©2021 Cardiovascular Systems, Inc. All Rights Reserved. Q3 FY21 Earnings Supplement May 6, 2021

Safe Harbor This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Report Act of 1995, which are provided under the protection of the safe harbor for forward-looking statements provided by that Act. For example, statements in this presentation regarding CSI’s strategy; growth; future financial measurements and investments; product development plans, milestones and introductions; geographic expansion; clinical trials and evidence; market estimates and opportunities; developments related to the COVID-19 pandemic; and anticipated product upgrades and reduced production volumes, and the impact thereof are forward-looking statements. These statements involve risks and uncertainties that could cause results differ materially from those projected, including, but not limited to, those described in CSI’s filings with the Securities and Exchange Commission, including its most recent annual report on Form 10-K and subsequent quarterly and annual reports. CSI encourages you to consider all of these risks, uncertainties and other factors carefully in evaluating the forward-looking statements contained in this presentation. As a result of these matters, changes in facts, assumptions not being realized or other circumstances, CSI’s actual results may differ materially from the expected results discussed in the forward-looking statements contained in this presentation. The forward-looking statements contained in this presentation are made only as of the date of this presentation, and CSI undertakes no obligation to update them to reflect subsequent events or circumstances. 2 FORWARD LOOKING STATEMENTS FINANCIAL INFORMATION This presentation includes calculations or figures that have been prepared internally and have not been reviewed or audited by CSI’s independent registered accounting firm. Use of different methods for preparing, calculating or presenting information may lead to differences, which may be material. In addition, this presentation also includes certain non-GAAP financial measures, such as Adjusted EBITDA. Reconciliations of the non-GAAP financial measures used in this presentation to the most comparable U.S. GAAP measures for the respective periods can be found in tables in the appendix to this presentation. Non-GAAP financial measures have limitations as analytical tools and should not be considered in isolation or as a substitute for CSI's financial results prepared in accordance with GAAP.
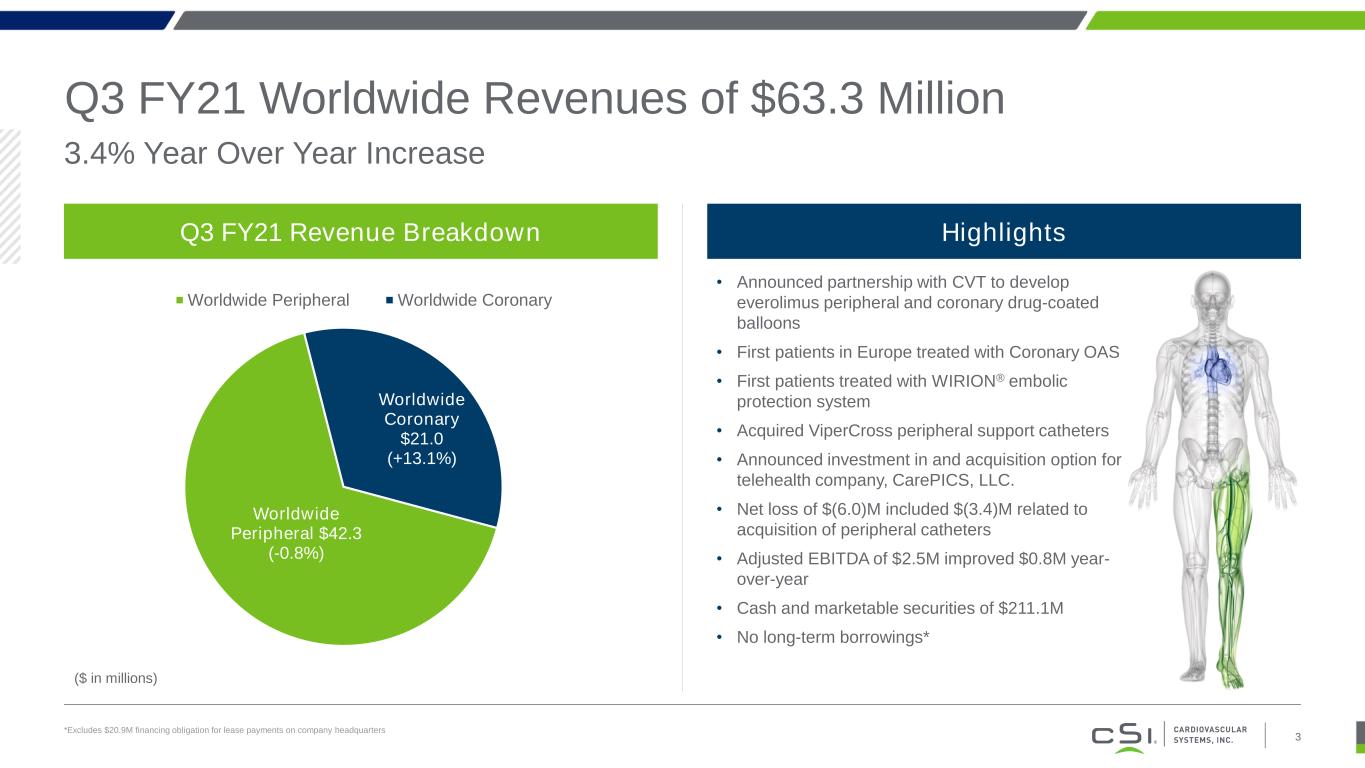
