||||||||||||||||||||||||
| Issuance of Series B redeemable convertible preferred stock | 10,401,845 | 26,005 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock, net of issuance costs of $70 | 1,504,825 | 7,454 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Accretion of Series A and Series B preferred stock to redemption | — | 2,879 | — | — | (2,879 | ) | — | — | — | (2,879 | ) | |||||||||||||||||||||||||
| Deferred compensation related to issuance of options granted to employees | — | — | — | — | 3,338 | (3,338 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation expense related to options issued to employees | — | — | — | — | — | 667 | — | — | 667 | |||||||||||||||||||||||||||
| Deferred compensation related to restricted stock issued to nonemployees | — | — | — | — | 3,458 | (3,458 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation expense related to restricted stock issued to nonemployees | — | — | — | — | — | 2,241 | — | — | 2,241 | |||||||||||||||||||||||||||
| Deferred compensation related to stock options issued to nonemployees | — | — | — | — | 1,178 | (1,178 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation expense related to stock options issued to nonemployees | — | — | — | — | — | 547 | — | — | 547 | |||||||||||||||||||||||||||
| Foreign currency translation | — | — | — | — | — | — | 76 | — | 76 | |||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | (25,033 | ) | (25,033 | ) | |||||||||||||||||||||||||
Balance at December 31, 2003 | 21,066,680 | 55,189 | 4,277,860 | 1 | 7,415 | (4,681 | ) | 76 | (29,518 | ) | (26,707 | ) | ||||||||||||||||||||||||
| Conversion of redeemable convertible preferred stock into common stock (unaudited) | (21,066,680 | ) | (55,189 | ) | 21,066,680 | 2 | 55,187 | — | — | — | 55,189 | |||||||||||||||||||||||||
Pro forma balance, December 31, 2003 (unaudited) | — | $ | — | 25,344,540 | $ | 3 | $ | 62,602 | $ | (4,681 | ) | $ | 76 | $ | (29,518 | ) | $ | 28,482 | ||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
Table of Contents
ALNYLAM PHARMACEUTICALS, INC.
Consolidated Statements of Cash Flows
| Period from | Cumulative | ||||||||||||||
| Inception | from Inception | ||||||||||||||
| (June 14, | (June 14, | ||||||||||||||
| 2002) | 2002) | ||||||||||||||
| through | Year Ended | through | |||||||||||||
| December 31, | December 31, | December 31, | |||||||||||||
| 2002 | 2003 | 2003 | |||||||||||||
Cash flows from operating activities | |||||||||||||||
| Net loss | $ | (4,136 | ) | $ | (25,033 | ) | $ | (29,169 | ) | ||||||
| Adjustments to reconcile net loss to net cash flows used in operating activities: | |||||||||||||||
| Depreciation and amortization | 36 | 838 | 874 | ||||||||||||
| Noncash stock-based compensation | 172 | 3,455 | 3,627 | ||||||||||||
| Series B preferred stock issued for Garching license | — | 397 | 397 | ||||||||||||
| Charge for purchased in-process research and development | — | 4,609 | 4,609 | ||||||||||||
| Changes in operating assets and liabilities; net of acquisition | |||||||||||||||
| Prepaid expenses and other current assets | (23 | ) | (558 | ) | (581 | ) | |||||||||
| Accounts payable | 674 | 794 | 1,468 | ||||||||||||
| Accrued expenses | 1,999 | 781 | 2,780 | ||||||||||||
| Deferred revenue | — | 1,889 | 1,889 | ||||||||||||
| Net cash used in operating activities | (1,278 | ) | (12,828 | ) | (14,106 | ) | |||||||||
Cash flows from investing activities | |||||||||||||||
| Purchases of property and equipment | (562 | ) | (3,119 | ) | (3,681 | ) | |||||||||
| Acquisition of Ribopharma AG, net of acquired cash | — | (121 | ) | (121 | ) | ||||||||||
| Increase in restricted cash | (19 | ) | (2,667 | ) | (2,686 | ) | |||||||||
| Net cash used in investing activities | (581 | ) | (5,907 | ) | (6,488 | ) | |||||||||
Cash flows from financing activities | |||||||||||||||
| Proceeds from the issuance of common stock | 9 | 21 | 30 | ||||||||||||
| Proceeds from issuance of Series A redeemable convertible preferred stock, net of issuance costs | 1,979 | 1,000 | 2,979 | ||||||||||||
| Proceeds from issuance of Series B redeemable convertible preferred stock, net of issuance costs | 15,357 | 19,005 | 34,362 | ||||||||||||
| Proceeds from issuance of Series C convertible preferred stock, net of issuance costs | — | 7,454 | 7,454 | ||||||||||||
| Proceeds of bank debt | — | 2,098 | 2,098 | ||||||||||||
| Repayment of bank debt | — | (239 | ) | (239 | ) | ||||||||||
| Repayment of debt assumed in acquisition | — | (2,964 | ) | (2,964 | ) | ||||||||||
| Deferred financing costs incurred in connection with the equipment line of credit | (9 | ) | — | (9 | ) | ||||||||||
| Net cash provided by financing activities | 17,336 | 26,375 | 43,711 | ||||||||||||
| Effect of exchange rate changes on cash | — | 76 | 76 | ||||||||||||
| Net increase in cash and cash equivalents | 15,477 | 7,716 | 23,193 | ||||||||||||
| Cash and cash equivalents, beginning of period | — | 15,477 | — | ||||||||||||
| Cash and cash equivalents, end of period | $ | 15,477 | $ | 23,193 | $ | 23,193 | |||||||||
F-6
Table of Contents
Consolidated Statements of Cash Flows — (Continued)
| Period from | Cumulative | |||||||||||
| Inception | from Inception | |||||||||||
| (June 14, | (June 14, | |||||||||||
| 2002) | 2002) | |||||||||||
| through | Year Ended | through | ||||||||||
| December 31, | December 31, | December 31, | ||||||||||
| 2002 | 2003 | 2003 | ||||||||||
Supplemental disclosure of cash flows | ||||||||||||
| Cash paid for interest | $ | — | $ | 503 | $ | 503 | ||||||
Supplemental disclosure of noncash financing activities | ||||||||||||
| Fair value of warrant issued in connection with equipment line of credit included as deferred financing costs | $ | 57 | $ | — | $ | 57 | ||||||
| Accretion of redeemable convertible preferred stock | 748 | 2,879 | 3,627 | |||||||||
| Series B preferred stock issued to Garching in 2003 for a license in 2002 included in accrued expenses | — | 2,205 | 2,205 | |||||||||
| Conversion of note payable and accrued interest into Series B preferred stock | — | 4,795 | 4,795 | |||||||||
| Beneficial conversion feature on issuance of Series A preferred stock | — | 260 | — | |||||||||
Acquisition of Ribopharma AG | ||||||||||||
| Fair valued of assets acquired | $ | — | $ | 12,256 | $ | 12,256 | ||||||
| Assumed liabilities | — | (8,390 | ) | (8,390 | ) | |||||||
| Cash paid | — | (1,500 | ) | (1,500 | ) | |||||||
| Acquisition costs incurred | — | (419 | ) | (419 | ) | |||||||
| Fair value of common stock issued | $ | — | $ | 1,947 | $ | 1,947 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-7
Table of Contents
ALNYLAM PHARMACEUTICALS, INC.
Notes to Consolidated Financial Statements
1. Nature of the Business
Alnylam (the “Company”) commenced operations on June 14, 2002 as a biopharmaceutical company seeking to develop and commercialize new drugs that work through a recently discovered system in cells known as RNA interference, or RNAi. Alnylam is focused on discovering, developing and commercializing RNAi therapeutics by establishing strategic alliances with leading pharmaceutical companies, establishing and maintaining a strong intellectual property position in the RNAi field and generating revenues through licensing agreements. Since its inception, the Company has devoted substantially all of its efforts to business planning, research and development, acquiring intellectual property rights, recruiting management and technical staff, and raising capital. Accordingly, the Company is considered a development stage enterprise as defined in the Statement of Financial Accounting Standards (“SFAS”) No. 7 and the accompanying financial statements represent those of a development stage enterprise.
On July 31, 2003, Alnylam acquired the outstanding shares of Ribopharma AG (a development stage enterprise) and accounted for the acquisition as an asset purchase. Under the terms of the agreement, shareholders of Ribopharma AG exchanged all their outstanding shares for $1,500 in cash and 1,549,216 shares of Alnylam common stock (Note 4).
The accompanying consolidated financial statements have been prepared on a basis which assumes that the Company will continue as a going concern and which contemplates the realization of assets and satisfaction of liabilities and commitments in the normal course of business. The Company has a limited operating history and has incurred losses from operations since its inception. The Company has an accumulated deficit of $29,518 at December 31, 2003, and the Company believes that its existing capital resources will enable it to maintain its current and planned operations through at least December 31, 2004. The future viability of the Company beyond 2004 is largely dependent on its ability to raise additional capital to finance its operations. Although management continues to pursue additional financing plans, there is no assurance that the Company will be successful in obtaining financing on terms acceptable to the Company, if at all.
The Company is subject to risks common to companies in the drug development and pharmaceutical industry, including but not limited to, the successful development and commercialization of drug candidates, fluctuations in operating results and financial risks, potential need for additional financing, ability to obtain licenses to technology to be used in the Company’s research, protection of proprietary technology and patent risks, compliance with government regulations, dependence on key personnel and collaborative partners, competition, customer demand, management of growth, and the effectiveness of marketing by the Company.
2. Summary of Significant Accounting Policies
Significant accounting policies applied by the Company in the preparation of its consolidated financial statements are as follows:
Basis of Presentation
The Company comprises three entities, Alnylam Pharmaceuticals, Inc. (the parent company) and two subsidiaries (Alnylam US and Ribopharma AG). Alnylam Pharmaceuticals, Inc is a Delaware corporation that was formed on May 8, 2003 and was formerly called Alnylam Holding Co. Alnylam US is also a Delaware corporation that was formed on June 14, 2002 and was previously called Alnylam Pharmaceuticals, Inc. On July 31, 2003, Alnylam Pharmaceuticals, Inc. (the parent company) and Alnylam US were reorganized and Alnylam US became a wholly owned subsidiary of Alnylam Pharmaceuticals, Inc. (the parent company).
F-8
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Since Alnylam US and Alnylam Pharmaceuticals, Inc. were under common control and Alnylam Pharmaceuticals, Inc. (the parent company) did not have independent operations prior to the reorganization, the combination of the two entities did not result in a new basis of accounting.
Principles of Consolidation
The accompanying consolidated financial statements reflect the operations of the Company and its wholly-owned subsidiaries Alnylam US and Ribopharma AG. All significant intercompany accounts and transactions have been eliminated.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Concentration of Credit Risk
Financial instruments which potentially expose the Company to concentration of credit risk consist primarily of cash and cash equivalents. As of December 31, 2002 and 2003, substantially all of the Company’s cash and cash equivalents was invested in money market mutual funds, primarily through a single highly rated financial institution.
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of three months or less at the date of purchase to be cash equivalents. Cash equivalents, which consist primarily of money market instruments and an overnight sweep account, amounted to $15,477 and $23,193 at December 31, 2002 and 2003, respectively. These investments are stated at cost plus accrued interest, which approximates fair market value.
Unaudited pro forma presentation
The unaudited pro forma balance sheet and the unaudited pro forma statement of redeemable convertible preferred stock and stockholders’ equity (deficit) as of December 31, 2003, reflect the automatic conversion of all outstanding shares of Series A, Series B and Series C convertible preferred stock into 21,066,680 shares of common stock upon the closing of the Company’s proposed initial public offering (IPO).
Restricted Cash
In June 2002, in conjunction with the lease for its 790 Memorial Drive, Cambridge facility, the Company arranged for the delivery to the landlord of a $19 letter of credit secured by restricted cash. At the end of the lease in April 2004, the cash will no longer be restricted. The remaining current restricted cash balance relates to a letter of credit the Company was obligated to enter into resulting from indemnity agreements that the Company entered into with the two Ribopharma shareholders for $354 (See Note 12).
In September 2003, the Company entered into a lease for a new facility at 300 Third Street, Cambridge, Massachusetts that it anticipates occupying in 2004. In connection with this lease, the Company arranged for the delivery to the landlord of a letter of credit in an amount equal to one year’s rent at the new facility, or
F-9
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
$2,313. This letter of credit was secured by restricted cash. Consistent with the terms of the lease, the Company has accounted for restricted cash as long-term in the accompanying consolidated balance sheets.
Fair Value of Financial Instruments
The carrying amounts of the Company’s financial instruments, which include cash equivalents, accounts payable, accrued expenses and notes payable, approximate their fair values at December 31, 2002 and 2003.
Property and Equipment and Long-Lived Assets
Property and equipment is recorded at cost and depreciated using the straight-line method over the estimated useful life of each asset. Expenditures for repairs and maintenance are charged to expense as incurred. On disposal, the related assets and accumulated depreciation are eliminated from the accounts and any resulting gain or loss is included in income. Leasehold improvements are amortized over the shorter of the useful life of the improvement or the remaining term of the lease.
The Company evaluates the recoverability of its property and equipment and other long-lived assets when circumstances indicate that an event of impairment may have occurred in accordance with the provisions of SFAS No. 144,Accounting for the Impairment of Disposal of Long-Lived Assets (“SFAS No. 144”). SFAS No. 144 further refines the requirements of SFAS No. 121,Accounting for the Impairment of Long-Lived Assets and Long-Lived Assets to be Disposed of, that companies (1) recognize an impairment loss only if the carrying amount of a long-lived asset is not recoverable based on its undiscounted future cash flows and (2) measure an impairment loss as the difference between the carrying amount and fair value of the asset. In addition, SFAS No. 144 provides guidance on accounting and disclosure issues surrounding long-lived assets to be disposed of by sale. No impairment was required to be recognized for any of the periods ended December 31, 2002 and 2003.
Revenue Recognition
The Company recognizes revenue in accordance with the Securities and Exchange Commission’s Staff Accounting Bulletin No. 104,Revenue Recognition in Financial Statements(“SAB 104”). Revenue is recognized when the following criteria have been met:
| 1. | Persuasive evidence of an arrangement exists; | |
| 2. | Delivery has occurred and risk of loss has passed; | |
| 3. | The seller’s price to the buyer is fixed or determinable; and | |
| 4. | Collectibility is reasonably assured. |
The Company has entered into a collaboration agreement with Merck. Revenue from this collaboration agreement includes nonrefundable license fees, milestones and royalties. When evaluating multiple element arrangements, the Company considers whether the components of the arrangement represent separate units of accounting as defined in Emerging Issues Task Force (EITF) Issue No. 00-21,Revenue Arrangements with Multiple Deliverables(“EITF 00-21”). EITF 00-21 requires the following criteria to be met for an element to represent a separate unit of accounting:
| 1. | The delivered items have value to a customer on a standalone basis; | |
| 2. | There is objective and reliable evidence of the fair value of the undelivered items; and |
F-10
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
| 3. | Delivery or performance is probable and within the control of the vendor for any delivered items that have a right of return. |
Nonrefundable license fees, which are not separable from the Company’s continuing performance obligations, are recognized as revenue as the Company performs under the collaboration agreement. Where the Company’s level of effort is relatively constant over the performance period, it recognizes total fixed or determined contract revenues on a straight-line basis over the development period set forth in the contract.
The Company recognizes milestone payments as revenue upon achievement of the milestone only if (1) it represents a separate unit of accounting as defined in EITF 00-21; (2) the milestone payments are nonrefundable; (3) substantive effort is involved in achieving the milestone; and (4) the amount of the milestone is reasonable in relation to the effort expended or the risk associated with achievement of the milestone. If any of these conditions are not met, the Company defers the milestone payments and recognizes them as revenue over the term of the contract as it completes its performance obligations.
Income Taxes
The Company uses the liability method of accounting for income taxes. Under the liability method, deferred tax assets and liabilities reflect the impact of temporary differences between amounts of assets and liabilities for financial reporting purposes and such amounts as measured under enacted tax laws. A valuation allowance is required to offset any net deferred tax assets if, based upon the available evidence, it is more likely than not that some or all of the deferred tax asset will not be realized.
Research and Development Costs
Research and development costs are expensed as incurred. Included in research and development costs are wages, benefits and other operating costs such as legal expenses to secure and defend patents (which are expensed as incurred), facilities, supplies and overhead directly related to the Company’s research and development department as well as costs to acquire technology licenses.
