Exhibit 99.1
 |
C. E. UNTERBERG, TOWBIN
LIFE SCIENCES
Life Sciences Conference
October 26, 2005
 Cautionary Statement We will make various remarks during this presentation that constitute “forward-looking statements” for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995, including statements regarding the focus of the Company on urology and sexual health; future Testim market share, prescriptions and sales growth and factors that may drive such growth; size and growth potential of the androgen market and the gel segment thereof and factors that may drive such growth; the effect the Company’ s co-promotion partnership will have on future Testim sales and gross profit; probability of the Company’ s prevailing in pending patent litigation and the timing of court decisions in these actions; the Company’s products in development for treatment of Peyronie’s and Dupuytren’ s Diseases; the Company’s products in development for androgen replacement, overactive bladder and pain using a transmucosal film delivery system; size, growth potential and competition within certain markets relevant to the Company’ s product candidates; market expansion opportunities from diabetic patients; interpretation of clinical results, including the efficacy and tolerability of the Company’ s product candidates as well as the reduction of side effects; the timing of the commencement and completion of clinical trials; the timing of manufacturing scale up for the Company’s product candidates; the timing of permission to enter Phase III trials for the Company’ s AA4500 product candidate; competitive developments affecting the Company’s products and product candidates; the success of the Company’s development activities; and the Company’ s development and operation goals for fiscal 2005, 2006 or 2007. All remarks other than statements of historical facts made during this presentation, including but not limited to, statements regarding future expectations, plans and prospects for the Company, statements regarding forward- looking financial information and other statements containing the words “believe,” “may,” “could,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” and similar expressions, as they relate to the Company, constitute forward- looking statements. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in the Company’s Quarterly Report on Form 10- Q for the quarterly period ended June 30, 2005 under the heading “Factors That May Affect Our Future Results” , which is on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “Investor Relations - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward- looking statements. Given these risks and uncertainties, any or all of these forward- looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’ s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward- looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 Auxilium Pharmaceuticals • Specialty pharma company currently focused on urology and sexual health • Testim ® – for the treatment of testosterone replacement in hypogonadal men – has grown rapidly since 2003 launch • Established sales and marketing organization • Pipeline of product candidates aimed at unmet medical needs |