3.4% Year Over Year Increase Q3 FY21 Worldwide Revenues of $63.3 Million *Excludes $20.9M financing obligation for lease payments on company headquarters Q3 FY21 Revenue Breakdown Worldwide Peripheral $42.3 (-0.8%) Worldwide Coronary $21.0 (+13.1%) Worldwide Peripheral Worldwide Coronary ($ in millions) • Announced partnership with CVT to develop everolimus peripheral and coronary drug-coated balloons • First patients in Europe treated with Coronary OAS • First patients treated with WIRION® embolic protection system • Acquired ViperCross peripheral support catheters • Announced investment in and acquisition option for telehealth company, CarePICS, LLC. • Net loss of $(6.0)M included $(3.4)M related to acquisition of peripheral catheters • Adjusted EBITDA of $2.5M improved $0.8M year- over-year • Cash and marketable securities of $211.1M • No long-term borrowings* Highlights 3
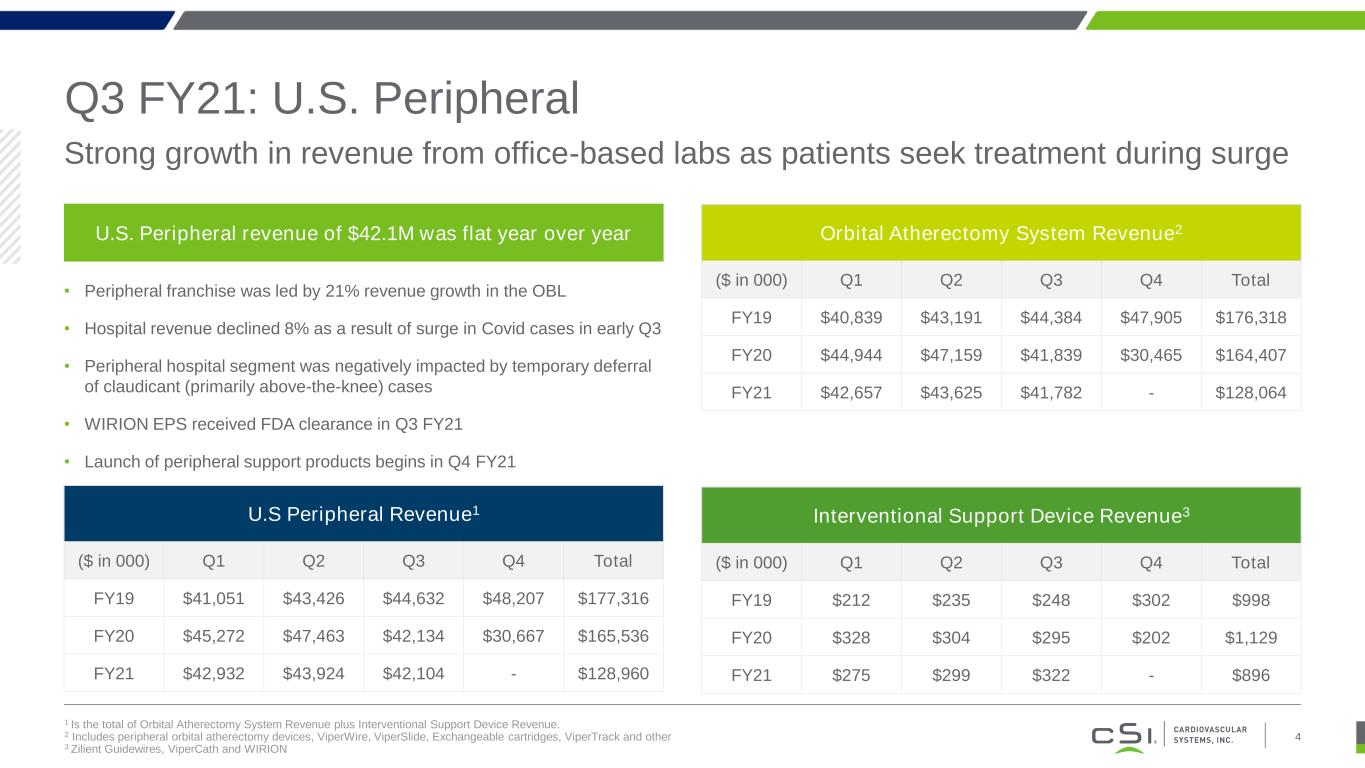
Q3 FY21: U.S. Peripheral 1 Is the total of Orbital Atherectomy System Revenue plus Interventional Support Device Revenue. 2 Includes peripheral orbital atherectomy devices, ViperWire, ViperSlide, Exchangeable cartridges, ViperTrack and other 3 Zilient Guidewires, ViperCath and WIRION Strong growth in revenue from office-based labs as patients seek treatment during surge 4 U.S. Peripheral revenue of $42.1M was flat year over year • Peripheral franchise was led by 21% revenue growth in the OBL • Hospital revenue declined 8% as a result of surge in Covid cases in early Q3 • Peripheral hospital segment was negatively impacted by temporary