During the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company included approximately $481, $1,357 and $1,838, respectively, of legal patent costs in research and development costs and expenses.
The Company has entered into several license agreements for rights to utilize certain technologies. The terms of the licenses may provide for up-front payments, annual maintenance payments, milestone payments based upon certain specified events being achieved and royalties on product sales. Costs to acquire and maintain licensed technology that has not reached technological feasibility and does not have alternative future use are charged to research and development expense as incurred. During the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company charged to research and development expense $1,918, $1,720 and $3,638, respectively, of costs associated with license fees (Note 14).
Accounting for Stock-Based Compensation
Employee stock awards granted under the Company’s compensation plans are accounted for in accordance with Accounting Principles Board Opinion No. 25,Accounting for Stock Issued to Employees(“APB 25”), and related interpretations. The Company has not adopted the fair value method of accounting for stock-based compensation. Accordingly, compensation expense is recorded for options issued to employees to the extent that the fair market value of the Company’s common stock exceeds the exercise price
F-11
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
of the option at the date granted and all other criteria for fixed accounting have been met. All stock-based awards granted to nonemployees are accounted for at their fair value in accordance with SFAS No. 123, as amended, and Emerging Issues Task Force (“EITF”) Issue No. 96-18,Accounting for Equity Instruments that are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling, Goods or Services, under which compensation expense is generally recognized over the vesting period of the award.
The Company provides the disclosure requirements of SFAS No. 148,Accounting for Stock Based Compensation — Transition and Disclosure, an amendment of FASB Statement No. 123 (“SFAS 148”).If compensation expense for the Company’s stock-based compensation plan had been determined based on the fair value at the grant dates as calculated in accordance with SFAS No. 123, the Company’s net loss attributable to common stockholders and net loss per common share would approximate the pro forma amounts below:
| Period from | |||||||||||||
| Inception | Cumulative from | ||||||||||||
| (June 14, | Inception | ||||||||||||
| 2002) | (June 14, 2002) | ||||||||||||
| through | Year Ended | through | |||||||||||
| December 31, | December 31, | December 31, | |||||||||||
| 2002 | 2003 | 2003 | |||||||||||
| Net loss attributable to common stockholders Net loss, as reported | $ | (4,884 | ) | $ | (27,939 | ) | $ | (32,823 | ) | ||||
| Add employee stock-based compensation expense included in reported net loss | — | 667 | 667 | ||||||||||
| Deduct stock-based compensation expense determined under fair value method | — | (697 | ) | (697 | ) | ||||||||
| Net loss — pro forma | $ | (4,884 | ) | $ | (27,969 | ) | $ | (32,853 | ) | ||||
| Net loss per common share (basic and diluted) | |||||||||||||
| As reported | $ | (7.76 | ) | $ | (15.60 | ) | $ | (19.01 | ) | ||||
| Pro forma | $ | (7.76 | ) | $ | (15.62 | ) | $ | (19.03 | ) | ||||
F-12
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
The Company has estimated the fair value of its granted stock options by applying a present value approach which does not consider expected volatility of the underlying stock (“minimum value method”) using the following weighted average assumptions for the period from inception (June 14, 2002) through December 31, 2002 and for the year ended December 31, 2003:
| Period from | ||||||||
| Inception | ||||||||
| (June 14, | ||||||||
| 2002) through | Year Ended | |||||||
| December 31, | December 31, | |||||||
| 2002 | 2003 | |||||||
| Risk-free interest rate | 3.02 | % | 3.19 | % | ||||
| Expected dividend yield | — | — | ||||||
| Expected option term | 5 years | 5 years | ||||||
| Volatility | 0 | % | 0 | % | ||||
Since options vest over several years and additional option grants are expected to be made in future years, the pro forma effects of applying the fair value method may be material to reported net income or loss in future years.
Redeemable Convertible Preferred Stock
Redeemable convertible preferred stock is treated as if it is mandatorily redeemable (classified in the mezzanine section of the balance sheet) if it may be redeemed by the holder based on facts and circumstances not in the Company’s control. If there is a specified redemption date, the carrying value is accreted to its redemption value over the term. These adjustments are affected through charges first against retained earnings, then against additional paid-in capital until it is reduced to zero and then to accumulated deficit.
Foreign Currency
Our foreign subsidiary, Ribopharma AG (a German based company), has designated their local currency, the Euro, as their functional currency. Financial statements of this foreign subsidiary are translated to U.S. dollars for consolidation purposes using current rates of exchange for assets and liabilities; equity is translated using historical exchange rates; and revenue and expense amounts are translated using the average exchange rate for the period. Net unrealized gains and losses resulting from foreign currency translation are included in other comprehensive loss which is a separate component of stockholders’ deficit. Net realized gains and losses from foreign currency transactions are included in the consolidated statement of operations.
F-13
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Comprehensive Loss
Comprehensive income (loss) is comprised of net income (loss) and certain changes in stockholders’ equity that are excluded from net income (loss). The Company includes foreign currency translation adjustments in other comprehensive income (loss) for Ribopharma AG as the functional currency is not the U.S. dollar.
Net Income (Loss) Per Common Share
The Company accounts for and discloses net income (loss) per common share in accordance with SFAS No. 128,Earnings Per Share(“SFAS No. 128”). Basic net income (loss) per common share is computed by dividing net income (loss) attributable to common stockholders by the weighted average number of common shares outstanding. Diluted net income (loss) per common share is computed by dividing net income (loss) attributable to common stockholders by the weighted average number of common shares and dilutive potential common share equivalents then outstanding. Potential common shares consist of shares issuable upon the exercise of stock options and warrants (using the treasury stock method), unvested restricted stock awards and the weighted average conversion of the preferred stock into shares of common stock (using the if-converted method).
Segment Information
Management uses consolidated financial information in determining how to allocate resources and assess financial performance. For this reason, the Company has determined that they are principally engaged in one industry segment.
The following table presents total long-lived tangible assets by geographic area as of December 31, 2002 and 2003:
| December 31, | |||||||||
| 2002 | 2003 | ||||||||
| Long-lived tangible assets | |||||||||
| United States | $ | 526 | $ | 2,342 | |||||
| Germany | — | 2,414 | |||||||
| Total long-lived tangible assets | $ | 526 | $ | 4,756 | |||||
Recent Accounting Pronouncements
In January 2003, the FASB issued FIN No. 46,Consolidation of Variable Interest Entities, an Interpretation of ARB No. 51(“FIN No. 46”). The primary objectives of FIN No. 46 are to provide guidance on the identification of entities for which control is achieved through means other than through voting rights (variable interest entities) and to determine when and which business enterprise should consolidate the variable interest entities. The new model for consolidation applies to an entity which either (1) the equity investors (if any) do not have a controlling financial interest or (2) the equity investment at risk is insufficient to finance the entity’s activities without receiving additional subordinated financial support from the other parties. FIN No. 46 also requires enhanced disclosures for variable interest entities. FIN No. 46 applies immediately to variable interest entities created after January 31, 2003, and to variable interest entities in which an enterprise obtains an interest after that date. The standard as amended by FIN 46R, applies to the first fiscal year or interim period beginning after March 15, 2004 to variable interest entities in which an
F-14
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
enterprise holds a variable interest that it acquired before February 1, 2003. The Company does not expect the adoption of FIN No. 46 to have a material impact on its consolidated financial statements.
In May 2003, the FASB issued SFAS No. 150,Accounting for Certain Instruments with Characteristics of both Liabilities and Equity(“SFAS No. 150”). This statement establishes standards for how an issuer classifies and measures certain financial instruments with characteristics of both liabilities and equity. It requires that an issuer classify a financial instrument that is within its scope as a liability (or an asset in some circumstances). Many of these instruments were previously classified as equity. This statement is effective for financial instruments entered into or modified after May 31, 2003, and otherwise is effective at the beginning of the first interim period beginning after June 15, 2003. The Company’s adoption of this statement did not have a material impact on its consolidated financial statements.
3. Net Loss Per Common Share
The following sets forth the computation of basic and diluted net loss per common share:
| Period from | Cumulative from | ||||||||||||
| Inception | Inception | ||||||||||||
| (June 14, 2002) | (June 14, 2002) | ||||||||||||
| through | Year Ended | through | |||||||||||
| December 31, | December 31, | December 31, | |||||||||||
| 2002 | 2003 | 2003 | |||||||||||
| Basic and diluted net loss per common share: | |||||||||||||
| Net loss attributable to common stockholders | $ | (4,884 | ) | $ | (27,939 | ) | $ | (32,823 | ) | ||||
| Basic and diluted net loss per common share | $ | (7.76 | ) | $ | (15.60 | ) | $ | (19.01 | ) | ||||
| Basic and diluted weighted average number of common shares outstanding | 629,548 | 1,791,063 | 1,726,521 | ||||||||||
| Pro forma basic and diluted net loss per common share (unaudited) | $ | (1.65 | ) | ||||||||||
| Basic and diluted pro forma number of common shares outstanding (unaudited) | 15,157,601 | ||||||||||||
As of December 31, 2002, 1,738,120 shares of restricted common stock remained subject to repurchase by the Company and, accordingly, were not included in the weighted average shares outstanding as of December 31, 2002. As of December 31, 2003, 1,226,235 shares of restricted common stock remained subject to repurchase by the Company and, accordingly, were not included in the weighted average shares outstanding as of December 31, 2003.
F-15
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
The following potentially dilutive, common share equivalents were excluded from the calculation of diluted net loss per common share because their effect was antidilutive for each of the periods presented:
| Period from | Cumulative from | |||||||||||
| Inception | Inception | |||||||||||
| (June 14, 2002) | (June 14, 2002) | |||||||||||
| through | Year Ended | through | ||||||||||
| December 31, | December 31, | December 31, | ||||||||||
| 2002 | 2003 | 2003 | ||||||||||
| Options | 155,600 | 3,217,756 | 3,217,756 | |||||||||
| Warrants | 25,000 | 25,000 | 25,000 | |||||||||
| Convertible preferred stock | 8,160,000 | 21,066,680 | 21,066,680 | |||||||||
| Unvested restricted stock | 1,738,120 | 1,226,235 | 1,226,235 | |||||||||
The Company’s historical capital structure is not indicative of its capital structure after the proposed IPO due to the anticipated automatic conversion of all shares of convertible preferred stock into shares of common stock concurrent with the closing of the Company’s proposed IPO. Accordingly, pro forma net loss per common share is presented for the year ended December 31, 2003.
Pro forma basic and diluted net loss per common share is computed by dividing the net loss for the period by the pro forma number of unrestricted common shares outstanding, assuming the conversion of the Company’s Series A, Series B, and Series C convertible preferred stock into shares of the Company’s common stock which will occur upon the closing of the Company’s proposed IPO, as if such conversion occurred at the date of the original issuance of the shares of convertible preferred stock. Pro forma diluted net loss per common share excludes options to purchase 3,217,756 shares of common stock, a warrant to purchase 25,000 shares of Series B redeemable convertible preferred stock which is assumed to be converted into a warrant to purchase common stock, and 1,226,235 shares of unvested restricted stock.
4. Acquisition of Ribopharma AG
On July 31, 2003, Alnylam acquired all the outstanding voting shares of Ribopharma AG (a German based company). Ribopharma’s results of operations are included in the operating results of the Company from the date of acquisition (July 31, 2003). Ribopharma is a development stage enterprise that is performing research and development associated with a new pharmaceutical active agent category siRNA. Alnylam purchased Ribopharma for access to its in-process research and development programs and its core technology. In addition, the acquisition of Ribopharma enabled Alnylam to satisfy the conditions in the technology license agreement with Garching Innovation GmbH (Note 14) to establish a German based company with comparable operational force and resources. Satisfaction of this condition enabled Alnylam to convert its co-exclusive rights under the Garching license to exclusive rights.
F-16
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
The consideration consisted of $1,500 in cash and 1,549,216 shares of common stock. Based on a valuation performed of Ribopharma AG and the intangible assets acquired, the purchase price is estimated at $3,866 and is comprised of the following:
| Cash paid | $ | 1,500 | ||
| Fair value of common stock issued | 1,947 | |||
| Acquisition costs | 419 | |||
| $ | 3,866 | |||
The fair value of the tangible and intangible assets acquired and liabilities assumed were recorded as follows:
| Cash | $ | 1,798 | ||
| Other current assets | 41 | |||
| Fixed assets | 1,733 | |||
| Intangible assets | 8,684 | |||
| Accounts payable and accrued expenses assumed | (1,300 | ) | ||
| Notes payable assumed | (7,090 | ) | ||
| $ | 3,866 | |||
The appraised value of intangible assets acquired was below the total fair value of intangible assets acquired and would generally result in the recognition of goodwill. However, since Ribopharma AG is a development stage company and not considered a “business” as defined by the applicable accounting rules, this residual value was allocated proportionately to the long-lived assets acquired as follows:
| Initial | Recorded | |||||||
| Value | Fair Value | |||||||
| Purchased in-process research and development | $ | 2,155 | $ | 4,609 | ||||
| Core technology | 1,700 | 3,638 | ||||||
| Workforce | 200 | 437 | ||||||
| Fixed assets | 552 | 1,733 | ||||||
| $ | 4,607 | $ | 10,417 | |||||
The fair market value of the intangible assets acquired was based on a valuation and determined using an income approach. The core technology was valued using a relief from royalty methodology, the purchased in-process research and development was valued based on a discounted cash flow analysis and the workforce was valued using the avoided cost method.
Purchased in-process technology was written off immediately upon the consummation of the acquisition and is included as a separate line in the Company’s statement of operations. Core technology and workforce are being amortized over their estimated useful lives of ten years and four years, respectively. The step up in the fixed assets is being amortized over four years, the remaining estimated useful life of these assets.
During the year ended December 31, 2003 and for the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company has recorded $197 of amortization expense related to the core technology and workforce intangibles of which the entire amount is included in research and development.
F-17
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
During the year ended December 31, 2003 and for the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company has recorded $123 of additional depreciation related to the increase in the recorded fair value of the fixed assets of which $111 is included in research and development and $12 is recorded in general and administrative expense.
Purchased In-Process Research and Development
In connection with the Company’s acquisition of Ribopharma AG, the Company acquired two systemic RNAi programs related to the development of drugs targeting cancers such as malignant melanoma and pancreatic carcinoma. The Company expensed $4,609 of purchased in-process research and development associated with these programs. Management’s plans contemplate that the Company will conduct the first phase of clinical trials and then out-license the programs to a partner. Upon out-licensing, the partner is expected to bear all development costs and control clinical development. The Company expects to earn payments upon the attainment of clinical milestones by our partner and royalties on product sales. Since our partner will control the clinical development, we will be unable to influence the timing of the achievement of the milestones, if at all, or the estimated year of the product launch, if at all. The Company’s valuation assumed a development period of approximately 10 years, with milestones being earned during that period, which management believes is a typical horizon to bring a therapeutic drug to market. Actual results will differ from these estimates due to the uncertainties surrounding drug development.
Management assumes responsibility for determining the in-process research and development valuation. The fair value assigned to purchase in-process research and development was estimated by discounting, to present value, the probability-adjusted net cash flows expected to result once the technology has reached technological feasibility. A discount rate of 32 percent was applied to estimate the present value of the cash flows and is consistent with the overall risks of developing these projects. As of December 31, 2003, the technological feasibility of the projects had not been reached and management believes the assumptions included in the valuation analysis continue to be valid. In the allocation of the purchase price, the concept of alternative future use was considered. The projects under development have no current alternative future uses for the underlying technology in the event the projects are unsuccessful.
5. Property and Equipment
| Estimated | December 31, | |||||||||||
| Useful Life | ||||||||||||
| (Years) | 2002 | 2003 | ||||||||||
| Laboratory equipment and software | 5 | $ | 398 | $ | 4,300 | |||||||
| Computer equipment | 3 | 59 | 270 | |||||||||
| Furniture and fixtures | 5 | 55 | 431 | |||||||||
| Leasehold improvements | * | 50 | 50 | |||||||||
| Construction in process | — | — | 363 | |||||||||
| 562 | 5,414 | |||||||||||
| Less — accumulated depreciation | (36 | ) | (658 | ) | ||||||||
| $ | 526 | $ | 4,756 | |||||||||
| * | shorter of asset life or lease term |
F-18
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Depreciation expense was $36, $622, and $658 for the period from June 14, 2002 (date of inception) to December 31, 2002, year ended December 31, 2003 and for the period from the June 14, 2002 (date of inception) to December 31, 2003, respectively.
