Exhibit 99.2
| 1Q22 Financial Results Conference Call & Webcast May 9, 2022 At the Forefront of Therapies for Rare Diseases |
| 2 Forward-Looking Statements This presentation contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995 relating to preclinical and clinical development of our product candidates, the timing and reporting of results from preclinical studies and clinical trials, the prospects and timing of the potential regulatory approval of our product candidates, commercialization plans, manufacturing and supply plans, financing plans, and the projected revenues and cash position for the Company. The inclusion of forward-looking statements should not be regarded as a representation by us that any of our plans will be achieved. Any or all of the forward-looking statements in this presentation may turn out to be wrong and can be affected by inaccurate assumptions we might make or by known or unknown risks and uncertainties. For example, with respect to statements regarding the goals, progress, timing, and outcomes of discussions with regulatory authorities, and in particular the potential goals, progress, timing, and results of preclinical studies and clinical trials, and revenue goals, including as they are impacted by COVID-19 related disruption, are based on current information. The potential impact on operations and/or revenue from the COVID-19 pandemic is inherently unknown and cannot be predicted with confidence and may cause actual results and performance to differ materially from the statements in this release, including without limitation, because of the impact on general political and economic conditions, including as a result of efforts by governmental authorities to mitigate COVID-19, such as travel bans, shelter in place orders and third-party business closures and resource allocations, manufacturing and supply chain disruptions and limitations on patient access to commercial or clinical product or to treatment sites. In addition to the impact of the COVID-19 pandemic, actual results may differ materially from those set