deferral of claudicant (primarily above-the-knee) cases • WIRION EPS received FDA clearance in Q3 FY21 • Launch of peripheral support products begins in Q4 FY21 U.S Peripheral Revenue1 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $41,051 $43,426 $44,632 $48,207 $177,316 FY20 $45,272 $47,463 $42,134 $30,667 $165,536 FY21 $42,932 $43,924 $42,104 - $128,960 Orbital Atherectomy System Revenue2 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $40,839 $43,191 $44,384 $47,905 $176,318 FY20 $44,944 $47,159 $41,839 $30,465 $164,407 FY21 $42,657 $43,625 $41,782 - $128,064 Interventional Support Device Revenue3 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $212 $235 $248 $302 $998 FY20 $328 $304 $295 $202 $1,129 FY21 $275 $299 $322 - $896
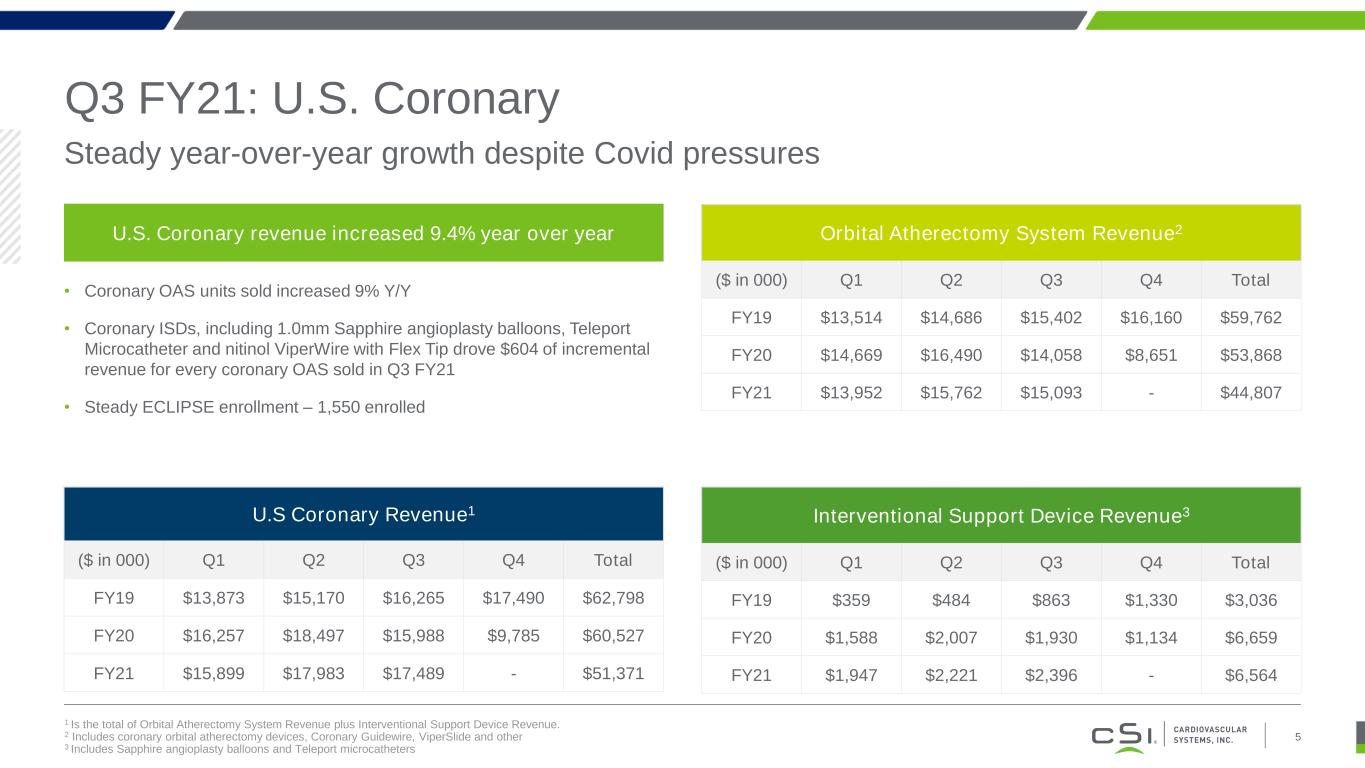
Q3 FY21: U.S. Coronary 1 Is the total of Orbital Atherectomy System Revenue plus Interventional Support Device Revenue. 2 Includes coronary orbital atherectomy devices, Coronary Guidewire, ViperSlide and other 3 Includes Sapphire angioplasty balloons and Teleport microcatheters Steady year-over-year growth despite Covid pressures 5 U.S. Coronary revenue increased 9.4% year over year • Coronary OAS units sold increased 9% Y/Y • Coronary ISDs, including 1.0mm Sapphire angioplasty balloons, Teleport Microcatheter and nitinol ViperWire with Flex Tip drove $604 of incremental revenue for