6. Accrued Liabilities
Accrued expenses consist of the following at December 31, 2002 and 2003:
| 2002 | 2003 | |||||||
| License | $ | 1,808 | $ | — | ||||
| Payroll and benefits | 18 | 823 | ||||||
| Professional fees | 112 | 344 | ||||||
| Other | 61 | 255 | ||||||
| Interest | — | 21 | ||||||
| $ | 1,999 | $ | 1,443 | |||||
Accrued license fees represents the value of stock owed to Garching Innovation GmbH as of December 31, 2002 for the rights to use certain patented technology (Note 14).
7. Notes Payable
Note Payable to a Bank
In December 2002, the Company entered into an agreement with Silicon Valley Bank to establish an equipment line of credit for $2,500. The termination date for the agreement is April 30, 2004 upon adherence to certain conditions. All borrowings under the line of credit are collateralized by all corporate assets, excluding intellectual property. Borrowings bear interest at prime rate plus 0.25 percent (4.25 percent at December 31, 2003) and each advance is repayable over a 42 month period from the date of each advance. On the maturity of each equipment advance under the line of credit, the Company shall pay, in addition to the paid principal and interest, an additional amount of eight percent of the original principal. The Company is accruing this eight percent over each of the respective drawdown repayment periods as additional interest expense, which amounted to $21 during the year ended December 31, 2003.
F-19
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
As of December 31, 2003, future cash payments under the note payable to a bank are as follows:
| Year Ended December 31, | ||||
| 2004 | $ | 645 | ||
| 2005 | 645 | |||
| 2006 | 589 | |||
| 2007 | 244 | |||
| 2,123 | ||||
| Less: portion representing interest | 264 | |||
| 1,859 | ||||
| Less: current portion of note payable | 558 | |||
| Note payable, net of current portion | $ | 1,301 | ||
The terms of the agreement include certain covenants which limit the Company’s ability to sell or transfer certain assets or businesses or to pay dividends or other distributions above specified limits. The Company made four drawdowns on the line of credit from January 1, 2003 to December 31, 2003, amounting to a total of $2,098, of which $239 has been paid as of December 31, 2003. As of December 31, 2003, $402 is available on the line of credit to finance additional equipment.
In connection with the agreement, the Company issued Silicon Valley Bank a warrant to purchase 25,000 shares of Series B redeemable convertible preferred stock at an exercise price of $2.50 per share. 12,500 of the shares were earned at closing and the remaining 12,500 were earned when the Company’s borrowings exceeded $1,250. The Company recorded the fair value of these warrants of $57 as a discount to the note payable which is being amortized to interest expense over the repayment term of the first advance of 42 months. The fair value of each warrant was calculated using the Black-Scholes option pricing model with the following assumptions: 100% volatility, risk-free interest rate of 3.8%, no dividend yield, and a ten-year term. During the year ended December 31, 2003 and the cumulative period from inception (June 14, 2002) through December 31, 2003, the Company recorded $16 of interest expense related to this deferred financing costs.
Note Payable to Ribopharma AG Shareholder
Upon the acquisition of Ribopharma AG, Alnylam Pharmaceuticals, Inc. assumed a note payable and accrued interest of $4,795 to a Ribopharma shareholder and an obligation to provide common shares based on prior terms of the note valued at $119, both of which were included in assumed liabilities upon the acquisition date. The note payable of $4,538 and accrued interest of $257 was exchanged for 1,917,857 shares of Series B preferred stock. Additionally, the 94,044 shares of Alnylam Pharmaceuticals, Inc.’s common stock were issued to satisfy the obligation to provide shares. There were no amounts outstanding under this note payable as of December 31, 2003.
Ribopharma AG Notes
Upon the acquisition of Ribopharma AG, Alnylam Pharmaceuticals, Inc. assumed notes payable amounting to $2,551. The Company repaid $2,964 in August 2003, which included accrued interest of $413. There are no amounts outstanding under these notes as of December 31, 2003.
F-20
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
| 8. | Redeemable Convertible Preferred Stock |
The Company’s Series A and Series B redeemable convertible preferred stock and Series C convertible preferred stock, $0.001 par value, consists of the following as of December 31, 2002 and 2003:
| 2002 | 2003 | |||||||
| Series A redeemable convertible preferred stock; 2,000,010 and 3,000,010 shares authorized, issued and outstanding at December 31, 2002 and 2003, respectively (liquidation preference of $3,000) | $ | 2,081 | $ | 3,094 | ||||
| Series B redeemable convertible preferred stock; 6,200,000 and 16,672,078 shares authorized, 6,160,000 and 16,561,845 issued and outstanding at December 31, 2002 and 2003, respectively (liquidation preference of $41,405) | 16,003 | 44,641 | ||||||
| Series C convertible preferred stock; no shares authorized, issued and outstanding at December 31, 2002, 2,600,000 shares authorized, 1,504,825 shares issued and outstanding at December 31, 2003 (liquidation preference of $7,524) | — | 7,454 | ||||||
| $ | 18,084 | $ | 55,189 | |||||
As of December 31, 2003, the Company has 22,272,088 authorized shares of preferred stock, of which 3,000,010 shares are designated as Series A redeemable convertible preferred stock (“Series A preferred stock”), 16,672,078 shares are designated as Series B redeemable convertible preferred stock (“Series B preferred stock”), and 2,600,000 shares are designated as Series C convertible preferred stock (“Series C preferred stock”) (collectively, the “preferred stock”).
The Company sold 1,000,000 and 2,000,010 shares of Series A preferred stock resulting in gross proceeds of $1,000 and $2,000 in July 2003 and June 2002, respectively. In July 2003 and July 2002, the Company sold 10,401,845 and 6,160,000 shares of Series B preferred stock for $2.50 per share resulting in total consideration of $26,005 and $15,400, respectively. In September 2003 and October 2003, the Company issued a total of 1,504,825 shares of Series C preferred stock for gross proceeds of $7,454.
The Company issued 1,000,000 shares of Series A preferred stock in July 2003, which was convertible into common stock at a value of $1,260 at the date of issuance. As a result, the Company was required to record a beneficial conversion feature charge of approximately $260 which was recorded as a discount on the Series A preferred stock and an increase to additional paid-in capital. This discount will be accreted over the period of the first available redemption of the Series A preferred stock through periodic charges. During the year ended December 31, 2003, the Company recorded $27 of accretion related to the amortization of this discount. This accretion will increase the net loss attributable to common stockholders.
As of December 31, 2003, the rights, preferences, and privileges of the Company’s preferred stock are listed below:
Voting
The holders of preferred stock are entitled to vote, together with the holders of common stock, on all matters submitted to stockholders for a vote. Each preferred stockholder is entitled to the number of votes equal to the number of shares of common stock into which each preferred share is then convertible at the time of such vote.
F-21
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Dividends
The Company may declare and pay dividends as and when determined by the Company’s Board of Directors. As of December 31, 2003, no dividends had been declared or paid by the Company.
Conversion
Each share of preferred stock is convertible at the option of the holder, at any time and from time to time into one share of common stock adjusted for certain dilutive events. The conversion price for the Series A, Series B and Series C preferred stock is equal to the purchase price of $1.00, $2.50 and $5.00, respectively. All shares of preferred stock shall be automatically converted into shares of common stock upon the closing of an underwritten IPO with gross proceeds of not less than $25,000.
Liquidation
In the event of any voluntary or involuntary liquidation, dissolution or winding-up of the corporation, as defined, holders of the Series A and Series B preferred stock then outstanding will be entitled to be paid an amount equal to $1.00 per share and $2.50 per share, respectively, plus any dividends declared but unpaid on such shares, prior to any payment to common shareholders. Holders of the Series C convertible preferred stock will receive liquidation preference equal to $5.00 per share only after the Series A and Series B shareholders have received full payment of the Series A and Series B preference amounts. Amounts remaining after the preference payments to the preferred stockholders, if any, will be shared ratably among the holders of common stock.
Redemption
Subject to a written election of holders of at least two-thirds in voting power of the outstanding shares of preferred stock, the Company will be required to redeem, subject to certain conditions, on July 25, 2007, July 25, 2008 and July 25, 2009, each a Mandatory Redemption Date, the percentage of Series A and Series B preferred stock, as listed in the following table, at a price per share equal to $1.00 and $2.50, respectively, plus an additional amount equal to simple interest of ten percent per annum per year from the issuance date.
| Mandatory | Maximum Portion of Shares of | |||
| Redemption Date | Preferred Stock to be Redeemed | |||
| July 25, 2007 | 33% | |||
| July 25, 2008 | 67% | |||
| July 25, 2009 | 100% | |||
The Company initially recorded redeemable convertible preferred stock at fair value at the date of issuance. Where the carrying amount of the redeemable convertible preferred stock was less than the redemption amount, the carrying amount is increased by periodic accretion so that the carrying amount would equal the redemption amount at the first available redemption date. The carrying amount is further periodically increased by amounts representing the ten percent per annum interest feature. Accretion of the Company’s redeemable convertible preferred stock, including the ten percent interest during the period from
F-22
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and for the period cumulative from inception (June 14, 2002) through December 31, 2003, was as follows:
| Period from | Cumulative from | |||||||||||
| Inception | Inception | |||||||||||
| (June 14, 2002) | (June 14, 2002) | |||||||||||
| through | Year Ended | through | ||||||||||
| December 31, | December 31, | December 31, | ||||||||||
| 2002 | 2003 | 2003 | ||||||||||
| Accretion of Series A preferred stock | $ | 102 | $ | 273 | $ | 375 | ||||||
| Accretion of Series B preferred stock | 646 | 2,633 | 3,279 | |||||||||
| $ | 748 | $ | 2,906 | $ | 3,654 | |||||||
As of December 31, 2003, the redemption value of the Series A and Series B preferred stock was $3,342 and $44,670, respectively.
The Series C convertible preferred stock is not subject to mandatory or optional redemption; however, there are circumstances outside the control of the Company that could result in the holders of the Series C preferred stock being redeemed upon certain deemed liquidation events in limited circumstances. Accordingly, the Series C preferred stock has been classified as redeemable convertible preferred stock. The Series C preferred stock is not being accreted because there is no mandatory redemption date. The Series C preferred stock does have a liquidation value of $7,524 at December 31, 2003.
| 9. | Common Stock |
Common Stock
As of December 31, 2003, the Company had 34,739,392 shares of common stock authorized, and 4,277,860 shares issued. As of December 31, 2003, the Company has reserved 21,091,680 shares for issuance to preferred stockholders in the event that the redeemable convertible preferred stock and warrant to purchase 25,000 shares of Series B preferred stock is converted into common stock. In addition, the Company has reserved 4,250,000 shares of common stock for future issuance upon the exercise of common stock options.
In July 2003, Alnylam Pharmaceuticals, Inc. issued 1,549,216 shares of common stock for the acquisition of Ribopharma AG (Note 4).
Founders’ Shares
In June 2002, the Company sold 2,460,000 shares of common stock to the Company’s founders, including certain nonemployees, in exchange for $0.0001 per share, which represented the fair market value of the common stock on the date of sale, as determined by management. The founders’ common stock is subject to restricted stock agreements, which include various restrictions, including the right of the Company to repurchase declining percentages of the shares at the original issuance price during the four-year period following issuance if the employee or nonemployee ceases to provide services to the Company for any reason. In July 2002, the Company sold 90,000 shares of common stock to a consultant for $0.10 per share, which represented the fair market value of the common stock on the date of sale, as determined by management. This common stock is subject to a restricted stock agreement, which includes various restrictions, including the right of the Company to repurchase declining percentages of the shares at the original issuance price during the four-year period following issuance if the consultant ceases to perform services.
F-23
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
In connection with the restricted stock award issued to nonemployees, the Company has recorded cumulative deferred compensation of $3,791, which represents the cumulative fair value of the restricted stock awards measured in accordance with SFAS No. 123 and EITF 96-18. Shares remaining unvested or subject to forfeiture for nonemployees still providing services are subject to a mark-to-market adjustment during each reporting period prior to vesting in full. The deferred compensation will be recorded as an expense over the vesting period of the underlying restricted stock using the method prescribed by FASB Interpretation No. 28 (FIN 28),Accounting for Stock Appreciation Rights and Other Variable Stock Options or Award Plans. The Company recorded $172, $2,241 and $2,413 of compensation expense during the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and for the period cumulative from inception (June 14, 2002) through December 31, 2003, respectively, related to the amortization of the deferred compensation. The deferred compensation balance at December 31, 2002 and 2003, respectively, related to these awards was $161 and $1,217. Since the fair market value of the common stock to nonemployees is subject to change in the future, the compensation expense recognized during the year ended December 31, 2003, and prior years may not be indicative of future compensation charges.
Board of Directors Actions
The Company’s board of directors approved, subject to stockholder approval, the following on February 23, 2004:
| • | Amendment to the certificate of incorporation increasing the number of authorized shares of common stock, $0.0001 par value per share, from 34,739,392 shares to 125,000,000 shares. | |
| • | Amendment and restatement of the certificate of incorporation, subject to the closing of the Company’s IPO, the number of authorized shares of common stock, $0.0001 par value per share, will be 125,000,000 and the number of authorized shares of preferred stock, $0.0001 par value per share, will be decreased to 5,000,000. The board of directors will have the authority to issue such shares of preferred stock in one or more series and fix the relative rights and preferences without vote or action by the stockholders. |
| 10. | Stock Option Plan |
In June 2002, the Company adopted the 2002 Stock Incentive Plan (the “2002 Stock Plan”), which was terminated in November 2002, and replaced with the Alnylam U.S., Inc. 2002 Employee, Director and Consultant Stock Plan (the “2002 Plan”). All options previously granted under the 2002 Stock Plan were canceled and new options for the same number of shares, vesting provisions and exercise price were granted under the 2002 Plan. In September 2003, the Company adopted the Alnylam Pharmaceuticals, Inc. 2003 Employee, Director and Consultant Stock Plan (the “2003 Plan”). As of December 31, 2003, an aggregate of 4,250,000 shares of common stock were authorized for issuance under the 2002 and 2003 plans, of which 947,644 were available for future grant at December 31, 2003.
The plans provide for the granting of incentive stock options (“ISOs”) and nonqualified stock options. Stock options may be granted to the Company’s employees, officers, directors, consultants and advisors, as defined. ISOs may be granted at no less than fair market value (“FMV”) on the date of grant, as determined by the Company’s Board of Directors (no less than 110 percent of FMV on the date of grant for 10 percent or greater stockholders), subject to limitations, as defined. Each option shall be exercisable at such times and subject to such terms as determined by the Board of Directors and expires within ten years of issuance.
Options granted generally vest at a rate of 25 percent on the last of the month in which the first anniversary of the grant date occurs and 6.25 percent of the shares at the end of each successive three-month
F-24
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
period until fully vested. In January 2004, the Company granted an option to the chief executive officer to purchase 200,000 shares of common stock at an exercise price of $0.50 per share that vest as to 25 percent of the shares on the first anniversary of the date of grant and as to an additional 6.25 percent of the shares at the end of each three-month period thereafter, except that it shall vest as to 100 percent of the shares upon an initial public offering.
The following table summarizes the activity of the Company’s stock option plans:
| Number of | ||||||||||||
| Options | Weighted | |||||||||||
| Available for | Average | |||||||||||
| Future | Number of | Exercise | ||||||||||
| Grant | Options | Price | ||||||||||
Outstanding at inception (June 14, 2002) | — | — | $ | — | ||||||||
| Granted | 311,200 | 0.25 | ||||||||||
| Exercised | — | — | ||||||||||
| Canceled | (155,600 | ) | 0.25 | |||||||||
Outstanding at December 31, 2002 | 1,294,400 | 155,600 | 0.25 | |||||||||
| Granted | 3,146,756 | 0.28 | ||||||||||
| Exercised | (84,600 | ) | 0.25 | |||||||||
| Canceled | — | |||||||||||
Outstanding at December 31, 2003 | 947,644 | 3,217,756 | $ | 0.28 | ||||||||
| Exercisable at December 31, 2003 | 366,151 | $ | 0.25 | |||||||||
| Exercisable at December 31, 2002 | 1,600 | $ | 0.10 | |||||||||
The Company has also committed to issuing an option to purchase 50,000 shares of common stock to an employee at the then fair market value upon entering into a strategic alliance, as defined. As of December 31, 2003, this event had not been achieved.