forth in this release due to the risks and uncertainties inherent in our business, including, without limitation: the potential that results of clinical or preclinical studies indicate that the product candidates are unsafe or ineffective; the potential that it may be difficult to enroll patients in our clinical trials; the potential that regulatory authorities, including the FDA, EMA, and PMDA, may not grant or may delay approval for our product candidates; the potential that we may not be successful in commercializing Galafold in Europe, U.K., Japan, the U.S. and other geographies or our other product candidates if and when approved; the potential that preclinical and clinical studies could be delayed because we identify serious side effects or other safety issues; the potential that we may not be able to manufacture or supply sufficient clinical or commercial products; and the potential that we will need additional funding to complete all of our studies, commercialization and manufacturing. Further, the results of earlier preclinical studies and/or clinical trials may not be predictive of future results. With respect to statements regarding corporate financial guidance and financial goals and the attainment of such goals and statements regarding projections of the Company's revenue and cash position, actual results may differ based on market factors and the Company's ability to execute its operational and budget plans. In addition, all forward-looking statements are subject to other risks detailed in our Annual Report on Form 10-K for the year ended December 31, 2021, and on Form 10-Q for the quarter ended March 31, 2022, to be filed today. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement, and we undertake no obligation to revise or update this news release to reflect events or circumstances after the date hereof. Non-GAAP Financial Measures In addition to financial information prepared in accordance with U.S. GAAP, this presentation also contains adjusted financial measures that we believe provide investors and management with