 Testosterone Replacement Therapy |
 Signs and Symptoms of Low Testosterone Brain Function Libido and erections Energy and vigor Irritability and depressed mood Cognitive function Sleep quality Body Composition Fat mass – abdominal obesity Muscle mass and strength Bone mineral density Male hairs and skin thickness |
 …within disease state 8% 12% 11% 20% 7% 20% 49% 70% 40-49 50-59 60-69 70-79 Prevalence of Low Testosterone Increases… …with age Total testosterone Free testosterone Age by Decade 20% 33% 30% 50% Erectile Dysfunction Type-2 Diabetes HIV (men) AIDS Source: Bodie J. et al, Dhinda S. et al, Dobbs, FDA Website |
 Gel Segment Continues to Outpace Androgen Market TRx Growth 2005 over 2004 Note: Total dollar value androgen market and gel segment increased 8.1% & 12.3%, respectively at YTD June 2005. Source: IMS, NPA & NSP data. -2.4% 1.3% 2.5% 1.3% 5.7% 7.3% Q1 Q2 Q3 Androgen TRT Market Gel Segment |
 Testim ® |
 Testim ® - a Leader in the Market • Proprietary, topical 1% testosterone gel – once-a-day application – self-marketed in U.S. • Launched in 2003 – 120 sales and marketing professionals – >26,000 script units in September 2005 (1) • Favorable clinical and commercial profile – comparative studies support advantages – cost-effective and convenient – broad prescription coverage – growing prescriber loyalty (1) Source: IMS Data |
              $2,893 $4,307 $5,634 $6,619 $7,157 $7,615 $8,797 $9,839 Q303 Q403 Q104 Q204 Q304 Q404 Q105 Q205 Quarterly Net Revenue Testim quarterly net revenue ($ in thousands) Growth Q2 2005 vs 2004: 49% Growth Q2 2005 vs Q1: 12% Growth Q2 2005 vs 2004: 49% Growth Q2 2005 vs Q1: 12% |
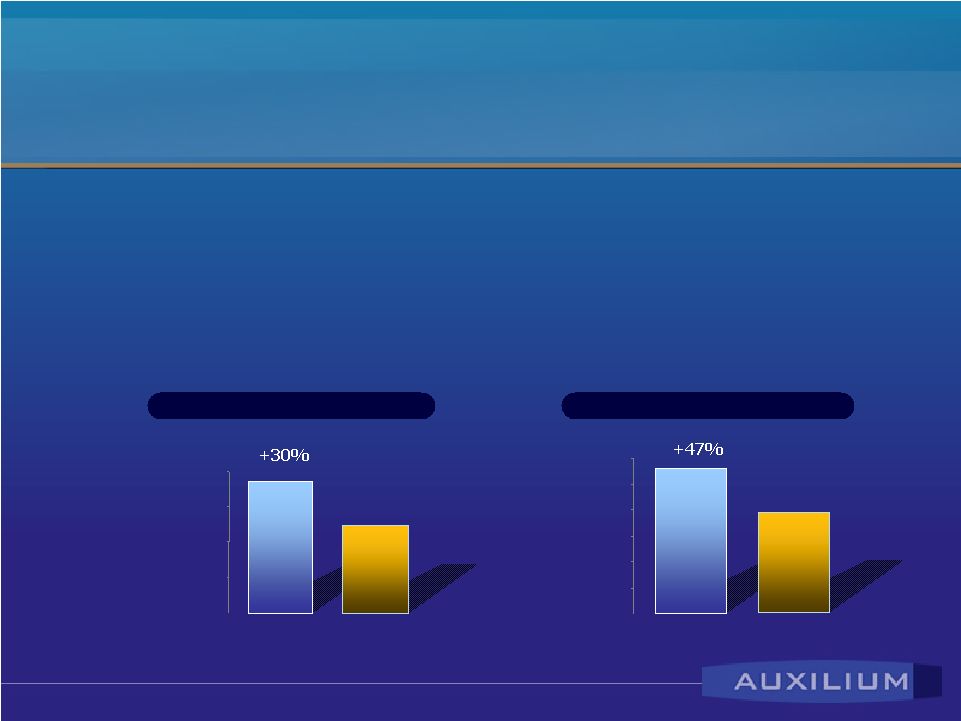       Note: Adjusted geometric means (CV b %) of a single-dose (50 mg testosterone), randomized, complete crossover study of 29 hypogonadal men. total testosterone 2,000 3,000 4,000 5,000 6,000 Mean AUC 0-24 (ng*h/dL) Testim AndroGel free testosterone 0 50 100 150 200 250 Mean AUC 0-24 (ng*h/dL) Testim AndroGel Data Demonstrates Testim ® ’s Advantages • 13 clinical studies involving 1,600 patients – largest placebo-controlled study ever conducted • Clinical trial vs. AndroGel – 30% higher testosterone absorption (p<0.001) |
 Sexual Activity Improved Source: Auxilium studies published in Journal of Clinical Metabolism & Review of Urology In placebo-controlled study, 90-day results included: • 59% increase in sexual activity • Statistically significant increases in sexual desire |