All options granted during the period from inception (June 14, 2002) through December 31, 2002, had an exercise price that was equal to the fair market value of common stock on the date of grant. The weighted average fair value of these options granted at fair market value during the period from inception (June 14, 2002) through December 31, 2002 was $0.03. All options granted during the year ended December 31, 2003, had an exercise price that was less than the fair value of common stock on the date of grant. The weighted average fair value of these options was $1.25.
F-25
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
The following table summarizes information about stock options outstanding and exercisable at December 31, 2003:
| Options Outstanding | ||||||||||||||||||||||
| Weighted | Options Exercisable | |||||||||||||||||||||
| Average | ||||||||||||||||||||||
| Remaining | Weighted | Weighted | ||||||||||||||||||||
| Contractual | Average | Average | ||||||||||||||||||||
| Exercise | Number | Life | Exercise | Number | Exercise | |||||||||||||||||
| Price | Outstanding | (in Years) | Price | Exercisable | Price | |||||||||||||||||
| $ | 0.25 | 2,777,756 | 9.27 | $ | 0.25 | 366,151 | $ | 0.25 | ||||||||||||||
| 0.50 | 440,000 | 9.83 | 0.50 | — | 0.50 | |||||||||||||||||
| 3,217,756 | 366,151 | |||||||||||||||||||||
During the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and the period cumulative from inception (June 14, 2002) through December 31, 2003, in connection with the grant of common stock options to employees, the Company recorded deferred stock compensation of approximately zero, $3,338 and $3,338, respectively, representing the difference between the exercise price and the fair market value of the Company’s common stock on the date the stock options were granted. During the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003 and the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company recorded amortization of deferred stock compensation of zero, $667 and $667, respectively, and $2,671 remains unamortized at December 31, 2003. The Company will be recognizing the fixed deferred stock compensation over the remaining vesting period of the options, subject to forfeitures should the employees terminate, in accordance with the method prescribed by FIN 28. The anticipated future amortization of deferred stock compensation related to employee option grants as of December 31, 2003 is as follows:
| Year Ended December 31, | ||||
| 2004 | $ | 1,416 | ||
| 2005 | 744 | |||
| 2006 | 381 | |||
| 2007 | 129 | |||
| $ | 2,670 | |||
In January 2004, the Company granted 687,543 common stock options to employees with an exercise price of $0.50 per share. The Company will record additional deferred compensation for the difference between the option exercise price and its fair value.
In connection with stock options granted to nonemployees for services during the year ended December 31, 2003, the Company has recorded aggregate deferred compensation of approximately $1,178, which represents the fair value of nonemployee grants. The deferred compensation will be recorded as an expense over the vesting period of the underlying stock options using the method prescribed by FIN 28. At the end of each financial reporting period prior to vesting, the value of these options (as calculated using the Black-Scholes option pricing model) will be remeasured using the then current fair value of the Company’s
F-26
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
common stock. At that point, deferred compensation and the noncash compensation recognized during that period will be adjusted accordingly. Accordingly, since the fair market value of the common stock options granted to nonemployees is subject to change in the future, the amount of future compensation expense recognized will be adjusted up until the stock options, are fully vested. Stock-based compensation expense related to these nonemployee options for the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and the period cumulative from inception (June 14, 2002) through December 31, 2003, was $0, $547 and $547, respectively.
| 11. | Income Taxes |
Deferred income taxes reflect the tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting and income tax purposes. A valuation allowance is established when uncertainty exists as to whether all or a portion of the net deferred tax assets will be realized. Components of the net deferred tax asset as of December 31, 2002 and 2003, are approximately as follows:
| 2002 | 2003 | ||||||||
| Deferred Tax Assets: | |||||||||
| Net operating loss carryforwards | $ | 243 | $ | 2,628 | |||||
| Research and development credits | 33 | 238 | |||||||
| Capitalized research and development and start-up costs | 585 | 6,239 | |||||||
| Deferred revenue | — | 761 | |||||||
| Other | 1 | 839 | |||||||
| Total deferred tax assets | 862 | 10,705 | |||||||
| Deferred Tax Liabilities: | |||||||||
| Intangible assets | — | (1,788 | ) | ||||||
| Deferred tax asset valuation allowance | (862 | ) | (8,917 | ) | |||||
| Net deferred tax assets | $ | — | $ | — | |||||
F-27
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
The Company’s effective income tax rate differs from the statutory federal income tax rate as follows for the period from inception (June 14, 2002) through December 31, 2002 and the year ended December 31, 2003:
| Period from | ||||||||
| Inception | ||||||||
| (June 14, | ||||||||
| 2002) | ||||||||
| through | Year Ended | |||||||
| December 31, | December 31, | |||||||
| 2002 | 2003 | |||||||
| Federal tax benefit rate | 34.0 | % | 34.0 | % | ||||
| State tax benefit, net of federal benefit | 6.2 | 5.0 | ||||||
| Permanent items | 0.8 | (2.2 | ) | |||||
| Purchased in-process research and development | — | (6.0 | ) | |||||
| Federal research credits | 0.9 | 0.6 | ||||||
| Valuation allowance | (41.9 | ) | (31.4 | ) | ||||
| Effective income tax rate | 0.0 | % | 0.0 | % | ||||
As required by SFAS No. 109, management of the Company has evaluated the positive and negative evidence bearing upon the realizability of its deferred tax assets. Management has concluded, in accordance with the applicable accounting standards, that it is more likely than not that the Company may not realize the benefit of its deferred tax assets. Accordingly, the deferred tax assets have been fully reserved. Management reevaluates the positive and negative evidence on an annual basis.
At December 31, 2003, the Company had federal and state net operating loss carryforwards of approximately $1,494 and $1,359 available, respectively, to reduce future taxable income and which will expire at various dates beginning in 2007 through 2023. At December 31, 2003, federal and state research and development credit carryforwards were approximately $164 and $112, respectively, available to reduce future tax liabilities, and, which expire at various dates beginning in 2017 through 2023. Ownership changes, as defined in the Internal Revenue Code, including those resulting from the issuance of common stock in connection with the Company’s planned initial public offering, may limit the amount of net operating loss and tax credit carryforwards that can be utilized to offset future taxable income or tax liability. The amount of the limitation is determined in accordance with Section 382 of the Internal Revenue Code.
12. Commitments and Contingencies
Operating Leases
The Company leases office and laboratory space in Cambridge, Massachusetts and Kulmbach, Germany (beginning on July 31, 2003 the date of acquisition of Ribopharma AG), under noncancelable operating lease agreements. Total rent expense under these operating leases was $92, $966, and $1,058 for the period from June 14, 2002 (date of inception) through December 31, 2002, for the year ended December 31, 2003 and for the period cumulative from inception (June 14, 2002) through December 31, 2003, respectively.
In September 2003, the Company entered into an operating lease to rent office space in Cambridge, Massachusetts through September 2011. Rental payments begin upon the earlier of April 1, 2004 or the date the Company takes occupancy. The Company can extend the lease for two successive five-year extensions.
F-28
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Pursuant to the terms of the lease agreement, the Company is required to deposit $2,313 and has thus classified this amount as long-term restricted cash in the accompanying consolidated balance sheet.
Future minimum lease payments under this noncancelable lease are approximately as follows:
| Minimum | |||||
| Lease | |||||
| Year Ending December 31, | Payments | ||||
| 2004 | $ | 1,637 | |||
| 2005 | 1,910 | ||||
| 2006 | 2,072 | ||||
| 2007 | 2,110 | ||||
| 2008 | 2,125 | ||||
| Thereafter | 5,392 | ||||
| Total | $ | 15,246 | |||
Commitment to Ribopharma AG Shareholders
In connection with the acquisition of Ribopharma AG, the Company agreed to provide the two shareholders of Ribopharma AG who received cash and common stock in the acquisition with a nonrecourse loan to cover any tax contingencies the shareholders may incur as a result of the acquisition. If the loan is provided, the note will bear interest at four percent per annum and be payable upon certain liquidity events, as defined. In addition to the loan commitment, the Company has also entered into an indemnity agreement whereby the Company will indemnify the shareholders for any taxes payable as a result of making the loan to the Ribopharma shareholders up to a maximum of approximately $179 for each shareholder as of December 31, 2003. As of December 31, 2003, the Company has not been required to provide the two shareholders with a loan or provide any amounts under the indemnity agreements. With respect to the indemnity, the Company has issued a letter of credit to the two shareholders amounting to $354 related to the potential indemnity that the Company has with the two shareholders. The required amount of the letter of credit is collateralized by restricted funds maintained by the Company at the bank issuing the letter of credit. As a result, the Company has classified this amount as restricted cash in the accompanying consolidated balance sheet.
In connection with the employment agreements of the same two Ribopharma AG employees, the Company has committed to paying a one-time payment to each employee of $250 upon the issuance of a specific patent in the United States of America. This contingent payment will be paid and expensed upon the issuance of the patent.
Consulting Agreements with Founders
The Company has consulting agreements with certain founding nonemployee stockholders, subject to certain termination rights. During the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, and the period cumulative from inception (June 14, 2002) through December 31, 2003, the Company paid the founders $128, $282 and $410, respectively, in connection with these consulting agreements. The Company’s aggregate remaining cash commitment pursuant to such consulting agreements amounted to approximately $360 at December 31, 2003.
F-29
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Guarantees
In November 2002, the FASB issued FIN No. 45,Guarantor’s Accounting and Disclosure Requirements for Guarantees, Including Indirect Guarantees of Indebtedness of Others an interpretation of FASB Statements No. 5, 57, and 107 and rescission of FASB Interpretation No 34. The Interpretation requires additional disclosures to be made by a guarantor in its annual financial statements about its obligations under certain guarantees it has issued. The accounting requirements for the initial recognition of guarantees are applicable on a prospective basis for guarantees issued or modified after December 31, 2002. The adoption of FIN No. 45 did not have a material effect on the Company’s financial statements.
The Company is also a party to a number of agreements entered into in the ordinary course of business, which contain typical provisions, which obligate the Company to indemnify the other parties to such agreements upon the occurrence of certain events. Such indemnification obligations are usually in effect from the date of execution of the applicable agreement for a period equal to the applicable statute of limitations. The aggregate maximum potential future liability of the Company under such indemnification provisions is uncertain. Since its inception, the Company has not incurred any expenses as a result of such indemnification provisions. Accordingly, the Company has determined that the estimated aggregate fair value of its potential liabilities under such indemnification provisions is minimal and has not recorded any liability related to such indemnification provisions as of December 31, 2003.
Legal Proceedings
The Company may periodically become subject to legal proceedings and claims arising in connection with on-going business activities, including being subject to claims or disputes around patents that have been issued or are pending in the field of research the Company is focused on. The Company does not believe that there were any material claims against the Company as of December 31, 2003.
13. 401(k) Savings Plan
The 401(k) Plan allows participants to defer up to 15 percent of their annual compensation and with proper notice up to 100 percent of their compensation in the final month of the plan year on a pretax basis and subject to IRC limits. The plan covers substantially all of the employees who meet minimum age and service requirements. The Company may make matching contributions to the 401(k) Plan in amounts determined by the Company’s Board of Directors. The Company did not contribute to the 401(k) plan during the period from inception (June 14, 2002) through December 31, 2002, the year ended December 31, 2003, or the period cumulative from inception (June 14, 2002) through December 31, 2003.
14. Significant Agreements
Garching Innovation GmbH License Agreement
In December 2002, the Company entered into a co-exclusive license with Garching Innovation GmbH (“Garching”) for the worldwide rights to use and sublicense certain patented technology to develop and commercialize therapeutic products and related applications. The Company also obtained the rights to use without the right to sublicense, the technology for all diagnostic uses other than for the purposes of therapeutic monitoring. In consideration for the rights to license this technology, the Company issued to Garching 723,240 shares of Series B redeemable convertible preferred stock in 2003. As of December 31, 2002, the Company valued this consideration at the Series B redeemable convertible preferred stock issuance price of $2.50 per share for total consideration of $1,808. As the Company had not issued the shares as of December 31, 2002, the Company has included this amount in accrued expenses within the accompanying
F-30
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
balance sheet and recorded the consideration as license fee expense as the technology has not reached technological feasibility and does not have any alternative future use. In July 2003, the Company issued the 723,240 shares of Series B redeemable preferred stock to Garching. The Company will also be required to pay future royalties on net sales of all therapeutic and prophylactic products developed with the technology.
The Company was also given the ability to acquire the remaining 50 percent exclusive rights to the technology that had not been previously granted to the Company by Garching upon the establishment of a German-based company with comparable operational work force and resources. The Company successfully obtained the remaining 50 percent exclusive rights upon the acquisition of Ribopharma AG in July 2003 (Note 4) and in consideration for the remaining rights to this technology, issued 158,605 shares of Series B redeemable convertible preferred stock. These shares were determined to have a fair value of $397 and the value was recorded as license fee expense. The Company is also reserving an additional 16,328 shares of Series B redeemable convertible preferred stock which is contingently issuable if a specified claim is obtained related to one of its licensed patents.
Collaboration Agreement with Merck & Co.
In September 2003, the Company entered into a five-year strategic alliance with Merck & Co. (“Merck”) to develop RNAi-based technology and therapeutics. For technology development, Merck and Alnylam each committed to devote resources, including full-time equivalents and expertise to the collaborative development of advanced RNAi technology. Merck will have rights to use this technology solely for the identification and validation of drug targets; Alnylam will have rights to use it for these purposes and also for therapeutic purposes. For therapeutics development, Merck agreed to provide Alnylam with twelve proprietary drug targets as potential targets for siRNA therapeutics. Alnylam has the right, but not the obligation, to develop siRNA drug candidates against each target provided by Merck. If Alnylam advances a candidate to a defined point in preclinical development, Alnylam and Merck will then decide whether Alnylam, Merck or the two companies together will proceed with the further development and commercialization of that candidate. For each drug candidate in whose development Merck decides to participate, it will make a cash payment to Alnylam at the time of its decision, and will also reimburse Alnylam for a portion of the costs Alnylam has so far incurred on that candidate.
In connection with this alliance, Merck made an upfront cash payment of $2,000 and a $5,000 equity investment in Alnylam during 2003. Further cash payments are due from Merck in each of 2004 and 2005, based upon the continuation of the alliance. In addition, if a prespecified technology milestone is achieved in the RNAi technology development program, Merck will make an additional cash payment and an additional equity investment. The Company is recognizing the revenue related to the $2,000 upfront payment ratably over the period of continuing performance obligations, which is estimated as six years. As of December 31, 2003, the Company has recognized approximately $111 as revenue and has deferred approximately $1,900.
15. Subsequent Event
Equipment Line of Credit
On March 30, 2004, the Company entered into an agreement with Lighthouse Capital Partners V, L.P. to establish an equipment line of credit for $10,000. The company has the ability to draw down amounts under the line of credit through June 30, 2005 upon adherence to certain conditions. All borrowings under the line of credit are collateralized by the assets financed. Borrowings bear interest at prime rate plus 3 percent and each advance is repayable over at least a 48 month period from the date of each advance. On the maturity of each equipment advance under the line of credit, the Company is required to pay, in addition to the paid principal and interest, an additional amount of 11.5 percent of the original principal. Alnylam also issued
F-31
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
Lighthouse a warrant to purchase 100,000 shares of Series C convertible preferred stock at an exercise price of $5.00 per share.
In conjunction with entering into the agreement, Alnylam paid off the remaining balance of the loan with Silicon Valley Bank via an initial draw in the amount of the payoff balance. As a result of the early termination of the Silicon Valley Bank loan, Alnylam paid additional interest of $168.
Isis Pharmaceuticals, Inc. Collaboration and License Agreement
In March 2004, Alnylam entered into a collaboration and license agreement with Isis Pharmaceuticals, Inc. Isis granted Alnylam licenses to its current and future patents and patent applications relating to chemistry and to RNA-targeting mechanisms for the research, development and commercialization of double-stranded RNA products. Alnylam has the right to use Isis technologies in its development programs or in collaborations and Isis has agreed not to grant licenses under these patents to any other organization for the discovery, development and commercialization of double-stranded RNA products designed to work through a RNAi mechanism, except in the context of a collaboration in which Isis plays an active role. Alnylam granted Isis non-exclusive licenses to its current and future patents and patent applications relating to RNA-targeting mechanisms and to chemistry for research use. Alnylam also granted Isis the exclusive or co-exclusive right to develop and commercialize double-stranded RNA products developed using RNAi technology against a limited number of targets. In addition, Alnylam granted Isis non-exclusive rights to research, develop and commercialize single-stranded RNA products.