supplemental information relating to operating performance and trends that facilitate comparisons between periods and with respect to projected information. These adjusted financial measures are non-GAAP measures and should be considered in addition to, but not as a substitute for, the information prepared in accordance with U.S. GAAP. We typically exclude certain GAAP items that management does not believe affect our basic operations and that do not meet the GAAP definition of unusual or non-recurring items. Other companies may define these measures in different ways. When we provide our expectation for non-GAAP operating expenses on a forward-looking basis, a reconciliation of the differences between the non-GAAP expectation and the corresponding GAAP measure generally is not available without unreasonable effort due to potentially high variability, complexity and low visibility as to the items that would be excluded from the GAAP measure in the relevant future period, such as unusual gains or losses. The variability of the excluded items may have a significant, and potentially unpredictable, impact on our future GAAP results. |
| 3 A Rare Company Patient-Dedicated, Rare Disease Biotechnology Company with Sustained Double-Digit Revenue Growth, a Global Commercial Infrastructure, and Late-stage Development Capabilities AT-GAA a Two-Component Therapy Under Global Regulatory Reviews for Pompe Disease GLOBAL COMMERCIAL ORGANIZATION World-Class CLINICAL DEVELOPMENT Capabilities EMPLOYEES in 27 Countries GALAFOLD & AT-GAA Gene Therapy PLATFORM Leveraging Experience in Protein Engineering & Glycobiology $411M Cash as of 3/31/22 $350M-$365M FY22 Global Galafold Revenue at CER PROFITABILITY expected in 2023 Cumulative $2B Peak Potential |
| 2022 Strategic Priorities to Drive Value 1 2 3 4 5 Double-digit Galafold growth (15-20%) with revenue of $350M to $365M at CER1 Secure FDA approval and positive CHMP opinion for AT-GAA Initiate successful, rapid launch in U.S. for AT-GAA Advance best-in-class next-generation genetic medicines and capabilities Maintain strong financial position on path to profitability 4 12022 Galafold revenue guidance is based on the average exchange rates for 2021 |