 12-month Study Reveals Lasting Results Changes in body composition: Lean Body Mass +4.8 lbs Fat Mass -4.0 lbs Bone Mineral Density +2.6 % Source: Auxilium studies published in Journal of Clinical Metabolism & Review of Urology |
 Specialized Sales Focus • 94 territories, 10 regions: covers 85% of specialty prescribers • Targeting urologists, endocrinologists and select primary care physicians • Higher frequency of calls among the most important prescribers – pyramid of influence • Share levels attained highest among the most important prescribers |
 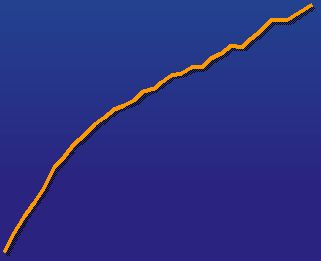   Testim ® share of TRx of gel market Testim ® is The Growth Story Strong market share with high-prescribing urologists (27% gel market share in August 2005) 1.0% 14.3% Mar-03 Aug-05 0% 2% 4% 6% 8% 10% 12% 14% Absolute Growth in Scripts YTD Aug 2005 vs 2004 Testim AndroGel Source: IMS data |
 Testim ® Co-Promote Adds SOV • Co-promotion agreement with Oscient began in May 2005 • Addition of 300 Oscient representatives improves “Share of Promotional Voice” for Testim ® , bringing total representatives to almost 400 |
 Testim ® Co-Promotion Partnership • Oscient to promote Testim ® to primary care physicians • Auxilium to promote Testim ® to specialists, select PCPs • Companies share out-of-pocket marketing costs • Two year agreement, with possibility to extend for six years pending OSCI achievement of milestones |
                   How the Profit Split Works gross profit sales PCP Gross Profit Gross Profit to Cover Marketing Costs Below Threshold Gross Profit Specialist Gross Profit Split based on sales being achieved Split 50/50 (except 2005: 65/35 OSCI) Retained 100% by Auxilium (illustrative only) PCP Sales PCP Sales to Cover Marketing Costs PCP Sales below Threshold Specialist Sales |
 Phase IV to Support Growth • Core market support – Testim ® co-administration with PDE-5 inhibitors • Market expansion – Diabetes: first study • 40% of diabetic men have low testosterone • Study impact on blood sugar control |
 market drivers product drivers Testim ® Growth Strategy • Male sexual health awareness • ED promotional impact • Increased screening and testing – normal “T” enhances outcome in ED patients – other health parameters improved with normal “T” • Increase awareness through thought leaders • Leverage clinical data • Improve sales force efficiencies • Modest sales force expansion • Leverage third-party payer coverage • Line extension |
 Gel Market Dynamics • Two gels on the market, including Testim ® – limited competition in development • Current Testim ® patent (exp. 2008) – new formulation patent filed in 4/03 (exp. 2020) • Testim ® inequivalent to AndroGel by virtue of greater absorption – AndroGel Paragraph IV challenge: Q1 ’06 – Opportunity to prevail via patent actions and court – Testim ® not substitutable • Impact of transmucosal film product candidates |