Under the terms of the agreement, Alnylam agreed to pay Isis an upfront license fee of $5,000, $3,000 of which was paid upon signing of the agreement and the remaining $2,000 of which is due on January 3, 2005. Alnylam also agreed to pay royalties and milestones to Isis on any products that Alnylam or a collaborator develop utilizing Isis intellectual property. In addition, Alnylam agreed to pay to Isis a percentage of some fees from strategic collaborations it may enter into that include access to the Isis intellectual property. In conjunction with the agreement, Isis purchased 1,666,667 shares of series D preferred stock of Alnylam for $10,000. Isis also agreed to pay Alnylam a license fee, royalty and milestone payments on any product developed by Isis or a collaborator that utilizes its intellectual property. The agreement also gives Alnylam an option to use Isis’ manufacturing services for RNA-based therapeutics.
The agreement with Isis also gives Alnylam the exclusive right to grant sub-licenses for Isis technology to third parties with whom Alnylam is not collaborating. Alnylam may include these sub-licenses in its InterfeRx licenses and research reagent and services licenses. If a license includes rights to Isis’ intellectual property, Alnylam will share revenues from that license equally with Isis.
Contractor Commitment
On March 15, 2004, the Company entered into an agreement with a contractor for the build out of the Company’s new facility in Cambridge, Massachusetts. The contract contains a guaranteed maximum price of $5,401. As a part of the lease agreement signed with the landlord, the landlord will reimburse the Company for certain of the costs of the build out, up to a maximum of $2,978.
Actions of the Board of Directors
The Company’s Board of Directors approved the following on March 30, 2004:
| • | Effective upon the closing of an initial public offering, no further stock options or other equity awards will be granted under the 2002 Plan or 2003 Plan; |
F-32
Table of Contents
Notes to Consolidated Financial Statements — (Continued)
| • | Effective upon the effective date of the registration statement relating to the IPO and subject to shareholder approval, authorization of the 2004 Stock Incentive Plan (the “2004 Plan”) with 3,000,000 shares authorized for future issuance under the plan plus an additional amount authorized based on the remaining shares available under the 2003 plan as of the closing of the IPO. The 2004 Plan includes a nonemployee director stock option program under which each eligible nonemployee director will be entitled to receive an annual grant of options to purchase 13,500 shares of common stock upon his or her initial appointment to the board of directors and a subsequent annual grant of an option to purchase 10,000 shares of common stock based on continued service; and | |
| • | Authorization of a 2004 Employee Stock Purchase Plan with 600,000 shares authorized for issuance under the plan commencing after the closing of an initial public offering and subject to shareholder approval. | |
F-33
Table of Contents
Report of Independent Auditors
To the Supervisory Board and Board of Directors of
In our opinion, the accompanying balance sheets and the related statements of operations, stockholders’ deficit and cash flows present fairly, in all material aspects, the financial position of Ribopharma AG (a development stage enterprise) at December 31, 2001 and 2002 and the results of its operations and its cash flows for the years then ended and for the period from June 16, 2000 (inception) through December 31, 2002, in accordance with accounting principles generally accepted in the United States of America. These financial statements are the responsibility of the Company’s management; our responsibility is to express an opinion on these financial statements based on our audits. We conducted our audit of these financial statements in accordance with auditing standards generally accepted in the United States of America, which require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
PricewaterhouseCoopers
| /s/ PRICE | /s/ MCMAHON | |||
| Chartered Accountant | Chartered Accountant | |||
Munich, December 23, 2003
F-34
Table of Contents
RIBOPHARMA AG
| December 31, | June 30, | ||||||||||||||
| 2001 | 2002 | 2003 | |||||||||||||
| € | |||||||||||||||
| € | € | (Unaudited) | |||||||||||||
| Assets | |||||||||||||||
| Current assets | |||||||||||||||
| Cash and cash equivalents | 119,598 | 2,025,601 | 1,092,815 | ||||||||||||
| Prepaid expenses and other current assets | 18,211 | 46,606 | 56,536 | ||||||||||||
| Total current assets | 137,809 | 2,072,207 | 1,149,351 | ||||||||||||
| Property and equipment, net | 310,684 | 573,759 | 702,161 | ||||||||||||
| Total assets | 448,493 | 2,645,966 | 1,851,512 | ||||||||||||
| Liabilities and Stockholders’ Deficit | |||||||||||||||
| Current liabilities | |||||||||||||||
| Accounts payable | 25,861 | 77,438 | 59,391 | ||||||||||||
| Accrued liabilities | 78,995 | 102,100 | 227,233 | ||||||||||||
| Other current liabilities | 40,976 | 38,207 | 69,369 | ||||||||||||
| Accrued interest on notes payable | 35,885 | 131,540 | 509,446 | ||||||||||||
| Current portion of note payable from stockholder | — | 2,000,000 | 3,000,000 | ||||||||||||
| Current portion of notes payable (Note 6) | 464,000 | — | 2,249,584 | ||||||||||||
| Deferred revenue | — | — | 35,650 | ||||||||||||
| Deferred grant | 60,594 | 73,983 | 73,983 | ||||||||||||
| Total current liabilities | 706,311 | 2,423,268 | 6,224,656 | ||||||||||||
| Notes payable, net of current portion (Note 6) | 849,469 | 2,249,584 | — | ||||||||||||
| Deferred grant, net of current portion | 177,293 | 177,887 | 140,440 | ||||||||||||
| Total liabilities | 1,026,762 | 2,427,471 | 140,440 | ||||||||||||
| Stockholders’ deficit | |||||||||||||||
Common stock of€ 1.00 par value | |||||||||||||||
| Number of shares issued and outstanding 55,560, 68,560, and 68,560 at December 31, 2001, 2002, and June 30, 2003 (unaudited), respectively | 55,560 | 68,560 | 68,560 | ||||||||||||
| Additional paid-in capital | — | 1,208,440 | 1,208,440 | ||||||||||||
| Deficit accumulated during the development stage | (1,340,140 | ) | (3,481,773 | ) | (5,790,584 | ) | |||||||||
| Total stockholders’ deficit | (1,284,580 | ) | (2,204,773 | ) | (4,513,584 | ) | |||||||||
| Total liabilities and stockholders’ deficit | 448,493 | 2,645,966 | 1,851,512 | ||||||||||||
The accompanying notes are an integral part of these financial statements.
F-35
Table of Contents
RIBOPHARMA AG
| Period from | |||||||||||||||||||||||||
| June 16, 2000 | |||||||||||||||||||||||||
| (Inception) | Period From | ||||||||||||||||||||||||
| For the Year Ended | through | For the Six Months | June 16, 2000 | ||||||||||||||||||||||
| December 31, | December 31, | Ended June 30, | (inception) | ||||||||||||||||||||||
| through | |||||||||||||||||||||||||
| 2001 | 2002 | 2002 | 2002 | 2003 | June 30, 2003 | ||||||||||||||||||||
| € | € | € | € | € | € | ||||||||||||||||||||
| (Unaudited) | |||||||||||||||||||||||||
| (Unaudited) | |||||||||||||||||||||||||
| Revenues from licenses | — | 43,166 | 43,166 | 18,166 | 35,650 | 78,816 | |||||||||||||||||||
| Costs and expenses | |||||||||||||||||||||||||
| Research and development | (609,037 | ) | (1,251,614 | ) | (1,937,153 | ) | (573,355 | ) | (1,125,016 | ) | (3,062,169 | ) | |||||||||||||
| General and administrative | (490,081 | ) | (745,155 | ) | (1,359,085 | ) | (313,501 | ) | (780,484 | ) | (2,139,569 | ) | |||||||||||||
| Total costs and expenses | (1,099,118 | ) | (1,996,769 | ) | (3,296,238 | ) | (886,856 | ) | (1,905,500 | ) | (5,201,738 | ) | |||||||||||||
| Loss from operations | (1,099,118 | ) | (1,953,603 | ) | (3,253,072 | ) | (868,690 | ) | (1,869,850 | ) | (5,122,922 | ) | |||||||||||||
| Interest income | 15,039 | 22,869 | 41,946 | 5,234 | 17,509 | 59,455 | |||||||||||||||||||
| Interest expense | (59,748 | ) | (210,899 | ) | (270,647 | ) | (56,104 | ) | (456,470 | ) | (727,117 | ) | |||||||||||||
| Net loss | (1,143,827 | ) | (2,141,633 | ) | (3,481,773 | ) | (919,560 | ) | (2,308,811 | ) | (5,790,584 | ) | |||||||||||||
The accompanying notes are an integral part of these financial statements.
F-36
Table of Contents
RIBOPHARMA AG
| Deficit | ||||||||||||||||||||
| Common Stock | Accumulated | |||||||||||||||||||
| Additional | During the | |||||||||||||||||||
| Number of | Paid-in | Development | ||||||||||||||||||
| Shares | Amount | Capital | Stage | Total | ||||||||||||||||
| € | € | € | € | |||||||||||||||||
| Balance at inception (June 16, 2000) | 50,000 | 50,000 | — | — | 50,000 | |||||||||||||||
| Stock issuance | 5,560 | 5,560 | — | — | 5,560 | |||||||||||||||
| Net loss | — | — | — | (196,313 | ) | (196,313 | ) | |||||||||||||
| Balance December 31, 2000 | 55,560 | 55,560 | — | (196,313 | ) | (140,753 | ) | |||||||||||||
| Net loss | — | — | — | (1,143,827 | ) | (1,143,827 | ) | |||||||||||||
| Balance December 31, 2001 | 55,560 | 55,560 | — | (1,340,140 | ) | (1,284,580 | ) | |||||||||||||
| Stock issuance | 13,000 | 13,000 | 1,208,440 | — | 1,221,440 | |||||||||||||||
| Net loss | — | — | — | (2,141,633 | ) | (2,141,633 | ) | |||||||||||||
| Balance December 31, 2002 | 68,560 | 68,560 | 1,208,440 | (3,481,773 | ) | (2,204,773 | ) | |||||||||||||
| Net loss (unaudited) | — | — | — | (2,308,811 | ) | (2,308,811 | ) | |||||||||||||
| Balance June 30, 2003 (unaudited) | 68,560 | 68,560 | 1,208,440 | (5,790,584 | ) | (4,513,584 | ) | |||||||||||||
The accompanying notes are an integral part of these financial statements.
F-37
Table of Contents
RIBOPHARMA AG
| Period from | |||||||||||||||||||||||||
| June 16, 2000 | Period from | ||||||||||||||||||||||||
| For the Year Ended | (Inception) | For the Six Months | June 16, 2000 | ||||||||||||||||||||||
| December 31, | through | Ended June 30, | (Inception) | ||||||||||||||||||||||
| December 31, | through | ||||||||||||||||||||||||
| 2001 | 2002 | 2002 | 2002 | 2003 | June 30, 2003 | ||||||||||||||||||||
| € | € | € | € | € | € | ||||||||||||||||||||
| (Unaudited) | |||||||||||||||||||||||||
| (Unaudited) | |||||||||||||||||||||||||
| Cash flows from operating activities | |||||||||||||||||||||||||
| Net loss | (1,143,827 | ) | (2,141,633 | ) | (3,481,773 | ) | (919,560 | ) | (2,308,811 | ) | (5,790,584 | ) | |||||||||||||
| Adjustments to reconcile net loss to net cash flows used in operating activities | |||||||||||||||||||||||||
| Depreciation and amortization | 68,043 | 127,397 | 209,680 | 56,325 | 90,823 | 300,503 | |||||||||||||||||||
| Changes in assets and liabilities | |||||||||||||||||||||||||
| Prepaid expenses and other current assets | (1,820 | ) | (28,395 | ) | (46,606 | ) | 542,477 | (9,930 | ) | (56,536 | ) | ||||||||||||||
| Accounts payable and accrued liabilities | 60,231 | 170,337 | 311,078 | 4,048 | 484,992 | 796,070 | |||||||||||||||||||
| Other liabilities | 7,689 | (2,769 | ) | 38,207 | 4,525 | 66,812 | 105,019 | ||||||||||||||||||
| Deferred equipment grant | 70,764 | 13,983 | 251,870 | 55,481 | (37,447 | ) | 214,423 | ||||||||||||||||||
| Net cash used in operating activities | (938,920 | ) | (1,861,080 | ) | (2,717,544 | ) | (256,704 | ) | (1,713,561 | ) | 4,431,105 | ||||||||||||||
| Cash flows from investing activities | |||||||||||||||||||||||||
| Purchases of property and equipment | (136,530 | ) | (390,472 | ) | (783,439 | ) | (245,784 | ) | (219,225 | ) | (1,002,664 | ) | |||||||||||||
| Net cash used in investing activities | (136,530 | ) | (390,472 | ) | (783,439 | ) | (245,784 | ) | (219,225 | ) | (1,002,664 | ) | |||||||||||||
| Cash flows from financing activities | |||||||||||||||||||||||||
| Proceeds from issuance of common stock | — | 13,000 | 68,560 | — | — | 68,560 | |||||||||||||||||||
| Proceeds from notes payable | 849,469 | 2,144,555 | 3,458,024 | 1,012,163 | — | 3,458,024 | |||||||||||||||||||
| Proceeds from note payable from stockholders | — | 2,000,000 | 2,000,000 | — | 1,000,000 | 3,000,000 | |||||||||||||||||||
| Net cash provided by financing activities | 849,469 | 4,157,555 | 5,526,584 | 1,012,163 | 1,000,000 | 6,526,584 | |||||||||||||||||||
| Net (decrease) increase in cash and cash equivalents | (225,981 | ) | 1,906,003 | 2,025,601 | 509,675 | (932,786 | ) | 1,092,815 | |||||||||||||||||
| Cash and cash equivalents, beginning | 345,579 | 119,598 | — | 119,598 | 2,025,601 | 2,025,601 | |||||||||||||||||||
| Cash and cash equivalents, ending | 119,598 | 2,025,601 | 2,025,601 | 629,273 | 1,092,815 | 3,118,416 | |||||||||||||||||||
| Supplemental disclosure of noncash cash flows: | |||||||||||||||||||||||||
| Interest paid | 23,863 | 115,244 | 139,107 | 22,293 | 78,564 | 217,671 | |||||||||||||||||||
| Supplemental disclosure of noncash financing activities: | |||||||||||||||||||||||||
| Conversion of note payable into shares of common stock | — | 1,208,440 | 1,208,440 | — | — | 1,208,440 | |||||||||||||||||||
The accompanying notes are an integral part of these financial statements.
F-38
Table of Contents
RIBOPHARMA AG
Notes to the Financial Statements
1. Nature of the Business
Ribopharma AG (the “Company”) was incorporated on June 16, 2000 and is registered in the Commercial Register at the Amtsgericht (Local Court) in Bayreuth.
The Company pursues innovative, application-related research and development in the field of Life Science, Agro-biotechnology, Medical Biotechnology and Molecular Medicine. Since its inception, the Company has devoted substantially all of its efforts to business planning, research and development, recruiting management and technical staff, and raising capital. Accordingly, the Company is considered a development stage enterprise as defined in the Statement of Financial Accounting Standards (“SFAS”) No. 7 and the accompanying financial statements represent those of a development stage enterprise.
The accompanying financial statements have been prepared on a basis which assumes that the Company will continue as a going concern and which contemplates the realization of assets and satisfaction of liabilities and commitments in the normal course of business. The Company has a limited operating history and has incurred losses from operations since its inception.
On July 31, 2003, Alnylam Holding Co., now Alnylam Pharmaceuticals, Inc., and subsequently referred to herein as “Alnylam”, signed a Share Exchange Agreement with Ribopharma AG. Under the terms of the agreement, shareholders of Ribopharma AG exchanged substantially all their outstanding shares for 1,549,216 shares of Alnylam common stock and approximately€ 1,322,000 in cash.
The future viability of the Company is largely dependent on the ability of Alnylam to raise additional capital to finance its operations. Although management continues to pursue additional financing plans, there is no assurance that the Company will be successful in obtaining financing on terms acceptable to the Company.