| 5 Positioned for Significant Value Growth Focused on Execution and Driving Sustainable Double-Digit Revenue Growth on Path to Profitability Continue to bring Galafold® to as many patients as possible, sustain double-digit revenue growth Successful launch of AT-GAA for people living with Pompe disease Advance next-generation gene therapies in Fabry and Pompe diseases Fully leverage global capabilities and infrastructure as a leader in rare diseases Achieve self- sustainability and profitability in 2023 |
| 6 Galafold® (migalastat) Continued Growth... … building a leadership position in the treatment of Fabry disease |
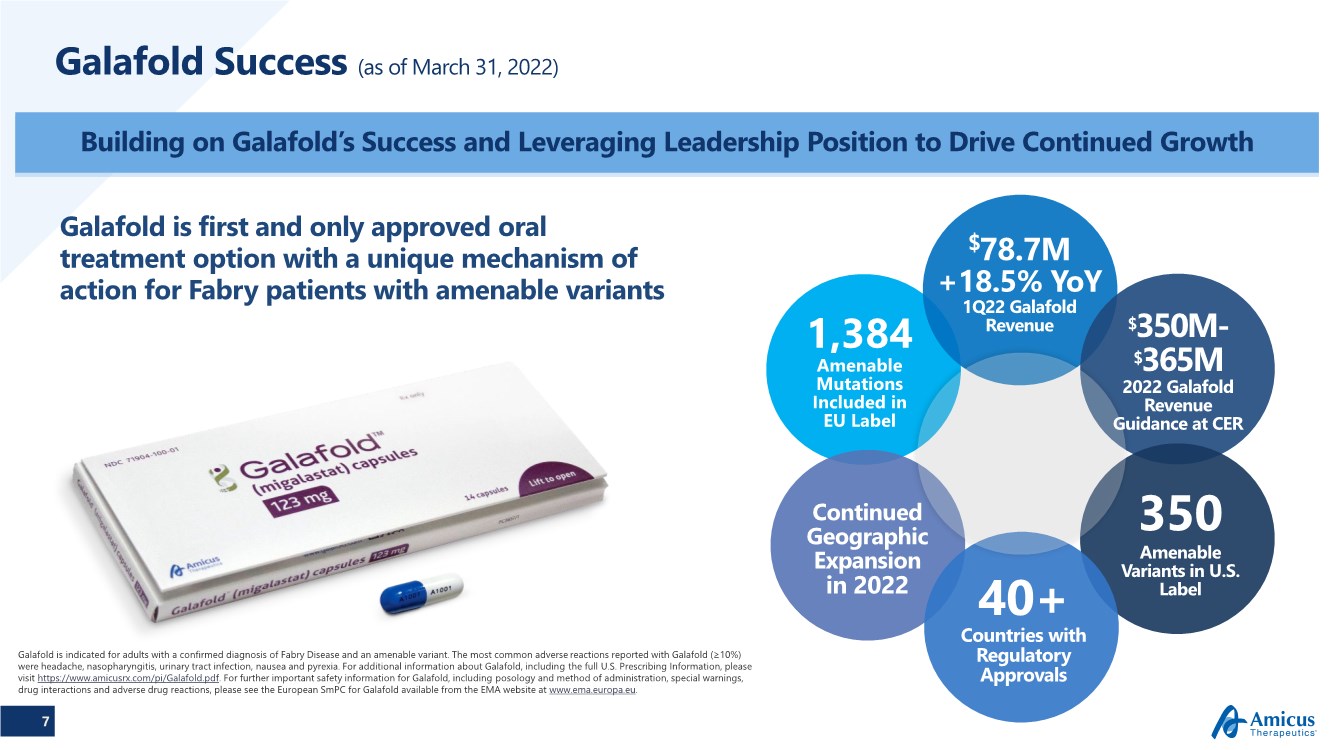
| 7 Galafold Success (as of March 31, 2022) Building on Galafold’s Success and Leveraging Leadership Position to Drive Continued Growth Galafold is indicated for adults with a confirmed diagnosis of Fabry Disease and an amenable variant. The most common adverse reactions reported with Galafold (≥10%) were headache, nasopharyngitis, urinary tract infection, nausea and pyrexia. For additional information about Galafold, including the full U.S. Prescribing Information, please visit https://www.amicusrx.com/pi/Galafold.pdf. For further important safety information for Galafold, including posology and method of administration, special warnings, drug interactions and adverse drug reactions, please see the European SmPC for Galafold available from the EMA website at www.ema.europa.eu. Galafold is first and only approved oral treatment option with a unique mechanism of action for Fabry patients with amenable variants 350 Amenable Variants in U.S. Label 40+ Countries with Regulatory Approvals Continued Geographic Expansion in 2022 $78.7M +18.5% YoY 1Q22 Galafold Revenue $350M- $365M 2022 Galafold Revenue Guidance at CER 1,384 Amenable Mutations Included in EU Label |
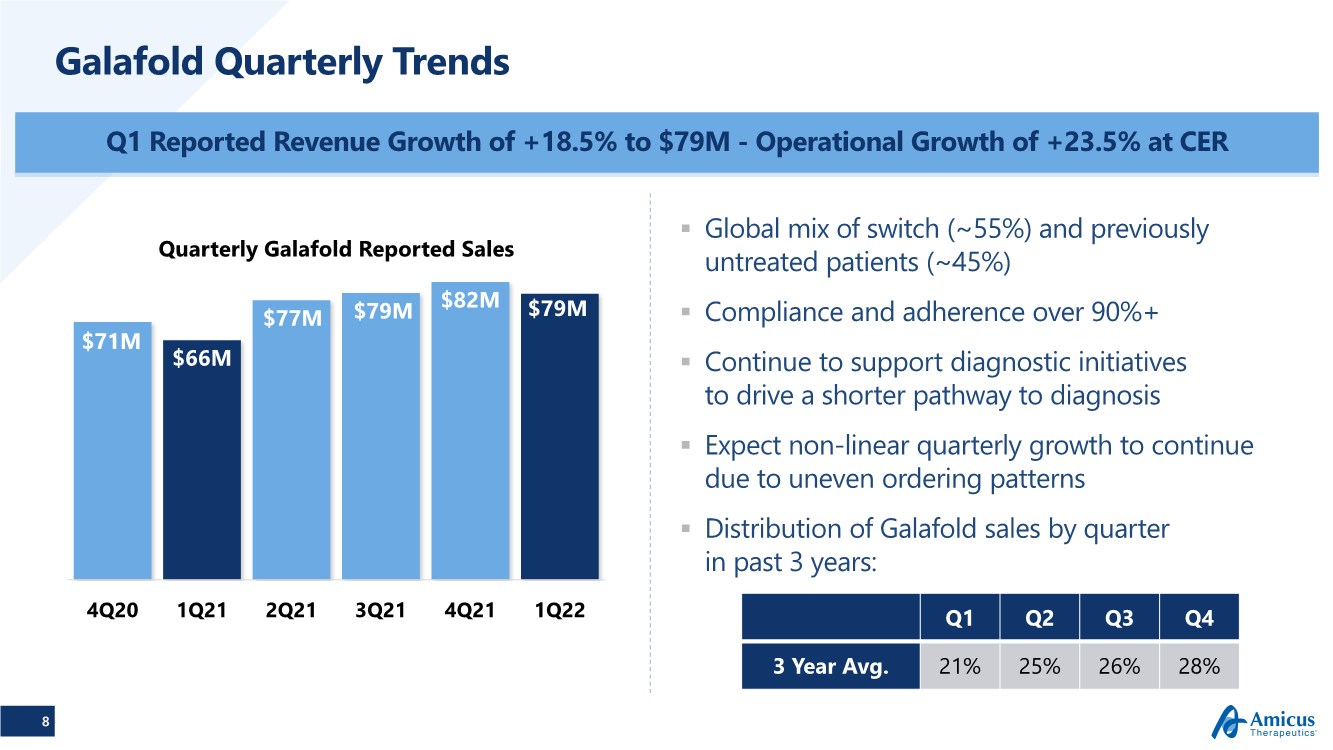