every coronary OAS sold in Q3 FY21 • Steady ECLIPSE enrollment – 1,550 enrolled U.S Coronary Revenue1 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $13,873 $15,170 $16,265 $17,490 $62,798 FY20 $16,257 $18,497 $15,988 $9,785 $60,527 FY21 $15,899 $17,983 $17,489 - $51,371 Orbital Atherectomy System Revenue2 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $13,514 $14,686 $15,402 $16,160 $59,762 FY20 $14,669 $16,490 $14,058 $8,651 $53,868 FY21 $13,952 $15,762 $15,093 - $44,807 Interventional Support Device Revenue3 ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $359 $484 $863 $1,330 $3,036 FY20 $1,588 $2,007 $1,930 $1,134 $6,659 FY21 $1,947 $2,221 $2,396 - $6,564
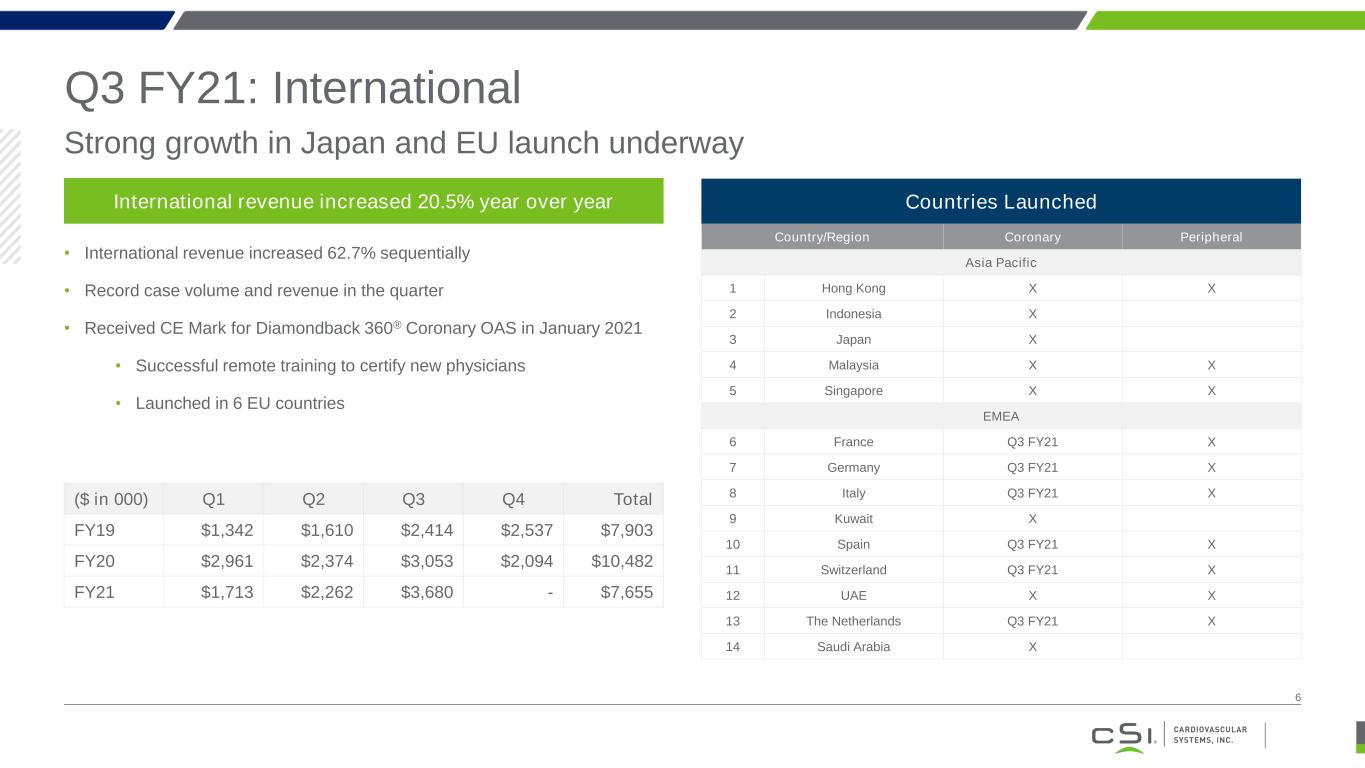
Q3 FY21: International Strong growth in Japan and EU launch underway 6 International revenue increased 20.5% year over year • International revenue increased 62.7% sequentially • Record case volume and revenue in the quarter • Received CE Mark for Diamondback 360® Coronary OAS in January 2021 • Successful remote training to certify new physicians • Launched in 6 EU countries ($ in 000) Q1 Q2 Q3 Q4 Total FY19 $1,342 $1,610 $2,414 $2,537 $7,903 FY20 $2,961 $2,374 $3,053 $2,094 $10,482 FY21 $1,713 $2,262 $3,680 - $7,655 Country/Region Coronary Peripheral Asia Pacific 1 Hong Kong X X 2 Indonesia X 3 Japan X 4 Malaysia X X 5 Singapore X X EMEA 6 France Q3 FY21 X 7 Germany Q3 FY21 X 8 Italy Q3 FY21 X 9 Kuwait X 10 Spain Q3 FY21 X 11 Switzerland Q3 FY21 X 12 UAE X X 13 The Netherlands Q3 FY21 X 14 Saudi Arabia X Countries Launched