The Company is subject to risks common to companies in the drug development and pharmaceutical industry, including but not limited to, the successful development and commercialization of drug candidates, fluctuations in operating results and financial risks, potential need for additional financing, protection of proprietary technology and patent risks, compliance with government regulations, dependence on key personnel and collaborative partners, competition, customer demand, management of growth, and the effectiveness of marketing by the Company.
2. Summary of Significant Accounting Policies
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Unaudited Interim Financial Information
The accompanying balance sheet as of June 30, 2003, and the statements of operations and cash flows for the six months ended June 30, 2002 and 2003 and the period from June 16, 2000 (inception) through June 30, 2003, and the statement of changes in stockholders’ deficit for the six months ended June 30, 2003 are unaudited, but in the opinion of management, include all adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of results for these interim periods. Certain information and footnote disclosures normally included in financial statements prepared in accordance with generally accepted accounting principles in the United States of America have been omitted, although the Company believes that the disclosures included are adequate to make the information presented not misleading. The results of
F-39
Table of Contents
Notes to the Financial Statements — (Continued)
operations for the six months ended June 30, 2003 are not necessarily indicative of the results expected for the entire year. The data disclosed in the notes to these financial statements for these periods is unaudited.
Concentration of Credit Risk
Financial instruments which potentially expose the Company to concentration of credit risk consist primarily of cash and cash equivalents. The Company places its cash in highly rated financial institutions.
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original or remaining maturity of three months or less at the date of purchase to be cash equivalents. Cash equivalents consist primarily of money market instruments at December 31, 2001 and 2002 and June 30, 2003 (unaudited).
Fair Value of Financial Instruments
The carrying amounts of the Company’s financial instruments, which include cash equivalents, accounts payable and accrued expenses, approximate their fair values at December 31, 2001 and 2002 and June 30, 2003 (unaudited).
Revenue Recognition
The Company recognizes revenue in accordance with the Securities and Exchange Commission’s Staff Accounting Bulletin No. 104,Revenue Recognition in Financial Statements(“SAB 104”). Revenue is recognized when the following criteria have been met:
1. Persuasive evidence of an arrangement exists;
2. Delivery has occurred and risk of loss has passed;
3. The seller’s price to the buyer is fixed or determinable; and
4. Collectibility is reasonably assured.
The Company’s revenue to date has been generated primarily through the licensing of the Company’s patents to certain collaborators. The Company recognized revenue related to nonrefundable license fees over the related performance period or at the time the Company has no remaining obligations.
Property and Equipment and Long-Lived Assets
Property and equipment is recorded at cost and depreciated using the straight-line method over the estimated useful life of each asset. Expenditures for repairs and maintenance are charged to expense as incurred. On disposal, the related assets and accumulated depreciation are eliminated from the accounts and any resulting gain or loss is included in income. Leasehold improvements are amortized over the shorter of the useful life of the improvement or the remaining term of the lease.
The Company evaluates the recoverability of its property and equipment and other long-lived assets when circumstances indicate that an event of impairment may have occurred. Impairment is measured based on the difference between the carrying value of the related assets or businesses and the discounted future cash flows of such assets or businesses. No impairment was required to be recognized for any of the years ended December 31, 2002 and 2003, or six months ended June 30, 2003 (unaudited).
F-40
Table of Contents
Notes to the Financial Statements — (Continued)
Income Taxes
The Company uses the liability method of accounting for income taxes. Under the liability method, deferred tax assets and liabilities reflect the impact of temporary differences between amounts of assets and liabilities for financial reporting purposes and such amounts as measured under enacted tax laws. A valuation allowance is required to offset any net deferred tax assets if, based upon the available evidence, it is more likely than not that some or all of the deferred tax asset will not be realized.
Research and Development Costs
Research and development costs are expensed as incurred. Included in research and development costs are wages, benefits and other operating costs such as facilities, supplies and overhead directly related to the Company’s research and development department as well as costs to acquire technology licenses.
Comprehensive Loss
Comprehensive income is defined as the change in equity of a business enterprise during a period from transactions and other events and circumstances from nonowner sources. During the years ended December 31, 2001 and 2002 and the six months ended June 30, 2002 and 2003 (unaudited), reported net loss was equal to comprehensive loss.
| 3. | Property and Equipment |
| Estimated | December 31, | ||||||||||||||||
| Useful Life | June 30, | ||||||||||||||||
| Years | 2001 | 2002 | 2003 | ||||||||||||||
| € | € | € | |||||||||||||||
| (Unaudited) | |||||||||||||||||
| Laboratory equipment | 5 to 8 | 351,092 | 709,045 | 910,589 | |||||||||||||
| Other equipment | 4 | 41,875 | 74,394 | 92,075 | |||||||||||||
| 392,967 | 783,439 | 1,002,664 | |||||||||||||||
| Less: accumulated depreciation | |||||||||||||||||
| Laboratory equipment | (70,131 | ) | (181,592 | ) | (262,130 | ) | |||||||||||
| Other equipment | (12,152 | ) | (28,088 | ) | (38,373 | ) | |||||||||||
| 310,684 | 573,759 | 702,161 | |||||||||||||||
Depreciation expense was€ 68,043,€ 127,397,€ 209,680,€ 56,325, and€ 90,823 for the year ended December 31, 2001 and 2002, for the period from June 16, 2000 (inception) to December 31, 2002 and for the six months ended June 30, 2002 and 2003 (unaudited).
F-41
Table of Contents
Notes to the Financial Statements — (Continued)
| 4. | Accrued Liabilities and Other Current Liabilities |
Accrued expenses and other current liabilities comprise the following:
| December 31, | ||||||||||||
| June 30, | ||||||||||||
| 2001 | 2002 | 2003 | ||||||||||
| € | € | € | ||||||||||
| (Unaudited) | ||||||||||||
| Vacation | 21,986 | 35,500 | 45,500 | |||||||||
| Legal support and consultancy | 24,644 | 46,000 | 179,171 | |||||||||
| Supervisory board services | 14,316 | 5,000 | — | |||||||||
| Other | 18,049 | 15,600 | 2,562 | |||||||||
| Total accrued expenses | 78,995 | 102,100 | 227,233 | |||||||||
| Employment tax | 19,829 | 14,280 | 23,316 | |||||||||
| Social security | 19,115 | 17,974 | 28,296 | |||||||||
| Other | 2,032 | 5,953 | 17,757 | |||||||||
| Total other current liabilities | 40,976 | 38,207 | 69,369 | |||||||||
| 5. | Deferred Equipment Grant |
In May 2000, the Company received a grant totaling€515,791 from the High-Tech-Offensive Zukunft Bayern-HTO Program (“HTO”) for the purchase of equipment to be used in the Company’s operations and to fund three Company scientists. The grant was remitted in three installments of€299,926 in 2000,€155,893 in 2001, and€59,972 in 2002. The Company recorded amounts received as deferred grant income and is recognizing the deferred amounts as an offset to the related depreciation of the fixed assets over the useful life of the related assets and as an offset to the salary expense of the related scientists. As a condition to the grant related to the purchase of equipment, for a period of 5 years after the grant date, if the Company sells the equipment purchased with the proceeds from the grant and realizes proceeds in excess of€400,000, the Company is obligated to refund the gain to the HTO.
During the year ended December 31, 2000, 2001, 2002, and the six months ended June 30, 2002 (unaudited) and 2003 (unaudited), the Company recorded€22,055,€195,877,€45,989,€4,492, and€37,447, respectively, against the related fixed asset depreciation expense and salary costs. For the period from June 16, 2000 (inception) through December 31, 2003 and for the period from June 16, 2000 (inception) through June 30, 2003 (unaudited), the Company recorded€273,921 and€301,368 as an offset against the related costs. The Company has classified the deferred grant amounts of€177,877 and€140,440 as long-term as of December 31, 2002 and June 30, 2003 (unaudited), as these amounts will be recognized beyond one year.
F-42
Table of Contents
Notes to the Financial Statements — (Continued)
| 6. | Notes Payable |
As of December 31, 2001, 2002 and June 30, 2003, the Company had approximately the following notes payable outstanding:
| December 31, | |||||||||||||
| June 30, | |||||||||||||
| 2001 | 2002 | 2003 | |||||||||||
| € | € | € | |||||||||||
| (Unaudited) | |||||||||||||
| Note Payable from Stockholder | — | 2,000,000 | 3,000,000 | ||||||||||
| Note Payable to GUB | 464,000 | — | — | ||||||||||
| Note Payable to TGB | 466,000 | 1,227,000 | 1,227,000 | ||||||||||
| Note Payable to TBFB | 383,000 | 1,023,000 | 1,023,000 | ||||||||||
| Total | 1,313,000 | 4,250,000 | 5,250,000 | ||||||||||
Note Payable from Stockholder
On October 11, 2002, the Company entered into an agreement with Abingworth Bioventures for a note payable of€ 2,000,000. The note was due for repayment 12 months after the date of the agreement in full, including all outstanding accrued interest. The loan accrued interest at the rate of 8 percent per annum. In April 2003, the loan amount available to the Company was increased to€ 4,000,000, and the Company drew down an additional€ 1,000,000 during the six months ended June 2003. As a result of this additional borrowing, the Company committed to issuing 5,000 shares of common stock to Abingworth Bioventures. The fair value of these shares is being amortized over the life of the debt as additional interest expense. Following the share exchange agreement with Alnylam, the loan and accrued interest of€ 4,225,844 was assigned to Alnylam, and subsequently paid in full. Additionally in July 2003, the 5,000 shares of Ribopharma owed to Abingworth Bioventures were converted into 94,044 shares of Alnylam common stock.
Note Payable to GUB
On September 6, 2000, the Company entered into a note payable with GUB Glasauer Unternehmensbeteiligung-KG (“GUB”) totalling approximately€ 1,208,000. The Company received approximately€ 464,000 in 2000, approximately€ 550,000 in January 2002 and approximately€ 194,000 in October 2002 for a total note payable of approximately€ 1,208,000 as of December 31, 2002. There was no interest due on the note payable, however, GUB was entitled to profit sharing. Furthermore, either GUB or the Company shareholders could require the note payable to be converted into ordinary shares of the Company. If certain milestones had been reached, 10,230 shares would be due, if they had not then 20,200. In October 2002, the agreement was amended, and the profit sharing agreement was ended, and GUB contributed an additional€ 13,000 and received 13,000 shares. As the estimated value of the common shares that were issued approximated the total amount provided to the Company under the note payable, the Company converted the note payable of approximately€ 1,208,000 into additional paid in capital upon issuance of the 13,000 shares of common stock.
Note Payable to TBG
Effective November 23, 2000 and December 18, 2000, the Company entered into a note payable with an aggregate amount available to the Company of approximately€ 1,277,000 with Technologie-Beteiligungs-Gesellschaft mbH der Deutschen Ausgleichsbank, Bonn (“TBG”), of which approximately€ 466,000 was
F-43
Table of Contents
Notes to the Financial Statements — (Continued)
provided to the Company in 2001 and approximately€ 761,000 was provided in 2002. As of December 31, 2003, there was€ 50,000 available to the Company under the agreement.
Borrowings under the note payable bear interest at the rate of 7 percent per annum and are due in December 2010. In addition, TBG is entitled to participate in a profit share of 15 percent of any profits of the Company. These profit share amounts will be paid out in the event that certain profit levels are met. The note payable to TBG also receives a final interest payment of 30 percent of the principal amount paid out together with an additional interest payment of 6 percent of the principal amount paid out for every year after the fifth year of the signing of the agreement. The final interest payment is being accrued over the term of the note as additional interest expense. These payments are due either on maturity or on early repayment. Any profit share received is deducted from the final payment. As of December 31, 2002,€ 1,227,000 was outstanding under this note payable to TBG. As of December 31, 2001, 2002 and June 30, 2003 (unaudited), there was€ 7,854,€ 38,298, and€ 184,050 of accrued interest recorded related to the final payment on the note payable. In August 2003, the outstanding amount of€ 1,227,000 and the final amounts due of accrued interest of€ 184,050 was repaid to TBG.
Note Payable to TBFB
Effective January 17 and January 22, 2001, the Company entered into a note payable with an aggregate amount available to the Company of approximately€ 1,023,000 with the Technologie Beteiligungsfonds Bayern GmbH & Co. KG (“TBFB”) of which approximately€ 383,000 was provided to the Company in 2001 and approximately€ 640,000 was provided in 2002.
Borrowings under the note payable bear interest at the rate of 8 percent per annum and are due in December 2008. In addition, TBFB is entitled to participate in a profit share of 15 percent of any profits of the Company. These profit share amounts will be paid out in the event that certain profit levels are met. The note payable to TBFB also receives a final interest payment of 35 percent of the principal amount paid out together with an additional interest payment of 9 percent of the principal amount paid out for every year after the fifth year of the signing of the agreement. The final interest payment is being accrued over the term of the note as additional interest expense. These payments are due either on maturity or on early repayment. Any profit share received is deducted from the final payment. As of December 31, 2002, approximately€ 1.023.000 was outstanding under this note payable to TBFB. As of December 31, 2001, 2002 and June 30, 2003 (unaudited), there was€11,671,€42,841, and€178,952 of accrued interest recorded related to the final payment on the note payable. In August 2003, the outstanding amount of€ 1,023,000 and the final amounts due of accrued interest of€178,952 was repaid to TBFB.
7. Common Stock
As of December 31, 2002, the Company has 68,560 shares of common stock authorized and 68,560 shares issued and outstanding. During the year ended December 31, 2002, the Company converted the note payable from GUB into 13,000 shares of common stock (Note 6).
8. Income Taxes
As a result of net operating losses there has been no income taxes payable during the year ended December 31, 2001 and 2002.
Deferred income taxes reflect the tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting and income tax purposes. A valuation allowance is established
F-44
Table of Contents
Notes to the Financial Statements — (Continued)
when uncertainty exists as to whether all or a portion of the net deferred tax assets will be realized. Components of the net deferred tax asset as of December 31, 2001 and 2002, are approximately as follows:
| December 31, | ||||||||
| 2001 | 2002 | |||||||
| € | € | |||||||
| Net operating loss carryforwards | 7,000 | 558,000 | ||||||
| Statutory to US GAAP differences | 108,000 | 154,000 | ||||||
| 115,000 | 712,000 | |||||||
| Deferred tax asset valuation allowance | (115,000 | ) | (712,000 | ) | ||||
| Net deferred tax assets | — | — | ||||||
As required by SFAS No. 109,Accounting for Income Taxes, management of the Company has evaluated the positive and negative evidence bearing upon the realizability of its deferred tax assets. Management has concluded, in accordance with the applicable accounting standards, that it is more likely than not that the Company may not realize the benefit of its deferred tax assets. Accordingly, the deferred tax assets have been fully reserved. Management reevaluates the positive and negative evidence on an annual basis.
At December 31, 2001 and 2002, the Company had net operating loss carryforwards for corporate income tax purposes of approximately€ 27,000 and€ 1,849,000, respectively, and for trade income tax purposes of approximately none and€ 385,000, respectively.
Under German tax law, tax losses can be carried forward indefinitely. However, ownership changes of greater than 50% within 5 years of the loss periods may result in the disallowance of tax losses.
9. Commitments and Contingencies
Operating Leases
Total rent expense under operating leases for the years ended December 31, 2001, 2002, for the period from June 16, 2000 (inception) to December 31, 2002 and for the six months ended June 30, 2002 and 2003 (unaudited) was approximately€ 20,000 ,€ 80,000,€ 100,000,€ 40,000, and€ 92,000, respectively.
Effective February 1, 2002, a rental agreement was entered into with Anterra Projektentwicklungs-GmbH & Co. Objekt Spinnerei Kulmbach KG for offices in Kulmbach, Germany. This rental agreement runs for a fixed period which expires on June 30, 2008. However, the Company may exercise its option to extend the agreement.