| 8 Galafold Quarterly Trends Q1 Reported Revenue Growth of +18.5% to $79M - Operational Growth of +23.5% at CER $71M $66M $77M $79M $82M $79M 4Q20 1Q21 2Q21 3Q21 4Q21 1Q22 Quarterly Galafold Reported Sales .. Global mix of switch (~55%) and previously untreated patients (~45%) .. Compliance and adherence over 90%+ .. Continue to support diagnostic initiatives to drive a shorter pathway to diagnosis .. Expect non-linear quarterly growth to continue due to uneven ordering patterns .. Distribution of Galafold sales by quarter in past 3 years: Q1 Q2 Q3 Q4 3 Year Avg. 21% 25% 26% 28% |
| 9 Galafold Success and FY22 Revenue Guidance Galafold Momentum on Track to Achieve Full-Year 2022 Revenue Guidance at CER FY16 FY17 FY18 FY19 FY20 FY21 FY22 $5.0M Q1 $78.7M FY22 Rev. Guidance $350M-$365M at CER1 $36.9M $91.2M $182.2M $260.9M $305.5M 12022 Galafold revenue guidance utilizes the average actual rates for 2021 |
| 10 Penetration of the diagnosed untreated population Increase in newborn screening and diagnostic initiatives Strong intellectual property rights, including COM protection through 2038 Continued penetration into existing markets Expansion into new geographies Broadening of labels Galafold Growth Opportunity $1B Annual Sales Opportunity at Peak Sustained double-digit revenue growth: Near-term growth to $500M driven by: Long-term growth towards peak sales potential driven by: 1Q operational revenue growth of +24% |
| 11 Galafold Initiatives Building the Body of the Evidence around Galafold Broadening Labels: Adolescents and Additional Variants Publications and Medical Presentations Ongoing and Planned Phase IV Studies Over 500 Patients Enrolled in a Global Registry Strengthening our IP Portfolio |
| 12 AT-GAA (cipaglucosidase alfa + miglustat) … potential to establish a new standard of care for people living with Pompe disease |