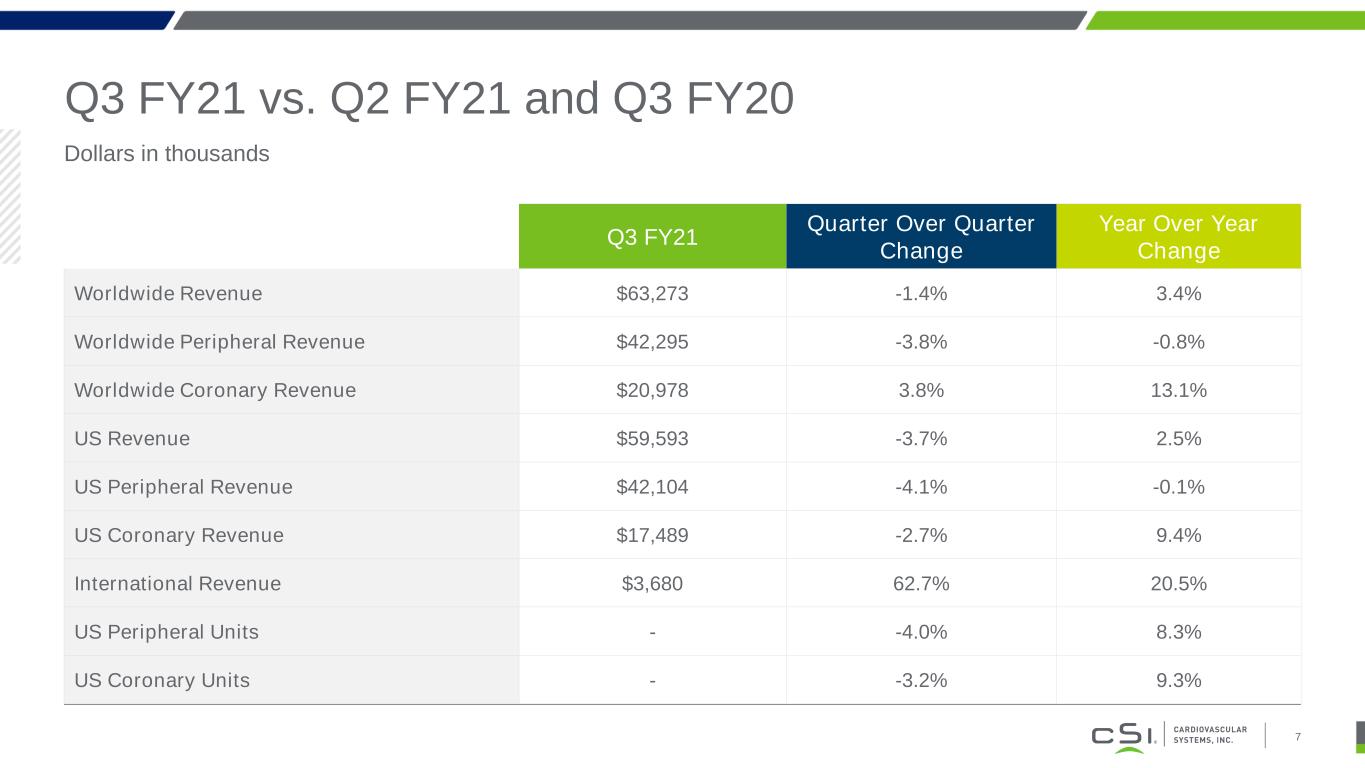
Q3 FY21 vs. Q2 FY21 and Q3 FY20 Dollars in thousands 7 Q3 FY21 Quarter Over Quarter Change Year Over Year Change Worldwide Revenue $63,273 -1.4% 3.4% Worldwide Peripheral Revenue $42,295 -3.8% -0.8% Worldwide Coronary Revenue $20,978 3.8% 13.1% US Revenue $59,593 -3.7% 2.5% US Peripheral Revenue $42,104 -4.1% -0.1% US Coronary Revenue $17,489 -2.7% 9.4% International Revenue $3,680 62.7% 20.5% US Peripheral Units - -4.0% 8.3% US Coronary Units - -3.2% 9.3%
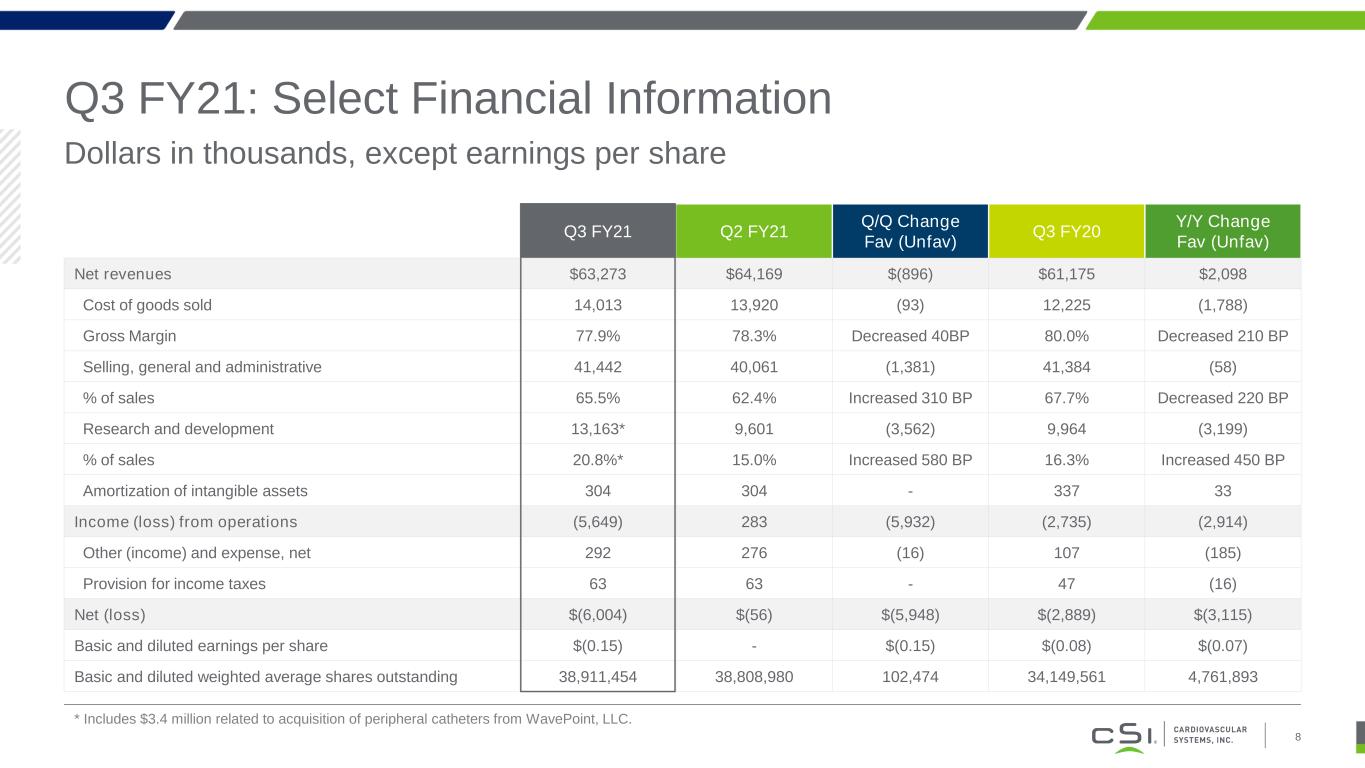
Q3 FY21: Select Financial Information Dollars in thousands, except earnings per share 8 Q3 FY21 Q2 FY21 Q/Q Change Fav (Unfav) Q3 FY20 Y/Y Change Fav (Unfav) Net revenues $63,273 $64,169 $(896) $61,175 $2,098 Cost of goods sold 14,013 13,920 (93) 12,225 (1,788) Gross Margin 77.9% 78.3% Decreased 40BP 80.0% Decreased 210 BP Selling, general and administrative 41,442 40,061 (1,381) 41,384 (58) % of sales 65.5% 62.4% Increased 310 BP 67.7% Decreased 220 BP Research and development 13,163* 9,601 (3,562) 9,964 (3,199) % of sales 20.8%* 15.0% Increased 580 BP 16.3% Increased 450 BP Amortization of intangible assets 304 304 - 337 33 Income (loss) from operations (5,649) 283 (5,932) (2,735) (2,914) Other (income) and expense, net 292 276 (16) 107 (185) Provision for income taxes 63 63 - 47 (16) Net (loss) $(6,004) $(56) $(5,948) $(2,889) $(3,115) Basic and diluted earnings per share $(0.15) - $(0.15) $(0.08) $(0.07) Basic and diluted weighted average shares outstanding 38,911,454 38,808,980 102,474 34,149,561 4,761,893 * Includes $3.4 million related to acquisition of peripheral catheters from WavePoint, LLC.

Q4 FY21: Guidance 9 For the fiscal 2021 fourth quarter ending June 30, 2021, CSI anticipates: Revenue of $67 million to $70 million; Gross profit as a percentage of revenues in the 70% to 71% range*; Operating expenses in a range of $53.5 million to $55.5 million; Net loss in a range of $6 million to $7 million; and Adjusted EBITDA loss of $1 million to $2 million. * Two factors will result in temporary lower gross margins during Q4: 1. CSI will incur a one-time charge to cost of goods sold related to the upgrade of saline pumps that will be reaching end of service over the coming 24-36 months. 2. Throughout the pandemic, CSI operated its production facilities to ensure adequate safety stock. With pandemic conditions stabilizing, CSI will temporarily reduce production during Q4 to restore normal inventory levels. Combined, these factors are expected to result in gross margin of approximately 70% to 71% during Q4.