 Testim ® - Summary We believe: • Testim ® - fastest growing androgen gel • Data demonstrates Testim’s ® advantages • Specialized sales focus • OSCIENT co-promote: 300 additional reps! • Phase IV to support growth • Gel market dynamics – gel therapy is fastest-growing segment of TRT market – Under-treated market with growth potential • Rapid script units growth; >26,000 script units in September 2005 Source: IMS data |
 Pipeline |
 Pipeline Overview • We believe: – AA4500 franchise offers “first mover” options for unmet medical needs – Transmucosal film technology provides dosage formulations for a variety of product candidates – Robust pipeline includes focused projects for short- term and additional candidates for mid and long-term |
 AA4500 - A Transforming Opportunity • We believe this is a high potential product: – First mover value – No effective non-surgical treatments available – Orphan drug status granted • Currently licensed for two indications; options for licensing other indications |
 AA4500 – An injectible enzyme that breaks down plaque TBD Evaluation Other Phase III: commence 1st half of year Phase II complete Dupuytren's Disease Phase IIb: 1st half Phase III: commence Q4/Q1 2007 Phase IIa complete Peyronie’s Disease 2006 Goals Current Status Indication |
 Peyronie’s Disease • Affects men predominantly over 50 • Plaque, or hard scar area on the penis • Pain during erection and distortion of the penis • Treated by urologists • No good treatment options – PABA, Vitamin E, Verapamil ineffective – surgical treatment – may shorten the penis; cause scarring Source: Mulhall, J. et al., June 2004 |
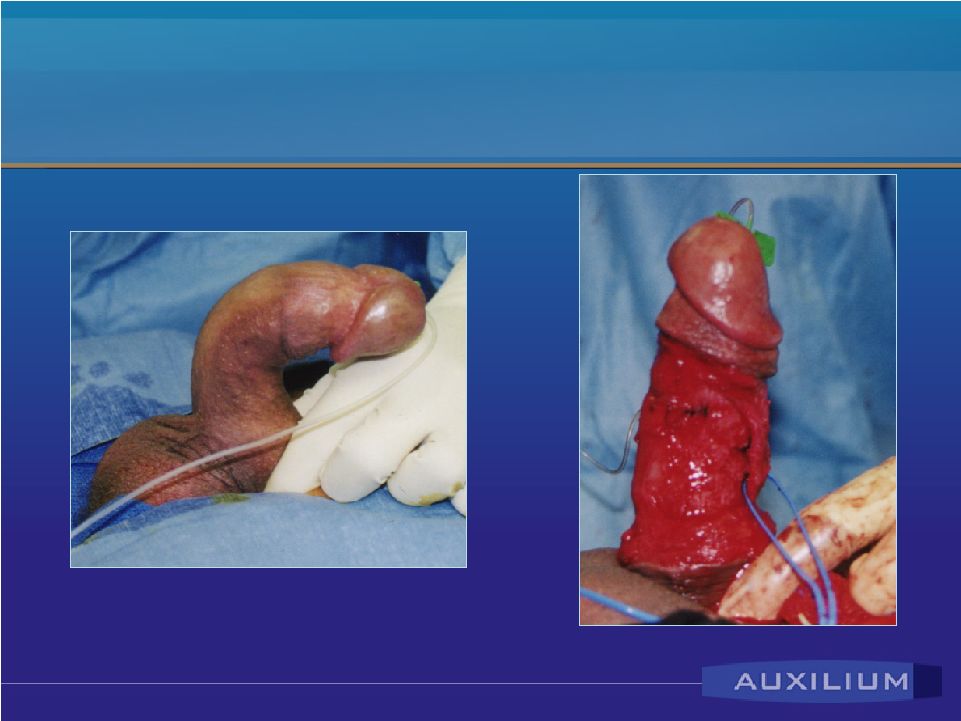 Peyronie’s Disease & Current Treatment Modality Before Surgery Surgery Confidential |
 Dupuytren’s Disease • Affects Caucasian population – prevalence higher among people of northern European descent • Ring and small fingers become permanently flexed • Surgery is the only approved treatment; expensive and characterized by numerous complications • Early Phase II data showed efficacy and tolerability |
 Reported Dupuytren’s Results • Mean degree of contracture: – MP Joints – 50 degrees – PIP Joints – 49 degrees • Ultimate patient outcomes – success = zero to five degrees - (mean of 3 injections): – MP Joints 90% success – PIP Joints 70% success Source: Badalamente, M. et al. J. Hand Surgery, September 2002 |
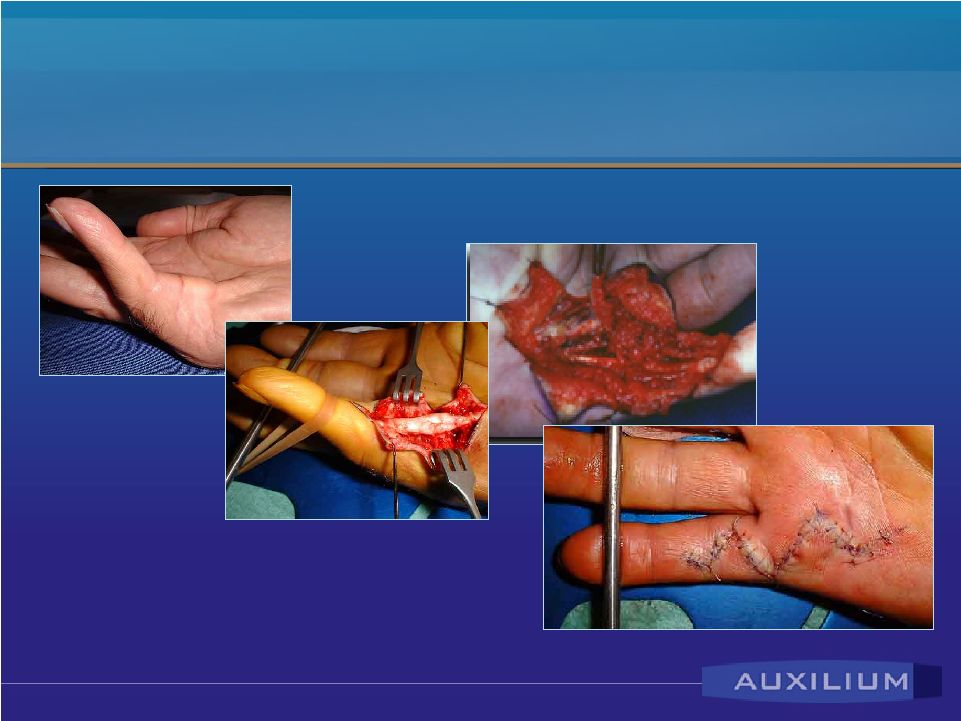 Confidential Dupuytren’s Disease & Current Treatment Modality |
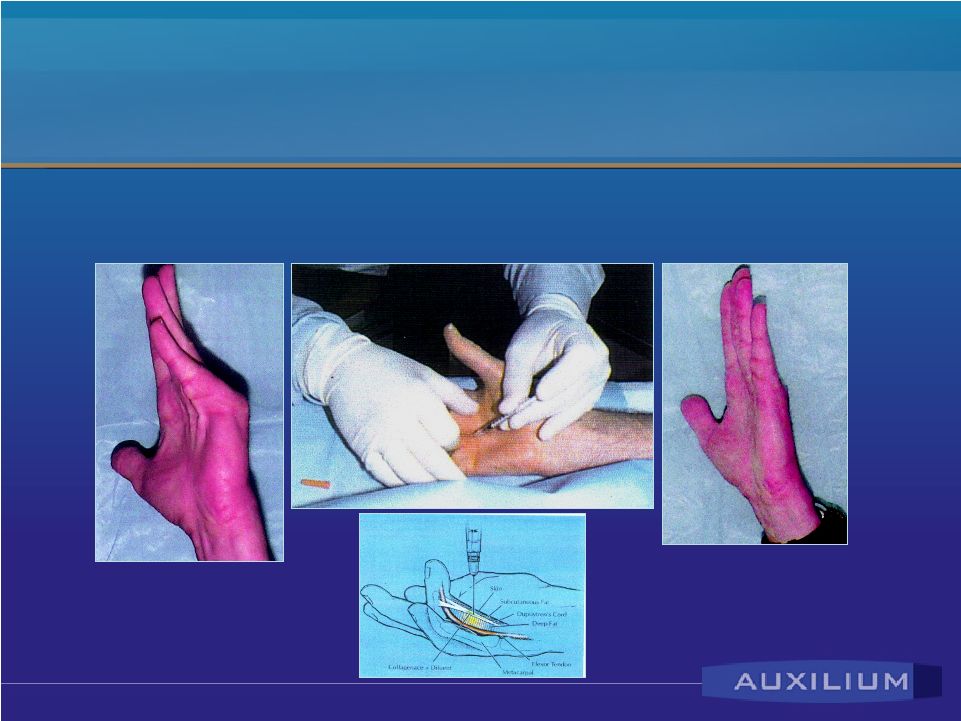 AA4500 For Dupuytren’s Disease Confidential |
 AA4500: Next Steps • Complete Manufacturing Scale-up • Request permission to enter Phase III for Dupytren’s Disease Trial • Then pursue Phase IIB – Peyronie’s Disease Study to optimize dosing |