| 13 Deficiency of GAA leading to lysosomal glycogen accumulation and cellular dysfunction Age of onset ranges from infancy to adulthood Symptoms include muscle weakness, respiratory failure, and cardiomyopathy Respiratory and cardiac failure are leading causes of morbidity and mortality Estimated incidence of ~1:28,000 ; newborn screening suggests significant underdiagnosis ~$1.2B+ global Pompe ERT sales1 Majority of patients on current standard of care decline after ~2 years Pompe Disease Overview 1. Based on 12 months ended December 31, 2021. Source: Sanofi Press Release Pompe is a Severe and Fatal Neuromuscular Disease Caused by the Deficiency of Lysosomal Enzyme GAA |
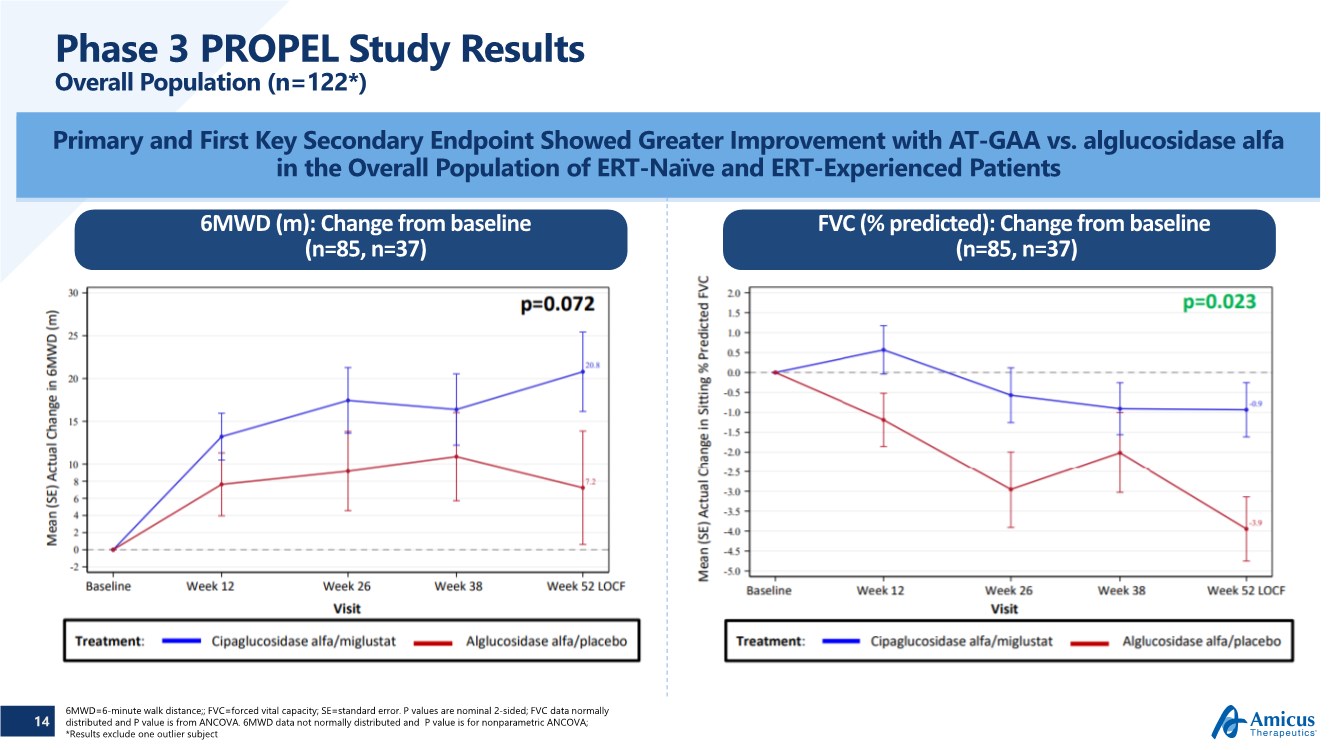
| 14 Phase 3 PROPEL Study Results Overall Population (n=122*) Primary and First Key Secondary Endpoint Showed Greater Improvement with AT-GAA vs. alglucosidase alfa in the Overall Population of ERT-Naïve and ERT-Experienced Patients 6MWD=6-minute walk distance;; FVC=forced vital capacity; SE=standard error. P values are nominal 2-sided; FVC data normally distributed and P value is from ANCOVA. 6MWD data not normally distributed and P value is for nonparametric ANCOVA; *Results exclude one outlier subject 6MWD (m): Change from baseline (n=85, n=37) FVC (% predicted): Change from baseline (n=85, n=37) |
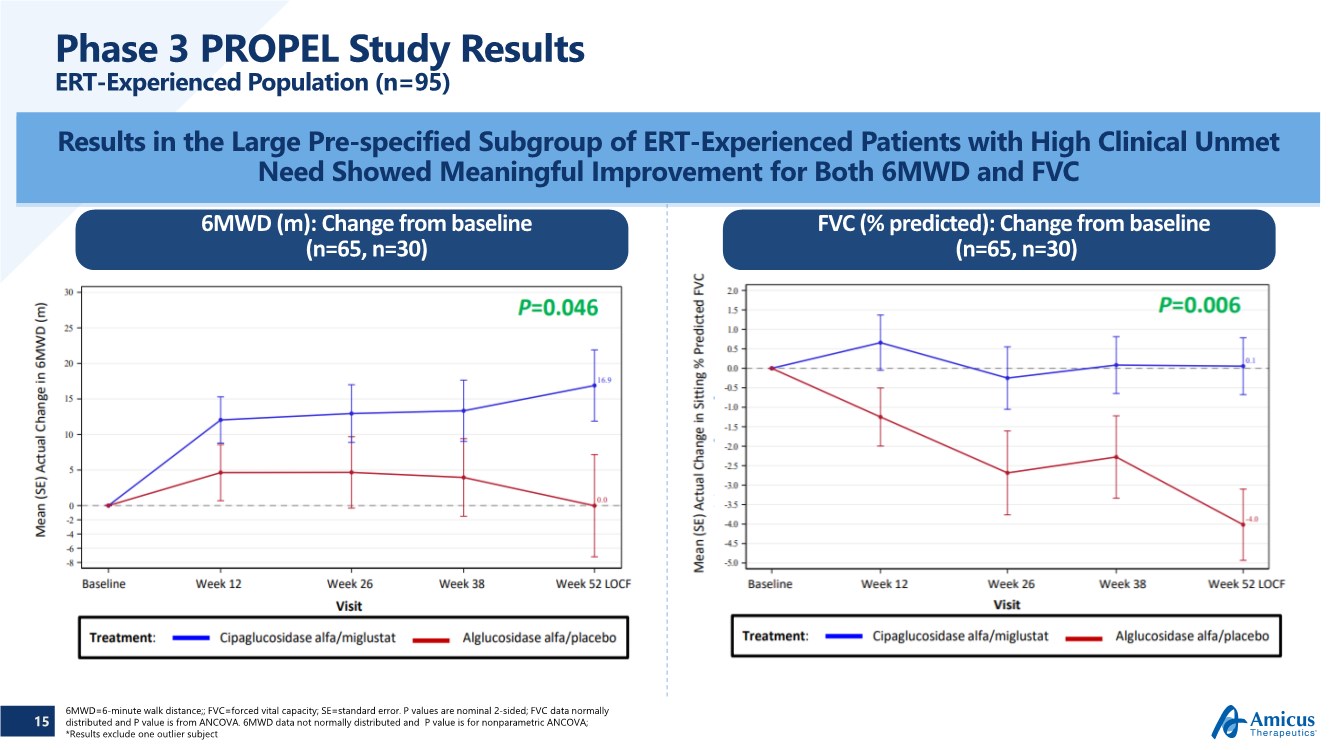
| 15 Phase 3 PROPEL Study Results ERT-Experienced Population (n=95) Results in the Large Pre-specified Subgroup of ERT-Experienced Patients with High Clinical Unmet Need Showed Meaningful Improvement for Both 6MWD and FVC 6MWD (m): Change from baseline (n=65, n=30) FVC (% predicted): Change from baseline (n=65, n=30) 6MWD=6-minute walk distance;; FVC=forced vital capacity; SE=standard error. P values are nominal 2-sided; FVC data normally distributed and P value is from ANCOVA. 6MWD data not normally distributed and P value is for nonparametric ANCOVA; *Results exclude one outlier subject |
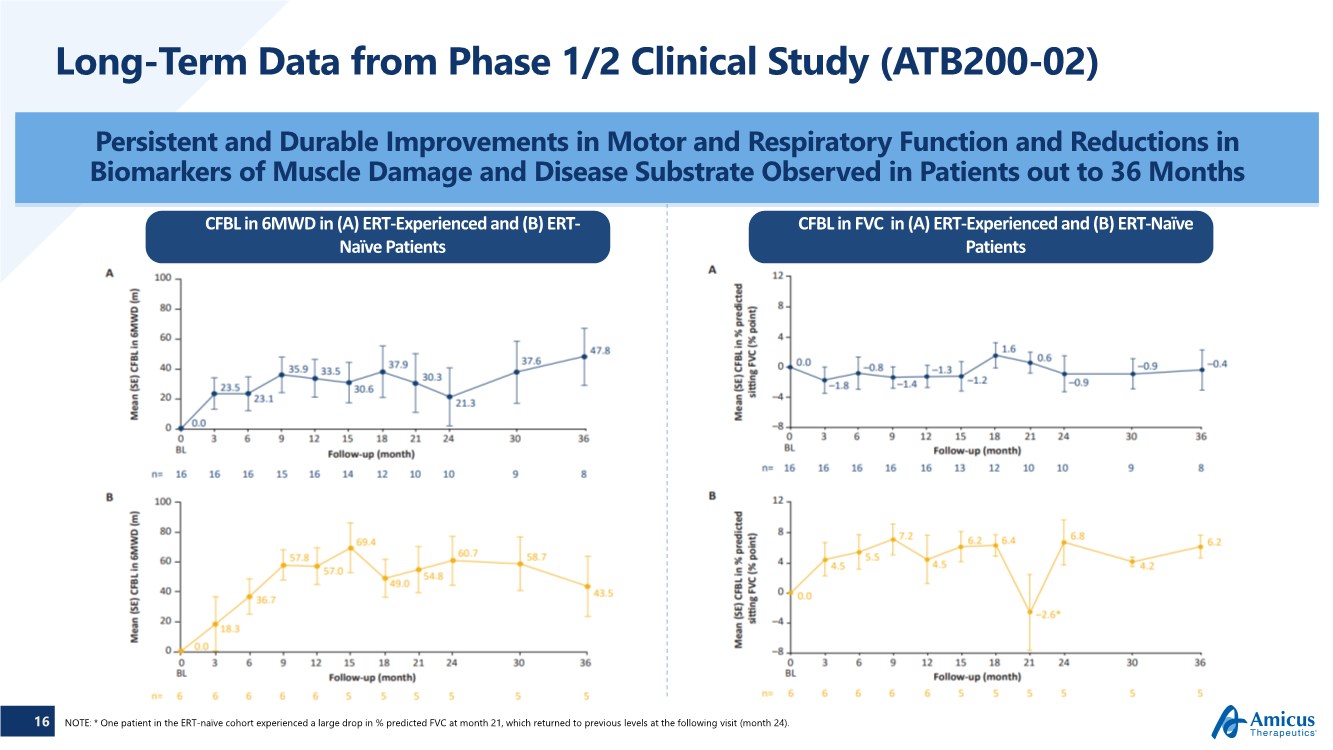
| 16 Long-Term Data from Phase 1/2 Clinical Study (ATB200-02) Persistent and Durable Improvements in Motor and Respiratory Function and Reductions in Biomarkers of Muscle Damage and Disease Substrate Observed in Patients out to 36 Months NOTE: * One patient in the ERT-naïve cohort experienced a large drop in % predicted FVC at month 21, which returned to previous levels at the following visit (month 24). CFBL in 6MWD in (A) ERT-Experienced and (B) ERT- Naïve Patients CFBL in FVC in (A) ERT-Experienced and (B) ERT-Naïve Patients |