The future minimum annual rental commitments under operating leases as of December 31, 2002 are as follows:
| Year Ending December 31, | Operating Leases | ||||
| € | |||||
| 2003 | 286,000 | ||||
| 2004 | 365,000 | ||||
| 2005 | 144,000 | ||||
| 2006 | 137,000 | ||||
| 2007 and thereafter | 197,000 | ||||
| Total | 1,129,000 | ||||
F-45
Table of Contents
Notes to the Financial Statements — (Continued)
10. Significant Agreements
Licence and Cooperation Agreement with Merck KGaA, Darmstadt
In March and April 2002, the Company entered into a licence and collaboration agreement with Merck KGaA, Darmstadt. The licence allows Merck KGaA to use the Company’s RNAi technology for pre-clinical validation of potential therapeutic molecular targets. The licence allows Merck KGaA to use the results of the target validating. Merck KGaA is to provide the Company with a number of gene sequences. As consideration for the agreement, Merck KGaA is to make a one-time payment of€ 10,000 and an annual licence fee of€ 35,000. If a patent is granted by either the European or U.S. patent office, the annual licence fee is increased to€ 70,000.
F-46
Table of Contents
PRO FORMA FINANCIAL DATA
The following unaudited pro forma combined financial statements give effect to the acquisition of Ribopharma AG (“Ribopharma”) by Alnylam Pharmaceuticals, Inc. (“Alnylam”) that occurred on July 31, 2003 in a transaction which was accounted for as an asset purchase. The unaudited pro forma combined statements of operations combine the historical consolidated statements of operations of Alnylam for the year ended December 31, 2003 included elsewhere in this prospectus, and Ribopharma for the seven months ended July 31, 2003, giving effect to the merger as if it occurred on January 1, 2003, reflecting only pro forma adjustments expected to have a continuing impact on the combined results.
These unaudited pro forma combined financial statements are for informational purposes only. They do not purport to indicate the results that would have actually been obtained had the merger been completed on the assumed date or for the periods presented, or which may be realized in the future. To produce the pro forma financial information, Alnylam allocated the purchase price using its best estimates of fair value, based in part on a valuation that was performed. The unaudited pro forma combined financial statements should be read in conjunction with Management’s Discussion and Analysis of Financial Condition and Results of Operations and the historical consolidated financial statements, including the related notes, of Alnylam and Ribopharma covering these periods, included elsewhere in this prospectus.
F-47
Table of Contents
ALNYLAM PHARMACEUTICALS
Unaudited Pro Forma Combined Statements of Operations
| Year Ended | Seven Months | ||||||||||||||||||||
| December 31, | Ended July 31, | Pro | |||||||||||||||||||
| 2003 | 2003 | Pro Forma | Note | Forma | |||||||||||||||||
| Alnylam | Ribopharma | Adjustments | Reference | Combined | |||||||||||||||||
| ($ in thousands, except per share amounts) | |||||||||||||||||||||
| Revenue from research collaborators | $ | 176 | $ | 47 | $ | 223 | |||||||||||||||
| Costs and expenses | |||||||||||||||||||||
| Research and development | 13,097 | 1,795 | 155 | (A) | |||||||||||||||||
| 197 | (B) | 15,244 | |||||||||||||||||||
| General and administrative | 7,527 | 1,027 | 17 | (A) | |||||||||||||||||
| (484 | ) | (C) | 8,087 | ||||||||||||||||||
| Purchased in-process research and development | 4,609 | — | (4,609 | ) | (D) | — | |||||||||||||||
| Total operating costs and expenses | 25,233 | 2,822 | 23,331 | ||||||||||||||||||
| Loss from operations | (25,057 | ) | (2,775 | ) | (23,108 | ) | |||||||||||||||
| Interest income | 179 | 20 | (11 | ) | (E) | 188 | |||||||||||||||
| Interest expense | (127 | ) | (498 | ) | (625 | ) | |||||||||||||||
| Other expense | (28 | ) | — | (28 | ) | ||||||||||||||||
| Net loss | (25,033 | ) | (3,253 | ) | (23,573 | ) | |||||||||||||||
| Accretion of redeemable convertible preferred stock | (2,906 | ) | — | (2,906 | ) | ||||||||||||||||
| Net loss attributable to common stockholders | $ | (27,939 | ) | $ | (3,253 | ) | $ | (26,479 | ) | ||||||||||||
| Net loss per share | |||||||||||||||||||||
| Net loss per common share (basic and diluted) | $ | (15.60 | ) | $ | (9.84 | ) | |||||||||||||||
| Weighted average shares used to compute basic and diluted net loss per common share | 1,791,063 | 899,819 | (F) | 2,690,882 | |||||||||||||||||
The accompanying notes are an integral part of these unaudited pro forma combined statements of operations.
F-48
Table of Contents
Notes to Unaudited Pro Forma Combined Financial Statements of Operations
Note 1. Basis of Presentation and Pro Forma Information
The unaudited pro forma combined statements of operations data contained herein describes the pro forma effect of the acquisition of Ribopharma by Alnylam that took place on July 31, 2003, as if it occurred on January 1, 2003. The acquisition was completed as of July 31, 2003 and the results of operations of Ribopharma from August 1, 2003 through December 31, 2003 are included in Alnylam’s historical results of operations. Ribopharma’s historical results represent its results of operations for the seven months ended July 31, 2003, prior to its acquisition by Alnylam.
Note 2. Description of the Business
Alnylam commenced operations on June 14, 2002 as an early stage biopharmaceutical company seeking to develop and commercialize new drugs that work through a recently discovered system in cells known as RNA interference, or RNAi. Alnylam is focused on discovering, developing and commercializing RNAi therapeutics by establishing strategic alliances with leading pharmaceutical companies, establishing and maintaining a strong intellectual property position in the RNAi field and generating revenues through licensing agreements. Since its inception, the Company has devoted substantially all of its efforts to business planning, research and development, recruiting management and technical staff, and raising capital.
Note 3. Description of Transaction
On July 31, 2003, Alnylam acquired the outstanding shares of Ribopharma and accounted for the acquisition as an asset purchase. Under the terms of the agreement, shareholders of Ribopharma AG exchanged all their outstanding shares for $1,500 in cash and 1,549,216 shares of common stock of Alnylam. Ribopharma is a development stage enterprise that is performing research and development associated with a new pharmaceutical active agent category siRNA. The consideration consisted of $1,500 in cash and 1,549,216 shares of common stock. Based on a valuation performed of Ribopharma AG and the intangible assets acquired, the purchase price is estimated at $3,866 and is comprised of the following:
| Cash paid | $ | 1,500 | ||
| Fair value of common stock issued | 1,947 | |||
| Acquisition costs | 419 | |||
| $ | 3,866 | |||
The purchase price has been allocated to the acquired tangible and intangible assets and liabilities based on their estimated fair values as of July 31, 2003 as follows:
| Cash | $ | 1,798 | ||
| Other current assets | 41 | |||
| Fixed assets | 1,733 | |||
| Intangible assets | 8,684 | |||
| Accounts payable and accrued expenses assumed | (1,300 | ) | ||
| Notes payable assumed | (7,090 | ) | ||
| $ | 3,866 | |||
F-49
Table of Contents
The appraised value of intangible assets acquired was below the total fair value of intangible assets acquired and would generally result in the recognition of goodwill. However, since Ribopharma AG is a development stage company and not considered a “business” as defined by the applicable accounting rules, this residual value was allocated proportionately to the long-lived assets acquired as follows:
| Initial | Recorded | |||||||
| Value | Fair Value | |||||||
| Purchased in-process research and development | $ | 2,155 | $ | 4,609 | ||||
| Core technology | 1,700 | 3,638 | ||||||
| Workforce | 200 | 437 | ||||||
| Fixed assets | 552 | 1,733 | ||||||
| $ | 4,607 | $ | 10,417 | |||||
In connection with the Company’s acquisition of Ribopharma AG, the Company acquired two systemic RNAi programs related to the development of drugs targeting cancers such as malignant melanoma and pancreatic carcinoma. The Company expensed $4,609 of purchased in-process research and development associated with these programs. Management’s plans contemplate the Company would conduct the first phase of clinical trials and then out-license the programs to a partner. Upon out-licensing, we expect the partner to bear all development costs and control clinical development. We expect to earn payments upon the attainment of clinical milestones by our partner and royalties on product sales. Since our partner will control the clinical development, we will be unable to influence the timing of the achievement of the milestones, if at all, or the estimated year of the product launch, if at all. Our valuation assumed a development period of approximately 10 years, with milestones being earned during that period, which management believes is a typical horizon to bring a therapeutic drug to market. Actual results will differ from these estimates due to the uncertainties surrounding drug development.
Management assumes responsibility for determining the in-process research and development valuation. The fair value assigned to purchase in-process research and development was estimated by discounting, to present value, the probability-adjusted net cash flows expected to result once the technology has reached technological feasibility. A discount rate of 32 percent was applied to estimate the present value of the cash flows and is consistent with the overall risks of developing these projects. As of December 31, 2003, the technological feasibility of the projects had not been reached and management believes the assumptions included in the valuation analysis continue to be valid. In the allocation of the purchase price, the concept of alternative future use was considered. The projects under development have no current alternative future uses for the underlying technology in the event the projects are unsuccessful.
Core technology and workforce are being amortized over their estimated useful lives of ten years and four years, respectively. The step up in the fixed assets is being amortized over four years, the remaining estimated useful life of these assets.
Note 4. Pro Forma Adjustments
| (A) | To record the amortization of the step-up of Ribopharma’s fixed assets book value to recorded fair value to reflect the full year of 2003. The step up in the fixed assets is being amortized over four years, the remaining estimated useful life of these assets. |
| (B) | To record the amortization of acquired identifiable intangible assets including core technology and workforce to reflect the full year of 2003. Core technology and workforce are being amortized over their estimated useful lives of ten years and four years, respectively. | |
| (C) | To reverse Ribopharma’s acquisition related expenses. This was amount was eliminated as it reflects a non-recurring charge directly resulting from the acquisition. |
F-50
Table of Contents
| (D) | To eliminate the charge for purchased in-process research and development that was recorded in July 2003 in connection with the acquisition. This was amount was eliminated as it reflects a non-recurring charge directly resulting from the acquisition. |
| (E) | To reduce investment income for the period from January 1, 2003 through July 31, 2003 to reflect the payment of $1,900 of cash consideration and acquisition related expenses paid in the acquisition at a rate of return of 1.0% per annum. |
| (F) | The unaudited pro forma basic and diluted net loss per share is based on the weighted average number of shares of Alnylam common stock outstanding to reflect the issuance of 1,549,216 shares of common stock issued in the acquisition as if the shares were issued on January 1, 2003. Included in the Alnylam basic and diluted weighted average number of shares outstanding for the year ended December 31, 2003, is 649,397 shares, which represents the 1,549,216 shares outstanding from the period July 31, 2003 through December 31, 2003. Accordingly, an additional 899,819 shares are included in the pro forma adjusted weighted average basic and diluted net loss per share to reflect the shares outstanding from January 1, 2003 through July 31, 2003. Options, redeemable convertible preferred stock and warrants outstanding have not been included in the computation of pro forma diluted net loss per share because their effect would be antidilutive. |
F-51
Table of Contents

Common Stock
Prospectus
Banc of America Securities LLC
Until , 2004, all dealers that buy, sell or trade our common stock may be required to deliver a prospectus, regardless of whether they are participating in this offering. This is in addition to the dealers’ obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
Table of Contents
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
The following table indicates the expenses to be incurred in connection with the offering described in this Registration Statement, other than underwriting discounts and commissions, all of which will be paid by Alnylam. All amounts are estimates, other than the SEC registration fee, the NASD filing fee and the NASDAQ National Market listing fee.
| SEC registration fee | $ | 10,928 | |||
| NASD Filing fee | 9,125 | ||||
| NASDAQ National Market listing fee | 100,000 | ||||
| Printing and engraving expenses | * | ||||
| Legal fees and expenses | * | ||||
| Accounting fees and expenses | * | ||||
| Blue Sky fees and expenses | * | ||||
| Transfer agent and registrar fees and expenses | * | ||||
| Miscellaneous | * | ||||
Total | $ | * | |||
| * | To be filed by amendment. |
Item 14. Indemnification of Directors and Officers.
Section 102 of the Delaware General Corporation Law allows a corporation to eliminate the personal liability of directors of a corporation to the corporation or its stockholders for monetary damages for a breach of fiduciary duty as a director, except where the director breached his duty of loyalty, failed to act in good faith, engaged in intentional misconduct or knowingly violated a law, authorized the payment of a dividend or approved a stock repurchase in violation of Delaware corporate law or obtained an improper personal benefit. Alnylam has included such a provision in its Certificate of Incorporation.
Section 145 of the General Corporation Law of Delaware provides that a corporation has the power to indemnify a director, officer, employee or agent of the corporation and certain other persons serving at the request of the corporation in related capacities against amounts paid and expenses incurred in connection with an action or proceeding to which he is or is threatened to be made a party by reason of such position, if such person shall have acted in good faith and in a manner he reasonably believed to be in or not opposed to the best interests of the corporation, and, in any criminal proceeding, if such person had no reasonable cause to believe his conduct was unlawful; provided that, in the case of actions brought by or in the right of the corporation, no indemnification shall be made with respect to any matter as to which such person shall have been adjudged to be liable to the corporation unless and only to the extent that the adjudicating court determines that such indemnification is proper under the circumstances.
Our Amended and Restated Certificate of Incorporation includes a provision that eliminates the personal liability of its directors for monetary damages for breach of fiduciary duty as a director, except for liability:
| • | for any breach of the director’s duty of loyalty to Alnylam or its stockholders; | |
| • | for acts or omissions not in good faith or that involve intentional misconduct or a knowing violation of law; | |
| • | under section 174 of the Delaware General Corporation Law regarding unlawful dividends and stock purchases; or | |
| • | for any transaction from which the director derived an improper personal benefit. |
II-1
Table of Contents
These provisions are permitted under Delaware law. Our Amended and Restated Certificate of Incorporation provides that:
| • | we must indemnify our directors and officers to the fullest extent permitted by Delaware law; | |
| • | we may indemnify our other employees and agents to the same extent that we indemnified our officers and directors, unless otherwise determined by our Board of Directors; and | |
| • | we must advance expenses, as incurred, to our directors and executive officers in connection with a legal proceeding to the fullest extent permitted by Delaware law. |
The indemnification provisions contained in our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws are not exclusive of any other rights to which a person may be entitled by law, agreement, vote of stockholders or disinterested directors or otherwise.
In addition, we maintain insurance on behalf of our directors and executive officers insuring them against any liability asserted against them in their capacities as directors or officers or arising out of such status.
Item 15. Recent Sales of Unregistered Securities.