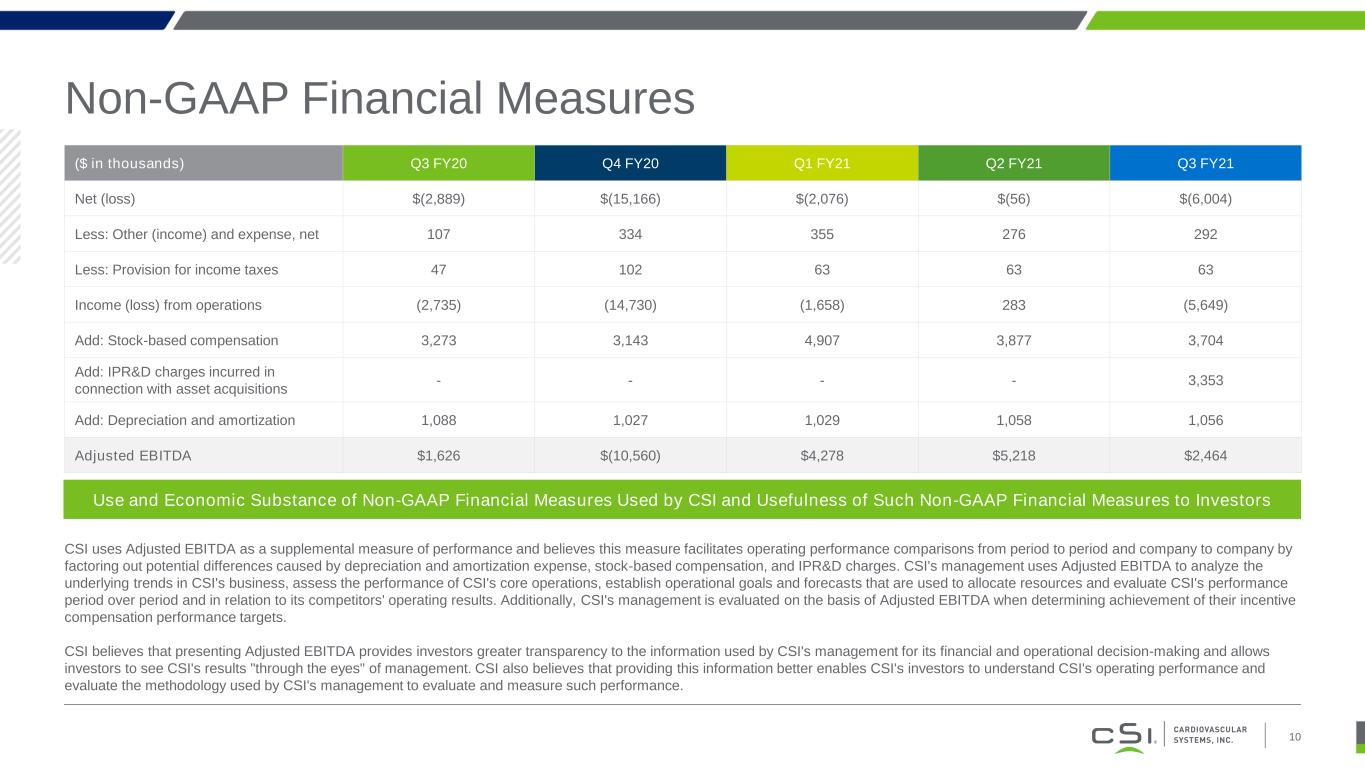
Non-GAAP Financial Measures 10 CSI uses Adjusted EBITDA as a supplemental measure of performance and believes this measure facilitates operating performance comparisons from period to period and company to company by factoring out potential differences caused by depreciation and amortization expense, stock-based compensation, and IPR&D charges. CSI's management uses Adjusted EBITDA to analyze the underlying trends in CSI's business, assess the performance of CSI's core operations, establish operational goals and forecasts that are used to allocate resources and evaluate CSI's performance period over period and in relation to its competitors' operating results. Additionally, CSI's management is evaluated on the basis of Adjusted EBITDA when determining achievement of their incentive compensation performance targets. CSI believes that presenting Adjusted EBITDA provides investors greater transparency to the information used by CSI's management for its financial and operational decision-making and allows investors to see CSI's results "through the eyes" of management. CSI also believes that providing this information better enables CSI's investors to understand CSI's operating performance and evaluate the methodology used by CSI's management to evaluate and measure such performance. ($ in thousands) Q3 FY20 Q4 FY20 Q1 FY21 Q2 FY21 Q3 FY21 Net (loss) $(2,889) $(15,166) $(2,076) $(56) $(6,004) Less: Other (income) and expense, net 107 334 355 276 292 Less: Provision for income taxes 47 102 63 63 63 Income (loss) from operations (2,735) (14,730) (1,658) 283 (5,649) Add: Stock-based compensation 3,273 3,143 4,907 3,877 3,704 Add: IPR&D charges incurred in connection with asset acquisitions - - - - 3,353 Add: Depreciation and amortization 1,088 1,027 1,029 1,058 1,056 Adjusted EBITDA $1,626 $(10,560) $4,278 $5,218 $2,464 Use and Economic Substance of Non-GAAP Financial Measures Used by CSI and Usefulness of Such Non-GAAP Financial Measures to Investors

Investor Contact: Jack Nielsen 651-202-4919 j.nielsen@csi360.com CSI®, Diamondback®, Diamondback 360®, GlideAssist®, ViperWire®, WIRION® and ViperWire Advance® are trademarks of Cardiovascular Systems, Inc. © 2021 Cardiovascular Systems, Inc. OrbusNeich®, Teleport® and Sapphire® are trademarks of OrbusNeich Medical, Inc. For more information: www.csi360.com Cardiovascular Systems, Inc. CSII @csi360 11