| 17 AT-GAA: Key Takeaways .. Regulatory status update: – U.S. PDUFA date mid 20221 – CHMP opinion late 2022 – Planning for additional regulatory submissions .. Multiple expanded access mechanisms in place, including in the U.K., Germany, France, Japan, and others .. 150+ people living with Pompe disease are on AT-GAA today across our clinical extension studies and expanded access programs .. Ongoing supportive studies: – Late-Onset Pompe Disease (LOPD) in children and adolescents aged 0 to <18 – Infantile-Onset Pompe Disease (IOPD) Focused on Advancing AT-GAA to as Many Patients as Possible through Global Regulatory Pathways and Expanded Access Mechanisms 1FDA PDUFA date of May 29, 2022 for miglustat NDA and July 29, 2022 for cipaglucosidase alfa BLA |
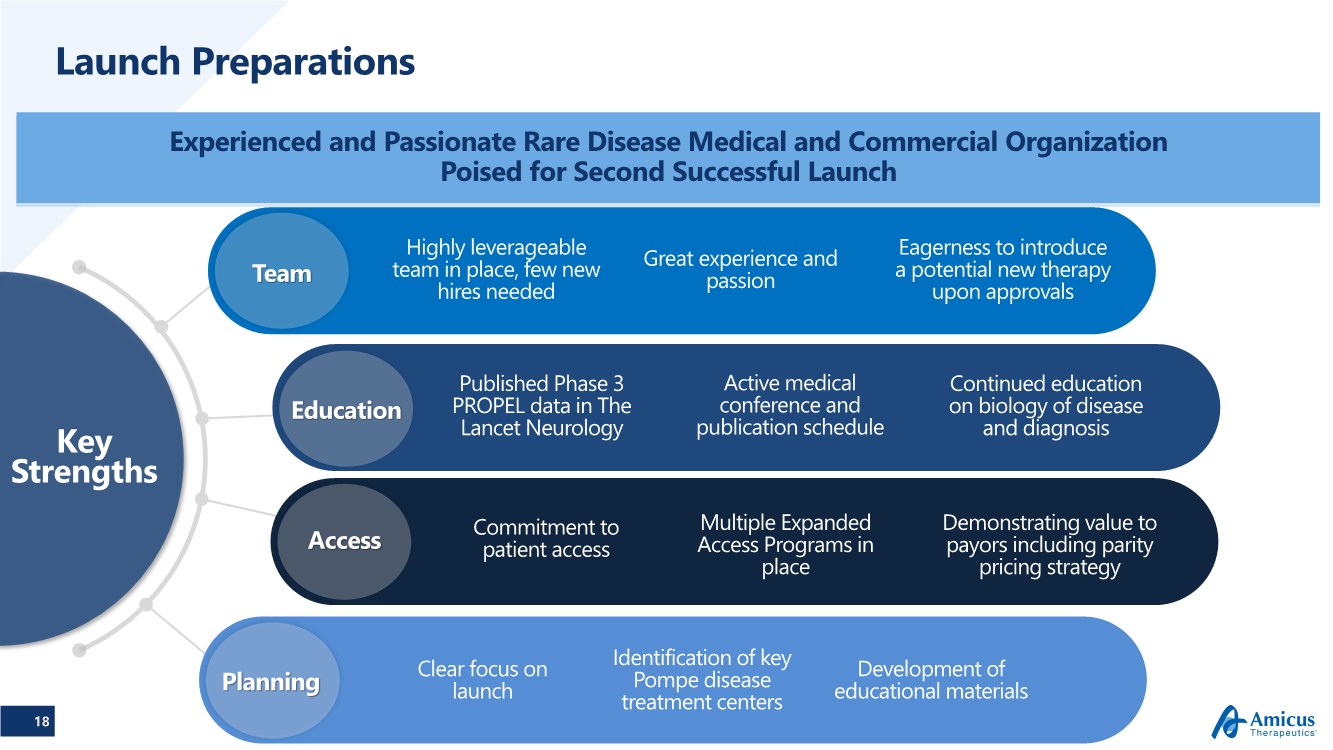
| 18 Launch Preparations Experienced and Passionate Rare Disease Medical and Commercial Organization Poised for Second Successful Launch Key Strengths Commitment to patient access Clear focus on launch Identification of key Pompe disease treatment centers Development of educational materials Planning Access Education Team Great experience and passion Eagerness to introduce a potential new therapy upon approvals Highly leverageable team in place, few new hires needed Published Phase 3 PROPEL data in The Lancet Neurology Active medical conference and publication schedule Multiple Expanded Access Programs in place Continued education on biology of disease and diagnosis Demonstrating value to payors including parity pricing strategy |
| 19 Financial & Operational Strategy … maintaining a strong financial outlook |
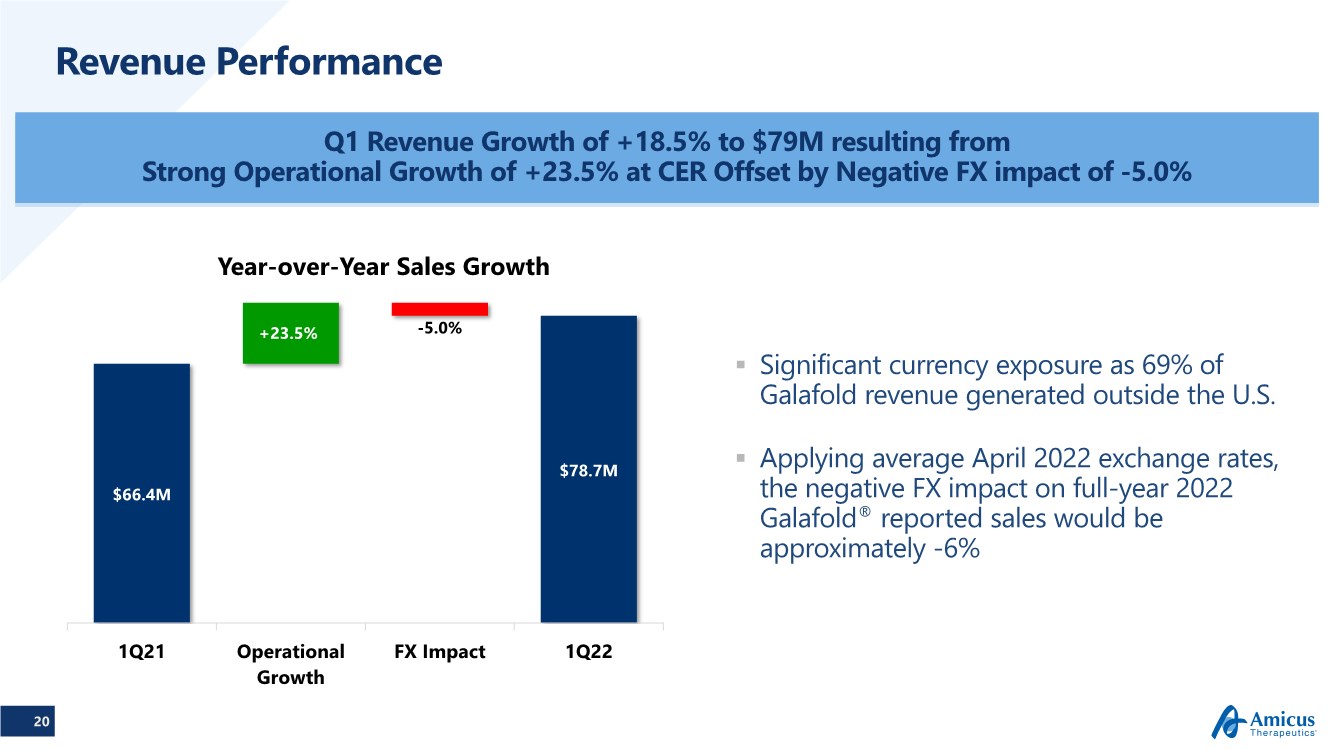