Set forth below is information regarding shares of common stock and preferred stock issued, and options and warrants granted, by the Registrant within the past three years. Also included is the consideration, if any, received by the Registrant for such shares, options and warrants and information relating to the section of the Securities Act, or rule of the Securities and Exchange Commission under which exemption from registration was claimed.
| (1) | In July 2003, in connection with its acquisition of Alnylam Europe, AG, the Registrant issued an aggregate of 1,549,216 shares of its common stock to the former stockholders of Ribopharma AG in exchange for the outstanding shares of common stock of Alnylam Europe, AG and issued 94,044 shares of its common stock to former holders of Alnylam Europe, AG debt upon settlement of Alnylam Europe, AG accrued interest. | |
| (2) | In July 2003, the Registrant issued and sold an aggregate of 3,000,010 shares of its Series A Preferred Stock to a group of eight investors at a price per share of $1.00. Upon the closing of this offering, these shares will convert into 3,000,010 shares of common stock. These investors consisted of Polaris Venture Partners III, Polaris Entrepreneurs Fund III, Polaris Founders Fund III, Abingworth BioVentures III A, Abingworth BioVentures III B, Abingworth BioVentures III C, Abingworth BioVentures Executives III and CHP II, L.P. | |
| (3) | In July 2003, the Registrant issued and sold an aggregate of 16,561,845 shares of its Series B Preferred Stock to a group of 25 investors at a price per share of $2.50. Upon the closing of this offering, these shares will convert into 16,561,845 shares of common stock. These investors consisted of Polaris Venture Partners III, Polaris Entrepreneurs Fund III, Polaris Founders Fund III, Atlas Venture Fund V, Atlas Venture Parallel Fund V-A, Atlas Venture Parallel Fund V-B, Atlas Venture Entrepreneurs Fund V, Atlas Venture Fund VI, Atlas Venture Entrepreneur Fund VI, Atlas Venture Fund VI GmbH, Arch Venture Fund V, Arch Entrepreneurs Fund L.P., Arch V Entrepreneurs Fund, L.P., Abingworth BioVentures III A, Abingworth BioVentures III B, Abingworth BioVentures III C, Abingworth BioVentures Executives III, Cambridge Science Equities, CHP II, L.P., Phillip A. Sharp, Ph.D., Paul R. Schimmel, Ph.D. and John G. Conley. | |
| (4) | In July 2003, the Registrant issued an aggregate of 881,845 shares of Series B Preferred Stock to a group of three institutions in connection with a certain license agreement. Upon the closing of this offering, these shares will convert into 881,845 shares of common stock. These institutions consisted of the Massachusetts Institute of Technology, the Whitehead Institute for Biomedical Research and Max Planck-Gesellschaft zur Förderung der Wissenschaften e.V. | |
| (5) | In July 2003, the Registrant assumed the obligations of Alnylam U.S., Inc. under a warrant issued to Silicon Valley Bank to purchase 25,000 shares of Series B Preferred Stock at an exercise price | |
II-2
Table of Contents
| of $2.50 per share. Upon the closing of this offering, this warrant will become exercisable for 25,000 shares of common stock. |
| (6) | In September 2003, the Registrant issued and sold an aggregate of 1,000,000 shares of its Series C Preferred Stock to Merck & Co., Inc. at a price per share of $5.00. Upon the closing of this offering, these shares will convert into 1,000,000 shares of common stock. | |
| (7) | In October 2003, the Registrant issued and sold an aggregate of 504,825 shares of its Series C Preferred Stock to a group of 17 investors at a price per share of $5.00. Upon the closing of this offering, these shares will convert into 504,825 shares of common stock. These investors included Polaris Venture Partners III, Polaris Entrepreneurs Fund III, Polaris Founders Fund III, Atlas Venture Fund V, Atlas Venture Parallel Fund V-A, Atlas Venture Parallel Fund V-B, Atlas Venture Entrepreneurs Fund V, Atlas Venture Fund VI, Atlas Venture Entrepreneur Fund VI, Atlas Venture Fund VI GmbH, Arch Venture Fund V, Arch V Entrepreneurs Fund, Abingworth BioVentures III A, Abingworth BioVentures III B, Abingworth BioVentures III C and Abingworth BioVentures Executives III. | |
| (8) | On March 11, 2004, the Registrant issued and sold 1,666,667 shares of Series D Preferred Stock to Isis Pharmaceuticals, Inc. at a price per share of $6.00. Upon the closing of this offering, these shares will convert into 1,666,667 shares of common stock. | |
| (9) | On March 30, 2004, the Registrant issued and sold warrants to purchase an aggregate of 100,000 shares of Series C Preferred Stock at an exercise price of $5.00 per share to Lighthouse Capital Partners V, L.P. and Lighthouse Capital Partners IV, L.P. Upon the closing of this offering, these shares will convert into 100,000 shares of common stock. | |
| (10) | The Registrant has granted stock options under its stock option plans for an aggregate of 3,691,618 shares of Common Stock (net of exercises, expirations and cancellations) at exercise prices of $0.10 to $0.50 per share. Options to purchase 369,281 shares of Common Stock have been exercised for an aggregate purchase price of $132,987. |
No underwriters were involved in the foregoing sales of securities. The securities described in paragraph 1 of Item 15 were issued to a combination of foreign and U.S. investors in reliance upon exemptions from the registration provisions of the Securities Act set forth in Section 3(b) and Regulation S. The securities described in paragraphs 2 through 9 of Item 15 were issued to a combination of foreign and U.S. investors in reliance upon exemptions from the registration provisions of the Securities Act set forth in Section 4(2) or Regulation S thereof relative to sales by an issuer not involving any public offering, to the extent an exemption from such registration was required. All purchasers of shares of our convertible preferred stock described above represented to us in connection with their purchase that they were accredited investors and were acquiring the shares for investment and not distribution, that they could bear the risks of the investment and could hold the securities for an indefinite period of time. Such purchasers received written disclosures that the securities had not been registered under the Securities Act and that any resale must be made pursuant to a registration or an available exemption from such registration.
The issuance of stock options and the common stock issuable upon the exercise of such options as described in paragraph 10 of Item 15 were issued pursuant to written compensatory plans or arrangements with our employees, directors and consultants, in reliance on the exemption provided by Rule 701 promulgated under the Securities Act.
All of the foregoing securities are deemed restricted securities for purposes of the Securities Act. All certificates representing the issued shares of common stock described in this Item 15 included appropriate legends setting forth that the securities had not been registered and the applicable restrictions on transfer.
II-3
Table of Contents
Item 16. Exhibits and Financial Statement Schedules.
(a) Exhibits
| Exhibit | ||||
| No. | Description | |||
| 1.1* | Underwriting Agreement | |||
| 3.1** | Certificate of Incorporation of the Registrant, as amended | |||
| 3.2** | Bylaws of the Registrant | |||
| 3.3 | Amended and Restated Certificate of Incorporation of the Registrant to be effective upon closing of the offering | |||
| 3.4 | Amended and Restated Bylaws of the Registrant to be effective upon closing of the offering | |||
| 4.1* | Specimen certificate evidencing shares of common stock | |||
| 5.1* | Opinion of Hale and Dorr LLP | |||
| 10.1 | 2002 Employee, Director and Consultant Stock Plan, as amended | |||
| 10.2 | 2003 Employee, Director and Consultant Stock Plan, as amended | |||
| 10.3 | 2004 Stock Incentive Plan | |||
| 10.4 | 2004 Employee Stock Purchase Plan | |||
| 10.5** | Registration Rights Agreement dated as of July 31, 2003 and amended as of October 9, 2003 and February 26, 2004 by and among the Registrant and the parties listed on Schedule A thereto | |||
| 10.6** | Investor Rights Agreement dated as of September 8, 2003 and amended on February 26, 2004 by and between the Registrant and Merck & Co., Inc. | |||
| 10.7** | Letter Agreement between the Registrant and John M. Maraganore, Ph.D. dated October 30, 2002 | |||
| 10.8** | Letter Agreement between the Registrant and Vincent J. Miles, Ph.D. dated June 16, 2003 | |||
| 10.9** | Letter Agreement between the Registrant and Thomas R. Ulich, M.D. dated June 15, 2003 | |||
| 10.10** | Letter Agreement between the Registrant and Barry E. Greene dated September 29, 2003 | |||
| 10.11 | Loan and Security Agreement by and between Lighthouse Capital Partners V, L.P. and the Registrant dated as of March 26, 2004, together with the Negative Pledge Agreement by and between Lighthouse Capital Partners V, L.P. and the Registrant dated as of March 26, 2004. | |||
| 10.12 | Warrants to Purchase Preferred Stock effective as of March 30, 2004 issued to Lighthouse Capital Partners V, L.P. and Lighthouse Capital Partners IV, L.P. | |||
| 10.13** | Warrant to Purchase Stock dated December 18, 2002 issued to Silicon Valley Bank | |||
| 10.14** | Lease, dated as of August 5, 2003, between the Registrant and ARE-770/784/790 Memorial Drive, LLC, as amended | |||
| 10.15** | Lease, dated as of September 26, 2003 by and between the Registrant and Three Hundred Third Street LLC | |||
| 10.16†* | * | License Agreement between Cancer Research Technology Limited and Alnylam U.S., Inc. dated July 18, 2003 | ||
| 10.17†* | * | License Agreement between the Carnegie Institution of Washington and Alnylam Europe, AG, effective March 1, 2002, as amended by letter agreements dated September 2, 2002 and October 28, 2003. | ||
| 10.18†* | * | License Agreement by and between the Cold Spring Harbor Laboratory and Alnylam U.S., Inc. dated December 30, 2003 | ||
| 10.19†* | * | Co-exclusive License Agreement between Garching Innovation GmbH and Alnylam U.S., Inc. dated December 20, 2002, as amended by Amendment dated July 8, 2003 | ||
| 10.20†* | * | Co-exclusive License Agreement between Garching Innovation GmbH and Alnylam Europe, AG dated July 30, 2003 | ||
| 10.21†* | * | Agreement between The Board of Trustees of the Leland Stanford Junior University and Alnylam U.S., Inc. effective as of September 17, 2003 | ||
| 10.22†* | * | Research Collaboration and License Agreement by and among Merck & Co., Inc., Alnylam U.S., Inc. and Registrant dated September 8, 2003 | ||
| 10.23†* | * | Sponsored Research Agreement among Mayo Foundation for Medical Education and Research, Mayo Clinic Jacksonville and Alnylam Pharmaceuticals, Inc. effective as of October 1, 2003 | ||
II-4
Table of Contents
| Exhibit | ||||
| No. | Description | |||
| 10.24† | Strategic Collaboration and License Agreement effective as of March 11, 2004 between Isis Pharmaceuticals, Inc. and the Registrant | |||
| 10.25 | Investor Rights Agreement entered into as of March 11, 2004 by and between the Registrant and Isis Pharmaceuticals, Inc. | |||
| 21.1 | Subsidiaries of the Registrant | |||
| 23.1 | Consent of PricewaterhouseCoopers LLP, Independent Auditors | |||
| 23.2 | Consent of PricewaterhouseCoopers Gesellschaft mit beschränkter Haftung Wirtschaftsprüfungsgesellschaft, Independent Auditors | |||
| 23.3* | Consent of Hale and Dorr LLP (included in Exhibit 5.1) | |||
| 24.1** | Power of Attorney | |||
| * | To be filed by amendment. |
| ** | Previously filed. |
| † | Confidential treatment requested as to certain portions, which portions have been filed separately with the Securities and Exchange Commission. |
(b) Financial Statement Schedules.
None
| Item 17. | Undertakings. |
The undersigned registrant hereby undertakes to provide to the underwriters at the closing specified in the underwriting agreement certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser.
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers, and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable. In the event that a claim for indemnification by the registrant against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer, or controlling person of the registrant in the successful defense of any action, suit, or proceeding) is asserted by such director, officer, or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
The undersigned registrant hereby undertakes that:
| (1) For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b) (1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. | |
| (2) For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
II-5
Table of Contents
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Cambridge, Commonwealth of Massachusetts on this 6th day of April, 2004.
| ALNYLAM PHARMACEUTICALS, INC. |
| By: | /s/ JOHN M. MARAGANORE |
| John M. Maraganore, Ph.D. | |
| President and Chief Executive Officer |
| Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated. |
| Signature | Title | Date | ||||
| /s/ JOHN M. MARAGANORE JOHN M. MARAGANORE, PH.D. | President, Chief Executive Officer and Director (Principal Executive Officer) | April 6, 2004 | ||||
| /s/ BARRY E. GREENE BARRY E. GREENE | Chief Operating Officer and Treasurer (Principal Financial and Accounting Officer) | April 6, 2004 | ||||
| * PETER BARRETT, PH.D. | Director | April 6, 2004 | ||||
| * JOHN BERRIMAN | Director | April 6, 2004 | ||||
| * JOHN CLARKE | Director | April 6, 2004 | ||||
| * PAUL SCHIMMEL, PH.D. | Director | April 6, 2004 | ||||
| * PHILLIP A. SHARP, PH.D. | Director | April 6, 2004 | ||||
| * KEVIN STARR | Director | April 6, 2004 | ||||
| * CHRISTOPH H. WESTPHAL, M.D., PH.D. | Director | April 6, 2004 | ||||
| *By: | /s/ JOHN M. MARAGANORE JOHN M. MARAGANORE, PH.D. Attorney-in-fact | |||||
II-6
Table of Contents
EXHIBIT INDEX
| Exhibit | ||||
| No. | Description | |||
| 1.1* | Underwriting Agreement | |||
| 3.1** | Certificate of Incorporation of the Registrant, as amended | |||
| 3.2** | Bylaws of the Registrant | |||
| 3.3 | Amended and Restated Certificate of Incorporation of the Registrant to be effective upon closing of the offering | |||
| 3.4 | Amended and Restated Bylaws of the Registrant to be effective upon closing of the offering | |||
| 4.1* | Specimen certificate evidencing shares of common stock | |||
| 5.1* | Opinion of Hale and Dorr LLP | |||
| 10.1 | 2002 Employee, Director and Consultant Stock Plan, as amended | |||
| 10.2 | 2003 Employee, Director and Consultant Stock Plan, as amended | |||
| 10.3 | 2004 Stock Incentive Plan | |||
| 10.4 | 2004 Employee Stock Purchase Plan | |||
| 10.5** | Registration Rights Agreement dated as of July 31, 2003 and amended as of October 9, 2003 and February 26, 2004 by and among the Registrant and the parties listed on Schedule A thereto | |||
| 10.6** | Investor Rights Agreement dated as of September 8, 2003 and amended on February 26, 2004 by and between the Registrant and Merck & Co., Inc. | |||
| 10.7** | Letter Agreement between the Registrant and John M. Maraganore, Ph.D. dated October 30, 2002 | |||
| 10.8** | Letter Agreement between the Registrant and Vincent J. Miles, Ph.D. dated June 16, 2003 | |||
| 10.9** | Letter Agreement between the Registrant and Thomas R. Ulich, M.D. dated June 15, 2003 | |||
| 10.10** | Letter Agreement between the Registrant and Barry E. Greene dated September 29, 2003 | |||
| 10.11 | Loan and Security Agreement by and between Lighthouse Capital Partners V, L.P. and the Registrant dated as of March 26, 2004, together with the Negative Pledge Agreement by and between Lighthouse Capital Partners V, L.P. and the Registrant dated as of March 26, 2004. | |||
| 10.12 | Warrants to Purchase Preferred Stock effective as of March 30, 2004 issued to Lighthouse Capital Partners V, L.P. and Lighthouse Capital Partners IV, L.P. | |||
| 10.13** | Warrant to Purchase Stock dated December 18, 2002 issued to Silicon Valley Bank | |||
| 10.14** | Lease, dated as of August 5, 2003, between the Registrant and ARE-770/784/790 Memorial Drive, LLC, as amended | |||
| 10.15** | Lease, dated as of September 26, 2003 by and between the Registrant and Three Hundred Third Street LLC | |||
| 10.16†* | * | License Agreement between Cancer Research Technology Limited and Alnylam U.S., Inc. dated July 18, 2003 | ||
| 10.17†* | * | License Agreement between the Carnegie Institution of Washington and Alnylam Europe, AG, effective March 1, 2002, as amended by letter agreements dated September 2, 2002 and October 28, 2003. | ||
| 10.18†* | * | License Agreement by and between the Cold Spring Harbor Laboratory and Alnylam U.S., Inc. dated December 30, 2003 | ||
| 10.19†* | * | Co-exclusive License Agreement between Garching Innovation GmbH and Alnylam U.S., Inc. dated December 20, 2002, as amended by Amendment dated July 8, 2003 | ||
| 10.20†* | * | Co-exclusive License Agreement between Garching Innovation GmbH and Alnylam Europe, AG dated July 30, 2003 | ||
II-1
Table of Contents
| Exhibit | ||||
| No. | Description | |||
| 10.21†* | * | Agreement between The Board of Trustees of the Leland Stanford Junior University and Alnylam U.S., Inc. effective as of September 17, 2003 | ||
| 10.22†* | * | Research Collaboration and License Agreement by and among Merck & Co., Inc., Alnylam U.S., Inc. and Registrant dated September 8, 2003 | ||
| 10.23†* | * | Sponsored Research Agreement among Mayo Foundation for Medical Education and Research, Mayo Clinic Jacksonville and Alnylam Pharmaceuticals, Inc. effective as of October 1, 2003 | ||
| 10.24† | Strategic Collaboration and License Agreement effective as of March 11, 2004 between Isis Pharmaceuticals, Inc. and the Registrant | |||
| 10.25 | Investor Rights Agreement entered into as of March 11, 2004 by and between the Registrant and Isis Pharmaceuticals, Inc. | |||
| 21.1 | Subsidiaries of the Registrant | |||
| 23.1 | Consent of PricewaterhouseCoopers LLP, Independent Auditors | |||
| 23.2 | Consent of PricewaterhouseCoopers Gesellschaft mit beschränkter Haftung Wirtschaftsprüfungsgesellschaft, Independent Auditors | |||
| 23.3* | Consent of Hale and Dorr LLP (included in Exhibit 5.1) | |||
| 24.1** | Power of Attorney | |||
| * | To be filed by amendment. |
| ** | Previously filed. |
| † | Confidential treatment requested as to certain portions, which portions have been filed separately with the Securities and Exchange Commission. |