 Transmucosal Film Technology --- Under evaluation Other Urologics --- Under evaluation Other Hormones Complete prototype formulation for two candidates Formulation Pain Products TBD Phase I complete Overactive Bladder Phase III: commence 1st quarter of year Phase II complete Hypogonadism 2006 Expectations Current Status Indication |
 Testosterone Replacement Film • Transmucosal film treatment for hypogonadism • Testim ® line extension with lower COGS • For patients who require: – more androgen than topical products – alternative dosage form • Phase II POC for Testosterone Replacement Transmucosal Film complete. Data demonstrated: – Increased serum testosterone levels – Product candidate was well tolerated – 97% patient acceptability: 49% rating film as desirable; 48% with acceptable rating |
 Overactive Bladder Film • OAB affects approximately 13 million people in the U.S.¹ – underreported by affected population – Est. (according to IMS) >$1 billion market – AA4010 API - oxybutynin • Goal: Equal or improved efficacy with fewer side effects • Phase I study completed Q2 2005 confirmed oxybutinin can be delivered via transmucosal film • Existing therapeutics’ side effects include: – dry mouth – gastrointestinal disorders – visual disturbances (1) Source: Goldenberg, M., 1999 |
 Corporate Partners • Testim ® marketing partners – Oscient – Co-Promote in U.S. – Bayer – Canada – Ipsen – worldwide outside U.S., Canada, Mexico and Japan • Drug delivery – Bentley Pharmaceuticals – transdermal gel – PharmaForm – transmucosal film • Contract manufacturers – Manufacture of development stage and commercial products |
 Financials |
 Summary Income Statement Operating expenses: 8,148 3,762 122 Cost of goods sold 15,993 7,175 14,130 Research & development 197,200 68,300 Dispensed Units (28,518) (28,875) (19,544) Net Loss (28,326) (29,590) (19,878) Operating Loss 55,351 38,412 19,878 Total Operating Expenses 31,210 27,475 5,626 S,G&A $ 27,025 $ 8,822 $ -- Revenue 2004 2003 2002 Year Ended December 31, ($ in thousands) Six Months 5,977 13,327 130,000 (20,141) (20,381) 39,017 19,713 $ 18,636 Ended 6/30/05 |
 Select Balance Sheet Information 2Q 2005 67,637 $ 86,186 18,527 11,486 50,012 as of Dec 31, 2004 Cash and cash equivalents and S/T Inv. 46,807 $ Total assets 61,040 Current liabilities 15,973 Long-term liabilities 8,823 Accumulated deficit (99,257) Total stockholders' equity 36,244 ($ in thousands) (119,398) 6,161 Financing-related liabilities - |
 Status of Goals for 2005 • Co-Promotion Partnership for Testim ® : Underway in Q2 • Increase Testim ® market penetration : Underway • Further develop urologic and sexual health products : Underway • In-license additional products : Evaluating Opportunities |
 |