| 20 Revenue Performance Q1 Revenue Growth of +18.5% to $79M resulting from Strong Operational Growth of +23.5% at CER Offset by Negative FX impact of -5.0% $66.4M $78.7M -5.0% +23.5% 1Q21 Operational Growth FX Impact 1Q22 Year-over-Year Sales Growth .. Significant currency exposure as 69% of Galafold revenue generated outside the U.S. .. Applying average April 2022 exchange rates, the negative FX impact on full-year 2022 Galafold® reported sales would be approximately -6% |
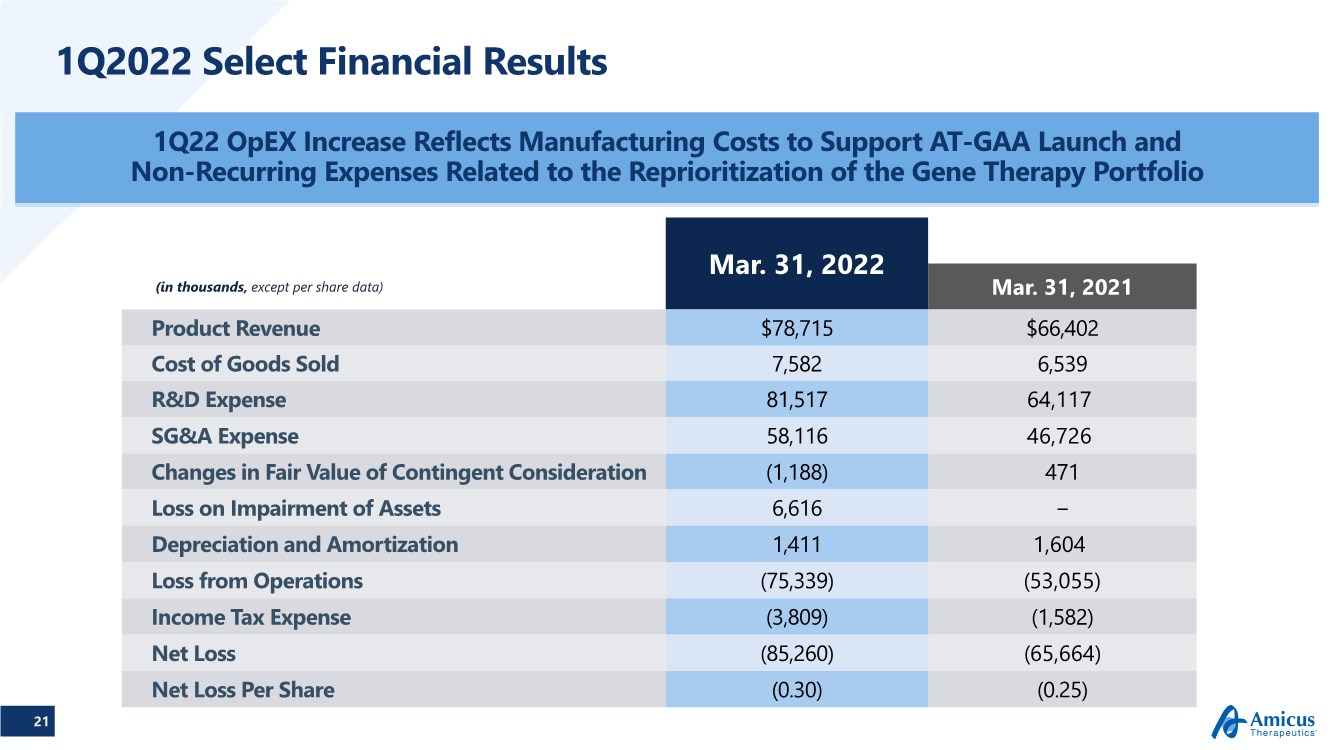
| 21 1Q2022 Select Financial Results 1Q22 OpEX Increase Reflects Manufacturing Costs to Support AT-GAA Launch and Non-Recurring Expenses Related to the Reprioritization of the Gene Therapy Portfolio Mar. 31, 2022 (in thousands, except per share data) Mar. 31, 2021 Product Revenue $78,715 $66,402 Cost of Goods Sold 7,582 6,539 R&D Expense 81,517 64,117 SG&A Expense 58,116 46,726 Changes in Fair Value of Contingent Consideration (1,188) 471 Loss on Impairment of Assets 6,616 − Depreciation and Amortization 1,411 1,604 Loss from Operations (75,339) (53,055) Income Tax Expense (3,809) (1,582) Net Loss (85,260) (65,664) Net Loss Per Share (0.30) (0.25) |
| Financial Outlook and Path to Profitability Clear Strategy to Build our Business, Advance our Portfolio, and Achieve Profitability 22 Sustain Galafold Revenue Growth Deliver on Financial Goals Secure Approvals of AT-GAA $79M 1Q2022 revenue, +24% YoY Operational Growth 2022 Galafold revenue guidance of $350M-$365M at CER, +15-20% YoY Growth Galafold and AT-GAA expected to drive strong double-digit growth long term Focused on prudent expense management Achieve profitability1 in 2023 1Based on projections of Amicus non-GAAP Net Income under current operating plans, which includes successful AT-GAA regulatory approvals and continued Galafold growth. We define non-GAAP Net Income as GAAP Net Income excluding the impact of share-based compensation expense, changes in fair value of contingent consideration, depreciation and amortization, acquisition related income (expense), loss on extinguishment of debt, loss on impairment of assets, restructuring charges and income taxes. 2022 Non-GAAP operating expense guidance of $470M-$485M |
| Thank You |
| Appendix |
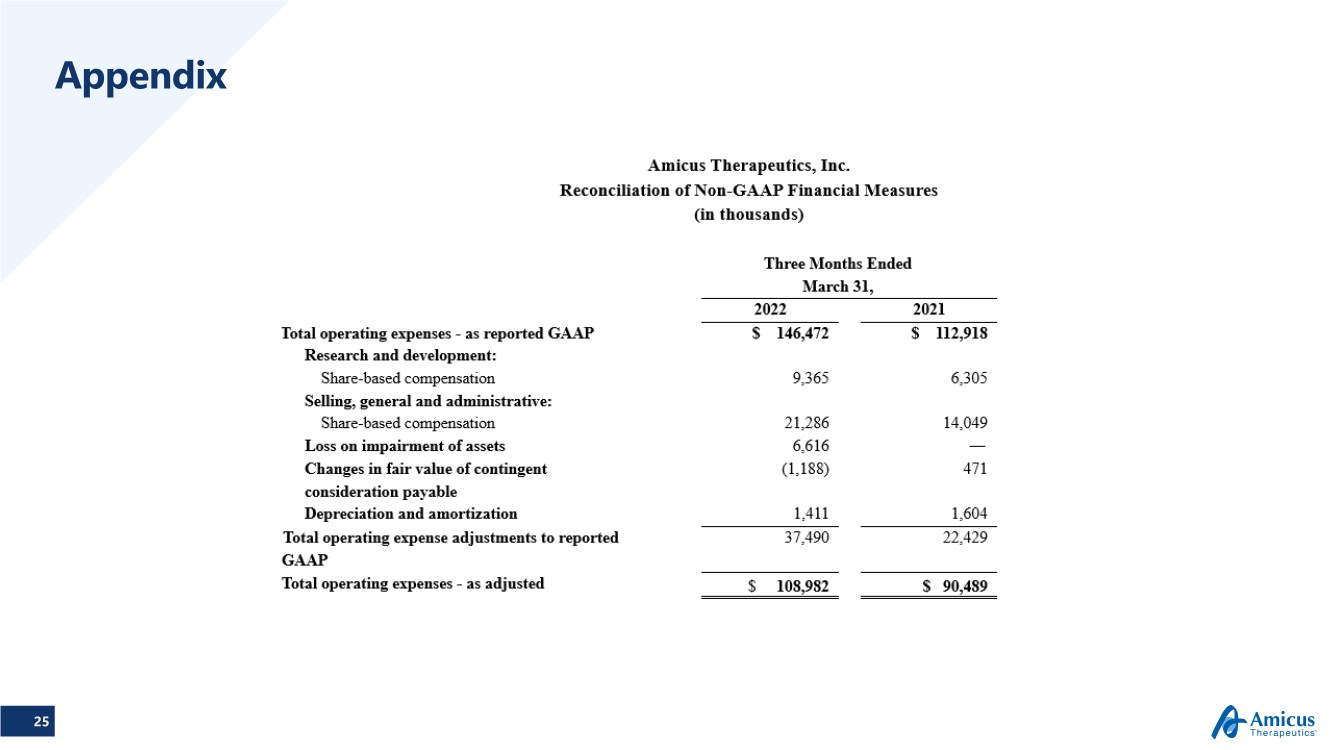
| 25 Appendix |